Center for Molecular Medicine. University of Cologne
|
|
|
- Phillip Willis
- 10 years ago
- Views:
Transcription
1 Center for Molecular Medicine University of Cologne
2 Center for Molecular Medicine University of Cologne (CMMC) Scientific Report covering the research activities of the CMMC from January 2005 until May 2007 The research activities of the CMMC were made possible through funding by the State NRW.
3 Imprint Editor: Editorial Office: Layout / Design: Prof. Dr. Martin Krönke Center for Molecular Medicine University of Cologne Dr. Debora Grosskopf-Kroiher Susanne Haase / Ute Fiehne-Reiß Cornelius Evers Printing: Print Office of the University Hospital of Cologne Date of Print: June 2007
4 Table of Contents Introduction 1 External Scientific Advisory Board 10 Board of Trustees 11 Internal Reviewing Board 12 Executive Board 13 Presentation of the Research Groups 15 Research Area A Cardiac and Vascular Disorders A 1 A 2 A 3 A 4 A 5 A 6 A 7 A 8 NG V Molecular Mechanisms of Insulin Action and Insulin Resistance Jens C. Brüning 1 / Wilhelm Krone 2 (Institute for Genetics 1 / Dept. II of Internal Medicine 2 ) Aspects of the Submembraneous Region: The Role of Actin-Associated Proteins and their Role in Human Disease Angelika A. Noegel (Institute for Biochemistry I) Molecular Dissection of the Hyperpolarization-Activated Inward Current I f Uta C. Hoppe / Erland Erdmann (Department III of Internal Medicine) Endothelial Cell Damage and Repair in the Retina and Choroid Antonia M. Joussen / Sascha Fauser / Bernd Kirchhof (Department of Ophtalmology) Role of β-subunit Genes and Splice Variants for Pathological Gating of Human Cardiac L-Type Calcium Channels in Heart Failure Stefan Herzig 1 / Uta C. Hoppe 2 (Institute for Pharmacology 1 / Dept. III of Internal Medicine 2 ) Characterization of Atherogenic Signaling Pathways by the βpdgf Receptor In Vivo Stephan Rosenkranz (Department III of Internal Medicine) Commitment of the Extraneuronal Monoamine Transporter (EMT) in Cardiovascular Inactivation of Noradrenaline Dirk Gründemann / Edgar Schömig (Institute for Pharmacology) Switch-on and -off Kinetics of the Troponin (Tn) in Cardiac Myofibrils and the Effect of Familial Cardiomyopythy-Linked Mutations of Tn on these Kinetics Robert Stehle / Gabriele Pfitzer (Institute for Vegetative Physiology) Molecular Pathogenesis of Arrhythmogenic Diseases Bernd Wollnik (CMMC Junior Research Group, hosted by the Institute for Human Genetics) Research Area B Host Response to Tumor Growth, Inflammation and Infection B 1 B 2 Characterization of Melanoma-Stroma Interactions Cornelia Mauch / Thomas Krieg (Department of Dermatology and Venerology) The Consequences of Mutations of Genes that Contribute to Basement Membrane Function Neil Smyth / Mats Paulsson (Institute for Biochemistry II) 34 36
5 Table of Contents B 3 B 4 B 5 B 6 B 7 B 8 B 9 B 10 B 11 B 12 NG I NG IV NG VI NG-F Liaison- Group Role of Papillomaviruses in Non-Melanoma Skin Cancer: Viral Oncoproteins, UV-Light as Cofactor Herbert J. Pfister (Institute for Virology) Modular Composition of Recombinant Immunoreceptors Hinrich Abken (Department I of Internal Medicine) The Role of NFκB Dependent Epidermal-Dermal Interactions in the Pathogenesis of Inflammatory Skin Disease Ingo Haase (Department of Dermatology and Venerology) Function of α3β1 and α6β1 Integrins and their Binding Proteins FHL2 and GIPC in Skin Biology and Pathology Monique Aumailley (Institute for Biochemistry II) RhoBTB Proteins: Role in Oncogenesis Francisco J. Rivero Crespo (Institute for Biochemistry I) Resistance to Apoptosis in Hodgkin s Lymphoma: From Molecular Mechanisms to Therapeutical Approaches Hamid Kashkar / Martin Krönke (Inst. for Medical Microbiology, Immunology and Hygiene) Search for Low Penetrance Genes in Hereditary Breast Cancer Rita Schmutzler / Barbara Wappenschmidt (Department of Gynaecology and Obstetrics) Optimization of AAV Vectors for Gene Therapy of Solid Tumors Hildegard Büning / Ulrich Hacker / Michael Hallek (Department I of Internal Medicine) Molecular Genetics of Alopecia Areata Hans Christian Hennies / Peter Nürnberg (Cologne Center for Genomics) Function and Regulation of Lipid Droplets Stefan Höning (Institute for Biochemistry I) Cell-Cell Interactions and Polarity in Development and Tumorigenesis Carien Niessen (Junior Research Group I, hosted by the Department II of Internal Medicine, Department of Dermatology and Department of Surgery) Regulation of Lineage Commitment in Mammalian Skin Catherin Niemann (Junior Research Group IV, hosted by the Institute for Pathology) Function and Regulation of PML Nuclear Bodies Gerrit Praefcke (Junior Research Group VI, hosted by the Institute for Genetics) Hepatitis B Virus-Host Interaction Ulrike Protzer (Junior Research Group of the Medical Faculty, hosted by the Institute for Med. Microbiology, Immunology and Hygiene) A Comprehensive Clinical and Molecular Approach to a Pediatric Model Tumor Frank Berthold (Department of Pediatric Oncology and Hematology) Research Area C Molecular Neurobiology C 1 The Role of Neuronal Insulin Resistance in Neurodegenerative Diseases Markus Schubert 1 / Jens Brüning 2 / Wilhelm Krone 1 (Dept. II of Internal Medicine 1, Institute for Genetics 2 ) 68
6 Table of Contents Table of Contents C3 C 4 C 5 C 6 C 7 C 8 C 9 C 10 C 11 Molecular Pathology Induced by Targeted Modifications of the Lipid Bilayer of Cell Membranes in Mouse Mutant Models Wilhelm Stoffel (Laboratory of Molecular Neuroscience) Molecular Mechanisms of Rhythm Generation and Synchronization in Electrically Coupled Cells Structure & Function of Voltage-Gated Ca 2+ Channels in Cardiac and Neuronal Pacemaker Centers and Related Neuronal Circuits, and their Role for Spontaneous Excitability and Memory Formation Toni Schneider / Jürgen Hescheler (Institute for Neurophysiology) Gene Therapy of Brain Tumors Andreas H. Jacobs / Wolf-Dieter Heiss (Department of Neurology) Role of an Impaired Protein Quality Control in Mitochondria for the Pathogenesis of Spastic Paraplegia Thomas Langer (Institute for Genetics) Functional Role of PACSIN Proteins in the Regulation of Glutamate Receptors in Healthy and Degenerating Neurons Markus Plomann (Institute for Biochemistry II) In-Depth Analysis of Axonal Guidance by Netrins Manuel Koch (Institute for Biochemistry II) Molecular Pathomechanisms of a Skeletal-Muscle-Specific Lethal Mutation of Mitochondrial DNA in a Muscle Specific Cybrid Model Rudolf J. Wiesner 1 / Jürgen-Christoph von Kleist-Retzow 2 (Institute for Vegetative Physiology 1 / Department of Pediatrics 2 ) Spinal Muscular Atrophy (SMA): Drug Validation for Therapy and Identification of Disease Modifying Genes Brunhilde Wirth / Eric Hahnen (Institute for Human Genetics) Comprehensive Genetic Analysis of the Dopaminergic and Serotonergic Systems in Migrane with Aura Christian Kubisch (Institute for Human Genetics) Core Units CBA AFN Central Bioanalytics Franz-Georg Hanisch Animal Facility Network Bettina Wagner Publications 92 Patents 111 Career Development 112 Seminars & Meetings 126 Addresses 138
7 Introduction The Center for Molecular Medicine at the University of Cologne (CMMC) From Basic Molecular Research to Clinical Application The Center for Molecular Medicine at the University of Cologne (CMMC) offers a forum for competitive basic, disease-oriented research for scientists from the Faculty of Medicine and the Faculty of Mathematics and Natural Sciences. In their biomedical projects the scientists of the CMMC investigate at the molecular level the pathogenetic mechanisms of cardiovascular disorders, inflammatory and infectious diseases, cancer and neurological disorders. This innovative concept of bringing together the expertise of both basic and clinical scientists supports the development of novel and effective diagnostic as well as therapeutic interventions. The Center for Molecular Medicine at the University of Cologne (CMMC) was established in June 1994 as a central interdisciplinary institution of the University of Cologne. The CMMC supports state-of-the-art medically orientated as well as translational research in molecular life sciences in order to gain insight into pathogenic mechanisms of diseases and to develop new and more efficient therapeutic modalities. The CMMC supports individual research projects focused within the following three biomedical research areas: A. Cardiac and Vascular Disorders B. Host Response to Tumor Growth, Inflammation and Infection C. Molecular Neurobiology Apart from advancing the knowledge of the pathogenesis of diseases at the molecular level, another major facet of the CMMC is the promotion of young talented scientists. Furthermore, the CMMC holds core facilities that provide technical platforms, e.g. the laboratory for protein bioanalytics and support services, e.g. the generation and breeding of transgenic animals. Scientific Research At present, the CMMC supports a total of 37 research projects including four Junior Research Groups and - as an associated group - the Junior Research Group of the Faculty of Medicine co-opted by the CMMC. In 2004, the center integrated the neuroblastoma research group as a liaison group. The many interactions among the groups within the Center have led to the transfer of methods and increasingly to the setting up of collaborative research projects including the establishment of animal disease models. The generation and analysis of such transgenic model systems is an essential component of the research within the CMMC. Recently, the translational research in cancer was strengthened by providing a position for the coordination of a clinical phase I/II study for analyzing a therapeutical concept of antigen-specific targeting of T-cells for treating a particular type of cutaneous lymphoma. The current research activities and scientific achievements of the CMMC are presented in the progress reports of the individual research groups which is complemented by the list of original articles published. As a major highlight, in 2006 Prof. Brüning was awarded the Gottfried Wilhelm Leibniz Prize from the Deutsche Forschungsgemeinschaft (DFG) for his outstanding research into better defining the underlying molecular mechanisms of diabetes mellitus.
8 Introduction A. Cardiac and Vascular Disorders In this research area the molecular mechanisms of cardiovascular risk factors such as hypercholesterinemia, insulin resistance and diabetes mellitus are experimentally addressed. The overriding aim is to identify novel strategies both for the development of diagnostics and therapies of these diseases. This research area is traditionally based on close cooperation and interchange between the Departments of Internal Medicine II and III, the Center of Ophthalmology, the Institute of Pharmacology, the Institute for Biochemistry I, the Institute for Physiology, the Institute for Human Genetics and the Institute for Genetics, and is concentrated on two major research areas: Molecular mechanisms in the pathogenesis and complications of cardiovascular disease and Growth and functions of cardiac muscle cells, vascular smooth muscle cells and endothelial cells. The individual projects aim at identifying the clinical and pathophysiological relevance of signaling mechanisms in disease. They also provide insights into the basic, cellbiological mechanisms of signal transduction, which involve growth factor receptors, ion channels and transcription factors. B. Host Response to Tumor Growth, Inflammation and Infection Within this research area, the CMMC has placed special onus on investigating the biology of, and immunity to tumors, mechanisms of apoptosis deficiency, and cellcell interactions. Since the human defense mechanisms against tumors of viral etiology resemble in many ways immunity to infection, general aspects of inflammation biology including therapeutic gene transfer using viral vectors, as well as cytokine signaling, cell adhesion, and angiogenesis are major research foci. Recently, the neuroblastoma research group led by Prof. Frank Berthold (Department of Pediatric Oncology and Hematology, University Medical Center) was integrated as a liaison research group into this research area, thereby reinforcing the CMMC s commitment in advancing understanding in the field of oncology. The aim of this liaison group is to unravel the molecular pathogenesis of neuroblastoma, the most frequent malignant tumor in children and adolescents. The insights gained are expected to be instrumental developing novel diagnostic tools and therapeutic modalities such as humanized, bispecific monoclonal antibodies and T cell-based vaccines. Finally, this liaison research group has forged close ties with members of the CMMC, which strengthened the oncological research network associated with the recently established Center of Integrated Oncology (CIO). C. Molecular Neurobiology The molecular biology and pathology of the central and peripheral nervous system has attracted major attention within the CMMC. Researchers investigate the molecular pathogenesis of neurodegenerative diseases like migraine with aura, Alzheimer s and Huntingtons disease as well as hereditary forms of dysmyelinoses and spastic paraplegia. Channelopathies causing epilepsy through disturbances of thalamocortical-cortico-thalamic neuronal circuits are are also being studied. Moreover, the development of gene therapy strategies is being pursued using in an experimental glioma model. By combining of gene-and cell-based therapies with non-invasive molecular imaging technology, strategies will be developed to improve conventional therapies of malignant brain tumors with high incidence yet poor prognosis. Finally, drug evaluation for the identification and therapy of disease-modifying genes are pursued in spinal muscular atrophy. Structure and Organization of the CMMC All of the CMMC s individual research projects are grouped within these three CMMC core research areas as are the junior investigator groups. At present, the principal investigator scientists are affiliated with eight clinical departments and nine pre-clinical institutes of the Faculty of Medicine, and the Institute for Genetics of the Faculty of Mathematics and Natural Sciences. Four core units provide the structural and administrative research infrastructure of the CMMC. The services of the Scientific Management Office, the Bioanalytical Laboratory, the Animal Facility Network as well as the Graphics and Art Unit are available to all CMMC scientists. The financial administration of the Center is handled by the unit for external funding at the Medical Center of the Uni-
9 Introduction University of Cologne Rectorate / Senate Faculty of Medicine and Faculty of Mathematics & Natural Sciences External Scientific Advisory Board Center for Molecular Medicine (CMMC) Executive Board Board of Trustees Internal Reviewing Board Board of Principal Investigators Board of CMMC-Members Research Area A Cardiac and Vascular Disorders Research Area B Host Response to Tumor Growth, Inflammation and Infection Research Area C Molecular Neurobiology 8 Projects 12 Projects 11 Projects 1 Junior Research Group (NG) 3 Junior Research Groups (NG) 1 Associated Junior Research Group Liaison Group Neuroblastoma Core Units Figure 1: Structure and Organization of the CMMC
10 Introduction versity of Cologne. The expert services of the core units of the CMMC are highly appreciated by the members of the CMMC and are instrumental in advancing for their high-quality research. The activities of the Management Boards and the Reviewing Boards guarantee the effectiveness of the CMMC. The Executive Board, the Board of Principal Investigators and the Board of Members are defined as Management Boards of the CMMC. The Reviewing Boards include the External Scientific Advisory Board, the Board of Trustees and the Internal Reviewing Board. The Executive Board, consisting of the chairman, two vice chairmen, four scientific coordinators and the deans of both faculties, is responsible for and takes action with regard to scientific as well as administrative matters and represents the interests of the CMMC in all external affairs. The Executive Board shares with the Board of Principal Investigators the responsibility for the scientific direction of the CMMC. The Board of Principal Investigators consists of all principal investigators of CMMC-funded projects, the deans of both faculties, and in a consulting function, two elected representatives each of academic and nonacademic staff as well as students. Finally, every person working in the CMMC belongs to the Board of Members. Funding of the CMMC From 1995 until 2003, the research activities and infrastructure core units were financially supported by the program Health Research 2000: Interdisciplinary Center of Clinical Research of the Federal Ministry of Education and Research and by the State of North Rhine Westphalia. During this time approximately 40 million flowed into the CMMC. As the federal support gradually decreased from 70% to 0%, the State of North Rhine Westphalia increased the funding from 30% to 100%. Since 2004, the CMMC is extensively funded by the State of North Rhine Westphalia with an overall budget of approximately 4.65 million per year. Reviewing Structures at the CMMC The External Scientific Advisory Board continuously assesses the scientific performance of the CMMC and has executive power over the funding of individual research groups, the establishment of junior research groups as well as major structural changes within the CMMC. The Board of Trustees advises the CMMC in all matters concerning its integration into the Cologne community and the local region. Consistent with the Universityembedded status of the CMMC, the members of the External Scientific Advisory Board and the Board of Trustees are appointed by the rector of the University of Cologne. In addition the Center maintains an Internal Reviewing Board that closely follows and evaluates all research projects. The development and maintenance of a stringent internal and external evaluation procedure performed by the External Scientific Advisory Board and the Internal Reviewing Board have been instrumental in setting high standards for CMMC c research activities. Support of Young and Junior Scientists The continuous support of young and junior scientists is a major CMMC commitment and involves a wide spectrum of sponsoring activities: Implementation of four Junior Research Groups Implementation of Rotation Positions Gain-of-brain Stipends Interdisciplinary Training Establishment of an Interdisciplinary Postgraduate Program in Molecular Medicine The implementation of junior research groups (NG) is an integral part of the junior career program of the CMMC. Excellent young scientists are selected and supported over a period of five years including a substantial running budget. In addition to their own salary they are supported with three additional positions (postdoctoral position, PhD student, technical assistant) and adequate laboratory facilities. In this span of time the junior researchers are expected to establish their scientific reputation within the scientific community and to qualify for academic faculty positions. The CMMC currently supports four junior research groups. In addition, a junior research group of the Faculty of Medicine has been co-opted as a full member of the CMMC and is thereby integrated into all scientific
11 Introduction and infrastructural activities of the CMMC. The junior research groups are allocated to the CMMC s research area that best matches their respective scientific expertise and are accommodated in the clinical departments or institutes that most closely relate to their respective research interests, a concept that fosters scientific interactions. NG I NG IV NG V NG VI NG-F Carien Niessen (hosted by the Dept. II of Internal Medicine, the Dept. of Dermatology and the Dept. of Surgery) Catherin Niemann (hosted by the Department of Pathology) Bernd Wollnik (hosted by the Institute for Human Genetics) Gerrit Praefcke (hosted by the Institute for Genetics) Ulrike Protzer (associated group of the Faculty of Medicine hosted by the Institute for Medical Microbiology, Immunology and Hygiene) To ensure that young physician-scientists have enough time to devote to their research endeavors, the CMMC provides five to six rotation positions for physicians who take over the clinical tasks and responsibilities of the researchers, thereby freeing the young physicianscientists from their daily clinical duties. The rotation positions are granted for six to twelve months and need to be re-applied for on a yearly basis. The CMMC also provides post-doctoral positions for young scientists working abroad who plan to continue their scientific career in a CMMC project when returning to Germany ( gain-of-brain stipends). After positive evaluation of the research proposal the position is granted for 12 months and provides an optimal basis for re-integrating postdoctoral scientists into the German science community after their temporary scientific employment abroad. Scientific Training Postdoctoral, PhD, and diploma research positions are offered within the CMMC along with structured research training in the form of courses and seminars. From January 2005 until the end of March 2007, 10 CMMC scientists completed their Habilitation, 39 were awarded the academic degree Dr. rer. nat., 5 the degree Dr. nat. med. (equivalent to a Dr. rer. nat.) and 30 the degree Dr. med. In addition, 56 students completed their diploma research work in CMMC groups. The gender distribution of young and junior scientists having completed or who are still enrolled in the different academic programs is summarized in table 1. As a consequence of the interfaculty nature of the CMMC, Academic Levels Completed Jan March 2007 On-going March 2007 total male female (%) total male female (%) Habilitation (10%) (17%) PhD Thesis Dr. rer. nat. / Dr. nat. med. Medical Dissertation Dr. med. Diploma Thesis (Master) Diploma / Master (48%) (53%) (43%) (42%) (46%) (70%) Table 1: Gender distribution of junior scientists at the different academic levels
12 Introduction University of Cologne Faculty of Medicine Interdisciplinary Postgraduate Program Physicians (2 nd state examination or equivalent ) Experimental Research Work + Seminars, Lectures- & Training Courses (individually tailored) CMMC -Molecular Medicine- Running period of three years MD-PhD-Student Graduation and Award of the Academic Degree Dr. nat. med. Faculty of Mathematics & Natural Sciences Postgraduate Natural Scientists Evaluation of the Application through the MD-PhD - Committee consisting of Members of both Faculties involved Advised and Instructed by one Tutor of the two Faculties Involved Doktor der naturwissenschaftlichen Medizin Figure 2: Postgraduate Program Molecular Medicine the Faculty of Medicine and the Faculty of Mathematics and Natural Sciences established an interdisciplinary doctoral program Molecular Medicine (IPMM) with its own postgraduate curriculum as summarized in Fig. 2. The IPMM is aimed at young physicians (after their 2 nd state examination or equivalent) and postgraduate natural scientists and is designed to provide projectgeared training in the field of biomedical research to doctoral candidates from both faculties. Each candidate is advised and instructed by one tutor from each of the two faculties; these senior scientists help to develop and tailor an individual study program according to the preexisting knowledge and field of interest of the candidate. In addition to their research work, doctoral candidates are required to participate in individually tailored teaching courses of a minimum of four hours per week over four terms. The successful completion of the IPMM consists of a positively evaluated doctoral thesis in combination with the completed study program and a passed oral doctoral examination. Successful participants are awarded the academic grade of Doktor der naturwissenschaftlichen Medizin (Dr. nat. med.) being equivalent to the academic grade Dr. rer. nat. and the Anglo-saxon PhD degree. The first doctoral students started the program in the winter term of 2002/2003. The CMMC supported the initiation of the program by providing five internationally advertised doctoral fellowships to the best among 135 applicants. At present 30 doctoral students are enrolled. PhD students funded by project grants can join the program after an intensive evaluation by the program committee consisting of members of both faculties. So far, five postgraduate students have successfully completed the program. Due to its interfaculty character, the scientific management office of the CMMC coordinates the program. For further information, please visit the website: Conclusion and Further Perspectives Since its establishment in 1994, the CMMC has initiated innovative biomedical research programs and catalyzed numerous structural changes in both the Faculty of Medicine and the Faculty of Mathematics and Natural Sciences leading to closer cooperation between researchers and clinicians. The implication of the CMMC for the development of research structures is illustrated in Fig. 3. The CMMC aims at creating a broader network of research activities in Cologne, serving the purpose of improving the scientific output and the third party funding in the University. In the last two years the departments and the institutes with CMMC-research projects contributed
13 Introduction CIO CRCs Educational Programs PhD-Program Molecular Medicine CCG CMMC Aging MPI Faculty of Medicine Faculty of Math. & Nat. Sciences Neurology Figure 3: The implications of the CMMC for the development of research structures. CIO - Center for Integrated Oncology, CCG - Cologne Center for Genomics, MPI - Max Planck Institute, CRCs - Collaborative Research Centers (SFB, SPP, BMBF and EU Programs or Networks) with approximately 60 % to the overall sum of third party funding. The proportion of this contribution gradually increased since 1999 as shown in Fig. 4. CMMC members have also established three Collaborative Research Centers of the German Research Foundation ( DFG-Sonderforschungsbereiche ): SFB 589 Molecular basis of mechanical and functional barriers in the skin co-ordinated by Prof. Thomas Krieg (Vice Chairman of the CMMC) is running since January 2002 and the SFB 635 Post-translational control of protein function, coordinated by Prof. Thomas Langer (Vice Chairman of the CMMC) operates since July In July 2006, a third SFB 670 Cell-autonomous Immunity was granted by the Deutsche Forschungsgemeinschaft, co-ordinated by Prof. Martin Krönke (Chairman of the CMMC). The CMMC is actively involved in all SFB activities, which reflected by close scientific co-operations and shared use of the research infrastructure developed by the CMMC. In addition, two DFG Priority Programs ( Schwerpunktprogramme ) are co-ordinated by members of the CMMC: SPP Genetic and molecular analysis of basement membranes and basement anchorage (Prof. Mats Paulsson) and SPP Innate Immunity (Prof. Martin Krönke). In March 2007, the Center of Integrated Oncology (CIO) at the Medical Center headed by Prof. Michael Hallek, a CMMC member, was chosen by the German Cancer Research Aid ( Deutsche Krebshilfe ) as one of four Oncological Centers of Excellence in a nationwide competition. Transferring the innovations from cancer research into practice is one major goal of the CIO. An
14 Introduction Overall Volume of the Third Party Funding of the Medical Faculty (Million Euro) CMMC-associated Departments and Institutes Other Departments and Institutes Years Figure 4: Contribution of the CMMC to the third party funding activities of the Faculty of Medicine. overview of third party funded projects of the Faculty of Medicine indicating the involvement of members of the CMMC is shown in table 2. In 2002, the University of Cologne, the CMMC and the Institute for Genetics jointly established the Cologne Center for Genomics (CCG) headed by Prof. Peter Nürnberg. The objective of the CCG is to link genetic analysis of human disease to the use of animal model systems (e.g. mouse, zebra fish, dictyostelium, drosophila). Furthermore, the Institute for Human Genetics headed by Prof. Brunhilde Wirth was recently established at the Medical Faculty. The interfaculty postgraduate program Molecular Medicine is an additional achievement of the CMMC together with the two faculties. Beyond the borders of the Cologne Campus, the CMMC has participated in creating a biomedical research cluster, BioRiver, which brings together the scientific communities of the Universities of Aachen, Bonn, Cologne and Düsseldorf as well as the Helmholtz Research Center in Jülich. Finally, the Max Planck Society will build a new Max Planck Institute for Aging Research (Biology of Aging) thereby further strengthening and stimulating research activities at the biomedical campus of the University of Cologne. The new Research Building of the CMMC / CCG Since the foundation of the CMMC, research groups and central core facilities and support services have been spread over the various departments and institutes of the Faculty of Medicine. Therefore, the necessity for a new research building was already evident from the very first funding period. Moreover, over the past years with the expansion of the CMMC, the shortage of high quality laboratory space as well as support facilities, e.g. for animal keeping, has increasingly hampered the research activities of the CMMC. In 2002, the Ministry of Innovation, Science, Research and Technological Transfer of North Rhine Westphalia (NRW) decided to provide financial support for a new research building. The Spatenstich for the new research building was held in combination with the celebration of the 10th anniversary of the CMMC on December 19, On this occasion, Prof. Franz Hofmann, the chairman of the External Scientific Advisory Board of the CMMC, recalled the beginning of the Center and the instrumental roles played in its successful development by Prof. Wilhelm Stoffel, Prof. Thomas Krieg and Prof. Walther Rüssman, the former dean of the Medical Faculty during the status nascendi of the CMMC. The construction of the five-story building located on the medical campus of the University in close proximity to the University Medical Center, the departments and institutes of the Faculty of Medicine, and the future Max Planck Institute for Aging Research began in March 2006 and is due for completion in May When completed it will provide a total floor space of sqm to scientists of the CMMC and CCG. It will occupy sqm floor space predominantly holding state-of-the-art laboratories. In addition, a library and other common services will support the flow of scientific communication. As stated at the topping out ceremony held on May 9, 2007, the new building housing 160 scientists from the CMMC and the CCG will be important for the further development of life science research at the University of Cologne. M. Krönke
15 Introduction Table 2: Participation of the CMMC in Third Party Funded Programs and Integrated Projects - individual third party funding and projects funded by industry are not included - 1. DFG Collaborative Research Centers ( SFBs ) and DFG Research Units ( FG ) SFB 670 Cell-autonomous immunity, Univ. Köln SFB 589 Molecular basis of structural and functional barriers in the skin, Univ. Köln SFB 635 Posttranslational control of protein function, Univ. Köln Participation in SFBs at other Universities SFB 704 Molecular mechanisms and chemical modulation of local immunity - Univ. Bonn SFB 612 Molecular analysis of cardiovascular function and dysfunction - Univ. Düsseldorf SFB 577 Molecular basis of clinical variability in mendelian disorders - Univ. Berlin SFB 490 Invasion and persistence of infectious agents - Univ. Mainz Participation in DFG Research Units ( FG ) at other Universities FG 427 Epidemiology and genetic of complex diseases, Bonn FG 535 Xenotransplantation, München 2. DFG Priority Programs ( SPP ) SPP 1086 Genetic and molecular analysis of basement membranes / basement membrane anchorage SPP 1110 Innate Immunity SPP 1230 Mechanisms of gene vector entry and persistence 3. BMBF Competence Networks and Networks Ichthyosis & disorders of cornification Impetus Malignant Lymphomas MD-Net-Muscular Dystrophy NGfN Animal Modells 4. European Framework Program Projects and Networks Amis: Anti-microbial drug resistence Attack: T-cell mediated immunotherapy (Program Coordination) Clinigene: Clinical gene transfer and therapy Crystal: Cryo-banking of stem cells, therapeutic application DiMI: Diagnostic molecular imaging (Program Coordination) ELN: European Leukemia-Net Emil: Molecular imaging techniques in oncology Eurohead: Migraine genes and neurobiological pathway FunGenES: Functional genomics in embryonic stem cells (Program Coordination) GeneExpression GeneSkin: Rare genetic diseases of the skin Nest: New and emerging science and technologies Secretory Pathway 5. Educational Programs Interdisciplinary postgraduate program Molecular Medicine University Köln (Program Coordination) NRW-Graduate School for Genetics and Functional Genomics Faculty of Mathematics and Natural Sciences
16 External Scientific Advisory Board Current Members Prof. Dr. Thomas Braun Max-Planck-Institute for Heart and Lung Research W.G. Kerckhoff-Institute Bad Nauheim CMMC Member since 2002 Prof. Dr. Stefan H.E. Kaufmann Department of Immunology Max-Planck-Institute for Infection Biology Berlin CMMC Member since 1997 Prof. Dr. Helmut Drexler Department of Internal Medicine University of Hannover CMMC Member since 1997 Prof. Dr. Hans Konrad Müller-Hermelink Institute for Pathology University of Würzburg CMMC Member since 1997 Prof. Dr. Bernd Groner Georg Speyer Haus Institute for Biomedical Research Frankfurt am Main CMMC Member since 2002 Prof. Dr. Klaus-Armin Nave Department of Neurogenetics Max-Planck-Institute for Experimental Medicine Göttingen CMMC Member since 2004 Prof. Dr. Dr. Jürgen Heesemann Max von Pettenkofer-Institute for Hygiene and Medical Microbiology München CMMC Member since 2005 Prof. Dr. Christian Peschel III rd Department of Medicine Technical University München CMMC Member since 2004 Prof. Dr. Franz Hofmann Chairman Institute for Pharmacology and Toxicology Technical University, München CMMC Member since 1997 Prof. Dr. Dietmar Vestweber Max-Planck-Institute for Molecular Biomedicine Münster CMMC Member since 2002 Former Members Prof. Dr. Uta Francke Howard Hughes Medical Institute Stanford University Medical Center, USA CMMC Member Prof. Dr. Christian Haass Department of Biochemistry University of München CMMC Member Prof. Dr. Wieland B. Huttner Max-Planck-Institute of Molecular Cell Biology and Genetics Dresden CMMC Member Prof. Dr. Rolf Knippers Department of Biology University of Konstanz CMMC Member Prof. Dr. Peter Herrlich Research Unit Molecular Cell Biology University of Jena CMMC Member Prof. Dr. Ulrich Walter Institute for Biochemistry and Pathobiochemistry University of Würzburg CMMC Member Prof. Dr. Wolfgang Hiddemann Department of Internal Medicine III University Hospital Grosshadern University of München CMMC Member
17 Board of Trustees Extramural Members Prof. Dr. Klaus Bergdolt Director Institute for History and Ethics of Medicine University of Cologne CMMC Member since 2005 Prof. Dr. Herbert Jäckle Vice-President Max-Planck-Society, München Director Max-Planck-Institute for Biophysical Chemistry, Göttingen CMMC Member since 2005 Stefan Miltenyi Director Miltenyi Biotec GmbH Bergisch Gladbach CMMC Member since 2004 Prof. Alfred Neven DuMont Publisher Kölner Stadtanzeiger CMMC Member since 1997 Dr. Heinrich Pfeiffer Retired Secretary General Alexander von Humboldt-Foundation Bonn CMMC Member since 1997 Dr. Bernd Pulverer Editor Nature Cell Biology London CMMC Member since 2004 Gustav Adolf Schröder Chairman Board Member SK Stiftung Kultur, Cologne Board Member Ruhrkohle Aktiengesellschaft Foundation, Essen CMMC Member since 1997 Prof. Dr. Walter Tokarski Rector German Sport University Cologne CMMC Member since 2004 Former Members Hartmut Krebs former State Secretary Ministry for Science and Research of the State NRW Chairman of the CMMC-Board of Trustees CMMC Member Norbert Heinen former Chief Executive Officer and Vice President of Gerling Life Insurance CMMC Member Prof. Dr. Walter Rüssmann Professor Emeritus Department of Ophthalmology, University of Cologne former Dean of the Faculty of Medicine and Medical Director of the University Hospital Cologne CMMC Member Intramural Members Permanent members of the Board of Trustees representing the University are the Dean of the Faculty of Medicine and the Dean of the Faculty of Mathematics and Natural Sciences as well as the Chancellor; the members representing the CMMC are the Chairman and the Vice Chairmen as well as one Research Area Coordinator. 11
18 Internal Reviewing Board Members and their Substitutes elected by the Board of Principal Investigators Member Dr. Hildegard Büning Department I of Internal Medicine & CMMC Dr. Carien Niessen (NG I) hosted by: Department II of Internal Medicine, Department of Dermatology, Department of Surgery & CMMC Prof. Dr. Angelika Noegel (Chairman) Institute for Biochemistry I & CMMC Prof. Dr. Gabriele Pfitzer Institute for Vegetative Physiology & CMMC Substitute Prof. Dr. Christian Kubisch Institute of Human Genetics & CMMC Prof. Dr. Frank Berthold Department of Pediatric Oncology and Hematology & CMMC Dr. Ulrike Protzer (NG-F) hosted by: Institute for Medical Microbiology, Immunology and Hygiene & CMMC Prof. Dr. Rudolf Wiesner Institute for Vegetative Physiology & CMMC Members and their Substitutes elected by the Medical Faculty ( Fachbereichsrat ) Member Prof. Dr. Uwe Fuhr Institute for Pharmacology Prof. Dr. Arnulf Hölscher Department of Visceral and Vascular Surgery Prof. Dr. Wolfram Neiss Institute I for Anatomy Substitute Prof. Dr. Peter Mallmann Department of Gynaecology and Obstetrics Prof. Dr. Tobias Goeser Department IV of Internal Medicine Prof. Dr. Martina Deckert Department of Neuropathology 12
19 Executive Board Chairmen Prof. Dr. Martin Krönke Institute for Medical Microbiology, Immunology and Hygiene Prof. Dr. Thomas Krieg Department of Dermatology and Venerology Vice Chairman Prof. Dr. Thomas Langer Institute for Genetics Vice Chairman Research Area-Coordinators Research Area A: Cardiac and Vascular Disorders Prof. Dr. Wilhelm Krone Department II of Internal Medicine Research Area B: Host Response to Tumor Growth, Inflammation and Infection Prof. Dr. Mats Paulsson Institute for Biochemistry II Prof. Dr. Herbert J. Pfister Institute for Virology Research Area C: Molecular Neurobiology Prof. Dr. Dr. Wilhelm Stoffel Laboratory for Molecular Neuroscience Prof. Dr. Jens Brüning Institute for Genetics 13
20 Research Area A Research Area B Research Area C 15
21 Research Area A A 1 Molecular Mechanisms of Insulin Action and Insulin Resistance Jens C. Brüning 1 / Wilhelm Krone 2 Institute for Genetics 1 / Department II of Internal Medicine 2 Resistance to the metabolic action of insulin underlies the development of type 2 diabetes mellitus and associated complications. Recently, it has been recognized that inflammatory signals arise from increased body weight to inhibit insulin action thus leading to type 2 diabetes mellitus. Therefore, the aim of this project is to determine the nature of inflammatory pathways inhibiting insulin action and in turn to directly address the role of insulin signaling in inflammatory cells with respect to numerous diabetes-associated complications. Introduction Type 2 diabetes mellitus represents the most frequent endocrine disease affecting more than 6 % of modern western populations. A hallmark in the pathogenesis of this disease is the resistance to the metabolic effects of insulin, i.e. insulin-stimulated glucose transport in skeletal muscle and adipose tissue and insulin s ability to suppress hepatic gluconeogenesis. The development of type 2 diabetes mellitus is increasingly associated with that of obesity. Therefore, defining the interaction between obesity and insulin action is critical to the understanding of the pathophysiology of this disease, thus, ultimately leading to the development of novel therapeutic interventions. Over the last ten years, new insights into the development of obesity-associated insulin resistance have been gained. It has been demonstrated that under conditions of obesity there is an increased recruitment of Deciphering divergent intracellular signaling pathways evoked by pro-inflammatory cytokines and their effect on insulin action As mentioned above, increased release of proinflammatory cytokines such as tumor necrosis factor (TNF) and interleukin (IL)-6 result in the occurrence of insulin resistance in skeletal muscle and liver. One aspect of our project is to define the relative contribution of TNF- and IL-6-evoked intracellular signaling cascades in mediating inhibition of insulin action in classical insulin target tissues. Along this line, we have successfully inactivated the inhibitor of NFκB kinase (IKK)-2 gene specifically in skeletal muscle of mice. Exposing these animals to high fat diet revealed that skeletal musclerestricted IKK2-deficiency does not influence the occurrence of obesity-associated insulin resistance, thus underlining the relative importance of other TNF-evoked Aggravated atherosclerosis in IRS-2 -/- ApoE -/- mice: Cross section analysis of atherosclerotic lesions in the aortic root. Left panel demonstrates a representative result obtained in IRS-2 +/+ ApoE -/- and IRS-2 -/- ApoE -/- mice. The graph shows the quantitative assessment of lesion area in these animals for animals in each genotype. the innate immune response system, i.e. monocytes and macrophages into white adipose tissues, which in turn release pro-inflammatory cytokines, which interfere with insulin action in classical insulin target tissue. Therefore, the aim of our project is to define the relative contribution of individual cytokine-evoked signaling pathways to inhibit insulin action and in turn to investigate the role of insulin signaling itself in the innate immune response system. intracellular signaling cascades in mediating obesityassociated insulin resistance in skeletal muscle. Candidate pathways for this phenomenon comprise the activation of JNK among others. On the other hand, we could demonstrate that liver-restricted inactivation of the NFκB essential modulator (NEMO) protects from the occurrence of obesity-associated insulin resistance both systemically and in liver. Nevertheless, surprisingly we could demonstrate that hepatic NEMO-deficiency synergizes 16
22 Research Area A with high fat feeding in the development of liver tumors, thus unraveling a complex, potentially harmful side effect of the NFκB interfering therapeutic concept for obesityassociated insulin resistance, i.e. liver tumorigenesis. Further work will have to define a molecular interaction between nutrient signaling as present in high fat diet condition and the inflammatory signaling cascade in the development of this condition. Macrophage-autonomous insulin action To define the relative contribution of insulin action itself in components of the innate immune response system, we have specifically disrupted the insulin receptor gene in myeloid lineage cells by crossing mice carrying a flox insulin receptor allele with those expressing the Cre recombinase under control of the Lysozym M promoter. Resulting myeloid cell-restricted insulin knockout mice were then analyzed with respect to the development of atherosclerosis upon feeding of a western diet on an ApoEdeficient background. Surprisingly, we could demonstrate that myeloid cell-restricted insulin resistance protects from the development of atherosclerosis further revealing a so far unknown role for insulin action in the regulation of innate immune responses. Further developments in this project aim to define the molecular mechanisms, how insulin action regulates macrophage function. Perspectives Taken together, experiments over the last funding period have revealed important bilateral interactions between components of the innate immune response system and the metabolic insulin signaling cascade. Thus, further definition of the interaction of both signaling components may set the ground for the development of novel therapeutic interventions with obesity and insulinresistant type 2 diabetes mellitus. Selected publications Wunderlich FT, Luedde T, Singer S, Schmidt-Supprian M, Baumgartl J, Schirmacher P, Pasparakis P, Brüning JC. Hepatic NEMO-Deficiency Prevents Obesity Induced Insulin Resistance But Synergizes With High Fat Feeding in Liver Tumorigenesis. Proc Natl Acad Sci USA (2007), in press. Hovelmeyer N, Wunderlich FT, Massoumi R, Jakobsen CG, Song J, Worns MA, Merkwirth C, Kovalenko A, Aumailley M, Strand D, Bruning JC, Galle PR, Wallach D, Fassler R, Waisman A. Regulation of B cell homeostasis and activation by the tumor suppressor gene Cyld. J Exp Med (2007), in press. Ma X, Bruning J, Ashcroft F. (2007) Glucagon-like peptide 1 stimulates hypothalamic proopio-melanocortin neurons. J Neurosci (2007) 27, Konner C, Janoschek R, Plum L, Jordan S, Rother E, Ma X, Xu C, Enriori P, Hampel B, Barsh G, Kahn CR, Cowley MA, Ashcroft F, Bruning JC. Insulin action in AgRP-expressing neurons is required for suppression of hepatic glucose production. Cell Met (2007) 5, Seibler J, Kleinridders A, Kuter-Luks B, Niehaves S, Bruning JC, Schwenk F. Reversible gene knockdown in mice using a tight, inducible shrna expression system. Nucleic Acids Res (2007) 35, e54. Ma X, Zubcevic L, Bruning JC, Ashcroft FM, Burdakov D. Electrical inhibition of identified anorexigenic POMC neurons by orexin/ hypocretin. J Neurosci (2007) 27, Sheffler DJ, Kroeze WK, Garcia BG, Deutch AY, Hufeisen SJ, Leahy P, Bruning JC, Roth BL. p90 Ribosomal S6 Kinase 2 Exerts a Tonic Brake on G Protein-Coupled Receptor Signaling. Proc Natl Acad Sci USA (2006) 103, Plum L, Belgardt BF, Bruning JC. Central insulin action in energy and glucose homeostasis. J Clin Invest (2006) 116, Plum L, Ma X, Hampel B, Balthasar N, Coppari R, Muenzberg H, Shanabrough M, Burdakov D, Rother E, Janoschek R, Alber J, Belgardt B, Koch L, Seibler J, Schwenk F, Fekete C, Suzuki A, Mak TW, Krone W, Horvath TL, Ashcroft FM, Bruning JC. Enhanced PIP(3) signaling in POMC neurons causes KATP channel activation and leads to diet-sensitive obesity. J Clin Invest (2006) 116, Janoschek R, Plum L, Koch L, Muenzberg H, Diano S, Shanabrough M, Muller W, Horvath TL, Bruning JC. gp130 signaling in proopiomelanocortin neurons mediates the acute anorectic response to centrally applied ciliary neurotrophic factor. Proc Natl Acad Sci U S A (2006) 103, Horvath TL, Bruning JC. Developmental programming of the hypothalamus: a matter of fat. Nat Med (2006) 12, Baumgartl J, Baudler S, Scherner M, Babaev V, Makowski L, Suttles L, McDuffie M, Tobe K, Kadowaki T, Fazio S, Kahn CR, Hotamisligil GS, Krone W, Linton MR, Bruning JC. Myeloid lineage cellrestricted insulin resistance protects apolipoproteine-deficient mice against atherosclerosis. Cell Met (2006) 3, Seibler J, Kuter-Luks B, Kern H, Streu S, Plum L, Mauer J, Kühn R, Bruning JC, Schwenk F. Single copy shrna configuration for ubiquitous gene knockdown in mice. Nucleic Acids Res (2005) 33, e67. Plum L, Schubert M, Bruning JC. The role of insulin receptor signaling in the brain. TEM (2005) 16, (Review) Gropp G, Shanabrough M, Borok E, Xu AW, Janoschek R, Buch T, Plum L, Balthasar N, Hampel B, Waisman A, Barsh GS, Horvath TL, Bruning JC Agouti-related peptide-expressing neurons are mandatory for feeding. Nature Neurosci (2005) 8, Freude S, Schnitker J, Leeser U, Plum L, Udelhoven M, Krone W, Bruning JC, Schubert M. Peripheral hyperinsulinemia promotes promotes tau phosphorylation in vivo. Diabetes (2005) 54, Baudler S, Baumgartl J, Hampel, B, Buch T, Waisman A, Snapper CM, Krone W, Bruning JC Insulin-like growth factor-1 controls type 2 T cell-independent B cell response. J Immunol (2005) 174,
23 Research Area A A 2 Aspects of the Submembranous Region: The Role of Actin-Associated Proteins and their Role in Human Disease Angelika A. Noegel Institute for Biochemistry I The actin cytoskeleton is involved in various cellular aspects such as stability of a cell, cell motility, signal transduction, exo- and endocytosis. We are investigating components of the actin cytoskeleton that have both structural as well as regulatory roles. The main focus is on proteins of the alpha-actin in superfamily and the aim of this study is to unravel their physiological role as well as their relevance for human disease states. Introduction The region between the plasma membrane and the cortical cytoskeleton is a highly dynamic area which is involved in a variety of cellular activities. It organizes the cell to cell contacts, the cell to extracellular matrix interactions, and in motile cells it regulates cell motility by controlling the extension of pseudopods. It is involved in transmitting external signals to the inside of cells and it regulates exocytotic and endocytotic processes. This variety is based on the presence of a highly coordinated network of proteins representing membrane proteins, signaling proteins and structural proteins. The actin cytoskeleton and the nuclear membrane Until recently, nuclear positioning was thought to be controlled and maintained by microtubule motors and microtubules. This view has changed because genetic evidence in C. elegans demonstrated a role for the actin cytoskeleton in nuclear migration and positioning. Mutations in the gene Anc-1 resulted in irregular spacing and clumping of nuclei. ANC-1 was subsequently identified as a protein of enormous size which had an N-terminal actin binding domain (ABD) and a C-terminal transmembrane domain separated by a long rod domain. ANC-1 resembles in its overall structure Drosophila s MSP- 300 protein and the mammalian Syne-1, Myne-1 and NUANCE (also known as Nesprins). Novel actin binding proteins In our search for novel components of the actin cytoskeleton that carry an actin binding site of the alphaactinin type and are related to spectrin and dystrophin we isolated Enaptin and NUANCE, now designated Nesprin 1 and Nesprin 2. Both proteins are encoded by single genes composed of many exons and giving rise to a multitude of isoforms. Nesprin 1 and 2 are of enormous size (> kda), harbour an N-terminal ABD, a long coiled coil region containing spectrin repeats and a transmembrane domain with which they are anchored in the nuclear membrane. Immunoelectron microscopy showed their presence at the inner (INM) and outer nuclear membrane (ONM). At the inner face of the nuclear envelope (NE) they bind to emerin and lamin A/C, and lamin A/C deficiency leads to loss of Nesprins from the NE. In skin, Nesprin 2 is expressed in all epidermal layers including the basal layer where only lamin C is expressed indicating that lamin C is sufficient for localisation of Nesprin-2 at the NE. The localisation of Nesprins at the NE and their interaction with lamins opens the possibility that they are involved in laminopathies. Laminopathies cover a wide variety of diseases including muscular dystrophies, cardiomyopathies, premature aging and skin diseases such as restrictive dermopathy (RD). They are caused by mutations in lamins, lamin processing enzymes as in RD, where the ZMPSTE gene encoding an endoprotease is mutated, and in lamin-associated proteins such as emerin in Emery-Dreifuss muscular dystrophy. These defects are often associated with nuclear shape changes and other nuclear defects. The mechanisms underlying the diseases are not clear. Both transcriptional defects and/or mechanical defects have been proposed to explain their pathology which anchors them in the nuclear membrane. Generation of a nucleocytoskeletal link Nesprins are type II transmembrane proteins. Their C-terminal domain is rather short and is composed of about 30 amino acids that extend into the perinuclear space. This domain is highly conserved. It is found in all Nesprin homologues including the proteins in lower eukaryotes, and in other proteins of the nuclear envelope. We could show that through this region Nesprins associate with SUN-proteins in the perinuclear space, which is surrounded by the INM and OMN, and are retained in the nuclear membrane. SUN proteins are highly conserved proteins of the INM. Lower eukaryotes have one gene coding for a SUN protein, higher eukaryotes have two SUN genes. Moreover, there exist isoforms of SUN. SUN proteins are anchored in the INM through a variable number of transmembane domains. The N-terminus extends into the nucleoplasm and can interact with nuclear proteins such as lamins and presumably with chromatin. The large C-terminal domain has a coiled-coil domain, through which the protein can dimerise, and a large SUN-domain that is highly conserved among the SUN proteins. The Nesprin-SUN-linkage provides a continuum between the nucleus and the cytoskeleton in the cytoplasm and can therefore link the plasma membrane to the nucleus (Figure 1). Furthermore, the Nesprin-SUN-interaction might ensure, that the spacing of the nuclear membranes is kept constant. 18
24 Research Area A Fig. 1: Model illustrating the interactions of Nesprins and SUN proteins in the cell. The scheme depicts (not drawn to scale) the interaction between Sun1 (green), Sun2 (pink) and KASH domain (orange) proteins (Nesprin-1, -2, -3) in the perinuclear space. Sun1 binds to the lamins (blue strands) and chromatin (black) and associates with emerin (purple) and LAP2β (brown) directly or indirectly. Nesprin-1 and -2 in the outer nuclear membrane connect the nucleus to the actin cytoskeleton (red) and Nesprin-3, a novel Nesprin lacking the N-terminal ABD, may associate with Sun1 to connect the nucleus to the intermediate filaments (blue bar) and actin filaments via its interaction with plectin (light blue). SUN domain proteins in C. elegans and fission yeast bind to the MTOC or MTOC equivalent structures, therefore in higher eukaryotes SUN proteins may also bind to the MTOC or to microtubules via unknown protein (brown oval with question mark). IF: intermediate filament; ONM: outer nuclear membrane; PNS: perinuclear space; INM: inner nuclear membrane; MTOC: microtubule organizing center. Question marks indicate unknown protein or hypothetical interactions (modified from Padmakumar et al., 2005). Perspectives The relevance of actin-binding proteins for correct functioning of a cell is underlined by a variety of human diseases that are linked to mutations in genes such as the BPAG1 gene, the dystrophin or the plectin gene which are involved in the generation of neurological disorders or cardiac and skeletal myopathies. Moreover, several such diseases have been linked to the nuclear envelope. They have been called laminopathies or nuclear envelopathies because proteins that are associated with the nuclear lamina carry disease-linked mutations. Frequently these defects are associated with structural nuclear abnormalities. Mutations in nuclear envelope proteins such as emerin or in nuclear lamina proteins such as the lamins have been identified as causing a variety of disease states. The disease mechanisms are however not known. Based on their presence at the nuclear envelope and their interactions with nuclear envelope proteins and cytoplasmic components the Nesprins are involved in organizing the interaction between nucleus and cytoplasm. They present further candidates for nuclear envelopathies and exploring their functions and interactions might help to unravel the mechanisms causing nuclear envelopathies. Selected publications Chu H, Thievessen I, Sixt M, Lämmermann T, Waisman A, Braun, A, Noegel AA, Fässler R: γ-parvin is dispensable for hematopoiesis, leukocyte trafficking, and T-cell-dependent antibody response. Mol Cell Biol (2006) 26, Padmakumar VC, Libotte T, Lu W, Zaim H, Abraham S, Noegel AA, Gotzmann J, Foisner R, Karakesisoglou I: Sun1, an inner nuclear membrane protein, mediates the anchorage of Nesprin-2 to the nuclear envelope. J Cell Sci (2005) 118, Libotte T, Zaim H, Abraham S, Padmakumar VC, Schneider M, Lu W, Munck M, Hutchison C, Wehnert M, Fahrenkrog B, Sauder U, Aebi U, Noegel AA, Karakesisoglou I: Lamin A/C dependent localization of Nesprin-2, a giant scaffolder at the nuclear envelope. Mol Biol Cell (2005) 16, Rick M, Ramos Garrido SI, Herr C, Thal DR, Noegel AA, Clemen CS: Nuclear localization of Annexin A7 during murine brain development. BMC Neuroscience (2005) 6, 25 (10 April 2005). 19
25 Research Area A A 3 Molecular Dissection of the Hyperpolarization-Activated Inward Current I f Uta C. Hoppe / Erland Erdmann Department III of Internal Medicine The hyperpolarization-activated inward current If is essential for the spontaneous cardiac beating activity. In the present project we will analyse the single channel function of the coding genes HCN1-4. We will determine the relevance of specific ion channel regions for the observed single channel characteristics using site-directed mutagenesis. Secondly we will determine the effect of modulating auxiliary subunits on IHCN and If properties and spontaneous cardiac action potential rate. Introduction In sinus node and Purkinje cells the hyperpolarizationactivated cation current I f is considered to contribute significantly to the spontaneous pacemaker activity. An I f -like current has also been recorded in ventricular and atrial myocytes of mammalian species and in human myocardial tissue. In heart failure and in spontaneously hypertensive rats I f density is increased compared to undiseased controls. This led to the hypothesis that an overexpression of I f might contribute to the increased propensity of ventricular arrhythmias in diseased states. Conversely, dysfunction or current reduction of I f in sinus node cells might cause bradyarrhythmias. Four HCN gene family members have been cloned, three of which are present in heart (HCN1, HCN2, HCN4) with varying message levels in different cardiac regions. Heterologous expression of these channel subunits results in a hyperpolarization-activated inward current with similar, though not identical properties compared to native I f. Single-channel properties support a potential contribution of HCN channels and I f to cardiac arrhythmias The pacemaker current I f is present in atrial and ventricular myocytes. However, it remains controversial whether I f overexpression in diseased states might play a role for arrhythmogenesis, since first I f activation in whole-cell recordings hardly overlapped the diastolic voltage of working myocardium. To obtain further insight into IHCN and I f properties we provide for the first time detailed single-channel analysis of heterologously expressed HCN isoforms and native human I f. HCN subtypes differed significantly in singlechannel amplitude, conductance, and activation kinetics. Interestingly, threshold potentials of HCN isoforms were more positive than would have been expected from whole-cell measurements. Single-channel properties of cells cotransfected with HCN2 and HCN4 were distinct from cells expressing HCN2 or HCN4 alone, demonstrating that different HCN isoforms can influence current properties of one single HCN channel complex, thus, providing direct functional evidence for HCN heteromerization. Pooled data of homomeric and heteromeric HCN channels and of native I f extrapolated from maximum likelihood fits indicated a multistate gating scheme comprising five closed and four open channel states. Single-channel characteristics of I f in human atrial myocytes closely resembled those of HCN4 or HCN2+HCN4, supporting the hypothesis that native I f channels in atrial myocardium are heteromeric complexes composed of HCN4 and/or HCN2. Most interestingly, halfmaximal activation of single-channel atrial I f (-68.3±4.9 mv, k=-9.9±1.5, n=8) was well within the diastolic voltage range of human atrial myocardium. These observations support a potential contribution of HCN/I f to the arrhythmogenesis of working myocardium under pathologic conditions. Fig. 1: Single channel currents and voltage-dependence of activation of I HCN2+HCN4 and atrial I f. 20
26 Research Area A KCR1 profoundly modulates gating of the hyperpolarization-activated pacemaker channel and spontaneous action potential activity HCN channel subunits (HCN1-4) are believed to be modulated by additional regulatory proteins. So far only KCNE2 has been reported to serve as a modulating subunit of HCN channels. KCNE2 enhanced the expression and accelerated activation of I HCN. Since these effects are not sufficient to explain I f properties in native tissue, it is likely that HCN currents are being modulated by one or more additional proteins. Using biochemistry, molecularbiology and electrophysiology methods we demonstrated a protein-protein interaction between HCN2 and the K + channel regulator protein 1, named KCR1. In coimmunoprecipitation experiments we showed that KCR1 and HCN2 proteins are able to associate. Heterologously expressed HCN2 whole-cell current density (79.6±13.9 pa/pf at -130 mv, n=9) was significantly decreased by KCR1 (3.6±2.1 pa/pf, ratio 1:2, n=10; p<0.001). KCR1 profoundly suppressed I HCN2 singlechannel activity and shifted the half-maximal activation, indicating a direct interaction between KCR1 and the HCN2 channel subunit. Endogenous KCR1 could be detected on mrna-level in adult and neonatal rat ventriculocytes. Adenoviral-mediated overexpression of KCR1 in rat cardiomyocytes (i) reduced I f whole-cell currents from 4.7±0.3 pa/pf (n=14) to 0.9±0.1 pa/pf (n=9) in KCR1- infected cells when measured at -130 mv (p<0.001), (ii) suppressed most single-channel gating parameters, (iii) altered the activation kinetics, and (iv) entirely suppressed spontaneous action potential activity. Figure 2 (middle) illustrates a representative recording of an action potential that was induced artificially by a short depolarizing current pulse in a KCR1-infected myocyte. More importantly, sirna-based knock-down of endogenous KCR1 increased the native I f current density (6.09±0.6 pa/pf n=11; p=0.04 vs. control) and single-channel activity. KCR1 profoundly altered gating kinetics of both recombinant HCN2 and native I f in both adult and neonatal ventriculocytes. Moreover, KCR1 knock-down markedly accelerated spontaneous beating rate from 80.4±5.8 bpm to 100.2±4.6 bpm (n=17; p<0.005 vs. control) without significantly changing the maximal diastolic potential. These results support a direct inhibitory action of KCR1 on native I f. Our observations demonstrate for the first time that KCR1 directly modulates I HCN2 /I f channel gating and indicate that KCR1 might serve as a β-subunit of native I f. Fig. 2: Effect of enhanced KCR1 expression (KCR1-infected) and knock-down of endogenous KCR1 (KCR1 sirna - infected) on I f whole-cell currents and spontaneous action potentials of neonatal cardiomyocytes. Perspectives Given the obvious inhibitory effects of KCR1 on HCN channels and native I f, alterations of KCR1 expression might cause changes in heart rate or ectopic activity in patients, though our in-vitro observations cannot readily be generalized to humans in-vivo. Further potential KCR1- induced regulatory mechanisms might involve alteration of the intracellular camp level, HCN trafficking and cellsurface stability. Although such effects could not explain the changes of I f single-channel gating, they might contribute to the observed KCR1-mediated actions and will be addressed in future studies. Interacting proteins like KCR1 inhibiting I f and, thus, depressing spontaneous activity may provide a novel therapeutic target of cardiac arrhythmias. Selected publications Er F, Michels G, Gassanov N, Rivero F, Hoppe UC. Testosterone induces cardioprotection by activating mitochondrial ATP-sensitive K + channels. Circulation (2004) 110, Michels G, Er F, Khan I, Südkamp M, Herzig S, Hoppe UC. Singlechannel properties support a potential contribution of HCN channels and I f to cardiac arrhythmias. Circulation (2005) 111, Michels G, Er F, Eicks M, Herzig S, Hoppe UC. Long-term and immediate effect of testosterone on single T-type calcium channel in neonatal rat cardiomyocytes. Endocrinology (2006) 147,
27 Research Area A A 4 Endothelial Cell Damage and Repair in the Retina and Choroid Antonia M. Joussen / Sascha Fauser / Bernd Kirchhof Department of Ophtalmology Currently, the inbalace of pro-and antiangiogenic factors such as VEGF and PEDF is thought to be causal for several retinal and choroidal neovasculari diseases including diabetic retinopathy, uveitis, and choroidal neovascularization. A common feature of these diseases seems to be endothelial cell apoptosis that is antagonized by natural repair mechanisms such as the recruitment of endothelial precursor cells (EPCs) to the vascular walls. We aim to determine the role of inflammatory mediators and endothelial precursor cells in retinal and choroidal vascular pathology. Introduction Emerging evidence suggests that bone marrow derived endothelial, hematopoietic stem and progenitor cells contribute to tissue vascularization during both embryonic and postnatal physiological processes. Corecruitment of angiocompetent hematopoietic cells delivering specific angiogenic factors facilitates incorporation of endothelial progenitor cells (EPCs) into newly sprouting blood vessels but also at sites of tissue repair (Rafii et al 2003, Mathews et al. 2004, Grant et al. 2002). We aim to identify the cellular mediators and tissue-specific chemokines, which alter the recruitment of bone marrow derived stem and progenitor cells to pathological retinal and choroidal neovascularization. These investigations help to eludicate new effective treatment targets in the prevention of diabetic macular edema and choroidal neovascularization. Lethal Irradiation May Lead To Long-Term Radiation Retinopathy In A GFP+-Chimeric Mouse Model In order to create a reliable model for investigation of long-term retinal and choroidal disease, the construction of chimeric GFP+ mice has been investigated. The construction of chimeric GFP+-mice has become a powerful tool in the field of stem cell-participation and - homing in various models of ocular pathology. Lethal irradiation was achieved at a total dose of 11 Gy, higher doses were lethal notwithstanding successful transplantation procedure. FACS-analysis revealed average transformation rates of 75% with a trend towards higher rates with increasing age of recipient mice, ranging as high as 90%. Perfused flatmounts 8 weeks after transplantation were almost free of extravasal GFP+-cells, whereas in the 5-months group cluster cell infiltrations, around the posterior pole and in the peripheral retina, became apparent. Cell morphology appeared from oval to cells with few extensions to dendritiform cells. In the 7- month group a variable cell number was found, and cells with a typical macrophage-like morphology were noted. Colocalization staining showed GFP+- as well as GFP-- macrophages and dendritic cells. Following lethal irradiation, bone marrow-derived leukocytes seem to infiltrate the retina, commencing at about 3 months after irradiation. With regard to the observed radiation-related inflammatory reaction after long-term transplantation, this model may be unsuitable for investigation of prolonged retinal diseases. Thus further investigation focussed on short-time experiments in inflammatory eye disease and choroidal neovascularization. Fig. 1: Retinal aspects of posterior pole and periphery at 2, 5 and 7 months after irradiation and GFP+ bone marrow transplantation. 22
28 Research Area A To investigate the role of stationary and migrated immunocompetent cells in ocular inflammation This study is to determine to which extent local retinal - and bloodstream derived immunocompetent cells participate after inflammation. In order to create retinal vascular inflammation, a model of LPS-induced uveitis was used after stable transplantation of GFP positive bone marrow cells. FACS-analysis of peripheral blood draws showed an average of 70.68% GFP+ transformation rate after bone marrow substitution. Perfused retinal flatmounts of nonuveitic control animals were free of extravasal GFP-positive cells. A significant difference (p<0,001) in the amount of CD11b-positive cells was found between uveitic and non uveitic mice and between cells in patches and outside (p<0.005). FACS-analysis of the blood showed an increase in CD11b and F4/80-positive cells as well as a decrease in CD3-positive cells in the LPS treated animals compared to controlls. These findings suggest an activation of residential immunocompetent retinal cells together with an influx of bloodstream-derived cells during LPS induced inflammation. To investigate the role of inflammatory mediators and bone-marrow-derived stem cells in a model of experimental choroidal neovascularization Chroidal neovascularization is the main feature of wet age-related macular degeneration. In the recent past, an inflammatory component has been shown to be involved in the pathogenesis of choroidal and retinal neovascularization. Macrophage-derived TNF-a increases the expression of vascular adhesion molecules (ICAM-1, VCAM-1, E-selectin) and VEGF. In current study, we investigate the role of bone marrow cells in laser-induced CNV in TNFR1b -/- mice. Lethally irradiated adult TNFR1b -/- mice as well as C57BL/6 mice were used as recipients for bone marrow transplantation. Whole bone marrow transplants from C57BL/6 and TNFR1b -/- mice served as controls. One month after transplantation, FACS analysis showed 70% gfp + circulating cells in both C57BL/6 and TNFR1b -/- mice. Gfp + -cells were localized predominantly within and around the edge of the CNV lesion and within the optic nerve head. Some gfp + -cells with branching processes (F4/80-positive) were observed to infiltrate the overlying neurosensory retina. The area of neovascularization (µm 2 ) was significantly reduced in TNFR1b -/- in comparison to C57BL/6 recipients. Gfp + fluorescence areas within the CNV were significantly reduced in TNFR1b -/- in comparison to C57BL/6 recipients. Laser injury induced recruitment of bone marrow-derived cells to the CNV lesions in both TNFR1b and C57BL/6 mice. CNV injury was reduced in TNFR1b -/- in comparison to C57BL/6 recipients. It remains to be determined whether decreased CNV formation in TNFR1b recipients is due to decreased involvement of transplanted cells to the formation of new vessels or to other mechanisms associated with the absence of TNFR1b. Fig. 2: Distribution and morhology of the gfp+-cells in the neurosensory retina over the laser burns. A: Gfp+labeled cells infiltrated the overlying neurosensory retina over the CNV lesions. Many of these cells expressed typical macrophage morphology and therefore seems to be circulating macrophages. Such bone-marrow-derived macrophages were often associated with retinal blood vessels. B: confocal microscopy image of laser burn and surrounding retinal vessels. The laser scar can be seen under the deeper layer of retinal vessels. C,D: Gfp+-cells were present surrounding the radial retinal vessels. Most of these cells had an elongated shape and were oriented parallel to the vessel walls but outside from the blood vessels. Perspectives Our studies have demonstrated the role of inflammatory cells in retinal vascular pathology. While most of the current data investigates whole bone marrow responses, we will now investigate the role of selected precursor cells. Based on our previous investigations, diabetic retinopathy has many features of a low-grade inflammation. Further studies will focus on the role of bone marrow derived cells in the pathogenesis of diabetic retinopathy using different models of genetically determined and pharmacologically induced diabetes. Selected publications Kociok N, Radetzky S, Krohne TU, Gavranic C, Joussen AM: Pathological but not physiological retinal neovascularization is altered in TNF-Rp55-receptor-deficient mice. Invest Ophthalmol Vis Sci (2006) 47, Shi X, Semkova I, Muther PS, Dell S, Kociok N, Joussen AM: Inhibition of TNF-alpha reduces laser-induced choroidal neovascularization. Exp Eye Res (2006) 83, Semkova I, Fauser S, Lappas A, Smyth N, Kociok N, Kirchhof B, Paulsson M, Poulaki V, Joussen AM: Overexpression of FasL in retinal pigment epithelial cells reduces choroidal neovascularization. FASEB J (2006) 20,
29 Research Area A A 5 Role of β-subunit Genes and Splice Variants for Pathological Gating of Human Cardiac L-Type Calcium Channels in Heart Failure Stefan Herzig 1 / Uta C. Hoppe 2 Institute for Pharmacology 1 / Department III of Internal Medicine 2 The L-type calcium channel triggers cellular responses as diverse as muscle contraction, exocytosis, and gene transcription. We focus on the structural and functional heterogeneity of this heteromultimeric channel complex in human heart failure. The molecular composition of heteromultimeric channels in general, and the role and the regulation of β-subunits (β 1, β 2, β 3 ) in particular are studied in human tissue and genetic mouse models to gain detailed molecular information on these channels, and to derive novel therapeutic strategies. Introduction The pathophysiological role of β-subunits in human heart has been illustrated by the differential expression pattern found in failing human heart (Hullin et al., 2003), leading to hyperactive single channels (Schröder et al., 1998). Guided by the molecular and functional diversity of β-subunit genes and splice variants we pursued three specific aims: Assess the functional relevance of differential β-subunit expression in diseased human heart Define the biophysical structure-function analysis of the N-terminus of β-subunits Generate and use models (in vitro, ex vivo, in vivo) to assess the therapeutic potential of β-subunits, N-terminal peptides and small molecules Functional relevance of differential β-subunit expression in diseased human heart The differential expression of β-subunits is now convincingly demonstrated in human failing heart (Hullin et al., 2007), and in a genetic mouse model of heart failure (Groner et al., 2004, Hullin et al., 2007). The β 2 subunit messenger RNA (Northern blot, real-time PCR) and protein expression (Western) increase along with the development of hypertrophy and failure in both models. No changes or opposite effects were observed with respect to β 1 - and β 3 -subunits. Given the functional heterogeneity of these subunits when assessed in recombinant channels (Hullin et al., 2003, 2007), we are now able to explain the pathological phentopype associated with human heart failure (Schröder et al., 1998). Biophysical structure-function analysis of the N- terminus of β-subunits We recently revealed the structural basis of differential effects of the five β 2 -subunits isoforms expressed in human heart (β 2a-e ) on the single L-type calcium channel current. These splice variants differ only by N-terminal length and amino acid composition (Herzig et al., 2007). β 2 -subunit effects were investigated in HEK293 cells expressing the recombinant L-type ion conducting pore. All β 2 -subunits increased open probability, availability and peak current with a highly consistent rank order (β 2a β 2b >β 2e β 2c >β 2d ). We show graded modulation of transition rates within and between deep-closed and inactivated states (Figure 1). The extent of modulation correlates strongly with the length of N-terminal domains. Two mutant β 2 -subunits which imitate the natural span related to length confirm this conclusion. The data show that the length of N-termini is a relevant physiological mechanism for channel closure and inactivation, and that alternative splicing exploits this principle for the modulation of gating properties of calcium channels. Fig. 1: Quantitative modeling of single-channel gating behavior with β 2b (780 sweeps from n= 4 experiments), β 2c (2460 sweeps from n= 10 experiments) and β 2d (1200 sweeps from n= 7 experiments): Idealized data from highquality experiments were used to compute the time course of open probability within the sweep, and to construct cumulative distribution functions of first latency, open times, and closed times (black and gray). Fits to the Markov model (top) are displayed as smooth red curves. Confirmingly, introduction of similar N-terminal alterations in the β 1 -subunit (where no alternative splicing exists) by site-directed mutagenenis reveals the same structurefunction relationship at the level of single-channel biophysics after heterologous expression (Jangsangthong W, Akhavan-Malyer R et al., not shown). This indicates that the N-terminus of β-subunits serves as structural motif 24
30 Research Area A facilitating inactivation, possibly caused simply by its own polypeptide mass. The next study will focus on the β 4 - subunit, of which high-resolution structural data of the N-terminus is already available. Generation of an in vivo model to assess the therapeutic potential of β-subunits To prove the causal relationship between β 2 -subunit overexpression and pathological gating of single L-type channels in heart failure, we generated a new genetic mouse model, designed to imitate the scenario in human heart: mice overexpressing the human cardiac calcium channel pore subunit reveal downregulated β 2 - subunits and low channel activity at young age (Groner et al., 2004). Only at an age of 9 months, they develop β 2 -subunit overexpression, pathological channel gating and heart failure. However, we can acutely (48 hours) induce β 2 -subunit protein overexpression and the channel gating pattern typical of heart failure in young animals (Hullin et al., 2007) bearing a second additional transgene, enabling ecdysone-dependent cardiac-specific overexpression of a rat β 2a -subunit (Figure 2). Experiments are underway to prove the principle of this approach by using competition assays between functionally distinct β-subunits. We will furthermore use our methodological armentarium to assess the structural and functional alterations of calcium channels in other diseases of the cardiovascular system, including diabetic cardiomyopathy and atherosclerosis. Selected publications Schröder F, Handrock R, Beuckelmann DJ, Hirt S, Hullin R, Priebe L, Schwinger RHG, Weil J, Herzig S Increased availability and open probability of single L-type calcium channels from failing compared with nonfailing human ventricle. Circulation (1998) 98, Hullin R, Khan IF, Wirtz S, Mohacsi P, Varadi G, Schwartz A, Herzig S Differential effects of recombinant cardiac calcium channel β-subunits revealed by single-channel analysis. J Biol Chem (2003) 278, Groner F, Rubio M, Schulte-Euler P, Matthes J, Khan IFY, Bodi I, Koch SE, Schwartz A, Herzig S (2004) Single-channel gating and regulation of human L-type calcium channels in cardiomyocytes of transgenic mice. Biochem Biophys Res Comm (2004) 314, Fig. 2: Characterisation of double transgenic mouse (constitutive cardiac overexpression of human Cav1.2, plus cardiac-specific, inducible overexpression of rat β2a-subunit). Left: Southern blots of an ecdysone receptor transgene coupled to α-mhc promotor, and of β 2a -transgene coupled to ecdyson-responsive element. Middle: Induction of β 2a - subunit protein expression, and Right: Increase of single channel activity, both taken 48 hours after in vivo-application of tebufenocide ( 9 mg i.p.). Perspectives We have now proven the causative role of β 2 -subunit overexpression for the altered single channel behaviour in human and mouse heart failure. The next step will be to assess whether this phenotype is adaptive or maladaptive, i.e. whether β 2 -subunit overexpression (used as independent experimental variable) will cause or prevent hypertrophy, arrhythmia and failure in double transgenic mice after chronic (4 weeks) induction. The methods for this approach have been established and validated. The novel mechanistic understanding of β 2 -subunit modulation will allow to design disease-specific (pharmacological, peptide) tools to ameliorate the pathological gating phenotype of cardiac calcium channels. Herzig S, Khan IFY, Gründemann D, Matthes J, Ludwig A, Michels G, Hoppe UC, Chaudhuri D, Schwartz A,Yue DT, Hullin R Mechanism of Cav1.2 channel modulation by the N-terminus of cardiac β 2 -subunits. FASEB J (2007) 21, Hullin R, Matthes J, von Vietinghoff S, Bodi I, Rubio M, D Souza K, Khan IF, Rottländer D, Hoppe UC, Mohacsi P, Schmitteckert E, Gilsbach R, Bünemann M, Hein L, Schwartz A, Herzig S (2007) Increased expression of the auxiliary β 2 -subunit of ventricular L- type Ca 2+ channels leads to single-channel activity characteristic of heart-failure. PLoS ONE 2(3): e292. doi: /journal. pone
31 Research Area A A 6 Characterization of Atherogenic Signaling Pathways by the βpdgf Receptor in vivo Stephan Rosenkranz Department III of Internal Medicine Platelet-derived growth factor (PDGF) plays a pivotal role in atherogenesis. We investigate the relative importance of PDGF-induced signals in vitro by the use of βpdgf receptor mutants and in vivo by selective deletion of the βpdgfr gene in VSMCs and macrophages in an atherosclerosis mouse model. Our in vitro data revealed that PI 3-kinase (PI3K) and phospholipase Cγ (PLCγ) are crucial for cellular proliferation, migration and apoptosis. Therefore, we currently analyse knockin mice expressing βpdgfr mutants which are selectively unable to bind / activate PI3K and / or PLCγ Introduction Atherosclerosis and its consequences such as myocardial infarction, stroke, and peripheral artery disease remains the leading cause of death in developed countries. In particular, migration, proliferation and apoptosis of vascular smooth muscle cells (VSMCs) are crucial for the development and progression of atherosclerotic plaques. Plateletderived growth factor (PDGF) is one of the most potent mitogens and chemoattractants for VSMC. PDGF elicits its biological responses via two receptor tyrosine kinases, termed α and β PDGF receptor (PDGFR). Recent studies revealed that of the PDGFR subtypes, βpdgfr-induced signals are particularly important for vascular development and remodeling. Upon ligand binding, PDGFRs dimerize, autophosphorylate on tyrosine residues, and the phosphorylated tyrosines serve as specific docking sites for Src homology 2 (SH2) domain-containing signal relay enzymes. In the βpdgfr, seven major phosphorylation sites have been identified, through which the activated receptor associates with and thus activates Src family kinases (Src), phosphatidylinositol 3-kinase (PI3K), RasGAP, the phosphotyrosine phosphatase SHP-2, and phospholipase Cγ1 (PLCγ). Signal relay of βpdgfr-dependent proliferation, migration, and survival To systematically evaluate the role of each of the signaling molecules for βpdgfr-mediated cellular responses in VSMCs, we created a series of βpdgfr mutants in which the tyrosine residues required for the association of either one of the signaling enzymes were mutated to phenylalanine. To bypass endogenous PDGFRs in VSMCs, we used chimeric βpdgfrs (ChiR) with altered ligand binding specificity. ChiRs contain the extracellular domain of the M-CSF receptor, which is not expressed in VSMCs, and the cytoplasmic domain of the wild type (WT) or mutated βpdgfr. Hence, ChiR mutants are selectively activated with M-CSF in transfected cells. To characterize the role of each signal relay enzyme for the various PDGF-dependent cellular responses, we compared the ability of each ChiR mutant to mediate VSMC proliferation, migration, and survival to that of the WT receptor. Using this system, we were able to demonstrate that PI3K and PLCγ mediate PDGF-dependent VSMC proliferation, and that PI3K, PLCγ and Src are involved in VSMC migration, whereas other βpdgfr-associated signaling molecules such as SHP-2 and RasGAP do not play a significant role for these cellular responses. A combination mutant lacking the binding sites for PI3K and PLCγ was insufficient to mediate VSMC proliferation and migration indicating that these two signaling enzymes are critical for atherogenic cellular responses. In addition, we were able to show that the anti-apoptotic effect of PDGF is exclusively mediated via PI3K/Akt (Fig. 1). Fig. 1: Summary of our results identifying the signal relay enzymes that mediate βpdgfr-induced DNA synthesis, chemotaxis, and regulation of apoptosis in VSMCs in vitro. The βpdgfr mediates pro-apoptotic signals in VSMC via PI 3 kinase dependent pathways Analysis of the time course of βpdgfr-mediated survival revealed that protection against apoptosis is only transient. Furthermore, long-term activation of the βpdgfr led to an increase of apoptosis. To determine the signaling mechanisms underlying the proapoptotic effect of PDGF- BB on VSMC we analysed the expression of Bcl-2 proteins which constitute central regulators of cellular apoptosis. In human atherosclerotic plaques the expression levels of antiapoptotic Bcl-2 and proapoptotic Bax seems to 26
32 Research Area A be particularly important for VSMC survival Western blot analysis revealed that the protein expression of Bcl-2 began to decrease 1 day upon stimulation with PDGF-BB and remained low until 4 days. Using the chimeric mutant system, we were able to demonstrate that induction of apoptosis and Bcl-2 downregulation solely depends on PI3K. These data for the first time indicate that growth factor receptors like the βpdgfr are able to induce apoptosis. Characterization of atherogenic βpdgfr-dependent signaling in vivo To investigate whether progression of atherosclerotic lesions can take place in the absence of βpdgfr signaling in macrophages or VSMCs, or in the absence of PDGFdependent activation of PI3K and PLCγ, we have created mice with the background of an atherosclerosis model (Fig. 2). Fig. 2: Characterization of atherogenic βpdgfr-dependent signaling in vivo. Mice are generated on the background of an atherosclerosis model (LDLR -/- ). LDLR -/- /mph PDGFR -/- mice and LDLR -/- /smpdgfr -/- mice contain a conditional inactivation of the βpdgfr gene in macrophages or VSMC, respectively. In LDLR -/- /LRP1 -/- mice the LRP1 gene is inactivated specifically in VSMC causing an overexpression of the βpdgfr. LDLR -/- /PDGFR F3/F4 or F2/F2 mice express a F3 or a F2 βpdgfr mutant instead of the βpdgfr WT receptor. To investigate the pathophysiological relevance of PDGFmediated responses for atherogenesis in macrophages vs. VSMCs in vivo we have generated mice with a selective deletion of the βpdgfr gene in VSMC (LDLR -/- /smpdgfr - /- ) or in macrophages(ldlr -/- /mphpdgfr -/- mice) on the background of an atherosclerosis model (LDLR -/- -mice). The generation of LDLR -/- mice lacking the βpdgfr specifically in macrophages is performed by adoptive transfer. The creation of LDLR-deficient, VSMC-specific βpdgfr knockout mice is performed by using the Cre-loxP system. Based on our in vitro findings, we currently investigate the role of PI3K and PLCγ for the formation and progression of atherosclerotic plaques in vivo. Therefore, we use knockin mice expressing mutant PDGFRs which are unable to bind or activate PI3K and / or PLCγ (F3 and F2 mutants). These animals are crossed with LDLR -/- -mice, and the resulting LDLR -/- /PDGFR F3/F3 -mice are evaluated in terms of atherosclerotic lesion formation. All mice are fed a cholesterol-rich diet, and quantification of atherosclerotic lesion size is performed at 15 weeks. The quantification of atherosclerotic plaques is performed on histological cross-sections of the aortic arch. In addition to the established methods for quantification of atherosclerotic lesions in mice, we utilize a novel approach enabling us to visualize and measure plaque size in vivo by magnetic resonance imaging (MRI). To investigate βpdgfr-mediated pro-apoptotic effects we use LDLR -/- /LRP1 -/- mice, which specifically overexpress the βpdgfr in VSMC due to the VSMC-specific lack of LRP1. The quantification of apoptosis and Bcl-2 expression in plaque VSMC is performed on histological crosssections of the aortic arch. Perspectives The use of specific βpdgfr mutants along with conditional βpdgfr knock out/in mice provide evidence of the role of βpdgfr signaling in the development of atherosclerosis and its consequences such as myocardial infarction, stroke, and peripheral artery disease. The identification of the signaling molecules which are responsible for βpdgfr-mediated atherogenic signaling provide potential targets for pharmacological intervention in the treatment of atherosclerosis. Selected publications Vantler M, Huntgeburth M, ten Freyhaus H, Schnabel P, Rosenkranz S. Antiapoptotic signaling by the PDGF α receptor is mediated by PI 3-kinase and negatively regulated by Src family kinases. FEBS Lett. (2006) 580, Ten Freyhaus H, Huntgeburth M, Wingler K, Schnitker J, Bäumer AT, Vantler M, Bekhite M, Wartenberg M, Sauer H, Rosenkranz S. Novel NOX inhibitor VAS2870 attenuates PDGF-dependent smooth muscle cell chemotaxis, but not proliferation. Cardiovasc. Res. (2006) 71, Vantler M, Caglayan E, Zimmermann WH, Baumer AT, Rosenkranz S. Systematic evaluation of anti-apoptotic growth factor signaling in vascular smooth muscle cells. Only phosphatidylinositol 3 -kinase is important. J. Biol. Chem. (2005) 280, Rosenkranz S, Knirel D, Dietrich H, Flesch M, Erdmann E, Böhm M. Inhibition of the PDGF receptor by red wine flavonoids provides a molecular explanation for the French paradox. FASEB J. (2002) 16,
33 Research Area A A 7 Commitment of the Extraneuronal Monoamine Transporter (EMT) in Cardiovascular Inactivation of Noradrenaline Dirk Gründemann / Edgar Schömig Institute for Pharmacology In cardiovascular disease, high levels of circulating catecholamines exacerbate pathophysiology. EMT (gene symbol SLC22A3), which was cloned by our group, is a key system for the inactivation of catecholamines. To assess the significance of EMT in disorders of the cardiovascular system and the potential for pharmacological intervention, we attempt to (i) generate an EMT-knock-out mouse and analyze the consequences of EMT ablation and (ii) explore the molecular mechanism underlying transporter activation. Introduction The sympathetic nervous system controls many cardiovascular functions, e.g. blood pressure, heart frequency, and inotropy. In pathophysiological situations like myocardial ischemia and heart failure, levels of free catecholamines often rise dramatically. Indeed, for heart failure, the concentration of circulating catecholamines correlates inversely with disease prognosis. This overload of free catecholamines causes excessive myocardial stimulation which in turn further advances damage of the heart muscle. For ischemia, high levels of noradrenaline promote, by means of an increase of myocyte energy consumption and Ca 2+ influx, cell death in ischemic borderline areas. Likewise in an animal model, high concentrations of exogenous noradrenaline induce myocardial necrosis. Interestingly, pharmacological blockade of sympathetic overstimulation with ß- adrenoceptor antagonists significantly reduces mortality in heart failure and in acute myocardial infarction. catecholamine carriers, which after all directly control the levels of free noradrenaline and adrenaline. Interestingly, radiotracer imaging suggests that in chronic heart failure neuronal reuptake of noradrenaline is reduced. On the non-neuronal side, detailed information on the molecular function, localization, regulation and genetic variability of the extraneuronal monoamine transporter EMT is only beginning to emerge. Besides receptor stimulation, removal of transmitter from extracellular space is a major factor to determine synaptic signalling intensity. Noradrenaline, the major transmitter of postganglionic sympathetic neurons, is inactivated by two distinct transport mechanisms (Fig. 1). The first uptake system (NET), located at the plasma membrane of releasing neurons, is driven by the gradients of Na +, K +, and Cl -, and is sensitive to antidepressants like desipramine and reboxetine. The second uptake system is located at the plasma membrane of non-neuronal effector cells of sympathetically innervated organs. It supplies the intracellular metabolizing enzymes, i.e. monoamine oxidase and catechol-o-methyl transferase, with catecholamines. This carrier, now called extraneuronal monoamine transporter or EMT, has been detected functionally in many sympathetically innervated tissues, e.g. myocardium, smooth vascular muscle, and salivary gland. EMT supposedly limits the spreading of transmitter in organ space and removes circulating catecholamines from the blood. Because of the link between cardiovascular disorders and sympathetic overstimulation, adrenoceptors continue to receive intense scrutiny. However, much less is known about the pathophysiological involvement of the Fig. 1: Noradrenaline transporters. Work from our group In the past, we have established new tools to analyze extraneuronal monoamine transport, i.e. (i) the first cell culture model (Caki-1 cell line), (ii) an efficiently transported, non-metabolized substrate (1-methyl- 4-phenyl pyridinium, MPP + ), and (iii) specific, highaffinity inhibitors (iso- and pseudoisocyanines, e.g. disprocynium24). With the latter we have demonstrated in the conscious rat model that non-neuronal monoamine transport is principally important for the inactivation of circulating catecholamines. Moreover, we have succeeded to elucidate the primary structure of EMT by targeted molecular cloning from human Caki-1 cells. EMT from human (EMTh) consists of 556 amino acids and is predicted to form 12 transmembrane segments. It is a member of the ASF (amphiphilic solute facilitator) transporter family (gene symbol group SLC22). We 28
34 Research Area A have studied, through heterologous expression in 293 cells, functional characteristics, substrate specificity, and pharmacological profile of EMT from human, rat and mouse, and we are analyzing localization and gene structure. Since EMT (gene symbol SLC22A3) represents an interesting candidate gene for a number of diseases, we have started to analyze genetic variability in humans. By direct sequencing of all 11 EMT exons and the putative core promoter, we have come upon a limited number of SNPs (single nucleotide polymorphisms) including a single amino acid variant (1110G>T resp. Met370Ile). The latter has been characterized as part of the CMMC project. Goals In cardiovascular disease, high levels of circulating catecholamines exacerbate pathophysiology and impair prognosis. EMT is a key system for the inactivation of catecholamines in the cardiovascular system. Thus, in order to assess the significance of EMT in disorders of the cardiovascular system and the potential for pharmacological intervention, it is essential to acquire detailed knowledge on the structure and function of this transport system. Following molecular cloning and functional characterization of EMT in vitro, we now explore its physiological function and regulation. The current project has two major goals: Targeted inactivation of EMT in the mouse model We expect important insights into the physiological role of EMT to emerge from a knock-out mouse. The group of D. Barlow has generated a knock-out of exon 1, but there is some doubt whether EMT is totally inactive in this mouse. We have therefore set out to create a knock-out of exons 6 and 7. With the homozygous animals, we will analyze the consequences of EMT ablation in a set of cardiovascular paradigms. Molecular mechanism of transporter activation We have made the puzzling observation that the extraneuronal monoamine transporter from rat (EMTr), when heterologously expressed in human embryonic kidney 293 cells, is about two orders of magnitude more resistant to most inhibitors including disprocynium24 and corticosterone than the human orthologue EMTh. Strikingly and despite the high similarity of primary structures, inhibitors and substrates at low concentration stimulate transport above control levels only with EMTr, but not with EMTh (Fig. 2). To explain this activation phenomenon, we have proposed a kinetic model which assumes two binding sites for substrate or inhibitor per transporter entity. Our sequence analyses and pilot experiments suggest that different C-termini are responsible for differential activation. In fact, we have noticed that all EMT orthologs carry PDZ ligand motives, and we now favour the notion of a PDZ-protein-provided cross-link of carrier units. It is our aim to identify the molecular details of transporter activation by targeted exchange of C-terminal peptides between EMTh and EMTr and functional characterization of these chimera. Fig. 2: Inhibition by compound DMPBI of MPP + uptake into 293 cells expressing EMTr or EMTh. Perspectives Generation of an EMT-knock-out mouse provides us with a basis for in vivo analysis of the physiological function of EMT. With NET still active at the releasing neurons, the functional consequences of a lack of EMT may not be instantly apparent. We are confident, however, that by putting stress on the heart and circulation, we will see differences between mutant and wild-type. To fully understand EMT activity, it is necessary to analyze tissue-specific modulation by other proteins. If we succeed to demonstrate that the C-terminus of EMTr is indeed responsible for activation, then we have to identify the corresponding binding partner. Eventually, activation of human EMT by pharmacological means would help to reduce levels of circulating catecholamines and thus prevent the associated toxic effects. Selected publications Gründemann D, Schechinger B, Rappold GA, Schömig E: Molecular identification of the corticosterone-sensitive extraneuronal monoamine transporter. Nature Neurosci. (1998) 1, Gründemann D, Koschker A-C, Haag C, Honold C, Zimmermann T, Schömig E: Activation of the extraneuronal monoamine transporter (EMT) from rat expressed in 293 cells. Br. J. Pharmacol. (2002) 137, Haag C, Berkels R, Gründemann D, Lazar A, Taubert D, Schömig E: The localisation of the extraneuronal monoamine transporter (EMT) in rat brain. J. Neurochem. (2004) 88, Schömig E, Lazar A, Gründemann D (2006) Extraneuronal Monoamine Transporter and Organic Cation Transporters 1 and 2 - A Review of Transport Efficiency. In: Handbook of Experimental Pharmacology - Neurotransmitter Transporters (Sitte HH & Freissmuth M, eds), pp Springer, Heidelberg. 29
35 Research Area A A 8 Switch-on and -off Kinetics of Troponin (Tn) in Cardiac Myofibrils and the Effect of Familial Cardiomyopathy-Linked Mutations of Tn on these Kinetics Robert Stehle / Gabriele Pfitzer Institute for Vegetative Physiology Familial hypertrophic cardiomyopathy (FHC) is linked to mutations in Tn. An early hallmark of FHC is diastolic dysfunction. To gain insight how these mutations impair cardiac relaxation we have developed a method which allows us to determine the kinetics of the Ca 2+ -controlled regulatory conformational change (switch) of cardiac Tn within its sarcomeric environment. We could demonstrate that each of six investigated FHClinked mutations lead to a slowdown of switch-off-kinetics. Because FHC is also associated with an alteration of the Tn-phosphorylation we want to investigate how this interacts with the switch kinetics. Introduction Point mutations in sarcomeric proteins like cardiac Troponin (Tn) have been linked to FHC. Among others symptoms FHC is associated with diastolic dysfunction and sudden cardiac death. Tn regulates contraction of the myocardium by the binding of Ca 2+. Tn is composed of three subunits: the Ca 2+ -binding TnC, the inhibitory TnI, and the Tropomyosin-binding TnT. Ca 2+ -binding to TnC leads to a conformational change (switch) which is translated to tropomyosin via TnI and TnT. Now tropomyosin uncovers the myosin-binding sites on actin and myosin crossbridges interact with actin in a strongly-bound, force generating manner. Up to now, it is not known whether the kinetics of the Tn-switch ratelimit the force development or relaxation. It is also not known whether the Tn-mutations linked to FHC cause the diastolic dysfunction because of a direct effect of the mutations on the switch kinetics of Tn or whether the symptom is due to secondary, maladaptational effects. Characterization of the switch-on and -off kinetics of ctn In this project we developed a novel technique which allows us to study the kinetics of the Ca 2+ -induced switch of Tn in its sarcomeric environment. For this purpose we labelled human Tn selectively with a dye which changes its fluorescence properties in response to the switch. To provide the sarcomeric environment we incorporated the labelled Tn into MF by exchanging the endogenous Tn against it. Kinetics of the fluorescence changes of labelled Tn were measured with stopped-flow-technique by mixing the MF with different [Ca 2+ ] or a Ca 2+ -buffer to either induce the switch-on or the switch-off of the Tncomplex. Fig.1: Ca 2+ -controlled kinetics of Tn-switch and force. A switch-on kinetics of isolated Tn and Tn in MF. B Ca 2+ -dependence of switch-on of incorporated Tn. MF were mixed at time = 0s and fluorescence traces were fitted monoexponentially. C Ca 2+ - dependence of rate-constants of switch-on and contraction. D Kinetics of switch-off and relaxation after Ca 2+ -removal at time = 0s. At saturating pca4.6 fluorescence increases biphasically in myofibrils. The first phase has a rate constant of > 2000s -1 and is likely to represent the fast Ca 2+ -binding. The second phase has a rate constant of 330s -1 and is likely to be the regulatory switch. Switch-on kinetics of Tn incorporated in MF are ~4-fold faster than those of isolated Tn (Fig.1A), showing that previous measurements on isolated Tn do not represent the kinetics of Tn as in the muscle. Fig. 2: Modelling of Ca 2+ -binding to Tn. A 3-state model of Ca 2+ -binding; * indicates fluorescent states of Tn. B Simulation of A at different pca. 30
36 Research Area A Detailed analysis gave the 3-state model described in Fig.2A. Simulation of this model yielded traces (Fig.2B) which describe the experimentally obtained traces in close agreement (Fig.1B) thus supporting the kinetic constants of the model. To find out whether the switch kinetics rate limit force the switch kinetics were compared with parallel measurements of myofibrillar contraction. Even at very low Ca 2+ -activations the Tn-switch turned out to be still ~50- fold faster than contraction (Fig.1C). Relation of the switchoff kinetics (koff=38s -1 ) with kinetics of relaxation showed that the switch-off is ~5-fold faster (Fig.1D). Thus we have shown that neither kinetics of contraction nor relaxation is rate limited directly by the switch of Tn. Effect of FHC-linked mutations on switch-kinetics of Tn To investigate whether FHC-linked TnI-mutations could directly cause diastolic dysfunction we determined their effect on switch-off kinetics of Tn. 40±1s -1 ) nor on the pca 50 (6.3±0.1 vs 6.3±0.1) or to WT-Tn, although the neighbouring FHC-mutation R21C induces a significant slowdown. Also measurements on isolated Tn showed no significant effect excluding that the environment of the guinea pig MF abolishes the effects in an unknown way. Perspectives For fully characterization of the different FHC-linked mutants we also want to determine their on-kinetics. To exclude that the lack of effect with the phosphorylation could be due to the use of S23/24D-Tn, we want to measure the switch-kinetics with PKA-phosphorylated Tn and with labelling site in TnI (S149C). Thereafter we want to investigate the crosstalk between the effects of phosphorylation and FHC-linked mutations. Selected publications Kruger M, Zittrich S, Redwood C, Blaudeck N, James J, Robbins J, Pfitzer G, Stehle R. Effects of the mutation R145G in human cardiac troponin I on the kinetics of the contraction-relaxation cycle in of the contraction-relaxation cycle in isolated cardiac myofibrils. J Physiol. (2005) 564, Poggesi C, Tesi C, Stehle R. Sarcomeric determinants of striated muscle relaxation kinetics. Pflugers Arch. (2005) 449, Stehle R, Solzin J, Iorga B, Gomez D, Blaudeck N, Pfitzer G. Mechanical properties of sarcomeres during cardiac myofibrillar relaxation: stretch-induced cross-bridge detachment contributes to early diastolic filling. J Muscle Res Cell Motil. (2006) 27, Fig. 3: Location of FHC-linked mutations of TnI and their effect on switch-off kinetics. A yellow: TnC, green: TnI, red: TnT, blue: tropomyosin, grey: actin Fig. 3B illustrates that all mutations lead to a significant slow-down of the switch-off - especially the ones at the important regulatory and inhibitory domain and next to the PKA phosphorylation site. Thus the data suggest that the slowed down kinetics could contribute at least indirectly to impairment of cardiac relaxation kinetics and thus to diastolic dysfunction. Stehle R, Iorga B, Pfitzer G. Calcium regulation of troponin and its role in the dynamics of contraction and relaxation. Am J Physiol Regul Integr Comp Physiol. (2007) 292, Neulen A, Blaudeck N, Zittrich S, Metzler D, Pfitzer G, Stehle R. Mn 2+ -dependent protein phosphatase 1 enhances protein kinase A-dependent Ca 2+ -sensitisation in skinned murine myocardium. Cardiovasc research (2007) 74, PKA-dependent phosphorylation of Tn There is evidence that phosphorylation of TnI is involved in pathogenesis of FHC. Hence, we wanted to investigate whether PKA-dependent phosphorylation of Ser23/24 of TnI changes the switch-kinetics of Tn and thus could contribute to impaired cardiac relaxation. According to this we first had to characterize the effect of phosphorylation itself. Therefore we mimicked phosphorylation by using a S23/24D-mutant of TnI and compared its kinetics with the one of an unphosphorylated S23/24A-mutant. Surprisingly, compared to S23/24A-Tn, S23/24D-Tn shows neither an effect on the switch-off kinetics (37±3s -1 vs 31
37 Research Area A Junior Research Group (NG) V Molecular Pathogenesis Of Arrhythmogenic Diseases Bernd Wollnik Hosted by the Institute for Human Genetics The aim of our research group is to analyse the molecular pathogenesis of cardiac disorders associated with arrhythmias by combining interdisciplinary research fields such as clinical science, molecular genetics, bioinformatics, molecular biology, and physiology. The identification and functional analysis of disease causing, predisposing and modifying genetic factors responsible for inherited forms of cardiac disorders will find its application in new molecular genetic tests, genetic counselling services, development of new therapeutic strategies. In addition, our group has a great interest in human genetics and therefore, we are working on the identification of disease-causing genes associated with various other phenotypes. Introduction Cardiac disorders associated with arrhythmias are a leading cause of morbidity and mortality. Recent advances in molecular genetics have led to the identification of the first genes responsible for some types of arrhythmias, e.g. ion channel genes in the long QT syndrome. Alterations of cardiac excitability as a manifestation of a primary electrical abnormality are known in different idiopathic forms of cardiac arrhythmias. Furthermore, arrhythmias may arise on the basis of structural heart diseases (e.g. as seen in patients with cardiomyopathy), coronary heart disease and myocardial infarction. The aim of the research group is to analyse the molecular pathogenesis of cardiac arrhythmias and other inherited disorders by combining clinical science, molecular genetics, bioinformatics, molecular biology, and physiology. Our work programme consists of: (I) ascertainment of well-characterized families and sporadic patients; (II) identification of the molecular defects in families with Mendelian inherited diseases by systematic genomewide mapping efforts and candidate gene approaches (fig. 1); (III) functional characterization of novel genes and their products; (IV) detailed analysis of physiological and pathophysiological mechanisms. Autosomal recessive ARVC Arrhythmogenic right ventricular cardio-myopathy (ARVC) is a heart muscle disease that causes arrhythmias, heart failure, and sudden cardiac death. The transmission of the non-syndromic form of the disease is autosomal dominant. Furthermore, a syndromic form of ARVC exists, called Naxos disease. Mutations in desmosomal components have been identified in some families and sporadic patients with ARVC. Desmosomes are cellular junctions with the dual function of intercellular adhesion and anchoring of the intermediate filament cytoskeleton to the cell membrane. We ascertained a large Turkish family with an autosomal recessive, non-syndromic form of ARVC and found statistically significant linkage to a chromosomal region and refined the critical interval. Efforts are continuing to identify the disease-causing gene in this region. In addition, we identified a second locus for recessive ARVC in another consanguineous family. CPVT and ARVC A specific form of ARVC associated with catecholamineinduced polymorphic ventricular (CPVT) arrhythmias has been described and dominant mutations in the cardiac ryanodine- receptor-2 gene (RYR2) were identified disturbing the calcium handling in the sarcoplasmic reticulum of cardiac myocytes. We have now clinically characterized a family with a CPVT/ARVC phenotype. Mutation screening of genes involved in cardiac calcium homeostasis identified for the first time a heterozygous mutation in the CASQ2 gene for this phenotype. Functional studies of the mutation showed a calcium leak of the sarco-plasmatic reticulum explaining the patho-physiological mechanism of the disease. Fig. 1: Example of mapping a locus for an autosomal recessive form of cardiac arrhythmia: a) pedigree and haplotype analysis; b) ECG of an affected family member; c) statistical analysis and chromosomal localization of linked genetic markers. 32
38 Research Area A Novel Naxos-like syndromes Loss of junctional integrity by mutations in desmosomal proteins causes skin, hair and heart defects in humans. A 2-bp deletion in the plakoglobin gene (JUP, 17q21) causes autosomal recessive Naxos disease characterized by ARVC, epidermolytic palmplantar keratoderma, and woolly hair. Recessive mutations in desmoplakin gene (DSP, 6p24) have been associated with Carvajal syndrome that presents with dilated left ventricular cardiomyopathy, striate palmoplantar keratoderma, and woolly hair. We studied a family with a novel Naxos-like syndrome with a severe ARVC and an early onset and rapid progression of the disease. A homozygous mutation was found in the DSP gene that results in the loss of one DSP isoform. Immunohistochemistry on biopsy material confirmed these results. Further molecular analysis of other Naxos-like families is currently performed and we have evidence for further genetic heterogeneity. Molecular basis of the Levi-Hollister syndrome The Levi-Hollister syndrome, also known as LADD syndrome, is a multiple congenital anomaly mainly characterized by lacrimal duct aplasia, malformed ears and deafness, small teeth, and digital anomalies. An association with cardiac arrhythmias (LQT syndrome) was described in a family with LADD syndrome. We mapped the LADD1 locus in three large families to chromosome 10q26 and identified heterozygous missense mutations in the tyrosine-kinase (TK) domain of the fibroblast-growthfactor-receptor 2 (FGFR2). After exclusion of FGFR2 in two additional LADD families we found a causative mutation in the tyrosine-kinase domain of FGFR3 in one family and a missense mutation in the gene encoding fibroblast-growth-factor 10 (FGF10), a known FGFR ligand, in the other family. Previous studies have shown that different craniosynostosis syndromes are caused by gain-of-function mutations in FGFR2/3, which result in elevated tyrosine kinase activity of FGFR2/3. A reduced functional activity of FGFR2/3 LADD mutations seems to be an attractive and plausible mechanism underlying the molecular basis of LADD syndrome. Using transient and stable FGFR2 expression studies, we are analyzing the activation properties of the receptor as well as the phosphorylation efficiency of know substrates. We hope that the results will give us new insights into mechanisms of FGF signaling and the pathophysiology of LADD syndrome. Furthermore, additional genes causing LADD syndrome are currently investigated. Perspectives The extraordinary clinical and genetic heterogeneity of inherited disorders requires the development of new and complementary strategies in order to effectively and successfully undertake the approach to analyse the molecular pathogenesis of these diseases. Therefore, our research group combines both, the classical strategy of gene identification in large families with inherited disorders and system genetics in sporadic cases and small families. Fig. 2: The molecular basis of LADD syndrome. Typical ear and hand malformations are shown. The location of different mutations is marked by a red dot in the schematic FGFR2/3 receptor model and its ligand FGF10. The TK-domains are shown in orange. Selected publications Antzelevitch C, others, Wollnik B, others, Wolpert C: Loss-offunction mutations in the cardiac calcium channel underlie a new clinical entity characterized by ST-segment elevation, short QT intervals, and sudden cardiac death. Circulation (2007) 115, Rohmann E, others, Nurnberg P, Kubisch C, Schlessinger J, van Bokhoven H, Wollnik B: Mutations in different components of FGF signaling in LADD syndrome. Nat Genet (2006) 38, Uzumcu A, others, Wollnik B: Loss of desmoplakin isoform I causes early onset cardiomyopathy and heart failure in a Naxoslike syndrome. J Med Genet (2006) 43, e05. Kalay E, Li Y, others, Nurnberg P, others, Kubisch C, Kremer H, Wollnik B: Mutations in the lipoma HMGIC fusion partner-like 5 (LHFPL5) gene cause autosomal recessive nonsyndromic hearing loss. Hum Mutat (2006) 27, Tukel T, others, Hennies HC, Nurnberg P, Desnick RJ, Wollnik B. A new syndrome, congenital extraocular muscle fibrosis with ulnar hand anomalies, maps to chromosome 21qter. J Med Genet (2005) 42,
39 Research Area B B 1 Characterization of Melanoma-Stroma Interactions Cornelia Mauch / Thomas Krieg Department of Dermatology and Venerology Interactions between tumor cells and the surrounding stromal cells have an important impact on the invasion of tumor cells into the connective tissue. In melanoma, we could demonstrate that matrix degradation occurs at the tumor-stroma border and involves the release of cytokines and growth factors by melanoma which activate the stromal cells. Characterization of these interactions will allow to develop new therapeutic strategies to specifically target tumor or stromal cells. Introduction During invasion and metastasis of malignant melanoma tumor cells detach from the primary tumor, migrate and penetrate several structural barriers, e.g. basement membranes and invade the surrounding connective tissue of the dermis. Degradation of stromal ECM is also considered to be essential for tumor-induced angiogenesis. A series of recent studies revealed that tumor cells are not individual units within a tumor, but rather depend on the cross-talk with neighboring stroma cells and matrix components. Of particular importance are interactions between melanoma cells and neigh-boring fibroblasts and microvascular endothelial cells. During embryonic development various growth factors, cytokines, matrix proteins and proteinases were identified as crucial regulators of mesench-ymal-epithelial cross-talk. It can be assumed that similar mechanisms are also important in the regulation of tumor growth. Recent studies with stromaoriented cancer therapy were remarkably successful and are considered as a particularly promising novel approach for the treatment of malignancy. Nevertheless, the knowledge in this area is still rudimentary. Molecular analysis of melanoma-cell interactions in different culture systems and in mouse models In order to elucidate the role of direct or indirect interactions between human melanoma cells and human dermal fibroblasts, we cultured cells in different co-culture systems either in direct or indirect contact (transwell chambers). As compared to controls, we observed a strong induction of MMP-1 synthesis in both, direct or indirect co-culture conditions and by medium conditioned by the melanoma cells suggesting that soluble factors released by melanoma cells mediate this induction. In contrast, media collected from low invasive melanoma cells showed no effect. Using recombinant IL-1 receptor antagonist (IL-1A), and neutralizing antibodies against IL-1α and bfgf, we also showed that IL-1 and bfgf both of which are secreted in high amounts by the melanoma cells are involved in this induction while IL-6 and EGF were not. Interestingly, treatment of mice with IL-1RA significantly reduced tumor growth of intracutanously injected melanoma cells and inhibited metastasis to lymph nodes und lungs showing that also in vivo agents that interfere with melanoma-stroma interaction indeed inhibit tumor growth. Fig. 1: Influence of IL1-RA on tumor growth and metastasis To identify candidate genes which are regulated in tumor cells or host tissue by IL1-RA treatment we performed DNA array analysis of tumors, lymph nodes and lungs of animals after melanoma cell inoculation. Beside MMP-13, the murine orthologue of human MMP-1 genes of several melanoma-specific proteins, proliferation-associated proteins and pro-inflammatory cytokines and chemokines as well as matrix components were altered. Some of the changes suggested alterations in tumor angiogenesis. Preliminary data show that vessel density in the peritumoral stroma and intratumoral lymphangiogenesis are affected by IL1-RA treatment. In addition we could show that high but not low invasive melanoma induce tube formation of microvascular endothelial cells in vitro. This induction could also be induced by incubating microvascular endothelial cells with supernatants collected from melanoma cells. Several secreted mediators were identified to be released by high but not invasive melanoma cells. VEGF-A was identified as the most important factor for the induction of tubulogenesis since this process was completely abolished in the presence of neutralizing antibodies. 34
40 Research Area B Perspectives It is well known that therapy of malignant tumors often hampers by the development of resistency to chemotherapeutical agents. Therefore agents which interfere with tumor-stroma cross-talk may become important features in the development of new therapies. Neutralizing antibodies and/or cytokine receptor antagonists, reduction of cytokine release as well as delivering of effector molecules by direct targeting the peritumoral environment e.g. by recombinant soluble fusion proteins. In addition, by inhibition of de novo synthesis of these factors we will analyze whether invasion of melanoma is inhibited. The role of stromal cells-derived MMP-13 and MT1-MMP will be further characterized using animals with inactivation of these enzymes. The characterization of the underlying mechanisms will allow to develop new therapeutic strategies to specifically target tumor or stromal cells. Fig. 2: Melanoma-induced tube-like formation in vitro is mediated by VEGF-A Role of MMP-13 in tumor invasion and metastasis In mice collagenolysis is believed to involve MMP-13 and possibly MT1-MMP both of which are induced by cytokines e.g. IL-1. In order to prove the impor-tance of the induction of MMP-13 by melanoma cells directly observed in murine fibroblasts in vitro we established melanoma cell invasion model using various mouse strains. To facilitate detection of melanoma metastases in mice we stably transfected murine B16 melanoma cells by the CMV-GFP gene and injected these cells intradermally into the back of the mice. Pigmented tumors developed within 5 to 10 days. Regional lymph nodes, and different organs (lungs, kidney, heart, liver, brain) were prepared. In mice with complete inactivation of MMP-13 melanoma cells metastasized less frequently into lung and lymph nodes and surprisingly no metastasis into the heart while in wildtype animals heart metastasis was detected in 50% of the animals. Selected publications Klose A., Wilbrand-Hennes A, Zigrino P, Weber E, Krieg T, Mauch C, Hunzelmann N. Identification and discrimination of extracellularly active cathepsins B and L in high-invasive melanoma cells Anal Biochem (2006) 353, Rhiemeier V, Breitenbach U, Richter KH, Gebhardt C, Hartenstein B, Fürstenberger G, Mauch C, Hess J, Angel P. A novel aspartic proteinase-like gene expressed in stratified epithelia and squamous cell carcinoma of the skin. Am J Pathol (2006) 168: Chometon G, Rubinstein E, Mauch C, Aumailley M. Dissociation of the complex between CD151 and laminin-binding integrins induces migration of epithelial cells. Exp Cell Res (2006) 312, Billion H, Ibrahim H, Mauch C, Niessen C. Increased soluble E-cadherin in melanoma patients. Skin Pharmacol Physiol (2006) 19, Löffek S, Zigrino P, Steiger J, Kurschat P, Smola H, Mauch C. Melanoma cell derived VEGF induces endothelial tubulogenesis within fibrin gels by a metalloproteinases mediated mechanism. Eur J Cell Biol (2006) 85, Braun S, Mauch C, Boukamp P, Werner S. Novel roles of NM23 proteins in skin homeostasis, repair and disease. Oncogene (2007) 26, Kurschat P, Nashan D, Krieg T, Mauch C. Early increase in serum levels of the angiogenesis-inhibitor endostatin and of basic fibroblast growth factor in melanoma patients during disease progression. Br J Dermatol (2007) 156, Fig. 3: Tumor growth and metastasis of melanoma in MMP-13 -/- mice 35
41 Research Area B B 2 The Consequences of Mutations of Genes that Contribute to Basement Membrane Function Neil Smyth / Mats Paulsson Institute for Biochemistry II The laminins and nidogens are the main protein families present in basement membranes. We have produced knockout mice lacking either the laminin γ1 chain or the nidogen isoforms. In these models we study the role of the basement membrane, of the individual proteins, and the significance of their interaction in development. In the past two years the particular emphasis has been on the role of nidogen-1 and -2 in limb development and in the formation of the central nervous system. Introduction The basement membrane is a specialised extracellular matrix structure contacting many cell and tissue types. Basement membranes have physical and signalling functions that alter with the tissue and stage of development. In addition to forming surfaces to which cells attach, they also transmit force between the cell and the extracellular matrix. Basement membranes also act as barriers, for instance in the kidney the highly specialised glomerular basement membrane is an important filter for plasma proteins. Elsewhere they define the structural limits of many tissues, for example the basement membrane of the pia or at the dermo-epidermal junction. Basement membranes may also regulate many aspects of intracellular activity by signalling through cellular receptors of the integrin, dystroglycan and syndecan families and, finally, act as a reservoir for cytokines and growth factors. Hence basement membrane induced signalling plays a highly important role in cell survival and differentiation as well as in providing cell and axonal migration cues. Analysis of mice lacking expression of one or both nidogen genes Mice null for nidogen-1 show little phenotypic alteration. Most basement membranes appear unaltered and there is little evident change in cellular or tissue morphology (Murshed et al., 2000). However, they do show neurological deficits, in particular a hindlimb ataxia and spontaneous seizure activity. While nidogen-1 is found in all basement membranes, the expression of its homologue nidogen-2 is more limited, being found in only trace amounts in the basement membranes of striated muscles. In the nidogen-1 -/- animals nidogen-2 is increased in these tissues and shows redistribution in its staining pattern suggesting that it may be compensating for the loss of nidogen-1. A detailed analysis of skeletal muscle fibres showed no evident morphological changes. However, there is a gradual loss of myelinated axons and signs of axonal degeneration, although this is probably not sufficient to explain the pronounced changes in locomotion as sciatic nerve conduction velocities are maintained. While nidogen-1 and -2 show a divergent expression pattern in certain adult tissues, both have a similar distribution during development. Gene knockout studies in mice demonstrated that the loss of nidogen-2, similar to nidogen-1, has no drastic effect on basement membrane formation and organ development, suggesting that these proteins have complementary functions. Deficiency of both nidogens in mice resulted in perinatal lethality (Bader et al., 2005). Nidogen-1 and -2 do not appear to be crucial in establishing tissue architecture during organ development; instead, they are essential for late stages of lung development and for maintenance and/or integrity of cardiac tissue. These organ defects are not compatible with postnatal survival. Ultrastructural analysis suggests that the phenotypes directly result from basement membrane changes. However, despite the ubiquitous presence of nidogens in basement membranes, defects do not occur in all tissues or in all basement membranes, suggesting a varying spectrum of roles for nidogens in the basement membrane. Limb development requires the complex interplay of numerous growth factors whose expression is dependent upon the apical ectodermal ridge. The lack of nidogen-1 and -2 results in a specific and time-limited failure in the ectodermal basement membrane of the limb bud (Böse et al., 2006). The absence of this basement membrane leads to aberrant apical ectodermal ridge formation. It also causes altered distribution of growth factors, such as fibroblast growth factors and leads to a fully penetrant soft tissue syndactyly caused by the dysregulation of interdigital apoptosis. Further, in certain animals more severe changes in bone formation occur, providing evidence for the interplay between growth factors and the extracellular matrix. Nidogen-1 appears to subserve a function in neuronal plasticity. Nidogen-1 ablation leads to epileptic activity in vivo and the appearance of spontaneous epileptiform activity in vitro (Kohling et al., 2006). This raises the intriguing question whether nidogens may directly influence neuronal function in the CNS, opening the possibility of modulatory mechanisms of synaptic plasticity and excitability reaching beyond classical processes confined to cellular interactions. In a search for 36
42 Research Area B mechanisms leading to the neurological phenotype we have analysed brain development in nidogen-1 /-2 double knockout mouse embryos. These show disturbances in cerebral layering which are probably due to a loss of attachment of radial glia cells to the basement membrane at the cerebral surface. Because of this migration of neuronal precursor cells is retarded. In double deficient mice many basement membranes are poorly formed and, in addition to nidogens, laminins are lost from the basement membrane structures, presumably due to the lacking anchorage to collagen IV through nidogen. A surprising finding is that despite basement membrane disruptions blood vessels are formed and the blood-brain barrier appears intact. Perspectives Our studies will reveal the role of nidogens in the formation of the central nervous system and also lead to elucidation of the mechanism through which the lack of nidogen-1 causes epilepsy. As mutations in genes coding for basement membrane proteins often lead to human disease, the phenotypes found in nidogen deficient mice may help identifying patients suffering from inherited nidogen changes. Fig. 2: Dual immunofluorescent staining for basement membrane and endothelial cells in E12.5 mouse brains Coronal sections of wt (a, f), nid1 -/- (b, g), nid2 -/- (c, h), nid1 +/- ;nid2 -/- (d, i) and nid1 -/- ;nid2 -/- (e, j) at E15.5 were stained for basement membrane markers (laminin γ1 chain, panels a-e; laminin α4 chain, panels f-j, green) and an endothelial cell marker (PECAM-1; red). The capillary basement membrane staining intensity decreased with the absence of nidogen-2 protein. There is virtually no capillary basement membrane structure in the nid1 -/- ;nid2 -/- cerebral cortex; however, the formation of blood vessels and capillaries appears not to be affected. Selected publications Böse K, Nischt R, Page A, Bader BL, Paulsson M, Smyth N. Loss of nidogen-1 and -2 results in syndactyly and changes in limb development. J Biol Chem (2006) 281, Bader BL*, Smyth N*, Nedbal S, Miosge, N, Baranowsky A, Mokkapati S, Murshed M, Nischt R. Compound genetic ablation of nidogen 1 and nidogen 2 causes basement membrane defects and perinatal lethality in mice. Mol Cell Biol (2005) 25, *equal contribution Fig. 1: Dual immunofluorescent staining for the laminin γ1 chain and the glia markerrc2 in E15.5 mouse brains Coronal sections of wt (a), nid1 -/- (b), nid2 -/- - (c), nid1 +/- ; nid2 -/- (d) and nid1 -/- ; nid2 -/- (e) at E15.5 were stained for both a basement marker (laminin γ1 chains; red) and a radial glial marker (RC2; green). The pial basement membrane structure is absent at the cortical surface of the nid1 -/- ; nid2 -/- brain and the extension of the radial glia is affected. However, subtle changes in the radial glia extension are also noted in mutant brains lacking nidogen-2 (c, d), even when the pial basement membrane is present. Kohling R, Nischt R, Vasudevan A, Ho M, Weiergräber M, Schneider T, Smyth N. Nidogen and nidogen-associated basement membrane proteins and neuronal plasticity. Neurodegener Dis (2006) 3, Murshed M, Smyth N, Miosge N, Karolat J, Krieg T, Paulsson M, Nischt R. The absence of nidogen 1 does not affect murine basement membrane formation. Mol Cell Biol (2000)
43 Research Area B B 3 Role of Papillomaviruses in Non-Melanoma Skin Cancer: Viral Oncoproteins and UV-Light as Cofactor Herbert J. Pfister Institute for Virology This project deals with the role of human papillomaviruses (HPV) in the development of non-melanoma skin cancer (NMSC). We established transgenic mice, which express the E2, E6 and E7 genes of HPV8 separately under the control of the human keratin 14 promoter. In mice, HPV8-E6 is the major oncogene, necessary and sufficient for carcinogenesis. HPV8-E2 on its own is a weaker oncogene. UV-light plays a synergistic role in HPV8 initiated oncogenesis Introduction HPV are well known tumor viruses, which induce a spectrum from benign to (pre)malignant lesions of the cutaneous and mucosal epithelia. The association between HPV and skin cancer was first identified in patients with the rare, inherited disorder epidermodysplasia verruciformis (EV). In contrast to the plurality of EV-associated HPV in benign tumors, only few virus types, predominantly HPV5 or 8 are found in the cancers and are regarded as high risk types. Recent studies have shown that EV-HPV are not restricted to EV-patients but are also found in NMSC of the general population. By generating transgenic mice with the complete early region (CER) of HPV8 under control of the human keratin-14 promoter, which activates the expression of viral genes in the basal keratinocytes, we could demonstrate the oncogenic capacity of HPV8 in vivo (Schaper et al., 2005). To dissect the role of the early genes in tumor development we generated transgenic mice, who express the HPV8-E2, -E6 and E7 genes separately. HPV8-E2, -E6 and -E7 transgenic mice The E6 protein alone consistently induced singular or multifocal tumors without any further treatment with chemical or physical carcinogens. These tumors were characterized by papillomatosis, acanthosis, hyperkeratosis and varying degrees of epidermal dysplasia. In 6% of the cases the tumors became malignant, comparable with the results of the HPV8-CER mice (Fig. 1). It was surprising to see that the separate expression of HPV8-E2 also induced partially dysplastic papillomas. However, tumor development took about 6 times as long as in E6 transgenics, only 60% to 80% of the animals were affected, and no cancers were observed (Fig. 1). None of the HPV8-E7 positive mice developed skin tumors after an observation period of up to 130 weeks. The presence of E7 mrna was confirmed by quantitative real time RT-PCR with similar levels as in HPV8 transgenic mice with all the early genes. UV light activates EV-HPV promoters The UV component of sunlight is the best established physical carcinogen for the development of human NMSC. To determine whether UVB irradiation modulates the transcriptional activity of EV-HPV-types we performed transient transfection assays in primary human keratinocytes Fig. 1: Time course of tumor development in different generations of three HPV8 mouse lines: HPV8-CER line 9 FVB/N, HPV8-E6 Line 637 FVB/N and HPV8-E2 line 2. Mice were checked every 3 days to monitor the development of skin tumors. with luciferase reporter gene constructs driven by the viral promoters. Various EV-HPVs showed different basal promoter activity and reacted differently upon UVB treatment. The high-risk cutaneous HPV types 5 and 8 were the only ones to be activated by UVB, which may be relevant to the development of NMSC (Akgül et al., 2005a). UV irradiation of HPV8 transgenic mice In order to evaluate synergistic effects of UV light on tumor formation in HPV8 transgenic animals, the shaved skin was irradiated with UVA and UVB. Immunhistochemical stainings of p53 showed similar levels of p53 in the epidermis of HPV8 positive and negative mice after UV irradiation. A single treatment was sufficient to induce papillomatosis in HPV8-CER positive FVB/N and BL6 mice after 3 weeks (Fig. 2). In contrast the irradiated skin of the HPV8 negative littermates healed completely within that time. The same development of papillomas was observed with 38
44 Research Area B HPV8 CER + HPV8 CER - 4 days 21 days Fig. 2: UVA/B irradiation of an HPV8-CER FVB/N mouse and an HPV8 negative littermate at the age of 3 weeks with 10J/cm 2 UVA and 1J/cm 2 UVB. After three weeks the transgene positive mouse developed papillomas on the entire irradiated area, whereas the irradiated skin of the HPV8 negative littermate healed completely within that time. UV irradiated HPV8-E6 mice while HPV8-E7 mice did not develop any skin lesions. The irradiation with UVA alone did not have an inducing effect and the irradiation with UVB alone led to a weak induction of papillomatosis. Histology revealed no differences between UV induced and spontaneous tumors, which appeared later on non-irradiated skin. UV-irradiated E2 mice developed papillomas within 3-4 weeks and in one case a squamous cell carcinoma after 3 months. Perspectives The single irradiation of HPV8 transgenics with an erythemal dose had a dramatic effect on papilloma induction but usually did not affect tumor progression. To address a synergism of UVB and HPV infection in malignant conversion, young mice will be irradiated with multiple sub-erythemal doses and tested for DNA damage and apoptosis/ proliferation. By vaccination of non-transgenic animals we will try to inhibit the growth and proliferation of tumors in transplanted skin from transgenic litter-mates. Selected publications Schaper ID, Marcuzzi GP, Weissenborn SJ, Kasper HU, Dries V, Smyth N, Fuchs P, Pfister H. Development of skin tumors in mice transgenic for early genes of human papillomavirus type 8. Cancer Res (2005) 65, Akgül B, Lemme W, Garcia-Escudero R, Storey A, Pfister HJ. UV-B irradiation stimulates the promoter activity of the high-risk, cutaneous human papillomavirus 5 and 8 in primary keratinocytes. Arch Virol (2005a)150, Akgül B, Garcia-Escudero R, Ghali L, Pfister HJ, Fuchs PG, Navsaria H, Storey A. The E7 protein of cutaneous human papillomavirus type 8 causes invasion of human keratinocytes into the dermis in organotypic cultures of skin. Cancer Res (2005b) 65, Akgül B, Pfefferle R, Marcuzzi GP, Zigrino P, Krieg T, Pfister H, Mauch C. Expression of matrix metalloproteinase (MMP)-2, MMP- 9, MMP-13, and MT1-MMP in skin tumors of human papillomavirus type 8 transgenic mice. Exp Dermatol (2006)15,
45 Research Area B B 4 Modular Composition of Recombinant Immunoreceptors Hinrich Abken Department I of Internal Medicine Adoptive immunotherapy aims to redirect the cellular immune response specifically towards target cells. To graft cytolytic T cells with pre-defined specificity, we express by retroviral gene transfer a recombinant T cell receptor ( immunoreceptor ) that has an extracellular scfv antibody domain for antigen binding and an intracellular signaling domain for cellular activation. Our main focus during the last year was the establishment of a rational design for optimized immunoreceptors and the engineering of redirected T cells that are resistant to tumor mediated immunosuppression. Results provide the basis for the evaluation of the immunoreceptor strategy in a clinical trial. Introduction Cellular immunity plays a major role in the control of tumor growth. Cellular immunotherapy of malignant diseases has therefore attracted growing interest, particularly in order to target cytolytic T cells specifically towards tumor cells. The combination of T cell receptor (TCR) driven T cells with the targeting specificity of antibodies gave rise to chimeric receptor molecules (immunoreceptors) that can be used to engineer antigen-specific T cells. Modular composition of recombinant immunoreceptors The immunoreceptor strategy aims to generate antigenspecific T cells by expression of a recombinant immunoreceptor whose antigen binding domain is composed of an scfv antibody und whose intracellular signaling moiety of the CD3ζ chain for T cell activation (Fig. 1). The immunoreceptor concept has a number of advantages for use in adoptive immunotherapy (1), including the use of autologous effector cells, active tissue penetration of the engineered cells, MHC-independent binding to the target molecule, induction of complex immune reactions and self-limitation in the absence of antigen. Fig. 1: Modular composition of recombinant immunoreceptors. scfv, single chain fragment of variable region binding domain, TM transmembrane domain. The modular composition of the receptor molecule facilitates modification of both the antigen binding and signaling properties. Accordingly, we have generated immunoreceptors with specificities for a variety of tumor-associated antigens and with various intracellular signaling domains. Binding to antigen induces crosslinking of the recombinant immunoreceptor forming an immunological synapse complex (Fig. 2) and initiation of an antigen-specific immune response. Notably, engineered T cells break tolerance towards autologous tumor cells, as exemplarily demonstrated for CD30 + cutaneous lymphoma and CEA + colon carcinoma (2). Fig. 2: Clustering and synapse formation of recombinant immunoreceptors. GFP-linked immunoreceptors expressed on the surface of T cells are clustered by specific antigen (A) or randomly distributed by contact with irrelevant antigen (B). Receptor activated T cells secrete IFN-γ, proliferate and drive granzyme/perforin dependent cytolysis of antigenpositive cells. We identified several items that affect the efficacy of receptor mediated cellular activation, some of which are the binding affinity, the targeted epitope of the tumor associated antigen, the sensitivity of engineered T cells towards tumor induced immunosuppression as well as the sensitivity of tumor cells towards a cytolytic T cell attack. Affinity By means of a panel of immunoreceptors with different affinities targeting the same epitope of ErbB2, we demonstrated that immunoreceptors of higher affinity do not necessarily induce a more potent activation of T cells than low affinity immunoreceptors (3). Whereas upon binding to solid phase bound ErbB2 the T cell activation inverse- 40
46 Research Area B ly correlates with the binding affinity, upon binding to ErbB2 + cells, however, there is a clear activation threshold above that T cells are activated with the same kinetics. Notably, the higher affinity immunoreceptors exhibit less discrimination between target cells with high or low antigen expression levels making low affinity immunoreceptors more attractive for immunotherapy. Binding epitope We explored the impact of the target epitope on the efficacy of receptor-mediated T cell activation (4). Upon binding to isolated, solid phase immobilized CEA as target antigen, receptor-mediated T cell activation correlates with the binding efficiency, irrespectively of the targeted epitope. Upon binding to CEA expressed on the cell membrane, in contrast, the membrane proximal A3 epitope mediates more efficiently T cell activation than the distal N epitope, although the N epitope is bound with higher affinity. The N epitope when in a more membrane proximal position on the target cell, however, activated receptor grafted T cells with higher efficiency than in the distal position. The position of the targeted epitope has obviously major impact on the efficacy of T cell activation independently of the binding efficiency of the immunoreceptor. CD4 + cells as cytolytic effector cells Immune elimination of tumor cells requires the close cooperation between CD8 + cytotoxic T lymphocytes and CD4 + T helper cells. Expression of a recombinant immunoreceptor with antibody derived binding domain circumvents MHC class II-restriction. Human CD4 + T cells with a CEA specific immunoreceptor are activated upon binding to proliferate and secrete cytokines. Notably, redirected CD4 + T cells mediate cytolysis of targeted tumor cells independently of death receptor signaling, but mediated by perforin and granzyme. Recruiting CD4 + T cells substantially extends the power of the adoptive immunotherapy by both CD4 + cell help and cytolytic effector functions (5). Sensitizing tumor cells for a redirected T cell attack In collaboration with Kashkar and Krönke, we aimed to develop strategies to overcome the resistance of Hodgkin s lymphoma (HL) cells against T cell mediated cytolysis. In HL cells granzyme B induced mitochondrial release of pro-apoptotic Smac is blocked, which results in abrogation of CTL cytotoxicity. Cytosolic expression of recombinant Smac enhanced caspase activity and restored the apoptotic response of HL cells. Similarly, down-regulation of XIAP by RNA interference enhanced the susceptibility of HL cells for cytotoxicity by antigen-specific CTLs redirected by a recombinant immunoreceptor. XIAP targeting by Smac agonists or XIAP-siRNA sensitizes HL cells for a redirected cytolytic T cell attack (6). CD28 costimulation overcomes TGF-β mediated T cell repression Tumors are frequently protected from a T cell attack by an immunological barrier, key factors of which are IL-10 and TGF-β. Whereas IL-10 has no direct impact on redirected T cell effector functions, TGF-β represses antigen-triggered T cell proliferation but does not alter specific cytolytic activities or cytokine secretion. The TGF-β effect can be overcome by CD28 costimulation showing promise to break tumor associated immunosuppression and to increase efficacy of T cell based immunotherapy (7). Perspectives and clinical trial Our analyses have expanded the knowledge on the structural prerequisites of transmembrane binding-signaling molecules giving a rational to design recombinant immunoreceptor molecules for use in adoptive immunotherapy. One of the major steps in the near future will be the initiation of a clinical trial to explore safety issues of locally applied, engineered T cells grafted with a combined CD28-CD3ζ signaling immunoreceptor. Selected publications Abken H, Hombach A, Heuser C, Immune response manipulation: recombinant immunoreceptors endow T-cells with predefined specificity. Curr Pharm Des (2003) 9: Hombach A, Schlimper C, Sieves E, Frank S, Schild HH, Sauerbruch T, Schmidt-Wolf IGH, Abken H, A recombinant anti-cea immunoreceptor with combined CD3ζ-CD28 signalling targets T cells from colorectal cancer patients against their tumor cells. Gut (2006) 55, Chmielewski M, Hombach A, Heuser C, Adams GP, Abken H, T cell activation by antibody-like immunoreceptors: increase in affinity of the scfv domain above threshold does not increase T cell activation against antigen-positive target cells but decreases selectivity. J Immunol (2004) 173, Hombach A, Schildgen V, Heuser C, Finnern R, Gilham D, Abken H, T cell activation by antibody-like immunoreceptors: the position of the binding epitope within the target molecule determines the efficiency of activation of redirected T cells. J Immunol (2007) 178, Hombach A, Koehler H, Rappl G, Abken H, Human CD4 + T cells lyse target cells via granzyme/perforin upon circumvention of MHC class II-restriction by an antibody-derived immunoreceptor. J Immunol (2006) 177, Kashkar H, Seeger J-M, Hombach A, Deggerich A, Yazdanpanah B, Utermöhlen O, Heimlich G, Abken H, Krönke M, XIAP targeting sensitizes Hodgkin s lymphoma cells for cytolytic T cell attack. Blood (2006) 108: Koehler H, Kofler D, Hombach A, Abken H, CD28 costimulation overcomes TGF-β mediated repression of proliferation of redirected human CD4 + and CD8 + T-cells in an anti-tumor cell attack. Cancer Res (2007) 67,
47 Research Area B B 5 The Role of NFκB Dependent Epidermal- Dermal Interactions in the Pathogenesis of Inflammatory Skin Disease Ingo Haase Department of Dermatology and Venerology Chronic inflammatory skin diseases are very common and are accompanied by dramatic impairments of the quality of life in the affected individuals. Our work indicates that skin specific interactions between the epidermis and cell populations in the underlying dermis are relevant to the pathogenesis of these diseases. The goal of this project is to identify and analyse the mechanisms that govern and translate such interactions. Introduction Mice with epidermis specific deletion of IκB kinase 2 (IKK2), a component of the IκB kinase complex (IKK complex) and a key enzyme in the NFκB signaling pathway, develop an inflammatory- hyperpro-liferative skin disease. The skin changes observed are similar to the human skin disease psoriasis. We use these mice as a disease model for psoriasis to investigate mechanisms leading to inflammation and hyperproliferation. We are particularly interested in the interactions between different skin cell types: epidermal keratinocytes, the cells that form the outer layer of the skin (epidermis) and the various resident cells of the dermis, the inner skin layer. In addition, we have shown that mice with epidermis specific deletion of NEMO, another component of the IKK complex, suffer from dramatic, widespread skin inflammation. In contrast, epidermis specific deletion of IκB kinase 1, a third component of the complex, causes an impairment of the skin barrier function, but not skin inflammation. Interestingly, although deletion of IKK2 and NEMO both inhibit NFκB signaling, constitutive activaton of NFκB signaling in keratinocytes following epidermis specific deletion of IκBα is also sufficient to initiate an inflammatory- hyperproliferative skin disease. This indicates that a balance of signals regulating NFκB activity in the epidermis is essential for the maintenance of skin homeostasis. Keratinocytes can initiate psoriasis like skin disease Our previous results show that a single genetic modification which is restricted to keratinocytes can be sufficient to initiate a dramatic, generalized inflammatoryhyperproliferative skin disease. The extent and severity of the disease can be limited by deleting IKK2 in the epidermis in a locally restricted manner. We have achieved this by crossing mice homozygous for floxed IKK2 alleles to mice that express an inducible Cre recombinase construct under the control of the cytokeratin K14 promoter. The results of these experiments show that there is temporal and spatial correlation between IKK2 deletion and psoriasis like skin inflammation. In the context of recent results from other laboratories these findings highlight the role of the epidermis in the pathogenesis of skin inflammation. They also give a paradigm for the potential role of epithelia in the pathogenesis of organ inflammation, e.g. in the gut, the lungs or the urinary tract. Psoriasis like skin disease in the absence of αβ T lymphocytes In humans, T lymphocytes carrying the αβ type of the T cell receptor are widely believed to make an essential contribution to the development of psoriasis. Based on this hypothesis, therapeutics targeting T cells have been developed and tested in clinical studies. The results of these studies have recently revealed that T cell targeting drugs show only limited efficacy in the treatment of psoriasis. In mice with epidermis specific deletion of IKK2, elimination of αβ T cells does not lead to major changes in the disease phenotype, demonstrating that the presence of these immune cells is not necessarily required for the development of the condition. This is in contrast to mice deficient for IκBα, which show a psoriasis like skin phenotype that depends on the presence of T lymphocytes. These findings demonstrate that different molecular changes can lead to a psoriatic skin phenotype; they also have important implications for the design of future psoriasis treatments. Important role for Macrophages in psoriasis like skin inflammation To address the role of other inflammatory cells in the development of the skin disease we have selectively eliminated granulocytes and macrophages from the skin of mice with epidermis specific IKK2 deletion. Elimination of granulocytes was achieved by deleting beta2 integrin (CD18), an adhesion receptor essential for the migration of these cells. The inflammatory- hyperproliferative skin phenotype developed in the absence of granulocytes, indicating that their presence is not essential in the pathogenesis of the skin disease. On the contrary, elimination of skin macrophages by injection of clodronate liposomes caused a dramatic improvement oft the skin phenotype, indicating an important role for macrophages in its pathogenesis. Staining for surface markers on macrophages indicates that the activation of these cells does not follow the classical pathways of macrophage activation. This is in line with our results showing that elimination of the receptor for interferon gamma, an essential cytokine in the classical macrophage activation pathway, has no major effect on the development of the psoriasis like skin condition. 42
48 Research Area B Fig. 2: Expression of CD83 (green) in mice with epidermis specific deletion of IKK2 (right) and in control mice (left) seven days after birth. Nuclei are shown in red. Fig. 1: Injection of clodronat liposomes normalizes epidermal differentiation and prevents immigration of immune cells. H/E stainings and immunostainings (green) of sections of paraffin embedded skin with antibodies against the epidermal differentiation markers keratin 14 (K14), keratin 10 (K10), loricrin (Lor), filaggrin (Fil) or of frozen skin with antibodies against the immune cell markers GR-1 for granulocytes and CD3 for T lymphocytes and with a phosphorylation specific antibody against STAT3. Red staining shows nuclei. Skin samples were obtained at day 7 after birth from mice with epidermis specific deletion of IKK2 injected with control liposomes (left), clodronat liposomes (middle) of from uninjected control mice (right). Investigating interactions between keratinocytes and macrophages Our results suggest a pathogenic role for macrophages in the hyperproliferative- inflammatory skin disease. Previous reports have identified a population of dermal macrophages that line up at the epidermal- dermal interface in psoriatic skin. These are known as epithelium lining macrophages which are able to directly communicate with epidermal keratinocytes by filopodia like extensions through the epidermal basement membrane. We have found that this population of macrophages shows strong expression of CD83, a surface marker for myeloid dendritic cells. It is therefore possible that epidermal keratinocytes in mice with epidermis specific IKK2 deletion stimulate the maturation of macrophages to CD83 positive dendritic cells which may then exert pro inflammatory functions. Perspectives The analysis of interactions between epidermal keratinocytes and cells in the dermis is an important issue that will be continued in the future. We will also develop model systems which allow to directly test the relevance of our findings in the human system. Future work will aim at identifying basic pathogenic mechanisms of inflammatory skin disease and at identifying new potential therapeutic targets in skin inflammation. Selected publications Gareus R, Huth M, Breiden B, Nenci A, Rösch N, Haase I, Bloch W, Sandhoff K, Pasparakis M: Normal epidermal differentiation but impaired skin barrier formation in mice with keratinocyterestricted knockout of IKK1. Nat Cell Biol (2007) 9, Haase I: Immunologic misbehaviour in psoriasis. Response to Clark and Kupper. J Clin Invest (2006) 116, Wang H, Peters T, Kess D, Sindrilaru A, Oreshkova T, Van Rooijen N, Stratis A, Renkl AC, Sunderkotter C, Wlaschek M, Haase I, Scharffetter-Kochanek K: Activated macrophages are essential in a murine model for T cell-mediated chronic psoriasiform skin inflammation. J Clin Invest (2006) 116, Stratis A, Pasparakis M, Rupec RA, Markur D, Hartmann K, Scharffetter-Kochanek K, Peters T, van Rooijen N, Krieg T, Haase I: Pathogenic role for skin macrophages in a mouse model of keratinocyte-induced psoriasis-like skin inflammation. J Clin Invest (2006) 116, Nenci A, Huth M, Funteh A, Schmidt-Supprian M, Bloch W, Metzger D, Chambon P, Rajewsky K, Krieg T, Haase I, Pasparakis M: Skin lesion development in a mouse model of incontinentia pigmenti is triggered by NEMO deficiency in epidermal keratinocytes and requires TNF signaling. Hum Mol Genet (2006) 15, Stratis A, Pasparakis M, Markur D, Knaup R, Pofahl R, Metzger D, Chambon P, Krieg T, Haase I: Localized inflammatory skin disease following inducible ablation of I kappa B kinase 2 in murine epidermis. J Invest Dermatol (2006) 126, Pasparakis M, Courtois G, Hafner M, Schmidt-Supprian M, Toksoy A, Krampert M, Goebeler M, Gillitzer R, Israel A, Krieg T, Rajewsky K, Haase I: TNF-mediated inflammatory skin disease in mice with epidermis-specific ablation of IKK2. Nature (2002) 417,
49 Research Area B B 6 Function of α3β1 and α6β1 Integrins and their Binding Proteins FHL2 and GIPC in Skin Biology and Pathology Monique Aumailley Institute for Biochemistry II Integrin signalling involves determinants from both the extracellular cellular space (extracellular matrix proteins) and the cell interior (focal adhesions and cytoskeleton-associated proteins, including Rho-GTPases). We are currently investigating the role of Rho-GTPases in laminin-induced signal transduction, i.e. upon activation of the α3β1 and α6β1 integrins. Introduction Laminins (LM) are essential for a myriad of cellular activities because by interacting with their integrin receptors they provide cells with positional information and control cell adhesion, polarisation, differentiation, migration, survival and expression of specific genes. All 15 known LM isoforms have a similar C-terminus interacting with integrins (Aumailley et al., 2003; 2005). LM111, highly expressed during development, is repressed and replaced by other isoforms in adult tissues, for example LM332 and 511 in the basement membrane of the skin. LM111 and other isoforms are ligands for α6β1 integrins, while α3β1 integrins bind LM332 and 511/521 only, and negatively regulate α6β1 integrins. The β1 integrins are connected to the actin cytoskeleton at cell-matrix adhesions, which organisation and turnover is regulated by small GTP-binding proteins of the Rho family, RhoA, Cdc42 and Rac1. which serve as binary molecular switches in the transduction of upstream signals, including those elicited by integrins, to downstream effectors. During migration, activation of Cdc42 and Rac1 induces de novo actin polymerisation and cellular protusions, i.e. filopodia and membrane ruffling, respectively, and the formation of small adhesion complexes. Subsequent activation of RhoA promotes bundling of pre-existing actin micofilaments and actin-myosin interactions leading to the formation of contractile actin stress fibers and mature focal adhesions. This sequential scenario has been largely deduced from studies performed with mouse 3T3 fibroblasts interacting with a fibronectin-rich matrix with fibronectin-binding integrins. We previously showed that binding of α3β1 and α6β1 integrins to LM332 and LM111, respectively, leads to specifc cellular phenotypes differing from that induced by fibronectin, suggesting activation of different signaling pathways. LM111 and LM332 induce different cell migration patterns Analysis by time-lapse video-microscopy of Wi26 fibroblasts and HaCaT epithelial cells exposed to LM111 and LM332, showed that Wi26 fibroblasts adopt an elongated morphology and migrate longer distances than HaCaT epithelial cells. Moreover, computer-assisted analysis of the cell migration tracks revealed that LM111 and LM332 induce undirected and directed migration, respectively. At a molecular level, this is related to different activation of Cdc42 and Rac1 by the two LMs. hence to changes in the balance between activated Rho-GTPases. Fig. 1: Migration of HaCaT epithelial cells (A,C) and Wi26 fibroblasts (B,D) exposed to LM111 (A,B) and LM332 (C,D). The images represent phase contrast microscopy photographs of the cells after 6 hours of migration. The black line indicate the position of the cells at the onset of the recordings (From Chometon et al., 2006). Locking Rho-GTPases in active conformation profoundly affect cell behavior The GTP-binding proteins RhoA, Cdc42 and Rac1 regulate organization and turnover of the cytoskeleton and cellmatrix adhesions, structures bridging cells to their support and translating forces, external or generated within the cell. We have engeeniered Wi26 fibroblasts expressing constitutively active RhoA, Cdc42 or Rac1, and analyzed their biomechanical properties. The actin cytoskeleton as well as the maturation of focal adhesions were specifically modified depending on which of RhoA, Cdc42 and Rac1 was locked in the active conformation. The cell adhesion was, however, not affected. In contrast, the mechanical properties of the cells were dramatically restricted by constitutively active RhoA on bi- and in tri-dimensional 44
50 Research Area B Fig. 2: Wi26 fibroblasts engineered to stably express constitutively active RhoA were stained with antibodies against FHL2 (red) and with FITC-conjugated phalloidin (geen). The fibroblasts display many thick actin stress fibers, as expected upon activation of RhoA, and in addition, distinct clusters of aggregated FHL2 protein. From Zhang et al., surroundings, boosted by constitutively active Rac1 on bi-dimensional surroundings only and not or marginally affected by constitutively active Cdc42. It indicates that the action of Rho GTPases depends on the task cells are performing. Perspectives Altogether, our studies suggest that a strictly regulated and coordinated cross-talk is taking place between lamininbinding integrins, Rho GTPases and FHL2 (four and a half LIM domains) The role of these proteins and a possible dysregulation in their cross-talk is currently investigated in several models of invasive growth of skin tumors in a collaboration with the Department of Dermatology. Selected publications Aumailley M, Bruckner-Tuderman L, Carter WG, Deutzmann R, Edgar D, Ekblom P, Engel J, Engvall E, Hohenester E, Jones JC, Kleinman HK, Marinkovich MP, Martin GR, Mayer U, Meneguzzi G, Miner JH, Miyazaki K, Patarroyo M, Paulsson M, Quaranta V, Sanes JR, Sasaki T, Sekiguchi K, Sorokin LM, Talts JF, Tryggvason K, Uitto J, Virtanen I, von der Mark K, Wewer UM, Yamada Y, Yurchenco PD. A simplified laminin nomenclature. Matrix Biol. (2005) 24, Zhang ZG, Lambert CA, Servotte S, Chometon G, Eckes B, Krieg T, Lapiere CM, Nusgens BV, Aumailley M. Effects of constitutively active GTPases on fibroblast behavior. Cell Mol Life Sci. (2006) 63, Chometon G, Zhang ZG, Rubinstein E, Boucheix C, Mauch C, Aumailley M. Dissociation of the complex between CD151 and laminin-binding integrins permits migration of epithelial cells. Exp Cell Res. (2006) 312, Canault M, Tellier E, Bonardo B, Mas E, Aumailley M, Juhan-Vague I, Nalbone G, Peiretti F. FHL2 interacts with both ADAM-17 and the cytoskeleton and regulates ADAM-17 localization and activity. J Cell Physiol. (2006) 208,
51 Research Area B B 7 RhoBTB Proteins: Role in Oncogenesis Francisco J. Rivero Crespo Institute for Biochemistry I Ras-related small GTPases of the Rho family, which are involved mainly in the regulation of the cytoskeleton, also participate in tumorigenesis. Rho GTPases of the RhoBTB subfamily might function as tumor suppressors, probably through their role as adaptors in cullin 3-dependent ubiquitin ligase complexes. Our goal is to investigate the role of these GTPases in tumorigenesis and to uncover the signal transduction pathways in which these proteins are involved. Introduction Signaling molecules play key roles in the development and progression of malignancies, either upon activation (oncogenes) or upon silencing (tumor suppressors). Among the most widely studied tumor-related genes are small GTPases of the Ras superfamily. The Ras superfamily also comprises Rho proteins, which are involved mainly in the regulation of the cytoskeleton, but also participate in a wide diversity of processes, like vesicle trafficking, phagocytosis, mitogenesis and transcriptional activation. There is increasing evidence supporting a role of Rho GTPases in tumorigenesis. Rho GTPases can influence the motile and invasive properties of tumor cells and deregulate the cell cycle progression, probably acting in concert with Ras. In contrast to all other proteins of the Ras superfamily described so far, RhoBTB proteins display a modular architecture in which the Rho GTPase is followed by a short proline-rich region, two BTB domains and a conserved carboxyl-terminal region. In human, as in other vertebrates, three isoforms have been identified. RHOBTB1 and RHOBTB3, which appear ubiquitously expressed, display a differential pattern of expression: RHOBTB1 shows high levels in stomach, skeletal muscle, placenta, kidney, testis and ovary, whereas RHOBTB3 is highly expressed in neural and cardiac tissues, pancreas, placenta, testis and ovary. RHOBTB2 is very weakly expressed, with relatively high levels in neural tissue, heart and uterus (Ramos et al., 2002). RhoBTB as scaffold proteins: a role as adaptors in cullin 3-dependent ubiquitin ligase complexes The remarkable modular architecture of the members of the RhoBTB family suggests that they could play a pivotal role as scaffold proteins, functioning as docking points for molecules participating in diverse signal transduction cascades. The proline-rich region linking the GTPase to the first BTB domain could act as a SH3 or WW domainbinding site. These two domains are frequently associated with other domains typical for proteins involved in signal transduction processes. The BTB domain is an evolutionarily conserved domain, initially thought to be involved exclusively in protein-protein interaction, participating in homomeric and heteromeric associations with other BTB domains. Common to all RhoBTB proteins is the presence of two BTB domains in tandem and the interruption of the first BTB domain by an insert region of variable length. The sequences of this region are unrelated among the different RhoBTB proteins. Recent reports have established a role of BTB domains in general as adaptors in cullin 3-dependent ubiquitin ligase complexes. Ubiquitin ligases catalyze the addition of ubiquitin, a highly conserved 76-amino acid globular protein, to a number of target proteins. This post-translational modification usually targets proteins for degradation by the proteasome. Similar to cullin 1-dependent SCF complexes, cullin 3-dependent complexes might be targets for auto-ubiquitination, and in fact, it has been reported that RhoBTB2 is ubiquitinated and degraded by the proteasome (Wilkins et al., 2004). Our own results indicate that RhoBTB3 also becomes degraded in the proteasome. We have verified the interaction of RhoBTB proteins with cullin 3 by means of co-immunoprecipitation and yeast two-hybrid experiments. We have also shown that interaction of RhoBTB3 with cullin 3 takes place through the first BTB domain. Moreover, we have also found that RhoBTB proteins are able to build both homodimers and heterodimers with other RhoBTB isoforms, and that dimerization requires the BTB domains. In the course of these experiments we observed that the GTPase domain is able to interact with the region encompassing the BTB domains, resulting in a decreased proteasomal degradation. This has led us to propose a model in which RhoBTB proteins exist in an autoinhibited state, perhaps as dimers. Upon interaction with targets the intramolecular interaction is releaved, allowing the formation of cullin 3-dependent ubiquitin ligase complexes (Berthold et al., in preparation). A two hybrid screening of a mouse brain cdna library has yielded several potential binding partners of RhoBTB that may constitute targets for ubiquitinylation. Interestingly, several of these interaction partners are components of the cytoskeleton. Work is underway to elucidate the role of RhoBTB in the homeostasis of these targets. RhoBTB proteins and the endocytic pathway RhoBTB3 appears to localize at vesicles of a compartment that partially overlaps with early endosomes. These vesicles are frequently found in association with microtubules (Fig. 1) (Berthold et al., in preparation). Interestingly, RhoBTB2 has been reported recently to display a similar localization and to be required for transport of VSVG (vesicular stomatitis virus glycoprotein) (Chang et al., 2006), suggesting that RhoBTB proteins might play roles in microtubule-mediated transport. 46
52 Research Area B Fig. 1: RhoBTB3-decorated vesicles localize along microtubules. COS7 cells were transfected with a plasmid that allows expression of GFP-tagged RhoBTB3, fixed with methanol and stained with an anti β-tubulin antibody. Arrows indicate vesicles in close proximity to a microtubule. RhoBTB as tumor suppressors? Since the first report proposing RHOBTB2 as a tumor suppressor gene (Hamaguchi et al., 2002), additional evidence has accumulated that extends this role to RHOBTB1 (Beder et al., 2006). We have studied the expression pattern of RHOBTB3 and CUL3 in a tumor profiling array and have observed decreased expression of both genes in more than 90% of kidney and more than 74% of breast tumors. This suggests that also RHOBTB3 might be a candidate tumor suppressor gene. In general, expression levels of RHOBTB and CUL3 correlated significantly, indicating that these genes are co-regulated (Berthold et al., in preparation). Target Normal RhoBTB Degradation of Target and RhoBTB Cul3 E2 Target Tumor RhoBTB Fig. 2: Model of the role of RhoBTB3 proteins in tumorigenesis. RhoBTB proteins are required for regulated targeting and degradation of proteins involved in control of cell proliferation and migration. In the absence of RhoBTB these targets accumulate, leading to disregulated proliferation and invasion. Cul3 E2 Accumulation of Target Cell Proliferation Invasivenses Perspectives In the light of the role of BTB domains as adaptors in cullin 3-dependent ubiquitin ligase complexes we envision RhoBTB proteins as elements involved in targeting for degradation of specific proteins that would participate in the control of cell proliferation or migration (Fig. 2). Several regions of RhoBTB could act as targeting or regulatory domains. The function of RhoBTB might be restricted to particular membrane compartments, where these proteins might be required for microtubule-dependent transport of vesicular compartments. The challenge will be linking this molecular mechanisms to the roles of RhoBTB in tumorigenesis. Selected publications Beder LB, Gunduz M, Ouchida M, Gunduz E, Sakai A, Fukushima K, Nagatsuka H, Ito S, Honjo N, Nishizaki K, Shimizu K. Identification of a candidate tumor suppressor gene RHOBTB1 located at a novel allelic loss region 10q21 in head and neck cancer. J. Cancer Res. Clin. Oncol. (2006) 132, Berthold J, Ramos S, Xiong, Y, Aspenström P, Rivero F. Prevention of formation of RhoBTB dependent Cul3 ubiquitin ligase complexes by an intramolecular interaction. Manuscript in preparation. Chang FK, Sato N, Kobayashi-Simorowski N, Yoshihara T, Meth JL, Hamaguchi M. DBC2 is essential for transporting vesicular stomatitis virus glycoprotein. J. Mol. Biol. (2006) 364, Hamaguchi M, Meth JL, von Klitzing C, Wei, W, Esposito D, Rodgers L, Walsh T, Welsch P, King M-C, Wigler MH. DBC2, a candidate for a tumor suppressor gene involved in breast cancer. Proc. Natl. Acad. Sci. USA. (2002) 99, Ramos S, Khademi F, Somesh BP, Rivero F. Genomic organization and expression profile of the small GTPases of the RhoBTB family in human and mouse. Gene (2002) 298, Wilkins A, Ping, Q, Carpenter, CL. RhoBTB2 is a substrate of the mammalian Cul3 ubiquitin ligase complex. Genes Dev. (2004) 18,
53 Research Area B B 8 Resistance to Apoptosis in Hodgkin s Lymphoma: From Molecular Mechanisms to Therapeutical Approaches Hamid Kashkar / Martin Krönke Institute for Medical Microbiology, Immunology and Hygiene Resisitance to cytostatic drug-induced programmed cell death (apoptosis) is a hallmark of neoplastic progression. The malignant Hodgkin and Reed-Sternberg (H-RS) cells of Hodgkin s lymphoma (HL) are characterized by suppressed proapoptotic proteases, the caspase cascade. XIAP, a potent caspase-inhibitor, is uniformly up-regulated in H-RS cells and has been suggested to be the key mediator of apoptosis resistance in HL. The aim of this project is to develop XIAP antagonizing strategies that are suitable to neutralize XIAP action as a therapeutic modality in HL. Introduction Apoptosis is a highly regulated cellular response that ultimately results in the elimination and disposal of unwanted or damaged cells. Apoptosis is brought about by a family of proteases known as the caspases, the activity of which is responsible for the organized destruction of the cell. At least 2 distinct major apoptotic signaling pathways initiating caspase activity can be distinguished (Fig. 1). The triggering of death domain-containing cell surface receptors of the TNF receptor superfamily results in the activation of the initiator caspase, caspase-8. A second apoptotic signaling pathway involves mitochondria and results in the release of cytochrome c, which subsequently initiates the activation of another initiator caspase, caspase-9. Once active, these initiator caspases cleave and activate the zymogens of executioner caspases such as caspases-3 and -7, which in turn are responsible for the majority of proteolytic events that ultimately result in the destruction of the cell. Caspase activation and activity can be modulated by direct interaction with members of the IAPs (inhibitor of apoptosis protein) family, such as XIAP. XIAP binds to and inhibits caspases-3, -7, and -9. The inhibitory action of XIAP, in turn, is regulated by Smac (second mitochondria-derived activator of caspases). In intact cells, Smac is a mitochondrial protein that is released during apoptosis along with cytochrome c. Once in the cytosol, Smac protein binds to XIAP and disrupts its activity. Resistance to apoptotic stimuli is one of the main mechanisms of cancer cell survival. H-RS cells are derived mainly from germinal center or post-germinal center B cells, while a very small minority (< 2%) is derived from T cells. H-RS cells lack specific functional markers of mature B or T cells and therefore should be physiologically prone to undergo apoptosis. The mechanisms of apoptotic resistance in HL cells have been intensively investigated. It has been shown that HL cells are resistant to CD95-mediated apoptosis due to the constitutive expression of cflip. In addition, HL cells display a defective mitochondrial apoptotic pathway and uniformly show up-regulated XIAP expression. Fig. 1: Apoptotic pathways XIAP targeting sensitizes Hodgkin lymphoma cells for cytolytic T-cell attack The immunosurveillance of HL by cytotoxic T lymphocytes (CTLs) is insufficient, and the clinical experience with adoptive transfer of CTLs is limited. CTLs are known to initiate target cell death by employing the apoptotic machinery of the target cells. Mitochondria and mitochondrial release of Smac have been shown to play a crucial role in CTL-mediated cytotoxicity by overcoming the inhibitory effect of XIAP. Our data indicated that mitochondria derived from HL cells have a general defect to receive and/or transduce pro-apoptotic signals and failed to release Smac which results in complete abrogation of cytotoxicity mediated by CTLs. Cytosolic expression of mature Smac or down-regulation of XIAP by short interfering (si) RNA markedly enhanced the susceptibility of HL cells for CTL-mediated cytotoxicity. XIAP gene knockdown sensitized HL cells for killing by antigen-specific CTLs redirected by grafting with a chimeric anti-cd30scfv-cd3zeta immunoreceptor (CD30 is a specific HL marker) (Fig. 2A). The results suggest that XIAP targeting by Smac agonists or XIAP-siRNA can be used as a synergistic strategy for cellular immunotherapy of Hodgkin lymphoma. 48
54 Research Area B Fig. 2: XIAP down-regulation promotes cytotoxicy A) Enhancement of CD30 specific CTL cytotoxicity against XIAP down-regulated HL cells. Isolated peripheral blood CD3 + T cells grafted by retroviral gene transfer with the CD30 specific HRS3scFv-Fc-zeta immunoreceptor and nontransduced T cells were incubated (24 h) with CD30 + L540 or L540-XIAP-shRNA HL cell lines. Viability of target cells was monitored by a XTT-based colorimetric assay. B) Bortezomib reduces XIAP expression. HL B-cell lines were treated (48 h) with increasing concentrations of bortezomib. Indicated modulators of apoptosis were detected by western blotting. C) Bortezomib promotes cytotoxicity by cytostatic agents. Control B-cell line L1309, control Jurkat T-cell line and HL B-cell lines (untreated or pre-treated with bortezomib, 5 nm, 48 h) were treated with indicated cytostatic agents and incubated for 24 h. Viable cell number was determined using an XTT assay. NF-κB independent down-regulation of XIAP by bortezomib sensitizes HL B-cells against cytotoxic drugs The proteasome inhibitor, bortezomib, has been shown to possess unique anti-tumor activity and significant efficacy against a variety of malignancies. Different studies demonstrated that bortezomib breaks the chemoresistance in different tumor cells basically by altering nuclear factor-κb (NF-κB) activity. NF-κB has been shown to be constitutively active in the vast majority of primary Hodgkin-Reed-Sternberg (H-RS) cells in lymph node sections and in Hodgkin s lymphoma (HL) cell lines and was suggested to be a central molecular switch in apoptosis resistance in HL. Our data showed that low-dose bortezomib sensitized HL cells against a variety of cytotoxic agents without altering NF-κB action (Fig. 2B). Strikingly, bortezomib specifically induced marked XIAP down-regulation at the post-translational level (Fig. 2C) which promoted cytotoxicity mediated by various cytostatic agents. Similarly, RNA interference-mediated XIAP down-regulation generated susceptibility to cytostatic agents. The results identify XIAP as an NF-κB independent target of bortezomib action that controls the chemoresistant phenotype of HL cells. The results of this study suggest that a combination of cytostatic agents with bortezomib or XIAP-targeting strategies might become a valuable therapeutic modality in HL. Perspectives The in vitro observations that XIAP targeting promoted cytotoxicity by CTLs and a variety of cytostatic drugs and the fact that XIAP is strongly expressed in majority of HL cases suggest therapeutic implications of XIAP targeting in HL. XIAP antagonizing protocols that have been proved successfull in vitro will be evaluated in vivo using a HL xenograft SCID mouse tumor model to develop significant in vivo efficacy as novel therapeutic modality. Selected Publications Kashkar H, Krönke M, Jürgensmeier JM. Defective Bax activation in Hodgkin B-cell lines confers resistance to staurosporine-induced apoptosis. Cell Death Differ. (2002) 9, Kashkar H, Haefs C, Shin H, Hamilton-Dutoit SJ, Salvesen GS, Krönke M, Jürgensmeier JM. XIAP-mediated Caspase Inhibition in Hodgkin s Lymphoma-derived B Cells. J. Exp. Med. (2003) 198, Kashkar H, Wiegmann K, Yazdanpanah B, Haubert D, Krönke M. Acid sphingomyelinase is indispensable for UV light-induced Bax conformational change at the mitochondrial membrane. J. Biol. Chem. (2005) 280, Kashkar H, Seeger JM, Hombach A, Deggerich A, Yazdanpanah B, Utermöhlen O, Heimlich G, Abken H, Krönke M. XIAP targeting sensitizes Hodgkin`s Lymphoma cells for cytolytic T cell attack. Blood (2006) 108, Kashkar H, Deggerich A, Seeger JM, Yazdanpanah B, Wiegmann K, Haubert D, Pongratz C, Krönke M. NF-κB independent downregulation of XIAP by bortezomib sensitizes HL B-cells against cytotoxic drugs. Blood (2007) 109,
55 Research Area B B 9 Search for Low Penetrance Genes in Hereditary Breast Cancer Rita Schmutzler / Barbara Wappenschmidt Department of Gynaecology and Obstetrics Germline mutations in BRCA1 and BRCA2 are found in the majority of families with evidence for an autosomal dominant transmission of breast cancer. The genetic and phenotypic variability of the disease suggest the existence of modifier genes. Moreover, it is suspected that single nucleotide polymorphisms (SNPs) in these genes may also confer an increased risk for sporadic breast cancer. Using 1) candidate gene and 2) whole genome approach we have identified a set of relevant variants. Introduction Hereditary breast cancer due to mutations in the BRCA1 or BRCA2 genes is a heterogeneous disorder. Besides genetic and allelic heterogeneity variable expression of the disease is obvious. Even in women carrying the same BRCA mutation age at onset, organ specificity (breast or ovarian), histopathological characteristics, associated cancers and course of the disease vary considerably. There is accumulating evidence that modifying genetic and environmental factors contribute to this phenotypic appearance. It is hypothesised that variants in genes affecting the penetrance of BRCA mutations may also be associated with sporadic breast cancer. Hereditary breast cancer families therefore constitute an excellent model for the identification of low penetrance genes that may be relevant for the vast majority of breast cancers. Mutations in BRCA1 and BRCA2 account for about 50% of hereditary breast cancers. Since to date segregation analysis failed to detect additional high risk genes, a polygenic mechanism appears more plausible. Under a polygenic model, a large number of variants each conferring a small genotypic risk ( ) combine additively or multiplicatively thus leading to an increased lifetime risk. Such low penetrance genes may also be responsible for the variable expression of BRCA1 or BRCA2 induced breast cancers thereby acting as modifier genes. Candidate low penetrance genes are usually chosen on the basis of biological plausibility in that alterations of their protein sequence could affect pathways involved in carcinogenesis. The BRCA genes are key players in DNA double strand repair. It has also been shown, that BRCA1 or BRCA2 dysfunction affect chromosomal stability, cell cycle arrest, and apoptosis (figure 1). Besides this genes involved in oestrogen / androgen synthesis and metabolism as well as corresponding hormone receptors and their co-activators are interesting candidates for breast cancer association studies. Modifier genes in BRCA1/2 positive cases In the first part we focus on modifier genes in BRCA1/2 mutation carriers. Until now about 950 mutation carriers have been collected within the German Consortium of Hereditary Breast and Ovarian Cancer and subjected to association studies. To generate a sample set of high statistical power, we started a cooperation with the international Consortium of Investigators of Modifiers of BRCA1 and BRCA2 (CIMBA). First results in a cohort of over patients revealed, that the Rad51 SNP is associated with an increased risk of breast cancer while the STK15 SNP did not reach significance. Our group which is in charge for the 3. call of CIMBA is presently analysing caspase 8 and 10. Gen SNP rs status quo STK15 F31I rs accepted Rad51 5 UTR -135 g > c rs submitted p53 Caspase 8 TGFb DR4 RNASEL ins16 D302H P10L T209R Q462R rs rs rs rs p53 Caspase 10 MTHFR DR4 ARLTS1 R72P V410I C677T E228A C148R rs rs rs rs evaluation of data evaluation of data evaluation of data evaluation of data evaluation of data Table 1: SNPs investigated as pilot studies in our cohort are listed in green. SNPs analysed in collaboration with CIMBA are listed in blue and red. The caspase-study is under our responsibility. Fig. 1: BRCA1 and BRCA2 interaction partners as potential candidate genes. 50
56 Research Area B Gene Gene function SNP Outcome OR (95% confidence interval) P-Value Reference NCOA3 ER-Coactivator 1758G/C MC 0.78 (95% CI = ) B. Burwinkel et al, 2880A/G MC 0.78 (95% CI = ) Clin Cancer Res G/G,1758G/C MC 0.79 (95% CI = ) DR4 Apoptosis signaling 626C/G MC 0.84 (95% CI = ) 0.18 B. Frank et al., 683A/C MC 0.89 (95% CI = ) 0.3 Carcinogenesis C, 683A/C MC 3.52 (95% CI = ) ERBB2 cwel growth, differentation Ile654Val MC 2.56 (95% CI = ) B. Frank et al., Ile 654Val: dimerization Carcinogenesis 2005 CASP8 apoptosis mediator Asp302His MC 0.87 (95% CI = ) 0.35 B. Frank et al., JNCI 2005 CASP10 apoptosis mediator Val410Ile MC 0.62 (95% CI = ) B. Frank et al., CASP10, 8 410Ile, 302His MC 0.37 (95% CI = ) Carcinogenesis 2006 TP53BP1 TP53 enhancer Asp353Glu MC 0.98 (95% CI = ) B. Frank et al., Gly412Ser MC 1.12 (95% CI = ) Breast Cancer Research _1352del6 MC 0.65 (95% CI = ) 0.35 SMYD3 VNTRs B. Frank et al., 3/3 vs (2/3+2/2) MC 0.96 (95% CI = ) 0.78 Int J Cancer 2006 hr MC 0.96 (95% CI = ) 0.77 c-myc proliferation, apoptosis Asn11Ser MC 1.62 (95% CI = ) M. Wirtenberger et al, MC > (95% CI = ) Int J Cancer 2005 PPARGC1A ER-Coactivator Thr612Met MC 1.35 (95% CI = ) M. Wirtenberger et al., hr MC 1.51 (95% CI = ) Carcinogenesis 2006 MC bil 2.3 (95% CI = ) PPARGC1B ER-Coactivator Ala203Pro MC % CI = ) PPARGC1A,B Thr612Met, Ala203Pro MC P trend WRN genome stability Cys1367Arg, TC+CC MC 1.28 (95% CI = ) M. Wirtenberger et al., Cys1367Arg, CC MC 1.39 (95% CI = ) Carcinogenesis 2006 Cys1367Arg, TC MC 1.26 (95% CI = ) p53 genome stability MspI 1798G/A MC 2.15 (95% CI = ) WRN, p53pin3 MC 3.39 (95% CI = ) AKAP13 camp PK regulation Lys526Gln MC 1.63 (95% CI = ) M. Wirtenberger et al., hr MC 1.85 (95% CI = ) Carcinogenesis 2006 AKAP10 camp PK regulation Ile646Val MC 1.25 (95% CI = ) M. Wirtenberger et al., Carcinogenesis 2006 AKAP10,13 Ile646Val, Lys526Gln MC 2.41 (95% CI = ) PRL Prolactin gene TGTG MC 1.42 (95% CI = ) 0.02 A. Vaclavicek et al., PRL Prom. Prolactin promotor TT MC 1.67 (95% CI = ) 0.01 J Clin Endocrinol Metab 2006 GG MC 2.09 (95% CI = ) PRLR PRL receptor TTC MC 0.69 (95% CI = ) PRL, PRLR TGTG/AG, TTC MC P trend ARLTS1 Apoptosis signaling Cys148Arg MC 1.48 (95% CI = ) B. Frank et al., Trp149Stop MC bil 4.11 (95% CI = ) Int J Cancer 2006 MDM2 Inhibition of p53 function IVS1+309T/G >=2 MC< (/95% CI = ) 0.48 S. Wilkening et al., (p53-dep. transcription) >=1MC/OC 1.00 (95% CI = ) 0.98 Cancer Research 2006 >= 2MC>50, 1.05 (95% CI = ) 0.74 Aurora-B Mitosis regulation 885A/G MC 0.84 (95% CI = ) 0.17 S. Tchatchou et al., G/G vs. (A/A+A/G) MC 1.45 (95% CI = ) 0.02 Cancer Lett TCF7L2 Wnt/ß-catenin signaling T allele, rs MC 1.19 (95% CI = ) 0.04 B. Burwinkel et al., GT+TT MC 1.24 (95% CI = ) 0.05 BMC Cancer 2006 SST Prom. GH1 release TC repeat, IVS1+46A/G MC 0.65 (95% CI = ) 0.02 K. Wagner et al., TC repeat MC > (95% CI = ) Carcinogenesis 2006 IVS1+46A/G MC > (95% CI = ) 0.01 TC repeat, IVS1+46A/G MC > (95% CI = ) SSTR2 Prom. TG repeat MC 0.62 (95% CI = ) 0.02 GHRHR C-261T, CT+TT v. CC MC 0.80 (95% CI = ) 0.04 MTUS1 cellular proliferation CNV, Del exon 4 MC 0.58 (95% CI = ) 0.01 B. Frank et al., hr MC 0.41 (95% CI = ) Carcinogenesis 2007 BUB 1B spindle checkpoint 9 different SNPs MC n.s. n.s. A. Vaclavicek et al., BUB 3 TT MC 2.58 (95% CI ) 0.06 Breast Cancer Res Treat 2007 CENPE, TTK n.s. n.s. MAD2L 1,2 n.s. n.s. Table 2: List of genes that have been analysed as low penetrance genes in BRCA1/2 negative families within a collaborative project. The odds ratios and p-values are given. MC, breast cancer; hr, high risk, bil, bilateral Low penetrance genes in BRCA1/2 negative families In our center we identified over 500 high risk breast cancer families for association studies to search for new susceptibility genes. Genes involved in the different signal transduction pathways of BRCA1 and BRCA2 were selected. In a conjoint project 30 genes have been analysed so far and revealed several intragenic SNPs that are associated with either an increased or decreased risk (table 2). The findings confirmed that these genes only exert a small genotypic risk indicating that several of such genes have to act in concert in order to induce cancer. Therefore, studies analysing multiple genes are warranted in order to elucidate their interactions. Perspectives In order to identify new candidate gene we now apply a hypothesis-free approach through adequately powered genome wide association studies using 500 k SNP chips Selected publications Modifier genes: Couch FJ et al. AURKA F31I Polymorphism and Breast Cancer Risk in BRCA1 and BRCA2 Mutation Carriers: A CIMBA study. Cancer Epidemiol Biomarkers Prev, in press. Antoniou et al. RAD51 135G>C modifies breast cancer risk among BRCA2 mutation carriers: Results from a combined analysis of 19 studies, submitted. Low penetrance genes: see publications, table 2. 51
57 Research Area B B 10 Optimization of AAV Vectors for Gene Therapy of Solid Tumors Hildegard Büning / Ulrich Hacker / Michael Hallek Department I of Internal Medicine The insufficient efficiency of gene vectors and the lack of knowledge concerning the most suited combination of vector and target cell are major impediments for successful cancer gene therapies. Our project targets these hurdles by developing special tailored AAV based vectors for transduction of hepatocytes, tumor and tumor endothelial cells, respectively, followed by a direct comparison of their transduction efficiency and specificity in vivo allowing thereby the identification of the best and most efficient combination of AAV vector and target cell for gene therapy of solid tumors. Introduction Gene therapy of solid tumors is a novel area of therapeutic treatment in which the active agent is a nucleic acid sequence rather than a protein or small molecule. Beside the malignant cell, tumor endothelial cells and hepatocytes are gene transfer targets for anti-tumor therapies. An ideal vector for tumor gene therapy would be one that selectively targets the above mentioned cells, is non-toxic to non-target cells and that could be systemically delivered, thereby effecting metastases as well as primary tumors. The adeno-associated virus (AAV) contains already by nature some very promising abilities which suit this virus to be developed as gene therapy vector: AAV is nonpathogenic for humans and elicits only a mild immune reaction. Moreover, AAV shows the potential to integrate site-specific, which would be ideal if stable gene transfer is needed. From AAV a portfolio of different recombinant AAV vector types have been developed with the aim to optimize efficiency or/and specificity of in vitro and in vivo gene transfer. Our group aims to contribute to these goals by insertion of receptor-specific peptides into the AAV2 capsid to retarget the vector tropism (raav targeting vector), by applying combinatorial approaches for the selection of vectors with desired abilities, by analysing different AAV serotypes and AAV:cell interactions. Principle targets for gene therapy of solid tumors and how to reach them Histological analyses of solid tumors reveal that tumor nodes contain malignant cells including a limited number of cancer stem cells, and non-malignant cells like endothelial, stromal and immune cells. The interplay between tumor cells and their environment is of crucial importance for survival of the tumor and metastases formation. Thus, beside the tumor cells non-malignant cells especially endothelial cells have become an interesting target for tumor gene therapy. Tumor cells release a number of angiogenic factors by which endothelial cells are attracted to the tumor site and forced to generate new blood vessels. Tumors exceeding a size of 1-2 mm 2 need these tumor vessels to be supplied with oxygen and nutrients. Furthermore, these vessels are used for tumor cell spreading. Thus, anti-angiogenic treatments should interfere with tumor growth and metastases formation. This has been shown by applying anti-angiogenic factors like angiostatin or endostatin in vivo. Since these factors are needed continuously, but are degraded rapidly in the plasma, a stable transfer of genes coding for such factors would be a significant improvement. Since anti-angiogenic factors are secreted into the blood stream, local as well as systemic anti-angiogenic strategies are possible. Thus, at least two different cell types, hepatocytes and endothelial cells of the tumor vessels (tumor endothelial cells), are suitable gene transfer targets. Beside this, the tumor cells itself is still a valuable target for anti-cancer treatments. Two of the most often used strategies are modulation of the tumor cell to express suicide genes, which will directly kill the tumor cell after systemic application of a prodrug, or immune modulatory genes, expression of which activates the patients immune system to identify and eradicate the malignant cells. All these gene therapy strategies are critical dependent on efficient and specific gene transfer vectors. Furthermore, nothing is known about which strategy is best suited for a given vector. Therefore, we are developing optimized vectors for the transduction of tumor endothelial cells, tumor cells and hepatoytes, respectively, and use these optimized vectors to identify the best combination of vector and target cell. Targeting AAV vectors By insertion of peptide ligands into the AAV2 capsid vector tropism can be modified. Two different peptides, RGD4c and NGR, selected by phage display and described to target tumor endothelial cells were inserted at amino acid position 587 of AAV2 capsid proteins. This site has been proven by us and others to be the most efficient region for insertion of receptor specific peptide ligands. However, the two different targeting vectors (raav- RGD4c and raav-ngr) showed neither an accumulation at the tumor site nor superior transduction of tumor endothelial cells in comparison to raav2 with unmodified capsid when analysed in animal tumor models. Thus these two peptides seem to be unable of mediating tumor endothelial transduction when inserted within the rigid conformation of a viral capsid. To circumvent such limitations, we invented the AAV peptide display library technology. We and others could show that such libraries 52
58 Research Area B in combination with high throughput selections can be used to identify peptides which mediate receptor specific cell entry and efficient intracellular processing of the respective targeting vector in the target cell. As already shown in small animal models, targeting vectors are able to transduce their target cells in vivo even after systemic application. However, target cells can only efficiently be reached if unspecific retention in liver and spleen can be circumvented. For AAV2 based vectors liver retention could be correlated with the ability to bind to heparan sulphate proteoglycan, which functions as AAVs primary receptor. Of interest in this regard is that insertions at 587 are likely to interfere with binding to two of the five amino acids of the recently described HSPG binding motif, explaining the ablation of HSPG binding of some targeting vectors. However, in some cases binding was only partially affected or even restored. We used our AAV peptide display library to explain this observation (for details see Perabo et al., 2006) and optimized this library to enable selections of AAV targeting mutants which transduce cells independent of HSPG binding increasing thereby their in vivo targeting ability and avoiding their retention in liver and spleen. Currently selections for tumor and tumor endothelial selective vectors utilizing this optimized library are performed. Gene transfer into hepatocytes Vectors based on the adeno-associated virus accumulate like most viral and non-viral vectors after i.v. injection in the liver, which is the target organ for systemic antiangiogenic treatments. However, gene expression from hepatocytes mediated by AAV vectors based on the serotype 2 is inefficient. Therefore, we investigated the possible use of an alternative AAV serotype, AAV8, for our application. Recombinant AAV vectors based on the serotype 2 and 8 encoding marker genes like green fluorescent protein (GFP) and luciferase controlled by CMV or tissue specific promoters were generated and systemically applied into different mouse strains. promoter was more efficient in hepatocytes than the CMV promoter. raav8 vectors coding for endostatin and a soluble truncated version of vascular endothelial growth factor receptor-2 (Flk-1), respectively, are currently tested in different mouse models for their efficiency in inducing systemic anti-angiogenesis and thereby a reduction in tumor growth and metastases formation. Results obtained by us and others reveal that raav8 is currently the best available AAV vector type if hepatocytes are the target cells. However, raav8 is not restricted to hepatocytes resulting in transduction of non-targeting cells after systemic application. Thus selection of a cell type specific in this case hepatocyte selective - AAV targeting vector would be advantageous. The respective selections utilizing our AAV display library are ongoing. Beside a gain in specificity we expect a gain in transduction efficiency for these vectors since AAV peptide display approaches select for efficient entry and intracellular processing. Perspectives Gene therapy holds the promise to be a potent and valid strategy to treat cancer. However, the efficiency of such an intervention is critical dependent on the best possible combination of target cell and gene transfer vector. The development of optimized AAV based vectors for gene transfer into the three main targets of anti-tumor therapy tumor cells, tumor endothelial cells and hepatocytes, followed by a direct comparison of their specificity and efficiency in in vivo tumor models, will smooth the way for a prosperous application of AAV vectors in cancer gene therapy. Selected publications Perabo L, Goldnau D, White K, Endell J, Boucas J, Humme S, Work L, Janicki H, Hallek M, Baker AH, Büning H. Heparan sulphate proteoglycan binding properties of adeno-associated virus retargeting mutants and consequences for their in vivo tropism. J. Virol. (2006) 80, Perabo L, Endell J, King SBS, Lux K, Goldnau D, Hallek M, Büning H. Combinatorial engineering of a gene therapy vector: directed evolution of adeno-associated virus. J. Gene Medicine (2006) 8, Work LM, Büning H, Hunt E, Nicklin SA, Denby L, Britton N, Leike K, Odenthal M, Drebber U, Hallek M, Baker AH. Vascular bedtargeted in vivo gene delivery using tropism-modified adenoassociated viruses. Mol. Ther. (2006) 13, Fig. 1: Gene transfer into liver. 5x10e11 genomic particles of raav2 (A) or raav8 (B), coding for GFP controlled by CMV promoter were applied systemically into mice. Liver were harvested two weeks later and stained with anti-gfp antibodies. In all cases, raav8 mediated liver gene transfer was superior to raav2 (Figure 1) and the liver specific Hacker UT, Wingenfeld L, Kofler DM, Schuhmann NK, Lutz S, Herold T, King SBS, Gerner FM, Perabo L, Rabinowitz J, McCarty DM, Samulski RJ, Hallek M, Büning H. Adenoassociated virus serotype 1 to 5 mediated tumor cell directed gene transfer and improvement of transduction efficiency. J. Gene Medicine (2005) 7: Perabo L, Büning H, Kofler DM, Ried MU, Girod A, Wendtner CM, Enssle J, Hallek M. In vitro selection of viral vectors with modified tropism: the adeno-associated virus display. Mol. Ther. (2003) 8,
59 Research Area B B 11 Molecular Genetics of Alopecia Areata Hans Christian Hennies / Peter Nürnberg Cologne Center for Genomics Alopecia areata is a frequent and often chronic autoimmune disease of the hair follicle. Familial occurrence clearly points to a multifactorial disorder with a strong genetic basis. The aim of our project is the molecular characterization of the pathogenesis of alopecia areata. The approach used is a combination of molecular analyses in man and rats, which will decisively contribute to developing pathogenesis-based therapeutic approaches for alopecia areata. Introduction Alopecia areata (MIM ) is a frequent and often chronic autoimmune disorder of the hair follicles in the actively growing anagen stage (Fig. 1). Especially the fast and extensive hair loss in adolescence often leads to social isolation and thoughts of suicide, making care and therapy of these patients a real challenge. The pathogenesis of the disorder is completely unknown. Numerous studies have indicated that alopecia areata is a multifactorial disease with a strong genetic basis, as it is much more frequently found in familial cases and 10-20% of patients have at least one other affected family member. Linkage analysis and association studies have proven to be powerful strategies for identifying not only genes underlying monogenic disorders but also factors involved in common, multifactorial diseases. This has been done using microsatellites for several years; the current method of choice is whole-genome analysis of single nucleotide polymorphisms (SNPs), which is feasible by chip-based genotyping. Moreover, increasing numbers of SNPs on commercially available chips facilitate both linkage and association analysis with the same experimental methodology. DEBR as an animal model for alopecia areata In order to elucidate the molecular pathogenesis of alopecia areata, we follow a combined approach of molecular analyses in humans and in an animal model. We use the Dundee Experimental Bald (DEB) rat for the genetic analysis of alopecia areata (Fig. 1). We cross bred DEB rats of the brown hooded substrain with PVG/OlaHsd rats, yielding 21% F1 offspring with a limited dystrophic hair loss phenotype (Fig. 2). F1 animals were then intercrossed and the F2 generation produced. A total of 430 female F2 rats were analyzed 12 months later, skin biopsies were taken, organ tissue samples were stored, and DNA was isolated. Of the 430 F2 generation rats, 130 were affected with overt hair loss. An additional 62 rats were classified as a subclinical group: these animals presented with a full pelage coat, i.e., an absence of overt hair loss at 12 months of age, but histological analysis showed a mild inflammatory infiltrate of anagen hair follicles, consistent with alopecia areata. For the genetic analysis, we first established an informative microsatellite marker panel for DEB and PVG/Ola, based on the publicly available SHRSPxBN linkage map. Samples of a total of 320 animals were genotyped with 176 markers distributed throughout the rat genome. Linkage results were calculated using model-based and non-parametric statistics. Non-parametric analysis revealed three different candidate regions: two with suggestive and one with significant scores for linkage. Analysis of both autosomal recessive and autosomal dominant models confirmed Fig. 1: Presentation of alopecia areata in humans and in the rat model. Episodic occurrence includes patchy (A), extended, subtotalis, and totalis forms. A similar presentation is found in the DEB rat: patchy (B), extensive (C), and universal hair loss (D). Histopathology includes dystrophic presentation and focal inflammation of hair follicles (E). 54
60 Research Area B pair study design, we are collecting samples from trio families, i.e., affected individuals and their parents, and also from single cases for further studies to follow, including association studies that are supposed both to confirm and refine mapping results obtained by linkage analysis and to provide another, independent approach into gene mapping in multifactorial diseases. Perspectives Our long-term goal is to elucidate the genetic basis of alopecia areata, a common multifactorial, disfiguring disease with autoimmune etiology that results in distressing hair loss. The strategy of analysing affected sib pairs and an animal model promises synergistic effects and provides us with a system for further characterizing genes and proteins involved in pathophysiology. These are important steps and valuable resources for the development of pathogenesis-based therapeutic approaches for alopecia areata. Fig. 2: Breeding strategy for genetic analysis of DEB rats. 130 F2 animals with hair loss were obtained after breeding with PVG/Ola rats. Additonally, animals with a subclinical, histologic phenotype were obtained. the results. In order to establish an optimized penetrance model, we varied the parameters for linkage analysis. This pointed to a partly dominant effect of the underlying gene, which further narrowed down the region (Fig. 3). As this is a very gene-rich interval, we have established several new markers, including both microsatellites and SNPs, for saturated mapping in the candidate region. Alopecia areata as a multifactorial disease The goal of our research is to exploit the rodent model data in the understanding of human alopecia areata. To this end, we are building a human DNA repository from alopecia areata affected people. An international Alopecia Areata Study Group has been established to collect DNA from affected persons and their relatives, and more than 800 affected individuals have been assessed so far. Our approach into the genetic investigation is the analysis of affected sib pairs, i.e., genotyping samples from at least two affected siblings and their parents in each family. We consider this strategy the most promising approach since extended pedigrees are rare and the impact of genetic heterogeneity cannot yet be estimated, and, on the other hand, affected sib pair families are still powerful for linkage analysis. We have collected more than 120 affected sib pair families so far, who have been clinically characterized, their data added to an in-house database, and blood samples taken. Based on epidemiological data available so far, we assume these families suitable for the first whole-genome analysis for linkage. The linkage study is conducted with 500,000 SNPs using established SNP chips commercially available (Affymetrix). In addition to the affected sib Fig. 3: Parametric and non-paramteric analysis of F2 animals from the DEBxPVG/Ola breeding experiment. The dotted line shows the parametric score with a dominant model, the red line the non-parametric score. Saturation mapping is performed first with microsatellite, then with SNP markers. Selected publications Kulle B, Schirmer M, Toliat MR, Suk A, Becker C, Tzvetkov MV, Brockmoller J, Bickeboller H, Hasenfuss G, Nürnberg P, Wojnowski L. Application of genomewide SNP arrays for detection of simulated susceptibility loci. Hum Mutat (2005) 25, Zur Stadt U, Schmidt S, Kasper B, Beutel K, Diler AS, Henter JI, Kabisch H, Schneppenheim R, Nürnberg P, Janka G, Hennies HC. Linkage of familial hemophagocytic lymphohistiocytosis (FHL) type-4 to chromosome 6q24 and identification of mutations in syntaxin 11. Hum Mol Genet (2005) 14, Mizrachi-Koren M, Shemer S, Morgan M, Indelman M, Khamaysi Z, Petronius D, Bitterman-Deutsch O, Hennies HC, Bergman R, Sprecher E. Homozygosity mapping as a screening tool for the molecular diagnosis of hereditary skin diseases in consanguineous populations. J Am Acad Dermatol (2006) 55, Hinkes B, Hennies HC, Nürnberg P, Hildebrandt F. Positional cloning uncovers mutations in PLCE1 responsible for a nephrotic syndrome variant that may be reversible. Nat Genet (2006) 38,
61 Research Area B B 12 Function and Regulation of Lipid Droplets Stefan Höning Institute for Biochemistry I The bulk of excess energy in our body is stored as triacylglycerol (TAG) in lipid droplets of adipocytes. Although it is becoming clear that imbalances of TAG deposition and mobilization contribute to the pathogenesis of dyslipidemia, insulin resistance, ß-cell failure and hypertension, little is known about the molecular mechanisms that control lipid lipid droplet biogenesis and turnover. Here we focus on 2 major lipid droplet proteins, Adipophilin and TIP47 and analyse their role in formation and metabolism of lipid droplets. Introduction TAG is the most efficient energy store and accumulates mainly in lipid droplets of adipose tissue, although almost any cell type in the body seems to be able to form lipid droplets, too. Only a small number of proteins that associate with the phospholipid monolayer of lipid droplets have been identified until now. They constitute the PAT family of proteins and include Perilipin, Adipophilin, TIP47, S3-12 and the myocardial lipid droplet protein MLDP. The expression of Perilipin is restricted to professional lipid storing cells and control TAG storage, protects it from hydrolysis and orchestrates the hormone-stimulated lipolysis. Much less is known about the other lipid droplet associated proteins, but accumulating evidence suggests that Adipophilin has a Perilipin-like function in non-adipose tissue while TIP47 may participate in the biogenesis of lipid droplets. TIP47 was also identified as a binding partner of the mannose-6-phosphate receptors and further work led to the concept that TIP47 in concert with rab9 is mediates retrograde sorting of mannose-6-phosphate receptors from endosomes to the trans-golgi network. In contrast to this view, our own experiments question this view and only support a function of TIP47 on lipid droplets. To further elucidate the function of TIP47 and its closest relative Adipophilin (47% sequence identity), we modulate their expression by RNAi and monitor the effects on lipid droplet biogenesis, turnover as well as consequences for membrane trafficking. Knock-down of TIP47 does not affect mannose-6-phosphate receptor function To analyse the function of TIP47 in cells, we established a HeLa cell line that lacks TIP47 expression (less than 15% residual protein) as a result of vector-based RNAi. In combination with transient RNAi for Adipophilin we have the option to suppress the expression of both main lipid droplet associated proteins. In cells lacking either Adipophilin or TIP47, we did not observe any changes in the localisation of mannose-6-phosphate receptors, or alterations of lysosomal morphology. In addition, we were unable to detect elevated levels of lysosomal enzymes in the cell culture supernatants, which would be indicative of defects in mannose-6-phosphate receptor function. It was also notable that HeLa cells under standard cell culture conditions did not exhibit a pronounced TIP47 staining on membranous structures, which one would expect for a protein functioning as a sorting device. In conclusion, these data suggest that TIP47 does not play a significant role in the sorting of mannose-6-phosphate receptors. Lack of TIP47 and Adipophilin interferes with the maturation of lipid droplets TIP47 is mainly found in the cytosolic fraction of cells, while only very little is detectable in the membrane fraction. This pattern changes very rapidly after incubation of cells in the presence of oleic acid complexed to albumin, which results in TIP47 redistribution to membranous structures identified as lipid droplets due to their staining with lipophilic dyes such as NileRed. We never observed any localisation of TIP47 to structures of the endocytic pathway including the plasma membrane, endosomes or lysosomes. Any colocalisation with markers of the Golgi complex failed. We therefore conclude that TIP47 shuttles between the cytosol and nascent lipid droplets that form in response to fatty acid feeding. Fig. 1: RNAi for TIP47 and Adipophilin block lipid droplet maturation Control HeLa cells (A) and cells with RNAi for Adipophilin and TIP47 (B) were incubated with oleic acid for 12 hours followed by fixation and staining of neutral lipid (red), of DNA (blue) and TIP47 by using a TIP47 specific rabbit serum followed by detection with an Alexa488-konjugated anti-rabbit antibody (green). When cells were observed that lack TIP47 due to RNAi, the morphology and number of lipid droplets was altered. This effect was most dramatic when both, Adipophilin and TIP47 were suppressed by RNAi. As shown in figure 1, fatty acid feeding of HeLa cells led to the formation of lipid droplets of relative homogenous size and distribution (figure 1A), while lack of Adipophilin and TIP47 resulted in a significant loss of incorporated neutral lipid as indicated by the very poor NileRed staining (figure 1B). Staining of the residual amount of TIP47 (green in figure 1) revealed 56
62 Research Area B that the maturation of lipid droplets is altered since they never gain the size of droplets found in control cells. In conclusion, TIP47 and Adipophilin are important for the formation of mature lipid droplets. For further analysis we have cloned GFP-variants of Adipophilin and TIP47 to monitor the formation of lipid droplets in living cells. By using markers of other organelles we should also be able to detect whether an exchange of TIP47 and Adipophilin with other intracellular membranes occurs. we set up the purification of LDs from cells. Figure 2 shows the SDS-PAGE of HeLa cell derived lipid droplet proteins. Based on these preliminary results we can now start to modulate the formation or metabolism of lipid droplets followed by their purification and analysis of the respective proteome. These experiments should show how the protein composition of lipid droplets may change during maturation and in response to physiological changes. In vitro analysis of TIP47 and Adipophilin Although both proteins are known to be recruited to the lipid droplet hemimembrane, the molecular mechanisms are unclear. We therefore cloned recombinant TIP47 and mutants thereof as well as Adipophilin. The pure proteins are currently used to analyse their binding to biosensorimmobilized lipid droplets and lipid-droplet like synthetic membranes. Furthermore we verify whether TIP47 binds directly to Adipophilin and we use pulldown experiments to identify putative novel binding partners. Perspectives The protein assisted formation of lipid droplets may be an important control mechanism for efficient TAG storage. Thus, pharmacological modulation of lipid droplet proteins may help to control pathological imbalances of the lipid droplet-associated metabolism. It is therefore of great importance to elucidate the interplay of lipid droplet-associated proteins and intermediates of the lipid metabolism in the biogenesis and turnover of lipid droplets. Selected publications Höning, S., D. Ricotta, M. Krauss, K. Späte, B. Spolaore, A. Motley, M. Robinson, C. Robinson, V. Haucke and D. Owen. Phosphatidylinositol-(4,5)-bisphosphate regulates sorting signal recognition by the clathrin-associated adaptor complex AP2. Mol. Cell (2005) 18, Bulankina, A., A. Deggerich, J. Wittmann, M. Rudolph and S. Höning. TIP47 is not a sorting device for mannose-6-phosphate receptors but a major protein component of lipid droplets and involved in organelle biogenesis and function. (2007), in revision. Fig. 2: Identified proteins of Hela cell lipid droplets. Cells were incubated with oleic acid for 12 hours to induce lipid droplet formation. Subsequently, the cells were collected and subjected to subcellular fractionation to yield lipid droplets. After washing of the floating lipid droplets, the proteins were resolved by SDS-PAGE and identified by peptide mass fingerprinting. The protein composition of lipid droplets Only a few proteins have been identified that associate with lipid droplet membranes. It is also unclear whether lipid droplets contain a core set of proteins always associated with them, e.g. to control import of fatty acids, formation of TAG as well as for lipolysis. To address this issue 57
63 Research Area B Junior Research Group (NG) I Cell-Cell Interactions and Polarity in Development and Tumorigenesis Carien Niessen Hosted by the Department II of Internal Medicine, the Department of Dermatology and the Department of Surgery Tissue rearrangements require that cells interpret cues from their environment and translate these into appropriate cell shape changes to allow for movement. Our group is interested how intercellular adhesion and polarity contribute to tissue morphogenesis and how altered function of key players in these processes contributes to disease, such as e.g. the formation and progression of (epithelial) cancers. Introduction Proper regulation of intercellular adhesion and polarity is crucial for tissue morphogenesis and maintenance of tissue boundaries. The cadherin family of Ca 2+ -dependent intercellular adhesion can affect a wide range of cellular functions, including activation of signaling pathways, regulation of the cytoskeleton, growth and control of cell polarity (5). Polarity is a fundamental property of cells and crucial for morphogenesis, directed migration and cell identity by regulating asymmetric cell division, apicalbasolateral and planar polarity. The atypical PKCs (apkc) are key players in almost all forms of polarity. Importantly, loss or alterations in cadherin/catenin or polarity protein function contribute to human cancer (3, 5). The goal of our research is to identify mechanisms by which cadherins and polarity proteins like apkc are collaborating in the establishment and maintenance of epithelia and to unravel key regulatory steps by which alterations in these proteins contribute to cancer initiation and invasion. We therefore develop in vivo model systems that address the role of adhesion in both morphogenetic and neoplastic processes. We use murine epidermis as one in vivo model system since this is a selfrenewing, polarized tissue with continuous upward cell movement. Primary cells are also easily obtained from this model system, allowing molecular analysis of underlying mechanisms. The other in vivo model system is the early development of the frog Xenopus laevis because of its dynamic tissue rearrangements. An advantage is that development occurs externally, is rapid and function can be studied in vivo by injection of DNA or RNA. Cadherin signalling regulates vertebrate morphogenesis via atypical PKC The release of the cadherin extracellular domain by proteolytic cleavage occurs both in cancer and during development and is implicated in regulating migration and invasion, and β-catenin signaling. It is still unclear if this a direct effect of the released extracellular domain. To directly test if the cadherin extracellular domain has the capacity to interfere with tissue morphogenesis, we have overexpressed cadherin extracellular domains during early development of the frog Xenopus laevis. Expression of CEC1-5 interfered with gastrulation by inhibiting convergent extension movements, a process driven by polarized cell rearrangements. Since dynamic regulation of cadherin adhesive activity is crucial for convergent extension, we hypothesized that CEC1-5 would directly interfere with adhesion. Surprisingly, we found no difference in adhesive activity of endogenous C-cadherin upon expression of the extracellular domain. Instead, gastrulation defects could be rescued by either dominant active Rac or the cell polarity protein apkc. Our data show that cadherin extracellular domains inhibit vertebrate gastrulation by affecting a cadherin-signaling pathway that includes the small GTpase Rac and apkcs, (Billion et al., manuscript submitted). We propose that cadherins regulate convergent extension movements not only by their adhesive activity but also by setting up the planar polarization of cells via proper positioning of polarity proteins such as apkc. Fig. 1: cadherin extracellular domains cause gastrulation defects (A): blastopure closure in embryos expression control or CEC1-5 extracellular domain (B) CEC1-5 (green) is associated with endogenous cadherins (red)at the cell surface E-cadherin serum levels as a tumor marker in melanoma In collaboration with the group of Cornelia Mauch (B1) we examined if release of E-cadherin ectodomain can serve as an early marker in melanoma, in which metastasis is associated with poor prognosis necessitating early detection. The most commonly used marker, S-100B, has low early sensitivity. E- to N-cadherin switching is thought to contribute to melanoma progression and proteolytic cleavage may serve as the first step in E-cadherin downregulation. We analyzed serum collected from 58
64 Research Area B Fig. 2: E-cadherin impairs epidermal barrier function via regulation of tight junctions. (A) Impaired inside-out barrier function in E-cad epi-/- mice (B) No functional tight junctions as measured transepithelial resistance in E-cad -/- keratinocytes (C) normal recruitment of ZO-1 to tight junctions in E-cad -/- keratinocytes. over 70 melanoma patients for soluble E-cadherin. And found a significant increase in serum E-cadherin levels of melanoma patients with advanced disease compared to the control group (2). Our data also show that levels of soluble E-cadherin are increased in a subset of melanoma patients with normal S-100B values, indicating that serum E-cadherin may be used as an early tumor marker for malignant melanoma (Filonenko, manuscript in preparation). E-cadherin is crucial for in vivo skin barrier function To address the in vivo role of cadherins in stratified epithelia we have conditionally inactivated E-cadherin in mice. Loss of E-cadherin in the epidermis results in perinatal death due to the inability to retain a functional water barrier because of abnormal tight junction function (Fig.1). Surprisingly, intercellular adhesion is not obviously altered. Instead we found that signaling molecules such as Rac and apkc are inappropriately distributed in the epidermis (1). This is the first evidence that E-cadherin is crucial for in vivo tight junction formation. In vitro studies using primary keratinocytes showed that barrier dysfunction is a direct consequence of E-cadherin loss. We are presently testing the molecular and spatial requirements of E-cadherin in epidermal barrier formation and are asking if Rac and apkc are indeed downstream components of the pathway by which E-cadherin affects tight junctions. Atypical PKCs regulate the epidermal barrier Our results with E-cadherin in the epidermis suggested that apkc regulates epidermal barrier formation. Indeed, using either pharmacological inhibitors or dominant negative apkc constructs, we found that interference with apkc activity blocked in vitro barrier formation (4). Two isoforms of apkc exist, encoded by different genes, apkcζ and apkcλ, although it unclear which one regulates the different functions assigned to apkc activity. We found that apkcλ but not zeta is associated with intercellular junctions in the epidermis and that loss results in stratum corneum alterations, implicating this isoform in barrier function. At present, we are examining the mechanisms and substrates by which apkcλ regulates epidermal barrier function. IGF-1 and IR regulate epidermal morphogenesis One pathway that potentially could converge with E- cadherin at the level of apkc to regulate epidermal morphogenesis is the insulin and related IGF-1 pathway. In collaboration with Jens Brüning (A1) we therefore specifically inactivated the insulin receptor or IGF-1 receptor in the epidermis and found that these receptors regulate epidermal morphogenesis cell autonomously. We also did observe alterations in tight junction function in the IGF-1R -/- keratinocytes, suggesting that this pathway is important for barrier function. Wound healing experiments on the IR epidermal specific knockouts revealed no obvious difference, showing that epidermal specific insulin signaling does not contribute to impaired wound healing in diabetic patients (Ibrahim et al., manuscript in preparation). Perspectives Intercellular adhesion and polarity are at the interface of translating cues from within cells and from the environment into cell shape changes. By understanding the basic mechanisms by which cell-cell adhesion and polarity contribute to tissue morphogenesis and integrity, and addressing the molecular function of these processes in pathogenesis we hope to uncover the key molecular pathways by which these pathways contribute to e.g. cancer initiation and progression. Ultimately, this may provide us with potential new targets for therapeutic strategies. Selected publications Tunggal* JA, Helfrich* I, Schmitz A, Schwarz H, Günzel D, Fromm M, Kemler R, Krieg T and Niessen CM: E-cadherin is essential for in vivo skin barrier function by regulating tight junctions. EMBO J. (2005) 24, Billion K, Ibrahim H, Mauch C, and Niessen CM: Increased soluble E-cadherin in melanoma patients. Skin Pharm. Phys. (2006) 19, Niessen, C.M. and Yap, A.S. Another job for the talented p120- catenin. Cell (2006) 127, Helfrich I, Schmitz A, Zigrino P, Michels C, Haase I, Le Bivic A, Leitges M and Niessen CM: The role of apkc isotypes and their binding partners Par3 and Par6 in epidermal barrier formation. J. Invest. Dermat. (2007)127, Niessen CM: Tight junctions/adherens junctions: basic structure and function. J. Invest. Derm. (2007), in press. 59
65 Research Area B Junior Research Group (NG) IV Regulation of Lineage Commitment in Mammalian Skin Catherin Niemann Hosted by the Institute for Pathology The skin is renewed throughout adult life by a population of stem cells that have the ability to generate the differentiated cell types of the interfollicular epidermis, hair follicles and sebaceous glands. We are investigating how lineage selection is regulated and how stem cells and lineage progenitor cells contribute to tumour development in the skin. Our studies of stem cell regulatory pathways focus on the hedgehog and Wnt/βcatenin signalling cascades. Introduction The epidermis forms the outermost layer of the skin and provides the protective covering of the body. It comprises a multilayered stratified epithelium, the interfollicular epidermis, and is associated with epidermal appendages, the hair follicles and the sebaceous and sweat glands. The epidermis is constantly renewed and hair follicles are remodelled by the hair cycle. Diffusible factors, cell to cell contacts and extracellular matrix proteins are all components of the microenvironment of individual stem cells and profoundly affect the differentiation pathway selected by their progeny. Unbalanced renewal and remodelling can lead to a variety of diseases including the development of skin cancer. Many of the molecules regulating epidermal self-renewal and differentiation have now been identified. A key role has emerged for Wnt/β-catenin signalling in response to the diverse repertoire of Wnt ligands expressed in different regions of the skin. A second important signalling cascade in the cutaneous epithelium involves the secreted protein Sonic hedgehog, its receptors Patched and Smoothened, and downstream transcription factors of the Gli family. Both pathways also play an important role in epidermal carcinogenesis. Our work aims for a better understanding of how β-catenin and hedgehog signalling function during lineage selection, normal differentiation and the development of skin tumours. Wnt/β-catenin signalling in skin development and skin homeostasis It has been shown recently that the developmentally important Wnt/β-catenin signalling pathway is critical for epidermal morphogenesis. We have generated transgenic mice in which β-catenin signalling is repressed in the epidermis by expressing a dominant-negative transcription factor Lef1 under control of the keratin14 promoter. Interestingly, keratinocytes of the hair follicle transdifferentiate into sebocytes and keratinocytes forming the interfollicular epidermis, leading to the formation of epidermal cysts in these mice. This reflects the mirror image of the phenotype seen in mice over-expressing a stabilised version of β-catenin in the skin which have an unscheduled de novo formation of hair follicles. The level of activation of β-catenin thus determines whether keratinocytes differentiate into hair or interfollicular epidermis and sebocytes. Therefore, manipulating Wnt/β-catenin signalling can change the differentiation pathway of keratinocytes even if cell fate decisions have already been made by the cells. This is particularly interesting because in addition to the newly recognised plasticity of adult stem cells, there are an increasing number of examples of cells changing fate after commitment to a particular program of differentiation. Wnt/β-catenin signalling in skin tumourigenesis Aberrant activation of Wnt/β-catenin signalling occurs in many tumours. Stabilising mutations within the NH 2 terminus of β-catenin or genetic defects in molecules regulating β-catenin degradation lead to nuclear accumulation of β-catenin and activation of TCF/Lef1 target genes. Transgenic mice that over-express an active, truncated mutant of β-catenin in the skin develop hair follicle tumours, and activating mutations in β-catenin have been found at high frequency in the corresponding human tumours. Surprisingly, an association between tumours and inhibition of Wnt/β-catenin signalling has also been reported. Mutation or deletion of the NH 2 terminus of Lef1 prevents β-catenin binding and inhibits expression of β-catenin target genes. Transgenic mice expressing N-terminally deleted Lef1 in the skin (K14 NLef1 transgenics) develop spontaneous skin tumours. Consistent with the role of β-catenin activation levels in controlling lineage selection, the tumours in K14 NLef1 transgenic mice show sebaceous and squameous differentiation rather than hair follicle differentiation. A high proportion of human sebaceous adenomas and sebeomas have double nucleotide mutations within the β-catenin binding domain of the lef1 gene. In an attempt to investigate how Lef1 mutations contribute to tumour formation we found out that keratinocytes of K14 NLef1 transgenic mice failed to upregulate p53 and p21 proteins during tumourigenesis and in response to UV irradiation, and this correlated with impaired p19arf induction. Two-stage chemical carcinogenesis experiments revealed that mutations within the NH 2 terminus of Lef1 play a dual role in skin cancer, specifying the tumour type (sebaceous tumours) by inhibiting Wnt/β-catenin signalling and acting as a tumour promoter by preventing induction of the tumour suppressor p53. 60
66 Research Area B Hedgehog signalling It has been well documented that hedgehog signalling is required for skin development. Hair follicle development in Sonic hedgehog null mutant mice arrests after the initial epidermal-dermal interactions indicating an important function for Sonic hedgehog signalling for normal advancement beyond the hair germ stage of development. Constitutive activation of Sonic hedgehog signalling in the skin of transgenic mouse models and humans leads to the formation of Basal cell carcinoma (BCC). Smoothened, a member of the hedgehog receptor complex, is required for activating transcription of hedgehog target genes, and over-expression of constitutively active Smoothened results in skin tumours similar to those caused by Sonic hedgehog over-expression. The receptor Patched is mutated in human nevoid Basal cell carcinoma syndrome (Gorlin-Goltz s syndrome), a hereditary predisposition to BCC, medulloblastomas, and rhabdomyosarcomas. Patched mutation results in overexpression and activation of the transcription factor Gli1, and there is evidence that activation of Gli1 and Gli2 leads to the development of BCC. In collaboration with Rune Toftgård from the Karolinska institute we found that multiple components of the Indian hedgehog network are up-regulated in the spontaneous sebaceous tumours developing in K14 NLef1 transgenic mice and also in normal sebocytes of the sebaceous glands. Our data suggest that Indian hedgehog signalling is active in sebocytes and imply a role for Indian hedgehog for sebocyte differentiation and sebaceous tumour formation in the skin. Perspectives We want to investigate the cellular mechanisms leading to tumour development in K14 NLef1 transgenic mice in more detail. Our aim is to find out if potential stem cells are contributing to tumour formation in the skin and we would like to isolate tumour stem cells from skin tissue. Furthermore, we want to understand the contribution of Indian hedgehog signalling for normal sebocyte differentiation and analyse if Indian hedgehog signalling is required for sebaceous tumour formation. Selected publications Niemann C, Owens DM, Schettina P, Watt FM. Dual role of inactivating Lef1 mutations in epidermis: tumour promotion and specification of tumour type. Cancer Res (2007) 67, Niemann C, Unden AB, Lyle S, Zouboulis CC, Toftgård R, Watt FM. Indian hedgehog and β-catenin signaling: Role in the sebaceous lineage of normal and neoplastic mammalian epidermis. PNAS (2003) 100 suppl.1, Braun KM, Niemann C, Jensen UB, Sundberg JP, Silva-Vargas V, Watt FM. Manipulation of stem cell proliferation and lineage commitment : visualisation of label-retaining cells in wholemounts of mouse epidermis. Development (2003) 130, Fig. 1: Wholemount of tail skin from a wild-type mouse stained with hematoxylin. Two hair follicles (HF) with stem cell bulge region (B) and associated sebaceous glands (SG) are shown. The basal layer of the interfollicular epidermis is labelled IFE. Niemann C, Owens DM, Hülsken J, Birchmeier W, Watt FM. Expression of DeltaNLef1 in mouse epidermis results in differentiation of hair follicles into squamous epidermal cysts and formation of skin tumours. Development (2002) 129,
67 Research Area B Junior Research Group (NG) VI Function and Regulation of PML Nuclear Bodies Gerrit Praefcke Hosted by the Institute for Genetics PML nuclear bodies are sub-nuclear organelles involved in the control of gene transcription. Upon attachment of the small ubiquitin related modifier (SUMO) protein to the promyelocytic leukaemia (PML) protein it serves as a scaffold for the recruitment of other proteins such as transcription factors which often contain SUMO interaction motifs (SIM) The aim of this project is to resolve the relation between the assembly of PMLbodies and their biological function on a molecular level Introduction Nuclei of eukaryotic cells contain a number of specialized domains or compartments such as nucleoli, speckles, Cajal bodies, gems and promyelocytic leukaemia (PML) nuclear bodies (NBs). PML NBs have been implicated in diverse functions ranging from an active role in transcription, DNA repair, DNA replication and RNA transport to a purely passive role as intranuclear protein depots. Like other nuclear compartments PML-NBs are not static but vary in size between 0.2 to 1 micrometer and in number from ten to thirty per nucleus. They are defined by the presence of the promyelocytic leukaemia (PML) protein which is essential for the formation of the NBs due to its multimerisation and the subsequent recruitment of other proteins. This process is regulated by the covalent attachment of the small ubiquitin-like modifier (SUMO) protein to the protein PML and several associated proteins which creates the binding sites for proteins containing short peptide sequences called SUMO interaction motifs (SIM). Fig. 1: Schematic model for the assembly of PML nuclear bodies. After the conjugation of SUMO to the proposed scaffold proteins PML and Sp100 these proteins oligomerise and recruit their respective binding partners via the interaction with SUMO interaction motifs leading to the formation of PML NBs. PML bodies also play a role in a number of human diseases. In patients suffering from acute promyelocytic leukaemia (APL) a genomic translocation leads to the fusion of the PML to the retinoic acid alpha receptor (RARα) and PML NBs are absent. Treatment with arsenic leads to the proteasomal degradation of the fusion protein, restoration of PML NBs and remission of the disease. Furthermore, certain viruses such as adenovirus, human cytomegalovirus or herpes simplex virus which need the cellular machinery of the host for their own replication target PML NBs and lead to their dispersal by interfering with the SUMO modification of PML or other PML NB associated proteins. Thus, PML NBs are frequently juxtaposed to the initial sites of transcription of nuclear-replicating DNA viruses. So far, it is not clear whether these viruses extract the proteins they need for their own purposes from PML NBs or whether PML NBs are part of a defense mechanism, for example by blocking the replication of the viral genome. Interaction mapping In order to understand the regulation of PML NB assembly we studied the interactions between several NB-associated proteins. The N-temini of both Sp100 and PML were found to be important for homodimerisation. Although these two proteins are two main components of PML NBs we did not find a direct interaction between them. The C-terminal part of Sp100 interacts with heterochromatin protein 1 (HP1) proteins. Although N-termini of the leukocyte specific Sp110 and Sp140 proteins share a high sequence homology with the related Sp100 protein, we found only weak evidence for the formation of homoand heterodimers. Also we found no evidence for an interaction with HP1. Since the binding of Sp100 to HP1 is enhanced by SUMO modification we will study the possible effects of SUMO conjugation on Sp110 and Sp140 function which both contain possible attachment sites for SUMO. Distinct functions of SUMO isoforms Within the recent years it has become clear that the SUMO interaction motifs which are present in several PML associated proteins including PML, Sp100 and Daxx mediate the interaction with SUMO-modified proteins. Furthermore, SUMO modification has been described to compete with ubiquitin and proteasomal degradation of these proteins and the pattern of SUMO modification changes in response to external stimuli leading to the reversible release and recruitment of proteins involved in transcriptional control. 62
68 Research Area B We could confirm this result for PML as an individual SUMO substrate. Treatment of SUMO3 transfected but not SUMO1 transfected cells with arsenic results in formation conjugated PML (Fig. 3). The effect was slightly weaker for the K11R mutant of SUMO3 which cannot form chains. No large conjugates were observed for the K160R mutant of PML, where one of the major SUMOylation sites is abolished and the protein appeared to be more stable. Fig. 2: Accumulation of SUMO2/3 conjugates after proteasome inhibition. High molecular weight conjugates of SUMO2/3 but not SUMO1 are stabilised by the proteasome inhibitor MG132. Higher eukaryotes contain four SUMO isoforms with at least partially distinct functions. One major difference is that SUMO2 and 3 are themselves substrates for SUMO modification leading to formation of SUMO2/3 chains while SUMO1 stays monomeric. In most studies on PML NBs only the effects of modification by SUMO1 have been studied. Therefore we want to compare the non-modified forms of PML NB associated to those which are covalently linked to the mammalian SUMO1 or SUMO2/3 isoforms. We observed that blocking the proteasomal degradation in HeLa cells leads to an accumulation of SUMO2/3 but not SUMO1 conjugates (Fig. 2). This finding together with data from yeast hints at a link between SUMO chain formation and ubiquitin mediated degradation, possibly via the activity of so far unidentified SUMO ligases for SUMO substrates (ULS) proteins. SUMOylation systems In order to dissect the individual steps of SUMOylation, SIM mediated SUMO binding, assembly of PML NBs as well as desumoylation or degradation it is necessary to obtain SUMO modified proteins for in vitro studies. To this end we have established the preparation of SUMO conjugates. Using recombinantly expressed proteins we can reconstitute the enzymatic cascade for SUMOylation in vitro. This allows us to directly investigate the stimulatory effect of the specific E3 SUMO ligases involved in PML NB assembly. Alternatively we coexpress SUMO substrates with all other proteins necessary for SUMOylation thus obtaining large amounts of SUMOylated proteins for further analyses of binding to other components, desumoylation or further modifications. Perspectives SUMO conjugation controls the assembly of the PML NBs core but also the recruitment of several effector proteins. The differential effects of protein modification by the SUMO isoforms and the link to the ubiquitin mediated proteasomal protein degradation have functional implications for the function of PML NBs. Depending on the transcriptional activity of the cell the may work as checkpoint hubs. Signalling proteins can either be stored until they are needed, released upon activation to fulfil they function in the nucleus or targeted for degradation. We want to identify the components involved in these regulatory pathways and the mechanisms controlling them in order to understand the pathological consequences of PML NB depletion. Fig. 3: SUMO conjugation to PML. SUMO3 but not SUMO1 conjugated wildtype PML accumulated upon treatment with arsenic but not the PML mutant K160R. 63
69 Research Area B Junior Research Group of the Medical Faculty (NG-F) Hepatitis B Virus-Host Interaction Ulrike Protzer Hosted by the Molecular Infectiology Institute for Medical Microbiology, Immunology and Hygiene The aim of our work is to understand in more detail the interaction between human hepatitis B virus (HBV), and its host cell, the hepatocyte. Hereby, we try to understand how HBV is controlled by cellular factors and by the systemic immune response. Based on this knowledge, we want to develop new treatment strategies for chronic viral hepatitis, such as a liver-directed cytokine gene transfer, or re-targeting of T-cells to infected cells. Introduction Hepatitis B viruses (family hepadnaviridae) are small, enveloped DNA viruses which cause acute and chronic liver infections in mammals and birds. Their prototype, the human hepatitis B virus (HBV), is the causative agent of a major public health problem with about 350 million chronic HBV carriers worldwide who are at high risk of developing liver cirrhosis and hepatocellular carcinoma. Genome replication and gene expression of HBV greatly vary in response to extra cellular stimuli such as cytokines to the status of the host hepatocyte. A better understanding of HBV-host interaction is still urgently needed for the development of targeted therapies. A prerequisite for this was to establish infection systems that allow studying (and interfering with) all steps of HBV replication. Models of hepatitis B virus infection The human HBV infects only humans and hominoids. That precludes studies in a convenient animal model. To bypass the species barrier, we have established adenoviral vectors that transfer replication competent HBV genomes (AdHBV) into a broad range of cultured cells and in vivo into animals. Following infection of mice with AdHBV via the tail vein, we efficiently established HBV replication in the mouse liver and mice secreted HBV into their sera. This system now allows for the first time to mimic acute HBV infection, to monitor an HBV specific immune response and determine factors essential for control of HBV infection in a well-characterized small animal model (Oberwinkler 2005; Svorcova, Untergasser et al., unpublished). Using the Ad-HBV model, we demonstrated a differential immune response against the secreted HBsAg in peripheral and in liver associated lymphocytes with the latter being turned off with respect of IFNγ secretion in the liver after a few days (Isogawa et al. 2005). As a model for persistent HBV infection, we use HBV transgenic mice. To study HBV infection in cell culture, we established the isolation and culture of primary human hepatocytes and developed a novel hepatoma cell line that establishes HBV cccdna, the persistence form of HBV: HepG2-H1.3 cells (Webb, Hösel, Bohne et al, submitted). HBV requires highly differentiated hepatocytes for efficient replication Not all hepatocytes containing HBV genomes support viral replication. We hypothesized that cellular differentiation state influences the efficiency of HBV replication. To proof this, we used freshly isolated primary human hepatocytes (PHH) and compared them with well differentiated hepatoma cell lines HepG2, HepaRG and HuH7 and hepatocyte cell line pop10 with respect to differentiation markers, liver specific transcription factor profile and efficiency of HBV replication. PHH replicated HBV at high, HepG2 - at medium and HuH7 - at low levels, whereas pop10 do not support viral replication at all. HepaRG cells showed marked differences related to their differentiation state. Since hepatocyte differentiation markers as well as HBV gene expression relied on hepatocyte nuclear transcription factors (HNF), we tested their role by knocking down HNF1α, HNF4α and HNF3γ. We could proof that high level expression of HNF1α and especially HNF4α driving transcription of HBV pregenomic RNA are necessary for efficient HBV replication and provide the link to a high hepatocyte differentiation state (Quasdorff et al., submitted). HBV pattern recognition by non-parenchymal liver cells: induction of an NFkB dependent cellular response Infection of primary human liver cell cultures with HBV leads to an acute phase response and the upregulation of antiapoptotic genes, which may prevent apoptosis deleterious to the virus (Hösel & Arzberger, manuscripts in preparation). By more detailed analysis, we found that HBV is recognized by pattern recognition receptors in non-parenchymal liver cells. Candidate cells are Kupffer cells or sinusoidal endothelial cells, in which the virus is rapidly taken up (Esser, unpublished). Contact with HBV activates within 3 h NFkB, and leads subsequently to the release of IL-6 and other cytokines, but not to the secretion of IFNs. IL-6 suppresses early HBV gene expression by downregulating HNF1α and HNF4α and probably stabilizes the infected hepatocyte before HBV replication starts (Hösel et al. submitted). 64
70 Research Area B Hepatitis B virus-based vectors for a liver-directed gene transfer As we previously demonstrated, HBV can be converted into a viral vector, which allows for a hepatocyte specific gene transfer (Protzer et al. 1999). HBV-based vectors, however, are not only candidate gene therapy vectors, but also are a very useful tool for experimental purposes. To improve HBV-based vectors, we completely eliminated HBV gene expression, improved transgene expression levels and established packaging cell lines (Untergasser & Protzer 2004). Using improved HBV-based vectors, we performed a first preclinical evaluation of human IFNγ expressing vectors in chimpanzees with chronic hepatitis C in collaboration with scientists from the NIH. Hereby, we could show that HBV-based vectors target the liver after intravenous application and were well tolerated. However, HCV viremia was not controlled, although transient expression of IFNγ induced hepatitis C virus (HCV)-specific CD4- and CD8- T-cell responses (Shin et al. 2005). Cytokine gene transfer as a potential therapeutic option for chronic hepatitis B and C HBV as well as HCV replication have been shown to be extremely sensitive to antiviral cytokines e.g. IFN α and γ. We expressed IFN α and γ in the livers of HBV-transgenic mice under control of a tetracyclin-controlled promoter using adenoviral vectors to further investigate which step of the HBV replication cycle is influenced by the respective cytokine, and to develop a new therapeutic options. Whereas expression of constitutively active STAT-1 did have no antiviral effect (Siebler et al., 2006), following expression of IFN, we found that HBV replication was almost completely blocked. However, HBV transcripts remained unaffected (Dumortier et al., 2005). This was confirmed by in vitro studies using HepG3-H1.3 cells (Webb et al., submitted). Thus, we concluded, that it would be hard to obtain virus elimination purely by interferon gamma expression in the liver, and that an additional killing of hepatocytes will be necessary. Retargeting T cells against HBV infected cells The final goal in hepatitis B therapy is eradication of the HBV rseplication template, the so-called covalently closed circular DNA (cccdna). Current antiviral treatment of chronic hepatitis B depends on IFNα or nucleoside analogues inhibiting the viral reverse transcriptase. These control replication but the extrachromosomal cccdna mostly persists in the host cell nucleus and continues to produce hepatitis B surface antigen (HBsAg). In the absence of a strong T-cell response, persisting cccdna is sufficient to cause relapsing disease. Together with H. Abken s group, we therefore designed chimeric T-cell receptors directed against HBV surface proteins present on infected cells and used them to graft primary human T cells with antibody-like specificity against these cells. The receptors are composed of a single chain antibody fragment fused to transmembrane and cytoplasmic regions of the costimulatory CD28 molecule and to the CD3 z-signalling domain. Our results show that these chimeric receptors, when retrovirally delivered and expressed on the cell surface (Fig. 1), enable primary human T cells to recognize HBsAgpositive hepatocytes, release proinflammatory cytokines and most importantly - eliminate HBV cccdna positive target cells (Bohne et al. in revision) Fig. 1: Fluorescently labelled HBV-particles bind to T-cells grafted with chimeric T-cell receptors Perspectives Based on our findings, we will try to identify the pattern recognition receptor, which recognizes HBV and the molecular viral pattern recognized. We will try to understand how HBV infection is cleared. In addition, we will further develop HBV-based vectors and investigate whether induction of HO-1 or retargeting T-cells towards HBV infected cells can be used as a therapeutic approach for hepatitis B. Selected publications Untergasser A, Zedler U, Langenkamp A, Hösel M, Quasdorff M, Esser K, Tapperzhofen B, Kolanus W and Protzer U. Dendritic cells take up viral antigens but do not support the early steps of hepatitis B virus infection. Hepatology (2006) 43, Siebler J, Protzer U, Wirtz S, Schuchmann M, Höhler T, Galle PR, Neurath M. Adenoviral over expression of STAT-1 does not inhibit Hepatitis B-Virus replication. Eur J Gastroenterol (2006) 18, 167. Dumortier J, Schönig K, Giese T, Schirmacher P, Bujard H, Protzer U. Liver-specific expression of interferon gamma following adenoviral gene transfer controls hepatitis B virus replication in mice. Gene Therapy (2005) 12, Shin E-C, Protzer U, Untergasser A, Feinstone SM, Rice CM, Hasselschwert D, Rehermann B. Liver-Directed Interferon-γ Gene Delivery in Chronic Hepatitis C. J Virol (2005), 79: Isogawa M, Kakimi K, Kamamoto H, Moriyasu F, Protzer U and Chisari FV. Differential Dynamics of the Peripheral and Intrahepatic Cytotoxic T Lymphocyte Response to a Secreted Viral Antigen Produced in the Liver. Virology (2005) 333,
71 Research Area B Liaison-Group Neuroblastoma A Comprehensive Clinical and Molecular Approach to a Pediatric Model Tumor Frank Berthold Department of Pediatric Oncology and Hematology Instead of identical histological patterns neuroblastoma tumors may demonstrate opposite biological features: spontaneous regression without cytotoxic therapy and mainly treatment-resistant tumor progression. A national acquisition of quality-controlled tumor samples was established with a > 80 % success rate. The Tumorbox for shipping samples improved handling and tissue quality significantly. A new molecular gene signature was defined and added important discriminative power to distinguish regressive from progressive courses of the disease. The clinical trial NB2004 spares now cytotoxic chemotherapy for half of all neuroblastoma patients. Introduction The local network for a nationwide clinical trial, the tumor bank and the molecular investigation of that tumor tissue provides a unique opportunity for a better understanding of the specifically complex tumor biology. The basic phenomena regression, progression and maturation may serve as a model for other cancers. The outreach of new insights is facilitated by the close networking of the Liaison Group Neuroblastoma within CMMC, the competence net Pediatric Oncology and Hematology (KPOH: tumor banking), the National Genome Research Net (NGFN2: gene profiling) and the International Society of Pediatric Oncology (SIOP, clinical trials). Tumor bank neuroblastoma The availability of statistically sufficient numbers of tumor samples and other biomaterial in high quality with corresponding clinical data is crucial for biomedical research. The samples are collected after informed consent of the parents and/ or patients, shipped by the specially designed tumorbox (figure 1), and collected with pathological review of tumor cell content and according to the Fig. 1: Tumorbox for simultaneous shipping of frozen and unfrozen material accepted standards for DNA and RNA content and quality. The number of unfixed tumor material (fresh frozen tissue, touch smears) increased form 54 % in 2000 to 82 % of all clinical trial patients in This represents approximately 5000 samples of 850 patients. Of ten applications from other research groups, nin were approved by an independent board. Progression and regression in the clinical trial Amplification of the MYCN oncogene (2p24) is internationally the only molecular marker used to predict poor outcome. The introduction of deletion or imbalance of 1p36.2 as a second adverse stratifying marker into the national treatment protocol separated additional high risk progression patients, but lead also to a more distinct description of the favourable biology and resulted in a small but significant increase of the number of patients in the observation group. If the incidence of recurrences and death from tumor will not increase during the study period then as much as half (54 % in 2006) of all patients may finally be spared from cytotoxic chemo- and radiation therapy. Improved prognostic classification by gene expression profiling of neuroblastoma To comprehensively characterize the transcriptome of neuroblastoma and to identify a predictive gene expression signature, a disease-specific oligonucleotide-microarray comprising about genes has been developed in collaboration with the German Cancer Research Center. Using the prediction analysis of microarrays (PAM) algorithm, it was demonstrated in 251 primary neuroblastoma samples that tumors showing regression are readily distinguishable from aggressive neuroblastoma by characteristic gene expression patterns (Fig. 2). Survival curve analysis and multivariate analysis suggested that outcome prediction using our PAM classifier may be more accurate than risk assessment using established prognostic markers such as stage, age and MYCN amplification (Fig. 3). In addition, gene expression data obtained from the microarray analyses were utilized to identify transcripts that are putatively involved in the molecular mechanisms of spontaneous regression and progression of neuroblas- 66
72 Research Area B Fig. 2: Hierarchical cluster analysis of neuroblastoma patients using the 144 genes of the PAM classifier. Lines represent genes, columns represent patients. Gene expression levels are visualized as log-values ranging from blue (+1.0) to red (-1.0). Results of the PAM prediction (green: favourable, red: unfavourable) are indicated at the bottom of the figure. Fig. 3: Kaplan-Meier estimates for event-free survival of neuroblastoma patients (subdivided into low-, intermediate and high-risk groups according to trial NB2004). The PAM classification discriminated high rish patients out of the low risk group and low risk patients out of the high risk group. toma. Using various mrna quantifying techniques, it was shown that the putative tumor suppressor gene IGSF4 is down-regulated in aggressive stage 4 neuroblastoma. Reconstitution of IGSF4 expression in four neuroblastoma cell lines resulted in decreased proliferation and an increase of cell death. Thus, IGSF4 may have a function as a tumor suppressor in neuroblastoma. Moreover, a novel gene ( DEIN ) has been identified and characterized at the genomic level. Expression of DEIN strongly correlates with neuroblastoma of stage 4S, which usually follows spontaneous regression. These data indicate that DEIN may play a role in the process of spontaneous tumor involution observed regularly in neuroblastoma. Perspectives The comprehensive approach to one Pediatric model tumor resulted in a new quality of risk estimation and had already clinical consequences. First steps in identification of potentially relevant genes (IGSF4, DEIN) promise a better understanding of basic biological processes like spontaneous tumor regression and malignant progression. Selected publications Berthold F et al.: Myeloablative megatherapy with autologous stem-cell rescue versus oral maintenance chemotherapy as consolidation treatment in patients with high-risk neuroblastoma: a randomised controlled trial. Lanc Oncol (2005) 6, Berthold F, Simon T: Clinical Presentation in neuroblastoma. In: Cheung NK and Cohn SL, Springer, ISBN: X p Boensch M, others, Berthold F, Spitz R: Quantitative real-time PCR for quick simultaneous determination of therapy-stratifying markers MYCN amplification, deletion 1p and 11q. Diagn Mol Pathol (2005) 14, Oberthuer A, others, Berthold F, Fischer M. Characterization of a complex genomic alteration on chromosome 2p that leads to four alternatively spliced fusion transcripts in the neuroblastoma cell lines IMR-5, IMR-5/75 and IMR-32.Gene (2005) 363, Oberthuer A, others, Berthold F, Fischer M: Classification of neuroblastoma patients by published gene-expression markers reveals a low sensitivity for unfavorable courses of MYCN non-amplified disease. Cancer Lett (2007) 250, Otto C, others, Berthold F, Schicha H, Schomacker K: Localization of 131I-labelled monoclonal antibody ERIC1 in a subcutaneous xenograft model of neuroblastoma in SCID mice. Nucl Med Commun (2006) 27, Ernestus K, others, Berthold F: Structure, use, and risks of biomaterial repositories of embryonal tumors. Clin Padiatr, 2006; 218: Spitz R, others, Berthold F: Oligonucleotide array-based comparative genomic hybridization (acgh) of 90 neuroblastomas reveals aberration patterns closely associated with relapse pattern and outcome. Genes Chromosomes Cancer (2006) 45, Spitz R, Hero B, Simon T, Berthold F: Loss in chromosome 11q identifies tumors with increased risk for metastatic relapses in localized and 4S neuroblastoma. Clin Cancer Res (2006) 12, Spitz R, others, Berthold F, Hero B: Favorable outcome of triploid neuroblastomas: a contribution to the special oncogenesis of neuroblastoma. Cancer Genet Cytogenet (2006) 167, Oberthuer A, Berthold F, others, Fischer M: Customized oligonucleotide microarray gene expression-based classification of neuroblastoma patients outperforms current clinical risk stratification. J Clin Oncol (2006) 24,
73 Research Area C C 1 The Role of Neuronal Insulin Resistance in Neurodegenerative Disease Markus Schubert 1 / Jens C. Brüning 2 / Wilhelm Krone 1 Department II of Internal Medicine 1 / Institute for Genetics 2 Cerebral insulin receptors play an important role in regulation of energy homeostasis and development of neurodegeneration. Accordingly, type 2 diabetes characterized by insulin resistance is associated with an increased risk of developing Alzheimer s disease. The underlying molecular mechanisms are not well understood. In the current project we investigated the influence of insulin resistance on cerebral amyloid load, plaque formation and longevity in mice. Introduction The physiological and pathological role of insulin receptor (IR) signaling in the central nervous system (CNS) is still unclear. In different clinical studies, an association of type 2 diabetes and neurodegenerative diseases has been described. One explanation could be, that vascular complications of diabetes result in neurodegeneration. Alternatively, the mechanism might be directly related to insulin signaling, suggesting a common cerebral signaling pathway in type 2 diabetes and Alzheimer s disease, leading to the proposal that Alzheimer s disease is a brain-type diabetes. Alzheimer s disease is characterized by the development of amyloid plaques containing the βa1-42 and the βa1-40 cleavage products of the amyloid precursor protein (APP). These peptides are capable of forming plaques, are cytotoxic, and their accumulation represents a key step in the development of Alzheimer s disease. In Alzheimer patients and healthy subjects, hyperinsulinemic-euglycemic clamp studies revealed an improving effect of insulin on cognitive function. Interestingly, administration of large doses of insulinlike growth factor-1 (IGF-1) reduced amyloid load in the brains of mice overexpressing the Swedish mutation of APP (APPsw) a well established mouse model of an Alzheimer s disease like pathology (Tg2576). On the other hand IGF1 resistance in drosophila, C. elegans and mice increases longevity, whereas expression of βa1-42 in C. elegans reduces life span. However, this phenomenon can be reversed by knocking down daf2, the homolog of the IR/IGF1 receptor in C. elegans, suggesting that IR/IGF1 resistance might reverse βa1-42 toxicity. IRS2 knockout mice are a well investigated model for insulin resistance and diabetes. Male and female mice are insulin resistant. However, male mice develop hyperglycemia due to pancreatic β-cell failure. In order to investigate the role of insulin resistance and hyperglycemia in CNS amyloid load and longevity we crossed IRS2 deficient mice with Tg2576 mice. IRS2 and longevity of Tg2576 mice In the current project we characterized and compared four different genotypes: wild type (WT) mice, IRS2 knockout mice (IRS2 -/- ), Tg2576 transgenic mice and mice deficient for IRS2 which express the APPsw transgen (IRS2 -/- Tg2576). In order to gain information about general health of the different genotypes we generated growth curves. Interestingly, we found that in females the Tg2576 mice gained less body weight starting at about 8-10 weeks of age compared to the other genotypes. However, male Tg2576 and IRS2 -/- Tg2576 mice grow less than WT and IRS2 -/- mice. The growth curves of Tg2576 and IRS2 -/- Tg2576 mice branch out at about 10 weeks of age. These data indicate that female Tg2576 as well as male Tg2576 and IRS2 -/- Tg2576 mice develop health impairment. To further analyze this point we generated Kaplan- Meier curves for all genotypes.as described previously Tg2576 mice suffer from increased mortality, which is more pronounced in male than in female mice. Surprisingly, in females, IRS2 -/- Tg2576 mice do not show premature death. This means, that IRS2 deficiency reverses the premature mortality of Tg2576 mice completely. Interestingly, in males IRS2 knockout Fig. 1: Kaplan-Meier survival curves from the different genotypes. The upper panel represents male, the lower panel female mice. 68
74 Research Area C does not influence mortality of Tg2576 mice, indicating a specific function of the IRS2 branch of the IR/IGF1R signaling cascade in regulating longevity in females (Fig 1). Role for IRS2 in regulation of CNS amyloid load In order to study the role of IRS2 in regulating amyloid load and apoptosis we used western blots and histological investigations to analyze the brains of the different genotypes. At first we quantified APPsw transgen expression of Tg2576 mice using an antibody exclusively detecting human APP (6E10). There were no differences in transgenic expression comparing Tg2576 and IRS2 -/- Tg2576 mice, excluding artifacts due to altered APPsw expression. We furthermore investigated the accumulation of βa1-42 and βa1-40 in the brains of the different genotypes. Using specific antibodies for βa1-42 and βa1-40 we could detect an about 50% reduction of these peptides in 3 months old IRS2 -/- Tg2575 mice compared to Tg2575 mice (Fig 2). Fig. 2: Western blot using the antibody 6E10 revealed unaltered expression of the full length APPsw transgen expression in IRS2 -/- Tg2576 compared to Tg2576 mice. Immunoblots for βa1-42 and the βa1-40 showed a 50% reduction of these peptides in IRS2 -/- Tg2576 compared to Tg2576 mice IR-β chain expression serves as loading control. Consistent with these analyses histological investigations revealed a decreased amyloid load in IRS2 -/- Tg2576 compared to Tg2576 mice (Fig 3). There are at least 3 different ways how βa1-42 and the βa1-40 peptides accumulate in the CNS i) increased cleavage from amyloid precursor protein, ii) decreased degradation, iii) decreased transport. So far we analyzed expression of important enzymes involved in the degradation of βa1-42 and βa1-40. We could not detect any differences between the expression of insulin degrading enzyme (IDE) and neprilysin the quantitative most important enzymes involved in amyloid degradation. Furthermore we checked for expression of ApoE and transthyretin which are involved in the transport of βa peptides over the blood brain barrier. There were no differences in the expression of these proteins in brain lysates from the different genotypes. Fig. 3: Total amyloid staining using the 6E10 antibody. Surprisingly, the staining is much weaker in IRS2 -/- Tg2576 compared to Tg2576 mice (in collaboration with Dr. Köhler, Prof. Schröder, Institute for Anatomy II). Perspectives Currently, we are investigating the expression and the activity of β- and γ-secretases in order to elucidate the mechanism how IRS2 influences amyloid load in the CNS in the different genotypes. Furthermore, we analyze Tg2576 and IRS2 -/- Tg2575 mice at the age of 12 months for studying the influence of IRS2 on plaque formation. We have generated neuron-specific IR and neuronspecific IGF1 receptor knockout mice and crossed them into the Tg2576 background to identify the receptor mediating these effects. The analysis of these mice is currently running. Since our data suggest a role for IRS2 in regulating amyloid accumulation and longevity we plan to overexpress IRS2 specifically in neurons. These mice will be crossed with Tg2576 mice. The resulting genotypes will be analyzed in respect to learning and memory, plaque formation and apoptosis. Selected publications Schubert M, Brazil DP, Burks DJ, Kushner JA, Jing Y, Flint CL, Butte A, Kohane I, Dikkes P, Warot XW, Rio C Corfas G, White MF: Insulin receptor substrate-2 deficiency impairs brain growth and promotes tau phosphorylation. J Neuroscience (2003) 23, Schubert M, Gautam D, Surjo D, Ueki K, Baudler S, Schubert D, Alber J, Galldiks N, Küstermann E, Jacobs AH, Krone W, Kahn CR, Brüning JC: Role for neuronal insulin resistance in neurodegenerative diseases. Proc Natl Acad Sci USA (2004) 101, Freude S, Plum L, Schnitker J, Leeser U, Udelhoven M, Krone W, Bruning JC, Schubert M: Peripheral Hyperinsulinemia Promotes Tau Phosphorylation in vivo. Diabetes (2005) 54,
75 Research Area C C 3 Molecular Pathology Induced by Targeted Modifications of the Lipid Bilayer of Cell Membranes in Mouse Mutant Models Wilhelm Stoffel Laboratory of Molecular Neuroscience Complex phospho- and sphingolipids in the membrane bilayer determine the structure and function of mammalian cell membranes as well as intra- and intercellular communication. Using strategies of molecular biology we try to answer the basic questions, how alterations of membrane lipids influence cellular functions in neuronal and extraneuronal tissues. These studies give insight into basic mechanisms in the molecular pathology of the brain, skin and metabolic diseases. Introduction We focus in two reseach areas our studies on a) the role of sphingolipids and their metabolites in cellular metabolism and intra- and intercellular signaling. b) Unsaturated fatty acids determine the lipid environment of membrane proteins. We study the impact of mono- and polyunsaturated fatty acids on membrane functions and Deletion of neutral sphingomyelinase disrupts Golgi vesicular transport and causes a new form combined pituitary hormone deficiency. Neutral sphingomyelinases (SMPD2 and 3) belong to a multi-gene family and are ubiquitously expressed. We discovered that gene ablation of neutral sphingomyelinases leads to a prolongation of the cell cycle causing late embryonic and juvenile hypoplasia fig. 1A and B, a novel form of combined pituitary hormone deficiency (CPHD). The loss of SMPD3 in the Golgi apparatus of hypothalamic has implications for future research into dwarfism and particularly, growth and development of the skeletal system, as well as for current screening and future treatment of dwarfism. Molecular pathology of targeted deletion of mono- and polyunsaturated fatty acid synthesis Targeted deletion of the stearoyl-coa desaturase 1 (scd1) gene in mouse causes obesity resistance and a severe skin phenotype. Here, we demonstrate that scd1 deficiency disrupts the epidermal lipid barrier and leads to uncontrolled transepidermal water loss, breakdown of adaptive thermoregulation and cold resistance as well as a metabolic wasting syndrome. Disruption of the epidermal lipid barrier resulted from protein-bound ceramides being replaced with ω-hydroxylated very long chain fatty acids covalently linked to corneocyte surface proteins. Fig. 1: Cell cycle prolongation causes growth retardation (A) juvenile dwarfism, (B) general hypoplasia and (C) short limbs and chondrodysplasia neurosecretory neurons perturbs the hypothalamuspituitary growth and -epiphyseal growth plate axis. Retarded chondrocyte development and delayed enchondral ossification lead to short-limbed dwarfism and severe skeletal dysplasia fig 1C. We introduced the full-length smpd3 cdna transgene under the control of the chondrocyte-specific promoter Col2a1and completely rescued the smpd3-/- mouse from its phenotype. The smpd3-/- mouse shares its dwarf and chondrodysplasia phenotype with human achondrodysplasia, the most common form of dwarfism combined with skeletal deformations in humans. The rescue of smpd3 in vivo Fig. 2: SCD1-deficiency leads to obesity resistance (A) caused by breakdown of epidermal lipid barrier (B), severe transepidermal waterloss (C), and heat loss (D). Artificial occlusion of the skin by topical lipid application reconstituted the epidermal barrier and reversed dysregulation of thermogenesis and cold resistance as well as the metabolic disturbances. Interestingly, SCD1 deficiency abolished expression of the key transcription factor Lef1 that is essential for interfollicular epidermis, sebaceous glands, and hair follicle development. Finally, the occurrence of SCD1 and a newly described hscd5 (ACOD4) gene in human suggests that the scd1-/- mouse mutant might be a valuable animal model for the study of human skin diseases associated with epidermal barrier defects. 70
76 Research Area C Polyunsaturated fatty acids are present as substituents of different phospholipid classes in all mammalian membranes and contribute in a complex way to membrane structures and functions.. Furthermore, they serve as precursors for numerous physiologically and pharmacologically highly active signaling molecules from eicosanoids to endo-cannabinoids. So far their essential role for cell viability has remained elusive. Fig. 3: Loss of Δ6-desaturase prohibits polyunsaturated fatty acid and eicosanoid synthesis. Lack of thromboxane synthesis deletes platelet aggregation and prohibits induction of thrombus formation(a), prologes bleeding time (B), and causes male (azospermia) (C) and (D). female sterility follicle atresia) causes by disruption of tight junctions of Sertoli cells (blood-testis barrier) (E), and gap junctions of follicle cells (F). Our research focuses on the role of fatty acid Δ-6 desaturase (FADS2). FADS2 is regarded as the key enzyme in the transformation of the two essential fatty acids linoleic and α- linolenic acid into polyunsaturated fatty acids (PUFAs) with ω6-arachidonic, ω3-eicosapentaenoic and ω3-docosahexaenoic acid as most abundant members, and therefore, since arachidonic and eicosapentaenoic acids are the precursors of eicosanoids, the essential step in their transformation to prostaglandins, thromboxanes, prostacyclins, leukotrienes and lipoxins. We studied the systemic role of FADS2 in the Δ6- desaturase deficient mouse (fasd2-/-). Targeted deletion of fads2 expression abolishes completely the transformation of the two essential fatty acids linoleic and α-linolenic acid to arachidonic, eicosapentaenoic and docosahexaenoic acid. FADS2 is solely responsible for the synthesis of eicosanoids, e.g. the synthesis of prostaglandins in vesicular glands, of thromboxanes in platelets and leukotrienes in stimulated macrophages. Surprisingly, male and female fads2-/- mice have a normal life span. Moreover, male and female fads2 null mice are sterile. We show that the Δ6-desaturase is critically required for spermiogenesis and ovary foliculogenesis. The disruption of spermiogenesis in fads2-/- testis with complete lack of mature sperm cells (azoospermia) led us to study the organization of the junctions in Sertoli cell polarity of fads2-/- testes. Defects in these structures are a major cause of male infertility in the human population. The loss of ω6- and ω3- polyunsaturated fatty acid and eicosanoid synthesis disrupts gap junction formation between follicle cells and ovary follicle development. The fads2-/- mutant is an auxotrophic mutant. Diets supplemented with arachidonic or eicosapenta- and docosahexaenoic acid, respectively, rescues eicosanoid synthesis, normal hemostasis and male and female fertility. We anticipate the fads2-/- mouse to be a well defined genetic model for studies in vivo to further our insight into the role of ω6- and ω3-polyunsaturated fatty acids in membrane structure dynamics, including the junctional systems regulating blood-testis barrier and follicle development, eicosanoid functions in cell signaling, in thromboembolic and inflammatory conditions and the development of atherosclerotic lesions. Perspectives The genetic mouse models established in our laboratory open new insight into the molecular mechanisms governing the hypothalamus directed prenatal and juvenile growth and development (SMPD3 deficiency), regulating the epidermal permeability barrier and its role in several skin diseases but also on the body energy metabolism (SCD1-deficiency) and the fads2-/- mouse, which allow for the first time a rational approach to study the role of essential and polyunsaturated fatty acids. Selected publications Stoffel, W., Jenke, B., Block, B., Zumbansen, M. & Koebke, J. Neutral sphingomyelinase 2 (smpd3) in the control of postnatal growth and development. Proc Natl Acad Sci USA (2005) 120, Stoffel, W., Jenke, B., Holz, B., Binczek, E., Günter, R., Knifka, J., Koebke, J. and Niehoff, A. Neutral sphingomyelinase (SMPD3) deficiency causes a novel form of chondrodysplasia and dwarfism that is rescued by Col2A1-driven smpd3 transgene expression. Am J Path (2007) 171, Binczek E, Jenke B, Holz B, Gunter RH, Thevis M, Stoffel W. Obesity resistance of the stearoyl-coa desaturase-deficient (scd1-/-) mouse results from disruption of the epidermal lipid barrier and adaptive thermoregulation. Biol Chem (2007) 388, Stoffel W, Holz B, Jenke B, Binczek E, Günter RH, Kiss C, Karakesisoglou I, Thevis M, Weber AA, Arnhold S and Addicks K: Targeted deletion of fatty acid D6-desaturase (fads2) abolishes polyunsaturated fatty acid and eicosanoid synthesis and causes male and female sterility. Nat Cell Biol (2007), under revision. 71
77 Research Area C C 4 Molecular Mechanisms of Rhythm Generation and Synchronization in Electrically Coupled Cells Structure & Function of Voltage-Gated Ca 2+ Channels in Cardiac and Neuronal Pacemaker Centers and Related Neuronal Circuits, and their Role for Spontaneous Excitability and Memory Formation Toni Schneider / Jürgen Hescheler Institute for Neurophysiology Ablation of pharmacoresistant Ca v 2.3 containing E-/R-type voltage-gated Ca 2+ channels caused cardiac and neuronal disturbances of normal rhythm generation. In adult mice, telemetric ECG recording revealed a complex cardiac phenotype. Further, generalized and brain-specific Ca v 2.3 deficient mice were analyzed for spontaneous epileptiform discharges, and tested for their susceptibility towards convulsive drugs (PTZ, 4-AP, kainate, NMDA) showing hippocampal seizure resistance and reduced neuronal excitotoxicity. Introduction Cardiac pacemaking and oscillations in neuronal networks may rely on similar sets of ion channels. For both, cardiac and neuronal pacemaking, voltage-gated Ca 2+ channels are important for a normal function. For the murine heart, at least 4 out of 10 voltage-gated Ca 2+ channel types contribute to a regular heart beat, including Ca v 2.3 E-/R-type Ca 2+ channels. Interestingly, while the ablation of Ca v 3.1, a low voltage-gated Ca 2+ channel, only leads to bradycardia, the ablation of Ca v 2.3 causes a more complex cardiac phenotype (Weiergräber et al., 2005b). Normal brain function is based on a well balanced release of excitatory and inhibitory neurotransmitters. Ca v 2.3 E-/ R-type Ca 2+ channels are highly expressed in regions containing inhibitory interneurons as the thalamic reticular nucleus and the hippocampus (Dietrich et al., 2003;Weiergräber et al., 2006a). In both regions, the neurotransmission of GABA is the most important inhibitory control for spike timing and sculpting neuronal rhythms. For example, disturbances of the GABA A receptor in the central nervous system cause disorders like anxiety, epilepsy, insomnia and schizophrenia. Ablation of E-type voltage-gated Ca 2+ channels results in cardiac arrhythmia, also in adult Ca v 2.3 deficient mice Amplified cdna fragments from murine heart and single cardiomyocytes reveal the expression of three different Ca v 2.3 splice variants. The ablation of Ca v 2.3 was found to be accompanied by a compensatory upregulation of the Ca v 3.1 T-type calcium channel. Telemetric ECG recordings from Ca v 2.3 deficient mice displayed a subsidiary escape rhythm, altered atrial activation patterns, atrioventricular conduction disturbances and alteration in the QRS-morphology. Furthermore, time domain analysis of heart rate variability in Ca v 2.3-deficient mice exhibited a significant increase in heart rate as well as in the coefficient of variance compared to control mice (Weiergräber et al., 2005b). Heart rate variability, autonomic modulation and functional cardiovascular capacity Heart rate variability is a clinically relevant predictor of cardiac morbidity and mortality indicating systemic shifts in cardiac sympathovagal balance. We demonstrate that Ca v 2.3 deficient mice exhibit not only qualitative ECG abnormalities related to Ca v 2.3 expression in cardiomyocytes but also alterations in autonomic nervous system activity. Autonomic block revealed that intrinsic heart rate does not differ between both genotypes and that increased heart rate in Ca v 2.3(- -) mice is due to increased sympathetic tone. Systemic administration of isoproterenol resulted in a significant reduction in interbeatinterval in both genotypes, whereas Ca v 2.3(- -) mice remained unaffected by selective inhibition of the sympathetic limb using propranolol, indicative of β-adrenoreceptor desensitisation under increased sympathetic tone. Our results indicate that the pharmacologically inducible heart rate range and heart rate reserve (Fig. 1) are severely impaired in Ca v mice suggesting that the Ca v 2.3 voltage-gated Ca 2+ channel is essential not only for normal impulse propagation and conduction in the murine heart, but also for functional cardiovascular capacity. Fig. 1: Functional cardiovascular capability in Ca v 2.3 (+/+) and Ca v 2.3( - - ) mice. Heart rate range is significantly reduced in Ca v 2.3 deficient mice (p<0.05) compared to controls. Heart rate reserve exhibits the same tendency (p = 0.067). 72
78 Research Area C Altered seizure susceptibility in Ca v 2.3 -/- mice (PTZ, 4-AP) Ca v 2.3 E-/R-type voltage-gated Ca 2+ channels were recently shown to be blocked by antiepileptic drugs (lamotrigine, topiramate) (Hainsworth et al., 2003). Further, Ca v 2.3 is thought to induce plateau potentials in CA1 pyramidal cells which can trigger epileptiform activity. The aim of our study was to perform an electroencephalographic analysis, seizure susceptibility testing, and histomorphological characterization of Ca v 2.3 / mice to unravel the functional relevance of E-/R-type Ca 2+ channels in ictogenesis (Weiergräber et al., 2005a). Generalized and brain-specific Ca v 2.3 knockout mice were analyzed for spontaneous epileptiform discharges by using both electrocorticographic and deep intracerebral recordings. In addition, convulsive seizure activity was induced by systemic administration of either 4-aminopyridine (4-AP; 10 mg/kg, i.p.) or pentylenetetrazol (PTZ; 80 mg/kg, s.c.) to reveal possible alterations in seizure susceptibility. Besides histomorphological analysis, expression studies of other voltage-gated Ca 2+ channels in Ca v 2.3 / brains were carried out by using semiquantitative reverse transcription polymerase chain reaction (RT-PCR). Both electrocorticographic and deep intrahippocampal recordings exhibited no spontaneous epileptiform discharges indicative of convulsive or nonconvulsive seizure activity during long-term observation. Gross histology and expression levels of other voltage-gated Ca 2+ channels remained unchanged in various brain regions. Surprisingly, Ca v 2.3-deficient mice were shown to be more resistant to seizures induced by the chemoconvulsant PTZ (impairs γ -aminobutyric acid (GABA)A neurotransmission). In particular, Ca v 2.3 -/- mice exhibited a significantly lower lethality after PTZ combined with a significant reduction of phase 4 latency, duration, and frequency (Weiergräber et al., 2006a). Interestingly, the 4-AP sensitivity remained unchanged. Fig. 2: Deep intrahippocampal (CA1) EEG recording from Ca v 2.3 +/+ (A) and Ca v 2.3 -/- mice (B) following kainate administration (30 mg/kg) (Weiergräber et al., 2007, revised & resubm). Note here, seizure activity is predominantly characterized by repetitive and delimitable high-amplitude exacerbation of spike and/or spikewave activity (*) followed by postictal depression (red arrows). Hippocampal seizure resistance and reduced neuronal excitotoxicity in Ca v 2.3 deficient mice (kainate, NMDA) For kainic acid (= KA; 30 mg/kg i.p.) we tested hippocampal seizure susceptibility in Ca v 2.3 deficient mice using surface and deep intrahippocampal telemetric EEG recordings as well as phenotypic seizure video analysis. Administration of KA revealed clear alteration in behavioural seizure architecture and dramatic resistance to limbic seizures in Ca v 2.3 / mice compared to controls whereas no difference in hippocampal EEG seizure activity between both genotypes could be detected at this suprathreshold dosage (Fig. 2). The same tendency was observed for NMDA seizure susceptibility (150 mg/kg i.p.) approaching the level of significance. In addition, histochemical analysis within the hippocampus revealed that excitotoxic effects following KA administration are absent in Ca v 2.3 / mice, whereas Ca v 2.3 +/+ animals exhibited clear and typical signs of excitotoxic cell death (Fig. 3). These findings clearly indicate that the Ca v 2.3 voltage-gated calcium channel plays a crucial role in both hippocampal ictogenesis and seizure generalization and is of central importance in neuronal degeneration following excitotoxic events (Weiergräber et al., 2006b). Fig. 3: Assessment of excitotoxic cell injury and neuronal degeneration in the hippocampus (CA3 region) of Ca v 2.3 +/+ (left) and Ca v 2.3 -/- mice (right) after systemic kainate (30 mg/kg) treatment. Seven days after injection brains were extirpated and HEstained. In Ca v 2.3 +/+ mice neuronal cell degeneration is present but absent in corresponding sections of Ca v 2.3 -/- animals. Note, the strong granulation of cells and gaps between pyramidal neurons in the CA3 region. Selected publications Dietrich D, Kirschstein T, Kukley M, Pereverzev A, von der Brelie C, Schneider T, Beck H: Functional specialization of presynaptic Ca v 2.3 Ca 2+ channels. Neuron (2003) 39, Hainsworth AH, McNaughton NC, Pereverzev A, Schneider T, Randall AD: Actions of sipatrigine, 202W92 and lamotrigine on R-type and T-type Ca 2+ channel currents. Eur J Pharmacol (2003) 467, Weiergräber M, Henry M, Hescheler J, Smyth N, Schneider T: Electrocorticographic and deep intracerebral EEG recording in mice using a telemetry system. Brain Res Brain Res Protoc (2005a) 14, Weiergräber M, Henry M, Krieger A, Kamp MA, Radhakrishnan K, Hescheler J, Schneider T; Altered seizure susceptibility in mice lacking the Ca v 2.3 E-type Ca 2+ channel. Epilepsia (2006a) 47, Weiergräber M, Henry M, Südkamp M, De Vivie ER, Hescheler J, Schneider T: Ablation of Ca v 2.3 / E-type voltage-gated calcium channel results in cardiac arrhythmia and altered autonomic control within the murine cardiovascular system. Basic Res Cardiol (2005b) 100, Weiergräber M, Kamp MA, Radhakrishnan K, Hescheler J, Schneider T: The Ca v 2.3 Voltage-gated calcium channel in epileptogenesis. Shedding new light on an enigmatic channel. Neurosci Biobehav Rev (2006b) 30,
79 Research Area C C5 Gene Therapy of Brain Tumors Andreas H. Jacobs / Wolf-Dieter Heiss Department of Neurology The overall aim is to further develop imaging-guided gene-, cell-based and molecular targeted therapies for gliomas. We made considerable achievements with regards to (i) imaging-guided gene therapy (Jacobs et al. Cancer Res 2007); (ii) imaging-guided cell-based therapies (Miletic et al. Molecular Therapy 2007) (iii) improved marker gene / marker substrate combinations (Kummer et al. Molecular Imaging 2007); (iv) imaging regulated gene expression (Winkeler et al. PlosOne accepted); (v) improved HSV/EBV/RV and lentivirus vectors (Miletic et al., Winkeler et al.); and (vi) imaging E2F-1 signal transduction (Monfared et al.). Introduction In view of the high incidence and poor prognosis of malignant brain tumors treated by conventional therapies, research focusing on alternative therapies such as gene-, cell-based and molecular targeted therapy seems to be of utmost importance. In the past years, we have established a universal HSV-1 amplicon vector system supporting the proportional coexpression of therapeutic and noninvasive imaging marker genes (e.g. cdirestk39gfp; Jacobs et al. 2003). Molecular imaging technology is being used to localize and quantify (i) the tumor extend (MRI), (ii) the viable target tissue ([ 18 F]FLT-PET), (iii) the transduction efficiency after imaging-guided vector application ([ 18 F]FHBG-PET), and (iv) the induced therapeutic effect ([ 18 F]FLT-PET). We have now made considerable advances in the topics listed in the summary above. Imaging-guided gene therapy in gliomas A detailed analysis of gene therapy induced changes within s.c. growing gliomas revealed similar findings to that made in clinical application (Jacobs et al. Lancet 2001): (i) tissue-dose of vector mediated gene expression (FHBG- PET) can be correlated to induced therapeutic effect (FLT- PET); (ii) complete responses are indicated by reduction in proliferation (FLT-PET) and tumor volume; (iii) partial responses are indicted by change in proliferation (FLT- PET) in parts of the tumor without effects in overall tumor volume; (iv) failing response are indicated by progressive tumor growth (FLT, volumetry)(jacobs et al. Cancer Res 2007; Fig. 1). Using this paradigm we evaluated the effects of HSV-CITG and, in collaboration with Dorothee van Laer (Frankfurt) lentiviral-tk vectors in intracranial gliomas with an improved therapeutic response with 100% long-term survivors (Miletic et al. submitted). Imaging-guided cell-based therapies Mouse neural progenitor cells (C17.2) have been transduced by tkiresgfp (TIG) and lucirestkgfp (LITG) constructs for visualisation by BLI and PET. The detection limit was determined with 100 C17.2-LITG cells for BLI and 5000 C17.2-TIG cells for FHBG-PET. Dynamic imaging studies in i.c. glioma models over an extended period of time (up to 100 days post implantation) revealed (i) limited migratory potential of C17.2-LITG cells towards contralateral growing gliomas; and (ii) uncontrolled growth and migration to cerebellum and spine when coinjected into gliomas (Waerzeggers et al. submitted, Fig. 2). These studies were complemented by a collaboration with Bernd Neumann (Bonn) who generated bone-marrow tumor infiltrating cells expressing TKGFP (BM-TIC-TG cells) which could be localized by PET and which exhibited a therapeutic response in 9L gliomas (Miletic et al. Molecular Therapy 2007). Improved marker gene / marker substrate combinations Detection of HSV-1-TK expression in the intact brain is currently not possible as the specific marker substrate FHBG does not cross the intact BBB. We therefore further evaluated more lipophilic marker substrates for HSV-1-TK expression, such as IVFAU and IVFRU (Li et al. submitted), as well as a combination of a mutated dopamine receptor as marker gene and 11C-racloprid as BBB-permeable 74
80 Research Area C ligand with a good correlation between TK quantification by FHBG-PET and D2R quantification by RAC-PET after transduction of HSV-d2RIREStk39gfp vectors in vivo (Kummer et al. Molecular Imaging 2007). Imaging regulated gene expression. In the attempt to achieve imaging assessment of regulated gene expression in vivo, a tet-regulated HSV-1 amplicon vector was generated (HET) serving tet-regulated LUC and/ or TK expression. We could demonstrate that localization and quantification of tet-regulated gene expression is possible by PET and BLI in s.c. and i.c. growing gliomas enabling the non-invasive assessment of repeated onand off-switching of the regulated gene (Winkeler et al. PLoSOne accepted, Fig. 3). Improved HSV/EBV/RV (HET) and lentivirus vectors Tribrid HSV-1 amplicon vectors have been generated which serve for highly efficient transduction of tumor cells in vivo with subsequent transformation into retrovirus producer cells. Cell culture analysis and characterization imaging response to chemo- and molecular targeted therapies (e.g. tyrosine kinase and VEGF inhibition, PPARg agonists) by FLT-PET for the assessment of glioma proliferation. Selected publications Galldiks et al. Effects of temozolomide in malignant gliomas monitored by MET-PET. Eur J Nucl Med Mol Imaging (2006) 33, Grommes C et al. Inhibition of in vivo glioma growth and invasion by PPAR {gamma} agonist treatment. Mol Pharmacol (2006) 70, Jacobs AH et al. 18 F-Fluoro-L-thymidine and MET as markers of increased transport and proliferation in brain tumors. J Nucl Med (2005) 46, Jacobs AH et al. Imaging-guided gene therapy of experimental gliomas. Cancer Res (2007) 67, Kummer C et al. Multitracer PET of exogeneous gene expression mediated by a universal HSV-1 amplicon vector. Molecular Imaging (2007) 6, show the dynamic increase of gene expression over time which is currently being further investigated in vivo by imaging (Winkeler et al.). The imaging-guided gene paradigm based on lentivirus vectors has been described above (Miletic et al. submitted). Imaging E2F-1 signal transduction The final goal in molecular imaging of gliomas is the development of paradigms for the non-invasive assessment of disease-specific molecular events in vivo. As E2F-1 is one of the key transcriptional regulators in glioma development we constructed glioma cells expressing the LITG-construct under transcriptional control of a E2F- 1 responsive element and could demonstrate that the difference between basal expression of E2F-1 and E2F- 1 expression induced by sublethal chemotherapeutic doses can be non-invsively assessed by BLI (Monfared et al. submitted; Fig. 4). Perspectives To further improve the efficiency of gene and cell-based therapies for gliomas a library of HSV-1 based vectors and tumor-infiltrating cells (TIC) serving expression of multiple therapeutic genes (cd, tk, il4) in conjunction with replication-conditional HSV-1 vectors serving tumor oncolysis is being established and investigated under imaging-guidance. This work is complemented by early Miletic H et al. Bystander killing of malignant glioma by bone marrow-derived tumor infiltrating progenitor cells. Molecular Therapy (2007) 15, Miletic H et al. Normal brain cells contribute to the bystander effect in a suicide gene therapy - approach for malignant glioma using lentiviral pseudotyped vectors. (2007), in revision. Monfared P et al. Non-invasive assessment of E2F-1 mediated transcriptional regulation in vivo. (2007), submitted. Rueger MA et al. The inter-individual variability of infectivity of HSV-1 amplicon vectors in primary human cell cultures. Gene Therapy (2005)12, Rueger MA et al. 18F-FLT-PET for non-invasive monitoring of experimental gliomas during antiproliferative therapy with suicide genes in vivo. EJNMMI (2007), in revision. Saydam O et al. HSV-1 amplicon vector-mediated sirna targeting EGFR inhibits growth of human gliomas. Molecular Therapy (2005) 12, Uhl M et al. Migratory neural stem cells for improved TK-based gene therapy of malignant gliomas. Biochem Biophys Res Commun (2005) 328, Waerzeggers Y et al. Multi-modal imaging of neural progenitor cells in the rodent brain. (2007), submitted. Winkeler A et al. Switching on the lights for gene therapy. PlosOne (2007) 2, e
81 Research Area C C 6 Role of an Impaired Protein Quality Control in Mitochondria for the Pathogenesis of Spastic Paraplegia Thomas Langer Institute for Genetics Hereditary spastic paraplegia is a neurodegenerative disorder characterized by progressive and cell specific axonal degeneration. An autosomal recessive form of the disease is caused by mutations in paraplegin, a conserved subunit of the ubiquitous and ATP-dependent m-aaa protease in mitochondria. We want to characterise the functional link between axonal degeneration, mitochondrial dysfunction, and impaired mitochondrial proteolysis in spastic paraplegia employing yeast and mice as model organisms. Introduction Hereditary spastic paraplegia (HSP) represents a novel category of neurodegenerative disorders which is characterized by the slow degeneration of a subset of axons in a `dying-back manner from the synaptic terminal and processes to the cell body. Axons of the cortical motor neurons degenerate from their distal extremities, leading to rigidity and weakness in the lower limbs. In the pure form of HSP, these symptoms are isolated, indicating that there is a selective involvement of motor axons. In the complicated forms of HSP, other axonal systems are affected and patients have a more complex phenotype, where spastic paraparesis is associated to cortical and cerebellar atrophy, amyotrophy, otpic atrophy, peripheral neuropathy, and deafness. HSP is a genetically heterogeneous disease with more than 30 identified loci. In most cases, the reason for the selective vulnerability of motor axons is still unknown. However, based on their putative function, HSP genes can be divided into thre categories: (1) genes that affect myelination or neuronal recognition; (2) genes that are involved in axonal trafficking; and (3) genes that have mitochondrial functions. The mitochondrial m-aaa protease is involved in HSP Mitochondria have been shown to be involved in HSP with the identification of mutations in the spastic-paraplegia-gene 7 (SPG7) in families affected by a complicated form of autosomal recessive HSP. SPG7 encodes paraplegin, highly homologous (~55% sequence identity) to a group of well-characterized yeast mitochondrial metalloproteases the AAA proteases that belong to the AAA family of P-loop ATPases and form large proteolytic complexes in the inner membrane of mitochondria. The nature of the mutations that were identified in human HSP patients suggests a complete loss of paraplegin function. Therefore, cloning of SPG7 implicated defective proteolysis within mitochondria as one of the pathological alterations that are involved in HSP. Mitochondria are essential for cell survival and exert multiple anabolic and catabolic functions, the production of energy being the most prominent one. Their functionally depends on the coordinated expression of the two cellular genomes, the nuclear and the mitochondrial genome, both of which code for core subunits of the respiratory chain in the inner membrane. In case of an imbalance of gene expression, non-assembled membrane proteins are degraded by AAA proteases which conduct the surveillance of protein quality control. Studies in yeast led to the identification of two AAA proteases in the inner membrane of mitochondria: the hetero-oligomeric m- AAA protease, which is active on the matrix side of the inner membrane, and the homo-oligomeric i-aaa protease, which protrudes to the intermembrane space. A complex of paraplegin and ATPase family gene 3-like 2 (AFG3L2), a closely related protein in the inner membrane of mitochondria, exerts proteolytic activity and can functionally replace the yeast m-aaa protease demonstrating functional conservation. Proteolytic activation or complete degradation: the Janus face of the m-aaa protease Given the role of the m-aaa protease in protein quality control in mitochondria, it is conceivable that non-native membrane proteins accumulating in the absence of paraplegin might be deleterious for mitochondrial activities in neurons and ultimately lead to axonal degeneration. Alternatively, there might be specific substrates of the m-aaa protease, the proteolysis of which affects mitochondrial biogenesis in a tissue-specific manner. We therefore employed a proteolytically inactive variant of the yeast m-aaa protease to purify associated polypeptides biochemically. This approach led to the identification of MrpL32, a conserved nuclear-encoded subunit of mitochondrial ribosomes. Surprisingly, the m-aaa protease does not affect the stability of this protein in mitochondria but cleaves the N-terminal mitochondrial targeting sequence upon import into this organelle. Complementation studies revealed that this function is conserved in human and that the human m-aaa protease composed of paraplegin and AFG3L2 mediates maturation of human MrpL32 in a similar manner. Thus, whereas AAA proteases degrade non-assembled membrane proteins to peptides in a processive manner, they degrade MrpL32 only partially so that they become mature proteins. MrpL32 processing is a prerequisite for its assembly into ribosomes anf or mitochondrial translation. The m-aaa protease therefore controls the completion of ribosome assembly and, consequently, the activation of mitochondrial protein synthesis. This function explains phenotypes 76
82 Research Area C associated with a loss of the m-aaa protease in yeast and may also be of pathogenic relevance in human. However, it should be pointed out that protein activation and degradation by the m-aaa protease are interdependent events and might interfere with each other during the pathogenesis of HSP. Fig. 1: Role of the m-aaa protease in protein activation and degradation. The m-aaa protease controls ribosome assembly in mitochondria by processing of the ribosomal subunit MrpL32. Moreover, it carries out protein quality control and degrades non-assembled or damaged inner membrane proteins. The relative contribution of both activities for mitochondrial dysfunction and axonal degeneration in HSP remains to be determined. IM, inner membrane; IMS, intermembrane space; M, matrix; OM, outer membrane; OXA, Oxa1 complex; TIM, translocase of inner membrane; TOM, translocase of outer membrane. Tissue-specific assembly of mammalian m-aaa proteases These studies established basic functions of AAA proteases within mitochondria, but left the most puzzling question unanswered: how can the loss of an AAA protease with housekeeping functions within mitochondria be reconciled with the tissue-specific defects observed in HSP patients lacking paraplegin? First evidence for the molecular basis of this phenomenon, came from our studies on the assembly of m-aaa proteases in different murine tissues. In addition to paraplegin and Afg3l2, an additional subunit, Afg3l1, is expressed in mice. We observed the assembly of these m-aaa protease subunits into proteolytic complexes which vary in their subunit composition in different tissues and according to subunit availability. Whereas Afg3l2 and Afg3l1 can homo-oligomerize, we did not obtain any evidence for the homo-oligomerization of paraplegin. Rather, paraplegin was found only as part of hetero-oligomeric complexes also containing Afg3l2, Afg3l1, or both. Complementation studies in yeast documented the activity of the different AAA protease isoforms both in protein turnover and processing. These findings reveal that the loss of paraplegin in HSP does not lead to the loss of m-aaa protease activity per se, but rather to the formation of proteolytic complexes with different subunit compositions. In the absence of paraplegin, homo-oligomeric Afg3l2 complexes replace hetero-oligomeric Afg3l2/paraplegin complex in brain mitochondria suggesting that differences in the substrate specificity of these m-aaa proteases are causative for cellspecific axonal degeneration in HSP. Notably, the relative expression of m-aaa protease subunits varies in different tissues, with Afg3l2 and paraplegin being expressed in significantly higher levels in the brain. This explains why the loss of paraplegin has similar phenotypic consequences in human and mouse, although in the latter with Afg3l1 an additional subunit is expressed. Perspectives Our recent findings revealed versatile activities of AAA proteases within mitochondria and provided first insight into the striking tissue-specificity of HSP. However, other mechanisms of pathogenesis have to be considered. In view of large variations in the mitochondrial proteome in different tissues and the variability of mitochondria within neurons, specific substrates of the paraplegin-containing m-aaa protease may exist which are expressed or may affect the functionality of mitochondria in a tissue-specific manner. Notably, enlarged and structurally abnormal mitochondria in the synaptic terminals of motor axons are the first pathogenic symptoms in paraplegin-deficient mice. Recently, paraplegin has been functionally linked to the processing of the dynamin-like GTPase OPA1, which controls mitochondrial fusion. Interestingly, loss of function mutations in OPA1 is causative for dominant optic atrophy. Thus, mitochondrial dysfunction caused by impaired proteolysis may lead to different but functionally interdependent neurodegenerative disorders. Selected publications Koppen M, Metodiev M, Casari G, Rugarli E and Langer T: Variable and tissue-specific subunit composition of mitochondrial m-aaa protease complexes linked to hereditary spastic paraplegia. Mol. Cell. Biol. (2007) 27, Tatsuta T, Augustin S, Nolden M, Friedrichs B and Langer T: m- AAA protease-driven mem-brane dislocation allows intramembrane cleavage by rhomboid in mitochondria. EMBO J. (2007) 26, Rugarli EI and Langer T: Translating AAA protease function in mitochondria to hereditary spastic paraplegia. Trends Mol. Med. (2006) 12, Nolden M, Ehses S, Koppen M, Bernacchia A, Rugarli EI and Langer T: The m-aaa protease defective in hereditary spastic paraplegia controls ribosome assembly in mitochondria. Cell (2005) 123,
83 Research Area C C 7 Functional Role of PACSIN Proteins in the Regulation of Glutamate Receptors in Healthy and Degenerating Neurons Markus Plomann Institute for Biochemistry II Synapses are specialized intercellular junctions between neurons where signals are propagated from one cell to another with high spatial precision and speed. The basic feature of a chemical synapse is a close apposition of specialised regions of the plasma membranes of the two participating cells to form the synaptic interface. At glutamatergic synapses, two major classes of receptors, NMDA- and AMPA-receptors, are localized in the central and peripheral regions, respectively, accompanied by other proteins needed for the formation and maintenance of the synaptic junction. These scaffolding proteins mediate receptor clustering, regulate receptor function, control receptor internalization and turnover, and coordinate electrical responses induced by neurotransmitter gating of ion channels with longer-lasting cellular responses. Recent findings point to the fact that abnormal synaptic function underlies early symptoms of neurodegenerative diseases and can contribute to the triggering of cell death in later stages. Introduction Huntington s disease (HD) is clinically characterized by insidious onset and a slowly progressive course. Individuals with HD, typically show marked specific neuronal loss in the neocortex and a defined region of the basal ganglia, namely, the neostriatum. Neuropathological abnormalities and onset of symptoms occur prior to cell loss, suggesting that cortical neuronal dysfunction may underlie some critical aspects of the HD phenotype. Experiments using transgenic HD mouse models have noted alterations in long-term potentiation (LTP), long term depression (LTD) and neurotransmitter levels within the cortex and striatum of HD models. The mechanisms of pathogenesis and cellular specificity have remained elusive, aberrant interactions of the affected mutant huntingtin with target proteins have been suggested as a potential cause. Mutant huntingtin has been demonstrated to form protein aggregates in vitro, and a variety of proteins including wild type huntingtin are sequestered within aggregates in cells. Experiments using transgenic HD mouse models have demonstrated alterations in synaptic plasticity and neurotransmitter levels within the striatum of HD models which may lead to excitotoxicity. This possibility is particularly intriguing since excitotoxicity is discussed as a potential mechanism for HD. The theory of excitotoxicity also raises the issue of synaptic function and neurotransmitter release in HD. Regulatory proteins with altered distribution or expression in Huntington s disease We earlier had identified PACSIN 1, which specifically interacts with huntingtin, like the endophilins, via its C-terminal SH3 domain. This interaction is glutamine repeat-length dependent and is enhanced by mutant huntingtin, possibly causing the sequestration of PACSIN 1. Normally, PACSIN 1 is located along neurites and within synaptic boutons (Figure 1), but in HD patient neurons, there is a progressive loss of PACSIN 1 immunostaining in synaptic varicosities beginning in presymptomatic and early stage HD. Fig. 1: PACSIN 1 localization in synapses. Cultured hippocampal neurons show endogenous PACSIN 1 (red) colocalized with AMPA receptors (green) in synaptic boutons. Further, PACSIN 1 immunostaining of HD patient tissue revealed a predominantly cytoplasmic distribution of the protein with particular concentration in the perinuclear region, coincident with mutant huntingtin. Thus, the specific interaction of huntingtin with the neuronal PACSIN isoform, PACSIN 1, and its altered intracellular distribution in pathological tissue together with the observed differences in the binding behavior suggest a role for PACSIN 1 during early stages of the selective neuropathology of HD (Modregger et al. 2002, DiProspero et al. 2004). PACSIN 1 specifically interacts with the NR3A chain of the NMDA receptor in young synapses and regulates its surface expression (Pérez-Otaño et al. 2006). We now have evidence that PACSIN 1 also participates in the recycling of AMPA receptors in mature neurons (Figure 1). In healthy neurons synaptic plasticity is a fundamental process in learning and memory. AMPA receptors have been shown to be one of the keyplayers in these processes and their regulation has been extensively studied. We and others could show that the protein kinase C (PKC) plays a special role in cerebellar long term depression and associates with the GluR-binding protein PICK1 (Leitges et al. 2004). Furthermore, PACSIN 1 is present in complexes with PKC and AMPA receptors. In a mouse model for HD we observed a drastic decrease in the expression levels for PAC- SIN 1, PKC and AMPA receptors (Figure 2). 78
84 Research Area C Fig. 2: Reduced expression of AMPA receptor complexes in HD mouse model. Cerebellar sections of normal mice (top) and HD mice (bottom) were stained for endogenous PKC (green), AMPAR (blue) and PACSIN 1 (red). A marked reduction is seen in Purkinje cells. Experiments using transgenic HD mouse models have demonstrated alterations in synaptic plasticity and neurotransmitter levels within the striatum of HD models which may lead to excitotoxicity. Changes in intracellular trafficking in HD models To gain insights into the early pathological changes in HD we established cell models, in which mutant huntingtin fragments containing extended polyglutamine repeats are expressed upon induction. The broad reticular pattern of PACSIN 1 localization throughout the cytosol of control cells is shifted towards a more restricted distribution in the periphery of PC12 cells inducibly expressing a huntingtin exon 1 protein with a pathological polyglutamine expansion. (Figure 3). and vesicles. The ability of PACSINs to form dimers and tetramers enables PACSIN aggregates to interact in parallel with several SH3 domain binding partners. PACSINs also act as linkers between the endocytic protein dynamin and the actin regulator N-WASP, thereby directing the actin propulsion machinery to the site of vesicle fission. This role might be important in dendritic spines, where PACSIN 1 might act at recycling vesicles containing glutamate receptors and regulates their postsynaptic membrane localization and/or removal (Pérez-Otaño et al. 2006). Abnormal localizations of specific proteins appear to be a common phenomenon in degenerative diseases (Laser et al. 2006). In HD this relocalization in combination with the reduced expression of other important key factors may contribute to excitotoxic processes at presymptomatic stages. Perspectives We suggest that a deficient presynaptic neurotransmitter release as well as an impaired recycling of postsynaptic neurotransmitter receptors may underlie central symptoms at the early and mid-stage of HD, like cognitive defects, dementia and hypokinesia. As excitotoxicity is one process proposed to be involved in early pathological changes in HD neurons, it is intriguing to speculate that the synaptic removal of PACSIN 1 impairs proper regulation of glutamate receptors and that this may gradually lead to the observed toxic effects in individual cells. Selected publications Modregger J, DiProspero N, Charles V, Tagle DA and Plomann M: PACSIN 1 interacts with huntingtin and is absent from synaptic varicosities in presymptomatic Huntington s disease brains. Hum Mol Gen (2002) 11, Leitges M, Kovac J, Plomann M and Linden DA: Unique PDZ ligand in PKCα confers induction of cerebellar longterm synaptic depression. Neuron (2004) 44, DiProspero N, Chen EY, Charles V, Plomann M, Kordower JH and Tagle DA: Early changes in Huntington s disease patient brains involve alterations in cytoskeletal and synaptic elements. J Neurocytol (2004) 32, Fig. 3: PACSIN 1 redistribution in cells expressing pathological huntingtin. Endogenous PACSIN 1 is relocated from a broad reticular distribution in uninduced cells (left) to a more peripheral localization 72 h after induction. The neuron-specific PACSIN 1 protein is involved in endocytosis on both, pre- and postsynaptic membranes. Via its C-terminal SH3 domain, PACSIN 1 binds to proline-rich domains of dynamin, synapsin, synaptojanin and N-WASP, four proteins involved in vesicle endocytosis and transport. In presynaptic vesicle endocytosis PACSIN 1 contributes to the formation of membrane invaginations Laser H, Conforti L, Morreale G, Mack TGM, Heyer M, Haley JE, Thomas M, Wishart TM, Beirowski B, Walker SA, Haase G, Celik A, Adalbert R, Wagner D, Grumme D, Ribchester RR, Plomann M, and Coleman MP: The slow Wallerian degeneration protein, WldS, binds directly to VCP/p97 and partially redistributes it within the nucleus. Mol Biol Cell (2006) 17, Pérez-Otaño I, Lujan R, Tavalin SJ, Plomann M, Modregger J, Liu X-B, Jones EG, Heinemann SF, and Ehlers MD: Endocytosis and synaptic removal of NR3A-containing NMDA receptors by PAC- SIN1/syndapin1. Nat Neurosci (2006) 9,
85 Research Area C C 8 In-Depth Analysis of Axonal Guidance by Netrins Manuel Koch Institute for Biochemistry II During development, a complex network of connections is made between the neurons to form the nervous system. These wirings emerge as neuronal growth cones are guided through their environment by extracellular matrix proteins and cell surface molecules. The aim of this project is to dissect the function of netrins and other extracellular matrix proteins during development and in diseases. Introduction Extracellular matrix proteins are essential for the structure of all multicellular organisms. These proteins provide the physical properties typical for tissues and can be seen as polymers, which form the structure around the cells. Furthermore, these proteins help to establish barriers between tissue compartments. Basement membranes, which are found underlying all endothelia and epithelia, form such structures. Genetic defects in human and mouse nicely demonstrate the importance of these barriers. Mutations in one of the components, laminin, cause blistering disease in human as well as muscular dystrophies. Beside this important structural function, the matrix also plays a crucial role during development. For example, the correct wiring of the nervous system depends on extracellular matrices as well as on their cell surface receptors. It is thought that axons, which send out their processes, migrate along a permissive track towards an attractive source. The growth cones express cell surface receptors that enable them to migrate on the extracellular matrix. In addition, the axons are not only guided by attractive but also repulsive cues and such signals are mediated by extracellular matrix associated proteins (e.g. netrins, slit, semaphorins) or by cell surface molecules (e.g. ephrins). It is thought that only about 400 nervous system related molecules are necessary in the extracellular space to build a nervous system in a vertebrate. Effects of netrin-4 on neurite outgrowth Because of the similarity in structure of netrin-4 to netrins 1 3, and because netrin-4 expression was detected in the output pathways of the olfactory bulb and the retina, we tested the ability of netrin-4 to promote neurite outgrowth. Comparison of the domain structure of netrins and laminin-111. All netrins share homology to the N-terminal LN and LEa domains of laminin chain short arms. Netrin-4 contains a laminin-like LN domain, 3.5 LE repeats, and a netrin C domain. Netrin-4 produces neurite outgrowth from olfactory bulb explants. Representative cultures treated as indicated. Treatment with full-length or truncated netrin-4 proteins increases the number of neurites produced by the explants. Protein additions are noted below the bars. 80
86 Research Area C Explants of E15 rat olfactory bulb were incubated with soluble recombinant netrin-4. In control cultures, neurites extend around the circumference of the explants. Treatment with netrin-4 increases both the neurite length and the number of neurites extending from cultures. With the addition of netrin-4, there was a dose-dependent increase in both the sum length of all neurites and the number of neurites (normalized to explant circumference) of up to 350% of control measurements. The addition of affinity-purified antibodies against netrin-4 antagonized this effect, reducing neurite length and number to control levels. Netrin-4 is localized in basement membranes Netrin-4 is widely expressed outside the nervous system, most abundantly in vasculature, kidney, ovary, and heart, and is often deposited in basement membranes. Netrin-4 interactions with laminin LN domains It is well known that laminins from polymers through their LN domains. Since netrin-4 also contains such a domain we speculated that the integration into the basement membrane is mediated through the LN domain. Several binding assays based on different principles revealed strong interactions between netrin-4 and both the laminin γ1 chain and the whole, trimeric laminin-111. In contrast, netrin-1 does not bind to either protein. Ca2+ ions are not required for the interaction and circular dichroism spectroscopy showed no detectable conformational change in netrin-4 induced by adding Ca2+ or removing divalent cations with EDTA. Since the laminin short arm self-interaction is Ca2+ dependant, it appears that netrin-4 binding to laminin-111 does not occur through the same interaction site. Biacore binding studies showed that the netrin-4 LN domain is crucial for interaction with the laminin γ1 subunit. Perspectives Most studies on netrins are on their role in neuronal guidance within the CNS. Our functional analysis of netrin- 4 in the nervous system in mutant mice is still ongoing. As pointed out, netrins are also widely expressed outside the nervous system and their functions in these tissues are not known. In the future, we will expand our research project and will also include the study of the role of basement membranes. The netrin-4 localization in newborn and adult mouse kidney was visualized by indirect immunofluorescence staining. Positive signals were detected in the glomerular (gl) basement membrane and in vasculature (bl). In the adult kidney netrin-4 localizes also in the tubules (tu). Co-localization studies in mouse kidney and salivary gland revealed overlapping spatial expression patterns of the laminin γ1 chain and netrin-4 in basement membranes of post-natal and adult animals. However, the temporal regulation is independent, as early embryonic expression does not show extensive co-localization. Netrin-4 expression increases from the newborn to the adult animal. Netrin-4 affects branching morphogenesis To analyze the effect of netrin-4 on epithelial branching, the salivary gland was studied. The salivary gland, which also depends upon branching morphogenesis for its development can be studied in vivo. Since netrin-4 is expressed first at late stages of salivary gland developmental this is an excellent model to study the effect of exogenous netrin-4. Treatment of submandibular gland explants with netrin-4 (50 µg/ml) lead to a drastic suppression of epithelial branching. Significantly, this effect was achieved using the full-length netrin-4 protein and tnetrin-4, the truncated form of netrin-4 lacking the C domain. Selected publications Koch M, Murrell JR, Hunter DD, Olson PF, Jin W, Keene DR, Brunken WJ, Burgeson RE: A novel member of the netrin family, beta-netrin, shares homology with the beta chain of laminin: identification, expression, and functional characterization. J. Cell Biol. (2000) 151, Liu Y, Stein E, Oliver T, Li Y, Brunken WJ, Koch M, Tessier-Lavigne M, Hogan BL: Novel role for netrins in regulating epithelial behavior during lung branching morphogenesis. Curr. Biol. (2004) 14, Mechai N, Wenzel M, Koch M, Lucka L, Horstkorte R, Reutter W, Danker K: The cytoplasmic tail of the alpha3 integrin subunit promotes neurite outgrowth in PC12 cells. J. Neurosci. Res. (2005) 82, Veit G, Kobbe B, Keene DR, Paulsson M, Koch M, Wagener R: Collagen XXVIII, a novel von Willebrand factor A domaincontaining protein with many imperfections in the collagenous domain. J. Biol. Chem. (2006) 281, Brandt-Bohne U, Keene DR, White FA, Koch M: MEGF9: a novel transmembrane protein with a strong and developmentally regulated expression in the nervous system. Biochem. J. (2007) 401, Maertens B, Hopkins D, Franzke CW, Keene DR, Bruckner- Tuderman L, Greenspan DS, Koch M: Cleavage and oligomerization of gliomedin, a transmembrane collagen required for node of Ranvier formation. J. Biol. Chem. (2007) 282,
87 Research Area C C 9 Molecular Pathomechanisms of a Skeletal-Muscle-Specific Lethal Mutation of Mitochondrial DNA in a Muscle Specific Cybrid Model Rudolf J. Wiesner 1 / Jürgen-Christoph von Kleist-Retzow 2 Institute for Vegetative Physiologie 1 / Department of Pediatrics 2 We studied how pathogenic mt DNA point mutations affects energy producing pathways, oxidative stress response and Ca 2+ homeostasis in model cell lines. The defect in oxidative phosphorylation was completely compensated by an appropriate rise of anaerobic glycolysis, so that the final total ATP production rate was preserved. This rise of glycolytic flux was enabled by a decrease of cellular ATP-levels and of the ATP/ADP+AMP ratio. Increased production of reactive oxygen species (ROS) was fully compensated by cellular antioxidant defence systems, so no persistent damage to proteins and lipids was found. However, intramitochondrial Ca 2+ homeostasis was disturbed due to a significant decrease in the mitochondrial inner membrane potential. We postulate that this, and not an energetic deficit or increased oxidative damage, most likely is a major pathomechanism in mitochondrial diseases. Introduction Disturbances in the energy generating capacity have been accused for a long time to account essentially for the clinical phenotypes of respiratory chain dysfunction, in particular as lactic acidemia is one of the hallmarks of mitochondrial disease in patients. However, elevated lactate levels are not mandatory at all and the most energy demanding tissues, e.g. the central nervous system and the heart, are not universally affected. In order to further elucidate the pathophysiologic consequences of mtdna mutations downstream of mitochondrial transcription, protein translation and respiratory chain function, we used cybrid model cells incorporating patient derived mitochondria. We used a set of four 143B osteosarcoma cell lines, two of them with point mutations in trna Leu(UUR), and compared those to two control cell lines, the acceptor wild-type cell line, and Rho0 cells, completely devoid of mtdna. The 3243A>G mutation represents the classical MELAS mutation, by far the most common mtdna point-mutation, which has been identified in surprisingly variable clinical contexts, while the other mutation, 3302A>G, has been observed only in patients presenting with a severe isolated myopathy, which is ultimately lethal. Enhancement of anaerobic glycolysis compensates failing oxidative phosphorylation Measurement of oxygen consumption and lactate release allowed us to estimate the ATP production rate via these two major pathways, oxidative phosphorylation and anaerobic glycolysis. We calculated that the sum of ATP generated by both pathways is similar in all cell lines (Fig. 1). In keeping with this, both cybrid as well as Rho0 cells lines retain their capacity to proliferate in culture as fast as their parental 143B control cell line (not shown). Fig. 1: ATP production rate during exponential growth Decrease in Adenine Nucleotide steady state levels permits the high glycolytic flux We found that total adenine nucleotide levels (TAN), mostly ATP, are drastically lowered in all respiratory chain (RC) deficient cell lines (Fig. 2), however a high energy charge potential is maintained (Fig. 2, Inset II). Fig. 2: Adenine nucleotide steady state levels TAN: Total Adenine-Nucleotides (= AMP+ADP+ATP); Inset I.: ratio of AMP + ADP / ATP in percent; Inset II.: Energy charge potential (ECP) [= ATP + (0,5 x ADP) / (AMP + ADP + ATP)] ns: p > 0,05; *: p 0,05; **: p 0,01; ***: p 0,001 82
88 Research Area C The remarkable decrease in TAN is unlikely the result of an impaired capacity for synthesis, as the amount of purine bases needed for the synthesis of the TAN pool is negligible compared to the amount needed for production of total DNA and RNA in a proliferating cell (only about 1% for TAN synthesis). However, while cellular proliferation is obviously not impaired in RC-deficient cells, they show a slight but significant increase of the ratio of AMP+ADP/ATP (Fig. 2, Inset I). The rate limiting enzyme of glycolysis, phosphofructokinase, is tightly regulated through allosteric inhibition by ATP and activation by AMP. We assume that the estimated duplication of glycolytic ATP production rate seen in all RC-deficient cell lines is enabled by an adaptive, intentional down-regulation of the nucleotide pools, consisting mainly of ATP, and a shift in their relative ratios. Consequently, the cytosolic ATP turnover can be considerably increased (estimation: x 4) but this apparently does not cause any major problem for glycolytic ATP production. Impaired mitochondrial Calcium homeostasis Mitochondrial Ca ++ handling was studied using mitochondrially targeted aequorine. We recorded a characteristic pattern of luminescence after stimulation with the IP3- generating agonist histamine: all cell lines showed a first peak which was followed, however only in RC-deficient cell lines, by a second peak (Fig. 3). We hypothesize that the first peak is due to Ca ++ flowing into mitochondria located closely to the sites of the endoplasmic reticulum where IP3 liberates Ca ++. This signal is most likely saturated and therefore its amplitude is not proportional to the amount of Ca ++ taken up by mitochondria. Accordingly, if present, the second peak should originate from mitochondria located more distantly from the release sites. These mitochondria are expected to be exposed to lower Ca ++ levels and with an appreciable delay due to slow cytoplasmic Ca ++ spreading. While mitochondria in 143B control cells were apparently capable of handling the Ca ++ load with a single response revealed by a single luminescence peak, in all 3 other cell lines this first response was followed by a second peak and luminescence returned to baseline with an appreciable delay. This indicates a disturbance in the Ca ++ -handling properties and/or mitochondrial-er interactions in RC-deficient cell lines. Conclusion Despite major respiratory chain dysfunction, the cellular growth potential is basically preserved in the cybrid lines incorporating trna Leu(UUR) mutations as well as in Rho0 cells. Lactate production is considerably enhanced showing that ATP generated by anaerobic glycolysis apparently allows for sufficient energy supply to keep up with the demands of proliferating cells. A dramatic decrease in total adenine nucleotide steady state levels, with preserved energy charge potential, most likely permits an appropriately high glycolytic flux. This underlines that the impairment of aerobic ATP production by RC-dysfunction apparently does not result inevitably in energy deficiency, and that low ATP steady state levels do not necessarily reflect a critical defect of cellular energy supply. On the other hand, we identified a disturbed mitochondrial Ca ++ homeostasis due to a lowered mitochondrial membrane potential in cells with a respiratory chain defect. These findings probably also mirror the situation under many circumstances in patients with mitochondrial disease. Perspectives Currently, there are almost no options for treatment of mitochondrial diseases, thus it is of major importance to understand the impact of RC-dysfunction on cellular physiology. Our findings are supported by recent observations in tissue specific k.o. animal models, which show that the absence of the respiratory chain can be tolerated for long time periods. Thus, other processes like Ca ++ homeostasis seem to be critically affected and represent new and promising targets for treatment. Selected publications Möllers M, others, Wiesner RJ, von Kleist-Retzow JC. Impairment of post-transcriptional maturation and severe depletion in mitochondrial trna Ser(UCN) caused by T7512C and G7497A point mutations. Nucl Acids Res (2005) 33, Thierbach M, others, von Kleist-Retzow JC, Wiesner RJ, others, Ristow M. Targeted disruption of hepatic frataxin expression causes impaired mitochondrial function, decreased life span and tumor growth in mice. Hum Mol Genet (2005) 14, Maniura-Weber K, others, von Kleist-Retzow JC, Wiesner RJ. Molecular dysfunction associated with the human mitochondrial 3302A>G mutation in the MTTL1 (mt-trna Leu(UUR) ) gene. Nucl Acids Res (2006) 34, Fig. 3: Mitochondrial Ca ++ handling (mt-aequorine) following an intracellular Ca ++ transient evoked by histamine. Mean of all luminescence curves for the cell lines as indicated. 83
89 Research Area C C 10 Spinal Muscular Atrophy (SMA): Drug Validation for Therapy and Identification of Disease Modifying Genes Brunhilde Wirth / Eric Hahnen Institute for Human Genetics Spinal muscular atrophy (SMA) is caused by the loss of the survival motor neuron gene 1 (SMN1) function. A nearly identical copy of SMN1, SMN2, modulates the disease severity and represents a promising therapeutic target. We identified histone deacetylase inhibitors (HDACis) to increase SMN2-derived SMN protein levels. A pilot trial in SMA carriers/patients with the HDACi valproic acid provided a first in vivo evidence for the efficiency of HDACis for SMA treatment. Finally, we identified a novel SMN1/2 unrelated gene that might protect individuals from developing SMA. Introduction Proximal spinal muscular atrophy (SMA) is the leading genetic cause of death in infants and toddlers. SMA is one of the most common autosomal recessive diseases with an incidence of approximately 1 in 6,000 live births and a carrier frequency of 1:35. The molecular basis of SMA is the homozygous absence or loss of function of SMN1, whereas the disease severity is mainly modulated by the copy number of SMN2 genes and in rare cases, by other, yet not known modifying factors. SMN has an important function in many pathways: snrnp biogenesis and assembly of the spliceosome, pre-mrna splicing, transcription, translation and axonal transport of RNA along the axons. Both SMN genes (SMN1 and SMN2) are ubiquitously expressed and encode identical proteins, but differ in their splicing patterns: While SMN1 produces only fulllength (FL) transcripts, SMN2 transcripts mainly lack exon 7. The skipping of exon 7 is caused by a single silent mutation within an exonic splicing enhancer (ESE) in exon 7 of SMN2. We have shown that Htra2-ß1 (an SR-like splicing factor) together with two hnrnp proteins (hnrnp-g and RMBX) bind to an additional ESE in exon 7, and are responsible for about 10% of correctly spliced SMN2 mrna. Over-expression of these splicing factors restores the correctly spliced FL-SMN2 mrna to almost 80%. From both, SMA patients and transgenic SMA-like mice, we learned that the SMN2 copy number mainly determines the severity of the SMA phenotype. This suggests that an increase in SMN protein level will be beneficial for SMA patients. Therefore, drugs that may activate and thus increase the amount of endogenous protein produced by the SMN2 gene copies in SMA patients may be beneficial for SMA patients. The identification of SMA modifying factor(s) may open new therapeutic perspectives. SMN2 is up-regulated by histone deacetylase inhibitors Transcriptional activation and repression is a dynamic process in all our cells and is influenced by many factors, the most important one is the structure of the chromatin. DNA together with histones forms the chromatin that has an open or relaxed structure when histones are acetylated and a closed or condensed structure when the acetyl groups are removed. Histone acetyl transferases (HATs) add acetyl groups to the histones and enable transcription, while histone deacetylases (HDACs) remove acetyl groups and repress transcription. Meanwhile, an increasing number of compounds that inhibit the activity of HDACs were identified. Some of these drugs are already used in human therapy or under clinical phase II/III investigation, while others are still at the beginning of the drug development process. We have been able to show that the transcription and restoration of correct splicing of SMN2 can be increased by three HDAC inhibitors (HDACi) belonging to different chemical categories: fatty acids (valproic acid, VPA), hydroxamic acids (suberoylanilide hydroxamic acid, SAHA) and benzamides (M344). All three compounds showed significant up-regulation of FL-SMN2 transcripts, SMN protein and number of gems/100 cells following treatment of fibroblast cell lines derived from SMA patients, organotypic hippocampal brain slice cultures originated from rats and humans (from epilepsy patients after surgery) and motor neuron enriched cell fractions derived from E15 rats (Fig 1). Fig. 1: VPA markedly increases SMN protein levels in: (A) SMA patient-derived fibroblast cell lines (SMN1-deleted), (B) human hippocampal brain slice cultures (derived from epilepsy surgery), and (C) motor neuron enriched cell cultures (isolated from rat embryos). Development of a biomarker Of particular relevance was the development of a biomarker for the measurement of the SMN2 transcription levels in SMA patients and carriers. This was an extremely demanding task because most genes in blood show high intra-individual expression variability in addition to the variability caused by HDACi. It is known that about 2% of the human transcripts change their expression under HDACi treatment. Based on a real-time RT-PCR on a LightCycler machine, we have been able to develop a highly reliable 84
90 Research Area C method to quantify FL- and Δ7-SMN2 transcript levels in blood. By analyzing blood samples from type I, II and III SMA patients we found out that there is no clear correlation between amount of full-length SMN-RNA levels and SMN2 copy number or severity of SMA, in contrast to primary fibroblast cell lines. Nevertheless, blood is the only material, which can repetitively be obtained before and during a clinical trial. Pilot trial in SMA carriers and patients with valproic acid Analyzing FL-SMN2 and SMN protein levels in 10 SMA carriers (Fig. 2) and 20 type I-III SMA patients treated with VPA, we have been able to show significantly increased FL-SMN levels in about 1/3 of patients. However, 1/3 of patients did not show any changes and 1/3 were negative-responders. These data clearly provide a first proof of principle on one hand, but also show the need for further research on drug discovery for more efficient drugs. Furthermore, our results clearly demonstrate the usefulness of testing a biomarker along with the evaluation of clinical out-come measures in further clinical trial. Based on our results a large clinical trial in type II and III SMA patients with VPA has been started in the USA and a further one is planed in type I SMA patients in Europe. tissue affected in SMA patients. Knock-down of the modifier by sirna in NGF stimulated PC12 cells severely affected the axonal growth, while overexpression stimulated the axonal growth. Simultaneous knock-down of Smn and over-expression of the modifier partially rescued the axonal growth. Analysis of the coding region, promoter and haplotype blocks by sequencing, methylation and single nucleotide polymorphism analysis strongly supported a trans-regulation of the expression of the modifier and excluded a differential expression due to cis-regulating domains. Perspectives The project will focus on the evaluation of several drugs, which we already showed to be effective in increasing SMN levels in SMA animal models. Furthermore, we will search for the signaling pathway responsible for the differential response under VPA by comparing the expression pattern on Affymetrix chips between positive, negative and non-responders. We will search for the modifications in the SMN promotor region produced by HDAC inhibitor treatment by using chromatin immunoprecipitation assays as well as by further epigenetic modifications of DNA methylation. Further funding for the analysis of the modifying gene will be applied from the Deutsche Forschungsgemeinschaft. Selected publications Sun Y, Grimmler M, Schwarzer V, Fischer U, Wirth B: Molecular and functional analysis of intragenic SMN1 mutations in patients with spinal muscular atrophy. Hum Mut, (2005) 25, Fig. 2: VPA activates the transcription of SMN protein levels in human. Western blot analysis of SMA carriers treated with VPA. Pilot trials in SMA animal models Of particularly high value is the availability of SMA-like mice (Smn -/- SMN2 +/+ ) for the investigation of the pathomechanisms and motor neuron cell death in SMA on one hand, and the step-by step analysis of various therapeutical drugs in these animals on the other hand. We expanded SMA transgenic/knockout mice in order to obtain a reasonable number of heterozygous mice (Smn +/- SMN2 +/+ ) which will serve for further studies of new drugs. The evaluation of SAHA for SMA treatment using SMA-like mice is currently ongoing. Identification of a SMA modifying gene(s) Based on a differential expression analysis using 6 phenotypically discordant SMA families having siblings with identical SMN1 mutations, SMN2 copies and 5q13 haplotypes but different phenotypes (SMA vs unaffected), we identified a first modifying gene in the unaffected siblings. All unaffected individuals are females and express the modifier as compared to their affected sibling. The gene is highly expressed in spinal cord, the major Brichta L, Holker I, Haug K, Klockgether T, Wirth B: In vivo activation of SMN in spinal muscular atrophy carriers and patients treated with valproate. Ann Neurol. (2006) 59, Hahnen E, Eyupoglu IY, Brichta L, Haastert K, Trankle C, Siebzehnrubl FA, Riessland M, Hölker I, Claus P, Romstock J, Buslei R, Wirth B, Blumcke I: In vivo and ex vivo evaluation of second-generation histone deacetylase inhibitors for the treatment of spinal muscular atrophy. J Neurochem (2006) 98, Riessland M, Brichta l, Hahnen E, Wirth B: The benzamide M344, a novel histone deacetylase inhibitor, significantly increases SMN2 RNA/protein levels in spinal muscular atrophy (SMA) cells. Hum Genet (2006) 120, Wirth B, Brichta L, Schrank B, Lochmüller H, Blick S, Baasner A, Heller R: Mildly affected patients with spinal muscular atrophy are partially protected by an increased SMN2 copy number. Hum Genet (2006) 119,
91 Research Area C C 11 Comprehensive Genetic Analysis of the Dopaminergic and Serotonergic Systems in Migraine with Aura Christian Kubisch Institute for Human Genetics Migraine with aura (MA) is a genetically complex brain disorder of unknown etiology. There is physiological evidence for an involvement of the serotonin and dopamine neurotransmitter systems in MA pathogenesis, which however has not been systematically tested in genetic studies. We are therefore comprehensively analyzing the genes encoding the neuronal subunits of these systems by linkage, association, or mutation screening approaches in a very large MA patient and family sample. Introduction Migraine is a common CNS disorder characterized by attacks of severe headache and reversible autonomic and neurological symptoms. The lifetime prevalence is about 15% with a pronounced female preponderance. The pain strongly disables the migraineur in his/her private and professional life causing a major loss in life quality and productivity. Migraine is a leading cause of chronic use/abuse of analgesic drugs (approx. 4 billion tablets per year in Germany) and concomitant secondary medical complications (e. g. renal insufficiency) and shows considerable comorbidity with other neuro-psychiatric disorders (e. g. epilepsy and depression). Disease definition and genetic epidemiology The two main types of migraine are migraine without aura (MO) and migraine with aura (MA) representing about 75-80% (MO) and 20% (MA) of all cases. The headache is typically unilateral, pulsating, and aggravated by physical activity. It is often accompanied by nausea, vomiting, or photo- and phonophobia. The attacks in MA are preceded by an aura consisting of reversible visual (99% of patients, see Figure 1), sensory (31%), and aphasic (18%) symptoms usually lasting less than 60 minutes. Fig. 1: Typical visual aura in MA with fortification spectrum as drawn by a MA patient. There is no causal cure for this chronic disorder, yet, migraine is in principle treatable by a variety of drugs. The introduction of triptans, a group of drugs modifying the serotonin pathway, means a major relief for many patients. Nevertheless, there is still a need for better drugs with less adverse effects since a significant number of patients cannot be treated sufficiently up to now. Twin and family studies prove a high degree of genetic determination in migraine with an estimate of heritability of about 0.65 for MA. Neurotransmitter systems in MA No major causative genes for complex forms of migraine have been identified, however, for both the serotonergic and dopaminergic neurotransmitter systems there are convincing clinical, experimental, and pharmacological arguments to support a causative involvement in disease etiology. For example, during a migraine attack both platelet release of serotonin and urinary excretion of serotonin-breakdown products are increased. Moreover, the serotonergic brainstem nuclei locus coeruleus and nucleus raphe represent migraine generators. Last but not least, a number of serotonin receptors are the principle targets of drugs alleviating migraine symptoms and especially the most potent migraine drugs, i. e. the triptans, are serotonin 1B/1D-receptor agonists. Likewise, there are also good experimental/clinical data concerning a role of dopamine in MA pathogenesis and migraine symptoms like nausea and vomiting specifically support the involvement of brain stem dopaminergic neurotransmission in migraine attacks. Moreover, dopamine antagonists are effective in suppressing and preventing migraine symptoms. Despite this convincing functional evidence, there is a lack of genetic studies applying up-to-date technology testing the serotonergic and dopaminergic genes in migraine etiology. Patient sample During the last years, we have collected one of the world largest patient and family samples for the systematic genetic study of migraine applying the revised diagnostic criteria of the International Headache Society, comprising (i) more than 100 multiplex families with several affected 86
92 Research Area C persons for linkage and association studies, (ii) 150 caseparent trios for family-based association studies, and more than 650 cases and 650 controls for association studies. Both the family and the association samples have a statistical power of >95% to identify or exclude larger genetic effects. In the family sample, a genome-scan with approx. 450 microsatellite markers is currently performed and analyzed (collaboration with the Cologne Centre for Genomics). We could also show by identifying and functionally characterizing missense alterations in highly conserved residues of the ATP1A2 gene in families with clustering of MA/MO that rare genetic variants confer susceptibility to complex forms of migraine. Genetic studies in neurotransmitter systems We are now systematically analyzing the neuronal subunits of the dopaminergic and serotonergic pathways by a combination of complimentary genetic methods including linkage, association and mutation screening in our patient sample. Together, the genes analyzed span more than 1 MB of genomic sequence making this study one of the largest systematic association studies in migraine genetics. To investigate the extended loci and their genetic variability in a systematic way, we use the data from public genome databases (like HapMap) to determine the genetic variability and linkage disequilibrium structure. Using these data, we divide the loci into haplotype blocks (an example of this is given in figure 2 for the dopamine receptor gene type 2). neurotransmitter systems. Indeed, we could for example already exclude a role of the serotonin transporter gene SLC6A4 in the molecular etiology of MA whereas we could find good evidence for a causative involvement of several components of the dopaminergic system in disease pathogenesis. For example, we obtained allelic P-values in the order of 5.0 x 10-4 for a combined sample of 650 cases and 650 controls for an SNP in the dopamine beta-hydroxylase gene which could also be confirmed in a haplotype-based association study. Further genetic studies are currently performed to identify the most significantly associated haplotypes/polymorphisms and to possibly identify the causative genetic variant(s). If feasible, additional functional studies will be performed to e. g. analyze whether the splicing pattern or expression level is changed or - in case the associated variant alters an amino-acid - biochemical or electrophysiological assays will be performed. Perspectives Our complimentary genetic approaches in one of the world largest patient and family samples with migraine with aura will enable us to define or exclude the role of genes in the dopamine and serotonin pathways in the pathogenesis of this common brain disorder. Furthermore, pharmacogenetic and functional analyses of associated genetic variants may help to better understand the molecular basis of this neuronal hyperexcitability and to improve the medical management of migraineurs by defining the individual risk basis more specifically. This genetic approach can also be applied for different physiological pathways and systems and in the long term on a genome-wide basis. Selected publications Todt U, Dichgans M, Jurkat-Rott K, Heinze A, Zifarelli G, Koenderink JB, Goebel I, Zumbroich V, Stiller A, Ramirez A, Friedrich T, Göbel H, Kubisch C. Rare missense variants in ATP1A2 in families with clustering of common forms of migraine. Hum Mutation (2005) 26: Netzer C, Todt U, Heinze A, Freudenberg J, Zumbroich V, Becker T, Goebel I, Ohlraun S, Göbel H, Kubisch C. Haplotype-based systematic association studies of ATP1A2 in migraine with aura. Am J Med Genet (2006) 141B, Fig. 2: Representation of linkage disequilibrium (LD) structure in the DRD2 region. Pairwise D -values between markers are shown (red fields without numbers denote complete LD with D = 1; weaker LD is encoded by light red or white shading). Note the presence of three distinct haplotype blocks in this region. Block partitioning requires that at least 90% of total haplotype diversity is covered by common haplotypes. We select those SNPs necessary for discrimination of the different haplotypes as tagging SNPs, altogether we genotype SNPs to completely cover both Todt U, Freudenberg J, Goebel I, Netzer C, Heinze A, Heinze-Kuhn K, Göbel H, Kubisch C. The MTHFR C677T polymorphism and migraine with aura. Ann Neurol (2006) 60, Todt U, Freudenberg J, Goebel I, Heinze A, Heinze-Kuhn K, Rietschel M, Göbel H, Kubisch C. Variation of the serotonin transporter gene SLC6A4 in the susceptibility to migraine with aura. Neurology (2006) 67, Netzer C, Freudenberg J, Toliat MR, Heinze A, Heinze-Kuhn K, Thiele H, Goebel I, Nürnberg P, Ptáček LJ, Göbel H, Todt U, Kubisch C. Genetic association studies of the chromosome 15 GABA-A receptor cluster in migraine with aura. Am J Med Genet Part B (2007), in press. 87
93 Core Units CBA Central Bioanalytics Franz-Georg Hanisch In extension of standard analytical services the Central Bioanalytics (CBA) offers state-of-the-art technical equipment and skill in protein analysis, particularly in the analysis of post-translationally modified (PTM) proteins. Our method developments resulted in a couple of new service offers and is still ongoing in the context of differential proteomics applications covering gel-based (2D-DIGE) and multidimensional LC-based strategies (SILAC). New mass spectrometric equipment, the HCT-ultra with the Electron-Transfer-Dissociation technology, will provide a technological platform for the site-specific analysis of labile protein modifications, like phosphorylation and O-glycosylation Introduction The Central Bioanalytics in the CMMC offers a variety of standardized analytical services to customers of the CMMC, including the automated sequencing of DNA and peptides, and the analysis of proteins or peptides by mass spectrometry. During 2002/2003 we were able to build-up the apparative instrumentation of a proteomics workstation and to establish methodological and technical protocols in the performance of gel-based proteomic applications. The instrumentation covers equipment for 2D gel electrophoresis of proteins, UV- and fluorescence scanners for gel imaging, software for the quantitative analysis of protein spots, robots for the picking of protein spots and for the in-gel digestion of proteins prior to their mass spectrometric analysis. Protein identification is performed by peptide mass fingerprinting (PMF) on a Reflex IV MALDI-TOF mass spectrometer (Bruker) or by partial sequencing of peptides on a Waters QTOF2 electrospray mass spectrometer (MS/MS mode) coupled online with a nanoflow LC from LC Packings. Beyond these standardized applications the CBA offers a variety of special service in the analysis of post-translationally modified proteins. Service Statistics Service in DNA sequence analysis stabilized in the order of samples per year. Total numbers in protein samples increased from 2005 to 2006 by a factor of two and reached now a level of samples per year. These protein analytical services covered internal and external customers, the latter with increasing proportion. Due to external service and special pricing the CBA was able to cover the costs for one technician from incoming money. Protein analysis was mainly restricted to peptide mass fingerprinting for the identification of proteins in gel-based techniques. Protein fine characteri zation was confined to the site-specific analysis of a variety of post-translational modifications (PTMs), including phosphorylation, glycosylation, ubiquiti nation, SUMOylation and farnesylation. New Service Offers The list of service offers was extended recently by inclusion of a variety of new services. These comprize standardized protocols for the analysis of post-translationally modified proteins, in particular for the detection/identification and quantification of low molecular weight modifications, like fatty acids, isoprenoids, and sugars by GC/MS. N- and O- linked can now be profiled by ESI-MS/MS after enzymatic or chemical cleavage from the protein as their methylated derivatives. Protein Analysis Statistic Total DNA Total Protein Cologne External CMMC Others PMF ESI n.d. 227 Gels Edman Chromatography Phosphopeptides are enriched either offline or online by affinity chroma tography on titanium dioxide and analysed by ESI-MS (neutral loss experiments for detection of phosphopeptides) and ESI-MS/MS (for site identification). Site-specific analysis of phospho peptides will be considerably improved on availability of the HCTultra PTM discovery ESI-IT mass spectrometer, which will be installed in The instrument will provide a technological platform for customers interested in the site-specific analysis of other labile bound protein modifications, like O-glycosylation (O-GalNAc, O-GlcNAc, O-Man, O-Fuc).The development of further new service offers in the context of differential proteomics (SILAC, 2D- DIGE) is in progress. 88
94 Core Units Selected publications Schwientek, T., Mandel, U., Roth, U., Müller, S., Hanisch, F.-G. (2007) Proteomics, in press Popov, A., Abdullah, Z., Wickenhauser, C., Saric, T., Driesen, J., Hanisch, F.-G., Domann, E., Raven, E.L., Dehus, O., Hermann, C., Eggle, D., Debey, S., Chakraborty, T., Krönke, M., Utermöhlen, O., Schultze, J.L. (2006), J. Clin. Invest. 116, Spitzer, C., Schellmann, S., Sabovliev, A., Shahriari, M., Keshavaiah, C., Bechthold, N., Herzog, M., Müller, S., Hanisch, F.-G., Hülskamp, M. (2006) Development 133, Investments in Mass Spectrometry The CBA is always interested to keep up with the technological standard, which will support the requested analytical service for members of the CMMC. Particular emphasis will be layed on improvement of PTM analysis. In this context it is of utmost importance to complement existing mass spectrometric equipment in the facility by advanced, state-of-the-art instrumentation using ion trap technology and novel peptide dissociation modes, which combines robustness, with increased speed and analytical depth and introduces unique features in site specific PTM analysis. The HCT-ultra (Bruker) fulfils these demands and has been successfully applied for in the context of a Collaborative Research Proposal (SFB 635) to specifically support the research on modified proteins (positively reviewed in February 2007). At the end of this funding period the MALDI mass spectrometer (Reflex IV) will be 10 years old and need substitution by a MALDI-TOF-TOF instrument to keep the mass spectrometric equipment at a competitive state. For this reason we applied in May 2006 for an Ultraflex II mass spectrometer from Bruker (HBFG application). Collaborative Research and Scientific Networks The CBA is involved as a technological platform in one Collaborative Research Proposal (SFB 635) and in a DFG-Schwerpunkt application (Plant Micro). The CBA collaborates in the context of proteome research with groups at the MPI for Neuro physiology, the Medical Clinic I, the University Clinic of Erlangen, and the University of Bristol. Roth, U., Müller, S., Hanisch, F.-G. (2006) Methods Mol. Biol. 346, Von Bergwelt-Baildon, M.S., Popov, A., Saric, T., Chemnitz, J., Classen, S., Stoffel, M.S., Fiore, F., Roth, U., Beyer, M., Debey, S., Wickenhauser, C., Hanisch, F.-G., Schultze, J.L. (2006) Blood 108, Stalz, H., Roth, U., Schleuder, D., Macht, M., Haebel, S., Strupat, K., Peter-Katalinic, J., Hanisch, F.-G. (2006) Glycobiology 16, Lehr, S., Kotzka, J., Avci, H., Knebel, B., Müller, S., Hanisch, F.- G., Jacob, S., Haak, C., Susanto, F., Müller-Wieland, D. (2005) Biochemistry 44, Kaufmann, B., Müller, S., Hanisch, F.-G., Hartmann, U., Paulsson, M., Maurer, P., Zaucke, F. (2005) Glycobiology 14, Kambacheld M., Augustin, S., Tatsuta, T., Müller, S., Langer, T. (2005) J. Biol. Chem. 280, Augustin, S., Nolden, M., Müller, S., Hardt, O., Arnold, I., Langer, T. (2005). J. Biol. Chem. 280, Kobayashi, N., Kostka, G., Garbe, J.H.O., Keene, D.R., Bächinger, H.P., Hanisch, F.-G., Markova, D., Tsuda, T., Timpl, R., Chu, M.-L., Sasaki, T. (2007) J. Biol. Chem., in press 89
95 Core Units AFN Animal Facility Network Bettina Wagner Generation and analysis of animal models provide insights into gene regulation, development, pathogenesis and treatment of human diseases and thus form an essential component of research within the CMMC. Being core unit of the CMMC, the animal facility network provides access to carefully controlled animal housing to its members. In addition to routine animal keeping and breeding, the facility offers assistance in the generation of new genetically modified mouse lines through its microinjection laboratory, and provides help in the analysis of certain phenotypes. Introduction The animal facility network was composed by joining existing animal facilities into a coordinated network to provide the infrastructure for high quality animal research. The animal facility network focuses on the breeding and maintenance of laboratory mouse strains, particularly genetically modified. Rats, including transgenic strains, are also kept in this unit at a low extend. The inherent value and increasing numbers of genetically modified mouse strains, in part produced in our facility, emphasizes the need of cryopreservation techniques. The preservation of embryos is also performed at our facility enabling the storage of mouse strains in liquid nitrogen for future use without the ongoing costs of maintaining a mouse colony. Since different experimental settings have individual requirements, the facility offers different levels of containment, ranging from (semi)barrier conditions to conventional housing. In this way we are able to meet specified needs for breeding and experimental housing, including infectious studies and quarantine. Quality and well being of animals is ensured by highly motivated and well trained personnel as well as routine sentinel testing of the health status of the mice. New incoming mouse strains of questionable health status are transferred via embryo transfer into the animal units to insure and protect the health status of the animals present. Microinjection laboratory The use and development of transgenic (over expression of a single gene) and knock out (deletion of a single gene) technology offer a great potential for basic biomedical research. Fig. 1: Microinjection of DNA into the male pronucleus (W. Krzyzak, CMMC) In vivo imaging The Xenogen IVIS 200 Imaging System can be used to image both bioluminescence and fluorescence noninvasively in living animals, and to perform quantitative in vitro and in vivo assays using reporter cells tagged with a wide range of bioluminescent or fluorescent probes. The CMMC microinjection lab creates transgenic mice by pronuclear injection of B6D2F1 embryos with transgenic expression cassettes prepared by the investigator. Other mouse strains may be used upon request. Support in tail biopsies for genotyping and further breeding of founders to establish the transgenic line is provided. Generation of chimeric mice by introducing embryonic stem cells into preimplantation embryos (mostly blastocysts) is one approach to produce genetically modified mice. The facility is injecting mutant ES cell clones derived by the collaborating scientist into C57BL/6 host blastocysts. Fig. 2: Chemiluminescence due to luciferase reporter gene expression in a living anaesthetised mouse using the Xenogen IVIS 200 camera system (U. Protzer, NG-F) 90
96 Core Units This system is capable of providing images in real-time to monitor and record cellular and genetic activity within a living organism. Light emitted from cells inside a living tissue or mouse can be detected, offering unique opportunities for diverse applications in molecular research such as monitoring the roles of certain genes in disease and the measurement of tumour growth in a mouse. Perspectives The rapid development of transgenic technologies led to a massive increase in mouse lines and thus multiplies the space required for mouse breeding and maintenance. To meet this demand, a modern animal house for the housing of a calculated additional mice under SPF conditions is under construction. Fig. 3: Mouse Chimera (T. Schneider, J. Hescheler, C 4) 91
97 Publications Summary of Original Articles Published by CMMC Members Selected CMMC-project related original publications are listed that were generated during the time interval January 2005 until May 2007 and that are already published or in press. Authors indicated in bold are principal investigators of the CMMC Akgül B, García-Escudero R, Ghali L, Pfister HJ, Fuchs PG, Navsaria H, Storey A: The E7 protein of cutaneous human papillomavirus type 8 causes invasion of human keratinocytes into the dermis in organotypic cultures of skin. Cancer Res (2005) 65, Akgül B, Lemme W, García-Escudero R, Storey A, Pfister HJ: UV-B irradiation stimulates the promoter activity of the high-risk, cutaneous human papillomavirus 5 and 8 in primary keratinocytes. Arch Virol (2005) 150, Augustin S, Nolden M, Müller S, Hardt O, Arnold I, Langer T: Characterization of peptides released from mitochondria: evidence for constant proteolysis and peptide efflux. J Biol Chem (2005) 280, Bader BL, Smyth N, Nedbal S, Miosge N, Baranowsky A, Mokkapati S, Murshed M, Nischt R: Compound genetic ablation of nidogen 1 and 2 causes basement membrane defects and perinatal lethality in mice. Mol Cell Biol (2005) 25, Baudler S, Baumgartl J, Hampel B, Buch T, Waisman A, Snapper CM, Krone W, Bruning JC: Insulin-like growth factor-1 controls type 2 T cell-independent B cell response. J Immunol (2005) 174, Berthold F, Boos J, Burdach S, Erttmann R, Henze G, Hermann J, Klingebiel T, Kremens B, Schilling FH, Schrappe M, Simon T, Hero B: Myeloablative megatherapy with autologous stem-cell rescue versus oral maintenance chemotherapy as consolidation treatment in patients with high-risk neuroblastoma: a randomised controlled trial. Lanc Oncol (2005) 6, Boensch M, Oberthuer A, Fischer M, Skowron M, Oestreich J, Berthold F, Spitz R: Quantitative real-time PCR for quick simultaneous determination of therapy-stratifying markers MYCN amplification, deletion 1p and 11q. Diagn Mol Pathol (2005) 14, Burwinkel B, Wirtenberger M, Klaes R, Schmutzler RK, Grzybowska E, Forsti A, Frank B, Bermejo JL, Bugert P, Wappenschmidt B, Butkiewicz D, Pamula J, Pekala W, Zientek H, Mielzynska D, Siwinska E, Bartram CR, Hemminki K: Association of NCOA3 polymorphisms with breast cancer risk. Clin Cancer Res (2005) 11, Carlos C, Dong HF, Howard OMZ, Oppenheim JJ, Hanisch F-G, Finn OJ: Human tumor antigen MUC1 is chemotactic for immataure dendritic cells and elicits maturation but does not promoteth1 type immunity. J Immunol (2005) 175, Chefetz I, Heller R, Galli-Tsinopoulou A, Richard G, Wollnik B, Indelman M, Koerber F, Topaz O, Bergman R, Sprecher E, Schoenau E: A novel homozygous missense mutation in FGF23 causes Familial Tumoral Calcinosis associated with disseminated visceral calcification. Hum Genet (2005) 118, Clemen CS, Fischer D, Roth U, Simon S, Vicart P, Kato K, Kaminska AM, Vorgerd M, Goldfarb LG, Eymard B, Romero NB, Gourdeau B, Eggermann T, Zerres K, Noegel AA, Schröder R: Hsp27-2D-gel electrophoresis is a diagnostic tool to differentiate primary desminopathies from myofibrillar myopathies. FEBS Lett (2005) 579, Dorn A, Zhao H, Granberg F, Hösel M, Webb D, Svensson C, Pettersson U, Doerfler W: Identification of specific cellular genes up-regulated late in adenovirus type 12 infection. J Virol (2005) 79, Dumortier J, Schönig K, Giese T, Schirmacher P, Bujard H, Protzer U: Liver-specific expression of interferon gamma following adenoviral gene transfer controls hepatitis B virus replication in mice. Gene Therapy (2005) 12, Engelmann K, Kinlough CL, Müller S, Razaw, H, Baldus SE, Hughey RP, Hanisch F-G: Transmembrane and secreted MUC1 probes show trafficking-dependent changes in O- glycan core profiles. Glycobiology (2005) 15, Eyupoglu IY, Hahnen E, Buslei R, Siebzehnrubl FA, Savaskan NE, Luders M, Trankle C, Wick W, Weller M, Fahlbusch R, Blumcke I: Suberoylanilide hydroxamic acid (SAHA) has potent anti-glioma properties in vitro, ex vivo and in vivo. J Neurochem (2005) 93, Fank B, Hemminki K, Bermejo JL, Klaes R, Bugert P, Wappenschmidt B, Schmutzler RK, Burwinkel B: TP53- binding protein variants and breast cancer risk: a casecontrol study. Breast Cancer Res (2005) 7, Fischer M, Skowron M, Berthold F: Reliable transcript quantification by real-time reverse transcriptasepolymerase chain reaction in primary neuroblastoma using normalization to averaged expression levels of the control genes HPRT1 and SDHA. J Mol Diag (2005) 7, Fisher SJ, Bruning JC, Lannon S, Kahn CR: Insulin signaling in the central nervous system is critical for the normal sympathoadrenal response to hypoglycemia. Diabetes (2005) 54,
98 Publications Frank B, Bermejo JL, Hemminki K, Klaes R, Bugert P, Wappenschmidt B, Schmutzler RK, Burwinkel B: Re: Association of a common variant of the CASP8 gene with reduced risk of breast cancer. J Natl Cancer Inst (2005) 97, Frank B, Hemminki K, Shanmugam KS, Meindl A, Klaes R, Schmutzler RK, Wappenschmidt B, Untch M, Bugert P, Bartram CR, Burwinkel B: Association of death receptor 4 haplotype 626C-683C with an increased breast cancer risk. Carciogenesis (2005) 11, Frank B, Hemminki K, Wirtenberger M, Bermejo JL, Bugert P, Klaes R, Schmutzler RK, Wappenschmidt B, Bartram CR, Burwinkel B: The rare ERBB2 variant Ile654Val is associated with an increased familial breast cancer risk. Carciogenesis (2005) 26, Freude S, Plum L, Schnitker J, Leeser U, Udelhoven M, Krone W, Bruning JC, Schubert M: Peripheral Hyperinsulinemia Promotes Tau Phosphorylation in vivo. Diabetes (2005) 54, Gallagher PG, Bao Y, Prorock A, Zigrino P, Nischt R, Politi V, Mauch C, Deagulev B, Fox JW: Gene expression profiling reveals cross-talk between melanoma and fibroblasts: Implications for host-tumor interactions in metastasis. Cancer Res (2005) 65, Gallagher PG, Bao Y, Serrano SMT, Laing GD, Theakston RDG, Gutiérrez JM, Escalante T, Zigrino P, Moura-da-Silva A, Nischt R, Mauch C, Moskaluk C, Fox JW: Role of the snake venom toxin Jararhagin in proinflammatory pathogenesis: in vitro and in vivo gene expression analysis of the effects of the toxin. Arch Biochem Biophys (2005) 441, Gebhardt C, Breitenbach U, Richter KH, Furstenberger G, Mauch C, Angel P, Hess J: C-Fos-dependent induction of the small ras-related GTPase Rab11a in skin carcinogenesis. Am J Pathol (2005) 167, Gotte M, Bernfield M, Joussen AM: Increased leukocyteendothelial interactions in syndecan-1-deficient mice involve heparan sulfate-dependent and -independent steps. Curr Eye Res (2005) 30, Gouni-Berthold I, Giannakidou E, Muller-Wieland D, Faust M, Kotzka J, Berthold HK, Krone W: The Pro387Leu variant of protein tyrosine phosphatase-1b is not associated with diabetes mellitus type 2 in a German population. J Intern Med (2005) 257, Gouni-Berthold I, Giannakidou E, Muller-Wieland D, Faust M, Kotzka J, Berthold HK, Krone W: Peroxisome proliferator-activated receptor-gamma2 Pro12Ala and endothelial nitric oxide synthase-4a/b gene polymorphisms are not associated with hypertension in diabetes mellitus type 2. J Hypertens (2005) 23, Gropp E*, Shanabrough M, Borok E, Xu AW, Janoschek R, Buch T, Plum L, Balthasar N, Hampel B, Waisman A, Barsh GS, Horvath TL, Bruning JC: Agouti-related peptide-expressing neurons are mandatory for feeding. Nat Neurosci (2005) 8, (*MD-PhD Program) Gründemann D, Harlfinger S, Golz S, Geerts A, Lazar A, Berkels R, Jung N, Rubbert A, Schömig E: Discovery of the ergothioneine transporter. Proc Natl Acad Sci USA (2005) 102, Hacker UT, Wingenfeld L, Kofler DM, Schuhmann NK, Lutz S, Herold T, King SBS, Gerner FM, Perabo L, Rabinowitz J, McCarty DM, Samulski RJ, Hallek M, Büning H: Adeno-associated virus serotypes 1 to 5 mediated tumor cell directed gene transfer and improvement of transduction efficiency. J Gene Med (2005) 7, Harlfinger S, Fork C, Lazar A, Schömig E, Gründemann D: Are organic cation transporters capable of transporting prostaglandins? Naunyn Schmiedebergs Arch Pharmacol (2005) 372, Hengstermann A, D silva MA, Kuballa P, Butz K, Hoppe-Seyler F, Scheffner M: Growth suppression induced by downregulation of E6-AP expression in HPV-positive cancer cell lines depends on p53. J Virol (2005) 79, Hennige AM, Ozcan U, Okada T, Jhala US, Schubert M, White MF, Kulkarni RN: Alterations in growth and apoptosis of insulin receptor substrate-1-deficient beta-cells. Am J Physiol Endocrinol Metab (2005) 289, E Hombach A, Heuser C, Stallmach A, Abken H: Re-combinant interleukin-12 for immunotherapy: a double-sided sword. Curr Med Chem - Immun, Endoc & Metab Agents (2005) 5, Isogawa M, Kakimi K, Kamamoto H, Protzer U, Chisari FV: Differential dynamics of the peripheral and intrahepatic cytotoxic T lymphocyte response to hepatitis B surface antigen. Virology (2005) 333, Jacobs AH, Thomas A, Kracht LW, Li H, Dittmar C, Garlip G, Galliks N, Klein JC, Herholz K, Wienhard K, Heiss W-D: 18 F-Fluoro-L-thymidine and 11 C-methyl-methionine as markers of increased transport and proliferation in brain tumors. J Nucl Med (2005) 46, Jing X, Li D-Q, Olofsson CS, Salehi A, Surve VV, Caballero J, Ivarsson R, Lundquist I, Pereverzev A, Schneider T, Rorsman P, Renström E: Ca v 2.3 calcium channels control second phase insulin release. J Clin Invest (2005) 115, Jockusch WJ, Praefcke GJ, McMahon HT, Lagnado L: Clathrin-dependent and clathrin-independent retrieval of synaptic vesicles in retinal bipolar cells. Neuron (2005) 46,
99 Publications Kalay E, de Brouwer AP, Caylan R, Nabuurs SB, Wollnik B, Karaguzel A, Heister JG, Erdol H, Cremers FP, Cremers CW, Brunner HG, Kremer H: A novel D458V mutation in the SANS PDZ binding motif causes atypical Usher syndrome. J Mol Med (2005) 83, Kambacheld M, Augustin S, Tatsuta T, Müller S, Langer T: Role of the novel metallopeptidase Mop112 and saccharolysin for the complete degradation of proteins residing in different subcompartments of mitochondria. J Biol Chem (2005) 280, Kashkar H, Wiegmann K, Yazdanpanah B, Haubert D, Kronke M: Acid sphingomyelinase is indispensable for UV light-induced Bax conformational change at the mitochondrial membrane. J Biol Chem (2005) 280, Ko Y-P, Kobbe B, Paulsson M, Wagener R: Zebrafish (Danio rerio) matrilins: Shared and divergent characteristics with their mammalian counterparts. Biochem J (2005) 386, Kociok N, Gavranic C, Kirchhof B, Joussen AM: Influence on membrane-mediated cell activation by vesicles of silicone oil or perfluorohexyloctane. Graefes Arch Clin Exp Ophthalmol (2005) 243, Kronfeld K, Abken H, Seliger B: B7-1 and B7-2 act differentially in the induction of a T-cell response: their impact for a HLA-matched and HLA-mismatched antitumor immunotherapy. Int J Cancer (2005) 117, Kronfeld K, Hochleitner E, Mendler S, Goldschmitdt J, Lichtenfels R, Lottspeich F, Abken H, Seliger B: B7/CD28 costimulation of T cells induces a distinct proteome pattern. Mol Cell Proteomics (2005) 4, Krüger M, Zittrich S, Redwood C, Blaudeck N, James J, Robbins J, Pfitzer G, Stehle R: Effects of the mutation R145G in human cardiac troponin I on the kinetics of the contraction-relaxation cycle in isolated cardiac myofibrils. J Physiol (2005) 564, Kunzelmann S, Praefcke GJ, Herrmann C: Nucleotide binding and self-stimulated GTPase activity of human guanylate-binding protein 1 (hgbp1). Methods Enzymol (2005) 404, Lehr S, Kotzka J, Avci H, Knebel B, Muller S, Hanisch F- G, Jacob S, Haak C, Susanto F, Muller-Wieland D: Effect of sterol regulatory element binding protein-1a on the mitochondrial protein pattern in human liver cells detected by 2D-DIGE. Biochemistry (2005) 44, Libotte T, Zaim H, Abraham S, Padmakumar VC, Schneider M, Lu W, Munck M, Hutchison C, Wehnert M, Fahrenkrog B, Sauder U, Aebi U, Noegel AA, Karakesisoglou I: Lamin A/C dependent localization of Nesprin-2, a giant scaffolder at the nuclear envelope. Mol Biol Cell (2005) 16, Löffek S, Zigrino P, Angel P, Anwald B, Krieg T, Mauch C: High invasive melanoma cells induce MMP-1 synthesis and activation in fibroblasts by interleukin-1alpha and basic fibroblast growth factor-mediated mechanisms. J Invest Dermatol (2005) 124, Lüke M, Henry M, Lingohr T, Maghsoodian M, Hescheler J, Weiergräber M*, Sickel W, Schneider T: A Ni 2+ -sensitive component of the ERG-b-wave from the isolated bovine retina is related to E-type voltage-gated Ca 2+ channels. Graefe Arch Clin Experim Ophthalmol (2005) 243, (*MD-PhD Program) Lüke M, Lüke C, Hescheler J, Schneider T, Sickel W: Effects of Phosphodiesterase Type 5 Inhibitor Sildenafil on Retinal Function in Isolated Superfused Retina. J Ocul Pharmacol & Ther (2005) 21, Lüke M, Weiergräber M*, Brand C, Siapich SA, Banat M, Hescheler J, Lüke C, Schneider T: The isolated perfused bovine retina - a sensitive tool for pharma-cological research on retinal function. Brain Res & Brain Res Protoc (2005) 16, (*MD-PhD Program) von Mensdorff-Pouilly S, Kinarsky L, Engelmann K, Baldus SE, Verheijen RH, Hollingsworth MA, Pisarev V, Sherman S, Hanisch F-G: Sequence-variant repeats of MUC1 show higher conformational flexibility, are less densely O-glycosylated and induce differential B lymphcyte responses. Glycobiology (2005) 15, Meyer PT*, Elmenhorst D, Zilles K, Bauer A: Simplified Quantification of Cerebral A1 Adenosine Receptors using [18F]CPFPX and PET: Analyses Based on Venous Blood Sampling. Synapse (2005) 55, (*MD-PhD Program) Meyer PT*, Elmenhorst D, Bier D, Holschbach MH, Matusch A, Coenen HH, Zilles K, Bauer A: Quantification of cerebral A1 adenosine receptors in humans using [18F]CPFPX and PET: an equilibrium approach. NeuroImage (2005) 24, (*MD-PhD Program) Michels G, Er F, Khan I, Südkamp M, Herzig S, Hoppe UC: Single-channel properties support a potential contribution of HCN channels and I f to cardiac arrhythmias. Circulation (2005) 111, Miletic H, Utermöhlen O, Wedekind C, Hermann M, Stenzel W, Lassmann H, Schlüter D, Deckert M: P is a neuritogenic epitope of the peripheral myelin protein P0 and induces autoimmune neuritis in C57BL/6 mice. J Neuropathol Neurol (2005) 64, Möllers M, Maniura-Weber K, Kiseljakovic E, Bust M, Hayrapetyan A, Jaksch M, Helm M, Wiesner RJ, von Kleist- 94
100 Publications Retzow JC: Impairment of post-transcriptional maturation and severe depletion in mitochondrial trna Ser(UCN) caused by T7512C and G7497A point mutations. Nucl Acids Res (2005) 33, Nolden M, Ehses S, Koppen M, Bernacchia A, Rugarli EI, Langer T: The m-aaa protease defective in hereditary spastic paraplegia controls ribosome assembly in mitochondria. Cell (2005) 123, Oberthuer A, Skowron M, Spitz R, Kahlert Y, Westermann F, Mehler K, Berthold F, Fischer M. Characterization of a complex genomic alteration on chromosome 2p that leads to four alternatively spliced fusion transcripts in the neuroblastoma cell lines IMR-5, IMR-5/75 and IMR-32. Gene (2005) 363, Padmakumar VC, Libotte T, Lu W, Zaim H, Abraham S, Noegel AA, Gotzmann J, Foisner R, Karakesisoglou I: Sun1, an inner nuclear membrane protein, mediates the anchorage of Nesprin-2 to the nuclear envelope. J cell Sci (2005) 118, Pereverzev A, Salehi A, Mikhna M, Renstrom E, Hescheler J, Weiergräber M*, Smyth N, Schneider T: The ablation of the Ca(v)2.3/E-type voltage-gated Ca 2+ channel causes a mild phenotype despite an altered glucose induced glucagon response in isolated islets of Langerhans. Eur J Pharmacol (2005) 511, (*MD-PhD Program) Plum L, Schubert M, Bruning JC: The role of insulin receptor signaling in the brain. Trends Endocrinol Metab (2005) 16, Remus R, Kanzaki A, Nakanishi H, Wada H, Yawata A, Zeschnigk M, Zuther I, Muiznieks I, Schmitz B, Doerfler W, Yawata Y: DNA methylation in promoter regions of red cell membrane protein genes in healthy individuals and patients with hereditary membrane disorders. Int J Hematol (2005) 81, Rick M, Ramos Garrido SI, Herr C, Thal DR, Noegel AA, Clemen CS: Nuclear localization of Annexin A7 during murine brain development. BMC Neurosci (2005) 6, 25. Rueger MA*, Winkeler A, Miletic H, Kaestle C, Richter R, Schneider G, Hampl JA, Fraefel C, Jacobs AH: The interindividual variability of the infectivity of HSV-1 amplicon vectors in primary cell cultures of human gliomas. Gene Therapy (2005) 12, (*MD-PhD Program) Rueger MA*, Winkeler A, Miletic HP*, Kaestle C, Richter R, Schneider G, Hilker R, Heneka MT, Ernestus RI, Hampl JA, Fraefel C, Jacobs AH: Variability in infectivity of primary cell cultures of human brain tumors with HSV-1 amplicon vectors. Gene Ther (2005) 12: (*MD- PhD Program) Saydam O, Glauser DL, Heid I, Turkeri G, Jacobs AH, Ackermann M, Fraefel C: Herpes simplex virus 1 amplicon vector-mediated sirna targeting epidermal growth factor receptor inhibits growth of human glioma cells in culture and in vivo. Molecular Therapy (2005) 12, Schaper ID, Marcuzzi GP, Weissenborn SJ, Kasper HU, Dries V, Smyth N, Fuchs P, Pfister H: Development of skin tumors in mice transgenic for early genes of human papillomavirus type 8. Cancer Res (2005) 65, Schnepp A, Komp Lindgren P, Hülsmann H, Kröger S, Paulsson M, Hartmann, U: Mouse testican-2: Expression, glycosylation and effects on neurite outgrowth. J Biol Chem (2005) 280, Seibler J, Kuter-Luks B, Kern H, Streu S, Plum L, Mauer J, Kuhn R, Bruning JC, Schwenk F: Single copy shrna configuration for ubiquitous gene knockdown in mice. Nucleic Acids Res (2005) 33, e67. Shin E-C, Protzer U, Untergasser A, Hasselschwert D, Rice CM, Feinston SM, Rehermann B: Liver-Directed Interferon-γ Gene Delivery in Chronic Hepatitis C infected chimpanzees. Journal of Virology (2005) 79, Siebolts U*, Ates M, Spitz R, Thiele J, Wickenhauser C: Quantification of clonal hematopoiesis in polycythemia vera. Virchows Arch (2005) 447, (*MD-PhD Program) Stoffel W, Jenke B, Block B, Zumbansen M, Koebke J: Neutral sphingomyelinase 2 (smpd3) in the control of postnatal growth and development. Proc Natl Acad Sci USA (2005) 120, Sun Y, Grimmler M, Schwarzer V, Schoenen F, Fischer U, Wirth B: Molecular and functional analysis of intragenic SMN1 mutations in patients with spinal muscular atrophy. Hum Mutat (2005) 25, Tatsuta T, Model K, Langer T: Formation of membranebound ring complexes by prohibitins in mitochondria. Mol Biol Cell (2005) 16, Tscharntke M, Pofahl R, Krieg T, Haase I: Ras induced spreading and wound closure in human epidermal keratinocytes. FASEB J (2005) 19, Tukel T, Uzumcu A, Gezer A, Kayserili H, Yuksel-Apak M, Uyguner O, Gultekin SH, Hennies HC, Nurnberg P, Desnick RJ, Wollnik B. A new syndrome, congenital extraocular muscle fibrosis with ulnar hand anomalies, maps to chromosome 21qter. J Med Genet (2005) 42, Tunggal JA, Helfrich I, Schmitz A, Schwarz H, Günzel D, Fromm M, Kemler R, Krieg T, Niessen CM: E-cadherin is essential for in vivo skin barrier function by regulating tight junctions. EMBO J 24,
101 Publications Uhl M, Weiler M, Wick W, Jacobs AH, Weller M, Herrlinger U: Migratory neural stem cells for improved thymidine kinase-based gene therapy of malignant gliomas. Biochem Biophys Res Commun (2005) 328, Vantler M, Caglayan E, Zimmermann WH, Baumer AT, Rosenkranz S: Systematic evaluation of anti-apoptotic growth factor signaling in vascular smooth muscle cells. Only phosphatidylinositol 3 -kinase is important. J Biol Chem (2005) 280, Wappenschmidt B, Fimmers R, Rhiem K, Brosig M, Wardelmann E, Meindl A, Arnold N, Mallmann P, Schmutzler RK: Strong evidence that the common variant S384F in BRCA2 has no pathogenic relevance in hereditary breast cancer. Breast Cancer Res (2005) 7, Weiergräber M*, Henry M, Hescheler J, Smyth N, Schneider T: Electrocorticographic and deep intracerebral EEG recording in mice using a telemetry system. Brain Res Protoc (2005) 14, (*MD-PhD Program) Weiergräber M*, Henry M, Südkamp M, de Vivie E- R, Hescheler J, Schneider T: Ablation of Ca v 2.3 / E- type voltage-gated calcium channel results in cardiac arrhythmia and altered autonomic control within the murine cardiovascular system. Basic Res Cardiol (2005) 100, (*MD-PhD Program) Wirtenberger M, Hemminki K, Forsti A, Klaes R, Schmutzler RK, Grzybowska E, Bermejo JL, Wappenschmidt B, Bugert P, Butkiewicz D, Pamula J, Pekala W, Zientek H, Bartram CR, Burwinkel B: c-myc Asn1 1Ser is associated with increased risk for familial breast cancer. Int J Cancer (2005) 11, Wölfl M, Jungbluth AA, Garrido F, Cabrera T, Meyen- Southard S, Spitz R, Ernestus K, Berthold F: Expression of MHC class I, MHC class II, and cancer germline antigens in neuroblastoma. Cancer Immunol Immunother (2005) 54, Yi X, Schubert M, Peachey NS, Suzuma K, Burks DJ, Kushner JA, Suzuma I, Cahill C, Flint CL, Dow MA, Leshan RL, King GL, White MF: Insulin receptor substrate 2 is essential for maturation and survival of photoreceptor cells. J Neurosci (2005) 2, Zigrino P, Mauch C, Fox JW, Nischt R: ADAM-9 expression and regulation in human skin melanoma and melanoma cell lines. Int J Cancer (2005) 116, Akgül B, Cürten M, Haigis H, Rogosz I, Pfister H: Interferon regulatory factor 5.2 acts as a transcription repressor of Epidermodysplasia verruciformis-associated human papillomaviruses. Arch Virol (2006) 151, Akgül B, Pfefferle R, Marcuzzi GP, Zigrino P, Krieg T, Pfister H, Mauch C: Expression of matrix metalloproteinase (MMP)-2, MMP-9, MMP-13, and MT1-MMP in skin tumors of human papillomavirus type 8 transgenic mice. Exp Dermatol (2006) 15, Arnold I, Wagner-Ecker M, Ansorge W, Langer T: Evidence for a novel mitochondria-to-nucleus signalling pathway in respiring cells lacking i-aaa protease and the ABCtransporter Mdl1. Gene (2006) 367, Aumailley M, Has C, Tunggal L, Bruckner-Tuderman L: Molecular basis of inherited skin-blistering disorders, and therapeutic implications. Expert Rev Mol Med (2006) 8, Bakthavatsalam D, Meijer HJ, Noegel AA, Govers F: Novel phosphatidylinositol phosphate kinases with a G-protein coupled receptor signature are shared by Dictyostelium and Phytophthora. Trends Microbiol (2006) 14, Baldus SE, Monig SP, Zirbes TK, Thakran J, Kothe D, Koppel M, Hanisch FG, Thiele J, Schneider PM, Holscher AH, Dienes HP: Lewis(y) antigen (CD174) and apoptosis in gastric and colorectal carcinomas: correlations with clinical and prognostic parameters. Histol Histopathol (2006) 21, Baumgartl J, Baudler S, Scherner M, Babaev V, Makowski L, Suttles J, McDuffie M, Tobe K, Kadowaki T, Fazio S, Kahn CR, Hotamisligil GS, Krone W, Linton M, Bruning JC: Myeloid lineage cell-restricted insulin resistance protects apolipoproteine-deficient mice against atherosclerosis. Cell Metab (2006) 3, Becker J, Erdlenbruch B, Noskova I, Schramm A, Aumailley M, Schorderet DF, Schweigerer L: Keratoepithelin suppresses the progression of experimental human neuroblastomas. Cancer Res (2006) 66, Bergmann C, Senderek J, Anhuf D, Thiel CT, Ekici AB, Poblete-Gutierrez P, van Steensel M, Seelow D, Nürnberg G, Schild HH, Nürnberg P, Reis A, Frank J, Zerres K: Mutations in the gene encoding the Wnt-signaling component R- spondin 4 (RSPO4) cause autosomal recessive anonychia. Am J Hum Genet (2006) 79, von Bergwelt-Baildon MS, Popov A, Saric T, Chemnitz J, Classen S, Stoffel MS, Fiore F, Roth U, Beyer M, Debey S, Wickenhauser C, Hanisch FG, Schultze JL: CD25 and indoleamine 2,3-dioxygenase are up-regulated by prostaglandin E2 and expressed by tumor-associated dendritic cells in vivo: additional mechanisms of T-cell inhibition. Blood (2006) 108, Billion K, Ibrahim H, Mauch C, Niessen CM: Increased soluble E-cadherin in melanoma patients. Skin Pharmacol Physiol (2006) 19,
102 Publications Bose K, Nischt R, Page A, Bader BL, Paulsson M, Smyth N: Loss of nidogen-1 and -2 results in syndactyly and changes in limb development. J Biol Chem (2006) 281, Braunschweig F, Fahrleitner A, Mangiavacchi M, Ghio S, Fotuhi P, Hoppe UC, Linde C: Correlation between serial measurements of N-terminal pro brain natriuretic peptide and ambulatory cardiac filling pressures in outpatients with chronic heart failure. Eur J Heart Fail (2006) 8, Breuhahn K, Nussbaum T*, Singer S, Schirmacher P: The Insulin-like Grwoth Factor (IGF) Signaling Pathway: Strategies for successful therapeutics tasks in cancer treatment. Current Cancer Therapy Reviews (2006) 2, (*MD-PhD Program) Brichta L, Holker I, Haug K, Klockgether T, Wirth B: In vivo activation of SMN in spinal muscular atrophy carriers and patients treated with valproate. Ann Neurol (2006) 59, Burwinkel B, Shanmugam KS, Hemminki K, Meindl A, Schmutzler RK, Sutter C, Wappenschmidt B, Kiechle M, Bartram CR, Frank B: Transcription factor 7-like 2 (TCF7L2) variant is associated with familial breast cancer risk: a case-control study. BMC Cancer (2006) 6, 268. Buslei R, Kreutzer J, Hofmann B, Schmidt V, Siebzehnrubl F, Hahnen E, Eyupoglu IY, Fahlbusch R, Blumcke I: Abundant hypermethylation of SOCS-1 in clinically silent pituitary adenomas. Acta Neuropathol (Berl) (2006) 111, Caglayan E, Huntgeburth M, Karasch T, Weihrauch J, Hunzelmann N, Krieg T, Erdmann E, Rosenkranz S: Phosphodiesterase type 5 inhibition is a novel therapeutic option in Raynaud disease. Arch Intern Med (2006) 166, Canault M, Tellier E, Bonardo B, Mas E, Aumailley M, Juhan- Vague I, Nalbone G, Peiretti F: FHL2 interacts with both ADAM-17 and the cytoskeleton and regulates ADAM-17 localization and activity. J Cell Physiol (2006) 208, Chometon G, Zhang ZG, Rubinstein E, Boucheix C, Mauch C, Aumailley M: Dissociation of the complex between CD151 and laminin-binding integrins permits migration of epithelial cells. Exp Cell Res (2006) 312, Chrostek A, Wu X, Quondamatteo F, Hu R, Sanecka A, Niemann C, Langbein L, Haase I, Brakebusch C: Rac1 is crucial for hair follicle integrity but is not essential for maintenance of the epidermis. Mol Cell Biol (2006) 26, Chu H, Thievessen I, Sixt M, Lämmermann T, Waisman A, Braun, A, Noegel AA, Fässler R: γ-parvin is dispensable for hematopoiesis, leukocyte trafficking, and T-celldependent antibody response. Mol Cell Biol (2006) 26, Cichon S, Martin L, Hennies HC, Müller F, Van Driessche K, Karpushova A, Stevens W, Colombo R, Renne T, Drouet C, Bork K, Nöthen MM: Increased activity of coagulation factor XII (Hageman factor) causes hereditary angioedema type III. Am J Hum Genet (2006) 79, Cleven BE, Palka-Santini M, Gielen J, Meembor S, Kronke M, Krut O: Identification and characterization of bacterial pathogens causing bloodstream infections by DNA microarray. J Clin Microbiol (2006) 44, Cuajungco MP, Grimm C, Oshima K, D hoedt D, Nilius B, Mensenkamp AR, Bindels RJ, Plomann M, Heller S: PACSINs bind to the TRPV4 cation channel. PACSIN 3 modulates the subcellular localization of TRPV4. J Biol Chem (2006) 281, Dell S, Peters S, Muther P, Kociok N, Joussen AM: The role of PDGF receptor inhibitors and PI3-kinase signaling in the pathogenesis of corneal neovascularization. Invest Ophthalmol Vis Sci (2006) 47, Dempfle A, Wudy SA, Saar K, Hagemann S, Friedel S, Scherag A, Berthold LD, Alzen G, Gortner L, Blum WF, Hinney A, Nürnberg P, Schafer H, Hebebrand J: Evidence for involvement of the vitamin D receptor gene in idiopathic short stature via a genome-wide linkage study and subsequent association studies. Hum Mol Genet (2006) 15, Drumea-Mirancea M, Wessels JT, Muller CA, Essl M, Eble JA, Tolosa E, Koch M, Reinhardt DP, Sixt M, Sorokin L, Stierhof YD, Schwarz H, Klein G: Characterization of a conduit system containing laminin-5 in the human thymus: a potential transport system for small molecules. J Cell Sci (2006) 119, Durr M, Escobar-Henriques M, Merz S, Geimer S, Langer T, Westermann B: Nonredundant roles of mitochondriaassociated F-box proteins Mfb1 and Mdm30 in maintenance of mitochondrial morphology in yeast. Mol Biol Cell (2006) 17, Eckes B, Zweers MC, Zhang ZG, Hallinger R, Mauch C, Aumailley M, Krieg T: Mechanical tension and integrin alpha 2 beta 1 regulate fibroblast functions. J Investig Dermatol Symp Proc (2006) 11, Eichenauer DA, Simhadri VL, von Strandmann EP, Ludwig A, Matthews V, Reiners KS, von Tresckow B*, Saftig P, Rose-John S, Engert A, Hansen HP: ADAM10 inhibition of human CD30 shedding increases specificity of targeted immunotherapy in vitro. Cancer Res (2007) 67, (*MD-PhD Program) 97
103 Publications Ellinor PT, Sasse-Klaassen S, Probst S, Gerull B, Shin JT, Toeppel A, Heuser A, Michely B, Yoerger DM, Song BS, Pilz B, Krings G, Coplin B, Lange PE, Dec GW, Hennies HC, Thierfelder L, MacRae CA: A novel locus for dilated cardiomyopathy, diffuse myocardial fibrosis, and sudden death on chromosome 10q J Am Coll Cardiol (2006) 48, Entz P, Blaumeiser B, Betz RC, Lambert J, Seymons K, Eigelshoven S, Hanneken S, Kruse R, Nürnberg P, Nagy M, Nöthen MM: Investigation of the HLA-DRB1 locus in alopecia areata. Eur J Dermatol (2006) 16, Ernestus K, Pietsch T, Gessler M, Simon T, Hero B, Berthold F: Structure, use, and risks of biomaterial repositories of embryonal tumors. Clin Padiatr (2006) 218, Escobar-Henriques M, Langer T: Mitochondrial shaping cuts. Biochim Biophys Acta (2006), 1763, Escobar-Henriques M, Westermann B, Langer T: Regulation of mitochondrial fusion by the F-box protein Mdm30 involves proteasome-independent turnover of Fzo1. J Cell Biol (2006) 173, Eyupoglu IY, Hahnen E, Trankle C, Savaskan NE, Siebzehnrubl FA, Buslei R, Lemke D, Wick W, Fahlbusch R, Blumcke I: Experimental therapy of malignant gliomas using the inhibitor of histone deacetylase MS-275. Mol Cancer Ther (2006) 5, Fischer M, Oberthuer A, Brors B, Kahlert Y, Skowron M, Voth H, Warnat P, Ernestus K, Hero B, Berthold F: Differential expression of neuronal genes defines subtypes of disseminated neuroblastoma with favorable and unfavorable outcome. Clin Cancer Res (2006) 12, Frank B, Hemminki K, Meindl A, Wappenschmidt B, Klaes R, Schmutzler RK, Untch M, Bugert P, Bartram CR, Burwinkel B: Association of the ARLTS1 Cys148Arg variant with familial breast cancer risk. Int J Cancer (2006) 118, Frank B, Hemminki K, Wappenschmidt B, Klaes R, Meindl A, Schmutzler RK, Bugert P, Untch M, Bartram CR, Burwinkel B: A variable number of tandem repeats polymorphism in the SMYD3 promoter region and the risk of familial breast cancer. Int J Cancer (2006) 118, Frank B, Hemminki K, Wappenschmidt B, Meindl A, Klaes R, Schmutzler RK, Bugert P, Untch M, Bartram CR, Burwinkel B: Association of the CASP10 V410I variant with reduced familial breast cancer risk and interaction with the CASP8 D302H variant. Carcinogenesis (2006) 27, Franzke CW, Has C, Schulte C, Huilaja L, Tasanen K, Aumailley M, Bruckner-Tuderman L: C-terminal truncation impairs glycosylation of transmembrane collagen XVII and leads to intracellular accumulation. J Biol Chem (2006) 281, Fyfe JC, Menotti-Raymond M, David VA, Brichta L, Schaffer AA, Agarwala R, Murphy WJ, Wedemeyer WJ, Gregory BL, Buzzell BG, Drummond MC, Wirth B, O Brien SJ: An approximately 140-kb deletion associated with feline spinal muscular atrophy implies an essential LIX1 function for motor neuron survival. Genome Res (2006) 16, Galldiks N, Kracht LW, Burghaus L, Thomas A, Jacobs AH, Heiss WD, Herholz K: Use of 11C-methionine PET to monitor the effects of temozolomide chemotherapy in malignant gliomas. Eur J Nucl Med Mol Imaging (2006) 33, Gassanov N, Brandt MC, Michels G, Lindner M, Er F, Hoppe UC: Angiotensin II - induced changes of calcium sparks and ionic currents in human atrial myocytes: potential role for early remodeling in atrial fibrillation. Cell Calcium (2006) 39, Ghosh A, Praefcke GJ, Renault L, Wittinghofer A, Herrmann C. How guanylate-binding proteins achieve assemblystimulated processive cleavage of GTP to GMP. Nature (2006) 440, Goffart S, Franko A, Clemen CS, Wiesner RJ: alpha-actinin 4 and BAT1 interaction with the Cytochrome c promoter upon skeletal muscle differentiation. Curr Genet (2006) 49, Goser S, Andrassy M, Buss SJ, Leuschner F, Volz CH, Ottl R, Zittrich S, Blaudeck Hardt SE, Pfitzer G, Rose NR, Katus HA, Kaya Z: Cardiac troponin I but not cardiac troponin T induces severe autoimmune inflammation in the myocardium. Circulation (2006) 114, Graef M, Langer T: Substrate specific consequences of central pore mutations in the i-aaa protease Yme1 on substrate engagement. J Struct Biol (2006) 156, 101. Haase I: Immunologic misbehaviour in psoriasis. Response to Clark and Kupper. J Clin Invest (2006) 116, Hacker UT, Schildhauer I, Barroso MC, Kofler DM, Gerner FM, Mysliwietz J, Buening H, Hallek M, King SB: Gene transfer preferentially selects MHC class I positive tumour cells and enhances tumour immunogenicity. Cancer Immunol Immunother (2006) 55, Hahnen E, Eyupoglu IY, Brichta L, Haastert K, Trankle C, Siebzehnrubl FA, Riessland M, Holker I, Claus P, Romstock J, Buslei R, Wirth B, Blumcke I: In vitro and ex vivo evaluation of second-generation histone deacetylase inhibitors for the treatment of spinal muscular atrophy. J Neurochem (2006) 98,
104 Publications Han JW, Leeper L, Rivero F, Chung CY: Role of RacC for the regulation of WASP and PI3 kinase during chemotaxis of Dictyostelium. J Biol Chem (2006) 281, Hanisch FG, Ninkovic T: Immunology of O-glycosylated proteins: approaches to the design of a MUC1 glycopeptide-based tumor vaccine. Curr Protein Pept Sci (2006) 7, Hebebrand J, Dempfle A, Saar K, Thiele H, Herpertz- Dahlmann B, Linder M, Kiefl H, Remschmidt H, Hemminger U, Warnke A, Knolker U, Heiser P, Friedel S, Hinney A, Schafer H, Nürnberg P, Konrad K: A genome-wide scan for attention-deficit/hyperactivity disorder in 155 German sib-pairs. Mol Psychiatry (2006) 11, Helfrich I, Schmitz A, Zigrino P, Michels C, Haase I, Bivic AL, Leitges M, Niessen CM: Role of apkc isoforms and their binding partners Par3 and Par6 in epidermal barrier formation. J Invest Dermatol (2006) 127, Heneka MT, Ramanathan M, Jacobs AH, Dumitrescu- Ozimek L, Bilkei-Gorzo A, Debeir T, Sastre M, Galldiks N, Zimmer A, Hoehn M, Heiss WD, Klockgether T, Staufenbiel M: Locus ceruleus degeneration promotes Alzheimer pathogenesis in amyloid precursor protein 23 transgenic mice. J Neurosci (2006) 26, Henrich KO, Fischer M, Mertens D, Benner A, Wiedemeyer R, Brors B, Oberthuer A, Berthold F, Wei JS, Khan J, Schwab M, Westermann F: Reduced expression of CAMTA1 correlates with adverse outcome in neuroblastoma patients. Clin Cancer Res (2006) 12, Herz C, Aumailley M, Schulte C, Schlotzer-Schrehardt U, Bruckner-Tuderman L, Has C: Kindlin-1 is a phosphoprotein involved in regulation of polarity, proliferation, and motility of epidermal keratinocytes. J Biol Chem (2006) 281, Hinkes B, Wiggins RC, Gbadegesin R, Vlangos CN, Seelow D, Nürnberg G, Garg P, Verma R, Chaib H, Hoskins BE, Ashraf S, Becker C, Hennies HC, Goyal M, Wharram BL, Schachter AD, Mudumana S, Drummond I, Kerjaschki D, Waldherr R, Dietrich A, Ozaltin F, Bakkaloglu A, Cleper R, Basel-Vanagaite L, Pohl M, Griebel M, Tsygin AN, Soylu A, Muller D, Sorli CS, Bunney TD, Katan M, Liu J, Attanasio M, O toole JF, Hasselbacher K, Mucha B, Otto EA, Airik R, Kispert A, Kelley GG, Smrcka AV, Gudermann T, Holzman LB, Nürnberg P, Hildebrandt F: Positional cloning uncovers mutations in PLCE1 responsible for a nephrotic syndrome variant that may be reversible. Nat Genet (2006) 38, Holsken A, Eyupoglu IY, Lueders M, Trankle C, Dieckmann D, Buslei R, Hahnen E, Blumcke I, Siebzehnrubl FA: Ex vivo therapy of malignant melanomas transplanted into organotypic brain slice cultures using inhibitors of histone deacetylases. Acta Neuropathol (Berl) (2006) 112, Hombach A, Koehler H, Rappl G, Abken H: Human CD 4+ T cells lyse target cells via granzyme/perforin upon circumvention of MHC class II-restriction by an antibodyderived immunoreceptor. J Immunol (2006) 177, Hombach A, Schlimper C, Sieves E, Frank S, Schild HH, Sauerbruch T, Schmidt-Wolf IGH, Abken H: A recombinant anti-cea immunoreceptor with combined CD3ζ-CD28 signalling targets T cells from colorectal cancer patients against their tumor cells. Gut (2006) 55, Hoppe UC, Casares JM, Eiskjær H, Hagemann A, Cleland JGF, Freemantle N, Erdmann E: Effect of cardiac resynchronization on the incidence of atrial fibrillation in patients with severe heart failure. Circulation (2006) 114, Horvath TL, Bruning JC: Developmental programming of the hypothalamus: a matter of fat. Nat Med (2006) 12, Hummerich L, Müller R, Hess J, Kokocinski F, Hahn M, Fürstenberger G, Mauch C, Lichter P, Angel P: Identification of novel tumour-associated genes differentially expressed in the process of squamous cell cancer development. Oncogene (2006) 25, Janoschek R, Plum L, Koch L, Munzberg H, Diano S, Shanabrough M, Muller W, Horvath TL, Bruning JC: gp130 signaling in proopiomelanocortin neurons mediates the acute anorectic response to centrally applied ciliary neurotrophic factor. Proc Natl Acad Sci (2006) 103, Julien S, Adriaenssens E, Ottenberg K, Fulan A, Courtand G, Edouart AS, Hanisch FG, Delannoy P, Bourhis XL: ST6GalNAc I expression in MDA-MB-231 breast cancer cells greatly modifies their O-glycosylation pattern and enhances their tumourigenicity. Glycobiology (2006) 16, Kalay E, Li Y, Uzumcu A, Uyguner O, Collin RW, Çaylan R, Ulubil-Emiroglu M, Kersten FF, Hafiz G, van Wijk E, Kayserili H, Rohmann E*, Wagenstaller J, Hoefsloot LH, Strom TM, Nurnberg G, Başerer N, den Hollander AI, Cremers FP, Cremers CW, Becker C, Brunner HG, Nurnberg P, Karaguzel A, Başaran S, Kubisch C, Kremer H, Wollnik B: Mutations in the lipoma HMGIC fusion partner-like 5 (LHFPL5) gene cause autosomal recessive nonsyndromic hearing loss. Hum Mutat (2006) 27, (*MD-PhD Program) Kappert K, Caglayan E, Huntgeburth M, Baumer AT, Sparwel J, Uebel M, Rosenkranz S: 17Beta-estradiol attenuates PDGF signaling in vascular smooth muscle cells at the 99
105 Publications postreceptor level. Am J Physiol Heart Circ Physiol (2006) 290, Kappert K, Sparwel J, Sandin A, Seiler A, Siebolts U, Leppanen O, Rosenkranz S, Ostman A: Antioxidants relieve phosphatase inhibition and reduce PDGF signaling in cultured VSMCs and in restenosis. Arterioscler Thromb Vasc Biol (2006) 26, Kashkar H, Seeger JM, Hombach A, Deggerich A, Yazdanpanah B, Utermohlen O, Heimlich G, Abken H, Kronke M: XIAP targeting sensitizes Hodgkin lymphoma cells for cytolytic T-cell attack. Blood (2006) 108, Kess D, Lindqvist AK, Peters T, Wang H, Zamek J, Nischt R, Broman KW, Blakytny R, Krieg T, Holmdahl R, Scharffetter- Kochanek K: Identification of susceptibility loci for skin disease in a murine psoriasis model. J Immunol (2006) 177, Kirchheiner J, Grundemann D, Schomig E: Contribution of allelic variations in transporters to the phenotype of drug response. J Psychopharmacol (2006) 20, Klose A, Wilbrand-Hennes A, Zigrino P, Weber E, Krieg T, Mauch C, Hunzelmann N: Contact of high-invasive, but not low-invasive, melanoma cells to native collagen I induces the release of mature cathepsin B. Int J Cancer (2006) 118, Klose A, Zigrino P, Dennhofer R, Mauch C, Hunzelmann N: Identification and discrimination of extracellularly active cathepsins B and L in high-invasive melanoma cells. Anal Biochem (2006) 353, Koch M, Veit G, Stricker S, Bhatt P, Kutsch S, Zhou P, Reinders E, Hahn RA, Song R, Burgeson RE, Gerecke DR, Mundlos S, Gordon MK: Expression of type XXIII collagen mrna and protein. J Biol Chem (2006) 281, Kociok N, Krohne TU, Poulaki V, Joussen AM: Geldanamycin treatment reduces neovascularization in a mouse model of retinopathy of prematurity. Graefes Arch Clin Exp Ophthalmol (2006), in press. Kociok N, Radetzky S, Krohne TU, Gavranic C, Joussen AM: Pathological but not physiological retinal neovascularization is altered in TNF-Rp55-receptordeficient mice. Invest Ophthalmol Vis Sci (2006) 47, Köhling R, Nischt R, Vasudevan A, Ho M, Weiergraber M, Schneider T, Smyth N: Nidogen and nidogen-associated basement membrane proteins and neuronal plasticity. Neurodegener Dis (2006) 3, Konrad M, Schaller A, Seelow D, Pandey AV, Waldegger S, Lesslauer A, Vitzthum H, Suzuki Y, Luk JM, Becker C, Schlingmann KP, Schmid M, Rodriguez-Soriano J, Ariceta G, Cano F, Enriquez R, Juppner H, Bakkaloglu SA, Hediger MA, Gallati S, Neuhauss SC, Nürnberg P, Weber S: Mutations in the tight-junction gene claudin 19 (CLDN19) are associated with renal magnesium wasting, renal failure, and severe ocular involvement. Am J Hum Genet (2006) 79, Koppold B, Sauer G, Buening H, Hallek M, Kreienberg R, Deissler H, Kurzeder C: Chemotherapeutic agents enhance AAV2-mediated gene transfer into breast cancer cells promoting CD40 ligand-based immunotherapy. J Cancer Res Clin Oncol (2006) 132, Krieger A, Radhakrishnan K, Pereverzev A, Siapich SA, Banat M, Kamp MA, Leroy J, Klockner U, Hescheler J, Weiergraber M, Schneider T: The molecular chaperone hsp70 interacts with the cytosolic II-III loop of the Ca v 2.3 E-type voltage-gated Ca 2+ channel. Cell Physiol Biochem (2006) 17, Krones-Herzig A, Mesaros A, Metzger D, Ziegler A, Lemke U, Bruning JC, Herzig S: Signal-dependent control of gluconeogenic key enzyme genes through coactivatorassociated arginine methyltransferase 1. J Biol Chem (2006) 281, Krut O, Wiegmann K, Kashkar H, Yazdanpanah B, Kronke M: Novel tumor necrosis factor-responsive mammalian neutral sphingomyelinase-3 is a C-tail-anchored protein. J Biol Chem (2006) 281, Kunzelmann S, Praefcke GJ, Herrmann C: Transient kinetic investigation of GTP hydrolysis catalyzed by interferongamma-induced hgbp1 (human guanylate binding protein 1). J Biol Chem (2006) 281, Ladage D, Brixius K, Hoyer H, Steingen C*, Wesseling A, Malan D, Bloch W, Schwinger RH: Mechanisms underlying nebivolol-induced endothelial nitric oxide synthase activation in human umbilical vein endothelial cells. Clin Exp Pharmacol Physiol (2006) 33, (*MD-PhD Program) Laser H, Conforti L, Morreale G, Mack TG, Heyer M, Haley JE, Wishart TM, Beirowski B, Walker SA, Haase G, Celik A, Adalbert R, Wagner D, Grumme D, Ribchester RR, Plomann M, Coleman MP: The slow Wallerian degeneration protein, WldS, binds directly to VCP/p97 and partially redistributes it within the nucleus. Mol Biol Cell (2006) 17, Lazar A, Zimmermann T, Koch W, Grundemann D, Schomig A, Kastrati A, Schomig E: Lower prevalence of the OCT2 Ser270 allele in patients with essential hypertension. Clin Exp Hypertens (2006) 28, Lebbink RJ, de Ruiter T, Adelmeijer J, Brenkman AB, van Helvoort JM, Koch M, Farndale RW, Lisman T, Sonnenberg A, Lenting PJ, Meyaard L: Collagens are functional, high 100
106 Publications affinity ligands for the inhibitory immune receptor LAIR-1. J Exp Med (2006) 203, Li Y, Wollnik B, Pabst S, Lennarz M, Rohmann E, Gillissen A, Vetter H, Grohé C: BTNL2 gene variant and sarcoidosis. Thorax (2006) 61, Lichtenauer U*, Duchniewicz M, Kolanczyk M, Hoeflich A, Hahner S, Else T, Bicknell A, Zemojtel T, Stallings N, Schulte D, Kamps M, Hammer G, Scheele J, Beuschlein F: Pbx1 and SF-1 synergistically regulate adrenocortical growth and steroidogenesis. Endocrinology (2007) 148, (*MD-PhD Program) Loffek S, Zigrino P, Steiger J, Kurschat P, Smola H, Mauch C: Melanoma cell-derived vascular endothelial growth factor induces endothelial tubulogenesis within fibrin gels by a metalloproteinase-mediated mechanism. Eur J Cell Biol (2006) 85, Lugassy J, Itin P, Ishida-Yamamoto A, Holland K, Huson S, Geiger D, Hennies HC, Indelman M, Bercovich D, Uitto J, Bergman R, McGrath JA, Richard G, Sprecher E: Naegeli- Franceschetti-Jadassohn syndrome and dermatopathia pigmentosa reticularis: two allelic ectodermal dysplasias caused by dominant mutations in KRT14. Am J Hum Genet (2006) 79, Luke M, Krott R, Warga M, Szurman P, Grisanti S, Bartz- Schmidt KU, Schneider T, Luke C: Effects of the protein tyrosine kinase inhibitor genistein and taurine on retinal function in isolated superfused retina. Graefes Arch Clin Exp Ophthalmol (2006), in press. Mak GZ, Kavanaugh GM, Buschmann MM, Stickley SM, Koch M, Goss KH, Waechter H, Zuk A, Matlin KS: Regulated synthesis and functions of laminin 5 in polarized madindarby canine kidney epithelial cells. Mol Biol Cell (2006) 17, Maniura-Weber K, Helm M, Engemann K, Eckertz S, Mollers M, Schauen M, Hayrapetyan A, von Kleist-Retzow JC, Lightowlers RN, Bindoff LA, Wiesner RJ: Molecular dysfunction associated with the human mitochondrial 3302A>G mutation in the MTTL1 (mt-trnaleu(uur)) gene. Nucleic Acids Res (2006) 22, Matusch A 1, Meyer PT 1 *, Bier D, Holschbach M, Elmenhorst D, Winz OH, Zilles K, Coenen HH, Bauer A: Metabolism of the A1 adenosine PET ligand [18F]CPFPX by CYP1A2: implications for bolus/infusion PET studies. Nucl Med Biol (2006) 33, (*MD-PhD Program, 1 both authors contributed equally) Marko M, Prabhu Y, Muller R, Blau-Wasser R, Schleicher M, Noegel AA: The annexins of Dictyostelium. Eur J Cell Biol (2006) 85, Meyer PT*, Circiumaru V, Cardi CA, Thomas DH, Bal H, Acton PD: Simplified quantification of small animal [18F]FDG PET studies using a standard arterial input function. Eur J Nucl Med Mol Imaging (2006) 33, (*MD-PhD Program) Meyer PT*, Elmenhorst D, Matusch A, Winz O, Zilles K, Bauer A: 18F-CPFPX PET: On the generation of parametric images and the effect of scan duration. J Nucl Med (2006) 47, (*MD-PhD Program) Meyer PT*, Elmenhorst D, Matusch A, Winz O, Zilles K, Bauer A: A1 adenosine receptor PET using [18F]CPFPX: displacement studies in humans. Neuroimage (2006) 32, (*MD-PhD Program) Michels G, Er F, Eicks M, Herzig S, Hoppe UC: Long-term and immediate effect of testosterone on single T-type calcium channel in neonatal rat cardiomyocytes. Endocrinology (2006) 147, Mizrachi-Koren M, Shemer S, Morgan M, Indelman M, Khamaysi Z, Petronius D, Bitterman-Deutsch O, Hennies HC, Bergman R, Sprecher E: Homozygosity mapping as a screening tool for the molecular diagnosis of hereditary skin diseases in consanguineous populations. J Am Acad Dermatol (2006) 55, Natrajan R, Little SE, Reis-Filho JS, Hing L, Messahel B, Grundy PE, Dome JS, Schneider T, Vujanic GM, Pritchard- Jones K, Jones C: Amplification and overexpression of CACNA1E correlates with relapse in favorable histology Wilms tumors. Clin Cancer Res (2006) 12, Nenci A, Huth M, Funteh A, Schmidt-Supprian M, Bloch W, Metzger D, Chambon P, Rajewsky K, Krieg T, Haase I, Pasparakis M: Skin lesion development in a mouse model of incontinentia pigmenti is triggered by NEMO deficiency in epidermal keratinocytes and requires TNF signaling. Hum Mol Genet (2006) 15, Netzer C, Todt U, Heinze A, Freudenberg J, Zumbroich V, Becker T, Goebel I, Ohlraun S, Goebel H, Kubisch C: Haplotype-based systematic association studies of ATP1A2 in migraine with aura. Am J Med Genet B Neuropsychiatr Genet (2006) 141, Niemann C: Controlling the stem cell niche: right time, right place, right strength. Bioessays (2006) 28, 1-5. Niessen C, Yap AS: Another job for the talented p120- catenin. Cell (2006) 127, Oberthuer A, Berthold F, Warnat P, Hero B, Kahlert Y, Spitz R, Ernestus K, Konig R, Haas S, Eils R, Schwab M, Brors B, Westermann F, Fischer M: Customized oligonucleotide microarray gene expression-based classification of neuroblastoma patients outperforms current clinical risk stratification. J Clin Oncol (2006) 24, Oji V, Hautier JM, Ahvazi B, Hausser I, Aufenvenne K, Walker T, Seller N, Steijlen PM, Küster W, Hovnanian 101
107 Publications A, Hennies HC, Traupe H: Bathing suit ichthyosis is caused by transglutaminase-1 deficiency: evidence for a temperature-sensitive phenotype. Hum Mol Genet (2006) 15, Otto C, Jensen M, Dietlein M, Fischer T, Schmidt M, Tawadros S, Borner SM, Weber SA, Spitz R, Bloch W, Berthold F, Schicha H, Schomacker K: Localization of 131Ilabelled monoclonal antibody ERIC1 in a subcutaneous xenograft model of neuroblastoma in SCID mice. Nucl Med Commun (2006) 27, Parry S, Hanisch FG, Leir SH, Sutton-Smith M, Morris HR, Dell A, Harris A: N-Glycosylation of the MUC1 mucin in epithelial cells and secretions. Glycobiology (2006) 16, Pashenkov M, Goess G, Wagner C, Hormann M, Jandl T, Moser A, Britten CM, Smolle J, Koller S, Mauch C, Tantcheva-Poor I, Grabbe S, Loquai C, Esser S, Franckson T, Schneeberger A, Haarmann C, Krieg T, Stingl G, Wagner SN: Phase II trial of a toll-like receptor 9-activating oligonucleotide in patients with metastatic melanoma. J Clin Oncol (2006) 24, Perabo L, Endell J, King S, Lux K, Goldnau D, Hallek M, Buning H: Combinatorial engineering of a gene therapy vector: directed evolution of adeno-associated virus. J Gene Med (2006) 8, Perabo L, Goldnau D, White K, Endell J, Boucas J, Humme S, Work LM, Janicki H, Hallek M, Baker AH, Buning H: Heparan sulfate proteoglycan binding properties of adenoassociated virus retargeting mutants and consequences for their in vivo tropism. J Virol (2006) 80, Pereira L, Matthes J, Schuster I, Valdivia HH, Herzig S, Richard S, Gomez AM: Mechanisms of Ca 2+ i transient decrease in cardiomyopathy of db/db type 2 diabetic mice. Diabetes (2006) 55, Pérez-Otaño I, Lujan R, Tavalin SJ, Plomann M, Modregger J, Liu XB, Jones EG, Heinemann SF, Lo DC, Ehlers MD: Endocytosis and synaptic removal of NR3A-containing NMDA receptors by PACSIN1/syndapin1. Nat Neurosci (2006) 9, Peters T, Bloch W, Wickenhauser C, Tawadros S, Oreshkova T, Kess D, Krieg T, Muller W, Scharffetter-Kochanek K: Terminal B cell differentiation is skewed by deregulated interleukin-6 secretion in beta2 integrin-deficient mice. J Leukoc Biol (2006) 80, Plum L, Belgardt BF, Bruning JC: Central insulin action in energy and glucose homeostasis. J Clin Invest (2006) 116, Plum L, Ma X, Hampel B, Balthasar N, Coppari R, Munzberg H, Shanabrough M, Burdakov D, Rother E*, Janoschek R, Alber J, Belgardt BF, Koch L, Seibler J, Schwenk F, Fekete C, Suzuki A, Mak TW, Krone W, Horvath TL, Ashcroft FM, Bruning JC: Enhanced PIP 3 signaling in POMC neurons causes K ATP channel activation and leads to diet-sensitive obesity. J Clin Invest (2006) 116, (*MD-PhD Program) Popov A, Abdullah Z, Wickenhauser C, Saric T, Driesen J, Hanisch FG, Domann E, Raven EL, Dehus O, Hermann C, Eggle D, Debey S, Chakraborty T, Kronke M, Utermohlen O, Schultze JL: Indoleamine 2,3-dioxygenase-expressing dendritic cells form suppurative granulomas following Listeria monocytogenes infection. J Clin Invest (2006) 116, Popowicz GM, Schleicher M, Noegel AA, Holak TA: Filamins: promiscuous organizers of the cytoskeleton. Trends Biochem Sci (2006) 31, Ramirez A, Heimbach A, Grundemann J, Stiller B, Hampshire D, Cid LP, Goebel I, Mubaidin AF, Wriekat AL, Roeper J, Al-Din A, Hillmer AM, Karsak M, Liss B, Woods CG, Behrens MI, Kubisch C: Hereditary parkinsonism with dementia is caused by mutations in ATP13A2, encoding a lysosomal type 5 P-type ATPase. Nat Genet (2006) 38, Riessland M, Brichta L, Hahnen E, Wirth B: The benzamide M344, a novel histone deacetylase inhibitor, significantly increases SMN2 RNA/protein levels in spinal muscular atrophy cells. Hum Genet (2006) 120, Rivero F, Maniak M: Quantitative and microscopic methods for studying the endocytic pathway. Methods Mol Biol (2006) 346, Rohmann E*, Brunner HG, Kayserili H, Uyguner O, Nurnberg G, Lew ED, Dobbie A, Eswarakumar VP, Uzumcu A, Ulubil- Emeroglu M, Leroy JG, Li Y, Becker C, Lehnerdt K, Cremers CW, Yuksel-Apak M, Nurnberg P, Kubisch C, Schlessinger J, van Bokhoven H, Wollnik B: Mutations in different components of FGF signaling in LADD syndrome. Nat Genet (2006) 38, (*MD-PhD Program) Roll S, Seul J, Paulsson M, Hartmann U: Testican-1 is dispensable for mouse development. Matrix Biol (2006) 25, Rosenkranz S, Brixius K, Halbach R, Diedrichs H, Schwinger RH: Phosphodiesterase type 5 inhibitor sildenafil citrate does not potentiate the vasodilative properties of nebivolol in rat aorta. Life Sci (2006) 78, Roth D, Piekarek M, Paulsson M, Christ H, Krieg T, Bloch W, Davidson JM, Eming SA: Plasmin modulates vascular endothelial growth factor-a-mediated angiogenesis during wound repair. Am J Pathol (2006) 168, Roth U, Muller S, Hanisch FG: Proteomic analysis of 102
108 Publications Dictyostelium discoideum. Methods Mol Biol (2006) 346, Rugarli EI, Langer T: Translating m-aaa protease function in mitochondria to hereditary spastic paraplegia. Trends Mol Med (2006) 12, Rybakin V, Gounko NV, Spate K, Honing S, Majoul IV, Duden R, Noegel AA: Crn7 interacts with AP-1 and is required for the maintenance of Golgi morphology and protein export from the Golgi. J Biol Chem (2006) 281, Sayer JA, Otto EA, O toole JF, Nürnberg G, Kennedy MA, Becker C, Hennies HC, Helou J, Attanasio M, Fausett BV, Utsch B, Khanna H, Liu Y, Drummond I, Kawakami I, Kusakabe T, Tsuda M, Ma L, Lee H, Larson RG, Allen SJ, Wilkinson CJ, Nigg EA, Shou C, Lillo C, Williams DS, Hoppe B, Kemper MJ, Neuhaus T, Parisi MA, Glass IA, Petry M, Kispert A, Gloy J, Ganner A, Walz G, Zhu X, Goldman D, Nürnberg P, Swaroop A, Leroux MR, Hildebrandt F: The centrosomal protein nephrocystin-6 is mutated in Joubert syndrome and activates transcription factor ATF4. Nat Genet (2006) 38, Schauen M, Hornig-Do HT, Schomberg S, Herrmann G, Wiesner RJ: Mitochondrial electron transport chain activity is not involved in ultraviolet A (UVA)-induced cell death. Free Radic Biol Med (2007) 42, Schauen M, Spitkovsky D, Schubert J, Fischer JH, Hayashi J, Wiesner RJ: Respiratory chain deficiency slows down cell-cycle progression via reduced ROS generation and is associated with a reduction of p21cip1/waf1. J Cell Physiol (2006) 209, Schmid EM, Ford MG, Burtey A, Praefcke GJ, Peak-Chew SY, Mills IG, Benmerah A, McMahon HAT: Role of the AP2 beta-appendage hub in recruiting partners for clathrincoated vesicle assembly. PLoS Biol (2006) 4, e262. Schmidt A, Ladage D, Steingen C*, Brixius K, Schinkothe T, Klinz FJ, Schwinger RH, Mehlhorn U, Bloch W: Mesenchymal stem cells transmigrate over the endothelial barrier. Eur J Cell Biol (2006) 85, (*MD-PhD Program) Schmidt M, Simon T, Hero B, Eschner W, Dietlein M, Sudbrock F, Bongartz R, Berthold F, Schicha H: Is there a benefit of 131 I-MIBG therapy in the treatment of children with stage 4 neuroblastoma? A retrospective evaluation of The German Neuroblastoma Trial NB97 and implications for The German Neuroblastoma Trial NB2004. Nuklearmedizin (2006) 45, Schmitz M, Becker A, Schmitz A, Weirich C, Paulsson M, Zaucke F, Dinser R: Disruption of extracellular matrix structure may cause pseudoachondroplasia phenotypes in the absence of impaired cartilage oligomeric matrix protein secretion. J Biol Chem (2006) 281, Schneider-Brachert W, Tchikov V, Merkel O, Jakob M, Hallas C, Kruse ML, Groitl P, Lehn A, Hildt E, Held-Feindt J, Dobner T, Kabelitz D, Kronke M, Schutze S: Inhibition of TNF receptor 1 internalization by adenovirus 14.7K as a novel immune escape mechanism. J Clin Invest (2006) 116, Schoenen F, Wirth B: The zinc finger protein ZNF297B interacts with BDP1, a subunit of TFIIIB. Biol Chem (2006) 387, Semkova I, Fauser S, Lappas A, Smyth N, Kociok N, Kirchhof B, Paulsson M, Poulaki V, Joussen AM: Overexpression of FasL in retinal pigment epithelial cells reduces choroidal neovascularization. FASEB J (2006) 20, Servotte S, Zhang Z, Lambert CA, Ho TT, Chometon G, Eckes B, Krieg T, Lapiere CM, Nusgens BV, Aumailley M: Establishment of stable human fibroblast cell lines constitutively expressing active Rho-GTPases. Protoplasma (2006) 229, Sheffler DJ, Kroeze WK, Garcia BG, Deutch AY, Hufeisen SJ, Leahy P, Bruning JC, Roth BL: p90 ribosomal S6 kinase 2 exerts a tonic brake on G protein-coupled receptor signaling. Proc Natl Acad Sci USA (2006) 103, Shi X, Semkova I, Muther PS, Dell S, Kociok N, Joussen AM: Inhibition of TNF-alpha reduces laser-induced choroidal neovascularization. Exp Eye Res (2006) 83, Siebler J, Protzer U, Wirtz S, Schuchmann M, Höhler T, Galle PR, Neurath M: Adenoviral overexpression of STAT- 1 does not inhibit Hepatitis B-Virus replication. Euro J Gastro (2006) 18, Simon T, Hero B, Bongartz R, Schmidt M, Muller RP, Berthold F: Intensified external-beam radiation therapy improves the outcome of stage 4 neuroblastoma in children > 1 year with residual local disease. Strahlenther Onkol (2006) 182, Simon T, Spitz R, Hero B, Berthold F, Faldum A: Risk estimation in localized unresectable single copy MYCN neuroblastoma by the status of chromosomes 1p and 11q. Cancer Lett (2006) 237, Sodersten F, Ekman S, Schmitz M, Paulsson M, Zaucke F: Thrombospondin-4 and cartilage oligomeric matrix protein form heterooligomers in equine tendon. Connect Tissue Res (2006) 47, Somesh BP, Neffgen C, Iijima M, Devreotes P, Rivero F: Dictyostelium RacH regulates endocytic vesicular trafficking and is required for localization of vacuolin. Traffic (2006) 7, Somesh BP, Vlahou G, Iijima M, Insall RH, Devreotes P, Rivero F: RacG regulates morphology, phagocytosis and chemotaxis. Euk Cell (2006) 5,
109 Publications Spitz R, Betts DR, Simon T, Boensch M, Oestreich J, Niggli FK, Ernestus K, Berthold F, Hero B: Favorable outcome of triploid neuroblastomas: a contribution to the special oncogenesis of neuroblastoma. Cancer Genet Cytogenet (2006) 167, Spitz R, Hero B, Simon T, Berthold F: Loss in chromosome 11q identifies tumors with increased risk for metastatic relapses in localized and 4S neuroblastoma. Clin Cancer Res (2006) 12, Spitz R, Oberthuer A, Zapatka M, Brors B, Hero B, Ernestus K, Oestreich J, Fischer M, Simon T, Berthold F: Oligonucleotide array-based comparative genomic hybridization (acgh) of 90 neuroblastomas reveals aberration patterns closely associated with relapse pattern and outcome. Genes Chromosomes Cancer (2006) 45, Spitzer C, Schellmann S, Sabovljevic A, Shahriari M, Keshavaiah C, Bechtold N, Herzog M, Muller S, Hanisch FG, Hulskamp M: The Arabidopsis elch mutant reveals functions of an ESCRT component in cytokinesis. Development (2006) 133, Stalz H, Roth U, Schleuder D, Macht M, Haebel S, Strupat K, Peter-Katalinic J, Hanisch FG: The Geodia cydonium galectin exhibits prototype and chimera-type characteristics and a unique sequence polymorphism within its carbohydrate recognition domain. Glycobiology (2006) 16, Stehle R, Solzin J, Iorga B, Gomez D, Blaudeck N, Pfitzer G: Mechanical properties of sarcomeres during cardiac myofibrillar relaxation: stretch-induced cross-bridge detachment contributes to early diastolic filling. J Muscle Res Cell Motil (2006) 27, Stratis A, Pasparakis M, Markur D, Knaup R, Pofahl R, Metzger D, Chambon P, Krieg T, Haase I: Localized inflammatory skin disease following inducible ablation of I kappa B kinase 2 in murine epidermis. J Invest Dermatol (2006) 126, Stratis A, Pasparakis M, Rupec RA, Markur D, Hartmann K, Scharffetter-Kochanek K, Peters T, van Rooijen N, Krieg T, Haase I: Pathogenic role for skin macrophages in a mouse model of keratinocyte-induced psoriasis-like skin inflammation. J Clin Invest (2006) 116, Telley IA, Denoth J, Stüssi E, Pfitzer G, Stehle R: Halfsarcomere dynamics in myofibril during activation and relaxation studied by tracking fluorescent markers. Biophys J (2006) 90, Telley IA, Stehle R, Ranatunga KW, Pfitzer G, Stüssi E, Denoth J: Dynamic behaviour of half-sarcomeres during and after stretch in activated psoas myofibrils: sarcomere asymmetry but no sarcomere popping. J Physiol (2006) 573, Ten Freyhaus H, Huntgeburth M, Wingler K, Schnitker J, Baumer AT, Vantler M, Bekhite MM, Wartenberg M, Sauer H, Rosenkranz S: Novel Nox inhibitor VAS2870 attenuates PDGF-dependent smooth muscle cell chemotaxis, but not proliferation. Cardiovasc Res (2006) 71, Theisen DM, Pongratz C, Wiegmann K, Rivero F, Krut O, Kronke M: Targeting of HIV-1 Tat traffic and function by transduction-competent single chain antibodies. Vaccine (2006) 24, Todt U, Freudenberg J, Goebel I, Netzer C, Heinze A, Heinze- Kuhn K, Gobel H, Kubisch C: MTHFR C677T polymorphism and migraine with aura. Ann Neurol (2006) 60, Untergasser A, Zedler U*, Langenkamp A, Hosel M, Quasdorff M*, Esser K*, Dienes HP, Tappertzhofen B, Kolanus W, Protzer U: Dendritic cells take up viral antigens but do not support the early steps of hepatitis B virus infection. Hepatology (2006) 43, (*MD-PhD Program) Uyguner O, Siva A, Kayserili H, Saip S, Tasyurekli M, Apak MY, Isik N, Yilmaz, Akman-Deniz G, Albayram S, Altintas A, Oz B, Wollnik B: The R110C mutation in Notch3 causes variable clinical features in two Turkish families with CADASIL syndrome. J Neurol Sci (2006) 246, Uzumcu A, Norgett EE, Dindar A, Uyguner O, Nisli K, Kayserili H, Sahin SE, Dupont E, Severs NJ, Leigh IM, Yuksel- Apak M, Kelsell DP, Wollnik B: Loss of desmoplakin isoform I causes early onset cardiomyopathy and heart failure in a Naxos-like syndrome. J Med Genet (2006) 43, e05. Vaclavicek A, Hemminki K, Bartram CR, Wagner K, Wappenschmidt B, Meindl A, Schmutzler RK, Klaes R, Untch M, Burwinkel B, Forsti A: Association of prolactin and its receptor gene regions with familial breast cancer. J Clin Endocrinol Metab (2006) 91, Vantler M, Huntgeburth M, Caglayan E, Ten Freyhaus H, Schnabel P, Rosenkranz S: PI3-kinase/Akt-dependent antiapoptotic signaling by the PDGF alpha receptor is negatively regulated by Src family kinases. FEBS Lett (2006) 580, Veit G, Hansen U, Keene DR, Bruckner P, Chiquet-Ehrismann R, Chiquet M, Koch M: Collagen XII interacts with avian tenascin-x through its NC3 domain. J Biol Chem (2006) 281, Veit G, Kobbe B, Keene DR, Paulsson M, Koch M, Wagener R: Collagen XXVIII, a novel von Willebrand factor A domain-containing protein with many imperfections in the collagenous domain. J Biol Chem (2006) 281, Vlahou G, Rivero F: Rho GTPase signaling in Dictyostelium discoideum: insights from the genome. Eur J Cell Biol (2006) 85,
110 Publications Wagner K, Hemminki K, Grzybowska E, Klaes R, Burwinkel B, Bugert P, Schmutzler RK, Wappenschmidt B, Butkiewicz D, Pamula J, Pekala W, Forsti A: Polymorphisms in genes involved in GH1 release and their association with breast cancer risk. Carcinogenesis (2006) 27, Walcher D, Babiak C, Poletek P, Rosenkranz S, Bach H, Betz S, Durst R, Grub M, Hombach V, Strong J, Marx N: C- Peptide induces vascular smooth muscle cell proliferation: involvement of SRC-kinase, phosphatidylinositol 3-kinase, and extracellular signal-regulated kinase 1/2. Circ Res (2006) 99, Wang H, Peters T, Kess D, Sindrilaru A, Oreshkova T, Van Rooijen N, Stratis A, Renkl AC, Sunderkotter C, Wlaschek M, Haase I, Scharffetter-Kochanek K: Activated macrophages are essential in a murine model for T cell-mediated chronic psoriasiform skin inflammation. J Clin Invest (2006) 116, Weiergraber M*, Henry M, Krieger A, Kamp M, Radhakrishnan K, Hescheler J, Schneider T: Altered seizure susceptibility in mice lacking the Ca v 2.3 E-type Ca 2+ channel. Epilepsia (2006) 47, (*MD-PhD Program) Weiergraber M*, Kamp MA, Radhakrishnan K, Hescheler J, Schneider T: The Ca(v)2.3 voltage-gated calcium channel in epileptogenesis--shedding new light on an enigmatic channel. Neurosci Biobehav Rev (2006) 30, (*MD-PhD Program) Wieland T, Herzig S: Specificity and diversity in Gi/omediated signaling: how the heart operates the RGS brake pedal. Circ Res (2006) 98, Wiesner RJ, Zsurka G, Kunz WS: Mitochondrial DNA damage and the aging process: facts and imaginations. Free Radic Res (2006) 40, Wilkening S, Bermejo JL, Burwinkel B, Klaes R, Wappenschmidt B, Schmutzler RK, Meindl A, Bugert P, Untch M, Hemminki K, Försti A: The single nucleotide poly-morphism IVS1+309 in MDN2 does not affect risk of familial breast cancer. Cancer Res (2006) 66, Wirtenberger M, Frank B, Hemminki K, Klaes R, Schmutzler RK, Wappenschmidt B, Meindl A, Kiechle M, Arnold N, Weber BH, Niederacher D, Bartram CR, Burwinkel B: Interaction of Werner and Bloom syndrome genes with p53 in familial breast cancer. Carcinogenesis (2006) 27, Wirtenberger M, Tchatchou S, Hemminki K, Schmutzhard J, Sutter C, Schmutzler RK, Meindl A, Wappenschmidt B, Kiechle M, Arnold N, Weber BH, Niederacher D, Bartram CR, Burwinkel B: Associations of genetic variants in the estrogen receptor coactivators PPARGC1A, PPARGC1B and EP300 with familial breast cancer. Carcinogenesis (2006) 27, Wirth B, Brichta L, Hahnen E: Spinal muscular atrophy and therapeutic prospects. Prog Mol Subcell Biol (2006) 44, Wirth B, Brichta L, Hahnen E: Spinal muscular atrophy: from gene to therapy. Semin Pediatr Neurol (2006) 13, Wirth B, Brichta L, Schrank B, Lochmuller H, Blick S, Baasner A, Heller R: Mildly affected patients with spinal muscular atrophy are partially protected by an increased SMN2 copy number. Hum Genet (2006) 119, Work LM, Büning H, Hunt E, Nicklin SA, Denby L, Britton N, Leike K, Odenthal M, Drebber U, Hallek M, Baker AH: Vascular bed-targeted in vivo gene delivery using tropismmodified adeno-associated viruses. Mol Ther (2006) 13, Yusof AM, Jaenicke E, Pedersen JS, Noegel AA, Schleicher M, Hofmann A: Studies on oligomerisation of cyclaseassociated protein from Dictyostelium discoideum in solution. J Mol Biol (2006) 362, Zhang ZG, Bothe I, Hirche F, Zweers M, Gullberg D, Pfitzer G, Krieg T, Eckes B, Aumailley M: Interactions of primary fibroblasts and keratinocytes with extracellular matrix proteins: contribution of alpha2beta1 integrin. J Cell Sci (2006) 119, Zhang ZG, Lambert CA, Servotte S, Chometon G, Eckes B, Krieg T, Lapière CM, Nusgens BV, Aumailley M: Effects of constitutively active GTPases on fibroblast behavior. Cell Mol Cell Sci (2006) 63, Zinn M, Aumailley M, Krieg T, Smola H: Expression of laminin 5 by parental and c-ha-ras-transformed HaCaT keratinocytes in organotypic cultures. Eur J Cell Biol (2006) 85, Abdullah Z, Saric T, Kashkar H, Baschuk N, Yazdanpanah B, Fleischmann BK, Hescheler J, Kronke M, Utermohlen O: Serpin-6 expression protects embryonic stem cells from lysis by antigen-specific CTL. J Immunol (2007) 178, Abken H: Chimeric T cell receptor. Encyclopedia of Cancer (2007), in press. Antzelevitch C, Pollevick GD, Cordeiro JM, Casis O, Sanguinetti MC, Aizawa Y, Guerchicoff A, Pfeiffer R, Oliva A, Wollnik B, Gelber P, Bonaros EP Jr, Burashnikov E, Wu Y, 105
111 Publications Sargent JD, Schickel S, Oberheiden R, Bhatia A, Hsu LF, Haissaguerre M, Schimpf R, Borggrefe M, Wolpert C: Lossof-function mutations in the cardiac calcium channel underlie a new clinical entity characterized by ST-segment elevation, short QT intervals, and sudden cardiac death. Circulation (2007) 115, Arasada R, Gloss A, Tunggal B, Joseph J M, Rieger D, Mondal S, Schleicher M, Noegel AA: Profilin isoforms in Dictyostelium discoideum. Biochem Biophysc Acta (2007), in press. Bach M, Grigat S, Pawlik B, Fork C, Utermöhlen O, Pal S, Banczyk D, Lazar A, Schömig E, Gründemann D: Fast setup of doxycycline-inducible protein expression in human cell lines with a single plasmid based on Epstein-Barr virus replication and the simple tetracycline repressor. FEBS J (2007) 274, Bakthavatsalam D, Brazill D, Gomer RH, Eichinger L, Rivero F, Noegel AA: A G protein-coupled receptor with a lipid kinase domain is involved in cell-density sensing. Curr Biol (2007) 17, Baschuk N, Utermohlen O, Gugel R, Warnecke G, Karow U, Paulsen D, Brombacher F, Kronke M, Deppert W: Interleukin-4 impairs granzyme-mediated cytotoxicity of Simian virus 40 large tumor antigen-specific CTL in BALB/c mice. Cancer Immunol Immunother (2007), in press. Binczek E, Jenke B, Holz B, Gunter RH, Thevis M, Stoffel W: Obesity resistance of the stearoyl-coa desaturase-deficient (scd1-/-) mouse results from disruption of the epidermal lipid barrier and adaptive thermoregulation. Biol Chem (2007) 388, Blume JJ, Halbach A, Behrendt D, Paulsson M, Plomann M: EHD proteins are associated with tubular and vesicular compartments and interact with specific phospholipids. Exp Cell Res (2007) 313, Brandt-Bohne U, Keene DR, White FA, Koch M: MEGF9: a novel transmembrane protein with a strong and developmentally regulated expression in the nervous system. Biochem J (2007) 401, Collin RW, Kalay E, Oostrik J, Çaylan R, Wollnik B, Arslan S, den Hollander A, Birinci Y, Strom TM, Lichtner P, Toraman B, Hoefsloot LH, Cremers CWR, Brunner HG, Cremers FP, Karaguzel A, Kremer H: The involvement of DFNB59 mutations in autosomal recessive non-syndromic hearing impairment. Hum Mutat (2007), in press. Coras R, Holsken A, Seufert S, Hauke J, Eyupoglu IY, Reichel M, Trankle C, Siebzehnrubl FA, Buslei R, Blumcke I, Hahnen E: The peroxisome proliferator-activated receptor-{gamma} agonist troglitazone inhibits transforming growth factor-{beta}-mediated glioma cell migration and brain invasion. Mol Cancer Ther (2007) 6, Crisponi L, Crisponi G, Meloni A, Toliat MR, Nürnberg G, Usala U, Uda M, Masala M, Höhne W, Becker C, Marongiu M, Chiappe F, Kleta R, Rauch A, Wollnik B, Strasser F, Reese R, Jakobs C, Kurlemann G, Cao A, Nürnberg P, Rutsch F: Crisponi syndrome is caused by mutations in the CRLF1 gene and is allelic to cold-induced sweating syndrome. Am J Hum Genet (2007), in press. Dressman D, Ahearn ME, Yariz KO, Basterrecha H, Martinez F, Palau F, Barmada MM, Clark RD, Meindl A, Wirth B, Hoffman EP, Baumbach-Reardon L: X-linked infantile spinal muscular atrophy: clinical definition and molecular mapping. Genet Med (2007) 9, Eichenauer DA, Simhadri VL, von Strandmann EP, Ludwig A, Matthews V, Reiners KS, von Tresckow B*, Saftig P, Rose-John S, Engert A, Hansen HP: ADAM10 inhibition of human CD30 shedding increases specificity of targeted immunotherapy in vitro. Cancer Res (2007) 67, (*MD-PhD Program) Elcioglu NH, Akin B, Toker E, Elcioglu M, Kaya A, Tuncali T, Wollnik B, Hornby S, Akarsu NA: Colobomatous macrophthalmia with microcornea syndrome maps to 2p23-p16 region. Am J Med Genet (2007), in press. Elmenhorst D, Meyer PT*, Matusch A, Winz OH, Zilles K, Bauer A: Test-retest stability of cerebral A(1) adenosine receptor quantification using [(18)F]CPFPX and PET. J Nucl Med Mol Imaging (2007), in press. (*MD-PhD Program) Elmenhorst D, Meyer PT*, Winz OH, Matusch A, Ermert J, Coenen HH, Basheer R, Haas HL, Zilles K, Bauer A: Sleep deprivation increases A1 adenosine receptor binding in the human brain: a positron emission tomography study. J Neurosci (2007) 27, (*MD-PhD Program) Er F, Michels G, Brandt MC, Khan I, Haase H, Eicks M, Lindner M, Hoppe UC: Impact of testosterone on cardiac L-type calcium channels and Ca 2+ sparks: Acute actions antagonize chronic effects. Cell Calcium (2007) 41, Fischer YH 1, Miletic H 1 *, Litwak S, Giroglou T, Stenzel W, Neumann H, von Laer D: A retroviral packaging cell line for pseudotype vectors based on glioma-infiltrating progenitor cells. J Gene Med (2007), in press. (*MD-PhD Program, 1 both authors contributed equally to these papers) Frank B, Bermejo JL, Hemminki K, Sutter C, Wappenschmidt B, Meindl A, Kiechle M, Bugert P, Schmutzler RK, Bartram CR, Burwinkel B: Copy number variant in the candidate tumor suppressor gene MTUS1 and familial breast cancer risk. Carcinogenesis (2007), in press. Frank B, Hemminki K, Meindl A, Wappenschmidt B, Sutter C, Kiechle M, Bugert P, Schmutzler RK, Bartram CR, Burwinkel B: BRIP1 (BACH1) variants and familial breast cancer risk: a case-control study. BMC Cancer (2007) 15,
112 Publications Gareus R, Huth M, Breiden B, Nenci A, Rösch N, Haase I, Bloch W, Sandhoff K, Pasparakis M: Normal epidermal differentiation but impaired skin barrier formation in mice with keratinocyte-restricted knockout of IKK1. Nat Cell Biol (2007) 9, Gassanov N, Jankowski M, Danalache B, Wang D, Grygorczyk R, Hoppe UC, Gutkowska J: Arginine vasopressin-mediated cardiac differentiation: new insight into the role of its receptors and nitric oxide signaling. J Biol Chem (2007), in press. Gerharz M, Baranowsky A, Siebolts U*, Eming S, Nischt R, Krieg T, Wickenhauser C: Morphometric analysis of murine skin wound healing: standardization of experimental procedures and impact of an advanced multitissue array technique. Wound Repair Regen (2007) 15, (*MD-PhD Program) Graef M, Seewald G, Langer T: Substrate Recognition by AAA+ ATPases: distinct substrate binding modes in ATPdependent protease Yme1 of the mitochondrial intermembrane space. Mol Cell Biol (2007) 27, Grigat S, Harlfinger S, Pal S, Striebinger R, Golz S, Geerts A, Lazar A, Schomig E, Grundemann D: Probing the substrate specificity of the ergothioneine transporter with methimazole, hercynine, and organic cations. Biochem Pharmacol (2007) 74, Halbach A, Mörgelin M, Baumgarten M, Milbrandt M, Paulsson M, Plomann M: PACSIN 1 forms tetramers via its N-terminal F-BAR domain. FEBS J (2007) 274, Haubert D, Gharib N, Rivero F, Wiegmann K, Hosel M, Kronke M, Kashkar H: PtdIns(4,5)P-restricted plasma membrane localization of FAN is involved in TNF-induced actin reorganization. EMBO J (2007), in press. Heiss WD, Sorensen AG: Advances in imaging Stroke (2007) 38, Herzig S, Khan IF R, Gründemann D, Matthes J, Ludwig A, Michels G, HOPPE UC, Chaudhuri D, Schwartz A, Yue DT, Hullin R: Mechanism of Ca v 1.2 channel modulation by the amino terminus of cardiac β 2 -subunits. FASEB J (2007), in press. Hombach A, Abken H: Costimulation tunes tumor-specific activation of redirected T cells in adoptive immunotherapy. Cancer Immunol Immunother (2007) 56, Hombach A, Schildgen V, Heuser C, Finnern R, Gilham D, Abken H: T cell activation by antibody-like immunoreceptors: the position of the binding epitope within the target molecule determines the efficiency of activation of redirected T cells. J Immunol (2007) 178, Hoppe UC, Freemantle N, Cleland JGF, Marijianowski M, Erdmann E: Effect of cardiac resynchronization on morbidity and mortality of diabetic patients with severe heart failure. Diabetes Care (2007) 30, Hornig-Do HT, von Kleist-Retzow JC, Lanz K, Wickenhauser C, Kudin AP, Kunz WS, Wiesner RJ, Schauen M: Human epidermal keratinocytes accumulate superoxide due to low activity of Mn-SOD, leading to mitochondrial functional impairment. J Invest Dermatol (2007), in press. Hovelmeyer N, Wunderlich FT, Massoumi R, Jakobsen CG, Song J, Worns MA, Merkwirth C, Kovalenko A, Aumailley M, Strand D, Bruning JC, Galle PR, Wallach D, Fassler R, Waisman A. Regulation of B cell homeostasis and activation by the tumor suppressor gene Cyld. J Exp Med (2007). In press. Hubbers CU, Clemen CS, Kesper K, Boddrich A, Hofmann A, Kamarainen O, Tolksdorf K, Stumpf M, Reichelt J, Roth U, Krause S, Watts G, Kimonis V, Wattjes MP, Reimann J, Thal DR, Biermann K, Evert BO, Lochmuller H, Wanker EE, Schoser BG, Noegel AA, Schroder R: Pathological consequences of VCP mutations on human striated muscle. Brain (2007) 130, Hullin R, Matthes J, von Vietinghoff S, Bodi I, Rubio M, D Souza K, Khan IF, Rottländer D, HOPPE UC, Mohacsi P, Schmitteckert E, Gilsbach R, Bünemann M, Hein L, Schwartz A, Herzig S: Increased expression of the auxiliary β2-subunit of ventricular L-type Ca channels leads to single-channel activity characteristic of heart-failure. PLoS ONE (2007) 2, e292. Jacobs AH, Rueger MA, Winkeler A, Li H, Vollmar S, Waerzeggers Y, Rueckriem B, Kummer C, Dittmar C, Klein M, Heneka MT, Herrlinger U, Fraefel C, Graf R, Wienhard K, Heiss WD: Imaging-guided gene therapy of experimental gliomas. Cancer Res (2007) 67, Joussen AM, Smyth N, Niessen C: Pathophysiology of diabetic macular edema. Dev Ophthalmol (2007) 39, Kalay E, Uzumcu A, Krieger E, Çaylan R, Uyguner O, Ulubil-Emiroglu M, Kayserili H, Hafiz G, Başerer N, Heister AJ, Hennies HC, Nürnberg P, Başaran A, Brunner HG, Cremers CW, Karagüzel A, Wollnik B, Kremer H: MYO15A (DFNB3) mutations in Turkish hearing loss families and functional modeling of a novel motor domain mutation. Am J Med Genet (2007), in press. Kashkar H, Deggerich A, Seeger JM, Yazdanpanah B, Wiegmann K, Haubert D, Pongratz C, Kronke M: NF-kB independent down-regulation of XIAP by bortezomib sensitizes HL B-cells against cytotoxic drugs. Blood (2007) 109, Khaire N, Muller R, Blau-Wasser R, Eichinger L, Schleicher M, Rief M, Holak TA, Noegel AA: Filamin-regulated F-actin assembly is essential for morphogenesis and controls phototaxis in Dictyostelium. J Biol Chem (2007) 282,
113 Publications von Kleist-Retzow JC, Hornig-Do HT, Schauen M, Eckertz S, Dong Dinh T, Stassen F, Lottmann N, Bust M, Galunska B, Wielckens K, Hein W, Beuth J, Braun JM, Fischer JH, Ganitkevich VY, Maniura-Weber K, Wiesner RJ: Impaired mitochondrial Ca 2+ homeostasis in respiratory chain deficient cells but efficient compensation of energetic disadvantage by enhanced anaerobic glycolysis due to low ATP steady state levels. Exp Cell Res (2007), in press. Kobayashi N, Kostka G, Garbe JH, Keene DR, Bachinger HP, Hanisch FG, Markova D, Tsuda T, Timpl R, Chu ML, Sasaki T: A comparative analysis of the fibulin protein family: Biochemical characterization, binding interactions and tissue localization. J Biol Chem (2007), in press. Koch A, Raidl M, Lux M, Muller K, Buning H, Humme S, Erdmann E: IL-12-induced T-bet expression and IFNgamma release in lymphocytes from asthmatics-role of MAPkinases ERK-1/-2, p38(mapk) and effect of dexamethasone. Respir Med (2007) 101, Kociok N, Joussen AM: Varied expression of functionally important genes of RPE and choroid in the macula and in the periphery of normal human eyes. Graefes Arch Clin Exp Ophthalmol (2007) 245, Koehler H, Kofler D, Hombach A, Abken H: CD28 costimulation overcomes transforming growth factor-{beta}-mediated repression of proliferation of redirected human CD4 + and CD8 + T cells in an antitumor cell attack. Cancer Res (2007) 67, Koizumi N, Rigby H, Fullwood NJ, Kawasaki S, Tanioka H, Koizumi K, Kociok N, Joussen AM, Kinoshita S: Comparison of intact and denuded amniotic membrane as a substrate for cell-suspension culture of human limbal epithelial cells. Graefes Arch Clin Exp Ophthalmol (2007) 245, Konner AC, Janoschek R, Plum L, Jordan SD, Rother E, Ma X, Xu C, Enriori P, Hampel B, Barsh GS, Kahn CR, Cowley MA, Ashcroft FM, Bruning JC: Insulin Action in AgRP-Expressing Neurons Is Required for Suppression of Hepatic Glucose Production. Cell Metab (2007) 5, Koppen M, Langer T: Protein Degradation within Mitochondria: Versatile Activities of AAA Proteases and Other Peptidases. Crit Rev Biochem Mol Biol (2007) 42, Koppen M, Metodiev MD, Casari G, Rugarli EI, Langer T: Variable and tissue-specific subunit composition of mitochondrial m-aaa protease complexes linked to hereditary spastic paraplegia. Mol Cell Biol (2007) 27, Kornblum C, Zsurka G, Wiesner RJ, Schröder R, Kunz WS: Double kill by two novel pathogenic mitochondrial trna point mutations in chronic progressive external ophthalmoplegia. J Neuropathol Exp Neurol (2007), in press. Kummer C, Winkeler A, Dittmar C, Bauer B, Rueger MA, Rueckriem B, Heneka MT, Vollmar S, Wienhard K, Fraefel C, Heiss WD, Jacobs AH: Multitracer positron emission tomographic imaging of exogenous gene expression mediated by a universal herpes simplex virus 1 amplicon vector. Mol Imaging (2007) 6, Kurschat P, Eming S, Nashan D, Krieg T, Mauch C: Early increase in serum levels of the angiogenesis-inhibitor endostatin and of basic fibroblast growth factor in melanoma patients during disease progression. Br J Dermatol (2007), in press. Lichtenauer DU*, Duchniewicz M, Kolanczyk M, Hoeflich A, Hahner S, Else T, Bicknell AB, Zemojtel T, Stallings NR, Schulte DM, Kamps MP, Hammer GD, Scheele JS, Beuschlein F: Pre-B-cell transcription factor 1 and steroidogenic factor 1 synergistically regulate adrenocortical growth and steroidogenesis. Endocrinology (2007) 148, (*MD-PhD Program) Luke M, Januschowski K, Warga M, Beutel J, Leitritz M, Gelisken F, Grisanti S, Schneider T, Luke C, Bartz-Schmidt KU, Szurman P: The retinal tolerance to bevacizumab in co-application with a recombinant tissue plasminogen activator. Br J Ophthalmol (2007), in press. Lüke M, Szurman P, Schneider T, Lüke C: The effects of the phosphodiesterase type V inhibitor sildenafil on human and bovine retinal function in vitro. Graefes Arch Clin Exp Ophthalmol (2007), in press. Ma X, Bruning J, Ashcroft F. (2007) Glucagon-like peptide 1 stimulates hypothalamic proopio-melanocortin neurons. J Neurosci (2007) 27(27): Ma X, Zubcevic L, Bruning JC, Ashcroft FM, Burdakov D. Electrical inhibition of identified anorexigenic POMC neurons by orexin/hypocretin. J Neurosci (2007) 27, Maaijwee KJ, van Meurs JC, Kirchhof B, Mooij CM, Fischer JH, Mackiewicz J, Kobuch K, Joussen AM: Histological evidence for revascularisation of an autologous retinal pigment epithelium-choroid graft in the pig. Br J Ophthalmol (2007) 91, Mackiewicz J, Muhling B, Hiebl W, Meinert H, Maaijwee K, Kociok N, Luke C, Zagorski Z, Kirchhof B, Joussen AM: In vivo retinal tolerance of various heavy silicone oils. Invest Ophthalmol Vis Sci (2007) 48, Maertens B, Hopkins D, Franzke CW, Keene DR, Bruckner-Tuderman L, Greenspan DS, Koch M: Cleavage and oligomerization of gliomedin, a transmembrane collagen required for node of Ranvier formation. J Biol Chem (2007) 282, Mann HH, Sengle G, Gebauer JM, Eble JA, Paulsson M, Wagener R: Matrilins mediate weak cell attachment without promoting focal adhesion formation. Matrix Biol (2007) 26,
114 Publications Miletic H*, Fischer YH, Litwak S, Giroglou T, Waerzeggers Y, Winkeler S, Li H, Himmelreich U, Stenzel W, Deckert M, Lange C, Neumann H, Jacobs AH, von Laer D: Bystander Killing of Malignant Glioma by Bone Marrow-Derived Tumor Infiltrating Progenitor Cells Expressing a Suicide Gene. Mol Ther (2007) 15, (*MD-PhD Program) Menotta M, Amicucci A, Basili G, Rivero F, Polidori E, Sisti D, Stocchi V: Molecular characterisation of the small GT- Pase CDC42 in the ectomycorrhizal fungus Tuber borchii Vittad. Protoplasma (2007), in press. Meyer PT*, Elmenhorst D, Boy C, Winz O, Matusch A, Zilles K, Bauer A: Effect of aging on cerebral A1 adenosine receptors: a [18F]CPFPX PET study in humans. Neurobiol Aging (2007), in press. (*MD-PhD Program) Moser N, Mechawar N, Jones I, Gochberg-Sarver A, Orr- Urtreger A, Plomann M, Salas R, Molles B, Marubio L, Roth U, Maskos U, Winzer-Serhan U, Bourgeois JP, Le Sourd AM, De Biasi M, Schroder H, Lindstrom J, Maelicke A, Changeux JP, Wevers A: Evaluating the suitability of nicotinic acetylcholine receptor antibodies for standard immunodetection procedures. J Neurochem (2007), in press. Netzer C, Freudenberg J, Toliat MR, Heinze A, Heinze- Kuhn K, Thiele H, Goebel I, Nürnberg P, Ptáček LJ, Göbel H, Todt U, Kubisch C. Genetic association studies of the chromosome 15 GABA-A receptor cluster in migraine with aura. Am J Med Genet Part B (2007), in press. Neulen A, Blaudeck N, Zittrich S, Metzler D, Pfitzer G, Stehle R: Mn 2+ -dependent protein phosphatase 1 enhances protein kinase A-induced Ca 2+ desensitisation in skinned murine myocardium. Cardiovasc Res (2007) 74, Niemann C, Owens DM, Schettina P, Watt FM: Dual role of inactivating Lef1 mutations in epidermis: tumour promotion and specification of tumour type. Cancer Res (2007) 67, Niessen CM: Tight junctions/adherens junctions: basic structure and function. J Invest Derm (2007), in press. Oberthuer A, Warnat P, Kahlert Y, Westermann F, Spitz R, Brors B, Hero B, Eils R, Schwab M, Berthold F, Fischer M: Classification of neuroblastoma patients by published gene-expression markers reveals a low sensitivity for unfavorable courses of MYCN non-amplified disease. Cancer Lett (2007) 250, Opel D, Poremba C, Simon T, Debatin KM, Fulda S: Activation of akt predicts poor outcome in neuroblastoma. Cancer Res (2007) 67, Osman C, Wilmes C, Tatsuta T, Langer T: Prohibitins interact genetically with Atp23, a novel processing peptidase and chaperone for the F1Fo-ATP synthase. Mol Biol Cell (2007) 18, Palka-Santini M, Putzfeld S, Cleven BE, Kronke M, Krut O: Rapid identification, virulence analysis and resistance profiling of Staphylococcus aureus by gene segment-based DNA microarrays: application to blood culture postprocessing. J Microbiol Methods (2007) 68, Pasutto F, Sticht H, Hammersen G, Gillessen-Kaesbach G, Fitzpatrick DR, Nürnberg G, Brasch F, Schirmer- Zimmermann H, Tolmie JL, Chitayat D, Houge G, Fernandez-Martinez L, Keating S, Mortier G, Hennekam RC, Wense A von der, Slavotinek A, Meinecke P, Bitoun P, Becker C, Nürnberg P, Reis A, Rauch A: Mutations in STRA6 cause a broad spectrum of malformations including anophthalmia, congenital heart defects, diaphragmatic hernia, alveolar capillary dysplasia, lung hypoplasia, and mental retardation. Am J Hum Genet (2007) 80, Pivarcsi A, Müller A, Rieker J, Steinhoff M, Kubitza R, Meller S, Sonkoly E, van Lierop A, Hoffmann TK, Schneider L, Piekorz R, Enderlein E, Rohr U-P, Haas R, Haase I, Nürnberg B, Ruzicka T, Zlotnik A, Homey B: Tumor immune escape by the loss of homeostatic chemokine expression. J Exp Med (2007), in press. Prabhu Y, Muller R, Anjard C, Noegel AA: GrlJ, a Dictyostelium GABAB-like receptor with roles in post-aggregation development. BMC Dev Biol (2007) 7, 4. Reimers N, Kasper HU, Weissenborn SJ, Stutzer H, Preuss SF, Hoffmann TK, Speel EJ, Dienes HP, Pfister HJ, Guntinas- Lichius O, Klussmann JP: Combined analysis of HPV-DNA, p16 and EGFR expression to predict prognosis in oropharyngeal cancer. Int J Cancer (2007) 120, Rhiem K, Flucke U, Engel C, Wappenschmidt B, Reinecke- Luthge A, Buttner R, Schmutzler RK: Association of the BRCA1 missense variant R1699W with a malignant phyllodes tumor of the breast. Cancer Genet Cytogenet (2007) 176, Roach W, Plomann M: PACSIN3 overexpression increases adipocyte glucose transport through GLUT1. Biochem Biophys Res Commun (2007) 355, Rosentreter A, Hofmann A, Xavier CP, Stumpf M, Noegel AA, Clemen CS: Coronin 3 involvement in F-actin-dependent processes at the cell cortex. Exp Cell Res (2007) 313, Rottlaender D, Matthes J, Vatner SF, Seifert R, Herzig S: Functional adenylyl cyclase inhibition in murine cardiomyocytes by 2 (3 )-O-(N-methylanthraniloyl)- guanosine 5 -[{gamma}-thio]triphosphate. J Pharmacol Exp Ther (2007), in press. Rudnik-Schoneborn S, Botzenhart E, Eggermann T, Senderek J, Schoser BG, Schroder R, Wehnert M, Wirth B, Zerres K: Mutations of the LMNA gene can mimic autosomal 109
115 Publications dominant proximal spinal muscular atrophy. Neurogenetics (2007) 13, Rueger MA*, Kracht LW, Hilker R, Thiel A, Sobesky J, Winkeler A, Thomas AV, Heneka MT, Graf R, Herholz K, Heiss WD, Jacobs AH: Role of in vivo imaging of the central nervous system for developing novel drugs. Q J Nucl Med Mol Imaging (2007) 51, (*MD-PhD Program) Ruhparwar A, Er F, Martin U, Radke K, Gruh I, Niehaus M, Karck M, Haverich A, HOPPE UC: Enrichment of cardiac pacemaker-like cells: Neuregulin-1 and cyclic AMP increase If-current density and Connexin 40 mrna levels in fetal cardiomyocytes. Med Biol Eng Comput (2007) 45, Sardy M, Csikos M, Geisen C, Preisz K, Kornsee Z, Tomsits E, Tox U, Hunzelmann N, Wieslander J, Karpati S, Paulsson M, Smyth N: Tissue transglutaminase ELISA positivity in autoimmune disease independent of gluten-sensitive disease. Clin Chim Acta (2007) 376, Saydam O, Saydam N, Glauser DL, Pruschy M, Dinh-Van V, Hilbe M, Jacobs AH, Ackermann M, Fraefel C: HSV-1 amplicon-mediated post-transcriptional inhibition of Rad51 sensitizes human glioma cells to ionizing radiation. Gene Ther (2007), in press. Schnaith A, Kashkar H, Leggio SA, Addicks K, Kronke M, Krut O: Staphylococcus aureus subvert autophagy for induction of caspase-independent host cell death. J Biol Chem (2007) 282, Schwientek T, Mandel U, Roth U, Müller S, Hanisch FG: A serial lectin approach to the mucin-type O-glycoproteome of Drosophila melanogaster S2 cells. Proteomics (2007), in press. Seibler J, Kleinridders A, Kuter-Luks B, Niehaves S, Bruning JC, Schwenk F: Reversible gene knockdown in mice using a tight, inducible shrna expression system. Nucleic Acids Res (2007) 35, e54. Siebzehnrubl FA, Buslei R, Eyupoglu IY, Seufert S, Hahnen E, Blumcke I: Histone deacetylase inhibitors increase neuronal differentiation in adult forebrain precursor cells. Exp Brain Res (2007) 176, Simon T, Langler A, Berthold F, Klingebiel T, Hero B: Topotecan and etoposide in the treatment of relapsed highrisk neuroblastoma: Results of a phase 2 trial. J Pediatr Hematol Oncol (2007) 29, Stehle R, Iorga B, Pfitzer G: Calcium regulation of troponin and its role in the dynamics of contraction and relaxation. Am J Physiol Regul Integr Comp Physiol (2007) 292, Stoffel W, Jenke B, Holz B, Binczek E, Günter RH, Knifka J, Koebke J, Niehoff A: Targeted deletion of the neutral sphingomyelinase (SMPD3) gene causes a novel form of chondrodysplasia. American J Path (2007), in press. Tatsuta T, Augustin S, Nolden M, Friedrichs B, Langer T: m-aaa protease-driven membrane dislocation allows intramembrane cleavage by rhomboid in mitochondria. EMBO J (2007) 26, Tchatchou S, Wirtenberger M, Hemminki K, Sutter C, Meindl A, Wappenschmidt B, Kiechle M, Bugert P, Schmutzler RK, Bartram CR, Burwinkel B: Aurora kinases A and B and familial breast cancer risk. Cancer Lett (2007) 247, Timmann C, Evans JA, Konig IR, Kleensang A, Ruschendorf F, Lenzen J, Sievertsen J, Becker C, Enuameh Y, Kwakye KO, Opoku E, Browne EN, Ziegler A, Nürnberg P, Horstmann RD: Genome-Wide Linkage Analysis of Malaria Infection Intensity and Mild Disease. PLoS Genet (2007) 3, e48. Tscharnke M, Pofahl R, Chrostek A, Smyth N, Niessen CM, Niemann C, Hartwig B, Herzog V, Klein HW, Krieg T, Brakebusch C, Haase I: Impaired epidermal wound healing in vivo upon inhibition or deletion of Rac1. J Cell Science (2007) 120, Uyguner O, Kayserili H, Li Y, Karaman B, Nurnberg G, Hennies H, Becker C, Nurnberg P, Basaran S, Apak M, Wollnik B: A new locus for autosomal recessive nonsyndromic mental retardation maps to 1p21.1-p13.3. Clin Genet (2007) 71, Vaclavicek A, Bermejo JL, Wappenschmidt B, Meindl A, Sutter C, Schmutzler RK, Kiechle M, Bugert P, Burwinkel B, Bartram CR, Hemminki K, Forsti A: Genetic variation in the major mitotic checkpoint genes does not affect familial breast cancer risk. Breast Cancer Res Treat (2007), in press. Versmold B, Felsberg J, Mikeska T, Ehrentraut D, Kohler J, Hampl JA, Rohn G, Niederacher D, Betz B, Hellmich M, Pietsch T, Schmutzler RK, Waha A: Epigenetic silencing of the candidate tumor suppressor gene PROX1 in sporadic breast cancer. Int J Cancer (2007) 121, Waisman A, Kraus M, Ghosh S, Seagal J, Melamed D, Song J*, Sasaki Y, Classen S, Lutz C, Brombacher F, Nitschke L, Rajewsky K: The IgG1 B cell receptor signaling is inhibited by CD22 and promotes the development of B cells whose survival is less dependent on Igalpha/beta. JEM (2007), in press. (*MD-PhD Program) Weiergräber M*, Henry M, Radhakrishnan K*, Hescheler J, Schneider T: Hippocampal seizure resistance and reduced neuronal excitotoxicity in mice lacking the Ca v
116 Publications E/R-type voltage-gated calcium channel. J Neurophysiology (2007) 97, (*MD-PhD Program) Winkeler A, Sena-Esteves M, Paulis LE, Li H, Waerzeggers Y, Ruckriem B, Himmelreich U, Klein M, Monfared P, Rueger MA, Heneka M, Vollmar S, Hoehn M, Fraefel C, Graf R, Wienhard K, Heiss WD, Jacobs AH: Switching on the lights for gene therapy. PLoS ONE (2007) 2, e528. Wirtenberger M, Schmutzhard J, Hemminki K, Meindl A, Sutter C, Schmutzler RK, Wappenschmidt B, Kiechle M, Arnold N, Weber BH, Niederacher D, Bartram CR, Burwinkel B: The functional genetic variant Ile646Val located in the kinase binding domain of the A-kinase anchoring protein 10 is associated with familial breast cancer. Carcinogenesis (2007) 28, Zhang Q, Bengtsson M, Partridge C, Salehi A, Braun M, Eliasson L, Johnson PRV, Renström E, Schneider T, Berggren PO, Göpel S, Ashcroft FM, Rorsman P: R-type Ca 2+ -channel-evoked CICR regulates glucose-induced somatostatin secretion. Nature Cell Biol (2007), in press. Zobel C, Rana OR, Saygili E, Bolck B, Diedrichs H, Reuter H, Frank K, Muller-Ehmsen J, Pfitzer G, Schwinger RH: Mechanisms of Ca 2+ -dependent calcineurin activation in mechanical stretch-induced hypertrophy. Cardiology (2007) 107, Ziskoven C, Grafweg S, Bolck B, Wiesner RJ, Jimenez M, Giacobino JP, Bloch W, Schwinger RH, Brixius K: Increased Ca 2+ sensitivity and protein expression of SERCA 2a in situations of chronic beta(3)-adrenoceptor deficiency. Pflugers Arch (2007) 453, Patents Overview of technological transfer activities generated during the time interval January 2005 March Liaison-Group Frank Berthold (Department of Pediatric Oncology and Hematology) Patent title: Tumorbox for Simultaneous Shipping of Frozen and Unfrozen Material Authors: Frank Berthold, Birgit Kleinhenz, Karen Ernestus, Markus Jensen Date of filing: January 2005 By: German Patent and Trade Mark Office C 10 Brunhilde Wirth / Eric Hahnen (Institute for Human Genetics) Patent title: Means and Methods for the Treatment of Head Injuries and Stroke Authors: Ingmar Blümcke, Eric Hahnen, Florian A. Siebzehnrübl, Rolf Buslei, Ilker Y. Eyüpoglu Date of filing: April 2006 Date of registration: November 2006 By: Patent Cooperation Treaty 111
117 Career Development Habilitations / Doctorates / Diploma Thesis Overview of completed and on-going Habilitations and Dissertations of the year 2005 until May Habilitations Bolz, Hanno C 10 Die molekulare Genetik sensorischer Erkrankungen: Schwerhörigkeit, Retinitis pigmentosa und Usher Syndrom; completed in Dec Büning, Hildegard B 10 Development of viral vectors based on the adenoassociated virus for human gene therapy; since April Fauser, Sascha A 4 Genetic approaches to retinal disease; completed in Oct Fischer, Matthias Liaison-Group Novel insights into the pathogenesis and improved outcome prediction of neuroblastoma by gene expression profiling analyses; since June Haase, Ingo B 5 The pathogenic potential of signals regulating biological repair and defence mechanisms in epidermal keratinocytes; completed in Dec Hacker, Ulrich B 10 Entwicklung viraler Vektoren auf der Basis des rekombinanten Adeno-assoziierten Virus (AAV) zur Gentherapie kolorektaler Karzinome; completed in Oct Hahnen, Eric C 10 Epigenetik und epigenetische Therapieoptionen zur Behandlung zentralnervöser Erkrankungen; since March Kashkar, Hamid B 8 Apoptosis resistance in Hodgkin Lymphoma and therapeutic approaches; since Aug von Kleist-Retzow, Jürgen-Christoph C 9 Cytochrome c Oxidase Defekte der mitochondrialen Atmungskette im Kindesalter; completed in April Kociok, Norbert A 4 Retinal Angiogenesis; since Oct Kurschat, Peter B 1 Mechanismen der Tumorprogression am Beispiel des malignen Melanoms - Untersuchungen zur Bedeutung von Metalloproteinasen und Angiogenese; completed in Nov Matthes, Jan A 5 Disease relevance of cardiovascular calcium channels; since Jan Netzer, Christian C 11 Genetic studies in hereditary forms of neurological disorders; since July Plomann, Markus C 7 Regulation of vesicular transport: Role of PACSIN proteins and their interaction partners; since Jan Rosenkranz, Stephan A 6 Signalmechanismen von PDGF-Rezeptoren im Rahmen kardiovaskulärer Erkrankungen; completed in April Schubert, Markus C 1 Role of insulin receptor signaling in the central nervous system; since Jan Semkova, Irina A 4 Pathogenesis of age-related macular degeneration; since Aug Simon, Thorsten Liaison-Group Critical evaluation of clinical and molecular risk factors in neuroblastoma; completed in April Solzin, Johannes A 8 Molecular mechanism of troponin in regulation of cardiac and skeletal contraction and relaxation; since July Todt, Unda C 11 The genetics of migraine with aura; since Jan Weiergräber, Marco C 4 Funktion neuronaler spannungsgesteuerter Ca 2+ -Kanäle bei der Umwandlung normaler zu pathophysiologischen Hirnrhythmen; since April Wollnik, Bernd C 10 Molecular and pathophysiological mechanisms underlying different forms of cardiac arrhythmias with and without associated structural heart disease; since May PhD-Theses Alef, Thomas B 11 Ichthyose-Syndrome; since July Augustin, Steffen C 6 Charakterisierung des mitochondrialen Peptidexports; since Aug Bach, Markus A 7 UST3 & OAT5 from human; since July
118 Career Development Behnke, Kerstin A 5 Effekt und Subtyp-Selektivität von Lachgas auf die Einzelkanal-Eigenschaften rekombinanter T-Typ Calciumkanäle; completed in March Behrendt, Kristina B 5 Rac1 in Wundheilung und Tumorentstehung der Haut; since March Berger, Marcus CBA Identifizierung biologisch aktiver Peptide und Proteine in den Sekreten von Lucilia sericata im Wundheilungsgeschehen; completed in July Berthold, Jessica B 7 Rho GTPasen der RhoBTB Familie: Charakterisierung und Rolle in der Tumorentstehung; completed in May Bilkovski, Roman C 1 Molekulare Charakterisierung der Determinierung humaner mesenchymaler Stammzellen in Präadipozyten; since Nov Billion, Karla NG I Proteolytic cleavage of cadherins: Functional role of the cleaved extracellular and cytoplasmic domains; completed in April Blume, Jessica C 7 Funktionale Analyse der EHD-Proteine bei tubulären Transportprozessen; completed in July Bohne, Felix NG-F Spezifische Elimination Hepatitis B Virus infizierter Hepatozyten durch modifizierte humane T-Zellen die einen chimären T-Zellrezeptor exprimieren und eine zytotoxische Immunantwort etablieren; completed in Nov Böll, Boris Expression regulatorischer Oberflächenmoleküle von NK-Zellen sowie die Bestimmung des cytotoxischen Potentials und die Zytokinsekretion nach Inhibition oder Aktivierung des CD30 Signalwegs; since Sept (MD PhD-Thesis) Bonn, Florian C 6 Proteolytic control of ribosomal biogenesis in mitochondria; since July Böse, Kerstin B 2 Funktionelle Charakterisierung von Nidogen anhand gewebespezifischer Entwicklungsvorgänge; completed in Nov Boucas, Jorge Miguel Martins B 10 Single chain antibodies fused to the N -terminus of VP2 for directing cell-specific AAV-mediated gene transfer into colorectal cancer; since Aug Brandt-Bohne, Ulrike C 8 Untersuchung zur Rolle von EGFL5 und DNER bei neuronalen Entwicklungsprozessen; completed in Jan Breitenkamp, Alexandra A 5 Populationsgenetische Untersuchungen zur Rolle von Calciumkanaluntereinheiten bei Autismus; since Sept Brichta, Lars C 10 Molecular genetic investigations of histone deacetylase inhibitors as potential neurotherapeutics for autosomal recessive proximal spinal muscular atrophy (SMA); completed in Oct Chaurasia, Bhagirath A 1 Role of PDK1 in Macrophage Function; since April Cherukupalli, Anil A 1 Role of IL-6 in obesity-induced insulin resistance; since April Chmielewski, Markus B 4 Murine Immunrezeptoren in Maus-Tumor Modellen; completed in June Chung, Boidinh C 11 Cloning and characterization of the causative gene for a novel MCA/MR syndrome; since July Dargazanli, Sascha C 6 Regulation of apoptosis by prohibitins in mitochondria; since Aug Deggerich, Anke B 12 Analysis of TIP47 function in the biogenesis and metabolism of lipid droplets; since Feb Dizayee, Sara A 5 Regulation des kardialen Calciumkanals durch das inhibitorische G-Protein G αi3 ; since Jan Ebermann, Inga C 10 Postitionsklonierung und funktionelle Charakterisierung neuer Gene für syndromale und nicht-syndromale Taubheit; since Oct Ebert, Gregor NG-F Hepatitis B Virus-basierende Vektoren; since Oct Eckertz, Sabrina C 9 Molecular pathomechanisms of a skeletal-muscle-specific lethal mutation of mitochondrial DNA in a muscle specific cybrid model; since April Eckl, Katja B 11 Kongenitale Ichthyosen; completed in July
119 Career Development Ehses, Sarah C 6 Functional analysis of the mammalian m-aaa protease; since Nov El Allaki, Zin-Eddin B 6 Functional characterisation of the CYLD protein; since Feb Elhamine, Fatiha Contractile and proteomic changes of the myocard at tetralogy of Fallot, a congenital heart defect; since May (MD PhD-Thesis) Endell, Jan B 10 Generation of enhanced gene delivery vectors by directed evolution of adeno-associated virus; completed in May Engmann, Tanja C 6 Analyzing of substrates of the yeast i-aaa protease Yme1; since Sept Ernst, Marianne A 1 Role of FOXO1 in skin homeostasis; since Nov (MD PhD-Thesis) Esser, Knud NG-F Visualization of hepatitis B virus infection; since July (MD PhD-Thesis) Fabisch, Christian A 5 Calciumkanalexpression bei genetischen Modellen der murinen diabetischen Kardiomyopathie; since Oct Fein, Nadin B 4 Generierung transgener Mäuse mit rekombinanten Immunrezeptoren; since March Fork, Christian A 7 SLC22A13 from human; since July Franko, Andras C 9 The role of AMPK and CREB-1 in the regulation of mitochondrial biogenesis during muscle differentiation; since Aug Fres, Julia NG VI Lipidmodifikation von humanem Guanylat-bindenden Proteinen; since March Garbes, Lutz C 10 Spinale Muskelatrophie: Auf dem Wege zum besseren Verständnis der individuellen Reaktion auf Histon- Deacetylase Inhibitoren; since Oct Gardemann, Thomas B 4 IL-12 erhöht die lytische Aktivität der Immunrezeptor vermittelten T-Zell Aktivierung; completed in March Goldnau, Daniela B 10 New strategies for the application of Adeno-Associated Virus type 2 targeting vectors; completed in May Graef, Martin C 6 Mechanismen der Substraterkennung der mitochondrialen AAA-Protease Yme1 aus Saccharomyces cerevisiae; completed in May Grigat, Silke A 7 OCNT2 from human; since Jan Günter, Robert C 3 Characterisation of the glial glutamate transporter GlT1; completed in Jan Haehn, Sebastian B 2 Funktionelle und strukturelle Aspekte bei Selbstinteraktionen der N-terminalen Domänen von Lamininen; completed in Dec Hamels, Ina C 3 Generation and characterization of the serine-plamitoyl- CoA transferase null allelic mouse; since Oct Harlfinger, Stephanie A 7 Identifizierung des physiologischen Substrates und funktionelle Charakterisierung des organischen Kationentransporters OCTN1 mittels LC-MS/MS; completed in Sept Hauke, Jan C 10 Molekulare Analyse der epigenetischen Regulation des survival of motor neuron 2 (SMN2) Gens und der Stabilität des SMN2 Proteins; since April Heimbach, André C 11 Functional analyses of the human ATP13A2 gene; since March Henkes, Liliane A 7 Klonierung und funktionelle Charakterisierung des Extraneuronalen Monoamin-Transporters (EMT) des Hausschweins; completed in July Hettich, Moritz C 1 Einfluss neuronaler Insulin-like growth factor-1 Signaltransduktion auf die Entstehung des Morbus Alzheimer in vivo; since Dec (MD PhD-Thesis) Ho, Matthew B 2 Neurological phenotypes in nidogen knockout mice; since April Hübbers, Christian A 2 In vitro und in vivo Studien zur Analyse von VCP-Mutanten, die die Multisystemerkrankung IBMPFD verursachen; since Nov
120 Career Development Huber, Anke B 10 Generation of B-CLL specific Adeno-associated virus vectors; since Oct Jakubik, Miriam C 10 Functional analysis of the Bdp1 protein by generating conditional knockout mice; since April Janoschek, Ruth A 1 Role of POMC-expressing neurons in the regulation of energy homeostasis; since Oct Jansangthong, Wanchana A 5 Calciumkanal-Hilfsuntereinheiten und ihre Veränderung im Herzmuskel; since May (MD PhD-Thesis) Jordan, Sabine A 1 Modulation of hepatic insulin action by micrornas; since Dec Kakanj, Parisa NG IV Hedgehog signalling in skin tumourigenesis; since Feb Kelter, Arndt-René C 10 Klonierung und funktionelle Analyse des transcription factor-like nuclear regulator (TFNR), ein essentieller Transkriptionsfaktor aller RNS-Polymerase III-abhängiger Gene und potentielles Kandidatengen für die atypische spinale Muskelatrophie; completed in Feb Keusekotten, Kirstin NG VI Regulation der Bildung von nukleären PML-Körpern; since Nov Kiss, Christine C 3 Generation of a conditional knock out of the glutamate transporter GLT1 (EAAT2) and the astrocyte inducible transgenic Cre mouse; since Jan Klein-Gonzales, Nela Migration der CD40 aktivierten B-Zelle; since Nov (MD PhD-Thesis) Kleinridders, André A 1 PLRG1 regulates mitosis and apoptosis; since July Knolle, Sandra Role of caldesmon in the regulation of smooth muscle actomyosin interaction; since May (MD PhD-Thesis) Koç-Schmitz, Yeliz C 7 Charkterisierung der PACSIN/PICK1/GluR-Komplexe in gesunden und kranken Neuronen; since Oct Koch, Linda A 1 Analysis of IGF-1-receptor action in the central nervous system by conditional mutagenesis in mice, since April Köhler, Heike B 4 Impact of TGF-b on immunoreceptor triggered T cell activation; since June Könner, Christine A 1 Role of PI3-kinase signaling in AgRP-neurons; since Dec (MD PhD-Thesis) Kopacek, Anke Liaison-Group Herstellung, Expression und funktionelle Analyse rekombinater bispezifischer CD3x NCAM Antikörper für die Therapie von Patienten mit rezidiviertem Neuroblastom; since Jan (MD PhD-Thesis) Kopecky, Caroline B 4 Rekombinante Immunrezeptoren als molekulare Schaltersysteme; since Jan Koppen, Mirko C 6 Assembly and function of mammalian m-aaa proteases upon reconstitution in yeast; since Sept Koula, Mina C 3 Characterization of the S-acylation deficient proteolipid protein mouse model and it role in myelin membrane compaction; since Feb Kübbeler, Marc A 4 Junction proteins in diabetic retinopathy; since March Kunze, Alexander B 2 Basement membrane changes leading to retinopathy; since Nov Lichtenauer, Urs Adulte Stammzellen der Nebenniere; since July (MD PhD-Thesis) Liebig, Tanja In vivo Evaluation CD40-aktivierter B Zell basierter Immuntherapie; since Dec (MD PhD-Thesis) Löffek, Stefanie B 1 Die Regulation und Aktivierung von Matrix Metalloproetinasen durch Interaktionen von humanen Melanomzellen mit dermalen Fibroblasten und mikrovaskulären Endothelzellen; completed in Feb Lopez-Davila, Alfredo Jesus A 8 Effect of myosin-crossbridges on the Ca 2+ -controlled switch kinetics of Troponin; since March Lu, Wenshu A 2 SUN proteins in higher eukaryotes, structural bridges connecting the nuclear interior with the cytoskeleton; since April
121 Career Development Lüke, Yvonne A 2 A novel lamin A/C S143F mutation causing Hutchinson- Gilford progeria syndrome and myopathy in humans, is associated with structural changes of the nuclear envelope; since April Lux, Kerstin B 10 Visualization of the intracellular trafficking of incoming Adeno-Associated Virus type 2; completed in May Maertens, Barbara C 8 Gliomedin: ein neues transmembranes Kollagen an den Ranvier schen Schnuerringen; completed in March Majczak, Joanna C 6 Functional analysis of the mammalian i-aaa protease; since Apr Marcuzzi, Gian Paolo B 3 Analyse des onkogenen Potentials der frühen Gene E6 und E7 des humanen Papillomvirus Typ 8 durch die Etablierung transgener Mausmodelle; completed in Dec Märsch, Stephan B 10 Optimization of AAV vectors for human gene therapy by in vitro evolution; since Sept Mauer, Jan A 1 Insulin regulates innate immune responses; since Dec Mauler, Daniela A 4 Integration of endothelial precursor cells into the diabetic retina; since April Mende, Ylva C 10 Htra2-beta1 a splicing regulator of full-length SMN2 transcripts: functional studies in transgenic and knock out mice; since Jan Merkwirth, Carsten C 6 Functional analysis of murine prohibitins; since Oct Mesaros, Andrea A 1 Role of coactivator-associated methyltransferase (CARM)- 1 in energy homeostasis; since April Metodiev, Metodi C 6 Role of prohibitins for proteolysis in yeast and murine mitochondria; completed in Nov Meyer, Philipp In vivo Quantifizierung zerebraler A 1 -Adenosin- Rezeptoren mittels [ 18 F]CPFPX u. der Positronen- Emissions-Tomographie: Quantifizierungsmodelle und erste Untersuchungen physiologischer u. pathologischer Prozesse; completed in March (MD PhD-Thesis) Michels, Christian NG I Cadherin mediated regulation of epidermal barrier function; since Oct Milbrandt, Mark C 7 Analyse zellulärer Transportprozesse nach PACSIN Inaktivierung; completed in Jan Miletic, Hrvoje Gentherapie von malignen Gliomen mit lentiviralen Vektoren; since July (MD PhD-Thesis) Miteva, Maria Rolle der SUMO-Modifikation in der zellulären Antwort auf DNS-Schäden; since Jan (MD PhD-Thesis) Monfared, Parisa C 5 Imaging transcriptional regulation in vivo; completed in March Muniyappa, Nagesh NG I Structural and functional regulation of cell-cell junctions in the diabetic retina: the role of insulin and IGF-1-receptors; since Jan Neumann, Sascha A 2 Alpha-N-Catenin a novel binding partner of Nesprin-2; since Jan Nolden, Mark C 6 Funktion der m-aaa Protease bei der Regulation mitochondrialer Translation; completed in Oct Nussbaum, Tanja Funktionelle Aspekte der IGF-II Inhibierung im humanen hepatozellulären Karzinom unter Berücksichtigung möglicher therapeutischer Ansätze, completed in July (MD PhD-Thesis) Oprea, Gabriela C 10 T-plastin, a cytoskeletal protein with important function in axonal growth acts as a modifier of spinal muscular atrophy; since Jan Osman, Christof C 6 Characterization of prohibitin function in mitochondria; since March Pawlik, Barbara NG V Molecular mechanisms of congenital heart diseases; since Dec (MD PhD-Thesis) Peche, Vivek A 2 Characterisation of CAP2, a dual compartment protein; since Jan Petersson, Monika NG IV Regulation of morphogenesis and tumourigenesis in the skin; since Aug (MD PhD-Thesis) 116
122 Career Development Pfefferle, Regina B 3 Charakterisierung des onkogenen Potentials von HPV8 E2 in transgenen Mäusen und Analyse der Matrix- Metalloproteinase Expression in HPV8 induzierten Hauttumoren; since Jan Quasdorff, Maria NG-F Die Bedeutung von Wirtsfaktoren für die Hepatitis B und Hepatitis B Virus-assoziierte hepatozelluläre Karzinome und ihre therapeutische Beeinflussung; since Oct (MD PhD-Thesis) Radhakrishnan, Kayalvizhi C 4 Interaction partners of the E-/R-type volte gated calcium channels which are involved in the phorbolester mediated stimulation of Ca v 2.3c calcium channel; since May (MD PhD-Thesis) Raschke, Ines C 6 Tissue-specific functions of AAA proteases in mice; since Jan Redemann, Nora A 1 Regulation of energy homeostasis via micrornas expressed in hypothalamus; since Oct Riessland, Markus C 10 In vivo characterization of new drugs for SMA therapy using a SMA mouse model; since Oct Riet, Tobias B 4 Interaktionen rekombinanter Immunrezeptoren; since April Rohmann, Edyta NG V Identification of the molecular pathogenesis of LADD syndrome; since July (MD PhD-Thesis) Rother, Eva A 1 Neuroendocrine control of energy balance; since Nov (MD PhD-Thesis) Rueger, Adele C 5 Activity and survival of dopamine neurons derived from embryonic stem cells; since July (MD PhD-Thesis) Rybakin, Vasily A 2 Characterization of Crn7, a novel mammalian Golgi protein; completed in Dec Sahin, Selen NG V Identification and functional analysis of novel genes underlying arrhythmogenic right ventricular cardiomyopathy; since Feb (under a PhD program from Istanbul University, Istanbul, Turkey) Schael, Sylvia C 7 AMPA-Rezeptor Regulation durch PACSIN 1 unter physiologischen und pathophysiologischen Bedingungen; since March Schenková, Kristína B 7 Characterization of RhoBTB binding partners; since Nov Schievenbusch, Stephanie Interaction of epithelial and mesenchymal cells in renal and hepatic fibrogenesis; since Feb (MD PhD- Thesis) Schlemminger, Stephanie B 10 Intracellular anti-aav defence mechanisms using hepatocytes as model; since Dec Schmidt, Patrick B 4 Anti-Melanom Immunrezeptoren in Maus Modellen; since April Schnaith, Annabelle Identification and characterization of cytotxic factors produced by Staphylococcus aureus; completed in March (MD PhD-Thesis) Schneider, Maria A 2 Charakterisierung von Nesprinen als Bindeglieder des Aktin- und Mikrotubuli-Zytoskelett der Kernmembran; since May Schneiders, Fiona C 8 Identifikation neuer Bindungspartner von Netrin-4 in der extrazellulären Matrix; completed in July Schnickmann, Stephanie Bedeutung des HGF-MET Signalsystems im Rahmen der epidermalen Wundheilung, completed in July (MD PhD-Thesis) Schoenen, Frank C 10 Molekularbiologische Analyse des BDP1, einer essentiellen Komponente des Polymerase-III- Transkriptionskomplexes und ZNF297B, seines neu identifizierten Interaktionspartners; completed in March Schuhmann, Natascha B 10 raav mediated gene transfer into pig organs for immune modulation of xenotransplants; since April Schulte, Carsten B 6 Regulation of keratinocyte migration Role of collagen XVII, Cyld and Kindlin-1; completed in Feb Schulze, Jörg C 8 Untersuchungen neuer Kollagene am Muskel-Sehnen Ansatz; completed in Jan Seeger, Jens-Michael B 8 XIAP-mediated chemoresistance and the role of mitochondria; since April
123 Career Development Seifert, Wenke B 11 Charakterisierung des Cohen-Syndrom-Gens VPS13B; since April Sequaris, Gilles NG IV Functional analysis of Hedgehog signalling during sebocyte proliferation and differentiation; since Oct Shekar, Sunil A 2 Characterisation of cyclase associated protein homologue, CAP2; completed in Feb Shina, Maria-Christina A 2 Die Rolle von Coronin 7; since Oct Siebolts, Udo Die Bedeutung gemeinsamer Transduktionsfaktoren bei der Signalübermittlung für maligne Transformation von Vorläuferzellen im Rahmen der polyzythemia vera; since July (MD PhD-Thesis) Skowron, Matthias Liaison-Group Gene expression analysis of spontaneously regressing and aggressive neuroblastoma identifies IGSF4 as a candidate tumor suppressor gene; since Aug Song, Jian The role of the cytoplasmatic tail of IgG1 B cell receptors in signalling and B cell development; since July (MD PhD-Thesis) Steingen, Caroline Untersuchungen zur Interaktion von mesenchymalen Stammzellen mit Endothelzellen; since Jan (MD PhD-Thesis) Stöhr, Oliver C 1 Einfluss der Insulinrezeptorsubstrat-1-Signaltransduktion auf die Energiehomöostase; since Sept Stratis, Athanasios B 5 Die Rolle von I kappa B Kinase 2 in der Entstehung entzündlicher Hautkrankheiten; completed in March Svorcova, Miriam NG-F Kontrolle der HBV-Infektion: Studien in einem neuen Mausmodel; since March Taranum, Surraya A 2 Characterization of Nesprin-1/Enaptin N-terminal variant Enaptin-165; since April Thomas, Anne C 5 Interaktion von amyloid precursor Protein (APP) und presenilin 1 (PS1) bei Demenz vom Alzheimer-Typ (AD); since Jan (MD PhD-Thesis) von Tresckow, Bastian Bedeutung des Membranproteins CD30 Untersuchung der Zusammenhänge zwischen cholesterinabhängigem shedding von CD30 u. der zellulären Immunregulation; since June (MD PhD-Thesis) Tscharntke, Michael B 5 Funktionen von Rac1 in der Epidermis der Maus; completed in October Uhrig, Silke B 10 Entry and genotoxicity of AAV vectors in the hematopoietic and lymphatic system; since April Ullrich, Roland C 5 Investigation of neuro-navigation guided and PET-guided biospsis of human tumors and their response to therapy in corralation to CD133 expression; since May (MD PhD-Thesis) Urban, Anita A 7 SLC6A14 from human; since Sept Veit, Guido C 8 Functional analysis of novel collagens; since May Weber, Ute B 4 Interaktionen rekombinanter Immunrezeptoren; completed in Feb Weiergräber, Marco C 4 Funktionelle Charakterisierung des neuronalen E-Typ Ca 2+ - Kanals im kardiovaskulären System der Maus; completed in June (MD PhD-Thesis) Weißhaar, Stefan NG VI Die Rolle der nukleären PML-Körper in der zellautonomen Verteidigung gegenüber Viren; since June (MD PhD- Thesis) Wilmes, Claudia C 6 Regulatory role of proteases for the assembly of respiratory complexes; since July Woeste, Andreas C 8 Analysis of the LYVE-1 hyaluronan receptor in tumor progression; since Dec Wurth, Stephanie C 6 Genetische und biochemische Charakterisierung der m-aaa Proteasefunktion in Mitochondrien von Saccharomyces cerevisiae; completed in March Xavier, Charles Peter A 2 Expression, localiszation, regulation, and interactions of the WD40-protein-Coronin 3; since Oct Zaim, Hafida A 2 Analyse der Nesprin-2 defizienten Mäuse; completed in Feb
124 Career Development Zedler, Uta NG-F Analyse des HBV-Eintritts in die Hepatozyten sowie den weiteren Weg der Viren zu visualisieren; since Feb (MD PhD-Thesis) Zhao, Yuan B 6 Role of Rho GTPases in extracellular matrix assembly; since June Zuther, Martin B 4 Antikörper-Zytokin Fusionsproteine; since April Zwolanek, Daniela C 8 FACIT collagens: novel insights; since Jan Medical Dissertations Ameli, Mitra C 5 Imaging-guided therapy assessment using 18F-FLT; completed in Dec Anadol, Evrim C 10 Sequenzierung und immunhistochemische Charakterisierung des murinen Tra2-beta1-Gens; completed in Nov Banat, Mohammed C 4 Elektroretinographische Aufzeichungen der retinalen Erregungsfortleitung in der Mausretina; since Aug Becker, Katrin C 1 Einfluss von chronischer Hyperinsulinämie und Alter auf die tau-phosphorylierung in vivo; since Nov Behrendt, Dieter C 7 Charakterisierung von PAST1 und PAST2 und deren Bindung an PACSIN 1; completed in July Beisel, Claudia Liaison-Group Identification of direct and indirect targets of HOXC9 in neuroblastoma using ChIP-on-chip and gene expression profiling; since Feb Betzler, Christopher C 10 Untersuchung zum Krankheitsverlauf bei Patienten mit molekular-genetisch gesicherter autosomal-rezessiver proximaler spinaler Muskelatrophie Typ I; since March Brandt, Carl C 4 Pharmakologische Untersuchungen der isolierten und umströmten Vertebratennetzhaut; since Dec (in cooperation with Prof. Dr. W. Sickel) Dalg, Nora A 7 SLC6A15 from human; since June Dick, Michelle B 9 Modifier genes in BRCA1/2 mutation carriers - single nucleotide polymorphisms (SNPs) in DR4, STK15, and Rad51; since Dec Filonenko, Inga NG I Soluble E-cadherin as an early prognostic marker in melanoma; since May Funteh, Alfred B 5 Functions of nuclear factor kappa B essential modulator (NEMO) in murine epidermis; completed in April Galetin, Thomas C 4 Strukturelle und funktionelle Untersuchungen zur Expression von Ca v 2.3 E-/R-Typ Kalziumkanälen im Herzen der Maus; since July Gekeler, Julia C 9 Polymorphismen der mtdna als Tumormarker zur Identifikation von minimal residual disease in Kopf-Hals- Tumoren; since July Gerhardt, Felix A 6 Charakterisierung der differenziellen Bedeutung von PI3K und PLCγ für die PDGF-induzierte Zellzyklusprogression in glatten Gefäßmuskelzellen; since July Gomez-Alcazar, Diana A 8 Effects of familial hypertrophic cardiomyopathy-linked Troponin I mutations on the Ca 2+ -controlled switch kinetics of Troponin in cardiac myofibrils; since June Gomolka, Mathias C 1 Adipogenese - Transkripitonelle Regulation bei humanen und 3T3-L1 Präadipozyten; since Oct Görlitz, Nico B 10 Visualisierung zellulärer Strukturen in lebenden Zellen und Markierung eines Kapsidproteins von Adeno-assoziierten Virus Typ 2 mit Fluoreszenzproteinen; completed in Feb Groneck, Volker B 4 Einfluß von Rezeptoraffinität und Adhäsionsmolekülen auf die zelluläre Aktivierung von T-Lymphozyten durch chimäre Rezeptoren gegen CEA; completed in Feb Halbach, René A 6 Interaktionen zwischen dem Phosphodiesterase 5- Hemmer Sildenafil und dem ß-Blocker Nebivolol auf den Gefäßtonus in Rattenaorta; since Oct Henry, Margit C 4 Funktionelle Bedeutung neuer Ca 2+ -bindender Protein- Interaktionspartner spannungsgesteuerte Ca 2+ -Kanäle; since April
125 Career Development Heussen, Florian A 4 Autologous translocation of choroid and retinal pigment epithelium in age-related macular degeneration; completed in Aug Hoyer, Caroline Liaison-Group Verstärkung der antigenspezifischen T-Zell-Antwort gegen Tetanustoxoid durch einen bispezifischen CD3xNCAM Antikörper; completed in June Huntgeburth, Michael A 6 Funktionelle Bedeutung des Arg389Gly ß1-Adrenozeptor- Genpolymorphismus am menschlichen Herzen Unterschiedliche kontraktile Antwort auf Katecholamine und unterschiedliches Ansprechen auf ß-Blocker; completed in May Jahn, Tobias B 4 Antikörper-IL12 Fusionsproteine; since May Kaess, Bernhard B 10 Entwicklung von Kapsidmutanten des Adenoassoziierten Virus 2 mit potentiellem Tropismus für KDR; completed in April Kallwass, Caroline Liaison-Group Genexpressions-profiling of a primary stage 4S neuroblastoma by serial analysis of gene expression; since Nov Kamp, Marcel A. C 4 Charakterisierung von carboxyterminalen Interaktionspartnern des spannungsgesteuerten Kalziumkanals Ca v 2.3; since Sept Klehr, Martin Liaison-Group Ch.MK1: Entwicklung eines humanisierten anti-ncam Antikörpers zur therapeutischen Anwendung gegen das Neuroblastom; completed in Nov Klein, Christoph A 5 Signaltransduktion des β2-adrenozeptors im Mausherzen; since March Knöß, Naomi B 6 Isolation and function of processed and unprocessed laminin 5; completed in Jan Köhle, Julia B 1 Charakterisierung des Tumorwachstums humaner Melanomzellen im Nacktmausmodell; since Feb Köskeroglu, Ibrahim C 10 Molekulare Ursachen bei familiärer distaler Arthrogryposis; since March Krieger, Andreas C 4 Identifzierung von Protein-Interaktionspartnern der zytosolischen II-III Schleife spannungsabhängiger E-Typ Ca 2+ -Kanäle; since Dec Krone, Florian A 6 Korrelation zwischen Serum-Testosteronspiegeln und dem Schweregrad der koronaren Herzerkrankung; since Jan Kubacki, Torsten A 8 Effect of Ca 2+ -sensitizers on the kinetics of the Ca 2+ - controlled switch of Troponin in cardiac myofibrils; since Oct Kuhn, Isolde B 1 Analyse des invasiven Wachstums von Melanomzellen nach gezielter Inaktivierung von MMPs in Tumorzellen und Fibroblasten; since July Kuper, Bettina A 6 Signaltransduktion PDGF-vermittelter Reaktionen von glatten Gefäßmuskelzellen im Rahmen der Atherogenese; completed in July Larbig, Robert A 3 Dominant-negative suppression of HCN channels markedly reduces the native pacemaker current I f and undermines spontaneous beating of neonatal cardiomyocytes; completed in July Lehnartz, Patrizia C 9 Transfektion von Mitochondrien mit humaner mtdna in intakten Zellen; since Jan Leuchs, Maike A 6 Einfluss von PI3K und PLCγ auf das PDGF-vermittelte vaskuläre Remodeling in Pulmonalgefässen und die Ausbildung einer pulmonalarteriellen Hypertonie; since Feb Lingohr, Thea C 4 Strukturelle und funktionelle Untersuchungen zur Expression spannungsabhängiger Ca 2+ -Kanäle in der Rinder-, Ratten- und Maus-Retina; completed in June Lösch, Corinna B 4 Generierung und Charakterisierung der rekombinanten anti-melanom T-Zell Rezeptoren scfv61-fc-γ und scfv61- Fc-ζ; completed in Jan Lüke, Matthias C 4 Effekte des Phosphodiesterasehemmers Sildenafil auf die Funktion der isolierten umströmten Vertebratennetzhaut. Elektroretinographische Untersuchungen; completed in March Lutz, Sandra B 10 Optimierung Adeno-assoziierter Viren als Vektoren für die Gentherapie solider Tumoren; completed in Oct
126 Career Development Maghsoodian, Mehran C 4 Morphometrische Analyse hippokampaler und retinaler Gewebeschnitte von E-Typ - Ca 2+ -Kanal defizienten Mäusen und Kontrolltieren; since Sept Maier, Annakarina A 4 Die Rolle des Oberflächenintegrins α5β1 in der Entstehung der hypoxisch bedingten Retinopathie; completed in Oct Manista, Klaudia B 4 Generierung und Charakterisierung rekombinanter Immunrezeptoren gegen das Melanom; completed in Oct Marquardt, Thomas B 4 Spezifische Aktivierung von T-Zellen durch rekombinante Immunrezeptoren mit antikörperähnlicher Spezifität: Einfluss von Antigendichte und B7-Kostimulation auf die Immunrezeptor-vermittelte Aktivierung von CD4+ und CD8+ T-Zellen; completed in Jan Matar, Nora C 4 Einfluss neuerer Antiepileptika auf spannungsgesteuerte T-Typ Ca 2+ -Kanäle; since Aug Möllers, Myriam C 9 Pathomechanismen der Punktmutationen G7497A und T7512C des mitochondrialen trna Ser(UCN) -Gens; completed in Jan Motloch, Lukas A 3 Modulation of HCN channels by the beat-subunit MiRP; since Jan Müller-Wellensieck, Kassandra A 6 Effekte von Aldosteron auf die kardialale Hypertrophie und Funktion in TGFbeta-transgenen Mäusen; since Oct Nass, Daniel A 5 Krankheitsrelevante Mutationen von Genen der Calciumkanaluntereinheiten bei Autismus; since Jan Ndolumingo, Maxime A 6 Effekte von Rot- und Weißwein auf die Atherosklerose- Entstehung in LDLR -/- /smlrp1 -/- -Mäusen; since Feb Nolting, Jens Liaison-Group Functional relevance of the tumor-associated antigen PRAME for proliferation, apotosis and differention in neuroblastoma; since Oct Ollenschläger, Julia Liaison-Group Identification of genes associated with progressive neuroblastoma of stage 4 using SAGE; since Jan von Olshausen, Gesa NG-F E-cadherin Regulation in der HBV-Infektion; since May Quabach, Ralf Alexander A 7 Untersuchungen zur Funktion und Lokalisation eines neuen, noch nicht näher spezifizierten Membrantransporters (UST3h) des Menschen; completed in Dec Ramos Garrido, Soraya A 2 Localisation studies of Annexin A7 in the brain, and analysis of an AnxA6/A7 double knockout mouse; completed in Nov Reintjes, Christian A 5 Einfluss pathophysiologisch relevanter Spleissvarianten des Calciumkanalporenprotein Ca v 1.2 auf Dihydropyidineffekte; since Aug Rottländer, Dennis A 5 Der Effekt von MANT-substitutierten Nukleosidanaloga auf den Adenylatzyklase-stimulierten Calciumstrom; since Aug Ruess, Daniel Friedrich A 8 Ca 2+ -controlled switch kinetics of rabbit skeletal Troponin in psoas myofibrils; since April Schlapakow, Elena C 4 Definition und Nachweis pharmakoresistenter spannungsgesteuerter Kalziumkanäle; since March Schramm, Christoph A 3 Subtype specific effects of ivabradine on HCN1-4; since Jan Schwab, Moritz A 6 Signalmechanismen des PDGF-induzierten pro-apoptotischen Effektes in vitro und in vivo; since Dec Schwarzer, Verena C 10 Molekulargenetische Untersuchungen zur autosomalrezessiven proximalen spinalen Muskelatrophie (SMA); completed in Aug Shahverdyan, Robert A 6 Einfluss von Transforming Growth Factor-ß auf den mitochondrialen Energiemetabolismus in Kardiomyozyten; since Dec Shiue, Jai-Jün B 9 Modifier genes in BRCA1/2 mutation carriers - single nucleotide polymorphisms (SNPs) in ARLTS1, AKAP10, and AKAP13; since March Siapich, Sergej C 4 Suche nach Antagonisten der Interaktionspartner 121
127 Career Development spannungsgesteuerter Ca 2+ -Kanäle, welche die durch Ni 2+ -induzierte Stimulierung der retinalen Erregungsfortleitung beeinflussen; since Sept Sierakowski, Eva A 8 Development of a method to determine the Ca 2+ - controlled switch kinetics of Troponin in cardiac myofibrils; since Sept Simion, Oana NG IV Analysis of RNA expression of hedgehog signalling components during skin development; since March Sparwel, Jan A 6 Vergleichende Untersuchung der Effekte verschiedener Rot- und Weißweine auf die Inhibition PDGF-vermittelter Signale in glatten Gefäßmuskelzellen; completed in Nov Stindt, Daniel A 5 Calciumkanalexpression in diabetischen Mausmodellen; since June Sun, Yuli C 10 Molecular and functional analysis of intragenic SMN1 mutations in patients with spinal muscular atrophy; completed in April Szyman, Agnieszka B 9 Modifier genes in BRCA1/2 mutation carriers - single nucleotide polymorphisms (SNPs) in RNASEL, p53, and TFGβ; since Aug Taghavi, Jubin B 4 Immunrezeptoren mit Spezifität für MDR; since April Van Aaken, Sebastian A 3 Single channel properties and conductance of HCN subtypes; since Aug Vogel, Anne Liaison-Group Herstellung humanisierter Antikörper gegen GD2; since Vogel, Dennis A 6 Prävalenz der HIV-assoziierten Kardiomyopathie in der Prä-HAART-Ära und der HAART-Ära; since Jan Voth, Harald Liaison-Group Identification and charaterization of novel transcripts with differential expression in neuroblastoma stage 4 and 4S; since Jan Weiland, Fabian A 1 Analyse der Insulinwirkung im zentralen Nervensystem durch konditionale Inaktivierung des Insulinrezeptorgens in Mäusen; completed in Nov Werner, Mario A 6 Prävalenz der pulmonalarteriellen Hypertonie bei HIV- Patienten Eine Screening-Untersuchung; since Jan Wieczarkowiecz, Anja B 4 MHC-unabhängige tumorspezifische T-Zellaktivierung durch rekombinante Immunrezeptoren mit kombinierter CD28-CD3ζ Signalleitung; completed in June Wollmer, Iris B 1 Vergleichende Untersuchungen von Metalloproteinasen und Integrinrezeptoren in Melanomzellen von humanen Primärtumoren, Lymphknoten- und Organmetastasen; completed in Sept Diploma-Theses ( Diplom-Biology ) Anton, Fabian C 6 Mitochondrial dynamics: stability and assembly of the dynamin-like GTPase Fzo1; since May Bach, Markus A 7 Funktionelle Charakterisierung von Einzelnukleotidpolymorphismen des humanen Ergothionein-Transporters; completed in June Bartels, Peter A 5 Die biophysikalische Charakterisierung von spannungsabhängigen T-Typ Calciumkanälen (Ca v 3.1 und Ca v 3.2) in einer transient transfizierten Zelllinie (HEK 293) mittels Patch-Clamp-Technik unter rauscharmen Bedingungen durch die Verwendung von Quarzglas; completed in Feb Becirovic, Elvir C 10 Identifizierung und funktionelle Charakterisierung von syndromalen und nicht-syndromalen Taubheitsgenen; completed in Sept Behringer, Arnica B 4 Generierung und funktionelle Charakterisierung eines rekombinanten anti-cea T-Zell-Rezeptors mit einer Maus- Spacer Domäne; completed in March Belgard, Bengt A 1 Role of PDK-1 in energy homeostasis; completed in May Bienemann, Manuela B 4 TCR-like Immunoreceptors; since Dec Boucas, Jorge Miguel Martins B 10 Anti-proliferative properties of adeno-associated virus: potential uses in cancer therapy; completed in June
128 Career Development Boujtat, Fouzia C 8 Analyse der Bindungsstellen von Netrin-1 an seine Rezeptoren; completed in Aug Brinkmann, Kerstin B 8 Proteasome inhibitor-mediated XIAP down-regulation; since March Broxterman, Matthias NG-F Patter recognition of hepatitis B viruses; since Sept Choi, Su B 4 Antibody-IL7 fusion proteins; since Aug Dargazanli, Sascha C 6 Subcellular sorting of prohibitins in mouse embryonic fibroblasts; completed in July Deggerich, Anke B 8 RNA interference (RNAi)-mediated XIAP knock-down in Hodgkin Lymphoma; completed in Jan Dip, Phat Vinh C 6 Characterization of Ylr168c; since Feb Ebermann, Inga C 10 Expression von Stopp-Mutationen des MYO7A-Gens von Patienten mit Usher-Syndrom Typ 1B; completed in Sept Eckertz, Sabrina C 9 Einfluss von Punktmutationen der mitochondrialen DNA auf Protein-turnover und Eigenschaften der trna Leu(UUR) in Zybridzellen; completed in March Ehling, Manuel B 4 Anti-CD25 Immunrezeptoren und Interaktion mit regulatorischen T-Zellen; since Aug El Allaki, Zin-Eddin B 6 Functional characterisation of wild type and mutated CYLD protein; completed in Jan Ernst, Marianne A 1 Conditional expression of FOXO1 mutants in the mouse; completed in Nov (master of science) Fein, Nadin A 1 (1) Conditional expression of K ATPchannel mutants in the mouse. (2) Generation of genetics reporter systems for cytokine signaling; completed in Oct (master of science) Fork, Christian A 7 Substratsuche und Untersuchung der Lokalisation des humanen Transporters SLC22A13; completed in June Fres, Julia NG VI Lipidmodifikation von humanem Guanylat-bindenden Protein 1; completed in Feb Garbes, Lutz C 10 Aufklärung der molekularen Grundlage des alternativen Spleißens von SMN Exon 3 und dessen Einfluß auf den Phänotyp; completed in Sept Gernemann, Christian NG VI Untersuchung der Interaktionen von Proteinen der Sp100 Familie; completed in May Gückel, Eva NG-F Bindungscharakteristika von Single-chain Antikörper- Fragmenten gegen HBV Hüllproteine; completed in March Günther (Osman), Christof C 6 Identification of genetic interactions of PHB1 and PHB2; completed in Feb Hardt, Olaf C 6 Einfluss der mitochondrialen Aktivität auf den Export von Peptiden aus Mitochondrien; completed in Feb Hartmann, Daniel B 4 Antibody-IL12-IL2 fusion proteins; since Aug Hasenbach, Kathy C 10 Modulationen der AMPA-Rezeptor vermittelten Exzitoxozität durch die pharmakologische Expressionssteigerung des GluR2-Gens; since June Hauke, Jan C 10 Analyse von Inhibitoren der Histon-Deacetylasen auf die Expression des Survival of Motor Neuron Gens 2; completed in March Heinrichsdorff, Jan B 4 Generierung eines rekombinanten Immunrezeptors mit Spezifität für IL-15 exprimierende Zellen; completed in May Hettich, Moritz NG I Untersuchungen zur Rolle der extrazellulären Cadherin-Domäne 1 bei Cadherin-vermitteltem Zellsortierungsverhalten; completed in May Hohn, Gregor NG-F Chimäre T-Zellrezeptoren gegen Hepatitis B Hüllproteine: Evaluation in vivo; completed in Feb Jäckel, Sandra A 1 TAP-tag knockin of the protein-arginin-methyltransferase CARM1 in vivo, completed in Nov Jakubik, Miriam C 10 Funktionelle Analyse des Bdp1-Proteins mit Hilfe von 123
129 Career Development konditionalen knock-out Mäusen; completed in March Jangsangthong, Wanchana A 5 Mutation of a human cardiac L-type Ca2+ channel β1- subunit by truncating the N-terminus to understand its functional properties; completed in Feb Jordan, Sabine A 1 Identification and characterisation of metabolically regulated micrornas in liver; completed in Nov Kadow, Stefanie NG-F Spezifische Elimination Hepatitis B Virus infizierter Hepatozyten durch modifizierte humane T-Zellen, die einen chimären T-Zellrezeptor exprimieren und eine zytotoxische Immunantwort etablieren; completed in Dec Klisch, Bruno A 1 Conditional expression of Pi3-kinase mutants in the mouse; completed in Sept Klöckener, Tim A 1 Hormonelle Kontrolle hypothalamischer Funktionen und Plastizität; since March Kocak, Hayriye Liaison-Group Functional role of HOXC09 in neuorblastoma pathogenesis; since Feb Könner, Christine A 1 Role of insulin-signal transduction in AgRP expressing neurons; completed in Dec Kopp, Marion B 7 Charakterisierung von Bindungspartnern der atypischen GTPase RhoBTB3; completed in April Kulozik, Florentine Pia A 8 Effects of phosphorylation and cardiomyopathy-linked Troponin I mutations on the Ca2+-controlled switch kinetics of Troponin in cardiac myofibrils; since June Kwasny, Thomas A 1 Konditionale Mutagenese des hairy enhancer of split 1 (Hes-1) Gens in Adipozyten der Maus; since July Lazic, Daliborka A 7 Charakterisierung of the chicken ergothioneine transporter (ETT); since Aug Lucas, Tina A 7 SLC22A14 from human; since Aug Makhnatkina, Olga B 4 Murine Immunrezeptoren; completed in March Meinecke, Anke NG VI Regulation von SUMO und Ubiquitin Konjugation; since March Neumann, Sascha C 7 Etablierung und Charakterisierung induzierbarer Huntingtin Fibroblastenlinien; completed in Dec Noetel, Andrea A 7 Charakterisierung extranneuronaler Monoaminetransporter (EMT) in salivary glands; since Aug Önal-Akan, Aysel B 11 Genkartierung bei Palmoplantarkeratosen; since Sept Pawlik, Barbara A 7 Substratsuche für den renalen humanen Transporter OCT2; completed in June Peche, Vivek A 2 Characterization of CAP2, Cyclase Associated Protein 2; completed in Aug Pietsch, Julia A 7 Charakterisierung of the ergothioneine transporter (ETT) in human monocytes; since Aug Plume, Nadine NG V Pathophysiological mechanisms of FGF10 mutations causing Levi-Hollister syndrome; completed in Feb Rastetter, Raphael A 2 Expression, localization, regulation, and interactions of the WD40-protein Coronin 3; since Oct Redemann, Nora A 1 Cloning and expression analysis of hypothalamic micrornas; completed in Sept Riessland, Markus C 10 Untersuchung von Substanzen, die die Expression der/ des Survival Motor Neuron RNA/Proteins bei Patienten mit spinaler Muskelatrophie heraufregulieren; completed in Sept Riet, Tobias B 4 Funktionelle Interaktion zwischen rekombinanten Antikörper-basierten Immunrezeptoren und dem nativen T-Zell-Rezeptor-Komplex in humanen T-Lymphozyten; completed in March Rosenbaum, Sabrina C 7 Identifizierung und Charakterisierung von EH Domänen Interaktionspartnern der EHD Proteinfamilie; completed in July Sallach, Jessica B 10 Detailed analysis of the AAV: hepatocyte interaction; since March (master of science) 124
130 Career Development Schael, Sylvia C 7 Regulation der PACSIN Proteine durch Phosphorylierung; completed in Feb Schmidt, Patrick B 4 Funktion der anti-melanom Immunrezeptoren bei primären Melanomzellen; completed in March Schneider, Maria A 2 Ermittlung neuer Noance und Enaptin bindender Proteine mittels des Zwei-Hybrid-Systems; completed in March Schulz, Katharina B 4 TCR-like Immunoreceptors; since Feb Sequaris, Gilles NG I Proteolytic cleavage and nuclear access of cadherins; completed in April Seufert, Sebastian C 10 In vitro Identifizierung und Charakterisierung neuer Zielgene zur Behandlung maligner Gliome; completed in March Silke Arzberger NG-F Apoptose-Sensitivität HBV infizierter Zellen; completed in Sept Stodtmeister, Torben NG I Role of atypical PKC and the small GTPase Rac1 in cadherin mediated gastrulation movements in Xenopus laevis; completed in Aug Thiem, Stefan C 7 Charakterisierung der Interaktion zwischen PACSIN 2 und der Metalloprotease ADAM9; completed in July Tsiockas, Wasiliki B 10 Mikroarray-Analysen von AAV-Vektoren des Serotyps 2; completed in Dec Vierkotten, Sarah C 7 Charakterisierung von PACSIN 2 - Ionenkanal Interaktionen; since March Wilmes, Claudia C 6 Characterization of a novel metalloprotease, Atp23, important for mitochondrial respiratory function; completed in July Willmes, Diana A 1 The Generation of Akt1 and Akt2 Tap Tag vectors using the Red/ET Recombination; since May Witschas, Katja A 5 Einfluß einer γ-untereinheit auf die Kinetik und Pharmakologie von T-Typ Calciumkanälen Eine Untersuchung des Einzelkanalschaltverhaltens mittels Patch-Clamp-Technik; completed in March Zhang, Yaxin A 5 Effect of obesity and diabetes on murine cardiac L-type calcium channels; since March Zwolanek, Daniela C 8 KOLLAGEN XII UND XIV: Modulatoren der Stabilität der Extrazellulären Matrix; completed in Nov
131 Seminars & Meetings The CMMC gives high priority to scientific exchange. This aim is achieved at different levels: 1. the annual retreats of the center and 2. the CMMC seminar series, 3. the yearly international symposia of the CMMC (Ernst Klenk Symposium) and 4. other international meetings (for example the Sweden Symposium 2005), the annual meetings in cooperation with the other Interdisziplinären Zentren für klinische Forschung (IZKF) in Germany. In addition the CMMC participates also in the scientific organization of the Cologne Spring Meetings organized by the Institute for Genetics. The CMMC provides also a platform to communicate biomedical science to visiting undergraduates from universities abroad and to the public in particular to school classes. A summary of these meetings is given in section Annual Retreats of the CMMC In 2000, the first CMMC annual retreat was organized at the Bayer conference center in Wermelskirchen. This internal CMMC event, where all CMMC groups alternately present progress reports, has become highly instrumental in the exchange of scientific information, expertise and ideas, often leading to the initiation of new co-operations. In recent meetings there has been an increasing participation of diploma and graduate students. 2. CMMC Seminar Series CMMC seminars are open to staff from all departments and institutes of the Faculty of Medicine and the Faculty of Mathematics and Natural Sciences. Upon recommendation by individual principal investigators, outside scientists are invited to present their results in the CMMC seminar series. In order to facilitate scientific communication, the guest speaker is asked to reserve time for scientific discussion with individual members of the CMMC. Occasionally, the CMMC also invites biotechnology companies for presentations of novel equipment on recent technologies. An overview of the CMMC-seminars from January 2005 until beginning of June 2007 is given below January Thierry VandenDriessche Center for Transgene Technology & Gene Therapy University of Leuven, Belgium Gene Therapy for Hemophilia February Walter Doerfler former CMMC principal investigator Institute for Clinical and Molecular Virology, Erlangen University Medical School, Germany The Adenovirus Type 12 Hamster Tumor Model: Alterations in Methylation and Transcription Patterns March Klaus-Armin Nave Max-Planck-Institute of Experimental Medicine, Göttingen, Germany Axon-Glia Interactions and Genetic Diseases of Myelin Formation March Max Topp Internal Medicine, University Hospital Tübingen, Germany Targeting B-cell Malignancies by T-cells April Karl Matter Division of Cell Biology, Institute of Ophthalmology, University College London, UK Tight Junctions in the Regulation of Epithelial Proliferation and Differentiation April Wolfgang Uckert Max Delbrück Center for Molecular Medicine (MDC), Berlin, Germany Reprogramming of T Lymphocyte Specificity by Retrovirus mediated T Cell Receptor Gene Transfer: Prospects and Problems May Carmen Alvarez-Dominguez Servicio de Immunologia, Marqués de Valdecilla University Hospital, Santander, Catabria, Spain Listeria Monocytogenes: An Infection Model to Investigate Phagosomal Microbicidal Mechanisms May Inger Helene Madshus Institute of Pathology, University of Oslo, Norway Endocytic Trafficking of the Epidermal Growth Factor Receptor May Peter Schirmacher former CMMC Principal Investigator Institute of Pathology, University of Heidelberg, Germany Hepatocellular Carcinoma - Novel Molecular Approaches to tackle an Orphan Disease June Dirk Dietrich Department of Neurosurgery, University of Bonn Medical Center, Germany Synaptic Innervation of Proliferating and Mature Oligodendroglial Cells 126
132 Seminars & Meetings July Ludwig Eichinger Institute for Biochemistry I, University of Cologne, Germany The Genome of the Social Amoeba Dictyostelium Discoideum July Christoph Renner Internal Medicine I, Saarland University Hospital, Germany Do MHC-Peptide Specific Antibodies Mimic the T-cell Receptor? September Jan-Åke Gustafsson Department of Medical Nutrition, Center for Biotechnology, Karolinska Institute, Stockholm, Sweden Estrogen Receptor Beta - a new Paradigm in Estrogen Signaling September Masataka Kawai Department of Anatomy & Cell Biology, College of Medicine, University of Iowa, USA Reconstitution of the Thin Filament in Skinned Cardiac Muscle Fibers, and the Role of Regulatory Proteins in Force Generation October Thomas Brunner Division of Immunopathology, Institute of Pathology, University of Berne, Switzerland Death Ligand-Mediated Apoptosis in Immuno- Pathologies October Martin Scheffner Laboratory of Biochemistry, Department of Biology, University of Konstanz, Germany The Ubiquitin-Protein Ligase E6-AP and its role in Cervical Carcinogenesis November Walter Birchmeier Max Delbrück Center for Molecular Medicine, Berlin, Germany The Role of Catenins in Cell Adhesion and Signal Transduction November Edgar Dahl Institute of Pathology, Molecular Oncology Group, University Hospital of Aachen, Germany Exploring the Molecular Basis of Breast and Ovarian Cancer: Expression based Methods identify Novel Markers and Gene Signatures potentially applicable in Tumor Diagnosis November Paul Saftig Unit of Molecular Cell Biology and Transgenic Research, University of Kiel, Germany Ectodomain Shedding and Cell Adhesion 2006 January Klaus Ebnet Institute of Cell Biology, Center for Molecular Biology of Inflammation, University of Muenster, Germany Regulation of Cell Polarity by Cell Adhesion Molecules January Jennifer Louise Gallop Neurobiology Division, MRC-Laboratory for Molecular Biology, Cambridge, UK Endophilin Promotes Membrane Curvature in a fast, Clathrin-Independent Pathway of Synaptic Vesicle Endocytosis January Nikolaus Pfanner Institute for Biochemistry and Molecular Cell Biology, University of Freiburg, Germany Import and Assembly of Mitochondrial Proteins February Pontus Aspenstrom Ludwig Institute for Cancer Research, Uppsala, Sweden Atypikal Rho GTPases February Harald Bär German Cancer Research Center, Heidelberg, Germany The Functional Role of Inherited Desmin Mutations for the Development of Cardiomyopahies February Harald Herrmann-Lerdon German Cancer Research Center, Heidelberg, Germany Mechanics of Intermediate Filament Assembly: Striking Differences between Nuclear and Cytoplasmic Systems February John Rice Functional Genomics and Systems Biology, IBM T.J. Watson Research Center, New York, USA A Spatially-Detailed Model of Actin and Myosin Interaction in the Cardiac Myofilament including Strain-Based Rates and Cooperative Mechanisms March Gerrit van Meer Membrane Enzymology Bijvoet Center / Institute of Biomembranes Utrecht University, Netherlands Cellular Lipidomics, Domains, Flippases, and Cell Physiology 127
133 Seminars & Meetings April Stephan Urban Department of Molecular Virology, Otto Meyerhof Centrum, University of Heidelberg, Germany Towards a Molecular Understanding of the Early Events of Hepatitis B Virus Infection: Entry Inhibition as a new Therapeutic Concept for the Treatment of Chronic Hepatitis B? May Helmut Jonuleit Department of Dermatology, University of Mainz, Germany Human CD4+CD25+ Regulatory T Cells: Subsets and Functional Properties May Sylvia Krobitsch Max Planck Institute for Molecular Genetics, Berlin, Germany Spinocerebellar Ataxia Type 2: Functional Characterization of Ataxin-2 May Ramin Massoumi Division of Experimental Pathology, Lund University, Sweden Cyld Inhibits Tumor Cell Proliferation by Blocking Bcl-3- Dependent NF-kappaB Signalling May Hans Spelbrink Institute of Medical Technology, FinMIT Centre of Excellence, University of Tampere, Finland Mitochondrial DNA Mutations in Disease and Ageing June Andreas Waha Institute for Neuropathology, University of Bonn, Germany Genome Wide Analysis of DNA Methylation by CpG Island Microarrays June Cornelis J. Weijer Wellcome Trust Biocentre University of Dundee, UK Chemotactic Cell Movement During Dictyostelium Development and Chick Gastrulation June Benedict Yen Department of Pathology, University of California, San Francisco, USA Role of Endoplasmic Reticulum Stress in the Pathogenesis of Hepatitis B and the Development of Hepatocellular Carcinoma August Yasuyuki Fujita MRC Laboratory for Molecular Cell Biology & Cell Biology Unit, London, UK Dynamic Regulation of the Cadherin-Based Cell-Cell Contacts September Francois Fagotto Department of Biology, McGill University Montreal, Canada How Embryonic Tissues Separate from each other: Insights from Amphibian Embryos September Christos Mantzoros Clinical Research Overseer, Endocrinology Diabetes and Metabolism, Harvard Medical School, Boston, USA The Adipocyte as an Endocrine Organ: Clinical Significance of Adipocytokines in Humans September Albert Wang Boston Biomedical Research Institute, USA Caldesmon: From Smooth Muscle Contraction to Cell Migration October Alpha Yap Institute for Molecular Bioscience, Univ. of Queensland, Brisbane, Australia A Cadherin User s Manual: Coordinating Adhesion, Signaling and the Cytoskeleton November Rainer Mußmann DeveloGen AG, Göttingen, Germany The Discovery and Development of Small Molecules Promoting Growth and Survival of Pancreatic Beta Cells 2007 January Pawel Ciborowski Center for Neurovirology and Neurodegenerative Disorders, University of Nebraska Medical Center, Omaha, USA Biomarkers for HIV-1 Associated Dementia: Proteomics Approach January Martina Müller-Schilling Department of Internal Medicine IV, University of Heidelberg, Germany The p53 Family and Apoptosis - Roles in Tumor Formation and Treatment Response February Elior Peles Department of Molecular Cell Biology, The Weizman Institute of Science, Rehovot, Israel Neuron-glia Interaction at the Nodes of Ranvier 128
134 Seminars & Meetings February Celine Revenu Institute Curie, Paris, France Villin Severing Activity Enhances Actin-based Motility in vivo March Jonathan Schneck Department of Pathology, Johns Hopkins School of Medicine, Baltimore, USA Setting the Terms of Engagement: Characterization of TCR Membrane Organization and Development of artificial Antigen Presenting Complexes, aapc April Björn Chapuy Department of Hematology and Onkology, University Hospital Göttingen, Germany ABCA3 expression in AML blast cells - Association with poor disease outcome and intracellular drug sequestration April Anthony Corfield Division of Clinical Sciences, University of Bristol, UK Sugar Codes in Mucosal Protection and Human Gastrointestinal Host-Microflora Interaction April Jonathan Yewdell National Institute of Allergy and Infectious Diseases, NIH, Bethesda, USA How to Succeed in Science without Really Trying organized with the Interdisciplinary Postgraduate Program Molecular Medicine of the Univ. Of Cologne May Peter Brouckaert Ghent University / Department for Molecular Biomedical Research, Gent, Belgium Molecular mechanisms of the in vivo tumor destructive and shock inducing properties of Tumor Necrosis Factor June José M. Cuezva Universidad Autónoma de Madrid, Spain The Bioenergetic Signature of Cancer June Annette Müller-Taubenberger Institute for Cell Biology, Ludwig Maximilians University Munich, Germany Regulation of Cytokinesis Investigated in the Eukaryotic Model Organism Dictyostelium discoideum June Zena Werb Department of Anatomy, University of California, San Francisco, USA Of Mice and Women-Common Themes in Mammary Development and Breast Cancer 3. International Scientific Meetings of the CMMC The CMMC organizes a yearly international scientific meeting. Since 2005, these meetings named Ernst Klenk Symposia in Molecular Medicine are organized together with the Ernst Klenk Foundation, Marburg. These two-day meetings are intended to provide a forum for discussion of the state-of-the-art research in the fields of molecular biology related to biomedical research for interested scientists, physicians and students from academia and industry. Organizational Structures of the Meetings A committee consisting of both members of the executive board of the CMMC and international scientists decide on the topic of the meetings. Subsequently, the committee asks an outstanding scientist to structure the conference on the selected topic and to invite 15 to 20 scientists in this research area as guest speakers. The intention is that the guest speakers should not only present their most recent research results but should also discuss fundamental problems and provide ideas to stimulate future research. Following the tradition, the scientific coordinator of the meeting is giving the Ernst Klenk-Lecture, which serves as one highlight of the symposium. 21 st Ernst Klenk Symposium in Molecular Medicine on Atherosclerosis - Mediators, Mechanisms and Interventions December 02-03, 2005 in Cologne Jorge Plutzky - Boston - USA Scientific Organizer and Speaker of the Ernst Klenk Lecture Diabetic Atherossclerosis: New Targets, New Pathways Session I Metabolic Aspects of Atherogenesis Jens C. Brüning - Cologne - Germany Alan Chait - Seattle - USA Matthias H. Tschöp - Cincinnati - USA Bart Staels - Lille - France Erik S. Stroes - Amsterdam - Netherlands Session II Inflammation and Molecular Mediators Paresh Dandona - Buffalo - USA Ulrich Knitscher - Berlin - Germany Stephan Rosenkranz - Cologne - Germany Russell P.Tracy - Burlington - USA Menno P.J. de Winther - Maastricht - Netherlands Session III Acute Coronary Syndromes Erland Erdmann - Cologne - Germany Heinz-Peter Schultheiss - Berlin - Germany 129
135 Seminars & Meetings Christian Torp-Pedersen - Copenhagen - Denmark Nicholas J. Wareham - Cambridge - UK Robert G. Wilcox - Nottingham - UK Organization Board Erland Erdmann - Cologne - Germany Wilhelm Krone - Cologne - Germany Uta C. Hoppe - Cologne - Germany Markus Schubert - Cologne - Germany Stephan Rosenkranz - Cologne - Germany 22 nd Ernst Klenk Symposium in Molecular Medicine on Emerging Infectious Diseases November 05-07, 2006 in Cologne Peter Palese, New York - USA Scientific Organizer and Speaker of the Ernst Klenk Lecture What Can We Learn from Reconstructing the Extinct 1918 Pandemic Influenza Virus? Key Note Lecture Claire M. Fraser-Liggett - Rockville - USA Session I The Innate Immune Systems and Pathogens Karl-Klaus Conzelmann - Munich - Germany Otto Haller - Freiburg - Germany Philippe J. Sansonetti - Paris - France Session II Smallpox, Filo- and Influenza Virus Heinz Feldmann - Winnipeg - Canada Grant McFadden - London, Ontario - Canada Elke Mühlberger - Marburg - Germany Albert D. Osterhaus - Rotterdam - Netherlands Session III Host-pathogen Interactions Charles M. Rice - New York - USA Kunitada Shimotohno - Kyoto - Japan Stanley M. Lemon - Galveston - USA Session IV Evolving Pathogens I Ralf Bartenschlager - Heidelberg - Germany Reinhard Kurth - Berlin - Germany Willy J.M. Spaan - Leiden - Netherlands Session V Evolving pathogens II Vincent Deubel - Shanghai - China Stefan H.E. Kaufmann - Berlin - Germany Hans-G. Kräusslich - Heidelberg - Germany W. Ian Lipkin - New York - USA Linda J. Miller - New York - USA Session VI Mechanisms of Pathogenicitiy R. John Collier - Boston - USA Bryan R. Cullen - Durham - USA Franz X. Heinz - Vienna - Austria Jonathan W. Yewdell - Bethesda - USA Session VII New Pathogens Caroline Ash - Geneva - Switzerland Don Ganem - San Francisco - USA Organization Board Martin Krönke - Cologne - Germany Hans-Dieter Klenk - Cologne - Germany Herbert Pfister - Cologne - Germany 23 rd Ernst Klenk Symposium in Molecular Medicine on Non - Coding RNA September 30 - October 02, 2007 in Cologne Thomas Tuschl - New York - USA Scientific Organizer and Speaker of the Ernst Klenk Lecture Mechanisms of Small RNA Guided Gene Silencing in Mammals Sessions I Mechanism of Small RNA Guided Gene Silencing in Fungi, Plants, and Protists Philippe Bastin - Paris - France David Baulcombe - Norwich - Great Britain Craig P. Hunter - Cambridge MA - USA Giuseppe Macino - Rome - Italy Wolfgang Nellen - Kassel - Germany Session II Other Non-Coding RNAs Jürgen Brosius - Münster - Germany Ivo L. Hofacker - Vienna - Austria Alexander Hüttenhofer - Innsbruck - Austria Jörg Vogel - Berlin - Germany Session III micrornas, sirnas and Disease Reuven Agami - Amsterdam - The Netherlands René Bernards - Amsterdam - The Netherlands Markus Stoffel - Zurich - Switzerland Mihaela Zavolan - Basel - Switzerland Session IV Biochemical Mechanism of RNA Silencing Witold Filipowicz - Basel - Switzerland Matthias W. Hentze - Heidelberg - Germany Elisa Izaurralde - Tübingen - Germany Javier Martinez - Vienna - Austria Mikiko C. Siomi - Tokushima - Japan Organization Board Martin Krönke - Cologne - Germany Hans-Dieter Klenk - Cologne - Germany 130
136 Seminars & Meetings 4. International Scientific Meetings in cooperation with the CMMC Sweden North Rhine - Westphalia Scientific Symposium in Molecular Medicine March 13-14, 2005 in Cologne The symposium originated from an initiative of scientists located at different Universities and other institutions of North Rhine-Westphalia (NRW) supported by the Ministerium für Innovation, Wissenschaft, Forschung und Technologie des Landes NRW to promote the biomedical cooperation between Sweden and NRW within the following research areas: 1. Neurobiology and Neuropsychiatric Diseases, 2. Inflammation and Extracellular Matrix, 3. Tumorbiology, 4. Vascular Diseases and 5. Biomaterials. In charge of the organization of this meeting was the CMMC. Session I Inflammation and Extracellular Matrix Peter Bruckner - Münster - Germany Dick Heinegard - Lund - Sweden Peter Heinrich - Aachen - Germany Mats Paulsson - Cologne - Germany Session II Biomaterials Alfons Fischer - Duisburg-Essen - Germany Thomas Groth - Halle-Wittenberg - Germany Willi Jahnen-Dechent - Aachen - Germany Session III Vascular Diseases Bernd Fleischmann - Bonn - Germany Lydia Sorokin - Lund - Sweden Christer Sylvén - Huddinge - Sweden Session IV Neurobiology Ansgar Büschges - Cologne - Germany Frank Edenhofer - Bonn - Germany Andreas Jacobs - Cologne - Germany Tadeusz Wieloch - Lund - Sweden Session V Tumorbiology Michael Hallek - Cologne - Germany Carl-Hendrik Heldin - Uppsala - Sweden Bernhard Lüscher - Aachen - Germany Rune Toftgard - Huddinge - Sweden Organizer CMMC Cologne Swedish-NRW Research Initiative Molecular Medicine AG Biomaterials NRW IZKF Aachen Biomat IZKF Münster in cooperation with the Ministerium für Wissenschaft und Forschung des Landes NRW ACRC Workshops and Symposia In order to promote the interaction between the nine IZKF distributed all over Germany the Association of Clinical Research Centers at Germany (ACRC) of IZKF organizes annual workshops or symposia. In most cases project leaders of the IZKF are chosen to present their research project. Based on a rotation system the IZKF Leipzig and the IZKF Aachen (BIOMAT) were in charge of the organization of the meeting in 2005 and 2006, respectively. ACRC Workshop Regenerative Medicine March 16-18, 2005 in Meissen Session I Stem Cell Biology and Stem Cell Therapy Michael A. Cross - Leipzig - Germany Hans Jörg Fehling - Ulm - Germany Jürgen Hescheler - Köln - Germany Michael Kühl - Ulm - Germany Catherin Niemann - Köln - Germany Sabine Neuss-Stein - Aachen - Germany Hans Schöler - Münster - Germany Thomas Skutella - Tübingen - Germany Olaf Utermöhlen - Köln - Germany Session II Transplantation and Regeneration Frank Emmrich - Leipzig - Germany Stefan Isenmann - Jena - Germany Ulrich Nöth - Würzburg - Germany Holm Schneider - Erlangen - Germany Michael Sittinger - Berlin - Germany David Pfander - Erlangen - Germany Solon Thanos - Münster - Germany Karin Ulrichs - Würzburg - Germany Session III Biomaterials and Tissue Engineering Augustinus Bader - Leipzig - Germany Holger Fuchs - Aachen - Germany Anke Hartmann - Würzburg - Germany Andreas Lendlein - Teltow - Germany Sven Möllers - Aachen - Germany Christoph Redecker - Jena - Germany Karl-Dietrich Sievert - Tübingen - Germany Christian Weber - Aachen - Germany Organizer: Association of Clinical Research Centres (ACRC) 131
137 Seminars & Meetings ACRC - Symposium Cell-Cell- and Cell-Matrix- Interactions in Tumor Progression and Immunity May 21-23, 2006 in Bamberg Honorary Lecture Dick Heinegard - Lund - Sweden Session I Cell-cell-interactions in inflammation and immunity Jörg Galle - Leipzig - Germany Hans-Martin Jäck - Erlangen - Germany Albrecht Müller - Würzburg - Germany Sussan Nourshargh - London - UK Liliana Schaefer - Münster - Germany Michael Sixt - Martinsried - Germany Dietmar Vestweber - Münster - Germany Session II Cell-matrix-interactions and matrix destruction Rolf Bräuer - Jena - Germany Harald Burkhardt - Erlangen - Germany Willi Jahnen-Dechent - Aachen - Germany Raimund Kinne - Jena - Germany Elke Kunisch - Jena - Germany Manfred Lutz - Erlangen - Germany Russel Taichman - Michigan - USA Lydia Sorokin - Münster - Germany Frank Zaucke - Köln - Germany Wolfgang Ziegler - Leipzig - Germany Session III Cell-cell and cell-matrix-interactions in cell migration and tumorigenesis and invasion Monique Aumailley - Köln - Germany Alexander Berndt - Jena - Germany Johannes Eble - Münster - Germany Beate Eckes - Köln - Germany Peter Friedl - Würzburg - Germany Carsten Hagemann - Würzburg - Germany Dick Heinegard - Lund - Sweden Andreas Ludwig - Aachen - Germany Cornelia Mauch - Köln - Germany Frans Van Roy - Ghent - Belgium Organizer IZKF-Erlangen Klaus von der Mark - Erlangen - Germany Joachim R. Kalden - Erlangen - Germany Jürgen Behrens - Erlangen - Germany Katrin Faber - Erlangen - Germany ACRC Workshop Regenerative Medizin / Extrazelluläre Matrix March 28-30, 2007 in Kerkrade (Abtei Rolduc), NL Sessions / Speakers Session I Kardiales Tissue Engineering Wulf Ito - Lübeck - Germany (Key note lecture) Stefan Jockenhövel - Aachen -Germany Stephan Rosenkranz - Köln - Germany Session II Extrazelluläre Matrix Jöns Hilborn - Uppsala - Schweden (Key note lecture) Mats Paulsson - Köln - Germany (Key note lecture) Monique Aumailley - Köln - Germany Ingo Flesch - Tübingen - Germany Gerd Klein - Tübingen - Germany Manuel Koch - Köln - Germany Carien Niessen - Köln - Germany Session III Funktion dendritischer Zellen Martin Lipp - Berlin - Germany (Key note lecture) Kolja Gelse - Erlangen - Germany Hartmut Schmidt - Münster - Germany Alexander Steinkasserer - Erlangen - Germany Thomas Thum - Würzburg - Germany Attila Yilmaz - Erlangen - Germany Martin Zenke - Aachen - Germany Session IV Neuroretinale Degeneration Charlotte Remé - Zürich - Schweiz Gabriele Thumann - Aachen - Germany Session VI Regeneration im zentralen Nervensystem Hans Werner Müller - Aachen - Germany Gary Brook - Aachen - Germany Stefan Isenmann - Jena - Germany Joachim Weis - Aachen -Germany Organizer IZKF BIOMAT Aachen First Joint Meeting of the French and German Connective Tissue Societies March 10-12, 2005 in Cologne Plenary lectures Attila Aszódi - Martinsried - Germany Jacky Bonaventure - Paris - France Leena Bruckner-Tuderman - Freiburg - Germany Ernst Pöschl - Erlangen - Germany Florence Ruggiero - Lyon - France Walter Stöcker - Mainz - Germany Ellen Van Obberghen-Schilling - Nice - France Laurence Vico - Saint-Etienne - France Oral presentations selected from abstracts Dirk Breitkreutz - Heidelberg - Germany Ehab El-Hallous - Lübeck - Germany Françoise Fauvel-Lafève - Paris - France 132
138 Seminars & Meetings Kay Grobe - Münster - Germany Heinz-Jürgen Hausser - Ulm - Germany Sylvie Ricard-Blum - Lyon - France Patricia Rousselle - Lyon - France Karin Scharffetter-Kochanek - Ulm - Germany Oral presentations selected from young investigator abstracts Ulrike Brandt - Cologne - Germany Salin A. Chakkalakal - Cologne - Germany Mihaela Drumea-Mirancea - Tübingen - Germany Leslie Heckmann - Ulm - Germany Delphine Javelaud - Paris - France Catherine Moali - Lyon - France Benoit Porée - Caen - France Delphine Quélard - Rennes - France Aurelia Raducanu - Martinsried - Germany Léa Ritié - Strasbourg - France Organizers Monique Aumailley - Cologne - Germany Sylvie Ricard-Blum - Lyon - France Cologne Spring Meetings The Cologne Spring Meetings have a long tradition at the University of Cologne. Previously, the spring meetings were organized only by the Institute for Genetics. Since 2001, members of the CMMC have participated in the organization of this meeting series (as indicated with an asterisk*). Cologne Spring Meeting Signal Transduction and Disease February 23-25, 2005 in Cologne Speakers Domenico Accili - New York - USA Johan Auwerx - Illkirch - France Mark T. Bedford - Smithville - USA Warner Green - San Francisco - USA Stephan Herzig - Heidelberg - Germany Randall S. Johnson - La Jolla - USA C. Ronald Kahn - Boston - USA Rüdiger Klein - Martinsried - Germany Peter Krammer - Heidelberg - Germany Werner Müller - Braunschweig - Germany Manolis Pasparakis - Monterotondo - Italy Klaus Rajewsky - Boston - USA Stefan Rose-John - Kiel - Germany Nadia Rosenthal - Monterotondo - Italy Günther Schütz - Heidelberg - Germany Stefan Schütze - Kiel - Germany Bruce M. Spiegelman - Boston - USA Sasha Tarakhovsky - New York - USA Axel Ullrich - Martinsried - Germany Erwin Wagner - Vienna - Austria Organizers Jens Brüning* - Cologne - Germany Martin Krönke* - Cologne - Germany Ursula Lichtenberg - Cologne - Germany Cologne Spring Meeting The Control of Protein Function - Cellular Regulation, Dysfunction and Disease March 01-03, 2006 in Cologne Speakers Bruno André - Bruxelles - Belgium John M. Christie - Glasgow - United Kingdom Ivan Dikic - Frankfurt/Main - Germany Jürgen Dohmen - Cologne - Germany Michael Ehrmann - Essen - Germany Matthew Freeman - Cambridge - United Kingdom Randy Hampton - La Jolla - USA Regine Hengge - Berlin - Germany Mark Hochstrasser - New Haven - USA Jonathan Howard - Cologne - Germany Stefan Jentsch - Martinsried - Germany Thomas Jenuwein - Vienna - Austria Harvey McMahon - Cambridge - United Kingdom Frauke Melchior - Göttingen - Germany Jacques Neefjes - Amsterdam - Netherlands Hugh Pelham - Cambridge - United Kingdom Didier Picard - Genève - Switzerland Titia K. Sixma - Amsterdam - Netherlands Brian J. Staskawicz - Berkeley - USA Helle Ulrich - London - United Kingdom Alfred Wittinghofer - Dortmund - Germany Organizers Thomas Langer* - Cologne - Germany Jonathan Howard - Cologne - Germany Maria Leptin - Cologne - Germany George Coupland - Cologne - Germany Cologne Spring Meeting Evolution of Pathogens and Immunity February 21-23, 2007 in Cologne Speakers Mark Achtman - Berlin - Germany Thomas Boehm - Freiburg - Germany Mike Boots - Sheffield - United Kingdom Stewart Cole - Paris - France Max D. Cooper - Birmingham - USA George Dimopoulos - Baltimore - USA Dieter Ebert - Basel - Switzerland Jonathan Ewbank - Marseille - France Martin Flajnik - Baltimore - USA Neill Hall - Rockville - USA Sheng Yang He - East Lansing - USA Bruno Lemaitre - Gif-sur-Yvette - France Polly Matzinger - Bethesda - USA 133
139 Seminars & Meetings Manfred Milinski - Ploen - Germany Dietmar Schmucker - Boston - USA Joana da Silva - Rockville - USA Paul Schulze-Lefert - Cologne - Germany Michiel Van Boven - Wageningen - Netherlands Irving Weisman - Stanford - USA Brendan Wren - London - United Kingdom Organizers Jonathan Howard - Cologne - Germany Maria Leptin - Cologne - Germany Diethard Tautz - Cologne - Germany Matthias Cramer - Cologne - Germany Kevin Johnson - Cologne - Germany Röbbe Wünschiers - Cologne - Germany 5. Public Relations Events relating to the new Research Building of the CMMC / CCG Spatenstich and celebration of the 10th anniversary of the CMMC The Spatenstich of the new research building was organized in connection with the celebration of the 10th anniversary of the CMMC held on December 19, Celebration of the 10th anniversary of the CMMC Welcome Address Prof. Dr. Thomas Krieg Vice Chancellor, University of Cologne Speeches Prof. Dr. Martin Krönke Chairman of the CMMC Prof. Dr. Wilhelm Stoffel Founder Member of the CMMC Prof. Dr. Franz Hofmann Chairman of the External Scientific Advisory Board of the CMMC Commemorative Speech Is Cancer transmittable? Prof. Dr. Dr. h.c. Harald zur Hausen German Cancer Research Center, Heidelberg Laudation: Prof. Dr. Herbert Pfister CMMC Board Member Spatenstich ceremony of the new research building Welcome Address Prof. Dr. Axel Freimuth Rector of the University of Cologne Speeches Prof. Dr. Andreas Pinkwart Minister of Innovation, Science, Research and Technological Transfer of the State of NRW Fritz Schramma Mayor of the City of Cologne Prof. Dr. Edgar Schömig Dean of the Faculty of Medicine Prof. Dr. Ulrich Radtke Dean of the Faculty of Mathematics and Natural Sciences Prof. Dr. Martin Krönke Chairman of the CMMC Dr. Jörg Blattmann Director of the University Hospital Festakt Spatenstich Bernadel-Trio of the Collegium Musicum Get together with Live Music of the Band Tribute to Richtfest ceremony of the new research building of the CMMC / CCG At the topping-out ceremony (Richtfest) held on May 9th, 2007 the Vice Chancellor of the University of Cologne Prof. Axel Freimuth welcomed more than 300 guests. The mayor of the City of Cologne Fritz Schramma and the representative of the Minister of Building and Transport of the State of NRW, Oliver Wittke, Ministerialdirigent Rüdiger Stallberg acknowledged the importance the landmark building at the University Campus with its biomedical research activities for the BioRiver Region and the State of NRW. The social program was created by five students of the Art Department of the University of Cologne who arranged the art exhibition Betonale Impressionen. The musical accompaniment was held by the quartet Vier Gewinnt of the school of music of Montabaur. Welcome Address Prof. Dr. Axel Freimuth Rector of the University of Cologne Speeches Fritz Schramma Mayor of the City of Cologne Rüdiger Stallberg Ministerialdirigent of the Ministry of Building and Transport of the State of NRW Prof. Dr. Joachim Klosterkötter Dean of the Faculty of Medicine Prof. Dr. Ulrich Radtke Dean of the Faculty of Mathematics and Natural Sciences Prof. Dr. Martin Krönke Chairman of the CMMC Festakt Richtspruch Klaus Schwetzke Foreman, Company BAM Musical Performance: Quartet Vier Gewinnt of the Musikgymnasium, Montabaur Supervised by Martin Schwind 134
140 Seminars & Meetings Studientag Medizin of the Medical Faculty KinderUni at the University of Cologne The medical faculty of the University of Cologne together with the equal opportunities commissioner organizes on an annual basis a one-day event for female high school students who are in their final years of their school education. The aim of this meeting is to provide information of the study courses in Human Medicine at the Medical Faculty and to show the different facets of the duties of a medical doctor in particular at a University Hospital. The students have the possibility to learn about the different clinics and institutes of the Medical Faculty and to gain insights into patient care and treatment as well as into the biomedical research environment. In general, 80 female students from high schools located in the Cologne region participate at the Studientag Medizin. After introductory lectures given predominantly by female Professors of the Faculty the participants are guided through different departments and institutes of the Medical Faculty. Since 2004, the CMMC as an example of a biomedical research center takes part in this event and offers after a short introduction guided tours to research groups and the Central Bioanalytical Facility of the Center. Studientag Medizin 2005 Guided Tours through CMMC Research Groups Frank Berthold - Liaison-Group - Department of Pediatric Oncology and Hematology Franz-Georg Hanisch - Central Bioanalytics Catherin Niemann - NG IV - Institute for Pathology Wilhelm Krone / Markus Schubert - C 1 - Department II for Internal Medicine Bernd Wollnik - NG V - Institute for Human Genetics In 2007, the CMMC participated the first time in the KinderUni organized by the coordination office for science and public relations of the University of Cologne. Carien Niessen and Gerrit Praefcke - both junior research group leaders of the CMMC - took the challenge and presented a lecture (45 min.) for school kids. 15. May Carien Niessen (NG I) Warum fällt mein Körper nicht auseinander? Wie Zellen einander erkennen 21. May Gerrit Praefcke (NG VI) Von der Seifenblase zur Zelle - Membranen als Grenzflächen Studientag Medizin 2006 Guided Tours through CMMC Research Groups Franz-Georg Hanisch - Central Bioanalytics Uta C. Hoppe - A 3 - Department III of Internal Medicine Carien Niessen - NG I - Department II of Internal Medicine, Department of Surgery and Dermatology Ulrike Protzer - NG-F - Institute for Medical Microbiology, Immunology and Hygiene Studientag Medizin 2007 Guided Tours through CMMC Research Groups Franz-Georg Hanisch - Central Bioanalytics Uta C. Hoppe - A 3 - Department III of Internal Medicine Cornelia Mauch - B 1 - Department of Dermatology and Venerology Rita Schmutzler / Babara Wappenschmidt - B 9 - Molecular Gyneco-Oncology, Department of Gynecology 135
141 Photo Art Anika Sturm Annina Laufer Annina Laufer Betonale Impressionen Shahrouz Yazdanyar 136
142 Photo Art Carsten Klett Carsten Klett Jan Klein 137
143 Addresses Prof. Dr. Hinrich Abken B 4 Department I of Internal Medicine Center for Molecular Medicine Cologne (CMMC) Medical Center, University of Cologne Kerpener Str. 62 D Cologne Phone: Fax: [email protected] Prof. Dr. Dr. Monique Aumailley B 6 Institute for Biochemistry II Center for Molecular Medicine Cologne (CMMC) Medical Center, University of Cologne Joseph-Stelzmann-Str. 52 D Cologne Phone: Fax: [email protected] Konstantin Belostotski System Administrator Center for Molecular Medicine Cologne (CMMC) Medical Center, University of Cologne Joseph-Stelzmann-Str. 52 D Cologne Phone: Fax: [email protected] Prof. Dr. Frank Berthold B / Liaison Group Department of Pediatric and Oncology Center for Molecular Medicine Cologne (CMMC) Medical Center, University of Cologne Kerpener Str. 69 D Cologne Phone: Fax: [email protected] Prof. Dr. Jens Brüning A 1/ C 1 Institute for Genetics Center for Molecular Medicine Cologne (CMMC) University of Cologne Zülpicher Str. 47 D Cologne Phone: Fax: [email protected] Dr. Hildegard Büning B 10 Department I of Internal Medicine Center for Molecular Medicine Cologne (CMMC) Medical Center, University of Cologne Kerpener Str. 62 D Cologne Phone: Fax: [email protected] Prof. Dr. Erland Erdmann A 3 Department III of Internal Medicine Center for Molecular Medicine Cologne (CMMC) Medical Center, University of Cologne Kerpener Str. 62 D Cologne Phone: Fax: [email protected] Cornelius Evers Graphics and Art Unit Center for Molecular Medicine Cologne (CMMC) Medical Center, University of Cologne Kerpener Str. 62 D Cologne Phone: Fax: [email protected] Dr. Sascha Fauser A 4 Department of Ophthalmology Center for Molecular Medicine Cologne (CMMC) Medical Center, University of Cologne Kerpener Str. 62 D Cologne Phone: Fax: [email protected] Ute Fiehne-Reiß Secretary Scientific Management Office Center for Molecular Medicine Cologne (CMMC) Medical Center, University of Cologne Joseph-Stelzmann-Str. 52 D Cologne Phone: Fax: [email protected] Ruth Fladerer Secretary Postgraduate Program Molecular Medicine Center for Molecular Medicine Cologne (CMMC) Medical Center, University of Cologne Joseph-Stelzmann-Str. 52 D Cologne Phone: Fax: [email protected] Dr. Debora Grosskopf-Kroiher Scientific Management Office Center for Molecular Medicine Cologne (CMMC) Medical Center, University of Cologne Joseph-Stelzmann-Str. 52 D Cologne Phone: Fax: [email protected] PD Dr. Dirk Gründemann A 7 Institute for Pharmacology Center for Molecular Medicine Cologne (CMMC) Medical Center, University of Cologne Gleueler Str. 24 D Cologne Phone: Fax: [email protected] PD Dr. Dr. Ingo Haase B 5 Department of Dermatology and Venerology Center for Molecular Medicine Cologne (CMMC) Medical Center, University of Cologne Kerpener Str. 62 D Cologne Phone: Fax: [email protected] Susanne Haase Secretary Scientific Management Office Center for Molecular Medicine Cologne (CMMC) Medical Center, University of Cologne Joseph-Stelzmann-Str. 52 D Cologne Phone: Fax: [email protected] 138
144 Seminars & Addresses Meetings Dr. Ulrich Hacker B 10 Department I of Internal Medicine Center for Molecular Medicine Cologne (CMMC) Medical Center, University of Cologne Kerpener Str. 62 D Cologne Phone: Fax: [email protected] Dr. Eric Hahnen C 10 Institute for Human Genetics Center for Molecular Medicine Cologne (CMMC) Medical Center, University of Cologne Kerpener Str. 34 D Cologne Phone: Fax: [email protected] Prof. Dr. Michael Hallek B 10 Department I of Internal Medicine Center for Molecular Medicine Cologne (CMMC) Medical Center, University of Cologne Kerpener Str. 62 D Cologne Phone: Fax: [email protected] Prof. Dr. Franz-Georg Hanisch Bioanalytics Center for Molecular Medicine Cologne (CMMC) Medical Center, University of Cologne Joseph-Stelzmann-Str. 52 D Cologne Phone: Fax: [email protected] Prof. Dr. Wolf-Dieter Heiss C 5 Department of Neurology Center for Molecular Medicine Cologne (CMMC) Medical Center, University of Cologne Kerpener Str. 62 D Cologne Phone: Fax: [email protected] Dr. Hans-Christian Hennies B 11 Cologne Center for Genomics Center for Molecular Medicine Cologne (CMMC) Medical Center, University of Cologne Zülpicher Str. 47 D Cologne Phone: Fax: [email protected] Prof. Dr. Stefan Herzig A 5 Institute for Pharmacology Center for Molecular Medicine Cologne (CMMC) Medical Center, University of Cologne Gleueler Str. 24 D Cologne Phone: Fax: [email protected] Prof. Dr. Jürgen Hescheler C 4 Institute for Neurophysiology Center for Molecular Medicine Cologne (CMMC) Medical Center, University of Cologne Robert-Koch-Str. 39 D Cologne Phone: Fax: [email protected] Prof. Dr. Stefan Höning B 12 Institute for Biochemistry I Center for Molecular Medicine Cologne (CMMC) Medical Center, University of Cologne Joseph-Stelzmann-Str. 52 D Cologne Phone: Fax: [email protected] Prof. Dr. Uta C. Hoppe A 3 / A 5 Department III of Internal Medicine Center for Molecular Medicine Cologne (CMMC) Medical Center, University of Cologne Kerpener Str. 62 D Cologne Phone: Fax: [email protected] Prof. Dr. Andreas Jacobs C 5 Department of Neurology Center for Molecular Medicine Cologne (CMMC) Medical Center, University of Cologne Kerpener Str. 62 D Cologne Present address: Klinikum Fulda gag Institut für Neurologie, Pacelliallee 4, Fulda Phone: Fax: [email protected] Prof. Dr. Antonia Joussen A 4 Department of Ophthalmology Center for Molecular Medicine Cologne (CMMC) Medical Center, University of Cologne Kerpener Str. 62 D Cologne Present address: Universitätsklinikum Düsseldorf Augenklinik, Moorenstr. 5, Düsseldorf Phone: [email protected] Dr. Hamid Kashkar B 8 Institute for Medical Microbiology, Immunology and Hygiene Center for Molecular Medicine Cologne (CMMC) Medical Center, University of Cologne Goldenfelsstr D Cologne Phone: Fax: [email protected] Prof. Dr. Bernd Kirchhof A 4 Department of Ophthalmology Center for Molecular Medicine Cologne (CMMC) Medical Center, University of Cologne Kerpener Str. 62 D Cologne Phone: Fax: [email protected] 139
145 Addresses PD Dr. Jürgen-Christoph von Kleist-Retzow C 9 Department of Pediatrics Center for Molecular Medicine Cologne (CMMC) Medical Center, University of Cologne Kerpener Str. 62 D Cologne Phone: Fax: [email protected] Prof. Dr. Manuel Koch C 8 Institute for Biochemistry II Center for Molecular Medicine Cologne (CMMC) Medical Center, University of Cologne Joseph-Stelzmann-Str. 52 D Cologne Phone: Fax: [email protected] Prof. Dr. Dr. Thomas Krieg B 1 Department of Dermatology and Venerology Center for Molecular Medicine Cologne (CMMC) Medical Center, University of Cologne Kerpener Str. 62 D Cologne Phone: Fax: [email protected] Prof. Dr. Martin Krönke B 8 Institute for Medical Microbiology, Immunology and Hygiene Center for Molecular Medicine Cologne (CMMC) Medical Center, University of Cologne Goldenfelsstr D Cologne Phone: Fax: [email protected] Prof. Dr. Wilhelm Krone A 1 / C 1 Department II of Internal Medicine Center for Molecular Medicine Cologne (CMMC) Medical Center, University of Cologne Kerpener Str. 62 D Cologne Phone: Fax: [email protected] Wieslaw Krzyzak Microinjection-Laboratory Animal Facility Network Center for Molecular Medicine Cologne (CMMC) Joseph-Stelzmann-Str. 52 D Cologne Phone.: or 3887 Fax: [email protected] Prof. Dr. Christian Kubisch C 11 Institute of Human Genetics Center for Molecular Medicine Cologne (CMMC) Medical Center, University of Cologne Kerpener Str. 34 D Cologne Phone: Fax: [email protected] Gertrud Lang Secretary Animal Facility Network Center for Molecular Medicine Cologne (CMMC) Center for Physiology Robert-Koch-Str. 39 D Cologne Phone: Fax: [email protected] Prof. Dr. Thomas Langer C 6 Institute for Genetics Center for Molecular Medicine Cologne (CMMC) University of Cologne Zülpicher Str. 47 D Cologne Phone: Fax: [email protected] Prof. Dr. Dr. Cornelia Mauch B 1 Department of Dermatology and Venerology Center for Molecular Medicine Cologne (CMMC) Medical Center, University of Cologne Kerpener Str. 62 D Cologne Phone: Fax: [email protected] Dr. Catherin Niemann B / NG IV Hosted by: Institute for Pathology Center for Molecular Medicine Cologne (CMMC) Medical Center, University of Cologne Kerpener Str. 62 D Cologne Phone: Fax: [email protected] Dr. Carien Niessen B / NG I Hosted by: Department II of Internal Medicine, Department of Dermatology and Department of Surgery Center for Molecular Medicine Cologne (CMMC) Medical Center, University of Cologne Kerpener Str. 62 D Cologne Phone: Fax: [email protected] Prof. Dr. Angelika A. Noegel A 2 Institute for Biochemistry I Center for Molecular Medicine Cologne (CMMC) Medical Center, University of Cologne Joseph-Stelzmann-Str. 52 D Cologne Phone: Fax: [email protected] Prof. Dr. Peter Nürnberg B 11 Cologne Center for Genomics Center for Molecular Medicine Cologne (CMMC) Medical Center, University of Cologne Zülpicher Str. 47 D Cologne Phone: Fax: [email protected] Prof. Dr. Mats Paulsson B 2 Institute for Biochemistry II Center for Molecular Medicine Cologne (CMMC) Medical Center, University of Cologne Joseph-Stelzmann-Str. 52 D Cologne 140
146 Addresses Phone: Fax: Prof. Dr. Herbert Pfister B 3 Institute for Virology Center for Molecular Medicine Cologne (CMMC) Medical Center, University of Cologne Fürst-Pückler-Str. 56 D Cologne Phone: Fax: [email protected] Prof. Dr. Gabriele Pfitzer A 8 Institute for Vegetative Physiology Center for Molecular Medicine Cologne (CMMC) Medical Center, University of Cologne Robert-Koch-Str. 39 D Cologne Phone: Fax: [email protected] Dr. Markus Plomann C 7 Institute for Biochemistry II Center for Molecular Medicine Cologne (CMMC) Medical Center, University of Cologne Joseph-Stelzmann-Str. 52 D Cologne Phone: Fax: [email protected] Dr. Gerrit Praefcke B / NG VI Hosted by: Institute for Genetics Center for Molecular Medicine Cologne (CMMC) University of Cologne Zülpicher Str. 47 D Cologne Phone: Fax: [email protected] PD Dr. Ulrike Protzer B / NG-F Institute for Medical Microbiology, Immunology and Hygiene Center for Molecular Medicine Cologne (CMMC) Medical Center, University of Cologne Goldenfelsstr D Cologne Phone: Fax: [email protected] PD Dr. Francisco J. Rivero Crespo B 7 Institute for Biochemistry I Center for Molecular Medicine Cologne (CMMC) Medical Center, University of Cologne Joseph-Stelzmann-Str. 52 D Cologne Phone: Fax: [email protected] PD Dr. Stephan Rosenkranz A 6 Department of Internal Medicine III Center for Molecular Medicine Cologne (CMMC) Medical Center, University of Cologne Kerpener Str. 62 D Cologne Phone: Fax: [email protected] Prof. Dr. Rita Schmutzler B 9 Division of Molecular Gyneco-Oncology Department of Gynaecology and Obstetrics Center for Molecular Medicine Cologne (CMMC) Medical Center, University of Cologne Kerpener Str. 34 D Cologne Phone: Fax: [email protected] Prof. Dr. Toni Schneider C 4 Institute for Neurophysiology Center for Molecular Medicine Cologne (CMMC) Medical Center, University of Cologne Robert-Koch-Str. 39 D Cologne Phone: Fax: [email protected] Prof. Dr. Edgar Schömig A 7 Institute for Pharmacology Center for Molecular Medicine Cologne (CMMC) Medical Center, University of Cologne Gleueler Str. 24 D Cologne Phone: Fax: [email protected] Dr. Markus Schubert C 1 Department of Internal Medicine II Center for Molecular Medicine Cologne (CMMC) Medical Center, University of Cologne Kerpener Str. 62 D Cologne Phone: Fax: [email protected] Dr. Neil Smyth B 2 Institute for Biochemistry II Center for Molecular Medicine Cologne (CMMC) Medical Center, University of Cologne Joseph-Stelzmann-Str. 52 D Cologne Present address School of Biological Sciences University of Southampton Bassett Crescent East Southampton, SO16 7PX, UK Phone: Fax: [email protected] Dr. Robert Stehle A 8 Institute for Vegetative Physiology Center for Molecular Medicine Cologne (CMMC) Medical Center, University of Cologne Robert-Koch-Str. 39 D Cologne Phone: Fax: [email protected] Prof. Dr. Dr. Wilhelm Stoffel C 2 / C 3 Laboratory of Molecular Neuroscience Center for Molecular Medicine Cologne (CMMC) Medical Center, University of Cologne Joseph-Stelzmann-Str. 52 D Cologne 141
147 Addresses Photo Credits Phone: Fax: Dr. Bettina Wagner Animal Facility Network Center for Molecular Medicine Cologne (CMMC) Center for Physiology Robert-Koch-Str. 39 D Cologne Phone: Fax: Dr. Barbara Wappenschmidt B 9 Division of Molecular Gyneco-Oncology Department of Gynaecology and Obstetrics Center for Molecular Medicine Cologne (CMMC) Medical Center, University of Cologne Kerpener Str. 34 D Cologne Phone: Fax: [email protected] Introduction: Cornelius Evers Carsten Klett Annina Laufer Friedemann Reinhold Thies Schöning Shahrouz Yazdanyar Photo Art: Students, lectured by Shahrouz Yazdanyar, from the photography course of the Institute of Art Theory and Didactics of the Faculty of Education, University of Cologne (Jan Klein, Carsten Klett, Annina Laufer, Anika Sturm). Prof. Dr. Rudolf Wiesner C 9 Institute for Vegetative Physiology Center for Molecular Medicine Cologne (CMMC) Medical Center, University of Cologne Robert-Koch-Str. 39 D Cologne Phone: Fax: [email protected] Prof. Dr. Brunhilde Wirth C 10 Institute for Human Genetics Center for Molecular Medicine Cologne (CMMC) Medical Center, University of Cologne Kerpener Str. 34 D Cologne Phone: Fax: [email protected] Dr. Bernd Wollnik A / NG V Hosted by: Institute for Human Genetics Center for Molecular Medicine Cologne (CMMC) Medical Center, University of Cologne Kerpener Str. 34 D Cologne Phone: Fax: [email protected] 142
148 Contact Address Scientific Management Office Dr. Debora Grosskopf-Kroiher Scientific Coordination Phone: Fax: MTI-Building, Room 8a Center for Molecular Medicine Cologne (CMMC) Medical Center, University of Cologne Joseph-Stelzmann-Str Cologne, Germany Secretary Ute Fiehne-Reiß Phone: Fax: Susanne Haase Phone: Fax: Secretary - Interdisciplinary Postgraduate Program Molecular Medicine Ruth Fladerer Phone : +49 (0) Fax: +49 (0) [email protected] 143
Version 1 2015. Module guide. Preliminary document. International Master Program Cardiovascular Science University of Göttingen
 Version 1 2015 Module guide International Master Program Cardiovascular Science University of Göttingen Part 1 Theoretical modules Synopsis The Master program Cardiovascular Science contains four theoretical
Version 1 2015 Module guide International Master Program Cardiovascular Science University of Göttingen Part 1 Theoretical modules Synopsis The Master program Cardiovascular Science contains four theoretical
Graduate and Postdoctoral Affairs School of Biomedical Sciences College of Medicine. Graduate Certificate. Metabolic & Nutritional Medicine
 Graduate and Postdoctoral Affairs School of Biomedical Sciences College of Medicine Graduate Certificate in Metabolic & Nutritional Medicine Graduate Certificate Metabolic & Nutritional Medicine Purpose
Graduate and Postdoctoral Affairs School of Biomedical Sciences College of Medicine Graduate Certificate in Metabolic & Nutritional Medicine Graduate Certificate Metabolic & Nutritional Medicine Purpose
SACKLER SCHOOL OF GRADUATE BIOMEDICAL SCIENCES CATALOG 2015-2016 PROGRAMS OF STUDY, COURSES AND REQUIREMENTS FOR ALL GRADUATE PROGRAMS
 SACKLER SCHOOL OF GRADUATE BIOMEDICAL SCIENCES CATALOG 2015-2016 PROGRAMS OF STUDY, COURSES AND REQUIREMENTS FOR ALL GRADUATE PROGRAMS Graduate Programs CELL, MOLECULAR, AND DEVELOPMENTAL BIOLOGY CLINICAL
SACKLER SCHOOL OF GRADUATE BIOMEDICAL SCIENCES CATALOG 2015-2016 PROGRAMS OF STUDY, COURSES AND REQUIREMENTS FOR ALL GRADUATE PROGRAMS Graduate Programs CELL, MOLECULAR, AND DEVELOPMENTAL BIOLOGY CLINICAL
Course Curriculum for Master Degree in Medical Laboratory Sciences/Clinical Biochemistry
 Course Curriculum for Master Degree in Medical Laboratory Sciences/Clinical Biochemistry The Master Degree in Medical Laboratory Sciences /Clinical Biochemistry, is awarded by the Faculty of Graduate Studies
Course Curriculum for Master Degree in Medical Laboratory Sciences/Clinical Biochemistry The Master Degree in Medical Laboratory Sciences /Clinical Biochemistry, is awarded by the Faculty of Graduate Studies
PGY 206 ELEMENTARY PHYSIOLOGY. (3) An introductory survey course in basic human physiology. Prereq: One semester of college biology.
 206 ELEMENTARY PHYSIOLOGY. (3) An introductory survey course in basic human physiology. Prereq: One semester of college biology. 207 CASE STUDIES IN PHYSIOLOGY. (1) Group discussions of clinical cases
206 ELEMENTARY PHYSIOLOGY. (3) An introductory survey course in basic human physiology. Prereq: One semester of college biology. 207 CASE STUDIES IN PHYSIOLOGY. (1) Group discussions of clinical cases
Degree Programmes. International Graduate School in Molecular Medicine Ulm IGradU. Bachelor Master PhD/Dr. rer. nat.
 Degree Programmes Bachelor Master PhD/Dr. rer. nat. International Graduate School in Molecular Medicine Ulm IGradU Funded by the Excellence Initiative of the German federal and state governments www.uni-ulm.de/mm
Degree Programmes Bachelor Master PhD/Dr. rer. nat. International Graduate School in Molecular Medicine Ulm IGradU Funded by the Excellence Initiative of the German federal and state governments www.uni-ulm.de/mm
Course Curriculum for Master Degree in Medical Laboratory Sciences/Clinical Microbiology, Immunology and Serology
 Course Curriculum for Master Degree in Medical Laboratory Sciences/Clinical Microbiology, Immunology and Serology The Master Degree in Medical Laboratory Sciences / Clinical Microbiology, Immunology or
Course Curriculum for Master Degree in Medical Laboratory Sciences/Clinical Microbiology, Immunology and Serology The Master Degree in Medical Laboratory Sciences / Clinical Microbiology, Immunology or
FACULTY OF ALLIED HEALTH SCIENCES
 FACULTY OF ALLIED HEALTH SCIENCES 102 Naresuan University FACULTY OF ALLIED HEALTH SCIENCES has focused on providing strong professional programs, including Medical established as one of the leading institutes
FACULTY OF ALLIED HEALTH SCIENCES 102 Naresuan University FACULTY OF ALLIED HEALTH SCIENCES has focused on providing strong professional programs, including Medical established as one of the leading institutes
Gene Therapy. The use of DNA as a drug. Edited by Gavin Brooks. BPharm, PhD, MRPharmS (PP) Pharmaceutical Press
 Gene Therapy The use of DNA as a drug Edited by Gavin Brooks BPharm, PhD, MRPharmS (PP) Pharmaceutical Press Contents Preface xiii Acknowledgements xv About the editor xvi Contributors xvii An introduction
Gene Therapy The use of DNA as a drug Edited by Gavin Brooks BPharm, PhD, MRPharmS (PP) Pharmaceutical Press Contents Preface xiii Acknowledgements xv About the editor xvi Contributors xvii An introduction
Study Program Handbook Biochemistry and Cell Biology
 Study Program Handbook Biochemistry and Cell Biology Bachelor of Science Jacobs University Undergraduate Handbook BCCB - Matriculation Fall 2015 Page: ii Contents 1 The Biochemistry and Cell Biology (BCCB)
Study Program Handbook Biochemistry and Cell Biology Bachelor of Science Jacobs University Undergraduate Handbook BCCB - Matriculation Fall 2015 Page: ii Contents 1 The Biochemistry and Cell Biology (BCCB)
Support Program for Improving Graduate School Education Advanced Education Program for Integrated Clinical, Basic and Social Medicine
 Support Program for Improving Graduate School Education Advanced Education Program for Integrated Clinical, Basic and Social Medicine January 27, 2009 Dear Professors (representative) of departments, Subject:
Support Program for Improving Graduate School Education Advanced Education Program for Integrated Clinical, Basic and Social Medicine January 27, 2009 Dear Professors (representative) of departments, Subject:
Department of Food and Nutrition
 Department of Food and Nutrition Faculties Professors Lee-Kim, Yang Cha, Ph.D. (M.I.T., 1973) Nutritional biochemistry, Antioxidant vitamins, Fatty acid metabolism, Brain development, and Hyperlipidemia
Department of Food and Nutrition Faculties Professors Lee-Kim, Yang Cha, Ph.D. (M.I.T., 1973) Nutritional biochemistry, Antioxidant vitamins, Fatty acid metabolism, Brain development, and Hyperlipidemia
Intra- and interorgan communication of the cardiovascular system
 International Research Training Group 1902 Intra- and interorgan communication of the cardiovascular system Heinrich-Heine-Universität Düsseldorf, Germany University of Virginia, Charlottesville, USA Educational
International Research Training Group 1902 Intra- and interorgan communication of the cardiovascular system Heinrich-Heine-Universität Düsseldorf, Germany University of Virginia, Charlottesville, USA Educational
Graduate Studies in Biomedical Sciences
 Graduate Studies in Biomedical Sciences The graduate program in Biomedical Sciences is designed to provide a multidisciplinary educational and training environment that will prepare them for independent
Graduate Studies in Biomedical Sciences The graduate program in Biomedical Sciences is designed to provide a multidisciplinary educational and training environment that will prepare them for independent
M110.726 The Nucleus M110.727 The Cytoskeleton M340.703 Cell Structure and Dynamics
 of Biochemistry and Molecular Biology 1. Master the knowledge base of current biochemistry, molecular biology, and cellular physiology Describe current knowledge in metabolic transformations conducted
of Biochemistry and Molecular Biology 1. Master the knowledge base of current biochemistry, molecular biology, and cellular physiology Describe current knowledge in metabolic transformations conducted
MD/Ph.D. Degree Requirements
 COMBINED MD/Ph.D. DEGREE REQUIREMENTS MD/Ph.D. Program Eligibility Application to the Program General Requirements of the Program T he combined MD/Ph.D. degree program at the University of Toledo offers
COMBINED MD/Ph.D. DEGREE REQUIREMENTS MD/Ph.D. Program Eligibility Application to the Program General Requirements of the Program T he combined MD/Ph.D. degree program at the University of Toledo offers
Weill Graduate School of Medical Sciences of Cornell University Program in Pharmacology. Graduate School Curriculum
 Weill Graduate School of Medical Sciences of Cornell University Program in Pharmacology Graduate School Curriculum Weill Graduate School of Medical Sciences Partnership between Weill Medical College and
Weill Graduate School of Medical Sciences of Cornell University Program in Pharmacology Graduate School Curriculum Weill Graduate School of Medical Sciences Partnership between Weill Medical College and
Biostatistics Credit 15 5 10 1. Integrated and Interdisciplinary Training 2/7 Credit 24 24 1
 1st Year Hours SEMESTER I Exam or credit Total Lecture Seminar Classes ECTS Anatomy 1/2* Credit 100 20 80 9 Biophysics Exam 55 15 10 30 4 First Aid in Emergency Situations Credit 20 6 4 10 1 Histology
1st Year Hours SEMESTER I Exam or credit Total Lecture Seminar Classes ECTS Anatomy 1/2* Credit 100 20 80 9 Biophysics Exam 55 15 10 30 4 First Aid in Emergency Situations Credit 20 6 4 10 1 Histology
Master in Biology Faculty of Natural Sciences February 2010
 Master in Biology Faculty of Natural Sciences February 2010 University of Ulm There are many good reasons to pursue a master degree at the University of Ulm. One of the most important ones, alongside the
Master in Biology Faculty of Natural Sciences February 2010 University of Ulm There are many good reasons to pursue a master degree at the University of Ulm. One of the most important ones, alongside the
POSTGRADUATE CLINICAL TRAINING PROGRAMS BIOMEDICAL SCIENCES DIVISION GRADUATE PROGRAMS
 52 The student may select, with the approval of the Committee for the Clinical Component, another course among the primary areas of the Junior Internship, or in areas such as Surgery, Psychiatry, and Emergency
52 The student may select, with the approval of the Committee for the Clinical Component, another course among the primary areas of the Junior Internship, or in areas such as Surgery, Psychiatry, and Emergency
Master BioMedical Sciences (BMS) Track Cell Biology and Advanced Microscopy
 Master BioMedical Sciences (BMS) Track Cell Biology and Advanced Microscopy The five tracks offered in the Medical Biology cluster are: Biochemistry and Metabolic Diseases Cell Biology and Advanced Microscopy
Master BioMedical Sciences (BMS) Track Cell Biology and Advanced Microscopy The five tracks offered in the Medical Biology cluster are: Biochemistry and Metabolic Diseases Cell Biology and Advanced Microscopy
Human Health Sciences
 Human Health Sciences WITH PLYMOUTH UNIVERSITY DISCOVER MORE If you would like to visit Plymouth and meet our staff, then why not come along to one of our open days. Human Health Sciences WITH PLYMOUTH
Human Health Sciences WITH PLYMOUTH UNIVERSITY DISCOVER MORE If you would like to visit Plymouth and meet our staff, then why not come along to one of our open days. Human Health Sciences WITH PLYMOUTH
MOLECULAR PHARMACOLOGY AND EXPERIMENTAL THERAPEUTICS
 MOLECULAR PHARMACOLOGY AND EXPERIMENTAL THERAPEUTICS R. M. Weinshilboum, M.D., Program Director L. Wang, M.D., Ph.D., Program Co-Director D. C. Mays, Ph.D., Associate Program Director Ph.D. Degree Course
MOLECULAR PHARMACOLOGY AND EXPERIMENTAL THERAPEUTICS R. M. Weinshilboum, M.D., Program Director L. Wang, M.D., Ph.D., Program Co-Director D. C. Mays, Ph.D., Associate Program Director Ph.D. Degree Course
FACULTY OF MEDICAL SCIENCE
 Doctor of Philosophy Program in Microbiology FACULTY OF MEDICAL SCIENCE Naresuan University 171 Doctor of Philosophy Program in Microbiology The time is critical now for graduate education and research
Doctor of Philosophy Program in Microbiology FACULTY OF MEDICAL SCIENCE Naresuan University 171 Doctor of Philosophy Program in Microbiology The time is critical now for graduate education and research
ATIP Avenir Program 2014. Applicant s guide
 ATIP Avenir Program 2014 Applicant s guide Important dates: - November 29 th 2013: deadline for the online submission, the mailing of the hard copy of the scientific project, and the letters of recommendation
ATIP Avenir Program 2014 Applicant s guide Important dates: - November 29 th 2013: deadline for the online submission, the mailing of the hard copy of the scientific project, and the letters of recommendation
Course Curriculum for Master Degree in Clinical Pharmacy
 Course Curriculum for Master Degree in Clinical Pharmacy The Master Degree in Clinical Pharmacy is awarded by the Faculty of Graduate studies at Jordan University of Science and Technology (JUST) upon
Course Curriculum for Master Degree in Clinical Pharmacy The Master Degree in Clinical Pharmacy is awarded by the Faculty of Graduate studies at Jordan University of Science and Technology (JUST) upon
UPBM CURRICULAR BROCHURE
 UPBM CURRICULAR BROCHURE Undergraduate Program in Biology and Medicine Contents Academic Year 2015-16 About the Undergraduate Program in Biology and Medicine...pg. 1 Undergraduate Majors...pg. 2-3 Getting
UPBM CURRICULAR BROCHURE Undergraduate Program in Biology and Medicine Contents Academic Year 2015-16 About the Undergraduate Program in Biology and Medicine...pg. 1 Undergraduate Majors...pg. 2-3 Getting
How To Identify A Novel Pathway Leading To Myocardial Infarction
 Press Release Embargo: 10 November 2013 at 1800 London time / 1300 US Eastern Time Genetic research identifies novel pathway leading to myocardial infarction Starting with a severely affected family, a
Press Release Embargo: 10 November 2013 at 1800 London time / 1300 US Eastern Time Genetic research identifies novel pathway leading to myocardial infarction Starting with a severely affected family, a
The Evolution of Graduate Education in the College of Medicine. Office of Graduate & Postdoctoral Affairs School of Basic Biomedical Sciences
 The Evolution of Graduate Education in the College of Medicine Office of Graduate & Postdoctoral Affairs Graduate Education in the College of Medicine Graduate Programs in Integrated Graduate Education
The Evolution of Graduate Education in the College of Medicine Office of Graduate & Postdoctoral Affairs Graduate Education in the College of Medicine Graduate Programs in Integrated Graduate Education
Admissions...713/500-5116 Alumni Relations...713/500-5065 Continuing Medical Education...713/500-5249 Educational Programs...
 fact book The University of Texas Health Science Center at Houston in the Texas Medic I-45 59 Lyndon B. Johnson Gen Hospital I-10 I-10 Rice University Campus Downtown Houston 59 Texas Medical Center I-45
fact book The University of Texas Health Science Center at Houston in the Texas Medic I-45 59 Lyndon B. Johnson Gen Hospital I-10 I-10 Rice University Campus Downtown Houston 59 Texas Medical Center I-45
POLICIES AND CUSTOMS Ph.D. PROGRAM DEPARTMENT OF MOLECULAR & CELLULAR BIOCHEMISTRY UNIVERSITY OF KENTUCKY
 - 1 - POLICIES AND CUSTOMS Ph.D. PROGRAM DEPARTMENT OF MOLECULAR & CELLULAR BIOCHEMISTRY UNIVERSITY OF KENTUCKY Revised: March 2010 The following is intended to serve as a guide for graduate students in
- 1 - POLICIES AND CUSTOMS Ph.D. PROGRAM DEPARTMENT OF MOLECULAR & CELLULAR BIOCHEMISTRY UNIVERSITY OF KENTUCKY Revised: March 2010 The following is intended to serve as a guide for graduate students in
FACULTY OF MEDICAL SCIENCE
 Doctor of Philosophy in Biochemistry FACULTY OF MEDICAL SCIENCE Naresuan University 73 Doctor of Philosophy in Biochemistry The Biochemistry Department at Naresuan University is a leader in lower northern
Doctor of Philosophy in Biochemistry FACULTY OF MEDICAL SCIENCE Naresuan University 73 Doctor of Philosophy in Biochemistry The Biochemistry Department at Naresuan University is a leader in lower northern
ANATOMY & PHYSIOLOGY MODULE 2015/16
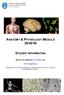 ANATOMY & PHYSIOLOGY MODULE 2015/16 STUDENT INFORMATION MODULE CO-ORDINATOR: Email: [email protected] DEPARTMENT OF PHYSIOLOGY, BIOMEDICAL SCIENCES INSTITUTE, TRINITY COLLEGE, PEARSE STREET, DUBLIN 2. Module
ANATOMY & PHYSIOLOGY MODULE 2015/16 STUDENT INFORMATION MODULE CO-ORDINATOR: Email: [email protected] DEPARTMENT OF PHYSIOLOGY, BIOMEDICAL SCIENCES INSTITUTE, TRINITY COLLEGE, PEARSE STREET, DUBLIN 2. Module
University of Oklahoma Health Sciences Center Graduate Program in Biomedical Sciences (GPiBS) Summer Undergraduate Research Programs (SURP)
 University of Oklahoma Health Sciences Center Graduate Program in Biomedical Sciences (GPiBS) Summer Undergraduate Research Programs (SURP) Katie Hudson Ph.D., Assistant Director of GPiBS and SURP My Path
University of Oklahoma Health Sciences Center Graduate Program in Biomedical Sciences (GPiBS) Summer Undergraduate Research Programs (SURP) Katie Hudson Ph.D., Assistant Director of GPiBS and SURP My Path
1. Program Title Master of Science Program in Biochemistry (International Program)
 1 Program Structure and Specification Master of Science Program in Biochemistry (International Program) Curriculum Last Revised in 2012 for Students Entering in Academic Year 2016 -----------------------------------------
1 Program Structure and Specification Master of Science Program in Biochemistry (International Program) Curriculum Last Revised in 2012 for Students Entering in Academic Year 2016 -----------------------------------------
The Ohio State University College of Medicine. Biomedical Sciences Graduate Program (BSGP) The Biology of Human Disease. medicine.osu.
 (BSGP) The Biology of Human Disease medicine.osu.edu/bsgp Welcome from BSGP Leadership Thank you for your interest in the at The Ohio State University Wexner Medical Center. Our goal is to train talented,
(BSGP) The Biology of Human Disease medicine.osu.edu/bsgp Welcome from BSGP Leadership Thank you for your interest in the at The Ohio State University Wexner Medical Center. Our goal is to train talented,
Never Stand Stil Faculties of Science and Medicine
 Medical Sciences Never Stand Still Faculties of Science and Medicine School of Medical Sciences The School of Medical Sciences provides teaching in four major disciplines within the health sciences - Anatomy,
Medical Sciences Never Stand Still Faculties of Science and Medicine School of Medical Sciences The School of Medical Sciences provides teaching in four major disciplines within the health sciences - Anatomy,
STEM CELL FELLOWSHIP
 Module I: The Basic Principles of Stem Cells 1. Basics of Stem Cells a. Understanding the development of embryonic stem cells i. Embryonic stem cells ii. Embryonic germ cells iii. Differentiated stem cell
Module I: The Basic Principles of Stem Cells 1. Basics of Stem Cells a. Understanding the development of embryonic stem cells i. Embryonic stem cells ii. Embryonic germ cells iii. Differentiated stem cell
Your Life Your Health Cariodmetabolic Risk Syndrome Part VII Inflammation chronic, low-grade By James L. Holly, MD The Examiner January 25, 2007
 Your Life Your Health Cariodmetabolic Risk Syndrome Part VII Inflammation chronic, low-grade By James L. Holly, MD The Examiner January 25, 2007 The cardiometabolic risk syndrome is increasingly recognized
Your Life Your Health Cariodmetabolic Risk Syndrome Part VII Inflammation chronic, low-grade By James L. Holly, MD The Examiner January 25, 2007 The cardiometabolic risk syndrome is increasingly recognized
BSc in Medical Sciences with PHARMACOLOGY
 BSc in Medical Sciences with PHARMACOLOGY Course Director Dr Christopher John Module Leaders Dr Robert Dickinson (Module 1) Dr Anabel Varela Carver (Module 2) Dr Sohag Saleh (Module 3) Course Administrator
BSc in Medical Sciences with PHARMACOLOGY Course Director Dr Christopher John Module Leaders Dr Robert Dickinson (Module 1) Dr Anabel Varela Carver (Module 2) Dr Sohag Saleh (Module 3) Course Administrator
PH.D. PROGRAM IN BIOMEDICAL NEUROSCIENCE
 PH.D. PROGRAM IN BIOMEDICAL NEUROSCIENCE Department of Neuroscience, Temple University School of Medicine www.temple.edu/medicine/biomedical_neuroscience [email protected] PROGRAM OVERVIEW ABOUT THE
PH.D. PROGRAM IN BIOMEDICAL NEUROSCIENCE Department of Neuroscience, Temple University School of Medicine www.temple.edu/medicine/biomedical_neuroscience [email protected] PROGRAM OVERVIEW ABOUT THE
Summary of Discussion on Non-clinical Pharmacology Studies on Anticancer Drugs
 Provisional Translation (as of January 27, 2014)* November 15, 2013 Pharmaceuticals and Bio-products Subcommittees, Science Board Summary of Discussion on Non-clinical Pharmacology Studies on Anticancer
Provisional Translation (as of January 27, 2014)* November 15, 2013 Pharmaceuticals and Bio-products Subcommittees, Science Board Summary of Discussion on Non-clinical Pharmacology Studies on Anticancer
Course Curriculum for Master Degree in Food Hygiene /Faculty of Veterinary Medicine
 Course Curriculum for Master Degree in Food Hygiene /Faculty of Veterinary Medicine The Master Degree Food Hygiene /Veterinary Medicine is awarded by the Faculty of Graduate Studies at Jordan University
Course Curriculum for Master Degree in Food Hygiene /Faculty of Veterinary Medicine The Master Degree Food Hygiene /Veterinary Medicine is awarded by the Faculty of Graduate Studies at Jordan University
MD-PhD: Is it Right for Me? Communications Committee of the MD-PhD Section of the Group of Research, Education, and Training (GREAT) AAMC
 2014 MD-PhD: Is it Right for Me? Communications Committee of the MD-PhD Section of the Group of Research, Education, and Training (GREAT) AAMC Biomedical Scientists: Multiple Pathways MD graduates pursue
2014 MD-PhD: Is it Right for Me? Communications Committee of the MD-PhD Section of the Group of Research, Education, and Training (GREAT) AAMC Biomedical Scientists: Multiple Pathways MD graduates pursue
Stem Cell Quick Guide: Stem Cell Basics
 Stem Cell Quick Guide: Stem Cell Basics What is a Stem Cell? Stem cells are the starting point from which the rest of the body grows. The adult human body is made up of hundreds of millions of different
Stem Cell Quick Guide: Stem Cell Basics What is a Stem Cell? Stem cells are the starting point from which the rest of the body grows. The adult human body is made up of hundreds of millions of different
Curriculum Overview:
 Curriculum Overview: The following courses must be completed by all Certified Nutrition Specialist candidates. Courses cannot fulfill multiple curriculum requirements, however, excess nutrition science
Curriculum Overview: The following courses must be completed by all Certified Nutrition Specialist candidates. Courses cannot fulfill multiple curriculum requirements, however, excess nutrition science
Graduate School of Excellence Exzellenz-Graduiertenschule
 Jena School for Microbial Communication Graduate School of Excellence Exzellenz-Graduiertenschule Microbial Communication Talking Microbes? Microorganisms are present everywhere. They are often considered
Jena School for Microbial Communication Graduate School of Excellence Exzellenz-Graduiertenschule Microbial Communication Talking Microbes? Microorganisms are present everywhere. They are often considered
GRADUATE EDUCATION DEPARTMENT OF INTEGRATIVE PHYSIOLOGY UNIVERSITY OF NORTH TEXAS HEALTH SCIENCE CENTER AT FORT WORTH POLICIES AND GUIDELINES
 1 GRADUATE EDUCATION DEPARTMENT OF INTEGRATIVE PHYSIOLOGY UNIVERSITY OF NORTH TEXAS HEALTH SCIENCE CENTER AT FORT WORTH POLICIES AND GUIDELINES Revised August, 2005 Course Numbers Edited August, 2009 2
1 GRADUATE EDUCATION DEPARTMENT OF INTEGRATIVE PHYSIOLOGY UNIVERSITY OF NORTH TEXAS HEALTH SCIENCE CENTER AT FORT WORTH POLICIES AND GUIDELINES Revised August, 2005 Course Numbers Edited August, 2009 2
UMDNJ New Jersey Medical School
 UMDNJ New Jersey Medical School NJMS History: Founded in 1954 as the Seton Hall College of Medicine and Dentistry, New Jersey Medical School is the state s oldest medical school. 1956 First class of 80
UMDNJ New Jersey Medical School NJMS History: Founded in 1954 as the Seton Hall College of Medicine and Dentistry, New Jersey Medical School is the state s oldest medical school. 1956 First class of 80
Medical Science Building, Rm C696 185 South Orange Ave. Newark, New Jersey 07101. www.umdnj.edu/gsbsnweb
 Medical Science Building, Rm C696 185 South Orange Ave. Newark, New Jersey 07101 Newark Division www.umdnj.edu/gsbsnweb University of Medicine & Dentistry of New Jersey Graduate School of Biomedical Sciences
Medical Science Building, Rm C696 185 South Orange Ave. Newark, New Jersey 07101 Newark Division www.umdnj.edu/gsbsnweb University of Medicine & Dentistry of New Jersey Graduate School of Biomedical Sciences
FACULTY OF MEDICINE - UNIVERSITY OF LISBON
 Egas Moniz Cerebral Arteriography (1927) Pre-frontal Leucotomy (1933) Nobel Prize (1949) Reynaldo dos Santos Translumbar Aortography (1929) J. Cid dos Santos Phlebography (1937) Endarterectomy (1946) Strengthen
Egas Moniz Cerebral Arteriography (1927) Pre-frontal Leucotomy (1933) Nobel Prize (1949) Reynaldo dos Santos Translumbar Aortography (1929) J. Cid dos Santos Phlebography (1937) Endarterectomy (1946) Strengthen
Graduate Certificate Pre-Med Program Course Descriptions For Year 2015-2016 FALL
 Graduate Certificate Pre-Med Program Course Descriptions For Year 2015-2016 FALL COURSE TITLE: BIOCHEMISTRY COURSE NUMBER: 5104 This course emphasizes biochemical compounds, processes and systems, designed
Graduate Certificate Pre-Med Program Course Descriptions For Year 2015-2016 FALL COURSE TITLE: BIOCHEMISTRY COURSE NUMBER: 5104 This course emphasizes biochemical compounds, processes and systems, designed
A Career in Pediatric Hematology-Oncology? Think About It...
 A Career in Pediatric Hematology-Oncology? Think About It... What does a pediatric hematologist-oncologist do? What kind of training is necessary? Is there a future need for specialists in this area? T
A Career in Pediatric Hematology-Oncology? Think About It... What does a pediatric hematologist-oncologist do? What kind of training is necessary? Is there a future need for specialists in this area? T
Future Oncology: Technology, Products, Market and Service Opportunities
 Brochure More information from http://www.researchandmarkets.com/reports/296370/ Future Oncology: Technology, Products, Market and Service Opportunities Description: Future Oncology is an analytical newsletter
Brochure More information from http://www.researchandmarkets.com/reports/296370/ Future Oncology: Technology, Products, Market and Service Opportunities Description: Future Oncology is an analytical newsletter
Pharmacology Curriculum Transition. 1990-Present
 Pharmacology Curriculum Transition 1990-Present Curriculum ~1990 Introductory Biochemistry Biology of Bacteria and Mammalian Cells Human Physiology General and Special Pharmacology Advanced Pharmacology
Pharmacology Curriculum Transition 1990-Present Curriculum ~1990 Introductory Biochemistry Biology of Bacteria and Mammalian Cells Human Physiology General and Special Pharmacology Advanced Pharmacology
PHYSIOLOGY. THE STUDY OF LIFE, and how genes, cells, tissues, and organisms function.
 PHYSIOLOGY THE STUDY OF LIFE, and how genes, cells, tissues, and organisms function. What is PHYSIOLOGY? Physiologists teach and mentor students in both the classroom and laboratory. Physiologists apply
PHYSIOLOGY THE STUDY OF LIFE, and how genes, cells, tissues, and organisms function. What is PHYSIOLOGY? Physiologists teach and mentor students in both the classroom and laboratory. Physiologists apply
ITT Advanced Medical Technologies - A Programmer's Overview
 ITT Advanced Medical Technologies (Ileri Tip Teknolojileri) ITT Advanced Medical Technologies (Ileri Tip Teknolojileri) is a biotechnology company (SME) established in Turkey. Its activity area is research,
ITT Advanced Medical Technologies (Ileri Tip Teknolojileri) ITT Advanced Medical Technologies (Ileri Tip Teknolojileri) is a biotechnology company (SME) established in Turkey. Its activity area is research,
A Genetic Analysis of Rheumatoid Arthritis
 A Genetic Analysis of Rheumatoid Arthritis Introduction to Rheumatoid Arthritis: Classification and Diagnosis Rheumatoid arthritis is a chronic inflammatory disorder that affects mainly synovial joints.
A Genetic Analysis of Rheumatoid Arthritis Introduction to Rheumatoid Arthritis: Classification and Diagnosis Rheumatoid arthritis is a chronic inflammatory disorder that affects mainly synovial joints.
The National Centre for Biomedical Engineering Science
 The How this Integrated Technology Platform has impacted the development of R&D Clusters in Galway Jacinta Thornton, PhD Executive Manager, NCBES National Centre for Biomedical Engineering g Science Mission
The How this Integrated Technology Platform has impacted the development of R&D Clusters in Galway Jacinta Thornton, PhD Executive Manager, NCBES National Centre for Biomedical Engineering g Science Mission
Public Health and. Interdisciplinary Context. Birgitta Edlund RNT
 1 Public Health and Caring Science Nursing Education in an Extended Interdisciplinary Context Birgitta Edlund RNT Associate Professor 2 Uppsala 3 University Main Building 4 Uppsala University Research
1 Public Health and Caring Science Nursing Education in an Extended Interdisciplinary Context Birgitta Edlund RNT Associate Professor 2 Uppsala 3 University Main Building 4 Uppsala University Research
Master of Physician Assistant Studies Course Descriptions for Year I
 FALL TERM COURSES: Master of Physician Assistant Studies Course Descriptions for Year I PHAC 7230 Fundamentals in Pharmacology for Health Care I Credit Hrs: 3 This course will build on foundational knowledge
FALL TERM COURSES: Master of Physician Assistant Studies Course Descriptions for Year I PHAC 7230 Fundamentals in Pharmacology for Health Care I Credit Hrs: 3 This course will build on foundational knowledge
Ph.D. in Molecular Medicine
 Ph.D. in Molecular Medicine and Translational Research College of Medicine 1. Introduction: The College of Medicine, while consolidating on its undergraduate innovative educational programs, decided to
Ph.D. in Molecular Medicine and Translational Research College of Medicine 1. Introduction: The College of Medicine, while consolidating on its undergraduate innovative educational programs, decided to
Summer School 2011 Berlin SFB Transregio 19
 Summer School 2011 Berlin SFB Transregio 19 27th October 29th October 2011 Molecular Mechanisms and Consequences of Inflammatory Cardiomyopathy: State of the Art and Future Challenges Welcome Address
Summer School 2011 Berlin SFB Transregio 19 27th October 29th October 2011 Molecular Mechanisms and Consequences of Inflammatory Cardiomyopathy: State of the Art and Future Challenges Welcome Address
Course Curriculum for Master Degree in Veterinary Epidemiology/Faculty of Veterinary Medicine
 Course Curriculum for Master Degree in Veterinary Epidemiology/Faculty of Veterinary Medicine The Master Degree Veterinary Epidemiology/ Faculty of Veterinary Medicine is awarded by the Faculty of Graduate
Course Curriculum for Master Degree in Veterinary Epidemiology/Faculty of Veterinary Medicine The Master Degree Veterinary Epidemiology/ Faculty of Veterinary Medicine is awarded by the Faculty of Graduate
Master of Science in Biomedical Sciences
 Master of Science in Biomedical Sciences Faculty of Medicine Good health is our greatest treasure. Understanding the human body, healthy and diseased, is the stepping stone to finding tools to improving
Master of Science in Biomedical Sciences Faculty of Medicine Good health is our greatest treasure. Understanding the human body, healthy and diseased, is the stepping stone to finding tools to improving
Positions Available in SINAPSE INSTITUTE in Singapore
 Positions Available in SINAPSE INSTITUTE in Singapore The SINAPSE Institute (www.sinapseinstitute.org), under the direction of Prof. Nitish Thakor (www.jhu.edu/nthakor), is a newly launched research institute
Positions Available in SINAPSE INSTITUTE in Singapore The SINAPSE Institute (www.sinapseinstitute.org), under the direction of Prof. Nitish Thakor (www.jhu.edu/nthakor), is a newly launched research institute
Biochemistry. Entrance Requirements. Requirements for Honours Programs. 148 Bishop s University 2015/2016
 148 Bishop s University 2015/2016 Biochemistry The Biochemistry program at Bishop s is coordinated through an interdisciplinary committee of chemists, biochemists and biologists, providing students with
148 Bishop s University 2015/2016 Biochemistry The Biochemistry program at Bishop s is coordinated through an interdisciplinary committee of chemists, biochemists and biologists, providing students with
Faculty of Applied Sciences. Bachelor s degree programme. Nanobiology. Integrating Physics with Biomedicine
 Faculty of Applied Sciences Bachelor s degree programme Nanobiology Integrating Physics with Biomedicine We have just begun to explore life on this level Nanobiology uses the language of maths in the context
Faculty of Applied Sciences Bachelor s degree programme Nanobiology Integrating Physics with Biomedicine We have just begun to explore life on this level Nanobiology uses the language of maths in the context
University of Medicine and Dentistry of New Jersey (UMDNJ)
 University of Medicine and Dentistry of New Jersey (UMDNJ) Dual-Degree Program between the UMDNJ Graduate School of Biomedical Sciences (GSBS) And the UMDNJ School of Public Health (SPH) Leading to the:
University of Medicine and Dentistry of New Jersey (UMDNJ) Dual-Degree Program between the UMDNJ Graduate School of Biomedical Sciences (GSBS) And the UMDNJ School of Public Health (SPH) Leading to the:
Course Curriculum for Master Degree in Medical Laboratory Sciences/Hematology and Blood Banking
 Course Curriculum for Master Degree in Medical Laboratory Sciences/Hematology and Blood Banking The Master Degree in Medical Laboratory Sciences /Hematology & Blood Banking, is awarded by the Faculty of
Course Curriculum for Master Degree in Medical Laboratory Sciences/Hematology and Blood Banking The Master Degree in Medical Laboratory Sciences /Hematology & Blood Banking, is awarded by the Faculty of
Diabetes and Insulin Signaling
 Diabetes and Insulin Signaling NATIONAL CENTER FOR CASE STUDY TEACHING IN SCIENCE by Kristy J. Wilson School of Mathematics and Sciences Marian University, Indianapolis, IN Part I Research Orientation
Diabetes and Insulin Signaling NATIONAL CENTER FOR CASE STUDY TEACHING IN SCIENCE by Kristy J. Wilson School of Mathematics and Sciences Marian University, Indianapolis, IN Part I Research Orientation
MD-PhD: Is it Right for Me? Training & Career Paths
 MD-PhD: Is it Right for Me? Training & Career Paths What is a physician-scientist? Men and women who are physicians and investigators (and mentors and teachers and inventors and ) Spend most of their professional
MD-PhD: Is it Right for Me? Training & Career Paths What is a physician-scientist? Men and women who are physicians and investigators (and mentors and teachers and inventors and ) Spend most of their professional
October 17, 2005. Elias Zerhouni, M.D. Director National Institutes of Health One Center Drive Suite 126 MSC 0148 Bethesda, MD 20892
 October 17, 2005 Elias Zerhouni, M.D. Director National Institutes of Health One Center Drive Suite 126 MSC 0148 Bethesda, MD 20892 Dear Dr. Zerhouni: The undersigned nonprofit medical and scientific societies
October 17, 2005 Elias Zerhouni, M.D. Director National Institutes of Health One Center Drive Suite 126 MSC 0148 Bethesda, MD 20892 Dear Dr. Zerhouni: The undersigned nonprofit medical and scientific societies
MSBS Degree Requirements
 MASTER OF SCIENCE IN BIOMEDICAL SCIENCES DEGREE REQUIREMENTS T he degree of Master of Science in Biomedical Sciences is offered in various areas of concentration, both departmental and interdisciplinary;
MASTER OF SCIENCE IN BIOMEDICAL SCIENCES DEGREE REQUIREMENTS T he degree of Master of Science in Biomedical Sciences is offered in various areas of concentration, both departmental and interdisciplinary;
The College of Liberal Arts and Sciences
 The College of Liberal Arts and Sciences Proposal for a Biomedical Sciences Major (BS) Professor Bernd Fritzsch, DEO, Department of Biology Associate Professor Bryant McAllister, DUS, Department of Biology
The College of Liberal Arts and Sciences Proposal for a Biomedical Sciences Major (BS) Professor Bernd Fritzsch, DEO, Department of Biology Associate Professor Bryant McAllister, DUS, Department of Biology
The Physiology Undergraduate Major in the University of Arizona College of Medicine Tucson, AZ
 The Physiology Undergraduate Major in the University of Arizona College of Medicine Tucson, AZ Erik J. Henriksen, Ph.D. Professor Director, Physiology Undergraduate Program Department of Physiology [email protected]
The Physiology Undergraduate Major in the University of Arizona College of Medicine Tucson, AZ Erik J. Henriksen, Ph.D. Professor Director, Physiology Undergraduate Program Department of Physiology [email protected]
1. The student shall be a student enrolled at McMurry University
 3 + 4 DUAL DEGREE AND DENTAL EARLY ADMISSION PROGRAM (DEAP) JOINT AGREEMENT BETWEEN McMURRY UNIVERSITY AND THE UNIVERSITY OF TEXAS HEALTH SCIENCE CENTER AT SAN ANTONIO DENTAL SCHOOL PURPOSE OF THE PROGRAM
3 + 4 DUAL DEGREE AND DENTAL EARLY ADMISSION PROGRAM (DEAP) JOINT AGREEMENT BETWEEN McMURRY UNIVERSITY AND THE UNIVERSITY OF TEXAS HEALTH SCIENCE CENTER AT SAN ANTONIO DENTAL SCHOOL PURPOSE OF THE PROGRAM
Program of Study: Bachelor of Science in Athletic Training
 Program of Study: Bachelor of Science Training Program Description Athletic training, as defined by the National Athletic Trainer s Association, is practiced by athletic trainers, health care professionals
Program of Study: Bachelor of Science Training Program Description Athletic training, as defined by the National Athletic Trainer s Association, is practiced by athletic trainers, health care professionals
Compartmentalization of the Cell. Objectives. Recommended Reading. Professor Alfred Cuschieri. Department of Anatomy University of Malta
 Compartmentalization of the Cell Professor Alfred Cuschieri Department of Anatomy University of Malta Objectives By the end of this session the student should be able to: 1. Identify the different organelles
Compartmentalization of the Cell Professor Alfred Cuschieri Department of Anatomy University of Malta Objectives By the end of this session the student should be able to: 1. Identify the different organelles
ALPHA (TNFa) IN OBESITY
 THE ROLE OF TUMOUR NECROSIS FACTOR ALPHA (TNFa) IN OBESITY Alison Mary Morris, B.Sc (Hons) A thesis submitted to Adelaide University for the degree of Doctor of Philosophy Department of Physiology Adelaide
THE ROLE OF TUMOUR NECROSIS FACTOR ALPHA (TNFa) IN OBESITY Alison Mary Morris, B.Sc (Hons) A thesis submitted to Adelaide University for the degree of Doctor of Philosophy Department of Physiology Adelaide
The Genetic Basis of Neurological Disorders
 The Genetic Basis of Neurological Disorders A complete advanced undergraduate/graduate course with: 22 online lectures by leading authorities Resources for workshops, tutorials, journal clubs, projects
The Genetic Basis of Neurological Disorders A complete advanced undergraduate/graduate course with: 22 online lectures by leading authorities Resources for workshops, tutorials, journal clubs, projects
Implementing Structured Doctoral Programmes at Faculty-Level: Benefits, Challenges, Perspectives
 EUA Workshop, London, 11-12 November 2005 Implementing Structured Doctoral Programmes at Faculty-Level: Benefits, Challenges, Perspectives Thomas Koch, Scientific Coordinator Graduate School of Chemistry
EUA Workshop, London, 11-12 November 2005 Implementing Structured Doctoral Programmes at Faculty-Level: Benefits, Challenges, Perspectives Thomas Koch, Scientific Coordinator Graduate School of Chemistry
The National Institute of Genomic Medicine (INMEGEN) was
 Genome is...... the complete set of genetic information contained within all of the chromosomes of an organism. It defines the particular phenotype of an individual. What is Genomics? The study of the
Genome is...... the complete set of genetic information contained within all of the chromosomes of an organism. It defines the particular phenotype of an individual. What is Genomics? The study of the
Programme Specification (Undergraduate) Date amended: August 2012
 Programme Specification (Undergraduate) Date amended: August 2012 1. Programme Title(s) and UCAS code(s): BSc Biological Sciences C100 BSc Biological Sciences (Biochemistry) C700 BSc Biological Sciences
Programme Specification (Undergraduate) Date amended: August 2012 1. Programme Title(s) and UCAS code(s): BSc Biological Sciences C100 BSc Biological Sciences (Biochemistry) C700 BSc Biological Sciences
College of Pharmacy & Pharmaceutical Sciences Graduate Catalog 2015-2016
 College of Pharmacy & Pharmaceutical Sciences Graduate Catalog 2015-2016 Degrees Offered Doctor of Philosophy in Experimental Therapeutics Doctor of Philosophy in Medicinal Chemistry Master of Science
College of Pharmacy & Pharmaceutical Sciences Graduate Catalog 2015-2016 Degrees Offered Doctor of Philosophy in Experimental Therapeutics Doctor of Philosophy in Medicinal Chemistry Master of Science
Molecular Biology And Biotechnology
 in Molecular Biology And Biotechnology National Institute of Molecular Biology and Biotechnology College of Science University of the Philippines Diliman, Quezon City 1101 National Institute of Molecular
in Molecular Biology And Biotechnology National Institute of Molecular Biology and Biotechnology College of Science University of the Philippines Diliman, Quezon City 1101 National Institute of Molecular
Curriculum for Structured Doctoral Candidate Training at the Heinrich Pette Institute Leibniz Institute for Experimental Virology
 Curriculum for Structured Doctoral Candidate Training at the Heinrich Pette Institute Leibniz Institute for Experimental Virology Please choose lectures and courses according to your interests and requirements.
Curriculum for Structured Doctoral Candidate Training at the Heinrich Pette Institute Leibniz Institute for Experimental Virology Please choose lectures and courses according to your interests and requirements.
Biochemistry Major Talk 2014-15. Welcome!!!!!!!!!!!!!!
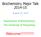 Biochemistry Major Talk 2014-15 August 14, 2015 Department of Biochemistry The University of Hong Kong Welcome!!!!!!!!!!!!!! Introduction to Biochemistry A four-minute video: http://www.youtube.com/watch?v=tpbamzq_pue&l
Biochemistry Major Talk 2014-15 August 14, 2015 Department of Biochemistry The University of Hong Kong Welcome!!!!!!!!!!!!!! Introduction to Biochemistry A four-minute video: http://www.youtube.com/watch?v=tpbamzq_pue&l
Masters Learning mode (Форма обучения)
 Program Title (Название программы): Pharmacology Degree (Степень) Masters Learning mode (Форма обучения) Full-time and part-time Duration of study (Продолжительность программы) 2 years (4 years part time)
Program Title (Название программы): Pharmacology Degree (Степень) Masters Learning mode (Форма обучения) Full-time and part-time Duration of study (Продолжительность программы) 2 years (4 years part time)
