PharmaSUG 2015 Paper SS10-SAS
|
|
|
- Hubert Cole
- 8 years ago
- Views:
Transcription
1 ABSTRACT PharmaSUG 2015 Paper SS10-SAS Using SAS Clinical Data Integration to Roundtrip a Complete Study Study Metadata (Define-XML) and Study Data (Dataset-XML) Ken Ellis, SAS Institute Inc., Cary, NC, USA SAS Clinical Data Integration 2.6 now supports the complete CDISC Define-XML 2.0 specification for both import from, and creation of define.xml files. Support for value-level metadata and supplemental documents, as well as support for the new CDISC Dataset-XML specification have been added and give the user a powerful means by which to import and create complete study definitions. Topics covered include: import of CDISC Define-XML file metadata, including domains, value-level metadata, supplemental documents, computational algorithms and code lists import of accompanying domain data from CDISC Dataset-XML files creation of a complete CDISC Define-XML file including all imported metadata (roundtrip) creation of CDISC Dataset-XML files from the imported domain tables (roundtrip) INTRODUCTION The CDISC Define-XML specification (define.xml) provides a machine readable description of datasets used for submission to the FDA. The new CDISC Dataset-XML 2 specification (dataset-xml) provides a means for describing the underlying data associated with CDISC Define-XML 2.0 compliant dataset definitions. SAS Clinical Data Integration 2.6 3, in combination with SAS Clinical Standards Toolkit 1.7 4, provides a rich set of tools for reading and writing both define.xml and dataset-xml files. This paper will focus on those tools and the processes that facilitate working with these files. A sample work flow will be developed that roundtrips a define.xml file and its associated dataset-xml files. It is assumed that the user has some familiarity with SAS Clinical Data Integration and the CDISC specifications referenced as in-depth discussions of each is beyond the scope of this paper. (See the References section for more information.)
2 THE TOOLS SAS Clinical Data Integration provides the following tools for reading and writing define.xml and dataset.xml files. We will look at them more in-depth as we encounter them in our workflow example: 1. Study Level a. Study from Define... Creates a basic study shell using the selected folder template. Values for the study name, description and protocol title are derived from the define.xml file. The user can optionally associate a data standard and library with the new study. b. Domain(s) from Define Creates empty SAS table shells for each SDTM domain selected for import. All table level and column metadata are derived from the define.xml file. c. Analysis Dataset(s) from Define Creates empty SAS table shells for each ADaM table selected for import. All table level and column metadata are derived from the define.xml file. d. Codelist Table from Define Creates a single SAS table for all codelists selected for import. The generated table can be associated with a terminology package that is associated with the study. 2. Table Level a. Value Level Metadata from Define Imports value level metadata found in the define.xml file into a pre-defined SAS data table that can be associated with the study via the study properties. b. Supplemental Documents from Define Imports supplemental document data found in the define.xml file into a pre-defined SAS data table that can be associated with the study via the study properties. 3. Clinical Transforms a. CDISC-Define Creation Generates a new define.xml file from the selected inputs. b. CDISC-Define Validation Validates that a define.xml file adheres to a schema. c. CDISC-Dataset-XML Creation Creates new dataset-xml files for each selected table. d. CDISC-Dataset-XML Import Creates a SAS data table representation of a dataset-xml file. SETUP There are several administrative tasks that must first be done before continuing. They are typically the responsibility of a Clinical Administrator and are beyond the scope of this paper. The basic necessities are as follows: 1. Import and activate a Data Standard 2. Create a suitable folder structure template for your study data 3. Create a Terminology Package for any codelist tables and associate it with the study 4. Create a Value Level Metadata table (empty) and associate it with the study 5. Create a Supplemental Documents table (empty) and associate it with the study. 2
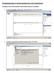
3 THE IMPORT PROCESS We will develop a basic process that imports the data and metadata associated with a define.xml file and its associated dataset-xml files. We will examine the parts of each where applicable so that you gain some familiarity with not only the process, but what is being created. Note that the tools provided for importing a define.xml file are very granular and thus allow the user to pick and choose the parts of the define.xml that he or she might want to import. Finally, we will export (roundtrip) our study data back into a new define.xml file and associated dataset-xml files and will validate that the generated define.xml conforms to a schema. Let s get started 1. Generate a new SAS Clinical Data Integration Study Use the New Study from Define context menu to launch the New Study from Define wizard (Figure 1). The wizard will first prompt for the location of a define.xml to use as an input source. Once selected, the wizard will validate that the file is indeed a define.xml file and then import the study name, description and protocol title values from the define.xml file s GlobalVariables element (Figure 2). Subsequent pages of the wizard allow the opportunity to change these default values, associate a folder template, and optionally associate a data standard (recommended), a library and a terminology package with the study. Once created, the new study will be displayed in the SAS Clinical Data Integration Folder tree. <GlobalVariables> <StudyName>CDISC01</StudyName> <StudyDescription> CDISC Test Study </StudyDescription> <ProtocolName>CDISC01</ProtocolName> </GlobalVariables> Figure 1. New Study from Define Wizard Figure 2. GlobalVariables Element 2. Import the SDTM Domain definitions Next, import the SDTM domain definitions and create empty SAS data table shells for each domain definition found in the define.xml file. All metadata associated at both the table and column level will be imported and stored in the SAS Clinical Data Integration metadata repository. Note that in order to reference permanent data tables on the file system that are associated with the metadata representations in SAS Clinical Data Integration, you will need to associate a Base SAS Library definition as part of the stored table metadata. It is also important to note that physical tables will not be created as part of the domain import process. Use the New Domain(s) from Define context menu wizard to launch the New Domain(s) from Define wizard (Figure 3). The wizard will prompt you for the location of a define.xml to use as an input source and a location to store the table metadata definition within SAS Clinical Data Integration. Next you will be required to select a data standard to associate with the table(s) you are about to import. Subsequent wizard pages allow you to select any (or all) domain definitions found and to optionally associate a SAS library definition for reference to permanent table storage. Upon completion of the wizard your domain table definitions will be created in the SAS Clinical Data Integration metadata repository (Figure 3, 4). 3
4 Figure 3. New Domain(s) from Define Wizard Figure 4. Partial Listing of Domains A quick look at the AE domain s column properties (Figure 5) from within SAS Clinical Data Integration shows that the ItemGroupDef element s attributes were imported correctly (Figure 6). Figure 5. Column Properties for the AE Domain 4
5 <ItemGroupDef OID="IG.AE" Name="AE" Repeating="Yes" IsReferenceData="No" SASDatasetName="AE" Domain="AE" Purpose="Tabulation" def:class="events" def:structure="one record per adverse event per subject" def:archivelocationid="lf.ae"> <Description> <TranslatedText xml:lang="en">adverse Events</TranslatedText> </Description> <ItemRef ItemOID="IT.AE.STUDYID" Mandatory="Yes" OrderNumber="1" KeySequence="1"/> <ItemRef ItemOID="IT.AE.DOMAIN" Mandatory="Yes" OrderNumber="2"/> <ItemRef ItemOID="IT.AE.USUBJID" Mandatory="Yes" OrderNumber="3" KeySequence="2" MethodOID="MT.AE.USUBJID" <ItemRef ItemOID="IT.AE.AESEQ" Mandatory="Yes" OrderNumber="4" MethodOID="MT.AE.AESEQ"/> <ItemRef ItemOID="IT.AE.AESPID" Mandatory="No" OrderNumber="5"/> <ItemRef ItemOID="IT.AE.AETERM" Mandatory="Yes" OrderNumber="6"/> <ItemRef ItemOID="IT.AE.AEMODIFY" Mandatory="No" OrderNumber="7"/> <ItemRef ItemOID="IT.AE.AEDECOD" Mandatory="Yes" OrderNumber="8" KeySequence="3"/> <ItemRef ItemOID="IT.AE.AEBODSYS" Mandatory="No" OrderNumber="9"/> <ItemRef ItemOID="IT.AE.AESEV" Mandatory="No" OrderNumber="10"/> <ItemRef ItemOID="IT.AE.AESER" Mandatory="No" OrderNumber="11"/> <ItemRef ItemOID="IT.AE.AEACN" Mandatory="No" OrderNumber="12"/> <ItemRef ItemOID="IT.AE.AEREL" Mandatory="No" OrderNumber="13"/> <ItemRef ItemOID="IT.AE.AESTDTC" Mandatory="No" OrderNumber="14" KeySequence="4"/> <ItemRef ItemOID="IT.AE.AEENDTC" Mandatory="No" OrderNumber="15"/> <ItemRef ItemOID="IT.AE.AESTDY" Mandatory="No" OrderNumber="16" MethodOID="MT.AE.AESTDY"/> <ItemRef ItemOID="IT.AE.AEENDY" Mandatory="No" OrderNumber="17" MethodOID="MT.AE.AEENDY"/> <ItemRef ItemOID="IT.AE.AEENRF" Mandatory="No" OrderNumber="18"/> <def:leaf ID="LF.AE" xlink:href="ae.xpt"> <def:title>ae.xpt</def:title> </def:leaf> </ItemGroupDef> Figure 6. The ItemGroupDef Element for the AE Domain 3. Import the Codelists SAS Clinical Data Integration can manage two distinct types of codelist data; those imported directly from SAS Clinical Standards Toolkit, and external codelist tables such as those imported from a define.xml or other source. The process of associating a codelist table with a study is: a. Create a Terminology Package b. Associate the Terminology Package with the study c. Associate the imported codelist table with the Terminology Package These steps are not within the scope of this paper and it is assumed that the user is familiar with the processes involved. It is also important to note SAS Clinical Data Integration does not manage any metadata associated with an imported codelist table other than the SAS Base Library association to the physical data table itself. Use the New Codelist Table from Define context menu wizard to launch the New Codelist Table from Define wizard. The wizard will prompt you for the location of a define.xml to use as input and a location to store the codelist table s metadata definition within SAS Clinical Data Integration. The next wizard page will display all of the codelists that are available to import. Select any or all depending on your need (Figure 7). The final wizard page asks you to provide a codelist name, optional description and a required SAS Library used to create the permanent physical SAS data table. Upon completion of the wizard the new codelist table is created and registered with SAS Clinical Data Integration. Remember that you must also register the new codelist table with a SAS Terminology Package for it to be associated with a study. 5
6 Figure 7. Codelist Selection Wizard Page Finally, a quick look at a sample of the generated codelist table (Figure 8) shows that the CodeList element was imported correctly from the define.xml file (figure 9). Figure 8. Partial Listing of the Generated Codelist Table <CodeList OID="CL.ACN" Name="Action Taken with Study Treatment" DataType="text" SASFormatName="$ACN"> <EnumeratedItem CodedValue="DOSE NOT CHANGED" OrderNumber="1"> <Alias Context="nci:ExtCodeID" Name="C49504"/> </EnumeratedItem> <EnumeratedItem CodedValue="DOSE REDUCED" OrderNumber="2"> <Alias Context="nci:ExtCodeID" Name="C49505"/> </EnumeratedItem> <EnumeratedItem CodedValue="DRUG INTERRUPTED" OrderNumber="3"> <Alias Context="nci:ExtCodeID" Name="C49501"/> <EnumeratedItem CodedValue="DRUG WITHDRAWN" OrderNumber="4"> <Alias Context="nci:ExtCodeID" Name="C49502"/> </EnumeratedItem> <Alias Context="nci:ExtCodeID" Name="C66767"/> </CodeList> Figure 9. The CodeList Element for Action Taken with Study Treatment 6
7 4. Import Value Level Metadata and Supplemental Documents The processes for importing Value Level Metadata and Supplemental Documents from a define.xml file are identical other than the wizards used. The process steps are: a. Create an empty Value Level Metadata or Supplemental Documents table b. Associate the table with the study c. Import data from a define.xml into the table Note that SAS Clinical Data Integration does not manage any metadata associated with the table or table columns other than the metadata link to the table itself. Users are free to edit the data within the tables as long as the table column integrity remains intact. A Clinical Administrator would first create an empty table by selecting Study tab from within the Study Properties page, then selecting the appropriate Add button for Value Level Metadata Table or Supplemental Documents Table (Figure 10). For this example we will trace the path of creating a new empty Value Level Metadata table. Figure 10: The Study Properties Study Tab 7
8 The Available Value Level Metadata Tables window is displayed and the user can select a preexisting table to associate with the study, or create a new one by selecting the New button (Figure 11). Figure 11. The Available Value Level Metadata Tables Window If New is selected, the New Value Level Metadata Table wizard is launched. The user must select a location within SAS Clinical Data Integration to store the metadata link to the physical table that will be created on the user s file system. Once chosen, the user is prompted for a table name, optional description and SAS Library used to create the permanent physical SAS data table (Figure 12). Figure 12. The Table and Library Specification Wizard Page 8
9 Upon completion of the wizard, the new empty table is created and pre-selected for association with the study. And, as was noted earlier, this same process is used to create and associate a Supplemental Documents table with a study, and after creating and adding both tables the Study Properties, Study tab would appear as in Figure 13. Figure 13. The Study Properties Page A quick look at the study s folders shows that the tables have been created (Figure 14). Figure 14. The Empty Tables have been created 9
10 The last step of the process involves importing data into the empty SAS data tables created in the previous steps. To do this, launch the Value Level Metadata from Define or Supplemental Documents from Define context menu by right-clicking on the table icon. The appropriate wizard will be launched and once again prompt you for the path to the define.xml file from which you wish to import the data. Upon completion of the wizard, the data will be imported into the appropriate table (Figure 15). Figure 15. Partial Listing of imported Value Level Metadata 5. Import Define-XML Data into the Domain Table Shells SAS Clinical Data Integration provides a pre-defined Clinical transform that allows you to import CDISC Dataset-XML files. The transform relies on functionality provided by SAS Clinical Standards Toolkit to first create the domain tables (SAS data sets) on the file system and to then load data into them. The domain tables are linked to their counterparts within SAS Clinical Data Integration via the SAS Library that was initially associated with the metadata representation of the domain when it was imported from the define.xml file. This same define.xml file must be used as an input to the transformation when creating and loading the dataset-xml files as metadata definitions within it are used to validate the incoming data. First, create a new Document within SAS Clinical Data Integration that references the define.xml by selecting the New->Document context menu from within the study and pointing the document to the define.xml file that was used to import the domain metadata definitions (Figures 16, 17). Figure 16. Document Definition for the define.xml File Figure 17. Document Folder 10
11 Next, create a new Job and drop the CDISC-Dataset-XML Import transform onto the Job palette. The transform takes one input; the Document (created above) that references the define.xml file that was initially used to import the domain metadata definitions (Figure 18). Figure 18. The CDISC-Dataset-XML Import Job Palette Next, set the input source (where the dataset-xml files reside that you will be importing) and output destination (folder where the physical SAS data tables will be created). To do so, double-click on the transform to open its properties window and then select the Generation tab. Browse to, and select the folder where the dataset-xml files reside on your file system that you wish to import. Next select the output library (destination for the SAS data tables that will be created). Note that you should use the same library definition that was used to import the domain metadata definitions in step 2. By doing so, you will establish the link between the metadata definitions and the physical tables (Figure 19). Figure 19. The Completed Transform s Generation Tab 11
12 Finally, select OK button on the transform s properties window and then run the transform. When the Job completes, there will be a SAS data table created for every dataset-xml file that was found in the input folder (Figure 20). Figure 20. Partial Listing of Generated Domain Data Tables To verify the link between the domain metadata definitions created in step 2, and the physical tables created via the CDISC-Dataset-XML Import transform, simply right click on any of the domain definitions inside of SAS Clinical Data Integration that have a corresponding physical table and select the Open context menu item. (Figures 21, 22). Figure 21. Partial Listing of AE Domain 12
13 <ItemGroupData ItemGroupOID="IG.AE" data:itemgroupdataseq="1"><itemdata ItemOID="IT.AE.STUDYID" Value="CDISC01"/> <ItemData ItemOID="IT.AE.DOMAIN" Value="AE"/> <ItemData ItemOID="IT.AE.USUBJID" Value="CDISC "/> <ItemData ItemOID="IT.AE.AESEQ" Value="1"/> <ItemData ItemOID="IT.AE.AESPID" Value="1"/> <ItemData ItemOID="IT.AE.AETERM" Value="AGITATED"/> <ItemData ItemOID="IT.AE.AEMODIFY" Value="AGITATION"/> <ItemData ItemOID="IT.AE.AEDECOD" Value="Agitation"/> <ItemData ItemOID="IT.AE.AEBODSYS" Value="Psychiatric disorders"/> <ItemData ItemOID="IT.AE.AESEV" Value="MILD"/> <ItemData ItemOID="IT.AE.AESER" Value="N"/> <ItemData ItemOID="IT.AE.AEACN" Value="DOSE NOT CHANGED"/> <ItemData ItemOID="IT.AE.AEREL" Value="POSSIBLY RELATED"/> <ItemData ItemOID="IT.AE.AESTDTC" Value=" "/> <ItemData ItemOID="IT.AE.AESTDY" Value="3"/> <ItemData ItemOID="IT.AE.AEENRF" Value="AFTER"/> </ItemGroupData> Figure 22. The ItemGroupData Element for the AE Domain Corresponding to the First Row THE EXPORT PROCESS SAS Clinical Data Integration provides a set of pre-defined Clinical Transforms that can be used to create define.xml and dataset-xml files, and a transform that can be used to validate a define.xml file against a standard schema. The transforms rely on functionality provided by SAS Clinical Standards Toolkit. We will first create a define.xml file that incorporates the domain, codelist, value level metadata and supplemental documents tables that we created in steps 2-4 of the Import Process. Next we will validate the newly created define.xml file against the standard schema for CDISC-Define-XML Version 2.0. Finally, we will export the physical domain table data into corresponding dataset-xml files, thus completing the roundtrip. 1. Create Source and Destination Documents The clinical transforms used in the following steps require Document objects that reference physical files on your file system. The output document (Output Define.xml) will serve as an output for the CDISC-Define- XML Creation transform. This document should reference the file that you want to use as the output destination for the define.xml file that will be created in step 2 below. A separate input document (Input Define.xml) will serve as an input to the CDISC-Dataset-XML Creation transformation and should reference the define.xml file that was used during the import process. To create a SAS Clinical Data Integration Document, select the New->Document context menu from within the study. (Figures 23, 24). Figure 23. The Input define.xml file Document Definition Figure 24. Document Folder 13
14 2. Export the Study Domains to a Define.xml File To create a define.xml file you must first create a new Job and drop the CDISC-Define-Creation transform onto the Job s palette. The transform takes the Output Define.xml document created in step 1 above (Figure 25). Figure 25. The CDISC-Define Creation Job Palette Double-click on the transform to open its properties window and then select the Tables tab. Next, in the Data Standard Type pull-down menu, select the data standard that you initially associated with your study and the domain table shells that you created earlier. The Study/Submission pull-down menu will populate with all studies associated with the selected data standard. Select the study created in step 1 and the Tables list will be populated with all domain definitions that are defined within the study for this data standard. Select the domains that should be included in the new define.xml that will be created (Figure 26). Figure 26. The Tables Tab with all Domains Selected for a Study 14
15 Next select the Generation tab for the transform. This page allows you to set various options for the new define.xml file you are about to create. SAS Clinical Data Integration supports creation of both define versions 1 and 2 (note that the default is version 1), as well as creation of a Define.pdf (version 1 only) and support for default or custom stylesheets. Select 2.0 in the Define version pull-down, and select the Use default output stylesheet radio button (Figure 27). Figure 27. The Generation Tab Finally, select the OK button on the transform s properties window and then run the transform. When the Job completes, the new define.xml file will be created using the physical file referenced by the output document. 3. Validate the Define.xml File SAS Clinical Data Integration can also be used to validate any CDISC compliant define.xml file against its standard schema. To validate a define.xml file, first create new Job and drop the CDISC-Define-Validation transform onto the Job s palette. The transform takes only one external input, a document object that points to physical define.xml file to be validated. In our case, this would be the Output Define.xml document created in step 1 above as it references the define file created in step 2 above. (Figure 28). Figure 28. The CDISC-Define-Validation Job Palette 15
16 Double-click on the transform to open its properties window and then select the Validation tab. Note that the transformation can validate either define versions 1 or 2. In the Define version pull-down box select 2.0, then close the properties window by selecting the OK button. Run the transform. When finished, open the CRT-DDS Validation Results (Work Table) and verify that no errors were generated (Figure 29). Figure 29. The Validation Results Table 4. Export the Domains to Dataset-XML files To export dataset-xml files you must first create a new Job and drop the CDISC-Dataset-XML Creation transform onto the Job palette. The transform takes one input; a Document such as the one created in step 1 that references a define.xml. Note that the metadata from the referenced define.xml file must match that of the domain table(s) that are to be written out as dataset-xml files. For the purposes of this paper, we could use either the input or output define.xml documents since we did not alter any of the domain metadata. For completeness, we will use the Output Define.xml document containing the current metadata definitions. (Figure 30). Figure 30. The CDISC-Dataset-XML Creation Job Palette 16
17 Double-click on the transform to open its properties window and then select the Tables tab. In the Data Standard Type pull-down menu, select the data standard that you initially associated with your study and the domain table shells that you created earlier. The Study/Submission pull-down menu will populate with all studies associated with the selected data standard. Select the study whose table data you wish to output as dataset-xml files and the Tables list will populate. Finally, select the tables for which dataset-xml files will be created. (Figure 31). Figure 31. The Tables Tab with all Domains Selected for a Study Next, select the Generation tab in the transform s properties window. Select a physical Output folder location for writing the dataset-xml files and then select the OK button to close the properties window. Run the transform. When the transform has completed, the output folder will be populated with individual dataset-xml files for each table selected (Figure 32). Finally, a quick visual comparison of the output datasetxml file s contents with the content of the corresponding original dataset-xml file that was imported will show that the correct data was written to the dataset-xml file (Figures 33, 24) Figure 32: Partial listing of the dataset-xml Files Created 17
18 <?xml version="1.0" encoding="utf-8"?> <!-- Produced from SAS data using SAS Clinical Standards Toolkit --> <ODM xmlns=" xmlns:data=" ODMVersion="1.3.2" FileType="Snapshot" FileOID="STUDY1.AE" PriorFileOID="DEFINE" CreationDateTime=" T17:08:26" data:datasetxmlversion="1.0.0"> <ClinicalData StudyOID="STUDY1" MetaDataVersionOID="MDV.CDISC01.SDTMIG SDTM.1.2"> <ItemGroupData ItemGroupOID="IG.AE" data:itemgroupdataseq="1"> <ItemData ItemOID="IT.AE.STUDYID" Value="CDISC01"/> <ItemData ItemOID="IT.AE.DOMAIN" Value="AE"/> <ItemData ItemOID="IT.AE.USUBJID" Value="CDISC "/> <ItemData ItemOID="IT.AE.AESEQ" Value="1"/> <ItemData ItemOID="IT.AE.AESPID" Value="1"/> <ItemData ItemOID="IT.AE.AETERM" Value="AGITATED"/> <ItemData ItemOID="IT.AE.AEMODIFY" Value="AGITATION"/> <ItemData ItemOID="IT.AE.AEDECOD" Value="Agitation"/> <ItemData ItemOID="IT.AE.AEBODSYS" Value="Psychiatric disorders"/> <ItemData ItemOID="IT.AE.AESEV" Value="MILD"/> <ItemData ItemOID="IT.AE.AESER" Value="N"/> <ItemData ItemOID="IT.AE.AEACN" Value="DOSE NOT CHANGED"/> <ItemData ItemOID="IT.AE.AEREL" Value="POSSIBLY RELATED"/> <ItemData ItemOID="IT.AE.AESTDTC" Value=" "/> <ItemData ItemOID="IT.AE.AESTDY" Value="3"/> <ItemData ItemOID="IT.AE.AEENRF" Value="AFTER"/> </ItemGroupData> Figure 33: First Row of Input Dataset-xml File Data for the AE Domain <?xml version="1.0" encoding="utf-8"?> <!-- Produced from SAS data using the SAS Clinical Standards Toolkit. --> <ODM xmlns=" xmlns:data=" ODMVersion="1.3.2" FileType="Snapshot" FileOID="STUDY1.AE" PriorFileOID="DEFINE" CreationDateTime=" T15:44:13" data:datasetxmlversion="1.0.0"> <ClinicalData StudyOID="STUDY1" MetaDataVersionOID="MDV.CDISC01"> <ItemGroupData ItemGroupOID="IG.AE" data:itemgroupdataseq="1"> <ItemData ItemOID="IT.AE.STUDYID" Value="CDISC01"/> <ItemData ItemOID="IT.AE.DOMAIN" Value="AE"/> <ItemData ItemOID="IT.AE.USUBJID" Value="CDISC "/> <ItemData ItemOID="IT.AE.AESEQ" Value="1"/> <ItemData ItemOID="IT.AE.AESPID" Value="1"/> <ItemData ItemOID="IT.AE.AETERM" Value="AGITATED"/> <ItemData ItemOID="IT.AE.AEMODIFY" Value="AGITATION"/> <ItemData ItemOID="IT.AE.AEDECOD" Value="Agitation"/> <ItemData ItemOID="IT.AE.AEBODSYS" Value="Psychiatric disorders"/> <ItemData ItemOID="IT.AE.AESEV" Value="MILD"/> <ItemData ItemOID="IT.AE.AESER" Value="N"/> <ItemData ItemOID="IT.AE.AEACN" Value="DOSE NOT CHANGED"/> <ItemData ItemOID="IT.AE.AEREL" Value="POSSIBLY RELATED"/> <ItemData ItemOID="IT.AE.AESTDTC" Value=" "/> <ItemData ItemOID="IT.AE.AESTDY" Value="3"/> <ItemData ItemOID="IT.AE.AEENRF" Value="AFTER"/> </ItemGroupData> Figure 33: First Row of Output Dataset-xml File Data for the AE Domain 18
19 CONCLUSION SAS Clinical Data Integration Version 2.6, coupled with the underlying functionality of SAS Clinical Standards Toolkit Version 1.7 provides a powerful set of tools for managing both the import and export of clinical trials metadata and data. These tools, in combination with others provided for metadata management within SAS Clinical Data Integration, facilitate the easy exchange of information between a CRO and a study sponsor, and can be used to generate key pieces of a submission to the FDA. REFERENCES 1. CDISC Define-XML Specification, Version 2.0, March 5, 2013 ( 2. CDISC Dataset-xml, Version 1.0 ( 3. SAS Clinical Data Integration 2.6: User s Guide ( 4. SAS Clinical Standards Toolkit 1.7: User s Guide ( CONTACT INFORMATION Your comments and questions are valued and encouraged. Contact the author at: Ken Ellis SAS Institute Inc. SAS Campus Drive Cary, NC 27519, USA SAS and all other SAS Institute Inc. product or service names are registered trademarks or trademarks of SAS Institute Inc. in the USA and other countries. indicates USA registration. Other brand and product names are trademarks of their respective companies. 19
SDTM, ADaM and define.xml with OpenCDISC Matt Becker, PharmaNet/i3, Cary, NC
 PharmaSUG 2012 - Paper HW07 SDTM, ADaM and define.xml with OpenCDISC Matt Becker, PharmaNet/i3, Cary, NC ABSTRACT Standards are an ongoing focus of the health care and life science industry. Common terms
PharmaSUG 2012 - Paper HW07 SDTM, ADaM and define.xml with OpenCDISC Matt Becker, PharmaNet/i3, Cary, NC ABSTRACT Standards are an ongoing focus of the health care and life science industry. Common terms
New features in SDTM-ETL v.1.2 1 SDTM-ETL TM. New Features in version 1.2
 New features in SDTM-ETL v.1.2 1 SDTM-ETL TM New Features in version 1.2 This document describes the new features in version 1.2 of the SDTM-ETL TM software. It can be used in addition to the STDM-ETL
New features in SDTM-ETL v.1.2 1 SDTM-ETL TM New Features in version 1.2 This document describes the new features in version 1.2 of the SDTM-ETL TM software. It can be used in addition to the STDM-ETL
Instructions for Configuring a SAS Metadata Server for Use with JMP Clinical
 Instructions for Configuring a SAS Metadata Server for Use with JMP Clinical These instructions describe the process for configuring a SAS Metadata server to work with JMP Clinical. Before You Configure
Instructions for Configuring a SAS Metadata Server for Use with JMP Clinical These instructions describe the process for configuring a SAS Metadata server to work with JMP Clinical. Before You Configure
How to easily convert clinical data to CDISC SDTM
 How to easily convert clinical data to CDISC SDTM Ale Gicqueau, Clinovo, Sunnyvale, CA Miki Huang, Clinovo, Sunnyvale, CA Stephen Chan, Clinovo, Sunnyvale, CA INTRODUCTION Sponsors are receiving clinical
How to easily convert clinical data to CDISC SDTM Ale Gicqueau, Clinovo, Sunnyvale, CA Miki Huang, Clinovo, Sunnyvale, CA Stephen Chan, Clinovo, Sunnyvale, CA INTRODUCTION Sponsors are receiving clinical
USE CDISC SDTM AS A DATA MIDDLE-TIER TO STREAMLINE YOUR SAS INFRASTRUCTURE
 USE CDISC SDTM AS A DATA MIDDLE-TIER TO STREAMLINE YOUR SAS INFRASTRUCTURE Kalyani Chilukuri, Clinovo, Sunnyvale CA WUSS 2011 Annual Conference October 2011 TABLE OF CONTENTS 1. ABSTRACT... 3 2. INTRODUCTION...
USE CDISC SDTM AS A DATA MIDDLE-TIER TO STREAMLINE YOUR SAS INFRASTRUCTURE Kalyani Chilukuri, Clinovo, Sunnyvale CA WUSS 2011 Annual Conference October 2011 TABLE OF CONTENTS 1. ABSTRACT... 3 2. INTRODUCTION...
WHITE PAPER. CONVERTING SDTM DATA TO ADaM DATA AND CREATING SUBMISSION READY SAFETY TABLES AND LISTINGS. SUCCESSFUL TRIALS THROUGH PROVEN SOLUTIONS
 WHITE PAPER CONVERTING SDTM DATA TO ADaM DATA AND CREATING SUBMISSION READY SAFETY TABLES AND LISTINGS. An innovative approach to deliver statistical analysis and data in a CDISC ADaM complient manner
WHITE PAPER CONVERTING SDTM DATA TO ADaM DATA AND CREATING SUBMISSION READY SAFETY TABLES AND LISTINGS. An innovative approach to deliver statistical analysis and data in a CDISC ADaM complient manner
Learn how to create web enabled (browser) forms in InfoPath 2013 and publish them in SharePoint 2013. InfoPath 2013 Web Enabled (Browser) forms
 Learn how to create web enabled (browser) forms in InfoPath 2013 and publish them in SharePoint 2013. InfoPath 2013 Web Enabled (Browser) forms InfoPath 2013 Web Enabled (Browser) forms Creating Web Enabled
Learn how to create web enabled (browser) forms in InfoPath 2013 and publish them in SharePoint 2013. InfoPath 2013 Web Enabled (Browser) forms InfoPath 2013 Web Enabled (Browser) forms Creating Web Enabled
Managing Custom Data Standards in SAS Clinical Data Integration
 PharmaSUG 2015 - Paper DS19-SAS Managing Custom Data Standards in SAS Clinical Data Integration ABSTRACT Melissa R. Martinez, SAS Institute, Inc., Round Rock, Texas, United States SAS Clinical Data Integration
PharmaSUG 2015 - Paper DS19-SAS Managing Custom Data Standards in SAS Clinical Data Integration ABSTRACT Melissa R. Martinez, SAS Institute, Inc., Round Rock, Texas, United States SAS Clinical Data Integration
Technical Paper. Defining an ODBC Library in SAS 9.2 Management Console Using Microsoft Windows NT Authentication
 Technical Paper Defining an ODBC Library in SAS 9.2 Management Console Using Microsoft Windows NT Authentication Release Information Content Version: 1.0 October 2015. Trademarks and Patents SAS Institute
Technical Paper Defining an ODBC Library in SAS 9.2 Management Console Using Microsoft Windows NT Authentication Release Information Content Version: 1.0 October 2015. Trademarks and Patents SAS Institute
«How we did it» Implementing CDISC LAB, ODM and SDTM in a Clinical Data Capture and Management System:
 Implementing CDISC LAB, ODM and SDTM in a Clinical Data Capture and Management System: «How we did it» 27 April 2006 Berlin CDISC Interchange Xavier Bessette, [i-clinics] Jozef Aerts, XML4Pharma Who are
Implementing CDISC LAB, ODM and SDTM in a Clinical Data Capture and Management System: «How we did it» 27 April 2006 Berlin CDISC Interchange Xavier Bessette, [i-clinics] Jozef Aerts, XML4Pharma Who are
Business & Decision Life Sciences
 Business & Decision Life Sciences Define-it: Demo webinar Anne-Sophie Bekx / 20th May 2014 INSERT COMPANY LOGO IN MASTER SLIDE HERE History of Define.xml Define-it Software Demo Define-it versions Q &
Business & Decision Life Sciences Define-it: Demo webinar Anne-Sophie Bekx / 20th May 2014 INSERT COMPANY LOGO IN MASTER SLIDE HERE History of Define.xml Define-it Software Demo Define-it versions Q &
SAS 9.3 Foundation for Microsoft Windows
 Software License Renewal Instructions SAS 9.3 Foundation for Microsoft Windows Note: In this document, references to Microsoft Windows or Windows include Microsoft Windows for x64. SAS software is licensed
Software License Renewal Instructions SAS 9.3 Foundation for Microsoft Windows Note: In this document, references to Microsoft Windows or Windows include Microsoft Windows for x64. SAS software is licensed
SDTM-ETL TM. The user-friendly ODM SDTM Mapping software package. Transforming operational clinical data into SDTM datasets is not an easy process.
 SDTM-ETL TM The user-friendly ODM SDTM Mapping software package Transforming operational clinical data into SDTM datasets is not an easy process. Therefore, XML4Pharma has developed a visual, easy-to-use
SDTM-ETL TM The user-friendly ODM SDTM Mapping software package Transforming operational clinical data into SDTM datasets is not an easy process. Therefore, XML4Pharma has developed a visual, easy-to-use
Practical application of SAS Clinical Data Integration Server for conversion to SDTM data
 Paper DM03 Practical application of SAS Clinical Data Integration Server for conversion to SDTM data Peter Van Reusel, Business & Decision Life Sciences, Brussels, Belgium Mark Lambrecht, SAS, Tervuren,
Paper DM03 Practical application of SAS Clinical Data Integration Server for conversion to SDTM data Peter Van Reusel, Business & Decision Life Sciences, Brussels, Belgium Mark Lambrecht, SAS, Tervuren,
Sanofi-Aventis Experience Submitting SDTM & Janus Compliant Datasets* SDTM Validation Tools - Needs and Requirements
 In-Depth Review of Tools to Check Compliance of CDISC - Ready Clinical s Bhavin Busa March 2 nd, 2011 Managing Clinical in the Age of CDISC BASUG Quarterly Meeting Presentation Outline Brief introduction
In-Depth Review of Tools to Check Compliance of CDISC - Ready Clinical s Bhavin Busa March 2 nd, 2011 Managing Clinical in the Age of CDISC BASUG Quarterly Meeting Presentation Outline Brief introduction
SDTM-ETL 3.1 New Features
 SDTM-ETL 3.1 New Features SDTM-ETL 3.1 has a lot of new features. The most important ones are listed and explained in this document which lists all major new features of versions 3.0 and 3.1. Support for
SDTM-ETL 3.1 New Features SDTM-ETL 3.1 has a lot of new features. The most important ones are listed and explained in this document which lists all major new features of versions 3.0 and 3.1. Support for
SAS CLINICAL STANDARDS TOOKIT
 You can read the recommendations in the user guide, the technical guide or the installation guide for SAS CLINICAL STANDARDS TOOKIT 1.3. You'll find the answers to all your questions on the SAS CLINICAL
You can read the recommendations in the user guide, the technical guide or the installation guide for SAS CLINICAL STANDARDS TOOKIT 1.3. You'll find the answers to all your questions on the SAS CLINICAL
Getting started with 2c8 plugin for Microsoft Sharepoint Server 2010
 Getting started with 2c8 plugin for Microsoft Sharepoint Server 2010... 1 Introduction... 1 Adding the Content Management Interoperability Services (CMIS) connector... 1 Installing the SharePoint 2010
Getting started with 2c8 plugin for Microsoft Sharepoint Server 2010... 1 Introduction... 1 Adding the Content Management Interoperability Services (CMIS) connector... 1 Installing the SharePoint 2010
9.1 SAS/ACCESS. Interface to SAP BW. User s Guide
 SAS/ACCESS 9.1 Interface to SAP BW User s Guide The correct bibliographic citation for this manual is as follows: SAS Institute Inc. 2004. SAS/ACCESS 9.1 Interface to SAP BW: User s Guide. Cary, NC: SAS
SAS/ACCESS 9.1 Interface to SAP BW User s Guide The correct bibliographic citation for this manual is as follows: SAS Institute Inc. 2004. SAS/ACCESS 9.1 Interface to SAP BW: User s Guide. Cary, NC: SAS
ArcGIS Business Analyst Premium* ~ Help Guide ~ Revised October 3, 2012
 ArcGIS Business Analyst Premium* ~ Help Guide ~ Revised October 3, 2012 ArcGIS Business Analyst Premium is an Esri software package that combines GIS analysis and visualization with data to provide a better
ArcGIS Business Analyst Premium* ~ Help Guide ~ Revised October 3, 2012 ArcGIS Business Analyst Premium is an Esri software package that combines GIS analysis and visualization with data to provide a better
How do I create a Peachtree (Sage 50) Payroll export file?
 How do I create a Peachtree (Sage 50) Payroll export file? Steps to complete prior to running the payroll export in HBS: In Peachtree: Prior to setting up the Peachtree (Sage 50) Payroll export, you will
How do I create a Peachtree (Sage 50) Payroll export file? Steps to complete prior to running the payroll export in HBS: In Peachtree: Prior to setting up the Peachtree (Sage 50) Payroll export, you will
Rationale and vision for E2E data standards: the need for a MDR
 E2E data standards, the need for a new generation of metadata repositories Isabelle de Zegher, PAREXEL Informatics, Belgium Alan Cantrell, PAREXEL, United Kingdom Julie James, PAREXEL Informatics, United
E2E data standards, the need for a new generation of metadata repositories Isabelle de Zegher, PAREXEL Informatics, Belgium Alan Cantrell, PAREXEL, United Kingdom Julie James, PAREXEL Informatics, United
Setting up the Oracle Warehouse Builder Project. Topics. Overview. Purpose
 Setting up the Oracle Warehouse Builder Project Purpose In this tutorial, you setup and configure the project environment for Oracle Warehouse Builder 10g Release 2. You create a Warehouse Builder repository
Setting up the Oracle Warehouse Builder Project Purpose In this tutorial, you setup and configure the project environment for Oracle Warehouse Builder 10g Release 2. You create a Warehouse Builder repository
Interact for Microsoft Office
 Interact for Microsoft Office Installation and Setup Guide Perceptive Content Version: 7.0.x Written by: Product Knowledge, R&D Date: October 2014 2014 Perceptive Software. All rights reserved Perceptive
Interact for Microsoft Office Installation and Setup Guide Perceptive Content Version: 7.0.x Written by: Product Knowledge, R&D Date: October 2014 2014 Perceptive Software. All rights reserved Perceptive
DEPLOYING A VISUAL BASIC.NET APPLICATION
 C6109_AppendixD_CTP.qxd 18/7/06 02:34 PM Page 1 A P P E N D I X D D DEPLOYING A VISUAL BASIC.NET APPLICATION After completing this appendix, you will be able to: Understand how Visual Studio performs deployment
C6109_AppendixD_CTP.qxd 18/7/06 02:34 PM Page 1 A P P E N D I X D D DEPLOYING A VISUAL BASIC.NET APPLICATION After completing this appendix, you will be able to: Understand how Visual Studio performs deployment
Release 2.1 of SAS Add-In for Microsoft Office Bringing Microsoft PowerPoint into the Mix ABSTRACT INTRODUCTION Data Access
 Release 2.1 of SAS Add-In for Microsoft Office Bringing Microsoft PowerPoint into the Mix Jennifer Clegg, SAS Institute Inc., Cary, NC Eric Hill, SAS Institute Inc., Cary, NC ABSTRACT Release 2.1 of SAS
Release 2.1 of SAS Add-In for Microsoft Office Bringing Microsoft PowerPoint into the Mix Jennifer Clegg, SAS Institute Inc., Cary, NC Eric Hill, SAS Institute Inc., Cary, NC ABSTRACT Release 2.1 of SAS
2009 Braton Groupe sarl, All rights reserved.
 D A T A B A S E M A N A G E R U S E R M A N U A L The information in this manual is not binding and may be modified without prior notice. Supply of the software described in this manual is subject to a
D A T A B A S E M A N A G E R U S E R M A N U A L The information in this manual is not binding and may be modified without prior notice. Supply of the software described in this manual is subject to a
Customer Tips. Xerox Network Scanning HTTP/HTTPS Configuration using Microsoft IIS. for the user. Purpose. Background
 Xerox Multifunction Devices Customer Tips June 5, 2007 This document applies to these Xerox products: X WC Pro 232/238/245/ 255/265/275 for the user Xerox Network Scanning HTTP/HTTPS Configuration using
Xerox Multifunction Devices Customer Tips June 5, 2007 This document applies to these Xerox products: X WC Pro 232/238/245/ 255/265/275 for the user Xerox Network Scanning HTTP/HTTPS Configuration using
2012 Teklynx Newco SAS, All rights reserved.
 D A T A B A S E M A N A G E R DMAN-US- 01/01/12 The information in this manual is not binding and may be modified without prior notice. Supply of the software described in this manual is subject to a user
D A T A B A S E M A N A G E R DMAN-US- 01/01/12 The information in this manual is not binding and may be modified without prior notice. Supply of the software described in this manual is subject to a user
Trend Micro Incorporated reserves the right to make changes to this document and to the products described herein without notice.
 Trend Micro Incorporated reserves the right to make changes to this document and to the products described herein without notice. Before installing and using the software, please review the readme files,
Trend Micro Incorporated reserves the right to make changes to this document and to the products described herein without notice. Before installing and using the software, please review the readme files,
SAS Marketing Automation 5.1. User s Guide
 SAS Marketing Automation 5.1 User s Guide The correct bibliographic citation for this manual is as follows: SAS Institute Inc. 2007. SAS Marketing Automation 5.1: User s Guide. Cary, NC: SAS Institute
SAS Marketing Automation 5.1 User s Guide The correct bibliographic citation for this manual is as follows: SAS Institute Inc. 2007. SAS Marketing Automation 5.1: User s Guide. Cary, NC: SAS Institute
Intellicus Enterprise Reporting and BI Platform
 Intellicus Cluster and Load Balancing (Windows) Intellicus Enterprise Reporting and BI Platform Intellicus Technologies info@intellicus.com www.intellicus.com Copyright 2014 Intellicus Technologies This
Intellicus Cluster and Load Balancing (Windows) Intellicus Enterprise Reporting and BI Platform Intellicus Technologies info@intellicus.com www.intellicus.com Copyright 2014 Intellicus Technologies This
Working with Office Applications and ProjectWise
 Working with Office Applications and ProjectWise The main Microsoft Office Applications (Word, Excel, PowerPoint and Outlook) are all integrated with ProjectWise. These applications are aware that ProjectWise
Working with Office Applications and ProjectWise The main Microsoft Office Applications (Word, Excel, PowerPoint and Outlook) are all integrated with ProjectWise. These applications are aware that ProjectWise
Scribe Online Integration Services (IS) Tutorial
 Scribe Online Integration Services (IS) Tutorial 7/6/2015 Important Notice No part of this publication may be reproduced, stored in a retrieval system, or transmitted in any form or by any means, photocopying,
Scribe Online Integration Services (IS) Tutorial 7/6/2015 Important Notice No part of this publication may be reproduced, stored in a retrieval system, or transmitted in any form or by any means, photocopying,
Windows Firewall Configuration with Group Policy for SyAM System Client Installation
 with Group Policy for SyAM System Client Installation SyAM System Client can be deployed to systems on your network using SyAM Management Utilities. If Windows Firewall is enabled on target systems, it
with Group Policy for SyAM System Client Installation SyAM System Client can be deployed to systems on your network using SyAM Management Utilities. If Windows Firewall is enabled on target systems, it
Package R4CDISC. September 5, 2015
 Type Package Title Read CDISC Data Files Version 0.4 Date 2015-9-5 Depends R (>= 3.0.3, XML Author Package R4CDISC September 5, 2015 Maintainer URL https://github.com/i-akiya/r4cdisc
Type Package Title Read CDISC Data Files Version 0.4 Date 2015-9-5 Depends R (>= 3.0.3, XML Author Package R4CDISC September 5, 2015 Maintainer URL https://github.com/i-akiya/r4cdisc
File Management Utility User Guide
 File Management Utility User Guide Legal Notes Unauthorized reproduction of all or part of this guide is prohibited. The information in this guide is subject to change without notice. We cannot be held
File Management Utility User Guide Legal Notes Unauthorized reproduction of all or part of this guide is prohibited. The information in this guide is subject to change without notice. We cannot be held
PharmaSUG2010 - Paper CD13
 PharmaSUG2010 - Paper CD13 In-Depth Review of Validation Tools to Check Compliance of CDISC SDTM-Ready Clinical Datasets Bhavin Busa, Cubist Pharmaceuticals, Inc., Lexington, MA Kim Lindfield, Cubist Pharmaceuticals,
PharmaSUG2010 - Paper CD13 In-Depth Review of Validation Tools to Check Compliance of CDISC SDTM-Ready Clinical Datasets Bhavin Busa, Cubist Pharmaceuticals, Inc., Lexington, MA Kim Lindfield, Cubist Pharmaceuticals,
ProjectWise Explorer V8i User Manual for Subconsultants & Team Members
 ProjectWise Explorer V8i User Manual for Subconsultants & Team Members submitted to Michael Baker International Subconsultants & Team Members submitted by Michael Baker International ProjectWise Support
ProjectWise Explorer V8i User Manual for Subconsultants & Team Members submitted to Michael Baker International Subconsultants & Team Members submitted by Michael Baker International ProjectWise Support
Software Application Tutorial
 Software Application Tutorial Copyright 2005, Software Application Training Unit, West Chester University. No Portion of this document may be reproduced without the written permission of the authors. For
Software Application Tutorial Copyright 2005, Software Application Training Unit, West Chester University. No Portion of this document may be reproduced without the written permission of the authors. For
CDISC Roadmap Outline: Further development and convergence of SDTM, ODM & Co
 CDISC Roadmap Outline: Further development and convergence of SDTM, ODM & Co CDISC Ausblick: Weitere Entwicklung und Konvergenz der CDISC-Standards SDTM, ODM & Co. Jozef Aerts - XML4Pharma Disclaimer Views
CDISC Roadmap Outline: Further development and convergence of SDTM, ODM & Co CDISC Ausblick: Weitere Entwicklung und Konvergenz der CDISC-Standards SDTM, ODM & Co. Jozef Aerts - XML4Pharma Disclaimer Views
Technical Paper. Provisioning Systems and Other Ways to Share the Wealth of SAS
 Technical Paper Provisioning Systems and Other Ways to Share the Wealth of SAS Table of Contents Abstract... 1 Introduction... 1 System Requirements... 1 Deploying SAS Enterprise BI Server... 6 Step 1:
Technical Paper Provisioning Systems and Other Ways to Share the Wealth of SAS Table of Contents Abstract... 1 Introduction... 1 System Requirements... 1 Deploying SAS Enterprise BI Server... 6 Step 1:
Implementing the CDISC standards into an existing CDMS
 Implementing the CDISC standards into an existing CDMS Presented by Ueng-Cheng Yang (yang@ym.edu.tw) Institute of Biomedical Informatics National Yang-Ming University 1 TCT is a resource center to support
Implementing the CDISC standards into an existing CDMS Presented by Ueng-Cheng Yang (yang@ym.edu.tw) Institute of Biomedical Informatics National Yang-Ming University 1 TCT is a resource center to support
NETWRIX FILE SERVER CHANGE REPORTER
 NETWRIX FILE SERVER CHANGE REPORTER ADMINISTRATOR S GUIDE Product Version: 3.3 April/2012. Legal Notice The information in this publication is furnished for information use only, and does not constitute
NETWRIX FILE SERVER CHANGE REPORTER ADMINISTRATOR S GUIDE Product Version: 3.3 April/2012. Legal Notice The information in this publication is furnished for information use only, and does not constitute
Implementing a SAS 9.3 Enterprise BI Server Deployment TS-811. in Microsoft Windows Operating Environments
 Implementing a SAS 9.3 Enterprise BI Server Deployment TS-811 in Microsoft Windows Operating Environments Table of Contents Introduction... 1 Step 1: Create a SAS Software Depot..... 1 Step 2: Prepare
Implementing a SAS 9.3 Enterprise BI Server Deployment TS-811 in Microsoft Windows Operating Environments Table of Contents Introduction... 1 Step 1: Create a SAS Software Depot..... 1 Step 2: Prepare
SAS Business Data Network 3.1
 SAS Business Data Network 3.1 User s Guide SAS Documentation The correct bibliographic citation for this manual is as follows: SAS Institute Inc. 2014. SAS Business Data Network 3.1: User's Guide. Cary,
SAS Business Data Network 3.1 User s Guide SAS Documentation The correct bibliographic citation for this manual is as follows: SAS Institute Inc. 2014. SAS Business Data Network 3.1: User's Guide. Cary,
Legal Notes. Regarding Trademarks. 2012 KYOCERA Document Solutions Inc.
 Legal Notes Unauthorized reproduction of all or part of this guide is prohibited. The information in this guide is subject to change without notice. We cannot be held liable for any problems arising from
Legal Notes Unauthorized reproduction of all or part of this guide is prohibited. The information in this guide is subject to change without notice. We cannot be held liable for any problems arising from
Intellicus Cluster and Load Balancing (Windows) Version: 7.3
 Intellicus Cluster and Load Balancing (Windows) Version: 7.3 Copyright 2015 Intellicus Technologies This document and its content is copyrighted material of Intellicus Technologies. The content may not
Intellicus Cluster and Load Balancing (Windows) Version: 7.3 Copyright 2015 Intellicus Technologies This document and its content is copyrighted material of Intellicus Technologies. The content may not
DropSend Getting Started Guide
 DropSend Getting Started Guide DropSend Ltd. 2012 Step 1: How to send a file without registration If you want to quickly send a large file, you can do it from the homepage in just a couple of clicks. Here
DropSend Getting Started Guide DropSend Ltd. 2012 Step 1: How to send a file without registration If you want to quickly send a large file, you can do it from the homepage in just a couple of clicks. Here
Follow these procedures for QuickBooks Direct or File Integration: Section 1: Direct QuickBooks Integration [Export, Import or Both]
![Follow these procedures for QuickBooks Direct or File Integration: Section 1: Direct QuickBooks Integration [Export, Import or Both] Follow these procedures for QuickBooks Direct or File Integration: Section 1: Direct QuickBooks Integration [Export, Import or Both]](/thumbs/25/5226259.jpg) Follow these procedures for QuickBooks Direct or File Integration: Section 1: Direct QuickBooks Integration [Export, Import or Both] Part A - Configuration Step 1. During installation of the Amano Time
Follow these procedures for QuickBooks Direct or File Integration: Section 1: Direct QuickBooks Integration [Export, Import or Both] Part A - Configuration Step 1. During installation of the Amano Time
UF Health SharePoint 2010 Document Libraries
 UF Health SharePoint 2010 Document Libraries Email: training@health.ufl.edu Web Page: http://training.health.ufl.edu Last Updated 2/7/2014 SharePoint 2010 Document Libraries 1.5 Hours 1.0 Shared Network
UF Health SharePoint 2010 Document Libraries Email: training@health.ufl.edu Web Page: http://training.health.ufl.edu Last Updated 2/7/2014 SharePoint 2010 Document Libraries 1.5 Hours 1.0 Shared Network
How To Set Up An Intellicus Cluster And Load Balancing On Ubuntu 8.1.2.2 (Windows) With A Cluster And Report Server (Windows And Ubuntu) On A Server (Amd64) On An Ubuntu Server
 Intellicus Cluster and Load Balancing (Windows) Intellicus Enterprise Reporting and BI Platform Intellicus Technologies info@intellicus.com www.intellicus.com Copyright 2014 Intellicus Technologies This
Intellicus Cluster and Load Balancing (Windows) Intellicus Enterprise Reporting and BI Platform Intellicus Technologies info@intellicus.com www.intellicus.com Copyright 2014 Intellicus Technologies This
Legal Notes. Regarding Trademarks. 2011 KYOCERA MITA Corporation
 Legal Notes Unauthorized reproduction of all or part of this guide is prohibited. The information in this guide is subject to change without notice. We cannot be held liable for any problems arising from
Legal Notes Unauthorized reproduction of all or part of this guide is prohibited. The information in this guide is subject to change without notice. We cannot be held liable for any problems arising from
Juris Installation / Upgrade Guide
 Juris Installation / Upgrade Guide Version 2.7 2015 LexisNexis. All rights reserved. Copyright and Trademark LexisNexis, Lexis, and the Knowledge Burst logo are registered trademarks of Reed Elsevier Properties
Juris Installation / Upgrade Guide Version 2.7 2015 LexisNexis. All rights reserved. Copyright and Trademark LexisNexis, Lexis, and the Knowledge Burst logo are registered trademarks of Reed Elsevier Properties
SAS Information Delivery Portal: Organize your Organization's Reporting
 SAS Information Delivery Portal: Organize your Organization's Reporting Kevin Davidson Texas Institute for Measurement, Evaluation, and Statistics University of Houston, Houston, TX ABSTRACT The SAS Information
SAS Information Delivery Portal: Organize your Organization's Reporting Kevin Davidson Texas Institute for Measurement, Evaluation, and Statistics University of Houston, Houston, TX ABSTRACT The SAS Information
Exporting emails from Outlook Version 1.00
 Exporting emails from Outlook Version 1.00 The rapid growth in volume of emails means that there is a growing need to archive old emails to media such as external hard disks and DVD s. The document will
Exporting emails from Outlook Version 1.00 The rapid growth in volume of emails means that there is a growing need to archive old emails to media such as external hard disks and DVD s. The document will
ProSeries DMS. Getting Started Guide
 ProSeries DMS Getting Started Guide Copyright 00 Intuit Inc. All Rights Reserved. Intuit, the Intuit logo, Intuit ProLine, ProSeries, and QuickBooks, among others, are trademarks or registered trademarks
ProSeries DMS Getting Started Guide Copyright 00 Intuit Inc. All Rights Reserved. Intuit, the Intuit logo, Intuit ProLine, ProSeries, and QuickBooks, among others, are trademarks or registered trademarks
3 Setting up Databases on a Microsoft SQL 7.0 Server
 3 Setting up Databases on a Microsoft SQL 7.0 Server Overview of the Installation Process To set up GoldMine properly, you must follow a sequence of steps to install GoldMine s program files, and the other
3 Setting up Databases on a Microsoft SQL 7.0 Server Overview of the Installation Process To set up GoldMine properly, you must follow a sequence of steps to install GoldMine s program files, and the other
MY WORLD GIS. Installation Instructions
 MY WORLD GIS Version 4.1 Installation Instructions Copyright 2007 by Northwestern University. All rights reserved. Created August 15, 2002 Last Revised April 14, 2008 2. Launching the Installer On the
MY WORLD GIS Version 4.1 Installation Instructions Copyright 2007 by Northwestern University. All rights reserved. Created August 15, 2002 Last Revised April 14, 2008 2. Launching the Installer On the
User Installation Guide for SAS 9.1 Foundation for 64-bit Microsoft Windows
 User Installation Guide for SAS 9.1 Foundation for 64-bit Microsoft Windows Installation Instructions Where to Begin SAS Setup Wizard Repair or Remove SAS Software Glossary Where to Begin Most people who
User Installation Guide for SAS 9.1 Foundation for 64-bit Microsoft Windows Installation Instructions Where to Begin SAS Setup Wizard Repair or Remove SAS Software Glossary Where to Begin Most people who
Tool Tip. SyAM Management Utilities and Non-Admin Domain Users
 SyAM Management Utilities and Non-Admin Domain Users Some features of SyAM Management Utilities, including Client Deployment and Third Party Software Deployment, require authentication credentials with
SyAM Management Utilities and Non-Admin Domain Users Some features of SyAM Management Utilities, including Client Deployment and Third Party Software Deployment, require authentication credentials with
Shavlik Patch for Microsoft System Center
 Shavlik Patch for Microsoft System Center User s Guide For use with Microsoft System Center Configuration Manager 2012 Copyright and Trademarks Copyright Copyright 2014 Shavlik. All rights reserved. This
Shavlik Patch for Microsoft System Center User s Guide For use with Microsoft System Center Configuration Manager 2012 Copyright and Trademarks Copyright Copyright 2014 Shavlik. All rights reserved. This
Scheduling in SAS 9.3
 Scheduling in SAS 9.3 SAS Documentation The correct bibliographic citation for this manual is as follows: SAS Institute Inc 2011. Scheduling in SAS 9.3. Cary, NC: SAS Institute Inc. Scheduling in SAS 9.3
Scheduling in SAS 9.3 SAS Documentation The correct bibliographic citation for this manual is as follows: SAS Institute Inc 2011. Scheduling in SAS 9.3. Cary, NC: SAS Institute Inc. Scheduling in SAS 9.3
Configuration Guide. Remote Backups How-To Guide. Overview
 Configuration Guide Remote Backups How-To Guide Overview Remote Backups allow you to back-up your data from 1) a ShareCenter TM to either a Remote ShareCenter or Linux Server and 2) Remote ShareCenter
Configuration Guide Remote Backups How-To Guide Overview Remote Backups allow you to back-up your data from 1) a ShareCenter TM to either a Remote ShareCenter or Linux Server and 2) Remote ShareCenter
Rational Quality Manager. Quick Start Tutorial
 Rational Quality Manager Quick Start Tutorial 1 Contents 1. Introduction... 2 2. Terminology... 3 3. Project Area Preparation... 4 3.1 Adding Users and specifying Roles... 4 3.2 Managing Tool Associations...
Rational Quality Manager Quick Start Tutorial 1 Contents 1. Introduction... 2 2. Terminology... 3 3. Project Area Preparation... 4 3.1 Adding Users and specifying Roles... 4 3.2 Managing Tool Associations...
EMC Documentum Composer
 EMC Documentum Composer Version 6.5 User Guide P/N 300 007 217 A02 EMC Corporation Corporate Headquarters: Hopkinton, MA 01748 9103 1 508 435 1000 www.emc.com Copyright 2008 EMC Corporation. All rights
EMC Documentum Composer Version 6.5 User Guide P/N 300 007 217 A02 EMC Corporation Corporate Headquarters: Hopkinton, MA 01748 9103 1 508 435 1000 www.emc.com Copyright 2008 EMC Corporation. All rights
Using Group Policies to Install AutoCAD. CMMU 5405 Nate Bartley 9/22/2005
 Using Group Policies to Install AutoCAD CMMU 5405 Nate Bartley 9/22/2005 Before we get started This manual provides a step-by-step process for creating a Group Policy that will install AutoCAD to a Windows
Using Group Policies to Install AutoCAD CMMU 5405 Nate Bartley 9/22/2005 Before we get started This manual provides a step-by-step process for creating a Group Policy that will install AutoCAD to a Windows
Sage Intelligence Financial Reporting for Sage ERP X3 Version 6.5 Installation Guide
 Sage Intelligence Financial Reporting for Sage ERP X3 Version 6.5 Installation Guide Table of Contents TABLE OF CONTENTS... 3 1.0 INTRODUCTION... 1 1.1 HOW TO USE THIS GUIDE... 1 1.2 TOPIC SUMMARY...
Sage Intelligence Financial Reporting for Sage ERP X3 Version 6.5 Installation Guide Table of Contents TABLE OF CONTENTS... 3 1.0 INTRODUCTION... 1 1.1 HOW TO USE THIS GUIDE... 1 1.2 TOPIC SUMMARY...
Generating a Custom Bill of Materials
 Summary Tutorial TU0104 (v2.3) May 16, 2008 This tutorial describes how to use the Report Manager to set up a Bill of Materials (BOM) report. The manipulation of data and columns and exporting to an Excel
Summary Tutorial TU0104 (v2.3) May 16, 2008 This tutorial describes how to use the Report Manager to set up a Bill of Materials (BOM) report. The manipulation of data and columns and exporting to an Excel
PharmaSUG 2013 - Paper DG06
 PharmaSUG 2013 - Paper DG06 JMP versus JMP Clinical for Interactive Visualization of Clinical Trials Data Doug Robinson, SAS Institute, Cary, NC Jordan Hiller, SAS Institute, Cary, NC ABSTRACT JMP software
PharmaSUG 2013 - Paper DG06 JMP versus JMP Clinical for Interactive Visualization of Clinical Trials Data Doug Robinson, SAS Institute, Cary, NC Jordan Hiller, SAS Institute, Cary, NC ABSTRACT JMP software
Setting up Remote Desktop
 Setting up Remote Desktop Terminology: Host PC the computer you want to access remotely; the destination Client PC the computer you are actually using to access another PC; the source IP Address the numerical
Setting up Remote Desktop Terminology: Host PC the computer you want to access remotely; the destination Client PC the computer you are actually using to access another PC; the source IP Address the numerical
MS Excel Template Building and Mapping for Neat 5
 MS Excel Template Building and Mapping for Neat 5 Neat 5 provides the opportunity to export data directly from the Neat 5 program to an Excel template, entering in column information using receipts saved
MS Excel Template Building and Mapping for Neat 5 Neat 5 provides the opportunity to export data directly from the Neat 5 program to an Excel template, entering in column information using receipts saved
Customer Tips. Configuring Color Access on the WorkCentre 7328/7335/7345 using Windows Active Directory. for the user. Overview
 Xerox Multifunction Devices Customer Tips February 13, 2008 This document applies to the stated Xerox products. It is assumed that your device is equipped with the appropriate option(s) to support the
Xerox Multifunction Devices Customer Tips February 13, 2008 This document applies to the stated Xerox products. It is assumed that your device is equipped with the appropriate option(s) to support the
Producing Listings and Reports Using SAS and Crystal Reports Krishna (Balakrishna) Dandamudi, PharmaNet - SPS, Kennett Square, PA
 Producing Listings and Reports Using SAS and Crystal Reports Krishna (Balakrishna) Dandamudi, PharmaNet - SPS, Kennett Square, PA ABSTRACT The SAS Institute has a long history of commitment to openness
Producing Listings and Reports Using SAS and Crystal Reports Krishna (Balakrishna) Dandamudi, PharmaNet - SPS, Kennett Square, PA ABSTRACT The SAS Institute has a long history of commitment to openness
Application. 1.1 About This Tutorial. 1.1.1 Tutorial Requirements. 1.1.2 Provided Files
 About This Tutorial 1Creating an End-to-End HL7 Over MLLP Application 1.1 About This Tutorial 1.1.1 Tutorial Requirements 1.1.2 Provided Files This tutorial takes you through the steps of creating an end-to-end
About This Tutorial 1Creating an End-to-End HL7 Over MLLP Application 1.1 About This Tutorial 1.1.1 Tutorial Requirements 1.1.2 Provided Files This tutorial takes you through the steps of creating an end-to-end
Business Insight Report Authoring Getting Started Guide
 Business Insight Report Authoring Getting Started Guide Version: 6.6 Written by: Product Documentation, R&D Date: February 2011 ImageNow and CaptureNow are registered trademarks of Perceptive Software,
Business Insight Report Authoring Getting Started Guide Version: 6.6 Written by: Product Documentation, R&D Date: February 2011 ImageNow and CaptureNow are registered trademarks of Perceptive Software,
Configuring Color Access on the WorkCentre 7120 Using Microsoft Active Directory Customer Tip
 Configuring Color Access on the WorkCentre 7120 Using Microsoft Active Directory Customer Tip October 21, 2010 Overview This document describes how to limit access to color copying and printing on the
Configuring Color Access on the WorkCentre 7120 Using Microsoft Active Directory Customer Tip October 21, 2010 Overview This document describes how to limit access to color copying and printing on the
Using SAS Data Integration Studio to Convert Clinical Trials Data to the CDISC SDTM Standard Barry R. Cohen, Octagon Research Solutions, Wayne, PA
 Using SAS Data Integration Studio to Convert Clinical Trials Data to the CDISC SDTM Standard Barry R. Cohen, Octagon Research Solutions, Wayne, PA ABSTRACT A new industry standard for clinical trials data,
Using SAS Data Integration Studio to Convert Clinical Trials Data to the CDISC SDTM Standard Barry R. Cohen, Octagon Research Solutions, Wayne, PA ABSTRACT A new industry standard for clinical trials data,
Oracle SOA Suite 11g Oracle SOA Suite 11g HL7 Inbound Example
 Oracle SOA Suite 11g Oracle SOA Suite 11g HL7 Inbound Example michael.czapski@oracle.com June 2010 Table of Contents Introduction... 1 Pre-requisites... 1 Prepare HL7 Data... 1 Obtain and Explore the HL7
Oracle SOA Suite 11g Oracle SOA Suite 11g HL7 Inbound Example michael.czapski@oracle.com June 2010 Table of Contents Introduction... 1 Pre-requisites... 1 Prepare HL7 Data... 1 Obtain and Explore the HL7
Kaspersky Password Manager USER GUIDE
 Kaspersky Password Manager USER GUIDE Dear User! Thank you for choosing our product. We hope that this documentation helps you in your work and provides answers you may need. Any type of reproduction or
Kaspersky Password Manager USER GUIDE Dear User! Thank you for choosing our product. We hope that this documentation helps you in your work and provides answers you may need. Any type of reproduction or
Hamline University Administrative Computing Page 1
 User Guide Banner Handout: BUSINESS OBJECTS ENTERPRISE (InfoView) Document: boxi31sp3-infoview.docx Created: 5/11/2011 1:24 PM by Chris Berry; Last Modified: 8/31/2011 1:53 PM Purpose:... 2 Introduction:...
User Guide Banner Handout: BUSINESS OBJECTS ENTERPRISE (InfoView) Document: boxi31sp3-infoview.docx Created: 5/11/2011 1:24 PM by Chris Berry; Last Modified: 8/31/2011 1:53 PM Purpose:... 2 Introduction:...
Statistical Operations: The Other Half of Good Statistical Practice
 Integrating science, technology and experienced implementation Statistical Operations: The Other Half of Good Statistical Practice Alan Hopkins, Ph.D. Theravance, Inc. Presented at FDA/Industry Statistics
Integrating science, technology and experienced implementation Statistical Operations: The Other Half of Good Statistical Practice Alan Hopkins, Ph.D. Theravance, Inc. Presented at FDA/Industry Statistics
Implementing a SAS Metadata Server Configuration for Use with SAS Enterprise Guide
 Implementing a SAS Metadata Server Configuration for Use with SAS Enterprise Guide Step 1: Setting Up Required Users and Groups o Windows Operating Systems Only Step 2: Installing Software Using the SAS
Implementing a SAS Metadata Server Configuration for Use with SAS Enterprise Guide Step 1: Setting Up Required Users and Groups o Windows Operating Systems Only Step 2: Installing Software Using the SAS
Context-sensitive Help Guide
 MadCap Software Context-sensitive Help Guide Flare 11 Copyright 2015 MadCap Software. All rights reserved. Information in this document is subject to change without notice. The software described in this
MadCap Software Context-sensitive Help Guide Flare 11 Copyright 2015 MadCap Software. All rights reserved. Information in this document is subject to change without notice. The software described in this
GFI LANguard 9.0 ReportPack. Manual. By GFI Software Ltd.
 GFI LANguard 9.0 ReportPack Manual By GFI Software Ltd. http://www.gfi.com E-mail: info@gfi.com Information in this document is subject to change without notice. Companies, names, and data used in examples
GFI LANguard 9.0 ReportPack Manual By GFI Software Ltd. http://www.gfi.com E-mail: info@gfi.com Information in this document is subject to change without notice. Companies, names, and data used in examples
Releasing blocked email in Data Security
 Releasing blocked email in Data Security IN-TopicInfo:Topic 41101/ Updated: 02-May-2011 Applies To: Websense Data Security v7.1.x Websense Data Security v7.5.x Websense Data Security v7.6.x - v7.8x SMTP
Releasing blocked email in Data Security IN-TopicInfo:Topic 41101/ Updated: 02-May-2011 Applies To: Websense Data Security v7.1.x Websense Data Security v7.5.x Websense Data Security v7.6.x - v7.8x SMTP
System Center Service Manager
 System Center Service Manager E-mail Configuration Version: 0.1 Date: October, 2010 Authors: David Baur Feedback: Send suggestions and comments about this document to David.Baur@infrontconsulting.com Contents
System Center Service Manager E-mail Configuration Version: 0.1 Date: October, 2010 Authors: David Baur Feedback: Send suggestions and comments about this document to David.Baur@infrontconsulting.com Contents
Provisioning Hyper-V virtual machines for a test environment
 Provisioning Hyper-V virtual machines for a test environment Creating a new virtual machine which will be used as a template 1. In Hyper-V Manager, select New->Virtual Machine in the Actions pane. 2. The
Provisioning Hyper-V virtual machines for a test environment Creating a new virtual machine which will be used as a template 1. In Hyper-V Manager, select New->Virtual Machine in the Actions pane. 2. The
Tech Tips Helpful Tips for Pelco Products
 DX4104 UPDATING FROM THE NETWORK SERVER October 29, 2009 DX4104 UPDATING FROM THE NETWORK SERVER The Operation/Configuration manual provides users an option for updating the server through the network
DX4104 UPDATING FROM THE NETWORK SERVER October 29, 2009 DX4104 UPDATING FROM THE NETWORK SERVER The Operation/Configuration manual provides users an option for updating the server through the network
GFI Product Manual. ReportPack Manual
 GFI Product Manual ReportPack Manual http://www.gfi.com info@gfi.com The information and content in this document is provided for informational purposes only and is provided "as is" with no warranty of
GFI Product Manual ReportPack Manual http://www.gfi.com info@gfi.com The information and content in this document is provided for informational purposes only and is provided "as is" with no warranty of
Installing and Configuring Login PI
 Installing and Configuring Login PI Login PI Hands-on lab In this lab, you will configure Login PI to provide performance insights for a Windows Server 2012 R2 Remote Desktop Services installation. To
Installing and Configuring Login PI Login PI Hands-on lab In this lab, you will configure Login PI to provide performance insights for a Windows Server 2012 R2 Remote Desktop Services installation. To
Visual Studio.NET Database Projects
 Visual Studio.NET Database Projects CHAPTER 8 IN THIS CHAPTER Creating a Database Project 294 Database References 296 Scripts 297 Queries 312 293 294 Visual Studio.NET Database Projects The database project
Visual Studio.NET Database Projects CHAPTER 8 IN THIS CHAPTER Creating a Database Project 294 Database References 296 Scripts 297 Queries 312 293 294 Visual Studio.NET Database Projects The database project
Scheduling in SAS 9.4 Second Edition
 Scheduling in SAS 9.4 Second Edition SAS Documentation The correct bibliographic citation for this manual is as follows: SAS Institute Inc. 2015. Scheduling in SAS 9.4, Second Edition. Cary, NC: SAS Institute
Scheduling in SAS 9.4 Second Edition SAS Documentation The correct bibliographic citation for this manual is as follows: SAS Institute Inc. 2015. Scheduling in SAS 9.4, Second Edition. Cary, NC: SAS Institute
IBM Configuring Rational Insight 1.0.1.1 and later for Rational Asset Manager
 IBM Configuring Rational Insight 1.0.1.1 and later for Rational Asset Manager Rational Insight and Rational Asset Manager...4 Prerequisites...5 Configuring the XML data configuration for Rational Asset
IBM Configuring Rational Insight 1.0.1.1 and later for Rational Asset Manager Rational Insight and Rational Asset Manager...4 Prerequisites...5 Configuring the XML data configuration for Rational Asset
Document Management Getting Started Guide
 Document Management Getting Started Guide Version: 6.6.x Written by: Product Documentation, R&D Date: February 2011 ImageNow and CaptureNow are registered trademarks of Perceptive Software, Inc. All other
Document Management Getting Started Guide Version: 6.6.x Written by: Product Documentation, R&D Date: February 2011 ImageNow and CaptureNow are registered trademarks of Perceptive Software, Inc. All other
Business Process Management IBM Business Process Manager V7.5
 Business Process Management IBM Business Process Manager V7.5 Federated task management for BPEL processes and human tasks This presentation introduces the federated task management feature for BPEL processes
Business Process Management IBM Business Process Manager V7.5 Federated task management for BPEL processes and human tasks This presentation introduces the federated task management feature for BPEL processes
William E Benjamin Jr, Owl Computer Consultancy, LLC
 So, You ve Got Data Enterprise Wide (SAS, ACCESS, EXCEL, MySQL, Oracle, and Others); Well, Let SAS Enterprise Guide Software Point-n-Click Your Way to Using It. William E Benjamin Jr, Owl Computer Consultancy,
So, You ve Got Data Enterprise Wide (SAS, ACCESS, EXCEL, MySQL, Oracle, and Others); Well, Let SAS Enterprise Guide Software Point-n-Click Your Way to Using It. William E Benjamin Jr, Owl Computer Consultancy,
OnDemand for Academics
 SAS OnDemand for Academics User s Guide SAS Documentation The correct bibliographic citation for this manual is as follows: SAS Institute Inc. 2015. SAS OnDemand for Academics: User's Guide. Cary, NC:
SAS OnDemand for Academics User s Guide SAS Documentation The correct bibliographic citation for this manual is as follows: SAS Institute Inc. 2015. SAS OnDemand for Academics: User's Guide. Cary, NC:
Talend Open Studio for MDM. Getting Started Guide 6.0.0
 Talend Open Studio for MDM Getting Started Guide 6.0.0 Talend Open Studio for MDM Adapted for v6.0.0. Supersedes previous releases. Publication date: July 2, 2015 Copyleft This documentation is provided
Talend Open Studio for MDM Getting Started Guide 6.0.0 Talend Open Studio for MDM Adapted for v6.0.0. Supersedes previous releases. Publication date: July 2, 2015 Copyleft This documentation is provided
EMC Smarts Network Configuration Manager
 EMC Smarts Network Configuration Manager Version 9.4.1 Advisors User Guide P/N 302-002-279 REV 01 Copyright 2013-2015 EMC Corporation. All rights reserved. Published in the USA. Published October, 2015
EMC Smarts Network Configuration Manager Version 9.4.1 Advisors User Guide P/N 302-002-279 REV 01 Copyright 2013-2015 EMC Corporation. All rights reserved. Published in the USA. Published October, 2015
