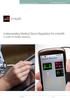
Regulation of Medical Apps FDA and European Union
|
|
|
- Myra Booker
- 8 years ago
- Views:
Transcription
1 JHW Comment - The content of below article is intended to provide a general guide to the subject matter. Specialist advice should be sought about your specific circumstances. Erik Vollebregt Erik is a Dutch lawyer specialised in EU legal issues relating to medical devices Erik s blog can be found at: FDA Draft Guidance for Mobile Medical Applications can be found at: Article by Erik Vollebregt 27 July 2011 On 21 July the FDA released Draft Guidance for Industry and Food and Drug Administration Staff Mobile Medical Applications, a document that I think has great significance for the regulation of the EU ehealth and medical device markets, even if it is presently only a draft. If adopted by the FDA its importance will only increase. Important guidance for the EU too I have read the document and the reasoning behind it can, as you will read in the following, almost for a 100% be transplanted to the EU medical devices regulatory environment and the document describes in a very detailed and practical way what is regulated as device (and what not) while giving lots of examples. This is a significant benefit for stakeholders in the EU (and in the US). Why is this document so significant for regulation of ehealth and medical devices markets in the EU? Firstly, the EU regulatory environment, although software is regulated in very general terms, is way behind in terms of the sophistication and level of detail provided. While the different stakeholders and regulators are going back and forth to agree on possible guidance to be published after the publication of the 'Swedish report' and whether this should be in a MEDDEV or guidance document, the industry and in particular the clinical institutions in the EU seem to have no clue about how apps, websites and software with diagnostic and/or therapeutic functionality are regulated and more often than not act like it is not regulated at all. Secondly, because the reasoning of the FDA fits so well into the EU regulatory system, medical technology and software companies can rely on the same things being regulated as medical device. Of course, the clinical substantiation and the way devices are regulated differs significantly between the EU and the US, but knowing that the same things are regulated as device in the first place is already a big step forwards. Scope of software regulated The FDA proposes that the guidance is applicable to mobile medical applications or "mobile medical apps", defined as "a software application that can be executed (run) on a mobile platform, or a web-based software application that is tailored to a mobile platform but is JHW Editorial Info-Note Regulatory Affairs of Medical Apps Page 1
2 executed on a server"; and has an intended use within the scope of the concept of medical 'device' as regulated by the FDA; and are used as an accessory to a regulated medical device; or transform a mobile platform into a regulated medical device. Although the wording "app" is misleading here because that seems to refer to software that is stored and run locally, it is quite clear that also web applications are covered by the definition. Such regulation applications are: Mobile apps that are an extension of one or more medical device(s) by connecting to such device(s) for purposes of controlling the device(s) or displaying, storing, analyzing, or transmitting patient-specific medical device data Mobile apps that transform the mobile platform into a medical device by using attachments, display screens, or sensors or by including functionalities similar to those of currently regulated medical devices. Mobile apps that allow the user to input patient-specific information and using formulae or processing algorithms output a patient-specific result, diagnosis, or treatment recommendation to be used in clinical practice or to assist in making clinical decisions. The FDA plans to address in a separate issuance mobile medical apps intended to analyze, process, or interpret medical device data (electronically collected or manually entered) from more than one medical device. Excluded however are "mobile apps that are electronic "copies" of medical textbooks, teaching aids or reference materials, or are solely used to provide clinicians with training or reinforce training previously received. These types of apps do not contain any patient-specific information, but could show examples for a specific medical specialty." In EU regulation terms, these devices quite clearly do no achieve a therapeutic or diagnostic effect "in or on the human body" as required by the definition of 'medical device' under the Medical Devices Directive (MDD); Mobile apps that are solely used to log, record, track, evaluate, or make decisions or suggestions related to developing or maintaining general health and wellness, provided that they are not intended for curing, treating, seeking treatment for mitigating, or diagnosing a specific disease, disorder, patient state, or any specific, identifiable health condition. This outcome would be largely similar under EU law as there is no intended therapeutic or diagnostic use except that the EU definition also covers "investigation [...] of the anatomy or of a physiological process" which makes these apps borderline cases under EU law. Mobile apps that only automate general office operations with functionalities that include billing, inventory, appointments, or insurance transactions. These are also excluded under the EU MDD. Mobile apps that are generic aids that assist users but are not commercially marketed for a specific medical indication, such as recording audio, note-taking, replaying audio with amplification, and other similar functionalities. These are currently also excluded under the EU MDD. Mobile apps that perform the functionality of an electronic health record system or personal health record system. These are also excluded under the EU MDD. JHW Editorial Info-Note Regulatory Affairs of Medical Apps Page 2
3 Manufacturer A mobile medical device manufacturer may include anyone who initiates specifications, designs, labels, or creates a software system or application in whole or from multiple software components. Examples of mobile medical device manufacturers include any person or entity that: Creates, designs, develops, labels, re-labels, remanufactures, modifies, or creates a software system from multiple components; Provides mobile medical app functionality through a "web service" or "web support" for use on a mobile platform; Initiates specifications or requirements for mobile medical apps or procures product development/manufacturing services from other individuals or entities (second party) for subsequent commercial distribution; Creates a mobile medical app intended to be used on a mobile platform, or that manufactures a mobile app to be supported by hardware attachments to the mobile platform with a device intended use. All of this fits perfectly in the definition of manufacturer under the MDD, defined as "the natural or legal person with responsibility for the design, manufacture, packaging and labelling of a device before it is placed on the market under his own name, regardless of whether these operations are carried out by that person himself or on his behalf by a third party." An important point is that the FDA urges manufacturers of applications that do not meet the definition of 'device' still apply a quality system to the design and development. A standard that would work for the EU is the EN standard on life cycle requirements for medical devices software harmonised under the MDD. Distributors A very important final point in the draft guidance is that the FDA expects distributors of mobile medical apps who may or may not be a platform or service provider will cooperate with manufacturers in conducting corrections and removal actions and requires medical app manufacturers to make timely reports of corrections and removals made to reduce a health risk or remedy a violation of the FD&C Act that presents a health risk, and to keep records regarding other corrections and removals. In EU wording: the manufacturer must exercise control over its supply chain and be able to implement Field Safety Corrective Action through its supply chain, while the supply chain is expected to cooperate. This is a very important point, because in my experience manufacturers distributing apps via larger stores will have no control whatsoever over these stores, like the itunes Store or the Android Market. They will have very limited options to implement field safety corrective action (in US wording: recall) in case of serious issues. If the FDA and the EU authorities are serious on safety in this respect, they should require design functionality that gives the manufacturer sufficient reach through to the user (e.g. by means of push messages to discontinue use or to upgrade the app to a new version), but also make it clear to the middle men that they have a duty to cooperate as they may be facilitating illegal / unsafe medical devices. In the EU that would mean that the store would for example lose its 'only intermediary' immunity under the e-commerce directive. Conclusion A lot more can be said about the subject, like how to implement in practice all the consequences of a mobile app being regulated as medical device in practice but I will leave it at this for the moment. In any event this draft guidance should be welcomed on both sides of the Atlantic and JHW Editorial Info-Note Regulatory Affairs of Medical Apps Page 3
4 the EU should use it to its advantage to take a much-needed and due step in the clarification of its regulation of software under the MDD. ehealth applications and websites developed by clinicians: There are rules for that! ehealth is booming, hospitals, doctors, clinics, SMEs, big companies, everybody seems to be developing an app nowadays. It is great to see how many people are throwing their weight behind ehealth initiatives in every possible way: remote monitoring of demented senior citizens by means of image interpreting software, decision support systems,, you name it. I am however constantly baffled by the fact that no one ever considers that this type of software, which has either an important role within a medical process or has a medical function in itself (treatment or diagnosis), should be validated on another level than the purely functional one (the does it work? level). This seems to be part of a bigger problem, because physicians have recently repeatedly demonstrated that they are not that familiar with medical technology rules (but still criticise them). A case in point is the recently launched online Health risk test from the Dutch Bronovo hospital. For a mere 19,95 (for the expert test, there s a free version too) you can fill in a host of information about your yourself, your medical history and your lifestyle after which you receive back an overview outlining a number of highly prevalent diseases and the risk of you contracting them. A lot of doctors seem to have cooperated on this software and have lent their name to it, given the number of pictures of people in white coats on the website. In my view, we are talking about a medical device here: there is a piece of software running on a server which applies a model or decision tree, the software produces a result based on this decision tree, the application serves up the information for medical purposes (diagnosis of health risks). In other words, the application fits the definition of software as standalone medical device. Realising that we are dealing with a medical device, I started looking for the CE sign on the website of this application. No CE sign to be found. So I decided to take the test, hoping that somewhere during the process the CE sign would appear. Still no CE sign. So how can I be sure that the manufacturer of the test has properly tested the software (which includes compiling a technical file supporting the way the software reaches its conclusions and presents them)? Soon after, I heard a radio broadcast on Dutch radio channel BNR Nieuwsradio in which it became clear that the Dutch Healthcare Inspectorate (IGZ) also had its doubts about the test as it had not been properly validated and might therefore be in violation of the Dutch Act on Population Investigation (Wet op het Bevolkingsonderzoek). Apparently the possibility of other infringements, like the Act on Medical Devices, has not occurred even to the Inspectorate yet, or the press has not picked up on it but I couldn t locate an official statement of IGZ on the subject. Another case which underlines my point is an app from a Dutch public clinic which claims to support treatment of anxiety disorders. The app shows movies of frightening situations after which viewers are asked to describe how they felt when watching these movies. At a congress, I asked a representative of the company developing the app (a psychiatrist) if it was clinically validated and whether medical devices risk management processes had been applied. What if a patient saw the wrong movie, got an anxiety attack and decided to jump off a building? After all, the goal of the app was therapy, one of the intended purposes that makes software a medical JHW Editorial Info-Note Regulatory Affairs of Medical Apps Page 4
5 device covered by medical devices rules. The company representative reacted very annoyed with my irrelevant questions and decided to avoid me for the remainder of the congress. These are just examples from the Dutch market that I ran into myself but I am sure these cases occur in other markets too. It is strange to see how clinicians that are working on ehealth solutions outside a medical devices company seem to have a tendency to completely overlook the possibility that they are putting medical devices on the market and risk being charged with illegal behaviour. In this particular case they may also have contravened some other rules in the process. Maybe I have met the wrong clinicians who have clouded my judgement. However, as mentioned before, clinicians sometimes do turn out to be very unfamiliar with the medical devices regulatory framework. Therefore, it seems to me that for ehealth to truly take off in a legally compliant fashion, the medical profession has some serious catching up to do. JHW Editorial Info-Note Regulatory Affairs of Medical Apps Page 5
Medical Device Software: Establishing FDA Authority and Mobile Medical Apps
 Medical Device Software: Establishing FDA Authority and Mobile Medical Apps Seth A. Mailhot, Partner Lead, FDA Regulatory Practice Overview Applying the Definition of a Device to Software Special Categories
Medical Device Software: Establishing FDA Authority and Mobile Medical Apps Seth A. Mailhot, Partner Lead, FDA Regulatory Practice Overview Applying the Definition of a Device to Software Special Categories
Mobile Medical Application Development: FDA Regulation
 Mobile Medical Application Development: FDA Regulation Mobile Medical Applications: Current Environment Currently over 100,000 mobile health related applications are available for download; there is an
Mobile Medical Application Development: FDA Regulation Mobile Medical Applications: Current Environment Currently over 100,000 mobile health related applications are available for download; there is an
Regulatory Considerations for Medical Device Software. Medical Device Software
 Medtec Ireland 2015 Wireless Medical Devices Regulatory Considerations for Medical Device Software Kenneth L. Block, RAC October 7, 2015 Galway, Ireland Offices: Dallas, Texas (12 employees) Tokyo, Japan
Medtec Ireland 2015 Wireless Medical Devices Regulatory Considerations for Medical Device Software Kenneth L. Block, RAC October 7, 2015 Galway, Ireland Offices: Dallas, Texas (12 employees) Tokyo, Japan
How To Know If A Mobile App Is A Medical Device
 The Regulation of Medical Device Apps Prepared for West of England Academic Health Science Network and University of Bristol June 2014 June 2014 1 Table of Contents 1 Purpose...3 2 Scope...3 3 The Regulation
The Regulation of Medical Device Apps Prepared for West of England Academic Health Science Network and University of Bristol June 2014 June 2014 1 Table of Contents 1 Purpose...3 2 Scope...3 3 The Regulation
WHITEPAPER: SOFTWARE APPS AS MEDICAL DEVICES THE REGULATORY LANDSCAPE
 WHITEPAPER: SOFTWARE APPS AS MEDICAL DEVICES THE REGULATORY LANDSCAPE White paper produced by Maetrics For more information, please contact global sales +1 610 458 9312 +1 877 623 8742 globalsales@maetrics.com
WHITEPAPER: SOFTWARE APPS AS MEDICAL DEVICES THE REGULATORY LANDSCAPE White paper produced by Maetrics For more information, please contact global sales +1 610 458 9312 +1 877 623 8742 globalsales@maetrics.com
Mobile Medical Applications: FDA s Final Guidance. M. Elizabeth Bierman Anthony T. Pavel Morgan, Lewis & Bockius, LLP
 Mobile Medical Applications: FDA s Final Guidance Michele L. Buenafe M. Elizabeth Bierman Anthony T. Pavel Morgan, Lewis & Bockius, LLP 1 Background FDA has a long-standing policy to regulate any computer
Mobile Medical Applications: FDA s Final Guidance Michele L. Buenafe M. Elizabeth Bierman Anthony T. Pavel Morgan, Lewis & Bockius, LLP 1 Background FDA has a long-standing policy to regulate any computer
CENTER FOR CONNECTED HEALTH POLICY
 CENTER FOR CONNECTED HEALTH POLICY The Center for Connected Health Policy (CCHP) is a public interest nonprofit organization that develops and advances telehealth policy solutions to promote improvements
CENTER FOR CONNECTED HEALTH POLICY The Center for Connected Health Policy (CCHP) is a public interest nonprofit organization that develops and advances telehealth policy solutions to promote improvements
Regulation of Mobile Medical Apps
 Regulation of Mobile Medical Apps May 30, 2014 Copyright 2014 Software Quality Consulting Inc. Slide 1 Speaker Bio Steven R. Rakitin has over 35 years experience as a software engineer and 25 years in
Regulation of Mobile Medical Apps May 30, 2014 Copyright 2014 Software Quality Consulting Inc. Slide 1 Speaker Bio Steven R. Rakitin has over 35 years experience as a software engineer and 25 years in
Navigating the New EU Rules for Medical Device Software
 Navigating the New EU Rules for Medical Device Software Mathias Klümper and Erik Vollebregt examine how European Directive 2007/47/EC will affect requirements for medical device software. Directive 2007/47/EC
Navigating the New EU Rules for Medical Device Software Mathias Klümper and Erik Vollebregt examine how European Directive 2007/47/EC will affect requirements for medical device software. Directive 2007/47/EC
FDA Regulation of Health IT
 FDA Regulation of Health IT September 2014 Marian J. Lee Partner +1 (202) 661 7955 mlee@kslaw.com Agenda FDA s Mobile Medical Applications Guidance FDA Draft Guidance on Medical Device Data Systems (MDDS),
FDA Regulation of Health IT September 2014 Marian J. Lee Partner +1 (202) 661 7955 mlee@kslaw.com Agenda FDA s Mobile Medical Applications Guidance FDA Draft Guidance on Medical Device Data Systems (MDDS),
FDA Issues Final Guidance on Mobile Medical Apps
 ADVISORY September 2013 FDA Issues Final Guidance on Mobile Medical Apps On September 23, 2013, the U.S. Food & Drug Administration (FDA or the Agency) issued its final Guidance for Industry and Food and
ADVISORY September 2013 FDA Issues Final Guidance on Mobile Medical Apps On September 23, 2013, the U.S. Food & Drug Administration (FDA or the Agency) issued its final Guidance for Industry and Food and
FDA Regulation of Health IT
 FDA Regulation of Health IT May 2014 Marian J. Lee Partner King & Spalding +1 (202) 661 7955 mlee@kslaw.com Agenda FDA s Mobile Medical Applications Guidance FDASIA Health IT Report 2 FDA s Mobile Medical
FDA Regulation of Health IT May 2014 Marian J. Lee Partner King & Spalding +1 (202) 661 7955 mlee@kslaw.com Agenda FDA s Mobile Medical Applications Guidance FDASIA Health IT Report 2 FDA s Mobile Medical
Mobile Medical Applications
 Mobile Medical Applications What Is the Impact of FDA s New MMA Guidance for the Life Science Industry? June 6, 2014, 11:15 AM 12:15 PM Presented by: Mark Gardner, M.B.A., J.D. Agenda 1. How does FDA regulate
Mobile Medical Applications What Is the Impact of FDA s New MMA Guidance for the Life Science Industry? June 6, 2014, 11:15 AM 12:15 PM Presented by: Mark Gardner, M.B.A., J.D. Agenda 1. How does FDA regulate
Regulatory Landscape For Mobile Medical Apps (MMAs)
 Regulatory Landscape For Mobile Medical Apps (MMAs) Date: December 6, 2013 Place: Erie County Bar Association, MidDay Learning Program Where value is law. Monika Bhatt mbhatt@hodgsonruss.com Blog: GatewayFDA.com
Regulatory Landscape For Mobile Medical Apps (MMAs) Date: December 6, 2013 Place: Erie County Bar Association, MidDay Learning Program Where value is law. Monika Bhatt mbhatt@hodgsonruss.com Blog: GatewayFDA.com
The Shifting Sands of Medical Software Regulation
 The Shifting Sands of Medical Software Regulation Suzanne O Shea Ralph Hall September 10, 2014 What Software is Regulated by FDA? FDA regulates medical devices. FDA regulates software that meets the definition
The Shifting Sands of Medical Software Regulation Suzanne O Shea Ralph Hall September 10, 2014 What Software is Regulated by FDA? FDA regulates medical devices. FDA regulates software that meets the definition
How To Regulate A Medical Device From A Cell Phone
 On Behalf of: InTouch Health White Paper FDA Regulation of Mobile Health Technologies The Current Regulatory Framework as Applied to InTouch Health s Telemedicine Solution June 15, 2012 TABLE OF CONTENTS
On Behalf of: InTouch Health White Paper FDA Regulation of Mobile Health Technologies The Current Regulatory Framework as Applied to InTouch Health s Telemedicine Solution June 15, 2012 TABLE OF CONTENTS
Use of Mobile Medical Applications in Clinical Research
 Use of Mobile Medical Applications in Clinical Research Erin K. O Reilly, PhD RAC Associate Director, Regulatory Affairs Duke Translational Medicine Institute erin.oreilly@duke.edu September 10, 2014 (919)
Use of Mobile Medical Applications in Clinical Research Erin K. O Reilly, PhD RAC Associate Director, Regulatory Affairs Duke Translational Medicine Institute erin.oreilly@duke.edu September 10, 2014 (919)
On Behalf of: InTouch Health
 On Behalf of: InTouch Health White Paper FDA Regulation of Mobile Health Technologies The Current Regulatory Framework as Applied to InTouch Health s Telehealth Solutions June 15, 2012; Updated June 15,
On Behalf of: InTouch Health White Paper FDA Regulation of Mobile Health Technologies The Current Regulatory Framework as Applied to InTouch Health s Telehealth Solutions June 15, 2012; Updated June 15,
Final Document. Software as a Medical Device (SaMD): Key Definitions. Date: 9 December 2013. Despina Spanou, IMDRF Chair. IMDRF/SaMD WG/N10FINAL:2013
 Final Document Title: Authoring Group: Software as a Medical Device (SaMD): Key Definitions IMDRF SaMD Working Group Date: 9 December 2013 Despina Spanou, IMDRF Chair This document was produced by the
Final Document Title: Authoring Group: Software as a Medical Device (SaMD): Key Definitions IMDRF SaMD Working Group Date: 9 December 2013 Despina Spanou, IMDRF Chair This document was produced by the
Proposed Definition for Clinical Decision Support Software
 May 15, 2013 Proposed Definition for Clinical Decision Support Software The International Medical Device Regulators Forum (IMDRF) has recently announced a Work Item on Standalone Software. As we understand
May 15, 2013 Proposed Definition for Clinical Decision Support Software The International Medical Device Regulators Forum (IMDRF) has recently announced a Work Item on Standalone Software. As we understand
Robert Jarrin Senior Director, Government Affairs. May 22, 2013
 Robert Jarrin Senior Director, Government Affairs May 22, 2013 1 Section 201(h) FD&C Act "an instrument, apparatus, implement, machine, contrivance, implant, in vitro reagent, or other similar or related
Robert Jarrin Senior Director, Government Affairs May 22, 2013 1 Section 201(h) FD&C Act "an instrument, apparatus, implement, machine, contrivance, implant, in vitro reagent, or other similar or related
Regulated Mobile Applications
 Regulated Mobile Applications Sion Wyn Conformity +[44] (0) 1492 642622 sion.wyn@conform-it.com Waters, Wilmslow, November 2014 1 Introduction According to industry estimates, 500 million smartphone users
Regulated Mobile Applications Sion Wyn Conformity +[44] (0) 1492 642622 sion.wyn@conform-it.com Waters, Wilmslow, November 2014 1 Introduction According to industry estimates, 500 million smartphone users
Loss Control Webinar Series. Mobile Medical Apps: FDA Regulation and Products Liability Implications 10-23-2013
 Loss Control Webinar Series Mobile Medical Apps: FDA Regulation and Products Liability Implications 10-23-2013 www.goldbergsegalla.com NEW YORK PENNSYLVANIA CONNECTICUT NEW JERSEY UNITED KINGDOM Mobile
Loss Control Webinar Series Mobile Medical Apps: FDA Regulation and Products Liability Implications 10-23-2013 www.goldbergsegalla.com NEW YORK PENNSYLVANIA CONNECTICUT NEW JERSEY UNITED KINGDOM Mobile
The U.S. FDA s Regulation and Oversight of Mobile Medical Applications
 The U.S. FDA s Regulation and Oversight of Mobile Medical Applications The U.S. FDA s Regulation and Oversight of Mobile Medical Applications As smart phones and portable tablet computers become the preferred
The U.S. FDA s Regulation and Oversight of Mobile Medical Applications The U.S. FDA s Regulation and Oversight of Mobile Medical Applications As smart phones and portable tablet computers become the preferred
Which Apps Does FDA Regulate?
 Which Apps Does FDA Regulate? Bradley Merrill Thompson 2015 EPSTEIN BECKER & GREEN, P.C. ALL RIGHTS RESERVED.. 1 Topics for Discussion Which apps does FDA regulate? Enforcement Discretion Pharma Apps Future
Which Apps Does FDA Regulate? Bradley Merrill Thompson 2015 EPSTEIN BECKER & GREEN, P.C. ALL RIGHTS RESERVED.. 1 Topics for Discussion Which apps does FDA regulate? Enforcement Discretion Pharma Apps Future
Developing a Mobile Medical App? How to determine if it is a medical device and get it cleared by the US FDA
 Developing a Mobile Medical App? How to determine if it is a medical device and get it cleared by the US FDA In this presentation: App stats: Explosive growth Examples already cleared by the US FDA Is
Developing a Mobile Medical App? How to determine if it is a medical device and get it cleared by the US FDA In this presentation: App stats: Explosive growth Examples already cleared by the US FDA Is
COMMISSION STAFF WORKING DOCUMENT. on the existing EU legal framework applicable to lifestyle and wellbeing apps. Accompanying the document
 EUROPEAN COMMISSION Brussels, 10.4.2014 SWD(2014) 135 final COMMISSION STAFF WORKING DOCUMENT on the existing EU legal framework applicable to lifestyle and wellbeing apps Accompanying the document GREEN
EUROPEAN COMMISSION Brussels, 10.4.2014 SWD(2014) 135 final COMMISSION STAFF WORKING DOCUMENT on the existing EU legal framework applicable to lifestyle and wellbeing apps Accompanying the document GREEN
EU Law and Life Sciences Mobile Health Apps. Webinar - Brussels, 9 July 2014
 EU Law and Life Sciences Mobile Health Apps Webinar - Brussels, 9 July 2014 Dr. Dr. Adem Koyuncu, M.D. Partner Prof. Damien Geradin Partner Agenda Covington at a glance Mobile health apps - opportunities
EU Law and Life Sciences Mobile Health Apps Webinar - Brussels, 9 July 2014 Dr. Dr. Adem Koyuncu, M.D. Partner Prof. Damien Geradin Partner Agenda Covington at a glance Mobile health apps - opportunities
Mobile Medical Apps. Purpose. Diane Romza Kutz Fredric E. Roth V. Regulation and Risks. Purpose of today s presentation
 Mobile Medical Apps Regulation and Risks Diane Romza Kutz Fredric E. Roth V Purpose Purpose of today s presentation Identify the newly-regulated industry Identify the newly regulated products and the basis
Mobile Medical Apps Regulation and Risks Diane Romza Kutz Fredric E. Roth V Purpose Purpose of today s presentation Identify the newly-regulated industry Identify the newly regulated products and the basis
Key Challenges and Unmet Needs in Bipolar Disorder
 Key Challenges and Unmet Needs in Bipolar Disorder Bridging the Gap Between Healthcare Professionals and Patients A Pilot Malaysian Study March and April 2010 WHAT THE REPORT WILL COVER Executive Summary
Key Challenges and Unmet Needs in Bipolar Disorder Bridging the Gap Between Healthcare Professionals and Patients A Pilot Malaysian Study March and April 2010 WHAT THE REPORT WILL COVER Executive Summary
The iphone as a Medical Device
 The iphone as a Medical Device Presented by: Melissa L. Markey, Esq. Hall, Render, Killian, Heath & Lyman, PLLC 201 West Big Beaver Rd, Suite 315 Troy, Michigan (248) 740-7505 Hall, Render, Killian, Heath
The iphone as a Medical Device Presented by: Melissa L. Markey, Esq. Hall, Render, Killian, Heath & Lyman, PLLC 201 West Big Beaver Rd, Suite 315 Troy, Michigan (248) 740-7505 Hall, Render, Killian, Heath
Medical Device Data Systems, Medical Image Storage Devices, and Medical Image Communications Devices
 Medical Device Data Systems, Medical Image Storage Devices, and Medical Image Communications Devices Draft Guidance for Industry and Food and Drug Administration Staff DRAFT GUIDANCE This guidance document
Medical Device Data Systems, Medical Image Storage Devices, and Medical Image Communications Devices Draft Guidance for Industry and Food and Drug Administration Staff DRAFT GUIDANCE This guidance document
Breakout Sessions: FDA s Regulation of Mobile Health and Medical Applications
 Breakout Sessions: FDA s Regulation of Mobile Health and Medical Applications 2015 Annual Conference Washington, DC Bakul Patel, Associate Director for Digital Health, Office of Center Director, Center
Breakout Sessions: FDA s Regulation of Mobile Health and Medical Applications 2015 Annual Conference Washington, DC Bakul Patel, Associate Director for Digital Health, Office of Center Director, Center
Risk based 12/1/2015. Digital Health Bakul Patel Associate Director for Digital Health Office of Center Director.
 Digital Health Bakul Patel Associate Director for Digital Health Office of Center Director Center for Devices and Radiological Health 1 Oversight Approach Platform independent Promote innovation Promote
Digital Health Bakul Patel Associate Director for Digital Health Office of Center Director Center for Devices and Radiological Health 1 Oversight Approach Platform independent Promote innovation Promote
Mobile Medical Applications: An Overview of FDA Regulation
 Mobile Medical Applications: An Overview of FDA Regulation RAPS Annual Convention 2014 Austin, Texas Michael A. Swit, Esq. Special Counsel, FDA Law Practice Duane Morris LLP Standard Disclaimers The views
Mobile Medical Applications: An Overview of FDA Regulation RAPS Annual Convention 2014 Austin, Texas Michael A. Swit, Esq. Special Counsel, FDA Law Practice Duane Morris LLP Standard Disclaimers The views
MOBILE MEDICAL APPLICATIONS
 October 7, 2013 EVOKE HEALTH POINT OF VIEW MOBILE MEDICAL APPLICATIONS FDA GUIDANCE FOR INDUSTRY FOR MORE INFORMATION: Mark McConaghy, VP, Strategy Evoke Health 267.765.4998 mark.mcconaghy@evokehealth.com
October 7, 2013 EVOKE HEALTH POINT OF VIEW MOBILE MEDICAL APPLICATIONS FDA GUIDANCE FOR INDUSTRY FOR MORE INFORMATION: Mark McConaghy, VP, Strategy Evoke Health 267.765.4998 mark.mcconaghy@evokehealth.com
Medical Technologies Market in Turkey
 U.S. Commercial Service Medical Technologies Market in Turkey Brief Market Report Prepared by Ebru Olcay, Medical Sector Specialist Turkey: Medical Technologies and Healthcare Market and What Is The Size
U.S. Commercial Service Medical Technologies Market in Turkey Brief Market Report Prepared by Ebru Olcay, Medical Sector Specialist Turkey: Medical Technologies and Healthcare Market and What Is The Size
A Design of Mobile Convergence Architecture for U-healthcare
 , pp. 253-260 http://dx.doi.org/10.14257/ijseia.2015.9.1.22 A Design of Mobile Convergence Architecture for U-healthcare Regin Joy Conejar 1 and Haeng-Kon Kim 1* 1 School of Information Technology Catholic
, pp. 253-260 http://dx.doi.org/10.14257/ijseia.2015.9.1.22 A Design of Mobile Convergence Architecture for U-healthcare Regin Joy Conejar 1 and Haeng-Kon Kim 1* 1 School of Information Technology Catholic
Rethinking the FDA s Regulation of. By Scott D. Danzis and Christopher Pruitt
 Rethinking the FDA s Regulation of Mobile Medical Apps By Scott D. Danzis and Christopher Pruitt Smartphones and mobile devices have rapidly become part of everyday life in the United States. It is no
Rethinking the FDA s Regulation of Mobile Medical Apps By Scott D. Danzis and Christopher Pruitt Smartphones and mobile devices have rapidly become part of everyday life in the United States. It is no
REFERENCE DOCUMENT. (DRAFT for Public Comments) Title: White Paper on Medical Device Software Regulation Software Qualification and Classification
 AHWP/WG1/R001:2014 REFERENCE DOCUMENT (DRAFT for Public Comments) Title: White Paper on Medical Device Software Regulation Software Qualification and Classification Author: Work Group 1, Pre-Market Submission
AHWP/WG1/R001:2014 REFERENCE DOCUMENT (DRAFT for Public Comments) Title: White Paper on Medical Device Software Regulation Software Qualification and Classification Author: Work Group 1, Pre-Market Submission
Websites & Social Media. in the Professional Environment. A practical guide to navigating the world of social media
 Websites & Social Media in the Professional Environment @ A practical guide to navigating the world of social media istockphoto.com/andrearoad Introduction Your Online Presence An online presence is essential
Websites & Social Media in the Professional Environment @ A practical guide to navigating the world of social media istockphoto.com/andrearoad Introduction Your Online Presence An online presence is essential
Draft Guidance for Industry and Food and Drug Administration Staff
 Draft Guidance for Industry and Food and Drug Administration Staff Mobile Medical Applications DRAFT GUIDANCE This guidance document is being distributed for comment purposes only. Document issued on:
Draft Guidance for Industry and Food and Drug Administration Staff Mobile Medical Applications DRAFT GUIDANCE This guidance document is being distributed for comment purposes only. Document issued on:
An introduction to the regulation of apps and wearables as medical devices
 An introduction to the regulation of apps and wearables as medical devices Introduction Phones are increasingly equipped with a range of devices capable of being used as sensors, including gyroscopes,
An introduction to the regulation of apps and wearables as medical devices Introduction Phones are increasingly equipped with a range of devices capable of being used as sensors, including gyroscopes,
Testimony of Denise Lea Elliott, DPM Member, American Podiatric Medical Association Solo Practitioner Marrero, Louisiana
 Testimony of Denise Lea Elliott, DPM Member, American Podiatric Medical Association Solo Practitioner Marrero, Louisiana Before the Subcommittee on Healthcare and Technology, House Small Business Committee
Testimony of Denise Lea Elliott, DPM Member, American Podiatric Medical Association Solo Practitioner Marrero, Louisiana Before the Subcommittee on Healthcare and Technology, House Small Business Committee
Scope of Practice and Qualifications - Nurse Practitioner 26 February 2014
 Scope of Practice and Qualifications - Nurse Practitioner 26 February 2014 New Zealand Society of Anaesthetists feedback 6. Do you support nurse practitioner being a broad generic scope of practice (like
Scope of Practice and Qualifications - Nurse Practitioner 26 February 2014 New Zealand Society of Anaesthetists feedback 6. Do you support nurse practitioner being a broad generic scope of practice (like
Secure, diagnostically-cleared, mobile access to imaging and information.
 Secure, diagnostically-cleared, mobile access to imaging and information. WHY THINK MOBILE FIRST? A mobile first approach offers quicker access to medical information to enable more informed decisions,
Secure, diagnostically-cleared, mobile access to imaging and information. WHY THINK MOBILE FIRST? A mobile first approach offers quicker access to medical information to enable more informed decisions,
Position Paper. Orgalime response to the Public consultation on the. collaborative economy - Digital Single Market Strategy follow up assessment
 Position Paper Brussels, 23 December 2015 Orgalime response to the Public consultation on the regulatory environment for platforms, online intermediaries, data and cloud computing and the collaborative
Position Paper Brussels, 23 December 2015 Orgalime response to the Public consultation on the regulatory environment for platforms, online intermediaries, data and cloud computing and the collaborative
Market Entry Toolkit. Mobile Health Technology and Architecture: A Practical Framework for Technology Assessment for Mobile Health
 Market Entry Toolkit Mobile Health Technology and Architecture: Kai-Lik Foh, Mobile Health Programme Manager, GSMA kfoh@gsm.org September 2011 Contents Introduction...2 ehealth vs mhealth Architecture...2
Market Entry Toolkit Mobile Health Technology and Architecture: Kai-Lik Foh, Mobile Health Programme Manager, GSMA kfoh@gsm.org September 2011 Contents Introduction...2 ehealth vs mhealth Architecture...2
Mobile Medical Applications. Guidance for Industry and Food and Drug Administration Staff
 Mobile Medical Applications Guidance for Industry and Food and Drug Administration Staff Document issued on: September 25, 2013 The draft of this guidance was issued on July 21, 2011. For questions regarding
Mobile Medical Applications Guidance for Industry and Food and Drug Administration Staff Document issued on: September 25, 2013 The draft of this guidance was issued on July 21, 2011. For questions regarding
EHRs and Contexts of Use
 2 EHRs and Contexts of Use A Providers Perspective on EHR User Experience Presented by March 2013 1 EHRs and Contexts of Use It s not a new concept. Most modern EHRs, descending from products designed
2 EHRs and Contexts of Use A Providers Perspective on EHR User Experience Presented by March 2013 1 EHRs and Contexts of Use It s not a new concept. Most modern EHRs, descending from products designed
Draft guidance for registered pharmacies providing internet and distance sale, supply or service provision
 Draft guidance for registered pharmacies providing internet and distance sale, supply or service provision September 2014 1 The General Pharmaceutical Council is the regulator for pharmacists, pharmacy
Draft guidance for registered pharmacies providing internet and distance sale, supply or service provision September 2014 1 The General Pharmaceutical Council is the regulator for pharmacists, pharmacy
A Primer: Solving the Unique Problems of Orphan Drug Communications Programs
 A Primer: Solving the Unique Problems of Orphan Drug Communications Programs Introduction When communicating about orphan diseases and development of pharmaceuticals, it is essential to recognize the special
A Primer: Solving the Unique Problems of Orphan Drug Communications Programs Introduction When communicating about orphan diseases and development of pharmaceuticals, it is essential to recognize the special
Night Owl 8 channel DVR system with HDMI output, 960H recording and a Free Night Owl Pro App with Owl Scan (No Hard Drive Included)
 Night Owl 8 channel DVR system with HDMI output, 960H recording and a Free Night Owl Pro App with Owl Scan (No Hard Drive Included) Night Owl s PE-DVR8 is an advanced 8 channel 960H Security DVR. The DVR
Night Owl 8 channel DVR system with HDMI output, 960H recording and a Free Night Owl Pro App with Owl Scan (No Hard Drive Included) Night Owl s PE-DVR8 is an advanced 8 channel 960H Security DVR. The DVR
Web-Based Software Tools Enable Lean Sales and Distribution Fred Noble
 Web-Based Software Tools Enable Lean Sales and Distribution Fred Noble During the last 25 years manufacturers have devoted much time to studying the concepts and practices of the Lean Enterprise, a continuous
Web-Based Software Tools Enable Lean Sales and Distribution Fred Noble During the last 25 years manufacturers have devoted much time to studying the concepts and practices of the Lean Enterprise, a continuous
Filename: P4P 016 Todd: Kim: Todd: Kim:
 Filename: P4P 016 Todd: [0:00:18] Hey everybody, welcome to another edition of The Prosperity Podcast, this is No BS Money Guy Todd Strobel. Once again, we have my cohost, bestselling financial author
Filename: P4P 016 Todd: [0:00:18] Hey everybody, welcome to another edition of The Prosperity Podcast, this is No BS Money Guy Todd Strobel. Once again, we have my cohost, bestselling financial author
Look at what innovation can do
 Clinic Information System Practice Management Tool Look at what innovation can do Despite all the innovative solutions we have put into CIS, the most amazing result might just be what you can get out of
Clinic Information System Practice Management Tool Look at what innovation can do Despite all the innovative solutions we have put into CIS, the most amazing result might just be what you can get out of
Legal compliance for developers. Training materials (prepared by Tilburg University)
 Legal compliance for developers using FI-STAR ehealth platform Training materials (prepared by Tilburg University) Target audience: Target audience and objectives developers & other potential users of
Legal compliance for developers using FI-STAR ehealth platform Training materials (prepared by Tilburg University) Target audience: Target audience and objectives developers & other potential users of
Unleashing the Potential of Cloud Computing in Europe - What is it and what does it mean for me?
 EUROPEAN COMMISSION MEMO Brussels, 27 September 2012 Unleashing the Potential of Cloud Computing in Europe - What is it and what does it mean for me? See also IP/12/1025 What is Cloud Computing? Cloud
EUROPEAN COMMISSION MEMO Brussels, 27 September 2012 Unleashing the Potential of Cloud Computing in Europe - What is it and what does it mean for me? See also IP/12/1025 What is Cloud Computing? Cloud
ilinc Legal & Technology Briefs The Liability of Internet Intermediaries In the EU
 ilinc Legal & Technology Briefs The Liability of Internet Intermediaries In the EU Many start-ups fulfil an intermediary role within the online environment. These intermediary start-ups offer their users
ilinc Legal & Technology Briefs The Liability of Internet Intermediaries In the EU Many start-ups fulfil an intermediary role within the online environment. These intermediary start-ups offer their users
The Challenge of Implementing Interoperable Electronic Medical Records
 Annals of Health Law Volume 19 Issue 1 Special Edition 2010 Article 37 2010 The Challenge of Implementing Interoperable Electronic Medical Records James C. Dechene Follow this and additional works at:
Annals of Health Law Volume 19 Issue 1 Special Edition 2010 Article 37 2010 The Challenge of Implementing Interoperable Electronic Medical Records James C. Dechene Follow this and additional works at:
ASHVINS Group. Mobile Application Testing Summary
 ASHVINS Group Mobile Application Testing Summary Mobile Application Testing Challenges Unique challenges with mobile applications; device compatibility, OS compatibility, UI compatibility, browser compatibility
ASHVINS Group Mobile Application Testing Summary Mobile Application Testing Challenges Unique challenges with mobile applications; device compatibility, OS compatibility, UI compatibility, browser compatibility
ELECTRONIC MEDICAL RECORDS. Selecting and Utilizing an Electronic Medical Records Solution. A WHITE PAPER by CureMD.
 ELECTRONIC MEDICAL RECORDS Selecting and Utilizing an Electronic Medical Records Solution A WHITE PAPER by CureMD CureMD Healthcare 55 Broad Street New York, NY 10004 Overview United States of America
ELECTRONIC MEDICAL RECORDS Selecting and Utilizing an Electronic Medical Records Solution A WHITE PAPER by CureMD CureMD Healthcare 55 Broad Street New York, NY 10004 Overview United States of America
Thomas Conroy, RPh., J.D. Director, Promotion Compliance Global Regulatory Affairs MARCH 11, 2015
 Thomas Conroy, RPh., J.D. Director, Promotion Compliance Global Regulatory Affairs MARCH 11, 2015 All slides and accompanying comments, ideas, arguments and other statements of any kind are personal to
Thomas Conroy, RPh., J.D. Director, Promotion Compliance Global Regulatory Affairs MARCH 11, 2015 All slides and accompanying comments, ideas, arguments and other statements of any kind are personal to
Medical Device Software
 Medical Device Software Bakul Patel Senior Policy Advisor 1 Overview Medical devices and software Oversight principles and Current approach Trends, Challenges and opportunities Addressing challenges 2
Medical Device Software Bakul Patel Senior Policy Advisor 1 Overview Medical devices and software Oversight principles and Current approach Trends, Challenges and opportunities Addressing challenges 2
Enabling Integrated Care
 Enabling Integrated Care Harnessing personal health systems for better outcomes across the care continuum Briefing Note for a SmartPersonalHealth Workshop WoHIT, Thursday 18 March 2010, 13:00-17:00, Barcelona
Enabling Integrated Care Harnessing personal health systems for better outcomes across the care continuum Briefing Note for a SmartPersonalHealth Workshop WoHIT, Thursday 18 March 2010, 13:00-17:00, Barcelona
The Personality Disorders Service
 The Personality Disorders Service Developing Skills to build A life worth living Page 1 PERSONALITY DISORDERS SERVICE TREATMENT STRUCTURE The recent restructuring of services in Camden & Islington Foundation
The Personality Disorders Service Developing Skills to build A life worth living Page 1 PERSONALITY DISORDERS SERVICE TREATMENT STRUCTURE The recent restructuring of services in Camden & Islington Foundation
FREQUENTLY ASKED QUESTIONS ABOUT THE CVS CAREMARK PRESCRIPTION DRUG PROGRAM
 FREQUENTLY ASKED QUESTIONS ABOUT THE CVS CAREMARK PRESCRIPTION DRUG PROGRAM ABBVIE EMPLOYEES WANT TO KNOW What s New in 2015? AbbVie is making changes in its pharmacy benefit program to ensure our medical
FREQUENTLY ASKED QUESTIONS ABOUT THE CVS CAREMARK PRESCRIPTION DRUG PROGRAM ABBVIE EMPLOYEES WANT TO KNOW What s New in 2015? AbbVie is making changes in its pharmacy benefit program to ensure our medical
Regulations and compliance for enterprise mhealth applications
 Regulations and compliance for enterprise mhealth applications Contents Mobilizing healthcare applications 4 Security Concerns and Challenges 5 Defining the application 'Does your mobile app need FDA approval?
Regulations and compliance for enterprise mhealth applications Contents Mobilizing healthcare applications 4 Security Concerns and Challenges 5 Defining the application 'Does your mobile app need FDA approval?
ehealth The issues that matter
 ehealth The issues that matter Contents Technology outpacing regulation 4 A new frontier for data privacy 6 Product liability and jurisdictional issues 8 Cyber security rules under observation 10 Your
ehealth The issues that matter Contents Technology outpacing regulation 4 A new frontier for data privacy 6 Product liability and jurisdictional issues 8 Cyber security rules under observation 10 Your
Getting Clinicians Involved: Testing Smartphone Applications to Promote Behavior Change in Health Care
 Getting Clinicians Involved: Testing Smartphone Applications to Promote Behavior Change in Health Care Xiaomu Zhou 4 Huntington Street xmyzhou@rutgers.edu Lora Appel 4 Huntington Street lappel@eden.rutgers.edu
Getting Clinicians Involved: Testing Smartphone Applications to Promote Behavior Change in Health Care Xiaomu Zhou 4 Huntington Street xmyzhou@rutgers.edu Lora Appel 4 Huntington Street lappel@eden.rutgers.edu
Testing Mobile Application using Device Cloud
 Testing Mobile Application using Device Cloud Device Cloud and Benefits CSC has partnerships with mobile device cloud services such as DeviceAnywhere Device cloud platform provides an end-to-end solution
Testing Mobile Application using Device Cloud Device Cloud and Benefits CSC has partnerships with mobile device cloud services such as DeviceAnywhere Device cloud platform provides an end-to-end solution
Regulation and Risk Management of Combination Products
 Regulation and Risk Management of Combination Products Katherine Ulman Associate Scientist Global Regulatory Compliance Manager Dow Corning Healthcare Jim Curtis Senior Specialist, Healthcare Applications
Regulation and Risk Management of Combination Products Katherine Ulman Associate Scientist Global Regulatory Compliance Manager Dow Corning Healthcare Jim Curtis Senior Specialist, Healthcare Applications
CDRH Regulated Software
 CDRH Regulated Software An Introduction John F. Murray Jr. CDRH Software Compliance Expert CDRH Regulates Software in the following areas Medical Devices Automation of Production Systems Automation of
CDRH Regulated Software An Introduction John F. Murray Jr. CDRH Software Compliance Expert CDRH Regulates Software in the following areas Medical Devices Automation of Production Systems Automation of
Bipolar Disorder. Some people with these symptoms have bipolar disorder, a serious mental illness. Read this brochure to find out more.
 Bipolar Disorder Do you go through intense moods? Do you feel very happy and energized some days, and very sad and depressed on other days? Do these moods last for a week or more? Do your mood changes
Bipolar Disorder Do you go through intense moods? Do you feel very happy and energized some days, and very sad and depressed on other days? Do these moods last for a week or more? Do your mood changes
Guidance for Industry
 Guidance for Industry Consumer-Directed Broadcast Advertisements Questions and Answers U.S. Department of Health and Human Services Food and Drug Administration Center for Drug Evaluation and Research
Guidance for Industry Consumer-Directed Broadcast Advertisements Questions and Answers U.S. Department of Health and Human Services Food and Drug Administration Center for Drug Evaluation and Research
A Sample Radio Interview
 A Sample Radio Interview By Erik R, 7/5/00 The following is a sample interview that has been put together to help show how to provide a positive approach to answering questions about Narcotics Anonymous.
A Sample Radio Interview By Erik R, 7/5/00 The following is a sample interview that has been put together to help show how to provide a positive approach to answering questions about Narcotics Anonymous.
Breakthrough Lung Cancer Treatment Approved Webcast September 9, 2011 Renato Martins, M.D., M.P.H. Introduction
 Breakthrough Lung Cancer Treatment Approved Webcast September 9, 2011 Renato Martins, M.D., M.P.H. Please remember the opinions expressed on Patient Power are not necessarily the views of Seattle Cancer
Breakthrough Lung Cancer Treatment Approved Webcast September 9, 2011 Renato Martins, M.D., M.P.H. Please remember the opinions expressed on Patient Power are not necessarily the views of Seattle Cancer
ICD-10 Frequently Asked Questions
 ICD-10 Frequently Asked Questions ICD-10 General Overview... 3 What is ICD-10?... 3 Why are we adopting ICD-10?... 3 What are the benefits of the ICD code expansion?... 3 What does ICD-10 compliance mean?...
ICD-10 Frequently Asked Questions ICD-10 General Overview... 3 What is ICD-10?... 3 Why are we adopting ICD-10?... 3 What are the benefits of the ICD code expansion?... 3 What does ICD-10 compliance mean?...
02- DEPARTMENT OF PROFESSIONAL AND FINANCIAL REGULATION. Chapter 8 REGULATIONS RELATING TO ADVANCED PRACTICE REGISTERED NURSING
 02- DEPARTMENT OF PROFESSIONAL AND FINANCIAL REGULATION 380 BOARD OF NURSING Chapter 8 REGULATIONS RELATING TO ADVANCED PRACTICE REGISTERED NURSING SUMMARY: This chapter identifies the role of a registered
02- DEPARTMENT OF PROFESSIONAL AND FINANCIAL REGULATION 380 BOARD OF NURSING Chapter 8 REGULATIONS RELATING TO ADVANCED PRACTICE REGISTERED NURSING SUMMARY: This chapter identifies the role of a registered
Giuseppe Busia Segretario generale Garante per la protezione dei dati personali
 mhealth enablers panel The Health & Wellness @ Mobile World Congress 2015 Giuseppe Busia Segretario generale Garante per la protezione dei dati personali 1 mhealth main concern Mobile Health (mhealth)
mhealth enablers panel The Health & Wellness @ Mobile World Congress 2015 Giuseppe Busia Segretario generale Garante per la protezione dei dati personali 1 mhealth main concern Mobile Health (mhealth)
General Wellness: Policy for Low Risk Devices. Draft Guidance for Industry and Food and Drug Administration Staff
 General Wellness: Policy for Low Risk Devices Draft Guidance for Industry and Food and Drug Administration Staff DRAFT GUIDANCE This guidance document is being distributed for comment purposes only. Document
General Wellness: Policy for Low Risk Devices Draft Guidance for Industry and Food and Drug Administration Staff DRAFT GUIDANCE This guidance document is being distributed for comment purposes only. Document
The role of big data in medicine
 The role of big data in medicine November 2015 Technology is revolutionizing our understanding and treatment of disease, says the founding director of the Icahn Institute for Genomics and Multiscale Biology
The role of big data in medicine November 2015 Technology is revolutionizing our understanding and treatment of disease, says the founding director of the Icahn Institute for Genomics and Multiscale Biology
User Agreement. Quality. Value. Efficiency.
 User Agreement Quality. Value. Efficiency. Welcome to QVuE, the Leaders Network on Quality, Value and Efficiency website sponsored by The Medicines Company. The information provided in this Webinar Series
User Agreement Quality. Value. Efficiency. Welcome to QVuE, the Leaders Network on Quality, Value and Efficiency website sponsored by The Medicines Company. The information provided in this Webinar Series
MEPI Connect An Open Source Graduate Tracking Software System: Resource Requirements to Customize and Implement the System
 MEPI Connect An Open Source Graduate Tracking Software System: Resource Requirements to Customize and Implement the System Version 1.0: January 2015 Michael Drane, Rebecca Bailey, and Rachel Deussom, IntraHealth
MEPI Connect An Open Source Graduate Tracking Software System: Resource Requirements to Customize and Implement the System Version 1.0: January 2015 Michael Drane, Rebecca Bailey, and Rachel Deussom, IntraHealth
NetSuite. Goes Natural. We have spoken to dozens of small business executives about their move to adopt technology.
 NetSuite Goes Natural We have spoken to dozens of small business executives about their move to adopt technology. Certain companies, though, stand out as powerful examples for others who may be considering
NetSuite Goes Natural We have spoken to dozens of small business executives about their move to adopt technology. Certain companies, though, stand out as powerful examples for others who may be considering
Leveraging Partners and Open Source Technology in your Mobility Strategy. emids webinar Thursday, August 11, 2011 1:00 pm 2:00 pm EDT
 Leveraging Partners and Open Source Technology in your Mobility Strategy emids webinar Thursday, August 11, 2011 1:00 pm 2:00 pm EDT Presenters Jerry Buchanan Account Director emids Technologies Ambarish
Leveraging Partners and Open Source Technology in your Mobility Strategy emids webinar Thursday, August 11, 2011 1:00 pm 2:00 pm EDT Presenters Jerry Buchanan Account Director emids Technologies Ambarish
Understanding Medical Device Regulation for mhealth A Guide for Mobile Operators
 for mhealth A Guide for Mobile Operators 1 Foreword Mobile technologies will make a significant contribution to addressing the enormous challenges of healthcare provision worldwide. Early efforts in mobile
for mhealth A Guide for Mobile Operators 1 Foreword Mobile technologies will make a significant contribution to addressing the enormous challenges of healthcare provision worldwide. Early efforts in mobile
Getting Started with iphone SDK, Android and others: Mobile Application Development Create your Mobile Applications Best Practices Guide
 Getting Started with iphone SDK, Android and others: Mobile Application Development Create your Mobile Applications Best Practices Guide Copyright 2008 Mobile Application Development 100 Success Secrets
Getting Started with iphone SDK, Android and others: Mobile Application Development Create your Mobile Applications Best Practices Guide Copyright 2008 Mobile Application Development 100 Success Secrets
EMR Adoption Survey. Instructions. This survey contains a series of multiple-choice questions corresponding to the 5-stage EMR Adoption Model.
 EMR Adoption Survey Instructions This survey contains a series of multiple-choice questions corresponding to the -stage EMR Adoption Model. If the respondent is a physician, ask all questions. If the respondent
EMR Adoption Survey Instructions This survey contains a series of multiple-choice questions corresponding to the -stage EMR Adoption Model. If the respondent is a physician, ask all questions. If the respondent
Guidance for registered pharmacies providing pharmacy services at a distance, including on the internet
 Guidance for registered pharmacies providing pharmacy services at a distance, including on the internet April 2015 The General Pharmaceutical Council is the regulator for pharmacists, pharmacy technicians
Guidance for registered pharmacies providing pharmacy services at a distance, including on the internet April 2015 The General Pharmaceutical Council is the regulator for pharmacists, pharmacy technicians
REGULATIONS AND COMPLIANCE FOR ENTERPRISE MOBILE HEALTH APPLICATIONS
 REGULATIONS AND COMPLIANCE FOR ENTERPRISE MOBILE HEALTH APPLICATIONS Author: Dilip Chatulingath A RapidValue Solutions Whitepaper Contents Mobilizing healthcare applications 01 Security concerns and challenges
REGULATIONS AND COMPLIANCE FOR ENTERPRISE MOBILE HEALTH APPLICATIONS Author: Dilip Chatulingath A RapidValue Solutions Whitepaper Contents Mobilizing healthcare applications 01 Security concerns and challenges
GUIDANCE NOTES FOR MANUFACTURERS OF CLASS I MEDICAL DEVICES
 Foreword GUIDANCE NOTES FOR MANUFACTURERS OF CLASS I MEDICAL DEVICES These guidance notes do not aim to be a definite interpretation of National Laws and/or regulations and are for guidance purpose only.
Foreword GUIDANCE NOTES FOR MANUFACTURERS OF CLASS I MEDICAL DEVICES These guidance notes do not aim to be a definite interpretation of National Laws and/or regulations and are for guidance purpose only.
OPEN MINDS 2012 Planning & Innovation Institute
 Panel: Trish Cavestany, MSN, RN, CCM Director, Clinical Products and Services, Robert Bosch Healthcare Systems Steve Daviss, MD Chair, Dept Psychiatry, Baltimore Washington Medical Center Chair, Committee
Panel: Trish Cavestany, MSN, RN, CCM Director, Clinical Products and Services, Robert Bosch Healthcare Systems Steve Daviss, MD Chair, Dept Psychiatry, Baltimore Washington Medical Center Chair, Committee
To host Ryan Holsopple at Performance Space 122 to imagine and explore new approaches to demand building for contemporary dance and theater
 Legal Name Performance Space 122 Project Title To host Ryan Holsopple at Performance Space 122 to imagine and explore new approaches to demand building for contemporary dance and theater DDCF Grant Number
Legal Name Performance Space 122 Project Title To host Ryan Holsopple at Performance Space 122 to imagine and explore new approaches to demand building for contemporary dance and theater DDCF Grant Number
Organising and planning services for people with a personality disorder
 Organising and planning services for people with a personality disorder A NICE pathway brings together all NICE guidance, quality standards and materials to support implementation on a specific topic area.
Organising and planning services for people with a personality disorder A NICE pathway brings together all NICE guidance, quality standards and materials to support implementation on a specific topic area.
Regulation of Medical Devices involving Software in Australia an Overview
 Regulation of Medical Devices involving Software in Australia an Overview John Jamieson Conformity Assessment Branch Therapeutic Goods Administration PO Box 100, Woden ACT 2606 John.Jamieson@health.gov.au
Regulation of Medical Devices involving Software in Australia an Overview John Jamieson Conformity Assessment Branch Therapeutic Goods Administration PO Box 100, Woden ACT 2606 John.Jamieson@health.gov.au
Certified National Pharmaceutical Representative
 Certified National Pharmaceutical Representative 120 hours Course Overview/Description The Certified National Pharmaceutical Representative (CNPR) online training program was developed in partnership with
Certified National Pharmaceutical Representative 120 hours Course Overview/Description The Certified National Pharmaceutical Representative (CNPR) online training program was developed in partnership with
ENROLLED SENATE BILL No. 92
 Act No. 285 Public Acts of 2014 Approved by the Governor September 23, 2014 Filed with the Secretary of State September 23, 2014 EFFECTIVE DATE: December 22, 2014 STATE OF MICHIGAN 97TH LEGISLATURE REGULAR
Act No. 285 Public Acts of 2014 Approved by the Governor September 23, 2014 Filed with the Secretary of State September 23, 2014 EFFECTIVE DATE: December 22, 2014 STATE OF MICHIGAN 97TH LEGISLATURE REGULAR
Medical App Checker:
 Medical App Checker: Evaluation of Mobile Medical Apps Medical App Checker 1 Explanation 9 Medical App Checker Medical App Checker There are now more than 120,000 mobile medical apps available in the various
Medical App Checker: Evaluation of Mobile Medical Apps Medical App Checker 1 Explanation 9 Medical App Checker Medical App Checker There are now more than 120,000 mobile medical apps available in the various
Under European law teleradiology is both a health service and an information society service.
 ESR statement on the European Commission Staff Working Document on the applicability of the existing EU legal framework to telemedicine services (SWD 2012/413). The European Society of Radiology (ESR)
ESR statement on the European Commission Staff Working Document on the applicability of the existing EU legal framework to telemedicine services (SWD 2012/413). The European Society of Radiology (ESR)
