Mobile Medical Applications
|
|
|
- Patience Chase
- 10 years ago
- Views:
Transcription
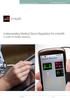
1 Mobile Medical Applications What Is the Impact of FDA s New MMA Guidance for the Life Science Industry? June 6, 2014, 11:15 AM 12:15 PM Presented by: Mark Gardner, M.B.A., J.D.
2 Agenda 1. How does FDA regulate mobile medical applications (MMAs) in light of FDA s new guidance? 2. Practical implications of new guidance What should companies consider when designing, manufacturing and selling MMAs? 3. Updates on MMAs and FDA 4. Questions at the end; please ask as we go too!
3 Martin Cooper invented the cellular phone at Motorola in the 1970s. Things have obviously changed a lot since then!
4 4 Credit: Hykano
5 Mobile Growth is Exponential 500 million users of health care apps by billion users of health care apps by 2018! Some for professional use others for personal
6 HOW DOES FDA REGULATE MOBILE MEDICAL APPLICATIONS?
7
8 At a Base Not all apps are medical devices just because they have health related functions Calorie counters and fitness apps are NOT MMAs Mobile apps that are medical devices by definition may not be regulated as such due to FDA enforcement policy FDA is focusing on high-risk apps apps that will hurt people if they malfunction
9 A Little History 1989 FDA put out its policy on computer products and software Called the FDA Policy for the Regulation of Computer Products, a.k.a., Draft Software Policy It declared software products were medical devices It was withdrawn in 2005
10 More Recent History 2011 Medical Device Data Systems (MDDS) downclassified from Class III medical devices (high-risk) to Class I (low-risk) on 2/15/ Has led to a great deal of interest in the MDDS space Sept. 25: MMA guidance finalized draft guidance was issued 9/21/2011
11 Definitions (are important) Mobile platform Commercial off-the-shelf (COTS) computing platforms Handheld in nature, e.g., mobile phones, tablets, laptops. Mobile Application (Mobile App), e.g.: My Fitness Pal ihealth WebMD Mobile Medical Application (MMA) Meets definition of a medical device under 201(h) of the FD&C Act; AND either: Accessory to medical device; OR Transforms a mobile platform into a medical device
12 Intended Use is Key in Determining Regulatory Classification Intended use determines if an app is a medical device Medical devices are: intended to treat, cure, prevent, mitigate or diagnose disease; OR intended to affect the structure or any function of the body Device definition at 201(h) of the FD&C Act Intended use (21 CFR 801.4) shown by : Labeling claims Advertising materials Oral / written statements Implied or expressed claims
13 Definitions MMA Manufacturer Includes those that create, design, develop, label, re-label, etc., MMAs Or people that commission the creation of MMAs Includes MMAs that can be accessed over the web Non-MMA manufacturers Makers of apps that have no medical device functions Distributors, e.g., itunes, Android market, are not MMA manufacturers Internet providers, cloud hosting HCPs, making apps for patients Research and teaching medical apps (consider IDE regs.)
14 Scope of FDA Guidance FDA focuses on mobile apps that: (1) meet the statutory definition of a device, under 201(h)*; and (2) are either intended: to be used as an accessory to a regulated medical device; or to transform a mobile platform (e.g., iphone) into a regulated medical device *Recall intended use discussion
15 Practical Reality Many app makers out there have no idea they are actually MMA manufacturers They are making medical devices (MMAs) and do not know it! As a result, many MMAs are misbranded and adulterated These companies face legal and regulatory exposure They are also at a competitive disadvantage
16 Practical Reality Companies that market and manufacture MMAs improperly face FDA enforcement risk Warning Letters Consent Decrees Seizure Publicity Business disruption or effective shut-down can be catastrophic
17 Practical Reality If this is news Get into compliance! Determine regulatory classification MMA? Unregulated mobile app? An app FDA will exercise enforcement discretion regarding? MMAs can be Class I (low risk), II (medium) or III (high) depending on FDA Product Code Among other things, register/list, follow Quality System (QS) requirements, 21 CFR 820 Good Manufacturing Practices (GMPs)
18 Types of Apps that are the Focus of FDA s Regulatory Oversight (1) Apps that are an extension of a medical device via connection (wire or wireless) used to control a device or displaying, storing, analyzing or transmitting patient specific device data Examples: Picture Archiving Communication Systems (PACS) Medical Device Data Systems (MDDS) Apps that control devices, e.g., delivery of insulin via a pump determined by an app blood pressure cuffs being run by apps
19 Example Accessory to a Regulated Medical Device Users can check their blood pressure using a regulated medical device and their iphone or ipad The app controls the cuff via the touchscreen The app also tracks information, e.g., daily read outs
20 Types of Apps that are the Focus of FDA s Regulatory Oversight (2) Apps that transform a mobile platform, e.g., your iphone, into a regulated medical device by using: Attachments Display screens Sensors Or other functions of a phone, e.g., the LED light Some that exist now Others on the horizon!
21 Example Transforming a Mobile Platform into a Regulated Medical Device iphones have an LED light that theoretically could be used by a doctor to examine patients Is my iphone now a medical device? It depends! How is the product marketed? If it is marketed to illuminate general objects, then no, probably not a device If it is marketed as a light source for ophthalmologists to examine patients, e.g., an ophthalmoscope, then the app has transformed my phone into a regulated medical device!
22 More Examples Transforming a Mobile Platform into a Regulated Medical Device Attaching: a blood glucose strip reader to my iphone to function as a glucose meter electrocardiograph (ECG) electrodes to my tablet to measure, store and display ECG signals Using built-in components: phone accelerometer for sleep apnea monitoring sensors for creating an electronic stethoscope Displaying radiographic images for diagnosis
23 Types of Apps that are the Focus of FDA s Regulatory Oversight (3) Apps that become regulated medical devices by performing patient-specific diagnosis or treatment recommendations Apps that calculate dosage plans Apps that give treatment recommendations If you want to avoid being regulated under this classification, consider whether you need to provide patient-specific advice
24 What does FDA enforcement discretion actually mean?
25 Apps for which FDA Intends to Exercise Enforcement Discretion Apps that: (1) help patients self-manage their disease without providing specific treatment or suggestions (general coaching for patients); e.g.: Skill of the Day apps for patients with mental challenges (2) provide patients with simple tools to help them track and organize health data e.g., blood pressure tracking tool (3) provide easy access to information related to patients health conditions or treatments e.g., drug allergy look-up tools
26 Apps for which FDA Intends to Exercise Enforcement Discretion Apps that: (4) help patients document, show, or communicate medical conditions to providers; e.g. a camera used for documenting a wound (5) automate simple tasks for providers; OR e.g., BMI calculation (6) enable patients or providers to interact with records. e.g., Personal Health Record (PHR) or Electronic Health Record (EHR) systems
27 Apps that are NOT MMAs Apps that are generic aids or general purpose products, For example: Using an Android phone as a magnifying glass Such apps cannot be not intended for medical purposes not regulated under 21 CFR , Magnifying Spectacles Record audio, amplify audio
28 Apps that are NOT MMAs Apps that: Provide education/training tools not intended to treat, diagnose or be used clinically Maps to special gluten free restaurants Automate general business functions in an office, e.g., apps that: Determine billing codes and facilitate insurance claims Generate appointment reminders Provide wait times in facilities Help patients pay their medical bills
29 Summary of Apps and FDA Regulatory Oversight Mobile apps that FDA regulates Enforcement discretion mobile apps Mobile apps that FDA does NOT regulate Risk to patients? Low, Medium or High Low Low/none Considered a mobile medical application, i.e., a medical device? FDA Clearance or Approval Required? Additional Regulatory Requirements? Yes Maybe No Perhaps, depending on FDA product code Yes QS, register/list, MDR P&Ps, etc. No No, but encouraged to by FDA No No Example of functions? Intended to diagnose, treat, cure, prevent or mitigate disease? -Controls a regulated medical device -Transforms a mobile platform into a device -Data management -Coaching -Reminders -Documentation -Simple calculations Maybe Maybe No -Audio recording, amplification -Education -Business operations
30 Additional Regulatory Requirements for Class I MMA Makers MMR makers of Class I* devices must follow general controls : Register annually and list devices with FDA (21 CFR 807); Registration fees, 2014: $3313 and 2015: $3750 Comply with the Quality System (QS) regulation (21 CFR 820) Following labeling requirements (21 CFR 801); Medical Device Reporting (21 CFR 803); Must report device failures which caused or contributed to death or serious injury or will in the future *Class is dependent on product code Know your code
31 Additional Regulatory Requirements for Class I MMA Makers (continued) MMR makers of Class I devices must follow general controls : Provide premarket notification if necessary (21 CFR 807); Report corrections and removals (21 CFR 806); and Follow the Investigational Device Exemption (IDE) regulations (21 CFR 812)
32 Additional Regulatory Requirements for Class II & III MMA Makers Class II and III MMA makers must follow all general controls for Class I devices which we just covered Plus: Special controls for most Class II devices Premarket Approval (21 CFR 814) for Class III devices
33 PRACTICAL IMPLICATIONS OF NEW GUIDANCE
34 MMA Company Question Checklist Classification Is the app the type that FDA cares about? Or will FDA use enforcement discretion? Can the app be safely used by a patient without active oversight by a medical professional? If something goes wrong with the app, is it going to hurt someone? Documentation supporting: i) the assigned app product code, ii) why the app is not regulated, or iii) reasons why it fits under the enforcement discretion bucket?
35 MMA Company Question Checklist More on Product Codes If my device is acting as an accessory to a regulated device, or is transforming a mobile platform into a regulated medical device then what Product Code does it fall under? Are the appropriate regulations being followed?
36 MMA Company Question Checklist Claims What is the intended use of the app? If it is an MMA, do the claims match my product code? If it is a Class I app, are claims made which go beyond the CFR it is regulated under? Off-label claims?
37 MMA Company Question Checklist Other regulatory requirements met? Quality System established? ISO 9001, ISO 13485, Special Controls? Policies and procedures in place? Registered and products listed? MDR reporting and documentation in place? Following FDA s recommendation to follow Quality System requirements for enforcement discretion apps? Necessary or not?
38 NEWS
39 Latest Intel on MMAs MMA FDA clearance/approval process is lagging The Agency recently defended its tardiness Bakul Patel told the audience that in 80 percent of the cases the agency has met the statutory 90-day timeframe under the 510(k) process Health Data Management Feb. 25, 2014 Some calling for a new legal definition and therefore regulatory class of products, i.e., medical software
40 Latest Intel on MMAs Many new MMAs have been cleared ~100 have been cleared and approved since 1997 Picking up See examples of MMAs FDA Has Cleared or Approved at: ocedures/connectedhealth/mobilemedicalapplications/uc m htm. Many app makers are participating illegally in the market place Investors continue to invest in this new space area of great growth in Silicon Valley
41 The end. Questions? Contact info: Mark Gardner, M.B.A., J.D. (612) Copyright 2014 DuVal & Associates, P.A. All rights reserved.
FDA Issues Final Guidance on Mobile Medical Apps
 ADVISORY September 2013 FDA Issues Final Guidance on Mobile Medical Apps On September 23, 2013, the U.S. Food & Drug Administration (FDA or the Agency) issued its final Guidance for Industry and Food and
ADVISORY September 2013 FDA Issues Final Guidance on Mobile Medical Apps On September 23, 2013, the U.S. Food & Drug Administration (FDA or the Agency) issued its final Guidance for Industry and Food and
CENTER FOR CONNECTED HEALTH POLICY
 CENTER FOR CONNECTED HEALTH POLICY The Center for Connected Health Policy (CCHP) is a public interest nonprofit organization that develops and advances telehealth policy solutions to promote improvements
CENTER FOR CONNECTED HEALTH POLICY The Center for Connected Health Policy (CCHP) is a public interest nonprofit organization that develops and advances telehealth policy solutions to promote improvements
Medical Device Software: Establishing FDA Authority and Mobile Medical Apps
 Medical Device Software: Establishing FDA Authority and Mobile Medical Apps Seth A. Mailhot, Partner Lead, FDA Regulatory Practice Overview Applying the Definition of a Device to Software Special Categories
Medical Device Software: Establishing FDA Authority and Mobile Medical Apps Seth A. Mailhot, Partner Lead, FDA Regulatory Practice Overview Applying the Definition of a Device to Software Special Categories
Regulation of Mobile Medical Apps
 Regulation of Mobile Medical Apps May 30, 2014 Copyright 2014 Software Quality Consulting Inc. Slide 1 Speaker Bio Steven R. Rakitin has over 35 years experience as a software engineer and 25 years in
Regulation of Mobile Medical Apps May 30, 2014 Copyright 2014 Software Quality Consulting Inc. Slide 1 Speaker Bio Steven R. Rakitin has over 35 years experience as a software engineer and 25 years in
Mobile Medical Applications: An Overview of FDA Regulation
 Mobile Medical Applications: An Overview of FDA Regulation RAPS Annual Convention 2014 Austin, Texas Michael A. Swit, Esq. Special Counsel, FDA Law Practice Duane Morris LLP Standard Disclaimers The views
Mobile Medical Applications: An Overview of FDA Regulation RAPS Annual Convention 2014 Austin, Texas Michael A. Swit, Esq. Special Counsel, FDA Law Practice Duane Morris LLP Standard Disclaimers The views
Mobile Medical Application Development: FDA Regulation
 Mobile Medical Application Development: FDA Regulation Mobile Medical Applications: Current Environment Currently over 100,000 mobile health related applications are available for download; there is an
Mobile Medical Application Development: FDA Regulation Mobile Medical Applications: Current Environment Currently over 100,000 mobile health related applications are available for download; there is an
The U.S. FDA s Regulation and Oversight of Mobile Medical Applications
 The U.S. FDA s Regulation and Oversight of Mobile Medical Applications The U.S. FDA s Regulation and Oversight of Mobile Medical Applications As smart phones and portable tablet computers become the preferred
The U.S. FDA s Regulation and Oversight of Mobile Medical Applications The U.S. FDA s Regulation and Oversight of Mobile Medical Applications As smart phones and portable tablet computers become the preferred
The Shifting Sands of Medical Software Regulation
 The Shifting Sands of Medical Software Regulation Suzanne O Shea Ralph Hall September 10, 2014 What Software is Regulated by FDA? FDA regulates medical devices. FDA regulates software that meets the definition
The Shifting Sands of Medical Software Regulation Suzanne O Shea Ralph Hall September 10, 2014 What Software is Regulated by FDA? FDA regulates medical devices. FDA regulates software that meets the definition
Mobile Medical Applications. Guidance for Industry and Food and Drug Administration Staff
 Mobile Medical Applications Guidance for Industry and Food and Drug Administration Staff Document issued on: September 25, 2013 The draft of this guidance was issued on July 21, 2011. For questions regarding
Mobile Medical Applications Guidance for Industry and Food and Drug Administration Staff Document issued on: September 25, 2013 The draft of this guidance was issued on July 21, 2011. For questions regarding
Use of Mobile Medical Applications in Clinical Research
 Use of Mobile Medical Applications in Clinical Research Erin K. O Reilly, PhD RAC Associate Director, Regulatory Affairs Duke Translational Medicine Institute [email protected] September 10, 2014 (919)
Use of Mobile Medical Applications in Clinical Research Erin K. O Reilly, PhD RAC Associate Director, Regulatory Affairs Duke Translational Medicine Institute [email protected] September 10, 2014 (919)
Mobile Medical Apps. Purpose. Diane Romza Kutz Fredric E. Roth V. Regulation and Risks. Purpose of today s presentation
 Mobile Medical Apps Regulation and Risks Diane Romza Kutz Fredric E. Roth V Purpose Purpose of today s presentation Identify the newly-regulated industry Identify the newly regulated products and the basis
Mobile Medical Apps Regulation and Risks Diane Romza Kutz Fredric E. Roth V Purpose Purpose of today s presentation Identify the newly-regulated industry Identify the newly regulated products and the basis
Templates. FDA Mobile Medical App Regulations. Your own sub headline This is an example text. Your Logo
 Templates FDA Mobile Medical App Regulations Your own sub headline This is an example text Your Logo FDA Oversight of Medical Devices The latest Guidance from the FDA Tom Richards MD/MS [email protected]
Templates FDA Mobile Medical App Regulations Your own sub headline This is an example text Your Logo FDA Oversight of Medical Devices The latest Guidance from the FDA Tom Richards MD/MS [email protected]
Risk based 12/1/2015. Digital Health Bakul Patel Associate Director for Digital Health Office of Center Director.
 Digital Health Bakul Patel Associate Director for Digital Health Office of Center Director Center for Devices and Radiological Health 1 Oversight Approach Platform independent Promote innovation Promote
Digital Health Bakul Patel Associate Director for Digital Health Office of Center Director Center for Devices and Radiological Health 1 Oversight Approach Platform independent Promote innovation Promote
Mobile Medical Applications: FDA s Final Guidance. M. Elizabeth Bierman Anthony T. Pavel Morgan, Lewis & Bockius, LLP
 Mobile Medical Applications: FDA s Final Guidance Michele L. Buenafe M. Elizabeth Bierman Anthony T. Pavel Morgan, Lewis & Bockius, LLP 1 Background FDA has a long-standing policy to regulate any computer
Mobile Medical Applications: FDA s Final Guidance Michele L. Buenafe M. Elizabeth Bierman Anthony T. Pavel Morgan, Lewis & Bockius, LLP 1 Background FDA has a long-standing policy to regulate any computer
Developing a Mobile Medical App? How to determine if it is a medical device and get it cleared by the US FDA
 Developing a Mobile Medical App? How to determine if it is a medical device and get it cleared by the US FDA In this presentation: App stats: Explosive growth Examples already cleared by the US FDA Is
Developing a Mobile Medical App? How to determine if it is a medical device and get it cleared by the US FDA In this presentation: App stats: Explosive growth Examples already cleared by the US FDA Is
Medical Device Software
 Medical Device Software Bakul Patel Senior Policy Advisor 1 Overview Medical devices and software Oversight principles and Current approach Trends, Challenges and opportunities Addressing challenges 2
Medical Device Software Bakul Patel Senior Policy Advisor 1 Overview Medical devices and software Oversight principles and Current approach Trends, Challenges and opportunities Addressing challenges 2
Medical Device Data Systems, Medical Image Storage Devices, and Medical Image Communications Devices
 Medical Device Data Systems, Medical Image Storage Devices, and Medical Image Communications Devices Draft Guidance for Industry and Food and Drug Administration Staff DRAFT GUIDANCE This guidance document
Medical Device Data Systems, Medical Image Storage Devices, and Medical Image Communications Devices Draft Guidance for Industry and Food and Drug Administration Staff DRAFT GUIDANCE This guidance document
Breakout Sessions: FDA s Regulation of Mobile Health and Medical Applications
 Breakout Sessions: FDA s Regulation of Mobile Health and Medical Applications 2015 Annual Conference Washington, DC Bakul Patel, Associate Director for Digital Health, Office of Center Director, Center
Breakout Sessions: FDA s Regulation of Mobile Health and Medical Applications 2015 Annual Conference Washington, DC Bakul Patel, Associate Director for Digital Health, Office of Center Director, Center
Thomas Conroy, RPh., J.D. Director, Promotion Compliance Global Regulatory Affairs MARCH 11, 2015
 Thomas Conroy, RPh., J.D. Director, Promotion Compliance Global Regulatory Affairs MARCH 11, 2015 All slides and accompanying comments, ideas, arguments and other statements of any kind are personal to
Thomas Conroy, RPh., J.D. Director, Promotion Compliance Global Regulatory Affairs MARCH 11, 2015 All slides and accompanying comments, ideas, arguments and other statements of any kind are personal to
Cutting Edge Issues in Health Care Technology & mhealth. Agenda
 Cutting Edge Issues in Health Care Technology & mhealth Vernessa Pollard July 2014 Agenda The Regulatory Landscape Jurisdiction and Authority Over Mobile Health Products Requirements, Compliance and Enforcement
Cutting Edge Issues in Health Care Technology & mhealth Vernessa Pollard July 2014 Agenda The Regulatory Landscape Jurisdiction and Authority Over Mobile Health Products Requirements, Compliance and Enforcement
How To Regulate A Medical Device From A Cell Phone
 On Behalf of: InTouch Health White Paper FDA Regulation of Mobile Health Technologies The Current Regulatory Framework as Applied to InTouch Health s Telemedicine Solution June 15, 2012 TABLE OF CONTENTS
On Behalf of: InTouch Health White Paper FDA Regulation of Mobile Health Technologies The Current Regulatory Framework as Applied to InTouch Health s Telemedicine Solution June 15, 2012 TABLE OF CONTENTS
Regulatory Considerations for Medical Device Software. Medical Device Software
 Medtec Ireland 2015 Wireless Medical Devices Regulatory Considerations for Medical Device Software Kenneth L. Block, RAC October 7, 2015 Galway, Ireland Offices: Dallas, Texas (12 employees) Tokyo, Japan
Medtec Ireland 2015 Wireless Medical Devices Regulatory Considerations for Medical Device Software Kenneth L. Block, RAC October 7, 2015 Galway, Ireland Offices: Dallas, Texas (12 employees) Tokyo, Japan
MOBILE MEDICAL APPLICATIONS
 October 7, 2013 EVOKE HEALTH POINT OF VIEW MOBILE MEDICAL APPLICATIONS FDA GUIDANCE FOR INDUSTRY FOR MORE INFORMATION: Mark McConaghy, VP, Strategy Evoke Health 267.765.4998 [email protected]
October 7, 2013 EVOKE HEALTH POINT OF VIEW MOBILE MEDICAL APPLICATIONS FDA GUIDANCE FOR INDUSTRY FOR MORE INFORMATION: Mark McConaghy, VP, Strategy Evoke Health 267.765.4998 [email protected]
On Behalf of: InTouch Health
 On Behalf of: InTouch Health White Paper FDA Regulation of Mobile Health Technologies The Current Regulatory Framework as Applied to InTouch Health s Telehealth Solutions June 15, 2012; Updated June 15,
On Behalf of: InTouch Health White Paper FDA Regulation of Mobile Health Technologies The Current Regulatory Framework as Applied to InTouch Health s Telehealth Solutions June 15, 2012; Updated June 15,
Rethinking the FDA s Regulation of. By Scott D. Danzis and Christopher Pruitt
 Rethinking the FDA s Regulation of Mobile Medical Apps By Scott D. Danzis and Christopher Pruitt Smartphones and mobile devices have rapidly become part of everyday life in the United States. It is no
Rethinking the FDA s Regulation of Mobile Medical Apps By Scott D. Danzis and Christopher Pruitt Smartphones and mobile devices have rapidly become part of everyday life in the United States. It is no
US & CANADA: REGULATION AND GUIDELINES ON MEDICAL SOFTWARE AND APPS OR
 US & CANADA: REGULATION AND GUIDELINES ON MEDICAL SOFTWARE AND APPS OR A MEDICAL DEVICE IS A MEDICAL DEVICE IS A MEDICAL DEVICE AHWP Medical SW Workshop Taipei, Taiwan November 3, 2012 John G. Abbott,
US & CANADA: REGULATION AND GUIDELINES ON MEDICAL SOFTWARE AND APPS OR A MEDICAL DEVICE IS A MEDICAL DEVICE IS A MEDICAL DEVICE AHWP Medical SW Workshop Taipei, Taiwan November 3, 2012 John G. Abbott,
FDA Regulation of Whole Slide Imaging (WSI) Devices: Current Thoughts
 FDA Regulation of Whole Slide Imaging (WSI) Devices: Current Thoughts Clinical Laboratory Improvement Advisory Committee Meeting Centers for Disease Control and Prevention February 15, 2012 Tremel A. Faison,
FDA Regulation of Whole Slide Imaging (WSI) Devices: Current Thoughts Clinical Laboratory Improvement Advisory Committee Meeting Centers for Disease Control and Prevention February 15, 2012 Tremel A. Faison,
EVALUATION OF AUTOMATIC CLASS III DESIGNATION FOR STUDIO on the Cloud Data Management Software DECISION SUMMARY
 A. DEN Number: DEN140016 EVALUATION OF AUTOMATIC CLASS III DESIGNATION FOR STUDIO on the Cloud Data Management Software B. Purpose for Submission: DECISION SUMMARY De novo request for adjunct data management
A. DEN Number: DEN140016 EVALUATION OF AUTOMATIC CLASS III DESIGNATION FOR STUDIO on the Cloud Data Management Software B. Purpose for Submission: DECISION SUMMARY De novo request for adjunct data management
International Medical Device Regulators Forum (IMDRF) US FDA Center for Devices and Radiological Health - Update
 International Medical Device Regulators Forum (IMDRF) US FDA Center for Devices and Radiological Health - Update Kimberly A. Trautman Associate Director, International Affairs Office of the Center Director
International Medical Device Regulators Forum (IMDRF) US FDA Center for Devices and Radiological Health - Update Kimberly A. Trautman Associate Director, International Affairs Office of the Center Director
CDRH Regulated Software
 CDRH Regulated Software An Introduction John F. Murray Jr. CDRH Software Compliance Expert CDRH Regulates Software in the following areas Medical Devices Automation of Production Systems Automation of
CDRH Regulated Software An Introduction John F. Murray Jr. CDRH Software Compliance Expert CDRH Regulates Software in the following areas Medical Devices Automation of Production Systems Automation of
CDRH Regulated Software Looking back, looking forward
 CDRH Regulated Software Looking back, looking forward Medical Device Software Compliance Expert US Food & Drug Administration at the Regulatory Affairs Professional Society Indianapolis, Indiana Goal of
CDRH Regulated Software Looking back, looking forward Medical Device Software Compliance Expert US Food & Drug Administration at the Regulatory Affairs Professional Society Indianapolis, Indiana Goal of
Understanding Medical Device Regulation for mhealth A Guide for Mobile Operators
 for mhealth A Guide for Mobile Operators 1 Foreword Mobile technologies will make a significant contribution to addressing the enormous challenges of healthcare provision worldwide. Early efforts in mobile
for mhealth A Guide for Mobile Operators 1 Foreword Mobile technologies will make a significant contribution to addressing the enormous challenges of healthcare provision worldwide. Early efforts in mobile
Activating Standardization Bodies Around Medical Apps
 Activating Standardization Bodies Around Medical Apps Michael J. Ackerman, Ph.D. Assistant Director High Performance Computing and Communications U.S. National Library of Medicine The views and opinions
Activating Standardization Bodies Around Medical Apps Michael J. Ackerman, Ph.D. Assistant Director High Performance Computing and Communications U.S. National Library of Medicine The views and opinions
REFERENCE DOCUMENT. (DRAFT for Public Comments) Title: White Paper on Medical Device Software Regulation Software Qualification and Classification
 AHWP/WG1/R001:2014 REFERENCE DOCUMENT (DRAFT for Public Comments) Title: White Paper on Medical Device Software Regulation Software Qualification and Classification Author: Work Group 1, Pre-Market Submission
AHWP/WG1/R001:2014 REFERENCE DOCUMENT (DRAFT for Public Comments) Title: White Paper on Medical Device Software Regulation Software Qualification and Classification Author: Work Group 1, Pre-Market Submission
The Human Experiment- Electronic Medical/Health Records
 The Human Experiment- Electronic Medical/Health Records Patient safety is one of the primary stated intentions behind the push for computerized medical records. To the extent illegible handwriting leads
The Human Experiment- Electronic Medical/Health Records Patient safety is one of the primary stated intentions behind the push for computerized medical records. To the extent illegible handwriting leads
Regulation of Medical Apps FDA and European Union
 JHW Comment - The content of below article is intended to provide a general guide to the subject matter. Specialist advice should be sought about your specific circumstances. Erik Vollebregt Erik is a
JHW Comment - The content of below article is intended to provide a general guide to the subject matter. Specialist advice should be sought about your specific circumstances. Erik Vollebregt Erik is a
General Wellness: Policy for Low Risk Devices. Draft Guidance for Industry and Food and Drug Administration Staff
 General Wellness: Policy for Low Risk Devices Draft Guidance for Industry and Food and Drug Administration Staff DRAFT GUIDANCE This guidance document is being distributed for comment purposes only. Document
General Wellness: Policy for Low Risk Devices Draft Guidance for Industry and Food and Drug Administration Staff DRAFT GUIDANCE This guidance document is being distributed for comment purposes only. Document
KIZ 2,33-3 p8 ~ 510(k) Summary. As required by 21 CFR 807.92(c) 510(k) Premarket Notification Submission AFrame Digital MobileCare Monitor.
 KIZ 2,33-3 p8 ~ 510(k) Premarket Notification Submission AFrame Digital MobileCare Monitor 510(k) Summary f 1 As required by 21 CFR 807.92(c) Submitter: 510(k) Owner: AFrame Digital, Inc. Address:.1889
KIZ 2,33-3 p8 ~ 510(k) Premarket Notification Submission AFrame Digital MobileCare Monitor 510(k) Summary f 1 As required by 21 CFR 807.92(c) Submitter: 510(k) Owner: AFrame Digital, Inc. Address:.1889
The Legal Aspects of Regulatory Compliance
 The Legal Aspects of Regulatory Compliance Terry G. Mahn Fish & Richardson P.C. 14-Dec-04 202-783-5070 www.fr.com I. General Theories of Legal Liability Administrative Law FCC * EMC, radio standards, equipment
The Legal Aspects of Regulatory Compliance Terry G. Mahn Fish & Richardson P.C. 14-Dec-04 202-783-5070 www.fr.com I. General Theories of Legal Liability Administrative Law FCC * EMC, radio standards, equipment
Inspections, Compliance, Enforcement, and Criminal Investigations
 Page 1 of 7 Home Inspections, Compliance, Enforcement, and Criminal Investigations Compliance Actions and Activities Warning Letters 2014 Inspections, Compliance, Enforcement, and Criminal Investigations
Page 1 of 7 Home Inspections, Compliance, Enforcement, and Criminal Investigations Compliance Actions and Activities Warning Letters 2014 Inspections, Compliance, Enforcement, and Criminal Investigations
Sarah Chandler A/Head, Regulatory and Scientific Section Medical Devices Bureau [email protected]
 Software Regulated as a Medical Device Sarah Chandler A/Head, Regulatory and Scientific Section Medical Devices Bureau [email protected] Therapeutic Products Directorate Director General S. Sharma
Software Regulated as a Medical Device Sarah Chandler A/Head, Regulatory and Scientific Section Medical Devices Bureau [email protected] Therapeutic Products Directorate Director General S. Sharma
How To Know If A Mobile App Is A Medical Device
 The Regulation of Medical Device Apps Prepared for West of England Academic Health Science Network and University of Bristol June 2014 June 2014 1 Table of Contents 1 Purpose...3 2 Scope...3 3 The Regulation
The Regulation of Medical Device Apps Prepared for West of England Academic Health Science Network and University of Bristol June 2014 June 2014 1 Table of Contents 1 Purpose...3 2 Scope...3 3 The Regulation
Medical Product Software Development and FDA Regulations Software Development Practices and FDA Compliance
 Medical Product Development and FDA Regulations IEEE Orange County Computer Society March 27, 2006 Carl R. Wyrwa Medical Product Development and FDA Regulations Introduction Regulated FDA Overview Medical
Medical Product Development and FDA Regulations IEEE Orange County Computer Society March 27, 2006 Carl R. Wyrwa Medical Product Development and FDA Regulations Introduction Regulated FDA Overview Medical
510(k) Summary May 7, 2012
 510(k) Summary Medicalgorithmics 510(k) Premarket Notification 510(k) Summary May 7, 2012 1. Submitter Name and Address Medicalgorithmics LLC 245 West 107th St., Suite 11A New York, NY 10025, USA Contact
510(k) Summary Medicalgorithmics 510(k) Premarket Notification 510(k) Summary May 7, 2012 1. Submitter Name and Address Medicalgorithmics LLC 245 West 107th St., Suite 11A New York, NY 10025, USA Contact
Guidance for Industry Labeling for Bronchodilators: Cold, Cough, Allergy, Bronchodilator,
 Reprinted from FDA s website by Guidance for Industry Labeling for Bronchodilators: Cold, Cough, Allergy, Bronchodilator, Guidance And Antiasthmatic for Drug Industry Products for Over-the-Counter Labeling
Reprinted from FDA s website by Guidance for Industry Labeling for Bronchodilators: Cold, Cough, Allergy, Bronchodilator, Guidance And Antiasthmatic for Drug Industry Products for Over-the-Counter Labeling
Marketed Unapproved Drugs: FDA to Take Immediate Enforcement Action at Any Time, Without Prior Notice
 Marketed Unapproved Drugs: FDA to Take Immediate Enforcement Action at Any Time, Without Prior Notice Kurt R. Karst Hyman, Phelps & McNamara, P.C. 700 Thirteenth Street, N.W., Suite 1200 Washington, D.C.
Marketed Unapproved Drugs: FDA to Take Immediate Enforcement Action at Any Time, Without Prior Notice Kurt R. Karst Hyman, Phelps & McNamara, P.C. 700 Thirteenth Street, N.W., Suite 1200 Washington, D.C.
Guidance for Industry. Further Amendments to General Regulations of the Food and Drug Administration to Incorporate Tobacco Products
 Guidance for Industry Further Amendments to General Regulations of the Food and Drug Administration to Incorporate Tobacco Products Small Entity Compliance Guide Written comments and suggestions may be
Guidance for Industry Further Amendments to General Regulations of the Food and Drug Administration to Incorporate Tobacco Products Small Entity Compliance Guide Written comments and suggestions may be
ORACLE CONSULTING GROUP
 ORACLE CONSULTING GROUP An Official United States Agent Firm for Foreign Establishments CONSULTING MEMORANDUM: DEALING WITH A MEDICAL DEVICE IN THE U.S. 5398 Golder Ranch Rd., Ste. 1 Tucson, Arizona 85739
ORACLE CONSULTING GROUP An Official United States Agent Firm for Foreign Establishments CONSULTING MEMORANDUM: DEALING WITH A MEDICAL DEVICE IN THE U.S. 5398 Golder Ranch Rd., Ste. 1 Tucson, Arizona 85739
Mobile Medical Apps. 34% of Adults 34% App = Application; = a self contained program or piece of software designed to fulfill a particular purpose;
 Mobile Medical Apps CONSTANTINE GEAN, MD, MS, MBA, FACOEM AOHC 2014 April 30, 2014 DISCLAIMERS ( Aka, My lawyer told me to say nothing I say matters and it s not my fault :) THESE APPS ARE PRESENTED FOR
Mobile Medical Apps CONSTANTINE GEAN, MD, MS, MBA, FACOEM AOHC 2014 April 30, 2014 DISCLAIMERS ( Aka, My lawyer told me to say nothing I say matters and it s not my fault :) THESE APPS ARE PRESENTED FOR
Health Care 2.0: How Technology is Transforming Health Care
 Health Care 2.0: How Technology is Transforming Health Care Matthew Kaiser, CEBS, SPHR Director, HR Technology and Outsourcing Lockton Kansas City, Missouri The opinions expressed in this presentation
Health Care 2.0: How Technology is Transforming Health Care Matthew Kaiser, CEBS, SPHR Director, HR Technology and Outsourcing Lockton Kansas City, Missouri The opinions expressed in this presentation
Building 6, Suite 300 RyeBrook, NY 10573 C-ontact-N~ame: --- William H-. Schofield Director of TeleHealth Operations. Signal,_Radiofrequency
 Optimealth Care Solutions, Inc. Traditional.51/0(k) Opion Telel-ealib Application 510(k) Summary: K130971 Manufacturer Name: Optum Health Care Solutions, Inc. Address:T1oo King Street Building 6, Suite
Optimealth Care Solutions, Inc. Traditional.51/0(k) Opion Telel-ealib Application 510(k) Summary: K130971 Manufacturer Name: Optum Health Care Solutions, Inc. Address:T1oo King Street Building 6, Suite
Leveraging Partners and Open Source Technology in your Mobility Strategy. emids webinar Thursday, August 11, 2011 1:00 pm 2:00 pm EDT
 Leveraging Partners and Open Source Technology in your Mobility Strategy emids webinar Thursday, August 11, 2011 1:00 pm 2:00 pm EDT Presenters Jerry Buchanan Account Director emids Technologies Ambarish
Leveraging Partners and Open Source Technology in your Mobility Strategy emids webinar Thursday, August 11, 2011 1:00 pm 2:00 pm EDT Presenters Jerry Buchanan Account Director emids Technologies Ambarish
AgaMatrucx. NV2 21 Salem, NH 030794oz 2 1. 510(k) Sumnmary
 AgaMatrucx Aga~arix, nc. -7C Ramond venu -, 007S 510(k) Sumnmary This summary of 5 10k) substantial equivalence is being submitted in accordance with the requirements of 21 CER 807.92. Prepared: 05 September
AgaMatrucx Aga~arix, nc. -7C Ramond venu -, 007S 510(k) Sumnmary This summary of 5 10k) substantial equivalence is being submitted in accordance with the requirements of 21 CER 807.92. Prepared: 05 September
Considerations for using the Web for Medical Device Applications
 Considerations for using the Web for Medical Device Applications MEDS, San Diego August 23 rd, 2012 Daniel Sterling, President Who is Sterling? Your Partner in Medical Device Development What we do: o
Considerations for using the Web for Medical Device Applications MEDS, San Diego August 23 rd, 2012 Daniel Sterling, President Who is Sterling? Your Partner in Medical Device Development What we do: o
Regulation and Risk Management of Combination Products
 Regulation and Risk Management of Combination Products Katherine Ulman Associate Scientist Global Regulatory Compliance Manager Dow Corning Healthcare Jim Curtis Senior Specialist, Healthcare Applications
Regulation and Risk Management of Combination Products Katherine Ulman Associate Scientist Global Regulatory Compliance Manager Dow Corning Healthcare Jim Curtis Senior Specialist, Healthcare Applications
Tidepool FDA Pre-submission Draft Meeting Minutes
 Tidepool FDA Pre-submission Draft Meeting Minutes October 14, 2014, 2:00pm EDT Food and Drug Administration, 10903 New Hampshire Ave., WO 66, Silver Spring, MD 20993 Pre submission Document ID: Pre submission
Tidepool FDA Pre-submission Draft Meeting Minutes October 14, 2014, 2:00pm EDT Food and Drug Administration, 10903 New Hampshire Ave., WO 66, Silver Spring, MD 20993 Pre submission Document ID: Pre submission
Regulated Mobile Applications
 Regulated Mobile Applications Sion Wyn Conformity +[44] (0) 1492 642622 [email protected] Waters, Wilmslow, November 2014 1 Introduction According to industry estimates, 500 million smartphone users
Regulated Mobile Applications Sion Wyn Conformity +[44] (0) 1492 642622 [email protected] Waters, Wilmslow, November 2014 1 Introduction According to industry estimates, 500 million smartphone users
User Agreement. Quality. Value. Efficiency.
 User Agreement Quality. Value. Efficiency. Welcome to QVuE, the Leaders Network on Quality, Value and Efficiency website sponsored by The Medicines Company. The information provided in this Webinar Series
User Agreement Quality. Value. Efficiency. Welcome to QVuE, the Leaders Network on Quality, Value and Efficiency website sponsored by The Medicines Company. The information provided in this Webinar Series
Draft Guidance for Industry, Food and Drug Administration Staff, and Clinical Laboratories
 14 1 2 3 4 5 6 7 8 9 10 11 12 13 15 16 17 18 19 20 21 22 23 24 25 26 27 Draft Guidance for Industry, Food and Drug Administration Staff, and Clinical Laboratories Framework for Regulatory Oversight of
14 1 2 3 4 5 6 7 8 9 10 11 12 13 15 16 17 18 19 20 21 22 23 24 25 26 27 Draft Guidance for Industry, Food and Drug Administration Staff, and Clinical Laboratories Framework for Regulatory Oversight of
FDA s Final Compliance Policy Guide for Marketed Unapproved Drugs Is Agency Enforcement at a Crossroads, or Stuck in a Traffic Circle
 FDA s Final Compliance Policy Guide for Marketed Unapproved Drugs Is Agency Enforcement at a Crossroads, or Stuck in a Traffic Circle Kurt R. Karst Associate Hyman, Phelps & McNamara, P.C. 700 Thirteenth
FDA s Final Compliance Policy Guide for Marketed Unapproved Drugs Is Agency Enforcement at a Crossroads, or Stuck in a Traffic Circle Kurt R. Karst Associate Hyman, Phelps & McNamara, P.C. 700 Thirteenth
Turning Patient Portals into Major EHR Assets Edward Fotsch, M.D. Douglas Gentile, M.D.
 Turning Patient Portals into Major EHR Assets Edward Fotsch, M.D. Douglas Gentile, M.D. DISCLAIMER: The views and opinions expressed in this presentation are those of the author and do not necessarily
Turning Patient Portals into Major EHR Assets Edward Fotsch, M.D. Douglas Gentile, M.D. DISCLAIMER: The views and opinions expressed in this presentation are those of the author and do not necessarily
[DOCKET NO.96N-0002] DRAFT
![[DOCKET NO.96N-0002] DRAFT [DOCKET NO.96N-0002] DRAFT](/thumbs/26/7603707.jpg) [DOCKET NO.96N-0002] DRAFT DRAFT DOCUMENT CONCERNING THE REGULATION OF PLACENTAL/UMBILICAL CORD BLOOD STEM CELL PRODUCTS INTENDED FOR TRANSPLANTATION OR FURTHER MANUFACTURE INTO INJECTABLE PRODUCTS DECEMBER,
[DOCKET NO.96N-0002] DRAFT DRAFT DOCUMENT CONCERNING THE REGULATION OF PLACENTAL/UMBILICAL CORD BLOOD STEM CELL PRODUCTS INTENDED FOR TRANSPLANTATION OR FURTHER MANUFACTURE INTO INJECTABLE PRODUCTS DECEMBER,
FDA Regulation of Hearing Aids. Eric A. Mann, MD, PhD Clinical Deputy Director Division of Ophthalmic and ENT Devices ODE/CDRH/FDA
 FDA Regulation of Hearing Aids Eric A. Mann, MD, PhD Clinical Deputy Director Division of Ophthalmic ENT ODE/CDRH/FDA U.S. Food Drug Administration Presentation Outline Overview of device regulations riskbased
FDA Regulation of Hearing Aids Eric A. Mann, MD, PhD Clinical Deputy Director Division of Ophthalmic ENT ODE/CDRH/FDA U.S. Food Drug Administration Presentation Outline Overview of device regulations riskbased
WHITEPAPER: SOFTWARE APPS AS MEDICAL DEVICES THE REGULATORY LANDSCAPE
 WHITEPAPER: SOFTWARE APPS AS MEDICAL DEVICES THE REGULATORY LANDSCAPE White paper produced by Maetrics For more information, please contact global sales +1 610 458 9312 +1 877 623 8742 [email protected]
WHITEPAPER: SOFTWARE APPS AS MEDICAL DEVICES THE REGULATORY LANDSCAPE White paper produced by Maetrics For more information, please contact global sales +1 610 458 9312 +1 877 623 8742 [email protected]
FDA Pre-Submission Cover Letter
 3 physical copies and 1 ecopy to: U.S. Food and Drug Administration Center for Devices and Radiological Health Document Control Center WO66 G609 10903 New Hampshire Avenue Silver Spring, MD 20993 0002
3 physical copies and 1 ecopy to: U.S. Food and Drug Administration Center for Devices and Radiological Health Document Control Center WO66 G609 10903 New Hampshire Avenue Silver Spring, MD 20993 0002
MOBILE TECHNOLOGY IN PATIENT CARE
 MOBILE TECHNOLOGY IN PATIENT CARE Elizabeth Urteaga, Pharm.D. National Wellness Conference June 23, 2014 Learning Objectives Identify mobile instruments available to monitor a patient s disease states
MOBILE TECHNOLOGY IN PATIENT CARE Elizabeth Urteaga, Pharm.D. National Wellness Conference June 23, 2014 Learning Objectives Identify mobile instruments available to monitor a patient s disease states
Lessons Learned from Expanding the Boundaries of the EMR
 Lessons Learned from Expanding the Boundaries of the EMR Phyllis Teater and Thomas Bentley DISCLAIMER: The views and opinions expressed in this presentation are those of the author and do not necessarily
Lessons Learned from Expanding the Boundaries of the EMR Phyllis Teater and Thomas Bentley DISCLAIMER: The views and opinions expressed in this presentation are those of the author and do not necessarily
Section 5: Quality Systems Regulation (QSR) and Good Manufacturing Practice (GMP)
 Section 5: Quality Systems Regulation (QSR) and Good Manufacturing Practice (GMP) 1 Quality Systems Regulation (QSR) 2 Quality Systems Regulation (QSR) and Good Manufacturing Practice (GMP) Sets of checks
Section 5: Quality Systems Regulation (QSR) and Good Manufacturing Practice (GMP) 1 Quality Systems Regulation (QSR) 2 Quality Systems Regulation (QSR) and Good Manufacturing Practice (GMP) Sets of checks
Datrix, Inc NOV - 1 2005. Appendix G. 510(k) Summary of Safety and Effectiveness for Datrix, Inc CardioServer ECG Management System
 Appendix G NOV - 1 2005 510(k) Summary of Safety and Effectiveness for CardioServer ECG Management System 1. DATE SUMMARY PREPARED: 9-26-2005 2. SUBMITTER'S NAME AND ADDRESS: Linda Gluckman 340 State Place
Appendix G NOV - 1 2005 510(k) Summary of Safety and Effectiveness for CardioServer ECG Management System 1. DATE SUMMARY PREPARED: 9-26-2005 2. SUBMITTER'S NAME AND ADDRESS: Linda Gluckman 340 State Place
EU and US regulation of health information technology, software and mobile apps
 EU and US regulation of health information technology, software and mobile apps www.practicallaw.com/3-518-3154 Lincoln Tsang, Vernessa Pollard and Daniel Kracov Arnold & Porter LLP The emergence of integrated
EU and US regulation of health information technology, software and mobile apps www.practicallaw.com/3-518-3154 Lincoln Tsang, Vernessa Pollard and Daniel Kracov Arnold & Porter LLP The emergence of integrated
PREP Course #27: Medical Device Clinical Trial Management
 PREP Course #27: Medical Device Clinical Trial Management Presented by: Evelyn Huang Jeffrey Revello Office of Research Compliance North Shore-LIJ Health System CME Disclosure Statement The North Shore
PREP Course #27: Medical Device Clinical Trial Management Presented by: Evelyn Huang Jeffrey Revello Office of Research Compliance North Shore-LIJ Health System CME Disclosure Statement The North Shore
February 5, 2015. Dear Kristin Pabst,
 DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service Food and Drug Administration 10903 New Hampshire Avenue Document Control Center - WO66-G609 Silver Spring, MD 20993-0002 February 5, 2015, Inc.
DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service Food and Drug Administration 10903 New Hampshire Avenue Document Control Center - WO66-G609 Silver Spring, MD 20993-0002 February 5, 2015, Inc.
Privacy Policy Version 1.0, 1 st of May 2016
 Privacy Policy Version 1.0, 1 st of May 2016 THIS PRIVACY POLICY APPLIES TO PERSONAL INFORMATION COLLECTED BY GOCIETY SOLUTIONS FROM USERS OF THE GOCIETY SOLUTIONS APPLICATIONS (GoLivePhone and GoLiveAssist)
Privacy Policy Version 1.0, 1 st of May 2016 THIS PRIVACY POLICY APPLIES TO PERSONAL INFORMATION COLLECTED BY GOCIETY SOLUTIONS FROM USERS OF THE GOCIETY SOLUTIONS APPLICATIONS (GoLivePhone and GoLiveAssist)
Introduction to Compliance with FDA Labeling and Advertising Requirements
 Introduction to Compliance with FDA Labeling and Advertising Requirements Second Annual Pharmaceutical Industry Regulatory and Compliance Summit Dick Kenny FDA History Basic function of government Oldest
Introduction to Compliance with FDA Labeling and Advertising Requirements Second Annual Pharmaceutical Industry Regulatory and Compliance Summit Dick Kenny FDA History Basic function of government Oldest
