Vision with one eye: a review of visual function following unilateral enucleation
|
|
|
- Theresa Miles
- 10 years ago
- Views:
Transcription
1 Spatial Vision, Vol. 21, No. 6, pp (2008) Koninklijke Brill NV, Leiden, Also available online - Vision with one eye: a review of visual function following unilateral enucleation JENNIFER K. E. STEEVES 1,2,3,4,, ESTHER G. GONZÁLEZ 1,3,4,5 and MARTIN J. STEINBACH 1,2,3,4,5 1 Centre for Vision Research, York University, Toronto, Canada 2 Department of Psychology, York University, Toronto, Canada 3 Department of Ophthalmology and Vision Sciences, University of Toronto, Canada 4 The Hospital for Sick Children, Toronto, Canada 5 Vision Science Research Program, Toronto Western Hospital, Toronto, Canada Received 23 April 2007; accepted 14 June 2007 Abstract What happens to vision in the remaining eye following the loss of vision in the fellow eye? Does the one-eyed individual have supernormal visual ability with the remaining eye in order to adapt and compensate for the loss of binocularity and the binocular depth cue, stereopsis? There are subtle changes in visual function following the complete loss of one eye from unilateral enucleation. Losing binocularity early in life results in a dissociation in form perception and motion processing: some aspects of visual spatial ability are enhanced, whereas motion processing and oculomotor behaviour appear to be adversely affected suggesting they are intrinsically linked to the presence of binocularity in early life. These differential effects may be due to a number of factors, including plasticity through recruitment of resources to the remaining eye; the absence of binocular inhibitory interactions; and/or years of monocular practice after enucleation. Finally, despite this dissociation of spatial vision and motion processing, research that has examined visual direction and performance on monocular tasks shows adaptive effects as a result of the loss of one eye. Practically speaking, one-eyed individuals maintain perfectly normal lives and are not limited by their lack of binocularity. Keywords: Enucleation; monocular deprivation; spatial vision; motion processing; visual direction. INTRODUCTION It is a popular belief that losing the ability to use one sensory system results in a sharpening of the other remaining senses. For example, the typical layperson might hold the belief that a person who is completely blind will have more acute hearing than someone with full vision. This is an empirical question are blind To whom correspondence should be addressed. [email protected]
2 510 J. K. E. Steeves et al. people more perceptive to barely audible sounds or are they able to hear a wider range of frequencies than normally sighted individuals? Looking at another sensory modality and using functional brain imaging, research on visual ability in deaf individuals with hearing loss early in life has demonstrated cortical changes in functional activation for visual stimuli. Specifically, enhanced sensitivity in multimodal areas in early deaf individuals has been shown (Bavelier et al., 2001). This would suggest that losing hearing early in life has allowed for adaptive cortical reorganization. In a more recent paper, however, Bavelier and colleagues (2006) note that the story is not completely straightforward deaf individuals exhibit both enhanced and diminished visual ability that is selective to specific visual capacities compared to hearing controls. In short, enhanced visual skills in deaf individuals is not widespread but rather, limited to those visual abilities that are attentionally demanding and that would normally benefit from a convergence of sensory information from both the auditory and visual domains. Consider the case of losing just one eye. A similar question can be asked; does the remaining eye compensate for the loss of binocularity and lead to enhanced visual function with the remaining eye? Here we review human behavioural studies of visual performance in individuals who have complete monocularity following the loss of one eye (unilateral enucleation). The majority of the research has been done on individuals who have lost one eye early in life, during postnatal visual development, but a few studies have examined the loss of one eye later in life. It is important to note that unilateral enucleation is unique in that it results in the most complete form of deprivation because the brain has absolutely no visual input from that eye once the end organ has been removed. This is unlike other forms of monocular visual deprivation such as cataract, strabismus, ptosis or anisometropia that leave some, frequently abnormal, visual input. Complete monocular deafferentation provides a unique human model for examining the consequences of the loss of binocularity. Do one-eyed individuals see better with the remaining eye? The answer is similar to the findings from Bavelier s work on early deaf individuals, both yes and no. Losing one eye leads to both enhanced and reduced visual function depending upon the visual capacity that is being measured and also on the age of the individual at the time of the loss. This dissociation in visual performance appears to lie in whether one is measuring visual spatial ability or visual motion processing and oculomotor systems. That is, some aspects of visual spatial ability appear to be enhanced by the loss of binocularity; however, motion processing and oculomotor behaviour appear to be intrinsically linked to normal binocularity. Here, we review the findings of studies that have specifically examined the visual consequences of unilateral enucleation, the complete loss of one eye, on spatial vision and motion systems as well as on visual direction and performance during monocular tasks. We also discuss the issue of what is the appropriate control comparison group and how best to test this group. Finally, we conclude with a brief overview of physiological mechanisms that could account for cortical changes
3 Vision with one eye 511 following deafferentation of one eye and could lead to either enhanced or impaired visual function in the remaining eye. We begin with spatial vision, which on the whole shows either enhanced or equivalent performance in one-eyed observers compared to binocularly-intact controls. SPATIAL VISION All of the studies that have examined visual performance in one-eyed individuals and are described below and are outlined in Table 1A (spatial vision) and Table 1B (motion processing tasks). Nicholas et al. (1996) tested contrast sensitivity in unilaterally enucleated adults and found that they had higher contrast sensitivity than controls viewing monocularly at 2, 4 and 8 c/deg. At 4 c/deg, those who were enucleated at 13 months or earlier had a peak sensitivity that was on the order of 3.5 times better than controls viewing with the better eye. Table 2 shows the approximate improvement in performance of one-eyed individuals compared to controls for spatial vision tasks. Moreover, when the enucleated observers were categorized by age at enucleation there was a developmental relationship those who had been enucleated before two years of age had better contrast sensitivity at 4 c/deg than those enucleated much later in life and control observers viewing binocularly. These differences in contrast sensitivity show a critical period such that earlier enucleation leads to larger improvement in contrast sensitivity. These findings suggest that contrast sensitivity develops at different rates for different spatial frequencies and, further, that there is a period in early postnatal visual development where removal of an eye is followed by an improvement in contrast sensitivity at some spatial frequencies. In a similar vein, Reed et al. (1996) examined acuity for letters defined by low to high luminance contrast (Regan, 1988). Unilaterally enucleated observers showed better letter recognition than normally sighted controls viewing monocularly and strabismic observers viewing with their better eye. At the lower contrasts, the one-eyed individuals had better acuity compared to controls by 41 60% (see Table 2). Enucleated observers, however, had similar performance to controls viewing binocularly (Reed et al., 1997). Similarly, González et al. (2002) tested acuity for luminance contrast-defined illiterate E optotypes, both centrally and peripherally (7 eccentricity). Consistent with the findings of Reed and colleagues (1996, 1997), for central viewing the enucleated observers demonstrated better acuity at high and low contrast levels than controls viewing monocularly but comparable acuity to that of controls viewing binocularly. With peripheral viewing, enucleated observers showed a small acuity difference in favour of the temporal hemifield. These one-eyed observers had better acuity than the better eye of strabismic observers (amblyopic or non-amblyopic) even though all groups had comparable foveal decimal acuity of 1.0 or better. Moreover, the enucleated group had better low contrast acuity, by approximately 95% (see Table 2) and similar high contrast acuity compared to the control group viewing monocularly. Because
4 512 J. K. E. Steeves et al. Table 1A. A summary of visual ability of one-eyed observers compared to binocularly-intact controls with binocular or monocular viewing for spatial vision. Performance is grouped into enhanced equivalent performance Visual ability Performance relative to controls Enhanced Equivalent Binocular Monocular Binocular Monocular Contrast sensitivity at 4 cpd if enucleated before age 2 at 2, 4 and 8 cpd (Nicholas et al., Cowey, 1996) Low-high contrast letter acuity (Reed et al., 1996, 1997) Eccentric (7 ) illiterate E acuity at low contrast at high contrast (González et al., 2002) Vernier acuity high contrast (Freeman and Bradley, 1980; Schwartz et al., 1987) Vernier acuity medium contrast poorest for children <8 years of age (González et al., 1992) Global pattern discrimination low-medium contrast (Steeves et al., 2004) Texture-defined (second-order) Trend for better detection letter detection/recognition (Steeves et al., 2002) Troxler fading (González et al., 2007)
5 Vision with one eye 513 Table 1B. A summary of visual ability of one-eyed observers compared to binocularly-intact controls with binocular or monocular viewing for motion processing tasks. Performance is grouped into equivalent or reduced performance. Checkmarks indicate how performance compares for each visual ability relative to controls Visual ability Performance relative to controls Equivalent Reduced Binocular Monocular Binocular Monocular Relative motion (shear) sensitivity motion detection reversed velocity discrim. bias in upper/lower VF (Bowns et al., 1994) Motion coherence direction discrimination nasalward bias (Steeves et al., 2002) Motion-defined (second-order) worse recognition Trend for worse recognition letter detection/recognition (Steeves et al., 2002) Motion in depth (time to collision estimation) (Steeves et al., 2000) mveps (Day, 1995) OKN (Day, 1995; Reed et al., 1997)
6 514 J. K. E. Steeves et al. Table 2. Performance ratios of one-eyed individuals relative to controls where there was a significant enhancement in visual ability for spatial vision tasks. Ratios were calculated as the mean performance of the one-eyed group/mean performance of the control group. Approximate ratios from published tables and graphs were computed when raw data were unavailable Visual ability Approximate performance ratio of one-eyed individuals relative to controls (mean one-eyed group performance/ mean performance controls) Contrast sensitivity At 4 cpd Eye-patched controls (better eye) Binocular controls (Nicholas et al.,1996) Very early enucleated (0 13 mos) Early enucleated (16 44 mos) Late enucleated (11 13 years) 1.58 n.s. Low-high contrast letter acuity Contrast (%) Eye-patched controls (Reed et al., 1996, 1997) Eccentric (7 ) illiterate E acuity Contrast (%) Eye-patched controls (González et al., 2002) Vernier acuity high contrast Eye-patched controls (poorer eye) Eye-patched controls (better eye) (Freeman and Bradley, 1980) Global pattern discrimination Contrast (%) Dichoptic controls Eye-patched controls (Steeves et al., 2004) Troxler fading times Contrast Eye-patched controls Binocular controls (González et al., 2007) Exp. 1 High 1.43 n.s. Medium 1.28 n.s. Low Exp. 2 Low
7 Vision with one eye 515 the temporal hemifield has been shown to develop earlier than the nasal hemifield (Lewis and Maurer, 1992) it is likely that early unilateral enucleation has a greater effect on the earlier-developing nasal retina, to its benefit. This favourable effect may be due to the complete absence of binocular competitive mechanisms during its maturation thereby allowing the nasal retina a neural advantage and therefore, enhanced temporal field acuity. Freeman and Bradley (1980) measured high contrast vernier acuity in adults with unilateral enucleation or unilateral amblyopia who had normal Snellen acuity in the functional or remaining eye. They found that the monocular individuals had higher vernier acuities than binocularly normal observers tested monocularly. Their oneeyed individual had a vernier offset threshold that was 55% better than the mean of controls viewing with the better eye (see Table 2 for a summary). This finding of enhanced vernier acuity in monocular individuals, however, was not replicated in unilaterally enucleated children when vernier acuity was tested at a medium level of contrast (González et al., 1992). Vernier acuity was also no different in the non-deprived eye of an individual with unilateral cataract compared to the patient s identical twin (Johnson et al., 1982). Since vernier acuity is known to greatly improve with practice (McKee and Westheimer, 1978; Poggio et al., 1992) it is possible that the findings of Freeman and Bradley are the result of using highly trained monocular observers (optometry students) and relatively untrained controls. González and colleagues, however, demonstrated a significant effect of age at testing such that younger observers had poorer resolution than older observers. From these data, it appears that vernier acuity reaches adult levels at 8 9 years of age and others have shown that it approaches adult levels at 5 years of age (Zanker et al., 1992). Similar findings were found by Schwartz et al. (1987) regarding age at enucleation and vernier acuity. Steeves et al. (2004) tested another form of hyperacuity in adult one-eyed observers. They tested performance for global shape discrimination by measuring discrimination of small deviations from circularity using radial frequency patterns at high and low contrast (Wilkinson et al., 1998). Control observers were tested in two monocular conditions: (1) dichoptic viewing a luminance-matched grey field was shown to the non-viewing eye in an attempt to optimize monocular performance and (2) wearing a black eye patch over the non-viewing eye. Sensitivity to low-contrast global shape was equivalent in unilaterally enucleated observers and binocularlyviewing controls. However, both binocularly-viewing controls and enucleated observers showed superior performance compared to controls viewing in either monocular condition. While enhancement in performance was not as large as that shown with contrast sensitivity or low contrast letter acuity, significant improvement relative to controls on this task ranged from 2 to 11% (see Table 2). At low contrast, the dichoptic control group was more sensitive than controls wearing the black eye patch, which suggests that dichoptic viewing is a superior method for testing controls monocularly. If differences in retinal illumination degrade monocular performance for binocularly-intact controls, presenting a feature-
8 516 J. K. E. Steeves et al. less field of equivalent brightness might be a better way of testing control observers monocularly rather than using the traditional black eye patch, which lowers the illumination to the non-viewing eye. This is likely the case since hyperacuity thresholds at low contrast were improved for the dichoptic compared to the eye patch monocular control condition. It is remarkable that, nonetheless, the enucleated observers exhibited superior performance at low contrast compared to this dichoptic viewing control group. High-contrast second-order texture-defined letter detection and discrimination (Regan and Hong, 1994) was compared between enucleated observers and monocularly- and binocularly-viewing controls (Steeves et al., 2002). On this spatial vision task, the enucleated observers showed no significant difference in performance compared to controls, although the one-eyed observers did show a trend for better detection of texture-defined letters compared to monocularly-viewing controls. It would be worthwhile to measure low-contrast performance for this task since it requires somewhat higher-level spatial integration, being a second-order spatial task. Recently, Troxler fading was measured in enucleated observers compared to controls (González et al., 2007). Time to fading for all observers was a function of brightness contrast. It was significantly faster with monocular (i.e. patched) than with binocular viewing, for the controls. In contrast, one-eyed observers showed equivalent time to fading compared to binocular viewing controls. Moreover, they showed significantly longer fading times than the two-eyed observers viewing monocularly, on the order of three times longer for low contrast stimuli (see Table 2). A control experiment showed that these findings were not due to worse fixation stability, larger pupil sizes, or an unusually large blinking rate in the enucleated group. Enucleated observers exhibited a slight miosis which, if replicated with a larger sample, would actually indicate that, rather than mimicking the consensual pupilary response of closing one eye, the visual system opts instead for a larger depth of field and reduced optical aberrations for the remaining eye. One would expect, therefore, that the fading times of the enucleated observers would be shorter than those of the monocularly viewing controls because the retinal image is, in fact, dimmer and as a result, likely to fade faster. This, however, is not the case, suggesting differences between the groups at a cortical level. To summarize the data from studies of spatial vision, visual ability of one-eyed observers is by and large enhanced compared to two-eyed controls. This is most often the case for tests that have used low contrast stimuli. Enhanced visual ability has been demonstrated for detection tasks such as contrast sensitivity, letter acuity and for some hyperacuity tasks as well as for the phenomenon of Troxler fading. The findings from these studies are summarized in Tables 1A and 2. It is possible that binocular interactions in the controls may have little effect at higher contrast but are more evident with low contrast stimuli. It is important to note that when experimenters attempted to minimize binocular interactions for the controls with the use of dichoptic viewing, the enucleated observers nevertheless performed better than the controls. Enhanced spatial visual ability in one-eyed observers could be
9 Vision with one eye 517 due to the removal of inhibitory binocular interactions that are known to underlie the tuning of retinal disparity (Poggio et al., 1998) and binocular rivalry (Fox, 1991). With respect to several of the lower contrast spatial vision tasks, enucleated observers indeed appear to have compensated for the loss of binocularity. These data are opposite from the deficits in spatial vision that are seen with other forms of visual deprivation such as cataract, strabismus, ptosis and anisometropia (e.g. Mansouri and Hess, 2006; McKee et al., 2003), suggesting that unilateral enucleation is a very distinct form of monocular visual deprivation. MOTION PROCESSING AND OCULOMOTOR SYSTEMS While visual spatial ability tends to be enhanced with the loss of one eye, motion processing and oculomotor performance tends to be adversely affected by it. Motion processing has been examined in one-eyed observers on a number of different tasks and is summarized in Table 1B. Bowns et al. (1994) examined relative motion discrimination for shearing stimuli in the upper and lower visual field. Stimuli were a textured surface with a discontinuity between the upper and lower field that was defined by relative speed differences but moved in the same direction. They found that early enucleated adults and binocularly intact age-matched controls have similar thresholds for detecting relative motion. These groups, however, exhibit opposite biases in the perceived velocity of stimuli in the upper and lower hemifields. Controls were more likely to judge the upper hemifield as faster while enucleated observers judged the bottom hemifield as faster. Bowns et al. hypothesized that if one-eyed observers or other subjects with weak stereopsis attempt to use motion parallax (a system used for far space) as a substitute for stereoscopic information (a system used for near space), a reversal of the normal visual field bias (Previc, 1990; Skrandies, 1987) could occur. In fact, González et al. (1989) measured depth perception in a test modified from the standard Howard Dolman depth perception test in which the only cue for depth was motion parallax. One might expect that enucleated observers would be superior in the use of monocular cues for depth, since depth from stereopsis is not available to them. Surprisingly, one-eyed young children do not spontaneously make lateral movements of the head when determining depth and as a result they show poorer depth perception compared to controls. Subsequently, however, González and colleagues instructed the children to move the head from side to side and retested their thresholds. In this case, depth perception from motion parallax in these younger one-eyed observers was comparable to that of an older control group viewing binocularly. These findings stand in contrast with those of Marotta et al. (1995), who found larger and faster head movements in enucleated subjects in a reaching and grasping task. It is important to note, however, that this discrepancy can be explained by the significant age difference in participants in the two studies the subjects in the study by González et al. (1989) were young (mean age = 12 years) while those
10 518 J. K. E. Steeves et al. in the paper by Marotta et al., were much older (mean age = 32.4 years). Further, Marotta and colleagues remarked that the proportion of self-generated lateral and vertical head movements versus forward head movements increases as a function of post-enucleation time, which is also consistent with the age differences between the two studies. It is likely that one-eyed individuals learn to increase the proportion of lateral and vertical head movements to make better use of motion parallax while reducing forward head movements that produce less helpful information for estimating depth (Marotta et al., 1995; Simpson, 1993). Similar age-related findings with depth perception in one-eyed observers were reported by Schwartz et al. (1987). The use of the monocular motion-in-depth cue tau (τ), the angular rate of expansion of a looming object (Hoyle, 1957), for the perception of time to contact was measured in one-eyed observers by Steeves et al. (2000). Again, as with motion parallax, this monocular cue for motion in depth might sometimes be useful in everyday life since many objects have familiar sizes. Surprisingly, however, enucleated observers cannot estimate time to collision (TTC) of an approaching object based on this monocular cue better than the controls. In fact, the majority of oneeyed observers were worse, showing larger errors in estimating TTC than controls viewing monocularly. Interestingly, most one-eyed individuals relied on other taskirrelevant variables such as the stimulus starting size. Such variables, although not relevant to this task, would be useful in the real world where objects have familiar sizes. This study provides evidence that one-eyed observers learn to use as many optical variables as possible to compensate for the lack of binocular information. The ability of one-eyed observers to recognize letters defined by second order motion the relative motion of elements within the boundary of the letter to that of elements in the background (Regan and Hong, 1990) is also significantly poorer than binocularly viewing controls (Steeves et al., 2002). When compared to monocularly viewing controls, their performance is equivalent but shows a trend for poorer recognition. Developmental data have shown that sensitivity to form-frommotion contrast has a longer developmental time course than that for luminance contrast (Giaschi and Regan, 1997). Together with the developmental evidence, the data from one-eyed observers suggest that the loss of binocularity early in life is operating on two distinct processing mechanisms (spatial vision versus motion processing) and as a result affects them differentially. Other findings with respect to motion processing and oculomotor performance demonstrate that, unlike spatial vision, these systems are adversely affected by the loss of binocularity. Poor motion processing is consistent with data from subjects with deficient stereopsis (see Tychsen, 1993, for a review). Steeves et al. (2002) examined left right direction discrimination in a motion coherence task. While binocularly intact controls showed no asymmetry in direction discrimination, the enucleated group showed significantly higher temporalward than nasalward motion coherence thresholds. Moreover, this nasalward bias was absent in the subject who had undergone enucleation at the oldest age (43 months). All other one-eyed
11 Vision with one eye 519 observers were enucleated before 36 months of age, suggesting a critical period for the role of binocularity in the development of horizontal motion discrimination. This perceptual asymmetry in direction discrimination is consistent with both sensorimotor and cortical motion processing asymmetries that have been demonstrated in early enucleated observers by others. For example, Reed et al. (1991) measured optokinetic nystagmus (OKN) in early unilaterally enucleated observers and found that 63% had small but significant asymmetries of OKN, favouring nasally-directed motion in the visual field. Day (1995) compared OKN and motion visual evoked potentials (mveps) to horizontally moving vertical sinusoidal gratings in different monocular populations. Day found that 25% of observers with early enucleation had asymmetrical OKN and further that 17% of observers showed no optokinetic response at all. Higher motion VEP asymmetries were seen in enucleated observers who lost the eye at a young age compared to those who had lost vision as an adult or to those who were congenitally monocular. These sensorimotor and cortical motion asymmetries reflect a critical period that has been demonstrated for the development of symmetrical motion processing in young infants. For instance, normal human infants tend to show more OKN to motion that is moving nasally than temporally when viewing moving stimuli monocularly (Atkinson and Braddick, 1981; Naegle and Held, 1982) and develop more symmetrical OKN (similar to that of an adult) at around 5 to 6 months of age (Naegle and Held, 1982). Similarly, Norcia et al. (1991) examined mveps to horizontally moving vertical sinusoidal gratings in young infants from 2 to 26 weeks of age compared to normal adults. They also found directional asymmetries in favour of nasally-directed stimuli. This evidence suggests that the maturation of cortical mechanisms is involved in the development of symmetrical motion responses and that there is a critical period for normal maturation. VISUAL DIRECTION AND PERFORMANCE ON MONOCULAR TASKS We have two eyes yet experience a singular view of the world the signals from the two eyes are integrated and projected to this egocentre without the observer s having any conscious eye of origin information (Steinbach et al., 1985). What does the one-eyed observer experience, since the only viewing eye is displaced laterally with respect to the midline of the body? Do they adapt the egocentre accordingly? While developing the laws of visual direction Hering (1942/1879) predicted that the origin of visual direction, or egocenter, would shift towards the remaining eye, if one eye was lost. Moidell et al. (1988) measured the position of the midline relative to the remaining eye in children with one eye. Children were asked to align a nonvisible rod, using their hands, with a visible fireman s hose that was aligned to the visual axis of the remaining eye. Binocular children with one eye covered, aligned the rod with their midline. The one-eyed children (five years of age and older), however, aligned the rod close to the position of the remaining eye (approximately 75% of the distance between the midline and the remaining eye), suggesting that the midline was plastic and had shifted toward the visual centre relative to the
12 520 J. K. E. Steeves et al. body. In this study, children were enucleated up to four years of age and there was no relationship to egocentre position and age at enucleation. This suggests that the egocentre is plastic at least up to four years of age. To test this hypothesis, healthy binocular adults were monocularly patched for a one month period, and showed virtually no change in egocentre location (Dengis et al., 1992). Further, adults who underwent monocular enucleation after a lifetime of normal binocular vision show limited plasticity in egocentre location (unpublished observations, Dengis and Steinbach). Barbeito (1983) confirmed a clinical observation that young children will place a tube between their two eyes when asked to look through it at a target. He found this cyclops effect in children aged 3 4 years. How do age and visual experience play a role in the cyclops effect, since most adults can use a tube for sighting by effortlessly placing it over one eye? Dengis and colleagues (1993) used the look through a tube technique in three groups binocularly normal; unilaterally enucleated; and strabismic children. They found that the egocentre was essentially built-in and children in all groups sighted with the tube midway between the two eyes. This effect was present in the youngest children who could be tested (1.1 years). Interestingly, the presence of normal binocular experience was not necessary for the cyclops response. For all groups, the cyclops effect diminished as they grew older and children sighted monocularly by about four years of age. Using different types of tubes for sighting, Dengis et al. (1996) examined the emergence of monocular sighting as it develops from the cyclops effect. Children progress through a series of behavioral stages that ultimately lead to the adult performance, which is monocular sighting with the non-sighting eye closed and the head straight. Turning the head, or face turn as it is sometimes called in the clinical literature, frequently occurs in one-eyed children (Goltz et al., 1997). Head turn almost always occurred with the head turned so as to bring the remaining eye closer to the midline of the body. This head turn also has the beneficial effect of reducing occlusion of the visual field by the nose. Others have described a head turn in the opposite direction in five patients with esotropia following unilateral enucleation (Helveston et al., 1985). Unlike the one-eyed observers in the study by Goltz et al. (1997) who showed no nystagmus, these patients had esotropia and the presence of an abduction nystagmus with an extreme adduction null point (a position of the eye in the orbit where the nystagmus dampens). Every monocular task we perform includes immediate feedback about its success. If we try to look through a microscope, we know from tactile and visual feedback whether or not the preferred eye is aligned with the eye piece. What happens in the absence of this feedback? Dengis et al. (1998) instructed adults and children to monocularly sight through a tube but they had placed a liquid crystal shutter directly in front of the subject s face. As soon as the subject initiated the movement of the tube toward the eye, the shutter became opaque, thereby preventing any visual feedback. At the same time, the glass plate of the shutter also prevented any tactile
13 Vision with one eye 521 feedback about where on the face the tube might have touched. The results were surprising: the tube was placed at the midline by adults and children with normal binocular visual development as well as by those with strabismus. The enucleated subjects, on the other hand, all placed the tube over their remaining eye. This finding suggests that binocular observers (both normal and strabismic) revert to egocentre sighting in the absence of visual and tactile feedback but that the egocentre shift in one-eyed observers has truly been re-wired toward the remaining eye. These results suggest that our orienting responses, when moving ourselves through space, use the midline egocentre as the origin from which we judge direction (Ono and Mapp, 1995; Ono et al., 2002). Only when we are forced into a monocular task, and have feedback about how we perform that task, do we use a learned pattern of responses developed with a preferred eye. It would be worthwhile to study orienting behaviour in one-eyed observers using more natural tasks this was tried in the past with some success (Dengis et al., 1995; González et al., 1999), showing better accuracy in one-eyed observers consistent with a shift in egocentre toward the remaining eye. POSSIBLE MECHANISMS OF VISUAL PLASTICITY FOLLOWING ENUCLEATION At least three kinds of processes may lie behind the dissociation in visual performance of enucleated observers in spatial vision and motion processing systems, some of which have been mentioned above. These are: (a) plasticity through recruitment by the remaining eye of the resources normally assigned to the missing eye, (b) the absence of binocular inhibitory interactions resulting from the removal of one eye and (c) monocular practice over the years after enucleation. Plasticity The visual system has been shown to exhibit a remarkable plasticity in response to visual deprivation in animals (see reviews in Daw, 1995; Fox and Wong, 2005; Karmarkar and Dan, 2006; Kiorpes and Movshon, 2003; Krahe et al., 2005). Specifically, various physiological changes have been demonstrated in animals following unilateral enucleation, many of which suggest that the remaining eye may make use of the deafferented cells to its advantage. This has been called recruitment, since visually driven brain cells driven normally by the enucleated eye may have been recruited by the remaining eye. There is evidence of recruitment after deprivation, which increases the cortical space innervated by the remaining eye. Cells dominated partially or completely by one eye undergo a reorganization after lack of visual input and become primarily responsive to the other eye (Gilbert and Wiesel, 1992; Hubel and Wiesel, 1962; Hubel et al., 1977; Kratz and Spear, 1976). In addition, monocular enucleation reduces apoptosis in ganglion cells in the remaining eye and preserves or even expands their central connections (Guillery,
14 522 J. K. E. Steeves et al. 1989). This reorganization can occur within hours, depending on the nature of the lesion (Schmid et al., 1995) and can involve other sensory modalities (Kahn and Krubitzer, 2002; Kujala et al., 2000). One study has shown physiological evidence in humans for cortical reorganization following unilateral enucleation during infancy for tumour. Horton and Hocking (1998a) showed a lack of ocular dominance columns in the striate cortex of children with early monocular enucleation. Binocular competition that is required for normal formation of the visual system does not necessarily require visual experience, since competition for cortical space and synaptic formation may begin before birth. For example, monocular enucleation that takes place prenatally rather than postnatally appears to produce more significant changes in the functional properties of cortical neurons in favour of the remaining eye. Prenatally enucleated macaque monkeys show an absence of ocular dominance columns (Rakic, 1981). Prenatally enucleated ferrets show a disruption in the formation of the fibres in the uncrossed pathways (Taylor and Guillery, 1995). In short, competition between the two eyes for synaptic space is necessary for the normal formation of the visual pathways and this operates at both cortical and subcortical levels. Although the animal results are complex and extrapolation to humans is made difficult by both empirical and technical considerations, the anatomical and physiological changes found in nonhuman species as a result of enucleation or deprivation and changes in binocular competition suggest the possibility of psychophysical correlates in humans. On one hand, the deficits in motion processing in one-eyed observers that were outlined earlier may be accounted for by an interruption in binocularity during a postnatal sensitive period. In order to establish symmetrical motion processing, normal levels of binocularity, in particular binocular competition, may be required during development of its neural substrates. Other cases of binocular interruption or an imbalance in binocular competition in early visual development show asymmetrical motion processing. For example, OKN is asymmetrical in children and adults with strabismus with an onset before two years of age (e.g. Atkinson and Braddick, 1981; Reed et al., 1991; Steeves et al., 1999). In the case of unilateral enucleation, it appears that removing an eye at an early age has lead to an imbalance in (or rather, a complete absence of) the normal binocular competitive interactions that are necessary for the establishment of symmetrical motion perception. On the other hand, the enhancement in spatial vision that is seen in one-eyed observers can also be explained by the anatomical consequences of changes in binocular competition. Factors other than recruitment could also play a role in enhancing spatial processing including an absence of binocular interactions and monocular practice. Absence of inhibitory binocular interactions Neurons in primary and secondary cortical visual areas are binocular and exhibit an intracortical system of inhibitory interactions. Nicholas et al. (1996) found that
15 Vision with one eye 523 the peak contrast sensitivity at 4 c/deg of early-enucleated subjects was greater than the binocular performance of controls by a factor greater than 2, the theoretical limit attainable if all the cortical cells were driven by the remaining eye (Campbell and Green, 1965). The authors proposed that the enucleated subjects performance may be enhanced by the removal of the inhibitory binocular interactions, which are known to underlie the tuning to retinal disparity (Poggio et al., 1998) and binocular rivalry (Fox, 1991; Mueller, 1990). A reduction in horizontal connections for binocular vision in V1 has been observed in naturally strabismic monkeys (Tychsen et al., 2004). The removal, by enucleation, of this intracortical inhibitory system may make individual neurons more sensitive to contrast. In addition, it is also likely that the performance of normally binocular subjects under monocular viewing conditions may be adversely affected by the binocular rivalry that is produced by an eye patch, which is commonly used for such tests. Although this view contradicts Levelt s (1965a, 1965b) proposition that a contourless stimulus cannot suppress a patterned one and should remain suppressed indefinitely, it has received ample support from a number of studies (see González et al., 2007; Howard, 2002, for reviews). The rivalry from the occluded eye does not exhibit the classic temporal and spatial features of rivalry from a textured field, and although binocularly normal observers exhibit very few changes in a variety of visual functions after a month of monocular patching (Dengis et al., 1992), they report frequent and annoying blackouts during occlusion which is very likely a consequence of the rivalry from the patched eye. Even when visual disturbances are either not perceived or reported by observers, the type of monocular occlusion used has distinctive effects on binocular performance, including acuity (Horowitz, 1949) and contrast sensitivity and acuity (Wildsoet et al., 1998). Binocular acuity and contrast sensitivity deteriorate as a function of interocular illuminance differences, and increasing the density of the filter in front of one eye eventually leads to binocular inhibition (i.e. binocular viewing becomes worse than monocular viewing). That the perceived brightness of a target can be lower when one eye is covered by a neutral density filter than when the filtered eye is closed, is a phenomenon called Fechner s paradox (Fechner, 1860/1966). The absence of inhibitory binocular interactions in the enucleated group may explain in part their resistance to fading (González et al., 2007) and their superior performance in other contrast-defined tasks, but does not rule out the effects of plasticity through recruitment and imbalanced binocular competition. In the discrimination of radial frequency patterns, Steeves et al. (2004) found that despite the superiority of dichoptic over patched viewing for the controls, performance was still not equivalent to that with binocular viewing or to that of the enucleated observers. This finding may reflect the consequences of recruitment and plasticity after enucleation that has been observed anatomically in humans by Horton and Hocking (1998a). Plasticity after enucleation is not limited to an immature visual system, however. Klaeger-Manzanell et al. (1994) reported a two-step recovery of visual function in
16 524 J. K. E. Steeves et al. an amblyopic adult whose acuity in the amblyopic eye improved after vision loss in the fixing eye. After remaining stable for 13 months, there was a further increase in acuity after the formerly better eye was enucleated. Improvements in visual function in the amblyopic eye after a reduction of visual function in the non-amblyopic eye have been documented in adults by others (Hamid et al., 1991; Romero-Apis et al., 1982) and are consistent with data from animals (Harwerth et al., 1986; Horton and Hocking, 1998b; Prusky et al., 2006; Smith, 1982). Finally, the superior monocular sensitivity for spatial vision of one-eyed observers could also be predicted by a model involving simple cortical pooling and a winnertake-all-rule as proposed by McKee et al. (2003). This model can explain some of the enucleated data showing better spatial sensitivity but fails to predict the diminished acuity of non-amblyopic and amblyopic strabismic observers using their preferred eye. This suggests that complete deafferentation with enucleation has a very distinct operation on the visual system when compared to non-deafferenting forms of monocular deprivation. Monocular practice Even though monocular practice may be an important component of the superior visual spatial performance of enucleated observers, González et al. (1998) found that age at enucleation rather than years since enucleation is a better predictor of visual performance. When comparing two early (under 2 years of age) and one late (in adulthood) enucleated patient with two normal controls viewing monocularly, the early enucleated observers had better improvement in performance with practice for recognition of motion-defined letters. In fact, the learning rate of the early enucleated observers was higher than that of the controls and the late enucleated subject. Similarly, other studies have also seen more plasticity in early compared to late enucleated observers (Nicholas et al., 1996). The data described here highlight the problems in studying the effects of binocular deprivation and the difficulties in selecting appropriate control groups for one-eyed observers. On one hand, while strabismic amblyopia, anisometropia, and unilateral cataracts all involve monocular deprivation, they also involve abnormal binocular interactions which result in inferior performance in many visual functions. On the other hand, in binocularly normal observers, patching or closing one eye does not produce monocular vision but rather a condition of weak binocular rivalry which, in addition to probability and neural summation explains the superiority of their binocular over monocular, that is, patched-viewing (Howard, 2002). In the study of visual plasticity and visual development, one-eyed observers provide us with a useful model to study the roles of binocularity in visual processing. Further, this monocular model leads to very different effects compared to other models of monocular deprivation. Complete monocular deafferentation, particularly when it occurs early in life, results in a divergence of two visual subsystems. The evidence suggests that spatial vision and motion systems are distinct visual subsystems that are differentially affected by the loss of an eye early in life. While
17 Vision with one eye 525 spatial vision appears somewhat enhanced following the loss of one eye, motion processing and oculomotor systems are poorer. Motion processing and oculomotor function must require balanced binocular input to establish these systems. Finally, despite the dissociation that is seen in these visual subsystems, one-eyed individuals maintain perfectly normal lives and are not limited by their lack of binocularity. Acknowledgements We are grateful to Linda Lillakas for her comments and editorial assistance. Support for this review comes from the Natural Sciences and Engineering Research Council, The Sir Jules Thorn Charitable Trust, The Krembil Family Foundation, Atkinson Faculty and the Vision Sciences Research Program at the Toronto Western Hospital. REFERENCES Atkinson, J. and Braddick, O. (1981). Development of optokinetic nystagmus in infants: an indicator of cortical binocularity? in: Eye Movements: Cognition and Visual Perception, Fischer, D. F., Monty, R. A. and Senders, J. W. (Eds), pp Lawrence Erlbaum, Hillsdale, NJ, USA. Barbeito, R. (1983). Sighting from the cyclopean eye: the cyclops effect in preschool children, Perception and Psychophysics 33, Bavelier, D., Brozinsky, C., Tomann, A., Mitchell, T., Neville, H. and Liu, G. (2001). Impact of early deafness and early exposure to sign language on the cerebral organization for motion processing, J. Neurosci. 15, Bavelier, D., Dye, M. W. and Hauser, P. C. (2006). Do deaf individuals see better? Trends Cognit. Sci. 10, Bowns, L., Kirshner, E. L. and Steinbach, M. J. (1994). Shear sensitivity in normal and monocularly enucleated adults, Vision Research 34, Campbell, F. W. and Green, D. G. (1965). Optical and retinal factors affecting visual resolution, J. Physiol. 181, Daw, N. W. (1995). Visual Development. Plenum, New York, USA. Day, S. (1995). Vision development in the monocular individual: implications for the mechanisms of normal binocular vision development and the treatment of infantile esotropia, Trans. Amer. Ophthalmol. Soc. XCVII, Dengis, C. A., Steinbach, M. J. and Kraft, S. P. (1992). Monocular occlusion for one month: lack of effect on a variety of visual functions in normal adults, Invest. Ophthalmol. Vis. Sci. Supp. 33, Dengis, C. A., Steinbach, M. J., Goltz, H. C. and Stager, C. (1993). Visual alignment from the midline: a declining developmental trend in normal, strabismic and monocularly enucleated children, J. Pediat. Ophthalmol. Strabismus 30, Dengis, C. A., Steinbach, M. J., Ono, H., Gunther, L. N. and Postiglione, S. (1995). Eye hand coordination tasks in normals, strabismics and enucleates, Invest. Ophthalmol. Vis. Sci. Supp. 36, S645. Dengis, C. A., Steinbach, M. J., Ono, H., Gunther, L. N., Fanfarillo, R., Steeves, J. K. E. and Postiglione, S. (1996). Learning to look with one eye: the use of head turn by normals and strabismics, Vision Research 36, Dengis, C. A., Steinbach, M. J., Ono, H. and Gunther, L. (1997). Learning to wink voluntarily and to master monocular tasks: a comparison of normal vs strabismic children, Binocular Vision 12,
18 526 J. K. E. Steeves et al. Dengis, C. A., Simpson, T., Steinbach, M. J. and Ono, H. (1998). The cyclops effect in adults: sighting without visual feedback, Vision Research 38, Fechner, G. (1966). Elements of Psychophysics. (Translated by Adler H. E.). Holt, New York, USA. Rinehart, Winston. (Original work published 1860.) Fox, R. (1991). Binocular rivalry, in: Vision and Visual Dysfunction, Vol. IX: Binocular Vision, Regan, D. (Ed.), pp CRC Press, Boca Raton, Florida, USA. Fox, K. and Wong, R. O. (2005). A comparison of experience-dependent plasticity in the visual and somatosensory systems, Neuron 48, Freeman, R. D. and Bradley, A. (1980). Monocularly deprived humans: nondeprived eye has supernormal vernier acuity, J. Neurophys. 43, Giaschi, D. and Regan, D. (1997). Development of motion-defined figure ground segregation in preschool and older children, using a letter-identification task, Optom. Vis. Sci. 74, Gilbert, C. D. and Wiesel, T. N. (1992). Receptive field dynamics in adult primary visual cortex, Nature 356, Goltz, H. C., Steinbach, M. J. and Gallie, B. L. (1997). Head turn in 1-eyed and normally sighted individuals during monocular viewing, Arch. Ophthalmol. 115, González, E. G., Steinbach, M. J., Ono, H. and Wolf, M. (1989). Depth perception in humans enucleated at an early age, Clin. Vis. Sci. 4, González, E. G., Steinbach, M. J., Ono, H. and Rush-Smith, N. (1992). Vernier acuity in monocular and binocular children, Clin. Vis. Sci. 7, González, E. G., Steeves, J. K. E. and Steinbach, M. J. (1998). Perceptual learning for motiondefined letters in unilaterally enucleated observers and monocularly viewing normal controls, Invest. Ophthalmol. Vis. Sci. Supp. 39, S400. González, E. G., Steinbach, M. J., Ono, H. and Gallie, B. L. (1999). Localization of facial landmarks in binocular and monocular children, Binocular Vision 14, González, E. G., Steeves, J. K. E., Kraft, S. P., Gallie, B. L. and Steinbach, M. J. (2002). Foveal and eccentric acuity in one-eyed observers, Behav. Brain Res. 128, González, E. G., Weinstock, M. and Steinbach, M. J. (2007). Peripheral fading with monocular and binocular viewing, Vision Research 47, Guillery, R. W. (1989). Competition in the development of the visual pathways, in: The Making of the Nervous System, Parnavelas, J. G., Stern, C. D. and Stirling, R. V. (Eds), pp Oxford University Press, Oxford, UK. Hamid, L. M., Glaser, J. S. and Schatz, N. J. (1991). Improvement of vision in the amblyopic eye following visual loss in the contralateral normal eye: a report of three cases, Binoc. Vis. 6, Harwerth, R. S., Smith, E. L., Duncan, G. C., Crawford, M. L. J. and von Noorden, G. K. (1986). Effects of enucleation of the fixing eye in strabismic amblyopic monkeys, Invest. Ophthalmol. Vis. Sci. 27, Helveston, E. M., Pinchoff, B., Ellis, F. D. and Miller, K. (1985). Unilateral esotropia after enucleation in infancy, Amer. J. Ophthalmol. 100, Hering, E. (1942). Spatial Sense and Movements of the Eye, p. 38. (Translated by Radde, C. A.) American Academy of Optometry, Baltimore, USA. (Original work published 1879.) Horowitz, M. W. (1949). An analysis of the superiority of binocular over monocular visual acuity, J. Exper. Psychol. 39, Horton, J. C. and Hocking, D. R. (1998a). Effect of early monocular enucleation upon ocular dominance columns and cytochrome oxidase activity in monkey and human visual cortex, Vis. Neurosci. 15, Horton, J. C. and Hocking, D. R. (1998b). Monocular core zones and binocular border strips in primate striate cortex revealed by the contrasting effects of enucleation, eyelid suture, and retinal laser lesions on cytochrome oxidase activity, J. Neurosci. 18, Howard, I. P. (2002). Seeing in Depth, Vol. 1. Porteus, Thornhill, Ontario, Canada. Howard, I. P. and Rogers, B. J. (1995). Binocular Vision and Stereopsis. Oxford, New York, USA.
19 Vision with one eye 527 Hoyle, F. (1957). The Black Cloud. Penguin, London, UK. Hubel, D. H. and Wiesel, T. N. (1962). Receptive fields binocular interaction and functional architecture in the cat s visual cortex, J. Physiol. 160, Hubel, D. H., Wiesel, T. N. and LeVay, S. (1977). Plasticity of ocular dominance columns in monkey striate cortex, Philosoph. Trans. Roy. Soc. London B: Biol. Sci. 278, Johnson, C. A., Post, R. B., Chalupa, L. M. and Lee, T. J. (1982). Monocular deprivation in humans: a study of identical twins, Invest. Ophthalmol. Vis. Sci. 23, Kahn, D. M. and Krubitzer, L. (2002). Massive cross-modal cortical plasticity and the emergence of a new cortical area in developmentally blind mammals, Proc. Nat. Acad. Sci. USA 99, Karmarkar, U. R. and Dan, Y. (2006). Experience-dependent plasticity in adult visual cortex, Neuron 52, Kiorpes, L. and Movshon, S. P. (2003). Neural limitations on visual development in primates, in: The Visual Neurosciences, Chalupa, L. M. and Werner, J. S. (Eds), pp MIT Press, Cambridge, MA, USA. Klaeger-Manzanell, C., Hoyt, C. S. and Good, W. V. (1994). Two-step recovery of vision in the amblyopic eye after visual loss and enucleation of the fixing eye, Brit. J. Ophthalmol. 78, Krahe, T. E., Medina, A. E., de Bittencourt-Navarrete, R. E., Colello, R. J. and Ramoa, A. S. (2005). Protein synthesis-independent plasticity mediates rapid and precise recovery of deprived eye responses, Neuron 48, Kratz, K. E. and Spear, P. D. (1976). Effects of visual deprivation and alterations in binocular competition on responses of striate cortex neurons in the cat, J. Comp. Neurol. 170, Kujala, T., Alho, K. and Naatanen, R. (2000). Cross-modal reorganization of human cortical functions, Trends Neurosci. 23, Lee, S. H. and Blake, R. (2002). V1 activity is reduced during binocular rivalry, J. Vision 2, Levelt, W. J. M. (1965a). Binocular brightness averaging and contour information, Brit. J. Psychol. 56, Levelt, W. J. M. (1965b). On Binocular Rivalry. Institute for Perception, Soesterberg, The Netherlands. Lewis, T. and Maurer, D. (1992). The development of the temporal and nasal visual fields during infancy, Vision Research 32, Mansouri, B. and Hess, R. F. (2006). The global processing deficit in amblopia involves noise segregation, Vision Research 46, Marotta, J. J., Perrot, T. S., Nicolle, D. and Goodale, M. A. (1995). The development of adaptive head movements following enucleation, Eye 9, McKee, S. P. and Westheimer, G. (1978). Improvement in vernier acuity with practice, Perception and Psychophysics 24, McKee, S. P., Levi, D. M. and Movshon, J. A. (2003). The pattern of visual deficits in amblyopia, J. Vision 3, Moidell, B., Steinbach, M. J. and Ono, H. (1988). Egocenter location in children enucleated at an early age, Invest. Ophthalmol. Vis. Sci. 29, Mueller, T. J. (1990). A physiological model of binocular rivalry, Vis. Neurosci. 4, Naegle, J. R. and Held, R. (1982). The postnatal development of monocular optokinetic nystagmus in infants, Vision Research 22, Nicholas, J., Heywood, C. A. and Cowey, A. (1996). Contrast sensitivity in one-eyed subjects, Vision Research 26, Norcia, A. M., Garcia, H., Humphry, R., Holmes, A., Hamer, R. D. and Orel-Bixler, D. (1991). Anomalous motion VEPs in infants and in infantile esotropia, Invest. Ophthalmol. Vis. Sci. 32, Ono, H. and Mapp, A. P. (1995). A restatement and modification of Wells Hering s laws of visual direction, Perception 24,
20 528 J. K. E. Steeves et al. Ono, H., Mapp, A. P. and Howard, I. P. (2002). The cyclopean eye in vision: the new and old data continue to hit you right between the eyes, Vision Research 42, Erratum in: Vision Research 42, 2331 (2002). Poggio, G. F., Fahle, M. and Edelman, S. (1992). Fast perceptual learning in visual hyperacuity, Science 256, Poggio, G. F., Gonzalez, F. and Krause, F. (1998). Stereoscopic mechanisms in monkey visual cortex: binocular correlation and disparity selectivity, J. Neurosci. 8, Previc, F. H. (1990). Functional specialization in the lower and upper visual fields in humans: its ecological origins and neurophysiological implications, Behav. Brain Sci. 13, Prusky, G. T., Alam, N. M. and Douglas, R. M. (2006). Enhancement of vision by moncular deprivation in adult mice, J. Neurosci. 26, Rakic, P. (1981). Development of visual centers in the primate brain depends on binocular competition before birth, Science 214, Reed, M. J., Steinbach, M. J., Anstis, S. M., Gallie, B. L., Smith, D. R. and Kraft, S. P. (1991). The development of optokinetic nystagmus in strabismic and monocularly enucleated subjects, Behav. Brain Res. 46, Reed, M. J., Steeves, J. K. E., Kraft, S. P., Gallie, B. L. and Steinbach, M. J. (1996). Contrast letter thresholds in the non-affected eye of strabismic and unilateral eye enucleated children, Vision Research 36, Reed, M. J., Steeves, J. K. E. and Steinbach, M. J. (1997). A comparison of contrast letter thresholds in unilateral eye enucleated subjects and binocular and monocular control subjects, Vision Research 37, Regan, D. (1988). Low-contrast visual acuity test for pediatric use, Can. J. Ophthalmol. 23, 224. Regan, D. and Hong, X. H. (1990). Visual acuity for optotypes made visible by relative motion, Optic. Vis. Sci. 67, Regan, D. and Hong, X. H. (1994). Recognition and detection of texture-defined letters, Vision Research 34, Romero-Apis, D., Rabayán-Mena, J. I., Fonte-Vázquez, A., Gutiérrez-Pérez, D., Martínez- Oropeza, S. and Murillo-Murillo, L. (1982). Pérdida del ojo fijador en adulto con ambliopia estrábica, An. Soc. Mex. de Oft. 56, Schmid, L. M., Rosa, M. G. P. and Calford, M. B. (1995). Retinal detachment induces massive immediate reorganization in visual cortex, Neuroreport 6, Schwartz, T. L., Linberg, J. V., Tillman, W. and Odom, J. V. (1987). Monocular depth and vernier acuities: a comparison of binocular and uniocular subjects, Invest. Ophthalmol. Vis. Sci. Supp. 28, 304. Simpson, W. A. (1993). Optic flow and depth perception, Spatial Vision 7, Skrandies, W. (1987). The upper and lower visual field of man: electrophysiological and functional differences, in: Progress in Sensory Physiology, Ottoson, D. (Ed.), pp Springer, Berlin, Germany. Smith, D. C. (1982). Functional restoration of vision in the cat after long-term monocular deprivation, Science 213, Steeves, J. K. E., Reed, M. J., Steinbach, M. J. and Kraft, S. P. (1999). Monocular horizontal optokinetic nystagmus in observers with early- and late-onset strabismus, Behav. Brain Res. 103, Steeves, J. K. E., Gray, R., Steinbach, M. J. and Regan, D. (2000). Accuracy of estimating time to collision using only monocular information in unilaterally enucleated observers and monocularly visewing normal controls, Vision Research 40, Steeves, J. K. E., González, E. G., Gallie, B. L. and Steinbach, M. J. (2002). Early unilateral enucleation disrupts motion processing, Vision Research 42, Steeves, J. K. E., Wilkinson, F., González, E. G., Wilson, H. R. and Steinbach, M. J. (2004). Global shape discrimination at reduced contrast in enucleated observers, Vision Research 44,
21 Vision with one eye 529 Steinbach, M. J., Howard, I. P. and Ono, H. (1985). Monocular asymmetries in vision: we don t see eye-to-eye, Can. J. Psychol. 39, Syken, J., GrandPre, T., Kanold, P. O. and Shatz, C. J. (2006). PirB restricts ocular-dominance plasticity in the visual cortex, Science 313, Taylor, J. S. and Guillery, R. W. (1995). Effect of a very early monocular enucleation upon the development of the uncrossed retinofugal pathway in ferrets, J. Comp. Neurol. 357, Tychsen, L. (1993). Motion sensitivity and the origins of infantile strabismus, in: Early Visual Development: Normal and Abnormal, Simons, K. (Ed.), pp Oxford University Press, New York, USA. Tychsen, L., Wong, A. M. and Burkhalter, A. (2004). Paucity of horizontal connections for binocular vision in V1 of naturally-strabismic macaques: cytochrome-oxidase compartment specificity, J. Comp. Neurol. 474, Wildsoet, C., Wood, J., Maag, H. and Sabdia, S. (1998). The effect of different forms of monocular occlusion on measures of central visual function, Ophthal. Physiol. Opt. 18, Wilkinson, F., Wilson, H. R. and Habak, C. (1998). Detection and recognition of radial frequency patterns, Vision Research 38, Zanker, J., Mohne, G., Weber, U., Zeitler-Driess, K. and Fahle, M. (1992). The development of vernier acuity in human infants, Vision Research 32,
The Visual Cortex 0 http://www.tutis.ca/neuromd/index.htm 20 February 2013
 T he Visual Cortex 0 Chapter contents Contents Chapter 2... 0 T he Visual Cortex... 0 Chapter Contents... 1 Introduction... 2 Optic Chiasm... 2 Where do the eye's ganglion cells project to?... 3 To where
T he Visual Cortex 0 Chapter contents Contents Chapter 2... 0 T he Visual Cortex... 0 Chapter Contents... 1 Introduction... 2 Optic Chiasm... 2 Where do the eye's ganglion cells project to?... 3 To where
Esotropia (Crossed Eye(s))
 Esotropia (Crossed Eye(s)) Esotropia is a type of strabismus or eye misalignment in which the eyes are "crossed," that is, while one eye looks straight ahead, the other eye is turned in toward the nose.
Esotropia (Crossed Eye(s)) Esotropia is a type of strabismus or eye misalignment in which the eyes are "crossed," that is, while one eye looks straight ahead, the other eye is turned in toward the nose.
CHAPTER 6 PRINCIPLES OF NEURAL CIRCUITS.
 CHAPTER 6 PRINCIPLES OF NEURAL CIRCUITS. 6.1. CONNECTIONS AMONG NEURONS Neurons are interconnected with one another to form circuits, much as electronic components are wired together to form a functional
CHAPTER 6 PRINCIPLES OF NEURAL CIRCUITS. 6.1. CONNECTIONS AMONG NEURONS Neurons are interconnected with one another to form circuits, much as electronic components are wired together to form a functional
Chapter 8: Perceiving Depth and Size
 Chapter 8: Perceiving Depth and Size Cues to Depth Perception Oculomotor - cues based on sensing the position of the eyes and muscle tension 1. Convergence knowing the inward movement of the eyes when
Chapter 8: Perceiving Depth and Size Cues to Depth Perception Oculomotor - cues based on sensing the position of the eyes and muscle tension 1. Convergence knowing the inward movement of the eyes when
Visual development in babies and infants
 Visual development in babies and infants Marko Nardini UCL Institute of Ophthalmology Vision a major function of the primate brain vision develops rapidly in early life and serves as a base for development
Visual development in babies and infants Marko Nardini UCL Institute of Ophthalmology Vision a major function of the primate brain vision develops rapidly in early life and serves as a base for development
Visual area MT responds to local motion. Visual area MST responds to optic flow. Visual area STS responds to biological motion. Macaque visual areas
 Visual area responds to local motion MST a Visual area MST responds to optic flow MST a Visual area STS responds to biological motion STS Macaque visual areas Flattening the brain What is a visual area?
Visual area responds to local motion MST a Visual area MST responds to optic flow MST a Visual area STS responds to biological motion STS Macaque visual areas Flattening the brain What is a visual area?
Fourth Nerve Palsy (a.k.a. Superior Oblique Palsy)
 Hypertropia Hypertropia is a type of strabismus characterized by vertical misalignment of the eyes. Among the many causes of vertical strabismus, one of the most common is a fourth nerve palsy (also known
Hypertropia Hypertropia is a type of strabismus characterized by vertical misalignment of the eyes. Among the many causes of vertical strabismus, one of the most common is a fourth nerve palsy (also known
Clinical Professor of Ophthalmology, John A. Moran Eye Center, University of Utah
 1 NeuroAdaptation Robert M. Kershner, M.D., M.S., F.A.C.S. Clinical Professor of Ophthalmology, John A. Moran Eye Center, University of Utah School of Medicine, Salt Lake City, Utah IK HO Visiting Professor
1 NeuroAdaptation Robert M. Kershner, M.D., M.S., F.A.C.S. Clinical Professor of Ophthalmology, John A. Moran Eye Center, University of Utah School of Medicine, Salt Lake City, Utah IK HO Visiting Professor
6 Space Perception and Binocular Vision
 Space Perception and Binocular Vision Space Perception and Binocular Vision space perception monocular cues to 3D space binocular vision and stereopsis combining depth cues monocular/pictorial cues cues
Space Perception and Binocular Vision Space Perception and Binocular Vision space perception monocular cues to 3D space binocular vision and stereopsis combining depth cues monocular/pictorial cues cues
GAZE STABILIZATION SYSTEMS Vestibular Ocular Reflex (VOR) Purpose of VOR Chief function is to stabilize gaze during locomotion. Acuity declines if
 GAZE STABILIZATION SYSTEMS Vestibular Ocular Reflex (VOR) Purpose of VOR Chief function is to stabilize gaze during locomotion. Acuity declines if slip exceeds 3-5 deg/sec. Ex: Head bobbing and heel strike
GAZE STABILIZATION SYSTEMS Vestibular Ocular Reflex (VOR) Purpose of VOR Chief function is to stabilize gaze during locomotion. Acuity declines if slip exceeds 3-5 deg/sec. Ex: Head bobbing and heel strike
Space Perception and Binocular Vision
 Space Perception and Binocular Vision Space Perception Monocular Cues to Three-Dimensional Space Binocular Vision and Stereopsis Combining Depth Cues 9/30/2008 1 Introduction to Space Perception Realism:
Space Perception and Binocular Vision Space Perception Monocular Cues to Three-Dimensional Space Binocular Vision and Stereopsis Combining Depth Cues 9/30/2008 1 Introduction to Space Perception Realism:
Understanding astigmatism Spring 2003
 MAS450/854 Understanding astigmatism Spring 2003 March 9th 2003 Introduction Spherical lens with no astigmatism Crossed cylindrical lenses with astigmatism Horizontal focus Vertical focus Plane of sharpest
MAS450/854 Understanding astigmatism Spring 2003 March 9th 2003 Introduction Spherical lens with no astigmatism Crossed cylindrical lenses with astigmatism Horizontal focus Vertical focus Plane of sharpest
Chapter 14: The Cutaneous Senses
 Chapter 14: The Cutaneous Senses Skin - heaviest organ in the body Cutaneous System Epidermis is the outer layer of the skin, which is made up of dead skin cells Dermis is below the epidermis and contains
Chapter 14: The Cutaneous Senses Skin - heaviest organ in the body Cutaneous System Epidermis is the outer layer of the skin, which is made up of dead skin cells Dermis is below the epidermis and contains
Binocular Vision and The Perception of Depth
 Binocular Vision and The Perception of Depth Visual Perception How one visually interprets a scene 4 forms of perception to be studied: Depth Color Temporal Motion Depth Perception How does one determine
Binocular Vision and The Perception of Depth Visual Perception How one visually interprets a scene 4 forms of perception to be studied: Depth Color Temporal Motion Depth Perception How does one determine
Research. Investigation of Optical Illusions on the Aspects of Gender and Age. Dr. Ivo Dinov Department of Statistics/ Neuroscience
 RESEARCH Research Ka Chai Lo Dr. Ivo Dinov Department of Statistics/ Neuroscience Investigation of Optical Illusions on the Aspects of Gender and Age Optical illusions can reveal the remarkable vulnerabilities
RESEARCH Research Ka Chai Lo Dr. Ivo Dinov Department of Statistics/ Neuroscience Investigation of Optical Illusions on the Aspects of Gender and Age Optical illusions can reveal the remarkable vulnerabilities
Education and the Brain: A Bridge Too Far John T. Bruer. Key Concept: the Human Brain and Learning
 Education and the Brain: A Bridge Too Far John T. Bruer Key Concept: the Human Brain and Learning John T. Bruer Scholar in cognitivist approaches to human learning and instruction. His argument refers
Education and the Brain: A Bridge Too Far John T. Bruer Key Concept: the Human Brain and Learning John T. Bruer Scholar in cognitivist approaches to human learning and instruction. His argument refers
Definitions, Identification, and Supportive Professionals
 Definitions, Identification, and Supportive Professionals Dolly Bhargava, M. Spec. Ed. with Supportive Information from Elmwood Visual Resource Centre, Christchurch, New Zealand Vision plays an important
Definitions, Identification, and Supportive Professionals Dolly Bhargava, M. Spec. Ed. with Supportive Information from Elmwood Visual Resource Centre, Christchurch, New Zealand Vision plays an important
Exotropias: A Brief Review. Leila M. Khazaeni, MD November 2, 2008
 Exotropias: A Brief Review Leila M. Khazaeni, MD November 2, 2008 Exotropia Myths Myth #1 He/she will grow out of it FALSE 75% of XTs show progression over a 3 year period Myth #2 The only treatment choice
Exotropias: A Brief Review Leila M. Khazaeni, MD November 2, 2008 Exotropia Myths Myth #1 He/she will grow out of it FALSE 75% of XTs show progression over a 3 year period Myth #2 The only treatment choice
Motion processing: the most sensitive detectors differ in temporally localized and extended noise
 ORIGINAL RESEARCH ARTICLE published: 15 May 2014 doi: 10.3389/fpsyg.2014.00426 Motion processing: the most sensitive detectors differ in temporally localized and extended noise Rémy Allard 1,2,3 * and
ORIGINAL RESEARCH ARTICLE published: 15 May 2014 doi: 10.3389/fpsyg.2014.00426 Motion processing: the most sensitive detectors differ in temporally localized and extended noise Rémy Allard 1,2,3 * and
CONGENITAL NYSTAGMUS WHEN TO RECORD HOW TO TREAT 2009
 CONGENITAL NYSTAGMUS WHEN TO RECORD HOW TO TREAT 2009 LIONEL KOWAL Royal Victorian Eye and Ear Hospital Center for Eye Research Australia Melbourne, Australia TYPES OF CONGENITAL NYSTAGMUS cn cn: any type
CONGENITAL NYSTAGMUS WHEN TO RECORD HOW TO TREAT 2009 LIONEL KOWAL Royal Victorian Eye and Ear Hospital Center for Eye Research Australia Melbourne, Australia TYPES OF CONGENITAL NYSTAGMUS cn cn: any type
THE EYES IN MARFAN SYNDROME
 THE EYES IN MARFAN SYNDROME Marfan syndrome and some related disorders can affect the eyes in many ways, causing dislocated lenses and other eye problems that can affect your sight. Except for dislocated
THE EYES IN MARFAN SYNDROME Marfan syndrome and some related disorders can affect the eyes in many ways, causing dislocated lenses and other eye problems that can affect your sight. Except for dislocated
Programs for diagnosis and therapy of visual field deficits in vision rehabilitation
 Spatial Vision, vol. 10, No.4, pp. 499-503 (1997) Programs for diagnosis and therapy of visual field deficits in vision rehabilitation Erich Kasten*, Hans Strasburger & Bernhard A. Sabel Institut für Medizinische
Spatial Vision, vol. 10, No.4, pp. 499-503 (1997) Programs for diagnosis and therapy of visual field deficits in vision rehabilitation Erich Kasten*, Hans Strasburger & Bernhard A. Sabel Institut für Medizinische
Masters research projects. 1. Adapting Granger causality for use on EEG data.
 Masters research projects 1. Adapting Granger causality for use on EEG data. Background. Granger causality is a concept introduced in the field of economy to determine which variables influence, or cause,
Masters research projects 1. Adapting Granger causality for use on EEG data. Background. Granger causality is a concept introduced in the field of economy to determine which variables influence, or cause,
The Binding Problem Solutions to the spatial binding problem
 The Binding Problem Objects have different features such as color, shape, sound, and smell. Some, such as color and sound, are represented separately from the instant they hit our sensory receptors. Other
The Binding Problem Objects have different features such as color, shape, sound, and smell. Some, such as color and sound, are represented separately from the instant they hit our sensory receptors. Other
Processing the Image or Can you Believe what you see? Light and Color for Nonscientists PHYS 1230
 Processing the Image or Can you Believe what you see? Light and Color for Nonscientists PHYS 1230 Optical Illusions http://www.michaelbach.de/ot/mot_mib/index.html Vision We construct images unconsciously
Processing the Image or Can you Believe what you see? Light and Color for Nonscientists PHYS 1230 Optical Illusions http://www.michaelbach.de/ot/mot_mib/index.html Vision We construct images unconsciously
Auditory neuroanatomy: the Spanish heritage. Santiago Ramón y Cajal, 1852 1934
 Auditory neuroanatomy: the Spanish heritage Santiago Ramón y Cajal, 1852 1934 Rafael Lorente de Nó, 1902 1990 3 The nervous system is made up of cells. Estimates of the number of cells vary from
Auditory neuroanatomy: the Spanish heritage Santiago Ramón y Cajal, 1852 1934 Rafael Lorente de Nó, 1902 1990 3 The nervous system is made up of cells. Estimates of the number of cells vary from
Curriculum Vitae. Adriane E. Seiffert. January 5, 2010
 Curriculum Vitae Adriane E. Seiffert January 5, 2010 Degrees Earned Bachelors of Science, Honors, Co-op in Psychology earned in 1994 University of Waterloo, Waterloo, Ontario, Canada MA in Psychology,
Curriculum Vitae Adriane E. Seiffert January 5, 2010 Degrees Earned Bachelors of Science, Honors, Co-op in Psychology earned in 1994 University of Waterloo, Waterloo, Ontario, Canada MA in Psychology,
Guidelines for the Management of Amblyopia
 Guidelines for the Management of Amblyopia 1. Introduction a. Background Guidelines for the management of strabismus and amblyopia were published by the Royal College of Ophthalmologists in 2000. Since
Guidelines for the Management of Amblyopia 1. Introduction a. Background Guidelines for the management of strabismus and amblyopia were published by the Royal College of Ophthalmologists in 2000. Since
Insights into Visual Impairments
 Insights into Visual Impairments Definition of Visual Impairment Washington State WAC 392 172A 01035 Child with a disability or student eligible for special education. (n) Visual impairment including blindness
Insights into Visual Impairments Definition of Visual Impairment Washington State WAC 392 172A 01035 Child with a disability or student eligible for special education. (n) Visual impairment including blindness
Problem-Based Group Activities for a Sensation & Perception Course. David S. Kreiner. University of Central Missouri
 -Based Group Activities for a Course David S. Kreiner University of Central Missouri Author contact information: David Kreiner Professor of Psychology University of Central Missouri Lovinger 1111 Warrensburg
-Based Group Activities for a Course David S. Kreiner University of Central Missouri Author contact information: David Kreiner Professor of Psychology University of Central Missouri Lovinger 1111 Warrensburg
Hearing and Vision Program. Public Health Muskegon County
 Hearing and Vision Program Public Health Muskegon County H&V Screening: Two of the Required Public Health Services in Michigan The Hearing and Vision programs are required by the Michigan Public Health
Hearing and Vision Program Public Health Muskegon County H&V Screening: Two of the Required Public Health Services in Michigan The Hearing and Vision programs are required by the Michigan Public Health
Cerebral Palsy and Visual Impairment
 CP Factsheet Cerebral Palsy and Visual Impairment Although cerebral palsy mainly causes difficulty with movement, other conditions may also occur. This is because other parts of the brain may also be affected
CP Factsheet Cerebral Palsy and Visual Impairment Although cerebral palsy mainly causes difficulty with movement, other conditions may also occur. This is because other parts of the brain may also be affected
The Information Processing model
 The Information Processing model A model for understanding human cognition. 1 from: Wickens, Lee, Liu, & Becker (2004) An Introduction to Human Factors Engineering. p. 122 Assumptions in the IP model Each
The Information Processing model A model for understanding human cognition. 1 from: Wickens, Lee, Liu, & Becker (2004) An Introduction to Human Factors Engineering. p. 122 Assumptions in the IP model Each
Video-Based Eye Tracking
 Video-Based Eye Tracking Our Experience with Advanced Stimuli Design for Eye Tracking Software A. RUFA, a G.L. MARIOTTINI, b D. PRATTICHIZZO, b D. ALESSANDRINI, b A. VICINO, b AND A. FEDERICO a a Department
Video-Based Eye Tracking Our Experience with Advanced Stimuli Design for Eye Tracking Software A. RUFA, a G.L. MARIOTTINI, b D. PRATTICHIZZO, b D. ALESSANDRINI, b A. VICINO, b AND A. FEDERICO a a Department
Drawing in blind and visually impaired children*
 1 Terra Haptica is a journal published by Les Doigts Qui Rêvent (Dreaming Fingers) Here the front and back cover pages. Size A4. Drawing in blind and visually impaired children* Annie VINTER a, Viviane
1 Terra Haptica is a journal published by Les Doigts Qui Rêvent (Dreaming Fingers) Here the front and back cover pages. Size A4. Drawing in blind and visually impaired children* Annie VINTER a, Viviane
Next Generation Artificial Vision Systems
 Next Generation Artificial Vision Systems Reverse Engineering the Human Visual System Anil Bharath Maria Petrou Imperial College London ARTECH H O U S E BOSTON LONDON artechhouse.com Contents Preface xiii
Next Generation Artificial Vision Systems Reverse Engineering the Human Visual System Anil Bharath Maria Petrou Imperial College London ARTECH H O U S E BOSTON LONDON artechhouse.com Contents Preface xiii
Basic Cranial Nerve Examination
 Basic Cranial Nerve Examination WIPE Wash hands Introduce yourself Permission Position (Patient sitting facing you, maintain comparable eye level) Exposure (Face exposed only, i.e. remove hats etc) Identify
Basic Cranial Nerve Examination WIPE Wash hands Introduce yourself Permission Position (Patient sitting facing you, maintain comparable eye level) Exposure (Face exposed only, i.e. remove hats etc) Identify
Fundus Photograph Reading Center
 Modified 7-Standard Field Digital Color Fundus Photography (7M-D) 8010 Excelsior Drive, Suite 100, Madison WI 53717 Telephone: (608) 410-0560 Fax: (608) 410-0566 Table of Contents 1. 7M-D Overview... 2
Modified 7-Standard Field Digital Color Fundus Photography (7M-D) 8010 Excelsior Drive, Suite 100, Madison WI 53717 Telephone: (608) 410-0560 Fax: (608) 410-0566 Table of Contents 1. 7M-D Overview... 2
The Effect of Video Game Training on the Vision of Adults with Bilateral Deprivation Amblyopia
 Seeing and Perceiving (2012) DOI:10.1163/18784763-00002391 brill.nl/sp The Effect of Video Game Training on the Vision of Adults with Bilateral Deprivation Amblyopia Seong Taek Jeon 1,, Daphne Maurer 1,2
Seeing and Perceiving (2012) DOI:10.1163/18784763-00002391 brill.nl/sp The Effect of Video Game Training on the Vision of Adults with Bilateral Deprivation Amblyopia Seong Taek Jeon 1,, Daphne Maurer 1,2
PRIMING OF POP-OUT AND CONSCIOUS PERCEPTION
 PRIMING OF POP-OUT AND CONSCIOUS PERCEPTION Peremen Ziv and Lamy Dominique Department of Psychology, Tel-Aviv University [email protected] [email protected] Abstract Research has demonstrated
PRIMING OF POP-OUT AND CONSCIOUS PERCEPTION Peremen Ziv and Lamy Dominique Department of Psychology, Tel-Aviv University [email protected] [email protected] Abstract Research has demonstrated
Visual & Auditory Skills Lab
 Visual & Auditory Skills Lab Name: Score: Introduction This lab consists of a series of experiments that explore various perceptual, vision, and balance skills that help us understand how we perform motor
Visual & Auditory Skills Lab Name: Score: Introduction This lab consists of a series of experiments that explore various perceptual, vision, and balance skills that help us understand how we perform motor
Exp. 1 Pathways of sound conduction. Yu Yanqin, PhD Zhejiang University, School of Medicine
 Exp. 1 Pathways of sound conduction Yu Yanqin, PhD Zhejiang University, School of Medicine [Purpose] To learn how to use a tuning fork to generate sound; To understand the function of the auditory organ;
Exp. 1 Pathways of sound conduction Yu Yanqin, PhD Zhejiang University, School of Medicine [Purpose] To learn how to use a tuning fork to generate sound; To understand the function of the auditory organ;
In normal vision, both eyes are precisely aligned on an object
 3 Binocular Vision and Introduction to Strabismus Kenneth W. Wright In normal vision, both eyes are precisely aligned on an object of regard, so the images from that object fall on the fovea of each eye.
3 Binocular Vision and Introduction to Strabismus Kenneth W. Wright In normal vision, both eyes are precisely aligned on an object of regard, so the images from that object fall on the fovea of each eye.
THE HUMAN BRAIN. observations and foundations
 THE HUMAN BRAIN observations and foundations brains versus computers a typical brain contains something like 100 billion miniscule cells called neurons estimates go from about 50 billion to as many as
THE HUMAN BRAIN observations and foundations brains versus computers a typical brain contains something like 100 billion miniscule cells called neurons estimates go from about 50 billion to as many as
The Physiology of the Senses Lecture 11 - Eye Movements www.tutis.ca/senses/
 The Physiology of the Senses Lecture 11 - Eye Movements www.tutis.ca/senses/ Contents Objectives... 2 Introduction... 2 The 5 Types of Eye Movements... 2 The eyes are rotated by 6 extraocular muscles....
The Physiology of the Senses Lecture 11 - Eye Movements www.tutis.ca/senses/ Contents Objectives... 2 Introduction... 2 The 5 Types of Eye Movements... 2 The eyes are rotated by 6 extraocular muscles....
Amy Berryman, OTR MSHSA Karen Rasavage, OTR. Combined Sections Meeting Las Vegas, NV February 5, 2014
 Clinical Evaluation and Management of Visual and Perceptual Impairments following Brain Injury Amy Berryman, OTR MSHSA Karen Rasavage, OTR Combined Sections Meeting Las Vegas, NV February 5, 2014 Objectives
Clinical Evaluation and Management of Visual and Perceptual Impairments following Brain Injury Amy Berryman, OTR MSHSA Karen Rasavage, OTR Combined Sections Meeting Las Vegas, NV February 5, 2014 Objectives
Cortical Visual Impairment An introduction
 Cortical Visual Impairment An introduction This is a general introduction to Cortical Visual Impairment intended for families and support workers who may live with or support children and adults with learning
Cortical Visual Impairment An introduction This is a general introduction to Cortical Visual Impairment intended for families and support workers who may live with or support children and adults with learning
Physical and Mental Conditions Guidelines VISION CONDITIONS AND ACTIONS Page 5.4
 Physical and Mental Conditions Guidelines VISION CONDITIONS AND ACTIONS Page 5.4 AMBLYOPIA (Lazy Eye) A reduction in the acuteness of vision without apparent eye disease. This condition cannot be entirely
Physical and Mental Conditions Guidelines VISION CONDITIONS AND ACTIONS Page 5.4 AMBLYOPIA (Lazy Eye) A reduction in the acuteness of vision without apparent eye disease. This condition cannot be entirely
What constitutes good visualization research?
 Visualization Viewpoints Editor: TheresaMarie Rhyne Toward a Perceptual Theory of Flow Visualization Colin Ware University of New Hampshire What constitutes good visualization research? Are researchers
Visualization Viewpoints Editor: TheresaMarie Rhyne Toward a Perceptual Theory of Flow Visualization Colin Ware University of New Hampshire What constitutes good visualization research? Are researchers
2 Neurons. 4 The Brain: Cortex
 1 Neuroscience 2 Neurons output integration axon cell body, membrane potential Frontal planning control auditory episodes soma motor Temporal Parietal action language objects space vision Occipital inputs
1 Neuroscience 2 Neurons output integration axon cell body, membrane potential Frontal planning control auditory episodes soma motor Temporal Parietal action language objects space vision Occipital inputs
How Plastic Is Spatial Hearing?
 Auditory CNS Processing and Plasticity Audiol Neurootol 2001;6:182 186 How Plastic Is Spatial Hearing? Andrew J. King Oliver Kacelnik Thomas D. Mrsic-Flogel Jan W.H. Schnupp Carl H. Parsons David R. Moore
Auditory CNS Processing and Plasticity Audiol Neurootol 2001;6:182 186 How Plastic Is Spatial Hearing? Andrew J. King Oliver Kacelnik Thomas D. Mrsic-Flogel Jan W.H. Schnupp Carl H. Parsons David R. Moore
Subjects. Subjects were undergraduates at the University of California, Santa Barbara, with
 Category-specific visual attention 1 SI Appendix 1 Method Subjects. Subjects were undergraduates at the University of California, Santa Barbara, with normal or corrected-to-normal vision. Exp 1: n=30;
Category-specific visual attention 1 SI Appendix 1 Method Subjects. Subjects were undergraduates at the University of California, Santa Barbara, with normal or corrected-to-normal vision. Exp 1: n=30;
Primary Motor Pathway
 Understanding Eye Movements Abdullah Moh. El-Menaisy, MD, FRCS Chief, Neuro-ophthalmology ophthalmology & Investigation Units, Dhahran Eye Specialist Hospital, Dhahran, Saudi Arabia Primary Motor Pathway
Understanding Eye Movements Abdullah Moh. El-Menaisy, MD, FRCS Chief, Neuro-ophthalmology ophthalmology & Investigation Units, Dhahran Eye Specialist Hospital, Dhahran, Saudi Arabia Primary Motor Pathway
ADDENDUM to the Informed Consent for Cataract Surgery with Intraocular Lens Implant
 ADDENDUM to the Informed Consent for Cataract Surgery with Intraocular Lens Implant INTRODUCTION Except for unusual situations, a cataract operation is indicated only when you cannot function satisfactorily
ADDENDUM to the Informed Consent for Cataract Surgery with Intraocular Lens Implant INTRODUCTION Except for unusual situations, a cataract operation is indicated only when you cannot function satisfactorily
Detection Sensitivity and Response Bias
 Detection Sensitivity and Response Bias Lewis O. Harvey, Jr. Department of Psychology University of Colorado Boulder, Colorado The Brain (Observable) Stimulus System (Observable) Response System (Observable)
Detection Sensitivity and Response Bias Lewis O. Harvey, Jr. Department of Psychology University of Colorado Boulder, Colorado The Brain (Observable) Stimulus System (Observable) Response System (Observable)
Brain Maps The Sensory Homunculus
 Brain Maps The Sensory Homunculus Our brains are maps. This mapping results from the way connections in the brain are ordered and arranged. The ordering of neural pathways between different parts of the
Brain Maps The Sensory Homunculus Our brains are maps. This mapping results from the way connections in the brain are ordered and arranged. The ordering of neural pathways between different parts of the
STROKE CARE NOW NETWORK CONFERENCE MAY 22, 2014
 STROKE CARE NOW NETWORK CONFERENCE MAY 22, 2014 Rehabilitation Innovations in Post- Stroke Recovery Madhav Bhat, MD Fort Wayne Neurological Center DISCLOSURE Paid speaker for TEVA Neuroscience Program.
STROKE CARE NOW NETWORK CONFERENCE MAY 22, 2014 Rehabilitation Innovations in Post- Stroke Recovery Madhav Bhat, MD Fort Wayne Neurological Center DISCLOSURE Paid speaker for TEVA Neuroscience Program.
Designing eye tracking experiments to measure human behavior
 Designing eye tracking experiments to measure human behavior Eindhoven, The Netherlands August, 2010 Ricardo Matos Tobii Technology Steps involved in measuring behaviour 1. Formulate and initial question
Designing eye tracking experiments to measure human behavior Eindhoven, The Netherlands August, 2010 Ricardo Matos Tobii Technology Steps involved in measuring behaviour 1. Formulate and initial question
Agent Simulation of Hull s Drive Theory
 Agent Simulation of Hull s Drive Theory Nick Schmansky Department of Cognitive and Neural Systems Boston University March 7, 4 Abstract A computer simulation was conducted of an agent attempting to survive
Agent Simulation of Hull s Drive Theory Nick Schmansky Department of Cognitive and Neural Systems Boston University March 7, 4 Abstract A computer simulation was conducted of an agent attempting to survive
Care of the Patient with Strabismus: Esotropia and Exotropia
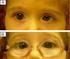 OPTOMETRIC CLINICAL PRACTICE GUIDELINE OPTOMETRY: THE PRIMARY EYE CARE PROFESSION Doctors of optometry are independent primary health care providers who examine, diagnose, treat, and manage diseases and
OPTOMETRIC CLINICAL PRACTICE GUIDELINE OPTOMETRY: THE PRIMARY EYE CARE PROFESSION Doctors of optometry are independent primary health care providers who examine, diagnose, treat, and manage diseases and
Recommended Vision Standards for Police Officers (Revised 1985)
 Recommended Vision Standards for Police Officers (Revised 1985) James E. Sheedy, O.D., Ph.D. Jeffrey T. Keller, O.D., M.P.H. Donald Pitts, O.D., Ph.D. Gerald Lowther, O.D., Ph.D. Stephen C. Miller, O.D.
Recommended Vision Standards for Police Officers (Revised 1985) James E. Sheedy, O.D., Ph.D. Jeffrey T. Keller, O.D., M.P.H. Donald Pitts, O.D., Ph.D. Gerald Lowther, O.D., Ph.D. Stephen C. Miller, O.D.
Retinal Imaging Biomarkers for Early Diagnosis of Alzheimer s Disease
 Retinal Imaging Biomarkers for Early Diagnosis of Alzheimer s Disease Eleonora (Nora) Lad, MD, PhD Assistant Professor of Ophthalmology, Vitreoretinal diseases Duke Center for Macular Diseases Duke University
Retinal Imaging Biomarkers for Early Diagnosis of Alzheimer s Disease Eleonora (Nora) Lad, MD, PhD Assistant Professor of Ophthalmology, Vitreoretinal diseases Duke Center for Macular Diseases Duke University
The Limits of Human Vision
 The Limits of Human Vision Michael F. Deering Sun Microsystems ABSTRACT A model of the perception s of the human visual system is presented, resulting in an estimate of approximately 15 million variable
The Limits of Human Vision Michael F. Deering Sun Microsystems ABSTRACT A model of the perception s of the human visual system is presented, resulting in an estimate of approximately 15 million variable
Quantifying Spatial Presence. Summary
 Quantifying Spatial Presence Cedar Riener and Dennis Proffitt Department of Psychology, University of Virginia Keywords: spatial presence, illusions, visual perception Summary The human visual system uses
Quantifying Spatial Presence Cedar Riener and Dennis Proffitt Department of Psychology, University of Virginia Keywords: spatial presence, illusions, visual perception Summary The human visual system uses
Torsional Diplopia 1,2
 Supplement Handout American Orthoptic Council Workshop: When the Patient Sees Double and the Doctor Sees Nothing A Guide to Double Vision AAPOS 37 th Annual Meeting San Diego, CA April 1, 2011 Torsional
Supplement Handout American Orthoptic Council Workshop: When the Patient Sees Double and the Doctor Sees Nothing A Guide to Double Vision AAPOS 37 th Annual Meeting San Diego, CA April 1, 2011 Torsional
Glossary of commonly used Occupational Therapy terms
 Glossary of commonly used Occupational Therapy terms Adaptive Response: An action that is appropriate and successful in meeting some environmental demand. Adaptive responses demonstrate adequate sensory
Glossary of commonly used Occupational Therapy terms Adaptive Response: An action that is appropriate and successful in meeting some environmental demand. Adaptive responses demonstrate adequate sensory
Internuclear ophthalmoplegia: recovery and plasticity
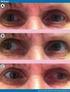 Internuclear ophthalmoplegia: recovery and plasticity M. J. Doslak, L. B. Kline, L. F. Dell'Osso, and R. B. Daroff We studied refixational eye movements of a patient during the gradual resolution of an
Internuclear ophthalmoplegia: recovery and plasticity M. J. Doslak, L. B. Kline, L. F. Dell'Osso, and R. B. Daroff We studied refixational eye movements of a patient during the gradual resolution of an
Pair B Two tasks selected from: evaluation of research data analysis essay media response annotated folio of practical activities oral presentation
 2011 School-assessed Coursework Report Psychology (2011 2014): Units 3 and 4 This report is provided for the first year of implementation of this study and is based on the coursework audit and Victorian
2011 School-assessed Coursework Report Psychology (2011 2014): Units 3 and 4 This report is provided for the first year of implementation of this study and is based on the coursework audit and Victorian
The role of context in Müller-Lyer illusion: The case of negative Müller-Lyer illusion
 Review of Psychology, 2013, Vol. 20, No. 1-2, 29-36 UDC 159.9 The role of context in Müller-Lyer illusion: The case of negative Müller-Lyer illusion PAVLE VALERJEV and TANJA GULAN The Müller-Lyer illusion
Review of Psychology, 2013, Vol. 20, No. 1-2, 29-36 UDC 159.9 The role of context in Müller-Lyer illusion: The case of negative Müller-Lyer illusion PAVLE VALERJEV and TANJA GULAN The Müller-Lyer illusion
Bernice E. Rogowitz and Holly E. Rushmeier IBM TJ Watson Research Center, P.O. Box 704, Yorktown Heights, NY USA
 Are Image Quality Metrics Adequate to Evaluate the Quality of Geometric Objects? Bernice E. Rogowitz and Holly E. Rushmeier IBM TJ Watson Research Center, P.O. Box 704, Yorktown Heights, NY USA ABSTRACT
Are Image Quality Metrics Adequate to Evaluate the Quality of Geometric Objects? Bernice E. Rogowitz and Holly E. Rushmeier IBM TJ Watson Research Center, P.O. Box 704, Yorktown Heights, NY USA ABSTRACT
Functions of the Brain
 Objectives 0 Participants will be able to identify 4 characteristics of a healthy brain. 0 Participants will be able to state the functions of the brain. 0 Participants will be able to identify 3 types
Objectives 0 Participants will be able to identify 4 characteristics of a healthy brain. 0 Participants will be able to state the functions of the brain. 0 Participants will be able to identify 3 types
AMPHETAMINE AND COCAINE MECHANISMS AND HAZARDS
 AMPHETAMINE AND COCAINE MECHANISMS AND HAZARDS BARRY J. EVERITT Department of Experimental Psychology, University of Cambridge Stimulant drugs, such as cocaine and amphetamine, interact directly with dopamine
AMPHETAMINE AND COCAINE MECHANISMS AND HAZARDS BARRY J. EVERITT Department of Experimental Psychology, University of Cambridge Stimulant drugs, such as cocaine and amphetamine, interact directly with dopamine
PLEASE SCROLL DOWN FOR ARTICLE
 This article was downloaded by:[universite Rene Descartes Paris 5] On: 2 May 28 Access Details: [subscription number 773444237] Publisher: Psychology Press Informa Ltd Registered in England and Wales Registered
This article was downloaded by:[universite Rene Descartes Paris 5] On: 2 May 28 Access Details: [subscription number 773444237] Publisher: Psychology Press Informa Ltd Registered in England and Wales Registered
13) In Piaget's theory, are psychological structures that organize experience. A) schemes B) accommodations C) assimilations D) equilibrations
 Review for Exam 2 1) When Roy first received a new toy, he played with it all the time. The longer he had it, the less he played with it. This change in Roy's behavior is an example of A) priming. B) habituation.
Review for Exam 2 1) When Roy first received a new toy, he played with it all the time. The longer he had it, the less he played with it. This change in Roy's behavior is an example of A) priming. B) habituation.
Wiring optimization in the brain
 Wiring optimization in the brain Dmitri B. Chklovskii Sloan Center for Theoretical Neurobiology The Salk Institute La Jolla, CA 92037 [email protected] Charles F. Stevens Howard Hughes Medical Institute and
Wiring optimization in the brain Dmitri B. Chklovskii Sloan Center for Theoretical Neurobiology The Salk Institute La Jolla, CA 92037 [email protected] Charles F. Stevens Howard Hughes Medical Institute and
Lecture 2, Human cognition
 Human Cognition An important foundation for the design of interfaces is a basic theory of human cognition The information processing paradigm (in its most simple form). Human Information Processing The
Human Cognition An important foundation for the design of interfaces is a basic theory of human cognition The information processing paradigm (in its most simple form). Human Information Processing The
Interpretive Report of WMS IV Testing
 Interpretive Report of WMS IV Testing Examinee and Testing Information Examinee Name Date of Report 7/1/2009 Examinee ID 12345 Years of Education 11 Date of Birth 3/24/1988 Home Language English Gender
Interpretive Report of WMS IV Testing Examinee and Testing Information Examinee Name Date of Report 7/1/2009 Examinee ID 12345 Years of Education 11 Date of Birth 3/24/1988 Home Language English Gender
Have you ever missed a call while moving? : The Optimal Vibration Frequency for Perception in Mobile Environments
 Have you ever missed a call while moving? : The Optimal Vibration Frequency for Perception in Mobile Environments Youngmi Baek and Rohae Myung Dept. of Industrial and Information Engineering Korea University
Have you ever missed a call while moving? : The Optimal Vibration Frequency for Perception in Mobile Environments Youngmi Baek and Rohae Myung Dept. of Industrial and Information Engineering Korea University
Strategic Plan Proposal: Learning science by experiencing science: A proposal for new active learning courses in Psychology
 Strategic Plan Proposal: Learning science by experiencing science: A proposal for new active learning courses in Psychology Contacts: Jacob Feldman, ([email protected], 848-445-1621) Eileen Kowler
Strategic Plan Proposal: Learning science by experiencing science: A proposal for new active learning courses in Psychology Contacts: Jacob Feldman, ([email protected], 848-445-1621) Eileen Kowler
The Physiology of the Senses Lecture 1 - The Eye www.tutis.ca/senses/
 The Physiology of the Senses Lecture 1 - The Eye www.tutis.ca/senses/ Contents Objectives... 2 Introduction... 2 Accommodation... 3 The Iris... 4 The Cells in the Retina... 5 Receptive Fields... 8 The
The Physiology of the Senses Lecture 1 - The Eye www.tutis.ca/senses/ Contents Objectives... 2 Introduction... 2 Accommodation... 3 The Iris... 4 The Cells in the Retina... 5 Receptive Fields... 8 The
PASSENGER/PEDESTRIAN ANALYSIS BY NEUROMORPHIC VISUAL INFORMATION PROCESSING
 PASSENGER/PEDESTRIAN ANALYSIS BY NEUROMORPHIC VISUAL INFORMATION PROCESSING Woo Joon Han Il Song Han Korea Advanced Science and Technology Republic of Korea Paper Number 13-0407 ABSTRACT The physiological
PASSENGER/PEDESTRIAN ANALYSIS BY NEUROMORPHIC VISUAL INFORMATION PROCESSING Woo Joon Han Il Song Han Korea Advanced Science and Technology Republic of Korea Paper Number 13-0407 ABSTRACT The physiological
Tonal Detection in Noise: An Auditory Neuroscience Insight
 Image: http://physics.ust.hk/dusw/ Tonal Detection in Noise: An Auditory Neuroscience Insight Buddhika Karunarathne 1 and Richard H.Y. So 1,2 1 Dept. of IELM, Hong Kong University of Science & Technology,
Image: http://physics.ust.hk/dusw/ Tonal Detection in Noise: An Auditory Neuroscience Insight Buddhika Karunarathne 1 and Richard H.Y. So 1,2 1 Dept. of IELM, Hong Kong University of Science & Technology,
WHAT IS CEREBRAL PALSY?
 WHAT IS CEREBRAL PALSY? Cerebral Palsy is a dysfunction in movement resulting from injury to or poor development of the brain prior to birth or in early childhood. Generally speaking, any injury or disease
WHAT IS CEREBRAL PALSY? Cerebral Palsy is a dysfunction in movement resulting from injury to or poor development of the brain prior to birth or in early childhood. Generally speaking, any injury or disease
