TRANSPARENCY COMMITTEE OPINION. 5 May 2010
|
|
|
- Joleen Stokes
- 7 years ago
- Views:
Transcription
1 The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 5 May 2010 XILANIK 100 mg/20 mg, modified-release capsule B/14 (CIP: ) XILANIK 200 mg/20 mg, modified-release capsule B/14 (CIP: ) Applicant: PIERRE FABRE MEDICAMENT Ketoprofen, omeprazole ATC code: M01AE53 List II Date of Marketing Authorisation: 15 September 2009 (national procedure) Reason for request: Inclusion on the list of medicines reimbursed by National Health Insurance and approved for hospital use. Medical, Economic and Public Health Assessment Division 1
2 1 CHARACTERISTICS OF THE MEDICINAL PRODUCT 1.1. Active ingredient Ketoprofen, omeprazole 1.2. Indications "For symptomatic treatment, following on from a previous association of ketoprofen and omeprazole: chronic inflammatory rheumatic conditions, particularly rheumatoid arthritis and ankylosing spondylitis, osteoarthritis. In the following types of patient: - patients with a previous history of NSAID-associated gastroduodenal erosions in whom continued treatment with a NSAID is essential; - patients who are at risk of developing gastroduodenal erosions (particularly those aged > 65 years or with a history of gastroduodenal ulcer) in whom continued treatment with a NSAID is essential. This fixed-dose combination of ketoprofen and omeprazole should not be used as an initial symptomatic treatment" Dosage "Undesirable effects may be minimised by using the lowest effective dose for the shortest duration necessary to control symptoms. Method of administration Oral use. Adults and adolescents over the age of 15 years: This product is not recommended for use in children below 15 years due to a lack of data on tolerance and efficacy in these indications. The capsule should be swallowed whole with food once daily, preferably in the morning, with a large glass of water. Dose and administration route The usual daily dose is 200 mg of ketoprofen and 20 mg omeprazole; however, 100 mg of ketoprofen in combination with 20 mg of omeprazole may be sufficient for some patients. At-risk populations: It is recommended that treatment be started with an initial daily dose of 100 mg / 20 mg of XILANIK prolonged-release capsules for such patients. Use of XILANIK 200 mg / 20 mg prolonged-release capsules must be guided by efficacy and renal safety. The 100 mg ketoprofen dose should be favoured in elderly patients, those with chronic heart failure and those with renal failure (creatinine clearance ml/min) or liver failure." 2
3 1.4 Contraindications - Hypersensitivity to ketoprofen or to omeprazole or to any of the excipients. - This product contains sucrose and is contraindicated in patients with fructose intolerance, glucose-galactose malabsorption or sucrase-isomaltase deficiency. o Linked to ketoprofen - Beyond the 24th week of pregnancy (5 months); - history of asthma induced by administration of ketoprofen or similar acting substances, such as other non-steroidal anti-inflammatory agents (NSAIDs) or acetylsalicylic acid; - history of haemorrhage or digestive tract perforation during previous NSAID treatment; - progressive peptic ulcer, history of peptic ulcer or recurrent bleeding (2 or more separate episodes of bleeding or ulceration observed); - severe hepatic failure; - severe renal failure; - severe heart failure; - gastrointestinal bleeding, cerebrovascular bleeding or other active bleeding. Note: these contraindications contradict the indication wording in the marketing authorisation for XILANIK. 2 SIMILAR MEDICINAL PRODUCTS 2.1. ATC Classification (2010) M : Musculo-skeletal system M01 : Antiinflammatory and antirheumatic products M01A : Antiinflammatory and antirheumatic products, non-steroids M01AE : Propionic acid derivatives M01AE53 : Ketoprofen, combinations 2.2. Medicines in the same therapeutic category Comparator medicines There are no other fixed-dose combinations of a non-steroidal anti-inflammatory (NSAID) and a proton pump inhibitor (PPI) included in the list of medicines reimbursed by National Insurance Medicines with a similar therapeutic aim - Fixed-dose combination: diclofenac + misoprostol (ARTOTEC), currently being reassessed by the Transparency Committee. - All NSAIDs prescribed in combination with misoprostol or PPIs. 3
4 3 ANALYSIS OF AVAILABLE DATA 3.1. Efficacy The application does not include any clinical studies assessing the efficacy of a fixed-dose combination of ketoprofen and omeprazole (XILANIK) in the indication given in the Marketing Authorisation. The Marketing Authorisation was granted on the basis of a bibliography-based application alone, which included six clinical studies that evaluated the efficacy of omeprazole in the treatment and prevention of recurrence of gastric and duodenal lesions in patients on NSAID ketoprofen (1, 2, 3, 4, 5, 6). treatment, including 3.2. Adverse effects The tolerance of XILANIK has not been evaluated specifically. According to the conclusions of the EMEA 2006 reassessment of the adverse effects of NSAIDs on the cardiovascular and gastrointestinal systems and the skin7, the benefits of ketoprofen outweigh its risks for daily doses up to a maximum of 200 mg. The SPC for XILANIK includes special warnings concerning gastrointestinal effects, as follows: "Omeprazole is an anti-secretory medicine that acts against ulcers. However, because of the possible serious nature of gastrointestinal bleeding, particular caution should be advised in patients receiving concomitant medications which could increase the risk of ulceration or bleeding, such as oral corticosteroids, anticoagulants such as warfarin, selective serotonin reuptake inhibitors or anti-platelet agents such as aspirin. When GI bleeding or ulceration occurs in patients receiving XILANIK, the treatment should be withdrawn. XILANIK modifiedrelease capsules should be given with care and under close monitoring to patients with a history of gastrointestinal disease (ulcerative colitis, Crohn's disease) as these conditions may be exacerbated." However, according to the AFSSAPS 2007 guidelines 8, the following at-risk populations may require NSAID treatment in combination with a PPI: - persons aged over 65 years - history of complicated or uncomplicated gastric or duodenal ulcer. In such cases, Helicobacter pylori should be tested for and treated. - combination with anti-platelet therapy, particularly low-dose aspirin and clopidogrel, and/or corticosteroids and/or an anticoagulant (bearing in mind that such combinations should in principle be avoided). 1 Hawkey C J et al. Omeprazole compared with misoprostol for ulcers associated with nonsteroidal antiinflammatory drugs. N Engl J Med 1998; 338 : Yeomans N D et al. A comparison of omeprazole with ranitidine for ulcers associated with nonsteroidal antiinflammatory drugs. N Engl J Med 1998; 338 : Bianchi Porro M et al. Prevention of gastroduodenal damage with omeprazole in patients receiving continuous NSAIDs treatment. A double blind placebo controlled study. Ital.J Gastroenterol. Hepatol 1998;30: Cullen et al. Primary gastroduodenal prophylaxis with omeprazole for non-steroidal anti-inflammatory drug users. Aliment pharmacol Ther 1998; 12: Ekstrom P et al. Prevention of peptic ulcer and dyspeptic symptoms with omeprazole in patients receiving continuous non-steroidal anti-inflammatory drug therapy. Scand J Gastrol. 1996; 31: Massimo C G et al. Omeprazole 20 or 40 mg for healing gastroduodenal ulcers in patients receiving nonsteroidal anti-inflammatory drugs. Aliment pharmacol Ther 1998; 12: EMEA. Public CHMP assessment report for medicinal products containing non-selective non steroidal antiinflammatory drugs (NSAIDs) - November AFSSAPS. Recommandations de bonne pratique : les antisécrétoires gastriques chez l adulte. [Good practice guidelines: gastric secretion inhibitors in adults.] November
5 3.3. Conclusion In the absence of studies conducted specifically on XILANIK products, it is not possible to assess the clinical benefit of these products in comparison with separate dosing with a NSAID and a PPI. 4 TRANSPARENCY COMMITTEE CONCLUSIONS 4.1. Actual benefit Rheumatic conditions can lead to disability and deterioration in quality of life. XILANIK products are indicated for the conditions stated in the wording of the marketing authorisation, as a continuation treatment following previous combination treatment with ketoprofen and omeprazole for symptomatic treatment of pain and inflammation in at-risk patients for whom NSAID treatment is essential. In the absence of any studies of their efficacy and tolerance, the efficacy/adverse effect ratio of these products cannot be stated. Public health benefit The public health burden caused by chronic inflammatory rheumatic conditions and osteoarthritis, which are chronic and disabling diseases, is considered to be significant. The burden represented by the sub-population of patients who are likely to benefit from this treatment (patients aged over 65 or with a history of gastric or duodenal ulcer) is moderate, because of the more restricted patient numbers involved. Improvement in management and quality of life of patients with these rheumatic conditions remains a public health need which is included in the list of established priorities (Public Health Law 2004, Plan to improve quality of life of patients with chronic diseases). However, existing treatments (including flexible combinations of ketoprofen and omeprazole) already help to meet this need. There is no indication that these fixed-dose combinations have any added benefit (even in terms of increased compliance) over flexible combinations of the two active substances. As a result, XILANIK is not expected to benefit public health. In the absence of comparative data, the role of the fixed-dose combination of ketoprofen + omeprazole (XILANIK) in therapeutic strategy in comparison with flexible NSAID + PPI combinations cannot be assessed. 5
6 The Transparency Committee also notes that: - this fixed-dose combination carries the risk of unnecessary use of ketoprofen, which has been shown to have poor digestive tract tolerance. - making this fixed-dose combination available entails a risk of over-prescription and misuse of PPIs. On the one hand, the ease of use of a dual therapy contained in one capsule carries the risk that prescription of PPIs will shift away from short courses for the treatment of acute conditions, which is contrary to the guidelines on the correct use of PPIs published by HAS 9. On the other hand, there is the risk that prescription may extend to patients who do not risk developing NSAID-induced gastric and duodenal lesions as defined in the marketing authorisation; - other treatment options are available. As a consequence of this, the Transparency Committee considers that the actual clinical benefit of these products, in comparison with the treatments that are already available, is not sufficient to allow it to be reimbursed by National Insurance Transparency Committee recommendations The Transparency Committee does not recommend inclusion on the list of medicines reimbursed by National Health Insurance and on the list of medicines approved for use by hospitals and various public services. 9 Bon usage des Médicaments. Les inhibiteurs de la pompe à protons chez l adulte. Haute Autorité de Santé. Décembre
A. Ketorolac*** B. Naproxen C. Ibuprofen D. Celecoxib
 1. A man, 66 years of age, with a history of knee osteoarthritis (OA) is experiencing increasing pain at rest and with physical activity. He also has a history of depression and coronary artery disease.
1. A man, 66 years of age, with a history of knee osteoarthritis (OA) is experiencing increasing pain at rest and with physical activity. He also has a history of depression and coronary artery disease.
TRANSPARENCY C OMMITTEE
 The legally binding text is the original French version TRANSPARENCY C OMMITTEE Opinion 20 November 2013 VOLTAFLEX 625 mg, film-coated tablet B/60 tablets (CIP: 34009 384 573 2) Applicant: NOVARTIS SANTÉ
The legally binding text is the original French version TRANSPARENCY C OMMITTEE Opinion 20 November 2013 VOLTAFLEX 625 mg, film-coated tablet B/60 tablets (CIP: 34009 384 573 2) Applicant: NOVARTIS SANTÉ
What s the Deal with NSAIDs?
 Focus on CME at the Memorial xxx University of Newfoundland What s the Deal with NSAIDs? Majed M. Khraishi, MB, BCh, FRCPC Presented at Wednesday at Noon Ask the Consultant Arthritis is one of the most
Focus on CME at the Memorial xxx University of Newfoundland What s the Deal with NSAIDs? Majed M. Khraishi, MB, BCh, FRCPC Presented at Wednesday at Noon Ask the Consultant Arthritis is one of the most
Essential Shared Care Agreement Drugs for Dementia
 Ref No. E040 Essential Shared Care Agreement Drugs for Dementia Please complete the following details: Patient s name, address, date of birth Consultant s contact details (p.3) And send One copy to: 1.
Ref No. E040 Essential Shared Care Agreement Drugs for Dementia Please complete the following details: Patient s name, address, date of birth Consultant s contact details (p.3) And send One copy to: 1.
Psoriasis and Psoriatic Arthritis Alliance
 Psoriasis and Psoriatic Arthritis Alliance A principal source of information on psoriasis and psoriatic arthritis ) Treatments for Psoriatic Arthritis overview Although psoriatic arthritis is a chronic
Psoriasis and Psoriatic Arthritis Alliance A principal source of information on psoriasis and psoriatic arthritis ) Treatments for Psoriatic Arthritis overview Although psoriatic arthritis is a chronic
Articles Presented. Journal Presentation. Dr Albert Lo. Dr Albert Lo
 * This presentation is prepared by the author in one s personal capacity for the purpose of academic exchange and does not represent the views of his/her organisations on the topic discussed. Journal Presentation
* This presentation is prepared by the author in one s personal capacity for the purpose of academic exchange and does not represent the views of his/her organisations on the topic discussed. Journal Presentation
understanding GI bleeding
 understanding GI bleeding a consumer education brochure American College of Gastroenterology 4900B South 31st Street, Arlington, VA 22206 703-820-7400 www.acg.gi.org American College of Gastroenterology
understanding GI bleeding a consumer education brochure American College of Gastroenterology 4900B South 31st Street, Arlington, VA 22206 703-820-7400 www.acg.gi.org American College of Gastroenterology
patient group direction
 DICLOFENAC v01 1/8 DICLOFENAC PGD Details Version 1.0 Legal category Staff grades Approved by POM Paramedic (Non-ECP) Nurse (Non-ECP) Emergency Care Practitioner (Paramedic) Emergency Care Practitioner
DICLOFENAC v01 1/8 DICLOFENAC PGD Details Version 1.0 Legal category Staff grades Approved by POM Paramedic (Non-ECP) Nurse (Non-ECP) Emergency Care Practitioner (Paramedic) Emergency Care Practitioner
Ask Your Doctor if There May Be a SMARTER CHOICE
 If you have osteoarthritis, rheumatoid arthritis or ankylosing spondylitis, Could Your NSAID Pain Medicine Be Hurting Your Stomach? Ask Your Doctor if There May Be a SMARTER CHOICE 1 of 8 Making Smart
If you have osteoarthritis, rheumatoid arthritis or ankylosing spondylitis, Could Your NSAID Pain Medicine Be Hurting Your Stomach? Ask Your Doctor if There May Be a SMARTER CHOICE 1 of 8 Making Smart
Over the Counter Drugs (OTCs): Considerations for Physical Therapy Practice in Canada
 Background Over the Counter Drugs (OTCs): Considerations for Physical Therapy Practice in Canada The use of medications or drugs by non-physician health professionals is evolving and is linked to collaboration
Background Over the Counter Drugs (OTCs): Considerations for Physical Therapy Practice in Canada The use of medications or drugs by non-physician health professionals is evolving and is linked to collaboration
Gloucestershire Hospitals
 TRUST GUIDELINE In the case of hard copies of this policy the content can only be assured to be accurate on the date of issue marked on the document. The Policy framework requires that the policy is fully
TRUST GUIDELINE In the case of hard copies of this policy the content can only be assured to be accurate on the date of issue marked on the document. The Policy framework requires that the policy is fully
There is a risk of renal impairment in dehydrated children and adolescents.
 PACKAGE LEAFLET: INFORMATION FOR THE USER MELFEN 200mg FILM-COATED TABLETS MELFEN 400mg FILM-COATED TABLETS Ibuprofen Read all of this leaflet carefully before you start taking this medicine because it
PACKAGE LEAFLET: INFORMATION FOR THE USER MELFEN 200mg FILM-COATED TABLETS MELFEN 400mg FILM-COATED TABLETS Ibuprofen Read all of this leaflet carefully before you start taking this medicine because it
What can I eat? Peptic ulcers. What are peptic ulcers? What tests are needed? Will the ulcer come back? What causes a peptic ulcer?
 In association with: INFORMATION ABOUT Peptic ulcers www.corecharity.org.uk What are peptic ulcers? What causes a peptic ulcer? How are NSAIDs and aspirin involved? How do I know if I ve got an ulcer?
In association with: INFORMATION ABOUT Peptic ulcers www.corecharity.org.uk What are peptic ulcers? What causes a peptic ulcer? How are NSAIDs and aspirin involved? How do I know if I ve got an ulcer?
New Anticoagulants and GI bleeding
 New Anticoagulants and GI bleeding DR DANNY MYERS MD FRCP(C) CLINICAL ASSISTANT PROFESSOR OF MEDICINE, UBC Conflicts of Interest None I am unbiased in the use of NOAC s vs Warfarin based on risk benefit
New Anticoagulants and GI bleeding DR DANNY MYERS MD FRCP(C) CLINICAL ASSISTANT PROFESSOR OF MEDICINE, UBC Conflicts of Interest None I am unbiased in the use of NOAC s vs Warfarin based on risk benefit
UpRight Aceclofenac 100 mg and Paracetamol 500 mg fixed dose combination
 For the use of a Registered Medical Practitioner or a Hospital or a Laboratory only UpRight Aceclofenac 100 mg and Paracetamol 500 mg fixed dose combination DESCRIPTION UPRIGHT is a fixed dose combination
For the use of a Registered Medical Practitioner or a Hospital or a Laboratory only UpRight Aceclofenac 100 mg and Paracetamol 500 mg fixed dose combination DESCRIPTION UPRIGHT is a fixed dose combination
Appendix C Factors to consider when choosing between anticoagulant options and FAQs
 Appendix C Factors to consider when choosing between anticoagulant options and FAQs Choice of anticoagulant for non-valvular* atrial fibrillation: Clinical decision aid Patients should already be screened
Appendix C Factors to consider when choosing between anticoagulant options and FAQs Choice of anticoagulant for non-valvular* atrial fibrillation: Clinical decision aid Patients should already be screened
Dabigatran: Amber Drug Guidance for the prevention of stroke and systemic embolism in patients with non-valvular AF
 Leeds Dabigatran: Amber Drug Guidance for the prevention of stroke and systemic embolism in patients with non-valvular AF Amber Drug Level 3 (amber drug with monitoring requirements) We have started your
Leeds Dabigatran: Amber Drug Guidance for the prevention of stroke and systemic embolism in patients with non-valvular AF Amber Drug Level 3 (amber drug with monitoring requirements) We have started your
Mucodyne-Clear 250 mg/5 ml Syrup PL 04425/0665
 Mucodyne-Clear 250 mg/5 ml Syrup PL 04425/0665 UKPAR TABLE OF CONTENTS Lay summary Page 2 Scientific discussion Page 3 Steps taken for assessment Page 9 Summary of product characteristics Page 10 Patient
Mucodyne-Clear 250 mg/5 ml Syrup PL 04425/0665 UKPAR TABLE OF CONTENTS Lay summary Page 2 Scientific discussion Page 3 Steps taken for assessment Page 9 Summary of product characteristics Page 10 Patient
PACKAGE INSERT TEMPLATE FOR ACETYLSALICYLIC ACID/ASPIRIN TABLET
 PACKAGE INSERT TEMPLATE FOR ACETYLSALICYLIC ACID/ASPIRIN TABLET Brand or Product Name [Product name] tablet 500mg Name and Strength of Active Substance(s) Acetylsalicylic acid.500mg Product Description
PACKAGE INSERT TEMPLATE FOR ACETYLSALICYLIC ACID/ASPIRIN TABLET Brand or Product Name [Product name] tablet 500mg Name and Strength of Active Substance(s) Acetylsalicylic acid.500mg Product Description
Acetylcholinesterase Inhibitors and Memantine Clinical Indication: Treatment of Dementia in Alzheimer s Disease (AD)
 SHARED CARE PROTOCOL AND INFORMATION FOR GPS Acetylcholinesterase Inhibitors and Memantine Clinical Indication: Treatment of Dementia in Alzheimer s Disease (AD) Version: 3 Date Approved: June 2011 Review
SHARED CARE PROTOCOL AND INFORMATION FOR GPS Acetylcholinesterase Inhibitors and Memantine Clinical Indication: Treatment of Dementia in Alzheimer s Disease (AD) Version: 3 Date Approved: June 2011 Review
Donepezil (Aricept ), Galantamine (Reminyl XL ), Rivastigmine (Exelon ) and Memantine (Ebixa )
 Donepezil (Aricept ), Galantamine (Reminyl XL ), Rivastigmine (Exelon ) and Memantine (Ebixa ) ESCA: For the treatment of Alzheimer s disease. SECONDARY CARE SECTION TO BE COMPLETED BY INITIATING DOCTOR
Donepezil (Aricept ), Galantamine (Reminyl XL ), Rivastigmine (Exelon ) and Memantine (Ebixa ) ESCA: For the treatment of Alzheimer s disease. SECONDARY CARE SECTION TO BE COMPLETED BY INITIATING DOCTOR
National Digestive Diseases Information Clearinghouse
 Gastritis National Digestive Diseases Information Clearinghouse U.S. Department of Health and Human Services NATIONAL INSTITUTES OF HEALTH What is gastritis? Gastritis is a condition in which the stomach
Gastritis National Digestive Diseases Information Clearinghouse U.S. Department of Health and Human Services NATIONAL INSTITUTES OF HEALTH What is gastritis? Gastritis is a condition in which the stomach
Patient Group Direction Hospital: Bristol Royal Infirmary Department: UHBristol Thrombosis Service University Hospitals Bristol NHS Foundation Trust.
 Patient Group Direction Hospital: Bristol Royal Infirmary Department: UHBristol Thrombosis Service University Hospitals Bristol NHS Foundation Trust. This Patient Group Direction (PGD) has been written
Patient Group Direction Hospital: Bristol Royal Infirmary Department: UHBristol Thrombosis Service University Hospitals Bristol NHS Foundation Trust. This Patient Group Direction (PGD) has been written
NSAID PREPARATIONS. COMPOSITION : Each enteric coated tablet contains Diclofenac Sodium BP 25 mg. Clofenac 50. Clofenac SR. Sodium BP 50 mg.
 Diclofenac Analgesic & Anti-inflammatory COMPOSITION Clofenac 25 : Each enteric coated tablet contains Diclofenac Sodium BP 25 mg. Clofenac 50 : Each enteric coated tablet contains Diclofenac Sodium BP
Diclofenac Analgesic & Anti-inflammatory COMPOSITION Clofenac 25 : Each enteric coated tablet contains Diclofenac Sodium BP 25 mg. Clofenac 50 : Each enteric coated tablet contains Diclofenac Sodium BP
PATIENT INFORMATION LEAFLET Phor Migraine (Ibuprofen)
 PATIENT INFORMATION LEAFLET Phor Migraine (Ibuprofen) Read all of this leaflet carefully before you start taking this medicine. Keep this leaflet. You may need to read it again. If you have any further
PATIENT INFORMATION LEAFLET Phor Migraine (Ibuprofen) Read all of this leaflet carefully before you start taking this medicine. Keep this leaflet. You may need to read it again. If you have any further
HERTFORDSHIRE MEDICINES MANAGEMENT COMMITTEE (HMMC) RIVAROXABAN RECOMMENDED see specific recommendations for licensed indications below
 Name: generic (trade) Rivaroxaban (Xarelto ) HERTFORDSHIRE MEDICINES MANAGEMENT COMMITTEE (HMMC) RIVAROXABAN RECOMMENDED see specific recommendations for licensed indications below What it is Indications
Name: generic (trade) Rivaroxaban (Xarelto ) HERTFORDSHIRE MEDICINES MANAGEMENT COMMITTEE (HMMC) RIVAROXABAN RECOMMENDED see specific recommendations for licensed indications below What it is Indications
National Institute for Clinical Excellence
 National Institute for Clinical Excellence 11 Strand London WC2N 5HR Web: www.nice.org.uk N0016 50k 1P July 01 (ABA) Technology Appraisal Guidance - No.27 Guidance on the use of cyclo-oxygenase (Cox) II
National Institute for Clinical Excellence 11 Strand London WC2N 5HR Web: www.nice.org.uk N0016 50k 1P July 01 (ABA) Technology Appraisal Guidance - No.27 Guidance on the use of cyclo-oxygenase (Cox) II
Comparative Effectiveness Review Number 4. Comparative Effectiveness and Safety of Analgesics for Osteoarthritis
 This report is based on research conducted by the Oregon Evidence-based Practice Center (EPC) under contract to the Agency for Healthcare Research and Quality (AHRQ), Rockville, MD (Contract No. 290-02-0024).
This report is based on research conducted by the Oregon Evidence-based Practice Center (EPC) under contract to the Agency for Healthcare Research and Quality (AHRQ), Rockville, MD (Contract No. 290-02-0024).
Prescriber Guide. 20mg. 15mg. Simply Protecting More Patients. Simply Protecting More Patients
 Prescriber Guide 20mg Simply Protecting More Patients 15mg Simply Protecting More Patients 1 Dear Doctor, This prescriber guide was produced by Bayer Israel in cooperation with the Ministry of Health as
Prescriber Guide 20mg Simply Protecting More Patients 15mg Simply Protecting More Patients 1 Dear Doctor, This prescriber guide was produced by Bayer Israel in cooperation with the Ministry of Health as
Dabigatran (Pradaxa) for stroke prevention in patients with non-valvular atrial fibrillation (da-big-a-tran)
 Dabigatran (Pradaxa) 1 Dabigatran (Pradaxa) for stroke prevention in patients with non-valvular atrial fibrillation (da-big-a-tran) Similar rate of major bleeding to warfarin KEY POINTS Dabigatran is an
Dabigatran (Pradaxa) 1 Dabigatran (Pradaxa) for stroke prevention in patients with non-valvular atrial fibrillation (da-big-a-tran) Similar rate of major bleeding to warfarin KEY POINTS Dabigatran is an
Canadian consensus guidelines on long-term nonsteroidal anti-inflammatory drug therapy and the need for gastroprotection: benefits versus risks
 Alimentary Pharmacology & Therapeutics Canadian consensus guidelines on long-term nonsteroidal anti-inflammatory drug therapy and the need for gastroprotection: benefits versus risks A. ROSTOM*, P. MOAYYEDI
Alimentary Pharmacology & Therapeutics Canadian consensus guidelines on long-term nonsteroidal anti-inflammatory drug therapy and the need for gastroprotection: benefits versus risks A. ROSTOM*, P. MOAYYEDI
SUMMARY OF PRODUCT CHARACTERISTICS BREXIN. 3. Pharmaceutical Form Tablet Pale yellow, hexagonal tablet with a median score line on one side
 פורמט עלון זה נקבע ע"י משרד הבריאות ותוכנו נבדק ואושר SUMMARY OF PRODUCT CHARACTERISTICS 1. Name of the Medicinal Product BREXIN 20 mg TABLETS BREXIN 2. Qualitative and Quantitative Composition Each tablet
פורמט עלון זה נקבע ע"י משרד הבריאות ותוכנו נבדק ואושר SUMMARY OF PRODUCT CHARACTERISTICS 1. Name of the Medicinal Product BREXIN 20 mg TABLETS BREXIN 2. Qualitative and Quantitative Composition Each tablet
Three new/novel oral anticoagulants (NOAC) have been licensed in Ireland since 2008:
 Key Points to consider when prescribing NOACs Introduction Three new/novel oral anticoagulants (NOAC) have been licensed in Ireland since 2008: Dabigatran Etexilate (Pradaxa ) 75mg, 110mg, 150mg. Rivaroxaban
Key Points to consider when prescribing NOACs Introduction Three new/novel oral anticoagulants (NOAC) have been licensed in Ireland since 2008: Dabigatran Etexilate (Pradaxa ) 75mg, 110mg, 150mg. Rivaroxaban
Peptic Ulcer. Anatomy The stomach is a hollow organ. It is located in the upper abdomen, under the ribs.
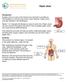 Peptic Ulcer Introduction A peptic ulcer is a sore in the lining of your stomach or duodenum. The duodenum is the first part of your small intestine. Peptic ulcers may also develop in the esophagus. Nearly
Peptic Ulcer Introduction A peptic ulcer is a sore in the lining of your stomach or duodenum. The duodenum is the first part of your small intestine. Peptic ulcers may also develop in the esophagus. Nearly
Updates to the Alberta Human Services Drug Benefit Supplement
 Updates to the Alberta Human Services Drug Benefit Supplement Effective January 1, 2016 Inquiries should be directed to: Pharmacy Services Alberta Blue Cross 10009 108 Street NW Edmonton AB T5J 3C5 Telephone
Updates to the Alberta Human Services Drug Benefit Supplement Effective January 1, 2016 Inquiries should be directed to: Pharmacy Services Alberta Blue Cross 10009 108 Street NW Edmonton AB T5J 3C5 Telephone
Rivaroxaban (Xarelto) for preventing venous thromboembolism after hip or knee replacement surgery
 for preventing venous thromboembolism after hip or knee replacement surgery (riv-ah-rocks-ah-ban) Summary Rivaroxaban is an oral anticoagulant and the first direct factor Xa inhibitor. Rivaroxaban has
for preventing venous thromboembolism after hip or knee replacement surgery (riv-ah-rocks-ah-ban) Summary Rivaroxaban is an oral anticoagulant and the first direct factor Xa inhibitor. Rivaroxaban has
THE COMPARATIVE OUTCOME OF TOPICAL FORMULATIONS IN RHEUMATOID ARTHRITIS: A PROSPECTIVE STUDY IN 125 PATIENTS OF AGE GROUPS 40-60 YEARS:
 Page972 Indo American Journal of Pharmaceutical Research, 2015 ISSN NO: 2231-6876 THE COMPARATIVE OUTCOME OF TOPICAL FORMULATIONS IN RHEUMATOID ARTHRITIS: A PROSPECTIVE STUDY IN 125 PATIENTS OF AGE GROUPS
Page972 Indo American Journal of Pharmaceutical Research, 2015 ISSN NO: 2231-6876 THE COMPARATIVE OUTCOME OF TOPICAL FORMULATIONS IN RHEUMATOID ARTHRITIS: A PROSPECTIVE STUDY IN 125 PATIENTS OF AGE GROUPS
Rivaroxaban: Amber Drug Guidance for the prevention of stroke and systemic embolism in patients with non-valvular AF
 Leeds Rivaroxaban: Amber Drug Guidance for the prevention of stroke and systemic embolism in patients with non-valvular AF Amber Drug Level 3 (amber drug with monitoring requirements) We have started your
Leeds Rivaroxaban: Amber Drug Guidance for the prevention of stroke and systemic embolism in patients with non-valvular AF Amber Drug Level 3 (amber drug with monitoring requirements) We have started your
Evidence-based Management of Rheumatoid Arthritis (2009)
 CPLD reviews its distance learning programmes every twelve months to ensure currency. This update has been produced by an expert and should be read in conjunction with the Evidencebased Management of distance
CPLD reviews its distance learning programmes every twelve months to ensure currency. This update has been produced by an expert and should be read in conjunction with the Evidencebased Management of distance
SUMMARY OF PRODUCT CHARACTERISTICS
 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE VETERINARY MEDICINAL PRODUCT Prednicare Tablets 5mg 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each tablet contains: Active Substance(s) Prednisolone
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE VETERINARY MEDICINAL PRODUCT Prednicare Tablets 5mg 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each tablet contains: Active Substance(s) Prednisolone
Updates to the Alberta Drug Benefit List. Effective January 1, 2016
 Updates to the Alberta Drug Benefit List Effective January 1, 2016 Inquiries should be directed to: Pharmacy Services Alberta Blue Cross 10009 108 Street NW Edmonton AB T5J 3C5 Telephone Number: (780)
Updates to the Alberta Drug Benefit List Effective January 1, 2016 Inquiries should be directed to: Pharmacy Services Alberta Blue Cross 10009 108 Street NW Edmonton AB T5J 3C5 Telephone Number: (780)
Package leaflet: Information for the user. < ASPROFLASH and associated names > 500 mg coated tablet Acetylsalicylic acid
 PACKAGE LEAFLET 1 Package leaflet: Information for the user < ASPROFLASH and associated names > 500 mg coated tablet Acetylsalicylic acid Read all of this leaflet carefully before you start using this
PACKAGE LEAFLET 1 Package leaflet: Information for the user < ASPROFLASH and associated names > 500 mg coated tablet Acetylsalicylic acid Read all of this leaflet carefully before you start using this
THE CLINICAL USE OF NATURAL ANTI-INFLAMMATORY HERBS AND SUPPLEMENTS JAMES MESCHINO DC, MS, ND
 THE CLINICAL USE OF NATURAL ANTI-INFLAMMATORY HERBS AND SUPPLEMENTS JAMES MESCHINO DC, MS, ND As reported by Gottlieb in 1997, the management of osteoarthritis should include specific dietary and supplementation
THE CLINICAL USE OF NATURAL ANTI-INFLAMMATORY HERBS AND SUPPLEMENTS JAMES MESCHINO DC, MS, ND As reported by Gottlieb in 1997, the management of osteoarthritis should include specific dietary and supplementation
TRANSPARENCY COMMITTEE OPINION. 26 November 2008
 The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 26 November 2008 PIASCLEDINE 300 mg capsules Pack of 15 capsules (CIP: 321 495-4) Applicant : EXPANSCIENCE Avocado
The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 26 November 2008 PIASCLEDINE 300 mg capsules Pack of 15 capsules (CIP: 321 495-4) Applicant : EXPANSCIENCE Avocado
TSOAC Initiation Checklist
 Task Establish appropriate dose based on anticoagulant selected, indication and patient factors such as renal function. Evaluate for medication interactions that may necessitate TSOAC dose adjustment.
Task Establish appropriate dose based on anticoagulant selected, indication and patient factors such as renal function. Evaluate for medication interactions that may necessitate TSOAC dose adjustment.
Single Blind, Comparative Study of Ketoprofen Cream Vs Diclofenac and Piroxicam Cream in Management of Rheumatoid Arthritis Patients
 International Journal of Pharma Sciences Vol. 4, No. 5 (2014): 697-701 Research Article Open Access ISSN: 2320-6810 Single Blind, Comparative Study of Ketoprofen Vs Diclofenac and Piroxicam in Management
International Journal of Pharma Sciences Vol. 4, No. 5 (2014): 697-701 Research Article Open Access ISSN: 2320-6810 Single Blind, Comparative Study of Ketoprofen Vs Diclofenac and Piroxicam in Management
EMEA PUBLIC STATEMENT ON LEFLUNOMIDE (ARAVA) - SEVERE AND SERIOUS HEPATIC REACTIONS -
 The European Agency for the Evaluation of Medicinal Products Post-authorisation evaluation of medicines for human use London, 12 March 2001 Doc. Ref: EMEA/H/5611/01/en EMEA PUBLIC STATEMENT ON LEFLUNOMIDE
The European Agency for the Evaluation of Medicinal Products Post-authorisation evaluation of medicines for human use London, 12 March 2001 Doc. Ref: EMEA/H/5611/01/en EMEA PUBLIC STATEMENT ON LEFLUNOMIDE
Rivaroxaban shared care guidelines for the prevention of stroke and embolism in adult patients with nonvalvular atrial fibrillation.
 South West Essex Rivaroxaban Shared Care Guideline (SCG) Rivaroxaban shared care guidelines for the prevention of stroke and embolism in adult patients with nonvalvular atrial fibrillation. Introduction
South West Essex Rivaroxaban Shared Care Guideline (SCG) Rivaroxaban shared care guidelines for the prevention of stroke and embolism in adult patients with nonvalvular atrial fibrillation. Introduction
PACKAGE LEAFLET: INFORMATION FOR THE USER Omeprazol XXX 40 mg powder for solution for infusion omeprazole
 PACKAGE LEAFLET: INFORMATION FOR THE USER Omeprazol XXX 40 mg powder for solution for infusion omeprazole Read all of this leaflet carefully before you start using this medicine. - Keep this leaflet. You
PACKAGE LEAFLET: INFORMATION FOR THE USER Omeprazol XXX 40 mg powder for solution for infusion omeprazole Read all of this leaflet carefully before you start using this medicine. - Keep this leaflet. You
MEDICAL ASSISTANCE HANDBOOK PRIOR AUTHORIZATION OF PHARMACEUTICAL SERVICES. A. Prescriptions That Require Prior Authorization
 MEDICAL ASSISTANCE HBOOK I. Requirements for Prior Authorization of Anticoagulants A. Prescriptions That Require Prior Authorization Prescriptions for Anticoagulants which meet any of the following conditions
MEDICAL ASSISTANCE HBOOK I. Requirements for Prior Authorization of Anticoagulants A. Prescriptions That Require Prior Authorization Prescriptions for Anticoagulants which meet any of the following conditions
Improving Appropriate Use of Proton Pump Inhibitors as Gastrointestinal Prophylaxis in the Hospital Setting
 Improving Appropriate Use of Proton Pump Inhibitors as Gastrointestinal Prophylaxis in the Hospital Setting DATE Educating for Quality Improvement & Patient Safety 1 The Team Division CS&E Participants
Improving Appropriate Use of Proton Pump Inhibitors as Gastrointestinal Prophylaxis in the Hospital Setting DATE Educating for Quality Improvement & Patient Safety 1 The Team Division CS&E Participants
Digestive System (continued) Digestive System. Stomach. Peptic Ulcer Disease
 Digestive System Digestive System (continued) Responsible for breaking down food, absorbing nutrients, eliminating wastes Alimentary canal Also known as gastrointestinal tract Reaches from mouth to anus
Digestive System Digestive System (continued) Responsible for breaking down food, absorbing nutrients, eliminating wastes Alimentary canal Also known as gastrointestinal tract Reaches from mouth to anus
Medication Management Improvement System Protocol #4 Potentially Inappropriate Use of NSAIDs
 Medication Management Improvement System Protocol #4 Potentially Inappropriate Use of NSAIDs Problem: Use of Non-Steroidal Anti-Inflammatory Drugs (NSAIDs) in clients with any one of the following risk
Medication Management Improvement System Protocol #4 Potentially Inappropriate Use of NSAIDs Problem: Use of Non-Steroidal Anti-Inflammatory Drugs (NSAIDs) in clients with any one of the following risk
VOLTAROL Suppositories 12.5, 25, 50 and 100 mg (diclofenac sodium)
 VOLTAROL Suppositories 12.5, 25, 50 and 100 mg (diclofenac sodium) Patient Information Leaflet What you need to know about Voltarol Suppositories Your doctor has decided that you need this medicine to
VOLTAROL Suppositories 12.5, 25, 50 and 100 mg (diclofenac sodium) Patient Information Leaflet What you need to know about Voltarol Suppositories Your doctor has decided that you need this medicine to
NHS FORTH VALLEY Rivaroxaban for Stroke Prevention in Atrial Fibrillation
 NHS FORTH VALLEY Rivaroxaban for Stroke Prevention in Atrial Fibrillation Date of First Issue 06/06/2012 Approved 06/06/2012 Current Issue Date 29/10/2014 Review Date 29/10/2016 Version 1.4 EQIA Yes 01/06/2012
NHS FORTH VALLEY Rivaroxaban for Stroke Prevention in Atrial Fibrillation Date of First Issue 06/06/2012 Approved 06/06/2012 Current Issue Date 29/10/2014 Review Date 29/10/2016 Version 1.4 EQIA Yes 01/06/2012
Enoxaparin for long term anticoagulation in patients unsuitable for oral anticoagulants
 Enoxaparin for long term anticoagulation in patients unsuitable for oral anticoagulants Traffic light classification- Amber 2 Information sheet for Primary Care Prescribers Relevant Licensed Indications
Enoxaparin for long term anticoagulation in patients unsuitable for oral anticoagulants Traffic light classification- Amber 2 Information sheet for Primary Care Prescribers Relevant Licensed Indications
Medications for chronic pain
 Medications for chronic pain When it comes to treating chronic pain with medications, there are many to choose from. Different types of pain medications are used for different pain conditions. You may
Medications for chronic pain When it comes to treating chronic pain with medications, there are many to choose from. Different types of pain medications are used for different pain conditions. You may
It can therefore be bought without a prescription but should be used correctly to ensure its efficacy and to reduce side effects.
 140 mg Medicated plaster Diclofenac Sodium BEFORE USE, CAREFULLY READ ALL THE INFORMATION GIVEN ON THE INFORMATIVE LEAFLET This medicinal product is intended for SELF-MEDICATION. It can be used to treat
140 mg Medicated plaster Diclofenac Sodium BEFORE USE, CAREFULLY READ ALL THE INFORMATION GIVEN ON THE INFORMATIVE LEAFLET This medicinal product is intended for SELF-MEDICATION. It can be used to treat
Nonsteroidal. Drugs (NSAIDs) Anti-Inflammatory. North American Spine Society Public Education Series
 Nonsteroidal Anti-Inflammatory Drugs (NSAIDs) North American Spine Society Public Education Series Nonsteroidal Anti- Inflammatory Drugs Your healthcare provider has recommended that you take a nonsteroidal
Nonsteroidal Anti-Inflammatory Drugs (NSAIDs) North American Spine Society Public Education Series Nonsteroidal Anti- Inflammatory Drugs Your healthcare provider has recommended that you take a nonsteroidal
FOR THE PREVENTION OF ATRIAL FIBRILLATION RELATED STROKE
 www.bpac.org.nz keyword: warfarinaspirin FOR THE PREVENTION OF ATRIAL FIBRILLATION RELATED STROKE Key Concepts In atrial fibrillation (AF) warfarin is more effective than aspirin for stroke prevention.
www.bpac.org.nz keyword: warfarinaspirin FOR THE PREVENTION OF ATRIAL FIBRILLATION RELATED STROKE Key Concepts In atrial fibrillation (AF) warfarin is more effective than aspirin for stroke prevention.
Dorset Cardiac Centre
 P a g e 1 Dorset Cardiac Centre Patients with Atrial Fibrillation/Flutter undergoing DC Cardioversion or Ablation procedures- Guidelines for Novel Oral Anti-coagulants (NOACS) licensed for this use February
P a g e 1 Dorset Cardiac Centre Patients with Atrial Fibrillation/Flutter undergoing DC Cardioversion or Ablation procedures- Guidelines for Novel Oral Anti-coagulants (NOACS) licensed for this use February
CHAPTER 1 INTRODUCTION
 CHAPTER 1 INTRODUCTION Rheumatoid Arthritis (RA) is a chronic syndrome characterised by non-specific, usually symmetric inflammation of the peripheral joints, potentially resulting in progressive destruction
CHAPTER 1 INTRODUCTION Rheumatoid Arthritis (RA) is a chronic syndrome characterised by non-specific, usually symmetric inflammation of the peripheral joints, potentially resulting in progressive destruction
MEDICAL ASSISTANCE HANDBOOK PRIOR AUTHORIZATION OF PHARMACEUTICAL SERVICES. A. Prescriptions That Require Prior Authorization
 MEDICAL ASSISTANCE HBOOK PRI AUTHIZATION OF PHARMACEUTICAL SERVICES I. Requirements for Prior Authorization of Anticoagulants A. Prescriptions That Require Prior Authorization Prescriptions for Anticoagulants
MEDICAL ASSISTANCE HBOOK PRI AUTHIZATION OF PHARMACEUTICAL SERVICES I. Requirements for Prior Authorization of Anticoagulants A. Prescriptions That Require Prior Authorization Prescriptions for Anticoagulants
NHS FORTH VALLEY Guidelines for use of high dose Intravenous Esomeprazole in Adults (Previously called the Hong Kong Protocol)
 NHS FORTH VALLEY Guidelines for use of high dose Intravenous Esomeprazole in Adults (Previously called the Hong Kong Protocol) Date of First Issue 10/05/2010 Approved 16/06/2010 Current Issue Date 18/11/2015
NHS FORTH VALLEY Guidelines for use of high dose Intravenous Esomeprazole in Adults (Previously called the Hong Kong Protocol) Date of First Issue 10/05/2010 Approved 16/06/2010 Current Issue Date 18/11/2015
A study to Evaluate PPI s effect on vitamin D levels. Rani Hanna M.D., M.S. PGY-3 Joseph Grisanti, MD
 A study to Evaluate PPI s effect on vitamin D levels Rani Hanna M.D., M.S. PGY-3 Joseph Grisanti, MD The sunshine vitamin Existed over 500 million years. Prehormone, not only a vitamin. Two major sources:
A study to Evaluate PPI s effect on vitamin D levels Rani Hanna M.D., M.S. PGY-3 Joseph Grisanti, MD The sunshine vitamin Existed over 500 million years. Prehormone, not only a vitamin. Two major sources:
Stowe School Medications Policy
 INTRODUCTION Most pupils will need medication at some stage of their school life. Although this will mainly be for short periods there are a few pupils with chronic conditions who may require regular medication
INTRODUCTION Most pupils will need medication at some stage of their school life. Although this will mainly be for short periods there are a few pupils with chronic conditions who may require regular medication
Dorset Medicines Advisory Group
 Shared Care Guideline for prescribing rivaroxaban in the prevention of adverse outcomes after acute management of acute coronary syndrome in adults INDICATION In accordance with NICE TA335 rivaroxaban
Shared Care Guideline for prescribing rivaroxaban in the prevention of adverse outcomes after acute management of acute coronary syndrome in adults INDICATION In accordance with NICE TA335 rivaroxaban
Public Assessment Report. Pharmacy to General Sales List Reclassification. Nexium Control 20mg Gastro-Resistant Tablets.
 Public Assessment Report Pharmacy to General Sales List Reclassification Nexium Control 20mg Gastro-Resistant Tablets (Esomeprazole) EMA Agency number: Pfizer Consumer Healthcare Ltd TABLE OF CONTENTS
Public Assessment Report Pharmacy to General Sales List Reclassification Nexium Control 20mg Gastro-Resistant Tablets (Esomeprazole) EMA Agency number: Pfizer Consumer Healthcare Ltd TABLE OF CONTENTS
MEDICAL ASSISTANCE BULLETIN
 ISSUE DATE June 22, 2015 SUBJECT EFFECTIVE DATE MEDICAL ASSISTANCE BULLETIN NUMBER *See below BY Prior Authorization of Anticoagulants Pharmacy Service Leesa M. Allen, Deputy Secretary Office of Medical
ISSUE DATE June 22, 2015 SUBJECT EFFECTIVE DATE MEDICAL ASSISTANCE BULLETIN NUMBER *See below BY Prior Authorization of Anticoagulants Pharmacy Service Leesa M. Allen, Deputy Secretary Office of Medical
Rivaroxaban for the treatment of deep vein thrombosis and prevention of recurrent deep vein thrombosis and pulmonary embolism
 Implementation of NICE TA 261 Rivaroxaban for the treatment of deep vein thrombosis and prevention of recurrent deep vein thrombosis and pulmonary embolism Contents 1. Executive summary 2. Introduction
Implementation of NICE TA 261 Rivaroxaban for the treatment of deep vein thrombosis and prevention of recurrent deep vein thrombosis and pulmonary embolism Contents 1. Executive summary 2. Introduction
NIMULID MD. 1. Introduction. 2. Nimulid MD Drug delivery system
 NIMULID MD 1. Introduction Nimulid MD is a flavoured dispersible Nimesulide tablet with fast mouth dissolving characteristics thereby providing immediate relief. Nimesulide is a non-steroidal antiinflammatory
NIMULID MD 1. Introduction Nimulid MD is a flavoured dispersible Nimesulide tablet with fast mouth dissolving characteristics thereby providing immediate relief. Nimesulide is a non-steroidal antiinflammatory
Antiplatelet and Antithrombotic Therapy. Dr Curry Grant Stroke Prevention Clinic Quinte Health Care
 Antiplatelet and Antithrombotic Therapy Dr Curry Grant Stroke Prevention Clinic Quinte Health Care Disclosure of Potential for Conflict of Interest Dr. F.C. Grant Atrial Fibrillation FINANCIAL DISCLOSURE:
Antiplatelet and Antithrombotic Therapy Dr Curry Grant Stroke Prevention Clinic Quinte Health Care Disclosure of Potential for Conflict of Interest Dr. F.C. Grant Atrial Fibrillation FINANCIAL DISCLOSURE:
Biologic Treatments for Rheumatoid Arthritis
 Biologic Treatments Rheumatoid Arthritis (also known as cytokine inhibitors, TNF inhibitors, IL 1 inhibitor, or Biologic Response Modifiers) Description Biologics are new class of drugs that have been
Biologic Treatments Rheumatoid Arthritis (also known as cytokine inhibitors, TNF inhibitors, IL 1 inhibitor, or Biologic Response Modifiers) Description Biologics are new class of drugs that have been
NEUROTONE THR 00904/0005 UKPAR
 NEUROTONE THR 00904/0005 UKPAR TABLE OF CONTENTS Lay summary Page 2 Scientific discussion Page 3 Steps taken for assessment Page 10 Summary of Product Characteristics Page 11 Product Information Leaflet
NEUROTONE THR 00904/0005 UKPAR TABLE OF CONTENTS Lay summary Page 2 Scientific discussion Page 3 Steps taken for assessment Page 10 Summary of Product Characteristics Page 11 Product Information Leaflet
NHS FORTH VALLEY Rivaroxaban for Stroke Prevention in Atrial Fibrillation
 NHS FORTH VALLEY Rivaroxaban for Stroke Prevention in Atrial Fibrillation Date of First Issue 06/06/2012 Approved 06/06/2012 Current Issue Date 06/06/2012 Review Date 06/06/2014 Version 1.1 EQIA Yes /
NHS FORTH VALLEY Rivaroxaban for Stroke Prevention in Atrial Fibrillation Date of First Issue 06/06/2012 Approved 06/06/2012 Current Issue Date 06/06/2012 Review Date 06/06/2014 Version 1.1 EQIA Yes /
Using big data to identify opportunity and drive informed change in secondary care
 Using big data to identify opportunity and drive informed change in secondary care Exploring the changing patterns of Non-Steroidal Anti-Inflammatory Drug dispensing in UK hospitals ABOUT IMS HEALTH IMS
Using big data to identify opportunity and drive informed change in secondary care Exploring the changing patterns of Non-Steroidal Anti-Inflammatory Drug dispensing in UK hospitals ABOUT IMS HEALTH IMS
DERBYSHIRE JOINT AREA PRESCRIBING COMMITTEE (JAPC) MANAGEMENT of Atrial Fibrillation (AF)
 DERBYSHIRE JOINT AREA PRESCRIBING COMMITTEE (JAPC) MANAGEMENT of Atrial Fibrillation (AF) Key priorities Identification and diagnosis Treatment for persistent AF Treatment for permanent AF Antithrombotic
DERBYSHIRE JOINT AREA PRESCRIBING COMMITTEE (JAPC) MANAGEMENT of Atrial Fibrillation (AF) Key priorities Identification and diagnosis Treatment for persistent AF Treatment for permanent AF Antithrombotic
Duration of Dual Antiplatelet Therapy After Coronary Stenting
 Duration of Dual Antiplatelet Therapy After Coronary Stenting C. DEAN KATSAMAKIS, DO, FACC, FSCAI INTERVENTIONAL CARDIOLOGIST ADVOCATE LUTHERAN GENERAL HOSPITAL INTRODUCTION Coronary artery stents are
Duration of Dual Antiplatelet Therapy After Coronary Stenting C. DEAN KATSAMAKIS, DO, FACC, FSCAI INTERVENTIONAL CARDIOLOGIST ADVOCATE LUTHERAN GENERAL HOSPITAL INTRODUCTION Coronary artery stents are
Guidelines for Use of Clopidogrel (Plavix )
 East Lancashire Medicines Management Board representing East Lancashire Hospitals NHS Trust, Lancashire Care Trust, Blackburn with Darwen PCT, East Lancs PCT Licensed Indications Guidelines for Use of
East Lancashire Medicines Management Board representing East Lancashire Hospitals NHS Trust, Lancashire Care Trust, Blackburn with Darwen PCT, East Lancs PCT Licensed Indications Guidelines for Use of
How To Choose A Biologic Drug
 North Carolina Rheumatology Association Position Statements I. Biologic Agents A. Appropriate delivery, handling, storage and administration of biologic agents B. Indications for biologic agents II. III.
North Carolina Rheumatology Association Position Statements I. Biologic Agents A. Appropriate delivery, handling, storage and administration of biologic agents B. Indications for biologic agents II. III.
New Oral Anticoagulants
 New Oral Anticoagulants Tracy Minichiello, MD Associate Professor of Medicine Chief, San FranciscoVA Anticoagulation and Thrombosis Service Ansell, J. Hematology Copyright 2010 American Society of Hematology.
New Oral Anticoagulants Tracy Minichiello, MD Associate Professor of Medicine Chief, San FranciscoVA Anticoagulation and Thrombosis Service Ansell, J. Hematology Copyright 2010 American Society of Hematology.
The Role of the Newer Anticoagulants
 The Role of the Newer Anticoagulants WARFARIN = Coumadin DAGIBATRAN = Pradaxa RIVAROXABAN = Xarelto APIXABAN = Eliquis INDICATION DABIGATRAN (Pradaxa) RIVAROXABAN (Xarelto) APIXABAN (Eliquis) Stroke prevention
The Role of the Newer Anticoagulants WARFARIN = Coumadin DAGIBATRAN = Pradaxa RIVAROXABAN = Xarelto APIXABAN = Eliquis INDICATION DABIGATRAN (Pradaxa) RIVAROXABAN (Xarelto) APIXABAN (Eliquis) Stroke prevention
Rivaroxaban: Prescribing Guidance for the treatment of provoked venous thromboembolism (VTE)
 Rivaroxaban: Prescribing Guidance for the treatment of provoked venous thromboembolism (VTE) Amber Drug Level 2 Leeds We have started your patient on rivaroxaban for the treatment of provoked VTE (deep
Rivaroxaban: Prescribing Guidance for the treatment of provoked venous thromboembolism (VTE) Amber Drug Level 2 Leeds We have started your patient on rivaroxaban for the treatment of provoked VTE (deep
Painkillers (analgesics)
 Drug information (analgesics) This leaflet provides information on painkillers and will answer any questions you have about the treatment. Arthritis Research UK produce and print our booklets entirely
Drug information (analgesics) This leaflet provides information on painkillers and will answer any questions you have about the treatment. Arthritis Research UK produce and print our booklets entirely
Survey conducted into patient experience of life with osteoarthritis. A collaboration between Pro Bono Bio, Arthritis Research UK, and LloydsPharmacy
 Survey conducted into patient experience of life with osteoarthritis. A collaboration between Pro Bono Bio, Arthritis Research UK, and LloydsPharmacy Executive summary Over 400 people with joint conditions
Survey conducted into patient experience of life with osteoarthritis. A collaboration between Pro Bono Bio, Arthritis Research UK, and LloydsPharmacy Executive summary Over 400 people with joint conditions
SUMMARY OF THE RISK MANAGEMENT PLAN (by medicinal product)
 PART VI SUMMARY OF THE RISK MANAGEMENT PLAN (by medicinal product) Format and content of the summary of the RMP The summary of the RMP part VI contains information based on RMP modules SI, SVIII and RMP
PART VI SUMMARY OF THE RISK MANAGEMENT PLAN (by medicinal product) Format and content of the summary of the RMP The summary of the RMP part VI contains information based on RMP modules SI, SVIII and RMP
Safety Information Card for Xarelto Patients
 Safety Information Card for Xarelto Patients 15mg Simply Protecting More Patients 20mg Simply Protecting More Patients Keep this card with you at all times Present this card to every physician or dentist
Safety Information Card for Xarelto Patients 15mg Simply Protecting More Patients 20mg Simply Protecting More Patients Keep this card with you at all times Present this card to every physician or dentist
The monthly newsletter from the MHRA and its independent advisor the Commission on Human Medicines
 Latest advice for medicines users The monthly newsletter from the MHRA and its independent advisor the Commission on Human Medicines Volume 6, Issue 9, April 2013 Drug safety advice Hot topic Stop press
Latest advice for medicines users The monthly newsletter from the MHRA and its independent advisor the Commission on Human Medicines Volume 6, Issue 9, April 2013 Drug safety advice Hot topic Stop press
TA 256: Rivaroxaban for the prevention of stroke and systemic embolism in people with atrial fibrillation
 Service Notification in response to DHSSPS endorsed NICE Technology Appraisals TA 256: Rivaroxaban for the prevention of stroke and systemic embolism in people with atrial fibrillation 1 Name of Commissioning
Service Notification in response to DHSSPS endorsed NICE Technology Appraisals TA 256: Rivaroxaban for the prevention of stroke and systemic embolism in people with atrial fibrillation 1 Name of Commissioning
NSAIDs and Peptic Ulcers
 NSAIDs and Peptic Ulcers National Digestive Diseases Information Clearinghouse U.S. Department of Health and Human Services NATIONAL INSTITUTES OF HEALTH What is a peptic ulcer? A peptic ulcer is a sore
NSAIDs and Peptic Ulcers National Digestive Diseases Information Clearinghouse U.S. Department of Health and Human Services NATIONAL INSTITUTES OF HEALTH What is a peptic ulcer? A peptic ulcer is a sore
STROKE PREVENTION IN ATRIAL FIBRILLATION. TARGET AUDIENCE: All Canadian health care professionals. OBJECTIVE: ABBREVIATIONS: BACKGROUND:
 STROKE PREVENTION IN ATRIAL FIBRILLATION TARGET AUDIENCE: All Canadian health care professionals. OBJECTIVE: To guide clinicians in the selection of antithrombotic therapy for the secondary prevention
STROKE PREVENTION IN ATRIAL FIBRILLATION TARGET AUDIENCE: All Canadian health care professionals. OBJECTIVE: To guide clinicians in the selection of antithrombotic therapy for the secondary prevention
NOAC Prescribing in Patients with Non-Valvular Atrial Fibrillation: Frequently Asked Questions
 AC Prescribing in Patients with Non-Valvular Atrial Fibrillation: Frequently Asked Questions FAQ document jointly prepared by NHSGGC Haematology Service & Medicines Infmation On behalf of the Heart MCN
AC Prescribing in Patients with Non-Valvular Atrial Fibrillation: Frequently Asked Questions FAQ document jointly prepared by NHSGGC Haematology Service & Medicines Infmation On behalf of the Heart MCN
Update on Antiplatelets and anticoagulants. Outlines. Antiplatelets and Anticoagulants 1/23/2013. Timir Paul, MD, PhD
 Update on Antiplatelets and anticoagulants Timir Paul, MD, PhD Antiplatelets Indications Doses Long term use (beyond 12 months) ASA and combination use of NSAIDS ASA resistance Plavix resistance Plavix
Update on Antiplatelets and anticoagulants Timir Paul, MD, PhD Antiplatelets Indications Doses Long term use (beyond 12 months) ASA and combination use of NSAIDS ASA resistance Plavix resistance Plavix
The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION. 10 March 2010
 The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 10 March 2010 ARIXTRA 1.5 mg/0.3 ml, solution for injection in pre-filled syringe Box of 2 (CIP: 363 500-6) Box of
The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 10 March 2010 ARIXTRA 1.5 mg/0.3 ml, solution for injection in pre-filled syringe Box of 2 (CIP: 363 500-6) Box of
DUAL ANTIPLATELET THERAPY. Dr Robert S Mvungi, MD(Dar), Mmed (Wits) FCP(SA), Cert.Cardio(SA) Phy Tanzania Cardiac Society Dar es Salaam Tanzania
 DUAL ANTIPLATELET THERAPY Dr Robert S Mvungi, MD(Dar), Mmed (Wits) FCP(SA), Cert.Cardio(SA) Phy Tanzania Cardiac Society Dar es Salaam Tanzania DUAL ANTIPLATELET THERAPY (DAPT) Dual antiplatelet regimen
DUAL ANTIPLATELET THERAPY Dr Robert S Mvungi, MD(Dar), Mmed (Wits) FCP(SA), Cert.Cardio(SA) Phy Tanzania Cardiac Society Dar es Salaam Tanzania DUAL ANTIPLATELET THERAPY (DAPT) Dual antiplatelet regimen
Inhibit terminal acid secretion from parietal cells by blocking H + /K + - ATPase pump
 Chris J. Taylor, Pharm.D., BCPS Clinical Pharmacist Phoenix VA Health Care System Review pharmacology of PPIs Discuss possible association between PPI use and development of the following: Pneumonia (community-acquired
Chris J. Taylor, Pharm.D., BCPS Clinical Pharmacist Phoenix VA Health Care System Review pharmacology of PPIs Discuss possible association between PPI use and development of the following: Pneumonia (community-acquired
Cyclooxygenase-2 selective non-steroidal anti-inflammatory drugs (etodolac, meloxicam, celecoxib, rofecoxib, etoricoxib, valdecoxib and
 Health Technology Assessment 2008; Vol. 12: No. 11 Cyclooxygenase-2 selective non-steroidal anti-inflammatory drugs (etodolac, meloxicam, celecoxib, rofecoxib, etoricoxib, valdecoxib and lumiracoxib) for
Health Technology Assessment 2008; Vol. 12: No. 11 Cyclooxygenase-2 selective non-steroidal anti-inflammatory drugs (etodolac, meloxicam, celecoxib, rofecoxib, etoricoxib, valdecoxib and lumiracoxib) for
All Wales Risk/Benefit Assessment Tool for Oral Anticoagulant Treatment in People with Atrial Fibrillation
 All Wales Risk/Benefit Assessment Tool for Oral Anticoagulant Treatment in People with Atrial Fibrillation October 2013 This report has been prepared by a multiprofessional collaborative group, with support
All Wales Risk/Benefit Assessment Tool for Oral Anticoagulant Treatment in People with Atrial Fibrillation October 2013 This report has been prepared by a multiprofessional collaborative group, with support
Learn More About Product Labeling
 Learn More About Product Labeling Product label The product label is developed during the formal process of review and approval by regulatory agencies of any medicine or medical product. There are specific
Learn More About Product Labeling Product label The product label is developed during the formal process of review and approval by regulatory agencies of any medicine or medical product. There are specific
