What s New in 2016? Prakash Krishnan MD FACC Director of Endovascular Services Cardiac Catheterization Laboratory Mount Sinai Medical Center New York
|
|
|
- Bertram Parsons
- 7 years ago
- Views:
Transcription
1 What s New in 2016? Prakash Krishnan MD FACC Director of Endovascular Services Cardiac Catheterization Laboratory Mount Sinai Medical Center New York
2 Topics!! New Devices Data Algorithm based on data Question that need to be answered!
3 Where do we start! Fem-Pop segment- Patency in real world cohorts- Long lesion,cto Randomized data ISR Ca
4 Where do we start! BTK- We need to define endpoints as a whole Perform studies that are relevant to our patients We still have a long way to go!
5 Problems Lesion length CTO ISR Calcium
6 Answers Lesion length- DEB/DES Randomized Trials Zilver PTX InPact Levant II
7 Baseline Lesion Characteristics PTA Zilver PTX p-value Lesions Normal-to-normal lesion length (mm) 63 ± ± Stenosed lesion length (mm) 1,2 53 ± ± Diameter stenosis (%) 1 78 ± ± Total occlusions 27% 33% 0.20 De novo lesions 94% 95% 0.68 Lesion calcification 1 None 5% 2% 1 Angiographic core lab assessment 2 Region with > 20% diameter stenosis * Statistically significant Little 38% 26% Moderate 22% 35% Severe 35% 37% < 0.01*
8 5-year Primary Patency (PSVR < 2.0) Zilver PTX vs. Standard Care 66.4% Zilver PTX p < 0.01 log-rank 43.4% Optimal PTA + BMS At 5 years, Zilver PTX demonstrates a 41% reduction in restenosis compared to standard care
9 5-year Primary Patency (PSVR < 2.0) Provisional Zilver PTX vs. BMS 72.4% Provisional Zilver PTX p = 0.03 log-rank 53.0% Provisional BMS At 5 years, Zilver PTX demonstrates a 41% reduction in restenosis compared to BMS
10 5-year Freedom from TLR Zilver PTX vs. Standard Care 83.1% 67.6% Zilver PTX Optimal PTA + BMS p < 0.01 log-rank At 5 years, Zilver PTX demonstrates a 48% reduction in reintervention compared to standard care
11 Drug-Coated Balloon
12 SC A Current DCB Landscape 12-Month Patency 24-Month Patency 83.7% 89.9% 67.0% 73.5% 72.4% 55% 53.7% LEVANT I Lutonix TM* LEVANT 2 Lutonix TM* Italian Registry IN.PACTTM* IN.PACT SFA IN.PACT TM*. Scheinert, D., et al., J Am Coll Cardiol Inv, 2014;7: p Micari, A., et al., J Am Coll Cardiol Inv, 2012;5: p Tepe G. Presentation, Charing Cross London. US Dept of HHS FDA Executive Summary: Circulatory System Devices Advisory Panel June 12,2014: Bard Lutonix 035 Drug Coated Balloon PTA Catheter.
13 No Class Effect With DCBs Noticeable difference (15.8%) in patency rates and effect size between LEVANT 2 and IN.PACT SFA 12-Month Patency 89.8% LEVANT 2 IN.PACT SFA Num. of lesions Mean Length (mm) % 17.2% 56.8% 23% 66.8% Baseline stenosis 80.5% 81% Mean RVD Severe Calcification 10.4% 8.1% Occlusions 20.6% 25.8% De novo 83.9% 95% LEVANT 2 Lutonix 2 µg/mm2 TM* LEVANT 2 Unocated PTA Arm IN.PACT SFA IN.PACT 3 µg/mm2 TM* IN.PACT SFA Uncoated PTA Arm 1. US, Dept HHS. FDA Executive Summary: Circulatory System Devices Advisory Panel June 12,2014: Bard Lutonix 035 Drug Coated Balloon PTA Catheter. 2. Tepe G. Presentation. Charing Cross 2014, London, England. April SC A
14 1 IN.PACT SFA TRIAL EFFICACY OUTCOMES THROUGH 2 YEARS SUSTAINED PERFORMANCE BENEFIT OF IN.PACT ADMIRAL DCB OVER PTA THROUGH TWO YEARS 2 1. Freedom from core laboratory-assessed restenosis (duplex ultrasound PSVR 2.4) or clinically-driven target lesion revascularization through 24 months (adjudicated by a Clinical Events Committee blinded to the assigned treatment). 2. Number at risk represents the number of evaluable subjects at the beginning of the 30-day window prior to each follow-up interval. 14
15 1 IN.PACT SFA TRIAL EFFICACY OUTCOMES THROUGH 2 YEARS SUSTAINED PERFORMANCE BENEFIT OF IN.PACT ADMIRAL DCB OVER PTA THROUGH TWO YEARS 2 1. Freedom from core laboratory-assessed restenosis (duplex ultrasound PSVR 2.4) or clinically-driven target lesion revascularization through 24 months (adjudicated by a Clinical Events Committee blinded to the assigned treatment). 2. Number at risk represents the number of evaluable subjects at the beginning of the 30-day window prior to each follow-up interval.
16 IN.PACT SFA TRIAL ADDITIONAL EFFICACY OUTCOMES THROUGH 2 YEARS SUSTAINED PERFORMANCE BENEFIT OF IN.PACT ADMIRAL DCB OVER PTA THROUGH TWO YEARS 2-Year Outcome IN.PACT Admiral DCB (n = 220) PTA (n = 111) P-value 1 Clinically-driven TLR 2 9.1% (18/198) 28.3% (30/106) <0.001 All TLR % (20/198) 29.2% (31/106) <0.001 Primary Sustained Clinical Improvement % (133/173) 59.2% (61/103) ABI / TBI ± ± Unless otherwise indicated, all tests were for superiority using the Fisher s exact test for binary variables and t-test for continuous variables. 2.Clinically-driven TLR adjudicated by an independent Clinical Event Committee, blinded to the assigned treatment based on any re-intervention at the target lesion due to symptoms or drop of ABI of 20% or >0.15 when compared to post-procedure baseline ABI. 3.All TLR includes clinically-driven and incidental or duplex driven TLR. 4.Freedom from target limb amputation, target vessel revascularization (TVR), and increase in Rutherford class. 5.TBI allowed in cases of incompressible vessels in IN.PACT SFA II phase. 16
17 What about real world lesions? No randomized data!! Zilver PTx Japanese post market study InPact/Lutonix Real world registry
18 Japan PMS Compared to RCT and SAS Zilver PTX RCT Zilver PTX SAS Zilver PTX Japan PMS No significant untreated inflow tract stenosis Key Sudy Criteria At least one patent runoof vessel Maximum 2 Zilver PTX stents Maximum 4 Zilver PTX stents per lesion per patient Lesion length < 14cm No exclusions One lesion per limb No Prior stent in SFA In-stent restenosis included All Patients treated with Zilver PTX enrolled (up to enrollment limit), NO exclusion criteria Excluded if serum creatinine > 2.0, renal failure, or dialysis No exclusions Antiplatelets Clopidogrel or ticlopidine recommended for 60 days, aspirin indefinitely Follow-up 5 Years 2 Years 5 Years Patency Core laboratory analysis Site analysis Stent Integrity X-ray core laboratory analysis Increasingly complex patients and lesions
19 Baseline Lesion Characteristics Zilver PTX RCT Zilver PTX SAS Zilver PTX Japan PMS Lesions Lesion length (cm) * 6.6 ± ± ± 9.7 Diameter stenosis (%) 80 ± ± ± 11 Total occlusions 30% 38% 42% In-stent restenosis (ISR) ** 0% 15% 19% Patent runoff vessels * Zilver PTX is indicated for lesions up to 140mm per leg and 280mm per patient ** Zilver PTX is not indicated for the treatment of in-stent restensosis 0 0% 0% 7% 1 22% 19% 32% 2 35% 35% 32% 3 42% 45% 29%
20 Freedom from TLR Months Freedom from TLR Zilver PTX RCT Zilver PTX SAS Zilver PTX Japan PMS 6 months 98.0% 97.6% 97.3% 1 year 91.6% 89.5% 91.4% 5 years 83.1% - TBD
21 Primary Patency by Duplex Ultrasonography Months Primary Patency by Duplex Ultrasonography Zilver PTX RCT (PSVR 2.0) Zilver PTX SAS (PSVR 2.0) Japan PMS (PSVR 2.4) 6 months 95.6% 96.3% 96.7% 1 year 90.2% 82.8% 84.8% 2 years 83.4% - TBD 5 years 72.4% - TBD
22 High stent integrity Fracture Rates Years Zilver PTX RCT (457 stents) Zilver PTX SAS (1889 stents) Japan PMS (1066 stents) 1 0.9% 1.5% 1.6% 3 1.9% - TBD 5 1.9% - TBD
23 Eluvia Drug-Eluting Vascular Stent System Design Goals Eluvia Drug-Eluting Vascular Stent System The Eluvia Drug-Eluting Vascular Stent System Design Goals: Ptx drug in a biostable polymer matrix loaded on to the Innova stent system Drug elution release of 85% at 1 year to match restenotic cascade Size matrix to accommodate various lesions Utilize a stent platform and triaxial delivery system BSC Data on File. Innova Vascular Stent System is an investigational device. Innova and Eluvia devices are not for sale in the U.S.
24 Drug Coated Balloon
25 Lesion Length: One of Many Challenges Unmet need in complex lesion types including long lesions, chronic total occlusions and in-stent restenosis No current treatment standard identified. Longer lesion length is a predictor of lower patency at 12 months post-procedure (35-65%) when associated with gold standard therapies of PTA and stenting subjects enrolled in the IN.PACT Global Study Long Lesion cohort intended to characterize performance of IN.PACT Admiral DCB in these types of long, complex lesions Clinical Cohort Total enrolment: 1,538 subjects, including >100 pts treated with 150 mm device Three Imaging Cohorts >450 subjects De NOVO ISR subcohort >150 subjects Long Lesion subcohort 157 subjects CTO sub-cohort >150 subjects 1. Lammer J et al. J Am Coll Cardiol 2013;62: Bosiers M et al. J Vasc Surg 2011;54:
26 IN.PACT Global Study Long Lesion Imaging Cohort: Baseline Clinical Characteristics N=157 Subjects Age (Y±SD) 69.5 ± 10.7 Male Gender (%) 66.2% (104/157) Diabetes (%) 41.0% (64/156) Insulin Dependent Diabetes Mellitus (%) 21.8% (34/156) Hypertension (%) 87.9% (138/157) Hyperlipidemia (%) 76.7% (115/150) Current Smoker (%) 34.4% (54/157) Obesity (BMI 30 kg/m²) (%) 22.1% (34/154) Coronary Heart Disease (%) 52.3% (80/153) Carotid Artery Disease (%) 22.4% (30/134) Renal Insufficiency 1 (%) 14.4% (21/146) Previous Peripheral Revasc. (%) 55.4% (87/157) Concomitant BTK Disease (%) 47.5% (67/141) ABI (mean ± SD) ± (147) Rutherford Clinical Category RCC4 10.3% RCC5 6.4% RCC3 61.5% RCC2 21.8% 1. Baseline serum creatinine 1.5 mg/dl Dierk Scheinert, MD, Drug coated balloon treatment for patients with intermittent claudication: new insights from the IN.PACT Global Study long lesion (>15cm) imaging cohort, europcr 2015
27 IN.PACT Global Study Long Lesion Imaging Cohort: Lesion and Procedural Characteristics Lesions (N) 164 Lesion Type: - de novo - restenotic (no ISR) - ISR Lesion Length (mean ±SD) 83.2% (134/161) 16.8% (27/161) 0.0% (0/161) ± 8.61 cm Total Occlusions 60.4% (99/164) Calcification Severe 71.8% (117/163) 19.6% (32/163) RVD (mm ±SD) ± Pre-treatment Diameter Stenosis (% ±SD) 90.9% ± 14.2 Device Success % (442/444) Procedure Success % (155/156) Clinical Success % (155/156) Pre-dilatation 89.8% (141/157) Post-dilatation 39.1% (61/156) Provisional Stent - LL cm: - LL > 25 cm: 40.4% (63/156) 33.3% (33/99) 52.6% (30/57) 1. Device success: successful delivery, inflation, deflation and retrieval of the intact study balloon device without burst below the RBP 2. Procedure success: residual stenosis of 50% (non-stented subjects) or 30% (stented subjects) by core lab (if core lab was not available then the site reported estimate was used) 3. Clinical success: procedural success without procedural complications (death, major target limb amputation, thrombosis of the target lesion, or TVR) prior to discharge Dissections: % (61/161) A-C 47.2% (76/161) D-F 14.9% (24/161) Dierk Scheinert, MD, Drug coated balloon treatment for patients with intermittent claudication: new insights from the IN.PACT Global Study long lesion (>15cm) imaging cohort, europcr 2015
28 IN.PACT Global Study Long Lesion Imaging Cohort: Kaplan-Meier Estimate of Primary Patency Number of subjects at risk* Baseline (day 0) 6 mo 12 mo IN.PACT Admiral DCB
29 IN.PACT Global Study Long Lesion Imaging Cohort: Primary Patency in Non-stented Subgroup Number of subjects at risk* Baseline (day 0) 6 mo 12 mo Non-Stented Pure LL Imaging Cohort
30 IN.PACT Global Study Long Lesion Imaging Cohort: Primary Patency by Lesion Length Subgroup Number of subjects at risk Baseline (day 0) 6 mo 12 mo Lesion Length cm Lesion Length >25 cm
31 IN.PACT Global Study Long Lesion Imaging Cohort: Safety & Effectiveness Outcomes by Lesion Length Subgroup LL cm (N=100) LL > 25 cm (N=57) Clinically-Driven TLR 1 2.3% (2/87) 12.8% (6/47) Primary Safety Endpoint % (85/87) 87.2% (41/47) Major Adverse Events 3 8.0% (7/87) 19.1% (9/47) Death (all-cause) 3.4% (3/87) 6.4% (3/47) Major Target Limb Amputation 0.0% (0/87) 0.0% (0/47) Thrombosis 2.3% (2/87) 6.4% (3/47) Any TLR 2.3% (2/87) 12.8% (6/47) Any TVR 2.3% (2/87) 12.8% (6/47) 1. Any re-intervention within the target lesion(s) due to symptoms or drop of ABI of 20% or > 0.15 when compared to post-index procedure baseline ABI 2. Composite of 30-day freedom from device- and procedure-related mortality and 12-month freedom from major target limb amputation and clinically-driven TVR 3. Major Adverse Events: Composite of death, major target limb amputation, clinically-driven TVR, and thrombosis Dierk Scheinert, MD, Drug coated balloon treatment for patients with intermittent claudication: new insights from the IN.PACT Global Study long lesion (>15cm) imaging cohort, europcr 2015
32 Results Across IN.PACT Clinical Studies Consistent clinical outcomes with the IN.PACT Admiral DCB across studies and complex femoropopliteal lesions. IN.PACT SFA Trial (DCB Arm) IN.PACT Global Study Long Lesion Cohort IN.PACT Global Study N=220 N=157 N=655 Lesion Length (cm) CD-TLR 2.4% 6.0% 8.7% CD-TVR 4.3% 6.0% 9.5% Thrombosis 1.4% 3.7% 3.8% Target Limb Major Amputation 0.0% 0.0% 0.3% Dierk Scheinert, MD, Drug coated balloon treatment for patients with intermittent claudication: new insights from the IN.PACT Global Study long lesion (>15cm) imaging cohort, europcr 2015
33 Problems Lesion length CTO ISR Calcium
34 Lesion Length: One of Many Challenges Unmet need in complex lesion types including long lesions, chronic total occlusions and in-stent restenosis No current treatment standard identified. Longer lesion length is a predictor of lower patency at 12 months post-procedure (35-65%) when associated with gold standard therapies of PTA and stenting subjects enrolled in the IN.PACT Global Study Long Lesion cohort intended to characterize performance of IN.PACT Admiral DCB in these types of long, complex lesions Clinical Cohort Total enrolment: 1,538 subjects, including >100 pts treated with 150 mm device Three Imaging Cohorts >450 subjects De NOVO ISR subcohort >150 subjects Long Lesion subcohort 157 subjects CTO sub-cohort >150 subjects 1. Lammer J et al. J Am Coll Cardiol 2013;62: Bosiers M et al. J Vasc Surg 2011;54:
35 Primary Patency at 1 Year ISR SUBGROUP ISR SUBGROUP 1 YEAR PRIMARY PATENCY CD-TLR AVERAGE LESION LENGTH 131 de novo ISR Lesions 88.7% 7.3% 17.2±10.5cm 35 UC IE 2015 Medtronic. All Rights Reserved. Medtronic, Medtronic logo and Further. Together are trademarks of Medtronic. Not for distribution in France, or Japan. 10/15
36 Design EXCITE ISR Trial 250 patients enrolled between June 2011 and February 2014 in 40 clinical sites in United States DESIGN: Prospective, randomized, multi-center clinical evaluation of excimer laser atherectomy (ELA) OBJECTIVE: To evaluate safety and efficacy of ELA with adjunctive PTA (ELA+PTA) versus PTA alone for treating femoropopliteal in-stent restenosis PRINCIPAL INVESTIGATORS Eric J Dippel, MD Craig Walker, MD 250 lesions crossable by guidewire 169 ELA + PTA Primary Safety endpoint at 37 days (n=155) 81 PTA 7 lesions uncrossable Primary Safety endpoint at 37 days (n=73) Primary Efficacy endpoint at 212 days (n=117) Primary Efficacy endpoint at 212 days (n=56)
37 Excimer Laser Atherectomy Catheters Turbo Elite Pilot channel creation Turbo Tandem Biased laser catheter for large lumen ablation
38 Survival Probability Freedom from TLR Product-Limit Survival Estimates With number of subjects at risk p < Days from Index Procedure
39 Survival Probability Primary Patency Product-Limit Survival Estimates With number of subjects at risk p < Days from Index Procedure
40 Data is upcoming!!! DEB/DES-CTO
41 Problems Lesion length CTO ISR Calcium
42 DCB and Calcium IN.PACT DCB and Calcium Prospective Study (n=60) 12 month Results 100% 100% 100% 100% 90% 90% % % % % 50% % Primary Patency LLL % 0.0 1a 1b 2a 2b 3a 3b 4a 4b Calcium distribution and severity affect LLL and primary patency Ca ++ represents a barrier to optimal drug absorption Calcium distribution evaluation by CTA (circumf.) and DSA (longitud.) F. Fanelli Cardiovasc Interv Radiol 2014
43 Levant 2 Trial Angiographic Characteristics (ITT) D. Scheinert LINC 2015
44 Levant 2 Trial Sub-analysis Calcified Lesions Kaplan Meier analysis, 38 events 67 at risk D. Scheinert LINC 2015
45 Correct choice of stent design can minimize struts stress while maintaining a patent lumen High circumferential stress values at the internal elastic lamina are most likely to disrupt this structure, provoking neintimal growth leading to restenosis
46 SUPERA stent Interwoven self-expanding nitinol stent Six pairs of super-elastic nitinol wires which are interwoven in a helical pattern in a closed cell geometry 1) Radial strength 2) Flexibility and Durability 3) Conformability 4) Kink, Crush and Fracture resistant Supports the natural movement of the vessel
47 SUPERB Results Primary Patency (VIVA) Primary Patency (K-M) Freedom from TLR (K-M) 79.0% 86.3% 89% 84% 83% 12 Months 24 Months 36 Months Patient Baseline 264 Ave. Lesion Length (cm) 7.8 Fracture Rate (%) 0* % Distal SFA/PPA 34 Occlusions (%) 25 Severe Calcium (%) 45 Rutherford 1-3 (%) 95 Rutherford 4 (%) 5 PSVR 2.0
48 SUPERB Results in Long Lesions Consistent results across lesion lengths Non-Restenosis Rate by Lesion Length (12 months SUPERB IDE TRIAL) PSVR 2.0
49 Freedom from TLR (K-M) by Percent Compression / Elongation at 12, 24, and 36 months Optimal Deployment Leads to Low Re-intervention Rate Out to 3 Years SUPERB freedom from TLR at 1, 2, and 3 years 100% 90% 80% 90% 90% 90% 91% 87% 87% 97% 96% 94% 84% 78% 87% 82% 78% 78% 77% 12 months 24 months 36 months 70% 60% 63% 50% 40% 42% 30% 20% 10% 0% Moderate (21-40%) Minimal (11-20%) Nominal (± 10%) Minimal (11-20%) Moderate (21-40%) Severe (>40%) n=6 n=22 n=74 n=38 n=39 n=26 Compressed Nominal Elongated
50 Comparative Data Between DCB and DES Therapies 89.8% Primary Patency (KM Day 360) 73.5% 82.7% CD-TLR (12 month) 12.3% 9.6% 2.4% IN.PACT SFA LEVANT II Zilver PTX IN.PACT SFA LEVANT II* Zilver PTX Note: Primary patency and TLR rates are not directly comparable; chart is for illustration only. IN.PACT SFA/LEVANT II represent DCB arms of trial; ZILVER PTX represents DES arm of trial. * Data from LEVANT II represents total TLR.
51 Comparative Data Between DCB and DES Therapies 12-month Binary Restenosis 36.0% 16.5% 23.5% IN.PACT SFA (DCB) LEVANT II (DCB) Zilver PTX PSVR >2.4 PSVR 2.5 PSVR >2.0 Source: FDA SSED Table 16 Source: FDA Exec Summary Table 37 Source: FDA SSED Table 25 Note: Binary restenosis rates are not directly comparable; chart is for illustration only; IN.PACT SFA and LEVANT II binary restenosis rates determined by same independent core laboratory.
52 New in 2016! Algorithm based on DATA
53 SFA Treatment Strategy - Proposal Stenosis PTA predilatation Flow-limiting dissection or >50% residual stenosis? Yes DES/Supera No Is the lesion severely calcified? No DCB Flow-limiting dissection or >50% residual stenosis? No Finished Yes Post-Dilatation Stent (spot supera)
54 SFA Treatment Strategy - Proposal Stenosis PTA predilatation Flow-limiting dissection or >50% residual stenosis? Yes DES/Supera No Is the lesion severely calcified? No Yes Consider atherectomy Debulk successful? DCB Yes No Flow-limiting dissection or >50% residual stenosis? Supera No Finished Yes Post-Dilatation Spot Supera)
55 SFA Treatment Strategy - Proposal Stenosis CTO PTA predilatation Required (US); Optional (Europe) Yes Can it be crossed? Flow-limiting dissection or >50% residual stenosis? Yes Stent No No Bypass Is the lesion severely calcified? No Yes Consider atherectomy Debulk successful? DCB Yes No Flow-limiting dissection or >50% residual stenosis? Stent No Finished Yes Post-Dilatation Stent (spot stent)
56 Thank You
57 What s New in 2016? Prakash Krishnan MD FACC Director of Endovascular Services Cardiac Catheterization Laboratory Mount Sinai Medical Center New York
Majestic Trial 12 Month Results
 Majestic Trial 12 Month Results S.Müller-Hülsbeck, MD, EBIR, FCIRSE, FICA ACADEMIC HOSPITALS Flensburg of Kiel University Ev.-Luth. Diakonissenanstalt zu Flensburg Knuthstraße 1, 24939 FLENSBURG Dept.
Majestic Trial 12 Month Results S.Müller-Hülsbeck, MD, EBIR, FCIRSE, FICA ACADEMIC HOSPITALS Flensburg of Kiel University Ev.-Luth. Diakonissenanstalt zu Flensburg Knuthstraße 1, 24939 FLENSBURG Dept.
Popliteal artery: to stent or not to stent?
 Popliteal artery: to stent or not to stent? Karl-Ludwig Schulte Vascular Center Berlin Ev. Hospital Königin Elisabeth St. Gertrauden Hospital University Hospital Charité, CC11 Humboldt-University Berlin
Popliteal artery: to stent or not to stent? Karl-Ludwig Schulte Vascular Center Berlin Ev. Hospital Königin Elisabeth St. Gertrauden Hospital University Hospital Charité, CC11 Humboldt-University Berlin
Fundación Favaloro. Fundación Favaloro
 Superficial Femoral Artery: Which h Factors Influence Patency? Oscar A. Mendiz.MD.FACC.FSCAI Chairman hi of Interventional Cardiology dil March 2010 Name: Oscar A. Mendiz Disclosure I have the following
Superficial Femoral Artery: Which h Factors Influence Patency? Oscar A. Mendiz.MD.FACC.FSCAI Chairman hi of Interventional Cardiology dil March 2010 Name: Oscar A. Mendiz Disclosure I have the following
EXCITE ISR: Initial Results. Eric J. Dippel, MD FACC On behalf of the EXCITE ISR Investigators
 EXCITE ISR: Initial Results Eric J. Dippel, MD FACC On behalf of the EXCITE ISR Investigators Disclosure Statement of Financial Interest Within the past 12 months, I or my spouse/partner have had a financial
EXCITE ISR: Initial Results Eric J. Dippel, MD FACC On behalf of the EXCITE ISR Investigators Disclosure Statement of Financial Interest Within the past 12 months, I or my spouse/partner have had a financial
IN.PACT SFA: A Prospective Randomized Trial of a Drug- Coated Balloon for Femoropopliteal Lesions Two-Year Outcomes
 IN.PACT SFA: A Prospective Randomized Trial of a Drug- Coated Balloon for Femoropopliteal Lesions Two-Year Outcomes John Laird, MD UC Davis, Sacramento, California USA On behalf of the IN.PACT SFA Trial
IN.PACT SFA: A Prospective Randomized Trial of a Drug- Coated Balloon for Femoropopliteal Lesions Two-Year Outcomes John Laird, MD UC Davis, Sacramento, California USA On behalf of the IN.PACT SFA Trial
Supera Peripheral Stent System Instructions for Use
 Supera Peripheral Stent System Instructions for Use Table of Contents 1.0 DEVICE DESCRIPTION 2.0 HOW SUPPLIED 3.0 INDICATIONS 4.0 CONTRAINDICATIONS 5.0 WARNINGS 6.0 PRECAUTIONS 6.1 Stent Delivery System
Supera Peripheral Stent System Instructions for Use Table of Contents 1.0 DEVICE DESCRIPTION 2.0 HOW SUPPLIED 3.0 INDICATIONS 4.0 CONTRAINDICATIONS 5.0 WARNINGS 6.0 PRECAUTIONS 6.1 Stent Delivery System
First Experience With Drug-Eluting Balloons in Infrapopliteal Arteries
 Journal of the American College of Cardiology Vol. 58, No. 11, 2011 2011 by the American College of Cardiology Foundation ISSN 0735-1097/$36.00 Published by Elsevier Inc. doi:10.1016/j.jacc.2011.05.034
Journal of the American College of Cardiology Vol. 58, No. 11, 2011 2011 by the American College of Cardiology Foundation ISSN 0735-1097/$36.00 Published by Elsevier Inc. doi:10.1016/j.jacc.2011.05.034
The Pantera Lux Paclitaxel DEB Device Description and Clinical Studies. Christoph Hehrlein, University Clinic Freiburg i.br.
 The Pantera Lux Paclitaxel DEB Device Description and Clinical Studies Christoph Hehrlein, University Clinic Freiburg i.br. Germany Disclosure Statement of Financial Interest Within the past 12 months,
The Pantera Lux Paclitaxel DEB Device Description and Clinical Studies Christoph Hehrlein, University Clinic Freiburg i.br. Germany Disclosure Statement of Financial Interest Within the past 12 months,
A Post-market Study to Assess the STENTYS Self-exPanding COronary Stent In AcuTe myocardial InfarctiON in Real Life APPOSITION III
 A Post-market Study to Assess the STENTYS Self-exPanding COronary Stent In AcuTe myocardial InfarctiON in Real Life APPOSITION III Gilles Montalescot, MD, PhD Pitié-Salpêtrière Hospital, Paris, France
A Post-market Study to Assess the STENTYS Self-exPanding COronary Stent In AcuTe myocardial InfarctiON in Real Life APPOSITION III Gilles Montalescot, MD, PhD Pitié-Salpêtrière Hospital, Paris, France
Drug-Eluting Balloons. Klaus Bonaventura Department of Cardiology and Angiology Heart Thorax Vascular Center, Klinikum Ernst von Bergmann, Potsdam
 Drug-Eluting Balloons Klaus Bonaventura Department of Cardiology and Angiology Heart Thorax Vascular Center, Klinikum Ernst von Bergmann, Potsdam Potential conflicts of interest Speaker s name: Klaus Bonaventura
Drug-Eluting Balloons Klaus Bonaventura Department of Cardiology and Angiology Heart Thorax Vascular Center, Klinikum Ernst von Bergmann, Potsdam Potential conflicts of interest Speaker s name: Klaus Bonaventura
LEADERS: 5-Year Follow-up
 LEADERS: -Year Follow-up from a Prospective, Randomized Trial of Biolimus A9-eluting Stents with a Biodegradable Polymer vs. Sirolimus-eluting Stents with a Durable Polymer : Final Report of the LEADERS
LEADERS: -Year Follow-up from a Prospective, Randomized Trial of Biolimus A9-eluting Stents with a Biodegradable Polymer vs. Sirolimus-eluting Stents with a Durable Polymer : Final Report of the LEADERS
Evolu'on of Balloon Angioplasty From POBA to newer technologies NCVH May 26, 2015
 Evolu'on of Balloon Angioplasty From POBA to newer technologies NCVH May 26, 2015 Cezar Staniloae, MD Heart and Vascular Ins7tute New York University Medical Center Overview 1. Physics of balloon angioplasty
Evolu'on of Balloon Angioplasty From POBA to newer technologies NCVH May 26, 2015 Cezar Staniloae, MD Heart and Vascular Ins7tute New York University Medical Center Overview 1. Physics of balloon angioplasty
Cilostazol versus Clopidogrel after Coronary Stenting
 Cilostazol versus Clopidogrel after Coronary Stenting Seong-Wook Park, MD, PhD, FACC Division of Cardiology, Asan Medical Center University of Ulsan College of Medicine Seoul, Korea AMC, 2004 Background
Cilostazol versus Clopidogrel after Coronary Stenting Seong-Wook Park, MD, PhD, FACC Division of Cardiology, Asan Medical Center University of Ulsan College of Medicine Seoul, Korea AMC, 2004 Background
Antiplatelet and anticoagulation treatment of patients undergoing carotid and peripheral artery angioplasty
 Round Table: Antithrombotic therapy beyond ACS Antiplatelet and anticoagulation treatment of patients undergoing carotid and peripheral artery angioplasty M. Matsagkas, MD, PhD, EBSQ-Vasc Associate Professor
Round Table: Antithrombotic therapy beyond ACS Antiplatelet and anticoagulation treatment of patients undergoing carotid and peripheral artery angioplasty M. Matsagkas, MD, PhD, EBSQ-Vasc Associate Professor
UNITED STATES SECURITIES AND EXCHANGE COMMISSION Washington, D.C. 20549 Form 10-K
 UNITED STATES SECURITIES AND EXCHANGE COMMISSION Washington, D.C. 20549 Form 10-K x ANNUAL REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 For the year ended December 31,
UNITED STATES SECURITIES AND EXCHANGE COMMISSION Washington, D.C. 20549 Form 10-K x ANNUAL REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 For the year ended December 31,
PRECOMBAT Trial. Seung-Whan Lee, MD, PhD On behalf of the PRECOMBAT Investigators
 Premier of Randomized Comparison of Bypass Surgery versus Angioplasty Using Sirolimus-Eluting Stent in Patients with Left Main Coronary Artery Disease PRECOMBAT Trial Seung-Whan Lee, MD, PhD On behalf
Premier of Randomized Comparison of Bypass Surgery versus Angioplasty Using Sirolimus-Eluting Stent in Patients with Left Main Coronary Artery Disease PRECOMBAT Trial Seung-Whan Lee, MD, PhD On behalf
Steven J. Yakubov, MD FACC For the CoreValve US Clinical Investigators
 Long-Term Outcomes Using a Self- Expanding Bioprosthesis in Patients With Severe Aortic Stenosis Deemed Extreme Risk for Surgery: Two-Year Results From the CoreValve US Pivotal Trial Steven J. Yakubov,
Long-Term Outcomes Using a Self- Expanding Bioprosthesis in Patients With Severe Aortic Stenosis Deemed Extreme Risk for Surgery: Two-Year Results From the CoreValve US Pivotal Trial Steven J. Yakubov,
Coronary Bifurcation Treatment: Update from the European Bifurcation Club. Remo Albiero, MD Ist. Clinico S. Rocco Brescia (Italy)
 Coronary Bifurcation Treatment: Update from the European Bifurcation Club Remo Albiero, MD Ist. Clinico S. Rocco Brescia (Italy) Disclosure Statement of Financial Interest Within the past 12 months, I
Coronary Bifurcation Treatment: Update from the European Bifurcation Club Remo Albiero, MD Ist. Clinico S. Rocco Brescia (Italy) Disclosure Statement of Financial Interest Within the past 12 months, I
INSTRUCTIONS FOR USE
 LUTONIX 035 Drug Coated Balloon PTA Catheter INSTRUCTIONS FOR USE Caution: Federal Law (USA) restricts this device to sale by or on the order of a physician. Table of Contents 1 DEVICE DESCRIPTION... 2
LUTONIX 035 Drug Coated Balloon PTA Catheter INSTRUCTIONS FOR USE Caution: Federal Law (USA) restricts this device to sale by or on the order of a physician. Table of Contents 1 DEVICE DESCRIPTION... 2
Antonio Colombo MD on behalf of the SECURITY Investigators
 Second Generation Drug-Eluting Stents Implantation Followed by Six Versus Twelve-Month - Dual Antiplatelet Therapy - The SECURITY Randomized Clinical Trial Antonio Colombo MD on behalf of the SECURITY
Second Generation Drug-Eluting Stents Implantation Followed by Six Versus Twelve-Month - Dual Antiplatelet Therapy - The SECURITY Randomized Clinical Trial Antonio Colombo MD on behalf of the SECURITY
MEDICAL POLICY No. 91580-R1 DRUG-ELUTING STENTS FOR ISCHEMIC HEART DISEASE
 DRUG-ELUTING STENTS FOR ISCHEMIC HEART DISEASE Effective Date: October 1, 2015 Review Dates: 10/11, 10/12, 10/13, 8/14, 8/15 Date Of Origin: October 12, 2011 Status: Current Summary of Changes Clarifications:
DRUG-ELUTING STENTS FOR ISCHEMIC HEART DISEASE Effective Date: October 1, 2015 Review Dates: 10/11, 10/12, 10/13, 8/14, 8/15 Date Of Origin: October 12, 2011 Status: Current Summary of Changes Clarifications:
Endovascular treatment of symptomatic atherosclerotic
 Drug-Coated Balloon Versus Standard Percutaneous Transluminal Angioplasty for the Treatment of Superficial Femoral and Popliteal Peripheral Artery Disease 12-Month Results From the IN.PACT SFA Randomized
Drug-Coated Balloon Versus Standard Percutaneous Transluminal Angioplasty for the Treatment of Superficial Femoral and Popliteal Peripheral Artery Disease 12-Month Results From the IN.PACT SFA Randomized
Ostial LAD: Single stent approach is the best. Antonio A. Pocoví, MD, FSCAI, MTSAC, Advisory Council Member, CACI
 Ostial LAD: Single stent approach is the best Antonio A. Pocoví, MD, FSCAI, MTSAC, Advisory Council Member, CACI Chair, Interventional Cardiology Sanatorio San Lucas Instituto Alexander Fleming Buenos
Ostial LAD: Single stent approach is the best Antonio A. Pocoví, MD, FSCAI, MTSAC, Advisory Council Member, CACI Chair, Interventional Cardiology Sanatorio San Lucas Instituto Alexander Fleming Buenos
Renal artery stenting: are there any indications left?
 there any indications left? Luís Mendes Pedro, MD. PhD, FEBVS Lisbon Academic Medical Centre (University of Lisbon and Hospital Santa Maria) Instituto Cardiovascular de Lisboa Disclosures Speaker name:
there any indications left? Luís Mendes Pedro, MD. PhD, FEBVS Lisbon Academic Medical Centre (University of Lisbon and Hospital Santa Maria) Instituto Cardiovascular de Lisboa Disclosures Speaker name:
Duration of Dual Antiplatelet Therapy After Coronary Stenting
 Duration of Dual Antiplatelet Therapy After Coronary Stenting C. DEAN KATSAMAKIS, DO, FACC, FSCAI INTERVENTIONAL CARDIOLOGIST ADVOCATE LUTHERAN GENERAL HOSPITAL INTRODUCTION Coronary artery stents are
Duration of Dual Antiplatelet Therapy After Coronary Stenting C. DEAN KATSAMAKIS, DO, FACC, FSCAI INTERVENTIONAL CARDIOLOGIST ADVOCATE LUTHERAN GENERAL HOSPITAL INTRODUCTION Coronary artery stents are
The Minvasys Amazonia Pax & Nile Pax Polymer Free Paclitaxel Eluting Stent Program
 The Minvasys Amazonia Pax & Nile Pax Polymer Free Paclitaxel Eluting Stent Program Jean Fajadet, MD, FESC Clinique Pasteur - Toulouse - France Disclosure statement Nothing to disclose Dedicated Delivery
The Minvasys Amazonia Pax & Nile Pax Polymer Free Paclitaxel Eluting Stent Program Jean Fajadet, MD, FESC Clinique Pasteur - Toulouse - France Disclosure statement Nothing to disclose Dedicated Delivery
IN.PACT ADMIRAL DRUG-COATED BALLOON NEW TECHNOLOGY ADD-ON PAYMENT (NTAP)
 IN.PACT ADMIRAL DRUG-COATED BALLOON NEW TECHNOLOGY ADD-ON PAYMENT (NTAP) New Technology Add-on (NTAP) for DCB OVERVIEW Effective October 1, 2015, hospital inpatient cases using a drug-coated balloon (DCB)
IN.PACT ADMIRAL DRUG-COATED BALLOON NEW TECHNOLOGY ADD-ON PAYMENT (NTAP) New Technology Add-on (NTAP) for DCB OVERVIEW Effective October 1, 2015, hospital inpatient cases using a drug-coated balloon (DCB)
Review of Intracoronary Radiation for In-Stent Restenosis
 Review of Intracoronary Radiation for In-Stent Restenosis Robert Lew, MBBS, FRACP, Andrew Ajani, MD, Ron Waksman, MD, FACC Vascular Brachytherapy using beta and gamma radiation is the standard care for
Review of Intracoronary Radiation for In-Stent Restenosis Robert Lew, MBBS, FRACP, Andrew Ajani, MD, Ron Waksman, MD, FACC Vascular Brachytherapy using beta and gamma radiation is the standard care for
1 Table of Contents 1.1 List of Tables 1.2 List of Figures 2 Introduction 3 Global Peripheral Vascular Devices Market - Market Characterization 3.
 1 Table of Contents 1.1 List of Tables 1.2 List of Figures 2 Introduction 3 Global Peripheral Vascular Devices Market - Market Characterization 3.1 Peripheral Vascular Devices Market, Revenue ($m), 2004-3.2
1 Table of Contents 1.1 List of Tables 1.2 List of Figures 2 Introduction 3 Global Peripheral Vascular Devices Market - Market Characterization 3.1 Peripheral Vascular Devices Market, Revenue ($m), 2004-3.2
Intracoronary Stenting and. Robert A. Byrne, Julinda Mehilli, Salvatore Cassese, Franz-Josef Neumann, Susanne Pinieck, Tomohisa Tada,
 Prospective, Randomized Trial of Paclitaxel-Eluting Balloon versus Paclitaxel-Eluting Stent versus Balloon Angioplasty for Treatment of Coronary Restenosis in Limus- Eluting Stents Intracoronary Stenting
Prospective, Randomized Trial of Paclitaxel-Eluting Balloon versus Paclitaxel-Eluting Stent versus Balloon Angioplasty for Treatment of Coronary Restenosis in Limus- Eluting Stents Intracoronary Stenting
Carotid Artery Stenting? Comparison of the outcome of octogenarian patients according to the stent type. Data from a multicentre registry
 Closed- or Open-cell stents in Carotid Artery Stenting? Comparison of the outcome of octogenarian patients according to the stent type. Data from a multicentre registry Nikas D 1, Reimers B 2, Iakovou
Closed- or Open-cell stents in Carotid Artery Stenting? Comparison of the outcome of octogenarian patients according to the stent type. Data from a multicentre registry Nikas D 1, Reimers B 2, Iakovou
Health Policy Advisory Committee on Technology Technology Brief
 Health Policy Advisory Committee on Technology Technology Brief LifeStent vascular stent for symptomatic lesions of the superficial femoral or proximal popliteal artery August 2012 State of Queensland
Health Policy Advisory Committee on Technology Technology Brief LifeStent vascular stent for symptomatic lesions of the superficial femoral or proximal popliteal artery August 2012 State of Queensland
Copenhagen University Hospital Rigshospitalet Aarhus University Hospital Skejby Denmark
 Long-term outcome after drug-eluting versus bare-metal stent implantation in patients with ST-elevation myocardial infarction 3 year follow-up of the randomised trial Peter Clemmensen, Henning Kelbæk,
Long-term outcome after drug-eluting versus bare-metal stent implantation in patients with ST-elevation myocardial infarction 3 year follow-up of the randomised trial Peter Clemmensen, Henning Kelbæk,
Yukon Choice DES + Translumina - The polymer-free DES solution. www.translumina.de. Coronary Stent System for Drug Application
 Coronary Stent System for Drug Application Yukon Choice DES + new catheter polymer-free DES Translumina - The polymer-free DES solution www.translumina.de A First Class Stent The YUKON Choice DES+ is the
Coronary Stent System for Drug Application Yukon Choice DES + new catheter polymer-free DES Translumina - The polymer-free DES solution www.translumina.de A First Class Stent The YUKON Choice DES+ is the
Rethink Atherectomy: Expert Insights Into Clinical Application and Use of the JETSTREAM System
 , LLC an HMP Communications Holdings Company September 2013 Volume 25/ Supplement B www.invasivecardiology.com The Official Journal of the International Andreas Gruentzig Society Rethink Atherectomy: Expert
, LLC an HMP Communications Holdings Company September 2013 Volume 25/ Supplement B www.invasivecardiology.com The Official Journal of the International Andreas Gruentzig Society Rethink Atherectomy: Expert
University of Ulsan College of Medicine, Asan Medical Center on behalf of the REAL-LATE and the ZEST-LATE trial
 Duration of Dual Antiplatelet Therapy After Drug-Eluting Stent Implantation A Pooled Analysis of the REAL-LATE and the ZEST-LATE Trial Seung-Jung Park MD PhD Seung-Jung Park, MD, PhD, University of Ulsan
Duration of Dual Antiplatelet Therapy After Drug-Eluting Stent Implantation A Pooled Analysis of the REAL-LATE and the ZEST-LATE Trial Seung-Jung Park MD PhD Seung-Jung Park, MD, PhD, University of Ulsan
Measure #257 (NQF 1519): Statin Therapy at Discharge after Lower Extremity Bypass (LEB) National Quality Strategy Domain: Effective Clinical Care
 Measure #257 (NQF 1519): Statin Therapy at Discharge after Lower Extremity Bypass (LEB) National Quality Strategy Domain: Effective Clinical Care 2016 PQRS OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY
Measure #257 (NQF 1519): Statin Therapy at Discharge after Lower Extremity Bypass (LEB) National Quality Strategy Domain: Effective Clinical Care 2016 PQRS OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY
2014 A N N U A L R E P O R T
 2 014 A N N U A L R E P O R T 2014: A Year of Transformation Spectranetics delivers innovative, minimally invasive cardiovascular technologies that are transforming patient care in more than 65 countries
2 014 A N N U A L R E P O R T 2014: A Year of Transformation Spectranetics delivers innovative, minimally invasive cardiovascular technologies that are transforming patient care in more than 65 countries
Clinical Research Intracoronary Stenting with Crushing in Coronary Artery Bifurcation Lesions: Initial Results and Medium-Term Follow Up
 Hellenic J Cardiol 45: 379-383, 2004 Clinical Research Intracoronary Stenting with Crushing in Coronary Artery Bifurcation Lesions: Initial Results and Medium-Term Follow Up PETROS S. DARDAS, DIMITRIS
Hellenic J Cardiol 45: 379-383, 2004 Clinical Research Intracoronary Stenting with Crushing in Coronary Artery Bifurcation Lesions: Initial Results and Medium-Term Follow Up PETROS S. DARDAS, DIMITRIS
Rotational Atherectomy for the Treatment of In-Stent Restenosis
 Home SVCC Area: English - Español - Português Rotational Atherectomy for the Treatment of In-Stent Restenosis Steven L. Goldberg, MD Cardiac Catheterization Laboratory, University of Washington Medical
Home SVCC Area: English - Español - Português Rotational Atherectomy for the Treatment of In-Stent Restenosis Steven L. Goldberg, MD Cardiac Catheterization Laboratory, University of Washington Medical
Editorial. Adult Cardiology - Meta-analysis
 Editorial Adult Cardiology - Meta-analysis Simple Versus Complex Bifurcation Stenting Strategies A Meta Analysis of Randomized Controlled Trials in the Drug Eluting Stent Era Ana Beatriz R. Medrano, MD
Editorial Adult Cardiology - Meta-analysis Simple Versus Complex Bifurcation Stenting Strategies A Meta Analysis of Randomized Controlled Trials in the Drug Eluting Stent Era Ana Beatriz R. Medrano, MD
ESC Guidelines on the diagnosis and treatment of peripheral artery diseases Lower extremity artery disease. Erich Minar Medical University Vienna
 ESC Guidelines on the diagnosis and treatment of peripheral artery diseases Lower extremity artery disease Erich Minar Medical University Vienna for the Task Force on the Diagnosis and Treatment of Peripheral
ESC Guidelines on the diagnosis and treatment of peripheral artery diseases Lower extremity artery disease Erich Minar Medical University Vienna for the Task Force on the Diagnosis and Treatment of Peripheral
Overview of Newer Stent Devices for Aneurysm Treatment
 Overview of Newer Stent Devices for Aneurysm Treatment Randall C. Edgell, M.D. Associate Professor Vascular and Interventional Neurology Saint Louis University Disclosure Outcome adjudication for THERAPY,
Overview of Newer Stent Devices for Aneurysm Treatment Randall C. Edgell, M.D. Associate Professor Vascular and Interventional Neurology Saint Louis University Disclosure Outcome adjudication for THERAPY,
Vascular Stent System (6F, 20 100mm stents)
 100000000922.2 Instructions for Use Cordis S.M.A.R.T. CONTROL Vascular Stent System (6F, 20 100mm stents) Cordis S.M.A.R.T. Vascular Stent System (6F, 120 150mm stents) Symbols Catalog Number LOT NonPyrogenic
100000000922.2 Instructions for Use Cordis S.M.A.R.T. CONTROL Vascular Stent System (6F, 20 100mm stents) Cordis S.M.A.R.T. Vascular Stent System (6F, 120 150mm stents) Symbols Catalog Number LOT NonPyrogenic
Clinical Programs. Medtronic Coronary Stent Systems. driver BMS
 Clinical Programs Medtronic Coronary Stent Systems Endeavor DES driver BMS July 2010 Contents Overview of Clinical Programs... 2 Drug-Eluting Stents ENDEAVOR I*... 4 ENDEAVOR II*... 6 ENDEAVOR II Continued
Clinical Programs Medtronic Coronary Stent Systems Endeavor DES driver BMS July 2010 Contents Overview of Clinical Programs... 2 Drug-Eluting Stents ENDEAVOR I*... 4 ENDEAVOR II*... 6 ENDEAVOR II Continued
Overview. Total Joint Replacement in the U.S. KP National Total Joint Registry EMR Tools and Outcome Assessment: A Model for Vascular Surgery?
 KP National Total Joint Registry EMR Tools and Outcome Assessment: A Model for Vascular Surgery? Liz Paxton Director of Surgical Outcomes and Analysis Overview KP Total Joint Replacement Registry Background
KP National Total Joint Registry EMR Tools and Outcome Assessment: A Model for Vascular Surgery? Liz Paxton Director of Surgical Outcomes and Analysis Overview KP Total Joint Replacement Registry Background
WHY DO MY LEGS HURT? Veins, arteries, and other stuff.
 WHY DO MY LEGS HURT? Veins, arteries, and other stuff. Karl A. Illig, MD Professor of Surgery Chief, Division of Vascular Surgery Mitzi Ekers, ARNP April 2013 Why do my legs hurt? CONFLICTS OF INTEREST
WHY DO MY LEGS HURT? Veins, arteries, and other stuff. Karl A. Illig, MD Professor of Surgery Chief, Division of Vascular Surgery Mitzi Ekers, ARNP April 2013 Why do my legs hurt? CONFLICTS OF INTEREST
JUL 2 2008. Ms. Kendra Basler Regulatory Affairs Associate Abbott Vascular Cardiac Therapies 3200 Lakeside Drive Santa Clara, CA 95054-2807
 DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service JUL 2 2008 Food and Drug Administration 9200 Corporate Boulevard Rockville MD 20850 Ms. Kendra Basler Regulatory Affairs Associate Abbott Vascular
DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service JUL 2 2008 Food and Drug Administration 9200 Corporate Boulevard Rockville MD 20850 Ms. Kendra Basler Regulatory Affairs Associate Abbott Vascular
SeQuent Please balloon catheter for in-stent coronary restenosis. NICE medical technology guidance 1. Issue date: December 2010
 Issue date: December 2010 SeQuent Please balloon catheter for in-stent coronary restenosis NICE medical technology guidance 1 1 NICE medical technology guidance 1 NICE medical technology guidance 1 SeQuent
Issue date: December 2010 SeQuent Please balloon catheter for in-stent coronary restenosis NICE medical technology guidance 1 1 NICE medical technology guidance 1 NICE medical technology guidance 1 SeQuent
Is Stenting or Coronary Artery By-pass Grafting the Better Treatment for This Patient?
 Is Stenting or Coronary Artery By-pass Grafting the Better Treatment for This Patient? --- NIRS-IVUS TVC Imaging Adds Additional Information for the Heart Team Dr. Luis Tami Memorial Regional Hospital
Is Stenting or Coronary Artery By-pass Grafting the Better Treatment for This Patient? --- NIRS-IVUS TVC Imaging Adds Additional Information for the Heart Team Dr. Luis Tami Memorial Regional Hospital
Main Effect of Screening for Coronary Artery Disease Using CT
 Main Effect of Screening for Coronary Artery Disease Using CT Angiography on Mortality and Cardiac Events in High risk Patients with Diabetes: The FACTOR-64 Randomized Clinical Trial Joseph B. Muhlestein,
Main Effect of Screening for Coronary Artery Disease Using CT Angiography on Mortality and Cardiac Events in High risk Patients with Diabetes: The FACTOR-64 Randomized Clinical Trial Joseph B. Muhlestein,
Renovascular Hypertension
 Renovascular Hypertension Philip Stockwell, MD Assistant Professor of Medicine (Clinical) Warren Alpert School of Medicine Cardiology for the Primary Care Provider September 28, 201 Renovascular Hypertension
Renovascular Hypertension Philip Stockwell, MD Assistant Professor of Medicine (Clinical) Warren Alpert School of Medicine Cardiology for the Primary Care Provider September 28, 201 Renovascular Hypertension
Talent Thoracic Stent Graft with THE Xcelerant Delivery System. Expanding the Indications for TEVAR
 Talent Thoracic with THE Xcelerant Delivery System Expanding the Indications for TEVAR Talent Thoracic Precise placement 1 Broad patient applicability 1 Excellent clinical outcomes 1, a + Xcelerant Delivery
Talent Thoracic with THE Xcelerant Delivery System Expanding the Indications for TEVAR Talent Thoracic Precise placement 1 Broad patient applicability 1 Excellent clinical outcomes 1, a + Xcelerant Delivery
RX Herculink Elite Renal and Biliary Stent system Instructions for Use. RX Herculink Elite Renal Stent System. Table of Contents
 RX Herculink Elite Renal and Biliary Stent system Instructions for Use Table of Contents RX Herculink Elite Renal Stent System 1.0 DEVICE DESCRIPTION Table 1. In Vitro* Device Specifications 2.0 HOW SUPPLIED
RX Herculink Elite Renal and Biliary Stent system Instructions for Use Table of Contents RX Herculink Elite Renal Stent System 1.0 DEVICE DESCRIPTION Table 1. In Vitro* Device Specifications 2.0 HOW SUPPLIED
CORONARY ARTERY BYPASS GRAFTS, STENTS, AND EXTRACORONARY CARDIAC DZ. Charles White MD
 CORONARY ARTERY BYPASS GRAFTS, STENTS, AND EXTRACORONARY CARDIAC DZ Charles White MD Director of Thoracic Imaging Department of Radiology University of Maryland CORONARY ARTERY BYPASS GRAFTS First performed
CORONARY ARTERY BYPASS GRAFTS, STENTS, AND EXTRACORONARY CARDIAC DZ Charles White MD Director of Thoracic Imaging Department of Radiology University of Maryland CORONARY ARTERY BYPASS GRAFTS First performed
Interventional Cardiology Peripheral Interventions Rhythm Management
 FY2016 Hospital Inpatient Final Rule (IPPS) Interventional Cardiology Peripheral Interventions Rhythm Management On July 31, 2015, the Centers for Medicare and Medicaid Services (CMS) released the Final
FY2016 Hospital Inpatient Final Rule (IPPS) Interventional Cardiology Peripheral Interventions Rhythm Management On July 31, 2015, the Centers for Medicare and Medicaid Services (CMS) released the Final
Early healing after treatment of coronary lesions by everolimus, or biolimus eluting bioresorbable polymer stents
 Early healing after treatment of coronary lesions by everolimus, or biolimus eluting bioresorbable polymer stents One-month results of the study Ida Riise Balleby 1, Trine Krejberg Ørhøj 1, Christian Juhl
Early healing after treatment of coronary lesions by everolimus, or biolimus eluting bioresorbable polymer stents One-month results of the study Ida Riise Balleby 1, Trine Krejberg Ørhøj 1, Christian Juhl
Perspectives on the Selection and Duration of Dual Antiplatelet Therapy
 Perspectives on the Selection and Duration of Dual Antiplatelet Therapy Dominick J. Angiolillo, MD, PhD, FACC, FESC, FSCAI Director of Cardiovascular Research Associate Professor of Medicine University
Perspectives on the Selection and Duration of Dual Antiplatelet Therapy Dominick J. Angiolillo, MD, PhD, FACC, FESC, FSCAI Director of Cardiovascular Research Associate Professor of Medicine University
Coronary Stents. What is a Coronary Artery stent?
 What is a Coronary Artery stent? Coronary Stents A coronary stent is stainless tube with slots. It is mounted on a balloon catheter in a "crimped" or collapsed state. When the balloon of is inflated, the
What is a Coronary Artery stent? Coronary Stents A coronary stent is stainless tube with slots. It is mounted on a balloon catheter in a "crimped" or collapsed state. When the balloon of is inflated, the
Innova Vascular OVER-THE-WIRE
 POTENTIAL ADVERSE EVENTS... HOW SUPPLIED...6 909580-0 Innova Vascular OVER-THE-WIRE Self-Expanding Stent System TABLE OF CONTENTS 05-07 < en > WARNING... DEVICE DESCRIPTION... Contents... INTENDED USE
POTENTIAL ADVERSE EVENTS... HOW SUPPLIED...6 909580-0 Innova Vascular OVER-THE-WIRE Self-Expanding Stent System TABLE OF CONTENTS 05-07 < en > WARNING... DEVICE DESCRIPTION... Contents... INTENDED USE
Iliac Artery Disease: A Case-Based Approach To Stent Selection
 Society for Vascular Medicine Iliac Artery Disease: A Case-Based Approach To Stent Selection Annotated Cited Reference Material (2002). "MRC/BHF Heart Protection Study of cholesterol lowering with simvastatin
Society for Vascular Medicine Iliac Artery Disease: A Case-Based Approach To Stent Selection Annotated Cited Reference Material (2002). "MRC/BHF Heart Protection Study of cholesterol lowering with simvastatin
Orsiro Hybrid Drug Eluting Stent Industry's first hybrid DES
 Vascular Intervention Drug Eluting Stents Orsiro Orsiro Hybrid Drug Eluting Stent Industry's first hybrid DES Orsiro An ideal combination of passive and active components The Orsiro Hybrid Drug Eluting
Vascular Intervention Drug Eluting Stents Orsiro Orsiro Hybrid Drug Eluting Stent Industry's first hybrid DES Orsiro An ideal combination of passive and active components The Orsiro Hybrid Drug Eluting
Wingspan Stent System with Gateway PTA Balloon Catheter
 Wingspan System with Gateway PTA Balloon Catheter Directions for Use AUS Australian Sponsor Address Boston Scientific (Australia) Pty Ltd PO Box 332 BOTANY NSW 1455 Australia Free Phone 1800 676 133 Free
Wingspan System with Gateway PTA Balloon Catheter Directions for Use AUS Australian Sponsor Address Boston Scientific (Australia) Pty Ltd PO Box 332 BOTANY NSW 1455 Australia Free Phone 1800 676 133 Free
GUIDELINES ON MEDICAL DEVICES EVALUATION OF CLINICAL DATA - A GUIDE FOR MANUFACTURERS AND NOTIFIED BODIES -
 EUROPEAN COMMISSION ENTERPRISE AND INDUSTRY DIRECTORATE-GENERAL Consumer goods Cosmetics and Medical Devices MEDDEV 2.7.1 Appendix 1 December 2008 GUIDELINES ON MEDICAL DEVICES EVALUATION OF CLINICAL DATA
EUROPEAN COMMISSION ENTERPRISE AND INDUSTRY DIRECTORATE-GENERAL Consumer goods Cosmetics and Medical Devices MEDDEV 2.7.1 Appendix 1 December 2008 GUIDELINES ON MEDICAL DEVICES EVALUATION OF CLINICAL DATA
Repeat Coronary Revascularization Procedures after Successful Bare-Metal or Drug-Eluting Stent Implantation
 Original Contribution Repeat Coronary Revascularization Procedures after Successful Bare-Metal or Drug-Eluting Stent Implantation Cynthia A. Yock, MS, J. Michael Isbill, MS, Spencer B. King III, MD, Mark
Original Contribution Repeat Coronary Revascularization Procedures after Successful Bare-Metal or Drug-Eluting Stent Implantation Cynthia A. Yock, MS, J. Michael Isbill, MS, Spencer B. King III, MD, Mark
European Heart Journal (1999) 20, 1014 1019 Article No. euhj.1998.1395, available online at http://www.idealibrary.com on
 European Heart Journal (1999) 20, 1014 1019 Article No. euhj.1998.1395, available online at http://www.idealibrary.com on Working Group Report Indications for intracoronary stent placement: the European
European Heart Journal (1999) 20, 1014 1019 Article No. euhj.1998.1395, available online at http://www.idealibrary.com on Working Group Report Indications for intracoronary stent placement: the European
OPINION OPtical frequency domain imaging vs. INtravascular ultrasound in percutaneous coronary InterventiON
 OPINION OPtical frequency domain imaging vs. INtravascular ultrasound in percutaneous coronary InterventiON - Baseline preliminary results - Takashi Akasaka, MD, PhD, FESC Wakayama Medical University,
OPINION OPtical frequency domain imaging vs. INtravascular ultrasound in percutaneous coronary InterventiON - Baseline preliminary results - Takashi Akasaka, MD, PhD, FESC Wakayama Medical University,
Interventional Cardiology Peripheral Interventions Rhythm Management
 FY2016 Hospital Inpatient Rule (IPPS) Interventional Cardiology Peripheral Interventions Rhythm Management On April 17, 2015 the Centers for Medicare and Medicaid Services (CMS) released the Hospital Inpatient
FY2016 Hospital Inpatient Rule (IPPS) Interventional Cardiology Peripheral Interventions Rhythm Management On April 17, 2015 the Centers for Medicare and Medicaid Services (CMS) released the Hospital Inpatient
Bard LifeStar Vascular Stent System
 Bard LifeStar Vascular Stent System Instructions For Use (IFU) Bard LifeStar Vascular Stent System Delivery System Diagram B05685 Vers.1/08-11 1 Instructions for use Read the Bard LifeStar Vascular Stent
Bard LifeStar Vascular Stent System Instructions For Use (IFU) Bard LifeStar Vascular Stent System Delivery System Diagram B05685 Vers.1/08-11 1 Instructions for use Read the Bard LifeStar Vascular Stent
Using Clinical Registries to Create Evidence-based Health Care Policy : Experiences from Ontario, Canada
 Using Clinical Registries to Create Evidence-based Health Care Policy : Experiences from Ontario, Canada April 2009 Jack V. Tu, MD PhD FRCPC CANADA RESEARCH CHAIR IN HEALTH SERVICES RESEARCH Institute
Using Clinical Registries to Create Evidence-based Health Care Policy : Experiences from Ontario, Canada April 2009 Jack V. Tu, MD PhD FRCPC CANADA RESEARCH CHAIR IN HEALTH SERVICES RESEARCH Institute
Advances in Stent Technology
 Nurse/Technologist Symposium Advances in Stent Technology Antonietta Tolentino, CCRN, MSN, ANP-C Adult Nurse Practitioner, Cardiac Catheterization Laboratory The Zena and Michael A. Weiner Cardiovascular
Nurse/Technologist Symposium Advances in Stent Technology Antonietta Tolentino, CCRN, MSN, ANP-C Adult Nurse Practitioner, Cardiac Catheterization Laboratory The Zena and Michael A. Weiner Cardiovascular
Non-surgical treatment of severe varicose veins
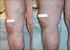 Non-surgical treatment of severe varicose veins Yasu Harasaki UCHSC Department of Surgery General Surgery Grand Rounds March 19, 2007 Definition Dilated, palpable, subcutaneous veins generally >3mm in
Non-surgical treatment of severe varicose veins Yasu Harasaki UCHSC Department of Surgery General Surgery Grand Rounds March 19, 2007 Definition Dilated, palpable, subcutaneous veins generally >3mm in
EXAMINATION trial. Manel Sabaté Hospital Clínic, Barcelona (On behalf of the Examination Investigators)
 EXAMINATION trial Manel Sabaté Hospital Clínic, Barcelona (On behalf of the Examination Investigators) EXAMINATION trial Background and Rationale (I) Acute coronary syndromes repeatedly appear as independent
EXAMINATION trial Manel Sabaté Hospital Clínic, Barcelona (On behalf of the Examination Investigators) EXAMINATION trial Background and Rationale (I) Acute coronary syndromes repeatedly appear as independent
Endovascular Repair of the Superficial Femoral Artery
 Supplement to Produced under an educational grant from W. L. Gore & Associates September 2008 Endovascular Repair of the Superficial Femoral Artery A roundtable discussion of the challenges in treating
Supplement to Produced under an educational grant from W. L. Gore & Associates September 2008 Endovascular Repair of the Superficial Femoral Artery A roundtable discussion of the challenges in treating
Patients suffering from critical limb ischemia (CLI)
 Building a Successful Amputation Prevention Program Our single-center experience implementing an amputation prevention algorithm and how it has led to a trend in reduced amputation rates. By Jihad A. Mustapha,
Building a Successful Amputation Prevention Program Our single-center experience implementing an amputation prevention algorithm and how it has led to a trend in reduced amputation rates. By Jihad A. Mustapha,
How To Determine Pad
 Process Representation #1 : The PAD algorithm as a sequential flow thru all sections An exploded version of the above scoped section flow is shown below. Notes: The flow presupposes existing services (
Process Representation #1 : The PAD algorithm as a sequential flow thru all sections An exploded version of the above scoped section flow is shown below. Notes: The flow presupposes existing services (
St Lucia Diabetes and Hypertension Screening and Disease Management Programs
 St Lucia Diabetes and Hypertension Screening and Disease Management Programs Michael Graven, MD, MSc,, MPH, FAAP Health Informatics and Neonatal Pediatrics Dalhousie University Halifax, Nova Scotia CANADA
St Lucia Diabetes and Hypertension Screening and Disease Management Programs Michael Graven, MD, MSc,, MPH, FAAP Health Informatics and Neonatal Pediatrics Dalhousie University Halifax, Nova Scotia CANADA
Polymer-Based, Paclitaxel-Eluting TAXUS Liberté Stent in De Novo Lesions: The Pivotal TAXUS ATLAS Trial
 Polymer-Based, Paclitaxel-Eluting Stent in De Novo Lesions: The Pivotal TAXUS ATLAS Trial Mark A. Turco, John A. Ormiston, Jeffrey J. Popma, Lazar Mandinov, Charles D. O'Shaughnessy, Tift Mann, Thomas
Polymer-Based, Paclitaxel-Eluting Stent in De Novo Lesions: The Pivotal TAXUS ATLAS Trial Mark A. Turco, John A. Ormiston, Jeffrey J. Popma, Lazar Mandinov, Charles D. O'Shaughnessy, Tift Mann, Thomas
RATE VERSUS RHYTHM CONTROL OF ATRIAL FIBRILLATION: SPECIAL CONSIDERATION IN ELDERLY. Charles Jazra
 RATE VERSUS RHYTHM CONTROL OF ATRIAL FIBRILLATION: SPECIAL CONSIDERATION IN ELDERLY Charles Jazra NO CONFLICT OF INTEREST TO DECLARE Relationship Between Atrial Fibrillation and Age Prevalence, percent
RATE VERSUS RHYTHM CONTROL OF ATRIAL FIBRILLATION: SPECIAL CONSIDERATION IN ELDERLY Charles Jazra NO CONFLICT OF INTEREST TO DECLARE Relationship Between Atrial Fibrillation and Age Prevalence, percent
Presenter: Marco Valgimigli, MD PhD, FESC Erasmus MC, Thoraxcenter Rotterdam The Netherlands
 Comparing zotarolimus-eluting and bare-metal stent efficacy in selected high bleeding risk patients treated with a short dual antiplatelet therapy duration. A pre-specified analysis from the The Zotarolimuseluting
Comparing zotarolimus-eluting and bare-metal stent efficacy in selected high bleeding risk patients treated with a short dual antiplatelet therapy duration. A pre-specified analysis from the The Zotarolimuseluting
Summary. Signature: Pol J Radiol, 2013; 78(3): 74-79 DOI: 10.12659/PJR.889245
 Signature: Pol J Radiol, 2013; 78(3): 74-79 DOI: 10.12659/PJR.889245 CASE REPORT Received: 2013.04.02 Accepted: 2013.07.16 Mechanical thrombectomy using Rotarex system and stent-in-stent placement for
Signature: Pol J Radiol, 2013; 78(3): 74-79 DOI: 10.12659/PJR.889245 CASE REPORT Received: 2013.04.02 Accepted: 2013.07.16 Mechanical thrombectomy using Rotarex system and stent-in-stent placement for
Systolic Blood Pressure Intervention Trial (SPRINT) Principal Results
 Systolic Blood Pressure Intervention Trial (SPRINT) Principal Results Paul K. Whelton, MB, MD, MSc Chair, SPRINT Steering Committee Tulane University School of Public Health and Tropical Medicine, and
Systolic Blood Pressure Intervention Trial (SPRINT) Principal Results Paul K. Whelton, MB, MD, MSc Chair, SPRINT Steering Committee Tulane University School of Public Health and Tropical Medicine, and
Understanding Coronary Artery Disease, Cardiac Catheterization, and Treatment Options. A Guide for Patients
 Understanding Coronary Artery Disease, Cardiac Catheterization, and Treatment Options A Guide for Patients Coronary Artery Disease If you or a member of your family has been diagnosed with coronary artery
Understanding Coronary Artery Disease, Cardiac Catheterization, and Treatment Options A Guide for Patients Coronary Artery Disease If you or a member of your family has been diagnosed with coronary artery
Cardiac Assessment for Renal Transplantation: Pre-Operative Clearance is Only the Tip of the Iceberg
 Cardiac Assessment for Renal Transplantation: Pre-Operative Clearance is Only the Tip of the Iceberg 2 nd Annual Duke Renal Transplant Symposium March 1, 2014 Durham, NC Joseph G. Rogers, M.D. Associate
Cardiac Assessment for Renal Transplantation: Pre-Operative Clearance is Only the Tip of the Iceberg 2 nd Annual Duke Renal Transplant Symposium March 1, 2014 Durham, NC Joseph G. Rogers, M.D. Associate
Clinical Study Synopsis
 Clinical Study Synopsis This Clinical Study Synopsis is provided for patients and healthcare professionals to increase the transparency of Bayer's clinical research. This document is not intended to replace
Clinical Study Synopsis This Clinical Study Synopsis is provided for patients and healthcare professionals to increase the transparency of Bayer's clinical research. This document is not intended to replace
Journal Club: Niacin in Patients with Low HDL Cholesterol Levels Receiving Intensive Statin Therapy by the AIM-HIGH Investigators
 Journal Club: Niacin in Patients with Low HDL Cholesterol Levels Receiving Intensive Statin Therapy by the AIM-HIGH Investigators Shaikha Al Naimi Doctor of Pharmacy Student College of Pharmacy Qatar University
Journal Club: Niacin in Patients with Low HDL Cholesterol Levels Receiving Intensive Statin Therapy by the AIM-HIGH Investigators Shaikha Al Naimi Doctor of Pharmacy Student College of Pharmacy Qatar University
Pooled RESOLUTE Clinical Program
 The Relationship Between Short and Long-term Antiplatelet Therapy Use and Stent Thrombosis Following Percutaneous Coronary Intervention With the Resolute Zotarolimus-eluting Stent Pooled RESOLUTE Clinical
The Relationship Between Short and Long-term Antiplatelet Therapy Use and Stent Thrombosis Following Percutaneous Coronary Intervention With the Resolute Zotarolimus-eluting Stent Pooled RESOLUTE Clinical
Mechanical Properties of Nitinol Stents and Stent-grafts. Comparison of 6 mm Diameter Devices
 Mechanical Properties of Nitinol Stents and Stent-grafts Comparison of 6 mm Diameter Devices P E R F O R M A N C E b y d e s i g n Purpose The association of target vessel restenosis with stent fractures
Mechanical Properties of Nitinol Stents and Stent-grafts Comparison of 6 mm Diameter Devices P E R F O R M A N C E b y d e s i g n Purpose The association of target vessel restenosis with stent fractures
QT VASCULAR REPORTS 9M2015 REVENUE OF US$9.1M
 MEDIA RELEASE QT VASCULAR REPORTS 9M2015 REVENUE OF US$9.1M Highlights: Gross profit margin improved to 35.4% in 9M2015, up from 12.6% in 9M2014 Working capital position will strengthen with conversion
MEDIA RELEASE QT VASCULAR REPORTS 9M2015 REVENUE OF US$9.1M Highlights: Gross profit margin improved to 35.4% in 9M2015, up from 12.6% in 9M2014 Working capital position will strengthen with conversion
When Procedural Support really matters. Navien TM A+ Intracranial Support Catheter
 When Procedural Support really matters Navien TM A+ Intracranial Support Catheter Coils / Liquid Embolics DURABLE ovalization resistance, Maintains patent lumen in tortuosity for smooth micro catheter
When Procedural Support really matters Navien TM A+ Intracranial Support Catheter Coils / Liquid Embolics DURABLE ovalization resistance, Maintains patent lumen in tortuosity for smooth micro catheter
PATIENT INFORMATION BOOKLET
 PATIENT INFORMATION BOOKLET Wingspan Stent System with Gateway PTA Balloon Catheter TABLE OF CONTENTS Definitions... 2 What is the Purpose of This Booklet?... 3 What is an Intracranial Lesion?... 3 Who
PATIENT INFORMATION BOOKLET Wingspan Stent System with Gateway PTA Balloon Catheter TABLE OF CONTENTS Definitions... 2 What is the Purpose of This Booklet?... 3 What is an Intracranial Lesion?... 3 Who
Is There A LIfe for DES after discontinuation of Clopidogrel
 Chicago 2014 Is There A LIfe for DES after discontinuation of Clopidogrel Six-month versus 24-month dual antiplatelet therapy after implantation of drug eluting stents in patients non-resistant to aspirin:
Chicago 2014 Is There A LIfe for DES after discontinuation of Clopidogrel Six-month versus 24-month dual antiplatelet therapy after implantation of drug eluting stents in patients non-resistant to aspirin:
on behalf of the AUGMENT-HF Investigators
 One Year Follow-Up Results from AUGMENT-HF: A Multicenter Randomized Controlled Clinical Trial of the Efficacy of Left Ventricular Augmentation with Algisyl-LVR in the Treatment of Heart Failure* Douglas
One Year Follow-Up Results from AUGMENT-HF: A Multicenter Randomized Controlled Clinical Trial of the Efficacy of Left Ventricular Augmentation with Algisyl-LVR in the Treatment of Heart Failure* Douglas
Stroke: Major Public Health Burden. Stroke: Major Public Health Burden. Stroke: Major Public Health Burden 5/21/2012
 Faculty Prevention Sharon Ewer, RN, BSN, CNRN Stroke Program Coordinator Baptist Health Montgomery, Alabama Satellite Conference and Live Webcast Monday, May 21, 2012 2:00 4:00 p.m. Central Time Produced
Faculty Prevention Sharon Ewer, RN, BSN, CNRN Stroke Program Coordinator Baptist Health Montgomery, Alabama Satellite Conference and Live Webcast Monday, May 21, 2012 2:00 4:00 p.m. Central Time Produced
DEPARTMENT OF HEALTH & HUMAN SERVICES
 DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service April 26, 2016 Food and Drug Administration 10903 New Hampshire Avenue Document Control Center - WO66-G609 Silver Spring, MD 20993-0002 Bard
DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service April 26, 2016 Food and Drug Administration 10903 New Hampshire Avenue Document Control Center - WO66-G609 Silver Spring, MD 20993-0002 Bard
Understanding your Renal Stent Procedure. A patient Guide (COVER PAGE) TABLE OF CONTENTS (inside front page)
 Understanding your Renal Stent Procedure. A patient Guide (COVER PAGE) TABLE OF CONTENTS (inside front page) The Kidney and the Renal Arteries... 1 Renal Artery Disease... 2 Diagnosis of Renal.Artery Disease...
Understanding your Renal Stent Procedure. A patient Guide (COVER PAGE) TABLE OF CONTENTS (inside front page) The Kidney and the Renal Arteries... 1 Renal Artery Disease... 2 Diagnosis of Renal.Artery Disease...
SAMPLE. Asia-Pacific Interventional Cardiology Procedures Outlook to 2020. Reference Code: GDMECR0061PDB. Publication Date: May 2014
 Asia-Pacific Interventional Cardiology Procedures Outlook to 2020 Reference Code: GDMECR0061PDB Publication Date: May 2014 Page 1 1 Table of Contents 1 Table of Contents... 2 1.1 List of Tables... 4 1.2
Asia-Pacific Interventional Cardiology Procedures Outlook to 2020 Reference Code: GDMECR0061PDB Publication Date: May 2014 Page 1 1 Table of Contents 1 Table of Contents... 2 1.1 List of Tables... 4 1.2
Biliary and Endovascular Product Catalog
 Biliary and Endovascular Product Catalog To navigate within this catalog: - Single click on a chapter or section letter - Type the product name or catalog number in the Find box on the Adobe tool bar Table
Biliary and Endovascular Product Catalog To navigate within this catalog: - Single click on a chapter or section letter - Type the product name or catalog number in the Find box on the Adobe tool bar Table
OPTIMIZE: A Prospective, Randomized Trial of 3 Months Versus 12 Months of Dual Antiplatelet Therapy with the Endeavor Zotarolimus-Eluting Stent
 Main Arena III - Plenary Sessions XVI Late Breaking Clinical Trials III - Featured Trial of the Day: OPTIMIZE: A Prospective, Randomized Trial of 3 Months Versus 12 Months of Dual Antiplatelet Therapy
Main Arena III - Plenary Sessions XVI Late Breaking Clinical Trials III - Featured Trial of the Day: OPTIMIZE: A Prospective, Randomized Trial of 3 Months Versus 12 Months of Dual Antiplatelet Therapy
