Focus on PSA Screening for Prostate Cancer Vol. 28 Supplement, February Prostate Cancer: Should We Be Screening?
|
|
|
- Sydney Haynes
- 8 years ago
- Views:
Transcription
1 Focus on PSA Screening for Prostate Cancer Vol. 28 Supplement, February 2012 Prostate Cancer: Should We Be Screening? INSIDE THIS ISSUE 2 Why the Controversy? 3 Active Surveillance 4 The Radical Prostatectomy Registry at Mayo Clinic 4 Discovery of PSA and Evolution of PSA Screening 6 After a Diagnosis 7 Mayo Clinic Department of Urology Perspective on Prostate Cancer Screening 8 Related Articles Do you want to know? A simple blood test and digital rectal exam can help determine if a patient may have prostate cancer. Screening may well prevent death and morbidity from prostate cancer, but it will also detect many cancers that are not likely to threaten patients at all. That is the conundrum in which many men and their physicians find themselves with prostate cancer screening. Prostate-specific antigen (PSA) is a protein that is produced by the prostate. Small amounts of this protein can leak into the blood and be detected by a simple blood test. In the decades before the Food and Drug Administration (FDA) approved PSA testing, prostate cancer was the most common source of Acancer death in men, and even more men suffered from the complications of metastatic prostate cancer. Given this, the development of the PSA screening test was revolutionary, and the 1990s saw countless men diagnosed with and treated for prostate cancer. PSA screening caused many men to be diagnosed with prostate cancer at an earlier stage than ever before, which led to improved survival. But, doctors soon began to notice that although cure rates were improved, some men suffered serious long-term difficulties with urinary control and sexual function. Many doctors began to question the value of diagnosing and treating a cancer that was causing no symptoms. This led to the development of two large cancer-screening trials the Prostate, Lung, Colorectal and Ovarian (PLCO) Cancer Screening Trial and the European Randomized Study of Screening for Prostate Cancer (ERSPC) the early results of which are available (see Why the Controversy? on page 2). Despite randomizing more than 250,000 men and spending millions of research dollars, significant questions remain. It continues to be unclear whether the PSA blood test saves lives or whether it exposes men to unnecessary physical and emotional anguish. Currently, multiple medical associations and government task forces have issued recommendations regarding prostate cancer screening. These range from suggesting absolutely no screening to offering annual screening starting at age 40. In this special edition of Clinical Update, we explain some of the debate and controversy surrounding prostate cancer screening and provide recommendations for patients and physicians who are perplexed by the controversy. Matthew K. Tollefson, MD Urologic Surgeon, Department of Urology Medical Editor, Clinical Update: Special Edition Mayo Clinic Bladder Rectum Urethra Normal prostate Cancerous prostate Tumor Figure. Prostate cancer occurs in the prostate gland, which is located just below a male s bladder and surrounds the top portion of the urethra, the tube that drains urine from the bladder. This illustration shows a normal prostate gland and a prostate with a tumor.
2 Why the Controversy? Two Large Studies, No Clear Conclusions In 2009, early results from two major studies regarding prostate cancer screening were released in the New England Journal of Medicine. In these studies, a total of more than 250,000 men were randomized to screening and followed for prostate cancer death. These studies have formed the basis for most prostate cancer screening recommendations, yet they are commonly misunderstood. To better assess the implications of these studies, it s important to review study design, strengths and weaknesses in order to place them in context within the larger debate on prostate cancer screening. Prostate, Lung, Colorectal and Ovarian (PLCO) Cancer Screening Trial PLCO, a study based in the United States, randomized 76,693 men ages 55 to 74 to either annual screening with PSA and digital rectal examination or usual care. In this study, approximately 17% more cancers were detected in the screening arm than in the control arm, but there was no difference in cancer-specific death. Although technically randomized, a criticism of the study is that it doesn t appear to have been particularly well-controlled. Nearly two-thirds of men randomized in the study underwent PSA screening prior to entry into the study, a factor that undoubtedly eliminated high-risk cancers before the study even began. This limitation has led many to suggest that the Table. Comparing PLCO with ERSPC PLCO ERSPC Origin United States Europe Patients 76, ,000 Age range years years Randomization % screened before entering study Contamination (controls screened) Annual PSA and DRE vs. usual care Nearly 70% 52% 15% Median follow-up 7 years 9 years Increased chance of diagnosing prostate cancer with screening 17% 39% Outcome No significant difference in prostate cancer death PSA and DRE every 4 years vs. no screening Unknown, but likely very small 20% reduction in prostate cancer death (increasing with time) trial was destined to fail from the start. Further, only 85% of men in the screening group were compliant with screening, while 52% of men randomized to the control arm underwent screening as a part of usual care. And finally, only 31% of men with an abnormal digital rectal examination and a PSA of more than 4 ng/ml underwent prostate biopsy. These data call into question the reliability of the study s findings the screening and control groups essentially blended together, reducing the study s ability to detect a difference between the two groups. European Randomized Study of Screening for Prostate Cancer (ERSPC) The ERSPC trial was a randomized screening study based in Europe. In this study, 182,000 men were randomized to be either controls (no screening) or receive screening with digital rectal exam and PSA every four years. In this study, 39% more cancers were detected in men in the screening group than the control group. Further, there was a 20-31% reduction in prostate cancer death in the screening arm compared with the control arm. ERSPC suffers from fewer of the limitations of PLCO. Specifically, very few men were screened prior to entry into the study and contamination of the control group was significantly lower at 15%. Compliance with biopsy recommendation was also much higher at 85%. However, ERSPC has been criticized for lack of informed consent in many of the countries, and many more screened men opted for active surveillance in ERSPC than in PLCO (18% vs. 11%). Finally, although there was a significant benefit to screening, in order to save one life after eight years, nearly 1,400 men needed to be screened and 48 men treated. It should be noted, though, that with longer follow-up and further prostate cancer-related deaths, the number of men needed to be screened and treated to prevent one death will very likely decrease. Practical considerations While ERSPC and PLCO both had similar endpoints and objectives, the manner in which the studies were performed and their interpretation were widely divergent. Nonetheless, there are important, complementary data that emerged from both trials. 2 MAYO CLINIC ClinicalUpdate
3 First, prostate cancer investigators now have a much better understanding of PSA dynamics on a population scale. This includes the complicated issues surrounding contamination and the difficulties in performing large studies where researchers are trying to obtain something as simple as a serum PSA. Also, both studies have demonstrated that men with a very low PSA (0-1.0 ng/ml) are at low risk for developing a clinically significant prostate cancer within the next several years. The diversity of methodology and data from these trials allows for significant flexibility in their interpretation, which makes it difficult to use them to substantiate across-the-board recommendations. Rather, the decision of whether to screen or not screen using PSA testing and/or other means is a decision best made between physicians and their individual patients. In making this decision, both physicians and their patients should be informed of the benefits and risks of screening or not screening. Other clinical factors, such as age, comorbidities, 10-year life expectancy and patient preferences, should also be taken into account. By being fully informed, patients and physicians are better armed to combat prostate cancer, says Erik P. Castle, MD, a urologic surgeon at Mayo Clinic s campus in Arizona. References Andriole GL, et al. Mortality results from a randomized prostatecancer screening trial. New England Journal of Medicine. 2009;360:1310. Andriole GL, et al. Prostate cancer screening in the randomized Prostate, Lung, Colorectal and Ovarian Cancer Screening Trial: Mortality results after 13 years of follow-up. Journal of the National Cancer Institute. 2012;104:125. Schroder FH, et al. Screening and prostate-cancer mortality in a randomized European study. New England Journal of Medicine. 2009;360:1320. Active Surveillance Does the Tumor Need Treatment? A growing number of men are being diagnosed with low-risk prostate cancer. For these men, the best initial treatment may be no treatment at all. Active surveillance is a treatment approach that recognizes the tumor as a long-term and likely slow-growing disease. An active surveillance approach may help select patients avoid potential complications from aggressive treatments that might negatively affect their current quality of life, explains Michael J. Wehle, MD, a urologic surgeon at Mayo Clinic s campus in Florida. At the same time, it keeps the door open for treatment at a later date if necessary. Each patient needs to be evaluated carefully to see if he s a candidate for the active surveillance approach. Active surveillance does not mean simply forgetting about prostate cancer. Rather, it involves closely following the tumor with regularly scheduled PSA tests and prostate biopsies. Given that the majority of men with low-risk prostate cancer will not have their cancer spread outside the prostate for many years, this approach enables doctors to follow the cancer to see if it shows any fast-growing characteristics. If the tumor should start to grow faster than expected, curative treatment usually radiation or surgery should be offered. On the other hand, if the tumor appears to be indolent, any potential side effects associated with treatment can be avoided. Using this approach, many men have successfully avoided cancer progression and treatment side effects for more than a decade. Best candidates Active surveillance is not for everyone, but it s an option that should be discussed with and considered for all patients with low-risk disease. Decision-making criteria include both PSA levels and biopsy results. The best candidates are patients with Low PSA levels Nonaggressive prostate cancer (as determined by biopsy) Small amounts of cancer (as determined by biopsy) At Mayo Clinic, patients who choose active surveillance are being followed longterm in a prospective database to see which tumors needed treatment and how successful that treatment has been. In addition, blood, urine and tissue samples are collected from these patients, which will be used to study new biomarkers that may help identify the tumors that are truly indolent. Urethra Bladder Cancerous prostate Tumor Rectum Figure. This illustration shows a prostate gland with a tumor. MAYO CLINIC ClinicalUpdate 3
4 The Radical Prostatectomy Registry at Mayo Clinic Translating Today s Surgery into Tomorrow s Discoveries Since 1985, Mayo Clinic has been collecting and storing clinical, pathological and follow-up data on men who have undergone radical prostatectomies at its campus in Rochester, Minn. Because Mayo Clinic is such a high-volume urologic surgery center on average over the past five years, urologic surgeons at its three campuses have performed an annual total of more than 1,600 prostatectomies it s possible to estimate morbidity and mortality associated with prostatectomy, monitor long-term outcomes, and investigate the best predictors of outcome. Today, Mayo Clinic s radical prostatectomy registry includes more than 20,000 patients, making it one of the largest registries of its kind in the world. More than 18,000 of these patients were seen from 1987 and onward the period during which PSA screening has been standard practice at Mayo. The other several thousand patients were seen at Mayo between 1966 and 1986; their data were retrospectively added to the registry when it was established. Abstractors prospectively maintain the registry by following up annually with patients after surgery (Table, on page 5). In addition to these data, tissue samples are also collected at the time of surgery, enabling researchers to study potential biomarkers and associate them with clinical outcomes. Contributing to this research are not only Mayo urologic surgeons, but also Mayo investigators with expertise in molecular biology, bioinformatics, pathology and epidemiology. A diverse team of biostatisticians, postdoctoral fellows and many others also support this research and its translation into clinical applications. Insights from the registry Since the introduction of PSA screening, Mayo has seen fewer cases of lymph node positive prostate cancer. Prostate cancer screening practices, which include the use of the PSA test when appropriate, enable detection of early-stage, treatable prostate cancer. Without judicious use of the PSA test, it s possible that clinically localized prostate cancer would not have been detected until it had metastasized; such cases are rarely considered curable (Figure, on page 6). Discovery of PSA The discovery and characterization of prostate-specific antigen (PSA), a protein produced primarily by the prostate gland, came about during the course of research related to cancer, forensic science and infertility Dr. Richard Ablin and colleagues find an uncharacterized antigen that appears to be specific to the prostate Dr. Mitsuwo Hara and colleagues discover gammaseminoprotein, another prostatespecific protein, in seminal plasma Drs. Tien Shun Li and Carl Beling isolate antigens known as E1 and E Dr. George Sensabaugh identifies a protein called p A team including Drs. Tsann Ming Chu and Ming Wang is the first to purify a prostate tissue-specific antigen. It s later shown to be the same as gamma-seminoprotein and p30. Evolution of PSA Screening Although it took several years after its introduction for researchers to demonstrate its utility as a screening tool, the PSA test has been available for 25 years Drs. Alexander Gutman and Ethel Gutman find that levels of prostatic acid phosphatase are elevated in people with metastatic prostate cancer. 1950s-1980s Prostatic acid phosphatase testing is used for prostate cancer detection, staging and treatment-response monitoring. The test has low sensitivity, particularly for early-stage prostate cancer. 4 MAYO CLINIC ClinicalUpdate
5 Table. Registry Variables and Data Data collected and recorded at time of surgery Demographics Preoperative PSA value Clinical stage Biopsy grade Pathological stage and grade Presurgery therapy Margin positivity Dimensions/volume of largest tumor Capsular perforation Seminal vesicle involvement Number of nodes involved DNA ploidy Data collected and recorded after surgery Use of adjuvant therapy (within 90 days of surgery) Use of salvage therapy (beyond 90 days of surgery) Complications, including potency and continence, at 30 days and 1 year after surgery Potency and continence at 30 days and 1 year after surgery Presence of elevated PSA value (biochemical recurrence) after surgery Occurrence of local or systemic clinical progression Mortality/cause of death After radical prostatectomy, prostate cancer recurs in only a small percentage of men. The vast majority of men tracked in the registry remain cancer-free. An analysis of 12,000 cases of radical prostatectomy from 1987 through 2004 shows systemic recurrence rates of 5.6% after 10 years and 8.2% after 15 years, with prostate cancer death rates of 2.9% after 10 years and 5.3% after 15 years. These outcomes are markedly better than the cure rates seen before PSA screening, supporting the sensible use of the PSA test. Continued on page Dr. Lawrence Papsidero and colleagues discover that PSA can be detected in the serum of men with prostate cancer, leading to the first PSA screening test Dr. Thomas Stamey and colleagues publish findings showing that serum PSA level is a marker for prostate cancer. Information in this timeline was gathered from various sources, including articles published in the British Journal of Urology International and Reviews in Urology Serum PSA testing is introduced and FDA approves it for monitoring prostate cancer recurrence after treatment, leading to reduced reliance on the prostatic acid phosphatase test Dr. William Catalona and colleagues show that PSA testing in addition to DRE is superior for prostate cancer detection than DRE alone Prostate cancer deaths peak in U.S FDA approves the PSA test as a method for prostate cancer screening U.S. Preventive Services Task Force recommends that healthy men no longer undergo PSA testing. MAYO CLINIC ClinicalUpdate 5
6 The Radical Prostatectomy Registry at Mayo Clinic Translating Today s Surgery into Tomorrow s Discoveries Continued from page 5 Percentage Incidence of Node-Positive Prostate Cancer as a Percentage of Total Diagnoses Figure. From 1988 to 2009, there has been a decline in the number of men who had node-positive prostate cancer at their time of surgery at Mayo Clinic. Source: Mayo Clinic radical prostatectomy registry Long-term side effects after surgery are rare. Multiple studies have demonstrated that high-volume centers have improved outcomes after radical prostatectomy. Mayo data from 1987 through 2004 show that few patients have severe urinary incontinence after surgery; less than 0.2% have gone on to have additional surgery for problematic leakage within one year. Furthermore, when preservation of nerve bundles is possible, most men are capable of erectile function after surgery with or without assistance. Year Looking ahead Using patient tissue samples linked to data from the radical prostatectomy registry, Mayo clinicians and researchers are investigating new diagnostic and prognostic molecular markers for prostate cancer. Such research is likely to lead to More reliable diagnostic tools. The best screening and diagnostic tools available today a combination of the PSA test, digital rectal examinations and core needle biopsies miss some clinically significant cancers. Further biomarker research will hopefully enable the development of a highly sensitive, highly specific, potentially noninvasive diagnostic test that can more reliably and effectively detect significant prostate cancer, says R. Jeffrey Karnes, MD, a urologic surgeon at Mayo Clinic s campus in Rochester. A better ability to personalize therapy. Separating cases of indolent cancer and aggressive cancer remains a clinical challenge. At Mayo Clinic, research based on data from the radical prostatectomy registry has already led to the identification of several promising biomarkers that are likely to prove useful in identifying patients with life-threatening prostate cancer. More aggressive treatment may be indicated for patients with high-risk cancers, while active surveillance may be sufficient for those with insignificant cancers. After a Diagnosis What Next? Prostate cancer is usually a slow-growing disease. Unlike some other cancers, prostate cancer rarely requires immediate treatment. This means the physician and the patient have time to thoroughly evaluate the many available treatment options, which include surgery, radiation therapy, hormone therapy and other interventions. Depending on the patient s overall health and disease stage, no treatment may be advisable (see Active Surveillance on page 3). A consultation with a urologist who practices within a large academic medical system is a good next step. As prostate cancer surgery and other therapies are continually evolving, this provides the patient with access to the widest array of evidence-based treatment options and highly trained surgeons. These centers also have the most experience with monitoring after surgery and, should it be needed, follow-up treatment. 6 MAYO CLINIC ClinicalUpdate
7 Mayo Clinic Department of Urology Perspective on Prostate Cancer Screening Balancing Risks and Benefits Screening for prostate cancer is a complex and ultimately personal decision it positions the hopeful prevention of prostate cancer death and morbidity against the possibility of treating an insignificant tumor. That said, most experts agree that prostate cancer is a serious disease and that screening is rather straightforward and detects high-risk prostate cancer. The debate about prostate cancer screening centers around the fact that it inevitably results in diagnosing some patients with indolent tumors that may never have become clinically significant. The ideal cancer-screening test would be one that is inexpensive and easy to perform, detects clinically significant cancers with high sensitivity and fails to detect slowgrowing, indolent tumors. Unfortunately, such a test isn t available today. This means that practitioners must rely on the best available options: PSA screening, digital rectal examinations and prostate biopsies. It is important to recognize that the benefit of screening is critically tied to downstream factors such as treatment success and morbidity. The identification of a slow-growing prostate cancer should not be viewed as a failure of prostate cancer screening. Rather, it should represent an opportunity to discuss appropriate treatment options and select the right management approach for each patient. Active surveillance has emerged as a preferred treatment option for many patients who are diagnosed with low-risk prostate cancer. For men who need treatment, advances are continuing to reduce the morbidity associated with surgery and other interventions. Prostate cancer surveillance, including the sensible use of the PSA test, reduces the morbidity and mortality associated with this disease. What remains to be determined is how often and at what time such screening needs to be performed to maximize its effectiveness and minimize adverse effects. Individualized screening approach The Department of Urology at Mayo Clinic recommends an individualized, multifactorial approach to determining whether or not to screen a particular patient for prostate cancer. Points to Remember The purpose of prostate cancer screening with the PSA test is to detect prostate cancer when it is localized and therefore most treatable. Prostate cancer screening is controversial because no trials have conclusively demonstrated that the benefits of screening outweigh its risks. The majority of prostate cancers detected with screening are low-risk and not immediately life-threatening. While prostate cancer screening identifies most prostate cancers, it does not discriminate between high-risk, life-threatening tumors and low-risk, indolent tumors. The death rate from prostate cancer has consistently declined approximately 4% per year since prostate cancer screening became commonplace. This approach, which should begin at age 40, incorporates an individual patient s risk of developing prostate cancer, a comprehensive physical examination, consideration of existing medical comorbidities, and a discussion regarding the benefits and potential risks of screening. When determining whether to screen: Discuss with the patient the pros and cons of screening Conduct a physical exam, staying alert for comorbidities that can affect PSA score Take a comprehensive family medical history, noting previous biopsy history of the patient and his family members Consider the patient s age, recognizing the age-related increase in cancer risk Consider the patient s ethnic background, noting that African-American men have the highest risk of prostate cancer MAYO CLINIC ClinicalUpdate 7
8 Mayo Clinic Clinical Update Medical Editor: Matthew K. Tollefson, MD Editorial Board: Paul E. Andrews, MD Douglas A. Husmann, MD Todd C. Igel, MD R. Jeffrey Karnes, MD Clinical Update is written for physicians and should be relied upon for medical education purposes only. It does not provide a complete overview of the topics covered and should not replace the independent judgment of a physician about the appropriateness or risks of a procedure for a given patient. Contact Us Mayo Clinic welcomes inquiries and referrals, and a request to a specific physician is not required to refer a patient. Arizona Florida Minnesota Resources mayoclinic.org/medicalprofs Related Articles Andriole GL, et al. Mortality results from a randomized prostate-cancer screening trial. New England Journal of Medicine. 2009;360:1310. Andriole GL, et al. Prostate cancer screening in the randomized Prostate, Lung, Colorectal and Ovarian Cancer Screening Trial: Mortality results after 13 years of follow-up. Journal of the National Cancer Institute. 2012;104:125. Boorjian SA, et al. Long-term survival after radical prostatectomy versus external-beam radiotherapy for patients with high-risk prostate cancer. Cancer. 2011;117:2883. Dwyer ME, Nehra A. Defining sexual function after radical retropubic prostatectomy. Urologic Oncology. 2010;28:469. Karnes RJ, et al. The ability of biomarkers to predict systemic progression in men with high-risk prostate cancer treated surgically is dependent on ERG status. Cancer Research. 2010;70:8994. Klein EA, et al. Vitamin E and the risk of prostate cancer: The Selenium and Vitamin E Cancer Prevention Trial (SELECT). Journal of the American Medical Association. 2011;306:1549. Mitchell CR, et al. Does body mass index dilute the predictive property of prostate-specific antigen for tumor volume at radical prostatectomy? Urology. 2011;78:868. Schroder FH, et al. Screening and prostate-cancer mortality in a randomized European study. New England Journal of Medicine. 2009;360:1320. Tollefson MK, et al. Lifelong yearly prostate-specific antigen surveillance is not necessary for low-risk prostate cancer treated with radical prostatectomy. The Journal of Urology. 2010;184:925. Tollefson MK, et al. Robotic-assisted radical prostatectomy decreases the incidence and morbidity of surgical site infections. Urology. 2011;78:827. Tollefson MK, et al. The effect of Gleason score on the predictive value of prostate-specific antigen doubling time. British Journal of Urology International. 2010;105:1381. Woodrum DA, et al. 3.0T MR-guided laser ablation of a prostate cancer recurrence in the postsurgical prostate bed. Journal of Vascular and Interventional Radiology. 2011;22:929. Clinical trials CME Grand Rounds Scientific videos Online referrals MC
PSA Screening for Prostate Cancer Information for Care Providers
 All men should know they are having a PSA test and be informed of the implications prior to testing. This booklet was created to help primary care providers offer men information about the risks and benefits
All men should know they are having a PSA test and be informed of the implications prior to testing. This booklet was created to help primary care providers offer men information about the risks and benefits
1. What is the prostate-specific antigen (PSA) test?
 1. What is the prostate-specific antigen (PSA) test? Prostate-specific antigen (PSA) is a protein produced by the cells of the prostate gland. The PSA test measures the level of PSA in the blood. The doctor
1. What is the prostate-specific antigen (PSA) test? Prostate-specific antigen (PSA) is a protein produced by the cells of the prostate gland. The PSA test measures the level of PSA in the blood. The doctor
Early Prostate Cancer: Questions and Answers. Key Points
 CANCER FACTS N a t i o n a l C a n c e r I n s t i t u t e N a t i o n a l I n s t i t u t e s o f H e a l t h D e p a r t m e n t o f H e a l t h a n d H u m a n S e r v i c e s Early Prostate Cancer:
CANCER FACTS N a t i o n a l C a n c e r I n s t i t u t e N a t i o n a l I n s t i t u t e s o f H e a l t h D e p a r t m e n t o f H e a l t h a n d H u m a n S e r v i c e s Early Prostate Cancer:
PSA Testing 101. Stanley H. Weiss, MD. Professor, UMDNJ-New Jersey Medical School. Director & PI, Essex County Cancer Coalition. weiss@umdnj.
 PSA Testing 101 Stanley H. Weiss, MD Professor, UMDNJ-New Jersey Medical School Director & PI, Essex County Cancer Coalition weiss@umdnj.edu September 23, 2010 Screening: 3 tests for PCa A good screening
PSA Testing 101 Stanley H. Weiss, MD Professor, UMDNJ-New Jersey Medical School Director & PI, Essex County Cancer Coalition weiss@umdnj.edu September 23, 2010 Screening: 3 tests for PCa A good screening
Thomas A. Kollmorgen, M.D. Oregon Urology Institute
 Thomas A. Kollmorgen, M.D. Oregon Urology Institute None 240,000 new diagnosis per year, and an estimated 28,100 deaths (2012) 2 nd leading cause of death from cancer in U.S.A. Approximately 1 in 6 men
Thomas A. Kollmorgen, M.D. Oregon Urology Institute None 240,000 new diagnosis per year, and an estimated 28,100 deaths (2012) 2 nd leading cause of death from cancer in U.S.A. Approximately 1 in 6 men
Oncology Annual Report: Prostate Cancer 2005 Update By: John Konefal, MD, Radiation Oncology
 Oncology Annual Report: Prostate Cancer 25 Update By: John Konefal, MD, Radiation Oncology Prostate cancer is the most common cancer in men, with 232,9 new cases projected to be diagnosed in the U.S. in
Oncology Annual Report: Prostate Cancer 25 Update By: John Konefal, MD, Radiation Oncology Prostate cancer is the most common cancer in men, with 232,9 new cases projected to be diagnosed in the U.S. in
7. Prostate cancer in PSA relapse
 7. Prostate cancer in PSA relapse A patient with prostate cancer in PSA relapse is one who, having received a primary treatment with intent to cure, has a raised PSA (prostate-specific antigen) level defined
7. Prostate cancer in PSA relapse A patient with prostate cancer in PSA relapse is one who, having received a primary treatment with intent to cure, has a raised PSA (prostate-specific antigen) level defined
Prostate Cancer Screening. A Decision Guide
 Prostate Cancer Screening A Decision Guide This booklet was developed by the U.S. Department of Health and Human Services, Centers for Disease Control and Prevention (CDC). Is screening right for you?
Prostate Cancer Screening A Decision Guide This booklet was developed by the U.S. Department of Health and Human Services, Centers for Disease Control and Prevention (CDC). Is screening right for you?
PSA screening in asymptomatic men the debate continues www.bpac.org.nz keyword: psa
 PSA screening in asymptomatic men the debate continues www.bpac.org.nz keyword: psa Key messages: PSA is present in the benign and malignant prostate There is currently no national screening programme
PSA screening in asymptomatic men the debate continues www.bpac.org.nz keyword: psa Key messages: PSA is present in the benign and malignant prostate There is currently no national screening programme
These rare variants often act aggressively and may respond differently to therapy than the more common prostate adenocarcinoma.
 Prostate Cancer OVERVIEW Prostate cancer is the second most common cancer diagnosed among American men, accounting for nearly 200,000 new cancer cases in the United States each year. Greater than 65% of
Prostate Cancer OVERVIEW Prostate cancer is the second most common cancer diagnosed among American men, accounting for nearly 200,000 new cancer cases in the United States each year. Greater than 65% of
Cancer in Primary Care: Prostate Cancer Screening. How and How often? Should we and in which patients?
 Cancer in Primary Care: Prostate Cancer Screening How and How often? Should we and in which patients? PLCO trial (Prostate, Lung, Colorectal and Ovarian) Results In the screening group, rates of compliance
Cancer in Primary Care: Prostate Cancer Screening How and How often? Should we and in which patients? PLCO trial (Prostate, Lung, Colorectal and Ovarian) Results In the screening group, rates of compliance
Screening for Prostate Cancer
 Screening for Prostate Cancer It is now clear that screening for Prostate Cancer discovers the disease at an earlier and more curable stage. It is not yet clear whether this translates into reduced mortality
Screening for Prostate Cancer It is now clear that screening for Prostate Cancer discovers the disease at an earlier and more curable stage. It is not yet clear whether this translates into reduced mortality
Prostate Cancer Screening. A Decision Guide for African Americans
 Prostate Cancer Screening A Decision Guide for African Americans This booklet was developed by the U.S. Department of Health and Human Services, Centers for Disease Control and Prevention (CDC). Published
Prostate Cancer Screening A Decision Guide for African Americans This booklet was developed by the U.S. Department of Health and Human Services, Centers for Disease Control and Prevention (CDC). Published
Official reprint from UpToDate www.uptodate.com 2013 UpToDate
 Official reprint from UpToDate www.uptodate.com 2013 UpToDate The content on the UpToDate website is not intended nor recommended as a substitute for medical advice, diagnosis, or treatment. Always seek
Official reprint from UpToDate www.uptodate.com 2013 UpToDate The content on the UpToDate website is not intended nor recommended as a substitute for medical advice, diagnosis, or treatment. Always seek
Analysis of Prostate Cancer at Easter Connecticut Health Network Using Cancer Registry Data
 The 2014 Cancer Program Annual Public Reporting of Outcomes/Annual Site Analysis Statistical Data from 2013 More than 70 percent of all newly diagnosed cancer patients are treated in the more than 1,500
The 2014 Cancer Program Annual Public Reporting of Outcomes/Annual Site Analysis Statistical Data from 2013 More than 70 percent of all newly diagnosed cancer patients are treated in the more than 1,500
Prostate Cancer Screening
 Prostate Cancer Screening The American Cancer Society and Congregational Health Ministry Team June Module To access this module via the Web, visit www.cancer.org and type in congregational health ministry
Prostate Cancer Screening The American Cancer Society and Congregational Health Ministry Team June Module To access this module via the Web, visit www.cancer.org and type in congregational health ministry
An Introduction to PROSTATE CANCER
 An Introduction to PROSTATE CANCER Being diagnosed with prostate cancer can be a life-altering experience. It requires making some very difficult decisions about treatments that can affect not only the
An Introduction to PROSTATE CANCER Being diagnosed with prostate cancer can be a life-altering experience. It requires making some very difficult decisions about treatments that can affect not only the
Prostate Cancer Screening in Taiwan: a must
 Prostate Cancer Screening in Taiwan: a must 吳 俊 德 基 隆 長 庚 醫 院 台 灣 醫 學 會 105 th What is the PSA test? The blood level of PSA is often elevated in men with prostate cancer, and the PSA test was originally
Prostate Cancer Screening in Taiwan: a must 吳 俊 德 基 隆 長 庚 醫 院 台 灣 醫 學 會 105 th What is the PSA test? The blood level of PSA is often elevated in men with prostate cancer, and the PSA test was originally
Prostate Cancer. Treatments as unique as you are
 Prostate Cancer Treatments as unique as you are UCLA Prostate Cancer Program Prostate cancer is the second most common cancer among men. The UCLA Prostate Cancer Program brings together the elements essential
Prostate Cancer Treatments as unique as you are UCLA Prostate Cancer Program Prostate cancer is the second most common cancer among men. The UCLA Prostate Cancer Program brings together the elements essential
HEALTH NEWS PROSTATE CANCER THE PROSTATE
 HEALTH NEWS PROSTATE CANCER THE PROSTATE Prostate comes from the Greek meaning to stand in front of ; this is very different than prostrate which means to lie down flat. The prostate is a walnut-sized
HEALTH NEWS PROSTATE CANCER THE PROSTATE Prostate comes from the Greek meaning to stand in front of ; this is very different than prostrate which means to lie down flat. The prostate is a walnut-sized
FAQ About Prostate Cancer Treatment and SpaceOAR System
 FAQ About Prostate Cancer Treatment and SpaceOAR System P. 4 Prostate Cancer Background SpaceOAR Frequently Asked Questions (FAQ) 1. What is prostate cancer? The vast majority of prostate cancers develop
FAQ About Prostate Cancer Treatment and SpaceOAR System P. 4 Prostate Cancer Background SpaceOAR Frequently Asked Questions (FAQ) 1. What is prostate cancer? The vast majority of prostate cancers develop
Prostate cancer screening. It s YOUR decision!
 Prostate cancer screening It s YOUR decision! For many years now, a test has been available to screen for. The test is called the prostate-specific antigen blood test (or PSA test). It is used in combination
Prostate cancer screening It s YOUR decision! For many years now, a test has been available to screen for. The test is called the prostate-specific antigen blood test (or PSA test). It is used in combination
PSA Testing for Prostate Cancer An information sheet for men considering a PSA Test
 PSA Testing for Prostate Cancer An information sheet for men considering a PSA Test What is the aim of this leaflet? Prostate cancer is a serious condition. The PSA test, which can give an early indication
PSA Testing for Prostate Cancer An information sheet for men considering a PSA Test What is the aim of this leaflet? Prostate cancer is a serious condition. The PSA test, which can give an early indication
4/8/13. Pre-test Audience Response. Prostate Cancer 2012. Screening and Treatment of Prostate Cancer: The 2013 Perspective
 Pre-test Audience Response Screening and Treatment of Prostate Cancer: The 2013 Perspective 1. I do not offer routine PSA screening, and the USPSTF D recommendation will not change my practice. 2. In light
Pre-test Audience Response Screening and Treatment of Prostate Cancer: The 2013 Perspective 1. I do not offer routine PSA screening, and the USPSTF D recommendation will not change my practice. 2. In light
Beyond the PSA: Genomic Testing in Localized Prostate Cancer
 Beyond the PSA: Genomic Testing in Localized Prostate Cancer Kelvin A. Moses, MD, PhD Vanderbilt University Medical Center Wednesday, December 2, 2015 5:00 p.m. ET/2:00 p.m. PT About ZERO ZERO s mission
Beyond the PSA: Genomic Testing in Localized Prostate Cancer Kelvin A. Moses, MD, PhD Vanderbilt University Medical Center Wednesday, December 2, 2015 5:00 p.m. ET/2:00 p.m. PT About ZERO ZERO s mission
Treating Prostate Cancer
 Treating Prostate Cancer A Guide for Men With Localized Prostate Cancer Most men have time to learn about all the options for treating their prostate cancer. You have time to talk with your family and
Treating Prostate Cancer A Guide for Men With Localized Prostate Cancer Most men have time to learn about all the options for treating their prostate cancer. You have time to talk with your family and
Cancer research in the Midland Region the prostate and bowel cancer projects
 Cancer research in the Midland Region the prostate and bowel cancer projects Ross Lawrenson Waikato Clinical School University of Auckland MoH/HRC Cancer Research agenda Lung cancer Palliative care Prostate
Cancer research in the Midland Region the prostate and bowel cancer projects Ross Lawrenson Waikato Clinical School University of Auckland MoH/HRC Cancer Research agenda Lung cancer Palliative care Prostate
TO SCREEN OR NOT TO SCREEN: THE PROSTATE CANCER
 TO SCREEN OR NOT TO SCREEN: THE PROSTATE CANCER DILEMMA Thomas J Stormont MD January 2012 http://www.youtube.com/watch?v=8jd 7bAHVp0A&feature=related related INTRODUCTION A government health panel (the
TO SCREEN OR NOT TO SCREEN: THE PROSTATE CANCER DILEMMA Thomas J Stormont MD January 2012 http://www.youtube.com/watch?v=8jd 7bAHVp0A&feature=related related INTRODUCTION A government health panel (the
The PSA Test for Prostate Cancer Screening:
 For more information, please contact your local VA Medical Center or Health Clinic. U.S. Department of Veterans Affairs Veterans Health Administration Patient Care Services Health Promotion and Disease
For more information, please contact your local VA Medical Center or Health Clinic. U.S. Department of Veterans Affairs Veterans Health Administration Patient Care Services Health Promotion and Disease
A Woman s Guide to Prostate Cancer Treatment
 A Woman s Guide to Prostate Cancer Treatment Supporting the man in your life Providing prostate cancer support and resources for women and families WOMEN AGAINST PROSTATE CANCER A Woman s Guide to Prostate
A Woman s Guide to Prostate Cancer Treatment Supporting the man in your life Providing prostate cancer support and resources for women and families WOMEN AGAINST PROSTATE CANCER A Woman s Guide to Prostate
Bard: Prostate Cancer Treatment. Bard: Pelvic Organ Prolapse. Prostate Cancer. An overview of. Treatment. Prolapse. Information and Answers
 Bard: Prostate Cancer Treatment Bard: Pelvic Organ Prolapse Prostate Cancer An overview of Pelvic Treatment Organ Prolapse Information and Answers A Brief Overview Prostate Anatomy The prostate gland,
Bard: Prostate Cancer Treatment Bard: Pelvic Organ Prolapse Prostate Cancer An overview of Pelvic Treatment Organ Prolapse Information and Answers A Brief Overview Prostate Anatomy The prostate gland,
PROSTATE CANCER. Get the facts, know your options. Samay Jain, MD, Assistant Professor,The University of Toledo Chief, Division of Urologic Oncology
 PROSTATE CANCER Get the facts, know your options Samay Jain, MD, Assistant Professor,The University of Toledo Chief, Division of Urologic Oncology i What is the Prostate? Unfortunately, you have prostate
PROSTATE CANCER Get the facts, know your options Samay Jain, MD, Assistant Professor,The University of Toledo Chief, Division of Urologic Oncology i What is the Prostate? Unfortunately, you have prostate
Prostate cancer. Christopher Eden. The Royal Surrey County Hospital, Guildford & The Hampshire Clinic, Old Basing.
 Prostate cancer Christopher Eden The Royal Surrey County Hospital, Guildford & The Hampshire Clinic, Old Basing. Screening Screening men for PCa (prostate cancer) using PSA (Prostate Specific Antigen blood
Prostate cancer Christopher Eden The Royal Surrey County Hospital, Guildford & The Hampshire Clinic, Old Basing. Screening Screening men for PCa (prostate cancer) using PSA (Prostate Specific Antigen blood
Us TOO University Presents: Understanding Diagnostic Testing
 Us TOO University Presents: Understanding Diagnostic Testing for Prostate Cancer Patients Today s speaker is Manish Bhandari, MD Program moderator is Pam Barrett, Us TOO International Made possible by
Us TOO University Presents: Understanding Diagnostic Testing for Prostate Cancer Patients Today s speaker is Manish Bhandari, MD Program moderator is Pam Barrett, Us TOO International Made possible by
Prostate Cancer Screening Guideline
 Prostate Cancer Screening Guideline Contents Prevention 2 Screening Recommendations 2 Shared Decision Making 2 Test Recommendations 3 Follow-up/Referral 3 Treatment Overview 4 Evidence Summary 4 References
Prostate Cancer Screening Guideline Contents Prevention 2 Screening Recommendations 2 Shared Decision Making 2 Test Recommendations 3 Follow-up/Referral 3 Treatment Overview 4 Evidence Summary 4 References
CMScript. Member of a medical scheme? Know your guaranteed benefits! Issue 7 of 2014
 Background CMScript Member of a medical scheme? Know your guaranteed benefits! Issue 7 of 2014 Prostate cancer is second only to lung cancer as the leading cause of cancer-related deaths in men. It is
Background CMScript Member of a medical scheme? Know your guaranteed benefits! Issue 7 of 2014 Prostate cancer is second only to lung cancer as the leading cause of cancer-related deaths in men. It is
Treating Localized Prostate Cancer A Review of the Research for Adults
 Treating Localized Prostate Cancer A Review of the Research for Adults Is this information right for me? Yes, this information is right for you if: Your doctor * said all tests show you have localized
Treating Localized Prostate Cancer A Review of the Research for Adults Is this information right for me? Yes, this information is right for you if: Your doctor * said all tests show you have localized
Update on Prostate Cancer Screening Guidelines
 www.medscape.com Update on Prostate Cancer Screening Guidelines Christine Gonzalez, PharmD, CHHC US Pharmacist Abstract and Introduction Introduction In the United States, prostate cancer is the most common
www.medscape.com Update on Prostate Cancer Screening Guidelines Christine Gonzalez, PharmD, CHHC US Pharmacist Abstract and Introduction Introduction In the United States, prostate cancer is the most common
Prostate Cancer Screening: Are We There Yet? March 2010 Andrew M.D. Wolf, MD University of Virginia School of Medicine
 Prostate Cancer Screening: Are We There Yet? March 2010 Andrew M.D. Wolf, MD University of Virginia School of Medicine Case #1 A 55 yo white man with well-controlled hypertension presents for his annual
Prostate Cancer Screening: Are We There Yet? March 2010 Andrew M.D. Wolf, MD University of Virginia School of Medicine Case #1 A 55 yo white man with well-controlled hypertension presents for his annual
Report with statistical data from 2007
 2008 Cancer Program Annual Report with statistical data from 2007 Lake Cumberland Regional Hospital 305 Langdon Streett Somerset, KY 42503 Telephone: 606-679-7441 Fax: 606-678-9919 Cancer Committee Mullai,
2008 Cancer Program Annual Report with statistical data from 2007 Lake Cumberland Regional Hospital 305 Langdon Streett Somerset, KY 42503 Telephone: 606-679-7441 Fax: 606-678-9919 Cancer Committee Mullai,
The PSA Controversy: Defining It, Discussing It, and Coping With It
 The PSA Controversy: Defining It, Discussing It, and Coping With It 11 TH ANNUAL SYMPOSIUM ON MEN S HEALTH June 12, 2013 The PSA Controversy Defining It, Discussing It and Coping With It As of May 2012,
The PSA Controversy: Defining It, Discussing It, and Coping With It 11 TH ANNUAL SYMPOSIUM ON MEN S HEALTH June 12, 2013 The PSA Controversy Defining It, Discussing It and Coping With It As of May 2012,
The 4Kscore blood test for risk of aggressive prostate cancer
 The 4Kscore blood test for risk of aggressive prostate cancer Prostate cancer tests When to use the 4Kscore Test? Screening Prior to 1 st biopsy Prior to negative previous biopsy Prognosis in Gleason 6
The 4Kscore blood test for risk of aggressive prostate cancer Prostate cancer tests When to use the 4Kscore Test? Screening Prior to 1 st biopsy Prior to negative previous biopsy Prognosis in Gleason 6
Screening for Prostate Cancer
 Screening for Prostate Cancer Review against programme appraisal criteria for the UK National Screening Committee (UK NSC) Version 1: This document summarises the work of ScHARR 1 2 and places it against
Screening for Prostate Cancer Review against programme appraisal criteria for the UK National Screening Committee (UK NSC) Version 1: This document summarises the work of ScHARR 1 2 and places it against
PSA screening: Controversies and Guidelines
 PSA screening: Controversies and Guidelines John Phillips, MD, FACS Department of Urology Urology Center of Westchester New York Medical College Historical PerspecGve Cancer of the prostate, although rare,
PSA screening: Controversies and Guidelines John Phillips, MD, FACS Department of Urology Urology Center of Westchester New York Medical College Historical PerspecGve Cancer of the prostate, although rare,
DECISION AID TOOL PROSTATE CANCER SCREENING WITH PSA TESTING
 DECISION AID TOOL PROSTATE CANCER SCREENING WITH PSA TESTING This booklet is what is often called a decision aid. The goals of a decision aid are to help people better understand their medical choices
DECISION AID TOOL PROSTATE CANCER SCREENING WITH PSA TESTING This booklet is what is often called a decision aid. The goals of a decision aid are to help people better understand their medical choices
Role of MRI in the Diagnosis of Prostate Cancer, A Proposal
 Clinical and Experimental Medical Sciences, Vol. 1, 2013, no. 3, 111 116 HIKARI Ltd, www.m-hikari.com Role of MRI in the Diagnosis of Prostate Cancer, A Proposal W. Akhter Research Fellow Urology, Bartshealth
Clinical and Experimental Medical Sciences, Vol. 1, 2013, no. 3, 111 116 HIKARI Ltd, www.m-hikari.com Role of MRI in the Diagnosis of Prostate Cancer, A Proposal W. Akhter Research Fellow Urology, Bartshealth
Advice to patients about the PSA (prostate-specific antigen) blood test: frequently-asked questions
 Advice to patients about the PSA (prostate-specific antigen) blood test: frequently-asked questions What is the PSA blood test? If you want more information before deciding to have this test, it is important
Advice to patients about the PSA (prostate-specific antigen) blood test: frequently-asked questions What is the PSA blood test? If you want more information before deciding to have this test, it is important
Historical Basis for Concern
 Androgens After : Are We Ready? Mohit Khera, MD, MBA Assistant Professor of Urology Division of Male Reproductive Medicine and Surgery Scott Department of Urology Baylor College of Medicine Historical
Androgens After : Are We Ready? Mohit Khera, MD, MBA Assistant Professor of Urology Division of Male Reproductive Medicine and Surgery Scott Department of Urology Baylor College of Medicine Historical
PROSTATE CANCER. Normal-risk men: No family history of prostate cancer No history of prior screening Not African-American
 PROSTATE CANCER 1. Guidelines for Screening Risk Factors Normal-risk men: No family history of prostate cancer No history of prior screening Not African-American High-risk men: Family history of prostate
PROSTATE CANCER 1. Guidelines for Screening Risk Factors Normal-risk men: No family history of prostate cancer No history of prior screening Not African-American High-risk men: Family history of prostate
Newly Diagnosed Prostate Cancer: Understanding Your Risk
 Newly Diagnosed Prostate Cancer: Understanding Your Risk When the urologist calls with the life-changing news that your prostate biopsy is positive for prostate cancer, an office appointment is made to
Newly Diagnosed Prostate Cancer: Understanding Your Risk When the urologist calls with the life-changing news that your prostate biopsy is positive for prostate cancer, an office appointment is made to
da Vinci Prostatectomy Information Guide (Robotically-Assisted Radical Prostatectomy)
 da Vinci Prostatectomy Information Guide (Robotically-Assisted Radical Prostatectomy) Prostate Cancer Overview Prostate cancer is a disease in which malignant (cancer) cells form in the tissues of the
da Vinci Prostatectomy Information Guide (Robotically-Assisted Radical Prostatectomy) Prostate Cancer Overview Prostate cancer is a disease in which malignant (cancer) cells form in the tissues of the
Summary 1. KEY FINDINGS The Office of Technology Assessment (OTA) concludes that research has not yet been completed to determine CHAPTER
 CHAPTER 1 Summary 1 rostate cancer is a common and serious malignancy among Medicare-age men. 1 In 1995, 244,000 new cases and 40,400 deaths are anticipated from this disease; men age 65 and older bear
CHAPTER 1 Summary 1 rostate cancer is a common and serious malignancy among Medicare-age men. 1 In 1995, 244,000 new cases and 40,400 deaths are anticipated from this disease; men age 65 and older bear
PROSTATE CANCER 101 WHAT IS PROSTATE CANCER?
 PROSTATE CANCER 101 WHAT IS PROSTATE CANCER? Prostate cancer is cancer that begins in the prostate. The prostate is a walnut-shaped gland in the male reproductive system located below the bladder and in
PROSTATE CANCER 101 WHAT IS PROSTATE CANCER? Prostate cancer is cancer that begins in the prostate. The prostate is a walnut-shaped gland in the male reproductive system located below the bladder and in
PCA3 DETECTION TEST FOR PROSTATE CANCER DO YOU KNOW YOUR RISK OF HAVING CANCER?
 PCA3 DETECTION TEST FOR PROSTATE CANCER DO YOU KNOW YOUR RISK OF HAVING CANCER? PCA3 DETECTION TEST FOR PROSTATE CANCER There is a range of methods available to your healthcare professional to verify the
PCA3 DETECTION TEST FOR PROSTATE CANCER DO YOU KNOW YOUR RISK OF HAVING CANCER? PCA3 DETECTION TEST FOR PROSTATE CANCER There is a range of methods available to your healthcare professional to verify the
Prostate Cancer. Screening and Diagnosis. Screening. Pardeep Kumar Consultant Urological Surgeon
 The Royal Marsden Prostate Cancer Screening and Diagnosis Pardeep Kumar Consultant Urological Surgeon Prostate Cancer Screening and Diagnosis 08 02 2013 2 Screening 1 3 Q1.Lots of men have prostate cancer
The Royal Marsden Prostate Cancer Screening and Diagnosis Pardeep Kumar Consultant Urological Surgeon Prostate Cancer Screening and Diagnosis 08 02 2013 2 Screening 1 3 Q1.Lots of men have prostate cancer
PATIENT GUIDE. Localized Prostate Cancer
 PATIENT GUIDE Localized Prostate Cancer The prostate* is part of the male reproductive system. It is about the same size as a walnut and weighs about an ounce. As pictured in Figure 1, the prostate is
PATIENT GUIDE Localized Prostate Cancer The prostate* is part of the male reproductive system. It is about the same size as a walnut and weighs about an ounce. As pictured in Figure 1, the prostate is
DIAGNOSIS OF PROSTATE CANCER
 DIAGNOSIS OF PROSTATE CANCER Determining the presence of prostate cancer generally involves a series of tests and exams. Before starting the testing process, the physician will ask questions about the
DIAGNOSIS OF PROSTATE CANCER Determining the presence of prostate cancer generally involves a series of tests and exams. Before starting the testing process, the physician will ask questions about the
Advanced Prostate Cancer Treatments
 Advanced Prostate Cancer Treatments Guest Expert: Kevin, DO Associate Professor of Medical Oncology, Yale Cancer Center www.wnpr.org www.yalecancercenter.org Welcome to Yale Cancer Center Answers with
Advanced Prostate Cancer Treatments Guest Expert: Kevin, DO Associate Professor of Medical Oncology, Yale Cancer Center www.wnpr.org www.yalecancercenter.org Welcome to Yale Cancer Center Answers with
Prostate cancer is the most common cause of death from cancer in men over age 75. Prostate cancer is rarely found in men younger than 40.
 A.D.A.M. Medical Encyclopedia. Prostate cancer Cancer - prostate; Biopsy - prostate; Prostate biopsy; Gleason score Last reviewed: October 2, 2013. Prostate cancer is cancer that starts in the prostate
A.D.A.M. Medical Encyclopedia. Prostate cancer Cancer - prostate; Biopsy - prostate; Prostate biopsy; Gleason score Last reviewed: October 2, 2013. Prostate cancer is cancer that starts in the prostate
Does my patient need more therapy after prostate cancer surgery?
 Does my patient need more therapy after prostate cancer surgery? Contact the GenomeDx Patient Care Team at: 1.888.792.1601 (toll-free) or e-mail: client.service@genomedx.com Prostate Cancer Classifier
Does my patient need more therapy after prostate cancer surgery? Contact the GenomeDx Patient Care Team at: 1.888.792.1601 (toll-free) or e-mail: client.service@genomedx.com Prostate Cancer Classifier
Science Highlights. To PSA or not to PSA: That is the Question.
 Science Highlights June 2012 by Ann A. Kiessling, PhD at the To PSA or not to PSA: That is the Question. The current raucous debate over the commonly used PSA blood test to screen for prostate cancer,
Science Highlights June 2012 by Ann A. Kiessling, PhD at the To PSA or not to PSA: That is the Question. The current raucous debate over the commonly used PSA blood test to screen for prostate cancer,
The 4Kscore blood test for risk of aggressive prostate cancer
 The 4Kscore blood test for risk of aggressive prostate cancer Early detection of aggressive prostate cancer Challenges Serum PSA has a high false positive rate Over 1 million prostate biopsies performed
The 4Kscore blood test for risk of aggressive prostate cancer Early detection of aggressive prostate cancer Challenges Serum PSA has a high false positive rate Over 1 million prostate biopsies performed
Prostate Cancer Action Plan: Choosing the treatment that s right for you
 Prostate Cancer Action Plan: Choosing the treatment that s right for you Segment 1: Introduction Trust me, there's a better way to choose a treatment for your prostate cancer. Watching this program is
Prostate Cancer Action Plan: Choosing the treatment that s right for you Segment 1: Introduction Trust me, there's a better way to choose a treatment for your prostate cancer. Watching this program is
Advances in Diagnostic and Molecular Testing in Prostate Cancer
 Advances in Diagnostic and Molecular Testing in Prostate Cancer Ashley E. Ross MD PhD Assistant Professor Urology, Oncology, Pathology Johns Hopkins School of Medicine September 24, 2015 1 Disclosures
Advances in Diagnostic and Molecular Testing in Prostate Cancer Ashley E. Ross MD PhD Assistant Professor Urology, Oncology, Pathology Johns Hopkins School of Medicine September 24, 2015 1 Disclosures
Prostate Cancer. There is no known association with an enlarged prostate or benign prostatic hyperplasia (BPH).
 Prostate Cancer Definition Prostate cancer is cancer that starts in the prostate gland. The prostate is a small, walnut-sized structure that makes up part of a man's reproductive system. It wraps around
Prostate Cancer Definition Prostate cancer is cancer that starts in the prostate gland. The prostate is a small, walnut-sized structure that makes up part of a man's reproductive system. It wraps around
Questions to ask my doctor: About prostate cancer
 Questions to ask my doctor: About prostate cancer Being diagnosed with prostate cancer can be scary and stressful. You probably have a lot of questions and concerns. Learning about the disease, how it
Questions to ask my doctor: About prostate cancer Being diagnosed with prostate cancer can be scary and stressful. You probably have a lot of questions and concerns. Learning about the disease, how it
Robert Bristow MD PhD FRCPC
 Robert Bristow MD PhD FRCPC Clinician-Scientist and Professor, Radiation Oncology and Medical Biophysics, University of Toronto and Ontario Cancer Institute/ (UHN) Head, PMH-CFCRI Prostate Cancer Research
Robert Bristow MD PhD FRCPC Clinician-Scientist and Professor, Radiation Oncology and Medical Biophysics, University of Toronto and Ontario Cancer Institute/ (UHN) Head, PMH-CFCRI Prostate Cancer Research
Clinical Practice Guidelines
 Clinical Practice Guidelines Prostate Cancer Screening CareMore Quality Management CareMore Health System adopts Clinical Practice Guidelines for the purpose of improving health care and reducing unnecessary
Clinical Practice Guidelines Prostate Cancer Screening CareMore Quality Management CareMore Health System adopts Clinical Practice Guidelines for the purpose of improving health care and reducing unnecessary
Update on Prostate Cancer: Screening, Diagnosis, and Treatment Making Sense of the Noise and Directions Forward
 Update on Prostate Cancer: Screening, Diagnosis, and Treatment Making Sense of the Noise and Directions Forward 33 rd Annual Internal Medicine Update December 5, 2015 Ryan C. Hedgepeth, MD, MS Chief of
Update on Prostate Cancer: Screening, Diagnosis, and Treatment Making Sense of the Noise and Directions Forward 33 rd Annual Internal Medicine Update December 5, 2015 Ryan C. Hedgepeth, MD, MS Chief of
AFTER DIAGNOSIS: PROSTATE CANCER Understanding Your Treatment Options
 AFTER DIAGNOSIS: PROSTATE CANCER Understanding Your Treatment Options INTRODUCTION This booklet describes how prostate cancer develops, how it affects the body and the current treatment methods. Although
AFTER DIAGNOSIS: PROSTATE CANCER Understanding Your Treatment Options INTRODUCTION This booklet describes how prostate cancer develops, how it affects the body and the current treatment methods. Although
Prostate Cancer Guide. A resource to help answer your questions about prostate cancer
 Prostate Cancer Guide A resource to help answer your questions about prostate cancer Thank you for downloading this guide to prostate cancer treatment. We know that all the information provided online
Prostate Cancer Guide A resource to help answer your questions about prostate cancer Thank you for downloading this guide to prostate cancer treatment. We know that all the information provided online
PSA: Prostate Cancer Screening
 PSA: Prostate Cancer Screening 42 nd Annual Convention of the Philippines College of Physicians May 7, 2012, SMX Convention Center Jose Albert Cruz Reyes III, MD, FPCS, FPUA, DPBU Institute of Urology
PSA: Prostate Cancer Screening 42 nd Annual Convention of the Philippines College of Physicians May 7, 2012, SMX Convention Center Jose Albert Cruz Reyes III, MD, FPCS, FPUA, DPBU Institute of Urology
Roswell Park scientists and clinicians:
 The Prostate Cancer Center at Roswell Park Connects You to Nationally Recognized Experts for State-of-the-Art Treatment Options and Compassionate, Evidence-based Care Founded in 1898, Roswell Park Cancer
The Prostate Cancer Center at Roswell Park Connects You to Nationally Recognized Experts for State-of-the-Art Treatment Options and Compassionate, Evidence-based Care Founded in 1898, Roswell Park Cancer
2010 SITE REPORT St. Joseph Hospital PROSTATE CANCER
 2010 SITE REPORT St. Joseph Hospital PROSTATE CANCER Humboldt County is located on the Redwood Coast of Northern California. U.S census data for 2010 reports county population at 134,623, an increase of
2010 SITE REPORT St. Joseph Hospital PROSTATE CANCER Humboldt County is located on the Redwood Coast of Northern California. U.S census data for 2010 reports county population at 134,623, an increase of
Cancer Association of South Africa (CANSA)
 Cancer Association of South Africa (CANSA) Fact Sheet on the Role of Prostate Specific Antigen (PSA) Screening on Prostate Cancer Diagnosis and Treatment Introduction Prostate Specific Antigen (PSA), also
Cancer Association of South Africa (CANSA) Fact Sheet on the Role of Prostate Specific Antigen (PSA) Screening on Prostate Cancer Diagnosis and Treatment Introduction Prostate Specific Antigen (PSA), also
Prostatectomy, pelvic lymphadenect. Med age 63 years Mean followup 53 months No other cancer related therapy before recurrence. Negative.
 Adjuvante und Salvage Radiotherapie Ludwig Plasswilm Klinik für Radio-Onkologie, KSSG CANCER CONTROL WITH RADICAL PROSTATECTOMY ALONE IN 1,000 CONSECUTIVE PATIENTS 1983 1998 Clinical stage T1 and T2 Mean
Adjuvante und Salvage Radiotherapie Ludwig Plasswilm Klinik für Radio-Onkologie, KSSG CANCER CONTROL WITH RADICAL PROSTATECTOMY ALONE IN 1,000 CONSECUTIVE PATIENTS 1983 1998 Clinical stage T1 and T2 Mean
Guidelines for Cancer Prevention, Early detection & Screening. Prostate Cancer
 Guidelines for Cancer Prevention, Early detection & Screening Prostate Cancer Intervention Comments & Recommendations For primary prevention, it has been suggested that diets low in meat & other fatty
Guidelines for Cancer Prevention, Early detection & Screening Prostate Cancer Intervention Comments & Recommendations For primary prevention, it has been suggested that diets low in meat & other fatty
Understanding the. Controversies of. testosterone replacement. therapy in hypogonadal men with prostate cancer. controversies surrounding
 Controversies of testosterone replacement therapy in hypogonadal men with prostate cancer Samuel Deem, DO CULTURA CREATIVE (RF) / ALAMY Understanding the controversies surrounding testosterone replacement
Controversies of testosterone replacement therapy in hypogonadal men with prostate cancer Samuel Deem, DO CULTURA CREATIVE (RF) / ALAMY Understanding the controversies surrounding testosterone replacement
Prostate Cancer. Patient Information
 Prostate Cancer Patient Information 1 The Prostate & Prostate Cancer The prostate is a small gland in the male reproductive system, approximately the size and shape of a walnut. It is located directly
Prostate Cancer Patient Information 1 The Prostate & Prostate Cancer The prostate is a small gland in the male reproductive system, approximately the size and shape of a walnut. It is located directly
Controversites: Screening for Prostate Cancer in Older Adults
 Controversites: Screening for Prostate Cancer in Older Adults William Dale, MD, PhD University of Chicago Sections of Geriatrics & Palliative Medicine and Hematology/Oncology Director, Specialized Oncology
Controversites: Screening for Prostate Cancer in Older Adults William Dale, MD, PhD University of Chicago Sections of Geriatrics & Palliative Medicine and Hematology/Oncology Director, Specialized Oncology
to Know About Your Partner s
 What You Need to Know About Your Partner s Prostate Cancer. A Guide for Wives, Partners, and the Men They Love. Providing Support for Your Partner Prostate cancer can be a physical and emotional challenge
What You Need to Know About Your Partner s Prostate Cancer. A Guide for Wives, Partners, and the Men They Love. Providing Support for Your Partner Prostate cancer can be a physical and emotional challenge
Prostate Cancer Screening. Dr. J. McCracken, Urologist
 Prostate Cancer Screening Dr. J. McCracken, Urologist USPSTF Lifetime risk for diagnosis currently estimated at 15.9% Llifetime risk of dying of prostate cancer is 2.8% Seventy percent of deaths due to
Prostate Cancer Screening Dr. J. McCracken, Urologist USPSTF Lifetime risk for diagnosis currently estimated at 15.9% Llifetime risk of dying of prostate cancer is 2.8% Seventy percent of deaths due to
PSA Screening and the USPSTF Understanding the Controversy
 PSA Screening and the USPSTF Understanding the Controversy Peter C. Albertsen Division of Urology University of Connecticut Farmington, CT, USA USPSTF Final Report 1 Four Key Questions 1. Does PSA based
PSA Screening and the USPSTF Understanding the Controversy Peter C. Albertsen Division of Urology University of Connecticut Farmington, CT, USA USPSTF Final Report 1 Four Key Questions 1. Does PSA based
Questions to Ask My Doctor About Prostate Cancer
 Questions to Ask My Doctor Being told you have prostate cancer can be scary and stressful. You probably have a lot of questions and concerns. Learning about the disease, how it s treated, and how this
Questions to Ask My Doctor Being told you have prostate cancer can be scary and stressful. You probably have a lot of questions and concerns. Learning about the disease, how it s treated, and how this
For further information on screening and early detection of prostate cancer, see the Section entitled Screening for Prostate Cancer.
 Prostate Cancer For many older men, prostate cancer may be present but never cause symptoms or problems and many men will die with their prostate cancer rather than of their prostate cancer. Yet it remains
Prostate Cancer For many older men, prostate cancer may be present but never cause symptoms or problems and many men will die with their prostate cancer rather than of their prostate cancer. Yet it remains
PCa Commentary. Volume 73 January-February 2012 PSA AND TREATMENT DECISIONS:
 1101 Madison Street Suite 1101 Seattle, WA 98104 P 206-215-2480 www.seattleprostate.com PCa Commentary Volume 73 January-February 2012 CONTENTS PSA SCREENING & BASIC SCIENCE PSA AND TREATMENT 1 DECISIONS
1101 Madison Street Suite 1101 Seattle, WA 98104 P 206-215-2480 www.seattleprostate.com PCa Commentary Volume 73 January-February 2012 CONTENTS PSA SCREENING & BASIC SCIENCE PSA AND TREATMENT 1 DECISIONS
Prostate Cancer In-Depth
 Prostate Cancer In-Depth Introduction Prostate cancer is the most common visceral malignancy among American men. In the year 2003, there are expected to be 220,000 new cases and nearly 29,000 deaths in
Prostate Cancer In-Depth Introduction Prostate cancer is the most common visceral malignancy among American men. In the year 2003, there are expected to be 220,000 new cases and nearly 29,000 deaths in
Screening for Prostate Cancer
 Cancer Expert Working Group on Cancer Prevention and Screening Screening for Prostate Cancer Information for men and their families 1 What is the prostate? 2 What is prostate cancer? prostate The prostate
Cancer Expert Working Group on Cancer Prevention and Screening Screening for Prostate Cancer Information for men and their families 1 What is the prostate? 2 What is prostate cancer? prostate The prostate
Cancer doesn t care but we do. 2010 Cancer Annual Report
 Cancer doesn t care but we do. 2010 Cancer Annual Report The Cancer Committee of CHRISTUS St. Patrick Hospital is proud to present its 2010 Annual Report. The Community Hospital Comprehensive Cancer Program
Cancer doesn t care but we do. 2010 Cancer Annual Report The Cancer Committee of CHRISTUS St. Patrick Hospital is proud to present its 2010 Annual Report. The Community Hospital Comprehensive Cancer Program
Testing for Prostate Cancer. Should I be tested? Is it the right choice for me?
 Testing for Prostate Cancer Should I be tested? Is it the right choice for me? Prostate cancer affects many men. There are tests to find it early. There may be benefits and risks with testing. Research
Testing for Prostate Cancer Should I be tested? Is it the right choice for me? Prostate cancer affects many men. There are tests to find it early. There may be benefits and risks with testing. Research
The PLCO Trial Not a comparison of Screening vs no Screening
 PSA Screening: Science, Politics and Uncertainty David F. Penson, MD, MPH Hamilton and Howd Chair of Urologic Oncology Professor and Chair, Department of Urologic Surgery Director, Center for Surgical
PSA Screening: Science, Politics and Uncertainty David F. Penson, MD, MPH Hamilton and Howd Chair of Urologic Oncology Professor and Chair, Department of Urologic Surgery Director, Center for Surgical
Your Health Matters. Localized Prostate Cancer and Its Treatment
 Your Health Matters Localized Prostate Cancer and Its Treatment Greetings! Understanding prostate cancer and choosing among the various treatment options can be a difficult and anxiety-arousing process.
Your Health Matters Localized Prostate Cancer and Its Treatment Greetings! Understanding prostate cancer and choosing among the various treatment options can be a difficult and anxiety-arousing process.
Facing Prostate Cancer Surgery? Learn about minimally invasive da Vinci Surgery
 Facing Prostate Cancer Surgery? Learn about minimally invasive da Vinci Surgery The Condition: Prostate Cancer Your prostate is a walnut-sized gland that is part of the male reproductive system. The prostate
Facing Prostate Cancer Surgery? Learn about minimally invasive da Vinci Surgery The Condition: Prostate Cancer Your prostate is a walnut-sized gland that is part of the male reproductive system. The prostate
Prostate Cancer Treatment: What s Best for You?
 Prostate Cancer Treatment: What s Best for You? Prostate Cancer: Radiation Therapy Approaches I. Choices There is really a variety of options in prostate cancer management overall and in radiation therapy.
Prostate Cancer Treatment: What s Best for You? Prostate Cancer: Radiation Therapy Approaches I. Choices There is really a variety of options in prostate cancer management overall and in radiation therapy.
Get all the Information about Prostate Cancer Treatment
 Volume Article.13-05 Publish Date: 28 th January 2013 Author(s): MPUH - CRS Team Get all the Information about Prostate Cancer Treatment ARTICLE hhh Muljibhai Patel Urological Hospital (MPUH) Centre For
Volume Article.13-05 Publish Date: 28 th January 2013 Author(s): MPUH - CRS Team Get all the Information about Prostate Cancer Treatment ARTICLE hhh Muljibhai Patel Urological Hospital (MPUH) Centre For
Radiation Therapy for Prostate Cancer
 Radiation Therapy for Prostate Cancer Introduction Cancer of the prostate is the most common form of cancer that affects men. About 240,000 American men are diagnosed with prostate cancer every year. Your
Radiation Therapy for Prostate Cancer Introduction Cancer of the prostate is the most common form of cancer that affects men. About 240,000 American men are diagnosed with prostate cancer every year. Your
Gleason Score. Oncotype DX GPS. identified for. about surveillance. time to get sophisticated
 patient: MARK SMITH PSA 6.2 Gleason Score 6 Oncotype DX GPS 8 identified for active surveillance time to get sophisticated about surveillance Accurate prediction of prostate cancer risk is needed at the
patient: MARK SMITH PSA 6.2 Gleason Score 6 Oncotype DX GPS 8 identified for active surveillance time to get sophisticated about surveillance Accurate prediction of prostate cancer risk is needed at the
Draft Clinical Practice Guidelines. PSA Testing and Early Management of Test-Detected Prostate Cancer
 Draft Clinical Practice Guidelines PSA Testing and Early Management of Test-Detected Prostate Cancer Draft November 2014 Prostate Cancer Foundation of Australia and Cancer Council Australia ISBN: This
Draft Clinical Practice Guidelines PSA Testing and Early Management of Test-Detected Prostate Cancer Draft November 2014 Prostate Cancer Foundation of Australia and Cancer Council Australia ISBN: This
Prostate Cancer. What is prostate cancer?
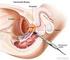 Scan for mobile link. Prostate Cancer Prostate cancer is a tumor of the prostate gland, which is located in front of the rectum and below the bladder. Your doctor may perform a physical exam, prostate-specific
Scan for mobile link. Prostate Cancer Prostate cancer is a tumor of the prostate gland, which is located in front of the rectum and below the bladder. Your doctor may perform a physical exam, prostate-specific
November 25, 2015. Albert L Siu, MD, MSPH Chair, US Preventive Services Task Force 5600 Fishers Lane, Mail Stop 06E53A Rockville, MD 20857
 November 25, 2015 Men's Health Network P. O. Box 75972 Washington, D.C. 20013 202-543-MHN-1 (6461) Fax 202-543-2727 Albert L Siu, MD, MSPH Chair, US Preventive Services Task Force 5600 Fishers Lane, Mail
November 25, 2015 Men's Health Network P. O. Box 75972 Washington, D.C. 20013 202-543-MHN-1 (6461) Fax 202-543-2727 Albert L Siu, MD, MSPH Chair, US Preventive Services Task Force 5600 Fishers Lane, Mail
