Use of Mode of Action in Human Health Risk Assessment
|
|
|
- August Morton
- 7 years ago
- Views:
Transcription
1 Use of Mode of Action in Human Health Vicki L. Dellarco, Ph.D. US Environmental Protection Program Office of Pesticide Programs This talk represents the view of presenter & does not necessarily represent the decisions or the stated policies of the EPA. MoA Analyses by Office of Pesticide Programs Chloro-s-triazines (neuroendocrine disruption & developmental/reproductive effects & mammary gland tumors) Conazole fungicides (CAR activation & liver tumors) Diphenyl ethers (PPAR" agonism & liver tumors) Dicarboximide fungicides (inhibition of androgen receptor & reproductive malformations/leydig leydig cell tumors) Organophosphorus pesticides (cholinesterase inhibition & cholinergic toxicity) Chloroacaetanilide herbicides (cytoxicity/regenerative proliferation & nasal tumors) Different classess (disruption of Thyroid hormone homeostasis & neurodevelopmental effects & follicular cell tumors) AND SO FORTH
2 How Do We Assess Human Health Risks? NAS 4-Step Paradigm Hazard Assessment & Characterization Dose Response Assessment & Characterization Exposure Assessment & Characterization FQPA 10 X Safety Factor, Aggregate Exposure & Cumulative Risk & Characterization How Do We Assess Human Health Risks? Relies heavily on laboratory animal data Relies on extrapolations, inference methods, safety factors, etc Animal Biology = Human Biology Effects found at high animal doses predict effects at environmental levels of exposure Current animal assays provide adequate coverage for predicting effects on human health including susceptible groups (?)
3 Mode of Action (MoA( MoA) ) Data in Inform Human Relevance Dose Response Susceptibilities Toxicity Pathways Identify Key Biological Events Understand Chemical Mixture or Cumulative Risk Promote Harmonized Approach For All Health Endpoints MoA/Human Relevance Framework History USEPA 96 draft Cancer Guidelines Defines MoA & contrasts with mechanism USEPA 99 draft Cancer Guidelines Introduces Framework Bradford Hill criteria for causality for weight of evidence for MoA MoA = Plausible hypothesis with measured key events (vs detailed molecular description of causality) Key Event: Critical, Rate Limiting, Quantifiable
4 MoA/Human Relevance Framework History continued ILSI RSI develops human relevance (HR) component & case studies for cancer and noncancer outcomes Critical Reviews in Toxicology (Meek et al., 2003; Cohen et al., 2004; Seed et al., 2005) IPCS refines & adds case studies Critical Reviews in Toxicology (Boobis et al., 2006; Boobis et al., 2008) Human Relevance Framework for Analyzing MoA Why? Provides Transparency clarifies extent of weight of evidence as a basis for decision making not prescriptive but designed to organize information Ensures Rigor of Evaluations & Consistency of Documentation Aids in Identification of Critical Data or Research Needs impact on conclusions basis for iterative dialogue between risk assessors/researchers
5 MoA/Human Relevance Framework Key Events established based on Hill Criteria Is the WoE Sufficient to establish a MoA in animals? MoA t Relevant MoA t Relevant Are the key events in the animal MoA (qualitatively) plausible in humans? Taking into account Kinetic & dynamic factors, is the animal MoA plausible in humans? Comparability analysis of Key Events & relevant biology between animals & humans MoA: Sufficiency of Evidence Interdisciplinary Teams Critical Analysis How much is enough? Peer Engagement *Burden of proof is reduced for well established MoAs
6 Q2. & 3.The Concordance Analysis Key Event Animals Humans Strength Atrazine as an Example Rat mode of action for mammary gland tumors not expected to occur in humans but mode of action should be assumed relevant for effects on reproductive development (FIFRA Science Advisory Panel, 2000)
7 MoA/Human Relevance Framework Is the WoE Sufficient to establish a MoA in animals? MoA t Relevant Are the key events in the animal MoA (qualitatively) plausible in humans? MoA t Relevant Taking into account Kinetic & dynamic factors, is the animal MoA plausible in humans? MoA/Human Relevance Framework: Q1. Is the WoE Sufficient to Establish MoA in Animals? Bradford Hill Criteria Postulated mode of action Identify sequence of key events on the path to cancer Experimental support Concordance of dose-response for key events with that for tumors Temporal relationships for key events & tumors Biological plausibility & Coherence Strength, consistency & specificity Other modes of action Identify uncertainties Conclusion
8 Postulated MoA: Consider rmal Biological Process Reproductive Aging (prolong estrus) in SD female Rats plays role in high mammary gland tumor incidence Establishes a hormonal environment conducive to tumor development prolonged exposure to endogenous estrogens MoA/Human Relevance Framework: Q1.Weight of Evidence Sufficient to Establish MoA? Postulated MoA - Description of Key Events Latency/ Incidence Mammary Gland Tumors Exposure to estrogens Anovulation (extended estrus) Pituitary LH Surge Atrazine Alters Hypothalamic Control of Pituitary-Ovarian Function GnRH
9 Human Relevance Framework: Q 1. Weight of Evidence Sufficient to Establish MoA Experimental Support Dose-Response Relationship Concordance of LH, extended estrus, latency/incidence of mammary tumors Temporal Relationship LH, disrupted cyclicity precede tumors LH (6 mo.) Effect LOAEL mg/kg/d 3.65 Reference Morseth,, 1996 Days in Estrus (6 mo.) Latency in Mammary Tumors (12 mo.) Incidence in Mammary Tumors (24 mo.) Morseth,, 1996 Thakur,, 1997 Mayhew, 1986 Strength, Consistency, Specificity of Association Consistent evidence from many studies Strong correlation between dose & effect ( LH, estrous cycle disruption, mammary gland tumors) ATZ-treated OVX females do not develop mammary tumors, ectopic pituitary & GnRH experiments support hypothalamus as a primary site Biological Plausibility & Coherence Substantial literature on role of estrogen in pathogenesis of mammary tumors in rats Morphological changes indicative of estrogen stimulation Other LH-dependent events affected pregnancy loss, delayed puberty Assessment of Uncertainties LH data variable, acute studies for GnRH levels, limited data on estrogen levels Do not discount postulated neuroendocrine MoA
10 Other MoAs t a DNA reactive mutagenic intrinsic estrogen potential Induction of aromatase activity but only in vitro support in vivo (rat) Disruption of adrenal steroidogenesis in rats Higher doses than those required to LH MoA/Human Relevance Framework Is the WoE Sufficient to establish a MoA in animals? MoA t Relevant Are the key events in the animal MoA (qualitatively) plausible in humans? MoA t Relevant Taking into account Kinetic & dynamic factors, is the animal MoA plausible in humans?
11 Q2.Qualitative Concordance Analysis Key Event GnRH pulses Rat (SD), Ectopic Pituitary & GnRH expts; GnRH restores LH in atrazine-blocked rats Humans Unknown, possible of LH surge % days d in estrus exposure to estrogen Mammary Gland Tumors, several expts in rats at different dose/durations, several expts in rats at different dose/durations, questionable data, incidence & early onset Unknown, possible Unknown, possible Unknown, NOT plausible t plausible, no convincing evidence that atrazine results in cancer in humans MoA/Human Relevance Framework Is the WoE Sufficient to establish a MoA in animals? MoA t Relevant MoA t Relevant Are the key events in the animal MoA (qualitatively) plausible in humans? Taking into account Kinetic & dynamic factors, is the animal MoA plausible in humans? Developmental effects should be be assumed
12 MoA: : Implications for Dose Response R e s p o n s e Consider Key Events to protect human health from neuroendocrine effects of Atrazine Human Exposure RfD = PoD/UFs PoD Based on xlh Suppression x x PoD = 1.8mg/kg/day x MoE NOAEL Dose PoD = Point of Departure RfD = Reference Dose MoE = Margin of Exposure Cumulative Risk of Compounds that Share a Common Mechanism of Toxicity (FQPA 96 ) OH C C 2 3 C C l l OH C C 2 3 OH Pesticide Inventory C C l l OH C C 2 3 Triazines: Reproductive & developmental effects based on GnRH & LH suppression C C l l C C 2 3 OH
13 Contribution of MoA Human Relevance Framework Transparent Consideration of Weight of Evidence Basis for MoA Promotes Use of All Relevant (including non chemical specific) Data Considers of Lifestage Responses MoA Defines the Key Events (Critical Endpoints) Relevant to dose response extrapolation method & point of departure Concordance Tables Help Identify Kinetic & Dynamic Differences Between Species (interspecies scaling) Delineates Types of Data that are Preferred over Defaults Paradigm Change Next Steps Reinforced by NRC, 2007 (Toxicity Testing in the 21 st Century) Move toward hypothesis/tiered approaches Use knowledge of toxicity pathways Use computational approaches, genomics, HTP, in vitro technologies for tailoring in vivo testing In vivo testing focuses on relevant endpoints, mode of action & kinetics/metabolism
Edward Odenkirchen, Ph.D. Office of Pesticide Programs US Environmental Protection Agency
 Edward Odenkirchen, Ph.D. Office of Pesticide Programs US Environmental Protection Agency 1 Mission Statement Best possible regulatory decisions to protect public health and environment. Rely on all best
Edward Odenkirchen, Ph.D. Office of Pesticide Programs US Environmental Protection Agency 1 Mission Statement Best possible regulatory decisions to protect public health and environment. Rely on all best
Communication of Uncertainty in Hazard & Risk Assessment: Panel Discussion. Lynn H. Pottenger, PhD, DABT The Dow Chemical Company
 Communication of Uncertainty in Hazard & Risk Assessment: Panel Discussion Lynn H. Pottenger, PhD, DABT The Dow Chemical Company Uncertainty Workshop Focus: Focus on identification of sources & communication
Communication of Uncertainty in Hazard & Risk Assessment: Panel Discussion Lynn H. Pottenger, PhD, DABT The Dow Chemical Company Uncertainty Workshop Focus: Focus on identification of sources & communication
Risk Assessment in Chemical Food Safety. Dept. of Food Safety and Zoonoses (FOS) http://www.who.int/foodsafety/en/
 Risk Assessment in Chemical Food Safety Dept. of Food Safety and Zoonoses (FOS) http://www.who.int/foodsafety/en/ Risk Analysis Paradigm Internationally Scientific data analysis Risk Assessment WHO & FAO
Risk Assessment in Chemical Food Safety Dept. of Food Safety and Zoonoses (FOS) http://www.who.int/foodsafety/en/ Risk Analysis Paradigm Internationally Scientific data analysis Risk Assessment WHO & FAO
Risk Assessment of Pesticides Residue by JMPR - To set ADI and ARfD-
 The FAO Regional Training Course: Strengthening Capacity of Data Collection and Generation for Food Safety Risk Analysis Support to Capacity Building and Implementation of International Safety Standards
The FAO Regional Training Course: Strengthening Capacity of Data Collection and Generation for Food Safety Risk Analysis Support to Capacity Building and Implementation of International Safety Standards
Chemical Name: Acetone CAS: 67-64-1 Synonyms: propanone, β-ketopropane, dimethyl ketone, dimethylformaldehyde, DMK, 2-propanone, propan-2-one
 2011 Health Risk Limits for Groundwater Health Risk Assessment Unit, Environmental Health Division 651-201-4899 651-201-5797 TDD Web Publication Date: March 21, 2011 Chemical Name: Acetone CAS: 67-64-1
2011 Health Risk Limits for Groundwater Health Risk Assessment Unit, Environmental Health Division 651-201-4899 651-201-5797 TDD Web Publication Date: March 21, 2011 Chemical Name: Acetone CAS: 67-64-1
Chemical safety and big data: the industry s demands
 Chemical safety and big data: the industry s demands Richard CURRIE Senior Technical Expert; Group Leader & Global Predictive and Computational Toxicology Lead Valid results Useful results Credit Money/Grants
Chemical safety and big data: the industry s demands Richard CURRIE Senior Technical Expert; Group Leader & Global Predictive and Computational Toxicology Lead Valid results Useful results Credit Money/Grants
Guidance on Cumulative Risk Assessment of Pesticide Chemicals That Have a Common Mechanism of Toxicity
 Guidance on Cumulative Risk Assessment of Pesticide Chemicals That Have a Common Mechanism of Toxicity Office of Pesticide Programs U.S. Environmental Protection Agency Washington, D.C. 20460 January 14,
Guidance on Cumulative Risk Assessment of Pesticide Chemicals That Have a Common Mechanism of Toxicity Office of Pesticide Programs U.S. Environmental Protection Agency Washington, D.C. 20460 January 14,
The Science of Chemical Safety Essential Toxicology - 4. Hazard and Risk. John Duffus & Howard Worth. IUPAC Educators Resource Material IUPAC
 The Science of Chemical Safety Essential Toxicology - 4 Hazard and Risk John Duffus & Howard Worth IUPAC Educators Resource Material IUPAC Hazard and Risk - 1 Hazard is the potential of a substance to
The Science of Chemical Safety Essential Toxicology - 4 Hazard and Risk John Duffus & Howard Worth IUPAC Educators Resource Material IUPAC Hazard and Risk - 1 Hazard is the potential of a substance to
Introduction. MassDEP, Office of Research and Standards 1 Tetrachloroethylene
 Summary of the Basis of Cancer Risk Values for Massachusetts Department of Environmental Protection (MassDEP) Office of Research and Standards January 22, 2014 Introduction In February 2012 the United
Summary of the Basis of Cancer Risk Values for Massachusetts Department of Environmental Protection (MassDEP) Office of Research and Standards January 22, 2014 Introduction In February 2012 the United
Chemical Risk Assessment in Absence of Adequate Toxicological Data
 Chemical Risk Assessment in Absence of Adequate Toxicological Data Mark Cronin School of Pharmacy and Chemistry Liverpool John Moores University England m.t.cronin@ljmu.ac.uk The Problem Risk Analytical
Chemical Risk Assessment in Absence of Adequate Toxicological Data Mark Cronin School of Pharmacy and Chemistry Liverpool John Moores University England m.t.cronin@ljmu.ac.uk The Problem Risk Analytical
DRAFT THE OFFICE OF PESTICIDE PROGRAMS POLICY DETERMINATION OF THE APPROPRIATE FQPA SAFETY FACTOR(S) FOR USE IN THE TOLERANCE-SETTING PROCESS
 5/10/99 DRAFT THE OFFICE OF PESTICIDE PROGRAMS POLICY ON DETERMINATION OF THE APPROPRIATE FQPA SAFETY FACTOR(S) FOR USE IN THE TOLERANCE-SETTING PROCESS OFFICE OF PESTICIDE PROGRAMS U.S. ENVIRONMENTAL
5/10/99 DRAFT THE OFFICE OF PESTICIDE PROGRAMS POLICY ON DETERMINATION OF THE APPROPRIATE FQPA SAFETY FACTOR(S) FOR USE IN THE TOLERANCE-SETTING PROCESS OFFICE OF PESTICIDE PROGRAMS U.S. ENVIRONMENTAL
Wellmark International Dog Collar 34 g (1.2 oz) 5986 0.0051 55 15. 0.28 1000 Cat Collar 10.5 g (0.37oz)
 revised risk estimates which are provided in Table 1 below. These corrections do not alter the risk conclusions as described in D371250. The Agency is also anticipating additional data from the registrants
revised risk estimates which are provided in Table 1 below. These corrections do not alter the risk conclusions as described in D371250. The Agency is also anticipating additional data from the registrants
Regulatory Risk Assessment
 Regulatory Risk Assessment Richard B. Belzer PhD Regulatory Checkbook Mt. Vernon, VA Belzer@RegulatoryCheckbook.Org What is the Purpose of Risk Assessment? For regulatory analysis [A] risk assessment should
Regulatory Risk Assessment Richard B. Belzer PhD Regulatory Checkbook Mt. Vernon, VA Belzer@RegulatoryCheckbook.Org What is the Purpose of Risk Assessment? For regulatory analysis [A] risk assessment should
NATIONAL INSTITUTE OF ENVIRONMENTAL HEALTH SCIENCES Division of Extramural Research and Training
 NATIONAL INSTITUTE OF ENVIRONMENTAL HEALTH SCIENCES Division of Extramural Research and Training NATIONAL ADVISORY ENVIRONMENTAL HEALTH SCIENCES COUNCIL May 12-13, 2010 Concept Clearance Small Business
NATIONAL INSTITUTE OF ENVIRONMENTAL HEALTH SCIENCES Division of Extramural Research and Training NATIONAL ADVISORY ENVIRONMENTAL HEALTH SCIENCES COUNCIL May 12-13, 2010 Concept Clearance Small Business
Evaluation of Human Relevance and Mode of Action for Tunica. Vaginalis Mesotheliomas Resulting from Oral Exposure to. Acrylamide
 Evaluation of Human Relevance and Mode of Action for Tunica Vaginalis Mesotheliomas Resulting from Oral Exposure to Acrylamide Lynne T. Haber a*, Andrew Maier a, Oliver L. Kroner a, and Melissa J. Kohrman
Evaluation of Human Relevance and Mode of Action for Tunica Vaginalis Mesotheliomas Resulting from Oral Exposure to Acrylamide Lynne T. Haber a*, Andrew Maier a, Oliver L. Kroner a, and Melissa J. Kohrman
Issues Relevant to Endocrine Disruptor Screening
 Concepts of Endocrinology: Issues Relevant to Endocrine Disruptor Screening Raphael J. Witorsch, Ph.D. Professor Emeritus Physiology and Biophysics School of Medicine Virginia i i Commonwealth University
Concepts of Endocrinology: Issues Relevant to Endocrine Disruptor Screening Raphael J. Witorsch, Ph.D. Professor Emeritus Physiology and Biophysics School of Medicine Virginia i i Commonwealth University
ITRC PROJECT PROPOSAL
 ITRC PROJECT PROPOSAL Risk Assessment Guidance and Training: State-of-the-Art Principles and Practice Please use brief statements or bullet items to input the requested information PROPOSAL DATE: March
ITRC PROJECT PROPOSAL Risk Assessment Guidance and Training: State-of-the-Art Principles and Practice Please use brief statements or bullet items to input the requested information PROPOSAL DATE: March
In Vivo and In Vitro Screening for Thyroid Hormone Disruptors
 In Vivo and In Vitro Screening for Thyroid Hormone Disruptors Kevin M. Crofton National lhealth and Environmental Research Laboratory California Environmental Protection Agency Human Health Hazard Indicators
In Vivo and In Vitro Screening for Thyroid Hormone Disruptors Kevin M. Crofton National lhealth and Environmental Research Laboratory California Environmental Protection Agency Human Health Hazard Indicators
NAFTA TWG Sound Science and Policy
 NAFTA TWG Sound Science and Policy Presentation to the NAFTA Technical Working Group on Pesticides Stakeholder-Government Meeting November 17, 2011 Dr. Peter Chan, Director General Health Evaluation Directorate
NAFTA TWG Sound Science and Policy Presentation to the NAFTA Technical Working Group on Pesticides Stakeholder-Government Meeting November 17, 2011 Dr. Peter Chan, Director General Health Evaluation Directorate
AGENDA REQUEST AGENDA ITEM NO: IV.B.5. Consent Agenda No. 2. March 21, 2011 BY City Auditor and Clerk Pamela M. Nadalini Mayor Kirschner SUBJECT:
 AGENDA REQUEST AGENDA HEADING: Consent Agenda No. 2 COMMISSION MEETING DATE: March 21, 2011 BY City Auditor and Clerk Pamela M. Nadalini Mayor Kirschner AGENDA ITEM NO: IV.B.5. Originating Department SUBJECT:
AGENDA REQUEST AGENDA HEADING: Consent Agenda No. 2 COMMISSION MEETING DATE: March 21, 2011 BY City Auditor and Clerk Pamela M. Nadalini Mayor Kirschner AGENDA ITEM NO: IV.B.5. Originating Department SUBJECT:
Biological importance of metabolites. Safety and efficacy aspects
 Biological importance of metabolites Safety and efficacy aspects Bernard Walther Technologie Servier Biological importance of metabolites Safety testing of drug metabolites Bioanalytical strategy Structural
Biological importance of metabolites Safety and efficacy aspects Bernard Walther Technologie Servier Biological importance of metabolites Safety testing of drug metabolites Bioanalytical strategy Structural
An Evidence-Based Approach to Reviewing the Science on the Safety of Chemicals in Foods
 An Evidence-Based Approach to Reviewing the Science on the Safety of Chemicals in Foods In considering the safety of chemicals added to foods, or present in foods due to environmental circumstances, we
An Evidence-Based Approach to Reviewing the Science on the Safety of Chemicals in Foods In considering the safety of chemicals added to foods, or present in foods due to environmental circumstances, we
ICH Topic S 1 A The Need for Carcinogenicity Studies of Pharmaceuticals. Step 5
 European Medicines Agency July 1996 CPMP/ICH/140/95 ICH Topic S 1 A The Need for Carcinogenicity Studies of Pharmaceuticals Step 5 NOTE FOR GUIDANCE ON THE NEED FOR CARCINOGENICITY STUDIES OF PHARMACEUTICALS
European Medicines Agency July 1996 CPMP/ICH/140/95 ICH Topic S 1 A The Need for Carcinogenicity Studies of Pharmaceuticals Step 5 NOTE FOR GUIDANCE ON THE NEED FOR CARCINOGENICITY STUDIES OF PHARMACEUTICALS
SUMMARY: This regulation establishes a tolerance for residues of propyzamide, also
 This document is scheduled to be published in the Federal Register on 01/13/2016 and available online at http://federalregister.gov/a/2016-00534, and on FDsys.gov BILLING CODE 6560-50-P ENVIRONMENTAL PROTECTION
This document is scheduled to be published in the Federal Register on 01/13/2016 and available online at http://federalregister.gov/a/2016-00534, and on FDsys.gov BILLING CODE 6560-50-P ENVIRONMENTAL PROTECTION
Mode of Action Frameworks in Toxicity Testing and Chemical Risk Assessment
 Mode of Action Frameworks in Toxicity Testing and Chemical Risk Assessment Mary Elizabeth Meek 2009 Research included in this thesis was conducted as part of the author s responsibilities at Health Canada.
Mode of Action Frameworks in Toxicity Testing and Chemical Risk Assessment Mary Elizabeth Meek 2009 Research included in this thesis was conducted as part of the author s responsibilities at Health Canada.
R. Julian Preston NHEERL U.S. Environmental Protection Agency Research Triangle Park North Carolina. Ninth Beebe Symposium December 1, 2010
 Low Dose Risk Estimation: The Changing Face of Radiation Risk Assessment? R. Julian Preston NHEERL U.S. Environmental Protection Agency Research Triangle Park North Carolina Ninth Beebe Symposium December
Low Dose Risk Estimation: The Changing Face of Radiation Risk Assessment? R. Julian Preston NHEERL U.S. Environmental Protection Agency Research Triangle Park North Carolina Ninth Beebe Symposium December
WHAT IS RISK? Definition: probability of harm or loss Risk = Hazard x Exposure Risk can be voluntary or involuntary Interpretation of risk differs for
 BASELINE RISK ASSESSMENT OVERVIEW Dawn A. Ioven Senior Toxicologist U.S. EPA Region III WHAT IS RISK? Definition: probability of harm or loss Risk = Hazard x Exposure Risk can be voluntary or involuntary
BASELINE RISK ASSESSMENT OVERVIEW Dawn A. Ioven Senior Toxicologist U.S. EPA Region III WHAT IS RISK? Definition: probability of harm or loss Risk = Hazard x Exposure Risk can be voluntary or involuntary
A Tiered Approach to Systemic Toxicity Testing for Agricultural Chemical Safety Assessment
 Critical Reviews in Toxicology, 36:37 68, 2006 Copyright c Taylor and Francis Group, LLC ISSN: 1040-8444 print / 1547-6898 online DOI: 10.1080/10408440500534370 A Tiered Approach to Systemic Toxicity Testing
Critical Reviews in Toxicology, 36:37 68, 2006 Copyright c Taylor and Francis Group, LLC ISSN: 1040-8444 print / 1547-6898 online DOI: 10.1080/10408440500534370 A Tiered Approach to Systemic Toxicity Testing
GUIDELINES FOR THE REGISTRATION OF BIOLOGICAL PEST CONTROL AGENTS FOOD AND AGRICULTURE ORGANIZATION OF THE UNITED NATIONS
 GUIDELINES FOR THE REGISTRATION OF BIOLOGICAL PEST CONTROL AGENTS FOOD AND AGRICULTURE ORGANIZATION OF THE UNITED NATIONS -ii- GUIDELINES ON THE REGISTRATION OF BIOLOGICAL PEST CONTROL AGENTS FOOD AND
GUIDELINES FOR THE REGISTRATION OF BIOLOGICAL PEST CONTROL AGENTS FOOD AND AGRICULTURE ORGANIZATION OF THE UNITED NATIONS -ii- GUIDELINES ON THE REGISTRATION OF BIOLOGICAL PEST CONTROL AGENTS FOOD AND
MATERIAL SAFETY DATA SHEET
 Page 1 of 6 1. IDENTIFICATION OF THE SUBSTANCE/PREPARATION AND THE COMPANY/UNDERTAKING Pfizer Inc Pfizer Pharmaceuticals Group 235 East 42nd Street New York, New York 10017 1-212-573-2222 Emergency telephone
Page 1 of 6 1. IDENTIFICATION OF THE SUBSTANCE/PREPARATION AND THE COMPANY/UNDERTAKING Pfizer Inc Pfizer Pharmaceuticals Group 235 East 42nd Street New York, New York 10017 1-212-573-2222 Emergency telephone
INTERNATIONAL CONFERENCE ON HARMONISATION OF TECHNICAL REQUIREMENTS FOR REGISTRATION OF PHARMACEUTICALS FOR HUMAN USE S1A. Current Step 4 version
 INTERNATIONAL CONFERENCE ON HARMONISATION OF TECHNICAL REQUIREMENTS FOR REGISTRATION OF PHARMACEUTICALS FOR HUMAN USE ICH HARMONISED TRIPARTITE GUIDELINE GUIDELINE ON THE NEED FOR CARCINOGENICITY STUDIES
INTERNATIONAL CONFERENCE ON HARMONISATION OF TECHNICAL REQUIREMENTS FOR REGISTRATION OF PHARMACEUTICALS FOR HUMAN USE ICH HARMONISED TRIPARTITE GUIDELINE GUIDELINE ON THE NEED FOR CARCINOGENICITY STUDIES
Module Three. Risk Assessment
 Module Three Risk Assessment 136 Module Three Introduction to Risk Assessment Time Allotted: 90 Minutes Objectives: Upon completion of this module, the learner will be able to # Define and understand the
Module Three Risk Assessment 136 Module Three Introduction to Risk Assessment Time Allotted: 90 Minutes Objectives: Upon completion of this module, the learner will be able to # Define and understand the
Uncertainty factors:
 The Interdepartmental Group on Health Risks from Chemicals Uncertainty factors: Their use in human health risk assessment by UK Government cr9 The Interdepartmental Group on Health Risks from Chemicals
The Interdepartmental Group on Health Risks from Chemicals Uncertainty factors: Their use in human health risk assessment by UK Government cr9 The Interdepartmental Group on Health Risks from Chemicals
AOP Knowledge Base/ Wiki Tool Set
 AOP Knowledge Base/ Wiki Tool Set Stephen W. Edwards Adverse Outcome Pathways: From Research to Regulation This talk does not necessarily reflect the views of the Environmental Protection Agency. Photo
AOP Knowledge Base/ Wiki Tool Set Stephen W. Edwards Adverse Outcome Pathways: From Research to Regulation This talk does not necessarily reflect the views of the Environmental Protection Agency. Photo
Probabilistic Risk Assessment Methodology for Criteria Calculation Versus the Standard (Deterministic) Methodology
 Probabilistic Risk Assessment Methodology for Criteria Calculation Versus the Standard (Deterministic) Methodology State of Idaho Department of Environmental Quality March 2014 Background The equation
Probabilistic Risk Assessment Methodology for Criteria Calculation Versus the Standard (Deterministic) Methodology State of Idaho Department of Environmental Quality March 2014 Background The equation
Risk Assessment Report on (3-CHLORO-2-HYDROXYPROPYL)TRIMETHYLAMMONIUM CHLORIDE (CHPTAC)
 Scientific Committee on Health and Environmental Risks SCHER Risk Assessment Report on (3-CHLORO-2-HYDROXYPROPYL)TRIMETHYLAMMONIUM CHLORIDE (CHPTAC) Human Health Part CAS No.: 3327-22-8 EINECS No.: 222-048-3
Scientific Committee on Health and Environmental Risks SCHER Risk Assessment Report on (3-CHLORO-2-HYDROXYPROPYL)TRIMETHYLAMMONIUM CHLORIDE (CHPTAC) Human Health Part CAS No.: 3327-22-8 EINECS No.: 222-048-3
WHAT S NEW IN ALTERNATIVES?
 WHAT S NEW IN ALTERNATIVES? Joanne Zurlo, PhD Center for Alternatives to Animal Testing (CAAT) Johns Hopkins Bloomberg School of Public Health Baltimore, MD What are alternatives? Alternative are sometimes
WHAT S NEW IN ALTERNATIVES? Joanne Zurlo, PhD Center for Alternatives to Animal Testing (CAAT) Johns Hopkins Bloomberg School of Public Health Baltimore, MD What are alternatives? Alternative are sometimes
Reproduction and its Hormonal Control
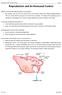 Reproduction and its Hormonal Control Page 1 Reproduction and its Hormonal Control Different mammals have different patterns of reproduction Eg mammals, rats and mice can breed all year round, whereas
Reproduction and its Hormonal Control Page 1 Reproduction and its Hormonal Control Different mammals have different patterns of reproduction Eg mammals, rats and mice can breed all year round, whereas
ICH M3 (R2) Guideline on Nonclinical Safety Studies for the Conduct of Human Clinical Trials and Marketing Authorization for Pharmaceuticals
 ICH M3 (R2) Guideline on Nonclinical Safety Studies for the Conduct of Human Clinical Trials and Marketing Authorization for Pharmaceuticals International Conference on Harmonisation of Technical Requirements
ICH M3 (R2) Guideline on Nonclinical Safety Studies for the Conduct of Human Clinical Trials and Marketing Authorization for Pharmaceuticals International Conference on Harmonisation of Technical Requirements
Summary and conclusion 2013
 The work presented in the thesis is focused on the problems related to the prostate gland. Benign prostatic hyperplasia (BPH) and prostate cancer (PCa) are the two major problems associated with prostate.
The work presented in the thesis is focused on the problems related to the prostate gland. Benign prostatic hyperplasia (BPH) and prostate cancer (PCa) are the two major problems associated with prostate.
NATIONAL INSTITUTE OF ENVIRONMENTAL HEALTH SCEINCES Division of Extramural Research and Training
 NATIONAL INSTITUTE OF ENVIRONMENTAL HEALTH SCEINCES Division of Extramural Research and Training NATIONAL ADVISORY ENVIRONMENTAL HEALTH SCIENCES COUNCIL May 20, 2009 Introduction Concept Clearance For
NATIONAL INSTITUTE OF ENVIRONMENTAL HEALTH SCEINCES Division of Extramural Research and Training NATIONAL ADVISORY ENVIRONMENTAL HEALTH SCIENCES COUNCIL May 20, 2009 Introduction Concept Clearance For
In Silico Models: Risk Assessment With Non-Testing Methods in OSIRIS Opportunities and Limitations
 In Silico Models: Risk Assessment With Non-Testing Methods in OSIRIS Opportunities and Limitations Mark Cronin, Mark Hewitt, Katarzyna Przybylak School of Pharmacy and Chemistry Liverpool John Moores University
In Silico Models: Risk Assessment With Non-Testing Methods in OSIRIS Opportunities and Limitations Mark Cronin, Mark Hewitt, Katarzyna Przybylak School of Pharmacy and Chemistry Liverpool John Moores University
Tier 3 Toxicity Value White Paper
 U.S. ENVIRONMENTAL PROTECTION AGENCY OSWER 9285.7-86 Tier 3 Toxicity Value White Paper Regional Tier 3 Toxicity Value Workgroup OSWER Human Health Regional Risk Assessors Forum 5/16/2013 Disclaimer: This
U.S. ENVIRONMENTAL PROTECTION AGENCY OSWER 9285.7-86 Tier 3 Toxicity Value White Paper Regional Tier 3 Toxicity Value Workgroup OSWER Human Health Regional Risk Assessors Forum 5/16/2013 Disclaimer: This
Guidelines for Ecological Risk Assessment
 EPA/630/R-95/002F April 1998 Guidelines for Ecological Risk Assessment (Published on May 14, 1998, Federal Register 63(93):26846-26924) Risk Assessment Forum U.S. Environmental Protection Agency Washington,
EPA/630/R-95/002F April 1998 Guidelines for Ecological Risk Assessment (Published on May 14, 1998, Federal Register 63(93):26846-26924) Risk Assessment Forum U.S. Environmental Protection Agency Washington,
Frequently asked questions on the procedure for the re-assessment of glyphosate within the framework of the EU active substance review
 Frequently asked questions on the procedure for the re-assessment of glyphosate within the framework of the EU active substance review BfR FAQ, 12 November 2015 Active substances used in pesticides are
Frequently asked questions on the procedure for the re-assessment of glyphosate within the framework of the EU active substance review BfR FAQ, 12 November 2015 Active substances used in pesticides are
19/03/2012 Toxicologicalcharacterizationof PM10: case study Genk-Zuid. R. Van Den Heuvel
 19/03/2012 Toxicologicalcharacterizationof PM10: case study Genk-Zuid. R. Van Den Heuvel Introduction» Assessment of potential risk to human health associated with environmental air» Fysico-chemical characterization»
19/03/2012 Toxicologicalcharacterizationof PM10: case study Genk-Zuid. R. Van Den Heuvel Introduction» Assessment of potential risk to human health associated with environmental air» Fysico-chemical characterization»
Anatomy and Physiology of Human Reproduction. Module 10a
 This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike License. Your use of this material constitutes acceptance of that license and the conditions of use of materials on this
This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike License. Your use of this material constitutes acceptance of that license and the conditions of use of materials on this
ATRAZINE & HEALTH: KEY STUDIES
 ATRAZINE & HEALTH: KEY STUDIES December, 2009 Pesticide Action Network North America (PANNA) has compiled and summarized some of the key studies documenting the known and potential health effects of the
ATRAZINE & HEALTH: KEY STUDIES December, 2009 Pesticide Action Network North America (PANNA) has compiled and summarized some of the key studies documenting the known and potential health effects of the
Risikovurdering: Karsinogenitet kl 15.15 16.00 tirsdag 27 april
 Risikovurdering: Karsinogenitet kl 5.5 6.00 tirsdag 27 april Tore Sanner Institutt for Kreftforskning Det Norske Radiumhospital IN EU Category. Substances known to be carcinogenic to man. There is sufficient
Risikovurdering: Karsinogenitet kl 5.5 6.00 tirsdag 27 april Tore Sanner Institutt for Kreftforskning Det Norske Radiumhospital IN EU Category. Substances known to be carcinogenic to man. There is sufficient
MATERIAL SAFETY DATA SHEET
 Page 1 of 7 1. IDENTIFICATION OF THE SUBSTANCE/PREPARATION AND THE COMPANY/UNDERTAKING Pfizer Inc Pfizer Pharmaceuticals Group 235 East 42nd Street New York, New York 10017 1-212-573-2222 Emergency telephone
Page 1 of 7 1. IDENTIFICATION OF THE SUBSTANCE/PREPARATION AND THE COMPANY/UNDERTAKING Pfizer Inc Pfizer Pharmaceuticals Group 235 East 42nd Street New York, New York 10017 1-212-573-2222 Emergency telephone
Guidelines for Neurotoxicity Risk Assessment
 EPA/630/R-95/001F April 1998 Guidelines for Neurotoxicity Risk Assessment (Published on May 14, 1998, Federal Register 63(93):26926-26954) Risk Assessment Forum U.S. Environmental Protection Agency Washington,
EPA/630/R-95/001F April 1998 Guidelines for Neurotoxicity Risk Assessment (Published on May 14, 1998, Federal Register 63(93):26926-26954) Risk Assessment Forum U.S. Environmental Protection Agency Washington,
September 19, 1984 FOOD PRODUCTION AND DIRECTION GÉNÉRALE, SECTION INSPECTION BRANCH PRODUCTION ET INSPECTION PESTICIDES DES ALIMENTS TRADE MEMORANDUM
 Agriculture Canada September 19, 1984 T-1-245 FOOD PRODUCTION AND DIRECTION GÉNÉRALE, SECTION INSPECTION BRANCH PRODUCTION ET INSPECTION PESTICIDES DES ALIMENTS TRADE MEMORANDUM RE: Guidelines for Developing
Agriculture Canada September 19, 1984 T-1-245 FOOD PRODUCTION AND DIRECTION GÉNÉRALE, SECTION INSPECTION BRANCH PRODUCTION ET INSPECTION PESTICIDES DES ALIMENTS TRADE MEMORANDUM RE: Guidelines for Developing
REVIEW OF EXPOSURE LIMITS AND HEALTH CONCERNS SANTA ANA. Base Station Telecommunication Transmitters UNIFIED SCHOOL DISTRICT
 SANTA ANA UNIFIED SCHOOL DISTRICT REVIEW OF EXPOSURE LIMITS AND HEALTH CONCERNS Base Station Telecommunication Transmitters Dr. Karl Rodenbaugh, Senior Scientist SCOPE OF SERVICES Review, Describe, Discuss,
SANTA ANA UNIFIED SCHOOL DISTRICT REVIEW OF EXPOSURE LIMITS AND HEALTH CONCERNS Base Station Telecommunication Transmitters Dr. Karl Rodenbaugh, Senior Scientist SCOPE OF SERVICES Review, Describe, Discuss,
SUMMARY: This regulation establishes tolerances for residues of pendimethalin in or on
 This document is scheduled to be published in the Federal Register on 12/21/2015 and available online at http://federalregister.gov/a/2015-31655, and on FDsys.gov BILLING CODE 6560-50-P ENVIRONMENTAL PROTECTION
This document is scheduled to be published in the Federal Register on 12/21/2015 and available online at http://federalregister.gov/a/2015-31655, and on FDsys.gov BILLING CODE 6560-50-P ENVIRONMENTAL PROTECTION
Overview of Phase 1 Oncology Trials of Biologic Therapeutics
 Overview of Phase 1 Oncology Trials of Biologic Therapeutics Susan Jerian, MD ONCORD, Inc. February 28, 2008 February 28, 2008 Phase 1 1 Assumptions and Ground Rules The goal is regulatory approval of
Overview of Phase 1 Oncology Trials of Biologic Therapeutics Susan Jerian, MD ONCORD, Inc. February 28, 2008 February 28, 2008 Phase 1 1 Assumptions and Ground Rules The goal is regulatory approval of
M110.726 The Nucleus M110.727 The Cytoskeleton M340.703 Cell Structure and Dynamics
 of Biochemistry and Molecular Biology 1. Master the knowledge base of current biochemistry, molecular biology, and cellular physiology Describe current knowledge in metabolic transformations conducted
of Biochemistry and Molecular Biology 1. Master the knowledge base of current biochemistry, molecular biology, and cellular physiology Describe current knowledge in metabolic transformations conducted
8. REGULATIONS AND ADVISORIES
 ARSENIC 395 The international and national regulations and guidelines pertaining to arsenic and its metabolites in air, water, and other media are summarized in Table 8-1. ATSDR has not derived inhalation
ARSENIC 395 The international and national regulations and guidelines pertaining to arsenic and its metabolites in air, water, and other media are summarized in Table 8-1. ATSDR has not derived inhalation
MATERIAL SAFETY DATA SHEET
 Page 1 of 5 1. IDENTIFICATION OF THE SUBSTANCE/PREPARATION AND THE COMPANY/UNDERTAKING Pfizer Inc Pfizer Pharmaceuticals Group 235 East 42nd Street New York, New York 10017 1-212-573-2222 Emergency telephone
Page 1 of 5 1. IDENTIFICATION OF THE SUBSTANCE/PREPARATION AND THE COMPANY/UNDERTAKING Pfizer Inc Pfizer Pharmaceuticals Group 235 East 42nd Street New York, New York 10017 1-212-573-2222 Emergency telephone
3. Quantitative Risk Assessment of Combined Exposure to Food Contaminants and Natural Toxins
 3. Quantitative Risk Assessment of Combined Exposure to Food Contaminants and Natural Toxins Objectives: To perform probabilistic risk modelling (exposure as well as effects) of food contaminants and natural
3. Quantitative Risk Assessment of Combined Exposure to Food Contaminants and Natural Toxins Objectives: To perform probabilistic risk modelling (exposure as well as effects) of food contaminants and natural
Management and Disposal of Hazardous Pharmaceutical Waste
 Management and Disposal of Hazardous Pharmaceutical Waste The Defining Regulations Federal- The Defining Regulation 40 CFR 260-265 Resource Conservation and Recovery Act (RCRA) Federal regulation of the
Management and Disposal of Hazardous Pharmaceutical Waste The Defining Regulations Federal- The Defining Regulation 40 CFR 260-265 Resource Conservation and Recovery Act (RCRA) Federal regulation of the
THRESHOLD OF TOXICOLOGICAL CONCERN (TTC) IN RISK ASSESSMENT
 THRESHOLD OF TOXICOLOGICAL CONCERN (TTC) IN RISK ASSESSMENT Prof. Em.. Dr. Robert Kroes Institute for Risk Assessment Sciences (IRAS) Utrecht University The Netherlands PQRI December 5, 2005 1 THRESHOLD
THRESHOLD OF TOXICOLOGICAL CONCERN (TTC) IN RISK ASSESSMENT Prof. Em.. Dr. Robert Kroes Institute for Risk Assessment Sciences (IRAS) Utrecht University The Netherlands PQRI December 5, 2005 1 THRESHOLD
SYNCHRONIZATION OF CATTLE
 UNDER ESTRUS SYNCHRONIZATION OF CATTLE FS921C Robin Salverson, Extension Livestock Educator, Harding County, and George Perry, Extension Beef Reproduction and Management Specialist Reproductive failure
UNDER ESTRUS SYNCHRONIZATION OF CATTLE FS921C Robin Salverson, Extension Livestock Educator, Harding County, and George Perry, Extension Beef Reproduction and Management Specialist Reproductive failure
Name of Chemical: Tebufenpyrad Reason for Issuance: Unconditional Registration Date Issued: August 26, 2002
 United States Environmental Protection Agency Office of Prevention, Pesticides and Toxic Substances (7501C) Pesticide Fact Sheet Name of Chemical: Tebufenpyrad Reason for Issuance: Unconditional Registration
United States Environmental Protection Agency Office of Prevention, Pesticides and Toxic Substances (7501C) Pesticide Fact Sheet Name of Chemical: Tebufenpyrad Reason for Issuance: Unconditional Registration
Subject Index. Bariatric surgery, obesity management 134
 Subject Index Acromegaly, PCOS differential diagnosis 149, 150, 154, 155 Adipokines, see specific adipokines Adiponectin, metabolic syndrome role 41 43 Adolescents, PCOS diagnosis 16, 17 Adrenal hyperplasia,
Subject Index Acromegaly, PCOS differential diagnosis 149, 150, 154, 155 Adipokines, see specific adipokines Adiponectin, metabolic syndrome role 41 43 Adolescents, PCOS diagnosis 16, 17 Adrenal hyperplasia,
MATERIAL SAFETY DATA SHEET
 Page 1 of 7 1. IDENTIFICATION OF THE SUBSTANCE/PREPARATION AND THE COMPANY/UNDERTAKING Pfizer Inc Pfizer Pharmaceuticals Group 235 East 42nd Street New York, New York 10017 1-212-573-2222 Emergency telephone
Page 1 of 7 1. IDENTIFICATION OF THE SUBSTANCE/PREPARATION AND THE COMPANY/UNDERTAKING Pfizer Inc Pfizer Pharmaceuticals Group 235 East 42nd Street New York, New York 10017 1-212-573-2222 Emergency telephone
MATERIAL SAFETY DATA SHEET
 Page 1 of 7 1. IDENTIFICATION OF THE SUBSTANCE/PREPARATION AND THE COMPANY/UNDERTAKING Pfizer Inc Pfizer Pharmaceuticals Group 235 East 42nd Street New York, New York 10017 1-212-573-2222 Emergency telephone
Page 1 of 7 1. IDENTIFICATION OF THE SUBSTANCE/PREPARATION AND THE COMPANY/UNDERTAKING Pfizer Inc Pfizer Pharmaceuticals Group 235 East 42nd Street New York, New York 10017 1-212-573-2222 Emergency telephone
TESTIMONY OF DR. STEVEN BRADBURY DIRECTOR, OFFICE OF PESTICIDE PROGRAMS U.S. ENVIRONMENTAL PROTECTION AGENCY
 TESTIMONY OF DR. STEVEN BRADBURY DIRECTOR, OFFICE OF PESTICIDE PROGRAMS U.S. ENVIRONMENTAL PROTECTION AGENCY BEFORE THE SUBCOMMITTEE ON NUTRITION AND HORTICULTURE OF THE AGRICULTURE COMMITTEE AND SUBCOMMITTEE
TESTIMONY OF DR. STEVEN BRADBURY DIRECTOR, OFFICE OF PESTICIDE PROGRAMS U.S. ENVIRONMENTAL PROTECTION AGENCY BEFORE THE SUBCOMMITTEE ON NUTRITION AND HORTICULTURE OF THE AGRICULTURE COMMITTEE AND SUBCOMMITTEE
Combined effects of carcinogens
 Combined effects of carcinogens Institute of Environmental Medicine ulla.stenius@ki.se National Cancer Institute Ulla Stenius, IMM, KI 1 477 chemicals tested in NTP cancer test analyzed 278 rat carcinogens
Combined effects of carcinogens Institute of Environmental Medicine ulla.stenius@ki.se National Cancer Institute Ulla Stenius, IMM, KI 1 477 chemicals tested in NTP cancer test analyzed 278 rat carcinogens
Basics of Risk Assessment
 Basics of Risk Assessment Christopher J. Portier, Ph.D. Director National Center for Environmental Health Agency for Toxic Substances and Disease Registry International Conference on EMF and Health Brussels,
Basics of Risk Assessment Christopher J. Portier, Ph.D. Director National Center for Environmental Health Agency for Toxic Substances and Disease Registry International Conference on EMF and Health Brussels,
Programming effects of endocrine disrupting compounds in mice
 Programming effects of endocrine disrupting compounds in mice Joantine van Esterik 19 June 2014 1 Perinatal bisphenol A in mice 18 January 2011 Developmental Origins of Health and Disease (DOHaD) Adverse
Programming effects of endocrine disrupting compounds in mice Joantine van Esterik 19 June 2014 1 Perinatal bisphenol A in mice 18 January 2011 Developmental Origins of Health and Disease (DOHaD) Adverse
Chapter 12: SPECIFIC TARGET ORGAN SYSTEMIC TOXICITY (TOST) FOLLOWING A SINGLE EXPOSURE
 Chapter 12: SPECIFIC TARGET ORGAN SYSTEMIC TOXICITY (TOST) FOLLOWING A SINGLE EXPOSURE DEFINITIONS 1. Classification identifies the chemical substance as being a specific target organ/systemic toxicant
Chapter 12: SPECIFIC TARGET ORGAN SYSTEMIC TOXICITY (TOST) FOLLOWING A SINGLE EXPOSURE DEFINITIONS 1. Classification identifies the chemical substance as being a specific target organ/systemic toxicant
3M HB Quat Disinfectant Cleaner Ready-to-Use (Product No. 25, 3M Chemical Management Systems) 09/15/14
 Safety Data Sheet Copyright, 2014, 3M Company. All rights reserved. Copying and/or downloading of this information for the purpose of properly utilizing 3M products is allowed provided that: (1) the information
Safety Data Sheet Copyright, 2014, 3M Company. All rights reserved. Copying and/or downloading of this information for the purpose of properly utilizing 3M products is allowed provided that: (1) the information
Risk Assessment for Chemicals in Drinking Water
 Brochure More information from http://www.researchandmarkets.com/reports/2181361/ Risk Assessment for Chemicals in Drinking Water Description: A comprehensive reference on state of the art risk assessment
Brochure More information from http://www.researchandmarkets.com/reports/2181361/ Risk Assessment for Chemicals in Drinking Water Description: A comprehensive reference on state of the art risk assessment
Overview: Human Health Effects of Environmental Contaminants
 Overview: Human Health Effects of Environmental Contaminants 1 1 OVERVIEW: HUMAN HEALTH EFFECTS OF ENVIRONMENTAL CONTAMINANTS Human activity has led to the production and release into the environment of
Overview: Human Health Effects of Environmental Contaminants 1 1 OVERVIEW: HUMAN HEALTH EFFECTS OF ENVIRONMENTAL CONTAMINANTS Human activity has led to the production and release into the environment of
Harmonization Project Document No. 2
 This report contains the collective views of an international group of experts and does not necessarily represent the decisions or the stated policy of the World Health Organization, the International
This report contains the collective views of an international group of experts and does not necessarily represent the decisions or the stated policy of the World Health Organization, the International
Systematic review by: Dr. Ashraf Ahmed ElDaly, M.Sc., M.D.
 Role of luteinizing hormone supplementation in the follicular phase and pregnancy, during controlled ovarian hyperstimulation in in vitro fertilization Systematic review by: Dr. Ashraf Ahmed ElDaly, M.Sc.,
Role of luteinizing hormone supplementation in the follicular phase and pregnancy, during controlled ovarian hyperstimulation in in vitro fertilization Systematic review by: Dr. Ashraf Ahmed ElDaly, M.Sc.,
Basic Overview of Preclinical Toxicology Animal Models
 Basic Overview of Preclinical Toxicology Animal Models Charles D. Hebert, Ph.D., D.A.B.T. December 5, 2013 Outline Background In Vitro Toxicology In Vivo Toxicology Animal Models What is Toxicology? Background
Basic Overview of Preclinical Toxicology Animal Models Charles D. Hebert, Ph.D., D.A.B.T. December 5, 2013 Outline Background In Vitro Toxicology In Vivo Toxicology Animal Models What is Toxicology? Background
MATERIAL SAFETY DATA SHEET
 Page 1 of 7 1. IDENTIFICATION OF THE SUBSTANCE/PREPARATION AND THE COMPANY/UNDERTAKING Pfizer Inc Pfizer Pharmaceuticals Group 235 East 42nd Street New York, New York 10017 1-212-573-2222 Emergency telephone
Page 1 of 7 1. IDENTIFICATION OF THE SUBSTANCE/PREPARATION AND THE COMPANY/UNDERTAKING Pfizer Inc Pfizer Pharmaceuticals Group 235 East 42nd Street New York, New York 10017 1-212-573-2222 Emergency telephone
Tech Prep Articulation
 Tech Prep Articulation Agriculture & Natural Resources Tech Prep Education: Tech Prep education in Missouri is an articulated two-year secondary and two or more year post-secondary education program which:
Tech Prep Articulation Agriculture & Natural Resources Tech Prep Education: Tech Prep education in Missouri is an articulated two-year secondary and two or more year post-secondary education program which:
Chapter 45: Hormones and the Endocrine System
 Name Period Overview 1. What is a hormone? 2. Why does a hormone elicit a response only with target cells? 3. The body has two long-distance regulating systems. Which involves chemical signals by hormones?
Name Period Overview 1. What is a hormone? 2. Why does a hormone elicit a response only with target cells? 3. The body has two long-distance regulating systems. Which involves chemical signals by hormones?
Glyphosate Levels in Breakfast Foods: What is safe?
 Glyphosate Levels in Breakfast Foods: What is safe? The Alliance for Natural Health USA April 19, 2016 Glyphosate is the active ingredient in the world s most widely used herbicide, its use being largely
Glyphosate Levels in Breakfast Foods: What is safe? The Alliance for Natural Health USA April 19, 2016 Glyphosate is the active ingredient in the world s most widely used herbicide, its use being largely
ALPHABETICAL LIST OF SELECTED GENERIC TERMS IN HAZARD AND RISK ASSESSMENT AND THEIR DEFINITIONS. Related terms: Reference Dose, Tolerable Daily Intake
 ALPHABETICAL LIST OF SELECTED GENERIC TERMS IN HAZARD AND RISK ASSESSMENT AND THEIR DEFINITIONS Term Acceptable Daily Intake Acceptable Risk Adverse Effect Analysis Assessment Assessment Endpoint Assessment
ALPHABETICAL LIST OF SELECTED GENERIC TERMS IN HAZARD AND RISK ASSESSMENT AND THEIR DEFINITIONS Term Acceptable Daily Intake Acceptable Risk Adverse Effect Analysis Assessment Assessment Endpoint Assessment
Answer Key. Vocabulary Practice
 Answer Key Vocabulary Practice Copyright by McDougal Littell, a division of Houghton Mifflin Company A. Categorize Words 1. organism, L; cell, L; species, L; transgenic, B; biotechnology, T; molecular
Answer Key Vocabulary Practice Copyright by McDougal Littell, a division of Houghton Mifflin Company A. Categorize Words 1. organism, L; cell, L; species, L; transgenic, B; biotechnology, T; molecular
The Menstrual Cycle. Model 1: Ovarian Cycle follicular cells
 The Menstrual Cycle REVIEW questions to complete before starting this POGIL activity 1. Gonads produce both gametes and sex steroid hormones. For the female, name the: A. gonads ovaries B. gametes oocyte/ovum/egg
The Menstrual Cycle REVIEW questions to complete before starting this POGIL activity 1. Gonads produce both gametes and sex steroid hormones. For the female, name the: A. gonads ovaries B. gametes oocyte/ovum/egg
Bayer Environmental Science
 Revision Date: 07/02/2008 SECTION 1. CHEMICAL PRODUCT AND COMPANY INFORMATION Product Name READY-TO-SPREAD GRANULES MSDS Number 102000016526 EPA Registration No. 72155-83 Bayer Environmental Science 2
Revision Date: 07/02/2008 SECTION 1. CHEMICAL PRODUCT AND COMPANY INFORMATION Product Name READY-TO-SPREAD GRANULES MSDS Number 102000016526 EPA Registration No. 72155-83 Bayer Environmental Science 2
NONCLINICAL EVALUATION FOR ANTICANCER PHARMACEUTICALS
 INTERNATIONAL CONFERENCE ON HARMONISATION OF TECHNICAL REQUIREMENTS FOR REGISTRATION OF PHARMACEUTICALS FOR HUMAN USE ICH HARMONISED TRIPARTITE GUIDELINE NONCLINICAL EVALUATION FOR ANTICANCER PHARMACEUTICALS
INTERNATIONAL CONFERENCE ON HARMONISATION OF TECHNICAL REQUIREMENTS FOR REGISTRATION OF PHARMACEUTICALS FOR HUMAN USE ICH HARMONISED TRIPARTITE GUIDELINE NONCLINICAL EVALUATION FOR ANTICANCER PHARMACEUTICALS
What is Cancer? Cancer is a genetic disease: Cancer typically involves a change in gene expression/function:
 Cancer is a genetic disease: Inherited cancer Sporadic cancer What is Cancer? Cancer typically involves a change in gene expression/function: Qualitative change Quantitative change Any cancer causing genetic
Cancer is a genetic disease: Inherited cancer Sporadic cancer What is Cancer? Cancer typically involves a change in gene expression/function: Qualitative change Quantitative change Any cancer causing genetic
Endocrine System: Practice Questions #1
 Endocrine System: Practice Questions #1 1. Removing part of gland D would most likely result in A. a decrease in the secretions of other glands B. a decrease in the blood calcium level C. an increase in
Endocrine System: Practice Questions #1 1. Removing part of gland D would most likely result in A. a decrease in the secretions of other glands B. a decrease in the blood calcium level C. an increase in
Safety Data Sheet. Document Group: 10-4849-5 Version Number: 11.01 Issue Date: 05/26/15 Supercedes Date: 10/09/14
 3M FT-10 Qualitative Fit Test Kit, Sweet 05/26/15 Safety Data Sheet Copyright,2015,3M Company. All rights reserved. Copying and/or downloading of this information for the purpose of properly utilizing
3M FT-10 Qualitative Fit Test Kit, Sweet 05/26/15 Safety Data Sheet Copyright,2015,3M Company. All rights reserved. Copying and/or downloading of this information for the purpose of properly utilizing
Endocrine Disruption Injury Claims
 Science and Strategy By Benjamin R. Dwyer and Samuel Goldblatt Endocrine Disruption Injury Claims Defense counsel should recognize that the novel theories advanced by proponents of these claims do not
Science and Strategy By Benjamin R. Dwyer and Samuel Goldblatt Endocrine Disruption Injury Claims Defense counsel should recognize that the novel theories advanced by proponents of these claims do not
Safety Data Sheet. SECTION 1: Identification. SECTION 2: Hazard identification
 Safety Data Sheet Copyright,2016,3M Company. All rights reserved. Copying and/or downloading of this information for the purpose of properly utilizing 3M products is allowed provided that: (1) the information
Safety Data Sheet Copyright,2016,3M Company. All rights reserved. Copying and/or downloading of this information for the purpose of properly utilizing 3M products is allowed provided that: (1) the information
2007 WHO Research Agenda for Extremely Low Frequency Fields
 2007 WHO Research Agenda for Extremely Low Frequency Fields Introduction In 1997, the WHO International EMF Project developed a Research Agenda in order to facilitate and coordinate research worldwide
2007 WHO Research Agenda for Extremely Low Frequency Fields Introduction In 1997, the WHO International EMF Project developed a Research Agenda in order to facilitate and coordinate research worldwide
Human Risk Assessment
 UNEP/IPCS Training Module No. 3 Section A Human Risk Assessment Prepared by The Edinburgh Centre for Toxicology UNEP/IPCS TRAINING MODULE SECTION A Human Risk Assessment TABLE OF CONTENTS PAGE EDUCATIONAL
UNEP/IPCS Training Module No. 3 Section A Human Risk Assessment Prepared by The Edinburgh Centre for Toxicology UNEP/IPCS TRAINING MODULE SECTION A Human Risk Assessment TABLE OF CONTENTS PAGE EDUCATIONAL
From Menses to Menopause: How Hormones Can Affect Blood Glucose Levels. Christine Day, RN, MS, CNS-BC Lake Superior College
 From Menses to Menopause: How Hormones Can Affect Blood Glucose Levels Christine Day, RN, MS, CNS-BC Lake Superior College Overview Will review hormonal changes over the female lifespan Discuss the effects
From Menses to Menopause: How Hormones Can Affect Blood Glucose Levels Christine Day, RN, MS, CNS-BC Lake Superior College Overview Will review hormonal changes over the female lifespan Discuss the effects
Reproductive System & Development: Practice Questions #1
 Reproductive System & Development: Practice Questions #1 1. Which two glands in the diagram produce gametes? A. glands A and B B. glands B and E C. glands C and F D. glands E and F 2. Base your answer
Reproductive System & Development: Practice Questions #1 1. Which two glands in the diagram produce gametes? A. glands A and B B. glands B and E C. glands C and F D. glands E and F 2. Base your answer
DiNP Dose-Response Studies:
 DiNP Dose-Response Studies: Gestation PK and Developmental Effects Postnatal Effects Final Project Summary October 2011 Rebecca Clewell, PhD Research Investigator Institute for Chemical Safety Sciences
DiNP Dose-Response Studies: Gestation PK and Developmental Effects Postnatal Effects Final Project Summary October 2011 Rebecca Clewell, PhD Research Investigator Institute for Chemical Safety Sciences
Fluoride. Introduction
 Fluoride Introduction Most fluoride in the body is found in bones and teeth, due to its high affinity for calcium and calcium phosphate. Ingestion of and topical treatment with fluoride is effective in
Fluoride Introduction Most fluoride in the body is found in bones and teeth, due to its high affinity for calcium and calcium phosphate. Ingestion of and topical treatment with fluoride is effective in
We continue to believe that ICCVAM should focus its limited resources on methods that have applicability to more than one member agency.
 June 25, 2009 Dr Mary S. Wolfe National Institute of Environmental Health Sciences PO Box 12233, MD A3-01 Research Triangle Park, NC 27709 Re: 74 FR 19562; April 29, 2009; National Toxicology Program (NTP);
June 25, 2009 Dr Mary S. Wolfe National Institute of Environmental Health Sciences PO Box 12233, MD A3-01 Research Triangle Park, NC 27709 Re: 74 FR 19562; April 29, 2009; National Toxicology Program (NTP);
RADIOPHARMACEUTICALS BASED ON MONOCLONAL ANTIBODIES
 RADIOPHARMACEUTICALS BASED ON MONOCLONAL ANTIBODIES Guideline Title Radiopharmaceuticals based on Monoclonal Antibodies Legislative basis Directives 65/65/EEC, 75/318/EEC as amended, Directive 89/343/EEC
RADIOPHARMACEUTICALS BASED ON MONOCLONAL ANTIBODIES Guideline Title Radiopharmaceuticals based on Monoclonal Antibodies Legislative basis Directives 65/65/EEC, 75/318/EEC as amended, Directive 89/343/EEC
