PATENTS ACT t970 SECTION 15 DECISION
|
|
|
- Alice Thomas
- 10 years ago
- Views:
Transcription
1 PATENTS ACT t970 SECTION 15 IN THE MATTER OF AN APPLICATION FOR PATENT, NUMBER I752/CIJW2OO6 FILED ON 25th SEPTE,MBER M / s. GLYCOSCIENCE LABORATORIES, INC, JAPAN... The Applicants. HEARING HELD ON 23'd JULY.2OO9 Present: Ms. Vaishnavi S K of DePenning & DePenning, Agent for the Applicants. Mr. R. Rajini, Examiner of Patents & Designs DECISION An application for Patent for an invention titled "PHARMACEUTICAL AGENT CONTAINING HYALURONAN AS AN ACTIVE INGREDIENT', WAS fi Ed by M / s. DePenning & DePenning on behalf of M / s. GLYCOSCIENCE LABORATORIES, INC, JAPAN and the same was allotted convention Appfication Number. 1752!CHE12OO6 dated 25th September The applicants have also claimed priority from Japanese patent applications identified as dated 26th September 2005, dated 12th October 2005 and dated 01't December 2005.
2 The complete specification of the instant application at the time of filing had claims as follows: 1' A therapeutic or prophyractic agent for an inframmation and a neural dysfunction, comprising hyaruronan as an active ingredient. 2' The therapeutic or prophylactic agent according to Claim 1, wherein said hyaluronan is a tetrasaccharide containing 2 units, with a singre unit being _D_ glucuronic acid- p -1,3-D_N_acetylglucosamine_ 13 _1,4_. 3. The therapeutic or inflammation and the prophylactic agent according to craim 1, wherein the neural dysfunction is due to an autoimmune disease. 4' The therapeutic or prophylactic agent according to Claim 1, wherein the inflammation and the neurar dysfunction is due to a neurar disease. 5' The therapeutic or prophylactic agent according to Claim 1, wherein the inflammation and the neurar dysfunction is due to spinar cord injury. 6. The therapeutic or prophylactic agentaccording to Claim 1, wherein the inflammation and the neural dysfunctionis due to asthma. 7. The therapeutic or prophylactic agent according to claim 1, wherein the inflammation and the neural dysfunction is due to a murtiple sclerosis. 8' A method for treating or preventing an inflammation comprising a step of administering an effective amount of hyaluronan to a subject in need of a treatment.
3 g. The method for treating or preventing an autoimmune disease according to Claim 8, wherein said hyaluronan is a tetrasaccharide containing 2 units, with a single unit being -D-glucuronic acid-b -1,3-D-N-acetylglucosamine-B -1, The method for treating or preventing an autoimmune disease according to Claim B, wherein the inflammation and the neural dysfunction is due to an autoimmune disease. 11. The method for treating or preventing an autoimmune disease according to Claim 8, wherein the inflammation and the neural dysfunction is due to a neural disease. 12. The method for treating or preventing an autoimmune disease according to Claim B, wherein the inflammation and the neural dysfunction is due to a spinal cord injury. 13. The method for treating or preventing an autoimmune disease according to Claim 8, wherein the inflammation and the neural dysfunction is due to asthma. 14. The method for treating or preventing an autoimmune disease according to Claim 8, wherein the inflammation and the neural dysfunction is due to a multiple sclerosis. 15. A cytokine-associated gene expression inhibitor comprising hyaluronan as an active ingredient. 16. The cytokine-associated gene expression inhibitor according to Claim 15, wherein said hyaluronan is a tetrasaccharide containing 2 units, with a single unit being -D-glucuronic acid-b -1,3-D-N-acetylglucosamine-B -1,4-'
4 17. The cytokine-associated gene expression inhibitor according to Claim15, wherein said cytokine-associated gene is a gene associated with an inflammatory cytokine. 18' The cytokine-associated gene expression inhibitor according to Claim15 which is an injection formulation and an oral formulation. 19' A chemokine-associated gene expression inhibitor comprising hyaluronan as an active ingredient. 20' The chemokine-associated gene expression inhibitor according to Claim 1g, wherein said hyaluronan is a tetrasaccharide containing 2 units, with a single unit being -D-glucuronic acid-b -1,3-D-N-acetylglucosamine_B _1,4_. 21. The chemokine-associated gene expression inhibitor according to Claim 1g, which is an injection formulation and an oral formulation. 22' A cell viability enhancer comprising hyaluronan as an active ingredient. 23' The cell viability enhancer according to Claim 22, wherein said hyaluronan is a tetrasaccharide containing 2 units, with a single unit being -D-glucuronic acid-b -1,3-D-N-acetylglucosamine-B -1, fhe cell viability enhancer according to Claim 23 which is an oral formulation. 25. A synaptic transmission promoter, comprising hyaluronan as an active ingredient. 26' The synaptic transmission promoter according to Claim 25, wherein said hyaluronan is a tetrasaccharide containing 2 units, with a single unit being -Dglucuronic acid-b -1,3-D-N-acetylglucosamine-B -1,4-.
5 27.The synaptic transmission promoter according to Claim 25 which is an intraduralformulation, a subcutaneous formulation, an intravenous formulation or an oralformulation. 28. A synaptic protector comprising hyaluronan as an active ingredient. 29. The synaptic protector according to Claim 28, wherein said hyaluronan is a tetrasaccharide containing 2 units, with a single unit being -D-glucuronic acid-b - 1,3-D-N-acetylglucosamine-B -1, The synaptic protector according to Claim 28 which is an intradural formulation, a subcutaneous formulation, an intravenous formulation, intranasal or an oral formulation. The request for examination was filed by the agent on 25'n September 2006 and the same was examined under the provision of section 12 of the patents Act, The First Examination Report was issued to the agent on 07th July 2008 which inter-alia included objections lack of novelty, inventive step, and under section 3(d), (i) and (e). The agents submitted their reply to the First Examination Report on 19th June 2009 wherein they amended the claims as, 1. A medicament for intraspinal administration for the treatment of an inflammation due to a multiple sclerosis or a neural dysfunction due to a multiple sclerosis comprising a tetrasaccharide containing 2 units, with a single unit being D-glucuronic acid-b-1,3-d-n-acetylglucosamine-b-1,4-' for the manufacture of a medicament.
6 The medicament as claimed in patient. The medicament as claimed in administration once a day. The medicament as claimed in any preceding claims, wherein it is an injection formulation containing a ph modifier, an osmotic agent and distilled water for injection. The medicament as claimed in claim 4, wherein the ph modifier is selected from the group consisting of hydrochloric acid, sodium hydroxide, lactic acid, sodium lactate, sodium monohydrogen phosphate and sodium dihydrogen phosphate" The medicament as claimed in claims 4 or 5, wherein the osmotic agent is sodium chloride or glucose. The medicament as claimed in any of claims 4 to 6, wherein the injection formulation is supplemented also with mannitol, dextrin, cyclodextrin or gelatin. 8. The medicament as claimed in any of claims 1 to 3, wherein the medicament is an emulsion for injection containing an emulsifier and water. 9. The medicament as claimed in claim 8, wherein the emulsifier is selected from the group consisting of lecithin, polysorbate 80, and polyethylene hydrogenated castor oil. claim 1, wherein it contains the tetrasaccharide in a dosage of from0.05 to 50 mg/kg body weight of the claims 1 or 2, wherein it is for The agents also requested for an opportunity of being heard before any decision adverse to the applicant is taken. As requested by the agents, a hearing was offered and held on 23'd July During the hearing, the agent for the applicants submitted that "in CA (D1), nerve damage due to spinal cord'injury'disclosed D1, and a neural dysfunction due to 'disease' such as multiple sclerosis, which is disclosed
7 in the present specification, are different in their pathophysiologies. D1 discloses well-known examples of neural dysfunction including multiple sclerosis; however, their pathogenic mechanisms are completely different. The description of D1 is incorrect with no scientific basis. The differences between multiple sclerosis and traumatic spinal cord injury are as follows: Multiple sclerosis Traumatic spinal co Category Autoimmune disease Traumatic iniu IFN-beta effective ineffective T-cell invasion + Animal models EAE model Injury Regarding the category of disease, multiple sclerosis is classified as an autoimmune disease and traumatic spinal cord injury is classified as trauma. The immune-mediated pathology as seen in multiple sclerosis is not typical for traumatic spinal cord injury. Regarding the effective treatment, INF-beta is commonly used for the treatment of multiple sclerosis, whereas INF-beta is ineffective for traumatic spinal cord injury. Regarding T-cell invasion, the most common form of multiple sclerosis is what is known as relapsing-remitting, and myelinated axons are destroyed as consequence of T-cell invasion in relapsing phase. On the other hand, traumatic spinal cord injury shows neither T cell invasion nor demyelination. Regarding animal models, experimental allergic encephalomyelitis (EAE) is widely used as a model of multiple sclerosis, which is induced by stimulating T- cell mediated immunity to myelin proteins via immunization with myelin antigens. On the other hand, crushing the spinal cord using tweezers produces traumatic injury. Only the therapeutic efficacy of intradural administration of HA for traumatic spinal cord injury is supported with the working examples of D1, and
8 D1 neither discloses nor suggests the therapeutic efficacy of HA4 for autoimmune disease such as multiple sclerosis. D1 does not disclose the mechanism of action of HA4 in the treatment of traumatic spinal cord injury. However, this efficacy may be due to promotion of the heat shock protein expression and inhibition of apoptosis. None of the references disclose the therapeutic efficacy of intraspinal administration of HA4 for multiple sclerosis. Only DG discloses the therapeutic efficacy of HA for multiple sclerosis. However, the pharmaceutical composition of D6 comprises a high-molecular-weight Zn-HA complex, which is a different kind of compound from HA without Zn. HA4 is more effective than high-molecularweight HA. Further, the therapeutic efficacy of HA4 for multiple sclerosis is unforeseen and is remarkable. None of the references disclose the therapeutic efficacy of intraspinal administration of HA4 for multiple sclerosis. The claimed medicament is as such novel and inventive". Therefore, they requested to reconsider the objection favorably and allow the application to proceed to grant. follows, After hearing all the arguments, it was explained to the agent as Claims do not involve inventive step under section 2(1)( ja ) of the patents Act Principal claim is directed to "A medicament for intraspinal administration for the treatment of an inflammation due to a multiple sclerosis or a neural dysfunction due to a multiple sclerosis comprising a tetrasaccharide containing 2 units, with a single unit being D-glucuronic acid-b-1,3-d-n_ acetylglucosamine-b-1,4-, for the manufacture of a medicament". Though the claim is directed to medicament the one and the only active ingredient is HA4 (tetarasaccharide containing 2 units, with a single unit being D-glucuronic acid-b- 1, 3-D-N-acetylglucosamine-B-1, 4-,).tnjection formulation of the medicament
9 is a conventional formulation. Excipients used in the formulation are inactive substances used as a carrier for the active ingredients of a medication' Activity of the medicament is only due to the presence of the active ingredient i'e'' HA4' Therefore claims relate to mere application / use' compound HA4 is well known from prior art. For example ca (family memberof wo ) and JP , were cited in the First Examination Report itself. The same has been accepted by the instant application. ln page 3 of the instant specification, it is given that "HA4 was reported to have therapeutic and inhibitory effect in an organ preservation' hepatic disorder and gastric ulcer (see, wo 2oo )' HA4 is also known to have a stress protein expression enhancing effect and a cell death inhibiting effect... In addition, HA4 is known to be effective therapeutically in a spinal cord injured model (see, " ' HA4 i.e., tetrasaccharide is well known from ca ' In page 5 it is given thatlhe glycoside bond between GtcA and GlcNAc is preferably a F 1-3 bond, and the glycosidic bond between GlcNAc and GlcA is preferabty F 1-4 bond'. In page 1 it is given that GlcA is glucuronic acid and GlcNAc is N- acety g ucosamine. npagel0firstparagraphitisgiventhatlow molecular-weight saccharide composed of at least GlcA and/or G[cNAc or a pharmaceutically acceptable salt thereof may be 100 micro g to 1000 mg per adult per administration. ln page 7 last paragraph it is given that 'the administration method of the therapeutic agent of the present invention is not particularly limited as long as the therapeutic agent of the present invention can exert an effect on nerve damage. Examples of administration routes include injection (intradural, intravenous, intramuscular, subcutaneous' intraculataneous, intraperitoneal, or the likel, transnasal' oral' percutaneous, and inhalation. The administration method such as direct administration by injection to a certain site or drip administration is appropriately selected depending on a disease or a site to be applied' ln the case of intradural administration or the like, an implantable pump for drug infusion may be
10 implanted in the body to per{orm continuous administration'. In page B it is given that'the liquid formulation may be produced by dissolving a low molecular-weight saccharide composed of at least GlcA and/or GlcNAc or a pharmaceutically acceptable salt thereof, for example, an appropriate aqueous solvent or a solvent commonly used for drugs. Example of such solvents include distilled water, buffers, physiological saline and water containing a water-miscible organic solvent or the like'. ln the case that the therapeutic agent of the present invention is provided as an injectable agent, its form may be a solution, frozen product, or freeze-dried product. The therapeutic agent is filled and sealed in an appropriate container such as an ampule, vial or syringe for injection, for distribution or preservation, and it may be administered as an iniection. For formulating the therapeutic agent of the present invention, a known method may be used. When the treatment agent is formulated, other active ingredients (such as anti-inflammatory drugs, analgesics, vitamin preparations, antibacterial agents, growth factors and adhesion factors), or ingredients generally used in medicines such as conventional stabilizing agents, emulsifiers, osmotic regutators, ph regulators, buffers, tonicity agents, preservatives, soothing agents, colorants, diluents, binders, lubricants and disintegrators may be used, as long as those ingredients exert no unfavorable influence on the above described saccharide or a pharmaceutically acceptable salt thereof and exert no influence on the effects of the present invention'. In page 1 paragraph 2, it is given that JP discloses an aqueous solution containing hyaluronic acid (HA) or a pharmaceutically acceptable salt thereof for spinal cord perfusion, and it describes that the solution for perfusion may be used in the spinal cord perfusion therapy for spinal cord injury...'. In page 9 paragraph 2 it is given that "the'condition where a treatment for nerve damage is desired' is not particularly limited, but examples thereof include spinal cord injury or nerve trauma such as head trauma, cerebral (infantile) paralysis, spinal vascular damage, cervical spondylosis, senile dementia, Alzheimer's disease, Parkinson's disease, and spinocerebellar degeneration (hereditary spastic paraparesis). Of these' the treatment agent is preferably applied to spinal cord injury include traumatic spinal l0
11 cord injury, vertebral degenerative disease (spondylosis or the like), vetevral inflammatory disease (spondylitis, chronic rheumatoid arthritis or the like), tumor (spinal corci tumor, vertebral tumor or the like), vascular disease (spinal cord bleeding, cerebral embolism, spinal paralysis caused by extramedullary vascular damage or the like), myelitis (arachnoiditis, viral myelitis bacterial myelitis, or the like), multiple sclerosis, and amyotrophic lateral sclerosis. In particular, the therapeutic agent is effective for traumatic spinal cord injury". EP discloses the claimed hyaluronic acids as being Hsp expression promoting agents responsible for the inhibition of cell death, cell injury, tissue protection ll- 10 production promoter and ll-8 production inhibitor. JP discloses hyaluronic acids as being stress protein expression enhancer, cell lesion suppressor and cell death suppressor. WO and US disclose hyaluronic acid as being inhibitors of T-cells. WO disclose pharmaceutical composition comprising a Zinc-hyaluronate complex for the treatment of multiple sclerosis" From the above discussion it is clear that the compound HA4, its therapeutic activity, quantity for administration, administration routes, injection formulation, use in spinal cord perfusion therapy, use in spinal cord injury, use for nerve damage and use for multiple sclerosis are known. From the above disclosure, it is obvious to the person skilled in the art to use the same known compound for the same known use for multiple sclerosis or neural dysfunction by varying the quantity and administering by different route such as intraspinal administration. Therefore, claims do not constitute an invention under section 2(1)( j ) of the Patents Act In connection with the above argument, claims relate to new use for a known compound therefore fall within the scope of section 3(d) of the Patents Act {Section 3 (d): the mere discovery of a new form of a known substance which does not result in the enhancement of the known efficacy of that substance or the mere discovery of any new property or new use for a known l1
12 substance or of the mere use of a known process, machine or apparatus unlessuch known process results in a new product or employs at least one new reactant. Explanation: For the purposes of this clause, salts, esters, ethers, polymorphs, metabolites, pure form, particle size, isomers, mixtures of isomers, complexes, combinations and other derivatives of known substance shall be considered to be the same substance, unless they differ significantly in properties with regard to efficacy;). From the above disclosure it is clear that the compound HA4, its therapeutic activity, quantity for administration, administration routes, injection formulation, use in spinal cord perfusion therapy, use in spinal cord injury, use for nerve damage and use for multiple sclerosis are known from the prior art" Medicament for intraspinal administration for the treatment of an inflammation due to a multiple sclerosis or a neural dysfunction due to a multiple sclerosis of instant application is a new use for known compound, which fall within the scope of section 3(d) of the Patents Act 1970, therefore cannot be allowed. ln the seven examples given in the specification, not even a single injection formulation has been disclosed as claimed in the instant application. The examples must contain the actual medicament formulation with exact ratio/weight o/o of the excipients for which the test has been carried out. The invention and its operation or use and the method by which it is to be performed are not fully and particularly described in the complete specification. The complete specification should disclose the best method of performing the invention, which is known to the applicant and for which he is entitled to claim protection. Also claims do not sufficiently define the invention for which protection is claimed. Claims relate to mere application/use. The presently claimed medicament does not disclose any injection formulation explicitly with the ratio/weight % of the excipients. Therefore the complete specification do not t2
13 1970. requirementsunder section 10 (4) (a), (b) and (c) of the Patents Act ln connection with the above arguments, claims are not fairly based on the matter discrosed in the specification. And therefore do not meet the requirements under section 10 (5) of the Patents Act 1970' Therefore, l refuse to proceed with this application for Patent, No' 1752 CHE 2O06 u nder section15 of the Patents Act, Dated this 06th day of August 2009' (8. AHILAN) Assistant Controtler of Patents & Designs Copy to: DePenning and DePenning' 31 South Bank Road, Chennai ' IJ
Clinical and Therapeutic Cannabis Information. Written by Cannabis Training University (CTU) All rights reserved
 Clinical and Therapeutic Cannabis Information Written by Cannabis Training University (CTU) All rights reserved Contents Introduction... 3 Chronic Pain... 6 Neuropathic Pain... 8 Movement Disorders...
Clinical and Therapeutic Cannabis Information Written by Cannabis Training University (CTU) All rights reserved Contents Introduction... 3 Chronic Pain... 6 Neuropathic Pain... 8 Movement Disorders...
Demystifying Stem Cells. Brent Bost M.D., CPA, MBA, FACOG
 Demystifying Stem Cells Brent Bost M.D., CPA, MBA, FACOG Disclaimers Author currently serves on Board of Directors of CordTrack Most applications discussed today are not currently FDA approved and are,
Demystifying Stem Cells Brent Bost M.D., CPA, MBA, FACOG Disclaimers Author currently serves on Board of Directors of CordTrack Most applications discussed today are not currently FDA approved and are,
Understanding How Existing and Emerging MS Therapies Work
 Understanding How Existing and Emerging MS Therapies Work This is a promising and hopeful time in the field of multiple sclerosis (MS). Many new and different therapies are nearing the final stages of
Understanding How Existing and Emerging MS Therapies Work This is a promising and hopeful time in the field of multiple sclerosis (MS). Many new and different therapies are nearing the final stages of
 CAMBRIDGE UNIVERSITY CENTRE FOR BRAIN REPAIR A layman's account of our scientific objectives What is Brain Damage? Many forms of trauma and disease affect the nervous system to produce permanent neurological
CAMBRIDGE UNIVERSITY CENTRE FOR BRAIN REPAIR A layman's account of our scientific objectives What is Brain Damage? Many forms of trauma and disease affect the nervous system to produce permanent neurological
Coventry Health Care of Georgia, Inc. Coventry Health and Life Insurance Company
 Coventry Health Care of Georgia, Inc. Coventry Health and Life Insurance Company PRESCRIPTION DRUG RIDER This Prescription Drug Rider is an attachment to the Coventry Health Care of Georgia, Inc. ( Health
Coventry Health Care of Georgia, Inc. Coventry Health and Life Insurance Company PRESCRIPTION DRUG RIDER This Prescription Drug Rider is an attachment to the Coventry Health Care of Georgia, Inc. ( Health
the product for which protection was sought as "Centoxin (HA-lA Monoclonal Antibody";
 COUNCIL REGULATION (EEC) No 1768/92 IN TiiE MATTER OF Application No SPC/GB93/172 for a Supplementary Protection Certificate in the name of Centocor, Inc DECISION Application No SPC/GB93/172 for a Supplementary
COUNCIL REGULATION (EEC) No 1768/92 IN TiiE MATTER OF Application No SPC/GB93/172 for a Supplementary Protection Certificate in the name of Centocor, Inc DECISION Application No SPC/GB93/172 for a Supplementary
Intrathecal Baclofen for CNS Spasticity
 Intrathecal Baclofen for CNS Spasticity Last Review Date: November 13, 2015 Number: MG.MM.ME.31bC5 Medical Guideline Disclaimer Property of EmblemHealth. All rights reserved. The treating physician or
Intrathecal Baclofen for CNS Spasticity Last Review Date: November 13, 2015 Number: MG.MM.ME.31bC5 Medical Guideline Disclaimer Property of EmblemHealth. All rights reserved. The treating physician or
The Prospect of Stem Cell Therapy in Multiple Sclerosis. Multiple sclerosis is a multifocal inflammatory disease of the central
 The Prospect of Stem Cell Therapy in Multiple Sclerosis Multiple sclerosis is a multifocal inflammatory disease of the central nervous system that generally affects young individuals, causing paralysis
The Prospect of Stem Cell Therapy in Multiple Sclerosis Multiple sclerosis is a multifocal inflammatory disease of the central nervous system that generally affects young individuals, causing paralysis
New Treatment Options for MS Patients: Understanding risks versus benefits
 New Treatment Options for MS Patients: Understanding risks versus benefits By Michael A. Meyer, MD Department of Neurology, Sisters Hospital, Buffalo, NY Objectives: 1. to understand fundamentals of MS
New Treatment Options for MS Patients: Understanding risks versus benefits By Michael A. Meyer, MD Department of Neurology, Sisters Hospital, Buffalo, NY Objectives: 1. to understand fundamentals of MS
A Genetic Analysis of Rheumatoid Arthritis
 A Genetic Analysis of Rheumatoid Arthritis Introduction to Rheumatoid Arthritis: Classification and Diagnosis Rheumatoid arthritis is a chronic inflammatory disorder that affects mainly synovial joints.
A Genetic Analysis of Rheumatoid Arthritis Introduction to Rheumatoid Arthritis: Classification and Diagnosis Rheumatoid arthritis is a chronic inflammatory disorder that affects mainly synovial joints.
Figure 14.2 Overview of Innate and Adaptive Immunity
 I M M U N I T Y Innate (inborn) Immunity does not distinguish one pathogen from another Figure 14.2 Overview of Innate and Adaptive Immunity Our first line of defense includes physical and chemical barriers
I M M U N I T Y Innate (inborn) Immunity does not distinguish one pathogen from another Figure 14.2 Overview of Innate and Adaptive Immunity Our first line of defense includes physical and chemical barriers
IF YOU ARE RECEIVING TREATMENT WITH TYSABRI FOR RELAPSING-REMITTING MS (NATALIZUMAB)
 IF YOU ARE RECEIVING (NATALIZUMAB) TREATMENT WITH TYSABRI FOR RELAPSING-REMITTING MS Read the patient information leaflet that accompanies the medicine carefully. 1 This brochure is a supplement to the
IF YOU ARE RECEIVING (NATALIZUMAB) TREATMENT WITH TYSABRI FOR RELAPSING-REMITTING MS Read the patient information leaflet that accompanies the medicine carefully. 1 This brochure is a supplement to the
Disclosures. Consultant and Speaker for Biogen Idec, TEVA Neuroscience, EMD Serrono, Mallinckrodt, Novartis, Genzyme, Accorda Therapeutics
 Mitzi Joi Williams, MD Neurologist MS Center of Atlanta, Atlanta, GA Disclosures Consultant and Speaker for Biogen Idec, TEVA Neuroscience, EMD Serrono, Mallinckrodt, Novartis, Genzyme, Accorda Therapeutics
Mitzi Joi Williams, MD Neurologist MS Center of Atlanta, Atlanta, GA Disclosures Consultant and Speaker for Biogen Idec, TEVA Neuroscience, EMD Serrono, Mallinckrodt, Novartis, Genzyme, Accorda Therapeutics
San Diego Stem Cell Treatment Center Frequently Asked Questions
 San Diego Stem Cell Treatment Center Frequently Asked Questions What is a Stem Cell? A stem cell is basically any cell that can replicate and differentiate. This means the cell can not only multiply, but
San Diego Stem Cell Treatment Center Frequently Asked Questions What is a Stem Cell? A stem cell is basically any cell that can replicate and differentiate. This means the cell can not only multiply, but
ICD-9-CM coding for patients with Spinal Cord Injury*
 ICD-9-CM coding for patients with Spinal Cord Injury* indicates intervening codes have been left out of this list. OTHER DISORDERS OF THE CENTRAL NERVOUS SYSTEM (340-349) 344 Other paralytic syndromes
ICD-9-CM coding for patients with Spinal Cord Injury* indicates intervening codes have been left out of this list. OTHER DISORDERS OF THE CENTRAL NERVOUS SYSTEM (340-349) 344 Other paralytic syndromes
Gene Therapy. The use of DNA as a drug. Edited by Gavin Brooks. BPharm, PhD, MRPharmS (PP) Pharmaceutical Press
 Gene Therapy The use of DNA as a drug Edited by Gavin Brooks BPharm, PhD, MRPharmS (PP) Pharmaceutical Press Contents Preface xiii Acknowledgements xv About the editor xvi Contributors xvii An introduction
Gene Therapy The use of DNA as a drug Edited by Gavin Brooks BPharm, PhD, MRPharmS (PP) Pharmaceutical Press Contents Preface xiii Acknowledgements xv About the editor xvi Contributors xvii An introduction
PACKAGE LEAFLET: INFORMATION FOR THE USER. VITAMINE B12 STEROP 1mg/1ml Solution for injection / oral solution. Cyanocobalamin
 PACKAGE LEAFLET: INFORMATION FOR THE USER VITAMINE B12 STEROP 1mg/1ml Solution for injection / oral solution Cyanocobalamin Read all of this leaflet carefully before you start using this medicine because
PACKAGE LEAFLET: INFORMATION FOR THE USER VITAMINE B12 STEROP 1mg/1ml Solution for injection / oral solution Cyanocobalamin Read all of this leaflet carefully before you start using this medicine because
INSULIN PRODUCTS. Jack DeRuiter
 INSULIN PRODUCTS Jack DeRuiter The number and types of insulin preparations available in the United States is constantly changing, thus students should refer to recent drug resources for a current list
INSULIN PRODUCTS Jack DeRuiter The number and types of insulin preparations available in the United States is constantly changing, thus students should refer to recent drug resources for a current list
An introduction to modern MS treatments
 BETAFERON is a Prescription Medicine. Use strictly as directed. Consult your pharmacist or other health professional in case of side effects. BETAFERON is reimbursed for some patients. See your neurologist
BETAFERON is a Prescription Medicine. Use strictly as directed. Consult your pharmacist or other health professional in case of side effects. BETAFERON is reimbursed for some patients. See your neurologist
ORTHOPAEDIC RESEARCH AND INNOVATION DAY, April 6, 2015
 ORTHOPAEDIC RESEARCH AND INNOVATION DAY, April 6, 2015 SAFETY STUDY OF INTRAVENOUSLY ADMINISTERED HUMAN CORD BLOOD STEM CELLS IN THE TREATMENT OF SYMPTOMS RELATED TO CHRONIC INFLAMATION Brian Mehling,
ORTHOPAEDIC RESEARCH AND INNOVATION DAY, April 6, 2015 SAFETY STUDY OF INTRAVENOUSLY ADMINISTERED HUMAN CORD BLOOD STEM CELLS IN THE TREATMENT OF SYMPTOMS RELATED TO CHRONIC INFLAMATION Brian Mehling,
For Educational Use Only - Not for Detailing or Distribution
 This document is intended for healthcare professionals practicing in the United States and may contain information that has not been approved by the FDA. It is supplied to you as a professional courtesy
This document is intended for healthcare professionals practicing in the United States and may contain information that has not been approved by the FDA. It is supplied to you as a professional courtesy
How To Become A Physio And Rehabilitation Medicine Specialist
 EUROPEAN BOARD OF PHYSICAL AND REHABILITATION MEDICINE LOGBOOK EUROPEAN UNION OF MEDICAL SPECIALISTS UEMS IDENTIFICATION... 2 INSTRUCTIONS FOR USE... 3 THE TRAINING COURSE... 3 TRAINING PROGRAMME... 4
EUROPEAN BOARD OF PHYSICAL AND REHABILITATION MEDICINE LOGBOOK EUROPEAN UNION OF MEDICAL SPECIALISTS UEMS IDENTIFICATION... 2 INSTRUCTIONS FOR USE... 3 THE TRAINING COURSE... 3 TRAINING PROGRAMME... 4
Promising Treatments for SCI: What s on The Horizon. SCI: A Devastating Injury. Case: Mr. MC 9/21/2015. Epidemiology: Costs:
 Promising Treatments for SCI: What s on The Horizon Shawn Song, MD SCI Fellow University of Washington/VA Puget Sound Healthcare System SCI: A Devastating Injury Epidemiology: Incidence of ~12,000 patients/year.
Promising Treatments for SCI: What s on The Horizon Shawn Song, MD SCI Fellow University of Washington/VA Puget Sound Healthcare System SCI: A Devastating Injury Epidemiology: Incidence of ~12,000 patients/year.
Report of the Veal Calf Vaccination Study with Rispoval IntraNasal (Zoetis)
 Report of the Veal Calf Vaccination Study with Rispoval IntraNasal (Zoetis) Date: February 18, 2014 H. A. Vahl, H. Bekman and J. van Riel Introduction: Substantial percentages of antibiotics applied in
Report of the Veal Calf Vaccination Study with Rispoval IntraNasal (Zoetis) Date: February 18, 2014 H. A. Vahl, H. Bekman and J. van Riel Introduction: Substantial percentages of antibiotics applied in
Multiple Sclerosis. Matt Hulvey BL A - 615
 Multiple Sclerosis Matt Hulvey BL A - 615 Multiple Sclerosis Multiple Sclerosis (MS) is an idiopathic inflammatory disease of the central nervous system (CNS) MS is characterized by demyelination (lesions)
Multiple Sclerosis Matt Hulvey BL A - 615 Multiple Sclerosis Multiple Sclerosis (MS) is an idiopathic inflammatory disease of the central nervous system (CNS) MS is characterized by demyelination (lesions)
Omega-3 fatty acids improve the diagnosis-related clinical outcome. Critical Care Medicine April 2006;34(4):972-9
 Omega-3 fatty acids improve the diagnosis-related clinical outcome 1 Critical Care Medicine April 2006;34(4):972-9 Volume 34(4), April 2006, pp 972-979 Heller, Axel R. MD, PhD; Rössler, Susann; Litz, Rainer
Omega-3 fatty acids improve the diagnosis-related clinical outcome 1 Critical Care Medicine April 2006;34(4):972-9 Volume 34(4), April 2006, pp 972-979 Heller, Axel R. MD, PhD; Rössler, Susann; Litz, Rainer
FORM 6-K. SECURITIES AND EXCHANGE COMMISSION Washington, D.C. 20549. Report of Foreign Private Issuer
 FORM 6-K SECURITIES AND EXCHANGE COMMISSION Washington, D.C. 20549 Report of Foreign Private Issuer Pursuant to Rule 13a-16 or 15d-16 under the Securities Exchange Act of 1934 For the month of September
FORM 6-K SECURITIES AND EXCHANGE COMMISSION Washington, D.C. 20549 Report of Foreign Private Issuer Pursuant to Rule 13a-16 or 15d-16 under the Securities Exchange Act of 1934 For the month of September
NIMULID MD. 1. Introduction. 2. Nimulid MD Drug delivery system
 NIMULID MD 1. Introduction Nimulid MD is a flavoured dispersible Nimesulide tablet with fast mouth dissolving characteristics thereby providing immediate relief. Nimesulide is a non-steroidal antiinflammatory
NIMULID MD 1. Introduction Nimulid MD is a flavoured dispersible Nimesulide tablet with fast mouth dissolving characteristics thereby providing immediate relief. Nimesulide is a non-steroidal antiinflammatory
ICH Topic S 1 A The Need for Carcinogenicity Studies of Pharmaceuticals. Step 5
 European Medicines Agency July 1996 CPMP/ICH/140/95 ICH Topic S 1 A The Need for Carcinogenicity Studies of Pharmaceuticals Step 5 NOTE FOR GUIDANCE ON THE NEED FOR CARCINOGENICITY STUDIES OF PHARMACEUTICALS
European Medicines Agency July 1996 CPMP/ICH/140/95 ICH Topic S 1 A The Need for Carcinogenicity Studies of Pharmaceuticals Step 5 NOTE FOR GUIDANCE ON THE NEED FOR CARCINOGENICITY STUDIES OF PHARMACEUTICALS
Chapter 3. Immunity and how vaccines work
 Chapter 3 Immunity and how vaccines work 3.1 Objectives: To understand and describe the immune system and how vaccines produce immunity To understand the differences between Passive and Active immunity
Chapter 3 Immunity and how vaccines work 3.1 Objectives: To understand and describe the immune system and how vaccines produce immunity To understand the differences between Passive and Active immunity
your complete stem cell bank
 your complete stem cell bank HYDERABAD - 88985 000 888, WARANGAL - 8297 256 777 VISAKHAPATNAM - 7799 990 774 VIJAYAWADA AND GUNTUR - 7799 990 771 NELLORE - 7799 990 772, KADAPA - 8297 256 700 RAJAHMUNDRY
your complete stem cell bank HYDERABAD - 88985 000 888, WARANGAL - 8297 256 777 VISAKHAPATNAM - 7799 990 774 VIJAYAWADA AND GUNTUR - 7799 990 771 NELLORE - 7799 990 772, KADAPA - 8297 256 700 RAJAHMUNDRY
Hypromellose Eye Drops BP 0.3% w/v PL 23097/0006
 Hypromellose Eye Drops BP 0.3% w/v PL 23097/0006 UKPAR TABLE OF CONTENTS Lay summary Page 2 Scientific discussion Page 3 Steps taken for assessment Page 9 Summary of product characteristics Page 10 Patient
Hypromellose Eye Drops BP 0.3% w/v PL 23097/0006 UKPAR TABLE OF CONTENTS Lay summary Page 2 Scientific discussion Page 3 Steps taken for assessment Page 9 Summary of product characteristics Page 10 Patient
5 Frequently Asked Questions About Adult Stem Cell Research
 5 Frequently Asked Questions About Adult Stem Cell Research Stem cells are often referred to in the sociopolitical realm with some level of controversy and beyond that, some level of confusion. Many researchers
5 Frequently Asked Questions About Adult Stem Cell Research Stem cells are often referred to in the sociopolitical realm with some level of controversy and beyond that, some level of confusion. Many researchers
Management in the pre-hospital setting
 Management in the pre-hospital setting Inflammation of the joints Two main types: Osteoarthritis - cartilage loss from wear and tear Rheumatoid arthritis - autoimmune disorder Affects all age groups,
Management in the pre-hospital setting Inflammation of the joints Two main types: Osteoarthritis - cartilage loss from wear and tear Rheumatoid arthritis - autoimmune disorder Affects all age groups,
PHENYLEPHRINE HYDROCHLORIDE INJECTION USP
 PRESCRIBING INFORMATION PHENYLEPHRINE HYDROCHLORIDE INJECTION USP 10 mg/ml Sandoz Canada Inc. Date of Preparation: September 1992 145 Jules-Léger Date of Revision : January 13, 2011 Boucherville, QC, Canada
PRESCRIBING INFORMATION PHENYLEPHRINE HYDROCHLORIDE INJECTION USP 10 mg/ml Sandoz Canada Inc. Date of Preparation: September 1992 145 Jules-Léger Date of Revision : January 13, 2011 Boucherville, QC, Canada
Programa Cooperación Farma-Biotech Neurociencias NT-KO-003
 Programa Cooperación Farma-Biotech Neurociencias NT-KO-003 A new oral treatment for Multiple Sclerosis based on a novel mechanism of action Barcelona, 15 de febrero 2011 Programa Cooperación Farma-Biotech
Programa Cooperación Farma-Biotech Neurociencias NT-KO-003 A new oral treatment for Multiple Sclerosis based on a novel mechanism of action Barcelona, 15 de febrero 2011 Programa Cooperación Farma-Biotech
ANIMALS FORM & FUNCTION BODY DEFENSES NONSPECIFIC DEFENSES PHYSICAL BARRIERS PHAGOCYTES. Animals Form & Function Activity #4 page 1
 AP BIOLOGY ANIMALS FORM & FUNCTION ACTIVITY #4 NAME DATE HOUR BODY DEFENSES NONSPECIFIC DEFENSES PHYSICAL BARRIERS PHAGOCYTES Animals Form & Function Activity #4 page 1 INFLAMMATORY RESPONSE ANTIMICROBIAL
AP BIOLOGY ANIMALS FORM & FUNCTION ACTIVITY #4 NAME DATE HOUR BODY DEFENSES NONSPECIFIC DEFENSES PHYSICAL BARRIERS PHAGOCYTES Animals Form & Function Activity #4 page 1 INFLAMMATORY RESPONSE ANTIMICROBIAL
4.1 Objectives of Clinical Trial Assessment
 L1 4.1 Objectives of Clinical Trial Assessment Presentation to APEC Preliminary Workshop on Review of Drug Development in Clinical Trials Celia Lourenco, PhD, Manager, Clinical Group I Office of Clinical
L1 4.1 Objectives of Clinical Trial Assessment Presentation to APEC Preliminary Workshop on Review of Drug Development in Clinical Trials Celia Lourenco, PhD, Manager, Clinical Group I Office of Clinical
INTERNATIONAL CONFERENCE ON HARMONISATION OF TECHNICAL REQUIREMENTS FOR REGISTRATION OF PHARMACEUTICALS FOR HUMAN USE S1A. Current Step 4 version
 INTERNATIONAL CONFERENCE ON HARMONISATION OF TECHNICAL REQUIREMENTS FOR REGISTRATION OF PHARMACEUTICALS FOR HUMAN USE ICH HARMONISED TRIPARTITE GUIDELINE GUIDELINE ON THE NEED FOR CARCINOGENICITY STUDIES
INTERNATIONAL CONFERENCE ON HARMONISATION OF TECHNICAL REQUIREMENTS FOR REGISTRATION OF PHARMACEUTICALS FOR HUMAN USE ICH HARMONISED TRIPARTITE GUIDELINE GUIDELINE ON THE NEED FOR CARCINOGENICITY STUDIES
How To Take Methotrexate By Injection
 How To Take Methotrexate By Injection 1 how long does it take for methotrexate to work for abortion 2 methotrexate 15 mg hair loss 3 methotrexate injection dosage for rheumatoid arthritis 4 order methotrexate
How To Take Methotrexate By Injection 1 how long does it take for methotrexate to work for abortion 2 methotrexate 15 mg hair loss 3 methotrexate injection dosage for rheumatoid arthritis 4 order methotrexate
Chapter 10. Summary & Future perspectives
 Summary & Future perspectives 123 Multiple sclerosis is a chronic disorder of the central nervous system, characterized by inflammation and axonal degeneration. All current therapies modulate the peripheral
Summary & Future perspectives 123 Multiple sclerosis is a chronic disorder of the central nervous system, characterized by inflammation and axonal degeneration. All current therapies modulate the peripheral
Biologic Treatments for Rheumatoid Arthritis
 Biologic Treatments Rheumatoid Arthritis (also known as cytokine inhibitors, TNF inhibitors, IL 1 inhibitor, or Biologic Response Modifiers) Description Biologics are new class of drugs that have been
Biologic Treatments Rheumatoid Arthritis (also known as cytokine inhibitors, TNF inhibitors, IL 1 inhibitor, or Biologic Response Modifiers) Description Biologics are new class of drugs that have been
Autoimmunity. Autoimmunity. Genetic Contributions to Autoimmunity. Targets of Autoimmunity
 Autoimmunity Factors predisposing an individual to autoimmune disease Mechanisms of initiation of autoimmunity Pathogenesis of particular autoimmune disease Animal models of autoimmune disease Treatment
Autoimmunity Factors predisposing an individual to autoimmune disease Mechanisms of initiation of autoimmunity Pathogenesis of particular autoimmune disease Animal models of autoimmune disease Treatment
WHEREAS updates are required to the Compensation Plan for Pharmacy Services;
 M.O. 23/2014 WHEREAS the Minister of Health is authorized pursuant to section 16 of the Regional Health Authorities Act to provide or arrange for the provision of health services in any area of Alberta
M.O. 23/2014 WHEREAS the Minister of Health is authorized pursuant to section 16 of the Regional Health Authorities Act to provide or arrange for the provision of health services in any area of Alberta
Intravenous Fluid Selection
 BENNMC03_0131186116.qxd 3/9/05 18:24 Page 20 seema Seema-3:Desktop Folder:PQ731: CHAPTER 3 Intravenous Fluid Selection LEARNING OBJECTIVES By the end of this chapter, you should be able to: Describe and
BENNMC03_0131186116.qxd 3/9/05 18:24 Page 20 seema Seema-3:Desktop Folder:PQ731: CHAPTER 3 Intravenous Fluid Selection LEARNING OBJECTIVES By the end of this chapter, you should be able to: Describe and
PHOSPHATE-SANDOZ Tablets (High dose phosphate supplement)
 1 PHOSPHATE-SANDOZ Tablets (High dose phosphate supplement) PHOSPHATE-SANDOZ PHOSPHATE-SANDOZ Tablets are a high dose phosphate supplement containing sodium phosphate monobasic. The CAS registry number
1 PHOSPHATE-SANDOZ Tablets (High dose phosphate supplement) PHOSPHATE-SANDOZ PHOSPHATE-SANDOZ Tablets are a high dose phosphate supplement containing sodium phosphate monobasic. The CAS registry number
Roche s marketing applications for review of OCREVUS (ocrelizumab) in two forms of multiple sclerosis accepted by EMA and FDA
 Media Release Basel, 28 June 2016 Roche s marketing applications for review of OCREVUS (ocrelizumab) in two forms of multiple sclerosis accepted by EMA and FDA OCREVUS is the first investigational medicine
Media Release Basel, 28 June 2016 Roche s marketing applications for review of OCREVUS (ocrelizumab) in two forms of multiple sclerosis accepted by EMA and FDA OCREVUS is the first investigational medicine
Part II. Application not searched. Application not searched due to the presence of certain subject matter
 II.7. II.7.1. Application not searched This section relates to cases where the application relates entirely or in part to subject matter which the ISA is not required to search, or where all or part of
II.7. II.7.1. Application not searched This section relates to cases where the application relates entirely or in part to subject matter which the ISA is not required to search, or where all or part of
If you were diagnosed with cancer today, what would your chances of survival be?
 Q.1 If you were diagnosed with cancer today, what would your chances of survival be? Ongoing medical research from the last two decades has seen the cancer survival rate increase by more than 40%. However
Q.1 If you were diagnosed with cancer today, what would your chances of survival be? Ongoing medical research from the last two decades has seen the cancer survival rate increase by more than 40%. However
http://images.tutorvista.com/content/control5coordination/human5brain.jpeg!! 387
 http://images.tutorvista.com/content/control5coordination/human5brain.jpeg!! 387! 388! http://my.fresnounified.org/personal/lygonza/gonzalez/neuron/neuron5synapse%20communication.png!! http://www.urbanchildinstitute.org/sites/all/files/databooks/2011/ch15fg25communication5between5neurons.jpg!!
http://images.tutorvista.com/content/control5coordination/human5brain.jpeg!! 387! 388! http://my.fresnounified.org/personal/lygonza/gonzalez/neuron/neuron5synapse%20communication.png!! http://www.urbanchildinstitute.org/sites/all/files/databooks/2011/ch15fg25communication5between5neurons.jpg!!
How To Use An Antibody
 GUIDELINES FOR THE PRODUCTION OF ANTIBODIES IN LABORATORY ANIMALS Table of Contents 1. Purpose 2. Choice of Species and Strain 3. Immunizing Antigen 4. Procedures for Polyclonal Antibody Production 5.
GUIDELINES FOR THE PRODUCTION OF ANTIBODIES IN LABORATORY ANIMALS Table of Contents 1. Purpose 2. Choice of Species and Strain 3. Immunizing Antigen 4. Procedures for Polyclonal Antibody Production 5.
Autoimmunity and immunemediated. FOCiS. Lecture outline
 1 Autoimmunity and immunemediated inflammatory diseases Abul K. Abbas, MD UCSF FOCiS 2 Lecture outline Pathogenesis of autoimmunity: why selftolerance fails Genetics of autoimmune diseases Therapeutic
1 Autoimmunity and immunemediated inflammatory diseases Abul K. Abbas, MD UCSF FOCiS 2 Lecture outline Pathogenesis of autoimmunity: why selftolerance fails Genetics of autoimmune diseases Therapeutic
Outpatient Prescription Drug Benefit
 Outpatient Prescription Drug Benefit GENERAL INFORMATION This supplemental Evidence of Coverage and Disclosure Form is provided in addition to your Member Handbook and Health Plan Benefits and Coverage
Outpatient Prescription Drug Benefit GENERAL INFORMATION This supplemental Evidence of Coverage and Disclosure Form is provided in addition to your Member Handbook and Health Plan Benefits and Coverage
The Most Common Autoimmune Disease: Rheumatoid Arthritis. Bonita S. Libman, M.D.
 The Most Common Autoimmune Disease: Rheumatoid Arthritis Bonita S. Libman, M.D. Disclosures Two googled comics The Normal Immune System Network of cells and proteins that work together Goal: protect against
The Most Common Autoimmune Disease: Rheumatoid Arthritis Bonita S. Libman, M.D. Disclosures Two googled comics The Normal Immune System Network of cells and proteins that work together Goal: protect against
Stepping toward a different treatment option LEARN WHAT ACTHAR CAN DO FOR YOU
 FOR MS RELAPSES Stepping toward a different treatment option LEARN WHAT ACTHAR CAN DO FOR YOU As a person with multiple sclerosis (MS), you know firsthand the profound impact MS relapses can have on your
FOR MS RELAPSES Stepping toward a different treatment option LEARN WHAT ACTHAR CAN DO FOR YOU As a person with multiple sclerosis (MS), you know firsthand the profound impact MS relapses can have on your
Bile Duct Diseases and Problems
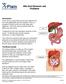 Bile Duct Diseases and Problems Introduction A bile duct is a tube that carries bile between the liver and gallbladder and the intestine. Bile is a substance made by the liver that helps with digestion.
Bile Duct Diseases and Problems Introduction A bile duct is a tube that carries bile between the liver and gallbladder and the intestine. Bile is a substance made by the liver that helps with digestion.
GUIDELINES FOR THE REGISTRATION OF BIOLOGICAL PEST CONTROL AGENTS FOOD AND AGRICULTURE ORGANIZATION OF THE UNITED NATIONS
 GUIDELINES FOR THE REGISTRATION OF BIOLOGICAL PEST CONTROL AGENTS FOOD AND AGRICULTURE ORGANIZATION OF THE UNITED NATIONS -ii- GUIDELINES ON THE REGISTRATION OF BIOLOGICAL PEST CONTROL AGENTS FOOD AND
GUIDELINES FOR THE REGISTRATION OF BIOLOGICAL PEST CONTROL AGENTS FOOD AND AGRICULTURE ORGANIZATION OF THE UNITED NATIONS -ii- GUIDELINES ON THE REGISTRATION OF BIOLOGICAL PEST CONTROL AGENTS FOOD AND
Perfalgan 10 mg/ml, solution for infusion
 PACKAGE LEAFLET: INFORMATION FOR THE USER Perfalgan 10 mg/ml, solution for infusion Paracetamol Read all of this leaflet carefully before you start using this medicine. Keep this leaflet. You may need
PACKAGE LEAFLET: INFORMATION FOR THE USER Perfalgan 10 mg/ml, solution for infusion Paracetamol Read all of this leaflet carefully before you start using this medicine. Keep this leaflet. You may need
AUBAGIO (teriflunomide) oral tablet
 AUBAGIO (teriflunomide) oral tablet Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit plan. This Pharmacy
AUBAGIO (teriflunomide) oral tablet Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit plan. This Pharmacy
AUBMC Multiple Sclerosis Center
 AUBMC Multiple Sclerosis Center 1 AUBMC Multiple Sclerosis Center The vision of the American University of Beirut Medical Center (AUBMC) is to be the leading academic medical center in Lebanon and the
AUBMC Multiple Sclerosis Center 1 AUBMC Multiple Sclerosis Center The vision of the American University of Beirut Medical Center (AUBMC) is to be the leading academic medical center in Lebanon and the
GENENTECH S OCRELIZUMAB FIRST INVESTIGATIONAL MEDICINE TO SHOW EFFICACY IN PEOPLE WITH PRIMARY PROGRESSIVE MULTIPLE SCLEROSIS IN LARGE PHASE III STUDY
 NEWS RELEASE Media Contact: Tara Iannuccillo (650) 467-6800 Investor Contacts: Stefan Foser Karl Mahler (650) 467-2016 011 41 61 687 8503 GENENTECH S OCRELIZUMAB FIRST INVESTIGATIONAL MEDICINE TO SHOW
NEWS RELEASE Media Contact: Tara Iannuccillo (650) 467-6800 Investor Contacts: Stefan Foser Karl Mahler (650) 467-2016 011 41 61 687 8503 GENENTECH S OCRELIZUMAB FIRST INVESTIGATIONAL MEDICINE TO SHOW
OHTAC Recommendation
 OHTAC Recommendation Multiple Sclerosis and Chronic Cerebrospinal Venous Insufficiency Presented to the Ontario Health Technology Advisory Committee in May 2010 May 2010 Issue Background A review on the
OHTAC Recommendation Multiple Sclerosis and Chronic Cerebrospinal Venous Insufficiency Presented to the Ontario Health Technology Advisory Committee in May 2010 May 2010 Issue Background A review on the
What is Multiple Sclerosis? Gener al information
 What is Multiple Sclerosis? Gener al information Kim, diagnosed in 1986 What is MS? Multiple sclerosis (or MS) is a chronic, often disabling disease that attacks the central nervous system (brain and spinal
What is Multiple Sclerosis? Gener al information Kim, diagnosed in 1986 What is MS? Multiple sclerosis (or MS) is a chronic, often disabling disease that attacks the central nervous system (brain and spinal
virun clean label claim emulsifiers and technologies
 virun nutrabiosciences - delivering technology clean label claim emulsifiers and technologies Esolv A patent and patents-pending naturally derived emulsifier composition to solubilize non-polar compounds
virun nutrabiosciences - delivering technology clean label claim emulsifiers and technologies Esolv A patent and patents-pending naturally derived emulsifier composition to solubilize non-polar compounds
Non-Oral Routes of Drug Administration
 Non-Oral Routes of Drug Administration Reading: Ansel 9 th edition, p. 161-170 Routes of Drug Administration Over the next series of lectures, we will talk about the biopharmaceutics of several non-oral
Non-Oral Routes of Drug Administration Reading: Ansel 9 th edition, p. 161-170 Routes of Drug Administration Over the next series of lectures, we will talk about the biopharmaceutics of several non-oral
FGF-1 as Cosmetic Supplement
 FGF-1 as Cosmetic Supplement Ing-Ming Chiu, Ph.D. Professor, Internal Medicine and Molecular and Cellular Biochemistry The Ohio State University Columbus, Ohio, U.S.A. GENTEON USA Fibroblast Growth Factor
FGF-1 as Cosmetic Supplement Ing-Ming Chiu, Ph.D. Professor, Internal Medicine and Molecular and Cellular Biochemistry The Ohio State University Columbus, Ohio, U.S.A. GENTEON USA Fibroblast Growth Factor
Disease Modifying Therapies for MS
 Disease Modifying Therapies for MS The term disease-modifying therapy means a drug that can modify or change the course of a disease. In other words a DMT should be able to reduce the number of attacks
Disease Modifying Therapies for MS The term disease-modifying therapy means a drug that can modify or change the course of a disease. In other words a DMT should be able to reduce the number of attacks
Supplemental Material CBE Life Sciences Education. Su et al.
 Supplemental Material CBE Life Sciences Education Su et al. APPENDIX Human Body's Immune System Test This test consists of 31 questions, with only 1 answer to be selected for each question. Please select
Supplemental Material CBE Life Sciences Education Su et al. APPENDIX Human Body's Immune System Test This test consists of 31 questions, with only 1 answer to be selected for each question. Please select
RULES OF DEPARTMENT OF COMMUNITY HEALTH HEALTHCARE FACILITY REGULATION
 Disclaimer: This is an unofficial copy of the rules that has been provided for the convenience of the public by the Department of Community Health. The official rules for this program are on record with
Disclaimer: This is an unofficial copy of the rules that has been provided for the convenience of the public by the Department of Community Health. The official rules for this program are on record with
ELECTROLYTE SOLUTIONS (Continued)
 ELECTROLYTE SOLUTIONS (Continued) Osmolarity Osmotic pressure is an important biologic parameter which involves diffusion of solutes or the transfer of fluids through semi permeable membranes. Per US Pharmacopeia,
ELECTROLYTE SOLUTIONS (Continued) Osmolarity Osmotic pressure is an important biologic parameter which involves diffusion of solutes or the transfer of fluids through semi permeable membranes. Per US Pharmacopeia,
MULTIPLE SCLEROSIS AUSTRALIA MULTIPLE SCLEROSIS RESEARCH AUSTRALIA
 MULTIPLE SCLEROSIS AUSTRALIA MULTIPLE SCLEROSIS RESEARCH AUSTRALIA Submission to the ACT Legislative Assembly Health, Ageing, Community and Social Services Inquiry into the exposure draft of the Drugs
MULTIPLE SCLEROSIS AUSTRALIA MULTIPLE SCLEROSIS RESEARCH AUSTRALIA Submission to the ACT Legislative Assembly Health, Ageing, Community and Social Services Inquiry into the exposure draft of the Drugs
CODEX STANDARD FOR FOLLOW-UP FORMULA CODEX STAN 156-1987. This standard applies to the composition and labelling of follow-up formula.
 CODEX STAN 156-1987 Page 1 of 9 CODEX STANDARD FOR FOLLOW-UP FORMULA CODEX STAN 156-1987 1. SCOPE This standard applies to the composition and labelling of follow-up formula. It does not apply to foods
CODEX STAN 156-1987 Page 1 of 9 CODEX STANDARD FOR FOLLOW-UP FORMULA CODEX STAN 156-1987 1. SCOPE This standard applies to the composition and labelling of follow-up formula. It does not apply to foods
MEDICATION GUIDE. ACTEMRA (AC-TEM-RA) (tocilizumab) Solution for Intravenous Infusion
 MEDICATION GUIDE ACTEMRA (AC-TEM-RA) (tocilizumab) Solution for Intravenous Infusion ACTEMRA (AC-TEM-RA) (tocilizumab) Injection, Solution for Subcutaneous Administration Read this Medication Guide before
MEDICATION GUIDE ACTEMRA (AC-TEM-RA) (tocilizumab) Solution for Intravenous Infusion ACTEMRA (AC-TEM-RA) (tocilizumab) Injection, Solution for Subcutaneous Administration Read this Medication Guide before
Media Release. Basel, 8 October 2015
 Media Release Basel, 8 October 2015 Roche s ocrelizumab first investigational medicine to show positive pivotal study results in both relapsing and primary progressive forms of multiple sclerosis Ocrelizumab
Media Release Basel, 8 October 2015 Roche s ocrelizumab first investigational medicine to show positive pivotal study results in both relapsing and primary progressive forms of multiple sclerosis Ocrelizumab
dependent independent claims
 Mechanics of claim drafting Karin Pramberger Belgrade 16/17 Nov 2006 1 dependent independent claims 1. all essential features in independent claims with at least 1 novelty conferring feature 2. fall back
Mechanics of claim drafting Karin Pramberger Belgrade 16/17 Nov 2006 1 dependent independent claims 1. all essential features in independent claims with at least 1 novelty conferring feature 2. fall back
THE SPINAL CORD AND THE INFLUENCE OF ITS DAMAGE ON THE HUMAN BODY
 THE SPINAL CORD AND THE INFLUENCE OF ITS DAMAGE ON THE HUMAN BODY THE SPINAL CORD. A part of the Central Nervous System The nervous system is a vast network of cells, which carry information in the form
THE SPINAL CORD AND THE INFLUENCE OF ITS DAMAGE ON THE HUMAN BODY THE SPINAL CORD. A part of the Central Nervous System The nervous system is a vast network of cells, which carry information in the form
Standards of Practice for Pharmacists and Pharmacy Technicians
 Standards of Practice for Pharmacists and Pharmacy Technicians Introduction These standards are made under the authority of Section 133 of the Health Professions Act. They are one component of the law
Standards of Practice for Pharmacists and Pharmacy Technicians Introduction These standards are made under the authority of Section 133 of the Health Professions Act. They are one component of the law
How To Choose A Biologic Drug
 North Carolina Rheumatology Association Position Statements I. Biologic Agents A. Appropriate delivery, handling, storage and administration of biologic agents B. Indications for biologic agents II. III.
North Carolina Rheumatology Association Position Statements I. Biologic Agents A. Appropriate delivery, handling, storage and administration of biologic agents B. Indications for biologic agents II. III.
Drug Pricing System in Japan
 Reference Chuikyo - 1 June 6, 2012 Drug Pricing System in Japan April 2012 (Underlined phrases in red in this text: New features and modifications to the current pricing system adopted by the 2012 system
Reference Chuikyo - 1 June 6, 2012 Drug Pricing System in Japan April 2012 (Underlined phrases in red in this text: New features and modifications to the current pricing system adopted by the 2012 system
Laser Treatment Policy
 Laser Treatment Policy Pursuant to federal law 21 CFR 812.2(c)7 and 812.3(b), physician(s) at this pain center may advise and use unapproved laser s on patients under one or more of the following conditions:
Laser Treatment Policy Pursuant to federal law 21 CFR 812.2(c)7 and 812.3(b), physician(s) at this pain center may advise and use unapproved laser s on patients under one or more of the following conditions:
Summary HTA. Interferons and Natalizumab for Multiple Sclerosis Clar C, Velasco-Garrido M, Gericke C. HTA-Report Summary
 Summary HTA HTA-Report Summary Interferons and Natalizumab for Multiple Sclerosis Clar C, Velasco-Garrido M, Gericke C Health policy background Multiple sclerosis (MS) is a chronic inflammatory disease
Summary HTA HTA-Report Summary Interferons and Natalizumab for Multiple Sclerosis Clar C, Velasco-Garrido M, Gericke C Health policy background Multiple sclerosis (MS) is a chronic inflammatory disease
Medicare Drug Coverage Under Part A, Part B, and Part D
 Medicare Drug Coverage Under Part A, Part B, and Part D Medicare Part A and Part B generally do not cover outpatient prescription drugs, most of which are now covered under Part D. This document and the
Medicare Drug Coverage Under Part A, Part B, and Part D Medicare Part A and Part B generally do not cover outpatient prescription drugs, most of which are now covered under Part D. This document and the
PATIENT INFORMATION: PATIENT CONTACT PHONE NUMBERS: PHYSICIAN INFORMATION: HEALTH INSURANCE INFORMATION:
 PATIENT INFORMATION: TODAY S DATE: HOW DID YOU HEAR ABOUT US?: LAST NAME: FIRST NAME: STREET CITY: STATE: ZIP: EMAIL MARTIAL STATUS: SINGLE MARRIED DIVORCED WIDOWED SEPARATED BIRTHDATE: AGE: SEX: MALE
PATIENT INFORMATION: TODAY S DATE: HOW DID YOU HEAR ABOUT US?: LAST NAME: FIRST NAME: STREET CITY: STATE: ZIP: EMAIL MARTIAL STATUS: SINGLE MARRIED DIVORCED WIDOWED SEPARATED BIRTHDATE: AGE: SEX: MALE
Absorption of Drugs. Transport of a drug from the GI tract
 Absorption of Drugs Absorption is the transfer of a drug from its site of administration to the bloodstream. The rate and efficiency of absorption depend on the route of administration. For IV delivery,
Absorption of Drugs Absorption is the transfer of a drug from its site of administration to the bloodstream. The rate and efficiency of absorption depend on the route of administration. For IV delivery,
Severe rheumatoid arthritis (a disease that causes inflammation of the joints),where MabThera is given intravenously together with methotrexate.
 EMA/614203/2010 EMEA/H/C/000165 EPAR summary for the public rituximab This is a summary of the European public assessment report (EPAR) for. It explains how the Committee for Medicinal Products for Human
EMA/614203/2010 EMEA/H/C/000165 EPAR summary for the public rituximab This is a summary of the European public assessment report (EPAR) for. It explains how the Committee for Medicinal Products for Human
Compensation Plan for Pharmacy Services
 Compensation Plan for Pharmacy Services Attachment A Section 1 - Definitions ABC Pharmacy Agreement means an agreement between ABC and a Community Pharmacy as described in Schedule 2.1 of the Alberta Blue
Compensation Plan for Pharmacy Services Attachment A Section 1 - Definitions ABC Pharmacy Agreement means an agreement between ABC and a Community Pharmacy as described in Schedule 2.1 of the Alberta Blue
Princeton University Prescription Drug Plan Summary Plan Description
 Princeton University Prescription Drug Plan Summary Plan Description Princeton University Prescription Drug Plan Summary Plan Description January 2015 Contents Introduction... 1 How the Plan Works... 2
Princeton University Prescription Drug Plan Summary Plan Description Princeton University Prescription Drug Plan Summary Plan Description January 2015 Contents Introduction... 1 How the Plan Works... 2
SUMMARY OF PRODUCT CHARACTERISTICS
 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE VETERINARY MEDICINAL PRODUCT Prednicare Tablets 5mg 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each tablet contains: Active Substance(s) Prednisolone
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE VETERINARY MEDICINAL PRODUCT Prednicare Tablets 5mg 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each tablet contains: Active Substance(s) Prednisolone
Winter 2013. Changing landscapes, pipeline products and plan sponsor impact
 Winter 2013 Changing landscapes, pipeline products and plan sponsor impact Changing landscapes, pipeline products and plan sponsor impact The pharmaceutical landscape is changing as is the profile of blockbuster
Winter 2013 Changing landscapes, pipeline products and plan sponsor impact Changing landscapes, pipeline products and plan sponsor impact The pharmaceutical landscape is changing as is the profile of blockbuster
Nursing 113. Pharmacology Principles
 Nursing 113 Pharmacology Principles 1. The study of how drugs enter the body, reach the site of action, and are removed from the body is called a. pharmacotherapeutics b. pharmacology c. pharmacodynamics
Nursing 113 Pharmacology Principles 1. The study of how drugs enter the body, reach the site of action, and are removed from the body is called a. pharmacotherapeutics b. pharmacology c. pharmacodynamics
