DEPARTMENT OF OPHTHALMOLOGY UNIVERSITY OF IOWA HOSPITALS AND CLINICS OCULAR PATHOLOGY LABORATORY
|
|
|
- Felicia Sharleen Pope
- 8 years ago
- Views:
Transcription
1 DEPARTMENT OF OPHTHALMOLOGY UNIVERSITY OF IOWA HOSPITALS AND CLINICS OCULAR PATHOLOGY LABORATORY SPECIMEN COLLECTION MANUAL FOR IN-HOUSE PHYSICIANS, SURGEONS AND STAFF The purpose of this collection manual is to provide specimen collection information to the Ophthalmology Department Nursing Station, Ambulatory Surgery Center, General OR, Iowa River Landing Clinic, Frozen Section Room and any other areas providing patient specimens to the F. C. Blodi Eye Pathology Laboratory, room 233, Medical Research Center building (MRC). This manual is found on the Ophthalmology Department s website at: Click on Patient Care, click on Labs and Screening, click on Ophthalmic Pathology. Notifications of annual review and updates are sent by to all ophthalmology staff, the General O.R., Frozen Section Room, the Ambulatory Surgical Center and Iowa River Landing Clinic. Specimen collection begins with the removal of a fluid or a piece of tissue from a patient during a surgical procedure. If the procedure requires stat/rush attention, (i.e: Acanthamoeba scraping, temporal artery biopsy, etc.) the F.C. Blodi Eye Pathology Laboratory should be contacted in advance of beginning the procedure as the Eye Pathology Laboratory must prepare equipment and materials for these particular procedures prior to receiving the patient's specimen. Dr. Syed (pager 2438) needs to be called for all fine needle aspirate procedures. GENERAL INFORMATION ON COLLECTION AND HANDLING OF SPECIMENS 1. ROUTINE PICK-UP LABORATORY HOURS a. The Ocular Pathology Laboratory Hours are Monday-Friday, 7:30 am-5:00 PM. b. Ocular Pathology has one routine pick-up time between 2:30 and 3 PM Monday-Friday. 2. EXCEPTIONS TO ROUTINE PICK-UP TIME: a. Temporal artery biopsies: i. See pages if a temporal artery biopsy is to be processed for stat/same day results. b. Fine Needle Aspirations: i. See pages c. Corneal Epithelial Scrapings: i. See pages Revised: April 2014 Page 1 of 26
2 HANDLING, LABELING AND TRANSPORTATION OF SPECIMENS AND OCULAR PATHOLOGY CONSULTATION REQUEST FORMS 1. ATTENDING PHYSICIANS RESPONSIBILITIES: a. Special procedure specimen collection i. Prior to surgery, refer to the Special Procedure Specimen Collection section (Prot: 1.1.1, page 9-17) regarding handling instructions for the following procedures and specimen types: a) Acanthamoeba scraping b) Electron microscopy c) Fine needle aspirations d) Frozen sections e) Immunohistochemistry f) Metabolic diseases (i.e.: gout, cystinosis, storage disease) g) Temporal artery biopsy h) Vitreous b. Routine Specimen Collection by Specimen Type i. Prior to surgery, refer to the Routine Specimen Collection by Specimen Type (Prot: 1.1.1, page 18) for handling instructions concerning specimens not mentioned in the Special Procedure Specimen Collection. c. University of Iowa Hospitals and Clinics By-laws i. All tissues removed in the Ophthalmology minor surgery room, Iowa River Landing Ambulatory Surgery, Operating rooms pertaining to the eye must be sent to the F.C. Blodi Eye Pathology Laboratory by virtue of the by-laws of the University of Iowa Hospitals and Clinics. d. Exceptions to Tissue Submittal i. All ocular tissues removed surgically must be sent to the F.C. Blodi Eye Pathology Laboratory unless prior written approval has been obtained by the Director of the Ophthalmology Department and the Director of the F.C. Blodi Eye Pathology Laboratory. ii. The following list of routine types of tissue need not be sent to the F.C. Blodi Eye Pathology Laboratory. a) Crystalline Lens (solid or aspirate) Cataract extractions b) Vitreous Routine c) Iris Routine iridectomy d) Trabecular Meshwork Routine Trabeculectomy Revised: April 2014 Page 2 of 26
3 iii. It is at the surgeon s discretion to determine whether pathologic examination of routine tissues will aid in patient management/diagnoses. The surgeon may elect to submit routine tissue to the F.C. Blodi Eye Pathology Laboratory as needed. iv. When these tissues are not submitted to the F.C. Blodi Eye Pathology Laboratory, they are discarded by the Main Operating Room and the Ambulatory Surgical Center per their specimen disposal protocol. e. Questions or Concerns i. Any questions concerning the handling of Ocular specimens should be directed to the Ocular Pathologist or the Ocular Pathology Supervisor ( ), prior to performing the surgery. 2. OPERATING ROOM, AMBULATORY SURGERY AND OPHTHALMOLOGY NURSING STAFF RESPONSIBILITIES: a. Special procedure specimen collection i. Refer to the each of the following under the Special Procedure Specimen Collection (Prot: 1.1.1, pages 9-17) for specific handling instructions: ii. iii. iv. Acanthamoeba scraping Electron microscopy Fine needle aspirations v. Frozen sections vi. Immunohistochemistry vii. Metabolic diseases (i.e.: gout, cystinosis, storage disease) viii. Temporal artery biopsy ix. Vitreous b. Routine Specimen Collection by Specimen Type i. Specimens that do not fall under the Special Procedure Specimen Collection portion of this manual should be considered "routine" and should be handled in the following manner (Refer to: Routine Specimen Collection by Specimen Type (Prot: 1.1.1, pages 18-25) for specific handling instructions): a) Materials needed prior to beginning the surgery: i) A leak proof specimen container with the correct type and amount of fixative (if a fixative is required), please refer to each specimen type under the Routine Specimen Collection by Specimen Type portion of this manual the for specific fixation instructions. ii) If formalin is used as the fixative, a formalin warning label must be placed on the specimen container. iii) An Ocular Pathology Consultation Request form. Revised: April 2014 Page 3 of 26
4 ii. Immediately after the "routine" tissue specimen has been removed from the patient do the following: a) Place the tissue into the container with the proper fixative. b) Make sure the tissue is completely covered by the fixative. i) DO NOT wrap the tissue in gauze or other types of toweling as they will absorb the fixative and parts of the tissue may not be covered by the fixative causing the tissue to decompose. c) The amount of fixative in the container will vary with the size of the specimen; however, the fixative should be a minimum of ten times the volume of the specimen being fixed. i) Example 1: If the specimen is a trabeculectomy, corneal biopsy, corneal button, small skin biopsy, conjunctival biopsy, etc. there should be at least ml of fixative in the container. ii) Example 2: If the specimen is an eyeball (enucleation) there should be at least 100 ml of fixative in the container. iii) Example 3: If the specimen is an exenteration (eye, eyelids and surrounding tissue) the container used should be large enough so that the fixative will completely cover the entire specimen. d) Place the lid on the container properly and tightly to avoid a fixative spill. i) If the fixatives are either formalin, glutaraldehyde or 100% alcohol and a spill occurs, refer to the respective Safety Data Sheet (SDS). *** NOTE *** The Ambulatory Surgery Center and the General O.R. provide specimen containers from their areas. The Ocular Pathology Laboratory provides the Ophthalmology Nurses Station with pre-filled specimen containers. c. Identification and labeling of specimen containers i. Identify and label all ophthalmology specimen containers with the following information: a) Two unique patient identifiers (name and medical record number or date of birth) b) Physician's name c) Type and location of the specimen: (i.e.: corneal button, left eye) d) Formalin warning label ii. Make sure that a well-constructed container with a secure lid is used. iii. Specimen containers must be labeled with the above information at the time of the procedure and before the container leaves the procedure room. Revised: April 2014 Page 4 of 26
5 d. Filling out the ocular pathology consultation request form i. The Ocular Pathology Consult Request form must be prepared in the room where the procedure is performed (College of American Pathologist regulation). ii. The following information must be on the ocular pathology consultation request form. The laboratory will be unable to process the specimen until this information is received (College of American Pathologist regulation). a) Material submitted (wet tissue, slides or blocks) b) Type and location of the tissue submitted: (i.e: Corneal button, left eye) c) Clinical history, data and operative findings d) Clinical diagnosis Note: Whenever possible use the UIHC patient label ID sticker for the following information. If the sticker is not available, the following information must be handwritten on the Ocular Pathology Consultation Request form: e) 2 unique patient identifiers (name and medical record number or date of birth) f) Patient's hospital location (Intensive care, Ambulatory surgery, etc.) g) Date of surgery h) Name and physician code number of the primary staff physician responsible for the patient i) Name of other staff physicians, fellows or residents should be placed in the designated space on the request form. j) Any special instructions or communications to the laboratory (ie: rush or stat cases, instructions to understand margins, etc.) iii. Place the container into a biohazard bag for transportation to avoid leakage during transportation. a) The requisition should be placed in the side pocket of the biohazard bag in case of a leaking container. e. Delivering specimens and ocular pathology consultation request forms i. If the specimens are from the operating room, ambulatory surgery, or Iowa River Landing, deliver all specimens with their respective Ocular Pathology Consultation Request forms to the Frozen Section Room. ii. If the specimen is removed in the Ophthalmology minor room the specimen should be placed in the ocular pathology box found on the desk at Ophthalmology's nursing station. Revised: April 2014 Page 5 of 26
6 3. OCULAR PATHOLOGY LABORATORY RESPONSIBILITIES: a. Routine Specimen Pick-up i. Routinely specimens are picked up from the Frozen Section Room and Ophthalmology's nursing station by the Ocular Pathology personnel between 2:30-3:00 PM, Monday-Friday. a) Ocular pathology personnel will check to assure that each specimen has an ocular pathology consultation request form. b) When picking up specimens from the Nurses Station, Ocular Pathology personnel will initial the log book for each specimen taken. ii. Ocular pathology personnel will bring all specimens back to the laboratory in a carrier labeled as Biohazard. Specimen containers are placed into a slotted box to keep the containers upright. b. Accessioning of Specimens i. Specimens will be accessioned (given an ocular pathology identification number). a) During accessioning the ocular pathology personnel will check the ocular pathology consultation request form and specimen container for pertinent patient information. ii. If any pertinent patient information is missing from either the specimen container or the ocular pathology consultation request form the attending physician will be contacted (see Rejection and Exemption of Specimens below for further instructions). c. Grossing of Specimens i. Specimens will be grossed by the pathologist or by the technician under the supervision of the pathologist. d. Processing, Embedding, Cutting and Staining of Specimens i. Specimens will be processed, embedded, cut and stained then given to the pathologist for diagnosis. Revised: April 2014 Page 6 of 26
7 REJECTION AND EXEMPTION OF SPECIMENS 1. INCOMPLETE PATIENT OR SPECIMEN INFORMATION a. Specimens received without an Ocular Pathology Consultation Request form, incomplete information on the form or the specimen container, cannot be processed until the proper information is received from the primary staff physician. i. A diagnosis cannot be rendered until the missing information is received from the primary staff physician (College of American Pathologists (CAP) regulations). ii. All requests for consultation must be done by the Ocular Pathology Consultation Request Form. Verbal or phone orders are not accepted. b. Ocular pathology lab personnel inform the staff physician and head nurse of the respective area of problems. (QA: 1.2.1) Monthly Problem QA notices will be sent out to involved staff. i. The following information is documented in the Specimen Rejection Notebook for each problems case: a) Date the problem occurred b) Staff doctor s name c) Nursing unit involved d) Patient name e) Patient medical record number f) Patient accession number g) Description of the problem h) How specimen may be affected i) Explanation of how future problems may be avoided j) Problem resolution k) Lab staff who note problems will handwrite a description of the problem and initial on original requisition form c. A copy of each letter is kept in the Specimen Rejection/Problem notebook. d. Specimen rejections are also a part of Ocular Pathology s Quality Management Program. Revised: April 2014 Page 7 of 26
8 2. SENDING INFORMATION TO THE OCULAR PATHOLOGY LABORATORY a. Due to possible miscommunications and laboratory regulations patient information cannot be received over the phone, it must be received in written form from the staff physician attending to the patient in question. i. The staff physician must provide the ocular pathology laboratory with the pertinent patient information in the one of the following ways, the same day it was requested: a) Using the computer, (Harris, Peggy) b) Fax (319) c) Physically coming to the laboratory and filling out a requisition form (233 MRC) ii. Any or fax received is stapled to the original patient consultation request form. Note: The case cannot be processed or signed out until this information is received (CAP regulation). iii. If the information is not received from the staff physician after two attempts, the Clerk IV and Director of the laboratory will be made aware of the situation. a) The Clerk IV or Director of the laboratory will contact the attending physician to inform them of the problems again and will let them know a diagnosis cannot be rendered until the information is received for our records. 3. GIVING PATIENT INFORMATION TO PHYSICIANS a. Secretaries, laboratory supervisor, technicians and students cannot give patient information over the phone to a physician; only the ocular pathologist can give this information to the attending physician. i. If a conversation occurs between the attending physician and the pathologist it will be documented in the final patient report for permanent record. b. If you wish to know the status of a case, please phone the ocular pathology laboratory ( ). 4. COPIES OF FINAL REPORTS a. A copy of the final report is available in the F.C. Blodi Eye Pathology laboratory on the Cerner computer system and a hard copy is printed out, bound and retained indefinitely. b. When pathology reports are signed final electronically in the Cerner system they are automatically sent to the Epic system as Anatomic Pathology Results. A Resulted Test notice is sent to the Epic in-box of the submitting physician. Ocular pathology reports remain on the Epic system as part of the electronic patient record. Revised: April 2014 Page 8 of 26
9 SPECIAL PROCEDURE SPECIMEN COLLECTION 1. CORNEAL EPITHELIAL SCRAPING FOR ACANTHAMOEBA a. Special instructions: i. For STAT results, scrapings should be done between the hours of 7:30 am and 3:00 PM, if they are done before or after these hours technicians may not be available to complete the procedure. a) Scrapings need to be delivered to the Ocular Pathology Laboratory by 3:00. ii. Routine scrapings will be picked up at the nurses station by lab personnel between 2:30 and 3:00 PM. iii. Make sure the following materials are in the room prior to performing the scraping: b. Procedure a) Spatula for scraping the cornea b) Sterile container (centrifuge tube, specimen container) c) Wash bottle containing Saccomanno fixative d) Ocular Pathology Consultation Request form with the proper basic patient information, clinical data, and specimen location (ie: corneal scraping right eye) written on the form. e) UIHC patient ID sticker label for the specimen container i. Have an open labeled sterile container (centrifuge tube, specimen container) available prior to scraping the patient. ii. Using a spatula scrape the patient as usual. iii. Immediately after scraping the patient, using wash bottle containing Saccomanno fixative, wash cells and tissue off spatula and into the sterile container. iv. Make sure not to use more than 0.5 ml of Saccomanno to wash the cells off the spatula. v. Take tip of spatula and swirl in the Saccomanno to assure that all of the cells have been removed from the spatula. vi. Place the lid on the container tightly vii. Make sure the Ocular Pathology Consultation Request form has been filled out completely and placed in the outside pouch of the bio-hazard bag. c. SDS i. An updated copy of the SDS for Saccomanno fluid will be provided by the Ocular Pathology Lab (5-7095). Revised: April 2014 Page 9 of 26
10 d. Saccomanno Fluid i. Saccomanno fluid will be provided by the Ocular Pathology Lab (5-7095) when: ii. The supply in the Cornea Clinic has been depleted. iii. The Saccomanno Fluid has expired. e. Lab Instructions i. Clinic copies of lab instructions for Corneal Epithelial Scraping will be provided by the Ocular Pathology Lab (5-7095) Prot: ELECTRON MICROSCOPY a. Fixative: Glutaraldehyde b. Special instructions: i. Prior to surgery contact the ocular pathologist to determine if Electron Microscopy is necessary for the patients care or if another test would be more informative. ii. If Electron Microscopy is to be performed on the tissue the following materials should be obtained prior to surgery: a) A container of Glutaraldehyde which can be obtained by contacting the Electron Microscopy laboratory at or i) 81 EMRB ii) The bottle of Glutaraldehyde must be labeled with the UIHC patient label sticker and percentage of Glutaraldehyde in the bottle. b) A container of 10% Neutral Buffered Formalin i) The specimen container must be labeled with the UIHC patient label ID sticker label c) An Ocular Pathology Consultation Request form. i. After removal of the tissue from the patient place half of the tissue into the Glutaraldehyde and the other half into the 10% Neutral Buffered Formalin. ii. Fill out the Ocular Pathology Consultation Request form. a) When filling out the request form make sure to indicate that one specimen is in 10% neutral buffered formalin and that the other is in Glutaraldehyde: (i.e.: A. corneal biopsy, left eye in 10% neutral buffered formalin, B. corneal biopsy, left eye in Glutaraldehyde, for EM studies). iii. Send both specimens and requisition to the Ocular Pathology Laboratory as the ocular pathologist will have to request the test for Electron Microscopy after the routine slides have been reviewed. a) The Electron Microscopy specimen will be refrigerated by the ocular pathology laboratory until it is sent to the Electron Microscopy facility for study. Revised: April 2014 Page 10 of 26
11 iv. If the EM specimen is not in the frozen section room by 2:30 PM it will not be picked up by the Ocular Pathology Laboratory that day, and it is the responsibility of the staff physician to assure the specimen is refrigerated until our next specimen pick-up or the laboratory is contacted and special arrangements are made to pick up the specimen. 3. FINE NEEDLE ASPIRATIONS a. Arrangements: Special arrangements must be made at least one day prior to this procedure being performed with the pathologist to assure that the pathologist and a technician are available to process the specimen. If this is an emergency please try to give the laboratory at least one hours notice prior to the removal of the specimen. i. At this time arrangements should be made with the pathologist. ii. Fine needle aspirations must be performed no later than 2:30 p.m., Monday-Friday, for same day diagnosis, unless special arrangements have been made with the pathologist. b. Fixation: This specimen requires immediate fixation. The following materials should be in the surgical suite prior to beginning this procedure. i. An FNA kit from the nurse s station. ii. Small gauge needle (22). iii. Syringe. iv. A completed Ocular Pathology Consultation Request form. a) The Ocular Pathology Consultation Request form should be filled out completely prior to the removal of the specimen to decrease the delay in getting the specimen to the laboratory. iv. Label the 50 ml conical tube with the UIHC patient label ID sticker. a) With 2 unique patient identifiers (name and MRN or DOB). v. The following three steps must be followed exactly as written in order to meet JCAHO, OSHA and CAP regulations. a) The physician will remove the specimen using the small gauge needle and syringe and retain the specimen in the syringe. b) After the removal of the specimen the physician will carefully draw an equal amount of Saccomanno fixative into the syringe. c) The physician will then dispense the fixed specimen into the empty 50 ml conical tube. d) Gently mix the specimen with the Saccomanno fluid for complete fixation. e) Place the conical tube into the BioHazard bag. Revised: April 2014 Page 11 of 26
12 vi. The specimen will be delivered to the laboratory for immediate processing. vii. Once in the laboratory, the specimen will be accessioned, described and processed within 1.5 hours. viii. The finished slides will be given to the pathologist for an immediate diagnosis. ix. The pathologist will inform the staff physician as soon as a diagnosis has been rendered. Note: If the Pathologist is unavailable the slides will be prepared by a technician and read by the pathologist upon her return. d. FNA Tap Kits i. FNA Tap Kits are located in the Ophthalmology Nurse s Station. ii. A kit should be obtained prior to the scheduled procedure. iii. Each kit contains: a) Saccomanno fluid (fixative) b) 50 ml conical tube (specimen container) c) BioHazard specimen bag d) Ocular Pathology Consultation Request Form e) Clinic copy of instructions for FNA tap procedure (Prot: 1.1.4) iv. The FNA tap kits in the ophthalmology nurses station are provided by the Ocular Pathology Laboratory (5-7095) when: a) The supply of kits has run out b) The Saccomanno fluid has expired v. An updated copy of the SDS for the Saccomanno fluid will be provided by the Ocular Pathology Laboratory (5-7095). The SDS is located in the Vitreous Tap Kit container. 4. FROZEN SECTIONS ON OCULAR TISSUE a. Arrangements: For ALL ophthalmic cases, the ocular pathologist should be paged (2438) directly from the operating room to notify her of the frozen. She will then notify Surgical Pathology of whether or not she will personally be available to interpret the case. If not, Surgical Pathology will provide backup coverage. In the event that the Surgical Pathologist does not feel comfortable with interpreting that particular case and the Ocular Pathologist is not available, they will contact Dr. Patricia Kirby (Pathology) page #3414. SURGEONS ARE REQUIRED TO MAKE PRIOR ARRANGEMENTS WITH DR. SYED TO MAKE SURE SHE IS AVAILABLE FOR CASES THAT ARE OPHTHALMIC PATHOLOGY SPECIFIC. b. Fixation: Fresh tissue placed on a saline soaked gauze pad and immediately sent to the frozen section room, 5804 JPP, to produce permanently stained and mounted slide/s. Revised: April 2014 Page 12 of 26
13 i. Fill out the Ocular Pathology Consultation Request form completely. ii. After the frozen section has been diagnosed, take the residual tissue, wrap it in lens paper and place in a cassette. iii. Place the cassette into a container with enough 10% neutral buffered formalin to completely cover the tissue filled cassette. a) The container will be labeled with 2 patient identifiers. iv. Place the lid on the container firmly. v. Give the specimen container and the Ocular Pathology Consultation Request form to the frozen section room personnel. vi. The frozen section slides (labeled with 2 patient identifiers) will be submitted to the F.C. Blodi Eye Pathology Laboratory. vii. The F.C. Blodi Eye Pathology laboratory will pick up the residual tissue and/or slides on their routine (3:00 PM) run, as mentioned under the General Information on Collection and Handling of Specimens. viii. The slides and residual tissue will be accessioned in the F.C. Blodi Eye Pathology Laboratory. ix. All residual tissue is processed for paraffin section comparison. 5. IMMUNOHISTOCHEMISTRY a. Arrangements: If an attending physician feels there may be a need for immunohistochemistry on a case they should contact the ocular pathologist prior to the surgery being performed to determine if the case needs the immunohistochemical techniques to diagnose the case. If it is necessary to perform this test the pathologist can determine whether fresh frozen tissue should be obtained during the surgery or if fixed, processed, paraffin embedded tissue can be used for the technique. b. Fixation: Determined by the ocular pathologist, either fresh frozen tissue given to the immunopathology laboratory and/or tissue placed into 10% neutral buffered formalin and sent to the frozen section room to be picked up by the F.C. Blodi Eye Pathology laboratory for processing. i. If fresh frozen section material is going to be supplied contact the Immunology laboratory ( ) prior to surgery for special instruction on how to handle the specimen properly. a) If there is more tissue to come on the case indicate this on the Ocular Pathology Consultation Request form and inform the Frozen Section Room Attendant. ii. If the tissue is placed into 10% neutral buffered formalin, send it and the Ocular Pathology Consultation form to the Frozen Section room for the routine 2:30 PM pick-up by the laboratory. Revised: April 2014 Page 13 of 26
14 a) Fill out the Ocular Pathology Consultation Request form as mentioned under General Information on Collection and Handling of Specimens. b) The specimen will be picked up by the F.C. Blodi Eye Pathology laboratory on the usual 2:30-3:00 PM run, unless other arrangements have been made with the Ocular Pathology laboratory. 6. METABOLIC DISEASES (ie: gout, cystinosis, storage disease) a. Arrangements: If the specimen has a suspected metabolic disease, contact the laboratory prior to removal of the specimen for special fixation instructions. DO NOT remove the specimen until the special instructions have been received or the disease process may be destroyed because the proper fixation was not performed in the proper amount of time. b. Each specimen is different and must be handled on a case by case basis. Contact the F.C. Blodi Eye Pathology laboratory for instructions (319) TEMPORAL ARTERY BIOPSIES a. If a doctor wants a same day diagnosis, THEY must call the lab themselves. Nursing is not responsible for telling the lab that there is a stat lab. The lab can only do same day results if they have the specimen by 9:30 AM again the rationale for the notice of the late specimen to be picked up early the next business day. Transportation of the specimen is to be arranged by the MD when the lab is called. c. The lab will continue to pick up specimens on their usual 2:30 PM rounds unless otherwise directed by Dr. Syed. i. If a TA biopsy is received after 4:30 PM it will be processed the following day (ie: in Tuesday after 4:30 PM will be given to the ocular pathologist by 1:00 PM Thursday). Note: Be aware that times may vary somewhat depending on the workload, availability of the Lab Director, technical coverage, and the number of stat cases on that particular day. Revised: April 2014 Page 14 of 26
15 TEMPORAL ARTERY BIOPSY DIAGNOSIS NEXT BUSINESS DAY (ROUTINE) DIAGNOSIS NEEDED SAME DAY IF PROCEDURE COMPLETE BEFORE 2:30 PM MON-FRI LEAVE SPECIMEN AND PAPERWORK AT NURSING STATION SPECIMEN PICK-UP AREA BY 2:30 PM TO INSURE SAME DAY PICK UP IF PROCEDURE COMPLETE BETWEEN 2:30 PM & 4:30 PM MON-FRI NOTE 1: SPECIMENS REACHING LAB AFTER 4:30 PM WILL BE LOGGED IN & PROCESSED NEXT DAY -- DIAGNOSIS WILL BE AVAILABLE 2 BUSINESS DAYS AFTER PROCEDURE LABORATORY STAFF WILL PICK UP SPECIMEN AT ROUTINE PICK-UP TIME 2:30 PM CALL LAB TO NOTIFY OF BIOPSY AND ARRANGE FOR SPECIMEN TRANSPORT TO LAB MUST SPEAK TO LAB PERSONNEL IF PROCEDURE COMPLETE AFTER 4:30 PM MON-FRI OR ON WEEKEND/HOLIDAY NOTE: DIAGNOSIS WILL BE AVAILABLE 2 BUSINESS DAYS AFTER PROCEDURE LEAVE SPECIMEN AND PAPERWORK AT NURSING STATION SPECIMEN PICK-UP AREA LABORATORY STAFF WILL PICK UP SPECIMEN NEXT BUSINESS DAY AT ROUTINE PICK-UP TIME 2:30 PM NOTE: SAME DAY DIAGNOSIS SHOULD BE RESERVED FOR WHEN THE NEED FOR DIAGNOSIS IS EXTREMELY URGENT AND IS AT THE DISCRETION OF THE ATTENDING PHYSICIAN MD MUST CALL PATHOLOGIST TO ARRANGE SPECIMEN MUST BE IN LABORATORY BY 9:30 AM BLODI EYE PATHOLOGY LABORATORY PHONE NUMBER: PATHOLOGIST PAGERS: LAB ADMIN ASST. -- PEG HARRIS LAB SUPERVISOR -- CHRISTY BALLARD SYED 2438 KIRBY 3414 Revised: April 2014 Page 15 of 26
16 d. Fixation: 10% Neutral buffered formalin e. Special Instructions: i. Prior to beginning the surgery for a TA biopsy the following should be in the surgical room: a) a specimen container with the correct amount of 10% neutral buffered formalin in it b) a formalin warning sticker c) UIHC patient label ID sticker d) An Ocular Pathology Consultation Request form. ii. Immediately after the TA biopsy specimen has been removed from the patient, place the tissue into the container of 10% neutral buffered formalin, make sure the tissue is completely covered with the 10% neutral buffered formalin. iii. The amount of formalin in the container will vary with the size of the specimen, however the fixative should be a minimum of ten times the volume of the specimen being fixed. a) The amount for TA biopsies should be at least ml of 10% neutral buffered formalin in the container. iv. Place the lid on the container properly and tightly to avoid a formalin spill. v. Follow the procedure under General Information on Collection and Handling of Specimens for proper labeling of the specimen and how to properly fill out the Ocular Pathology Consultation Request forms. 8. VITREOUS TAP (Intraocular biopsy, fluid and tissue fragments from the eye) a. Arrangements: A vitreous tap is a concentrated specimen collected during the same procedure as a vitrectomy to diagnose lymphoma or malignancy. i. The Ocular Pathology Lab should be notified at least one hour before the procedure, preferably the day before for stat results. b. Fixation: The specimen must be fixed immediately. c. Vitreous Tap Kits i. Vitreous Tap Kits are located in the Ophthalmology Nurse s Station. ii. A kit should be obtained prior to the scheduled procedure. iii. Each kit contains: a) Saccomanno fluid (fixative) b) 50 ml conical tube (specimen container) c) BioHazard specimen bag Revised: April 2014 Page 16 of 26
17 d) Ocular Pathology Consultation Request Form e) Clinic copy of instructions for vitreous tap procedure (Prot: 1.1.5) iv. The vitreous tap kits in the ophthalmology nurses station are provided by the Ocular Pathology Laboratory (5-7095) when: a) The supply of kits has run out b) The Saccomanno fluid has expired v. An updated copy of the SDS for the Saccomanno fluid will be provided by the Ocular Pathology Laboratory (5-7095). The SDS is located in the Vitreous Tap Kit container. d. Special Instructions: i. Prior to the beginning of surgery for a vitreous tap, the following should be in the surgical room: a) A syringe. i) Vitreous fluid is tapped out of a vitrectomy. There is no needle. b) A Vitreous Tap Kit from the nurse s station c) A completed Ocular Pathology Consultation Request Form. ii. Label the 50 ml conical tube with the UIHC patient label ID sticker. iii. Add an equal amount of Saccomanno fixative to the syringe and then dispense into the 50 ml conical tube. iv. Gently mix the specimen and the Saccomanno fluid for complete fixation. v. For stat processing, immediately deliver the specimen to the Ocular Pathology Lab, room #233, MRC. a) Vitreous taps must be in the Ocular Pathology Laboratory by 3:00 p.m. for same day results. vi. For routine processing, the Ocular Pathology Lab will pick up the specimen on the daily run at 2:30 p.m. vii. VITRECTOMY CASSETTE BAGS: a) If the biopsy is being done to rule-out lymphoma, the vitrectomy cassette bag (dilute aspirate) should be labeled with a UIHC patient label ID sticker and sent immediately to the Flow Cytometry Laboratory. Deliver the cassette bag to Specimen Control and they will deliver the cassette bag to the Flow Cytometry Laboratory (6 TH FLOOR CARVER BETWEEN ELEVATORS E &F, TAKE WEST HALLWAY NEXT TO GLASS ATRIUM TO PATHOLOGY LABORATORIES, 1 ST RIGHT TO SPECIMEN CONTROL ). i) A Flow Cytometry request form (WHITE WITH GREEN PRINT) should be completed and sent with the specimen. Revised: April 2014 Page 17 of 26
18 b) If the biopsy is done for other reasons, label with the UIHC patient label ID sticker and transport the vitrectomy cassette bag to the Blodi Eye Pathology Lab as soon as possible with the YELLOW request form. 1. AUTOPSY EYE ROUTINE SPECIMEN COLLECTION BY SPECIMEN TYPE a. Arrangements: Contact the F.C. Blodi Eye Pathology laboratory in advance of the removal of the eyes. i. Immerse the specimen in a sufficient quantity of fixative so that the eye is covered completely by the fixative. Make no holes or incisions in the globe as this will complicate the diagnosis and may destroy pertinent diagnostic information. ii. Follow the instructions listed under General Information on Collection and 2. CILIARY BODY (iridocyclectomy specimen) a. Arrangements: No special arrangements necessary i. Because this specimen is usually taken for tumor, a study of the resection margins is important. Please make a sketch on the Ocular Pathology Consultation Request form of the location of the tumor in the eye so that the medial and temporal resection margins can be distinguished in the specimen. ii. Follow the instructions listed under General Information on Collection and 3. CONJUNCTIVA a. Arrangements: No special arrangements are necessary. i. Conjunctival tissue should be handled according to the following protocol to prevent curling of the tissue: ii. Spread the conjunctiva onto a flat, absorbent surface such as the paper wrapping for gloves, the file-card envelope for sutures, or filter paper. iii. Allow the conjunctival tissue to become adherent to the surface for approximately seconds. Revised: April 2014 Page 18 of 26
19 iv. When the tissue is adherent to its support, float the supporting surface and tissue onto formalin with the tissue surface facing up. The absorbent material will soak up fixative and sink. The specimen will be received in the pathology laboratory with its proper orientation preserved. d. It is important that an accurate clinical diagnosis accompany the specimen. For example, conjunctival biopsies to rule out sarcoid are handled differently by the grossing pathologist from biopsies for suspected neoplasms. i. Follow the instructions listed under General Information on Collection and ii. When a conjunctival biopsy is done to rule out OCP (ocular cicatricial pemphigoid), half of the specimen should be sent fresh to the Immuno Lab and the other half in formalin to our lab. Both specimens should be accessioned under our accession number. The Immuno Lab should be notified that they should proceed with doing a pemphigoid panel on the tissue. They should put their portion of the report in the Additional Procedures section of our report. iii. Please indicate on the Ocular Pathology Consultation Request form if tissue has been sent to the Immuno Pathology Laboratory. 4. CORNEAL BIOPSY (for infectious disease ) a. Arrangements: Please contact the F.C. Blodi Eye Pathology laboratory ( ) before surgery when the anticipated diagnosis will require: (1) electron microscopy, (2) immunopathology, (3) the detection of crystalline substances, especially urate crystals., unless specimen was for electron microscopy, immunopathology, or the detection of crystalline substances, especially urate crystals in these cases contact the Ocular Pathology Laboratory for fixation instructions. i. It is generally not possible to divide small specimens less than 1.5 mm into two pieces, one for histology and one for microbiology. Dividing such small specimens may make it difficult for both histology and microbiology laboratories to perform adequate examinations on the specimen. ii. Follow the instructions listed under General Information on Collection and 5. CORNEAL BUTTONS (tissue from transplant procedures) a. Arrangements: Please contact the F.C. Blodi Eye Pathology laboratory ( ) before surgery when the anticipated diagnosis will require: (1) electron microscopy, (2) immunopathology, (3) the detection of crystalline substances, especially urate crystals., unless specimen was for electron microscopy, immunopathology, or the detection of crystalline substances, especially urate crystals in these cases contact the Ocular Pathology Laboratory for fixation instructions. Revised: April 2014 Page 19 of 26
20 i. Corneal buttons should not be allowed to desiccate in the operating room before being placed into fixative. Because it may be important to wait until donor tissue is secured into place, put the host material into tissue culture medium until the surgeon considers the circumstances of the operation "safe enough" to permit host tissue fixation. ii. Follow the instructions listed under General Information on Collection and, unless the specimen is for electron microscopy, immunopathology or detection of crystalline substances (urate crystals). 6. CORNEAL BUTTON (routine) a. Arrangements: Please contact the F.C. Blodi Eye Pathology laboratory ( ) before surgery when the anticipated diagnosis will require: (1) electron microscopy, (2) immunopathology, (3) the detection of crystalline substances, especially urate crystals., unless specimen was for electron microscopy, immunopathology, or the detection of crystalline substances, especially urate crystals in these cases contact the Ocular Pathology Laboratory for fixation instructions. i. Routine corneal buttons should be placed into the 10% neutral buffered formalin immediately after removal. ii. Follow the instructions listed under General Information on Collection and. 7. DESCEMET S MEMBRANE a. Arrangements: No special arrangements are necessary. i. Follow the instructions listed under General Information on Collection and. 8. ENUCLEATION a. Arrangements: Contact the F.C. Blodi Eye Pathology laboratory ( ) in advance of the removal of the eye if special studies (such as electron microscopy or immunology) are to be performed on the tissue. ii. Immerse the specimen in sufficient quantity of fixative so that the eye is covered. Make no holes or incisions into the globe. Revised: April 2014 Page 20 of 26
21 iii. It is important that a complete clinical history accompany the specimen (ie: a list of any previous operations performed on the eye may alert the pathologist to the presence of an intraocular lens or a surgical wound. iv. If an eye is removed for tumor, provide a copy of the fundus drawings so that the eye may be opened in an appropriate plane. v. Follow the instructions listed under General Information on Collection and 9. EVISCERATION a. Arrangements: No special arrangements are necessary i. Follow the instructions listed under General Information on Collection and 10. EXENTERATION a. Arrangements: No special arrangements are necessary 11. EYELID i. Please be certain to have all aspects of this large specimen completely covered by fixative. ii. Follow the instructions listed under General Information on Collection and a. Arrangements: No special arrangements are necessary i. If the resection margins are important, please make a sketch on the Ocular Pathology Consultation Request form as to the location of the tumor or area of interest in the eyelid so that the resection margins can be distinguished in the specimen. ii. Follow the instructions listed under General Information on Collection and 12. FOREIGN BODY a. Arrangements: The specimen is sent to microbiology for study then after microbiology is finished with the specimen it is sent to the F.C. Blodi Eye Pathology laboratory for gross and saved for archival studies. b. Fixation: None, sometimes received in microbiology broth. Revised: April 2014 Page 21 of 26
22 i. These specimens are stored indefinitely as archival material and such information may be important for future medical-legal considerations. 13. INTRAOCULAR LENS a. Arrangements: No special arrangements are necessary. b. Fixation: None 14. IRIS i. Please provide the name of the manufacturer of the lens together with the model number or style of the lens. These specimens are stored for archival purposes and such information may be important for future medical-legal considerations. a. Arrangements: If the iris is removed for a suspected tumor, please see instructions listed above under ciliary body (iridocyclectomy) specimens. i. There are no special instructions if the iris is removed at the time of glaucoma filtering procedures. ii. Follow the instructions listed under General Information on Collection and 15. LACRIMAL SYSTEM (lacrimal gland or sac) a. Arrangements: If material is needed for immunopathology please contact the F.C. Blodi Eye Pathology laboratory for instructions prior to surgery, unless specimen is for immunopathology then contact the F.C. Blodi Eye Pathology laboratory for instructions on how to handle the tissue prior to surgery 16. MUSCLE i. Please indicate on the Ocular Pathology Consultation Request form if there is tissue to be sent to the immunopathology laboratory. ii. If the tissue is for the F.C. Blodi Eye Pathology laboratory, follow the instructions listed under General Information on Collection and Handling of Specimens (beginning on page 1) a. Arrangements: No special arrangements are necessary Revised: April 2014 Page 22 of 26
23 i. Follow the instructions listed under General Information on Collection and 17. OPTIC NERVE a. Arrangements: No special arrangements are necessary i. Follow the instructions listed under General Information on Collection and 18. OPTIC NERVE SHEATH a. Arrangements: No special arrangements are necessary i. Follow the instructions listed under General Information on Collection and 19. ORBITAL BIOPSY a. Arrangements: If material is needed for immunopathology please contact the F.C. Blodi Eye Pathology laboratory for instructions prior to surgery, unless specimen is for immunopathology then contact the F.C. Blodi Eye Pathology laboratory for instructions on how to handle the tissue prior to surgery i. Please indicate on the Ocular Pathology Consultation Request form if there is tissue to be sent to the immunopathology laboratory. ii. If the tissue is for the F.C. Blodi Eye Pathology laboratory, follow the instructions listed under General Information on Collection and Handling of Specimens (beginning on page 1) 20. ORBITAL TUMOR/LYMPHOID LESION a. Arrangements: Material will be needed for Flow Cytometry. Please contact the F.C. Blodi Eye Pathology Laboratory for instructions prior to surgery, unless specimen is for immunopathology then contact the F.C. Blodi Eye Pathology laboratory for instructions on how to handle the tissue prior to surgery i. At least 3 cubic mm of tissue will be needed for flow cytometry Revised: April 2014 Page 23 of 26
24 ii. Please indicate on the Ocular Pathology Consultation Request Form when tissue has been sent to the Flow Cytometry Lab. (See pg 17 #8-d-vii for directions to this lab.) iii. Please mark flow on the immunopath sheet iv. If the tissue is for the F.C. Blodi Eye Pathology laboratory, follow the instructions listed under General Information on Collection and Handling of Specimens (beginning on page 1) 21. ORBITAL TUMOR/PEDIATRIC TUMOR a. Arrangements: Material will be needed for cytogenetics studies. Pleas contact the F.C. Blodi Eye Pathology laboratory for instructions prior to surgery. for the F.C. Blodi Eye Pathology laboratory i. Fresh tissue to be sent to the Cytogenetics Laboratory. i. Please indicate on the Ocular Pathology Consultation Request form when fresh tissue has been sent to the cytogenetics laboratory. ii. If the tissue is for the F.C. Blodi Eye Pathology laboratory, follow the instructions listed under General Information on Collection and Handling of Specimens (beginning on page 1) 22. PTERYGIUM a. Special Arrangements: No special arrangements are necessary 23. SKIN i. Follow the procedure under General Information on Collection and Handling of Specimens (beginning on page 1). a. Arrangements: No special arrangements are necessary i. Follow the instructions listed under General Information on Collection and 24. TRABECULAR MESHWORK a. Arrangements: No special arrangements are necessary Revised: April 2014 Page 24 of 26
25 i. Follow the instructions listed under General Information on Collection and 25. VITRECTOMY a. Arrangements: No arrangements are necessary, unless the specimen is collected in a syringe. i. See #9, Vitreous Tap, page 16. b. If the specimen is collected in the usual vitreous container, take it and the Ocular Pathology Consultation Request form to the frozen section room. i. If the tissue is for the F.C. Blodi Eye Pathology laboratory, follow the instructions listed under General Information on Collection and Handling of Specimens (beginning on page 1). ****NOTE**** Never refrigerate or place vitrectomy specimens on ice as cells degenerate faster. PHYSICIAN NOTIFICATION 1. If the diagnosis of the patient's tissue is critical to the patients treatment and requires immediate attention the ocular pathologist will contact the attending physician as soon as the diagnosis is complete. a. The above information is noted in the Comment section of the final report along with the following information: i. Date ii. Time iii. First and last name of person notified 2. An electronic copy is sent to the attending physician s Epic in-box as a Resulted Test. 3. A hard copy is printed out, bound, and retained indefinitely in the F.C. Blodi Eye Pathology Laboratory. REFERENCES: Sheehan, Dezna C. and Barbara B. Hrapchak: Theory and Practice of Histotechnology, 2nd Ed.; C.V. Mosby Co., St. Louis, MO; Preece, Ann: A Manual for Histologic Technicians, 3rd Ed.; Little, Brown & Co., Inc.; Boston, MA; Revised: April 2014 Page 25 of 26
26 Aldrich: Catalog and Handbook of Fine Chemicals; Aldrich Chemical Co., Inc.; Milwaukee, WI; Safety Data Sheets. Revised: April 2014 Page 26 of 26
BSM Connection elearning Course
 BSM Connection elearning Course Scope of the Eye Care Practice 2008, BSM Consulting All Rights Reserved. Table of Contents OVERVIEW...1 THREE O S IN EYE CARE...1 ROUTINE VS. MEDICAL EXAMS...2 CONTACT LENSES/GLASSES...2
BSM Connection elearning Course Scope of the Eye Care Practice 2008, BSM Consulting All Rights Reserved. Table of Contents OVERVIEW...1 THREE O S IN EYE CARE...1 ROUTINE VS. MEDICAL EXAMS...2 CONTACT LENSES/GLASSES...2
Descemet s Stripping Endothelial Keratoplasty (DSEK)
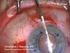 Descemet s Stripping Endothelial Keratoplasty (DSEK) Your doctor has decided that you will benefit from a corneal transplant operation. This handout will explain your options to you. It explains the differences
Descemet s Stripping Endothelial Keratoplasty (DSEK) Your doctor has decided that you will benefit from a corneal transplant operation. This handout will explain your options to you. It explains the differences
MUSCLE BIOPSY Information For Clinicians Information For Pathology Residents Muscle Biopsy Video Tutorial
 Modified 2-24-11 by Dr. McKeever and Dr. Camelo-Piragua MUSCLE BIOPSY Information For Clinicians Information For Pathology Residents Muscle Biopsy Video Tutorial Procedure (for Main OR, Mott OR, Treatment
Modified 2-24-11 by Dr. McKeever and Dr. Camelo-Piragua MUSCLE BIOPSY Information For Clinicians Information For Pathology Residents Muscle Biopsy Video Tutorial Procedure (for Main OR, Mott OR, Treatment
Infectious Waste Management Plan
 Infectious Waste Management Plan Infectious Waste Management Plan USC Health & Safety Programs Unit 777-5269 POLICY: A. In keeping with the University of South Carolina's policy of providing protection
Infectious Waste Management Plan Infectious Waste Management Plan USC Health & Safety Programs Unit 777-5269 POLICY: A. In keeping with the University of South Carolina's policy of providing protection
Please note: Contact Coppe Laboratories at 262-574-0701 if archival plasma samples need to be tested.
 Collecting a Coppe Laboratories Sample The Coppe Laboratories Sample Kit contains: Test Request Form (TRF) Heparin tube Absorbent sheet Biohazard bag Foil pouch Label for blood tube Box Seal Instructions
Collecting a Coppe Laboratories Sample The Coppe Laboratories Sample Kit contains: Test Request Form (TRF) Heparin tube Absorbent sheet Biohazard bag Foil pouch Label for blood tube Box Seal Instructions
UCLA Henry Samueli School of Engineering and Applied Science STANDARD OPERATING PROCEDURE MEDICAL WASTE (BIOHAZARD WASTE) MANAGEMENT
 UCLA Henry Samueli School of Engineering and Applied Science STANDARD OPERATING PROCEDURE MEDICAL WASTE (BIOHAZARD WASTE) MANAGEMENT PURPOSE The purpose of this document is to specify the procedures used
UCLA Henry Samueli School of Engineering and Applied Science STANDARD OPERATING PROCEDURE MEDICAL WASTE (BIOHAZARD WASTE) MANAGEMENT PURPOSE The purpose of this document is to specify the procedures used
The University of Texas at San Antonio Office of Environmental Health, Safety and Risk Management. Part A. Biological Waste Management Safety Plan
 The University of Texas at San Antonio Office of Environmental Health, Safety and Risk Management Part A Biological Waste Management Safety Plan i. SIGNATURE PAGE This Biological Waste Management Safety
The University of Texas at San Antonio Office of Environmental Health, Safety and Risk Management Part A Biological Waste Management Safety Plan i. SIGNATURE PAGE This Biological Waste Management Safety
Getting Ready for ICD-10. Dianna Hoskins, OCS Cincinnati Eye Institute
 Dianna Hoskins, OCS Cincinnati Eye Institute Chart Documentation: Will your documentation stand up to ICD-10? Do you always mark which eye, severity or status of the disease (chronic or acute), site, etiology
Dianna Hoskins, OCS Cincinnati Eye Institute Chart Documentation: Will your documentation stand up to ICD-10? Do you always mark which eye, severity or status of the disease (chronic or acute), site, etiology
WHICH SAMPLES SHOULD BE SUBMITTED WHEN LYMPHOID NEOPLASIA IS SUSPECTED?
 WHICH SAMPLES SHOULD BE SUBMITTED WHEN LYMPHOID NEOPLASIA IS SUSPECTED? Which test should be submitted? The answer to this depends on the clinical signs, and the diagnostic question you are asking. If
WHICH SAMPLES SHOULD BE SUBMITTED WHEN LYMPHOID NEOPLASIA IS SUSPECTED? Which test should be submitted? The answer to this depends on the clinical signs, and the diagnostic question you are asking. If
Annual Biomedical Waste Code Training
 Annual Biomedical Waste Code Training Provided by: Barbara D. Will, MPH Biomedical Waste Program Supervisor To protect, promote and improve the health of all people in Florida through integrated state,
Annual Biomedical Waste Code Training Provided by: Barbara D. Will, MPH Biomedical Waste Program Supervisor To protect, promote and improve the health of all people in Florida through integrated state,
Introduction BIOMEDICAL WASTE
 Page 1 of 4 Title: Chemical Waste Disposal Guidelines-Research Program or Department: Research Document Type: PROCEDURE Effective Date: January 01,2015 Author Steven Hayes Next Review Date: January 01,2016
Page 1 of 4 Title: Chemical Waste Disposal Guidelines-Research Program or Department: Research Document Type: PROCEDURE Effective Date: January 01,2015 Author Steven Hayes Next Review Date: January 01,2016
Carnegie Mellon University s Policy and Procedures for Recombinant and Synthetic Nucleic Acid Materials Spills
 Carnegie Mellon University s Policy and Procedures for Recombinant and Synthetic Nucleic Acid Materials Spills Background In accordance with Section IV-B-2-b-(6) of the NIH Guidelines for Research Involving
Carnegie Mellon University s Policy and Procedures for Recombinant and Synthetic Nucleic Acid Materials Spills Background In accordance with Section IV-B-2-b-(6) of the NIH Guidelines for Research Involving
NEW YORK UNIVERSITY SCHOOL OF MEDICINE DEPARTMENT OF OPHTHALMOLOGY SUPERVISION OF RESIDENTS POLICY
 NEW YORK UNIVERSITY SCHOOL OF MEDICINE DEPARTMENT OF OPHTHALMOLOGY SUPERVISION OF RESIDENTS POLICY Applies to: All Ophthalmology Residents, including those in ACGME- and non-acgme-accredited specialty
NEW YORK UNIVERSITY SCHOOL OF MEDICINE DEPARTMENT OF OPHTHALMOLOGY SUPERVISION OF RESIDENTS POLICY Applies to: All Ophthalmology Residents, including those in ACGME- and non-acgme-accredited specialty
INSTITUTIONAL POLICY AND PROCEDURE (IPP)
 HOSPITAL NAME INSTITUTIONAL POLICY AND PROCEDURE (IPP) Department: Manual: Section: TITLE/DESCRIPTION POLICY NUMBER HAZARDOUS WASTE: HANDLING AND DISPOSAL EFFECTIVE DATE REVIEW DUE REPLACES NUMBER NO.
HOSPITAL NAME INSTITUTIONAL POLICY AND PROCEDURE (IPP) Department: Manual: Section: TITLE/DESCRIPTION POLICY NUMBER HAZARDOUS WASTE: HANDLING AND DISPOSAL EFFECTIVE DATE REVIEW DUE REPLACES NUMBER NO.
Appendix H IBC Managing Biohazardous Waste SOP
 Biohazardous waste is managed under the State of Rhode Island s Regulated Medical Waste Regulations (Regulation DEM-OWM-MW-1-2009, amended July, 2010). http://www.dem.ri.gov/pubs/regs/regs/waste/medwaste10.pdf
Biohazardous waste is managed under the State of Rhode Island s Regulated Medical Waste Regulations (Regulation DEM-OWM-MW-1-2009, amended July, 2010). http://www.dem.ri.gov/pubs/regs/regs/waste/medwaste10.pdf
Biohazardous Waste and Sharps Disposal
 Biohazardous Waste and Sharps Disposal Federal OSHA Occupational Exposure to Bloodborne Pathogens Standard 29 CFR 1910.1030 State California Code of Regulations (CCR), Medical Waste Management Act, Chapter
Biohazardous Waste and Sharps Disposal Federal OSHA Occupational Exposure to Bloodborne Pathogens Standard 29 CFR 1910.1030 State California Code of Regulations (CCR), Medical Waste Management Act, Chapter
ENVIRONMENTAL HEALTH, SAFETY & RISK MANAGEMENT. Hazardous Materials &Waste Management Plan at
 ا السلامة و معالجة المخاطر لصحة البيي ية ENVIRONMENTAL HEALTH, SAFETY & RISK MANAGEMENT Hazardous Materials &Waste Management Plan at AUB-MC 1 Outline 1. Objectives 2. Definitions of Hazardous Waste 3.
ا السلامة و معالجة المخاطر لصحة البيي ية ENVIRONMENTAL HEALTH, SAFETY & RISK MANAGEMENT Hazardous Materials &Waste Management Plan at AUB-MC 1 Outline 1. Objectives 2. Definitions of Hazardous Waste 3.
Revision 5. Calvin College Medical Waste Management Plan. Date: Health and Safety
 Calvin College Medical Waste Management Plan Prepared by: Date: Environmental 6/10/1 Health and Safety Approved By: Date: Revision 1.0 Policy The following medical waste management plan has been established
Calvin College Medical Waste Management Plan Prepared by: Date: Environmental 6/10/1 Health and Safety Approved By: Date: Revision 1.0 Policy The following medical waste management plan has been established
Some of the ophthalmic surgeries performed at the DMV Center.
 Some of the ophthalmic surgeries performed at the DMV Center. This document presents some types of the surgeries performed by the ophthalmology service at the DMV veterinary center as well as the equipment
Some of the ophthalmic surgeries performed at the DMV Center. This document presents some types of the surgeries performed by the ophthalmology service at the DMV veterinary center as well as the equipment
Division of Laboratory Medicine Department of Pathology and Laboratory Medicine Hospital of the University of Pennsylvania
 Division of Laboratory Medicine Department of Pathology and Laboratory Medicine Hospital of the University of Pennsylvania General Laboratory Specimen Collection Guidelines The Laboratory will perform
Division of Laboratory Medicine Department of Pathology and Laboratory Medicine Hospital of the University of Pennsylvania General Laboratory Specimen Collection Guidelines The Laboratory will perform
Clinical Histology Procedure histo09.03. Embedding Paraffin Processed Tissue
 Clinical Histology Procedure histo09.03 Final Approval: May 2010 Effective: May 2010 Next Review Date: April 2015 List all stakeholder(s) and dates of approval: Stakeholder Name(s): Shelly Siegel HT Date:
Clinical Histology Procedure histo09.03 Final Approval: May 2010 Effective: May 2010 Next Review Date: April 2015 List all stakeholder(s) and dates of approval: Stakeholder Name(s): Shelly Siegel HT Date:
Biosafety Level 2 (BSL-2) Safety Guidelines
 BLS-4 Biosafety Level 2 (BSL-2) Safety Guidelines BSL-3 BSL-2 BSL-1 BSL-2 builds upon BSL-1. If you work in a lab that is designated a BSL-2, the microbes used pose moderate hazards to laboratory staff
BLS-4 Biosafety Level 2 (BSL-2) Safety Guidelines BSL-3 BSL-2 BSL-1 BSL-2 builds upon BSL-1. If you work in a lab that is designated a BSL-2, the microbes used pose moderate hazards to laboratory staff
Caring for a Tenckhoff Catheter
 Caring for a Tenckhoff Catheter UHN A Patient s Guide What is a Pleural Effusion? There is a small space between the outside of your lung and the chest wall (ribs). This space is called the pleural space.
Caring for a Tenckhoff Catheter UHN A Patient s Guide What is a Pleural Effusion? There is a small space between the outside of your lung and the chest wall (ribs). This space is called the pleural space.
2.3. The management in each HCF shall be responsible for ensuring good waste management practices in their premises.
 1. PURPOSE Health-care activities lead to production of medical waste that may lead to adverse health effects. Most of this waste is not more dangerous than regular household waste. However, some types
1. PURPOSE Health-care activities lead to production of medical waste that may lead to adverse health effects. Most of this waste is not more dangerous than regular household waste. However, some types
Biohazardous, Medical & Biological Waste Guidance Chart
 CSULA Environmental Health and Safety Biohazardous, Medical & Biological Waste Guidance Chart The chart below provides information on how to handle most, if not all, of the items that frequently are collectively
CSULA Environmental Health and Safety Biohazardous, Medical & Biological Waste Guidance Chart The chart below provides information on how to handle most, if not all, of the items that frequently are collectively
Leica CEREBRO Sample Tracking. Patient Safety. Enhanced Productivity. Complete Flexibility.
 P P P Leica CEREBRO Sample Tracking Patient Safety. Enhanced Productivity. Complete Flexibility. Patient Safety Protect their health, and your reputation. Don't let identification errors lead to patient
P P P Leica CEREBRO Sample Tracking Patient Safety. Enhanced Productivity. Complete Flexibility. Patient Safety Protect their health, and your reputation. Don't let identification errors lead to patient
Archived. Gloves should be changed frequently during the analysis.
 Introduction Gloves and laboratory coats Small tools Specific clean-up and housekeeping procedures are used to help protect evidence samples from conditions and agents that might serve to destroy, deteriorate,
Introduction Gloves and laboratory coats Small tools Specific clean-up and housekeeping procedures are used to help protect evidence samples from conditions and agents that might serve to destroy, deteriorate,
Content Sheet 5-1: Overview of Sample Management
 Content Sheet 5-1: Overview of Management Role in quality management system management is a part of process control, one of the essentials of a quality management system. The quality of the work a laboratory
Content Sheet 5-1: Overview of Management Role in quality management system management is a part of process control, one of the essentials of a quality management system. The quality of the work a laboratory
UltraClean Forensic DNA Isolation Kit (Single Prep Format)
 UltraClean Forensic DNA Isolation Kit (Single Prep Format) Catalog No. Quantity 14000-10 10 preps 14000-S 1 prep Instruction Manual Please recycle Version: 10302012 1 Table of Contents Introduction...
UltraClean Forensic DNA Isolation Kit (Single Prep Format) Catalog No. Quantity 14000-10 10 preps 14000-S 1 prep Instruction Manual Please recycle Version: 10302012 1 Table of Contents Introduction...
Radiology Services Policies and Procedures
 Radiology Services Policies and Procedures November 2011 Table of Contents - Alphabetical Please Note! Clicking on a title in the Table of Contents will take you to that document. To find a particular
Radiology Services Policies and Procedures November 2011 Table of Contents - Alphabetical Please Note! Clicking on a title in the Table of Contents will take you to that document. To find a particular
DIRECTED UMBILICAL CORD BLOOD AND TISSUE COLLECTION IN THEATRE (CELLCARE)
 WOMEN AND NEWBORN HEALTH SERVICE King Edward Memorial Hospital CLINICAL GUIDELINES PERIOPERATIVE GUIDELINES DIRECTED CORD CELL COLLECTION IN THEATRE DIRECTED UMBILICAL CORD BLOOD AND TISSUE COLLECTION
WOMEN AND NEWBORN HEALTH SERVICE King Edward Memorial Hospital CLINICAL GUIDELINES PERIOPERATIVE GUIDELINES DIRECTED CORD CELL COLLECTION IN THEATRE DIRECTED UMBILICAL CORD BLOOD AND TISSUE COLLECTION
MODERN CLINICAL OPTOMETRY BILLING & CODING THE MEDICAL EYE EXAMINATION. Definitions of Eye Examinations. Federal Government Definition
 MODERN CLINICAL OPTOMETRY BILLING & CODING THE MEDICAL EYE EXAMINATION Craig Thomas, O.D. 3900 West Wheatland Road Dallas, Texas 75237 972-780-7199 thpckc@yahoo.com Definitions of Eye Examinations Optometry
MODERN CLINICAL OPTOMETRY BILLING & CODING THE MEDICAL EYE EXAMINATION Craig Thomas, O.D. 3900 West Wheatland Road Dallas, Texas 75237 972-780-7199 thpckc@yahoo.com Definitions of Eye Examinations Optometry
PATIENT GUIDE. Care and Maintenance Drainage Frequency: Max. Drainage Volume: Dressing Option: Clinician s Signature: Every drainage Weekly
 PATIENT GUIDE Care and Maintenance Drainage Frequency: Max. Drainage Volume: Dressing Option: Every drainage Weekly Clinician s Signature: ACCESS SYSTEMS Pleural Space Insertion Site Cuff Exit Site Catheter
PATIENT GUIDE Care and Maintenance Drainage Frequency: Max. Drainage Volume: Dressing Option: Every drainage Weekly Clinician s Signature: ACCESS SYSTEMS Pleural Space Insertion Site Cuff Exit Site Catheter
A Guide to Managing Your Biological Waste at the University at Albany
 A Guide to Managing Your Biological Waste at the University at Albany Section 1 - What you need to know: Definition: "Regulated Medical Waste (RMW) shall mean any of the following waste which is generated
A Guide to Managing Your Biological Waste at the University at Albany Section 1 - What you need to know: Definition: "Regulated Medical Waste (RMW) shall mean any of the following waste which is generated
Oklahoma Facts CPT. Definitions. Mohs Micrographic Surgery. What Does That Mean? Billing and Coding for Mohs Surgery
 Billing and Coding for Mohs Surgery Cindy L. Wilson Dermatology Associates of Tulsa Oklahoma Facts Per square mile, Oklahoma has more tornadoes than any other place in the world. The highest wind speed
Billing and Coding for Mohs Surgery Cindy L. Wilson Dermatology Associates of Tulsa Oklahoma Facts Per square mile, Oklahoma has more tornadoes than any other place in the world. The highest wind speed
Connecticut Biomedical Waste (BMW) Requirements (22a-209-15) and Common Industry Practices
 Connecticut Biomedical Waste (BMW) Requirements (22a-209-15) and Common Industry Practices Mark Latham CT DEP, Waste Engineering and Enforcement Division Common Synonyms for BMW Regulated Medical Waste
Connecticut Biomedical Waste (BMW) Requirements (22a-209-15) and Common Industry Practices Mark Latham CT DEP, Waste Engineering and Enforcement Division Common Synonyms for BMW Regulated Medical Waste
Appendix J IBC Biohazard Spill Management Plan
 OVERVIEW Prevention is the most important part of any spill management plan. Be sure to read and understand standard operating procedures (SOP s) and protocols for safe manipulation of biohazards before
OVERVIEW Prevention is the most important part of any spill management plan. Be sure to read and understand standard operating procedures (SOP s) and protocols for safe manipulation of biohazards before
BREAST CANCER PATHOLOGY
 BREAST CANCER PATHOLOGY FACT SHEET Version 4, Aug 2013 This fact sheet was produced by Breast Cancer Network Australia with input from The Royal College of Pathologists of Australasia I m a nurse and know
BREAST CANCER PATHOLOGY FACT SHEET Version 4, Aug 2013 This fact sheet was produced by Breast Cancer Network Australia with input from The Royal College of Pathologists of Australasia I m a nurse and know
Managing Regulated Medical Waste in New Mexico
 Managing Regulated Medical Waste in New Mexico Prepared by: Dr. Joe King Camino Real Environmental Research Center Sunland Park, New Mexico History of Medical Waste Regulations 1988 Legislation in response
Managing Regulated Medical Waste in New Mexico Prepared by: Dr. Joe King Camino Real Environmental Research Center Sunland Park, New Mexico History of Medical Waste Regulations 1988 Legislation in response
"ADOPTED STANDARDS FOR THE REGULATION OF MEDICAL WASTE" IN HEALTH CARE FACILITIES LICENSED BY THE MISSISSIPPI STATE DEPARTMENT OF HEALTH
 "ADOPTED STANDARDS FOR THE REGULATION OF MEDICAL WASTE" IN HEALTH CARE FACILITIES LICENSED BY THE MISSISSIPPI STATE DEPARTMENT OF HEALTH REGULATED MEDICAL WASTE "Infectious medical wastes" includes solid
"ADOPTED STANDARDS FOR THE REGULATION OF MEDICAL WASTE" IN HEALTH CARE FACILITIES LICENSED BY THE MISSISSIPPI STATE DEPARTMENT OF HEALTH REGULATED MEDICAL WASTE "Infectious medical wastes" includes solid
MEDICAL WASTE MANAGEMENT
 MEDICAL WASTE MANAGEMENT Biological Safety INTRODUCTION PURPOSE Regulated medical waste is a designation for wastes that may contain pathogenic microorganisms which was previously termed infectious waste.
MEDICAL WASTE MANAGEMENT Biological Safety INTRODUCTION PURPOSE Regulated medical waste is a designation for wastes that may contain pathogenic microorganisms which was previously termed infectious waste.
OCCUPATIONAL SAFETY AND ENVIRONMENTAL HEALTH GUIDELINE
 OSEH Occupational Safety & Environmental Health OCCUPATIONAL SAFETY AND ENVIRONMENTAL HEALTH GUIDELINE Subject: Biohazardous (Medical) Waste Disposal Date: 08/19/09 Revision: 03 Page: 1 of 7 TABLE OF Section
OSEH Occupational Safety & Environmental Health OCCUPATIONAL SAFETY AND ENVIRONMENTAL HEALTH GUIDELINE Subject: Biohazardous (Medical) Waste Disposal Date: 08/19/09 Revision: 03 Page: 1 of 7 TABLE OF Section
UCSF Pediatric Dermatology Inpatient Consultation Service Guidelines for Res B Wards (Res Bw)
 UCSF atology Inpatient Consultation Service Guidelines for Res B Wards (Res Bw) PEDIATRIC DERMATOLOGY Ilona Frieden, MD Kelly Cordoro, MD, MD Fellows Pristine Lee, MD Barrett Zlotoff, MD Resident and Conference
UCSF atology Inpatient Consultation Service Guidelines for Res B Wards (Res Bw) PEDIATRIC DERMATOLOGY Ilona Frieden, MD Kelly Cordoro, MD, MD Fellows Pristine Lee, MD Barrett Zlotoff, MD Resident and Conference
IRMC. P.C. No.: 167. SUBJECT: Universal Labeling and Procurement of Specimens
 IRMC P.C. No.: 167 SUBJECT: Universal Labeling and Procurement of Specimens Purpose: Laboratory specimens must be collected and labeled in accordance with all regulatory requirements and standards of Clinical
IRMC P.C. No.: 167 SUBJECT: Universal Labeling and Procurement of Specimens Purpose: Laboratory specimens must be collected and labeled in accordance with all regulatory requirements and standards of Clinical
(800) 786-7235 Cryo-Cell.com
 HEALTHCARE PROVIDER Instructions Keep these instructions in the kit [ When handling blood products, use personal protective measures, maintain aseptic techniques and adequate room for collection processes.
HEALTHCARE PROVIDER Instructions Keep these instructions in the kit [ When handling blood products, use personal protective measures, maintain aseptic techniques and adequate room for collection processes.
STEP-BY-STEP INSTRUCTIONS FOR INVESTIGATIONAL USE. Rapid HCV Antibody Test FOR ORAQUICK RAPID HCV ANTIBODY TEST
 Before performing testing, all operators MUST read and become familiar with Universal Precautions for Prevention of Transmission of Human Immunodeficiency Virus, Hepatitis B Virus, and other Blood-borne
Before performing testing, all operators MUST read and become familiar with Universal Precautions for Prevention of Transmission of Human Immunodeficiency Virus, Hepatitis B Virus, and other Blood-borne
Surgical Coding Errors & English 101. Riva Lee Asbell. Fort Lauderdale, FL
 Surgical Coding Errors & English 101 Riva Lee Asbell Fort Lauderdale, FL INTRODUCTION Many surgical coding mistakes result from misinterpretation of CPT (Current Procedural Terminology) wording. When the
Surgical Coding Errors & English 101 Riva Lee Asbell Fort Lauderdale, FL INTRODUCTION Many surgical coding mistakes result from misinterpretation of CPT (Current Procedural Terminology) wording. When the
Medical Compliance with Billing and Coding 2013: Will your Records Survive an Audit from a Third Party Payer or the OIG?
 Medical Compliance with Billing and Coding 2013: Will your Records Survive an Audit from a Third Party Payer or the OIG? Michael J. McGovern, OD, FAAO; Richard Soden, OD, FAAO American Academy of Optometry
Medical Compliance with Billing and Coding 2013: Will your Records Survive an Audit from a Third Party Payer or the OIG? Michael J. McGovern, OD, FAAO; Richard Soden, OD, FAAO American Academy of Optometry
NC DIVISION OF SERVICES FOR THE BLIND POLICIES AND PROCEDURES VOCATIONAL REHABILITATION
 NC DIVISION OF SERVICES FOR THE BLIND POLICIES AND PROCEDURES VOCATIONAL REHABILITATION Section: E Revision History Revised 01/97; 05/03; 02/08; 04/08; 03/09; 05/09; 12/09; 01/11; 12/14 An individual is
NC DIVISION OF SERVICES FOR THE BLIND POLICIES AND PROCEDURES VOCATIONAL REHABILITATION Section: E Revision History Revised 01/97; 05/03; 02/08; 04/08; 03/09; 05/09; 12/09; 01/11; 12/14 An individual is
ArtisanLink Staining System is an automated special stains slide
 Technical Tips Tips on using the ArtisanLink Special Staining System Jamie Nowacek, BS, HT(ASCP) CM, QIHC, PMP Dako North America, Inc. Carpinteria, CA, USA ArtisanLink Staining System is an automated
Technical Tips Tips on using the ArtisanLink Special Staining System Jamie Nowacek, BS, HT(ASCP) CM, QIHC, PMP Dako North America, Inc. Carpinteria, CA, USA ArtisanLink Staining System is an automated
RADIATION CONTROL TECHNIQUE #2 INSTRUCTIONS FOR PREPARATION OF RADIOACTIVE WASTE FOR DISPOSAL
 RADIATION CONTROL TECHNIQUE #2 INSTRUCTIONS FOR PREPARATION OF RADIOACTIVE WASTE FOR DISPOSAL I. PURPOSE: To establish a standard procedure for preparing Radioactive Waste and Radioactive Mixed Waste (RMW)
RADIATION CONTROL TECHNIQUE #2 INSTRUCTIONS FOR PREPARATION OF RADIOACTIVE WASTE FOR DISPOSAL I. PURPOSE: To establish a standard procedure for preparing Radioactive Waste and Radioactive Mixed Waste (RMW)
PROCUREMENT OF BONE MARROW SPECIMENS
 Page 1 of 8 SITES AFFECTED _X Enterprise Chandler Good Samaritan PROCUREMENT OF BONE MARROW SPECIMENS A technologist at Chandler CORE and Samaritan are assigned to Bone Marrow duties Monday through Friday
Page 1 of 8 SITES AFFECTED _X Enterprise Chandler Good Samaritan PROCUREMENT OF BONE MARROW SPECIMENS A technologist at Chandler CORE and Samaritan are assigned to Bone Marrow duties Monday through Friday
PATIENT GUIDE. Care and Maintenance Drainage Frequency: Max. Drainage Volume: Dressing Option: Clinician s Signature: Every drainage Weekly
 PATIENT GUIDE Care and Maintenance Drainage Frequency: Max. Drainage Volume: Dressing Option: Every drainage Weekly Clinician s Signature: ACCESS SYSTEMS Pleural Space Insertion Site Cuff Exit Site Catheter
PATIENT GUIDE Care and Maintenance Drainage Frequency: Max. Drainage Volume: Dressing Option: Every drainage Weekly Clinician s Signature: ACCESS SYSTEMS Pleural Space Insertion Site Cuff Exit Site Catheter
Introduction A JP Drain is a soft tube and container used to drain fluids that build up under the skin after surgery.
 JP Drain Introduction A JP Drain is a soft tube and container used to drain fluids that build up under the skin after surgery. This reference summary explains what a JP Drain is and discusses how to take
JP Drain Introduction A JP Drain is a soft tube and container used to drain fluids that build up under the skin after surgery. This reference summary explains what a JP Drain is and discusses how to take
How To Understand And Understand The Rules Of Hazardous Waste
 Understanding Regulated Medical Waste & Best Management Practices Regulations & References The information provided in this presentation is based on the referenced Code of Federal Regulations and State
Understanding Regulated Medical Waste & Best Management Practices Regulations & References The information provided in this presentation is based on the referenced Code of Federal Regulations and State
Biohazardous Waste Disposal. Table of Contents
 1 of 6 The purpose of these guidelines is to ensure compliance with legislation related to the disposal of biohazardous waste. Table of Contents 1. Definitions... 1 2. Biohazardous waste assessment...
1 of 6 The purpose of these guidelines is to ensure compliance with legislation related to the disposal of biohazardous waste. Table of Contents 1. Definitions... 1 2. Biohazardous waste assessment...
Cataracts. Cataract and Primary Eye Care Service...215-928-3041. Main Number...215-928-3000. Physician Referral...1-877-AT-WILLS 1-877-289-4557
 Main Number...215-928-3000 Physician Referral...1-877-AT-WILLS 1-877-289-4557 Emergency Service...215-503-8080 Cataract and Primary Eye Care Service...215-928-3041 Retina Service... 215-928-3300 Cataract
Main Number...215-928-3000 Physician Referral...1-877-AT-WILLS 1-877-289-4557 Emergency Service...215-503-8080 Cataract and Primary Eye Care Service...215-928-3041 Retina Service... 215-928-3300 Cataract
Appendix H Managing Biohazardous Waste SOP
 Biohazardous waste is managed under the State of Rhode Island s Regulated Medical Waste Regulations (Regulation DEM-OWM-MW-1-2009, amended July, 2010). http://www.dem.ri.gov/pubs/regs/regs/waste/medwaste10.pdf
Biohazardous waste is managed under the State of Rhode Island s Regulated Medical Waste Regulations (Regulation DEM-OWM-MW-1-2009, amended July, 2010). http://www.dem.ri.gov/pubs/regs/regs/waste/medwaste10.pdf
Welcome to our Office!
 Randy Burks, MD, FACS Raymond Gailitis, MD, FACS Northwest Medical Plaza 5800 Colonial Drive, Suite 100-103 Margate, Florida 33063 954-977-8770 Comprehensive Eye Care 954-969-0090 LASIK 954-977-8774 Fax
Randy Burks, MD, FACS Raymond Gailitis, MD, FACS Northwest Medical Plaza 5800 Colonial Drive, Suite 100-103 Margate, Florida 33063 954-977-8770 Comprehensive Eye Care 954-969-0090 LASIK 954-977-8774 Fax
Biohazardous Waste Management Plan
 Central Michigan University Biohazardous Waste Management Plan This document has been prepared to provide guidance to Central Michigan University (CMU) employees in the use and disposal of biohazardous
Central Michigan University Biohazardous Waste Management Plan This document has been prepared to provide guidance to Central Michigan University (CMU) employees in the use and disposal of biohazardous
Safe Handling of Cytotoxic Materials
 Safe Handling of Cytotoxic Materials Kara Henman RN, MN, CON(C) Oncology Practice Consultant Cancer Care Nova Scotia What are hazardous drugs? Chemotherapy Immunosuppressive agents Biological agents Antiviral
Safe Handling of Cytotoxic Materials Kara Henman RN, MN, CON(C) Oncology Practice Consultant Cancer Care Nova Scotia What are hazardous drugs? Chemotherapy Immunosuppressive agents Biological agents Antiviral
6 Body Fluid Stains and Standards
 6 Body Fluid Stains and Standards Laboratory examination of body fluids (i.e., blood, semen, saliva, etc.) may produce significant information in certain investigations. This chapter considers the recognition,
6 Body Fluid Stains and Standards Laboratory examination of body fluids (i.e., blood, semen, saliva, etc.) may produce significant information in certain investigations. This chapter considers the recognition,
INSTITUTIONAL POLICY AND PROCEDURE (IPP) Department: Manual: Section:
 HOSPITAL NAME INSTITUTIONAL POLICY AND PROCEDURE (IPP) Department: Manual: Section: TITLE/DESCRIPTION POLICY NUMBER HANDLING OF H AZARDOUS DRUGS EFFECTIVE DATE REVIEW DUE REPLACES NUMBER NO. OF PAGES APPROVED
HOSPITAL NAME INSTITUTIONAL POLICY AND PROCEDURE (IPP) Department: Manual: Section: TITLE/DESCRIPTION POLICY NUMBER HANDLING OF H AZARDOUS DRUGS EFFECTIVE DATE REVIEW DUE REPLACES NUMBER NO. OF PAGES APPROVED
Biological Safety Program
 Risk Management & Safety Main Office, Wyoming Hall Phone: (307) 766-3277 Fax: (307)766-6116 Regulated Materials Management Center Phone: (307)766-3696 Fax: (307)766-3699 Web: www.uwyo.edu/ehs Email: UWEHS@uwyo.edu
Risk Management & Safety Main Office, Wyoming Hall Phone: (307) 766-3277 Fax: (307)766-6116 Regulated Materials Management Center Phone: (307)766-3696 Fax: (307)766-3699 Web: www.uwyo.edu/ehs Email: UWEHS@uwyo.edu
Management Plan For Control of Blood-borne Pathogens, Infectious Wastes and Other Potentially Hazardous Biological Agents
 Management Plan For Control of Blood-borne Pathogens, Infectious Wastes and Other Potentially Hazardous Biological Agents 1.0 Executive Summary 1.1 The Bloodborne Pathogen standard, initiated by OSHA,
Management Plan For Control of Blood-borne Pathogens, Infectious Wastes and Other Potentially Hazardous Biological Agents 1.0 Executive Summary 1.1 The Bloodborne Pathogen standard, initiated by OSHA,
MEDICAL WASTE DEFINITION OF TERMS HEALTH & SAFETY CODE, PART 14; COUNTY ORDINANCE #7646
 GARY W. ERBECK DIRECTOR County of San D iego DEPARTMENT OF ENVIRONMENTAL HEALTH HAZARDOUS MATERIALS DIVISION P.O. BOX 129261, SAN DIEGO, CA 92112-9261 (619) 338-2222 FAX (619) 338-2377 1-800 - 253-9933
GARY W. ERBECK DIRECTOR County of San D iego DEPARTMENT OF ENVIRONMENTAL HEALTH HAZARDOUS MATERIALS DIVISION P.O. BOX 129261, SAN DIEGO, CA 92112-9261 (619) 338-2222 FAX (619) 338-2377 1-800 - 253-9933
Laser Procedure Note
 Laser Procedure Note Patient Name Date 1. Pre procedure diagnosis 2. Procedure and Eye Eye: 3. Pre procedure topical medications administered (and time) 4. Vital signs BP / HR Acuity OD: OS: 5. IOP: OD
Laser Procedure Note Patient Name Date 1. Pre procedure diagnosis 2. Procedure and Eye Eye: 3. Pre procedure topical medications administered (and time) 4. Vital signs BP / HR Acuity OD: OS: 5. IOP: OD
ATTACHMENT I TO APPENDIX B OF UNOS BYLAWS
 ATTACHMENT I TO APPENDIX B OF UNOS BYLAWS Designated Transplant Program Criteria XIII. Transplant Programs. A. In order to qualify for membership, a transplant program must utilize, for its histocompatibility
ATTACHMENT I TO APPENDIX B OF UNOS BYLAWS Designated Transplant Program Criteria XIII. Transplant Programs. A. In order to qualify for membership, a transplant program must utilize, for its histocompatibility
Information for patients and nurses
 Information for patients and nurses Rocket IPC Pleural Catheter Indwelling Catheter Rocket Indwelling Pleural Catheter (IPC) Contents Contact Information...03 What s in the Rocket Dressing Pack and Bottle
Information for patients and nurses Rocket IPC Pleural Catheter Indwelling Catheter Rocket Indwelling Pleural Catheter (IPC) Contents Contact Information...03 What s in the Rocket Dressing Pack and Bottle
After Glaucoma Surgery
 2011 After Glaucoma Surgery Eye Care Centre Please bring this booklet to the hospital with you on the day of your operation. After Glaucoma Surgery What is in this booklet? How is the surgery done?...
2011 After Glaucoma Surgery Eye Care Centre Please bring this booklet to the hospital with you on the day of your operation. After Glaucoma Surgery What is in this booklet? How is the surgery done?...
PEAC CENTRAL LABORATORY SAMPLING MANUAL
 PEAC CENTRAL LABORATORY SAMPLING MANUAL FINAL REVISED VERSION (9.0) PEAC Clinical Study Final 1.0 2 TABLE OF CONTENTS Page No. 1. PEAC CENTRAL LABORATORY - CONTACT NUMBERS... 3 2. IMPORTANT INFORMATION...
PEAC CENTRAL LABORATORY SAMPLING MANUAL FINAL REVISED VERSION (9.0) PEAC Clinical Study Final 1.0 2 TABLE OF CONTENTS Page No. 1. PEAC CENTRAL LABORATORY - CONTACT NUMBERS... 3 2. IMPORTANT INFORMATION...
Biomedical Waste Disposal Procedures
 UNIVERSITY OF OTTAWA Subject: Biomedical Waste Disposal Procedures No.: 1 Issued by: Office of Risk Management (ORM) Revised: Sept 2007 Target Group: Faculties and Services Effective: Oct 2004 Biomedical
UNIVERSITY OF OTTAWA Subject: Biomedical Waste Disposal Procedures No.: 1 Issued by: Office of Risk Management (ORM) Revised: Sept 2007 Target Group: Faculties and Services Effective: Oct 2004 Biomedical
Beaver-Visitec International. Ophthalmic and Microsurgical Product Catalog
 Beaver-Visitec International Ophthalmic and Microsurgical Product Catalog Eye Spears Beaver-Visitec offers an extensive range of Eye Spears that are designed to meet the needs of clinicians for a variety
Beaver-Visitec International Ophthalmic and Microsurgical Product Catalog Eye Spears Beaver-Visitec offers an extensive range of Eye Spears that are designed to meet the needs of clinicians for a variety
CONSENT FOR CATARACT SURGERY REQUEST FOR SURGICAL OPERATION / PROCEDURE AND ANAESTHETIC
 CONSENT FOR CATARACT SURGERY REQUEST FOR SURGICAL OPERATION / PROCEDURE AND ANAESTHETIC Your doctor has indicated that the condition of your eye appears stable and your cataract surgery and/or implantation
CONSENT FOR CATARACT SURGERY REQUEST FOR SURGICAL OPERATION / PROCEDURE AND ANAESTHETIC Your doctor has indicated that the condition of your eye appears stable and your cataract surgery and/or implantation
We Believe the Possibilities. Delphic AP
 We Believe the Possibilities. Delphic AP Designer Software for your pathology workflow. Delphic AP Tracks the specimen process cycle Labels cassettes and slides Enables sophisticated report creation Send
We Believe the Possibilities. Delphic AP Designer Software for your pathology workflow. Delphic AP Tracks the specimen process cycle Labels cassettes and slides Enables sophisticated report creation Send
How To Remove An Eye From An Eye
 p r o c e d u r e s p r o O P H T H L M O L O G Y Sheryl G. Krohne, DVM, MS, Diplomate CVO, Purdue University Subconjunctival Enucleation Surgery in Dogs & Cats Enucleation surgery, a common procedure
p r o c e d u r e s p r o O P H T H L M O L O G Y Sheryl G. Krohne, DVM, MS, Diplomate CVO, Purdue University Subconjunctival Enucleation Surgery in Dogs & Cats Enucleation surgery, a common procedure
Ministry Saint Michael s Hospital Laboratory LABORATORY POLICY AND PROCEDURE
 Ministry Saint Michael s Hospital Laboratory LABORATORY POLICY AND PROCEDURE TITLE/SUBJECT: FILE NUMBER: ASSIGNED MANUALS: ORIGINATION DATE: 11/21/97 EFFECTIVE DATE: 10/31/2012 DISTRIBUTION: SMH Main Laboratory
Ministry Saint Michael s Hospital Laboratory LABORATORY POLICY AND PROCEDURE TITLE/SUBJECT: FILE NUMBER: ASSIGNED MANUALS: ORIGINATION DATE: 11/21/97 EFFECTIVE DATE: 10/31/2012 DISTRIBUTION: SMH Main Laboratory
HAEMATOLOGY LABORATORY
 HAEMATOLOGY LABORATORY Head of Unit : Dr Raudhawati Osman Phone : +603-26155281 Email : raudhaosman@yahoo.com 1. INTRODUCTION The Haematology Unit, Department of Pathology, HKL provides tertiary diagnostic
HAEMATOLOGY LABORATORY Head of Unit : Dr Raudhawati Osman Phone : +603-26155281 Email : raudhaosman@yahoo.com 1. INTRODUCTION The Haematology Unit, Department of Pathology, HKL provides tertiary diagnostic
General Information and Guidelines. Submission of blood tubes
 General Information and Guidelines Submission of blood tubes Note: Do not submit blood samples in syringes (especially with needles attached). Syringes that are submitted to the lab with the needles attached
General Information and Guidelines Submission of blood tubes Note: Do not submit blood samples in syringes (especially with needles attached). Syringes that are submitted to the lab with the needles attached
Policy for the Disposal of Biological Waste
 Policy for the Disposal of Biological Waste I. Biological Waste II. Regulated Medical Waste Prepared by: Rutgers Environmental Health and Safety 24 Street 1603 Building 4127, Livingston Campus Piscataway,
Policy for the Disposal of Biological Waste I. Biological Waste II. Regulated Medical Waste Prepared by: Rutgers Environmental Health and Safety 24 Street 1603 Building 4127, Livingston Campus Piscataway,
Delphic AP. Designer software for your pathology workflow. We Believe the Possibilities.
 We Believe the Possibilities. Delphic AP Designer software for your pathology workflow. Sysmex Software Solutions. Shaping the advancement of healthcare. Delphic AP 1 Delphic AP Delphic AP has been specifically
We Believe the Possibilities. Delphic AP Designer software for your pathology workflow. Sysmex Software Solutions. Shaping the advancement of healthcare. Delphic AP 1 Delphic AP Delphic AP has been specifically
RPCI 004 v.002 Staining Procedure For all Directly Conjugated Reagents (Whole Blood Method)
 Immune Tolerance Network RPCI 004 v.002 Staining Procedure For all Directly Conjugated Reagents (Whole Blood Method) Author: Paul Wallace, Director, RPCI Laboratory of Flow Cytometry Approved by: Paul
Immune Tolerance Network RPCI 004 v.002 Staining Procedure For all Directly Conjugated Reagents (Whole Blood Method) Author: Paul Wallace, Director, RPCI Laboratory of Flow Cytometry Approved by: Paul
Utah Division of Solid and Hazardous Waste Solid Waste Management Program
 Utah Division of Solid and Hazardous Waste Solid Waste Management Program Mailing Address Office Location Phone (801) 536-0200 P.O. Box 144880 195 North 1950 West Fax (801) 536-0222 Salt Lake City, Utah
Utah Division of Solid and Hazardous Waste Solid Waste Management Program Mailing Address Office Location Phone (801) 536-0200 P.O. Box 144880 195 North 1950 West Fax (801) 536-0222 Salt Lake City, Utah
Guidelines for the Safe Transport of Infectious Substances and Diagnostic Specimens
 Distr.: General Orig.: English Guidelines for the Safe Transport of Infectious Substances and Diagnostic Specimens WORLD HEALTH ORGANIZATION Division of Emerging and Other Communicable Diseases Surveillance
Distr.: General Orig.: English Guidelines for the Safe Transport of Infectious Substances and Diagnostic Specimens WORLD HEALTH ORGANIZATION Division of Emerging and Other Communicable Diseases Surveillance
UNIVERSITY OF NORTH FLORIDA BIOMEDICAL WASTE MANAGEMENT PLAN DEVELOPED BY: ENVIRONMENTAL HEALTH, SAFETY, INSURANCE & RISK MANAGEMENT
 UNIVERSITY OF NORTH FLORIDA BIOMEDICAL WASTE MANAGEMENT PLAN DEVELOPED BY: ENVIRONMENTAL HEALTH, SAFETY, INSURANCE & RISK MANAGEMENT June 2008 Table of Contents Section Page Background 1 Definitions 1-2
UNIVERSITY OF NORTH FLORIDA BIOMEDICAL WASTE MANAGEMENT PLAN DEVELOPED BY: ENVIRONMENTAL HEALTH, SAFETY, INSURANCE & RISK MANAGEMENT June 2008 Table of Contents Section Page Background 1 Definitions 1-2
HARVESTING AND CRYOPRESERVATION OF HUMAN EMBRYONIC STEM CELLS (hescs)
 HARVESTING AND CRYOPRESERVATION OF HUMAN EMBRYONIC STEM CELLS (hescs) OBJECTIVE: can be cryopreserved in a liquid nitrogen (LN 2 ) freezer for long-term storage. This Standard Operating Procedure (SOP)
HARVESTING AND CRYOPRESERVATION OF HUMAN EMBRYONIC STEM CELLS (hescs) OBJECTIVE: can be cryopreserved in a liquid nitrogen (LN 2 ) freezer for long-term storage. This Standard Operating Procedure (SOP)
MEDICAL WASTE DISPOSAL POLICY. EFFECTIVE DATE: January 31, 1991 REVISED DATE: April 1, 2008 I. AUTHORITY TO ESTABLISH DISPOSAL POLICY:
 MEDICAL WASTE DISPOSAL POLICY EFFECTIVE DATE: January 31, 1991 REVISED DATE: April 1, 2008 I. AUTHORITY TO ESTABLISH DISPOSAL POLICY: The authority by which the Municipality of Anchorage, Solid Waste Services
MEDICAL WASTE DISPOSAL POLICY EFFECTIVE DATE: January 31, 1991 REVISED DATE: April 1, 2008 I. AUTHORITY TO ESTABLISH DISPOSAL POLICY: The authority by which the Municipality of Anchorage, Solid Waste Services
To provide direction for the safe handling, administration and disposal of hazardous drugs.
 Subsection: MEDICATION Related terms: Cytotoxic Drugs, Antineoplastic Drugs Authorized by: Clinical Directors CS-04-02-01 Page 1 of 9 Date Established: October 2006 Date For Review: September 2014 Dates
Subsection: MEDICATION Related terms: Cytotoxic Drugs, Antineoplastic Drugs Authorized by: Clinical Directors CS-04-02-01 Page 1 of 9 Date Established: October 2006 Date For Review: September 2014 Dates
Particularly Hazardous Substances (PHS) Standard Operating Procedure (SOP)
 Particularly Hazardous Substances (PHS) Standard Operating Procedure (SOP) Formaldehyde, formalin, paraformaldehyde solutions, and paraformaldehyde solids Principal Investigator: Room & Building #: Department:
Particularly Hazardous Substances (PHS) Standard Operating Procedure (SOP) Formaldehyde, formalin, paraformaldehyde solutions, and paraformaldehyde solids Principal Investigator: Room & Building #: Department:
Caring for Your PleurX Pleural Catheter
 Caring for Your PleurX Pleural Catheter A PleurX Pleural Catheter has been placed in your chest through a small incision in your skin into the pleural space (see picture below). This allows you to drain
Caring for Your PleurX Pleural Catheter A PleurX Pleural Catheter has been placed in your chest through a small incision in your skin into the pleural space (see picture below). This allows you to drain
Nick Strouthidis MBBS MD PhD FRCS FRCOphth FRANZCO CONSULTANT OPHTHALMIC SURGEON
 TUBES/SHUNTS TUBES/SHUNTS Implantation of a glaucoma drainage device (GDD - also known as a tube or aqueous shunt) works by diverting aqueous from the front of the eye via a tube to a drainage plate stitched
TUBES/SHUNTS TUBES/SHUNTS Implantation of a glaucoma drainage device (GDD - also known as a tube or aqueous shunt) works by diverting aqueous from the front of the eye via a tube to a drainage plate stitched
DRAFT OVERDOSE ASSESSMENT, RESPONSE AND TREATMENT POLICY FOR SELECTED ADULT FACILITES
 1 DRAFT OVERDOSE ASSESSMENT, RESPONSE AND TREATMENT POLICY FOR SELECTED ADULT FACILITES PURPOSE To provide guidelines for staff response when a client is found unconscious or not breathing. APPLICABILITY
1 DRAFT OVERDOSE ASSESSMENT, RESPONSE AND TREATMENT POLICY FOR SELECTED ADULT FACILITES PURPOSE To provide guidelines for staff response when a client is found unconscious or not breathing. APPLICABILITY
COW S EYE dissection. Dissecting a Cow s Eye Step-by-Step Instructions. Safety first!
 COW S EYE dissection Dissecting a Cow s Eye Step-by-Step Instructions One way to figure out how something works is to look inside it. To learn about how your eyes work, you can dissect, or take apart,
COW S EYE dissection Dissecting a Cow s Eye Step-by-Step Instructions One way to figure out how something works is to look inside it. To learn about how your eyes work, you can dissect, or take apart,
Laboratory Biosafty In Molecular Biology and its levels
 Laboratory Biosafty In Molecular Biology and its levels Workshop 16-17 Oct..2012 Guidelines Does not mean optional Laboratory Biosafety The Laboratory Biosafety Manual is an important WHO publication
Laboratory Biosafty In Molecular Biology and its levels Workshop 16-17 Oct..2012 Guidelines Does not mean optional Laboratory Biosafety The Laboratory Biosafety Manual is an important WHO publication
ILLINOIS DEPARTMENT OF CENTRAL MANAGEMENT SERVICES CLASS SPECIFICATION CLINICAL LABORATORY TECHNICIAN SERIES
 ILLINOIS DEPARTMENT OF CENTRAL MANAGEMENT SERVICES CLASS SPECIFICATION CLINICAL LABORATORY TECHNICIAN SERIES CLASS TITLE POSITION CODE CLINICAL LABORATORY TECHNICIAN I 08215 CLINICAL LABORATORY TECHNICIAN
ILLINOIS DEPARTMENT OF CENTRAL MANAGEMENT SERVICES CLASS SPECIFICATION CLINICAL LABORATORY TECHNICIAN SERIES CLASS TITLE POSITION CODE CLINICAL LABORATORY TECHNICIAN I 08215 CLINICAL LABORATORY TECHNICIAN
TEST UPDATE: Ova and Parasites Effective: September 2008
 TEST UPDATE: Ova and Parasites Effective: September 2008 Related Information CPT Cryptosporidium by FA Giardia by FA 87177 (ova and parasite exam); 87209 (complex special stain) Synonyms Fecal Parasite
TEST UPDATE: Ova and Parasites Effective: September 2008 Related Information CPT Cryptosporidium by FA Giardia by FA 87177 (ova and parasite exam); 87209 (complex special stain) Synonyms Fecal Parasite
Biosafety Spill Response Guide
 Yale University Office of Environmental Health & Safety Biosafety Spill Response Guide Office of Environmental Health & Safety 135 College Street, 1 st Floor, New Haven, CT 06510 Telephone: 203-785-3550
Yale University Office of Environmental Health & Safety Biosafety Spill Response Guide Office of Environmental Health & Safety 135 College Street, 1 st Floor, New Haven, CT 06510 Telephone: 203-785-3550
BOSTON COLLEGE ENVIRONMENTAL HEALTH AND SAFETY LABORATORY GUIDE FOR RENOVATIONS, REMODELS, MOVES AND TERMINATIONS
 BOSTON COLLEGE ENVIRONMENTAL HEALTH AND SAFETY LABORATORY GUIDE FOR RENOVATIONS, REMODELS, MOVES AND TERMINATIONS I. Policy for Termination of Laboratory Use of Hazardous Materials The Principal Investigator
BOSTON COLLEGE ENVIRONMENTAL HEALTH AND SAFETY LABORATORY GUIDE FOR RENOVATIONS, REMODELS, MOVES AND TERMINATIONS I. Policy for Termination of Laboratory Use of Hazardous Materials The Principal Investigator
Attachment D Infection Control Policy METHODS OF IMPLEMENTATION AND CONTROL
 Attachment D Infection Control Policy METHODS OF IMPLEMENTATION AND CONTROL OSHA requires that the ECP include a schedule and method of implementation for the various requirements of the standard. The
Attachment D Infection Control Policy METHODS OF IMPLEMENTATION AND CONTROL OSHA requires that the ECP include a schedule and method of implementation for the various requirements of the standard. The
WHAT IS A CATARACT, AND HOW IS IT TREATED?
 4089 TAMIAMI TRAIL NORTH SUITE A103 NAPLES, FL 34103 TELEPHONE (239) 262-2020 FAX (239) 435-1084 DOES THE PATIENT NEED OR WANT A TRANSLATOR, INTERPRETOR OR READER? YES NO TO THE PATIENT: You have the right,
4089 TAMIAMI TRAIL NORTH SUITE A103 NAPLES, FL 34103 TELEPHONE (239) 262-2020 FAX (239) 435-1084 DOES THE PATIENT NEED OR WANT A TRANSLATOR, INTERPRETOR OR READER? YES NO TO THE PATIENT: You have the right,
