Australian guidelines for drug donations to developing countries
|
|
|
- Dominick Sims
- 9 years ago
- Views:
Transcription
1 Australian guidelines for drug donations to developing countries Endorsed by the Australian Pharmaceutical Advisory Council November 1996 Amendments endorsed November 2000 APAC AUSTRALIAN PHARMACEUTICAL ADVISORY COUNCIL
2 Australian guidelines for drug donations to developing countries APAC AUSTRALIAN PHARMACEUTICAL ADVISORY COUNCIL Endorsed by the Australian Pharmaceutical Advisory Council NOVEMBER 1996 Amendments endorsed November 2000 Australian Government Publishing Service Canberra
3 Commonwealth of Australia 2001 ISBN This work is copyright. It may be reproduced in whole or part subject to the inclusion of an acknowledgment of the source and no commercial usage or sale. Reproduction for purposes other than those indicated above requires the written permission of the Commonwealth available through AusInfo. Requests and inquiries should be addressed to the Manager, Legislative Services, AusInfo, GPO Box 1920, Canberra ACT First printed 1997 Reprinted with amendments 2001 Publication approval number: 2488 Publications Production Unit (Public Affairs, Parliamentary and Access Branch) Commonwealth Department of Health and Aged Care Canberra ii
4 Acknowledgments APAC wishes to acknowledge the valuable assistance provided in the development of the guidelines by: Beverley Snell, Jonathan Dartnell, Virginia Ford, and Margaret Duguid (Writing Working Party, Society of Hospital Pharmacists of Australia). Dr Hans Hogerzeil (Medical Officer, WHO Drug Action Program). Dr Wilbert Bannenberg (Consultants for Health and Development, The Netherlands). Dr Edelisa Carandang (Philippine National Drug Policy, Bureau of Food and Drugs, Department of Health, Philippines). iii
5 Introduction These Australian guidelines are based on the international Guidelines for Drug Donations developed by the World Health Organisation (WHO) in The WHO s guidelines reflect a consensus between the major international agencies active in humanitarian emergency relief: WHO, Office of the United Nations High Commission for Refugees (UNHCR), United Nations Children s Fund (UNICEF), International Committee of the Red Cross, Medecins sans Frontieres, Churches Action for Health and the World Council of Churches. They aim to improve the quality of drug donations and are intended to serve as a basis for national and institutional guidelines. Scenarios associated with drug donations range from acute emergencies to non-emergency development programs. Although there are differences between these scenarios, there are basic rules for an appropriate response that apply to all. As with the guidelines prepared by WHO, this Australian document starts with a discussion on the need for guidelines, followed by the presentation of the core principles for appropriate donations. The Australian guidelines have been endorsed by the Australian Pharmaceutical Advisory Council (APAC). APAC expresses its appreciation to the Society of Hospital Pharmacists of Australia for its assistance in developing these guidelines. 1
6 Why do we need guidelines for donations of drugs to developing countries? The contribution of medicines in the form of donations to developing countries or in disaster situations is often viewed by communities in developed countries, such as Australia, as a useful way to provide much needed pharmaceutical supplies. Televised pictures of refugee camps or rural areas lacking the most basic health services often give rise to emotional appeals to do something. As the lack of medicines is often presented as a pressing problem, an immediate donation of drugs is often perceived as the most pragmatic and direct response. Unfortunately not all drug donations are helpful and inappropriate drug donations can be dangerous or useless, and a source of problems for the recipients as the following examples illustrate. Guatemala, 1976 In the first week after the earthquake in Guatemala, 7000 cartons of mixed drugs arrived. These were sorted by a group of 40 pharmacy students at a rate of boxes per day (it took about six months to finish the job). Only 10% of the drugs sent were relevant to the health needs in Guatemala and were adequately labelled. Sudan, 1990 A large consignment of drugs was sent from France to war-devastated southern Sudan. Each box contained a collection of small packets of drugs, some partly used. All were labelled in French, a language not spoken in Sudan. Most were inappropriate, some could be dangerous. There were: contact lens solutions, appetite stimulants, antidepressants and expired antibiotics. Of 50 boxes, 12 contained drugs of some use. It would have been much better to use the money spent on transport to purchase penicillin and other essential drugs from Kenya and to have them sent from there. Eritrea, 1980s During the war for independence, despite careful wording of appeals, much time and energy had to be spent on sorting drug consignments. Examples of inappropriate donations were: seven truck loads of expired aspirin tablets that took six months to burn; a whole container of unsolicited cardiovascular drugs with two months to expiry; and 30,000 half-litre bottles of expired amino-acid infusion that could not be disposed of anywhere near a settlement because of the smell. How else can drug donations cause problems? Most developing countries have national essential drugs lists covering major health problems. Training programs for health personnel focus on the correct use of these drugs. Such developments need support. Donated drugs can frustrate and undermine national drug policies which aim to secure reliable supplies of appropriate, good quality, essential drugs and to promote standard treatment guidelines as a tool to encourage rational drug use. There are also problems with the quality of donated drugs, donation of expired drugs being a common practice. This practice is not acceptable. If the quality of a drug is not suitable for use in Australia, it is not suitable for donation to an overseas country. Apart from the issues of the quality of the products, eg expired/damaged drugs, and the relevance of the drugs to the situation, logistical difficulties can also arise. These difficulties are associated with sorting, storage and distribution which may waste valuable human and financial resources. In addition, transport costs may be higher than the value of the drugs, and local purchase can be more cost effective. 2
7 Guidelines for donating drugs Core principles A donation is intended to assist the recipient. Donations should be based on an expressed need. Donor-initiated donations where the recipients advice has not been sought are unhelpful. Donations must support essential drug policies. Respect must be given for the authority of the recipient and the receiving country s administrative arrangements. There should be no double standards. If the quality of an item is not acceptable in Australia it is unacceptable as a donation. Therefore the collection and redistribution of patients unused medicines is not permitted. Selection of drugs 1. All drug donations should be based on an expressed need. Drugs should not be sent without prior clearance by the recipient. A written request should be obtained from a competent authority in the recipient country. The health facility receiving the drugs should be required to acknowledge receipt of the donated medicines. This provision excludes donor-driven donations, or donations which arrive unannounced and unwanted. It encourages recipients to specify their needs, and empowers them to refuse unwanted gifts. 2. All donated drugs should be on the national list of essential drugs of the recipient country or, if such a national list is not available, on the WHO Model List of Essential Drugs. This provision ensures that drug donations comply with existing government policies and with the essential drugs concept. It aims at maximising the positive impact of the donation, and excludes the donation of unnecessary or dangerous drugs, and drugs which are not specified for use in that country. Copies of the WHO Model List of Essential Drugs are available from Hunter Publications, 58 Gipps Street, Collingwood, Telephone: The presentation and strength of donated drugs should, as much as possible, be similar to those commonly used in the recipient country. Most staff working at different health care levels in the recipient country have been trained to use a certain dosage schedule and dosage forms. Treatment guidelines cannot be constantly changed. Note: Vaccine donations The donation of vaccines is not appropriate because of the logistical problems associated with transport and storage. Donations of money for purchasing vaccines are more helpful, so cold chain facilities can be put in place, and the right quantity and type of vaccine and associated supplies can be ordered at the right time. Donations for vaccine purchase should only be directed to a responsible agency which has an accepted role in immunisation programs such as Australian Red Cross, Medecins Sans Frontieres, UNICEF, Save The Children Fund and World Vision Australia. 3
8 Quality and shelf-life 4. All donated drugs should comply with quality standards in both the donor and recipient country. This provision prevents double standards. For example, donations of unused drugs (returned drugs from patients or doctors free samples), which would not be acceptable for use in Australia should not be donated to another country. 5. All donated drugs should have a remaining shelf-life of at least one year after the arrival in the recipient country, with an exception for direct donations to a specific health program in response to an urgent and special request. This exception may be made provided; the date of arrival and the expiry dates of the drugs is advised to the responsible recipient professional, and that they acknowledge that they are aware of the shelf-life; and, that prior to expiration, the quantity and remaining shelf-life allow for proper administration. Considerable periods of time may elapse before donated drugs are transported, cleared through customs, relocated to a warehouse, contents identified and recorded, and the drugs dispatched to the place of use. Distribution may take many months as drugs are moved from a central store, to provincial stores, to district hospitals and other peripheral health facilities. Very few possibilities for immediate distribution exist. Presentation, packing and labelling 6. All drugs should be labelled in a language that is easily understood in the recipient country. The label should contain at least the International Non-proprietary Name (generic name), dosage form, strength, name of manufacturer, storage conditions and expiry date. Product Information and Consumer Product Information (where available) should be included for each of the drugs sent. Generic names can be understood in any country where Roman script is being used. Training programs are based on the use of generic names. Receiving the same drug under many different and often unknown brand names is very confusing to the health workers and can even be dangerous. The use of generic names also prevents brand-name loyalty being inadvertently promoted by the donor. Many health facilities in developing countries do not have access to current, complete and accurate drug information. Where national treatment guidelines are not in use, it is important that prescribers and dispensers are provided with this information to ensure safe use. 7. As much as possible, donated drugs should be presented in larger quantity units. Larger quantity packs are more cost-effective as they are cheaper and less bulky to transport. This should not be taken as encouragement to repack drugs from different batch lots and expiry dates into one container. Export and transport 8. All drug donations should be packed in strong outer cartons and be accompanied by a detailed packing list which should specify the contents of each carton by generic name, quantity and expiry date. Cartons should be numbered and the contents of each carton listed in detail in the accompanying documents. In addition, the contents of cartons should be marked on the outside of cartons, preferably using a code system. In addition to knowing what is coming the recipient is assisted when the consignment arrives. S/he will know just where to find items which may be needed urgently (instead of having to unpack the 4
9 whole shipment) and will be able to decide where to direct items that may be intended for different centres. A code numbering system for listing contents on the outside of packs is better than the use of the names of items which can facilitate pilferage. 9. Preparation for consignment of goods must be undertaken in close co-operation with the recipient to determine the transport and clearance arrangements, documents needed by the recipient, and the costs that need to be met by the donor. The recipient will require advance details of the content of the shipment and its time of arrival. In most cases international transport, customs warehousing, clearance costs and internal transport will need to be paid by the donor. However, in some cases the recipient is able to cover the cost of clearance and internal transport. Agencies should make their own arrangements to comply with customs documentation requirements at all dispatch, transit and entry points. The recipient will know the local requirements for receiving and clearing consignments and will be able to make preparatory arrangements for prompt clearance in advance. It is most important that shipments are met on arrival. Having a consignment lying on the docks or in a store somewhere, without attention, is also potentially disastrous, and can be very expensive. In addition, these provisions prevent the recipient having to spend effort and money on the clearance and transport of unannounced consignments of unwanted items and enables the recipient to review the list of donated items at an early stage. However, it does not exclude costs being paid by the recipient, if prior notice has been given and agreement has been reached. Federal Permits 10. The export of certain drugs, including drugs of addiction, ie, drugs listed in Schedule 8 of the Standard for the Uniform Scheduling of Drugs and Poisons, psychotropic substances such as stimulants, barbiturates and benzodiazepines as well as drugs and chemicals of concern such as ephedrine and pseudoephedrine, requires a written permission to export from the Austalian Government Department of Health and Ageing. This written permission must be obtained prior to the shipment leaving Australia. Special conditions apply to the issue of a written permission to cover an immediate response to an emergency situation. The person responsible for shipping the donation should contact Therapeutic Goods Administration The Treaties and Monitoring Team PO Box 100 WODEN ACT 2606 Telephone: (06) Facsimile: (06) with a list of drugs proposed to be donated, to confirm whether a licence and export permit is required. If the items do not require an export permit, and provided the drugs have been legally acquired within Australia, are not for commercial supply and are not intended for use in clinical trials, there is no requirement to provide any documentation to the Federal authorities. 5
10 How can Australians help? The most appropriate response to appeals for help or ongoing assistance to developing countries is a financial contribution. It is usually cheaper for drugs to be purchased locally, or from specialist non-profit procuring agencies which are closer to the scene. Local procurement, which involves only a fraction of the transport costs, encourages locally sustainable drug availability and support for local industry is a more development oriented approach. Provision of funds for direct procurement from specialist non-profit agencies such as IDA - International Dispensary Association, is the most helpful strategy when supplies are not available locally. In emergencies, the most appropriate action may be the purchase of the World Health Organisation Emergency Health Kits which include drugs and medical supplies for a population of 10,000 people for a period of three months. Delivery within 48 hours can be arranged through: 1. IDA - International Dispensary Association, PO Box AB Amsterdam THE NETHERLANDS Telephone: Facsimile: [email protected] or 2. UNICEF - United Nations Children s Fund Unicef Plads - Freeport DK-2100 Copenhagen 0 DENMARK Telephone: (35) Facsimile: (35) Telex: Donations of funds for purchase of these kits can be very helpful. Donors wishing to identify the coordinating agency for a current emergency can find information on the Internet (World Wide Web) under United Nations Departments UNDHA - UN Department of Humanitarian Affairs: UNDHA Relief Alternatively information can be sought from: Medical Advisor, Australian Red Cross National Office, telephone , Medical Advisor, Medecins sans Frontieres, National Office, telephone ; Emergency Program Officer, AusAID Human Relief Programs, telephone
11 References 1. WHO. Guidelines for Drug Donations. Geneva: WHO, (WHO/DAP/96.2) 2. Interagency Guidelines for Drug Donations, Published on behalf of agencies by the World Health Organization. 3. Guidelines for donors and recipients of pharmaceutical donations. Geneva: Christian Medical Commission of the World Council of Churches, The New Emergency Health Kit. Geneva: World Health Organization, The use of essential drugs. Geneva: World Health Organization, Technical Report Series
GUIDELINES FOR DRUG DONATIONS. A WHO/ Government of Vanuatu guide for those accepting and making donations of drugs.
 INTRODUCTION GUIDELINES FOR DRUG DONATIONS A WHO/ Government of Vanuatu guide for those accepting and making donations of drugs. Why do we need guidelines on the donation of drugs Donations of medicines
INTRODUCTION GUIDELINES FOR DRUG DONATIONS A WHO/ Government of Vanuatu guide for those accepting and making donations of drugs. Why do we need guidelines on the donation of drugs Donations of medicines
SADC GUIDELINES ON IMPORT AND EXPORT PROCEDURES FOR PHARMACEUTICAL PRODUCTS
 SADC GUIDELINES ON IMPORT AND EXPORT PROCEDURES FOR PHARMACEUTICAL PRODUCTS June 2006 1 INTRODUCTION Public health concerns demand that the manufacture of pharmaceutical products and their subsequent handling
SADC GUIDELINES ON IMPORT AND EXPORT PROCEDURES FOR PHARMACEUTICAL PRODUCTS June 2006 1 INTRODUCTION Public health concerns demand that the manufacture of pharmaceutical products and their subsequent handling
Guidelines for Medicine Donations Revised 2010
 Guidelines for Medicine Donations Revised 2010 World Health Organization (WHO) Ecumenical Pharmaceutical Network (EPN) International Pharmaceutical Federation (FIP) International Federation of Red Cross
Guidelines for Medicine Donations Revised 2010 World Health Organization (WHO) Ecumenical Pharmaceutical Network (EPN) International Pharmaceutical Federation (FIP) International Federation of Red Cross
Novartis disaster relief process
 Novartis disaster relief process Why a Disaster Relief Process? 2 Novartis disaster relief process 2014 Tailored approach based on the scale of disaster Global scale Regional scale Local scale Indicators
Novartis disaster relief process Why a Disaster Relief Process? 2 Novartis disaster relief process 2014 Tailored approach based on the scale of disaster Global scale Regional scale Local scale Indicators
Assessment of Drugs Donation Practices in Sudan
 Pharmacology & Pharmacy, 2012, 3, 113-118 http://dx.doi.org/10.4236/pp.2012.31017 Published Online January 2012 (http://www.scirp.org/journal/pp) 113 Jacob L. Doro 1, Asim F. Mustafa 2, Ahmed S. Eldalo
Pharmacology & Pharmacy, 2012, 3, 113-118 http://dx.doi.org/10.4236/pp.2012.31017 Published Online January 2012 (http://www.scirp.org/journal/pp) 113 Jacob L. Doro 1, Asim F. Mustafa 2, Ahmed S. Eldalo
chapter 15 Pharmaceutical donations
 Part I: Policy and economic issues Part II: Pharmaceutical management Part III: Management support systems Policy and legal framework Financing and sustainability 9 Pharmaceutical pricing policy 10 Economics
Part I: Policy and economic issues Part II: Pharmaceutical management Part III: Management support systems Policy and legal framework Financing and sustainability 9 Pharmaceutical pricing policy 10 Economics
GUIDELINES FOR THE PREPARATION OF A TRANSPORT EMERGENCY RESPONSE PLAN. Endorsed by ACTDG
 GUIDELINES FOR THE PREPARATION OF A TRANSPORT EMERGENCY RESPONSE PLAN Endorsed by ACTDG Commonwealth of Australia 2003 ISBN 0 642 45037 4 This work is copyright. You may download, display, print and reproduce
GUIDELINES FOR THE PREPARATION OF A TRANSPORT EMERGENCY RESPONSE PLAN Endorsed by ACTDG Commonwealth of Australia 2003 ISBN 0 642 45037 4 This work is copyright. You may download, display, print and reproduce
Medical Mission Pack Program
 Medical Mission Pack Program For over 125 years, Johnson & Johnson has been committed to improving the health and well-being of mothers and children around the world. With the support of its operating
Medical Mission Pack Program For over 125 years, Johnson & Johnson has been committed to improving the health and well-being of mothers and children around the world. With the support of its operating
Formulary notes for the List of Approved Medicines
 Formulary notes for the List of Approved Medicines Last updated May 2016 Contents Introduction... 2 Formulary notes... 2 Generic listing of medicines... 2 PBS items... 2 Restrictions... 3 Applications
Formulary notes for the List of Approved Medicines Last updated May 2016 Contents Introduction... 2 Formulary notes... 2 Generic listing of medicines... 2 PBS items... 2 Restrictions... 3 Applications
msupply Pharmaceutical Supply Chain Software October 2013
 msupply Pharmaceutical Supply Chain Software October 2013 Ease-of-use msupply has almost 15 years experience in providing solutions for medicines and vaccines supply chain, msupply is downloadable for
msupply Pharmaceutical Supply Chain Software October 2013 Ease-of-use msupply has almost 15 years experience in providing solutions for medicines and vaccines supply chain, msupply is downloadable for
CHAPTER 6 PROCUREMENT AND SUPPLY OF PHARMACEUTICAL PRODUCTS IN THE PUBLIC AND PRIVATE MEDICAL SECTORS
 CHAPTER 6 PROCUREMENT AND SUPPLY OF PHARMACEUTICAL PRODUCTS IN THE PUBLIC AND PRIVATE MEDICAL SECTORS Overview 6.1 This chapter sets out the Review Committee s findings and recommendations on procurement
CHAPTER 6 PROCUREMENT AND SUPPLY OF PHARMACEUTICAL PRODUCTS IN THE PUBLIC AND PRIVATE MEDICAL SECTORS Overview 6.1 This chapter sets out the Review Committee s findings and recommendations on procurement
Annex 9 Guide to good storage practices for pharmaceuticals 1
 World Health Organization WHO Technical Report Series, No. 908, 2003 Annex 9 Guide to good storage practices for pharmaceuticals 1 1. Introduction 125 2. Glossary 126 3. Personnel 128 4. Premises and facilities
World Health Organization WHO Technical Report Series, No. 908, 2003 Annex 9 Guide to good storage practices for pharmaceuticals 1 1. Introduction 125 2. Glossary 126 3. Personnel 128 4. Premises and facilities
HIGHLY SPECIALISED DRUGS PROGRAM AND HERCEPTIN PROGRAM. Western Australia Administrative Guidelines
 HIGHLY SPECIALISED DRUGS PROGRAM AND HERCEPTIN PROGRAM Western Australia Administrative Guidelines Pharmaceutical Services Branch Health Protection Group Table of Contents BACKGROUND...1 Overview...1 AHMAC
HIGHLY SPECIALISED DRUGS PROGRAM AND HERCEPTIN PROGRAM Western Australia Administrative Guidelines Pharmaceutical Services Branch Health Protection Group Table of Contents BACKGROUND...1 Overview...1 AHMAC
Schedule 8 and Declared Schedule 4 Medicines Management Policy
 Department of Health and Human Services SYSTEM PURCHASING AND PERFORMANCE - MEDICATION STRATEGY AND REFORM Schedule 8 and Declared Schedule 4 Medicines Management Policy SDMS Id Number: Effective From:
Department of Health and Human Services SYSTEM PURCHASING AND PERFORMANCE - MEDICATION STRATEGY AND REFORM Schedule 8 and Declared Schedule 4 Medicines Management Policy SDMS Id Number: Effective From:
Pharmacy Policy (General)
 WORKSAFE VICTORIA Pharmacy Policy (General) WorkSafe can pay the reasonable costs of medications and other pharmacy items required as a result of a work-related injury or illness in accordance with Victorian
WORKSAFE VICTORIA Pharmacy Policy (General) WorkSafe can pay the reasonable costs of medications and other pharmacy items required as a result of a work-related injury or illness in accordance with Victorian
11 MEDICATION MANAGEMENT
 1 11 MEDICATION MANAGEMENT OVERVIEW OF MEDICATION MANAGEMENT Depending on the size, structure and functions of the health facility, there may be a pharmacy with qualified pharmacists to dispense medication,
1 11 MEDICATION MANAGEMENT OVERVIEW OF MEDICATION MANAGEMENT Depending on the size, structure and functions of the health facility, there may be a pharmacy with qualified pharmacists to dispense medication,
GUIDELINES FOR THE CONTROL AND ADMINISTRATION OF MEDICINES DOMICILIARY CARE AGENCIES
 GUIDELINES FOR THE CONTROL AND ADMINISTRATION OF MEDICINES DOMICILIARY CARE AGENCIES January 2009 Contents Page Number 1.0 Introduction 3 2.0 Background 4 3.0 Criteria 5 3.1 Referral 5 3.2 Levels of assistance/consent
GUIDELINES FOR THE CONTROL AND ADMINISTRATION OF MEDICINES DOMICILIARY CARE AGENCIES January 2009 Contents Page Number 1.0 Introduction 3 2.0 Background 4 3.0 Criteria 5 3.1 Referral 5 3.2 Levels of assistance/consent
UNICEF s Quality Assurance System for procurement of medicines
 UNICEF s Quality Assurance System for procurement of medicines QA Specialist Peter Svarrer Jakobsen Joint UNICEF, UNFPA and WHO Meeting with Manufacturers and Suppliers UN City, Copenhagen 23 September
UNICEF s Quality Assurance System for procurement of medicines QA Specialist Peter Svarrer Jakobsen Joint UNICEF, UNFPA and WHO Meeting with Manufacturers and Suppliers UN City, Copenhagen 23 September
Guidelines for dispensing of medicines 6585 08/10
 for dispensing of medicines 6585 08/10 for dispensing of medicines Contents Introduction 1 Who needs to use these guidelines? 1 Summary of guidelines 1 1 1 Dispensing precaution safety of prescriptions
for dispensing of medicines 6585 08/10 for dispensing of medicines Contents Introduction 1 Who needs to use these guidelines? 1 Summary of guidelines 1 1 1 Dispensing precaution safety of prescriptions
Medication Management Guidelines for Nurses and Midwives
 Medication Management Guidelines for Nurses and Midwives 1. Introduction As the statutory body responsible for the regulation of nursing and midwifery practice in Western Australia (WA), the Nurses & Midwives
Medication Management Guidelines for Nurses and Midwives 1. Introduction As the statutory body responsible for the regulation of nursing and midwifery practice in Western Australia (WA), the Nurses & Midwives
NHS Lanarkshire Care Homes Protocol Group. Care Home Prescriptions - Good Practice Guide
 NHS Lanarkshire Care Homes Protocol Group Care Home Prescriptions - Good Practice Guide Date of Publication Review Date August 2015 Responsible Author Francesca Aaen Care Homes Pharmacist on behalf of
NHS Lanarkshire Care Homes Protocol Group Care Home Prescriptions - Good Practice Guide Date of Publication Review Date August 2015 Responsible Author Francesca Aaen Care Homes Pharmacist on behalf of
Bringing health within your reach. Chronic Medication Designated Service Provider (DSP) Ruby. Emerald. Onyx
 Bringing health within your reach 2009 Chronic Medication Designated Service Provider (DSP) Ruby Emerald Onyx An Overview: The GEMS Chronic Medication DSP Medipost Pharmacy is the Designated Service Provider
Bringing health within your reach 2009 Chronic Medication Designated Service Provider (DSP) Ruby Emerald Onyx An Overview: The GEMS Chronic Medication DSP Medipost Pharmacy is the Designated Service Provider
Note that the following document is copyright, details of which are provided on the next page.
 Please note that the following document was created by the former Australian Council for Safety and Quality in Health Care. The former Council ceased its activities on 31 December 2005 and the Australian
Please note that the following document was created by the former Australian Council for Safety and Quality in Health Care. The former Council ceased its activities on 31 December 2005 and the Australian
AER reference: 52454; D14/54321 ACCC_09/14_865
 Commonwealth of Australia 2014 This work is copyright. In addition to any use permitted under the Copyright Act 1968, all material contained within this work is provided under a Creative Commons Attribution
Commonwealth of Australia 2014 This work is copyright. In addition to any use permitted under the Copyright Act 1968, all material contained within this work is provided under a Creative Commons Attribution
Technical University of Mombasa Faculty of Applied and Health Sciences
 Technical University of Mombasa Faculty of Applied and Health Sciences DEPARTMENT OF MEDICAL SCIENCES DIPLOMA IN PHARMACEUTICAL TECHNOLOGY (DPT 11M) APM 2210 : DRUG SUPPLY & MANAGEMENT II SPECIAL/SUPPLEMENTARY:
Technical University of Mombasa Faculty of Applied and Health Sciences DEPARTMENT OF MEDICAL SCIENCES DIPLOMA IN PHARMACEUTICAL TECHNOLOGY (DPT 11M) APM 2210 : DRUG SUPPLY & MANAGEMENT II SPECIAL/SUPPLEMENTARY:
Statement of Mary L. Hendrickson,PharmD,MBA,RAC Director of Quality & Regulatory Affairs Capital Returns Inc., d/b/a Genco Pharmaceutical Services
 Statement of Mary L. Hendrickson,PharmD,MBA,RAC Director of Quality & Regulatory Affairs Capital Returns Inc., d/b/a Genco Pharmaceutical Services Before The Special Committee on Aging United States Senate
Statement of Mary L. Hendrickson,PharmD,MBA,RAC Director of Quality & Regulatory Affairs Capital Returns Inc., d/b/a Genco Pharmaceutical Services Before The Special Committee on Aging United States Senate
How To Understand Your Role In A Pharmacy
 Element 1 Element 2 Assist with the maintenance of pharmaceutical Issue pharmaceutical 277 278 Element 1 Element 2 Assist with the maintenance of pharmaceutical Issue pharmaceutical Background See background
Element 1 Element 2 Assist with the maintenance of pharmaceutical Issue pharmaceutical 277 278 Element 1 Element 2 Assist with the maintenance of pharmaceutical Issue pharmaceutical Background See background
Understanding Alberta s Drug Schedules
 Understanding Alberta s Drug Schedules Preface In May 2002, the provincial drug schedules to the Pharmaceutical Profession Act were amended. In April 2007, the Alberta Regulation 66/2007 to the Pharmacy
Understanding Alberta s Drug Schedules Preface In May 2002, the provincial drug schedules to the Pharmaceutical Profession Act were amended. In April 2007, the Alberta Regulation 66/2007 to the Pharmacy
DEPARTMENT OF LICENSING AND REGULATORY AFFAIRS DIRECTOR S OFFICE PHARMACY PROGRAM FOR UTILIZATION OF UNUSED PRESCRIPTION DRUGS
 DEPARTMENT OF LICENSING AND REGULATORY AFFAIRS DIRECTOR S OFFICE PHARMACY PROGRAM FOR UTILIZATION OF UNUSED PRESCRIPTION DRUGS (By authority conferred on the director of the department of licensing and
DEPARTMENT OF LICENSING AND REGULATORY AFFAIRS DIRECTOR S OFFICE PHARMACY PROGRAM FOR UTILIZATION OF UNUSED PRESCRIPTION DRUGS (By authority conferred on the director of the department of licensing and
CHAPTER 61-03-02 CONSULTING PHARMACIST REGULATIONS FOR LONG-TERM CARE FACILITIES (SKILLED, INTERMEDIATE, AND BASIC CARE)
 CHAPTER 61-03-02 CONSULTING PHARMACIST REGULATIONS FOR LONG-TERM CARE FACILITIES (SKILLED, INTERMEDIATE, AND BASIC CARE) Section 61-03-02-01 Definitions 61-03-02-02 Absence of Provider or Consulting Pharmacist
CHAPTER 61-03-02 CONSULTING PHARMACIST REGULATIONS FOR LONG-TERM CARE FACILITIES (SKILLED, INTERMEDIATE, AND BASIC CARE) Section 61-03-02-01 Definitions 61-03-02-02 Absence of Provider or Consulting Pharmacist
A guide to applying for your trade mark overseas
 A guide to applying for your trade mark overseas Copyright All content in this publication is provided under a Creative Commons Attribution 4.0 International (CC BY 4.0) licence, with the exception of:
A guide to applying for your trade mark overseas Copyright All content in this publication is provided under a Creative Commons Attribution 4.0 International (CC BY 4.0) licence, with the exception of:
Illinois Emergency Management Agency (IEMA)
 Primary Agency Support Agencies Illinois Emergency Management Agency (IEMA) Governor's Office of Citizens' Assistance (GOCA) Illinois Department of Agriculture (IDA) Illinois Department of Central Management
Primary Agency Support Agencies Illinois Emergency Management Agency (IEMA) Governor's Office of Citizens' Assistance (GOCA) Illinois Department of Agriculture (IDA) Illinois Department of Central Management
QUM and Continuity of Care A Prescribed Medicines Services and Programs
 Community Pharmacy Roadmap Program Development Template Program/Service: Quadrant: 1. Program/Service Description QUM and Continuity of Care A Prescribed Medicines Services and Programs a) Background The
Community Pharmacy Roadmap Program Development Template Program/Service: Quadrant: 1. Program/Service Description QUM and Continuity of Care A Prescribed Medicines Services and Programs a) Background The
JOB DESCRIPTION. Date this JD written/updated : Sep 11 (Updated Organisational Position April 2014)
 JOB DESCRIPTION 1. JOB IDENTIFICATION Job Title: Pharmacy ATO Responsible to: Lead Procurement Pharmacy Technician Department & Base: BGH Pharmacy Date this JD written/updated : Sep 11 (Updated Organisational
JOB DESCRIPTION 1. JOB IDENTIFICATION Job Title: Pharmacy ATO Responsible to: Lead Procurement Pharmacy Technician Department & Base: BGH Pharmacy Date this JD written/updated : Sep 11 (Updated Organisational
Annex 5 WHO good distribution practices for pharmaceutical products
 World Health Organization WHO Technical Report Series, No. 957, 2010 Annex 5 WHO good distribution practices for pharmaceutical products 1. Introduction 2. Scope of the document 3. Glossary 4. General
World Health Organization WHO Technical Report Series, No. 957, 2010 Annex 5 WHO good distribution practices for pharmaceutical products 1. Introduction 2. Scope of the document 3. Glossary 4. General
Assessment of Drug Utilization Patterns in Some Health Insurance Outpatient Clinics in Alexandria 1Ibrahem, Samaa Zenhom; 1Amer, N.; 2Ghoneim, M.
 Assessment of Drug Utilization Patterns in Some Health Insurance Outpatient Clinics in Alexandria 1Ibrahem, Samaa Zenhom; 1Amer, N.; 2Ghoneim, M.; 1Abou El Enein N 1High Institute of Public Health, Egypt
Assessment of Drug Utilization Patterns in Some Health Insurance Outpatient Clinics in Alexandria 1Ibrahem, Samaa Zenhom; 1Amer, N.; 2Ghoneim, M.; 1Abou El Enein N 1High Institute of Public Health, Egypt
Pharmacy Apprenticeships
 Pharmacy Apprenticeships JOB TITLE: BAND: BASE: Pharmacy Technician Pharmacy Department Pharmacy Assistant Pharmacy Department Apprentice Leicester Royal Infirmary Leicester General Hospital Glenfield
Pharmacy Apprenticeships JOB TITLE: BAND: BASE: Pharmacy Technician Pharmacy Department Pharmacy Assistant Pharmacy Department Apprentice Leicester Royal Infirmary Leicester General Hospital Glenfield
How To Amend The Health Insurance Bill Of Insurance In Australia
 Parliament of Australia Department of Parliamentary Services Parliamentary Library Information analysis and advice for the Parliament BILLS DIGEST 18 September 2007, no. 49, 2007 08, ISSN 1328-8091 Health
Parliament of Australia Department of Parliamentary Services Parliamentary Library Information analysis and advice for the Parliament BILLS DIGEST 18 September 2007, no. 49, 2007 08, ISSN 1328-8091 Health
APMI CODE OF PRACTICE ON ADVERTISING OF MEDICINAL PRODUCTS ISSUE 1
 APMI CODE OF PRACTICE ON ADVERTISING OF MEDICINAL PRODUCTS ISSUE 1 August 2010 1. Introduction... 2 2. Scope... 3 3. Definitions... 4 4. General rules of advertising of medicinal products... 6 5. Advertising
APMI CODE OF PRACTICE ON ADVERTISING OF MEDICINAL PRODUCTS ISSUE 1 August 2010 1. Introduction... 2 2. Scope... 3 3. Definitions... 4 4. General rules of advertising of medicinal products... 6 5. Advertising
Medical College of Georgia SOP NUMBER: 03 INVESTIGATIONAL DRUG HANDLING Version Number: 1.0, 1.1 Effective Date: 09/12/06, 08/02/10, 3/2/11
 Effective Date: 09/12/06, 08/02/10, 3/2/11 Title: 1.0 OBJECTIVE: 1.1 This SOP describes the methods and policies for: Handling investigational drug Dispensing investigational drug 1.2. This procedure applies
Effective Date: 09/12/06, 08/02/10, 3/2/11 Title: 1.0 OBJECTIVE: 1.1 This SOP describes the methods and policies for: Handling investigational drug Dispensing investigational drug 1.2. This procedure applies
Value added services of pharmaceutical fullline wholesalers. Contact GIRP for discussing partnerships + 32 2 777 99 77 girp@girp.
 Value added services of pharmaceutical fullline wholesalers Contact GIRP for discussing partnerships + 32 2 777 99 77 [email protected] Selected examples from GIRP s integrated member companies: Please note
Value added services of pharmaceutical fullline wholesalers Contact GIRP for discussing partnerships + 32 2 777 99 77 [email protected] Selected examples from GIRP s integrated member companies: Please note
Ensuring Best Practice in the Notification of Prescription Medical Product Discontinuations
 Ensuring Best Practice in the Notification of Prescription Medical Product Discontinuations TGA and Medicines Australia Conjoint Best Practice Guidelines October 2007 1 Ensuring Best Practice in the Notification
Ensuring Best Practice in the Notification of Prescription Medical Product Discontinuations TGA and Medicines Australia Conjoint Best Practice Guidelines October 2007 1 Ensuring Best Practice in the Notification
INSPECTORS CHECKLIST MONITORING AND INSPECTION VISITS
 INSPECTORS CHECKLIST MONITORING AND INSPECTION VISITS About this document This document is a guide to show what the Inspectorate may look for during an inspection visit. It is intended for use by the GPhC
INSPECTORS CHECKLIST MONITORING AND INSPECTION VISITS About this document This document is a guide to show what the Inspectorate may look for during an inspection visit. It is intended for use by the GPhC
Records Authority. Australian Security Intelligence Organisation
 Records Authority Australian Security Intelligence Organisation This is an accurate reproduction of the authorised records authority content, created for accessibility purposes CONTENTS INTRODUCTION 3
Records Authority Australian Security Intelligence Organisation This is an accurate reproduction of the authorised records authority content, created for accessibility purposes CONTENTS INTRODUCTION 3
Accreditation of a Dispensing and Pharmacy Assistant programme, Boots UK
 Accreditation of a Dispensing and Pharmacy Assistant programme, Boots UK Report of an accreditation event, 19 November 2010 Introduction The General Pharmaceutical Council (GPhC) is the statutory regulator
Accreditation of a Dispensing and Pharmacy Assistant programme, Boots UK Report of an accreditation event, 19 November 2010 Introduction The General Pharmaceutical Council (GPhC) is the statutory regulator
Purpose... 2. What s new?... 2. Role of pharmacists and pharmacy technicians in physician-assisted death... 3
 Table of Contents Purpose... 2 What s new?... 2 Role of pharmacists and pharmacy technicians in physician-assisted death... 3 Complying with ACP s Standards of Practice for Pharmacists and Pharmacy Technicians...
Table of Contents Purpose... 2 What s new?... 2 Role of pharmacists and pharmacy technicians in physician-assisted death... 3 Complying with ACP s Standards of Practice for Pharmacists and Pharmacy Technicians...
Ethiopian Food, Medicine and Healthcare Administration and Control Authority. Medical Equipment Donation Directive
 Ethiopian Food, Medicine and Healthcare Administration and Control Authority Medical Equipment Donation Directive December 2012 Addis Ababa I Contents Contents... II Part One... 1 General... 1 1. Short
Ethiopian Food, Medicine and Healthcare Administration and Control Authority Medical Equipment Donation Directive December 2012 Addis Ababa I Contents Contents... II Part One... 1 General... 1 1. Short
Guide to The Notification System for Exempt Medicinal Products
 Guide to The Notification System for Exempt Medicinal Products AUT-G0090-1 10 JULY 2014 This guide does not purport to be an interpretation of law and/or regulations and is for guidance purposes only.
Guide to The Notification System for Exempt Medicinal Products AUT-G0090-1 10 JULY 2014 This guide does not purport to be an interpretation of law and/or regulations and is for guidance purposes only.
Good Practice Guidance: The administration of medicines in domiciliary care
 Good Practice Guidance: The administration of medicines in domiciliary care Medicines Management Social Care Support Team Reviewed February 2014 This guidance is based on documents that were on CQC s website
Good Practice Guidance: The administration of medicines in domiciliary care Medicines Management Social Care Support Team Reviewed February 2014 This guidance is based on documents that were on CQC s website
16.19.10.11 PUBLIC HEALTH CLINICS: A. CLINIC LICENSURE: (1) All clinics where dangerous drugs are administered, distributed or dispensed shall obtain
 16.19.10.11 PUBLIC HEALTH CLINICS: A. CLINIC LICENSURE: (1) All clinics where dangerous drugs are administered, distributed or dispensed shall obtain a limited drug permit as described in Section 61-11-14
16.19.10.11 PUBLIC HEALTH CLINICS: A. CLINIC LICENSURE: (1) All clinics where dangerous drugs are administered, distributed or dispensed shall obtain a limited drug permit as described in Section 61-11-14
Iamey/AbWEST AFRICAN HEATH ORGANIZATION PROCEDURE GUIDELINE FOR THE MANAGEMENT OF THE REGIONAL ANTIRETROVIRAL DRUGS BUFFER STOCK
 Page 1 of 54 Iamey/AbWEST AFRICAN HEATH ORGANIZATION PROCEDURE GUIDELINE FOR THE MANAGEMENT OF THE REGIONAL ANTIRETROVIRAL DRUGS BUFFER STOCK Addressee: West African Heath Organization (WAHO), Central
Page 1 of 54 Iamey/AbWEST AFRICAN HEATH ORGANIZATION PROCEDURE GUIDELINE FOR THE MANAGEMENT OF THE REGIONAL ANTIRETROVIRAL DRUGS BUFFER STOCK Addressee: West African Heath Organization (WAHO), Central
Development of Medication Barcode System to Ensure Patient Safety in Taiwan
 Development of Medication Barcode System to Ensure Patient Safety in Taiwan Chairman, Taiwan Healthcare Automation Association IPP, Taiwan Society of Health-System Pharmacists Director, Dept. of Pharmacy,
Development of Medication Barcode System to Ensure Patient Safety in Taiwan Chairman, Taiwan Healthcare Automation Association IPP, Taiwan Society of Health-System Pharmacists Director, Dept. of Pharmacy,
Abdul Zahir Siddiqui, 1 Fahima Habibi, 2, Noorulhaq Yousufzai, 3 Lutfullah Ehsaas, 1 M. Zafar Omari, 1 Niranjan Konduri, 4 Terry Green 4
 Pharmaceutical Management Interventions Through a Drug and Therapeutics Committee (DTC) in Kabul, Afghanistan: Initial Experience After Decades of Neglect Abdul Zahir Siddiqui, 1 Fahima Habibi, 2, Noorulhaq
Pharmaceutical Management Interventions Through a Drug and Therapeutics Committee (DTC) in Kabul, Afghanistan: Initial Experience After Decades of Neglect Abdul Zahir Siddiqui, 1 Fahima Habibi, 2, Noorulhaq
DANGEROUS GOODS TRAINING FOR EMPLOYEES
 Advisory Circular AC 92-01(1) AUGUST 2009 DANGEROUS GOODS TRAINING FOR EMPLOYEES CONTENTS 1. References 1 2. Purpose 1 3. Status of this AC 2 4. Accronyms 2 5. Who is Required to Undertake Dangerous Goods
Advisory Circular AC 92-01(1) AUGUST 2009 DANGEROUS GOODS TRAINING FOR EMPLOYEES CONTENTS 1. References 1 2. Purpose 1 3. Status of this AC 2 4. Accronyms 2 5. Who is Required to Undertake Dangerous Goods
Emergency Operations Plan ANNEX R - DONATIONS MANAGEMENT ESF #14 I. MNWALK REQUIREMENTS. Item #: 1, 4, 83, 93, 94, 95, 97
 ANNEX R - DONATIONS MANAGEMENT ESF #14 I. MNWALK REQUIREMENTS Item #: 1, 4, 83, 93, 94, 95, 97 II. PURPOSE The purpose of this annex is to explain the responsibilities for coordinating volunteers, donated
ANNEX R - DONATIONS MANAGEMENT ESF #14 I. MNWALK REQUIREMENTS Item #: 1, 4, 83, 93, 94, 95, 97 II. PURPOSE The purpose of this annex is to explain the responsibilities for coordinating volunteers, donated
Regulatory Impact Statement. Drugs, Poisons and Controlled Substances (Nurse Practitioner and Miscellaneous Amendments 2004)
 Regulatory Impact Statement Drugs, Poisons and Controlled Substances (Nurse Practitioner and Miscellaneous Amendments 2004) Regulatory Impact Statement Drugs, Poisons and Controlled Substances (Nurse Practitioner
Regulatory Impact Statement Drugs, Poisons and Controlled Substances (Nurse Practitioner and Miscellaneous Amendments 2004) Regulatory Impact Statement Drugs, Poisons and Controlled Substances (Nurse Practitioner
Welcome to OptumRx Your Prescription Benefit Program
 Welcome to OptumRx Your Prescription Benefit Program OptumRx offers you more ways to improve your health, while keeping medications more affordable and accessible. Welcome to OptumRx OptumRx manages your
Welcome to OptumRx Your Prescription Benefit Program OptumRx offers you more ways to improve your health, while keeping medications more affordable and accessible. Welcome to OptumRx OptumRx manages your
Contents General Information... 1. General Information
 Contents General Information... 1 Preferred Drug List... 2 Pharmacies... 3 Prescriptions... 4 Generic and Preferred Drugs... 5 Express Scripts Website and Mobile App... 5 Specialty Medicines... 5 Prior
Contents General Information... 1 Preferred Drug List... 2 Pharmacies... 3 Prescriptions... 4 Generic and Preferred Drugs... 5 Express Scripts Website and Mobile App... 5 Specialty Medicines... 5 Prior
EC/64/SC/CRP.4. Report on supply management. Executive Committee of the High Commissioner s Programme. Standing Committee 56 th meeting.
 Executive Committee of the High Commissioner s Programme Standing Committee 56 th meeting Distr. : Restricted 12 February 2013 English Original : English and French Report on supply management Summary
Executive Committee of the High Commissioner s Programme Standing Committee 56 th meeting Distr. : Restricted 12 February 2013 English Original : English and French Report on supply management Summary
MEDICAL DEVICES SECTOR
 MEDICAL DEVICES SECTOR MDS - G1 GUIDANCE FOR MEDICAL DEVICE IMPORTERS Version 2 Our mission is to ensure the safety of food; the safety, quality and efficacy of drugs; and the safety and effectiveness
MEDICAL DEVICES SECTOR MDS - G1 GUIDANCE FOR MEDICAL DEVICE IMPORTERS Version 2 Our mission is to ensure the safety of food; the safety, quality and efficacy of drugs; and the safety and effectiveness
Australian Safety and Quality Framework for Health Care
 Activities for MANAGERS Australian Safety and Quality Framework for Health Care Putting the Framework into action: Getting started Contents Principle: Consumer centred Area for action: 1.1 Develop methods
Activities for MANAGERS Australian Safety and Quality Framework for Health Care Putting the Framework into action: Getting started Contents Principle: Consumer centred Area for action: 1.1 Develop methods
Pharmacy Technician Structured Practical Training Program Logbook
 Pharmacy Technician Structured Practical Training Program Logbook This logbook outlines the activities that pharmacy technician learners are required to complete in order to demonstrate competencies as
Pharmacy Technician Structured Practical Training Program Logbook This logbook outlines the activities that pharmacy technician learners are required to complete in order to demonstrate competencies as
Professional Standards and Guidance for the Sale and Supply of Medicines
 Professional Standards and Guidance for the Sale and Supply of Medicines About this document The Code of Ethics sets out seven principles of ethical practice that you must follow as a pharmacist or pharmacy
Professional Standards and Guidance for the Sale and Supply of Medicines About this document The Code of Ethics sets out seven principles of ethical practice that you must follow as a pharmacist or pharmacy
BAXTER HEALTHCARE PTY LIMITED TERMS AND CONDITIONS OF SALE September 1, 2011
 BAXTER HEALTHCARE PTY LIMITED TERMS AND CONDITIONS OF SALE September 1, 2011 1. OPERATION OF THESE TERMS These Terms apply to Orders made by Customers directly to Baxter and do not apply to Orders for
BAXTER HEALTHCARE PTY LIMITED TERMS AND CONDITIONS OF SALE September 1, 2011 1. OPERATION OF THESE TERMS These Terms apply to Orders made by Customers directly to Baxter and do not apply to Orders for
Introduction 2. 1. The Role of Pharmacy Within a NHS Trust 3. 2. Pharmacy Staff 4. 3. Pharmacy Facilities 5. 4. Pharmacy and Resources 6
 Index Index Section Page Introduction 2 1. The Role of Pharmacy Within a NHS Trust 3 2. Pharmacy Staff 4 3. Pharmacy Facilities 5 4. Pharmacy and Resources 6 5. Prescription Charges 7 6. Communication
Index Index Section Page Introduction 2 1. The Role of Pharmacy Within a NHS Trust 3 2. Pharmacy Staff 4 3. Pharmacy Facilities 5 4. Pharmacy and Resources 6 5. Prescription Charges 7 6. Communication
Rivaroxaban (Xarelto) for preventing blood clots after hip or knee replacement surgery
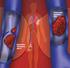 medicineupdate Asking the right questions about new medicines Rivaroxaban (Xarelto) for preventing blood clots after hip or knee replacement surgery This Medicine Update is for people who have been prescribed
medicineupdate Asking the right questions about new medicines Rivaroxaban (Xarelto) for preventing blood clots after hip or knee replacement surgery This Medicine Update is for people who have been prescribed
DRUG REPOSITORY PROGRAM
 OHIO STATE BOARD OF PHARMACY 77 S. High Street, Room 1702; Columbus, OH 43215-6126 Tel: 614-466-4143 Email: [email protected] DRUG REPOSITORY PROGRAM ORC - Ohio Revised Code OAC - Ohio Administrative
OHIO STATE BOARD OF PHARMACY 77 S. High Street, Room 1702; Columbus, OH 43215-6126 Tel: 614-466-4143 Email: [email protected] DRUG REPOSITORY PROGRAM ORC - Ohio Revised Code OAC - Ohio Administrative
BOARD NOTICE 271 OF 2013 THE SOUTH AFRICAN PHARMACY COUNCIL RULES RELATING TO GOOD PHARMACY PRACTICE
 STAATSKOERANT, 20 DESEMBER 2013 No. 37193 3 BOARD NOTICES BOARD NOTICE 271 OF 2013 THE SOUTH AFRICAN PHARMACY COUNCIL RULES RELATING TO GOOD PHARMACY PRACTICE The South African Pharmacy Council herewith
STAATSKOERANT, 20 DESEMBER 2013 No. 37193 3 BOARD NOTICES BOARD NOTICE 271 OF 2013 THE SOUTH AFRICAN PHARMACY COUNCIL RULES RELATING TO GOOD PHARMACY PRACTICE The South African Pharmacy Council herewith
Minimize overspend by gaining visibility of total demand
 Minimize overspend by gaining visibility of total demand Patti Seymour 9th Annual Clinical Trials Supplies and Packaging October 10-12, 2011 BioProcess Technology Consultants www.bptc.com Supply and Demand
Minimize overspend by gaining visibility of total demand Patti Seymour 9th Annual Clinical Trials Supplies and Packaging October 10-12, 2011 BioProcess Technology Consultants www.bptc.com Supply and Demand
Safeguarding your organisation against terrorism financing. A guidance for non-profit organisations
 Safeguarding your organisation against terrorism financing A guidance for non-profit organisations Safeguarding your organisation against terrorism financing A guidance for non-profit organisations ISBN:
Safeguarding your organisation against terrorism financing A guidance for non-profit organisations Safeguarding your organisation against terrorism financing A guidance for non-profit organisations ISBN:
GENERAL INFORMATION. With Express Scripts, you have access to:
 CONTENTS GENERAL INFORMATION... 1 PREFERRED DRUG LIST....2 PHARMACIES... 3 PRESCRIPTIONS... 4 GENERIC AND PREFERRED DRUGS... 5 EXPRESS SCRIPTS WEBSITE AND MOBILE APP... 5 SPECIALTY MEDICATIONS... 6 PRIOR
CONTENTS GENERAL INFORMATION... 1 PREFERRED DRUG LIST....2 PHARMACIES... 3 PRESCRIPTIONS... 4 GENERIC AND PREFERRED DRUGS... 5 EXPRESS SCRIPTS WEBSITE AND MOBILE APP... 5 SPECIALTY MEDICATIONS... 6 PRIOR
Move Management. Enter
 Movers Storers Shippers Enter Exceeding expectations Seafreight European Moving Airfreight Packing Storage Insurance Surveys and Quotes > Contents Contents Moving overseas is a personal service that is
Movers Storers Shippers Enter Exceeding expectations Seafreight European Moving Airfreight Packing Storage Insurance Surveys and Quotes > Contents Contents Moving overseas is a personal service that is
Administrative Policy
 Administrative Policy Number: 411 Effective: 4/7/2011 Supersedes: n/a Page: 1 of 8 Subject: Use of Controlled Substances in Research 1.0. PURPOSE: The purpose of this policy is to set forth procedures
Administrative Policy Number: 411 Effective: 4/7/2011 Supersedes: n/a Page: 1 of 8 Subject: Use of Controlled Substances in Research 1.0. PURPOSE: The purpose of this policy is to set forth procedures
Rehabilitation Guidelines for Employers. Issued under section 41 of the Safety, Rehabilitation and Compensation Act 1988
 Rehabilitation Guidelines for Employers Issued under section 41 of the Safety, Rehabilitation and Compensation Act 1988 Publication details These guidelines are issued under section 41 of the Safety, Rehabilitation
Rehabilitation Guidelines for Employers Issued under section 41 of the Safety, Rehabilitation and Compensation Act 1988 Publication details These guidelines are issued under section 41 of the Safety, Rehabilitation
RMIP Prescription Plan FAQ's
 RMIP Prescription Plan FAQ's A new U.S. Pharmacy Benefit Manager has been selected for January 1, 2015 - CVS/caremark. 1) Why is the RMIP changing to CVS/caremark? The Express Scripts contract ends on
RMIP Prescription Plan FAQ's A new U.S. Pharmacy Benefit Manager has been selected for January 1, 2015 - CVS/caremark. 1) Why is the RMIP changing to CVS/caremark? The Express Scripts contract ends on
Health Professions Act BYLAWS SCHEDULE F. PART 3 Residential Care Facilities and Homes Standards of Practice. Table of Contents
 Health Professions Act BYLAWS SCHEDULE F PART 3 Residential Care Facilities and Homes Standards of Practice Table of Contents 1. Application 2. Definitions 3. Supervision of Pharmacy Services in a Facility
Health Professions Act BYLAWS SCHEDULE F PART 3 Residential Care Facilities and Homes Standards of Practice Table of Contents 1. Application 2. Definitions 3. Supervision of Pharmacy Services in a Facility
JOB TITLE: Trainee Pharmacy Technician
 JOB TITLE: Trainee Pharmacy Technician 1. PURPOSE OF POSITION This position is responsible for helping to provide a customer focused pharmacy service using best practice guidelines to achieve optimal outcomes.
JOB TITLE: Trainee Pharmacy Technician 1. PURPOSE OF POSITION This position is responsible for helping to provide a customer focused pharmacy service using best practice guidelines to achieve optimal outcomes.
POLICY STATEMENT 5.17
 POLICY STATEMENT 5.17 DENTAL RECORDS 1 (Including ADA Guidelines for Dental Records) 1. Introduction 1.1 Dentists have a professional and a legal obligation to maintain clinically relevant, accurate and
POLICY STATEMENT 5.17 DENTAL RECORDS 1 (Including ADA Guidelines for Dental Records) 1. Introduction 1.1 Dentists have a professional and a legal obligation to maintain clinically relevant, accurate and
Draft guidance for registered pharmacies providing internet and distance sale, supply or service provision
 Draft guidance for registered pharmacies providing internet and distance sale, supply or service provision September 2014 1 The General Pharmaceutical Council is the regulator for pharmacists, pharmacy
Draft guidance for registered pharmacies providing internet and distance sale, supply or service provision September 2014 1 The General Pharmaceutical Council is the regulator for pharmacists, pharmacy
Office of Clinical Standards and Quality / Survey & Certification Group
 DEPARTMENT OF HEALTH & HUMAN SERVICES Centers for Medicare & Medicaid Services 7500 Security Boulevard, Mail Stop C2-21-16 Baltimore, Maryland 21244-1850 Office of Clinical Standards and Quality / Survey
DEPARTMENT OF HEALTH & HUMAN SERVICES Centers for Medicare & Medicaid Services 7500 Security Boulevard, Mail Stop C2-21-16 Baltimore, Maryland 21244-1850 Office of Clinical Standards and Quality / Survey
Pharmacy Handbook. Understanding Your Prescription Benefit
 Pharmacy Handbook Understanding Your Prescription Benefit 1 Welcome to Your Prescription Drug Plan! Health Republic Insurance of New York has partnered with US Script to manage your prescription drug benefits.
Pharmacy Handbook Understanding Your Prescription Benefit 1 Welcome to Your Prescription Drug Plan! Health Republic Insurance of New York has partnered with US Script to manage your prescription drug benefits.
SHARPS COLLECTION PROGRAM PLAN FOR THE PRINCE EDWARD ISLAND MEDICAL SHARP STEWARDSHIP PROGRAM
 SHARPS COLLECTION PROGRAM PLAN FOR THE PRINCE EDWARD ISLAND MEDICAL SHARP STEWARDSHIP PROGRAM DECEMBER 2014 EXECUTIVE SUMMARY On June 10, 2014 Prince Edward Island approved the Environmental Protection
SHARPS COLLECTION PROGRAM PLAN FOR THE PRINCE EDWARD ISLAND MEDICAL SHARP STEWARDSHIP PROGRAM DECEMBER 2014 EXECUTIVE SUMMARY On June 10, 2014 Prince Edward Island approved the Environmental Protection
Draft guidance for registered pharmacies preparing unlicensed medicines
 Draft guidance for registered pharmacies preparing unlicensed medicines January 2014 1 The General Pharmaceutical Council is the regulator for pharmacists, pharmacy technicians and registered pharmacies
Draft guidance for registered pharmacies preparing unlicensed medicines January 2014 1 The General Pharmaceutical Council is the regulator for pharmacists, pharmacy technicians and registered pharmacies
International emergency response
 International emergency response Mission: RedR Australia relieves suffering in disasters by selecting, training and providing competent and committed personnel to humanitarian relief agencies worldwide.
International emergency response Mission: RedR Australia relieves suffering in disasters by selecting, training and providing competent and committed personnel to humanitarian relief agencies worldwide.
Marken For more information visit: www.marken.com
 ceocfointerviews.com All rights reserved! Issue: January 18, 2016 The Most Powerful Name in Corporate News Global Supply Chain Service Provider to the Pharmaceutical and Healthcare Industry We are tracking
ceocfointerviews.com All rights reserved! Issue: January 18, 2016 The Most Powerful Name in Corporate News Global Supply Chain Service Provider to the Pharmaceutical and Healthcare Industry We are tracking
the world moves with us
 the art of moving he art of moving the world moves with us Chess Moving Australia is the proud amalgamation of several leading Australian owned moving companies, with industry experience dating back to
the art of moving he art of moving the world moves with us Chess Moving Australia is the proud amalgamation of several leading Australian owned moving companies, with industry experience dating back to
When an earthquake erupts A crisis is called a disaster A number of small and large donors have adopted
 When an earthquake erupts or when a tsunami causes death and destruction, when thousands are displaced by war, or when yet another drought leads to acute famine in a developing country Denmark is ready
When an earthquake erupts or when a tsunami causes death and destruction, when thousands are displaced by war, or when yet another drought leads to acute famine in a developing country Denmark is ready
Take As Directed. Take As Directed
 Take As Directed Take As Directed Peter got some new medication on prescription. When he got home and read the label, it read, TAKE AS DIRECTED. But what did TAKE AS DIRECTED mean? ISBN 0-7306-5646-2 9
Take As Directed Take As Directed Peter got some new medication on prescription. When he got home and read the label, it read, TAKE AS DIRECTED. But what did TAKE AS DIRECTED mean? ISBN 0-7306-5646-2 9
File No.: 20100701. Guidelines for the Administration of certain substances by aged-care workers in residential aged care services
 File No.: 20100701 Guidelines for the Administration of certain substances by aged-care workers in residential aged care services 1 September 2010 Contents 1. Introduction...4 2. Regulation 95EA...5 3.
File No.: 20100701 Guidelines for the Administration of certain substances by aged-care workers in residential aged care services 1 September 2010 Contents 1. Introduction...4 2. Regulation 95EA...5 3.
