Pancreaticoduodenectomy (Pylorus-Preserving Whipple Procedure)
|
|
|
- Cecil Johnson
- 8 years ago
- Views:
Transcription
1 Pancreaticoduodenectomy (Pylorus-Preserving Whipple Procedure) Operative Indications: Pancreaticoduodenectomy, or the Whipple procedure, may be indicated for a variety of benign and malignant diseases. It is most commonly performed for one of the four periampullary adenocarcinomas arising in the head of the pancreas, the ampulla, the distal bile duct, or the duodenum. The procedure is also utilized for less common neoplasms that may arise in the head of the pancreas. These include the cystic neoplasms, both serous and mucinous cystadenomas and mucinous cystadenocarcinomas, intraductal papillary mucinous neoplasms, islet cell tumors (both benign and malignant), and solid and pseudopapillary neoplasms (Hamoudi tumor). Benign ampullary and duodenal adenomas may also occasionally require a pancreaticoduodenectomy for management. There are also a handful of rare tumors, including gastrointestinal stromal tumors and acinar cell tumors, that are treatable by pancreaticoduodenectomy. Some pancreatic surgeons feel that pancreaticoduodenectomy is the procedure of choice for chronic pancreatitis when a dilated duct is not present and a Puestow procedure, therefore, cannot be performed. It is a particularly attractive operation for chronic pancreatitis when the disease is most severe in the head and uncinate process, with less extensive involvement of the body and tail of the gland. Rarely, pancreaticoduodenectomy may be indicated for extensive pancreatic and duodenal trauma, when it is felt that duodenal repair and pancreatic drainage would be inadequate surgical management. In most instances, however, a pancreaticoduodenectomy is performed for a malignant neoplasm arising in the periampullary region. The pylorus-preserving modification of the classic Whipple procedure has become our standard, and it is utilized in over 80% of the pancreaticoduodenectomies done for neoplasms. Operative Technique: The operative procedure can be performed through either a bilateral subcostal or an upper abdominal midline incision. Once the abdomen is entered, a thorough exploration must be
2 Pancreaticoduodenectomy (Pylorus-Preserving Whipple Procedure) 285 Gastrohepatic ligament Liver Celiac axis Spleen Gallbladder Stomach Duodenum 1 carried out to detect any evidence of tumor spread outside the limits of resection. The liver is carefully examined, as are all serosal surfaces for metastatic spread or peritoneal dissemination. Ultrasound can be further used to clear the liver. In addition, lymph node spread outside the boundaries of resection should be determined (1). Involvement of the periportal and celiac axis lymph nodes used to be considered a contraindication for resection. Today, however, these areas can easily be included in the lymphadenectomy accompanying a pancreaticoduodenectomy. The root of the transverse mesocolon also should be examined to determine whether there is direct tumor extension into this area. The root of the transverse mesocolon, if involved, can often be excised along with a segment of the middle colic artery. Generally, the marginal artery of the transverse colon will continue to supply adequate blood to the transverse colon, even in the instance when a segment of middle colic artery has to be excised.
3 286 Atlas of Gastrointestinal Surgery: Pancreas 2 Stomach Kocherized duodenum Tumor Duodenum Head of pancreas Inferior vena cava v. and a. Uncinate process 3 Aorta Pancreas Once tumor dissemination has been ruled out, the duodenum is extensively mobilized. The duodenum, head of the pancreas, and tumor are generally easily elevated off the inferior vena cava and aorta. Direct extension posteriorly into these structures is very unusual. This maneuver is important, however, to be certain the tumor has not extended beyond the uncinate process to involve the superior artery. For this reason, an extensive kocherization should be performed so that one can palpate the superior artery and be reasonably comfortable that there is normal uncinate process adjacent to it (2, 3). If, upon performing this maneuver, one feels tumor extending over to and involving the superior artery, the lesion is not resectable.
4 Pancreaticoduodenectomy (Pylorus-Preserving Whipple Procedure) 287 One next needs to identify the portal vein and to be certain the portal and superior veins are not involved with tumor. The quickest way to accomplish this is to mobilize the gallbladder and divide the common hepatic duct (4). The gallbladder is no longer considered an acceptable means of decompressing the biliary tree into a jejunal loop if the tumor proves to be unresectable. The hepaticojejunostomy is the biliary bypass of choice even if a palliative double bypass is to be carried out. Therefore, the common hepatic duct can be divided early. This allows one to immediately come down upon and identify the portal vein. Gallbladder Common hepatic duct divided Common bile duct 4 Gastroduodnal a. divided 5 The next step usually involves dividing the gastroduodenal artery, passing inferiorly from the common hepatic artery (5). This vessel passes anterior to the portal vein, just at the point where the portal vein passes posterior to the duodenum and neck of the pancreas. Prior to ligating and dividing the gastroduodenal artery, it should be occluded with either a vessel loop or a bulldog clamp, to be certain that a good pulse remains in the hepatic artery. In some instances, when the celiac axis is partially or completely occluded either by atherosclerosis or the arcuate
5 288 Atlas of Gastrointestinal Surgery: Pancreas ligament, the hepatic artery is fed by the gastroduodenal artery through the arcade originating from the superior artery. In this instance, if one divides the gastroduodenal artery, there is risk of liver ischemia and necrosis, and serious lifethreatening morbidity. One therefore has to be certain that a good pulse remains in the hepatic artery before division of the gastroduodenal artery. At this point, one should also check for a replaced right hepatic artery (6) a right hepatic artery originating from the superior artery rather than from the common hepatic artery. In the past, angiography was performed to determine this anomaly prior to surgery. That is no longer felt necessary because, with great accuracy, one can easily identify a replaced right hepatic artery at the time of surgery. This vessel will be found originating from the superior artery (SMA), just after the take-off of the SMA from the aorta. The replaced right hepatic artery then passes up to the liver just lateral to, and posterior to, the biliary tree. With the use of three dimensional CT scans, this anomaly can usually be identified preoperatively. Another anomaly that is less frequent but even more difficult to recognize, is a replaced common hepatic artery off the superior artery. In this instance, a sizable vessel passes anterior to the portal vein just at the point where the portal vein passes behind the first portion of the duodenum and the neck of the pancreas (7). This is in the usual location of the gastroduodenal artery. This also has to be identified and carefully preserved, because division of this vessel would be particularly disastrous, disrupting the blood supply to the liver. Replaced right hepatic a. Replaced common heaptic a. L. hepatic a. R. gastric a. L. gastric a. Splenic a. Hepatic aa. Replaced right hepatic a. Gastroduodenal a. Gastroduodenal a. Replaced common hepatic a. (off superior a.) 6 7
6 Pancreaticoduodenectomy (Pylorus-Preserving Whipple Procedure) 289 Once the common hepatic duct and the gastroduodenal artery have been divided, one can easily dissect the anterior surface of the portal vein off the posterior surface of the neck of the pancreas (8). The insertion of a vein retractor on the neck of the pancreas will allow excellent exposure for this dissection (inset). It is unusual for veins to originate from the anterior surface of the portal vein and enter the posterior neck of the pancreas, so this dissection can often proceed fairly rapidly and bluntly, without risk. Occasionally, however, a coronary vein or a superior pancreaticoduodenal vein (vein of Belcher), will come off the anterior surface of the portal vein, and one has to be aware of these possibilities. Portal v. Neck of pancreas Portal v. v. v. 8 Neck of pancreas v. 9 a. R. gastroepiploic v. Next, the third portion of the duodenum is kocherized extensively. The first structure that is identified crossing anterior to the third portion of the duodenum is the superior vein. This is a much easier route by which to identify the superior vein, rather than going through the lesser sac. We do not enter the lesser sac during a pyloruspreserving pancreaticoduodenectomy. Once the superior vein is identified as it passes anterior to the third portion of the duodenum, its anterior surface is cleaned up under the neck of the pancreas (9). Again, the use of a vein retractor on the neck of the pancreas is helpful in exposing the superior vein for this dissection. The anterior surface of the vein is usually free of significant venous branches, except for the origin of the right gastroepiploic vein.
7 290 Atlas of Gastrointestinal Surgery: Pancreas This venous structure is almost always present, coming off the anterior, or anterior left lateral, aspect of the superior vein just below the neck of the pancreas, and has to be carefully dissected, ligated and divided. Once this large venous structure has been divided, the dissections between the portal vein from above and the superior vein from below can easily be connected (10). After this is accomplished without any evidence of direct involvement by tumor of these major structures, proceeding with the operative procedure is appropriate. Dissection of pancreatic neck off portal v. 10 Portal v. Splenic v. Dissection of pancreas off superior v. v. The first portion of the duodenum is then mobilized and dissected free, off the neck of the pancreas. It is divided with a gastrointestinal anastomosis GIA stapler (11). At times, the dissection of the neck of the pancreas off the portal and superior veins can be enhanced by dividing the duodenum at an earlier stage. Gastroduodenal a. 11 First portion of duodenum divided
8 Pancreaticoduodenectomy (Pylorus-Preserving Whipple Procedure) 291 At this point, the neck of the pancreas can be divided. We generally pass a small Penrose drain underneath the neck of the pancreas, and then, using the electrocautery, divide the gland down to the Penrose drain (12). If the pancreatic duct has been occluded by tumor, and the gland is fibrotic, this division is relatively bloodless. But if the patient has an ampullary tumor, or another neoplasm that has not obstructed the pancreatic duct, and the pancreatic parenchymal is relatively normal, care has to be taken to achieve hemostasis from a variety of bleeding vessels. Duodenum Pancreatic head Tumor Portal v. 12 Pancreas divided v. Once the neck of the pancreas has been divided, a neck margin should be sent for frozen section to be certain that the margin is free of tumor (13). 13
9 292 Atlas of Gastrointestinal Surgery: Pancreas a. First jejunal branch divided (first time) Vein of Belcher v. 14 Hepatic a. Neck of pancreas The superior and portal veins are then dissected off the uncinate process. Again, this is generally done with very little need to ligate and divide substantial vessels. The superior pancreaticoduodenal vein draining into the portal vein, also known as the vein of Belcher, is a fairly constant landmark and needs to be identified, doubly ligated and divided. In addition, more distally on the superior vein, near the inferior border of the uncinate process, the first jejunal branch is a constant landmark, arising from the right lateral border of the superior vein, and coursing around and posterior to the superior artery to the proximal jejunum, to the left of the vessels. This generally has to be ligated and divided (14). A good deal of the dissection of the superior and portal veins off the uncinate process, however, can be done gently, but bluntly. The uncinate process of the pancreas ends flush against the right lateral border of the superior artery. In most instances, the dissection and division of the uncinate process should be flush with the superior artery, cleaning approximately 180 degrees of the artery s circumference (15). There are several sizable arterial branches that have to be identified, ligated and divided. Finally, the first jejunal venous branch, as it courses underneath the superior artery to pass to the proximal jejunum, often a. has to be doubly ligated and divided for a second time (16). v. Uncinate process divided Vein of Belcher v. 15 a. 16 First jejunal branch divided (second time)
10 Pancreaticoduodenectomy (Pylorus-Preserving Whipple Procedure) 293 One then retracts cephalad the transverse colon and identifies the ligament of Treitz and the proximal jejunum. The proximal jejunum at a convenient point distal to the ligament of Treitz, in the middle of a large arcade, is divided with a GIA stapler (17). The mesentery to the proximal jejunum and fourth and third portions of the duodenum are then doubly clamped, divided and ligated with 2-0 silk. Since the third portion of the duodenum has been extensively kocherized and mobilized during the earlier part of the dissection, and since the uncinate process has been completely divided during the earlier dissection, this dissection below the transverse mesocolon to completely mobilize the specimen proceeds easily and quickly. Once the jejunum is divided and the mesentery is clamped, divided and ligated down to the ligament of Treitz, the proximal jejunum is passed underneath the superior vessels over to the right side of the abdomen, and the specimen is removed from the operative field (18). Common hepatic duct Jejunum divided Common hepatic a. Mesentery divided 17 First portion of duodenum Transverse colon 18 Duodenum Transverse colon Pancreatic head Uncinate process Jejunum
11 294 Atlas of Gastrointestinal Surgery: Pancreas Gallbladder Distal biliary tree Head and neck of pancreas The specimen consists of the distal Uncincate portion of the first part of the duodenum, all of the second, third and process fourth portions of the duodenum, and approximately 10 cm of proximal jejunum. In addition, the neck, head Duodenum and all of the uncinate process of the pancreas are included, as are the gallbladder and distal biliary tree (19). 19 Proximal There are a variety of ways to jejunum perform the pancreaticojejunostomy. Resection specimen Many have been used very effectively, with a reasonably low incidence of pancreatic leakage and with low morbidity and mortality. We prefer to invaginate the end of the pancreas into the side of the jejunum. Other pancreatic surgeons prefer to perform a duct to mucosa pancreaticojejunostomy, also in an end-to-side fashion. Whether or not to stent the anastomosis with a small polyethylene tube is still under debate. We will demonstrate the invagination technique with an end-to-side pancreaticojejunostomy (20), which also includes a duct-tojejunal mucosal anastomosis; we will also demonstrate a duct-to-mucosa anastomosis; finally, we will demonstrate teh invagination technique. The end-to-side invagination anastomosis is carried out in two layers: an outer interrupted layer of 3-0 silk and an inner continuous layer of 3-0 synthetic absorbable material. The jejunum is brought up into the lesser sac through a rent in the transverse mesocolon, generally through the bare area of transverse mesocolon that resided over the junction of the second and third portions of the duodenum. The outer row of the posterior layer is placed first. The 3-0 silks are placed through the posterior surface of the pancreas, and then through the jejunum (21). 20 Outer layer of posterior row Jejunum Pancreas Pancreaticojejunostomy 21
12 Pancreaticoduodenectomy (Pylorus-Preserving Whipple Procedure) 295 Inner layer of 22 posterior row 23 Pancreatic duct included in inner layer Inner layer oif anterior row When all sutures have been placed, they are secured. A jejunotomy is then performed. The inner posterior layer of the anastomosis consists of a continuous 3-0 synthetic absorbable suture placed in a locking fashion (22). The pancreatic duct is included in this inner layer. If it is a normal-sized duct, only two or three throws are placed through the duct. If the duct is dilated, however, several throws are placed through and through the dilated pancreatic duct. Thus, this technique combines both the invagination technique as well as a duct-to-mucosa anastomosis. The inner layer of the anterior row of the anastomosis is performed next. This consists of an over-and-over suture passing from above to below, through the capsule of the pancreas and out through the divided parenchyma, and then from inside out on the jejunum. Again, the pancreatic duct is incorporated in several throws of this inner layer of the anterior row (23). When this has been completed, the outer interrupted layer of 3-0 silk sutures is placed such that some of the jejunum is drawn over to cover the anastomosis (24). 24 Outer layer of anterior row
13 296 Atlas of Gastrointestinal Surgery: Pancreas 25 Jejunotomy (small) Jejunum Invaginated pancreas This is performed by passing the silk sutures through the capsule of the pancreas about 1 cm from the anastomosis, then out at the anastomosis, and then through and through the jejunum about a centimeter away from the anastomosis. This anastomosis results in invagination of the end of the pancreas into the side of the jejunum, utilizing two layers, and also incorporates the pancreatic duct (25). This anastomosis can also be performed in an end-to-end fashion, when the pancreatic remnant has a relatively small diameter, and the jejunal diameter is of sufficient size. The anastomosis is performed in an identical fashion as the end-to-side anastomosis. 26 Posterior duct-to-mucosa row For the duct-to-mucosa anastomosis, the outer layer is placed exactly as it is for the invagination technique. The next step creates a jejunotomy the same size and exactly adjacent to the pancreatic duct (26). The ductto-mucosa anastomosis is performed with interrupted 5-0 synthetic absorbable suture material. The posterior row is placed first, the sutures passing from inside out on the duct side and outside in on the mucosal side. Once all posterior row sutures have been placed, they are secured (27). 27
14 Pancreaticoduodenectomy (Pylorus-Preserving Whipple Procedure) 297 Stent placed in pancreatic duct and secured Anterior duct-to-mucosa row 30 Completed anastmosis Next, one can choose to place a stent or not. We demonstrate a stent created from a No. 8 French pediatric feeding tube, 8 cm in length, being secured with one of the 5-0 sutures previously placed in the posterior row (28 29). The anterior row of the duct-tomucosa anastomosis is completed by placing a row of interrupted 5-0 synthetic absorbable sutures from outside in on the duct, and inside out on the jejunal mucosa (30). The outer anterior layer is completed with an interrupted row of 3-0 silk sutures placed in a Lembert fashion (31). Stent 31
15 298 Atlas of Gastrointestinal Surgery: Pancreas 32 Finally, in the unusual situation of a very thin, soft gland in which the pancreatic duct can not be identified, we resort to placing the end of the pancreas into the end of the jejunum for a 5- cm distance and then tacking the jejunum circumferentially to the body of the gland. This is accomplished by mobilizing the end of the pancreas for a 5-cm distance and then placing stay sutures 2.5 cm from the cut end on the superior and inferior borders. These stay sutures are tied, then passed into the end of the jejunum and out the side, 2.5 cm from its end (32). When these are secured, the pancreas is inserted into the jejunum for a 5- cm distance. The end of the jejunum is then sutured to the body of the pancreas circumferentially with a series of interrupted 3-0 silk sutures (33). 33
16 Pancreaticoduodenectomy (Pylorus-Preserving Whipple Procedure) 299 Two to five centimeters distal to the pancreaticojejunostomy, the hepaticojejunostomy is performed (34). This is carried out with a single layer of interrupted 4-0 synthetic absorbable suture material. An appropriately sized enterotomy is performed, and the posterior row is placed with through-and-through sutures passing from inside out on the jejunum and outside in on the hepatic duct (35). When all sutures are placed, they are secured. The anterior layer is then carried out, again first placing all sutures before securing them. They pass from outside in on the jejunum, and inside out on the hepatic duct (36). Hepaticojejunostomy 34 Posterior row Hepatic duct Anterior row 35 36
17 300 Atlas of Gastrointestinal Surgery: Pancreas Hepaticojejunostomy Duodenojejunostomy Jejunum 37 Outer layer of posterior row The final anastomosis is an end-to-side duodenojejunostomy (37). If one leaves only a 2-cm cuff of duodenum on the pylorus, it is not necessary to preserve the right gastric artery, although this is always preferable. The right gastric artery is often small, arises from the hepatic artery, and actually joins the first portion of the duodenum. In most instances, the right gastric artery can be identified and preserved. The duodenojejunostomy is performed with an outer interrupted layer of 3-0 silk and an inner continuous layer of 3-0 synthetic absorbable suture (38). Once the outer layer of the back row of the anastomosis has been placed, these sutures are secured and an enterotomy is made on the side of the jejunum (39). First portion of duodenum Stomach Stent placed in pancreatic duct and secured 38 Enterotomy 39
18 Pancreaticoduodenectomy (Pylorus-Preserving Whipple Procedure) 301 The staple line on the first portion of the duodenum is then excised. The inner layer on the posterior row is a continuous locking suture of 3-0 synthetic absorbable material (40). This is continued onto the anterior row and placed in a Connell fashion (41). The duodenojejunostomy is completed with an outer layer of interrupted 3-0 silk sutures (42). Inner layer of posterior row Inner layer of anterior row Outer layer of anterior row 42
19 302 Atlas of Gastrointestinal Surgery: Pancreas Resection Reconstruction Gallbladder End-to-side hepaticojejunostomy Distal biliary tree End-to-side duodenojejunostomy End-to-side pancreaticojejunostomy Duodenum Proximal jejunum Tumor Pancreas When performing the pylorus-preserving Whipple procedure, a portion of the first part of the duodenum; all of the second, third, and fourth parts of the duodenum; and the proximal jejunum are resected, along with neck, head, and uncinate process of the pancreas. Also removed are the gallbladder and distal biliary tree (43). Although there are many ways to perform the reconstruction following the resection, as just demonstrated, we prefer the end-to-side pancreaticojejunostomy, the end-to-side hepaticojejunostomy, and then an end-to-side duodenojejunostomy (44). Many surgeons continue to prefer the classic Whipple, in which a hemigastrectomy is performed instead of pylorus preservation (45). 45 Alternate: Hemigastrectomy with gastrojejunostomy
20 Pancreaticoduodenectomy (Pylorus-Preserving Whipple Procedure) 303 Portal v. 47 Splenic v. v. Portal v. Veinotomy closed v. Inferior v. 46 Tumor involvement of superior v. 48 Many pancreatic surgeons also feel that, in selected patients with tumor involvement of the portal and/or superior vein (but with otherwise favorable tumors), resection of a segment of these venous structures should be performed, along with venous reconstruction. If the tumor involves just a very small area of the portal vein or superior vein, a Satinsky clamp or DeBakey clamp can partially occlude the venous structure, and a small ellipse of the vein that is involved with the tumor can be excised (46). If this ellipse is small, the venotomy can be closed with a continuous 5-0 synthetic non-absorbable suture (47). If the ellipse is larger and its direct closure would result in narrowing of the superior vein, a vein patch can be utilized to maintain diameter (48). Patch
21 304 Atlas of Gastrointestinal Surgery: Pancreas 49 Portal v. Tumor v. In some patients, enough of the vein is involved so that tangential resection is not feasible. In these instances, a segment of portal and/or superior vein generally has to be excised (49). In these instances, if the segment includes the splenic vein, the splenic vein can be ligated and divided with impunity. After the segment of superior vein and portal vein is excised, if it is 3 cm or less in length, a direct end-to-end anastomosis can be performed (50) If the segment is longer than 3 to 4 cm, an interposition vein graft of either saphenous vein or jugular vein is preferable (51). It is not necessary to reimplant the splenic vein. It has been controversial as to whether or not a retroperitoneal lymphadenectomy accompanying a pancreaticoduodenectomy is of benefit in prolonging survival after a resection for cancer of the pancreas. Some pancreatic surgeons have felt an extensive retroperitoneal dissection, as well as dissection of the nodes surrounding the celiac axis and porta hepatis, has resulted in prolonged survival. When we perform a retroperitoneal lymphadenectomy, we first perform a classic Whipple including a hemigastrectomy so that the prepyloric and pyloric lymph nodes are included. The retroperitoneal dissection then starts at the medial aspect of the right kidney hilum and proceeds laterally to the left side of the aorta. It Portal v. Portal v. End-to-end v. anastomosis Interposition vein graft v.
22 Pancreaticoduodenectomy (Pylorus-Preserving Whipple Procedure) 305 Alternative: Retroperitoneal lymphdenctomy v. Hepatic duct (clamped) Inferior vena cava Aorta Stomach a. Right kidney Inferior a. 52 extends superiorly from the portal vein to inferiorly at the takeoff of the inferior artery (52). In our experience, when retroperitoneal nodes are positive, they generally are in the caval-aortic groove. This dissection also includes the tissues surrounding 180 degrees of the circumference of the superior artery. In addition, the celiac axis lymph nodes are dissected (53), and the dissection can pass laterally along the hepatic artery into the porta hepatis. It remains controversial as to whether or not such a retroperitoneal lymphadenectomy prolongs survival. In the largest single-institution, prospective randomized study carried out, radical retroperitoneal lymphadenectomy was of no survival benefit. Retroperitoneal nodes Celiac axis nodes 53
The Whipple Procedure. Sally Hodges, Ph.D.(c) Given the length and difficulty of the procedure, regardless of the diagnosis, certain
 The Whipple Procedure Sally Hodges, Ph.D.(c) Preoperative procedures Given the length and difficulty of the procedure, regardless of the diagnosis, certain assurances must occur prior to offering a patient
The Whipple Procedure Sally Hodges, Ph.D.(c) Preoperative procedures Given the length and difficulty of the procedure, regardless of the diagnosis, certain assurances must occur prior to offering a patient
PANCREATIC AND PERIAMPULLARY TUMORS: PANCREATICODUODENECTOMY. Dr. Shailesh V. Shrikhande
 PANCREATIC AND PERIAMPULLARY TUMORS: PANCREATICODUODENECTOMY Dr. Shailesh V. Shrikhande Associate Professor & Consultant Surgeon GI and HPB Surgical Oncology Tata Memorial Hospital, Mumbai INDIA HELICAL
PANCREATIC AND PERIAMPULLARY TUMORS: PANCREATICODUODENECTOMY Dr. Shailesh V. Shrikhande Associate Professor & Consultant Surgeon GI and HPB Surgical Oncology Tata Memorial Hospital, Mumbai INDIA HELICAL
Cancer of the Exocrine Pancreas, Ampulla of Vater and Distal Common Bile Duct Proforma
 Cancer of the Exocrine, Ampulla of Vater and Distal Coon Bile Duct Proforma Mandatory questions (i.e. protocol standards) are in bold (e.g. S1.03). Family name Given name(s) Sex Male Female Intersex/indeterminate
Cancer of the Exocrine, Ampulla of Vater and Distal Coon Bile Duct Proforma Mandatory questions (i.e. protocol standards) are in bold (e.g. S1.03). Family name Given name(s) Sex Male Female Intersex/indeterminate
Pancreaticoduodenectomy in Patients with a History of Roux-en Y Gastric Bypass Surgery
 ORIGINAL ARTICLE Pancreaticoduodenectomy in Patients with a History of Roux-en Y Gastric Bypass Surgery Mehrdad Nikfarjam 1,2, Kevin F Staveley-O Carroll 2, Eric T Kimchi 2, Jeffrey M Hardacre 1 1 Department
ORIGINAL ARTICLE Pancreaticoduodenectomy in Patients with a History of Roux-en Y Gastric Bypass Surgery Mehrdad Nikfarjam 1,2, Kevin F Staveley-O Carroll 2, Eric T Kimchi 2, Jeffrey M Hardacre 1 1 Department
Extended Radical Whipple Resection for Cancer of the Pancreatic Head: Operative Procedure and Results
 How I Do It Dig Surg 1998;15:299 307 Received: February 17, 1997 Accepted: March 20, 1997 T. Imaizumi F. Hanyu N. Harada T. Hatori A. Fukuda Department of Gastroenterological Surgery, Tokyo Women s Medical
How I Do It Dig Surg 1998;15:299 307 Received: February 17, 1997 Accepted: March 20, 1997 T. Imaizumi F. Hanyu N. Harada T. Hatori A. Fukuda Department of Gastroenterological Surgery, Tokyo Women s Medical
David R. DeHaas, Jr. MD Sacred Heart Medical Center at RiverBend
 David R. DeHaas, Jr. MD Sacred Heart Medical Center at RiverBend 1. To review the surgical history of the Whipple procedure for periampullary tumors. 2. To review the differential diagnosis for patients
David R. DeHaas, Jr. MD Sacred Heart Medical Center at RiverBend 1. To review the surgical history of the Whipple procedure for periampullary tumors. 2. To review the differential diagnosis for patients
To Whipple or Not to Whipple, that is the Question: Evaluating the Resectability of Pancreatic Adenocarcinoma
 August 2009 To Whipple or Not to Whipple, that is the Question: Evaluating the Resectability of Pancreatic Adenocarcinoma Christina Ramirez, Harvard Medical School Year III Gillian Lieberman, MD Agenda
August 2009 To Whipple or Not to Whipple, that is the Question: Evaluating the Resectability of Pancreatic Adenocarcinoma Christina Ramirez, Harvard Medical School Year III Gillian Lieberman, MD Agenda
WHIPPLE PROCEDURE FOR PANCREATIC CANCER UNIVERSITY OF MARYLAND MEDICAL CENTER BALTIMORE, MD January 7, 2008
 WHIPPLE PROCEDURE FOR PANCREATIC CANCER UNIVERSITY OF MARYLAND MEDICAL CENTER BALTIMORE, MD January 7, 2008 00:00:11 ANNOUNCER: This year, an estimated 37,000 new cases of pancreatic cancer will be diagnosed
WHIPPLE PROCEDURE FOR PANCREATIC CANCER UNIVERSITY OF MARYLAND MEDICAL CENTER BALTIMORE, MD January 7, 2008 00:00:11 ANNOUNCER: This year, an estimated 37,000 new cases of pancreatic cancer will be diagnosed
Cancer Surgery Volume Study: ICD-9 and CPT Codes
 This paper contains the ICD-9 diagnostic and procedure codes and the CPT procedure codes used by researchers for a project of the California HealthCare Foundation (CHCF) and the California Office of Statewide
This paper contains the ICD-9 diagnostic and procedure codes and the CPT procedure codes used by researchers for a project of the California HealthCare Foundation (CHCF) and the California Office of Statewide
Bile Leaks After Laparoscopic Cholecystectomy. Kings County Hospital Center Eliana A. Soto, MD
 Bile Leaks After Laparoscopic Cholecystectomy Kings County Hospital Center Eliana A. Soto, MD Biliary Injuries during Cholecystectomy In the 1990s, high rate of biliary injury was due in part to learning
Bile Leaks After Laparoscopic Cholecystectomy Kings County Hospital Center Eliana A. Soto, MD Biliary Injuries during Cholecystectomy In the 1990s, high rate of biliary injury was due in part to learning
The Whipple Operation for Pancreatic Cancer: Optimism vs. Reality. Franklin Wright UCHSC Department of Surgery Grand Rounds September 11, 2006
 The Whipple Operation for Pancreatic Cancer: Optimism vs. Reality Franklin Wright UCHSC Department of Surgery Grand Rounds September 11, 2006 Overview Pancreatic ductal adenocarcinoma Pancreaticoduodenectomy
The Whipple Operation for Pancreatic Cancer: Optimism vs. Reality Franklin Wright UCHSC Department of Surgery Grand Rounds September 11, 2006 Overview Pancreatic ductal adenocarcinoma Pancreaticoduodenectomy
ATLAS FOR THE DELINEATION OF THE POSTOPERATIVE TREATMENT OF PANCREATIC CANCER
 CONSENSUS PANEL CONTOURING ATLAS FOR THE DELINEATION OF THE CLINICAL TARGET VOLUME IN THE POSTOPERATIVE TREATMENT OF PANCREATIC CANCER Collaborators Ross A. Abrams, M.D. 1, William F. Regine, M.D. 2, Karyn
CONSENSUS PANEL CONTOURING ATLAS FOR THE DELINEATION OF THE CLINICAL TARGET VOLUME IN THE POSTOPERATIVE TREATMENT OF PANCREATIC CANCER Collaborators Ross A. Abrams, M.D. 1, William F. Regine, M.D. 2, Karyn
The digestive system. Medicine and technology. Normal structure and function Diagnostic methods Example diseases and therapies
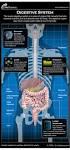 The digestive system Medicine and technology Normal structure and function Diagnostic methods Example diseases and therapies The digestive system An overview (1) Oesophagus Liver (hepar) Biliary system
The digestive system Medicine and technology Normal structure and function Diagnostic methods Example diseases and therapies The digestive system An overview (1) Oesophagus Liver (hepar) Biliary system
Robotic Whipple Procedure for Pancreatic Cancer: The Moffitt Cancer Center Pathway
 The robotic Whipple procedure is a minimally invasive option for resectable pancreatic cancer and other tumors of the pancreas head, distal common bile duct, and ampulla. Sofía Cáceres Nazario. Zinnia,
The robotic Whipple procedure is a minimally invasive option for resectable pancreatic cancer and other tumors of the pancreas head, distal common bile duct, and ampulla. Sofía Cáceres Nazario. Zinnia,
ADVANCES IN SURGERY. Robotic-Assisted Major Pancreatic Resection. H.J. Zeh III, MD*, David L. Bartlett, MD, A. James Moser, MD*
 Advances in Surgery 45 (2011) 323 340 ADVANCES IN SURGERY Robotic-Assisted Major Pancreatic Resection H.J. Zeh III, MD*, David L. Bartlett, MD, A. James Moser, MD* Division of Surgical Oncology, Department
Advances in Surgery 45 (2011) 323 340 ADVANCES IN SURGERY Robotic-Assisted Major Pancreatic Resection H.J. Zeh III, MD*, David L. Bartlett, MD, A. James Moser, MD* Division of Surgical Oncology, Department
restricted to certain centers and certain patients, preferably in some sort of experimental trial format.
 Managing Pancreatic Cancer, Part 4: Pancreatic Cancer Surgery, Complications, & the Importance of Surgical Volume Dr. Matthew Katz, Surgeon, MD Anderson Cancer Center, Houston, TX I m going to talk a little
Managing Pancreatic Cancer, Part 4: Pancreatic Cancer Surgery, Complications, & the Importance of Surgical Volume Dr. Matthew Katz, Surgeon, MD Anderson Cancer Center, Houston, TX I m going to talk a little
Clinical Anatomy of the Biliary Apparatus: Relations & Variations
 Clinical Anatomy of the Biliary Apparatus: Relations & Variations Handout download: http://www.oucom.ohiou.edu/dbms-witmer/gs-rpac.htm 24 January 2012 Lawrence M. Witmer, PhD Professor of Anatomy Department
Clinical Anatomy of the Biliary Apparatus: Relations & Variations Handout download: http://www.oucom.ohiou.edu/dbms-witmer/gs-rpac.htm 24 January 2012 Lawrence M. Witmer, PhD Professor of Anatomy Department
PATIENT INFORMATION ABOUT ADJUVANT THERAPY AFTER THE WHIPPLE OPERATION FOR ADENOCARCINOMA ( CANCER ) OF THE PANCREAS AND RELATED SITES.
 PATIENT INFORMATION ABOUT ADJUVANT THERAPY AFTER THE WHIPPLE OPERATION FOR ADENOCARCINOMA ( CANCER ) OF THE PANCREAS AND RELATED SITES. Radiation Oncology Sidney Kimmel Cancer Center at Johns Hopkins Last
PATIENT INFORMATION ABOUT ADJUVANT THERAPY AFTER THE WHIPPLE OPERATION FOR ADENOCARCINOMA ( CANCER ) OF THE PANCREAS AND RELATED SITES. Radiation Oncology Sidney Kimmel Cancer Center at Johns Hopkins Last
Pancreatic Cancer. The Killer that must be discovered early. Dr Alfred Kow Wei Chieh
 Pancreatic Cancer The Killer that must be discovered early 27 th June 2015 Dr Alfred Kow Wei Chieh Consultant Department of Surgery Division of HPB Surgery & Liver Transplantation & Assistant Dean (Education)
Pancreatic Cancer The Killer that must be discovered early 27 th June 2015 Dr Alfred Kow Wei Chieh Consultant Department of Surgery Division of HPB Surgery & Liver Transplantation & Assistant Dean (Education)
Laparoscopic extended right hemicolectomy for transverse colon
 Laparoscopic extended right hemicolectomy for transverse colon cancer Elsa B. Valsdottir, MD Department of General Surgery University Hospital of Iceland Hringbraut 101 Reykjavik Iceland Phone +354 543
Laparoscopic extended right hemicolectomy for transverse colon cancer Elsa B. Valsdottir, MD Department of General Surgery University Hospital of Iceland Hringbraut 101 Reykjavik Iceland Phone +354 543
Minimally Invasive Spine Surgery
 Chapter 1 Minimally Invasive Spine Surgery 1 H.M. Mayer Primum non nocere First do no harm In the long history of surgery it always has been a basic principle to restrict the iatrogenic trauma done to
Chapter 1 Minimally Invasive Spine Surgery 1 H.M. Mayer Primum non nocere First do no harm In the long history of surgery it always has been a basic principle to restrict the iatrogenic trauma done to
Understanding. Pancreatic Cancer
 Understanding Pancreatic Cancer Understanding Pancreatic Cancer The Pancreas The pancreas is an organ that is about 6 inches long. It s located deep in your belly between your stomach and backbone. Your
Understanding Pancreatic Cancer Understanding Pancreatic Cancer The Pancreas The pancreas is an organ that is about 6 inches long. It s located deep in your belly between your stomach and backbone. Your
How to treat early gastric cancer. Surgery
 How to treat early gastric cancer Surgery Mark I. van Berge Henegouwen Department of Surgery, AMC, Amsterdam Director upper GI surgical unit Academic Medical Center Upper GI surgery at AMC 100 oesophagectomies
How to treat early gastric cancer Surgery Mark I. van Berge Henegouwen Department of Surgery, AMC, Amsterdam Director upper GI surgical unit Academic Medical Center Upper GI surgery at AMC 100 oesophagectomies
Preoperative drainage is always indicated in malignant CBD strictures PRO. Horst Neuhaus Evangelisches Krankenhaus Düsseldorf, Germany
 Preoperative drainage is always indicated in malignant CBD strictures PRO Horst Neuhaus Evangelisches Krankenhaus Düsseldorf, Germany Background Jaundice is associated with high perioperative morbidity
Preoperative drainage is always indicated in malignant CBD strictures PRO Horst Neuhaus Evangelisches Krankenhaus Düsseldorf, Germany Background Jaundice is associated with high perioperative morbidity
A Patient s Guide to. Pancreatic Cysts. University of Michigan Comprehensive Cancer Center
 A Patient s Guide to Pancreatic Cysts University of Michigan Comprehensive Cancer Center Staff of the Comprehensive Cancer Center s Multidisciplinary Pancreatic Cancer Program provided information for
A Patient s Guide to Pancreatic Cysts University of Michigan Comprehensive Cancer Center Staff of the Comprehensive Cancer Center s Multidisciplinary Pancreatic Cancer Program provided information for
Oxford University Hospitals
 Oxford University Hospitals NHS Trust Department of Hepatobiliary and Pancreatic Surgery About Pancreatic Surgery A guide for patients and relatives Introduction This booklet has been written to provide
Oxford University Hospitals NHS Trust Department of Hepatobiliary and Pancreatic Surgery About Pancreatic Surgery A guide for patients and relatives Introduction This booklet has been written to provide
Laparoscopic Sleeve gastrectomy
 Restrictive procedure Laparoscopic Sleeve gastrectomy Dr. R. Peterli Professional Education 1 2 Introduction Gastric sleeve resection is the restrictive part of the biliopancreatic diversion duodenal switch,
Restrictive procedure Laparoscopic Sleeve gastrectomy Dr. R. Peterli Professional Education 1 2 Introduction Gastric sleeve resection is the restrictive part of the biliopancreatic diversion duodenal switch,
Lenox Hill Hospital Department of Surgery General Surgery Goals and Objectives
 Lenox Hill Hospital Department of Surgery General Surgery Goals and Objectives Medical Knowledge and Patient Care: Residents must demonstrate knowledge and application of the pathophysiology and epidemiology
Lenox Hill Hospital Department of Surgery General Surgery Goals and Objectives Medical Knowledge and Patient Care: Residents must demonstrate knowledge and application of the pathophysiology and epidemiology
Vascular resection during pancreaticoduodenectomy. Ryan Turley, MD Research Fellow, Duke University
 Vascular resection during pancreaticoduodenectomy. Ryan Turley, MD Research Fellow, Duke University Vascular Resection during PD Outline Review of pancreatic adenocarcinoma Role of PD in treatment of pancreatic
Vascular resection during pancreaticoduodenectomy. Ryan Turley, MD Research Fellow, Duke University Vascular Resection during PD Outline Review of pancreatic adenocarcinoma Role of PD in treatment of pancreatic
WHAT S WRONG WITH MY GALL BLADDER? GALL BLADDER POLYPS
 WHAT S WRONG WITH MY GALL BLADDER? GALL BLADDER POLYPS This is a patient information booklet providing specific practical information about gall bladder polyps in brief. Its aim is to provide the patient
WHAT S WRONG WITH MY GALL BLADDER? GALL BLADDER POLYPS This is a patient information booklet providing specific practical information about gall bladder polyps in brief. Its aim is to provide the patient
Facing Pancreatic Surgery? Learn about minimally invasive da Vinci Surgery
 Facing Pancreatic Surgery? Learn about minimally invasive da Vinci Surgery The Condition: Pancreatitis/Pancreatic Cancer The pancreas is an organ that produces enzymes and hormones to help your body digest
Facing Pancreatic Surgery? Learn about minimally invasive da Vinci Surgery The Condition: Pancreatitis/Pancreatic Cancer The pancreas is an organ that produces enzymes and hormones to help your body digest
LAPAROSCOPIC GASTRIC BYPASS AND SLEEVE GASTRECTOMY FLAGLER HOSPITAL AND HOLY CROSS HOSPITAL ST. AUGUSTINE, FL AND FT. LAUDERDALE, FL January 15, 2008
 LAPAROSCOPIC GASTRIC BYPASS AND SLEEVE GASTRECTOMY FLAGLER HOSPITAL AND HOLY CROSS HOSPITAL ST. AUGUSTINE, FL AND FT. LAUDERDALE, FL January 15, 2008 00:00:10 ANNOUNCER: This event is being sponsored by
LAPAROSCOPIC GASTRIC BYPASS AND SLEEVE GASTRECTOMY FLAGLER HOSPITAL AND HOLY CROSS HOSPITAL ST. AUGUSTINE, FL AND FT. LAUDERDALE, FL January 15, 2008 00:00:10 ANNOUNCER: This event is being sponsored by
Luis D. Carcorze Soto, MD PGY-3
 Luis D. Carcorze Soto, MD PGY-3 Peritoneal Surface Malignancies Peritoneum Patient Selection Operative Technique HIPEC EPIC Primary: Primary Peritoneal Carcinoma Malignant Peritoneal Mesothelioma Metastatic:
Luis D. Carcorze Soto, MD PGY-3 Peritoneal Surface Malignancies Peritoneum Patient Selection Operative Technique HIPEC EPIC Primary: Primary Peritoneal Carcinoma Malignant Peritoneal Mesothelioma Metastatic:
Pancreatic Cancer Basics. Meghan McGurk, PA-C, MMSc Physician Assistant Department of Medical Oncology Yale Cancer Center
 Pancreatic Cancer Basics Meghan McGurk, PA-C, MMSc Physician Assistant Department of Medical Oncology Yale Cancer Center Case Study Mr.T is a 36 yo male who presented in the fall of 2003, at the age of
Pancreatic Cancer Basics Meghan McGurk, PA-C, MMSc Physician Assistant Department of Medical Oncology Yale Cancer Center Case Study Mr.T is a 36 yo male who presented in the fall of 2003, at the age of
Digestive System AKA. GI System. Overview. GI Process Process Includes. G-I Tract Alimentary Canal
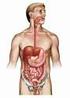 Digestive System AKA G-I Tract Alimentary Canal Overview GI System Consists of Mouth, pharynx, esophagus, stomach, small intestine, large intestine, anus About 30 in length Accessory Organs Teeth, tongue,
Digestive System AKA G-I Tract Alimentary Canal Overview GI System Consists of Mouth, pharynx, esophagus, stomach, small intestine, large intestine, anus About 30 in length Accessory Organs Teeth, tongue,
Surgery and cancer of the pancreas
 Surgery and cancer of the pancreas This information is an extract from the booklet Understanding cancer of the pancreas. You may find the full booklet helpful. We can send you a free copy see page 8. Introduction
Surgery and cancer of the pancreas This information is an extract from the booklet Understanding cancer of the pancreas. You may find the full booklet helpful. We can send you a free copy see page 8. Introduction
Functional Human Morphology (2040) & Functional Anatomy of the Head, Neck and Trunk (2130)
 Functional Human Morphology (2040) & Functional Anatomy of the Head, Neck and Trunk (2130) Gastrointestinal & Urogenital Systems Recommended Text: TEXTBOOK OF ANATOMY: ROGERS Published by Churchill Livingstone
Functional Human Morphology (2040) & Functional Anatomy of the Head, Neck and Trunk (2130) Gastrointestinal & Urogenital Systems Recommended Text: TEXTBOOK OF ANATOMY: ROGERS Published by Churchill Livingstone
Journal Name: International Journal of Hepatobiliary and Pancreatic Diseases (IJHPD)
 ACCEPTED MAN U SCRIPT EARLY VIEW ARTICLE Early View Article: Online published version of an accepted article before inclusion in an issue of International Journal of Hepatobiliary and Pancreatic Diseases
ACCEPTED MAN U SCRIPT EARLY VIEW ARTICLE Early View Article: Online published version of an accepted article before inclusion in an issue of International Journal of Hepatobiliary and Pancreatic Diseases
Sonographic Demonstration of Couinaud s Liver Segments
 PICTORIL ESSY Sonographic Demonstration of Couinaud s Liver Segments Dean Smith, MD, FRCPC, Donal Downey, M, Ch, FRCPC, lison Spouge, MD, FRCPC, Sue Soney, RT, RDMS, RCMS The segmental localization of
PICTORIL ESSY Sonographic Demonstration of Couinaud s Liver Segments Dean Smith, MD, FRCPC, Donal Downey, M, Ch, FRCPC, lison Spouge, MD, FRCPC, Sue Soney, RT, RDMS, RCMS The segmental localization of
ANTERIOR LUMBAR INTERBODY FUSION (ALIF) Basic Anatomical Landmarks: Anterior Lumbar Spine
 (ALIF) Anterior In human anatomy, referring to the front surface of the body or the position of one structure relative to another Lumbar Relating to the loins or the section of the back and sides between
(ALIF) Anterior In human anatomy, referring to the front surface of the body or the position of one structure relative to another Lumbar Relating to the loins or the section of the back and sides between
SURGICAL TREATMENT OF PANCREATIC CANCER: FROM MAJOR RESECTION TO CELLULAR SURGERY
 SOCIETAD CATALANA DE CIRURGIA Barcelona, October 2013 SURGICAL TREATMENT OF PANCREATIC CANCER: FROM MAJOR RESECTION TO CELLULAR SURGERY Prof. Claudio Bassi FACS,FRCS,FEBS Verona, Italy Personal convinctions
SOCIETAD CATALANA DE CIRURGIA Barcelona, October 2013 SURGICAL TREATMENT OF PANCREATIC CANCER: FROM MAJOR RESECTION TO CELLULAR SURGERY Prof. Claudio Bassi FACS,FRCS,FEBS Verona, Italy Personal convinctions
Treatment of Hepatic Neoplasm
 I. Policy University Health Alliance (UHA) will reimburse for treatment of hepatic neoplasm outside of systemic chemotherapy alone when determined to be medically necessary and within the medical criteria
I. Policy University Health Alliance (UHA) will reimburse for treatment of hepatic neoplasm outside of systemic chemotherapy alone when determined to be medically necessary and within the medical criteria
PRACTICE GUIDELINE TITLE: INTRAVENOUS LINE INSERTION: PERIPHERAL AND CENTRAL
 PRACTICE GUIDELINE Effective Date: 9-17-04 Manual Reference: Deaconess Trauma Services TITLE: INTRAVENOUS LINE INSERTION: PERIPHERAL AND CENTRAL PURPOSE: To outline the indications and options for intravenous
PRACTICE GUIDELINE Effective Date: 9-17-04 Manual Reference: Deaconess Trauma Services TITLE: INTRAVENOUS LINE INSERTION: PERIPHERAL AND CENTRAL PURPOSE: To outline the indications and options for intravenous
Integumentary System Individual Exercises
 Integumentary System Individual Exercises 1. A physician performs an incision and drainage of a subcutaneous abscess in his office for a particularly uncooperative established patient. How should this
Integumentary System Individual Exercises 1. A physician performs an incision and drainage of a subcutaneous abscess in his office for a particularly uncooperative established patient. How should this
Cystic Neoplasms of the Pancreas: A multidisciplinary approach to the prevention and early detection of invasive pancreatic cancer.
 This lecture is drawn from the continuing medical education program Finding Hope: Prevention, Early Detection and Treatment of Pancreatic Cancer, Nov, 2011. Robert P. Jury, MD Cystic Neoplasms of the Pancreas:
This lecture is drawn from the continuing medical education program Finding Hope: Prevention, Early Detection and Treatment of Pancreatic Cancer, Nov, 2011. Robert P. Jury, MD Cystic Neoplasms of the Pancreas:
Divisions of Digestive System. Organs of the Alimentary Canal. Anatomy of the Digestive System: Organs of the Alimentary Canal. CHAPTER 14 p.
 Divisions of Digestive System Anatomy of the Digestive System: Organs of the Alimentary Canal CHAPTER 14 p. 412-423 1. Alimentary Canal or Gastrointestinal Tract (GI)-digests and absorbs food coiled hollow
Divisions of Digestive System Anatomy of the Digestive System: Organs of the Alimentary Canal CHAPTER 14 p. 412-423 1. Alimentary Canal or Gastrointestinal Tract (GI)-digests and absorbs food coiled hollow
Aehlert: Paramedic Practice Today PowerPoint Lecture Notes Chapter 50: Abdominal Trauma
 Aehlert: Paramedic Practice Today PowerPoint Lecture Notes Chapter 50: Abdominal Trauma Chapter 50 Abdominal Trauma 1 Describe the epidemiology, including morbidity, mortality rates, and prevention strategies,
Aehlert: Paramedic Practice Today PowerPoint Lecture Notes Chapter 50: Abdominal Trauma Chapter 50 Abdominal Trauma 1 Describe the epidemiology, including morbidity, mortality rates, and prevention strategies,
BERGEN COMMUNITY COLLEGE DIAGNOSTIC MEDICAL SONOGRAPHY PROGRAM Division of Health Professions DMS 213 SYLLABUS
 BERGEN COMMUNITY COLLEGE DIAGNOSTIC MEDICAL SONOGRAPHY PROGRAM Division of Health Professions DMS 213 SYLLABUS Course Title: DMS 213 - Abdominal Sonography 2 2 lec. 3 lab. 3 credits (5 hours) Required
BERGEN COMMUNITY COLLEGE DIAGNOSTIC MEDICAL SONOGRAPHY PROGRAM Division of Health Professions DMS 213 SYLLABUS Course Title: DMS 213 - Abdominal Sonography 2 2 lec. 3 lab. 3 credits (5 hours) Required
Cracking CPT Codes: An Interactive Discussion Presented by Tom Loughrey, CCS-P. Jumping Right In!
 Cracking CPT Codes: An Interactive Discussion Presented by Tom Loughrey, CCS-P Jumping Right In! Code the following: 38 year old female for right breast biopsy with percutaneous needle core using image
Cracking CPT Codes: An Interactive Discussion Presented by Tom Loughrey, CCS-P Jumping Right In! Code the following: 38 year old female for right breast biopsy with percutaneous needle core using image
Acute Abdominal Pain following Bariatric Surgery. Disclosure. Objectives 8/17/2015. I have nothing to disclose
 Acute Abdominal Pain following Bariatric Surgery Kathy J. Morris, DNP, APRN, FNP C, FAANP University of Nebraska Medical Center College of Nursing Disclosure I have nothing to disclose Objectives Pathophysiology
Acute Abdominal Pain following Bariatric Surgery Kathy J. Morris, DNP, APRN, FNP C, FAANP University of Nebraska Medical Center College of Nursing Disclosure I have nothing to disclose Objectives Pathophysiology
Basic Laparoscopy and Lap. Suturing and Stapling course Course Contents
 Online Courses on Laparoscopic GI Surgery for GISurgery.info Lap Skills course Harshad Soni 1. Basic Laparoscopy and Lap. Suturing and Stapling course H. Soni 2. Laparoscopic UGI Surgery Course J Mistry
Online Courses on Laparoscopic GI Surgery for GISurgery.info Lap Skills course Harshad Soni 1. Basic Laparoscopy and Lap. Suturing and Stapling course H. Soni 2. Laparoscopic UGI Surgery Course J Mistry
PATIENT CONSENT TO PROCEDURE - ROUX-EN-Y GASTRIC BYPASS
 As a patient you must be adequately informed about your condition and the recommended surgical procedure. Please read this document carefully and ask about anything you do not understand. Please initial
As a patient you must be adequately informed about your condition and the recommended surgical procedure. Please read this document carefully and ask about anything you do not understand. Please initial
Dept. of Medical Imaging University of Ottawa
 ED Visits Related to Bariatric Surgery: Review of Normal Post-Surgical Anatomy as Well as Complications Dept. of Medical Imaging University of Ottawa Disclosures Background Roux-en-Y Gastric Bypass Surgery
ED Visits Related to Bariatric Surgery: Review of Normal Post-Surgical Anatomy as Well as Complications Dept. of Medical Imaging University of Ottawa Disclosures Background Roux-en-Y Gastric Bypass Surgery
REVIEW ARTICLE. Left-Sided Pancreatic Cancer. Steven M. Strasberg, MD and Ryan Fields, MD
 REVIEW ARTICLE Left-Sided Pancreatic Cancer Distal Pancreatectomy and its Variants: Radical Antegrade Modular Pancreatosplenectomy and Distal Pancreatectomy With Celiac Axis Resection Steven M. Strasberg,
REVIEW ARTICLE Left-Sided Pancreatic Cancer Distal Pancreatectomy and its Variants: Radical Antegrade Modular Pancreatosplenectomy and Distal Pancreatectomy With Celiac Axis Resection Steven M. Strasberg,
Smoking and misuse of certain pain medicines can affect the risk of developing renal cell cancer.
 Renal cell cancer Renal cell cancer is a disease in which malignant (cancer) cells form in tubules of the kidney. Renal cell cancer (also called kidney cancer or renal adenocarcinoma) is a disease in which
Renal cell cancer Renal cell cancer is a disease in which malignant (cancer) cells form in tubules of the kidney. Renal cell cancer (also called kidney cancer or renal adenocarcinoma) is a disease in which
Cancer of the Cardia/GE Junction: Surgical Options
 Cancer of the Cardia/GE Junction: Surgical Options Michael A Smith, MD Associate Chief Thoracic Surgery Center for Thoracic Disease St Joseph s Hospital and Medical Center Phoenix, AZ Michael Smith, MD
Cancer of the Cardia/GE Junction: Surgical Options Michael A Smith, MD Associate Chief Thoracic Surgery Center for Thoracic Disease St Joseph s Hospital and Medical Center Phoenix, AZ Michael Smith, MD
PHaSES: Practical Hands-on Surgical Education System
 U.S. Toll Free 866-GOLIMBS PHaSES Range PHaSES: Practical Hands-on Surgical Education System Limbs & Things is pleased to introduce the PHaSES Range. The range is based upon our well known basic & general
U.S. Toll Free 866-GOLIMBS PHaSES Range PHaSES: Practical Hands-on Surgical Education System Limbs & Things is pleased to introduce the PHaSES Range. The range is based upon our well known basic & general
CLINICAL MANAGEMENT OF INTRADUCTAL PAPILLARY MUCINOUS NEOPLASMS OF THE PANCREAS
 1 CLINICAL MANAGEMENT OF INTRADUCTAL PAPILLARY MUCINOUS NEOPLASMS OF THE PANCREAS Carlos Fernández-del Castillo, M.D. Associate Professor of Surgery Massachusetts General Hospital Harvard Medical School,
1 CLINICAL MANAGEMENT OF INTRADUCTAL PAPILLARY MUCINOUS NEOPLASMS OF THE PANCREAS Carlos Fernández-del Castillo, M.D. Associate Professor of Surgery Massachusetts General Hospital Harvard Medical School,
Evidence tabel Lokaal palliatieve behandelingen
 Auteurs, jaartal Mate van bewijs Studie type Follow-up Populatie (incl. steekproef-grootte) Patienten kenmerken Interventie Controle Resultaten Conclusie Opmerkingen, commentaar Hartgrink, 2002 The Netherlands
Auteurs, jaartal Mate van bewijs Studie type Follow-up Populatie (incl. steekproef-grootte) Patienten kenmerken Interventie Controle Resultaten Conclusie Opmerkingen, commentaar Hartgrink, 2002 The Netherlands
Abdominal Surgery Rotation Section 7- Whipple resection. 1. Surgical considerations
 Abdominal Surgery Rotation Section 7- Whipple resection 1. Surgical considerations The Whipple resection consists of a pancreaticoduodenectomy followed by anastamosis of the pancreas, liver and stomach
Abdominal Surgery Rotation Section 7- Whipple resection 1. Surgical considerations The Whipple resection consists of a pancreaticoduodenectomy followed by anastamosis of the pancreas, liver and stomach
ERCP in Post Surgical Anatomy
 ERCP in Post Surgical Anatomy ACG Western Regional Course, 2013 John G. Lee, MD Division of Gastroenterology University of California, Irvine Medical Center Common surgical alterations Intact pancreaticobiliary
ERCP in Post Surgical Anatomy ACG Western Regional Course, 2013 John G. Lee, MD Division of Gastroenterology University of California, Irvine Medical Center Common surgical alterations Intact pancreaticobiliary
Surgical Treatment of Various GI Tract Cancers
 Surgical Treatment of Various GI Tract Cancers By James Ouellette, DO, FACS, Surgical Oncology, Hepatobiliary Surgery Surgical treatment for most gastrointestinal (GI) cancers requires multidisciplinary
Surgical Treatment of Various GI Tract Cancers By James Ouellette, DO, FACS, Surgical Oncology, Hepatobiliary Surgery Surgical treatment for most gastrointestinal (GI) cancers requires multidisciplinary
Pancreatic Cancer Understanding your diagnosis
 Pancreatic Cancer Understanding your diagnosis Let s Make Cancer History 1 888 939-3333 cancer.ca Pancreatic Cancer Understanding your diagnosis When you first hear that you have cancer you may feel alone
Pancreatic Cancer Understanding your diagnosis Let s Make Cancer History 1 888 939-3333 cancer.ca Pancreatic Cancer Understanding your diagnosis When you first hear that you have cancer you may feel alone
Protocol for the Examination of Specimens From Patients With Carcinoma of the Exocrine Pancreas
 Protocol for the Examination of Specimens From Patients With Carcinoma of the Exocrine Pancreas Protocol applies to all epithelial tumors of the exocrine pancreas. Endocrine tumors and tumors of the ampulla
Protocol for the Examination of Specimens From Patients With Carcinoma of the Exocrine Pancreas Protocol applies to all epithelial tumors of the exocrine pancreas. Endocrine tumors and tumors of the ampulla
Whipple Procedure: A guide for patients and families UHN
 Whipple Procedure: A guide for patients and families UHN Please visit the UHN Patient Education website for more health information: www.uhnpatienteducation.ca 2014 University Health Network. All rights
Whipple Procedure: A guide for patients and families UHN Please visit the UHN Patient Education website for more health information: www.uhnpatienteducation.ca 2014 University Health Network. All rights
9 TUMORS OF THE PANCREAS, BILIARY TRACT, AND LIVER
 9 Tumors of the Pancreas, Biliary Tract, and Liver 1 9 TUMORS OF THE PANCREAS, BILIARY TRACT, AND LIVER Steven M. Strasberg, M.D., F.A.C.S., and David C. Linehan, M.D., F.A.C.S. Numerous types of tumors
9 Tumors of the Pancreas, Biliary Tract, and Liver 1 9 TUMORS OF THE PANCREAS, BILIARY TRACT, AND LIVER Steven M. Strasberg, M.D., F.A.C.S., and David C. Linehan, M.D., F.A.C.S. Numerous types of tumors
Lesions, and Masses, and Tumors Oh My!!
 Lesions, and Masses, and Tumors Oh My!! Presented by: Susan Ward, CPC, CPC-H, CPC-I, CPCD, CEMC, CPRC 1 1 CPT GUIDELINES Agenda CPT DEFINITIONS OP REPORT CASES 2 Definitions Cyst - a closed sac having
Lesions, and Masses, and Tumors Oh My!! Presented by: Susan Ward, CPC, CPC-H, CPC-I, CPCD, CEMC, CPRC 1 1 CPT GUIDELINES Agenda CPT DEFINITIONS OP REPORT CASES 2 Definitions Cyst - a closed sac having
Practice Anatomy Questions Semester 2
 1 - Which muscle layer does NOT wrap around the abdomen? a) external oblique b) internal oblique c) transversus abdominus d) rectus abdominus 2 - Which statement is correct? a) The fibres of the internal
1 - Which muscle layer does NOT wrap around the abdomen? a) external oblique b) internal oblique c) transversus abdominus d) rectus abdominus 2 - Which statement is correct? a) The fibres of the internal
Metastatic Renal Cell Carcinoma: Staging and Prognosis of Three Separate Cases.
 Metastatic Renal Cell Carcinoma: Staging and Prognosis of Three Separate Cases. Abstract This paper describes the staging, imaging, treatment, and prognosis of renal cell carcinoma. Three case studies
Metastatic Renal Cell Carcinoma: Staging and Prognosis of Three Separate Cases. Abstract This paper describes the staging, imaging, treatment, and prognosis of renal cell carcinoma. Three case studies
Abdominal Injuries. Chapter 17
 Abdominal Injuries Chapter 17 Abdominal Injuries Introduction Changing patterns of warfare coupled with improvements in protective body armor have combined synergistically to decrease truncal and abdominal
Abdominal Injuries Chapter 17 Abdominal Injuries Introduction Changing patterns of warfare coupled with improvements in protective body armor have combined synergistically to decrease truncal and abdominal
Basic Abdominal Sonographic Anatomy and Protocol
 Basic Abdominal Sonographic Anatomy and Protocol Sandra Hagen-Ansert, M.S., RDMS, RDCS Ultrasound Education Specialist and Clinical Consultant Charleston, South Carolina Table of Contents 1. Objectives
Basic Abdominal Sonographic Anatomy and Protocol Sandra Hagen-Ansert, M.S., RDMS, RDCS Ultrasound Education Specialist and Clinical Consultant Charleston, South Carolina Table of Contents 1. Objectives
A Left Paraduodenal Hernia Causing Recurrent Small Bowel Obstruction : A Case Report
 Article ID: WMC001308 2046-1690 A Left Paraduodenal Hernia Causing Recurrent Small Bowel Obstruction : A Case Report Corresponding Author: Dr. Prashanth Shetty, Associate Professor, Kasturba Medical College,
Article ID: WMC001308 2046-1690 A Left Paraduodenal Hernia Causing Recurrent Small Bowel Obstruction : A Case Report Corresponding Author: Dr. Prashanth Shetty, Associate Professor, Kasturba Medical College,
Complex treatment of pancreatic tumours
 Complex treatment of pancreatic tumours PhD Thesis Dr. Róbert Papp Head of Doctorate School : Head of Program : Head of the Theme: Prof. Dr. Sámuel Komoly Prof. Örs Péter Horváth Dr. Dezső Kelemen Department
Complex treatment of pancreatic tumours PhD Thesis Dr. Róbert Papp Head of Doctorate School : Head of Program : Head of the Theme: Prof. Dr. Sámuel Komoly Prof. Örs Péter Horváth Dr. Dezső Kelemen Department
Distance Learning Program Anatomy of the Human Heart/Pig Heart Dissection Middle School/ High School
 Distance Learning Program Anatomy of the Human Heart/Pig Heart Dissection Middle School/ High School This guide is for middle and high school students participating in AIMS Anatomy of the Human Heart and
Distance Learning Program Anatomy of the Human Heart/Pig Heart Dissection Middle School/ High School This guide is for middle and high school students participating in AIMS Anatomy of the Human Heart and
Open Ventral Hernia Repair
 Ventral Hernias Open Ventral Hernia Repair UCSF Postgraduate Course in General Surgery Maui, HI March 21, 2011 Hobart W. Harris, MD, MPH Ventral Hernias: National Experience Occur following 11-23% of laparotomies,
Ventral Hernias Open Ventral Hernia Repair UCSF Postgraduate Course in General Surgery Maui, HI March 21, 2011 Hobart W. Harris, MD, MPH Ventral Hernias: National Experience Occur following 11-23% of laparotomies,
Types of Bariatric Procedures. Tejal Brahmbhatt, MD General Surgery Teaching Conference April 18, 2012
 Types of Bariatric Procedures Tejal Brahmbhatt, MD General Surgery Teaching Conference April 18, 2012 A Brief History of Bariatric Surgery First seen in pts with short bowel syndrome weight loss First
Types of Bariatric Procedures Tejal Brahmbhatt, MD General Surgery Teaching Conference April 18, 2012 A Brief History of Bariatric Surgery First seen in pts with short bowel syndrome weight loss First
Procedure Information Guide
 Procedure Information Guide Surgery to remove the pancreas (whipple's procedure) Brought to you in association with EIDO and endorsed by the The Royal College of Surgeons of England Discovery has made
Procedure Information Guide Surgery to remove the pancreas (whipple's procedure) Brought to you in association with EIDO and endorsed by the The Royal College of Surgeons of England Discovery has made
sufficiently during choledochostomy to result in stricture formation; however, the biliary tract in the prevention of these lesions.
 STRICTURES OF THE COMMON DUCT* WARREN H. COLE, M.D., JOHN T. REYNOLDS, M.D., AND CARL IRENEUS, JR., M.D. CHICACO, ILL. ALTHOUGH THERE HAS BEEN GREAT IMPROVEMENT in the results following repair of strictures
STRICTURES OF THE COMMON DUCT* WARREN H. COLE, M.D., JOHN T. REYNOLDS, M.D., AND CARL IRENEUS, JR., M.D. CHICACO, ILL. ALTHOUGH THERE HAS BEEN GREAT IMPROVEMENT in the results following repair of strictures
CHAPTER 23 DIGESTIVE
 CHAPTER 23 DIGESTIVE nutrition requires : getting nutrients digesting nutrients transporting nutrients Digestive System musculo-skeletal digestive circulatory Digestive System alimentary canal ~ gastrointestinal
CHAPTER 23 DIGESTIVE nutrition requires : getting nutrients digesting nutrients transporting nutrients Digestive System musculo-skeletal digestive circulatory Digestive System alimentary canal ~ gastrointestinal
Gallbladder Cancer. What is gallbladder cancer? About the gallbladder
 Gallbladder Cancer What is gallbladder cancer? Cancer starts when cells in the body begin to grow out of control. Cells in nearly any part of the body can become cancer, and can spread to other areas of
Gallbladder Cancer What is gallbladder cancer? Cancer starts when cells in the body begin to grow out of control. Cells in nearly any part of the body can become cancer, and can spread to other areas of
Billing Guideline. Subject: Colorectal Cancer Screening Exams (Invasive Procedures) Effective Date: 1/1/2012 Last Update Effective: 4/16
 Subject: Colorectal Cancer Screening Exams (Invasive Procedures) Effective Date: 1/1/2012 Last Update Effective: 4/16 Billing Guideline Background Health First administers benefit packages with full coverage
Subject: Colorectal Cancer Screening Exams (Invasive Procedures) Effective Date: 1/1/2012 Last Update Effective: 4/16 Billing Guideline Background Health First administers benefit packages with full coverage
Overview of Bariatric Surgery
 Overview of Bariatric Surgery To better understand how weight loss surgery works, it is helpful to know how the normal digestive process works. As food moves along the digestive tract, special digestive
Overview of Bariatric Surgery To better understand how weight loss surgery works, it is helpful to know how the normal digestive process works. As food moves along the digestive tract, special digestive
4 th Edition Paul H. Sugarbaker, MD, FACS, FRCS
 December 5, 2005 Technical Handbook for the Integration of Cytoreductive Surgery and Perioperative Intraperitoneal Chemotherapy into the Surgical Management of Gastrointestinal and Gynecologic Malignancy
December 5, 2005 Technical Handbook for the Integration of Cytoreductive Surgery and Perioperative Intraperitoneal Chemotherapy into the Surgical Management of Gastrointestinal and Gynecologic Malignancy
Stomach (Gastric) Cancer. Prof. M K Mahajan ACDT & RC Bathinda
 Stomach (Gastric) Cancer Prof. M K Mahajan ACDT & RC Bathinda Gastric Cancer Role of Radiation Layers of the Stomach Mucosa Submucosa Muscularis Serosa Stomach and Regional Lymph Nodes Stomach and Regional
Stomach (Gastric) Cancer Prof. M K Mahajan ACDT & RC Bathinda Gastric Cancer Role of Radiation Layers of the Stomach Mucosa Submucosa Muscularis Serosa Stomach and Regional Lymph Nodes Stomach and Regional
TRAUMA SURGERY Dr. Michal Cheatham Orlando Regional Health PGY-4
 ROTATION LIAISON: INSTITUTION: LEVEL(S): TRAUMA SURGERY Dr. Michal Cheatham Orlando Regional Health PGY-4 I. GENERAL INFORMATION The General Surgery Department at Orlando Regional Health has three full
ROTATION LIAISON: INSTITUTION: LEVEL(S): TRAUMA SURGERY Dr. Michal Cheatham Orlando Regional Health PGY-4 I. GENERAL INFORMATION The General Surgery Department at Orlando Regional Health has three full
Diagnosis and Prognosis of Pancreatic Cancer
 Main Page Risk Factors Reducing Your Risk Screening Symptoms Diagnosis Treatment Overview Chemotherapy Radiation Therapy Surgical Procedures Lifestyle Changes Managing Side Effects Talking to Your Doctor
Main Page Risk Factors Reducing Your Risk Screening Symptoms Diagnosis Treatment Overview Chemotherapy Radiation Therapy Surgical Procedures Lifestyle Changes Managing Side Effects Talking to Your Doctor
Diseases of peritoneum Lect. Al Qassim University, Faculty of Medicine Phase II Year III, CMD 332 Pathology Department 31-32
 Diseases of peritoneum Lect Al Qassim University, Faculty of Medicine Phase II Year III, CMD 332 Pathology Department 31-32 Describe the etiology, pathogenesis and types of peritonitis Define ascites and
Diseases of peritoneum Lect Al Qassim University, Faculty of Medicine Phase II Year III, CMD 332 Pathology Department 31-32 Describe the etiology, pathogenesis and types of peritonitis Define ascites and
Non-coronary Brachytherapy
 Non-coronary Brachytherapy I. Policy University Health Alliance (UHA) will reimburse for non-coronary brachytherapy when it is determined to be medically necessary and when it meets the medical criteria
Non-coronary Brachytherapy I. Policy University Health Alliance (UHA) will reimburse for non-coronary brachytherapy when it is determined to be medically necessary and when it meets the medical criteria
DEPARTMENT OF SURGERY GENERAL SURGERY SECTION
 Privilege Request Form DIRECTIONS: This Privilege Request Form must accompany all initial applications for appointment to the General Surgery Section, Department of Surgery. Please indicate those privileges
Privilege Request Form DIRECTIONS: This Privilege Request Form must accompany all initial applications for appointment to the General Surgery Section, Department of Surgery. Please indicate those privileges
Urinary Diversion: Ileovesicostomy/Ileal Loop/Colon Loop
 Urinary Diversion: Ileovesicostomy/Ileal Loop/Colon Loop Why do I need this surgery? A urinary diversion is a surgical procedure that is performed to allow urine to safely pass from the kidneys into a
Urinary Diversion: Ileovesicostomy/Ileal Loop/Colon Loop Why do I need this surgery? A urinary diversion is a surgical procedure that is performed to allow urine to safely pass from the kidneys into a
A912: Kidney, Renal cell carcinoma
 A912: Kidney, Renal cell carcinoma General facts of kidney cancer Renal cell carcinoma, a form of kidney cancer that involves cancerous changes in the cells of the renal tubule, is the most common type
A912: Kidney, Renal cell carcinoma General facts of kidney cancer Renal cell carcinoma, a form of kidney cancer that involves cancerous changes in the cells of the renal tubule, is the most common type
Kidney Cancer OVERVIEW
 Kidney Cancer OVERVIEW Kidney cancer is the third most common genitourinary cancer in adults. There are approximately 54,000 new cancer cases each year in the United States, and the incidence of kidney
Kidney Cancer OVERVIEW Kidney cancer is the third most common genitourinary cancer in adults. There are approximately 54,000 new cancer cases each year in the United States, and the incidence of kidney
Mesothelioma. 1995-2013, The Patient Education Institute, Inc. www.x-plain.com ocft0101 Last reviewed: 03/21/2013 1
 Mesothelioma Introduction Mesothelioma is a type of cancer. It starts in the tissue that lines your lungs, stomach, heart, and other organs. This tissue is called mesothelium. Most people who get this
Mesothelioma Introduction Mesothelioma is a type of cancer. It starts in the tissue that lines your lungs, stomach, heart, and other organs. This tissue is called mesothelium. Most people who get this
ANTERIOR CERVICAL DISCECTOMY AND FUSION. Basic Anatomical Landmarks: Anterior Cervical Spine
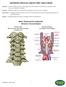 Anterior In the human anatomy, referring to the front surface of the body or position of one structure relative to another Cervical Relating to the neck, in the spine relating to the first seven vertebrae
Anterior In the human anatomy, referring to the front surface of the body or position of one structure relative to another Cervical Relating to the neck, in the spine relating to the first seven vertebrae
CEU Update. Pancreatic Cancer
 CEU Update A semi-annual publication of the National Association for Health Professionals June 2015 Issue #0615 Pancreatic Cancer The Pancreatic Cancer Action Network, Inc. (PanCAN), established in 1999,
CEU Update A semi-annual publication of the National Association for Health Professionals June 2015 Issue #0615 Pancreatic Cancer The Pancreatic Cancer Action Network, Inc. (PanCAN), established in 1999,
Recurrent & Persistent Papillary Thyroid Cancer Central Nodal Dissection vs. Node-Picking Patterns of Nodal Metastases Recurrent Laryngeal Nerve,
 Recurrent & Persistent Papillary Thyroid Cancer Central Nodal Dissection vs. Node-Picking Patterns of Nodal Metastases Recurrent Laryngeal Nerve, Larynx, Trachea, & Esophageal Management Robert C. Wang,
Recurrent & Persistent Papillary Thyroid Cancer Central Nodal Dissection vs. Node-Picking Patterns of Nodal Metastases Recurrent Laryngeal Nerve, Larynx, Trachea, & Esophageal Management Robert C. Wang,
LIVER CANCER AND TUMOURS
 LIVER CANCER AND TUMOURS LIVER CANCER AND TUMOURS Healthy Liver Cirrhotic Liver Tumour What causes liver cancer? Many factors may play a role in the development of cancer. Because the liver filters blood
LIVER CANCER AND TUMOURS LIVER CANCER AND TUMOURS Healthy Liver Cirrhotic Liver Tumour What causes liver cancer? Many factors may play a role in the development of cancer. Because the liver filters blood
Lip Cancer: Treatment & Reconstruction
 Lip Cancer: Treatment & Reconstruction GBMC - Head & Neck Cancer Grand Rounds Elizabeth E. Redd, M.D. With the assistance of Ira Papel, M.D. Patrick Byrne, M.D. Lip Cancer: Treatment & Reconstruction Anatomic
Lip Cancer: Treatment & Reconstruction GBMC - Head & Neck Cancer Grand Rounds Elizabeth E. Redd, M.D. With the assistance of Ira Papel, M.D. Patrick Byrne, M.D. Lip Cancer: Treatment & Reconstruction Anatomic
Gastrointestinal Bleeding
 Gastrointestinal Bleeding Introduction Gastrointestinal bleeding is a symptom of many diseases rather than a disease itself. A number of different conditions can cause gastrointestinal bleeding. Some causes
Gastrointestinal Bleeding Introduction Gastrointestinal bleeding is a symptom of many diseases rather than a disease itself. A number of different conditions can cause gastrointestinal bleeding. Some causes
