Prostate Cancer and Radiation Therapy
|
|
|
- Janis Morton
- 5 years ago
- Views:
Transcription
1 UNIVERSITY OF PATRAS SCHOOLS OF HEALTH SCIENCE - SCIENCE FACULTIES OF MEDICINE - PHYSICS MSc Thesis Clinically derived Dose-Response relations for Rectum and Penile Bulb from combined photon and proton Radiotherapy of prostate cancer CHRISTIANA VALIANTI Examination Committee: Nikiforidis Georgios Sakellaropoulos Georgios Panayiotakis Georgios Supervisor: Mavroidis Panagiotis Advisor Supervisor: Bengt K. Lind Patras, September 2008, Greece
2 ΠΑΝΕΠΙΣΤΗΜΙΟ ΠΑΤΡΩΝ ΣΧΟΛΕΣ ΕΠΙΣΤΗΜΩΝ ΥΓΕΙΑΣ - ΘΕΤΙΚΩΝ ΕΠΙΣΤΗΜΩΝ ΤΜΗΜΑΤΑ ΙΑΤΡΙΚΗΣ - ΦΥΣΙΚΗΣ ΔΙΠΛΩΜΑΤΙΚΗ ΕΡΓΑΣΙΑ Κλινικά προσδιορισμένες σχέσεις Δόσης Απόκρισης για το ορθό και το βολβό του πέους σε συνδυασμένη ακτινοθεράπεια φωτονίων και πρωτoνίων στον καρκινο του προστάτη XΡΙΣΤΙΑΝΑ ΒΑΛΙΑΝΤΗ Εξεταστική επιτροπή: Νικηφορίδης Γεώργιος Σακελλαρόπουλος Γεώργιος Παναγιωτάκης Γεώργιος Επιβλέπων: ΜαυροειδήςΠαναγιώτης Συμβουλευτικός Επιβλέπων: Bengt K. Lind Πάτρα, Σεπτέμβρης 2009, Ελλάδα 1
3 ABSTRACT Purpose Recent advances in radiation therapy have provided the possibility of combining different modalities, energies and particles in different parts of the treatment. However, there are very limited data on the clinical effectiveness of these modalities. The purpose of this study was the determination of the parameters D 50 and γ for the prostate adenocarcinoma, the rectum and the penile bulb using different radiobiological models, when proton and photon beams are combined. Material and Methods Of 189 patients, who were treated for prostate cancer at the Academic Hospital in Uppsala, Sweden from 2002 to 2006, 110 were used in this study, and 100 patients are presented here. The patients were treated with a conventional photon beam configuration to a dose of 50 Gy in 2 Gy fractions. Additionally, a proton boost of 20 Gy in 5 Gy fractions was applied. In some patients, the distance between prostate and rectum was increased during proton therapy by retraction of the rectum in the dorsal direction by the use of a cylindrical rod of Perspex inside the rectum. The photon and proton doses were corrected for the fractionation effects using the BED (biologically effective dose) concept. In the calculation of the proton dose, we considered RBE=1.1. For each patient, the clinical treatment outcome (follow-up time of 24 months) and the 3-D dose distribution delivered to the target, the rectum and the penile bulb were available. The best estimates of the parameters D 50 and γ of the Binomial, Poisson and Probit models were determined by applying a maximum likelihood fitting. Results Of the 100 patients, 94 had tumor control (94.0%) whereas 6 patients had treatment failure (6.0%). The mean doses in the prostate in these two groups of patients were 72.97Gy and 62.60Gy, respectively. Of the 98 patients used for the rectal analysis, 33 produced rectal dysfunction (33.7%). The mean doses in the rectum for responders and non responders were 49.43Gy Vs 52.18Gy respectively. The analysis also shows that the treatment plans with rectal retraction significantly reduces the dose in the rectum and as a consequence the responders of 2
4 the treatment (27.3% vs. 36.9% with and without the rod, respectively). The estimated values of the parameters for the Poisson and Binomial models are D 50 =49.55Gy (68%CI= Gy) and γ=2.25 (68%CI= ) for the tumour, D 50 =61.18Gy (68%CI= Gy) and γ =0.81 (68%CI= ) for the rectum. The estimated values for the Probit model are D 50 =47.27Gy (68%CI= Gy) and γ=1.31 (68%CI= ) for the tumour, D 50 =60.45Gy (68%CI= Gy) and γ=0.97 (68%CI= ) for the rectum. Of the 89 patients used or the penile bulb analysis, 25 had sexual dysfunction (28.1%). The very small difference in the mean doses for the responders and non-responders for the case of penile bulb (55.45 Gy vs Gy) indicates that there may exist other factors apart from dose, that affect the potency of the patients. Conclusions The estimated parameters are able to describe the dose-response relation of prostate adenocarcinoma and rectum. The additional use of the cylindrical rod in the proton treatment appears to reduce rectal side effects. Since there are very limited data on the clinical effectiveness of radiation therapy using combinations of photon and proton beams, the determined parameters can be useful in estimating the expected clinical outcome during treatment planning and perform optimization of the delivered treatment. Future studies must consider other factors apart from dose in the penile bulb, in order to describe erectile dysfunction after radiotherapy of prostate cancer. 3
5 ΠΕΡΙΛΗΨΗ Σκοπός Πρόσφατες εξελίξεις στην ακτινοθεραπεία παρέχουν την δυνατότητα συνδυασμού διαφόρων μονάδων, ενεργειών και σωματιδίων σε διαφορετικά τμήματα της θεραπείας. Ωστόσο, υπάρχουν πολύ περιορισμένα δεδομένα που αφορούν στην κλινική αποτελεσματικότητα αυτών των συνδυασμών. Ο σκοπός αυτής της εργασίας ήταν ο προσδιορισμός των παραμέτρων D 50 και γ για το αδενοκαρκίνωμα του προστάτη το ορθό καθώς και τον βολβό του πέους με τη χρήση διαφόρων ραδιοβιολογικών μοντέλων, στην περίπτωση της συνδυασμένης χρήσης δέσμεων φωτονίων και πρωτονίων. Υλικά και Μέθοδοι Από τους 189 ασθενείς που δέχτηκαν θεραπεία για καρκίνο του προστάτη στο Ακαδημαικό Νοσοκομείο της Ουπσάλας, Σουηδίας από το έτος 2002 μέχρι 2006, οι 110 χρησιμοποιήθηκαν στην παρούσα εργασία, εκ των οποίων οι 100 χρησιμοποιήθηκαν στην στατιστική ανάλυση. Οι ασθενείς δέχτηκαν συμβατική θεραπεία φωτονιών δόσεως 50Gy σε δόσεις των 2Gy και μία επιπλέον δόση των 20Gy από δέσμη πρωτονίων σε δόσεις των 5 Gy. Η χρήση μίας κυλινδρικής ράβδου από Πλεξιγκλάς εντός του ορθού σε μερικούς ασθενείς, απομάκρυνε το ορθό από τον προστάτη, αυξάνοντας έτσι την μεταξύ τους απόσταση. Οι δόσεις φωτονίων και προτωνίων διορθώθηκαν από την επίδραση των συνεδρειών με τη χρήση της έννοιας της Βιολογικά Ενεργής Δόσης (BED). Στον υπολογισμό της δόσης πρωτονίων η σχετική βιολογική επίδραση (RBE) θεωρήθηκε ίση με 1.1. Για την ανάλυση ήταν διαθέσιμο το κλινικό αποτέλεσμα σε διάστημα 24 μηνών από την ολοκλήρωση της θεραπείας καθώς και η τρισδιάστατη κατανομή της δόσης στον προσάτη, το ορθό και το βολβό του πέους. Με τη βοήθεια της προσέγγισης μέγιστης πιθανοφάνειας (maximum likelihood fitting), εκτιμήθηκαν οι βέλτιστες τιμές των παραμέτρων D 50 και γ των μοντέλων Binomial, Poisson and Probit. Αποτελέσματα Από τους 100 ασθενείς, οι 94 είχαν αποδοτική θεραπεία, (94%), ενώ στους υπόλοιπους 6 η θεραπεία δεν απέδωσε (6%). Η μέση δόσε στον προστάτη στις δύο πιο πάνω ομάδες ασθενών ήταν 72.97Gy και 62.60Gy αντίστοιχα. Από τους 98 ασθενείς που χρησιμοποιήθηκαν για την 4
6 ανάλυση του ορθού, οι 33 παρουσίασαν επιπλοκές (33.7%). Η μέση δόση στο ορθό για τους ασθενείς που παρουσιάσαν ή όσι επιπλοκές ήταν 49.43Gy και 52.18Gy αντίστοιχα. Η ανάλυση έδειξε επίσης ότι η χρήση της κυλιδρικής ράβδου μείωσε σημαντικά τη δόση στο ορθό και συνεπώς μείωσε και το ποσοστό των επιπλοκών στο ορθό (27.3% Vs. 36.9% με και χωρίς τη ράβδο αντίσοιχα). Οι τιμες των παραμέτρεων για τα μοντέλα Poisson και Binomial εκτιμήθηκαν ως εξής: D 50 =49.55Gy (68%CI= Gy) και γ=2.25 (68%CI= ) για τον προστάτη, D 50 =61.18Gy (68%CI= Gy) και γ =0.81 (68%CI= ) για το ορθό. Οι αντίσοιχες τιμές για το μοντέλο Probit είναι: D 50 =47.27Gy (68%CI= Gy) και γ=1.31 (68%CI= ) για τον προστάτηr, D 50 =60.45Gy (68%CI= Gy) και γ=0.97 (68%CI= ) για το ορθό. Από τους 89 ασθενείς που χρησιοποιήθηκαν για την ανάλυση για την ανάλυση του βολβού του πέους, οι 25 παρουσίασαν στυτική δυσλειτουρία (28.1%). Στην περίπτωση αυτή, η πολύ μικρή διαφορά στην μέση δόση για τους ασθενείς που παρουσιάσαν ή όχι επιπλοκές (55.45 Gy vs Gy) αντανακλά την ύπαρξη επιπλέων παραγόντων πέρα από την δόση, οι οποίοι ενδέχονται να επηρεάζουν την σεξουαλική δραστηριότητα των ασθενών. Συμπεράσματα Οι εκτιμημένες παράμετροι ειναι ικανές να περιγράψουν την σχέση δόσης-απόκρισης για τον προστάτη και το ορθό. Η επιπλέον χρήση της κυλινδρικής ράβδου στην θεραπέια πρωτονίων μειώνει την πιθανότητα επιπλοκών στο ορθό. Oι καθορισμένες παράμετροι μπορούν να είναι πολύ χρήσιμες στον υπολογισμό του αναμενόμενου κλινικού αποτελέσματος, κατά τη διάρκεια του σχεδιασμού της θεραπείας, ώστε να παρέχεται βελτιστοποίηση της θεραπείας. Μελλοντικές μελέτες που ασκοπούν στην περιγραφή της στυτικής δυσλειτουργίας σαν αποτέλεσμα της ακτινοθεραπείας του προστάτη, πρέπει να λάβουν υπόψη επιπλέον παράγοντες, πέρα από την δόση στο βολβό του πέους 5
7 List of abbreviations AUC CT CTV DNA DRE DSB DVH EBRT EUD FSU GTV HDR HIFU IMRT LET LQ LN MLC MRI NTCP OAR PTV PSA RBE ROC RTOG SOBP TCP TNM TRUS Area Under ROC curve Computed Tomography Clinical Target Volume Deoxyribonucleic Acid Digital Rectal Exam Double Strand Break Dose Volume Histogram External Beam Radiation Therapy Equivalent Uniform Dose Functional Subunit Gross Tumour Volume High Dose Rate High Intensity Focused Ultrasound Intensity Modulated Radiation Therapy Linear Energy Transfer Linear Quadratic Model Lymph Nodes Multi-Leaf Collimator Magnetic Resonance Imaging Normal Tissue Complication Probability Organ At Risk Planning Target Volume Prostate Specific Antigen Relative Biological Effectiveness Receiver Operating Characteristic Radiation Therapy Oncology Group Spread Out the Bragg Peak Tumour Control Probability Tumour Nodes Metastasis Trans-Rectal Ultrasound Scan 6
8 Table of contents Introduction Background Normal Anatomy of Prostate Prostate cancer Incidence Diagnosis of prostate cancer Classification Gleason score Overall staging Side effects after radiation therapy Rectal side effects Erectile Dysfunction Management options of prostate tumour Prostate Surgery Radiation Therapy Three-dimensional Conformal Radiotherapy Intensity modulated Radiation Therapy (IMRT) Proton Therapy Radiation Therapy in Uppsala Brachytherapy Cryosurgery High Intensity Focused Ultrasound (HIFU) Hormonal Therapy Chemotherapy Watchful Waiting Dose Volume Histograms The Radiobiological Basis of Radiotherapy Normal and malignant tissue response to radiation The 5 R s of radiotherapy Dose-Response curves
9 1.6 Radiobiological Models Cell Survival Models Target Theory The Linear Quadratic Model Relative seriality model Dose Response Models The Probit Model The Poisson Model The Binomial Model...59 Materials & Methods Study Bases Hospital records Target Volume and Organs at risk Treatment technique and dose planning Patient fixation and positioning Follow up Dose-Volume Histograms Methods for evaluation Maximum likelihood fitting of the response model to clinical data Fractionation correction ROC Curves...76 Results Dose Distributions Prostate Adenocarcinoma Rectum Subgroups concerning the use of Perspex rod Penile Bulb Summation of photon & proton doses Dose-response parameters ROC curves...95 Discussion
10 4.1 Dose distributions Dose response parameters ROC curves Uncertainties...99 Conclusions Acknowledgments
11 Introduction 1.1 Background Normal Anatomy of Prostate The normal prostate gland is a part of the male reproductive system. It is a quite small, squishy gland and has nearly the same size and shape as a walnut. The normal gland is volume is about cm 3. It is located in front of the rectum, or the lower end of the bowel, and between the bladder and urogenital diaphragm (figure1).the urethra, the narrow tube that runs the length of the penis and that carries both urine and semen out of the body, runs directly through the prostate. Sitting just above the prostate are the seminal vesicles, two little glands that secrete about 60% of the substances that makes up a thick white fluid called semen; running alongside and attached to the sides of the prostate are the nerves that control erectile function. The prostate gland is surrounded by a sheet of smooth muscle that helps expel semen during ejaculation [89]. The prostate is divided into five histologically distinct lobes (anterior, posterior, median and two laterals) and three zones, a central, a peripheral and a transitional zone [59, 89]. Figure 1: The anatomy of male s reproductive system. The prostate is located in front of the rectum, or the lower end of the bowel, and between the bladder and urogenital diaphragm 10
12 The peripheral zone consisting of 70% of the glandular prostate is the side of the most carcinomas of the prostate [59]. One of prostate main roles is to squeeze fluid into the urethra as sperm move through during sexual climax. This fluid, which helps make up semen, energizes the sperm and makes the vaginal canal less acidic. It also produces a protein called prostate-specific antigen (PSA) that turns the semen into liquid. In addition to the prostate's role in producing ejaculate, it also plays a part in controlling the flow of urine, since the prostate wraps itself around the urethra as it passes from the bladder to the penis. In particular, muscular fibres in the prostate contract to slow the flow of urine Prostate cancer Prostate cancer typically is comprised of multiple very small, primary tumours within the prostate (figure 2). At this stage, the disease is often curable (rates of 90% or better) with standard interventions such as surgery or radiation that aim to remove or kill all cancerous cells in the prostate. Unfortunately, at this stage the cancer produces few or no symptoms and can be difficult to detect. In addition, many men who develop prostate cancer undergo no therapy, and they often die of causes unrelated to the prostate cancer, such as heart/circulatory disease, pneumonia, other unconnected cancers, or old age. Sometimes, however, prostate cancer does cause symptoms, often similar to those of diseases such as benign prostatic hypertrophy. These include frequent urination, increased urination at night, difficulty starting and maintaining a steady stream of urine, blood in the urine, and painful urination. Prostate cancer is associated with urinary dysfunction as the prostate gland surrounds the prostatic urethra. Changes within the gland therefore directly affect urinary function. Because the vas deferens deposits seminal fluid into the prostatic urethra, and secretions from the prostate gland itself are included in semen content, prostate cancer may also cause problems with sexual function and performance, such as difficulty achieving erection or painful ejaculation [145]. If untreated and allowed to grow, the cells from primary tumours can spread to other parts of the body and this may cause additional symptoms. The spread of cancer outside the prostate 11
13 can be detected by the presence of prostate cancer cells in areas surrounding the prostate such as the seminal vesicle, lymph nodes in the groin area, the rectum and bones. The most common symptom is bone pain, often in the vertebrae (bones of the spine), pelvis or ribs. Spread of cancer into other bones such as the femur is usually to the proximal part of the bone. Prostate cancer in the spine can also compress the spinal cord, causing leg weakness and urinary and fecal incontinence [159]. Figure 2: A normal prostate and a prostate with located tumour cancer. Prostate cancer typically is comprised of multiple very small, primary tumours within the prostate. Because approximately 90% of all prostate cancers are detected in the local and regional stages, the cure rate for prostate cancer is very high nearly 100% of men diagnosed at this stage will be disease-free after five years. By contrast, in the 1970s, only 67% of men diagnosed with local or regional prostate cancer were disease-free after five years. Once the cancer has spread beyond the prostate, cure rates drop dramatically. The decision whether or not to treat localized prostate cancer with curative intent is a patient trade-off between the expected beneficial and harmful effects in terms of patient survival and quality of life Incidence Prostate cancer is one of the most common types of cancer in men [19]. It Rates of detection of prostate cancers vary widely across the world with South and East Asia detecting less 12
14 frequently than in Europe, and especially the United States [6, 75]. In the USA the estimated incidence of prostate cancer and the mortality rate in 2003 are and respectively [163]. In Sweden, with a total population of 8.9 million people, 786 new cases of prostate cancer were diagnosed in 2002 making it the most common cancer among Sweden men. The incidence of prostate cancer increased by 40% compared with the incidence in 1992 [101]. Many factors, including genetics and diet, have been implicated in the development of prostate cancer. The strongest risk factor for prostate cancer is age. The incidence of prostate cancer is extremely low for men under 50 years of age; it rises exponentially with advancing age and reaches a maximum after the age of 80. For families in which the disease occurs in men under the age of 60 genetic factors appear to play a significant role [25]. The risk for prostate cancer also rises with the number of close relatives who have the disease [25]. Nutritional factors have been hypothesized to be associated with the incidence of prostate cancer [138]. There is a marked difference in the incidence of prostate cancer in various countries. Asian men have much lower incidences of prostate cancer than their Western counterparts. A reason for that may be the low-fat, high fibber diet which is rich in phytoestrogens [37, 50] Diagnosis of prostate cancer Digital Rectal examination (DRE) As the rectum (back passage) is close to the prostate gland, the doctor can feel for any abnormalities in the prostate by inserting a gloved finger into the rectum. If cancer is present in the prostate gland it may feel hard and knobbly, whereas with benign prostatic hyperplasia it is usually enlarged, firm and smooth. However, often the prostate may feel normal, even when cancer cells are present. The DRE cannot diagnose prostate cancer [141]. It is used to examine the size and surface of the prostate to determine if further investigation is needed. Experts recommend that all men over the age of 50 have a DRE yearly along with a PSA test. 13
15 PSA test The pronounced increase in incidence of prostate cancer in the last 20 years is probably primarily due to the widespread use of PSA testing [6]. The presence of prostate cancer may be indicated by an elevated PSA (prostate specific antigen) noticed during a routine checkup. PSA is a glycoprotein produced by the epithelial cells of the prostate gland. A blood test measures the amount of PSA circulating in the blood [144], expressed in nanograms per milliliter (ng/ml). The resulting PSA level is used to assess cancer risk (table 1). PSA level Probability of cancer 0-2 ng/ml 1% 2-4 ng/ml 15% 4-10 ng/ml 25% >10 ng/ml >50% Table 1: PSA levels and there corresponding probabilities of cancer. However, there is no simple correlation between PSA level and disease stage [120, 151], and elevated PSA can also indicate non-cancerous conditions such as infection or benign prostatic hyperplasia (enlarged prostate). Additionally, low PSA levels don't always mean there's no cancer. Many studies have failed to show improvement in mortality or morbidity from PSAscreening for prostate cancer [34,104,121]. PSA is not specific to cancer, but rather to prostate tissue. Suspected prostate cancer is typically confirmed by taking a biopsy of the prostate and examining it under a microscope. Further tests, such as CT scans and bone scans, may be performed to determine whether prostate cancer has spread. Trans-rectal ultrasounds scan (TRUS) Ultrasound scans use sound waves to build up a picture of part of the inside of the body. In this test, a small cylindrical tube about the size of a finger is inserted into the anus. This is an ultrasound probe which emits high-frequency sound waves. These waves bounce back from 14
16 internal structures and are computed. The computations allow an image to be produced on a screen. This image gives the radiologist an idea as to the outline of the prostate and the look of the internal structure of the gland. This type of scan is used to measure the size and density of the prostate [120]. A sample of cells (biopsy) can be taken at the same time for examination under the microscope by a pathologist. The scan may be uncomfortable but it only takes a few minutes. Biopsy If the initial tests (rectal examination, PSA or ultrasound) show that there is a possibility of cancer, the patient may be offered a biopsy, in which several samples of tissue (usually around 10) are taken from the prostate to be looked at under a microscope [140]. The biopsy is normally done at the same time as the ultrasound. A needle is passed through the wall of the back passage (rectum) and into the prostate. The specimens that are taken from the prostate are processed in the pathology department of the hospital. This test is usually uncomfortable, and can sometimes be painful, but it does not need a general anaesthetic. The patient may be given a local anaesthetic to reduce the discomfort. Antibiotics are given to reduce the risk of infection. In the 24 hours following this test it is important to drink plenty of fluids. For up to a few weeks the patient may notice bleeding when passing water, when opening his bowels or in his semen after sex. Unfortunately, even if there is cancer in the prostate it may not be found by biopsy [140]. This will happen in approximately one in 10 or 20 men tested (5 10%). If a biopsy is negative it may need to be repeated (this may detect a cancer that was missed first time). Sometimes the PSA may be measured again after a few months and the biopsy repeated if the PSA level starts to rise. CT and MRI scanning A CT (Computerized Tomography) scan can show whether the cancer has spread to the lymph nodes near the prostate. The patient may have this scan if there is a risk of his cancer spreading 15
17 and he is considering active treatment options such as radiotherapy or radical prostatectomy. The scanner takes X-rays of his pelvis, which are fed into a computer to create an image of the prostate and the surrounding tissues, including the lymph nodes. This process takes approximately quarter of an hour. The doctor can then look more closely for possible signs of spread. The films are then reviewed by a radiologist and assessments made on the possibility of spread of the cancer. Defining the degree of spread allows the doctor to make a judgment as to treatment which can then be discussed with the patient. As with CT, the patient can have an MRI if there is a risk of his cancer spreading and he is considering active treatment options such as radiotherapy or radical prostatectomy. MRI (Magnetic Resonance Imaging) scans can also create a clear picture of the prostate gland. MRI uses magnetic fields rather than X-rays to create a detailed picture of the prostate and surrounding tissues. In addition, a substantial advantage of MRI, is that the use of non ionizing radiation for the creation of the images, minimizes the potential risk for the patient compared to CT scan. It also provides images in multiple imaging planes (not only in the transverse like CT) Classification In order to choose the most appropriate type of management for a particular patient, the prostate tumour is assigned a stage. Staging forms the basis of initial patient management and also provides a guide to prognosis. The most commonly used staging method is the TNM (Tumour Nodes Metastasis) system [157], which recognizes four stages of local tumour growth. Each stage describes the state of pathological development of the tumour. The TNM staging system considers the disease only at diagnosis and has been suggested to use the clinical state from diagnosis to death as a dynamic model of disease progression [97,136]. The TNM system classifies prostate cancer by looking at three factors. Each factor is given a number. Generally, lower numbers mean cancer is smaller and hasn't spread far (if at all): T is for tumour: The number tells how big the cancer is and whether it has spread outside the prostate. 16
18 N is for lymph nodes: The number tells whether prostate cancer has spread to the lymph nodes M is for metastasis: The number tells whether prostate cancer has spread to other areas of the body, such as the hip bones or the back. Figure 3: The four stages of the TNM stage. The number tells how big the cancer is and whether it has spread outside the prostate. T1 represents an incidental state whereas T4 represents advanced disease. T1 represents an incidental state, where the tumour is detected by chance following transurethral resection or by biopsy following PSA testing. At this stage, the tumour will be undetectable by palpation (digital rectal examination [DRE]) or ultrasonography. T4 represents advanced disease, where the tumour has invaded neighbouring organs (figure 3). The nodal stage (N0 N1) and the metastatic stage (M0 M1c) reflect the clinical spread of the disease to lymph nodes and distant sites (metastasis), respectively. A full description of the TNM staging system is shown in table 2. The numbers are then sometimes followed by letters. These tell the doctor how the cancer was found and they give more details about how far it has spread. 17
19 T- Primary Tumour Stage Tx T0 T1 Description The primary tumour was not or could not be assessed. There is no evidence of a primary tumour. The tumour could not be found by examination or with the use of imaging, b was incidentally found during a biopsy or surgery. T1a: The tumour is found in 5% or less of the tissue that was taken. T1b: The tumour is found in more than 5% of tissue that was taken. T1c: The tumour was found by needle biopsy after an elevated level. The tumour is found only within the prostate itself. T2 T3 T2a: The tumour is found in 50% or less of one lobe. T2b: The tumour is found in more than 50% of one lobe. T2c: The tumour is found in both lobes. The tumour has extended through the capsule that surrounds the prostate. T3a: The tumour has only gone through the capsule without invading the seminal vesicles. T3b: The tumour has invaded the seminal vesicles. T4 The tumour has invaded structures or tissues near the prostate other than th seminal vesicles. These include the bladder neck, the rectum, and the pelvi wall along with other structures. (a) N- Regional Lymph Nodes Stage Description Nx The lymph nodes were not or could not be assessed. N0 The nodes do not show evidence of cancer. N1 The nodes show evidence of cancer. (b) 18
20 M- Distant Metastasis Stage Description Mx The presence of metastases was not or could not be assessed. M0 There is no evidence of distant metastasis. There is evidence of distant metastasis. M1 M1a: Cancer has been found in lymph nodes far from the prostate. M1b: Cancer has been found in the bone. M1c: Cancer has been found in another area of the body. (c) Table 2: A full description of the TNM staging system (a) T: adjudicate about the size of primary tumour and whether it has spread outside the tumour (b) N: tell as whether the tumour has spread into the lymph nodes (c) M: indicates about distant metastasis of the tumour Gleason score The Gleason score is used to help evaluate the prognosis of men with prostate cancer. Together with other parameters, the Gleason score is incorporated into a strategy of prostate cancer staging which predicts prognosis and helps guide therapy. The scoring system is named after Donald F. Gleason, M.D., a pathologist at the Minneapolis Veterans Affairs Hospital who developed it with other colleagues at that facility in the 1960s [52].Gleason score remains an important tool. A Gleason score is given to prostate cancer and is based exclusively on the architectural pattern of the glands of the prostate tumour. It evaluates how effectively the cells of any particular cancer are able to structure themselves into glands resembling those of the normal prostate. The ability of a tumour to mimic normal gland architecture is called its differentiation, and experience has shown that a tumour whose structure is nearly normal (well differentiated) will probably have a biological behavior relatively close to normal that is not very aggressively malignant. In addition, higher Gleason scores are given to cancer which is more aggressive. 19
21 To assign a Gleason score, a piece of prostatic tissue must be obtained (a biopsy). This is done either by removing the entire prostate gland (prostatectomy) or by sampling the gland with a needle introduced through the rectum. A pathologist examines the biopsy specimen and attempts to give a score to the two patterns. Figure 4: The five stages of the Gleason score. Score 1 corresponds to a tumour whose structure is nearly normal (well differentiated), while score 5 corresponds to an aggressive tumour (poorly differentiated). First called the primary grade, represents the majority of tumour (has to be greater than 50% of the total pattern seen). Second - a secondary grade - relates to the minority of the tumour (has to be less than 50%, but at least 5%, of the pattern of the total cancer observed). The final score is a combination of the two different scores which each range from one to five. In addition, the Gleason score ranges from two to ten [51,53,97]. A Gleason score of two is associated with the best prognosis and a score of ten with the worst. Gleason scores are associated with the following features (figure 4): G1: The cancerous prostate closely resembles normal prostate tissue. The glands are small, well-formed, and closely packed G2: The tissue still has well-formed glands, but they are larger and have more tissue between them. 20
22 G3: The tissue still has recognizable glands, but the cells are darker. At high magnification, some of these cells have left the glands and are beginning to invade the surrounding tissue. G4: The tissue has few recognizable glands. Many cells are invading the surrounding tissue G5: The tissue does not have recognizable glands. There are often just sheets of cells throughout the surrounding tissue Overall staging The tumor, lymph node, metastasis, and grade status can be combined into four stages of worsening severity (Table 3). Each of these four prostate cancer stages has sub-stages. The meaning of these stages is: Stage I: The cancer is still very small and is located completely inside the area of the prostate gland. This prostate cancer stage is generally curable. Stage II: The cancer is still inside the prostate gland, but is larger and a lump or hard area can be felt during a rectal examination. Most Stage II prostate cancers are curable. Stage III: The cancer has broken through the covering of the prostate and may have grown into the tubes which carry semen. Only a few cases of stage C cancers can be cured. Stage IV: The cancer that has already grown and spread into the rectum, pelvic wall, neck of the bladder, lymph nodes, bones, liver, lungs and other parts of the body. While stage IV cancer is not curable, it could be treated using a variety of therapies and surgery. 21
23 Overall staging Stage Tumour Nodes Metastasis Grade Stage I T1a N0 M0 G1 Stage II T1a N0 M0 G2-4 T1b N0 M0 Any G T1c N0 M0 Any G T2 N0 M0 Any G Stage III T3 N0 M0 Any G Stage IV T4 N0 M0 Any G Any T N1 M0 Any G Any T Any N M1 Any G Table 3: An illustration of the overall staging of a prostate cancer. The tumor, lymph node, metastasis, and grade status can be combined into four stages of worsening severity Side effects after radiation therapy In cancer radiotherapy, the prescribed dose needed to eradicate the involved tumours is limited by the risk of frequent and severe unwanted effects. Late side effects associated with radiotherapy of prostate cancer can permanently deteriorate the patient s quality of life [7, 55, 117] An outline of the side effects of radiation therapy that are related with the rectum and the bulb is shown here, since this thesis deals only with the above organs at risk. It must be noted that, symptoms of urinary tract dysfunction, such as bladder emptying (voiding) symptoms, dysuria (painful burning urination), bladder neck obstruction, a weak stream, bleeding (hematuria), fistula, incontinence and urethral stricture are also due to the large portion of the bladder and urethra included in the PTV during radiation therapy of prostate cancer [108] Rectal side effects Rectum is the lower part of the large intestine, about 10 cm long, continuous with the descending sigmoid colon, proximal to the anal canal (figure 5). It follows the sacrococcygeal 22
24 curve, ends in the anal canal, and usually contains three transverse semilunar folds: one situated proximally on the right side, a second one extending inward from the left side, and the third and largest fold projecting caudally. Each fold is about 12 mm wide. The folds overlap when the intestine is empty or defecation occurs. Figure 5: Normal anatomy of the rectum. The internal and external anal sphincters are rings of muscle at the opening of the anus. The sphincters keep the anus closed as stool collects in the rectum. The rectum acts as a temporary storage for stool prior to defecation. After food has passed through the stomach and intestines and been digested, the leftover material, in the form of feces, enters the rectum, where it stays until defecation. When the rectum becomes full the increase in intrarectal pressure forces the walls of the anal canal apart allowing the fecal matter to enter the canal. Eventually the pressure on the rectum wall causes the internal anal sphincter to relax. Conscious control over the external anal sphincter allows stool to pass out of the body through the anus [154]. In external beam radiation therapy of prostate cancer, a large portion of the cecum, ileum sigmoid colon, rectum and anal sphincter is involved in the treatment. About 75% of men experience no early rectal side effects. About 20% experience either some rectal discomfort or a tendency to diarrhea. Diarrhea is a common side effect of external radiotherapy. It is caused by radiation irritating the lining of the bowel. The remainder may suffer from more severe 23
25 diarrhea or abdominal pains and will need some medication to deal with the problems. Rectal bleeding at an early stage is extremely unlikely. Long term or chronic complications may develop 1 to 2 years following treatment and they are of substantial concern to patients [155]. Most late reactions are seen within 2 years of the completion of radiotherapy [128]. After radiation therapy, the intestine with long-term radiation damage has been described as showing fibrosis, ischemia, stenosis, ulceration, fistulas, telangiectasis, strictures and fibloblasts [15,28,82,155]. The clinical signs and symptoms of the long-term effect of radiation therapy of prostate cancer include urgency, diarrhea, tenesmus, excessive flatulence, soiling, anal sphincter dysfunction (fecal leakage), constipation, mucus in stools, blood in stools or bleeding with ulceration [28,82,29,124, 125,174,165]. The most common long term side effect is proctitis. Prostate cancer radiation therapy which is administered externally may cause proctitis after repeated exposure to radiation. Proctitis is the inflammation of the rectum and the anal sphincter [13]. Proctitis can be short term (acute) or long term (chronic). A randomized trial has shown that conformal techniques reduce the risk of long term proctitis [35]. The most common symptom is a frequent or continuous sensation or urge to have a bowel movement. Other symptoms include constipation, a feeling of rectal fullness, left-sided abdominal pain, passage of mucus through the rectum, rectal bleeding, and anorectal pain. Some men only have the straining feeling. Some only have bleeding. Bleeding is usually slight, but can be more severe for some men. The bowel movements may continue to be looser or more frequent than they were before the treatment. The occurrence of sundries of side effects is certainly a function of the dose. The prevalence of rectal bleeding and diarrhea was 14-34% and 9-25% respectively, after 3-D conformal therapy with Gy [35,98]. In contrast, the prevalence of defecation-urgency, rectal bleeding and diarrhea was 26-33%, 12-50%, and 12-17% respectively, using conventional radiotherapy with Gy [98,35,365]. In a normal population, the prevalence of fecal leakage, defecationurgency, diarrhea and blood or phlegm in the stools has been found to be 2-4 %, 2-11%, 11% and 2% respectively [2,70,165]. It has also been shown that a history of diabetes mellitus, hypertension and adjuvant hormonal therapy has a significant impact on the likelihood of rectal 24
26 bleeding [5,71,140]. It is also likely that other factors, like a large prostate, that make it difficult to avoid subjecting the rectum to high doses of radiation will also influence the occurrence of rectal bleeding Erectile Dysfunction Penile erection is a neurovascular vent modulated by neurotransmitters and the hormonal status. The body of the penis consists of the paired corpora cavernosa and the midline corpus spongiosum (figure 6). Followed posteriorly into the perineum, the two corpora cavernosa separate and form the crura of the penis, attached to the inferior pubic rami. Between the crura, the corpus spongiosum enlarges to form the bulb of the penis, which is attached superiorly to the inferior surface of the urogenital diaphragm. The penis is innervated by autonomic and somatic nerves [67]. Since radiotherapy is usually given in conjunction with hormone therapies, the side effects from the two therapies become entangled. The impact of radiotherapy on sexual function seems less well documented than urinary and rectal side effects [129,69,98]. The most immediate effect that develops over a few months to a year is that orgasms become largely 'dry'. A small amount of ejaculatory fluid may continue to be produced probably by the Cowper glands [173] that are not irradiated. These glands are normally responsible for the production of a small amount of pre-ejaculatory fluid. Figure 6: Normal anatomy of male s reproductive system 25
27 In the longer term, the radiation is thought to damage the nerves that control erections. Erectile function appears to diminish with advancing time after treatment, with 33% to 61% of patients maintaining their erectile function at 5 years or longer after irradiation [142]. These run down either side of the prostate. It has been found that a dose of 50 Gy or more to 50% of the bulb of the penis was associated with increased erectile dysfunction, but there was no relationship between the radiation dose to the neurovascular bundles and erectile dysfunction after brachytherapy [111,112,113]. It has also been reported that a dose escalation higher than 76 Gy was associated with an increased the risk of erectile dysfunction [176]. Because prostate cancer affects mainly older men, it is difficult to distinguish between effects due to general ageing and those due to the radiation [6, 69]. Men are more at risk of having erection problems after radiation therapy if the quality of erections before treatment was borderline [16,137]. In addition, many factors such as age, a history of diabetes mellitus, hypertension, myocardial infraction and drugs, must be associated with the waning of sexual function after radiotherapy [69]. However, for men in their sixties who were potent at the start of their treatment, about half will retain their potency at three years after treatment. For younger men, the figure is probably higher. Because the radiation damages rather than destroys the nerves responsible for erections, drugs like Viagra and Cialis can help to mitigate against 'creeping' impotence [109]. 1.2 Management options of prostate tumour Treatment options for prostate cancer with intent to cure are primarily surgery and radiation therapy. Other treatments such as hormonal therapy, chemotherapy, proton therapy, cryosurgery, high intensity focused ultrasound (HIFU) also exist depending on the clinical scenario and desired outcome. The choice of treatment is a very difficult issue for both physician and patient. Every case is unique and therefore the physician must always consider the exact problem of the patient as much as his history. However, recently statistics show that there are some benefits of each treatment in perspective with the others. Surgical patient have higher rates of urinary incontinence and probably also erectile dysfunction than irradiated patients [2,33,123,129,148]. In contrast, anal sphincter and large 26
28 bowel dysfunction develops as a consequence of radiation therapy but not radical prostatectomy [14,33,65,165,123,129,148]. It has been suggested that the addition of hormone therapy to radiation therapy or prostate cancer does not increase the risk of sexual dysfunction [27,126]. A guarded analysis of single patient series showed that radical prostatectomy, as compared to watchful waiting, halves the risk of dying of prostate cancer among men with moderately or highly differentiated localized prostate cancer [1,72]. In a population based study the 10 year prostate cancer-specific survival in patients with clinically localized prostate cancer for patients analyzed as intention to treat, was 83%, 75% and 82% respectively, after radical prostatectomy, radiotherapy and watchful waiting [103]. It has also been found that differential staging can significantly influence the observed outcomes [103] Prostate Surgery Surgery is one of two main treatments for early-stage prostate cancer. Surgery may be done to remove the entire prostate gland, with resection of both seminal vesicles [10]. Surgery may completely remove the prostate cancer. But it is not possible to know ahead of time whether the cancer has spread beyond the prostate and is not curable with surgery alone. Good candidates for surgery to treat prostate cancer have one or more of the following characteristics: Good health No spread of cancer to bone Tumour confined to the prostate gland (stage T1 and T2) Under the age of 70 Expected to live another 10 years or longer Radical prostatectomy can be done as open surgery through an incision (cut) in the belly, or as laparoscopic surgery through several very small incisions in the belly. Laparoscopic surgery is done with a tiny camera and special instruments to remove the prostate [10]. Sometimes lymph nodes in the area also are removed so that they can be checked for signs of cancer. This is called a lymph node biopsy. A few doctors now do this surgery by guiding robotic arms that hold the surgery tools. This is called robot-assisted prostatectomy. Radical prostatectomy is major surgery, so it carries the same general risks as other major operations, including heart 27
29 problems, blood clots, allergic reaction to anaesthesia, blood loss, and infection of the wound. Survival is very good after surgery, cancer-specific survival is about 90% at ten years and 82% at 15 years [54,130,179]. Up to 80% of men experience erection problems after a radical prostatectomy [168]. The nerves that control a man's ability to have an erection lie next to the prostate gland. They often are damaged or removed during surgery. In the months and years after surgery, most men who had erection problems after prostatectomy are able to regain their ability to have erections [38]. Recovery depends on, whether the man was able to have an erection before surgery, how the surgery affected the nerves that control erections, how old the man was at the time of surgery [38]. With nerve-sparing surgery, a procedure introduced by Walsh and Donker in 1982 [160], erectile function can be preserved [137]. This is only done when there is little chance of leaving cancer cells behind. If the patient already has sexual function issues, nerve-sparing surgery may not be the best choice. In order to remove the prostate, the surgeon must cut the urethra and later reconnect it to the bladder. In addition, up to half of all men who have a radical prostatectomy develop urinary incontinence, ranging from a need to wear urinary incontinence pads to occasional dribbling. Studies show that one year later, between 15% and 50% of men report urinary problems [168] Radiation Therapy Radiotherapy (RT) is one of the primary treatment modalities for localized prostate cancer, especially in patients with the disease well confined to the prostate, in which it often achieves a reasonable probability of tumour control with good cosmetic results. Although local control is more likely to be achieved with high radiation therapy dose, one must always consider the tolerance dose of the organs at risk and thus the radiation dose that can be given is limited. In addition, the aim of RT is to deliver as high and homogenous dose as possible to the tumour target without causing unwanted and unnecessary side effects to the patient. Most commonly doses of Gy are prescribed when conventional treatment plans are used [68]. Conformal techniques including three-dimensional-conformal RT (3D-CRT) and intensity modulated RT 28
30 (IMRT) have allowed dose escalation to the prostate beyond 70Gy with improved PSA control and acceptable toxicity [128,63,89]. Moreover, data form Radiation Therapy Oncology Group (RTOG) studies have shown that for high grade tumours, a radiation dose of 66Gy or more decreased the risk of death from prostate cancer to 29% compared with men treated with lower doses [59]. A pioneer method to reduce the dose to OR, and especially to the rectum is combined proton and photon conformal radiotherapy [77], where the total dose of 70Gy to the prostate is given by additional doses from separate photon and proton beams. Generally, proton radiotherapy may represent a major advance in radiation therapy in part because of excellent dose distributions around the tumour, and in part because of the potentially lower whole body dose compared with photon radiotherapy Three-dimensional Conformal Radiotherapy The aim of 3-D conformal radiotherapy is to reduce the dose load to normal tissue by exactly tailoring the dose distribution to match the planning target volume. It is obvious that 3D conformal radiotherapy requires that PTV is properly defined [78]. Thus, a 3-D conformal radiation therapy treatment plan is based on 3-D imaging in the delineation of target volumes and OARs, and three dimensional treatment planning systems, and the dose is delivered by combining a number of beams from different directions to irradiate the PTV (figure 7). The 3- D description of the patient s internal anatomy is achieved with the use of CT scanners [48].With the additional help of computer-controlled treatment machines equipped with multileaf collimators and contemporary computer power and software sophistication the clinical implementation of conformal treatment planning can be accomplished [48]. Figure 7: In 3-D conformal radiotherapy, the dose is delivered by combining a number of beams from different directions to irradiate the PTV 29
31 Production and modulation of photon beams Generally, in photon radiotherapy the patients are treated with accelerator-produced radiation (figure 8). Electrons are produced from an electron gun and are guided to the entrance of the linear accelerator, where they enter in pulses. In the same time a magnetron produces electromagnetic waves, in the frequencies of microwaves, which are guided in the entrance of an accelerating tube. If each electron pulse enters the accelerating tube when the microwave has its maximum value, then the electrons will be accelerated by the axial component of the electric field. In that way the electrons receive very high velocities, near the velocity of light (e.g. electrons with energies 1 MeV and 10 MeV reach velocities equal to 0.941c and 0.999c respectively). Afterwards the electrons are focalized and focused via centering and focusing coils. The accelerated electrons are then passing through a strong bending magnetic field where they forced to turn from the horizontal to the vertical direction and trip to a tungsten target where photons are produced. The produced photons, pass through a primary collimator, through a field flattering filter and then through ion chambers to check the homogeneity of the beam. The beam is shaped by a block collimator to define the target area, resulting in a square or rectangular field. The beam shaping can also be achieved by the backup-jaws, cerrobend blocks, or a Multi-leaf collimator (MLC) [161,162]. A Multi-leaf collimator consists of a set f parallel focused opposed metal leafs. Each leaf can be controlled separately in the forward or transverse direction in to order to fix the dose distribution to the shape of the planning target volume. Multi-leaf collimators have the advantage that they shape the radiation field from the beam seye view, which is the prospect of patient s anatomy as it would appear to a viewer located at the radiation source and looking toward the isocenter of the PTV. Randomized clinical trials have demonstrated a clinically significant reduction of late effects in patients with prostate cancer treated with 3-D conformal radiotherapy as compared with conventional radiotherapy [35]. Also, other unrandomized clinical studies have suggested that dose escalation in 3-D conformal radiotherapy improves tumour control [64,175] 30
32 Figure 8: A design of the linear accelerator used for 3-D conformal radiotherapy Intensity modulated Radiation Therapy (IMRT) A modern technique of a three dimensional conformal radiotherapy is called Intensity Modulated Radiation Therapy (IMRT) [80]. IMRT with associated computerised optimization of fluence profiles is a technology that has been developed dramatically since the end of 1980s [20,21,23]. IMRT could be used for the whole duration of radiotherapy or as a boost. To obtain the desired dose distribution to the target and at the same time spare specific normal tissues, the fluence profiles generally have to be non-uniform. This can be achieved with an interactive 31
33 computer optimization procedure that calculates, for all the selected beam directions, the fluence profiles necessary to obtain the desired dose distribution within the patient. Thus, with IMRT the intensity of radiation varies in a controlled way across the beams (figure 9) [95,162]. Figure 9: In IMRT the intensity of radiation varies in a controlled way across the beams in order to obtain the desired dose distribution. The beam intensity will have the largest value at the point where the target has the largest diameter whereas the beam intensity will have the smallest value where the target has the smallest diameter. The beam intensity will have the largest value at the point where the target has the largest diameter whereas the beam intensity will have the smallest value where the target has the smallest diameter. So, different subfields in different segments will combine (figure 10) to make the three dimensional IMRT field. In this way, the spatial distribution conforms as tightly as possible to the spatial distribution of cancer cells, thereby reducing the dose to the adjacent normal tissues even if they lie within a concavity surrounded by the planning target volume [23,162]. There are two main IMRT delivery techniques, the cone beam IMRT and tomotherapy. Cone beam uses standard C-arm linear accelerators equipped with MLCs that can be used by two different techniques. The first technique which is called step and shoot, builds up the fluence profiles from multiple static fields from each beam direction [110,162]. MLC does not change shape during a radiation beam-on session. Thus, the gantry of the linear accelerator steps in a specific angle and shoots the corresponding static field. 32
34 Figure 10: Different subfields in different segments are combined to make a three dimensional IMRT field. In this way, the spatial distribution conforms as tightly as possible to the spatial distribution of cancer cells The second technique is called sliding window where MLCs change shapes as a function of monitor units or gantry angle during a radiation beam-on sequence [102]. In that way, dynamic movements of the MLC are used to obtain the fluence profiles from each beam direction. In contrast, tomotherapy can be described as slice or fan beam IMRT [100,105]. With serial tomotherapy one slice of the tumour is treated at a time with a series of gantry angles up to a full rotation after which the patient is moved to treat the next slice Proton Therapy In 1946, Robert Wilson suggested that protons could be used for medical purposes [167]. The first use of proton beams for treatment of human patients occurred at the University of California, Berkeley, California, USA I the mid-1950s [153]. The proton beam (340 Mev) was used to irradiate the pituitary gland since its well-localized structure is an ideal target for proton therapy. Proton treatment was followed by others in the 1950s and 1960s by other centres: The Svedberg Laboratory (TSL), Uppsala (Sweden), the Harvard Cyclotron Laboratory, Boston Massachusetts (USA), Dubna and Institute of Theoretical and Experimental Physics (ITEP), Moscow (Russia). 33
35 Figure 11: An outline of The Svedberg Laboratory (TSL), Uppsala, Sweden The proton beam from the synchrocyclotron at the TSL was developed both for neurosurgery [90] and for large-field proton treatments [58,149]. Many of the physical beam delivery techniques in use today were first implement by the Uppsala group. These include the first use of scanned proton beams for production of large treatment fields and the use of range modulation to spread out the Bragg-Peak (SOBP) [94]. The first medically dedicated proton beam facility was build at Loma Linda University in California (LLUMC), USA in 1990 [143] and several other medically dedicated facilities around the world are in the construction and planning stages. The physical properties of proton beams Protons are positively charged ions (H + ) and thus the dominant effect of energy loss is Coulomb interaction with the atomic electrons. However, protons also deposit energy in matter by Coulomb interactions with nuclei, as much as by bremsstrahlung and by nuclear reactions. These interactions lead to a slowing down of the primary protons. The dose that is absorbed by the matter is proportional to the energy deposition per unit path length, i.e. the stopping power de dx. The stopping power increases as the proton energy decreases until very little energy remains. The path of protons was first described by Bragg [24], which proposed that as the 34
36 protons penetrate into the material, the absorbed dose increases gradually and near the end of the range the dose increases rapidly, creating a peak (figure 12) that bears Braggs name. Figure 12: Depth-dose curves for a 173 MeV proton beam, 21 MV photon beam and 16 MeV electron beam. All protons have approximately the same range due to the small fluctuations in energy loss over the path (energy strangling) and thus the dose distribution has a sharp fall-off near the maximum range. Moreover, the much greater mass of the protons in perspective with electrons (approximately 1835 times heavier) prevent the creation of substantial penumbra during the scattering of protons by the atomic electrons. In addition, the sharp fall-off of the dose distribution in combination with the relatively sharp penumbra will enable the use of proton beams to conform the high-dose region to the target volume (figure 13) and thus minimize the dose the adjacent normal tissue. 35
37 Figure 13: Parallel Opposed Beam Techniques for different types of particle beams (Brahme et al 2001). Protons have the advantage to conform the high region of the dose to the tumour For that reason, studies showed that RBE tends to increase with depth of the beam [18]. Specifically, it has been reported [42,49] that RBE varies between 1.1 and 1.6 at the end of their range. The RBE corrected dose can be calculated using the linear-quadratic cell survival model with tissue specific parameters for cell survival as a function of LET [152]. Figure 14 shows the results of a RBE corrected depth dose cuve. Although RBE correction in the dose is important, uncertainties in the in vitro and the clinical in vitro data are still large, and until there are solved a clinical RBE of 1.1 is used [62]. Figure 14: Central-axis depth dose curves for one spread out Bragg peak from the monoenergetic 173 MeV proton beam (black line) and one corrected for RBE (red line). 36
38 Production and modulation of proton beams Protons are produced and accelerated in a cyclotron. With the help of an electrical field, protons are extracted from plasma contained within an ion-source, located at the centre of the cyclotron. The particles are then captured in a circular trajectory by the strong magnetic field and accelerated in the high-tension gap between the D-shaped electrodes. This gap is passed many thousands of times, successively increasing the energy of the particles and causing them to spiral outwards. At the pole edge there is a system of electromagnetic elements which guide the particles out of the cyclotron in a well defined and bunched beam.the cyclotron used in TSL is a synchrocyclotron. A synchrocyclotron is a cyclotron that achieves relativistic velocities by modulating the frequency of the accelerating electric field. More specific, synchrocyclotron is a cyclotron in which the frequency of the acceleration voltage decreases after a while so that it adjusts exactly to the orbits of the accelerated particles to compensate for relativistic increases in particle mass at high speeds. Since only one proton bunch can be accelerated through the synchrocyclotron before the acceleration of the next bunch begins, the proton beam is pulsed. Figure 15 shows the optical bench where the beam leaves the vacuum pipe and passes through the modulation phase. Figure 15: The optical bench where the beam leaves the vacuum pipe and passes through ion chambers, degraders and collimators, to adjust its shape to the tumour. 37
39 The proton beam that is extracted from the synchrocyclotron is a narrow pencil with a sharp Bragg peak. To broaden the beam a first scatterer is used (figure 16). The proton beam is passed through a scattering foil, and produced a proton beam with Gaussian profile. To flatten the beam, a second scatterer is used, namely a Gaussian scatterer (figure 16). The tails of the beam are cut off with the help of collimators that are positioned from both sides of the beam. Now the sharp Bragg-peak of the proton beam must modulated in range in order to give a homogenous dose to an extend volume. In the TSL the spread out of Bragg-peak is achieved passively by using a rotating stepped absorber [87]. This is an absorber that its thickness varies radially in specific steps. During the therapy the absorber is rotating and thus, depending on the phase that the beam meets the absorber, it passes through different thicknesses and so the beam is modulated in equal number of steps. Figure 16: A plot of the scatterers that the proton beam passes through before it reaches the patient. A first scatterer is used to broaden the beam and a Gaussian scatterer flatters the beam. The tails of the beam are cut off with the help of collimators that are positioned from both sides of the beam. The Bragg peak will widen, according to the number of the absorber s steps. More steps are needed for a wider tumour, and fewer steps are needed for a tighter tumour. In figure 17, the Bragg peak of a 173 MeV proton beam has been modulated in 10 steps to create a uniform dose over the last 54 mm of the depth dose curve, together with a 6 MV photon beam for comparison. The figure shows that photons give much higher dose both on the entrance and exit sides of the target. 38
40 Figure 17: Central-axis depth dose curves for one mono-energetic 178 MeV proton beam, and a 6 MV photon beam. The SOBP of the proton beam is also shown. It is obvious that photons give much higher dose both on the entrance and exit sides of the target. Apart form the passive scanning, active scanning is used for the SOBP by means of magnetic deflection. The use of scanning to achieve large uniform treatment fields was first implemented by the Uppsala group, where scanning magnets produced diamond patterns of variable size to cover the treatment field [94]. The narrow proton beam can also be scanned in a raster pattern or in concentric rings to produce uniform fields of variable sizes [132]. Dynamic scanning minimizes energy losses and the maximum useful range is preserved. Since the proton beam extracted from the synchrocyclotron is monoenergetic (178 MeV) and thus it has a specific range of 20cm, the range must be modulated accordingly to the distance of the volume. Thereby, the range of a passively scattered therapy beam is adapted to the distal end of the target volume by applying a range compensator or bolus (figure 18). This filter is machined such that the maximum range is conformed to the shape of the target. 39
41 Figure 18: (a) The monoenergetic beam that is extracted form the synchrocyclotron is a narrow pencil beam with a sharp Bragg peak. (b) SOBP aims in widen the Bragg peak and (c) A range compensator or bolus adapts the range of the beam to the distal ed of the target volume Radiation Therapy in Uppsala In 1957, in Uppsala, Sweden, for the first time a patient with cancer was irradiated with a proton beam [43]. Also stereotactic radiosurgery in the brain with the 185 MeV proton beam was performed in Uppsala [91]. The first patient was treated with stereotactic radiosurgery in 1958 [93]. In addition, beam scanning and the use of range-modulated beams were first developed in Uppsala [58, 94,147,57]. More than patients receive radiotherapy in Sweden every year. From those patients, 99% are treated with conventional beam qualities. However, more than 100 patients per year are treated with Protons at The Svedberg laboratory in Uppsala. Between the years , 73 patients were treated with protons at TSL. This number was rapidly increased between the years where 418 patients had been treated with protons. Now, approximately patients per year in Sweden are benefit from proton therapy, and that corresponds to the % of all radiotherapy patients. It must be noticed that light ions are not used in Sweden Brachytherapy Brachytherapy (the term derived from the Greek word brachys, which means brief or short) refers to cancer treatment with electromagnetic radiation delivered via radioactive material placed a short distance from, or within, the tumour. As it said before, the prostate gland is located under the bladder and in front of the rectum, and so it is vital that the radiation be 40
42 focused in the prostate to avoid serious side effects. The prostate gland is also close enough to the skin that it can be easily reached by brachytherapy needles. Brachytherapy works more effectively on small and moderately sized prostate glands. Men with very large prostates often undergo a 3- to 6-month course of hormone therapy to shrink the prostate prior to implantation. Patients are excluded from brachytherapy as monotherapy if their Gleason grade is grader than 7, their PSA levels are greater than 18 ng/ml or if their clinical stage is greater than T 2 [9,41]. The target volume, the volume to be implanted, includes the whole prostate within the capsule plus a 2-3 mm margin [41]. Depending on the circumstances, either radioactive Iodine (I-125) or palladium (Pd-103) will be used. The dose prescribed is 145 Gy for I-125 and 125 Gy for Pd-103 at the periphery of the target volume [9,41]. There are two major methods of prostate brachytherapy, permanent seed implantation and high dose rate (HDR) temporary brachytherapy (figure 19). Permanent seed implants involve injecting approximately 100 radioactive seeds into the prostate gland. They give off their radiation at a low dose rate over several weeks or months, and then the seeds remain in the prostate gland permanently. The prescribed dose for temporal brachytherapy is usually Gy/2 fractions added to 4050 Gy using external beam radiation therapy [88]. HDR temporary brachytherapy instead involves placing very tiny plastic catheters into the prostate gland, and then giving a series of radiation treatments through these catheters. The catheters are then easily pulled out, and no radioactive material is left in the prostate gland. A computercontrolled machine pushes a single highly radioactive iridium seed into the catheters one by one. Because the computer can control how long this single seed remains in each of the catheters, it is able to control the radiation dose in different regions of the prostate. Tumour can be given a higher dose, and yet the urine passage (urethra) and rectum will receive a lower dose. This ability to modify the dose after the needles are placed is one of the main advantages of temporary brachytherapy over permanent seed implants. Brachytherapy may have some minimal side effects, such as a burning sensation during urination or ejaculation and increase in urinary frequency. These symptoms are usually treated supportively and go away rather quickly. In few patients, temporary difficulties with urination may be experienced for a few weeks to a few months after the implant. The chances of developing impotence or incontinence with the brachytherapy procedure are less when 41
43 compared to other treatments [46]. Impotence has been reported by various groups of patients, although most patients receiving brachytherapy have often reported similar levels of potency before their treatment. However, the risk of impotence does increase with age. Figure 19: There are two major methods of prostate brachytherapy, permanent seed implantation and high dose rate (HDR) temporary brachytherapy. (a)permanent seed implants involve injecting approximately 100 radioactive seeds into the prostate gland. (b)hdr temporary brachytherapy involves placing very tiny plastic catheters into the prostate gland, and then giving a series of radiation treatments through these catheters Cryosurgery Cryosurgery (also known as cryotherapy, cryoablation, or cryosurgical ablation) is a procedure in which the prostate gland is frozen under controlled conditions in order to kill cancer cells. It is recommended for patients who cannot tolerate surgery or radiation, have prostate-confined tumours (stage T3 or lower), do not respond to radiation (both external-beam and brachytherapy), and are elderly. Transrectal ultrasound and prostate biopsy are performed prior to cryosurgery to determine the exact size and location of the tumour(s). Cryosurgery works best on prostates 40 grams or less in size as measured by ultrasound. Three to six months prior to the procedure, the patient is placed on hormone therapy. Hormone therapy shrinks the prostate and cancer prior to cryosurgery and improves the chances of freezing the entire prostate. The procedure is performed under regional (e.g., epidural nerve block) or general anesthesia. With the man on his back, the surgeon inserts a warming catheter into the urethra to protect it 42
44 from freezing temperatures. An ultrasound transducer is inserted into the rectum, so the surgeon can see the prostate and surrounding tissue and monitor placement of the cryoprobes. The surgeon then makes 5 to 8 needle punctures in the perineum and advances the needles to preselected locations in the prostate tumour. Liquid nitrogen or argon gas circulates through the probes and freezes cancer cells to -40ºC. The temperature in and around the prostate is monitored with thermosensors, also inserted through the perineum. Once the spheres of tissue surrounding the cryoprobes are covered with ice, the liquid nitrogen or argon circulation is stopped and the area is allowed to thaw. The freeze-thaw cycle is repeated and then the instruments are removed. The procedure takes about 2 hours. The urethral warming device causes incontinence or urethral obstruction in about 1% of patients. There is an 85% chance that freezing will result in nerve damage and cause impotence (erectile dysfunction). However, nerve-sparing techniques are being developed to help reduce that risk High Intensity Focused Ultrasound (HIFU) HIFU is a noninvasive treatment that uses precision-focused ultrasound waves to heat and destroy (ablate) targeted prostatic tissue without affecting healthy surrounding tissue. HIFU is a method of focusing ultrasound waves in the same way as a convex mirror can focus the sun s rays to set light to a piece of paper. Thus, ultrasound energy, or sound waves, is focused at a specific location and at that "focal point" the temperature raises to 90 degrees Celsius in a matter of seconds. Because the prostate is deep within the pelvis, the patient has HIFU for prostate cancer by putting an ultrasound probe into his back passage (rectum). Doctors call this a transrectal probe. From that position, the ultrasound can direct beams more accurately at the prostate.it has been shown to effectively treat localized prostate cancer as well as benign prostatic hyperplasia (BPH). In clinical trials, HIFU is performed on an outpatient basis, under anesthesia. HIFU can be repeated as necessary, and each treatment takes 1 3 hours. Following treatment, a catheter is necessary for about 1 week and most patients are able to resume regular activities within days. Impotence occurs in 1 7% of patients. 43
45 1.2.5 Hormonal Therapy Prostate cancer cells are just like all other living organisms, they need fuel to grow and survive. Because the hormone testosterone serves as the main fuel for prostate cancer cell growth, it is a common target for therapeutic intervention in men with prostate cancer. Hormone therapy, also known as androgen deprivation therapy, is the use of drugs or surgery to decrease the production of male hormones, or androgens, in order to stop or limit the growth of prostate cancer. Once the testosterone is out of patients body, the prostate cancer usually shrinks. Hormone treatments are most often used in patients with cancer that has already spread beyond the prostate gland, or in men whose cancer has returned after radical prostatectomy or radiation therapy. The majority of cells in prostate cancer tumours respond to the removal of testosterone. But some cells grow independent of testosterone, and therefore remain unaffected by hormone therapy. Thus, hormonal therapy may allow a reduction in the radiotherapy target volume of 20-50% [45,178]. On comparing patients with T2-T4 primary tumours treated with combined androgen blockade for 2 months before and during radiotherapy to a group treated with radiotherapy alone, the improvements were in both local disease control at 5 years (75% vs. 64%) and freedom from metastasis (71% vs. 61%) [127]. Hormone therapy is therefore not a perfect strategy in the fight against prostate cancer, but it remains an important step in the process of managing advancing disease, and will likely be a part of every man s therapeutic regimen at some point during his fight against recurrent or advanced prostate cancer Chemotherapy Chemotherapy is the third most important treatment modality, after radiotherapy and surgery. Chemotherapy makes use of anti-cancer (cytotoxic) drugs to destroy cancer cells. Although it is often used to treat cancer, it is less commonly used to treat cancer of the prostate. Prostate cancer chemotherapy is almost always a salvage therapy for advanced prostate cancer. Usually when treatments like cryotherapy or prostate surgery fail the next step is either another type of treatment or prostate cancer hormone therapy. Once the patient begins hormone therapy, the disease can be effectively controlled and even halted for a period of time. Hormone therapy 44
46 will eventually fail however and the prostate cancer will continue to grow and spread. The next step is usually chemotherapy. Like radiation therapies, chemotherapy does not destroy the entire body because only cells that divide soon after being treated will die. Unfortunately, chemotherapy cannot be focused to any particular area of the body. All quickly-dividing cells of the body therefore are affected, including, those in the hair follicles, skin, gastrointestinal tract, and bone marrow. The severe and sometimes dangerous side effects of chemotherapy drugs have often outweighed their benefits as an early prostate cancer treatment. However, for patients with advanced disease, chemotherapy can be beneficial in both extending the life and decreasing pain. Sometimes the disease will metastasize to the bones and is called advanced prostate cancer with bone metastasis. Bone metastasis can occur during hormone therapy treatment but may not be apparent without a bone scan. Chemotherapy can be used to treat the cells in the bones that are now dividing malignantly. The pain associated with bone metastasis can be greatly relieved by the use of chemotherapy drugs. External radiation therapy is also sometimes used in conjunction with chemotherapy as a way to alleviate pain associated with advanced prostate cancer Watchful Waiting One of the management options for prostate cancer is a treatment called watchful waiting. It's a reasonable option in men who, based upon their tumour-specific variables, their age and their overall health, have a low risk of dying from their prostate cancer. Because many men with a slow-growing tumour have the same life expectancy as men who don't even have prostate cancer, it may not be necessary to treat very small, very slow-growing prostate tumours. Also, some men feel that the side effects of treatment outweigh the benefits. In watchful waiting, the patient gets no treatment, but sees his doctor often. During the watchful waiting period, the physician keeps the cancer under close watch and this is why it sometimes is referred to as active surveillance. Watchful waiting entails regular prostate-specific antigen (PSA) tests, digital rectal exams (DRE) and/or other tests. "Waiting" means being alert for any indication 45
47 that the cancer has developed to the point that it may require surgery or other treatment. If there's no sign the cancer is growing, he continues to get no treatment. Hormone therapy can be started if the cancer starts to grow. The major risk of watchful waiting is that without treatment, prostate cancer can grow and spread outside the prostate capsule before the next doctor's visit. Even small, slow-growing tumours may become rapidly growing tumours if left untreated. And sometimes prostate cancer that appears to be small and slow growing may be larger and more aggressive than originally thought. 1.3 Dose Volume Histograms Dose volume histograms (DVHs) are extremely powerful tools for quantitative evaluation of treatment plans. They take advantage of the fact that the dose distribution information in a 3-D treatment plan can be reproduced over a 3-D matrix of points over the patient s anatomy and they summarize this information in a 2-D plot of the volume V as a function of dose D ( V = F( D) ). Thus a DVH represents the frequency distribution of dose values within a defined volume that may be the PTV itself or a specific organ at risk. There are two types of DVHs in use; the direct or differential DVH and the cumulative or integral DVH. It must be noticed that an important disadvantage of DVHs is that they contain no spatial information. Differential Dose Volume Histogram The differential DVH can be created if we answer to the question what percentage of the defined volume received an average dose d i within a specific range d1 di d 2. Thus, the computer sums the number of voxels with an average dose within the given range and gives a plot of the Volume as a function of Dose. Figure 20 shows an example of a differential DVH for a PTV and for an organ at risk. The resulting volume is a percentage of the total organ volume ( VV) whereas the Dose is normalized to 100%. The ideal DVH for a PTV would be 0 a single vertical line that crosses the dose-axis at the value d = 100, indicating that 100% of 46
48 the volume receives the prescribed dose. For an organ at risk, the DVH may contain several peaks indicating that different parts of the organ receive different doses. Figure 20: (a) The differential DVH for a PTV (continuous line) and the ideal differential DVH for a PTV (dot line), which indicates that 100% of the volume receives the prescribed dose (b) The differential DVH for a normal tissue indicates that different parts of the organ receive different doses. Cumulative Dose Volume Histogram The cumulative DVH is just the integral of the differential DVH. It can be created if we answer to the question what percentage of the defined volume receives more than an average dose d i, i.e, d d i. Thus, the computer sums the number of voxels that receive at least the given dose and plots this volume as a function of dose. The above sentence can be more coherent from the fact that all cumulative DVHs start at 100% of the volume for zero doses that indicate that all of the volume receives at least no dose. Figure 21 shows an example of a cumulative DVH for a PTV and for an organ at risk. The ideal cumulative DVH for a PTV would be a straight line parallel to the dose-axis that falls sharply to the point d = 100%. In addition, the corresponding ideal case for an organ risk is that the 100% of the volume receives zero total dose. 47
49 Figure 21: (a) The cumulative DVH for a PTV (continuous line) and the ideal cumulative DVH for a PTV that falls sharply to the point d = 100% (dot line). (b) The cumulative DVH for an organ at risk. The corresponding ideal case for an organ risk is that the 100% of the volume receives zero total dose. 1.4 The Radiobiological Basis of Radiotherapy Considerable efforts are being devote at the present time to the improvement of radiotherapy. The development of radiobiology the last years resort in the understanding of how the different tissues respond to radiation [12,66]. Thus an exploitation of radiobiological initiatives will help us in finding the optimized delivered dose for each individual patient [146]. Radiobiology provides a conceptual basis for radiotherapy, identifying mechanisms and processes that underlie the response of tumour and normal tissues to irradiation and which help to explain observed phenomena, like hypoxia, reoxygenation, tumour cell repopulation and mechanisms of repair DNA damage. Radiobiology has been very fruitful in the generation of new ideas and in the identification of potentially exploitable mechanisms. Moreover, radiobiology led to the development of specific new approaches in radiotherapy. Examples are hypoxic cell sensitizers, high-let radiotherapy, accelerated radiotherapy and hyperfractionation. Radiobiology can also help to the choice of schedules for clinical radiotherapy. For instance, conversion formulae for changes in fractionation or dose rate, or advice on whether to use a chemical radiosensitizer at the start, or at the end of a course of radiotherapy. However, the choice of specific protocols is limited by the inadequacy of the theoretical and experimental models: it will always be necessary to rely on clinical trials for the final choice of a protocol. 48
50 1.4.1 Normal and malignant tissue response to radiation The volume response of an uncontrolled tumour is the resultant of two processes: regression and regrowth. If the tumour fails to regrow during the normal lifespan of the patient then we have cure or local control. The response of normal tissues to therapeutic radiation exposure range from those that are life-threatening. The speed at which the response develops varies widely form one tissue to another and depends on the dose of radiation which the tissue receives. Early and late normal tissue responses are differently modified by a change in dose fractionation and this gives rise to the current interest in hyperfractionation [73,135,150] The 5 R s of radiotherapy There are 5 biological factors that influence the response of normal and malignant tissues [147,169]. These five factors modify the response of a tissue to repeated doses of radiation. Two of these factors will tend to make the tissue more resistant to a second dose of radiation: Repair: Damaged cells tend to recover i.e. restore their functions, during the few hours after exposure. Repopulation: Cells that survive irradiation may proliferate during a 5-7 weeks course of radiotherapy and thus increase the number of cells that must be killed. In contrast, the other three factors will tend to make the tissue more sensitive to a second dose of radiation: Redistribution: The Radiosensitivity of cells varies through the cell cycles. There seems to be a general tendency for cells in the S-phase to be the most resistant and for cells in G 2 and mitosis to be the most sensitive. Thus, cells that survive a first dose of radiation will tend to be in the S phase of the cell cycle and within a few hours they may progress into the more sensitive phases G 2 and mitosis. Reoxygenation: In general hypoxic cells tend to be more resistant. Thus cells that survive a first dose of radiation tend to be hypoxic, but thereafter their oxygen supply may improve, leading to an increase in Radiosensitivity. (Intrinsic) Radiosensitivity: The response of a tissue depends on its intrinsic Radiosensitivity, and thus it differs among different tissues. Similarly, some tumours are 49
51 more radioresponsive than others and this is largely due to differences in Radiosensitivity [147] Dose-Response curves When we study the radiation response of a tissue, is very investigate a dose-response curve by varying the radiation dose. An example of a dose-response curve for a target and for normal tissue is shown in figure 22. There are two parameters that characterize the shape of the curve. The first parameter is D 50 that indicates the position of the curve on the dose scale, and thus the sensitivity of the tissue to radiation. The other parameter is γ that shows the steepness of the curve and thus it gives a direct indication of the change in response that will accompany an increase or decrease in radiation dose. Figure 22: (a) An example of a dose-response curve for a target and for normal tissue. As radiation dose is increased, there is always a tendency for tumour response to increase, and the same is true for normal tissue damage (b) Parameter D 50 indicates the position of the curve on the dose scale and parameter γ shows the steepness of the curve. An increase of the radiation dose results not only to an increase of the Tumour Control Probability (TCP) but also to an increase of the Normal Tissue Complication Probability (NTCP) (figure 22). Thus, the sigmoid shape of the NTCP curve is unlikely the same as TCP curve, and since a high frequency of damage is unacceptable, we probably will not wish to determine more than the initial part of the NTCP curve. Treatment plannings aim at finding the 50
52 optimum dose that maximizes TCP and at the same time minimizes NTCP. In addition, we can fix a notional upper limit of tolerance, meaning the upper limit of radiation dose that can be tolerated by the normal tissues. The tumour response for a fixed level of normal tissue damage is called therapeutic index. Therapeutic index caries the notion of cost-benefit analysis 1.6 Radiobiological Models Cell Survival Models Radiotherapy treatment plan evaluation relies on an implicit estimation of the tumor control probability (TCP) and normal tissue complication probability (NTCP) arising from a given dose distribution. A potential application of radiobiological modeling to radiotherapy is the ranking of treatment plans via a more explicit determination of TCP and NTCP values [139]. Although the limited predictive capabilities of current radiobiological models prevent their use as a primary evaluative tool, radiobiological models are a necessary part of radiobiology research, to provide a framework in which to analyze and compare data and ultimately to assist in building up a consistent theory of radiation action both in vitro and in vivo Target Theory Generally target theory, is a theory that predicts response curves based on the number of events required to cause the phenomenon. In biology, there exists an idea that there may be regions of the DNA that are crucial to maintain the reproductive ability of the cells [26,81,133]. In addition, these sensitive regions could be thought of as specific targets for radiation damage so that the survival of a cell after radiation exposure will be related to the number of targets inactivated. There are two interpretations of this idea that have been used commonly [30,56]. The first interpretation of the theory proposes that just one hit by radiation to a single target can give rise to a lethal lesion by itself. Most common underlying biological rationale is that radiation produces a double strand DNA break (DSB) by using this single radiation track. This is called single-target inactivation. The second and more general interpretation of target theory proposes that just one hit by radiation of each of n sensitive targets in the cell is required for 51
53 death of the cell. This interpretation is called multi target inactivation. In both versions, we assume that all the target cells respond identically. Cell survival after irradiation is binomial, but since during irradiation there are a very large number of hits on different cells taking place, but the probability of the next hit occurring in a given cell is very small, Poisson Statistics can be applied [156]. In addition, for the simple case of single target inactivation, the probability of survival is equal to the probability of having zero hits in the cell, and thus: nd D D0 D0 (, ) ( ) ( ) Sdn= e = e = Sdn= SD (1) where D 0 is the radiation resistance (the dose that gives an average of one hit per target and reduces survival from 1 to 0,37), n is the number of fractions, d is the dose per fraction and D is the total dose. D/D 0 is the number of hits per target The Linear Quadratic Model The Linear Quadratic model (LQ Model), is a mechanistic model which describes cell killing, both for tumor control and for normal tissue complications. In this formalism, the yield of lethal lesions is the sum of lethal lesions produced from a single radiation track (which are linearly related to dose, by a factor αd) and lethal lesions produced from two radiation tracks (which are quadratically related to dose, by a factor βd 2 ). Thereby, the survival of target cells after a dose per fraction d is given as: 2 d ad β Sd ( ) = e (2) This is a continuously-bending survival curve with no straight portion at high radiation doses. ad Curves for the individual LQ components and e β d intersect at the dose where the ad and e 2 βd 2 components of cell killing are equal. This intersection happens to occur at a dose equal to the radio a/β. For acutely responding tissues which express their damage within a period of days to weeks after irradiation, the a/β ratio is in the range of 7-20 Gy while, for late responding tissues which express their damage months to years after irradiation, a/β generally ranges from Gy [115,166]. Thus, the shape of the survival curve (figure 23) is determined by the radio a/β [84,171]. A low a/β ratio indicates a rapid increase of total dose with 52
54 decreasing dose per fraction and a survival curve for the putative target cells that is significantly curved. A higher a/β ratio indicates a less rapid increase in total dose with decreasing dose per fraction and a less curved survival response for the target cells. Figure 23: (a) Lethal lesions produced from a single radiation track are linearly related to dose by a factor αd and lethal lesions produced from two radiation tracks are quadratically related to dose, by a factor βd 2 (b) The shape of the survival curve is determined by the radio a/β. A low a/β ratio implies a steep curved, while a higher a/β ratio indicates a less curved survival response. The ratio a/β is usually known, and so the parameters a and β can be expressed as a function of this ratio, under the assumption that the responses for the linear model (eq 1) and the LQ model (eq 2) are the same for the dose per fraction d used in the clinical data: a = β = D 1 D ( d + a β ) (3) d a β If is assumed that full repair occurs between fractions and consequently each successive fraction in a series is equally effective [32,44], the biological effect of n fractions can be expressed as: n 2 E = Ln[ S( d)] = nln[ S( d)] = n( ad + βd ) = ad + βdd (5) (4) 53
55 The quantity E a is called biologically effective dose (BED) and is an approximate quantity by which different radiotherapy fractionations regimens may be intercompared. Equation (5) may be rearranged into the form: E d BED = = D 1 + (6) a a β If the dose per fraction is changed from d 1 d 2 in a fractionated schedule then is required an additional change in total dose (D 1 D 2 ) that is given by the formula: D D 2 d a 1 + β = d + a β 1 2 (7) Relative seriality model To calculate the injury caused by partial irradiation, the organ could be divided into morphological or functional subunits (FSU). In that way, the radiation induced normal tissue complications is described in terms of inactivation of functional subunits [171]. The FSU structure has been described in terms of serial organization, parallel and more general a combination of these two. When defining a functional subunit one should keep in mind that the target cells of an organ is not only the functional cells but the tissue regenerating cells may be even more important [164]. The division of an organ into functional subunits is therefore quite complex if the centers of function and regeneration does not coincide. The functional subunits are arranged structurally to give the functional properties of an organ. A purely serial organ would contain m subunits in series, while a purely parallel organ would contain n subunits in parallel (figure 24). The simplest non trivial second order structure is a nm matrix of parallel and serial subunits. 54
56 Figure 24: Functional subunits are arranged structurally into serial and parallel organs (a) a purely serial organ that contains m subunits in series (b) a purely parallel organ that contains n subunits in parallel (c) the simplest non trivial second order structure is a nm matrix of parallel and serial subunits. The response of such an organ will be the product of the responses for a purely serial and a purely parallel organ [22,85,99]: n m P = 1 (1 Pij ) j= 1 i= 1 (8) Assuming that the sensitivity of each subunit is identical and the absorbed dose distribution is homogenous (that is P ij =P s ): m n or 1 ( ) 1 m n Ps 1 1 P P= 1 (1 P s ) = (9) where P s is the response of a single subunit. For a fraction ab (where 0 α 1, 0 b 1 are the relative fractions of the parallel and serial tissue subunits being irradiated) of the whole organ (figure 24), the corresponding response is: 1 am bn n a Pab = 1 (1 Ps ) = 1 (1 P ) The above formula gives a response that is critically dependent on the geometrical alignment of the organ structures relative to the incident beam. Furthermore, the exact location of the various structures has to be known in relation to the therapy beam. A simpler geometrical structure is achieved under the assumption that b=1. Under this assumption it is sufficient to use a single parameter s, which essentially describe the relative seriality of the tissue (s=m/(nm)=1/n). A relative seriality close to zero (s 0) corresponds to a parallel organ like lung or liver, whereas a relative seriality cloze to unit (s 1) corresponds to a closely serial bn (10) 55
57 organ like esophagus or spinal cord. The response functional P v of a subunit v (v=ab=a) is given by: Pv 1 s v s or for homogenous dose distribution to all functional subunits: = 1 (1 P ) (11) s P = 1 (1 P v ) Now the response of the entire organ can be expressed for a non uniform dose distribution [172] but homogenous cell density, for a fractional volume element Δv equal to one over the number of voxels M of the whole organ. Eq 12 can be rewritten as: 1 v 1 s 1 s (12) M s v P = 1 [1 P( Di ) ] Δ (13) i= 1 Eq 13 gives the response of the entire organ P to a non uniform dose distribution as a function of the response of the whole organ for the dose D i in each compartment i [22,84,85] (figure 25). Figure 25: (a) Organs are represented by 3-D matrices of voxels which have a monotonic relationship with the dose matrix. The dose to each voxel v i is D i. (b) The relative seriality model takes into account the different internal organization of the FSUs in each organ, and thus the different value of parameter s since this affects significantly the response to radiation. Organs with serial infrastructure have small volume dependence since every subunit is vital for organ function. For organs with parallel infrastructure strong volume dependence can be expected since the organ can maintain most of its function even when a large portion of its 56
58 subunits are damaged. From a structural point of view the tumour is a parallel tissue because all clonogenic cells have to be eliminated to control the tumour. Instead the difficulty with the tumour is to know the density and radiation sensitivity of the most resistant clonogenic cells [4,170] Dose Response Models The Probit Model This model uses a normal distribution that is a probability density function: φ μσ 2, 2 ( x μ ) σ ( x) = e (8) σ 2π If the mean value μ and the standard deviation σ have respectively the values: then the response probability P(D) is equal to: D50 μ = D 50 & σ = (14) γ π D 1 x D50 γ γ 50 π D50 PD ( ) e dx = D (15) 50 0 where D 50 is the dose that gives 50% response probability (i.e. P(D 50 )=0.5), γ the normalized dose response gradient [100]: PD ( ) γ D (16) D and γ 50 is the normalized dose response gradient which is defined as: γ D P ( D ) (17) Parameters D 50 and γ are specific for every organ and specific for the kind of injury (endpoint) considered and can be calculated only from clinical data. This model has a sigmoid doseresponse curve (figure 26). 57
59 The Poisson Model This model uses Poisson statistics and assumes that the probability of cell survival S(D) is given by eq 1. In addition the response probability is given by: D NS 0 0 ( D) Ne D 0 PD ( ) = e = e (18) where N 0 is the number of clonogenic cells (or functional subunits for healthy tissue). Solving PD ( ) eq. 18 for D 50 and γ% (where % γ = D% max D ) [27] arises that: D D = D (ln N ln ln 2) (19) ln N % γ = 0 (20) e Sometimes it is helpful to use γ 50 in order to compare with other response models, and so the following transformation can be used [118]: The shape of the dose-response curve is sigmoid (figure 26) γ 50 ln 2 = ( e % γ lnln2 ) (21) P Poisson Binomial Probit D /Gy Figure 26: Dose response curves for Poisson model (blue line), Binomial model (black line) and Probit model (red line). Poisson and Binomial models are represented by the same curve, since Poisson model, is just a special case of the Binomial model. 58
60 The Binomial Model For a smaller number of clonogenic cells, Poisson statistics must be replaced by Binomial Statistics. In addition, assuming that the probability of cell survival S(D) is given by eq 1 the response probability is given by: Solving eq. 22 for D 50 and γ% arises that: D N0 D PD ( ) = (1 SD ( )) = 1 e N0 0 (22) 1 D = D ln N (23) N0 1 1 % γ = ln N0 1 (24) N0 The dose response shape of the Binomial model is also sigmoid (figure 26) 59
61 Materials & Methods 2.1 Study Bases In this study there were included 111 patients with biopsy proven and localized adenocarcinoma of the prostate. The value of pre-treatment PSA was (mean 13.1, median 9.1), the Gleason Score was 3-9 (mean 6.22, median 6.0) and T1-T4. All patients were treated with a proton boost to the prostate alone of 20 Gy given in 5 Gy fractions during four consecutive days, followed by a treatment with x-rays to a dose of 50 Gy in 2 Gy fractions five days per week. The patients were treated with External Beam Radiotherapy (EBRT) at the department of Oncology at Akademiska Sjukhuset, Uppsala, Sweden, between the years The proton treatment was given with the fixed horizontal proton beam at the The Svedberg Laboratory in Uppsala [116]. At the proton treatment the prostate is positioned with high precision, using x-ray imaging of radiopaque gold markers previously implanted in the prostate [36,61]. A new method, where the distance between prostate and rectum is increased during proton therapy, was introduced during November 2005 and due to this fact two subgroups were made according to the treatment time. In particular, in this method a cylindrical rod of Perspex was inserted into the rectum and this device allows the rectum to be retracted posteriorly. In addition the first subgroup (2000 October 2005) consists of 65 patients and the second subgroup (November ) consists of 33 patients. Two additional subgroups were also made for the study of erectile dysfunction after the treatment. The criterion was whether the patient undergone hormonal therapy additional to radiation therapy. Thus the first subgroup consists of 53 patients, that undergone hormonal therapy before radiotherapy, while the second subgroup consists of 36 patients that were treated only with radiation therapy. The study was not anonymous according to the need of following up the patients and in order to relate the symptoms to the treatment techniques, dose and treatment volume. Therefore the patients were coded to allow additional information which is related to the investigated variables. However the study was approved by ethical comity. 60
62 2.2 Hospital records Information concerning the prescribed dose, the fractionation, the treatment period, and the tumour stage were retrieved from the hospital records. To evaluate the dose plans the treatment plans for each patient, both for photon and proton therapy were retrieved. The image data were introduced into Helax-TMS [117] (Treatment Management System, MDS Nordion Therapy System, Uppsala, Sweden) and stored on tapes, which have been restored in order to collect the data needed for this research. The data collected for each treatment were images of the pelvic area both for the treatment of photon and proton, since the targets delineated for proton and photon treatment were not identical. The CT slices have 2mm and 5mm distance from each other, in the case of protons and photons treatment plans respectively. The much smaller distance in slices for proton treatment plan reflects the need for more accurate dose calculation. The whole bulb was delineated in each CT image of each patient, both for protons and photons plans (figure 27, 28). The penile bulb appears as an oval-shaped, hyper intense midline structure [86]. The top of the bulb is located approximately 10 mm inferior to the apex of the prostate gland. Between the apex of the prostate and the penile bulb, there is the urogenital diaphragm. Additionally 10cm of the rectum (± 5cm from the middle of the prostate) were also delineated in each CT image of each patient, both for protons and photons plans (figure 29, 30). This area of the rectum has been chosen because we decided that the whole rectum was 10cm. To confirm the delineation, Oncologist Silvia checked the delineated organs. 61
63 Figure 27: Delineation of penile bulb in CT scanning of proton treatment plans (yellow contour) as it is demonstrated in the treatment planning system Helax-TMS. The rectum is also delineated with orange contour and the PTV with red contour. Blue lines represent the slices, while red line is the selected slice that is also shown in the middle red box. Left and right boxes are the preceding and the sequential slices respectively. 62
64 Figure 28: Delineation of penile bulb in CT scanning of photon treatment plans (yellow contour) as it is demonstrated in the treatment planning system Helax-TMS. The rectum is also delineated with orange contour and the PTV with red contour. Blue lines represent the slices, while red line is the selected slice that is also shown in the middle red box. Left and right boxes are the preceding and the sequential slices respectively. 63
65 Figure 29: Delineation of 10cm of the rectum in CT scanning of photon treatment plans (yellow contour) as it is demonstrated in the treatment planning system Helax-TMS. The prostate (GTV) is also delineated with orange contour and the PTV with red contour. 64
66 Figure 30: Delineation of 10cm of the rectum in CT scanning of proton treatment plans (yellow contour) as it is demonstrated in the treatment planning system Helax-TMS. The prostate (GTV) is also delineated with orange contour and the PTV with red contour. The presence of the cylindrical Perspex rod is obvious inside the rectum. 2.3 Target Volume and Organs at risk Radiotherapist must outline the gross tumour volume (GTV), that is the primary tumour volume with the highest tumour cell density, the clinical target volume (CTV) which includes the GTV, and in addition, a margin containing assumed subclinical i.e. non-detectable, disease. To account for organ movements and errors in patient set-up, a mrgin is applied around the 65
67 GTV creating the planning target volume (PTV). Radiation treatment fields are designed to cover the PTV entirely and deliver a uniform dose distribution to it. The dose delivered to PTV represents the dose delivered to the CTV and ensures that the prescribed dose is delivered to the PTV [76,79]. In the treatment of localized prostate cancer, the CTV is usually equivalent to the GTV. The CTV for the proton treatment was the prostate gland alone. Margins around the GTV must be applied to allow for geometrical uncertainties in its shape and variations in its location relative to the beams due to the organ mobility, organ deformation and patient set-up variations [77,78]. Moreover the margins around the GTV account for microscopic tumour spread a lymph node involvement [77,78]. Thus, to define the PTV for the proton treatment a margin of 5 mm was applied around the GTV in all directions except near the rectum, where a margin of 2 mm was used. In contrast, the CTV for the photon treatment was either the prostate gland alone or the prostate gland plus the seminal vesicles. The seminal vesicles were included if any of the pretreatment conditions PSA>10 ng/ml, Gleason>6 or >T2b were present. The PTV for the case of photon treatment was defined as the CTV with a margin of 15 mm in lateral and ventral direction and 10 mm in cranial, caudal and dorsal direction. The larger PTV margin for photon treatment was motivated by the less accurate positioning of the prostate compared to proton therapy. Radiotherapist must also define the organs at risk (OARs). Those are the organs whose radiation sensitivity and location in the vicinity of the CTV will give some restrictions accordingly to there tolerance dose. Thus, OARs may significantly influence treatment planning and/or the prescripted dose. In the treatment of prostate cancer, the OARs include the urinary bladder, the urethra, the anal-sphincter region, the rectum, sigmoid colon and small bowel, and the penile bulb as well as the nerves and vessels involved in erectile function. 2.4 Treatment technique and dose planning A computerized treatment planning system based on CT scanning allows a slice-by-slice delineation of the region that is to be irradiated. Therefore, each patient was CT scanned in a 66
68 specific treatment position (see section 2.4.1) over the pelvic region with a slice thickness of 2 mm for proton treatment and 5 mm for photon treatment respectively. The CT images were obtained from a Siemens Sensation 16 scanner. Also, all the patients underwent diagnostic MR imaging for guidance in delineation of the prostate gland in the CT-studies used for treatment planning. This is due to the fact that MRI provides a very rich tissue contrast range, and so a very high spatial resolution in the images. Thus, it helps us in the recognition of the different structures. The next step is the delineation of the CTV, the PTV and the organs at risk in the CT images. As it was said before, the targets delineated for proton treatment and photon treatment were not identical. Figure 31: Dose distributions in a transverse section as isodose contours in the CT for the two different treatment plans (a) with and (b) without the rectal retractor. 67
69 Treatment planning was performed on a three-dimensional conformal treatment planning system (TMS; Helax MDS-Nordion, Therapy System, Uppsala, Sweden). The system is provided with absorbed dose calculation algorithms for the different radiation modalities employed and has been subjected to adequate quality assurance tests [3,134,117]. Dose distributions were presented as isodose contours (figure 31) in the CT slices and as dose volume histograms (DVH). The dose distributions for photons and protons were calculated using particle specific pencil beam algorithms [1,68,134]. It must be noticed, that for the new technique in proton therapy, that is the retraction of the rectum in dorsal direction, new treatment plans were made, as now the greater distance between the rectal wall and the prostate release us from the high risk of giving to the rectum greater dose than the excepted. The photon treatment was given with a three field treatment technique (figure 32) using 15 MV photon beams from an Elekta Precise linear accelerator (Elekta AB, UK) equipped with a multileaf collimator (MLC). The prescribed dose was 50Gy, given in 2Gy fractions five days per week. Figure 32: For the photon treatment the method of 3-D conformal radiotherapy was used. The prescribed dose was given with a three field treatment technique. 68
70 Figure 33: The proton treatment was realized with a single perineal proton beam. An additional dose of 20Gy (physical dose) was given with a single 180 MeV perineal proton beam (figure 33) in 5Gy fractions. The range compensation filters were calculated to obtain a distal dose distribution conformal to the PTV with a 10 mm beam margin, allowing for range uncertainties in the variable entrance region (±5 mm), in bolus construction (±2 mm) and accelerator energy (±1 mm) Patient fixation and positioning The positioning of the target in the proton beam can be done with an accuracy of less than ±1 mm by using the method described by Grusell and co-workers [108,109]. Therefore, the patient must be placed in a very well immobilized position. Before fixation, imaging and treatment the patients are prescribed a laxative in order to remove gas from the rectum and so to make rectal volume as small as possible. In proton therapy the patients are lying in gynecological position (i.e. with the legs placed in adjustable leg supports) on a specially constructed fixation couch (figure 34). The couch allows the patients to be tilted in the cranial-caudal direction. The selected couch angle, and thus the irradiated angle follow the direction of the rectum. An error of ±3 is allowed in the couch angle; this error is taken into account in the determination of 69
71 PTV. The fixation couch is provided with a vertical Perspex plate onto which the patient s perineum is placed in contact. Figure 34: The position of the patients in proton therapy. The patients are lying in gynecological position (i.e. with the legs placed in adjustable leg supports) on a specially constructed fixation couch. This vertical Perspex has three uses. First, because of its strict placement the patient has no choice but stay immobilized. Second, it acts as an adjuster of the distal dose fall of to the distal (cranial) end of the prostate. That is because the proton beam passes through the Perspex plate on its way towards the prostate and thereby the distance from the beam entrance to the distal end of the prostate can be determined in a reproducible way. Finally, the vertical Perspex plate has apertures in order to insert the cylindrical Perspex rod into the rectum in a fixed angle (figure 35). Thus the rod is attached to the Perspex plate and positioned horizontally to be parallel with the fixed horizontal proton beam. It must be noticed that the patients were immobilised in the couch with identical rectal retraction both at the CT imaging before treatment and at each proton treatment. For this purpose, the prostate as well as the rectal retractor has 3 radiopaque markers so that its position can be verified by CT-imaging for treatment planning and by X-ray imaging for treatment positioning. Thus, the magnitude of the retraction of the rectum relative to the prostate was determined in the CT- planning study by measuring the position of the markers in the prostate and markers in the rectal retractor. 70
72 Figure 35: The cylindrical Perspex rod insert into the rectum through the vertical Perspex plate. The Perspex rod retracts the rectum in the dorsal direction, and thus increases the distance between the rectum and the prostate. For photon treatment the situation is much simpler. In photon treatment, the patients are positioned in a conventional supine position. MV portal images are used to verify the position by matching to bony structures. The gold markers that were used for proton treatment can not be used to verify the position as the small marker size used for proton therapy made them hard to visualize on a simple MV portal imaging. Therefore, for photon treatments, larger margins to the PTV are used. 2.5 Follow up The follow up time for all the patients was 24 months. This decision was due to the fact that a greater follow up time would exclude the patients that were treated during the year The baseline was considered as the first day that each patient began his treatment. Thus, in the time period of 24 months after the treatment, all the patients had had a PSA test to check for PSA progression. We defined PSA progression the case in which PSA level was increased after the completion of the treatment, even though that level was lower than the initial PSA (before the beginning of the treatment). Moreover, all the patients were given a questionnaire in order to estimate about the grade of their rectal complications, together with their potency. 71
73 2.6 Dose-Volume Histograms The dose (in step of 0.2Gy) in relation with the absolute volume i.e the DVHs were extracted from the treatment planning system for each patient, both for photon and proton treatment plans, for every defined structure (PTV, GTV, bladder, rectum and bulb). The DVHs helped as to have a first view of the dose distribution in the defined structures. The treatment planning system calculated differential DVHs, which were exported in a text file together with other personal and statistical information about the dose and the volume for each patient, for all the defined structures. That additional information was then used for the statistical analysis. The cumulative DVHs were also calculated, by integrating the differential DVHs. 2.7 Methods for evaluation Maximum likelihood fitting of the response model to clinical data The maximum likelihood method is perhaps the most powerful estimation in performing the fitting of the models to experimental data. This method determines the best estimates of the parameters by maximizing the likelihood to reproduce the given pattern of observations. The logarithm of the likelihood function is often used for computational convenience, and so the bigger the log-likelihood value, the better the respective fit. The fitting calculations were performed through the use of the repetitive method Newton-Raphson. A disadvantage of this method is that it finds local maxima, while we are looking for the global maximum of the loglikelihood function. In addition, the calculations were performed by changing both the initial values (starting points) and the allowed range of the parameters. Likelihood function In statistics, the likelihood function is a form of probability, but with the difference that probability allows us to predict unknown outcomes based on known parameters, whereas likelihood allows us to estimate unknown parameters based on unknown outcomes. Thus, a 72
74 likelihood function is a conditional probability function (formalized on Bayes theorem) considered as a function of its second argument with its first argument held fixed [39,122]. Definitely, given a family of r θ real parameters and N stochastic variables (x 1,..,x N ) with probability density functions f( X r r θ ), the likelihood function is by independence: The likelihood function LX ( r r θ ) r r N r LX ( θ ) = f( x i θ ) i= 1 (25) is considered as a function of θ r provided that variables X r are having fixed values observed in the experiment. The Maximum Likelihood Estimate The method of maximum likelihood estimates the parameters r θ by finding the value ˆ θ that maximizes LX ( r r θ ) for particular observations X r. This is the maximum likelihood estimator of θ: r r ˆ θ arg max LX ( θ ) = r θ Since maxima are unaffected by monotone transformations, one can take the logarithm of expression (25) to turn it into a sum: r r Ν r LX ( θ ) = ln f( x i θ ) (27) So the maximum likelihood can know be performed by maximizing equation (27): r r ˆ θ arg max ln LX ( θ ) = r θ It must be noticed that the maximum likelihood estimator may not be unique, or indeed may not even exist. ι= 1 (26) (28) Estimation of confidence intervals using log-likelihood function The likelihood of a normal distribution with variance σ and mean μ, for a set of N observations is given by: 73
75 Or more conveniently: N /2 N 2 r N ( ) x 1 i μ i= LX ( μσ, ) = f( xi μσ, ) = exp 2 (29) 2 i= 1 2πσ 2σ r LX ( μσ, ) exp 2πσ 2σ N /2 N ( x ) ( ) i 1 i x + N x μ = = 2 2 If we consider the case of normal distribution with unιt variance and unknown mean μ, then the r likelihood LX ( μ) has the same bell-shaped form as the normal probability density function. (30) Then the log-likelihood is a parabola in μ: r N 1 2 N LnL( X μ) = ln c ( xi x ) ( μ x ) The maximum likelihood estimator of equation (31) is μ = x. i= 1 (31) One of the properties of normal distribution is that only a very small amount of the area that is delimitated by f ( X r ) is out of the interval( x= μ 2 σ, x= μ+ 2σ). In particular the probability of the variance X r to be found between x = μ 2σ and x = μ + 2σ is In addition, the probability of the variance X r to be found between x = μ σ and x = μ + σ is For the case of σ = 1this is expressed by: P X 2 μ Χ+ 2 = 95.5% (32) P X 1 μ Χ+ 1 = 68.3% (33) In figure 36 we show the parabolic log likelihood after we changed the origin to have ln L = 0 at the maximum, and for the case of N=1. As we can see from figure 36 a line drew at ln L = 2 yields the 95.5% confidence interval for μ, whereas a line drawn at ln L = 0.5 yields the 68.3% confidence interval for μ. Thus the values of μ that change the value of loglikelihood an amount of ±2 and ±0.5 determine the 95% and 68.3% confidence interval respectively. 74
76 Figure 36: The parabolic log likelihood after a change of the origin to have ln the maximum, and for the case of N=1. A line drew at ln L = 2 and at ln yields the 95.5% and 68.3% confidence interval for μ respectively. L = 0 at L = Fractionation correction In the dose-response relation, the calculated radiobiological parameters refer to a certain uniform dose per fraction. Thus, in the case of combined photon and proton radiotherapy, fractionation correction must be applied before deriving these parameters [17,47,107], since the dose per fraction was different for proton and photon treatment plans (2Gy/fr and 5Gy/fr for photon and proton respectively). In this study the fractionation correction was applied using the linear quadratic model. Thus if the dose per fraction is changed from d 1 d 2 in a fractionated schedule then is required an additional change in total dose (D 1 D 2 ) that is given by the formula 7 and so: d1+ a d a 1+ β β D2 = D 1 = nd 1 1 d a a 2 + d β 2 + β In our case we considered that the dose per fraction for proton schedule is changed from 5 Gy / fr 2 Gy / fr (in order to be the same as photon schedule). Fractionation correction must also be applied in photon schedule. Although the dose per fraction is fixed at 2Gy/fr, that dose corresponds to the prescribed dose in the PTV. 75
77 Consequently, the mean dose per fraction for the normal tissues will vary in an interval between 0 d 2Gy and thus the dose per fraction must be changed to a uniform dose of 2Gy/fr ROC Curves The diagnostic performance or the accuracy of a test to discriminate diseased cases from normal cases is evaluated using Receiver Operating Characteristic (ROC) curve analysis [114,180]. ROC curves can also be used to compare the diagnostic performance of two or more laboratory or diagnostic tests [60]. When we want to separate the results of a particular test in two populations, one population with a disease and the other population without the disease, we have to consider a specific cut-off point or criterion value to discriminate between the two populations. However, there will be some cases that the disease will correctly classified as positive (TP = True Positive fraction), but some cases that the disease will be classified negative (FN = False Negative fraction). On the other hand, some cases without the disease will be correctly classified as negative (TN = True Negative fraction), but some cases without the disease will be classified as positive (FP = False Positive fraction). Thus we will rarely observe a perfect separation between the two groups. Indeed, the distribution of the test results will overlap (figure 37). The position of the cut-off point will determine the number of TP, TN, FP and FN. Figure 37: When we consider the results of a particular test in two populations, one population with a disease, the other population without the disease, we observe that the distribution of the test results will overlap. Thus we determine True Positive fraction (TP), False Positive fraction (FP), True Negative fraction (TN) and False Negative fraction (FN). The position of the cut-off point will determine the number of TP, TN, FP and FN. 76
78 Two definitions are very important; sensitivity and specificity. Sensitivity is the probability that a test result will be positive when the disease is present (True Positive Rate-TPR, expressed as a percentage). Specificity is the probability that a test result will be negative when the disease is not present (True Negative Rate-TNR, also expressed as a percentage). In a ROC curve the True Positive Rate is plotted as a function of the False Positive Rate (FPR=1-Specificity) for different cut-off points. In addition, each point on the ROC plot represents a sensitivity/specificity pair corresponding to a particular decision threshold (figure 38). Figure 38: An example of ROC curves where the True Positive Rate is plotted as a function of the False Positive Rate (FPR=1-Specificity) for different cut-off points. The Area under the ROC curve (AUC) measures the accuracy of the test. An area of 1 represents a perfect test whereas an area of 0.5 represents a worthless test. The accuracy of the test is measured by the Area under the ROC curve (AUC). An area of 1 represents a perfect test whereas an area of 0.5 represents a worthless test. Thus, a test with perfect discrimination (no overlap in the two distributions) has a ROC plot that passes through the upper left corner (100% sensitivity, 100% specificity). The closer the ROC plot is to the upper left corner, the higher the overall accuracy of the test [60]. 77
79 Results The clinical characteristics of the patients that were used for the study are illustrated in the table 4. From the 100 patients that were used for the study, 94 didn t appear PSA progression (responders) within the follow up time, while only 6 of them appeared PSA progression (nonresponders). All of the non-responders had prostate cancer at the stage T3 of the TNM system. The mean value of the Gleason score and the mean value of the initial PSA were 6 Vs 9 and 8.9 Vs 18.5 for the responders and non responders respectively. The mean volume of the prostate was greater for the non-responders comparatively with the responders (66.99mm 3 Vs 59.00mm 3 ). None of the non-responders was treated with the additional use of the cylindrical Perspex rod. It is also interesting to notice that from the patients that had hormonal therapy, only 6 of them (10.3%) appeared PSA progression. From the 98 patients that were used for the rectal analysis, 65 didn t appear rectal complications, while 33 of them had rectal complications within the follow up time. At total 33 patients were treated with the additional use of the cylindrical Perspex rod (33.7%) and 65 patients without the rod (66.3%). From the 33 patients that were treated with the Perspex rod, only 9 of them appeared rectal complications (27.3%). From the rest 65 patients that were treated without the Perspex rod 24 patients appeared rectal complications (36.9%). From the 89 that were used for the penile bulb analysis, 64 didn t have symptoms of sexual dysfunction (71.9%) while 25 had sexual dysfunction (28.1%) within the follow up time. At total 53 patients received hormonal therapy (30.2%). From the 53 patients that received hormonal therapy, 16 appeared sexual dysfunction (30.2%). From the rest 36 patients that didn t receive hormonal therapy, 9 patients appeared sexual dysfunction (25.0%). 78
80 Clinical Characteristics Total PSA Progression No PSA Progressio Number of patients (6.0%) 94 (94.0%) Age at treatment Range Median Mean TNM T0 T1 T2 T3 T4 Gleason Score Range Median Mean Initial PSA Range Median Mean (1.0%) 39 (39.0%) 33 (33.0%) 24 (24.0%) 3 (3.0%) (0.0%) 0 (0.0%) 0 (0.0%) 6 (100.0%) 0 (0.0%) (1.0%) 39 (41.5%) 33 (35.1%) 18 (19.1%) 3 (3.2%) Hormonal Therapy 58 6 (10.30%) 52 (89.7%) Perspex Rod 34 0 (0.0%) 34 (100%) Mean Dose in Prostate Photon Dose (Gy)(SD) Mean Max Min Proton Dose (Gy) (SD) Mean Max Min Total Dose in Prostate from photons and protons (Gy) (SD) Mean Volume of prostate mm 3 (SD) (0.30) (0.45) (0.59) (0.16) (0.37) (4.34) (0.31) (0.44) (0.59) (0.15) (0.23) 9.99 (3.94) (0.26) (0.59) (0.43) (0.32) (1.25) (7.26) (6.81) (9.95) (6.22) (27.20) (31.40) (27.03) Table 4 (a): The clinical characteristics of the study population. The population is spitted into those who appeared or not PSA progression. 79
81 Clinical Characteristics Total Rectal Complicatio No Rectal Complications Number of patients (33.7%) 65 (66.3%) Age at treatment Range Median Mean Hormonal Therapy (28.1%) 41 (71.9%) Mean Dose in Rectum Photon Dose (Gy) (SD) Mean Max Proton Dose (Gy) (SD) Mean Max Total Dose: Photons + protons (Gy) (SD) (5.15) (0.64) 1.11 (0.47) (1.97) (6.13) (0.17) 1.19 (0.53) (1.61) (4.47) (0.06) 1.07 (0.47) (2.14) (6.42) (6.28) (6.33) With Perspex Rod 33 9 (27.3%) 24 (72.7%) Mean Dose in Rectum Photon Dose (Gy) (SD) Mean Max Proton Dose (Gy) (SD) Mean Max Total Dose: Photons + protons (Gy) (SD) (4.66) (0.67) 0.86 (0.34) (2.36) (5.14) (0.78) 0.95 (0.41) (1.33) (4.43) (0.64) 0.82 (0.31) (2.65) Without Perspex Rod (36.9%) 41 (63.1%) Mean Dose in Rectum Photon Dose (Gy) (SD) Mean Max Proton Dose (Gy) (SD) Mean Max Total Dose: photons + protons (Gy) (SD) (5.26) (0.63) 1.23 (0.51) (1.46) (4.54) (0.58) 1.27 (0.49) (1.48) (5.73) (0.70) 1.21 (0.55) (1.42) (6.88) (7.11) (6.15) Table 4 (b): The clinical characteristics of the study population. The population is spitted into those who had or not rectal complications. 80
82 Clinical Characteristics Total Sexual Dysfunction No Sexual Dysfunction Number of patients (28.1%) 64 (71.9%) Age at treatment Range Median Mean Perspex Rod 32 4 (12.5%) 28 (87.5%) Mean Dose in Penile Bulb Photon Dose (Gy) Mean Max Proton Dose (Gy) Mean Max Total Dose: Photons + protons (Gy) 30.1 (13.55) (8.14) (0.58) (1.01) (13.84) (7.82) (1.65) (1.05) (12.52) (8.74) (1.14) (0.93) (14.60) (13.20) (15.20) Hormonal Therapy (30.2%) 37 (69.8%) Mean Dose in Penile Bulb Photon Dose (Gy) Mean Max Proton Dose (Gy) Mean Max Total Dose: Photons + protons (Gy) (13.49) (6.54) (1.33) (1.00) (13.91) (4.25) (1.42) (0.86) (13.48) (7.36) (1.29) (0.99) (14.83) (12.99) (15.70) No Hormonal Therapy 36 9 (25.0%) 27 (75.0%) Mean Dose in Penile Bulb Photon Dose (Gy) Mean Max Proton Dose (Gy) Mean Max (12.95) (9.85) (1.87) (1.04) (12.98) (10.73) (1.99) (1.07) (13.14) (6.82) (1.55) (0.93) Total Dose: Photons + protons (Gy) (13.73) (14.32) (13.58) Table 4 (c): The clinical characteristics of the study population. The population is spitted into those who had or not sexual dysfunction. 81
83 3.1 Dose Distributions All the cumulative DVHs that were collected both from the photon and proton treatment plans are presented here. This section will deal with the analysis of the DVHs from the prostate, the rectum and the penile bulb. In the case of rectal analysis, we will also deal with two subgroups considering the use of Perspex rod Prostate Adenocarcinoma Figure 39 shows the mean cumulative DVH for the prostate both for proton and photon treatment. Figures 40 (a) and (b) show the mean cumulative DVH for the prostate for the responders and non responders for photon and proton dose respectively. As it is illustrated in the table 4 (a) the mean photon dose in the prostate was 50.11Gy (SD=0.30), and the mean proton dose was 19.96Gy (SD=0.16). The mean photon dose in the prostate was 50.10Gy (SD=0.26) and 50.24Gy (SD=0.31) for the responders and non responders respectively. Moreover, the mean proton dose in the prostate was 19.97Gy (SD=0.32) and 19.83Gy (SD=0.15) for the responders and non responders respectively. 120 Mean DVH for prostate 100 % Volume % Dose photon proton Figure 39: The mean cumulative DVH for the prostate adenocarcinoma for photon and proton treatment for all the patients. 82
84 Prostate-Photon Dose % Volume PSA progression % Dose no PSA progression Figure 40 (a): The mean cumulative DVH for the prostate for photon treatment for the patients who appeared or not PSA progression. 120 Prostate-Proton Dose % Volume % Dose no PSA progression PSA progression Figure 40 (b): The mean cumulative DVH for the prostate for proton treatment for the patients who appeared or not PSA progression. 83
85 3.1.2 Rectum Figure 41 shows the mean cumulative DVH for the rectum for all the patients both for proton and photon treatment. Figures 42 (a) and (b) show the mean cumulative DVH for the rectum for the responders and non responders for photon and proton dose respectively. As it is illustrated in table 4(b) the mean photon dose in the rectum was 29.91Gy (SD=5.15) and the mean proton dose in rectum was 1.11Gy (SD=0.47). In addition, the mean photon dose in the rectum was 31.26Gy (SD=6.13) and 29.23Gy (SD=4.47) for the responders and non responders respectively. The mean proton dose in the rectum was 1.19Gy (SD=0.53) and 1.07Gy (0.47) for the responders and non responders respectively. 120 Rectum mean DVH % Volume % Dose proton photon Figure 41: The mean cumulative DVH for the rectum for photon and proton treatment for all the patients. 84
86 120 Rectum- Photon Dose % Volume % Dose rectal complications no rectal complications Figure 42 (a): The mean cumulative DVH for the rectum for photon treatment for the patients who appeared or not rectal complications. 120 Rectum- Proton Dose % Volume % Dose rectal complications no retal complications Figure 42 (b): The mean cumulative DVH for the rectum for proton treatment for the patients who appeared or not rectal complications. 85
87 Subgroups concerning the use of Perspex rod. Figure 43 shows the mean cumulative DVH for the patients that were treated with or without the use of the cylindrical Perspex rod during proton therapy. As it is illustrated in table 4 (b) the mean proton dose in the rectum was 0.86 Gy (SD=0.34) and 1.23 Gy (SD=0.51) with and without the use of rod respectively. Figures 44 (a) and (b) show the mean cumulative DVH for the responders and non responders for the two subgroups. The mean proton dose in the rectum for the case that the cylindrical rod was used was 0.95 Gy (SD=0.41) and 0.82 Gy (SD=0.31) for the responders and non responders respectively. In addition, the mean proton dose in the rectum for the case that no rod was used was 1.27 Gy (SD=0.49) and 1.21 Gy (SD=0.55) for the responders and non responders respectively 120 Rectum- subgroups % Volume % Dose 120 with rod without rod Figure 43: The mean cumulative DVH for the rectum for the two subgroups: with or without the use of cylindrical Perspex rod during proton therapy. 86
88 Rectum - without rod % Volume % Dose 120 rectal complications no rectal complications Figure 44 (a): The mean cumulative DVH for the rectum for proton treatment, for the case that no rod was used, for the patients who appeared or not rectal complications. 120 Rectum - with rod % Volume % Dose rectal complications no rectal complications 120 Figure 44 (b): The mean cumulative DVH for the rectum for proton treatment, for the case that the cylindrical Perspex rod was used, for the patients who appeared or not rectal complications. 87
89 3.1.3 Penile Bulb Figure 45 shows the mean cumulative DVH for the penile bulb for all the patients both for proton and photon treatment. As it is illustrated in table 4 the mean photon dose in the penile bulb was Gy (SD=13.55) and the mean proton dose in penile bulb was Gy (SD=0.58). Figures 46 (a) and (b) show the mean cumulative DVH for the penile bulb for the responders and non responders for photon and proton dose respectively. As it is illustrated in table 4 (c) the mean photon dose in the penile bulb was Gy (SD=13.84) and Gy (SD=1.65) for the responders and non responders respectively. The mean proton dose in the penile bulb was Gy (SD=12.52) and Gy (1.14) for the responders and non responders respectively. 120 Bulb mean DVH % Volume % Dose 120 protons photons Figure 45: The mean cumulative DVH for the penile bulb for photon and proton treatment for all the patients. 88
90 Penile Bulb-Proton Dose % Volume % Dose no sexual dysfunction sexual dysfunction 120 Figure 46 (a): The mean cumulative DVH for the penile bulb for photon treatment for the patients who appeared or not sexual dysfunction. Penile bulb-photon Dose % Volume % Dose 120 sexual dysfunction no sexual dysfunction Figure 46 (b): The mean cumulative DVH for the penile bulb for proton treatment for the patients who appeared or not sexual dysfunction. 89
91 3.2 Summation of photon & proton doses To combine photon and proton radiotherapy, fractionation correction must be first applied since the dose per fraction was different for proton and photon treatment plans (2Gy/fr and 5Gy/fr for photon and proton respectively). In this study the fractionation correction was applied using the linear quadratic model. The Relative Biological Effectiveness (RBE) was considered equal to 1 for photon beams and equal to 1.1 for proton beams. The decision of the summation of the different doses was based upon the value of the Coefficient Variance (CV). Coefficient Variance is a normalized measure of the dispersion, and thus a relatively small CV indicates a relatively small dispersion. Figure 47: Plots of the Log Likelihood as a function of the ratio a/b (a) for the prostate (b) for the rectum and (c) for the penile bulb. The value of CV was greater for protons than for photon doses, both for prostate adenocarcinoma and for normal tissues (rectum and penile bulb). This allows as to consider the 90
92 photon dose as a background and thus to use the mean photon dose for each patient. In addition, we decided to use the minimum proton dose in prostate adenocarcinoma and the maximum proton dose for the normal tissues. The choice of minimum dose for the prostate reflects its parallel nature. To summarize, the total doses from photon and proton beams were: Prostate: Mean Dose from Photons + Minimum Dose from Protons (a/b=8.2). Rectum: Mean Dose from Photons + Maximum Dose from Protons (a/b=6.2). Penile Bulb: Mean Dose from Photons + Maximum Dose from Protons (a/b=10). For each case, we utilized the value of a/b ratio that maximizes the Log-Likelihood function (figure 47). 3.3 Dose-response parameters The best estimates of the parameters D 50 and γ of the Binomial, Poisson and Probit models were determined for the prostate adenocarcinoma, the rectum and the penile bulb, by applying a maximum likelihood fitting. D 50 is the dose that gives 50% complication probability and γ is the maximum normalized gradient of the dose-response curve. The best estimates ( ˆP ) of the parameters with their 68% Confidence Intervals (68% CI) are illustrated in table 5. Figures 48 (a)-(e) show the dose-response curves for the prostate adenocarcinoma, the rectum, the rectum with and without the rod and the penile bulb. Prostate adenocarcinoma Parameters Poisson Binomial Probit ˆP 68% CI ˆP 68% CI ˆP 68% CI D 50 (Gy) γ Rectum Parameters Poisson Binomial Probit ˆP 68% CI ˆP 68% CI ˆP 68% CI D 50 (Gy) γ
93 Rectum with rod Parameters Poisson Binomial Probit ˆP 68% CI ˆP 68% CI ˆP 68% CI D 50 (Gy) γ Rectum without rod Parameters Poisson Binomial Probit ˆP 68% CI ˆP 68% CI ˆP 68% CI D 50 (Gy) γ Penile Bulb Parameters Poisson Binomial Probit ˆP 68% CI ˆP 68% CI ˆP 68% CI D 50 (Gy) γ Table 5: The best estimates of the parameters D 50 and γ of the Binomial, Poisson and Probit models for the prostate adenocarcinoma, the rectum and the penile bulb, that were resulted by applying a maximum likelihood fitting. D 50 is the dose that gives 50% complication probability and γ is the maximum normalized gradient of the dose-response curve. 1.0 Prostate Adenocarcinoma P Poisson Binomial Probit outcome Dose (Gy) Figure 48 (a): The dose-response curve for the prostate adenocarcinoma for all the patients of the study. 92
94 1 Rectum P Poisson Binomial Probit outcome Dose (Gy) Figure 48(b): The dose-response curve for the rectum for all the patients of the study Rectum with rod Poisson Binomial Probit outcome P Dose (Gy) Figure 48 (c): The dose-response curve for the rectum for the patients that were treated with the use of the cylindrical Perspex rod during proton treatment 93
95 Rectum without rod P Poisson Binomial Probit outcome Dose (Gy) Figure 48 (d): The dose-response curve for the rectum for the patients that were treated without the use of the cylindrical Perspex rod during proton treatment Penile Bulb 0.8 P Poisson Binomial Probit outcome Dose (Gy) Figure 48 (e): The dose-response curve for the penile bulb for all the patients of the study. 94
96 3.4 ROC curves The ROC curves for the responders and non responders are shown in figure 49. The area under the ROC curve (AUC) is 0.71 when the endpoint is PSA progression, 0.60 when the endpoint is rectal complications and 0.52 when the endpoint is sexual dysfunction (table 6). Clinical endpoint AUC PSA progression 0.71 Rectal complications 0.60 Sexual dysfunction 0.52 Table 6: The AUC for the different endpoints. Figure 49: The receiver operating characteristic (ROC) curve for the endpoint of (a) PSA progression (b) rectal complications and (c) sexual dysfunction. 95
97 Discussion 4.1 Dose distributions As we can see from figures 39, 40 (a) and (b) the corresponding DVHs of photon and proton dose distributions within the prostate adenocarcinoma are virtually the same. The DVHs are almost ideal; both treatments managed to give to the target approximately the 100% of the prescribed dose. Thus there is no significant difference between the responders and non responders neither for photon nor for proton treatment. Figure 41 shows a substantial difference between the dose distribution of protons and photons within the rectum. This is mainly due to the fact that the dose distribution of protons has a sharp fall-off after their maximum range, which is adjusted to the prostate, and thus the dose to the rectum is significantly reduced. This fact is the reason for the choice of combined proton and photon treatment. With the proton boost we manage to give an additional dose to the target without increasing significant the dose to the surrounded normal tissues. As we can also see from figures 42 (a) and (b) the DVHs of photon dose distribution for patients that appeared or not rectal complications show a small difference, with the patients that had rectal complications receiving more dose. On the contrary, there is no difference between the DVHs of proton distribution for patients that appeared or not rectal complications. We can thus say that rectal complications were mainly due to the photon treatment. As it is illustrated in figure 43, the use of the cylindrical Perspex rod during proton treatment reduces the dose within the rectum. In addition, figures 44 (a) and (b) show that in the case where no Perspex rod was used, there is a small correlation between the dose and rectal complications. On the contrary, in the case where the Perspex rod was used, there is no correlation between the dose and rectal complications. In this case other factors except from dose, such as the intrapatient Radiosensitivity and the small number of patients included in this group, insert an uncertainty. 96
98 Figure 45 shows the DVHs of photon and proton dose distributions within the penile bulb. It is obvious that proton treatment gives a much higher dose to the penile bulb in perspective with photon treatment. This is due to the anatomical position of the penile bulb, and the fact that proton boost is given by a single perineal beam. This fact is also an explanation of the choice of combined photon and proton treatment. If only proton treatment was used, the dose to the penile bulb (that is almost the same as the dose to the prostate) would be higher than the allowable. Figure 46 (a) and (b) show that there is only a small correlation between the dose and sexual dysfunction, both for proton and photon distributions. This means that there may be other factors except form dose that are responsible for impotency. For example the age of the patients is an important factor, as their age, or the sequences of their age may by the reason for their impotency. 4.2 Dose response parameters The results of the parameters D 50 and γ of the different models for the prostate adenocarcinoma, the rectum and the penile bulb are shown in Table 5. The estimated values of the parameter D 50 for the prostate adenocarcinoma are Gy (CI = Gy) for the Poisson and Binomial models and Gy (CI = Gy) for the Probit model. Those values are lower than the prescribed dose whose value is 70 Gy. Thus, the delivered dose to the prostate adenocarcinoma, it seems sufficient enough to control the tumour. The estimated values of the parameter γ, that shows the steepness of the curve, are 2.25 (CI = ) for the Poisson and Binomial models and 1.31 (CI = ) for the Probit model. Although the estimated values of γ are decent enough, usually tumours have higher γ values (γ 4) and this reflect the fact that the received dose is homogeneously distributed in the tumours. The estimated values of the parameter D 50 for the rectum are Gy (CI = Gy) for the Poisson model, Gy (CI = Gy) for the Binomial model and Gy (CI = Gy) for the Probit model. Theses values are closed to the prescribed dose (70 Gy), and that is because rectum is located close to the tumour. In addition, the technique 97
99 that was used with photon beams was not conformal enough, and thus most of the dose to the rectum is due to photon therapy. The corresponding values of parameter γ are 0.81(CI = ) for Poisson model, 0.78 (CI = ) for Binomial model and 0.97 (CI = ) for Probit model. That means that the dose-response curve of the rectum is not so steep, and this is generally true for normal tissues. The fact that the use of the cylindrical Perspex rod retracts the rectum form the tumour and thus reduces rectal complications is indicated by the estimated values of the parameter D 50 for the subgroup that the rod was used. The values of D 50 for this subgroup are Gy (CI = Gy) for the Poisson model, Gy (CI = Gy) for the Binomial model, and Gy (CI = Gy) for the Probit model. These values are greater than the prescribed dose (70 Gy), and this is because now we deal with a normal tissue that wasn t so close to the tumour. The estimated values of parameter γ are 1.12 (CI = ) for Poisson model, 1.09 (CI = ) for Binomial model and 1.21 (CI = ) for Probit model. General, normal tissues correspond to the lower part of the dose response curve and due to this fact there is an uncertainty about the rest of the curve, based on the lack of data. In this case the used model is very important. The estimated values of the parameter D 50 for the subgroup that no rod was used within the rectum are Gy (CI = Gy) for the Poisson model, Gy (CI = Gy) for the Binomial model and Gy (CI = Gy) for the Probit model. These values are lower than the corresponding values for the rectum when all patients were used, and this indicates that without the use of rod, the appearance of rectal complications (i.e. the NTCP) has a greater probability. The values of the parameter γ are1.20 (CI = ) for the Poisson model, 1.20 (CI = ) for Binomial model and 1.49 (CI = ) for Probit model. These values show again that the dose-response curve for this subgroup is not so steep. As it seems from the estimated values of the parameter D 50 for the case of penile bulb, there is no correlation between the dose and sexual dysfunction. The estimated values are Gy (CI = Gy) for the Poisson model, Gy (CI = Gy) for the Binomial model and Gy (CI = Gy) for the Probit model. These values 98
100 are very large, and if they were true, none of the patients would appear sexual dysfunction. In addition, the curves have no steepness, as it seems from the estimated values of the parameter γ that is 0.27 (CI = ) for Poison model, 0.30 (CI = ) for Binomial model and 0.31 (CI = ) for Probit model. 4.3 ROC curves The Area under the ROC curve measures the accuracy of the test, and in the particular case it is a measure of how well the test separates the patients that responded to the therapy, from the patients that didn t respond. From table 6 we can see that the patients that appeared or not PSA progression are well separated. The patients that appeared rectal complications are hardly separated, while the patients that appeared sexual dysfunction are not separated. These results are in agreement with the results that came up from the model analysis. 4.4 Uncertainties The most important uncertainty in this study concerns the summation of photon and proton doses. The best way to sum the doses would be a pixel-by-pixel summation of the 3D distributions. However, that could not be done, because the treatment position of the patients during photon therapy differs from the corresponding treatment position during proton therapy. In addition the anatomical position of the organs is different for each case. Nevertheless the hot-spots are approximately the same in the two treatments and so we simplified the summation. Another uncertainty is the fact that we considered the Relative Biological Effectiveness for protons constant and equal to 1.1. Studies showed that RBE tends to increase with depth of the beam [18] and varies between 1.1 and 1.6 at the end of protons range [42,49], and thus the total calculated dose may be lower than the real one. An important limitation in the current study is the uncertainty regarding how the organs were defined, i.e. the delineation of the rectum and the penile bulb. The CT scans has a relatively low space resolution, and that makes the separation of the tissues difficult, especially in the 99
101 case of penile bulb. Delineation was more accurate in proton treatment plans, where the slice distance in the CT scanning was 2mm while in photon treatment plans the corresponding slice distance was 5mm. It is obvious that in the case of proton treatment plans, it was easier to follow the organ. Another issue is that despite the fact that rectum is a tube, the whole volume of rectum has been included in the delineation. However, this does not insert big uncertainties to our results. Another important factor is that in the delineation of rectum and bulb we didn t take into account the probability of patient or organ movement, as we did with the tumour, and this adds an uncertainty in the definition of the organs. Set-up errors may be another possible uncertainty. The risk for the development of rectal complications and sexual dysfunction is not only influenced by radiation therapy but also by surgery and hormonal therapy. Therefore the fact that the collection of clinical data has not yet been completed introduces an additional uncertainty. 100
102 Conclusions The estimated parameters are able to describe the dose-response relation of prostate adenocarcinoma and rectum. Since there are very limited data on the clinical effectiveness of radiation therapy using combinations of photon and proton beams, the determined parameters can be useful in estimating the expected clinical outcome during treatment planning and perform optimization of the delivered treatment. Thus, we can achieve high Tumour Control Probability by justifying the dose distribution in the prostate adenocarcinoma as well as low Normal Tissue Complication Probability by justifying the dose distribution in the rectum. The small differences between the dose distributions of the patients that appeared or not sexual dysfunction, along with the ineffectual estimation of the dose-response parameters D 50 and γ, indicate that there are other factors except from dose, which influence the appearance of sexual dysfunction after combined photon and proton radiation therapy of prostate cancer. In addition, future studies must consider other factors apart from dose in the penile bulb, in order to describe erectile dysfunction after radiotherapy of prostate cancer. The choice of combined photon and proton radiotherapy in the treatment of prostate cancer seems to be very effective, since we can obtain the desirable dose to the tumour, without increasing the dose to the adjacent normal tissues. Moreover, the additional use of the cylindrical rod in the proton treatment appears to reduce the dose within the rectum and thus reduce rectal side effects. 101
103 Acknowledgments 102
104 References [1] Adolfsson J, SteineckG, Whitmore WF Jr. Recent results of management of palpable clinically localized prostate cancer. Cancer (1993);72(2): [2] Adolfsson J. Helgason AR, Dickman P, Steineck G. Urinary and bowel symptoms in men with and without prostate cancer: results from an observational study in the Stockholm area. Eur. Urol. (1998); 33(1): [3] Ahnesjo A, Saxner M, Trepp A. A pencil beam model for photon dose calculation. Med. Phys. (1992); 19: [4] Ågren, A., Brahme A. and Turesson I. Optimization of uncomplicated control for head and neck tumors. Int. J. Radiat. Oncol. Biol. Phys. (1990); 19: [5] Akimoto T., Muramatsu H., Takahashi M. et al. Rectal bleeding after hypofractionated radiotherapy for prostate cancer: correlation between clinical and dosimetric parameters and the incidence of grade 2 or worse rectal bleeding. Int. J. Radiat. Oncol, (2004); 60(4): [6] Al Abany M, Steineck G, Ågren Cronqvist AK, Helgason AR. Improving the preservation of erectile function after external beam radiation therapy for prostate cancer, Radiat. Oncol. (2000); 57: [7] Al-abany M, Helgason AR, Ågren Cronqvist AK, et al. Long term symptoms after external beam radiotherapy for prostate cancer with three or four fields. Acta. Oncol. (2002); 41: [8] Anseline PF, Lavery IC, Fazio VW, et al. Radiation injury of the rectum. Ann Surg. (1981); 194: [9] Ash D, Flynn A, Battermann J, de Reijke T, Lavagnini P, Blank L. ESTRO/EAU/EORTC recommendations on permanent seed implantation for localized prostate cancer. Radiot. Oncol. (2000); 57(3): [10] Aus G, Abbou CC, Heidenreich A, Schmid HP, van Poppel H, Wolff JM et al. EAU guidelines on prostate cancer. Eur Urol. (2003); 40(2): [11] Austin-Seymour M, Munzenrider JE, Goitein M et al. Progress in low-let heavy particle therapy: intracranial and paracranial tumours and uveal melanomas. Radiat. Res. Suppl. (1985); 8:S [12] Awwad HK. Radiation Oncology: Radiobiological and Physiological Perspectives. Kluwer Academic Peblishers (1990); Dordrecht, The Netherlands. [13] Babb RR. Radiation proctitis: a review. Am J Gastroenterol. (1996); 91(7): [14] Bacon CG, Giovannucci E, Testa M Kawachi I. The impact of cancer treatment on quality of life outcomes for patient with localized prostate cacer. J Urol (2001); 166(5): [15] Bacon CG, Giovannucci E, Testa M, Glass TA, Kawaski I. The association of treatment-related symptoms with quality of life outcomes for localized carcinoma patients. Cancer (2002); 94(3): [16] Banker FL. The preservation of potency after external beam irradiation for prostate cancer. Int J Radiat. Oncol Biol. Phys. 1(1988);5(1): [17] Barendesen G.W. Dose fractionation, dose rate and iso-effect relationships for normal tissue responses. Int. J. Radiat. Biol. Phys. (1982); 11: [18] Blomquist E, Russel KE, Stenerlöw B, et al. Relative biological effectiveness of intermediate energy protons. Comparison with 60Co gamma-irradiation using two cell lines. Radiother. Oncol. (1993); 98: [19] Boring C.C, Squires T.S, Tong T. et al. Cancer Statistics. CA-Cancer J. Clin. (1994) ;44:7-26. [20] Bortfeld T, Burkelbach J, Boesecke R, Schlegel W. Methods of image reconstruction from projections applied to conformation radiotherapy. Phys. Med. Biol. (1990); 35: [21] Brahme A, Roos JE, Lax I. Solution of an integral equation encountered in rotation therapy. Phys. Med. Biol. (1982); 27: [22] Brahme A, Ågren A-K. Optimal dose distribution foe eradication of heterogeneous tumour Acta Oncol. (1987); 26:
105 [23] Brahme A. Optimization of stationary and moving beam radiation therapy techniques. Radio. Oncol. (1988); 12:12940 [24] Bragg W. Studies in radioactivity. London, [25] Carter BS, Bova GS, Beaty TH, Steinberg GD, Childs B, Isaacs WB et al. Hereditary prostate cancer: epidemiology and clinical features. J Urol (1993); 150(3): [26] Chadwick KH and Leenhouts HP. A molecular theory of cell curvival. Phys Med Biol (1973); 13: [27] Chen CT, Valicenti RK, Lu J, Derose T, Dicker AP, Strup SE et al. Does hormonal therapy influence sexual function in men receiving 3D conformal radiation therapy for prostate cancer? Int J Radiat. Oncol Biol. Phys (2001); 50(3): [28] Coia LR, Myerson RJ, Tepper JE. Late effects of radiation therapy on the gastrointestinal tract. Int J Radiat. Oncol Biol. Phys. (1995); 31(5): [29] Cox JD, Stetz J, Pajak TF. Toxicity criteria of the Radiation Therapy Oncology Group (RTOG) and the European Organization of Research and Treatment of Cancer (EORTG). Int J Radiat. Oncol Biol. Phys. (1995); 31(5): [30] Curtis SB. Lethal and potentially lethal lesions induced by radiation: A unified repair model. Radiat. Res. (1986); 106: [31] D Amico AV, Whittinton R, Malkowicz SB et al. Biochemical outcome after radical prostatectomy, external beam radiotherapy, or interstitial radiation therapy for clinically localized prostate cancer. JAMA (1998); 280: [32] Dale RG. The application of the linear quadratic dose effect equation to fractionated and protracted radiotherapy. Br J Radiol. (1985); 58: [33] Davis JW, Kuban DA, Lynch DF, Schellhammer PF. Quality of life after prostate cancer: differences based on treatment mordality. J Urol (2001); 166(3): [34] De Koning HJ, Auvinen A, Berenguer SA, Calais ds, Ciatto S, Denis L et al. Large-scale randomized prostate cancer screening trials: program performances in the European Randomized Screening for prostate cancer trial and the Prostate, Lung, Colorectal and Ovary cancer trial. Int J Cancer (2002); 97(2): [35] Dearnaley DP, Khoo VS, Norman R, Meyer L, Naum A, Tait D et al. (1999) Comparison of radiation side-effects of conformal and conventional radiotherapy in prostate cancer: a randomized trial. Lancet 353(9149): [36] Dehnad H, Nederveen AJ, Van der Heide UA. Van Moorselaar RJA. Hofman P. and Lagendijk JJW. Clinical feasibility study for the use of implanted gold seeds in the prostate as reliable positioning markers during megavoltage irradiation, Radiot. Oncol. (2003); 67: [37] Denis L, Morton MS, Griffiths K. Diet and its preventive role in prostatic disease. Eur. Urol. (1999); 35(5-6): [38] Eastham JA, Scardino PT. Radical prostatectomy. In PC Walsh et al., eds., Campbell's Urology, 8th ed., (2002); Philadelphia: W.B. Saunders. [39] Edwards AWF. Likelihood: An account of the statistical concept of likelihood ad its applications to scientific inference. Cambridge University Press (1972). [40] Eichler K et al. Diagnostic value of systematic prostate biopsy methods in the investigation for prostate cancer: a systematic review. Centre for Reviews and Dissemination, University of York; (2005). [41] ESTRO. The GEC ESTRO Handbook of Brachytherapy. Leuven, Belgium:ESRTO (2002). [42] Falkmer S, Larsson B, Stenson S. Effects of single dose proton irradiation of normal skin and Vx2 carcinma in rabbit ears: a comparative investigation with protons and roentgen rays. Acta Oncol.(1959); 52: [43] Falkmer S, Fors B, Larsson B, Lindell A, Naeslud J, Stenson S. Pilot study on proton irradiation of human carcinoma. Acta Radiol. Therapy. Phys. Biol. (1962); 58:33-51 [44] Fowler JF. The linear quadratic formula and progress in fractionated radiotherapy. Br J Radiol(1989); 62:
106 [45] Forman JD, Kumar R, Haas G, Montie J, Porter AT, Mesina CF. Neoadjuvant hormonal downsizing of localized carcinoma of the prostate: effects on the volume of normal tissue irradiation. Cancer Invest. (1995); 13(1):8-15. [46] Fowler JE Jr, Barzell W, Hilaris BS, Whitmore WF Jr. Complications of 125-Iodine implantation and pelvic lymphadenectomy in the treatment of prostate cancer. J Uro. (1979); 121(4): [47] Fowler J.F. Brief summary of radiobiological principles in fractionated radiotherapy. Semin. Radiat. Oncol. (1992); 2: [48] Fraass BA. The development of conformal radiation therapy. Med. Phys. (1995); 22(11) Pt2: [49] Gerweck LE, Kozin SV. Relative biological effectiveness of proton beams in clinical therapy. Radiat. Oncol. (1999); 50: [50] Garnick MB, Fair WR. Combating prostate cancer. Sci. Am (1998); 279(6): [51] Gleason DF, Mellinger GT: Prediction of prognosis for prostatic adenocarcinoma by combined histological grading and clinical staging. J. Urol. (1974); 111(1):58-64, [PUBMED Abstract] [52] Gleason DF. The Veteran's Administration Cooperative Urologic Research Group: histologic grading and clinical staging of prostatic carcinoma. In Tannenbaum M (ed.) Urologic Pathology: The Prostate. Lea and Febiger, Philadelphia, (1977); [53] Gleason DF. Histologic grade, clinical stage, and patient age in prostate cancer. NCI Monogr: (1988); 7: [54] Gibbons RP, Correa RJ Jr Brannen GE, Weissman RM. Total prostatectomy for clinically prostatic cancer: long term results. J. Urol (1989); 141(3): [55] Goldner G, Wachter-Gerstner N, WachterS, et al. Acute side effects during #D-planned conformal radiotherapy of prostate cancer. Strahlenther Onkol. (2003); 179:320-7 [56] Goodhead DT. Saturable repair models of radiation action in mammalian cells. Radiat. Res. (1985); 104:S58-S67. [57] Graffman S, Haymaker W, Hugosson R, Jung B. High-energy protons in the postoperative treatment of malignant glioma Acta Radiol. Therapy. Phys. Biol. (1975); 14: [58] Graffman S, Brahme A, Larsson B. Proton radiotherapy with the Uppsala cyclotron. Experience and Plans. Strahlentherapie (1985); 161: [59] Greene DR, Fitzpatrick JM, Scardino PT. Anatomy of the prostate and distribution of early prostate cancer. Semin. Surg. Oncol. (1995); 11(1):9-22. [60] Griner PF, Mayewski RJ, Mushlin AI, Greenland P. Selection and interpretation of diagnostic tests and procedures. Annals of Internal Medicine (1981); 94: [61] Grusell E, Montelius A, Russell KR, Blomquist E, Pellettieri L, Lilja A, et al. Patient positioning for fractionated precision radiation treatment of targets in the head using fiducial markers. Radit Oncol (1994); 33: [62] Gueulette J, Bohm L, Slabbert JP et al. Proton relative biological effectiveness (RBE) for survival in mice after thoracic irradiation with fractionated doses. Int. J. Radiat. Biol. Phys. (2000); 47: [63] Hanks GE, Lee WR, Hanlon AL, et al. Conformal technique dose escalation in prostate cancer: Improved cancer control with higher doses in patients with pretreatment PSA>10ng/ml. Int. J. Radiat. Oncol. Bio. Phys. (1996); 35: [64] Hanks GE, Hanlon AL, Schultheiss TE, Pinover WH, Movsas B, Epstein BE, et al. Dose escalation with 3-D conformal treatment: five year outcomes, treatment optimization, at future directions. Int. J. Radiat. Oncol. Bio. Phys. (1998); 41(3): [65] Hanlon AL, Watkins BD, Peter R, Hanks GE. Quality of life study in prostate cancer patients treated with three dimensional conformal radiotherapy: comparing late bowel and bladder quality of life symptoms to that of the normal population. Int. J Radiat. Oncol. Biol. Phys. (2001); 49(1): [66] Hall EJ. Radiobiology for the Radiologist. JB Lippincott Co. (1988); Philadelphia. [67] Hall-Craggs ECB. Anatomy as a base for clinical medicine, (1995) 3 rd ed. London: Williams & Wilkins. [68] Helax AB. Dose Formalism and Models in Helax-TMS. Uppsala, Sweden; (1998). 105
107 [69] Helgason AC, Adolfsson J, Dickman P, Arver S, Fredrikson M, Steineck G. Factors associated with waning sexual function among elderly men and prostate cancer patients. J Urol. (1997); 158(1): [70] Henningsohn L, Wijkstrom H, Dickman PW, Bergmark K, Steinek G. Distresful symptoms after radical cystectomy with urinary diversion for urinary bladder cancer: A Swedish population-based study. Eur. Urol. (2001); 40(2): [71] Herold M, Hanlon AL, Hanks GE. Diabetes mellitus: a predictor for late radiation morbidity. Int J Radiat. Oncol Biol. Phys. (1999); 43(3): [72] Holmberg L, Bill-Axelson A, Helgesen F, Salo JO, Folmerz P, Haggman M et al. A randomized trial comparing radical prostatectomy with watchful waiting in early prostate cancer. N. Engl. J Med. (2002); 347(11): [73] Horiot JC, Le Fur R, N Guyen T et al. Hyperfractionation versus conventional fractionation in oropharyngeal carcinoma: final analysis of a randomized trial of the EORTC cooperative group of radiotherapy. Radia. Oncol (192); 25: [74] Hsing AW, Tsao L, Devesa SS. International trends and patterns of prostate cancer incidence and mortality. Int J Cancer (2000); 85 (1): [75] "IARC Worldwide Cancer Incidence Statistics Prostate"(2001) JNCI Cancer Spectrum. Oxford University Press. [76] ICRU50. Prescribing, recording and reporting photon beam therapy. Bethesda [77] ICRU. Prescribing, recording and reporting proton beam therapy. ICRU Report 50. Bethesda, Maryland: International Commission on Radiation Units and Measurements, [78] ICRU. Prescribing, recording and reporting proton beam therapy. (Supplement on ICRU 50), ICRU Report 62. Bethesda, Maryland: International Commission on Radiation Units and Measurements, (1999). [79] ICRU562 Prescribing, recording and reporting photon beam therapy (supplement to ICRU report 50). Bethesda [80] Intensity-modulated Radiotherapy: current status and issues of interest. Int. J. Radiat. Oncol. Bio. Phys. (2001); 51: [81] Joel S. Bedford. Radiological Health Sciences, Colorado State University, Fort Collins, Colorado Radiation Research (2003);159(1): [82] Johnston MJ, Robertson GM, Frizelle FA. Management of late complications of pelvic radiation in the rectum and anus: a review. Dis Colon Rectum (2003); 46(2): [83] Jung B, Montelius A, Dahlin H, Ekström P, Ahnesjö A, Högström B, et al. The conceptual design of a radiation oncology planning system. Comput Methods Programs Biomed (1997); 52: [84] Källman P, Lind B K and Brahme A. An algorithm for maximizing the probability of complication free tumour control in radiation therapy. Phys. Med Biol. (1992); 37: [85] Källman P. Optimization of radiation therapy planning using physical and biological objective functions Phd Thesis (1992); University of Stockholm, Sweden. [86] Kent E. Wallner, M.D. Gregory S. Merrick, M.D., Mark L. Benson, M.D., Wayne M. Butler, Ph.D., Jeffry Maki, M.D., and Brayan G. Tollenaar, M.S. Penile Bulb Imaging. Int. J. Radiat. Oncol. Biol. Phys. (2002); 53(4): [87] Koehler AM, Schneider RJ, Sisterson JM. Range modulators for protons and heavy ions. Nuclear Instrument and Methods (1975); 437. [88] Kovacs G, Galalae R. Fractionated perineal high dose rate temporary brachytherapy combined with external beam radiotherapy in the treatment of localized prostate cancer: is lymph node sampling necessary? Cancer Radiot. (2003); 7(2): [89] Kuban VL, Majumber PK. Prostate gland: structure, functions and regulation. Int. Urol. Nephrol. (1995); 27(3): [171] Larsson B, Leksell L, Rexed B, Sourander P, Mair W, Andersson B. The high-energy proton beam as a neurosurgical tool. Naure (1958); 182: [90] Larsson B, Leksell L, Rexed B et al. The high-energy proton beam as a neurosurgical tool. Nature (1958); 182:
108 [91] Larsson B, Leksell L, Rexed B, Sourander P. Effect of high-energy protons on the spinal cord. Acta Radiol. (1959); 51: [92] Larsson B. Radiobiological properties of beams of high-energy protons. Br J Radiol (1961); 34: [93] Larsson B, Leksell L, Rexed B. The use of high-energy protons for cerebral surgery in man. Acta Chir. Scandinavia (1963); 125:1-7. [94] Larsson B. Pre-therapeutic properties of beams of high-energy protons. Radiat. Res. Supp. (1967); 7: [95] Leibel SA, Fuks Z, Zelefsky MJ, Wolden SL, Rosenzweig KE, Alektiar KM, et al. Intensitymodulated radiotherapy. Cancer J. (2002); 8(2): [96] Leslie T. Yonemoto, Jerry D. Slater, Carl J. Rossi, John E. Antoine et al. Combine proton and photon conformal radiation therapy or locally advanced carcinoma of the prostate: Peliminary results of a phase I/II Study. Int. J. Radiat. Oncol. Bio. Phys. (1997); 37(1): [97] L.H. Sobin and Wittekind. (2002) TNM classification of malignant tumours, Willey-Liss, sixth edition. [98] Lilleby W, Fossa SD, Waehre HR, Olsen DR. Long term morbidity and quality of life in patients with localized prostate cancer undergoing definitive radiotherapy orradical prostatectomy. Int. J. Radiat. Oncol Biol. Phys. (1999); 43(4): [99] Lind, B. K. Radiation therapy planning and optimization studies as inverse problems. Thesis, (1991) University of Stockholm, Sweden. [100] Lind B. K., J. Nilsson, J. Lof and A. Brahme. Generalization of the normalized dose-response gradient to non-uniform dose delivery. Acta. Oncol. (2001); 40, [101] Lindholm C, Cavallin-Ståhl E, Ceberg J, Frödin JE, Littbrand B, Möller TR. Radiotherapy practices in Sweden compared to the scientific evidence. Acta Oncol (2003); 42(5-6): [102] Ling CC, Burman C, Chui CS, Kutcher GJ, Leibel Sa, LoSasso T et al. Conformal radiation treatment of prostate cancer using inversely-planned intensity modulated photon beams produced with dynamic multileaf collimation. Int. J. Radiat. Oncol. Bio. Phys. (1996); 35(4): [103] Lu-Yao GL, Yao SL. Population based study of long term survival in patients with clinically localized prostate cancer. Lancet. (1997); 349(9056): [104] Lu-Yao G, Albertsen PC, Stanford JL, Stukel TA, Walker-Corkery ES, Barry MJ. Natural experiment examining impact of aggressive screening and treatment on prostate cancer mortality in two fixed cohorts from Seattle area and Connecticut. BMJ (2002); 325(7367):740. [105] Mackie TR, Holmes T, Swerdloff S, Reckwerdt P, Deasy JO, Yang J et al. Tomotherapy: a new concept for the delivery of dynamic conformal radiotherapy. Med Phys. (1993); 20(6): [106] Mackie TR, Balog J, Ruchala K, Shepard D, Aldridge S, Fitchard E et al. Tomotherapy. Semin Radiat. Oncol. (1999); 9(1): [107] Mah K, van Dyk J, Keane T and Poon PY. Acute radiation-induced pulmonary damage. A clinical sudy on the response to fractionated radiotherapy. Int. J. Radiat. Biol. Phys. (1987); 13: [108] Marks LB, Carroll PR, Dugan TC, Anscher MS. The response of the urinary bladder, urethra, and ureter to radiation and chemotherapy. Int. J Radiat. Oncol Biol. Phys. (1995); 31(5): [109] McVary, K. T. Clinical practice. Erectile dysfunction. N Engl J Med.; (2007); 357(24): [110] Mejaddem Y. Photon and electron transport calculations for development of intensity modulated radiation therapy. Doctoral thesis: Stockholm University, University Press, [111] Merrick GS, Butler WM, Dorsey AT, Lief JH, Donzella JG. A comparison of radiation dose to the neurovascular bundles in men with and without prostate bachytherapy-induced erectile dysfunction. Int. J. Radiat. Oncol Biol. Phys. (2000); 48(4): [112] Merrick GS, Wallner KE, Butler WM, Galbreath RW, Lef JH, Benson ML. A comparison of radiation dose to the bulb of the penis in men with and without prostate brachytherapy-induced erectile dysfunction. Int. J. Radiat. Oncol Biol. Phys. (2001); 50(3): [113] Merrick GS, Butler WM, Wallner KE, Lief JH, Anderson RL, Smeiles BJ et al. The importance of radiation doses to the penile bulb vs. crura in the development of post-brachytherapy erectile 107
109 dysfunction. Int. J. Radiat. Oncol Biol. Phys. (2002); 54(4): [114] Metz CE. Basic principles of ROC analysis. Seminars in Nuclear Medicine, (1978); 8: [115] Michael BD. A simple graphical determination of a/β from reciprocal dose plots. Int J Radiat Biol (1985); 47: [116] Montelius A, Blomquist E, Naeser P, Brahme A, Carlsson J, Carlsson AC, et al. The narrow proton beam therapy unit at the Svedberg Laboratory in Uppsala. Acta Oncol (1991); 30: [117] Montelius A, Jung B, Rikner G, Murman A, Russell K. Quality assurance tests of the TMS-radix treatment planning system. In: Breit A, editor. Advanced radiation therapy tumour response monitoring and treatment planning. Berlin, Heidelberg: Springer-Verlag; (1992). p [118] Moiseenko V, Battista J and van Dyk J. Normal tissue complication probabilities: dependence on choice of biological model and dose-volume histogram reduction scheme Int. J. Radiat. Oncol. Biol. Phys. (2000); 46: [119] Moore, Keith L.; Dalley, Arthur F, Clinically Oriented Anatomy, (2006) 5th Edition, Lippincott Williams & Wilkins. [120] National Institute for Health and Clinical Excellence (2008). Prostate Cancer: Diagnosis and Treatment; Full Guideline. [121] Parkinson MC, Bott SR, Montironi R, Melia J. Screening for prostatic cancer and its evolution within Britain. J Pathol (2002); 197(2): [122] Pawitan, Yudi. In All Likelihood: Statistical Modeling and Inference Using Likelihood. Oxford University press (2001); ISBN [123] Penson DF. Quality of life following prostate cancer treatments Curr. Urol. Rep. (2000); 1:(1) [124] Pilepich MV, Asbell SO, Krall JM, Baerwald WH, Sause WT, Rubin P et al. Correlation of radiotherapeutic parameters and treatment related morbidity-analysis of RTOG Study Int J Radiat. Oncol Biol. Phys. (1987); 13(7): [125] Pilepich MV, Krall JM, Sause WT, Johnson RJ, Russ HH, Hanks GE et al. Correlation of radiotherapeutic parameters and treatment related morbidity in carcinoma of the prostate-analysis of RTOG Study Int J Radiat. Oncol Biol. Phys. (1987); 13(3): [126] Pilepich MV, Krall JM, al Sarrf M, John MJ, Doggett RL, Sause WT et al. Androgen deprivation with radiation therapy compared with radiation therapy alone for locally advanced prostatic carcinoma: a randomized comparative trial of The Radiation Therapy Oncology Group. Urology (1995); 45(4): [127] Pilepich MV, Winter K, John MJ, Mesic JB, Sause W, Rubin Pet al. Phase III radiation therapy oncology group (RTOG) trial of androgen deprivation adjuvant to definitive radiotherapy in locally advanced carcinoma of the prostate. Int. J, Radiat. Oncol. Biol. Phys. (2001); 50(5): [128] Pollack A, Zagars GK, Starkschall G, Antolak JA, Lee JJ, Huang E et al. Prostate cancer radiation dose response: results of the M.D. Anderson phase III randomized trial. Int J Radiat. Oncol Biol. Phys (2002); 53(5): [129] Potosky AL, Legler J, Albertsen PC, Standford JL, Gilliland FD, Hamilton AS et al. Health outcomes after prostatectomy or radiotherapy for prostate cancer: results from the Prostate Cancer Outcomes Study. J Natl. Cancer Inst. (2000); 92(19): [130] Pound CR, Partin AW, Eisenberger MA, Chan DW, Pearson JD, Walsh PC Natural history of progression after PSA elevation fllowing radical prostatectomy. JAMA (1999); 281(17) [131] Purdy JA, Harms WB, Matthews JW, DrzymalaR, Amemi B, Simpson JR, et al. Advances in 3- dimensional radiation treatment planning systems: room-view display with real time interactivity. Int. J. Radiat. Oncol. Bio. Phys. (1999); 43(1): [132] Renner TR, Chu WT. Wobbler facility for biomedical experiments. Med. Phys. (1987); 14: [133] Russell, W. L., in Advances in Radiation Research Biology and Medicine edit. by Duplan, J. F. and Chapiro, A. Gordon and Breach, New York. (1973);
110 [134] Russell KR, Isacsson U, Saxner M, Ahnesjö A, Montelius A, Grusell E, et al. Implementation of pencil kernel and depth penetration algorithms for treatment planning of proton beams. Phys Med Biol (2000); 45:9-27. [135] Saunders MI, Dische S, Hong A et al. Continuous hyperfractionated accelerated radiotherapy in locally advanced carcinoma of the head and neck region. Int. J. Radiat. Biol. Phys. (1989); 17: [136] Scher HI, Heller G. Clinical states in prostate cancer: toward a dynamic model of disease progression. Urol. (2000);55(3): [137] Schover LR. Sexual rehabilitation after treatment for prostate cancer. Cancer (1993); 71(3 Suppl): [138] Schulman CC. Ekane S, Zlotta AR. Nutrition and prostate cancer: evidence or suspicion? Urol. (2001); 58(3): [139] Schultheiss T E, Oorton C G and Peck R A. Models in radiotherapy, Volume effects Med. Phys. (1983);10: [140] Schultheiss TE, Lee WR, Hunt MA, Hanlon AL, Peter RS, Hanks GE. Late GI and GU complications in the treatment of prostate cancer. Int J Radiat. Oncol Biol. Phys. (1997); 37(1):3-11. [141] Seidel HM, Ball JW, Dains JE, Benedict GW. Mosby's Guide to Physical Examination. 6th ed. Philadelphia, (2006); Pa: C.V. Mosby;. [142] Shipley WU, Zietman AL, Hanks GE, Coen JJ, Caplan RJ, Won M et al. Treatment related sequelae following external beam radiation for prostate cancer: a review with an update in patients with stages T1 and T2 tumor. J Urol. (1994); 152( Pt 2): [143] Slater JM, Archambeau JO, Miller DW et al. The proton treatment center at Loma Linda University Medical Centre: rationale for and description of its development. Int. J. Radiat. Biol. Phys. (1992); 22: [144] Smith DS, Humphrey PA, Catalona WJ. The early detection of prostate carcinoma with prostate specific antigen: The Washington University experience. Cancer; (1997); 80(9): [145] Smith JA, Chan RC, Chang SS, et al. "A comparison of the incidence and location of positive surgical margins in robotic assisted laparoscopic radical prostatectomy and open retropubic radical prostatectomy". J. Urol. (2007); 178(6): ; discussion [146] Steel GG Adams GE and Horwich A. The biological Basis of Radiotherapy, second edition. Elsevier (1989); Amsterdam. [147] Steel GG, Mcmllan TJ and Peacock JH. The 5 R s of radiobiology. Int. J. Radiat. Biol. (1989); 56: [148] Steineck G, Helgesen F, Adolfsson J, Dickman PW, Johansson JE, Norlen BJ et al. Quality of life after radical prostatectomy or watchful waiting. N Engl. J Med. (2002); 347(11): [149] Sténson S. Effects of high-energy protons on healthy organs and malignant tumours. Including a study of radiation protection by local hypoxia, Doctoral dissertation. Uppsala University, [150] Thames HD, Withers HR, Peters LJ et al. Changes in early and late radiation responses with altered dose fractionation: implications for dose-survival relationships. Int. J. Radiat. Biol. Phys. (1982); 8: [151] Thompson IM, Pauler DK, Goodman PJ, et al. Prevalence of prostate cancer among men with a prostate-specific antigen level 4.0 ng per milliliter. New England Journal of Medicine; (2004); 350(22): [152] Tilly N, Johansson J, Isacsson U, et al. The influence of RBE variations in a clinical proton treatment plan for a hypopharynx cancer. Phys. Med. Biol. (2005); 50: [153] Tobias CA, Roberts JE, Lawrence JH et al. Irradiation hypophysectomy and related studies using 340 MeV protons and 190 MeV deuterons. Peaceful Use of Atomic Energy (1955); 10: [154] Tortora J, Grabowski R. The Digestive system. Principles of Anatomy ad Physiology, 8 th edition New York: Harber Collins College, (2003);
111 [55] Tubiana D, Dutreix J, Wambersie A. Effects of normal tissues. In: Tubiana D, Dutreix J, Wambersie A. eds, Introduction to Radiobiology, (1990) 1 st ed. London: Taylor and Francis Ltd, [156] Tucker, S.L., Thames, H.D., Taylor, J.M.G. How well is the probability of tumour cure after fractionated radiotherapy described by Poisson statistics? Rad. Res., (1990); 124: [157] UICC. (2002) TNM Classification of Malignant Tumours, In: Sobin LH, Wittekin Ch, eds, 6 th ed. New York: Wiley-Liss. [158] Valicenti R, Lu J, Pilepich M, Asbell S, Grignon D. Survival advantages from higher-dose radiation therapy for clinically localized prostate cancer treated on the Radiation Therapy Oncology Group trials. J. Clin. Oncol. (2000); 18(14): [159] Van der Cruijsen-Koeter, IW; Vis AN, Roobol MJ, Wildhagen MF, de Koning HJ, van der Kwast TH, Schroder FH. Comparison of screen detected and clinically diagnosed prostate cancer in the European randomized study of screening for prostate cancer, section Rotterdam. Urol. (2005); 174 (1): [160] Walsh PC, Donker PJ. Impotence following radical prostatectomy: insight into aetiology and prevention. J. Urol. (1982); 128(3): [161] Webb S. The Physics of Three-Dimensional Radiation Therapy, Conformal Radiotherapy, Radiosurgery and Treatment Planning, 1 st ed. London: IOP Publishing Ltd, [162] Webb S. The Physics of Conformal Radiotherapy: Advances in Technology. Bristol and Philadelphia: IOP Publishing Ltd, [163] Weir HK, Thun MJ, Hankey BF, Ries LA, Howe HL, Wingo PA et al.. Annual report to the nation of the status of cancer, , featuring the uses of surveillance data for cancer prevention and control. J Natl Cancer Inst (2003); 95(17): [164] Wheldon, T.E, Michalowski, A.S. and Kirk, J. The effect of irradiation on function in selfrenewing normal tissues with differing proliferative organization. British Journal of Radiology, (1982); 55: [165] Widmark A, Fransson P, Tavelin B. Self-assessment questionnaire for evaluating urinary and intestinal late side effects after pelvic radiotherapy in patients with prostate cancer compared with agematched control population. Cancer (1994);74(9): [166] Williams MV, Denekamp J and Fowler JF. A review of a/β ratios for experimental tumours: implication for clinical studies of altered fractionation.int J Radiat. Oncol. Biol. Phys. (1985); 11: [167] Wilson R.R. Radiological Use of fast Protons. Radiology (1946); 47:487-4 [168] Wilt T. Prostate cancer (non-metastatic). Clinical Evidence (2004); 11: [169] Withers HR. The four R s of radiotherapy. Adv, Radiat. Biol. (1975); 5: [170] Withers, H.R. and Peter L.J. Biological aspects of radiation therapy. In: Textbook of radiotherapy Ed: Fletcher G.H. (1980); ISBN [171] Withers H R, Taylor J M G and Maciejeski B. Treatment volume and tissue tolerance Int. J Radiat. Oncol. Biol. Phys. (1988); 14: [172] Yaes R.J. The biological effect of inhomogeneous dose distribution in fractionated radiotherapy. Int. J. Radiat. Oncol. Biol. Phys. (1990); 19: [173] Yaffe D, Zissin R: Cowper's glands duct: Radiographic findings. Urol. Radiol. (1991); 13: [174]Yeoh EK, Horowitz M. Radiation enteritis. Surg. Gynecol. Obster. (1987); 65(4): [175] Zelefsky MJ, Wallner KE, Ling CC, Raben A, Hollister T, Wolfe T, et al. Comparison of the 5- year outcome and morbidity of three-dimensional conformal radiotherapy versus transperineal permanent iodine-125 implantation for early-stage prostate cancer. J. Clin. Oncol. (1995); 17(2): [176] Zelefsky MJ, Cowen D, Fuks Z Shike M, Burman C, Jackson A et al. Long term tolerance of high dose three-dimensional conformal radiotherapy in patients with localized prostate carcinoma. Cancer (1999); 85(11):
112 [177] Zelefsky MJ, Fuks Z, Hunt M, et al. High dose radiation delivered by intensity modulated conformal radiotherapy improves the outcome of localize prostate cancer. J. Urol. (2001); 166: [178] Zietman AL, Prince EA, Nakfoor BM, Shipley WU. Neoadjuvant androgen suppression with radiation in the management of locally adenocarcinoma of he prostate: experimental and clinical results. Urol. (1997); 49(3A Suppl): [179] Zincke H, Oesterling JE, Blute ML, Bergstralh EJ, Myers RP, Barett DM. Long term (15 years) results after radical prostatectomy for clinical localized (stage T2c or lower) prostate cancer. J. Urol. (1994); 152(5 Pt 2): [180] Zweig MH, Campbell G Receiver-operating characteristic (ROC) plots: a fundamental evaluation tool in clinical medicine. Clinical Chemistry (1993); 39:
PATIENT GUIDE. Localized Prostate Cancer
 PATIENT GUIDE Localized Prostate Cancer The prostate* is part of the male reproductive system. It is about the same size as a walnut and weighs about an ounce. As pictured in Figure 1, the prostate is
PATIENT GUIDE Localized Prostate Cancer The prostate* is part of the male reproductive system. It is about the same size as a walnut and weighs about an ounce. As pictured in Figure 1, the prostate is
Early Prostate Cancer: Questions and Answers. Key Points
 CANCER FACTS N a t i o n a l C a n c e r I n s t i t u t e N a t i o n a l I n s t i t u t e s o f H e a l t h D e p a r t m e n t o f H e a l t h a n d H u m a n S e r v i c e s Early Prostate Cancer:
CANCER FACTS N a t i o n a l C a n c e r I n s t i t u t e N a t i o n a l I n s t i t u t e s o f H e a l t h D e p a r t m e n t o f H e a l t h a n d H u m a n S e r v i c e s Early Prostate Cancer:
These rare variants often act aggressively and may respond differently to therapy than the more common prostate adenocarcinoma.
 Prostate Cancer OVERVIEW Prostate cancer is the second most common cancer diagnosed among American men, accounting for nearly 200,000 new cancer cases in the United States each year. Greater than 65% of
Prostate Cancer OVERVIEW Prostate cancer is the second most common cancer diagnosed among American men, accounting for nearly 200,000 new cancer cases in the United States each year. Greater than 65% of
Radiation Therapy for Prostate Cancer
 Radiation Therapy for Prostate Cancer Introduction Cancer of the prostate is the most common form of cancer that affects men. About 240,000 American men are diagnosed with prostate cancer every year. Your
Radiation Therapy for Prostate Cancer Introduction Cancer of the prostate is the most common form of cancer that affects men. About 240,000 American men are diagnosed with prostate cancer every year. Your
Treating Localized Prostate Cancer A Review of the Research for Adults
 Treating Localized Prostate Cancer A Review of the Research for Adults Is this information right for me? Yes, this information is right for you if: Your doctor * said all tests show you have localized
Treating Localized Prostate Cancer A Review of the Research for Adults Is this information right for me? Yes, this information is right for you if: Your doctor * said all tests show you have localized
PSA Screening for Prostate Cancer Information for Care Providers
 All men should know they are having a PSA test and be informed of the implications prior to testing. This booklet was created to help primary care providers offer men information about the risks and benefits
All men should know they are having a PSA test and be informed of the implications prior to testing. This booklet was created to help primary care providers offer men information about the risks and benefits
Prostate Cancer Screening. A Decision Guide
 Prostate Cancer Screening A Decision Guide This booklet was developed by the U.S. Department of Health and Human Services, Centers for Disease Control and Prevention (CDC). Is screening right for you?
Prostate Cancer Screening A Decision Guide This booklet was developed by the U.S. Department of Health and Human Services, Centers for Disease Control and Prevention (CDC). Is screening right for you?
CMScript. Member of a medical scheme? Know your guaranteed benefits! Issue 7 of 2014
 Background CMScript Member of a medical scheme? Know your guaranteed benefits! Issue 7 of 2014 Prostate cancer is second only to lung cancer as the leading cause of cancer-related deaths in men. It is
Background CMScript Member of a medical scheme? Know your guaranteed benefits! Issue 7 of 2014 Prostate cancer is second only to lung cancer as the leading cause of cancer-related deaths in men. It is
FAQ About Prostate Cancer Treatment and SpaceOAR System
 FAQ About Prostate Cancer Treatment and SpaceOAR System P. 4 Prostate Cancer Background SpaceOAR Frequently Asked Questions (FAQ) 1. What is prostate cancer? The vast majority of prostate cancers develop
FAQ About Prostate Cancer Treatment and SpaceOAR System P. 4 Prostate Cancer Background SpaceOAR Frequently Asked Questions (FAQ) 1. What is prostate cancer? The vast majority of prostate cancers develop
Prostate Cancer. There is no known association with an enlarged prostate or benign prostatic hyperplasia (BPH).
 Prostate Cancer Definition Prostate cancer is cancer that starts in the prostate gland. The prostate is a small, walnut-sized structure that makes up part of a man's reproductive system. It wraps around
Prostate Cancer Definition Prostate cancer is cancer that starts in the prostate gland. The prostate is a small, walnut-sized structure that makes up part of a man's reproductive system. It wraps around
Prostate Cancer. Patient Information
 Prostate Cancer Patient Information 1 The Prostate & Prostate Cancer The prostate is a small gland in the male reproductive system, approximately the size and shape of a walnut. It is located directly
Prostate Cancer Patient Information 1 The Prostate & Prostate Cancer The prostate is a small gland in the male reproductive system, approximately the size and shape of a walnut. It is located directly
Prostate Cancer Screening. A Decision Guide for African Americans
 Prostate Cancer Screening A Decision Guide for African Americans This booklet was developed by the U.S. Department of Health and Human Services, Centers for Disease Control and Prevention (CDC). Published
Prostate Cancer Screening A Decision Guide for African Americans This booklet was developed by the U.S. Department of Health and Human Services, Centers for Disease Control and Prevention (CDC). Published
An Introduction to PROSTATE CANCER
 An Introduction to PROSTATE CANCER Being diagnosed with prostate cancer can be a life-altering experience. It requires making some very difficult decisions about treatments that can affect not only the
An Introduction to PROSTATE CANCER Being diagnosed with prostate cancer can be a life-altering experience. It requires making some very difficult decisions about treatments that can affect not only the
Prostate Cancer. What is prostate cancer?
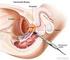 Scan for mobile link. Prostate Cancer Prostate cancer is a tumor of the prostate gland, which is located in front of the rectum and below the bladder. Your doctor may perform a physical exam, prostate-specific
Scan for mobile link. Prostate Cancer Prostate cancer is a tumor of the prostate gland, which is located in front of the rectum and below the bladder. Your doctor may perform a physical exam, prostate-specific
AFTER DIAGNOSIS: PROSTATE CANCER Understanding Your Treatment Options
 AFTER DIAGNOSIS: PROSTATE CANCER Understanding Your Treatment Options INTRODUCTION This booklet describes how prostate cancer develops, how it affects the body and the current treatment methods. Although
AFTER DIAGNOSIS: PROSTATE CANCER Understanding Your Treatment Options INTRODUCTION This booklet describes how prostate cancer develops, how it affects the body and the current treatment methods. Although
Treating Prostate Cancer
 Treating Prostate Cancer A Guide for Men With Localized Prostate Cancer Most men have time to learn about all the options for treating their prostate cancer. You have time to talk with your family and
Treating Prostate Cancer A Guide for Men With Localized Prostate Cancer Most men have time to learn about all the options for treating their prostate cancer. You have time to talk with your family and
A918: Prostate: adenocarcinoma
 A918: Prostate: adenocarcinoma General facts of prostate cancer The prostate is about the size of a walnut. It is just below the bladder and in front of the rectum. The tube that carries urine (the urethra)
A918: Prostate: adenocarcinoma General facts of prostate cancer The prostate is about the size of a walnut. It is just below the bladder and in front of the rectum. The tube that carries urine (the urethra)
HEALTH NEWS PROSTATE CANCER THE PROSTATE
 HEALTH NEWS PROSTATE CANCER THE PROSTATE Prostate comes from the Greek meaning to stand in front of ; this is very different than prostrate which means to lie down flat. The prostate is a walnut-sized
HEALTH NEWS PROSTATE CANCER THE PROSTATE Prostate comes from the Greek meaning to stand in front of ; this is very different than prostrate which means to lie down flat. The prostate is a walnut-sized
1. What is the prostate-specific antigen (PSA) test?
 1. What is the prostate-specific antigen (PSA) test? Prostate-specific antigen (PSA) is a protein produced by the cells of the prostate gland. The PSA test measures the level of PSA in the blood. The doctor
1. What is the prostate-specific antigen (PSA) test? Prostate-specific antigen (PSA) is a protein produced by the cells of the prostate gland. The PSA test measures the level of PSA in the blood. The doctor
For further information on screening and early detection of prostate cancer, see the Section entitled Screening for Prostate Cancer.
 Prostate Cancer For many older men, prostate cancer may be present but never cause symptoms or problems and many men will die with their prostate cancer rather than of their prostate cancer. Yet it remains
Prostate Cancer For many older men, prostate cancer may be present but never cause symptoms or problems and many men will die with their prostate cancer rather than of their prostate cancer. Yet it remains
Prostate Cancer Action Plan: Choosing the treatment that s right for you
 Prostate Cancer Action Plan: Choosing the treatment that s right for you Segment 1: Introduction Trust me, there's a better way to choose a treatment for your prostate cancer. Watching this program is
Prostate Cancer Action Plan: Choosing the treatment that s right for you Segment 1: Introduction Trust me, there's a better way to choose a treatment for your prostate cancer. Watching this program is
Bard: Prostate Cancer Treatment. Bard: Pelvic Organ Prolapse. Prostate Cancer. An overview of. Treatment. Prolapse. Information and Answers
 Bard: Prostate Cancer Treatment Bard: Pelvic Organ Prolapse Prostate Cancer An overview of Pelvic Treatment Organ Prolapse Information and Answers A Brief Overview Prostate Anatomy The prostate gland,
Bard: Prostate Cancer Treatment Bard: Pelvic Organ Prolapse Prostate Cancer An overview of Pelvic Treatment Organ Prolapse Information and Answers A Brief Overview Prostate Anatomy The prostate gland,
A Woman s Guide to Prostate Cancer Treatment
 A Woman s Guide to Prostate Cancer Treatment Supporting the man in your life Providing prostate cancer support and resources for women and families WOMEN AGAINST PROSTATE CANCER A Woman s Guide to Prostate
A Woman s Guide to Prostate Cancer Treatment Supporting the man in your life Providing prostate cancer support and resources for women and families WOMEN AGAINST PROSTATE CANCER A Woman s Guide to Prostate
PROSTATE CANCER. Get the facts, know your options. Samay Jain, MD, Assistant Professor,The University of Toledo Chief, Division of Urologic Oncology
 PROSTATE CANCER Get the facts, know your options Samay Jain, MD, Assistant Professor,The University of Toledo Chief, Division of Urologic Oncology i What is the Prostate? Unfortunately, you have prostate
PROSTATE CANCER Get the facts, know your options Samay Jain, MD, Assistant Professor,The University of Toledo Chief, Division of Urologic Oncology i What is the Prostate? Unfortunately, you have prostate
PROSTATE CANCER. Diagnosis and Treatment
 Diagnosis and Treatment Table of Contents Introduction... 1 Patient and Coach Information... 1 About Your Prostate... 1 What You Should Know About Prostate Cancer... 2 Screening and Diagnosis... 2 Digital
Diagnosis and Treatment Table of Contents Introduction... 1 Patient and Coach Information... 1 About Your Prostate... 1 What You Should Know About Prostate Cancer... 2 Screening and Diagnosis... 2 Digital
Prostate Cancer Treatment
 Scan for mobile link. Prostate Cancer Treatment Prostate cancer is a tumor of the prostate gland, which is located in front of the rectum and below the bladder. Your doctor may perform a physical exam,
Scan for mobile link. Prostate Cancer Treatment Prostate cancer is a tumor of the prostate gland, which is located in front of the rectum and below the bladder. Your doctor may perform a physical exam,
DIAGNOSIS OF PROSTATE CANCER
 DIAGNOSIS OF PROSTATE CANCER Determining the presence of prostate cancer generally involves a series of tests and exams. Before starting the testing process, the physician will ask questions about the
DIAGNOSIS OF PROSTATE CANCER Determining the presence of prostate cancer generally involves a series of tests and exams. Before starting the testing process, the physician will ask questions about the
PCA3 DETECTION TEST FOR PROSTATE CANCER DO YOU KNOW YOUR RISK OF HAVING CANCER?
 PCA3 DETECTION TEST FOR PROSTATE CANCER DO YOU KNOW YOUR RISK OF HAVING CANCER? PCA3 DETECTION TEST FOR PROSTATE CANCER There is a range of methods available to your healthcare professional to verify the
PCA3 DETECTION TEST FOR PROSTATE CANCER DO YOU KNOW YOUR RISK OF HAVING CANCER? PCA3 DETECTION TEST FOR PROSTATE CANCER There is a range of methods available to your healthcare professional to verify the
CONTENTS: WHAT S IN THIS BOOKLET
 Q Questions & A & Answers About Your Prostate Having a biopsy test to find out if you may have prostate cancer can bring up a lot of questions. This booklet will help answer those questions. CONTENTS:
Q Questions & A & Answers About Your Prostate Having a biopsy test to find out if you may have prostate cancer can bring up a lot of questions. This booklet will help answer those questions. CONTENTS:
The PSA Test for Prostate Cancer Screening:
 For more information, please contact your local VA Medical Center or Health Clinic. U.S. Department of Veterans Affairs Veterans Health Administration Patient Care Services Health Promotion and Disease
For more information, please contact your local VA Medical Center or Health Clinic. U.S. Department of Veterans Affairs Veterans Health Administration Patient Care Services Health Promotion and Disease
Understanding Prostate Cancer. The Urology Group Guide for Newly Diagnosed Patients. Advanced Care. Improving Lives.
 Understanding Prostate Cancer The Urology Group Guide for Newly Diagnosed Patients Advanced Care. Improving Lives. UNDERSTANDING PROSTATE CANCER The Urology Group has created this publication to provide
Understanding Prostate Cancer The Urology Group Guide for Newly Diagnosed Patients Advanced Care. Improving Lives. UNDERSTANDING PROSTATE CANCER The Urology Group has created this publication to provide
Radiation Therapy for Prostate Cancer: Treatment options and future directions
 Radiation Therapy for Prostate Cancer: Treatment options and future directions David Weksberg, M.D., Ph.D. PinnacleHealth Cancer Institute September 12, 2015 Radiation Therapy for Prostate Cancer: Treatment
Radiation Therapy for Prostate Cancer: Treatment options and future directions David Weksberg, M.D., Ph.D. PinnacleHealth Cancer Institute September 12, 2015 Radiation Therapy for Prostate Cancer: Treatment
Medical Tests for Prostate Problems
 Medical Tests for Prostate Problems National Kidney and Urologic Diseases Information Clearinghouse U.S. Department of Health and Human Services NATIONAL INSTITUTES OF HEALTH What is the prostate? The
Medical Tests for Prostate Problems National Kidney and Urologic Diseases Information Clearinghouse U.S. Department of Health and Human Services NATIONAL INSTITUTES OF HEALTH What is the prostate? The
Prostate Cancer & Its Treatment
 Your Health Matters Prostate Cancer & Its Treatment By the UCSF Medical Center Prostate Cancer Advocates Greetings! From our personal experience in dealing with our own prostate cancers, we UCSF Patient
Your Health Matters Prostate Cancer & Its Treatment By the UCSF Medical Center Prostate Cancer Advocates Greetings! From our personal experience in dealing with our own prostate cancers, we UCSF Patient
7. Prostate cancer in PSA relapse
 7. Prostate cancer in PSA relapse A patient with prostate cancer in PSA relapse is one who, having received a primary treatment with intent to cure, has a raised PSA (prostate-specific antigen) level defined
7. Prostate cancer in PSA relapse A patient with prostate cancer in PSA relapse is one who, having received a primary treatment with intent to cure, has a raised PSA (prostate-specific antigen) level defined
Prostate Cancer Treatment: What s Best for You?
 Prostate Cancer Treatment: What s Best for You? Prostate Cancer: Radiation Therapy Approaches I. Choices There is really a variety of options in prostate cancer management overall and in radiation therapy.
Prostate Cancer Treatment: What s Best for You? Prostate Cancer: Radiation Therapy Approaches I. Choices There is really a variety of options in prostate cancer management overall and in radiation therapy.
LIvING WITH Prostate Cancer
 Living with Prostate Cancer A Common Cancer in Men If you ve been diagnosed with prostate cancer, it s normal to be concerned about your future. It may help you to know that this disease can often be cured
Living with Prostate Cancer A Common Cancer in Men If you ve been diagnosed with prostate cancer, it s normal to be concerned about your future. It may help you to know that this disease can often be cured
Prostate Cancer. Understanding your diagnosis
 Prostate Cancer Understanding your diagnosis Prostate Cancer Understanding your diagnosis When you first hear that you have cancer, you may feel alone and afraid. You may be overwhelmed by the large amount
Prostate Cancer Understanding your diagnosis Prostate Cancer Understanding your diagnosis When you first hear that you have cancer, you may feel alone and afraid. You may be overwhelmed by the large amount
Us TOO University Presents: Understanding Diagnostic Testing
 Us TOO University Presents: Understanding Diagnostic Testing for Prostate Cancer Patients Today s speaker is Manish Bhandari, MD Program moderator is Pam Barrett, Us TOO International Made possible by
Us TOO University Presents: Understanding Diagnostic Testing for Prostate Cancer Patients Today s speaker is Manish Bhandari, MD Program moderator is Pam Barrett, Us TOO International Made possible by
Prostate Cancer In-Depth
 Prostate Cancer In-Depth Introduction Prostate cancer is the most common visceral malignancy among American men. In the year 2003, there are expected to be 220,000 new cases and nearly 29,000 deaths in
Prostate Cancer In-Depth Introduction Prostate cancer is the most common visceral malignancy among American men. In the year 2003, there are expected to be 220,000 new cases and nearly 29,000 deaths in
Frequently Asked Questions About Ovarian Cancer
 Media Contact: Gerri Gomez Howard Cell: 303-748-3933 [email protected] Frequently Asked Questions About Ovarian Cancer What is ovarian cancer? Ovarian cancer is a cancer that forms in tissues
Media Contact: Gerri Gomez Howard Cell: 303-748-3933 [email protected] Frequently Asked Questions About Ovarian Cancer What is ovarian cancer? Ovarian cancer is a cancer that forms in tissues
Smoking and misuse of certain pain medicines can affect the risk of developing renal cell cancer.
 Renal cell cancer Renal cell cancer is a disease in which malignant (cancer) cells form in tubules of the kidney. Renal cell cancer (also called kidney cancer or renal adenocarcinoma) is a disease in which
Renal cell cancer Renal cell cancer is a disease in which malignant (cancer) cells form in tubules of the kidney. Renal cell cancer (also called kidney cancer or renal adenocarcinoma) is a disease in which
How prostate cancer is diagnosed
 How prostate cancer is diagnosed This information is an extract from the booklet Having tests for prostate cancer. You may find the full booklet helpful. We can send you a free copy see page 7. Contents
How prostate cancer is diagnosed This information is an extract from the booklet Having tests for prostate cancer. You may find the full booklet helpful. We can send you a free copy see page 7. Contents
After Diagnosis: Prostate Cancer. Understanding Your Treatment Options
 After Diagnosis: Prostate Cancer Understanding Your Treatment Options What s inside How will this booklet help me?... 3 What is the prostate?... 4 What is prostate cancer?... 4 Who gets prostate cancer?...
After Diagnosis: Prostate Cancer Understanding Your Treatment Options What s inside How will this booklet help me?... 3 What is the prostate?... 4 What is prostate cancer?... 4 Who gets prostate cancer?...
Prostate cancer A guide for newly diagnosed men
 Prostate cancer A guide for newly diagnosed men 2 Prostate cancer A guide for newly diagnosed men About this booklet This booklet is for men who have recently been diagnosed with prostate cancer. It is
Prostate cancer A guide for newly diagnosed men 2 Prostate cancer A guide for newly diagnosed men About this booklet This booklet is for men who have recently been diagnosed with prostate cancer. It is
Incontinence. What is incontinence?
 Incontinence What is incontinence? Broadly speaking, the medical term incontinence refers to any involuntary release of bodily fluids, but many people associate it strongly with the inability to control
Incontinence What is incontinence? Broadly speaking, the medical term incontinence refers to any involuntary release of bodily fluids, but many people associate it strongly with the inability to control
Possible Side Effects of Radiation Therapy on the Prostate Gland
 Possible Side Effects of Radiation Therapy on the Prostate Gland Information compiled by Daniel J. Faber, Coordinator of the Warriors+one, a Breakout Group of the Prostate Cancer Canada Network- Ottawa,
Possible Side Effects of Radiation Therapy on the Prostate Gland Information compiled by Daniel J. Faber, Coordinator of the Warriors+one, a Breakout Group of the Prostate Cancer Canada Network- Ottawa,
Spine University s Guide to Cauda Equina Syndrome
 Spine University s Guide to Cauda Equina Syndrome 2 Introduction Your spine is a very complicated part of your body. It s made up of the bones (vertebrae) that keep it aligned, nerves that channel down
Spine University s Guide to Cauda Equina Syndrome 2 Introduction Your spine is a very complicated part of your body. It s made up of the bones (vertebrae) that keep it aligned, nerves that channel down
150640_Brochure_B 4/12/07 2:58 PM Page 2. Patient Information. Freedom From an Enlarged Prostate
 150640_Brochure_B 4/12/07 2:58 PM Page 2 Patient Information Freedom From an Enlarged Prostate 150640_Brochure_B 4/12/07 2:58 PM Page 3 GreenLight Laser Therapy 1 150640_Brochure_B 4/12/07 2:58 PM Page
150640_Brochure_B 4/12/07 2:58 PM Page 2 Patient Information Freedom From an Enlarged Prostate 150640_Brochure_B 4/12/07 2:58 PM Page 3 GreenLight Laser Therapy 1 150640_Brochure_B 4/12/07 2:58 PM Page
male sexual dysfunction
 male sexual dysfunction lack of desire Both men and women often lose interest in sex during cancer treatment, at least for a time. At first, concern for survival is so overwhelming that sex is far down
male sexual dysfunction lack of desire Both men and women often lose interest in sex during cancer treatment, at least for a time. At first, concern for survival is so overwhelming that sex is far down
Colorectal Cancer Treatment
 Scan for mobile link. Colorectal Cancer Treatment Colorectal cancer overview Colorectal cancer, also called large bowel cancer, is the term used to describe malignant tumors found in the colon and rectum.
Scan for mobile link. Colorectal Cancer Treatment Colorectal cancer overview Colorectal cancer, also called large bowel cancer, is the term used to describe malignant tumors found in the colon and rectum.
Oncology Annual Report: Prostate Cancer 2005 Update By: John Konefal, MD, Radiation Oncology
 Oncology Annual Report: Prostate Cancer 25 Update By: John Konefal, MD, Radiation Oncology Prostate cancer is the most common cancer in men, with 232,9 new cases projected to be diagnosed in the U.S. in
Oncology Annual Report: Prostate Cancer 25 Update By: John Konefal, MD, Radiation Oncology Prostate cancer is the most common cancer in men, with 232,9 new cases projected to be diagnosed in the U.S. in
Screening for Prostate Cancer
 Cancer Expert Working Group on Cancer Prevention and Screening Screening for Prostate Cancer Information for men and their families 1 What is the prostate? 2 What is prostate cancer? prostate The prostate
Cancer Expert Working Group on Cancer Prevention and Screening Screening for Prostate Cancer Information for men and their families 1 What is the prostate? 2 What is prostate cancer? prostate The prostate
Prostate Specific Antigen (PSA) Blood Test
 TO PROVIDE THE VERY BEST CARE FOR EACH PATIENT ON EVERY OCCASION Prostate Specific Antigen (PSA) Blood Test An information guide Prostate Specific Antigen (PSA) Blood Test Prostate Specific Antigen (PSA)
TO PROVIDE THE VERY BEST CARE FOR EACH PATIENT ON EVERY OCCASION Prostate Specific Antigen (PSA) Blood Test An information guide Prostate Specific Antigen (PSA) Blood Test Prostate Specific Antigen (PSA)
Understanding Your Diagnosis of Endometrial Cancer A STEP-BY-STEP GUIDE
 Understanding Your Diagnosis of Endometrial Cancer A STEP-BY-STEP GUIDE Introduction This guide is designed to help you clarify and understand the decisions that need to be made about your care for the
Understanding Your Diagnosis of Endometrial Cancer A STEP-BY-STEP GUIDE Introduction This guide is designed to help you clarify and understand the decisions that need to be made about your care for the
Prostate cancer is the most common cause of death from cancer in men over age 75. Prostate cancer is rarely found in men younger than 40.
 A.D.A.M. Medical Encyclopedia. Prostate cancer Cancer - prostate; Biopsy - prostate; Prostate biopsy; Gleason score Last reviewed: October 2, 2013. Prostate cancer is cancer that starts in the prostate
A.D.A.M. Medical Encyclopedia. Prostate cancer Cancer - prostate; Biopsy - prostate; Prostate biopsy; Gleason score Last reviewed: October 2, 2013. Prostate cancer is cancer that starts in the prostate
to Know About Your Partner s
 What You Need to Know About Your Partner s Prostate Cancer. A Guide for Wives, Partners, and the Men They Love. Providing Support for Your Partner Prostate cancer can be a physical and emotional challenge
What You Need to Know About Your Partner s Prostate Cancer. A Guide for Wives, Partners, and the Men They Love. Providing Support for Your Partner Prostate cancer can be a physical and emotional challenge
Thomas A. Kollmorgen, M.D. Oregon Urology Institute
 Thomas A. Kollmorgen, M.D. Oregon Urology Institute None 240,000 new diagnosis per year, and an estimated 28,100 deaths (2012) 2 nd leading cause of death from cancer in U.S.A. Approximately 1 in 6 men
Thomas A. Kollmorgen, M.D. Oregon Urology Institute None 240,000 new diagnosis per year, and an estimated 28,100 deaths (2012) 2 nd leading cause of death from cancer in U.S.A. Approximately 1 in 6 men
Prostate Cancer Screening
 Prostate Cancer Screening The American Cancer Society and Congregational Health Ministry Team June Module To access this module via the Web, visit www.cancer.org and type in congregational health ministry
Prostate Cancer Screening The American Cancer Society and Congregational Health Ministry Team June Module To access this module via the Web, visit www.cancer.org and type in congregational health ministry
Newly Diagnosed Prostate Cancer: Understanding Your Risk
 Newly Diagnosed Prostate Cancer: Understanding Your Risk When the urologist calls with the life-changing news that your prostate biopsy is positive for prostate cancer, an office appointment is made to
Newly Diagnosed Prostate Cancer: Understanding Your Risk When the urologist calls with the life-changing news that your prostate biopsy is positive for prostate cancer, an office appointment is made to
Introduction Breast cancer is cancer that starts in the cells of the breast. Breast cancer happens mainly in women. But men can get it too.
 Male Breast Cancer Introduction Breast cancer is cancer that starts in the cells of the breast. Breast cancer happens mainly in women. But men can get it too. Many people do not know that men can get breast
Male Breast Cancer Introduction Breast cancer is cancer that starts in the cells of the breast. Breast cancer happens mainly in women. But men can get it too. Many people do not know that men can get breast
da Vinci Prostatectomy Information Guide (Robotically-Assisted Radical Prostatectomy)
 da Vinci Prostatectomy Information Guide (Robotically-Assisted Radical Prostatectomy) Prostate Cancer Overview Prostate cancer is a disease in which malignant (cancer) cells form in the tissues of the
da Vinci Prostatectomy Information Guide (Robotically-Assisted Radical Prostatectomy) Prostate Cancer Overview Prostate cancer is a disease in which malignant (cancer) cells form in the tissues of the
Prostate Cancer Guide. A resource to help answer your questions about prostate cancer
 Prostate Cancer Guide A resource to help answer your questions about prostate cancer Thank you for downloading this guide to prostate cancer treatment. We know that all the information provided online
Prostate Cancer Guide A resource to help answer your questions about prostate cancer Thank you for downloading this guide to prostate cancer treatment. We know that all the information provided online
Screening for Prostate Cancer
 Screening for Prostate Cancer It is now clear that screening for Prostate Cancer discovers the disease at an earlier and more curable stage. It is not yet clear whether this translates into reduced mortality
Screening for Prostate Cancer It is now clear that screening for Prostate Cancer discovers the disease at an earlier and more curable stage. It is not yet clear whether this translates into reduced mortality
GYNECOLOGIC CANCERS Facts to Help Patients Make an Informed Decision
 RADIATION THERAPY FOR GYNECOLOGIC CANCERS Facts to Help Patients Make an Informed Decision TARGETING CANCER CARE AMERICAN SOCIETY FOR RADIATION ONCOLOGY FACTS ABOUT GYNECOLOGIC CANCERS Gynecologic cancers
RADIATION THERAPY FOR GYNECOLOGIC CANCERS Facts to Help Patients Make an Informed Decision TARGETING CANCER CARE AMERICAN SOCIETY FOR RADIATION ONCOLOGY FACTS ABOUT GYNECOLOGIC CANCERS Gynecologic cancers
Localised prostate cancer
 Diagnosis Specialist Nurses 0800 074 8383 prostatecanceruk.org 1 Localised prostate cancer In this fact sheet: What is localised prostate cancer? How is localised prostate cancer diagnosed? What do my
Diagnosis Specialist Nurses 0800 074 8383 prostatecanceruk.org 1 Localised prostate cancer In this fact sheet: What is localised prostate cancer? How is localised prostate cancer diagnosed? What do my
Esophageal Cancer Treatment
 Scan for mobile link. Esophageal Cancer Treatment What is Esophageal Cancer? Esophageal cancer occurs when cancer cells develop in the esophagus, a long, tube-like structure that connects the throat and
Scan for mobile link. Esophageal Cancer Treatment What is Esophageal Cancer? Esophageal cancer occurs when cancer cells develop in the esophagus, a long, tube-like structure that connects the throat and
Fecal Incontinence. What is fecal incontinence?
 Scan for mobile link. Fecal Incontinence Fecal incontinence is the inability to control the passage of waste material from the body. It may be associated with constipation or diarrhea and typically occurs
Scan for mobile link. Fecal Incontinence Fecal incontinence is the inability to control the passage of waste material from the body. It may be associated with constipation or diarrhea and typically occurs
Locally advanced prostate cancer
 Diagnosis Specialist Nurses 0800 074 8383 prostatecanceruk.org 1 Locally advanced prostate cancer In this fact sheet: What is locally advanced prostate cancer? How is locally advanced prostate cancer diagnosed?
Diagnosis Specialist Nurses 0800 074 8383 prostatecanceruk.org 1 Locally advanced prostate cancer In this fact sheet: What is locally advanced prostate cancer? How is locally advanced prostate cancer diagnosed?
Information for Men Receiving Radiation and Hormone Treatments for Prostate Cancer
 Information for Men Receiving Radiation and Hormone Treatments for Prostate Cancer Treatment Plan Stage of prostate cancer: RADIATION THERAPY Radiation Treatments: Date for planning CT: Date to start radiation
Information for Men Receiving Radiation and Hormone Treatments for Prostate Cancer Treatment Plan Stage of prostate cancer: RADIATION THERAPY Radiation Treatments: Date for planning CT: Date to start radiation
WHEN PROSTATE CANCER RETURNS: ADVANCED PROSTATE CANCER. How Will I Know If My Prostate Cancer Returns?
 WHEN PROSTATE CANCER RETURNS: ADVANCED PROSTATE CANCER (The following information is based on the general experiences of many prostate cancer patients. Your experience may be different.) How Will I Know
WHEN PROSTATE CANCER RETURNS: ADVANCED PROSTATE CANCER (The following information is based on the general experiences of many prostate cancer patients. Your experience may be different.) How Will I Know
Advice to patients about the PSA (prostate-specific antigen) blood test: frequently-asked questions
 Advice to patients about the PSA (prostate-specific antigen) blood test: frequently-asked questions What is the PSA blood test? If you want more information before deciding to have this test, it is important
Advice to patients about the PSA (prostate-specific antigen) blood test: frequently-asked questions What is the PSA blood test? If you want more information before deciding to have this test, it is important
Disease/Illness GUIDE TO ASBESTOS LUNG CANCER. What Is Asbestos Lung Cancer? www.simpsonmillar.co.uk Telephone 0844 858 3200
 GUIDE TO ASBESTOS LUNG CANCER What Is Asbestos Lung Cancer? Like tobacco smoking, exposure to asbestos can result in the development of lung cancer. Similarly, the risk of developing asbestos induced lung
GUIDE TO ASBESTOS LUNG CANCER What Is Asbestos Lung Cancer? Like tobacco smoking, exposure to asbestos can result in the development of lung cancer. Similarly, the risk of developing asbestos induced lung
PROSTATE CANCER 101 WHAT IS PROSTATE CANCER?
 PROSTATE CANCER 101 WHAT IS PROSTATE CANCER? Prostate cancer is cancer that begins in the prostate. The prostate is a walnut-shaped gland in the male reproductive system located below the bladder and in
PROSTATE CANCER 101 WHAT IS PROSTATE CANCER? Prostate cancer is cancer that begins in the prostate. The prostate is a walnut-shaped gland in the male reproductive system located below the bladder and in
BRACHYTHERAPY OR RADIOACTIVE SEED IMPLANTATION CANCER OF THE PROSTATE
 BRACHYTHERAPY OR RADIOACTIVE SEED IMPLANTATION CANCER OF THE PROSTATE Prostate cancer, like other cancers, is a disease of the body's cells. Normal cells reproduce themselves by dividing--facilitating
BRACHYTHERAPY OR RADIOACTIVE SEED IMPLANTATION CANCER OF THE PROSTATE Prostate cancer, like other cancers, is a disease of the body's cells. Normal cells reproduce themselves by dividing--facilitating
Prostate Cancer. What is cancer?
 What is cancer? Prostate Cancer The body is made up of trillions of living cells. Normal body cells grow, divide into new cells, and die in an orderly way. During the early years of a person s life, normal
What is cancer? Prostate Cancer The body is made up of trillions of living cells. Normal body cells grow, divide into new cells, and die in an orderly way. During the early years of a person s life, normal
Talking about Prostate Cancer
 Talking about Prostate Cancer Talking about Prostate Cancer If I can avoid going to a doctor, I will but not when we re talking about prostate cancer. Why take chances with a condition that might be serious?
Talking about Prostate Cancer Talking about Prostate Cancer If I can avoid going to a doctor, I will but not when we re talking about prostate cancer. Why take chances with a condition that might be serious?
Colon and Rectal Cancer
 Colon and Rectal Cancer What is colon or rectal cancer? Colon or rectal cancer is the growth of abnormal cells in your large intestine, which is also called the large bowel. The colon is the last 5 feet
Colon and Rectal Cancer What is colon or rectal cancer? Colon or rectal cancer is the growth of abnormal cells in your large intestine, which is also called the large bowel. The colon is the last 5 feet
CONTENTS. What is Cancer?... 2. What Should I Know About Prostate Cancer?... 3. Who's at Risk?... 3 Diagnosis & Staging Tests... 3
 CONTENTS Introduction................................. 1 What is Cancer?.............................. 2 What Should I Know About Prostate Cancer?....... 3 Who's at Risk?................................
CONTENTS Introduction................................. 1 What is Cancer?.............................. 2 What Should I Know About Prostate Cancer?....... 3 Who's at Risk?................................
Understanding. Pancreatic Cancer
 Understanding Pancreatic Cancer Understanding Pancreatic Cancer The Pancreas The pancreas is an organ that is about 6 inches long. It s located deep in your belly between your stomach and backbone. Your
Understanding Pancreatic Cancer Understanding Pancreatic Cancer The Pancreas The pancreas is an organ that is about 6 inches long. It s located deep in your belly between your stomach and backbone. Your
Urinary Incontinence FAQ Sheet
 Urinary Incontinence FAQ Sheet Are you reluctant to talk to your doctor about your bladder control problem? Don t be. There is help. Loss of bladder control is called urinary incontinence. It can happen
Urinary Incontinence FAQ Sheet Are you reluctant to talk to your doctor about your bladder control problem? Don t be. There is help. Loss of bladder control is called urinary incontinence. It can happen
