Randomized Controlled Trial of Cephalexin Versus Clindamycin for Uncomplicated Pediatric Skin Infections
|
|
|
- Lucinda Bennett
- 10 years ago
- Views:
Transcription
1 ARTICLES Randomized Controlled Trial of Cephalexin Versus Clindamycin for Uncomplicated Pediatric Skin Infections AUTHORS: Aaron E. Chen, MD, a Karen C. Carroll, MD, b Marie Diener-West, PhD, c Tracy Ross, MS, b Joyce Ordun, MS, CRNP, a Mitchell A. Goldstein, MD, a Gaurav Kulkarni, MD, a J. B. Cantey, MD, a and George K. Siberry, MD, MPH d Departments of a Pediatrics and b Pathology, Johns Hopkins Medical Institutions, Baltimore, Maryland; c Department of Biostatistics, Johns Hopkins Bloomberg School of Public Health, Baltimore, MD; and d Pediatric, Adolescent, and Maternal AIDS Branch, Eunice Kennedy Shriver National Institute of Child Health and Human Development, National Institutes of Health, Bethesda, Maryland KEY WORDS MRSA, skin infections, abscess, Staphylococcus aureus, clindamycin, cephalexin ABBREVIATIONS CA-MRSA community-associated methicillin-resistant Staphylococcus aureus SSTI skin and soft tissue infection ID incision and drainage ED emergency department D test disk-diffusion induction test MSSA methicillin-susceptible Staphylococcus aureus All authors participated in the concept and design of the study, analysis and interpretation of the data, and drafting or revision of the manuscript and have approved the manuscript as submitted. Registered with clinicaltrials.gov (identifier NCT ). doi: /peds Accepted for publication Dec 2, 2010 Address correspondence to Aaron E. Chen, MD, Johns Hopkins University, Department of Pediatrics, Division of Pediatric Emergency Medicine, CMSC 144, 600 N Wolfe St, Baltimore, MD [email protected] PEDIATRICS (ISSN Numbers: Print, ; Online, ). Copyright 2011 by the American Academy of Pediatrics FINANCIAL DISCLOSURE: The authors have indicated they have no financial relationships relevant to this article to disclose. Funded in part by the National Institutes of Health (NIH). WHAT S KNOWN ON THIS SUBJECT: Community-associated methicillin-resistant Staphylococcus aureus (MRSA) causes the majority of purulent skin infections in children. The choice of adjuvant antibiotic varies widely in clinical practice, with a recent trend toward the empiric use of antibiotics with activity against MRSA. WHAT THIS STUDY ADDS: This randomized controlled trial of cephalexin versus clindamycin for uncomplicated, purulent skin infections showed equally high rates of improvement at 48 to 72 hours and 7 days, which suggests that using antibiotics with in vitro MRSA activity may not be critical for successful treatment. abstract OBJECTIVE: To compare clindamycin and cephalexin for treatment of uncomplicated skin and soft tissue infections (SSTIs) caused predominantly by community-associated (CA) methicillin-resistant Staphylococcus aureus (MRSA). We hypothesized that clindamycin would be superior to cephalexin (an antibiotic without MRSA activity) for treatment of these infections. PATIENTS AND METHODS: Patients aged 6 months to 18 years with uncomplicated SSTIs not requiring hospitalization were enrolled September 2006 through May Eligible patients were randomly assigned to 7 days of cephalexin or clindamycin; primary and secondary outcomes were clinical improvement at 48 to 72 hours and resolution at 7 days. Cultures were obtained and tested for antimicrobial susceptibilities, pulsed-field gel electrophoresis type, and Panton-Valentine leukocidin status. RESULTS: Of 200 enrolled patients, 69% had MRSA cultured from wounds. Most MRSA were USA300 or subtypes, positive for Panton- Valentine leukocidin, and clindamycin susceptible, consistent with CA- MRSA. Spontaneous drainage occurred or a drainage procedure was performed in 97% of subjects. By 48 to 72 hours, 94% of subjects in the cephalexin arm and 97% in the clindamycin arm were improved (P.50). By 7 days, all subjects were improved, with complete resolution in 97% in the cephalexin arm and 94% in the clindamycin arm (P.33). Fevers and age less than 1 year, but not initial erythema 5 cm, were associated with early treatment failures, regardless of antibiotic used. CONCLUSIONS: There is no significant difference between cephalexin and clindamycin for treatment of uncomplicated pediatric SSTIs caused predominantly by CA-MRSA. Close follow-up and fastidious wound care of appropriately drained, uncomplicated SSTIs are likely more important than initial antibiotic choice. Pediatrics 2011;127: e573 e580 PEDIATRICS Volume 127, Number 3, March 2011 e573
2 Since the mid-1990s, an epidemic of community-associated methicillinresistant Staphylococcus aureus (CA- MRSA) infections in healthy, nonhospitalized children has spread throughout the United States and worldwide. 1 3 The majority of these CA-MRSA infections are purulent skin and soft tissue infections (SSTIs), whereas more invasive infections such as sepsis, necrotizing pneumonia, osteomyelitis, septic arthritis, and pyomyositis occur less frequently. 4 Although incision and drainage (ID) is considered essential for the management of abscesses, 5,6 the role of adjuvant antibiotics is unclear. Results of a small, blinded, placebo-controlled trial of adults undergoing ID for uncomplicated skin abscesses that was conducted the mid-1980s showed no advantage of adjuvant antibiotics compared with placebo. 7 Since the emergence of CA-MRSA, findings from observational studies have been inconsistent in regard to the advantage of treating CA-MRSA SSTIs with antibiotics selected on the basis of their in vitro activity. 8,9 During the CA-MRSA era, results of randomized trials in which cephalexin was compared with placebo in adults 10 and trimethoprimsulfamethoxazole was compared with placebo in children 11 have failed to demonstrate a substantial difference in outcome. Since the emergence of CA-MRSA, however, no trial has included a comparison of traditional antistaphylococcal antibiotics with MRSA-active adjuvant antibiotics in the treatment of adults or children. This trial was designed to enable us to compare the clinical outcomes of children with uncomplicated purulent SSTIs who were randomly assigned to receive treatment with cephalexin (a traditional antistaphylococcal antibiotic without activity against MRSA) or clindamycin (an antibiotic with high clinical activity against CA-MRSA in our area). METHODS This study was approved by the institutional review board of the Johns Hopkins Medical Institutions. Written informed consent with assent appropriate for age was obtained by either the research assistant or the treating attending physician from all subjects and/or parents/guardians. Enrollment occurred between September 2006 and May Eligible subjects included patients aged 6 months to 18 years (inclusive) who presented to a pediatric outpatient center at Johns Hopkins (pediatric emergency department [ED] or pediatric outpatient department) with an uncomplicated, purulent SSTI, defined as an abscess (with or without surrounding cellulitis), furuncle, or carbuncle for which outpatient management was anticipated. Exclusion criteria included: hospitalization on initial visit or previous 14 days; hypersensitivity to cephalosporin antibiotics or clindamycin; inherited or acquired altered immunity (such as HIV infection, uncontrolled diabetes mellitus, congenital immunodeficiency); skin infections related to surgical wounds or hardware; and current use of antibiotic therapy. Subjects were equally randomized to receive cephalexin (40 mg/kg per day, taken orally in divided doses administered 3 times per day) or clindamycin (20 mg/kg per day, taken orally in divided doses administered 3 times per day) Patient randomization and assignment to study groups were performed in permuted blocks of 4 by the investigational drug research pharmacy at Johns Hopkins, who also concealed and maintained all assignments. Study drugs were dispensed in amber glass bottles if liquid formulation was used, and both subjects and caregivers were blinded to the study drug received. The objective of this study was to compare the outcomes of children with uncomplicated SSTIs treated with either clindamycin or cephalexin. The primary outcome variable was clinical improvement at 48 to 72 hours from the initiation of treatment, defined as improvement in at least 1 of the measured parameters (overall improvement according to subject or parent/ guardian, fever, erythema, pain/ tenderness, and drainage) without worsening in any of those parameters. The secondary outcome variable was resolution of disease at 7 days, defined as overall improvement according to the subject or parent/guardian in addition to resolution of all variables (fever, erythema, pain/tenderness, and drainage). Our hypothesis was that clindamycin would be superior to cephalexin for both the primary and secondary outcome variables. The sample size calculations were performed on the basis of the main outcome variable of comparison of the proportion of subjects with clinical improvement at 48 to 72 hours in the 2 study groups. On the basis of the results of previous studies, 8 we expected clinical improvement at 48 to 72 hours to occur in 95% of subjects treated with cephalexin. Using sample size calculations for the comparison of the 2 proportions, we estimated a sample size of 178 (88 per group) needed to detect a 15% difference between improvement in the clindamycin and cephalexin groups with a power of 80% and a 2-sided significance level of 5%. We increased the planned sample size by 10% to 100 per group to account for subjects lost to follow-up. Clinical data for children who met inclusion criteria were collected during the initial encounter, and then during repeat visits or by telephone follow-up at 2 to 3 days, 1 week, and 3 months. For the 3-month follow-up, subjects or parents/guardians were interviewed e574 CHEN et al
3 ARTICLES by telephone, and hospital medical records were reviewed for all subjects to capture data regarding any visits for repeat infections. A standardized data abstraction form was used for all visits, and data were recorded by the research assistant, the attending physician treating the patient, or the study coordinator. Conditions for which historical information was obtained at the initial visit included: previous skin infections; asthma; eczema; antibiotics used in the preceding 6 months; and chronic medical problems. For the acute infection, clinical information obtained included: fever history; location of abscess; measured erythema; measured induration; presence of warmth, tenderness, fluctuance, and spontaneous drainage; number of abscesses; procedure performed; and amount of purulent debris obtained. Procedures were categorized as ID with packing, ID without packing, expressed pus without ID, or no purulent drainage. The longest diameter of both erythema and induration of the infection site were measured with a measuring tape. Subjects were instructed to return to either the pediatric ED or the outpatient center for reevaluation 48 to 72 hours after the initial encounter; however, if subjects did not return the follow-up information was obtained by telephone contact. Information obtained at the second visit included whether antibiotics were obtained and taken as prescribed and whether there was a need for change in antibiotics or for additional procedures. A 5-point Likert scale (1, much improved; 2, somewhat improved; 3, unchanged; 4, somewhat worsened; 5, much worsened) was used for the following assessments: overall subjective improvement (according to subject or parent/guardian); fever; erythema (actual repeat measurements were performed if patients were seen in the ED or clinic); pain/tenderness; and drainage. For the 1-week follow-up visit, the subjects/family were contacted by telephone to determine adherence to medications, occurrence of adverse events, and overall subjective improvement. The day of resolution was recorded for the following variables: fever; erythema; pain/tenderness; and drainage. For the 3-month follow-up, subjects/ family were contacted by telephone to obtain a history of interval complications from the initial infection or medications, and any interval SSTIs in subjects. For all subjects the electronic medical record from our institution was also reviewed to extract available data from intervening visits and laboratory results related to SSTIs. Wound specimens were obtained for culture by using the BactiSwab II culturette with modified Stuart s medium (Remel, Lenexa, KS) according to standard ED culturing practices. Specimens were then plated onto BBL Mac- Conkey II sheeps s blood, chocolate, and CNA (colimycin nalidixic) agar; after overnight incubation at 37 C, all cultures were read at 24 and 48 hours. Any potential pathogen was investigated according to standard laboratory protocol, which included identification and susceptibility testing using the BD Phoenix automated microbiology system (BD Diagnostics, Sparks, MD). 15 Isolates were tested for susceptibility to a standard array of antibiotic agents, including cotrimoxazole, clindamycin, erythromycin, tetracycline, moxifloxacin, and vancomycin. The disk-diffusion induction test (D test) 16 was used to detect inducible clindamycin resistance in erythromycinresistant, clindamycin-susceptible staphylococcal isolates. All S aureus isolates were additional subjected to pulsed-field gel electrophoresis performed with standard extraction methods 17 and evaluated for strain relatedness as described by Tenover et al. 18 The first 20 isolates and all subsequent isolates that were not identical to the USA300 strain were tested by polymerase chain reaction for the presence of the genes encoding LukS-PV and LukF-PV, 2 proteins that comprise the Panton-Valentine leukocidin toxin. 19 We used 2 or Fisher s exact test to compare proportions of outcomes or characteristics according to study drug assignment. Stata 11 (Stata Corp, College Station, Texas) was used for all analyses, which were performed on an intentionto-treat basis. A P-value.05 was considered statistically significant. RESULTS Enrollment and Demographic Characteristics Of 220 subjects screened, 200 were enrolled in the study. One hundred subjects were randomly assigned to receive cephalexin and 100 subjects were randomly assigned to receive clindamycin. Three subjects in each arm were lost to follow-up by the first visit, and 1 additional subject in the cephalexin arm and 2 subjects in the clindamycin arm were lost to follow-up at the 7-day visit. In the cephalexin arm 34 of 97 subjects (35%) and in the clindamycin arm 37 of 97 subjects (38%) were followed up at 48 to 72 hours by telephone instead of in the ED or outpatient clinic (P.67). Twenty-eight subjects in the cephalexin arm and 26 subjects in the clindamycin arm could not be reached for the 3-month followup. There were no statistically significant differences between subjects assigned to the cephalexin and clindamycin arms for any of the demographic variables (Table 1). Clinical Characteristics at Baseline There were no significant differences in any characteristics of infection at baseline between subjects in the cephalexin and clindamycin treatment arms (Table 2). PEDIATRICS Volume 127, Number 3, March 2011 e575
4 TABLE 1 Demographic Characteristics of Study Subjects According to Study Drug Assignment Overall Cephalexin Arm Clindamycin Arm P Total no. of subjects NA Prior skin infections Location of enrollment.55 ED Clinic Black Age mo y y y Female gender History of asthma History of eczema Antibiotic use 6 mo Underlying CMC Hospitalization in previous year NA indicates not applicable; CMC, chronic medical condition. TABLE 2 Clinical Characteristics of Study Subjects According to Study Drug Assignment Overall Cephalexin Arm Clindamycin Arm P Total number of subjects NA Fever present Infection characteristics: Tenderness present Fluctuance present Spontaneous drainage Location: below waist Erythema 5cm a Induration 5cm b Drainage procedure.72 No procedure EP without ID ID Needle aspiration Wound culture isolate.56 MRSA MSSA None/other NA indicates not applicable; EP, expressed pus. a Erythema: range, 0 20 cm, median, 4 cm, mean, 4.4 cm. b Induration: range, 0 16 cm, median, 3.5 cm, mean, 3.8 cm. Wound Isolates Of 200 specimens, 137 (69%) grew MRSA, 37 (19%) grew methicillinsusceptible S aureus (MSSA), 16 (8%) grew other organisms (Proteus spp, coagulase-negative staphylococci, streptococci [groups A, B and C], Actinomyces spp), and 10 (5%) were sterile or unable to be collected. According to results of pulsed-field gel electrophoresis, most MRSA (93%) and many MSSA (35%) isolates were identical to USA 300 or related subtypes, and Panton-Valentine leukocidin was detected at high rates in MRSA (99%) and MSSA (82%). Wound isolates demonstrated high rates of susceptibility to clindamycin (91%), tetracyclines (93%), cotrimoxazole (98%), and vancomycin (100%), and lower rates of susceptibility to erythromycin (19%) and moxifloxacin (25%). Three MRSA wound isolates and 1 MSSA wound isolate harbored inducible clindamycin resistance (D-test positive). The proportions of subjects with wound infections caused by MRSA, MSSA, and other pathogens were similar for the cephalexin and clindamycin treatment arms (Table 2). Outcome at 48 to 72 Hours Drug assignments of study subjects were unblinded only after interventions and outcomes were assigned at the end of the study. Three subjects in each study arm were lost to follow-up for this primary outcome variable. Of the remaining subjects, 91 of 97 subjects (94%) in the cephalexin arm and 94 of 97 subjects (97%) in the clindamycin arm showed improvement or resolution in their infection (P.50). The primary infection had worsened in 6 of 97 subjects (6%) in the cephalexin arm and 3 of 97 subjects (3%) in the clindamycin arm (Table 3). For the subset of subjects whose initial wound cultures grew MRSA and were not lost to follow-up (135), 9% (6 of 64) in the cephalexin arm and 3% (2 of 71) in the clindamycin arm had worsened by the 48-to-72 hour visit (P.15). For the subset of subjects for whom an organism was isolated from the initial wound culture and susceptibility data were known (183), 2% (2 of 111) of those who received an antibiotic with in vitro activity against the isolate versus 10% (7 of 72) of those who received an inactive antibiotic had worsened by the 48-to-72 hour visit (P.02). There was no association between gender, size of erythema, size of induration, location of infection, or type of initial drainage procedure on primary outcome; however, there was a significantly lower rate of improvement at 48 to 72 hours among subjects aged 1 year and those with fever (Table 4), results that were unrelated to the study drug given. Hospitalizations Four subjects were hospitalized, all within the first week after enrollment; 2 of these hospitalizations were unre- e576 CHEN et al
5 ARTICLES TABLE 3 Characteristics of Study Patients Whose Condition Was Worsened at the 48-to-72 Hour Follow-up Visit Hospitalized? Secondary Outcome: Status at 7 d Change in Antibiotics? Procedure Improvement at h Subsequent Procedure Spontaneous Drainage Induration, cm Location Erythema, cm Fever at Initial Visit Chronic Condition Gender Study Drug Wound Culture Age, y Patient Study No. Drainage Overall a Fever Erythema Pain/ tenderness 100% Improved or resolved Male Clindamycin MRSA No Thigh 8 5 Yes ID-NP Repeat ID Yes Yes Resolved Male Cephalexin MRSA No Buttocks 4 7 Yes ID-NP Wound Yes Yes Resolved repacked Female Clindamycin MRSA b No Thigh 3 4 Yes EP no ID None Yes No Improved Female Cephalexin MRSA No No Buttocks 6 5 No ID-P None Yes No Resolved 25 1 Male Cephalexin MRSA No Buttocks No ID-NP Repeat ID; Yes No Resolved sedation Female Cephalexin MRSA No Groin 6 5 No ID-NP Repeat ID Yes No Resolved attempted 71 8 Female Clindamycin MSSA No No Thigh 7 5 No ID-P Repeat ID Yes No Resolved 45 9 Female Cephalexin MRSA No No Leg 2 2 No ID-NP None No No Resolved Male Cephalexin MRSA No No Abdomen 5 3 Yes ID-P None No No Resolved Summary, 22% Hospitalized 78% Antibiotics changed 56% Repeat drainage procedure 100% Drainage procedure 5 cm 5 cm 44% Spontaneous drainage 56% Fever 100% Waist and below 100% No chronic condition 33% Clinda 89% MRSA 1 44% Male median or % ID-NP indicates ID without packing; ID-P, ID with packing; EP, expressed pus. a Overall improvement, fever, erythema, pain/tenderness, and drainage were assessed with a 5-point Likert scale: 0, never had symptoms; 1, much improved; 2, somewhat improved; 3, unchanged; 4, somewhat worse; 5, much worse. b Of the 9 patients whose condition had worsened at the 48-to-72 hour follow-up, only patient 170 had a wound-culture organism that was resistant to clindamycin (it was also D-test positive). lated to the SSTI (1 was for acute gastroenteritis and 1 for injuries from a motor vehicle crash). The other 2 subjects, 1 in each study arm, were hospitalized because of worsening of their initial infection (P.73, Table 3). Outcome at 7 Days For the secondary outcome variable of resolution of infection at 7 days, 4 subjects in the cephalexin arm and 5 subjects in the clindamycin arm were lost to follow-up. Of the remaining subjects, 93 of 96 subjects (97%) in the cephalexin arm and 89 of 95 subjects (94%) in the clindamycin arm had clinical resolution by 7 days (P.33). Only 1 subject developed a new SSTI while on therapy; this subject was on studyassigned clindamycin and had an infection caused by MRSA susceptible to clindamycin. Compliance with taking medications as directed was reported by subjects or parents/guardians for 84 of 96 subjects (88%) in the cephalexin arm and 81 of 95 (85%) in the clindamycin arm (P.66). For the subset of subjects whose initial wound cultures grew only MRSA and who were not lost to follow-up (133), 100% (63 of 63) in the cephalexin arm and 94% (66 of 70) in the clindamycin arm had clinical resolution of disease by 7 days (P.12). For the subset of subjects for whom an organism was isolated from the initial wound culture and susceptibility data were known (180), 95% (104 of 109) of those who received an active antibiotic versus 99% (70 of 71) of those who received an inactive antibiotic had clinical resolution of disease by 7 days (P.41). Only baseline fever was associated with a significantly lower rate of resolution at 7 days (Table 4). Three-Month Follow-up According to data obtained from telephone contact (73%) and chart review (100%) at the 3-month follow-up, 36 PEDIATRICS Volume 127, Number 3, March 2011 e577
6 TABLE 4 Primary and Secondary Outcomes According to Erythema, Induration, Fever, Type of Procedure, Location of Infection, Gender, and Age subjects (18%) had a recurrent SSTI. The risk of new SSTI did not differ according to isolation of MRSA versus MSSA from initial wound culture (21% MRSA versus 16% MSSA; P.51) or by cephalexin or clindamycin assignment (20% vs 16%; P.46). Adverse Events There were no serious adverse events related to study treatment. A 13- month-old child developed mild diarrhea with stool positive for Clostridium difficile antigen and toxin 1 week after completing study-assigned treatment with clindamycin. The diarrhea resolved without treatment and repeat stool specimens were negative for C. difficile. DISCUSSION Primary Outcome: Improved at h (n 194), % (n/n) a P Secondary Outcome: Resolved at 7 d (n 191), % (n/n) a Erythema cm 96% (134/139) 96% (132/137) 5 cm 93% (51/55) 93% (50/54) Induration cm 95% (156/164) 96% (154/161) 5 cm 97% (29/30) 93% (28/30) Fever present.03 b.03 b Yes 89% (42/47) 89% (42/47) No 97% (143/147) 97% (140/144) Procedure ID 94% (117/125) 95% (117/123) EP, no ID 98% (63/64) 95% (60/63) None 100% (5/5) 100% (5/5) Location of infection Above waist 99% (78/79) 97% (77/79) Below waist 93% (107/115) 94% (105/112) Gender Male 96% (79/83) 95% (77/81) Female 95% (106/111) 95% (105/110) Age.004 b mo 76% (13/17) 88% (14/16) 1 year 97% (172/177) 96% (168/175) EP, expressed pus. a The total N for both the primary and secondary outcome variable is 200 owing to subjects lost to follow-up. b Statistically significant. Although there is a limited body of evidence to indicate that antibiotics may not be helpful in the management of purulent SSTIs, especially those that have been treated with ID, many clinicians have expressed discomfort with withholding antibiotics for these patients. Results of a recent survey by the New England Journal of Medicine 20 revealed significant variability in clinical practice in the treatment of patients with uncomplicated SSTIs. When surveyed on their management of an uncomplicated, purulent SSTI in a college athlete, nearly 31% responded that they would perform ID alone, 28% responded that they would advocate use of ID plus an oral antibiotic with activity against MSSA, and 41% responded that they would advocate use of ID plus an oral antibiotic with activity against MRSA. Recently, Duong et al reported results of a randomized trial of cotrimoxazole versus placebo for children with uncomplicated SSTIs, which demonstrated no difference in treatment failures between the 2 groups but higher rates of recurrent SSTIs within the first 10 days (but not 3 months) in the pla- P cebo group. 11 At the time of our study design, we did not feel that there was sufficient evidence to justify a true placebo arm, and therefore for this study we chose to compare an antibiotic (clindamycin) with high in vitro activity against CA-MRSA, the predominant organism causing SSTIs in our area, with a traditional antistaphylococcal antibiotic (cephalexin) that has been associated with good clinical outcomes for CA-MRSA SSTIs in observational studies 3,8,21 despite in vitro resistance. Since mid-2004, it has been standard practice at our institution to use clindamycin for patients who present with uncomplicated SSTIs. This change in practice, which was instituted on the basis of reports of safety and efficacy in children with MRSA infections 22 and the perceived importance of antibiotics in the management of SSTIs, has resulted in the significantly increased use of clindamycin despite its numerous disadvantages over traditional antistaphylococcal antibiotics, including increased cost, poor palatability of oral formulations, greater concern for adverse effects (such as C difficile infection), and development of antibiotic resistance. In this double-blinded, randomized trial of children with uncomplicated purulent SSTIs caused predominantly by CA-MRSA we found no difference in clinical outcomes at 2 to 3 days or at 7 days between those children who received cephalexin and those who received clindamycin. This finding was consistent for the overall study and for only those infections caused by MRSA. Nearly all children had active or passive drainage of purulent collections before treatment with antibiotics. Adverse events, including hospitalization for treatment failure, were uncommon and equal in the 2 study arms. A small percentage of subjects with worsening infection at 48 to 72 hours ( 5% of all subjects) were managed with repeat e578 CHEN et al
7 ARTICLES drainage, with or without a change in antibiotics, and all had good outcomes at 7 days. Overall, 95% of SSTIs had resolved within 1 week of entry. One potentially important finding was a slight but statistically significant better outcome at 48 to 72 hours for subjects treated with an antibiotic with in vitro activity. An observational study by Ruhe et al 9 also showed a small increase in the incidence of early treatment failures for antibiotics without in vitro activity compared with antibiotics with in vitro activity. Such results must be interpreted with caution, however, because subjects in our study were randomly assigned to clindamycin versus cephalexin and not to active versus inactive antibiotic, and these results may have been influenced by confounding differences between groups. In practice, there is no way to reliably predict a priori the infecting organism and its susceptibilities. While active development of muchneeded technology for rapid organism identification and susceptibility testing is underway, the clinical scenario facing most practitioners today would in fact be that of choosing an empiric antibiotic on the basis of the local prevalence of MRSA and local susceptibility patterns. The results of this study should reassure clinicians that despite this small difference in early treatment failures according to in vitro antibiotic activity, adverse events were quite uncommon and outcomes at 7 days were uniformly good. Contrary to a previously reported study by Lee et al, 8 in our study we did not find a significant difference in adverse outcomes associated with the initial size of lesion ( 5 cm versus 5 cm). This lack of association of outcome with lesion size was observed even for subjects who had significant surrounding areas of cellulitis (erythema greater than induration). There did seem to be a statistically significant difference in early treatment failures for younger subjects (children aged less than 1 year) compared with older subjects, as well as those presenting with fever. These data suggest that younger age and presence of fever, rather than initial size of lesion, may be more important for the prediction of adverse outcomes and may be more important for predicting the subsequent need for hospitalization. There were several limitations to this study. Because of the relatively small sample size, we likely were unable to detect very small differences in outcomes, but the extremely high rate of favorable outcomes in both study arms (94% and 97%) was a reassuring indication that there was no clinically important difference. A second limitation was that not all infections were attributable to MRSA and not all MRSA were susceptible in vitro to clindamycin, which prevented a true random comparison of antibiotics with in vitro activity to those without. Third, the rates of recurrence by 3 months may have been underestimated because they were determined with data obtained from reports by patients or parents/guardians and abstraction of medical records only from our institution. Fourth, there was no formal method of assessing the success of blinding. Difficulty with the administration of clindamycin because of its unpalatability was considered as a potential contributor to poorer compliance (and thus effectiveness) of clindamycin, but there was not a significant difference in reported rates of compliance with medications. Another limitation was that this study lacked a true placebo arm. Some experts have hypothesized that antibiotics without in vitro activity against infecting organisms may exert some clinical benefit through effects such as immunomodulation, activity against coexisting organisms, or overcoming resistance in vivo by other mechanisms, 10 but evidence of these effects in the treatment of CA-MRSA is limited. The high rate of improvement in the placebo arm of the trial by Duong et al 11 was similar to the rate in this study, and highlights the uncertain benefit of adjunctive antibiotics and the appropriateness of placebo arms in future studies. In this randomized controlled trial of antibiotic treatment for children with uncomplicated SSTIs, the majority of our subjects had spontaneous drainage or a drainage procedure, the cornerstone of management of purulent infections. Contrary to our initial hypothesis, we did not find any significant difference in outcomes at 48 to 72 hours or 7 days that was attributable to the antibiotic used for initial treatment (cephalexin versus clindamycin), although there was a small but statistically significant difference in early treatment failures that was associated with non randomly assigned active versus inactive antibiotics. Until additional studies confirm that adjuvant antibiotics offer no benefit in the management of children with uncomplicated, purulent SSTIs, cephalexin remains a viable empiric antibiotic choice (even in areas with a high prevalence of CA-MRSA) in the context of management that already includes careful drainage of purulent collections, attention to wound care, and appropriate follow-up, especially in children of younger age and with fever. Additional studies are needed to define predictors and effective interventions for the prevention of recurrent infections. ACKNOWLEDGMENTS We thank the Thrasher Research Foundation (award NR-0001) and the Johns Hopkins University School of Medicine General Clinical Research Center (grant M01-RR0052, from the National Center for Research Resources/Na- PEDIATRICS Volume 127, Number 3, March 2011 e579
8 REFERENCES tional Institutes of Health) for their generous support of this project; the patients and families who participated; the faculty and staff of the Johns Hopkins ED and Harriet Lane Clinic; and Dr Neal Halsey, Dr Marie Diener-West, 1. Herold BC, Immergluck LC, Maranan MC, et al. Community-acquired methicillinresistant Staphylococcus aureus in children with no identified predisposing risk. JAMA. 1998;279(8): Naimi TS, LeDell KH, Como-Sabetti K, et al. Comparison of community- and health careassociated methicillin-resistant Staphylococcus aureus infection. JAMA. 2003; 290(22): Moran GJ, Krishnadasan A, Gorwitz RJ, et al. Methicillin-resistant S. aureus infections among patients in the emergency department. N Engl J Med. 2006;355(7): Kaplan SL, Hulten KG, Gonzalez BE, et al. Three-year surveillance of communityacquired Staphylococcus aureus infections in children. Clin Infect Dis. 2005;40(12): Miller LG, Quan C, Shay A, Mostafaie K. A prospective investigation of outcomes after hospital discharge for endemic, communityacquired methicillin-resistant and -susceptible Staphylococcus aureus skin infection. Clin Infect Dis. 2007;44(4): Daum RS. Clinical practice. Skin and softtissue infections caused by methicillinresistant Staphylococcus aureus. N Engl J Med. 2007;357(4): Llera JL, Levy RC. Treatment of cutaneous abscess: a double-blind clinical study. Ann Emerg Med. 1985;14(1): Lee MC, Rios AM, Aten MF, et al. Management and outcome of children with skin and soft tissue abscesses caused by communityacquired methicillin-resistant Staphylococcus aureus. Pediatr Infect Dis J. 2004;23(2): Ruhe JJ, Smith N, Bradsher RW, Menon A. Community-onset methicillin-resistant Staphylococcus aureus skin and soft-tissue infections: impact of antimicrobial therapy on outcome. Clin Infect Dis. 2007;44(6): Rajendran PM, Young D, Maurer T, et al. Randomized, double-blind, placebo-controlled trial of cephalexin for treatment of uncomplicated skin abscesses in a population at risk for community-acquired methicillinresistant Staphylococcus aureus infection. Antimicrob Agents Chemother. 2007;51(11): Duong M, Markwell S, Peter J, Barenkamp S. Randomized, controlled trial of antibiotics in the management of community-acquired skin abscesses in the Pediatric Patient. Ann Emerg Med. 2010;55(5): Feigin RD, Pickering LK, Anderson D et al. Clindamycin treatment of osteomyelitis and septic arthritis in children. Pediatrics. 1975; 55(2): American Academy of Pediatrics. Section 4: antimicrobial agents and related therapy: drug interactions. In: Pickering LK, Baker CJ, Long SS, eds. Red Book: 2009 Report of the Committee on Infectious Diseases. 28th ed. Elk Grove Village, IL: American Academy of Pediatrics; 2009: Greenstone Brand Clindamycin. [package insert] Peapack, NJ: Greenstone LLC; Carroll KC, Borek AP, Burger C, et al. Evaluation of the BD Phoenix automated microbiology system for identification and antimicrobial and Dr Tim Townsend for serving on the data safety and monitoring board for this study. susceptibility testing of staphylococci and enterococci. J Clin Microbiol. 2006;44(6): Lewis JS 2nd, Jorgensen JH. Inducible Clindamycin resistance in Staphylococci: should clinicians and microbiologists be concerned? Clin Infect Dis. 2005;40(2): Murray BE, Singh KV, Heath JD, Sharma BR, Weinstock GM. Comparison of genomic DNAs of different enterococcal isolates using restriction endonucleases with infrequent recognition sites. J Clin Microbiol. 1990;28(9): Tenover FC, Arbeit RD, Goering RV. Interpreting chromosomal DNA restriction patterns produced by pulsed-field gel electrophoresis: criteria for bacterial strain typing. J Clin Microbiol. 1995;33(9): Lina G, Piémont Y, Godail-Gamot F, et al. Involvement of Panton-Valentine leukocidinproducing Staphylococcus aureus in primary skin infections and pneumonia. Clin Infect Dis. 1999;29(5): Hammond SP, Baden LR. Management of skin and soft-tissue infection: polling results. N Engl J Med. 2008;359(15):e Chen AE, Goldstein M, Carroll K, Song X, Perl TM, Siberry GK. Evolving epidemiology of pediatric Staphylococcus aureus cutaneous infections in a Baltimore hospital. Pediatr Emerg Care. 2006;22(10): Frank AL, Marcinak JF, Mangat PD, et al. Clindamycin treatment of methicillin-resistant Staphylococcus aureus infections in children. Pediatr Infect Dis J. 2002;21(6): e580 CHEN et al
Treatment of skin and soft tissue infections due to methicillin-resistant Staphylococcus aureus in adults
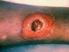 1 of 6 9/24/2010 11:16 AM Official reprint from UpToDate www.uptodate.com 2010 UpToDate Treatment of skin and soft tissue infections due to methicillin-resistant Staphylococcus aureus in adults Author
1 of 6 9/24/2010 11:16 AM Official reprint from UpToDate www.uptodate.com 2010 UpToDate Treatment of skin and soft tissue infections due to methicillin-resistant Staphylococcus aureus in adults Author
 Skin and Soft tissue Infections: new bugs, old drugs Disclosure Statement Sponsor: Goodman Photographic Presented by: Dr. Kristopher Wiebe, MD, CCFP (EM) Presented to: BC Chapter, Canadian Society of Hospital
Skin and Soft tissue Infections: new bugs, old drugs Disclosure Statement Sponsor: Goodman Photographic Presented by: Dr. Kristopher Wiebe, MD, CCFP (EM) Presented to: BC Chapter, Canadian Society of Hospital
Guidance for Industry Acute Bacterial Skin and Skin Structure Infections: Developing Drugs for Treatment
 Guidance for Industry Acute Bacterial Skin and Skin Structure Infections: Developing Drugs for Treatment U.S. Department of Health and Human Services Food and Drug Administration Center for Drug Evaluation
Guidance for Industry Acute Bacterial Skin and Skin Structure Infections: Developing Drugs for Treatment U.S. Department of Health and Human Services Food and Drug Administration Center for Drug Evaluation
Management of Methicillin-Resistant Staphylococcus aureus (MRSA) Infections
 Management of Methicillin-Resistant Staphylococcus aureus (MRSA) Infections Federal Bureau of Prisons Clinical Practice Guidelines April 2012 Clinical guidelines are made available to the public for informational
Management of Methicillin-Resistant Staphylococcus aureus (MRSA) Infections Federal Bureau of Prisons Clinical Practice Guidelines April 2012 Clinical guidelines are made available to the public for informational
Use of Packing for Surgical Wounds. Maggie Benson Clinical Problem Solving II
 Use of Packing for Surgical Wounds Maggie Benson Clinical Problem Solving II Purpose Present patient management s/p Incision and Drainage in an outpatient setting Examine evidence for the use of wound
Use of Packing for Surgical Wounds Maggie Benson Clinical Problem Solving II Purpose Present patient management s/p Incision and Drainage in an outpatient setting Examine evidence for the use of wound
Molluscum BOTE Sign: A Predictor of Imminent Resolution
 Molluscum BOTE Sign: A Predictor of Imminent Resolution abstract Molluscum contagiosum is a common self-limited viral skin infection. The course of the infection often includes tender, crusted, erythematous
Molluscum BOTE Sign: A Predictor of Imminent Resolution abstract Molluscum contagiosum is a common self-limited viral skin infection. The course of the infection often includes tender, crusted, erythematous
A P P E N D I X SAMPLE FORMS
 A P P E N D I X A SAMPLE FORMS Authorization for Disclosure Consent for HBV/HCV Antigens, HIV Antibody Documentation of Staff Education Employees Eligible for Hepatitis-B Vaccination Hepatitis-A Consent
A P P E N D I X A SAMPLE FORMS Authorization for Disclosure Consent for HBV/HCV Antigens, HIV Antibody Documentation of Staff Education Employees Eligible for Hepatitis-B Vaccination Hepatitis-A Consent
Bachir K. Younes, M.D., M.P.H.
 Work: 36923 Cook St. # 103 Palm Desert, CA 92211 Phone (760) 636-1336 Fax (760) 636-1335 Bachir K. Younes, M.D., M.P.H. Personal Born: Jan. 1 st, 1971 in Lebanon Marital Status: Married to Roula Sleilati
Work: 36923 Cook St. # 103 Palm Desert, CA 92211 Phone (760) 636-1336 Fax (760) 636-1335 Bachir K. Younes, M.D., M.P.H. Personal Born: Jan. 1 st, 1971 in Lebanon Marital Status: Married to Roula Sleilati
Hemodialysis catheter infection
 Hemodialysis catheter infection Scary facts In 2006, 82% of patients in the United States initiated dialysis via a catheter The overall likelihood of Tunneled cuffed catheters use was 35% greater in 2005
Hemodialysis catheter infection Scary facts In 2006, 82% of patients in the United States initiated dialysis via a catheter The overall likelihood of Tunneled cuffed catheters use was 35% greater in 2005
Antibiotic Guidelines: Ear Nose and Throat (ENT) Infections. Contents
 Antibiotic Guidelines: Ear Nose and Throat (ENT) Infections. Classification: Clinical Guideline Lead Author: Antibiotic Steering Committee Additional author(s): Authors Division: DCSS & Tertiary Medicine
Antibiotic Guidelines: Ear Nose and Throat (ENT) Infections. Classification: Clinical Guideline Lead Author: Antibiotic Steering Committee Additional author(s): Authors Division: DCSS & Tertiary Medicine
METHICILLIN-RESISTANT STAPHYLOCOCCUS AUREUS (MRSA) COMMUNITY ACQUIRED vs. HEALTHCARE ASSOCIATED
 METHICILLIN-RESISTANT STAPHYLOCOCCUS AUREUS (MRSA) COMMUNITY ACQUIRED vs. HEALTHCARE ASSOCIATED Recently, there have been a number of reports about methicillin-resistant Staph aureus (MRSA) infections
METHICILLIN-RESISTANT STAPHYLOCOCCUS AUREUS (MRSA) COMMUNITY ACQUIRED vs. HEALTHCARE ASSOCIATED Recently, there have been a number of reports about methicillin-resistant Staph aureus (MRSA) infections
C-Difficile Infection Control and Prevention Strategies
 C-Difficile Infection Control and Prevention Strategies Adrienne Mims, MD MPH VP, Chief Medical Officer [email protected] 1/18/2016 1 Disclosure This educational activity does not have commercial
C-Difficile Infection Control and Prevention Strategies Adrienne Mims, MD MPH VP, Chief Medical Officer [email protected] 1/18/2016 1 Disclosure This educational activity does not have commercial
Fungal Infection in Total Joint Arthroplasty. Dr.Wismer Dr.Al-Sahan
 Fungal Infection in Total Joint Arthroplasty Dr.Wismer Dr.Al-Sahan Delayed Reimplantation Arthroplasty for Candidal Prosthetic Joint Infection: A Report of 4 Cases and Review of the Literature David M.
Fungal Infection in Total Joint Arthroplasty Dr.Wismer Dr.Al-Sahan Delayed Reimplantation Arthroplasty for Candidal Prosthetic Joint Infection: A Report of 4 Cases and Review of the Literature David M.
Musculoskeletal Infection Care Process Model
 Musculoskeletal Infection Care Process Model Musculoskeletal infections are serious and potentially life-threatening. Musculoskeletal infections include necrotizing fasciitis, septic arthritis, osteomyelitis,
Musculoskeletal Infection Care Process Model Musculoskeletal infections are serious and potentially life-threatening. Musculoskeletal infections include necrotizing fasciitis, septic arthritis, osteomyelitis,
EAST CAROLINA UNIVERSITY INFECTION CONTROL POLICY. Methicillin-resistant Staph aureus: Management in the Outpatient Setting
 EAST CAROLINA UNIVERSITY INFECTION CONTROL POLICY Methicillin-resistant Staph aureus: Management in the Outpatient Setting Date Originated: Date Reviewed: Date Approved: Page 1 of Approved by: Department
EAST CAROLINA UNIVERSITY INFECTION CONTROL POLICY Methicillin-resistant Staph aureus: Management in the Outpatient Setting Date Originated: Date Reviewed: Date Approved: Page 1 of Approved by: Department
Develop an understanding of the differential diagnosis of pseudomembranous colitis
 Update on Clostridium difficile Colitis Clostridium difficile infection has recently emerged in populations without any known risk factors. This presentation will focus on the historical background, diagnosis,
Update on Clostridium difficile Colitis Clostridium difficile infection has recently emerged in populations without any known risk factors. This presentation will focus on the historical background, diagnosis,
Why Do Some Antibiotics Fail?
 Why Do Some Antibiotics Fail? Patty W. Wright, M.D. April 2010 Objective To outline common reasons why antibiotic therapy is not successful and how this can be avoided. And to teach you a little bit about
Why Do Some Antibiotics Fail? Patty W. Wright, M.D. April 2010 Objective To outline common reasons why antibiotic therapy is not successful and how this can be avoided. And to teach you a little bit about
HOMEOPATHY AS AN ALTERNATIVE TO ANTIBIOTICS
 HOMEOPATHY AS AN ALTERNATIVE TO ANTIBIOTICS Professor Dr. Michael FRASS Medical University of Vienna, Department Medicine I Doctor s Asscociation for Classical Homeopathy www.aekh.at President, Umbrella
HOMEOPATHY AS AN ALTERNATIVE TO ANTIBIOTICS Professor Dr. Michael FRASS Medical University of Vienna, Department Medicine I Doctor s Asscociation for Classical Homeopathy www.aekh.at President, Umbrella
Antimicrobial Stewardship for Hospital Acquired Infection Prevention: Focus on C. difficile infection
 Antimicrobial Stewardship for Hospital Acquired Infection Prevention: Focus on C. difficile infection Emi Minejima, PharmD Assistant Professor of Clinical Pharmacy USC School of Pharmacy [email protected]
Antimicrobial Stewardship for Hospital Acquired Infection Prevention: Focus on C. difficile infection Emi Minejima, PharmD Assistant Professor of Clinical Pharmacy USC School of Pharmacy [email protected]
- 301-17. SICKLE CELL SCREENING AND SELECT TOPICS IN PREVENTION OF COMPLICATIONS Mark Schuster, M.D., Ph.D.
 - 301-17. SICKLE CELL SCREENING AND SELECT TOPICS IN PREVENTION OF COMPLICATIONS Mark Schuster, M.D., Ph.D. We used the following sources to construct indicators for sickle cell disease screening for newborns
- 301-17. SICKLE CELL SCREENING AND SELECT TOPICS IN PREVENTION OF COMPLICATIONS Mark Schuster, M.D., Ph.D. We used the following sources to construct indicators for sickle cell disease screening for newborns
ORTHOPAEDIC INFECTION PREVENTION AND CONTROL: AN EMERGING NEW PARADIGM
 ORTHOPAEDIC INFECTION PREVENTION AND CONTROL: AN EMERGING NEW PARADIGM AMERICAN ACADEMY OF ORTHOPAEDIC SURGEONS 77th Annual Meeting March 9-12, 2010 New Orleans, Louisiana COMMITTEE ON PATIENT SAFETY PREPARED
ORTHOPAEDIC INFECTION PREVENTION AND CONTROL: AN EMERGING NEW PARADIGM AMERICAN ACADEMY OF ORTHOPAEDIC SURGEONS 77th Annual Meeting March 9-12, 2010 New Orleans, Louisiana COMMITTEE ON PATIENT SAFETY PREPARED
Antibiotic-Associated Diarrhea, Clostridium difficile- Associated Diarrhea and Colitis
 Antibiotic-Associated Diarrhea, Clostridium difficile- Associated Diarrhea and Colitis ANTIBIOTIC-ASSOCIATED DIARRHEA Disturbance of the normal colonic microflora Leading to alterations in bacterial degradation
Antibiotic-Associated Diarrhea, Clostridium difficile- Associated Diarrhea and Colitis ANTIBIOTIC-ASSOCIATED DIARRHEA Disturbance of the normal colonic microflora Leading to alterations in bacterial degradation
Blue Team Teaching Module: Periorbital/Orbital Infections
 Blue Team Teaching Module: Periorbital/Orbital Infections Format: 1. Case 2. Topic Summary 3. Questions 4. References Case: A 3-year-old boy presents with 2 days of increasing redness, swelling, and pain
Blue Team Teaching Module: Periorbital/Orbital Infections Format: 1. Case 2. Topic Summary 3. Questions 4. References Case: A 3-year-old boy presents with 2 days of increasing redness, swelling, and pain
GUIDELINES FOR THE MANAGEMENT OF PVL ASSOCIATED STAPHYLOCOCCUS AUREUS INFECTIONS IN THE HOSPITAL ENVIRONMENT
 GUIDELINES FOR THE MANAGEMENT OF PVL ASSOCIATED STAPHYLOCOCCUS AUREUS INFECTIONS IN THE HOSPITAL ENVIRONMENT Post holder responsible for Policy: Directorate/Department responsible for Policy: Lead Nurse
GUIDELINES FOR THE MANAGEMENT OF PVL ASSOCIATED STAPHYLOCOCCUS AUREUS INFECTIONS IN THE HOSPITAL ENVIRONMENT Post holder responsible for Policy: Directorate/Department responsible for Policy: Lead Nurse
If you are signing for a minor child, you refers to your child throughout the consent document.
 CONSENT TO PARTICIPATE IN A CLINICAL RESEARCH STUDY Adult Patient or Parent, for Minor Patient INSTITUTE: National Cancer Institute PRINCIPAL INVESTIGATOR: Raffit Hassan, M.D. STUDY TITLE: Tissue Procurement
CONSENT TO PARTICIPATE IN A CLINICAL RESEARCH STUDY Adult Patient or Parent, for Minor Patient INSTITUTE: National Cancer Institute PRINCIPAL INVESTIGATOR: Raffit Hassan, M.D. STUDY TITLE: Tissue Procurement
Treatment of Opioid Dependence: A Randomized Controlled Trial. Karen L. Sees, DO, Kevin L. Delucchi, PhD, Carmen Masson, PhD, Amy
 Category: Heroin Title: Methadone Maintenance vs 180-Day psychosocially Enriched Detoxification for Treatment of Opioid Dependence: A Randomized Controlled Trial Authors: Karen L. Sees, DO, Kevin L. Delucchi,
Category: Heroin Title: Methadone Maintenance vs 180-Day psychosocially Enriched Detoxification for Treatment of Opioid Dependence: A Randomized Controlled Trial Authors: Karen L. Sees, DO, Kevin L. Delucchi,
Illinois Department of Public Health STATEMENT OF DEFICIENCIES AND PLAN OF CORRECTION. Statement of LICENSURE Violations
 (X1) PROVER/SUPPLIER/CLIA ENTIFICATION NUMBER: (X3) SURVEY D NAME OF PROVER OR SUPPLIER (X4) SUMMARY REGULATORY OR LSC ENTIFYING INFORMATION) PROVER'S PLAN OF CORRECTION Final Observations Statement of
(X1) PROVER/SUPPLIER/CLIA ENTIFICATION NUMBER: (X3) SURVEY D NAME OF PROVER OR SUPPLIER (X4) SUMMARY REGULATORY OR LSC ENTIFYING INFORMATION) PROVER'S PLAN OF CORRECTION Final Observations Statement of
Principles of Disease and Epidemiology. Copyright 2010 Pearson Education, Inc.
 Principles of Disease and Epidemiology Pathology, Infection, and Disease Disease: An abnormal state in which the body is not functioning normally Pathology: The study of disease Etiology: The study of
Principles of Disease and Epidemiology Pathology, Infection, and Disease Disease: An abnormal state in which the body is not functioning normally Pathology: The study of disease Etiology: The study of
Sample Position Description Nurse Practitioner GS-12. Introduction
 Sample Position Description Nurse Practitioner GS-12 Introduction The Nurse Practitioner Position is located within the National Institutes of Health, (Institute, Branch). The nurse practitioner is a Masters
Sample Position Description Nurse Practitioner GS-12 Introduction The Nurse Practitioner Position is located within the National Institutes of Health, (Institute, Branch). The nurse practitioner is a Masters
A Phase 2 Study of Interferon Beta-1a (Avonex ) in Ulcerative Colitis
 A Phase 2 Study of (Avonex ) in Ulcerative Colitis - Study Results - ClinicalTrials.gov A Phase 2 Study of (Avonex ) in Ulcerative Colitis This study has been completed. Sponsor: Biogen Idec Information
A Phase 2 Study of (Avonex ) in Ulcerative Colitis - Study Results - ClinicalTrials.gov A Phase 2 Study of (Avonex ) in Ulcerative Colitis This study has been completed. Sponsor: Biogen Idec Information
Short Report: Failure of Burkholderia pseudomallei to Grow in an Automated Blood Culture System
 Accepted for Publication, Published online October 13, 2014; doi:10.4269/ajtmh.14-0018. The latest version is at http://ajtmh.org/cgi/doi/10.4269/ajtmh.14-0018 In order to provide our readers with timely
Accepted for Publication, Published online October 13, 2014; doi:10.4269/ajtmh.14-0018. The latest version is at http://ajtmh.org/cgi/doi/10.4269/ajtmh.14-0018 In order to provide our readers with timely
Guidance for Industry Diabetes Mellitus Evaluating Cardiovascular Risk in New Antidiabetic Therapies to Treat Type 2 Diabetes
 Guidance for Industry Diabetes Mellitus Evaluating Cardiovascular Risk in New Antidiabetic Therapies to Treat Type 2 Diabetes U.S. Department of Health and Human Services Food and Drug Administration Center
Guidance for Industry Diabetes Mellitus Evaluating Cardiovascular Risk in New Antidiabetic Therapies to Treat Type 2 Diabetes U.S. Department of Health and Human Services Food and Drug Administration Center
Antibiotic Prophylaxis for the Prevention of Infective Endocarditis and Prosthetic Joint Infections for Dentists
 PRACTICE ADVISORY SERVICE FAQ 6 Crescent Road, Toronto, ON Canada M4W 1T1 T: 416.961.6555 F: 416.961.5814 Toll Free: 1.800.565.4591 www.rcdso.org Antibiotic Prophylaxis for the Prevention of Infective
PRACTICE ADVISORY SERVICE FAQ 6 Crescent Road, Toronto, ON Canada M4W 1T1 T: 416.961.6555 F: 416.961.5814 Toll Free: 1.800.565.4591 www.rcdso.org Antibiotic Prophylaxis for the Prevention of Infective
PRIORITY RESEARCH TOPICS
 PRIORITY RESEARCH TOPICS Understanding all the issues associated with antimicrobial resistance is probably impossible, but it is clear that there are a number of key issues about which we need more information.
PRIORITY RESEARCH TOPICS Understanding all the issues associated with antimicrobial resistance is probably impossible, but it is clear that there are a number of key issues about which we need more information.
National Quality Forum Safe Practices for Better Healthcare
 National Quality Forum Safe Practices for Better Healthcare UCLA Health System advocates the National Quality Forum (NQF) endorsed safe practices.this set of safe Practices encompasses 34 practices that
National Quality Forum Safe Practices for Better Healthcare UCLA Health System advocates the National Quality Forum (NQF) endorsed safe practices.this set of safe Practices encompasses 34 practices that
SYNOPSIS. 2-Year (0.5 DB + 1.5 OL) Addendum to Clinical Study Report
 Name of Sponsor/Company: Bristol-Myers Squibb Name of Finished Product: Abatacept () Name of Active Ingredient: Abatacept () Individual Study Table Referring to the Dossier (For National Authority Use
Name of Sponsor/Company: Bristol-Myers Squibb Name of Finished Product: Abatacept () Name of Active Ingredient: Abatacept () Individual Study Table Referring to the Dossier (For National Authority Use
A case of concurrent deep venous thrombosis, pseudoaneurysm, and extremity abscess in an intravenous methamphetamine abuser
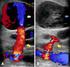 A case of concurrent deep venous thrombosis, pseudoaneurysm, and extremity abscess in an intravenous methamphetamine abuser BY MATTHEW L HARRISON Abstract Introduction. Intravenous drug abuse is a global
A case of concurrent deep venous thrombosis, pseudoaneurysm, and extremity abscess in an intravenous methamphetamine abuser BY MATTHEW L HARRISON Abstract Introduction. Intravenous drug abuse is a global
Diagnosis, Treatment and Prevention of Typhoid Fever in the Children of Bangladesh: A Microbiologist s View.
 Diagnosis, Treatment and Prevention of Typhoid Fever in the Children of Bangladesh: A Microbiologist s View. Samir K Saha, Ph.D. Department of Microbiology Dhaka Shishu Hospital Bangladesh Typhoid Fever:Salmonella
Diagnosis, Treatment and Prevention of Typhoid Fever in the Children of Bangladesh: A Microbiologist s View. Samir K Saha, Ph.D. Department of Microbiology Dhaka Shishu Hospital Bangladesh Typhoid Fever:Salmonella
COMPOUNDING PHARMACY SOLUTIONS PRESCRIPTION COMPOUNDING FOR DERMATOLOGY
 JUNE 2012 COMPOUNDING PHARMACY SOLUTIONS PRESCRIPTION COMPOUNDING WWW.CPSRXS. COM We customize individual prescriptions for the specific needs of our patients. INSIDE THIS ISSUE: Acne 2 Cutaneous Candidiasis
JUNE 2012 COMPOUNDING PHARMACY SOLUTIONS PRESCRIPTION COMPOUNDING WWW.CPSRXS. COM We customize individual prescriptions for the specific needs of our patients. INSIDE THIS ISSUE: Acne 2 Cutaneous Candidiasis
2. The prescribing clinician will register with the designated manufacturer.
 Clozapine Management Program Description Magellan of Arizona Pharmacy Program Background: Magellan Health Services of Arizona recognizes the importance of a clozapine program. Clozapine received increased
Clozapine Management Program Description Magellan of Arizona Pharmacy Program Background: Magellan Health Services of Arizona recognizes the importance of a clozapine program. Clozapine received increased
Ch 14: Principles of Disease and Epidemiology
 Ch 14: Principles of Disease and Epidemiology Learning Objectives Define pathology, etiology, infection, and disease Explain the difference between normal, transient, and opportunistic microbes Compare
Ch 14: Principles of Disease and Epidemiology Learning Objectives Define pathology, etiology, infection, and disease Explain the difference between normal, transient, and opportunistic microbes Compare
Laboratory confirmation requires isolation of Bordetella pertussis or detection of B. pertussis nucleic acid, preferably from a nasopharyngeal swab.
 Pertussis Epidemiology in New Zealand New Zealand has continued to experience outbreaks of pertussis in recent decades. This is in part due to historically low immunisation rates and in part because immunity
Pertussis Epidemiology in New Zealand New Zealand has continued to experience outbreaks of pertussis in recent decades. This is in part due to historically low immunisation rates and in part because immunity
Do general practitioners prescribe more antimicrobials when the weekend comes?
 DOI 10.1186/s40064-015-1505-6 RESEARCH Open Access Do general practitioners prescribe more antimicrobials when the weekend comes? Meera Tandan 1*, Sinead Duane 1 and Akke Vellinga 1,2 Abstract Inappropriate
DOI 10.1186/s40064-015-1505-6 RESEARCH Open Access Do general practitioners prescribe more antimicrobials when the weekend comes? Meera Tandan 1*, Sinead Duane 1 and Akke Vellinga 1,2 Abstract Inappropriate
GENERAL INFORMATION. Adverse Event (AE) Definition (ICH GUIDELINES E6 FOR GCP 1.2):
 Make copies of the blank SAE report form as needed. Retain originals with confirmation of all information faxed to DMID Pharmacovigilance Group Clinical Research Operations and Management Support (CROMS
Make copies of the blank SAE report form as needed. Retain originals with confirmation of all information faxed to DMID Pharmacovigilance Group Clinical Research Operations and Management Support (CROMS
HLA-Cw*0602 associates with a twofold higher prevalence. of positive streptococcal throat swab at the onset of
 1 HLA-Cw*0602 associates with a twofold higher prevalence of positive streptococcal throat swab at the onset of psoriasis: a case control study Lotus Mallbris, MD, PhD, Katarina Wolk, MD, Fabio Sánchez
1 HLA-Cw*0602 associates with a twofold higher prevalence of positive streptococcal throat swab at the onset of psoriasis: a case control study Lotus Mallbris, MD, PhD, Katarina Wolk, MD, Fabio Sánchez
Frequently Asked Questions
 Guidelines for Testing and Treatment of Gonorrhea in Ontario, 2013 Frequently Asked Questions Table of Contents Background... 1 Treatment Recommendations... 2 Treatment of Contacts... 4 Administration
Guidelines for Testing and Treatment of Gonorrhea in Ontario, 2013 Frequently Asked Questions Table of Contents Background... 1 Treatment Recommendations... 2 Treatment of Contacts... 4 Administration
If several different trials are mentioned in one publication, the data of each should be extracted in a separate data extraction form.
 General Remarks This template of a data extraction form is intended to help you to start developing your own data extraction form, it certainly has to be adapted to your specific question. Delete unnecessary
General Remarks This template of a data extraction form is intended to help you to start developing your own data extraction form, it certainly has to be adapted to your specific question. Delete unnecessary
2013 Indiana Healthcare Provider and Hospital Administrator Multi-Drug Resistant Organism Survey
 2013 Indiana Healthcare Provider and Hospital Administrator Multi-Drug Resistant Organism Survey Antibiotic resistance is a global issue that has significant impact in the field of infectious diseases.
2013 Indiana Healthcare Provider and Hospital Administrator Multi-Drug Resistant Organism Survey Antibiotic resistance is a global issue that has significant impact in the field of infectious diseases.
Clostridium Difficile Colitis. Presented by Mark Skains August 2003
 Clostridium Difficile Colitis Presented by Mark Skains August 2003 What is Clostridium Difficile Gram positive rod Produces spores (hang out in diverticula) Forms Endotoxin A + B which cause diarrhea.
Clostridium Difficile Colitis Presented by Mark Skains August 2003 What is Clostridium Difficile Gram positive rod Produces spores (hang out in diverticula) Forms Endotoxin A + B which cause diarrhea.
BD Phoenix Automated Microbiology System
 BD Phoenix Automated Microbiology System Resistance Detection Workflow Efficiency Analysis and Communication BD Diagnostics 7 Loveton Circle Sparks, MD 115-0999 800.638.8663 www.bd.com/ds CHROMagar is
BD Phoenix Automated Microbiology System Resistance Detection Workflow Efficiency Analysis and Communication BD Diagnostics 7 Loveton Circle Sparks, MD 115-0999 800.638.8663 www.bd.com/ds CHROMagar is
INFORMED CONSENT FOR SLEEVE GASTRECTOMY
 INFORMED CONSENT FOR SLEEVE GASTRECTOMY This informed-consent document has been prepared to help inform you about your Sleeve Gastrectomy including the risks and benefits, as well as alternative treatments.
INFORMED CONSENT FOR SLEEVE GASTRECTOMY This informed-consent document has been prepared to help inform you about your Sleeve Gastrectomy including the risks and benefits, as well as alternative treatments.
ESCMID Online Lecture Library. by author
 Do statins improve outcomes of patients with sepsis and pneumonia? Jordi Carratalà Department of Infectious Diseases Statins for sepsis & community-acquired pneumonia Sepsis and CAP are major healthcare
Do statins improve outcomes of patients with sepsis and pneumonia? Jordi Carratalà Department of Infectious Diseases Statins for sepsis & community-acquired pneumonia Sepsis and CAP are major healthcare
Mastitis and Breast Abscess
 1. Purpose This document outlines the guidelines for pharmacological and non pharmacological management of mastitis and breast abscess at the Women s. This guideline is related to the RWH Breastfeeding
1. Purpose This document outlines the guidelines for pharmacological and non pharmacological management of mastitis and breast abscess at the Women s. This guideline is related to the RWH Breastfeeding
White Paper. Medicare Part D Improves the Economic Well-Being of Low Income Seniors
 White Paper Medicare Part D Improves the Economic Well-Being of Low Income Seniors Kathleen Foley, PhD Barbara H. Johnson, MA February 2012 Table of Contents Executive Summary....................... 1
White Paper Medicare Part D Improves the Economic Well-Being of Low Income Seniors Kathleen Foley, PhD Barbara H. Johnson, MA February 2012 Table of Contents Executive Summary....................... 1
MEDICATION MANUAL Policy & Procedure
 MEDICATION MANUAL Policy & Procedure TITLE: Section: Initial Management of Anaphylaxis Following Immunization Medication Specific NUMBER: MM 20-005 Date Issued: October 2009 Source: Distribution: Capital
MEDICATION MANUAL Policy & Procedure TITLE: Section: Initial Management of Anaphylaxis Following Immunization Medication Specific NUMBER: MM 20-005 Date Issued: October 2009 Source: Distribution: Capital
Post-surgical V.A.C. VeraFlo Therapy with Prontosan Instillation on Inpatient Infected Wounds * COLLECTION OF CASE STUDIES
 COLLECTION OF CASE STUDIES Post-surgical V.A.C. VeraFlo Therapy with Prontosan Instillation on Inpatient Infected Wounds * *All patients were treated with systemic antibiotics Post-surgical V.A.C. VeraFlo
COLLECTION OF CASE STUDIES Post-surgical V.A.C. VeraFlo Therapy with Prontosan Instillation on Inpatient Infected Wounds * *All patients were treated with systemic antibiotics Post-surgical V.A.C. VeraFlo
MRSA. Living with. Acknowledgements. (Methicillin-Resistant Staphylococcus aureus)
 How can I keep myself healthy? Hand washing and use of an alcohol-based hand sanitizer are the primary way to prevent acquiring or transmitting bacteria. If you get a cut or scrape, wash it well with soap
How can I keep myself healthy? Hand washing and use of an alcohol-based hand sanitizer are the primary way to prevent acquiring or transmitting bacteria. If you get a cut or scrape, wash it well with soap
GLOBAL CONCERNS ABOUT HPV VACCINES FACT SHEET
 GLOBAL CONCERNS ABOUT HPV VACCINES FACT SHEET When detected, HPV infection is easily managed and rarely proceeds to cancer Very few women with HPV develop cervical cancer HPV infections are only one of
GLOBAL CONCERNS ABOUT HPV VACCINES FACT SHEET When detected, HPV infection is easily managed and rarely proceeds to cancer Very few women with HPV develop cervical cancer HPV infections are only one of
Local Clinical Trials
 Local Clinical Trials The Alzheimer s Association, Connecticut Chapter does not officially endorse any specific research study. The following information regarding clinical trials is provided as a service
Local Clinical Trials The Alzheimer s Association, Connecticut Chapter does not officially endorse any specific research study. The following information regarding clinical trials is provided as a service
Massachusetts Department of Developmental Services MRSA, VRE, and C. Diff Management Protocol
 Massachusetts Department of Developmental Services MRSA, VRE, and C. Diff Management Protocol PURPOSE: To provide guidance for personnel in order to prevent the spread of Antibiotic Resistant Microorganisms
Massachusetts Department of Developmental Services MRSA, VRE, and C. Diff Management Protocol PURPOSE: To provide guidance for personnel in order to prevent the spread of Antibiotic Resistant Microorganisms
The Minnesota Chlamydia Strategy: Action Plan to Reduce and Prevent Chlamydia in Minnesota Minnesota Chlamydia Partnership, April 2011
 The Minnesota Chlamydia Strategy: Action Plan to Reduce and Prevent Chlamydia in Minnesota Minnesota Chlamydia Partnership, April 2011 Section 5: Screening, Treating and Reporting Chlamydia While the information
The Minnesota Chlamydia Strategy: Action Plan to Reduce and Prevent Chlamydia in Minnesota Minnesota Chlamydia Partnership, April 2011 Section 5: Screening, Treating and Reporting Chlamydia While the information
Clinical Infectious Diseases Advance Access published January 4, 2011
 Clinical Infectious Diseases Advance Access published January 4, 2011 IDSA GUIDELINES Clinical Practice Guidelines by the Infectious Diseases Society of America for the Treatment of Methicillin-Resistant
Clinical Infectious Diseases Advance Access published January 4, 2011 IDSA GUIDELINES Clinical Practice Guidelines by the Infectious Diseases Society of America for the Treatment of Methicillin-Resistant
Nursing college, Second stage Microbiology Dr.Nada Khazal K. Hendi L14: Hospital acquired infection, nosocomial infection
 L14: Hospital acquired infection, nosocomial infection Definition A hospital acquired infection, also called a nosocomial infection, is an infection that first appears between 48 hours and four days after
L14: Hospital acquired infection, nosocomial infection Definition A hospital acquired infection, also called a nosocomial infection, is an infection that first appears between 48 hours and four days after
KENTUCKY ADMINISTRATIVE REGULATIONS TITLE 201. GENERAL GOVERNMENT CABINET CHAPTER 9. BOARD OF MEDICAL LICENSURE
 KENTUCKY ADMINISTRATIVE REGULATIONS TITLE 201. GENERAL GOVERNMENT CABINET CHAPTER 9. BOARD OF MEDICAL LICENSURE 201 KAR 9:260. Professional standards for prescribing and dispensing controlled substances.
KENTUCKY ADMINISTRATIVE REGULATIONS TITLE 201. GENERAL GOVERNMENT CABINET CHAPTER 9. BOARD OF MEDICAL LICENSURE 201 KAR 9:260. Professional standards for prescribing and dispensing controlled substances.
COMPOSITION: Each capsule contains clindamycin hydrochloride equivalent to 150 mg clindamycin base.
 APPROVED PACKAGE INSERT DALACIN C 150 mg CAPSULES SCHEDULING STATUS: S4 PROPRIETARY NAME (and dosage form): DALACIN C TM 150 mg (Capsules) COMPOSITION: Each capsule contains clindamycin hydrochloride equivalent
APPROVED PACKAGE INSERT DALACIN C 150 mg CAPSULES SCHEDULING STATUS: S4 PROPRIETARY NAME (and dosage form): DALACIN C TM 150 mg (Capsules) COMPOSITION: Each capsule contains clindamycin hydrochloride equivalent
Version History. Previous Versions. Drugs for MS.Drug facts box fingolimod Version 1.0 Author
 Version History Policy Title Drugs for MS.Drug facts box fingolimod Version 1.0 Author West Midlands Commissioning Support Unit Publication Date Jan 2013 Review Date Supersedes/New (Further fields as required
Version History Policy Title Drugs for MS.Drug facts box fingolimod Version 1.0 Author West Midlands Commissioning Support Unit Publication Date Jan 2013 Review Date Supersedes/New (Further fields as required
ALCHEMIST (Adjuvant Lung Cancer Enrichment Marker Identification and Sequencing Trials)
 ALCHEMIST (Adjuvant Lung Cancer Enrichment Marker Identification and Sequencing Trials) 3 Integrated Trials Testing Targeted Therapy in Early Stage Lung Cancer Part of NCI s Precision Medicine Effort in
ALCHEMIST (Adjuvant Lung Cancer Enrichment Marker Identification and Sequencing Trials) 3 Integrated Trials Testing Targeted Therapy in Early Stage Lung Cancer Part of NCI s Precision Medicine Effort in
Guidelines for the Management of Community-Acquired Methicillin-Resistant Staphylococcus aureus (CA-MRSA) Infections in the US Navy and Marine Corps
 Guidelines for the Management of Community-Acquired Methicillin-Resistant Staphylococcus aureus (CA-MRSA) Infections in the US Navy and Marine Corps AUGUST 2005 Navy Environmental Health Center 620 John
Guidelines for the Management of Community-Acquired Methicillin-Resistant Staphylococcus aureus (CA-MRSA) Infections in the US Navy and Marine Corps AUGUST 2005 Navy Environmental Health Center 620 John
Oral Zinc Supplementation as an Adjunct Therapy in the Management of Hepatic Encephalopathy: A Randomized Controlled Trial
 Oral Zinc Supplementation as an Adjunct Therapy in the Management of Hepatic Encephalopathy: A Randomized Controlled Trial Marcus R. Pereira A. Study Purpose Hepatic encephalopathy is a common complication
Oral Zinc Supplementation as an Adjunct Therapy in the Management of Hepatic Encephalopathy: A Randomized Controlled Trial Marcus R. Pereira A. Study Purpose Hepatic encephalopathy is a common complication
Malpractice and the Infectious Disease Any Physician WHAT YOU SHOULD KNOW! Why this talk? Why me?
 Malpractice and the Infectious Disease Any Physician WHAT YOU SHOULD KNOW! G.R. Donowitz 2015 Why this talk? Why me? Expert witness for a long time Defense and Plaintiff work Have said, No, no case and
Malpractice and the Infectious Disease Any Physician WHAT YOU SHOULD KNOW! G.R. Donowitz 2015 Why this talk? Why me? Expert witness for a long time Defense and Plaintiff work Have said, No, no case and
Drug Utilization and Evaluation of Cephalosporin s At Tertiary Care Teaching Hospital, Bangalore
 Research Article Drug Utilization and Evaluation of Cephalosporin s At Tertiary Care Teaching Hospital, Bangalore HS. Shekar 1*, HR. Chandrashekhar 2, M. Govindaraju 3, P. Venugopalreddy 1, Chikkalingiah
Research Article Drug Utilization and Evaluation of Cephalosporin s At Tertiary Care Teaching Hospital, Bangalore HS. Shekar 1*, HR. Chandrashekhar 2, M. Govindaraju 3, P. Venugopalreddy 1, Chikkalingiah
Basic Results Database
 Basic Results Database Deborah A. Zarin, M.D. ClinicalTrials.gov December 2008 1 ClinicalTrials.gov Overview and PL 110-85 Requirements Module 1 2 Levels of Transparency Prospective Clinical Trials Registry
Basic Results Database Deborah A. Zarin, M.D. ClinicalTrials.gov December 2008 1 ClinicalTrials.gov Overview and PL 110-85 Requirements Module 1 2 Levels of Transparency Prospective Clinical Trials Registry
Streptococcal Infections
 Streptococcal Infections Introduction Streptococcal, or strep, infections cause a variety of health problems. These infections can cause a mild skin infection or sore throat. But they can also cause severe,
Streptococcal Infections Introduction Streptococcal, or strep, infections cause a variety of health problems. These infections can cause a mild skin infection or sore throat. But they can also cause severe,
Community Health Administration
 Community Health Administration 300 North San Antonio Road Santa Barbara, CA 93110-1332 805/681-5439 FAX 805/681-5200 Takashi M. Wada, MD, MPH Director/Health Officer Anne M. Fearon Deputy Director Suzanne
Community Health Administration 300 North San Antonio Road Santa Barbara, CA 93110-1332 805/681-5439 FAX 805/681-5200 Takashi M. Wada, MD, MPH Director/Health Officer Anne M. Fearon Deputy Director Suzanne
Participating in Alzheimer s Disease Clinical Trials and Studies
 Participating in Alzheimer s Disease Clinical Trials and Studies FACT SHEET When Margaret was diagnosed with earlystage Alzheimer s disease at age 68, she wanted to do everything possible to combat the
Participating in Alzheimer s Disease Clinical Trials and Studies FACT SHEET When Margaret was diagnosed with earlystage Alzheimer s disease at age 68, she wanted to do everything possible to combat the
Santa Clara County Tuberculosis Screening Requirement for School Entrance Effective June 1, 2014. Frequently Asked Questions
 Frequently Asked Questions A child has history of BCG vaccination, should they have TST or IGRA? According to the American Academy of Pediatrics Red Book (2012), Interferon Gamma Release Assay (IGRA) is
Frequently Asked Questions A child has history of BCG vaccination, should they have TST or IGRA? According to the American Academy of Pediatrics Red Book (2012), Interferon Gamma Release Assay (IGRA) is
ANNUAL REPORT ON STAPHYLOCOCCUS AUREUS BACTERAEMIA CASES IN DENMARK 2009 (part I)
 ANNUAL REPORT ON STAPHYLOCOCCUS AUREUS BACTERAEMIA CASES IN DENMARK 2009 (part I) STAPHYLOCOCCUS LABORATORY, STATENS SERUM INSTITUT 1 Staphylococcus aureus Bacteraemia Annual Report, Part I The annual
ANNUAL REPORT ON STAPHYLOCOCCUS AUREUS BACTERAEMIA CASES IN DENMARK 2009 (part I) STAPHYLOCOCCUS LABORATORY, STATENS SERUM INSTITUT 1 Staphylococcus aureus Bacteraemia Annual Report, Part I The annual
PROTOCOL SYNOPSIS Evaluation of long-term opioid efficacy for chronic pain
 P a g e 1 PROTOCOL SYNOPSIS Evaluation of long-term opioid efficacy for chronic pain Clinical Phase 4 Study Centers Study Period 25 U.S. sites identified and reviewed by the Steering Committee and Contract
P a g e 1 PROTOCOL SYNOPSIS Evaluation of long-term opioid efficacy for chronic pain Clinical Phase 4 Study Centers Study Period 25 U.S. sites identified and reviewed by the Steering Committee and Contract
Non-Inpatient Parenteral Antibiotic Therapy : clinical and cost effective option
 Non-Inpatient Parenteral Antibiotic Therapy : clinical and cost effective option Dilip Nathwani Ninewells Hospital & Medical School Dundee DD1 9SY TAYSIDE OHPAT EXPERIENCE Case Study Seen in A&E or AMU
Non-Inpatient Parenteral Antibiotic Therapy : clinical and cost effective option Dilip Nathwani Ninewells Hospital & Medical School Dundee DD1 9SY TAYSIDE OHPAT EXPERIENCE Case Study Seen in A&E or AMU
Etiology and treatment of chronic bacterial prostatitis the Croatian experience
 Etiology and treatment of chronic bacterial prostatitis the Croatian experience Višnja Škerk University Hospital for Infectious Diseases "Dr. Fran Mihaljevic" Zagreb Croatia Milano, Malpensa, 14 Nov 2008
Etiology and treatment of chronic bacterial prostatitis the Croatian experience Višnja Škerk University Hospital for Infectious Diseases "Dr. Fran Mihaljevic" Zagreb Croatia Milano, Malpensa, 14 Nov 2008
A list of FDA-approved testosterone products can be found by searching for testosterone at http://www.accessdata.fda.gov/scripts/cder/drugsatfda/.
 FDA Drug Safety Communication: FDA cautions about using testosterone products for low testosterone due to aging; requires labeling change to inform of possible increased risk of heart attack and stroke
FDA Drug Safety Communication: FDA cautions about using testosterone products for low testosterone due to aging; requires labeling change to inform of possible increased risk of heart attack and stroke
Evaluation of a Morphine Weaning Protocol in Pediatric Intensive Care Patients
 Evaluation of a Morphine Weaning Protocol in Pediatric Intensive Care Patients Jennifer Kuhns, Pharm.D. Pharmacy Practice Resident Children s Hospital of Michigan **The speaker has no actual or potential
Evaluation of a Morphine Weaning Protocol in Pediatric Intensive Care Patients Jennifer Kuhns, Pharm.D. Pharmacy Practice Resident Children s Hospital of Michigan **The speaker has no actual or potential
Holy Family University, Student Health Services, Directions for Completion of Health Packet
 1 Holy Family University, Student Health Services, Directions for Completion of Health Packet All forms are to be returned to Health Services by Summer Orientation for the Fall Semester and the first day
1 Holy Family University, Student Health Services, Directions for Completion of Health Packet All forms are to be returned to Health Services by Summer Orientation for the Fall Semester and the first day
Hialeah Nursing and Rehabilitation Center Combines Technology and Best Practices to Improve Infection Control Specific to C.diff
 RESEARCH ARTICLE Page 1 of 5 Hialeah Nursing and Rehabilitation Center Combines Technology and Best Practices to Improve Infection Control Specific to C.diff ABSTRACT RB Health Partners, Inc., June 24,
RESEARCH ARTICLE Page 1 of 5 Hialeah Nursing and Rehabilitation Center Combines Technology and Best Practices to Improve Infection Control Specific to C.diff ABSTRACT RB Health Partners, Inc., June 24,
