SPINAL CORD STIMULATOR
|
|
|
- Jacob Lamb
- 9 years ago
- Views:
Transcription
1 SPINAL CORD STIMULATOR Policy Number: 2016M0002B Effective Date: February 1, 2016 Table of Contents: Page: Cross Reference Policy: POLICY DESCRIPTION 2 Deep Brain Stimulator, 2012M0005A COVERAGE RATIONALE/CLINICAL CONSIDERATION 2 Transcranial Magnetic Stimulation, BACKGROUND M0007A REGULATORY STATUS 6 CLINICAL EVIDENCE 7 Extracorporeal Magnetic Stimulation For APPLICABLE CODES 8 Urinary Incontinence, 2013M0012A REFERENCES 10 Gastric Electrical Stimulation For POLICY HISTORY/REVISION INFORMATION 13 Gastroparesis, 2013M0012A INSTRUCTIONS: Medical Policy assists in administering UCare benefits when making coverage determinations for members under our health benefit plans. When deciding coverage, all reviewers must first identify enrollee eligibility, federal and state legislation or regulatory guidance regarding benefit mandates, and the member specific Evidence of Coverage (EOC) document must be referenced prior to using the medical policies. In the event of a conflict, the enrollee's specific benefit document and federal and state legislation and regulatory guidance supersede this Medical Policy. In the absence of benefit mandates or regulatory guidance that govern the service, procedure or treatment, or when the member s EOC document is silent or not specific, medical policies help to clarify which healthcare services may or may not be covered. This Medical Policy is provided for informational purposes and does not constitute medical advice. In addition to medical policies, UCare also uses tools developed by third parties, such as the InterQual Guidelines, to assist us in administering health benefits. The InterQual Guidelines are intended to be used in connection with the independent professional medical judgment of a qualified health care provider and do not constitute the practice of medicine or medical advice. Other Policies and Coverage Determination Guidelines may also apply. UCare reserves the right, in its sole discretion, to modify its Policies and Guidelines as necessary and to provide benefits otherwise excluded by medical policies when necessitated by operational considerations. Page 1 of 14
2 POLICY DESCRIPTION: This medical policy provides information on the spinal cord stimulator devices which are used to produce electrical stimulation of the spinal nerves to block the sensation of pain. This therapy, also called dorsal column stimulation or neurostimulation, delivers low-voltage, electrical signals to the spinal cord through electrodes surgically placed in the epidural space of the spinal column, and a pulse generator (which contains a battery) implanted into a pocket in the abdomen. The generator and the electrodes are connected by a cable that is tunneled under the skin. The goal of this treatment is to relieve or control a variety of chronic, refractory pain of neurologic origin. COVERAGE RATIONALE / CLINICAL CONSIDERATIONS: A temporary trial of spinal cord stimulation may be CONSIDERED MEDICALLY NECESSARY for the relief of intractable, chronic, neuropathic pain when ALL of the following are present: A. At least ONE of the following conditions: 1. Lumbosacral radiculopathy or radiculitis, or 2. Failed back surgery syndrome (FBSS), post-surgical or posttraumatic, including that of postlaminectomy syndrome, or 3. Chronic pain caused by incomplete spinal cord injury/cauda equina injury, or 4. Chronic pain caused by complex regional pain syndrome I (also known as reflex sympathetic dystrophy), and complex regional pain syndrome II (also known as causalgia), or 5. Chronic pain caused by end-stage peripheral vascular disease (limb ischemia), when the patient cannot undergo revascularization or when revascularization has failed to relieve painful symptoms and the pain has not responded to medical management, or 6. Chronic pain caused by plexopathy, or 7. Chronic pain caused by intercostal neuralgia that did not respond to medical management and nerve blocks, or 8. Phantom limb syndrome/postamputation syndrome, or 9. Chronic pain due to chronic peripheral neuropathy, or 10. Chronic refractory angina pectoris, functional class III or class IV (New York Heart Association) in patients who are not considered candidates for a revascularization procedure, AND B. The patient has failed to achieve satisfactory pain control with treatment modalities over a period of at least 6 months which must include all of the following: 1. Medication management and 2. Conservative treatment which may include alternative treatments, such as acupuncture and chiropractic care, AND must include at least 8 weeks of intensive physical therapy in which the member actively and regularly participates AND C. The patient does not have a condition for which there is a surgical treatment, AND Page 2 of 14
3 D. The patient has undergone a psychological evaluation that has determined that unaddressed psychosocial factors or substance abuse factors do not contribute to the patient s intractable, chronic, neuropathic pain. Note: All facilities, equipment and personnel required for the proper diagnosis, treatment, training and follow-up of the patient must be available. Permanent implantation of a lumbar or thoracic spinal cord stimulator may be CONSIDERED MEDICALLY NECESSARY when the patient has met the above criteria for a temporary trial and has undergone a temporary trial of spinal cord stimulation for a minimum of 2 weeks, and this has resulted in significant improvement in symptoms of intractable, chronic, neuropathic pain. Generally, electronic analysis services (CPT codes ) are not CONSIDERED MEDICALLY NECESSARY when provided at a frequency more often than once every 30 days. More frequent analysis may be necessary in the first month after implantation. Treatment of all other diseases and disorders by an implanted epidural spinal cord stimulator (both temporary and permanent) IS CONSIDERED INVESTIGATIONAL AND NOT MEDICALLY NECESSARY, including, but not limited, to treatment of the following: 1. Central deafferentation pain (related to CNS damage from a stroke or complete spinal cord injury). 2. Chronic pelvic pain. 3. Critical limb ischemia as a technique to forestall amputation. 4. Headache, including but not limited to chronic cluster headaches. 5. Nociceptive pain (resulting from irritation, not damage to the nerves). 6. Postherpetic neuralgia. 7. Visceral pain. 8. Vulvodynia; vulvar vestibulitis. IMPLANTABLE SUBCUTANEOUS TARGET STIMULATOR DEVICES (both temporary and permanent) are CONSIDERED EXPERIMENTAL AND/OR INVESTIGATIONAL AND NOT MEDICALLY NECESSARY for all indications. Clinical Considerations: A. The goals of spinal cord stimulation (SCS) are to create a maximal overlap of SCS-induced paresthesia over the patient's painful area without undesired motor responses or paresthesia beyond the painful site. The devices work best when used on an intact nervous system and are the least effective for patients with damage to the nervous system. Clinical evidence supports the use of spinal cord stimulation for the treatment of intractable chronic pain of the trunk and limbs that has not responded to surgical, pharmacological, and conservative treatment modalities. Documentation in the medical record that medical management has been tried for at least six months and did not provide satisfactory pain control, or are judged unsuitable or contraindicated for the given patient. No medical contraindications to the implantation/spinal surgery (e.g., drug allergies, sepsis, coagulopathy, inability to cope with the technology). Page 3 of 14
4 Documentation from the patient s primary care physician or a mental health professional (i.e., psychiatrist or Ph.D psychologist) that identified any mental health or chemical dependency disorder, that would negatively impact the success of a SCS, is being or has been addressed. Demonstration of at least 50% pain relief with a temporarily implanted electrode precedes permanent implantation. Adverse Effects/Complications: In the evaluation of the risks for implantable devices, observational studies can provide data on the likelihood of potential complications. The following complications for spinal cord stimulation have been reported: o Lead migration, connection failure, and/ lead breakage o Superficial and deep infection with or without abscess o Hematoma o Nerve injury and paralysis o Lack of appropriate paresthesia coverage Medical contraindications to the use of SCS include: o Uncontrolled bleeding disorder o Ongoing anticoagulant therapy o Local or systemic sepsis o Presence of a demand pacemaker or implanted defibrillator o Immunosuppression o Pregnancy o Active or untreated abuse of alcohol, drugs, or medication BACKGROUND: Neuropathic pain refers to pain that is generated and perpetuated by the nervous system itself, without any ongoing stimuli from injury. Examples of neuropathic pain include diabetic neuropathy, post herpetic neuralgia, phantom limb pain, and trigeminal neuralgia. Neuropathic pain can also occur after spinal surgery; this condition is generally known as failed back surgery syndrome (FBSS). Another disorder that appears to be neuropathic in nature is complex regional pain syndrome (CRPS), which can develop spontaneously or after stroke, spinal cord injury, surgery, or peripheral trauma. CRPS is differentiated by type I and type II. Both types have the same basic symptoms, but type I, also known as reflex sympathetic dystrophy (RSD), describes cases with no nerve injury. CRPS type II, also called causalgia, refers to cases with a distinct nerve injury. Neuropathic pain responds poorly to standard pain therapies, can last indefinitely, increasing over time, and often results in severe disability. Therapies used for neuropathic pain include topical agents (capsaicin or topical anesthetics), local or regional nerve blocks, acupuncture or transcutaneous electrical nerve stimulation, physical therapy, behavioral therapy; and drugs, such as antiepileptic drugs, tricyclic antidepressants, and N-methyl-Daspartate (NMDA) receptor antagonists. Narcotic analgesics may be effective initially, but over time increasing doses may be required to control pain and dependency can result. Surgical procedures may be used to relieve nerves that are being compressed by tumors or other tissue masses. If chronic neuropathic pain fails to respond adequately to medical and/or surgical therapies, spinal cord Page 4 of 14
5 stimulation (SCS) may be attempted to provide pain relief. Spinal cord stimulation devices consist of several components: 1) the lead that delivers the electrical stimulation to the spinal cord; 2) an extension wire that conducts the electrical stimulation from the power source to the lead; and 3) a power source that generates the electrical stimulation. The lead may incorporate four to eight electrodes, with eight electrodes more commonly used for complex pain patterns, such as bilateral pain or pain extending from the limbs to the trunk. Two basic types of power sources may be used. In one type, the power source (i.e., battery) can be surgically implanted. In the second type, a radiofrequency receiver is implanted and the power source is worn externally with an antenna over the receiver. Totally implantable systems are most commonly used. The patient's pain distribution pattern dictates the placement level of the stimulation lead in the spinal cord. The pain pattern may influence the type of device used (e.g., a lead with eight electrodes may be selected for those with complex pain patterns or bilateral pain). Implantation of the spinal cord stimulator is typically a two-step process. Initially, the electrode is temporarily implanted percutaneously in the epidural space through a special needle, allowing a trial period of stimulation. Some patients may need an open procedure requiring laminectomy to place the electrodes. After placement of the electrodes, the patient is provided with an external neurostimulator, initially on a trial basis. The trial period may be extended up to four weeks. If during the trial period it is determined that the modality is not effective, or it is not acceptable to the patient, the electrodes may be removed. Once treatment effectiveness is confirmed (defined as at least 50% reduction in pain), the electrodes and radio-receiver/transducer are permanently implanted. Successful spinal cord stimulation may require extensive programming of the neurostimulators to identify the optimal electrode combinations and stimulation of channels. Computer-controlled programs are often used to assist the physician in studying the various programming options when complex systems are used. The first reports of pain relief by direct spinal cord stimulation were published in The first multiprogrammable stimulator was introduced in 1980, and the totally implantable neurostimulator was introduced in In 1988 a noninvasive programmable, implantable pulse generator with radiofrequency capabilities was introduced. Early spinal cord stimulator electrodes required a surgical laminectomy, two to four spinal segments superior to the patient's highest painful dermatome. Today the electrode is introduced through a Touhy needle into the epidural space at the appropriate spinal level for stimulation. Current systems use four or eight electrodes with a power source. This allows reversing the placement without altering the spinal architecture. Implantable Subcutaneous Target Stimulation. A new technique for treating chronic pain is the use of subcutaneous target stimulation (also referred to as peripheral subcutaneous field stimulation). This method involves the use of electrodes implanted directly at the painful area in the subcutaneous space, bypassing the spinal column and nerves. The electrodes are attached to a pulse generator which delivers permanent electrical stimulation. Like the implanted spinal cord stimulators, subcutaneous target stimulators are done in a two-phase process. The first phase involves a temporary trial with the second phase as a permanent implantation following at least a 50% reduction in pain following the temporary trial. Page 5 of 14
6 REGULATORY STATUS: 1. U.S. FOOD AND DRUG ADMINISTRATION (FDA): SCS devices are approved for use as an aid in the management of chronic intractable pain of the trunk and/or limbs, including unilateral or bilateral pain associated with the following: failed back surgery syndrome, intractable low back pain and leg pain (FDA, 2004). They are not specifically approved for the treatment of chronic stable angina. Totally implantable SCS systems are regulated by the FDA as Class III premarket-approval (PMA) devices. Examples of these devices include the PRECISION Plus SCS (Spinal Cord Stimulator) System (Boston Scientific, MA) and the Genesis IPG System (St. Jude Medical, Inc. previously Advanced Neuromodulation Systems, Inc.; Plano, TX). Systems with external transmitters (e.g., X-trel Neurostimulation Systems [Medtronic, Inc., Minneapolis MN]) are regulated by the FDA as Class II 510(k) devices. 2. CENTERS FOR MEDICARE AND MEDICAID SERVICES (CMS): Neuromuscular Electrical Stimulation (NMES) for Spinal Cord Injury (NCD #: 160.7). Two general classifications of electrical nerve stimulators are employed to treat chronic intractable pain: peripheral nerve stimulators and central nervous system stimulators. Relevant to this policy is the Central Nervous System Stimulators (Dorsal Column and Depth Brain Stimulators). The implantation of central nervous system stimulators may be covered as therapies for the relief of chronic intractable pain, subject to the following conditions: Types of Implantations. There are two types of implantations covered by this instruction: Dorsal Column (Spinal Cord) Neurostimulation - The surgical implantation of neurostimulator electrodes within the dura mater (endodural) or the percutaneous insertion of electrodes in the epidural space is covered. Depth Brain Neurostimulation - The stereotactic implantation of electrodes in the deep brain (e.g., thalamus and periaqueductal gray matter) is covered. Conditions for Coverage. No payment may be made for the implantation of dorsal column or depth brain stimulators or services and supplies related to such implantation, unless all of the conditions listed below have been met: The implantation of the stimulator is used only as a late resort (if not a last resort) for patients with chronic intractable pain; Other treatment modalities (pharmacological, surgical, physical, or psychological therapies) have been tried and did not prove satisfactory, or are judged to be unsuitable or contraindicated for the given patient; Patients have undergone careful screening, evaluation and diagnosis by a multidisciplinary team prior to implantation. (Such screening must include psychological, as well as physical evaluation); All the facilities, equipment, professional and support personnel required for the proper diagnosis, treatment, training, and follow up of the patient must be available; and Demonstration of pain relief with a temporarily implanted electrode precedes permanent implantation. Contractors may find it helpful to work with QIOs to obtain the information needed to apply these conditions to claims. Notice: CMS Local Coverage Determination for spinal cord stimulator imposes diagnosis limitations Page 6 of 14
7 that support diagnosis to procedure code automated denials. However, services performed for any given diagnosis must meet all of the indications and limitations stated in this policy, the general requirements for medical necessity as stated in CMS payment policy manuals, any and all existing CMS national coverage determinations, and all Medicare payment rules. 3. MINNESOTA DEPARTMENT OF HUMAN SERVICES (DHS): Minnesota DHS does not have a policy statement regarding spinal cord stimulators in its Provider Manual or other specific provider references. CLINICAL EVIDENCE SUMMARY: Neuropathic Pain: The available evidence for SCS as a treatment of neuropathic trunk and limb pain is mixed and limited by heterogeneity. However, systematic reviews, randomized controlled trials, numerous nonrandomized studies, and most clinical practice guidelines have found support for the use of SCS when all other treatment modalities have failed to adequately reduce symptoms in this patient population for whom there are limited options. There is some evidence that spinal cord stimulation (SCS) can reduce chronic, refractory, neuropathic pain and may improve quality of life (QOL) in patients who have failed back surgery syndrome (FBSS) or complex regional pain syndrome (CRPS). The available literature contained 3 randomized controlled trials (RCTs) or comparative controlled trials evaluating the efficacy of SCS in patients with FBSS, and significant pain reduction compared with reoperation and conventional medical management was evident for 2 years. In the only RCT in patients with CRPS, SCS was more efficacious than physical therapy for 2 years, but not at 3-, 4-, or 5-year follow-up times. Complications arising from SCS implantation required reoperation in 24% to 42% of patients. Overall, the evidence was limited by the small number of studies, small sample sizes and single-site studies, making it difficult to evaluate the generalizability of the results. In addition, none of the trials included a placebo control, which makes it difficult to evaluate the magnitude of the treatment effect. There is insufficient evidence to determine whether position-adaptive or high-frequency SCS improves outcomes or whether methods or factors other than a trial of SCS with temporary electrodes can improve the prognosis of SCS for neuropathic pain. Definitive patient selection criteria for SCS as a treatment for neuropathic pain could not be established due to the limited amount of evidence of low quality. Intractable Angina Pectoris: Evidence from a small number of randomized controlled or comparative trials and a limited number of nonrandomized or uncontrolled studies suggests that SCS can reduce the frequency and severity of anginal pain in some patients with chronic, medically refractory angina, and that some also experience an increase in exercise tolerance and improvement in quality-of-life measures. However, most of the available studies were limited by lack of adequate controls or blinding, small sample size, short duration of follow-up, and use of subjective outcome measures. SCS appeared to be a relatively safe therapy, with few serious adverse effects reported. There was no evidence that the analgesic effect of SCS masks the warning pain of myocardial infarction, and patients who have been treated with SCS have not been shown to be at increased risk for morbidity or mortality compared with their peers. Although there are some promising results from a small number of studies, data from larger, well-designed, randomized controlled trials are required to confirm that SCS is an effective pain management strategy for patients with refractory angina and to establish appropriate patient selection criteria. Implantable Subcutaneous Target Stimulation: Subcutaneous target stimulation appears to be a Page 7 of 14
8 promising alternative to implanted spinal cord stimulators for the treatment of chronic pain, however current literature is limited to small group sizes, case series, and retrospective reviews. Further long-term follow-up evaluations and controlled trials are necessary to determine safety and efficacy. APPLICABLE CODES: The Current Procedural Terminology (CPT ) codes and HCPCS codes listed in this policy are for reference purposes only. Listing of a service or device code in this policy does not imply that the service described by this code is a covered or non-covered health service. The inclusion of a code does not imply any right to reimbursement or guarantee claims payment. Other medical policies and coverage determination guidelines may apply. HCPCS Codes C1767 C1778 C1787 C1816 C1820 C1883 C1897 L8679 L8680 L8681 L8682 L8683 L8685 L8686 L8687 L8688 L8689 L8695 CPT Codes Description Generator, neurostimulator (implantable), nonrechargeable Lead, neurostimulator (implantable) Patient programmer, neurostimulator Receiver and/or transmitter, neurostimulator (implantable) Generator, neurostimulator (implantable), with rechargeable battery and charging system Adaptor/extension, pacing lead or neurostimulator lead (implantable) Lead, neurostimulator test kit (implantable) Implantable neurostimulator, pulse generator, any type Implantable neurostimulator electrode (with any number of contact points), each Patient programmer (external) for use with implantable programmable neurostimulator pulse generator Implantable neurostimulator radiofrequency receiver Radiofrequency transmitter (external) for use with implantable neurostimulator radiofrequency receiver Implantable neurostimulator pulse generator, single array, rechargeable, includes extension Implantable neurostimulator pulse generator, single, array, nonrechargeable, includes extension Implantable neurostimulator pulse generator, dual array, rechargeable, includes extension Implantable neurostimulator pulse generator, dual array, onrechargeable, includes extension External recharging system for battery (internal) for use with implantable neurostimulator External recharging system for battery (external) for use with implantable neurostimulator, replacement only Description Percutaneous implantation of neurostimulator electrode array, epidural Laminectomy for implantation of neurostimulator electrodes, plate/paddle, epidural Removal of spinal neurostimulator electrode percutaneous array(s), including fluoroscopy, when performed Removal of spinal neurostimulator electrode plate/paddle(s) placed via laminotomy or laminectomy, including fluoroscopy, when performed Revision including replacement, when performed, of spinal neurostimulator electrode percutaneous array(s), including fluoroscopy, when performed Revision including replacement, when performed, of spinal neurostimulator electrode plate/paddle(s) placed via laminotomy or laminectomy, including fluoroscopy, when performed Page 8 of 14
9 63685 Insertion or replacement of spinal neurostimulator pulse generator or receiver, direct or inductive coupling Revision or removal of implanted spinal neurostimulator pulse generator or receiver Electronic analysis of implanted neurostimulator pulse generator system (e.g., rate, pulse amplitude and duration, configuration of wave form, battery status, electrode selectability, output modulation, cycling, impedance and patient compliance measurements); simple or complex brain, spinal cord, or peripheral (i.e., cranial nerve, peripheral nerve, autonomic nerve, neuromuscular) neurostimulator pulse generator/transmitter, without reprogramming Electronic analysis of implanted neurostimulator pulse generator system (e.g., rate, pulse amplitude and duration, configuration of wave form, battery status, electrode selectability, output modulation, cycling, impedance and patient compliance measurement(s); simple spinal cord, or peripheral (i.e., peripheral nerve, autonomic nerve, neuromuscular) neurostimulator pulse generator/transmitter, with intraoperative or subsequent programming Electronic analysis of implanted neurostimulator pulse generator system (e.g., rate, pulse amplitude and duration, configuration of wave form, battery status, electrode selectability, output modulation, cycling, impedance and patient compliance measurement(s); complex spinal cord, or peripheral (except cranial nerve) neurostimulator pulse generator/transmitter, with intraoperative or subsequent programming, first hour Electronic analysis of implanted neurostimulator pulse generator system (e.g., rate, pulse amplitude and duration, configuration of wave form, battery status, electrode selectability, output modulation, cycling, impedance and patient compliance measurement(s); complex spinal cord, or peripheral (except cranial nerve) neurostimulator pulse generator/transmitter, with intraoperative or subsequent programming, each additional 30 minutes after first hour 0282T Percutaneous or open implantation of neurostimulator electrode array(s), subcutaneous (peripheral subcutaneous field stimulation), including imaging guidance, when performed, cervical, thoracic or lumbar; for trial, including removal at the conclusion of trial period 0283T Percutaneous or open implantation of neurostimulator electrode array(s), subcutaneous (peripheral subcutaneous field stimulation), including imaging guidance, when performed, cervical, thoracic or lumbar; permanent, with implantation of a pulse generator 0284T Revision or removal of pulse generator or electrodes, including imaging guidance, when performed, including addition of new electrodes, when performed 0285T Electronic analysis of implanted peripheral subcutaneous field stimulation pulse generator, with reprogramming when performed ICD-9 Codes Description Implantation or replacement of spinal neurostimulator lead(s) Removal of spinal neurostimulator lead(s) Incision with removal of foreign body or device from skin and subcutaneous tissue Insertion or replacement of single array neurostimulator pulse generator, not specified as rechargeable Insertion or replacement of dual array neurostimulator pulse generator, not specified as rechargeable Insertion or replacement of other neurostimulator pulse generator Insertion or replacement of single array rechargeable neurostimulator pulse generator Insertion or replacement of dual array rechargeable neurostimulator pulse generator ICD-10 Codes 00HU0MZ- 00HU4MZ 00HV0MZ- Description Insertion of neurostimulator lead into spinal canal [by approach; includes codes 00HU0MZ, 00HU3MZ, 00HU4MZ] Insertion of neurostimulator lead into spinal cord [by approach; includes codes 00HV0MZ, Page 9 of 14
10 00HV4MZ 00HV3MZ, 00HV4MZ] G03.0-G03.9 Meningitis due to other and unspecified causes (arachnoiditis) G54.0-G54.9 Nerve root and plexus disorders G55 Nerve root and plexus compressions in diseases classified elsewhere G56.00-G56.92 Mononeuropathies of upper limb G57.00-G57.92 Mononeuropathies of lower limb G58.0-G58.9 Other mononeuropathies G59 Mononeuropathy in diseases classified elsewhere G89.11 Acute pain due to trauma G89.21 Chronic pain due to trauma G89.4 Chronic pain syndrome G90.50-G90.59 Complex regional pain syndrome I (CRPS I) M50.00-M50.93 Cervical disc disorders M51.04-M51.9 Thoracic, thoracolumbar, and lumbosacral intervertebral disc disorders M53.0-M53.9 Other and unspecified dorsopathies, not elsewhere classified M54.00-M54.9 Dorsalgia CPT is a registered trademark of the American Medical Association. REFERENCES: 1. American Society of Anesthesiologists (ASA) Website. Statement on anesthetic care during interventional pain procedures for adults. October 20, Available at: Accessed November 17, American Society of Anesthesiologists, Inc. Practice guidelines for chronic pain management: an updated report by the American Society of Anesthesiologists task force on chronic pain management and the American Society of Regional Anesthesia and Pain Medicine. Anesthesiology. 2010; 112(4): Andréll P, Yu W, Gersbach P, Gillberg L, Pehrsson K, Hardy I, Ståhle A, Andersen C, Mannheimer C. Long-term effects of spinal cord stimulation on angina symptoms and quality of life in patients with refractory angina pectoris--results from the European Angina Registry Link Study (EARL). Heart Jul;96(14): Bell GK, Kidd D, North RB. Cost-effectiveness analysis of spinal cord stimulation in treatment of failed back surgery syndrome. J Pain Symptom Manage. 1997;13(5): Bondesson S, Pettersson T, Erdling A, Hallberg IR, Wackenfors A, Edvinsson L. Comparison of patients undergoing enhanced external counterpulsation and spinal cord stimulation for refractory angina pectoris. Coron Artery Dis Dec;19(8): Börjesson M, Andrell P, Lundberg D, Mannheimer C. Spinal cord stimulation in severe angina pectoris--a systematic review based on the Swedish Council on Technology assessment in health care report on long-standing pain. Pain Dec;140(3): Burgher AH, Huntoon MA, Turley TW, et al. Subcutaneous Peripheral Nerve Stimulation with Inter-lead Stimulation for Axial Neck and Low Back Pain: Case Series and Review of the Literature. Neuromodulation Aug Cameron T. Safety and efficacy of spinal cord stimulation for the treatment of chronic pain: a 20 year literature review. J Neurosurg (spine3). 2004; 100 (3 Suppl Spine): Centers for Medicare & Medicaid Services (CMS). National coverage determination (NCD) for electrical nerve stimulators (160.7). August 7, Available at: Accessed November 17, Chou R, Loeser J, Owens D, et al. Interventional therapies, surgery, and interdisciplinary rehabilitation for low back pain an evidence based clinical practice guideline from the American Pain Society. Spine. 2009;34: Page 10 of 14
11 11. de Jongste, MJ, Hautvast, RW, Hillege, HL, Lie, KI. Efficacy of spinal cord stimulation as adjuvant therapy for intractable angina pectoris: a prospective, randomized clinical study. Working Group on Neurocardiology. J Am Coll Cardiol Jun;23(7): de Vries J, Dejongste MJ, Durenkamp A, Zijlstra F, Staal MJ. The sustained benefits of long-term neurostimulation in patients with refractory chest pain and normal coronary arteries. Eur J Pain Apr;11(3): Deer TR, Raso LJ. Spinal cord stimulation for refractory angina pectoris and peripheral vascular disease. Pain Physician Oct;9(4): Diedrichs H, Zobel C, Theissen P, et al. Symptomatic relief precedes improvement of myocardial blood flow in patients under spinal cord stimulation. Curr Control Trials Cardiovasc Med. 2005; 6(1): Dyer MT, Goldsmith K, Khan S, Sharples L, Freeman C, Hardy I, Buxton M, Schofield P. Clinical and costeffectiveness analysis of an open label, single-centre, randomised trial of spinal cord stimulation (SCS) versus percutaneous myocardial laser revascularisation (PMR) in patients with refractory angina pectoris: The SPiRiT trial. Trials Jun 30;9: Eddicks S, Maier-Hauff K, Schenk M, Muller A, Baumann G, Theres H.Thoracic spinal cord stimulation improves functional status and relieves symptoms in patients with refractory angina pectoris: the first placebo-controlled randomised study. Heart May;93(5): Foletti A, Durrer A, Buchser E. Neurostimulation technology for the treatment of chronic pain: a focus on spinal cord stimulation. Expert Rev Med Devices Mar;4(2): Frey, ME, Manchikanti, L, Benyamin, RM, Schultz, DM, Smith, HS, Cohen, SP. Spinal cord stimulation for patients with failed back surgery syndrome: a systematic review. Pain Physician Mar-Apr;12(2): PMID: Hautvast, RW, DeJongste, MJ, Staal, MJ, van Gilst, WH, Lie, KI. Spinal cord stimulation in chronic intractable angina pectoris: a randomized, controlled efficacy study. Am Heart J Dec;136(6): Hayes, Winifred S. Medical Technology Directory. Spinal Cord Stimulation for Relief of Neuropathic Pain. October 8, Available at: Accessed November 17, Hayes, Winifred S. Medical Technology Directory. Electrical Spinal Cord Stimulation for the Treatment of Intractable Angina Pectoris. October 23, Reviewed: October 26, Available at: Accessed November 17, Institute for Clinical Systems Improvement (ICSI) Website. Health care guideline: assessment and management of chronic pain. Sixth edition. December Available at: Accessed November 17, Kemler MA, Barendse GA, van Kleef M, et al. Spinal cord stimulation in patients with chronic reflex sympathetic dystrophy. N Engl J Med. 2000; 343(9): Kemler MA, de Vet HC, Barendse GA et al. Spinal cord stimulation for chronic reflex sympathetic dystrophy--fiveyear follow-up. N Engl J Med. 2006; 354(22): Kemler MA, Furnee CA. Economic evaluation of spinal cord stimulation for chronic reflex sympathetic dystrophy. Neurology. 2002;59(8): Klomp HM, Spinc le GH, Steyerberg EW, et al. Spinal cord stimulation in critical limb ischemia: a randomized trial. Lancet.1999; 353(9158): Klomp HM, Steyerberg EW, Habbema JD, van Urk H; ESES study group. What is the evidence on efficacy of spinal cord stimulation in (subgroups of) patients with critical limb ischemia? Ann Vasc Surg. 2009; 23(3): Kumar K, Taylor RS, Jacques L, et al. Spinal cord stimulation versus conventional medical management for neuropathic pain: a multicentre randomized controlled trial in patients with failed back surgery syndrome Pain. 2007; 132(1-2): Lapenna E, Rapati D, Cardano P, De Bonis M, Lullo F, Zangrillo A, Alfieri O. Spinal cord stimulation for patients with refractory angina and previous coronary surgery. Ann Thorac Surg Nov;82(5): Mailis-Gagnon A, Furlan AD, Sandoval JA, Taylor R. Spinal cord stimulation for chronic pain. Cochrane Database Page 11 of 14
12 Syst Rev. 2004; (3):CD Manchikanti L, Boswell MV, Singh V, et al. Comprehensive evidence based guidelines for interventional techniques in the management of chronic spinal pain. Pain Physician. 2009;12(4): Mannheimer C, Camici P, Chester MR, Collins A, DeJongste M, Eliasson T, Follath F, Hellemans I, Herlitz J, Luscher T, Pasic M, Thelle D. The problem of chronic refractory angina; report from the ESC Joint Study Group on the Treatment of Refractory Angina. Eur Heart J Mar;23(5): Mannheimer, C, Eliasson, T, Augustinsson, LE, et al. Electrical stimulation versus coronary artery bypass surgery in severe angina pectoris: the ESBY study. Circulation Mar 31;97(12): McNab D, Khan SN, Sharples LD, Ryan JY, Freeman C, Caine N, Tait S, Hardy I, Schofield PM. An open label, singlecentre, randomized trial of spinal cord stimulation vs. percutaneous myocardial laser revascularization in patients with refractory angina pectoris: the SPiRiT trial. Eur Heart J May;27(9): Mekhail, NA, Mathews, M, Nageeb, F, Guirguis, M, Mekhail, MN, Cheng, J. Retrospective review of 707 cases of spinal cord stimulation: indications and complications. Pain Pract Mar;11(2): PMID: National Guideline Clearinghouse Website. Unstable angina and NSTEMI: the early management of unstable angina and non-st-segment-elevation myocardial infarction. Available at: Accessed November 17, National Guideline Clearinghouse Website. American College of Occupational and Environmental Medicine (ACOEM). Low back disorders. Occupational medicine practice guidelines: evaluation and management of common health problems and functional recovery in workers. Second edition; National Guideline Clearinghouse Website. American College of Occupational and Environmental Medicine (ACOEM). Chronic pain. Occupational medicine practice guidelines: evaluation and management of common health problems and functional recovery in workers National Guideline Clearinghouse Website. Work Loss Data Institute (WLDI). Low back lumbar & thoracic (acute & chronic) National Guideline Clearinghouse Website. Work Loss Data Institute (WLDI). Pain (chronic) National Institute for Health and Clinical Excellence (NICE) Website. Spinal cord stimulation for chronic pain of neuropathic or ischaemic origin. November North RB, Kidd DH, Lee MS, et al. A prospective, randomized study of spinal cord stimulation versus reoperation for failed back surgery syndrome: initial results. Stereotact Funct Neurosurg. 1994; 62(1-4): North, RB, Kidd, D, Shipley, J, Taylor, RS. Spinal cord stimulation versus reoperation for failed back surgery syndrome: a cost effectiveness and cost utility analysis based on a randomized, controlled trial. Neurosurgery Aug;61(2):361-8; discussion 8-9. PMID: North, RB, Kidd, DH, Farrokhi, F, Piantadosi, SA. Spinal cord stimulation versus repeated lumbosacral spine surgery for chronic pain: a randomized, controlled trial. Neurosurgery. 2005;56(1):98-106; discussion -7. PMID: Sator-Katzenschlager S, Fiala K, Kress HG, et al. Subcutaneous target stimulation (STS) in chronic noncancer pain: a nationwide retrospective study. Pain Pract. 2010; 10(4): Simpson EL, Duenas A, Holmes MW, Papaioannou D, Chilcott J. Spinal cord stimulation for chronic pain of neuropathic or ischaemic origin: systematic review and economic evaluation. Health Technol Assess Mar;13(17):iii, ix-x, Stanton-Hicks MD, Burton AW, Bruehl SP, Carr DB, Harden N, Hassenbusch SJ, et al. An Updated Interdisciplinary Clinical Pathway for CRPS: Report of an Expert Panel. Pain practice Nov (2): Taylor RS, De Vries J, Buchser E, Dejongste MJ. Spinal cord stimulation in the treatment of refractory angina: systematic review and meta-analysis of randomised controlled trials. BMC Cardiovasc Disord Mar 25;9: Taylor RS. Spinal cord stimulation in complex regional pain syndrome and refractory neuropathic back and leg pain/failed back surgery syndrome: results of a systematic review and meta-analysis. J Pain Symptom Manage. Page 12 of 14
13 2006; 31(4 Suppl):S UpToDate Website. Cancer pain management: interventional therapies. October Available at: Accessed November 17, UpToDate Website. Lower extremity peripheral arterial disease in end stage renal disease. April Available at: Accessed November 17, UpToDate Website. New therapies for angina pectoris. August Available at: Accessed November 17, UpToDate Website. Overview of the treatment of chronic pain. August Available at: Accessed November 17, UpToDate Website. Prevention and management of complex regional pain syndrome in adults. August Available at: Accessed November 17, UpToDate Website. Subacute and chronic low back pain: surgical treatment. March Available at: Accessed November 17, UpToDate Website. Treatment of lower extremity critical limb ischemia. July Available at: Accessed November 17, POLICY HISTORY: DATE ACTION/DESCRIPTION 09/25/2012 New policy 2012M0002A. Approved by the Interim Medical Policy Committee. 02/28/2013 Reviewed and approved by the Quality Improvement Advisory and Credentialing Council (QIACC). 11/15/2013 Published to UCare.org 03/01/2014 Policy Revision : Policy Number has changed to 2014M0002B Revised Coverage Rationale: Added implantable subcutaneous target stimulator devices to Investigational and Not Medically Necessary statement. Clarified medically necessary criteria for Chronic Refractory Angina Pectoris. Updated Background, Clinical Evidence, and References sections. Updated Coding section to include 01/01/2014 HCPCS changes and ICD-10 codes. 03/12/2014 New Policy. Reviewed by the Medical Policy Committee. 03/27/2014 Reviewed and approved by the Quality Improvement Advisory and Credentialing Committee (QIACC). 12/7/2015 Revised Policy reviewed by the Medical Policy Committee. Coverage rationale unchanged but rewritten for clarity. 12/17/2015 Reviewed and approved by the Quality Improvement Advisory and Credentialing Committee (QIACC). 01/01/2016 Published to UCare.org Page 13 of 14
14 QUESTIONS AND ANSWERS: Q1: A1: ATTACHMENTS: A- New York Heart Association and Canadian Cardiovascular Society Functional Classifications Class I II III IV New York Heart Association Functional Classification Patients with cardiac disease but without resulting limitations of physical activity. Ordinary physical activity does not cause undue fatigue, palpitation, dyspnea, or anginal pain. Patients with cardiac disease resulting in slight limitation of physical activity. They are comfortable at rest. Ordinary physical activity results in fatigue, palpitation, dyspnea, or anginal pain. Patients with cardiac disease resulting in marked limitation of physical activity. They are comfortable at rest. Less than ordinary physical activity causes fatigue, palpitation, dyspnea, or anginal pain. Patient with cardiac disease resulting in inability to carry on any physical activity without discomfort. Symptoms of cardiac insufficiency or of the anginal syndrome may be present even at rest. If any physical activity is undertaken, discomfort is increased. Canadian Cardiovascular Society Functional Classification Ordinary physical activity does not cause angina, such as walking and climbing stairs. Angina occurs with strenuous or rapid or prolonged exertion at work or recreation. Slight limitation of ordinary activity. Walking or climbing stairs rapidly, walking uphill, walking or stair climbing after meals, in cold, in wind, or under emotional stress, or only during the few hours after awakening. Walking more than two blocks on the level and climbing more than one flight of ordinary stairs at a normal pace and in normal conditions. Marked limitation of ordinary physical activity. Walking one to two blocks on the level and climbing one flight in normal conditions and at a normal pace. Inability to carry on any physical activity without discomfort anginal syndrome may be present at rest. (Heart Failure Society of America [HFSA], 2006; Gibbons, et al., 2002; American Heart Association [AHA], 1994; Canadian Cardiovascular Society [CCS], 1976). Page 14 of 14
Corporate Medical Policy
 Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: spinal_cord_stimulation 3/1980 10/2015 10/2016 10/2015 Description of Procedure or Service Spinal cord stimulation
Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: spinal_cord_stimulation 3/1980 10/2015 10/2016 10/2015 Description of Procedure or Service Spinal cord stimulation
NEUROMODULATION THERAPY ACCESS COALITION POSITION STATEMENT ON SPINAL CORD NEUROSTIMULATION
 NEUROMODULATION THERAPY ACCESS COALITION POSITION STATEMENT ON SPINAL CORD NEUROSTIMULATION Introduction: Spinal cord stimulation (SCS) is an established treatment for chronic neuropathic pain, which can
NEUROMODULATION THERAPY ACCESS COALITION POSITION STATEMENT ON SPINAL CORD NEUROSTIMULATION Introduction: Spinal cord stimulation (SCS) is an established treatment for chronic neuropathic pain, which can
Local Coverage Determination (LCD): Spinal Cord Stimulation (Dorsal Column Stimulation) (L34705)
 Local Coverage Determination (LCD): Spinal Cord Stimulation (Dorsal Column Stimulation) (L34705) Contractor Information Contractor Name Novitas Solutions, Inc. LCD Information Document Information LCD
Local Coverage Determination (LCD): Spinal Cord Stimulation (Dorsal Column Stimulation) (L34705) Contractor Information Contractor Name Novitas Solutions, Inc. LCD Information Document Information LCD
Spinal Cord Stimulation (SCS) Therapy: Fact Sheet
 Spinal Cord Stimulation (SCS) Therapy: Fact Sheet What is SCS Therapy? Spinal cord stimulation (SCS) may be a life-changing 1 surgical option for patients to control their chronic neuropathic pain and
Spinal Cord Stimulation (SCS) Therapy: Fact Sheet What is SCS Therapy? Spinal cord stimulation (SCS) may be a life-changing 1 surgical option for patients to control their chronic neuropathic pain and
Spinal Cord Stimulation Business Case for
 Spinal Cord Stimulation Business Case for Failed Back Surgery Syndrome Understanding How Various Stakeholders Gauge Success in Treating Chronic Pain FBSS is a subset of chronic low back pain, which is
Spinal Cord Stimulation Business Case for Failed Back Surgery Syndrome Understanding How Various Stakeholders Gauge Success in Treating Chronic Pain FBSS is a subset of chronic low back pain, which is
Contractor Number 11302. Oversight Region Region IV
 Local Coverage Determination (LCD): Spinal Cord Stimulators for Chronic Pain (L32549) Contractor Information Contractor Name Palmetto GBA opens in new window Contractor Number 11302 Contractor Type MAC
Local Coverage Determination (LCD): Spinal Cord Stimulators for Chronic Pain (L32549) Contractor Information Contractor Name Palmetto GBA opens in new window Contractor Number 11302 Contractor Type MAC
Pain Management. Practical Applications in Electrotherapy
 Pain Management Practical Applications in Electrotherapy The TENS Advantage Deliver Immediate Pain Relief using a unique waveform designed to help prevent nerve accommodation. Manage Dynamic Pain by adjusting
Pain Management Practical Applications in Electrotherapy The TENS Advantage Deliver Immediate Pain Relief using a unique waveform designed to help prevent nerve accommodation. Manage Dynamic Pain by adjusting
National Coverage Determination. Vagus Nerve Stimulation (VNS)
 National Coverage Determination Vagus Nerve Stimulation (VNS) Number NEURO-004 Contractor Name Wisconsin Physicians Service Insurance Corporation AMA CPT Copyright Statement CPT codes, descriptions and
National Coverage Determination Vagus Nerve Stimulation (VNS) Number NEURO-004 Contractor Name Wisconsin Physicians Service Insurance Corporation AMA CPT Copyright Statement CPT codes, descriptions and
Neurostimulation: Orthopaedic Institute of Ohio 801 Medical Drive Lima, Ohio 45804 419-222-6622
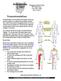 801 Medical Drive Lima, Ohio 45804 419-222-6622 Neurostimulation is the stimulation of the spinal cord by tiny electrical impulses. An implanted lead (a flexible insulated wire), which is powered by an
801 Medical Drive Lima, Ohio 45804 419-222-6622 Neurostimulation is the stimulation of the spinal cord by tiny electrical impulses. An implanted lead (a flexible insulated wire), which is powered by an
 Epimed Would Like To Congratulate The 20th Annual Budapest Conference The New Shape of Pain Relief SmalleST. ThiNNeST. CoNTouRed design. Precision Novi is the world s smallest, thinnest 16 contact primary
Epimed Would Like To Congratulate The 20th Annual Budapest Conference The New Shape of Pain Relief SmalleST. ThiNNeST. CoNTouRed design. Precision Novi is the world s smallest, thinnest 16 contact primary
Physician Coding and Payment Guide 2015
 Targeted Drug Delivery Physician Coding and Payment Guide 2015 Flowonix Medical has compiled this coding information for your convenience. This information is gathered from third party sources and is subject
Targeted Drug Delivery Physician Coding and Payment Guide 2015 Flowonix Medical has compiled this coding information for your convenience. This information is gathered from third party sources and is subject
Ambulatory Surgery Center Coding and Payment Guide 2015
 Targeted Drug Delivery Ambulatory Surgery Center Coding and Payment Guide 2015 Flowonix Medical has compiled this coding information for your convenience. This information is gathered from third party
Targeted Drug Delivery Ambulatory Surgery Center Coding and Payment Guide 2015 Flowonix Medical has compiled this coding information for your convenience. This information is gathered from third party
Spinal cord stimulation
 Spinal cord stimulation This leaflet aims to answer your questions about having spinal cord stimulation. It explains the benefits, risks and alternatives, as well as what you can expect when you come to
Spinal cord stimulation This leaflet aims to answer your questions about having spinal cord stimulation. It explains the benefits, risks and alternatives, as well as what you can expect when you come to
Intrathecal Baclofen for CNS Spasticity
 Intrathecal Baclofen for CNS Spasticity Last Review Date: November 13, 2015 Number: MG.MM.ME.31bC5 Medical Guideline Disclaimer Property of EmblemHealth. All rights reserved. The treating physician or
Intrathecal Baclofen for CNS Spasticity Last Review Date: November 13, 2015 Number: MG.MM.ME.31bC5 Medical Guideline Disclaimer Property of EmblemHealth. All rights reserved. The treating physician or
MEDICAL CONTESTED CASE HEARING NO 12090 M6-12-38043-01 DECISION AND ORDER
 MEDICAL CONTESTED CASE HEARING NO 12090 M6-12-38043-01 DECISION AND ORDER This case is decided pursuant to Chapter 410 of the Texas Workers Compensation Act and Rules of the Division of Workers Compensation
MEDICAL CONTESTED CASE HEARING NO 12090 M6-12-38043-01 DECISION AND ORDER This case is decided pursuant to Chapter 410 of the Texas Workers Compensation Act and Rules of the Division of Workers Compensation
Electroconvulsive Therapy - ECT
 Electroconvulsive Therapy - ECT Introduction Electroconvulsive therapy, or ECT, is a safe and effective treatment that may reduce symptoms related to depression or mental illness. During ECT, certain parts
Electroconvulsive Therapy - ECT Introduction Electroconvulsive therapy, or ECT, is a safe and effective treatment that may reduce symptoms related to depression or mental illness. During ECT, certain parts
Department of Neurosciences Dorsal Root Ganglion (DRG) Stimulation Information for patients
 Oxford University Hospitals NHS Trust Department of Neurosciences Dorsal Root Ganglion (DRG) Stimulation Information for patients We have recently seen you in clinic as you have had pain for a long period
Oxford University Hospitals NHS Trust Department of Neurosciences Dorsal Root Ganglion (DRG) Stimulation Information for patients We have recently seen you in clinic as you have had pain for a long period
Sample Treatment Protocol
 Sample Treatment Protocol 1 Adults with acute episode of LBP Definition: Acute episode Back pain lasting
Sample Treatment Protocol 1 Adults with acute episode of LBP Definition: Acute episode Back pain lasting
CODING SHEETS CHRONIC INTRACTABLE SPASTICITY. Effective January 1, 2009 CODMAN 3000 NEUROMODULATION AND ONCOLOGY REIMBURSEMENT HOTLINE
 CODING SHEETS CHRONIC INTRACTABLE SPASTICITY Effective January 1, 2009 CODMAN 3000 NEUROMODULATION AND ONCOLOGY REIMBURSEMENT HOTLINE Phone: 800-609-1108 Email: [email protected] Fax: 303-703-1572 CODMAN
CODING SHEETS CHRONIC INTRACTABLE SPASTICITY Effective January 1, 2009 CODMAN 3000 NEUROMODULATION AND ONCOLOGY REIMBURSEMENT HOTLINE Phone: 800-609-1108 Email: [email protected] Fax: 303-703-1572 CODMAN
Effectiveness of spinal cord stimulation for the management of neuropathic pain
 Effectiveness of spinal cord stimulation for the management of neuropathic pain August 2009 Author: Peter Larking PhD Important Note This evidence-based review summarises information on spinal cord stimulation
Effectiveness of spinal cord stimulation for the management of neuropathic pain August 2009 Author: Peter Larking PhD Important Note This evidence-based review summarises information on spinal cord stimulation
How To Treat Pain With Pain Management
 SUTTER PHYSICIANS ALLIANCE (SPA) 2800 L Street, 7 th Floor Sacramento, CA 95816 SPA PCP Treatment & Referral Guidelines Pain Management Developed June 1, 2003 Revised - (Format Revisions) November 13,
SUTTER PHYSICIANS ALLIANCE (SPA) 2800 L Street, 7 th Floor Sacramento, CA 95816 SPA PCP Treatment & Referral Guidelines Pain Management Developed June 1, 2003 Revised - (Format Revisions) November 13,
Coverage and Authorization Services is available to respond to your coding questions toll-free at 800-292-2903.
 For Urinary Control Commonly Billed Codes October 2010 Medtronic provides this information for your convenience only. It is not intended as a recommendation regarding clinical practice. It is the responsibility
For Urinary Control Commonly Billed Codes October 2010 Medtronic provides this information for your convenience only. It is not intended as a recommendation regarding clinical practice. It is the responsibility
EPIDURAL STEROID AND FACET INJECTIONS FOR SPINAL PAIN
 MEDICAL POLICY EPIDURAL STEROID AND FACET INJECTIONS FOR SPINAL PAIN Policy Number: 2015T0004W Effective Date: December 1, 2015 Table of Contents BENEFIT CONSIDERATIONS COVERAGE RATIONALE APPLICABLE CODES..
MEDICAL POLICY EPIDURAL STEROID AND FACET INJECTIONS FOR SPINAL PAIN Policy Number: 2015T0004W Effective Date: December 1, 2015 Table of Contents BENEFIT CONSIDERATIONS COVERAGE RATIONALE APPLICABLE CODES..
Aetna Nerve Conduction Study Policy
 Aetna Nerve Conduction Study Policy Policy Aetna considers nerve conduction velocity (NCV) studies medically necessary when both of the following criteria are met: 1. Member has any of the following indications:
Aetna Nerve Conduction Study Policy Policy Aetna considers nerve conduction velocity (NCV) studies medically necessary when both of the following criteria are met: 1. Member has any of the following indications:
CODING SHEETS CHRONIC INTRACTABLE PAIN MANAGEMENT. Effective January 1, 2011 CODMAN 3000 NEUROMODULATION AND ONCOLOGY REIMBURSEMENT HOTLINE
 CODING SHEETS CHRONIC INTRACTABLE PAIN MANAGEMENT Effective January 1, 2011 CODMAN 3000 NEUROMODULATION AND ONCOLOGY REIMBURSEMENT HOTLINE Phone: 800-609-1108 Email: [email protected] Fax: 303-703-1572
CODING SHEETS CHRONIC INTRACTABLE PAIN MANAGEMENT Effective January 1, 2011 CODMAN 3000 NEUROMODULATION AND ONCOLOGY REIMBURSEMENT HOTLINE Phone: 800-609-1108 Email: [email protected] Fax: 303-703-1572
EPIDURAL STEROID AND FACET INJECTIONS FOR SPINAL PAIN
 CLINICAL POLICY EPIDURAL STEROID AND FACET INJECTIONS FOR SPINAL PAIN Policy Number: PAIN 019.16 T2 Effective Date: December 1, 2015 Table of Contents CONDITIONS OF COVERAGE... BENEFIT CONSIDERATIONS..
CLINICAL POLICY EPIDURAL STEROID AND FACET INJECTIONS FOR SPINAL PAIN Policy Number: PAIN 019.16 T2 Effective Date: December 1, 2015 Table of Contents CONDITIONS OF COVERAGE... BENEFIT CONSIDERATIONS..
REHABILITATION SERVICES (OUTPATIENT)
 REHABILITATION SERVICES (OUTPATIENT) Protocol: MSC028 Effective Date: March 1, 2016 Table of Contents Page COMMERCIAL COVERAGE RATIONALE... 1 DEFINITIONS... 2 APPLICABLE CODES... 4 REFERENCES... 7 POLICY
REHABILITATION SERVICES (OUTPATIENT) Protocol: MSC028 Effective Date: March 1, 2016 Table of Contents Page COMMERCIAL COVERAGE RATIONALE... 1 DEFINITIONS... 2 APPLICABLE CODES... 4 REFERENCES... 7 POLICY
ECG may be indicated for patients with cardiovascular risk factors
 eappendix A. Summary for Preoperative ECG American College of Cardiology/ American Heart Association, 2007 A1 2002 A2 European Society of Cardiology and European Society of Anaesthesiology, 2009 A3 Improvement,
eappendix A. Summary for Preoperative ECG American College of Cardiology/ American Heart Association, 2007 A1 2002 A2 European Society of Cardiology and European Society of Anaesthesiology, 2009 A3 Improvement,
Perioperative Cardiac Evaluation
 Perioperative Cardiac Evaluation Caroline McKillop Advisor: Dr. Tam Psenka 10-3-2007 Importance of Cardiac Guidelines -Used multiple times every day -Patient Safety -Part of Surgical Care Improvement Project
Perioperative Cardiac Evaluation Caroline McKillop Advisor: Dr. Tam Psenka 10-3-2007 Importance of Cardiac Guidelines -Used multiple times every day -Patient Safety -Part of Surgical Care Improvement Project
Information for the Patient About Surgical
 Information for the Patient About Surgical Decompression and Stabilization of the Spine Aging and the Spine Daily wear and tear, along with disc degeneration due to aging and injury, are common causes
Information for the Patient About Surgical Decompression and Stabilization of the Spine Aging and the Spine Daily wear and tear, along with disc degeneration due to aging and injury, are common causes
Minimally Invasive Spine Surgery For Your Patients
 Minimally Invasive Spine Surgery For Your Patients Lukas P. Zebala, M.D. Assistant Professor Orthopaedic and Neurological Spine Surgery Department of Orthopaedic Surgery Washington University School of
Minimally Invasive Spine Surgery For Your Patients Lukas P. Zebala, M.D. Assistant Professor Orthopaedic and Neurological Spine Surgery Department of Orthopaedic Surgery Washington University School of
ELECTROMYOGRAPHY (EMG), NEEDLE, NERVE CONDUCTION STUDIES (NCS) AND QUANTITATIVE SENSORY TESTING (QST)
 AND QUANTITATIVE SENSORY TESTING (QST) Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit plan. This Medical
AND QUANTITATIVE SENSORY TESTING (QST) Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit plan. This Medical
BOTOX Injection (Onabotulinumtoxin A) for Migraine Headaches [Preauthorization Required]
![BOTOX Injection (Onabotulinumtoxin A) for Migraine Headaches [Preauthorization Required] BOTOX Injection (Onabotulinumtoxin A) for Migraine Headaches [Preauthorization Required]](/thumbs/29/13417512.jpg) BOTOX Injection (Onabotulinumtoxin A) for Migraine Headaches [Preauthorization Required] Medical Policy: MP-RX-01-11 Original Effective Date: March 24, 2011 Reviewed: Revised: This policy applies to products
BOTOX Injection (Onabotulinumtoxin A) for Migraine Headaches [Preauthorization Required] Medical Policy: MP-RX-01-11 Original Effective Date: March 24, 2011 Reviewed: Revised: This policy applies to products
Measure Title X RAY PRIOR TO MRI OR CAT SCAN IN THE EVAULATION OF LOWER BACK PAIN Disease State Back pain Indicator Classification Utilization
 Client HMSA: PQSR 2009 Measure Title X RAY PRIOR TO MRI OR CAT SCAN IN THE EVAULATION OF LOWER BACK PAIN Disease State Back pain Indicator Classification Utilization Strength of Recommendation Organizations
Client HMSA: PQSR 2009 Measure Title X RAY PRIOR TO MRI OR CAT SCAN IN THE EVAULATION OF LOWER BACK PAIN Disease State Back pain Indicator Classification Utilization Strength of Recommendation Organizations
Coding and Payment Guide for Anesthesia Services. An essential coding, billing, and reimbursement resource for anesthesiology and pain management
 Coding and Payment Guide for Anesthesia Services An essential coding, billing, and reimbursement resource for anesthesiology and pain management 2011 Contents Introduction...1 Coding Systems... 1 Claim
Coding and Payment Guide for Anesthesia Services An essential coding, billing, and reimbursement resource for anesthesiology and pain management 2011 Contents Introduction...1 Coding Systems... 1 Claim
Michigan Guidelines for the Use of Controlled Substances for the Treatment of Pain
 Michigan Guidelines for the Use of Controlled Substances for the Treatment of Pain Section I: Preamble The Michigan Boards of Medicine and Osteopathic Medicine & Surgery recognize that principles of quality
Michigan Guidelines for the Use of Controlled Substances for the Treatment of Pain Section I: Preamble The Michigan Boards of Medicine and Osteopathic Medicine & Surgery recognize that principles of quality
Transmittal 55 Date: MAY 5, 2006. SUBJECT: Changes Conforming to CR3648 for Therapy Services
 CMS Manual System Pub 100-03 Medicare National Coverage Determinations Department of Health & Human Services (DHHS) Centers for Medicare & Medicaid Services (CMS) Transmittal 55 Date: MAY 5, 2006 Change
CMS Manual System Pub 100-03 Medicare National Coverage Determinations Department of Health & Human Services (DHHS) Centers for Medicare & Medicaid Services (CMS) Transmittal 55 Date: MAY 5, 2006 Change
ICD-9-CM coding for patients with Spinal Cord Injury*
 ICD-9-CM coding for patients with Spinal Cord Injury* indicates intervening codes have been left out of this list. OTHER DISORDERS OF THE CENTRAL NERVOUS SYSTEM (340-349) 344 Other paralytic syndromes
ICD-9-CM coding for patients with Spinal Cord Injury* indicates intervening codes have been left out of this list. OTHER DISORDERS OF THE CENTRAL NERVOUS SYSTEM (340-349) 344 Other paralytic syndromes
Denver Spine Surgeons David Wong, MD, Sanjay Jatana, MD, Gary Ghiselli, MD
 Cervical and Lumbar Spine Health History Name: Today s Date: Referring Provider: How did you find us: (Please circle) Primary care physician, Google search, Facebook, Friend or Family member, Website (JatanaSpine
Cervical and Lumbar Spine Health History Name: Today s Date: Referring Provider: How did you find us: (Please circle) Primary care physician, Google search, Facebook, Friend or Family member, Website (JatanaSpine
Subject: Functional Electrical Stimulation for Spinal Cord Injury 8/27/14
 Subject: Functional Electrical Stimulation for Spinal Cord Injury Guidance Number: MCG-205 Revision Date(s): Original Effective Date: 8/27/14 SUMMARY Spinal Cord Injury (SCI): According to the international
Subject: Functional Electrical Stimulation for Spinal Cord Injury Guidance Number: MCG-205 Revision Date(s): Original Effective Date: 8/27/14 SUMMARY Spinal Cord Injury (SCI): According to the international
ESC Guidelines on the diagnosis and treatment of peripheral artery diseases Lower extremity artery disease. Erich Minar Medical University Vienna
 ESC Guidelines on the diagnosis and treatment of peripheral artery diseases Lower extremity artery disease Erich Minar Medical University Vienna for the Task Force on the Diagnosis and Treatment of Peripheral
ESC Guidelines on the diagnosis and treatment of peripheral artery diseases Lower extremity artery disease Erich Minar Medical University Vienna for the Task Force on the Diagnosis and Treatment of Peripheral
ANESTHESIA - Medicare
 ANESTHESIA - Medicare Policy Number: UM14P0008A2 Effective Date: August 19, 2014 Last Reviewed: January 1, 2016 PAYMENT POLICY HISTORY Version DATE ACTION / DESCRIPTION Version 2 January 1, 2016 Under
ANESTHESIA - Medicare Policy Number: UM14P0008A2 Effective Date: August 19, 2014 Last Reviewed: January 1, 2016 PAYMENT POLICY HISTORY Version DATE ACTION / DESCRIPTION Version 2 January 1, 2016 Under
Medical Affairs Policy
 Medical Affairs Policy Service: Sacroiliac Joint Treatments and Coccydynia Injections: Sacroiliac Joint Injections, Sacroiliac Joint Ablation, Sacroiliac Neuroablation, Sacroiliac Fusion, Lateral Branch
Medical Affairs Policy Service: Sacroiliac Joint Treatments and Coccydynia Injections: Sacroiliac Joint Injections, Sacroiliac Joint Ablation, Sacroiliac Neuroablation, Sacroiliac Fusion, Lateral Branch
MEDICAL POLICY No. 91580-R1 DRUG-ELUTING STENTS FOR ISCHEMIC HEART DISEASE
 DRUG-ELUTING STENTS FOR ISCHEMIC HEART DISEASE Effective Date: October 1, 2015 Review Dates: 10/11, 10/12, 10/13, 8/14, 8/15 Date Of Origin: October 12, 2011 Status: Current Summary of Changes Clarifications:
DRUG-ELUTING STENTS FOR ISCHEMIC HEART DISEASE Effective Date: October 1, 2015 Review Dates: 10/11, 10/12, 10/13, 8/14, 8/15 Date Of Origin: October 12, 2011 Status: Current Summary of Changes Clarifications:
Present and Potential Use of Spinal Cord Stimulation to Control Chronic Pain
 Pain Physician 2014; 17:235-246 ISSN 1533-3159 Narrative Review Present and Potential Use of Spinal Cord Stimulation to Control Chronic Pain Jason J. Song, MD, PhD, Adrian Popescu, MD, and Russell L. Bell,
Pain Physician 2014; 17:235-246 ISSN 1533-3159 Narrative Review Present and Potential Use of Spinal Cord Stimulation to Control Chronic Pain Jason J. Song, MD, PhD, Adrian Popescu, MD, and Russell L. Bell,
Corporate Medical Policy Spinal Manipulation under Anesthesia
 Corporate Medical Policy Spinal Manipulation under Anesthesia File Name: Origination: Last CAP Review: Next CAP Review: Last Review: spinal_manipulation_under_anesthesia 5/1998 1/2015 1/2016 1/2015 Description
Corporate Medical Policy Spinal Manipulation under Anesthesia File Name: Origination: Last CAP Review: Next CAP Review: Last Review: spinal_manipulation_under_anesthesia 5/1998 1/2015 1/2016 1/2015 Description
Spinal Decompression
 Spinal Decompression Spinal decompression is just one more tool we have to treat radiculopathy. With appropriate education and exercises, this modality has been proven to assist in the resolution of symptoms
Spinal Decompression Spinal decompression is just one more tool we have to treat radiculopathy. With appropriate education and exercises, this modality has been proven to assist in the resolution of symptoms
33 % of whiplash patients develop. headaches originating from the upper. cervical spine
 33 % of whiplash patients develop headaches originating from the upper cervical spine - Dr Nikolai Bogduk Spine, 1995 1 Physical Treatments for Headache: A Structured Review Headache: The Journal of Head
33 % of whiplash patients develop headaches originating from the upper cervical spine - Dr Nikolai Bogduk Spine, 1995 1 Physical Treatments for Headache: A Structured Review Headache: The Journal of Head
Billing and Coding Guidance Co-morbidities associated with morbid obesity
 Billing and Coding Guidance Co-morbidities associated with morbid obesity AMA CPT / ADA CDT / AHA NUBC Copyright Statement CPT only copyright 2002-2014 American Medical Association. All Rights Reserved.
Billing and Coding Guidance Co-morbidities associated with morbid obesity AMA CPT / ADA CDT / AHA NUBC Copyright Statement CPT only copyright 2002-2014 American Medical Association. All Rights Reserved.
Significant nerve damage is uncommonly associated with a general anaesthetic
 Risks associated with your anaesthetic Section 10: Nerve damage associated with an operation under general anaesthetic Section 10: Significant nerve damage is uncommonly associated with a general anaesthetic
Risks associated with your anaesthetic Section 10: Nerve damage associated with an operation under general anaesthetic Section 10: Significant nerve damage is uncommonly associated with a general anaesthetic
Khaled s Radiology report
 Khaled s Radiology report Patient Name: Khaled Adli Moustafa Date 06/15/2014 The patient is not present. And the following report is based upon what was in the MRI of the cervical and lumbar spine report
Khaled s Radiology report Patient Name: Khaled Adli Moustafa Date 06/15/2014 The patient is not present. And the following report is based upon what was in the MRI of the cervical and lumbar spine report
Automatic External Defibrillators
 Last Review Date: May 27, 2016 Number: MG.MM.DM.10dC2 Medical Guideline Disclaimer Property of EmblemHealth. All rights reserved. The treating physician or primary care provider must submit to EmblemHealth
Last Review Date: May 27, 2016 Number: MG.MM.DM.10dC2 Medical Guideline Disclaimer Property of EmblemHealth. All rights reserved. The treating physician or primary care provider must submit to EmblemHealth
Management of spinal cord compression
 Management of spinal cord compression (SUMMARY) Main points a) On diagnosis, all patients should receive dexamethasone 10mg IV one dose, then 4mg every 6h. then switched to oral dose and tapered as tolerated
Management of spinal cord compression (SUMMARY) Main points a) On diagnosis, all patients should receive dexamethasone 10mg IV one dose, then 4mg every 6h. then switched to oral dose and tapered as tolerated
Sacral Nerve Neuromodulation/Stimulation
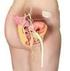 MEDICAL POLICY POLICY RELATED POLICIES POLICY GUIDELINES DESCRIPTION SCOPE BENEFIT APPLICATION RATIONALE REFERENCES CODING APPENDIX HISTORY Sacral Nerve Neuromodulation/Stimulation Number 7.01.69 Effective
MEDICAL POLICY POLICY RELATED POLICIES POLICY GUIDELINES DESCRIPTION SCOPE BENEFIT APPLICATION RATIONALE REFERENCES CODING APPENDIX HISTORY Sacral Nerve Neuromodulation/Stimulation Number 7.01.69 Effective
Marc A. Cohen, MD, FAAOS, FACS Diplomate American Board of Spinal Surgery Fellow American College of Spinal Surgery
 Marc A. Cohen, MD, FAAOS, FACS Diplomate American Board of Spinal Surgery Fellow American College of Spinal Surgery 221 Madison Ave Morristown, New Jersey 07960 (973) 538 4444 Fax (973) 538 0420 Patient
Marc A. Cohen, MD, FAAOS, FACS Diplomate American Board of Spinal Surgery Fellow American College of Spinal Surgery 221 Madison Ave Morristown, New Jersey 07960 (973) 538 4444 Fax (973) 538 0420 Patient
Measure #257 (NQF 1519): Statin Therapy at Discharge after Lower Extremity Bypass (LEB) National Quality Strategy Domain: Effective Clinical Care
 Measure #257 (NQF 1519): Statin Therapy at Discharge after Lower Extremity Bypass (LEB) National Quality Strategy Domain: Effective Clinical Care 2016 PQRS OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY
Measure #257 (NQF 1519): Statin Therapy at Discharge after Lower Extremity Bypass (LEB) National Quality Strategy Domain: Effective Clinical Care 2016 PQRS OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY
6/3/2011. High Prevalence and Incidence. Low back pain is 5 th most common reason for all physician office visits in the U.S.
 High Prevalence and Incidence Prevalence 85% of Americans will experience low back pain at some time in their life. Incidence 5% annual Timothy C. Shen, M.D. Physical Medicine and Rehabilitation Sub-specialty
High Prevalence and Incidence Prevalence 85% of Americans will experience low back pain at some time in their life. Incidence 5% annual Timothy C. Shen, M.D. Physical Medicine and Rehabilitation Sub-specialty
Michigan Board of Nursing Guidelines for the Use of Controlled Substances for the Treatment of Pain
 JENNIFER M. GRANHOLM GOVERNOR STATE OF MICHIGAN DEPARTMENT OF COMMUNITY HEALTH LANSING JANET OLSZEWSKI DIRECTOR Michigan Board of Nursing Guidelines for the Use of Controlled Substances for the Treatment
JENNIFER M. GRANHOLM GOVERNOR STATE OF MICHIGAN DEPARTMENT OF COMMUNITY HEALTH LANSING JANET OLSZEWSKI DIRECTOR Michigan Board of Nursing Guidelines for the Use of Controlled Substances for the Treatment
NON SURGICAL SPINAL DECOMPRESSION. Dr. Douglas A. VanderPloeg
 NON SURGICAL SPINAL DECOMPRESSION Dr. Douglas A. VanderPloeg CONTENTS I. Incidence of L.B.P. II. Anatomy Review III. IV. Disc Degeneration, Bulge, and Herniation Non-Surgical Spinal Decompression 1. History
NON SURGICAL SPINAL DECOMPRESSION Dr. Douglas A. VanderPloeg CONTENTS I. Incidence of L.B.P. II. Anatomy Review III. IV. Disc Degeneration, Bulge, and Herniation Non-Surgical Spinal Decompression 1. History
ICD-10 Cheat Sheet Frequently Used ICD-10 Codes for Musculoskeletal Conditions *
 ICD-10 Cheat Sheet Frequently Used ICD-10 Codes for Musculoskeletal Conditions * Finding the ICD-10 equivalent for an ICD-9 code can be a challenge. This resource of frequently used codes can help when
ICD-10 Cheat Sheet Frequently Used ICD-10 Codes for Musculoskeletal Conditions * Finding the ICD-10 equivalent for an ICD-9 code can be a challenge. This resource of frequently used codes can help when
PSYCHOLOGICAL AND NEUROPSYCHOLOGICAL TESTING
 Status Active Medical and Behavioral Health Policy Section: Behavioral Health Policy Number: X-45 Effective Date: 01/22/2014 Blue Cross and Blue Shield of Minnesota medical policies do not imply that members
Status Active Medical and Behavioral Health Policy Section: Behavioral Health Policy Number: X-45 Effective Date: 01/22/2014 Blue Cross and Blue Shield of Minnesota medical policies do not imply that members
Does the pain radiating down your legs, buttocks or lower back prevent you from walking long distances?
 Does the pain radiating down your legs, buttocks or lower back prevent you from walking long distances? Do you experience weakness, tingling, numbness, stiffness, or cramping in your legs, buttocks or
Does the pain radiating down your legs, buttocks or lower back prevent you from walking long distances? Do you experience weakness, tingling, numbness, stiffness, or cramping in your legs, buttocks or
User Manual Table of Contents. Instructions for Using the LaserTouchOne Charging the Unit... 5 Holding the Device... 5 Treatment Steps...
 User Manual User Manual Table of Contents Introduction The LaserTouchOne... 3 Device Features and Controls (Photo of device)... 4 Product Contents... 4 Laser Safety... 4 Electrical Safety... 5 Instructions
User Manual User Manual Table of Contents Introduction The LaserTouchOne... 3 Device Features and Controls (Photo of device)... 4 Product Contents... 4 Laser Safety... 4 Electrical Safety... 5 Instructions
Nursing. Management of Spinal Trauma. Content. Objectives. Objectives
 7 cervical vertebrae Content 12 thoracic vertebrae Nursing 5 sacral vertebrae Management of Spinal Trauma Kwai Fung Betty Siu Ward Manager O&T Dept TKOH Date : 22nd April 2007 5 lumbar vertebrae 4 coccygeal
7 cervical vertebrae Content 12 thoracic vertebrae Nursing 5 sacral vertebrae Management of Spinal Trauma Kwai Fung Betty Siu Ward Manager O&T Dept TKOH Date : 22nd April 2007 5 lumbar vertebrae 4 coccygeal
TABLE OF CONTENTS CPT
 TABLE OF CONTENTS CPT Coding Basics...1 National Correct Coding Initiative Edits...1 How to Use NCCI Edits...2 Evaluation and Management...3 Integumentary System... 20 Debridement... 20 Surgery/Musculoskeletal...
TABLE OF CONTENTS CPT Coding Basics...1 National Correct Coding Initiative Edits...1 How to Use NCCI Edits...2 Evaluation and Management...3 Integumentary System... 20 Debridement... 20 Surgery/Musculoskeletal...
NeuroStar TMS Therapy Patient Guide for Treating Depression
 NeuroStar TMS Therapy Patient Guide for Treating Depression This NeuroStar TMS Therapy Patient Guide for Treating Depression provides important safety and use information for you to consider about treating
NeuroStar TMS Therapy Patient Guide for Treating Depression This NeuroStar TMS Therapy Patient Guide for Treating Depression provides important safety and use information for you to consider about treating
Protocol. Cardiac Rehabilitation in the Outpatient Setting
 Protocol Cardiac Rehabilitation in the Outpatient Setting (80308) Medical Benefit Effective Date: 07/01/14 Next Review Date: 09/15 Preauthorization No Review Dates: 07/07, 07/08, 05/09, 05/10, 05/11, 05/12,
Protocol Cardiac Rehabilitation in the Outpatient Setting (80308) Medical Benefit Effective Date: 07/01/14 Next Review Date: 09/15 Preauthorization No Review Dates: 07/07, 07/08, 05/09, 05/10, 05/11, 05/12,
Guidelines for the Use of Controlled Substances in the Treatment of Pain Adopted by the New Hampshire Medical Society, July 1998
 Guidelines for the Use of Controlled Substances in the Treatment of Pain Adopted by the New Hampshire Medical Society, July 1998 Section I: Preamble The New Hampshire Medical Society believes that principles
Guidelines for the Use of Controlled Substances in the Treatment of Pain Adopted by the New Hampshire Medical Society, July 1998 Section I: Preamble The New Hampshire Medical Society believes that principles
BRYAN. Cervical Disc System. Patient Information
 BRYAN Cervical Disc System Patient Information 3 BRYAN Cervical Disc System PATIENT INFORMATION BRYAN Cervical Disc System PATIENT INFORMATION 1 BRYAN Cervical Disc System This patient information brochure
BRYAN Cervical Disc System Patient Information 3 BRYAN Cervical Disc System PATIENT INFORMATION BRYAN Cervical Disc System PATIENT INFORMATION 1 BRYAN Cervical Disc System This patient information brochure
Webinar title: Know Your Options for Treating Severe Spasticity
 Webinar title: Know Your Options for Treating Severe Spasticity Presented by: Dr. Gerald Bilsky, Physiatrist Medical Director of Outpatient Services and Associate Medical Director of Acquired Brain Injury
Webinar title: Know Your Options for Treating Severe Spasticity Presented by: Dr. Gerald Bilsky, Physiatrist Medical Director of Outpatient Services and Associate Medical Director of Acquired Brain Injury
Neck Pain Overview Causes, Diagnosis and Treatment Options
 Neck Pain Overview Causes, Diagnosis and Treatment Options Neck pain is one of the most common forms of pain for which people seek treatment. Most individuals experience neck pain at some point during
Neck Pain Overview Causes, Diagnosis and Treatment Options Neck pain is one of the most common forms of pain for which people seek treatment. Most individuals experience neck pain at some point during
MDC 1 DISEASES AND DISORDERS OF THE NERVOUS SYSTEM Implantation of chemotherapeutic agent Intracranial stents
 To assist the readers in identifying all changes that were made to the MS-DRGs as a result of comments, we developed the attached table that summaries those changes. MS-DRG Summary Table PRE-MDC Intestinal
To assist the readers in identifying all changes that were made to the MS-DRGs as a result of comments, we developed the attached table that summaries those changes. MS-DRG Summary Table PRE-MDC Intestinal
Migraine and the greater occipital nerve
 Migraine and the greater occipital nerve What is the greater occipital nerve? The nerves that travel from your spine (in the neck) to the back of the head and scalp are known as the occipital nerves. There
Migraine and the greater occipital nerve What is the greater occipital nerve? The nerves that travel from your spine (in the neck) to the back of the head and scalp are known as the occipital nerves. There
Managing Chronic Pain
 Managing Chronic Pain Chronic pain can cripple the body, mind and spirit. Feeling broken? You may benefit from Cleveland Clinic s Section of Pain Medicine, which tailors comprehensive, innovative treatment
Managing Chronic Pain Chronic pain can cripple the body, mind and spirit. Feeling broken? You may benefit from Cleveland Clinic s Section of Pain Medicine, which tailors comprehensive, innovative treatment
Issued and entered this _6th_ day of October 2010 by Ken Ross Commissioner ORDER I PROCEDURAL BACKGROUND
 STATE OF MICHIGAN DEPARTMENT OF ENERGY, LABOR & ECONOMIC GROWTH OFFICE OF FINANCIAL AND INSURANCE REGULATION Before the Commissioner of Financial and Insurance Regulation In the matter of XXXXX Petitioner
STATE OF MICHIGAN DEPARTMENT OF ENERGY, LABOR & ECONOMIC GROWTH OFFICE OF FINANCIAL AND INSURANCE REGULATION Before the Commissioner of Financial and Insurance Regulation In the matter of XXXXX Petitioner
Pennsylvania Trauma Nursing Core Curriculum. Posted to PTSF Website: 10/30/2014
 Pennsylvania Trauma Nursing Core Curriculum Posted to PTSF Website: 10/30/2014 PREFACE Care of the trauma patient has evolved since 1985, when the Pennsylvania Trauma Systems Foundation (PTSF) Board of
Pennsylvania Trauma Nursing Core Curriculum Posted to PTSF Website: 10/30/2014 PREFACE Care of the trauma patient has evolved since 1985, when the Pennsylvania Trauma Systems Foundation (PTSF) Board of
Antiplatelet and anticoagulation treatment of patients undergoing carotid and peripheral artery angioplasty
 Round Table: Antithrombotic therapy beyond ACS Antiplatelet and anticoagulation treatment of patients undergoing carotid and peripheral artery angioplasty M. Matsagkas, MD, PhD, EBSQ-Vasc Associate Professor
Round Table: Antithrombotic therapy beyond ACS Antiplatelet and anticoagulation treatment of patients undergoing carotid and peripheral artery angioplasty M. Matsagkas, MD, PhD, EBSQ-Vasc Associate Professor
Treating Bulging Discs & Sciatica. Alexander Ching, MD
 Treating Bulging Discs & Sciatica Alexander Ching, MD Disclosures Depuy Spine Teaching and courses K2 Spine Complex Spine Study Group Disclosures Take 2 I am a spine surgeon I like spine surgery I believe
Treating Bulging Discs & Sciatica Alexander Ching, MD Disclosures Depuy Spine Teaching and courses K2 Spine Complex Spine Study Group Disclosures Take 2 I am a spine surgeon I like spine surgery I believe
Medical Treatment Guidelines Washington State Department of Labor and Industries
 Complex regional pain syndrome (CRPS) Formerly known as reflex sympathetic dystrophy 1. Introduction This bulletin outlines the Department of Labor and Industries guidelines for diagnosing and treating
Complex regional pain syndrome (CRPS) Formerly known as reflex sympathetic dystrophy 1. Introduction This bulletin outlines the Department of Labor and Industries guidelines for diagnosing and treating
Corporate Medical Policy
 Corporate Medical Policy Diagnosis and Treatment of Sacroiliac Joint Pain File Name: Origination: Last CAP Review: Next CAP Review: Last Review: diagnosis_and_treatment_of_sacroiliac_joint_pain 8/2010
Corporate Medical Policy Diagnosis and Treatment of Sacroiliac Joint Pain File Name: Origination: Last CAP Review: Next CAP Review: Last Review: diagnosis_and_treatment_of_sacroiliac_joint_pain 8/2010
A list of FDA-approved testosterone products can be found by searching for testosterone at http://www.accessdata.fda.gov/scripts/cder/drugsatfda/.
 FDA Drug Safety Communication: FDA cautions about using testosterone products for low testosterone due to aging; requires labeling change to inform of possible increased risk of heart attack and stroke
FDA Drug Safety Communication: FDA cautions about using testosterone products for low testosterone due to aging; requires labeling change to inform of possible increased risk of heart attack and stroke
MEDICAL POLICY SUBJECT: CARDIAC REHABILITATION. POLICY NUMBER: 8.01.14 CATEGORY: Therapy/ Rehabilitation
 MEDICAL POLICY SUBJECT: CARDIAC REHABILITATION PAGE: 1 OF: 6 If the member's subscriber contract excludes coverage for a specific service it is not covered under that contract. In such cases, medical policy
MEDICAL POLICY SUBJECT: CARDIAC REHABILITATION PAGE: 1 OF: 6 If the member's subscriber contract excludes coverage for a specific service it is not covered under that contract. In such cases, medical policy
Nonoperative Management of Herniated Cervical Intervertebral Disc With Radiculopathy. Spine Volume 21(16) August 15, 1996, pp 1877-1883
 Nonoperative Management of Herniated Cervical Intervertebral Disc With Radiculopathy 1 Spine Volume 21(16) August 15, 1996, pp 1877-1883 Saal, Joel S. MD; Saal, Jeffrey A. MD; Yurth, Elizabeth F. MD FROM
Nonoperative Management of Herniated Cervical Intervertebral Disc With Radiculopathy 1 Spine Volume 21(16) August 15, 1996, pp 1877-1883 Saal, Joel S. MD; Saal, Jeffrey A. MD; Yurth, Elizabeth F. MD FROM
a) Nerve conduction studies (NCS) test the peripheral nervous system for:
 Health Plan Coverage Policy ARBenefits Approval: 11/02/2011 Effective Date: 01/01/2012 Revision Date: 09/18/2013 Comments: Code additions only. Title: Electrodiagnostic Testing Document: ARB0256 Public
Health Plan Coverage Policy ARBenefits Approval: 11/02/2011 Effective Date: 01/01/2012 Revision Date: 09/18/2013 Comments: Code additions only. Title: Electrodiagnostic Testing Document: ARB0256 Public
Stuart B Black MD, FAAN Chief of Neurology Co-Medical Director: Neuroscience Center Baylor University Medical Center at Dallas
 Billing and Coding in Neurology and Headache Stuart B Black MD, FAAN Chief of Neurology Co-Medical Director: Neuroscience Center Baylor University Medical Center at Dallas CPT Codes vs. ICD Codes Category
Billing and Coding in Neurology and Headache Stuart B Black MD, FAAN Chief of Neurology Co-Medical Director: Neuroscience Center Baylor University Medical Center at Dallas CPT Codes vs. ICD Codes Category
Offering Solutions for The Management of Pain
 Integrative Pain Treatment Center Integrative Pain Treatment Center Offering Solutions for The Management of Pain ADACHES BACK AND NECK PAIN FIBROMYALGIA MYOFASCIAL PAIN ARTHRITIS SPINAL STENOSIS JOINT
Integrative Pain Treatment Center Integrative Pain Treatment Center Offering Solutions for The Management of Pain ADACHES BACK AND NECK PAIN FIBROMYALGIA MYOFASCIAL PAIN ARTHRITIS SPINAL STENOSIS JOINT
LOW BACK PAIN; MECHANICAL
 1 ORTHO 16 LOW BACK PAIN; MECHANICAL Background This case definition was developed by the Armed Forces Health Surveillance Center (AFHSC) for the purpose of epidemiological surveillance of a condition
1 ORTHO 16 LOW BACK PAIN; MECHANICAL Background This case definition was developed by the Armed Forces Health Surveillance Center (AFHSC) for the purpose of epidemiological surveillance of a condition
Spine Trauma: When to Transfer. Alexander Ching, MD Director, Orthopaedic Spine Trauma OHSU
 Spine Trauma: When to Transfer Alexander Ching, MD Director, Orthopaedic Spine Trauma OHSU Disclosures Depuy Spine Consultant (teaching and courses) Department education and research funds Atlas Spine
Spine Trauma: When to Transfer Alexander Ching, MD Director, Orthopaedic Spine Trauma OHSU Disclosures Depuy Spine Consultant (teaching and courses) Department education and research funds Atlas Spine
PAIN MANAGEMENT AT UM/SYLVESTER
 PAIN MANAGEMENT AT UM/SYLVESTER W HAT IS THE PURPOSE OF THIS BROCHURE? We created this brochure for patients receiving care from the University of Miami Sylvester Comprehensive Cancer Center and their
PAIN MANAGEMENT AT UM/SYLVESTER W HAT IS THE PURPOSE OF THIS BROCHURE? We created this brochure for patients receiving care from the University of Miami Sylvester Comprehensive Cancer Center and their
Laser Treatment Policy
 Laser Treatment Policy Pursuant to federal law 21 CFR 812.2(c)7 and 812.3(b), physician(s) at this pain center may advise and use unapproved laser s on patients under one or more of the following conditions:
Laser Treatment Policy Pursuant to federal law 21 CFR 812.2(c)7 and 812.3(b), physician(s) at this pain center may advise and use unapproved laser s on patients under one or more of the following conditions:
surg urin Surgery: Urinary System 1
 Surgery: Urinary System 1 This section contains information to assist providers in billing for surgical procedures related to the urinary system. Extracorporeal Shock Wave Lithotripsy Medi-Cal covers Extracorporeal
Surgery: Urinary System 1 This section contains information to assist providers in billing for surgical procedures related to the urinary system. Extracorporeal Shock Wave Lithotripsy Medi-Cal covers Extracorporeal
Intraoperative Nerve Monitoring Coding Guide. March 1, 2010
 Intraoperative Nerve Monitoring Coding Guide March 1, 2010 Please direct any questions to: Kim Brew Manager Reimbursement and Therapy Access Medtronic ENT (904) 279-7569 Rev 9/10 KB TO OUR PARTNERS IN
Intraoperative Nerve Monitoring Coding Guide March 1, 2010 Please direct any questions to: Kim Brew Manager Reimbursement and Therapy Access Medtronic ENT (904) 279-7569 Rev 9/10 KB TO OUR PARTNERS IN
Electrodiagnostic Testing
 Electrodiagnostic Testing Electromyogram and Nerve Conduction Study North American Spine Society Public Education Series What Is Electrodiagnostic Testing? The term electrodiagnostic testing covers a
Electrodiagnostic Testing Electromyogram and Nerve Conduction Study North American Spine Society Public Education Series What Is Electrodiagnostic Testing? The term electrodiagnostic testing covers a
A Patient s Guide to Artificial Cervical Disc Replacement
 A Patient s Guide to Artificial Cervical Disc Replacement Each year, hundreds of thousands of adults are diagnosed with Cervical Disc Degeneration, an upper spine condition that can cause pain and numbness
A Patient s Guide to Artificial Cervical Disc Replacement Each year, hundreds of thousands of adults are diagnosed with Cervical Disc Degeneration, an upper spine condition that can cause pain and numbness
