Trauma/Reconstruction/Diversion. Assessing the Impact of Ureteral Stent Design on Patient Comfort
|
|
|
- Monica Brittney Shepherd
- 7 years ago
- Views:
Transcription
1 Trauma/Reconstruction/Diversion Assessing the Impact of Ureteral Stent Design on Patient Comfort James E. Lingeman,*, Glenn M. Preminger, Evan R. Goldfischer and Amy E. Krambeck (The Comfort Study Team) From the Methodist Hospital Institute for Kidney Stone Disease (JEL, AEK), Indianapolis, Indiana, Division of Urology, Duke University (GMP), Durham, North Carolina, and Hudson Valley Urology Center (ERG), Poughkeepsie, New York Purpose: We assessed the near term comfort of newly designed ureteral study stents or marketed control stents, including Polaris and Percuflex stents. Study stents had distal 6Fr pigtail ends with 3Fr or less loops. Decreased material in situ was hypothesized to enhance comfort. Usefulness of the patient self-administered Ureteral Stent Symptoms Questionnaire (Stone Management Unit, Southmead Hospital, United Kingdom) was assessed. Materials and Methods: This 4-arm multicenter study enrolled adults requiring retrograde unilateral ureteral stent placement for 4 to 28 days. Ureteral Stent Symptoms Questionnaire administration was done before placement (baseline), on day 4 after placement and on day 30 after removal. A total of 236 patients were randomized in a 1:1:1:1 ratio to the short loop tail stent (60), the long loop tail stent (59), the Percuflex Plus stent (64) and the Polaris stent (53). Results: Overall pain worsened from baseline to day 4 and improved from days 4 to 30. Mean pain medication use peaked for all stents on day 1 after placement. Common device related symptoms were mild or moderate in severity, including flank pain in 47 patients, hematuria in 39, dysuria in 34, frequent urination in 30 and urinary urgency in 27. Six patients experienced a total of 9 device related adverse events requiring hospitalization. All adverse events resolved, including most within 3 days of inpatient treatment. Conclusions: Although it was not statistically significant, patients stented with the short loop tail had lower questionnaire pain scores on day 4 after placement and lower pain medication use on day 1 after placement when pain peaked in all stent groups, suggesting that ureteral stent comfort, especially pain, may be improved by less material in situ. The Ureteral Stent Symptoms Questionnaire may be better suited for longer term comparisons in stented vs nonstented patients, rather than in this short-term ureteral stent trial. Abbreviations and Acronyms AE adverse event LLT long loop tail PC Polaris stent PPC Percuflex Plus stent SLT short loop tail USSQ Ureteric Stent Symptoms Questionnaire VAS visual analog scale Submitted for publication October 9, Study received approval from local institutional review boards. * Correspondence: Methodist Hospital Institute for Kidney Stone Disease, 1801 Senate Blvd., Suite 220, Indianapolis, Indiana (telephone: ; FAX: ; jlingeman@clarian.org). Financial interest and/or other relationship with Lumenis, Boston Scientific, Olympus, Karl Storz Medical and Cook Urological. Financial interest and/or other relationship with Boston Scientific. Key Words: ureter, stents, pain, complications, questionnaires THE precise etiology of pain during ureteral stent indwell remains elusive. Previously groups have assessed the impact of ureteral stent composition, caliber, length and configuration on patient discomfort. 1 5 With the introduction of USSQ in 2003 a standardized instrument became available for use in clinical trials to evaluate patient comfort after stent placement. 6,7 USSQ validation in the United Kingdom was done in a clinical trial setting, comparing symptoms in stented and nonstented patients who were evaluated 1 and 4 weeks after stent placement, and /09/ /0 Vol. 181, , June 2009 THE JOURNAL OF UROLOGY Printed in U.S.A. Copyright 2009 by AMERICAN UROLOGICAL ASSOCIATION DOI: /j.juro
2 2582 URETERAL STENT DESIGN AND COMFORT weeks after stent removal. This timing was consistent with standard practice in the United Kingdom. Near term (less than 1 week) comfort was not assessed in the USSQ validation study. Until recently near term comfort had not been assessed in a large clinical population involving a comparison of multiple stent types. We examined the hypothesis that substituting the distal pigtail end of a ureteral stent with an SLT or LLT design would enhance patient comfort by decreasing mass and material in situ. USSQ was used in this study and we secondarily evaluated it as a tool for assessing stent discomfort. MATERIALS AND METHODS This was a prospective, 4-arm multicenter study comparing stent related comfort in patients 18 years old or older who had undergone uncomplicated ureteroscopy requiring retrograde unilateral placement of a ureteral stent for 4 to 28 days inclusive. Study exclusion criteria were active urinary tract infection, urinary malignancy, nonstone related pain or stricture, spinal cord injury, bilateral stent placement or chronic anti-inflammatory medication. Patients provided signed informed consent before study entry at study centers and the study was approved by local institutional review boards. The study was designed to compare differences in patient ureteral stent related comfort, as assessed by the patient self-administered USSQ. USSQ is the only standardized instrument available to assess patient comfort (pain and quality of life) after ureteral stent placement. 6 8 Appropriate to anatomical differences, male and female versions of USSQ were administered. The timing of instrument administration involved certain time points, including baseline (immediately before stent placement), 4 days after stent placement and 30 5 days after stent removal. The Appendix lists USSQ components. Changes in the pain score were assessed between day 4, when peak pain was expected, and day 30, when patient comfort levels were expected to return to an almost normal level. 9 Patients were randomized in a 1:1:1:1 ratio to receive 1 of 4 stents. All patients were blinded to the stent received. The 2 study stents were the SLT and LLT stents, and the 2 control stents were the PC and PPC stents. The proximal portions of all stents were configured in identical 6Fr pigtail designs of the same basic composition but they differed in the distal third portion. Control PC and PPC were standard double-pigtail designs, while SLT and LLT study stents had substitution of the distal pigtail ends with 2, 5 cm 3Fr or less loops (SLT) or 2, 8 cm 3Fr or less loops (LLT) (see figure). The distal ends of SLT and LLT stents each had 70% less mass by volume than a standard pigtail end. The length of stent placed was determined by patient height. During the postoperative period patients had unrestricted access to pain medications such as oxycodone or hydrocodone under principal investigator guidance. At discharge home the pain management regimen included 1 Study stents were substituted for control stent distal pigtail ends with 2, 5 cm 3Fr or less loops (SLT design) or 2, 8 cm 3Fr or less loops (LLT design). or 2 tablets of 325 mg/5 mg oxycodone or 500 mg/5 mg hydrocodone every 4 to 6 hours as needed for pain. To compare patient comfort during stent placement pairwise comparisons were made among stent groups. Comparisons were performed using multiple comparison tests at an overall 5% significance level. The 2-sided F-test was used to determine significant differences in USSQ scores or pain medication use among the 4 stent groups, followed by the Tukey HSD pairwise test when F-test p Additionally, a comparison was made of the pooled pair of study stents with the pooled pair of control stents. Statistical analysis was performed using SAS software. RESULTS From April 2003 to February 2004, 236 patients were eligible for ureteral stent placement and were assigned randomly to 1 of the 4 stent groups, including 60 to the SLT group, 59 to the LLT group, 64 to the PPC group and 53 to the PC group. Since the study was stopped after interim analysis, an unequal number of patients were in each group. The 4 treatment groups were comparable demographically (table 1). A 17.7-year-old patient was enrolled in the study as an inadvertent protocol deviation. Table 2 lists surgical procedure details. Operative time and anesthesia type were not collected as part of the protocol. In 231 of 236 cases (97.9%) mean SD device indwelling time was days, including days in 64 PPC, days in 53 PC, days in 57 LLT and days in 57 SLT cases. In the remaining 5 of 236 patients (2.1%) a protocol deviation occurred due to AEs and the stents were removed before day 4, including in 1
3 URETERAL STENT DESIGN AND COMFORT 2583 Table 1. Patient characteristics in each treatment cohort PPC PC LLT Stent SLT Stent Totals No. pts Age: No. pts Mean (range) ( ) ( ) ( ) ( ) ( ) No. sex (%): M 38 (59.4) 34 (64.2) 34 (57.6) 37 (61.7) 143 (60.6) F 26 (40.6) 19 (35.8) 25 (42.4) 23 (38.3) 93 (39.4) Wt: No. pts Mean lbs (range) ( ) ( ) ( ) ( ) ( ) Ht: No. pts Mean in (range) ( ) ( ) ( ) ( ) ( ) patient each in the PC and SLT groups, and in 3 in the LLT group. Many device related AEs were expected side effects of ureteral stents. Tables 3 and 4 list procedure and device related AEs. Of device related AEs 86.5% were judged by the investigators to be mild or moderate in severity. Comparison of the overall AE rate among the 4 stent types, including 43.8% for PPC, 64.2% for PC, 52.5% for LLT and 40.0% for SLT, showed no statistical significance (p 0.051). Comparison of the device related AE rate among the 4 stent types, including 37.5% for PPC, 49.1% for PC, 37.3% for LLT and 36.7% for SLT, was also not significant (p 0.495). Six patients (2.5%), including 1, 4 and 1 with a SLT stent, LLT stent and PC, respectively, experienced a total of 9 device related AEs that led to hospitalization. These events were flank pain in 3 patients (3 events), stent migration in 1 (1 event), urinary tract infection in 2 (2 events), urosepsis and concurrent fever in 1 (2 events, respectively) and suicidal thoughts in 1 (1 event). All events resolved after hospitalization, including most within 3 days of inpatient treatment. In 10 patients, including 4, 4, 1 and 1 with LLT stent, SLT stent, PC and PPC, respectively, stents were removed due to AEs. Stents in 5 patients were removed within 4 days of placement, while in 4 the stents remained in place for 9 to 28 days. The duration of stenting in 1 patient was not recorded. AEs leading to stent removal were typically flank or suprapubic pain and urinary urgency or frequency. The patterns of change in overall USSQ comfort scores (combined body pain questions P3 to P9) were similar in all stent cohorts (table 5). Comparison of the individual USSQ components, body pain question P3 (VAS) and body pain questions P4 to P9 (kidney pain related to daily activity) again demonstrated no change from days 4 to 30 among the different stent types (p and 0.898, respectively). Overall patients experienced worsening of pain from baseline to day 4 (increasing USSQ score) and then improvement in pain from days 4 to 30 (decreasing USSQ score). Patients with an SLT stent had lower USSQ pain scores day 4 after placement. However, differences in pain scores among stent types and between stent groups (study vs control) at each time point were not statistically significant. In all stent groups more patients reported pain localized to the groin, bladder or flank areas, although no statistically significant differences were observed among the stents. Pain medication use was measured daily from baseline to day 4 (table 6). Mean use peaked in all stent groups on day 1 after placement and patients who received a SLT stent had the lowest mean use at this point. Mean pain tablet counts on days 2 and 3 were significantly higher in LLT cases than in others (Tukey HSD test p 0.05), although mean use on day 4 was clinically and statistically the same as that of the other stents. Patients in the SLT and LLT groups experienced the largest percent decrease from peak use to day 4 (53% and 57%, respectively). DISCUSSION Multiple ureteral stent shapes, sizes, compositions (durometers) and designs have been studied to decrease patient discomfort during ureteral stent placement and indwelling time. However, the precise etiology of pain during ureteral stent indwelling remains elusive. Clinical evaluation of 2 investigational stents (SLT and LLT) and currently available double pigtail PC and PPC showed similar comfort and safety profiles. The absence of statistically significant differences between USSQ pain scores in patients with different stents may have been the result of several factors related to stent design, study design and the choice of USSQ as the instrument with which to measure near term comfort.
4 2584 URETERAL STENT DESIGN AND COMFORT Table 2. Surgical procedure in each patient cohort No. PPC (%) No. PC (%) No. LLT No. SLT Total No. (%) Overall Antibiotic regimen before procedure: 120 mg Gentamicin ampicillin 20 (31.3) 17 (32.1) 20 (33.9) 19 (31.7) 76 (32.2) 120 mg Gentamicin, no ampicillin 8 (12.5) 6 (11.3) 5 (8.5) 4 (6.7) 23 (9.7) 400 mg Ciprofloxacin 3 (4.7) 2 (3.8) 5 (8.5) 3 (5.0) 13 (5.5) 1 gm Cefazolin 26 (40.6) 22 (41.5) 22 (37.3) 31 (51.7) 101 (42.8) Other 6 (9.4) 6 (11.3) 5 (8.5) 3 (5.0) 20 (8.5) Missing data 1 (1.6) 0 2 (3.4) 0 3 (1.3) Ureteroscopy purpose: Stone 60 (93.8) 49 (92.5) 58 (98.3) 55 (91.7) 222 (94.1) Diagnosis 4 (6.3) 3 (5.7) 1 (1.7) 5 (8.3) 13 (5.5) Other 0 1 (1.9) (0.4) Ureteroscopy: Flexible 24 (37.5) 23 (43.4) 25 (42.4) 28 (46.7) 100 (42.4) Rigid 27 (42.2) 21 (39.6) 27 (45.8) 26 (43.3) 101 (42.8) Flexible rigid 13 (20.3) 9 (17.0) 7 (11.9) 6 (10.0) 35 (14.8) Dilation: None 45 (70.3) 41 (77.4) 48 (81.4) 42 (70.0) 176 (74.6) Balloon 9 (14.1) 4 (7.5) 0 5 (8.3) 18 (7.6) Rigid 4 (6.3) 6 (11.3) 7 (11.9) 8 (13.3) 25 (10.6) Other 6 (9.4) 2 (3.8) 4 (6.8) 5 (8.3) 17 (7.2) Dilator size (Fr): 12 or Less 9 (14.1) 6 (11.3) 7 (11.9) 10 (16.7) 32 (13.6) Greater than 12 less than 18 4 (6.3) 3 (5.7) 4 (6.8) 7 (11.7) 18 (7.6) 18 or Greater 6 (9.4) 3 (5.7) 0 1 (1.7) 10 (4.2) Procedure sheath: Yes 32 (50.0) 23 (43.4) 24 (40.7) 25 (41.7) 104 (44.1) No 32 (50.0) 29 (54.7) 35 (59.3) 35 (58.3) 131 (55.5) Intracorporeal lithotripsy:* Not applicable 21 (32.8) 21 (39.6) 21 (35.6) 26 (43.3) 89 (37.7) Holmium laser 43 (67.2) 31 (58.5) 38 (64.4) 33 (55.0) 145 (61.4) Other 0 1 (1.9) 0 1 (1.7) 2 (0.8) Stone removal device: Not applicable 27 (42.2) 26 (49.1) 20 (33.9) 21 (35.0) 94 (39.8) Stone basket 33 (51.6) 24 (45.3) 34 (57.6) 35 (58.3) 126 (53.4) Stone cone 2 (3.1) 1 (1.9) 2 (3.4) 2 (3.3) 7 (3.0) 3-Prong graspers 1 (1.6) 2 (3.8) 2 (3.4) 0 5 (2.1) Other 1 (1.6) 0 1 (1.7) 2 (3.3) 4 (1.7) Residual fragments: None visible 34 (53.1) 28 (52.8) 34 (57.6) 33 (55.0) 129 (54.7) 2 mm or Less 30 (46.9) 23 (43.4) 24 (40.7) 25 (41.7) 102 (43.2) Greater than 2 mm 0 2 (3.8) 1 (1.7) 0 3 (1.3) Foley catheter: Yes 11 (17.2) 6 (11.3) 3 (5.1) 3 (5.0) 23 (9.7) No 53 (82.8) 47 (88.7) 56 (94.9) 57 (95.0) 213 (90.3) Stent side: Lt 20 (31.3) 19 (35.8) 21 (35.6) 18 (30.0) 78 (33.1) Rt 21 (32.8) 13 (24.5) 13 (22.0) 19 (31.7) 66 (28.0) * Electrohydraulic, pneumatic and pulsed dye laser lithotripsy was not used. Alligator graspers were not used. The ureteral stent designs may have been too similar to generate statistically significant differences in USSQ pain scores and, thus, each stent was equally comfortable. However, other data potentially support the hypothesis that less material in the bladder was associated with enhanced comfort. Patients who received an SLT stent had lower USSQ pain scores on day 4 after placement and lower pain medication use on day 1 after placement, when pain peaked in all stent groups. Compared to all other stent recipients, patients who received an LLT stent appeared to experience more pain, in that mean pain medication use was higher on days 1 and 2, and 3 (each p 0.05). Furthermore, a greater proportion of patients who received an LLT stent reported flank pain and suprapubic pain compared to the other patients. Finally, all AEs of flank pain that led to hospitalization were experienced by patients who received a LLT stent. These observations seem contradic-
5 URETERAL STENT DESIGN AND COMFORT 2585 Table 3. Breakdown of 103 procedure related AEs Condition No. PPC (%) No. PC (%) No. LLT No. SLT Overall Abdominal pain 1 (4.2) 0 1 (3.7) 4 (17.3) Nausea 2 (8.3) 4 (13.8) 0 1 (4.3) Vomiting 0 2 (6.9) 0 1 (4.3) Rigors 0 1 (3.4) 0 0 Fever 0 2 (6.9) 0 1 (4.3) Catheter migration (4.3) Fungal infection (3.7) 0 Urosepsis 0 1 (3.4) 0 0 Urinary tract 3 (12.5) 0 2 (7.4) 1 (4.3) infection Bladder/suprapubic 6 (25.0) 6 (20.6) 4 (14.8) 5 (21.7) pain/spasm Back pain 0 1 (3.4) 0 1 (4.3) Flank pain/colic 16 (66.7) 15 (51.7) 20 (74.1) 10 (43.5) Groin/testicular 0 2 (6.9) 0 1 (4.3) pain Headache/dizziness 0 2 (6.9) 1 (3.7) 0 Suicidal ideation (3.7) 0 Dysuria 11 (45.8) 10 (34.5) 7 (25.9) 7 (30.4) Incontinence not 3 (12.5) otherwise specified Urgency 7 (29.2) 2 (6.9) 6 (22.2) 8 (34.8) Frequency 9 (37.5) 3 (10.3) 6 (22.2) 5 (21.7) Urethral pain (8.7) Urinary retention 1 (4.2) 0 1 (3.7) 1 (4.3) Vesicoureteral (3.7) 0 reflux Acute renal failure (3.7) 0 Ureteral (3.7) 0 perforation Hematuria 15 (62.5) 13 (44.8) 12 (44.4) 12 (52.2) Nocturia 1 (4.2) 0 1 (3.7) 0 Respiratory problem (4.3) tory to the study central hypothesis of less stent material in the bladder or intramural ureter resulting in less patient discomfort, especially since the SLT and LLT stents maintain an identical mass in the bladder. However, the additional loop length of the LLT stent resides in the ureter. Therefore, the etiology of pain caused by loop tailed ureteral stents may involve a combination of the stent distal end profile, length, mass and movement within the bladder or ureter, or at the ureteral orifice. The study design validating the USSQ was based on a comparison of USSQ scores in stented and nonstented cases. 6 In contrast to this all or none study design, in the current study we assessed patient comfort among 4 stents with only subtle design differences. Therefore, USSQ by design may not have been sufficiently sensitive to detect differences in comfort among patients receiving similarly designed stents. Furthermore, post hoc statistical modeling in the current investigation indicated that a study population of approximately 1,000 patients was needed to show statistical separation among the 4 study and control ureteral stents. Therefore, only a quarter of the necessary number of patients were analyzed and no statistically or clinically significant differences in patient comfort could have been observed among the stents at any time point. Recently Lee et al reported no statistically significant differences in USSQ pain scores in 44 patients who completed the questionnaire 1, 3 and 5 days after ureteral stent placement. 10 USSQ is a lengthy and complicated instrument. Patient psychological and physical status in the days immediately following ureteral stent placement can create a motivational challenge for completing USSQ. Some groups who have studied comfort have acknowledged the issue of patient discomfort in regard to USSQ complexity and they have used ad hoc questionnaires, while others have adapted existing instruments validated for other symptoms, eg following prostate biopsy, 11,12 although the USSQ was available. Modifying USSQ by abridging it or validating its modules for discrete or improvisational use may enhance its usefulness in more diverse clinical study settings. Table 4. Breakdown of 94 device related AEs Condition No. PPC (%) No. PC (%) No. LLT No. SLT Overall Abdominal pain 1 (4.2) 1 (3.8) 1 (4.5) 4 (18.1) Nausea 2 (8.3) 3 (11.5) 0 1 (4.5) Vomiting (4.5) Rigors 0 1 (3.8) 0 0 Fever 0 2 (7.7) 0 1 (4.5) Catheter migration (4.5) 1 (4.5) Urosepsis 0 1 (3.8) 0 0 Urinary tract 3 (12.5) 0 1 (4.5) 1 (4.5) infection Bladder/suprapubic 6 (25.0) 5 (19.2) 6 (27.2) 5 (22.7) pain/spasm Back pain 0 1 (3.8) 0 1 (4.5) Flank pain/colic 15 (62.5) 14 (53.8) 18 (81.8) 9 (40.9) Groin/testicular 0 1 (3.8) 0 1 (4.5) pain Headache/dizziness 0 2 (6.9) 0 0 Suicidal ideation (4.5) 0 Dysuria 11 (45.8) 10 (38.5) 7 (25.9) 7 (31.8) Incontinence not 4 (16.7) 1 (3.8) 2 (9.1) 0 otherwise specified Urgency 10 (41.7) 3 (11.5) 9 (40.9) 8 (36.4) Frequency 12 (50.0) 4 (15.4) 9 (40.9) 6 (27.3) Urethral pain 0 1 (3.8) 0 2 (9.1) Urinary retention (4.5) Vesicoureteral 0 1 (3.8) 1 (4.5) 0 reflux Hematuria 14 (58.3) 10 (38.5) 10 (45.5) 10 (45.5) Nocturia 1 (4.2) 1 (3.8) 2 (9.1) 0
6 2586 URETERAL STENT DESIGN AND COMFORT Table 5. USSQ P3 to P9 combined body pain scores for each stent from days 4 to 30 Mean Score (range) PPC PC LLT Stent SLT Stent Totals p Value No. pts Day ( ) ( ) ( ) ( ) ( ) Day ( ) 7.75 ( ) 7.74 ( ) 6.81 ( ) 6.98 ( ) Day 4 30 change ( ) ( ) ( ) ( ) ( ) The timing of USSQ administration may have a role in obtaining robust data. In its validation study USSQ administration time points reflected management regimens in the United Kingdom, 6 where ureteral stents are typically placed for an extended duration between physician visits. The current study was performed primarily in the United States, where stents are typically placed for a shorter duration. In the current study pain was assessed by USSQ on baseline, on day 4 after placement and on day 30 after removal. However, data on pain medication use indicated that patients experienced peak pain 1 day after stent placement. Therefore, USSQ was administered when patient pain was subsiding. Had maximum potential pain scores been obtained, differences among the stents on day 30 may have shown greater separation. In a study comparing comfort among multiple similarly designed stents the optimal timing for USSQ administration appears to be 1 to 2 days after the stent is placed. Certain study limitations must be recognized. Each study arm included an unequal number of patients because the study was terminated early after interim statistical analysis demonstrated no significant differences among the study arms. Stenting duration was not fixed among the study arms to allow variations in clinical practices and treatment scenarios. Finally, 2 control stents were used, although PC and PPC have similar designs and materials. Using 2 different control groups we could expand the study by further comparing different standard double pigtail stents. Despite these limitations to our knowledge the current study is the first to compare the near term comfort of indwelling ureteral stents using USSQ in a large clinical study population involving a comparison of multiple stent types. In future investigations comparing stent comfort study design considerations must include appropriate statistical powering and clinical assessments. The required population size will create challenges from the perspectives of multiple investigational centers, patient enrollment, the effect on study duration and total cost. Survey instruments designed to measure patient subjective assessments of comfort and pain must be appropriately timed and administered to coincide with expected maximum and minimum periods of comfort. Objective study assessments should be measured, such as events of early stent removal, new or increased pain medication use or pill counts, or the need for unscheduled physician Table 6. Analgesia with acetaminophen plus 325 mg/5 mg oxycodone or 500 mg/5 mg hydrocodone PPC PC LLT Stent SLT Stent Totals p Value No. pts Day 0: No. pts Mean No. tablets (range) 2.07 ( ) 1.78 ( ) 2.61 ( ) 1.98 ( ) 2.11 ( ) Day 1: No. pts Mean No. tablets (range) 3.87 ( ) 4.00 ( ) 5.05 ( ) 3.60 ( ) 4.12 ( ) Day 2: No. pts Mean No. tablets (range) 3.70 ( ) 3.40 ( ) 4.93 ( ) 3.15 ( ) 3.79 ( ) Day 3: No. pts Mean No. tablets (range) 3.17 ( ) 2.56 ( ) 4.44 ( ) 2.83 ( ) 3.25 ( ) Day 4: No. pts Mean No. tablets (range) 2.28 ( ) 2.06 ( ) 2.18 ( ) 1.68 ( ) 2.05 ( ) Overall: No. pts Mean No. tablets (range) ( ) ( ) ( ) ( ) ( ) 0.035
7 URETERAL STENT DESIGN AND COMFORT 2587 visits. Clearly there are many considerations for future clinical investigations of patient comfort after ureteral stent placement. APPENDIX USSQ Components USSQ Component Parameters CONCLUSIONS We examined the hypothesis that substituting the distal pigtail end of a ureteral stent with an SLT or LLT design would enhance patient comfort in regard to less mass and material in situ. Patients who received an SLT stent had lower USSQ pain scores on day 4 after placement and lower pain medication use on day 1 after placement, when pain peaked in all stent groups. The absence of statistically significant differences among USSQ pain scores in patients with the different stents suggests that USSQ may be better suited for longer term comparisons among stented and nonstented cases, rather than in this short-term study design. Future studies of ureteral stent comfort will require balancing subjective and objective end points. Opportunities exist for further studies and survey instrument development. Extent of body pain 7 Questions: P3, a VAS (0 no pain/discomfort to 10 worst possible pain)* P4 to P9, multiple choice questions Location of body pain VAS rating (P3) at 4 locations (females) and 5 locations (males): Kidney front/side area Groin area Bladder area Kidney back (flank) area Penis (males only) Urinary tract 11 Multiple choice questions (U1 to U11) symptoms Other USSQ information General health, work performance, sexual matters and additional problems * The VAS was read by 2 readers. When the 2 readings were not within 0.5 points, the readings were adjudicated by a third reader. The pictorial question P3 (extent of body pain) asked the patient to rate pain on the VAS at 4 locations (females) and 5 locations (males). Therefore, the maximum score in females was 40 and in males it was 50. The scale in women was normalized to 50 to have the same scale in all patients, including the 2 sexes. REFERENCES 1. Pollard SG and Macfarlane R: Symptoms arising from Double-J ureteral stents. J Urol 1988; 139: Pryor JL, Langley MJ and Jenkins AD: Comparison of symptom characteristics of indwelling ureteral catheters. J Urol 1991; 145: Riehle RA: Selective use of ureteral stents before extracorporeal shock-wave lithotripsy. Urol Clin North Am 1988; 15: Saltzman B: Ureteral stents. Indications, variations, and complications. Urol Clin North Am 1988; 15: Lennon GM, Thornhill JA, Sweeny PA, Grainger R, McDermott TE and Butler MR: Firm versus soft double pigtail ureteric stents: a randomized blind comparative trial. Eur Urol 1995; 28: Joshi HB, Newns N, Stainthorpe A, MacDonagh RP, Keeley FX Jr and Timoney AG: Ureteral stent symptom questionnaire: development and validation of a multidimensional quality of life measure. J Urol 2003; 169: Joshi HB, Stainthorpe A, MacDonagh RP, Keeley FX Jr, Timoney AG and Barry MJ: Indwelling ureteral stents: evaluation of symptoms, quality of life and utility. J Urol 2003; 169: Joshi HB, Stainthorpe A, Keeley FX, MacDonagh R and Timoney AG: Indwelling ureteral stents: evaluation of quality of life to aid outcome analysis. J Endourol 2001; 15: Damiano R, Autorino R, De Sio M, Cantiello F, Quarto G, Perdona S et al: Does the size of ureteral stent impact urinary symptoms and quality of life? A prospective randomized study. Eur Urol 2005; 48: Lee C, Kuskowski M, Premoli J, Skemp N and Monga M: Randomized evaluation of ureteral stents using validated symptom questionnaire. J Endourol 2005; 19: Leibovici D, Cooper A, Lindner A, Ostrowsky R, Kleinmann J and Velikanov S et al: Ureteral stents: morbidity and impact on quality of life. Isr Med Assoc J 2005; 7: El-Nahas AR, El-Assmy AM, Shoma AM, Eraky I, El-Kenawy MR and El-Kappany HA: Self-retaining ureteral stents: analysis of factors responsible for patients discomfort. J Endourol 2006; 20: 33.
Urolithiasis/Endourology
 Urolithiasis/Endourology Use of Ureteral Stent in Extracorporeal Shock Wave Lithotripsy for Upper Urinary Calculi: A Systematic Review and Meta-Analysis Shen Pengfei, Jiang Min, Yang Jie, Li Xiong, Li
Urolithiasis/Endourology Use of Ureteral Stent in Extracorporeal Shock Wave Lithotripsy for Upper Urinary Calculi: A Systematic Review and Meta-Analysis Shen Pengfei, Jiang Min, Yang Jie, Li Xiong, Li
Open the Flood Gates Urinary Obstruction and Kidney Stones. Dr. Jeffrey Rosenberg Dr. Emilio Lastarria Dr. Richard Kasulke
 Open the Flood Gates Urinary Obstruction and Kidney Stones Dr. Jeffrey Rosenberg Dr. Emilio Lastarria Dr. Richard Kasulke Nephrology vs. Urology Nephrologist a physician who has been trained in the diagnosis
Open the Flood Gates Urinary Obstruction and Kidney Stones Dr. Jeffrey Rosenberg Dr. Emilio Lastarria Dr. Richard Kasulke Nephrology vs. Urology Nephrologist a physician who has been trained in the diagnosis
URETEROSCOPY (AND TREATMENT OF KIDNEY STONES)
 URETEROSCOPY (AND TREATMENT OF KIDNEY STONES) AN INFORMATION LEAFLET Written by: Department of Urology May 2011 Stockport: 0161 419 5698 Website: w w w. s t o c k p o r t. n h s. u k Tameside: 0161 922
URETEROSCOPY (AND TREATMENT OF KIDNEY STONES) AN INFORMATION LEAFLET Written by: Department of Urology May 2011 Stockport: 0161 419 5698 Website: w w w. s t o c k p o r t. n h s. u k Tameside: 0161 922
surg urin Surgery: Urinary System 1
 Surgery: Urinary System 1 This section contains information to assist providers in billing for surgical procedures related to the urinary system. Extracorporeal Shock Wave Lithotripsy Medi-Cal covers Extracorporeal
Surgery: Urinary System 1 This section contains information to assist providers in billing for surgical procedures related to the urinary system. Extracorporeal Shock Wave Lithotripsy Medi-Cal covers Extracorporeal
2.0 Synopsis. Vicodin CR (ABT-712) M05-765 Clinical Study Report R&D/07/095. (For National Authority Use Only) to Part of Dossier: Volume:
 2.0 Synopsis Abbott Laboratories Name of Study Drug: Vicodin CR Name of Active Ingredient: Hydrocodone/Acetaminophen Extended Release (ABT-712) Individual Study Table Referring to Part of Dossier: Volume:
2.0 Synopsis Abbott Laboratories Name of Study Drug: Vicodin CR Name of Active Ingredient: Hydrocodone/Acetaminophen Extended Release (ABT-712) Individual Study Table Referring to Part of Dossier: Volume:
A PRINTED copy of this guideline may not be the most recent version. The OFFICIAL version is located on IHNET at the Policies & Procedures Home Page
 A PRINTED copy of this guideline may not be the most recent version. The OFFICIAL version is located on IHNET at the Policies & Procedures Home Page IX0200: Prevention & Control of Catheter Associated
A PRINTED copy of this guideline may not be the most recent version. The OFFICIAL version is located on IHNET at the Policies & Procedures Home Page IX0200: Prevention & Control of Catheter Associated
Prevention and Recognition of Obstetric Fistula Training Package. Module 8: Pre-repair Care and Referral for Women with Obstetric Fistula
 Prevention and Recognition of Obstetric Fistula Training Package Module 8: Pre-repair Care and Referral for Women with Obstetric Fistula Early detection and treatment If a woman has recently survived a
Prevention and Recognition of Obstetric Fistula Training Package Module 8: Pre-repair Care and Referral for Women with Obstetric Fistula Early detection and treatment If a woman has recently survived a
POAC CLINICAL GUIDELINE
 POAC CLINICAL GUIDELINE Acute Pylonephritis DIAGNOSIS COMPLICATED PYELONEPHRITIS EXCLUSION CRITERIA: Male Known or suspected renal impairment (egfr < 60) Abnormality of renal tract Known or suspected renal
POAC CLINICAL GUIDELINE Acute Pylonephritis DIAGNOSIS COMPLICATED PYELONEPHRITIS EXCLUSION CRITERIA: Male Known or suspected renal impairment (egfr < 60) Abnormality of renal tract Known or suspected renal
Antibiotic Prophylaxis for Short-term Catheter Bladder Drainage in adults. A Systematic Review (Cochrane database August 2013)
 Antibiotic Prophylaxis for Short-term Catheter Bladder Drainage in adults A Systematic Review (Cochrane database August 2013) Gail Lusardi, Senior Lecturer Dr Allyson Lipp, Principal Lecturer, Dr Chris
Antibiotic Prophylaxis for Short-term Catheter Bladder Drainage in adults A Systematic Review (Cochrane database August 2013) Gail Lusardi, Senior Lecturer Dr Allyson Lipp, Principal Lecturer, Dr Chris
Medical Malpractice in Endourology: Analysis of Closed Cases From the State of New York
 Medical Malpractice in Endourology: Analysis of Closed Cases From the State of New York Brian Duty,* Zhamshid Okhunov, Zeph Okeke and Arthur Smith From the Department of Urology, North Shore-Long Island
Medical Malpractice in Endourology: Analysis of Closed Cases From the State of New York Brian Duty,* Zhamshid Okhunov, Zeph Okeke and Arthur Smith From the Department of Urology, North Shore-Long Island
Ureteroscopy with Laser Lithotripsy
 Ureteroscopy with Laser Lithotripsy Introduction Kidney stones are fairly common. Although kidney stones can be very painful, they are treatable, and in many cases preventable. Your doctor may recommend
Ureteroscopy with Laser Lithotripsy Introduction Kidney stones are fairly common. Although kidney stones can be very painful, they are treatable, and in many cases preventable. Your doctor may recommend
Urinary Tract Infections
 Urinary Tract Infections Overview A urine culture must ALWAYS be interpreted in the context of the urinalysis and patient symptoms. If a patient has no signs of infection on urinalysis, no symptoms of
Urinary Tract Infections Overview A urine culture must ALWAYS be interpreted in the context of the urinalysis and patient symptoms. If a patient has no signs of infection on urinalysis, no symptoms of
To decrease and/or prevent the incidence of catheter associated infections and other complications associated with IUC.
 Patient Care Manual Standardized Procedure Number: PC-SP.115 Latest Revision Date: 01/27/2015 Effective Date: 10/10/2011 Standardized Procedure: Urethral Catheter (IUC), Adult, Discontinuance of FUNCTION
Patient Care Manual Standardized Procedure Number: PC-SP.115 Latest Revision Date: 01/27/2015 Effective Date: 10/10/2011 Standardized Procedure: Urethral Catheter (IUC), Adult, Discontinuance of FUNCTION
MANAGEMENT OF PROSTATE ENLARGEMENT/BPH
 MANAGEMENT OF PROSTATE ENLARGEMENT/BPH J E S S I C A E. P A O N E S S A, M. D. A S S I S T A N T P R O F E S S O R D E P A R T M E N T O F U R O L O G Y S U N Y U P S T A T E M E D I C A L U N I V E R
MANAGEMENT OF PROSTATE ENLARGEMENT/BPH J E S S I C A E. P A O N E S S A, M. D. A S S I S T A N T P R O F E S S O R D E P A R T M E N T O F U R O L O G Y S U N Y U P S T A T E M E D I C A L U N I V E R
Effectiveness of Day-case Surgery in Urology: Single Surgeon Experience
 Bahrain Medical Bulletin 29, No. 3, September 2007 Effectiveness of Day-case Surgery in Urology: Single Surgeon Experience Mohamed H. Durazi, FRCS ED, FRCSI* Reem Al-Bareeq, MRCSI, CAB(Urol)** Mohamed
Bahrain Medical Bulletin 29, No. 3, September 2007 Effectiveness of Day-case Surgery in Urology: Single Surgeon Experience Mohamed H. Durazi, FRCS ED, FRCSI* Reem Al-Bareeq, MRCSI, CAB(Urol)** Mohamed
Prevention of catheter associated urinary tract infections
 Prevention of catheter associated urinary tract infections Dr. Suzan Sanavi, Nephrologist, M.D University of Social Welfare and Rehabilitation Akhavan Physical Spine Center INTRODUCTION Urinary bladder
Prevention of catheter associated urinary tract infections Dr. Suzan Sanavi, Nephrologist, M.D University of Social Welfare and Rehabilitation Akhavan Physical Spine Center INTRODUCTION Urinary bladder
150640_Brochure_B 4/12/07 2:58 PM Page 2. Patient Information. Freedom From an Enlarged Prostate
 150640_Brochure_B 4/12/07 2:58 PM Page 2 Patient Information Freedom From an Enlarged Prostate 150640_Brochure_B 4/12/07 2:58 PM Page 3 GreenLight Laser Therapy 1 150640_Brochure_B 4/12/07 2:58 PM Page
150640_Brochure_B 4/12/07 2:58 PM Page 2 Patient Information Freedom From an Enlarged Prostate 150640_Brochure_B 4/12/07 2:58 PM Page 3 GreenLight Laser Therapy 1 150640_Brochure_B 4/12/07 2:58 PM Page
A Summary of the Guideline for the Diagnosis and Management of. Urinary Tract Infections in Long Term Care
 A Summary of the Guideline for the Diagnosis and Management of Urinary Tract Infections in Long Term Care Exclusions Community acquired UTIs UTIs in acute care Prevention Limit use of catheters Ensure
A Summary of the Guideline for the Diagnosis and Management of Urinary Tract Infections in Long Term Care Exclusions Community acquired UTIs UTIs in acute care Prevention Limit use of catheters Ensure
Prostate Cancer Patients Report on Benefits of Proton Therapy
 Prostate Cancer Patients Report on Benefits of Proton Therapy Dobson DaVanzo & Associates, LLC Vienna, VA 703.260.1760 www.dobsondavanzo.com Prostate Cancer Patients Report on Benefits of Proton Therapy
Prostate Cancer Patients Report on Benefits of Proton Therapy Dobson DaVanzo & Associates, LLC Vienna, VA 703.260.1760 www.dobsondavanzo.com Prostate Cancer Patients Report on Benefits of Proton Therapy
PREVENTION OF CATHETER ASSOCIATED URINARY TRACT INFECTIONS. (CAUTIs)
 PREVENTION OF CATHETER ASSOCIATED URINARY TRACT INFECTIONS (CAUTIs) CAUTIs A UTI where an indwelling urinary catheter was in place for >2 calendar days on the date of event. OR If an indwelling urinary
PREVENTION OF CATHETER ASSOCIATED URINARY TRACT INFECTIONS (CAUTIs) CAUTIs A UTI where an indwelling urinary catheter was in place for >2 calendar days on the date of event. OR If an indwelling urinary
Urinary Tract Infections
 1 Infections in the urinary tract are relatively common. These infections are often referred to as bladder infections. They are also known as UTI s or urinary tract infections. When an infection is confined
1 Infections in the urinary tract are relatively common. These infections are often referred to as bladder infections. They are also known as UTI s or urinary tract infections. When an infection is confined
Refer to Coaptite Injectable Implant Instructions for Use provided with product for complete instructions for use.
 Questions for my Doctor Refer to Coaptite Injectable Implant Instructions for Use provided with product for complete instructions for use. INDICATIONS: Coaptite Injectable Implant is indicated for soft
Questions for my Doctor Refer to Coaptite Injectable Implant Instructions for Use provided with product for complete instructions for use. INDICATIONS: Coaptite Injectable Implant is indicated for soft
Long-term result of Memokath urethral sphincter stent in. spinal cord injury patients
 1 Long-term result of Memokath urethral sphincter stent in spinal cord injury patients Subramanian Vaidyanathan 1 E-mail: vaidyanathansiu@hotmail.com Bakul M Soni 1 E-mail: Bakul.Soni@mail.soh-tr.nwest.nhs.uk
1 Long-term result of Memokath urethral sphincter stent in spinal cord injury patients Subramanian Vaidyanathan 1 E-mail: vaidyanathansiu@hotmail.com Bakul M Soni 1 E-mail: Bakul.Soni@mail.soh-tr.nwest.nhs.uk
HELPFUL STEP-BY-STEP ILLUSTRATIONS ARE LOCATED IN THIS BOOKLET
 HELPFUL STEP-BY-STEP ILLUSTRATIONS ARE LOCATED IN THIS BOOKLET DESCRIPTION The Vet Sent Ureter comes as a sterile stent system comprised of two components: 1) the radioopaque, double pigtail multi-fenestrated
HELPFUL STEP-BY-STEP ILLUSTRATIONS ARE LOCATED IN THIS BOOKLET DESCRIPTION The Vet Sent Ureter comes as a sterile stent system comprised of two components: 1) the radioopaque, double pigtail multi-fenestrated
Riesa Gusewelle, MNSc, RN, APRN, GNP-BC. OBJECTIVES Identify early warning signs of urinary tract
 Detection, Early Management & Prevention, of Urinary Tract Infections in Older Adults Riesa Gusewelle, MNSc, RN, APRN, GNP-BC OBJECTIVES Identify early warning signs of urinary tract infections (UTIs)
Detection, Early Management & Prevention, of Urinary Tract Infections in Older Adults Riesa Gusewelle, MNSc, RN, APRN, GNP-BC OBJECTIVES Identify early warning signs of urinary tract infections (UTIs)
NICE Pathways bring together all NICE guidance, quality standards and other NICE information on a specific topic.
 Initial assessment and investigation of urinary incontinence bring together all NICE guidance, quality standards and other NICE information on a specific topic. are interactive and designed to be used
Initial assessment and investigation of urinary incontinence bring together all NICE guidance, quality standards and other NICE information on a specific topic. are interactive and designed to be used
VITAMIN C AND INFECTIOUS DISEASE: A REVIEW OF THE LITERATURE AND THE RESULTS OF A RANDOMIZED, DOUBLE-BLIND, PROSPECTIVE STUDY OVER 8 YEARS
 39 Chapter 3 VITAMIN C AND INFECTIOUS DISEASE: A REVIEW OF THE LITERATURE AND THE RESULTS OF A RANDOMIZED, DOUBLE-BLIND, PROSPECTIVE STUDY OVER 8 YEARS Maxine Briggs TABLE OF CONTENTS I. Review of the
39 Chapter 3 VITAMIN C AND INFECTIOUS DISEASE: A REVIEW OF THE LITERATURE AND THE RESULTS OF A RANDOMIZED, DOUBLE-BLIND, PROSPECTIVE STUDY OVER 8 YEARS Maxine Briggs TABLE OF CONTENTS I. Review of the
CARE PROCESS STEP EXPECTATIONS RATIONALE
 URINARY INCONTINENCE CARE PROCESS STEP EXPECTATIONS RATIONALE ASSESSMENT/PROBLEM RECOGNITION 1. Did the staff and physician seek and document risk factors for urinary incontinence and any history of urinary
URINARY INCONTINENCE CARE PROCESS STEP EXPECTATIONS RATIONALE ASSESSMENT/PROBLEM RECOGNITION 1. Did the staff and physician seek and document risk factors for urinary incontinence and any history of urinary
Urinary tract and perineum
 9 Urinary tract and perineum Key Points 9.1 9.1 THE URINARY BLADDER URINARY RETENTION Acute retention of urine is an indication for emergency drainage of the bladder The common causes of acute retention
9 Urinary tract and perineum Key Points 9.1 9.1 THE URINARY BLADDER URINARY RETENTION Acute retention of urine is an indication for emergency drainage of the bladder The common causes of acute retention
Postoperative. Voiding Dysfunction
 Postoperative Voiding Trial Voiding Dysfunction Stephanie Pickett, MD Fellow Female Pelvic Medicine and Reconstructive Surgery Objectives Define postoperative voiding dysfunction Describe how to evaluate
Postoperative Voiding Trial Voiding Dysfunction Stephanie Pickett, MD Fellow Female Pelvic Medicine and Reconstructive Surgery Objectives Define postoperative voiding dysfunction Describe how to evaluate
Etiology and treatment of chronic bacterial prostatitis the Croatian experience
 Etiology and treatment of chronic bacterial prostatitis the Croatian experience Višnja Škerk University Hospital for Infectious Diseases "Dr. Fran Mihaljevic" Zagreb Croatia Milano, Malpensa, 14 Nov 2008
Etiology and treatment of chronic bacterial prostatitis the Croatian experience Višnja Škerk University Hospital for Infectious Diseases "Dr. Fran Mihaljevic" Zagreb Croatia Milano, Malpensa, 14 Nov 2008
PROTOCOL SYNOPSIS Evaluation of long-term opioid efficacy for chronic pain
 P a g e 1 PROTOCOL SYNOPSIS Evaluation of long-term opioid efficacy for chronic pain Clinical Phase 4 Study Centers Study Period 25 U.S. sites identified and reviewed by the Steering Committee and Contract
P a g e 1 PROTOCOL SYNOPSIS Evaluation of long-term opioid efficacy for chronic pain Clinical Phase 4 Study Centers Study Period 25 U.S. sites identified and reviewed by the Steering Committee and Contract
MINIMALLY INVASIVE MANAGEMENT OF FELINE LOWER URINARY TRACT DISEASE:
 MINIMALLY INVASIVE MANAGEMENT OF FELINE LOWER URINARY TRACT DISEASE: LASERS, STENTS AND BALLOONS Dr. Allyson Berent, DVM, DACVIM The Animal Medical Center, New York, NY Feline lower urinary tract disease
MINIMALLY INVASIVE MANAGEMENT OF FELINE LOWER URINARY TRACT DISEASE: LASERS, STENTS AND BALLOONS Dr. Allyson Berent, DVM, DACVIM The Animal Medical Center, New York, NY Feline lower urinary tract disease
INTERDISCIPLINARY CLINICAL MANUAL Practice Guideline
 INTERDISCIPLINARY CLINICAL MANUAL Practice Guideline TITLE: Insertion of a curved tip Urinary Catheter (Coude/Tiemann) NUMBER: CC 50-013 Effective Date: August 2015 Page 1 of 5 Applies To: All Preamble:
INTERDISCIPLINARY CLINICAL MANUAL Practice Guideline TITLE: Insertion of a curved tip Urinary Catheter (Coude/Tiemann) NUMBER: CC 50-013 Effective Date: August 2015 Page 1 of 5 Applies To: All Preamble:
Adult Urodynamics: American Urological Association (AUA)/Society of Urodynamics, Female Pelvic Medicine & Urogenital Reconstruction (SUFU) Guideline
 Adult Urodynamics: American Urological Association (AUA)/Society of Urodynamics, Female Pelvic Medicine & Urogenital Reconstruction (SUFU) Guideline RECOMMENDATIONS 1 Conditional: Clinicians who are making
Adult Urodynamics: American Urological Association (AUA)/Society of Urodynamics, Female Pelvic Medicine & Urogenital Reconstruction (SUFU) Guideline RECOMMENDATIONS 1 Conditional: Clinicians who are making
Psoriasis, Incidence, Quality of Life, Psoriatic Arthritis, Prevalence
 1.0 Abstract Title Prevalence and Incidence of Articular Symptoms and Signs Related to Psoriatic Arthritis in Patients with Psoriasis Severe or Moderate with Adalimumab Treatment (TOGETHER). Keywords Psoriasis,
1.0 Abstract Title Prevalence and Incidence of Articular Symptoms and Signs Related to Psoriatic Arthritis in Patients with Psoriasis Severe or Moderate with Adalimumab Treatment (TOGETHER). Keywords Psoriasis,
LOSS OF BLADDER CONTROL IS TREATABLE TAKE CONTROL AND RESTORE YOUR LIFESTYLE
 LOSS OF BLADDER CONTROL IS TREATABLE TAKE CONTROL AND RESTORE YOUR LIFESTYLE TALKING ABOUT STRESS INCONTINENCE (SUI) Millions of women suffer from stress incontinence (SUI). This condition results in accidental
LOSS OF BLADDER CONTROL IS TREATABLE TAKE CONTROL AND RESTORE YOUR LIFESTYLE TALKING ABOUT STRESS INCONTINENCE (SUI) Millions of women suffer from stress incontinence (SUI). This condition results in accidental
By James D. Gould, MD FACS
 By James D. Gould, MD FACS BACKGROUND Balloon devices enlarge narrowed sinus ostia and outflow tracts by remodeling the surrounding bone and paranasal sinus structures. Multiple studies have demonstrated
By James D. Gould, MD FACS BACKGROUND Balloon devices enlarge narrowed sinus ostia and outflow tracts by remodeling the surrounding bone and paranasal sinus structures. Multiple studies have demonstrated
Bard: Intermittent Catheters. A guide to. Bard: Pelvic Organ Prolapse. An REIMBURSEMENT. overview of OF INTERMITTENT. Prolapse CATHETERS
 Bard: Intermittent Catheters A guide to Bard: Pelvic Organ Prolapse An REIMBURSEMENT overview of Pelvic OF INTERMITTENT Organ Prolapse CATHETERS 1 Intermittent catheterization is a covered Medicare benefit
Bard: Intermittent Catheters A guide to Bard: Pelvic Organ Prolapse An REIMBURSEMENT overview of Pelvic OF INTERMITTENT Organ Prolapse CATHETERS 1 Intermittent catheterization is a covered Medicare benefit
Ceftriaxone Therapy Vs. Ciprofloxacin In Treatment Of Typhoid Fever In Adult Patients.
 Dec QMJ VOL.5 No.8 Ceftriaxone Therapy Vs. Ciprofloxacin In Treatment Of Typhoid Fever In Adult Patients. Radhi F.Alshaibani* الخلاصھ لا زالت حمى التایفوید سببا مھما من اسباب الامراض وفقدان ساعات العمل
Dec QMJ VOL.5 No.8 Ceftriaxone Therapy Vs. Ciprofloxacin In Treatment Of Typhoid Fever In Adult Patients. Radhi F.Alshaibani* الخلاصھ لا زالت حمى التایفوید سببا مھما من اسباب الامراض وفقدان ساعات العمل
FAQ About Prostate Cancer Treatment and SpaceOAR System
 FAQ About Prostate Cancer Treatment and SpaceOAR System P. 4 Prostate Cancer Background SpaceOAR Frequently Asked Questions (FAQ) 1. What is prostate cancer? The vast majority of prostate cancers develop
FAQ About Prostate Cancer Treatment and SpaceOAR System P. 4 Prostate Cancer Background SpaceOAR Frequently Asked Questions (FAQ) 1. What is prostate cancer? The vast majority of prostate cancers develop
THE KIDNEY. Bulb of penis Abdominal aorta Scrotum Adrenal gland Inferior vena cava Urethra Corona glandis. Kidney. Glans penis Testicular vein
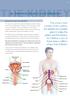 29 THE KIDNEY 9. Recurrent urinary tract infections Recurrent urinary tract infections The urinary tract consists of the urethra, the bladder, the ureters, the kidneys and in men the prostate gland. An
29 THE KIDNEY 9. Recurrent urinary tract infections Recurrent urinary tract infections The urinary tract consists of the urethra, the bladder, the ureters, the kidneys and in men the prostate gland. An
Allium Ureteral Stent (URS)
 Allium Ureteral Stent (URS) Instructions For Use Manufactured by Allium Ltd. DEVICE NAME: ALLIUM Ureteral Stent (URS) is intended to be inserted into the lower ureter to allow free flow of urine from the
Allium Ureteral Stent (URS) Instructions For Use Manufactured by Allium Ltd. DEVICE NAME: ALLIUM Ureteral Stent (URS) is intended to be inserted into the lower ureter to allow free flow of urine from the
CHAPTER 13 Pediatric Urologic Diseases
 CHAPTER 13 Pediatric Urologic Diseases Urinary Incontinence Hypospadias Ureterocele Ureteropelvic Junction Obstruction Posterior Urethral Valves Vesicoureteral Reflux Disease Upper Urinary Tract Stones
CHAPTER 13 Pediatric Urologic Diseases Urinary Incontinence Hypospadias Ureterocele Ureteropelvic Junction Obstruction Posterior Urethral Valves Vesicoureteral Reflux Disease Upper Urinary Tract Stones
Patient Information:
 Patient Information: Care of an indwelling Urinary Catheter What is a Urinary Catheter? A catheter is a hollow, flexible tube designed to drain urine from the bladder. Following insertion into the bladder,
Patient Information: Care of an indwelling Urinary Catheter What is a Urinary Catheter? A catheter is a hollow, flexible tube designed to drain urine from the bladder. Following insertion into the bladder,
URINARY CATHETER CARE
 URINARY CATHETER CARE INTRODUCTION Urinary catheter care is a very important skill, and it is a skill that many certified nursing assistants (CNAs) must know. Competence at providing urinary catheter care
URINARY CATHETER CARE INTRODUCTION Urinary catheter care is a very important skill, and it is a skill that many certified nursing assistants (CNAs) must know. Competence at providing urinary catheter care
PROCEDURE- SPECIFIC INFORMATION FOR PATIENTS
 The British Association of Urological Surgeons 35-43 Lincoln s Inn Fields London WC2A 3PE Phone: Fax: Website: E- mail: +44 (0)20 7869 6950 +44 (0)20 7404 5048 www.baus.org.uk admin@baus.org.uk PROCEDURE-
The British Association of Urological Surgeons 35-43 Lincoln s Inn Fields London WC2A 3PE Phone: Fax: Website: E- mail: +44 (0)20 7869 6950 +44 (0)20 7404 5048 www.baus.org.uk admin@baus.org.uk PROCEDURE-
1.0 Abstract. Title: Real Life Evaluation of Rheumatoid Arthritis in Canadians taking HUMIRA. Keywords. Rationale and Background:
 1.0 Abstract Title: Real Life Evaluation of Rheumatoid Arthritis in Canadians taking HUMIRA Keywords Rationale and Background: This abbreviated clinical study report is based on a clinical surveillance
1.0 Abstract Title: Real Life Evaluation of Rheumatoid Arthritis in Canadians taking HUMIRA Keywords Rationale and Background: This abbreviated clinical study report is based on a clinical surveillance
What s new? INVESTIGATIVE PROTOCOL FOR URINARY INCONTINENCE & CATHETERS. The Revised Guidance Includes: Interpretive Guidelines
 INVESTIGATIVE PROTOCOL FOR URINARY INCONTINENCE & CATHETERS The Revised Guidance Includes: Interpretive Guidelines Investigative Protocols Compliance & Severity Guidance What s new? The new guidance for
INVESTIGATIVE PROTOCOL FOR URINARY INCONTINENCE & CATHETERS The Revised Guidance Includes: Interpretive Guidelines Investigative Protocols Compliance & Severity Guidance What s new? The new guidance for
CAUTI-The Challenge Continues IHA-Coalition for Care April 23, 2014 Presented by Linda Doerflein, BS, RN, CPHRM Director of Quality/Risk HealthSouth
 CAUTI-The Challenge Continues IHA-Coalition for Care April 23, 2014 Presented by Linda Doerflein, BS, RN, CPHRM Director of Quality/Risk HealthSouth Deaconess Rehabilitation Hospital Did You Know?? The
CAUTI-The Challenge Continues IHA-Coalition for Care April 23, 2014 Presented by Linda Doerflein, BS, RN, CPHRM Director of Quality/Risk HealthSouth Deaconess Rehabilitation Hospital Did You Know?? The
Urinary Diversion: Ileovesicostomy/Ileal Loop/Colon Loop
 Urinary Diversion: Ileovesicostomy/Ileal Loop/Colon Loop Why do I need this surgery? A urinary diversion is a surgical procedure that is performed to allow urine to safely pass from the kidneys into a
Urinary Diversion: Ileovesicostomy/Ileal Loop/Colon Loop Why do I need this surgery? A urinary diversion is a surgical procedure that is performed to allow urine to safely pass from the kidneys into a
Radiation Therapy for Prostate Cancer: Treatment options and future directions
 Radiation Therapy for Prostate Cancer: Treatment options and future directions David Weksberg, M.D., Ph.D. PinnacleHealth Cancer Institute September 12, 2015 Radiation Therapy for Prostate Cancer: Treatment
Radiation Therapy for Prostate Cancer: Treatment options and future directions David Weksberg, M.D., Ph.D. PinnacleHealth Cancer Institute September 12, 2015 Radiation Therapy for Prostate Cancer: Treatment
Oral Zinc Supplementation as an Adjunct Therapy in the Management of Hepatic Encephalopathy: A Randomized Controlled Trial
 Oral Zinc Supplementation as an Adjunct Therapy in the Management of Hepatic Encephalopathy: A Randomized Controlled Trial Marcus R. Pereira A. Study Purpose Hepatic encephalopathy is a common complication
Oral Zinc Supplementation as an Adjunct Therapy in the Management of Hepatic Encephalopathy: A Randomized Controlled Trial Marcus R. Pereira A. Study Purpose Hepatic encephalopathy is a common complication
Received June 18, 2007; Revised August 30, 2007; Accepted August 30, 2007; Published September 17, 2007
 Case Study TheScientificWorldJOURNAL (2007) 7, 1575 1578 TSW Urology ISSN 1537-744X; DOI 10.1100/tsw.2007.253 Unusual Complication of Suprapubic Cystostomy in a Male Patient with Tetraplegia: Traction
Case Study TheScientificWorldJOURNAL (2007) 7, 1575 1578 TSW Urology ISSN 1537-744X; DOI 10.1100/tsw.2007.253 Unusual Complication of Suprapubic Cystostomy in a Male Patient with Tetraplegia: Traction
1 in 3 women experience Stress Urinary Incontinence.
 A PATIENT S GUIDE 1 in 3 women experience Stress Urinary Incontinence. It s time to talk about SUI Get the facts. This Patient s Guide is intended as a public resource on the issue of Stress Urinary Incontinence
A PATIENT S GUIDE 1 in 3 women experience Stress Urinary Incontinence. It s time to talk about SUI Get the facts. This Patient s Guide is intended as a public resource on the issue of Stress Urinary Incontinence
510(k) SUMMARY. OPTION-vyM Urinary Catheter
 510(k) SUMMARY OPTICON MEDICAL, INC. OPTION-vyM Urinary Catheter Submitter s Name and Contact Information Opticon Medical, Inc. 7001 Post Road, Suite 100 Dublin, OH 430 16 Primary Contact: Glenn D. Brunner,
510(k) SUMMARY OPTICON MEDICAL, INC. OPTION-vyM Urinary Catheter Submitter s Name and Contact Information Opticon Medical, Inc. 7001 Post Road, Suite 100 Dublin, OH 430 16 Primary Contact: Glenn D. Brunner,
Improving access and reducing costs of care for overactive bladder through a multidisciplinary delivery model
 Improving access and reducing costs of care for overactive bladder through a multidisciplinary delivery model Trafford Crump, Ph.D. Department of Surgery, University of Calgary Presentation to: Canadian
Improving access and reducing costs of care for overactive bladder through a multidisciplinary delivery model Trafford Crump, Ph.D. Department of Surgery, University of Calgary Presentation to: Canadian
Study Design Of Medical Research
 Study Design Of Medical Research By Ahmed A.Shokeir, MD,PHD, FEBU Prof. Urology, Urology & Nephrology Center, Mansoura, Egypt Study Designs In Medical Research Topics Classification Case series studies
Study Design Of Medical Research By Ahmed A.Shokeir, MD,PHD, FEBU Prof. Urology, Urology & Nephrology Center, Mansoura, Egypt Study Designs In Medical Research Topics Classification Case series studies
Are Urinary Catheters necessary during Endovascular Procedures? A prospective randomized pilot study. Medical Student Research Project.
 Are Urinary Catheters necessary during Endovascular Procedures? A prospective randomized pilot study Medical Student Research Project Jordan Knepper Faculty advisor: Mark Langsfeld, MD Introduction Background
Are Urinary Catheters necessary during Endovascular Procedures? A prospective randomized pilot study Medical Student Research Project Jordan Knepper Faculty advisor: Mark Langsfeld, MD Introduction Background
International Journal of Case Reports in Medicine
 International Journal of Case Reports in Medicine Vol. 2013 (2013), Article ID 340925, 18 minipages. DOI:10.5171/2013.340925 www.ibimapublishing.com Copyright 2013. Massinde Anthony. Distributed under
International Journal of Case Reports in Medicine Vol. 2013 (2013), Article ID 340925, 18 minipages. DOI:10.5171/2013.340925 www.ibimapublishing.com Copyright 2013. Massinde Anthony. Distributed under
Systolic Blood Pressure Intervention Trial (SPRINT) Principal Results
 Systolic Blood Pressure Intervention Trial (SPRINT) Principal Results Paul K. Whelton, MB, MD, MSc Chair, SPRINT Steering Committee Tulane University School of Public Health and Tropical Medicine, and
Systolic Blood Pressure Intervention Trial (SPRINT) Principal Results Paul K. Whelton, MB, MD, MSc Chair, SPRINT Steering Committee Tulane University School of Public Health and Tropical Medicine, and
Bladder Health Promotion
 Bladder Health Promotion Community Awareness Presentation Content contributions provided by the Society of Urologic Nurses (SUNA) National Association for Continence (NAFC) Simon Foundation for Continence
Bladder Health Promotion Community Awareness Presentation Content contributions provided by the Society of Urologic Nurses (SUNA) National Association for Continence (NAFC) Simon Foundation for Continence
X-Plain Kidney Stones Reference Summary
 X-Plain Kidney Stones Reference Summary Introduction Kidney stones are fairly common. Although they can be very painful, they are treatable, and in many cases preventable. This reference summary will help
X-Plain Kidney Stones Reference Summary Introduction Kidney stones are fairly common. Although they can be very painful, they are treatable, and in many cases preventable. This reference summary will help
Series 1 Case Studies Adverse Events that Represent Unanticipated Problems: Reporting Required
 Welcome! This document contains three (3) series of Case Study examples that will demonstrate all four OHSU reporting categories (#1 4) as well as examples of events that are considered not reportable.
Welcome! This document contains three (3) series of Case Study examples that will demonstrate all four OHSU reporting categories (#1 4) as well as examples of events that are considered not reportable.
A Phase 2 Study of Interferon Beta-1a (Avonex ) in Ulcerative Colitis
 A Phase 2 Study of (Avonex ) in Ulcerative Colitis - Study Results - ClinicalTrials.gov A Phase 2 Study of (Avonex ) in Ulcerative Colitis This study has been completed. Sponsor: Biogen Idec Information
A Phase 2 Study of (Avonex ) in Ulcerative Colitis - Study Results - ClinicalTrials.gov A Phase 2 Study of (Avonex ) in Ulcerative Colitis This study has been completed. Sponsor: Biogen Idec Information
Introduction to Statistics Used in Nursing Research
 Introduction to Statistics Used in Nursing Research Laura P. Kimble, PhD, RN, FNP-C, FAAN Professor and Piedmont Healthcare Endowed Chair in Nursing Georgia Baptist College of Nursing Of Mercer University
Introduction to Statistics Used in Nursing Research Laura P. Kimble, PhD, RN, FNP-C, FAAN Professor and Piedmont Healthcare Endowed Chair in Nursing Georgia Baptist College of Nursing Of Mercer University
SECTION H: BLADDER AND BOWEL. H0100: Appliances. Item Rationale Health-related Quality of Life. Planning for Care
 SECTION H: BLADDER AND BOWEL Intent: The intent of the items in this section is to gather information on the use of bowel and bladder appliances, the use of and response to urinary toileting programs,
SECTION H: BLADDER AND BOWEL Intent: The intent of the items in this section is to gather information on the use of bowel and bladder appliances, the use of and response to urinary toileting programs,
KELLI DEWITT WHITEHEAD, RN, MS, ARNP
 CIRRICULUM VITAE KELLI DEWITT WHITEHEAD, RN, MS, ARNP Associated Urologist, PA 341 Wheatfield, Suite 180 Sunnyvale, TX 75182 972-270-8859 EDUCATION Master of Science, Family Nurse Practitioner University
CIRRICULUM VITAE KELLI DEWITT WHITEHEAD, RN, MS, ARNP Associated Urologist, PA 341 Wheatfield, Suite 180 Sunnyvale, TX 75182 972-270-8859 EDUCATION Master of Science, Family Nurse Practitioner University
Oncology Nursing Society Annual Progress Report: 2008 Formula Grant
 Oncology Nursing Society Annual Progress Report: 2008 Formula Grant Reporting Period July 1, 2011 June 30, 2012 Formula Grant Overview The Oncology Nursing Society received $12,473 in formula funds for
Oncology Nursing Society Annual Progress Report: 2008 Formula Grant Reporting Period July 1, 2011 June 30, 2012 Formula Grant Overview The Oncology Nursing Society received $12,473 in formula funds for
NURSE DRIVEN FOLEY CATHETER PROTOCOL
 NURSE DRIVEN FOLEY CATHETER PROTOCOL BACKGROUND There are over 1.7 million hospital-acquired infections in US hospitals annually, 40% of which are urinary tract infections. 80% of hospital-acquired UTIs
NURSE DRIVEN FOLEY CATHETER PROTOCOL BACKGROUND There are over 1.7 million hospital-acquired infections in US hospitals annually, 40% of which are urinary tract infections. 80% of hospital-acquired UTIs
Kidney Stones. This reference summary will help you understand kidney stones and how to treat and prevent them. Kidney
 Introduction A kidney stone is a solid piece of material that forms in the kidney from substances in the urine. Kidney stones are fairly common. Although kidney stones can be painful, they are treatable.
Introduction A kidney stone is a solid piece of material that forms in the kidney from substances in the urine. Kidney stones are fairly common. Although kidney stones can be painful, they are treatable.
Endoscopic therapy for obesity and complications of bariatric surgery
 Endoscopic therapy for obesity and complications of bariatric surgery Jacques Devière, MD, PhD Erasme University Hospital Brussels Belgium jacques.deviere@erasme.ulb.ac.be Obesity Affects 300 millions
Endoscopic therapy for obesity and complications of bariatric surgery Jacques Devière, MD, PhD Erasme University Hospital Brussels Belgium jacques.deviere@erasme.ulb.ac.be Obesity Affects 300 millions
Pyelonephritis: Kidney Infection
 Pyelonephritis: Kidney Infection National Kidney and Urologic Diseases Information Clearinghouse U.S. Department of Health and Human Services NATIONAL INSTITUTES OF HEALTH What is pyelonephritis? Pyelonephritis
Pyelonephritis: Kidney Infection National Kidney and Urologic Diseases Information Clearinghouse U.S. Department of Health and Human Services NATIONAL INSTITUTES OF HEALTH What is pyelonephritis? Pyelonephritis
Tara Stevermuer (MAppStat), Centre for Health Service Development, University of Wollongong.
 ENAR - PAIN RELIEF THAT S FAST AND LASTS Tara Stevermuer (MAppStat), Centre for Health Service Development, University of Wollongong. Introducing ENAR The ENAR (Electro-Neuro-Adaptive-Regulator) is a Russian
ENAR - PAIN RELIEF THAT S FAST AND LASTS Tara Stevermuer (MAppStat), Centre for Health Service Development, University of Wollongong. Introducing ENAR The ENAR (Electro-Neuro-Adaptive-Regulator) is a Russian
Measure #257 (NQF 1519): Statin Therapy at Discharge after Lower Extremity Bypass (LEB) National Quality Strategy Domain: Effective Clinical Care
 Measure #257 (NQF 1519): Statin Therapy at Discharge after Lower Extremity Bypass (LEB) National Quality Strategy Domain: Effective Clinical Care 2016 PQRS OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY
Measure #257 (NQF 1519): Statin Therapy at Discharge after Lower Extremity Bypass (LEB) National Quality Strategy Domain: Effective Clinical Care 2016 PQRS OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY
PATIENT CARE MANUAL PROCEDURE
 PATIENT CARE MANUAL PROCEDURE NUMBER VII-E-5 PAGE 1 OF 7 APPROVED BY: CATEGORY: Tri-site Nursing Policy and Procedures Review Committee Body Systems; Genitourinary 1.0 GOALS To influence patient care providers
PATIENT CARE MANUAL PROCEDURE NUMBER VII-E-5 PAGE 1 OF 7 APPROVED BY: CATEGORY: Tri-site Nursing Policy and Procedures Review Committee Body Systems; Genitourinary 1.0 GOALS To influence patient care providers
Thomas A. Kollmorgen, M.D. Oregon Urology Institute
 Thomas A. Kollmorgen, M.D. Oregon Urology Institute None 240,000 new diagnosis per year, and an estimated 28,100 deaths (2012) 2 nd leading cause of death from cancer in U.S.A. Approximately 1 in 6 men
Thomas A. Kollmorgen, M.D. Oregon Urology Institute None 240,000 new diagnosis per year, and an estimated 28,100 deaths (2012) 2 nd leading cause of death from cancer in U.S.A. Approximately 1 in 6 men
Palm Beach Obstetrics & Gynecology, PA
 Palm Beach Obstetrics & Gynecology, PA 4671 S. Congress Avenue, Lake Worth, FL 33461 561.434.0111 4631 N. Congress Avenue, Suite 102, West Palm Beach, FL 33407 Urinary Tract Infection About one of every
Palm Beach Obstetrics & Gynecology, PA 4671 S. Congress Avenue, Lake Worth, FL 33461 561.434.0111 4631 N. Congress Avenue, Suite 102, West Palm Beach, FL 33407 Urinary Tract Infection About one of every
190.12 - Urine Culture, Bacterial Summary Information Highlighted items indicate most frequently used codes.
 Medicare National Coverage Determination (NCD) (Jan 2011) Source: CMS: Centers for Medicare & Medicaid Services To view full document: www.cms.gov/coveragegeninfo. Select Lab NCDs, then select NCD Coding
Medicare National Coverage Determination (NCD) (Jan 2011) Source: CMS: Centers for Medicare & Medicaid Services To view full document: www.cms.gov/coveragegeninfo. Select Lab NCDs, then select NCD Coding
GENERAL INFORMATION. Adverse Event (AE) Definition (ICH GUIDELINES E6 FOR GCP 1.2):
 Make copies of the blank SAE report form as needed. Retain originals with confirmation of all information faxed to DMID Pharmacovigilance Group Clinical Research Operations and Management Support (CROMS
Make copies of the blank SAE report form as needed. Retain originals with confirmation of all information faxed to DMID Pharmacovigilance Group Clinical Research Operations and Management Support (CROMS
Sponsor. Novartis Generic Drug Name. Vildagliptin. Therapeutic Area of Trial. Type 2 diabetes. Approved Indication. Investigational.
 Clinical Trial Results Database Page 1 Sponsor Novartis Generic Drug Name Vildagliptin Therapeutic Area of Trial Type 2 diabetes Approved Indication Investigational Study Number CLAF237A2386 Title A single-center,
Clinical Trial Results Database Page 1 Sponsor Novartis Generic Drug Name Vildagliptin Therapeutic Area of Trial Type 2 diabetes Approved Indication Investigational Study Number CLAF237A2386 Title A single-center,
Primary Care Management of Male Lower Urinary Tract Symptoms. Matthew B.K. Shaw Consultant Urological Surgeon
 Primary Care Management of Male Lower Urinary Tract Symptoms Matthew B.K. Shaw Consultant Urological Surgeon NICE LUTS Guidelines Lower Urinary Tract Symptoms (LUTS) in men. NICE Clinical Guideline 97
Primary Care Management of Male Lower Urinary Tract Symptoms Matthew B.K. Shaw Consultant Urological Surgeon NICE LUTS Guidelines Lower Urinary Tract Symptoms (LUTS) in men. NICE Clinical Guideline 97
Spotlight on Success: Implementing Nurse-Driven Protocols to Reduce CAUTIs
 Spotlight on Success: Implementing Nurse-Driven Protocols to Reduce CAUTIs Many hospitals today are increasingly allowing nurses to make decisions regarding patients care, treatment, and services without
Spotlight on Success: Implementing Nurse-Driven Protocols to Reduce CAUTIs Many hospitals today are increasingly allowing nurses to make decisions regarding patients care, treatment, and services without
Childhood Urinary Tract Infections
 Childhood Urinary Tract Infections What is a UTI? Urinary tract infection (UTI) is one of the most common infections in childhood. It can cause distress to the child, concerns to the parents, and may
Childhood Urinary Tract Infections What is a UTI? Urinary tract infection (UTI) is one of the most common infections in childhood. It can cause distress to the child, concerns to the parents, and may
10. Treatment of peritoneal dialysis associated fungal peritonitis
 10. Treatment of peritoneal dialysis associated fungal peritonitis Date written: February 2003 Final submission: July 2004 Guidelines (Include recommendations based on level I or II evidence) The use of
10. Treatment of peritoneal dialysis associated fungal peritonitis Date written: February 2003 Final submission: July 2004 Guidelines (Include recommendations based on level I or II evidence) The use of
Bladder and Bowel Control
 Bladder and Bowel Control Dr Sue Woodward Lecturer, Florence Nightingale School of Nursing and Midwifery 2 Why do we need to understand anatomy? Normal physiology Normal adult bladder capacity = 450-500mls
Bladder and Bowel Control Dr Sue Woodward Lecturer, Florence Nightingale School of Nursing and Midwifery 2 Why do we need to understand anatomy? Normal physiology Normal adult bladder capacity = 450-500mls
Case Based Urology Learning Program
 Case Based Urology Learning Program Resident s Corner: UROLOGY Case Number 21 CBULP 2011 068 Case Based Urology Learning Program Editor: Associate Editors: Manager: Case Contributors: Steven C. Campbell,
Case Based Urology Learning Program Resident s Corner: UROLOGY Case Number 21 CBULP 2011 068 Case Based Urology Learning Program Editor: Associate Editors: Manager: Case Contributors: Steven C. Campbell,
Omega-3 fatty acids improve the diagnosis-related clinical outcome. Critical Care Medicine April 2006;34(4):972-9
 Omega-3 fatty acids improve the diagnosis-related clinical outcome 1 Critical Care Medicine April 2006;34(4):972-9 Volume 34(4), April 2006, pp 972-979 Heller, Axel R. MD, PhD; Rössler, Susann; Litz, Rainer
Omega-3 fatty acids improve the diagnosis-related clinical outcome 1 Critical Care Medicine April 2006;34(4):972-9 Volume 34(4), April 2006, pp 972-979 Heller, Axel R. MD, PhD; Rössler, Susann; Litz, Rainer
Blood in the urine (hematuria)
 Blood in the urine (hematuria) Hematuria refers to the presence of blood in the urine. It is important to investigate the cause of hematuria because rarely, it is caused by a serious condition, such as
Blood in the urine (hematuria) Hematuria refers to the presence of blood in the urine. It is important to investigate the cause of hematuria because rarely, it is caused by a serious condition, such as
GU Assessment, Diagnosis and Case Studies. Jami Windhorn, RN BSN SOAR Session #7
 GU Assessment, Diagnosis and Case Studies Jami Windhorn, RN BSN SOAR Session #7 GU System Kidneys: Ultrafiltration of the blood, maintain water and electrolyte homeostasis Pelvicalyceal systems: urine
GU Assessment, Diagnosis and Case Studies Jami Windhorn, RN BSN SOAR Session #7 GU System Kidneys: Ultrafiltration of the blood, maintain water and electrolyte homeostasis Pelvicalyceal systems: urine
Scientific Method. 2. Design Study. 1. Ask Question. Questionnaire. Descriptive Research Study. 6: Share Findings. 1: Ask Question.
 Descriptive Research Study Investigation of Positive and Negative Affect of UniJos PhD Students toward their PhD Research Project : Ask Question : Design Study Scientific Method 6: Share Findings. Reach
Descriptive Research Study Investigation of Positive and Negative Affect of UniJos PhD Students toward their PhD Research Project : Ask Question : Design Study Scientific Method 6: Share Findings. Reach
PATIENT URINARY CATHETER PASSPORT
 n PATIENT URINARY CATHETER PASSPORT A guide on how to look after your Catheter NHS Hertfordshire Patient Experience Team Charter House Parkway Welwyn Garden City Hertfordshire AL8 6JL Telephone: 01707
n PATIENT URINARY CATHETER PASSPORT A guide on how to look after your Catheter NHS Hertfordshire Patient Experience Team Charter House Parkway Welwyn Garden City Hertfordshire AL8 6JL Telephone: 01707
6/3/2011. High Prevalence and Incidence. Low back pain is 5 th most common reason for all physician office visits in the U.S.
 High Prevalence and Incidence Prevalence 85% of Americans will experience low back pain at some time in their life. Incidence 5% annual Timothy C. Shen, M.D. Physical Medicine and Rehabilitation Sub-specialty
High Prevalence and Incidence Prevalence 85% of Americans will experience low back pain at some time in their life. Incidence 5% annual Timothy C. Shen, M.D. Physical Medicine and Rehabilitation Sub-specialty
Treatment of Opioid Dependence: A Randomized Controlled Trial. Karen L. Sees, DO, Kevin L. Delucchi, PhD, Carmen Masson, PhD, Amy
 Category: Heroin Title: Methadone Maintenance vs 180-Day psychosocially Enriched Detoxification for Treatment of Opioid Dependence: A Randomized Controlled Trial Authors: Karen L. Sees, DO, Kevin L. Delucchi,
Category: Heroin Title: Methadone Maintenance vs 180-Day psychosocially Enriched Detoxification for Treatment of Opioid Dependence: A Randomized Controlled Trial Authors: Karen L. Sees, DO, Kevin L. Delucchi,
PROCEEDINGS INCIDENCE AND PREVALENCE OF STRESS URINARY INCONTINENCE * Ananias C. Diokno, MD ABSTRACT
 INCIDENCE AND PREVALENCE OF STRESS URINARY INCONTINENCE * Ananias C. Diokno, MD ABSTRACT Urinary incontinence is a worldwide problem that affects millions of women, although the magnitude of the problem
INCIDENCE AND PREVALENCE OF STRESS URINARY INCONTINENCE * Ananias C. Diokno, MD ABSTRACT Urinary incontinence is a worldwide problem that affects millions of women, although the magnitude of the problem
Advances In Spine Care. James D. Bruffey M.D. Scripps Clinic Division of Orthopaedic Surgery Section of Spinal Surgery
 Advances In Spine Care James D. Bruffey M.D. Scripps Clinic Division of Orthopaedic Surgery Section of Spinal Surgery Introduction The Spine - A common source of problems Back pain is the #2 presenting
Advances In Spine Care James D. Bruffey M.D. Scripps Clinic Division of Orthopaedic Surgery Section of Spinal Surgery Introduction The Spine - A common source of problems Back pain is the #2 presenting
Service delivery interventions
 Service delivery interventions S A S H A S H E P P E R D D E P A R T M E N T O F P U B L I C H E A L T H, U N I V E R S I T Y O F O X F O R D CO- C O O R D I N A T I N G E D I T O R C O C H R A N E E P
Service delivery interventions S A S H A S H E P P E R D D E P A R T M E N T O F P U B L I C H E A L T H, U N I V E R S I T Y O F O X F O R D CO- C O O R D I N A T I N G E D I T O R C O C H R A N E E P
2. Does the patient have one of the following appropriate indications for placing indwelling urinary catheters?
 A. Decision to Insert a Urinary Catheter: 1. Before placing an indwelling catheter, please consider if these alternatives would be more appropriate: Bladder scanner: to assess and confirm urinary retention,
A. Decision to Insert a Urinary Catheter: 1. Before placing an indwelling catheter, please consider if these alternatives would be more appropriate: Bladder scanner: to assess and confirm urinary retention,
Implementing the Long-Term Care UTI Toolkit Wisconsin Coalition on HealthCare Association Infection in Long Term Care
 Implementing the Long-Term Care UTI Toolkit Wisconsin Coalition on HealthCare Association Infection in Long Term Care MetaStar Health Care Quality Symposium: The Value of Better Health Paula Kock RN paulak@parkmanorwi.com
Implementing the Long-Term Care UTI Toolkit Wisconsin Coalition on HealthCare Association Infection in Long Term Care MetaStar Health Care Quality Symposium: The Value of Better Health Paula Kock RN paulak@parkmanorwi.com
COMMITTEE FOR PROPRIETARY MEDICINAL PRODUCTS (CPMP)
 The European Agency for the Evaluation of Medicinal Products Evaluation of Medicines for Human Use London, 18 December 2002 COMMITTEE FOR PROPRIETARY MEDICINAL PRODUCTS (CPMP) NOTE FOR GUIDANCE ON THE
The European Agency for the Evaluation of Medicinal Products Evaluation of Medicines for Human Use London, 18 December 2002 COMMITTEE FOR PROPRIETARY MEDICINAL PRODUCTS (CPMP) NOTE FOR GUIDANCE ON THE
