Irish Medicines Board
|
|
|
- Myra Sutton
- 7 years ago
- Views:
Transcription
1 IRISH MEDICINES BOARD ACTS 1995 AND 2006 MEDICINAL PRODUCTS(CONTROL OF PLACING ON THE MARKET)REGULATIONS,2007 (S.I. No.540 of 2007) PA0030/038/001 Case No: The Irish Medicines Board in exercise of the powers conferred on it by the above mentioned Regulations hereby grants to Novartis Consumer Health UK Limited Wimblehurst Road, Horsham, West Sussex RH12 5AB, United Kingdom an authorisation, subject to the provisions of the said Regulations, in respect of the product Calcium-D-Sandoz 600 mg IU, effervescent tablets The particulars of which are set out in Part I and Part II of the attached Schedule. The authorisation is also subject to the general conditions as may be specified in the said Regulations as listed on the reverse of this document. This authorisation, unless previously revoked, shall continue in force from 30/03/2010. Signed on behalf of the Irish Medicines Board this A person authorised in that behalf by the said Board. Date Printed 31/03/2010 CRN page number: 1
2 1 NAME OF THE MEDICINAL PRODUCT Calcium-D-Sandoz 600 mg IU, effervescent tablets. Part II Summary of Product Characteristics 2 QUALITATIVE AND QUANTITATIVE COMPOSITION 1 effervescent tablet contains 1500 mg of calcium carbonate (equivalent to 600 mg or 15 mmol of calcium) 400 I.U. or 10 µg cholecalciferol (vitamin D 3 ) as cholecalciferol concentrate powder form. Excipient(s): 52 mg of sodium, 1.52 mg of sucrose, 50 mg of lemon flavouring (contains: sorbitol (E 420)), 0.30 mg of partially hydrogenated soybean oil For a full list of excipients, see section PHARMACEUTICAL FORM Effervescent tablet White-round tablet, smooth and not vaulted. 4 CLINICAL PARTICULARS 4.1 Therapeutic Indications Correction of combined vitamin D 3 and calcium deficiencies. Vitamin D 3 and calcium supplementation as an adjunct to specific treatment for osteoporosis in patients where combined calcium and vitamin D deficiencies have been diagnosed or at high risk of such deficiencies. 4.2 Posology and method of administration Adults take 1-2 effervescent tablets daily (equivalent to mg of calcium and I.U. of vitamin D 3 ). For pregnancy and lactation, see section 4.6 Pregnancy and Lactation. The effervescent tablets should be dissolved in a glass of water (approx. 200 ml) and drunk immediately. Dosage in hepatic impairment: No dose adjustment is required. Dosage in renal impairment: Calcium-D-Sandoz should not be used in patients with severe renal impairment. Oral use - For adults only. 4.3 Contraindications Hypersensitivity to the active substances, soya, peanut or to any of the excipients, Hypercalcaemia, hypercalciuria, Nephrocalcinosis, nephrolithiasis Date Printed 31/03/2010 CRN page number: 2
3 Disease and/or conditions resulting in hypercalcaemia and/or hypercalciuria (e.eg. primary hyperparathyroidism, myeloma, bone metastases) Hypervitaminosis D Renal failure 4.4 Special warnings and precautions for use During long-term treatment, serum calcium levels should be followed and renal function should be monitored through measurements of serum creatinine. Monitoring is especially important in patients on concomitant treatment with cardiac glycosides or thiazide diuretics (see section 4.5) and in patients with a high tendency to calculus formation. In case of hypercalcaemia or signs of impaired renal function the dose should be reduced or the treatment discontinued. Therapy should be reduced or preliminary interrupted, if urinary calcium level exceeds 7.5 mmol/24 h (300 mg/24 h). Vitamin D should be used with caution in patients with impairment of renal function and the effect on calcium and phosphate levels should be monitored. The risk of soft tissue calcification should be taken into account. In patients with severe renal insufficiency, vitamin D in the form of cholecalciferol is not metabolised normally and other forms of vitamin D should be used. Calcium-D-Sandoz should be prescribed with caution to patients suffering from sarcoidosis, due to the risk of increased metabolism of vitamin D into its active form. These patients should be monitored with regard to the calcium content in serum and urine. Calcium-D-Sandoz should be used cautiously in immobilised patients with osteoporosis due to increased risk of hypercalcaemia. The content of vitamin D (400 IU) in Calcium-D-Sandoz should be considered when prescribing other medicinal products containing vitamin D. Additional doses of calcium or vitamin D should be taken under close medical supervision. In such cases it is necessary to monitor serum calcium levels and urinary calcium excretion frequently. Calcium and Vitamin D intake from other sources (food, dietary supplements) should be estimated, before prescribing the product. Calcium-D-Sandoz is not intended for use in children and adolescents. Calcium-D-Sandoz contains 2.26 mmol (corresponding to 52 mg) of sodium per tablet. The product contains sorbitol, therefore patients with rare hereditary problems of fructose intolerance should not take this medicine. The product contains sucrose, therefore patients with rare hereditary problems of fructose intolerance, glucosegalactose malabsorption or sucrase-isomaltase insufficiency should not take this medicine. Information for diabetics: 1 effervescent tablet contains 0.01 Bread Units and is therefore suitable for diabetics. 4.5 Interaction with other medicinal products and other forms of interaction Concomitant use of phenytoin or other barbiturates may reduce the effect of vitamin D3 since the metabolism increases. Thiazide diuretics reduce the urinary excretion of calcium. Due to increased risk of hypercalcaemia, serum calcium should be regularly monitored during concomitant use of thiazide diuretics. Systemic corticosteroids reduce calcium absorption. During concomitant use, it may be necessary to increase the dose of Calcium-D-Sandoz. Date Printed 31/03/2010 CRN page number: 3
4 Simultaneous treatment with ion exchange resins such as cholestyramine or laxatives such as paraffin oil may reduce the gastrointestinal absorption of vitamin D. Therefore a time interval as long as possible between the intakes should be recommended. Calcium carbonate may interfere with the absorption of concomitantly administered tetracycline preparations. For this reason, tetracycline preparations should be administered at least two hours before or four to six hours after oral intake of calcium. Hypercalcaemia may increase the toxicity of cardiac glycosides during treatment with calcium and vitamin D. Patients should be monitored with regard to electrocardiogram (ECG) and serum calcium levels. If a bisphosphonate or sodium fluoride is used concomitantly, this preparation should be administered at least three hours before the intake of Calcium-D-Sandoz since gastrointestinal absorption of bisphosphonate may be reduced. Oxalic acid (found in spinach and rhubarb) and phytic acid (found in whole cereals) may inhibit calcium absorption through formation of insoluble compounds with calcium ions. The patient should not take calcium products within two hours of eating foods high in oxalic acid and phytic acid. 4.6 Pregnancy and lactation During pregnancy and lactation, combined vitamin D and calcium deficiencies can be corrected. The daily intake should not exceed 1,500 mg of calcium and 600 I.U. of vitamin D 3. Therefore, the daily dose must not exceed 1 tablet. Overdoses of vitamin D have been shown to have teratogenic effects in animal experiments. In pregnant women, overdosage of vitamin D 3 should be avoided, since prolonged hypercalcaemia has been sometimes associated with retardation of physical and mental development, supravalvular aortic stenosis and retinopathy in the child. There are, however, several case reports of administration of very high vitamin D doses in hypoparathyroidism in the mother where normal children were born. Calcium passes slightly into breast-milk, without having a negative effect on children. Vitamin D and its metabolites also pass into breast-milk. This should be considered when giving additional vitamin D to the child. In pregnant and lactating women, the calcium preparation should be taken at a distance of two hours from a meal due to a possible decrease of iron absorption. 4.7 Effects on ability to drive and use machines An unfavourable effect of the preparation on the ability to drive or operate machines is very unlikely. 4.8 Undesirable effects Immune system disorders Very Rare (<1/10,000): Hypersensitivity reactions such as angioedema or laryngeal oedema. Metabolism and nutrition disorders Uncommon (>1/1,000, <1/100): hypercalcaemia, hypercalciuria. Unknown: milk-alkali syndrome Gastrointestinal disorders Rare (>1/10,000, <1/1,000): nausea, diarrhea, abdominal pain, constipation, flatulence, abdominal distension. Unknown: vomiting Date Printed 31/03/2010 CRN page number: 4
5 Skin and subcutaneous tissue disorders Rare (>1/10,000, <1/1,000): rash, pruritus, urticaria. 4.9 Overdose Overdosage leads to hypervitaminosis and hypercalcaemia with the following symptoms: Anorexia, nausea, vomiting, thirst, polydipsia, polyuria, dehydration, constipation, abdominal pain, muscle weakness, fatigue, mental disturbances, bone pain, nephrocalcinosis and in severe cases cardiac arrythmias. Extreme hypercalcemia may result in coma and death. Chronic overdosage with resulting hypercalcaemia can cause irreversible renal damage and soft tissue calcification. The threshold for vitamin D intoxication is between 40,000 and 100,000 I.U./day for 1-2 months in persons with normal parathyroid function, for calcium in excess of 2,000 mg per day. Treatment In the case of an intoxication, treatment should be stopped immediately and the fluid deficiency should be balanced. Treatment with thiazide diuretics, lithium, Vitamin A and cardiac glycosides must also be discontinued. Emptying of the stomach in patients with impaired consciousness. Rehydration and, according to severity, isolated or combined treatment with loop diuretics, bisphosphonates, calcitonin and corticosteroids. Serum electrolytes, renal function and diuresis must be monitored. In severe cases ECG and CVP should be followed. 5 PHARMACOLOGICAL PROPERTIES 5.1 Pharmacodynamic properties Pharmacotherapeutic group: Mineral supplements ATC code: Calcium, combinations with other drugs (A12AX) Calcium-D-Sandoz is a fixed combination of calcium and vitamin D. The high calcium and vitamin D concentration in each dose unit enables sufficient absorption of calcium with a limited number of doses. Vitamin D is involved in calcium-phosphorus metabolism. It allows the active absorption of calcium and phosphorus from the intestine and their uptake by bone. Supplementation with calcium and vitamin D 3 corrects latent vitamin D deficiency and secondary hyperparathyroidism. The optimal amount of Vitamin D in the elderly is IU/day. The commonly accepted requirement of calcium in the elderly is 1500 mg/day. In a double-blind placebo controlled study of 18 months, including 3270 women aged 84 ± 6 and living in nursing homes, supplemented with cholecalciferol (800 IU/day) + Calcium (1.2 g/day), a significant decrease in PTH secretion has been observed. After 18 months, the results of the intent to treat analysis showed 80 hip fractures in the calcium vitamin D group and 110 hip fractures in the placebo-group (p=0.004). So in the conditions of this study, the treatment of 1387 women prevented 30 hip fractures. After 36 months of follow-up,137 women presented at least one hip fracture in the calcium-vitamin D group (n=1176) and 178 in the placebo group (n=1127) (p 0.02). 5.2 Pharmacokinetic properties Calcium carbonate: Absorption: On dissolution of the effervescent tablet, the calcium carbonate is converted in the presence of citric acid to soluble calcium citrate. Some 30-40% of the ingested dose of calcium is absorbed, predominantly in the proximal part of the small intestine, through Vitamin D dependant saturable active transport. Date Printed 31/03/2010 CRN page number: 5
6 Elimination: Calcium is excreted in the urine, faeces and in sweat. Urinary excretion depends on glomerular filtration and tubular resorption. Vitamin D 3 : Absorption: Vitamin D 3 is absorbed in the intestine and transported by protein binding in the blood to the liver (where it undergoes the first hydroxylation to 25-hydroxycholecalciferol) and to the kidneys (second hydroxylation to 1,25- dihydroxycholecalciferol), the actual active metabolite of vitamin D 3. Non-hydroxylated vitamin D 3 is stored in muscle and adipose tissues. Elimination: The plasma half-life is in the order of several days; vitamin D 3 is eliminated in the faeces and urine. 5.3 Preclinical safety data No other relevant data is available that has not been mentioned elsewhere in the Summary of Product Characteristics (see 4.6 Pregnancy and lactation; 4.9 Overdose). 6 PHARMACEUTICAL PARTICULARS 6.1 List of excipients Citric acid anhydrous, Malic acid, Sodium hydrogen carbonate, Sodium cyclamate, Lemon flavouring (contains: lemon oil, mannitol, sorbitol, dextrin, D-glucono-1,5-lactone, acacia), Sodium carbonate, Maltodextrin, Saccharin sodium, Sucrose, Gelatin, Maize starch, Partially hydrogenated soya oil, α-tocopherol. 6.2 Incompatibilities Not applicable. 6.3 Shelf Life 3 years. Date Printed 31/03/2010 CRN page number: 6
7 6.4 Special precautions for storage Do not store above 30 C. Keep the container tightly closed. 6.5 Nature and contents of container Packs of 20, 40 (2 x 20), 60 (3 x 20) and 100 (5 packs of 20) effervescent tablets. Each unit of 20 tablets is in an aluminium or polypropylene tube with polyethylene stopper. Not all pack sizes may be marketed. 6.6 Special precautions for disposal of a used medicinal product or waste materials derived from such medicinal product and other handling of the product No special requirements. 7 MARKETING AUTHORISATION HOLDER Novartis Consumer Health UK Limited Trading as Novartis Consumer Health Wimblehurst Road Horsham RH12 5AB West Sussex England 8 MARKETING AUTHORISATION NUMBER PA 0030/038/001 9 DATE OF FIRST AUTHORISATION/RENEWAL OF THE AUTHORISATION Date of first authorisation: 28 th April 2000 Date of last renewal: 14 th January DATE OF REVISION OF THE TEXT August 2009 Date Printed 31/03/2010 CRN page number: 7
(NON-PRESCRIPTION) LEAFLET: USER INFORMATION. CALTRATE VITAMIN D3 600mg/400 IU, film-coated tablet Calcium and Cholecalciferol
 (NON-PRESCRIPTION) LEAFLET: USER INFORMATION CALTRATE VITAMIN D3 600mg/400 IU, film-coated tablet Calcium and Cholecalciferol Read all of this leaflet carefully because it contains important information
(NON-PRESCRIPTION) LEAFLET: USER INFORMATION CALTRATE VITAMIN D3 600mg/400 IU, film-coated tablet Calcium and Cholecalciferol Read all of this leaflet carefully because it contains important information
For the use of a Registered Medical Practitioner or a Hospital or a Laboratory only OR for Specialist Use only
 For the use of a Registered Medical Practitioner or a Hospital or a Laboratory only OR for Specialist Use only Cholecalciferol Granules VITOMIN D3 COMPOSITION Each sachet of 1 g contains: Cholecalciferol
For the use of a Registered Medical Practitioner or a Hospital or a Laboratory only OR for Specialist Use only Cholecalciferol Granules VITOMIN D3 COMPOSITION Each sachet of 1 g contains: Cholecalciferol
PHOSPHATE-SANDOZ Tablets (High dose phosphate supplement)
 1 PHOSPHATE-SANDOZ Tablets (High dose phosphate supplement) PHOSPHATE-SANDOZ PHOSPHATE-SANDOZ Tablets are a high dose phosphate supplement containing sodium phosphate monobasic. The CAS registry number
1 PHOSPHATE-SANDOZ Tablets (High dose phosphate supplement) PHOSPHATE-SANDOZ PHOSPHATE-SANDOZ Tablets are a high dose phosphate supplement containing sodium phosphate monobasic. The CAS registry number
Calcium and Colecalciferol
 30042015 PACKAGE LEAFLET: INFORMATION FOR THE USER Calcium and Colecalciferol Read all of this leaflet carefully before you start taking this medicine.
30042015 PACKAGE LEAFLET: INFORMATION FOR THE USER Calcium and Colecalciferol Read all of this leaflet carefully before you start taking this medicine.
PHARMACOLOGICAL PROPERTIES
 ANTİ-FOSFAT ca 700 mg Film Tablet COMPOSITION 1 film tablet contains 700 mg Calcium acetate, as an active ingredient. Excipients are Microcrystalline cellulose, Povidone K30, Crosspovidone, Magnesium stearate,
ANTİ-FOSFAT ca 700 mg Film Tablet COMPOSITION 1 film tablet contains 700 mg Calcium acetate, as an active ingredient. Excipients are Microcrystalline cellulose, Povidone K30, Crosspovidone, Magnesium stearate,
Package leaflet: Information for the patient. Cholecalciferol mibe 500 IE, tabletten Cholecalciferol. For use in infants, children and adults
 Package leaflet: Information for the patient Cholecalciferol mibe 500 IE, tabletten Cholecalciferol For use in infants, children and adults Read all of this leaflet carefully before you start taking this
Package leaflet: Information for the patient Cholecalciferol mibe 500 IE, tabletten Cholecalciferol For use in infants, children and adults Read all of this leaflet carefully before you start taking this
MRP-No. DE/H/0279/001/P/002 Dr. Scheffler Vitamin C, 1000mg, effervescent tablets
 PACKAGE LEAFLET: INFORMATION FOR THE USER Dr. Scheffler Vitamin C, 1000 mg, effervescent tablets Ascorbic Acid Read all of this leaflet carefully because it contains important information for you. This
PACKAGE LEAFLET: INFORMATION FOR THE USER Dr. Scheffler Vitamin C, 1000 mg, effervescent tablets Ascorbic Acid Read all of this leaflet carefully because it contains important information for you. This
ALPHA D 3 0.5 mcg Capsules
 "פורמט עלון זה נקבע ע"י משרד הבריאות ותוכנו נבדק ואושר". עלון מאושר: אפריל 2010 This leaflet format has been determined by the Ministry of Health and the content thereof has been checked and approved.
"פורמט עלון זה נקבע ע"י משרד הבריאות ותוכנו נבדק ואושר". עלון מאושר: אפריל 2010 This leaflet format has been determined by the Ministry of Health and the content thereof has been checked and approved.
PATIENT INFORMATION LEAFLET. Calcium Sandoz Syrup calcium glubionate and calcium lactobionate
 PATIENT INFORMATION LEAFLET Calcium Sandoz Syrup calcium glubionate and calcium lactobionate Read all of this leaflet carefully before you start taking this medicine Keep this leaflet. You may need to
PATIENT INFORMATION LEAFLET Calcium Sandoz Syrup calcium glubionate and calcium lactobionate Read all of this leaflet carefully before you start taking this medicine Keep this leaflet. You may need to
NEUROTONE THR 00904/0005 UKPAR
 NEUROTONE THR 00904/0005 UKPAR TABLE OF CONTENTS Lay summary Page 2 Scientific discussion Page 3 Steps taken for assessment Page 10 Summary of Product Characteristics Page 11 Product Information Leaflet
NEUROTONE THR 00904/0005 UKPAR TABLE OF CONTENTS Lay summary Page 2 Scientific discussion Page 3 Steps taken for assessment Page 10 Summary of Product Characteristics Page 11 Product Information Leaflet
Always take this medicine exactly as described in this leaflet or as your doctor, pharmacist or nurse have told you.
 leaflet: Information for the user Macrogol 4000 10 g powder for oral solution in sachet Macrogol 4000
leaflet: Information for the user Macrogol 4000 10 g powder for oral solution in sachet Macrogol 4000
Public Assessment Report. UK National Procedure. (colecalciferol) PL 16508/0047 PL 16508/0048. ProStrakan Ltd.
 Public Assessment Report UK National Procedure STEXEROL-D 3 1,000 IU FILM-COATED TABLETS STEXEROL-D 3 25,000 IU FILM-COATED TABLETS (colecalciferol) PL 16508/0047 PL 16508/0048 ProStrakan Ltd. 1 LAY SUMMARY
Public Assessment Report UK National Procedure STEXEROL-D 3 1,000 IU FILM-COATED TABLETS STEXEROL-D 3 25,000 IU FILM-COATED TABLETS (colecalciferol) PL 16508/0047 PL 16508/0048 ProStrakan Ltd. 1 LAY SUMMARY
EFFIMET 1000 XR Metformin Hydrochloride extended release tablet
 BRAND NAME: Effimet XR. THERAPEUTIC CATEGORY: Anti-Diabetic PHARMACOLOGIC CLASS: Biguanides EFFIMET 1000 XR Metformin Hydrochloride extended release tablet COMPOSITION AND PRESENTATION Composition Each
BRAND NAME: Effimet XR. THERAPEUTIC CATEGORY: Anti-Diabetic PHARMACOLOGIC CLASS: Biguanides EFFIMET 1000 XR Metformin Hydrochloride extended release tablet COMPOSITION AND PRESENTATION Composition Each
SUMMARY OF THE RISK MANAGEMENT PLAN (by medicinal product)
 PART VI SUMMARY OF THE RISK MANAGEMENT PLAN (by medicinal product) Format and content of the summary of the RMP The summary of the RMP part VI contains information based on RMP modules SI, SVIII and RMP
PART VI SUMMARY OF THE RISK MANAGEMENT PLAN (by medicinal product) Format and content of the summary of the RMP The summary of the RMP part VI contains information based on RMP modules SI, SVIII and RMP
Public Assessment Report. Scientific discussion. Calcium and Vitamine D3 Alpex 1000 mg/880 IE, effervescent granules
 Public Assessment Report Scientific discussion Calcium and Vitamine D3 Alpex 1000 mg/880 IE, effervescent granules (calcium carbonate and cholecalciferol) NL License RVG: 111783 Date: 12 March 2015 This
Public Assessment Report Scientific discussion Calcium and Vitamine D3 Alpex 1000 mg/880 IE, effervescent granules (calcium carbonate and cholecalciferol) NL License RVG: 111783 Date: 12 March 2015 This
SUMMARY OF PRODUCT CHARACTERISTICS
 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE VETERINARY MEDICINAL PRODUCT Prednicare Tablets 5mg 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each tablet contains: Active Substance(s) Prednisolone
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE VETERINARY MEDICINAL PRODUCT Prednicare Tablets 5mg 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each tablet contains: Active Substance(s) Prednisolone
PACKAGE LEAFLET: INFORMATION FOR THE USER Paracetamol 500 mg Effervescent Tablets Paracetamol
 PACKAGE LEAFLET: INFORMATION FOR THE USER Paracetamol 500 mg Effervescent Tablets Paracetamol Read all of this leaflet carefully because it contains important information for you. This medicine is available
PACKAGE LEAFLET: INFORMATION FOR THE USER Paracetamol 500 mg Effervescent Tablets Paracetamol Read all of this leaflet carefully because it contains important information for you. This medicine is available
PACKAGE LEAFLET: INFORMATION FOR THE USER. VITAMINE B12 STEROP 1mg/1ml Solution for injection / oral solution. Cyanocobalamin
 PACKAGE LEAFLET: INFORMATION FOR THE USER VITAMINE B12 STEROP 1mg/1ml Solution for injection / oral solution Cyanocobalamin Read all of this leaflet carefully before you start using this medicine because
PACKAGE LEAFLET: INFORMATION FOR THE USER VITAMINE B12 STEROP 1mg/1ml Solution for injection / oral solution Cyanocobalamin Read all of this leaflet carefully before you start using this medicine because
Mucodyne-Clear 250 mg/5 ml Syrup PL 04425/0665
 Mucodyne-Clear 250 mg/5 ml Syrup PL 04425/0665 UKPAR TABLE OF CONTENTS Lay summary Page 2 Scientific discussion Page 3 Steps taken for assessment Page 9 Summary of product characteristics Page 10 Patient
Mucodyne-Clear 250 mg/5 ml Syrup PL 04425/0665 UKPAR TABLE OF CONTENTS Lay summary Page 2 Scientific discussion Page 3 Steps taken for assessment Page 9 Summary of product characteristics Page 10 Patient
Public Assessment Report. Decentralised Procedure. CosmoCol Half 6.9 g, powder for oral solution CosmoCol Paediatric 6.9 g, powder for oral solution
 Public Assessment Report Decentralised Procedure CosmoCol Half 6.9 g, powder for oral solution CosmoCol Paediatric 6.9 g, powder for oral solution (Macrogol, sodium chloride, sodium hydrogen carbonate
Public Assessment Report Decentralised Procedure CosmoCol Half 6.9 g, powder for oral solution CosmoCol Paediatric 6.9 g, powder for oral solution (Macrogol, sodium chloride, sodium hydrogen carbonate
Clinical Aspects of Hyponatremia & Hypernatremia
 Clinical Aspects of Hyponatremia & Hypernatremia Case Presentation: History 62 y/o male is admitted to the hospital with a 3 month history of excessive urination (polyuria) and excess water intake up to
Clinical Aspects of Hyponatremia & Hypernatremia Case Presentation: History 62 y/o male is admitted to the hospital with a 3 month history of excessive urination (polyuria) and excess water intake up to
Adcal-D3 Caplets, 750 mg/200 I.U, film-coated tablets (calcium carbonate, vitamin D 3 ) PL 16508/0039
 Adcal-D3 Caplets, 750 mg/200 I.U, film-coated tablets (calcium carbonate, vitamin D 3 ) PL 16508/0039 UKPAR TABLE OF CONTENTS Lay Summary Page 2 Scientific discussion Page 3 Steps taken for assessment
Adcal-D3 Caplets, 750 mg/200 I.U, film-coated tablets (calcium carbonate, vitamin D 3 ) PL 16508/0039 UKPAR TABLE OF CONTENTS Lay Summary Page 2 Scientific discussion Page 3 Steps taken for assessment
4 Clinical Particulars
 SUMMARY OF PRODUCT CHARACTERISTICS 1 Name of the Medicinal Product Procyclidine Syrup 5mg/5ml 2. Qualitative and Quantitative Composition Each 5ml dose contains 5mg Procyclidine Hydrochloride BP. 3. Pharmaceutical
SUMMARY OF PRODUCT CHARACTERISTICS 1 Name of the Medicinal Product Procyclidine Syrup 5mg/5ml 2. Qualitative and Quantitative Composition Each 5ml dose contains 5mg Procyclidine Hydrochloride BP. 3. Pharmaceutical
NEW ZEALAND DATA SHEET
 Presentation Vit.D3 1.25 mg (equivalent to 50,000IU) capsules are presented as natural coloured, transparent, oval shaped, soft gelatin capsules, containing pale yellow coloured oil. Uses INDICATIONS Colecalciferol
Presentation Vit.D3 1.25 mg (equivalent to 50,000IU) capsules are presented as natural coloured, transparent, oval shaped, soft gelatin capsules, containing pale yellow coloured oil. Uses INDICATIONS Colecalciferol
Kalms Tablets THR 01074/0235 UKPAR
 Kalms Tablets THR 01074/0235 UKPAR TABLE OF CONTENTS Lay summary Page 2 Scientific discussion Page 3 Steps taken for assessment Page 10 Summary of Product Characteristics Page 11 Patient Information Leaflet
Kalms Tablets THR 01074/0235 UKPAR TABLE OF CONTENTS Lay summary Page 2 Scientific discussion Page 3 Steps taken for assessment Page 10 Summary of Product Characteristics Page 11 Patient Information Leaflet
DVITA PLUS Vitamin D 1000 IU Tablet INDICATED CLAIMS:
 DVITA PLUS Vitamin D 1000 IU Tablet INDICATED CLAIMS: Vitamin D is the principal regulator of calcium homeostasis in the body Vitamin D is particularly important in skeletal development and bone mineralization.
DVITA PLUS Vitamin D 1000 IU Tablet INDICATED CLAIMS: Vitamin D is the principal regulator of calcium homeostasis in the body Vitamin D is particularly important in skeletal development and bone mineralization.
Calcium Folinate Ebewe Data Sheet
 NAME OF THE MEDICINE Calcium folinate injection Composition Active: Calcium folinate (equivalent to 10 mg folinic acid per ml) Inactive: Sodium chloride (7.7mg/mL), qs Water for Injections. Preservative
NAME OF THE MEDICINE Calcium folinate injection Composition Active: Calcium folinate (equivalent to 10 mg folinic acid per ml) Inactive: Sodium chloride (7.7mg/mL), qs Water for Injections. Preservative
Package leaflet: Information for the patient. Laxido Orange, powder for oral solution
 Package leaflet: Information for the patient Laxido Orange, powder for oral solution Read all of this leaflet carefully before you start taking this medicine because it contains important information for
Package leaflet: Information for the patient Laxido Orange, powder for oral solution Read all of this leaflet carefully before you start taking this medicine because it contains important information for
Corporate Medical Policy
 Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: testing_serum_vitamin_d_levels 9/2015 2/2016 2/2017 2/2016 Description of Procedure or Service Vitamin D,
Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: testing_serum_vitamin_d_levels 9/2015 2/2016 2/2017 2/2016 Description of Procedure or Service Vitamin D,
HYPERTENSION ASSOCIATED WITH RENAL DISEASES
 RENAL DISEASE v Patients with renal insufficiency should be encouraged to reduce dietary salt and protein intake. v Target blood pressure is less than 135-130/85 mmhg. If patients have urinary protein
RENAL DISEASE v Patients with renal insufficiency should be encouraged to reduce dietary salt and protein intake. v Target blood pressure is less than 135-130/85 mmhg. If patients have urinary protein
Calcium (serum, plasma, blood)
 Calcium (serum, plasma, blood) 1 Name and description of analyte 1.1 Name of analyte Calcium (total in serum, plasma, ionised in blood (see 2.1 (2)). 1.2 Alternative names None 1.3 NMLC code 1.4 Description
Calcium (serum, plasma, blood) 1 Name and description of analyte 1.1 Name of analyte Calcium (total in serum, plasma, ionised in blood (see 2.1 (2)). 1.2 Alternative names None 1.3 NMLC code 1.4 Description
PATIENT INFORMATION LEAFLET: CENTRUM. Read the contents of this leaflet carefully before you start using CENTRUM, because it
 PATIENT INFORMATION LEAFLET: CENTRUM Page 1 of 7 This leaflet tells you about CENTRUM tablets. Read the contents of this leaflet carefully before you start using CENTRUM, because it contains important
PATIENT INFORMATION LEAFLET: CENTRUM Page 1 of 7 This leaflet tells you about CENTRUM tablets. Read the contents of this leaflet carefully before you start using CENTRUM, because it contains important
INITIATING ORAL AUBAGIO (teriflunomide) THERAPY
 FOR YOUR PATIENTS WITH RELAPSING FORMS OF MS INITIATING ORAL AUBAGIO (teriflunomide) THERAPY WARNING: HEPATOTOXICITY AND RISK OF TERATOGENICITY Severe liver injury including fatal liver failure has been
FOR YOUR PATIENTS WITH RELAPSING FORMS OF MS INITIATING ORAL AUBAGIO (teriflunomide) THERAPY WARNING: HEPATOTOXICITY AND RISK OF TERATOGENICITY Severe liver injury including fatal liver failure has been
P AC K AG E L E AF L E T: INFORMAT I ON FO R THE USER. 500 mg, film-coated tablet Active substance: metformin hydrochloride
 P AC K AG E L E AF L E T: INFORMAT I ON FO R THE USER Siofor 500 500 mg, film-coated tablet Active substance: metformin hydrochloride For use in children above 10 years and adults Read all of this leaflet
P AC K AG E L E AF L E T: INFORMAT I ON FO R THE USER Siofor 500 500 mg, film-coated tablet Active substance: metformin hydrochloride For use in children above 10 years and adults Read all of this leaflet
Summary of Product Characteristics
 1 NAME OF THE MEDICINAL PRODUCT Librium 10mg Hard capsule Summary of Product Characteristics 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each capsule contains 10 mg of chlordiazepoxide hydrochloride. Excipients:
1 NAME OF THE MEDICINAL PRODUCT Librium 10mg Hard capsule Summary of Product Characteristics 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each capsule contains 10 mg of chlordiazepoxide hydrochloride. Excipients:
SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT 2 QUALITATIVE AND QUANTITATIVE COMPOSITION
 SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Eskazole Tablets 400 mg 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Albendazole 400 mg Excipients with known effect: Lactose, sunset yellow
SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Eskazole Tablets 400 mg 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Albendazole 400 mg Excipients with known effect: Lactose, sunset yellow
D. Vitamin D. 1. Two main forms; vitamin D2 and D3
 D. Vitamin D. Two main forms; vitamin D2 and D3 H H D3 - Cholecalciferol D2 - Ergocalciferol Technically, vitamin D is not a vitamin. It is the name given to a group of fat-soluble prohormones (substances
D. Vitamin D. Two main forms; vitamin D2 and D3 H H D3 - Cholecalciferol D2 - Ergocalciferol Technically, vitamin D is not a vitamin. It is the name given to a group of fat-soluble prohormones (substances
Considerations With Calcium And Vitamin D Supplementation
 Considerations With Calcium And Vitamin D Supplementation Activity Preview Calcium is an electrolyte involved in many systems in the body including bone health, nerve signaling pathways, muscle contractions,
Considerations With Calcium And Vitamin D Supplementation Activity Preview Calcium is an electrolyte involved in many systems in the body including bone health, nerve signaling pathways, muscle contractions,
One vial contains 500 U* of C1-inhibitor** After reconstitution the product contains 500 U/5 ml which correspond to a concentration of 100 U/ml.
 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Cetor 100 U/ml powder and solvent for solution for injection 2. QUALITATIVE AND QUANTITATIVE COMPOSITION One vial contains 500 U* of
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Cetor 100 U/ml powder and solvent for solution for injection 2. QUALITATIVE AND QUANTITATIVE COMPOSITION One vial contains 500 U* of
Week 30. Water Balance and Minerals
 Week 30 Water Balance and Minerals Water: more vital to life than food involved in almost every body function is not stored--excreted daily largest single constituent of the human body, averaging 60% of
Week 30 Water Balance and Minerals Water: more vital to life than food involved in almost every body function is not stored--excreted daily largest single constituent of the human body, averaging 60% of
Public Assessment Report Scientific discussion. Tenofovir disoproxil Teva (tenofovir disoproxil) SE/H/1432/01/DC
 Public Assessment Report Scientific discussion Tenofovir disoproxil Teva (tenofovir disoproxil) SE/H/1432/01/DC This module reflects the scientific discussion for the approval of Tenofovir disoproxil Teva.
Public Assessment Report Scientific discussion Tenofovir disoproxil Teva (tenofovir disoproxil) SE/H/1432/01/DC This module reflects the scientific discussion for the approval of Tenofovir disoproxil Teva.
ZOVIRAX Cold Sore Cream
 Data Sheet ZOVIRAX Cold Sore Cream Aciclovir 5% w/w Presentation Topical cream Indications ZOVIRAX Cold Sore Cream is indicated for the treatment of Herpes simplex virus infections of the lips and face
Data Sheet ZOVIRAX Cold Sore Cream Aciclovir 5% w/w Presentation Topical cream Indications ZOVIRAX Cold Sore Cream is indicated for the treatment of Herpes simplex virus infections of the lips and face
ENZAR FORTE TABLETS. (derived from Pancreatin USP) Sodium tauroglycocholate BPC 65mg (with sugar coating containing essential carminative oils)
 ENZAR FORTE TABLETS COMPOSITION Each enteric-coated tablet contains: Amylase activity -15,000 USP units Lipase activity -4,000 USP units Protease activity -15,000 USP units of (derived from Pancreatin
ENZAR FORTE TABLETS COMPOSITION Each enteric-coated tablet contains: Amylase activity -15,000 USP units Lipase activity -4,000 USP units Protease activity -15,000 USP units of (derived from Pancreatin
DEXTROMETHORPHAN HYDROBROMIDE 10MG/5ML ORAL SOLUTION PL 17907/0314
 DEXTROMETHORPHAN HYDROBROMIDE 10MG/5ML ORAL SOLUTION PL 17907/0314 UKPAR TABLE OF CONTENTS Lay Summary Page 2 Scientific discussion Page 3 Steps taken for assessment Page 10 Summary of Product Characteristics
DEXTROMETHORPHAN HYDROBROMIDE 10MG/5ML ORAL SOLUTION PL 17907/0314 UKPAR TABLE OF CONTENTS Lay Summary Page 2 Scientific discussion Page 3 Steps taken for assessment Page 10 Summary of Product Characteristics
Thioctacid 600 T Solution for Injection contains 600 mg alpha-lipoic acid
 Package Leaflet: Information for the User Thioctacid 600 T Solution for Injection contains 600 mg alpha-lipoic acid For use in adults Active substance: Alpha-lipoic acid, Trometamol salt (1:1) Read all
Package Leaflet: Information for the User Thioctacid 600 T Solution for Injection contains 600 mg alpha-lipoic acid For use in adults Active substance: Alpha-lipoic acid, Trometamol salt (1:1) Read all
Adams Memorial Hospital Decatur, Indiana EXPLANATION OF LABORATORY TESTS
 Adams Memorial Hospital Decatur, Indiana EXPLANATION OF LABORATORY TESTS Your health is important to us! The test descriptions listed below are for educational purposes only. Laboratory test interpretation
Adams Memorial Hospital Decatur, Indiana EXPLANATION OF LABORATORY TESTS Your health is important to us! The test descriptions listed below are for educational purposes only. Laboratory test interpretation
VISTARIL (hydroxyzine pamoate) Capsules and Oral Suspension
 VISTARIL (hydroxyzine pamoate) Capsules and Oral Suspension DESCRIPTION Hydroxyzine pamoate is designated chemically as 1-(p-chlorobenzhydryl) 4- [2-(2-hydroxyethoxy) ethyl] diethylenediamine salt of 1,1
VISTARIL (hydroxyzine pamoate) Capsules and Oral Suspension DESCRIPTION Hydroxyzine pamoate is designated chemically as 1-(p-chlorobenzhydryl) 4- [2-(2-hydroxyethoxy) ethyl] diethylenediamine salt of 1,1
PARACETAMOL REXIDOL. 600 mg Tablet. Analgesic-Antipyretic. Paracetamol 600 mg
 (Insert Text) UL Consumer Health PARACETAMOL REXIDOL 600 mg Tablet Analgesic-Antipyretic FORMULATION Each tablet contains: Paracetamol 600 mg PRODUCT DESCRIPTION Rexidol is a round, yellow, flat, bevel-edged
(Insert Text) UL Consumer Health PARACETAMOL REXIDOL 600 mg Tablet Analgesic-Antipyretic FORMULATION Each tablet contains: Paracetamol 600 mg PRODUCT DESCRIPTION Rexidol is a round, yellow, flat, bevel-edged
PL 17871/0208 UKPAR TABLE OF CONTENTS
 Fultium-D 3 3,200 IU Capsules PL 17871/0208 UKPAR TABLE OF CONTENTS Lay Summary Page 2 Scientific discussion Page 4 Steps taken for assessment Page 11 Summary of Product Characteristics Page 12 Patient
Fultium-D 3 3,200 IU Capsules PL 17871/0208 UKPAR TABLE OF CONTENTS Lay Summary Page 2 Scientific discussion Page 4 Steps taken for assessment Page 11 Summary of Product Characteristics Page 12 Patient
SR 5. W2364A_NT.XATRAL SR5 PAK/SA 5/12/05 9:47 Page 1
 W2364A_NT.XATRAL SR5 PAK/SA 5/12/05 9:47 Page 1 alfuzosin SR 5 mg (Alfuzosin HCI) COMPOSITION Each sustained-release, film-coated tablet contains: Alfuzosin hydrochloride......... 5 mg Excipients... q.s.
W2364A_NT.XATRAL SR5 PAK/SA 5/12/05 9:47 Page 1 alfuzosin SR 5 mg (Alfuzosin HCI) COMPOSITION Each sustained-release, film-coated tablet contains: Alfuzosin hydrochloride......... 5 mg Excipients... q.s.
Nursing 113. Pharmacology Principles
 Nursing 113 Pharmacology Principles 1. The study of how drugs enter the body, reach the site of action, and are removed from the body is called a. pharmacotherapeutics b. pharmacology c. pharmacodynamics
Nursing 113 Pharmacology Principles 1. The study of how drugs enter the body, reach the site of action, and are removed from the body is called a. pharmacotherapeutics b. pharmacology c. pharmacodynamics
PATIENT INFORMATION LEAFLET. Forceval Junior Capsules
 PATIENT INFORMATION LEAFLET Forceval Junior Capsules Read all of this leaflet carefully because it contains important information for you or your child. Please note this leaflet has been written as if
PATIENT INFORMATION LEAFLET Forceval Junior Capsules Read all of this leaflet carefully because it contains important information for you or your child. Please note this leaflet has been written as if
SUMMARY OF PRODUCT CHARACTERISTICS
 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Aknemycin Solution 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 10 g of solution contains 0.2 g of erythromycin. Structural formula of
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Aknemycin Solution 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 10 g of solution contains 0.2 g of erythromycin. Structural formula of
Summary of Product Characteristics
 Summary of Product Characteristics 1 NAME OF THE VETERINARY MEDICINAL PRODUCT ACEGON, 50 microgram/ml, solution for injection for cattle. 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each ml contains: Active
Summary of Product Characteristics 1 NAME OF THE VETERINARY MEDICINAL PRODUCT ACEGON, 50 microgram/ml, solution for injection for cattle. 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each ml contains: Active
UKPAR Goodnight THR 00904/0003. Goodnight THR 00904/0003 UKPAR TABLE OF CONTENTS. Lay Summary Page 2. Scientific discussion Page 3
 Goodnight THR 00904/0003 UKPAR TABLE OF CONTENTS Lay Summary Page 2 Scientific discussion Page 3 Steps taken for assessment Page 12 Steps taken after authorisation Page 13 Summary of Product Characteristics
Goodnight THR 00904/0003 UKPAR TABLE OF CONTENTS Lay Summary Page 2 Scientific discussion Page 3 Steps taken for assessment Page 12 Steps taken after authorisation Page 13 Summary of Product Characteristics
PATIENT INFORMATION LEAFLET. Forceval Capsules
 PATIENT INFORMATION LEAFLET Forceval Capsules Read all of this leaflet carefully because it contains important information for you. This medicine is available without prescription. However, you still need
PATIENT INFORMATION LEAFLET Forceval Capsules Read all of this leaflet carefully because it contains important information for you. This medicine is available without prescription. However, you still need
Doxylamine succinate belongs to the ethanolamine class of antihistamines with sedative properties.
 Data Sheet MERSYNDOL Tablet Paracetamol 450mg per tablet Codeine Phosphate 9.75mg per tablet Doxylamine Succinate 5mg per tablet MERSYNDOL FORTE Tablet Paracetamol 450mg per tablet Codeine Phosphate 30mg
Data Sheet MERSYNDOL Tablet Paracetamol 450mg per tablet Codeine Phosphate 9.75mg per tablet Doxylamine Succinate 5mg per tablet MERSYNDOL FORTE Tablet Paracetamol 450mg per tablet Codeine Phosphate 30mg
NUBONE +D Calcium/Vitamin D 500mg/200 IU Tablet
 NUBONE +D Calcium/Vitamin D 500mg/200 IU Tablet INDICATED CLAIMS: Helps in the maintenance of Bones and teeth. Calcium intake, when combined with Sufficient vitamin D, a healthy diet, and regular exercise,
NUBONE +D Calcium/Vitamin D 500mg/200 IU Tablet INDICATED CLAIMS: Helps in the maintenance of Bones and teeth. Calcium intake, when combined with Sufficient vitamin D, a healthy diet, and regular exercise,
See 17 for PATIENT COUNSELING INFORMATION. Revised: 3/2016 FULL PRESCRIBING INFORMATION: CONTENTS* WARNING: ADRENAL CRISIS IN THE SETTING OF SHOCK OR
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use LYSODREN safely and effectively. See full prescribing information for LYSODREN. LYSODREN (mitotane)
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use LYSODREN safely and effectively. See full prescribing information for LYSODREN. LYSODREN (mitotane)
SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT 2 QUALITATIVE AND QUANTITATIVE COMPOSITION
 SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Methadone 10mg/ml Injection / Physeptone 10mg/ml Injection 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Contains: Methadone Hydrochloride
SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Methadone 10mg/ml Injection / Physeptone 10mg/ml Injection 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Contains: Methadone Hydrochloride
SUMMARY OF PRODUCT CHARACTERISTICS. Paracetamol...10.00 mg for 1 ml of solution for infusion
 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT PARACETAMOL MACOPHARMA 10 mg/ml, solution for infusion 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Paracetamol...10.00 mg for 1 ml of
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT PARACETAMOL MACOPHARMA 10 mg/ml, solution for infusion 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Paracetamol...10.00 mg for 1 ml of
The format of this leaflet was determined by the Ministry of Health and its content was checked and approved in May 2013.
 The format of this leaflet was determined by the Ministry of Health and its content was checked and approved in May 2013 Havrix 720 Junior 1 NAME OF THE MEDICINAL PRODUCT Havrix 720 Junior 2 QUALITATIVE
The format of this leaflet was determined by the Ministry of Health and its content was checked and approved in May 2013 Havrix 720 Junior 1 NAME OF THE MEDICINAL PRODUCT Havrix 720 Junior 2 QUALITATIVE
INFORMATION FOR PATIENT. WARNINGS Using more than one enema in 24 hours can be harmful.
 FLEET ENEMA, A SALINE LAXATIVE - ready-to-use squeeze bottle FLEET ENEMA EXTRA, A SALINE LAXATIVE - ready-to-use squeeze bottle FLEET PEDIA-LAX ENEMA, A SALINE LAXATIVE - ready-to-use squeeze bottle FLEET
FLEET ENEMA, A SALINE LAXATIVE - ready-to-use squeeze bottle FLEET ENEMA EXTRA, A SALINE LAXATIVE - ready-to-use squeeze bottle FLEET PEDIA-LAX ENEMA, A SALINE LAXATIVE - ready-to-use squeeze bottle FLEET
FOR ORAL OR RECTAL ADMINISTRATION FOR THE PREVENTION AND TREATMENT OF PORTAL-SYSTEMIC ENCEPHALOPATHY
 FORM NO. 1360 VC3074 Rev.2/08 LACTULOSE SOLUTION, USP 10 g/15 ml FOR ORAL OR RECTAL ADMINISTRATION FOR THE PREVENTION AND TREATMENT OF PORTAL-SYSTEMIC ENCEPHALOPATHY Rx Only DESCRIPTION Lactulose is a
FORM NO. 1360 VC3074 Rev.2/08 LACTULOSE SOLUTION, USP 10 g/15 ml FOR ORAL OR RECTAL ADMINISTRATION FOR THE PREVENTION AND TREATMENT OF PORTAL-SYSTEMIC ENCEPHALOPATHY Rx Only DESCRIPTION Lactulose is a
COMMITTEE ON HERBAL MEDICINAL PRODUCTS (HMPC) FINAL COMMUNITY HERBAL MONOGRAPH ON HYPERICUM PERFORATUM L., HERBA (TRADITIONAL USE)
 European Medicines Agency Evaluation of Medicines for Human Use London, 12 November 2009 Doc. Ref.: EMEA/HMPC/745582/2009 COMMITTEE ON HERBAL MEDICINAL PRODUCTS (HMPC) FINAL COMMUNITY HERBAL MONOGRAPH
European Medicines Agency Evaluation of Medicines for Human Use London, 12 November 2009 Doc. Ref.: EMEA/HMPC/745582/2009 COMMITTEE ON HERBAL MEDICINAL PRODUCTS (HMPC) FINAL COMMUNITY HERBAL MONOGRAPH
SODIUM CHLORIDE 0.9% W/V SOLUTION FOR INJECTION PL 01502/0068 UKPAR TABLE OF CONTENTS
 SODIUM CHLORIDE 0.9% W/V SOLUTION FOR INJECTION PL 01502/0068 UKPAR TABLE OF CONTENTS Lay Summary Page 2 Scientific discussion Page 3 Steps taken for assessment Page 10 Steps taken after authorisation
SODIUM CHLORIDE 0.9% W/V SOLUTION FOR INJECTION PL 01502/0068 UKPAR TABLE OF CONTENTS Lay Summary Page 2 Scientific discussion Page 3 Steps taken for assessment Page 10 Steps taken after authorisation
Vitamin D Deficiency and Thyroid Disease. Theodore C. Friedman, M.D., Ph.D.
 Vitamin D Deficiency and Thyroid Disease Theodore C. Friedman, M.D., Ph.D. Vitamin D deficiency and thyroid diseases Vitamin D is an important vitamin that not only regulates calcium, but also has many
Vitamin D Deficiency and Thyroid Disease Theodore C. Friedman, M.D., Ph.D. Vitamin D deficiency and thyroid diseases Vitamin D is an important vitamin that not only regulates calcium, but also has many
PACKAGE LEAFLET: INFORMATION FOR THE USER. PARACETAMOL MACOPHARMA 10 mg/ml, solution for infusion. Paracetamol
 PACKAGE LEAFLET: INFORMATION FOR THE USER PARACETAMOL MACOPHARMA 10 mg/ml, solution for infusion Paracetamol Read all of this leaflet carefully before you start using this medicine. Keep this leaflet.
PACKAGE LEAFLET: INFORMATION FOR THE USER PARACETAMOL MACOPHARMA 10 mg/ml, solution for infusion Paracetamol Read all of this leaflet carefully before you start using this medicine. Keep this leaflet.
Teriflunomide is the active metabolite of Leflunomide, a drug employed since 1994 for the treatment of rheumatoid arthritis (Baselt, 2011).
 Page 1 of 10 ANALYTE NAME AND STRUCTURE TERIFLUNOMIDE Teriflunomide TRADE NAME Aubagio CATEGORY Antimetabolite TEST CODE PURPOSE Therapeutic Drug Monitoring GENERAL RELEVANCY BACKGROUND sclerosis. The
Page 1 of 10 ANALYTE NAME AND STRUCTURE TERIFLUNOMIDE Teriflunomide TRADE NAME Aubagio CATEGORY Antimetabolite TEST CODE PURPOSE Therapeutic Drug Monitoring GENERAL RELEVANCY BACKGROUND sclerosis. The
White, circular, biconvex, uncoated tablets with a score line on one side, plain on the other.
 Nausicalm Cyclizine hydrochloride Ph. Eur. 50 mg Presentation White, circular, biconvex, uncoated tablets with a score line on one side, plain on the other. Uses Actions The active ingredient-cyclizine
Nausicalm Cyclizine hydrochloride Ph. Eur. 50 mg Presentation White, circular, biconvex, uncoated tablets with a score line on one side, plain on the other. Uses Actions The active ingredient-cyclizine
PACKAGE LEAFLET VITAMIN C
 PACKAGE LEAFLET VITAMIN C ATC code: A11GA01 PHARMACOTHERAPEUTIC GROUP Vitamins. COMPOSITION Ascorbic acid 100 mg or 500 mg in 1 tablet; 600 mg or 1000 mg in 1 effervescent tablet. ACTION As a redoxy-compound,
PACKAGE LEAFLET VITAMIN C ATC code: A11GA01 PHARMACOTHERAPEUTIC GROUP Vitamins. COMPOSITION Ascorbic acid 100 mg or 500 mg in 1 tablet; 600 mg or 1000 mg in 1 effervescent tablet. ACTION As a redoxy-compound,
3% Sodium Chloride Injection, USP 5% Sodium Chloride Injection, USP
 PRESCRIBING INFORMATION 3% Sodium Chloride Injection, USP 5% Sodium Chloride Injection, USP IV Fluid and Electrolyte Replenisher Baxter Corporation Mississauga, Ontario L5N 0C2 Canada Date of Revision:
PRESCRIBING INFORMATION 3% Sodium Chloride Injection, USP 5% Sodium Chloride Injection, USP IV Fluid and Electrolyte Replenisher Baxter Corporation Mississauga, Ontario L5N 0C2 Canada Date of Revision:
SUMMARY OF PRODUCT CHARACTERISTICS. Albuman 200 g/l is a solution containing 200 g/l (20%) of total protein of which at least 95% is human albumin.
 Albuman 200 g/l SPC 01 December 2015 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Albuman 200 g/l solution for infusion 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Albuman 200 g/l
Albuman 200 g/l SPC 01 December 2015 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Albuman 200 g/l solution for infusion 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Albuman 200 g/l
PRESCRIBING INFORMATION K-10. (Potassium Chloride Oral Solution, 10% USP) POTASSIUM REPLACEMENT THERAPY
 PRESCRIBING INFORMATION K-10 (Potassium Chloride Oral Solution, 10% USP) POTASSIUM REPLACEMENT THERAPY GlaxoSmithKline Inc Date of Revision: 7333 Mississauga Road North April 14, 2015 Mississauga, OntarioL5N
PRESCRIBING INFORMATION K-10 (Potassium Chloride Oral Solution, 10% USP) POTASSIUM REPLACEMENT THERAPY GlaxoSmithKline Inc Date of Revision: 7333 Mississauga Road North April 14, 2015 Mississauga, OntarioL5N
0.9% Sodium Chloride Injection, USP In VIAFLEX Plastic Container
 Page 1 of 8 PRESCRIBING INFORMATION 0.9% Sodium Chloride Injection, USP In VIAFLEX Plastic Container IV Fluid and Electrolyte Replenisher Baxter Corporation Mississauga, Ontario L5N 0C2 Canada Date of
Page 1 of 8 PRESCRIBING INFORMATION 0.9% Sodium Chloride Injection, USP In VIAFLEX Plastic Container IV Fluid and Electrolyte Replenisher Baxter Corporation Mississauga, Ontario L5N 0C2 Canada Date of
Maintenance of abstinence in alcohol dependence
 Shared Care Guideline for Prescription and monitoring of Acamprosate Calcium Author(s)/Originator(s): (please state author name and department) Dr Daly - Consultant Psychiatrist, Alcohol Services Dr Donnelly
Shared Care Guideline for Prescription and monitoring of Acamprosate Calcium Author(s)/Originator(s): (please state author name and department) Dr Daly - Consultant Psychiatrist, Alcohol Services Dr Donnelly
Interpretation of Laboratory Values
 Interpretation of Laboratory Values Konrad J. Dias PT, DPT, CCS Overview Electrolyte imbalances Renal Function Tests Complete Blood Count Coagulation Profile Fluid imbalance Sodium Electrolyte Imbalances
Interpretation of Laboratory Values Konrad J. Dias PT, DPT, CCS Overview Electrolyte imbalances Renal Function Tests Complete Blood Count Coagulation Profile Fluid imbalance Sodium Electrolyte Imbalances
Kidney Disease WHAT IS KIDNEY DISEASE? TESTS TO DETECT OR DIAGNOSE KIDNEY DISEASE TREATMENT STRATEGIES FOR KIDNEY DISEASE
 Kidney Disease WHAT IS KIDNEY DISEASE? Kidney disease is when your kidneys are damaged and not functioning as they should. When kidney disease is not going away it is called chronic kidney disease or CKD.
Kidney Disease WHAT IS KIDNEY DISEASE? Kidney disease is when your kidneys are damaged and not functioning as they should. When kidney disease is not going away it is called chronic kidney disease or CKD.
Acid-Base Balance and the Anion Gap
 Acid-Base Balance and the Anion Gap 1. The body strives for electrical neutrality. a. Cations = Anions b. One of the cations is very special, H +, and its concentration is monitored and regulated very
Acid-Base Balance and the Anion Gap 1. The body strives for electrical neutrality. a. Cations = Anions b. One of the cations is very special, H +, and its concentration is monitored and regulated very
CODEINE PHOSPHATE 15 MG TABLETS CODEINE PHOSPHATE 30 MG TABLETS CODEINE PHOSPHATE 60 MG TABLETS PL 24837/0023-5 UK PAR TABLE OF CONTENTS
 CODEINE PHOSPHATE 15 MG TABLETS CODEINE PHOSPHATE 30 MG TABLETS CODEINE PHOSPHATE 60 MG TABLETS PL 24837/0023-5 UK PAR TABLE OF CONTENTS Lay Summary Page 2 Scientific discussion Page 3 Steps taken for
CODEINE PHOSPHATE 15 MG TABLETS CODEINE PHOSPHATE 30 MG TABLETS CODEINE PHOSPHATE 60 MG TABLETS PL 24837/0023-5 UK PAR TABLE OF CONTENTS Lay Summary Page 2 Scientific discussion Page 3 Steps taken for
Glycopyrronium Bromide 0.5mg/mL and Neostigmine Metilsulfate 2.5mg/mL Solution for Injection
 NEW ZEALAND DATA SHEET Glycopyrronium Bromide 0.5mg/mL and Neostigmine Metilsulfate 2.5mg/mL Solution for Injection Each 1mL contains Glycopyrrolate USP 0.5mg (Glycopyrronium Bromide) and Neostigmine Metilsulfate
NEW ZEALAND DATA SHEET Glycopyrronium Bromide 0.5mg/mL and Neostigmine Metilsulfate 2.5mg/mL Solution for Injection Each 1mL contains Glycopyrrolate USP 0.5mg (Glycopyrronium Bromide) and Neostigmine Metilsulfate
Perfalgan 10 mg/ml, solution for infusion
 PACKAGE LEAFLET: INFORMATION FOR THE USER Perfalgan 10 mg/ml, solution for infusion Paracetamol Read all of this leaflet carefully before you start using this medicine. Keep this leaflet. You may need
PACKAGE LEAFLET: INFORMATION FOR THE USER Perfalgan 10 mg/ml, solution for infusion Paracetamol Read all of this leaflet carefully before you start using this medicine. Keep this leaflet. You may need
1. NAME OF THE MEDICINAL PRODUCT. Impavido 10 mg capsules Impavido 50 mg capsules 2. QUALITATIVE AND QUANTITATIVE COMPOSITION. 1 capsule contains:
 1. NAME OF THE MEDICINAL PRODUCT Impavido 10 mg capsules Impavido 50 mg capsules 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 1 capsule contains: Impavido 10 mg capsules 10 mg Miltefosine. Impavido 50 mg
1. NAME OF THE MEDICINAL PRODUCT Impavido 10 mg capsules Impavido 50 mg capsules 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 1 capsule contains: Impavido 10 mg capsules 10 mg Miltefosine. Impavido 50 mg
NEW ZEALAND DATA SHEET NAPHCON-A Naphazoline hydrochloride and pheniramine maleate.
 NEW ZEALAND DATA SHEET NAPHCON-A Naphazoline hydrochloride and pheniramine maleate. PRESENTATION Eye Drops: NAPHCON-A Eye Drops are a combination of an antihistamine (pheniramine maleate) and a decongestant
NEW ZEALAND DATA SHEET NAPHCON-A Naphazoline hydrochloride and pheniramine maleate. PRESENTATION Eye Drops: NAPHCON-A Eye Drops are a combination of an antihistamine (pheniramine maleate) and a decongestant
OSTEOPOROSIS REHABILITATION PROGRAM
 OSTEOPOROSIS REHABILITATION PROGRAM Tricia Orme, R.N. BSc(N) Mary Pack Arthritis Program Victoria i Arthritis i Centre Objectives Participants will gain an understanding of what Osteoporosis is and how
OSTEOPOROSIS REHABILITATION PROGRAM Tricia Orme, R.N. BSc(N) Mary Pack Arthritis Program Victoria i Arthritis i Centre Objectives Participants will gain an understanding of what Osteoporosis is and how
Phosphate (serum, plasma, urine)
 Phosphate (serum, plasma, urine) 1 Name and description of analyte 1.1 Name of analyte Phosphate 1.2 Alternative names Phosphorus, inorganic phosphorus (Pi), PO 4. 1.3 NMLC number 1.4 Description of analyte
Phosphate (serum, plasma, urine) 1 Name and description of analyte 1.1 Name of analyte Phosphate 1.2 Alternative names Phosphorus, inorganic phosphorus (Pi), PO 4. 1.3 NMLC number 1.4 Description of analyte
ACID- BASE and ELECTROLYTE BALANCE. MGHS School of EMT-Paramedic Program 2011
 ACID- BASE and ELECTROLYTE BALANCE MGHS School of EMT-Paramedic Program 2011 ACID- BASE BALANCE Ions balance themselves like a see-saw. Solutions turn into acids when concentration of hydrogen ions rises
ACID- BASE and ELECTROLYTE BALANCE MGHS School of EMT-Paramedic Program 2011 ACID- BASE BALANCE Ions balance themselves like a see-saw. Solutions turn into acids when concentration of hydrogen ions rises
EMEA PUBLIC STATEMENT ON LEFLUNOMIDE (ARAVA) - SEVERE AND SERIOUS HEPATIC REACTIONS -
 The European Agency for the Evaluation of Medicinal Products Post-authorisation evaluation of medicines for human use London, 12 March 2001 Doc. Ref: EMEA/H/5611/01/en EMEA PUBLIC STATEMENT ON LEFLUNOMIDE
The European Agency for the Evaluation of Medicinal Products Post-authorisation evaluation of medicines for human use London, 12 March 2001 Doc. Ref: EMEA/H/5611/01/en EMEA PUBLIC STATEMENT ON LEFLUNOMIDE
Calcium and Vitamin D: Important at Every Age
 Calcium and Vitamin D: Important at Every Age National Institutes of Health Osteoporosis and Related Bone Diseases ~ National Resource Center 2 AMS Circle Bethesda, MD 20892-3676 Tel: (800) 624-BONE or
Calcium and Vitamin D: Important at Every Age National Institutes of Health Osteoporosis and Related Bone Diseases ~ National Resource Center 2 AMS Circle Bethesda, MD 20892-3676 Tel: (800) 624-BONE or
SUMMARY OF PRODUCT CHARACTERISTICS. 100 mg
 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Neurobion ampoules 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 3 ml of aqueous solution (1 ampoule) contains: Thiamine chloride hydrochloride
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Neurobion ampoules 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 3 ml of aqueous solution (1 ampoule) contains: Thiamine chloride hydrochloride
Calcium. 1995-2013, The Patient Education Institute, Inc. www.x-plain.com nuf40101 Last reviewed: 02/19/2013 1
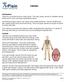 Calcium Introduction Calcium is a mineral found in many foods. The body needs calcium to maintain strong bones and to carry out many important functions. Not having enough calcium can cause many health
Calcium Introduction Calcium is a mineral found in many foods. The body needs calcium to maintain strong bones and to carry out many important functions. Not having enough calcium can cause many health
Vitamin D and Calcium Guideline
 Vitamin D and Calcium Guideline Vitamin D Functions of Vitamin D 2 Serum Levels 2 Deficiency 2 Toxicity 2 Dietary Reference Intakes 3 Sources of Vitamin D 3 Therapeutic Options 4 Adults 4 Children (0 18
Vitamin D and Calcium Guideline Vitamin D Functions of Vitamin D 2 Serum Levels 2 Deficiency 2 Toxicity 2 Dietary Reference Intakes 3 Sources of Vitamin D 3 Therapeutic Options 4 Adults 4 Children (0 18
Summary of Product Characteristics
 1 NAME OF THE MEDICINAL PRODUCT Diamox SR 250mg Modified Release Hard Capsules. Summary of Product Characteristics 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each capsule contains Acetazolamide 250 mg.
1 NAME OF THE MEDICINAL PRODUCT Diamox SR 250mg Modified Release Hard Capsules. Summary of Product Characteristics 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each capsule contains Acetazolamide 250 mg.
Open the Flood Gates Urinary Obstruction and Kidney Stones. Dr. Jeffrey Rosenberg Dr. Emilio Lastarria Dr. Richard Kasulke
 Open the Flood Gates Urinary Obstruction and Kidney Stones Dr. Jeffrey Rosenberg Dr. Emilio Lastarria Dr. Richard Kasulke Nephrology vs. Urology Nephrologist a physician who has been trained in the diagnosis
Open the Flood Gates Urinary Obstruction and Kidney Stones Dr. Jeffrey Rosenberg Dr. Emilio Lastarria Dr. Richard Kasulke Nephrology vs. Urology Nephrologist a physician who has been trained in the diagnosis
YOUR TRUSTED SOURCE - FOR ALL YOUR VITAMIN D 3 NEEDS
 Vitamin D YOUR TRUSTED SOURCE - FOR ALL YOUR VITAMIN D 3 NEEDS ü Natural form of vitamin D 3, cholecalciferol (from lanolin) ü Strengths from 400 iu to 10,000 iu ü Capsule and liquid forms available ü
Vitamin D YOUR TRUSTED SOURCE - FOR ALL YOUR VITAMIN D 3 NEEDS ü Natural form of vitamin D 3, cholecalciferol (from lanolin) ü Strengths from 400 iu to 10,000 iu ü Capsule and liquid forms available ü
ISOPLEX 4% W/V SOLUTION FOR INFUSION (Succinylated gelatin) PL 13538/0017 UKPAR TABLE OF CONTENTS
 ISOPLEX 4% W/V SOLUTION FOR INFUSION (Succinylated gelatin) PL 13538/0017 UKPAR TABLE OF CONTENTS Lay Summary Page 2 Scientific discussion Page 3 Steps taken for assessment Page 11 Steps taken after authorisation
ISOPLEX 4% W/V SOLUTION FOR INFUSION (Succinylated gelatin) PL 13538/0017 UKPAR TABLE OF CONTENTS Lay Summary Page 2 Scientific discussion Page 3 Steps taken for assessment Page 11 Steps taken after authorisation
PACKAGE LEAFLET: INFORMATION FOR THE USER Omeprazol XXX 40 mg powder for solution for infusion omeprazole
 PACKAGE LEAFLET: INFORMATION FOR THE USER Omeprazol XXX 40 mg powder for solution for infusion omeprazole Read all of this leaflet carefully before you start using this medicine. - Keep this leaflet. You
PACKAGE LEAFLET: INFORMATION FOR THE USER Omeprazol XXX 40 mg powder for solution for infusion omeprazole Read all of this leaflet carefully before you start using this medicine. - Keep this leaflet. You
Vitamin D (serum, plasma)
 Vitamin D (serum, plasma) 1 Name and description of analyte 1.1 Name of analyte Vitamin D 1.2 Alternative names The term vitamin D covers a group of closely related naturally occurring lipid soluble compounds
Vitamin D (serum, plasma) 1 Name and description of analyte 1.1 Name of analyte Vitamin D 1.2 Alternative names The term vitamin D covers a group of closely related naturally occurring lipid soluble compounds
Hypromellose Eye Drops BP 0.3% w/v PL 23097/0006
 Hypromellose Eye Drops BP 0.3% w/v PL 23097/0006 UKPAR TABLE OF CONTENTS Lay summary Page 2 Scientific discussion Page 3 Steps taken for assessment Page 9 Summary of product characteristics Page 10 Patient
Hypromellose Eye Drops BP 0.3% w/v PL 23097/0006 UKPAR TABLE OF CONTENTS Lay summary Page 2 Scientific discussion Page 3 Steps taken for assessment Page 9 Summary of product characteristics Page 10 Patient
OMNIC OCAS Film coated Tablets SAJA PHARMA
 08-15 OMNIC OCAS Film coated Tablets SAJA PHARMA Tamsulosin Hydrochloride 0.4 mg film-coated prolonged release tablets 1. NAME OF THE MEDICINAL PRODUCT OMNIC OCAS, 0.4 mg prolonged release tablets, film-coated.
08-15 OMNIC OCAS Film coated Tablets SAJA PHARMA Tamsulosin Hydrochloride 0.4 mg film-coated prolonged release tablets 1. NAME OF THE MEDICINAL PRODUCT OMNIC OCAS, 0.4 mg prolonged release tablets, film-coated.
