Standard Operating Procedure (SOP)
|
|
|
- Lee Bennett
- 9 years ago
- Views:
Transcription
1 Department of Biology, Chemistry, Pharmacy Institute for Pharmacy (Pharmacology and Toxicology) Königin-Luise-Str Berlin Germany Standard Operating Procedure (SOP) Title Isolation of Keratinocytes and Fibroblasts from Human Specimens Documentenno. First edition Zoschke, C.; Gonska, H. Version Page 1 von 17 Version valid from Description of changes Erstellung Selbst hergestellte Medien, mikrobielle Kontamination Trypsin-EDTA-Konzentration, Zählung Translation Trypsin-EDTA-working and -stock solutions Zoschke, C.; Gonska, H. Kral, V.; Kapfer, C. Scope Scientific projects C02 (Schäfer-Korting, Hedtrich) and Z01 (Kleuser, Ma, Schäfer-Korting) of the CRC 1112; scientific projects Development of human-based skin disease models (Schäfer- Korting) and Integration of Langerhans cells into reconstructed human skin (Weindl) of BB3R.
2 2 Directory 1 Aims Scope Equipment... 4 Consumables... Custom-made media Step-to-step protocol... 8 Prepare specimen... 8 Cell isolation... 9 Cell culture Cell count Change cell culture media Cell subculture Cell freezing Cell thawing Cell transport Cell morphology... 1 Specimen documentation Cell documentation Trouble shooting... 17
3 3 AIMS Standardized cell isolation from human skin specimens with maximal cell yield. SCOPE This SOP applies to the scientific projects C02 (Schäfer-Korting, Hedtrich) and Z01 (Kleuser, Ma, Schäfer-Korting) of the collaborative research center 1112 as well as to the scientific projects Development of human-based skin disease models (Schäfer-Korting) and Integration of Langerhans cells into reconstructed human skin (Weindl) of the Berlin-Brandenburg research platform BB3R with integrated graduate education.
4 4 MATERIALS Equipment Designation Autoklave Centrifuge (Eppendorf) Centrifuge (Megafuge 1.0R) Cryotome (Leica CM 110S) Drying cabinet Fluorescence microscope (BZ-8000K) Freezer (-20 C) Freezer (-80 C) Freezing container (Nalgene, Mr. Frosty) Neubauer counting chamber Incubator (BB6220) Magnetic stirrer RCT basic Microtome (Hyrax M40) Nitrogen tank (Arpege 70) Paraffinization station (Microm EC 30) Phase contrast inverted microscope (Axiovert 40C) ph meter (766 Calimatic) Pipette (Eppendorf Reference ) Pipetting aid (Easypet ) Refrigerator (4 C) Sterile working bench (LaminAir ) Water bath Water processing unit (SG LaboStar) Manufacturer Systec, Wettenberg Eppendorf, Hamburg Thermo Fisher Scientific, Waltham, MA, USA Leica Microsystems, Wetzlar Memmert, Schwabach Keyence, Osaka, JAP Siemens, München Thermo Fisher Scientific, Waltham, MA, USA Thermo Fisher Scientific, Waltham, MA, USA Zeiss, Jena Thermo Fisher Scientific, Waltham, MA, USA IKA-Werke, Staufen Zeiss, Jena Air Liquide, Paris, F Zeiss, Jena Zeiss, Jena Knick, Nürnberg Eppendorf, Hamburg Eppendorf, Hamburg Siemens, München Thermo Fisher Scientific, Waltham, MA, USA Gesellschaft für Labortechnik, Burgwedel SG Wasseraufberitung und Regenerierstation, Barsbüttel Equivalent equipment of other suppliers can be used as well.
5 Consumables Designation 2-Propanol Calibration solutions (ph 7 / ph 9) Cell culture flask (7 cm²) Cell strainer (70 µm pore size) Centrifuge tubes (0 ml) Cryo vial Dimethyl sulfoxide (plant cell culture tested) Disodium hydrogenphosphate (pro analysi) Dispase II (Cat. No ) Dulbecco's Modified Eagle Medium (DMEM) Ethylenediaminetetraacetic acid (EDTA) Fetal calf serum Forceps (anatomical) Keratinocytes Growth Medium (KGM) L-Glutamine Nitrogen (liquid) Parafilm (Nescofilm ) Penicillin-Streptomycin-solution (100x, U/mL Penicillin, 10 mg/ml Streptomycin) Pipette tips Pipettes ( ml, 10 ml, 2 ml) Potassium chloride (KCl, cell culture tested) Potassium dihydrogen orthophosphate (KH 2 PO 4 ) Supplier VWR, Darmstadt Hanna Instruments, Woonsocket, RI, USA TPP, Trasadingen, Schweiz BD, Franklin Lakes, NJ, USA TPP, Trasadingen, Schweiz Almeco, Esbjerg N, DK Sigma-Aldrich, München Merck, Darmstadt Roche Diagnostics, Mannheim Sigma-Aldrich, München Sigma-Aldrich, Schnelldorf Biochrom, Berlin Carl Roth, Karlsruhe Lonza, Köln Sigma-Aldrich, München Air Liquide, Paris, F Carl Roth, Karlsruhe Sigma-Aldrich, München Eppendorf, Hamburg Sarstedt, Nümbrecht Sigma-Aldrich, München Carl Roth, Karlsruhe
6 6 Consumables (continued) Designation Scalpel (No. 24) Sodium chloride (NaCl, pro analysi) Stirring bar Syringe (10 ml, 20 ml) Syringe filter (Cellulose acetate, 0.2 µm pore size, 1 ) Syringe filter (Reconstituted cellulose, 0.2 µm pore size, 1 ) Tissue culture dish Tissue culture test plate (6-well) Transport vial for preputial specimen Trypsin Supplier Carl Roth, Karlsruhe Carl Roth, Karlsruhe Carl Roth, Karlsruhe BBraun, Melsungen Sarstedt, Nümbrecht Carl Roth, Karlsruhe TPP, Trasadingen, Schweiz TPP, Trasadingen, Schweiz Sarstedt, Nümbrecht Sigma-Aldrich, München Equivalent consumables of other suppliers can be used as well. 1 ) Filtrate is sterile according to United States Pharmacopeia (USP)
7 7 Custom-made media Description Ingredients Remarks Dispase stock Dispase 10 mg c(dispase)=10 U/mL solution PBS 1 ml -20 C, 6 weeks 2 ) Dispase working Dispase stock solution 300 µl c(dispase)=1.2 U/mL solution PBS 2. ml do not store Freezing FGM 4 ml protect from light (tin foil) medium Dimethyl sulfoxide ml 4 C, 6 weeks 2,3 ) FGM DMEM 00 ml Fetal Calf Serum L-Glutamine 0 ml ml Penicillin/Streptomycin ml 4 C, 6 weeks 2 ) PBS KCl 200 mg NaCl KH 2 PO 4 8,000 mg 200 mg Na 2 HPO 4 1,148 mg autoclave, check ph 4 ) Aq. bidest. 1 L 4 C, 6 weeks 2 ) Trypsin-EDTA- Trypsin 2,00 mg c(trypsin)=139 mg/ml stock solution EDTA mg sterile filtration, PBS 18 ml Aliquot: -20 C, 6 weeks 2 ) Trypsin-EDTA- Trypsin-EDTA stock solution 1.8 ml c(trypsin)=2. mg/ml = 0.2% c(edta)=0.3 mmol/l working solution PBS 99 ml 4 C, 2 weeks 2 ) 2 ) maximal duration of storage when media are stored at 2-8 C, media need to be warmed to room temperature before use (except freezing medium) 3 ) use reconstituted cellulose filters for the filtration of DMSO 4 ) perform calibration of ph meter before use (ph 7, ph 9, stirring bar)
8 8 STEP-TO-STEP PROTOCOL Prepare specimen Day 01 Dispase working solution, forceps, Parafilm, PBS, scalpel, tissue culture dish Step-to-step protocol Prepare specimens separately, one after each other Evaluate specimen and discard, if - specimen transport took longer than 48 h or - cooling chain was interrupted or - donor age exceeds 10 years (only applicable for preputial specimen) or - specimen shows pus, tumor, or necrosis Wash specimen (3- times) in transport vial with 10 ml PBS until the specimen is cleared from blood Transfer specimen in tissue culture dish Remove mucosal, vascular, and adipose tissue from preputial specimen using a scalpel Wash specimen (1- times) until PBS is clear Subdivide specimen into 3 mm wide pieces using a scalpel and forceps Place all pieces into another tissue culture dish with the dermis attaching the dish. Pieces should be placed without touching each other (remove any liquid) Pipette Dispase working solution into tissue culture dish, but not onto the pieces of the preputial specimen (2.8 ml per 60 cm² growth surface of the tissue culture dish) Label tissue culture dish with specimen number and time of adding Dispase working solution specimen number: year (yy) und serial number (xx) within the respective year (e.g. 16-1: first specimen in 2016) Seal the tissue culture dish and its lid with Parafilm and incubate the specimen for h at 4 C Specimen documentation Record specimen number, date of receipt, and remarks (e.g. colored skin, female donor, specimen site) in the specimen documentation
9 9 Cell isolation Day 01 Cell culture flask, cell strainer, centrifuge tube, FGM, forceps, KGM, PBS, tissue culture test plate, Trypsin-EDTA working solution, materials for cell count Step-to-step protocol Dilute 4 ml Trypsin-EDTA working solution with 12 ml PBS (in centrifuge tube, do not store diluted Trypsin-EDTA working solution) Fill ml PBS in the lid of the tissue culture dish Separate epidermis and dermis using forceps and collect the epidermal pieces in the lid of the tissue culture dish Collect the epidermal pieces in diluted Trypsin-EDTA working solution in the centrifuge tube; epidermal pieces must not stick to the rim of the centrifuge tube Incubate the epidermal suspension for 20 min in the incubator. Wave the centrifuge tube twice every min to re-suspend the epidermal pieces Place dermal pieces upside down into the tissue culture test plate. Use one plate per donor and place max. 6 dermal pieces into one well Allow the dermal pieces to attach the bottom of the tissue culture test plate for at least 30 min at room temperature After at least 30 min, fill 1. ml FGM per well into the tissue culture test plate using an Eppendorf pipette without detaching the dermal pieces. Incubate the dermal pieces in the incubator Stop enzymatic reaction of Trypsin with 10 ml FGM and pipette the cell suspension three times up and down Filtrate the cell suspension using a cell strainer and wash the cell strainer with ml PBS Centrifuge cell suspension at 130 g for min at 2 C and discard the supernatant Re-suspend NHK pellet in 10 ml PBS Count cells and centrifuge cell suspension again CRITICAL STEPs Discard cells, if viable cell count is below 700,000 NHK or dead cell count exceeds 0% Otherwise use cells and Label cell culture flask with cell type, passage, and specimen number Re-suspend NHK pellet in ml KGM and seed 2 x10 6 cells per cell culture flask (7 cm²) Use 12 ml KGM per cell culture flask (7 cm²), except on Friday (18 ml KGM)
10 10 Cell culture Days Cell culture flask, centrifuge tube, FGM, forceps, KGM, PBS, Trypsin-EDTA working solution, materials for cell count Step-to-step protocol Day Evaluate NHK (Figure 1a, c) adherent cells without microbial contamination change medium on Mo-Tu-Fr until day 08 non-adherent cells or microbial contamination discard cells Day 07 Evaluate NHDF (Figure 1b, c) adherent cells without microbial contamination aspirate FGM, remove dermal pieces using forceps, add 2 ml FGM per well but do not wash cells on day 07, change medium on Mo-Tu-Fr until day 29 non-adherent cells or microbial contamination discard cells Day 08 Evaluate NHK (Figure 1a) slack NHK colonies with cell membranes appearing as bright rims freeze cells (label: p1) differentiated NHK appearing as cobblestones discard cells Day 29 Evaluate NHDF (Figure 1) huge NHDF bundles subculture cells from 6 wells in 3 cell culture flasks (7 cm²), label: p1 small or absent NHDF bundles discard cells Cell count Days 01, 08, 29 Cell suspension in PBS, Neubauer counting chamber Step-to-step protocol Pipette 10 µl cell suspension into Neubauer counting chamber Determine the mean cell count of all four quadrants Calculate the absolute cell count per ml: mean cell count of all four quadrants x 10 4
11 11 Change cell culture media FGM, KGM, pasteur pipettes, PBS Step-to-step protocol Evaluate cell morphology (Figure 1) Aspirate KGM or FGM using pasteur pipettes without touching the cell monolayer Wash cells twice with 10 ml PBS (cell culture flask, 7 cm²) or with 2 ml per well of a tissue culture test plate; do not pipette the liquids directly onto the cell monolayer CRITICAL STEPs Use 12 ml KGM/FGM per cell culture flask (7 cm²), except on Friday (18 ml KGM/FGM) Use 2 ml KGM/FGM per well of a tissue culture test plate. Avoid foaming of media Change KGM three times a week (Monday-Wednesday-Friday) Change FGM twice a week (Monday-Thursday) Cell subculture (passaging) Cell culture flask, centrifuge tube, FGM, KGM, tissue culture test plates, Trypsin-EDTA working solution, PBS, materials for cell count Step-to-step protocol Evaluate cell morphology Aspirate KGM or FGM using pasteur pipettes without touching the cell monolayer Wash cells twice with 10 ml PBS (cell culture flask, 7 cm²) or with 2 ml per well of a tissue culture test plate; do not pipette the liquids directly onto the cell monolayer Incubate cells with 1. ml Trypsin-EDTA working solution per cell culture flask (7 cm²) or 0. ml per well of a tissue culture test plate for min in the incubator; close the cell culture flask gas tight to ensure optimal ph (ph ) for Trypsin activity
12 12 Cell subculture (continued) CRITICAL STEPs To prevent cell damage to NHDF, Trypsin-EDTA working solution can be alternatively spread out to the cell monolayer and aspirated after 1 min using pasteur pipettes; close the cell culture flask gas tight and incubate for min in the incubator Promote cell detachment by gently tapping at the side of the cell culture flask Check cell detachment microscopically (detached cells appear as floating spheres) Let as many cells as possible detach to prevent selection processes, but cells should be exposed not longer than 10 min to the Trypsin-EDTA working solution Inactivate Trypsin-EDTA working solution by adding FGM (FGM volume equals at least threefold of the Trypsin-EDTA working solution volume) Pipette the cell suspension into a centrifuge tube, wash the cell culture flask or the tissue culture plate with 10 ml PBS and transfer the wash fluid into the centrifuge tube Centrifuge the cell suspension at 130 g for min at 2 C and discard the supernatant Re-suspend the cell pellet in 10 ml PBS and count cells Centrifuge the cell suspension again at 130 g for min at 2 C and discard the supernatant Label cell culture flask with cell type, passage, and specimen number Re-suspend cells in ml KGM or FGM, seed 2 x10 6 cells per cell culture flask (7 cm²), and add KGM or FGM Allow the cells to attach to the cell culture flask and avoid agitation for the next 24 h Freeze passage 2 of NHK or NHDF and discard passage 4 or higher
13 13 Cell freezing 2-Propanol, cryo vial, freezing container, freezing medium, materials for cell subculture Step-to-step protocol Change medium 24 h before cell freezing; evaluate morphology directly before cell freezing Fill 20 ml 2-Propanol into the freezing container (change 2-Propanol every freeze-thawcycles) Label cell culture flask with cell type, passage (+1), specimen number, and date of freezing; (optimal cell count: 3x10 6 NHK or 1.x10 6 NHDF per cryo vial) Subculture cells Re-suspend cells in freezing medium, fill every cryo vial with 1.8 ml cell suspension Store freezing container with closed cryo vials for 24 h in freezer (-80 C) and transfer cryo vials to nitrogen tank Specimen and cell documentation Record date of freezing in the specimen documentation Record cell type, passage, specimen number, count, date of freezing in the cell documentation Cell thawing 2-Propanol (70%), cell culture flask, centrifuge tube, FGM, KGM, PBS Step-to-step protocol Take cryo vial from nitrogen tank, vent cryo vials, and thaw them using a water bath (37 C) Wipe cryo vial with 2-Propanol (70%) Pipette cell suspension from cryo vial into centrifuge tube and add up to 20 ml PBS Centrifuge the cell suspension at 130 g for min at 2 C and discard the supernatant Re-suspend the cell pellet in 10 ml PBS and count cells Centrifuge the cell suspension again at 130 g for min at 2 C and discard the supernatant Label cell culture flask with cell type, passage, and specimen number Re-suspend cells in ml KGM or FGM, seed 2 x10 6 cells per cell culture flask (7 cm²), and add KGM or FGM Allow the cells to attach to the cell culture flask and avoid agitation for the next 24 h Cell documentation Delete cells from cell documentation
14 14 Cells transport cell culture flask, centrifuge tube, dry ice, FGM, KGM, PBS Step-to-step protocol send cells Use dry ice to ensure continuous cooling chain for 3 days and order overnight express Inform the recipient prior to cell delivery about cell type, passage, cell count, and culture conditions CRITICAL STEPs Observe country specific quarantine rules and guidance on dangerous goods If required, send cells in cell culture flasks Grow cells until 70% confluence Fill the cell culture flask completely with KGM or FGM Close the cell culture flask gas tight Cell documentation Delete cells from cell documentation Step-to-step protocol receive cells Separate cells from routine cell culture to avoid cross contamination Culture cells according to the information of the sender If cells arrive in cell culture flasks Aspirate cell culture media from cell culture flask, but keep approximately 1 ml culture medium in a cell culture flask (7 cm²) Change medium and evaluate morphology 24 h upon receipt
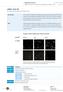
15 1 Cell morphology a) NHK b) NHDF c) Microbial contamination (Gstraunthaler & Lindl, 2013) (Gstraunthaler & Lindl, 2013) Figure 1 Morphology of cutaneous cells. a) polygonal, adherent NHK that grow in colonies. b) bipolar, elongated, adherent NHDF c) circles: round, adherent cells (NHK/NHDF) arrows: bacteria, suspended in medium. Scale bar = 0 µm.
16 16 SPECIMEN DOCUMENTATION Specimen of freezing No. of receipt Remarks NHK NHDF yy-01 yy-02 yy-03 yy-04 yy-0 yy-06 yy-07 yy-08 yy-09 yy-10 CELL DOCUMENTATION Box No Cell type passage Vial count Specimen number Cell count (mio per cryo vial) Remarks of freezing <NHDF p2> <3> <16-01> <1.> < > 2 <NHK p2> <3> <16-01> <3.0> < > 3
17 17 TROUBLE SHOOTING Problem Epidermis cannot be separated from dermis Melanocytes grow within NHK monolayer Microbial contamination NHDF grow within NHK monolayer Cells do not detach Cells proliferate sparsely Solution Incubate for additional 2 h (4 C) or 1 h (2 C) Discard cells, if the proportion of melanocytes exceeds 10% Discard cells; if microbial contamination occurs in monolayers from more than one donor, than take actions to root out the causes Aspirate KGM, using pasteur pipettes Incubate cells with 2 ml Trypsin-EDTA working solution per cell culture flask (7 cm²) until the NHDF detach Observe this process using a microscope Wash cells with 10 ml PBS and discard the PBS Incubate cells with 1. ml Trypsin-EDTA working solution per cell culture flask (7 cm²) for min in the incubator as described in cell subculture Discard cells 30% confluence: subculture cells immediately < 30% confluence: discard cells
Standard Operating Procedure (SOP)
 Department of Biology, Chemistry, Pharmacy Institute for Pharmacy (Pharmacology and Toxicology) Königin-Luise-Str. 2+ 1195 Berlin Germany Standard Operating Procedure (SOP) Title Production of Organotypic
Department of Biology, Chemistry, Pharmacy Institute for Pharmacy (Pharmacology and Toxicology) Königin-Luise-Str. 2+ 1195 Berlin Germany Standard Operating Procedure (SOP) Title Production of Organotypic
HARVESTING AND CRYOPRESERVATION OF HUMAN EMBRYONIC STEM CELLS (hescs)
 HARVESTING AND CRYOPRESERVATION OF HUMAN EMBRYONIC STEM CELLS (hescs) OBJECTIVE: can be cryopreserved in a liquid nitrogen (LN 2 ) freezer for long-term storage. This Standard Operating Procedure (SOP)
HARVESTING AND CRYOPRESERVATION OF HUMAN EMBRYONIC STEM CELLS (hescs) OBJECTIVE: can be cryopreserved in a liquid nitrogen (LN 2 ) freezer for long-term storage. This Standard Operating Procedure (SOP)
Follicle Dermal Papilla Cell
 Follicle Dermal Papilla Cell Instruction Manual Product Size Catalog Number Human Follicle Dermal Papilla Cells (HFDPC) 500,000 cryopreserved cells 500,000 proliferating cells C-12071 C-12072 Product Description
Follicle Dermal Papilla Cell Instruction Manual Product Size Catalog Number Human Follicle Dermal Papilla Cells (HFDPC) 500,000 cryopreserved cells 500,000 proliferating cells C-12071 C-12072 Product Description
Standard Operating Procedure (SOP) Work Package 8. Sample Collection and Storage
 Standard Operating Procedure (SOP) Work Package 8 Sample Collection and Storage May 2008 SOP WP 8 - Sample collection Table of Contents 1 SAMPLE COLLECTION... 3 1.1 Sample collection -summary... 3 1.2
Standard Operating Procedure (SOP) Work Package 8 Sample Collection and Storage May 2008 SOP WP 8 - Sample collection Table of Contents 1 SAMPLE COLLECTION... 3 1.1 Sample collection -summary... 3 1.2
STANDARD OPERATING PROCEDURE
 STANDARD OPERATING PROCEDURE Title: Antibody Production at Strategic Diagnostics Inc. SOP#: M-119 Version #: 1 Date Approved: August 6, 2009 Author: Strategic Diagnostic Inc. Date Modified: 1. PURPOSE
STANDARD OPERATING PROCEDURE Title: Antibody Production at Strategic Diagnostics Inc. SOP#: M-119 Version #: 1 Date Approved: August 6, 2009 Author: Strategic Diagnostic Inc. Date Modified: 1. PURPOSE
Cellartis Protocol Culturing of hes Cells
 Cellartis Protocol Culturing of hes Cells This material was cultured and frozen using Cellartis protocols. WiCell recommends that stem cells should be thawed and established in the conditions in which
Cellartis Protocol Culturing of hes Cells This material was cultured and frozen using Cellartis protocols. WiCell recommends that stem cells should be thawed and established in the conditions in which
Human Adult Mesothelial Cell Manual
 Human Adult Mesothelial Cell Manual INSTRUCTION MANUAL ZBM0025.01 SHIPPING CONDITIONS Human Adult Mesothelial Cells Orders are delivered via Federal Express courier. All US and Canada orders are shipped
Human Adult Mesothelial Cell Manual INSTRUCTION MANUAL ZBM0025.01 SHIPPING CONDITIONS Human Adult Mesothelial Cells Orders are delivered via Federal Express courier. All US and Canada orders are shipped
Mesenchymal Stem Cells
 Mesenchymal Stem Cells Instruction Manual Product Size Catalog Number from Bone Marrow (hmsc-bm) from Umbilical Cord Matrix (hmsc-uc) from Adipose Tissue (hmsc-at) 500,000 cryopreserved cells 500,000 proliferating
Mesenchymal Stem Cells Instruction Manual Product Size Catalog Number from Bone Marrow (hmsc-bm) from Umbilical Cord Matrix (hmsc-uc) from Adipose Tissue (hmsc-at) 500,000 cryopreserved cells 500,000 proliferating
GASTRIC ORGANOID CULTURE PROTOCOL
 GASTRIC ORGANOID CULTURE PROTOCOL THIS PROTOCOL PROVIDES THE PROCEDURE FOR SUBCULTURING NORMAL HUMAN GASTRIC ORGANOIDS WHICH WAS DERIVED FROM THE SUBMERGED METHOD AS DESCRIBED IN BARKER N, ET AL. LGR5+VE
GASTRIC ORGANOID CULTURE PROTOCOL THIS PROTOCOL PROVIDES THE PROCEDURE FOR SUBCULTURING NORMAL HUMAN GASTRIC ORGANOIDS WHICH WAS DERIVED FROM THE SUBMERGED METHOD AS DESCRIBED IN BARKER N, ET AL. LGR5+VE
Islet Viability Assessment by Single Cell Flow Cytometry
 Islet Viability Assessment by Single Cell Flow Cytometry Page 1 of 8 Purpose: To comprehensively assess the viability of the islet cell preparation prior to transplantation. Tissue Samples: A sample containing
Islet Viability Assessment by Single Cell Flow Cytometry Page 1 of 8 Purpose: To comprehensively assess the viability of the islet cell preparation prior to transplantation. Tissue Samples: A sample containing
Agrobacterium tumefaciens-mediated transformation of Colletotrichum graminicola and Colletotrichum sublineolum
 Agrobacterium tumefaciens-mediated transformation of Colletotrichum graminicola and Colletotrichum sublineolum Flowers and Vaillancourt, 2005. Current Genetics 48: 380-388 NOTE added by L. Vaillancourt:
Agrobacterium tumefaciens-mediated transformation of Colletotrichum graminicola and Colletotrichum sublineolum Flowers and Vaillancourt, 2005. Current Genetics 48: 380-388 NOTE added by L. Vaillancourt:
Taqman TCID50 for AAV Vector Infectious Titer Determination
 Page 1 of 8 Purpose: To determine the concentration of infectious particles in an AAV vector sample. This process involves serial dilution of the vector in a TCID50 format and endpoint determination through
Page 1 of 8 Purpose: To determine the concentration of infectious particles in an AAV vector sample. This process involves serial dilution of the vector in a TCID50 format and endpoint determination through
Amaxa 4D-Nucleofector Protocol for Mouse Embryonic Stem [ES] Cells For 4D-Nucleofector X Unit Transfection in suspension
![Amaxa 4D-Nucleofector Protocol for Mouse Embryonic Stem [ES] Cells For 4D-Nucleofector X Unit Transfection in suspension Amaxa 4D-Nucleofector Protocol for Mouse Embryonic Stem [ES] Cells For 4D-Nucleofector X Unit Transfection in suspension](/thumbs/30/14702458.jpg) For 4D-Nucleofector X Unit Transfection in suspension Cells derived from mouse blastocysts; round cells, growing in clumps s 1. This protocol is meant to provide an outline for the handling and the Nucleofection
For 4D-Nucleofector X Unit Transfection in suspension Cells derived from mouse blastocysts; round cells, growing in clumps s 1. This protocol is meant to provide an outline for the handling and the Nucleofection
Plant Genomic DNA Extraction using CTAB
 Plant Genomic DNA Extraction using CTAB Introduction The search for a more efficient means of extracting DNA of both higher quality and yield has lead to the development of a variety of protocols, however
Plant Genomic DNA Extraction using CTAB Introduction The search for a more efficient means of extracting DNA of both higher quality and yield has lead to the development of a variety of protocols, however
Classic Immunoprecipitation
 292PR 01 G-Biosciences 1-800-628-7730 1-314-991-6034 [email protected] A Geno Technology, Inc. (USA) brand name Classic Immunoprecipitation Utilizes Protein A/G Agarose for Antibody Binding (Cat.
292PR 01 G-Biosciences 1-800-628-7730 1-314-991-6034 [email protected] A Geno Technology, Inc. (USA) brand name Classic Immunoprecipitation Utilizes Protein A/G Agarose for Antibody Binding (Cat.
PROTOCOL. Immunocytochemistry (ICC) MATERIALS AND EQUIPMENT REQUIRED
 PROTOCOL Immunocytochemistry (ICC) 1850 Millrace Drive, Suite 3A Eugene, Oregon 97403 11-07 MATERIALS AND EQUIPMENT REQUIRED Materials: MitoSciences primary monoclonal antibody/antibodies Fluorophore-conjugated
PROTOCOL Immunocytochemistry (ICC) 1850 Millrace Drive, Suite 3A Eugene, Oregon 97403 11-07 MATERIALS AND EQUIPMENT REQUIRED Materials: MitoSciences primary monoclonal antibody/antibodies Fluorophore-conjugated
Measuring Cell Viability/Cytotoxicity: Cell Counting Kit-F
 Introduction The Cell Counting Kit-F is a fluorometic assay for the determination of viable cell numbers. Calcein-AM in this kit passes through the cell membrane and is hydrolized by the esterase in the
Introduction The Cell Counting Kit-F is a fluorometic assay for the determination of viable cell numbers. Calcein-AM in this kit passes through the cell membrane and is hydrolized by the esterase in the
Procedure for RNA isolation from human muscle or fat
 Procedure for RNA isolation from human muscle or fat Reagents, all Rnase free: 20% SDS DEPC-H2O Rnase ZAP 75% EtOH Trizol Chloroform Isopropanol 0.8M NaCitrate/1.2M NaCl TE buffer, ph 7.0 1. Homogenizer-probe
Procedure for RNA isolation from human muscle or fat Reagents, all Rnase free: 20% SDS DEPC-H2O Rnase ZAP 75% EtOH Trizol Chloroform Isopropanol 0.8M NaCitrate/1.2M NaCl TE buffer, ph 7.0 1. Homogenizer-probe
Human Peripheral Blood Mononuclear Cell (PBMC) Manual
 Human Peripheral Blood Mononuclear Cell (PBMC) Manual INSTRUCTION MANUAL ZBM0063.04 SHIPPING CONDITIONS Human Peripheral Blood Mononuclear Cells, cryopreserved Cryopreserved human peripheral blood mononuclear
Human Peripheral Blood Mononuclear Cell (PBMC) Manual INSTRUCTION MANUAL ZBM0063.04 SHIPPING CONDITIONS Human Peripheral Blood Mononuclear Cells, cryopreserved Cryopreserved human peripheral blood mononuclear
NCL Method ITA-6. Leukocyte Proliferation Assay
 NCL Method ITA-6 Leukocyte Proliferation Assay Nanotechnology Characterization Laboratory National Cancer Institute-Frederick SAIC-Frederick Frederick, MD 21702 (301) 846-6939 [email protected] December
NCL Method ITA-6 Leukocyte Proliferation Assay Nanotechnology Characterization Laboratory National Cancer Institute-Frederick SAIC-Frederick Frederick, MD 21702 (301) 846-6939 [email protected] December
Related topics: Application Note 27 Data Analysis of Tube Formation Assays.
 Tube Formation Assays in µ-slide Angiogenesis Related topics: Application Note 27 Data Analysis of Tube Formation Assays. Contents 1. General Information... 1 2. Material... 2 3. Work Flow Overview...
Tube Formation Assays in µ-slide Angiogenesis Related topics: Application Note 27 Data Analysis of Tube Formation Assays. Contents 1. General Information... 1 2. Material... 2 3. Work Flow Overview...
Poietics human mesenchymal stem cells Instructions for use
 Poietics human mesenchymal stem cells Instructions for use www.lonza.com U.S. Scientific Support: 800-521-0390 [email protected] EU/ROW Scientific Support: +49-221-99199-400 [email protected]
Poietics human mesenchymal stem cells Instructions for use www.lonza.com U.S. Scientific Support: 800-521-0390 [email protected] EU/ROW Scientific Support: +49-221-99199-400 [email protected]
Human Umbilical Cord Blood CD34 + Progenitor Cell Care Manual
 Human Umbilical Cord Blood CD34 + Progenitor Cell Care Manual INSTRUCTION MANUAL ZBM0065.03 SHIPPING CONDITIONS Human Umbilical Cord Blood CD34+ Progenitor Cells, cryopreserved Cryopreserved human umbilical
Human Umbilical Cord Blood CD34 + Progenitor Cell Care Manual INSTRUCTION MANUAL ZBM0065.03 SHIPPING CONDITIONS Human Umbilical Cord Blood CD34+ Progenitor Cells, cryopreserved Cryopreserved human umbilical
Required Materials. Stemgent Reprogramming-Qualified NuFF Cells GSC-3002G 4 x 10 6 cells/vial Liquid Nitrogen
 Page 1 Overview This protocol for microrna-enhanced mrna reprogramming describes the combined use of the Stemgent mrna Reprogramming Kit, the Stemgent microrna Booster Kit, and the Stemgent Stemfect RNA
Page 1 Overview This protocol for microrna-enhanced mrna reprogramming describes the combined use of the Stemgent mrna Reprogramming Kit, the Stemgent microrna Booster Kit, and the Stemgent Stemfect RNA
Blood Plasma Sample Collection and Handling for Proteomics Analysis - A guide to obtain optimal plasma samples -
 Blood Plasma Sample Collection and Handling for Proteomics Analysis - A guide to obtain optimal plasma samples - 1 Introduction Blood can be regarded as a complex liquid tissue that comprises cells and
Blood Plasma Sample Collection and Handling for Proteomics Analysis - A guide to obtain optimal plasma samples - 1 Introduction Blood can be regarded as a complex liquid tissue that comprises cells and
Xpert TM Animal Tissue Culture Teaching Kit
 Xpert TM Animal Tissue Culture Teaching Kit Product Code: CCK005 Contents 1. About the kit 2. Kit contents and storage instructions 3. Materials required but not provided in the kit 4. Aseptic techniques
Xpert TM Animal Tissue Culture Teaching Kit Product Code: CCK005 Contents 1. About the kit 2. Kit contents and storage instructions 3. Materials required but not provided in the kit 4. Aseptic techniques
BD PuraMatrix Peptide Hydrogel
 BD PuraMatrix Peptide Hydrogel Catalog No. 354250 Guidelines for Use FOR RESEARCH USE ONLY NOT FOR CLINICAL, DIAGNOSTIC OR THERAPEUTIC USE SPC-354250-G rev 2.0 1 TABLE OF CONTENTS Intended Use... 2 Materials
BD PuraMatrix Peptide Hydrogel Catalog No. 354250 Guidelines for Use FOR RESEARCH USE ONLY NOT FOR CLINICAL, DIAGNOSTIC OR THERAPEUTIC USE SPC-354250-G rev 2.0 1 TABLE OF CONTENTS Intended Use... 2 Materials
NimbleGen DNA Methylation Microarrays and Services
 NimbleGen DNA Methylation Microarrays and Services Sample Preparation Instructions Outline This protocol describes the process for preparing samples for NimbleGen DNA Methylation microarrays using the
NimbleGen DNA Methylation Microarrays and Services Sample Preparation Instructions Outline This protocol describes the process for preparing samples for NimbleGen DNA Methylation microarrays using the
Human CD4+T Cell Care Manual
 Human CD4+T Cell Care Manual INSTRUCTION MANUAL ZBM0067.02 SHIPPING CONDITIONS Human CD4+T Cells, cryopreserved Cryopreserved human CD4+T cells are shipped on dry ice and should be stored in liquid nitrogen
Human CD4+T Cell Care Manual INSTRUCTION MANUAL ZBM0067.02 SHIPPING CONDITIONS Human CD4+T Cells, cryopreserved Cryopreserved human CD4+T cells are shipped on dry ice and should be stored in liquid nitrogen
Day -1 RETRIEVAL OF OVARIES AND OOCYTE COLLECTION AND MATURATION
 Day -1 RETRIEVAL OF OVARIES AND OOCYTE COLLECTION AND MATURATION OVARY COLLECTION Materials and Equipment Needed Thermos with 2 containers with 0.5 L of transport saline in each. Appropriate attire as
Day -1 RETRIEVAL OF OVARIES AND OOCYTE COLLECTION AND MATURATION OVARY COLLECTION Materials and Equipment Needed Thermos with 2 containers with 0.5 L of transport saline in each. Appropriate attire as
Western Blot Protocol (updated on 05/20/14)
 Western Blot Protocol (updated on 05/20/14) Required Solutions 10x PBS (1L) 80 g NaCl 2 g KCl 14.4 g Na 2 HPO 4 or 22 g Na 2 HPO 4 7H 2 O 2.4 g KH 2 PO 4 or 2 g KH 2 PO4 Adjust ph to 7.4 Autoclave PBST
Western Blot Protocol (updated on 05/20/14) Required Solutions 10x PBS (1L) 80 g NaCl 2 g KCl 14.4 g Na 2 HPO 4 or 22 g Na 2 HPO 4 7H 2 O 2.4 g KH 2 PO 4 or 2 g KH 2 PO4 Adjust ph to 7.4 Autoclave PBST
Aseptic Technique. A GMP/GTP Training Module
 Aseptic Technique A GMP/GTP Training Module Aseptic Technique The GMP Facility manufactures products for clinical use These products must meet a number of requirements, one of which is that they are sterile
Aseptic Technique A GMP/GTP Training Module Aseptic Technique The GMP Facility manufactures products for clinical use These products must meet a number of requirements, one of which is that they are sterile
Human Hepatic Sinusoidal Endothelial Cell Manual
 Human Hepatic Sinusoidal Endothelial Cell Manual INSTRUCTION MANUAL SHIPPING CONDITIONS ZBM0093.00 Human Hepatic Sinusoidal Cells. Orders are delivered via Federal Express courier. All US and Canada orders
Human Hepatic Sinusoidal Endothelial Cell Manual INSTRUCTION MANUAL SHIPPING CONDITIONS ZBM0093.00 Human Hepatic Sinusoidal Cells. Orders are delivered via Federal Express courier. All US and Canada orders
BACTERIAL ENUMERATION
 BACTERIAL ENUMERATION In the study of microbiology, there are numerous occasions when it is necessary to either estimate or determine the number of bacterial cells in a broth culture or liquid medium.
BACTERIAL ENUMERATION In the study of microbiology, there are numerous occasions when it is necessary to either estimate or determine the number of bacterial cells in a broth culture or liquid medium.
Transfection reagent for visualizing lipofection with DNA. For ordering information, MSDS, publications and application notes see www.biontex.
 METAFECTENE FluoR Transfection reagent for visualizing lipofection with DNA For ordering information, MSDS, publications and application notes see www.biontex.com Description Cat. No. Size METAFECTENE
METAFECTENE FluoR Transfection reagent for visualizing lipofection with DNA For ordering information, MSDS, publications and application notes see www.biontex.com Description Cat. No. Size METAFECTENE
Flow cytometric Annexin V/Propidium Iodide measurement in A549 cells
 Project: VIGO Flow cytometric Annexin V/Propidium Iodide measurement in A549 cells Detecting apoptosis and necrosis in A549 cells AUTHORED BY: DATE: Cordula Hirsch 20.01.2014 REVIEWED BY: DATE: Harald
Project: VIGO Flow cytometric Annexin V/Propidium Iodide measurement in A549 cells Detecting apoptosis and necrosis in A549 cells AUTHORED BY: DATE: Cordula Hirsch 20.01.2014 REVIEWED BY: DATE: Harald
Transformation Protocol
 To make Glycerol Stocks of Plasmids ** To be done in the hood and use RNase/DNase free tips** 1. In a 10 ml sterile tube add 3 ml autoclaved LB broth and 1.5 ul antibiotic (@ 100 ug/ul) or 3 ul antibiotic
To make Glycerol Stocks of Plasmids ** To be done in the hood and use RNase/DNase free tips** 1. In a 10 ml sterile tube add 3 ml autoclaved LB broth and 1.5 ul antibiotic (@ 100 ug/ul) or 3 ul antibiotic
QuickZyme Soluble Collagen Assay
 QuickZyme Soluble Collagen Assay Version June 2012 FOR RESEARCH USE ONLY NOT FOR USE IN DIAGNOSTIC PROCEDURES This package insert must be read in its entirety before using this product. Introduction Collagen
QuickZyme Soluble Collagen Assay Version June 2012 FOR RESEARCH USE ONLY NOT FOR USE IN DIAGNOSTIC PROCEDURES This package insert must be read in its entirety before using this product. Introduction Collagen
Instructions. Torpedo sirna. Material. Important Guidelines. Specifications. Quality Control
 is a is a state of the art transfection reagent, specifically designed for the transfer of sirna and mirna into a variety of eukaryotic cell types. is a state of the art transfection reagent, specifically
is a is a state of the art transfection reagent, specifically designed for the transfer of sirna and mirna into a variety of eukaryotic cell types. is a state of the art transfection reagent, specifically
BioResearch. RAFT 3D Cell Culture Kit Protocol
 BioResearch RAFT 3D Cell Culture Kit Protocol RAFT 3D Cell Culture Kit This protocol describes in detail how to produce RAFT 3D Cell Cultures in 96-well plates, 24-well plates and 24-well inserts. It is
BioResearch RAFT 3D Cell Culture Kit Protocol RAFT 3D Cell Culture Kit This protocol describes in detail how to produce RAFT 3D Cell Cultures in 96-well plates, 24-well plates and 24-well inserts. It is
Optimized Protocol sirna Test Kit for Cell Lines and Adherent Primary Cells
 page 1 of 8 sirna Test Kit for Cell Lines and Adherent Primary Cells Kit principle Co-transfection of pmaxgfp, encoding the green fluorescent protein (GFP) from Pontellina p. with an sirna directed against
page 1 of 8 sirna Test Kit for Cell Lines and Adherent Primary Cells Kit principle Co-transfection of pmaxgfp, encoding the green fluorescent protein (GFP) from Pontellina p. with an sirna directed against
Cell Culture Protocol for Biogelx Peptide Hydrogel 2D and 3D Cell Culture PRO/BGX/001
 Protocol (PRO) Cell Culture Protocol for Hydrogel 2D and 3D Cell Culture PRO/BGX/001 Materials Provided Gel solution (stiffness of Gel will vary as per type of product requested), packaged in a glass vial.
Protocol (PRO) Cell Culture Protocol for Hydrogel 2D and 3D Cell Culture PRO/BGX/001 Materials Provided Gel solution (stiffness of Gel will vary as per type of product requested), packaged in a glass vial.
Bacterial Transformation with Green Fluorescent Protein. Table of Contents Fall 2012
 Bacterial Transformation with Green Fluorescent Protein pglo Version Table of Contents Bacterial Transformation Introduction..1 Laboratory Exercise...3 Important Laboratory Practices 3 Protocol...... 4
Bacterial Transformation with Green Fluorescent Protein pglo Version Table of Contents Bacterial Transformation Introduction..1 Laboratory Exercise...3 Important Laboratory Practices 3 Protocol...... 4
Agencourt RNAdvance Blood Kit for Free Circulating DNA and mirna/rna Isolation from 200-300μL of Plasma and Serum
 SUPPLEMENTAL PROTOCOL WHITE PAPER Agencourt RNAdvance Blood Kit for Free Circulating DNA and mirna/rna Isolation from 200-300μL of Plasma and Serum Bee Na Lee, Ph.D., Beckman Coulter Life Sciences Process
SUPPLEMENTAL PROTOCOL WHITE PAPER Agencourt RNAdvance Blood Kit for Free Circulating DNA and mirna/rna Isolation from 200-300μL of Plasma and Serum Bee Na Lee, Ph.D., Beckman Coulter Life Sciences Process
Standard Operating Procedures (SOPs) for the production and testing of embryonic stem cell lines in Taiwan Stem Cell Bank
 Standard Operating Procedures (SOPs) for the production and testing of embryonic stem cell lines in Taiwan Stem Cell Bank Bioresource Collection and Research Centre (BCRC) at Food Industry Research and
Standard Operating Procedures (SOPs) for the production and testing of embryonic stem cell lines in Taiwan Stem Cell Bank Bioresource Collection and Research Centre (BCRC) at Food Industry Research and
UltraClean Soil DNA Isolation Kit
 PAGE 1 UltraClean Soil DNA Isolation Kit Catalog # 12800-50 50 preps New improved PCR inhibitor removal solution (IRS) included Instruction Manual (New Alternative Protocol maximizes yields) Introduction
PAGE 1 UltraClean Soil DNA Isolation Kit Catalog # 12800-50 50 preps New improved PCR inhibitor removal solution (IRS) included Instruction Manual (New Alternative Protocol maximizes yields) Introduction
Xpert TM Karyotyping Teaching Kit
 Xpert TM Karyotyping Teaching Kit Product Code: CCK012 Contents 1. About the kit 2. Kit contents and storage instructions 3. Materials required but not provided in the kit 4. Aseptic techniques and good
Xpert TM Karyotyping Teaching Kit Product Code: CCK012 Contents 1. About the kit 2. Kit contents and storage instructions 3. Materials required but not provided in the kit 4. Aseptic techniques and good
Lab Exercise 3: Media, incubation, and aseptic technique
 Lab Exercise 3: Media, incubation, and aseptic technique Objectives 1. Compare the different types of media. 2. Describe the different formats of media, plate, tube etc. 3. Explain how to sterilize it,
Lab Exercise 3: Media, incubation, and aseptic technique Objectives 1. Compare the different types of media. 2. Describe the different formats of media, plate, tube etc. 3. Explain how to sterilize it,
Amaxa Mouse T Cell Nucleofector Kit
 Amaxa Mouse T Cell Nucleofector Kit For T cells isolated from C57BL/6 & BALB/c mice Evaluated for murine T cells isolated from C57BL/6 & BALB/c mice This protocol is designed for murine lymphocytes or
Amaxa Mouse T Cell Nucleofector Kit For T cells isolated from C57BL/6 & BALB/c mice Evaluated for murine T cells isolated from C57BL/6 & BALB/c mice This protocol is designed for murine lymphocytes or
MEF Starter Nucleofector Kit
 page 1 of 7 MEF Starter Nucleofector Kit for Mouse Embryonic Fibroblasts (MEF) MEF display significant phenotypic variations which depend on the strain, the genetic background of the mice they are isolated
page 1 of 7 MEF Starter Nucleofector Kit for Mouse Embryonic Fibroblasts (MEF) MEF display significant phenotypic variations which depend on the strain, the genetic background of the mice they are isolated
Inc. Wuhan. Quantity Pre-coated, ready to use 96-well strip plate 1 Plate sealer for 96 wells 4 Standard (liquid) 2
 Uscn Life Science Inc. Wuhan Website: www.uscnk.com Phone: +86 27 84259552 Fax: +86 27 84259551 E-mail: [email protected] ELISA Kit for Human Prostaglandin E1(PG-E1) Instruction manual Cat. No.: E0904Hu
Uscn Life Science Inc. Wuhan Website: www.uscnk.com Phone: +86 27 84259552 Fax: +86 27 84259551 E-mail: [email protected] ELISA Kit for Human Prostaglandin E1(PG-E1) Instruction manual Cat. No.: E0904Hu
ab83369 Alkaline Phosphatase Assay kit (Colorimetric)
 ab83369 Alkaline Phosphatase Assay kit (Colorimetric) Instructions for use: For the rapid, sensitive and accurate measurement of Alkaline Phosphatase in various samples. This product is for research use
ab83369 Alkaline Phosphatase Assay kit (Colorimetric) Instructions for use: For the rapid, sensitive and accurate measurement of Alkaline Phosphatase in various samples. This product is for research use
RPCI 004 v.002 Staining Procedure For all Directly Conjugated Reagents (Whole Blood Method)
 Immune Tolerance Network RPCI 004 v.002 Staining Procedure For all Directly Conjugated Reagents (Whole Blood Method) Author: Paul Wallace, Director, RPCI Laboratory of Flow Cytometry Approved by: Paul
Immune Tolerance Network RPCI 004 v.002 Staining Procedure For all Directly Conjugated Reagents (Whole Blood Method) Author: Paul Wallace, Director, RPCI Laboratory of Flow Cytometry Approved by: Paul
QIAGEN Supplementary Protocol
 QIAGEN Supplementary Protocol Isolation of Peripheral Blood Mononuclear Cells (PBMC) and Purification of Total RNA from PBMC Using the RNeasy Micro or Mini Kit This protocol describes how to isolate PBMC
QIAGEN Supplementary Protocol Isolation of Peripheral Blood Mononuclear Cells (PBMC) and Purification of Total RNA from PBMC Using the RNeasy Micro or Mini Kit This protocol describes how to isolate PBMC
RealLine HCV PCR Qualitative - Uni-Format
 Instructions for use PCR KIT FOR EXTRACTION OF RNA AND REAL TIME PCR DETECTION KIT FOR HEPATITIS C VIRUS RNA Research Use Only Qualitative Uni Format VBD0798 48 tests valid from: December 2013 Rev11122013
Instructions for use PCR KIT FOR EXTRACTION OF RNA AND REAL TIME PCR DETECTION KIT FOR HEPATITIS C VIRUS RNA Research Use Only Qualitative Uni Format VBD0798 48 tests valid from: December 2013 Rev11122013
RNA Extraction and Quantification, Reverse Transcription, and Real-time PCR (q-pcr)
 RNA Extraction and Quantification, Reverse Transcription, and Real-time Preparation of Samples Cells: o Remove media and wash cells 2X with cold PBS. (2 ml for 6 well plate or 3 ml for 6cm plate) Keep
RNA Extraction and Quantification, Reverse Transcription, and Real-time Preparation of Samples Cells: o Remove media and wash cells 2X with cold PBS. (2 ml for 6 well plate or 3 ml for 6cm plate) Keep
Human Umbilical Cord-derived Multipotent Mesenchymal Stromal Cells (huc-msc) Handling Instructions
 Human Umbilical Cord-derived Multipotent Mesenchymal Stromal Cells (huc-msc) Order No.: 19401-005, 19401-010 Handling Instructions For preclinical ex vivo use. Not intended for therapeutic use. Table of
Human Umbilical Cord-derived Multipotent Mesenchymal Stromal Cells (huc-msc) Order No.: 19401-005, 19401-010 Handling Instructions For preclinical ex vivo use. Not intended for therapeutic use. Table of
DNA Isolation Kit for Cells and Tissues
 DNA Isolation Kit for Cells and Tissues for 10 isolations of 00 mg each for tissue or 5 x 10 7 cultured cells Cat. No. 11 81 770 001 Principle Starting material Application Time required Results Benefits
DNA Isolation Kit for Cells and Tissues for 10 isolations of 00 mg each for tissue or 5 x 10 7 cultured cells Cat. No. 11 81 770 001 Principle Starting material Application Time required Results Benefits
Generation and Culture of Neural Progenitor Cells using the STEMdiff Neural System
 Generation and Culture of Neural Progenitor Cells using the STEMdiff Neural System i Table of Contents 1.0 Introduction... 1 2.0 Materials, Reagents and Equipment... 2 2.1 Materials Required for Neural
Generation and Culture of Neural Progenitor Cells using the STEMdiff Neural System i Table of Contents 1.0 Introduction... 1 2.0 Materials, Reagents and Equipment... 2 2.1 Materials Required for Neural
Proto col. GoClone Repor ter Construc ts: Sample Protocol for Adherent Cells. Tech support: 877-994-8240. Luciferase Assay System
 Luciferase Assay System Proto col GoClone Repor ter Construc ts: Sample Protocol for Adherent Cells LightSwitch Luciferase Assay System GoClone Reporter Assay Workflow Step 1: Seed cells in plate format.
Luciferase Assay System Proto col GoClone Repor ter Construc ts: Sample Protocol for Adherent Cells LightSwitch Luciferase Assay System GoClone Reporter Assay Workflow Step 1: Seed cells in plate format.
LAB 11 PLASMID DNA MINIPREP
 LAB 11 PLASMID DNA MINIPREP STUDENT GUIDE GOAL The objective of this lab is to perform extraction of plasmid DNA and analyze the results. OBJECTIVES After completion, the student should be able to: 1.
LAB 11 PLASMID DNA MINIPREP STUDENT GUIDE GOAL The objective of this lab is to perform extraction of plasmid DNA and analyze the results. OBJECTIVES After completion, the student should be able to: 1.
Especially developed for the transfection of mammalian cells with sirna or mirna
 METAFECTENE SI Especially developed for the transfection of mammalian cells with sirna or mirna For ordering information, MSDS, publications and application notes see www.biontex.com Description Cat. No.
METAFECTENE SI Especially developed for the transfection of mammalian cells with sirna or mirna For ordering information, MSDS, publications and application notes see www.biontex.com Description Cat. No.
HBV Quantitative Real Time PCR Kit
 Revision No.: ZJ0002 Issue Date: Aug 7 th, 2008 HBV Quantitative Real Time PCR Kit Cat. No.: HD-0002-01 For Use with LightCycler 1.0/LightCycler2.0/LightCycler480 (Roche) Real Time PCR Systems (Pls ignore
Revision No.: ZJ0002 Issue Date: Aug 7 th, 2008 HBV Quantitative Real Time PCR Kit Cat. No.: HD-0002-01 For Use with LightCycler 1.0/LightCycler2.0/LightCycler480 (Roche) Real Time PCR Systems (Pls ignore
Modulating Glucose Uptake in Skeletal Myotubes:
 icell Skeletal Myoblasts Application Protocol Introduction Modulating Glucose Uptake in Skeletal Myotubes: Insulin Induction with Bioluminescent Glucose Uptake Analysis The skeletal muscle is one of the
icell Skeletal Myoblasts Application Protocol Introduction Modulating Glucose Uptake in Skeletal Myotubes: Insulin Induction with Bioluminescent Glucose Uptake Analysis The skeletal muscle is one of the
Fast, easy and effective transfection reagent for mammalian cells
 METAFECTENE EASY + Fast, easy and effective transfection reagent for mammalian cells For ordering information, MSDS, publications and application notes see www.biontex.com Description Cat. No. Size METAFECTENE
METAFECTENE EASY + Fast, easy and effective transfection reagent for mammalian cells For ordering information, MSDS, publications and application notes see www.biontex.com Description Cat. No. Size METAFECTENE
Blood Collection and Processing SOP
 Brisbane Breast Bank Blood Collection and Processing SOP Breast Pathology Laboratory University of Queensland Centre for Clinical Research Blood Collection We collect 30ml of blood from patients who have
Brisbane Breast Bank Blood Collection and Processing SOP Breast Pathology Laboratory University of Queensland Centre for Clinical Research Blood Collection We collect 30ml of blood from patients who have
Evaporation-based Microfluidic Production of. Oil-free Cell-Containing Hydrogel Particles
 Supporting Information Evaporation-based Microfluidic Production of Oil-free Cell-Containing Hydrogel Particles Rong Fan, a Kubra Naqvi, b Krishna Patel, c Jun Sun d and Jiandi Wan *a a Microsystems Engineering
Supporting Information Evaporation-based Microfluidic Production of Oil-free Cell-Containing Hydrogel Particles Rong Fan, a Kubra Naqvi, b Krishna Patel, c Jun Sun d and Jiandi Wan *a a Microsystems Engineering
Rat creatine kinase MM isoenzyme (CK-MM) ELISA Kit
 Rat creatine kinase MM isoenzyme (CK-MM) ELISA Kit Catalog Number. CSB-E14405r For the quantitative determination of rat creatine kinase MM isoenzyme (CK-MM) concentrations in serum, plasma and tissue
Rat creatine kinase MM isoenzyme (CK-MM) ELISA Kit Catalog Number. CSB-E14405r For the quantitative determination of rat creatine kinase MM isoenzyme (CK-MM) concentrations in serum, plasma and tissue
MTT Cell Proliferation Assay
 ATCC 30-1010K Store at 4 C This product is intended for laboratory research purposes only. It is not intended for use in humans, animals or for diagnostics. Introduction Measurement of cell viability and
ATCC 30-1010K Store at 4 C This product is intended for laboratory research purposes only. It is not intended for use in humans, animals or for diagnostics. Introduction Measurement of cell viability and
TransformAid Bacterial Transformation Kit
 Home Contacts Order Catalog Support Search Alphabetical Index Numerical Index Restriction Endonucleases Modifying Enzymes PCR Kits Markers Nucleic Acids Nucleotides & Oligonucleotides Media Transfection
Home Contacts Order Catalog Support Search Alphabetical Index Numerical Index Restriction Endonucleases Modifying Enzymes PCR Kits Markers Nucleic Acids Nucleotides & Oligonucleotides Media Transfection
Growth and Maintenance of T-REx Cell Lines
 USER GUIDE Growth and Maintenance of T-REx Cell Lines Catalog Numbers R710-07, R714-07, R718-07, R722-07 Document Part Number 25-0272 Publication Number MAN0000106 Revision 3.0 For Research Use Only. Not
USER GUIDE Growth and Maintenance of T-REx Cell Lines Catalog Numbers R710-07, R714-07, R718-07, R722-07 Document Part Number 25-0272 Publication Number MAN0000106 Revision 3.0 For Research Use Only. Not
Chromatin Immunoprecipitation (ChIP)
 Chromatin Immunoprecipitation (ChIP) Day 1 A) DNA shearing 1. Samples Dissect tissue (One Mouse OBs) of interest and transfer to an eppendorf containing 0.5 ml of dissecting media (on ice) or PBS but without
Chromatin Immunoprecipitation (ChIP) Day 1 A) DNA shearing 1. Samples Dissect tissue (One Mouse OBs) of interest and transfer to an eppendorf containing 0.5 ml of dissecting media (on ice) or PBS but without
Minimal residual disease detection in Acute Myeloid Leukaemia on a Becton Dickinson flow cytometer
 Minimal residual disease detection in Acute Myeloid Leukaemia on a Becton Dickinson flow cytometer Purpose This procedure gives instruction on minimal residual disease (MRD) detection in patients with
Minimal residual disease detection in Acute Myeloid Leukaemia on a Becton Dickinson flow cytometer Purpose This procedure gives instruction on minimal residual disease (MRD) detection in patients with
Creatine Kinase (CK) Enzymatic Assay Kit Manual Catalog #: 3460-07
 Creatine Kinase (CK) Enzymatic Assay Kit Manual Catalog #: 3460-07 TABLE OF CONTENTS GENERAL INFORMATION... 2 Product Description... 2 Procedure Overview... 2 Kit Contents, Storage and Shelf Life... 3
Creatine Kinase (CK) Enzymatic Assay Kit Manual Catalog #: 3460-07 TABLE OF CONTENTS GENERAL INFORMATION... 2 Product Description... 2 Procedure Overview... 2 Kit Contents, Storage and Shelf Life... 3
Cell Culture Experiments
 Cell Culture Experiments Cell cultures are another way to observe and analyze biological systems. These in vitro (in glass; outside the living body and in an artificial environment) models allow experimental
Cell Culture Experiments Cell cultures are another way to observe and analyze biological systems. These in vitro (in glass; outside the living body and in an artificial environment) models allow experimental
Rat creatine kinase MM isoenzyme (CK-MM) ELISA Kit
 Rat creatine kinase MM isoenzyme (CK-MM) ELISA Kit Catalog Number. CSB-E14405r For the quantitative determination of rat creatine kinase MM isoenzyme (CK-MM) concentrations in serum, plasma, tissue homogenates.
Rat creatine kinase MM isoenzyme (CK-MM) ELISA Kit Catalog Number. CSB-E14405r For the quantitative determination of rat creatine kinase MM isoenzyme (CK-MM) concentrations in serum, plasma, tissue homogenates.
GRS Plasmid Purification Kit Transfection Grade GK73.0002 (2 MaxiPreps)
 1 GRS Plasmid Purification Kit Transfection Grade GK73.0002 (2 MaxiPreps) (FOR RESEARCH ONLY) Sample : Expected Yield : Endotoxin: Format : Operation Time : Elution Volume : 50-400 ml of cultured bacterial
1 GRS Plasmid Purification Kit Transfection Grade GK73.0002 (2 MaxiPreps) (FOR RESEARCH ONLY) Sample : Expected Yield : Endotoxin: Format : Operation Time : Elution Volume : 50-400 ml of cultured bacterial
Microbiological Testing of the Sawyer Mini Filter. 16 December 2013. Summary
 Microbiological Testing of the Sawyer Mini Filter 16 December 2013 Summary The Sawyer Mini Filter was tested for its ability to remove three microorganisms Raoultella terrigena, Bacillus subtilis, and
Microbiological Testing of the Sawyer Mini Filter 16 December 2013 Summary The Sawyer Mini Filter was tested for its ability to remove three microorganisms Raoultella terrigena, Bacillus subtilis, and
MEF Nucleofector Kit 1 and 2
 page 1 of 7 MEF Nucleofector Kit 1 and 2 for Mouse Embryonic Fibroblasts (MEF) MEF display significant phenotypic variations which depend on the strain, the genetic background of the they are isolated
page 1 of 7 MEF Nucleofector Kit 1 and 2 for Mouse Embryonic Fibroblasts (MEF) MEF display significant phenotypic variations which depend on the strain, the genetic background of the they are isolated
Running protein gels and detection of proteins
 Running protein gels and detection of proteins 1. Protein concentration determination using the BIO RAD reagent This assay uses a colour change reaction to give a direct measurement of protein concentration.
Running protein gels and detection of proteins 1. Protein concentration determination using the BIO RAD reagent This assay uses a colour change reaction to give a direct measurement of protein concentration.
EXPERIMENT 9 - IDENTIFYING FEATURES OF MUTANT EMBRYO USING NOMARSKI MICROSCOPY (GENE TWO)
 EXPERIMENT 9 - IDENTIFYING FEATURES OF MUTANT EMBRYO USING NOMARSKI MICROSCOPY (GENE TWO) Purpose: To introduce Differential Interference Contrast (DIC) or Nomarski Interference Contrast (NIC) microscopy
EXPERIMENT 9 - IDENTIFYING FEATURES OF MUTANT EMBRYO USING NOMARSKI MICROSCOPY (GENE TWO) Purpose: To introduce Differential Interference Contrast (DIC) or Nomarski Interference Contrast (NIC) microscopy
ENUMERATION OF MICROORGANISMS. To learn the different techniques used to count the number of microorganisms in a sample.
 ENUMERATION OF MICROORGANISMS I. OBJECTIVES To learn the different techniques used to count the number of microorganisms in a sample. To be able to differentiate between different enumeration techniques
ENUMERATION OF MICROORGANISMS I. OBJECTIVES To learn the different techniques used to count the number of microorganisms in a sample. To be able to differentiate between different enumeration techniques
HEK 293 K V 7.1/minK Cell Line
 B SYS GmbH HEK 293 K V 7.1/minK Cell Line Specification Sheet B SYS GmbH B SYS GmbH HEK 293 K V 7.1/minK Cells Page 2 TABLE OF CONTENTS 1. BACKGROUND...3 1.1. The cardiac IKs current is encoded by K V
B SYS GmbH HEK 293 K V 7.1/minK Cell Line Specification Sheet B SYS GmbH B SYS GmbH HEK 293 K V 7.1/minK Cells Page 2 TABLE OF CONTENTS 1. BACKGROUND...3 1.1. The cardiac IKs current is encoded by K V
Environmental Water Testing: Surface Water, Groundwater, Hard Water, Wastewater, & Seawater
 Document: AND Sol Env 08 2013 Environmental Water Testing: Surface Water, Groundwater, Hard Water, Wastewater, & Seawater Matrix specific sample preparation and testing methods for environmental waters
Document: AND Sol Env 08 2013 Environmental Water Testing: Surface Water, Groundwater, Hard Water, Wastewater, & Seawater Matrix specific sample preparation and testing methods for environmental waters
An In-Gel Digestion Protocol
 An In-Gel Digestion Protocol This protocol describes the digestion of a protein present in an SDS-PAGE gel band with trypsin. The band can be taken from either a 1D or 2D electrophoresis gel. Reagents
An In-Gel Digestion Protocol This protocol describes the digestion of a protein present in an SDS-PAGE gel band with trypsin. The band can be taken from either a 1D or 2D electrophoresis gel. Reagents
Effective Methods For Culturing Breast Cancer Cell Lines
 Abstract Effective Methods For Culturing Breast Cancer Cell Lines William H. Marshall II Life Science Team Cell culturing is one of the most useful and prolific techniques practiced in biological science
Abstract Effective Methods For Culturing Breast Cancer Cell Lines William H. Marshall II Life Science Team Cell culturing is one of the most useful and prolific techniques practiced in biological science
ab185915 Protein Sumoylation Assay Ultra Kit
 ab185915 Protein Sumoylation Assay Ultra Kit Instructions for Use For the measuring in vivo protein sumoylation in various samples This product is for research use only and is not intended for diagnostic
ab185915 Protein Sumoylation Assay Ultra Kit Instructions for Use For the measuring in vivo protein sumoylation in various samples This product is for research use only and is not intended for diagnostic
Western Blot Analysis with Cell Samples Grown in Channel-µ-Slides
 Western Blot Analysis with Cell Samples Grown in Channel-µ-Slides Polyacrylamide gel electrophoresis (PAGE) and subsequent analyses are common tools in biochemistry and molecular biology. This Application
Western Blot Analysis with Cell Samples Grown in Channel-µ-Slides Polyacrylamide gel electrophoresis (PAGE) and subsequent analyses are common tools in biochemistry and molecular biology. This Application
Biology 29 Cell Structure and Function Spring, 2009 Springer LABORATORY 2:CHLOROPLASTS AND PHOTOREDUCTION
 Biology 29 Cell Structure and Function Spring, 2009 Springer LABORATORY 2:CHLOROPLASTS AND PHOTOREDUCTION In this laboratory we will purify chloroplasts from spinach by differential centrifugation, then
Biology 29 Cell Structure and Function Spring, 2009 Springer LABORATORY 2:CHLOROPLASTS AND PHOTOREDUCTION In this laboratory we will purify chloroplasts from spinach by differential centrifugation, then
Annexin V-EGFP Apoptosis Detection Kit
 ab14153 Annexin V-EGFP Apoptosis Detection Kit Instructions for Use For the rapid, sensitive and accurate measurement of apoptosis in various samples This product is for research use only and is not intended
ab14153 Annexin V-EGFP Apoptosis Detection Kit Instructions for Use For the rapid, sensitive and accurate measurement of apoptosis in various samples This product is for research use only and is not intended
Western Blotting: Mini-gels
 Western Blotting: Mini-gels Materials a Protein Extraction Buffer (for callus or kernel), Solution Stock Final Volume Tris-HCl ph 80 1 M 200 mm 20 ml NaCl 4 M 100 mm 25 ml Sucrose 2 M 400 mm 20 ml EDTA
Western Blotting: Mini-gels Materials a Protein Extraction Buffer (for callus or kernel), Solution Stock Final Volume Tris-HCl ph 80 1 M 200 mm 20 ml NaCl 4 M 100 mm 25 ml Sucrose 2 M 400 mm 20 ml EDTA
Transformation of the bacterium E. coli. using a gene for Green Fluorescent Protein
 Transformation of the bacterium E. coli using a gene for Green Fluorescent Protein Background In molecular biology, transformation refers to a form of genetic exchange in which the genetic material carried
Transformation of the bacterium E. coli using a gene for Green Fluorescent Protein Background In molecular biology, transformation refers to a form of genetic exchange in which the genetic material carried
Dot Blot Analysis. Teacher s Guidebook. (Cat. # BE 502) think proteins! think G-Biosciences www.gbiosciences.com
 PR110 G-Biosciences 1-800-628-7730 1-314-991-6034 [email protected] A Geno Technology, Inc. (USA) brand name Dot Blot Analysis Teacher s Guidebook (Cat. # BE 502) think proteins! think G-Biosciences
PR110 G-Biosciences 1-800-628-7730 1-314-991-6034 [email protected] A Geno Technology, Inc. (USA) brand name Dot Blot Analysis Teacher s Guidebook (Cat. # BE 502) think proteins! think G-Biosciences
Malondialdehyde (MDA) ELISA
 Malondialdehyde (MDA) ELISA For the quantitative determination of MDA in serum, plasma and other biological fluids Cat. No. KT-21493 For Research Use Only. Not for use in diagnostic procedures. Page 1
Malondialdehyde (MDA) ELISA For the quantitative determination of MDA in serum, plasma and other biological fluids Cat. No. KT-21493 For Research Use Only. Not for use in diagnostic procedures. Page 1
Annexin V-FITC Apoptosis Detection Kit
 ab14085 Annexin V-FITC Apoptosis Detection Kit Instructions for Use For the rapid, sensitive and accurate measurement of Apoptosis in living cells (adherent and suspension). This product is for research
ab14085 Annexin V-FITC Apoptosis Detection Kit Instructions for Use For the rapid, sensitive and accurate measurement of Apoptosis in living cells (adherent and suspension). This product is for research
Growth and Maintenance of Insect Cell Lines
 Insect Cell Lines Version K July 12, 2002 25-0127 Growth and Maintenance of Insect Cell Lines www.invitrogen.com [email protected] ii Table of Contents Table of Contents... iii Important Information...iv
Insect Cell Lines Version K July 12, 2002 25-0127 Growth and Maintenance of Insect Cell Lines www.invitrogen.com [email protected] ii Table of Contents Table of Contents... iii Important Information...iv
Basics of Cell Culture A student laboratory manual
 Basics of Cell Culture A student laboratory manual By Golnar Afshar PhD Table of Contents: Introduction 3 Laboratory Exercises Laboratory Safety 3 Aseptic Technique Exercise30 30 Laboratory Notebooks5
Basics of Cell Culture A student laboratory manual By Golnar Afshar PhD Table of Contents: Introduction 3 Laboratory Exercises Laboratory Safety 3 Aseptic Technique Exercise30 30 Laboratory Notebooks5
UltraClean Forensic DNA Isolation Kit (Single Prep Format)
 UltraClean Forensic DNA Isolation Kit (Single Prep Format) Catalog No. Quantity 14000-10 10 preps 14000-S 1 prep Instruction Manual Please recycle Version: 10302012 1 Table of Contents Introduction...
UltraClean Forensic DNA Isolation Kit (Single Prep Format) Catalog No. Quantity 14000-10 10 preps 14000-S 1 prep Instruction Manual Please recycle Version: 10302012 1 Table of Contents Introduction...
