Hy's law is presented as the fundamental rule of thumb when assessing liver specific lab parameters (points summarized) :
|
|
|
- Franklin French
- 9 years ago
- Views:
Transcription
1 Referral concerning : Draft Guidance for Industry : Drug-induced liver injury: Premarketing Clinical Evaluation FDA, 25 October 2007 (Docket 2007D-0396) From: Swedish MPA Staff and consultants, 19 December Summary of comments of draft guidance document : Volk, ~ 0 This draft is mainly devoted to clinical assessment, thus not preclinical assessment, action and documentation on premarketing signs of Drug-Induced Liver Injuries, DILIs. It is pointed out that drugs which cause elevated AT as well as bilirubin levels or impaired function such as inhibition of synthesis of clotting factor are those at risk to cause severe DILI. It is also stated that drugs with, in retrospect, clear hepatotoxic potential have not shown any signal at all in animal studies, not shown dose-related toxicity and show a very low rate in humans, usuall y , all suggesting to reflect host specific factors. Another problem pointed at is that some drugs do not show any hepatotoxicity by elevation of AT. The frequency of AT elevation of a drug in a population is not predictive either, the degree of elevation and liver function tests may be better predictors. ALT is considered more liver specific, although expressed in other tissues. Increased bilirubin.levels is an indicator of severed liver function, especially accompanied with increased AT. Hy's law is presented as the fundamental rule of thumb when assessing liver specific lab parameters (points summarized) : Suspected liver toxic drug shows more frequent 3-fold or greater ULN o f ALT or AST than control or placebo. 2. Among these subjects as in 1), if they also show elevation of TBL more than 2xULN, without initial findings of cholestasis. 3. No other explanation, clinical condition, for increased AT and TBL. One case which fulfills the criteria of Hy's Law in a trial is ominous, two or more cases highly predictive of potential DILI. 0~ 0 0 7::1) - 0 3?b C_ -I'
2 The document further outlines clinical evaluation of DILI, by "General considerations", "Case Report Forms", "Interpretations of Signals of DILI or Acute Liver Failure", and "Analysis of Signals of DILI". Lastly, several illustrative cases of DILI are assembled in the Appendix A. General comments : The scope of the guidance document is clincal assessment/evaluation of possible occurence of liver injury and their predictive value for severe DILI. It is a conscientiously and well written document with several practical and useful rules of thumb and suggestions on how to investigate, differentiate, document and act on signs of hepatotoxicity in patients on study drug. It reflects the current thinking in the field of drug-induced liver injury and can be considered the state-of-the art for guidance of the management of DILI. It is recognized that the discussion on preclinical hepatotoxicity and its relevance to clinical hepatotoxicity lies outside the scope of the draft. However, it would have been interesting with some overview of the preclinical-clinical bridging information related to liver toxicity. Even though it is recognized that preclinical tests do not predict rare drug-induced hepatotoxicity in humans, they sometimes need to be revisited and/or extended when clinical signals occur. We suggest future update and inclusion of any new preclinical information which may be of relevance to the diagnosis or prevention of DILI. (See also European Draft guideline "Detection of early signals of hepatotoxicity from non-clinical data" EMEA/CHMP/SWP/ /2006 ). Please find minor points to consider related to structure and clarifications below. Specific comment 1 : On p. 2, line 62 : Suggested addition : "....gather, apart from detailed information of treatment with other drug(s), additional clinical and laboratory information : :" On p. 2, line 79 : Proposed addition: "...thousand subjects were studied. In addition, often patients with underlying risk factors for liver injury were excluded in the clinical trials ". Specific comment 2 : The wording on p.5, line 183, could be further clarified : Current wording : "As a rule of thumb, based on Zimmerman's original estimate of percent mortality associated with hepatocellular injury sufficient to impair the liver bilirubin excretory function, severe DILI can be estimated to occur at a rate of at least one-thenth the rate of socalled Hy's Law cases (Temple 2001)".
3 Proposed wording : "As a rule of thumb, since hepatocellular damage causing impaired liver bilirubin excreto ry function is associated with 10-50% mo rtality, based on Zimmernian's original estimate, the rate of severe DILI may be at least one tenth of the rate of so-called Hy's Law cases (Temple 2001). _ Specific comment 3 : On p.5, line 201. Insert subtitle, e.g. "Major findings" and number the bullet points in bold text (1, 2, 3) on p. 5 and 6. Specific comment 4 : On page 6, line 234 it is stated : " We are not aware of false positive Hy's Law findings". The authors should explain what they really mean by this. Do the authors mean that when a patient is found to have ALT?3 ULN and bilirubin >2 ULN that the drug is going to give severe hepatotoxicity or any hepatotoxicity? In the paper by Bjornsson and Olsson in Hepatology 2005, there were a total of 32 cases with erythromycin suspected liver injury (with concomitant jaundice) whereas no cases resulted in death or liver transplantation. Erythromycin has very rarely on its own been associated with a fatal outcome. Specific comment 5 : On p. 6, line 239: Proposed addition: " The degree of assurance depends on the population exposed for a long enough time, how discontinuation criteria were set in the clinical trials regarding liver test abnormalities and on the rate of severe DILI that would be of interest. " Specific comment 6 : On page 10, concerning elevations of aminotransferases (AT) in ischemic hepatitis it is stated that AT >10,000, it is perhaps better to keep on mentioning the multiples of the upper limit normal (e.g. >25xULN). Specific comment 7 : On p.8, section 5. "Decision to Stop Drug Administration" : The first sentence is suggested to be further clarified to avoid misunderstanding : Current w riting :
4 "It has been observed that dechallenge (stopping drug administration) does not always, or even usually, result in immediate improvement in abnormal lab values." Proposed writing : " Stopping drug administration, dechallenge, does not always result in immediate improvement in abnormal lab values". Specific comment 8 : On p 9 & 10, section 6. "Evaluating Data for Alternative Causes" : Although probably rare outside oncology trials, malignancy should be mentioned, as a possible cause for liver enzyme elevation. Proposed change p. 10, line 404 : Current wording: "Biliary tract disorders", Proposed wording : Hepatobiliary disorders. Proposed change p.10, line 406 : "Malignant interruption : Current wording of the biliary tract also should be considered. " Proposed wording : "Malignant infiltration of the liver and biliary tract should also be considered. " Specific comment 9 : The possible role of eosinophilia in assessment of the aetiology and severity of DILI could be further clarified somewhere in the draft. For example, on p. 9, line 374, eosinophilia is mentioned (as a co-finding with other symptoms and AT-elevations) suggestive of discontinuation of study drug. Also, eosinophilia is mentioned on p. 10, line 441, as suggestive of immunological mechanism and if present speaks against rechallenge. Specific comment 10 : On the top of page 14, line 505, it is mentioned that "several in vitro methods are available to detect and quantify binding for a drug or its metabolites to liver proteins, including radiochemical and immunological methods". Perhaps it should be acknowledged that unfortunately these methods are very rarely useful in prediction of hepatotoxicity or useful in diagnosing DILI. It would though be of interest to mention in which rare instances this can be of use, such as in treatment with isoniazid, where analysis of acetylation status is potentially useful.
5 Program Detecting and Investigating Drug-Induced Liver Injury During Clinical Trials What should we be doing about uncommon but potentially serious adverse events (e.g., DILI, Stevens- Johnson syndrome, rhabdomyolysis, prolonged QT interval, etc.) found during controlled clinical trials? A national/international discussion and debate on issues raised by the draft guidance of 25 October 2007 (Docket No. 2007D-0396) toward building consensus among interested parties of the pharmaceutical industry, regulatory bodies, academic investigators and consultants, and public groups or individuals. The program is being co-sponsored by the Food and Drug Administration/Center for Drug Evaluation and Research (FDA/CDER), the Pharmaceutical Research and Manufacturers of America (PhRMA), and the American Association for the Study of Liver Disease (AASLD). National Labor College, Silver Spring MD, March New Hampshire Avenue at Powder Mill Road, Silver Spring MD Wednesday, 26 March 7:30 Continental Breakfast 8:00 Introductions and Brief (5') Opening Statements Janet Woodcock, FDA/CDE R Alan Goldhammer, PhRM A John Vierling, AASL D Session 1 : When should an investigational drug be stopped during a trial? Moderator, Paul Watkins, U N C 8:15 Clinical meaning of elevated aminotransferase activity? Naga Chalasani, IN U 8:45 Liver test elevations seen in patients on placebo. Robert Tipping, Merck 9:15 Lessons from isoniazid - would it be approved today? John Senior, FDA/CDE R 9:45 Brea k 10:15 A simple tool for finding important cases in a clinical trial Kate Gelperin, FDA/CDE R 10:45 Hy's Law explained Adrian Reuben, MU SC 11 :15 Was it the drug, or a disease? How to determine it. Don Rockey, UT SW 11 :45 General discussion Al l 12:15 Lunch Moderator, Paul Seligman, FDA/CDE R Session IIA: Should rechallenge be used to prove the drug caused the reaction? 1 :30 Immune-allergic sensitization to a xenobiotic Jack Uetrecht, U Toronto 2:00 Hierarchy of evidence - how much do we need to know? Leonard Seeff, NI H 2:30 Balancing the risks and benefits of rechallenge Christine Hunt, GS K 3:00-3:30 General discussion -speakers and audience Al l Break Session IIB : Can we find a truly predictive biomarker to prevent serious adverse reactions? 4:00 What kind of a biomarker do we need? Mark Avigan, FDA/CDE R 4:30 How might we find one? Jack Bloom, Lilly 5:00 Role of clinical trials in cracking the nut Arthur Holden, SAEC * 5:30 General discussion -speakers and audience Al l 6:00-6:45 Reception: wine and cheese, mingle and relax --- *Serious Adverse Event Consortium 7:30 Reassemble for dinner. serve soup, salad, main cours e Discuss ethical, management, political issue s Ethical perspectives Dave Wendler, NI H Industry perspectives Jay Barth, Merc k International regulatory perspectives (to be confirmed ) ' Dessert, coffee
6 Thursday, 27 March 7:30 Continental Breakfast Session III : Should pat ients with stable underlying liver disease be incuded? Moderator, John Pears, AstraZeneca 8 :00 Study the patients who will be treated Robert Temple, FDA/CDER 8:30 Would this increase the risk of DILI? William Lee, UT S W 9:00 Case for continuing to exclude them Arie Regev, Lilly 9:30 General discussion, panelists and audienc e 10:00 Break 10:30 Breakout groups I, II, and II I Each group to formulate and specify the questions to be asked for consensus buildin g 12:00 Lunch 1 :00 Public statements : Brief 5" statements (arguments, questions, comments) from interested persons, groups 2:00 Asking the questions and discussion of eac h 1) What should be the stopping rules for study drug administration? 2) When should rechallenge be done/not done? 3) Should patients with preexisting liver disease be studied? 4) Other questions, issue s 3:00 Adjourn For details and changes follow information posted at website : www. fda.gov/cder/livertox Registration by AASLD : $350 for industry; $175 for government or academia Lodging reservations on your own at NLC or in Silver Spring
U.S. Food and Drug Administration
 U.S. Food and Drug Administration Notice: Archived Document The content in this document is provided on the FDA s website for reference purposes only. It was current when produced, but is no longer maintained
U.S. Food and Drug Administration Notice: Archived Document The content in this document is provided on the FDA s website for reference purposes only. It was current when produced, but is no longer maintained
Guidance for Industry Drug-Induced Liver Injury: Premarketing Clinical Evaluation
 Guidance for Industry Drug-Induced Liver Injury: Premarketing Clinical Evaluation U.S. Department of Health and Human Services Food and Drug Administration Center for Drug Evaluation and Research (CDER)
Guidance for Industry Drug-Induced Liver Injury: Premarketing Clinical Evaluation U.S. Department of Health and Human Services Food and Drug Administration Center for Drug Evaluation and Research (CDER)
INITIATING ORAL AUBAGIO (teriflunomide) THERAPY
 FOR YOUR PATIENTS WITH RELAPSING FORMS OF MS INITIATING ORAL AUBAGIO (teriflunomide) THERAPY WARNING: HEPATOTOXICITY AND RISK OF TERATOGENICITY Severe liver injury including fatal liver failure has been
FOR YOUR PATIENTS WITH RELAPSING FORMS OF MS INITIATING ORAL AUBAGIO (teriflunomide) THERAPY WARNING: HEPATOTOXICITY AND RISK OF TERATOGENICITY Severe liver injury including fatal liver failure has been
EMEA PUBLIC STATEMENT ON LEFLUNOMIDE (ARAVA) - SEVERE AND SERIOUS HEPATIC REACTIONS -
 The European Agency for the Evaluation of Medicinal Products Post-authorisation evaluation of medicines for human use London, 12 March 2001 Doc. Ref: EMEA/H/5611/01/en EMEA PUBLIC STATEMENT ON LEFLUNOMIDE
The European Agency for the Evaluation of Medicinal Products Post-authorisation evaluation of medicines for human use London, 12 March 2001 Doc. Ref: EMEA/H/5611/01/en EMEA PUBLIC STATEMENT ON LEFLUNOMIDE
Guidance for Clinical Investigators, Sponsors, and IRBs
 Guidance for Clinical Investigators, Sponsors, and IRBs Adverse Event Reporting to IRBs Improving Human Subject Protection U.S. Department of Health and Human Services Office of the Commissioner (OC) Center
Guidance for Clinical Investigators, Sponsors, and IRBs Adverse Event Reporting to IRBs Improving Human Subject Protection U.S. Department of Health and Human Services Office of the Commissioner (OC) Center
Approach to Abnormal Liver Tests
 Approach to Abnormal Liver Tests Naga P. Chalasani, MD, FACG Professor of Medicine and Cellular & Integrative Physiology Director, Division of Gastroenterology and Hepatology Indiana University School
Approach to Abnormal Liver Tests Naga P. Chalasani, MD, FACG Professor of Medicine and Cellular & Integrative Physiology Director, Division of Gastroenterology and Hepatology Indiana University School
LIVER FUNCTION TESTS AND STATINS
 LIVER FUNCTION TESTS AND STATINS Philippe J. Zamor and Mark W. Russo Current Opinion in Cardiology 2011,26:338 341 SUMMARY Purpose of review: To discuss recent data on statins in patients with elevated
LIVER FUNCTION TESTS AND STATINS Philippe J. Zamor and Mark W. Russo Current Opinion in Cardiology 2011,26:338 341 SUMMARY Purpose of review: To discuss recent data on statins in patients with elevated
NP/PA Clinical Hepatology Fellowship Summary of Year-Long Curriculum
 OVERVIEW OF THE FELLOWSHIP The goal of the AASLD NP/PA Fellowship is to provide a 1-year postgraduate hepatology training program for nurse practitioners and physician assistants in a clinical outpatient
OVERVIEW OF THE FELLOWSHIP The goal of the AASLD NP/PA Fellowship is to provide a 1-year postgraduate hepatology training program for nurse practitioners and physician assistants in a clinical outpatient
5.07.09. Aubagio. Aubagio (teriflunomide) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.07.09 Subject: Aubagio Page: 1 of 6 Last Review Date: December 5, 2014 Aubagio Description Aubagio (teriflunomide)
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.07.09 Subject: Aubagio Page: 1 of 6 Last Review Date: December 5, 2014 Aubagio Description Aubagio (teriflunomide)
Novartis Gilenya FDO Program Clinical Protocol and Highlights from Prescribing Information (PI)
 Novartis Gilenya FDO Program Clinical Protocol and Highlights from Prescribing Information (PI) Highlights from Prescribing Information - the link to the full text PI is as follows: http://www.pharma.us.novartis.com/product/pi/pdf/gilenya.pdf
Novartis Gilenya FDO Program Clinical Protocol and Highlights from Prescribing Information (PI) Highlights from Prescribing Information - the link to the full text PI is as follows: http://www.pharma.us.novartis.com/product/pi/pdf/gilenya.pdf
Latest advice for medicines users The monthly newsletter from the MHRA and its independent advisor the Commission on Human Medicines
 Latest advice for medicines users The monthly newsletter from the MHRA and its independent advisor the Commission on Human Medicines Volume 6, Issue 10, May 2013 Drug safety advice Yellow card scheme Stop
Latest advice for medicines users The monthly newsletter from the MHRA and its independent advisor the Commission on Human Medicines Volume 6, Issue 10, May 2013 Drug safety advice Yellow card scheme Stop
Patterns of abnormal LFTs and their differential diagnosis
 Patterns of abnormal LFTs and their differential diagnosis Professor Matthew Cramp South West Liver Unit and Peninsula Schools of Medicine and Dentistry, Plymouth Summary liver function / liver function
Patterns of abnormal LFTs and their differential diagnosis Professor Matthew Cramp South West Liver Unit and Peninsula Schools of Medicine and Dentistry, Plymouth Summary liver function / liver function
Introduction to Post marketing Drug Safety Surveillance: Pharmacovigilance in FDA/CDER
 Introduction to Post marketing Drug Safety Surveillance: Pharmacovigilance in FDA/CDER LT Andrew Fine, Pharm.D., BCPS Safety Evaluator Division of Pharmacovigilance Office of Pharmacovigilance and Epidemiology
Introduction to Post marketing Drug Safety Surveillance: Pharmacovigilance in FDA/CDER LT Andrew Fine, Pharm.D., BCPS Safety Evaluator Division of Pharmacovigilance Office of Pharmacovigilance and Epidemiology
A.P. Chen, MD Director, Developmental Therapeutics Clinic Division of Cancer Treatment and Diagnosis National Cancer Institute
 A.P. Chen, MD Director, Developmental Therapeutics Clinic Division of Cancer Treatment and Diagnosis National Cancer Institute Click to view Biosketch and Presentation Abstract or page down to review presentation
A.P. Chen, MD Director, Developmental Therapeutics Clinic Division of Cancer Treatment and Diagnosis National Cancer Institute Click to view Biosketch and Presentation Abstract or page down to review presentation
Liver Diseases. An Essential Guide for Nurses and Health Care Professionals
 Brochure More information from http://www.researchandmarkets.com/reports/1047385/ Liver Diseases. An Essential Guide for Nurses and Health Care Professionals Description: Liver disease is a rapidly growing
Brochure More information from http://www.researchandmarkets.com/reports/1047385/ Liver Diseases. An Essential Guide for Nurses and Health Care Professionals Description: Liver disease is a rapidly growing
Teriflunomide is the active metabolite of Leflunomide, a drug employed since 1994 for the treatment of rheumatoid arthritis (Baselt, 2011).
 Page 1 of 10 ANALYTE NAME AND STRUCTURE TERIFLUNOMIDE Teriflunomide TRADE NAME Aubagio CATEGORY Antimetabolite TEST CODE PURPOSE Therapeutic Drug Monitoring GENERAL RELEVANCY BACKGROUND sclerosis. The
Page 1 of 10 ANALYTE NAME AND STRUCTURE TERIFLUNOMIDE Teriflunomide TRADE NAME Aubagio CATEGORY Antimetabolite TEST CODE PURPOSE Therapeutic Drug Monitoring GENERAL RELEVANCY BACKGROUND sclerosis. The
GUIDELINE ON THE SUBMISSION OF RISK MANAGEMENT PLAN DOCUMENTS
 APPENDIX GUIDELINE ON THE SUBMISSION OF RISK MANAGEMENT PLAN DOCUMENTS This document is intended to provide guidance on the submission of risk management plan (RMP) documents in support of NDA, GDA and
APPENDIX GUIDELINE ON THE SUBMISSION OF RISK MANAGEMENT PLAN DOCUMENTS This document is intended to provide guidance on the submission of risk management plan (RMP) documents in support of NDA, GDA and
Recommended Warning for Overthe-Counter. Containing Drug Products and Labeling Statements Regarding Serious Skin Reactions
 Recommended Warning for Overthe-Counter Acetaminophen- Containing Drug Products and Labeling Statements Regarding Serious Skin Reactions Guidance for Industry DRAFT GUIDANCE This guidance document is being
Recommended Warning for Overthe-Counter Acetaminophen- Containing Drug Products and Labeling Statements Regarding Serious Skin Reactions Guidance for Industry DRAFT GUIDANCE This guidance document is being
Causality Assessment in Practice Pharmaceutical Industry Perspective. Lachlan MacGregor Senior Safety Scientist F. Hoffmann-La Roche Ltd.
 Causality Assessment in Practice Pharmaceutical Industry Perspective Lachlan MacGregor Senior Safety Scientist F. Hoffmann-La Roche Ltd., Basel Disclaimer: The opinions expressed in this presentation are
Causality Assessment in Practice Pharmaceutical Industry Perspective Lachlan MacGregor Senior Safety Scientist F. Hoffmann-La Roche Ltd., Basel Disclaimer: The opinions expressed in this presentation are
Guidance for Industry Determining the Extent of Safety Data Collection Needed in Late Stage Premarket and Postapproval Clinical Investigations
 Guidance for Industry Determining the Extent of Safety Data Collection Needed in Late Stage Premarket and Postapproval Clinical Investigations DRAFT GUIDANCE This guidance document is being distributed
Guidance for Industry Determining the Extent of Safety Data Collection Needed in Late Stage Premarket and Postapproval Clinical Investigations DRAFT GUIDANCE This guidance document is being distributed
Prof. of Tropical Medicine Faculty of Medicine Alexandria University
 prof. Dr. Ali El-Kady (MD) Prof. of Tropical Medicine Faculty of Medicine Alexandria University DRUGS THAT MAY CAUSE LIVER DYSFUNCTION DAMAGE The liver is the principal organ that is capable of converting
prof. Dr. Ali El-Kady (MD) Prof. of Tropical Medicine Faculty of Medicine Alexandria University DRUGS THAT MAY CAUSE LIVER DYSFUNCTION DAMAGE The liver is the principal organ that is capable of converting
Safety Assessment for IND Safety Reporting Guidance for Industry
 Safety Assessment for IND Safety Reporting Guidance for Industry DRAFT GUIDANCE This guidance document is being distributed for comment purposes only. Comments and suggestions regarding this draft document
Safety Assessment for IND Safety Reporting Guidance for Industry DRAFT GUIDANCE This guidance document is being distributed for comment purposes only. Comments and suggestions regarding this draft document
Summary of the risk management plan (RMP) for Ofev (nintedanib)
 EMA/738120/2014 Summary of the risk management plan (RMP) for Ofev (nintedanib) This is a summary of the risk management plan (RMP) for Ofev, which details the measures to be taken in order to ensure that
EMA/738120/2014 Summary of the risk management plan (RMP) for Ofev (nintedanib) This is a summary of the risk management plan (RMP) for Ofev, which details the measures to be taken in order to ensure that
Regulatory Issues in Genetic Testing and Targeted Drug Development
 Regulatory Issues in Genetic Testing and Targeted Drug Development Janet Woodcock, M.D. Deputy Commissioner for Operations Food and Drug Administration October 12, 2006 Genetic and Genomic Tests are Types
Regulatory Issues in Genetic Testing and Targeted Drug Development Janet Woodcock, M.D. Deputy Commissioner for Operations Food and Drug Administration October 12, 2006 Genetic and Genomic Tests are Types
How Public-Private Partnerships Benefit All Stakeholders Douglas C. Throckmorton, MD Deputy Director for Regulatory Programs, CDER, FDA
 How Public-Private Partnerships Benefit All Stakeholders Douglas C. Throckmorton, MD Deputy Director for Regulatory Programs, CDER, FDA September 10, 2013 Outline Need for improved science in support of
How Public-Private Partnerships Benefit All Stakeholders Douglas C. Throckmorton, MD Deputy Director for Regulatory Programs, CDER, FDA September 10, 2013 Outline Need for improved science in support of
Introduction to Post-marketing Drug Safety Surveillance Pharmacovigilance Practice:
 Introduction to Post-marketing Drug Safety Surveillance Pharmacovigilance Practice: US Perspective Min Chen, M.S. R.Ph. Pharmacovigilance Consulting, LLC The opinions expressed are those of the author.
Introduction to Post-marketing Drug Safety Surveillance Pharmacovigilance Practice: US Perspective Min Chen, M.S. R.Ph. Pharmacovigilance Consulting, LLC The opinions expressed are those of the author.
Alanine aminotransferase (serum, plasma)
 Alanine aminotransferase (serum, plasma) 1 Name and description of analyte 1.1 Name of analyte Alanine aminotransferase (ALT) 1.2 Alternative names Systematic name L alanine:2 oxoglutarate aminotransferase
Alanine aminotransferase (serum, plasma) 1 Name and description of analyte 1.1 Name of analyte Alanine aminotransferase (ALT) 1.2 Alternative names Systematic name L alanine:2 oxoglutarate aminotransferase
PHARMACOVIGILANCE GUIDELINES
 PHARMACOVIGILANCE GUIDELINES What is Pharmacovigilance? Pharmacovigilance is defined as the science and activities concerned with the detection, assessment, understanding and prevention of adverse reactions
PHARMACOVIGILANCE GUIDELINES What is Pharmacovigilance? Pharmacovigilance is defined as the science and activities concerned with the detection, assessment, understanding and prevention of adverse reactions
Learn More About Product Labeling
 Learn More About Product Labeling Product label The product label is developed during the formal process of review and approval by regulatory agencies of any medicine or medical product. There are specific
Learn More About Product Labeling Product label The product label is developed during the formal process of review and approval by regulatory agencies of any medicine or medical product. There are specific
Guidance for Industry
 Guidance for Industry Cancer Drug and Biological Products Clinical Data in Marketing Applications U.S. Department of Health and Human Services Food and Drug Administration Center for Drug Evaluation and
Guidance for Industry Cancer Drug and Biological Products Clinical Data in Marketing Applications U.S. Department of Health and Human Services Food and Drug Administration Center for Drug Evaluation and
Subject: No. Page PROTOCOL AND CASE REPORT FORM DEVELOPMENT AND REVIEW Standard Operating Procedure
 703 1 of 11 POLICY The Beaumont Research Coordinating Center (BRCC) will provide advice to clinical trial investigators on protocol development, content and format. Upon request, the BRCC will review a
703 1 of 11 POLICY The Beaumont Research Coordinating Center (BRCC) will provide advice to clinical trial investigators on protocol development, content and format. Upon request, the BRCC will review a
Preface. TTY: (888) 232-6348 or [email protected]. Hepatitis C Counseling and Testing, contact: 800-CDC-INFO (800-232-4636)
 Preface The purpose of this CDC Hepatitis C Counseling and Testing manual is to provide guidance for hepatitis C counseling and testing of individuals born during 1945 1965. The guide was used in draft
Preface The purpose of this CDC Hepatitis C Counseling and Testing manual is to provide guidance for hepatitis C counseling and testing of individuals born during 1945 1965. The guide was used in draft
Guidance on Reviewing and Reporting Unanticipated Problems Involving Risks to Subjects or Others and Adverse Events
 Office for Human Research Protections (OHRP) Department of Health and Human Services (HHS) Guidance on Reviewing and Reporting Unanticipated Problems Involving Risks to Subjects or Others and Adverse Events
Office for Human Research Protections (OHRP) Department of Health and Human Services (HHS) Guidance on Reviewing and Reporting Unanticipated Problems Involving Risks to Subjects or Others and Adverse Events
HEPATOLOGY CLERKSHIP
 College of Osteopathic Medicine HEPATOLOGY CLERKSHIP Office for Clinical Affairs 515-271-1629 FAX 515-271-1727 Elective Rotation General Description This elective rotation is a four (4) week introductory,
College of Osteopathic Medicine HEPATOLOGY CLERKSHIP Office for Clinical Affairs 515-271-1629 FAX 515-271-1727 Elective Rotation General Description This elective rotation is a four (4) week introductory,
First In Human Pediatric Trials and Safety Assessment for Rare and Orphan Diseases
 First In Human Pediatric Trials and Safety Assessment for Rare and Orphan Diseases Andrew E. Mulberg, MD, FAAP Division Deputy Director OND/ODE3/DGIEP FDA Partnership is the Key Coming together is a beginning;
First In Human Pediatric Trials and Safety Assessment for Rare and Orphan Diseases Andrew E. Mulberg, MD, FAAP Division Deputy Director OND/ODE3/DGIEP FDA Partnership is the Key Coming together is a beginning;
Evaluation of Liver Function tests in Primary Care. Abid Suddle Institute of Liver Studies, KCH
 Evaluation of Liver Function tests in Primary Care Abid Suddle Institute of Liver Studies, KCH Liver Function tests Markers of hepatocellular damage Cholestasis Liver synthetic function Markers of Hepatocellular
Evaluation of Liver Function tests in Primary Care Abid Suddle Institute of Liver Studies, KCH Liver Function tests Markers of hepatocellular damage Cholestasis Liver synthetic function Markers of Hepatocellular
Selected Requirements of Prescribing Information
 The Selected Requirements of Prescribing Information (SRPI) is a checklist of 42 important format prescribing information (PI) items based on labeling regulations [21 CFR 201.56(d) and 201.57] and guidances.
The Selected Requirements of Prescribing Information (SRPI) is a checklist of 42 important format prescribing information (PI) items based on labeling regulations [21 CFR 201.56(d) and 201.57] and guidances.
Summary of treatment benefits
 Risk Management Plan PEMETREXED Powder for concentrate for Solution for infusion Pemetrexed is also indicated as monotherapy for the maintenance treatment of locally advanced or metastatic non small cell
Risk Management Plan PEMETREXED Powder for concentrate for Solution for infusion Pemetrexed is also indicated as monotherapy for the maintenance treatment of locally advanced or metastatic non small cell
The use of the WHO-UMC system for standardised case causality assessment
 The use of the WHO-UMC system for standardised case causality assessment Why causality assessment? An inherent problem in pharmacovigilance is that most case reports concern suspected adverse drug reactions.
The use of the WHO-UMC system for standardised case causality assessment Why causality assessment? An inherent problem in pharmacovigilance is that most case reports concern suspected adverse drug reactions.
Guidance for Industry Diabetes Mellitus Evaluating Cardiovascular Risk in New Antidiabetic Therapies to Treat Type 2 Diabetes
 Guidance for Industry Diabetes Mellitus Evaluating Cardiovascular Risk in New Antidiabetic Therapies to Treat Type 2 Diabetes U.S. Department of Health and Human Services Food and Drug Administration Center
Guidance for Industry Diabetes Mellitus Evaluating Cardiovascular Risk in New Antidiabetic Therapies to Treat Type 2 Diabetes U.S. Department of Health and Human Services Food and Drug Administration Center
Summary of the risk management plan (RMP) for Orkambi (lumacaftor and ivacaftor)
 EMA/662624/2015 Summary of the risk management plan (RMP) for Orkambi (lumacaftor and ivacaftor) This is a summary of the risk management plan (RMP) for Orkambi, which details the measures to be taken
EMA/662624/2015 Summary of the risk management plan (RMP) for Orkambi (lumacaftor and ivacaftor) This is a summary of the risk management plan (RMP) for Orkambi, which details the measures to be taken
Service Definition with all Clinical Terms Service: Laprascopic Cholecystectomy Clinic (No Gallstones in bile duct)
 Service Definition with all Clinical Terms Service: Laprascopic Cholecystectomy Clinic (No Gallstones in bile duct) Section 1 Service Details Service ID: 7540540 Service Comments: Referrer Alert: Service
Service Definition with all Clinical Terms Service: Laprascopic Cholecystectomy Clinic (No Gallstones in bile duct) Section 1 Service Details Service ID: 7540540 Service Comments: Referrer Alert: Service
2.1 AST can be measured in heparin plasma or serum. 3 Summary of clinical applications and limitations of measurements
 Aspartate aminotransferase (serum, plasma) 1 Name and description of analyte 1.1 Name of analyte Aspartate aminotransferase (AST) 1.2 Alternative names Systematic name L aspartate:2 oxoglutarate aminotransferase
Aspartate aminotransferase (serum, plasma) 1 Name and description of analyte 1.1 Name of analyte Aspartate aminotransferase (AST) 1.2 Alternative names Systematic name L aspartate:2 oxoglutarate aminotransferase
ANALYSIS OF POISON CONTROL CENTER DATA FOR ACETAMINOPHEN-CONTAINING PRODUCTS 1998-2000
 ANALYSIS OF POISON CONTROL CENTER DATA FOR ACETAMINOPHEN-CONTAINING PRODUCTS 1998-2000 Prepared by Julie A. Skare, PhD. Health Care Research Center The Procter & Gamble Company 8700 Mason-Montgomery Road
ANALYSIS OF POISON CONTROL CENTER DATA FOR ACETAMINOPHEN-CONTAINING PRODUCTS 1998-2000 Prepared by Julie A. Skare, PhD. Health Care Research Center The Procter & Gamble Company 8700 Mason-Montgomery Road
Drugs Believed Capable of Inducing Autoimmune Hepatitis
 Drugs Believed Capable of Inducing Autoimmune Hepatitis Leonard B. Seeff, M.D. National Institutes of Diabetes and Digestive and Kidney Diseases National Institutes of Health Immune-Mediated Liver Diseases
Drugs Believed Capable of Inducing Autoimmune Hepatitis Leonard B. Seeff, M.D. National Institutes of Diabetes and Digestive and Kidney Diseases National Institutes of Health Immune-Mediated Liver Diseases
GUIDELINES ON MEDICAL DEVICES
 EUROPEAN COMMISSION DG Internal Market, Industry, Entrepreneurship and SMEs Consumer, Environmental and Health Technologies Health technology and Cosmetics MEDDEV 2.7/3 revision 3 May 2015 GUIDELINES ON
EUROPEAN COMMISSION DG Internal Market, Industry, Entrepreneurship and SMEs Consumer, Environmental and Health Technologies Health technology and Cosmetics MEDDEV 2.7/3 revision 3 May 2015 GUIDELINES ON
Corporate Medical Policy Genetic Testing for Fanconi Anemia
 Corporate Medical Policy Genetic Testing for Fanconi Anemia File Name: Origination: Last CAP Review: Next CAP Review: Last Review: genetic_testing_for_fanconi_anemia 03/2015 3/2016 3/2017 3/2016 Description
Corporate Medical Policy Genetic Testing for Fanconi Anemia File Name: Origination: Last CAP Review: Next CAP Review: Last Review: genetic_testing_for_fanconi_anemia 03/2015 3/2016 3/2017 3/2016 Description
Not All Clinical Trials Are Created Equal Understanding the Different Phases
 Not All Clinical Trials Are Created Equal Understanding the Different Phases This chapter will help you understand the differences between the various clinical trial phases and how these differences impact
Not All Clinical Trials Are Created Equal Understanding the Different Phases This chapter will help you understand the differences between the various clinical trial phases and how these differences impact
Guidance for Industry and Investigators
 Guidance for Industry and Investigators Safety Reporting Requirements for INDs and BA/BE Studies U.S. Department of Health and Human Services Food and Drug Administration Center for Drug Evaluation and
Guidance for Industry and Investigators Safety Reporting Requirements for INDs and BA/BE Studies U.S. Department of Health and Human Services Food and Drug Administration Center for Drug Evaluation and
Application for a Marketing Authorisation: Requirements and Criteria for the Assessment of QT Prolonging Potential
 Application for a Marketing Authorisation: Requirements and Criteria for the Assessment of QT Prolonging Potential Bundesinstitut für Arzneimittel Dr. med. Clemens Mittmann Bundesinstitut für Arzneimittel
Application for a Marketing Authorisation: Requirements and Criteria for the Assessment of QT Prolonging Potential Bundesinstitut für Arzneimittel Dr. med. Clemens Mittmann Bundesinstitut für Arzneimittel
Albumin. Prothrombin time. Total protein
 Hepatitis C Fact Sheet February 2016 www.hepatitis.va.gov Laboratory Tests and Hepatitis If you have hepatitis C, your doctor will use laboratory tests to about learn more about your individual hepatitis
Hepatitis C Fact Sheet February 2016 www.hepatitis.va.gov Laboratory Tests and Hepatitis If you have hepatitis C, your doctor will use laboratory tests to about learn more about your individual hepatitis
INTERNATIONAL CONFERENCE ON HARMONISATION OF TECHNICAL REQUIREMENTS FOR REGISTRATION OF PHARMACEUTICALS FOR HUMAN USE S1A. Current Step 4 version
 INTERNATIONAL CONFERENCE ON HARMONISATION OF TECHNICAL REQUIREMENTS FOR REGISTRATION OF PHARMACEUTICALS FOR HUMAN USE ICH HARMONISED TRIPARTITE GUIDELINE GUIDELINE ON THE NEED FOR CARCINOGENICITY STUDIES
INTERNATIONAL CONFERENCE ON HARMONISATION OF TECHNICAL REQUIREMENTS FOR REGISTRATION OF PHARMACEUTICALS FOR HUMAN USE ICH HARMONISED TRIPARTITE GUIDELINE GUIDELINE ON THE NEED FOR CARCINOGENICITY STUDIES
Understanding CA 125 Levels A GUIDE FOR OVARIAN CANCER PATIENTS. foundationforwomenscancer.org
 Understanding CA 125 Levels A GUIDE FOR OVARIAN CANCER PATIENTS foundationforwomenscancer.org Contents Introduction...1 CA 125................................... 1 The CA 125 Test...2 The Use of the CA
Understanding CA 125 Levels A GUIDE FOR OVARIAN CANCER PATIENTS foundationforwomenscancer.org Contents Introduction...1 CA 125................................... 1 The CA 125 Test...2 The Use of the CA
Docetaxel + Carboplatin + Trastuzumab (TCH) Adjuvant Breast Cancer
 Docetaxel + Carboplatin + Trastuzumab (TCH) Adjuvant Breast Cancer Background: A non-anthracycline based regimen for high-risk, HER 2 positive breast cancer in the adjuvant setting (BCIRG 006). Patient
Docetaxel + Carboplatin + Trastuzumab (TCH) Adjuvant Breast Cancer Background: A non-anthracycline based regimen for high-risk, HER 2 positive breast cancer in the adjuvant setting (BCIRG 006). Patient
Update and Review of Medication Assisted Treatments
 Update and Review of Medication Assisted Treatments for Opiate and Alcohol Use Disorders Richard N. Whitney, MD Medical Director Addiction Services Shepherd Hill Newark, Ohio Medication Assisted Treatment
Update and Review of Medication Assisted Treatments for Opiate and Alcohol Use Disorders Richard N. Whitney, MD Medical Director Addiction Services Shepherd Hill Newark, Ohio Medication Assisted Treatment
Bilirubin (serum, plasma)
 Bilirubin (serum, plasma) 1 Analyte 1.1 Name of analyte Bilirubin 1.2 Alternative names None 1.3 NLMC code 1.4 Description of analyte Bilirubin is a linear tetrapyrrole (MW 585 Da), the final product of
Bilirubin (serum, plasma) 1 Analyte 1.1 Name of analyte Bilirubin 1.2 Alternative names None 1.3 NLMC code 1.4 Description of analyte Bilirubin is a linear tetrapyrrole (MW 585 Da), the final product of
TB Drugs: Common Side Effects and Interactions. First-line Drugs 11/21/2012. Adverse Events of First-line TB Drugs
 TB Drugs: Common Side Effects and Interactions L. Beth Gadkowski MD MPH MS Assistant Professor Division of Infectious Diseases Eastern Virginia Medical School First-line Drugs Isoniazid (INH) Rifampin
TB Drugs: Common Side Effects and Interactions L. Beth Gadkowski MD MPH MS Assistant Professor Division of Infectious Diseases Eastern Virginia Medical School First-line Drugs Isoniazid (INH) Rifampin
Hepatitis C. Laboratory Tests and Hepatitis C
 Hepatitis C Laboratory Tests and Hepatitis C If you have hepatitis C, your doctor will use laboratory tests to check your health. This handout will help you understand what the major tests are and what
Hepatitis C Laboratory Tests and Hepatitis C If you have hepatitis C, your doctor will use laboratory tests to check your health. This handout will help you understand what the major tests are and what
BACKGROUND MEDIA INFORMATION Fast facts about liver disease
 BACKGROUND MEDIA INFORMATION Fast facts about liver disease Liver, or hepatic, disease comprises a wide range of complex conditions that affect the liver. Liver diseases are extremely costly in terms of
BACKGROUND MEDIA INFORMATION Fast facts about liver disease Liver, or hepatic, disease comprises a wide range of complex conditions that affect the liver. Liver diseases are extremely costly in terms of
Below, this letter outlines [patient name] s medical history, prognosis, and treatment rationale.
![Below, this letter outlines [patient name] s medical history, prognosis, and treatment rationale. Below, this letter outlines [patient name] s medical history, prognosis, and treatment rationale.](/thumbs/25/5563775.jpg) [Date] [Name of Contact] [Title] [Name of Health Insurance Company] [Address] [City, State, Zip Code] Insured: [Patient Name] Policy Number: [Number] Group Number: [Number] Diagnosis: [Diagnosis and ICD-9-CM
[Date] [Name of Contact] [Title] [Name of Health Insurance Company] [Address] [City, State, Zip Code] Insured: [Patient Name] Policy Number: [Number] Group Number: [Number] Diagnosis: [Diagnosis and ICD-9-CM
The Clinical Trials Process an educated patient s guide
 The Clinical Trials Process an educated patient s guide Gwen L. Nichols, MD Site Head, Oncology Roche TCRC, Translational and Clinical Research Center New York DISCLAIMER I am an employee of Hoffmann-
The Clinical Trials Process an educated patient s guide Gwen L. Nichols, MD Site Head, Oncology Roche TCRC, Translational and Clinical Research Center New York DISCLAIMER I am an employee of Hoffmann-
Medical management of CHF: A New Class of Medication. Al Timothy, M.D. Cardiovascular Institute of the South
 Medical management of CHF: A New Class of Medication Al Timothy, M.D. Cardiovascular Institute of the South Disclosures Speakers Bureau for Amgen Background Chronic systolic congestive heart failure remains
Medical management of CHF: A New Class of Medication Al Timothy, M.D. Cardiovascular Institute of the South Disclosures Speakers Bureau for Amgen Background Chronic systolic congestive heart failure remains
What to Do with the Patient With Abnormal Liver Enzymes? Nizar N. Zein, M.D. The Cleveland Clinic
 What to Do with the Patient With Abnormal Liver Enzymes? Nizar N. Zein, M.D. The Cleveland Clinic Introduction Elevated liver enzymes is often not a clinical problem by itself. However it is a warning
What to Do with the Patient With Abnormal Liver Enzymes? Nizar N. Zein, M.D. The Cleveland Clinic Introduction Elevated liver enzymes is often not a clinical problem by itself. However it is a warning
Patient Handbook on Stem Cell Therapies
 Patient Handbook on Stem Cell Therapies Appendix I of the Guidelines for the Clinical Translation of Stem Cells www.isscr.org 2008, International Society for Stem Cell Research 2 Introduction We have all
Patient Handbook on Stem Cell Therapies Appendix I of the Guidelines for the Clinical Translation of Stem Cells www.isscr.org 2008, International Society for Stem Cell Research 2 Introduction We have all
Original Policy Date
 MP 5.01.20 Tysabri (natalizumab) Medical Policy Section Prescription Drug Issue 12:2013 Original Policy Date 12:2013 Last Review Status/Date Local Policy/12:2013 Return to Medical Policy Index Disclaimer
MP 5.01.20 Tysabri (natalizumab) Medical Policy Section Prescription Drug Issue 12:2013 Original Policy Date 12:2013 Last Review Status/Date Local Policy/12:2013 Return to Medical Policy Index Disclaimer
Drug-Induced Liver Injury (DILI)
 Drug-Induced Liver Injury (DILI) CLINICAL INVESTIGATOR TRAINING COURSE Holiday Inn, College Park MD November 15, 2012 Lana L. Pauls, MPH John R. Senior, MD Office of Surveillance & Epidemiology Center
Drug-Induced Liver Injury (DILI) CLINICAL INVESTIGATOR TRAINING COURSE Holiday Inn, College Park MD November 15, 2012 Lana L. Pauls, MPH John R. Senior, MD Office of Surveillance & Epidemiology Center
Advanced Management Issues in HIV Medicine
 Is a Cure Possible for HIV? Department of Infectious Disease and R.J. Fasenmyer Center for Clinical Immunology 6th Annual Advanced Management Issues in HIV Medicine May 30 31, 2013 InterContinental Hotel
Is a Cure Possible for HIV? Department of Infectious Disease and R.J. Fasenmyer Center for Clinical Immunology 6th Annual Advanced Management Issues in HIV Medicine May 30 31, 2013 InterContinental Hotel
Participating in Alzheimer s Disease Clinical Trials and Studies
 Participating in Alzheimer s Disease Clinical Trials and Studies FACT SHEET When Margaret was diagnosed with earlystage Alzheimer s disease at age 68, she wanted to do everything possible to combat the
Participating in Alzheimer s Disease Clinical Trials and Studies FACT SHEET When Margaret was diagnosed with earlystage Alzheimer s disease at age 68, she wanted to do everything possible to combat the
Disclosures. Consultant and Speaker for Biogen Idec, TEVA Neuroscience, EMD Serrono, Mallinckrodt, Novartis, Genzyme, Accorda Therapeutics
 Mitzi Joi Williams, MD Neurologist MS Center of Atlanta, Atlanta, GA Disclosures Consultant and Speaker for Biogen Idec, TEVA Neuroscience, EMD Serrono, Mallinckrodt, Novartis, Genzyme, Accorda Therapeutics
Mitzi Joi Williams, MD Neurologist MS Center of Atlanta, Atlanta, GA Disclosures Consultant and Speaker for Biogen Idec, TEVA Neuroscience, EMD Serrono, Mallinckrodt, Novartis, Genzyme, Accorda Therapeutics
GT-020 Phase 1 Clinical Trial: Results of Second Cohort
 GT-020 Phase 1 Clinical Trial: Results of Second Cohort July 29, 2014 NASDAQ: GALT www.galectintherapeutics.com 2014 Galectin Therapeutics inc. Forward-Looking Statement This presentation contains, in
GT-020 Phase 1 Clinical Trial: Results of Second Cohort July 29, 2014 NASDAQ: GALT www.galectintherapeutics.com 2014 Galectin Therapeutics inc. Forward-Looking Statement This presentation contains, in
CLINICAL POLICY Department: Medical Management Document Name: Opdivo Reference Number: CP.PHAR.121 Effective Date: 07/15
 Page: 1 of 6 IMPORTANT REMINDER This Clinical Policy has been developed by appropriately experienced and licensed health care professionals based on a thorough review and consideration of generally accepted
Page: 1 of 6 IMPORTANT REMINDER This Clinical Policy has been developed by appropriately experienced and licensed health care professionals based on a thorough review and consideration of generally accepted
North of Tyne Area Prescribing Committee
 North of Tyne Area Prescribing Committee ANTIPSYCHOTICS IN PSYCHOSIS, BIPOLAR DISORDER AND AUGMENTATION THERAPY IN TREATMENT RESISTANT DEPRESSION Information for Primary Care Updated November 2013 This
North of Tyne Area Prescribing Committee ANTIPSYCHOTICS IN PSYCHOSIS, BIPOLAR DISORDER AND AUGMENTATION THERAPY IN TREATMENT RESISTANT DEPRESSION Information for Primary Care Updated November 2013 This
Guidance for Industry and Review Staff Target Product Profile A Strategic Development Process Tool
 Guidance for Industry and Review Staff Product Profile A Strategic Development Process Tool DRAFT GUIDANCE This guidance document is being distributed for comment purposes only. Comments and suggestions
Guidance for Industry and Review Staff Product Profile A Strategic Development Process Tool DRAFT GUIDANCE This guidance document is being distributed for comment purposes only. Comments and suggestions
Obstetric Cholestasis (itching liver disorder) Information for parents-to-be
 Oxford University Hospitals NHS Trust Obstetric Cholestasis (itching liver disorder) Information for parents-to-be page 2 You have been given this leaflet because you have been diagnosed with (or are suspected
Oxford University Hospitals NHS Trust Obstetric Cholestasis (itching liver disorder) Information for parents-to-be page 2 You have been given this leaflet because you have been diagnosed with (or are suspected
Summary of the risk management plan (RMP) for Otezla (apremilast)
 EMA/741412/2014 Summary of the risk management plan (RMP) for Otezla (apremilast) This is a summary of the risk management plan (RMP) for Otezla, which details the measures to be taken in order to ensure
EMA/741412/2014 Summary of the risk management plan (RMP) for Otezla (apremilast) This is a summary of the risk management plan (RMP) for Otezla, which details the measures to be taken in order to ensure
Adverse Events in Clinical Trials: Definitions and Documentation
 Clinical and Translational Science Institute / CTSI at the University of California, San Francisco Welcome to Online Training for Clinical Research Coordinators Adverse Events in Clinical Trials: Definitions
Clinical and Translational Science Institute / CTSI at the University of California, San Francisco Welcome to Online Training for Clinical Research Coordinators Adverse Events in Clinical Trials: Definitions
Preoperative Laboratory and Diagnostic Studies
 Preoperative Laboratory and Diagnostic Studies Preoperative Labratorey and Diagnostic Studies The concept of standardized testing in all presurgical patients regardless of age or medical condition is no
Preoperative Laboratory and Diagnostic Studies Preoperative Labratorey and Diagnostic Studies The concept of standardized testing in all presurgical patients regardless of age or medical condition is no
DRUG APPROVAL PROCESS FOR THE TREATMENT OF ALZHEIMER S DISEASE
 Research DRUG APPROVAL PROCESS FOR THE TREATMENT OF ALZHEIMER S DISEASE There are certain principles that should be followed when involving people with Alzheimer s disease in research. For more information,
Research DRUG APPROVAL PROCESS FOR THE TREATMENT OF ALZHEIMER S DISEASE There are certain principles that should be followed when involving people with Alzheimer s disease in research. For more information,
Guidelines for the Use of Controlled Substances in the Treatment of Pain Adopted by the New Hampshire Medical Society, July 1998
 Guidelines for the Use of Controlled Substances in the Treatment of Pain Adopted by the New Hampshire Medical Society, July 1998 Section I: Preamble The New Hampshire Medical Society believes that principles
Guidelines for the Use of Controlled Substances in the Treatment of Pain Adopted by the New Hampshire Medical Society, July 1998 Section I: Preamble The New Hampshire Medical Society believes that principles
Disclosures. Interpreting Liver Tests: What Do They Mean? Liver Function Tests. Objectives. Common Tests. Case 1
 Disclosures Interpreting Liver Tests: What Do They Mean? I have no financial disclosures to make. Roman Perri, MD Vanderbilt University Medical Center Nashville, TN Objectives Discuss tests commonly used
Disclosures Interpreting Liver Tests: What Do They Mean? I have no financial disclosures to make. Roman Perri, MD Vanderbilt University Medical Center Nashville, TN Objectives Discuss tests commonly used
Amylase and Lipase Tests
 Amylase and Lipase Tests Also known as: Amy Formal name: Amylase Related tests: Lipase The Test The blood amylase test is ordered, often along with a lipase test, to help diagnose and monitor acute or
Amylase and Lipase Tests Also known as: Amy Formal name: Amylase Related tests: Lipase The Test The blood amylase test is ordered, often along with a lipase test, to help diagnose and monitor acute or
The State of the Liver in the Adult Patient after Fontan Palliation
 The State of the Liver in the Adult Patient after Fontan Palliation Fred Wu, M.D. Boston Adult Congenital Heart Service Boston Children s Hospital/Brigham & Women s Hospital 7 th National Adult Congenital
The State of the Liver in the Adult Patient after Fontan Palliation Fred Wu, M.D. Boston Adult Congenital Heart Service Boston Children s Hospital/Brigham & Women s Hospital 7 th National Adult Congenital
Rare Diseases: Common Issues in Drug Development Guidance for Industry
 Rare Diseases: Common Issues in Drug Development Guidance for Industry DRAFT GUIDANCE This guidance document is being distributed for comment purposes only. Comments and suggestions regarding this draft
Rare Diseases: Common Issues in Drug Development Guidance for Industry DRAFT GUIDANCE This guidance document is being distributed for comment purposes only. Comments and suggestions regarding this draft
Guidance for Industry Migraine: Developing Drugs for Acute Treatment
 Guidance for Industry Migraine: Developing Drugs for Acute Treatment DRAFT GUIDANCE This guidance document is being distributed for comment purposes only. Comments and suggestions regarding this draft
Guidance for Industry Migraine: Developing Drugs for Acute Treatment DRAFT GUIDANCE This guidance document is being distributed for comment purposes only. Comments and suggestions regarding this draft
Southern Derbyshire. Shared Care Pathology Guidelines. Abnormal Liver Function Tests (LFTs) in Adults
 Southern Derbyshire Shared Care Pathology Guidelines Abnormal Liver Function Tests (LFTs) in Adults Purpose of Guideline The management of patients with abnormal liver function test results Scope This
Southern Derbyshire Shared Care Pathology Guidelines Abnormal Liver Function Tests (LFTs) in Adults Purpose of Guideline The management of patients with abnormal liver function test results Scope This
Prior Authorization Guideline
 Prior Authorization Guideline Guideline: PDP IBT Inj - Vivitrol Therapeutic Class: Central Nervous System Agents Therapeutic Sub-Class: Opiate Antagonist Client: 2007 PDP IBT Inj Approval Date: 2/20/2007
Prior Authorization Guideline Guideline: PDP IBT Inj - Vivitrol Therapeutic Class: Central Nervous System Agents Therapeutic Sub-Class: Opiate Antagonist Client: 2007 PDP IBT Inj Approval Date: 2/20/2007
SYNOPSIS. Risperidone: Clinical Study Report CR003274
 SYNOPSIS Protocol No: CR003274 Title of Study: An Open-Label, Long-Term Trial of Risperidone Long-Acting Microspheres in the Treatment of Subjects Diagnosed with Schizophrenia Coordinating Investigator:
SYNOPSIS Protocol No: CR003274 Title of Study: An Open-Label, Long-Term Trial of Risperidone Long-Acting Microspheres in the Treatment of Subjects Diagnosed with Schizophrenia Coordinating Investigator:
LEFLUNOMIDE (Adults)
 Shared Care Guideline DRUG: Introduction: LEFLUNOMIDE (Adults) Indication: Disease modifying drug for rheumatoid arthritis and psoriatic arthritis Licensing Information: Disease modifying drug for active
Shared Care Guideline DRUG: Introduction: LEFLUNOMIDE (Adults) Indication: Disease modifying drug for rheumatoid arthritis and psoriatic arthritis Licensing Information: Disease modifying drug for active
Achieving Regulatory Success: Areas of focus for biotechnology companies. Michael J. Schlosser, PhD, DABT April 21, 2013
 Achieving Regulatory Success: Areas of focus for biotechnology companies Michael J. Schlosser, PhD, DABT April 21, 2013 Regulatory Success Outline Regulatory Initiatives Regulatory Science Pre-Regulatory
Achieving Regulatory Success: Areas of focus for biotechnology companies Michael J. Schlosser, PhD, DABT April 21, 2013 Regulatory Success Outline Regulatory Initiatives Regulatory Science Pre-Regulatory
ICH Topic E 2 A Clinical Safety Data Management: Definitions and Standards for Expedited Reporting. Step 5
 European Medicines Agency June 1995 CPMP/ICH/377/95 ICH Topic E 2 A Clinical Safety Data Management: Definitions and Standards for Expedited Reporting Step 5 NOTE FOR GUIDANCE ON CLINICAL SAFETY DATA MANAGEMENT:
European Medicines Agency June 1995 CPMP/ICH/377/95 ICH Topic E 2 A Clinical Safety Data Management: Definitions and Standards for Expedited Reporting Step 5 NOTE FOR GUIDANCE ON CLINICAL SAFETY DATA MANAGEMENT:
MedDRA in clinical trials industry perspective
 MedDRA in clinical trials industry SFDA ICH MedDRA Workshop, Beijing, 13 14 May 2011 Christina Winter, MD, FFPM Medical director GlaxoSmithKline R&D / ICH MedDRA Management Board 2011 ICH International
MedDRA in clinical trials industry SFDA ICH MedDRA Workshop, Beijing, 13 14 May 2011 Christina Winter, MD, FFPM Medical director GlaxoSmithKline R&D / ICH MedDRA Management Board 2011 ICH International
Bile Duct Diseases and Problems
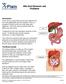 Bile Duct Diseases and Problems Introduction A bile duct is a tube that carries bile between the liver and gallbladder and the intestine. Bile is a substance made by the liver that helps with digestion.
Bile Duct Diseases and Problems Introduction A bile duct is a tube that carries bile between the liver and gallbladder and the intestine. Bile is a substance made by the liver that helps with digestion.
Presents: Insider tips for the life insurance medical exam. Know what they are testing for--and how to get the best results.
 Presents: Insider tips for the life insurance medical exam. Know what they are testing for--and how to get the best results. Copyright 2012 TheLifeInsuranceInsider.com The material Information presented
Presents: Insider tips for the life insurance medical exam. Know what they are testing for--and how to get the best results. Copyright 2012 TheLifeInsuranceInsider.com The material Information presented
Top Ten Things to Know About Stem Cell Treatments
 Top Ten Things to Know About Stem Cell Treatments Many clinics that are offering stem cell treatments make claims about what stem cells can and cannot do that are not supported by our understanding of
Top Ten Things to Know About Stem Cell Treatments Many clinics that are offering stem cell treatments make claims about what stem cells can and cannot do that are not supported by our understanding of
SUPPLEMENTARY MATERIALS. Rivaroxaban for Stroke Prevention in East Asian Patients from the ROCKET AF Trial
 1 SUPPLEMENTARY MATERIALS Rivaroxaban for Stroke Prevention in East Asian Patients from the ROCKET AF Trial Professor Ka Sing Lawrence Wong on behalf of The Executive Steering Committee and the ROCKET
1 SUPPLEMENTARY MATERIALS Rivaroxaban for Stroke Prevention in East Asian Patients from the ROCKET AF Trial Professor Ka Sing Lawrence Wong on behalf of The Executive Steering Committee and the ROCKET
