Botulinum toxin for the treatment of headache: a promising path on a dead end road?
|
|
|
- Elvin Sullivan
- 10 years ago
- Views:
Transcription
1 Acta Neurol. Belg., 2010, 110, Review article Botulinum toxin for the treatment of headache: a promising path on a dead end road? Stephanie DELSTANCHE and Jean SCHOENEN Headache Research Unit. Department of Neurology and GIGA-Neurosciences. Liège University. Abstract Available preventive treatments for primary headaches such as migraines and tension-type headaches have limited efficacy and often disabling side effects (Schoenen 2004, Schoenen 2000). There is thus room for new more effective and better tolerated therapeutic approaches as long as they can be proven superior to placebo. Based on pilot studies and open trials, botulinum toxin (BT) appeared in the headache armamentarium more than a decade ago and it remains widely used in North America since. The initial enthusiasm for BT was not confirmed by subsequent randomized controlled trials reviewed in this article, neither in tension-type headache, nor in episodic migraine and hence BT was considered a dead end road by certain headache experts. A promising path for BT may, however, exist. In two recent trials (PRE- EMPT 1 and 2), OnabotulinumtoxinA (Botox ) was found effective in chronic migraine. The therapeutic gain over placebo is modest (± 11%), but chronic migraine is most disabling, often drug resistant and a serious public health problem, as it affects 1-2% of people in the general population. Because the PREEMPT trials leave unsolved a number of clinically relevant questions, OnabotulinumtoxinA cannot become yet the pre-emptor of CM treatment. Although the path is promising, it seems wise, at this stage, to restrict its use to specialized headache centres where BT can be included in a multidisciplinary approach for chronic headache patients. Key words: Botulinum toxin, OnabotulinumtoxinA, migraine, chronic migraine, chronic tension-type headache Introduction Since more than a decade, Botulinum Toxin (BT) injections have proven therapeutic efficacy in neurologic disorders like dystonias. Their application to headache therapy stems from case reports of pain improvement after cosmetic use. Consequently, on the sole basis of some positive open-label trials, BT became a most popular (and profitable) therapy for headache patients in North America. The initial enthusiasm was not confirmed by subsequent randomized controlled trials (RCT) neither in migraine nor in tension-type headache which led certain headache experts (Evers and Olesen 2006) to consider that BT in headache therapy was a dead end road. There was evidence, however, from subanalyses of certain RCT and from clinical experience that subgroups of patients might benefit from BT, in particular those suffering from chronic migraine, the most disabling form of this disorder. Two recent large RCT (PRE- EMPT 1 and 2) confirm that OnabotulinumtoxinA (Botox ) is effective in chronic migraine patients with or without acute medication overuse, although its therapeutic gain over placebo is modest (Aurora et al., 2010, Diener et al., 2010). This could thus be a promising path for BT in migraine therapy, but a number of clinically relevant questions remain unsolved. In this article, we have done a critical review of the most published RCT with BT in headache. Before getting to the clinical data, it must be reminded that there are several commercial preparations of BT known to interfere at the neuromuscular junction with the molecular mechanisms of storage and release of acetylcholine. There are several subtypes of BT (A, B, C). BT A is the one most frequently used in medical applications. There are also various preparations of Botulinum toxin type-a (BT A), which have recently be renamed: OnabotulinumtoxinA (Botox ) and AbobotulinumtoxinA (Dysport ). BT acts on the SNARE complex that is involved in vesicular transmitter release at the nerve ending. BT A splits the protein SNAP-25 and acts also on synaptobrevin. The result is muscle relaxation and weakness by reduction of acetylcholine release. Most of the studies were realized with botulinum toxin marketed by Allergan under the name of BOTOX. In view of the muscle relaxant properties of BT, it is not surprising that it was first thought to have therapeutic potentials in tension-type
2 222 L. DELSTANCHE & J. SCHOENEN headache where myofascial factors may play a role. After some promising open-label studies, large multicenter RCT did not confirm the initial enthusiasm and migraineurs became a target for BT the more so that it was shown that the toxin could have analgesic properties in a rat model of inflammatory pain and was able to inhibit glutamate and CGRP release by nociceptive afferents in superficial laminae of the spinal cord dorsal horns (Cui et al., 2004, Meng et al., 2007). We will synoptically review RCT performed with BT in migraine (Mig), in chronic daily headaches (CDH) which chiefly comprise chronic migraine and medication overuse headache, and in chronic tension-type headaches (CTTH). The results of placebo-controlled trials are summarized in tables 1-3. Except one, all studies were performed with OnabotulinumtoxinA (Botox - Allergan Inc). In all studies a parallel group design was used with a BT arm and a saline arm. Chronic tension-type headaches. The chronic form of tension-type headache is characterized by the occurrence of 15 days of headache per month. The headache has no migraine features. As mentioned above, some open-label studies suggested that BT could be effective in CTTH. Most of these studies were published in sponsored journal supplements and thus not peer-reviewed. This was the case for two small sample trials that showed positive results for BT in CTTH (Smuts et al., 1999, Relja and Telarovic s 2004). All subsequent RCT of BT in CTTH found no significant difference between BT and placebo (Table 1). Three negative studies were performed with abobotulinumtoxina (Dysport ) (Rollnick et al., 2000, Schulte-Mattler et al., 2004, Straube et al., 2008). The study by Rollnick et al., (2000) is important because it shows that BT does not improve CTTH in spite of a clear reduction of EMG activity in the injected pericranial muscles. Schulte-Mattler et al s study (2004) included 112 patients who, after an observational baseline phase of 1 month, were treated once with BT injections in multiple fixed sites (total dose 500 U) or placebo and followed for 3 months. The area under the curve (AUC) (duration severity of headache) was analyzed, but also the number of headache days, of days with acute drug treatments and headache severity. The results showed no significant difference between BT (-8% AUC) and placebo (- 4%). Also, the comparison of 2 subgroups of patients differing by the degree of pericranial palpation tenderness disclosed no significant difference between the 2 treatment arms. In the last RCT performed with Dysport (Straube et al., 2008) 118 patients were included in the intention-to-treat analysis and injections of a total dose of 420U or 210U were performed at multiple head and neck sites. There was no difference between Dysport and placebo for the primary outcome measure: change in the number of headache-free days at 4-8 weeks after injection compared with 4 weeks before injection. Treatment with 420 units was, however, associated with significant improvements compared with placebo for two secondary efficacy parameters: mean change in headache duration from baseline to weeks 8-12 (P < 0.05) and improved global physician and patient assessment scores (P < 0.05). A large multicentre study included 279 patients randomized into 6 groups. 3 groups received respectively 50, 100 and 150 units of Botox into 5 muscles; 2 other groups received 86 or 100 units of Botox into 3 muscles and placebo in the 2 others; the last group had placebo injections in all 5 muscles (Silberstein et al., 2006). The results for all efficacy measures were negative. For the primary outcome parameter, number of headache-free days, results were even significantly better for placebo compared to 150 U BT (Plac.+4.5 vs BT +2.8). In two other RCT (Schmitt et al., 2001, Padberg et al., 2004), outcome was not significantly different between BT and placebo. By contrast, Kokoska et al., (2004) in a study of 40 patients suffering from frontal tension-type headaches, found that an injection of 50 U BT in frontal muscles significantly decreased headache intensity compared to placebo, but had no effect on headache frequency. Episodic migraine. The first studies of BT in migraine prevention were carried out in patients suffering from 2 to 8 headache episodes per month. In the first placebo-controlled study the results were surprising in so far that there was a significant decrease in the number of migraine episodes with 25U administered as multiple, fixed, pericranial injections, but not with the 75U dose (Silberstein et al., 2000) (Table 2). The study by Barrientos et Chana (2003) is limited to 30 subjects, half of them injected with 50U of BT, the other half with placebo. After a 3 months followup, there was a significant reduction of monthly migraine attacks in the BT group (BT vs Plac ) and of acute anti-migraine drug intake (BT 1.73 vs Plac. 5.60). The authors conclude therefore that BT is efficient in migraine prevention. This conclusion must be taken with reservation because of the small sample size and the quasi absence of placebo effect.
3 RCT and authors BOTULINUM TOXIN FOR THE TREATMENT OF HEADACHE: A PROMISING PATH ON A DEAD END ROAD? 223 Table 1 Randomized, double-blind, placebo-controlled studies about chronic tension-type headache. Clinical parameters and number of patients Doses and injection sites Rollnick et al CTTH + ETC N = U (Dysport ) (1 ) Followup 3 months Multiple fixed sites Schmitt et al CTTH N = U (Botox ) (1 ) Follow-up 2 months Forehead and temples Schulte-Mattler et al CTTH N = U (Dysport ) (1 ) Follow up 3 months Multiple fixed sites Results Negative (despite of EMG activity in injected muscles) Negative (no significant difference in responders: BT 54% vs plac. 38%) Negative (no significant area under the curve) of Kokoska et al CTTH (frontale) N = U (Botox ) (1 ) Multiple fixed sites Padberg et al CTTH N = 40 1U/Kg max. 100 U (Botox ) (1 ) Follow up 3 months Mode Follow-The-Pain (FTP) Silberstein et al CTTH N = U/100U/150U (Botox ) 5 fixed sites (1 ) OR 86U/100U + Plac fixed sites (1 ) follow up 4 months Straube et al CTTH N = U/210U /saline (Dysport ) 18 fixed sites Positive for intensity (signif for BT) Negative for frequency (no signif difference) Negative (no significant difference for headache intensity: VAS: BT vs Plac +7.1) Negative (Plac > Botox 150U for headache-free days/mth: +4.5 vs 2,8 ) Negative (headache-free days/mth: Plac = 1.93, 420U = 2.60, 210U = 2.87) The study by Evers et al., (2004) is the first negative study published for BT in migraine. This RCT was run for 11 months (1 month baseline, 1 month placebo treatment, followed by a 9 months doubleblind phase with 1 BT injection every 3 months). Sixty patients suffering from migraine since at least 1 year were included and randomized into 3 groups. The first group received 100U BT in frontalis and neck muscles, the second 16U BT in frontalis and placebo in neck muscles, the third one only placebo. The primary outcome measure was the percentage of responders, i.e. patients with at least 50% reduction of monthly migraine attack frequency. Secondary efficacy parameters were decrease of attack frequency per month, number of days with migraine, number of days with moderate or severe migraine, associated symptoms and days with use of acute anti-migraine drugs. Differences between groups were not significant (30% of responders in the 2 BT groups; 25% in the placebo group) except for associated symptoms which were more attenuated in the 16U group. This study does therefore not argue in favour of an effect of BT in migraine prevention, but its statistical power could have been too weak. The large European multicenter study had an observation period of 11 months (Relja et al., 2007). It included 495 patients suffering from at least 3 migraine attacks per month, but less than 15 headache days per month. The mean monthly frequency of attacks before randomization was between 4.3 and 4.7. At the end of the baseline month, patients received a placebo injection to identify placebo responders. After 1 month, placebo responders (n = 173) and non-responders (n = 322) were randomized into 4 groups: placebo, 75 U BT, 150 U BT and 225 U BT injected in 7 predefined pericranial sites. The primary outcome measure was the decrease of monthly migraine attack frequency in placebo responders and non-responders. There were no significant differences between treatment groups for neither parameter, even not after control for age, sex or illness duration. Similar negative results were obtained in two large North American trials (Aurora et al., 2006, Elkind et al., 2006) and in a smaller trial of drug-resistant migraine patients (Cady and Schreiber 2008), although the latter found improvement after BT in disability scales.
4 224 L. DELSTANCHE & J. SCHOENEN Table 2 Randomized, double-blind, placebo-controlled studies in episodic migraine. RCT and, authors Silberstein et al Clinical parameters and number of patients Migraine. (2-8 episodes/month) N = 42 Doses and injection sites 25 U/ 75 U (Botox ) (1 ) Multiple fixed sites Follow up 3 months Barrientos & Chana 2003 Migraine N = U (Botox ) (1 ) Follow up 3 months 6 fixed sites Evers et al Relja et al Migraine (2-8 episodes/month) N = 60 Migraine ( 3 episodes/month) N = 495 (n = 322 placebo nonresponders) 16 U/ 100 U (Botox ) (1 ) fixed sites forehead and neck Follow up 3 months 225 U/ 150 U / 75U (Botox ) 1 /3months during 9 months Multiple fixed sites Results Positive for 25 U ( 38% of days with acute treatments) Negative for 75U Positive (signif. of frequency at day 90: BT vs plac ) Negative (BT: 30% responders; Plac: 25%) Negative (no significant difference between 4 groups after 3 months Aurora et al.2007 Migraine ( 4-15 episodes/month) N = 369 (n = 203 placebo non-responders) U (Botox ) 1 /3months during 9 months Modified protocol Follow The Pain (FTP) Negative (no difference with placebo, except for patients with 12 headaches/month : n = 88) Elkind et al Migraine ( 4-8 episodes /month) N = injections at 4-month intervals (Botox ) - 0U/7.5U/25U/50U - 25U/50U - 0U/25U/50U fixed sites forehead and temples Negative (no difference with placebo; improvement with time in all groups) Cady & Schreiber 2008 Migraine (HIT-6 56) failing oral prophylactic treatment N = 61 (2 BT:1 pl.) 139 U (Botox ) (1 ) Multiple fixed sites Follow up 3 months Negative (no signif. difference for headache frequency or severity Positive Signif. difference of HIT-6 score, MIDAS and MIQ at month 3 in favour of BT One may wonder if the outcome measures used in most RCT, i.e. frequency and intensity of attacks, are sensitive enough to disclose differences between BT and placebo. In a randomized study comparing the preventive anti-migraine effect of valproic acid and BT, both treatments had equal beneficial effects on headache frequency, but BT was clearly superior for quality of life measures and disability (Blumenfeld and Schim 2008). This difference must be verified in placebo-controlled trials again, but it may explain why BT continues to be used by certain practitioners despite the lack of scientific evidence for its efficiency. Duration of illness might influence the response to BT treatment. This is suggested by a prospective, non-controlled study which evaluated 61 migraineurs and found that responders to BT (62%) differed from non-responders (38%) by illness duration (21.9 vs 31.4 years) (Eross et al., 2005). It has finally been suggested that episodic migraine patients with an imploding pattern of headache (external constriction) may respond to BT treatment contrary to those with an exploding (intracranial pressure) pattern (Jakubowski et al., 2006). Chronic daily headache. The chronic daily headache (CDH) group is defined by a headache frequency of at least 15 days per month. It includes different types of primary headaches, but most often chronic migraine with and without medication overuse (Fumal et al., 2006). In the large North-American multicenter study (Mathew et al., 2005) (table 3), 355 patients suffering from CDH were followed during 11 months. The
5 BOTULINUM TOXIN FOR THE TREATMENT OF HEADACHE: A PROMISING PATH ON A DEAD END ROAD? 225 Table 3 Randomized, double-blind, placebo-controlled studies inchronic daily headache (CDH) and chronic migraine (CM). RCT and authors Diagnosis and number of patients Doses and injection sites Ondo et al CDH N = U (Botox ) (1 ) Follow up 3 mths Mode FTP Mathew et al CDH (± 50% with analgesic overuse) N = 355 (n = 279 placebo non-responders) U (Botox ) 1 /3months during 9 months Mode FTP Results Negative (no significant decrease of headaches) Negative (1 param.) (no signif of headache-free days: BT 6,7 vs Plac 5,2) Positive (2 param.) (50% of headaches: BT 32,7% vs Plac 15%) Dodick et al (Mathew et al. sub-analysis) CDH without prophylactic treatments (± 50% with analgesic overuse) N = 228 (out of 355) U (Botox ) 1 /3months during 9 months Mode FTP Positive (better headaches in BT -7,8 vs Plac-4,5; 50% of headache after 3 inj: BT 50% vs Plac 35%) Silberstein et al CDH N = U/150U/75U (Botox ) Negative (1 param.) (no signif of headache-free days at day 180: 6/7,9/7,9/8 for BT225/BT150/BT75/Plac) Positive (2 param.) (signif headaches at day 240 for BT225-8,4 ; BT150-8,6 vs plac 6,4) Freitag et al Aurora et al (PREEMPT 1) Diener et al (PREEMPT 2) CM (without analgesic overuse) N = 36 CM N = U (Botox ) (1 ) Follow up 4 mois Multiple fixed sites 155U (up to 195U) (Botox ) (N = 341); placebo (N = 338) 31 fixed sites (5U/site) every 12 weeks 4 week baseline/ 24-week double blind/ 32-week open-label CM N = 705 Same protocol BT (N = 347); placebo (N = 358) Positive ( of headache: BT 31% vs Plac 8,1%; responders 33% vs 16.7%) Negative 1 endpoint Headache episodes : no difference -0.5%) Positive 2 endpoints Significant BT effect for headache days (-6.7%), migraine days (-7.9%), headache hours (-10.4%), HIT-6 score (- 3.4%) Positive 1 endpoint Headache days (-11%) (p < 0.001) Positive 2 endpoints Migraine days (-11.3%), headache episodes (-8%), headache hours (-13.4), HIT-6 score (-3.8) first period of 1 month was a baseline observational period. Thereafter, 279 subjects who did not improve during baseline, were included in the randomised phase. They received 3 BT injections separated by 3 months at a dose ranging from 105 U to 260 U or placebo in a follow the pain pattern meaning that the injections are made in tender points identified by palpation of pericranial and neck muscles. The primary outcome measure was the number of headache-free days per month. Secondary outcome measures were 50% decrease from baseline of headache days, number of acute headache medication used per month and quality of life. The primary endpoint was not reached. Indeed, there was no significant increase of headache free-days after BT injections (+6.7) by comparison with placebo (+5.2). The percentage of patients with a 50% decrease of headache days was, however, superior in the BT group (32.7% vs 15%). The authors speculated that the lack of efficacy of BT might be due to an overuse of acute headache medications and/or interference with concomitant preventive anti-migraine drugs. It must be pointed out that more than 50% of patients included in the BT arm overused analgesics com-
6 226 L. DELSTANCHE & J. SCHOENEN pared to ± 40% in the placebo arm. As it is well known that interruption of excessive analgesic intake can by itself improve headache frequency, the bias due to medication overuse, if any, would have been in favour of BT. To verify the possible interaction with preventive anti-migraine treatments, Dodick et al., (2005) analyzed a subgroup of patients coming from the Mathew et al., s study, i.e. those who did not receive any prophylactic treatment (n = 228/355). In this subgroup, BT induced a significant decrease in headache frequency with an improvement of 50% in 50% of subjects from day 150 onwards, i.e. after the 3 rd injection compared to 30% in placebo-treated patients. A decrease in headache severity was evident from the second BT injection onwards. Improvement in quality of life was, however, not superior in the BT group. Two other smaller studies are of interest (Table 3). The study by Ondo et al., (2004) also failed to show a beneficial effect of BT. Freitag et al., (2007) studied 36 chronic migraine patients without analgesic overuse and found evidence for the efficacy of BT. Because of the small sample size, this study cannot be taken as convincing evidence for the use of BT in chronic migraine. Chronic migraine (the PREEMPT trials) In 2006, broader diagnostic criteria for chronic migraine (CM) were proposed by an expert group of the International Headache Classification committee (CM-R A 1.5.1) (Olesen et al., 2006). This facilitated the launch of 2 large multicentre studies of OnabotulinumtoxinA in patients diagnosed with chronic migraine, the PREEMPT trials. As shown in table 3, both studies undoubtedly do support efficacy in the patients studied and have demonstrated undisputable strengths. They were well-designed, performed by experienced investigators and included large numbers of patients with a long blinded follow-up of 24 weeks and a subsequent open label period of 32 weeks. OnabotulinumtoxinA was statistically superior to placebo in a range of outcome measures, including quality of life and disability scales. These results appear encouraging, especially since they concern a most disabled patient population. They were confirmed by the pooled data analysis of PRE- EMPT 1 and 2 (Dodick et al., 2010) which also confirmed the excellent tolerability and safety of BT treatment. Adverse events, most of them mild to moderate in severity, occurred in 62.4% of onabotulinumtoxina patients and 51.7% of placebo patients. Few patients discontinued treatment due to adverse events (onabotulinumtoxina, 3.8%; placebo, 1.2%). No unexpected treatment-related adverse events were identified. The following considerations, however, may temper an excessive optimism. One major concern is that both trials recruited a majority of patients (up to 65%) fulfilling criteria for medication overuse headache (MOH-R A 8.2) (Olesen et al., 2006). Although this reflects real world life, it means that many patients may have been suffering from a secondary headache disorder and not, as stated in titles and methods section, from CM per se. Detoxification suffices in many patients to convert the chronic headache pattern into an episodic one and/or ineffective preventives to effective drugs. This may explain part of the beneficial effect, admittedly both in BT and placebo groups. Both groups had indeed an average decrease of ± 10 acute medication intakes at week 24, but there was a significantly greater decrease of triptan intake in OnabotulinumtoxinA-treated patients. It would have been informative to know whether the treatment response differed between patients with and without medication overuse. Along the same line of patient selection, it comes as a surprise that up to 40% of enrolled patients never received a preventive treatment before, the more so as mean duration of CM in both trials was 20 years and mean age of participants ± 40 years! Although the reliability of the historical data on migraine pattern and previous treatments may be questioned, knowing if prophylaxis-naive patients respond differently is of practical interest. Unfortunately, this information is not provided. A frequent question in RCTs concerns the clinical significance of statistical differences. There is no consensus on the minimal change in an outcome measure required to be considered clinically relevant and this minimum has to be balanced against the severity of the disorder. Because of the well-known high placebo response in headache trials using injections of drugs, in particular, of BT, the therapeutic gain of OnabotulinumtoxinA over placebo is not impressive in numerical terms: absolute gain of 6.7% and 11% for headache days and 7.9% and 11.3% for migraine days, respectively in PREEMPT 1 and 2 (see Table 3). Looking at the absolute mean change in headache days, there is little doubt, however, that a substantial number of migraine patients switched from a chronic to an episodic pattern. According to data from the American Migraine Prevalence and Prevention Study, conversion from CM to EM occurs over a 3- year period in 26% of patients, whereas CM persists unchanged in only 22% (Manack et al., 2009). Unfortunately, the percentage of patients who converted and the difference between treatment arms are not
7 BOTULINUM TOXIN FOR THE TREATMENT OF HEADACHE: A PROMISING PATH ON A DEAD END ROAD? 227 given in the PREEMPT articles, nor are the respective percentages of responders with a 50% or 25% of reduction in headache days, so that a comparison with the natural history of the disorder is not possible. These missing data, as those on the proportion of patients who would choose to continue the treatment, would have been useful to further estimate the clinical relevance of the results. The only other RCT performed in CM has studied topiramate (Silberstein et al., 2009) and it is somewhat surprising that the PREEMPT authors, several of whom also participated in the topiramate trial, do not compare the results in the discussion. Although preventive treatment-naive patients were excluded in the topiramate trial contrary to PREEMPT 1 and 2, the effect size in the three studies seems to be similar, e.g. 10.2% absolute gain over placebo for migraine days in the topiramate trial compared to 11.3% in PREEMPT 2. OnabotulinumtoxinA was directly compared to topiramate in a small randomized double-blind single-centre trial (Mathew and Jaffri 2009). No statistical difference was found between the 2 treatment groups, but the results are not based on an intent-to-treat analysis. Adverse events, however, were more numerous and severe for topiramate. The only question that really matters to patients and doctors is whether the PREEMPT 1-2 results are convincing enough to justify our present or change our future medical practice. The answer could be Yes with some reservations. OnabotulinumtoxinA was proven to be moderately superior to placebo, and probably to natural outcome, in a population of chronic migraine patients of whom the majority was overusing acute medication and a substantial proportion had never received preventive drugs. This is good news as CM is a most disabling disorder with a 1-2% prevalence in the general population in which even a small effect or an effect in a small percentage of patients can be of medical value. A major advantage of OnabotulinumtoxinA compared to other drug treatments is its excellent tolerance. Before it can become the pre-emptor of CM treatment, however, the abovementioned questions need to be addressed. The mechanisms by which BT improves CM are speculative. As central sensitization in the trigeminal sensory systems may play a role in migraine chronification, one may hypothesize that this might be reduced by BT thanks to its capacity to attenuate glutamate and CGRP release by trigeminal afferents (Cui et al., 2004; Kitamura et al., 2009). Phase IV studies should be able to provide answers to crucial open questions like How to identify responders? or Is the treatment cost-effective? For translational benefits, they should be paralleled by more research on the mode of action of OnabotulinumtoxinA in chronic migraine, which still remains a mystery. Tolerance and safety of use. All mentioned studies have assessed tolerance and safety of BT. The conclusion is that BT causes only few, non-disabling and transient side effects. As with its use in other disorders, the most frequent ones are muscular weakness, ptosis and neck pain. Because of the motor side effects, unblinding may be a problem in RCT of BT. Overall, tolerance seems to be superior to that of the most effective preventive antimigraine drugs such as the anticonvulsants valproate and topiramate. Conclusions Randomized placebo-controlled trials in primary headaches indicate convincingly that BT has no efficacy in chronic tension-type headache or in episodic migraine without aura. These disappointing results of BT treatment may have several explanations. First, it is clear that in CTTH, pericranial muscle activity which is supposed to be reduced by the BT, does not play a major pathophysiological role contrary to central sensitization and dysfunction of endogenous pain control systems (see review by Schoenen 2005). Episodic migraine attacks, on the other hand, are associated with activation of the trigemino-vascular system, the visceral portion of the 1st division of the trigeminal nerve (V1), and with a transient and reversible sensitization of the 1 st and 2nd order central trigeminal nociceptors. Subcutaneous injections of BT, by contrast, act on the somatic portion of V1. If BT is able to decrease the release of transmitters by somatic trigeminal afferents at the level of the spinal dorsal horns (Cui et al., 2004), it might reduce cutaneous allodynia which accompanies up to 30% of migraine attacks without preventing the attacks themselves. To our knowledge this has not been investigated up to now. Contrasting with the studies in CTTH and episodic migraine, those performed with BT in chronic migraine have provided evidence of efficacy. The evidence comes from subanalysis of chronic daily headache trials and, more convincingly, from the two recent PREEMPT trials. The Pro s and con s of these trials have been discussed. It seems of uttermost importance in future trials to develop methods which are able to better identify patients who may respond to BT. Taken together, OnabotulinumtoxinA (Botox ) cannot be considered yet as a standard treatment for
8 228 L. DELSTANCHE & J. SCHOENEN CM. it seems wise at this stage to restrict its use to specialized headache centres where it can be included in the multidisciplinary armamentarium recommended in chronic headache patients and where the indispensable supplementary studies on its target patient population, basic mechanisms of action and pharmaco-economic profile can be conducted. Thus, after several negative studies forecasting a dead end road, the CM path for BT is promising, but needs to be more clearly delineated and clarified in clinical practice, before anyone can take it with confidence. Acknowledgments The authors are grateful to Drs L Herroelen, P Louis, K Paemeleire, M Vandenheede and B Vandersmissen from the Belgian Headache Society for helpful discussions concerning the PREEMPT trial results. REFERENCES Aurora SK, Dodick DW, Turkel CC, DeGryse RE, Silberstein SD, et al. PREEMPT 1 Chronic Migraine Study Group - OnabotulinumtoxinA for treatment of chronic migraine: results from the double-blind, randomized, placebo-controlled phase of the PRE- EMPT 1 trial. Cephalalgia. 2010;30(7): Aurora SK, Gawel M, Brandes JL, Pokta S, VanDenburgh AM, for the BOTOX North American Episodic Migraine Study Group Botulinum Toxin type A Prophylactic Treatment of Episodic Migraine: A randomized, Double-blind, Placebo- Controlled Exploratory Study. Headache. 2007; 47(4): Barrientos N, Chana P. Botulinum toxin type A in prophylactic treatment of migraine headaches: a preliminary study. J Headache Pain. 2003;4(3): Blumenfeld A, Schim J. Botulinum toxin A compared with divalproex for the prophylactic treatment of migraine: a 9 month randomized, double-blind trial. Headache. 2008;48(2): Cady R, Schreiber C. Botulinum toxin type A as migraine preventive treatment in patients previously failing oral prophylactic treatment due to compliance issues. Headache. 2008;48(6): Cui M, Khanijou S, Rubino J, Aoki KR. Subcutaneous administration of botulinum toxin A reduces formalin-induced pain. Pain, 2004;107(1-2): Diener HC, Dodick DW, Aurora SK, Turkel CC, DeGryse RE, et al. PREEMPT 2 Chronic Migraine Study Group - OnabotulinumtoxinA for treatment of chronic migraine: results from the double-blind, randomized, placebo-controlled phase of the PRE- EMPT 2 trial. Cephalalgia. 2010;30(7): Dodick DW, Mauskop A, Elkind AH, DeGryse R, Brin MF, et al. BOTOX CDH Study Group. Botulinum toxin type A for the prophylaxis of chronic daily headache: subgroup analysis of patients not receiving other prophylactic medications: a randomized, double-blind, placebo-controlled study. Headache. 2005;45(4): Dodick DW, Turkel CC, DeGryse RE, Aurora SK, Silberstein SD, et al. PREEMPT Chronic Migraine Study Group - OnabotulinumtoxinA for treatment of chronic migraine: pooled results from the double-blind, randomized, placebo-controlled phases of the PREEMPT clinical program. Headache, 2010;50(6): Elkind HA, O Carroll P, Blumenfeld A, DeGryse R, Dimitrova R, for the BONTA Study Group. A series of three sequential, randomized, controlled studies of repeated treatments with botulinum toxin type A for migraine prophylaxis. J Pain. 2006;7(10): Eross EJ, Gladstone JP, Lewis S, Rogers R, Dodick DW. Duration of migraine is a predictor for response to botulinum toxin A. Headache, 2005;45(4): Evers S, Vollmer-Haase, Schwaag S, Rahmann A, Husstedt IW, et al. Botulinum toxin A in the prophylactic treatment of migraine- a randomized, double-blind, placebo-controlled study. Cephalalgia. 2004;24(10): Evers S, Olesen J. Botulinum toxin in headache treatment: the end of the road? Cephalalgia. 2006;26(7): Freitag FG, Diamond S, Diamond M, Urban G. Botulinum Toxin Type A in the Treatment of Chronic Migraine without Medication Overuse Headache. Headache. 2008;48(2): Fumal A, Magis D, Schoenen J. Les céphalées par abus d antalgiques. Rev Med Liège, 2006;61(4): Jakubowski M, McAllister PJ, Bajwa ZH, Ward TN, Smith P, et al. Exploding vs. imploding headache in migraine prophylaxis with Botulinum Toxin A. Pain. 2006;125(3): Kitamura Y, Matsuka Y, Spigelman I, Ishihara Y, Yamamoto Y, et al. Botulinum toxin type a (150 kda) decreases exaggerated neurotransmitter release from trigeminal ganglion neurons and relieves neuropathy behaviors induced by infraorbital nerve constriction. Neuroscience. 2009;10;159(4): Kokoska MS, Glaser DA, Burch CM, Hollenbeak CS. Botulinum toxin injections for the treatment of frontal tension headache. J Headache Pain. 2004; 5(2): Manack A, Turkel C, Silberstein S. The evolution of chronic migraine: classification and nomenclature. Headache. 2009;49(8): Mathew N, Frishberg B, Gawel M, Dimitrova R, Gibson J, et al. BOTOX CDH Study Group. Botulinum Toxin Type A (BOTOX) for the prophylactic treatment of chronic daily headache: a randomized, doubleblind, placebo-controlled trial. Headache. 2005;45(4):
9 BOTULINUM TOXIN FOR THE TREATMENT OF HEADACHE: A PROMISING PATH ON A DEAD END ROAD? 229 Mathew NT, Kailasam J, Meadors L. Predictors of Response to Botulinum Toxin Type A (BoNTA) in Chronic Daily Headache. Headache. 2008;48(2): Mathew NT, Jaffri SFA. A Double-Blind Comparison of OnabotulinumtoxinA (BOTOX ) and Topiramate (TOPAMAX ) for the Prophylactic Treatment of Chronic Migraine: A Pilot Study. Headache. 2009; 49(10): Meng J, Wang J, Lawrence G, Dolly JO. Synaptobrevin I mediates exocytosis of CGRP from sensory neurons and inhibition by botulinum toxins reflects their anti-nociceptive potential. J Cell Sci. 2007:15; 120(Pt 16): Ondo WG, Vuong KD, Derman HS. Botulinum toxin A for chronic daily headache: a randomized, placebocontrolled, parallel design study. Cephalalgia, 2004;24(1):60-5. Olesen J., Bousser M-G., Diener H-C., Dodick D., First M., et al. New appendix criteria open for a broader concept of chronic migraine. Cephalalgia, 2006; 26(6): Padberg M, de Bruijn SFMT, de Haan RJ, Tavy DLJ. Treatment of chronic tension-type headache with botulinum toxin: a double-blind, placebo-controlled clinical trial. Cephalalgia. 2004;24(8): Relja M, Telarovic S. Botulinum toxin in tension-type headache. J Neurol 2004;251 (suppl.1): I/12-I/14. Relja M, Poole AC, Schoenen J, Pascual J, Lei X, et al. For the European BoNTA Headache Study Group. A multicenter, double-blind, randomized, placebocontrolled, parallel group study of multiple treatments of botulinum toxin type A (BoNTA) for the prophylaxis of episodic migraine headaches. Céphalalgia. 2007;27(6): Rollnick JD, Tanneberger O, Schubert M, Schneider U, Dengler R. Treatment of tension-type headache with botulinum toxin type A: a double blind placebo controlled study. Headache. 2000;40(4): Schmitt WJ, Slowey E, Fravi N, Weber S, Burgunder J- M. Effect of botulinum toxin A injections in the treatment of chronic tension-type headache: a double -blind, placebo-controlled trial. Headache. 2001; 41(7): Schoenen J. Tension-type Headache, in MacMahon S & Koltzenburg M Eds., Wall & Melzack s Textbook of Pain. 5th Edition. Elsevier, Amsterdam, 2005, Chap.56. Schoenen J. Tension-type headache, in Diener H.C Ed., Drug Treatment of Migraine and Other Headaches. Monogr Clin Neurosci. Basel, Karger, 2000, vol 17, Schoenen J., Di Clemente L., Coppola G. Other prophylactic anti-migraine agents: riboflavin, feverfew, magnesium, Botulinum toxin, and calcium antagonists, in Olesen J, Silberstein SD., Tfelt-Hansen P Eds., Preventive pharmacotherapy of headache disorders. University Press, Oxford, 2004, Schulte-Mattler WJ, Krack P, BoNTTH study group. Treatment of chronic tension-type headache with botulinum toxin A: a randomized, double-blind, placebo-controlled multicenter study. Pain. 2004; 109(1-2): Silberstein SD, Göbel H, Jensen R, Elkind AH, DeGryse R. et al. Botulinum toxin type A in the prophylactic treatment of chronic tension-type headache: a multicentre, double-blind, randomized, placebo-controlled, parallel-group study. Cephalalgia. 2006; 26(7): Silberstein S, Mathew N, Saper J, Jenkins S. Botulinum toxin type A as a migraine preventive treatment. For the BOTOX Migraine Clinical Research Group. Headache. 2000;40(6): Silberstein SD, Stark SR, Lucas SM, Christie SN, Degryse RE, et al.; BoNTA-039 Study Group. Botulinum toxin type A for the prophylactic treatment of chronic daily headache: a randomized, doubleblind, placebo-controlled trial. Mayo Clin Proc. 2005;80(9): Silberstein S, Lipton R, Dodick D, Freitag F, Mathew N, et al. Topiramate treatment of chronic migraine: a randomized, placebo-controlled trial of quality of life and other efficacy measures. Headache. 2009; 49(8): Smuts JA, Baker MK, Smuts HM, Rheta Stassen JM, Rossouw E, et al. Prophylactic treatment of chronic tension-type headache using botulinum toxin type A. Eur J Neurol. 1999;6 (suppl 4):S99-S102. Straube A, Empl M, Ceballos-Baumann A, Tölle T, Stefenelli U, et al. Dysport Tension-Type Headache Study Group. - Pericranial injection of botulinum toxin type A (Dysport) for tension-type headache - a multicentre, double-blind, randomized, placebocontrolled study. Eur J Neurol. 2008;15(3):
Onabotulinumtoxin in the Preventive Treatment of Chronic Migraine and Other Headaches
 Onabotulinumtoxin in the Preventive Treatment of Chronic Migraine and Other Headaches While BoNTA shows promise in the management of chronic migraine, optimal outcomes will be acheived when clinicians
Onabotulinumtoxin in the Preventive Treatment of Chronic Migraine and Other Headaches While BoNTA shows promise in the management of chronic migraine, optimal outcomes will be acheived when clinicians
PROCEEDINGS BOTULINUM TOXIN: PARADIGMS FOR TREATMENT * Benjamin M. Frishberg, MD ABSTRACT
 BOTULINUM TOIN: PARADIGMS FOR TREATMENT Benjamin M. Frishberg, MD ABSTRACT Botulinum toxin type A has been studied for the treatment of headache disorders since the early 1990s when its usefulness for
BOTULINUM TOIN: PARADIGMS FOR TREATMENT Benjamin M. Frishberg, MD ABSTRACT Botulinum toxin type A has been studied for the treatment of headache disorders since the early 1990s when its usefulness for
Treatment of Tension-Type Headache with Botox:
 Treatment of Tension-Type Headache with Botox: A Review of the Literature DIMA ROZEN, M.D. 1, AND JOYTI SHARMA, M.D. 2 Abstract Botulinum toxin has been shown to effectively treat several types of neurological
Treatment of Tension-Type Headache with Botox: A Review of the Literature DIMA ROZEN, M.D. 1, AND JOYTI SHARMA, M.D. 2 Abstract Botulinum toxin has been shown to effectively treat several types of neurological
Neurotoxins and headache: A review of the evidence. Alan G Finkel MD FAHS FAAN Carolina Headache Institute University of North Carolina
 Neurotoxins and headache: A review of the evidence Alan G Finkel MD FAHS FAAN Carolina Headache Institute University of North Carolina Disclosures Allergan: Trainer 2010 present Glaxo Smith Kline: Speaker
Neurotoxins and headache: A review of the evidence Alan G Finkel MD FAHS FAAN Carolina Headache Institute University of North Carolina Disclosures Allergan: Trainer 2010 present Glaxo Smith Kline: Speaker
Botulinum Toxin: Could It Be An Effective Treatment For Chronic Tension Type Headache?
 Sherif M. Hamdy et al. Botulinum Toxin: Could It Be An Effective Treatment For Chronic Tension Type Headache? Sherif M. Hamdy 1, Hatem Samir 1, M. El-Sayed 1, Nermin Adel 1, Rasha Hassan 2 Department of
Sherif M. Hamdy et al. Botulinum Toxin: Could It Be An Effective Treatment For Chronic Tension Type Headache? Sherif M. Hamdy 1, Hatem Samir 1, M. El-Sayed 1, Nermin Adel 1, Rasha Hassan 2 Department of
WAT ALS HOOFDPIJN CHRONISCH WORDT? Prof Jan Versijpt, MD PhD Kliniek voor Hoofd- en Aangezichtspijn Neurologie, UZ Brussel
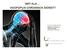 WAT ALS HOOFDPIJN CHRONISCH WORDT? Prof Jan Versijpt, MD PhD Kliniek voor Hoofd- en Aangezichtspijn Neurologie, UZ Brussel Stovner, IHC 2009 New daily persistent headache Primair = dagelijkse and continue
WAT ALS HOOFDPIJN CHRONISCH WORDT? Prof Jan Versijpt, MD PhD Kliniek voor Hoofd- en Aangezichtspijn Neurologie, UZ Brussel Stovner, IHC 2009 New daily persistent headache Primair = dagelijkse and continue
Tension Type Headaches
 Tension Type Headaches Research Review by : Dr. Ian MacIntyre Physiotherapy for tension-type Headache: A Controlled Study P. Torelli, R. Jenson, J. Olsen: Cephalalgia, 2004, 24, 29-36 Tension-type headache
Tension Type Headaches Research Review by : Dr. Ian MacIntyre Physiotherapy for tension-type Headache: A Controlled Study P. Torelli, R. Jenson, J. Olsen: Cephalalgia, 2004, 24, 29-36 Tension-type headache
Botulinum toxin in the treatment of chronic migraine. Gregory P. Hanes, MD Neuroscience Summit 5/14/15
 Botulinum toxin in the treatment of chronic migraine Gregory P. Hanes, MD Neuroscience Summit 5/14/15 Primary Headache Disorders: Frequency Classification After Secondary Causes Are Ruled Out Short-Duration
Botulinum toxin in the treatment of chronic migraine Gregory P. Hanes, MD Neuroscience Summit 5/14/15 Primary Headache Disorders: Frequency Classification After Secondary Causes Are Ruled Out Short-Duration
botulinum toxin type A, 50 unit, 100 unit and 200 unit powder for solution for injection (Botox ) SMC No. (692/11) Allergan Ltd
 Resubmission botulinum toxin type A, 50 unit, 100 unit and 200 unit powder for solution for injection (Botox ) SMC No. (692/11) Allergan Ltd 08 March 2013 The Scottish Medicines Consortium (SMC) has completed
Resubmission botulinum toxin type A, 50 unit, 100 unit and 200 unit powder for solution for injection (Botox ) SMC No. (692/11) Allergan Ltd 08 March 2013 The Scottish Medicines Consortium (SMC) has completed
New appendix criteria open for a broader concept of chronic migraine
 Blackwell Publishing LtdOxford, UKCHACephalalgia0333-1024Blackwell Science, 20062006266742746Original ArticleA broader concept of chronic migrainej Olesen et al. BRIEF REPORT New appendix criteria open
Blackwell Publishing LtdOxford, UKCHACephalalgia0333-1024Blackwell Science, 20062006266742746Original ArticleA broader concept of chronic migrainej Olesen et al. BRIEF REPORT New appendix criteria open
How Is OnabotulinumtoxinA Reimbursed For Chronic Migraine? Impact Of FDA Approval And The New CPT Code
 How Is OnabotulinumtoxinA Reimbursed For Chronic Migraine? Impact Of FDA Approval And The New CPT Code Effective January 1, 2013, physicians will be able to report the new CPT code 64615 when performing
How Is OnabotulinumtoxinA Reimbursed For Chronic Migraine? Impact Of FDA Approval And The New CPT Code Effective January 1, 2013, physicians will be able to report the new CPT code 64615 when performing
33 % of whiplash patients develop. headaches originating from the upper. cervical spine
 33 % of whiplash patients develop headaches originating from the upper cervical spine - Dr Nikolai Bogduk Spine, 1995 1 Physical Treatments for Headache: A Structured Review Headache: The Journal of Head
33 % of whiplash patients develop headaches originating from the upper cervical spine - Dr Nikolai Bogduk Spine, 1995 1 Physical Treatments for Headache: A Structured Review Headache: The Journal of Head
BOTOX Injection (Onabotulinumtoxin A) for Migraine Headaches [Preauthorization Required]
![BOTOX Injection (Onabotulinumtoxin A) for Migraine Headaches [Preauthorization Required] BOTOX Injection (Onabotulinumtoxin A) for Migraine Headaches [Preauthorization Required]](/thumbs/29/13417512.jpg) BOTOX Injection (Onabotulinumtoxin A) for Migraine Headaches [Preauthorization Required] Medical Policy: MP-RX-01-11 Original Effective Date: March 24, 2011 Reviewed: Revised: This policy applies to products
BOTOX Injection (Onabotulinumtoxin A) for Migraine Headaches [Preauthorization Required] Medical Policy: MP-RX-01-11 Original Effective Date: March 24, 2011 Reviewed: Revised: This policy applies to products
Frequent headache is defined as headaches 15 days/month and daily. Course of Frequent/Daily Headache in the General Population and in Medical Practice
 DISEASE STATE REVIEW Course of Frequent/Daily Headache in the General Population and in Medical Practice Egilius L.H. Spierings, MD, PhD, Willem K.P. Mutsaerts, MSc Department of Neurology, Brigham and
DISEASE STATE REVIEW Course of Frequent/Daily Headache in the General Population and in Medical Practice Egilius L.H. Spierings, MD, PhD, Willem K.P. Mutsaerts, MSc Department of Neurology, Brigham and
Will Botox help your migraines?
 Will Botox help your migraines? In case you hadn t heard, Botox can now be used and potentially claimed on insurance for migraines. Surprising I know. You re probably wondering how will an injection of
Will Botox help your migraines? In case you hadn t heard, Botox can now be used and potentially claimed on insurance for migraines. Surprising I know. You re probably wondering how will an injection of
Guidelines for controlled trials of prophylactic treatment of chronic migraine in adults
 doi:10.1111/j.1468-2982.2008.01555.x REVIEW Guidelines for controlled trials of prophylactic treatment of chronic migraine in adults S Silberstein 1 (Chairman, USA), P Tfelt-Hansen 2 (Co-Chairman, Denmark),
doi:10.1111/j.1468-2982.2008.01555.x REVIEW Guidelines for controlled trials of prophylactic treatment of chronic migraine in adults S Silberstein 1 (Chairman, USA), P Tfelt-Hansen 2 (Co-Chairman, Denmark),
Natural Modality in the Treatment of Primary Headaches. William S. Mihin, D.C. Catharine Helms, M.S. Michelle M. Anderson, M.S.N., F.N.P.
 Natural Modality in the Treatment of Primary Headaches William S. Mihin, D.C. Catharine Helms, M.S. Michelle M. Anderson, M.S.N., F.N.P. Abstract Headaches are both a prevalent and disabling condition.
Natural Modality in the Treatment of Primary Headaches William S. Mihin, D.C. Catharine Helms, M.S. Michelle M. Anderson, M.S.N., F.N.P. Abstract Headaches are both a prevalent and disabling condition.
Classification of Chronic Headache
 Chronic Headache Classification of Chronic Headache JMAJ 47(3): 112 117, 2004 Mitsunori MORIMATSU Professor, Department of Neurology and Clinical Neuroscience, Yamaguchi University School of Medicine Abstract:
Chronic Headache Classification of Chronic Headache JMAJ 47(3): 112 117, 2004 Mitsunori MORIMATSU Professor, Department of Neurology and Clinical Neuroscience, Yamaguchi University School of Medicine Abstract:
Headaches in Children How to Manage Difficult Headaches
 Headaches in Children How to Manage Difficult Headaches Peter Procopis Childhood headaches Differential diagnosis Migraine Psychological Raised Pressure Childhood headaches Other causes: Constitutional
Headaches in Children How to Manage Difficult Headaches Peter Procopis Childhood headaches Differential diagnosis Migraine Psychological Raised Pressure Childhood headaches Other causes: Constitutional
Guidance for Industry Migraine: Developing Drugs for Acute Treatment
 Guidance for Industry Migraine: Developing Drugs for Acute Treatment DRAFT GUIDANCE This guidance document is being distributed for comment purposes only. Comments and suggestions regarding this draft
Guidance for Industry Migraine: Developing Drugs for Acute Treatment DRAFT GUIDANCE This guidance document is being distributed for comment purposes only. Comments and suggestions regarding this draft
BOTOX Injection (Onabotulinumtoxin A) for Chronic Migraine Headaches
 BOTOX Injection (Onabotulinumtoxin A) for Chronic Migraine Headaches [For the list of services and procedures that need preauthorization, please refer to www.mcs.com.pr go to Comunicados a Proveedores,
BOTOX Injection (Onabotulinumtoxin A) for Chronic Migraine Headaches [For the list of services and procedures that need preauthorization, please refer to www.mcs.com.pr go to Comunicados a Proveedores,
Tension-type headache Non-pharmacological and pharmacological treatment
 Danish Headache Center Tension-type headache Non-pharmacological and pharmacological treatment Lars Bendtsen Associate professor, MD, PhD, Dr Med Sci Danish Headache Center, Department of Neurology Glostrup
Danish Headache Center Tension-type headache Non-pharmacological and pharmacological treatment Lars Bendtsen Associate professor, MD, PhD, Dr Med Sci Danish Headache Center, Department of Neurology Glostrup
Version History. Previous Versions. Drugs for MS.Drug facts box fampridine Version 1.0 Author
 Version History Policy Title Drugs for MS.Drug facts box fampridine Version 1.0 Author West Midlands Commissioning Support Unit Publication Date Jan 2013 Review Date Supersedes/New (Further fields as required
Version History Policy Title Drugs for MS.Drug facts box fampridine Version 1.0 Author West Midlands Commissioning Support Unit Publication Date Jan 2013 Review Date Supersedes/New (Further fields as required
Adoption by CHMP for release for consultation November 2010. End of consultation (deadline for comments) 31 March 2011
 1 2 3 November 2010 EMA/759784/2010 Committee for Medicinal Products for Human Use 4 5 6 7 Reflection paper on the need for active control in therapeutic areas where use of placebo is deemed ethical and
1 2 3 November 2010 EMA/759784/2010 Committee for Medicinal Products for Human Use 4 5 6 7 Reflection paper on the need for active control in therapeutic areas where use of placebo is deemed ethical and
BOTOX Treatment. for Chronic Migraine. Information for patients and their families. Botulinum Toxin Type A
 BOTOX Treatment Botulinum Toxin Type A for Chronic Migraine Information for patients and their families. Is Chronic Migraine the same as Migraine? Chronic Migraine is similar to migraine as sufferers experience
BOTOX Treatment Botulinum Toxin Type A for Chronic Migraine Information for patients and their families. Is Chronic Migraine the same as Migraine? Chronic Migraine is similar to migraine as sufferers experience
Chronic daily headache
 Chronic daily headache Chronic daily headache (CDH) is defined as any headache syndrome affecting more than half of the month, i.e. a headache on more than 15 days per month. It is also known as daily
Chronic daily headache Chronic daily headache (CDH) is defined as any headache syndrome affecting more than half of the month, i.e. a headache on more than 15 days per month. It is also known as daily
When the Pain Won t Stop: Managing Chronic Daily Headache
 When the Pain Won t Stop: Managing Chronic Daily Headache Arnolda Eloff, MB, ChB, Mmed Presented at the University of Calgary s Wednesday Evening Course Program, Calgary, Alberta. Copyright Chronic daily
When the Pain Won t Stop: Managing Chronic Daily Headache Arnolda Eloff, MB, ChB, Mmed Presented at the University of Calgary s Wednesday Evening Course Program, Calgary, Alberta. Copyright Chronic daily
Tension-type headache Non-pharmacological and pharmacological treatment
 Danish Headache Center Tension-type headache Non-pharmacological and pharmacological treatment Lars Bendtsen Associate professor, MD, PhD, Dr Med Sci Danish Headache Center, Department of Neurology Glostrup
Danish Headache Center Tension-type headache Non-pharmacological and pharmacological treatment Lars Bendtsen Associate professor, MD, PhD, Dr Med Sci Danish Headache Center, Department of Neurology Glostrup
Version History. Previous Versions. Policy Title. Drugs for MS.Drug facts box Glatiramer Acetate Version 1.0 Author
 Version History Policy Title Drugs for MS.Drug facts box Glatiramer Acetate Version 1.0 Author West Midlands Commissioning Support Unit Publication Date Jan 2013 Review Date Supersedes/New (Further fields
Version History Policy Title Drugs for MS.Drug facts box Glatiramer Acetate Version 1.0 Author West Midlands Commissioning Support Unit Publication Date Jan 2013 Review Date Supersedes/New (Further fields
Drugs for MS.Drug fact box cannabis extract (Sativex) Version 1.0 Author
 Version History Policy Title Drugs for MS.Drug fact box cannabis extract (Sativex) Version 1.0 Author West Midlands Commissioning Support Unit Publication Date Jan 2013 Review Date Supersedes/New (Further
Version History Policy Title Drugs for MS.Drug fact box cannabis extract (Sativex) Version 1.0 Author West Midlands Commissioning Support Unit Publication Date Jan 2013 Review Date Supersedes/New (Further
HEADACHE. as. MUDr. Rudolf Černý, CSc. doc. MUDr. Petr Marusič, Ph.D.
 HEADACHE as. MUDr. Rudolf Černý, CSc. doc. MUDr. Petr Marusič, Ph.D. Dpt. of Neurology Charles University in Prague, 2nd Faculty of Medicine Motol University Hospital History of headache 1200 years B.C.
HEADACHE as. MUDr. Rudolf Černý, CSc. doc. MUDr. Petr Marusič, Ph.D. Dpt. of Neurology Charles University in Prague, 2nd Faculty of Medicine Motol University Hospital History of headache 1200 years B.C.
PROTOCOL SYNOPSIS Evaluation of long-term opioid efficacy for chronic pain
 P a g e 1 PROTOCOL SYNOPSIS Evaluation of long-term opioid efficacy for chronic pain Clinical Phase 4 Study Centers Study Period 25 U.S. sites identified and reviewed by the Steering Committee and Contract
P a g e 1 PROTOCOL SYNOPSIS Evaluation of long-term opioid efficacy for chronic pain Clinical Phase 4 Study Centers Study Period 25 U.S. sites identified and reviewed by the Steering Committee and Contract
Behavioral and Physical Treatments for Tension-type and Cervicogenic Headache
 Evidence Report: Behavioral and Physical Treatments for Tension-type and Cervicogenic Headache Douglas C. McCrory, MD, MHSc Donald B. Penzien, PhD Vic Hasselblad, PhD Rebecca N. Gray, DPhil Duke University
Evidence Report: Behavioral and Physical Treatments for Tension-type and Cervicogenic Headache Douglas C. McCrory, MD, MHSc Donald B. Penzien, PhD Vic Hasselblad, PhD Rebecca N. Gray, DPhil Duke University
New daily persistent headache in the paediatric population
 doi:10.1111/j.1468-2982.2008.01647.x New daily persistent headache in the paediatric population E Kung 1, SJ Tepper 2, AM Rapoport 3, FD Sheftell 4 & ME Bigal 1,5 1 Department of Neurology, Albert Einstein
doi:10.1111/j.1468-2982.2008.01647.x New daily persistent headache in the paediatric population E Kung 1, SJ Tepper 2, AM Rapoport 3, FD Sheftell 4 & ME Bigal 1,5 1 Department of Neurology, Albert Einstein
A chiropractic approach to managing migraine
 A chiropractic approach to managing migraine What is chiropractic? Chiropractic is a primary healthcare profession that specialises in the diagnosis, treatment and overall management of conditions that
A chiropractic approach to managing migraine What is chiropractic? Chiropractic is a primary healthcare profession that specialises in the diagnosis, treatment and overall management of conditions that
C 2 / C 3 N E RVE BLOCKS AND GREATER OCCIPITAL NERVE BLOCK IN CERV I C O G E N I C HEADACHE TREATMENT
 C 2 / C 3 N E RVE BLOCKS AND GREATER OCCIPITAL NERVE BLOCK IN CERV I C O G E N I C HEADACHE TREATMENT N u rten Inan, Aysegul Ceyhan, Levent Inan*, Ozlem Kavaklioglu, Alp Alptekin, Nurten Unal Ministry
C 2 / C 3 N E RVE BLOCKS AND GREATER OCCIPITAL NERVE BLOCK IN CERV I C O G E N I C HEADACHE TREATMENT N u rten Inan, Aysegul Ceyhan, Levent Inan*, Ozlem Kavaklioglu, Alp Alptekin, Nurten Unal Ministry
Summary and general discussion
 Chapter 7 Summary and general discussion Summary and general discussion In this thesis, treatment of vitamin K antagonist-associated bleed with prothrombin complex concentrate was addressed. In this we
Chapter 7 Summary and general discussion Summary and general discussion In this thesis, treatment of vitamin K antagonist-associated bleed with prothrombin complex concentrate was addressed. In this we
Version History. Previous Versions. Drugs for MS.Drug facts box fingolimod Version 1.0 Author
 Version History Policy Title Drugs for MS.Drug facts box fingolimod Version 1.0 Author West Midlands Commissioning Support Unit Publication Date Jan 2013 Review Date Supersedes/New (Further fields as required
Version History Policy Title Drugs for MS.Drug facts box fingolimod Version 1.0 Author West Midlands Commissioning Support Unit Publication Date Jan 2013 Review Date Supersedes/New (Further fields as required
Which injectable medication should I take for relapsing-remitting multiple sclerosis?
 Which injectable medication should I take for relapsing-remitting multiple sclerosis? A decision aid to discuss options with your doctor This decision aid is for you if you: Have multiple sclerosis Have
Which injectable medication should I take for relapsing-remitting multiple sclerosis? A decision aid to discuss options with your doctor This decision aid is for you if you: Have multiple sclerosis Have
Version History. Previous Versions. for secondary progressive MS (SPMS) Policy Title. Drugs for MS.Drug facts box Interferon beta 1b
 Version History Policy Title Drugs for MS.Drug facts box Interferon beta 1b for secondary progressive MS (SPMS) Version 1.0 Author West Midlands Commissioning Support Unit Publication Date Jan 2013 Review
Version History Policy Title Drugs for MS.Drug facts box Interferon beta 1b for secondary progressive MS (SPMS) Version 1.0 Author West Midlands Commissioning Support Unit Publication Date Jan 2013 Review
THE INTERNET STROKE CENTER PRESENTATIONS AND DISCUSSIONS ON STROKE MANAGEMENT
 THE INTERNET STROKE CENTER PRESENTATIONS AND DISCUSSIONS ON STROKE MANAGEMENT Stroke Prevention in Atrial Fibrillation Gregory Albers, M.D. Director Stanford Stroke Center Professor of Neurology and Neurological
THE INTERNET STROKE CENTER PRESENTATIONS AND DISCUSSIONS ON STROKE MANAGEMENT Stroke Prevention in Atrial Fibrillation Gregory Albers, M.D. Director Stanford Stroke Center Professor of Neurology and Neurological
The Chronification of Migraine
 The Chronification of Migraine Jennifer Bickel, MD Medical Director, Comprehensive Headache Clinic Children s Mercy Hospital Division of Neurology Associate Professor of Pediatrics University of Missouri-Kansas
The Chronification of Migraine Jennifer Bickel, MD Medical Director, Comprehensive Headache Clinic Children s Mercy Hospital Division of Neurology Associate Professor of Pediatrics University of Missouri-Kansas
Double-Blind Trial of Fluoxetine: Chronic Daily Headache and Migraine
 Double-Blind Trial of Fluoxetine: Chronic Daily Headache and Migraine Joel R. Saper, M.D., 1 Stephen D. Silberstein, M.D., 2 Alvin E. Lake III, Ph.D., 1 Marjorie E. Winters, R.N., B.S.N. 1 1 Michigan Head-Pain
Double-Blind Trial of Fluoxetine: Chronic Daily Headache and Migraine Joel R. Saper, M.D., 1 Stephen D. Silberstein, M.D., 2 Alvin E. Lake III, Ph.D., 1 Marjorie E. Winters, R.N., B.S.N. 1 1 Michigan Head-Pain
THE COMMONWEALTH OF MASSACHUSETTS Department of Industrial Accidents
 THE COMMONWEALTH OF MASSACHUSETTS Department of Industrial Accidents 1 Congress Street, Suite 100 Boston, Massachusetts 02114-2017 DEVAL L. PATRICK Governor PHILIP L. HILLMAN Director TIMOTHY P. MURRAY
THE COMMONWEALTH OF MASSACHUSETTS Department of Industrial Accidents 1 Congress Street, Suite 100 Boston, Massachusetts 02114-2017 DEVAL L. PATRICK Governor PHILIP L. HILLMAN Director TIMOTHY P. MURRAY
Diagnosing and treating episodic migraine
 D A N I S H H E A D A C H E C E N T E R Danish Headache Center Disclosures Diagnosing and treating episodic migraine M.A. is a consultant or scientific adviser for Allergan, Alder, Amgen, ATI, Bayer and
D A N I S H H E A D A C H E C E N T E R Danish Headache Center Disclosures Diagnosing and treating episodic migraine M.A. is a consultant or scientific adviser for Allergan, Alder, Amgen, ATI, Bayer and
One Day at a Time: When Headaches Become Chronic. Robert Shapiro, MD, PhD
 One Day at a Time: When Headaches Become Chronic Robert Shapiro, MD, PhD Disclosures Scientific/Medical Advisory Boards (since 10/12) Transcept Pharmaceuticals Chronic Headaches: Overview What is a chronic
One Day at a Time: When Headaches Become Chronic Robert Shapiro, MD, PhD Disclosures Scientific/Medical Advisory Boards (since 10/12) Transcept Pharmaceuticals Chronic Headaches: Overview What is a chronic
Botox treatment for chronic migraine
 University Teaching Trust Botox treatment for chronic migraine Humphrey Booth Building Neurosciences 0161 206 2563 0161 206 2427 All Rights Reserved 2016. Document for issue as handout.. This information
University Teaching Trust Botox treatment for chronic migraine Humphrey Booth Building Neurosciences 0161 206 2563 0161 206 2427 All Rights Reserved 2016. Document for issue as handout.. This information
Guidance on Investigational Medicinal Products (IMPs) and other medicinal products used in Clinical Trials
 EUROPEAN COMMISSION ENTERPRISE AND INDUSTRY DIRECTORATE-GENERAL Consumer goods Pharmaceuticals Guidance on Investigational Medicinal Products (IMPs) and other medicinal products used in Clinical Trials
EUROPEAN COMMISSION ENTERPRISE AND INDUSTRY DIRECTORATE-GENERAL Consumer goods Pharmaceuticals Guidance on Investigational Medicinal Products (IMPs) and other medicinal products used in Clinical Trials
This clinical study synopsis is provided in line with Boehringer Ingelheim s Policy on Transparency and Publication of Clinical Study Data.
 abcd Clinical Study for Public Disclosure This clinical study synopsis is provided in line with s Policy on Transparency and Publication of Clinical Study Data. The synopsis which is part of the clinical
abcd Clinical Study for Public Disclosure This clinical study synopsis is provided in line with s Policy on Transparency and Publication of Clinical Study Data. The synopsis which is part of the clinical
XXXXX Petitioner v File No. 121439-001. Issued and entered this 27 TH day of October 2011 by R. Kevin Clinton Commissioner ORDER
 In the matter of STATE OF MICHIGAN DEPARTMENT OF ENERGY, LABOR & ECONOMIC GROWTH OFFICE OF FINANCIAL AND INSURANCE REGULATION Before the Commissioner of Financial and Insurance Regulation XXXXX Petitioner
In the matter of STATE OF MICHIGAN DEPARTMENT OF ENERGY, LABOR & ECONOMIC GROWTH OFFICE OF FINANCIAL AND INSURANCE REGULATION Before the Commissioner of Financial and Insurance Regulation XXXXX Petitioner
ESCMID Online Lecture Library. by author
 Do statins improve outcomes of patients with sepsis and pneumonia? Jordi Carratalà Department of Infectious Diseases Statins for sepsis & community-acquired pneumonia Sepsis and CAP are major healthcare
Do statins improve outcomes of patients with sepsis and pneumonia? Jordi Carratalà Department of Infectious Diseases Statins for sepsis & community-acquired pneumonia Sepsis and CAP are major healthcare
HEADACHES IN CHILDREN AND ADOLESCENTS. Brian D. Ryals, M.D.
 HEADACHES IN CHILDREN AND ADOLESCENTS Brian D. Ryals, M.D. Frequency and Type of Headaches in Schoolchildren 8993 children age 7-15 in Sweden Migraine in 4% Frequent Nonmigrainous in 7% Infrequent Nonmigrainous
HEADACHES IN CHILDREN AND ADOLESCENTS Brian D. Ryals, M.D. Frequency and Type of Headaches in Schoolchildren 8993 children age 7-15 in Sweden Migraine in 4% Frequent Nonmigrainous in 7% Infrequent Nonmigrainous
Medication Policy Manual. Topic: Aubagio, teriflunomide Date of Origin: November 9, 2012
 Medication Policy Manual Policy No: dru283 Topic: Aubagio, teriflunomide Date of Origin: November 9, 2012 Committee Approval Date: December 12, 2014 Next Review Date: December 2015 Effective Date: January
Medication Policy Manual Policy No: dru283 Topic: Aubagio, teriflunomide Date of Origin: November 9, 2012 Committee Approval Date: December 12, 2014 Next Review Date: December 2015 Effective Date: January
Post Traumatic and other Headache Syndromes. Danielle L. Erb, MD Brain Rehabilitation Medicine, LLC Brain Injury Rehab Center, PRA
 Post Traumatic and other Headache Syndromes Danielle L. Erb, MD Brain Rehabilitation Medicine, LLC Brain Injury Rehab Center, PRA Over 45 million Americans have chronic, recurring headaches 62% of these
Post Traumatic and other Headache Syndromes Danielle L. Erb, MD Brain Rehabilitation Medicine, LLC Brain Injury Rehab Center, PRA Over 45 million Americans have chronic, recurring headaches 62% of these
2.0 Synopsis. Vicodin CR (ABT-712) M05-765 Clinical Study Report R&D/07/095. (For National Authority Use Only) to Part of Dossier: Volume:
 2.0 Synopsis Abbott Laboratories Name of Study Drug: Vicodin CR Name of Active Ingredient: Hydrocodone/Acetaminophen Extended Release (ABT-712) Individual Study Table Referring to Part of Dossier: Volume:
2.0 Synopsis Abbott Laboratories Name of Study Drug: Vicodin CR Name of Active Ingredient: Hydrocodone/Acetaminophen Extended Release (ABT-712) Individual Study Table Referring to Part of Dossier: Volume:
If several different trials are mentioned in one publication, the data of each should be extracted in a separate data extraction form.
 General Remarks This template of a data extraction form is intended to help you to start developing your own data extraction form, it certainly has to be adapted to your specific question. Delete unnecessary
General Remarks This template of a data extraction form is intended to help you to start developing your own data extraction form, it certainly has to be adapted to your specific question. Delete unnecessary
Treatment of Opioid Dependence: A Randomized Controlled Trial. Karen L. Sees, DO, Kevin L. Delucchi, PhD, Carmen Masson, PhD, Amy
 Category: Heroin Title: Methadone Maintenance vs 180-Day psychosocially Enriched Detoxification for Treatment of Opioid Dependence: A Randomized Controlled Trial Authors: Karen L. Sees, DO, Kevin L. Delucchi,
Category: Heroin Title: Methadone Maintenance vs 180-Day psychosocially Enriched Detoxification for Treatment of Opioid Dependence: A Randomized Controlled Trial Authors: Karen L. Sees, DO, Kevin L. Delucchi,
Re: LUMIGAN 0.03% (bimatoprost ophthalmic solution 0.03%) and LUMIGAN 0.01% (bimatoprost ophthalmic solution 0.01%)
 Allergan Ltd, 1st Floor, Marlow International, Parkway, Marlow, Bucks SL7 1YL Tel: (01628) 494444 Facsimile: (01628) 494449 Ref No: UK15-000677 Mandeep Allingham NW Surrey CCG February 18, 2015 Dear Ms
Allergan Ltd, 1st Floor, Marlow International, Parkway, Marlow, Bucks SL7 1YL Tel: (01628) 494444 Facsimile: (01628) 494449 Ref No: UK15-000677 Mandeep Allingham NW Surrey CCG February 18, 2015 Dear Ms
Breaking the cycle of medication overuse headache
 REVIEW CME CREDIT EDUCATIONAL OBJECTIVE: Readers will recognize the risk of developing medication overuse headache with increasing use of medications to treat acute migraine Stewart J. Tepper, MD * Center
REVIEW CME CREDIT EDUCATIONAL OBJECTIVE: Readers will recognize the risk of developing medication overuse headache with increasing use of medications to treat acute migraine Stewart J. Tepper, MD * Center
Alison White Devang Rai Richard Chye
 Ketamine use in hospice patients before and after the sentinel randomised controlled trial of ketamine in cancer pain: A single centre retrospective review Alison White Devang Rai Richard Chye Overview
Ketamine use in hospice patients before and after the sentinel randomised controlled trial of ketamine in cancer pain: A single centre retrospective review Alison White Devang Rai Richard Chye Overview
Clinical Study Synopsis
 Clinical Study Synopsis This Clinical Study Synopsis is provided for patients and healthcare professionals to increase the transparency of Bayer's clinical research. This document is not intended to replace
Clinical Study Synopsis This Clinical Study Synopsis is provided for patients and healthcare professionals to increase the transparency of Bayer's clinical research. This document is not intended to replace
Neck Pain & Cervicogenic Headache Integrating Research into Practice: San Luis Sports Therapy s Approach to Evidence-Based Practice
 Neck Pain & Cervicogenic Headache Integrating Research into Practice: San Luis Sports Therapy s Approach to Evidence-Based Practice PROBLEM: Neck Pain and Cervicogenic Headache 66% Proportion of individuals
Neck Pain & Cervicogenic Headache Integrating Research into Practice: San Luis Sports Therapy s Approach to Evidence-Based Practice PROBLEM: Neck Pain and Cervicogenic Headache 66% Proportion of individuals
Presented by: Jean Yoo-Campbell, Matthew Konerman, Monica Konerman, Jean Yoo Campbell, Christian Gocke, Eunpi Cho Donald Lynch
 Bass N.M., et. al. N Engl J Med 2010; 362:1071-1081 Presented by: Jean Yoo-Campbell, Matthew Konerman, Monica Konerman, Jean Yoo Campbell, Christian Gocke, Eunpi Cho Donald Lynch Faculty Advisor: Dr. Fred
Bass N.M., et. al. N Engl J Med 2010; 362:1071-1081 Presented by: Jean Yoo-Campbell, Matthew Konerman, Monica Konerman, Jean Yoo Campbell, Christian Gocke, Eunpi Cho Donald Lynch Faculty Advisor: Dr. Fred
Oral riboflavin versus oral propranolol in migraine prophylaxis: An open label randomized controlled trial
 Neurology Asia 2011; 16(3) : 223 229 Oral riboflavin versus oral propranolol in migraine prophylaxis: An open label randomized controlled trial 1 *Natasha J Nambiar MBBS MD, 1 *Cuckoo Aiyappa PhD, 2 R
Neurology Asia 2011; 16(3) : 223 229 Oral riboflavin versus oral propranolol in migraine prophylaxis: An open label randomized controlled trial 1 *Natasha J Nambiar MBBS MD, 1 *Cuckoo Aiyappa PhD, 2 R
BEDFORDSHIRE AND LUTON JOINT PRESCRIBING COMMITTEE (JPC)
 BEDFORDSHIRE AND LUTON JOINT PRESCRIBING COMMITTEE (JPC) September 2014 Review date: September 2017 Bulletin 203: Tocilizumab (subcutaneous) in combination with methotrexate or as monotherapy for the treatment
BEDFORDSHIRE AND LUTON JOINT PRESCRIBING COMMITTEE (JPC) September 2014 Review date: September 2017 Bulletin 203: Tocilizumab (subcutaneous) in combination with methotrexate or as monotherapy for the treatment
Spine Vol. 30 No. 16; August 15, 2005, pp 1799-1807
 A Randomized Controlled Trial of an Educational Intervention to Prevent the Chronic Pain of Whiplash Associated Disorders Following Rear-End Motor Vehicle Collisions 1 Spine Vol. 30 No. 16; August 15,
A Randomized Controlled Trial of an Educational Intervention to Prevent the Chronic Pain of Whiplash Associated Disorders Following Rear-End Motor Vehicle Collisions 1 Spine Vol. 30 No. 16; August 15,
TUTORIAL on ICH E9 and Other Statistical Regulatory Guidance. Session 1: ICH E9 and E10. PSI Conference, May 2011
 TUTORIAL on ICH E9 and Other Statistical Regulatory Guidance Session 1: PSI Conference, May 2011 Kerry Gordon, Quintiles 1 E9, and how to locate it 2 ICH E9 Statistical Principles for Clinical Trials (Issued
TUTORIAL on ICH E9 and Other Statistical Regulatory Guidance Session 1: PSI Conference, May 2011 Kerry Gordon, Quintiles 1 E9, and how to locate it 2 ICH E9 Statistical Principles for Clinical Trials (Issued
Issues Regarding Use of Placebo in MS Drug Trials. Peter Scott Chin, MD Novartis Pharmaceuticals Corporation
 Issues Regarding Use of Placebo in MS Drug Trials Peter Scott Chin, MD Novartis Pharmaceuticals Corporation Context of the Guidance The draft EMA Guidance mentions placebo as a comparator for superiority
Issues Regarding Use of Placebo in MS Drug Trials Peter Scott Chin, MD Novartis Pharmaceuticals Corporation Context of the Guidance The draft EMA Guidance mentions placebo as a comparator for superiority
Review for NHS Surrey Prescribing Clinical Network. Treatment: Botox for chronic migraine. Prepared by: Victoria Overland
 Review for NHS Surrey Prescribing Clinical Network Treatment: Botox for chronic migraine Prepared by: Victoria Overland Date: To be considered at September 2012 PCN meeting 1. Purpose of the Review NICE
Review for NHS Surrey Prescribing Clinical Network Treatment: Botox for chronic migraine Prepared by: Victoria Overland Date: To be considered at September 2012 PCN meeting 1. Purpose of the Review NICE
