Institute Assessment & Analytics (BGA)
|
|
|
- Barbra Wright
- 10 years ago
- Views:
Transcription
1 Institute Assessment & Analytics (BGA) Dr. Gerhard Beck BGA AGES Medizinmarktaufsicht Erfahrung mit der neuen Struktur Wien, 15. Sept Austrian Agency for Health and Food Safety
2 Assessment & Analytics G. Beck Consultants: Friedrich Lackner Andreas Mayrhofer EMA EDQM A s s e s s m e n t CASE Clinical Assessment of Safety & Efficacy B. Tucek BPSV Biologicals, Preclinical & Statistical Assessment, Veterinary Medicinal Products B. Zemann APHV Assessment Pharmacovigilance B. Tucek BAMA Analytics of Biological Medicinal Products H. Schindl CPAA Analytics of Chemical-pharmaceutical Medicinal Products R. Macas O M C L 1200 Traisengasse 1160 Possingergasse 1090 Zimmermanng. 2
3 Duties and responsibilities Assessment: applications for: marketing authorisation, variation, renewal, re-examination, referral -> Centralised procedure: biological, chemical & herbal medicinal products -> MRP/DCP and national procedure: biological & herbal medicinal products -> Pharmacovigilance procedures -> Scientific Advice EMA and National OMCL: ->Marketing surveillance (quality control) of medicinal products (legal & illegal market) ->Official control authority batch release 3
4 Department Clinical Assessment of Safety & Efficacy (CASE) Head of department: Barbara Tucek (Jan Neuhauser) CP Admin Team Stefan Fringer, Tanja Knapp, Sylvia Pach Scientific Advice René Anour (Andreas Kirisits) CP chem, biosim Jan Neuhauser (Christina Kerscher) Biologika Brigitte Müller (Daniela Philadelphy) HERB (LCM/QUAL: Länger) René Anour, Mahdi Ben-Cheikh, Dominika Chrusciel, Christiane Cserveny, Zehra Dzanic, Katharina Hausteiner-Melichar, Samantha Hewavitharana, Mirjam Hinterleitner, Christina Kerscher, Andreas Kirisits, Lisa Koinig, Ulrike Michel, Brigitte Müller, Jan Neuhauser, Daniela Philadelphy, Iria Schubert, Barbara Szivacz, Johanna Wernsperger, Daniela Wöhrer, Tanja Zahlner, Elvira Zimre-Grabensteiner Beatrix Cihal, Regina Mekonnen, Brigitta Sabor 4
5 Duties and responsibilities CP Chemical-, biological-, herbal human medicinal products Clinical assessment Applications for marketing authorisation, variations, renewals, referrals, re-examinations National, MRP/DCP Biological-, herbal human MPs Clinical assessment Applications for marketing authorisation, variations, renewals, referrals Scientific advice NASA EMA Contact (NASA): Nationaler Scientific Advice EMA-Scientific Advice 5
6 Working Parties & Committees Blood Products Working Party (BPWP) European Pandemic Task Force (ETF) Brigitte Müller Pharmacokinetic Working Party (PKWP) Jan Neuhauser National Vaccination Committee ( Nationales Impfgremium - NIG), National Pandemic Task Force Barbara Tucek 6
7 Department Assessment-Pharmacovigilance (APHV) Head of department: Barbara Tucek (Jan Neuhauser) Assessment - Pharmacovigilance Expert Group Coordinator Sonja Hrabcik (Susanne Wolf) Marianne Lunzer, Ramona Mirzaei, Leonhard-Key Mien, Manuela Mondl, Claudia Reichelt, Irmgard Resch, Katja Valent, Susanne Wolf Contact: [email protected] 7
8 Pharmacovigilance 8
9 Duties and responsibilities National procedures, MRP/DCP, CP Risk Management Plans (RMP) Post Authorisation Safety Studies (PASS) Referrals Safety-driven Variations Urgent Safety Restrictions (USR) Re-examinations Periodic Safety Update Reports (PSUR) PSUR Single Assessment (PSUSA) Periodic Benefit-Risk Evaluation Report (PBRER) 9
10 Working Parties & Committees Pharmacovigilance Risk Assessment Committee PRAC until Harald Herkner /MedUni Wien alternate: Jan Neuhauser after Jan Neuhauser alternate: Marianne Lunzer PSUR Worksharing Working Party Irmgard Resch 10
11 Biologicals, Preclinical & Statistical Assessment, Veterinary Medicinal Products (BPSV) Head of department: Barbara Zemann (Ulrike Heißenberger) Group Biologicals Ulrike Heissenberger (deputy head of dpt) Christian Mayer Petra Falb Irmtraud Held Martina Joseph Manuela Leitner Petra Schlick Preclincal assessment Günter Waxenecker Klaudia Hettinger Group Statistics Thomas Lang Christian Gartner Support: Daniela Gutschi Group Veterinary Medicinal Products Eugen Obermayr Brigitte Hauser Anja Kotschwar Ingrid Langner Ines Lindner Gabriele Rothmüller (Petra Günter- Stadlmaier) 11
12 Duties and responsibilities Assessment veterinary medicinal products Bio-quality, preclinical, clinical, statistical and pharmacovigilance data Assessment human medicinal products Bio-quality, preclinical and statistical data Support of the Clinical Trials Department Scientific Advice EMA & National Scientific Advice ESVAC Surveillance of Veterinary Antimicrobial Consumption PHV (v) 12
13 Working Parties & Committees Committee for Medicinal Products for Veterinary Use Barbara Zemann Safety Working Party Günter Waxenecker (h)/barbara Zemann (v) Scientific Advice Working Party Biostatistics Working Party (Vice Chair) Thomas Lang Pharmacovigilance Working Party-V (veterinary) Brigitte Hauser Nationales Impfgremium & Pandemic Task Force, Ad-hoc Influenca Working Party Petra Falb Plasma Master File Manuela Leitner 13
14 Contact (EMA-SA) 14
15 Department Analytics of Biological Medicinal Products (BAMA) Head of department: Heidemarie Schindl (Dieter Pullirsch) Coagulation Factors and Protein Analytics Expert Group Coordinator: Christoph Kefeder Berger Renate Eichmeir Stefanie Hottowy Brigitte Staber Eva Wollein Gabriele Assistence & Support Lisa Löffler Maria Schwiretz Vaccines Plasma Pool Testing Expert Group Coordinator: Dieter Pullirsch Belouafa Fatima Farkas Isabella Hottowy Brigitte Ritter Renate Sammer Cornelia Stejskal Christoph Wetzinger Christine Zechmann Ulrike 15
16 Duties and responsibilities Official control authority batch release: plasma pools plasma derived medicinal products vaccines Participation: Proficiency testing studies (PTS) biological standardisation program (BSP) testing of centrally authorised medicinal products (CAP) Elaboration and assessment: monographs, methods and reference preparations Contact: 16
17 Working Parties & Committees European Pharmacopoeia Commission Friedrich Lackner European Pharmacopoeia Expert Groups Group 6B (Human Blood and Blood Products) P4 Bio Biologicals Friedrich Lackner Group 15 (Sera and Vaccines) Heidemarie Schindl Group 15V (Veterinary Sera and Vaccines) Monoclonal Antibodies, Gene Therapy Products Dieter Pullirsch Allergens Christoph Kefeder 17
18 Analysis Chemical-pharmaceutical Medicinal Products (CPAA) Head of department: Roman Macas (Thomas Lang) Support: Brigitte Kunz Brabenetz Andrea Chromatographic, Spectroscopic & Identification Methods (CSIM) Expert Group Coordinator: Thomas Lang Waltraud Doiber Andreas Hackl Andrea Hölzel Zdenek Janicek Franz Lechner Hermann Leth Rosemarie Schuch Martina Waismayer Pharmaceutical-Technical, Pharmacognostic & Volumetric Methods (PPMM) Expert Group Coordinator: Reinhard Stark Elisabeth Egerth Brigitte Nedved Gerhard Nesetril Silvester Ölzant Christine Schmidt Josef Steinhäusler Nicole Walter 18
19 Duties and responsibilities Market surveillance of legal/illegal Medicinal Products Analysis of products based on routine sampling plan or based on quality defect report Analysis of unknown or suspicious medicinal products Participation: Proficiency testing studies (PTS), testing of centrally authorised medicinal products (CAP) Elaboration and assessment: monographs, methods and reference preparations Stockpiled medicines: Analysis to monitor the quality of stockpiled medicines to extent the period of use Contact: 19
Class 1B. Form tutor: Mag. Ruth Plessl-Studeny. Deputy form tutor: Mag. Monika Pessicka
 Class 1B Form tutor: Mag. Ruth Plessl-Studeny Deputy form tutor: Mag. Monika Pessicka Arts Mag. Bettina Nagy-Rumpf Wednesday, 09:55-10:45 Biology Mag. Ruth Plessl-Studeny Tuesday, 08:55-09:45 Crafts (technical)
Class 1B Form tutor: Mag. Ruth Plessl-Studeny Deputy form tutor: Mag. Monika Pessicka Arts Mag. Bettina Nagy-Rumpf Wednesday, 09:55-10:45 Biology Mag. Ruth Plessl-Studeny Tuesday, 08:55-09:45 Crafts (technical)
COMMITTEE FOR MEDICINAL PRODUCTS FOR HUMAN USE (CHMP) GUIDELINE ON SIMILAR BIOLOGICAL MEDICINAL PRODUCTS
 European Medicines Agency Evaluation of Medicines for Human Use CHMP/437/04 London, 30 October 2005 COMMITTEE FOR MEDICINAL PRODUCTS FOR HUMAN USE (CHMP) GUIDELINE ON SIMILAR BIOLOGICAL MEDICINAL PRODUCTS
European Medicines Agency Evaluation of Medicines for Human Use CHMP/437/04 London, 30 October 2005 COMMITTEE FOR MEDICINAL PRODUCTS FOR HUMAN USE (CHMP) GUIDELINE ON SIMILAR BIOLOGICAL MEDICINAL PRODUCTS
QUESTIONS AND ANSWERS ABOUT THE EDQM ACTIVITIES
 QUESTIONS AND ANSWERS ABOUT THE EDQM ACTIVITIES Why are Pharmacopoeias so important in a globalised world? Pharmacopoeias have historically provided collections of medical recipes intended to ensure accurate
QUESTIONS AND ANSWERS ABOUT THE EDQM ACTIVITIES Why are Pharmacopoeias so important in a globalised world? Pharmacopoeias have historically provided collections of medical recipes intended to ensure accurate
Report from the CMDh meeting held on 21-23 September 2015. !!! 3 months to go until the mandatory use of the electronic application form!!!
 Report from the meeting held on 21-23 September 2015!!! 3 months to go until the mandatory use of the electronic application form!!! Pharmacovigilance positions following PSUSA procedure for only nationally
Report from the meeting held on 21-23 September 2015!!! 3 months to go until the mandatory use of the electronic application form!!! Pharmacovigilance positions following PSUSA procedure for only nationally
Public Assessment Report. Scientific discussion. Paracetamol Orifarm 500 mg film-coated tablets. (Paracetamol) DK/H/2271/001/DC.
 Public Assessment Report Scientific discussion Paracetamol Orifarm 500 mg film-coated tablets (Paracetamol) DK/H/2271/001/DC 15 October 2014 This module reflects the scientific discussion for the approval
Public Assessment Report Scientific discussion Paracetamol Orifarm 500 mg film-coated tablets (Paracetamol) DK/H/2271/001/DC 15 October 2014 This module reflects the scientific discussion for the approval
TRANSATLANTIC TRADE AND INVESTMENT PARTNERSHIP
 DISCLAIMER: The EU reserves the right to make subsequent modifications to this text and to complement its proposals at a later stage, by modifying, supplementing or withdrawing all, or any part, at any
DISCLAIMER: The EU reserves the right to make subsequent modifications to this text and to complement its proposals at a later stage, by modifying, supplementing or withdrawing all, or any part, at any
Volume 2B Notice to Applicants. Medicinal products for human use. Presentation and format of the dossier. Common Technical Document (CTD)
 Volume 2B Notice to Applicants Medicinal products for human use Presentation and format of the dossier Common Technical Document (CTD) Introduction Edition June 26 Module 1 Edition May 28 Module 2 Edition
Volume 2B Notice to Applicants Medicinal products for human use Presentation and format of the dossier Common Technical Document (CTD) Introduction Edition June 26 Module 1 Edition May 28 Module 2 Edition
Questions & answers on signal management
 23 October 2015 EMA/261758/2013 Inspections and Human Medicines Pharmacovigilance Division This document addresses a number of questions which stakeholders, in particular marketing authorisation holders
23 October 2015 EMA/261758/2013 Inspections and Human Medicines Pharmacovigilance Division This document addresses a number of questions which stakeholders, in particular marketing authorisation holders
VOLUME 2A Procedures for marketing authorisation CHAPTER 1 MARKETING AUTHORISATION. June 2013
 EUROPEAN COMMISSION HEALTH AND CONSUMERS DIRECTORATE-GENERAL Health systems and products Medicinal products Revision 4 NOTICE TO APPLICANTS VOLUME 2A Procedures for marketing authorisation CHAPTER 1 MARKETING
EUROPEAN COMMISSION HEALTH AND CONSUMERS DIRECTORATE-GENERAL Health systems and products Medicinal products Revision 4 NOTICE TO APPLICANTS VOLUME 2A Procedures for marketing authorisation CHAPTER 1 MARKETING
Clinical Trials Center
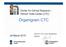 Clinical Trials Center Head CTC ZKF Managing Director Gregor Zünd ZKF Geschäftsführer Robin Schneider Head Quality Management QMR Jürg Lustenberger International Association Services Education and Training
Clinical Trials Center Head CTC ZKF Managing Director Gregor Zünd ZKF Geschäftsführer Robin Schneider Head Quality Management QMR Jürg Lustenberger International Association Services Education and Training
Implementation strategy for ISO IDMP in EU
 Implementation strategy for ISO IDMP in EU Paolo Alcini Head of Data Standardisation and Analytics Business Data & Analytics Information Management EMA An agency of the European Union The EU ISO IDMP Task
Implementation strategy for ISO IDMP in EU Paolo Alcini Head of Data Standardisation and Analytics Business Data & Analytics Information Management EMA An agency of the European Union The EU ISO IDMP Task
Regulatory approval routes in the European System for Medicinal Products
 Regulatory approval routes in the European System for Medicinal Products Cardiovascular Combination Pharmacotherapy Global Summit, Melbourne, 8 th May 2014 Presented by: Kevin Blake Human Medicines Research
Regulatory approval routes in the European System for Medicinal Products Cardiovascular Combination Pharmacotherapy Global Summit, Melbourne, 8 th May 2014 Presented by: Kevin Blake Human Medicines Research
Agence fédérale des médicaments et des produits de santé
 Agence fédérale des médicaments et des produits de santé From Risk Management Plan to Risk Minimization Activities New Royal Decree: Status and General Explanation Role of the Pharmacovigilance Department
Agence fédérale des médicaments et des produits de santé From Risk Management Plan to Risk Minimization Activities New Royal Decree: Status and General Explanation Role of the Pharmacovigilance Department
The information contained in these slides is for general purposes only and presents the state of knowledge at November 30, 2011
 1 The information contained in these slides is for general purposes only and presents the state of knowledge at November 30, 2011 No rights can be derived from this information The Medicines Evaluation
1 The information contained in these slides is for general purposes only and presents the state of knowledge at November 30, 2011 No rights can be derived from this information The Medicines Evaluation
Opinion/ Commission. Notification. Decision. Issued 2 / affected 3 amended on. 06/07/2015 SmPC, Labelling and PL
 Procedural steps taken and scientific information after the authorisation Application Scope Opinion/ Commission Product Summary number Notification Decision Information 1 issued on Issued 2 / affected
Procedural steps taken and scientific information after the authorisation Application Scope Opinion/ Commission Product Summary number Notification Decision Information 1 issued on Issued 2 / affected
A 4.5 Validity Period of a Marketing Authorisation
 104 A 4.5 Examples of situations in which the notification procedure according to Art. 61(3) of the Community Code will apply are as follows: Minor changes to the package leaflet resulting from readability
104 A 4.5 Examples of situations in which the notification procedure according to Art. 61(3) of the Community Code will apply are as follows: Minor changes to the package leaflet resulting from readability
COMMITTEE FOR PROPRIETARY MEDICINAL PRODUCTS (CPMP) COMMITTEE FOR VETERINARY MEDICINAL PRODUCTS (CVMP)
 European Medicines Agency Veterinary Medicines and Inspections London, 15 April 2005 EMEA/CVMP/134/02 Rev 1 CPMP/QWP/227/02 Rev 1 COMMITTEE FOR PROPRIETARY MEDICINAL PRODUCTS (CPMP) COMMITTEE FOR VETERINARY
European Medicines Agency Veterinary Medicines and Inspections London, 15 April 2005 EMEA/CVMP/134/02 Rev 1 CPMP/QWP/227/02 Rev 1 COMMITTEE FOR PROPRIETARY MEDICINAL PRODUCTS (CPMP) COMMITTEE FOR VETERINARY
Public Assessment Report. Scientific discussion. Calcium and Vitamine D3 Alpex 1000 mg/880 IE, effervescent granules
 Public Assessment Report Scientific discussion Calcium and Vitamine D3 Alpex 1000 mg/880 IE, effervescent granules (calcium carbonate and cholecalciferol) NL License RVG: 111783 Date: 12 March 2015 This
Public Assessment Report Scientific discussion Calcium and Vitamine D3 Alpex 1000 mg/880 IE, effervescent granules (calcium carbonate and cholecalciferol) NL License RVG: 111783 Date: 12 March 2015 This
EBE Position paper on labelling of biosimilars Summary of Product Characteristics (SmPC) and Patient Information Leaflet (PIL)- draft April 2013
 FINAL EBE 21 August 2013 EBE Position paper on labelling of biosimilars Summary of Product Characteristics (SmPC) and Patient Information Leaflet (PIL)- draft April 2013 Introduction This document seeks
FINAL EBE 21 August 2013 EBE Position paper on labelling of biosimilars Summary of Product Characteristics (SmPC) and Patient Information Leaflet (PIL)- draft April 2013 Introduction This document seeks
Opinion/ Commission. Notification. Decision. Issued 2 / affected 3 amended on. 30/07/2015 Annex II, Labelling and PL
 Procedural steps taken and scientific information after the authorisation Application Scope Opinion/ Commission Product Summary number Notification Decision Information 1 issued on Issued 2 / affected
Procedural steps taken and scientific information after the authorisation Application Scope Opinion/ Commission Product Summary number Notification Decision Information 1 issued on Issued 2 / affected
Non-clinical development of biologics
 Aurigon Life Science GmbH Non-clinical development of biologics Requirements, challenges and case studies Committed to Life. Sigrid Messemer vet. med. M4 Seminar March 10 th 2014 Aurigon - your full service
Aurigon Life Science GmbH Non-clinical development of biologics Requirements, challenges and case studies Committed to Life. Sigrid Messemer vet. med. M4 Seminar March 10 th 2014 Aurigon - your full service
Curriculum for Structured Doctoral Candidate Training at the Heinrich Pette Institute Leibniz Institute for Experimental Virology
 Curriculum for Structured Doctoral Candidate Training at the Heinrich Pette Institute Leibniz Institute for Experimental Virology Please choose lectures and courses according to your interests and requirements.
Curriculum for Structured Doctoral Candidate Training at the Heinrich Pette Institute Leibniz Institute for Experimental Virology Please choose lectures and courses according to your interests and requirements.
The European regulatory system for medicines and the European Medicines Agency
 The European regulatory system for medicines and the European Medicines Agency A consistent approach to medicines regulation across the European Union An agency of the European Union This booklet is intended
The European regulatory system for medicines and the European Medicines Agency A consistent approach to medicines regulation across the European Union An agency of the European Union This booklet is intended
The Human Medicines Regulations 2012
 STATUTORY INSTRUMENTS 2012 No. 1916 MEDICINES The Human Medicines Regulations 2012 Made - - - - 19th July 2012 Laid before Parliament 24th July 2012 Coming into force - - 14th August 2012 38.00 STATUTORY
STATUTORY INSTRUMENTS 2012 No. 1916 MEDICINES The Human Medicines Regulations 2012 Made - - - - 19th July 2012 Laid before Parliament 24th July 2012 Coming into force - - 14th August 2012 38.00 STATUTORY
1. Name of pharmacopoeia
 1 1. Name of pharmacopoeia European Pharmacopoeia 36 Member states (Austria, Belgium, Bosnia and Herzegovina, Bulgaria, Croatia, Cyprus, Czech Republic, Denmark, Estonia, Finland, France, Germany, Greece,
1 1. Name of pharmacopoeia European Pharmacopoeia 36 Member states (Austria, Belgium, Bosnia and Herzegovina, Bulgaria, Croatia, Cyprus, Czech Republic, Denmark, Estonia, Finland, France, Germany, Greece,
Public Assessment Report. Scientific discussion. Apotel 10 mg/ml, solution for infusion. (paracetamol) NL/H/2857/001/DC
 Public Assessment Report Scientific discussion Apotel 10 mg/ml, solution for infusion (paracetamol) NL/H/2857/001/DC Date: 16 December 2014 This module reflects the scientific discussion for the approval
Public Assessment Report Scientific discussion Apotel 10 mg/ml, solution for infusion (paracetamol) NL/H/2857/001/DC Date: 16 December 2014 This module reflects the scientific discussion for the approval
SPECIFICATIONS AND CONTROL TESTS ON THE FINISHED PRODUCT
 SPECIFICATIONS AND CONTROL TESTS ON THE FINISHED PRODUCT Guideline Title Specifications and Control Tests on the Finished Product Legislative basis Directive 75/318/EEC as amended Date of first adoption
SPECIFICATIONS AND CONTROL TESTS ON THE FINISHED PRODUCT Guideline Title Specifications and Control Tests on the Finished Product Legislative basis Directive 75/318/EEC as amended Date of first adoption
Report to the European Commission on Pharmacovigilance audits carried out in the Medicines Evaluation Board, The Netherlands period of time from
 Report to the European Commission on Pharmacovigilance audits carried out in the Medicines Evaluation Board, The Netherlands period of time from September 2013 to September 2015 1. INTRODUCTION Applicable
Report to the European Commission on Pharmacovigilance audits carried out in the Medicines Evaluation Board, The Netherlands period of time from September 2013 to September 2015 1. INTRODUCTION Applicable
Sustainable infrastructure? Selected physical, organizational and intelectual Austrian examples
 Civitas Forum 2015, Sharing the City October 8 th, 2015, Ljubljana, SLO Session 14: Designing sustainable urban mobility infrastructure Selected physical, organizational and intelectual Austrian examples
Civitas Forum 2015, Sharing the City October 8 th, 2015, Ljubljana, SLO Session 14: Designing sustainable urban mobility infrastructure Selected physical, organizational and intelectual Austrian examples
Opinion/ Commission. Notification 1. Decision. Information issued on. Issued 2 / affected 3 amended on
 Procedural steps taken and scientific information after the authorisation Application Scope Opinion/ Commission Product Summary number Notification 1 Decision Information issued on Issued 2 / affected
Procedural steps taken and scientific information after the authorisation Application Scope Opinion/ Commission Product Summary number Notification 1 Decision Information issued on Issued 2 / affected
One-year report on human medicines pharmacovigilance tasks of the European Medicines Agency
 8 April 2014 EMA/171322/2014 Pharmacovigilance Department One-year report on human medicines pharmacovigilance tasks of the European Medicines Agency Reporting period: 2 July 2012 to 1 July 2013 7 Westferry
8 April 2014 EMA/171322/2014 Pharmacovigilance Department One-year report on human medicines pharmacovigilance tasks of the European Medicines Agency Reporting period: 2 July 2012 to 1 July 2013 7 Westferry
Guideline on good pharmacovigilance practices (GVP)
 22 June 2012 EMA/827661/2011 Guideline on good pharmacovigilance practices (GVP) Module IX Signal management Draft finalised by the Agency in collaboration with Member States and submitted to ERMS FG 19
22 June 2012 EMA/827661/2011 Guideline on good pharmacovigilance practices (GVP) Module IX Signal management Draft finalised by the Agency in collaboration with Member States and submitted to ERMS FG 19
Data submission of authorised medicines in the European Union
 23 February 2015 EMA/471367/2014, Rev. 1 1 Business Data and Support Department Data submission of authorised medicines in the European Union Outlines on Article 57(2) of Regulation (EC) No 726/2004 1
23 February 2015 EMA/471367/2014, Rev. 1 1 Business Data and Support Department Data submission of authorised medicines in the European Union Outlines on Article 57(2) of Regulation (EC) No 726/2004 1
EDQM Certificates of Suitability: Cooperation Among Inspectorates in Europe And Beyond
 EDQM Certificates of Suitability: Cooperation Among Inspectorates in Europe And Beyond 14 th International Conference of Drug Regulatory Authorities Pre-ICDRA Meeting Dr Susanne Keitel European Directorate
EDQM Certificates of Suitability: Cooperation Among Inspectorates in Europe And Beyond 14 th International Conference of Drug Regulatory Authorities Pre-ICDRA Meeting Dr Susanne Keitel European Directorate
Leichtathletik Team Challenge München Olympiastadion München 08.08.2004
 100m Pupils Men Heat 1 Start Time: 13:00 Wind: -1,6 m/s 1 581 HAUST Sören HES 7 0.147 12.00 2 578 BESCHORNER Patrick HES 2 0.202 12.00 3 548 BRUNNER Thomas BAY 6 0.183 12.02 4 559 KROJER Julian BAY 8 0.210
100m Pupils Men Heat 1 Start Time: 13:00 Wind: -1,6 m/s 1 581 HAUST Sören HES 7 0.147 12.00 2 578 BESCHORNER Patrick HES 2 0.202 12.00 3 548 BRUNNER Thomas BAY 6 0.183 12.02 4 559 KROJER Julian BAY 8 0.210
HMPWG in the view of the NCA
 EMEA Homeopathic Workshop Bundesinstitut für Arzneimittel London, 27.10.2006 HMPWG in the view of the NCA Objectives, Achievements, Roles and Responsibilities Werner Knöss Head of Division Complementary
EMEA Homeopathic Workshop Bundesinstitut für Arzneimittel London, 27.10.2006 HMPWG in the view of the NCA Objectives, Achievements, Roles and Responsibilities Werner Knöss Head of Division Complementary
Public Assessment Report. Scientific discussion. Desloracell 5 mg, film-coated tablet. (desloratadine) NL License RVG: 112807
 Public Assessment Report Scientific discussion Desloracell 5 mg, film-coated tablet (desloratadine) NL License RVG: 112807 Date: 6 July 2015 This module reflects the scientific discussion for the approval
Public Assessment Report Scientific discussion Desloracell 5 mg, film-coated tablet (desloratadine) NL License RVG: 112807 Date: 6 July 2015 This module reflects the scientific discussion for the approval
PL 17871/0208 UKPAR TABLE OF CONTENTS
 Fultium-D 3 3,200 IU Capsules PL 17871/0208 UKPAR TABLE OF CONTENTS Lay Summary Page 2 Scientific discussion Page 4 Steps taken for assessment Page 11 Summary of Product Characteristics Page 12 Patient
Fultium-D 3 3,200 IU Capsules PL 17871/0208 UKPAR TABLE OF CONTENTS Lay Summary Page 2 Scientific discussion Page 4 Steps taken for assessment Page 11 Summary of Product Characteristics Page 12 Patient
Masters Learning mode (Форма обучения)
 Program Title (Название программы): Pharmacology Degree (Степень) Masters Learning mode (Форма обучения) Full-time and part-time Duration of study (Продолжительность программы) 2 years (4 years part time)
Program Title (Название программы): Pharmacology Degree (Степень) Masters Learning mode (Форма обучения) Full-time and part-time Duration of study (Продолжительность программы) 2 years (4 years part time)
LAW ON MEDICINES. Article 1. Article 2. Article 3 Manufacturing and marketing of medical devices is the activity of public interest.
 LAW ON MEDICINES I BASIC PROVISIONS Article 1 This Law regulates conditions for manufacturing and marketing medicines for human use and use in veterinary medicine, measures for quality assurance, safety
LAW ON MEDICINES I BASIC PROVISIONS Article 1 This Law regulates conditions for manufacturing and marketing medicines for human use and use in veterinary medicine, measures for quality assurance, safety
How To Understand The Paediatric Regulation
 Paediatric Use Marketing Authorisation (PUMA) from a National Competent Authority Point of View DGRA Annual Congress 2006 Bonn, 10. May 2006 Dr. Susanne Keitel Content PUMA The Future Background Legislative
Paediatric Use Marketing Authorisation (PUMA) from a National Competent Authority Point of View DGRA Annual Congress 2006 Bonn, 10. May 2006 Dr. Susanne Keitel Content PUMA The Future Background Legislative
HSA Consumer Guide. Understanding Vaccines, Vaccine Development and Production. www.hsa.gov.sg November 2009. How a Vaccine Works.
 November 2009 Understanding Vaccines, Vaccine Development and Production Vaccines, in general, help protect people from harmful infections before they come in contact with the disease. Vaccines may also
November 2009 Understanding Vaccines, Vaccine Development and Production Vaccines, in general, help protect people from harmful infections before they come in contact with the disease. Vaccines may also
Descendants of Friedrich Ohlhausen Sr. Family History
 Descendants of Friedrich Ohlhausen Sr. Family History Generation One 1. Friedrich OHLHAUSEN Sr. was born on 5 Apr 1863 in Belz, Bessarabia. He was confirmed on 9 Jun 1878 in Bortscheag, Bessarabia. Friedrich
Descendants of Friedrich Ohlhausen Sr. Family History Generation One 1. Friedrich OHLHAUSEN Sr. was born on 5 Apr 1863 in Belz, Bessarabia. He was confirmed on 9 Jun 1878 in Bortscheag, Bessarabia. Friedrich
MedDRA in pharmacovigilance industry perspective
 MedDRA in pharmacovigilance industry SFDA ICH MedDRA Workshop, Beijing, 13 14 May 2011 Christina Winter, MD, FFPM Medical director GlaxoSmithKline R&D / ICH MedDRA Management Board 2011 ICH International
MedDRA in pharmacovigilance industry SFDA ICH MedDRA Workshop, Beijing, 13 14 May 2011 Christina Winter, MD, FFPM Medical director GlaxoSmithKline R&D / ICH MedDRA Management Board 2011 ICH International
Guideline on good pharmacovigilance practices (GVP)
 1 2 20 February 2012 EMA/541760/2011 3 4 Guideline on good pharmacovigilance practices (GVP) Module I Pharmacovigilance systems and their quality systems Draft finalised by the Agency in collaboration
1 2 20 February 2012 EMA/541760/2011 3 4 Guideline on good pharmacovigilance practices (GVP) Module I Pharmacovigilance systems and their quality systems Draft finalised by the Agency in collaboration
Detailed guidance AEs reporting in clinical trials ( CT-3 ) Stefano Bonato, M.D. Pharmacovigilance Country Head Bayer HealthCare Pharmaceuticals
 Detailed guidance AEs reporting in clinical trials ( CT-3 ) Stefano Bonato, M.D. Pharmacovigilance Country Head Bayer HealthCare Pharmaceuticals Pharmacovigilance costs 0.12%-0.22% of hospital admissions
Detailed guidance AEs reporting in clinical trials ( CT-3 ) Stefano Bonato, M.D. Pharmacovigilance Country Head Bayer HealthCare Pharmaceuticals Pharmacovigilance costs 0.12%-0.22% of hospital admissions
Guideline on similar biological medicinal products containing interferon beta
 21 February 2013 EMA/CHMP/BMWP/652000/2010 Committee for Medicinal Products for Human Use (CHMP) Guideline on similar biological medicinal products containing interferon beta Draft Agreed by BMWP June
21 February 2013 EMA/CHMP/BMWP/652000/2010 Committee for Medicinal Products for Human Use (CHMP) Guideline on similar biological medicinal products containing interferon beta Draft Agreed by BMWP June
Official Journal of the European Union. (Acts whose publication is obligatory)
 27.12.2006 L 378/1 I (Acts whose publication is obligatory) REGULATION (EC) No 1901/2006 OF THE EUROPEAN PARLIAMT AND OF THE COUNCIL of 12 December 2006 on medicinal products for paediatric use and amending
27.12.2006 L 378/1 I (Acts whose publication is obligatory) REGULATION (EC) No 1901/2006 OF THE EUROPEAN PARLIAMT AND OF THE COUNCIL of 12 December 2006 on medicinal products for paediatric use and amending
VOLUME 6A Procedures for marketing authorisation
 EUROPEAN COMMISSION ENTERPRISE AND INDUSTRY DIRECTORATE-GENERAL Consumer goods Pharmaceuticals Brussels, 29.01.2007 ENTR/F2/KK D(2007) Revision 3.1 NOTICE TO APPLICANTS VETERINARY MEDICINAL PRODUCTS VOLUME
EUROPEAN COMMISSION ENTERPRISE AND INDUSTRY DIRECTORATE-GENERAL Consumer goods Pharmaceuticals Brussels, 29.01.2007 ENTR/F2/KK D(2007) Revision 3.1 NOTICE TO APPLICANTS VETERINARY MEDICINAL PRODUCTS VOLUME
Guide to Fees for Veterinary Products
 Guide to Fees for Veterinary Products FIN-G0003-14 04 FEBRUARY 2016 This guide does not purport to be an interpretation of law and/or regulations and is for guidance purposes only. CONTENTS ABBREVIATIONS
Guide to Fees for Veterinary Products FIN-G0003-14 04 FEBRUARY 2016 This guide does not purport to be an interpretation of law and/or regulations and is for guidance purposes only. CONTENTS ABBREVIATIONS
Guideline on dossier requirements for Type IA and IB notifications
 Guideline on dossier requirements for Type IA and IB notifications In accordance with Regulation (EC) No 726/2004 and Directives 2001/83/EC and 2001/82/EC, a common approach to the procedures for variations
Guideline on dossier requirements for Type IA and IB notifications In accordance with Regulation (EC) No 726/2004 and Directives 2001/83/EC and 2001/82/EC, a common approach to the procedures for variations
Pharmacovigilance System Master File (PSMF), QPPV and audits
 Federal Agency for Medicines and Health Products (FAMHP) Pharmacovigilance System Master File (PSMF), QPPV and audits Matthijs Nele, PhV Inspector, DG Inspection BRAS - FAMHP/NM/ 1 Content Introduction
Federal Agency for Medicines and Health Products (FAMHP) Pharmacovigilance System Master File (PSMF), QPPV and audits Matthijs Nele, PhV Inspector, DG Inspection BRAS - FAMHP/NM/ 1 Content Introduction
An túdarás Rialála Táirgí Sláinte Health Products Regulatory Authority
 ANNUAL REPORT 2014 CONTENTS 2014 STATISTICS AT A GLANCE 2 CHAIRMAN S STATEMENT 4 AUTHORITY MEMBERS 6 MANAGEMENT COMMITTEE 7 CHIEF EXECUTIVE S REPORT 8 AUTHORISATION, REGISTRATION 18 AND LICENSING ACTIVITIES
ANNUAL REPORT 2014 CONTENTS 2014 STATISTICS AT A GLANCE 2 CHAIRMAN S STATEMENT 4 AUTHORITY MEMBERS 6 MANAGEMENT COMMITTEE 7 CHIEF EXECUTIVE S REPORT 8 AUTHORISATION, REGISTRATION 18 AND LICENSING ACTIVITIES
Guideline on good pharmacovigilance practices (GVP)
 15 April 2014 EMA/876333/2011 Rev 3* Guideline on good pharmacovigilance practices (GVP) Annex I - Definitions (Rev 3) Date for coming into effect of first version 2 July 2012 Date for coming into effect
15 April 2014 EMA/876333/2011 Rev 3* Guideline on good pharmacovigilance practices (GVP) Annex I - Definitions (Rev 3) Date for coming into effect of first version 2 July 2012 Date for coming into effect
Risk management plans an overview
 Risk management plans an overview Elspeth Kay Director, Risk Management Plan Evaluation Section, Post-marketing Surveillance Branch, Monitoring and Compliance Division, TGA ARCS-TGA workshop 12 March 2015
Risk management plans an overview Elspeth Kay Director, Risk Management Plan Evaluation Section, Post-marketing Surveillance Branch, Monitoring and Compliance Division, TGA ARCS-TGA workshop 12 March 2015
Quality by Design Concept
 3rd Jerusalem Conference on Quality and Pharma Sciences 6-7 June, 2012 QbD in Clinical Research - Where Can QbD Impact Clinical Research Practices? Dr. Yafit Stark Vice President, TEVA Pharmaceutical Industries,
3rd Jerusalem Conference on Quality and Pharma Sciences 6-7 June, 2012 QbD in Clinical Research - Where Can QbD Impact Clinical Research Practices? Dr. Yafit Stark Vice President, TEVA Pharmaceutical Industries,
Lacidipine 2 mg Film-Coated Tablets PL 08553/0502. Lacidipine 4 mg Film-Coated Tablets PL 08553/0503. UK Public Assessment Report
 Lacidipine 2 mg Film-Coated Tablets PL 08553/0502 Lacidipine 4 mg Film-Coated Tablets PL 08553/0503 UK Public Assessment Report TABLE OF CONTENTS Lay Summary Page 2 Scientific discussion Page 4 Steps taken
Lacidipine 2 mg Film-Coated Tablets PL 08553/0502 Lacidipine 4 mg Film-Coated Tablets PL 08553/0503 UK Public Assessment Report TABLE OF CONTENTS Lay Summary Page 2 Scientific discussion Page 4 Steps taken
EUROPEAN COMMISSION ENTERPRISE DIRECTORATE-GENERAL. Brussels, ENTR/F2/BL D(2003) CT 1 Revision 2
 EUROPEAN COMMISSION ENTERPRISE DIRECTORATE-GENERAL Single market : management & legislation for consumer goods Pharmaceuticals : regulatory framework and market authorisations Brussels, ENTR/F2/BL D(2003)
EUROPEAN COMMISSION ENTERPRISE DIRECTORATE-GENERAL Single market : management & legislation for consumer goods Pharmaceuticals : regulatory framework and market authorisations Brussels, ENTR/F2/BL D(2003)
Compilation of individual product-specific guidance on demonstration of bioequivalence
 17 December 2014 EMA/CHMP/736403/2014 Committee for Medicinal Products for Human Use (CHMP) Compilation of individual product-specific guidance on demonstration of bioequivalence Initial batch of individual
17 December 2014 EMA/CHMP/736403/2014 Committee for Medicinal Products for Human Use (CHMP) Compilation of individual product-specific guidance on demonstration of bioequivalence Initial batch of individual
Review of CBER's Current and Planned Post-marketing Safety Practices
 Review of CBER's Current and Planned Post-marketing Safety Practices Robert Ball MD, MPH, ScM Director, Office of Biostatistics and Epidemiology (OBE), Center for Biologics Evaluation and Research (CBER),
Review of CBER's Current and Planned Post-marketing Safety Practices Robert Ball MD, MPH, ScM Director, Office of Biostatistics and Epidemiology (OBE), Center for Biologics Evaluation and Research (CBER),
EUROPEAN PHARMACOPOEIA REQUIREMENTS FOR FISH VACCINES
 EDQM WORKSHOP EUROPEAN PHARMACOPOEIA REQUIREMENTS FOR FISH VACCINES 8 SEPTEMBER 2015 17 th International Conference on Diseases of Fish and Shellfish, Las Palmas de Gran Canaria, Spain PRESENTATIONS Table
EDQM WORKSHOP EUROPEAN PHARMACOPOEIA REQUIREMENTS FOR FISH VACCINES 8 SEPTEMBER 2015 17 th International Conference on Diseases of Fish and Shellfish, Las Palmas de Gran Canaria, Spain PRESENTATIONS Table
Risk Management Plan (RMP) Guidance (Draft)
 Pharmaceutical and Food Safety Bureau, Ministry of Health, Labour and Welfare Translated by Pharmaceuticals and Medical Devices Agency Pharmaceutical and Food Safety Bureau, Ministry of Health, Labour
Pharmaceutical and Food Safety Bureau, Ministry of Health, Labour and Welfare Translated by Pharmaceuticals and Medical Devices Agency Pharmaceutical and Food Safety Bureau, Ministry of Health, Labour
Comparative analysis between the possible regulatory approaches to GMP compliance TITOLO PRESENTAZIONE
 Comparative analysis between the possible regulatory approaches to GMP compliance TITOLO PRESENTAZIONE Dr. Fulvio CARLOTTI, GNOSIS SpA, Corporate QA Director September 26, 2014 Scope of GMP GMP compliance
Comparative analysis between the possible regulatory approaches to GMP compliance TITOLO PRESENTAZIONE Dr. Fulvio CARLOTTI, GNOSIS SpA, Corporate QA Director September 26, 2014 Scope of GMP GMP compliance
A critical review of the current. marketing authorisation transfer procedure. in Europe
 A critical review of the current marketing authorisation transfer procedure in Europe Wissenschaftliche Prüfungsarbeit zur Erlangung des Titels Master of Drug Regulatory Affairs der Mathematisch-Naturwissenschaftlichen
A critical review of the current marketing authorisation transfer procedure in Europe Wissenschaftliche Prüfungsarbeit zur Erlangung des Titels Master of Drug Regulatory Affairs der Mathematisch-Naturwissenschaftlichen
Brand Management Tutorial
 Brand Management Tutorial Summer Term 2014 Master of Science Module Brand Management & Integrated Communication 1 List of Course Participants Please check whether you are already included on the list If
Brand Management Tutorial Summer Term 2014 Master of Science Module Brand Management & Integrated Communication 1 List of Course Participants Please check whether you are already included on the list If
Big Data in Medicine
 Symposium Big Data in Medicine Joint Symposium of the German National Academy of Sciences Leopoldina and the Hasso Plattner Institute for Software Systems Engineering July 1-2, 2015 Hasso Plattner Institute
Symposium Big Data in Medicine Joint Symposium of the German National Academy of Sciences Leopoldina and the Hasso Plattner Institute for Software Systems Engineering July 1-2, 2015 Hasso Plattner Institute
PHARMACEUTICAL REFERENCE STANDARDS
 PHARMACEUTICAL REFERENCE STANDARDS 11 th International Symposium organised by the European Directorate for the Quality of Medicines & HealthCare (EDQM), Council of Europe 3-4 September 2012, Strasbourg,
PHARMACEUTICAL REFERENCE STANDARDS 11 th International Symposium organised by the European Directorate for the Quality of Medicines & HealthCare (EDQM), Council of Europe 3-4 September 2012, Strasbourg,
The compassionate use of medicinal products.
 The compassionate use of medicinal products. The French ATU system C. Bélorgey Head of Department of evaluation of Clinical Trials and Medicinal products of special status ECRD 23 May 2012 Disclaimer Employee
The compassionate use of medicinal products. The French ATU system C. Bélorgey Head of Department of evaluation of Clinical Trials and Medicinal products of special status ECRD 23 May 2012 Disclaimer Employee
Guidelines. of 16.05.2013
 EUROPEAN COMMISSION Brussels, 16.05.2013 C (2013) 2804 Guidelines of 16.05.2013 on the details of the various categories of variations, on the operation of the procedures laid down in Chapters, a, I and
EUROPEAN COMMISSION Brussels, 16.05.2013 C (2013) 2804 Guidelines of 16.05.2013 on the details of the various categories of variations, on the operation of the procedures laid down in Chapters, a, I and
Risk Management Plan for Drug Establishments
 Republic of the Philippines Department of Health FOOD AND DRUG ADMINISTRATION Risk Management Plan for Drug Establishments Center for Drug Regulation and Research Food and Drug Administration 17 December
Republic of the Philippines Department of Health FOOD AND DRUG ADMINISTRATION Risk Management Plan for Drug Establishments Center for Drug Regulation and Research Food and Drug Administration 17 December
Minimum Information Model for Reporting
 Minimum Information Model for Reporting Dr. H. Sefiani - Pr. R. Soulaymani Bencheikh 1 Background WHO created the Department Safety and vigilances with ambition to integrate all Health Vigilances that
Minimum Information Model for Reporting Dr. H. Sefiani - Pr. R. Soulaymani Bencheikh 1 Background WHO created the Department Safety and vigilances with ambition to integrate all Health Vigilances that
Guideline on the clinical investigation of recombinant and human plasma-derived factor VIII products
 28 January 2016 EMA/CHMP/BPWP/144533/2009 rev. 1 Committee for medicinal products for human use (CHMP) Guideline on the clinical investigation of recombinant and human plasma-derived factor VIII products
28 January 2016 EMA/CHMP/BPWP/144533/2009 rev. 1 Committee for medicinal products for human use (CHMP) Guideline on the clinical investigation of recombinant and human plasma-derived factor VIII products
List of Special Topics
 Notes List of Special Topics Published in the Financial Stability Report Series The following contributions can be downloaded at www.oenb.at. Financial Stability Report 25 The Single Supervisory Mechanism
Notes List of Special Topics Published in the Financial Stability Report Series The following contributions can be downloaded at www.oenb.at. Financial Stability Report 25 The Single Supervisory Mechanism
MRP & DCP step by steb instructions how to apply and how the procedures are conducted
 MRP & DCP step by steb instructions how to apply and how the procedures are conducted 06 May 2013 Dubrovnik, Croatia Dr. Peter Bachmann Head of Unit Coordination Group European and International Affairs
MRP & DCP step by steb instructions how to apply and how the procedures are conducted 06 May 2013 Dubrovnik, Croatia Dr. Peter Bachmann Head of Unit Coordination Group European and International Affairs
GMP Issues for Start-Ups, quality in the supply chain and role of the Qualified Person (QP)
 GMP Issues for Start-Ups, quality in the supply chain and role of the Qualified Person (QP) Presented by: Patrick Costello Scientific Administrator, European Medicines Agency An agency of the European
GMP Issues for Start-Ups, quality in the supply chain and role of the Qualified Person (QP) Presented by: Patrick Costello Scientific Administrator, European Medicines Agency An agency of the European
exactly. The need for efficiency in developing effective new therapeutics has never been greater.
 exactly. The need for efficiency in developing effective new therapeutics has never been greater. As demands on the global healthcare system increase and treating disease becomes more complex, the research,
exactly. The need for efficiency in developing effective new therapeutics has never been greater. As demands on the global healthcare system increase and treating disease becomes more complex, the research,
Guideline on good pharmacovigilance practices (GVP)
 22 June 2012 EMA/541760/2011 Guideline on good pharmacovigilance practices (GVP) Module I Pharmacovigilance systems and their quality systems Draft finalised by the Agency in collaboration with Member
22 June 2012 EMA/541760/2011 Guideline on good pharmacovigilance practices (GVP) Module I Pharmacovigilance systems and their quality systems Draft finalised by the Agency in collaboration with Member
EMA and Progressive Multifocal Leukoencephalopathy.
 EMA and Progressive Multifocal Leukoencephalopathy. ENCePP Plenary, London 23 November 2011 Presented by: Henry Fitt Head of Coordination & Networking, Pharmacovigilance & Risk Management An agency of
EMA and Progressive Multifocal Leukoencephalopathy. ENCePP Plenary, London 23 November 2011 Presented by: Henry Fitt Head of Coordination & Networking, Pharmacovigilance & Risk Management An agency of
From Drug Discovery to First in Humans
 0 :: 2 v01 x y p.345 B xxx.:: B A x y pval«««[xy] -løøøø xxx.:: øøøø -0.02-0.01-0.00 0.00 0.01 0.02.1.2 A PharmaTrain Centre of Excellence Non-Clinical Testing, Pharmaceutical & Early Clinical Development:
0 :: 2 v01 x y p.345 B xxx.:: B A x y pval«««[xy] -løøøø xxx.:: øøøø -0.02-0.01-0.00 0.00 0.01 0.02.1.2 A PharmaTrain Centre of Excellence Non-Clinical Testing, Pharmaceutical & Early Clinical Development:
Die Zulassung von Ultra Orphans und die Rolle des COMP und CAT 14. DGRA Jahreskongress 30.-31.5.2012, Bonn
 Die Zulassung von Ultra Orphans und die Rolle des COMP und CAT 14. DGRA Jahreskongress 30.-31.5.2012, Bonn Dr. med. Christian K Schneider Committee for Advanced Therapies (CAT), EMA, London CHMP Biosimilar
Die Zulassung von Ultra Orphans und die Rolle des COMP und CAT 14. DGRA Jahreskongress 30.-31.5.2012, Bonn Dr. med. Christian K Schneider Committee for Advanced Therapies (CAT), EMA, London CHMP Biosimilar
The Product Review Life Cycle A Brief Overview
 Stat & Quant Mthds Pharm Reg (Spring 2, 2014) Lecture 2,Week 1 1 The review process developed over a 40 year period and has been influenced by 5 Prescription User Fee Act renewals Time frames for review
Stat & Quant Mthds Pharm Reg (Spring 2, 2014) Lecture 2,Week 1 1 The review process developed over a 40 year period and has been influenced by 5 Prescription User Fee Act renewals Time frames for review
EUROPEAN COMMISSION HEALTH AND CONSUMERS DIRECTORATE-GENERAL
 EUROPEAN COMMISSION HEALTH AND CONSUMERS DIRECTORATE-GENERAL Health systems and products Medicinal products authorisations, EMA Brussels, 27.03.2014 ENTR/6283/00 Rev 4 orphan\guidelines\format content
EUROPEAN COMMISSION HEALTH AND CONSUMERS DIRECTORATE-GENERAL Health systems and products Medicinal products authorisations, EMA Brussels, 27.03.2014 ENTR/6283/00 Rev 4 orphan\guidelines\format content
EU Clinical Trials Register
 EU Clinical Trials Register Disclaimer: The explanations are provided for the benefit of public users of the system and to enhance general understanding of terms used. They are not intended as the regulatory
EU Clinical Trials Register Disclaimer: The explanations are provided for the benefit of public users of the system and to enhance general understanding of terms used. They are not intended as the regulatory
Overview of the EU CTA Regulatory Framework and Future Direction of the EU Clinical Trial Directive. Features
 72 Features STEPHAN REYNIER Project Director Voisin Consulting, France Overview of the EU CTA Regulatory Framework and Future Direction of the EU Clinical Trial Directive Over the last few years, the European
72 Features STEPHAN REYNIER Project Director Voisin Consulting, France Overview of the EU CTA Regulatory Framework and Future Direction of the EU Clinical Trial Directive Over the last few years, the European
OMCL Network of the Council of Europe QUALITY MANAGEMENT DOCUMENT
 OMCL Network of the Council of Europe QUALITY MANAGEMENT DOCUMENT PA/PH/OMCL (11) 204 3R HANDLING AND USE OF REFERENCE STANDARDS IN THE OMCL NETWORK Full document title and reference Document type Legislative
OMCL Network of the Council of Europe QUALITY MANAGEMENT DOCUMENT PA/PH/OMCL (11) 204 3R HANDLING AND USE OF REFERENCE STANDARDS IN THE OMCL NETWORK Full document title and reference Document type Legislative
Medicine Safety Glossary
 The following definitions are provided as a resource to supplement the information provided in the Medicine Safety Education section of the Pfizer.com Web site; they are not intended as a comprehensive
The following definitions are provided as a resource to supplement the information provided in the Medicine Safety Education section of the Pfizer.com Web site; they are not intended as a comprehensive
Summary of the risk management plan (RMP) for Cerdelga (eliglustat)
 EMA/743948/2014 Summary of the risk management plan (RMP) for Cerdelga (eliglustat) This is a summary of the risk management plan (RMP) for Cerdelga, which details the measures to be taken in order to
EMA/743948/2014 Summary of the risk management plan (RMP) for Cerdelga (eliglustat) This is a summary of the risk management plan (RMP) for Cerdelga, which details the measures to be taken in order to
1. Inleiding...5 2. Samenstelling van de Nederlandse delegatie naar de EFC...6 3. Activiteiten en resultaten van de EFC...6
 2 !" #$%& 1. Inleiding...5 2. Samenstelling van de Nederlandse delegatie naar de EFC...6 3. Activiteiten en resultaten van de EFC...6 '((&...6 '(()...7 '('(*...7 '(+(...8 '(,(...8 '(-(./*0...8 '(1(.*...8
2 !" #$%& 1. Inleiding...5 2. Samenstelling van de Nederlandse delegatie naar de EFC...6 3. Activiteiten en resultaten van de EFC...6 '((&...6 '(()...7 '('(*...7 '(+(...8 '(,(...8 '(-(./*0...8 '(1(.*...8
Public Assessment Report. Decentralised Procedure. Cefuroxime 250mg and 500mg film-coated tablets. Cefuroxime 500mg film-coated tablets
 Public Assessment Report Decentralised Procedure Cefuroxime 250mg film-coated tablets Cefuroxime 500mg film-coated tablets Procedure No: UK Licence No: PL 35646/0020-0021 Alkem Pharma GmbH 1 LAY SUMMARY
Public Assessment Report Decentralised Procedure Cefuroxime 250mg film-coated tablets Cefuroxime 500mg film-coated tablets Procedure No: UK Licence No: PL 35646/0020-0021 Alkem Pharma GmbH 1 LAY SUMMARY
Guideline on good pharmacovigilance practices (GVP)
 1 2 24 February 2016 EMA/838713/2011 Rev 2* Draft for public consultation 3 4 Guideline on good pharmacovigilance practices (GVP) Module V Risk management systems (Rev 2) Date for coming into effect of
1 2 24 February 2016 EMA/838713/2011 Rev 2* Draft for public consultation 3 4 Guideline on good pharmacovigilance practices (GVP) Module V Risk management systems (Rev 2) Date for coming into effect of
Decentralised Procedure. Public Assessment Report
 Bundesinstitut für Arzneimittel und Medizinprodukte Decentralised Procedure Public Assessment Report ben-u-ron direkt Erdbeer/Vanille 250/500 mg Granulat in Beuteln ben-u-ron direkt Cappuccino 500/1000
Bundesinstitut für Arzneimittel und Medizinprodukte Decentralised Procedure Public Assessment Report ben-u-ron direkt Erdbeer/Vanille 250/500 mg Granulat in Beuteln ben-u-ron direkt Cappuccino 500/1000
APRS Symposium and Late Summer Meeting 2015
 Wednesday, August 26 Friday, August 28, 2015 Wednesday, August 26: Program 08:00 08:30-09:00 09:00-09:45 11:00-12:15 12:15-14:00 14:00-17:30 17:30-18:00 18:00 18:00-19:45 19:45 20:00 22:00 Symposium shuttle
Wednesday, August 26 Friday, August 28, 2015 Wednesday, August 26: Program 08:00 08:30-09:00 09:00-09:45 11:00-12:15 12:15-14:00 14:00-17:30 17:30-18:00 18:00 18:00-19:45 19:45 20:00 22:00 Symposium shuttle
Seminario SSFA. New Guideline on Good Pharmacovigilance Practice (GVP) Fare clic per modificare lo stile del sottotitolo dello schema
 Seminario SSFA New Guideline on Good Pharmacovigilance Practice (GVP) Pharmacovigilance Systems and their Quality Systems Milano, 20th April 2012 GIULIA M. VALSECCHI The proposed GVP - Module 1 Guidance
Seminario SSFA New Guideline on Good Pharmacovigilance Practice (GVP) Pharmacovigilance Systems and their Quality Systems Milano, 20th April 2012 GIULIA M. VALSECCHI The proposed GVP - Module 1 Guidance
Guideline on similar biological medicinal products containing biotechnology-derived proteins as active substance: quality issues (revision 1)
 22 May 2014 EMA/CHMP/BWP/247713/2012 Committee for Medicinal Products for Human Use (CHMP) Guideline on similar biological medicinal products containing biotechnology-derived proteins as active substance:
22 May 2014 EMA/CHMP/BWP/247713/2012 Committee for Medicinal Products for Human Use (CHMP) Guideline on similar biological medicinal products containing biotechnology-derived proteins as active substance:
Public Assessment Report. Decentralised Procedure
 Public Assessment Report Decentralised Procedure Levetiracetam 250 mg film-coated tablets PL 36390/0168; UK/H/5630/001/DC Levetiracetam 500 mg film-coated tablets PL 36390/0169; UK/H/5630/002/DC Levetiracetam
Public Assessment Report Decentralised Procedure Levetiracetam 250 mg film-coated tablets PL 36390/0168; UK/H/5630/001/DC Levetiracetam 500 mg film-coated tablets PL 36390/0169; UK/H/5630/002/DC Levetiracetam
COMMISSION STAFF WORKING DOCUMENT EXECUTIVE SUMMARY OF THE IMPACT ASSESSMENT. Accompanying the document
 EUROPEAN COMMISSION Brussels, 26.6.2013 SWD(2013) 235 final COMMISSION STAFF WORKING DOCUMENT EXECUTIVE SUMMARY OF THE IMPACT ASSESSMENT Accompanying the document Proposal for a Regulation of the European
EUROPEAN COMMISSION Brussels, 26.6.2013 SWD(2013) 235 final COMMISSION STAFF WORKING DOCUMENT EXECUTIVE SUMMARY OF THE IMPACT ASSESSMENT Accompanying the document Proposal for a Regulation of the European
