Gastrointestinal Involvement in Spinal Cord Injury: a Clinical Perspective
|
|
|
- Mervyn Atkins
- 8 years ago
- Views:
Transcription
1 Gastrointestinal Involvement in Spinal Cord Injury: a Clinical Perspective Ellen Ebert University of Medicine and Dentistry New Jersey, Robert Wood Johnson Medical School, New Brunswick, NJ, USA Abstract Bowel problems occur in 27% to 62% of patients with spinal cord injuries (SCI), most commonly constipation, distention, abdominal pain, rectal bleeding, hemorrhoids, bowel accidents, and autonomic hyperreflexia. The acute abdomen, with a mortality of 9.5%, does not present with rigidity or absent bowel sounds but rather with dull/poorlylocalized pain, vomiting, or restlessness, with tenderness, fever, and leukocytosis in up to 50% of patients. Fecal impaction may present with anorexia and nausea. Methods used for bowel care include laxatives, anal massage, manual evacuation, and enemas. Randomized, double-blind studies demonstrated the effectiveness of neostigmine, which increases cholinergic tone, combined with glycopyrrolate, an anticholinergic agent with minimal activity in the colon that reduces extracolonic side-effects. Improved bowel function occurs with anterior sacral root stimulators which may be combined with an S2 to S4 posterior sacral rhizotomy which interrupts the reflex arc by cutting the posterior roots carrying the spasticity-causing sensory nerves. For severe constipation, a colostomy reduces time for bowel care, providing a clean environment so decubitus ulcers may heal. Gallstones occur in 17% to 31% of patients, and acalculous cholecystitis in 3.7% of patients with acute SCI. A high index of suspicion is needed to properly diagnose bowel problems in SCI. Keywords Fecal impaction constipation acute abdomen neurogenic bowel. Received: Accepted: J Gastrointestin Liver Dis March 2012 Vol. 21 No 1, Address for correspondence: Ellen Ebert UMDNJ - Robert Wood Johnson Medical School New Brunswick, NJ USA jeydels@yahoo.com Introduction There are about 200,000 patients, mostly male, with spinal cord injury (SCI) in the United States with 10,000 to 15,000 new cases diagnosed annually [1]. One-third are tetraplegic, and 50% have complete lesions [2]. The mean age is 33 years old. With improvements in critical care and rehabilitation, SCI patients are living longer and becoming functional members of society. There are analogies between the genitourinary and the gastrointestinal (GI) tracts. Both are organs controlled by a combination of smooth internal and striated external sphincters. Their innervation, blood supply, and response to cholinergic and adrenergic stimuli are also similar [3]. Patients with difficulty in micturition are likely to have difficulties with defecation. Gastrointestinal tract involvement in SCI Bowel problems occur in 27% to 62% of SCI patients [3, 4], associated with depression or with injuries of highest levels, most severity, and longest duration [5]. Complaints include constipation (42-81%), distension (43-53%), incontinence (0-75%), abdominal pain (14-38%), bowel accidents (27%), nausea (19%), diarrhea (14%), straining (20%), rectal bleeding (40%), hemorrhoids (36%-75%), autonomic hyperreflexia (AH) (43%), and headaches or sweats relieved by a bowel movement (13-27%) [5-11]. Constipation is most common with complete lesions [9], previous urinary outlet surgery, and use of anticholinergic agents [6, 8, 11]. Fecal incontinence is associated with complete lesions [10] or anxiety [12]. Whether constipation [7, 10] or fecal incontinence [7, 12] varies with the level of injury is controversial. Abdominal pain may be exacerbated by eating and often improves with a bowel movement or passing flatus; it is associated with constipation and distention [3]. Abdominal distension, if severe, may cause restricted ventilation particularly with high cervical cord injuries. Gastrointestinal complications occur in 1.9% to 11% of patients [13, 14], and undiagnosed abdominal emergencies
2 76 Ebert E account for 10% of all fatalities among patients with SCI [15]. Risk factors include increasing age, male sex, head injury, multisystem trauma, and cervical spine injury [14]. Most complications occur within days after the injury, mainly GI hemorrhage, perforation, ileus, and intestinal obstruction [13, 14]. The lifetime prevalence of peptic ulcer disease is 10%, similar to the general population [3]. Spinal shock occurs for several weeks after injury when all autonomic and reflex activities are lost below the level of cord transaction. During the first four weeks, 4.7% have acute abdominal pathology [16], while 4.2% have acute gastroduodenal ulceration and hemorrhage [17], mainly with SCI above the sympathetic outflow. This may be related to the increase in serum gastrin levels seen within the first 12 to 48 hrs after paralysis caused by penetrating CNS injuries but not after closed-head injuries without paralysis [18]. The abdomen is distended and flaccid with no sensation below the level of the injury. Acute gastric dilatation with high cervical cord lesions may occur at this time [19]. There are absent or hypoactive bowel sounds due to a paralytic ileus which resolves within a week in most cases. Acute abdominal pathology is difficult to diagnose during this period without a high index of suspicion. The rectum should be evacuated using manual disimpaction or enemas [20]. Enteral feeding is safe in patients with acute SCI, the most common reason for interruption being large gastric aspirates. When spinal shock ends, there is a return and then exaggeration of reflex activity below the level of the injury. Autonomic hyperreflexia (AH) or dysreflexia, a condition often missed by medical personnel, is a potentially fatal syndrome in those whose SCI lies above T6 or the greater splanchnic outflow [21]. It results in intense reflex vasoconstriction below the level of the SCI, with tachycardia or bradycardia, hypertension, diaphoresis, headaches, bladder spasms, and diarrhea. It is triggered by processes distending a hollow viscus associated with pain or irritation with the colorectal area being the second most common trigger after bladder irritation. Anoscopy, with stretching of the anal sphincter, is a more potent stimulus than flexible sigmoidoscopy, which causes gaseous distension of the rectosigmoid, although both can raise blood pressure and trigger AH. Intersphincteric anal block with lidocaine before anoscopy or sigmoidoscopy may reduce the incidence of this syndrome. The acute abdomen has a 9.5% mortality in patients with SCI [22]. The loss of sensory, motor, and reflex functions early on after SCI or in patients with high SCI masks the usual signs of an acute abdomen, such as pain, tenderness, rigidity and guarding. Early clues to abdominal disease are nausea, vomiting, anorexia, malaise, restlessness, a vague feeling of being unwell, changes in the normal daily pattern of living, bowel or bladder incontinence, bladder spasms, or diarrhea [23]. Pain is present in 64% of patients, more common in low than high SCI [24]. It may be dull and poorly-localized from slow conduction velocities of unmyelinated class C fibers. A burning pain, localized at the border between sensate and insensate skin (described as a tight belt ) may represent a neurologic, dysesthetic pain syndrome. Pain may be referred to distant skin areas of the dermatome supplying the involved viscera due to convergence of visceral and somatic afferents onto the same spinal segments. Such radicular pain has a burning quality, and the skin is hypersensitive to pinprick. Shoulder pain occurs in 32%, particularly in high SCI [24]. This is due to pathologic processes irritating the diaphragm, perceived as pain in the neck or shoulder by way of afferent fibers of the phrenic nerve. As the process extends to the peritoneum, the pain may become sharp from transmission by the high conduction velocities of the large myelinated somatic sensory fibers. On physical exam, 32% are febrile, 27% are distended (all with high SCI), 50% have tenderness to palpation, 18% have decreased or absent bowel sounds, and 14% have rigidity or spasm (all with high SCI) [24]. A rise in pulse or a sudden temperature increase above F may be an early sign of abdominal pathology. High fevers of F, in contrast, are usually due to urinary tract infections (UTIs) [25], a diagnosis supported by the presence of pyuria. Distension from dilated loops of bowel is common and may be corrected by enemas. However, an increase in the distension or the formation of air-fluid levels may indicate pathology. Palpation of the abdomen is made difficult by spasticity of the abdominal wall muscles [24]. Tenderness is usually absent, but when present, may represent a perforated viscus; its location does not reliably indicate the location of the pathology. Leukocytosis is found in 55% of patients and radiographic studies leading to the diagnosis in 61.9% to 77% [22, 24]. Leukocytosis and fever are often ascribed to UTIs, soft tissue infections, or pneumonia. Peritoneal lavage showing cloudy fluid containing leukocytes may lead to early diagnosis of an abdominal catastrophe. Acute abdominal pathology includes a perforated viscus (such as peptic ulcer disease), cholecystitis [24], appendicitis, and bowel obstruction [23]. Appendicitis presents with abdominal discomfort and distention [26]. Signs of AH with SCI above T6, fever, and right lower quadrant abdominal mass may be present but rarely tenderness. The delay in diagnosis results in a perforated appendix in 92% to 100% of patients. Laparoscopy may hasten the diagnosis, thus reducing mortality and morbidity. Abdominal operations are challenging in patients with SCI. Patients may experience a drop in blood pressure with induction of anesthesia. Some patients have deformities, contractures, stomas, or spasticity that can technically compromise the procedures. In addition, they may be debilitated and chronically ill so that they make poor operative candidates. Retention sutures may be needed to close abdominal wounds due to increased tension that can occur from abdominal spasticity. In addition, the patients may have hyperreflexic hypertension, decreased pulmonary excursion, particularly in high-level lesions, and prolonged ileus due to poor visceral muscle tone. Pulmonary complications increase the morbidity in those undergoing surgery for appendicitis [26]. Patients often have chronic septic foci, such as UTIs, bronchitis, and decubitus ulcers that increase their risk of wound infections. If carefully
3 Gastrointestinal involvement in spinal cord injury 77 managed, however, the morbidity may be similar to that of normal individuals. Pathophysiology of bowel dysfunction With rectal distension, the smooth muscles of the internal anal sphincter (IAS) normally relax (rectoanal inhibitory reflex or RAIR). With rectal contractions, continence is maintained by voluntary contraction of the skeletal muscles of the external anal sphincter (EAS). Defecation occurs when the puborectalis muscle and the EAS relax. Neurogenic bowel consists of upper motor neuron (UMN) and lower motor neuron (LMN) lesions [27]. With UMN lesions above the conus medullaris (S2-S4), the defecatory maneuver cannot be performed due to lack of functioning abdominal musculature [28]. Patients with SCI above T5 generate abdominal pressure by intercostal and diaphragmatic contraction, while those with cervical lesions rely totally on diaphragmatic contraction [29]. Rectal distension causes brief EAS activityin high cord lesions, so that leakage of fluid may not occur. It also causes autonomic symptoms, such as flushing and sweating, above the level of the lesion. The bowel is hyperreflexic with a dysrhythmic or irregular electrical rhythm on electrorectogram [30]. The tone of the colonic wall increases. The voluntary control of the EAS is discontinued so the sphincter remains tight, thereby retaining stool. The result of the UMN bowel syndrome is constipation and mild incontinence. Half the patients have bowel movements on the toilet and almost half in bed. The LMN bowel syndrome or the areflexic bowel is due to a lesion in the conus medullaris (S2-S4), the cauda equina, or the pelvic nerve. Patients have voluntary control of part or all of their abdominal muscles. Electrorectogram shows no electrical events [30]. There is no spinal cord-mediated peristalsis but rather slow stool propulsion. The myenteric plexus coordinates segmental colonic peristalsis resulting in dry, round stools. The EAS is denervated, increasing the risk of incontinence. The levator ani muscles lack tone so the rectal angle is reduced causing the lumen of the rectum to open. The final result of the LMN lesion is constipation and incontinence-increased frequency of defecation [3] but with increased time required for defecation. Patients use the Valsalva maneuver and manual removal of stool from the rectum.with the loss of sphincter tone, the rectum must be kept empty by removal of feces with the finger to reduce incontinence. Low paraplegic patients can empty their bowels in a sitting position which reduces the anorectal angle and facilitates defecation. They have partially intact abdominal muscles needed to expel feces [8]. Patients with SCI have normal [28, 32], decreased [33], or increased [34] anal sphincter pressures. RAIR is normal in SCI since it is an entero-enteric reflex dependent on the myenteric nervous system and independent of spinal control [28, 34]. Phasic giant rectal contractions increase in SCI, perhaps predisposing to fecal incontinence [35]. From 19% to 42% of patients have a desire to defecate [10]. Signals of an upcoming defecation include abdominal pressure with incomplete lesions and unspecified abdominal complaints with complete lesions, such as a prickling sensation, increased spasticity, cutis anserine (goose bumps), and sweating [36]. The gastrocolic reflex, the increase in colonic motility with food, may be present [32] or reduced [37,38]. This response was seen only in the descending, not the rectosigmoid, colon [37] suggesting that differences in the studies may be due to placement of the recording electrodes. Other authors found an exaggerated response in the rectum and claimed that attaching recording clips to the mucosa in other studies inhibited bowel motility [32]. A normal pressure/volume relationship during ramp inflation of the rectal balloon causes a reversed-s shape with increase in pressure initially, followed by a plateau, and then further increase in pressure as the volume increases [29]. In SCI patients, however, this pressure/volume relationship is linear, indicating a tighter and less compliant rectum at high volumes. Using radio-opaque markers or ph-sensitive radiolabelled exchange resin pellets, prolonged transit time may be pancolonic [39] or predominantly in the left colon and rectum [28]. Transit times may [40] or may not [41] correlate with symptoms. Involvement of the esophagus, stomach, and duodenum in SCI Dysphagia is present in 22.5% to 30% of patients with SCI, related to age, presence of a tracheostomy, mechanical ventilation, and cervical spinal surgery, especially the anterior approach [42]. Factors predisposing to heartburn include delayed gastric emptying (GE), recumbency, immobilization, and certain drugs (such as anticholinergics and meperidine). Although heartburn may not be more common in SCI, these patients may undergo endoscopy less frequently than normal individuals and esophagitis is seen more commonly [43].While resting upper and lower esophageal sphincter pressures may be normal, there is incomplete upper esophageal sphincter relaxation in some patients, predisposing them to aspiration [44]. As the posterior wall of the pharynx lies adjacent to the cervical vertebrae, cervical trauma may impinge upon or even perforate the pharynx. Gastric emptying is either delayed [45, 46] or normal [47] in SCI patients. This discrepancy may be due to the different test meals used and whether the patients are supine [45] or sitting [47]. Gastric emptying times increase directly with the higher level of the SCI [45, 46] and the increased duration of injury [45]. Intravenous metoclopramide, a potent dopamine receptor antagonist with prokinetic properties, corrects impaired GE [47]. Functional magnetic stimulation decreases GE times by more than one-third in SCI patients with preserved LMN function, particularly during the first 30 min [48]. Superior mesenteric artery syndrome is a condition in which the third portion of the duodenum is intermittently compressed by the overlying superior mesenteric artery.
4 78 Ebert E Predisposing factors include rapid weight loss (with loss of mesenteric fat that shields the duodenum as it passes behind the superior mesenteric artery and in front of the lumbar spine and aorta), prolonged supine positioning, and the use of a spinal orthosis, all of which are common with acute quadriplegic patients [49]. Symptoms include epigastric pain and vomiting, especially when supine. A barium study shows a cutoff between the third and fourth portions of the duodenum in the supine position that disappears when the patient is upright. Treatment is upright body positioning, weight gain, and in resistant cases, a duodenojejunostomy. Involvement of the colon in SCI Fecal impaction is a common GI complication in SCI [13]. Patients may present with loss of appetite and nausea and are mistakenly given antinausea preparations, most of which have anticholinergic and constipating properties. Liquid stool may pass around the blockage. Plain films of the abdomen show the feces and abnormal air patterns. Digital removal of the stool should be done. Patients may develop stercoral ulcers in the rectum from pressure due to stool. Megacolon occurs in 18% or 73% of patients, depending on how it is defined [50, 51]; small bowel dilatation found in 39% of patients may be due to stool retention most prominent in the ascending colon [50]. Colonic volvulus, reported in 2.6% of hospitalized patients, is a common cause of intestinal obstruction in patients with chronic constipation due to the long redundant colon [52]. Hemorrhoids and hemorrhoidectomy are probably more prevalent in SCI than in normal individuals, particularly in those who strain and use suppositories, enemas, or chemical stimulation for bowel management [7, 9]; hemorrhoidal bleeding occurs in up to 76% of patients [3, 53]. Conservative therapy, banding, and sclerotherapy of hemorrhoids reduces bleeding and/or symptoms [54]. Colorectal carcinoma when discovered is more advanced in SCI than in non-paralyzed subjects and morbidity after surgery may be higher [55]. A delay in diagnosis may occur since the distension, constipation, and pain may be attributed to GI complications of SCI; in addition, the difficulty of bowel preparation in these patients may result in an avoidance of screening colonoscopies. Treatment of bowel problems Bowel management involves increasing fiber intake to maintain proper stool consistency, ensuring adequate fluid intake, and providing bowel training so that the colon is evacuated at regular intervals. This is best accomplished after breakfast to take advantage of the gastrocolic reflex which peaks about 30 minutes after eating. One study surprisingly showed that the mean colonic transit time increased and the evacuation time did not change with the addition of dietary fiber [56]. Methods used for bowel care include anal massage (35%), unaided self-defecation with or without oral medication and abdominal massage (29%), digital stimulation (18 to 82%), rectal suppositories (15 to 74%), oral laxatives (29 to 41%), supplemental fiber (17 to 43%), manual evacuation (65 to 68%), and enemas (11 to 89%), [4, 9,10]. A sequential schedule of evacuating stimuli reduces total but not segmental transit times, decreases the need for oral or rectal laxatives, and increases the frequency of bowel movements [57]. The bisacodyl suppository is the most commonly used laxative, while docusate is the most popular oral agent [6]. Polyethylene glycol-based bisacodyl suppositories or polyethylene glycol-based, glycerine, docusate sodium minienemas result in a decrease in the amount of time for bowel care, a shorter total defecation period, and stimulation of bowel reflex defecation sooner than hydrogenated vegetable oil-based bisacodyl suppositories [58]. Digital stimulation of the anal canal serves to manually disimpact the rectum, to dilate the anal canal, and to relax the puborectalis muscle; the last effect decreases the anorectal angle and reduces outflow resistance to the passage of stool, resulting in leftsided colonic activity [59]. A gloved finger in the rectum inducing sustained pressure towards the sacrum, relaxes the spastic EAS and pelvic muscles [60]. Rotation of the finger continues the stimulation until a reflex peristaltic wave is generated in the rectum, resulting in passage of flatus and stool. The resulting RAIR relaxes the IAS, while the rectocolic reflex stimulates pelvic nerve-mediated peristalsis. When the abdomen is massaged, following the course of the colon in an aboral direction, intraabdominal pressure increases [20]. Massage may improve abdominal distension, fecal incontinence, number of bowel movements, and mean total colonic transit time [61]. Megacolon may contribute to reduced responsiveness to both laxative therapy and physical interventions [62]. Saline enemas increase the proportion of continent stools and decrease the number of constipated stools although there is a significant drop-out rate due to stressful life events, inability to retain the balloon, and AH [63]. Due to the difficulty in retaining enemas from the loss of voluntary control of the EAS, the enemas should be placed above the rectosigmoid junction. With transanal irrigation, performed daily or every other day, a rectal catheter with an inflatable balloon holds the catheter in place while the enema is administered [64]. It empties the rectosigmoid and descending colon [64]. In a randomized, controlled trial where it was compared to conservative bowel management, transanal irrigation improves quality of life and the neurogenic bowel dysfunction score which includes frequency of bowel movements, time for defecation, frequency of digital stimulation, and fecal incontinence [64, 65]. After 3 years, however, the rate of success is only 35% [66]. The main reason for failure is leakage around the catheter, expulsion of the catheter, or bursting of the balloon [64]. The Malone Antegrade Continence Enema is an operation whereby the appendix is brought out to the skin forming an appendicostomy. Through the small stoma, patients can introduce a catheter and administer an enema that washes out the colon and rectum [67]. The overall success rate in
5 Gastrointestinal involvement in spinal cord injury 79 patients with mixed neurological diseases including SCI who were dissatisfied with conservative treatment was 74% and 87.5% after a mean follow-up period of 38 and 75 months, respectively [67,68]. Complications occur in 12.5% and 67% of patients, including wound infection, small bowel obstruction, and stomal stenosis. Cisapride acts locally on motor activity by facilitating the release of acetylcholine at the myenteric plexus.in two cross-over trials, cisapride did not alter the number of bowel movements, ease of evacuation, or oro-anal transit time [69, 70], although one study showed improvement in stool consistency [70]. Cisapride has since been taken off the market due to ventricular arrhythmias. Prucalopride, a serotonin 4 receptor agonist with enterokinetic properties, reduces total colonic transit time with an increase in bowel movements but does not affect other bowel care related parameters [71]. Adverse events include flatulence, abdominal pain, diarrhea, and headache, causing some patients to discontinue treatment. Neostigmine, which increases cholinergic tone by blocking the metabolism of acetylcholine by acetylcholinesterase, for bowel care in SCI patients was tested in a randomized, blinded design comparing intravenously neostigmine alone, neostigmine with glycopyrrolate, or normal saline [72]. Glycopyrrolate is an anticholinergic agent that reduces the side effects of bradycardia and bronchoconstriction caused by neostigmine with limited activity on the muscarinic receptors of the colon. Prompt bowel evacuation occurs with neostigmine alone or neostigmine with glycopyrrolate. In a subsequent double-blind, cross-over study, intramuscular neomycin/ glycopyrrolate given during three consecutive bowel evacuation sessions reduced total bowel evacuation times compared to placebo [73]. Trials are in progress to test the efficacy of intranasal neomycin and glycopyrrolate [74]. Fampridine, a selective potassium channel blocker, increases the number of days with a bowel movement in about onefifth of the subjects in a double-blind randomized placebocontrolled trial, also improving spasticity as well as bladder and sexual function [75]. For sacral nerve stimulation, an anterior sacral root nerve stimulator is implanted at the level of the cauda equina. This is usually preceded by a test period using a percutaneous needle electrode [76, 77]. This method causes increased perianal sensitivity and improved deliberate retention of feces [77]. It affects the left colon increasing frequency of defecation [78, 79], decreasing time dedicated to bowel movements [79], decreasing fecal incontinence [80], and improving quality of life. There may or may not be an increase in rectal and anal pressures [80, 81] and anorectal measurements do not predict which patients will benefit from this treatment [82]. Colonic transit times do not change [78]. Anterior sacral root stimulators can be combined with an S2 to S4 posterior sacral rhizotomy which interrupts the reflex arc by cutting the posterior roots carrying the spasticity-causing sensory nerves. It results in improved bowel function and increased bladder capacity and emptying [83]. With elimination by rhizotomy of spinal reflexes that control continence and defecation, incontinence is no longer a problem. Although reflex defecation is then impossible, evacuation of stools is more predictably induced by manual evacuation or the use of anterior root stimulators. Noninvasive magnetic stimulation, which produces an electrical field sufficient to depolarize nerves, increases anal and rectal pressures, decreases rectal volume [84], decreases mean colonic transit time, and improves bowel habits [85]. Surgical placement of a colostomy or ileostomy, the main indication being constipation, reduces the time spent in bowel care, increases independence, prevents fecal incontinence, relieves abdominal distension, enhances or maintains quality of life, and reduces hospitalizations for chronic bowel dysfunction by 70% according to pre-post or retrospective studies [86-89]. It provides a dry, clean environment so that pressure decubitus ulcers may heal. Most patients wished that the colostomy had been offered sooner, and none wanted it reversed [88]. Colon transit time and anorectal manometry can be used to identify the severity of bowel dysfunction and the site for the colostomy [89]. A left-sided colostomy with a Hartman s pouch is probably the most common approach used [87]. Management of complicated decubitus ulcers and perineal and pelvic wounds is the primary indication in 43% of patients with left-sided colostomies. A right-sided colostomy is less likely to have problems with emptying but results in more watery stools, more frequent stoma care, and increased risk of leakage compared to a left-sided stoma. The long segment of defunctionalized colon predisposes to diversion colitis and mucous impaction which can be prevented by a distal colonic resection, a procedure that increases operative morbidity. The most common complications of colostomies are leakage of mucus per rectum, stomal prolapse or parastomal hernias, bowel obstruction, and wound dehiscence [86, 89]. Gallbladder involvement in SCI Gallstones or previous cholecystectomy occur in 17-31% of SCI patients [90-92], some of whom have risk factors such as diabetes, obesity and/or family history of gallbladder disease [90]. The prevalence may be high even if patients with risk factors are excluded [91, 92]. Ultrasound examinations found gallstones in 25% or 26% of asymptomatic males compared to a control of 9% or 10% [93, 94]. Ultrasound exams performed within the first 6 months after SCI demonstrates a high incidence of biliary sludge (19%) but not gallstones (8%); all stones developed after four months and were small and not strongly echogenic. Cholelithiasis does not correlate with neurological level of injury, duration of SCI, age, obesity, or diabetes mellitus [94] although it may correlate with the severity of the SCI lesion [90]. The reason for the increase in gallstones in SCI is unknown. Gallbladder fasting volume is low, probably from impaired gallbladder filling due to impaired relaxation, generally with SCI lesions above T10 [95]. Ejection fraction
6 80 Ebert E is low especially in lesions above T10 [95]; this may be due to low resting volumes rather than a defect in contractility. By ultrasound, only 38% have abnormal emptying fractions but this was associated with diabetes and obesity. Contraction of the gallbladder is probably mediated by the vagus nerve and cholecystokinin, both intact in SCI patients. Gallstones may also be due to gastroduodenal and colonic motility disorders seen in these patients [96], altering bile acid pool size and biliary lipid secretion [97]. Gallstones may be due to events that occur around the time of the injury, such as rapid weight loss [90]. While there is no association between gallstone formation and body mass index (BMI) in SCI, the BMI may underestimate the percentage of body fat due to osteopenia and loss of muscle mass from severe atrophy of limbs. Due to the loss of normal sensory perception, the symptoms and signs of gallbladder disease may be atypical or even absent so that pathologic processes are often advanced at diagnosis [11]. Other authors indicate that the presentation is normal [98] and that atypical GI symptoms (bloating, nausea, and abdominal pain not found in the right upper quadrant or epigastrium) are not associated with gallstones [98]. Localization of pain was found in one study but in others only occurred in those with low SCI [11, 99]. Acute acalculous cholecystitis, found in 3.7% of patients with acute SCI admitted to the Intensive Care Unit of a referral hospital [100], presents with a right upper quadrant mass (100% of patients), steady fever (85.7%), tachycardia (100%), and cholestasis (85.7%). Predisposing factors include narcotics, mechanical ventilation with positive endexpiratory pressure, and hyperalimentation. Complications include a gangrenous gallbladder and an inflammatory mass. The need for cholecystectomy is normal if operations done prophylactically are removed from consideration (90). Although mortality rates are comparable to control individuals, SCI patients have a higher complication rate and a higher conversion from laparoscopic to open procedures. Some authors, whose threshold for elective cholecystectomy is low [90, 99], support routine screening ultrasounds to alert the clinician to the presence of gallbladder disease, while others believe that no specific interventions are warranted based on the findings. Conclusion A high index of suspicion is required to diagnose gastrointestinal involvement in SCI due to impaired sensory and motor functions. Conflicts of interest The author has no conflicts of interest regarding this article. References 1. Spinal cord injury: facts and figures at a glance. J Spinal Cord Med 2000;23: Wyndaele M, Wyndaele JJ. Incidence, prevalence and epidemiology of spinal cord injury: what learns a worldwide literature survey? Spinal Cord 2006;44: Stone JM, Nino-Murcia M, Wolfe VA, Perkash I. Chronic gastrointestinal problems in spinal cord injury patients: a prospective analysis. Am J Gastroenterology 1990;85: Han TR, Kim JH, Kwon BS. Chronic gastrointestinal problems and bowel dysfunction in patients with spinal cord injury. Spinal Cord 1998;36: Liu CW, Huang CC, Chen CH, Yang YH, Chen TW, Huang MH. Prediction of severe neurogenic bowel dysfunction in persons with spinal cord injury. Spinal Cord 2010;48: Harari D, Sarkarati M, Gurwitz JH, McGlinchey-Berroth G, Minaker KL. Constipation-related symptoms and bowel program concerning individuals with spinal cord injury. Spinal Cord 1997;35: Menter R, Weitzenkamp D, Cooper D, Bingley J, Charlifue S, Whiteneck G. Bowel management outcomes in individuals with long-term spinal cord injuries. Spinal Cord 1997;35: DeLooze D, Van Laere M, DeMuynck M, Beke R, Elewaut A. Constipation and other chronic gastrointestinal problems in spinal cord injury patients. Spinal Cord 1998;36: Lynch AC, Wong C, Anthony A, Dobbs BR, Frizelle FA. Bowel dysfunction following spinal cord injury: a description of bowel function in a spinal cord-injured population and comparison with age and gender matched controls. Spinal Cord 2000;38: Krogh K, Nielsen J, Djurhuus JC, Mosdal C, Sabroe S, Laurberg S. Colorectal function in patients with spinal cord lesions. Dis Colon Rectum 1997;40: Chen CY, Chuang TY, Tsai YA, et al. Loss of sympathetic coordination appears to delay gastrointestinal transit in patients with spinal cord injury. Dig Dis Sci 2004;49: Ng C, Prott G, Rutkowski S, et al. Gastrointestinal symptoms in spinal cord injury: relationships with level of injury and psychologic factors. Dis Colon Rect 2005;48: Gore RM, Mintzer RA, Calenoff L. Gastrointestinal complications of spinal cord injury. Spine 1981; 6: Matsumura JS, Prystowsky JB, Bresticker MA, Meyer PR Jr, Joehl RJ, Nahrwold DL. Gastrointestinal tract complications after acute spine injury. Arch Surg 1996;130: Juler GL, Eltorai IM. The acute abdomen in spinal cord injury patients. Paraplegia 1985;23: Berlly MH, Wilmot CB. Acute abdominal emergencies during the first four weeks after spinal cord injury. Arch Phys Med Rehabil 1984;65: Kewalramani LS. Neurogenic gastroduodenal ulceration and bleeding associated with spinal cord injuries. J Trauma 1979;19: Bowen JC, Fleming WH, Thompson JC. Increased gastrin release following penetrating central nervous system injury. Surgery 1974;75: Sutton RA, Macphail I, Bentley R, Nandy MK. Acute gastric dilatation as a relatively late complication of tetraplegia due to very high cervical cord injury. Paraplegia 1981;19: Longo WE, Ballantyne GH, Modlin IM. The colon, anorectum, and spinal cord patient. A review of the functional alterations of the denervated hindgut. Dis Colon Rectum 1989;32: Cosman BC, Vu TT. Lidocaine anal block limits autonomic dysreflexia during anorectal procedures in spinal cord injury: a randomized, double-blind, placebo-controlled trial. Dis Colon Rectum 2005;48: Miller BJ, Geraghty TJ, Wong CH, Hall DF, Cohen JR. Outcome of the acute abdomen in patients with previous spinal cord injury.anz J Surg 2001;71:
7 Gastrointestinal involvement in spinal cord injury Bar-On Z, Ohry A. The acute abdomen in spinal cord injury individuals. Paraplegia 1995;33: Neumayer LA, Bull DA, Mohr JD, Putham CW. The acutely affected abdomen in paraplegic spinal cord injury patients. Ann Surg 1990;212: Sugarman B, Brown D, Musher D. Fever and infection in spinal cord injury patients. JAMA 1982;248: Strauther GR, Longo WE, Virgo KS, Johnson FE. Appendicitis in patients with previous spinal cord injury. Am J Surg 1999;178: Nino-Murcia M, Friedland GW. Functional abnormalities of the gastrointestinal tract in patients with spinal cord injuries: evaluation with imaging procedures.ajr Am J Roentgenol 1992;158: Valles M, Mearin F. Pathophysiology of bowel dysfunction in patients with motor incomplete spinal cord injury: comparison with patients with motor complete spinal cord injury. Dis Colon Rectum 2009;52: MacDonagh R, Sun WM, Thomas DG, Smallwood R, Read NW. Anorectal function in patients with complete supraconal spinal cord lesions. Gut 1992;33: Shafik A. Electrorectogram study of the neuropathic rectum. Paraplegia 1995;33: Yim SY, Yoon SH, Lee IY, Rah EW, Moon HW. A comparison of bowel care patterns in patients with spinal cord injury: upper motor neuron bowel vs lower motor neuron bowel. Spinal Cord 2001;39: Walter SA, Morren GL, Ryn AK, Hallbook O. Rectal pressure response to a meal in patients with high spinal cord injury. Arch Phys Med Rehabil 2003;84: Greving I, Tegenthoff M, Nediat S, et al. Anorectal functions in patients with spinal cord injury. Neurogastroenterol Motil 1998;10: Beuret-Blanquart F, Weber J, Gouverneur JP, Demangeon S, Denis P. Colonic transit time and anorectal manometric anomalies in 19 patients with complete transaction of the spinal cord. J Auton Nerv Syst 1990;30: Krogh K, Mosdal C, Gregersen H, Laurberg S. Rectal wall properties in patients with acute and chronic spinal cord lesions. Dis Colon Rectum 2002;45: Haas U, Geng V. Sensation of defecation in patients with spinal cord injury. Spinal Cord 2008;46: Fajardo NR, Pasiliao RV, Modeste-Duncan R, Creasey G, Bauman WA, Korsten MA. Decreased colonic motility in persons with chronic spinal cord injury. Am J Gastroenterol 2003;98: Aaronson MJ, Freed MM, Burakoff R. Colonic myoelectric activity in persons with spinal cord injury. Dig Dis Sci 1985;30: Keshavarzian A, Barnes WE, Bruninga K, Nemchausky B, Mermall H, Bushnell D. Delayed colonic transit in spinal cord-injured patients measured by indium-111 Amberlite scintigraphy. Am J Gastroenterol 1995;90: De Looze DA, De Muynck MC, Van Laere M, De Vos MM, Elewaut AG. Pelvic floor function in patients with clinically complete spinal cord injury and its relation to constipation. Dis Colon Rectum 1998;41: Media S, Christensen P, Lauge I, Al-Hashimi M, Laurberg S, Krogh K. Reproducibility and validity of radiographically determined gastrointestinal and segmental colonic transit times in spinal cordinjured patients. Spinal Cord 2009; 47: Kirshblum S, Johnston MV, Brown J, O Connor KC, Jarosz P. Predictors of dysphagia after spinal cord injury. Arch Phys Med Rehabil 1999;80: Singh G, Triadafilopoulos G. Gastroesophageal reflux disease in patients with spinal cord injury. J Spinal Cord Med : Neville AL, Crookes P, Velmahos GC, Vlahos A, Theodorou D, Lucas CE. Esophageal dysfunction in cervical spinal cord injury: a potentially important mechanism of aspiration. J Trauma 2005;59: Segal JL, Milne N, Brunnemann SR, Lyons KP. Metoclopramideinduced normalization of impaired gastric emptying in spinal cord injury. Am J Gastroenterol 1987;82: Kao CH, Ho YJ, Changlai SP, Ding HJ. Gastric emptying in spinal cord injury patients. Dig Dis Sci 1999;44: Zhang RL, Chayes Z, Korsten MA, Bauman WA. Gastric emptying rates to liquid or solid meals appear to be unaffected by spinal cord injury. Am J Gastroenterol 1994;89: Lin VW, Kim KH, Hsiao I, Brown W. Functional magnetic stimulation facilitates gastric emptying. Arch Phys Med Rehabil 2002;83: Roth EJ, Fenton LL, Gaebler-Spira DJ, Frost FS, Yarkony GM. Superior mesenteric artery syndrome in acute traumatic quadriplegia: case reports and literature review. Arch Phys Med Rehabil 1991;72: Harari D, Minaker KL. Megacolon in patients with chronic spinal cord injury.spinal Cord 2000;38: Faaborg PM, Christensen P, Rosenkilde M, Laurberg S, Krogh K. Do gastrointestinal transit times and colonic dimensions change with time since spinal cord injury? Spinal Cord 2011;49: Fenton-Lee D, Yeo BW, Jones RF, Engel S. Colonic volvulus in the spinal cord injured patient. Paraplegia 1993;31: Connell AM, Frankel H, Guttmann L. The motility of the pelvic colon following complete lesions of the spinal cord. Paraplegia 1963;1: Scott D, Papa MZ, Sareli M, Velano A, Ben-Ari GY, Koller M. Management of hemorrhoidal disease in patients with chronic spinal cord injury. Tech Coloproctol 2002;6: Stratton MD, McKirgan LW, Wade TP, et al. Colorectal cancer in patients with previous spinal cord injury. Dis Colon Rectum 1996;39: Cameron KJ, Nyulasi IB, Collier GR, Brown DJ. Assessment of the effect of increased dietary fibre intake on bowel function in patients with spinal cord injury. Spinal Cord 1996;34: Badiali D, Bracci F, Castellano V, et al. Sequential treatment of chronic constipation in paraplegic subjects. Spinal Cord 1997;35: House JG, Stiens SA. Pharmacologically initiated defecation for persons with spinal cord injury: effectiveness of three agents. Arch Phys Med Rehabil 1997;78: Korsten MA, Singal AK, Monga A, et al. Anorectal stimulation causes increased colonic motor activity in subjects with spinal cord injury. J Spinal Cord Med 2007;30: Lynch AC, Anthony A, Dobbs BR, Frizelle FA. Anorectal physiology following spinal cord injury. Spinal Cord 2000;38: Ayas S, Leblebici B, Sozay S, Bayramoglu M, Niron EA. The effect of abdominal massage on bowel function in patients with spinal cord injury. Am J Phys Med Rehabil 2006;85: Coggrave MJ, Norton C. The need for manual evacuation and oral laxatives in the management of neurogenic bowel dysfunction after spinal cord injury: a randomized controlled trial of a stepwise protocol. Spinal Cord 2010;48: Liptak GS, Revell GM. Management of bowel dysfunction in children with spinal cord disease or injury by means of the enema continence catheter. J Pediatr 1992;120:
8 82 Ebert E 64. Christensen P, Bazzocchi G, Coggrave M, et al. A randomized, controlled trial of transanal irrigation versus conservative bowel management in spinal cord-injured patients. Gastroenterology 2006;131: Krogh K, Christensen P, Sabroe S, Laurberg S. Neurogenic bowel dysfunction score. Spinal Cord 2006;44: Faaborg PM, Christensen P, Kvitsau B, Buntzen S, Laurberg S, Krogh K. Long-term outcome and safety of transanal colonic irrigation for neurogenic bowel dysfunction. Spinal Cord 2009;47: Teichman JM, Zabihi N, Kraus SR, Harris JM, Barber DB. Longterm results for malone antegrade continence enema for adults with neurogenic bowel disease. Urology 2003;61: Worsoe J, Christensen P, Krogh K, Buntzen S, Laurberg S. Long-term results of antegrade colonic enema in adult patients: assessment of functional results. Dis Colon Rectum 2008;51: Badali D, Corazziari E, Habib FI, Bausano G, Viscardi A, Anzini F, et al. A double-blind controlled trial on the effect of cisapride in the treatment of constipation in paraplegic patients. Neurogastroenterol Motil 1991;3: de Both PSM, de Groot GH, Slootman HR. Effects of cisapride on constipation in paraplegic patients: a placebo-controlled randomized double-blind cross-over study. Eur J Gastroenterol Hepatol 1992;4: Krogh K, Jensen MB, Gandrup P, et al. Efficacy and tolerability of prucalopride in patients with constipation due to spinal cord injury. Scand J Gastroenterol 2002;37: Korsten MA, Rosman AS, Ng A, et al. Infusion of neostigmineglycopyrrolate for bowel evacuation in persons with spinal cord injury. Am J Gastroenterol 2005;100: Rosman AS, Chaparala G, Monga A, Spungen AM, Bauman WA, Korsten MA. Intramuscular neostigmine and glycopyrrolate safely accelerated bowel evacuation in patients with spinal cord injury and defecatory disorders. Dig Dis Sci 2008;53: ClinicalTrials.gov. Intranasal administration of neostigmine and glycopyrrolate for bowel evacuation. show/nct Cardenas DD, Ditunno J, Graziani V, et al. Phase 2 trial of sustainedrelease fampridine in chronic spinal cord injury. Spinal Cord 2007;45: Krogh K, Christensen P. Neurogenic colorectal and pelvic floor dysfunction. Best PractRes Clin Gastroenterol 2009;23: Gstaltner K, Rosen H, Hufgard J, Mark R, Schrei K. Sacral nerve stimulation as an option for the treatment of faecal incontinence in patients suffering from cauda equine syndrome. Spinal Cord 2008;46: Binnie NR, Smith AN, Creasey GH, Edmond P. Constipation associated with chronic spinal cord injury: the effect of pelvic parasympathetic stimulation by the Brindley stimulator. Paraplegia 1991;29: Valles M, Rodriguez A, Borau A, Mearin F. Effect of sacral anterior root stimulator on bowel dysfunction in patients with spinal cord injury. Dis Colon Rectum 2009;52: Holzer B, Rosen HR, Novi G, Ausch C, Holbling N, Schiessel R. Sacral nerve stimulation for neurogenic faecal incontinence. Br J Surg 2007;94: Chia YW, Lee TK, Kour NW, Tung KH, Tan ES. Microchip implants on the anterior sacral roots in patients with spinal trauma: does it improve bowel function? Dis Colon Rectum 1996;39: Lombardi G, Del Popolo G, Cecconi F, Surrenti E, Macchiarella A. Clinical outcome of sacral neuromodulation in incomplete spinal cord-injured patients suffering from neurogenic bowel dysfunctions. Spinal Cord 2010;48: Egon G, Barat M, Colombel P, Visentin C, Isambert JL, Guerin J. Implantation of anterior sacral root stimulators combined with posterior sacral rhizotomy in spinal injury patients. World J Urol 1998;16: Morren GL, Walter S, Hallbook O, Sjodahl R. Effects of magnetic sacral root stimulation on anorectal pressure and volume. Dis Colon Rectum 2001;44: Tsai PY, Wang CP, Chiu FY, Tsai YA, Chang YC, Chuang TY. Efficacy of functional magnetic stimulation in neurogenic bowel dysfunction after spinal cord injury. J Rehabil Med 2009;41: Branagan G, Tromans A, Finnis D. Effect of stoma formation on bowel care and quality of life in patients with spinal cord injury. Spinal Cord 2003;41: Safadi BY, Rosito O, Nino-Murcia M, Wolfe VA, Perkash I. Which stoma works better for colonic dysmotility in the spinal cord injured patient? Am J Surg 2003;186: Kelly SR, Shashidharan M, Borwell B, Tromans AM, Finnis D, Grundy DJ. The role of intestinal stoma in patients with spinal cord injury. Spinal Cord 1999;37: Rosito O, Nino-Murcia M, Wolfe VA, Kiratti BJ, Perkash I. The effects of colostomy on the quality of life in patients with spinal cord injury: a retrospective analysis. J Spinal Cord Med 2002;25: Moonka R, Stiens SA, Resnick WJ, et al. The prevalence and natural history of gallstones in spinal cord injured patients. J Am Coll Surg 1999;189: Ketover SR, Ansel HJ, Goldish G, Roche B, Gebhard RL. Gallstones in chronic spinal cord injury: is impaired gallbladder emptying a risk factor? Arch Phys Med Rehabil 1996;77: Park TH, Byun JH, Suh JH, et al. A study on the incidence of gallstones in patients with spinal cord injury. Korean J Gastroenterol 1998;31: Xia CS, Han YQ, Yang XY, Hong GX. Spinal cord injury and cholelithiasis. Hepatobiliary Pancreat Dis Int 2004;3: Rotter KP, Larrain CG. Gallstones in spinal cord injury (SCI): a late medical complication? Spinal Cord 2003;41: Tandon RK, Jain RK, Garg PK. Increased incidence of biliary sludge and normal gallbladder contractility in patients with high spinal cord injury. Gut 1997;41 : Fealey RD, Szurszewski JH, Merritt JL, DiMagno EP. Effect of traumatic spinal cord transaction on human upper gastrointestinal motility and gastric emptying. Gastroenterology 1984;87: Apstein MD, Dalecki-Chipperfield K. Spinal cord injury is a risk factor for gallstone disease Gastroenterology 1987;92: Moonka R, Stiens SA, Stelzner M. Atypical gastrointestinal symptoms are not associated with gallstones in patients with spinal cord injury. Arch Phys Med Rehabil 2000;81: Ahmed HU, Smith JB, Rudderow DJ, Longo WE, Virgo KS, Johnson FE. Cholecystectomy in patients with previous spinal cord injury. Am J Surg 2002;184: Romero Ganuza FJ, La Banda G, Montalvo R, Mazaira J. Acute acalculous cholecystitis in patients with acute traumatic spinal cord injury. Spinal Cord 1997;35:
LA MIELOLESIONE ATTUALITA' IN RIABILITAZIONE IX CORSO EMRSS Siracusa 21 24 Ottobre 2013 Fondazione Sant Angela Merici
 LA MIELOLESIONE ATTUALITA' IN RIABILITAZIONE IX CORSO EMRSS Siracusa 21 24 Ottobre 2013 Fondazione Sant Angela Merici Bowel Management S. Tiberti MD Prof. C. Foti MD Physical and Rehabilitation Medicine
LA MIELOLESIONE ATTUALITA' IN RIABILITAZIONE IX CORSO EMRSS Siracusa 21 24 Ottobre 2013 Fondazione Sant Angela Merici Bowel Management S. Tiberti MD Prof. C. Foti MD Physical and Rehabilitation Medicine
The Work Up of Pelvic Floor Dyssynergia and Fecal Incontinence. Gina R. Sam, MD/MPH Director, Mount Sinai Gastrointestinal Motility Center
 The Work Up of Pelvic Floor Dyssynergia and Fecal Incontinence Gina R. Sam, MD/MPH Director, Mount Sinai Gastrointestinal Motility Center Constipation Overview Constipation Normal Transit Constipation
The Work Up of Pelvic Floor Dyssynergia and Fecal Incontinence Gina R. Sam, MD/MPH Director, Mount Sinai Gastrointestinal Motility Center Constipation Overview Constipation Normal Transit Constipation
Fecal Incontinence. What is fecal incontinence?
 Scan for mobile link. Fecal Incontinence Fecal incontinence is the inability to control the passage of waste material from the body. It may be associated with constipation or diarrhea and typically occurs
Scan for mobile link. Fecal Incontinence Fecal incontinence is the inability to control the passage of waste material from the body. It may be associated with constipation or diarrhea and typically occurs
Spinal Cord Injury Education. An Overview for Patients, Families, and Caregivers
 Spinal Cord Injury Education An Overview for Patients, Families, and Caregivers Spinal Cord Anatomy A major component of the Central Nervous System (CNS) It is 15 to 16 inches long, and weighs 1 to 2 ounces
Spinal Cord Injury Education An Overview for Patients, Families, and Caregivers Spinal Cord Anatomy A major component of the Central Nervous System (CNS) It is 15 to 16 inches long, and weighs 1 to 2 ounces
CHOC CHILDREN SUROLOGY CENTER. Constipation
 Constipation What is constipation? Constipation is a condition in which a person has uncomfortable or infrequent bowel movements. Generally, a person is considered to be constipated when bowel movements
Constipation What is constipation? Constipation is a condition in which a person has uncomfortable or infrequent bowel movements. Generally, a person is considered to be constipated when bowel movements
Urodynamics in Neuro-Urology
 Urodynamics in Neuro-Urology R Hamid MD (Res), FRCS (Urol) Consultant Urologist London Spinal Injuries Unit, Stanmore & University College London Hospitals Micturition Micturition can be visualized as
Urodynamics in Neuro-Urology R Hamid MD (Res), FRCS (Urol) Consultant Urologist London Spinal Injuries Unit, Stanmore & University College London Hospitals Micturition Micturition can be visualized as
Acute abdominal conditions Key Points
 7 Acute abdominal conditions Key Points 7.1 ASSESSMENT AND DIAGNOSIS Referred abdominal pain Fore gut pain (stomach, duodenum, gall bladder) is referred to the upper abdomen Mid gut pain (small intestine,
7 Acute abdominal conditions Key Points 7.1 ASSESSMENT AND DIAGNOSIS Referred abdominal pain Fore gut pain (stomach, duodenum, gall bladder) is referred to the upper abdomen Mid gut pain (small intestine,
Nursing. Management of Spinal Trauma. Content. Objectives. Objectives
 7 cervical vertebrae Content 12 thoracic vertebrae Nursing 5 sacral vertebrae Management of Spinal Trauma Kwai Fung Betty Siu Ward Manager O&T Dept TKOH Date : 22nd April 2007 5 lumbar vertebrae 4 coccygeal
7 cervical vertebrae Content 12 thoracic vertebrae Nursing 5 sacral vertebrae Management of Spinal Trauma Kwai Fung Betty Siu Ward Manager O&T Dept TKOH Date : 22nd April 2007 5 lumbar vertebrae 4 coccygeal
Spinal Cord and Bladder Management Male: Intermittent Catheter
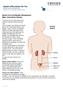 Spinal Cord and Bladder Management Male: Intermittent Catheter The 5 parts of the urinary system work together to get rid of waste and make urine. Urine is made in your kidneys and travels down 2 thin
Spinal Cord and Bladder Management Male: Intermittent Catheter The 5 parts of the urinary system work together to get rid of waste and make urine. Urine is made in your kidneys and travels down 2 thin
6/3/2011. High Prevalence and Incidence. Low back pain is 5 th most common reason for all physician office visits in the U.S.
 High Prevalence and Incidence Prevalence 85% of Americans will experience low back pain at some time in their life. Incidence 5% annual Timothy C. Shen, M.D. Physical Medicine and Rehabilitation Sub-specialty
High Prevalence and Incidence Prevalence 85% of Americans will experience low back pain at some time in their life. Incidence 5% annual Timothy C. Shen, M.D. Physical Medicine and Rehabilitation Sub-specialty
Urinary Incontinence in Women. Susan Hingle, M.D. Department of Medicine
 Urinary Incontinence in Women Susan Hingle, M.D. Department of Medicine Background Estimated 13 million Americans with urinary incontinence Women are affected twice as frequently as men Only 25% will seek
Urinary Incontinence in Women Susan Hingle, M.D. Department of Medicine Background Estimated 13 million Americans with urinary incontinence Women are affected twice as frequently as men Only 25% will seek
I can t empty my rectum without pressing my fingers in or near my vagina
 Since the birth of my baby, I can t control my bowel movements Normally bowel movements (stools) are stored in the rectum until the bowel sends a message to the brain that it is full, and the person finds
Since the birth of my baby, I can t control my bowel movements Normally bowel movements (stools) are stored in the rectum until the bowel sends a message to the brain that it is full, and the person finds
Appendix A. Rome III Diagnostic Criteria for Functional Gastrointestinal Disorders
 Appendix A Rome III Diagnostic Criteria for Functional Gastrointestinal Disorders 885 886 Appendix A: Rome III Diagnostic Criteria for FGIDs A. Functional Esophageal Disorders A1. Functional Heartburn
Appendix A Rome III Diagnostic Criteria for Functional Gastrointestinal Disorders 885 886 Appendix A: Rome III Diagnostic Criteria for FGIDs A. Functional Esophageal Disorders A1. Functional Heartburn
Alan Rosenberg, MD VP Medical Policy, Technology Assessment and Credentialing WellPoint, Inc. 233 S. Wacker Drive, Suite 3900 Chicago, IL 60606
 October 5, 2010 Alan Rosenberg, MD VP Medical Policy, Technology Assessment and Credentialing WellPoint, Inc. 233 S. Wacker Drive, Suite 3900 Chicago, IL 60606 Dear Dr. Rosenberg, The American Gastroenterological
October 5, 2010 Alan Rosenberg, MD VP Medical Policy, Technology Assessment and Credentialing WellPoint, Inc. 233 S. Wacker Drive, Suite 3900 Chicago, IL 60606 Dear Dr. Rosenberg, The American Gastroenterological
Surgical Weight Loss. Mission Bariatrics
 Surgical Weight Loss Mission Bariatrics Obesity is a major health problem in the United States, with more than one in every three people suffering from this chronic condition. Obese adults are at an increased
Surgical Weight Loss Mission Bariatrics Obesity is a major health problem in the United States, with more than one in every three people suffering from this chronic condition. Obese adults are at an increased
Basics on Bladder and Bowel
 Basics on Bladder and Bowel A quick journey through some of our recent new insights on the nervous system, the lower urinary tract and gastrointestinal tract Basics on Bladder and Bowel Basics on bladder
Basics on Bladder and Bowel A quick journey through some of our recent new insights on the nervous system, the lower urinary tract and gastrointestinal tract Basics on Bladder and Bowel Basics on bladder
Medical Nutrition Therapy for Upper Gastrointestinal Tract Disorders. By: Jalal Hejazi PhD, MSc.
 Medical Nutrition Therapy for Upper Gastrointestinal Tract Disorders By: Jalal Hejazi PhD, MSc. Digestive Disorders Common problem; more than 50 million outpatient visits per year Dietary habits and nutrition
Medical Nutrition Therapy for Upper Gastrointestinal Tract Disorders By: Jalal Hejazi PhD, MSc. Digestive Disorders Common problem; more than 50 million outpatient visits per year Dietary habits and nutrition
Understanding and managing your bowel program
 Understanding and managing your bowel program A guide for you after spinal cord injury Spinal Cord Injury Rehabilitation Program This booklet has been written by the health care providers who provide care
Understanding and managing your bowel program A guide for you after spinal cord injury Spinal Cord Injury Rehabilitation Program This booklet has been written by the health care providers who provide care
Autonomic Nervous System
 CHAPTER 4 Autonomic Nervous System Autonomic Dysreflexia What is Dysreflexia? Autonomic dysreflexia (or hyperreflexia) is a complication which occurs in people with spinal cord injuries at or above the
CHAPTER 4 Autonomic Nervous System Autonomic Dysreflexia What is Dysreflexia? Autonomic dysreflexia (or hyperreflexia) is a complication which occurs in people with spinal cord injuries at or above the
Symptoms of constipation are extremely common; the
 GASTROENTEROLOGY 2000;119:1761 1778 AMERICAN GASTROENTEROLOGICAL ASSOCIATION American Gastroenterological Association Medical Position Statement: Guidelines on Constipation This document presents the official
GASTROENTEROLOGY 2000;119:1761 1778 AMERICAN GASTROENTEROLOGICAL ASSOCIATION American Gastroenterological Association Medical Position Statement: Guidelines on Constipation This document presents the official
ICD-9-CM coding for patients with Spinal Cord Injury*
 ICD-9-CM coding for patients with Spinal Cord Injury* indicates intervening codes have been left out of this list. OTHER DISORDERS OF THE CENTRAL NERVOUS SYSTEM (340-349) 344 Other paralytic syndromes
ICD-9-CM coding for patients with Spinal Cord Injury* indicates intervening codes have been left out of this list. OTHER DISORDERS OF THE CENTRAL NERVOUS SYSTEM (340-349) 344 Other paralytic syndromes
Laparoscopic Colectomy. What do I need to know about my laparoscopic colorectal surgery?
 Laparoscopic Colectomy What do I need to know about my laparoscopic colorectal surgery? Traditionally, colon & rectal surgery requires a large, abdominal and/or pelvic incision, which often requires a
Laparoscopic Colectomy What do I need to know about my laparoscopic colorectal surgery? Traditionally, colon & rectal surgery requires a large, abdominal and/or pelvic incision, which often requires a
Topic review: Clinical presentation and diagnosis of urinary incontinence in the elderly. Prapa Pattrapornpisut 7 June 2012
 1 Topic review: Clinical presentation and diagnosis of urinary incontinence in the elderly Prapa Pattrapornpisut 7 June 2012 2 Urinary incontinence Definition the complaint of any involuntary leakage of
1 Topic review: Clinical presentation and diagnosis of urinary incontinence in the elderly Prapa Pattrapornpisut 7 June 2012 2 Urinary incontinence Definition the complaint of any involuntary leakage of
PATIENT CONSENT TO PROCEDURE - ROUX-EN-Y GASTRIC BYPASS
 As a patient you must be adequately informed about your condition and the recommended surgical procedure. Please read this document carefully and ask about anything you do not understand. Please initial
As a patient you must be adequately informed about your condition and the recommended surgical procedure. Please read this document carefully and ask about anything you do not understand. Please initial
United States Department of Transportation National Highway Traffic Safety Administration Paramedic: National Standard Curriculum 1
 UNIT TERMINAL OBJECTIVE 4-8 At the completion of this unit, the paramedic student will be able to integrate pathophysiologic principles and the assessment findings to formulate a field impression and implement
UNIT TERMINAL OBJECTIVE 4-8 At the completion of this unit, the paramedic student will be able to integrate pathophysiologic principles and the assessment findings to formulate a field impression and implement
Acute Abdominal Pain following Bariatric Surgery. Disclosure. Objectives 8/17/2015. I have nothing to disclose
 Acute Abdominal Pain following Bariatric Surgery Kathy J. Morris, DNP, APRN, FNP C, FAANP University of Nebraska Medical Center College of Nursing Disclosure I have nothing to disclose Objectives Pathophysiology
Acute Abdominal Pain following Bariatric Surgery Kathy J. Morris, DNP, APRN, FNP C, FAANP University of Nebraska Medical Center College of Nursing Disclosure I have nothing to disclose Objectives Pathophysiology
The Anatomy of Spinal Cord Injury (SCI)
 The Anatomy of Spinal Cord Injury (SCI) What is the Spinal Cord? The spinal cord is that part of your central nervous system that transmits messages between your brain and your body. The spinal cord has
The Anatomy of Spinal Cord Injury (SCI) What is the Spinal Cord? The spinal cord is that part of your central nervous system that transmits messages between your brain and your body. The spinal cord has
Care and Problems of the Digestive System. Chapter 18 Lesson 2
 Care and Problems of the Digestive System Chapter 18 Lesson 2 Care of the Digestive System Good eating habits are the best way to avoid or minimize digestive system problems. Eat a variety of foods Avoid
Care and Problems of the Digestive System Chapter 18 Lesson 2 Care of the Digestive System Good eating habits are the best way to avoid or minimize digestive system problems. Eat a variety of foods Avoid
Chapter 6 Gastrointestinal Impairment
 Chapter 6 Gastrointestinal This chapter consists of 2 parts: Part 6.1 Diseases of the digestive system Part 6.2 Abdominal wall hernias and obesity PART 6.1: DISEASES OF THE DIGESTIVE SYSTEM Diseases of
Chapter 6 Gastrointestinal This chapter consists of 2 parts: Part 6.1 Diseases of the digestive system Part 6.2 Abdominal wall hernias and obesity PART 6.1: DISEASES OF THE DIGESTIVE SYSTEM Diseases of
Are any artificial parts used in the ACE Malone surgery?
 ACE Malone (Antegrade Continence Enema) What is the ACE Malone? The Antegrade Continence Enema (ACE) is a type of surgery designed for the child who has chronic bowel problems with bouts of constipation,
ACE Malone (Antegrade Continence Enema) What is the ACE Malone? The Antegrade Continence Enema (ACE) is a type of surgery designed for the child who has chronic bowel problems with bouts of constipation,
Guide to Abdominal or Gastroenterological Surgery Claims
 What are the steps towards abdominal surgery? Investigation and Diagnosis It is very important that all necessary tests are undertaken to investigate the patient s symptoms appropriately and an accurate
What are the steps towards abdominal surgery? Investigation and Diagnosis It is very important that all necessary tests are undertaken to investigate the patient s symptoms appropriately and an accurate
Bowel and Bladder Dysfunction in MS. Tracy Walker, WOCN, MSCN, FNP C Nurse Practitioner MS Institute at Shepherd Center. Bladder Dysfunction
 Bowel and Bladder Dysfunction in MS Tracy Walker, WOCN, MSCN, FNP C Nurse Practitioner MS Institute at Shepherd Center Bladder Dysfunction Approximately 75% of people with MS experience bladder problems
Bowel and Bladder Dysfunction in MS Tracy Walker, WOCN, MSCN, FNP C Nurse Practitioner MS Institute at Shepherd Center Bladder Dysfunction Approximately 75% of people with MS experience bladder problems
Bowel Control Problems
 Bowel Control Problems WOMENCARE A Healthy Woman is a Powerful Woman (407) 898-1500 Bowel control problems affect at least 1 million people in the United States. Loss of normal control of the bowels is
Bowel Control Problems WOMENCARE A Healthy Woman is a Powerful Woman (407) 898-1500 Bowel control problems affect at least 1 million people in the United States. Loss of normal control of the bowels is
Understanding Laparoscopic Colorectal Surgery
 Understanding Laparoscopic Colorectal Surgery University Colon & Rectal Surgery A Problem with Your Colon Your doctor has told you that you have a colon problem. Now you ve learned that surgery is needed
Understanding Laparoscopic Colorectal Surgery University Colon & Rectal Surgery A Problem with Your Colon Your doctor has told you that you have a colon problem. Now you ve learned that surgery is needed
Learning Objectives. Introduction to Medical Careers. Vocabulary: Chapter 16 FACTS. Functions. Organs. Digestive System Chapter 16
 Learning Objectives Introduction to Medical Careers Digestive System Chapter 16 Define at least 10 terms relating to the digestive Describe the four functions of the digestive Identify different structures
Learning Objectives Introduction to Medical Careers Digestive System Chapter 16 Define at least 10 terms relating to the digestive Describe the four functions of the digestive Identify different structures
Normal Gastrointestinal Motility and Function
 Normal Gastrointestinal Motility and Function "Motility" is an unfamiliar word to many people; it is used primarily to describe the contraction of the muscles in the gastrointestinal tract. Because the
Normal Gastrointestinal Motility and Function "Motility" is an unfamiliar word to many people; it is used primarily to describe the contraction of the muscles in the gastrointestinal tract. Because the
THE SPINAL CORD AND THE INFLUENCE OF ITS DAMAGE ON THE HUMAN BODY
 THE SPINAL CORD AND THE INFLUENCE OF ITS DAMAGE ON THE HUMAN BODY THE SPINAL CORD. A part of the Central Nervous System The nervous system is a vast network of cells, which carry information in the form
THE SPINAL CORD AND THE INFLUENCE OF ITS DAMAGE ON THE HUMAN BODY THE SPINAL CORD. A part of the Central Nervous System The nervous system is a vast network of cells, which carry information in the form
NHS Greater Glasgow and Clyde Yorkhill Hospital CONSTIPATION IN CHILDREN
 NHS Greater Glasgow and Clyde Yorkhill Hospital CONSTIPATION IN CHILDREN Constipation in Children Version: 1 Page 1 of 8 Background Constipation is a common complaint in infants and children. The aetiology
NHS Greater Glasgow and Clyde Yorkhill Hospital CONSTIPATION IN CHILDREN Constipation in Children Version: 1 Page 1 of 8 Background Constipation is a common complaint in infants and children. The aetiology
Webinar title: Know Your Options for Treating Severe Spasticity
 Webinar title: Know Your Options for Treating Severe Spasticity Presented by: Dr. Gerald Bilsky, Physiatrist Medical Director of Outpatient Services and Associate Medical Director of Acquired Brain Injury
Webinar title: Know Your Options for Treating Severe Spasticity Presented by: Dr. Gerald Bilsky, Physiatrist Medical Director of Outpatient Services and Associate Medical Director of Acquired Brain Injury
11/10/2014. I have nothing to Disclose. Covered Stents discussed are NOT FDA approved for the indications covered in my presentation
 I have nothing to Disclose Ramsey Dallal, MD, FACS Vice Chair Department of Surgery Chief Bariatric i and Minimally i Invasive Surgery Einstein Healthcare Network Nemacolin, PA 2014 Covered Stents discussed
I have nothing to Disclose Ramsey Dallal, MD, FACS Vice Chair Department of Surgery Chief Bariatric i and Minimally i Invasive Surgery Einstein Healthcare Network Nemacolin, PA 2014 Covered Stents discussed
This document presents the official recommendations of
 GASTROENTEROLOGY 2013;144:211 217 American Gastroenterological Association Medical Position Statement on Constipation The Institute Medical Position Panel consisted of the lead technical review author
GASTROENTEROLOGY 2013;144:211 217 American Gastroenterological Association Medical Position Statement on Constipation The Institute Medical Position Panel consisted of the lead technical review author
Laparoscopic Surgery of the Colon and Rectum (Large Intestine) A Simple Guide to Help Answer Your Questions
 Laparoscopic Surgery of the Colon and Rectum (Large Intestine) A Simple Guide to Help Answer Your Questions What are the Colon and Rectum? The colon and rectum together make up the large intestine. After
Laparoscopic Surgery of the Colon and Rectum (Large Intestine) A Simple Guide to Help Answer Your Questions What are the Colon and Rectum? The colon and rectum together make up the large intestine. After
Care of Gastrostomy Tubes for Adults with IDD in Community Settings: The Nurse s Role. Lillian Khalil, BSN, RN Volunteers of America, Chesapeake
 Care of Gastrostomy Tubes for Adults with IDD in Community Settings: The Nurse s Role Lillian Khalil, BSN, RN Volunteers of America, Chesapeake Objectives The participants will be able to identify the
Care of Gastrostomy Tubes for Adults with IDD in Community Settings: The Nurse s Role Lillian Khalil, BSN, RN Volunteers of America, Chesapeake Objectives The participants will be able to identify the
D.U.C. Assist. Lec. Faculty of Dentistry General Physiology Ihsan Dhari. The Autonomic Nervous System
 The Autonomic Nervous System The portion of the nervous system that controls most visceral functions of the body is called the autonomic nervous system. This system helps to control arterial pressure,
The Autonomic Nervous System The portion of the nervous system that controls most visceral functions of the body is called the autonomic nervous system. This system helps to control arterial pressure,
Spine University s Guide to Cauda Equina Syndrome
 Spine University s Guide to Cauda Equina Syndrome 2 Introduction Your spine is a very complicated part of your body. It s made up of the bones (vertebrae) that keep it aligned, nerves that channel down
Spine University s Guide to Cauda Equina Syndrome 2 Introduction Your spine is a very complicated part of your body. It s made up of the bones (vertebrae) that keep it aligned, nerves that channel down
Normal bladder function requires a coordinated effort between the brain, spinal cord, and the bladder.
 .. Urinary Incontinence Urinary incontinence is not an inevitable part of aging, and it is not a disease. The loss of bladder control - called urinary incontinence - affects between 13 and 17 million adult
.. Urinary Incontinence Urinary incontinence is not an inevitable part of aging, and it is not a disease. The loss of bladder control - called urinary incontinence - affects between 13 and 17 million adult
Recovery After Stroke: Bladder & Bowel Function
 Recovery After Stroke: Bladder & Bowel Function Problems with bladder and bowel function are common but distressing for stroke survivors. Going to the bathroom after suffering a stroke may be complicated
Recovery After Stroke: Bladder & Bowel Function Problems with bladder and bowel function are common but distressing for stroke survivors. Going to the bathroom after suffering a stroke may be complicated
Emergencies in Post- Bariatric Surgery Patients
 Emergencies in Post- Patients Disclosures Dr. Birnbaumer has no financial disclosures Diane M. Birnbaumer, M.D., FACEP Professor of Medicine University of California, Los Angeles Senior Clinical Educator
Emergencies in Post- Patients Disclosures Dr. Birnbaumer has no financial disclosures Diane M. Birnbaumer, M.D., FACEP Professor of Medicine University of California, Los Angeles Senior Clinical Educator
Aehlert: Paramedic Practice Today PowerPoint Lecture Notes Chapter 50: Abdominal Trauma
 Aehlert: Paramedic Practice Today PowerPoint Lecture Notes Chapter 50: Abdominal Trauma Chapter 50 Abdominal Trauma 1 Describe the epidemiology, including morbidity, mortality rates, and prevention strategies,
Aehlert: Paramedic Practice Today PowerPoint Lecture Notes Chapter 50: Abdominal Trauma Chapter 50 Abdominal Trauma 1 Describe the epidemiology, including morbidity, mortality rates, and prevention strategies,
Overview of Bariatric Surgery
 Overview of Bariatric Surgery To better understand how weight loss surgery works, it is helpful to know how the normal digestive process works. As food moves along the digestive tract, special digestive
Overview of Bariatric Surgery To better understand how weight loss surgery works, it is helpful to know how the normal digestive process works. As food moves along the digestive tract, special digestive
Urinary Diversion: Ileovesicostomy/Ileal Loop/Colon Loop
 Urinary Diversion: Ileovesicostomy/Ileal Loop/Colon Loop Why do I need this surgery? A urinary diversion is a surgical procedure that is performed to allow urine to safely pass from the kidneys into a
Urinary Diversion: Ileovesicostomy/Ileal Loop/Colon Loop Why do I need this surgery? A urinary diversion is a surgical procedure that is performed to allow urine to safely pass from the kidneys into a
It s A Gut Feeling: Abdominal Pain in Children. David Deutsch, MD Pediatric Gastroenterology Rockford Health Physicians
 It s A Gut Feeling: Abdominal Pain in Children David Deutsch, MD Pediatric Gastroenterology Rockford Health Physicians Introduction Common Symptom Affects 10-15% of school-aged children Definition (Dr.
It s A Gut Feeling: Abdominal Pain in Children David Deutsch, MD Pediatric Gastroenterology Rockford Health Physicians Introduction Common Symptom Affects 10-15% of school-aged children Definition (Dr.
Gastrointestinal Bleeding
 Gastrointestinal Bleeding Introduction Gastrointestinal bleeding is a symptom of many diseases rather than a disease itself. A number of different conditions can cause gastrointestinal bleeding. Some causes
Gastrointestinal Bleeding Introduction Gastrointestinal bleeding is a symptom of many diseases rather than a disease itself. A number of different conditions can cause gastrointestinal bleeding. Some causes
Clinical Anatomy of the Biliary Apparatus: Relations & Variations
 Clinical Anatomy of the Biliary Apparatus: Relations & Variations Handout download: http://www.oucom.ohiou.edu/dbms-witmer/gs-rpac.htm 24 January 2012 Lawrence M. Witmer, PhD Professor of Anatomy Department
Clinical Anatomy of the Biliary Apparatus: Relations & Variations Handout download: http://www.oucom.ohiou.edu/dbms-witmer/gs-rpac.htm 24 January 2012 Lawrence M. Witmer, PhD Professor of Anatomy Department
FUNCTIONAL BOWEL DISORDERS
 FUNCTIONAL BOWEL DISORDERS Contributed by the International Foundation for Functional Gastrointestinal Disorders (IFFGD) and edited by the Patient Care Committee of the ACG. INTRODUCTION Doctors use the
FUNCTIONAL BOWEL DISORDERS Contributed by the International Foundation for Functional Gastrointestinal Disorders (IFFGD) and edited by the Patient Care Committee of the ACG. INTRODUCTION Doctors use the
Bladder and Bowel Problems Associated with Multiple Sclerosis
 Bladder and Bowel Problems Associated with Multiple Sclerosis Bladder Dysfunction Bladder dysfunction is one of the most common symptoms associated with Multiple Sclerosis Surveys have indicated that 60-90%
Bladder and Bowel Problems Associated with Multiple Sclerosis Bladder Dysfunction Bladder dysfunction is one of the most common symptoms associated with Multiple Sclerosis Surveys have indicated that 60-90%
Fecal incontinence (Encopresis) It is the fecal incontinence condition observed in children with chronic constipation over 1-2 years.
 WHAT IS DYSFUNCTIONAL URINATION (URINATION FUNCTION DISORDER)? It stands for the urination phase disorders, which appear due to wrongly acquired urination habits during the toilet training of some neurologically
WHAT IS DYSFUNCTIONAL URINATION (URINATION FUNCTION DISORDER)? It stands for the urination phase disorders, which appear due to wrongly acquired urination habits during the toilet training of some neurologically
Laparoscopic Cholecystectomy
 Laparoscopic Cholecystectomy Removal of Gall Bladder Page 12 Patient Information Further Information We endeavour to provide an excellent service at all times, but should you have any concerns please,
Laparoscopic Cholecystectomy Removal of Gall Bladder Page 12 Patient Information Further Information We endeavour to provide an excellent service at all times, but should you have any concerns please,
Overactive bladder is a common condition thought to. women, and is a serious condition that can lead to. significant lifestyle changes.
 Overactive bladder is a common condition thought to FADE UP TO WIDE SHOT OF FEMALE MODEL WITH TRANSPARENT SKIN. URINARY BLADDER VISIBLE IN PELVIC REGION affect over 16 percent of adults. It affects men
Overactive bladder is a common condition thought to FADE UP TO WIDE SHOT OF FEMALE MODEL WITH TRANSPARENT SKIN. URINARY BLADDER VISIBLE IN PELVIC REGION affect over 16 percent of adults. It affects men
Exercise and FES cycle fact sheet
 Exercise and FES cycle fact sheet FES = Functional Electrical Stimulation. An electrical current is passed through paralysed muscle causing it to contract. The electrical current uses the peripheral nerves
Exercise and FES cycle fact sheet FES = Functional Electrical Stimulation. An electrical current is passed through paralysed muscle causing it to contract. The electrical current uses the peripheral nerves
THE PRIMARY CARE MANAGEMENT OF CHRONIC CONSTIPATION. Dr Richard Stevens GP in Oxford and editor-in-chief of the Digest and Eurodigest
 THE PRIMARY CARE MANAGEMENT OF CHRONIC CONSTIPATION Dr Richard Stevens GP in Oxford and editor-in-chief of the Digest and Eurodigest Introduction Bowel functions are private. When bowels misfunction, privacy
THE PRIMARY CARE MANAGEMENT OF CHRONIC CONSTIPATION Dr Richard Stevens GP in Oxford and editor-in-chief of the Digest and Eurodigest Introduction Bowel functions are private. When bowels misfunction, privacy
III./8.4.2: Spinal trauma. III./8.4.2.1 Injury of the spinal cord
 III./8.4.2: Spinal trauma Introduction Causes: motor vehicle accidents, falls, sport injuries, industrial accidents The prevalence of spinal column trauma is 64/100,000, associated with neurological dysfunction
III./8.4.2: Spinal trauma Introduction Causes: motor vehicle accidents, falls, sport injuries, industrial accidents The prevalence of spinal column trauma is 64/100,000, associated with neurological dysfunction
1 ST JAMAICAN PAEDIATRIC NEPHROLOGY CONFERENCE
 in association with 1 ST JAMAICAN PAEDIATRIC NEPHROLOGY CONFERENCE Jamaica Conference Centre Kingston, Jamaica October 4 th 2014 VOIDING DISORDERS IN CHILDREN Dr. Colin Abel Paediatric Urologist Bustamante
in association with 1 ST JAMAICAN PAEDIATRIC NEPHROLOGY CONFERENCE Jamaica Conference Centre Kingston, Jamaica October 4 th 2014 VOIDING DISORDERS IN CHILDREN Dr. Colin Abel Paediatric Urologist Bustamante
Closed Automobile Insurance Third Party Liability Bodily Injury Claim Study in Ontario
 Page 1 Closed Automobile Insurance Third Party Liability Bodily Injury Claim Study in Ontario Injury Descriptions Developed from Newfoundland claim study injury definitions No injury Death Psychological
Page 1 Closed Automobile Insurance Third Party Liability Bodily Injury Claim Study in Ontario Injury Descriptions Developed from Newfoundland claim study injury definitions No injury Death Psychological
CLINICAL UPDATE. Irritable Bowel Syndrome IBS
 CLINICAL UPDATE Irritable Bowel Syndrome IBS 2nd Edition 2003, Reprinted 2006 Digestive Health Foundation 2006 Table of Contents 1 Digestive Health Foundation 2 What Is Irritable Bowel Syndrome? 2 How
CLINICAL UPDATE Irritable Bowel Syndrome IBS 2nd Edition 2003, Reprinted 2006 Digestive Health Foundation 2006 Table of Contents 1 Digestive Health Foundation 2 What Is Irritable Bowel Syndrome? 2 How
Eating, pooping, and peeing THE DIGESTIVE SYSTEM
 THE DIGESTIVE SYSTEM Ingested food is not technically in the body until it is absorbed so it needs to be: Mechanically and chemically reduced Transported by the blood to the cells Large portions are not
THE DIGESTIVE SYSTEM Ingested food is not technically in the body until it is absorbed so it needs to be: Mechanically and chemically reduced Transported by the blood to the cells Large portions are not
Spine Trauma: When to Transfer. Alexander Ching, MD Director, Orthopaedic Spine Trauma OHSU
 Spine Trauma: When to Transfer Alexander Ching, MD Director, Orthopaedic Spine Trauma OHSU Disclosures Depuy Spine Consultant (teaching and courses) Department education and research funds Atlas Spine
Spine Trauma: When to Transfer Alexander Ching, MD Director, Orthopaedic Spine Trauma OHSU Disclosures Depuy Spine Consultant (teaching and courses) Department education and research funds Atlas Spine
Traumatic injuries SPINAL CORD. Causes of Traumatic SCI SYMPTOMS. Spinal Cord trauma can be caused by:
 Traumatic injuries SPINAL CORD Jennie Trkulja RN, BScN, ENC (c) Causes of Traumatic SCI Spinal Cord trauma can be caused by: MVC (most injuries) Gunshots Falls Stabbings Assaults Industrial accidents Sports
Traumatic injuries SPINAL CORD Jennie Trkulja RN, BScN, ENC (c) Causes of Traumatic SCI Spinal Cord trauma can be caused by: MVC (most injuries) Gunshots Falls Stabbings Assaults Industrial accidents Sports
Management of Urinary Incontinence. Sheri J. Ross, BSc, DVM, PhD, Dipl. ACVIM (Internal Medicine)
 Management of Urinary Incontinence Sheri J. Ross, BSc, DVM, PhD, Dipl. ACVIM (Internal Medicine) Managing Urinary Incontinence Urinary incontinence is very prevalent among older animals. In this group
Management of Urinary Incontinence Sheri J. Ross, BSc, DVM, PhD, Dipl. ACVIM (Internal Medicine) Managing Urinary Incontinence Urinary incontinence is very prevalent among older animals. In this group
Colocutaneous Fistula. Disclosures
 Colocutaneous Fistula Madhulika G. Varma MD Associate Professor Chief, Colorectal Surgery University of California, San Francisco Honoraria Applied Medical Covidien Disclosures 1 Colocutaneous Fistula
Colocutaneous Fistula Madhulika G. Varma MD Associate Professor Chief, Colorectal Surgery University of California, San Francisco Honoraria Applied Medical Covidien Disclosures 1 Colocutaneous Fistula
Get the Facts, Be Informed, Make YOUR Best Decision. Pelvic Organ Prolapse
 Pelvic Organ Prolapse ETHICON Women s Health & Urology, a division of ETHICON, INC., a Johnson & Johnson company, is dedicated to providing innovative solutions for common women s health problems and to
Pelvic Organ Prolapse ETHICON Women s Health & Urology, a division of ETHICON, INC., a Johnson & Johnson company, is dedicated to providing innovative solutions for common women s health problems and to
Cervical Whiplash: Considerations in the Rehabilitation of Cervical Myofascial Injury. Canadian Family Physician
 Cervical Whiplash: Considerations in the Rehabilitation of Cervical Myofascial Injury 1 Canadian Family Physician Volume 32, September 1986 Arthur Ameis, MD Dr. Ames practices physical medicine and rehabilitation,
Cervical Whiplash: Considerations in the Rehabilitation of Cervical Myofascial Injury 1 Canadian Family Physician Volume 32, September 1986 Arthur Ameis, MD Dr. Ames practices physical medicine and rehabilitation,
Urinary Incontinence (Involuntary Loss of Urine) A Patient Guide
 Urinary Incontinence (Involuntary Loss of Urine) A Patient Guide Urinary Incontinence (Urine Loss) This booklet is intended to give you some facts on urinary incontinence - what it is, and is not, and
Urinary Incontinence (Involuntary Loss of Urine) A Patient Guide Urinary Incontinence (Urine Loss) This booklet is intended to give you some facts on urinary incontinence - what it is, and is not, and
03/20/12. Recognize the right of patients to appropriate assessment and management of pain
 Narcotic Bowel Syndrome Alvin Zfass M.D. M.D. Professor of Medicine Toufic Kachaamy M.D. GI Fellow Chronic Pain 110 million Americans suffer from chronic pain according to the NIH Cost of untreated t or
Narcotic Bowel Syndrome Alvin Zfass M.D. M.D. Professor of Medicine Toufic Kachaamy M.D. GI Fellow Chronic Pain 110 million Americans suffer from chronic pain according to the NIH Cost of untreated t or
Chapter 15. Autonomic Nervous System (ANS) and Visceral Reflexes. general properties Anatomy. Autonomic effects on target organs
 Chapter 15 Autonomic Nervous System (ANS) and Visceral Reflexes general properties Anatomy Autonomic effects on target organs Central control of autonomic function 15-1 Copyright (c) The McGraw-Hill Companies,
Chapter 15 Autonomic Nervous System (ANS) and Visceral Reflexes general properties Anatomy Autonomic effects on target organs Central control of autonomic function 15-1 Copyright (c) The McGraw-Hill Companies,
Facing Pancreatic Surgery? Learn about minimally invasive da Vinci Surgery
 Facing Pancreatic Surgery? Learn about minimally invasive da Vinci Surgery The Condition: Pancreatitis/Pancreatic Cancer The pancreas is an organ that produces enzymes and hormones to help your body digest
Facing Pancreatic Surgery? Learn about minimally invasive da Vinci Surgery The Condition: Pancreatitis/Pancreatic Cancer The pancreas is an organ that produces enzymes and hormones to help your body digest
Informed Consent for Laparoscopic Vertical Sleeve Gastrectomy. Patient Name
 Informed Consent for Laparoscopic Vertical Sleeve Gastrectomy Patient Name Please read this form carefully and ask about anything you may not understand. I consent to have a laparoscopic Vertical Sleeve
Informed Consent for Laparoscopic Vertical Sleeve Gastrectomy Patient Name Please read this form carefully and ask about anything you may not understand. I consent to have a laparoscopic Vertical Sleeve
The University of Hong Kong Department of Surgery Division of Esophageal and Upper Gastrointestinal Surgery
 Program Overview The University of Hong Kong Department of Surgery Division of Esophageal and Upper Gastrointestinal Surgery Weight Control and Metabolic Surgery Program The Weight Control and Metabolic
Program Overview The University of Hong Kong Department of Surgery Division of Esophageal and Upper Gastrointestinal Surgery Weight Control and Metabolic Surgery Program The Weight Control and Metabolic
Abdominal Pain. Charles Henley, DO, MPH Department of Family Medicine. OSU College of Osteopathic Medicine (Revised 9/2002)
 Abdominal Pain Charles Henley, DO, MPH Department of Family Medicine OSU College of Osteopathic Medicine (Revised 9/2002) Common Causes of Abdominal Pain Infants - colic, gastroenteritis, constipation,
Abdominal Pain Charles Henley, DO, MPH Department of Family Medicine OSU College of Osteopathic Medicine (Revised 9/2002) Common Causes of Abdominal Pain Infants - colic, gastroenteritis, constipation,
Case Based Urology Learning Program
 Case Based Urology Learning Program Resident s Corner: UROLOGY Case Number 21 CBULP 2011 068 Case Based Urology Learning Program Editor: Associate Editors: Manager: Case Contributors: Steven C. Campbell,
Case Based Urology Learning Program Resident s Corner: UROLOGY Case Number 21 CBULP 2011 068 Case Based Urology Learning Program Editor: Associate Editors: Manager: Case Contributors: Steven C. Campbell,
The Acute Abdomen. Dr. Ed Snyder Dr. Melanie Walker Huntington Memorial Hospital
 The Acute Abdomen Dr. Ed Snyder Dr. Melanie Walker Huntington Memorial Hospital Causes of the Acute Abdomen Hemorrhage in the GI tract Blood vessel GU tract Perforation of the GI tract Ulcer Infection
The Acute Abdomen Dr. Ed Snyder Dr. Melanie Walker Huntington Memorial Hospital Causes of the Acute Abdomen Hemorrhage in the GI tract Blood vessel GU tract Perforation of the GI tract Ulcer Infection
Section: Surgery Last Reviewed Date: December 2013. Policy No: 129 Effective Date: March 1, 2014
 Medical Policy Manual Topic: Transanal Radiofrequency Treatment of Fecal Incontinence Date of Origin: December 2003 Section: Surgery Last Reviewed Date: December 2013 Policy No: 129 Effective Date: March
Medical Policy Manual Topic: Transanal Radiofrequency Treatment of Fecal Incontinence Date of Origin: December 2003 Section: Surgery Last Reviewed Date: December 2013 Policy No: 129 Effective Date: March
Orthopaedic Spine Center. Anterior Cervical Discectomy and Fusion (ACDF) Normal Discs
 Orthopaedic Spine Center Graham Calvert MD James Woodall MD PhD Anterior Cervical Discectomy and Fusion (ACDF) Normal Discs The cervical spine consists of the bony vertebrae, discs, nerves and other structures.
Orthopaedic Spine Center Graham Calvert MD James Woodall MD PhD Anterior Cervical Discectomy and Fusion (ACDF) Normal Discs The cervical spine consists of the bony vertebrae, discs, nerves and other structures.
symptoms of Incontinence
 Types, causes and symptoms of Urinary Incontinence Aims and Objectives Aim: To have an understanding of the types and causes of urinary incontinence. Objectives: To be aware of the incidence and prevalence
Types, causes and symptoms of Urinary Incontinence Aims and Objectives Aim: To have an understanding of the types and causes of urinary incontinence. Objectives: To be aware of the incidence and prevalence
Multiple Sclerosis (MS)
 Multiple Sclerosis (MS) Purpose/Goal: Care partners will have an understanding of Multiple Sclerosis and will demonstrate safety and promote independence while providing care to the client with MS. Introduction
Multiple Sclerosis (MS) Purpose/Goal: Care partners will have an understanding of Multiple Sclerosis and will demonstrate safety and promote independence while providing care to the client with MS. Introduction
URINARY CATHETER CARE
 URINARY CATHETER CARE INTRODUCTION Urinary catheter care is a very important skill, and it is a skill that many certified nursing assistants (CNAs) must know. Competence at providing urinary catheter care
URINARY CATHETER CARE INTRODUCTION Urinary catheter care is a very important skill, and it is a skill that many certified nursing assistants (CNAs) must know. Competence at providing urinary catheter care
Normal bladder function. Aging of the bladder and urethra
 SECTION 10 Bladder and bowel control (continence) Section overview This section looks at: Normal bladder function Stroke and loss of bladder control (incontinence) Normal bowel function Stroke and bowel
SECTION 10 Bladder and bowel control (continence) Section overview This section looks at: Normal bladder function Stroke and loss of bladder control (incontinence) Normal bowel function Stroke and bowel
Introduction. Physiology of the Abdomen. Anatomy & Physiology. Abdominal Pain Introduction (2 of 2) Gastrointestional and Urologic Emergencies
 Gastrointestional and Urologic Emergencies Introduction Abdominal pain is a common complaint. Cause of abdominal pain is often difficult to determine. As an EMT: You do not need to determine exact cause.
Gastrointestional and Urologic Emergencies Introduction Abdominal pain is a common complaint. Cause of abdominal pain is often difficult to determine. As an EMT: You do not need to determine exact cause.
The Digestive System
 School of Social Work The Digestive System Undergraduate researcher: Nancy D. Bergerson (2010). 1 The Digestive System breaks down and absorbs food. When food is eaten, it is not in a form the body can
School of Social Work The Digestive System Undergraduate researcher: Nancy D. Bergerson (2010). 1 The Digestive System breaks down and absorbs food. When food is eaten, it is not in a form the body can
Gastrointestinal problems in children with Down's syndrome
 Gastrointestinal problems in children with Down's syndrome by Dr Liz Marder This article was written for parents for the Down s Syndrome Association newsletter and is reproduced here with the permission
Gastrointestinal problems in children with Down's syndrome by Dr Liz Marder This article was written for parents for the Down s Syndrome Association newsletter and is reproduced here with the permission
Recovery After Stroke: Bladder & Bowel Function. Treatments
 Recovery After Stroke: Bladder & Bowel Function Problems with bladder and bowel function are common but distressing for stroke survivors. Going to the bathroom after suffering a stroke may be complicated
Recovery After Stroke: Bladder & Bowel Function Problems with bladder and bowel function are common but distressing for stroke survivors. Going to the bathroom after suffering a stroke may be complicated
Digestive System Functions
 Digestive System Functions A. Gastrointestinal Processes 1. Ingestion: placing food in mouth (voluntary) 2. Propulsion: moving food through GI tract a. Peristalsis: alternating waves of contraction and
Digestive System Functions A. Gastrointestinal Processes 1. Ingestion: placing food in mouth (voluntary) 2. Propulsion: moving food through GI tract a. Peristalsis: alternating waves of contraction and
Registered Charity No. 5365
 THE MULTIPLE SCLEROSIS SOCIETY OF IRELAND Dartmouth House, Grand Parade, Dublin 6. Telephone: (01) 269 4599. Fax: (01) 269 3746 MS Helpline: 1850 233 233 E-mail: mscontact@ms-society.ie www.ms-society.ie
THE MULTIPLE SCLEROSIS SOCIETY OF IRELAND Dartmouth House, Grand Parade, Dublin 6. Telephone: (01) 269 4599. Fax: (01) 269 3746 MS Helpline: 1850 233 233 E-mail: mscontact@ms-society.ie www.ms-society.ie
Incontinence. What is incontinence?
 Incontinence What is incontinence? Broadly speaking, the medical term incontinence refers to any involuntary release of bodily fluids, but many people associate it strongly with the inability to control
Incontinence What is incontinence? Broadly speaking, the medical term incontinence refers to any involuntary release of bodily fluids, but many people associate it strongly with the inability to control
Overactive Bladder (OAB)
 Overactive Bladder (OAB) Overactive bladder is a problem with bladder storage function that causes a sudden urge to urinate. The urge may be difficult to suppress, and overactive bladder can lead to the
Overactive Bladder (OAB) Overactive bladder is a problem with bladder storage function that causes a sudden urge to urinate. The urge may be difficult to suppress, and overactive bladder can lead to the
Medical Physiology Z.H.Al-Zubaydi
 Lec.13 Medical Physiology Z.H.Al-Zubaydi Functions of the Digestive System The major functions of the digestive tract include the following six processes, summarized in Figure 1: 1. Ingestion Food must
Lec.13 Medical Physiology Z.H.Al-Zubaydi Functions of the Digestive System The major functions of the digestive tract include the following six processes, summarized in Figure 1: 1. Ingestion Food must
2.01.58. Description. Section: Medicine Effective Date: January 15, 2015 Subsection: Medicine Original Policy Date: June 7, 2012 Subject: Page: 1 of 6
 Page: 1 of 6 Last Review Status/Date: December 2014 Description Radiofrequency (RF) energy has been investigated as a minimally invasive treatment of fecal incontinence, referred to as the Secca procedure.
Page: 1 of 6 Last Review Status/Date: December 2014 Description Radiofrequency (RF) energy has been investigated as a minimally invasive treatment of fecal incontinence, referred to as the Secca procedure.
Constipation in the older child
 Constipation in the older child Definition Constipation in children is a common problem. Constipation in children is often characterized by infrequent bowl movements or hard, dry stools. Various factors
Constipation in the older child Definition Constipation in children is a common problem. Constipation in children is often characterized by infrequent bowl movements or hard, dry stools. Various factors

 More details >>> HERE
More details >>> HERE