Summary of the risk management plan (RMP) for Zydelig (idelalisib)
|
|
|
- Anabel Foster
- 7 years ago
- Views:
Transcription
1 EMA/476566/2014 Summary of the risk management plan (RMP) for Zydelig (idelalisib) This is a summary of the risk management plan (RMP) for Zydelig, which details the measures to be taken in order to ensure that Zydelig is used as safely as possible. For more information on RMP summaries, see here. This RMP summary should be read in conjunction with the EPAR summary and the product information for Zydelig, which can be found on Zydelig s EPAR page. Overview of disease epidemiology Zydelig is a cancer medicine that is used to treat two types of blood cancer: chronic lymphocytic leukaemia and follicular lymphoma. Chronic lymphocytic leukaemia (a cancer of a type of white blood cells called B lymphocytes) is a rare disease that affects around 4 in 10,000 people per year in the Western world. It mainly affects elderly people, typically around 70 years of age. The course of the disease is highly variable, and low-risk patients may expect to live for more than 10 years compared with 2 years in high-risk patients. Follicular lymphoma is an indolent (or slow-growing) form of another cancer of the B lymphocytes known as non-hodgkin lymphoma. Most cases of indolent non-hodgkin lymphoma affect people older than 60 years of age. It is also a rare disease and affects up to 4 people in 10,000. With treatment, patients can expect to live from 5 years to up to 10 years or more, depending on the extent (stage) and location of their cancer. Summary of treatment benefits In a main study of 220 patients with previously treated chronic lymphocytic leukaemia, Zydelig was shown to be more effective at treating the cancer than placebo (a dummy treatment) when both were given in combination with another medicine rituximab: 75% of patients taking Zydelig had an improvement in their disease compared with 15% of patients taking placebo. Zydelig was also more effective than placebo in the subgroup of patients who had a specific genetic mutation in their cancer cells that makes them unsuitable for chemo-immunotherapy. Another main study evaluated Zydelig in patients with different lymphomas, including 72 patients with follicular lymphoma that had failed two previous treatments. Zydelig was shown to be effective, with 54% of patients with follicular lymphoma having either a complete or partial response to treatment. Unknowns relating to treatment benefits Zydelig has not been studied in children. Zydelig has not been studied in patients with severe liver or kidney impairment. There are only limited data on the long-term use of Zydelig. Page 1/8
2 Summary of safety concerns Important identified risks Risk What is known Preventability Increased liver enzyme (transaminase) levels in the blood Watery diarrhoea (diarrhoea/colitis) Lung inflammation (pneumonitis) Decreased levels of white blood cells (neutropenia) Increases in liver enzyme levels that occur early in the course of therapy have been observed in clinical studies. In many cases, the liver enzyme levels return to normal without any treatment. In some cases the liver enzyme levels become too high requiring treatment with Zydelig to be stopped. Increased liver enzyme levels is listed as a very common side effect (occurring in more than 1 patient in 10) in the product information for Zydelig. Severe diarrhoea can occur after many months of therapy. More than 1 patient in 10 have experienced inflammation of the bowel and watery diarrhoea occurring several times a day and lasting for weeks. Watery diarrhoea is listed as a very common side effect in the product information for Zydelig Lung inflammation has been seen in some patients receiving Zydelig but it is not known whether it is caused by Zydelig or by other concomitant cancer treatments. This lung inflammation is sometimes severe enough to require mechanical ventilation and has, very rarely, led to death. Decreased levels of white blood cells have been seen in more than 1 patient in 10 treated with It is not known how an increase in liver enzyme levels can be prevented, but when it occurs stopping Zydelig can lead to the enzyme levels returning to normal. Regular blood tests should be carried out during the first few weeks and months of therapy to detect an increase in liver enzyme levels. Treatment may be restarted at a lower dose once the levels of enzymes have improved. It is not known how diarrhoea can be prevented, but when it occurs it can be managed by temporarily stopping Zydelig and by treatment with antidiarrhoeals, and/or anti-inflammatory medicines that are effective in the gastrointestinal tract. Treatment may be restarted at a lower dose once the diarrhoea has improved. It is not known how lung inflammation can be prevented, but when it occurs it can be treated. Patients who experience shortness of breath and cough without any signs of infection, should stop treatment and be treated with corticosteroids. Treatment can be restarted once the lung inflammation has resolved. If the lung symptoms come back, treatment with Zydelig needs to be permanently stopped. It is not known how a decrease in white blood cell levels can be prevented, but when it occurs stopping Zydelig can lead Page 2/8
3 Risk What is known Preventability Rash Zydelig in clinical studies. Decreased levels of white blood cells is listed as a very common side effect in the product information for Zydelig Patients have experienced rash after starting treatment with Zydelig. The rash is usually a flat, red area of the skin (on the trunk and extremities) associated with occasional fever or itching. The rash was generally mild to moderate and rarely resulted in discontinuation of treatment. It typically resolved with treatment and dose interruption for severe cases. to the white blood cells levels returning to normal. Treatment may be restarted once the levels of white blood cells have returned to normal. It is not known how rash can be prevented, but when it occurs it can be treated with antihistamine and/or topical (skin) or oral steroids. In the event of severe rash, treatment must be stopped. Important potential risks Risk Birth defects (including teratogenicity) What is known In animal studies with Zydelig harmful effects were seen on the foetus and on fertility. No human data are available. Zydelig is not recommended for use during pregnancy, and women taking the medicine should use a reliable method of contraception to avoid becoming pregnant during treatment and for 1 month after treatment. As it is not known whether Zydelig can make hormonal contraceptives less effective, women should also use a barrier method of contraception such as condoms. Use of Zydelig with certain medicines called CYP3A inducers Use of Zydelig with medicines normally processed by CYP3A (called CYP3A substrates) Zydelig is processed in the body by CYP3A enzymes. Certain medicines can increase the activity of these enzymes. They are called CYP3A inducers and include rifampicin (used to prevent and treat tuberculosis), phenytoin and carbamazepine (used to prevent seizures) and St. John s wort (a herbal remedy used for depression and anxiety). By increasing CYP3A activity, CYP3A inducers can reduce the levels of Zydelig in the blood thereby reducing its effectiveness. As Zydelig itself blocks CYP3A enzymes, it could potentially increase the effects of other medicines metabolised by the enzymes or make their side effects worse. Zydelig should be used with caution with these medicines. In particular, Zydelig should not be used concomitantly with any of the following medicinal products due to the potential for serious or life-threatening side effects: alfuzosin, amiodarone, cisapride, pimozide, quinidine, ergotamine, Page 3/8
4 Risk What is known dihydroergotamine, quetiapine, lovastatin, simvastatin, sildenafil, midazolam and triazolam. Sunburn-like reactions following exposure to light (photosensitivity) Studies in experimental models indicate that there could be a risk of photosensitivity with Zydelig. Photosensitivity effects of Zydelig in humans have not been established. Skin cancer Chronic lymphocytic leukaemia is associated with an increased incidence of skin cancer, which is thought to be linked to cancer s suppressing of the immune system as well as the treatment. Because Zydelig acts on the immune system, skin cancer is a potential risk. The potential for developing skin cancer with Zydelig has not yet been established. Missing information Risk Drug resistance (reduction in the medicine s effectiveness) Tumour inducing potential (carcinogenicity) Long-term safety Use in patients with severe liver impairment Use in patients with severe kidney impairment Use in patients with chronic active hepatitis Use in patients undergoing immunisation (vaccination) during treatment What is known The potential for development of drug resistance with Zydelig has not yet been established. The tumour inducing potential (carcinogenicity) of Zydelig has not yet been established. There is limited experience with prolonged use of Zydelig (over 1 year) in clinical trials. The long-term safety of Zydelig has not yet been established. The safety of Zydelig is not known in patients with severely decreased liver function. There is limited information on the use of the medicine in patients with severe kidney impairment. Increases in the levels in the body of Zydelig s active substance idelalisib and its major metabolite (the substance produced when idelalisib is broken down) were minimal and not clinically meaningful in patients with severely decreased kidney function. The safety of Zydelig is not known in patients with chronic (long-term) active hepatitis including viral hepatitis. The safety of Zydelig is not known in patients with concomitant immunisation. Page 4/8
5 Risk Immunological effects and auto-immunity Use in children Use while breastfeeding Interaction with oral contraceptive What is known The effects of idelalisib on the immune system, including how it may trigger immune reactions against the body s own cells (auto-immunity), are not known. The safety and efficacy of Zydelig have not yet been established in children. It is not known whether the ingredients in Zydelig pass into human breast milk. Breastfeeding should be discontinued during treatment with Zydelig.. It is not known whether Zydelig interacts with oral contraceptives. Women using hormonal contraceptives should also use a barrier method to avoid becoming pregnant while taking Zydelig and for a month after stopping treatment. Summary of risk minimisation measures by safety concern All medicines have a summary of product characteristics (SmPC) which provides physicians, pharmacists and other healthcare professionals with details on how to use the medicine, and also describes the risks and recommendations for minimising them. Information for patients is available in lay language in the package leaflet. The measures listed in these documents are known as routine risk minimisation measures. The SmPC and the package leaflet are part of the medicine s product information. The product information for Zydelig can be found on Zydelig s EPAR page. This medicine has no additional risk minimisation measures. Planned post-authorisation development plan List of studies in post-authorisation development plan Study/activity (including study number) Objectives Safety concerns/ efficacy issue addressed Status Planned date for submission of (interim and) final results GS-US To evaluate the safety and efficacy of idelalisib in combination with rituximab in patients with relapsed chronic lymphocytic leukaemia. Long-term safety and efficacy Ongoing December 2014 GS-US To evaluate the safety and efficacy of idelalisib in combination with rituximab in patients with relapsed chronic lymphocytic leukaemia. Long-term safety and efficacy Ongoing Fourth quarter To evaluate the safety and Long-term safety Ongoing December 2015 Page 5/8
6 Study/activity (including study number) Objectives Safety concerns/ efficacy issue addressed Status Planned date for submission of (interim and) final results efficacy of idelalisib monotherapy in patients with refractory indolent and efficacy non-hodgkin lymphoma. BP-US (Interventional clinical study) BP-US (Interventional clinical study) To evaluate what the body does to Zydelig and the extent to which Zydelig becomes available to the body and how children tolerate Zydelig. To evaluate safety and efficacy of Zydelig when used with standard of care multi-agent chemotherapy in children. Safety in children Planned Not determined Safety in children Planned Not determined Study BP-US An in vivo interaction (induction) study with oral To evaluate the effect of idelalisib co-administration on the pharmacokinetics of a representative oral contraceptive. Oral contraceptive drug-drug interaction study Planned February 2015 (Feasibility report and proposal for study design) contraceptive (PK study) AD Further studies on To determine the role of aldehyde oxidase in the Drug-drug interactions with Planned December 2014 the human enzymology of metabolism of idelalisib. aldehyde oxidase idelalisib oxidation AD In vitro assessment of GS-1101 as a substrate for human To determine the OATP1B1 and OATP1B3 substrate characteristics of idelalisib. Drug-drug interactions with transport substrates Planned December 2014 OATP1B1 and OATP1B3 over an extended concentration range TX To evaluate carcinogenicity Carcinogenicity Ongoing Second quarter Page 6/8
7 Study/activity (including study number) A 2-year oral (gavage) carcinogenicity study of idelalisib in sprague dawley rats Objectives Safety concerns/ efficacy issue addressed Status with idelalisib therapy Planned date for submission of (interim and) final results TX week oral gavage carcinogenicity and toxicokinetic study with idelalisib in RasH2 [ T (hemizygous), CByB6F1- Tg(HRAS)2Jic] mice To evaluate carcinogenicity with idelalisib therapy. Carcinogenicity Planned Second quarter 2017 TX week oral dose range-finding toxicity and toxicokinetic oral gavage study with idelalisib in W (wild type) RasH2 mice To determine the safety of the main human metabolite GS Safety of the main human metabolite GS Ongoing Fourth quarter 2014 PC Radioligand binding assay with GS To determine the safety of the main human metabolite GS Safety of the main human metabolite GS Ongoing Fourth quarter 2014 Multiple studies Mechanistic studies on the effect of idelalisib on immune function To evaluate data on immunological effects and auto-immunity. Immunological effects and autoimmunity Planned March 2015 Page 7/8
8 Study/activity (including study number) Objectives Safety concerns/ efficacy issue addressed Status Planned date for submission of (interim and) final results Drug mechanism of resistance studies for CLL (samples collected from completed and ongoing studies: GS-US , GS-US and GS-US ) and inhl To investigate the mechanism of drug resistance with idelalisib. Development of drug resistance Started December 2014 (CLL) To be determined (inhl) Studies which are a condition of the marketing authorisation As part of the marketing authorisation for Zydelig, the company is obliged to submit the final reports of two ongoing studies in patients with chronic lymphocytic leukaemia (GS-US and GS-US ) and of another study, , in patients with indolent B-cell non-hodgkin s lymphoma. Summary of changes to the risk management plan over time Not applicable. This summary was last updated in Page 8/8
Summary of the risk management plan (RMP) for Otezla (apremilast)
 EMA/741412/2014 Summary of the risk management plan (RMP) for Otezla (apremilast) This is a summary of the risk management plan (RMP) for Otezla, which details the measures to be taken in order to ensure
EMA/741412/2014 Summary of the risk management plan (RMP) for Otezla (apremilast) This is a summary of the risk management plan (RMP) for Otezla, which details the measures to be taken in order to ensure
Summary of the risk management plan (RMP) for Ofev (nintedanib)
 EMA/738120/2014 Summary of the risk management plan (RMP) for Ofev (nintedanib) This is a summary of the risk management plan (RMP) for Ofev, which details the measures to be taken in order to ensure that
EMA/738120/2014 Summary of the risk management plan (RMP) for Ofev (nintedanib) This is a summary of the risk management plan (RMP) for Ofev, which details the measures to be taken in order to ensure that
Summary of the risk management plan (RMP) for Orkambi (lumacaftor and ivacaftor)
 EMA/662624/2015 Summary of the risk management plan (RMP) for Orkambi (lumacaftor and ivacaftor) This is a summary of the risk management plan (RMP) for Orkambi, which details the measures to be taken
EMA/662624/2015 Summary of the risk management plan (RMP) for Orkambi (lumacaftor and ivacaftor) This is a summary of the risk management plan (RMP) for Orkambi, which details the measures to be taken
Summary of the risk management plan (RMP) for Rasagiline ratiopharm (rasagiline)
 EMA/744222/2014 Summary of the risk management plan (RMP) for Rasagiline ratiopharm (rasagiline) This is a summary of the risk management plan (RMP) for Rasagiline ratiopharm, which details the measures
EMA/744222/2014 Summary of the risk management plan (RMP) for Rasagiline ratiopharm (rasagiline) This is a summary of the risk management plan (RMP) for Rasagiline ratiopharm, which details the measures
Summary of the risk management plan (RMP) for Ionsys (fentanyl)
 EMA/764409/2015 Summary of the risk management plan (RMP) for Ionsys (fentanyl) This is a summary of the risk management plan (RMP) for Ionsys, which details the measures to be taken in order to ensure
EMA/764409/2015 Summary of the risk management plan (RMP) for Ionsys (fentanyl) This is a summary of the risk management plan (RMP) for Ionsys, which details the measures to be taken in order to ensure
Summary of the risk management plan (RMP) for Cerdelga (eliglustat)
 EMA/743948/2014 Summary of the risk management plan (RMP) for Cerdelga (eliglustat) This is a summary of the risk management plan (RMP) for Cerdelga, which details the measures to be taken in order to
EMA/743948/2014 Summary of the risk management plan (RMP) for Cerdelga (eliglustat) This is a summary of the risk management plan (RMP) for Cerdelga, which details the measures to be taken in order to
Summary of the risk management plan (RMP) for Sirturo (bedaquiline)
 EMA/16634/2014 Summary of the risk management plan (RMP) for Sirturo (bedaquiline) This is a summary of the risk management plan (RMP) for Sirturo, which details the measures to be taken in order to ensure
EMA/16634/2014 Summary of the risk management plan (RMP) for Sirturo (bedaquiline) This is a summary of the risk management plan (RMP) for Sirturo, which details the measures to be taken in order to ensure
Summary of the risk management plan (RMP) for Xultophy (insulin degludec / liraglutide)
 EMA/467911/2014 Summary of the risk management plan (RMP) for Xultophy (insulin degludec / liraglutide) This is a summary of the risk management plan (RMP) for Xultophy, which details the measures to be
EMA/467911/2014 Summary of the risk management plan (RMP) for Xultophy (insulin degludec / liraglutide) This is a summary of the risk management plan (RMP) for Xultophy, which details the measures to be
In non-hodgkin s lymphoma, MabThera is used to treat two types of the disease, both of which affect B-lymphocytes:
 EMA/614203/2010 EMEA/H/C/000165 EPAR summary for the public rituximab This is a summary of the European public assessment report (EPAR) for. It explains how the Committee for Medicinal Products for Human
EMA/614203/2010 EMEA/H/C/000165 EPAR summary for the public rituximab This is a summary of the European public assessment report (EPAR) for. It explains how the Committee for Medicinal Products for Human
Severe rheumatoid arthritis (a disease that causes inflammation of the joints),where MabThera is given intravenously together with methotrexate.
 EMA/614203/2010 EMEA/H/C/000165 EPAR summary for the public rituximab This is a summary of the European public assessment report (EPAR) for. It explains how the Committee for Medicinal Products for Human
EMA/614203/2010 EMEA/H/C/000165 EPAR summary for the public rituximab This is a summary of the European public assessment report (EPAR) for. It explains how the Committee for Medicinal Products for Human
INITIATING ORAL AUBAGIO (teriflunomide) THERAPY
 FOR YOUR PATIENTS WITH RELAPSING FORMS OF MS INITIATING ORAL AUBAGIO (teriflunomide) THERAPY WARNING: HEPATOTOXICITY AND RISK OF TERATOGENICITY Severe liver injury including fatal liver failure has been
FOR YOUR PATIENTS WITH RELAPSING FORMS OF MS INITIATING ORAL AUBAGIO (teriflunomide) THERAPY WARNING: HEPATOTOXICITY AND RISK OF TERATOGENICITY Severe liver injury including fatal liver failure has been
Summary of the risk management plan (RMP) for Accofil (filgrastim)
 EMA/475472/2014 Summary of the risk management plan (RMP) for Accofil (filgrastim) This is a summary of the risk management plan (RMP) for Accofil, which details the measures to be taken in order to ensure
EMA/475472/2014 Summary of the risk management plan (RMP) for Accofil (filgrastim) This is a summary of the risk management plan (RMP) for Accofil, which details the measures to be taken in order to ensure
Lung Pathway Group Nintedanib (Vargatef) in advanced Non-Small Cell Lung Cancer (NSCLC)
 Lung Pathway Group Nintedanib (Vargatef) in advanced Non-Small Cell Lung Cancer (NSCLC) Indication: In combination with docetaxel in locally advanced, metastatic or locally recurrent NSCLC of adenocarcinoma
Lung Pathway Group Nintedanib (Vargatef) in advanced Non-Small Cell Lung Cancer (NSCLC) Indication: In combination with docetaxel in locally advanced, metastatic or locally recurrent NSCLC of adenocarcinoma
I B2.4. Design of the patient information leaflet for VariQuin
 (English translation of official Dutch version) I B2.4. Design of the patient information leaflet for VariQuin Information for the Patient: Read this package leaflet carefully when you have some time to
(English translation of official Dutch version) I B2.4. Design of the patient information leaflet for VariQuin Information for the Patient: Read this package leaflet carefully when you have some time to
CICLOSPORIN. What are the aims of this leaflet?
 CICLOSPORIN What are the aims of this leaflet? This leaflet has been written to help you understand more about ciclosporin. It tells you what it is, how it works, how it is used to treat skin conditions,
CICLOSPORIN What are the aims of this leaflet? This leaflet has been written to help you understand more about ciclosporin. It tells you what it is, how it works, how it is used to treat skin conditions,
Elements for a Public Summary
 VI.2 Elements for a Public Summary VI.2.1 Overview of Disease Epidemiology Hunter syndrome is a rare genetic disease which mainly affects males of all ethnicities. The incidence rate ranges from 0.6 to
VI.2 Elements for a Public Summary VI.2.1 Overview of Disease Epidemiology Hunter syndrome is a rare genetic disease which mainly affects males of all ethnicities. The incidence rate ranges from 0.6 to
Teriflunomide (Aubagio) 14mg once daily tablet
 Teriflunomide (Aubagio) 14mg once daily tablet Exceptional healthcare, personally delivered Your Consultant Neurologist has suggested that you may benefit from treatment with Teriflunomide. The decision
Teriflunomide (Aubagio) 14mg once daily tablet Exceptional healthcare, personally delivered Your Consultant Neurologist has suggested that you may benefit from treatment with Teriflunomide. The decision
Summary of the risk management plan (RMP) for Aripiprazole Pharmathen (aripiprazole)
 EMA/303592/2015 Summary of the risk management plan (RMP) for Aripiprazole Pharmathen (aripiprazole) This is a summary of the risk management plan (RMP) for Aripiprazole Pharmathen, which details the measures
EMA/303592/2015 Summary of the risk management plan (RMP) for Aripiprazole Pharmathen (aripiprazole) This is a summary of the risk management plan (RMP) for Aripiprazole Pharmathen, which details the measures
ELEMENTS FOR A PUBLIC SUMMARY. Overview of disease epidemiology. Summary of treatment benefits
 VI: 2 ELEMENTS FOR A PUBLIC SUMMARY Bicalutamide (CASODEX 1 ) is a hormonal therapy anticancer agent, used for the treatment of prostate cancer. Hormones are chemical messengers that help to control the
VI: 2 ELEMENTS FOR A PUBLIC SUMMARY Bicalutamide (CASODEX 1 ) is a hormonal therapy anticancer agent, used for the treatment of prostate cancer. Hormones are chemical messengers that help to control the
Understanding How Existing and Emerging MS Therapies Work
 Understanding How Existing and Emerging MS Therapies Work This is a promising and hopeful time in the field of multiple sclerosis (MS). Many new and different therapies are nearing the final stages of
Understanding How Existing and Emerging MS Therapies Work This is a promising and hopeful time in the field of multiple sclerosis (MS). Many new and different therapies are nearing the final stages of
Risk Management Plan
 Risk Management Plan Active substance: Drospirenone/ethinylestradiol Version number: 4.0 VI.2 Elements for a Public Summary VI.2.1 Overview of disease epidemiology Oral contraception Effective control
Risk Management Plan Active substance: Drospirenone/ethinylestradiol Version number: 4.0 VI.2 Elements for a Public Summary VI.2.1 Overview of disease epidemiology Oral contraception Effective control
See 17 for PATIENT COUNSELING INFORMATION. Revised: 3/2016 FULL PRESCRIBING INFORMATION: CONTENTS* WARNING: ADRENAL CRISIS IN THE SETTING OF SHOCK OR
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use LYSODREN safely and effectively. See full prescribing information for LYSODREN. LYSODREN (mitotane)
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use LYSODREN safely and effectively. See full prescribing information for LYSODREN. LYSODREN (mitotane)
Multiple sclerosis disease-modifying drugs second line treatments
 Great Ormond Street Hospital for Children NHS Foundation Trust: Information for Families Multiple sclerosis disease-modifying drugs second line treatments The following information should be read in conjunction
Great Ormond Street Hospital for Children NHS Foundation Trust: Information for Families Multiple sclerosis disease-modifying drugs second line treatments The following information should be read in conjunction
5.07.09. Aubagio. Aubagio (teriflunomide) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.07.09 Subject: Aubagio Page: 1 of 6 Last Review Date: December 5, 2014 Aubagio Description Aubagio (teriflunomide)
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.07.09 Subject: Aubagio Page: 1 of 6 Last Review Date: December 5, 2014 Aubagio Description Aubagio (teriflunomide)
PACKAGE LEAFLET: INFORMATION FOR THE USER. Dalacin C 150 mg Capsules. clindamycin hydrochloride. Dalacin C 150mg Capsules clindamycin hydrochloride
 PACKAGE LEAFLET: INFORMATION FOR THE USER PFIZER Dalacin C 150 mg Capsules clindamycin hydrochloride Dalacin C 150mg Capsules clindamycin hydrochloride PFIZER Read all of this leaflet carefully before
PACKAGE LEAFLET: INFORMATION FOR THE USER PFIZER Dalacin C 150 mg Capsules clindamycin hydrochloride Dalacin C 150mg Capsules clindamycin hydrochloride PFIZER Read all of this leaflet carefully before
Disease Modifying Therapies for MS
 Disease Modifying Therapies for MS The term disease-modifying therapy means a drug that can modify or change the course of a disease. In other words a DMT should be able to reduce the number of attacks
Disease Modifying Therapies for MS The term disease-modifying therapy means a drug that can modify or change the course of a disease. In other words a DMT should be able to reduce the number of attacks
How long will it take to work? You may begin to feel better within a few days or it may take up to six weeks after your first treatment session.
 Crohn's and Colitis UK Drug Treatment Information Improving life for people affected by inflammatory bowel diseases Infliximab This information leaflet aims to answer common questions you may have if you
Crohn's and Colitis UK Drug Treatment Information Improving life for people affected by inflammatory bowel diseases Infliximab This information leaflet aims to answer common questions you may have if you
NCCP Chemotherapy Protocol. Afatinib Monotherapy
 Afatinib Monotherapy INDICATIONS FOR USE: INDICATION Treatment of Epidermal Growth Factor Receptor (EGFR) TKI- naïve adult patients with locally advanced or metastatic non-small cell lung cancer (NSCLC)
Afatinib Monotherapy INDICATIONS FOR USE: INDICATION Treatment of Epidermal Growth Factor Receptor (EGFR) TKI- naïve adult patients with locally advanced or metastatic non-small cell lung cancer (NSCLC)
2 What you need to know before you have Ampiclox
 Reason for update: GDS 14 & QRD Updates Response to questions for variation update section 4.1 of SPC MHRA Submission Date: 6 November 2014 MHRA Approval Date: Text Date: October 2014 Text Issue and Draft
Reason for update: GDS 14 & QRD Updates Response to questions for variation update section 4.1 of SPC MHRA Submission Date: 6 November 2014 MHRA Approval Date: Text Date: October 2014 Text Issue and Draft
Clinical Criteria for Hepatitis C (HCV) Therapy
 Diagnosis Clinical Criteria for Hepatitis C (HCV) Therapy Must have chronic hepatitis C (HCV infection > 6 months), genotype and sub-genotype specified to determine the length of therapy; Liver biopsy
Diagnosis Clinical Criteria for Hepatitis C (HCV) Therapy Must have chronic hepatitis C (HCV infection > 6 months), genotype and sub-genotype specified to determine the length of therapy; Liver biopsy
Gilenya. Exceptional healthcare, personally delivered
 Gilenya Exceptional healthcare, personally delivered Your Consultant Neurologist has suggested that you may benefit from treatment with Gilenya. The decision to start this form of treatment can be difficult.
Gilenya Exceptional healthcare, personally delivered Your Consultant Neurologist has suggested that you may benefit from treatment with Gilenya. The decision to start this form of treatment can be difficult.
Teriflunomide is the active metabolite of Leflunomide, a drug employed since 1994 for the treatment of rheumatoid arthritis (Baselt, 2011).
 Page 1 of 10 ANALYTE NAME AND STRUCTURE TERIFLUNOMIDE Teriflunomide TRADE NAME Aubagio CATEGORY Antimetabolite TEST CODE PURPOSE Therapeutic Drug Monitoring GENERAL RELEVANCY BACKGROUND sclerosis. The
Page 1 of 10 ANALYTE NAME AND STRUCTURE TERIFLUNOMIDE Teriflunomide TRADE NAME Aubagio CATEGORY Antimetabolite TEST CODE PURPOSE Therapeutic Drug Monitoring GENERAL RELEVANCY BACKGROUND sclerosis. The
Drug Treatment Information for patients with Inflammatory Bowel Disease. Infliximab
 Drug Treatment Information for patients with Inflammatory Bowel Disease Infliximab Improving life for people affected by Colitis and Crohn s Disease National Association For Colitis and Crohn s Disease
Drug Treatment Information for patients with Inflammatory Bowel Disease Infliximab Improving life for people affected by Colitis and Crohn s Disease National Association For Colitis and Crohn s Disease
MEDICATION GUIDE mitoxantrone (mito-xan-trone) for injection concentrate
 MEDICATION GUIDE mitoxantrone (mito-xan-trone) for injection concentrate Read this Medication Guide before you start receiving mitoxantrone and each time you receive mitoxantrone. There may be new information.
MEDICATION GUIDE mitoxantrone (mito-xan-trone) for injection concentrate Read this Medication Guide before you start receiving mitoxantrone and each time you receive mitoxantrone. There may be new information.
READ THIS FOR SAFE AND EFFECTIVE USE OF YOUR MEDICINE PATIENT MEDICATION INFORMATION. [new-ka la]
![READ THIS FOR SAFE AND EFFECTIVE USE OF YOUR MEDICINE PATIENT MEDICATION INFORMATION. [new-ka la] READ THIS FOR SAFE AND EFFECTIVE USE OF YOUR MEDICINE PATIENT MEDICATION INFORMATION. [new-ka la]](/thumbs/35/17235501.jpg) READ THIS FOR SAFE AND EFFECTIVE USE OF YOUR MEDICINE PATIENT MEDICATION INFORMATION Pr NUCALA [new-ka la] mepolizumab lyophilized powder for subcutaneous injection Read this carefully before you start
READ THIS FOR SAFE AND EFFECTIVE USE OF YOUR MEDICINE PATIENT MEDICATION INFORMATION Pr NUCALA [new-ka la] mepolizumab lyophilized powder for subcutaneous injection Read this carefully before you start
swine flu vaccination:
 swine flu vaccination: what you need to know Flu. Protect yourself and others. Contents What is swine flu?............... 3 About the swine flu vaccine....... 4 What else do I need to know?...... 8 What
swine flu vaccination: what you need to know Flu. Protect yourself and others. Contents What is swine flu?............... 3 About the swine flu vaccine....... 4 What else do I need to know?...... 8 What
There is a risk of renal impairment in dehydrated children and adolescents.
 PACKAGE LEAFLET: INFORMATION FOR THE USER MELFEN 200mg FILM-COATED TABLETS MELFEN 400mg FILM-COATED TABLETS Ibuprofen Read all of this leaflet carefully before you start taking this medicine because it
PACKAGE LEAFLET: INFORMATION FOR THE USER MELFEN 200mg FILM-COATED TABLETS MELFEN 400mg FILM-COATED TABLETS Ibuprofen Read all of this leaflet carefully before you start taking this medicine because it
Patient Guide. Important information for patients starting therapy with LEMTRADA (alemtuzumab)
 Patient Guide Important information for patients starting therapy with LEMTRADA (alemtuzumab) This medicinal product is subject to additional monitoring. This will allow quick identification of new safety
Patient Guide Important information for patients starting therapy with LEMTRADA (alemtuzumab) This medicinal product is subject to additional monitoring. This will allow quick identification of new safety
FAQs on Influenza A (H1N1-2009) Vaccine
 FAQs on Influenza A (H1N1-2009) Vaccine 1) What is Influenza A (H1N1-2009) (swine flu) 1? Influenza A (H1N1-2009), previously known as "swine flu", is a new strain of influenza virus that spreads from
FAQs on Influenza A (H1N1-2009) Vaccine 1) What is Influenza A (H1N1-2009) (swine flu) 1? Influenza A (H1N1-2009), previously known as "swine flu", is a new strain of influenza virus that spreads from
MEDICATION GUIDE REMICADE (Rem-eh-kaid) (infliximab) Read the Medication Guide that comes with REMICADE before you receive the first treatment, and
 MEDICATION GUIDE REMICADE (Rem-eh-kaid) (infliximab) Read the Medication Guide that comes with REMICADE before you receive the first treatment, and before each time you get a treatment of REMICADE. This
MEDICATION GUIDE REMICADE (Rem-eh-kaid) (infliximab) Read the Medication Guide that comes with REMICADE before you receive the first treatment, and before each time you get a treatment of REMICADE. This
LEFLUNOMIDE (Adults)
 Shared Care Guideline DRUG: Introduction: LEFLUNOMIDE (Adults) Indication: Disease modifying drug for rheumatoid arthritis and psoriatic arthritis Licensing Information: Disease modifying drug for active
Shared Care Guideline DRUG: Introduction: LEFLUNOMIDE (Adults) Indication: Disease modifying drug for rheumatoid arthritis and psoriatic arthritis Licensing Information: Disease modifying drug for active
Exposure. What Healthcare Personnel Need to Know
 Information from the Centers for Disease Control and Prevention National Center for Infectious Diseases Divison of Healthcare Quality Promotion and Division of Viral Hepatitis For additional brochures
Information from the Centers for Disease Control and Prevention National Center for Infectious Diseases Divison of Healthcare Quality Promotion and Division of Viral Hepatitis For additional brochures
ZOVIRAX Cold Sore Cream
 Data Sheet ZOVIRAX Cold Sore Cream Aciclovir 5% w/w Presentation Topical cream Indications ZOVIRAX Cold Sore Cream is indicated for the treatment of Herpes simplex virus infections of the lips and face
Data Sheet ZOVIRAX Cold Sore Cream Aciclovir 5% w/w Presentation Topical cream Indications ZOVIRAX Cold Sore Cream is indicated for the treatment of Herpes simplex virus infections of the lips and face
MEDICATION GUIDE STELARA
 MEDICATION GUIDE STELARA (stel ar a) (ustekinumab) Injection What is the most important information I should know about STELARA? STELARA is a medicine that affects your immune system. STELARA can increase
MEDICATION GUIDE STELARA (stel ar a) (ustekinumab) Injection What is the most important information I should know about STELARA? STELARA is a medicine that affects your immune system. STELARA can increase
A guide to the accelerated elimination procedure
 A guide to the accelerated elimination procedure Prescribing information and information on adverse event reporting can be found on the last page. What is the accelerated elimination procedure? An accelerated
A guide to the accelerated elimination procedure Prescribing information and information on adverse event reporting can be found on the last page. What is the accelerated elimination procedure? An accelerated
IMPORTANT DRUG WARNING Regarding Mycophenolate-Containing Products
 Dear Healthcare Provider: Mycophenolate REMS (Risk Evaluation and Mitigation Strategy) has been mandated by the FDA (Food and Drug Administration) due to postmarketing reports showing that exposure to
Dear Healthcare Provider: Mycophenolate REMS (Risk Evaluation and Mitigation Strategy) has been mandated by the FDA (Food and Drug Administration) due to postmarketing reports showing that exposure to
SHINGLES (Herpes zoster infection)
 SHINGLES (Herpes zoster infection) What are the aims of this leaflet? This leaflet has been written to help you understand more about shingles. It will tell you what it is, what causes it, what can be
SHINGLES (Herpes zoster infection) What are the aims of this leaflet? This leaflet has been written to help you understand more about shingles. It will tell you what it is, what causes it, what can be
INTERNATIONAL CONFERENCE ON HARMONISATION OF TECHNICAL REQUIREMENTS FOR REGISTRATION OF PHARMACEUTICALS FOR HUMAN USE S1A. Current Step 4 version
 INTERNATIONAL CONFERENCE ON HARMONISATION OF TECHNICAL REQUIREMENTS FOR REGISTRATION OF PHARMACEUTICALS FOR HUMAN USE ICH HARMONISED TRIPARTITE GUIDELINE GUIDELINE ON THE NEED FOR CARCINOGENICITY STUDIES
INTERNATIONAL CONFERENCE ON HARMONISATION OF TECHNICAL REQUIREMENTS FOR REGISTRATION OF PHARMACEUTICALS FOR HUMAN USE ICH HARMONISED TRIPARTITE GUIDELINE GUIDELINE ON THE NEED FOR CARCINOGENICITY STUDIES
IF YOU ARE RECEIVING TREATMENT WITH TYSABRI FOR RELAPSING-REMITTING MS (NATALIZUMAB)
 IF YOU ARE RECEIVING (NATALIZUMAB) TREATMENT WITH TYSABRI FOR RELAPSING-REMITTING MS Read the patient information leaflet that accompanies the medicine carefully. 1 This brochure is a supplement to the
IF YOU ARE RECEIVING (NATALIZUMAB) TREATMENT WITH TYSABRI FOR RELAPSING-REMITTING MS Read the patient information leaflet that accompanies the medicine carefully. 1 This brochure is a supplement to the
Leflunomide Leflunomide
 Drug information Leflunomide Leflunomide This leaflet provides information on leflunomide and will answer any questions you have about the treatment. Arthritis Research UK produce and print our booklets
Drug information Leflunomide Leflunomide This leaflet provides information on leflunomide and will answer any questions you have about the treatment. Arthritis Research UK produce and print our booklets
Bile Duct Diseases and Problems
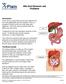 Bile Duct Diseases and Problems Introduction A bile duct is a tube that carries bile between the liver and gallbladder and the intestine. Bile is a substance made by the liver that helps with digestion.
Bile Duct Diseases and Problems Introduction A bile duct is a tube that carries bile between the liver and gallbladder and the intestine. Bile is a substance made by the liver that helps with digestion.
Temozolomide (oral) with concurrent radiotherapy to the brain
 Temozolomide (oral) with concurrent radiotherapy to the brain Temozolomide (oral) with concurrent radiotherapy to the brain This leaflet is offered as a guide to you and your family. You will find it useful
Temozolomide (oral) with concurrent radiotherapy to the brain Temozolomide (oral) with concurrent radiotherapy to the brain This leaflet is offered as a guide to you and your family. You will find it useful
ICH Topic S 1 A The Need for Carcinogenicity Studies of Pharmaceuticals. Step 5
 European Medicines Agency July 1996 CPMP/ICH/140/95 ICH Topic S 1 A The Need for Carcinogenicity Studies of Pharmaceuticals Step 5 NOTE FOR GUIDANCE ON THE NEED FOR CARCINOGENICITY STUDIES OF PHARMACEUTICALS
European Medicines Agency July 1996 CPMP/ICH/140/95 ICH Topic S 1 A The Need for Carcinogenicity Studies of Pharmaceuticals Step 5 NOTE FOR GUIDANCE ON THE NEED FOR CARCINOGENICITY STUDIES OF PHARMACEUTICALS
Understanding your Tecfidera treatment
 Understanding your Tecfidera treatment Information for patients who have been prescribed treatment with Tecfidera. (dimethyl fumarate) Contents About Multiple Sclerosis (MS) What is MS? Symptoms of MS
Understanding your Tecfidera treatment Information for patients who have been prescribed treatment with Tecfidera. (dimethyl fumarate) Contents About Multiple Sclerosis (MS) What is MS? Symptoms of MS
Understanding your Tecfidera treatment
 Understanding your Tecfidera treatment Information for patients who have been prescribed treatment with Tecfidera. (dimethyl fumarate) Contents About Multiple Sclerosis (MS) What is MS? Symptoms of MS
Understanding your Tecfidera treatment Information for patients who have been prescribed treatment with Tecfidera. (dimethyl fumarate) Contents About Multiple Sclerosis (MS) What is MS? Symptoms of MS
Chickenpox in pregnancy: what you need to know
 Chickenpox in pregnancy: what you need to know First published December 2003 Revised edition published November 2008 What is chickenpox? Chickenpox is a very infectious illness caused by a virus called
Chickenpox in pregnancy: what you need to know First published December 2003 Revised edition published November 2008 What is chickenpox? Chickenpox is a very infectious illness caused by a virus called
1.3.1.3 PACKAGE LEAFLET
 Sandoz Business use only Page 1 of 8 1.3.1.3 PACKAGE LEAFLET Sandoz Business use only Page 2 of 8 Package leaflet: Information for the patient Cefuhexal 250 mg film-coated tablets Cefuhexal 500 mg film-coated
Sandoz Business use only Page 1 of 8 1.3.1.3 PACKAGE LEAFLET Sandoz Business use only Page 2 of 8 Package leaflet: Information for the patient Cefuhexal 250 mg film-coated tablets Cefuhexal 500 mg film-coated
MEDICATION GUIDE JUXTAPID (JUKS-tuh-pid) (lomitapide) capsules
 MEDICATION GUIDE JUXTAPID (JUKS-tuh-pid) (lomitapide) capsules Read this Medication Guide before your treatment. What is the most important information I should know about JUXTAPID? JUXTAPID is available
MEDICATION GUIDE JUXTAPID (JUKS-tuh-pid) (lomitapide) capsules Read this Medication Guide before your treatment. What is the most important information I should know about JUXTAPID? JUXTAPID is available
Eastern Health MS Service. Tysabri Therapy. Information for People with MS and their Families
 Eastern Health MS Service Tysabri Therapy Information for People with MS and their Families The Eastern Health MS Service has developed this information for you as a guide through what will happen to you
Eastern Health MS Service Tysabri Therapy Information for People with MS and their Families The Eastern Health MS Service has developed this information for you as a guide through what will happen to you
MEDICATION GUIDE ACTOPLUS MET (ak-tō-plus-met) (pioglitazone hydrochloride and metformin hydrochloride) tablets
 MEDICATION GUIDE (ak-tō-plus-met) (pioglitazone hydrochloride and metformin hydrochloride) tablets Read this Medication Guide carefully before you start taking and each time you get a refill. There may
MEDICATION GUIDE (ak-tō-plus-met) (pioglitazone hydrochloride and metformin hydrochloride) tablets Read this Medication Guide carefully before you start taking and each time you get a refill. There may
Suspected pulmonary embolism (PE) in pregnant women
 Suspected pulmonary embolism (PE) in pregnant women What is a pulmonary embolus? A deep vein thrombosis (DVT) is a blood clot that forms in one of the deep veins of the leg. If the clot moves to the lung,
Suspected pulmonary embolism (PE) in pregnant women What is a pulmonary embolus? A deep vein thrombosis (DVT) is a blood clot that forms in one of the deep veins of the leg. If the clot moves to the lung,
Disease Modifying Therapies for MS
 Disease Modifying Therapies for MS The term disease-modifying therapy (DMT) means a drug that can modify or change the course of a disease. In other words a DMT should be able to reduce the number of attacks
Disease Modifying Therapies for MS The term disease-modifying therapy (DMT) means a drug that can modify or change the course of a disease. In other words a DMT should be able to reduce the number of attacks
A Patient s Guide to Treating Relapsed Chronic Lymphocytic Leukemia
 A Patient s Guide to Treating Relapsed Chronic Lymphocytic Leukemia Last week, my granddaughter asked me to take her fishing. Yesterday, we bought her a rod. Today, she caught her first speckled trout.
A Patient s Guide to Treating Relapsed Chronic Lymphocytic Leukemia Last week, my granddaughter asked me to take her fishing. Yesterday, we bought her a rod. Today, she caught her first speckled trout.
Oxford University Hospitals. NHS Trust. Department of Neurology Natalizumab (Tysabri) for Multiple Sclerosis. Information for patients
 Oxford University Hospitals NHS Trust Department of Neurology Natalizumab (Tysabri) for Multiple Sclerosis Information for patients page 2 What is Natalizumab and what is it used for? Natalizumab is an
Oxford University Hospitals NHS Trust Department of Neurology Natalizumab (Tysabri) for Multiple Sclerosis Information for patients page 2 What is Natalizumab and what is it used for? Natalizumab is an
MS Treatments Aubagio TM
 1 MSology Essentials Series Aubagio TM (teriflunomide) Developed by MSology with the invaluable assistance of multiple sclerosis nurse advisors: Bonnie Blain Central Alberta MS Clinic, Red Deer, Alberta
1 MSology Essentials Series Aubagio TM (teriflunomide) Developed by MSology with the invaluable assistance of multiple sclerosis nurse advisors: Bonnie Blain Central Alberta MS Clinic, Red Deer, Alberta
QUESTIONS TO ASK MY DOCTOR
 Be a part of the treatment decision by asking questions QUESTIONS TO ASK MY DOCTOR FOR PATIENTS WITH ADVANCED STOMACH OR GASTROESOPHAGEAL JUNCTION (GEJ) CANCER CYRAMZA (ramucirumab) is used alone or in
Be a part of the treatment decision by asking questions QUESTIONS TO ASK MY DOCTOR FOR PATIENTS WITH ADVANCED STOMACH OR GASTROESOPHAGEAL JUNCTION (GEJ) CANCER CYRAMZA (ramucirumab) is used alone or in
MEDICATION GUIDE POMALYST (POM-uh-list) (pomalidomide) capsules. What is the most important information I should know about POMALYST?
 MEDICATION GUIDE POMALYST (POM-uh-list) (pomalidomide) capsules What is the most important information I should know about POMALYST? Before you begin taking POMALYST, you must read and agree to all of
MEDICATION GUIDE POMALYST (POM-uh-list) (pomalidomide) capsules What is the most important information I should know about POMALYST? Before you begin taking POMALYST, you must read and agree to all of
FDA-Approved Patient Labeling IMPLANON (etonogestrel implant) Subdermal Use
 FDA-Approved Patient Labeling IMPLANON (etonogestrel implant) Subdermal Use IMPLANON does not protect against HIV infection (the virus that causes AIDS) or other sexually transmitted diseases. Read this
FDA-Approved Patient Labeling IMPLANON (etonogestrel implant) Subdermal Use IMPLANON does not protect against HIV infection (the virus that causes AIDS) or other sexually transmitted diseases. Read this
What You Need to Know About Lung Cancer Immunotherapy
 What You Need to Know About Lung Cancer Immunotherapy Lung.org/immunotherapy What is immunotherapy? Immunotherapy for cancer, sometimes called immune-oncology, is a type of medicine that treats cancer
What You Need to Know About Lung Cancer Immunotherapy Lung.org/immunotherapy What is immunotherapy? Immunotherapy for cancer, sometimes called immune-oncology, is a type of medicine that treats cancer
Treating Chronic Hepatitis C. A Review of the Research for Adults
 Treating Chronic Hepatitis C A Review of the Research for Adults Is This Information Right for Me? Yes, this information is right for you if: Your doctor* has told you that you have chronic hepatitis C.
Treating Chronic Hepatitis C A Review of the Research for Adults Is This Information Right for Me? Yes, this information is right for you if: Your doctor* has told you that you have chronic hepatitis C.
Hull & East Riding Prescribing Committee
 Hull & East Riding Prescribing Committee Guideline on Prescribing of Gonadorelin (GnRH) Analogues and Progesterone Receptor Modulators in treatment of Endometriosis or prior to Endometrial Ablation, or
Hull & East Riding Prescribing Committee Guideline on Prescribing of Gonadorelin (GnRH) Analogues and Progesterone Receptor Modulators in treatment of Endometriosis or prior to Endometrial Ablation, or
Rituximab. Drug information Rituximab. This leaflet provides information on rituximab and will answer any questions you have about the treatment.
 Drug information Rituximab Rituximab This leaflet provides information on rituximab and will answer any questions you have about the treatment. Arthritis Research UK produce and print our booklets entirely
Drug information Rituximab Rituximab This leaflet provides information on rituximab and will answer any questions you have about the treatment. Arthritis Research UK produce and print our booklets entirely
Answering your questions on Chronic Myeloid Leukaemia (CML)
 Answering your questions on Chronic Myeloid Leukaemia (CML) Your guide to understanding CML and Glivec (imatinib) treatment The information in this booklet is designed to help you understand chronic myeloid
Answering your questions on Chronic Myeloid Leukaemia (CML) Your guide to understanding CML and Glivec (imatinib) treatment The information in this booklet is designed to help you understand chronic myeloid
Your NHL Journey. RITUXAN for Follicular Lymphoma and Diffuse Large B-cell Lymphoma (DLBCL) Indications. Important Safety Information
 * Your NHL Journey RITUXAN for Follicular Lymphoma and Diffuse Large B-cell Lymphoma (DLBCL) * Non-Hodgkin s Lymphoma Indications RITUXAN (rituximab) is indicated for the treatment of: Follicular CD20-positive
* Your NHL Journey RITUXAN for Follicular Lymphoma and Diffuse Large B-cell Lymphoma (DLBCL) * Non-Hodgkin s Lymphoma Indications RITUXAN (rituximab) is indicated for the treatment of: Follicular CD20-positive
PACKAGE LEAFLET: INFORMATION FOR THE USER. PARACETAMOL MACOPHARMA 10 mg/ml, solution for infusion. Paracetamol
 PACKAGE LEAFLET: INFORMATION FOR THE USER PARACETAMOL MACOPHARMA 10 mg/ml, solution for infusion Paracetamol Read all of this leaflet carefully before you start using this medicine. Keep this leaflet.
PACKAGE LEAFLET: INFORMATION FOR THE USER PARACETAMOL MACOPHARMA 10 mg/ml, solution for infusion Paracetamol Read all of this leaflet carefully before you start using this medicine. Keep this leaflet.
IMMUNOTHERAPY FOR THE TREATMENT OF LUNG CANCER
 IMMUNOTHERAPY FOR THE TREATMENT OF LUNG CANCER BOOKLET SUPPLEMENT Immunotherapy Immunotherapy is one of the most exciting new approaches for treating several types of cancer, including lung cancer. Immunotherapies
IMMUNOTHERAPY FOR THE TREATMENT OF LUNG CANCER BOOKLET SUPPLEMENT Immunotherapy Immunotherapy is one of the most exciting new approaches for treating several types of cancer, including lung cancer. Immunotherapies
An overview of CLL care and treatment. Dr Dean Smith Haematology Consultant City Hospital Nottingham
 An overview of CLL care and treatment Dr Dean Smith Haematology Consultant City Hospital Nottingham What is CLL? CLL (Chronic Lymphocytic Leukaemia) is a type of cancer in which the bone marrow makes too
An overview of CLL care and treatment Dr Dean Smith Haematology Consultant City Hospital Nottingham What is CLL? CLL (Chronic Lymphocytic Leukaemia) is a type of cancer in which the bone marrow makes too
Medication Guide Enbrel (en-brel) (etanercept)
 Medication Guide Enbrel (en-brel) (etanercept) Read the Medication Guide that comes with Enbrel before you start using it and each time you get a refill. There may be new information. This Medication Guide
Medication Guide Enbrel (en-brel) (etanercept) Read the Medication Guide that comes with Enbrel before you start using it and each time you get a refill. There may be new information. This Medication Guide
PATIENT INFORMATION LEAFLET. CEFALEXIN 250 mg AND 500 mg CAPSULES CEFALEXIN
 PATIENT INFORMATION LEAFLET CEFALEXIN 250 mg AND 500 mg CAPSULES CEFALEXIN Read all of this leaflet carefully before you start taking this medicine. - Keep this leaflet. You may need to read it again.
PATIENT INFORMATION LEAFLET CEFALEXIN 250 mg AND 500 mg CAPSULES CEFALEXIN Read all of this leaflet carefully before you start taking this medicine. - Keep this leaflet. You may need to read it again.
Early Access to Medicines Scheme Treatment protocol Information for patients
 Early Access to Medicines Scheme Treatment protocol Information for patients Introduction The aim of the Early Access to Medicines Scheme (EAMS) is to provide earlier availability of promising new unlicensed
Early Access to Medicines Scheme Treatment protocol Information for patients Introduction The aim of the Early Access to Medicines Scheme (EAMS) is to provide earlier availability of promising new unlicensed
Series 1 Case Studies Adverse Events that Represent Unanticipated Problems: Reporting Required
 Welcome! This document contains three (3) series of Case Study examples that will demonstrate all four OHSU reporting categories (#1 4) as well as examples of events that are considered not reportable.
Welcome! This document contains three (3) series of Case Study examples that will demonstrate all four OHSU reporting categories (#1 4) as well as examples of events that are considered not reportable.
Biologic Treatments for Rheumatoid Arthritis
 Biologic Treatments Rheumatoid Arthritis (also known as cytokine inhibitors, TNF inhibitors, IL 1 inhibitor, or Biologic Response Modifiers) Description Biologics are new class of drugs that have been
Biologic Treatments Rheumatoid Arthritis (also known as cytokine inhibitors, TNF inhibitors, IL 1 inhibitor, or Biologic Response Modifiers) Description Biologics are new class of drugs that have been
This regimen has low emetogenic potential refer to local protocol None required routinely. Baseline results valid for 7 days. Results valid for 72 hrs
 Regimen : Ipilimumab for Advanced Melanoma ICD10 code Codes pre-fixed with C43. Indication Regimen detail Ipilimumab is recommended as an option for treating advanced (unresectable or metastatic) melanoma
Regimen : Ipilimumab for Advanced Melanoma ICD10 code Codes pre-fixed with C43. Indication Regimen detail Ipilimumab is recommended as an option for treating advanced (unresectable or metastatic) melanoma
Topical Tacrolimus or Pimecrolimus for the treatment of mild, moderate or severe atopic eczema. Effective Shared Care Agreement
 Topical Tacrolimus or Pimecrolimus for the treatment of mild, moderate or severe atopic eczema. Effective Shared Care Agreement A Copy of this page signed by all three parties should be retained in the
Topical Tacrolimus or Pimecrolimus for the treatment of mild, moderate or severe atopic eczema. Effective Shared Care Agreement A Copy of this page signed by all three parties should be retained in the
12.5 VI.2 ELEMENTS FOR A PUBLIC SUMMARY
 12.5 VI.2 ELEMENTS FOR A PUBLIC SUMMARY 12.5.1 VI.2.1 Overview of disease epidemiology Breast cancer affects 10 12% of women globally, and is more common among older people. Some cancer cells have proteins
12.5 VI.2 ELEMENTS FOR A PUBLIC SUMMARY 12.5.1 VI.2.1 Overview of disease epidemiology Breast cancer affects 10 12% of women globally, and is more common among older people. Some cancer cells have proteins
Like cocaine, heroin is a drug that is illegal in some areas of the world. Heroin is highly addictive.
 Heroin Introduction Heroin is a powerful drug that affects the brain. People who use it can form a strong addiction. Addiction is when a drug user can t stop taking a drug, even when he or she wants to.
Heroin Introduction Heroin is a powerful drug that affects the brain. People who use it can form a strong addiction. Addiction is when a drug user can t stop taking a drug, even when he or she wants to.
Update in Hematology Oncology Targeted Therapies. Mark Holguin
 Update in Hematology Oncology Targeted Therapies Mark Holguin 25 years ago Why I chose oncology People How to help people with possibly the most difficult thing they may have to deal with Science Turning
Update in Hematology Oncology Targeted Therapies Mark Holguin 25 years ago Why I chose oncology People How to help people with possibly the most difficult thing they may have to deal with Science Turning
AUBAGIO Conversation Starter
 AUBAGIO Conversation Starter When you are choosing a multiple sclerosis (MS) treatment for the first time or are considering switching your MS therapy, there can be a lot to think about for both you and
AUBAGIO Conversation Starter When you are choosing a multiple sclerosis (MS) treatment for the first time or are considering switching your MS therapy, there can be a lot to think about for both you and
Trileptal (Oxcarbazepine)
 Brand and Generic Names: Trileptal Tablets: 150mg, 300mg, 600mg Liquid Suspension: 300mg/5mL Generic name: oxcarbazepine What is Trileptal and what does it treat? Trileptal (Oxcarbazepine) Oxcarbazepine
Brand and Generic Names: Trileptal Tablets: 150mg, 300mg, 600mg Liquid Suspension: 300mg/5mL Generic name: oxcarbazepine What is Trileptal and what does it treat? Trileptal (Oxcarbazepine) Oxcarbazepine
Thioctacid 600 T Solution for Injection contains 600 mg alpha-lipoic acid
 Package Leaflet: Information for the User Thioctacid 600 T Solution for Injection contains 600 mg alpha-lipoic acid For use in adults Active substance: Alpha-lipoic acid, Trometamol salt (1:1) Read all
Package Leaflet: Information for the User Thioctacid 600 T Solution for Injection contains 600 mg alpha-lipoic acid For use in adults Active substance: Alpha-lipoic acid, Trometamol salt (1:1) Read all
PACKAGE LEAFLET: INFORMATION FOR THE USER PROPECIA 1 mg film-coated Tablets (finasteride)
 PACKAGE LEAFLET: INFORMATION FOR THE USER PROPECIA 1 mg film-coated Tablets (finasteride) This medicine is for use in men only Read all of this leaflet carefully before you start to take this medicine.
PACKAGE LEAFLET: INFORMATION FOR THE USER PROPECIA 1 mg film-coated Tablets (finasteride) This medicine is for use in men only Read all of this leaflet carefully before you start to take this medicine.
X-Plain Psoriasis Reference Summary
 X-Plain Psoriasis Reference Summary Introduction Psoriasis is a long-lasting skin disease that causes the skin to become inflamed. Patches of thick, red skin are covered with silvery scales. It affects
X-Plain Psoriasis Reference Summary Introduction Psoriasis is a long-lasting skin disease that causes the skin to become inflamed. Patches of thick, red skin are covered with silvery scales. It affects
1. What Daktarin oral gel is and what it is used for
 PACKAGE LEAFLET: INFORMATION FOR THE USER Daktarin oral gel Miconazole Daktarin is a registered trademark Read all of this leaflet carefully before you start using this medicine. Keep this leaflet. You
PACKAGE LEAFLET: INFORMATION FOR THE USER Daktarin oral gel Miconazole Daktarin is a registered trademark Read all of this leaflet carefully before you start using this medicine. Keep this leaflet. You
EMA and Progressive Multifocal Leukoencephalopathy.
 EMA and Progressive Multifocal Leukoencephalopathy. ENCePP Plenary, London 23 November 2011 Presented by: Henry Fitt Head of Coordination & Networking, Pharmacovigilance & Risk Management An agency of
EMA and Progressive Multifocal Leukoencephalopathy. ENCePP Plenary, London 23 November 2011 Presented by: Henry Fitt Head of Coordination & Networking, Pharmacovigilance & Risk Management An agency of
MS Treatments Gilenya
 1 MSology Essentials Series Gilenya (fingolimod) Developed by MSology with the invaluable assistance of multiple sclerosis nurse advisors: Trudy Campbell Dalhousie MS Research Unit, Capital Health, Halifax,
1 MSology Essentials Series Gilenya (fingolimod) Developed by MSology with the invaluable assistance of multiple sclerosis nurse advisors: Trudy Campbell Dalhousie MS Research Unit, Capital Health, Halifax,
EFFIMET 1000 XR Metformin Hydrochloride extended release tablet
 BRAND NAME: Effimet XR. THERAPEUTIC CATEGORY: Anti-Diabetic PHARMACOLOGIC CLASS: Biguanides EFFIMET 1000 XR Metformin Hydrochloride extended release tablet COMPOSITION AND PRESENTATION Composition Each
BRAND NAME: Effimet XR. THERAPEUTIC CATEGORY: Anti-Diabetic PHARMACOLOGIC CLASS: Biguanides EFFIMET 1000 XR Metformin Hydrochloride extended release tablet COMPOSITION AND PRESENTATION Composition Each
Summary of the risk management plan (RMP) for Tritanrix HB [Diphtheria, tetanus, pertussis (whole cell) and hepatitis B (rdna) vaccine (adsorbed)]
![Summary of the risk management plan (RMP) for Tritanrix HB [Diphtheria, tetanus, pertussis (whole cell) and hepatitis B (rdna) vaccine (adsorbed)] Summary of the risk management plan (RMP) for Tritanrix HB [Diphtheria, tetanus, pertussis (whole cell) and hepatitis B (rdna) vaccine (adsorbed)]](/thumbs/27/12172665.jpg) EMA/14365/2014 Summary of the risk management plan (RMP) for Tritanrix HB [Diphtheria, tetanus, pertussis (whole cell) and hepatitis B (rdna) vaccine (adsorbed)] Overview of disease epidemiology Diphtheria
EMA/14365/2014 Summary of the risk management plan (RMP) for Tritanrix HB [Diphtheria, tetanus, pertussis (whole cell) and hepatitis B (rdna) vaccine (adsorbed)] Overview of disease epidemiology Diphtheria
MINISTRY OF HEALTH PANDEMIC INFLUENZA A / H1N1 2009 VACCINE FREQUENTLY ASKED QUESTIONS
 Government of the Republic of Trinidad and Tobago MINISTRY OF HEALTH PANDEMIC INFLUENZA A / H1N1 2009 VACCINE FREQUENTLY ASKED QUESTIONS Influenza vaccines are one of the most effective ways to protect
Government of the Republic of Trinidad and Tobago MINISTRY OF HEALTH PANDEMIC INFLUENZA A / H1N1 2009 VACCINE FREQUENTLY ASKED QUESTIONS Influenza vaccines are one of the most effective ways to protect
What You Need to Know About LEMTRADA (alemtuzumab) Treatment: A Patient Guide
 For Patients What You Need to Know About LEMTRADA (alemtuzumab) Treatment: A Patient Guide Patients: Your doctor or nurse will go over this patient guide with you. It is important to ask any questions
For Patients What You Need to Know About LEMTRADA (alemtuzumab) Treatment: A Patient Guide Patients: Your doctor or nurse will go over this patient guide with you. It is important to ask any questions
Patient Information STRIBILD (STRY-bild) (elvitegravir, cobicistat, emtricitabine, and tenofovir disoproxil fumarate) Tablets Important: Ask your
 Patient Information STRIBILD (STRY-bild) (elvitegravir, cobicistat, emtricitabine, and tenofovir disoproxil fumarate) Tablets Important: Ask your healthcare provider or pharmacist about medicines that
Patient Information STRIBILD (STRY-bild) (elvitegravir, cobicistat, emtricitabine, and tenofovir disoproxil fumarate) Tablets Important: Ask your healthcare provider or pharmacist about medicines that
