GeNei TM GFP Cloning Teaching Kit Manual
|
|
|
- Marcus Porter
- 9 years ago
- Views:
Transcription
1 Teaching Kit Manual Cat No Revision No.:
2 CONTENTS Page No. Objective 3 Principle 3 Kit Description 7 Materials Provided 9 Procedure 11 Observation & Interpretation 15 Appendix 16 Ordering Information 17 1
3 Objective: To understand the basic concept of cloning and perform some of the steps involved, using the GFP gene (Green Fluorescent Protein). Principle: Molecular cloning or gene cloning involves insertion of a DNA fragment (gene of interest) into a cloning vector. The recombinant vector is subsequently transformed into a suitable host to generate the desired clones. Gene cloning basically involves the following steps: Isolation of vector & insert DNA. Cutting of DNA. Joining of DNA. Amplifying the recombinant DNA. Screening for clones. Isolation of vector and insert DNA: Insert DNA: Foreign DNA is isolated from cells by disrupting lipid membrane with detergents and is subsequently purified by RNase treatment and phenol/chloroform extractions. Insert DNA (having the gene of interest) is then obtained from the purified foreign DNA by shearing, PCR amplification or restriction enzyme digestion. 2 3
4 Vector DNA: Cloning vectors are circular DNA molecules in which DNA fragments/insert are maintained and amplified. Commonly used cloning vector include: Plasmids, Cosmids, Phagemids and Yeast artificial chromosomes etc. Vectors have the following features: o An origin of replication recognized by the host cell replication machinery. o A selection marker that enables the identification of transformants and/or transfectants. o One or multiple restriction endonuclease recognition sites that allow the insertion of gene of interest. Vector DNA purification protocols vary depending upon the type of vector chosen for cloning experiments. Smaller sized cloning vectors like plasmids, cosmids and phagemids are easier to purify and are usually done by alkaline lysis method and subsequent phenol/chloroform extractions. Cutting of DNA: In order to carry out recombination between the vector and the insert DNA, it is necessary to specifically and reproducibly cleave DNA. Restriction enzymes are the scissors of molecular genetics, which recognize specific nucleotide sequences and cut the DNA at these points to generate sticky or blunt ends. Generally, the same restriction enzyme is used to cleave both the insert and vector DNA. Following restriction enzyme digestion of vector, the DNA is dephosphorylated i.e., 5 phosphate group is removed by treating with alkaline phosphatase. This is done to reduce self ligation of vector molecules by T4 DNA ligase. However, this does not prevent ligation of insert to the vector as 5 phosphate groups of insert are intact and thus efficiency of insert ligation to the vector increases. Another method employed to prevent self ligation of vector is to cut the DNA with two different restriction enzymes resulting in mismatched ends that cannot ligate. Joining of DNA: Once the insert and vector DNA have been isolated and cut with specific restriction enzyme(s), they must be ligated. DNA ligase is the glue of molecular genetics that holds the DNA together by creating a phosphodiester bond between the two DNA ends to create a recombinant DNA molecule. When a single restriction enzyme is used to cut the vector and insert DNA, ligation occurs in either direction/orientation. However, insert can be forced to ligate to the vector in the desired orientation, by using two different restriction enzymes to cut both the vector and insert DNA. Amplifying the recombinant DNA: The recombinant DNA thus created is introduced into a compatible host such that the vector DNA is replicated resulting in amplification of the insert DNA. When plasmid DNA is introduced into host cell, the process is referred to as transformation and in case of viral DNA the process is transduction. A successful vector DNA transfer is monitored by selecting for the genetic marker present on the vector. For eg., plasmid vectors usually have dominant selectable markers such as antibiotic resistance genes that confer growth on media containing appropriate concentration of an antibiotic. In case of viral vectors, one observes for formation of plaques. 4 5
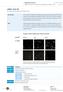
5 Screening for clones: The final step involved in cloning is the means to detect the right clone among a population of colonies or plaques. This screening mechanism will depend upon the vector chosen as the cloning vehicle. For example, pbr322 (plasmid vector) carries genes for ampicillin resistance and tetracycline resistance. Both these genes have sites for restriction enzyme that allow for cloning and selection of recombinants by insertional inactivation of one of the genes. Hence, cells with vector alone will be resistant to both the antibiotics, but cells with recombinant DNA will be resistant to only one antibiotic and can be selected by replica plating. Kit Description: In this kit, a vector and an insert DNA (GFP gene isolated from a bioluminescent jelly fish Aequorea victoria) are provided along with the host (E. coli strain DH5α). Using the kit students will carry out the following experiments: Ligation: GFP gene will be ligated to the vector using the enzyme T4 DNA ligase at 16 C. Since, the vector and insert DNA have been cleaved with the same single restriction enzyme, ligation of insert to vector will occur in either direction/orientation. Preparation of competent cells: Host cells will be made competent by using a solution having salts like calcium chloride, magnesium chloride, manganese chloride, etc. Transformation: The ligated sample will then be transformed into competent E.coli cells and plated on LB plates containing ampicillin. Cells having recombinant DNA or self ligated vector DNA will grow since the plasmid carrying ampicillin resistance gene confers antibiotic resistance to the host cells. Screening: The plates will then be visualized under UV light to screen for clones. This is because, the unique 3 dimensional conformation of the GFP releases energy in the form of visible green light on exposure to UV light. Hence, only cells having the insert in the right orientation will glow and these are the clones. However, there will be other clones where in the insert is ligated in the reverse orientation resulting in no expression of the protein. These clones will not glow on exposure to UV light. 6 7
6 KT60 : The kit is designed to carry out 5 ligation reactions of GFP gene to the vector and its transformation into competent E. coli (DH5α) cells. Note: Single batch of competent cells will be prepared, sufficient for 5 transformation experiments. Duration of experiment: Experiment is carried out over a span of 3 days, approximate time taken on each day is indicated below: Day 1 : 3 hours (Revival of host and Ligation of insert to vector) Day 2 : 6 hours (Preparation of competent cells and Transformation) Day 3 : 45 minutes (Observation and Interpretation) Materials Provided: The list below provides information about the materials supplied in the kit. The products should be stored as suggested. Use the kit within 6 months of arrival. Quantity Materials Cat # (5 expts.) Store Ampicillin 100 mg 4 C Host 1 vial 4 C Solution A 40 ml 4 C Control DNA 10 µl -20 C T4 DNA Ligase 5.0 µl -20 C 10X Ligase Assay Buffer 10 µl -20 C Insert 20 µl -20 C Vector DNA 10 µl -20 C LB Broth 15 g RT Agar 5 g RT 1.5 ml vials 25 Nos. RT Materials Required: Equipment : Centrifuge (preferably refrigerated), UV transilluminator (312 nm), Spectrophotometer. Glassware : Capped centrifuge tubes, Conical flask, Petri plates, Test tubes. Reagent : Distilled water. Other Requirements: Crushed ice, Cuvette (of 1 cm path length), Micropipette, Tips, Thermometer, Water bath. 8 9
7 Note: Read the entire procedure before starting the experiment. All microbiological procedures should be done under aseptic conditions. Revive the strain as soon as the lyophilized vial is opened. Prepare competent cells within 3 days of reviving the strain. Entire procedure of preparing competent cells should be done under aseptic conditions. Carry out transformation as soon as the competent cells are prepared. Storage of competent cells may result in poor/no transformants. Transformation efficiency of competent cells should be more than 1 x 10 5 µg of DNA. Efficiency lower than this may lead to lower number of transformants. Pre-cool tubes, pipettes, centrifuge tubes and Solution A, prior to preparation of competent cells. Solution A supplied is sterile, handle under aseptic conditions. 5 ligation reactions and transformations are to be done simultaneously. Ensure that all the required components are ready prior to starting the experiment. For preparation of media, antibiotic, etc., refer appendix. Procedure: Day 1: Revival of Host 1. Break open the lyophilized vial, add 0.1 ml of LB broth. 2. Streak a loopful of the suspension onto LB plate (in duplicates). 3. Incubate the plates at 37 C, overnight. Ligation of Vector to Insert 4. Thaw the ligase assay buffer, vector and insert DNA. Note: Thaw the ligase assay buffer vial on ice, store at -20 C immediately after use. 5. Set up ligation reaction as follows: Water : 11 µl Vector DNA : 2 µl Insert DNA : 4 µl Ligase assay buffer : 2 µl T4 DNA ligase : 1 µl Mix the contents by tapping gently and incubate at 16 C waterbath, overnight. Note: Set up five ligation reactions simultaneously. Day 2: Preparation of Competent Cells 6. Pick moderate sized colonies from the LB plate and inoculate into 100 ml of LB broth (in a 1 litre conical flask). 7. Incubate at 37 C, in a shaker. Grow until OD A 600 reaches 0.3, this takes about 2-3 hours. 8. Chill the culture flask on ice for minutes
8 9. Transfer the culture aseptically into sterile centrifuge tubes and spin down at 6000 rpm for 8 minutes, preferably in a refrigerated centrifuge at 4 C or spin at Room temperature (RT). 10. Discard the supernatant. 11. Resuspend the cell pellet very gently in small volume of ice-cold solution A (approximately 2 ml), using a pre-chilled pipette. Care must be taken not to remove the tubes from ice during resuspension. Add remaining 33 ml of solution A, resuspend gently. 12. Keep the centrifuge tubes on ice for 20 minutes. Centrifuge at 3500 rpm for 15 minutes at 4 C or spin at RT. 13. Discard the supernatant and chill the tube on ice. Resuspend the pellet in 3 ml of ice-cold solution A. Note: Resuspension is to be done gently as cells are very fragile at this stage. 14. Aliquot 0.6 ml of the above suspension into 5 different pre-chilled 1.5 ml vials, aseptically. 15. Heat inactivate the ligated samples at 65 C for 10 minutes. Spin at 5000 rpm for 2 minutes and keep the vials on ice. 16. Aliquot 2 µl (10 ng) of control DNA each into 5 prechilled 1.5 ml vials. Transformation: Note: Use single aliquot of competent cells (0.6 ml) for one set of transformation experiment. 17. Take a single vial of ligated sample and control DNA. To this add 200 µl each of competent cells. Tap the vials gently and incubate on ice for 20 minutes. 18. Label the remaining 200 µl of competent cells as nontransformed cells, place on ice till the plating step (step 24). 19. Tap all the vials gently and incubate on ice for 20 minutes. 20. Heat shock the cells by placing the vial(s) in 42 C water bath for 2 minutes, then return the vials to ice and chill for 5 minutes. 21. Add 0.5 ml of LB broth aseptically to the vial(s) and incubate at 37 C (shaker) for an hour. This is to allow bacteria to recover and express the protein. 22. Pipette 200 µl each from the vials transformed with the ligated mix onto LB-Amp plates and spread thoroughly using spreader/pipette. 23. Pipette 200 µl of LB onto a LB -Amp plate and spread 20 ìl of the cells transformed with control DNA. Label this as control plate. 24. Plate 200 µl of non-transformed cells onto another LB-Amp plate to check for contamination. Label this as non-transformed plate. 25. Incubate the plates at 37 C, overnight. 26. Observe the plates under UV-light (312 nm). Note: If observed add 254 nm of less intensity of glow could vary. 27. Count the number of colonies on the plate and calculate Transformation Efficiency. Note: Steps 17 to 27 explains the protocol for one set of transformation experiment. Carry out 5 such transformations simultaneously
9 Transformation Efficiency is expressed as: No. of transformants/µg of DNA. Observation: Record your observation as follows: Calculation: Transformation Efficiency is: No. of colonies x 1000 ng Amount of DNA plated (in ng) = /µg Plate Growth Glow Control plate Ligated plate Non-transformed plate For example: Amount of DNA transformed = 10 ng Volume of culture plated = 200 µl (of 1 ml) Thus, amount of DNA plated = 2 ng If no. of colonies observed on plating 2 ng = 200 Transformation Efficiency = 200 x = 1 x 10 5 /µg Denote : +ve: when growth or glow is observed. -ve: when growth or glow is not observed. Interpretation: On plating cells transformed with ligated sample/control DNA on LB-Amp plates, growth is observed as the vector DNA confers antibiotic resistance to the host E.coli cells. In contrast, no growth is observed on plating competent cells untransformed with vector DNA as the host E. coli is sensitive to ampicillin. On viewing the ligated plate under UV light, one observes both glowing and non-glowing colonies, indicating ligation of insert to the vector DNA has taken place in either orientation. Hence, about half the number of recombinant colonies would be non-glowing. Control plate will show no glowing colonies as they lack the GFP gene. Bangalore Genei, Bangalore Genei,
10 Appendix: Preparation of LB Agar/broth (1 litre): Dissolve 25 g of media in 800 ml of distilled water. Adjust the ph to 7.0 with 5N NaOH (if necessary) and make up the volume to 1000 ml. Sterilize by autoclaving. For LB agar, add 1.5% agar and autoclave. Ampicillin Preparation: Dissolve 100 mg of ampicillin in 1 ml sterile water to get a stock concentration of 100 mg/ml. Store at 4 C for 2 weeks. Use the antibiotic within this period. For Ampicillin LB media: Add ampicillin to LB broth or agar at a final concentration of 100 µg/ml, when the temperature of the media is around C. Ordering Information: Product Size Cat # 1 Pack Teaching Kit (Consumables for 5 experiments) Sales: [email protected] Customer Support: [email protected] Following aliquots of media are required for GFP cloning experiment: LB Broth 4 x 5 ml; 1 x 100 ml LB Agar 50 ml LB Agar with ampicillin 300 ml Note: Prepare 100 ml of LB broth in a 1 litre conical flask
11 Notes: Bangalore Genei, Bangalore Genei, 2008
TransformAid Bacterial Transformation Kit
 Home Contacts Order Catalog Support Search Alphabetical Index Numerical Index Restriction Endonucleases Modifying Enzymes PCR Kits Markers Nucleic Acids Nucleotides & Oligonucleotides Media Transfection
Home Contacts Order Catalog Support Search Alphabetical Index Numerical Index Restriction Endonucleases Modifying Enzymes PCR Kits Markers Nucleic Acids Nucleotides & Oligonucleotides Media Transfection
Bacterial Transformation with Green Fluorescent Protein. Table of Contents Fall 2012
 Bacterial Transformation with Green Fluorescent Protein pglo Version Table of Contents Bacterial Transformation Introduction..1 Laboratory Exercise...3 Important Laboratory Practices 3 Protocol...... 4
Bacterial Transformation with Green Fluorescent Protein pglo Version Table of Contents Bacterial Transformation Introduction..1 Laboratory Exercise...3 Important Laboratory Practices 3 Protocol...... 4
Lab 10: Bacterial Transformation, part 2, DNA plasmid preps, Determining DNA Concentration and Purity
 Lab 10: Bacterial Transformation, part 2, DNA plasmid preps, Determining DNA Concentration and Purity Today you analyze the results of your bacterial transformation from last week and determine the efficiency
Lab 10: Bacterial Transformation, part 2, DNA plasmid preps, Determining DNA Concentration and Purity Today you analyze the results of your bacterial transformation from last week and determine the efficiency
Transformation Protocol
 To make Glycerol Stocks of Plasmids ** To be done in the hood and use RNase/DNase free tips** 1. In a 10 ml sterile tube add 3 ml autoclaved LB broth and 1.5 ul antibiotic (@ 100 ug/ul) or 3 ul antibiotic
To make Glycerol Stocks of Plasmids ** To be done in the hood and use RNase/DNase free tips** 1. In a 10 ml sterile tube add 3 ml autoclaved LB broth and 1.5 ul antibiotic (@ 100 ug/ul) or 3 ul antibiotic
Transformation of the bacterium E. coli. using a gene for Green Fluorescent Protein
 Transformation of the bacterium E. coli using a gene for Green Fluorescent Protein Background In molecular biology, transformation refers to a form of genetic exchange in which the genetic material carried
Transformation of the bacterium E. coli using a gene for Green Fluorescent Protein Background In molecular biology, transformation refers to a form of genetic exchange in which the genetic material carried
Green Fluorescent Protein (GFP): Genetic Transformation, Synthesis and Purification of the Recombinant Protein
 Green Fluorescent Protein (GFP): Genetic Transformation, Synthesis and Purification of the Recombinant Protein INTRODUCTION Green Fluorescent Protein (GFP) is a novel protein produced by the bioluminescent
Green Fluorescent Protein (GFP): Genetic Transformation, Synthesis and Purification of the Recombinant Protein INTRODUCTION Green Fluorescent Protein (GFP) is a novel protein produced by the bioluminescent
LAB 16 Rapid Colony Transformation of E. coli with Plasmid DNA
 LAB 16 Rapid Colony Transformation of E. coli with Plasmid DNA Objective: In this laboratory investigation, plasmids containing fragments of foreign DNA will be used to transform Escherichia coli cells,
LAB 16 Rapid Colony Transformation of E. coli with Plasmid DNA Objective: In this laboratory investigation, plasmids containing fragments of foreign DNA will be used to transform Escherichia coli cells,
Bacterial Transformation and Plasmid Purification. Chapter 5: Background
 Bacterial Transformation and Plasmid Purification Chapter 5: Background History of Transformation and Plasmids Bacterial methods of DNA transfer Transformation: when bacteria take up DNA from their environment
Bacterial Transformation and Plasmid Purification Chapter 5: Background History of Transformation and Plasmids Bacterial methods of DNA transfer Transformation: when bacteria take up DNA from their environment
GENETIC TRANSFORMATION OF BACTERIA WITH THE GENE FOR GREEN FLUORESCENT PROTEIN (GFP)
 GENETIC TRANSFORMATION OF BACTERIA WITH THE GENE FOR GREEN FLUORESCENT PROTEIN (GFP) LAB BAC3 Adapted from "Biotechnology Explorer pglo Bacterial Transformation Kit Instruction Manual". (Catalog No. 166-0003-EDU)
GENETIC TRANSFORMATION OF BACTERIA WITH THE GENE FOR GREEN FLUORESCENT PROTEIN (GFP) LAB BAC3 Adapted from "Biotechnology Explorer pglo Bacterial Transformation Kit Instruction Manual". (Catalog No. 166-0003-EDU)
CLONING IN ESCHERICHIA COLI
 CLONING IN ESCHERICHIA COLI Introduction: In this laboratory, you will carry out a simple cloning experiment in E. coli. Specifically, you will first create a recombinant DNA molecule by carrying out a
CLONING IN ESCHERICHIA COLI Introduction: In this laboratory, you will carry out a simple cloning experiment in E. coli. Specifically, you will first create a recombinant DNA molecule by carrying out a
Transformation Kit BACTERIAL TRANSFORMATION: GREEN FLUORESCENT PROTEIN. Partnership for Biotechnology and Genomics Education
 Transformation Kit BACTERIAL TRANSFORMATION: GREEN FLUORESCENT PROTEIN Partnership for Biotechnology and Genomics Education Barbara Soots Linda Curro Education Coordinator University of California Davis
Transformation Kit BACTERIAL TRANSFORMATION: GREEN FLUORESCENT PROTEIN Partnership for Biotechnology and Genomics Education Barbara Soots Linda Curro Education Coordinator University of California Davis
GRS Plasmid Purification Kit Transfection Grade GK73.0002 (2 MaxiPreps)
 1 GRS Plasmid Purification Kit Transfection Grade GK73.0002 (2 MaxiPreps) (FOR RESEARCH ONLY) Sample : Expected Yield : Endotoxin: Format : Operation Time : Elution Volume : 50-400 ml of cultured bacterial
1 GRS Plasmid Purification Kit Transfection Grade GK73.0002 (2 MaxiPreps) (FOR RESEARCH ONLY) Sample : Expected Yield : Endotoxin: Format : Operation Time : Elution Volume : 50-400 ml of cultured bacterial
Cloning GFP into Mammalian cells
 Protocol for Cloning GFP into Mammalian cells Studiepraktik 2013 Molecular Biology and Molecular Medicine Aarhus University Produced by the instructors: Tobias Holm Bønnelykke, Rikke Mouridsen, Steffan
Protocol for Cloning GFP into Mammalian cells Studiepraktik 2013 Molecular Biology and Molecular Medicine Aarhus University Produced by the instructors: Tobias Holm Bønnelykke, Rikke Mouridsen, Steffan
LAB 11 PLASMID DNA MINIPREP
 LAB 11 PLASMID DNA MINIPREP STUDENT GUIDE GOAL The objective of this lab is to perform extraction of plasmid DNA and analyze the results. OBJECTIVES After completion, the student should be able to: 1.
LAB 11 PLASMID DNA MINIPREP STUDENT GUIDE GOAL The objective of this lab is to perform extraction of plasmid DNA and analyze the results. OBJECTIVES After completion, the student should be able to: 1.
DNA Isolation Kit for Cells and Tissues
 DNA Isolation Kit for Cells and Tissues for 10 isolations of 00 mg each for tissue or 5 x 10 7 cultured cells Cat. No. 11 81 770 001 Principle Starting material Application Time required Results Benefits
DNA Isolation Kit for Cells and Tissues for 10 isolations of 00 mg each for tissue or 5 x 10 7 cultured cells Cat. No. 11 81 770 001 Principle Starting material Application Time required Results Benefits
restriction enzymes 350 Home R. Ward: Spring 2001
 restriction enzymes 350 Home Restriction Enzymes (endonucleases): molecular scissors that cut DNA Properties of widely used Type II restriction enzymes: recognize a single sequence of bases in dsdna, usually
restriction enzymes 350 Home Restriction Enzymes (endonucleases): molecular scissors that cut DNA Properties of widely used Type II restriction enzymes: recognize a single sequence of bases in dsdna, usually
TIANquick Mini Purification Kit
 TIANquick Mini Purification Kit For purification of PCR products, 100 bp to 20 kb www.tiangen.com TIANquick Mini Purification Kit (Spin column) Cat no. DP203 Kit Contents Contents Buffer BL Buffer PB Buffer
TIANquick Mini Purification Kit For purification of PCR products, 100 bp to 20 kb www.tiangen.com TIANquick Mini Purification Kit (Spin column) Cat no. DP203 Kit Contents Contents Buffer BL Buffer PB Buffer
HiPer RT-PCR Teaching Kit
 HiPer RT-PCR Teaching Kit Product Code: HTBM024 Number of experiments that can be performed: 5 Duration of Experiment: Protocol: 4 hours Agarose Gel Electrophoresis: 45 minutes Storage Instructions: The
HiPer RT-PCR Teaching Kit Product Code: HTBM024 Number of experiments that can be performed: 5 Duration of Experiment: Protocol: 4 hours Agarose Gel Electrophoresis: 45 minutes Storage Instructions: The
HCS604.03 Exercise 1 Dr. Jones Spring 2005. Recombinant DNA (Molecular Cloning) exercise:
 HCS604.03 Exercise 1 Dr. Jones Spring 2005 Recombinant DNA (Molecular Cloning) exercise: The purpose of this exercise is to learn techniques used to create recombinant DNA or clone genes. You will clone
HCS604.03 Exercise 1 Dr. Jones Spring 2005 Recombinant DNA (Molecular Cloning) exercise: The purpose of this exercise is to learn techniques used to create recombinant DNA or clone genes. You will clone
Classic Immunoprecipitation
 292PR 01 G-Biosciences 1-800-628-7730 1-314-991-6034 [email protected] A Geno Technology, Inc. (USA) brand name Classic Immunoprecipitation Utilizes Protein A/G Agarose for Antibody Binding (Cat.
292PR 01 G-Biosciences 1-800-628-7730 1-314-991-6034 [email protected] A Geno Technology, Inc. (USA) brand name Classic Immunoprecipitation Utilizes Protein A/G Agarose for Antibody Binding (Cat.
Agrobacterium tumefaciens-mediated transformation of Colletotrichum graminicola and Colletotrichum sublineolum
 Agrobacterium tumefaciens-mediated transformation of Colletotrichum graminicola and Colletotrichum sublineolum Flowers and Vaillancourt, 2005. Current Genetics 48: 380-388 NOTE added by L. Vaillancourt:
Agrobacterium tumefaciens-mediated transformation of Colletotrichum graminicola and Colletotrichum sublineolum Flowers and Vaillancourt, 2005. Current Genetics 48: 380-388 NOTE added by L. Vaillancourt:
HiPer Ion Exchange Chromatography Teaching Kit
 HiPer Ion Exchange Chromatography Teaching Kit Product Code: HTC001 Number of experiments that can be performed: 5 Duration of Experiment: Protocol: 5-6 hours Storage Instructions: The kit is stable for
HiPer Ion Exchange Chromatography Teaching Kit Product Code: HTC001 Number of experiments that can be performed: 5 Duration of Experiment: Protocol: 5-6 hours Storage Instructions: The kit is stable for
HighPure Maxi Plasmid Kit
 HighPure Maxi Plasmid Kit For purification of high pure plasmid DNA with high yields www.tiangen.com PP120109 HighPure Maxi Plasmid Kit Kit Contents Storage Cat.no. DP116 Contents RNaseA (100 mg/ml) Buffer
HighPure Maxi Plasmid Kit For purification of high pure plasmid DNA with high yields www.tiangen.com PP120109 HighPure Maxi Plasmid Kit Kit Contents Storage Cat.no. DP116 Contents RNaseA (100 mg/ml) Buffer
LAB 7 DNA RESTRICTION for CLONING
 BIOTECHNOLOGY I DNA RESTRICTION FOR CLONING LAB 7 DNA RESTRICTION for CLONING STUDENT GUIDE GOALS The goals of this lab are to provide the biotech student with experience in DNA digestion with restriction
BIOTECHNOLOGY I DNA RESTRICTION FOR CLONING LAB 7 DNA RESTRICTION for CLONING STUDENT GUIDE GOALS The goals of this lab are to provide the biotech student with experience in DNA digestion with restriction
DNA CAN BE TRANSFERRED BETWEEN BACTERIA GENETIC ENGINEERING USING RECOMBINANT DNA TECHNOLOGY
 Bacterial Transformation DNA CAN BE TRANSFERRED BETWEEN BACTERIA Background Information Plasmid Transformed Cell Figure 1: Bacterial Transformation Quick Reference Abbreviations GFP pgfp gfp Green fl uorescent
Bacterial Transformation DNA CAN BE TRANSFERRED BETWEEN BACTERIA Background Information Plasmid Transformed Cell Figure 1: Bacterial Transformation Quick Reference Abbreviations GFP pgfp gfp Green fl uorescent
One Shot TOP10 Competent Cells
 USER GUIDE One Shot TOP10 Competent Cells Catalog Numbers C4040-10, C4040-03, C4040-06, C4040-50, and C4040-52 Document Part Number 280126 Publication Number MAN0000633 Revision A.0 For Research Use Only.
USER GUIDE One Shot TOP10 Competent Cells Catalog Numbers C4040-10, C4040-03, C4040-06, C4040-50, and C4040-52 Document Part Number 280126 Publication Number MAN0000633 Revision A.0 For Research Use Only.
Frozen-EZ Yeast Transformation II Catalog No. T2001
 INSTRUCTIONS Frozen-EZ Yeast Transformation II Catalog No. T2001 Highlights High transformation efficiency that yields approximately 10 5-10 6 transformants per μg plasmid DNA (circular). Frozen storage
INSTRUCTIONS Frozen-EZ Yeast Transformation II Catalog No. T2001 Highlights High transformation efficiency that yields approximately 10 5-10 6 transformants per μg plasmid DNA (circular). Frozen storage
LAB 10 DNA TRANSFORMATION
 LAB 10 DNA TRANSFORMATION STUDENT GUIDE GOAL The objective of this lab is to successfully perform DNA transformation of a recombinant plasmid and use blue-white selection to select recombinant clones.
LAB 10 DNA TRANSFORMATION STUDENT GUIDE GOAL The objective of this lab is to successfully perform DNA transformation of a recombinant plasmid and use blue-white selection to select recombinant clones.
Student Manual. pglo Transformation
 Student Manual pglo Transformation Lesson 1 Introduction to Transformation In this lab you will perform a procedure known as genetic transformation. Remember that a gene is a piece of DNA which provides
Student Manual pglo Transformation Lesson 1 Introduction to Transformation In this lab you will perform a procedure known as genetic transformation. Remember that a gene is a piece of DNA which provides
Transformation of E.coli with pgal
 The Biotechnology Education Company ATTENTION! This experiment includes either BactoBeads or LyphoCells. If you have received LyphoCells, please refer to the addendum posted on the last page of this literature.
The Biotechnology Education Company ATTENTION! This experiment includes either BactoBeads or LyphoCells. If you have received LyphoCells, please refer to the addendum posted on the last page of this literature.
CHAPTER 6: RECOMBINANT DNA TECHNOLOGY YEAR III PHARM.D DR. V. CHITRA
 CHAPTER 6: RECOMBINANT DNA TECHNOLOGY YEAR III PHARM.D DR. V. CHITRA INTRODUCTION DNA : DNA is deoxyribose nucleic acid. It is made up of a base consisting of sugar, phosphate and one nitrogen base.the
CHAPTER 6: RECOMBINANT DNA TECHNOLOGY YEAR III PHARM.D DR. V. CHITRA INTRODUCTION DNA : DNA is deoxyribose nucleic acid. It is made up of a base consisting of sugar, phosphate and one nitrogen base.the
The fastest spin-column based procedure for purifying up to 10 mg of ultra-pure endotoxin-free transfection-grade plasmid DNA.
 INSTRUCTION MANUAL ZymoPURE Plasmid Gigaprep Kit Catalog Nos. D4204 (Patent Pending) Highlights The fastest spin-column based procedure for purifying up to 10 mg of ultra-pure endotoxin-free transfection-grade
INSTRUCTION MANUAL ZymoPURE Plasmid Gigaprep Kit Catalog Nos. D4204 (Patent Pending) Highlights The fastest spin-column based procedure for purifying up to 10 mg of ultra-pure endotoxin-free transfection-grade
HiPer Total RNA Extraction Teaching Kit
 HiPer Total RNA Extraction Teaching Kit Product Code: HTBM012 Number of experiments that can be performed: 10 Duration of Experiment Protocol: 1 hour Agarose Gel Electrophoresis: 1 hour Storage Instructions:
HiPer Total RNA Extraction Teaching Kit Product Code: HTBM012 Number of experiments that can be performed: 10 Duration of Experiment Protocol: 1 hour Agarose Gel Electrophoresis: 1 hour Storage Instructions:
Recombinant DNA Technology
 Recombinant DNA Technology Dates in the Development of Gene Cloning: 1965 - plasmids 1967 - ligase 1970 - restriction endonucleases 1972 - first experiments in gene splicing 1974 - worldwide moratorium
Recombinant DNA Technology Dates in the Development of Gene Cloning: 1965 - plasmids 1967 - ligase 1970 - restriction endonucleases 1972 - first experiments in gene splicing 1974 - worldwide moratorium
Application Guide... 2
 Protocol for GenomePlex Whole Genome Amplification from Formalin-Fixed Parrafin-Embedded (FFPE) tissue Application Guide... 2 I. Description... 2 II. Product Components... 2 III. Materials to be Supplied
Protocol for GenomePlex Whole Genome Amplification from Formalin-Fixed Parrafin-Embedded (FFPE) tissue Application Guide... 2 I. Description... 2 II. Product Components... 2 III. Materials to be Supplied
Biotechnology Explorer
 arac ori pglo bla GFP Biotechnology Explorer pglo Bacterial Transformation Kit Catalog Number 166-0003EDU explorer.bio-rad.com Store components of this kit at room temperature. Duplication of any part
arac ori pglo bla GFP Biotechnology Explorer pglo Bacterial Transformation Kit Catalog Number 166-0003EDU explorer.bio-rad.com Store components of this kit at room temperature. Duplication of any part
Investigating a Eukaryotic Genome: Cloning and Sequencing a Fragment of Yeast DNA
 Investigating a Eukaryotic Genome: Cloning and Sequencing a Fragment of Yeast DNA Credits: This lab was created by Sarah C.R. Elgin and developed and written by Kathleen Weston-Hafer. Specific protocols
Investigating a Eukaryotic Genome: Cloning and Sequencing a Fragment of Yeast DNA Credits: This lab was created by Sarah C.R. Elgin and developed and written by Kathleen Weston-Hafer. Specific protocols
Recombinant DNA & Genetic Engineering. Tools for Genetic Manipulation
 Recombinant DNA & Genetic Engineering g Genetic Manipulation: Tools Kathleen Hill Associate Professor Department of Biology The University of Western Ontario Tools for Genetic Manipulation DNA, RNA, cdna
Recombinant DNA & Genetic Engineering g Genetic Manipulation: Tools Kathleen Hill Associate Professor Department of Biology The University of Western Ontario Tools for Genetic Manipulation DNA, RNA, cdna
BacReady TM Multiplex PCR System
 BacReady TM Multiplex PCR System Technical Manual No. 0191 Version 10112010 I Description.. 1 II Applications 2 III Key Features.. 2 IV Shipping and Storage. 2 V Simplified Procedures. 2 VI Detailed Experimental
BacReady TM Multiplex PCR System Technical Manual No. 0191 Version 10112010 I Description.. 1 II Applications 2 III Key Features.. 2 IV Shipping and Storage. 2 V Simplified Procedures. 2 VI Detailed Experimental
Plant Genomic DNA Extraction using CTAB
 Plant Genomic DNA Extraction using CTAB Introduction The search for a more efficient means of extracting DNA of both higher quality and yield has lead to the development of a variety of protocols, however
Plant Genomic DNA Extraction using CTAB Introduction The search for a more efficient means of extracting DNA of both higher quality and yield has lead to the development of a variety of protocols, however
Cloning and Transformation
 Cloning and Transformation 2 Exp. 2.1 Preparation of Competent Cells and Heat-Shock Transformation Objective To prepare competent cells of bacteria by chemical treatment and transformation by heat shock.
Cloning and Transformation 2 Exp. 2.1 Preparation of Competent Cells and Heat-Shock Transformation Objective To prepare competent cells of bacteria by chemical treatment and transformation by heat shock.
Molecular Biology Techniques: A Classroom Laboratory Manual THIRD EDITION
 Molecular Biology Techniques: A Classroom Laboratory Manual THIRD EDITION Susan Carson Heather B. Miller D.Scott Witherow ELSEVIER AMSTERDAM BOSTON HEIDELBERG LONDON NEW YORK OXFORD PARIS SAN DIEGO SAN
Molecular Biology Techniques: A Classroom Laboratory Manual THIRD EDITION Susan Carson Heather B. Miller D.Scott Witherow ELSEVIER AMSTERDAM BOSTON HEIDELBERG LONDON NEW YORK OXFORD PARIS SAN DIEGO SAN
Purification of Plasmid DNA
 Purification of Plasmid DNA Introduction: The growth of colonies on antibiotic medium provides phenotypic evidence that cells have been transformed. To confirm this at the genotypic level, plasmid DNA
Purification of Plasmid DNA Introduction: The growth of colonies on antibiotic medium provides phenotypic evidence that cells have been transformed. To confirm this at the genotypic level, plasmid DNA
Genomic DNA Extraction Kit INSTRUCTION MANUAL
 Genomic DNA Extraction Kit INSTRUCTION MANUAL Table of Contents Introduction 3 Kit Components 3 Storage Conditions 4 Recommended Equipment and Reagents 4 Introduction to the Protocol 4 General Overview
Genomic DNA Extraction Kit INSTRUCTION MANUAL Table of Contents Introduction 3 Kit Components 3 Storage Conditions 4 Recommended Equipment and Reagents 4 Introduction to the Protocol 4 General Overview
RNA Extraction and Quantification, Reverse Transcription, and Real-time PCR (q-pcr)
 RNA Extraction and Quantification, Reverse Transcription, and Real-time Preparation of Samples Cells: o Remove media and wash cells 2X with cold PBS. (2 ml for 6 well plate or 3 ml for 6cm plate) Keep
RNA Extraction and Quantification, Reverse Transcription, and Real-time Preparation of Samples Cells: o Remove media and wash cells 2X with cold PBS. (2 ml for 6 well plate or 3 ml for 6cm plate) Keep
First Strand cdna Synthesis
 380PR 01 G-Biosciences 1-800-628-7730 1-314-991-6034 [email protected] A Geno Technology, Inc. (USA) brand name First Strand cdna Synthesis (Cat. # 786 812) think proteins! think G-Biosciences
380PR 01 G-Biosciences 1-800-628-7730 1-314-991-6034 [email protected] A Geno Technology, Inc. (USA) brand name First Strand cdna Synthesis (Cat. # 786 812) think proteins! think G-Biosciences
Chromatin Immunoprecipitation
 Chromatin Immunoprecipitation A) Prepare a yeast culture (see the Galactose Induction Protocol for details). 1) Start a small culture (e.g. 2 ml) in YEPD or selective media from a single colony. 2) Spin
Chromatin Immunoprecipitation A) Prepare a yeast culture (see the Galactose Induction Protocol for details). 1) Start a small culture (e.g. 2 ml) in YEPD or selective media from a single colony. 2) Spin
Wizard DNA Clean-Up System INSTRUCTIONS FOR USE OF PRODUCT A7280.
 Technical Bulletin Wizard DNA Clean-Up System INSTRUCTIONS FOR USE OF PRODUCT A7280. PRINTED IN USA. Revised 4/06 AF9TB141 0406TB141 Wizard DNA Clean-Up System All technical literature is available on
Technical Bulletin Wizard DNA Clean-Up System INSTRUCTIONS FOR USE OF PRODUCT A7280. PRINTED IN USA. Revised 4/06 AF9TB141 0406TB141 Wizard DNA Clean-Up System All technical literature is available on
Thermo Scientific Phusion Site-Directed Mutagenesis Kit #F-541
 PRODUCT INFORMATION Thermo Scientific Phusion Site-Directed Mutagenesis Kit #F-541 Lot _ Store at -20 C Expiry Date _ www.thermoscientific.com/onebio CERTIFICATE OF ANALYSIS The Phusion Site-Directed Mutagenesis
PRODUCT INFORMATION Thermo Scientific Phusion Site-Directed Mutagenesis Kit #F-541 Lot _ Store at -20 C Expiry Date _ www.thermoscientific.com/onebio CERTIFICATE OF ANALYSIS The Phusion Site-Directed Mutagenesis
How To Shear Chromatin
 truchip Low Cell Chromatin Shearing Kit with Non-ionic Shearing Buffer Part Number: 010144 Rev C 1 Page INTRODUCTION The truchip Low Cell Chromatin Shearing Kit with Non-ionic Shearing Buffer (PN 520084)
truchip Low Cell Chromatin Shearing Kit with Non-ionic Shearing Buffer Part Number: 010144 Rev C 1 Page INTRODUCTION The truchip Low Cell Chromatin Shearing Kit with Non-ionic Shearing Buffer (PN 520084)
NimbleGen DNA Methylation Microarrays and Services
 NimbleGen DNA Methylation Microarrays and Services Sample Preparation Instructions Outline This protocol describes the process for preparing samples for NimbleGen DNA Methylation microarrays using the
NimbleGen DNA Methylation Microarrays and Services Sample Preparation Instructions Outline This protocol describes the process for preparing samples for NimbleGen DNA Methylation microarrays using the
Biotechnology and Recombinant DNA (Chapter 9) Lecture Materials for Amy Warenda Czura, Ph.D. Suffolk County Community College
 Biotechnology and Recombinant DNA (Chapter 9) Lecture Materials for Amy Warenda Czura, Ph.D. Suffolk County Community College Primary Source for figures and content: Eastern Campus Tortora, G.J. Microbiology
Biotechnology and Recombinant DNA (Chapter 9) Lecture Materials for Amy Warenda Czura, Ph.D. Suffolk County Community College Primary Source for figures and content: Eastern Campus Tortora, G.J. Microbiology
50 g 650 L. *Average yields will vary depending upon a number of factors including type of phage, growth conditions used and developmental stage.
 3430 Schmon Parkway Thorold, ON, Canada L2V 4Y6 Phone: 866-667-4362 (905) 227-8848 Fax: (905) 227-1061 Email: [email protected] Phage DNA Isolation Kit Product # 46800, 46850 Product Insert
3430 Schmon Parkway Thorold, ON, Canada L2V 4Y6 Phone: 866-667-4362 (905) 227-8848 Fax: (905) 227-1061 Email: [email protected] Phage DNA Isolation Kit Product # 46800, 46850 Product Insert
Enzymes: Amylase Activity in Starch-degrading Soil Isolates
 Enzymes: Amylase Activity in Starch-degrading Soil Isolates Introduction This week you will continue our theme of industrial microbiologist by characterizing the enzyme activity we selected for (starch
Enzymes: Amylase Activity in Starch-degrading Soil Isolates Introduction This week you will continue our theme of industrial microbiologist by characterizing the enzyme activity we selected for (starch
Genolution Pharmaceuticals, Inc. Life Science and Molecular Diagnostic Products
 Genolution Pharmaceuticals, Inc. Revolution through genes, And Solution through genes. Life Science and Molecular Diagnostic Products www.genolution1.com TEL; 02-3010-8670, 8672 Geno-Serum Hepatitis B
Genolution Pharmaceuticals, Inc. Revolution through genes, And Solution through genes. Life Science and Molecular Diagnostic Products www.genolution1.com TEL; 02-3010-8670, 8672 Geno-Serum Hepatitis B
DNA ligase. ATP (or NAD+)
 DNA Ligase enzyme catalysing formation of phosphodiesteric bound between group 3 -OH of one end of DNA molecule and group 5 -phosphate of the second end of DNA DNA ligase ATP (or NAD+) Ligase cofactors
DNA Ligase enzyme catalysing formation of phosphodiesteric bound between group 3 -OH of one end of DNA molecule and group 5 -phosphate of the second end of DNA DNA ligase ATP (or NAD+) Ligase cofactors
SOLIDscript Solid Phase cdna Synthesis Kit Instruction Manual
 Toll Free: 866-252-7771 752A Lincoln Blvd. Phone: 732-469-7771 Fax: 732-469-7782 Middlesex, NJ 08846 Web: www.purebiotechllc.com SOLIDscript Solid Phase cdna Synthesis Kit Instruction Manual Product: SOLIDscript
Toll Free: 866-252-7771 752A Lincoln Blvd. Phone: 732-469-7771 Fax: 732-469-7782 Middlesex, NJ 08846 Web: www.purebiotechllc.com SOLIDscript Solid Phase cdna Synthesis Kit Instruction Manual Product: SOLIDscript
ELUTION OF DNA FROM AGAROSE GELS
 ELUTION OF DNA FROM AGAROSE GELS OBTECTIVE: To isolate specific bands or regions of agarose-separated DNA for use in subsequent experiments and/or procedures. INTRODUCTION: It is sometimes necessary to
ELUTION OF DNA FROM AGAROSE GELS OBTECTIVE: To isolate specific bands or regions of agarose-separated DNA for use in subsequent experiments and/or procedures. INTRODUCTION: It is sometimes necessary to
Effects of Antibiotics on Bacterial Growth and Protein Synthesis: Student Laboratory Manual
 Effects of Antibiotics on Bacterial Growth and Protein Synthesis: Student Laboratory Manual I. Purpose...1 II. Introduction...1 III. Inhibition of Bacterial Growth Protocol...2 IV. Inhibition of in vitro
Effects of Antibiotics on Bacterial Growth and Protein Synthesis: Student Laboratory Manual I. Purpose...1 II. Introduction...1 III. Inhibition of Bacterial Growth Protocol...2 IV. Inhibition of in vitro
UltraClean Soil DNA Isolation Kit
 PAGE 1 UltraClean Soil DNA Isolation Kit Catalog # 12800-50 50 preps New improved PCR inhibitor removal solution (IRS) included Instruction Manual (New Alternative Protocol maximizes yields) Introduction
PAGE 1 UltraClean Soil DNA Isolation Kit Catalog # 12800-50 50 preps New improved PCR inhibitor removal solution (IRS) included Instruction Manual (New Alternative Protocol maximizes yields) Introduction
CHEF Genomic DNA Plug Kits Instruction Manual
 CHEF Genomic DNA Plug Kits Instruction Manual Catalog Numbers 170-3591 170-3592 170-3593 For Technical Service Call Your Local Bio-Rad Office or in the U.S. Call 1-800-4BIORAD (1-800-424-6723) Table of
CHEF Genomic DNA Plug Kits Instruction Manual Catalog Numbers 170-3591 170-3592 170-3593 For Technical Service Call Your Local Bio-Rad Office or in the U.S. Call 1-800-4BIORAD (1-800-424-6723) Table of
Introduction to cloning
 1 of 14 Introduction to cloning Aim The aim of this protocol is to serve as a general guideline to mainstream molecular cloning of Gene of Interest ( GOI ). Overview GOI Sequence Transformation into Bacteria
1 of 14 Introduction to cloning Aim The aim of this protocol is to serve as a general guideline to mainstream molecular cloning of Gene of Interest ( GOI ). Overview GOI Sequence Transformation into Bacteria
Recombinant DNA and Biotechnology
 Recombinant DNA and Biotechnology Chapter 18 Lecture Objectives What Is Recombinant DNA? How Are New Genes Inserted into Cells? What Sources of DNA Are Used in Cloning? What Other Tools Are Used to Study
Recombinant DNA and Biotechnology Chapter 18 Lecture Objectives What Is Recombinant DNA? How Are New Genes Inserted into Cells? What Sources of DNA Are Used in Cloning? What Other Tools Are Used to Study
Biology 29 Cell Structure and Function Spring, 2009 Springer LABORATORY 2:CHLOROPLASTS AND PHOTOREDUCTION
 Biology 29 Cell Structure and Function Spring, 2009 Springer LABORATORY 2:CHLOROPLASTS AND PHOTOREDUCTION In this laboratory we will purify chloroplasts from spinach by differential centrifugation, then
Biology 29 Cell Structure and Function Spring, 2009 Springer LABORATORY 2:CHLOROPLASTS AND PHOTOREDUCTION In this laboratory we will purify chloroplasts from spinach by differential centrifugation, then
MEF Nucleofector Kit 1 and 2
 page 1 of 7 MEF Nucleofector Kit 1 and 2 for Mouse Embryonic Fibroblasts (MEF) MEF display significant phenotypic variations which depend on the strain, the genetic background of the they are isolated
page 1 of 7 MEF Nucleofector Kit 1 and 2 for Mouse Embryonic Fibroblasts (MEF) MEF display significant phenotypic variations which depend on the strain, the genetic background of the they are isolated
Cloning Blunt-End Pfu DNA Polymerase- Generated PCR Fragments into pgem -T Vector Systems
 Promega Notes Number 71, 1999, p. 10 Blunt-End Pfu DNA Polymerase- Generated PCR Fragments into pgem -T Vector Systems By Kimberly Knoche, Ph.D., and Dan Kephart, Ph.D. Promega Corporation Corresponding
Promega Notes Number 71, 1999, p. 10 Blunt-End Pfu DNA Polymerase- Generated PCR Fragments into pgem -T Vector Systems By Kimberly Knoche, Ph.D., and Dan Kephart, Ph.D. Promega Corporation Corresponding
How To Make A Tri Reagent
 TRI Reagent For processing tissues, cells cultured in monolayer or cell pellets Catalog Number T9424 Store at room temperature. TECHNICAL BULLETIN Product Description TRI Reagent is a quick and convenient
TRI Reagent For processing tissues, cells cultured in monolayer or cell pellets Catalog Number T9424 Store at room temperature. TECHNICAL BULLETIN Product Description TRI Reagent is a quick and convenient
pmod2-puro A plasmid containing a synthetic Puromycin resistance gene Catalog # pmod2-puro For research use only Version # 11H29-MM
 pmod2-puro A plasmid containing a synthetic Puromycin resistance gene Catalog # pmod2-puro For research use only Version # 11H29-MM PrOduct information content: - 20 mg of lyophilized pmod2-puro plasmid
pmod2-puro A plasmid containing a synthetic Puromycin resistance gene Catalog # pmod2-puro For research use only Version # 11H29-MM PrOduct information content: - 20 mg of lyophilized pmod2-puro plasmid
GROWING BACTERIA INTRODUCTION
 GROWING BACTERIA INTRODUCTION E. coli is a normal part of the bacterial flora of the human gut. It is not generally considered pathogenic, although some strains are highly toxic (recent food poisonings
GROWING BACTERIA INTRODUCTION E. coli is a normal part of the bacterial flora of the human gut. It is not generally considered pathogenic, although some strains are highly toxic (recent food poisonings
DNA Scissors: Introduction to Restriction Enzymes
 DNA Scissors: Introduction to Restriction Enzymes Objectives At the end of this activity, students should be able to 1. Describe a typical restriction site as a 4- or 6-base- pair palindrome; 2. Describe
DNA Scissors: Introduction to Restriction Enzymes Objectives At the end of this activity, students should be able to 1. Describe a typical restriction site as a 4- or 6-base- pair palindrome; 2. Describe
Biotechnology: DNA Technology & Genomics
 Chapter 20. Biotechnology: DNA Technology & Genomics 2003-2004 The BIG Questions How can we use our knowledge of DNA to: diagnose disease or defect? cure disease or defect? change/improve organisms? What
Chapter 20. Biotechnology: DNA Technology & Genomics 2003-2004 The BIG Questions How can we use our knowledge of DNA to: diagnose disease or defect? cure disease or defect? change/improve organisms? What
Transfection-Transfer of non-viral genetic material into eukaryotic cells. Infection/ Transduction- Transfer of viral genetic material into cells.
 Transfection Key words: Transient transfection, Stable transfection, transfection methods, vector, plasmid, origin of replication, reporter gene/ protein, cloning site, promoter and enhancer, signal peptide,
Transfection Key words: Transient transfection, Stable transfection, transfection methods, vector, plasmid, origin of replication, reporter gene/ protein, cloning site, promoter and enhancer, signal peptide,
RevertAid Premium First Strand cdna Synthesis Kit
 RevertAid Premium First Strand cdna Synthesis Kit #K1651, #K1652 CERTIFICATE OF ANALYSIS #K1651 Lot QUALITY CONTROL RT-PCR using 100 fg of control GAPDH RNA and GAPDH control primers generated a prominent
RevertAid Premium First Strand cdna Synthesis Kit #K1651, #K1652 CERTIFICATE OF ANALYSIS #K1651 Lot QUALITY CONTROL RT-PCR using 100 fg of control GAPDH RNA and GAPDH control primers generated a prominent
Lab Exercise 3: Media, incubation, and aseptic technique
 Lab Exercise 3: Media, incubation, and aseptic technique Objectives 1. Compare the different types of media. 2. Describe the different formats of media, plate, tube etc. 3. Explain how to sterilize it,
Lab Exercise 3: Media, incubation, and aseptic technique Objectives 1. Compare the different types of media. 2. Describe the different formats of media, plate, tube etc. 3. Explain how to sterilize it,
Isolation and Purification of Total Genomic DNA from Gram-Negative Bacteria
 Isolation and Purification of Total Genomic DNA from Gram-Negative Bacteria INTRODUCTION The isolation and purification of DNA from cells is one of the most common procedures in contemporary molecular
Isolation and Purification of Total Genomic DNA from Gram-Negative Bacteria INTRODUCTION The isolation and purification of DNA from cells is one of the most common procedures in contemporary molecular
Optimized Protocol sirna Test Kit for Cell Lines and Adherent Primary Cells
 page 1 of 8 sirna Test Kit for Cell Lines and Adherent Primary Cells Kit principle Co-transfection of pmaxgfp, encoding the green fluorescent protein (GFP) from Pontellina p. with an sirna directed against
page 1 of 8 sirna Test Kit for Cell Lines and Adherent Primary Cells Kit principle Co-transfection of pmaxgfp, encoding the green fluorescent protein (GFP) from Pontellina p. with an sirna directed against
Biotechnology Explorer
 arac ori pglo bla GFP Biotechnology Explorer pglo Bacterial Transformation Kit Catalog #166-0003EDU explorer.bio-rad.com See individual components for storage temperature. Duplication of any part of this
arac ori pglo bla GFP Biotechnology Explorer pglo Bacterial Transformation Kit Catalog #166-0003EDU explorer.bio-rad.com See individual components for storage temperature. Duplication of any part of this
Determine Osmolarity Activity Using RFP under PompC
 Determine Osmolarity Activity Using RFP under PompC Materials and Equipment: E. coli : o containing RFP under Plac o (blank) Isogenic strains containing mrfp under PompC: o 21153 o 3367-3 ΔenvZ Isogenic
Determine Osmolarity Activity Using RFP under PompC Materials and Equipment: E. coli : o containing RFP under Plac o (blank) Isogenic strains containing mrfp under PompC: o 21153 o 3367-3 ΔenvZ Isogenic
Genetic Engineering and Biotechnology
 1 So, what is biotechnology?? The use of living organisms to carry out defined chemical processes for industrial or commercial application. The office of Technology Assessment of the U.S. Congress defines
1 So, what is biotechnology?? The use of living organisms to carry out defined chemical processes for industrial or commercial application. The office of Technology Assessment of the U.S. Congress defines
Procedure for RNA isolation from human muscle or fat
 Procedure for RNA isolation from human muscle or fat Reagents, all Rnase free: 20% SDS DEPC-H2O Rnase ZAP 75% EtOH Trizol Chloroform Isopropanol 0.8M NaCitrate/1.2M NaCl TE buffer, ph 7.0 1. Homogenizer-probe
Procedure for RNA isolation from human muscle or fat Reagents, all Rnase free: 20% SDS DEPC-H2O Rnase ZAP 75% EtOH Trizol Chloroform Isopropanol 0.8M NaCitrate/1.2M NaCl TE buffer, ph 7.0 1. Homogenizer-probe
Microbiological Testing of the Sawyer Mini Filter. 16 December 2013. Summary
 Microbiological Testing of the Sawyer Mini Filter 16 December 2013 Summary The Sawyer Mini Filter was tested for its ability to remove three microorganisms Raoultella terrigena, Bacillus subtilis, and
Microbiological Testing of the Sawyer Mini Filter 16 December 2013 Summary The Sawyer Mini Filter was tested for its ability to remove three microorganisms Raoultella terrigena, Bacillus subtilis, and
RT-PCR: Two-Step Protocol
 RT-PCR: Two-Step Protocol We will provide both one-step and two-step protocols for RT-PCR. We recommend the twostep protocol for this class. In the one-step protocol, the components of RT and PCR are mixed
RT-PCR: Two-Step Protocol We will provide both one-step and two-step protocols for RT-PCR. We recommend the twostep protocol for this class. In the one-step protocol, the components of RT and PCR are mixed
BaculoDirect Baculovirus Expression System Free your hands with the BaculoDirect Baculovirus Expression System
 BaculoDirect Baculovirus Expression System Free your hands with the BaculoDirect Baculovirus Expression System The BaculoDirect Baculovirus Expression System gives you: Unique speed and simplicity High-throughput
BaculoDirect Baculovirus Expression System Free your hands with the BaculoDirect Baculovirus Expression System The BaculoDirect Baculovirus Expression System gives you: Unique speed and simplicity High-throughput
ExpressArt Bacterial H-TR cdna synthesis kit. With extreme selectivity against rrnas
 ExpressArt Bacterial H-TR cdna synthesis kit With extreme selectivity against rrnas suitable for 2 to 4 µg total RNA Catalogue No. 8004-A30 (30 rxns) Reagents Materials are provided for 30 cdna synthesis
ExpressArt Bacterial H-TR cdna synthesis kit With extreme selectivity against rrnas suitable for 2 to 4 µg total RNA Catalogue No. 8004-A30 (30 rxns) Reagents Materials are provided for 30 cdna synthesis
DNA SPOOLING 1 ISOLATION OF DNA FROM ONION
 DNA SPOOLING 1 ISOLATION OF DNA FROM ONION INTRODUCTION This laboratory protocol will demonstrate several basic steps required for isolation of chromosomal DNA from cells. To extract the chromosomal DNA,
DNA SPOOLING 1 ISOLATION OF DNA FROM ONION INTRODUCTION This laboratory protocol will demonstrate several basic steps required for isolation of chromosomal DNA from cells. To extract the chromosomal DNA,
Microbiology BIOL 275 DILUTIONS
 DILUTIONS Occasionally a solution is too concentrated to be used as is. For example, when one is performing manual blood counts, the blood contains too many cells to be counted as such. Or when performing
DILUTIONS Occasionally a solution is too concentrated to be used as is. For example, when one is performing manual blood counts, the blood contains too many cells to be counted as such. Or when performing
NIH Mammalian Gene Collection (MGC)
 USER GUIDE NIH Mammalian Gene Collection (MGC) Catalog number FL1002 Revision date 28 November 2011 Publication Part number 25-0610 MAN0000351 For Research Use Only. Not for diagnostic procedures. ii Table
USER GUIDE NIH Mammalian Gene Collection (MGC) Catalog number FL1002 Revision date 28 November 2011 Publication Part number 25-0610 MAN0000351 For Research Use Only. Not for diagnostic procedures. ii Table
1) Vector Preparation sg 1371 w/blp1 Ef1α puro t29 BFP (602 ng/ul)
 1) Vector Preparation sg 1371 w/blp1 Ef1α puro t29 BFP (602 ng/ul) a) Digest 5 ug of vector with Thermo Scientific FastDigest BstX1 and Blp1 for 1h at 37ºC. Set up reaction as follows: 100 ul Reaction
1) Vector Preparation sg 1371 w/blp1 Ef1α puro t29 BFP (602 ng/ul) a) Digest 5 ug of vector with Thermo Scientific FastDigest BstX1 and Blp1 for 1h at 37ºC. Set up reaction as follows: 100 ul Reaction
QUANTITATIVE RT-PCR. A = B (1+e) n. A=amplified products, B=input templates, n=cycle number, and e=amplification efficiency.
 QUANTITATIVE RT-PCR Application: Quantitative RT-PCR is used to quantify mrna in both relative and absolute terms. It can be applied for the quantification of mrna expressed from endogenous genes, and
QUANTITATIVE RT-PCR Application: Quantitative RT-PCR is used to quantify mrna in both relative and absolute terms. It can be applied for the quantification of mrna expressed from endogenous genes, and
SAMPLE. Bacterial Transformation. Lab 8 BACKGROUND INFORMATION. Neo/SCI Student s Guide Name... Teacher/Section...
 1431489 REV 001 Neo/SCI Lab 8 Bacterial Transformation BACKGROUND INFORMATION What Is Biotechnology? Before you start doing biotechnology laboratory exercises, it is important to know exactly what biotechnology
1431489 REV 001 Neo/SCI Lab 8 Bacterial Transformation BACKGROUND INFORMATION What Is Biotechnology? Before you start doing biotechnology laboratory exercises, it is important to know exactly what biotechnology
A and B are not absolutely linked. They could be far enough apart on the chromosome that they assort independently.
 Name Section 7.014 Problem Set 5 Please print out this problem set and record your answers on the printed copy. Answers to this problem set are to be turned in to the box outside 68-120 by 5:00pm on Friday
Name Section 7.014 Problem Set 5 Please print out this problem set and record your answers on the printed copy. Answers to this problem set are to be turned in to the box outside 68-120 by 5:00pm on Friday
The E. coli Insulin Factory
 The E. coli Insulin Factory BACKGROUND Bacteria have not only their normal DNA, they also have pieces of circular DNA called plasmids. Plasmids are a wonderfully ally for biologists who desire to get bacteria
The E. coli Insulin Factory BACKGROUND Bacteria have not only their normal DNA, they also have pieces of circular DNA called plasmids. Plasmids are a wonderfully ally for biologists who desire to get bacteria
FOR REFERENCE PURPOSES
 BIOO LIFE SCIENCE PRODUCTS FOR REFERENCE PURPOSES This manual is for Reference Purposes Only. DO NOT use this protocol to run your assays. Periodically, optimizations and revisions are made to the kit
BIOO LIFE SCIENCE PRODUCTS FOR REFERENCE PURPOSES This manual is for Reference Purposes Only. DO NOT use this protocol to run your assays. Periodically, optimizations and revisions are made to the kit
2.1.2 Characterization of antiviral effect of cytokine expression on HBV replication in transduced mouse hepatocytes line
 i 1 INTRODUCTION 1.1 Human Hepatitis B virus (HBV) 1 1.1.1 Pathogenesis of Hepatitis B 1 1.1.2 Genome organization of HBV 3 1.1.3 Structure of HBV virion 5 1.1.4 HBV life cycle 5 1.1.5 Experimental models
i 1 INTRODUCTION 1.1 Human Hepatitis B virus (HBV) 1 1.1.1 Pathogenesis of Hepatitis B 1 1.1.2 Genome organization of HBV 3 1.1.3 Structure of HBV virion 5 1.1.4 HBV life cycle 5 1.1.5 Experimental models
Creatine Kinase Activity Colorimetric Assay Kit ABE5487 100 assays; Store at -20 C
 Creatine Kinase Activity Colorimetric Assay Kit ABE5487 100 assays; Store at -20 C I. Introduction: Creatine Kinase (CK) also known as creatine phosphokinase (CPK) and ATP: creatine N- phosphotransferase
Creatine Kinase Activity Colorimetric Assay Kit ABE5487 100 assays; Store at -20 C I. Introduction: Creatine Kinase (CK) also known as creatine phosphokinase (CPK) and ATP: creatine N- phosphotransferase
Mouse ES Cell Nucleofector Kit
 page 1 of 7 Mouse ES Cell Nucleofector Kit for Mouse Embryonic Stem Cells Cell type Origin Cells derived from mouse blastocysts. Morphology Round cells growing in clumps. Important remarks! 1. This protocol
page 1 of 7 Mouse ES Cell Nucleofector Kit for Mouse Embryonic Stem Cells Cell type Origin Cells derived from mouse blastocysts. Morphology Round cells growing in clumps. Important remarks! 1. This protocol
TransIT -2020 Transfection Reagent
 Quick Reference Protocol, MSDS and Certificate of Analysis available at mirusbio.com/5400 INTRODUCTION TransIT -2020 Transfection Reagent is a high-performance, animal-origin free, broad spectrum reagent
Quick Reference Protocol, MSDS and Certificate of Analysis available at mirusbio.com/5400 INTRODUCTION TransIT -2020 Transfection Reagent is a high-performance, animal-origin free, broad spectrum reagent
MEF Starter Nucleofector Kit
 page 1 of 7 MEF Starter Nucleofector Kit for Mouse Embryonic Fibroblasts (MEF) MEF display significant phenotypic variations which depend on the strain, the genetic background of the mice they are isolated
page 1 of 7 MEF Starter Nucleofector Kit for Mouse Embryonic Fibroblasts (MEF) MEF display significant phenotypic variations which depend on the strain, the genetic background of the mice they are isolated
PCR and Sequencing Reaction Clean-Up Kit (Magnetic Bead System) 50 preps Product #60200
 3430 Schmon Parkway Thorold, ON, Canada L2V 4Y6 Phone: 866-667-4362 (905) 227-8848 Fax: (905) 227-1061 Email: [email protected] PCR and Sequencing Reaction Clean-Up Kit (Magnetic Bead System)
3430 Schmon Parkway Thorold, ON, Canada L2V 4Y6 Phone: 866-667-4362 (905) 227-8848 Fax: (905) 227-1061 Email: [email protected] PCR and Sequencing Reaction Clean-Up Kit (Magnetic Bead System)
