FDA and Optical Imaging Device/Combinations
|
|
|
- Percival Palmer
- 7 years ago
- Views:
Transcription
1 FDA and Optical Imaging Device/Combinations Neil R.P. Ogden, BME Chief, General Surgery Devices Branch 1 Division of Surgical Devices Office of Device Evaluation Food and Drug Administration New Hampshire Ave. WO 66, Room G414 Silver Spring, MD Neil.ogden@fda.hhs.gov (fax)
2 None Notice of Conflicts I currently work for FDA.
3 Disclaimer This talk represents the professional opinions of the author and is not an official document, guidance, or policy of the US government, the Department of Health and Human Services or the Food and Drug Administration, nor should any official endorsement be inferred.
4 Presentation Overview What we have done What we want to do How we can do it Considerations
5 Who does What at FDA?
6 Center for Food Safety & Applied Nutrition Center for Tobacco Products Center for Drug Evaluation & Research Center for Biologics Evaluation & Research Center for Devices & Radiological Health Center for Veterinary Medicine National Center for Toxicological Research
7 Light based Imaging and Combination products
8 Cleared Device Indications with ICG 1. For use in intra-operative visual assessment of the coronary vasculature and bypass grafts during coronary artery bypass (CABG) surgery. 2. For visual assessment of blood flow and related tissue perfusion during cardiovascular surgical procedures.
9 Cleared Device Indications with ICG, continued 3. For visual assessment of blood flow as an adjunctive method for the evaluation of tissue perfusion and related tissue-transfer circulation in tissue and free flaps used in plastic, micro- and reconstructive surgery. 4. For visual assessment of blood flow as an adjunctive method for the evaluation of flow in the native and anastomosed vessels, tissue perfusion and related tissue-transfer circulation in implanted and surrounding organs; to visualize blood flow indicative of perfusion of the donor implant prior to transplantation and to provide indication of organ function after transplantation
10 Cleared Device Indications with ICG, continued 5. For visual assessment of vessels, blood flow and related tissue perfusion with near infrared fluorescence imaging during minimally invasive surgery. 6. For visual assessment of vessels, blood flow, and related tissue perfusion with near infrared fluorescence imaging during minimally invasive robotic surgery. 7. For visual assessment of blood flow and related tissue perfusion during gastrointestinal surgery. 8. For use as an imaging tool in the evaluation of human tissue microstructure by providing two-dimensional, cross-sectional, realtime depth visualization
11 Cleared Device Indications, Cont d 9. For use in diagnostic and operative arthroscopic and endoscopic procedures to provide illumination and visualization of an interior cavity of the body through either a natural or surgical opening. 10. Intended to allow confocal laser imaging of the internal microstructure of tissues in the anatomical tract; enables surgeons to perform minimally invasive surgery using standard endoscopic visible light as well as visual assessment of vessels, blood flow and related tissue perfusion. 11. The da Vinci Fluorescence Imaging Vision System is intended to provide real time endoscopic visible and near infrared fluorescence imaging. The da Vinci Fluorescence Imaging Vision System enables surgeons to perform minimally invasive surgery using standard endoscopic visible light as well as visual assessment of vessels, blood flow and related tissue perfusion, and at least one of the major extrahepatic bile ducts (cystic duct, common bile duct and common hepatic duct), using near infrared imaging.
12 Presentation Overview What we have done What we want to do How can we do it Considerations
13 What Imaging Clinical Strategy? Complex issues dependent on many variables: General visualization alone Specific disease detection Combination products
14 Devices indicated for imaging specific diseases Device Indication drives data needs. Stand alone clinical data showing S&E will be needed.
15 Devices indicated for margin detection When and where is margin detection occurring?
16 Imaging Combination products Device plus drug or biologic will be reviewed together. Combination products: Complicated Likely - stand alone clinical data showing Safety & Effectiveness. If there s a predicate technical comparison.
17 Imaging Combination products, Continued Regulatory Pathway: Established Drug, same dosing, admin. Route - 510(k) or PMA. (ICG, Sodium Fluorescein) New Molecular Entity New Drug Application (NDA) New drug indication - NDA or supplements
18 Combo Product - Which Center has lead - RFD? Request For Designation (RFD) to our Office of Combination Products (OCP) will review Primary Mode of Action and how the combination achieves its Primary Intended Purpose(s) and what are the major FDA review challenges.
19 OCP Guidance Copies are available from: Office of Combination Products Food and Drug Administration WO32, Hub/Mail Room # New Hampshire Avenue Silver Spring, MD (Tel) (Fax) t.htm.
20 Informational needs for FDA device review Device Labeling Performance specifications Valid scientific evidence Tissue effects Mechanism-of-Action, like to have Clinical outcomes
21 VSE - 21 CFR (2) Valid scientific evidence is evidence from well-controlled investigations, partially controlled studies, studies and objective trials without matched controls, well-documented case histories conducted by qualified experts, and reports of significant human experience with a marketed device, from which it can fairly and responsibly be concluded by qualified experts that there is reasonable assurance of the safety and effectiveness of a device under its conditions of use.
22 Presentation Overview What we have done What we want to do How can we do it Considerations
23 Phantoms
24 Standardization Efforts for Optical Imaging Consensus standards are corner stones for other imaging modalities, serving both the industry and the government agencies Increasing needs to standardize aspects of optical imaging - to facilitate product/clinical trial development
25 What to standardize? Optical Image calibration and performance evaluation Optical Image quality, # pixels, color rendering Optical Image size, resolution, contrast, precision Optical Image capture, CCD, ICCD, ultrasound, OCT, photoacoustic Optical Image creation
26 Summary Comments Optical imaging technology ---- non-ionizing, real-time microscopic observation An emerging field in medical applications, particularly coupled with general surgery / minimally invasive surgery Based on technologies and indications, regulated as Class I, II, and III devices Specific disease detection, e.g., cancer, in screening and for intraoperative guidance Challenges for combination products regulatory route with new drugs or approved drugs across-center efforts with CDER, CBER, and with Office of Combination Products
27
28 Extra slides
29
November 20, 2014. Vascular Flow Technologies Ltd. Edwin Lindsay VP of QA/RA Prospect Business Centre, Gemini Cresent Dundee DD2 1TY United Kingdom
 DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service Food and Drug Administration 10903 New Hampshire Avenue Document Control Center - WO66-G609 Silver Spring, MD 20993-0002 November 20, 2014 Vascular
DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service Food and Drug Administration 10903 New Hampshire Avenue Document Control Center - WO66-G609 Silver Spring, MD 20993-0002 November 20, 2014 Vascular
SPY ing on Coronaries: The Use Of Intraoperative Imaging to Avoid Coronary Artery Injury During Redo Congenital Cardiac Surgery
 SPY ing on Coronaries: The Use Of Intraoperative Imaging to Avoid Coronary Artery Injury During Redo Congenital Cardiac Surgery Avianne P. Bunnell, B.S. Kamal K. Pourmoghadam, M.D. William M. DeCampli,
SPY ing on Coronaries: The Use Of Intraoperative Imaging to Avoid Coronary Artery Injury During Redo Congenital Cardiac Surgery Avianne P. Bunnell, B.S. Kamal K. Pourmoghadam, M.D. William M. DeCampli,
da Vinci Mitral Valve Repair
 da Vinci Mitral Valve Repair Changing the Experience of Cardiac Surgery Are you a candidate for the latest treatment option for mitral valve disease? Your doctor may be able to offer you a new, minimally
da Vinci Mitral Valve Repair Changing the Experience of Cardiac Surgery Are you a candidate for the latest treatment option for mitral valve disease? Your doctor may be able to offer you a new, minimally
Guidance for Industry and Investigators Safety Reporting Requirements for INDs and BA/BE Studies- Small Entity Compliance Guide
 Guidance for Industry and Investigators Safety Reporting Requirements for INDs and BA/BE Studies- Small Entity Compliance Guide U.S. Department of Health and Human Services Center for Drug Evaluation and
Guidance for Industry and Investigators Safety Reporting Requirements for INDs and BA/BE Studies- Small Entity Compliance Guide U.S. Department of Health and Human Services Center for Drug Evaluation and
Meetings with CDER Judit Milstein
 Meetings with CDER Judit Milstein Division of Transplant and Ophthalmology Products (DTOP) Office of Antimicrobial Products (OAP), Office of New Drugs (OND) Center for Drug Evaluation and Research (CDER)
Meetings with CDER Judit Milstein Division of Transplant and Ophthalmology Products (DTOP) Office of Antimicrobial Products (OAP), Office of New Drugs (OND) Center for Drug Evaluation and Research (CDER)
University of Texas Medical School at Houston. April 14, 2015
 DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service Food and Drug Administration 10903 New Hampshire Avenue Document Control Center WO66-G609 Silver Spring, MD 20993-0002 University of Texas Medical
DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service Food and Drug Administration 10903 New Hampshire Avenue Document Control Center WO66-G609 Silver Spring, MD 20993-0002 University of Texas Medical
UCLA Asian Liver Program
 CLA Program Update Program Faculty Myron J. Tong, PhD, MD Professor of Medicine Hepatology Director, Asian Liver Program Surgery Ronald W. Busuttil, MD, PhD Executive Chair Department of Surgery Director,
CLA Program Update Program Faculty Myron J. Tong, PhD, MD Professor of Medicine Hepatology Director, Asian Liver Program Surgery Ronald W. Busuttil, MD, PhD Executive Chair Department of Surgery Director,
Combination Products Regulation in the United States
 Combination Products Regulation in the United States Presenter: Scott Sardeson RAC US/EU 3M Health Care St. Paul, MN USA 1 Presentation Outline Combination products Definitions and Regulations Jurisdiction
Combination Products Regulation in the United States Presenter: Scott Sardeson RAC US/EU 3M Health Care St. Paul, MN USA 1 Presentation Outline Combination products Definitions and Regulations Jurisdiction
Information Sheet Guidance For IRBs, Clinical Investigators, and Sponsors
 Information Sheet Guidance For IRBs, Clinical Investigators, and Sponsors FDA Institutional Review Board Inspections Additional copies are available from: Office of Good Clinical Practice Office of Special
Information Sheet Guidance For IRBs, Clinical Investigators, and Sponsors FDA Institutional Review Board Inspections Additional copies are available from: Office of Good Clinical Practice Office of Special
Neuro imaging: looking with lasers in the brain
 Neuro imaging: looking with lasers in the brain Aim: To image life cells, label free, with cellular resolution in deep tissue Marloes Groot Vrije Universiteit Amsterdam Faculteit Exacte Wetenschappen Natuurkunde
Neuro imaging: looking with lasers in the brain Aim: To image life cells, label free, with cellular resolution in deep tissue Marloes Groot Vrije Universiteit Amsterdam Faculteit Exacte Wetenschappen Natuurkunde
Facing Lung Cancer? Learn why da Vinci Surgery may be your best treatment option for lung cancer.
 Facing Lung Cancer? Learn why da Vinci Surgery may be your best treatment option for lung cancer. The Condition: Lung Cancer The lung is the organ that moves oxygen through your body. You have two lungs
Facing Lung Cancer? Learn why da Vinci Surgery may be your best treatment option for lung cancer. The Condition: Lung Cancer The lung is the organ that moves oxygen through your body. You have two lungs
Good Review Practice. October 2009 Labeling
 Guidance for Industry and Review Staff Labeling for Human Prescription Drug and Biological Products Determining Established Pharmacologic Class for Use in the Highlights of Prescribing Information Good
Guidance for Industry and Review Staff Labeling for Human Prescription Drug and Biological Products Determining Established Pharmacologic Class for Use in the Highlights of Prescribing Information Good
Guidance for Industry
 Guidance for Industry General/Specific Intended Use Document Issued: November 4, 1998 U.S. Department of Health and Human Services Food and Drug Administration Center for Devices and Radiological Health
Guidance for Industry General/Specific Intended Use Document Issued: November 4, 1998 U.S. Department of Health and Human Services Food and Drug Administration Center for Devices and Radiological Health
Guidance for Industry Bar Code Label Requirements Questions and Answers
 Guidance for Industry Bar Code Label Requirements Questions and Answers For questions on the content of this guidance, contact the Center for Drug Evaluation and Research or the Center for Biologics Evaluation
Guidance for Industry Bar Code Label Requirements Questions and Answers For questions on the content of this guidance, contact the Center for Drug Evaluation and Research or the Center for Biologics Evaluation
Phoenix Thera-Lase Systems, LLC Ms. Diane Rutherford Ken Block Consulting 1201 Richardson Drive, Suite 280 Richardson, Texas 75080
 DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service Food and Drug Administration 10903 New Hampshire Avenue Document Control Center WO66-G609 Silver Spring, MD 20993-0002 Phoenix Thera-Lase Systems,
DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service Food and Drug Administration 10903 New Hampshire Avenue Document Control Center WO66-G609 Silver Spring, MD 20993-0002 Phoenix Thera-Lase Systems,
Information Sheet Guidance For IRBs, Clinical Investigators, and Sponsors
 Information Sheet Guidance For IRBs, Clinical Investigators, and Sponsors Frequently Asked Questions About Medical Devices Additional copies are available from: Office of Good Clinical Practice Office
Information Sheet Guidance For IRBs, Clinical Investigators, and Sponsors Frequently Asked Questions About Medical Devices Additional copies are available from: Office of Good Clinical Practice Office
CDISC Journal. Regulatory Submissions for Medical Devices and Diagnostics: The Basics
 CDISC Journal Clinical Data Interchange Standards Consortium O ctober 2011 Regulatory Submissions for Medical Devices and Diagnostics: The Basics By Carey G. Smoak Abstract Medical devices and diagnostics
CDISC Journal Clinical Data Interchange Standards Consortium O ctober 2011 Regulatory Submissions for Medical Devices and Diagnostics: The Basics By Carey G. Smoak Abstract Medical devices and diagnostics
Clinical Practice Assessment Robotic surgery
 Clinical Practice Assessment Robotic surgery Background: Surgery is by nature invasive. Efforts have been made over time to reduce complications and the trauma inherently associated with surgery through
Clinical Practice Assessment Robotic surgery Background: Surgery is by nature invasive. Efforts have been made over time to reduce complications and the trauma inherently associated with surgery through
FDA STAFF MANUAL GUIDES, VOLUME I - ORGANIZATIONS AND FUNCTIONS FOOD AND DRUG ADMINISTRATION OFFICE OF OPERATIONS
 SMG 1117.2112 FDA STAFF MANUAL GUIDES, VOLUME I - ORGANIZATIONS AND FUNCTIONS FOOD AND DRUG ADMINISTRATION OFFICE OF OPERATIONS OFFICE OF INFORMATION MANAGEMENT AND TECHNOLOGY OFFICE OF INFORMATION MANAGEMENT
SMG 1117.2112 FDA STAFF MANUAL GUIDES, VOLUME I - ORGANIZATIONS AND FUNCTIONS FOOD AND DRUG ADMINISTRATION OFFICE OF OPERATIONS OFFICE OF INFORMATION MANAGEMENT AND TECHNOLOGY OFFICE OF INFORMATION MANAGEMENT
restricted to certain centers and certain patients, preferably in some sort of experimental trial format.
 Managing Pancreatic Cancer, Part 4: Pancreatic Cancer Surgery, Complications, & the Importance of Surgical Volume Dr. Matthew Katz, Surgeon, MD Anderson Cancer Center, Houston, TX I m going to talk a little
Managing Pancreatic Cancer, Part 4: Pancreatic Cancer Surgery, Complications, & the Importance of Surgical Volume Dr. Matthew Katz, Surgeon, MD Anderson Cancer Center, Houston, TX I m going to talk a little
Heart Center Packages
 Heart Center Packages For more information and appointments, Please contact The Heart Center of Excellence at the American Hospital Dubai Tel: +971-4-377-6571 Email: heartcenter@ahdubai.com www.ahdubai.com
Heart Center Packages For more information and appointments, Please contact The Heart Center of Excellence at the American Hospital Dubai Tel: +971-4-377-6571 Email: heartcenter@ahdubai.com www.ahdubai.com
Medical Device Data Systems, Medical Image Storage Devices, and Medical Image
 4164-01-P DEPARTMENT OF HEALTH AND HUMAN SERVICES Food and Drug Administration [Docket No. FDA-2014-D-0798] Medical Device Data Systems, Medical Image Storage Devices, and Medical Image Communication Devices;
4164-01-P DEPARTMENT OF HEALTH AND HUMAN SERVICES Food and Drug Administration [Docket No. FDA-2014-D-0798] Medical Device Data Systems, Medical Image Storage Devices, and Medical Image Communication Devices;
Guidance for Industry
 Guidance for Industry Internet/Social Media Platforms: Correcting Independent Third-Party Misinformation About Prescription Drugs and Medical Devices DRAFT GUIDANCE This guidance document is being distributed
Guidance for Industry Internet/Social Media Platforms: Correcting Independent Third-Party Misinformation About Prescription Drugs and Medical Devices DRAFT GUIDANCE This guidance document is being distributed
How To Write A Cer
 2014 CBER Update: Advertising & Promotional Labeling Compliance Alpita Popat, PharmD, MBA Consumer Safety Officer Advertising & Promotional Labeling Branch Division of Case Management Office of Compliance
2014 CBER Update: Advertising & Promotional Labeling Compliance Alpita Popat, PharmD, MBA Consumer Safety Officer Advertising & Promotional Labeling Branch Division of Case Management Office of Compliance
510(K) SUMMARY. 510(k) Number KOS'00-r
 " iirtual Ports Virtsua Ports Virtual Ports Ltd. 510(K) SUMMARY 510(k) Number KOS'00-r MAR 2 12008 Applicant's Name: Contact Person: Trade Name: Common name: Classification: Predicate Devices: Device Description:
" iirtual Ports Virtsua Ports Virtual Ports Ltd. 510(K) SUMMARY 510(k) Number KOS'00-r MAR 2 12008 Applicant's Name: Contact Person: Trade Name: Common name: Classification: Predicate Devices: Device Description:
Surgery Support System as a Surgeon s Advanced Hand and Eye
 Surgery Support System as a Surgeon s Advanced Hand and Eye 8 Surgery Support System as a Surgeon s Advanced Hand and Eye Kazutoshi Kan Michio Oikawa Takashi Azuma Shio Miyamoto OVERVIEW: A surgery support
Surgery Support System as a Surgeon s Advanced Hand and Eye 8 Surgery Support System as a Surgeon s Advanced Hand and Eye Kazutoshi Kan Michio Oikawa Takashi Azuma Shio Miyamoto OVERVIEW: A surgery support
Formal FDA Meeting Request: Guidance and Template
 Formal FDA Meeting Request: Guidance and Template ICTR Navigators July 23, 2011 Version 2.0 Page 1 of 20 1.0 Table of Contents Section Page 1.0 Table of Contents 2 2.0 Abbreviations 2 3.0 FDA Regulations
Formal FDA Meeting Request: Guidance and Template ICTR Navigators July 23, 2011 Version 2.0 Page 1 of 20 1.0 Table of Contents Section Page 1.0 Table of Contents 2 2.0 Abbreviations 2 3.0 FDA Regulations
Cancer treatment. TOP EUROPEAN CANCER EXPERTISE The path to recovery
 Cancer treatment TOP EUROPEAN CANCER EXPERTISE The path to recovery 0% LAND OF HIGH QUALITY HEALTHCARE Located in Finland, a land of high quality healthcare, Helsinki University Hospital is regarded as
Cancer treatment TOP EUROPEAN CANCER EXPERTISE The path to recovery 0% LAND OF HIGH QUALITY HEALTHCARE Located in Finland, a land of high quality healthcare, Helsinki University Hospital is regarded as
What is a medical device? Medical Devices: Roadmap to Market. Kathryn Klaus, Esq.
 Medical Devices: Roadmap to Market Kathryn Klaus, Esq. The last installment of Regulatory 360 discussed the FDA organization in general where it came from and a broad overview of how it operates, as well
Medical Devices: Roadmap to Market Kathryn Klaus, Esq. The last installment of Regulatory 360 discussed the FDA organization in general where it came from and a broad overview of how it operates, as well
Inova. Breast Care Institute
 Inova Breast Care Institute At the Inova Breast Care Institute, our commitment is to provide expert care for you, every step of the way. Our multidisciplinary team of more than 80 experts provides a full
Inova Breast Care Institute At the Inova Breast Care Institute, our commitment is to provide expert care for you, every step of the way. Our multidisciplinary team of more than 80 experts provides a full
NC DIVISION OF SERVICES FOR THE BLIND POLICIES AND PROCEDURES VOCATIONAL REHABILITATION
 NC DIVISION OF SERVICES FOR THE BLIND POLICIES AND PROCEDURES VOCATIONAL REHABILITATION Section: E Revision History Revised 01/97; 05/03; 02/08; 04/08; 03/09; 05/09; 12/09; 01/11; 12/14 An individual is
NC DIVISION OF SERVICES FOR THE BLIND POLICIES AND PROCEDURES VOCATIONAL REHABILITATION Section: E Revision History Revised 01/97; 05/03; 02/08; 04/08; 03/09; 05/09; 12/09; 01/11; 12/14 An individual is
GE Healthcare. Vivid 7 Dimension Cardiovascular Ultrasound System
 GE Healthcare Vivid 7 Dimension Cardiovascular Ultrasound System New dime Introducing multi-dimensional and 4D imaging. The ability to incorporate multi-dimensional and 4D imaging into your clinical routine
GE Healthcare Vivid 7 Dimension Cardiovascular Ultrasound System New dime Introducing multi-dimensional and 4D imaging. The ability to incorporate multi-dimensional and 4D imaging into your clinical routine
Guidance for Clinical Investigators, Sponsors, and IRBs
 Guidance for Clinical Investigators, Sponsors, and IRBs Adverse Event Reporting to IRBs Improving Human Subject Protection U.S. Department of Health and Human Services Office of the Commissioner (OC) Center
Guidance for Clinical Investigators, Sponsors, and IRBs Adverse Event Reporting to IRBs Improving Human Subject Protection U.S. Department of Health and Human Services Office of the Commissioner (OC) Center
L~l t l V~! MAY 23 200?
 L~l t l V~! MAY 23 200? 510 (k) Summay (As required by 21 CFR 807.92 and 21 CFR 807.93) NAME OF SPONSOR: 510(K) CONTACT: DePuy Orthopaedics, Inc. 700 Orthopaedic Drive Warsaw, Indiana 46582 Establishment
L~l t l V~! MAY 23 200? 510 (k) Summay (As required by 21 CFR 807.92 and 21 CFR 807.93) NAME OF SPONSOR: 510(K) CONTACT: DePuy Orthopaedics, Inc. 700 Orthopaedic Drive Warsaw, Indiana 46582 Establishment
SURGICAL SERVICES. Touching Lives
 SURGICAL SERVICES Touching Lives with MODERN TOOLS of EXPERT PHYSICIANS in the Hands and Staff FROM DISCOVERY TO RECOVERY St. Mary s Health System offers a full range of surgical services in almost every
SURGICAL SERVICES Touching Lives with MODERN TOOLS of EXPERT PHYSICIANS in the Hands and Staff FROM DISCOVERY TO RECOVERY St. Mary s Health System offers a full range of surgical services in almost every
DRAFT GUIDANCE. This guidance document is being distributed for comment purposes only.
 Guidance for Industry Fulfilling Regulatory Requirements for Postmarketing Submissions of Interactive Promotional Media for Prescription Human and Animal Drugs and Biologics DRAFT GUIDANCE This guidance
Guidance for Industry Fulfilling Regulatory Requirements for Postmarketing Submissions of Interactive Promotional Media for Prescription Human and Animal Drugs and Biologics DRAFT GUIDANCE This guidance
STUDY PLAN FOR THE CERTIFICATE OF THE HIGHER SPECIALIZATION IN ( Diagnostic Radiology)
 STUDY PLAN FOR THE CERTIFICATE OF THE HIGHER SPECIALIZATION IN ( Diagnostic Radiology) plan number :15/11/97/NT I-GENERAL RULES AND CONDITIONS: 1- This plan conforms to the regulations of granting the
STUDY PLAN FOR THE CERTIFICATE OF THE HIGHER SPECIALIZATION IN ( Diagnostic Radiology) plan number :15/11/97/NT I-GENERAL RULES AND CONDITIONS: 1- This plan conforms to the regulations of granting the
Specific Standards of Accreditation for Residency Programs in General Surgery
 Specific Standards of Accreditation for Residency Programs in General Surgery 2010 INTRODUCTION The purpose of this document is to provide program directors and surveyors with an interpretation of the
Specific Standards of Accreditation for Residency Programs in General Surgery 2010 INTRODUCTION The purpose of this document is to provide program directors and surveyors with an interpretation of the
PRODYNOV. Targeted Photodynamic Therapy of Ovarian Peritoneal Carcinomatosis ONCO-THAI. Image Assisted Laser Therapy for Oncology
 PRODYNOV Targeted Photodynamic Therapy of Ovarian Peritoneal Carcinomatosis ONCO-THAI Image Assisted Laser Therapy for Oncology Inserm ONCO-THAI «Image Assisted Laser Therapy for Oncology» Inserm ONCO-THAI
PRODYNOV Targeted Photodynamic Therapy of Ovarian Peritoneal Carcinomatosis ONCO-THAI Image Assisted Laser Therapy for Oncology Inserm ONCO-THAI «Image Assisted Laser Therapy for Oncology» Inserm ONCO-THAI
Guidance for Industry Pyrogen and Endotoxins Testing: Questions and Answers
 Guidance for Industry Pyrogen and Endotoxins Testing: Questions and Answers U.S. Department of Health and Human Services Food and Drug Administration Center for Drug Evaluation and Research (CDER) Center
Guidance for Industry Pyrogen and Endotoxins Testing: Questions and Answers U.S. Department of Health and Human Services Food and Drug Administration Center for Drug Evaluation and Research (CDER) Center
The lungs What is lung cancer? How common is it? Risks & symptoms Diagnosis & treatment options
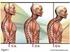 Why We re Here The lungs What is lung cancer? How common is it? Risks & symptoms Diagnosis & treatment options What Are Lungs? What Do They Do? 1 Located in the chest Allow you to breathe Provide oxygen
Why We re Here The lungs What is lung cancer? How common is it? Risks & symptoms Diagnosis & treatment options What Are Lungs? What Do They Do? 1 Located in the chest Allow you to breathe Provide oxygen
Guidance for Industry
 Guidance for Industry IND Exemptions for Studies of Lawfully Marketed Drug or Biological Products for the Treatment of Cancer U.S. Department of Health and Human Services Food and Drug Administration Center
Guidance for Industry IND Exemptions for Studies of Lawfully Marketed Drug or Biological Products for the Treatment of Cancer U.S. Department of Health and Human Services Food and Drug Administration Center
FACTS about. MENTOR MemoryGel. Silicone Gel Filled Breast Implants
 FACTS about MENTOR MemoryGel Silicone Gel Filled Breast Implants Are you considering breast implant surgery but not certain which type of implant to choose? You re not alone. Science-based information
FACTS about MENTOR MemoryGel Silicone Gel Filled Breast Implants Are you considering breast implant surgery but not certain which type of implant to choose? You re not alone. Science-based information
Information Sheet Guidance for Sponsors, Clinical Investigators, and IRBs
 Information Sheet Guidance for Sponsors, Clinical Investigators, and IRBs Frequently Asked Questions Statement of Investigator (Form FDA 1572) U.S. Department of Health and Human Services Food and Drug
Information Sheet Guidance for Sponsors, Clinical Investigators, and IRBs Frequently Asked Questions Statement of Investigator (Form FDA 1572) U.S. Department of Health and Human Services Food and Drug
A pretty picture, or a measurement? Retinal Imaging
 Big Data Challenges A pretty picture, or a measurement? Organelles Dynamics Cells Retinal Imaging Physiology Pathology Fundus Camera Optical coherence tomography Fluorescence Histology High Content Screening
Big Data Challenges A pretty picture, or a measurement? Organelles Dynamics Cells Retinal Imaging Physiology Pathology Fundus Camera Optical coherence tomography Fluorescence Histology High Content Screening
National Framework for Excellence in
 National Framework for Excellence in Lung Cancer Screening and Continuum of Care declaration of purpose Rights and Expectations THE RIGHTS OF THE PEOPLE Lung cancer kills more Americans than the next four
National Framework for Excellence in Lung Cancer Screening and Continuum of Care declaration of purpose Rights and Expectations THE RIGHTS OF THE PEOPLE Lung cancer kills more Americans than the next four
Guidance for Industry Classifying Resubmissions in Response to Action Letters
 Guidance for Industry Classifying Resubmissions in Response to Action Letters U.S. Department of Health and Human Services Food and Drug Administration Center for Drug Evaluation and Research (CDER) Center
Guidance for Industry Classifying Resubmissions in Response to Action Letters U.S. Department of Health and Human Services Food and Drug Administration Center for Drug Evaluation and Research (CDER) Center
Guidance for Industry Formal Meetings Between the FDA and Sponsors or Applicants
 Guidance for Industry Formal Meetings Between the FDA and Sponsors or Applicants U.S. Department of Health and Human Services Food and Drug Administration Center for Drug Evaluation and Research (CDER)
Guidance for Industry Formal Meetings Between the FDA and Sponsors or Applicants U.S. Department of Health and Human Services Food and Drug Administration Center for Drug Evaluation and Research (CDER)
Food and Drug Administration (FDA) Administrative Contacts DIS Liaisons for Visiting Scientist (FTE) cases
 Office of Research Services DIVISION OF INTERNATIONAL SERVICES DATA SHEET Food and Drug Administration (FDA) Administrative Contacts DIS Liaisons for Visiting Scientist (FTE) cases Tania Tse Director,
Office of Research Services DIVISION OF INTERNATIONAL SERVICES DATA SHEET Food and Drug Administration (FDA) Administrative Contacts DIS Liaisons for Visiting Scientist (FTE) cases Tania Tse Director,
Minimally Invasive Spine Surgery
 Chapter 1 Minimally Invasive Spine Surgery 1 H.M. Mayer Primum non nocere First do no harm In the long history of surgery it always has been a basic principle to restrict the iatrogenic trauma done to
Chapter 1 Minimally Invasive Spine Surgery 1 H.M. Mayer Primum non nocere First do no harm In the long history of surgery it always has been a basic principle to restrict the iatrogenic trauma done to
Use of Nucleic Acid Tests to Reduce the Risk of Transmission of Hepatitis B Virus from Donors
 This document is scheduled to be published in the Federal Register on 01/08/2016 and available online at http://federalregister.gov/a/2016-00149, and on FDsys.gov 4164-01-P DEPARTMENT OF HEALTH AND HUMAN
This document is scheduled to be published in the Federal Register on 01/08/2016 and available online at http://federalregister.gov/a/2016-00149, and on FDsys.gov 4164-01-P DEPARTMENT OF HEALTH AND HUMAN
Guidance for Industry FDA Approval of New Cancer Treatment Uses for Marketed Drug and Biological Products
 Guidance for Industry FDA Approval of New Cancer Treatment Uses for Marketed Drug and Biological Products U.S. Department of Health and Human Services Food and Drug Administration Center for Drug Evaluation
Guidance for Industry FDA Approval of New Cancer Treatment Uses for Marketed Drug and Biological Products U.S. Department of Health and Human Services Food and Drug Administration Center for Drug Evaluation
Preparing for the Pre-Approval Inspection What to do Before the FDA Arrives. Barry A. Friedman, Ph.D. Consultant
 Preparing for the Pre-Approval Inspection What to do Before the FDA Arrives Barry A. Friedman, Ph.D. Consultant FDA Overview FDA is a consumer protection agency within the Department of Health & Human
Preparing for the Pre-Approval Inspection What to do Before the FDA Arrives Barry A. Friedman, Ph.D. Consultant FDA Overview FDA is a consumer protection agency within the Department of Health & Human
Guidance Medication Guides Distribution Requirements and Inclusion in Risk Evaluation and Mitigation Strategies (REMS)
 Guidance s Distribution Requirements and Inclusion in Risk Evaluation and Mitigation Strategies (REMS) U.S. Department of Health and Human Services Food and Drug Administration Center for Drug Evaluation
Guidance s Distribution Requirements and Inclusion in Risk Evaluation and Mitigation Strategies (REMS) U.S. Department of Health and Human Services Food and Drug Administration Center for Drug Evaluation
Chapter 8 WHAT THE STATUTE AND REGULATIONS SAY. Combination Products. Key Points
 Chapter 8 Combination Products Suzanne O Shea, Baker & Daniels, LLP, Indianapolis, IN Key Points A combination product comprises two or more different types of regulated components. It can be a drug/device,
Chapter 8 Combination Products Suzanne O Shea, Baker & Daniels, LLP, Indianapolis, IN Key Points A combination product comprises two or more different types of regulated components. It can be a drug/device,
SAMPLE. Asia-Pacific Interventional Cardiology Procedures Outlook to 2020. Reference Code: GDMECR0061PDB. Publication Date: May 2014
 Asia-Pacific Interventional Cardiology Procedures Outlook to 2020 Reference Code: GDMECR0061PDB Publication Date: May 2014 Page 1 1 Table of Contents 1 Table of Contents... 2 1.1 List of Tables... 4 1.2
Asia-Pacific Interventional Cardiology Procedures Outlook to 2020 Reference Code: GDMECR0061PDB Publication Date: May 2014 Page 1 1 Table of Contents 1 Table of Contents... 2 1.1 List of Tables... 4 1.2
Guidance for Industry
 Guidance for Industry Cancer Drug and Biological Products Clinical Data in Marketing Applications U.S. Department of Health and Human Services Food and Drug Administration Center for Drug Evaluation and
Guidance for Industry Cancer Drug and Biological Products Clinical Data in Marketing Applications U.S. Department of Health and Human Services Food and Drug Administration Center for Drug Evaluation and
DICOM Grid, Inc. January 25, 2016. Senior Consultant Biologics Consulting Group, Inc. 400 N. Washington Street, Suite 100 ALEXANDRIA VA 22314
 DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service Food and Drug Administration 10903 New Hampshire Avenue Document Control Center WO66-G609 Silver Spring, MD 20993-0002 DICOM Grid, Inc. Senior
DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service Food and Drug Administration 10903 New Hampshire Avenue Document Control Center WO66-G609 Silver Spring, MD 20993-0002 DICOM Grid, Inc. Senior
Specific Basic Standards for Osteopathic Fellowship Training in Cardiology
 Specific Basic Standards for Osteopathic Fellowship Training in Cardiology American Osteopathic Association and American College of Osteopathic Internists BOT 07/2006 Rev. BOT 03/2009 Rev. BOT 07/2011
Specific Basic Standards for Osteopathic Fellowship Training in Cardiology American Osteopathic Association and American College of Osteopathic Internists BOT 07/2006 Rev. BOT 03/2009 Rev. BOT 07/2011
Guidance for Industry Direct-to-Consumer Television Advertisements FDAAA DTC Television Ad Pre- Dissemination Review Program
 Guidance for Industry Direct-to-Consumer Television Advertisements FDAAA DTC Television Ad Pre- Dissemination Review Program DRAFT GUIDANCE This guidance document is being distributed for comment purposes
Guidance for Industry Direct-to-Consumer Television Advertisements FDAAA DTC Television Ad Pre- Dissemination Review Program DRAFT GUIDANCE This guidance document is being distributed for comment purposes
da Vinci Prostatectomy Information Guide (Robotically-Assisted Radical Prostatectomy)
 da Vinci Prostatectomy Information Guide (Robotically-Assisted Radical Prostatectomy) Prostate Cancer Overview Prostate cancer is a disease in which malignant (cancer) cells form in the tissues of the
da Vinci Prostatectomy Information Guide (Robotically-Assisted Radical Prostatectomy) Prostate Cancer Overview Prostate cancer is a disease in which malignant (cancer) cells form in the tissues of the
The Whipple Procedure. Sally Hodges, Ph.D.(c) Given the length and difficulty of the procedure, regardless of the diagnosis, certain
 The Whipple Procedure Sally Hodges, Ph.D.(c) Preoperative procedures Given the length and difficulty of the procedure, regardless of the diagnosis, certain assurances must occur prior to offering a patient
The Whipple Procedure Sally Hodges, Ph.D.(c) Preoperative procedures Given the length and difficulty of the procedure, regardless of the diagnosis, certain assurances must occur prior to offering a patient
GROCERIES. Helps cover costs associated with heart attack, stroke, or heart disease
 What if you suffered from a heart attack or a stroke... could you pay for your out-of-pocket treatment expenses, plus cover daily living expenses? GROCERIES CAR HOME PRESCRIPTIONS Heart/Stroke Insurance
What if you suffered from a heart attack or a stroke... could you pay for your out-of-pocket treatment expenses, plus cover daily living expenses? GROCERIES CAR HOME PRESCRIPTIONS Heart/Stroke Insurance
Bile Duct Diseases and Problems
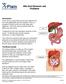 Bile Duct Diseases and Problems Introduction A bile duct is a tube that carries bile between the liver and gallbladder and the intestine. Bile is a substance made by the liver that helps with digestion.
Bile Duct Diseases and Problems Introduction A bile duct is a tube that carries bile between the liver and gallbladder and the intestine. Bile is a substance made by the liver that helps with digestion.
Welcome to our Office!
 Randy Burks, MD, FACS Raymond Gailitis, MD, FACS Northwest Medical Plaza 5800 Colonial Drive, Suite 100-103 Margate, Florida 33063 954-977-8770 Comprehensive Eye Care 954-969-0090 LASIK 954-977-8774 Fax
Randy Burks, MD, FACS Raymond Gailitis, MD, FACS Northwest Medical Plaza 5800 Colonial Drive, Suite 100-103 Margate, Florida 33063 954-977-8770 Comprehensive Eye Care 954-969-0090 LASIK 954-977-8774 Fax
Guidance for Industry. 21 CFR Part 11; Electronic. Records; Electronic Signatures. Time Stamps
 Guidance for Industry 21 CFR Part 11; Electronic Records; Electronic Signatures Time Stamps Draft Guidance This guidance document is being distributed for comment purposes only. Comments and suggestions
Guidance for Industry 21 CFR Part 11; Electronic Records; Electronic Signatures Time Stamps Draft Guidance This guidance document is being distributed for comment purposes only. Comments and suggestions
July 24, 2015. Dear Ms. Rhodes:
 DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service Food and Drug Administration 10903 New Hampshire Avenue Document Control Center WO66-G609 Silver Spring, MD 20993-0002 InnoVision, Incorporated
DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service Food and Drug Administration 10903 New Hampshire Avenue Document Control Center WO66-G609 Silver Spring, MD 20993-0002 InnoVision, Incorporated
NEW HORIZONS IN CORNEAL SURGERY VERSATILE FEMTOSECOND LASER WORKSTATION WE FOCUS ON PERFECTION
 NEW HORIZONS VERSATILE FEMTOSECOND IN CORNEAL LASER WORKSTATION SURGERY WE FOCUS ON PERFECTION ADVANCED FEMTOSECOND LASER TECHNOLOGY COMMITTED TO VERSATILITY > ONE SYSTEM FOR ALL FEMTO-APPLICATIONS > ANATOMICALLY
NEW HORIZONS VERSATILE FEMTOSECOND IN CORNEAL LASER WORKSTATION SURGERY WE FOCUS ON PERFECTION ADVANCED FEMTOSECOND LASER TECHNOLOGY COMMITTED TO VERSATILITY > ONE SYSTEM FOR ALL FEMTO-APPLICATIONS > ANATOMICALLY
August 12, 2014. Oertli Instrumente AG Ms. Karin Rohr Head of Quality Management & Regulatory Affairs Hafnerwisenstrasse 4 CH 9442 Berneck Switzerland
 DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service August 12, 2014 Food and Drug Administration 10903 New Hampshire Avenue Document Control Center WO66-G609 Silver Spring, MD 20993-0002 Oertli
DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service August 12, 2014 Food and Drug Administration 10903 New Hampshire Avenue Document Control Center WO66-G609 Silver Spring, MD 20993-0002 Oertli
FDA Regulation of Hearing Aids. Eric A. Mann, MD, PhD Clinical Deputy Director Division of Ophthalmic and ENT Devices ODE/CDRH/FDA
 FDA Regulation of Hearing Aids Eric A. Mann, MD, PhD Clinical Deputy Director Division of Ophthalmic ENT ODE/CDRH/FDA U.S. Food Drug Administration Presentation Outline Overview of device regulations riskbased
FDA Regulation of Hearing Aids Eric A. Mann, MD, PhD Clinical Deputy Director Division of Ophthalmic ENT ODE/CDRH/FDA U.S. Food Drug Administration Presentation Outline Overview of device regulations riskbased
Guidance for Industry
 Guidance for Industry Product Name Placement, Size, and Prominence in Advertising and Promotional Labeling DRAFT GUIDANCE This guidance document is being distributed for comment purposes only. Comments
Guidance for Industry Product Name Placement, Size, and Prominence in Advertising and Promotional Labeling DRAFT GUIDANCE This guidance document is being distributed for comment purposes only. Comments
Corporate Medical Policy Implantation of Intrastromal Corneal Ring Segments
 Corporate Medical Policy Implantation of Intrastromal Corneal Ring Segments File Name: Origination: Last CAP Review: Next CAP Review: Last Review: implantation_of_intrastromal_corneal_ring_segments 8/2008
Corporate Medical Policy Implantation of Intrastromal Corneal Ring Segments File Name: Origination: Last CAP Review: Next CAP Review: Last Review: implantation_of_intrastromal_corneal_ring_segments 8/2008
Guidance for Industry
 Guidance for Industry Expanded Access to Investigational Drugs for Treatment Use Qs & As DRAFT GUIDANCE This guidance document is being distributed for comment purposes only. Comments and suggestions regarding
Guidance for Industry Expanded Access to Investigational Drugs for Treatment Use Qs & As DRAFT GUIDANCE This guidance document is being distributed for comment purposes only. Comments and suggestions regarding
The Shape Of Things To Come. Dr. F.P. Wieringa LVO congres 7-8 feb 2013
 The Shape Of Things To Come Dr. F.P. Wieringa LVO congres 7-8 feb 2013 Long as you ve got your health, Bradley W. Schenck 2004 f.wieringa@tno.nl Medical technology innovation eco-system Hospitals Universities
The Shape Of Things To Come Dr. F.P. Wieringa LVO congres 7-8 feb 2013 Long as you ve got your health, Bradley W. Schenck 2004 f.wieringa@tno.nl Medical technology innovation eco-system Hospitals Universities
Ad-tech Medical Instrument Corporation. Principal Consultant Quality & Regulatory Associates, LLC. 800 Levanger Lane. Stoughton, Wisconsin 53589
 DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service March 4, 2016 Food and Drug Administration 10903 New Hampshire Avenue Document Control Center - WO66-G609 Silver Spring, MD 20993-0002 Ad-tech
DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service March 4, 2016 Food and Drug Administration 10903 New Hampshire Avenue Document Control Center - WO66-G609 Silver Spring, MD 20993-0002 Ad-tech
Keeping Your Eyes Healthy after Treatment for Childhood Cancer
 Keeping Your Eyes Healthy after Treatment for Childhood Cancer High doses of radiation to the brain, eye, or eye socket (orbit) during treatment for childhood cancer can have a long-lasting affect on the
Keeping Your Eyes Healthy after Treatment for Childhood Cancer High doses of radiation to the brain, eye, or eye socket (orbit) during treatment for childhood cancer can have a long-lasting affect on the
Providing Regulatory Submissions In Electronic Format Standardized Study Data
 Providing Regulatory Submissions In Electronic Format Standardized Study Data Guidance for Industry U.S. Department of Health and Human Services Food and Drug Administration Center for Drug Evaluation
Providing Regulatory Submissions In Electronic Format Standardized Study Data Guidance for Industry U.S. Department of Health and Human Services Food and Drug Administration Center for Drug Evaluation
August 18, 2015. Re: Section 1201 Rulemaking Proposed Exemption for Medical Devices
 DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service Food and Drug Administration 10903 New Hampshire Avenue Silver Spring, MD 20993 August 18, 2015 Ms. Jacqueline C. Charlesworth General Counsel
DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service Food and Drug Administration 10903 New Hampshire Avenue Silver Spring, MD 20993 August 18, 2015 Ms. Jacqueline C. Charlesworth General Counsel
Plamena Entcheva-Dimitrov, PhD, RAC On-line Course. www.preferredregulatoryconsulting.com 1
 Plamena Entcheva-Dimitrov, PhD, RAC On-line Course 1 The Preferred Regulatory Consulting content in this presentation is copyright protected under United States law and applicable international copyright
Plamena Entcheva-Dimitrov, PhD, RAC On-line Course 1 The Preferred Regulatory Consulting content in this presentation is copyright protected under United States law and applicable international copyright
May 5, 2015. Dear Mr. Courtney:
 DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service Food and Drug Administration 10903 New Hampshire Avenue Document Control Center WO66-G609 Silver Spring, MD 20993-0002 May 5, 2015 Terumo Cardiovascular
DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service Food and Drug Administration 10903 New Hampshire Avenue Document Control Center WO66-G609 Silver Spring, MD 20993-0002 May 5, 2015 Terumo Cardiovascular
Types of surgery for kidney cancer
 Useful information for cancer patients Contents This information is about the different operations that you may have for kidney cancer. Surgery can be used to treat almost any stage of kidney cancer. There
Useful information for cancer patients Contents This information is about the different operations that you may have for kidney cancer. Surgery can be used to treat almost any stage of kidney cancer. There
Guidance for Industry
 Guidance for Industry FDA Export Certificates Submit comments and suggestions regarding this document at anytime to Dockets Management Branch (HFA-305), Food and Drug Administration, 5630 Fishers Lane,
Guidance for Industry FDA Export Certificates Submit comments and suggestions regarding this document at anytime to Dockets Management Branch (HFA-305), Food and Drug Administration, 5630 Fishers Lane,
Breakthrough Therapy Program U.S. Food and Drug Administration (FDA)
 Breakthrough Therapy Program U.S. Food and Drug Administration (FDA) Presentation before the European Commission Expert Group on Safe and Timely Access to Medicines for Patients (STAMP) Jarilyn Dupont,
Breakthrough Therapy Program U.S. Food and Drug Administration (FDA) Presentation before the European Commission Expert Group on Safe and Timely Access to Medicines for Patients (STAMP) Jarilyn Dupont,
BASIC STANDARDS FOR RESIDENCY TRAINING IN CARDIOLOGY
 BASIC STANDARDS FOR RESIDENCY TRAINING IN CARDIOLOGY American Osteopathic Association and the American College of Osteopathic Internists Specific Requirements For Osteopathic Subspecialty Training In Cardiology
BASIC STANDARDS FOR RESIDENCY TRAINING IN CARDIOLOGY American Osteopathic Association and the American College of Osteopathic Internists Specific Requirements For Osteopathic Subspecialty Training In Cardiology
Surgical Treatment of Obesity: A Surgeon s View
 Surgical Treatment of Obesity: A Surgeon s View Jenny J. Choi, MD Director of Bariatrics Associate Director of Clinical Affairs Assistant Professor of Surgery Albert Einstein School of Medicine Montefiore
Surgical Treatment of Obesity: A Surgeon s View Jenny J. Choi, MD Director of Bariatrics Associate Director of Clinical Affairs Assistant Professor of Surgery Albert Einstein School of Medicine Montefiore
Disruptive Innovation in Health Care: Identifying Areas of Future Growth
 Disruptive Innovation in Health Care: Identifying Areas of Future Growth Jason Hwang, M.D., M.B.A. Executive Director, Healthcare jhwang@innosightinstitute.org The Disruptive Innovation Model Incumbents
Disruptive Innovation in Health Care: Identifying Areas of Future Growth Jason Hwang, M.D., M.B.A. Executive Director, Healthcare jhwang@innosightinstitute.org The Disruptive Innovation Model Incumbents
Imaging of Thoracic Endovascular Stent-Grafts
 Imaging of Thoracic Endovascular Stent-Grafts Tariq Hameed, M.D. Department of Radiology and Imaging Sciences, Indiana University School of Medicine, Indianapolis, Indiana Disclosures: No relevant financial
Imaging of Thoracic Endovascular Stent-Grafts Tariq Hameed, M.D. Department of Radiology and Imaging Sciences, Indiana University School of Medicine, Indianapolis, Indiana Disclosures: No relevant financial
Food and Drug Administration COMMISSIONER S. Fellowship Program. Protecting Public Health Through Science and Regulation
 DEPARTMENT OF HEALTH & HUMAN SERVICES USA Food and Drug Administration COMMISSIONER S Fellowship Program Protecting Public Health Through Science and Regulation FDA MISSION The FDA is responsible for protecting
DEPARTMENT OF HEALTH & HUMAN SERVICES USA Food and Drug Administration COMMISSIONER S Fellowship Program Protecting Public Health Through Science and Regulation FDA MISSION The FDA is responsible for protecting
Guidance for Cardiopulmonary Bypass Arterial Line Blood Filter 510(k) Submissions
 Guidance for Industry and FDA Guidance for Cardiopulmonary Bypass Arterial Line Blood Filter 510(k) Submissions Document issued on: February 21,200O US Department Of Health and Human Services Food and
Guidance for Industry and FDA Guidance for Cardiopulmonary Bypass Arterial Line Blood Filter 510(k) Submissions Document issued on: February 21,200O US Department Of Health and Human Services Food and
Guidance for Industry and FDA Staff: Current Good Manufacturing Practice Requirements for Combination Products
 Reprinted from FDA s website by EAS Consulting Group, LLC Guidance for Industry and FDA Staff: Current Good Manufacturing Practice Requirements for Combination Products DRAFT GUIDANCE This guidance document
Reprinted from FDA s website by EAS Consulting Group, LLC Guidance for Industry and FDA Staff: Current Good Manufacturing Practice Requirements for Combination Products DRAFT GUIDANCE This guidance document
The Comprehensive Cancer Care Team at Penn Vet s Ryan Hospital CORE TEAM
 The Comprehensive Cancer Care Team at Penn Vet s Ryan Hospital Ryan Hospital s Comprehensive Cancer Care Program is led by a world- class team of board- certified veterinarians who each offer a unique
The Comprehensive Cancer Care Team at Penn Vet s Ryan Hospital Ryan Hospital s Comprehensive Cancer Care Program is led by a world- class team of board- certified veterinarians who each offer a unique
January 12, 2016. Dear Amy Yang:
 DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service Food and Drug Administration 10903 New Hampshire Avenue Document Control Center - WO66-G609 Silver Spring, MD 20993-0002 January 12, 2016 Amy
DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service Food and Drug Administration 10903 New Hampshire Avenue Document Control Center - WO66-G609 Silver Spring, MD 20993-0002 January 12, 2016 Amy
Food and Drug Administration
 Center for Drug Evaluation and Research (CDER) Small Business Assistance Program The Information Source for Regulated Domestic and International Small Pharmaceutical Business CAPT Brenda Stodart, USPHS
Center for Drug Evaluation and Research (CDER) Small Business Assistance Program The Information Source for Regulated Domestic and International Small Pharmaceutical Business CAPT Brenda Stodart, USPHS
New Chemical Entity Exclusivity Determinations for Certain Fixed- Combination Drug Products
 New Chemical Entity Exclusivity Determinations for Certain Fixed- Combination Drug Products Guidance for Industry U.S. Department of Health and Human Services Food and Drug Administration Center for Drug
New Chemical Entity Exclusivity Determinations for Certain Fixed- Combination Drug Products Guidance for Industry U.S. Department of Health and Human Services Food and Drug Administration Center for Drug
Elenco dei periodici elettronici in Ovid Full text
 Academic Medicine Addictive Disorders & Their Treatment Advances in Anatomic Pathology Age & Ageing AIDS AIDS Patient Care & Stds AJN, American Journal of Nursing Alzheimer Disease & Associated Disorders
Academic Medicine Addictive Disorders & Their Treatment Advances in Anatomic Pathology Age & Ageing AIDS AIDS Patient Care & Stds AJN, American Journal of Nursing Alzheimer Disease & Associated Disorders
MViAKIO. V.A hn2 ATTACHMENT I 610(K) SUMMARY. Submitter:
 MViAKIO SURGICGAL CCWRP2 V.A hn2 ATTACHMENT I 610(K) SUMMARY Submitter: MAKO Surgical Corp. Address: 2555 Davie Road, Fort Lauderdale, FL, 33317 Phone number: 954-927-2044 x. 605 Fax number: 954-927-0446
MViAKIO SURGICGAL CCWRP2 V.A hn2 ATTACHMENT I 610(K) SUMMARY Submitter: MAKO Surgical Corp. Address: 2555 Davie Road, Fort Lauderdale, FL, 33317 Phone number: 954-927-2044 x. 605 Fax number: 954-927-0446
Guidance for Industry
 Guidance for Industry Providing Regulatory Submissions in Electronic Format Content of Labeling U.S. Department of Health and Human Services Food and Drug Administration Center for Drug Evaluation and
Guidance for Industry Providing Regulatory Submissions in Electronic Format Content of Labeling U.S. Department of Health and Human Services Food and Drug Administration Center for Drug Evaluation and
September 25, 2014. Dear Ms. McCoy:
 DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service Food and Drug Administration 10903 New Hampshire Avenue Document Control Center WO66-G609 Silver Spring, MD 20993-0002 September 25, 2014 Biomet
DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service Food and Drug Administration 10903 New Hampshire Avenue Document Control Center WO66-G609 Silver Spring, MD 20993-0002 September 25, 2014 Biomet
