Section 21 Ophthalmological Preparations. Latanoprost - Addition. Application submitted by International Council of Ophthalmology
|
|
|
- Ralph Sims
- 7 years ago
- Views:
Transcription
1 Section 21 Ophthalmological Preparations Latanoprost - Addition Application submitted by International Council of Ophthalmology
2 1. Summary statement of the proposal for inclusion, change or deletion We propose the inclusion of latanoprost, a prostaglandin F2α-analog that has been available for over a decade in most countries for the treatment of glaucoma. 1 It is one of the most potent and effective agents for the reduction of intraocular pressure currently available for the treatment of adult patients with glaucoma and ocular hypertension. 1-5 Latanoprost can be used as a first line therapy, but is also effective in reducing intraocular pressure in combination with other classes of eye medications as well as in cases where other medicines are not effective. 1-2 The March 2011 approval of generic latanoprost by the U.S. Food and Drug Administration and the consequent availability over generic latanoprost in other countries has significantly reduced the cost associated with the use of this drug and has made it a very practical option for reducing intraocular pressure in glaucoma and ocular hypertension patients in developing countries. 2. Name of the focal point in WHO submitting or supporting the application (where relevant) Ivo Kocur 3. Name of the organization(s) consulted and/or supporting the application International Council of Ophthalmology 4. International Nonproprietary Name (INN, generic name) of the medicine Latanoprost 5. Formulation proposed for inclusion; including adult and paediatric (if appropriate) Latanoprost 50mcg/ml 6. International availability - sources, if possible manufacturers and trade names Generic latanoprost is available in most countries, including the US, UK, France, Sweden, Ireland, Denmark, Germany, the Netherlands, Finland,Italy, Spain, Iceland, Switzerland, Hungary, Bulgaria, the Czech Republic and India. 7. Whether listing is requested as an individual medicine or as an example of a therapeutic group Individual medicine 8. Information supporting the public health relevance (epidemiological information on disease burden, assessment of current use, target population) Glaucoma is the second leading cause of blindness worldwide. Over 60 million people worldwide are affected by glaucoma, with 8.4 million of them being bilaterally blind. 6 By 2020, it is anticipated that nearly 80 million people will be affected by glaucoma. 7
3 In clinical practice, many patients are first treated using eye drops, most often using a prostaglandin analogue such as the proposed drug latanoprost or β-adrenergic antagonists. Pressure lowering is documented to slow the rate of glaucoma progression by half, and if the elevated IOP is left untreated in low and middle income countries, it may lead to severe vision loss. 9. Treatment details (dosage regimen, duration; reference to existing WHO and other clinical guidelines; need for special diagnostics, treatment or monitoring facilities and skills) Generic latanoprost is an eye drop that needs to be administered once a day, preferably in the evening. 10. Summary of comparative effectiveness in a variety of clinical settings: The articles discussed here were chosen because they summarize several notable clinical trials conducted involving latanoprost. A pooled data analysis of three phase III clinical trials conducted in Scandinavia, the UK and the United States showed that latanoprost was able to significantly reduce the mean intraocular pressure in a statistically significantly reduced from the untreated baseline. 2 In the 493 patients who were treated for 6 months, latanoprost reduced mean intraocular pressure 8.2 mm Hg and 8.6 mmhg after 0.5 and 6 months of treatment, respectively, from an untreated baseline of 25.8 mm Hg. In the 113 patients who were treated for 2 years, latanoprost reduced mean IOP 8.9 mm Hg 24 months after treatment. 2 Morning intraocular pressure at untreated baseline and after 0.5 to 24 months of treatment with latanoprost (mean ± 95% confidence interval, n = number of patients) The intraocular pressure was reduced 15% or more in 93%, 90%, 90%, and 84% of the total number of patients treated with latanoprost for 6, 12, 18, and 24 months, respectively. 2 A morning intraocular pressure reduction of about 32% was reached by 50% of the patients treated with latanoprost for 6, 12, 18, and 24 months, respectively. 2
4 Percent patients who reached specific morning intraocular pressure reduction levels (mm Hg) from untreated baseline after 6, 12, 18, and 24 months of latanoprost treatment. For patients who were withdrawn due to insufficient intraocular pressure reduction before specified visits, the last intraocular pressure value on latanoprost was carried forward (n = number of patients). No significant increase in intraocular pressure between the 0.5 month time point and the 24 month time point was observed, suggesting that the efficacy of the latanoprost did not decrease over time. Only 6% of the participants were deemed to have an insufficient intraocular pressure reduction as per the investigator (intraocular pressure treatment failure). 2 The overall risk of IOP treatment failure on latanoprost was 8%.
5 Life-table survival function of the risk of treatment failure on latanoprost due to insufficient IOP reduction and of withdrawal due to adverse event (n = number of patients at risk per month). Timolol, the leading topical beta-adrenergic antagonist, is often used as a first line therapy for the treatment of glaucoma. Dorzolamide, the first topical carbonic anhydrase inhibitor to become available on the market, is often prescribed as an add-on therapy. A review of studies comparing the efficacy of latanoprost to combined timolol and dorzolamide suggested that the intraocular pressure lowering effect of latanoprost is equivalent to that of concomitant timololdorzolamide therapy. In addition, data suggests that adding latanoprost to timolol and dorzolamide leads to a further 16% reduction of intraocular pressure. 1
6 Meta-analysis of five European studies comparing latanoprost monotherapy with timolol and dorzolamide. Comparison of the IOP reduction in mm Hg at 3 months between treatment groups (mean ± SEM). Percentage of patients who reached a specific reduction in diurnal IOP at 3 months. 11. Summary of comparative evidence on safety*: Estimate of total patient exposure to date Description of adverse effects/reactions Identification of variation in safety due to health systems and patient factors Summary of comparative safety against comparators Branded Xalatan was the most commonly prescribed glaucoma medication for over a decade and millions of people have been exposed to the drug globally. Almost all side effects are local
7 to the eye and these include conjunctival hyperemia which is almost never severe, iris color change for persons with light colored irides, changes in skin pigmentation, and growth of eyelashes. Ocular irritation also occurs. No systemic side effects are known to be caused by latanaprost (although patient reports occur in clinician studies, these are typically no greater than those reported as a background rate in the study population). A pooled data analysis from two trials showed that the risk of withdrawal from latanoprost before 24 months of treatment as a result of an adverse event was 20%. 2 However, in the clinical trials, this included the risk of developing an increase in iris pigmentation. Only 3% of patients were withdrawn due to ocular adverse events other than increased iris pigmentation and 3% due to non-ocular adverse events. 2 A detailed breakdown among 255 patients of withdrawals from latanoprost due to adverse events in one of the trials is as below. 8 Reasons for withdrawals during 2 years treatment with latanoprost. Patients withdrawn from Group B during the double-masked period then they were on timolol are not included. In a survey of literature comparing latanoprost and combined dorzolamide and timolol treatment, the overall safety profile of latanoprost was found to be as good as that of combined timolol and dorzolamide. 1 Adverse event Latanoprost n = 116 Timolol and dorzolamide n = 112 Ocular Eye irritation 27 55
8 Adverse event Latanoprost n = 116 Timolol and dorzolamide n = 112 Hyperaemia 13 6 Conjunctival disorders 10 5 Blurred vision 3 2 Increased IOP 3 Epiphora/photophobia 3 2 Blepharitis 2 4 Corneal disorders 2 2 Uveitis 1 Increased iris pigmentation 1 Dry eyes 1 Eye pain 3 Other 12 2 Non-ocular Respiratory disorders 7 4 Skin disorders 3 2 Influenza-like symptoms 4 3 Metabolic disorder 3 Cardiovascular disorder 3 Headache 3 5 Pharyngitis 3 1 Psychiatric disorder 3 1 Gastro-intestinal disorder 1 2 Musculo-skeletal disorder 1 3 Taste perversion 6 Other 7 4 Adverse Events Reported in the Meta-Analysis of Latanoprost Versus Timolol and Dorzolamide 12. Summary of available data on comparative cost** and cost-effectiveness within the pharmacological class or therapeutic group: range of costs of the proposed medicine comparative cost-effectiveness presented as range of cost per routine outcome (e.g. cost per case, cost per cure, cost per month of treatment, cost per case prevented, cost per clinical event prevented, or, if possible and relevant, cost per quality adjusted life year gained) Since latanoprost recently became a generic, the previous data on comparative cost and costeffectiveness is not valid. Generic latanoprost has been available in India for many years and the cost there is about two dollars per bottle (which lasts a month). Pressure lowering is documented to slow the rate of glaucoma progression by half, so widespread use of latanoprost should be able to delay or prevent vision loss in the vast majority of those who have glaucoma. The table below containing the approximate monthly cost of some of the most frequently used drugs for glaucoma treatment in the US suggests that latanoprost is an extremely cost-effective
9 option, especially when its efficacy and its ability to be used as both a first-line therapy and as an add-on drug are taken into account. Using GoodRx, a service that compares prices across nationwide pharmacy chains in the US: Timolol 3 Latanoprost 13 Acetazolamide 13 Dorzolamide (generic Trusopt) 15 Methazolamide 30 Azopt 38 Alphagan P 84 Combigan 87 Travatan Z 96 Acetazolamide ER (generic Diamox Sequels) 101 Xalatan 102 Lumigan 107 Price shown is the lowest price at select nationwide pharmacy chains for a 30-day supply of most-prescribed strength 13. Summary of regulatory status of the medicine (in country of origin, and preferably in other countries as well) Generic latanoprost was approved by the US Food and Drug Administration on March 22, 2011, and generic latanoprost is available in several places, including the US, Europe and India. 14. Availability of pharmacopoeial standards (British Pharmacopoeia, International Pharmacopoeia, United States Pharmacopoeia) Generic latanoprost met United States and Japanese pharmacopoeial standards. 15. Proposed (new/adapted) text for the WHO Model Formulary Latanoprost, a prostaglandin F2α-analog, has been available for several years in most countries for the treatment of glaucoma. It is one of the most potent and effective agents for the reduction of intraocular pressure currently available for the treatment of adult patients with glaucoma and ocular hypertension. From DailyMed: INDICATIONS AND USAGE Latanoprost ophthalmic solution is indicated for the reduction of elevated intraocular pressure in patients with open-angle glaucoma or ocular hypertension. HOW SUPPLIED Latanoprost Ophthalmic Solution is a sterile, clear, isotonic, buffered, preserved colorless solution of latanoprost 0.005% (50 mcg/ml).
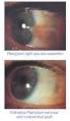
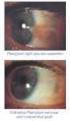
10 DOSAGE AND ADMINISTRATION The recommended dosage is one drop (1.5 mcg) in the affected eye(s) once daily in the evening. If one dose is missed, treatment should continue with the next dose as normal. The dosage of latanoprost ophthalmic solution should not exceed once daily; the combined use of two or more prostaglandins, or prostaglandin analogs including latanoprost ophthalmic solution is not recommended. It has been shown that administration of these prostaglandin drug products more than once daily may decrease the intraocular pressure lowering effect or cause paradoxical elevations in IOP. Reduction of the intraocular pressure starts approximately 3 to 4 hours after administration and the maximum effect is reached after 8 to 12 hours. Latanoprost ophthalmic solution may be used concomitantly with other topical ophthalmic drug products to lower intraocular pressure. If more than one topical ophthalmic drug is being used, the drugs should be administered at least five (5) minutes apart. CONTRAINDICATIONS Known hypersensitivity to latanoprost, benzalkonium chloride or any other ingredients in this product. PRECAUTIONS General Latanoprost ophthalmic solution may gradually increase the pigmentation of the iris. The eye color change is due to increased melanin content in the stromal melanocytes of the iris rather than to an increase in the number of melanocytes. This change may not be noticeable for several months to years. Typically, the brown pigmentation around the pupil spreads concentrically towards the periphery of the iris and the entire iris or parts of the iris become more brownish. Neither nevi nor freckles of the iris appear to be affected by treatment. While treatment with latanoprost can be continued in patients who develop noticeably increased iris pigmentation, these patients should be examined regularly. During clinical trials, the increase in brown iris pigment has not been shown to progress further upon discontinuation of treatment, but the resultant color change may be permanent. Eyelid skin darkening, which may be reversible, has been reported in association with the use of latanoprost. Latanoprost may gradually change eyelashes and vellus hair in the treated eye; these changes include increased length, thickness, pigmentation, the number of lashes or hairs and misdirected growth of eyelashes. Eyelash changes are usually reversible upon discontinuation of treatment.
11 Latanoprost should be used with caution in patients with a history of intraocular inflammation (iritis/uveitis) and should generally not be used in patients with active intraocular inflammation. Macular edema, including cystoid macular edema, has been reported during treatment with latanoprost. These reports have mainly occurred in aphakic patients, in pseudophakic patients with a torn posterior lens capsule or in patients with known risk factors for macular edema. Latanoprost should be used with caution in patients who do not have an intact posterior capsule or who have known risk factors for macular edema. There is limited experience with latanoprost in the treatment of angle closure, inflammatory or neovascular glaucoma. There have been reports of bacterial keratitis associated with the use of multiple-dose containers of topical ophthalmic products. These containers had been inadvertently contaminated by patients who, in most cases, had a concurrent corneal disease or a disruption of the ocular epithelial surface. Contact lenses should be removed prior to the administration of latanoprost and may be reinserted 15 minutes after administration. Information for Patients Patients should be advised about the potential for increased brown pigmentation of the iris, which may be permanent. Patients should also be informed about the possibility of eyelid skin darkening, which may be reversible after discontinuation of latanoprost. Patients should also be informed of the possibility of eyelash and vellus hair changes in the treated eye during treatment with latanoprost. These changes may result in a disparity between eyes in length, thickness, pigmentation, number of eyelashes or vellus hairs and/or direction of eyelash growth. Eyelash changes are usually reversible upon discontinuation of treatment. Patients should be instructed to avoid allowing the tip of the dispensing container to contact the eye or surrounding structures because this could cause the tip to become contaminated by common bacteria known to cause ocular infections. Serious damage to the eye and subsequent loss of vision may result from using contaminated solutions. Patients also should be advised that if they develop an intercurrent ocular condition (e.g., trauma or infection) or have ocular surgery, they should immediately seek their physician s advice concerning the continued use of the multiple-dose container. Patients should be advised that if they develop any ocular reactions, particularly conjunctivitis and lid reactions, they should immediately seek their physician s advice. Patients should also be advised that latanoprost contains benzalkonium chloride, which may be absorbed by contact lenses. Contact lenses should be removed prior to administration of the solution. Lenses may be reinserted 15 minutes following administration of latanoprost.
12 If more than one topical ophthalmic drug is being used, the drugs should be administered at least five (5) minutes apart. Drug Interactions In vitro studies have shown that precipitation occurs when eye drops containing thimerosal are mixed with latanoprost. If such drugs are used they should be administered at least five (5) minutes apart. Carcinogenesis, Mutagenesis, Impairment of Fertility Latanoprost was not mutagenic in bacteria, in mouse lymphoma or in mouse micronucleus tests. Chromosome aberrations were observed in vitro with human lymphocytes. Latanoprost was not carcinogenic in either mice or rats when administered by oral gavage at doses of up to 170 mcg/kg/day (approximately 2,800 times the recommended maximum human dose) for up to 20 and 24 months, respectively. Additional in vitro and in vivo studies on unscheduled DNA synthesis in rats were negative. Latanoprost has not been found to have any effect on male or female fertility in animal studies. Pregnancy Teratogenic Effects Pregnancy Category C Reproduction studies have been performed in rats and rabbits. In rabbits an incidence of 4 of 16 dams had no viable fetuses at a dose that was approximately 80 times the maximum human dose and the highest nonembryocidal dose in rabbits was approximately 15 times the maximum human dose. There are no adequate and well controlled studies in pregnant women. Latanoprost should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus. Nursing Mothers It is not known whether this drug or its metabolites are excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when latanoprost is administered to a nursing woman. Pediatric Use Safety and effectiveness in pediatric patients have not been established. Geriatric Use
13 No overall differences in safety or effectiveness have been observed between elderly and younger patients. ADVERSE REACTIONS Eyelash changes (increased length, thickness, pigmentation and number of lashes); eyelid skin darkening; intraocular inflammation (iritis/uveitis); iris pigmentation changes; and macular edema, including cystoid macular edema Controlled Clinical Trials The ocular adverse events and ocular signs and symptoms reported in 5% to 15% of the patients on latanoprost ophthalmic solution in the three 6-month, multicenter, double-masked, active-controlled trials were blurred vision, burning and stinging, conjunctival hyperemia, foreign body sensation, itching, increased pigmentation of the iris and punctuate epithelial keratopathy. Local conjunctival hyperemia was observed; however, less than 1% of the patients treated with latanoprost required discontinuation of therapy because of intolerance to conjunctival hyperemia. In addition to the above listed ocular events/signs and symptoms, the following were reported in 1% to 4% of the patients: dry eye, excessive tearing, eye pain, lid crusting, lid discomfort/pain, lid edema, lid erythema and photophobia. The following events were reported in less than 1% of the patients: conjunctivitis, diplopia and discharge from the eye. During clinical studies, there were extremely rare reports of the following: retinal artery embolus, retinal detachment and vitreous hemorrhage from diabetic retinopathy. The most common systemic adverse events seen with latanoprost were upper respiratory tract infection/cold/flu, which occurred at a rate of approximately 4%. Chest pain/angina pectoris, muscle/joint/back pain and rash/allergic skin reaction each occurred at a rate of 1% to 2%. Clinical Practice The following events have been identified during post-marketing use of latanoprost in clinical practice. Because they are reported voluntarily from a population of unknown size, estimates of frequency cannot be made. The events, which have been chosen for inclusion due to either their seriousness, frequency of reporting, possible causal connection to latanoprost or a combination of these factors, include: asthma and exacerbation of asthma; corneal edema and erosions; dyspnea; eyelash and vellus hair changes (increased length, thickness, pigmentation and number); eyelid skin darkening; herpes keratitis; intraocular inflammation (iritis/uveitis); keratitis; macular edema, including cystoid macular edema; misdirected eyelashes sometimes resulting in eye irritation; dizziness; headache; and toxic epidermal necrolysis.
14 References 1. Bron, Alain M., and Karl-Heinz Emmerich. "Latanoprost versus combined timolol and dorzolamide." Survey of ophthalmology 47 (2002): S Hedman, Katarina, Peter G. Watson, and Albert Alm. "The effect of latanoprost on intraocular pressure during 2 years of treatment." Survey of ophthalmology 47 (2002): S65-S Alm, Albert, and Johan Stjernschantz. "Effects on intraocular pressure and side effects of 0.005% latanoprost applied once daily, evening or morning. A comparison with timolol. Scandinavian Latanoprost Study Group."Ophthalmology (1995): Camras, Carl B. "Comparison of latanoprost and timolol in patients with ocular hypertension and glaucoma: a six-month masked, multicenter trial in the United States. The United States Latanoprost Study Group." Ophthalmology (1996): Lusky, Moshe, et al. "A comparative study of two dose regimens of latanoprost in patients with elevated intraocular pressure." Ophthalmology (1997): Quigley, Harry A. "Glaucoma." Lancet (2011): Quigley, Harry A., & Broman, Aimee T. "The number of people with glaucoma worldwide in 2010 and 2020." The British Journal of Ophthalmology 90.3 (2006): Alm, Albert, and Ingmar Widengård. "Latanoprost: Experience of 2 year treatment in Scandinavia." Acta Ophthalmologica Scandinavica 78.1 (2000):
Revised: 9/2014. LUMIGAN (bimatoprost ophthalmic solution) 0.01% for topical ophthalmic use. Initial U.S. Approval: 2001
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use LUMIGAN (bimatoprost ophthalmic solution) 0.01% safely and effectively. See full prescribing information
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use LUMIGAN (bimatoprost ophthalmic solution) 0.01% safely and effectively. See full prescribing information
Keep Up With Your Treatment and Keep Track of Your Eye Pressure
 Keep Up With Your Treatment and Keep Track of Your Eye Pressure Get into the habit of applying your XAATAN (latanoprost ophthalmic solution) eye drops at the same time every day DAY 1 DAY 2 DAY 3 DAY 4
Keep Up With Your Treatment and Keep Track of Your Eye Pressure Get into the habit of applying your XAATAN (latanoprost ophthalmic solution) eye drops at the same time every day DAY 1 DAY 2 DAY 3 DAY 4
Revised: 9/2014 HIGHLIGHTS OF PRESCRIBING INFORMATION
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use LATISSE safely and effectively. See full prescribing information for LATISSE. LATISSE (bimatoprost
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use LATISSE safely and effectively. See full prescribing information for LATISSE. LATISSE (bimatoprost
DO YOU HAVE ITCHY ALLERGY EYES? Find out about lasting relief
 DO YOU HAVE ITCHY ALLERGY EYES? Find out about lasting relief Common causes of itching due to eye allergies include: Pollen from trees, grasses, and ragweed Dust mites Cat dander Approximately 10 million
DO YOU HAVE ITCHY ALLERGY EYES? Find out about lasting relief Common causes of itching due to eye allergies include: Pollen from trees, grasses, and ragweed Dust mites Cat dander Approximately 10 million
Treatments for Open-Angle Glaucoma. A Review of the Research for Adults
 Treatments for Open-Angle Glaucoma A Review of the Research for Adults Is This Information Right for Me? Yes, this information is for you if: Your eye doctor has said that you have open-angle glaucoma,
Treatments for Open-Angle Glaucoma A Review of the Research for Adults Is This Information Right for Me? Yes, this information is for you if: Your eye doctor has said that you have open-angle glaucoma,
Re: LUMIGAN 0.03% (bimatoprost ophthalmic solution 0.03%) and LUMIGAN 0.01% (bimatoprost ophthalmic solution 0.01%)
 Allergan Ltd, 1st Floor, Marlow International, Parkway, Marlow, Bucks SL7 1YL Tel: (01628) 494444 Facsimile: (01628) 494449 Ref No: UK15-000677 Mandeep Allingham NW Surrey CCG February 18, 2015 Dear Ms
Allergan Ltd, 1st Floor, Marlow International, Parkway, Marlow, Bucks SL7 1YL Tel: (01628) 494444 Facsimile: (01628) 494449 Ref No: UK15-000677 Mandeep Allingham NW Surrey CCG February 18, 2015 Dear Ms
NEW ZEALAND DATA SHEET NAPHCON-A Naphazoline hydrochloride and pheniramine maleate.
 NEW ZEALAND DATA SHEET NAPHCON-A Naphazoline hydrochloride and pheniramine maleate. PRESENTATION Eye Drops: NAPHCON-A Eye Drops are a combination of an antihistamine (pheniramine maleate) and a decongestant
NEW ZEALAND DATA SHEET NAPHCON-A Naphazoline hydrochloride and pheniramine maleate. PRESENTATION Eye Drops: NAPHCON-A Eye Drops are a combination of an antihistamine (pheniramine maleate) and a decongestant
ZOVIRAX Cold Sore Cream
 Data Sheet ZOVIRAX Cold Sore Cream Aciclovir 5% w/w Presentation Topical cream Indications ZOVIRAX Cold Sore Cream is indicated for the treatment of Herpes simplex virus infections of the lips and face
Data Sheet ZOVIRAX Cold Sore Cream Aciclovir 5% w/w Presentation Topical cream Indications ZOVIRAX Cold Sore Cream is indicated for the treatment of Herpes simplex virus infections of the lips and face
See 17 for PATIENT COUNSELING INFORMATION. Revised: 3/2016 FULL PRESCRIBING INFORMATION: CONTENTS* WARNING: ADRENAL CRISIS IN THE SETTING OF SHOCK OR
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use LYSODREN safely and effectively. See full prescribing information for LYSODREN. LYSODREN (mitotane)
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use LYSODREN safely and effectively. See full prescribing information for LYSODREN. LYSODREN (mitotane)
DOSAGE FORMS AND STRENGTHS Intravitreal implant containing dexamethasone 0.7 mg in the NOVADUR solid polymer drug delivery system.
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use OZURDEX safely and effectively. See full prescribing information for OZURDEX. OZURDEX (dexamethasone
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use OZURDEX safely and effectively. See full prescribing information for OZURDEX. OZURDEX (dexamethasone
FULL PRESCRIBING INFORMATION: CONTENTS*
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use PHOTREXA VISCOUS and PHOTREXA safely and effectively. See full prescribing information for PHOTREXA
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use PHOTREXA VISCOUS and PHOTREXA safely and effectively. See full prescribing information for PHOTREXA
Learn More About Product Labeling
 Learn More About Product Labeling Product label The product label is developed during the formal process of review and approval by regulatory agencies of any medicine or medical product. There are specific
Learn More About Product Labeling Product label The product label is developed during the formal process of review and approval by regulatory agencies of any medicine or medical product. There are specific
Package leaflet: Information for the patient. Dorzolamide Mylan 20 mg/ml Eye drops, Solution Dorzolamide
 Package leaflet: Information for the patient Dorzolamide Mylan 20 mg/ml Eye drops, Solution Dorzolamide Read all of this leaflet carefully before you start using this medicine because it contains important
Package leaflet: Information for the patient Dorzolamide Mylan 20 mg/ml Eye drops, Solution Dorzolamide Read all of this leaflet carefully before you start using this medicine because it contains important
EXPANDING YOUR HORIZONS
 Glaucoma Therapy EXPANDING YOUR HORIZONS Learn how to control glaucoma with SLT therapy. SLTPATIENT EDUCATION PROGRAM Glaucoma Glaucoma is a degenerative disease that if left untreated can cause permanent
Glaucoma Therapy EXPANDING YOUR HORIZONS Learn how to control glaucoma with SLT therapy. SLTPATIENT EDUCATION PROGRAM Glaucoma Glaucoma is a degenerative disease that if left untreated can cause permanent
INITIATING ORAL AUBAGIO (teriflunomide) THERAPY
 FOR YOUR PATIENTS WITH RELAPSING FORMS OF MS INITIATING ORAL AUBAGIO (teriflunomide) THERAPY WARNING: HEPATOTOXICITY AND RISK OF TERATOGENICITY Severe liver injury including fatal liver failure has been
FOR YOUR PATIENTS WITH RELAPSING FORMS OF MS INITIATING ORAL AUBAGIO (teriflunomide) THERAPY WARNING: HEPATOTOXICITY AND RISK OF TERATOGENICITY Severe liver injury including fatal liver failure has been
How To Treat Eye Problems With A Laser
 1550 Oak St., Suite 5 1515 Oak St., St Eugene, OR 97401 Eugene, OR 97401 (541) 687-2110 (541) 344-2010 INFORMED CONSENT FOR PHOTOREFRACTIVE KERATECTOMY (PRK) This information is to help you make an informed
1550 Oak St., Suite 5 1515 Oak St., St Eugene, OR 97401 Eugene, OR 97401 (541) 687-2110 (541) 344-2010 INFORMED CONSENT FOR PHOTOREFRACTIVE KERATECTOMY (PRK) This information is to help you make an informed
VOLTAREN OPHTHA EYE DROPS
 פורמט עלון זה נקבע ע"ימשרד הבריאות ותוכנו נבדק ואושר על ידו באפריל 2008 PRESCRIBING INFORMATION VOLTAREN OPHTHA EYE DROPS 1 Name of the medicinal product Voltaren Ophtha Eye Drops. 2 Qualitative and quantitative
פורמט עלון זה נקבע ע"ימשרד הבריאות ותוכנו נבדק ואושר על ידו באפריל 2008 PRESCRIBING INFORMATION VOLTAREN OPHTHA EYE DROPS 1 Name of the medicinal product Voltaren Ophtha Eye Drops. 2 Qualitative and quantitative
Summary of the risk management plan (RMP) for Orkambi (lumacaftor and ivacaftor)
 EMA/662624/2015 Summary of the risk management plan (RMP) for Orkambi (lumacaftor and ivacaftor) This is a summary of the risk management plan (RMP) for Orkambi, which details the measures to be taken
EMA/662624/2015 Summary of the risk management plan (RMP) for Orkambi (lumacaftor and ivacaftor) This is a summary of the risk management plan (RMP) for Orkambi, which details the measures to be taken
GLAUCOMA. American Academy of Ophthalmology
 GLAUCOMA American Academy of Ophthalmology What is glaucoma? Glaucoma is a disease of the optic nerve, which is the part of the eye that carries the images we see to the brain. The optic nerve is made
GLAUCOMA American Academy of Ophthalmology What is glaucoma? Glaucoma is a disease of the optic nerve, which is the part of the eye that carries the images we see to the brain. The optic nerve is made
SUMMARY OF THE RISK MANAGEMENT PLAN (by medicinal product)
 PART VI SUMMARY OF THE RISK MANAGEMENT PLAN (by medicinal product) Format and content of the summary of the RMP The summary of the RMP part VI contains information based on RMP modules SI, SVIII and RMP
PART VI SUMMARY OF THE RISK MANAGEMENT PLAN (by medicinal product) Format and content of the summary of the RMP The summary of the RMP part VI contains information based on RMP modules SI, SVIII and RMP
11/11/2015. Glaucoma: Diagnosis and Treatment. Financial disclosures. Glaucoma: the problem. Joshua J. Ney, M.D. No disclosures to report
 Glaucoma: Diagnosis and Treatment Joshua J. Ney, M.D. No disclosures to report Financial disclosures Glaucoma: the problem Glaucoma is second most common cause of blindness worldwide 7 million people are
Glaucoma: Diagnosis and Treatment Joshua J. Ney, M.D. No disclosures to report Financial disclosures Glaucoma: the problem Glaucoma is second most common cause of blindness worldwide 7 million people are
Diabetes & blindness. due to DME BLINDNESS IN EUROPE
 Diabetes & blindness due to DME BLINDNESS IN EUROPE Blindness is a life-changing disability which puts a heavy strain on the daily lives of sufferers, their families, and society at large. Today, 284 million
Diabetes & blindness due to DME BLINDNESS IN EUROPE Blindness is a life-changing disability which puts a heavy strain on the daily lives of sufferers, their families, and society at large. Today, 284 million
There Are Millions of People at Risk For Dry Eye Who Is Likely to Develop This Irritating Condition?
 There Are Millions of People at Risk For Dry Eye Who Is Likely to Develop This Irritating Condition? Dry eye can be a temporary or chronic condition and occurs when the eye does not produce tears properly,
There Are Millions of People at Risk For Dry Eye Who Is Likely to Develop This Irritating Condition? Dry eye can be a temporary or chronic condition and occurs when the eye does not produce tears properly,
INFORMED CONSENT FOR PHOTOREFRACTIVE KERATECTOMY (PRK)
 INFORMED CONSENT FOR PHOTOREFRACTIVE KERATECTOMY (PRK) This information and the Patient Information booklet must be reviewed so you can make an informed decision regarding Photorefractive Keratectomy (PRK)
INFORMED CONSENT FOR PHOTOREFRACTIVE KERATECTOMY (PRK) This information and the Patient Information booklet must be reviewed so you can make an informed decision regarding Photorefractive Keratectomy (PRK)
VOLTAREN OPHTHA EYE DROPS
 2013 במאי משרד הבריאות ותוכנו נבדק ואושר על ידו ע"י עלון זה נקבע ע פורמט PRESCRIBING INFORMATION VOLTAREN OPHTHA EYE DROPS 1 Name of the medicinal product Voltaren Ophtha Eye Drops. 2 Qualitative and quantitative
2013 במאי משרד הבריאות ותוכנו נבדק ואושר על ידו ע"י עלון זה נקבע ע פורמט PRESCRIBING INFORMATION VOLTAREN OPHTHA EYE DROPS 1 Name of the medicinal product Voltaren Ophtha Eye Drops. 2 Qualitative and quantitative
Seeing Beyond the Symptoms
 Seeing Beyond the Symptoms Cataracts are one of the leading causes of vision impairment in the United States. 1 However, because cataracts form slowly and over a long period of time, many people suffer
Seeing Beyond the Symptoms Cataracts are one of the leading causes of vision impairment in the United States. 1 However, because cataracts form slowly and over a long period of time, many people suffer
FAQ. Possible Allergic Reaction
 FAQ Possible Allergic Reaction First, please be sure you are using RevitaLash as directed on the package insert. Remember: only a small quantity of RevitaLash is needed to enhance the beauty of your natural
FAQ Possible Allergic Reaction First, please be sure you are using RevitaLash as directed on the package insert. Remember: only a small quantity of RevitaLash is needed to enhance the beauty of your natural
1 Always test and record vision wearing distance spectacles test each eye separately A 1mm pinhole will improve acuity in refractive errors
 Golden eye rules Examination techniques 1 Always test and record vision wearing distance spectacles test each eye separately A 1mm pinhole will improve acuity in refractive errors Snellen chart (6 metre)
Golden eye rules Examination techniques 1 Always test and record vision wearing distance spectacles test each eye separately A 1mm pinhole will improve acuity in refractive errors Snellen chart (6 metre)
Sponsor Novartis Pharmaceuticals
 Clinical Trial Results Database Page 1 Sponsor Novartis Pharmaceuticals Generic Drug Name Indacaterol Therapeutic Area of Trial Chronic Obstructive Pulmonary Disease (COPD) Indication studied: COPD Study
Clinical Trial Results Database Page 1 Sponsor Novartis Pharmaceuticals Generic Drug Name Indacaterol Therapeutic Area of Trial Chronic Obstructive Pulmonary Disease (COPD) Indication studied: COPD Study
Eye and Vision Care in the Patient-Centered Medical Home
 1505 Prince Street, Alexandria, VA 22314 (703) 739-9200200 FAX: (703) 739-9497494 Eye and Vision Care in the Patient-Centered Medical Home The Patient Centered Medical Home (PCMH) is an approach to providing
1505 Prince Street, Alexandria, VA 22314 (703) 739-9200200 FAX: (703) 739-9497494 Eye and Vision Care in the Patient-Centered Medical Home The Patient Centered Medical Home (PCMH) is an approach to providing
SECONDARY GLAUCOMA: Pseudoexfoliation (PXF), Pigmentary Dispersion Syndrome (PDS), Neovascular (NV), Uveitic
 SECONDARY GLAUCOMA: Pseudoexfoliation (PXF), Pigmentary Dispersion Syndrome (PDS), Neovascular (NV), Uveitic Many roads can lead to glaucoma. With the exception of primary open-angle glaucoma (POAG) and
SECONDARY GLAUCOMA: Pseudoexfoliation (PXF), Pigmentary Dispersion Syndrome (PDS), Neovascular (NV), Uveitic Many roads can lead to glaucoma. With the exception of primary open-angle glaucoma (POAG) and
Eye Diseases. 1995-2014, The Patient Education Institute, Inc. www.x-plain.com otf30101 Last reviewed: 05/21/2014 1
 Eye Diseases Introduction Some eye problems are minor and fleeting. But some lead to a permanent loss of vision. There are many diseases that can affect the eyes. The symptoms of eye diseases vary widely,
Eye Diseases Introduction Some eye problems are minor and fleeting. But some lead to a permanent loss of vision. There are many diseases that can affect the eyes. The symptoms of eye diseases vary widely,
IR Conference Call on EYLEA (aflibercept) Injection in Diabetic Macular Edema. September 28, 2013
 IR Conference Call on EYLEA (aflibercept) Injection in Diabetic Macular Edema September 28, 2013 Safe Harbor Statement This presentation includes forward-looking statements that involve risks and uncertainties
IR Conference Call on EYLEA (aflibercept) Injection in Diabetic Macular Edema September 28, 2013 Safe Harbor Statement This presentation includes forward-looking statements that involve risks and uncertainties
Summary of the risk management plan (RMP) for Rasagiline ratiopharm (rasagiline)
 EMA/744222/2014 Summary of the risk management plan (RMP) for Rasagiline ratiopharm (rasagiline) This is a summary of the risk management plan (RMP) for Rasagiline ratiopharm, which details the measures
EMA/744222/2014 Summary of the risk management plan (RMP) for Rasagiline ratiopharm (rasagiline) This is a summary of the risk management plan (RMP) for Rasagiline ratiopharm, which details the measures
Summary of the risk management plan (RMP) for Ionsys (fentanyl)
 EMA/764409/2015 Summary of the risk management plan (RMP) for Ionsys (fentanyl) This is a summary of the risk management plan (RMP) for Ionsys, which details the measures to be taken in order to ensure
EMA/764409/2015 Summary of the risk management plan (RMP) for Ionsys (fentanyl) This is a summary of the risk management plan (RMP) for Ionsys, which details the measures to be taken in order to ensure
Eye Injuries. The Eyes The eyes are sophisticated organs. They collect light and focus it on the back of the eye, allowing us to see.
 Eye Injuries Introduction The design of your face helps protect your eyes from injury. But injuries can still damage your eyes. Sometimes injuries are severe enough that you could lose your vision. Most
Eye Injuries Introduction The design of your face helps protect your eyes from injury. But injuries can still damage your eyes. Sometimes injuries are severe enough that you could lose your vision. Most
REGISTRATION FORM PATIENT NAME: ADDRESS (STREET, CITY, STATE, ZIP): HOME PHONE: WORK PHONE: CELL PHONE: DATE OF BIRTH: / / AGE: SEX:
 REGISTRATION FORM PATIENT NAME: ADDRESS (STREET, CITY, STATE, ZIP): HOME PHONE: WORK PHONE: CELL PHONE: E-MAIL ADDRESS: OCCUPATION: DATE OF BIRTH: / / AGE: SEX: SOCIAL SECURITY NUMBER: MARITAL STATUS:
REGISTRATION FORM PATIENT NAME: ADDRESS (STREET, CITY, STATE, ZIP): HOME PHONE: WORK PHONE: CELL PHONE: E-MAIL ADDRESS: OCCUPATION: DATE OF BIRTH: / / AGE: SEX: SOCIAL SECURITY NUMBER: MARITAL STATUS:
PERIOCULAR (SUBTENON) STEROID INJECTION ERIC S. MANN M.D.,Ph.D.
 PERIOCULAR (SUBTENON) STEROID INJECTION ERIC S. MANN M.D.,Ph.D. A. INDICATIONS: Periocular steroid injection involves placement of steroid around the eye to treat intraocular inflammation or swelling of
PERIOCULAR (SUBTENON) STEROID INJECTION ERIC S. MANN M.D.,Ph.D. A. INDICATIONS: Periocular steroid injection involves placement of steroid around the eye to treat intraocular inflammation or swelling of
Public Assessment Report. Decentralised Procedure. Latanoprost 0.005%w/v Eye Drops, Solution
 Public Assessment Report Decentralised Procedure Latanoprost 0.005%w/v Eye Drops, Solution Latanoprost UK licence no: PL 27583/0098 Apotex Europe BV 1 Latanoprost 0.005% w/v Eye Drops, Solution PL 27583/0098
Public Assessment Report Decentralised Procedure Latanoprost 0.005%w/v Eye Drops, Solution Latanoprost UK licence no: PL 27583/0098 Apotex Europe BV 1 Latanoprost 0.005% w/v Eye Drops, Solution PL 27583/0098
Hypromellose Eye Drops BP 0.3% w/v PL 23097/0006
 Hypromellose Eye Drops BP 0.3% w/v PL 23097/0006 UKPAR TABLE OF CONTENTS Lay summary Page 2 Scientific discussion Page 3 Steps taken for assessment Page 9 Summary of product characteristics Page 10 Patient
Hypromellose Eye Drops BP 0.3% w/v PL 23097/0006 UKPAR TABLE OF CONTENTS Lay summary Page 2 Scientific discussion Page 3 Steps taken for assessment Page 9 Summary of product characteristics Page 10 Patient
Congratulations! You have just joined the thousands of people who are enjoying the benefits of laser vision correction.
 Dear Valued Patient, Thank you for choosing Shady Grove Ophthalmology for your laser vision correction procedure. Our excellent staff is committed to offering you the highest quality eye care using state
Dear Valued Patient, Thank you for choosing Shady Grove Ophthalmology for your laser vision correction procedure. Our excellent staff is committed to offering you the highest quality eye care using state
0.9% Sodium Chloride Injection, USP In VIAFLEX Plastic Container
 Page 1 of 8 PRESCRIBING INFORMATION 0.9% Sodium Chloride Injection, USP In VIAFLEX Plastic Container IV Fluid and Electrolyte Replenisher Baxter Corporation Mississauga, Ontario L5N 0C2 Canada Date of
Page 1 of 8 PRESCRIBING INFORMATION 0.9% Sodium Chloride Injection, USP In VIAFLEX Plastic Container IV Fluid and Electrolyte Replenisher Baxter Corporation Mississauga, Ontario L5N 0C2 Canada Date of
Adjunctive psychosocial intervention. Conditions requiring dose reduction. Immediate, peak plasma concentration is reached within 1 hour.
 Shared Care Guideline for Prescription and monitoring of Naltrexone Hydrochloride in alcohol dependence Author(s)/Originator(s): (please state author name and department) Dr Daly - Consultant Psychiatrist,
Shared Care Guideline for Prescription and monitoring of Naltrexone Hydrochloride in alcohol dependence Author(s)/Originator(s): (please state author name and department) Dr Daly - Consultant Psychiatrist,
A normal eye is protected by a layer of natural tears.
 A normal eye is protected by a layer of natural tears. If you need to use eye drops several times a day to make your eyes comfortably moist, you should consult your eye doctor. Helps increase tear production
A normal eye is protected by a layer of natural tears. If you need to use eye drops several times a day to make your eyes comfortably moist, you should consult your eye doctor. Helps increase tear production
Selected Requirements of Prescribing Information
 The Selected Requirements of Prescribing Information (SRPI) is a checklist of 42 important format prescribing information (PI) items based on labeling regulations [21 CFR 201.56(d) and 201.57] and guidances.
The Selected Requirements of Prescribing Information (SRPI) is a checklist of 42 important format prescribing information (PI) items based on labeling regulations [21 CFR 201.56(d) and 201.57] and guidances.
used to treat inflammation, corneal injury and bacterial infections in the external part of the eye.
 TOBRADEX * Eye Drops Tobramycin and Dexamethasone CONSUMER MEDICINE INFORMATION What is in this Leaflet This leaflet answers some common questions about TOBRADEX* Eye Drops. It does not contain all the
TOBRADEX * Eye Drops Tobramycin and Dexamethasone CONSUMER MEDICINE INFORMATION What is in this Leaflet This leaflet answers some common questions about TOBRADEX* Eye Drops. It does not contain all the
Nursing 113. Pharmacology Principles
 Nursing 113 Pharmacology Principles 1. The study of how drugs enter the body, reach the site of action, and are removed from the body is called a. pharmacotherapeutics b. pharmacology c. pharmacodynamics
Nursing 113 Pharmacology Principles 1. The study of how drugs enter the body, reach the site of action, and are removed from the body is called a. pharmacotherapeutics b. pharmacology c. pharmacodynamics
Medical Policy An independent licensee of the Blue Cross Blue Shield Association
 Afrezza Page 1 of 6 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: Afrezza (human insulin) Prime Therapeutics will review Prior Authorization requests Prior Authorization
Afrezza Page 1 of 6 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: Afrezza (human insulin) Prime Therapeutics will review Prior Authorization requests Prior Authorization
TABLE OF CONTENTS: LASER EYE SURGERY CONSENT FORM
 1 BoydVision TABLE OF CONTENTS: LASER EYE SURGERY CONSENT FORM Risks and Side Effects... 2 Risks Specific to PRK... 3 Risks Specific to LASIK... 4 Patient Statement of Consent... 5 Consent for Laser Eye
1 BoydVision TABLE OF CONTENTS: LASER EYE SURGERY CONSENT FORM Risks and Side Effects... 2 Risks Specific to PRK... 3 Risks Specific to LASIK... 4 Patient Statement of Consent... 5 Consent for Laser Eye
Explanation of the Procedure
 Informed Consent Cataract Surgery with Intraocular Lens Implant Please initial below indicating that you have read and understand each section Introduction The internal lens of the eye can become cloudy
Informed Consent Cataract Surgery with Intraocular Lens Implant Please initial below indicating that you have read and understand each section Introduction The internal lens of the eye can become cloudy
3% Sodium Chloride Injection, USP 5% Sodium Chloride Injection, USP
 PRESCRIBING INFORMATION 3% Sodium Chloride Injection, USP 5% Sodium Chloride Injection, USP IV Fluid and Electrolyte Replenisher Baxter Corporation Mississauga, Ontario L5N 0C2 Canada Date of Revision:
PRESCRIBING INFORMATION 3% Sodium Chloride Injection, USP 5% Sodium Chloride Injection, USP IV Fluid and Electrolyte Replenisher Baxter Corporation Mississauga, Ontario L5N 0C2 Canada Date of Revision:
Vitreo-Retinal and Macular Degeneration Frequently Asked Questions
 Vitreo-Retinal and Macular Degeneration Frequently Asked Questions What is a Vitreo-Retinal specialist? Retinal specialists are eye physicians and surgeons who focus on diseases in the back of the eye
Vitreo-Retinal and Macular Degeneration Frequently Asked Questions What is a Vitreo-Retinal specialist? Retinal specialists are eye physicians and surgeons who focus on diseases in the back of the eye
FOR ORAL OR RECTAL ADMINISTRATION FOR THE PREVENTION AND TREATMENT OF PORTAL-SYSTEMIC ENCEPHALOPATHY
 FORM NO. 1360 VC3074 Rev.2/08 LACTULOSE SOLUTION, USP 10 g/15 ml FOR ORAL OR RECTAL ADMINISTRATION FOR THE PREVENTION AND TREATMENT OF PORTAL-SYSTEMIC ENCEPHALOPATHY Rx Only DESCRIPTION Lactulose is a
FORM NO. 1360 VC3074 Rev.2/08 LACTULOSE SOLUTION, USP 10 g/15 ml FOR ORAL OR RECTAL ADMINISTRATION FOR THE PREVENTION AND TREATMENT OF PORTAL-SYSTEMIC ENCEPHALOPATHY Rx Only DESCRIPTION Lactulose is a
PHENYLEPHRINE HYDROCHLORIDE INJECTION USP
 PRESCRIBING INFORMATION PHENYLEPHRINE HYDROCHLORIDE INJECTION USP 10 mg/ml Sandoz Canada Inc. Date of Preparation: September 1992 145 Jules-Léger Date of Revision : January 13, 2011 Boucherville, QC, Canada
PRESCRIBING INFORMATION PHENYLEPHRINE HYDROCHLORIDE INJECTION USP 10 mg/ml Sandoz Canada Inc. Date of Preparation: September 1992 145 Jules-Léger Date of Revision : January 13, 2011 Boucherville, QC, Canada
ACTIVITY DISCLAIMER. Glaucoma DISCLOSURE. Learning Objectives. Audience Engagement System Step 1 Step 2 Step 3. Jeffrey SooHoo, MD. Jeffrey SooHoo, MD
 Glaucoma Jeffrey SooHoo, MD ACTIVITY DISCLAIMER The material presented here is being made available by the American Academy of Family Physicians for educational purposes only. This material is not intended
Glaucoma Jeffrey SooHoo, MD ACTIVITY DISCLAIMER The material presented here is being made available by the American Academy of Family Physicians for educational purposes only. This material is not intended
GENERAL CONSIDERATIONS FOR CLINICAL TRIALS E8
 INTERNATIONAL CONFERENCE ON HARMONISATION OF TECHNICAL REQUIREMENTS FOR REGISTRATION OF PHARMACEUTICALS FOR HUMAN USE ICH HARMONISED TRIPARTITE GUIDELINE GENERAL CONSIDERATIONS FOR CLINICAL TRIALS E8 Current
INTERNATIONAL CONFERENCE ON HARMONISATION OF TECHNICAL REQUIREMENTS FOR REGISTRATION OF PHARMACEUTICALS FOR HUMAN USE ICH HARMONISED TRIPARTITE GUIDELINE GENERAL CONSIDERATIONS FOR CLINICAL TRIALS E8 Current
Consent for Bilateral Simultaneous Refractive Surgery PRK
 Consent for Bilateral Simultaneous Refractive Surgery PRK Please sign and return Patient Copy While many patients choose to have both eyes treated at the same surgical setting, there may be risks associated
Consent for Bilateral Simultaneous Refractive Surgery PRK Please sign and return Patient Copy While many patients choose to have both eyes treated at the same surgical setting, there may be risks associated
Conjunctivitis - Pink Eye
 Conjunctivitis - Pink Eye Introduction Conjunctivitis is often called pink eye. It causes inflammation of the thin, clear lining inside the eyelid and on the white of the eye. The inflammation gives the
Conjunctivitis - Pink Eye Introduction Conjunctivitis is often called pink eye. It causes inflammation of the thin, clear lining inside the eyelid and on the white of the eye. The inflammation gives the
IMAGE ASSISTANT: OPHTHALMOLOGY
 IMAGE ASSISTANT: OPHTHALMOLOGY Summary: The Image Assistant has been developed to provide medical doctors with a software tool to search, display, edit and use medical illustrations of their own specialty,
IMAGE ASSISTANT: OPHTHALMOLOGY Summary: The Image Assistant has been developed to provide medical doctors with a software tool to search, display, edit and use medical illustrations of their own specialty,
For non-superficial eye injuries an individual may be considered an incident case only once per lifetime.
 1 OPHTH_15 EYE INJURIES Background This case definition was developed by the Armed Forces Health Surveillance Branch (AFHSB) and the Tri-Service Vision Conservation and Readiness Program (TSVCRP) at the
1 OPHTH_15 EYE INJURIES Background This case definition was developed by the Armed Forces Health Surveillance Branch (AFHSB) and the Tri-Service Vision Conservation and Readiness Program (TSVCRP) at the
Material Safety data sheet
 EMERGENCY OVERVIEW METFORMIN HYDROCHLORIDE TABLET contain Metformin and excipients generally considered to be non- toxic and non-hazardous in small quantities and under conditions of normal occupational
EMERGENCY OVERVIEW METFORMIN HYDROCHLORIDE TABLET contain Metformin and excipients generally considered to be non- toxic and non-hazardous in small quantities and under conditions of normal occupational
VISTARIL (hydroxyzine pamoate) Capsules and Oral Suspension
 VISTARIL (hydroxyzine pamoate) Capsules and Oral Suspension DESCRIPTION Hydroxyzine pamoate is designated chemically as 1-(p-chlorobenzhydryl) 4- [2-(2-hydroxyethoxy) ethyl] diethylenediamine salt of 1,1
VISTARIL (hydroxyzine pamoate) Capsules and Oral Suspension DESCRIPTION Hydroxyzine pamoate is designated chemically as 1-(p-chlorobenzhydryl) 4- [2-(2-hydroxyethoxy) ethyl] diethylenediamine salt of 1,1
Cataracts. Cataract and Primary Eye Care Service...215-928-3041. Main Number...215-928-3000. Physician Referral...1-877-AT-WILLS 1-877-289-4557
 Main Number...215-928-3000 Physician Referral...1-877-AT-WILLS 1-877-289-4557 Emergency Service...215-503-8080 Cataract and Primary Eye Care Service...215-928-3041 Retina Service... 215-928-3300 Cataract
Main Number...215-928-3000 Physician Referral...1-877-AT-WILLS 1-877-289-4557 Emergency Service...215-503-8080 Cataract and Primary Eye Care Service...215-928-3041 Retina Service... 215-928-3300 Cataract
Summary of the risk management plan (RMP) for Aripiprazole Pharmathen (aripiprazole)
 EMA/303592/2015 Summary of the risk management plan (RMP) for Aripiprazole Pharmathen (aripiprazole) This is a summary of the risk management plan (RMP) for Aripiprazole Pharmathen, which details the measures
EMA/303592/2015 Summary of the risk management plan (RMP) for Aripiprazole Pharmathen (aripiprazole) This is a summary of the risk management plan (RMP) for Aripiprazole Pharmathen, which details the measures
European Medicines Agency recommends restricting use of trimetazidine-containing medicines
 22 June 2012 EMA/CHMP/417861/2012 Press Office Press release European Medicines Agency recommends restricting use of trimetazidine-containing medicines Restricted indication for patients with stable angina
22 June 2012 EMA/CHMP/417861/2012 Press Office Press release European Medicines Agency recommends restricting use of trimetazidine-containing medicines Restricted indication for patients with stable angina
Chemical Name: (±)-9-Fluoro-2,3-dihydro-3-methyl-10-(4-methyl-1-piperazinyl)-7-oxo-7H-pyrido[1,2,3-de]-1,4-benzoxazine-6- carboxylic acid.
![Chemical Name: (±)-9-Fluoro-2,3-dihydro-3-methyl-10-(4-methyl-1-piperazinyl)-7-oxo-7H-pyrido[1,2,3-de]-1,4-benzoxazine-6- carboxylic acid. Chemical Name: (±)-9-Fluoro-2,3-dihydro-3-methyl-10-(4-methyl-1-piperazinyl)-7-oxo-7H-pyrido[1,2,3-de]-1,4-benzoxazine-6- carboxylic acid.](/thumbs/40/20977501.jpg) OCUFLOX (ofloxacin ophthalmic solution) 0.3% sterile DESCRIPTION OCUFLOX (ofloxacin ophthalmic solution) 0.3% is a sterile ophthalmic solution. It is a fluorinated carboxyquinolone anti-infective for topical
OCUFLOX (ofloxacin ophthalmic solution) 0.3% sterile DESCRIPTION OCUFLOX (ofloxacin ophthalmic solution) 0.3% is a sterile ophthalmic solution. It is a fluorinated carboxyquinolone anti-infective for topical
PRIORITY RESEARCH TOPICS
 PRIORITY RESEARCH TOPICS Understanding all the issues associated with antimicrobial resistance is probably impossible, but it is clear that there are a number of key issues about which we need more information.
PRIORITY RESEARCH TOPICS Understanding all the issues associated with antimicrobial resistance is probably impossible, but it is clear that there are a number of key issues about which we need more information.
Avastin (Bevacizumab) Intravitreal Injection
 Avastin (Bevacizumab) Intravitreal Injection This handout describes how Avastin may be used to treat wet age related macular degeneration (AMD) or macular edema due to retinal vascular disease such as
Avastin (Bevacizumab) Intravitreal Injection This handout describes how Avastin may be used to treat wet age related macular degeneration (AMD) or macular edema due to retinal vascular disease such as
Emory Eye Center New Patient Questionnaire
 Patient Name: Date: Current Address: Current Phone: Date of Birth: Primary Care Physician: Referring Physician: (First & Last Name) (First & Last Name) Pharmacy Name: Phone #: ( ) Please answer all questions
Patient Name: Date: Current Address: Current Phone: Date of Birth: Primary Care Physician: Referring Physician: (First & Last Name) (First & Last Name) Pharmacy Name: Phone #: ( ) Please answer all questions
MINISTRY OF HEALTH PANDEMIC INFLUENZA A / H1N1 2009 VACCINE FREQUENTLY ASKED QUESTIONS
 Government of the Republic of Trinidad and Tobago MINISTRY OF HEALTH PANDEMIC INFLUENZA A / H1N1 2009 VACCINE FREQUENTLY ASKED QUESTIONS Influenza vaccines are one of the most effective ways to protect
Government of the Republic of Trinidad and Tobago MINISTRY OF HEALTH PANDEMIC INFLUENZA A / H1N1 2009 VACCINE FREQUENTLY ASKED QUESTIONS Influenza vaccines are one of the most effective ways to protect
Topical Tacrolimus or Pimecrolimus for the treatment of mild, moderate or severe atopic eczema. Effective Shared Care Agreement
 Topical Tacrolimus or Pimecrolimus for the treatment of mild, moderate or severe atopic eczema. Effective Shared Care Agreement A Copy of this page signed by all three parties should be retained in the
Topical Tacrolimus or Pimecrolimus for the treatment of mild, moderate or severe atopic eczema. Effective Shared Care Agreement A Copy of this page signed by all three parties should be retained in the
White, circular, biconvex, uncoated tablets with a score line on one side, plain on the other.
 Nausicalm Cyclizine hydrochloride Ph. Eur. 50 mg Presentation White, circular, biconvex, uncoated tablets with a score line on one side, plain on the other. Uses Actions The active ingredient-cyclizine
Nausicalm Cyclizine hydrochloride Ph. Eur. 50 mg Presentation White, circular, biconvex, uncoated tablets with a score line on one side, plain on the other. Uses Actions The active ingredient-cyclizine
Health Professions Act BYLAWS SCHEDULE F. PART 3 Residential Care Facilities and Homes Standards of Practice. Table of Contents
 Health Professions Act BYLAWS SCHEDULE F PART 3 Residential Care Facilities and Homes Standards of Practice Table of Contents 1. Application 2. Definitions 3. Supervision of Pharmacy Services in a Facility
Health Professions Act BYLAWS SCHEDULE F PART 3 Residential Care Facilities and Homes Standards of Practice Table of Contents 1. Application 2. Definitions 3. Supervision of Pharmacy Services in a Facility
TALLAHASSEE EYE CENTER
 TALLAHASSEE EYE CENTER PATIENT INFORMATION Date: Name: Gender: M / F First MI Last Date of Birth: / / Address: City: State: ZIP: Phone Numbers: Home: Cellular: Work: E-Mail: SS#: - - What is the best way
TALLAHASSEE EYE CENTER PATIENT INFORMATION Date: Name: Gender: M / F First MI Last Date of Birth: / / Address: City: State: ZIP: Phone Numbers: Home: Cellular: Work: E-Mail: SS#: - - What is the best way
PATIENT HEALTH QUESTIONNAIRE Radiation Oncology (Patient Label)
 REVIEWED DATE / INITIALS SAFETY: Are you at risk for falls? Do you have a Pacemaker? Females; Is there a possibility you may be pregnant? ALLERGIES: Do you have any allergies to medications? If, please
REVIEWED DATE / INITIALS SAFETY: Are you at risk for falls? Do you have a Pacemaker? Females; Is there a possibility you may be pregnant? ALLERGIES: Do you have any allergies to medications? If, please
MOH CLINICAL PRACTICE GUIDELINES 2/2008 Prescribing of Benzodiazepines
 MOH CLINICL PRCTICE GUIELINES 2/2008 Prescribing of Benzodiazepines College of Family Physicians, Singapore cademy of Medicine, Singapore Executive summary of recommendations etails of recommendations
MOH CLINICL PRCTICE GUIELINES 2/2008 Prescribing of Benzodiazepines College of Family Physicians, Singapore cademy of Medicine, Singapore Executive summary of recommendations etails of recommendations
MECHANISM MEDICAL MANAGEMENT OF GLAUCOMA. CLASSIFICATION (Cholinergic) CLASSIFICATION 7/25/2014. Decreased aqueous production
 MEDICAL MANAGEMENT OF GLAUCOMA Dr. Liz Thomas Glaucoma Services Aravind Eye Hospital Madurai MECHANISM Decreased aqueous production Increased facility of outflow (trabecular / uveoscleral) Intraocular
MEDICAL MANAGEMENT OF GLAUCOMA Dr. Liz Thomas Glaucoma Services Aravind Eye Hospital Madurai MECHANISM Decreased aqueous production Increased facility of outflow (trabecular / uveoscleral) Intraocular
LATANOPROST/TIMOLOL SANDOZ Eye Drops Latanoprost /Timolol maleate Eye Drops
 LATANOPROST/TIMOLOL SANDOZ Eye Drops Latanoprost /Timolol maleate Eye Drops Consumer Medicine Information What is in this leaflet This leaflet answers some common questions about. It does not contain all
LATANOPROST/TIMOLOL SANDOZ Eye Drops Latanoprost /Timolol maleate Eye Drops Consumer Medicine Information What is in this leaflet This leaflet answers some common questions about. It does not contain all
IMPORTANT DRUG WARNING Regarding Mycophenolate-Containing Products
 Dear Healthcare Provider: Mycophenolate REMS (Risk Evaluation and Mitigation Strategy) has been mandated by the FDA (Food and Drug Administration) due to postmarketing reports showing that exposure to
Dear Healthcare Provider: Mycophenolate REMS (Risk Evaluation and Mitigation Strategy) has been mandated by the FDA (Food and Drug Administration) due to postmarketing reports showing that exposure to
EFFIMET 1000 XR Metformin Hydrochloride extended release tablet
 BRAND NAME: Effimet XR. THERAPEUTIC CATEGORY: Anti-Diabetic PHARMACOLOGIC CLASS: Biguanides EFFIMET 1000 XR Metformin Hydrochloride extended release tablet COMPOSITION AND PRESENTATION Composition Each
BRAND NAME: Effimet XR. THERAPEUTIC CATEGORY: Anti-Diabetic PHARMACOLOGIC CLASS: Biguanides EFFIMET 1000 XR Metformin Hydrochloride extended release tablet COMPOSITION AND PRESENTATION Composition Each
LEFLUNOMIDE (Adults)
 Shared Care Guideline DRUG: Introduction: LEFLUNOMIDE (Adults) Indication: Disease modifying drug for rheumatoid arthritis and psoriatic arthritis Licensing Information: Disease modifying drug for active
Shared Care Guideline DRUG: Introduction: LEFLUNOMIDE (Adults) Indication: Disease modifying drug for rheumatoid arthritis and psoriatic arthritis Licensing Information: Disease modifying drug for active
PACKAGE LEAFLET: INFORMATION FOR THE USER. VITAMINE B12 STEROP 1mg/1ml Solution for injection / oral solution. Cyanocobalamin
 PACKAGE LEAFLET: INFORMATION FOR THE USER VITAMINE B12 STEROP 1mg/1ml Solution for injection / oral solution Cyanocobalamin Read all of this leaflet carefully before you start using this medicine because
PACKAGE LEAFLET: INFORMATION FOR THE USER VITAMINE B12 STEROP 1mg/1ml Solution for injection / oral solution Cyanocobalamin Read all of this leaflet carefully before you start using this medicine because
Thioctacid 600 T Solution for Injection contains 600 mg alpha-lipoic acid
 Package Leaflet: Information for the User Thioctacid 600 T Solution for Injection contains 600 mg alpha-lipoic acid For use in adults Active substance: Alpha-lipoic acid, Trometamol salt (1:1) Read all
Package Leaflet: Information for the User Thioctacid 600 T Solution for Injection contains 600 mg alpha-lipoic acid For use in adults Active substance: Alpha-lipoic acid, Trometamol salt (1:1) Read all
Eye Trauma: Incidence
 Ocular Emergencies and Trauma Stuart Fourman, MD Director, Glaucoma Center Department of Ophthalmology Stony Brook University Eye Trauma: Incidence 1.3 million eye injuries per year in the United States.
Ocular Emergencies and Trauma Stuart Fourman, MD Director, Glaucoma Center Department of Ophthalmology Stony Brook University Eye Trauma: Incidence 1.3 million eye injuries per year in the United States.
Summary of the risk management plan (RMP) for Otezla (apremilast)
 EMA/741412/2014 Summary of the risk management plan (RMP) for Otezla (apremilast) This is a summary of the risk management plan (RMP) for Otezla, which details the measures to be taken in order to ensure
EMA/741412/2014 Summary of the risk management plan (RMP) for Otezla (apremilast) This is a summary of the risk management plan (RMP) for Otezla, which details the measures to be taken in order to ensure
SR 5. W2364A_NT.XATRAL SR5 PAK/SA 5/12/05 9:47 Page 1
 W2364A_NT.XATRAL SR5 PAK/SA 5/12/05 9:47 Page 1 alfuzosin SR 5 mg (Alfuzosin HCI) COMPOSITION Each sustained-release, film-coated tablet contains: Alfuzosin hydrochloride......... 5 mg Excipients... q.s.
W2364A_NT.XATRAL SR5 PAK/SA 5/12/05 9:47 Page 1 alfuzosin SR 5 mg (Alfuzosin HCI) COMPOSITION Each sustained-release, film-coated tablet contains: Alfuzosin hydrochloride......... 5 mg Excipients... q.s.
Laser Procedure Note
 Laser Procedure Note Patient Name Date 1. Pre procedure diagnosis 2. Procedure and Eye Eye: 3. Pre procedure topical medications administered (and time) 4. Vital signs BP / HR Acuity OD: OS: 5. IOP: OD
Laser Procedure Note Patient Name Date 1. Pre procedure diagnosis 2. Procedure and Eye Eye: 3. Pre procedure topical medications administered (and time) 4. Vital signs BP / HR Acuity OD: OS: 5. IOP: OD
MATERIAL SAFETY DATA SHEET
 Page 1 of 7 1. IDENTIFICATION OF THE SUBSTANCE/PREPARATION AND THE COMPANY/UNDERTAKING Pfizer Inc Pfizer Pharmaceuticals Group 235 East 42nd Street New York, New York 10017 1-212-573-2222 Emergency telephone
Page 1 of 7 1. IDENTIFICATION OF THE SUBSTANCE/PREPARATION AND THE COMPANY/UNDERTAKING Pfizer Inc Pfizer Pharmaceuticals Group 235 East 42nd Street New York, New York 10017 1-212-573-2222 Emergency telephone
Public Assessment Report. Pharmacy to General Sales List Reclassification. Pirinase Hayfever Relief for Adults 0.05% Nasal Spray.
 Public Assessment Report Pharmacy to General Sales List Reclassification Pirinase Hayfever Relief for Adults 0.05% Nasal Spray (Fluticasone) PL 00079/0688 Glaxo Wellcome UK Limited TABLE OF CONTENTS Introduction
Public Assessment Report Pharmacy to General Sales List Reclassification Pirinase Hayfever Relief for Adults 0.05% Nasal Spray (Fluticasone) PL 00079/0688 Glaxo Wellcome UK Limited TABLE OF CONTENTS Introduction
2 DOSAGE AND ADMINISTRATION 2.1 Dosing and Dose Adjustment
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use Auryxia safely and effectively. See full prescribing information for Auryxia. Auryxia (ferric citrate)
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use Auryxia safely and effectively. See full prescribing information for Auryxia. Auryxia (ferric citrate)
MATERIAL SAFETY DATA SHEET
 MATERIAL SAFETY DATA SHEET 1. IDENTIFICATION OF THE SUBSTANCE AND THE COMPANY Material Manufacturer Distributor Losartan Potassium and Hydrochlorothiazide Tablets 100 mg/12.5 mg Lupin Limited Goa 403 722
MATERIAL SAFETY DATA SHEET 1. IDENTIFICATION OF THE SUBSTANCE AND THE COMPANY Material Manufacturer Distributor Losartan Potassium and Hydrochlorothiazide Tablets 100 mg/12.5 mg Lupin Limited Goa 403 722
CONSENT FOR CATARACT SURGERY REQUEST FOR SURGICAL OPERATION / PROCEDURE AND ANAESTHETIC
 CONSENT FOR CATARACT SURGERY REQUEST FOR SURGICAL OPERATION / PROCEDURE AND ANAESTHETIC Your doctor has indicated that the condition of your eye appears stable and your cataract surgery and/or implantation
CONSENT FOR CATARACT SURGERY REQUEST FOR SURGICAL OPERATION / PROCEDURE AND ANAESTHETIC Your doctor has indicated that the condition of your eye appears stable and your cataract surgery and/or implantation
Eye Manifestations of Lupus And Sjogren s Syndrome
 Eye Manifestations of Lupus And Sjogren s Syndrome Based on a presentation by Dr. N. Kevin Wade at the BC Lupus Society Symposium held October 22, 2005. Background Dr. Wade completed his MD in 1998 at
Eye Manifestations of Lupus And Sjogren s Syndrome Based on a presentation by Dr. N. Kevin Wade at the BC Lupus Society Symposium held October 22, 2005. Background Dr. Wade completed his MD in 1998 at
Guidelines for the Use of Controlled Substances in the Treatment of Pain Adopted by the New Hampshire Medical Society, July 1998
 Guidelines for the Use of Controlled Substances in the Treatment of Pain Adopted by the New Hampshire Medical Society, July 1998 Section I: Preamble The New Hampshire Medical Society believes that principles
Guidelines for the Use of Controlled Substances in the Treatment of Pain Adopted by the New Hampshire Medical Society, July 1998 Section I: Preamble The New Hampshire Medical Society believes that principles
Preconception Clinical Care for Women Medical Conditions
 Preconception Clinical Care for Women All women of reproductive age are candidates for preconception care; however, preconception care must be tailored to meet the needs of the individual. Given that preconception
Preconception Clinical Care for Women All women of reproductive age are candidates for preconception care; however, preconception care must be tailored to meet the needs of the individual. Given that preconception
MATERIAL SAFETY DATA SHEET
 Page 1 of 5 1. IDENTIFICATION OF THE SUBSTANCE/PREPARATION AND THE COMPANY/UNDERTAKING Pfizer Inc Pfizer Pharmaceuticals Group 235 East 42nd Street New York, New York 10017 1-212-573-2222 Emergency telephone
Page 1 of 5 1. IDENTIFICATION OF THE SUBSTANCE/PREPARATION AND THE COMPANY/UNDERTAKING Pfizer Inc Pfizer Pharmaceuticals Group 235 East 42nd Street New York, New York 10017 1-212-573-2222 Emergency telephone
To: all optometrists and billing staff
 Number: Opto 27 Date: September 29, 2011 Page: 1 of 1 Subject: Schedule of Optometric Benefits amendments October 1, 2011/New explanatory code list Reference: Schedule of Optometric Benefits To: all optometrists
Number: Opto 27 Date: September 29, 2011 Page: 1 of 1 Subject: Schedule of Optometric Benefits amendments October 1, 2011/New explanatory code list Reference: Schedule of Optometric Benefits To: all optometrists
ALTERNATIVES TO LASIK
 EYE PHYSICIANS OF NORTH HOUSTON 845 FM 1960 WEST, SUITE 101, Houston, TX 77090 Office: 281 893 1760 Fax: 281 893 4037 INFORMED CONSENT FOR LASER IN-SITU KERATOMILEUSIS (LASIK) INTRODUCTION This information
EYE PHYSICIANS OF NORTH HOUSTON 845 FM 1960 WEST, SUITE 101, Houston, TX 77090 Office: 281 893 1760 Fax: 281 893 4037 INFORMED CONSENT FOR LASER IN-SITU KERATOMILEUSIS (LASIK) INTRODUCTION This information
Glaucoma. Quick reference guide. Issue date: April 2009. Diagnosis and management of chronic open angle glaucoma and ocular hypertension
 Issue date: April 2009 Glaucoma Diagnosis and management of chronic open angle glaucoma and ocular hypertension Developed by the National Collaborating Centre for Acute Care About this booklet This is
Issue date: April 2009 Glaucoma Diagnosis and management of chronic open angle glaucoma and ocular hypertension Developed by the National Collaborating Centre for Acute Care About this booklet This is
FDA approves Lucentis (ranibizumab injection) for treatment of diabetic macular edema
 Media Release Basel, 13 August 2012 FDA approves Lucentis (ranibizumab injection) for treatment of diabetic macular edema First major treatment advance in more than 25 years for sight-threatening condition
Media Release Basel, 13 August 2012 FDA approves Lucentis (ranibizumab injection) for treatment of diabetic macular edema First major treatment advance in more than 25 years for sight-threatening condition
