Chemical Name: (±)-9-Fluoro-2,3-dihydro-3-methyl-10-(4-methyl-1-piperazinyl)-7-oxo-7H-pyrido[1,2,3-de]-1,4-benzoxazine-6- carboxylic acid.
|
|
|
- Beverly Dawson
- 7 years ago
- Views:
Transcription
1 OCUFLOX (ofloxacin ophthalmic solution) 0.3% sterile DESCRIPTION OCUFLOX (ofloxacin ophthalmic solution) 0.3% is a sterile ophthalmic solution. It is a fluorinated carboxyquinolone anti-infective for topical ophthalmic use. Chemical Name: (±)-9-Fluoro-2,3-dihydro-3-methyl-10-(4-methyl-1-piperazinyl)-7-oxo-7H-pyrido[1,2,3-de]-1,4-benzoxazine-6- carboxylic acid. Contains: Active: ofloxacin 0.3% (3 mg/ml). Preservative: benzalkonium chloride (0.005%). Inactives: sodium chloride and purified water. May also contain hydrochloric acid and/or sodium hydroxide to adjust ph. OCUFLOX solution is unbuffered and formulated with a ph of 6.4 (range to 6.8). It has an osmolality of 300 mosm/kg. Ofloxacin is a fluorinated 4-quinolone which differs from other fluorinated 4-quinolones in that there is a six member (pyridobenzoxazine) ring from positions 1 to 8 of the basic ring structure. CLINICAL PHARMACOLOGY Pharmacokinetics Serum, urine and tear concentrations of ofloxacin were measured in 30 healthy women at various time points during a ten-day course of treatment with OCUFLOX solution. The mean serum ofloxacin concentration ranged from 0.4 ng/ml to 1.9 ng/ml. Maximum ofloxacin concentration increased from 1.1 ng/ml on day one to 1.9 ng/ml on day 11 after QID dosing for 10 1/2 days. Maximum serum ofloxacin concentrations after ten days of topical ophthalmic dosing were more than 1000 times lower than those reported after standard oral doses of ofloxacin. Tear ofloxacin concentrations ranged from 5.7 to 31 mcg/g during the 40 minute period following the last dose on day 11. Mean tear concentration measured four hours after topical ophthalmic dosing was 9.2 mcg/g. Corneal tissue concentrations of 4.4 mcg/ml were observed four hours after beginning topical ocular application of two drops of OCUFLOX ophthalmic solution every 30 minutes. Ofloxacin was excreted in the urine primarily unmodified. Microbiology Ofloxacin has in vitro activity against a broad range of gram-positive and gram-negative aerobic and anaerobic bacteria. Ofloxacin is bactericidal at concentrations equal to or slightly greater than inhibitory concentrations. Ofloxacin is thought to exert a bactericidal effect on susceptible bacterial cells by inhibiting DNA gyrase, an
2 essential bacterial enzyme which is a critical catalyst in the duplication, transcription, and repair of bacterial DNA. Cross-resistance has been observed between ofloxacin and other fluoroquinolones. There is generally no crossresistance between ofloxacin and other classes of antibacterial agents such as beta-lactams or aminoglycosides. Ofloxacin has been shown to be active against most strains of the following organisms both in vitro and clinically, in conjunctival and/or corneal ulcer infections as described in the INDICATIONS AND USAGE section. AEROBES, GRAM-POSITIVE: Staphylococcus aureus Staphylococcus epidermidis Streptococcus pneumoniae ANAEROBIC SPECIES: Propionibacterium acnes AEROBES, GRAM-NEGATIVE: Enterobacter cloacae Haemophilus influenzae Proteus mirabilis Pseudomonas aeruginosa Serratia marcescens* *Efficacy for this organism was studied in fewer than 10 infections The safety and effectiveness of OCUFLOX ophthalmic solution in treating ophthalmologic infections due to the following organisms have not been established in adequate and well-controlled clinical trials. OCUFLOX ophthalmic solution has been shown to be active in vitro against most strains of these organisms but the clinical significance in ophthalmologic infections is unknown. AEROBES, GRAM-POSITIVE: Enterococcus faecalis Listeria monocytogenes Staphylococcus capitis AEROBES, GRAM-NEGATIVE: Acinetobacter calcoaceticus var. anitratus Acinetobacter calcoaceticus var. lwoffii Citrobacter diversus Citrobacter freundii Enterobacter aerogenes Enterobacter agglomerans Escherichia coli Haemophilus parainfluenzae Klebsiella oxytoca OTHER: Chlamydia trachomatis Staphylococcus hominus Staphylococcus simulans Streptococcus pyogenes Klebsiella pneumoniae Moraxella (Branhamella) catarrhalis Moraxella lacunata Morganella morganii Neisseria gonorrhoeae Pseudomonas acidovorans Pseudomonas fluorescens Shigella sonnei Clinical Studies Conjunctivitis In a randomized, double-masked, multicenter clinical trial, OCUFLOX ophthalmic solution was superior to its vehicle after 2 days of treatment in patients with conjunctivitis and positive conjunctival cultures. Clinical outcomes for the trial demonstrated a clinical improvement rate of 86% (54/63) for the ofloxacin treated group
3 versus 72% (48/67) for the placebo treated group after 2 days of therapy. Microbiological outcomes for the same clinical trial demonstrated an eradication rate for causative pathogens of 65% (41/63) for the ofloxacin treated group versus 25% (17/67) for the vehicle treated group after 2 days of therapy. Please note that microbiologic eradication does not always correlate with clinical outcome in anti-infective trials. Corneal Ulcers In a randomized, double-masked, multi-center clinical trial of 140 subjects with positive cultures, OCUFLOX ophthalmic solution treated subjects had an overall clinical success rate (complete re-epithelialization and no progression of the infiltrate for two consecutive visits) of 82% (61/74) compared to 80% (53/66) for the fortified antibiotic group, consisting of 1.5% tobramycin and 10% cefazolin solutions. The median time to clinical success was 11 days for the ofloxacin treated group and 10 days for the fortified treatment group. INDICATIONS AND USAGE OCUFLOX ophthalmic solution is indicated for the treatment of infections caused by susceptible strains of the following bacteria in the conditions listed below: CONJUNCTIVITIS: Gram-positive bacteria: Staphylococcus aureus Staphylococcus epidermidis Streptococcus pneumoniae CORNEAL ULCERS: Gram-positive bacteria: Staphylococcus aureus Staphylococcus epidermidis Streptococcus pneumoniae Gram-negative bacteria: Enterobacter cloacae Haemophilus influenzae Proteus mirabilis Pseudomonas aeruginosa Gram-negative bacteria: Pseudomonas aeruginosa Serratia marcescens* Anaerobic species: Propionibacterium acnes *Efficacy for this organism was studied in fewer than 10 infections CONTRAINDICATIONS OCUFLOX solution is contraindicated in patients with a history of hypersensitivity to ofloxacin, to other quinolones, or to any of the components in this medication. WARNINGS NOT FOR INJECTION. OCUFLOX solution should not be injected subconjunctivally, nor should it be introduced directly into the anterior chamber of the eye. Serious and occasionally fatal hypersensitivity (anaphylactic) reactions, some following the first dose, have been reported in patients receiving systemic quinolones, including ofloxacin. Some reactions were accompanied by cardiovascular collapse, loss of consciousness, angioedema (including laryngeal, pharyngeal or facial edema), airway obstruction, dyspnea, urticaria, and itching. A rare occurrence of Stevens-Johnson syndrome, which progressed to toxic epidermal necrolysis, has been reported in a patient who was receiving topical
4 ophthalmic ofloxacin. If an allergic reaction to ofloxacin occurs, discontinue the drug. Serious acute hypersensitivity reactions may require immediate emergency treatment. Oxygen and airway management, including intubation should be administered as clinically indicated. PRECAUTIONS General As with other anti-infectives, prolonged use may result in overgrowth of nonsusceptible organisms, including fungi. If superinfection occurs discontinue use and institute alternative therapy. Whenever clinical judgment dictates, the patient should be examined with the aid of magnification, such as slit lamp biomicroscopy and, where appropriate, fluorescein staining. Ofloxacin should be discontinued at the first appearance of a skin rash or any other sign of hypersensitivity reaction. The systemic administration of quinolones, including ofloxacin, has led to lesions or erosions of the cartilage in weight-bearing joints and other signs of arthropathy in immature animals of various species. Ofloxacin, administered systemically at 10 mg/kg/day in young dogs (equivalent to 110 times the maximum recommended daily adult ophthalmic dose) has been associated with these types of effects. Information for Patients Avoid contaminating the applicator tip with material from the eye, fingers or other source. Systemic quinolones, including ofloxacin, have been associated with hypersensitivity reactions, even following a single dose. Discontinue use immediately and contact your physician at the first sign of a rash or allergic reaction. Drug Interactions Specific drug interaction studies have not been conducted with OCUFLOX ophthalmic solution. However, the systemic administration of some quinolones has been shown to elevate plasma concentrations of theophylline, interfere with the metabolism of caffeine, and enhance the effects of the oral anticoagulant warfarin and its derivatives, and has been associated with transient elevations in serum creatinine in patients receiving cyclosporine concomitantly. Carcinogenesis, Mutagenesis, Impairment of Fertility Long term studies to determine the carcinogenic potential of ofloxacin have not been conducted. Ofloxacin was not mutagenic in the Ames test, in vitro and in vivo cytogenic assay, sister chromatid exchange assay (Chinese hamster and human cell lines), unscheduled DNA synthesis (UDS) assay using human fibroblasts, the dominant lethal assay, or mouse micronucleus assay. Ofloxacin was positive in the UDS test using rat hepatocyte, and in the mouse lymphoma assay. In fertility studies in rats, ofloxacin did not affect male or female fertility or morphological or reproductive performance at oral dosing up to 360 mg/kg/day (equivalent to 4000 times the maximum recommended daily ophthalmic dose). Pregnancy Teratogenic Effects. Pregnancy Category C Ofloxacin has been shown to have an embryocidal effect in rats and in rabbits when given in doses of 810 mg/kg/day (equivalent to 9000 times the maximum recommended daily ophthalmic dose) and 160 mg/kg/day (equivalent to 1800 times the maximum recommended daily ophthalmic dose). These dosages resulted in decreased fetal body weight and increased fetal mortality in rats and rabbits, respectively. Minor fetal skeletal variations were reported in rats receiving doses of 810 mg/kg/day. Ofloxacin has not been shown to be
5 teratogenic at doses as high as 810 mg/kg/day and 160 mg/kg/day when administered to pregnant rats and rabbits, respectively. Nonteratogenic Effects Additional studies in rats with doses up to 360 mg/kg/day during late gestation showed no adverse effect on late fetal development, labor, delivery, lactation, neonatal viability, or growth of the newborn. There are, however, no adequate and well-controlled studies in pregnant women. OCUFLOX solution should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus. Nursing Mothers In nursing women a single 200 mg oral dose resulted in concentrations of ofloxacin in milk which were similar to those found in plasma. It is not known whether ofloxacin is excreted in human milk following topical ophthalmic administration. Because of the potential for serious adverse reactions from ofloxacin in nursing infants, a decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother. Pediatric Use Safety and effectiveness in infants below the age of one year have not been established. Quinolones, including ofloxacin, have been shown to cause arthropathy in immature animals after oral administration; however, topical ocular administration of ofloxacin to immature animals has not shown any arthropathy. There is no evidence that the ophthalmic dosage form of ofloxacin has any effect on weight bearing joints. Geriatric Use No overall differences in safety or effectiveness have been observed between elderly and younger patients. ADVERSE REACTIONS Ophthalmic Use The most frequently reported drug-related adverse reaction was transient ocular burning or discomfort. Other reported reactions include stinging, redness, itching, chemical conjunctivitis/keratitis, ocular/periocular/facial edema, foreign body sensation, photophobia, blurred vision, tearing, dryness, and eye pain. Rare reports of dizziness and nausea have been received. DOSAGE AND ADMINISTRATION The recommended dosage regimen for the treatment of bacterial conjunctivitis is: Days 1 and 2 Instill one to two drops every two to four hours in the affected eye(s). Days 3 through 7 Instill one to two drops four times daily. The recommended dosage regimen for the treatment of bacterial corneal ulcer is: Days 1 and 2 Instill one to two drops into the affected eye every 30 minutes, while awake. Awaken at approximately four and six hours after retiring and instill one to two drops. Days 3 through 7 to 9 Instill one to two drops hourly, while awake. Days 7 to 9 through treatment completion Instill one to two drops, four times daily.
6 HOW SUPPLIED OCUFLOX (ofloxacin ophthalmic solution) 0.3% is supplied sterile in opaque white LDPE plastic bottles and white dropper tips with beige high impact polystyrene (HIPS) caps as follows: 5mL in 10mL bottle - NDC Note: Store at C (59-77 F) Rx only Revised: 02/ Allergan, Inc., Irvine, CA 92612, U.S.A. marks owned by Allergan, Inc. Made in the U.S.A US12C
DO YOU HAVE ITCHY ALLERGY EYES? Find out about lasting relief
 DO YOU HAVE ITCHY ALLERGY EYES? Find out about lasting relief Common causes of itching due to eye allergies include: Pollen from trees, grasses, and ragweed Dust mites Cat dander Approximately 10 million
DO YOU HAVE ITCHY ALLERGY EYES? Find out about lasting relief Common causes of itching due to eye allergies include: Pollen from trees, grasses, and ragweed Dust mites Cat dander Approximately 10 million
FULL PRESCRIBING INFORMATION
 HIGHLIGHTS F PRESCRIBING INFRMATIN These highlights do not include all the information needed to use AzaSite safely and effectively. See full prescribing information for AzaSite. AzaSite (azithromycin
HIGHLIGHTS F PRESCRIBING INFRMATIN These highlights do not include all the information needed to use AzaSite safely and effectively. See full prescribing information for AzaSite. AzaSite (azithromycin
CEFA-DROPS AND CEFA-TABS
 Page 1 of 5 FORT DODGE ANIMAL HEALTH Division of Wyeth 800-5TH STREET N.W., P.O. BOX 518 FORT DODGE IA 50501 USA Telephone: 515-955-4600 Fax: 515-955-3730 Order Desk Telephone: 800-685-5656 Order Desk
Page 1 of 5 FORT DODGE ANIMAL HEALTH Division of Wyeth 800-5TH STREET N.W., P.O. BOX 518 FORT DODGE IA 50501 USA Telephone: 515-955-4600 Fax: 515-955-3730 Order Desk Telephone: 800-685-5656 Order Desk
Revised: 9/2014. LUMIGAN (bimatoprost ophthalmic solution) 0.01% for topical ophthalmic use. Initial U.S. Approval: 2001
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use LUMIGAN (bimatoprost ophthalmic solution) 0.01% safely and effectively. See full prescribing information
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use LUMIGAN (bimatoprost ophthalmic solution) 0.01% safely and effectively. See full prescribing information
skin and soft tissue infections (skinfold pyoderma, impetigo, folliculitis, furunculosis, cellulitis) caused by susceptible strains of organisms.
 Part II SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE VETERINARY MEDICINAL PRODUCT MARBOCYL P 5 mg tablet 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Active Ingredient Marbofloxacin 5mg per tablet
Part II SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE VETERINARY MEDICINAL PRODUCT MARBOCYL P 5 mg tablet 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Active Ingredient Marbofloxacin 5mg per tablet
FULL PRESCRIBING INFORMATION: CONTENTS*
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use PHOTREXA VISCOUS and PHOTREXA safely and effectively. See full prescribing information for PHOTREXA
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use PHOTREXA VISCOUS and PHOTREXA safely and effectively. See full prescribing information for PHOTREXA
ZOVIRAX Cold Sore Cream
 Data Sheet ZOVIRAX Cold Sore Cream Aciclovir 5% w/w Presentation Topical cream Indications ZOVIRAX Cold Sore Cream is indicated for the treatment of Herpes simplex virus infections of the lips and face
Data Sheet ZOVIRAX Cold Sore Cream Aciclovir 5% w/w Presentation Topical cream Indications ZOVIRAX Cold Sore Cream is indicated for the treatment of Herpes simplex virus infections of the lips and face
SUMMARY OF PRODUCT CHARACTERISTICS
 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Aknemycin Solution 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 10 g of solution contains 0.2 g of erythromycin. Structural formula of
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Aknemycin Solution 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 10 g of solution contains 0.2 g of erythromycin. Structural formula of
Revised: 9/2014 HIGHLIGHTS OF PRESCRIBING INFORMATION
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use LATISSE safely and effectively. See full prescribing information for LATISSE. LATISSE (bimatoprost
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use LATISSE safely and effectively. See full prescribing information for LATISSE. LATISSE (bimatoprost
NEW ZEALAND DATA SHEET NAPHCON-A Naphazoline hydrochloride and pheniramine maleate.
 NEW ZEALAND DATA SHEET NAPHCON-A Naphazoline hydrochloride and pheniramine maleate. PRESENTATION Eye Drops: NAPHCON-A Eye Drops are a combination of an antihistamine (pheniramine maleate) and a decongestant
NEW ZEALAND DATA SHEET NAPHCON-A Naphazoline hydrochloride and pheniramine maleate. PRESENTATION Eye Drops: NAPHCON-A Eye Drops are a combination of an antihistamine (pheniramine maleate) and a decongestant
INITIATING ORAL AUBAGIO (teriflunomide) THERAPY
 FOR YOUR PATIENTS WITH RELAPSING FORMS OF MS INITIATING ORAL AUBAGIO (teriflunomide) THERAPY WARNING: HEPATOTOXICITY AND RISK OF TERATOGENICITY Severe liver injury including fatal liver failure has been
FOR YOUR PATIENTS WITH RELAPSING FORMS OF MS INITIATING ORAL AUBAGIO (teriflunomide) THERAPY WARNING: HEPATOTOXICITY AND RISK OF TERATOGENICITY Severe liver injury including fatal liver failure has been
WARNING LETTER. According to its approved product labeling (PI) (in pertinent part, emphasis original):
 DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service Food and Drug Administration Silver Spring, MD 20993-0002 TRANSMITTED BY FACSIMILE Sapan A. Shah, Ph.D. President and Chief Executive Officer
DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service Food and Drug Administration Silver Spring, MD 20993-0002 TRANSMITTED BY FACSIMILE Sapan A. Shah, Ph.D. President and Chief Executive Officer
See 17 for PATIENT COUNSELING INFORMATION. Revised: 3/2016 FULL PRESCRIBING INFORMATION: CONTENTS* WARNING: ADRENAL CRISIS IN THE SETTING OF SHOCK OR
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use LYSODREN safely and effectively. See full prescribing information for LYSODREN. LYSODREN (mitotane)
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use LYSODREN safely and effectively. See full prescribing information for LYSODREN. LYSODREN (mitotane)
COMPOSITION: Each capsule contains clindamycin hydrochloride equivalent to 150 mg clindamycin base.
 APPROVED PACKAGE INSERT DALACIN C 150 mg CAPSULES SCHEDULING STATUS: S4 PROPRIETARY NAME (and dosage form): DALACIN C TM 150 mg (Capsules) COMPOSITION: Each capsule contains clindamycin hydrochloride equivalent
APPROVED PACKAGE INSERT DALACIN C 150 mg CAPSULES SCHEDULING STATUS: S4 PROPRIETARY NAME (and dosage form): DALACIN C TM 150 mg (Capsules) COMPOSITION: Each capsule contains clindamycin hydrochloride equivalent
CEFADROXIL CAPSULES, USP 500 mg Rx only
 CEFADROXIL CAPSULES, USP 500 mg Rx only To reduce the development of drug-resistant bacteria and maintain the effectiveness of cefadroxil capsules and other antibacterial drugs, cefadroxil capsules should
CEFADROXIL CAPSULES, USP 500 mg Rx only To reduce the development of drug-resistant bacteria and maintain the effectiveness of cefadroxil capsules and other antibacterial drugs, cefadroxil capsules should
PRODUCT INFORMATION. Silver sulfadiazine is a sulfonamide and has broad antimicrobial activity against both Grampositive and Gram-negative organisms.
 PRODUCT INFORMATION Name of the Medicine: FLAMAZINE CREAM 1.0% w/w Silver sulfadiazine 1% w/w Composition: Active ingredient. Silver sulfadiazine. Excipients. Polysorbate 60 Ph. Eur, Polysorbate 80 Ph.
PRODUCT INFORMATION Name of the Medicine: FLAMAZINE CREAM 1.0% w/w Silver sulfadiazine 1% w/w Composition: Active ingredient. Silver sulfadiazine. Excipients. Polysorbate 60 Ph. Eur, Polysorbate 80 Ph.
CIPRO (ciprofloxacin hydrochloride) TABLETS. CIPRO (ciprofloxacin*) ORAL SUSPENSION
 CIPRO (ciprofloxacin hydrochloride) TABLETS CIPRO (ciprofloxacin*) ORAL SUSPENSION 08918468, R.0 10/04 To reduce the development of drug-resistant bacteria and maintain the effectiveness of CIPRO Tablets
CIPRO (ciprofloxacin hydrochloride) TABLETS CIPRO (ciprofloxacin*) ORAL SUSPENSION 08918468, R.0 10/04 To reduce the development of drug-resistant bacteria and maintain the effectiveness of CIPRO Tablets
VOLTAREN OPHTHA EYE DROPS
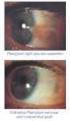 2013 במאי משרד הבריאות ותוכנו נבדק ואושר על ידו ע"י עלון זה נקבע ע פורמט PRESCRIBING INFORMATION VOLTAREN OPHTHA EYE DROPS 1 Name of the medicinal product Voltaren Ophtha Eye Drops. 2 Qualitative and quantitative
2013 במאי משרד הבריאות ותוכנו נבדק ואושר על ידו ע"י עלון זה נקבע ע פורמט PRESCRIBING INFORMATION VOLTAREN OPHTHA EYE DROPS 1 Name of the medicinal product Voltaren Ophtha Eye Drops. 2 Qualitative and quantitative
Teriflunomide is the active metabolite of Leflunomide, a drug employed since 1994 for the treatment of rheumatoid arthritis (Baselt, 2011).
 Page 1 of 10 ANALYTE NAME AND STRUCTURE TERIFLUNOMIDE Teriflunomide TRADE NAME Aubagio CATEGORY Antimetabolite TEST CODE PURPOSE Therapeutic Drug Monitoring GENERAL RELEVANCY BACKGROUND sclerosis. The
Page 1 of 10 ANALYTE NAME AND STRUCTURE TERIFLUNOMIDE Teriflunomide TRADE NAME Aubagio CATEGORY Antimetabolite TEST CODE PURPOSE Therapeutic Drug Monitoring GENERAL RELEVANCY BACKGROUND sclerosis. The
VOLTAREN OPHTHA EYE DROPS
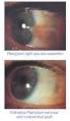 פורמט עלון זה נקבע ע"ימשרד הבריאות ותוכנו נבדק ואושר על ידו באפריל 2008 PRESCRIBING INFORMATION VOLTAREN OPHTHA EYE DROPS 1 Name of the medicinal product Voltaren Ophtha Eye Drops. 2 Qualitative and quantitative
פורמט עלון זה נקבע ע"ימשרד הבריאות ותוכנו נבדק ואושר על ידו באפריל 2008 PRESCRIBING INFORMATION VOLTAREN OPHTHA EYE DROPS 1 Name of the medicinal product Voltaren Ophtha Eye Drops. 2 Qualitative and quantitative
VISTARIL (hydroxyzine pamoate) Capsules and Oral Suspension
 VISTARIL (hydroxyzine pamoate) Capsules and Oral Suspension DESCRIPTION Hydroxyzine pamoate is designated chemically as 1-(p-chlorobenzhydryl) 4- [2-(2-hydroxyethoxy) ethyl] diethylenediamine salt of 1,1
VISTARIL (hydroxyzine pamoate) Capsules and Oral Suspension DESCRIPTION Hydroxyzine pamoate is designated chemically as 1-(p-chlorobenzhydryl) 4- [2-(2-hydroxyethoxy) ethyl] diethylenediamine salt of 1,1
FLAMAZINE CREAM 1.0% w/w
 PRODUCT INFORMATION NAME OF THE MEDICINE: FLAMAZINE CREAM 1.0% w/w Silver sulfadiazine 1% w/w Composition: Active ingredient. Silver sulfadiazine. Excipients. Polysorbate 60 Ph. Eur, Polysorbate 80 Ph.
PRODUCT INFORMATION NAME OF THE MEDICINE: FLAMAZINE CREAM 1.0% w/w Silver sulfadiazine 1% w/w Composition: Active ingredient. Silver sulfadiazine. Excipients. Polysorbate 60 Ph. Eur, Polysorbate 80 Ph.
PACKAGE LEAFLET. CLINDAMYCIN capsules Clidamycin. One capsule of 75 mg contains 75 mg Clindamycin (as hydrochloride).
 PACKAGE LEAFLET CLINDAMYCIN capsules Clidamycin COMPOSITION One capsule of 75 mg contains 75 mg Clindamycin (as hydrochloride). One capsule of 150 mg contains 150 mg Clindamycin (as hydrochloride). PROPERTIES
PACKAGE LEAFLET CLINDAMYCIN capsules Clidamycin COMPOSITION One capsule of 75 mg contains 75 mg Clindamycin (as hydrochloride). One capsule of 150 mg contains 150 mg Clindamycin (as hydrochloride). PROPERTIES
NIZORAL (KETOCONAZOLE) 2% SHAMPOO DESCRIPTION
 NIZORAL (KETOCONAZOLE) 2% SHAMPOO DESCRIPTION NIZORAL (ketoconazole) 2% Shampoo is a red-orange liquid for topical application, containing the broad spectrum synthetic antifungal agent ketoconazole in
NIZORAL (KETOCONAZOLE) 2% SHAMPOO DESCRIPTION NIZORAL (ketoconazole) 2% Shampoo is a red-orange liquid for topical application, containing the broad spectrum synthetic antifungal agent ketoconazole in
ZERBAXA (ceftolozane and tazobactam) for injection, for intravenous use Initial U.S. Approval: 2014
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use ZERBAXA safely and effectively. See full prescribing information for ZERBAXA. ZERBAXA (ceftolozane
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use ZERBAXA safely and effectively. See full prescribing information for ZERBAXA. ZERBAXA (ceftolozane
FRENCH AGENCY FOR VETERINARY MEDICINAL PRODUCTS DECENTRALISED PROCEDURE PUBLICLY AVAILABLE ASSESSMENT REPORT FOR A VETERINARY MEDICINAL PRODUCT
 FRENCH AGENCY FOR VETERINARY MEDICINAL PRODUCTS DECENTRALISED PROCEDURE FOR A VETERINARY MEDICINAL PRODUCT EMEDOG, 1 mg/ml, solution for injection for dogs Date: 20/08/2015 French agency for food, environnemental
FRENCH AGENCY FOR VETERINARY MEDICINAL PRODUCTS DECENTRALISED PROCEDURE FOR A VETERINARY MEDICINAL PRODUCT EMEDOG, 1 mg/ml, solution for injection for dogs Date: 20/08/2015 French agency for food, environnemental
Learn More About Product Labeling
 Learn More About Product Labeling Product label The product label is developed during the formal process of review and approval by regulatory agencies of any medicine or medical product. There are specific
Learn More About Product Labeling Product label The product label is developed during the formal process of review and approval by regulatory agencies of any medicine or medical product. There are specific
FOR ORAL OR RECTAL ADMINISTRATION FOR THE PREVENTION AND TREATMENT OF PORTAL-SYSTEMIC ENCEPHALOPATHY
 FORM NO. 1360 VC3074 Rev.2/08 LACTULOSE SOLUTION, USP 10 g/15 ml FOR ORAL OR RECTAL ADMINISTRATION FOR THE PREVENTION AND TREATMENT OF PORTAL-SYSTEMIC ENCEPHALOPATHY Rx Only DESCRIPTION Lactulose is a
FORM NO. 1360 VC3074 Rev.2/08 LACTULOSE SOLUTION, USP 10 g/15 ml FOR ORAL OR RECTAL ADMINISTRATION FOR THE PREVENTION AND TREATMENT OF PORTAL-SYSTEMIC ENCEPHALOPATHY Rx Only DESCRIPTION Lactulose is a
CIPRO CIPRO. AUC 0-12h b
 CIPRO (ciprofloxacin hydrochloride) TABLETS CIPRO (ciprofloxacin*) ORAL SUSPENSION 81532304, R.1 04/09 WARNING: Fluoroquinolones, including CIPRO, are associated with an increased risk of tendinitis and
CIPRO (ciprofloxacin hydrochloride) TABLETS CIPRO (ciprofloxacin*) ORAL SUSPENSION 81532304, R.1 04/09 WARNING: Fluoroquinolones, including CIPRO, are associated with an increased risk of tendinitis and
Hypromellose Eye Drops BP 0.3% w/v PL 23097/0006
 Hypromellose Eye Drops BP 0.3% w/v PL 23097/0006 UKPAR TABLE OF CONTENTS Lay summary Page 2 Scientific discussion Page 3 Steps taken for assessment Page 9 Summary of product characteristics Page 10 Patient
Hypromellose Eye Drops BP 0.3% w/v PL 23097/0006 UKPAR TABLE OF CONTENTS Lay summary Page 2 Scientific discussion Page 3 Steps taken for assessment Page 9 Summary of product characteristics Page 10 Patient
IMPORTANT DRUG WARNING Regarding Mycophenolate-Containing Products
 Dear Healthcare Provider: Mycophenolate REMS (Risk Evaluation and Mitigation Strategy) has been mandated by the FDA (Food and Drug Administration) due to postmarketing reports showing that exposure to
Dear Healthcare Provider: Mycophenolate REMS (Risk Evaluation and Mitigation Strategy) has been mandated by the FDA (Food and Drug Administration) due to postmarketing reports showing that exposure to
used to treat inflammation, corneal injury and bacterial infections in the external part of the eye.
 TOBRADEX * Eye Drops Tobramycin and Dexamethasone CONSUMER MEDICINE INFORMATION What is in this Leaflet This leaflet answers some common questions about TOBRADEX* Eye Drops. It does not contain all the
TOBRADEX * Eye Drops Tobramycin and Dexamethasone CONSUMER MEDICINE INFORMATION What is in this Leaflet This leaflet answers some common questions about TOBRADEX* Eye Drops. It does not contain all the
0.9% Sodium Chloride Injection, USP In VIAFLEX Plastic Container
 Page 1 of 8 PRESCRIBING INFORMATION 0.9% Sodium Chloride Injection, USP In VIAFLEX Plastic Container IV Fluid and Electrolyte Replenisher Baxter Corporation Mississauga, Ontario L5N 0C2 Canada Date of
Page 1 of 8 PRESCRIBING INFORMATION 0.9% Sodium Chloride Injection, USP In VIAFLEX Plastic Container IV Fluid and Electrolyte Replenisher Baxter Corporation Mississauga, Ontario L5N 0C2 Canada Date of
Lecture Outline. Quinolones
 Lecture Outline Quinolones Trimethoprim/Sulfamethoxazole Miscellaneous antimicrobials - Metronidazole Daptomycin Cases Quinolones Bactericidal broad spectrum antibiotics Increasingly used because of their
Lecture Outline Quinolones Trimethoprim/Sulfamethoxazole Miscellaneous antimicrobials - Metronidazole Daptomycin Cases Quinolones Bactericidal broad spectrum antibiotics Increasingly used because of their
8.5 Geriatric Use * Sections or subsections omitted from the full prescribing information are not listed.
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use Injectafer safely and effectively. See full prescribing information for Injectafer. INJECTAFER (ferric
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use Injectafer safely and effectively. See full prescribing information for Injectafer. INJECTAFER (ferric
The clinical studies have been performed in children, adolescents and adults, from 4 years up to 55 years of age.
 NAME OF THE MEDICINE TdaP-Booster. Diphtheria, tetanus and pertussis (acellular mono-component) vaccine (adsorbed, reduced antigen content). DESCRIPTION TdaP-Booster is a suspension for injection in pre-filled
NAME OF THE MEDICINE TdaP-Booster. Diphtheria, tetanus and pertussis (acellular mono-component) vaccine (adsorbed, reduced antigen content). DESCRIPTION TdaP-Booster is a suspension for injection in pre-filled
See 17 for PATIENT COUNSELING INFORMATION and FDAapproved. Revised: 4/2015
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use DUAC Gel safely and effectively. See full prescribing information for DUAC Gel. DUAC (clindamycin
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use DUAC Gel safely and effectively. See full prescribing information for DUAC Gel. DUAC (clindamycin
CHLORHEXIDINE ACETATE WITH CETRIMIDE (%) ANTISEPTIC SOLUTION
 CHLORHEXIDINE ACETATE WITH CETRIMIDE (%) ANTISEPTIC SOLUTION NAME OF THE MEDICINE Proprietary name: Non-proprietary name: Chemical Name: Molecular formula: Baxter Chlorhexidine Cetrimide Antiseptic Solution
CHLORHEXIDINE ACETATE WITH CETRIMIDE (%) ANTISEPTIC SOLUTION NAME OF THE MEDICINE Proprietary name: Non-proprietary name: Chemical Name: Molecular formula: Baxter Chlorhexidine Cetrimide Antiseptic Solution
2 DOSAGE AND ADMINISTRATION 2.1 Dosing and Dose Adjustment
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use Auryxia safely and effectively. See full prescribing information for Auryxia. Auryxia (ferric citrate)
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use Auryxia safely and effectively. See full prescribing information for Auryxia. Auryxia (ferric citrate)
Laboratory Exercise # 11: Differentiation of the Species Staphylococcus and Streptococcus
 Laboratory Exercise # 11: Differentiation of the Species Staphylococcus and Streptococcus Purpose: The purpose of this laboratory exercise is to explore the differences between Staphylococcal species and
Laboratory Exercise # 11: Differentiation of the Species Staphylococcus and Streptococcus Purpose: The purpose of this laboratory exercise is to explore the differences between Staphylococcal species and
160S01105, Page 1 of 7. Human Hepatitis B Immunoglobulin, solution for intramuscular injection.
 160S01105, Page 1 of 7 New Zealand Data Sheet Hepatitis B Immunoglobulin-VF NAME OF THE MEDICINE Human Hepatitis B Immunoglobulin, solution for intramuscular injection. DESCRIPTION Hepatitis B Immunoglobulin-VF
160S01105, Page 1 of 7 New Zealand Data Sheet Hepatitis B Immunoglobulin-VF NAME OF THE MEDICINE Human Hepatitis B Immunoglobulin, solution for intramuscular injection. DESCRIPTION Hepatitis B Immunoglobulin-VF
Clindamycin is a semisynthetic antibiotic produced by a 7(S)-chloro-substitution of the 7(R)- hydroxyl group of the parent compound lincomycin.
 DALACIN C - Oral Capsules 1. QUALITATIVE AND QUANTITATIVE COMPOSITION Active Ingredient: Clindamycin hydrochloride. Inactive ingredients: Magnesium stearate, maize starch, talc,lactose, hard gelatin capsule
DALACIN C - Oral Capsules 1. QUALITATIVE AND QUANTITATIVE COMPOSITION Active Ingredient: Clindamycin hydrochloride. Inactive ingredients: Magnesium stearate, maize starch, talc,lactose, hard gelatin capsule
PART B: METHODS FOR THE DETERMINATION OF TOXICITY AND OTHER HEALTH EFFECTS GENERAL INTRODUCTION: PART B
 PART B: METHODS FOR THE DETERMINATION OF TOXICITY AND OTHER HEALTH EFFECTS GENERAL INTRODUCTION: PART B A. EXPLANATORY NOTE For the purpose of this General Introduction the following nymbering applies:
PART B: METHODS FOR THE DETERMINATION OF TOXICITY AND OTHER HEALTH EFFECTS GENERAL INTRODUCTION: PART B A. EXPLANATORY NOTE For the purpose of this General Introduction the following nymbering applies:
Billing and Coding Guide
 Billing and Coding Guide INDICATIONS Injectafer is an iron replacement product indicated for the treatment of iron deficiency anemia (IDA) in adult patients: who have intolerance to or have had unsatisfactory
Billing and Coding Guide INDICATIONS Injectafer is an iron replacement product indicated for the treatment of iron deficiency anemia (IDA) in adult patients: who have intolerance to or have had unsatisfactory
HOW TO WRITE AN UNKNOWN LAB REPORT IN MICROBIOLOGY
 HOW TO WRITE AN UNKNOWN LAB REPORT IN MICROBIOLOGY GENERAL Unknown reports in microbiology are written in scientific format. Scientific writing is written differently from other types of writing. The results
HOW TO WRITE AN UNKNOWN LAB REPORT IN MICROBIOLOGY GENERAL Unknown reports in microbiology are written in scientific format. Scientific writing is written differently from other types of writing. The results
PARACETAMOL REXIDOL. 600 mg Tablet. Analgesic-Antipyretic. Paracetamol 600 mg
 (Insert Text) UL Consumer Health PARACETAMOL REXIDOL 600 mg Tablet Analgesic-Antipyretic FORMULATION Each tablet contains: Paracetamol 600 mg PRODUCT DESCRIPTION Rexidol is a round, yellow, flat, bevel-edged
(Insert Text) UL Consumer Health PARACETAMOL REXIDOL 600 mg Tablet Analgesic-Antipyretic FORMULATION Each tablet contains: Paracetamol 600 mg PRODUCT DESCRIPTION Rexidol is a round, yellow, flat, bevel-edged
5.07.09. Aubagio. Aubagio (teriflunomide) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.07.09 Subject: Aubagio Page: 1 of 6 Last Review Date: December 5, 2014 Aubagio Description Aubagio (teriflunomide)
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.07.09 Subject: Aubagio Page: 1 of 6 Last Review Date: December 5, 2014 Aubagio Description Aubagio (teriflunomide)
3% Sodium Chloride Injection, USP 5% Sodium Chloride Injection, USP
 PRESCRIBING INFORMATION 3% Sodium Chloride Injection, USP 5% Sodium Chloride Injection, USP IV Fluid and Electrolyte Replenisher Baxter Corporation Mississauga, Ontario L5N 0C2 Canada Date of Revision:
PRESCRIBING INFORMATION 3% Sodium Chloride Injection, USP 5% Sodium Chloride Injection, USP IV Fluid and Electrolyte Replenisher Baxter Corporation Mississauga, Ontario L5N 0C2 Canada Date of Revision:
White, circular, biconvex, uncoated tablets with a score line on one side, plain on the other.
 Nausicalm Cyclizine hydrochloride Ph. Eur. 50 mg Presentation White, circular, biconvex, uncoated tablets with a score line on one side, plain on the other. Uses Actions The active ingredient-cyclizine
Nausicalm Cyclizine hydrochloride Ph. Eur. 50 mg Presentation White, circular, biconvex, uncoated tablets with a score line on one side, plain on the other. Uses Actions The active ingredient-cyclizine
45813E/Revised: April 2008 CYANOCOBALAMIN INJECTION, USP
 45813E/Revised: April 2008 CYANOCOBALAMIN INJECTION, USP DESCRIPTION: Cyanocobalamin Injection, USP is a sterile solution of cyanocobalamin for intramuscular or subcutaneous use. Each ml contains 1000
45813E/Revised: April 2008 CYANOCOBALAMIN INJECTION, USP DESCRIPTION: Cyanocobalamin Injection, USP is a sterile solution of cyanocobalamin for intramuscular or subcutaneous use. Each ml contains 1000
Dechra Veterinary Products Limited (A business unit of Dechra Pharmaceuticals PLC) Sansaw Business Park Hadnall, Shrewsbury Shropshire SY4 4AS
 Dechra Veterinary Products Limited (A business unit of Dechra Pharmaceuticals PLC) Sansaw Business Park Hadnall, Shrewsbury Shropshire SY4 4AS Tel: 01939 211200 Fax: 01939 211201 Email: info.uk@dechra.com
Dechra Veterinary Products Limited (A business unit of Dechra Pharmaceuticals PLC) Sansaw Business Park Hadnall, Shrewsbury Shropshire SY4 4AS Tel: 01939 211200 Fax: 01939 211201 Email: info.uk@dechra.com
Material Safety data sheet
 EMERGENCY OVERVIEW METFORMIN HYDROCHLORIDE TABLET contain Metformin and excipients generally considered to be non- toxic and non-hazardous in small quantities and under conditions of normal occupational
EMERGENCY OVERVIEW METFORMIN HYDROCHLORIDE TABLET contain Metformin and excipients generally considered to be non- toxic and non-hazardous in small quantities and under conditions of normal occupational
PACKAGE LEAFLET: INFORMATION FOR THE USER. VITAMINE B12 STEROP 1mg/1ml Solution for injection / oral solution. Cyanocobalamin
 PACKAGE LEAFLET: INFORMATION FOR THE USER VITAMINE B12 STEROP 1mg/1ml Solution for injection / oral solution Cyanocobalamin Read all of this leaflet carefully before you start using this medicine because
PACKAGE LEAFLET: INFORMATION FOR THE USER VITAMINE B12 STEROP 1mg/1ml Solution for injection / oral solution Cyanocobalamin Read all of this leaflet carefully before you start using this medicine because
SUMMARY OF PRODUCT CHARACTERISTICS 2 QUALITATIVE AND QUANTITATIVE COMPOSITION
 SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Targocid 200mg Teicoplanin 200mg Powder for Injection Targocid 400mg Teicoplanin 400mg Powder for Injection 2 QUALITATIVE AND QUANTITATIVE
SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Targocid 200mg Teicoplanin 200mg Powder for Injection Targocid 400mg Teicoplanin 400mg Powder for Injection 2 QUALITATIVE AND QUANTITATIVE
Agencia Española de Medicamentos y Productos Sanitarios C/Campezo 1, Edificio 8 28022 Madrid España
 DEPARTAMENTO DE MEDICAMENTOS VETERINARIOS Medicamentos y Productos C/Campezo 1, Edificio 8 28022 Madrid España DECENTRALISED PROCEDURE PUBLICLY AVAILABLE ASSESSMENT REPORT FOR A VETERINARY MEDICINAL PRODUCT
DEPARTAMENTO DE MEDICAMENTOS VETERINARIOS Medicamentos y Productos C/Campezo 1, Edificio 8 28022 Madrid España DECENTRALISED PROCEDURE PUBLICLY AVAILABLE ASSESSMENT REPORT FOR A VETERINARY MEDICINAL PRODUCT
EFFIMET 1000 XR Metformin Hydrochloride extended release tablet
 BRAND NAME: Effimet XR. THERAPEUTIC CATEGORY: Anti-Diabetic PHARMACOLOGIC CLASS: Biguanides EFFIMET 1000 XR Metformin Hydrochloride extended release tablet COMPOSITION AND PRESENTATION Composition Each
BRAND NAME: Effimet XR. THERAPEUTIC CATEGORY: Anti-Diabetic PHARMACOLOGIC CLASS: Biguanides EFFIMET 1000 XR Metformin Hydrochloride extended release tablet COMPOSITION AND PRESENTATION Composition Each
Results of all scientific investigations with the A.Vogel Sore Throat Spray. Issue 2 - June 2008
 Results of all scientific investigations with the A.Vogel Sore Issue 2 - June 2008 Antiviral Antibacterial Anti-inflammatory Analgetic Immunemodulatory Authors: Andy Suter, Medical Department Roland Schoop,
Results of all scientific investigations with the A.Vogel Sore Issue 2 - June 2008 Antiviral Antibacterial Anti-inflammatory Analgetic Immunemodulatory Authors: Andy Suter, Medical Department Roland Schoop,
SELECTIVE AND DIFFERENTIAL MEDIA
 SELECTIVE AND DIFFERENTIAL MEDIA Selective and differential media are used to isolate or identify particular organisms. Selective media allow certain types of organisms to grow, and inhibit the growth
SELECTIVE AND DIFFERENTIAL MEDIA Selective and differential media are used to isolate or identify particular organisms. Selective media allow certain types of organisms to grow, and inhibit the growth
Urinary Tract Infections
 Urinary Tract Infections Leading cause of morbidity and health care expenditures in persons of all ages. An estimated 50 % of women report having had a UTI at some point in their lives. 8.3 million office
Urinary Tract Infections Leading cause of morbidity and health care expenditures in persons of all ages. An estimated 50 % of women report having had a UTI at some point in their lives. 8.3 million office
PACKAGE LEAFLET: INFORMATION FOR THE USER. PARACETAMOL MACOPHARMA 10 mg/ml, solution for infusion. Paracetamol
 PACKAGE LEAFLET: INFORMATION FOR THE USER PARACETAMOL MACOPHARMA 10 mg/ml, solution for infusion Paracetamol Read all of this leaflet carefully before you start using this medicine. Keep this leaflet.
PACKAGE LEAFLET: INFORMATION FOR THE USER PARACETAMOL MACOPHARMA 10 mg/ml, solution for infusion Paracetamol Read all of this leaflet carefully before you start using this medicine. Keep this leaflet.
APPENDIX B: UWHC SURGICAL ANTIMICROBIAL PROPHYLAXIS GUIDELINES
 APPENDIX B: UWHC SURGICAL ANTIMICROBIAL PROPHYLAXIS GUIDELINES Principles of prophylaxis 1) Use antimicrobials for surgical procedures where prophylactic antimicrobials have been found to be beneficial.
APPENDIX B: UWHC SURGICAL ANTIMICROBIAL PROPHYLAXIS GUIDELINES Principles of prophylaxis 1) Use antimicrobials for surgical procedures where prophylactic antimicrobials have been found to be beneficial.
1. NAME OF THE MEDICINAL PRODUCT. Impavido 10 mg capsules Impavido 50 mg capsules 2. QUALITATIVE AND QUANTITATIVE COMPOSITION. 1 capsule contains:
 1. NAME OF THE MEDICINAL PRODUCT Impavido 10 mg capsules Impavido 50 mg capsules 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 1 capsule contains: Impavido 10 mg capsules 10 mg Miltefosine. Impavido 50 mg
1. NAME OF THE MEDICINAL PRODUCT Impavido 10 mg capsules Impavido 50 mg capsules 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 1 capsule contains: Impavido 10 mg capsules 10 mg Miltefosine. Impavido 50 mg
One vial contains 500 U* of C1-inhibitor** After reconstitution the product contains 500 U/5 ml which correspond to a concentration of 100 U/ml.
 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Cetor 100 U/ml powder and solvent for solution for injection 2. QUALITATIVE AND QUANTITATIVE COMPOSITION One vial contains 500 U* of
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Cetor 100 U/ml powder and solvent for solution for injection 2. QUALITATIVE AND QUANTITATIVE COMPOSITION One vial contains 500 U* of
Microcyn Technology. Life-Altering Advance in Tissue Care
 Microcyn Technology Life-Altering Advance in Tissue Care The patented Microcyn Technology mimics the same oxychlorine composition as that manufactured by neutrophils in the body s immune system. Neutrophils
Microcyn Technology Life-Altering Advance in Tissue Care The patented Microcyn Technology mimics the same oxychlorine composition as that manufactured by neutrophils in the body s immune system. Neutrophils
Thioctacid 600 T Solution for Injection contains 600 mg alpha-lipoic acid
 Package Leaflet: Information for the User Thioctacid 600 T Solution for Injection contains 600 mg alpha-lipoic acid For use in adults Active substance: Alpha-lipoic acid, Trometamol salt (1:1) Read all
Package Leaflet: Information for the User Thioctacid 600 T Solution for Injection contains 600 mg alpha-lipoic acid For use in adults Active substance: Alpha-lipoic acid, Trometamol salt (1:1) Read all
PHOSPHATE-SANDOZ Tablets (High dose phosphate supplement)
 1 PHOSPHATE-SANDOZ Tablets (High dose phosphate supplement) PHOSPHATE-SANDOZ PHOSPHATE-SANDOZ Tablets are a high dose phosphate supplement containing sodium phosphate monobasic. The CAS registry number
1 PHOSPHATE-SANDOZ Tablets (High dose phosphate supplement) PHOSPHATE-SANDOZ PHOSPHATE-SANDOZ Tablets are a high dose phosphate supplement containing sodium phosphate monobasic. The CAS registry number
Basic Overview of Preclinical Toxicology Animal Models
 Basic Overview of Preclinical Toxicology Animal Models Charles D. Hebert, Ph.D., D.A.B.T. December 5, 2013 Outline Background In Vitro Toxicology In Vivo Toxicology Animal Models What is Toxicology? Background
Basic Overview of Preclinical Toxicology Animal Models Charles D. Hebert, Ph.D., D.A.B.T. December 5, 2013 Outline Background In Vitro Toxicology In Vivo Toxicology Animal Models What is Toxicology? Background
ORAL MEDICATIONS FOR MS! Gilenya and Aubagio
 ORAL MEDICATIONS FOR MS! Gilenya and Aubagio Champions against MS 4/20/13 Alexandra Goodyear, MD Stanford University Oral Medications Since 2010, 3 new oral medications for MS: Gilenya 2010 Aubagio 2012
ORAL MEDICATIONS FOR MS! Gilenya and Aubagio Champions against MS 4/20/13 Alexandra Goodyear, MD Stanford University Oral Medications Since 2010, 3 new oral medications for MS: Gilenya 2010 Aubagio 2012
SUMMARY OF PRODUCT CHARACTERISTICS. Albuman 200 g/l is a solution containing 200 g/l (20%) of total protein of which at least 95% is human albumin.
 Albuman 200 g/l SPC 01 December 2015 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Albuman 200 g/l solution for infusion 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Albuman 200 g/l
Albuman 200 g/l SPC 01 December 2015 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Albuman 200 g/l solution for infusion 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Albuman 200 g/l
Augmentin SR 1000mg Tablets
 The format of this leaflet as determined by the Ministry of Health and its content was checked and approved in December 2007 Physician Data Sheet Augmentin SR 1000mg Tablets TITLE Amoxicillin trihydrate
The format of this leaflet as determined by the Ministry of Health and its content was checked and approved in December 2007 Physician Data Sheet Augmentin SR 1000mg Tablets TITLE Amoxicillin trihydrate
CAPASTAT SULFATE CAPREOMYCIN FOR INJECTION, USP
 1 CAPASTAT SULFATE CAPREOMYCIN FOR INJECTION, USP PV 4801 AMP FOR INTRAMUSCULAR AND INTRAVENOUS INFUSION ONLY NOT FOR PEDIATRIC USE WARNINGS The use of Capastat Sulfate (Capreomycin for Injection, USP)
1 CAPASTAT SULFATE CAPREOMYCIN FOR INJECTION, USP PV 4801 AMP FOR INTRAMUSCULAR AND INTRAVENOUS INFUSION ONLY NOT FOR PEDIATRIC USE WARNINGS The use of Capastat Sulfate (Capreomycin for Injection, USP)
MERREM I.V. (meropenem for injection) FOR INTRAVENOUS USE ONLY
 Page 3 MERREM I.V. (meropenem for injection) FOR INTRAVENOUS USE ONLY To reduce the development of drug-resistant bacteria and maintain the effectiveness of MERREM I.V. (meropenem for injection) and other
Page 3 MERREM I.V. (meropenem for injection) FOR INTRAVENOUS USE ONLY To reduce the development of drug-resistant bacteria and maintain the effectiveness of MERREM I.V. (meropenem for injection) and other
Public Assessment Report. Decentralised Procedure. Cefixime 400 mg Film-coated Tablets PL 22805/0032 PL 22805/0033 UK/H/1532/001/DC UK/H/3965/001/DC
 Public Assessment Report Decentralised Procedure Cefixime 400 mg Film-coated Tablets PL 22805/0032 PL 22805/0033 UK/H/1532/001/DC UK/H/3965/001/DC Orchid Europe Ltd. MHRA PAR; CEFIXIME 400 MG FILM-COATED
Public Assessment Report Decentralised Procedure Cefixime 400 mg Film-coated Tablets PL 22805/0032 PL 22805/0033 UK/H/1532/001/DC UK/H/3965/001/DC Orchid Europe Ltd. MHRA PAR; CEFIXIME 400 MG FILM-COATED
OSMITROL - mannitol injection, solution Baxter Healthcare Corporation
 OSMITROL - mannitol injection, solution Baxter Healthcare Corporation DESCRIPTION OSMITROL Injection (Mannitol Injection, USP) is a sterile, nonpyrogenic solution of Mannitol, USP in a single dose container
OSMITROL - mannitol injection, solution Baxter Healthcare Corporation DESCRIPTION OSMITROL Injection (Mannitol Injection, USP) is a sterile, nonpyrogenic solution of Mannitol, USP in a single dose container
DOSAGE FORMS AND STRENGTHS Intravitreal implant containing dexamethasone 0.7 mg in the NOVADUR solid polymer drug delivery system.
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use OZURDEX safely and effectively. See full prescribing information for OZURDEX. OZURDEX (dexamethasone
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use OZURDEX safely and effectively. See full prescribing information for OZURDEX. OZURDEX (dexamethasone
1) Aug 2005 In Vitro Chromosomal Aberration Study Cytotoxicity (File: TS39) Conclusion No cytotoxicity can be detected at concentrations up to 100%.
 Endotoxicity and Cytotoxicity Studies 1) Aug 2005 In Vitro Chromosomal Aberration Study Cytotoxicity (File: TS39) Chinese Hamster Ovary (CHO) cells were grown in monolayer with and without metabolic activation.
Endotoxicity and Cytotoxicity Studies 1) Aug 2005 In Vitro Chromosomal Aberration Study Cytotoxicity (File: TS39) Chinese Hamster Ovary (CHO) cells were grown in monolayer with and without metabolic activation.
Certofix protect More certainty, more safety, more success
 Certofix protect More certainty, more safety, more success Highly effective, long-term protection against catheter infection Central Venous Catheters Antimicrobial modified hydrophilic surface We help
Certofix protect More certainty, more safety, more success Highly effective, long-term protection against catheter infection Central Venous Catheters Antimicrobial modified hydrophilic surface We help
RADIOPHARMACEUTICALS BASED ON MONOCLONAL ANTIBODIES
 RADIOPHARMACEUTICALS BASED ON MONOCLONAL ANTIBODIES Guideline Title Radiopharmaceuticals based on Monoclonal Antibodies Legislative basis Directives 65/65/EEC, 75/318/EEC as amended, Directive 89/343/EEC
RADIOPHARMACEUTICALS BASED ON MONOCLONAL ANTIBODIES Guideline Title Radiopharmaceuticals based on Monoclonal Antibodies Legislative basis Directives 65/65/EEC, 75/318/EEC as amended, Directive 89/343/EEC
A normal eye is protected by a layer of natural tears.
 A normal eye is protected by a layer of natural tears. If you need to use eye drops several times a day to make your eyes comfortably moist, you should consult your eye doctor. Helps increase tear production
A normal eye is protected by a layer of natural tears. If you need to use eye drops several times a day to make your eyes comfortably moist, you should consult your eye doctor. Helps increase tear production
INFUSE Bone Graft. Patient Information Brochure
 INFUSE Bone Graft Patient Information Brochure This Patient Guide is designed to help you decide whether or not to have surgery using INFUSE Bone Graft to treat your broken tibia (lower leg). There are
INFUSE Bone Graft Patient Information Brochure This Patient Guide is designed to help you decide whether or not to have surgery using INFUSE Bone Graft to treat your broken tibia (lower leg). There are
Buffered PFIZERPEN (penicillin G potassium) for Injection
 Buffered PFIZERPEN (penicillin G potassium) for Injection To reduce the development of drug-resistant bacteria and maintain the effectiveness of Pfizerpen and other antibacterial drugs, Pfizerpen should
Buffered PFIZERPEN (penicillin G potassium) for Injection To reduce the development of drug-resistant bacteria and maintain the effectiveness of Pfizerpen and other antibacterial drugs, Pfizerpen should
Analytical Specifications RIVAROXABAN
 Page 1 of 9 ANALYTE NAME AND STRUCTURE - RIVAROXABAN SYNONYMS Xarelto CATEGORY Anticoagulant TEST CODE PURPOSE Therapeutic Drug Monitoring GENERAL RELEVANCY BACKGROUND Xarelto (rivaroxaban) is an orally
Page 1 of 9 ANALYTE NAME AND STRUCTURE - RIVAROXABAN SYNONYMS Xarelto CATEGORY Anticoagulant TEST CODE PURPOSE Therapeutic Drug Monitoring GENERAL RELEVANCY BACKGROUND Xarelto (rivaroxaban) is an orally
Substance Abuse Treatment. Naltrexone for Extended-Release Injectable Suspension for Treatment of Alcohol Dependence
 Spring 2007 Volume 6 Issue 1 ADVISORY News for the Treatment Field Naltrexone for Extended-Release Injectable Suspension for Treatment of Alcohol Dependence What is naltrexone for extendedrelease injectable
Spring 2007 Volume 6 Issue 1 ADVISORY News for the Treatment Field Naltrexone for Extended-Release Injectable Suspension for Treatment of Alcohol Dependence What is naltrexone for extendedrelease injectable
FLOXIN Tablets (ofloxacin tablets)
 FLOXIN Tablets (ofloxacin tablets) WARNING: Fluoroquinolones, including FLOXIN, are associated with an increased risk of tendinitis and tendon rupture in all ages. This risk is further increased in older
FLOXIN Tablets (ofloxacin tablets) WARNING: Fluoroquinolones, including FLOXIN, are associated with an increased risk of tendinitis and tendon rupture in all ages. This risk is further increased in older
PHENYLEPHRINE HYDROCHLORIDE INJECTION USP
 PRESCRIBING INFORMATION PHENYLEPHRINE HYDROCHLORIDE INJECTION USP 10 mg/ml Sandoz Canada Inc. Date of Preparation: September 1992 145 Jules-Léger Date of Revision : January 13, 2011 Boucherville, QC, Canada
PRESCRIBING INFORMATION PHENYLEPHRINE HYDROCHLORIDE INJECTION USP 10 mg/ml Sandoz Canada Inc. Date of Preparation: September 1992 145 Jules-Léger Date of Revision : January 13, 2011 Boucherville, QC, Canada
Understanding your Tecfidera treatment
 Understanding your Tecfidera treatment Information for patients who have been prescribed treatment with Tecfidera. (dimethyl fumarate) Contents About Multiple Sclerosis (MS) What is MS? Symptoms of MS
Understanding your Tecfidera treatment Information for patients who have been prescribed treatment with Tecfidera. (dimethyl fumarate) Contents About Multiple Sclerosis (MS) What is MS? Symptoms of MS
Understanding your Tecfidera treatment
 Understanding your Tecfidera treatment Information for patients who have been prescribed treatment with Tecfidera. (dimethyl fumarate) Contents About Multiple Sclerosis (MS) What is MS? Symptoms of MS
Understanding your Tecfidera treatment Information for patients who have been prescribed treatment with Tecfidera. (dimethyl fumarate) Contents About Multiple Sclerosis (MS) What is MS? Symptoms of MS
Reference ID: 3482803
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use EVZIO safely and effectively. See full prescribing information for EVZIO. EVZIO (naloxone hydrochloride
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use EVZIO safely and effectively. See full prescribing information for EVZIO. EVZIO (naloxone hydrochloride
* Sections or subsections omitted from the full prescribing information are not listed
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use RECLAST safely and effectively. See full prescribing information for RECLAST. Reclast (zoledronic
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use RECLAST safely and effectively. See full prescribing information for RECLAST. Reclast (zoledronic
ALLERGENIC EXTRACT. Prescription Set of Serial Dilutions (or Maintenance Vial (s)) INSTRUCTIONS FOR USE. U.S. Government License No.
 ALLERGENIC EXTRACT Prescription Set of Serial Dilutions (or Maintenance Vial (s)) INSTRUCTIONS FOR USE U.S. Government License No. 308 Revised 07/04 PO Box 800 Lenoir, NC 28645 USA DESCRIPTION This set
ALLERGENIC EXTRACT Prescription Set of Serial Dilutions (or Maintenance Vial (s)) INSTRUCTIONS FOR USE U.S. Government License No. 308 Revised 07/04 PO Box 800 Lenoir, NC 28645 USA DESCRIPTION This set
Package leaflet: Information for the patient. Dorzolamide Mylan 20 mg/ml Eye drops, Solution Dorzolamide
 Package leaflet: Information for the patient Dorzolamide Mylan 20 mg/ml Eye drops, Solution Dorzolamide Read all of this leaflet carefully before you start using this medicine because it contains important
Package leaflet: Information for the patient Dorzolamide Mylan 20 mg/ml Eye drops, Solution Dorzolamide Read all of this leaflet carefully before you start using this medicine because it contains important
SUMMARY OF PRODUCT CHARACTERISTICS
 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Aknemycin Plus 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 100 g of solution contains: Erythromycin 4.0 g, Tretinoin 0.025 g. For a full
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Aknemycin Plus 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 100 g of solution contains: Erythromycin 4.0 g, Tretinoin 0.025 g. For a full
SUMMARY OF PRODUCT CHARACTERISTICS 2. QUALITATIVE AND QUANTITATIVE COMPOSITION
 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Levoxa 250 mg film-coated tablets Levoxa 500 mg film-coated tablets 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each film-coated tablet
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Levoxa 250 mg film-coated tablets Levoxa 500 mg film-coated tablets 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each film-coated tablet
Selected Requirements of Prescribing Information
 The Selected Requirements of Prescribing Information (SRPI) is a checklist of 42 important format prescribing information (PI) items based on labeling regulations [21 CFR 201.56(d) and 201.57] and guidances.
The Selected Requirements of Prescribing Information (SRPI) is a checklist of 42 important format prescribing information (PI) items based on labeling regulations [21 CFR 201.56(d) and 201.57] and guidances.
Primary Options for Acute Care: Management of Adult Cellulitis
 Primary Options for Acute Care: Management of Adult Cellulitis These guidelines provide support for IV Management of Cellulitis in the community for initial oral treatment has failed. This is to be used
Primary Options for Acute Care: Management of Adult Cellulitis These guidelines provide support for IV Management of Cellulitis in the community for initial oral treatment has failed. This is to be used
SUMMARY OF THE RISK MANAGEMENT PLAN (by medicinal product)
 PART VI SUMMARY OF THE RISK MANAGEMENT PLAN (by medicinal product) Format and content of the summary of the RMP The summary of the RMP part VI contains information based on RMP modules SI, SVIII and RMP
PART VI SUMMARY OF THE RISK MANAGEMENT PLAN (by medicinal product) Format and content of the summary of the RMP The summary of the RMP part VI contains information based on RMP modules SI, SVIII and RMP
UTILIZATION of PLASMA ACTIVATED WATER in Biotechnology, Pharmacology and Medicine. JSC TECHNOSYSTEM-ECO Moscow, Russia April, 2009
 UTILIZATION of PLASMA ACTIVATED WATER in Biotechnology, Pharmacology and Medicine JSC TECHNOSYSTEM-ECO Moscow, Russia April, 2009 METHOD of WATER ACTIVATION with PLASMA of GAS DISCHARGE ANODE VACUUM WATER
UTILIZATION of PLASMA ACTIVATED WATER in Biotechnology, Pharmacology and Medicine JSC TECHNOSYSTEM-ECO Moscow, Russia April, 2009 METHOD of WATER ACTIVATION with PLASMA of GAS DISCHARGE ANODE VACUUM WATER
ATROVENT HFA (ipratropium bromide HFA) Inhalation Aerosol FOR ORAL INHALATION ONLY. Initial U.S. Approval: 2004
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use ATROVENT HFA safely and effectively. See full prescribing information for ATROVENT HFA. ATROVENT
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use ATROVENT HFA safely and effectively. See full prescribing information for ATROVENT HFA. ATROVENT
AMOXICILLIN 250MG POWDER FOR SOLUTION FOR INJECTION OR INFUSION (PL 24610/0010)
 AMOXICILLIN 250MG POWDER FOR SOLUTION FOR INJECTION OR INFUSION (PL 24610/0010) AMOXICILLIN 500MG POWDER FOR SOLUTION FOR INJECTION OR INFUSION (PL 24610/0011) AMOXICILLIN 1G POWDER FOR SOLUTION FOR INJECTION
AMOXICILLIN 250MG POWDER FOR SOLUTION FOR INJECTION OR INFUSION (PL 24610/0010) AMOXICILLIN 500MG POWDER FOR SOLUTION FOR INJECTION OR INFUSION (PL 24610/0011) AMOXICILLIN 1G POWDER FOR SOLUTION FOR INJECTION
MATERIAL SAFETY DATA SHEET
 MATERIAL SAFETY DATA SHEET 1. IDENTIFICATION OF THE SUBSTANCE AND THE COMPANY Material Manufacturer Distributor Losartan Potassium and Hydrochlorothiazide Tablets 100 mg/12.5 mg Lupin Limited Goa 403 722
MATERIAL SAFETY DATA SHEET 1. IDENTIFICATION OF THE SUBSTANCE AND THE COMPANY Material Manufacturer Distributor Losartan Potassium and Hydrochlorothiazide Tablets 100 mg/12.5 mg Lupin Limited Goa 403 722
