Clopidogrel versus Aspirin and Esomeprazole to Prevent Recurrent Ulcer Bleeding
|
|
|
- Sara Carr
- 7 years ago
- Views:
Transcription
1 The new england journal of medicine original article Clopidogrel versus Aspirin and Esomeprazole to Prevent Recurrent Ulcer Bleeding Francis K.L. Chan, M.D., Jessica Y.L. Ching, M.P.H., Lawrence C.T. Hung, M.D., Vincent W.S. Wong, M.D., Vincent K.S. Leung, M.D., Nelson N.S. Kung, M.D., Aric J. Hui, M.D., Justin C.Y. Wu, M.D., Wai K. Leung, M.D., Vivian W.Y. Lee, Pharm.D., Kenneth K.C. Lee, Ph.D., Yuk T. Lee, M.D., James Y.W. Lau, M.D., Ka F. To, M.D., Henry L.Y. Chan, M.D., S.C. Sydney Chung, M.D., and Joseph J.Y. Sung, M.D., Ph.D. abstract From the Departments of Medicine and Therapeutics (F.K.L.C., J.Y.L.C., L.C.T.H., V.W.S.W., A.J.H., J.C.Y.W., W.K.L., Y.T.L., H.L.Y.C., J.J.Y.S.), the School of Pharmacy (V.W.Y.L., K.K.C.L.), and the Departments of Surgery (J.Y.W.L., S.C.S.C.) and Anatomical and Cellular Pathology (K.F.T.), Prince of Wales Hospital, Chinese University of Hong Kong; and the Medical Unit, United Christian Hospital (V.K.S.L., N.N.S.K.) all in Hong Kong. Address reprint requests to Dr. Francis K.L. Chan at the Department of Medicine and Therapeutics, Prince of Wales Hospital, Chinese University of Hong Kong, Ngan Shing St., Shatin, Hong Kong, China, or at edu.hk. N Engl J Med 2005;352: Copyright 2005 Massachusetts Medical Society. background Concurrent therapy with a proton-pump inhibitor is a standard treatment for patients receiving aspirin who are at risk for ulcer. Current U.S. guidelines also recommend clopidrogel for patients who have major gastrointestinal intolerance of aspirin. We compared clopidogrel with aspirin plus esomeprazole for the prevention of recurrent bleeding from ulcers in high-risk patients. methods We studied patients who took aspirin to prevent vascular diseases and who presented with ulcer bleeding. After the ulcers had healed, we randomly assigned patients who were negative for Helicobacter pylori to receive either 75 mg of clopidogrel daily plus esomeprazole placebo twice daily or 80 mg of aspirin daily plus 20 mg of esomeprazole twice daily for 12 months. The end point was recurrent ulcer bleeding. results We enrolled 320 patients (161 patients assigned to receive clopidogrel and 159 to receive aspirin plus esomeprazole). Recurrent ulcer bleeding occurred in 13 patients receiving clopidogrel and 1 receiving aspirin plus esomeprazole. The cumulative incidence of recurrent bleeding during the 12-month period was 8.6 percent (95 percent confidence interval, 4.1 to 13.1 percent) among patients who received clopidogrel and 0.7 percent (95 percent confidence interval, 0 to 2.0 percent) among those who received aspirin plus esomeprazole (difference, 7.9 percentage points; 95 percent confidence interval for the difference, 3.4 to 12.4; P=0.001). conclusions Among patients with a history of aspirin-induced ulcer bleeding whose ulcers had healed before they received the study treatment, aspirin plus esomeprazole was superior to clopidogrel in the prevention of recurrent ulcer bleeding. Our finding does not support the current recommendation that patients with major gastrointestinal intolerance of aspirin be given clopidogrel. 238
2 it is estimated that during the past two decades 50 million Americans have started taking aspirin for the prevention of heart attack and stroke. 1 However, aspirin doubles the risk of upper gastrointestinal bleeding even at doses as low as 75 mg daily. 2 A history of upper gastrointestinal bleeding from an ulcer is the most important risk factor for subsequent upper gastrointestinal bleeding in patients taking aspirin. 3,4 Up to 15 percent of those taking aspirin who have a history of bleeding from ulcers had recurrent bleeding within one year. 5 Proton-pump inhibitors reduce the risk of aspirin-induced ulcer bleeding, 5-7 and combination therapy with proton-pump inhibitors has been advocated for patients at high risk for ulcer bleeding who are taking aspirin. 8,9 However, compliance with the drug regimen may limit the usefulness of the combination therapy, especially among patients who are already receiving multiple drugs. An alternative strategy is to replace aspirin with another antiplatelet drug that does not induce ulcer. Clopidogrel, which inhibits the platelet adenosine diphosphate receptor, has been shown to prevent ischemic events The Food and Drug Administration has approved clopidogrel for the treatment of vascular diseases. 14 In healthy volunteers, clopidogrel did not induce gastric damage. 15 It was reported to be more efficacious and to induce fewer episodes of gastrointestinal bleeding than aspirin. 10 Although the combination of clopidogrel and aspirin increases the overall risk of bleeding, 11 a recent analysis indicated that the excess risk of bleeding was attributed to the dose-dependent ulcerogenic effect of aspirin. 16 The American College of Cardiology American Heart Association guidelines recommend the use of clopidogrel for hospitalized patients with a coronary syndrome who are unable to take aspirin because of major gastrointestinal intolerance (class IA recommendation). 17,18 However, there has been no prospective trial to assess whether clopidogrel is an alternative to aspirin plus a proton-pump inhibitor for patients at risk for ulcer. Our study was a 12-month, prospective, randomized, double-blind trial that compared clopidogrel with aspirin plus esomeprazole for patients who had previous aspirin-induced ulcer bleeding. We hypothesized that after the ulcers had healed, clopidogrel would not be inferior to aspirin plus esomeprazole in the prevention of recurrent ulcer bleeding among these high-risk patients. methods study population The study was conducted at the Prince of Wales Hospital in Hong Kong. We screened consecutive users of low-dose aspirin (325 mg or less per day) who presented with upper gastrointestinal bleeding. The patients underwent endoscopy within 24 hours after presentation to identify the site of the bleeding. During endoscopy, three biopsy specimens were obtained from the antrum and two from the body of the stomach for a rapid urease test (CLO, Delta West) and for histologic examination for Helicobacter pylori with the use of hematoxylin and eosin stain and Warthin Starry stain, if necessary. Patients with H. pylori infection were treated for one week with a triple-drug regimen that included a proton-pump inhibitor. Aspirin was withheld during this period. All patients received protonpump inhibitors to promote the healing of ulcers. Follow-up endoscopy was performed eight weeks after eradication therapy, while the patients were not taking acid-suppressing drugs. H. pylori was considered to be present if any portion of the specimen was positive; it was considered to be absent or eradicated when all above-noted test results were negative. Patients were considered eligible for inclusion if they had endoscopically confirmed ulcer healing, negative results on the test for H. pylori or successful eradication of H. pylori and anticipated regular use of antiplatelet therapy for the duration of the trial. The exclusion criteria were concomitant use of nonsteroidal antiinflammatory drugs (NSAIDs), cyclooxygenase-2 inhibitors, anticoagulant agents, other antiplatelet drugs, or corticosteroids; a history of gastric surgery other than a patch repair; allergy to aspirin or clopidogrel; and the presence of erosive esophagitis, gastric-outlet obstruction, renal failure requiring dialysis, terminal illness, or cancer. treatment Eligible patients were randomly assigned to receive either 75 mg of clopidogrel (Plavix, Sanofi-Synthelabo) daily plus esomeprazole placebo twice daily or 80 mg of aspirin daily plus 20 mg of esomeprazole (Nexium, AstraZeneca) twice daily for 12 months. Randomization was carried out with the use of a computer-generated list of random numbers. An independent staff member assigned the treatments according to consecutive numbers that were kept in sealed envelopes. We purchased the drugs and 239
3 The new england journal of medicine repackaged them so that clopidogrel and aspirin appeared as identical blue capsules and esomeprazole and its placebo appeared as identical red capsules, according to the International Good Manufacturing Practice Guidelines for Pharmaceuticals. Consecutively numbered, sealed bottles of the study drugs were dispensed by a research nurse. Anticoagulant agents, NSAIDs, cyclooxygenase-2 inhibitors, over-the-counter analgesics (including herbal products), corticosteroids, misoprostol, histamine H 2 -receptor antagonists, sucralfate, antiplatelet drugs other than the study drugs, and proton-pump inhibitors were prohibited. assessment After random assignment, the patients returned at month 1, month 3, and every three months thereafter until the end of the study. At each visit, hemoglobin levels and serum biochemical values were measured, and drug compliance, the use of other medications including over-the-counter drugs, and safety were assessed. Drug compliance was assessed with the use of pill counts. We also used a territory-wide electronic prescription database that captured all prescriptions written within the public health sector; and we retrieved over-the-counter drugs and prescriptions from the patients, their families, and their primary care doctors in order to identify any concomitant therapy with NSAIDs or aspirin. The assessment of safety was based on physical examination, laboratory tests, and observed or reported adverse events. A direct telephone line was provided for patients and physicians to use to report adverse events that occurred between the scheduled visits with the study physicians. Patients who discontinued the study drugs prematurely were followed until the end of the study, to determine whether gastrointestinal events had occurred. The local ethics committee approved the protocol of the study and monitored the patients safety data. All patients gave written informed consent. An independent, blinded adjudication committee reviewed the data to determine which patients had reached the study end points according to the prespecified criteria. end points The primary end point was recurrent ulcer bleeding as defined according to prespecified criteria namely, hematemesis or melena documented by the admitting physician, with ulcers or bleeding erosions confirmed on endoscopy, or a decrease in the hemoglobin level of at least 2 g per deciliter in the presence of endoscopically documented ulcers or bleeding erosions. An ulcer was defined as a circumscribed mucosal break at least 0.5 cm in diameter and with a perceptible depth; a bleeding erosion was defined as a flat mucosal break of any size that occurred in the presence of blood in the stomach. Endoscopy was performed in a treatment-blinded fashion. Only events that were confirmed by the adjudication committee and that occurred during treatment or within 28 days after the discontinuation of treatment were included in the analysis. The secondary end point was lower gastrointestinal bleeding, which was defined by either melena or rectal bleeding requiring hospitalization or transfusion, with negative results on upper endoscopy, or by a decrease in the hemoglobin level of at least 2 g per deciliter in association with a positive fecal occult blood test and negative results on upper endoscopy. Eligible patients underwent colonoscopy to locate the source of bleeding; those with a negative result were considered to have gastrointestinal bleeding of an obscure origin. Extragastrointestinal bleeding included intracranial hemorrhage and other bleeding disorders such as hematuria leading to hospitalization, hypotension, the need for transfusion, or the need to discontinue the study medication. statistical analysis We determined the size of the sample on the assumptions that about 1.5 percent of patients receiving aspirin plus esomeprazole would have recurrent ulcer bleeding within 12 months 9 and that clopidogrel would not be inferior to aspirin plus esomeprazole if the upper limit of the 95 percent confidence interval for the difference in the rates of recurrent ulcer bleeding did not exceed 4 percentage points. Accordingly, a sample size of 145 patients in each of the two treatment groups was necessary to give the study a power of 80 percent and a 5 percent level of significance with the use of a onesided equivalence test of proportions. 19 Assuming that 10 percent of patients did not complete followup, a total sample of 319 patients would be required. No interim analysis was performed. The data analysis was carried out exclusively by the data review committee. We used the Kaplan Meier method to estimate the likelihood of reaching the end points in the intention-to-treat population, 20 which was defined as all patients who had taken at least one dose of 240
4 the study medication. The log-rank test was used to compare time-to-event curves for the two groups (SPSS software, version 10.0). Failure to take at least 80 percent of the study drugs or the use of prohibited drugs was considered a violation of the protocol. All P values and 95 percent confidence intervals were two-sided. results patients Between September 2001 and June 2003, we screened 492 consecutive patients who were taking low-dose aspirin and who presented with hematemesis, melena, or both, and we enrolled a total of 320 of these patients. The reasons for exclusion were terminal illness (in 66 patients), cancer (43), end-stage renal failure (17), lower gastrointestinal bleeding (4), previous gastric surgery (2), gastric-outlet obstruction (1), erosive esophagitis (1), aspirin allergy (1), and concomitant treatment with anticoagulant agents (8), NSAIDs (3), or other antiplatelet drugs (1); in addition, 25 patients declined participation. The intention-to-treat analysis included all 320 patients: 161 patients were randomly assigned to receive clopidogrel, and 159 patients to receive aspirin plus esomeprazole (Table 1). The median followup was 12 months (range, 0.3 to 12) in both groups. All of the patients in the clopidogrel group and all but three patients in the aspirin-plus-esomeprazole group completed follow-up. Ninety-four percent of the patients in each group took at least 80 percent of the assigned study drugs. The rates of discontinuation, excluding patients who reached the primary end point, were similar in the two groups 11.8 percent in the clopidogrel group (4.3 percent because of adverse events, 1.9 percent because of recurrent ischemic events, 0.6 percent owing to withdrawal of consent, and 5.0 percent for other reasons) and 8.8 percent in the aspirin-plus-esomeprazole group (1.9 percent because of adverse events, 3.8 percent owing to withdrawal of consent, and 3.1 percent for other reasons). No patient who discontinued medications early had recurrent ulcer bleeding or anemia within the study period. gastrointestinal events Thirty-four cases of suspected serious gastrointestinal events were evaluated by the adjudication committee. The committee identified 14 cases of recurrent ulcer bleeding, 13 in the clopidogrel group (6 gastric ulcers, 5 duodenal ulcers, 2 both gastric and duodenal ulcers) and 1 (duodenal ulcer) in the aspirin-plus-esomeprazole group. All the patients with recurrent bleeding had presented with recurrent melena, hematemesis, or both, requiring hospitalization. The median diameter of the recurrent ulcers was 0.5 cm (range, 0.5 to 3.0 cm). Five patients required endoscopic control of active bleeding, and eight required transfusion (median, 3.5 units; range, 1 to 9). In 10 of the 14 patients with recurrent bleeding (71.4 percent), the ulcers recurred at their previous locations. None of the 14 patients had recurrent H. pylori infection. Two patients with recurrent ulcer bleeding in the clopidogrel group used concomitant NSAIDs. The cumulative incidence of recurrent ulcer bleeding during the 12-month study period was 8.6 percent (95 percent confidence interval, 4.1 to 13.1 percent) among patients who received clopidogrel and 0.7 percent (95 percent confidence interval, 0 to 2.0 percent) among those who received aspirin plus esomeprazole (difference, 7.9 percentage points; 95 percent confidence interval for the difference, 3.4 to 12.4; P=0.001) (Table 2 and Fig. 1). A perprotocol analysis of 293 patients showed that the cumulative incidence of recurrent bleeding was 7.5 percent (95 percent confidence interval, 3.0 to 11.9 percent) in the clopidogrel group and 0.7 percent (95 percent confidence interval, 0 to 2.2 percent) in the aspirin-plus-esomeprazole group (difference, 6.8 percentage points; 95 percent confidence interval for the difference, 2.3 to 11.3; P=0.005). Of the 20 patients who were found on adjudication not to have recurrent ulcer bleeding, 4 were found to have gastrointestinal cancer (3 had colon cancer and 1 had cholangiocarcinoma), and 2 had anemia that was not due to gastrointestinal blood loss. Of 14 patients who met the prespecified criteria for lower gastrointestinal bleeding, 7 received clopidogrel (6 had gastrointestinal bleeding of obscure origin and 1 had a bleeding rectal ulcer) and 7 received aspirin plus esomeprazole (5 had gastrointestinal bleeding of obscure origin, 1 had hemorrhoidal bleeding, and 1 had angiodysplasia). The cumulative incidence of lower gastrointestinal bleeding was 4.6 percent (95 percent confidence interval, 1.3 to 7.9 percent) in the clopidogrel group and 4.6 percent (95 percent confidence interval, 1.3 to 8.0 percent) in the aspirin-plus-esomeprazole group (P=0.98). 241
5 The new england journal of medicine Table 1. Baseline Characteristics of the 320 Patients.* Characteristic Clopidogrel (N=161) Aspirin plus Esomeprazole (N=159) Male sex no. (%) 108 (67.1) 103 (64.8) Age yr 72.1± ±9.5 Current smoking no. (%) 21 (13.0) 13 (8.2) Current alcohol consumption no. (%) 13 (8.1) 8 (5.0) Source of bleeding no. (%) Gastric ulcer 94 (58.4) 74 (46.5) Duodenal ulcer 48 (29.8) 61 (38.4) Gastric and duodenal ulcers 9 (5.6) 18 (11.3) Gastric erosions 8 (5.0) 6 (3.8) Duodenal erosions 2 (1.2) 0 Multiple episodes of ulcer bleeding no. (%) 28 (17.4) 33 (20.8) Diameter of ulcer cm 1.3± ±3.3 Diameter 2 cm no. (%) 20 (12.4) 21 (13.2) Ulcer with active bleeding or nonbleeding 45 (28.0) 54 (34.0) visible vessels no. (%) Transfusion required no. (%) 78 (48.4) 89 (56.0) Ischemic condition no. (%) Coronary heart disease 88 (54.7) 78 (49.1) Cerebrovascular insufficiency 55 (34.2) 66 (41.5) Peripheral vascular disease 8 (5.0) 6 (3.8) Multiple ischemic conditions no. (%) 10 (6.2) 9 (5.7) Serum creatinine >1.2 mg/dl no. (%) 49 (30.4) 47 (29.6) Previous H. pylori infection no. (%) 74 (46.0) 74 (46.5) * Plus minus values are means ±SD. To convert the value for creatinine to micromoles per liter, multiply by extragastrointestinal bleeding and other adverse events Three patients who received clopidogrel had extragastrointestinal bleeding: two patients had intracranial hemorrhage, and one had severe hematuria requiring hospitalization for transfusion. None of the patients who received aspirin plus esomeprazole had extragastrointestinal bleeding. Other adverse events occurred in 9.4 percent of the clopidogrel group (7.5 percent of patients had dyspepsia, and 1.9 percent had allergy) and in 4.4 percent of the aspirin-plus-esomeprazole group (2.5 percent of patients had dyspepsia and 1.9 percent had allergy). Recurrent ischemic events occurred in 9 patients in the clopidogrel group (1 patient had myocardial infarction, 6 had unstable angina, and 2 had cerebrovascular insufficiency) and in 11 patients in the aspirin-plus-esomeprazole group (1 had myocardial infarction, 7 had unstable angina, and 3 had cerebrovascular insufficiency). mortality Of 12 patients who died, 8 were in the clopidogrel group (1 patient died from myocardial infarction, 1 from an intracranial hemorrhage, 1 from heart failure, 3 from sepsis, and 2 from uncertain causes), and 4 were in the aspirin-plus-esomeprazole group (1 patient died from myocardial infarction, 1 from cerebrovascular insufficiency, 1 from renal failure, and 1 from uncertain causes). We tested the hypothesis that clopidogrel would not be inferior to aspirin plus esomeprazole in the prevention of recurrent ulcer bleeding in high-risk patients. The patients enrolled in this study had multiple risk factors, including a recent history of aspirin-induced ulcer bleeding, advanced age, and coexisting conditions. We found that among these high-risk patients who received clopidogrel after their ulcers had healed, the incidence of recurrent ulcer bleeding was unacceptably high: 8.6 percent of the patients had recurrent bleeding during the 12-month period of the study, as compared with only 0.7 percent of the patients receiving aspirin plus esomeprazole. This finding is not consistent with the current American College of Cardiology American Heart Association practice guidelines, which recommend the use of clopidogrel as an alternative antiplatelet agent for patients who have major gastrointestinal intolerance of aspirin. 18 Current evidence regarding the gastrointestinal safety of clopidogrel was derived from a secondary analysis of studies that did not use prespecified criteria to report gastrointestinal complications. 10,11 Although one study found a lower incidence of gastrointestinal bleeding among patients receiving clopidogrel than among those receiving aspirin, 10 a relatively high dose of aspirin (325 mg daily) was used as the comparator. Our results raise doubt about the gastrointestinal safety of clopidogrel even in the absence of active ulcers. Although all the patients had confirmed ulcer healing before undergoing randomization, those in whom upper gastrointestinal bleeding recurred actually had bleeding from recurrent ulcers. None had recurrent H. pylori infection. Only two patients with recurrent bleeding used concomitant NSAIDs. This finding was consistent with a retrodiscussion 242
6 Table 2. Kaplan Meier Estimates of the Likelihood of Recurrent Ulcer Bleeding and Lower Gastrointestinal (GI) Bleeding at 12 Months. Type of Bleeding Probability of Bleeding (95% CI)* P Value Recurrent ulcer bleeding Lower GI bleeding Cumulative Incidence of Recurrent Ulcer Bleeding (%) No. at Risk Clopidogrel 161 Aspirin plus 159 esomeprazole Clopidogrel * CI denotes confidence interval Aspirin plus Esomeprazole Follow-up (months) Figure 1. Cumulative Incidence of Recurrent Ulcer Bleeding in the Group Receiving Clopidogrel and the Group Receiving Aspirin plus Esomeprazole. The difference between the groups was significant (P=0.001 by the log-rank test). spective study that reported that 12 percent of patients with a history of ulcer who took clopidogrel had ulcer bleeding within one year. 21 The mechanisms leading to recurrent ulcer bleeding among patients receiving clopidogrel are unknown. Studies in animals have shown that platelet adenosine diphosphate receptor antagonists impair the healing of gastric ulcers by suppressing the release of platelet-derived growth factors. 22 We speculate that clopidogrel may induce recurrent ulcers in the previously damaged gastric mucosal barrier, as suggested by the high rate of recurrence at the previous location (71.4 percent) in our study. Alternatively, patients who have major coexisting conditions may have a predisposition to the development of ulcers even in the absence of H. pylori infection or the use of NSAIDs. 23 Clopidogrel probably provoked bleeding from recurrent ulcers in these high-risk patients. The optimal dose of proton-pump inhibitors for patients at high risk from the use of aspirin remains undefined. One study showed that among aspirin users with a history of ulcer bleeding who received lansoprazole at 30 mg once daily, the incidence of recurrent bleeding over 12 months was 1.6 percent. However, the upper limit of the 95 percent confidence interval was as high as 9.0 percent. 5 We used a twice-daily dose of a protonpump inhibitor to provide better acid control than the once-daily dose. 24,25 Our study has several limitations. First, the risk reduction achieved by clopidogrel or aspirin plus esomeprazole could not be determined, because we did not include a group of patients with a history of ulcer bleeding who used aspirin without prophylaxis. A previous study reported that about 15 percent of patients with a history of ulcer bleeding who used aspirin had recurrent bleeding within one year. 5 It would therefore be unethical to prescribe aspirin without prophylaxis for high-risk patients. Second, whether genetic variation in the metabolism of aspirin and proton-pump inhibitors among racial and ethnic groups has any effect on the risk of bleeding and the efficacy of treatment remains unknown. Third, because the study drugs were repackaged from the form available commercially, there may have been differences in uptake and absorption that changed the therapeutic efficacy of the drugs or had adverse effects. In summary, among patients with a history of aspirin-induced ulcer bleeding, aspirin plus esomeprazole was superior to clopidogrel for the prevention of recurrent bleeding. Our observations do not support the current recommendation that clopidogrel be used for patients who have major gastrointestinal intolerance of aspirin. Supported by a grant from the Division of Gastroenterology and Hepatology at the Chinese University of Hong Kong. Dr. Chan reports having received consulting fees from Pfizer and TAP Pharmaceutical Products, and Dr. J. Sung consulting fees from TAP Pharmaceutical Products. We are indebted to Dr. S.K. Lo, University of Sydney, for advice on the statistical analysis; to Bing-yee Suen, Priscilla Siu, M.Y. Yung, Jessica Leung, Franco Lai, and the Team Two surgical colleagues; and to the nursing staff of the Endoscopy Center at the Prince of Wales Hospital. Difference between the Groups percentage percent points 8.6 (4.1 to 13.1) 0.7 (0 to 2.0) 7.9 (3.4 to 12.4) (1.3 to 7.9) 4.6 (1.3 to 8.0) 0.0 ( 4.6 to 4.6) 0.98 Clopidogrel Aspirin plus esomeprazole
7 references 1. Derry S, Loke YK. Risk of gastrointestinal haemorrhage with long term use of aspirin: meta-analysis. BMJ 2000;321: Weil J, Colin-Jones D, Langman M, et al. Prophylactic aspirin and risk of peptic ulcer bleeding. BMJ 1995;310: Garcia Rodriguez LA, Jick H. Risk of upper gastrointestinal bleeding and perforation associated with individual non-steroidal anti-inflammatory drugs. Lancet 1994;343: [Erratum, Lancet 1994;343:1048.] 4. Lanas A, Bajador E, Serrano P, et al. Nitrovasodilators, low-dose aspirin, other nonsteroidal antiinflammatory drugs, and the risk of upper gastrointestinal bleeding. N Engl J Med 2000;343: Lai KC, Lam SK, Chu KM, et al. Lansoprazole for the prevention of recurrences of ulcer complications from long-term lowdose aspirin use. N Engl J Med 2002;346: Garcia Rodriguez LA, Ruigomez A. Secondary prevention of upper gastrointestinal bleeding associated with maintenance acidsuppressing treatment in patients with peptic ulcer bleed. Epidemiology 1999;10: Chan FK, Chung SC, Suen BY, et al. Preventing recurrent upper gastrointestinal bleeding in patients with Helicobacter pylori infection who are taking low-dose aspirin or naproxen. N Engl J Med 2001;344: Laine L. Approaches to nonsteroidal anti-inflammatory drug use in the high-risk patient. Gastroenterology 2001;120: Peura DA, Malfertheiner P. Dichotomies and directions in acid-related disorders. Aliment Pharmacol Ther 2004;19:Suppl 1: CAPRIE Steering Committee. A randomised, blinded, trial of clopidogrel versus aspirin in patients at risk of ischaemic events (CAPRIE). Lancet 1996;348: The Clopidogrel in Unstable Angina to Prevent Recurrent Events Trial Investigators. Effects of clopidogrel in addition to aspirin in patients with acute coronary syndromes without ST-segment elevation. N Engl J Med 2001;345: [Errata, N Engl J Med 2001;345:1506, 1716.] 12. Mehta SR, Yusuf S, Peters RJ, et al. Effects of pretreatment with clopidogrel and aspirin followed by long-term therapy in patients undergoing percutaneous coronary intervention: the PCI-CURE study. Lancet 2001;358: Steinhubl SR, Berger PB, Mann JT III, et al. Early and sustained dual oral antiplatelet therapy following percutaneous coronary intervention: a randomized controlled trial. JAMA 2002;288: [Erratum, JAMA 2003;289:987.] 14. Center for Drug Evaluation and Research. Approval package for application number /SE1-019 (Plavix). (Accessed December 27, 2004, at foi/nda/2002/20-839s019_clopidogrel%20 Bisulfate_ prntlbl.pdf ) 15. Fork FT, Lafolie P, Toth E, Lindgarde F. Gastroduodenal tolerance of 75 mg clopidogrel versus 325 mg aspirin in healthy volunteers: a gastroscopic study. Scand J Gastroenterol 2000;35: Peters RJ, Mehta SR, Fox KA, et al. Effects of aspirin dose when used alone or in combination with clopidogrel in patients with acute coronary syndromes: observations from the Clopidogrel in Unstable angina to prevent Recurrent Events (CURE) study. Circulation 2003;108: Braunwald E, Antman EM, Beasley JW, et al. ACC/AHA guideline update for the management of patients with unstable angina and non-st-segment elevation myocardial infarction 2002: summary article: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Committee on the Management of Patients with Unstable Angina). Circulation 2002;106: ACC/AHA 2002 guideline update for the management of patients with unstable angina and non-st-segment elevation myocardial infarction. (Accessed December 27, 2004, at guidelines/unstable/iii_hospital.htm#iii_ B1.) 19. Roebruck P, Kuhn A. Comparison of tests and sample-size formulae for proving therapeutic equivalence based on the difference of binomial probabilities. Stat Med 1995;14: Kaplan EL, Meier P. Nonparametric estimation from incomplete observations. J Am Stat Assoc 1958;53: Ng FH, Wong SY, Chang CM, et al. High incidence of clopidogrel-associated gastrointestinal bleeding in patients with previous peptic ulcer disease. Aliment Pharmacol Ther 2003;18: Ma L, Elliott SN, Cirino G, Buret A, Ignarro LJ, Wallace JL. Platelets modulate gastric ulcer healing: role of endostatin and vascular endothelial growth factor release. Proc Natl Acad Sci U S A 2001;98: Chan HL, Wu JC, Chan FK, et al. Is non- Helicobacter pylori, non-nsaid peptic ulcer a common cause of upper GI bleeding? A prospective study of 977 patients. Gastrointest Endosc 2001;53: Katz PO, Castell DO, Chen Y, Andersson T, Sostek MB. Intragastric acid suppression and pharmacokinetics of twice-daily esomeprazole: a randomized, three-way crossover study. Aliment Pharmacol Ther 2004; 20: Hammer J, Schmidt B. Effect of splitting the dose of esomeprazole on gastric acidity and nocturnal acid breakthrough. Aliment Pharmacol Ther 2004;19: Copyright 2005 Massachusetts Medical Society. 244
Guidelines for Use of Clopidogrel (Plavix )
 East Lancashire Medicines Management Board representing East Lancashire Hospitals NHS Trust, Lancashire Care Trust, Blackburn with Darwen PCT, East Lancs PCT Licensed Indications Guidelines for Use of
East Lancashire Medicines Management Board representing East Lancashire Hospitals NHS Trust, Lancashire Care Trust, Blackburn with Darwen PCT, East Lancs PCT Licensed Indications Guidelines for Use of
A. Ketorolac*** B. Naproxen C. Ibuprofen D. Celecoxib
 1. A man, 66 years of age, with a history of knee osteoarthritis (OA) is experiencing increasing pain at rest and with physical activity. He also has a history of depression and coronary artery disease.
1. A man, 66 years of age, with a history of knee osteoarthritis (OA) is experiencing increasing pain at rest and with physical activity. He also has a history of depression and coronary artery disease.
New Anticoagulants and GI bleeding
 New Anticoagulants and GI bleeding DR DANNY MYERS MD FRCP(C) CLINICAL ASSISTANT PROFESSOR OF MEDICINE, UBC Conflicts of Interest None I am unbiased in the use of NOAC s vs Warfarin based on risk benefit
New Anticoagulants and GI bleeding DR DANNY MYERS MD FRCP(C) CLINICAL ASSISTANT PROFESSOR OF MEDICINE, UBC Conflicts of Interest None I am unbiased in the use of NOAC s vs Warfarin based on risk benefit
Duration of Dual Antiplatelet Therapy After Coronary Stenting
 Duration of Dual Antiplatelet Therapy After Coronary Stenting C. DEAN KATSAMAKIS, DO, FACC, FSCAI INTERVENTIONAL CARDIOLOGIST ADVOCATE LUTHERAN GENERAL HOSPITAL INTRODUCTION Coronary artery stents are
Duration of Dual Antiplatelet Therapy After Coronary Stenting C. DEAN KATSAMAKIS, DO, FACC, FSCAI INTERVENTIONAL CARDIOLOGIST ADVOCATE LUTHERAN GENERAL HOSPITAL INTRODUCTION Coronary artery stents are
Apixaban Plus Mono vs. Dual Antiplatelet Therapy in Acute Coronary Syndromes: Insights from the APPRAISE-2 Trial
 Apixaban Plus Mono vs. Dual Antiplatelet Therapy in Acute Coronary Syndromes: Insights from the APPRAISE-2 Trial Connie N. Hess, MD, MHS, Stefan James, MD, PhD, Renato D. Lopes, MD, PhD, Daniel M. Wojdyla,
Apixaban Plus Mono vs. Dual Antiplatelet Therapy in Acute Coronary Syndromes: Insights from the APPRAISE-2 Trial Connie N. Hess, MD, MHS, Stefan James, MD, PhD, Renato D. Lopes, MD, PhD, Daniel M. Wojdyla,
Articles Presented. Journal Presentation. Dr Albert Lo. Dr Albert Lo
 * This presentation is prepared by the author in one s personal capacity for the purpose of academic exchange and does not represent the views of his/her organisations on the topic discussed. Journal Presentation
* This presentation is prepared by the author in one s personal capacity for the purpose of academic exchange and does not represent the views of his/her organisations on the topic discussed. Journal Presentation
Antiplatelet therapy:
 Balanced information for better care Antiplatelet therapy: Aggregating the latest evidence Evaluating the choices for a preventive therapy with impressive benefits and important risks Antiplatelet agents
Balanced information for better care Antiplatelet therapy: Aggregating the latest evidence Evaluating the choices for a preventive therapy with impressive benefits and important risks Antiplatelet agents
DUAL ANTIPLATELET THERAPY. Dr Robert S Mvungi, MD(Dar), Mmed (Wits) FCP(SA), Cert.Cardio(SA) Phy Tanzania Cardiac Society Dar es Salaam Tanzania
 DUAL ANTIPLATELET THERAPY Dr Robert S Mvungi, MD(Dar), Mmed (Wits) FCP(SA), Cert.Cardio(SA) Phy Tanzania Cardiac Society Dar es Salaam Tanzania DUAL ANTIPLATELET THERAPY (DAPT) Dual antiplatelet regimen
DUAL ANTIPLATELET THERAPY Dr Robert S Mvungi, MD(Dar), Mmed (Wits) FCP(SA), Cert.Cardio(SA) Phy Tanzania Cardiac Society Dar es Salaam Tanzania DUAL ANTIPLATELET THERAPY (DAPT) Dual antiplatelet regimen
Aspirin to Prevent Heart Attack and Stroke: What s the Right Dose?
 The American Journal of Medicine (2006) 119, 198-202 REVIEW Aspirin to Prevent Heart Attack and Stroke: What s the Right Dose? James E. Dalen, MD, MPH Professor Emeritus, University of Arizona, Tucson
The American Journal of Medicine (2006) 119, 198-202 REVIEW Aspirin to Prevent Heart Attack and Stroke: What s the Right Dose? James E. Dalen, MD, MPH Professor Emeritus, University of Arizona, Tucson
The largest clinical study of Bayer's Xarelto (rivaroxaban) Wednesday, 14 November 2012 07:38
 Bayer HealthCare has announced the initiation of the COMPASS study, the largest clinical study of its oral anticoagulant Xarelto (rivaroxaban) to date, investigating the prevention of major adverse cardiac
Bayer HealthCare has announced the initiation of the COMPASS study, the largest clinical study of its oral anticoagulant Xarelto (rivaroxaban) to date, investigating the prevention of major adverse cardiac
L'aspirina è diventata obsoleta nell'era dei nuovi inbitori P2Y12? Leonardo Bolognese MD, FESC, FACC Cardiovascular Department, Arezzo, Italy ISO 9001
 L'aspirina è diventata obsoleta nell'era dei nuovi inbitori P2Y12? Leonardo Bolognese MD, FESC, FACC Cardiovascular Department, Arezzo, Italy Scientific Advances and Cardiovascular Mortality Nabel and
L'aspirina è diventata obsoleta nell'era dei nuovi inbitori P2Y12? Leonardo Bolognese MD, FESC, FACC Cardiovascular Department, Arezzo, Italy Scientific Advances and Cardiovascular Mortality Nabel and
Journal Club: Niacin in Patients with Low HDL Cholesterol Levels Receiving Intensive Statin Therapy by the AIM-HIGH Investigators
 Journal Club: Niacin in Patients with Low HDL Cholesterol Levels Receiving Intensive Statin Therapy by the AIM-HIGH Investigators Shaikha Al Naimi Doctor of Pharmacy Student College of Pharmacy Qatar University
Journal Club: Niacin in Patients with Low HDL Cholesterol Levels Receiving Intensive Statin Therapy by the AIM-HIGH Investigators Shaikha Al Naimi Doctor of Pharmacy Student College of Pharmacy Qatar University
Perspectives on the Selection and Duration of Dual Antiplatelet Therapy
 Perspectives on the Selection and Duration of Dual Antiplatelet Therapy Dominick J. Angiolillo, MD, PhD, FACC, FESC, FSCAI Director of Cardiovascular Research Associate Professor of Medicine University
Perspectives on the Selection and Duration of Dual Antiplatelet Therapy Dominick J. Angiolillo, MD, PhD, FACC, FESC, FSCAI Director of Cardiovascular Research Associate Professor of Medicine University
Rivaroxaban for acute coronary syndromes
 Northern Treatment Advisory Group Rivaroxaban for acute coronary syndromes Lead author: Nancy Kane Regional Drug & Therapeutics Centre (Newcastle) May 2014 2014 Summary Current long-term management following
Northern Treatment Advisory Group Rivaroxaban for acute coronary syndromes Lead author: Nancy Kane Regional Drug & Therapeutics Centre (Newcastle) May 2014 2014 Summary Current long-term management following
FOR THE PREVENTION OF ATRIAL FIBRILLATION RELATED STROKE
 www.bpac.org.nz keyword: warfarinaspirin FOR THE PREVENTION OF ATRIAL FIBRILLATION RELATED STROKE Key Concepts In atrial fibrillation (AF) warfarin is more effective than aspirin for stroke prevention.
www.bpac.org.nz keyword: warfarinaspirin FOR THE PREVENTION OF ATRIAL FIBRILLATION RELATED STROKE Key Concepts In atrial fibrillation (AF) warfarin is more effective than aspirin for stroke prevention.
Dabigatran (Pradaxa) Guidelines
 Dabigatran (Pradaxa) Guidelines Dabigatran is a new anticoagulant for reducing the risk of stroke in patients with atrial fibrillation. Dabigatran is a direct thrombin inhibitor, similar to warfarin, without
Dabigatran (Pradaxa) Guidelines Dabigatran is a new anticoagulant for reducing the risk of stroke in patients with atrial fibrillation. Dabigatran is a direct thrombin inhibitor, similar to warfarin, without
Getting smart about dyspnea and life saving drug therapy in ACS patients. Kobi George Kaplan Medical Center Rehovot
 Getting smart about dyspnea and life saving drug therapy in ACS patients Kobi George Kaplan Medical Center Rehovot 78 year old female Case description Presented with resting chest pain and dyspnea Co morbidities:
Getting smart about dyspnea and life saving drug therapy in ACS patients Kobi George Kaplan Medical Center Rehovot 78 year old female Case description Presented with resting chest pain and dyspnea Co morbidities:
Dual Antiplatelet Therapy. Stephen Monroe, MD FACC Chattanooga Heart Institute
 Dual Antiplatelet Therapy Stephen Monroe, MD FACC Chattanooga Heart Institute Scope of Talk Identify the antiplatelet drugs and their mechanisms of action Review dual antiplatelet therapy in: The medical
Dual Antiplatelet Therapy Stephen Monroe, MD FACC Chattanooga Heart Institute Scope of Talk Identify the antiplatelet drugs and their mechanisms of action Review dual antiplatelet therapy in: The medical
Cilostazol versus Clopidogrel after Coronary Stenting
 Cilostazol versus Clopidogrel after Coronary Stenting Seong-Wook Park, MD, PhD, FACC Division of Cardiology, Asan Medical Center University of Ulsan College of Medicine Seoul, Korea AMC, 2004 Background
Cilostazol versus Clopidogrel after Coronary Stenting Seong-Wook Park, MD, PhD, FACC Division of Cardiology, Asan Medical Center University of Ulsan College of Medicine Seoul, Korea AMC, 2004 Background
ADVANCE: a factorial randomised trial of blood pressure lowering and intensive glucose control in 11,140 patients with type 2 diabetes
 ADVANCE: a factorial randomised trial of blood pressure lowering and intensive glucose control in 11,140 patients with type 2 diabetes Effects of a fixed combination of the ACE inhibitor, perindopril,
ADVANCE: a factorial randomised trial of blood pressure lowering and intensive glucose control in 11,140 patients with type 2 diabetes Effects of a fixed combination of the ACE inhibitor, perindopril,
Kevin Saunders MD CCFP Rivergrove Medical Clinic Wellness Institute @ SOGH April 17 2013
 Kevin Saunders MD CCFP Rivergrove Medical Clinic Wellness Institute @ SOGH April 17 2013 Family physician with Rivergrove Medical Clinic Practice in the north end since 1985 Medical Director of the Wellness
Kevin Saunders MD CCFP Rivergrove Medical Clinic Wellness Institute @ SOGH April 17 2013 Family physician with Rivergrove Medical Clinic Practice in the north end since 1985 Medical Director of the Wellness
Bayer Pharma AG 13342 Berlin Germany Tel. +49 30 468-1111 www.bayerpharma.com. News Release. Not intended for U.S. and UK Media
 News Release Not intended for U.S. and UK Media Bayer Pharma AG 13342 Berlin Germany Tel. +49 30 468-1111 www.bayerpharma.com Bayer Forms Collaboration with Academic and Governmental Institutions for Rivaroxaban
News Release Not intended for U.S. and UK Media Bayer Pharma AG 13342 Berlin Germany Tel. +49 30 468-1111 www.bayerpharma.com Bayer Forms Collaboration with Academic and Governmental Institutions for Rivaroxaban
Gruppo di lavoro: Malattie Tromboemboliche
 Gruppo di lavoro: Malattie Tromboemboliche 2381 Soluble Recombinant Thrombomodulin Ameliorates Hematological Malignancy-Induced Disseminated Intravascular Coagulation More Promptly Than Conventional Anticoagulant
Gruppo di lavoro: Malattie Tromboemboliche 2381 Soluble Recombinant Thrombomodulin Ameliorates Hematological Malignancy-Induced Disseminated Intravascular Coagulation More Promptly Than Conventional Anticoagulant
New Oral Anticoagulants
 New Oral Anticoagulants Tracy Minichiello, MD Associate Professor of Medicine Chief, San FranciscoVA Anticoagulation and Thrombosis Service Ansell, J. Hematology Copyright 2010 American Society of Hematology.
New Oral Anticoagulants Tracy Minichiello, MD Associate Professor of Medicine Chief, San FranciscoVA Anticoagulation and Thrombosis Service Ansell, J. Hematology Copyright 2010 American Society of Hematology.
Gloucestershire Hospitals
 TRUST GUIDELINE In the case of hard copies of this policy the content can only be assured to be accurate on the date of issue marked on the document. The Policy framework requires that the policy is fully
TRUST GUIDELINE In the case of hard copies of this policy the content can only be assured to be accurate on the date of issue marked on the document. The Policy framework requires that the policy is fully
What is an NNT? What is...? series Second edition Statistics. Supported by sanofi-aventis
 ...? series Second edition Statistics Supported by sanofi-aventis What is an NNT? Andrew Moore MA DPhil DSc CChem FRSC Senior Research Fellow, Pain Research and Nuffield Department of Anaesthetics, University
...? series Second edition Statistics Supported by sanofi-aventis What is an NNT? Andrew Moore MA DPhil DSc CChem FRSC Senior Research Fellow, Pain Research and Nuffield Department of Anaesthetics, University
The importance of adherence and persistence: The advantages of once-daily dosing
 The importance of adherence and persistence: The advantages of once-daily dosing Craig I. Coleman, PharmD Professor, University of Connecticut School of Pharmacy Storrs, CT, USA Conflicts of interest Dr
The importance of adherence and persistence: The advantages of once-daily dosing Craig I. Coleman, PharmD Professor, University of Connecticut School of Pharmacy Storrs, CT, USA Conflicts of interest Dr
Is There A LIfe for DES after discontinuation of Clopidogrel
 Chicago 2014 Is There A LIfe for DES after discontinuation of Clopidogrel Six-month versus 24-month dual antiplatelet therapy after implantation of drug eluting stents in patients non-resistant to aspirin:
Chicago 2014 Is There A LIfe for DES after discontinuation of Clopidogrel Six-month versus 24-month dual antiplatelet therapy after implantation of drug eluting stents in patients non-resistant to aspirin:
The author has no disclosures
 Mary Bradbury, PharmD, BCPS Clinical Pharmacy Specialist, Cardiac Surgery September 18, 2012 Mary.bradbury@inova.org This presentation will discuss unlabeled and investigational use of products The author
Mary Bradbury, PharmD, BCPS Clinical Pharmacy Specialist, Cardiac Surgery September 18, 2012 Mary.bradbury@inova.org This presentation will discuss unlabeled and investigational use of products The author
Rubén Casado Arroyo, 1,2 Mónica Polo-Tomas, 2 Maria P Roncalés, 2 James Scheiman, 3 Ángel Lanas 2,3,4 ORIGINAL ARTICLE. Antiplatelet therapy
 ORIGINAL ARTICLE Lower GI bleeding is more common than upper among patients on dual antiplatelet therapy: long-term follow-up of a cohort of patients commonly using PPI co-therapy Rubén Casado Arroyo,
ORIGINAL ARTICLE Lower GI bleeding is more common than upper among patients on dual antiplatelet therapy: long-term follow-up of a cohort of patients commonly using PPI co-therapy Rubén Casado Arroyo,
Committee Approval Date: September 12, 2014 Next Review Date: September 2015
 Medication Policy Manual Policy No: dru361 Topic: Pradaxa, dabigatran Date of Origin: September 12, 2014 Committee Approval Date: September 12, 2014 Next Review Date: September 2015 Effective Date: November
Medication Policy Manual Policy No: dru361 Topic: Pradaxa, dabigatran Date of Origin: September 12, 2014 Committee Approval Date: September 12, 2014 Next Review Date: September 2015 Effective Date: November
STROKE PREVENTION IN ATRIAL FIBRILLATION. TARGET AUDIENCE: All Canadian health care professionals. OBJECTIVE: ABBREVIATIONS: BACKGROUND:
 STROKE PREVENTION IN ATRIAL FIBRILLATION TARGET AUDIENCE: All Canadian health care professionals. OBJECTIVE: To guide clinicians in the selection of antithrombotic therapy for the secondary prevention
STROKE PREVENTION IN ATRIAL FIBRILLATION TARGET AUDIENCE: All Canadian health care professionals. OBJECTIVE: To guide clinicians in the selection of antithrombotic therapy for the secondary prevention
Series 1 Case Studies Adverse Events that Represent Unanticipated Problems: Reporting Required
 Welcome! This document contains three (3) series of Case Study examples that will demonstrate all four OHSU reporting categories (#1 4) as well as examples of events that are considered not reportable.
Welcome! This document contains three (3) series of Case Study examples that will demonstrate all four OHSU reporting categories (#1 4) as well as examples of events that are considered not reportable.
Antiplatelet and anticoagulation treatment of patients undergoing carotid and peripheral artery angioplasty
 Round Table: Antithrombotic therapy beyond ACS Antiplatelet and anticoagulation treatment of patients undergoing carotid and peripheral artery angioplasty M. Matsagkas, MD, PhD, EBSQ-Vasc Associate Professor
Round Table: Antithrombotic therapy beyond ACS Antiplatelet and anticoagulation treatment of patients undergoing carotid and peripheral artery angioplasty M. Matsagkas, MD, PhD, EBSQ-Vasc Associate Professor
Peptic Ulcer. Anatomy The stomach is a hollow organ. It is located in the upper abdomen, under the ribs.
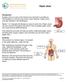 Peptic Ulcer Introduction A peptic ulcer is a sore in the lining of your stomach or duodenum. The duodenum is the first part of your small intestine. Peptic ulcers may also develop in the esophagus. Nearly
Peptic Ulcer Introduction A peptic ulcer is a sore in the lining of your stomach or duodenum. The duodenum is the first part of your small intestine. Peptic ulcers may also develop in the esophagus. Nearly
New Treatments for Stroke Prevention in Atrial Fibrillation. John C. Andrefsky, MD, FAHA NEOMED Internal Medicine Review course May 5 th, 2013
 New Treatments for Stroke Prevention in Atrial Fibrillation John C. Andrefsky, MD, FAHA NEOMED Internal Medicine Review course May 5 th, 2013 Classification Paroxysmal atrial fibrillation (AF) Last < 7
New Treatments for Stroke Prevention in Atrial Fibrillation John C. Andrefsky, MD, FAHA NEOMED Internal Medicine Review course May 5 th, 2013 Classification Paroxysmal atrial fibrillation (AF) Last < 7
Description of problem Description of proposed amendment Justification for amendment ERG response
 KEY INACCURACIES Issue 1 Distinguishing between groups of STEMI patients Key issue throughout the report The ERG distinguishes between groups of STEMI patients defining four patient groups: STEMI without
KEY INACCURACIES Issue 1 Distinguishing between groups of STEMI patients Key issue throughout the report The ERG distinguishes between groups of STEMI patients defining four patient groups: STEMI without
CDEC FINAL RECOMMENDATION
 CDEC FINAL RECOMMENDATION RIVAROXABAN (Xarelto Bayer Inc.) New Indication: Pulmonary Embolism Note: The Canadian Drug Expert Committee (CDEC) previously reviewed rivaroxaban for the treatment of deep vein
CDEC FINAL RECOMMENDATION RIVAROXABAN (Xarelto Bayer Inc.) New Indication: Pulmonary Embolism Note: The Canadian Drug Expert Committee (CDEC) previously reviewed rivaroxaban for the treatment of deep vein
understanding GI bleeding
 understanding GI bleeding a consumer education brochure American College of Gastroenterology 4900B South 31st Street, Arlington, VA 22206 703-820-7400 www.acg.gi.org American College of Gastroenterology
understanding GI bleeding a consumer education brochure American College of Gastroenterology 4900B South 31st Street, Arlington, VA 22206 703-820-7400 www.acg.gi.org American College of Gastroenterology
PRECOMBAT Trial. Seung-Whan Lee, MD, PhD On behalf of the PRECOMBAT Investigators
 Premier of Randomized Comparison of Bypass Surgery versus Angioplasty Using Sirolimus-Eluting Stent in Patients with Left Main Coronary Artery Disease PRECOMBAT Trial Seung-Whan Lee, MD, PhD On behalf
Premier of Randomized Comparison of Bypass Surgery versus Angioplasty Using Sirolimus-Eluting Stent in Patients with Left Main Coronary Artery Disease PRECOMBAT Trial Seung-Whan Lee, MD, PhD On behalf
Antiplatelet and Antithrombotics From clinical trials to guidelines
 Antiplatelet and Antithrombotics From clinical trials to guidelines Ashraf Reda, MD, FESC Prof and head of Cardiology Dep. Menofiya University Preisedent of EGYBAC Chairman of WGLVR One of the big stories
Antiplatelet and Antithrombotics From clinical trials to guidelines Ashraf Reda, MD, FESC Prof and head of Cardiology Dep. Menofiya University Preisedent of EGYBAC Chairman of WGLVR One of the big stories
4/9/2015. Risk Stratify Our Patients. Stroke Risk in AF: CHADS2 Scoring system JAMA 2001; 285: 2864-71
 Anticoagulation in the 21 st Century Adam Karpman, D.O. Saint Francis Medical Center/Oklahoma State University Medical Center Disclosures: None Atrial Fibrillation Most common arrhythmia in clinical practice.
Anticoagulation in the 21 st Century Adam Karpman, D.O. Saint Francis Medical Center/Oklahoma State University Medical Center Disclosures: None Atrial Fibrillation Most common arrhythmia in clinical practice.
Guidance for Industry Diabetes Mellitus Evaluating Cardiovascular Risk in New Antidiabetic Therapies to Treat Type 2 Diabetes
 Guidance for Industry Diabetes Mellitus Evaluating Cardiovascular Risk in New Antidiabetic Therapies to Treat Type 2 Diabetes U.S. Department of Health and Human Services Food and Drug Administration Center
Guidance for Industry Diabetes Mellitus Evaluating Cardiovascular Risk in New Antidiabetic Therapies to Treat Type 2 Diabetes U.S. Department of Health and Human Services Food and Drug Administration Center
Atrial Fibrillation, Chronic - Antithrombotic Treatment - OBSOLETE
 Atrial Fibrillation, Chronic - Antithrombotic Treatment - OBSOLETE Clinical practice guidelines serve as an educational reference, and do not supersede the clinical judgment of the treating physician with
Atrial Fibrillation, Chronic - Antithrombotic Treatment - OBSOLETE Clinical practice guidelines serve as an educational reference, and do not supersede the clinical judgment of the treating physician with
THE INTERNET STROKE CENTER PRESENTATIONS AND DISCUSSIONS ON STROKE MANAGEMENT
 THE INTERNET STROKE CENTER PRESENTATIONS AND DISCUSSIONS ON STROKE MANAGEMENT Stroke Prevention in Atrial Fibrillation Gregory Albers, M.D. Director Stanford Stroke Center Professor of Neurology and Neurological
THE INTERNET STROKE CENTER PRESENTATIONS AND DISCUSSIONS ON STROKE MANAGEMENT Stroke Prevention in Atrial Fibrillation Gregory Albers, M.D. Director Stanford Stroke Center Professor of Neurology and Neurological
HERTFORDSHIRE MEDICINES MANAGEMENT COMMITTEE (HMMC) RIVAROXABAN RECOMMENDED see specific recommendations for licensed indications below
 Name: generic (trade) Rivaroxaban (Xarelto ) HERTFORDSHIRE MEDICINES MANAGEMENT COMMITTEE (HMMC) RIVAROXABAN RECOMMENDED see specific recommendations for licensed indications below What it is Indications
Name: generic (trade) Rivaroxaban (Xarelto ) HERTFORDSHIRE MEDICINES MANAGEMENT COMMITTEE (HMMC) RIVAROXABAN RECOMMENDED see specific recommendations for licensed indications below What it is Indications
AHA/ASA Scientific Statement Oral Antithrombotic Agents for the Prevention of Stroke in Atrial Fibrillation
 AHA/ASA Scientific Statement Oral Antithrombotic Agents for the Prevention of Stroke in Atrial Fibrillation A Statement for Healthcare Professionals from the American Heart Association/American Stroke
AHA/ASA Scientific Statement Oral Antithrombotic Agents for the Prevention of Stroke in Atrial Fibrillation A Statement for Healthcare Professionals from the American Heart Association/American Stroke
Management of the new antiplatelets and anticoagulants
 Management of the new antiplatelets and anticoagulants Session No.: 1 Name: C. Boustiere, T Ponchon Guidelines : Anti-thrombotic agents and digestive endoscopy 2006 : French guideline (SFED) 2007 : Japanese
Management of the new antiplatelets and anticoagulants Session No.: 1 Name: C. Boustiere, T Ponchon Guidelines : Anti-thrombotic agents and digestive endoscopy 2006 : French guideline (SFED) 2007 : Japanese
Platelet Function Testing vs Genotyping : Focus on Pharmacogenomics of Clopidogrel. Kiyuk Chang, M.D., Ph.D.
 Platelet Function Testing vs Genotyping : Focus on Pharmacogenomics of Clopidogrel Kiyuk Chang, M.D., Ph.D. Cardiovascular Center & Cardiology Division Seoul St. Mary s Hospital The Catholic University
Platelet Function Testing vs Genotyping : Focus on Pharmacogenomics of Clopidogrel Kiyuk Chang, M.D., Ph.D. Cardiovascular Center & Cardiology Division Seoul St. Mary s Hospital The Catholic University
Rebleeding and survival after acute lower gastrointestinal bleeding
 The American Journal of Surgery 188 (2004) 485 490 Scientific paper Rebleeding and survival after acute lower gastrointestinal bleeding Thomas Anthony, M.D.*, Pradeep Penta, B.S., Robert D. Todd, B.S.,
The American Journal of Surgery 188 (2004) 485 490 Scientific paper Rebleeding and survival after acute lower gastrointestinal bleeding Thomas Anthony, M.D.*, Pradeep Penta, B.S., Robert D. Todd, B.S.,
Sponsor. Novartis Generic Drug Name. Vildagliptin. Therapeutic Area of Trial. Type 2 diabetes. Approved Indication. Investigational.
 Clinical Trial Results Database Page 1 Sponsor Novartis Generic Drug Name Vildagliptin Therapeutic Area of Trial Type 2 diabetes Approved Indication Investigational Study Number CLAF237A2386 Title A single-center,
Clinical Trial Results Database Page 1 Sponsor Novartis Generic Drug Name Vildagliptin Therapeutic Area of Trial Type 2 diabetes Approved Indication Investigational Study Number CLAF237A2386 Title A single-center,
Bayer Initiates Rivaroxaban Phase III Study to Support Dose Selection According to Individual Benefit-Risk Profile in Long- Term VTE Prevention
 Investor News Not intended for U.S. and UK Media Bayer AG Investor Relations 51368 Leverkusen Germany www.investor.bayer.com Long-term prevention of venous blood clots (VTE): Bayer Initiates Rivaroxaban
Investor News Not intended for U.S. and UK Media Bayer AG Investor Relations 51368 Leverkusen Germany www.investor.bayer.com Long-term prevention of venous blood clots (VTE): Bayer Initiates Rivaroxaban
WOEST TRIAL- NO ASPIRIN IN STENTED PATIENTS REQUIRING ANTICOAGULATION. Van Crisco, MD, FACC, FSCAI First Coast
 WOEST TRIAL- NO ASPIRIN IN STENTED PATIENTS REQUIRING ANTICOAGULATION Van Crisco, MD, FACC, FSCAI First Coast Conflicts of Interest I have been a paid consultant and speaker for AstraZeneca, makers of
WOEST TRIAL- NO ASPIRIN IN STENTED PATIENTS REQUIRING ANTICOAGULATION Van Crisco, MD, FACC, FSCAI First Coast Conflicts of Interest I have been a paid consultant and speaker for AstraZeneca, makers of
Inpatient Anticoagulation Safety. To provide safe and effective anticoagulation therapy through a collaborative approach.
 Inpatient Anticoagulation Safety Purpose: Policy: To provide safe and effective anticoagulation therapy through a collaborative approach. Upon the written order of a physician, Heparin, Low Molecular Weight
Inpatient Anticoagulation Safety Purpose: Policy: To provide safe and effective anticoagulation therapy through a collaborative approach. Upon the written order of a physician, Heparin, Low Molecular Weight
New Anticoagulation Options for Stroke Prevention in Atrial Fibrillation. Joy Wahawisan, Pharm.D., BCPS April 25, 2012
 New Anticoagulation Options for Stroke Prevention in Atrial Fibrillation Joy Wahawisan, Pharm.D., BCPS April 25, 2012 Stroke in Atrial Fibrillation % Stroke 1991;22:983. Age Range (years) CHADS 2 Risk
New Anticoagulation Options for Stroke Prevention in Atrial Fibrillation Joy Wahawisan, Pharm.D., BCPS April 25, 2012 Stroke in Atrial Fibrillation % Stroke 1991;22:983. Age Range (years) CHADS 2 Risk
Novel Anticoagulation Agents DISCLOSURES. Objectives ATRIAL FIBRILLATION TRIALS. NOAC Comparison 6/12/2015
 Novel Anticoagulation Agents DISCLOSURES James W. Haynes, MD Department of Family Medicine Univ of TN Health Science Center (Chattanooga) Objectives Understand mechanism of action behind the NOAC agents
Novel Anticoagulation Agents DISCLOSURES James W. Haynes, MD Department of Family Medicine Univ of TN Health Science Center (Chattanooga) Objectives Understand mechanism of action behind the NOAC agents
A Patient s Guide to Primary and Secondary Prevention of Cardiovascular Disease Using Blood-Thinning (Anticoagulant) Drugs
 A Patient s Guide to Primary and Secondary Prevention of PATIENT EDUCATION GUIDE What Is Cardiovascular Disease? Cardiovascular disease (CVD) is a broad term that covers any disease of the heart and circulatory
A Patient s Guide to Primary and Secondary Prevention of PATIENT EDUCATION GUIDE What Is Cardiovascular Disease? Cardiovascular disease (CVD) is a broad term that covers any disease of the heart and circulatory
ABOUT XARELTO CLINICAL STUDIES
 ABOUT XARELTO CLINICAL STUDIES FAST FACTS Xarelto (rivaroxaban) is a novel, oral direct Factor Xa inhibitor. On September 30, 2008, the European Commission granted marketing approval for Xarelto for the
ABOUT XARELTO CLINICAL STUDIES FAST FACTS Xarelto (rivaroxaban) is a novel, oral direct Factor Xa inhibitor. On September 30, 2008, the European Commission granted marketing approval for Xarelto for the
A rapid-release 50-mg tablet-based 13 C-urea breath test for the diagnosis of Helicobacter pylori infection
 Aliment Pharmacol Ther 2003; 17: 253 257. doi: 10.1046/j.0269-2813.2003.01417.x A rapid-release 50-mg tablet-based 13 C-urea breath test for the diagnosis of Helicobacter pylori infection W.M.WONG*,S.K.LAM*,K.C.LAI*,K.M.CHU,
Aliment Pharmacol Ther 2003; 17: 253 257. doi: 10.1046/j.0269-2813.2003.01417.x A rapid-release 50-mg tablet-based 13 C-urea breath test for the diagnosis of Helicobacter pylori infection W.M.WONG*,S.K.LAM*,K.C.LAI*,K.M.CHU,
Bios 6648: Design & conduct of clinical research
 Bios 6648: Design & conduct of clinical research Section 1 - Specifying the study setting and objectives 1. Specifying the study setting and objectives 1.0 Background Where will we end up?: (a) The treatment
Bios 6648: Design & conduct of clinical research Section 1 - Specifying the study setting and objectives 1. Specifying the study setting and objectives 1.0 Background Where will we end up?: (a) The treatment
ESCMID Online Lecture Library. by author
 Do statins improve outcomes of patients with sepsis and pneumonia? Jordi Carratalà Department of Infectious Diseases Statins for sepsis & community-acquired pneumonia Sepsis and CAP are major healthcare
Do statins improve outcomes of patients with sepsis and pneumonia? Jordi Carratalà Department of Infectious Diseases Statins for sepsis & community-acquired pneumonia Sepsis and CAP are major healthcare
The University of Hong Kong Department of Surgery Division of Esophageal and Upper Gastrointestinal Surgery
 Program Overview The University of Hong Kong Department of Surgery Division of Esophageal and Upper Gastrointestinal Surgery Weight Control and Metabolic Surgery Program The Weight Control and Metabolic
Program Overview The University of Hong Kong Department of Surgery Division of Esophageal and Upper Gastrointestinal Surgery Weight Control and Metabolic Surgery Program The Weight Control and Metabolic
An Introduction to the Improved FDA Prescription Drug Labeling
 An Introduction to the Improved FDA Prescription Drug Labeling 1 Introduction Mary E. Kremzner, Pharm.D. CDR, U.S. Public Health Service Deputy Director, Division of Drug Information Center for Drug Evaluation
An Introduction to the Improved FDA Prescription Drug Labeling 1 Introduction Mary E. Kremzner, Pharm.D. CDR, U.S. Public Health Service Deputy Director, Division of Drug Information Center for Drug Evaluation
ANESTHESIA FOR PATIENTS WITH CORONARY STENTS FOR NON CARDIAC SURGERY. Dr. Mahesh Vakamudi. Professor and Head
 ANESTHESIA FOR PATIENTS WITH CORONARY STENTS FOR NON CARDIAC SURGERY Dr. Mahesh Vakamudi Professor and Head Department of Anesthesiology, Critical Care and Pain Medicine Sri Ramachandra University INTRODUCTION
ANESTHESIA FOR PATIENTS WITH CORONARY STENTS FOR NON CARDIAC SURGERY Dr. Mahesh Vakamudi Professor and Head Department of Anesthesiology, Critical Care and Pain Medicine Sri Ramachandra University INTRODUCTION
Improving Appropriate Use of Proton Pump Inhibitors as Gastrointestinal Prophylaxis in the Hospital Setting
 Improving Appropriate Use of Proton Pump Inhibitors as Gastrointestinal Prophylaxis in the Hospital Setting DATE Educating for Quality Improvement & Patient Safety 1 The Team Division CS&E Participants
Improving Appropriate Use of Proton Pump Inhibitors as Gastrointestinal Prophylaxis in the Hospital Setting DATE Educating for Quality Improvement & Patient Safety 1 The Team Division CS&E Participants
VIOXX Gastrointestinal Outcomes Research Trial (VIGOR) Bonnie Goldmann, M.D. Regulatory Affairs Merck Research Laboratories
 VIOXX Gastrointestinal Outcomes Research Trial (VIGOR) Bonnie Goldmann, M.D. Regulatory Affairs Merck Research Laboratories 1 Arachidonic Acid CO 2 H COX-1 NSAIDs COX-2 Prostanoids Prostanoids Protection
VIOXX Gastrointestinal Outcomes Research Trial (VIGOR) Bonnie Goldmann, M.D. Regulatory Affairs Merck Research Laboratories 1 Arachidonic Acid CO 2 H COX-1 NSAIDs COX-2 Prostanoids Prostanoids Protection
Dabigatran (Pradaxa) for stroke prevention in patients with non-valvular atrial fibrillation (da-big-a-tran)
 Dabigatran (Pradaxa) 1 Dabigatran (Pradaxa) for stroke prevention in patients with non-valvular atrial fibrillation (da-big-a-tran) Similar rate of major bleeding to warfarin KEY POINTS Dabigatran is an
Dabigatran (Pradaxa) 1 Dabigatran (Pradaxa) for stroke prevention in patients with non-valvular atrial fibrillation (da-big-a-tran) Similar rate of major bleeding to warfarin KEY POINTS Dabigatran is an
Novel oral anticoagulant (NOAC) for stroke prevention in atrial fibrillation Special situations
 Novel oral anticoagulant (NOAC) for stroke prevention in atrial fibrillation Special situations Dardo E. Ferrara MD Cardiac Electrophysiology North Cascade Cardiology PeaceHealth Medical Group Which anticoagulant
Novel oral anticoagulant (NOAC) for stroke prevention in atrial fibrillation Special situations Dardo E. Ferrara MD Cardiac Electrophysiology North Cascade Cardiology PeaceHealth Medical Group Which anticoagulant
Cardiovascular Subcommittee of PTAC Meeting held 27 February 2014. (minutes for web publishing)
 Cardiovascular Subcommittee of PTAC Meeting held 27 February 2014 (minutes for web publishing) Cardiovascular Subcommittee minutes are published in accordance with the Terms of Reference for the Pharmacology
Cardiovascular Subcommittee of PTAC Meeting held 27 February 2014 (minutes for web publishing) Cardiovascular Subcommittee minutes are published in accordance with the Terms of Reference for the Pharmacology
Antiaggreganti. STEMI : cosa c è di nuovo? Heartline 2015. Genova 13 14 Novembre 2015
 Heartline 2015 Genova 13 14 Novembre 2015 STEMI : cosa c è di nuovo? Antiaggreganti Luigi Oltrona Visconti Divisione di Cardiologia IRCCS Fondazione Policlinico S. Matteo Pavia STEMI : cosa c è di nuovo?
Heartline 2015 Genova 13 14 Novembre 2015 STEMI : cosa c è di nuovo? Antiaggreganti Luigi Oltrona Visconti Divisione di Cardiologia IRCCS Fondazione Policlinico S. Matteo Pavia STEMI : cosa c è di nuovo?
prodigy study: duration of dual antiplatelet therapy under review In BrIef In Brief NPS RADAR AuguSt 2012
 3 In BrIef A digest of news items about NPS RADAR, new drugs and changes to PBS listings prodigy study: duration of dual antiplatelet therapy under review Dual antiplatelet therapy with aspirin and clopidogrel
3 In BrIef A digest of news items about NPS RADAR, new drugs and changes to PBS listings prodigy study: duration of dual antiplatelet therapy under review Dual antiplatelet therapy with aspirin and clopidogrel
Dabigatran etexilate for the treatment and secondary prevention of deep vein thrombosis and/or pulmonary embolism ERRATUM
 Dabigatran etexilate for the treatment and secondary prevention of deep vein thrombosis and/or pulmonary embolism ERRATUM This report was commissioned by the NIHR HTA Programme as project number 12/78
Dabigatran etexilate for the treatment and secondary prevention of deep vein thrombosis and/or pulmonary embolism ERRATUM This report was commissioned by the NIHR HTA Programme as project number 12/78
National Digestive Diseases Information Clearinghouse
 Gastritis National Digestive Diseases Information Clearinghouse U.S. Department of Health and Human Services NATIONAL INSTITUTES OF HEALTH What is gastritis? Gastritis is a condition in which the stomach
Gastritis National Digestive Diseases Information Clearinghouse U.S. Department of Health and Human Services NATIONAL INSTITUTES OF HEALTH What is gastritis? Gastritis is a condition in which the stomach
Systolic Blood Pressure Intervention Trial (SPRINT) Principal Results
 Systolic Blood Pressure Intervention Trial (SPRINT) Principal Results Paul K. Whelton, MB, MD, MSc Chair, SPRINT Steering Committee Tulane University School of Public Health and Tropical Medicine, and
Systolic Blood Pressure Intervention Trial (SPRINT) Principal Results Paul K. Whelton, MB, MD, MSc Chair, SPRINT Steering Committee Tulane University School of Public Health and Tropical Medicine, and
University of Ulsan College of Medicine, Asan Medical Center on behalf of the REAL-LATE and the ZEST-LATE trial
 Duration of Dual Antiplatelet Therapy After Drug-Eluting Stent Implantation A Pooled Analysis of the REAL-LATE and the ZEST-LATE Trial Seung-Jung Park MD PhD Seung-Jung Park, MD, PhD, University of Ulsan
Duration of Dual Antiplatelet Therapy After Drug-Eluting Stent Implantation A Pooled Analysis of the REAL-LATE and the ZEST-LATE Trial Seung-Jung Park MD PhD Seung-Jung Park, MD, PhD, University of Ulsan
GI Bleed. Steven Lichtenstein, D.O. Chief, Division of Gastroenterology Mercy Health System. Director, Endoscopy/GI Lab Mercy Fitzgerald Hospital
 October 3, 2015 GI Bleed Steven Lichtenstein, D.O. Chief, Division of Gastroenterology Mercy Health System Director, Endoscopy/GI Lab Mercy Fitzgerald Hospital Clinical Associate Professor of Medicine
October 3, 2015 GI Bleed Steven Lichtenstein, D.O. Chief, Division of Gastroenterology Mercy Health System Director, Endoscopy/GI Lab Mercy Fitzgerald Hospital Clinical Associate Professor of Medicine
Endoscopy & ACS. 8/1/2014 Dr. Whang
 Endoscopy & ACS 8/1/2014 Dr. Whang Outline I. Antiplatelets II. Coronary Artery Disease in U.S. III. GI Bleeding in ACS IV. ACC/AHA guidelines: Dual Antiplatelet Therapy in CAD V. PPI and Plavix Controversy
Endoscopy & ACS 8/1/2014 Dr. Whang Outline I. Antiplatelets II. Coronary Artery Disease in U.S. III. GI Bleeding in ACS IV. ACC/AHA guidelines: Dual Antiplatelet Therapy in CAD V. PPI and Plavix Controversy
This clinical study synopsis is provided in line with Boehringer Ingelheim s Policy on Transparency and Publication of Clinical Study Data.
 abcd Clinical Study for Public Disclosure This clinical study synopsis is provided in line with s Policy on Transparency and Publication of Clinical Study Data. The synopsis which is part of the clinical
abcd Clinical Study for Public Disclosure This clinical study synopsis is provided in line with s Policy on Transparency and Publication of Clinical Study Data. The synopsis which is part of the clinical
Medical management of CHF: A New Class of Medication. Al Timothy, M.D. Cardiovascular Institute of the South
 Medical management of CHF: A New Class of Medication Al Timothy, M.D. Cardiovascular Institute of the South Disclosures Speakers Bureau for Amgen Background Chronic systolic congestive heart failure remains
Medical management of CHF: A New Class of Medication Al Timothy, M.D. Cardiovascular Institute of the South Disclosures Speakers Bureau for Amgen Background Chronic systolic congestive heart failure remains
Cardiovascular Disease
 Cardiovascular Disease 1 Cardiovascular Disease 1. More target specific oral anticoagulants (TSOAC) 2. Vorapaxar (Zonivity) 3. Continued noise about a polypill 4. WATCHMAN 3 1 2 3 4 Left Atrial Appendage
Cardiovascular Disease 1 Cardiovascular Disease 1. More target specific oral anticoagulants (TSOAC) 2. Vorapaxar (Zonivity) 3. Continued noise about a polypill 4. WATCHMAN 3 1 2 3 4 Left Atrial Appendage
FDA Approved Oral Anticoagulants
 FDA Approved Oral Anticoagulants Generic (Trade Name) Warfarin (Coumadin, Jantoven ) 1 FDA approved indication Prophylaxis and treatment of venous thromboembolism (VTE) Prophylaxis and treatment of thromboembolic
FDA Approved Oral Anticoagulants Generic (Trade Name) Warfarin (Coumadin, Jantoven ) 1 FDA approved indication Prophylaxis and treatment of venous thromboembolism (VTE) Prophylaxis and treatment of thromboembolic
RR 0.88 (95% CI: 0.77 1.00) P=0.051 (superiority) 3.75
 ALL-CAUSE MORTALITY RR 0.88 (95% CI: 0.77 1.00) P=0.051 (superiority) Rate per year (%) 5.0 4.0 3.0 2.0 1.0 0 3.64 D150 mg BID 3.75 D110 mg BID RR 0.91 (95% CI: 0.80 1.03) P=0.13 (superiority) 4.13 Warfarin
ALL-CAUSE MORTALITY RR 0.88 (95% CI: 0.77 1.00) P=0.051 (superiority) Rate per year (%) 5.0 4.0 3.0 2.0 1.0 0 3.64 D150 mg BID 3.75 D110 mg BID RR 0.91 (95% CI: 0.80 1.03) P=0.13 (superiority) 4.13 Warfarin
Figure 2: Recurrent chest pain of suspected esophageal origin
 Figure 2: Recurrent chest pain of suspected esophageal origin 1 patient with chest pain of suspected esophageal origin 2 history and physical exam. suggestive of n-esophageal etiology? 3 evaluate and treat
Figure 2: Recurrent chest pain of suspected esophageal origin 1 patient with chest pain of suspected esophageal origin 2 history and physical exam. suggestive of n-esophageal etiology? 3 evaluate and treat
Breadth of indications matters One drug for multiple indications
 Breadth of indications matters One drug for multiple indications Sylvia Haas, MD, PhD Formerly of the Technical University of Munich Munich, Germany Disclosures: Sylvia Haas 1 Novel oral anticoagulants:
Breadth of indications matters One drug for multiple indications Sylvia Haas, MD, PhD Formerly of the Technical University of Munich Munich, Germany Disclosures: Sylvia Haas 1 Novel oral anticoagulants:
Thrombosis and Hemostasis
 Thrombosis and Hemostasis Wendy Lim, MD, MSc, FRCPC Associate Professor, Department of Medicine McMaster University, Hamilton, ON Overview To review the important developments in venous thromboembolism
Thrombosis and Hemostasis Wendy Lim, MD, MSc, FRCPC Associate Professor, Department of Medicine McMaster University, Hamilton, ON Overview To review the important developments in venous thromboembolism
Bayer Extends Clinical Investigation of Rivaroxaban into Important Areas of Unmet Medical Need in Arterial Thromboembolism
 Investor News Not intended for U.S. and UK Media Bayer AG Investor Relations 51368 Leverkusen Germany www.investor.bayer.com Bayer Extends Clinical Investigation of Rivaroxaban into Important Areas of
Investor News Not intended for U.S. and UK Media Bayer AG Investor Relations 51368 Leverkusen Germany www.investor.bayer.com Bayer Extends Clinical Investigation of Rivaroxaban into Important Areas of
Gastrointestinal bleeding: the management of acute upper gastrointestinal bleeding
 Gastrointestinal bleeding: the management of acute upper gastrointestinal bleeding NICE guideline Draft for consultation, December 2011 If you wish to comment on this version of the guideline, please be
Gastrointestinal bleeding: the management of acute upper gastrointestinal bleeding NICE guideline Draft for consultation, December 2011 If you wish to comment on this version of the guideline, please be
Drug treatments in upper gastrointestinal bleeding: value of endoscopic findings as surrogate end points
 372 Gut 2001;49:372 379 Drug treatments in upper gastrointestinal bleeding: value of endoscopic findings as surrogate end points G M Hawkey, A T Cole, A S McIntyre, R G Long, C J Hawkey Division of Gastroenterology,
372 Gut 2001;49:372 379 Drug treatments in upper gastrointestinal bleeding: value of endoscopic findings as surrogate end points G M Hawkey, A T Cole, A S McIntyre, R G Long, C J Hawkey Division of Gastroenterology,
Secondary Stroke Prevention Luke Bradbury, MD 10/4/14 Fall WAPA Conferfence
 Guidelines Secondary Stroke Prevention Luke Bradbury, MD 10/4/14 Fall WAPA Conferfence Stroke/TIA Nearly 700,000 ischemic strokes and 240,000 TIAs every year in the United States Currently, the risk for
Guidelines Secondary Stroke Prevention Luke Bradbury, MD 10/4/14 Fall WAPA Conferfence Stroke/TIA Nearly 700,000 ischemic strokes and 240,000 TIAs every year in the United States Currently, the risk for
Mohammad Reza Motamed MD 29, Seyed-Mohammad Fereshtehnejad 30, Nooshin Sadigh 2
 Dual antiplatelet therapy with clopidogrel and aspirin for stroke prevention following transient ischemic attack (TIA) or mild-to-moderate ischemic stroke Mohammad Reza Motamed MD 29, Seyed-Mohammad Fereshtehnejad
Dual antiplatelet therapy with clopidogrel and aspirin for stroke prevention following transient ischemic attack (TIA) or mild-to-moderate ischemic stroke Mohammad Reza Motamed MD 29, Seyed-Mohammad Fereshtehnejad
Anticoagulation in Atrial Fibrillation
 Anticoagulation in Atrial Fibrillation Parag P. Patel, MD FACC Disclosures Eliquis Speakers Bureau 1 Clinical Scenario Ms. L is a 76F admitted to the stroke service with a dense right sided hemiparesis
Anticoagulation in Atrial Fibrillation Parag P. Patel, MD FACC Disclosures Eliquis Speakers Bureau 1 Clinical Scenario Ms. L is a 76F admitted to the stroke service with a dense right sided hemiparesis
Clinical Study Synopsis
 Clinical Study Synopsis This Clinical Study Synopsis is provided for patients and healthcare professionals to increase the transparency of Bayer's clinical research. This document is not intended to replace
Clinical Study Synopsis This Clinical Study Synopsis is provided for patients and healthcare professionals to increase the transparency of Bayer's clinical research. This document is not intended to replace
Clinical Study Synopsis
 Clinical Study Synopsis This Clinical Study Synopsis is provided for patients and healthcare professionals to increase the transparency of Bayer's clinical research. This document is not intended to replace
Clinical Study Synopsis This Clinical Study Synopsis is provided for patients and healthcare professionals to increase the transparency of Bayer's clinical research. This document is not intended to replace
Treating AF: The Newest Recommendations. CardioCase presentation. Ethel s Case. Wayne Warnica, MD, FACC, FACP, FRCPC
 Treating AF: The Newest Recommendations Wayne Warnica, MD, FACC, FACP, FRCPC CardioCase presentation Ethel s Case Ethel, 73, presents with rapid heart beating and mild chest discomfort. In the ED, ECG
Treating AF: The Newest Recommendations Wayne Warnica, MD, FACC, FACP, FRCPC CardioCase presentation Ethel s Case Ethel, 73, presents with rapid heart beating and mild chest discomfort. In the ED, ECG
1/7/2012. Objectives. Epidemiology of Atrial Fibrillation(AF) Stroke in AF. Stroke Risk Stratification in AF
 Objectives Atrial Fibrillation and Prevention of Thrombotic Complications: Therapeutic Update Andrea C. Flores Pharm.D Pharmacy Resident at the Miami VA Healthcare System Review the epidemiology, pathophysiology
Objectives Atrial Fibrillation and Prevention of Thrombotic Complications: Therapeutic Update Andrea C. Flores Pharm.D Pharmacy Resident at the Miami VA Healthcare System Review the epidemiology, pathophysiology
Investor News. Not intended for U.S. and UK media
 Investor News Not intended for U.S. and UK media Bayer AG Investor Relations 51368 Leverkusen Germany www.investor.bayer.com Bayer s Xarelto (Rivaroxaban) Approved for the Treatment of Pulmonary Embolism
Investor News Not intended for U.S. and UK media Bayer AG Investor Relations 51368 Leverkusen Germany www.investor.bayer.com Bayer s Xarelto (Rivaroxaban) Approved for the Treatment of Pulmonary Embolism
DVT/PE Management with Rivaroxaban (Xarelto)
 DVT/PE Management with Rivaroxaban (Xarelto) Rivaroxaban is FDA approved for the acute treatment of DVT and PE and reduction in risk of recurrence of DVT and PE. FDA approved indications: Non valvular
DVT/PE Management with Rivaroxaban (Xarelto) Rivaroxaban is FDA approved for the acute treatment of DVT and PE and reduction in risk of recurrence of DVT and PE. FDA approved indications: Non valvular
GI Bleeding. Thomas S.Foster,Pharm.D. PHR 961 Integrated Therapeutics
 GI Bleeding Thomas S.Foster,Pharm.D. PHR 961 Integrated Therapeutics Overview Because GI bleeding is internal, it is possible for a person to have GI bleeding without symptoms. Important to recognize
GI Bleeding Thomas S.Foster,Pharm.D. PHR 961 Integrated Therapeutics Overview Because GI bleeding is internal, it is possible for a person to have GI bleeding without symptoms. Important to recognize
What s the Deal with NSAIDs?
 Focus on CME at the Memorial xxx University of Newfoundland What s the Deal with NSAIDs? Majed M. Khraishi, MB, BCh, FRCPC Presented at Wednesday at Noon Ask the Consultant Arthritis is one of the most
Focus on CME at the Memorial xxx University of Newfoundland What s the Deal with NSAIDs? Majed M. Khraishi, MB, BCh, FRCPC Presented at Wednesday at Noon Ask the Consultant Arthritis is one of the most
ECG may be indicated for patients with cardiovascular risk factors
 eappendix A. Summary for Preoperative ECG American College of Cardiology/ American Heart Association, 2007 A1 2002 A2 European Society of Cardiology and European Society of Anaesthesiology, 2009 A3 Improvement,
eappendix A. Summary for Preoperative ECG American College of Cardiology/ American Heart Association, 2007 A1 2002 A2 European Society of Cardiology and European Society of Anaesthesiology, 2009 A3 Improvement,
