Clinical Policy Title: Vagus Nerve Stimulation (VNS)
|
|
|
- Conrad Preston
- 9 years ago
- Views:
Transcription
1 Clinical Policy Title: Vagus Nerve Stimulation (VNS) Clinical Policy Number: Effective Date: Sept. 1, 2013 Initial Review Date: May 15, 2013 Most Recent Review Date: May 8, 2014 Next Review Date: May 2015 Policy contains: Generalized epilepsy with atonic or tonic-clonic seizures. Medically refractory seizures. Vagus nerve. ABOUT THIS POLICY: Keystone First has developed clinical policies to assist with making coverage determinations. Keystone First clinical policies are based on guidelines from established industry sources, such as the Centers for Medicare & Medicaid Services (CMS), state regulatory agencies, the American Medical Association (AMA), medical specialty professional societies, and peer-reviewed professional literature. These clinical policies, along with other sources, such as plan benefits and state and federal laws and regulatory requirements, including any state- or plan-specific definition of medically necessary, and the specific facts of the particular situation are considered by Keystone First when making coverage determinations. In the event of conflict between this clinical policy and plan benefits and/or state or federal laws and/or regulatory requirements, the plan benefits and/or state and federal laws and/or regulatory requirements shall control. Keystone First clinical policies are for informational purposes only and not intended as medical advice or to direct treatment. Physicians and other health care providers are solely responsible for the treatment decisions for their patients. Keystone First clinical policies are reflective of evidence-based medicine at the time of review. As medical science evolves, Keystone First will update its clinical policies as necessary. Keystone First clinical policies are not guarantees of payment. Coverage policy Keystone First considers the use of vagus nerve stimulation (VNS) to be clinically proven and therefore, medically necessary when the following criteria are met: PATIENT CRITERIA ALL criteria must be met The individual is age 12 years or older. The individual is diagnosed with severe generalized epilepsy with atonic or tonicclonic seizures. The individual experiences partial onset seizures that are refractory to antiepileptic (AEDs) medication therapy, including individuals who are allergic to, OR suffer debilitating side effects of AEDs. The individual has failed surgical intervention or is not a candidate for surgical resection. NOTE: In Pennsylvania requests for Vagus Nerve Stimulation will be considered on a case by case basis including for off-label uses. 1
2 Limitations: All other uses of VNS are not medically necessary. In addition: Individuals with a progressive disorder, such as malignant brain neoplasm or a progressive metabolic or degenerative disorder should not receive VNS. The U.S. Food and Drug Administration (FDA) states: o A NeuroCypernetic Prosthesis (NCP ) VNS system is contraindicated for individuals with a history of left or bilateral cervical vagotomy; o VNS devices are approved for individuals age 12 years and older; AND o Individuals with an implanted VNS device should avoid diatherpy, as this has the potential to cause the generator or leads to heat up and cause tissue damage.. Transmitter adjustments usually occur in an outpatient setting. NOTE: In Pennsylvania the use of Vagus Nerve Stimulation for treatment refractory depression would be managed under behavioral health benefits. Such use is still considered investigational despite past FDA approval. (George MS, 2013) Alternative covered services- Pharmacotherapy with anticonvulsant medications. Desensitization of nerve receptors. Background The vagus nerve is a paired cranial nerve that originates in the brainstem and ends in the abdominal region. It carries both somatic and visceral afferent and efferent signals. The majority of its fibers are visceral afferents with widespread distribution. Vagus nerve stimulation (VNS) works on the basis that the vagal visceral afferents provide a diffuse central nervous system projection. The activation of the diffuse pathways has an effect upon neural excitability. Although the exact mechanism of VNS on neuronal excitability in not fully defined, it is known to activate well-defined reflexes, and evokes potentials from the cerebellum, cerebral cortex, hippocampus, and the thalamus. VNS has been used with success for individuals with epilepsy. A VNS system is composed of an electrode array, an implantable generator, and an external programming device. The programming device is used to make adjustments and changes to the stimulation settings, which are transmitted to the implanted generator. The VNS generator must be surgically implanted in the upper left area of the chest, or under the left arm. The left vagus nerve is selected due to its limited cardiac effects. A thin, platinum-based wire is affixed to the vagus nerve and attached to the electrode array. The flexible electrodes are threaded under the skin and connected to the VNS generator. After implantation is completed the external programmer is coded with instructions to stimulate the vagus nerve at a rate determined by the individual and the physician (Heck 2002). The programming may be done for specific frequencies, and is designed to reduce the volume of the recipient s seizure activity. Revisions to the VNS system, such as replacing the battery-powered generator, usually occur every one to five years. 2
3 The US Food and Drug Administration (FDA) has approved several VNS systems. None are approved for children less than 12 years of age. The implantation of a VNS device in children less than 12 years of age is considered an off-label use. The National Institute for Health and Clinical Excellence (NICE) guidelines support the use of VNS for refractory epilepsy in children when all of the following criteria are met (NICE 2004): The VNS implantation procedure is performed by a specialized pediatric epilepsy team; The VNS implantation procedure is directed by developmentally appropriate pre- and post-device implantation; AND There is quality-of-life monitoring post VNS implantation. Epilepsy is diagnosed by the occurrence and type of recurrent, unprovoked seizure activity, and electroencephalography (EEG) readings. Seizures occur when there are errant electrical discharges within the brain. They manifest with different physical symptoms relating to the area of brain where the errant electrical activity is located. Seizures are classified and subtyped by EEG. They are classified as general onset, complex partial or simple partial, and are distinguished by level of consciousness. Partial seizures affect only one hemisphere of the brain. Generalized seizures have errant electrical activity in large areas, and may affect both sides of the brain. Even with the advent of new medications and surgical techniques for the treatment of epilepsy, 20 percent to 50 percent of individuals with epilepsy have breakthrough seizures or experience adverse effects of anti-seizure medications. Current peer-reviewed medical literature and accepted evidence support the efficacy of VNS as a useful palliative alternative to reduce the frequency of seizures for individuals who have severe generalized epilepsy with atonic or tonic-clonic seizures. Peer-reviewed literature also supports the use of VNS as an effective treatment for medically refractory partial-onset seizures, as well as in individuals who have failed surgery, are not candidates for surgery and are not able to benefit from anti-epileptic drugs (AEDS). Some of the benefits of using VNS may include a reduction in seizure frequency, improved recovery periods after seizures, and a lessening of seizure clusters. Although VNS may reduce the frequency and magnitude of seizure activity, individuals still need to remain on an anti-seizure medicinal regimen. In addition, individuals need to be apprised of the possible adverse effects of VNS therapy, including cough, headache, neck and incisional pain, and voice alterations (Schlaepfer 2008). A meta-analysis of VNS identified 74 clinical trials with 3,321 individuals with intractable epilepsy (Englot 2011a). Participants of studies showed an average 36% to 45% reduction in seizure frequency at three to 12 months after implantation, and a 51% reduction after more than one year of using the therapy. Posttraumatic epilepsy and tuberous sclerosis were positive predictors of a favorable outcome. Complete seizure abatement occurred in rare instances, and at least 25% of individuals did not respond to the VNS systems. An additional study by Englot (2011b) suggests that children age 18 years and younger, and individuals with generalized epilepsy, benefited significantly from VNS, in contrast to their exclusion from the initial approval of the device. Children younger than 12 years of age had responses similar to older counterparts with no increase in complications. Klinkenberg (2012) found similar results in 41 children with intractable epilepsy. However, the small size of the population samples limits the applicability of these conclusions to clinical practice. 3
4 In July of 2005, the FDA approved a VNS therapy system called NeuroCybernetic Prosthesis (NCP ) System (Cybertronics Inc., Houston, TX) for use as an adjunctive treatment in individuals age 18 and older with chronic or recurrent depression who have experienced a major depressive episode and have not adequately responded to four or more antidepressant treatments. VNS has been studied as a treatment for depression. Early available studies for VNS use in depression were subsidized by the leading manufacturer of the device and significantly flawed, particularly with regard to the lack of randomized controlled trials and statistical analysis. Several systematic reviews have examined the effectiveness of VNS for use in treatment resistant depression (TRD) (Mark 2006; Daban 2008; Gaynes 2011; Martin 2012; Mitchell 2013). However, only one double-blind, randomized study was identified with inconclusive results. There have been no studies to date that have demonstrated improved health outcomes with the use of VNS when compared to other treatment modalities for TRD. A systematic review by Martin and Martin-Sanchez (2012) identified a study with 235 participants that revealed no statistical differences between the control and the placebo groups, citing insufficient data were available to confirm that VNS was or was not effective in the treatment of depression. Though FDA approved, current peer-reviewed published literature does not at this time support the use of VNS devices for the treatment of depression. Clinical trials for the use of VNS for TRD do not demonstrate the effectiveness of VNS therapy on health outcomes in an investigational setting. Data from studies lacking a control group are of limited value (Bajbouj 2010). The American Psychiatric Association (APA) practice guideline for the treatment of major depressive disorders states that electroconvulsive therapy remains the best established efficacy above other stimulation therapies (Gelenberg 2010). In addition, there are no Class I evidence statements from any professional health care societies for the use of VNS in acute or chronic depression, and there is insufficient evidence to recommend the use of VNS for other neuropsychiatric disorders. (Fisher 1999; Kennedy 2009; National Institute for Clinical Excellence [NICE] 2009; Bauer 2013). Methods Searches: Keystone First searched PubMed and the databases of: UK National Health Services Centre for Reviews and Dissemination. Agency for Healthcare Research and Quality Guideline Clearinghouse and evidence-based practice centers. The Centers for Medicare & Medicaid Services. Searches were conducted on May 9, 2014, using the terms "Vagus nerve stimulation" [MeSH]. Included were: Systematic reviews, which pool results from multiple studies to achieve larger sample sizes and greater precision of effect estimation than in smaller primary studies. Systematic reviews use predetermined transparent methods to minimize bias, effectively treating the review as a scientific endeavor, and are thus rated highest in evidencegrading hierarchies. Guidelines based on systematic reviews. Economic analyses, such as cost-effectiveness, and benefit or utility studies (but not simple cost studies), reporting both costs and outcomes sometimes referred to as efficiency studies which also rank near the top of evidence hierarchies. 4
5 Findings No additional systematic reviews or meta-analyses published since 2013 were identified that addressed this policy. Glossary Afferent Conveying impulses toward the central nervous system. Efferent Conveying nerve impulses away from a part or organ. Medically Necessary- A service or benefit is Medically Necessary if it is compensable under the MA Program and if it meets any one of the following standards: The service or benefit will, or is reasonably expected to, prevent the onset of an illness, condition or disability. The service or benefit will, or is reasonably expected to, reduce or ameliorate the physical, mental or developmental effects of an illness, condition, injury or disability. The service or benefit will assist the Member to achieve or maintain maximum functional capacity in performing daily activities, taking into account both the functional capacity of the Member and those functional capacities that are appropriate for Members of the same age. Medically refractory seizures Seizures that are not controlled by medication, or cannot be treated with therapeutic levels of antiepileptic drugs (AEDs) because of intolerable side effects of these drugs. Partial seizure A seizure with a focal onset in one area of the brain, and may or may involve a loss of motor control or alteration of consciousness. These may be simple, complex or complex-partial, and secondarily generalized. Refractory Unrelenting, uncontrollable, resistant to therapy. Seizure Uncontrolled electrical activity in the brain that causes abnormal muscle movements, changes in behavior, or loss of consciousness. Somatic Involving or relating to skeletal muscle. Vagus nerve The tenth cranial nerve; a pair of nerves that originate in the brainstem and extend through the base of the skull, through the neck, down into the abdominal region. Visceral Relating to autonomic or involuntary functions. Related policies Keystone First Utilization Management program description. References Professional society guidelines/other: 5
6 Fisher R, Handforth A. Reassessment: VNS for epilepsy: a report of the Therapeutics and Technology Assessment Subcommittee of the American Academy of Neurology (AAN). Neurology. 1999; 53: Reaffirmed Oct 17, Gelenberg AJ, Freeman MP, Markowitz JC, Rosenbaum JF, Thase ME, Trivedi MH, et al. American Psychiatric Association (APA). Practice guideline for the treatment of patients with major depressive disorder. 3rd Edition. October Available at: Accessed May 9, Kennedy SH, Milev R, Giacobbe P, et al. Canadian Network for Mood and Anxiety Treatments (CANMAT) clinical guidelines for the management of major depressive disorder in adults: IV. Neurostimulation therapies. J Affect Disord. 2009;117:S44-S53. National Institute for Health and Clinical Excellence (NICE). IPG050 Vagus nerve stimulation for refractory epilepsy in children - guidance. [NICE Web site]. 03/16/04. Available at: Accessed May 9, National Institute for Health and Clinical Excellence (NICE). Vagus nerve stimulation for treatment-resistant depression. December Available at: Accessed May 9, Peer-reviewed references: Amar, A., Apuzzo, M., Liu C. Vagus nerve stimulation therapy after failed cranial surgery for intractable epilepsy: results from the vagus nerve stimulation therapy patient outcome registry. Neurosurgery Feb;62 Suppl 2: Bajbouj M, Merkl A, Schlaepfer TE, et al. Two-year outcome of vagus nerve stimulation in treatmentresistant depression. J Clin Psychopharmacol. 2010; 30(3): Bauer M, Pfennig A, Severus E, Whybrow PC, Angst J, Möller HJ, on behalf of the Task Force on Unipolar Depressive Disorders. World Federation of Societies of Biological Psychiatry (WFSBP) Guidelines for Biological Treatment of Unipolar Depressive Disorders, Part 1: Update 2013 on the acute and continuation treatment of unipolar depressive disorders. The World Journal of Biological Psychiatry. 2013; 14: Available at: orders_bauer_et_al_2013.pdf. Accessed May 8, Boggs JG. Simple partial seizures. [e-medicine Web site]. Original: 09/03/09. Updated: October 25, Available at: Accessed February 21, Daban C, Martinez-Aran A, Cruz N, Vieta E. Safety and efficacy of vagus nerve stimulation in treatmentresistant depression. A systematic review. J Affect Disord. 2008;110(1-2):1-15. Englot DJ, Chang EF, Auguste KI. Efficacy of vagus nerve stimulation for epilepsy by patient age, epilepsy duration, and seizure type. Neurosurg Clin N Am Oct;22(4):443-8, v. (b) 6
7 Englot DJ, Chang EF, Auguste KI. Vagus nerve stimulation for epilepsy: a meta-analysis of efficacy and predictors of response. J Neurosurg Dec;115(6): (a) Gaynes BN, Lux L, Lloyd S, Hansen RA, Gartlehner G, Thieda P, et al. Nonpharmacologic Interventions for Treatment-Resistant Depression in Adults. Comparative Effectiveness Review No. 33. (Prepared by RTI International-University of North Carolina (RTI-UNC) Evidence based Practice Center under Contract No I.) AHRQ Publication No. 11-EHC056-EF. Rockville, MD: Agency for Healthcare Research and Quality. September Available at: Accessed May 9, George MS, Aston-Jones G. Noninvasive techniques for probing neurocircuitry and treating illness: vagus nerve stimulation (VNS), transcranial magnetic stimulation (TMS) and transcranial direct current stimulation (tdcs). Neuropsychopharmacology Jan;35(1): George MS, Taylor JJ, Short B. Treating the depressions with superficial brain stimulation methods. Handb Clin Neurol. 2013;116: Heck C, Helmers S, DeGiorgio. VNS therapy, epilepsy, and device parameters: scientific parameters and recommendations for use. Neurology. 2002;59(6):1-12. Klinkenberg S, van den Bosch CN, Majoie HJ, Aalbers MW, Leenen L, Hendriksen J, et al. Behavioural and cognitive effects during vagus nerve stimulation in children with intractable epilepsy - A randomized controlled trial. Eur J Paediatr Neurol Aug 6. Kostov H, Larsson PG, Roste GK. Is vagus nerve stimulation a treatment option for patients with drugresistant idiopathic generalized epilepsy? Acta Neurol Scand Suppl. 2007;187:55-8. Mark D. Vagus nerve stimulation for treatment-resistant depression. Chicago, IL, USA: Blue Cross and Blue Shield Association, Technology Evaluation Center. TEC Assessment Program; 21(7) Martin JL, Martin-Sanchez E. Systematic review and meta-analysis of vagus nerve stimulation in the treatment of depression: variable results based on study designs. Eur Psychiatry Apr;27(3): Mitchell J, Trangle M, Degnan B, Gabert T, Haight B, Kessler D, et al. Institute for Clinical Systems Improvement. Adult Depression in Primary Care. Updated September Available at: Accessed May 8, 2014 Montavont A, Demarquay G, Ryvlin P, et al. Long-term efficiency of vagus nerve stimulation (VNS) in nonsurgical refractory epilepsies in adolescents and adults [article in French]. Rev Neurol (Paris). 2007;163(12): Murro AM. Complex partial seizures. [e-medicine website]. 10/11/06. Available at: Accessed May 3, National Institute of Neurological Disorders and Stroke (NINDS). National Institutes of Health. NINDS epilepsy information page. [NINDS Web site]. Updated April 8, Available at: Accessed May 9,
8 Rush AJ, Marangell LB, Sackeim HA, et al. Vagus nerve stimulation for treatment-resistant depression: a randomized, controlled acute phase trial. Biol Psychiatry. 2005;58(5): Schlaepfer TE, Frick C, Zobel A, et al. Vagus nerve stimulation for depression: efficacy and safety in a European study. Psychol Med 2008;38: Sperling W, Reulbach U, Kornhuber J. Clinical benefits and cost effectiveness of vagus nerve stimulation in a long-term treatment of patients with major depression. Pharmacopsychiatry. 2009;42(3): Tecoma ES, Iragui VJ. Vagus nerve stimulation use and effect in epilepsy: what have we learned? Epilepsy Behavior. 2006;8(1); U.S. Food and Drug Administration (FDA). New Device Approval VNS Therapy System. P970003/S050. Updated July 18, Available at: You SJ, Kang HC, Kim HD, et al. Vagus nerve stimulation in intractable childhood epilepsy: a Korean multicenter experience. J Korean Med Sci. 2007;22(3): Clinical Trials: Searched clinicaltrials.gov May 9, 2014 using term vagus nerve stimulation Open Studies: 25 studies found 20 relevant studies identified The Centers for Medicare & Medicaid Services (CMS) National Coverage Determination Centers for Medicare & Medicaid Services (CMS). National Coverage Determination (NCD) Vagus Nerve Stimulation (VNS). [CMS website]. Effective May 4, Available at: p=title&keywordlookup=title&keywordsearchtype=and&keywordsearchtype=and&bc=gaaaacaaaaa A&. Accessed May 9, Local Coverage Determinations None identified. 8
9 Commonly Submitted Codes Below are the most commonly submitted codes for the service(s)/item(s) subject to this policy. This is not an exhaustive list of codes. Providers are expected to consult the appropriate coding manuals and bill accordingly. CPT Code Description Comment Insertion or replacement of cranial neurostimulator pulse generator or receiver, direct or inductive coupling; with connection to a single electrode array Insertion or replacement of cranial neurostimulator pulse generator or receiver, direct or inductive coupling; with connection to two or more electrode arrays Revision or removal of cranial neurostimulator pulse generator or receiver. Do not report in conjunction with 61885, or Percutaneous implantation of neurostimulator electrode array; cranial nerve Incision for implantation of cranial nerve (e.g., vagus nerve) neurostimulator electrode array and pulse generator Revision or replacement of cranial nerve (e.g., vagus nerve) neurostimulator electrode array, including connection to existing pulse generator Removal of cranial nerve (e.g., vagus nerve neurostimulator electrode array and pulse generator Revision or removal of peripheral neurostimulator electrode array Electronic analysis of implanted neurostimulator pulse generator system (e.g., rate, pulse amplitude, pulse duration, configuration of wave form, battery status, electrode selectability, output modulation, cycling, impedance and patient compliance measurements); simple or complex brain, spinal cord, or peripheral (ie, cranial nerve, peripheral nerve, sacral nerve, neuromuscular) neurostimulator pulse generator/transmitter, without reprogramming Complex cranial nerve neurostimulator pulse generator/transmitter, with intraoperative or subsequent programming, with or without nerve interface testing, first hour Complex cranial nerve neurostimulator pulse generator/transmitter, with intraoperative or subsequent programming, each additional 30 minutes after first hour (list separately in addition to code for primary procedure). Use in conjunction with ICD-9 Code Description Comment Generalized nonconvulsive epilepsy with intractable epilepsy Generalized convulsive epilepsy with intractable epilepsy. 9
10 ICD-9 Code Description Comment Localization-related (focal) (partial) epilepsy and epileptic syndromes with complex partial seizures with intractable epilepsy. Localization-related (focal) (partial) epilepsy and epileptic syndromes with simple partial seizures, with intractable epilepsy. NCD: must be reported, as appropriate, to receive VNS NCD: must be reported, as appropriate, to receive VNS Infantile spasms with intractable epilepsy Epilepsia partialis continua with intractable epilepsy Other forms of epilepsy and recurrent seizures, with intractable epilepsy Unspecified epilepsy with intractable epilepsy Other convulsions. Policy # , Appendix ICD-10, for informational purposes only until the ICD-10 compliance date ICD-10 Code Description Comment G Generalized idiopathic epilepsy and epileptic syndromes, intractable, with status epilepticus. Related to ICD & G Generalized idiopathic epilepsy and epileptic syndromes, intractable, without status epilepticus. G Localization-related (focal) (partial) symptomatic epilepsy and epileptic syndromes with complex partial seizures, intractable, with status epilepticus. Related to ICD G Localization-related (focal) (partial) symptomatic epilepsy and epileptic syndromes with complex partial seizures, intractable, without status epilepticus. G Localization-related (focal) (partial) idiopathic epilepsy and epileptic syndromes with seizures of localized onset, intractable, with status epilepticus. Related to ICD-9s and , and G Localization-related (focal) (partial) idiopathic epilepsy and epileptic syndromes with seizures of localized onset, intractable, without status epilepticus. G Localization-related (focal) (partial) symptomatic epilepsy and epileptic syndromes with simple partial seizures, intractable, with status epilepticus. G Localization-related (focal) (partial) symptomatic epilepsy and epileptic syndromes with simple partial seizures, intractable, without status epilepticus. G Other generalized epilepsy and epileptic syndromes, intractable, with status epilepticus. Related to ICD
11 ICD-10 Code Description Comment G Other generalized epilepsy and epileptic syndromes, intractable, without status epilepticus. G Other epilepsy, intractable, with status epilepticus. G Other epilepsy, intractable, without status epilepticus. G Lennox-Gastaut syndrome, not intractable, with status epilepticus. Related to ICD G Lennox-Gastaut syndrome, intractable, with status epilepticus. G Lennox-Gastaut syndrome, intractable, without status epilepticus. G Epileptic spasms, intractable, with status epilepticus. G Epileptic spasms, intractable, without status epilepticus. G40.89 Other seizures. G40.A11 G40.A19 G40.B11 G40.B19 Absence epileptic syndrome, intractable, with status epilepticus. Absence epileptic syndrome, intractable, without status epilepticus. Juvenile myoclonic epilepsy, intractable, with status epilepticus. Juvenile myoclonic epilepsy, intractable, without status epilepticus. Related to ICD G Epilepsy, unspecified, intractable, with status epilepticus. Related to ICD G Epilepsy, unspecified, intractable, without status epilepticus. R56.9 Unspecified convulsions. Related to ICD
National Coverage Determination. Vagus Nerve Stimulation (VNS)
 National Coverage Determination Vagus Nerve Stimulation (VNS) Number NEURO-004 Contractor Name Wisconsin Physicians Service Insurance Corporation AMA CPT Copyright Statement CPT codes, descriptions and
National Coverage Determination Vagus Nerve Stimulation (VNS) Number NEURO-004 Contractor Name Wisconsin Physicians Service Insurance Corporation AMA CPT Copyright Statement CPT codes, descriptions and
Therapies for Treatment- Resistant Depression. A Review of the Research
 Therapies for Treatment- Resistant Depression A Review of the Research Is This Information Right for Me or Someone I Care For? Yes, if: A doctor or health care professional has told you that you have major
Therapies for Treatment- Resistant Depression A Review of the Research Is This Information Right for Me or Someone I Care For? Yes, if: A doctor or health care professional has told you that you have major
Corporate Medical Policy
 Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: spinal_cord_stimulation 3/1980 10/2015 10/2016 10/2015 Description of Procedure or Service Spinal cord stimulation
Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: spinal_cord_stimulation 3/1980 10/2015 10/2016 10/2015 Description of Procedure or Service Spinal cord stimulation
Local Coverage Determination (LCD): Special EEG Tests (L33699) Contractor Information
 Local Coverage Determination (LCD): Special EEG Tests (L33699) Contractor Information Contractor Name First Coast Service Options, Inc. Document Information LCD ID L33699 LCD Title Special EEG Tests Original
Local Coverage Determination (LCD): Special EEG Tests (L33699) Contractor Information Contractor Name First Coast Service Options, Inc. Document Information LCD ID L33699 LCD Title Special EEG Tests Original
VNS Therapy for Epilepsy
 VNS Therapy for Epilepsy This pamphlet provides general information for the public. It is not intended to cover all possible uses, directions, precautions, interactions or adverse effects involving any
VNS Therapy for Epilepsy This pamphlet provides general information for the public. It is not intended to cover all possible uses, directions, precautions, interactions or adverse effects involving any
Clinical Policy Title: Leiomyosarcoma and Laparoscopic Power Morcellation
 Clinical Policy Title: Leiomyosarcoma and Laparoscopic Power Morcellation Clinical Policy Number: 12.03.01 Effective Date: January 1, 2015 Initial Review Date: August 20, 2014 Most Recent Review Date:
Clinical Policy Title: Leiomyosarcoma and Laparoscopic Power Morcellation Clinical Policy Number: 12.03.01 Effective Date: January 1, 2015 Initial Review Date: August 20, 2014 Most Recent Review Date:
Spinal Cord Stimulation (SCS) Therapy: Fact Sheet
 Spinal Cord Stimulation (SCS) Therapy: Fact Sheet What is SCS Therapy? Spinal cord stimulation (SCS) may be a life-changing 1 surgical option for patients to control their chronic neuropathic pain and
Spinal Cord Stimulation (SCS) Therapy: Fact Sheet What is SCS Therapy? Spinal cord stimulation (SCS) may be a life-changing 1 surgical option for patients to control their chronic neuropathic pain and
Protocol. Vagus Nerve Stimulation
 Protocol Vagus Nerve Stimulation (70120) Medical Benefit Effective Date: 01/01/15 Next Review Date: 11/16 Preauthorization Yes Review Dates: 01/07, 05/08, 11/08, 03/09, 01/10, 01/11, 01/12, 01/13, 01/14,
Protocol Vagus Nerve Stimulation (70120) Medical Benefit Effective Date: 01/01/15 Next Review Date: 11/16 Preauthorization Yes Review Dates: 01/07, 05/08, 11/08, 03/09, 01/10, 01/11, 01/12, 01/13, 01/14,
NEUROMODULATION THERAPY ACCESS COALITION POSITION STATEMENT ON SPINAL CORD NEUROSTIMULATION
 NEUROMODULATION THERAPY ACCESS COALITION POSITION STATEMENT ON SPINAL CORD NEUROSTIMULATION Introduction: Spinal cord stimulation (SCS) is an established treatment for chronic neuropathic pain, which can
NEUROMODULATION THERAPY ACCESS COALITION POSITION STATEMENT ON SPINAL CORD NEUROSTIMULATION Introduction: Spinal cord stimulation (SCS) is an established treatment for chronic neuropathic pain, which can
BOTOX Injection (Onabotulinumtoxin A) for Migraine Headaches [Preauthorization Required]
![BOTOX Injection (Onabotulinumtoxin A) for Migraine Headaches [Preauthorization Required] BOTOX Injection (Onabotulinumtoxin A) for Migraine Headaches [Preauthorization Required]](/thumbs/29/13417512.jpg) BOTOX Injection (Onabotulinumtoxin A) for Migraine Headaches [Preauthorization Required] Medical Policy: MP-RX-01-11 Original Effective Date: March 24, 2011 Reviewed: Revised: This policy applies to products
BOTOX Injection (Onabotulinumtoxin A) for Migraine Headaches [Preauthorization Required] Medical Policy: MP-RX-01-11 Original Effective Date: March 24, 2011 Reviewed: Revised: This policy applies to products
VAGUS NERVE STIMULATION
 VAGUS NERVE STIMULATION Policy Number: SURGERY 073.12 T2 Effective Date: December 1, 2015 CLINICAL POLICY Table of Contents CONDITIONS OF COVERAGE... BENEFIT CONSIDERATIONS... COVERAGE RATIONALE... APPLICABLE
VAGUS NERVE STIMULATION Policy Number: SURGERY 073.12 T2 Effective Date: December 1, 2015 CLINICAL POLICY Table of Contents CONDITIONS OF COVERAGE... BENEFIT CONSIDERATIONS... COVERAGE RATIONALE... APPLICABLE
Clinical Policy Title: Air Ambulance Transport
 Clinical Policy Title: Air Ambulance Transport Clinical Policy Number: 18.02.02 Effective Date: Sept. 1, 2014 Initial Review Date: April 16, 2014 Most Recent Review Date: May 21, 2014 Next Review Date:
Clinical Policy Title: Air Ambulance Transport Clinical Policy Number: 18.02.02 Effective Date: Sept. 1, 2014 Initial Review Date: April 16, 2014 Most Recent Review Date: May 21, 2014 Next Review Date:
How To Determine If A Fall Prevention Program Is Effective
 Clinical Policy Title: Medical alert devices and other interventions for vulnerable peoples safety at home Clinical Policy Number: 17.01.02 Effective Date: April 1, 2015 Initial Review Date: January 21,
Clinical Policy Title: Medical alert devices and other interventions for vulnerable peoples safety at home Clinical Policy Number: 17.01.02 Effective Date: April 1, 2015 Initial Review Date: January 21,
Best Practices Treatment Guideline for Major Depression
 Best Practices Treatment Guideline for Major Depression Special Report on New Depression Treatment Technology Based on 2010 APA Practice Guidelines Best Practices Guideline for the Treatment of Patients
Best Practices Treatment Guideline for Major Depression Special Report on New Depression Treatment Technology Based on 2010 APA Practice Guidelines Best Practices Guideline for the Treatment of Patients
Neurological System Best Practice Documentation
 Neurological System Best Practice Documentation Click on the desired Diagnoses link or press Enter to view all information. Diagnoses: Dementia Delirium/Encephalopathy Parkinson s Epilepsy /Seizure Migraines
Neurological System Best Practice Documentation Click on the desired Diagnoses link or press Enter to view all information. Diagnoses: Dementia Delirium/Encephalopathy Parkinson s Epilepsy /Seizure Migraines
How To Decide If A Helicopter Is Right For A Patient
 Clinical Policy Title: Air Ambulance Transport Clinical Policy Number: 18.02.02 Effective Date: September 1, 2014 Initial Review Date: April 16, 2014 Most Recent Review Date: May 20, 2015 Next Review Date:
Clinical Policy Title: Air Ambulance Transport Clinical Policy Number: 18.02.02 Effective Date: September 1, 2014 Initial Review Date: April 16, 2014 Most Recent Review Date: May 20, 2015 Next Review Date:
Clinical guidance for MRI referral
 MRI for cervical radiculopathy Referral by a medical practitioner (excluding a specialist or consultant physician) for a scan of spine for a patient 16 years or older for suspected: cervical radiculopathy
MRI for cervical radiculopathy Referral by a medical practitioner (excluding a specialist or consultant physician) for a scan of spine for a patient 16 years or older for suspected: cervical radiculopathy
Neurostimulation: Orthopaedic Institute of Ohio 801 Medical Drive Lima, Ohio 45804 419-222-6622
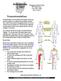 801 Medical Drive Lima, Ohio 45804 419-222-6622 Neurostimulation is the stimulation of the spinal cord by tiny electrical impulses. An implanted lead (a flexible insulated wire), which is powered by an
801 Medical Drive Lima, Ohio 45804 419-222-6622 Neurostimulation is the stimulation of the spinal cord by tiny electrical impulses. An implanted lead (a flexible insulated wire), which is powered by an
Behavioral Health Policy Phototherapy Light for the Treatment of Seasonal Affective (SAD) and Other Depressive Disorders
 Behavioral Health Policy Phototherapy Light for the Treatment of Seasonal Affective (SAD) and Other Depressive Disorders Table of Contents Policy: Commercial Coding Information Information Pertaining to
Behavioral Health Policy Phototherapy Light for the Treatment of Seasonal Affective (SAD) and Other Depressive Disorders Table of Contents Policy: Commercial Coding Information Information Pertaining to
Contractor Number 11302. Oversight Region Region IV
 Local Coverage Determination (LCD): Spinal Cord Stimulators for Chronic Pain (L32549) Contractor Information Contractor Name Palmetto GBA opens in new window Contractor Number 11302 Contractor Type MAC
Local Coverage Determination (LCD): Spinal Cord Stimulators for Chronic Pain (L32549) Contractor Information Contractor Name Palmetto GBA opens in new window Contractor Number 11302 Contractor Type MAC
Pain Management. Practical Applications in Electrotherapy
 Pain Management Practical Applications in Electrotherapy The TENS Advantage Deliver Immediate Pain Relief using a unique waveform designed to help prevent nerve accommodation. Manage Dynamic Pain by adjusting
Pain Management Practical Applications in Electrotherapy The TENS Advantage Deliver Immediate Pain Relief using a unique waveform designed to help prevent nerve accommodation. Manage Dynamic Pain by adjusting
Electroconvulsive Therapy - ECT
 Electroconvulsive Therapy - ECT Introduction Electroconvulsive therapy, or ECT, is a safe and effective treatment that may reduce symptoms related to depression or mental illness. During ECT, certain parts
Electroconvulsive Therapy - ECT Introduction Electroconvulsive therapy, or ECT, is a safe and effective treatment that may reduce symptoms related to depression or mental illness. During ECT, certain parts
Clinical Policy Title: Home uterine activity monitoring
 Clinical Policy Title: Home uterine activity monitoring Clinical Policy Number: 12.01.01 Effective Date: August 19, 2015 Initial Review Date: July 17, 2013 Most Recent Review Date: July 15, 2015 Next Review
Clinical Policy Title: Home uterine activity monitoring Clinical Policy Number: 12.01.01 Effective Date: August 19, 2015 Initial Review Date: July 17, 2013 Most Recent Review Date: July 15, 2015 Next Review
Intrathecal Baclofen for CNS Spasticity
 Intrathecal Baclofen for CNS Spasticity Last Review Date: November 13, 2015 Number: MG.MM.ME.31bC5 Medical Guideline Disclaimer Property of EmblemHealth. All rights reserved. The treating physician or
Intrathecal Baclofen for CNS Spasticity Last Review Date: November 13, 2015 Number: MG.MM.ME.31bC5 Medical Guideline Disclaimer Property of EmblemHealth. All rights reserved. The treating physician or
Spinal Cord Stimulation Business Case for
 Spinal Cord Stimulation Business Case for Failed Back Surgery Syndrome Understanding How Various Stakeholders Gauge Success in Treating Chronic Pain FBSS is a subset of chronic low back pain, which is
Spinal Cord Stimulation Business Case for Failed Back Surgery Syndrome Understanding How Various Stakeholders Gauge Success in Treating Chronic Pain FBSS is a subset of chronic low back pain, which is
VAGUS NERVE STIMULATION FOR PATIENTS IN RESIDENTIAL TREATMENT FACILITIES
 VAGUS NERVE STIMULATION FOR PATIENTS IN RESIDENTIAL TREATMENT FACILITIES Michael Frost, MD Roger Huf, MD John Gates, MD This paper has been prepared specifically for: American Epilepsy Society Annual Meeting
VAGUS NERVE STIMULATION FOR PATIENTS IN RESIDENTIAL TREATMENT FACILITIES Michael Frost, MD Roger Huf, MD John Gates, MD This paper has been prepared specifically for: American Epilepsy Society Annual Meeting
Background. Physiotherapy Management
 Mr. Alex Wong, Senior physiotherapist, QEH Mr. Barry Ma, Physiotherapist, QEH Background Trigeminal neuralgia is a neuropathic pain syndrome that affects the trigeminal or 5 th cranial nerve, one of the
Mr. Alex Wong, Senior physiotherapist, QEH Mr. Barry Ma, Physiotherapist, QEH Background Trigeminal neuralgia is a neuropathic pain syndrome that affects the trigeminal or 5 th cranial nerve, one of the
Webinar title: Know Your Options for Treating Severe Spasticity
 Webinar title: Know Your Options for Treating Severe Spasticity Presented by: Dr. Gerald Bilsky, Physiatrist Medical Director of Outpatient Services and Associate Medical Director of Acquired Brain Injury
Webinar title: Know Your Options for Treating Severe Spasticity Presented by: Dr. Gerald Bilsky, Physiatrist Medical Director of Outpatient Services and Associate Medical Director of Acquired Brain Injury
Alfredo Velez, M.D. ECT: A REVIEW
 Alfredo Velez, M.D. ECT: A REVIEW ECT is not a treatment of last resort!!! History Experiments with medical electricity begin in 18 th Century (!) 1934 Chemical convulsive therapy, camphor, metrazole Hungarian
Alfredo Velez, M.D. ECT: A REVIEW ECT is not a treatment of last resort!!! History Experiments with medical electricity begin in 18 th Century (!) 1934 Chemical convulsive therapy, camphor, metrazole Hungarian
NeuroPace. RNS System Patient Manual
 NeuroPace RNS System Patient Manual CAUTION: Federal law restricts this device to sale by or on the order of a physician. 01/2013 DN 1013555 Rev 2 1 2013 NeuroPace, Inc. 2 This Manual supports: RNS Neurostimulator
NeuroPace RNS System Patient Manual CAUTION: Federal law restricts this device to sale by or on the order of a physician. 01/2013 DN 1013555 Rev 2 1 2013 NeuroPace, Inc. 2 This Manual supports: RNS Neurostimulator
Neurology. Medical Staff: Children s Access Center. Outpatient Referral. Neurology Office Numbers
 Neurology The Neurology Practice at Children s Hospital Central California provides diagnostic services, medical treatment, and follow-up care to infants, children, and adolescents who have suspected or
Neurology The Neurology Practice at Children s Hospital Central California provides diagnostic services, medical treatment, and follow-up care to infants, children, and adolescents who have suspected or
What is epilepsy? English
 What is epilepsy? English WHAT IS EPILEPSY? An epileptic seizure is the term used for a temporary brain dysfunction due to a sudden and uncontrolled disturbance of the brain s electrical activity. Epilepsy
What is epilepsy? English WHAT IS EPILEPSY? An epileptic seizure is the term used for a temporary brain dysfunction due to a sudden and uncontrolled disturbance of the brain s electrical activity. Epilepsy
Epilepsy 101: Getting Started
 American Epilepsy Society 1 Epilepsy 101 for nurses has been developed by the American Epilepsy Society to prepare professional nurses to understand the general issues, concerns and needs of people with
American Epilepsy Society 1 Epilepsy 101 for nurses has been developed by the American Epilepsy Society to prepare professional nurses to understand the general issues, concerns and needs of people with
THE DEPRESSION RESEARCH CLINIC Department of Psychiatry and Behavioral Sciences Stanford University, School of Medicine
 THE DEPRESSION RESEARCH CLINIC Department of Psychiatry and Behavioral Sciences Stanford University, School of Medicine Volume 1, Issue 1 August 2007 The Depression Research Clinic at Stanford University
THE DEPRESSION RESEARCH CLINIC Department of Psychiatry and Behavioral Sciences Stanford University, School of Medicine Volume 1, Issue 1 August 2007 The Depression Research Clinic at Stanford University
Vagus nerve stimulation therapy in epilepsy
 Published on Epilepsy Action (http://www.epilepsy.org.uk/) Page generated on 15/05/2013 Vagus nerve stimulation therapy in epilepsy In this section Introduction About VNS therapy How VNS therapy works
Published on Epilepsy Action (http://www.epilepsy.org.uk/) Page generated on 15/05/2013 Vagus nerve stimulation therapy in epilepsy In this section Introduction About VNS therapy How VNS therapy works
MEDICAL POLICY SUBJECT: GASTRIC ELECTRICAL STIMULATION
 MEDICAL POLICY SUBJECT: GASTRIC ELECTRICAL PAGE: 1 OF: 6 If a product excludes coverage for a service, it is not covered, and medical policy criteria do not apply. If a commercial product, including an
MEDICAL POLICY SUBJECT: GASTRIC ELECTRICAL PAGE: 1 OF: 6 If a product excludes coverage for a service, it is not covered, and medical policy criteria do not apply. If a commercial product, including an
MEDICAL POLICY SUBJECT: COGNITIVE REHABILITATION. POLICY NUMBER: 8.01.19 CATEGORY: Therapy/Rehabilitation
 MEDICAL POLICY SUBJECT: COGNITIVE REHABILITATION PAGE: 1 OF: 5 If the member's subscriber contract excludes coverage for a specific service it is not covered under that contract. In such cases, medical
MEDICAL POLICY SUBJECT: COGNITIVE REHABILITATION PAGE: 1 OF: 5 If the member's subscriber contract excludes coverage for a specific service it is not covered under that contract. In such cases, medical
Coverage and Authorization Services is available to respond to your coding questions toll-free at 800-292-2903.
 For Urinary Control Commonly Billed Codes October 2010 Medtronic provides this information for your convenience only. It is not intended as a recommendation regarding clinical practice. It is the responsibility
For Urinary Control Commonly Billed Codes October 2010 Medtronic provides this information for your convenience only. It is not intended as a recommendation regarding clinical practice. It is the responsibility
Local Coverage Determination (LCD): Spinal Cord Stimulation (Dorsal Column Stimulation) (L34705)
 Local Coverage Determination (LCD): Spinal Cord Stimulation (Dorsal Column Stimulation) (L34705) Contractor Information Contractor Name Novitas Solutions, Inc. LCD Information Document Information LCD
Local Coverage Determination (LCD): Spinal Cord Stimulation (Dorsal Column Stimulation) (L34705) Contractor Information Contractor Name Novitas Solutions, Inc. LCD Information Document Information LCD
Clinical Policy Title: Medical alert devices and other interventions for vulnerable peoples safety at home
 Clinical Policy Title: Medical alert devices and other interventions for vulnerable peoples safety at home Clinical Policy Number: 17.01.02 Effective Date: April 1, 2015 Initial Review Date: January 21,
Clinical Policy Title: Medical alert devices and other interventions for vulnerable peoples safety at home Clinical Policy Number: 17.01.02 Effective Date: April 1, 2015 Initial Review Date: January 21,
MEDICAL POLICY POLICY TITLE POLICY NUMBER NEUROPSYCHOLOGICAL TESTING (FOR MEDICAL PURPOSES) MP-4.027
 Original Issue Date (Created): July 1, 2005 Most Recent Review Date (Revised): November 26, 2013 Effective Date: February 1, 2014 I. POLICY Neuropsychological testing may be considered medically necessary
Original Issue Date (Created): July 1, 2005 Most Recent Review Date (Revised): November 26, 2013 Effective Date: February 1, 2014 I. POLICY Neuropsychological testing may be considered medically necessary
Fit, (falls) and funny turns. Richard J Davenport Consultant Neurologist Edinburgh
 Fit, (falls) and funny turns Richard J Davenport Consultant Neurologist Edinburgh The plan Epilepsy nuggets 10 things I would like GPs to know This week s FS clinic What is epilepsy? Characterised by two
Fit, (falls) and funny turns Richard J Davenport Consultant Neurologist Edinburgh The plan Epilepsy nuggets 10 things I would like GPs to know This week s FS clinic What is epilepsy? Characterised by two
Transmittal 55 Date: MAY 5, 2006. SUBJECT: Changes Conforming to CR3648 for Therapy Services
 CMS Manual System Pub 100-03 Medicare National Coverage Determinations Department of Health & Human Services (DHHS) Centers for Medicare & Medicaid Services (CMS) Transmittal 55 Date: MAY 5, 2006 Change
CMS Manual System Pub 100-03 Medicare National Coverage Determinations Department of Health & Human Services (DHHS) Centers for Medicare & Medicaid Services (CMS) Transmittal 55 Date: MAY 5, 2006 Change
How To Get A Dbs Therapy For Obsessive Compulsive Disorder
 Sample Appeal Letters Appeals Payers who deny coverage usually do so for one of these three reasons: The therapy is investigational / experimental The therapy is not medically necessary The therapy is
Sample Appeal Letters Appeals Payers who deny coverage usually do so for one of these three reasons: The therapy is investigational / experimental The therapy is not medically necessary The therapy is
A systematic review of focused topics for the management of spinal cord injury and impairment
 A systematic review of focused topics for the management of spinal cord injury and impairment icahe, University of South Australia For the NZ Spinal Cord Impairment Strategy Introduction This was the third
A systematic review of focused topics for the management of spinal cord injury and impairment icahe, University of South Australia For the NZ Spinal Cord Impairment Strategy Introduction This was the third
NeuroStar TMS Therapy Patient Guide for Treating Depression
 NeuroStar TMS Therapy Patient Guide for Treating Depression This NeuroStar TMS Therapy Patient Guide for Treating Depression provides important safety and use information for you to consider about treating
NeuroStar TMS Therapy Patient Guide for Treating Depression This NeuroStar TMS Therapy Patient Guide for Treating Depression provides important safety and use information for you to consider about treating
A. Study Purpose and Rationale
 Effects of deep brain stimulation on seizure number and signal synchrony in epileptogenic brain regions A. Study Purpose and Rationale Epilepsy is categorized as any of various disorders marked by repetitive
Effects of deep brain stimulation on seizure number and signal synchrony in epileptogenic brain regions A. Study Purpose and Rationale Epilepsy is categorized as any of various disorders marked by repetitive
How To Cover Occupational Therapy
 Guidelines for Medical Necessity Determination for Occupational Therapy These Guidelines for Medical Necessity Determination (Guidelines) identify the clinical information MassHealth needs to determine
Guidelines for Medical Necessity Determination for Occupational Therapy These Guidelines for Medical Necessity Determination (Guidelines) identify the clinical information MassHealth needs to determine
Vagus Nerve Stimulation (V.N.S) Training for staff who support people with epilepsy and a vagus nerve implant
 Vagus Nerve Stimulation (V.N.S) Training for staff who support people with epilepsy and a vagus nerve implant Vagus Nerve Stimulation is used in people whose epilepsy has proved difficult to control using
Vagus Nerve Stimulation (V.N.S) Training for staff who support people with epilepsy and a vagus nerve implant Vagus Nerve Stimulation is used in people whose epilepsy has proved difficult to control using
1695 N.W. 9th Avenue, Suite 3302H Miami, FL. 33136. Days and Hours: Monday Friday 8:30a.m. 6:00p.m. (305) 355 9028 (JMH, Downtown)
 UNIVERSITY OF MIAMI, LEONARD M. MILLER SCHOOL OF MEDICINE CLINICAL NEUROPSYCHOLOGY UHEALTH PSYCHIATRY AT MENTAL HEALTH HOSPITAL CENTER 1695 N.W. 9th Avenue, Suite 3302H Miami, FL. 33136 Days and Hours:
UNIVERSITY OF MIAMI, LEONARD M. MILLER SCHOOL OF MEDICINE CLINICAL NEUROPSYCHOLOGY UHEALTH PSYCHIATRY AT MENTAL HEALTH HOSPITAL CENTER 1695 N.W. 9th Avenue, Suite 3302H Miami, FL. 33136 Days and Hours:
New Onset Seizure Clinic
 New Onset Seizure Clinic Timely Care from the Region s Experts The New Onset Seizure Clinic at UH Rainbow Babies & Children s Hospital provides expert diagnostic services, referrals, treatment and follow-up
New Onset Seizure Clinic Timely Care from the Region s Experts The New Onset Seizure Clinic at UH Rainbow Babies & Children s Hospital provides expert diagnostic services, referrals, treatment and follow-up
CHILDREN S NEUROSCIENCE CENTER
 CHILDREN S NEUROSCIENCE CENTER W hen families come to Children s Memorial Hermann Hospital, they expect to find the technological advances and healing expertise of a university-affiliated, academic hospital.
CHILDREN S NEUROSCIENCE CENTER W hen families come to Children s Memorial Hermann Hospital, they expect to find the technological advances and healing expertise of a university-affiliated, academic hospital.
MEDICAL CONTESTED CASE HEARING NO 12090 M6-12-38043-01 DECISION AND ORDER
 MEDICAL CONTESTED CASE HEARING NO 12090 M6-12-38043-01 DECISION AND ORDER This case is decided pursuant to Chapter 410 of the Texas Workers Compensation Act and Rules of the Division of Workers Compensation
MEDICAL CONTESTED CASE HEARING NO 12090 M6-12-38043-01 DECISION AND ORDER This case is decided pursuant to Chapter 410 of the Texas Workers Compensation Act and Rules of the Division of Workers Compensation
Clinical and Therapeutic Cannabis Information. Written by Cannabis Training University (CTU) All rights reserved
 Clinical and Therapeutic Cannabis Information Written by Cannabis Training University (CTU) All rights reserved Contents Introduction... 3 Chronic Pain... 6 Neuropathic Pain... 8 Movement Disorders...
Clinical and Therapeutic Cannabis Information Written by Cannabis Training University (CTU) All rights reserved Contents Introduction... 3 Chronic Pain... 6 Neuropathic Pain... 8 Movement Disorders...
2016 CODING FOR FETAL ALCOHOL SPECTRUM DISORDERS
 2016 CODING FOR FETAL ALCOHOL SPECTRUM DISORDERS Listed below are the most commonly used codes applicable to FASD patient care. Code Description ICD-10-CM Primary Diagnosis P04.3 Newborn (suspected to
2016 CODING FOR FETAL ALCOHOL SPECTRUM DISORDERS Listed below are the most commonly used codes applicable to FASD patient care. Code Description ICD-10-CM Primary Diagnosis P04.3 Newborn (suspected to
ALL ABOUT SPASTICITY. www.almirall.com. Solutions with you in mind
 ALL ABOUT SPASTICITY www.almirall.com Solutions with you in mind WHAT IS SPASTICITY? The muscles of the body maintain what is called normal muscle tone, a level of muscle tension that allows us to hold
ALL ABOUT SPASTICITY www.almirall.com Solutions with you in mind WHAT IS SPASTICITY? The muscles of the body maintain what is called normal muscle tone, a level of muscle tension that allows us to hold
Spinal Decompression
 Spinal Decompression Spinal decompression is just one more tool we have to treat radiculopathy. With appropriate education and exercises, this modality has been proven to assist in the resolution of symptoms
Spinal Decompression Spinal decompression is just one more tool we have to treat radiculopathy. With appropriate education and exercises, this modality has been proven to assist in the resolution of symptoms
VNS Therapy. 10th European Conference on Epilepsy and Society. Facing the Real Challenges of Difficult-to. to-treat treat Epilepsy
 10th European Conference on Epilepsy and Society Friday 4 August 2006 VNS Therapy Facing the Real Challenges of Difficult-to to-treat treat Epilepsy VNS Therapy Facing the Real Challenges of Difficult-to
10th European Conference on Epilepsy and Society Friday 4 August 2006 VNS Therapy Facing the Real Challenges of Difficult-to to-treat treat Epilepsy VNS Therapy Facing the Real Challenges of Difficult-to
Spinal cord stimulation
 Spinal cord stimulation This leaflet aims to answer your questions about having spinal cord stimulation. It explains the benefits, risks and alternatives, as well as what you can expect when you come to
Spinal cord stimulation This leaflet aims to answer your questions about having spinal cord stimulation. It explains the benefits, risks and alternatives, as well as what you can expect when you come to
The Brain and Spine CenTer
 The Br ain and Spine Center Choosing the right treatment partner is important for patients facing tumors involving the brain, spine or skull base. The Brain and Spine Center at The University of Texas
The Br ain and Spine Center Choosing the right treatment partner is important for patients facing tumors involving the brain, spine or skull base. The Brain and Spine Center at The University of Texas
The Detection of Neural Fatigue during intensive conditioning for football: The Potential of Transcranial Magnetic Stimulation
 The Detection of Neural Fatigue during intensive conditioning for football: The Potential of Transcranial Magnetic Stimulation Carl Wells PhD Sport Science Lead, Perform, National Football Centre, St.
The Detection of Neural Fatigue during intensive conditioning for football: The Potential of Transcranial Magnetic Stimulation Carl Wells PhD Sport Science Lead, Perform, National Football Centre, St.
Neck Pain & Cervicogenic Headache Integrating Research into Practice: San Luis Sports Therapy s Approach to Evidence-Based Practice
 Neck Pain & Cervicogenic Headache Integrating Research into Practice: San Luis Sports Therapy s Approach to Evidence-Based Practice PROBLEM: Neck Pain and Cervicogenic Headache 66% Proportion of individuals
Neck Pain & Cervicogenic Headache Integrating Research into Practice: San Luis Sports Therapy s Approach to Evidence-Based Practice PROBLEM: Neck Pain and Cervicogenic Headache 66% Proportion of individuals
Guidance on competencies for management of Cancer Pain in adults
 Guidance on competencies for management of Cancer Pain in adults Endorsed by: Contents Introduction A: Core competencies for practitioners in Pain Medicine B: Competencies for practitioners in Pain Medicine
Guidance on competencies for management of Cancer Pain in adults Endorsed by: Contents Introduction A: Core competencies for practitioners in Pain Medicine B: Competencies for practitioners in Pain Medicine
33 % of whiplash patients develop. headaches originating from the upper. cervical spine
 33 % of whiplash patients develop headaches originating from the upper cervical spine - Dr Nikolai Bogduk Spine, 1995 1 Physical Treatments for Headache: A Structured Review Headache: The Journal of Head
33 % of whiplash patients develop headaches originating from the upper cervical spine - Dr Nikolai Bogduk Spine, 1995 1 Physical Treatments for Headache: A Structured Review Headache: The Journal of Head
Corporate Medical Policy
 Corporate Medical Policy Electrical Stimulation for the Treatment of Arthritis File Name: Origination: Last CAP Review: Next CAP Review: Last Review: electrical_stimulation_for_the_treatment_of_arthritis
Corporate Medical Policy Electrical Stimulation for the Treatment of Arthritis File Name: Origination: Last CAP Review: Next CAP Review: Last Review: electrical_stimulation_for_the_treatment_of_arthritis
CLINICAL NEUROPHYSIOLOGY
 CLINICAL NEUROPHYSIOLOGY Barry S. Oken, MD, Carter D. Wray MD Objectives: 1. Know the role of EMG/NCS in evaluating nerve and muscle function 2. Recognize common EEG findings and their significance 3.
CLINICAL NEUROPHYSIOLOGY Barry S. Oken, MD, Carter D. Wray MD Objectives: 1. Know the role of EMG/NCS in evaluating nerve and muscle function 2. Recognize common EEG findings and their significance 3.
Chapter 7: The Nervous System
 Chapter 7: The Nervous System I. Organization of the Nervous System Objectives: List the general functions of the nervous system Explain the structural and functional classifications of the nervous system
Chapter 7: The Nervous System I. Organization of the Nervous System Objectives: List the general functions of the nervous system Explain the structural and functional classifications of the nervous system
Occupational Therapy Program
 Health Care Authority Occupational Therapy Program Billing Instructions [WAC 182-545-0300] About This Publication This publication supersedes all previous Agency Occupational Therapy Program Billing Instructions
Health Care Authority Occupational Therapy Program Billing Instructions [WAC 182-545-0300] About This Publication This publication supersedes all previous Agency Occupational Therapy Program Billing Instructions
PROVEN NON-INVASIVE TREATMENT FOR RHEUMATOID ARTHRITIS
 PROVEN NON-INVASIVE TREATMENT FOR RHEUMATOID ARTHRITIS BIONICARE HAND SYSTEM Non-drug, non-invasive Adjunctive therapy For patients symptomatic despite current therapy For patients intolerant to drug
PROVEN NON-INVASIVE TREATMENT FOR RHEUMATOID ARTHRITIS BIONICARE HAND SYSTEM Non-drug, non-invasive Adjunctive therapy For patients symptomatic despite current therapy For patients intolerant to drug
Reflex Physiology. Dr. Ali Ebneshahidi. 2009 Ebneshahidi
 Reflex Physiology Dr. Ali Ebneshahidi Reflex Physiology Reflexes are automatic, subconscious response to changes within or outside the body. a. Reflexes maintain homeostasis (autonomic reflexes) heart
Reflex Physiology Dr. Ali Ebneshahidi Reflex Physiology Reflexes are automatic, subconscious response to changes within or outside the body. a. Reflexes maintain homeostasis (autonomic reflexes) heart
STROKE CARE NOW NETWORK CONFERENCE MAY 22, 2014
 STROKE CARE NOW NETWORK CONFERENCE MAY 22, 2014 Rehabilitation Innovations in Post- Stroke Recovery Madhav Bhat, MD Fort Wayne Neurological Center DISCLOSURE Paid speaker for TEVA Neuroscience Program.
STROKE CARE NOW NETWORK CONFERENCE MAY 22, 2014 Rehabilitation Innovations in Post- Stroke Recovery Madhav Bhat, MD Fort Wayne Neurological Center DISCLOSURE Paid speaker for TEVA Neuroscience Program.
Nonoperative Management of Herniated Cervical Intervertebral Disc With Radiculopathy. Spine Volume 21(16) August 15, 1996, pp 1877-1883
 Nonoperative Management of Herniated Cervical Intervertebral Disc With Radiculopathy 1 Spine Volume 21(16) August 15, 1996, pp 1877-1883 Saal, Joel S. MD; Saal, Jeffrey A. MD; Yurth, Elizabeth F. MD FROM
Nonoperative Management of Herniated Cervical Intervertebral Disc With Radiculopathy 1 Spine Volume 21(16) August 15, 1996, pp 1877-1883 Saal, Joel S. MD; Saal, Jeffrey A. MD; Yurth, Elizabeth F. MD FROM
PSYCHOLOGICAL AND NEUROPSYCHOLOGICAL TESTING
 Status Active Medical and Behavioral Health Policy Section: Behavioral Health Policy Number: X-45 Effective Date: 01/22/2014 Blue Cross and Blue Shield of Minnesota medical policies do not imply that members
Status Active Medical and Behavioral Health Policy Section: Behavioral Health Policy Number: X-45 Effective Date: 01/22/2014 Blue Cross and Blue Shield of Minnesota medical policies do not imply that members
Step 4: Complex and severe depression in adults
 Step 4: Complex and severe depression in adults A NICE pathway brings together all NICE guidance, quality standards and materials to support implementation on a specific topic area. The pathways are interactive
Step 4: Complex and severe depression in adults A NICE pathway brings together all NICE guidance, quality standards and materials to support implementation on a specific topic area. The pathways are interactive
Nerves and Nerve Impulse
 Nerves and Nerve Impulse Terms Absolute refractory period: Period following stimulation during which no additional action potential can be evoked. Acetylcholine: Chemical transmitter substance released
Nerves and Nerve Impulse Terms Absolute refractory period: Period following stimulation during which no additional action potential can be evoked. Acetylcholine: Chemical transmitter substance released
Classification of Diseases and Disorders and Guidelines Chapters 4-7. Rebecca Herrera, CCS, CCS-P, CPC
 Classification of Diseases and Disorders and Guidelines Chapters 4-7 Rebecca Herrera, CCS, CCS-P, CPC Overview of. Endocrine, Nutritional and Metabolic Diseases Mental and Behavioral Disorders Diseases
Classification of Diseases and Disorders and Guidelines Chapters 4-7 Rebecca Herrera, CCS, CCS-P, CPC Overview of. Endocrine, Nutritional and Metabolic Diseases Mental and Behavioral Disorders Diseases
Update on guidelines on biological treatment of depressive disorder. Dr. Henry CHEUNG Psychiatrist in private practice
 Update on guidelines on biological treatment of depressive disorder Dr. Henry CHEUNG Psychiatrist in private practice 2013 update International Task Force of World Federation of Societies of Biological
Update on guidelines on biological treatment of depressive disorder Dr. Henry CHEUNG Psychiatrist in private practice 2013 update International Task Force of World Federation of Societies of Biological
ELECTROMYOGRAPHY (EMG), NEEDLE, NERVE CONDUCTION STUDIES (NCS) AND QUANTITATIVE SENSORY TESTING (QST)
 AND QUANTITATIVE SENSORY TESTING (QST) Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit plan. This Medical
AND QUANTITATIVE SENSORY TESTING (QST) Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit plan. This Medical
Behavioral and Physical Treatments for Tension-type and Cervicogenic Headache
 Evidence Report: Behavioral and Physical Treatments for Tension-type and Cervicogenic Headache Douglas C. McCrory, MD, MHSc Donald B. Penzien, PhD Vic Hasselblad, PhD Rebecca N. Gray, DPhil Duke University
Evidence Report: Behavioral and Physical Treatments for Tension-type and Cervicogenic Headache Douglas C. McCrory, MD, MHSc Donald B. Penzien, PhD Vic Hasselblad, PhD Rebecca N. Gray, DPhil Duke University
DEPRESSION CARE PROCESS STEP EXPECTATIONS RATIONALE
 1 DEPRESSION CARE PROCESS STEP EXPECTATIONS RATIONALE ASSESSMENT/PROBLEM RECOGNITION 1. Did the staff and physician seek and document risk factors for depression and any history of depression? 2. Did staff
1 DEPRESSION CARE PROCESS STEP EXPECTATIONS RATIONALE ASSESSMENT/PROBLEM RECOGNITION 1. Did the staff and physician seek and document risk factors for depression and any history of depression? 2. Did staff
Depression. Introduction Depression is a common condition that affects millions of people every year.
 Depression Introduction Depression is a common condition that affects millions of people every year. Depression has an impact on most aspects of everyday life. It affects eating and sleeping routines,
Depression Introduction Depression is a common condition that affects millions of people every year. Depression has an impact on most aspects of everyday life. It affects eating and sleeping routines,
BOTOX Treatment. for Chronic Migraine. Information for patients and their families. Botulinum Toxin Type A
 BOTOX Treatment Botulinum Toxin Type A for Chronic Migraine Information for patients and their families. Is Chronic Migraine the same as Migraine? Chronic Migraine is similar to migraine as sufferers experience
BOTOX Treatment Botulinum Toxin Type A for Chronic Migraine Information for patients and their families. Is Chronic Migraine the same as Migraine? Chronic Migraine is similar to migraine as sufferers experience
ADULT HEALTH AND WELLBEING LONG-TERM NEUROLOGICAL CONDITIONS
 ADULT HEALTH AND WELLBEING LONG-TERM NEUROLOGICAL CONDITIONS i. Summary The National Service Framework for long-term neurological conditions categorises neurological conditions as: Sudden-onset conditions
ADULT HEALTH AND WELLBEING LONG-TERM NEUROLOGICAL CONDITIONS i. Summary The National Service Framework for long-term neurological conditions categorises neurological conditions as: Sudden-onset conditions
PAIN MANAGEMENT AT UM/SYLVESTER
 PAIN MANAGEMENT AT UM/SYLVESTER W HAT IS THE PURPOSE OF THIS BROCHURE? We created this brochure for patients receiving care from the University of Miami Sylvester Comprehensive Cancer Center and their
PAIN MANAGEMENT AT UM/SYLVESTER W HAT IS THE PURPOSE OF THIS BROCHURE? We created this brochure for patients receiving care from the University of Miami Sylvester Comprehensive Cancer Center and their
A Rapidly Growing Population with. - Murray Goldstein, DO, MPH Chairman, Cerebral Palsy International Research CPIRF
 Adults with Cerebral Palsy A Rapidly Growing Population with Complex Health Issues - Murray Goldstein, DO, MPH Chairman, Cerebral Palsy International Research Foundation Scientific Advisory Council CPIRF
Adults with Cerebral Palsy A Rapidly Growing Population with Complex Health Issues - Murray Goldstein, DO, MPH Chairman, Cerebral Palsy International Research Foundation Scientific Advisory Council CPIRF
Deep Brain Stimulation for Essential Tremor and Parkinson s Disease (NCD 160.24)
 Policy Number Reimbursement Policy 160.24 Approved By UnitedHealthcare Medicare Reimbursement Policy Committee Current Approval Date 06/10/2015 IMPORTANT NOTE ABOUT THIS REIMBURSEMENT POLICY This policy
Policy Number Reimbursement Policy 160.24 Approved By UnitedHealthcare Medicare Reimbursement Policy Committee Current Approval Date 06/10/2015 IMPORTANT NOTE ABOUT THIS REIMBURSEMENT POLICY This policy
Chapter 15. Autonomic Nervous System (ANS) and Visceral Reflexes. general properties Anatomy. Autonomic effects on target organs
 Chapter 15 Autonomic Nervous System (ANS) and Visceral Reflexes general properties Anatomy Autonomic effects on target organs Central control of autonomic function 15-1 Copyright (c) The McGraw-Hill Companies,
Chapter 15 Autonomic Nervous System (ANS) and Visceral Reflexes general properties Anatomy Autonomic effects on target organs Central control of autonomic function 15-1 Copyright (c) The McGraw-Hill Companies,
Guidelines for Clinical Neurophysiology
 Guidelines for Clinical Neurophysiology These Guidelines have been drawn up along the lines of those of the Department of Clinical Radiology to help referring clinicians make the best use of Clinical Neurophysiology.
Guidelines for Clinical Neurophysiology These Guidelines have been drawn up along the lines of those of the Department of Clinical Radiology to help referring clinicians make the best use of Clinical Neurophysiology.
Diagnostic and Statistical Manual of Mental Disorders (DSM-IV-TR) (APA, 2001) 10
 5. Diagnosis Questions to be answered: 5.1. What are the diagnostic criteria for ADHD in children and adolescents? 5.2. How is ADHD diagnosed in children and adolescents? Who must diagnose it? 5.3. Which
5. Diagnosis Questions to be answered: 5.1. What are the diagnostic criteria for ADHD in children and adolescents? 5.2. How is ADHD diagnosed in children and adolescents? Who must diagnose it? 5.3. Which
Regarding Insurance Reimbursement
 Innovation in Health and Wellness Technology Regarding Insurance Reimbursement BEST-Pro1 BEST-RSI TM Enclosed is prescription form that documents necessity and support for coverage of the Avazzia Biofeedback
Innovation in Health and Wellness Technology Regarding Insurance Reimbursement BEST-Pro1 BEST-RSI TM Enclosed is prescription form that documents necessity and support for coverage of the Avazzia Biofeedback
Table of Contents. Preface...xv. Part I: Introduction to Mental Health Disorders and Depression
 Table of Contents Visit www.healthreferenceseries.com to view A Contents Guide to the Health Reference Series, a listing of more than 16,000 topics and the volumes in which they are covered. Preface...xv
Table of Contents Visit www.healthreferenceseries.com to view A Contents Guide to the Health Reference Series, a listing of more than 16,000 topics and the volumes in which they are covered. Preface...xv
