Ontario Drug Benefit Formulary/Comparative Drug Index
|
|
|
- Ariel Sullivan
- 8 years ago
- Views:
Transcription
1 Ontario Drug Benefit Formulary/Comparative Drug Index Edition 42 Summary of Changes - September 2014 Effective September 25, 2014 Ministry of Health and Long-Term Care
2 Table of Contents New Single Source Products... 3 New Multi-Source Products... 4 New Off-Formulary Interchangeable (OFI) Products... 8 New Nutrition Product... 9 Change to Reason For Use Content Addition of New Reason For Use Code Drug Benefit Price (DBP) Changes OFI Product Price Change Nutrition Product Name Change Discontinued Products Delisted Products
3 New Single Source Products DIN/PIN PRODUCT NAME GENERIC NAME MFR DBP Emend Tri-Pack 125mg & 80mg Cap APREPITANT MEK Reason For Use Code and Clinical Criteria Code 452 In combination with a 5HT3 receptor antagonist and dexamethasone for highly emetogenic chemotherapy (HEC) regimens: Cisplatin-based chemotherapy where a single daily dose is greater than or equal to 70 mg per meter squared Cisplatin and cyclophosphamide combinations where the single daily dose is greater than or equal to 50mg per meter squared Cisplatin (any dose) given for 3 to 5 consecutive days Non-cisplatin based highly emetogenic chemotherapy (such as those containing anthracycline greater than or equal to 60mg per meter squared plus cyclophosphamide) Dosage: Recommend aprepitant 125mg orally on Day 1 of HEC followed by 80mg orally on Days 2 to 3 post-chemotherapy for each cycle. LU Authorization Period: 1 year. Code 453 For patients receiving moderately emetogenic chemotherapy (MEC) regimens AND who have had inadequate symptom control using a 5HT3 antagonist and dexamethasone in a previous cycle. Dosage: Recommend aprepitant 125mg orally on Day 1 of MEC followed by 80mg orally on Days 2 to 3 post-chemotherapy for each cycle. LU Authorization Period: 1 year. 3
4 New Multi-Source Products DIN/PIN BRAND NAME STRENGTH DOSAGE FORM MFR DBP Abbott-Rabeprazole 20mg Tab ABB (Interchangeable with Pariet) DIN/PIN BRAND NAME STRENGTH DOSAGE FORM MFR DBP Act Dutasteride 0.5mg Cap ACV Apo-Dutasteride 0.5mg Cap APX PMS-Dutasteride 0.5mg Cap PMS Sandoz Dutasteride Capsule 0.5mg Cap SDZ Teva-Dutasteride 0.5mg Cap TEV (Interchangeable with Avodart) Reason For Use Code and Clinical Criteria Code 384 For use in combination with an alpha blocker for the treatment of men with symptomatic* Benign Prostatic Hyperplasia. LU Authorization Period: Indefinite. Code 385 For monotherapy, as a second line agent in patients with symptomatic* Benign Prostatic Hyperplasia following treatment failure or intolerance to an alpha blocker. * Symptomatic is defined as having moderate (about half the time) to severe (almost always) symptoms related to the prostate in at least 4 of the following domains: 1. feeling of incomplete emptying of the bladder after voiding 2. needing to urinate again less than 2 hours after previous void 3. stopping and starting urine several times while voiding 4. difficulty postponing urination 5. weak urinary stream 6. pushing or straining to begin voiding 7. the need to get up to void at least 3 times in the night. LU Authorization Period: Indefinite. 4
5 New Multi-Source Products (Cont d...) DIN/PIN BRAND NAME STRENGTH DOSAGE FORM MFR DBP Auro-Galantamine ER 8mg ER Cap AUR Auro-Galantamine ER 16mg ER Cap AUR Auro-Galantamine ER 24mg ER Cap AUR (Interchangeable with Reminyl ER) Reason For Use Code and Clinical Criteria Code 347 Initial Trial: For patients with mild to moderate Alzheimer's Disease (Mini-Mental State Exam [MMSE] 10-26). Patients will be reimbursed for a period of up to 3 months after which continued treatment must be reassessed. Network note: Maximum duration 3 months. LU Authorization Period: 1 year. Code 348 Continuation: Further reimbursement will be made available to those patients whose disease has not progressed/deteriorated while on this drug. Patients must continue to have a MMSE score of LU Authorization Period: 1 year. 5
6 New Multi-Source Products (Cont d...) DIN/PIN BRAND NAME STRENGTH DOSAGE FORM MFR DBP Septa-Losartan 25mg Tab SET Septa-Losartan 50mg Tab SET Septa-Losartan 100mg Tab SET (Interchangeable with Cozaar) DIN/PIN BRAND NAME STRENGTH DOSAGE FORM MFR DBP Telmisartan Tablets 40mg Tab ACH Telmisartan Tablets 80mg Tab ACH (Interchangeable with Micardis) DIN/PIN BRAND NAME STRENGTH DOSAGE MFR DBP FORM Telmisartan and 80mg & 12.5mg Tab ACH Hydrochlorothiazide Tablets Telmisartan and 80mg & 25mg Tab ACH Hydrochlorothiazide Tablets (Interchangeable with Micardis Plus) 6
7 New Multi-Source Products (Cont d...) DIN/PIN BRAND NAME STRENGTH DOSAGE FORM MFR DBP Zoledronic Acid Injection 5mg/100mL Inj Sol-100mL Pk DRR (Interchangeable with Aclasta) Reason For Use Code and Clinical Criteria Code 319 For the treatment of Paget's disease. LU Authorization Period: Indefinite. Code 436 For the treatment of osteoporosis in postmenopausal women who would otherwise be eligible for funding for oral bisphosphonates, but for whom bisphosphonates are contraindicated due to abnormalities of the esophagus (e.g. esophageal stricture or achalasia), AND have at least two of the following:. Age greater than 75 years old. A prior fragility fracture. A bone mineral density (BMD) T-score less than or equal to -2.5 NOTE: Patients receiving Aclasta should not be receiving concomitant bisphosphonate therapy. The recommended dose of Aclasta (zoledronic acid) is a single IV injection of 5mg, once yearly. LU Authorization Period: Indefinite. 7
8 New Off-Formulary Interchangeable (OFI) Products DIN/PIN BRAND NAME STRENGTH DOSAGE FORM MFR UNIT COST Mint-Hydrochlorothiazide 12.5mg Tab MIN (Interchangeable with PMS-Hydrochlorothiazide) DIN/PIN BRAND NAME STRENGTH DOSAGE FORM MFR UNIT COST PMS-Olanzapine ODT 20mg Rapid Dissolve Tab PMS (Interchangeable with Zyprexa Zydis) DIN/PIN BRAND NAME STRENGTH DOSAGE FORM MFR UNIT COST Ran-Memantine 10mg Tab RAN (Interchangeable with Ebixa) DIN/PIN BRAND NAME STRENGTH DOSAGE FORM MFR UNIT COST Teva-Modafinil 100mg Tab TEV (Interchangeable with Alertec) DIN/PIN BRAND NAME STRENGTH DOSAGE FORM MFR UNIT COST Zoledronic Acid for 4mg/5mL Inj Sol-5mL Pk DRR Injection Concentrate (Interchangeable with Zometa Concentrate) 8
9 New Nutrition Product DIN/PIN PRODUCT NAME STRENGTH DOSAGE FORM MFR COST/PKG AMOUNT MOH PAYS AMOUNT PATIENT PAYS KetoCal 4:1 (Unflavoured) 1.5kcal/mL Liq-237mL Tetra Pk NUT
10 Change to Reason For Use Content DIN/PIN BRAND NAME STRENGTH DOSAGE FORM MFR Xarelto 15mg Tab BAH Xarelto 20mg Tab BAH Updated Reason For Use Code 444 For the treatment of deep vein thrombosis (DVT) or pulmonary embolism (PE) for up to six (6) months. LU Authorization Period: 6 months. Notes: The recommended dose of rivaroxaban for patients initiating DVT or PE treatment is 15mg twice daily for 3 weeks, followed by 20mg once daily. ODB Program coverage for rivaroxaban is an alternative to heparin/warfarin for up to 6 months. When used for greater than 6 months, rivaroxaban is more costly than heparin/warfarin. As such, patients with an intended duration of therapy greater than 6 months should be considered for initiation on heparin/warfarin. Since renal impairment can increase bleeding risk, it is important to monitor renal function regularly. Other factors that increase bleeding risks should also be assessed and monitored (see product monograph). 10
11 Addition of New Reason For Use Code DIN/PIN BRAND NAME STRENGTH DOSAGE FORM MFR Granisetron 1mg Tab AAP Ondansetron 4mg/5mL O/L AAP Zofran 4mg/5mL O/L GSK Apo-Ondansetron 4mg Tab APX Co Ondansetron 4mg Tab COB Jamp-Ondansetron 4mg Tab JPC Mar-Ondansetron 4mg Tab MAR Mint-Ondansetron 4mg Tab MIN Mylan-Ondansetron 4mg Tab MYL Ondansetron-Odan 4mg Tab ODN PMS-Ondansetron 4mg Tab PMS Phl-Ondansetron 4mg Tab PHE Ran-Ondansetron 4mg Tab RAN Ratio-Ondansetron 4mg Tab RPH Sandoz Ondansetron 4mg Tab SDZ Septa-Ondansetron 4mg Tab SET Teva-Ondansetron 4mg Tab TEV Zofran 4mg Tab GSK Zym-Ondansetron 4mg Tab ZYN Ondissolve ODF (Film) 4mg Film TAK Zofran ODT (Tablet) 4mg Tab GSK New Reason For Use Code and Clinical Criteria in addition to the existing codes Code 454 For the treatment of emesis in cancer patients receiving moderately emetogenic chemotherapy (MEC) regimens. LU Authorization Period: 1 year. 11
12 Addition of New Reason For Use Code (Cont d ) DIN/PIN BRAND NAME STRENGTH DOSAGE MFR FORM Apo-Ondansetron 8mg Tab APX Co Ondansetron 8mg Tab COB Jamp-Ondansetron 8mg Tab JPC Mar-Ondansetron 8mg Tab MAR Mint-Ondansetron 8mg Tab MIN Mylan-Ondansetron 8mg Tab MYL Ondansetron-Odan 8mg Tab ODN PMS-Ondansetron 8mg Tab PMS Phl-Ondansetron 8mg Tab PHE Ran-Ondansetron 8mg Tab RAN Ratio-Ondansetron 8mg Tab RPH Sandoz Ondansetron 8mg Tab SDZ Septa-Ondansetron 8mg Tab SET Teva-Ondansetron 8mg Tab TEV Zofran 8mg Tab GSK Zym-Ondansetron 8mg Tab ZYN Ondissolve ODF (Film) 8mg Film TAK Zofran ODT (Tablet) 8mg Tab GSK New Reason For Use Code and Clinical Criteria in addition to the existing codes Code 454 For the treatment of emesis in cancer patients receiving moderately emetogenic chemotherapy (MEC) regimens. LU Authorization Period: 1 year. 12
13 Drug Benefit Price (DBP) Changes DIN/PIN BRAND NAME STRENGTH DOSAGE FORM MFR DBP Haloperidol LA 100mg/mL Oily Inj Sol-5mL Pk SDZ Humalog Mix25 Kwikpen 25% & 75% Inj Susp-5x3mL Pk LIL
14 OFI Product Price Change DIN/PIN BRAND NAME STRENGTH DOSAGE FORM MFR UNIT COST Apo-Adefovir 10mg Tab APX
15 Nutrition Product Name Change PIN/PIN CURRENT PRODUCT NAME NEW PRODUCT NAME KetoCal 4:1 KetoCal 4:1 (Vanilla Flavoured) STRENGTH DOSAGE FORM MFR 1.5kcal/mL Liq-237mL Tetra Pk NUT 15
16 Discontinued Products (Some products will remain on Formulary for six months to facilitate depletion of supply) DIN/PIN BRAND NAME STRENGTH DOSAGE FORM MFR Drisdol 8288IU/mL O/L SAV Ratio-IPRA SAL UDV 500mcg/2.5mg/2.5mL Inh Sol-2.5mL Pk RPH Tamofen 10mg Tab SAV Tamofen 20mg Tab SAV Trental 400mg SR Tab SAV 16
17 Delisted Products DIN/PIN BRAND NAME STRENGTH DOSAGE FORM MFR Cytosar 100mg Inj Pd-Vial Pk PFI Apo-Haloperidol 1mg Tab APX Apo-Haloperidol 10mg Tab APX Cafergot 1mg & 100mg Tab NOV * Monopril 10mg Tab BQU * Monopril 20mg Tab BQU Naprosyn 25mg/mL O/L HLR Simvastatin-Odan 5mg Tab ODN Simvastatin-Odan 10mg Tab ODN Simvastatin-Odan 20mg Tab ODN Simvastatin-Odan 40mg Tab ODN Simvastatin-Odan 80mg Tab ODN * Remain in the Formulary as reference product. 17
18 ISSN (PDF) September 2014 Queen s Printer for Ontario, 2014.
Updates to the Alberta Human Services Drug Benefit Supplement
 Updates to the Alberta Human Services Drug Benefit Supplement Effective December 1, 2014 Inquiries should be directed to: Pharmacy Services Alberta Blue Cross 10009 108 Street NW Edmonton AB T5J 3C5 Telephone
Updates to the Alberta Human Services Drug Benefit Supplement Effective December 1, 2014 Inquiries should be directed to: Pharmacy Services Alberta Blue Cross 10009 108 Street NW Edmonton AB T5J 3C5 Telephone
Updates to the Alberta Drug Benefit List. Effective January 1, 2016
 Updates to the Alberta Drug Benefit List Effective January 1, 2016 Inquiries should be directed to: Pharmacy Services Alberta Blue Cross 10009 108 Street NW Edmonton AB T5J 3C5 Telephone Number: (780)
Updates to the Alberta Drug Benefit List Effective January 1, 2016 Inquiries should be directed to: Pharmacy Services Alberta Blue Cross 10009 108 Street NW Edmonton AB T5J 3C5 Telephone Number: (780)
Updates to the Alberta Human Services Drug Benefit Supplement
 Updates to the Alberta Human Services Drug Benefit Supplement Effective January 1, 2016 Inquiries should be directed to: Pharmacy Services Alberta Blue Cross 10009 108 Street NW Edmonton AB T5J 3C5 Telephone
Updates to the Alberta Human Services Drug Benefit Supplement Effective January 1, 2016 Inquiries should be directed to: Pharmacy Services Alberta Blue Cross 10009 108 Street NW Edmonton AB T5J 3C5 Telephone
Indication: Indication: Protaxos is indicated in adults.
 Maklumat tambahan indikasi untuk upload pada laman web Year 2013 Products Approved For Additional Indication (DCA 262 28 Mac 2013) NO PRODUCT (ACTIVE INGREDIENT) 1. 1.1 EVISTA TABLET 60MG [ Raloxifene
Maklumat tambahan indikasi untuk upload pada laman web Year 2013 Products Approved For Additional Indication (DCA 262 28 Mac 2013) NO PRODUCT (ACTIVE INGREDIENT) 1. 1.1 EVISTA TABLET 60MG [ Raloxifene
UPDATE AJ Ontario Drug Benefit Formulary/Comparative Drug Index No. 41 Effective July 27, 2012 SUMMARY OF CHANGES
 UPDATE AJ Ontario Drug Benefit Formulary/Comparative Drug Index No. 41 Effective July 27, 2012 SUMMARY OF CHANGES TABLE OF CONTENTS Page New Single Source Drug(s) 2 New Multi-Source Drug(s) 5 Off Formulary
UPDATE AJ Ontario Drug Benefit Formulary/Comparative Drug Index No. 41 Effective July 27, 2012 SUMMARY OF CHANGES TABLE OF CONTENTS Page New Single Source Drug(s) 2 New Multi-Source Drug(s) 5 Off Formulary
SASKATCHEWAN FORMULARY BULLETIN Update to the 62nd Edition of the Saskatchewan Formulary
 July 1, 2013 Bulletin #141 Drug Plan and Extended Benefits Branch ISSN 1923-0761 SASKATCHEWAN FORMULARY BULLETIN Update to the 62nd Edition of the Saskatchewan Formulary Product DIN Pre-Markup ($) New
July 1, 2013 Bulletin #141 Drug Plan and Extended Benefits Branch ISSN 1923-0761 SASKATCHEWAN FORMULARY BULLETIN Update to the 62nd Edition of the Saskatchewan Formulary Product DIN Pre-Markup ($) New
KING KHALID UNIVERSITY HOSPITAL PHARMACY TEAM
 KING KHALID UNIVERSITY HOSPITAL PHARMACY TEAM MINUTES OF THE MEETING Month: 29 th September Location: Director of Pharmacy office Date/Time : 29-09-2012 (13-11-1433H) 12.30pm to 2.30pm Chairman of the
KING KHALID UNIVERSITY HOSPITAL PHARMACY TEAM MINUTES OF THE MEETING Month: 29 th September Location: Director of Pharmacy office Date/Time : 29-09-2012 (13-11-1433H) 12.30pm to 2.30pm Chairman of the
Updates to the Alberta Drug Benefit List. Effective March 1, 2015
 Updates to the Alberta Drug Benefit List Effective March 1, 2015 Inquiries should be directed to: Pharmacy Services Alberta Blue Cross 10009 108 Street NW Edmonton AB T5J 3C5 Telephone Number: (780) 498-8370
Updates to the Alberta Drug Benefit List Effective March 1, 2015 Inquiries should be directed to: Pharmacy Services Alberta Blue Cross 10009 108 Street NW Edmonton AB T5J 3C5 Telephone Number: (780) 498-8370
NHS FORTH VALLEY RIVAROXABAN AS TREATMENT FOR DEEP VEIN THROMBOSIS AND PULMONARY EMBOLISM IN ADULTS
 NHS FORTH VALLEY RIVAROXABAN AS TREATMENT FOR DEEP VEIN THROMBOSIS AND PULMONARY EMBOLISM IN ADULTS Date of First Issue 01/12/ 2012 Approved 15/11/2012 Current Issue Date 29/10/2014 Review Date 29/10/2016
NHS FORTH VALLEY RIVAROXABAN AS TREATMENT FOR DEEP VEIN THROMBOSIS AND PULMONARY EMBOLISM IN ADULTS Date of First Issue 01/12/ 2012 Approved 15/11/2012 Current Issue Date 29/10/2014 Review Date 29/10/2016
SECTION 3. Criteria for Special Authorization of Select Drug Products. Section 3 Criteria for Special Authorization of Select Drug Products
 SECTION 3 Criteria for Special Authorization of Select Drug Products Section 3 Criteria for Special Authorization of Select Drug Products CRITERIA FOR SPECIAL AUTHORIZATION OF SELECT DRUG PRODUCTS The
SECTION 3 Criteria for Special Authorization of Select Drug Products Section 3 Criteria for Special Authorization of Select Drug Products CRITERIA FOR SPECIAL AUTHORIZATION OF SELECT DRUG PRODUCTS The
Inpatient Anticoagulation Safety. To provide safe and effective anticoagulation therapy through a collaborative approach.
 Inpatient Anticoagulation Safety Purpose: Policy: To provide safe and effective anticoagulation therapy through a collaborative approach. Upon the written order of a physician, Heparin, Low Molecular Weight
Inpatient Anticoagulation Safety Purpose: Policy: To provide safe and effective anticoagulation therapy through a collaborative approach. Upon the written order of a physician, Heparin, Low Molecular Weight
Clinical Study Synopsis
 Clinical Study Synopsis This Clinical Study Synopsis is provided for patients and healthcare professionals to increase the transparency of Bayer's clinical research. This document is not intended to replace
Clinical Study Synopsis This Clinical Study Synopsis is provided for patients and healthcare professionals to increase the transparency of Bayer's clinical research. This document is not intended to replace
Guidelines for Prevention and Treatment of Chemotherapy-Induced Nausea and Vomiting in Adults
 Guidelines for Prevention and Treatment of Chemotherapy-Induced Nausea and Vomiting in Adults Protocol Code Tumour group Physician Contact SCNAUSEA SUPPORTIVE CARE Dr. Paul Hoskins ELIGIBILITY Adults receiving
Guidelines for Prevention and Treatment of Chemotherapy-Induced Nausea and Vomiting in Adults Protocol Code Tumour group Physician Contact SCNAUSEA SUPPORTIVE CARE Dr. Paul Hoskins ELIGIBILITY Adults receiving
Texas Medicaid/CHIP Vendor Drug Program Drug Utilization Criteria For Outpatient Use Guidelines
 About Information on indications for use or diagnosis is assumed to be unavailable. All criteria may be applied retrospectively; prospective application is indicated with an asterisk [*]. The information
About Information on indications for use or diagnosis is assumed to be unavailable. All criteria may be applied retrospectively; prospective application is indicated with an asterisk [*]. The information
To provide an evidenced-based approach to treatment of patients presenting with deep vein thrombosis.
 DEEP VEIN THROMBOSIS: TREATMENT TARGET AUDIENCE: All Canadian health care professionals. OBJECTIVE: To provide an evidenced-based approach to treatment of patients presenting with deep vein thrombosis.
DEEP VEIN THROMBOSIS: TREATMENT TARGET AUDIENCE: All Canadian health care professionals. OBJECTIVE: To provide an evidenced-based approach to treatment of patients presenting with deep vein thrombosis.
NB Drug Plans Formulary Update
 Bulletin #902 March 6, 2015 NB Drug Plans Formulary Update This update to the New Brunswick Drug Plans Formulary is effective March 6, 2015. Included in this bulletin: Regular Benefit Additions Special
Bulletin #902 March 6, 2015 NB Drug Plans Formulary Update This update to the New Brunswick Drug Plans Formulary is effective March 6, 2015. Included in this bulletin: Regular Benefit Additions Special
Objectives. Patient Background. Transitioning a Patient To & From a New Oral Anticoagulant
 Objectives Transitioning a Patient To & From a New Oral Anticoagulant How to switch from warfarin to rivaroxaban Discuss the Medicare donut hole How to switch from rivaroxaban to warfarin Home INR monitoring
Objectives Transitioning a Patient To & From a New Oral Anticoagulant How to switch from warfarin to rivaroxaban Discuss the Medicare donut hole How to switch from rivaroxaban to warfarin Home INR monitoring
CDEC FINAL RECOMMENDATION
 CDEC FINAL RECOMMENDATION RIVAROXABAN (Xarelto Bayer Inc.) New Indication: Pulmonary Embolism Note: The Canadian Drug Expert Committee (CDEC) previously reviewed rivaroxaban for the treatment of deep vein
CDEC FINAL RECOMMENDATION RIVAROXABAN (Xarelto Bayer Inc.) New Indication: Pulmonary Embolism Note: The Canadian Drug Expert Committee (CDEC) previously reviewed rivaroxaban for the treatment of deep vein
Medications for Prevention and Treatment of Osteoporosis
 1 Medications for Prevention and Treatment of Osteoporosis Osteoporosis is a disease where the strength of bones is less than normal, making them more susceptible to fracture, or breaking, than normal
1 Medications for Prevention and Treatment of Osteoporosis Osteoporosis is a disease where the strength of bones is less than normal, making them more susceptible to fracture, or breaking, than normal
CDEC RECORD OF ADVICE
 CDEC RECORD OF ADVICE RIVAROXABAN (Xarelto Bayer Inc.) Indication: Deep Vein Thrombosis Without Symptomatic Pulmonary Embolism This document summarizes the Canadian Drug Expert Committee (CDEC) response
CDEC RECORD OF ADVICE RIVAROXABAN (Xarelto Bayer Inc.) Indication: Deep Vein Thrombosis Without Symptomatic Pulmonary Embolism This document summarizes the Canadian Drug Expert Committee (CDEC) response
SUMMARY OF THE RISK MANAGEMENT PLAN (by medicinal product)
 PART VI SUMMARY OF THE RISK MANAGEMENT PLAN (by medicinal product) Format and content of the summary of the RMP The summary of the RMP part VI contains information based on RMP modules SI, SVIII and RMP
PART VI SUMMARY OF THE RISK MANAGEMENT PLAN (by medicinal product) Format and content of the summary of the RMP The summary of the RMP part VI contains information based on RMP modules SI, SVIII and RMP
Safety Information Card for Xarelto Patients
 Safety Information Card for Xarelto Patients 15mg Simply Protecting More Patients 20mg Simply Protecting More Patients Keep this card with you at all times Present this card to every physician or dentist
Safety Information Card for Xarelto Patients 15mg Simply Protecting More Patients 20mg Simply Protecting More Patients Keep this card with you at all times Present this card to every physician or dentist
What You Need to Know for Better Bone Health
 What You Need to Know for Better Bone Health A quick lesson about bones: Why healthy bones matter The healthier your bones The more active you can be Bone health has a major effect on your quality of life
What You Need to Know for Better Bone Health A quick lesson about bones: Why healthy bones matter The healthier your bones The more active you can be Bone health has a major effect on your quality of life
Lung Pathway Group Nintedanib (Vargatef) in advanced Non-Small Cell Lung Cancer (NSCLC)
 Lung Pathway Group Nintedanib (Vargatef) in advanced Non-Small Cell Lung Cancer (NSCLC) Indication: In combination with docetaxel in locally advanced, metastatic or locally recurrent NSCLC of adenocarcinoma
Lung Pathway Group Nintedanib (Vargatef) in advanced Non-Small Cell Lung Cancer (NSCLC) Indication: In combination with docetaxel in locally advanced, metastatic or locally recurrent NSCLC of adenocarcinoma
Common Drug Review Pharmacoeconomic Review Report
 Common Drug Review Pharmacoeconomic Review Report August 2015 Drug rivaroxaban (Xarelto) Indication Listing request Manufacturer Treatment of venous thromboembolic events (deep vein thrombosis [DVT], pulmonary
Common Drug Review Pharmacoeconomic Review Report August 2015 Drug rivaroxaban (Xarelto) Indication Listing request Manufacturer Treatment of venous thromboembolic events (deep vein thrombosis [DVT], pulmonary
Rivaroxaban: Prescribing Guidance for the treatment of provoked venous thromboembolism (VTE)
 Rivaroxaban: Prescribing Guidance for the treatment of provoked venous thromboembolism (VTE) Amber Drug Level 2 Leeds We have started your patient on rivaroxaban for the treatment of provoked VTE (deep
Rivaroxaban: Prescribing Guidance for the treatment of provoked venous thromboembolism (VTE) Amber Drug Level 2 Leeds We have started your patient on rivaroxaban for the treatment of provoked VTE (deep
Primary Care Management of Male Lower Urinary Tract Symptoms. Matthew B.K. Shaw Consultant Urological Surgeon
 Primary Care Management of Male Lower Urinary Tract Symptoms Matthew B.K. Shaw Consultant Urological Surgeon NICE LUTS Guidelines Lower Urinary Tract Symptoms (LUTS) in men. NICE Clinical Guideline 97
Primary Care Management of Male Lower Urinary Tract Symptoms Matthew B.K. Shaw Consultant Urological Surgeon NICE LUTS Guidelines Lower Urinary Tract Symptoms (LUTS) in men. NICE Clinical Guideline 97
MEDICAL ASSISTANCE HANDBOOK PRIOR AUTHORIZATION OF PHARMACEUTICAL SERVICES. A. Prescriptions That Require Prior Authorization
 MEDICAL ASSISTANCE HBOOK PRI AUTHIZATION OF PHARMACEUTICAL SERVICES I. Requirements for Prior Authorization of Anticoagulants A. Prescriptions That Require Prior Authorization Prescriptions for Anticoagulants
MEDICAL ASSISTANCE HBOOK PRI AUTHIZATION OF PHARMACEUTICAL SERVICES I. Requirements for Prior Authorization of Anticoagulants A. Prescriptions That Require Prior Authorization Prescriptions for Anticoagulants
11. CANCER PAIN AND PALLIATION. Jennifer Reifel, M.D.
 11. CANCER PAIN AND PALLIATION Jennifer Reifel, M.D. While supportive care was not one of the original clinical areas selected for the development of quality indicators, the Oncology and HIV Panel felt
11. CANCER PAIN AND PALLIATION Jennifer Reifel, M.D. While supportive care was not one of the original clinical areas selected for the development of quality indicators, the Oncology and HIV Panel felt
Osteoporosis Medications
 Osteoporosis Medications When does a doctor prescribe osteoporosis medications? Healthcare providers look at several pieces of information before prescribing a bone- preserving or bone- building medication.
Osteoporosis Medications When does a doctor prescribe osteoporosis medications? Healthcare providers look at several pieces of information before prescribing a bone- preserving or bone- building medication.
The importance of adherence and persistence: The advantages of once-daily dosing
 The importance of adherence and persistence: The advantages of once-daily dosing Craig I. Coleman, PharmD Professor, University of Connecticut School of Pharmacy Storrs, CT, USA Conflicts of interest Dr
The importance of adherence and persistence: The advantages of once-daily dosing Craig I. Coleman, PharmD Professor, University of Connecticut School of Pharmacy Storrs, CT, USA Conflicts of interest Dr
HERTFORDSHIRE MEDICINES MANAGEMENT COMMITTEE (HMMC) RIVAROXABAN RECOMMENDED see specific recommendations for licensed indications below
 Name: generic (trade) Rivaroxaban (Xarelto ) HERTFORDSHIRE MEDICINES MANAGEMENT COMMITTEE (HMMC) RIVAROXABAN RECOMMENDED see specific recommendations for licensed indications below What it is Indications
Name: generic (trade) Rivaroxaban (Xarelto ) HERTFORDSHIRE MEDICINES MANAGEMENT COMMITTEE (HMMC) RIVAROXABAN RECOMMENDED see specific recommendations for licensed indications below What it is Indications
SASKATCHEWAN FORMULARY BULLETIN Update to the 62nd Edition of the Saskatchewan Formulary
 November 1, 2014 Bulletin #150 ISSN 1923-0761 SASKATCHEWAN FORMULARY BULLETIN Update to the 62nd Edition of the Saskatchewan Formulary New Exception Drug Status (EDS) Listings Effective November 1, 2014
November 1, 2014 Bulletin #150 ISSN 1923-0761 SASKATCHEWAN FORMULARY BULLETIN Update to the 62nd Edition of the Saskatchewan Formulary New Exception Drug Status (EDS) Listings Effective November 1, 2014
Prescriber Guide. 20mg. 15mg. Simply Protecting More Patients. Simply Protecting More Patients
 Prescriber Guide 20mg Simply Protecting More Patients 15mg Simply Protecting More Patients 1 Dear Doctor, This prescriber guide was produced by Bayer Israel in cooperation with the Ministry of Health as
Prescriber Guide 20mg Simply Protecting More Patients 15mg Simply Protecting More Patients 1 Dear Doctor, This prescriber guide was produced by Bayer Israel in cooperation with the Ministry of Health as
Prevention of stroke and systemic embolism in adult patients with non-valvular atrial fibrillation (AF) with one or more risk factors
 News Release For use outside the US and UK only Bayer Pharma AG 13342 Berlin Germany Tel. +49 30 468-1111 www.bayerpharma.com Bayer s Xarelto Approved in the EU for the Prevention of Stroke in Patients
News Release For use outside the US and UK only Bayer Pharma AG 13342 Berlin Germany Tel. +49 30 468-1111 www.bayerpharma.com Bayer s Xarelto Approved in the EU for the Prevention of Stroke in Patients
Randomized, double-blind, parallel-group, multicenter, doubledummy
 ABOUT RECORD STUDIES FAST FACTS RECORD is a global program of four trials in more than 12,500 patients, comparing Xarelto (rivaroxaban) and enoxaparin in the prevention of venous thromboembolism (VTE)
ABOUT RECORD STUDIES FAST FACTS RECORD is a global program of four trials in more than 12,500 patients, comparing Xarelto (rivaroxaban) and enoxaparin in the prevention of venous thromboembolism (VTE)
National Guidance and New Protocols
 National Guidance and New Protocols Dr Jane Strong Consultant Haematologist DVT clinical lead Acute VTE chair DAWN AC Twentieth User Group Meeting 8 th October 2012 DVT patient pathway Assessment Diagnosis
National Guidance and New Protocols Dr Jane Strong Consultant Haematologist DVT clinical lead Acute VTE chair DAWN AC Twentieth User Group Meeting 8 th October 2012 DVT patient pathway Assessment Diagnosis
Antiplatelet and Antithrombotic Therapy. Dr Curry Grant Stroke Prevention Clinic Quinte Health Care
 Antiplatelet and Antithrombotic Therapy Dr Curry Grant Stroke Prevention Clinic Quinte Health Care Disclosure of Potential for Conflict of Interest Dr. F.C. Grant Atrial Fibrillation FINANCIAL DISCLOSURE:
Antiplatelet and Antithrombotic Therapy Dr Curry Grant Stroke Prevention Clinic Quinte Health Care Disclosure of Potential for Conflict of Interest Dr. F.C. Grant Atrial Fibrillation FINANCIAL DISCLOSURE:
Chemotherapy Induced Nausea & Vomiting
 Chemotherapy Induced Nausea & Vomiting A Nurse s Perspective Michael Flynn MSc, PG Cert, RGN Chemotherapy Nurse Consultant Guy s and St Thomas NHS Foundation Trust Guy s and St Thomas NHS Foundation Trust
Chemotherapy Induced Nausea & Vomiting A Nurse s Perspective Michael Flynn MSc, PG Cert, RGN Chemotherapy Nurse Consultant Guy s and St Thomas NHS Foundation Trust Guy s and St Thomas NHS Foundation Trust
How To Take Xarelto
 A patient's guide Your clinic's contact details are: Name: Contact number: Contents 2 Why have I been prescribed Xarelto? 2 What is Xarelto? 3 How do I take Xarelto? 3 What should I do if I miss a dose
A patient's guide Your clinic's contact details are: Name: Contact number: Contents 2 Why have I been prescribed Xarelto? 2 What is Xarelto? 3 How do I take Xarelto? 3 What should I do if I miss a dose
Guidelines for the Management of. Nausea and Vomiting in Cancer Patients
 Guidelines for the Management of Nausea and Vomiting in Cancer Patients Guidelines for the Management of Nausea and Vomiting in Cancer Patients Management of chemotherapy-induced nausea and vomiting includes
Guidelines for the Management of Nausea and Vomiting in Cancer Patients Guidelines for the Management of Nausea and Vomiting in Cancer Patients Management of chemotherapy-induced nausea and vomiting includes
Ontario Drug Benefit Formulary/Comparative Drug Index
 Ontario Drug Benefit Formulary/Comparative Drug Index Edition 42 Effective August 26, 2015 Ministry of Health and Long-Term Care Table of Contents Part I Introduction... I.1 Part II Preamble... II Part
Ontario Drug Benefit Formulary/Comparative Drug Index Edition 42 Effective August 26, 2015 Ministry of Health and Long-Term Care Table of Contents Part I Introduction... I.1 Part II Preamble... II Part
Investor News. Not intended for U.S. and UK media
 Investor News Not intended for U.S. and UK media Bayer AG Investor Relations 51368 Leverkusen Germany www.investor.bayer.com Bayer s Xarelto (Rivaroxaban) Approved for the Treatment of Pulmonary Embolism
Investor News Not intended for U.S. and UK media Bayer AG Investor Relations 51368 Leverkusen Germany www.investor.bayer.com Bayer s Xarelto (Rivaroxaban) Approved for the Treatment of Pulmonary Embolism
ABOUT XARELTO CLINICAL STUDIES
 ABOUT XARELTO CLINICAL STUDIES FAST FACTS Xarelto (rivaroxaban) is a novel, oral direct Factor Xa inhibitor. On September 30, 2008, the European Commission granted marketing approval for Xarelto for the
ABOUT XARELTO CLINICAL STUDIES FAST FACTS Xarelto (rivaroxaban) is a novel, oral direct Factor Xa inhibitor. On September 30, 2008, the European Commission granted marketing approval for Xarelto for the
Oxford Anticoagulation & Thrombosis Service Deep Vein Thrombosis (DVT) and Pulmonary Embolism (PE)
 Oxford University Hospitals NHS Trust Oxford Anticoagulation & Thrombosis Service Deep Vein Thrombosis (DVT) and Pulmonary Embolism (PE) Information for people with a blood clot (thrombus) What is this
Oxford University Hospitals NHS Trust Oxford Anticoagulation & Thrombosis Service Deep Vein Thrombosis (DVT) and Pulmonary Embolism (PE) Information for people with a blood clot (thrombus) What is this
Clinical Study Synopsis
 Clinical Study Synopsis This Clinical Study Synopsis is provided for patients and healthcare professionals to increase the transparency of Bayer's clinical research. This document is not intended to replace
Clinical Study Synopsis This Clinical Study Synopsis is provided for patients and healthcare professionals to increase the transparency of Bayer's clinical research. This document is not intended to replace
150640_Brochure_B 4/12/07 2:58 PM Page 2. Patient Information. Freedom From an Enlarged Prostate
 150640_Brochure_B 4/12/07 2:58 PM Page 2 Patient Information Freedom From an Enlarged Prostate 150640_Brochure_B 4/12/07 2:58 PM Page 3 GreenLight Laser Therapy 1 150640_Brochure_B 4/12/07 2:58 PM Page
150640_Brochure_B 4/12/07 2:58 PM Page 2 Patient Information Freedom From an Enlarged Prostate 150640_Brochure_B 4/12/07 2:58 PM Page 3 GreenLight Laser Therapy 1 150640_Brochure_B 4/12/07 2:58 PM Page
patient education Fact Sheet PFS003: Hormone Therapy APRIL 2015
 patient education Fact Sheet PFS003: Hormone Therapy APRIL 2015 Hormone Therapy Menopause is the time in a woman s life when she naturally stops having menstrual periods. Menopause marks the end of the
patient education Fact Sheet PFS003: Hormone Therapy APRIL 2015 Hormone Therapy Menopause is the time in a woman s life when she naturally stops having menstrual periods. Menopause marks the end of the
National Guidance and New Protocols
 National Guidance and New Protocols Dr Jane Strong Consultant Haematologist DVT clinical lead Acute VTE chair DAWN AC Twentieth User Group Meeting 8 th October 2012 Autumn dawn Restless geese take flight
National Guidance and New Protocols Dr Jane Strong Consultant Haematologist DVT clinical lead Acute VTE chair DAWN AC Twentieth User Group Meeting 8 th October 2012 Autumn dawn Restless geese take flight
2. Background This indication of rivaroxaban had not previously been considered by the PBAC.
 PUBLIC SUMMARY DOCUMENT Product: Rivaroxaban, tablets, 15mg and 20mg, Xarelto Sponsor: Bayer Australia Ltd Date of PBAC Consideration: March 2013 1. Purpose of Application The application requested the
PUBLIC SUMMARY DOCUMENT Product: Rivaroxaban, tablets, 15mg and 20mg, Xarelto Sponsor: Bayer Australia Ltd Date of PBAC Consideration: March 2013 1. Purpose of Application The application requested the
I will be having surgery and radiation treatment for breast cancer. Do I need drug treatment too?
 What is node-positive breast cancer? Node-positive breast cancer means that cancer cells from the tumour in the breast have been found in the lymph nodes (sometimes called glands ) in the armpit area.
What is node-positive breast cancer? Node-positive breast cancer means that cancer cells from the tumour in the breast have been found in the lymph nodes (sometimes called glands ) in the armpit area.
Pathway for the management of DVT in primary Care
 Pathway for the management of DVT in primary Care Final Version: Approved by NHS DGS CCG: June 2015 This document aims to support practices in DGS CCG in the Management & Treatment of patients with suspected
Pathway for the management of DVT in primary Care Final Version: Approved by NHS DGS CCG: June 2015 This document aims to support practices in DGS CCG in the Management & Treatment of patients with suspected
How To Take A Bone Marrow Transplant
 Drug treatments to protect your bones This information is an extract from the booklet, Bone health. You may find the full booklet helpful. We can send you a copy free see page 5. Contents Bisphosphonates
Drug treatments to protect your bones This information is an extract from the booklet, Bone health. You may find the full booklet helpful. We can send you a copy free see page 5. Contents Bisphosphonates
ORAL ANTICOAGULANTS RIVAROXABAN (XARELTO) FOR PULMONARY EMBOLISM (PE)
 ORAL ANTICOAGULANTS RIVAROXABAN (XARELTO) FOR PULMONARY EMBOLISM (PE) Information Leaflet Your Health. Our Priority. Page 2 of 6 What Are Anticoagulants And What Do They Do? This information leaflet has
ORAL ANTICOAGULANTS RIVAROXABAN (XARELTO) FOR PULMONARY EMBOLISM (PE) Information Leaflet Your Health. Our Priority. Page 2 of 6 What Are Anticoagulants And What Do They Do? This information leaflet has
Acetylcholinesterase Inhibitors and Memantine Clinical Indication: Treatment of Dementia in Alzheimer s Disease (AD)
 SHARED CARE PROTOCOL AND INFORMATION FOR GPS Acetylcholinesterase Inhibitors and Memantine Clinical Indication: Treatment of Dementia in Alzheimer s Disease (AD) Version: 3 Date Approved: June 2011 Review
SHARED CARE PROTOCOL AND INFORMATION FOR GPS Acetylcholinesterase Inhibitors and Memantine Clinical Indication: Treatment of Dementia in Alzheimer s Disease (AD) Version: 3 Date Approved: June 2011 Review
Rivaroxaban for the treatment of deep vein thrombosis and prevention of recurrent deep vein thrombosis and pulmonary embolism
 Rivaroxaban for the treatment of deep vein thrombosis and prevention of recurrent deep vein thrombosis and pulmonary Issued: July 2012 guidance.nice.org.uk/ta NHS Evidence has accredited the process used
Rivaroxaban for the treatment of deep vein thrombosis and prevention of recurrent deep vein thrombosis and pulmonary Issued: July 2012 guidance.nice.org.uk/ta NHS Evidence has accredited the process used
Bayer Initiates Rivaroxaban Phase III Study to Support Dose Selection According to Individual Benefit-Risk Profile in Long- Term VTE Prevention
 Investor News Not intended for U.S. and UK Media Bayer AG Investor Relations 51368 Leverkusen Germany www.investor.bayer.com Long-term prevention of venous blood clots (VTE): Bayer Initiates Rivaroxaban
Investor News Not intended for U.S. and UK Media Bayer AG Investor Relations 51368 Leverkusen Germany www.investor.bayer.com Long-term prevention of venous blood clots (VTE): Bayer Initiates Rivaroxaban
The Role of the Newer Anticoagulants
 The Role of the Newer Anticoagulants WARFARIN = Coumadin DAGIBATRAN = Pradaxa RIVAROXABAN = Xarelto APIXABAN = Eliquis INDICATION DABIGATRAN (Pradaxa) RIVAROXABAN (Xarelto) APIXABAN (Eliquis) Stroke prevention
The Role of the Newer Anticoagulants WARFARIN = Coumadin DAGIBATRAN = Pradaxa RIVAROXABAN = Xarelto APIXABAN = Eliquis INDICATION DABIGATRAN (Pradaxa) RIVAROXABAN (Xarelto) APIXABAN (Eliquis) Stroke prevention
VITAMIN C AND INFECTIOUS DISEASE: A REVIEW OF THE LITERATURE AND THE RESULTS OF A RANDOMIZED, DOUBLE-BLIND, PROSPECTIVE STUDY OVER 8 YEARS
 39 Chapter 3 VITAMIN C AND INFECTIOUS DISEASE: A REVIEW OF THE LITERATURE AND THE RESULTS OF A RANDOMIZED, DOUBLE-BLIND, PROSPECTIVE STUDY OVER 8 YEARS Maxine Briggs TABLE OF CONTENTS I. Review of the
39 Chapter 3 VITAMIN C AND INFECTIOUS DISEASE: A REVIEW OF THE LITERATURE AND THE RESULTS OF A RANDOMIZED, DOUBLE-BLIND, PROSPECTIVE STUDY OVER 8 YEARS Maxine Briggs TABLE OF CONTENTS I. Review of the
DVT/PE Management with Rivaroxaban (Xarelto)
 DVT/PE Management with Rivaroxaban (Xarelto) Rivaroxaban is FDA approved for the acute treatment of DVT and PE and reduction in risk of recurrence of DVT and PE. FDA approved indications: Non valvular
DVT/PE Management with Rivaroxaban (Xarelto) Rivaroxaban is FDA approved for the acute treatment of DVT and PE and reduction in risk of recurrence of DVT and PE. FDA approved indications: Non valvular
ORAL ANTICOAGULANTS - RIVAROXABAN (XARELTO) FOR DEEP VEIN THROMBOSIS (DVT)
 ORAL ANTICOAGULANTS - RIVAROXABAN (XARELTO) FOR DEEP VEIN THROMBOSIS (DVT) Information Leaflet Your Health. Our Priority. Page 2 of 6 What Are Anticoagulants And What Do They Do? This information leaflet
ORAL ANTICOAGULANTS - RIVAROXABAN (XARELTO) FOR DEEP VEIN THROMBOSIS (DVT) Information Leaflet Your Health. Our Priority. Page 2 of 6 What Are Anticoagulants And What Do They Do? This information leaflet
Newer Anticoagulants and Newer Diabetic Drug Classes. Nicole N. Nguyen, PharmD Senior Clinical Pharmacist Health Care Services August 21, 2013
 Newer Anticoagulants and Newer Diabetic Drug Classes Nicole N. Nguyen, PharmD Senior Clinical Pharmacist Health Care Services August 21, 2013 Apixaban Newer Anticoagulants Dabigatran etexilate Rivaroxaban
Newer Anticoagulants and Newer Diabetic Drug Classes Nicole N. Nguyen, PharmD Senior Clinical Pharmacist Health Care Services August 21, 2013 Apixaban Newer Anticoagulants Dabigatran etexilate Rivaroxaban
Drug treatment pathway for Osteoporosis in Postmenopausal Women
 Drug treatment pathway for Osteoporosis in Postmenopausal Women Version 1.0 Ratified by: East Sussex HEMC Date ratified: 26.01.2011 Job title of originator/author Gillian Ells, East Sussex HEMC Pharmacist
Drug treatment pathway for Osteoporosis in Postmenopausal Women Version 1.0 Ratified by: East Sussex HEMC Date ratified: 26.01.2011 Job title of originator/author Gillian Ells, East Sussex HEMC Pharmacist
Bios 6648: Design & conduct of clinical research
 Bios 6648: Design & conduct of clinical research Section 1 - Specifying the study setting and objectives 1. Specifying the study setting and objectives 1.0 Background Where will we end up?: (a) The treatment
Bios 6648: Design & conduct of clinical research Section 1 - Specifying the study setting and objectives 1. Specifying the study setting and objectives 1.0 Background Where will we end up?: (a) The treatment
SAVAYSA (edoxaban) U.S. Opportunity
 SAVAYSA (edoxaban) U.S. Opportunity February 17, 2015 Ken Keller President, U.S. Commercial DSI, Inc. Key Discussion Points Daiichi Sankyo, Inc. (DSI) in the U.S. SAVAYSA (edoxaban) U.S. Indication NOAC
SAVAYSA (edoxaban) U.S. Opportunity February 17, 2015 Ken Keller President, U.S. Commercial DSI, Inc. Key Discussion Points Daiichi Sankyo, Inc. (DSI) in the U.S. SAVAYSA (edoxaban) U.S. Indication NOAC
Rivaroxaban for the treatment of deep vein thrombosis and prevention of recurrent deep vein thrombosis and pulmonary embolism
 Rivaroxaban for the treatment of deep vein thrombosis and prevention of recurrent deep vein thrombosis and pulmonary Issued: July 2012 guidance.nice.org.uk/ta NICE has accredited the process used by the
Rivaroxaban for the treatment of deep vein thrombosis and prevention of recurrent deep vein thrombosis and pulmonary Issued: July 2012 guidance.nice.org.uk/ta NICE has accredited the process used by the
National Pharmaceutical Pricing Authority 3 rd Floor, YMCA Cultural Centre 1 Jai Singh Road New Delhi 110001 File No. 23(01)2014/Div.
 Dated 21 st November 2014 NPPA Invites Comments of Pharmaceutical Industry & Trade, Consumer Organisations, Public Health Experts and other Stakeholders on the Recommendations of Tata Memorial Centre under
Dated 21 st November 2014 NPPA Invites Comments of Pharmaceutical Industry & Trade, Consumer Organisations, Public Health Experts and other Stakeholders on the Recommendations of Tata Memorial Centre under
PROTOCOL FOR PATIENTS WITH ABNORMAL LAB AND X-RAY VALUES
 PROTOCOL FOR PATIENTS WITH ABNORMAL LAB AND X-RAY VALUES Patients newly diagnosed as osteopenic or osteoporotic on a radiology report or patients receiving abnormal lab values on the following lab tests
PROTOCOL FOR PATIENTS WITH ABNORMAL LAB AND X-RAY VALUES Patients newly diagnosed as osteopenic or osteoporotic on a radiology report or patients receiving abnormal lab values on the following lab tests
Clinical Study Synopsis
 Clinical Study Synopsis This Clinical Study Synopsis is provided for patients and healthcare professionals to increase the transparency of Bayer's clinical research. This document is not intended to replace
Clinical Study Synopsis This Clinical Study Synopsis is provided for patients and healthcare professionals to increase the transparency of Bayer's clinical research. This document is not intended to replace
MEDICAL ASSISTANCE HANDBOOK PRIOR AUTHORIZATION OF PHARMACEUTICAL SERVICES. A. Prescriptions That Require Prior Authorization
 MEDICAL ASSISTANCE HBOOK I. Requirements for Prior Authorization of Anticoagulants A. Prescriptions That Require Prior Authorization Prescriptions for Anticoagulants which meet any of the following conditions
MEDICAL ASSISTANCE HBOOK I. Requirements for Prior Authorization of Anticoagulants A. Prescriptions That Require Prior Authorization Prescriptions for Anticoagulants which meet any of the following conditions
Hormone. Replacement. Therapy. Information leaflet. This information is also available on request in other formats by phoning 01387 241053.
 Hormone Replacement Therapy This information is also available on request in other formats by phoning 01387 241053. Information leaflet Produced by Dr H Currie & Sr. K Martin May 2005 Updated Dec. 2013
Hormone Replacement Therapy This information is also available on request in other formats by phoning 01387 241053. Information leaflet Produced by Dr H Currie & Sr. K Martin May 2005 Updated Dec. 2013
Donepezil (Aricept ), Galantamine (Reminyl XL ), Rivastigmine (Exelon ) and Memantine (Ebixa )
 Donepezil (Aricept ), Galantamine (Reminyl XL ), Rivastigmine (Exelon ) and Memantine (Ebixa ) ESCA: For the treatment of Alzheimer s disease. SECONDARY CARE SECTION TO BE COMPLETED BY INITIATING DOCTOR
Donepezil (Aricept ), Galantamine (Reminyl XL ), Rivastigmine (Exelon ) and Memantine (Ebixa ) ESCA: For the treatment of Alzheimer s disease. SECONDARY CARE SECTION TO BE COMPLETED BY INITIATING DOCTOR
Drug name: RIVAROXABAN (Xarelto ) For the treatment of acute venous thromboembolism in adult patients.
 Drug name: RIVAROXABAN (Xarelto ) For the treatment of acute venous thromboembolism in adult patients. DOCUMENT TO BE SCANNED INTO ELECTRONIC RECORDS AS AND FILED IN NOTES Patient Name : Date of Birth:
Drug name: RIVAROXABAN (Xarelto ) For the treatment of acute venous thromboembolism in adult patients. DOCUMENT TO BE SCANNED INTO ELECTRONIC RECORDS AS AND FILED IN NOTES Patient Name : Date of Birth:
Dorset Cardiac Centre
 P a g e 1 Dorset Cardiac Centre Patients with Atrial Fibrillation/Flutter undergoing DC Cardioversion or Ablation procedures- Guidelines for Novel Oral Anti-coagulants (NOACS) licensed for this use February
P a g e 1 Dorset Cardiac Centre Patients with Atrial Fibrillation/Flutter undergoing DC Cardioversion or Ablation procedures- Guidelines for Novel Oral Anti-coagulants (NOACS) licensed for this use February
FDA Approved Oral Anticoagulants
 FDA Approved Oral Anticoagulants Generic (Trade Name) Warfarin (Coumadin, Jantoven ) 1 FDA approved indication Prophylaxis and treatment of venous thromboembolism (VTE) Prophylaxis and treatment of thromboembolic
FDA Approved Oral Anticoagulants Generic (Trade Name) Warfarin (Coumadin, Jantoven ) 1 FDA approved indication Prophylaxis and treatment of venous thromboembolism (VTE) Prophylaxis and treatment of thromboembolic
Chemotherapy Order Assessment and Review
 Chemotherapy Order Assessment and Review Contents Introduction... 2 Step 1: Verify Patient Information... 2 Step 2: Confirm Protocol Matches Clinical Indication and Eligibility for Treatment... 2 Step
Chemotherapy Order Assessment and Review Contents Introduction... 2 Step 1: Verify Patient Information... 2 Step 2: Confirm Protocol Matches Clinical Indication and Eligibility for Treatment... 2 Step
Patient Group Direction Hospital: Bristol Royal Infirmary Department: UHBristol Thrombosis Service University Hospitals Bristol NHS Foundation Trust.
 Patient Group Direction Hospital: Bristol Royal Infirmary Department: UHBristol Thrombosis Service University Hospitals Bristol NHS Foundation Trust. This Patient Group Direction (PGD) has been written
Patient Group Direction Hospital: Bristol Royal Infirmary Department: UHBristol Thrombosis Service University Hospitals Bristol NHS Foundation Trust. This Patient Group Direction (PGD) has been written
rivaroxaban 15 and 20mg film-coated tablets (Xarelto ) SMC No. (755/12) Bayer PLC
 rivaroxaban 15 and 20mg film-coated tablets (Xarelto ) SMC No. (755/12) Bayer PLC 13 January 2012 The Scottish Medicines Consortium (SMC) has completed its assessment of the above product and advises NHS
rivaroxaban 15 and 20mg film-coated tablets (Xarelto ) SMC No. (755/12) Bayer PLC 13 January 2012 The Scottish Medicines Consortium (SMC) has completed its assessment of the above product and advises NHS
Hull & East Riding Prescribing Committee
 Hull & East Riding Prescribing Committee Guideline on Prescribing of Gonadorelin (GnRH) Analogues and Progesterone Receptor Modulators in treatment of Endometriosis or prior to Endometrial Ablation, or
Hull & East Riding Prescribing Committee Guideline on Prescribing of Gonadorelin (GnRH) Analogues and Progesterone Receptor Modulators in treatment of Endometriosis or prior to Endometrial Ablation, or
Kevin Saunders MD CCFP Rivergrove Medical Clinic Wellness Institute @ SOGH April 17 2013
 Kevin Saunders MD CCFP Rivergrove Medical Clinic Wellness Institute @ SOGH April 17 2013 Family physician with Rivergrove Medical Clinic Practice in the north end since 1985 Medical Director of the Wellness
Kevin Saunders MD CCFP Rivergrove Medical Clinic Wellness Institute @ SOGH April 17 2013 Family physician with Rivergrove Medical Clinic Practice in the north end since 1985 Medical Director of the Wellness
The Role of Bisphosphonates in Multiple Myeloma: 2007 Update Clinical Practice Guideline
 The Role of Bisphosphonates in Multiple Myeloma: 2007 Update Clinical Practice Guideline Introduction ASCO convened an Update Committee to review and update the 2002 recommendations for the role of bisphosphonates
The Role of Bisphosphonates in Multiple Myeloma: 2007 Update Clinical Practice Guideline Introduction ASCO convened an Update Committee to review and update the 2002 recommendations for the role of bisphosphonates
Rivaroxaban for the treatment of deep vein thrombosis (DVT) and pulmonary embolism (PE)
 Rivaroxaban for the treatment of deep vein thrombosis (DVT) and pulmonary embolism (PE) This leaflet aims to answer your questions about rivaroxaban that may be prescribed for you when you are diagnosed
Rivaroxaban for the treatment of deep vein thrombosis (DVT) and pulmonary embolism (PE) This leaflet aims to answer your questions about rivaroxaban that may be prescribed for you when you are diagnosed
Notice from the Executive Officer: Supporting Sustainability and Access for the Ontario Drug Benefit Program
 Ontario Public Drug Programs, Ministry of Health and Long-Term Care Notice from the Executive Officer: Supporting Sustainability and Access for the Ontario Drug Benefit Program Responsible management of
Ontario Public Drug Programs, Ministry of Health and Long-Term Care Notice from the Executive Officer: Supporting Sustainability and Access for the Ontario Drug Benefit Program Responsible management of
The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION. 10 March 2010
 The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 10 March 2010 ARIXTRA 1.5 mg/0.3 ml, solution for injection in pre-filled syringe Box of 2 (CIP: 363 500-6) Box of
The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 10 March 2010 ARIXTRA 1.5 mg/0.3 ml, solution for injection in pre-filled syringe Box of 2 (CIP: 363 500-6) Box of
Executive Summary. Motive for the request for advice
 Executive Summary Motive for the request for advice Currently nearly 400,000 people in the Netherlands are being treated with anticoagulants of a type Vitamin K antagonists (VKAs). Although VKAs are very
Executive Summary Motive for the request for advice Currently nearly 400,000 people in the Netherlands are being treated with anticoagulants of a type Vitamin K antagonists (VKAs). Although VKAs are very
Technology appraisal guidance Published: 23 March 2011 nice.org.uk/guidance/ta217
 Donepezil, galantamine, rivastigmine and memantine for the treatment of Alzheimer's disease Technology appraisal guidance Published: 23 March 2011 nice.org.uk/guidance/ta217 NICE 2011. All rights reserved.
Donepezil, galantamine, rivastigmine and memantine for the treatment of Alzheimer's disease Technology appraisal guidance Published: 23 March 2011 nice.org.uk/guidance/ta217 NICE 2011. All rights reserved.
A PATIENT S GUIDE TO DEEP VEIN THROMBOSIS TREATMENT
 A PATIENT S GUIDE TO DEEP VEIN THROMBOSIS TREATMENT This medicine is subject to additional monitoring. This will allow quick identification of new safety information. If you get any side effects, talk
A PATIENT S GUIDE TO DEEP VEIN THROMBOSIS TREATMENT This medicine is subject to additional monitoring. This will allow quick identification of new safety information. If you get any side effects, talk
How To Take Methotrexate By Injection
 How To Take Methotrexate By Injection 1 how long does it take for methotrexate to work for abortion 2 methotrexate 15 mg hair loss 3 methotrexate injection dosage for rheumatoid arthritis 4 order methotrexate
How To Take Methotrexate By Injection 1 how long does it take for methotrexate to work for abortion 2 methotrexate 15 mg hair loss 3 methotrexate injection dosage for rheumatoid arthritis 4 order methotrexate
Rivaroxaban (Xarelto) for preventing blood clots after hip or knee replacement surgery
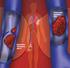 medicineupdate Asking the right questions about new medicines Rivaroxaban (Xarelto) for preventing blood clots after hip or knee replacement surgery This Medicine Update is for people who have been prescribed
medicineupdate Asking the right questions about new medicines Rivaroxaban (Xarelto) for preventing blood clots after hip or knee replacement surgery This Medicine Update is for people who have been prescribed
Shared Care Guideline-Use of Donepezil, Galantamine, Rivastigmine and Memantine in Dementia
 Shared Care Guideline-Use of Donepezil, Galantamine, Rivastigmine and Memantine in Dementia Version: 3.0 Ratified by: Medicines Committee Date ratified: 16 th November 2011 Name of originator/author: James
Shared Care Guideline-Use of Donepezil, Galantamine, Rivastigmine and Memantine in Dementia Version: 3.0 Ratified by: Medicines Committee Date ratified: 16 th November 2011 Name of originator/author: James
MANAGEMENT OF PROSTATE ENLARGEMENT/BPH
 MANAGEMENT OF PROSTATE ENLARGEMENT/BPH J E S S I C A E. P A O N E S S A, M. D. A S S I S T A N T P R O F E S S O R D E P A R T M E N T O F U R O L O G Y S U N Y U P S T A T E M E D I C A L U N I V E R
MANAGEMENT OF PROSTATE ENLARGEMENT/BPH J E S S I C A E. P A O N E S S A, M. D. A S S I S T A N T P R O F E S S O R D E P A R T M E N T O F U R O L O G Y S U N Y U P S T A T E M E D I C A L U N I V E R
Emergency and inpatient treatment of migraine: An American Headache Society
 Emergency and inpatient treatment of migraine: An American Headache Society survey. The objective of this study was to determine the practice preferences of AHS members for acute migraine treatment in
Emergency and inpatient treatment of migraine: An American Headache Society survey. The objective of this study was to determine the practice preferences of AHS members for acute migraine treatment in
Three new/novel oral anticoagulants (NOAC) have been licensed in Ireland since 2008:
 Key Points to consider when prescribing NOACs Introduction Three new/novel oral anticoagulants (NOAC) have been licensed in Ireland since 2008: Dabigatran Etexilate (Pradaxa ) 75mg, 110mg, 150mg. Rivaroxaban
Key Points to consider when prescribing NOACs Introduction Three new/novel oral anticoagulants (NOAC) have been licensed in Ireland since 2008: Dabigatran Etexilate (Pradaxa ) 75mg, 110mg, 150mg. Rivaroxaban
Fast Facts on Osteoporosis
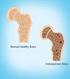 Fast Facts on Osteoporosis Definition Prevalence Osteoporosis, or porous bone, is a disease characterized by low bone mass and structural deterioration of bone tissue, leading to bone fragility and an
Fast Facts on Osteoporosis Definition Prevalence Osteoporosis, or porous bone, is a disease characterized by low bone mass and structural deterioration of bone tissue, leading to bone fragility and an
MEDICATION(S) SUBJECT TO STEP THERAPY
 ACE/ARB COMBO AZOR 5-20 MG TABLET, AZOR 5-40 MG TABLET, BENICAR HCT, MICARDIS HCT, TARKA, TEKTURNA HCT, TELMISARTAN-HYDROCHLOROTHIAZID, TRIBENZOR Claims for formulary step 2 ACE Inhibitor combination products
ACE/ARB COMBO AZOR 5-20 MG TABLET, AZOR 5-40 MG TABLET, BENICAR HCT, MICARDIS HCT, TARKA, TEKTURNA HCT, TELMISARTAN-HYDROCHLOROTHIAZID, TRIBENZOR Claims for formulary step 2 ACE Inhibitor combination products
DOACs. What s in a name? or TSOACs. Blood Clot. Darra Cover, Pharm D. Clot Formation DOACs work here. Direct Oral AntiCoagulant
 DOACs NOACs or TSOACs Generic Name DOACs Brand Name Mechanism of Action Direct Xa Inhibitor Direct Thrombin Inhibitor Dabigatran Pradaxa X Rivaroxaban Xarelto X Darra Cover, Pharm D Apixaban Eliquis X
DOACs NOACs or TSOACs Generic Name DOACs Brand Name Mechanism of Action Direct Xa Inhibitor Direct Thrombin Inhibitor Dabigatran Pradaxa X Rivaroxaban Xarelto X Darra Cover, Pharm D Apixaban Eliquis X
Enoxaparin for long term anticoagulation in patients unsuitable for oral anticoagulants
 Enoxaparin for long term anticoagulation in patients unsuitable for oral anticoagulants Traffic light classification- Amber 2 Information sheet for Primary Care Prescribers Relevant Licensed Indications
Enoxaparin for long term anticoagulation in patients unsuitable for oral anticoagulants Traffic light classification- Amber 2 Information sheet for Primary Care Prescribers Relevant Licensed Indications
NHS FORTH VALLEY Rivaroxaban for Stroke Prevention in Atrial Fibrillation
 NHS FORTH VALLEY Rivaroxaban for Stroke Prevention in Atrial Fibrillation Date of First Issue 06/06/2012 Approved 06/06/2012 Current Issue Date 29/10/2014 Review Date 29/10/2016 Version 1.4 EQIA Yes 01/06/2012
NHS FORTH VALLEY Rivaroxaban for Stroke Prevention in Atrial Fibrillation Date of First Issue 06/06/2012 Approved 06/06/2012 Current Issue Date 29/10/2014 Review Date 29/10/2016 Version 1.4 EQIA Yes 01/06/2012
Rivaroxaban for treating pulmonary embolism and preventing recurrent venous thromboembolism
 NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Final appraisal determination Rivaroxaban for treating pulmonary embolism and preventing recurrent venous thromboembolism This guidance was developed using
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Final appraisal determination Rivaroxaban for treating pulmonary embolism and preventing recurrent venous thromboembolism This guidance was developed using
Anticoagulation at the end of life. Rhona Maclean Rhona.maclean@sth.nhs.uk
 Anticoagulation at the end of life Rhona Maclean Rhona.maclean@sth.nhs.uk Content Anticoagulant Therapies Indications for anticoagulation Venous thromboembolism (VTE) Atrial Fibrillation Mechnical Heart
Anticoagulation at the end of life Rhona Maclean Rhona.maclean@sth.nhs.uk Content Anticoagulant Therapies Indications for anticoagulation Venous thromboembolism (VTE) Atrial Fibrillation Mechnical Heart
