Clinical Policy Title: Injectable bulking agents for fecal incontinence
|
|
|
- Sheila Davidson
- 8 years ago
- Views:
Transcription
1 Clinical Policy Title: Injectable bulking agents for fecal incontinence Clinical policy number: Effective Date: October 1, 2015 Initial Review Date: May 20, 2015 Most Recent Review Date: June 17, 2015 Next Review Date: May 2016 Related policies: None. Policy contains: Solesta injectable gel (Q-Med AB, Sweden for Salix Pharmaceuticals, Inc. Raleigh, NC). Non-animal stabilized hyaluronic acid/dextranomer (NASHA Dx). Fecal incontinence. ABOUT THIS POLICY: AmeriHealth Caritas Pennsylvania has developed clinical policies to assist with making coverage determinations. AmeriHealth Caritas Pennsylvania s clinical policies are based on guidelines from established industry sources, such as the Centers for Medicare & Medicaid Services (CMS), state regulatory agencies, the American Medical Association (AMA), medical specialty professional societies, and peer-reviewed professional literature. These clinical policies along with other sources, such as plan benefits and state and federal laws and regulatory requirements, including any state- or plan-specific definition of medically necessary, and the specific facts of the particular situation are considered by AmeriHealth Caritas Pennsylvania when making coverage determinations. In the event of conflict between this clinical policy and plan benefits and/or state or federal laws and/or regulatory requirements, the plan benefits and/or state and federal laws and/or regulatory requirements shall control. AmeriHealth Caritas Pennsylvania s clinical policies are for informational purposes only and not intended as medical advice or to direct treatment. Physicians and other health care providers are solely responsible for the treatment decisions for their patients. AmeriHealth Caritas Pennsylvania s clinical policies are reflective of evidence-based medicine at the time of review. As medical science evolves, AmeriHealth Caritas Pennsylvania will update its clinical policies as necessary. AmeriHealth Caritas Pennsylvania s clinical policies are not guarantees of payment. Coverage policy AmeriHealth Caritas Pennsylvania considers the use of injectable bulking agents for fecal incontinence to be investigational and, therefore, not medically necessary. Limitations: Other uses of injectable bulking agents may be medically necessary for other gastrourinary indications, such as urinary incontinence. NOTE: The following codes are not included in the Medicaid fee schedule in Pennsylvania: 0377T - Anoscopy with directed submucosal injection of bulking agent for fecal incontinence. L Injectable bulking agent dextranomer/hyaluronic acid copolymer implant, anal canal, 1 ml, includes shipping and necessary supplies. 1
2 Alternative covered services: Biofeedback. Dietary management. Pharmacotherapy. Strengthening exercises. Surgery (e.g., post-anal repair, sphincteroplasty, artificial anal sphincter implantation, total pelvic floor repair or bowel diversion). Sacral nerve stimulation. Background Fecal incontinence (FI), also called anal incontinence or accidental bowel leakage, is loss of control of the bowels resulting in involuntary loss of liquid or solid feces, or flatus, from the rectum. FI is a symptom of an extensive list of underlying causes. The prevalence of FI ranges from 7 percent to 15 percent in community-dwelling men and women and may be higher in institutionalized patients (Bharucha 2015). It is often underreported, as providers seldom screen for FI and patients are often embarrassed and do not volunteer the symptom. FI has a negative impact on activities of daily living and quality of life and is associated with a substantial economic burden, particularly in patients who require surgical therapy (Bharucha 2015). The strongest independent risk factors for FI in the community are bowel disturbances such as diarrhea, the symptom of rectal urgency and burden of chronic illness. Groups at high risk of FI include (Norton 2007, Bharucha 2015): Frail older people. Patients with loose stools or diarrhea from any cause. Symptom of rectal urgency. Women who have recently given birth (especially after third or fourth degree obstetric injury). Patients with neurological or spinal cord disease or injury. Patients with severe cognitive impairment or learning disabilities. Patients with urinary incontinence; pelvic organ or rectal prolapse; or perianal soreness, itching or pain. Patients who have had colonic resection, anal surgery or pelvic radiotherapy. The pathophysiological mechanisms responsible for FI include diarrhea, anal and pelvic floor weakness, reduced rectal compliance, and reduced or increased rectal sensation; many patients have multifaceted anorectal dysfunctions. The type (urge, passive or combined), etiology (anorectal disturbance, bowel symptoms or both) and severity classify FI (Bharucha 2015). The diagnosis of FI encompasses a detailed medical history, physical exam and a range of tests to assess the structure and function of the rectum, anus and pelvic floor muscles (NIDDK 2015). 2
3 Current treatments for FI range from conservative medical therapy aimed at reducing symptoms to surgical interventions intended to correct anal sphincter or pelvic floor abnormalities (NIDDK 2015). The goal of medical therapy is to reduce stool frequency and improve stool consistency. Conservative measures include dietary changes, fiber additives, anti-motility medications, bowel training and pelvic floor exercises with or without biofeedback. Electrical nerve stimulation (neuromodulation) is an emerging treatment option. Surgery may be an option for FI that fails to improve with other treatments or for FI caused by pelvic floor or anal sphincter muscle injuries. Several surgical procedures are available for the treatment of fecal incontinence: pelvic reconstructive surgery to restore normal anatomy, artificial anal sphincter implantation, and bowel diversion such as colonostomy or ileostomy (NIDDK 2015). Minimally invasive procedures such as radiofrequency ablation to the anal sphincter and injectable perianal bulking agents have emerged as potential treatment alternatives for FI (Wald 2014). Injectable, perianal bulking agents: Injectable bulking agents have emerged as a potential treatment for FI following their reported success in treating urinary incontinence. A biocompatible material is injected via the perianal skin or the anal mucosa into the anal submucosa or intersphincteric space to close the anal canal or raise the pressure inside the anal canal to avoid FI. Typically, a colorectal surgeon or gastroenterologist performs the procedure under local anesthesia, and the procedure may be done in an outpatient clinic setting. The simplicity and minimal invasiveness of this procedure make them an attractive treatment alternative for FI. Several different materials have been used to treat urinary incontinence, but to date U.S. Food and Drug Administration (FDA) has approved only one bulking agent for treatment of FI: dextranomer in stabilized sodium hyaluronate, also known as non-animal stabilized hyaluronic acid/dextranomer in stabilized hyaluronic acid or NASHA Dx. NASHA Dx is manufactured by Q- Med AB (Sweden) and distributed in the U.S. by Salix Pharmaceuticals Inc. (Raleigh, NC) under the trade name Solesta. FDA approved NASHA Dx as a class III medical device (product code LNM; agent, bulking, injectable for gastro-urology use) for the treatment of FI in patients 18 years and older who have failed conservative therapy (e.g., diet, fiber therapy, anti-motility medications) (FDA 2011). The physician injects one milliliter (ml) of NASHA Dx through an anoscope into each quadrant of the deep submucosal layer of the proximal anal canal, approximately 5 to 10 millimeters (mm) above the dentate line. According to product labeling, NASHA Dx is contraindicated in patients with the following conditions (FDA 2011): Active inflammatory bowel disease. Immunodeficiency disorders or ongoing immunosuppressive therapy. 3
4 Previous radiation treatment to the pelvic area. Significant mucosal or full-thickness rectal prolapse. Active anorectal conditions, including abscess, fissures, sepsis, bleeding, proctitis or other infections. Anorectal atresia, tumors, stenosis or malformation. Rectocele. Rectal varices. Patients who were pregnant, breast-feeding or without adequate contraception within the first year, or within one year postpartum. Presence of existing implant (other than NASHA Dx) in the anorectal region. Allergy to hyaluronic acid-based products. As a condition of approval, FDA requires manufacturers to provide data regarding numbers of devices sold and distributed with necessary context for FDA to ascertain the frequency and prevalence of adverse events. FDA also mandated two additional studies to assess the long-term safety and durability of NASHA Dx (FDA 2011): 1. A single-arm, multicenter observational study of safety and durability through 36 months. 2. A substudy to show the anatomic stability of NASHA Dx in at least 30 subjects by comparing anatomical positioning via transrectal ultrasonography at time of injection to positioning at six and 36 months. Methods Searches: AmeriHealth Caritas Pennsylvania searched PubMed and the databases of: UK National Health Services Centre for Reviews and Dissemination. Agency for Healthcare Research and Quality s (AHRQ) National Guideline Clearinghouse and other evidence-based practice centers. The Centers for Medicare & Medicaid Services (CMS). We conducted searches on April 13, 2015, using the terms fecal incontinence (MeSH) AND bulking agent OR NASHA OR dextranomer ). Included were: Systematic reviews, which pool results from multiple studies to achieve larger sample sizes and greater precision of effect estimation than in smaller primary studies. Systematic reviews use predetermined transparent methods to minimize bias, effectively treating the review as a scientific endeavor, and are thus rated highest in evidence-grading hierarchies. Guidelines based on systematic reviews. Economic analyses, such as cost-effectiveness, and benefit or utility studies (but not simple cost studies), reporting both costs and outcomes sometimes referred to as efficiency studies which also rank near the top of evidence hierarchies. 4
5 Findings: AmeriHealth Caritas Pennsylvania identified two systematic reviews (Hayes 2014, Maeda 2013), one cost- effectiveness analysis (Bernstein 2014) and two evidence-based guidelines (NICE 2007, Wald 2014) for this policy. The overall quality of the evidence is low given the scarcity of controlled studies and high risk of bias among uncontrolled studies. Methodological limitations of the uncontrolled studies included small sample sizes, lack of blinding and high numbers of dropouts. The majority of studies evaluated NASHA Dx for treating FI. The evidence consists of one randomized controlled trial (RCT) comparing NASHA Dx to sham controls (the PIVOTAL study; ClinicalTrials.gov identifier NCT ), one RCT comparing NASHA Dx to anal sphincter training with biofeedback (ClinicalTrials.gov identifier NCT ) and several small uncontrolled studies using NASHA Dx and other bulking agents. The Pivotal Study is the primary data set that demonstrated the safety and effectiveness of NASHA Dx to FDA, along with supporting evidence of safety and effectiveness from one uncontrolled, multisite open label study (ClinicalTrials.gov identifier NCT ) and one single site, proof of concept study (ClinicalTrials.gov identifier NCT ) (FDA 2011). All but one of the NASHA Dx studies was industry sponsored. The study populations comprised patients with FI who had not responded to conservative treatment (n = 21 to 206 patients). All patients received four injections of one ml of NASHA Dx in each quadrant of the anal submucosa. Patients were generally discharged from the treatment setting after a brief period of observation. After one month, patients without improvement of symptoms were offered a second treatment. Efficacy endpoints included the change in the number of incontinence episodes. A significant treatment response was defined as a 50 percent or greater decrease in FI episode frequency compared with baseline, the number of incontinencefree days and changes in incontinence scores using validated instruments. Patients recorded FI episodes and patterns in diaries when warranted. The duration of follow-up ranged from three months to three years. The evidence is insufficient to support the use of injectable bulking agents for treatment of FI in adults. A limited number of uncontrolled studies of bulking agents other than NASHA Dx are insufficient to permit conclusions regarding their safety and efficacy for FI. The results for NASHA Dx suggest the procedure was well tolerated, with the majority of treatment-related adverse events considered mild or moderate in intensity. These included mild or moderate pain or discomfort in the rectum or anus, minor to moderate bleeding or spotting from the rectum, fever, abdominal pain, diarrhea, and constipation after treatment. NASHA Dx is associated with some modest but statistically significant symptomatic improvements and may be a cost-effective alternative up to three years of follow up in persons who have not responded to conservative treatment. However, improvement in many incontinence scores and general health were not statistically significant, and it is unclear if 5
6 improvement in incontinence scores correlated with practical symptom improvements that mattered to the patients. Results of the sham controlled study suggest a significant placebo effect, and the other controlled study suggested comparable results between NASHA Dx and anal sphincter training with biofeedback. Additional research is needed to determine the clinical value of bulking agents for FI. Given the large placebo effect observed, larger, independent, randomized, sham-controlled studies are needed to further evaluate the efficacy, durability and safety of this treatment. Future studies should compare NASHA Dx with standard therapies such as sacral nerve stimulation and other minimally invasive alternatives. There is also a need to better define the patient selection criteria by examining variables that predict which patients will derive the most clinical benefit from this therapy. To fulfill FDA conditions for continued approval, one observational study is underway to evaluate the long term safety and effectiveness of NASHA Dx through three years in a real world setting (ClinicalTrials.gov identifier NCT ). According to an AHRQ horizon scanning report, tissue bulking agents have potential to improve health outcomes but would not always completely resolve FI (ECRI 2012). Those with muscle disruptions will probably need surgery. The intervention might become widely accepted because it is a noninvasive alternative to surgery that would appeal to patients, but most experts wanted to see additional trial results. Similarly, evidence-based guidelines confirmed the potential of bulking agents for treating FI in patients who are refractory to conservative therapy, but further studies are needed (NICE 2007, Wald 2014). Summary of clinical evidence: Citation Hayes (2014) Content, Methods, Recommendations Key points: Systematic review of two randomized controlled trials (RCTs) and six prospective uncontrolled studies. All but one study was manufacturer-sponsored. Overall quality: low due to low numbers of controlled studies, small study sizes, high numbers of dropouts, lack of blinding in uncontrolled studies. Uncontrolled studies (frequency of FI episodes, episode-free days or incontinence scores baseline vs. post-treatment): NASHA Dx (four injections of 1 ml in each quadrant of the anal submucosa) showed statistically significant improvements in 44% to 57.1% of patients at six months, 64% to 74% at one year, 59% to 63% at two years, and 45% at three years. No significant differences in incontinence scores or general health scores. NASHA Dx vs. sham control at six months after treatment: significant reduction ( 50%) in the number of incontinence episodes (53% vs. 32%); but similar median decrease in number of incontinence episodes between groups; significant benefit for only one quality-of-life (QOL) measure (coping behavior) at six months; no significant differences between groups in other QOL measures. NASHA Dx vs. anal sphincter training with biofeedback: no significant differences in improvements in FI and QOL during two years of follow-up. Strong likelihood of placebo effect noted in studies. Adverse events (AE): mostly mild or moderate and transient with low incidence of serious AEs (~4% to 12%), mainly infections that required antibiotics or surgical drainage, which resolved spontaneously or with treatment. No late complications 6
7 Citation Maeda (2013) Cochrane review Bernstein (2014) Content, Methods, Recommendations observed in studies reporting two- and three-year follow-up of patients. Key points: Systematic review of five RCTs (n = 382 patients) of injectable perianal bulking agents for passive fecal incontinence. Overall quality: low. Four of five trials had an uncertain or high risk of bias. Most trials reported a short-term benefit from injections regardless of the material used. Long term data lacking. Unclear if improvement in incontinence scores matched practical symptom improvements that mattered to the patients. One RCT of NASHA Dx vs. sham (n = 206): NASHA Dx was more effective than sham injection at six months but with more adverse effects. o 48% vs. 69% participants not improved, defined as < 50% reduction in FI episodes (RR 0.70, 95% CI 0.55 to 0.88). o More incontinence-free days (3.1 days vs. 1.7 days, MD 1.40 days, 95% CI 0.33 to 2.47). Key points: Three-year cost-effectiveness analysis. Expected costs in year 2013 for Solesta ($14,962), conservative therapy ($9053) and sacral nerve stimulation (SNS) ($33,201). The incremental costs per quality-adjusted life-year gained were: $37,036 for Solesta vs. conservative therapy; $244,509 for SNS vs. Solesta; and $103,066 for SNS vs. conservative therapy. The model did not include indirect costs. Funded by Salix Pharmaceuticals Inc. Other organizational policies: Organization CMS Policy No NCDs or LCDs for bulking agents for FI. Glossary Anus The opening at the lower end of the alimentary canal through which solid waste is eliminated from the body. Fecal incontinence Lack of bowel control, leading to involuntary loss of bowel contents including gas, liquid stool elements and mucus, and solid feces. Medically Necessary- A service or benefit is Medically Necessary if it is compensable under the MA Program and if it meets any one of the following standards: The service or benefit will, or is reasonably expected to, prevent the onset of an illness, condition or disability. The service or benefit will, or is reasonably expected to, reduce or ameliorate the physical, mental or developmental effects of an illness, condition, injury or disability. The service or benefit will assist the Member to achieve or maintain maximum functional capacity in performing daily activities, taking into account both the functional capacity of 7
8 the Member and those functional capacities that are appropriate for Members of the same age. Pelvic floor In women, the pelvic floor is the muscles, ligaments, connective tissues and nerves that support the bladder, uterus, vagina and rectum and help these pelvic organs function. In men, the pelvic floor includes the muscles, tissues and nerves that support the bladder, rectum and other pelvic organs. Pelvic floor exercises (also called Kegel exercises) A series of exercises designed to strengthen the muscles of the pelvic floor. Rectal (bowel or fecal) urgency A sudden, irresistible need to have a bowel movement. Rectum The comparatively straight, terminal section of the intestine, ending in the anus. Sphincter Ring-like band of muscle fibers that constricts a passage or closes a natural opening; also called a musculus sphincter. Urinary incontinence Lack of bladder control. Related policies AmeriHealth Caritas Pennsylvania Utilization Management program description. References Professional society guidelines/other: National Institute for Health and Clinical Excellence (NICE). Injectable bulking agents for faecal incontinence. NICE interventional procedure guidance 210. February Available at: Accessed April 14, Wald A, Bharucha AE, Cosman BC, Whitehead WE. ACG clinical guideline: management of benign anorectal disorders. Am J Gastroenterol. 2014;109(8): ; (Quiz) Peer-reviewed references: Bernstein MA, Purdy CH, Becker A, Magar R. Three-year cost-effectiveness model for non-animal stabilized hyaluronic acid and dextranomer copolymer compared with sacral nerve stimulation after conservative therapy for the management of fecal incontinence. Clin Ther. 2014;36(6): e893. 8
9 Bharucha AE, Dunivan G, Goode PS, et al. Epidemiology, pathophysiology, and classification of fecal incontinence: state of the science summary for the National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK) workshop. Am J Gastroenterol. 2015;110(1): ECRI Institute. "AHRQ Healthcare Horizon Scanning System Potential High-Impact Interventions: Priority Area 08: Functional Limitations. (Prepared by ECRI Institute under Contract No. HHSA C.)." Rockville, MD: Agency for Healthcare Research and Quality. December Available at: Accessed April 16, Hayes, Inc. Hayes Medical Technology Report. Solesta (NASHA Dx; Q-Med AB) for Treatment of Fecal Incontinence. Lansdale, PA: Hayes, Inc.; October Maeda Y, Laurberg S, Norton C. Perianal injectable bulking agents as treatment for faecal incontinence in adults. Cochrane Database Syst Rev. 2013;2:CD National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK). Fecal Incontinence. Available at: Accessed April 13, Norton C, Thomas L, Hill J. Management of faecal incontinence in adults: summary of NICE guidance. BMJ. 2007;334(7608): U.S. Food and Drug Administration (FDA). Solesta - P Pre-Market Approval Accessed April 13, Clinical trials Searched ClinicalTrials.gov on April 16, 2015 using terms solesta OR NASHA Dx fecal incontinence: A Randomized, Blinded, Multicenter Study to Evaluate NASHA/Dx for the Treatment of Fecal Incontinence. Available at: (Active.) Long Term Safety and Efficacy of Solesta Injectable Bulking Agent for the Treatment of Fecal Incontinence (SoFI). Available at: (Active.) A Randomized, Controlled, Clinical Trial of Biofeedback and Anal Injections as First Treatment of Fecal Incontinence. Available at: (Completed.) Safety and Efficacy of Anorectal Application of Dx-gel for Treatment of Anal Incontinence. Available at: (Completed.) 9
10 Study to Evaluate Solesta for Treatment of Fecal Incontinence. Available at: (Completed.) Pilot Study of NASHA/Dx Gel for Fecal Incontinence. Available at: (Completed.) CMS National Coverage Determinations (NCDs): No NCDs were identified as of the writing of this policy. Local Coverage Determinations (LCDs): No LCDs were identified as of the writing of this policy. Commonly submitted codes Below are the most commonly submitted codes for the service(s)/item(s) subject to this policy. This is not an exhaustive list of codes. Providers are expected to consult the appropriate coding manuals and bill accordingly. CPT Code Description Comment ICD-9 Code Description Comment Full incontinence of feces Fecal smearing ICD-10 Code Description Comment R15.9 Full incontinence of feces R15.1 Fecal smearing HCPCS Level II 0377T L8605 Description Anoscopy with directed submucosal injection of bulking agent for fecal incontinence Injectable bulking agent dextranomer/hyaluronic acid copolymer implant, anal canal, 1 ml, includes shipping and necessary supplies Comment 10
Fecal Incontinence. What is fecal incontinence?
 Scan for mobile link. Fecal Incontinence Fecal incontinence is the inability to control the passage of waste material from the body. It may be associated with constipation or diarrhea and typically occurs
Scan for mobile link. Fecal Incontinence Fecal incontinence is the inability to control the passage of waste material from the body. It may be associated with constipation or diarrhea and typically occurs
Clinical Policy Title: Home uterine activity monitoring
 Clinical Policy Title: Home uterine activity monitoring Clinical Policy Number: 12.01.01 Effective Date: August 19, 2015 Initial Review Date: July 17, 2013 Most Recent Review Date: July 15, 2015 Next Review
Clinical Policy Title: Home uterine activity monitoring Clinical Policy Number: 12.01.01 Effective Date: August 19, 2015 Initial Review Date: July 17, 2013 Most Recent Review Date: July 15, 2015 Next Review
Section: Surgery Last Reviewed Date: December 2013. Policy No: 129 Effective Date: March 1, 2014
 Medical Policy Manual Topic: Transanal Radiofrequency Treatment of Fecal Incontinence Date of Origin: December 2003 Section: Surgery Last Reviewed Date: December 2013 Policy No: 129 Effective Date: March
Medical Policy Manual Topic: Transanal Radiofrequency Treatment of Fecal Incontinence Date of Origin: December 2003 Section: Surgery Last Reviewed Date: December 2013 Policy No: 129 Effective Date: March
Clinical Policy Title: Air Ambulance Transport
 Clinical Policy Title: Air Ambulance Transport Clinical Policy Number: 18.02.02 Effective Date: Oct. 1, 2014 Initial Review Date: April 16, 2014 Most Recent Review Date: May 21, 2014 Next Review Date:
Clinical Policy Title: Air Ambulance Transport Clinical Policy Number: 18.02.02 Effective Date: Oct. 1, 2014 Initial Review Date: April 16, 2014 Most Recent Review Date: May 21, 2014 Next Review Date:
The Work Up of Pelvic Floor Dyssynergia and Fecal Incontinence. Gina R. Sam, MD/MPH Director, Mount Sinai Gastrointestinal Motility Center
 The Work Up of Pelvic Floor Dyssynergia and Fecal Incontinence Gina R. Sam, MD/MPH Director, Mount Sinai Gastrointestinal Motility Center Constipation Overview Constipation Normal Transit Constipation
The Work Up of Pelvic Floor Dyssynergia and Fecal Incontinence Gina R. Sam, MD/MPH Director, Mount Sinai Gastrointestinal Motility Center Constipation Overview Constipation Normal Transit Constipation
Clinical Policy Title: Air Ambulance Transport
 Clinical Policy Title: Air Ambulance Transport Clinical Policy Number: 18.02.02 Effective Date: Sept. 1, 2014 Initial Review Date: April 16, 2014 Most Recent Review Date: May 21, 2014 Next Review Date:
Clinical Policy Title: Air Ambulance Transport Clinical Policy Number: 18.02.02 Effective Date: Sept. 1, 2014 Initial Review Date: April 16, 2014 Most Recent Review Date: May 21, 2014 Next Review Date:
Bowel Control Problems
 Bowel Control Problems WOMENCARE A Healthy Woman is a Powerful Woman (407) 898-1500 Bowel control problems affect at least 1 million people in the United States. Loss of normal control of the bowels is
Bowel Control Problems WOMENCARE A Healthy Woman is a Powerful Woman (407) 898-1500 Bowel control problems affect at least 1 million people in the United States. Loss of normal control of the bowels is
2.01.58. Description. Section: Medicine Effective Date: January 15, 2015 Subsection: Medicine Original Policy Date: June 7, 2012 Subject: Page: 1 of 6
 Page: 1 of 6 Last Review Status/Date: December 2014 Description Radiofrequency (RF) energy has been investigated as a minimally invasive treatment of fecal incontinence, referred to as the Secca procedure.
Page: 1 of 6 Last Review Status/Date: December 2014 Description Radiofrequency (RF) energy has been investigated as a minimally invasive treatment of fecal incontinence, referred to as the Secca procedure.
Transanal Radiofrequency Treatment of Fecal Incontinence
 Transanal Radiofrequency Treatment of Fecal Incontinence Policy Number: 2.01.58 Last Review: 12/2015 Origination: 1/2012 Next Review: 1/2016 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will
Transanal Radiofrequency Treatment of Fecal Incontinence Policy Number: 2.01.58 Last Review: 12/2015 Origination: 1/2012 Next Review: 1/2016 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will
Normal bladder function requires a coordinated effort between the brain, spinal cord, and the bladder.
 .. Urinary Incontinence Urinary incontinence is not an inevitable part of aging, and it is not a disease. The loss of bladder control - called urinary incontinence - affects between 13 and 17 million adult
.. Urinary Incontinence Urinary incontinence is not an inevitable part of aging, and it is not a disease. The loss of bladder control - called urinary incontinence - affects between 13 and 17 million adult
Get the Facts, Be Informed, Make YOUR Best Decision. Pelvic Organ Prolapse
 Pelvic Organ Prolapse ETHICON Women s Health & Urology, a division of ETHICON, INC., a Johnson & Johnson company, is dedicated to providing innovative solutions for common women s health problems and to
Pelvic Organ Prolapse ETHICON Women s Health & Urology, a division of ETHICON, INC., a Johnson & Johnson company, is dedicated to providing innovative solutions for common women s health problems and to
Clinical Policy Title: Leiomyosarcoma and Laparoscopic Power Morcellation
 Clinical Policy Title: Leiomyosarcoma and Laparoscopic Power Morcellation Clinical Policy Number: 12.03.01 Effective Date: January 1, 2015 Initial Review Date: August 20, 2014 Most Recent Review Date:
Clinical Policy Title: Leiomyosarcoma and Laparoscopic Power Morcellation Clinical Policy Number: 12.03.01 Effective Date: January 1, 2015 Initial Review Date: August 20, 2014 Most Recent Review Date:
CHOC CHILDREN SUROLOGY CENTER. Constipation
 Constipation What is constipation? Constipation is a condition in which a person has uncomfortable or infrequent bowel movements. Generally, a person is considered to be constipated when bowel movements
Constipation What is constipation? Constipation is a condition in which a person has uncomfortable or infrequent bowel movements. Generally, a person is considered to be constipated when bowel movements
I can t empty my rectum without pressing my fingers in or near my vagina
 Since the birth of my baby, I can t control my bowel movements Normally bowel movements (stools) are stored in the rectum until the bowel sends a message to the brain that it is full, and the person finds
Since the birth of my baby, I can t control my bowel movements Normally bowel movements (stools) are stored in the rectum until the bowel sends a message to the brain that it is full, and the person finds
Urinary Incontinence FAQ Sheet
 Urinary Incontinence FAQ Sheet Are you reluctant to talk to your doctor about your bladder control problem? Don t be. There is help. Loss of bladder control is called urinary incontinence. It can happen
Urinary Incontinence FAQ Sheet Are you reluctant to talk to your doctor about your bladder control problem? Don t be. There is help. Loss of bladder control is called urinary incontinence. It can happen
Tone Up Your Pelvic Floor. A regular pelvic floor exercise ( Kegel ) routine can prevent symptoms before, during, and after childbirth.
 Will Breastfeeding Affect Incontinence and Pelvic Symptoms? Yes, often for as long as you re nursing. Many breastfeeding women are unaware that a natural drop in estrogen which persists for as long as
Will Breastfeeding Affect Incontinence and Pelvic Symptoms? Yes, often for as long as you re nursing. Many breastfeeding women are unaware that a natural drop in estrogen which persists for as long as
Corporate Medical Policy
 Corporate Medical Policy Injectable Bulking Agents for the Treatment of Urinary and Fecal File Name: Origination: Last CAP Review: Next CAP Review: Last Review: injectable_bulking_agents_for_the_treatment_of_urinary_and_fecal_incontinence
Corporate Medical Policy Injectable Bulking Agents for the Treatment of Urinary and Fecal File Name: Origination: Last CAP Review: Next CAP Review: Last Review: injectable_bulking_agents_for_the_treatment_of_urinary_and_fecal_incontinence
LOSS OF BLADDER CONTROL IS TREATABLE TAKE CONTROL AND RESTORE YOUR LIFESTYLE
 LOSS OF BLADDER CONTROL IS TREATABLE TAKE CONTROL AND RESTORE YOUR LIFESTYLE TALKING ABOUT STRESS INCONTINENCE (SUI) Millions of women suffer from stress incontinence (SUI). This condition results in accidental
LOSS OF BLADDER CONTROL IS TREATABLE TAKE CONTROL AND RESTORE YOUR LIFESTYLE TALKING ABOUT STRESS INCONTINENCE (SUI) Millions of women suffer from stress incontinence (SUI). This condition results in accidental
Female Urinary Disorders and Pelvic Organ Prolapse
 Female Urinary Disorders and Pelvic Organ Prolapse Richard S. Bercik, M.D. Director, Division of Urogynecology & Reconstruction Pelvic Surgery Department of Obstetrics, Gynecology & Reproductive Sciences
Female Urinary Disorders and Pelvic Organ Prolapse Richard S. Bercik, M.D. Director, Division of Urogynecology & Reconstruction Pelvic Surgery Department of Obstetrics, Gynecology & Reproductive Sciences
FAQ About Prostate Cancer Treatment and SpaceOAR System
 FAQ About Prostate Cancer Treatment and SpaceOAR System P. 4 Prostate Cancer Background SpaceOAR Frequently Asked Questions (FAQ) 1. What is prostate cancer? The vast majority of prostate cancers develop
FAQ About Prostate Cancer Treatment and SpaceOAR System P. 4 Prostate Cancer Background SpaceOAR Frequently Asked Questions (FAQ) 1. What is prostate cancer? The vast majority of prostate cancers develop
Sacral Nerve Neuromodulation/Stimulation
 MEDICAL POLICY POLICY RELATED POLICIES POLICY GUIDELINES DESCRIPTION SCOPE BENEFIT APPLICATION RATIONALE REFERENCES CODING APPENDIX HISTORY Sacral Nerve Neuromodulation/Stimulation Number 7.01.69 Effective
MEDICAL POLICY POLICY RELATED POLICIES POLICY GUIDELINES DESCRIPTION SCOPE BENEFIT APPLICATION RATIONALE REFERENCES CODING APPENDIX HISTORY Sacral Nerve Neuromodulation/Stimulation Number 7.01.69 Effective
How To Decide If A Helicopter Is Right For A Patient
 Clinical Policy Title: Air Ambulance Transport Clinical Policy Number: 18.02.02 Effective Date: September 1, 2014 Initial Review Date: April 16, 2014 Most Recent Review Date: May 20, 2015 Next Review Date:
Clinical Policy Title: Air Ambulance Transport Clinical Policy Number: 18.02.02 Effective Date: September 1, 2014 Initial Review Date: April 16, 2014 Most Recent Review Date: May 20, 2015 Next Review Date:
Acticon. Neosphincter. Getting Back to Life. Information Guide treatment for fecal incontinence
 Acticon Neosphincter Information Guide treatment for fecal incontinence Getting Back to Life Introduction If you suffer from loss of bowel control, you are not alone. Did you know that over 2% of the worldwide
Acticon Neosphincter Information Guide treatment for fecal incontinence Getting Back to Life Introduction If you suffer from loss of bowel control, you are not alone. Did you know that over 2% of the worldwide
Prevention and Recognition of Obstetric Fistula Training Package. Module 8: Pre-repair Care and Referral for Women with Obstetric Fistula
 Prevention and Recognition of Obstetric Fistula Training Package Module 8: Pre-repair Care and Referral for Women with Obstetric Fistula Early detection and treatment If a woman has recently survived a
Prevention and Recognition of Obstetric Fistula Training Package Module 8: Pre-repair Care and Referral for Women with Obstetric Fistula Early detection and treatment If a woman has recently survived a
Perianal Abscess and Fistula-in-ano. Background
 Perianal Abscess and Fistula-in-ano Background Anorectal abscesses are some of the more common anorectal conditions encountered, and they are potentially debilitating conditions. The current theory as
Perianal Abscess and Fistula-in-ano Background Anorectal abscesses are some of the more common anorectal conditions encountered, and they are potentially debilitating conditions. The current theory as
URINARY INCONTINENCE
 URINARY INCONTINENCE What is urinary incontinence? Urinary incontinence is the uncontrollable loss of urine. The amount of urine leaked can vary from only a few drops when you cough or sneeze to entirely
URINARY INCONTINENCE What is urinary incontinence? Urinary incontinence is the uncontrollable loss of urine. The amount of urine leaked can vary from only a few drops when you cough or sneeze to entirely
Regain Control of Your Active Life Treatment Options for Incontinence and Pelvic Organ Prolapse
 Regain Control of Your Active Life Treatment Options for Incontinence and Pelvic Organ Prolapse Nearly one quarter of all women in the United States have some sort of pelvic floor disorder such as urinary
Regain Control of Your Active Life Treatment Options for Incontinence and Pelvic Organ Prolapse Nearly one quarter of all women in the United States have some sort of pelvic floor disorder such as urinary
Pelvic Organ Prolapse FAQs
 What is prolapse? Prolapse is a hernia of the vagina that a woman may feel as a bulge or pressure. This is referred to in many different ways, including dropped bladder, dropped uterus, dropped vagina,
What is prolapse? Prolapse is a hernia of the vagina that a woman may feel as a bulge or pressure. This is referred to in many different ways, including dropped bladder, dropped uterus, dropped vagina,
Overactive Bladder (OAB)
 Overactive Bladder (OAB) Overactive bladder is a problem with bladder storage function that causes a sudden urge to urinate. The urge may be difficult to suppress, and overactive bladder can lead to the
Overactive Bladder (OAB) Overactive bladder is a problem with bladder storage function that causes a sudden urge to urinate. The urge may be difficult to suppress, and overactive bladder can lead to the
Urinary Incontinence in Women. Susan Hingle, M.D. Department of Medicine
 Urinary Incontinence in Women Susan Hingle, M.D. Department of Medicine Background Estimated 13 million Americans with urinary incontinence Women are affected twice as frequently as men Only 25% will seek
Urinary Incontinence in Women Susan Hingle, M.D. Department of Medicine Background Estimated 13 million Americans with urinary incontinence Women are affected twice as frequently as men Only 25% will seek
Female Urinary Incontinence
 Female Urinary Incontinence Molly Heublein, MD Assistant Professor Clinical Medicine UCSF Women s Health Primary Care Disclosures I have nothing to disclose. 1 Objectives Review the problem Feel confident
Female Urinary Incontinence Molly Heublein, MD Assistant Professor Clinical Medicine UCSF Women s Health Primary Care Disclosures I have nothing to disclose. 1 Objectives Review the problem Feel confident
Incontinence. What is incontinence?
 Incontinence What is incontinence? Broadly speaking, the medical term incontinence refers to any involuntary release of bodily fluids, but many people associate it strongly with the inability to control
Incontinence What is incontinence? Broadly speaking, the medical term incontinence refers to any involuntary release of bodily fluids, but many people associate it strongly with the inability to control
Direct Current Therapy for Treatment of Hemorrhoids
 Direct Current Therapy for Treatment of Hemorrhoids [For the list of services and procedures that need preauthorization, please refer to www.mcs.com.pr Go to Comunicados a Proveedores, and click Cartas
Direct Current Therapy for Treatment of Hemorrhoids [For the list of services and procedures that need preauthorization, please refer to www.mcs.com.pr Go to Comunicados a Proveedores, and click Cartas
Registered Charity No. 5365
 THE MULTIPLE SCLEROSIS SOCIETY OF IRELAND Dartmouth House, Grand Parade, Dublin 6. Telephone: (01) 269 4599. Fax: (01) 269 3746 MS Helpline: 1850 233 233 E-mail: mscontact@ms-society.ie www.ms-society.ie
THE MULTIPLE SCLEROSIS SOCIETY OF IRELAND Dartmouth House, Grand Parade, Dublin 6. Telephone: (01) 269 4599. Fax: (01) 269 3746 MS Helpline: 1850 233 233 E-mail: mscontact@ms-society.ie www.ms-society.ie
PREPARING FOR YOUR STOMA REVERSAL
 PREPARING FOR YOUR STOMA REVERSAL Information Leaflet Your Health. Our Priority. Page 2 of 6 Introduction- What you need to know As part of your bowel operation you may have had a temporary stoma formed.
PREPARING FOR YOUR STOMA REVERSAL Information Leaflet Your Health. Our Priority. Page 2 of 6 Introduction- What you need to know As part of your bowel operation you may have had a temporary stoma formed.
Radiation Therapy for Prostate Cancer
 Radiation Therapy for Prostate Cancer Introduction Cancer of the prostate is the most common form of cancer that affects men. About 240,000 American men are diagnosed with prostate cancer every year. Your
Radiation Therapy for Prostate Cancer Introduction Cancer of the prostate is the most common form of cancer that affects men. About 240,000 American men are diagnosed with prostate cancer every year. Your
Alan Rosenberg, MD VP Medical Policy, Technology Assessment and Credentialing WellPoint, Inc. 233 S. Wacker Drive, Suite 3900 Chicago, IL 60606
 October 5, 2010 Alan Rosenberg, MD VP Medical Policy, Technology Assessment and Credentialing WellPoint, Inc. 233 S. Wacker Drive, Suite 3900 Chicago, IL 60606 Dear Dr. Rosenberg, The American Gastroenterological
October 5, 2010 Alan Rosenberg, MD VP Medical Policy, Technology Assessment and Credentialing WellPoint, Inc. 233 S. Wacker Drive, Suite 3900 Chicago, IL 60606 Dear Dr. Rosenberg, The American Gastroenterological
in 1996 by the Agency for Health Care Policy and Research, 13 million Americans are
 Seeking Treatment: What? When? Why? Who? How? & Where? In recent membership surveys, the National Association For Continence (NAFC) was distressed to learn that on average people were waiting years before
Seeking Treatment: What? When? Why? Who? How? & Where? In recent membership surveys, the National Association For Continence (NAFC) was distressed to learn that on average people were waiting years before
Corporate Medical Policy
 Corporate Medical Policy Pelvic Floor Stimulation as a Treatment of Urinary and Fecal File Name: Origination: Last CAP Review: Next CAP Review: Last Review: pelvic_floor_stimulation_as_a_treatment_of_urinary_and_fecal_incontinence
Corporate Medical Policy Pelvic Floor Stimulation as a Treatment of Urinary and Fecal File Name: Origination: Last CAP Review: Next CAP Review: Last Review: pelvic_floor_stimulation_as_a_treatment_of_urinary_and_fecal_incontinence
Urinary Incontinence. Anatomy and Terminology Overview. Moeen Abu-Sitta, MD, FACOG, FACS
 Urinary Incontinence Anatomy and Terminology Overview Moeen Abu-Sitta, MD, FACOG, FACS Purpose Locate and describe the anatomy of the Female Urinary System Define terminology related to Incontinence Describe
Urinary Incontinence Anatomy and Terminology Overview Moeen Abu-Sitta, MD, FACOG, FACS Purpose Locate and describe the anatomy of the Female Urinary System Define terminology related to Incontinence Describe
2/21/2016. Prolapse Surgery after Transvaginal Mesh: The Evolving Landscape. Disclosures. Objectives. No Relevant Disclosures
 Prolapse Surgery after Transvaginal Mesh: The Evolving Landscape David R. Ellington, MD, FACOG Assistant Professor Division of Urogynecology and Pelvic Reconstructive Surgery Disclosures No Relevant Disclosures
Prolapse Surgery after Transvaginal Mesh: The Evolving Landscape David R. Ellington, MD, FACOG Assistant Professor Division of Urogynecology and Pelvic Reconstructive Surgery Disclosures No Relevant Disclosures
NHS. Surgical repair of vaginal wall prolapse using mesh. National Institute for Health and Clinical Excellence. 1 Guidance.
 Issue date: June 2008 NHS National Institute for Health and Clinical Excellence Surgical repair of vaginal wall prolapse using mesh 1 Guidance 1.1 The evidence suggests that surgical repair of vaginal
Issue date: June 2008 NHS National Institute for Health and Clinical Excellence Surgical repair of vaginal wall prolapse using mesh 1 Guidance 1.1 The evidence suggests that surgical repair of vaginal
Laparoscopic Colectomy. What do I need to know about my laparoscopic colorectal surgery?
 Laparoscopic Colectomy What do I need to know about my laparoscopic colorectal surgery? Traditionally, colon & rectal surgery requires a large, abdominal and/or pelvic incision, which often requires a
Laparoscopic Colectomy What do I need to know about my laparoscopic colorectal surgery? Traditionally, colon & rectal surgery requires a large, abdominal and/or pelvic incision, which often requires a
Spine Care Centre (SCC) protocols for Multiple Sclerosis Update 1 August 2015
 Spine Care Centre (SCC) protocols for Multiple Sclerosis Update 1 August 2015 Introduction Multiple sclerosis (MS) affects nerves in the brain and spinal cord, causing a wide range of symptoms including
Spine Care Centre (SCC) protocols for Multiple Sclerosis Update 1 August 2015 Introduction Multiple sclerosis (MS) affects nerves in the brain and spinal cord, causing a wide range of symptoms including
GLOSSARY of research terms
 GLOSSARY of research terms JLA Urinary Incontinence Working Partnership Workshop to set research priorities in Urinary Incontinence November 2008 Terms in bold are commonly used clinical/research terms.
GLOSSARY of research terms JLA Urinary Incontinence Working Partnership Workshop to set research priorities in Urinary Incontinence November 2008 Terms in bold are commonly used clinical/research terms.
Spine University s Guide to Cauda Equina Syndrome
 Spine University s Guide to Cauda Equina Syndrome 2 Introduction Your spine is a very complicated part of your body. It s made up of the bones (vertebrae) that keep it aligned, nerves that channel down
Spine University s Guide to Cauda Equina Syndrome 2 Introduction Your spine is a very complicated part of your body. It s made up of the bones (vertebrae) that keep it aligned, nerves that channel down
OVERACTIVE BLADDER DIAGNOSIS AND TREATMENT OF OVERACTIVE BLADDER IN ADULTS:
 2014 OVERACTIVE BLADDER DIAGNOSIS AND TREATMENT OF OVERACTIVE BLADDER IN ADULTS: AUA/SUFU Guideline (2012); Amended (2014) For Primary Care Providers OVERACTIVE BLADDER Diagnosis and Treatment of Overactive
2014 OVERACTIVE BLADDER DIAGNOSIS AND TREATMENT OF OVERACTIVE BLADDER IN ADULTS: AUA/SUFU Guideline (2012); Amended (2014) For Primary Care Providers OVERACTIVE BLADDER Diagnosis and Treatment of Overactive
Urinary Incontinence (Involuntary Loss of Urine) A Patient Guide
 Urinary Incontinence (Involuntary Loss of Urine) A Patient Guide Urinary Incontinence (Urine Loss) This booklet is intended to give you some facts on urinary incontinence - what it is, and is not, and
Urinary Incontinence (Involuntary Loss of Urine) A Patient Guide Urinary Incontinence (Urine Loss) This booklet is intended to give you some facts on urinary incontinence - what it is, and is not, and
Patient. Frequently Asked Questions. Transvaginal Surgical Mesh for Pelvic Organ Prolapse
 Patient Frequently Asked Questions Transvaginal Surgical Mesh for Pelvic Organ Prolapse Frequently Asked Questions WHAT IS PELVIC ORGAN PROLAPSE AND HOW IS IT TREATED? Q: What is pelvic organ prolapse
Patient Frequently Asked Questions Transvaginal Surgical Mesh for Pelvic Organ Prolapse Frequently Asked Questions WHAT IS PELVIC ORGAN PROLAPSE AND HOW IS IT TREATED? Q: What is pelvic organ prolapse
SOGC Recommendations for Urinary Incontinence
 The quality of evidence is rated, and recommendations are made using the criteria described by the Canadian Task Force on Preventive Health Care. Clinical Practice Guidelines: The Evaluation of Stress
The quality of evidence is rated, and recommendations are made using the criteria described by the Canadian Task Force on Preventive Health Care. Clinical Practice Guidelines: The Evaluation of Stress
Night frequency None 1 2 3 4 5 6 7 8 9 10. Not enough warning before needing to urinate. none mild moderate severe
 {Patient Label} SYMPTOMS SURVEY: FREQUENCY: How many times do you urinate during the day and get up from sleep to urinate at night? Day frequency 3 4 5 6 7 8 9 10 11 12 13 14 15 More Night frequency None
{Patient Label} SYMPTOMS SURVEY: FREQUENCY: How many times do you urinate during the day and get up from sleep to urinate at night? Day frequency 3 4 5 6 7 8 9 10 11 12 13 14 15 More Night frequency None
TRANSVAGINAL MESH IN PELVIC ORGAN PROLAPSE REPAIR.
 TRANSVAGINAL MESH IN PELVIC ORGAN PROLAPSE REPAIR. Spanish full text SUMMARY Introduction: Pelvic organ prolapse (POP) is characterised by the descent or herniation of the uterus, vaginal vault, bladder
TRANSVAGINAL MESH IN PELVIC ORGAN PROLAPSE REPAIR. Spanish full text SUMMARY Introduction: Pelvic organ prolapse (POP) is characterised by the descent or herniation of the uterus, vaginal vault, bladder
How To Determine If A Fall Prevention Program Is Effective
 Clinical Policy Title: Medical alert devices and other interventions for vulnerable peoples safety at home Clinical Policy Number: 17.01.02 Effective Date: April 1, 2015 Initial Review Date: January 21,
Clinical Policy Title: Medical alert devices and other interventions for vulnerable peoples safety at home Clinical Policy Number: 17.01.02 Effective Date: April 1, 2015 Initial Review Date: January 21,
Refer to Coaptite Injectable Implant Instructions for Use provided with product for complete instructions for use.
 Questions for my Doctor Refer to Coaptite Injectable Implant Instructions for Use provided with product for complete instructions for use. INDICATIONS: Coaptite Injectable Implant is indicated for soft
Questions for my Doctor Refer to Coaptite Injectable Implant Instructions for Use provided with product for complete instructions for use. INDICATIONS: Coaptite Injectable Implant is indicated for soft
Protocol. Injectable Bulking Agents for the Treatment of Urinary and Fecal Incontinence
 Injectable Bulking Agents for the Treatment of Urinary and Fecal (70119) Medical Benefit Effective Date: 07/01/15 Next Review Date: 05/16 Preauthorization No Review Dates: 01/08, 11/08, 09/09, 09/10, 09/11,
Injectable Bulking Agents for the Treatment of Urinary and Fecal (70119) Medical Benefit Effective Date: 07/01/15 Next Review Date: 05/16 Preauthorization No Review Dates: 01/08, 11/08, 09/09, 09/10, 09/11,
6/3/2011. High Prevalence and Incidence. Low back pain is 5 th most common reason for all physician office visits in the U.S.
 High Prevalence and Incidence Prevalence 85% of Americans will experience low back pain at some time in their life. Incidence 5% annual Timothy C. Shen, M.D. Physical Medicine and Rehabilitation Sub-specialty
High Prevalence and Incidence Prevalence 85% of Americans will experience low back pain at some time in their life. Incidence 5% annual Timothy C. Shen, M.D. Physical Medicine and Rehabilitation Sub-specialty
Bladder and Bowel Control
 Bladder and Bowel Control Dr Sue Woodward Lecturer, Florence Nightingale School of Nursing and Midwifery 2 Why do we need to understand anatomy? Normal physiology Normal adult bladder capacity = 450-500mls
Bladder and Bowel Control Dr Sue Woodward Lecturer, Florence Nightingale School of Nursing and Midwifery 2 Why do we need to understand anatomy? Normal physiology Normal adult bladder capacity = 450-500mls
FEMALE INCONTINENCE REVIEW
 200 S. Wenona Suite 298 Steven L. Jensen, M.D. 5400 Mackinaw, Suite 4302 Bay City, MI 48706 Frank H. Kim, M.D. Saginaw, MI 48604 Telephone (989) 895-2634 Adult & Pediatric Urologists (989) 791-4020 Fax
200 S. Wenona Suite 298 Steven L. Jensen, M.D. 5400 Mackinaw, Suite 4302 Bay City, MI 48706 Frank H. Kim, M.D. Saginaw, MI 48604 Telephone (989) 895-2634 Adult & Pediatric Urologists (989) 791-4020 Fax
Cerebral Palsy. 1995-2014, The Patient Education Institute, Inc. www.x-plain.com nr200105 Last reviewed: 06/17/2014 1
 Cerebral Palsy Introduction Cerebral palsy, or CP, can cause serious neurological symptoms in children. Thousands of children are diagnosed with cerebral palsy every year. This reference summary explains
Cerebral Palsy Introduction Cerebral palsy, or CP, can cause serious neurological symptoms in children. Thousands of children are diagnosed with cerebral palsy every year. This reference summary explains
surg urin Surgery: Urinary System 1
 Surgery: Urinary System 1 This section contains information to assist providers in billing for surgical procedures related to the urinary system. Extracorporeal Shock Wave Lithotripsy Medi-Cal covers Extracorporeal
Surgery: Urinary System 1 This section contains information to assist providers in billing for surgical procedures related to the urinary system. Extracorporeal Shock Wave Lithotripsy Medi-Cal covers Extracorporeal
URINARY INCONTINENCE IN WOMEN
 URINARY INCONTINENCE IN WOMEN Definition Urinary incontinence (UI) is defined as involuntary loss of urine that is a social or hygienic problem (International Continence Society, 1973) Magnitude of the
URINARY INCONTINENCE IN WOMEN Definition Urinary incontinence (UI) is defined as involuntary loss of urine that is a social or hygienic problem (International Continence Society, 1973) Magnitude of the
Recovery After Stroke: Bladder & Bowel Function
 Recovery After Stroke: Bladder & Bowel Function Problems with bladder and bowel function are common but distressing for stroke survivors. Going to the bathroom after suffering a stroke may be complicated
Recovery After Stroke: Bladder & Bowel Function Problems with bladder and bowel function are common but distressing for stroke survivors. Going to the bathroom after suffering a stroke may be complicated
Perianal injectable bulking agents as treatment for faecal incontinence in adults (Review)
 Perianal injectable bulking agents as treatment for faecal incontinence in adults (Review) Maeda Y, Laurberg S, Norton C This is a reprint of a Cochrane review, prepared and maintained by The Cochrane
Perianal injectable bulking agents as treatment for faecal incontinence in adults (Review) Maeda Y, Laurberg S, Norton C This is a reprint of a Cochrane review, prepared and maintained by The Cochrane
Bard: Continence Therapy. Stress Urinary Incontinence. Regaining Control. Restoring Your Lifestyle.
 Bard: Continence Therapy Stress Urinary Incontinence Regaining Control. Restoring Your Lifestyle. Stress Urinary Incontinence Becoming knowledgeable about urinary incontinence Uterus Normal Pelvic Anatomy
Bard: Continence Therapy Stress Urinary Incontinence Regaining Control. Restoring Your Lifestyle. Stress Urinary Incontinence Becoming knowledgeable about urinary incontinence Uterus Normal Pelvic Anatomy
Urinary Incontinence. Types
 Urinary Incontinence Leakage of urine is called urinary incontinence. It is a common problem in women. Some women occasionally leak small amounts of urine. At other times, leakage of urine is frequent
Urinary Incontinence Leakage of urine is called urinary incontinence. It is a common problem in women. Some women occasionally leak small amounts of urine. At other times, leakage of urine is frequent
Cerebral Palsy. In order to function, the brain needs a continuous supply of oxygen.
 Cerebral Palsy Introduction Cerebral palsy, or CP, can cause serious neurological symptoms in children. Up to 5000 children in the United States are diagnosed with cerebral palsy every year. This reference
Cerebral Palsy Introduction Cerebral palsy, or CP, can cause serious neurological symptoms in children. Up to 5000 children in the United States are diagnosed with cerebral palsy every year. This reference
URINARY INCONTINENCE CASE PRESENTATION #1. Urinary Incontinence - History 2014/10/07. Structure of the Female Lower Urinary Tract
 Bladder pressure 2014/10/07 Structure of the Female Lower Urinary Tract Ureter URINARY INCONTINENCE Clinical Clerkship Lecture Series Outer peritoneal coat Detrusor smooth muscle Mucosa Trigone Proximal
Bladder pressure 2014/10/07 Structure of the Female Lower Urinary Tract Ureter URINARY INCONTINENCE Clinical Clerkship Lecture Series Outer peritoneal coat Detrusor smooth muscle Mucosa Trigone Proximal
Urinary Incontinence Dr. Leffler
 Urinary Incontinence Dr. Leffler The involuntary loss of urine at socially unacceptable times occurs in both women and men, but more commonly in women. It has multiple, far-reaching effects on daily activities,
Urinary Incontinence Dr. Leffler The involuntary loss of urine at socially unacceptable times occurs in both women and men, but more commonly in women. It has multiple, far-reaching effects on daily activities,
Fecal incontinence (Encopresis) It is the fecal incontinence condition observed in children with chronic constipation over 1-2 years.
 WHAT IS DYSFUNCTIONAL URINATION (URINATION FUNCTION DISORDER)? It stands for the urination phase disorders, which appear due to wrongly acquired urination habits during the toilet training of some neurologically
WHAT IS DYSFUNCTIONAL URINATION (URINATION FUNCTION DISORDER)? It stands for the urination phase disorders, which appear due to wrongly acquired urination habits during the toilet training of some neurologically
Effects of Pregnancy & Delivery on Pelvic Floor
 Effects of Pregnancy & Delivery on Pelvic Floor 吳 銘 斌 M.D., Ph.D. 財 團 法 人 奇 美 醫 院 婦 產 部 婦 女 泌 尿 暨 骨 盆 醫 學 科 ; 台 北 醫 學 大 學 醫 學 院 婦 產 學 科 ; 古 都 府 城 台 南 Introduction Pelvic floor disorders (PFDs) include
Effects of Pregnancy & Delivery on Pelvic Floor 吳 銘 斌 M.D., Ph.D. 財 團 法 人 奇 美 醫 院 婦 產 部 婦 女 泌 尿 暨 骨 盆 醫 學 科 ; 台 北 醫 學 大 學 醫 學 院 婦 產 學 科 ; 古 都 府 城 台 南 Introduction Pelvic floor disorders (PFDs) include
Urinary Incontinence
 Urinary Incontinence Q: What is urinary Urinary (YOOR-in-air-ee) incontinence (in-kahn-tih-nens) is when urine leaks out before you can get to a bathroom. If you have urinary incontinence, you re not alone.
Urinary Incontinence Q: What is urinary Urinary (YOOR-in-air-ee) incontinence (in-kahn-tih-nens) is when urine leaks out before you can get to a bathroom. If you have urinary incontinence, you re not alone.
1 in 3 women experience Stress Urinary Incontinence.
 A PATIENT S GUIDE 1 in 3 women experience Stress Urinary Incontinence. It s time to talk about SUI Get the facts. This Patient s Guide is intended as a public resource on the issue of Stress Urinary Incontinence
A PATIENT S GUIDE 1 in 3 women experience Stress Urinary Incontinence. It s time to talk about SUI Get the facts. This Patient s Guide is intended as a public resource on the issue of Stress Urinary Incontinence
Treatment of Fistula in Ano. Johanna Basa M.D. SUNY Downstate Medical Center August 2, 2012
 Treatment of Fistula in Ano Johanna Basa M.D. SUNY Downstate Medical Center August 2, 2012 Case Presentation HPI:54 yr old male with PMH of HTN, presented to clinic with complaints of 3rd perianal abscess
Treatment of Fistula in Ano Johanna Basa M.D. SUNY Downstate Medical Center August 2, 2012 Case Presentation HPI:54 yr old male with PMH of HTN, presented to clinic with complaints of 3rd perianal abscess
PSA Screening for Prostate Cancer Information for Care Providers
 All men should know they are having a PSA test and be informed of the implications prior to testing. This booklet was created to help primary care providers offer men information about the risks and benefits
All men should know they are having a PSA test and be informed of the implications prior to testing. This booklet was created to help primary care providers offer men information about the risks and benefits
NHS Greater Glasgow and Clyde Yorkhill Hospital CONSTIPATION IN CHILDREN
 NHS Greater Glasgow and Clyde Yorkhill Hospital CONSTIPATION IN CHILDREN Constipation in Children Version: 1 Page 1 of 8 Background Constipation is a common complaint in infants and children. The aetiology
NHS Greater Glasgow and Clyde Yorkhill Hospital CONSTIPATION IN CHILDREN Constipation in Children Version: 1 Page 1 of 8 Background Constipation is a common complaint in infants and children. The aetiology
Clinical Policy Title: Medical alert devices and other interventions for vulnerable peoples safety at home
 Clinical Policy Title: Medical alert devices and other interventions for vulnerable peoples safety at home Clinical Policy Number: 17.01.02 Effective Date: April 1, 2015 Initial Review Date: January 21,
Clinical Policy Title: Medical alert devices and other interventions for vulnerable peoples safety at home Clinical Policy Number: 17.01.02 Effective Date: April 1, 2015 Initial Review Date: January 21,
Bladder Health Promotion
 Bladder Health Promotion Community Awareness Presentation Content contributions provided by the Society of Urologic Nurses (SUNA) National Association for Continence (NAFC) Simon Foundation for Continence
Bladder Health Promotion Community Awareness Presentation Content contributions provided by the Society of Urologic Nurses (SUNA) National Association for Continence (NAFC) Simon Foundation for Continence
Sample Treatment Protocol
 Sample Treatment Protocol 1 Adults with acute episode of LBP Definition: Acute episode Back pain lasting
Sample Treatment Protocol 1 Adults with acute episode of LBP Definition: Acute episode Back pain lasting
Case Based Urology Learning Program
 Case Based Urology Learning Program Resident s Corner: UROLOGY Case Number 21 CBULP 2011 068 Case Based Urology Learning Program Editor: Associate Editors: Manager: Case Contributors: Steven C. Campbell,
Case Based Urology Learning Program Resident s Corner: UROLOGY Case Number 21 CBULP 2011 068 Case Based Urology Learning Program Editor: Associate Editors: Manager: Case Contributors: Steven C. Campbell,
1 ST JAMAICAN PAEDIATRIC NEPHROLOGY CONFERENCE
 in association with 1 ST JAMAICAN PAEDIATRIC NEPHROLOGY CONFERENCE Jamaica Conference Centre Kingston, Jamaica October 4 th 2014 VOIDING DISORDERS IN CHILDREN Dr. Colin Abel Paediatric Urologist Bustamante
in association with 1 ST JAMAICAN PAEDIATRIC NEPHROLOGY CONFERENCE Jamaica Conference Centre Kingston, Jamaica October 4 th 2014 VOIDING DISORDERS IN CHILDREN Dr. Colin Abel Paediatric Urologist Bustamante
9/24/2015. Incontinence and Prolapse 2015 Primary Care Update CME Symposium. Objectives. Mark Memo, DO, FACS NEO Urology September 25-27
 Incontinence and Prolapse 2015 Primary Care Update CME Symposium Mark Memo, DO, FACS NEO Urology September 25-27 Objectives Incontinence Pelvic Organ Prolapse Vaginal Mesh Litigation 1 Urinary Incontinence
Incontinence and Prolapse 2015 Primary Care Update CME Symposium Mark Memo, DO, FACS NEO Urology September 25-27 Objectives Incontinence Pelvic Organ Prolapse Vaginal Mesh Litigation 1 Urinary Incontinence
Bladder and Pelvic Health
 A Woman s Guide to Bladder and Pelvic Health Symptoms Tests Conditions Treatments 937-277-8988 1-888-808-1016 Dayton Center Englewood Center Huber Heights Center Trotwood Satellite Table of Contents About
A Woman s Guide to Bladder and Pelvic Health Symptoms Tests Conditions Treatments 937-277-8988 1-888-808-1016 Dayton Center Englewood Center Huber Heights Center Trotwood Satellite Table of Contents About
Update on Incontinence in Women: Resources for the Health Professional Satellite Program - February 14, 2002 CME/CEU Sign-up Sheet
 Update on Incontinence in Women: Resources for the Health Professional Satellite Program - February 14, 2002 CME/CEU Sign-up Sheet Information will be used for CME/CEU application only. Please carefully
Update on Incontinence in Women: Resources for the Health Professional Satellite Program - February 14, 2002 CME/CEU Sign-up Sheet Information will be used for CME/CEU application only. Please carefully
Epi procolon The Blood Test for Colorectal Cancer Screening
 Epi procolon The Blood Test for Colorectal Cancer Screening Epi procolon is an approved blood test for colorectal cancer screening. The US Preventive Services Task Force, the American Cancer Society and
Epi procolon The Blood Test for Colorectal Cancer Screening Epi procolon is an approved blood test for colorectal cancer screening. The US Preventive Services Task Force, the American Cancer Society and
Bladder Health Promotion
 Bladder Health Promotion Community Awareness Presentation endorsed by the Society of Urologic Nurses (SUNA) National Association for Continence( NAFC) Simon Foundation for Continence This presentation
Bladder Health Promotion Community Awareness Presentation endorsed by the Society of Urologic Nurses (SUNA) National Association for Continence( NAFC) Simon Foundation for Continence This presentation
Millions of Americans suffer from abdominal pain, bloating, constipation and diarrhea. Now new treatments can relieve your pain and discomfort.
 3888-IBS Consumer Bro 5/8/03 10:38 AM Page 1 TAKE THE IBS TEST Do you have recurrent abdominal pain or discomfort? YES NO UNDERSTANDING IRRITABLE BOWEL SYNDROME A Consumer Education Brochure Do you often
3888-IBS Consumer Bro 5/8/03 10:38 AM Page 1 TAKE THE IBS TEST Do you have recurrent abdominal pain or discomfort? YES NO UNDERSTANDING IRRITABLE BOWEL SYNDROME A Consumer Education Brochure Do you often
Behavioral and Physical Treatments for Tension-type and Cervicogenic Headache
 Evidence Report: Behavioral and Physical Treatments for Tension-type and Cervicogenic Headache Douglas C. McCrory, MD, MHSc Donald B. Penzien, PhD Vic Hasselblad, PhD Rebecca N. Gray, DPhil Duke University
Evidence Report: Behavioral and Physical Treatments for Tension-type and Cervicogenic Headache Douglas C. McCrory, MD, MHSc Donald B. Penzien, PhD Vic Hasselblad, PhD Rebecca N. Gray, DPhil Duke University
Colocutaneous Fistula. Disclosures
 Colocutaneous Fistula Madhulika G. Varma MD Associate Professor Chief, Colorectal Surgery University of California, San Francisco Honoraria Applied Medical Covidien Disclosures 1 Colocutaneous Fistula
Colocutaneous Fistula Madhulika G. Varma MD Associate Professor Chief, Colorectal Surgery University of California, San Francisco Honoraria Applied Medical Covidien Disclosures 1 Colocutaneous Fistula
Is it time for a new drug development paradigm?
 Is it time for a new drug development paradigm? Robert McDonough, M.D. Senior Director, Clinical Policy Research and Development 1 The Aetna Way Our Cause To make quality health care more affordable and
Is it time for a new drug development paradigm? Robert McDonough, M.D. Senior Director, Clinical Policy Research and Development 1 The Aetna Way Our Cause To make quality health care more affordable and
MEDICAL COVERAGE POLICY SERVICE: Urinary Incontinence Treatments
 Important note Even though this policy may indicate that a particular service or supply may be considered covered, this conclusion is not based upon the terms of your particular benefit plan. Each benefit
Important note Even though this policy may indicate that a particular service or supply may be considered covered, this conclusion is not based upon the terms of your particular benefit plan. Each benefit
Women s Continence and Pelvic Health Center
 Women s Continence and Pelvic Health Center Committed to Caring 580-590 Court Street Keene, New Hampshire 03431 (603) 354-5454 Ext. 6643 URINARY INCONTINENCE QUESTIONNAIRE The purpose of this questionnaire
Women s Continence and Pelvic Health Center Committed to Caring 580-590 Court Street Keene, New Hampshire 03431 (603) 354-5454 Ext. 6643 URINARY INCONTINENCE QUESTIONNAIRE The purpose of this questionnaire
Overview of Urinary Incontinence in the Long Term Care Setting
 Overview of Urinary Incontinence in the Long Term Care Setting Management Strategies for the Nursing Assistant Ann M. Spenard RN, C, MSN Courtney Lyder ND, GNP Learning Objectives Describe common types
Overview of Urinary Incontinence in the Long Term Care Setting Management Strategies for the Nursing Assistant Ann M. Spenard RN, C, MSN Courtney Lyder ND, GNP Learning Objectives Describe common types
Urinary Incontinence. Causes of Incontinence. What s Happening?
 National Institute on Aging AgePage Urinary Incontinence Sarah loves to spend time with her friends talking about her grandchildren and going to exercise classes with neighbors. But she s started to have
National Institute on Aging AgePage Urinary Incontinence Sarah loves to spend time with her friends talking about her grandchildren and going to exercise classes with neighbors. But she s started to have
Consumer summary Minimally invasive techniques for the relief of stress urinary incontinence
 ASERNIP S Australian Safety and Efficacy Register of New Interventional Procedures Surgical Consumer summary Minimally invasive techniques for the relief of stress urinary incontinence (Adapted from the
ASERNIP S Australian Safety and Efficacy Register of New Interventional Procedures Surgical Consumer summary Minimally invasive techniques for the relief of stress urinary incontinence (Adapted from the
Appendicitis National Digestive Diseases Information Clearinghouse
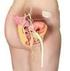
 Appendicitis National Digestive Diseases Information Clearinghouse National Institute of Diabetes and Digestive and Kidney Diseases NATIONAL INSTITUTES OF HEALTH The appendix is a small, tube-like structure
Appendicitis National Digestive Diseases Information Clearinghouse National Institute of Diabetes and Digestive and Kidney Diseases NATIONAL INSTITUTES OF HEALTH The appendix is a small, tube-like structure
A Physical Therapist s Perspective
 You Can Do Something About INCONTINENCE A Physical Therapist s Perspective American Physical Therapy Association 1 You Can Do Something About Urinary Incontinence Incontinence, involuntary loss of bladder
You Can Do Something About INCONTINENCE A Physical Therapist s Perspective American Physical Therapy Association 1 You Can Do Something About Urinary Incontinence Incontinence, involuntary loss of bladder
Pyelonephritis: Kidney Infection
 Pyelonephritis: Kidney Infection National Kidney and Urologic Diseases Information Clearinghouse U.S. Department of Health and Human Services NATIONAL INSTITUTES OF HEALTH What is pyelonephritis? Pyelonephritis
Pyelonephritis: Kidney Infection National Kidney and Urologic Diseases Information Clearinghouse U.S. Department of Health and Human Services NATIONAL INSTITUTES OF HEALTH What is pyelonephritis? Pyelonephritis
Anorectal Abscess and Fistula
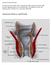 Anorectal Abscess/Fistula by: Robert K Cleary MD, John C Eggenberger MD, Amalia J Stefanou, MD location: Michigan Heart & Vascular Institute, 5325 Elliott Dr, Suite 104 mailing address: PO Box 974, Ann
Anorectal Abscess/Fistula by: Robert K Cleary MD, John C Eggenberger MD, Amalia J Stefanou, MD location: Michigan Heart & Vascular Institute, 5325 Elliott Dr, Suite 104 mailing address: PO Box 974, Ann
Prolapse Repair Systems. a guide To correcting PELVIC ORGAN PROLAPSE
 Anterior PROLAPSE & Posterior REPAIR SYSTEM Prolapse Repair Systems a guide To correcting PELVIC ORGAN PROLAPSE Restore Your Body Pelvic organ prolapse occurs when pelvic structures, like the bladder or
Anterior PROLAPSE & Posterior REPAIR SYSTEM Prolapse Repair Systems a guide To correcting PELVIC ORGAN PROLAPSE Restore Your Body Pelvic organ prolapse occurs when pelvic structures, like the bladder or
Bladder and Bowel Assessment Ann Yates Director of Continence Services. 18/07/2008 Cardiff and Vale NHS Trust
 Bladder and Bowel Assessment Ann Yates Director of Continence Services Types of continence problems Bladder Stress incontinence Urgency and urge Incontinence Mixed incontinence Obstructive incontinence
Bladder and Bowel Assessment Ann Yates Director of Continence Services Types of continence problems Bladder Stress incontinence Urgency and urge Incontinence Mixed incontinence Obstructive incontinence
