Late Stage, Novel Antibiotics
|
|
|
- Georgia Bradford
- 10 years ago
- Views:
Transcription
1 Late Stage, Novel Antibiotics November, /18/2015 1
2 Third-party information included herein has been obtained from sources believed to be reliable, but the accuracy or completeness of such information is not guaranteed by, and should not be construed as a representation by, Paratek Pharmaceuticals, Inc. ( Paratek ). The information contained in this presentation is accurate only as of the date hereof. Paratek and the Paratek logo are trademarks and service marks of Paratek. All other trademarks, service marks, trade names, logos and brand names identified in this presentation are the property of their respective owners. Forward-Looking Statements / Risk Factors This presentation contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act of These statements include, but are not limited to, statements about our strategy, future operations, future financial position, future revenue, clinical development plans and timing, projected costs, prospects, plans, objectives of management, potential use and effectiveness of our product candidates, expected market growth, the market opportunity for and the market acceptance of our product candidates, and the strength of, and protection offered by, our intellectual property position. Examples of such statements include, but are not limited to, statements relating to the potential clinical risks and efficacy of, and market opportunities for, our product candidates, including Omadacycline and Sarecycline, the timing of clinical development of, and regulatory approval for, our product candidates, and the nature and timing of our collaboration agreements with respect to our product candidates. The words anticipate, estimate, expect, potential, believe, will and similar terms and phrases are used to identify forward-looking statements. These statements are based on current information and belief and are not guarantees of future performance. Our ability to predict results, financial or otherwise, or the actual effect of future plans or strategies, is inherently uncertain and actual results may differ from those predicted depending on a variety of factors. Our operations involve risks and uncertainties, many of which are outside our control, and any one of which, or a combination of which, could materially affect our results of operations or whether the forward-looking statements ultimately prove to be correct. We undertake no obligation to publicly update forward-looking statements, whether as a result of new information, future events or otherwise. Among the risks and uncertainties that could cause actual results to differ materially from those indicated by such forwardlooking statements include: delays in clinical trials or unexpected results; the failure of collaborators to perform obligations under our collaboration agreements; our failure to obtain regulatory approval for our product candidates; delays in undertaking or completing clinical trials; our products not gaining the anticipated acceptance in the marketplace or acceptance being delayed; our products not receiving reimbursement from healthcare payors; the effects of competition; our inability to protect our intellectual property and proprietary technology through patents and other means; and the risks described in the Risk Factors section of our Quarterly Report Form 10-Q for the quarter ended June 30, 2015, and our other periodic reports filed with the SEC. 11/18/2015 2
3 Paratek Investment Highlights Developing novel tetracycline antibiotics to address unmet medical needs Omadacycline : Oral/IV antibiotic addressing bacterial resistance Enrolling two pivotal trials ABSSSI and CABP Developing for UTI Potential additional indications - Sinusitis Worldwide commercial rights retained Sarecycline : Antimicrobial and anti-inflammatory tetracycline for treating acne Partnered with Allergan in U.S. Ex-U.S. commercial rights retained Proven management team Strong IP position 11/18/2015 3
4 Proven Management Team Commercialized Major Antibiotics/Built Leading Companies Michael F. Bigham Chairman & CEO Doug Pagan Chief Financial Officer Evan Tzanis VP, Clinical Development Evan Loh, MD President & CMO Led Tygacil Development Adam Woodrow Chief Commercial Officer Led Tygacil Commercialization Steve Villano, MD VP, Clinical and Medical Affairs Randy Brenner SVP, Regulatory & Quality Susan Perkins VP, Intellectual Property William Haskel SVP, General Counsel & Corporate Secretary Sean M. Johnston, PhD VP, Manufacturing Yulii Bogatyrenko SVP, Business Development Jeanne Jew VP, Business Development 11/18/ S. Ken Tanaka, PhD VP, Research and Development Developed clarithromycin, temafloxacin
5 Developing Novel Tetracycline Antibiotics Addressing Unmet Medical Needs Research Preclinical Phase 1 Phase 2 Phase 3 Commercial Rights ABSSSI (Oral & IV) QIDP Status SPA Omadacycline (1) CABP (Oral & IV) QIDP Status UTI (Oral & IV) QIDP Stauts (2) SPA TM Acute Sinusitis (Oral) Sarecycline (3) (WC 3035) Acne Safety/Efficacy X2 (Oral) Acne Long Term Safety Follow up (Oral) (1) Paratek owns Worldwide Commercial Rights (2) UTI program in development + QIDP for cuti (3) Paratek owns Ex-U.S. Commercial Rights 11/18/2015 5
6 Critical Need for New Antibiotics Bacterial resistance renders generic products obsolete over time Bacterial resistance costs society billions $USD > $20 billion USD/year in excess health care costs ~ $35 billion USD societal costs in 2000 > 8 million additional patient days (1) (2) REPORT TO THE PRESIDENT ON COMBATING ANTIBIOTIC RESISTANCE Executive Office of the President President s Council of Advisors on Science and Technology September 2014 (1) Roberts RR, Hota B, Ahmad I, Scott RD 2nd, Foster SD, Abbasi F, Schabowski S, Kampe LM, Ciavarella GG, Supino M, Naples J, Cordell R, Levy SB, Weinstein RA. Clin Infect Dis Oct 15;49(8): doi: / /18/2015 6
7 11/18/ Scarcity of New Antibiotics in Development
8 Legislative Initiatives to Drive Antibiotic Development GAIN ACT (1) ADAPT ACT (2) DISARM ACT (3) Five years additional data exclusivity & priority review Passed Accelerated development pathway New Reimbursement framework to enable premium pricing (1) Provisions signed into law on July 9, 2012 as part of the Food and Drug Administration Safety and Innovation Act (FDASIA) (2) S (113 th ): ADAPT Act, Introduced Jul 10, 2014 (3) H.R DISARM Act of 2015, Introduced Jan 22, /18/2015 8
9 Developing Novel Tetracycline Antibiotics that Overcome Bacterial Resistance Efflux Pump Ribosomal Protection 11/18/2015 9
10 Aminomethylcyclines A New Generation Tetracycline Antibiotic 7-Position Modification: Overcomes Efflux Pump R1 R2 N R3 OH O H H O H O H N O OH NH 2 O 9-Position Modification: Overcomes Ribosomal Protection 11/18/
11 Omadacycline Overcoming Bacterial Resistance The Next Levofloxacin? Well Tolerated Once-Daily IV & Oral Potentially optimizes patient compliance No Drug-Drug Interactions Anticipated Reduces potential safety concerns Potentially Replaces Quinolones MRSA in ABSSSI PRSP/MRSA in CABP ESBL+ E. coli in UTI 11/18/
12 Omadacycline Vancomycin (Vancocin) Linezolid (Zyvox) Levofloxacin (Levaquin) Ceftriaxone (Rocephin) Amox-Clav (Augmentin) Azithromycin (Zithromax) Omadacycline Potent Against Key Resistant ABSSSI Pathogens (1) In-Vitro MIC90 (µg/ml) Organism (# Isolates) MRSA (111) >8 >64 >8 >8 MSSA (52) >8 S.pyogenes (104) >4 (1) CMI 2007 report to Paratek 11/18/
13 Omadacycline: Demonstrated Clinical Efficacy in Complicated Skin, Skin Structure Infections in both Phase 2 and a Truncated Phase 3 (1) (2) Clinical Success Rate (3) Phase 2 Population Omadacycline Linezolid Intent-to-Treat (ITT) 88.3% (98/111) 75.9% (82/108) Clinically Evaluable (CE) 98.0% (98/100) 93.2% (82/88) Phase 3 Population (2) Clinical Success Rate (3) Omadacycline Linezolid Intent-to-Treat (ITT) 85.3% (58/68) 88.9% (64/72) Clinically Evaluable (CE) 96.7% (58/60) 95.5% (64/67) (1) Data from PRTK s Phase 2 and truncated Phase 3 csssi trials (2) An Intent-to-Treat, or ITT, population refers to all enrolled subjects, as defined in the protocol, who received at least one dose of study drug. A Clinically Evaluable, or CE, population refers to all ITT subjects who had a qualifying infection, as defined in the protocol. (3) Clinical success refers to resolution of the infection such that no additional antibiotics were needed in the ITT or CE populations, assessed by the clinical investigator 10 to 17 days after the last dose of study drug. 11/18/
14 Omadacycline Levofloxacin (Levaquin) Azithromycin (Zithromax) Ceftriaxone (Rocephin) Amox-Clav (Augmentin) Vancomycin (Vancocin) Omadacycline Potent Against Key Resistant CABP Pathogens (1) In-Vitro MIC90 (µg/ml) Organism (# Isolates) MRSA (111) 0.25 >8 >8 >64 >8 1 PRSP (51) > H. influenzae (105) Legionella (25) (2) 0.5 Not Active Not Active Not Active Not Active (1) CMI 2007 report to Paratek (2) Indicates data is from moxifloxacin; J. Dubois et al /18/
15 Omadacycline SPA Approved Phase 3 Trial Designs (1) ABSSSI 650 patients Omadacycline IV Linezolid IV Omadacycline Oral Linezolid Oral d1 d2-3 Early Response Up to d14 End of Treatment d16-20 Post Treatment Evaluation, Test of Cure (2) CABP 750 patients Omadacycline IV Moxifloxacin IV Omadacycline Oral Moxifloxacin Oral (1) FDA approved strategy (2) TOC endpoint = Primary endpoint confirmed through EMA scientific advice 11/18/ d1 d3-5 Early Response Up to d14 End of Treatment d16-20 Post Treatment Evaluation, Test of Cure (2)
16 Omadacycline Addressing Resistant E. coli in an Oral Form 97.1% of surveyed physicians believe that their patients with resistant E. coli could benefit from a new antibiotic (1) Omadacycline: UTI Profile Activity against most prevalent UTI pathogen E.coli (2) >40% Renal clearance Once-daily oral dose; plus an IV formulation Safety and tolerability profile: anticipated to match community-based needs Clinical development plans being finalized (1) Medacorp survey 1Q 2013 (2) CMI 2007 report to Paratek 11/18/
17 Omadacycline Amox-Clav (Augmentin) Ceftriaxone (Rocephin) Linezolid (Zyvox) Levofloxacin (Levaquin) Omadacycline Potent Against Key Resistant UTI Pathogens (1) In-Vitro MIC90 (µg/ml) Organism (# Isolates) E. coli ESBL+ (102) Not Active >16 E. faecium, VS (56) 0.12 >8 >64 4 >8 E. faecium, VRE* (100) 0.12 >8 >64 2 >8 E. faecalis, VS (107) >64 2 >8 E. faecalis, VNS* (47) >64 2 >8 (1) CMI 2007 report to Paratek,; *VNS=Vancomycin MIC 16 µg/ml; VRE=Vancomycin MIC 32 µg/ml 11/18/
18 Omadacycline Levofloxacin (Levaquin) Azithromycin (Zithromax) Amox-Clav (Augmentin) Omadacycline Potent Against Key Resistant Sinusitis Pathogens (1) In-Vitro MIC90 (µg/ml) Organism (# Isolates) S. pneumonia (104) >4 8 PRSP (51) >4 8 H. influenzae (105) M. catarrhalis (105) /18/ (1) CMI 2007 report to Paratek
19 Omadacycline Favorable Safety and Tolerability Profile ~700 individuals treated to-date No known metabolites No CYP interactions identified No DDI effects anticipated No anticipated monitoring No herg channel effects: TQTc (1) study completed No effects on heart rate (HR) in patients Modest transient vagolytic HR effect in healthy volunteers (1) Thorough QTc study 11/18/
20 Omadacycline Commercial-Scale Formulations and Process Established Both Oral tablet and IV manufactured at commercial-scale Stability >3 years at room temp for both oral and IV Oral tablets are bioequivalent to the IV Cost effective 3 step manufacturing process 11/18/
21 Omadacycline Power of Oral and IV Dosing for the Big 3 Indications (1) Antibiotic Class Oral Frequency Big 3 Indications (2) 2010 Sales (3,4) Levofloxacin Quinolone Once Daily 3 $3.4B Co-Amoxy clav B-Lactam Twice Daily 3 $2.8B Azithromycin Macrolide Once Daily 2 (2) $1.8B Ciprofloxacin Quinolone Twice Daily 3 $1.4B Clarithromycin Macrolide Twice Daily 2 (2) $1.4B Oral Use ~Two-thirds of Total Sales (1) Skin, Respiratory, UTI (2) Both Azithromycin and Clarithromycin did not have UTI claim (3) IMS global sales data in 2010 (4) Major patents had expired for all products by 2010 except Levofloxacin 11/18/
22 Significant at risk patient population in U.S. hospitals and community (1) ABSSSI, CABP, UTI ~38M by 2028 Total Patients (2) Non-elevated risk 66% Elevated risk(3) 34% ~13M Elevated- Risk Patients (1) No suspected drug resistant pathogens (DRP) 8M or 62% Suspected or confirmed DRP 5M or 38% ~5M patients with suspected or confirmed DRP (1) Community 3.5M Hospital 1.5M (1) Paratek Research and Analysis, September 2015 (2) Projected Total Patient Popultaion in the U.S. with Omadacycline potential indications of ABSSSI, CABP and UTI both hospital and community in 2028 (3) Elevated risk defined as Elderly, Immuno-compromised, Co-morbidity e.g, diabetes, history of treatment failure, recent hospitalization, resident of a nursing home, suspected or confirmed drug-resistant pathogen 11/18/
23 Omadacycline Value Proposition Aligns Well with Current and Future Patient Needs (1) Unmet Need Lack of Bio- equivalent IV-to-oral step-down therapies Hospital Formulary Acceptance: In the hospital setting for ABSSSI and CABP, the IV and oral formulations of omadacycline allow for IV-oral stepdown therapy in hospital, greatly facilitating patient discharge (1.5M patients with known or suspected drug resistant pathogens DRP) Need for oral therapies covering drug-resistant pathogens (DRP) Hospital & Community Adoption Opportunity: The broad spectrum of activity with coverage against drug resistant pathogens (MRSA, Penicillin and macrolideresistant S. pneumonia & MDR E. coli), and an oral formulation, make Omadacycline especially suited for elevated risk patients with confirmed or suspected DRP (1.5M Hospital + 3.5M Community Patients) Improved safety, tolerability & Once-daily dosing Expansion Opportunity: A lack of drug-drug interactions, overall tolerability profile and once-daily oral dosing makes Omadacycline suitable for all elevated risk patients with or without DRP (8M patients) (1 Paratek Research and Analysis September /18/
24 Omadacycline Potential Broad Range of Major Indications Scarcity of Late-Stage IV/Oral Antibiotics in Development Late-Stage IV & Oral Omadacycline Hospital Community cuti ABSSSI CABP CABP ABSSSI UTI Other Sinusitis Delafloxacin Finafloxacin Lefamulin Solithromycin Neisseria 11/18/
25 Omadacycline Strong IP Through 2028 U.S. Base Composition of Matter plus anticipated patent term extension into 2028 And U.S. Hatch Waxman plus GAIN Act extension totaling 10 yrs. EU: 10 yrs. of market exclusivity expected 11/18/
26 Sarecycline: Narrow-spectrum Tetracycline Antibiotic Specifically Designed for Inflammatory Acne Novel, narrow-spectrum antibiotic Demonstrated anti-inflammatory activity Does not cross Blood-Brain Barrier Favorable GI tolerability Once-daily Oral formulation Composition of Matter IP protection U.S. Base Composition of Matter: 2031 EU: 10 years of market exclusivity expected 11/18/
27 Sarecycline Late-Stage Development Progressing as Planned U.S. commercial rights: Allergan Ex-U.S. commercial rights: Paratek Phase 2 Trials met primary endpoints for efficacy and safety (1) Phase 3 Trials in U.S. underway; Data expected in 2016 Milestones and royalties to Paratek Allergan estimates $ M peak U.S. revenue (1) Solodyn analogue supports sales potential Peak sales >$750M (reformulated minocycline) (2) (1) Allergan plc, Investor Day 2015, p. 142 (February 18, 2015) (2) IMS Sales data /18/
28 Phase 3 & NDA Filing Milestones for Omadacycline and Sarecycline Omadacycline Events Estimated Timing ABSSSI Phase 3 Study Initiated enrollment 2015 CABP Phase 3 Study Initiation Late 2015 ABSSSI Phase 3 Data 2H 2016 CABP Phase 3 Data 2H 2017 Omadacycline Filing 2018 Sarecycline Events (1) Estimated Timing Sarecycline Phase 3 Efficacy Data (2 trials) 2016 Sarecycline Phase 3 long term safety study Data 2016 Sarecycline Filing 2017 (1) Allergan owns U.S. development & commercial rights 11/18/
29 Key Financial Information Total Cash (1) (2) $146.4 Total Debt, Net of Issuance Costs Key Metrics (1) (2) $19.5 million million Basic Shares Outstanding (1) 17,608,615 Stock Options, Restricted Stock, and Warrants Outstanding (1) 2,412,813 Cash balance expected to fund operations through 2H 2017 Potential BD opportunities Sarecycline: Ex-U.S. development and commercialization rights Monetize potential U.S. royalties Omadacycline: Ex-U.S. partnerships (e.g., Asia) (1) As of September 30, 2015 (2) Note: On September 30, 2015, Company drew $20 million of $40 million debt line 11/18/
30 Paratek Investment Highlights Developing novel tetracyclines to address unmet medical needs Omadacycline: Oral/IV antibiotic addressing bacterial resistance Enrolling two pivotal trials ABSSSI and CABP Developing for UTI Potential additional indications - Sinusitis Worldwide commercial rights retained Sarecycline: Antimicrobial and anti-inflammatory tetracycline for treating acne Partnered with Allergan in U.S. Ex-U.S. commercial rights retained Proven management team Strong IP position 11/18/
31 11/18/ Back Up Slides
32 Omadacycline: Data supports Potential CABP Indication Tygacil Omadacycline Animal Pneumonia Model Efficacy Yes Yes AUC:MIC PK Target 12.8 (human) (1) (mouse) AUC (human; µg-hr/ml) (2) ~ 4.7 ~ 10 MIC 90 (µg/ml; S. pneumoniae) 0.06 (3) 0.12 AUC:MIC (human) Achieved ~ 80 ~ 80 Efficacy for CABP Approved Phase 3 11/18/ (1) Rubino et al Pharmacokinetics-Pharmacodynamics of Tigecycline in Patients with Community-Acquired Pneumonia. AAC56: ; (2) Tygacil AUC: Tygacil Product Label; Omadacycline AUC: Sun, H. et al A single dose study to evaluate the pharmacokinetics, safety and tolerability of multiple formulations of PTK 0796 in healthy subjects. ECCMID 2012, Poster P1423. (3) Tomic and Dowzicky Regional and Global Susceptibility among isolates of Streptococcus pneumoniae and Haemophilus influenzae collected as part of the Tigecycline Evaluation and Surveillance Trial (Table S1). Ann Clin Micro Antimicrob. 13:52.;
Flamel Technologies Provides Update on Corporate Progress
 Flamel Technologies Provides Update on Corporate Progress Clinical Programs Continue to Move Forward Outlines Full Year 2016 Revenue Guidance Lyon, France January 8, 2016 - Flamel Technologies (NASDAQ:
Flamel Technologies Provides Update on Corporate Progress Clinical Programs Continue to Move Forward Outlines Full Year 2016 Revenue Guidance Lyon, France January 8, 2016 - Flamel Technologies (NASDAQ:
Theravance Biopharma, Inc. Reports Second Quarter 2015 Financial Results and Provides Business Update
 August 10, 2015 Theravance Biopharma, Inc. Reports Second Quarter 2015 Financial Results and Provides Business Update DUBLIN, IRELAND -- (Marketwired) -- 08/10/15 -- Theravance Biopharma, Inc. (NASDAQ:
August 10, 2015 Theravance Biopharma, Inc. Reports Second Quarter 2015 Financial Results and Provides Business Update DUBLIN, IRELAND -- (Marketwired) -- 08/10/15 -- Theravance Biopharma, Inc. (NASDAQ:
Sanofi Reports Positive Phase 3 Results for Toujeo (insulin glargine [rdna origin] injection, 300 U/mL)
![Sanofi Reports Positive Phase 3 Results for Toujeo (insulin glargine [rdna origin] injection, 300 U/mL) Sanofi Reports Positive Phase 3 Results for Toujeo (insulin glargine [rdna origin] injection, 300 U/mL)](/thumbs/26/8616095.jpg) PRESS RELEASE Sanofi Reports Positive Phase 3 Results for Toujeo (insulin glargine [rdna origin] injection, 300 U/mL) Meta-analysis of three late-stage trials in people with type 2 diabetes shows decreases
PRESS RELEASE Sanofi Reports Positive Phase 3 Results for Toujeo (insulin glargine [rdna origin] injection, 300 U/mL) Meta-analysis of three late-stage trials in people with type 2 diabetes shows decreases
Flamel Technologies Announces Second Quarter Results of Fiscal Year 2015
 Flamel Technologies Announces Second Quarter Results of Fiscal Year 2015 Product revenue guidance for 2015 of $170-$185 million reaffirmed Conference call with management to take place at 10:00 am ET on
Flamel Technologies Announces Second Quarter Results of Fiscal Year 2015 Product revenue guidance for 2015 of $170-$185 million reaffirmed Conference call with management to take place at 10:00 am ET on
MEDICAL DEVICE & DIAGNOSTICS
 to Guide Strategic Medical Device and Introduction Medical device and diagnostic makers face myriad challenges when developing a new product, including working within resource and financial constraints,
to Guide Strategic Medical Device and Introduction Medical device and diagnostic makers face myriad challenges when developing a new product, including working within resource and financial constraints,
Paul K. Wotton, Ph.D President and Chief Executive Officer May 2012 AMEX: AIS
 Paul K. Wotton, Ph.D President and Chief Executive Officer May 2012 AMEX: AIS Safe Harbor Statement This presentation may contain forward-looking statements which are made pursuant to the safe harbor provisions
Paul K. Wotton, Ph.D President and Chief Executive Officer May 2012 AMEX: AIS Safe Harbor Statement This presentation may contain forward-looking statements which are made pursuant to the safe harbor provisions
Urinary Tract Infections
 Urinary Tract Infections Overview A urine culture must ALWAYS be interpreted in the context of the urinalysis and patient symptoms. If a patient has no signs of infection on urinalysis, no symptoms of
Urinary Tract Infections Overview A urine culture must ALWAYS be interpreted in the context of the urinalysis and patient symptoms. If a patient has no signs of infection on urinalysis, no symptoms of
U.S. Scientific Update Aricept 23 mg Tablets. Dr. Lynn Kramer President NeuroScience Product Creation Unit Eisai Inc.
 U.S. Scientific Update Aricept 23 mg Tablets Dr. Lynn Kramer President NeuroScience Product Creation Unit Eisai Inc. Unmet Need in Moderate to Severe Alzheimer s Disease (AD) Ongoing clinical deterioration
U.S. Scientific Update Aricept 23 mg Tablets Dr. Lynn Kramer President NeuroScience Product Creation Unit Eisai Inc. Unmet Need in Moderate to Severe Alzheimer s Disease (AD) Ongoing clinical deterioration
Q3 2015 Financial Results and Corporate Update. November 4, 2015
 Q3 2015 Financial Results and Corporate Update November 4, 2015 Introductions and Forward- Looking Statements Silvia Taylor, SVP Investor Relations and Corporate Affairs Agenda Introductions and Forward-Looking
Q3 2015 Financial Results and Corporate Update November 4, 2015 Introductions and Forward- Looking Statements Silvia Taylor, SVP Investor Relations and Corporate Affairs Agenda Introductions and Forward-Looking
A Leader in Medication Management and Patient Safety. March 17, 2015
 A Leader in Medication Management and Patient Safety March 17, 2015 Disclaimers This slide presentation contains certain estimates and other forward-looking statements (as defined under Federal securities
A Leader in Medication Management and Patient Safety March 17, 2015 Disclaimers This slide presentation contains certain estimates and other forward-looking statements (as defined under Federal securities
The Future of Consumer Health Care
 The Future of Consumer Health Care Coming Together To Lead The Consumer Health Care Industry 2 Creating a New Business Model in Consumer Health Care 3 Serve More Consumers In More Parts of the World, More
The Future of Consumer Health Care Coming Together To Lead The Consumer Health Care Industry 2 Creating a New Business Model in Consumer Health Care 3 Serve More Consumers In More Parts of the World, More
Credit Suisse Healthcare Conference
 Credit Suisse Healthcare Conference November 14, 2013 Safe Harbor Statement 2 Safe Harbor Statement under the Private Securities Litigation Reform Act of 1995: To the extent any statements made in this
Credit Suisse Healthcare Conference November 14, 2013 Safe Harbor Statement 2 Safe Harbor Statement under the Private Securities Litigation Reform Act of 1995: To the extent any statements made in this
Genzyme s Multiple Sclerosis Franchise Featured at AAN
 PRESS RELEASE Genzyme s Multiple Sclerosis Franchise Featured at AAN - Multiple Presentations Highlight Continuing Progress of AUBAGIO and LEMTRADA Programs - Paris, France March 13, 2013 Sanofi (EURONEXT:
PRESS RELEASE Genzyme s Multiple Sclerosis Franchise Featured at AAN - Multiple Presentations Highlight Continuing Progress of AUBAGIO and LEMTRADA Programs - Paris, France March 13, 2013 Sanofi (EURONEXT:
How To Understand And Understand Biosimilars
 Welcome to Biosimilars 20/20, presented by the Specialty Pharma Education Center (SPEC) in partnership with the Specialty Pharma Journal (SPJ). Please note that this is a preliminary agenda that is subject
Welcome to Biosimilars 20/20, presented by the Specialty Pharma Education Center (SPEC) in partnership with the Specialty Pharma Journal (SPJ). Please note that this is a preliminary agenda that is subject
AT&T to Acquire DIRECTV May 19, 2014
 AT&T to Acquire DIRECTV May 19, 2014 2014 AT&T Intellectual Property. All rights reserved. AT&T, the AT&T logo and all other marks contained herein are trademarks of AT&T Intellectual Property and/or AT&T
AT&T to Acquire DIRECTV May 19, 2014 2014 AT&T Intellectual Property. All rights reserved. AT&T, the AT&T logo and all other marks contained herein are trademarks of AT&T Intellectual Property and/or AT&T
Breakthrough oral drug delivery technology for diabetes, drugs and vaccines November 5, 2014
 Oramed Pharmaceuticals, Inc. Initiating Report Breakthrough oral drug delivery technology for diabetes, drugs and vaccines November 5, 2014 Key data Sector Rating Biotechnology BUY Target Price $45.00
Oramed Pharmaceuticals, Inc. Initiating Report Breakthrough oral drug delivery technology for diabetes, drugs and vaccines November 5, 2014 Key data Sector Rating Biotechnology BUY Target Price $45.00
2016 PQRS OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY
 Measure #65 (NQF 0069): Appropriate Treatment for Children with Upper Respiratory Infection (URI) National Quality Strategy Domain: Efficiency and Cost Reduction 2016 PQRS OPTIONS FOR INDIVIDUAL MEASURES:
Measure #65 (NQF 0069): Appropriate Treatment for Children with Upper Respiratory Infection (URI) National Quality Strategy Domain: Efficiency and Cost Reduction 2016 PQRS OPTIONS FOR INDIVIDUAL MEASURES:
Lessons for the United States: Biosimilar Market Development Worldwide
 Lessons for the United States: Biosimilar Market Development Worldwide Sumant Ramachandra, MD, PhD, MBA Senior Vice President, Chief Scientific Officer Hospira $67B+ of 2012 LMV is expected to face biosimilar
Lessons for the United States: Biosimilar Market Development Worldwide Sumant Ramachandra, MD, PhD, MBA Senior Vice President, Chief Scientific Officer Hospira $67B+ of 2012 LMV is expected to face biosimilar
ACCELERATING BIOTECHNOLOGY INNOVATION
 ACCELERATING BIOTECHNOLOGY INNOVATION FOR RARE DISEASES: Challenges and solutions Emil D. Kakkis, M.D., Ph.D. President and Founder, EFRD Also CEO, Ultragenyx Pharmaceutical Inc. April 17, 2013 1 EveryLife
ACCELERATING BIOTECHNOLOGY INNOVATION FOR RARE DISEASES: Challenges and solutions Emil D. Kakkis, M.D., Ph.D. President and Founder, EFRD Also CEO, Ultragenyx Pharmaceutical Inc. April 17, 2013 1 EveryLife
For personal use only
 ACRUX (ACR) - ASX ANNOUNCEMENT 20 FEBRUARY 2014 ACRUX PROVIDES HALF YEAR UPDATE 2013 Highlights: Axiron sales milestone achieved US$25 million receivable early March 2014 Half-year financials: o Revenue
ACRUX (ACR) - ASX ANNOUNCEMENT 20 FEBRUARY 2014 ACRUX PROVIDES HALF YEAR UPDATE 2013 Highlights: Axiron sales milestone achieved US$25 million receivable early March 2014 Half-year financials: o Revenue
OPKO Health to Acquire Bio-Reference Laboratories
 OPKO Health to Acquire Bio-Reference Laboratories - Complementary in-depth expertise in diagnostics business with state of the art experience in use of genomic data for personalized therapy - Acquisition
OPKO Health to Acquire Bio-Reference Laboratories - Complementary in-depth expertise in diagnostics business with state of the art experience in use of genomic data for personalized therapy - Acquisition
Third-Quarter 2015 Earnings Conference Call Executive Commentary Highlights. October 27, 2015
 Third-Quarter 2015 Earnings Conference Call Executive Commentary Highlights October 27, 2015 Forward-Looking Statement of Merck & Co., Inc., Kenilworth, N.J., USA This presentation of Merck & Co., Inc.,
Third-Quarter 2015 Earnings Conference Call Executive Commentary Highlights October 27, 2015 Forward-Looking Statement of Merck & Co., Inc., Kenilworth, N.J., USA This presentation of Merck & Co., Inc.,
Frequently Asked Questions
 Guidelines for Testing and Treatment of Gonorrhea in Ontario, 2013 Frequently Asked Questions Table of Contents Background... 1 Treatment Recommendations... 2 Treatment of Contacts... 4 Administration
Guidelines for Testing and Treatment of Gonorrhea in Ontario, 2013 Frequently Asked Questions Table of Contents Background... 1 Treatment Recommendations... 2 Treatment of Contacts... 4 Administration
Introduction to Enteris BioPharma
 Introduction to Enteris BioPharma Enteris BioPharma Intelligent Solutions for Oral Drug Delivery Privately held, New Jersey based biotech company Owned solely by Victory Park Capital, a large Chicago based
Introduction to Enteris BioPharma Enteris BioPharma Intelligent Solutions for Oral Drug Delivery Privately held, New Jersey based biotech company Owned solely by Victory Park Capital, a large Chicago based
THIS IS AN OFFICIAL NH DHHS HEALTH ALERT
 THIS IS AN OFFICIAL NH DHHS HEALTH ALERT Distributed by the NH Health Alert Network [email protected] August 13, 2015 1400 EDT (2:00 PM EDT) NH-HAN 20150813 Updated Centers for Disease Control (CDC)
THIS IS AN OFFICIAL NH DHHS HEALTH ALERT Distributed by the NH Health Alert Network [email protected] August 13, 2015 1400 EDT (2:00 PM EDT) NH-HAN 20150813 Updated Centers for Disease Control (CDC)
Ohr Pharmaceutical Reports Fiscal Year 2015 Financial and Business Results
 December 10, 2015 Ohr Pharmaceutical Reports Fiscal Year 2015 Financial and Business Results OHR-102 Phase 3 Program in Wet-AMD to be Initiated Upon Completion of SPA; Enroll Patients in Q1 2016 Conference
December 10, 2015 Ohr Pharmaceutical Reports Fiscal Year 2015 Financial and Business Results OHR-102 Phase 3 Program in Wet-AMD to be Initiated Upon Completion of SPA; Enroll Patients in Q1 2016 Conference
Immunovaccine Inc. (TSX-V: IMV) July 2011
 Immunovaccine Inc. (TSX-V: IMV) July 2011 2 Forward Looking Statements This document contains forward-looking information pursuant to applicable securities law. All information that addresses activities
Immunovaccine Inc. (TSX-V: IMV) July 2011 2 Forward Looking Statements This document contains forward-looking information pursuant to applicable securities law. All information that addresses activities
CAN-FITE BIOPHARMA LTD.
 UNITED STATES SECURITIES AND EXCHANGE COMMISSION Washington, D.C. 20549 FORM 6-K Report of Foreign Private Issuer Pursuant to Rule 13a-16 or 15d-16 Under the Securities Exchange Act of 1934 For the Month
UNITED STATES SECURITIES AND EXCHANGE COMMISSION Washington, D.C. 20549 FORM 6-K Report of Foreign Private Issuer Pursuant to Rule 13a-16 or 15d-16 Under the Securities Exchange Act of 1934 For the Month
Mid-Clinical Stage Antiviral Drug Development Company
 BIOTRON LIMITED (ASX:BIT) Mid-Clinical Stage Antiviral Drug Development Company Investor Update 20 August 2015 Dr Michelle Miller Managing Director +61 412 313329 [email protected] www.biotron.com.au
BIOTRON LIMITED (ASX:BIT) Mid-Clinical Stage Antiviral Drug Development Company Investor Update 20 August 2015 Dr Michelle Miller Managing Director +61 412 313329 [email protected] www.biotron.com.au
CDC 2015 STD Treatment Guidelines: Update for IHS Providers Sharon Adler M.D., M.P.H.
 CDC 2015 STD Treatment Guidelines: Update for IHS Providers Sharon Adler M.D., M.P.H. Clinical Faculty, CA Prevention Training Center Disclosure Information Sharon Adler MD, MPH I have no financial relationships
CDC 2015 STD Treatment Guidelines: Update for IHS Providers Sharon Adler M.D., M.P.H. Clinical Faculty, CA Prevention Training Center Disclosure Information Sharon Adler MD, MPH I have no financial relationships
New Advances in Cancer Treatments. March 2015
 New Advances in Cancer Treatments March 2015 Safe Harbour Statement This presentation document contains certain forward-looking statements and information (collectively, forward-looking statements ) within
New Advances in Cancer Treatments March 2015 Safe Harbour Statement This presentation document contains certain forward-looking statements and information (collectively, forward-looking statements ) within
FORM 6-K. SECURITIES AND EXCHANGE COMMISSION Washington, D.C. 20549. Report of Foreign Private Issuer
 FORM 6-K SECURITIES AND EXCHANGE COMMISSION Washington, D.C. 20549 Report of Foreign Private Issuer Pursuant to Rule 13a-16 or 15d-16 under the Securities Exchange Act of 1934 For the month of August 2011
FORM 6-K SECURITIES AND EXCHANGE COMMISSION Washington, D.C. 20549 Report of Foreign Private Issuer Pursuant to Rule 13a-16 or 15d-16 under the Securities Exchange Act of 1934 For the month of August 2011
Antares Pharma, Inc. Princeton South Corporate Center 100 Princeton South, Suite 300 Ewing, NJ 08628 NASDAQ : ATRS. Paul
 Antares Pharma, Inc. Princeton South Corporate Center 100 Princeton South, Suite 300 Ewing, NJ 08628 NASDAQ : ATRS Paul Antares Mission: To develop and commercialize self-administered parenteral medicines
Antares Pharma, Inc. Princeton South Corporate Center 100 Princeton South, Suite 300 Ewing, NJ 08628 NASDAQ : ATRS Paul Antares Mission: To develop and commercialize self-administered parenteral medicines
Appropriate Treatment for Children with Upper Respiratory Infection
 BCBS ACO Measure Appropriate Treatment for Children with Upper Respiratory Infection HEDIS Measure CPT II coding required: YES Click here to go to Table of Contents BCBS Measure: Page 50 of 234 Dated:
BCBS ACO Measure Appropriate Treatment for Children with Upper Respiratory Infection HEDIS Measure CPT II coding required: YES Click here to go to Table of Contents BCBS Measure: Page 50 of 234 Dated:
How To Buy Chubb For $28.3 Billion In Cash And Stock
 ACE TO ACQUIRE CHUBB FOR $28.3 BILLION IN CASH AND STOCK Complementary Businesses and Skills Will Create Global P&C Industry Leader with Superior Product, Customer and Distribution Channel Capabilities
ACE TO ACQUIRE CHUBB FOR $28.3 BILLION IN CASH AND STOCK Complementary Businesses and Skills Will Create Global P&C Industry Leader with Superior Product, Customer and Distribution Channel Capabilities
The 505(b)(2) Drug Development Pathway:
 The 505(b)(2) Drug Development Pathway: When and How to Take Advantage of a Unique American Regulatory Pathway By Mukesh Kumar, PhD, RAC and Hemant Jethwani, MS The 505(b)(2) regulation offers a less expensive
The 505(b)(2) Drug Development Pathway: When and How to Take Advantage of a Unique American Regulatory Pathway By Mukesh Kumar, PhD, RAC and Hemant Jethwani, MS The 505(b)(2) regulation offers a less expensive
February 2006 Procedural
 Guidance for Industry Reports on the Status of Postmarketing Study Commitments Implementation of Section 130 of the Food and Drug Administration Modernization Act of 1997 U.S. Department of Health and
Guidance for Industry Reports on the Status of Postmarketing Study Commitments Implementation of Section 130 of the Food and Drug Administration Modernization Act of 1997 U.S. Department of Health and
Vancomycin. Beta-lactams. Beta-lactams. Vancomycin (Glycopeptide) Rifamycins (rifampin) MID 4
 Antibiotic Classes Introduction to Antimicrobials Rachel J. Gordon, MD, MPH Assistant Professor of Clinical Medicine and Epidemiology Beta-lactams Inhibit cell wall synthesis Penicillins Cephalosporins
Antibiotic Classes Introduction to Antimicrobials Rachel J. Gordon, MD, MPH Assistant Professor of Clinical Medicine and Epidemiology Beta-lactams Inhibit cell wall synthesis Penicillins Cephalosporins
Disclosure. This presentation contains forward-looking statements.
 Disclosure This presentation contains forward-looking statements. These forward-looking statements are based on management's current expectations and assumptions as of the date of this presentation, and
Disclosure This presentation contains forward-looking statements. These forward-looking statements are based on management's current expectations and assumptions as of the date of this presentation, and
Antimicrobial Stewardship for Hospital Acquired Infection Prevention: Focus on C. difficile infection
 Antimicrobial Stewardship for Hospital Acquired Infection Prevention: Focus on C. difficile infection Emi Minejima, PharmD Assistant Professor of Clinical Pharmacy USC School of Pharmacy [email protected]
Antimicrobial Stewardship for Hospital Acquired Infection Prevention: Focus on C. difficile infection Emi Minejima, PharmD Assistant Professor of Clinical Pharmacy USC School of Pharmacy [email protected]
HUSRES Annual Report 2010 Martti Vaara www.huslab.fi www.intra.hus.fi
 HUSRES Annual Report 2010 Martti Vaara www.huslab.fi www.intra.hus.fi Martti Vaara, 2/2011 1 The basis of this HUSRES 2010 report is the HUSLAB/Whonet database 2010, which contains susceptibility data
HUSRES Annual Report 2010 Martti Vaara www.huslab.fi www.intra.hus.fi Martti Vaara, 2/2011 1 The basis of this HUSRES 2010 report is the HUSLAB/Whonet database 2010, which contains susceptibility data
BNC105 CANCER CLINICAL TRIALS REACH KEY MILESTONES CLINICAL PROGRAM TO BE EXPANDED
 ASX ANNOUNCEMENT 3 August 2011 ABN 53 075 582 740 BNC105 CANCER CLINICAL TRIALS REACH KEY MILESTONES CLINICAL PROGRAM TO BE EXPANDED Data from renal cancer trial supports progression of the trial: o Combination
ASX ANNOUNCEMENT 3 August 2011 ABN 53 075 582 740 BNC105 CANCER CLINICAL TRIALS REACH KEY MILESTONES CLINICAL PROGRAM TO BE EXPANDED Data from renal cancer trial supports progression of the trial: o Combination
Great Basin Reports 2015 Second Quarter Results and Business Update
 Great Basin Reports 2015 Second Quarter Results and Business Update Company Reports 122 Revenue-Generating Customers, Reaffirms Guidance of 170-180 Customers by Year End SALT LAKE CITY, August 12, 2015
Great Basin Reports 2015 Second Quarter Results and Business Update Company Reports 122 Revenue-Generating Customers, Reaffirms Guidance of 170-180 Customers by Year End SALT LAKE CITY, August 12, 2015
Ted Quiroz Director, Global Clinical Supply Chain Amgen Inc.
 Ted Quiroz Director, Global Clinical Supply Chain Amgen Inc. to serve patients Enbrel (etanercept) EPOGEN (Epoetin alfa) Aranesp (Darbepoetin alfa) Sensipar (cinacalcet HCl) Vectibix (panitumumab) We aspire
Ted Quiroz Director, Global Clinical Supply Chain Amgen Inc. to serve patients Enbrel (etanercept) EPOGEN (Epoetin alfa) Aranesp (Darbepoetin alfa) Sensipar (cinacalcet HCl) Vectibix (panitumumab) We aspire
UNITED STATES SECURITIES AND EXCHANGE COMMISSION WASHINGTON, D.C. 20549 FORM 8-K
 UNITED STATES SECURITIES AND EXCHANGE COMMISSION WASHINGTON, D.C. 20549 FORM 8-K CURRENT REPORT Pursuant To Section 13 or 15(d) of the Securities Exchange Act of 1934 Date of Report (date of earliest event
UNITED STATES SECURITIES AND EXCHANGE COMMISSION WASHINGTON, D.C. 20549 FORM 8-K CURRENT REPORT Pursuant To Section 13 or 15(d) of the Securities Exchange Act of 1934 Date of Report (date of earliest event
Guidance for Industry Antibacterial Therapies for Patients With Unmet Medical Need for the Treatment of Serious Bacterial Diseases
 Guidance for Industry Antibacterial Therapies for Patients With Unmet Medical Need for the Treatment of Serious Bacterial Diseases DRAFT GUIDANCE This guidance document is being distributed for comment
Guidance for Industry Antibacterial Therapies for Patients With Unmet Medical Need for the Treatment of Serious Bacterial Diseases DRAFT GUIDANCE This guidance document is being distributed for comment
2009 Foley & Lardner LLP Attorney Advertising Prior results do not guarantee a similar outcome Models used are not clients but may be representative
 Attorney Advertising Prior results do not guarantee a similar outcome Models used are not clients but may be representative of clients 321 N. Clark Street, Suite 2800, Chicago, IL 60654 312.832.4500 Life
Attorney Advertising Prior results do not guarantee a similar outcome Models used are not clients but may be representative of clients 321 N. Clark Street, Suite 2800, Chicago, IL 60654 312.832.4500 Life
Dec. 9, 2013, 11:00 a.m. EST
 Dec. 9, 2013, 11:00 a.m. EST Portola Pharmaceuticals Announces New Phase 2 Results Confirming Immediate, Dose-Dependent and Well-Tolerated Reversal of Anticoagulation Activity of XARELTO(R) (rivaroxaban)
Dec. 9, 2013, 11:00 a.m. EST Portola Pharmaceuticals Announces New Phase 2 Results Confirming Immediate, Dose-Dependent and Well-Tolerated Reversal of Anticoagulation Activity of XARELTO(R) (rivaroxaban)
Achieving Regulatory Success: Areas of focus for biotechnology companies. Michael J. Schlosser, PhD, DABT April 21, 2013
 Achieving Regulatory Success: Areas of focus for biotechnology companies Michael J. Schlosser, PhD, DABT April 21, 2013 Regulatory Success Outline Regulatory Initiatives Regulatory Science Pre-Regulatory
Achieving Regulatory Success: Areas of focus for biotechnology companies Michael J. Schlosser, PhD, DABT April 21, 2013 Regulatory Success Outline Regulatory Initiatives Regulatory Science Pre-Regulatory
How To Treat Mrsa From A Dead Body
 HUSRES Annual Report 2012 Martti Vaara www.huslab.fi www.intra.hus.fi Martti Vaara 2013 1 The basis of this HUSRES 2012 report is the HUSLAB/Whonet database 2012, which contains susceptibility data on
HUSRES Annual Report 2012 Martti Vaara www.huslab.fi www.intra.hus.fi Martti Vaara 2013 1 The basis of this HUSRES 2012 report is the HUSLAB/Whonet database 2012, which contains susceptibility data on
MOLOGEN AG. Q1 Results 2015 Conference Call Dr. Matthias Schroff Chief Executive Officer. Berlin, 12 May 2015
 Q1 Results 2015 Conference Call Dr. Matthias Schroff Chief Executive Officer Berlin, 12 May 2015 V1-6 Disclaimer Certain statements in this presentation contain formulations or terms referring to the future
Q1 Results 2015 Conference Call Dr. Matthias Schroff Chief Executive Officer Berlin, 12 May 2015 V1-6 Disclaimer Certain statements in this presentation contain formulations or terms referring to the future
Localised Cancer Treatment. PCI Biotech. Amphinex a new product for localised cancer treatment
 Localised Cancer Treatment PCI Biotech Amphinex a new product for localised cancer treatment Disclaimer This document (the Presentation ) has been produced by PCI Biotech Holding ASA (the Company ). The
Localised Cancer Treatment PCI Biotech Amphinex a new product for localised cancer treatment Disclaimer This document (the Presentation ) has been produced by PCI Biotech Holding ASA (the Company ). The
XENOPORT INC FORM 8-K. (Current report filing) Filed 05/08/14 for the Period Ending 05/08/14
 XENOPORT INC FORM 8-K (Current report filing) Filed 05/08/14 for the Period Ending 05/08/14 Address 3410 CENTRAL EXPRESSWAY SANTA CLARA, CA 95051 Telephone 4086167200 CIK 0001130591 Symbol XNPT SIC Code
XENOPORT INC FORM 8-K (Current report filing) Filed 05/08/14 for the Period Ending 05/08/14 Address 3410 CENTRAL EXPRESSWAY SANTA CLARA, CA 95051 Telephone 4086167200 CIK 0001130591 Symbol XNPT SIC Code
UNITED STATES SECURITIES AND EXCHANGE COMMISSION Washington, D.C. 20549 FORM 8-K CURRENT REPORT
 UNITED STATES SECURITIES AND EXCHANGE COMMISSION Washington, D.C. 20549 FORM 8-K CURRENT REPORT Pursuant to Section 13 OR 15(d) of The Securities Exchange Act of 1934 Date of Report (Date of earliest event
UNITED STATES SECURITIES AND EXCHANGE COMMISSION Washington, D.C. 20549 FORM 8-K CURRENT REPORT Pursuant to Section 13 OR 15(d) of The Securities Exchange Act of 1934 Date of Report (Date of earliest event
Exelixis Showcases R&D Pipeline at JPMorgan Healthcare Conference
 Exelixis Showcases R&D Pipeline at JPMorgan Healthcare Conference Two New Clinical Programs and Significant Expansion of Cancer Pipeline Planned for 2004 SOUTH SAN FRANCISCO, Calif., Jan. 13 /PRNewswire-FirstCall/
Exelixis Showcases R&D Pipeline at JPMorgan Healthcare Conference Two New Clinical Programs and Significant Expansion of Cancer Pipeline Planned for 2004 SOUTH SAN FRANCISCO, Calif., Jan. 13 /PRNewswire-FirstCall/
Expedited Partner Therapy (EPT) for Sexually Transmitted Diseases Protocol for Health Care Providers in Oregon
 Expedited Partner Therapy (EPT) for Sexually Transmitted Diseases Protocol for Health Care Providers in Oregon Oregon Health Authority Center for Public Health Practice HIV/STD/TB Section Principles of
Expedited Partner Therapy (EPT) for Sexually Transmitted Diseases Protocol for Health Care Providers in Oregon Oregon Health Authority Center for Public Health Practice HIV/STD/TB Section Principles of
- Patients treated with alemtuzumab in CARE-MS II were more than twice as likely to experience disability improvement compared to Rebif -
 PRESS RELEASE Significant Improvement in Disability Scores Observed in Multiple Sclerosis Patients Who Received Lemtrada TM* (Alemtuzumab) Compared With Rebif in Phase lll Trial - Patients treated with
PRESS RELEASE Significant Improvement in Disability Scores Observed in Multiple Sclerosis Patients Who Received Lemtrada TM* (Alemtuzumab) Compared With Rebif in Phase lll Trial - Patients treated with
HUSRES Annual Report 2008 Martti Vaara. www.huslab.fi www.intra.hus.fi
 HUSRES Annual Report 2008 Martti Vaara www.huslab.fi www.intra.hus.fi The basis of this HUSRES 2008 report is the HUSLAB/Whonet database 2008, which contains susceptibility data on about 180.000 bacteria
HUSRES Annual Report 2008 Martti Vaara www.huslab.fi www.intra.hus.fi The basis of this HUSRES 2008 report is the HUSLAB/Whonet database 2008, which contains susceptibility data on about 180.000 bacteria
A LEADING GLOBAL HEALTH CARE GROUP. Frankfurt Stock Exchange (DAX30): FRE US ADR program (OTC): FSNUY wwww.fresenius.com/investors
 A LEADING GLOBAL HEALTH CARE GROUP Frankfurt Stock Exchange (DAX30): FRE US ADR program (OTC): FSNUY wwww.fresenius.com/investors SAFE HARBOR STATEMENT This presentation contains forward-looking statements
A LEADING GLOBAL HEALTH CARE GROUP Frankfurt Stock Exchange (DAX30): FRE US ADR program (OTC): FSNUY wwww.fresenius.com/investors SAFE HARBOR STATEMENT This presentation contains forward-looking statements
Drug Utilization and Evaluation of Cephalosporin s At Tertiary Care Teaching Hospital, Bangalore
 Research Article Drug Utilization and Evaluation of Cephalosporin s At Tertiary Care Teaching Hospital, Bangalore HS. Shekar 1*, HR. Chandrashekhar 2, M. Govindaraju 3, P. Venugopalreddy 1, Chikkalingiah
Research Article Drug Utilization and Evaluation of Cephalosporin s At Tertiary Care Teaching Hospital, Bangalore HS. Shekar 1*, HR. Chandrashekhar 2, M. Govindaraju 3, P. Venugopalreddy 1, Chikkalingiah
ANTIBIOTICS IN SEPSIS
 ANTIBIOTICS IN SEPSIS Jennifer Curello, PharmD, BCPS Clinical Pharmacist, Infectious Diseases Antimicrobial Stewardship Program Ronald Reagan UCLA Medical Center October 27, 2014 The power of antibiotics
ANTIBIOTICS IN SEPSIS Jennifer Curello, PharmD, BCPS Clinical Pharmacist, Infectious Diseases Antimicrobial Stewardship Program Ronald Reagan UCLA Medical Center October 27, 2014 The power of antibiotics
Select Medical Holdings Corporation Announces Results for Second Quarter Ended June 30, 2015
 R E L E A S E FOR IMMEDIATE RELEASE 4714 Gettysburg Road Mechanicsburg, PA 17055 Select Medical Holdings Corporation Announces Results for Second Quarter Ended June 30, 2015 NYSE Symbol: SEM MECHANICSBURG,
R E L E A S E FOR IMMEDIATE RELEASE 4714 Gettysburg Road Mechanicsburg, PA 17055 Select Medical Holdings Corporation Announces Results for Second Quarter Ended June 30, 2015 NYSE Symbol: SEM MECHANICSBURG,
Information Services Group Agrees to Acquire TPI, Global Leader in Sourcing Advisory Services
 For immediate release Press Contacts: Barry Holt /ISG 203-517-3110 [email protected] Andrea Riffle/TPI 954-659-8616 [email protected] Information Services Group Agrees to Acquire TPI, Global
For immediate release Press Contacts: Barry Holt /ISG 203-517-3110 [email protected] Andrea Riffle/TPI 954-659-8616 [email protected] Information Services Group Agrees to Acquire TPI, Global
Quality Affordable Healthcare Products. Perrigo Company to Acquire Elan to Create Premier Global Healthcare Company July 31, 2013
 Perrigo Company to Acquire Elan to Create Premier Global Healthcare Company July 31, 2013 Important Information For Investors And Shareholders This announcement does not constitute an offer to sell, or
Perrigo Company to Acquire Elan to Create Premier Global Healthcare Company July 31, 2013 Important Information For Investors And Shareholders This announcement does not constitute an offer to sell, or
Jefferies Healthcare Conference
 Jefferies Healthcare Conference Marc Naughton Executive Vice President and Chief Financial Officer June 2, 2015 Safe Harbor Statement This presentation may contain forward-looking statements, including
Jefferies Healthcare Conference Marc Naughton Executive Vice President and Chief Financial Officer June 2, 2015 Safe Harbor Statement This presentation may contain forward-looking statements, including
SORRENTO THERAPEUTICS, INC.
 ˆ200F@WVSVmdG1Yng+Š 200F@WVSVmdG1Yng+ AZ0151AC599817 11.3.10 SWRvelaj0nb 31-Jul-2013 16:51 EST 577580 TX 1 3* UNITED STATES SECURITIES AND EXCHANGE COMMISSION Washington, DC 20549 CURRENT REPORT Pursuant
ˆ200F@WVSVmdG1Yng+Š 200F@WVSVmdG1Yng+ AZ0151AC599817 11.3.10 SWRvelaj0nb 31-Jul-2013 16:51 EST 577580 TX 1 3* UNITED STATES SECURITIES AND EXCHANGE COMMISSION Washington, DC 20549 CURRENT REPORT Pursuant
Presented at: Jefferies 2015 Global Healthcare Conference
 Presented at: Jefferies 2015 Global Healthcare Conference Agenda 1 Overview & Service Platforms 2 Adaptability, Scalability & Expansion Plans 3 Best Practices 4 Blue Chip Customer Base 5 Roadmap of Evolution
Presented at: Jefferies 2015 Global Healthcare Conference Agenda 1 Overview & Service Platforms 2 Adaptability, Scalability & Expansion Plans 3 Best Practices 4 Blue Chip Customer Base 5 Roadmap of Evolution
Treatment of skin and soft tissue infections due to methicillin-resistant Staphylococcus aureus in adults
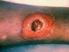 1 of 6 9/24/2010 11:16 AM Official reprint from UpToDate www.uptodate.com 2010 UpToDate Treatment of skin and soft tissue infections due to methicillin-resistant Staphylococcus aureus in adults Author
1 of 6 9/24/2010 11:16 AM Official reprint from UpToDate www.uptodate.com 2010 UpToDate Treatment of skin and soft tissue infections due to methicillin-resistant Staphylococcus aureus in adults Author
Investor Presentation
 Investor Presentation October 2015 Jay Hoffman Chief Strategy Officer Forward Looking Statements & Statutory Rights of Action FORWARD-LOOKING STATEMENTS Certain statements in this presentation (the "Presentation")
Investor Presentation October 2015 Jay Hoffman Chief Strategy Officer Forward Looking Statements & Statutory Rights of Action FORWARD-LOOKING STATEMENTS Certain statements in this presentation (the "Presentation")
PRIORITY RESEARCH TOPICS
 PRIORITY RESEARCH TOPICS Understanding all the issues associated with antimicrobial resistance is probably impossible, but it is clear that there are a number of key issues about which we need more information.
PRIORITY RESEARCH TOPICS Understanding all the issues associated with antimicrobial resistance is probably impossible, but it is clear that there are a number of key issues about which we need more information.
EINSTEIN PE Data Summary & Perspectives on XARELTO (rivaroxaban) in ORS & NVAF. Recorded Webcast Update for Analysts and Investors March 26, 2012
 EINSTEIN PE Data Summary & Perspectives on XARELTO (rivaroxaban) in ORS & NVAF Recorded Webcast Update for Analysts and Investors March 26, 2012 1 Webcast Presentation Agenda EINSTEIN PE Clinical Trial
EINSTEIN PE Data Summary & Perspectives on XARELTO (rivaroxaban) in ORS & NVAF Recorded Webcast Update for Analysts and Investors March 26, 2012 1 Webcast Presentation Agenda EINSTEIN PE Clinical Trial
Investor News. Phase III J-ROCKET AF Study of Bayer s Xarelto (rivaroxaban) Meets Primary Endpoint. Not intended for U.S.
 Investor News Not intended for U.S. and UK Media Bayer AG Investor Relations 51368 Leverkusen Germany www.investor.bayer.com Phase III J-ROCKET AF Study of Bayer s Xarelto (rivaroxaban) Meets Primary Endpoint
Investor News Not intended for U.S. and UK Media Bayer AG Investor Relations 51368 Leverkusen Germany www.investor.bayer.com Phase III J-ROCKET AF Study of Bayer s Xarelto (rivaroxaban) Meets Primary Endpoint
Corporate Overview. April 2014 OTCQB: LCDX
 Corporate Overview April 2014 OTCQB: LCDX Company Snapshot biopsy Provides non-invasive, point-of-care cellular imaging technologies VivaScope systems can improve patient outcomes while reducing costs
Corporate Overview April 2014 OTCQB: LCDX Company Snapshot biopsy Provides non-invasive, point-of-care cellular imaging technologies VivaScope systems can improve patient outcomes while reducing costs
First In Human Pediatric Trials and Safety Assessment for Rare and Orphan Diseases
 First In Human Pediatric Trials and Safety Assessment for Rare and Orphan Diseases Andrew E. Mulberg, MD, FAAP Division Deputy Director OND/ODE3/DGIEP FDA Partnership is the Key Coming together is a beginning;
First In Human Pediatric Trials and Safety Assessment for Rare and Orphan Diseases Andrew E. Mulberg, MD, FAAP Division Deputy Director OND/ODE3/DGIEP FDA Partnership is the Key Coming together is a beginning;
Daiichi Sankyo to Acquire Ambit Biosciences
 For Immediate Release Company name: DAIICHI SANKYO COMPANY, LIMITED Representative: Joji Nakayama, Representative Director, President and CEO (Code no.: 4568, First Section, Tokyo Stock Exchange) Please
For Immediate Release Company name: DAIICHI SANKYO COMPANY, LIMITED Representative: Joji Nakayama, Representative Director, President and CEO (Code no.: 4568, First Section, Tokyo Stock Exchange) Please
