Slik er planene for gjennomføring av byttestudien for biotilsvarende legemidler
|
|
|
- Damon Terry
- 10 years ago
- Views:
Transcription
1 Slik er planene for gjennomføring av byttestudien for biotilsvarende legemidler Tore K. Kvien Dept of Rheumatology Diakonhjemmet Hospital Oslo, Norway
2 Tore K Kvien disclosures Honorarium Institutional support NOR-DMARD Presentation Advice Previous Current AbbVie X X X BMS X X X X Eli Lilly X X MSD/ Schering-Plough X X X Pfizer/Wyeth X X X Roche X X X UCB X X X Hospira Orion X X Celltrion X Editor-in-Chief Annals of the Rheumatic Diseases
3 A great interest of biosimilar mabs in rheumatology
4
5
6 Polina P et al Ann Rheum Dis 2013 Epub ahead of print 12 August
7 Polina P et al Ann Rheum Dis 2013 Epub ahead of print 12 August
8 Polina P et al Ann Rheum Dis 2013 Epub ahead of print 12 August
9
10 Key question in Norway (and other parts of the world) Can patients on treatment with Remicade be switched/transitioned to CT-P13 (Remsima or Inflectra) to reduce medication cost (and improve access to bdmards for more patients in areas with major financial restrictions)? Is switching from from innovator to biosimilar infliximab safe? (efficacy, safety, immunogenicity) Limited switch data after 52 weeks are available in the PLANETRA and PLANETAS studies
11 Statsbudsjettet 2014: Det foreslås 20 mill. kroner til kliniske studier Det foreslås 20 mill. kroner til kliniske studier av bytte mellom biologiske og biotilsvarende legemidler. Når et biologisk legemiddel går av patent åpnes det for at andre produsenter kan lage tilvarende biologiske legemidler. Det er som oftest ikke mulig å lage helt identiske kopier av biologiske legemidler slik tilfellet er med syntetiske legemidler (generika), herav navnet biotilsvarende. Når europeiske legemiddelmyndigheter godkjenner et biotilsvarende legemiddel, er det en bekreftelse på at det har samme virkning som originallegemidlet. Ev. bytte mellom det originale biologiske legemidlet og det biotilsvarende legemidlet hos pasienter som allerede får behandling, er ikke en del av godkjenningen.
12 Statsbudsjettet 20 mill til byttestudier (2) Formålet med studiene er å dokumentere hvorvidt slikt bytte er trygt. Satsingen er i tråd med forslag om å styrke offentlig initierte kliniske studier innenfor andre fagområder enn kreftområdet, hvor det i dag pågår en satsing gjennom Norges forskningsråd. Behovet for offentlig initierte kliniske fase IV-studier på ulike fagområder er løftet fram av Nasjonalt råd for kvalitet og prioritering og i Meld. St. 10 ( ) God kvalitet trygge tjenester. Denne type forskning vil normalt ikke bli finansiert av kommersielle aktører og ofte heller ikke alltid være attraktiv for forskergrupper å initiere, men vil kunne gi resultater med stor samfunnsmessig verdi.
13 A RANDOMIZED, DOUBLE-BLIND, PARALLEL-GROUP STUDY TO EVALUATE THE SAFETY AND EFFICACY OF SWITCHING FROM INNOVATOR INFLIXIMAB TO BIOSIMILAR INFLIXIMAB COMPARED WITH CONTINUED TREATMENT WITH INNOVATOR INFLIXIMAB IN PATIENTS WITH RHEUMATOID ARTHRITIS, SPONDYLOARTHRITIS, PSORIATIC ARTHRITIS, ULCERATIVE COLITIS, CROHN S DISEASE AND CHRONIC PLAQUE PSORIASIS THE NOR-SWITCH STUDY
14 Feasibility The study has to be feasible should be part of daily clinical practice. Therefore, design and data collection must not be too complicated or comprehensive However, the study should be performed with a high quality from both the scientific and regulatory perspective
15 Objectives Primary: To assess if CT-P13 is non-inferior to innovator infliximab (INX) with regard to disease worsening in patients who have been on stable INX treatment for at least 6 months Secondary: To assess the safety and immunogenicity of CT-P13 compared to INX on patients who have been on stable INX treatment for at least 6 months To compare the efficacy of CT-P13 to INX in patients who have been on stable INX treatment for at least 6 months applying generic and disease-specific outcome measures Exploratory: To compare cost-effectiveness of CT-P13 to INX in patients who have been on stable INX treatment for at least 6 months
16 Patient population Patients RA, SpA inkl PsA IBD Psoriasis Adults on stable treatment with Remicade for at least 6 mnd Ethics Will patients on successful treatment be willing to be randomized? What will be the alternative treatment for patients who decline to participate (continue Remicade or switch to Remsima to reduce costs assuming that the drugs are similar)
17 Inclusion criteria A clinical diagnosis of either rheumatoid arthritis, spondyloarthritis, psoriatic arthritis, ulcerative colitis, Crohn s disease or psoriasis Male or non-pregnant, non-nursing female >18 years of age at screening Stable treatment of innovator infliximab (Remicade) during the last 6 months Subject capable of understanding and signing an informed consent form Provision of written informed consent
18 Exclusions Major co-morbidities, such as severe malignancies, severe diabetic mellitus, severe infections, uncontrollable hypertension, severe cardiovascular disease (NYHA class 3 or 4) and/or severe respiratory diseases Change of major co-medication during the last 2 months: RA, SpA and PsA: Initiation of systemic corticosteroids or synthetic DMARDs or other medication which according to the investigator would interfere with the stability of the disease. UC and CD: Initiation of systemic corticosteroids or an immunosuppressant or other medication which according to the investigator would interfere with the stability of the disease Psoriasis: Initiation of synthetic DMARDs or other medication which according to the investigator would interfere with the stability of the disease Inadequate birth control, pregnancy, and/or breastfeeding Psychiatric or mental disorders, alcohol abuse or other substance abuse, language barriers or other factors which makes adherence to the study protocol impossible Change in treatment with innovator infliximab (Remicade) during the last 6 months due to disease related factors
19 Medication- dosage and drug administration Biosimilar infliximab (Remsima or Inflectra): CT-P13 The dose of both drugs (Remicade or CT-P13) will be identical to the dose of innovator infliximab (Remicade) before randomization. Both drugs will be administered as intravenous infusions.
20 Schedule Modifications The main rule is that treatment will continue on the same dose and the same infusion intervals during the 52-week follow-up period. However changes in dose and interval may be permitted due to non disease related factors such as intercurrent infections, surgery, vacation, etc at the discretion of the treating physician. Drug levels will be measured at each assessment, but only reported on request. Changes in dose/interval based on drugs levels is not part of the treatment regimen.
21 Design RCT 1:1 (stratification according to disease, co-medication, other factors ) Blinding procedures Primary endpoint Occurrence of disease worsening during the 52-week study period based on disease specific efficacy assessment scores Several secondary study endpoints Follow-up 12 months Biobank
22 Endpoints Primary endpoint: Occurrence of disease worsening during the 52-week study period based on disease specific efficacy assessment scores Secondary endpoints: Generic: Time from randomization to disease worsening Patient and Physician Global assessment of disease activity Occurrence of drug discontinuation Time from randomization to drug discontinuation Disease-specific: Inflammation assessed by biochemical parameters RA and PsA: DAS28, M-HAQ, RAID, PsAID SpA: ASDAS, MHAQ, BASDAI Psoriasis: PASI, DLQI UC: Partial Mayo score, IBDQ CD: HBI, IBDQ Exploratory endpoints: EQ-5D SF-36 WPAI-GH Use of health care resources
23 Disease worsening Disease worsening in RA and PsA A disease worsening in RA and PsA is defined as an increase in DAS of 1.2 from randomization, a minimum DAS score of 1.6 Disease worsening in SpA A disease worsening in AS/SpA is defined as an increase in ASDAS of 1.1 from randomization Disease worsening in ulcerative colitis A disease worsening in ulcerative colitis is defined as an increase in partial Mayo score of 3 points from randomization and a minimum partial Mayo score of 5 points. Disease worsening in Crohn s disease A disease worsening in Crohn s disease is defined as an increase in HBI of 4 points from randomization and a minimum HBI score of 7 points. Disease worsening in psoriasis A disease worsening in psoriasis is defined as an increase in PASI of 3 points from randomization and a minimum PASI score of 5. Patient and investigator consensus on disease worsening If a patient does not fulfill any of the formal definitions above, experienced clinically significant worsening according to both the investigator and patient will be considered as fulfillment of the primary endpoint,but recorded separately in the CRF.
24 6. 2. Discontinuation Criteria for Patient Discontinuation Patients may be discontinued from study treatment and assessments at any time. Specific reasons for discontinuing a patient for this study are: Voluntary discontinuation by the patient who is at any time free to discontinue his/her participation in the study, without prejudice to further treatment. Safety reason as judged by the Principal Investigator Major protocol deviation Incorrect enrolment ie, the patient does not meet the required inclusion/exclusion criteria for the study Patient lost to follow-up A female patient becoming pregnant Major disease progression Deterioration in the patients condition which in the opinion of the Principal Investigator warrants study medication discontinuation (to be records as an AE or under Investigator Discretion) Patient s non-compliance to study treatment and/or procedures
25 Statistics/power Non-inferiority Primary endpoint estimated to occur in 30% during 52 weeks Equivalence criteria + 15% Power 90% 500 patients to be randomized (20% withdrawals)
26 Table 1: The numbers in the cells represent the total number of patients needed in total. All calculations are based on a power of 80% and alpha 2.5% Noninferiority Margin 10% disease worsening at 48 w 20% disease worsening at 48 w 30% disease worsening at 48 w 10% % % Table 2: The numbers in the cells represent the total number of patients needed in total. All calculations are based on a power of 90% and alpha 2.5%. Noninferiority Margin 10% disease worsening at 48 w 20% disease worsening at 48 w 30% disease worsening at 48 w 10% % %
27 Organisation Coordinating center: Dept of Rheumatology, Diakonhjemmet Hospital (on behalf of the South-East Health region authority) Multidisciplinary and multiregional project group with special competence in statistics (power calculations), immunogenicity, health economic evaluations, performance of strategy trials. Representation from NorCRIN Representation from the three relevant patient organisations Working group with a dermatologist, gastroenterologist, rheumatologist, statistician and expert in strategy studies (TKK, leader) The project will have a dedicated project coordinator and also three young fellows supporting the project (and being involved in publishing the results). Electronic CRF Monitoring of the study will most likely be performed by the regional health authorities
28 Conclusions Access for patients to biological disease-modifying antirheumatic drugs (bdmards) is strongly correlated to the economy in different countries. It can be expected that lower price of bdmards will improve accessibility and lead to better treatment. The availability of biosimilar infliximab (Remsima/Inflectra) is a new opportunity for lowering the cost of treatment with bdmards. Biosimilar infliximab (Remsima and Inflectra) was approved by the European Medicines Agency (EMA) and the Norwegian health authorities in the fall In the Norwegian tender system Orion Pharma offered a discount of 39% compared to the price of Remicade whereas Hospira offered a discount of 33%. Thus, Remsima is the preferred infliximab in 2014 based on this price system. Safety, efficacy and immunogenicity related to switching from Remicade to CT- P13 (Remsima/Inflectra) will be explored in a govermental supported study in Norway designed as a non-inferiority RCT.
April 2016. Guro Løvik Goll, MD PhD. Prosjektkoordinator revmatologi, Nor-Switch
 April 2016 Guro Løvik Goll, MD PhD Prosjektkoordinator revmatologi, Nor-Switch LIS TNF/BIOrecommendations 2015 http://www.lisnorway.no/ LIS TNF/BIO spesialistgruppe januar 2015 LIS 2015 Hospira offered
April 2016 Guro Løvik Goll, MD PhD Prosjektkoordinator revmatologi, Nor-Switch LIS TNF/BIOrecommendations 2015 http://www.lisnorway.no/ LIS TNF/BIO spesialistgruppe januar 2015 LIS 2015 Hospira offered
Current Rheumatoid Arthritis Treatment Options: Update for Managed Care and Specialty Pharmacists
 Current Rheumatoid Arthritis Treatment Options: Update for Managed Care and Specialty Pharmacists 1. Which of the following matches of biologic targets that contribute to rheumatoid arthritis (RA) and
Current Rheumatoid Arthritis Treatment Options: Update for Managed Care and Specialty Pharmacists 1. Which of the following matches of biologic targets that contribute to rheumatoid arthritis (RA) and
BEDFORDSHIRE AND LUTON JOINT PRESCRIBING COMMITTEE (JPC)
 BEDFORDSHIRE AND LUTON JOINT PRESCRIBING COMMITTEE (JPC) September 2014 Review date: September 2017 Bulletin 203: Tocilizumab (subcutaneous) in combination with methotrexate or as monotherapy for the treatment
BEDFORDSHIRE AND LUTON JOINT PRESCRIBING COMMITTEE (JPC) September 2014 Review date: September 2017 Bulletin 203: Tocilizumab (subcutaneous) in combination with methotrexate or as monotherapy for the treatment
MEDICAL ASSISTANCE HANDBOOK PRIOR AUTHORIZATION OF PHARMACEUTICAL SERVICES `I. Requirements for Prior Authorization of Cytokine and CAM Antagonists
 MEDICAL ASSISTANCE HBOOK `I. Requirements for Prior Authorization of Cytokine and CAM Antagonists A. Prescriptions That Require Prior Authorization All prescriptions for Cytokine and CAM Antagonists must
MEDICAL ASSISTANCE HBOOK `I. Requirements for Prior Authorization of Cytokine and CAM Antagonists A. Prescriptions That Require Prior Authorization All prescriptions for Cytokine and CAM Antagonists must
SYNOPSIS. 2-Year (0.5 DB + 1.5 OL) Addendum to Clinical Study Report
 Name of Sponsor/Company: Bristol-Myers Squibb Name of Finished Product: Abatacept () Name of Active Ingredient: Abatacept () Individual Study Table Referring to the Dossier (For National Authority Use
Name of Sponsor/Company: Bristol-Myers Squibb Name of Finished Product: Abatacept () Name of Active Ingredient: Abatacept () Individual Study Table Referring to the Dossier (For National Authority Use
Effectiveness and Drug Adherence in Rheumatoid Arthritis Patients on Biologic Monotherapy: A prospective observational study in Southern Sweden
 Effectiveness and Drug Adherence in Rheumatoid Arthritis Patients on Biologic Monotherapy: A prospective observational study in Southern Sweden Collaborators: Primary investigators Dr Lars Erik Kristensen,
Effectiveness and Drug Adherence in Rheumatoid Arthritis Patients on Biologic Monotherapy: A prospective observational study in Southern Sweden Collaborators: Primary investigators Dr Lars Erik Kristensen,
Biologic Treatments for Rheumatoid Arthritis
 Biologic Treatments Rheumatoid Arthritis (also known as cytokine inhibitors, TNF inhibitors, IL 1 inhibitor, or Biologic Response Modifiers) Description Biologics are new class of drugs that have been
Biologic Treatments Rheumatoid Arthritis (also known as cytokine inhibitors, TNF inhibitors, IL 1 inhibitor, or Biologic Response Modifiers) Description Biologics are new class of drugs that have been
Psoriasis, Incidence, Quality of Life, Psoriatic Arthritis, Prevalence
 1.0 Abstract Title Prevalence and Incidence of Articular Symptoms and Signs Related to Psoriatic Arthritis in Patients with Psoriasis Severe or Moderate with Adalimumab Treatment (TOGETHER). Keywords Psoriasis,
1.0 Abstract Title Prevalence and Incidence of Articular Symptoms and Signs Related to Psoriatic Arthritis in Patients with Psoriasis Severe or Moderate with Adalimumab Treatment (TOGETHER). Keywords Psoriasis,
ustekinumab 45mg solution for injection in pre-filled syringe (Stelara ) SMC No. (944/14) Janssen-Cilag Ltd
 ustekinumab 45mg solution for injection in pre-filled syringe (Stelara ) SMC No. (944/14) Janssen-Cilag Ltd 07 February 2014 The Scottish Medicines Consortium (SMC) has completed its assessment of the
ustekinumab 45mg solution for injection in pre-filled syringe (Stelara ) SMC No. (944/14) Janssen-Cilag Ltd 07 February 2014 The Scottish Medicines Consortium (SMC) has completed its assessment of the
Sponsor Novartis. Generic Drug Name Secukinumab. Therapeutic Area of Trial Psoriasis. Approved Indication investigational
 Clinical Trial Results Database Page 2 Sponsor Novartis Generic Drug Name Secukinumab Therapeutic Area of Trial Psoriasis Approved Indication investigational Clinical Trial Results Database Page 3 Study
Clinical Trial Results Database Page 2 Sponsor Novartis Generic Drug Name Secukinumab Therapeutic Area of Trial Psoriasis Approved Indication investigational Clinical Trial Results Database Page 3 Study
A Phase 2 Study of Interferon Beta-1a (Avonex ) in Ulcerative Colitis
 A Phase 2 Study of (Avonex ) in Ulcerative Colitis - Study Results - ClinicalTrials.gov A Phase 2 Study of (Avonex ) in Ulcerative Colitis This study has been completed. Sponsor: Biogen Idec Information
A Phase 2 Study of (Avonex ) in Ulcerative Colitis - Study Results - ClinicalTrials.gov A Phase 2 Study of (Avonex ) in Ulcerative Colitis This study has been completed. Sponsor: Biogen Idec Information
1.0 Abstract. Title: Real Life Evaluation of Rheumatoid Arthritis in Canadians taking HUMIRA. Keywords. Rationale and Background:
 1.0 Abstract Title: Real Life Evaluation of Rheumatoid Arthritis in Canadians taking HUMIRA Keywords Rationale and Background: This abbreviated clinical study report is based on a clinical surveillance
1.0 Abstract Title: Real Life Evaluation of Rheumatoid Arthritis in Canadians taking HUMIRA Keywords Rationale and Background: This abbreviated clinical study report is based on a clinical surveillance
Phenotypes and Classification of Psoriasis
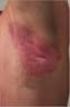 Rheumatology 2010 Birmingham 21 April 2010 Phenotypes and Classification of Psoriasis Christopher E.M. Griffiths Abbott Centocor Incyte Galderma Janssen-Cilag Leo Pharma Lynxx Novartis Pfizer Schering-Plough
Rheumatology 2010 Birmingham 21 April 2010 Phenotypes and Classification of Psoriasis Christopher E.M. Griffiths Abbott Centocor Incyte Galderma Janssen-Cilag Leo Pharma Lynxx Novartis Pfizer Schering-Plough
PROTOCOL SYNOPSIS Evaluation of long-term opioid efficacy for chronic pain
 P a g e 1 PROTOCOL SYNOPSIS Evaluation of long-term opioid efficacy for chronic pain Clinical Phase 4 Study Centers Study Period 25 U.S. sites identified and reviewed by the Steering Committee and Contract
P a g e 1 PROTOCOL SYNOPSIS Evaluation of long-term opioid efficacy for chronic pain Clinical Phase 4 Study Centers Study Period 25 U.S. sites identified and reviewed by the Steering Committee and Contract
How To Choose A Biologic Drug
 North Carolina Rheumatology Association Position Statements I. Biologic Agents A. Appropriate delivery, handling, storage and administration of biologic agents B. Indications for biologic agents II. III.
North Carolina Rheumatology Association Position Statements I. Biologic Agents A. Appropriate delivery, handling, storage and administration of biologic agents B. Indications for biologic agents II. III.
Shared care protocol for the management of patients with Rheumatoid Arthritis treated with disease modifying antirheumatic drugs (DMARDs)
 Tameside Hospital NHS Foundation Trust and NHS Tameside and Glossop Shared care protocol for the management of patients with Rheumatoid Arthritis treated with disease modifying antirheumatic drugs (DMARDs)
Tameside Hospital NHS Foundation Trust and NHS Tameside and Glossop Shared care protocol for the management of patients with Rheumatoid Arthritis treated with disease modifying antirheumatic drugs (DMARDs)
Immune modulation in rheumatology. Geoff McColl University of Melbourne/Australian Rheumatology Association
 Immune modulation in rheumatology Geoff McColl University of Melbourne/Australian Rheumatology Association A traditional start to a presentation on biological agents in rheumatic disease is Plasma cell
Immune modulation in rheumatology Geoff McColl University of Melbourne/Australian Rheumatology Association A traditional start to a presentation on biological agents in rheumatic disease is Plasma cell
Version History. Previous Versions. Drugs for MS.Drug facts box fingolimod Version 1.0 Author
 Version History Policy Title Drugs for MS.Drug facts box fingolimod Version 1.0 Author West Midlands Commissioning Support Unit Publication Date Jan 2013 Review Date Supersedes/New (Further fields as required
Version History Policy Title Drugs for MS.Drug facts box fingolimod Version 1.0 Author West Midlands Commissioning Support Unit Publication Date Jan 2013 Review Date Supersedes/New (Further fields as required
påçííáëü=jéçáåáåéë=`çåëçêíáìã==
 påçííáëü=jéçáåáåéë=`çåëçêíáìã== adalimumab 40mg pre-filled syringe for subcutaneous injection (Humira ) No. (218/05) Abbott New indication: treatment of active and progressive psoriatic arthritis in adults
påçííáëü=jéçáåáåéë=`çåëçêíáìã== adalimumab 40mg pre-filled syringe for subcutaneous injection (Humira ) No. (218/05) Abbott New indication: treatment of active and progressive psoriatic arthritis in adults
to Part of Dossier: Name of Active Ingredient: Title of Study: Quality of life study with adalimumab in rheumatoid arthritis. ESCALAR.
 2.0 Synopsis Abbott Laboratories Name of Study Drug: Individual Study Table Referring to Part of Dossier: Adalimumab (HUMIRA) (For National Authority Use Only) Name of Active Ingredient: Adalimumab Title
2.0 Synopsis Abbott Laboratories Name of Study Drug: Individual Study Table Referring to Part of Dossier: Adalimumab (HUMIRA) (For National Authority Use Only) Name of Active Ingredient: Adalimumab Title
Summary of the risk management plan (RMP) for Otezla (apremilast)
 EMA/741412/2014 Summary of the risk management plan (RMP) for Otezla (apremilast) This is a summary of the risk management plan (RMP) for Otezla, which details the measures to be taken in order to ensure
EMA/741412/2014 Summary of the risk management plan (RMP) for Otezla (apremilast) This is a summary of the risk management plan (RMP) for Otezla, which details the measures to be taken in order to ensure
Etanercept, infliximab and adalimumab for the treatment of psoriatic arthritis
 Etanercept, infliximab and adalimumab for the treatment of Issued: August 2010 guidance.nice.org.uk/ta199 NICE has accredited the process used by the Centre for Health Technology Evaluation at NICE to
Etanercept, infliximab and adalimumab for the treatment of Issued: August 2010 guidance.nice.org.uk/ta199 NICE has accredited the process used by the Centre for Health Technology Evaluation at NICE to
Subject: No. Page PROTOCOL AND CASE REPORT FORM DEVELOPMENT AND REVIEW Standard Operating Procedure
 703 1 of 11 POLICY The Beaumont Research Coordinating Center (BRCC) will provide advice to clinical trial investigators on protocol development, content and format. Upon request, the BRCC will review a
703 1 of 11 POLICY The Beaumont Research Coordinating Center (BRCC) will provide advice to clinical trial investigators on protocol development, content and format. Upon request, the BRCC will review a
Understanding and improving communications between people with rheumatoid arthritis and their healthcare professionals. RCUKCOMM00116w February 2014
 Understanding and improving communications between people with rheumatoid arthritis and their healthcare professionals RCUKCOMM00116w February 2014 This project has been funded by Roche Products Ltd &
Understanding and improving communications between people with rheumatoid arthritis and their healthcare professionals RCUKCOMM00116w February 2014 This project has been funded by Roche Products Ltd &
Understanding Rheumatoid Arthritis
 Understanding Rheumatoid Arthritis Understanding Rheumatoid Arthritis What Is Rheumatoid Arthritis? 1,2 Rheumatoid arthritis (RA) is a chronic autoimmune disease. It causes joints to swell and can result
Understanding Rheumatoid Arthritis Understanding Rheumatoid Arthritis What Is Rheumatoid Arthritis? 1,2 Rheumatoid arthritis (RA) is a chronic autoimmune disease. It causes joints to swell and can result
NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE. Health Technology Appraisal
 NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE Health Technology Appraisal Adalimumab, etanercept, infliximab, rituximab and abatacept for the treatment of rheumatoid arthritis after the failure
NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE Health Technology Appraisal Adalimumab, etanercept, infliximab, rituximab and abatacept for the treatment of rheumatoid arthritis after the failure
4.1 Objectives of Clinical Trial Assessment
 L1 4.1 Objectives of Clinical Trial Assessment Presentation to APEC Preliminary Workshop on Review of Drug Development in Clinical Trials Celia Lourenco, PhD, Manager, Clinical Group I Office of Clinical
L1 4.1 Objectives of Clinical Trial Assessment Presentation to APEC Preliminary Workshop on Review of Drug Development in Clinical Trials Celia Lourenco, PhD, Manager, Clinical Group I Office of Clinical
SECTION 3. Criteria for Special Authorization of Select Drug Products. Section 3 Criteria for Special Authorization of Select Drug Products
 SECTION 3 Criteria for Special Authorization of Select Drug Products Section 3 Criteria for Special Authorization of Select Drug Products CRITERIA FOR SPECIAL AUTHORIZATION OF SELECT DRUG PRODUCTS The
SECTION 3 Criteria for Special Authorization of Select Drug Products Section 3 Criteria for Special Authorization of Select Drug Products CRITERIA FOR SPECIAL AUTHORIZATION OF SELECT DRUG PRODUCTS The
REFERENCE CODE GDHC503DFR PUBLICAT ION DATE DECEMBER 2014 METHOTREXATE (RHEUMATOID ARTHRITIS) - FORECAST AND MARKET ANALYSIS TO 2023
 REFERENCE CODE GDHC503DFR PUBLICAT ION DATE DECEMBER 2014 METHOTREXATE (RHEUMATOID ARTHRITIS) - Executive Summary The table below provides the key metrics for Methotrexate in the 10MM (US, France, Germany,
REFERENCE CODE GDHC503DFR PUBLICAT ION DATE DECEMBER 2014 METHOTREXATE (RHEUMATOID ARTHRITIS) - Executive Summary The table below provides the key metrics for Methotrexate in the 10MM (US, France, Germany,
Patient Input Information Clinical Trials Outcomes Common Drug Review
 CDEC FINAL RECOMMENDATION USTEKINUMAB (Stelara Janssen Inc.) Indication: Psoriatic Arthritis Recommendation: The Canadian Drug Expert Committee (CDEC) recommends that ustekinumab not be listed at the submitted
CDEC FINAL RECOMMENDATION USTEKINUMAB (Stelara Janssen Inc.) Indication: Psoriatic Arthritis Recommendation: The Canadian Drug Expert Committee (CDEC) recommends that ustekinumab not be listed at the submitted
Morgan Schultz 1, Stephanie Keeling 2, Steven Katz 2, Walter Maksymowych 2, Dean Eurich 3, Jill Hall 1 1
 Morgan Schultz 1, Stephanie Keeling 2, Steven Katz 2, Walter Maksymowych 2, Dean Eurich 3, Jill Hall 1 1 Faculty of Pharmacy and Pharmaceutical Sciences, 2 Faciluty of Medicine and Dentistry, 3 School
Morgan Schultz 1, Stephanie Keeling 2, Steven Katz 2, Walter Maksymowych 2, Dean Eurich 3, Jill Hall 1 1 Faculty of Pharmacy and Pharmaceutical Sciences, 2 Faciluty of Medicine and Dentistry, 3 School
Patient Assistance Application for HUMIRA (adalimumab)
 The AbbVie Patient Assistance Foundation provides AbbVie medicines at no cost to patients experiencing financial difficulties. Eligible patients typically have no healthcare coverage for the requested
The AbbVie Patient Assistance Foundation provides AbbVie medicines at no cost to patients experiencing financial difficulties. Eligible patients typically have no healthcare coverage for the requested
New Evidence reports on presentations given at EULAR 2012. Rituximab for the Treatment of Rheumatoid Arthritis
 New Evidence reports on presentations given at EULAR 2012 Rituximab for the Treatment of Rheumatoid Arthritis Report on EULAR 2012 presentations Long-term safety of rituximab: 10-year follow-up in the
New Evidence reports on presentations given at EULAR 2012 Rituximab for the Treatment of Rheumatoid Arthritis Report on EULAR 2012 presentations Long-term safety of rituximab: 10-year follow-up in the
Sponsor Novartis Pharmaceuticals
 Clinical Trial Results Database Page 1 Sponsor Novartis Pharmaceuticals Generic Drug Name Indacaterol Therapeutic Area of Trial Chronic Obstructive Pulmonary Disease (COPD) Indication studied: COPD Study
Clinical Trial Results Database Page 1 Sponsor Novartis Pharmaceuticals Generic Drug Name Indacaterol Therapeutic Area of Trial Chronic Obstructive Pulmonary Disease (COPD) Indication studied: COPD Study
Media Release. Basel, 11 June 2009. RA patients with enhanced response identified
 Media Release Basel, 11 June 2009 New data demonstrate the ability of MabThera to reduce the progression of joint damage when used as a first-line biologic treatment in rheumatoid arthritis RA patients
Media Release Basel, 11 June 2009 New data demonstrate the ability of MabThera to reduce the progression of joint damage when used as a first-line biologic treatment in rheumatoid arthritis RA patients
Improvement in Quality of Life of Rheumatoid Arthritis Patients on Biologic Therapy
 Improvement in Quality of Life of Rheumatoid Arthritis Patients on Biologic Therapy R Adams 1, Ct Ng 2, A Gibbs 2, L Tilson 1, D Veale 2, B Bresnihan 2, O FitzGerald 2, M Barry 1 1. National Centre for
Improvement in Quality of Life of Rheumatoid Arthritis Patients on Biologic Therapy R Adams 1, Ct Ng 2, A Gibbs 2, L Tilson 1, D Veale 2, B Bresnihan 2, O FitzGerald 2, M Barry 1 1. National Centre for
CLINICAL RESEARCH PROTOCOL CHECKLIST
 CLINICAL RESEARCH PROTOCOL CHECKLIST [taken from ICH GCP : Guidance for Industry, Good Clinical Practice: Consolidated Guidance, Revision 1 (R1) June 1996] ICH GCP, Section 6. CLINICAL TRIAL PROTOCOL AND
CLINICAL RESEARCH PROTOCOL CHECKLIST [taken from ICH GCP : Guidance for Industry, Good Clinical Practice: Consolidated Guidance, Revision 1 (R1) June 1996] ICH GCP, Section 6. CLINICAL TRIAL PROTOCOL AND
Recommendations for Early RA Patients
 SUPPLEMENTARY APPENDIX 5: Executive summary of recommendations for patients with early RA, established RA, and high-risk comorbidities Recommendations for Early RA Patients We strongly recommend using
SUPPLEMENTARY APPENDIX 5: Executive summary of recommendations for patients with early RA, established RA, and high-risk comorbidities Recommendations for Early RA Patients We strongly recommend using
Guideline for the use of Biological Therapies in the Treatment of Psoriasis
 1 Date of Production: March1 st 2011 Date of 1 st review: July 10 th 2015 Date for next review: March1 st 2018 Local Contact Dermatology Consultant Shanti Ayob Patient group to which this applies: Patients
1 Date of Production: March1 st 2011 Date of 1 st review: July 10 th 2015 Date for next review: March1 st 2018 Local Contact Dermatology Consultant Shanti Ayob Patient group to which this applies: Patients
RHEUMATOID ARTHRITIS. Dr Bruce Kirkham Rheumatology Clinical Lead
 RHEUMATOID ARTHRITIS Dr Bruce Kirkham Rheumatology Clinical Lead RHEUMATOID ARTHRITIS (RA) RA is a common disease: 0.8 per cent of the population RA more common in females: female to male ratio 3:1 RA
RHEUMATOID ARTHRITIS Dr Bruce Kirkham Rheumatology Clinical Lead RHEUMATOID ARTHRITIS (RA) RA is a common disease: 0.8 per cent of the population RA more common in females: female to male ratio 3:1 RA
Pain Management Regulations Affect More Than Pain Management Specialists January 2012. Of counsel to
 Pain Management Regulations Affect More Than Pain Management Specialists January 2012 LINDA A. KEEN MSN, JD, LHCRM LAW OFFICE OF LINDA A. KEEN P.A. TALLAHASSEE, FL Of counsel to Pain Management Regulations
Pain Management Regulations Affect More Than Pain Management Specialists January 2012 LINDA A. KEEN MSN, JD, LHCRM LAW OFFICE OF LINDA A. KEEN P.A. TALLAHASSEE, FL Of counsel to Pain Management Regulations
Arthritis and Rheumatology Clinics of Kansas Patient Education. Reactive Arthritis (ReA) / Inflammatory Bowel Disease (IBD) Arthritis
 Arthritis and Rheumatology Clinics of Kansas Patient Education Reactive Arthritis (ReA) / Inflammatory Bowel Disease (IBD) Arthritis Introduction: For as long as scientists have studied rheumatic disease,
Arthritis and Rheumatology Clinics of Kansas Patient Education Reactive Arthritis (ReA) / Inflammatory Bowel Disease (IBD) Arthritis Introduction: For as long as scientists have studied rheumatic disease,
Original Policy Date
 MP 5.01.20 Tysabri (natalizumab) Medical Policy Section Prescription Drug Issue 12:2013 Original Policy Date 12:2013 Last Review Status/Date Local Policy/12:2013 Return to Medical Policy Index Disclaimer
MP 5.01.20 Tysabri (natalizumab) Medical Policy Section Prescription Drug Issue 12:2013 Original Policy Date 12:2013 Last Review Status/Date Local Policy/12:2013 Return to Medical Policy Index Disclaimer
Guidelines for the Use of Controlled Substances in the Treatment of Pain Adopted by the New Hampshire Medical Society, July 1998
 Guidelines for the Use of Controlled Substances in the Treatment of Pain Adopted by the New Hampshire Medical Society, July 1998 Section I: Preamble The New Hampshire Medical Society believes that principles
Guidelines for the Use of Controlled Substances in the Treatment of Pain Adopted by the New Hampshire Medical Society, July 1998 Section I: Preamble The New Hampshire Medical Society believes that principles
Biologics: Specific Drug Safety Challenges. Violetta B. Kyburz
 Biologics: Specific Drug Safety Challenges Violetta B. Kyburz 2012 2013 2014 Biologics: Specific Drug Safety Challenges Topics for discussion Particular issues in the preclinical development of biologics
Biologics: Specific Drug Safety Challenges Violetta B. Kyburz 2012 2013 2014 Biologics: Specific Drug Safety Challenges Topics for discussion Particular issues in the preclinical development of biologics
Information on Rheumatoid Arthritis
 Information on Rheumatoid Arthritis Table of Contents About Rheumatoid Arthritis 1 Definition 1 Signs and symptoms 1 Causes 1 Risk factors 1 Test and diagnosis 2 Treatment options 2 Lifestyle 3 References
Information on Rheumatoid Arthritis Table of Contents About Rheumatoid Arthritis 1 Definition 1 Signs and symptoms 1 Causes 1 Risk factors 1 Test and diagnosis 2 Treatment options 2 Lifestyle 3 References
Summary and general discussion
 Chapter 7 Summary and general discussion Summary and general discussion In this thesis, treatment of vitamin K antagonist-associated bleed with prothrombin complex concentrate was addressed. In this we
Chapter 7 Summary and general discussion Summary and general discussion In this thesis, treatment of vitamin K antagonist-associated bleed with prothrombin complex concentrate was addressed. In this we
Psoriasis Treatment Transition Pathway
 Psoriasis Treatment Transition Pathway A Treatment Support Tool Adapted from Circle Nottingham NHS Treatment Centre Psoriasis Pathway (under consultation) with support from Abbvie Ltd Treatment Pathways
Psoriasis Treatment Transition Pathway A Treatment Support Tool Adapted from Circle Nottingham NHS Treatment Centre Psoriasis Pathway (under consultation) with support from Abbvie Ltd Treatment Pathways
Early identification and treatment - the Norwegian perspective. Till Uhlig Dept of Rheumatology Diakonhjemmet Hospital Oslo, Norway
 Early identification and treatment - the Norwegian perspective Till Uhlig Dept of Rheumatology Diakonhjemmet Hospital Oslo, Norway Oslo Rheumatoid Arthritis Registry (ORAR) Very early Arthritis Clinic
Early identification and treatment - the Norwegian perspective Till Uhlig Dept of Rheumatology Diakonhjemmet Hospital Oslo, Norway Oslo Rheumatoid Arthritis Registry (ORAR) Very early Arthritis Clinic
biologics for the treatment of psoriasis
 How to contact us The Psoriasis Association Dick Coles House 2 Queensbridge Northampton NN4 7BF tel: 08456 760 076 (01604) 251 620 fax: (01604) 251 621 email: [email protected] www.psoriasis-association.org.uk
How to contact us The Psoriasis Association Dick Coles House 2 Queensbridge Northampton NN4 7BF tel: 08456 760 076 (01604) 251 620 fax: (01604) 251 621 email: [email protected] www.psoriasis-association.org.uk
Efficacy, safety and preference study of a insulin pen PDS290 vs. a Novo Nordisk marketed insulin pen in diabetics
 Efficacy, safety and preference study of a insulin pen PDS290 vs. a Novo Nordisk marketed insulin pen in diabetics This trial is conducted in the United States of America (USA). The aim of this clinical
Efficacy, safety and preference study of a insulin pen PDS290 vs. a Novo Nordisk marketed insulin pen in diabetics This trial is conducted in the United States of America (USA). The aim of this clinical
subcutaneous initially every 4 weeks then every 12 weeks Coverage Criteria: Express Scripts, Inc. monograph dated 02/24/2010
 BENEFIT DESCRIPTION AND LIMITATIONS OF COVERAGE ITEM: PRODUCT LINES: COVERED UNDER: DESCRIPTION: CPT/HCPCS Code: Company Supplying: Setting: Humira (adalimumab subcutaneous injection) Commercial HMO/PPO/CDHP
BENEFIT DESCRIPTION AND LIMITATIONS OF COVERAGE ITEM: PRODUCT LINES: COVERED UNDER: DESCRIPTION: CPT/HCPCS Code: Company Supplying: Setting: Humira (adalimumab subcutaneous injection) Commercial HMO/PPO/CDHP
Drugs for MS.Drug fact box cannabis extract (Sativex) Version 1.0 Author
 Version History Policy Title Drugs for MS.Drug fact box cannabis extract (Sativex) Version 1.0 Author West Midlands Commissioning Support Unit Publication Date Jan 2013 Review Date Supersedes/New (Further
Version History Policy Title Drugs for MS.Drug fact box cannabis extract (Sativex) Version 1.0 Author West Midlands Commissioning Support Unit Publication Date Jan 2013 Review Date Supersedes/New (Further
PRODUCT INN REFERENCE AUTHORIZATION Omnitrope
 PRODUCT INN REFERENCE AUTHORIZATION Omnitrope Genotropin 12/04/2006 somatropin Valtropin (W/D) Humatrope 24/04/2006 Abseamed Binocrit Epoetina alfa hexal Retacrit Silapo Biograstim Filgrastim ratiopharm
PRODUCT INN REFERENCE AUTHORIZATION Omnitrope Genotropin 12/04/2006 somatropin Valtropin (W/D) Humatrope 24/04/2006 Abseamed Binocrit Epoetina alfa hexal Retacrit Silapo Biograstim Filgrastim ratiopharm
Appendix to Tennessee Department of Health: Tennessee Clinical Practice Guidelines for Outpatient Management of Chronic Non- Malignant Pain
 Appendix to Tennessee Department of Health: Tennessee Clinical Practice Guidelines for Outpatient Management of Chronic Non- Malignant Pain Division of Workers Compensation 04.01.2015 Background Opioids
Appendix to Tennessee Department of Health: Tennessee Clinical Practice Guidelines for Outpatient Management of Chronic Non- Malignant Pain Division of Workers Compensation 04.01.2015 Background Opioids
Evidence-based Management of Rheumatoid Arthritis (2009)
 CPLD reviews its distance learning programmes every twelve months to ensure currency. This update has been produced by an expert and should be read in conjunction with the Evidencebased Management of distance
CPLD reviews its distance learning programmes every twelve months to ensure currency. This update has been produced by an expert and should be read in conjunction with the Evidencebased Management of distance
Key Findings. Use this report to... The Autoimmune Market Outlook to 2013
 Key Findings The global autoimmune market generated sales of $31.9bn in, an increase of 14.4% over 2006 sales. The market is forecast to grow at a CAGR of 8.1% to reach a total value of $51.0bn in 2013.
Key Findings The global autoimmune market generated sales of $31.9bn in, an increase of 14.4% over 2006 sales. The market is forecast to grow at a CAGR of 8.1% to reach a total value of $51.0bn in 2013.
CAN-FITE BIOPHARMA LTD.
 UNITED STATES SECURITIES AND EXCHANGE COMMISSION Washington, D.C. 20549 FORM 6-K Report of Foreign Private Issuer Pursuant to Rule 13a-16 or 15d-16 Under the Securities Exchange Act of 1934 For the Month
UNITED STATES SECURITIES AND EXCHANGE COMMISSION Washington, D.C. 20549 FORM 6-K Report of Foreign Private Issuer Pursuant to Rule 13a-16 or 15d-16 Under the Securities Exchange Act of 1934 For the Month
HealthCare Partners of Nevada. Heart Failure
 HealthCare Partners of Nevada Heart Failure Disease Management Program 2010 HF DISEASE MANAGEMENT PROGRAM The HealthCare Partners of Nevada (HCPNV) offers a Disease Management program for members with
HealthCare Partners of Nevada Heart Failure Disease Management Program 2010 HF DISEASE MANAGEMENT PROGRAM The HealthCare Partners of Nevada (HCPNV) offers a Disease Management program for members with
Minimum Insurance Benefits for Patients with Opioid Use Disorder The Opioid Use Disorder Epidemic: The Evidence for Opioid Treatment:
 Minimum Insurance Benefits for Patients with Opioid Use Disorder By David Kan, MD and Tauheed Zaman, MD Adopted by the California Society of Addiction Medicine Committee on Opioids and the California Society
Minimum Insurance Benefits for Patients with Opioid Use Disorder By David Kan, MD and Tauheed Zaman, MD Adopted by the California Society of Addiction Medicine Committee on Opioids and the California Society
Version history Version number Version date Effective date 01 dd-mon-yyyy dd-mon-yyyy 02 dd-mon-yyyy dd-mon-yyyy 03 (current) dd-mon-yyyy dd-mon-yyyy
 Trial name: HOVON xxx yyy Sponsor: HOVON Version history Version number Version date Effective date 01 dd-mon-yyyy dd-mon-yyyy 02 dd-mon-yyyy dd-mon-yyyy 03 (current) dd-mon-yyyy dd-mon-yyyy QRMP authors
Trial name: HOVON xxx yyy Sponsor: HOVON Version history Version number Version date Effective date 01 dd-mon-yyyy dd-mon-yyyy 02 dd-mon-yyyy dd-mon-yyyy 03 (current) dd-mon-yyyy dd-mon-yyyy QRMP authors
Prior Authorization, Pharmacy and Health Case Management Information. Prior Authorization. Pharmacy Information. Health Case Management
 Prior Authorization, Pharmacy and Health Case Management Information The purpose of this information sheet is to provide you with details on how Great-West Life will be assessing and managing your claim
Prior Authorization, Pharmacy and Health Case Management Information The purpose of this information sheet is to provide you with details on how Great-West Life will be assessing and managing your claim
Prior Authorization, Pharmacy and Health Case Management Information. Prior Authorization. Pharmacy Information. Health Case Management
 Prior Authorization, Pharmacy and Health Case Management Information The purpose of this information sheet is to provide you with details on how Great-West Life will be assessing and managing your claim
Prior Authorization, Pharmacy and Health Case Management Information The purpose of this information sheet is to provide you with details on how Great-West Life will be assessing and managing your claim
Disease Therapy Management (DTM) Enhances Rheumatoid Arthritis Treatment
 A WHITE PAPER BY Disease Therapy Management (DTM) Enhances Rheumatoid Arthritis Treatment Increased adherence rates deliver improved outcomes for patients FALL 2010 The Benefits of DTM for Rheumatoid Arthritis
A WHITE PAPER BY Disease Therapy Management (DTM) Enhances Rheumatoid Arthritis Treatment Increased adherence rates deliver improved outcomes for patients FALL 2010 The Benefits of DTM for Rheumatoid Arthritis
Robert Okwemba, BSPHS, Pharm.D. 2015 Philadelphia College of Pharmacy
 Robert Okwemba, BSPHS, Pharm.D. 2015 Philadelphia College of Pharmacy Judith Long, MD,RWJCS Perelman School of Medicine Philadelphia Veteran Affairs Medical Center Background Objective Overview Methods
Robert Okwemba, BSPHS, Pharm.D. 2015 Philadelphia College of Pharmacy Judith Long, MD,RWJCS Perelman School of Medicine Philadelphia Veteran Affairs Medical Center Background Objective Overview Methods
Is Monotherapy Treatment of Etanercept Effective Against Plaque Psoriasis?
 Philadelphia College of Osteopathic Medicine DigitalCommons@PCOM PCOM Physician Assistant Studies Student Scholarship Student Dissertations, Theses and Papers 2011 Is Monotherapy Treatment of Etanercept
Philadelphia College of Osteopathic Medicine DigitalCommons@PCOM PCOM Physician Assistant Studies Student Scholarship Student Dissertations, Theses and Papers 2011 Is Monotherapy Treatment of Etanercept
In the last decade, there have been major changes in the
 233 Promising New Treatments for Rheumatoid Arthritis The Kinase Inhibitors Yusuf Yazici, M.D., and Alexandra L. Regens, B.A. Abstract Three major advances over the last decade have impacted the way we
233 Promising New Treatments for Rheumatoid Arthritis The Kinase Inhibitors Yusuf Yazici, M.D., and Alexandra L. Regens, B.A. Abstract Three major advances over the last decade have impacted the way we
COMMITTEE FOR PROPRIETARY MEDICINAL PRODUCTS (CPMP)
 The European Agency for the Evaluation of Medicinal Products Evaluation of Medicines for Human Use London, 17 December 2003 CPMP/EWP/556/95 rev 1/Final COMMITTEE FOR PROPRIETARY MEDICINAL PRODUCTS (CPMP)
The European Agency for the Evaluation of Medicinal Products Evaluation of Medicines for Human Use London, 17 December 2003 CPMP/EWP/556/95 rev 1/Final COMMITTEE FOR PROPRIETARY MEDICINAL PRODUCTS (CPMP)
Michigan Guidelines for the Use of Controlled Substances for the Treatment of Pain
 Michigan Guidelines for the Use of Controlled Substances for the Treatment of Pain Section I: Preamble The Michigan Boards of Medicine and Osteopathic Medicine & Surgery recognize that principles of quality
Michigan Guidelines for the Use of Controlled Substances for the Treatment of Pain Section I: Preamble The Michigan Boards of Medicine and Osteopathic Medicine & Surgery recognize that principles of quality
FastTest. You ve read the book... ... now test yourself
 FastTest You ve read the book...... now test yourself To ensure you have learned the key points that will improve your patient care, read the authors questions below. Please refer back to relevant sections
FastTest You ve read the book...... now test yourself To ensure you have learned the key points that will improve your patient care, read the authors questions below. Please refer back to relevant sections
Assessment of Medication Adherence in Rheumatoid Arthritis Patients
 J.J. Appl. Sci. Vol. 10, No. 2 (2008) Assessment of Medication Adherence in Rheumatoid Arthritis Patients Kholoud Z. Qoul, Ikbal N. Thuheerat & Imad Al-Dogham Royal Medical Services, Amman, Jordan Received:
J.J. Appl. Sci. Vol. 10, No. 2 (2008) Assessment of Medication Adherence in Rheumatoid Arthritis Patients Kholoud Z. Qoul, Ikbal N. Thuheerat & Imad Al-Dogham Royal Medical Services, Amman, Jordan Received:
Rheumatoid Arthritis: Diagnosis, Management and Monitoring
 Rheumatoid Arthritis: Diagnosis, Management and Monitoring Effective Date: September 30, 2012 Scope This guideline is intended to aid in early recognition, intervention and management of patients with
Rheumatoid Arthritis: Diagnosis, Management and Monitoring Effective Date: September 30, 2012 Scope This guideline is intended to aid in early recognition, intervention and management of patients with
Advancing research: a physician s guide to clinical trials
 Advancing research: a physician s guide to clinical trials Recruiting and retaining trial participants is one of the greatest obstacles to developing the next generation of Alzheimer s treatments Alzheimer
Advancing research: a physician s guide to clinical trials Recruiting and retaining trial participants is one of the greatest obstacles to developing the next generation of Alzheimer s treatments Alzheimer
Trial Description. Organizational Data. Secondary IDs
 Trial Description Title A randomized, double-blind, multicenter study to demonstrate equivalent efficacy and to compare safety and immunogenicity of a biosimilar etanercept (GP2015) and Enbrel in patients
Trial Description Title A randomized, double-blind, multicenter study to demonstrate equivalent efficacy and to compare safety and immunogenicity of a biosimilar etanercept (GP2015) and Enbrel in patients
Treatment of Severe Rheumatoid Arthritis
 Treatment of Severe Rheumatoid Arthritis Zhanguo Li Department of Rheumatology and Immunology, People s Hospital Beijing University Medical School, China Contents Background Challenges Treatment strategies
Treatment of Severe Rheumatoid Arthritis Zhanguo Li Department of Rheumatology and Immunology, People s Hospital Beijing University Medical School, China Contents Background Challenges Treatment strategies
Medicines for Psoriatic Arthritis. A Review of the Research for Adults
 Medicines for Psoriatic Arthritis A Review of the Research for Adults Is This Information Right for Me? Yes, this information is right for you if: Your doctor* has told you that you have psoriatic (pronounced
Medicines for Psoriatic Arthritis A Review of the Research for Adults Is This Information Right for Me? Yes, this information is right for you if: Your doctor* has told you that you have psoriatic (pronounced
Care Management Council submission date: August 2013. Contact Information
 Clinical Practice Approval Form Clinical Practice Title: Acute use of Buprenorphine for the Treatment of Opioid Dependence and Detoxification Type of Review: New Clinical Practice Revisions of Existing
Clinical Practice Approval Form Clinical Practice Title: Acute use of Buprenorphine for the Treatment of Opioid Dependence and Detoxification Type of Review: New Clinical Practice Revisions of Existing
GENERAL INFORMATION. Adverse Event (AE) Definition (ICH GUIDELINES E6 FOR GCP 1.2):
 Make copies of the blank SAE report form as needed. Retain originals with confirmation of all information faxed to DMID Pharmacovigilance Group Clinical Research Operations and Management Support (CROMS
Make copies of the blank SAE report form as needed. Retain originals with confirmation of all information faxed to DMID Pharmacovigilance Group Clinical Research Operations and Management Support (CROMS
DEPRESSION Depression Assessment PHQ-9 Screening tool Depression treatment Treatment flow chart Medications Patient Resource
 E-Resource March, 2015 DEPRESSION Depression Assessment PHQ-9 Screening tool Depression treatment Treatment flow chart Medications Patient Resource Depression affects approximately 20% of the general population
E-Resource March, 2015 DEPRESSION Depression Assessment PHQ-9 Screening tool Depression treatment Treatment flow chart Medications Patient Resource Depression affects approximately 20% of the general population
HEALTH CARE COSTS 11
 2 Health Care Costs Chronic health problems account for a substantial part of health care costs. Annually, three diseases, cardiovascular disease (including stroke), cancer, and diabetes, make up about
2 Health Care Costs Chronic health problems account for a substantial part of health care costs. Annually, three diseases, cardiovascular disease (including stroke), cancer, and diabetes, make up about
Prior Authorization, Pharmacy and Health Case Management Information. Prior Authorization. Pharmacy Information. Health Case Management
 Prior Authorization, Pharmacy and Health Case Management Information The purpose of this information sheet is to provide you with details on how Great-West Life will be assessing and managing your claim
Prior Authorization, Pharmacy and Health Case Management Information The purpose of this information sheet is to provide you with details on how Great-West Life will be assessing and managing your claim
Can Rheumatoid Arthritis treatment ever be stopped?
 Can Rheumatoid Arthritis treatment ever be stopped? Robert L. DiGiovanni, DO, FACOI Program Director Largo Medical Center Rheumatology Fellowship [email protected] Do not pour strange medicines
Can Rheumatoid Arthritis treatment ever be stopped? Robert L. DiGiovanni, DO, FACOI Program Director Largo Medical Center Rheumatology Fellowship [email protected] Do not pour strange medicines
ADULT HYPERTENSION PROTOCOL STANFORD COORDINATED CARE
 I. PURPOSE To establish guidelines for the monitoring of antihypertensive therapy in adult patients and to define the roles and responsibilities of the collaborating clinical pharmacist and pharmacy resident.
I. PURPOSE To establish guidelines for the monitoring of antihypertensive therapy in adult patients and to define the roles and responsibilities of the collaborating clinical pharmacist and pharmacy resident.
Early Rehabilitation of Rheumatoid Arthritis (RA)
 Early Rehabilitation of Rheumatoid Arthritis (RA) Results and Hands-On Experiences with the Implementation of a Randomized Controlled Trial in Health Services Research Susanne Schlademann University of
Early Rehabilitation of Rheumatoid Arthritis (RA) Results and Hands-On Experiences with the Implementation of a Randomized Controlled Trial in Health Services Research Susanne Schlademann University of
Biostat Methods STAT 5820/6910 Handout #6: Intro. to Clinical Trials (Matthews text)
 Biostat Methods STAT 5820/6910 Handout #6: Intro. to Clinical Trials (Matthews text) Key features of RCT (randomized controlled trial) One group (treatment) receives its treatment at the same time another
Biostat Methods STAT 5820/6910 Handout #6: Intro. to Clinical Trials (Matthews text) Key features of RCT (randomized controlled trial) One group (treatment) receives its treatment at the same time another
DOCTOR DISCUSSION GUIDE FOR RHEUMATOID ARTHRITIS
 DOCTOR DISCUSSION GUIDE FOR RHEUMATOID ARTHRITIS Talking your Doctor About Rheumatoid Arthritis Preparing for your Doctor s Appointment Early and aggressive treatment can help you forestall the joint damage
DOCTOR DISCUSSION GUIDE FOR RHEUMATOID ARTHRITIS Talking your Doctor About Rheumatoid Arthritis Preparing for your Doctor s Appointment Early and aggressive treatment can help you forestall the joint damage
FREEDOM C: A 16-Week, International, Multicenter, Double-Blind, Randomized, Placebo-Controlled Comparison of the Efficacy and Safety of Oral UT-15C
 FREEDOM C: A 16-Week, International, Multicenter, Double-Blind, Randomized, Placebo-Controlled Comparison of the Efficacy and Safety of Oral UT-15C SR in Combination with an ERA and/or a PDE-5 Inhibitor
FREEDOM C: A 16-Week, International, Multicenter, Double-Blind, Randomized, Placebo-Controlled Comparison of the Efficacy and Safety of Oral UT-15C SR in Combination with an ERA and/or a PDE-5 Inhibitor
Completing your Personal Health Application New York Applicants
 Completing your Personal Health Application New York Applicants Purpose These instructions will help you to complete your Personal Health Application. This will help ensure that your application is processed
Completing your Personal Health Application New York Applicants Purpose These instructions will help you to complete your Personal Health Application. This will help ensure that your application is processed
