Protein kinase C alpha expression and resistance to neo-adjuvant gemcitabine-containing chemotherapy in non-small cell lung cancer
|
|
|
- Ethel Walters
- 8 years ago
- Views:
Transcription
1 Protein kinase C alpha expression and resistance to neo-adjuvant gemcitabine-containing chemotherapy in non-small cell lung cancer Dan Vogl Lay Abstract Early stage non-small cell lung cancer can be cured with surgery alone in approximately 60% of cases. Trials are currently underway to evaluate whether giving chemotherapy before surgery improves that cure rate. Protein kinase C alpha is a cellular protein involved in regulating cell proliferation and may play a role in resistance of non-small cell lung cancer to chemotherapy. This trial will investigate whether increased levels of protein kinase C alpha are associated with resistance of lung cancer to initial chemotherapy. We are currently conducting a trial in which patients receive chemotherapy before undergoing surgical removal of early stage non-small cell lung cancer. Patients who are already enrolled in the chemotherapy study will be eligible for this study, in which we will compare levels of protein kinase C alpha in the biopsy specimens from before patients begin chemotherapy and the post-chemotherapy surgical specimens. Up to 70 adults will be enrolled in the chemotherapy study and will therefore be eligible for this study. Patients will already have provided informed consent for the chemotherapy trial and will provide written informed consent for their biopsies to be used in this study. The decision whether to participate in this study will not affect participation in the chemotherapy study. No additional procedures, treatments, or questionnaires will be used in this study. There are neither additional risks nor additional benefits associated with participation in this study, and the study presents no ethical problems. A. Study Purpose and Rationale Lung cancer is a major cause of morbidity and.mortality. Approximately 75% of primary lung malignancies are non-small cell lung cancer (NSCLC). When found at an early stage, without spread to the mediastinal lymph nodes, surgery can provide a cure for NSCLC, but even patients with the best clinical prognosis have at best a 67% 5-year survival rate (for pathological stage T1 NO) with surgical therapy. 1 Adjuvant, or postoperative treatment with chemotherapy, radiation, or both has yielded minimal to no benefit in recurrence or mortality rates. Randomized trials of preoperative, or neoadjuvant, chemotherapy in more advanced disease have shown improvements in cure and survival. 2,3,4 Early experience with neoadjuvant chemotherapy in stage I and II NSCLC has been encouraging, and phase III randomized trials are underway to better define the possible benefit of neoadjuvant chemotherapy compared with surgery alone. We are currently conducting a multi-center, randomized, phase 11 trial of gemcitabinecontaining neo-adjuvant regimens. The study has received IRB approval and has begun accruing patients with histologically confirmed clinical stage I and 11 non-small cell lung cancers. Patients will receive gemcitabine and either cisplatin or carboplatin in a 6-week regimen of chemotherapy prior to initial surgery. The primary endpoint is pathologic complete response, or the absence of identifiable tumor cells in the postchemotherapy surgical specimen, and based on prior studies of neo-adjuvant regimens, a pathologic complete response rate of at least 20% will indicate a regimen of sufficient efficacy to warrant comparison with surgery alone in a phase III trial. The study will enroll up to 70 patients at three centers depending on preliminary results. Given this expected pathologic complete response rate, we expect 80% Columbia University College of Physicians and Surgeons 97
2 of our patients to have residual tumor at the time of surgery, with demonstrable in vivo resistance to gemcitabine and platinum. Protein kinase C alpha (PKCα) is one of 13 isoforms of a family of serine threonine kinases, appears to play a major antiapoptotic role in lung cancer, and may have some role in resistance to chemotherapy. Protein kinase C alpha and beta over-expression in cell lines from human non-small cell lung cancers is associated with in vitro resistance to doxorubicin. 6 An antisense nucleotide that reduces expression of PKCα is currently in phase III trials in late-stage lung cancer. 7,8 It may also be appropriate to use in neo adjuvant regimens for early-stage cancer. The role of PKCα in resistance to gemcitabine containing regimens in early stage lung cancer is unclear. Evidence that PKCα is over-expressed in non-small cell lung cancer that is resistant to initial chemotherapy would strengthen the rationale for using anti-pkcα therapy in early stage lung cancer. The purpose of this study is to: 1) describe the level of expression of PKCα in prechemotherapy and post-chemotherapy biopsy specimens of non-small cell lung cancer; 2) determine the association between level of PKCα expression and response to chemotherapy with gemcitabine and platinum; and 3) determine whether PKCα expression increases in tumors that are resistant to induction chemotherapy. B. Study Design and Statistical Analysis We will attempt to enroll all patients who are in the parent trial. Patients will therefore have histologically confirmed clinical stage I or 11 non-small cell lung cancer; will not have received any prior chemotherapy or radiation therapy for non-small cell lung cancer; will not be receiving other investigational therapy; will be 18 years of age or older, with a Karnofsky Performance Status of ~:70 and adequate pre-treatment lung, hematologic, and renal function; and will have given informed consent to participate in the randomized trial of neo-adjuvant chemotherapy. The parent study will enroll up to seventy patients overall, with up to thirty-five in each arm, and the sample size for this study is therefore limited to seventy patients. We will describe the mean and standard deviation of relative expression of PKC(X on Western blot of pre-chemotherapy specimens and post-chemotherapy (surgical) specimens. We will assess the relationship between pre-chemotherapy level of PKCOC expression and pathologic complete response with Student's t-test. For patients without pathologic complete response, we will assess the change in PKC(X expression from prechemotherapy to post-chemotherapy specimens with a paired t-test. All p values will be two-tailed, and alpha will be Assuming a pathologic complete response rate of 15%, we will have approximately ten patients with pathologic complete response and sixty patients with residual tumor after chemotherapy. Our study will therefore have the ability (with a power of 80% and alpha = 0.05) to detect a difference of 0.97 standard deviations in initial PKCa levels between patients who achieve pathologic complete response and those who do not. Similarly, we will have the ability to detect a difference of 0.37 standard deviations between levels of PKCa in pre-chemotherapy and post-chemotherapy specimens in the sixty patients without a pathologic complete response. The mean and standard deviation of Western blot levels for PKCa in biopsy specimens of NSCLC are not known prior to this study. C. Study Procedure Biopsy specimens will be available from most patients prior to starting chemotherapy, and from all patients after chemotherapy. We will have access to all specimens from surgery done at Columbia-Presbyterian, the primary site for the original study, and we may have access to specimens from surgery done at other sites. Pathologic complete response is defined as the absence of viable tumor cells on light microscopy of routine sections in the pathology laboratory. We will perform a Western blot on all specimens to quantify the relative expression of PKCa. Columbia University College of Physicians and Surgeons 98
3 We will not perform any medical procedures in addition to those necessary for the parent trial. No additional follow-up will be necessary. D. Study Drugs This study will not involve any drugs in addition to those used in the parent trial. E. Medical Devices This study will not involve any investigational medical devices. F. Study Questionnaires G. Study Subjects We will not gather any additional information other than PKCα levels. Inclusion criteria: 1) Patients enrolled in the parent study with an evaluable pre-chemotherapy biopsy specimen will be eligible for inclusion in this study. 2) Patients must be able to provide informed consent for participation in this study. Inclusion criteria for the parent study: 1) Histologically confirmed non-small cell lung cancer 2) Stage I or 11 disease with a negative mediastinal evaluation (all mediastinal lymph nodes < 1 cm by CT scan and negative PET scan if available, with negative mediastinoscopy if either CT or PET is positive) and no signs or symptoms of systemic spread 3) No prior chemotherapy or radiation therapy for non-small cell lung cancer. No prior resection of lung disease within the five years before registration 4) Not receiving other investigational therapy 5) Karnofsky Performance Status 70 6) Age 18 years or older 7) Calculated post-resection FEV 1 > 40% predicted and calculated post-resection D L CO/V A > 40% predicted. 8) Absolute granulocyte count 1.5 x 10 9 /L and platelet count 120 x 10 9 /L 9) Serum creatinine within normal limits, or estimated or calculated creatinine clearance 65 ml/min 10) No prior malignancy, except treated basal cell or squamous cell skin cancer, in situ cervical cancer, other cancer from which the patient has been disease-free for 5 years or in which the stage and nature of malignancy is unlikely to affect 3-year survival 11) Able to give written informed consent 12) Suitable candidate for surgery Exclusion criteria for the parent study: 1) Stage III tumor or stage II tumor involving the superior sulcus 2) Post-obstructive pneumonia or other serious infection, serious medical condition, or prior allergic reactions to drugs containing cremophor 3) Pregnancy or nursing H. Recruitment of Subjects Columbia University College of Physicians and Surgeons 99
4 The primary investigator for the parent trial will approach patients regarding inclusion in this trial. I. Confidentiality of Study Data All biopsy specimens will be labeled with the patient's unique code number from the parent study and not with the patient's name or other identifying information. Biopsy specimens will be destroyed after the completion of the study. J. Potential Conflict of Interest There are no potential conflicts of interest. K. Location of the Study L. Potential Risks The study will take place in the Department of Oncology at CPIVIC. There are no additional potential risks to subjects as a result of participating in this trial. M. Potential Benefits There are no benefits to the subjects from participation in this trial. N. Alternative Therapies No therapy is provided as part of this trial, and the alternative is not participating in the trial. O. Compensation to Subjects No compensation will be provided. P. Costs to Subjects Subjects will not incur any additional costs as a result of participating in this study. Q. Minors as Research Subjects This study does not involve minors. R. Radiation or Radioactive Substances S. References: This study does not involve exposure of subjects to radiation. 1. Mountain CF. Revisions in the International System for Staging Lung Cancer. Chest 1997;111: Martini N, Kris MG, Flehinger BJ, et al. Preoperative Chemotherapy for Stage IlIa (N2) Lung Cancer: The Sloan-Kettering Experience with 136 patients. Ann Thorac Surg 1993;55: Columbia University College of Physicians and Surgeons 100
5 3. Rosell et al. A randomized trial comparing preoperative chemotherapy plus surgery with surgery alone in patients with non-small cell lung cancer. N Engl J Med 1994;330: Roth et al. A randomized trial comparing perioperative chemotherapy and surgery with surgery alone in resectable stage IIIA non-small-cell lung cancer. J Natl Cancer Inst 1995; 86: Pisters KMW, Ginsberg RJ, Firoux DJ, et al. Induction chemotherapy before surgery for early-stage lung cancer: a novel approach. J Thorac Cariovasc Surg 2000; 119: Volm M, Pornmerenke EW. Associated expression of protein kinase C with resistance to doxorubicin in human lung cancer. Anticancer Research 1995;15: Marcusson EG, Yacyshyn BR, Shanahan WR, Dean NM. Preclinical and clinical pharmacology of antisense oligonucleotides. Molecular Biotechnology 1999; 12: McKay RA, Miraglia LJ, Cummins LL, et al. Characterizqation of a potent and specific class of antisense oligonucleotide inhibitor of human protein kinase C-α expression. J Biol Chem 1999;274: Columbia University College of Physicians and Surgeons 101
ALCHEMIST (Adjuvant Lung Cancer Enrichment Marker Identification and Sequencing Trials)
 ALCHEMIST (Adjuvant Lung Cancer Enrichment Marker Identification and Sequencing Trials) 3 Integrated Trials Testing Targeted Therapy in Early Stage Lung Cancer Part of NCI s Precision Medicine Effort in
ALCHEMIST (Adjuvant Lung Cancer Enrichment Marker Identification and Sequencing Trials) 3 Integrated Trials Testing Targeted Therapy in Early Stage Lung Cancer Part of NCI s Precision Medicine Effort in
Adjuvant Therapy Non Small Cell Lung Cancer. Sunil Nagpal MD Director, Thoracic Oncology Jan 30, 2015
 Adjuvant Therapy Non Small Cell Lung Cancer Sunil Nagpal MD Director, Thoracic Oncology Jan 30, 2015 No Disclosures Number of studies Studies Per Month 12 10 8 6 4 2 0 1 2 3 4 5 6 7 8 9 10 11 12 1 2 3
Adjuvant Therapy Non Small Cell Lung Cancer Sunil Nagpal MD Director, Thoracic Oncology Jan 30, 2015 No Disclosures Number of studies Studies Per Month 12 10 8 6 4 2 0 1 2 3 4 5 6 7 8 9 10 11 12 1 2 3
Brigham and Women s Hospital, Boston, MA, USA; 2 Verastem, Inc., Boston, MA, USA
 Determination of Biomarker Response in a Phase II Window of Opportunity Study of Defactinib (VS 6063), a Focal Adhesion Kinase (FAK) Inhibitor, in Patients with Resectable Malignant Pleural Mesothelioma
Determination of Biomarker Response in a Phase II Window of Opportunity Study of Defactinib (VS 6063), a Focal Adhesion Kinase (FAK) Inhibitor, in Patients with Resectable Malignant Pleural Mesothelioma
GUIDELINES FOR THE MANAGEMENT OF LUNG CANCER
 GUIDELINES FOR THE MANAGEMENT OF LUNG CANCER BY Ali Shamseddine, MD (Coordinator); as04@aub.edu.lb Fady Geara, MD Bassem Shabb, MD Ghassan Jamaleddine, MD CLINICAL PRACTICE GUIDELINES FOR THE TREATMENT
GUIDELINES FOR THE MANAGEMENT OF LUNG CANCER BY Ali Shamseddine, MD (Coordinator); as04@aub.edu.lb Fady Geara, MD Bassem Shabb, MD Ghassan Jamaleddine, MD CLINICAL PRACTICE GUIDELINES FOR THE TREATMENT
Targeted Therapy What the Surgeon Needs to Know
 Targeted Therapy What the Surgeon Needs to Know AATS Focus in Thoracic Surgery 2014 David R. Jones, M.D. Professor & Chief, Thoracic Surgery Memorial Sloan Kettering Cancer Center I have no disclosures
Targeted Therapy What the Surgeon Needs to Know AATS Focus in Thoracic Surgery 2014 David R. Jones, M.D. Professor & Chief, Thoracic Surgery Memorial Sloan Kettering Cancer Center I have no disclosures
Surgery. Wedge resection only part of the lung, not. not a lobe, is removed. Cancer Council NSW
 The treatment you receive will depend on your lung cancer type, for example, whether you have a non-small cell lung cancer Adenocarcinoma or Squamous cell carcinoma, and if this is a sub-type with a mutation.
The treatment you receive will depend on your lung cancer type, for example, whether you have a non-small cell lung cancer Adenocarcinoma or Squamous cell carcinoma, and if this is a sub-type with a mutation.
Non-Small Cell Lung Cancer Treatment Comparison to NCCN Guidelines
 Non-Small Cell Lung Cancer Treatment Comparison to NCCN Guidelines April 2008 (presented at 6/12/08 cancer committee meeting) By Shelly Smits, RHIT, CCS, CTR Conclusions by Dr. Ian Thompson, MD Dr. James
Non-Small Cell Lung Cancer Treatment Comparison to NCCN Guidelines April 2008 (presented at 6/12/08 cancer committee meeting) By Shelly Smits, RHIT, CCS, CTR Conclusions by Dr. Ian Thompson, MD Dr. James
A Practical Guide to Advances in Staging and Treatment of NSCLC
 A Practical Guide to Advances in Staging and Treatment of NSCLC Robert J. Korst, M.D. Director, Thoracic Surgery Medical Director, The Blumenthal Cancer Center The Valley Hospital Objectives Revised staging
A Practical Guide to Advances in Staging and Treatment of NSCLC Robert J. Korst, M.D. Director, Thoracic Surgery Medical Director, The Blumenthal Cancer Center The Valley Hospital Objectives Revised staging
The Need for Accurate Lung Cancer Staging
 The Need for Accurate Lung Cancer Staging Peter Baik, DO Thoracic Surgery Cancer Treatment Centers of America Oklahoma Osteopathic Association 115th Annual Convention Financial Disclosures: None 2 Objectives
The Need for Accurate Lung Cancer Staging Peter Baik, DO Thoracic Surgery Cancer Treatment Centers of America Oklahoma Osteopathic Association 115th Annual Convention Financial Disclosures: None 2 Objectives
POLICY A. INDICATIONS
 Alimta (pemetrexed) Line(s) of Business: HMO; PPO; QUEST Integration Akamai Advantage Original Effective Date: 09/01/2007 Current Effective Date: 10/01/2015 POLICY A. INDICATIONS The indications below
Alimta (pemetrexed) Line(s) of Business: HMO; PPO; QUEST Integration Akamai Advantage Original Effective Date: 09/01/2007 Current Effective Date: 10/01/2015 POLICY A. INDICATIONS The indications below
Lung Cancer and Mesothelioma
 Lung Cancer and Mesothelioma Robert Kratzke, M.D. John C. Skoglund Professor of Lung Cancer Research Section of Heme/Onc/Transplant Department of Medicine University of Minnesota Medical School Malignant
Lung Cancer and Mesothelioma Robert Kratzke, M.D. John C. Skoglund Professor of Lung Cancer Research Section of Heme/Onc/Transplant Department of Medicine University of Minnesota Medical School Malignant
SMALL CELL LUNG CANCER
 Protocol for Planning and Treatment The process to be followed in the management of: SMALL CELL LUNG CANCER Patient information given at each stage following agreed information pathway 1. DIAGNOSIS New
Protocol for Planning and Treatment The process to be followed in the management of: SMALL CELL LUNG CANCER Patient information given at each stage following agreed information pathway 1. DIAGNOSIS New
Lung Cancer Treatment Guidelines
 Updated June 2014 Derived and updated by consensus of members of the Providence Thoracic Oncology Program with the aid of evidence-based National Comprehensive Cancer Network (NCCN) national guidelines,
Updated June 2014 Derived and updated by consensus of members of the Providence Thoracic Oncology Program with the aid of evidence-based National Comprehensive Cancer Network (NCCN) national guidelines,
SAKK Lung Cancer Group. Current activities and future projects
 SAKK Lung Cancer Group Current activities and future projects SAKK Lung Cancer Group Open group of physicians interested in lung cancer Mostly Medical Oncologists, but also Thoracic Surgeons Radiation
SAKK Lung Cancer Group Current activities and future projects SAKK Lung Cancer Group Open group of physicians interested in lung cancer Mostly Medical Oncologists, but also Thoracic Surgeons Radiation
Small Cell Lung Cancer
 Small Cell Lung Cancer Types of Lung Cancer Non-small cell carcinoma (NSCC) (87%) Adenocarcinoma (38%) Squamous cell (20%) Large cell (5%) Small cell carcinoma (13%) Small cell lung cancer is virtually
Small Cell Lung Cancer Types of Lung Cancer Non-small cell carcinoma (NSCC) (87%) Adenocarcinoma (38%) Squamous cell (20%) Large cell (5%) Small cell carcinoma (13%) Small cell lung cancer is virtually
Rotation Specific Goals & Objectives: University Health Network-Princess Margaret Hospital/ Sunnybrook Breast/Melanoma
 Rotation Specific Goals & Objectives: University Health Network-Princess Margaret Hospital/ Sunnybrook Breast/Melanoma Medical Expert: Breast Rotation Specific Competencies/Objectives 1.0 Medical History
Rotation Specific Goals & Objectives: University Health Network-Princess Margaret Hospital/ Sunnybrook Breast/Melanoma Medical Expert: Breast Rotation Specific Competencies/Objectives 1.0 Medical History
Recruiting now. Could you help by joining this study?
 Non-Small Cell Lung Cancer Recruiting now AstraZeneca is looking for men and women with locally advanced or metastatic non-small cell lung cancer (NSCLC) to join ATLANTIC, a clinical study to help investigate
Non-Small Cell Lung Cancer Recruiting now AstraZeneca is looking for men and women with locally advanced or metastatic non-small cell lung cancer (NSCLC) to join ATLANTIC, a clinical study to help investigate
Stage I, II Non Small Cell Lung Cancer
 Stage I, II Non Small Cell Lung Cancer Best Results T1 (less 3 cm) N0 80% 5 year survival No Role Adjuvant Chemotherapy Radiation Therapy Reduces Local Recurrence No Improvement in Survival 1 Staging Mediastinal
Stage I, II Non Small Cell Lung Cancer Best Results T1 (less 3 cm) N0 80% 5 year survival No Role Adjuvant Chemotherapy Radiation Therapy Reduces Local Recurrence No Improvement in Survival 1 Staging Mediastinal
Survey on the treatment of non-small cell lung cancer (NSCLC) in England and Wales
 Eur Respir J 1997; 10: 1552 1558 DOI: 10.1183/09031936.97.10071552 Printed in UK - all rights reserved Copyright ERS Journals Ltd 1997 European Respiratory Journal ISSN 0903-1936 Survey on the treatment
Eur Respir J 1997; 10: 1552 1558 DOI: 10.1183/09031936.97.10071552 Printed in UK - all rights reserved Copyright ERS Journals Ltd 1997 European Respiratory Journal ISSN 0903-1936 Survey on the treatment
NCCN Non-Small Cell Lung Cancer V.1.2011 Update Meeting 07/09/10
 Guideline Page and Request NSCL-3 Stage IA, margins positive delete the recommendation for chemoradiation. Stage IB, IIA, margins positive delete the recommendation for chemoradiation + Stage IIA, Stage
Guideline Page and Request NSCL-3 Stage IA, margins positive delete the recommendation for chemoradiation. Stage IB, IIA, margins positive delete the recommendation for chemoradiation + Stage IIA, Stage
ORIGINAL ARTICLE THORACIC ONCOLOGY
 Ann Surg Oncol (2013) 20:1934 1940 DOI 10.1245/s10434-012-2800-x ORIGINAL ARTICLE THORACIC ONCOLOGY Predictors for Locoregional Recurrence for Clinical Stage III-N2 Non-small Cell Lung Cancer with Nodal
Ann Surg Oncol (2013) 20:1934 1940 DOI 10.1245/s10434-012-2800-x ORIGINAL ARTICLE THORACIC ONCOLOGY Predictors for Locoregional Recurrence for Clinical Stage III-N2 Non-small Cell Lung Cancer with Nodal
بسم هللا الرحمن الرحيم
 بسم هللا الرحمن الرحيم Updates in Mesothelioma By Samieh Amer, MD Professor of Cardiothoracic Surgery Faculty of Medicine, Cairo University History Wagner and his colleagues (1960) 33 cases of mesothelioma
بسم هللا الرحمن الرحيم Updates in Mesothelioma By Samieh Amer, MD Professor of Cardiothoracic Surgery Faculty of Medicine, Cairo University History Wagner and his colleagues (1960) 33 cases of mesothelioma
Table of Contents. Data Supplement 1: Summary of ASTRO Guideline Statements. Data Supplement 2: Definition of Terms
 Definitive and Adjuvant Radiotherapy in Locally Advanced Non-Small-Cell Lung Cancer: American Society of Clinical Oncology Clinical Practice Guideline Endorsement of the American Society for Radiation
Definitive and Adjuvant Radiotherapy in Locally Advanced Non-Small-Cell Lung Cancer: American Society of Clinical Oncology Clinical Practice Guideline Endorsement of the American Society for Radiation
3.0 With final Comments for presentation at Sub Group Meeting 24. 24.11.10
 Guideline for the Treatment of Lung Cancer Version History 2.0 Endorsed by the Governance Committee as treatment of lung cancer 27.07.09 with radiotherapy and chemotherapy. 2.1 Re-written to include the
Guideline for the Treatment of Lung Cancer Version History 2.0 Endorsed by the Governance Committee as treatment of lung cancer 27.07.09 with radiotherapy and chemotherapy. 2.1 Re-written to include the
Emerging Drug List GEFITINIB
 Generic (Trade Name): Manufacturer: Gefitinib (Iressa ) formerly referred to as ZD1839 AstraZeneca NO. 52 JANUARY 2004 Indication: Current Regulatory Status: Description: Current Treatment: Cost: Evidence:
Generic (Trade Name): Manufacturer: Gefitinib (Iressa ) formerly referred to as ZD1839 AstraZeneca NO. 52 JANUARY 2004 Indication: Current Regulatory Status: Description: Current Treatment: Cost: Evidence:
Principal Investigator: Valerie W. Rusch, MD, FACS, Chief, Thoracic Surgery Memorial Sloan-Kettering Cancer Center
 Protocol 1101-1088 Phase I study of intra-pleural administration of GL-ONC1 in patients with malignant pleural effusion: primary, metastases and mesothelioma Principal Investigator: Valerie W. Rusch, MD,
Protocol 1101-1088 Phase I study of intra-pleural administration of GL-ONC1 in patients with malignant pleural effusion: primary, metastases and mesothelioma Principal Investigator: Valerie W. Rusch, MD,
Treatment Algorithms for the Management of Lung Cancer in NSW Guide for Clinicians
 Treatment Algorithms for the Management of Lung Cancer in NSW Guide for Clinicians Background The Cancer Institute New South Wales Oncology Group Lung (NSWOG Lung) identified the need for the development
Treatment Algorithms for the Management of Lung Cancer in NSW Guide for Clinicians Background The Cancer Institute New South Wales Oncology Group Lung (NSWOG Lung) identified the need for the development
Summary of treatment benefits
 Risk Management Plan PEMETREXED Powder for concentrate for Solution for infusion Pemetrexed is also indicated as monotherapy for the maintenance treatment of locally advanced or metastatic non small cell
Risk Management Plan PEMETREXED Powder for concentrate for Solution for infusion Pemetrexed is also indicated as monotherapy for the maintenance treatment of locally advanced or metastatic non small cell
Moving forward, where are we with Clinical Trials?
 Moving forward, where are we with Clinical Trials? Dennis A. Wigle Division of Thoracic Surgery Mayo Clinic AATS/STS General Thoracic Surgery Symposium Sunday, April 27 th 2014 2012 MFMER slide-1 Where
Moving forward, where are we with Clinical Trials? Dennis A. Wigle Division of Thoracic Surgery Mayo Clinic AATS/STS General Thoracic Surgery Symposium Sunday, April 27 th 2014 2012 MFMER slide-1 Where
The expanding role of systemic treatment in non-small cell lung cancer neo-adjuvant therapy
 17 (Supplement 10): x108 x112, 2006 doi:10.1093/annonc/mdl247 The expanding role of systemic treatment in non-small cell lung cancer neo-adjuvant therapy E. Felip & E. Vilar Oncology Department, Vall d
17 (Supplement 10): x108 x112, 2006 doi:10.1093/annonc/mdl247 The expanding role of systemic treatment in non-small cell lung cancer neo-adjuvant therapy E. Felip & E. Vilar Oncology Department, Vall d
RESEARCH EDUCATE ADVOCATE. Just Diagnosed with Melanoma Now What?
 RESEARCH EDUCATE ADVOCATE Just Diagnosed with Melanoma Now What? INTRODUCTION If you are reading this, you have undergone a biopsy (either of a skin lesion or a lymph node) or have had other tests in which
RESEARCH EDUCATE ADVOCATE Just Diagnosed with Melanoma Now What? INTRODUCTION If you are reading this, you have undergone a biopsy (either of a skin lesion or a lymph node) or have had other tests in which
Adiuwantowe i neoadiuwantowe leczenie chorych na zaawansowanego raka żołądka
 Adiuwantowe i neoadiuwantowe leczenie chorych na zaawansowanego raka żołądka Neoadiuvant and adiuvant therapy for advanced gastric cancer Franco Roviello, IT Neoadjuvant and adjuvant therapy for advanced
Adiuwantowe i neoadiuwantowe leczenie chorych na zaawansowanego raka żołądka Neoadiuvant and adiuvant therapy for advanced gastric cancer Franco Roviello, IT Neoadjuvant and adjuvant therapy for advanced
Radiation Therapy in the Treatment of
 Lung Cancer Radiation Therapy in the Treatment of Lung Cancer JMAJ 46(12): 537 541, 2003 Kazushige HAYAKAWA Professor and Chairman, Department of Radiology, Kitasato University School of Medicine Abstract:
Lung Cancer Radiation Therapy in the Treatment of Lung Cancer JMAJ 46(12): 537 541, 2003 Kazushige HAYAKAWA Professor and Chairman, Department of Radiology, Kitasato University School of Medicine Abstract:
Targeting Specific Cell Signaling Pathways for the Treatment of Malignant Peritoneal Mesothelioma
 The Use of Kinase Inhibitors: Translational Lab Results Targeting Specific Cell Signaling Pathways for the Treatment of Malignant Peritoneal Mesothelioma Sheelu Varghese, Ph.D. H. Richard Alexander, M.D.
The Use of Kinase Inhibitors: Translational Lab Results Targeting Specific Cell Signaling Pathways for the Treatment of Malignant Peritoneal Mesothelioma Sheelu Varghese, Ph.D. H. Richard Alexander, M.D.
IF AT FIRST YOU DON T SUCCEED: TRIAL, TRIAL AGAIN
 + IF AT FIRST YOU DON T SUCCEED: TRIAL, TRIAL AGAIN Rena Buckstein MD FRCPC Head Hematology Site Group Sunnybrook Odette Cancer Center (OCC) Head of Hematology Clinical Trials Group at OCC + Outline Start
+ IF AT FIRST YOU DON T SUCCEED: TRIAL, TRIAL AGAIN Rena Buckstein MD FRCPC Head Hematology Site Group Sunnybrook Odette Cancer Center (OCC) Head of Hematology Clinical Trials Group at OCC + Outline Start
Tricia Cox on 7/18/2012 at Oncology Center. Sarah Randolf. Female
 SAMPLE This Survivorship Care Plan will facilitate cancer care following active treatment. It may include important contact information, a treatment summary, recommendations for follow-up care testing,
SAMPLE This Survivorship Care Plan will facilitate cancer care following active treatment. It may include important contact information, a treatment summary, recommendations for follow-up care testing,
CANCER PULMON: ESTADIOS INICIALES POSTMUNDIAL PULMON DENVER 2015. 8-10-2015.Manuel Cobo Dols S. Oncología Médica HU Málaga Regional y VV
 CANCER PULMON: ESTADIOS INICIALES POSTMUNDIAL PULMON DENVER 2015 8-10-2015.Manuel Cobo Dols S. Oncología Médica HU Málaga Regional y VV Meta-analisis LACE: adyuvancia vs no adyuvancia Pignon JP, et al.
CANCER PULMON: ESTADIOS INICIALES POSTMUNDIAL PULMON DENVER 2015 8-10-2015.Manuel Cobo Dols S. Oncología Médica HU Málaga Regional y VV Meta-analisis LACE: adyuvancia vs no adyuvancia Pignon JP, et al.
Probe: Could you tell me about when?
 PERIODIC ASSESSMENT OF TREATMENT AND VITAL/DISEASE STATUS Periodic Assessment of Cancer Treatment and Disease Status (To be administered to patient at 3 months and reviewed at 6, 9 and 12 months) Instructions:
PERIODIC ASSESSMENT OF TREATMENT AND VITAL/DISEASE STATUS Periodic Assessment of Cancer Treatment and Disease Status (To be administered to patient at 3 months and reviewed at 6, 9 and 12 months) Instructions:
The lungs What is lung cancer? How common is it? Risks & symptoms Diagnosis & treatment options
 Why We re Here The lungs What is lung cancer? How common is it? Risks & symptoms Diagnosis & treatment options What Are Lungs? What Do They Do? 1 Located in the chest Allow you to breathe Provide oxygen
Why We re Here The lungs What is lung cancer? How common is it? Risks & symptoms Diagnosis & treatment options What Are Lungs? What Do They Do? 1 Located in the chest Allow you to breathe Provide oxygen
PET/CT in Lung Cancer
 PET/CT in Lung Cancer Rodolfo Núñez Miller, M.D. Nuclear Medicine and Diagnostic Imaging Section Division of Human Health International Atomic Energy Agency Vienna, Austria GLOBOCAN 2012 #1 #3 FDG-PET/CT
PET/CT in Lung Cancer Rodolfo Núñez Miller, M.D. Nuclear Medicine and Diagnostic Imaging Section Division of Human Health International Atomic Energy Agency Vienna, Austria GLOBOCAN 2012 #1 #3 FDG-PET/CT
Prior Authorization Guideline
 Prior Authorization Guideline Guideline: PS Inj - Alimta Therapeutic Class: Antineoplastic Agents Therapeutic Sub-Class: Antifolates Client: PS Inj Approval Date: 8/2/2004 Revision Date: 12/5/2006 I. BENEFIT
Prior Authorization Guideline Guideline: PS Inj - Alimta Therapeutic Class: Antineoplastic Agents Therapeutic Sub-Class: Antifolates Client: PS Inj Approval Date: 8/2/2004 Revision Date: 12/5/2006 I. BENEFIT
General Information About Non-Small Cell Lung Cancer
 General Information About Non-Small Cell Lung Cancer Non-small cell lung cancer is a disease in which malignant (cancer) cells form in the tissues of the lung. The lungs are a pair of cone-shaped breathing
General Information About Non-Small Cell Lung Cancer Non-small cell lung cancer is a disease in which malignant (cancer) cells form in the tissues of the lung. The lungs are a pair of cone-shaped breathing
Round Table with Drs. Anne Tsao and Alex Farivar, Case 2: Mesothelioma
 Round Table with Drs. Anne Tsao and Alex Farivar, Case 2: Mesothelioma I d like to welcome everyone, thanks for coming out to our lunch with experts. The faculty today are great people in the thoracic
Round Table with Drs. Anne Tsao and Alex Farivar, Case 2: Mesothelioma I d like to welcome everyone, thanks for coming out to our lunch with experts. The faculty today are great people in the thoracic
TREATING LUNG CANCER AT VA PITTSBURGH HEALTHCARE SYSTEM A HERO S GUIDE
 Providing the best in cancer care for Veterans TREATING LUNG CANCER AT VA PITTSBURGH HEALTHCARE SYSTEM A HERO S GUIDE VA Pittsburgh s state-of-the-art TomoTherapy machine, which delivers radiation to patients.
Providing the best in cancer care for Veterans TREATING LUNG CANCER AT VA PITTSBURGH HEALTHCARE SYSTEM A HERO S GUIDE VA Pittsburgh s state-of-the-art TomoTherapy machine, which delivers radiation to patients.
New Trends & Current Research in the Treatment of Lung Cancer, Pt. II
 New Trends & Current esearch in the Treatment of Lung Cancer, Pt. II Howard (Jack) West, MD President & CEO, GACE Medical Director, Thoracic Oncology Program Swedish Cancer Institute Seattle, WA Cancer
New Trends & Current esearch in the Treatment of Lung Cancer, Pt. II Howard (Jack) West, MD President & CEO, GACE Medical Director, Thoracic Oncology Program Swedish Cancer Institute Seattle, WA Cancer
OI PARP ΑΝΑΣΤΟΛΕΙΣ ΣΤΟΝ ΚΑΡΚΙΝΟ ΤΟΥ ΜΑΣΤΟΥ ΝΙΚΟΛΑΙΔΗ ΑΔΑΜΑΝΤΙΑ ΠΑΘΟΛΟΓΟΣ-ΟΓΚΟΛΟΓΟΣ Β ΟΓΚΟΛΟΓΙΚΗ ΚΛΙΝΙΚΗ ΝΟΣ. ΜΗΤΕΡΑ
 OI PARP ΑΝΑΣΤΟΛΕΙΣ ΣΤΟΝ ΚΑΡΚΙΝΟ ΤΟΥ ΜΑΣΤΟΥ ΝΙΚΟΛΑΙΔΗ ΑΔΑΜΑΝΤΙΑ ΠΑΘΟΛΟΓΟΣ-ΟΓΚΟΛΟΓΟΣ Β ΟΓΚΟΛΟΓΙΚΗ ΚΛΙΝΙΚΗ ΝΟΣ. ΜΗΤΕΡΑ Study Overview Inhibition of poly(adenosine diphosphate [ADP]-ribose) polymerase
OI PARP ΑΝΑΣΤΟΛΕΙΣ ΣΤΟΝ ΚΑΡΚΙΝΟ ΤΟΥ ΜΑΣΤΟΥ ΝΙΚΟΛΑΙΔΗ ΑΔΑΜΑΝΤΙΑ ΠΑΘΟΛΟΓΟΣ-ΟΓΚΟΛΟΓΟΣ Β ΟΓΚΟΛΟΓΙΚΗ ΚΛΙΝΙΚΗ ΝΟΣ. ΜΗΤΕΡΑ Study Overview Inhibition of poly(adenosine diphosphate [ADP]-ribose) polymerase
Metastatic Breast Cancer 201. Carolyn B. Hendricks, MD October 29, 2011
 Metastatic Breast Cancer 201 Carolyn B. Hendricks, MD October 29, 2011 Overview Is rebiopsy necessary at the time of recurrence or progression of disease? How dose a very aggressive treatment upfront compare
Metastatic Breast Cancer 201 Carolyn B. Hendricks, MD October 29, 2011 Overview Is rebiopsy necessary at the time of recurrence or progression of disease? How dose a very aggressive treatment upfront compare
Disease/Illness GUIDE TO ASBESTOS LUNG CANCER. What Is Asbestos Lung Cancer? www.simpsonmillar.co.uk Telephone 0844 858 3200
 GUIDE TO ASBESTOS LUNG CANCER What Is Asbestos Lung Cancer? Like tobacco smoking, exposure to asbestos can result in the development of lung cancer. Similarly, the risk of developing asbestos induced lung
GUIDE TO ASBESTOS LUNG CANCER What Is Asbestos Lung Cancer? Like tobacco smoking, exposure to asbestos can result in the development of lung cancer. Similarly, the risk of developing asbestos induced lung
Mesothelioma. Malignant Pleural Mesothelioma
 Mesothelioma William G. Richards, PhD Brigham and Women s Hospital Malignant Pleural Mesothelioma 2,000-3,000 cases per year (USA) Increasing incidence Asbestos (50-80%, decreasing) 30-40 year latency
Mesothelioma William G. Richards, PhD Brigham and Women s Hospital Malignant Pleural Mesothelioma 2,000-3,000 cases per year (USA) Increasing incidence Asbestos (50-80%, decreasing) 30-40 year latency
Gemcitabine, Paclitaxel, and Trastuzumab in Metastatic Breast Cancer
 Gemcitabine, Paclitaxel, and Trastuzumab in Metastatic Breast Cancer Review Article [1] December 01, 2003 By George W. Sledge, Jr, MD [2] Gemcitabine (Gemzar) and paclitaxel show good activity as single
Gemcitabine, Paclitaxel, and Trastuzumab in Metastatic Breast Cancer Review Article [1] December 01, 2003 By George W. Sledge, Jr, MD [2] Gemcitabine (Gemzar) and paclitaxel show good activity as single
L Lang-Lazdunski, A Bille, S Marshall, R Lal, D Landau, J Spicer
 Pleurectomy/decortication, hyperthermic pleural lavage with povidone-iodine and systemic chemotherapy in malignant pleural mesothelioma. A 10-year experience. L Lang-Lazdunski, A Bille, S Marshall, R Lal,
Pleurectomy/decortication, hyperthermic pleural lavage with povidone-iodine and systemic chemotherapy in malignant pleural mesothelioma. A 10-year experience. L Lang-Lazdunski, A Bille, S Marshall, R Lal,
Avastin: Glossary of key terms
 Avastin: Glossary of key terms Adenocarcinoma Adenoma Adjuvant therapy Angiogenesis Anti-angiogenics Antibody Antigen Avastin (bevacizumab) Benign A form of carcinoma that originates in glandular tissue.
Avastin: Glossary of key terms Adenocarcinoma Adenoma Adjuvant therapy Angiogenesis Anti-angiogenics Antibody Antigen Avastin (bevacizumab) Benign A form of carcinoma that originates in glandular tissue.
REPORT ASCO 1998 LOS ANGELES : LUNG CANCER Johan F. Vansteenkiste, MD, PhD, Univ. Hospital and Leuven Lung Cancer Group
 REPORT ASCO 1998 LOS ANGELES : LUNG CANCER Johan F. Vansteenkiste, MD, PhD, Univ. Hospital and Leuven Lung Cancer Group Educational session Treatment of stage III non-small cell lung cancer (NSCLC) in
REPORT ASCO 1998 LOS ANGELES : LUNG CANCER Johan F. Vansteenkiste, MD, PhD, Univ. Hospital and Leuven Lung Cancer Group Educational session Treatment of stage III non-small cell lung cancer (NSCLC) in
Management of Non-Small Cell Lung Cancer Guide for General Practitioners
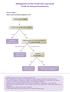 Management of n-small Cell Lung Cancer Guide for General Practitioners Clinical Stage I Cancer only in one lobe of lung and
Management of n-small Cell Lung Cancer Guide for General Practitioners Clinical Stage I Cancer only in one lobe of lung and
Update on Small Cell Lung Cancer
 Welcome to Master Class for Oncologists Session 3: 2:45 PM - 3:30 PM Washington, DC March 28, 2009 Small Cell Lung Cancer: Best Practices & Recent Advances Speaker: Bruce E. Johnson, MD Professor of Medicine,
Welcome to Master Class for Oncologists Session 3: 2:45 PM - 3:30 PM Washington, DC March 28, 2009 Small Cell Lung Cancer: Best Practices & Recent Advances Speaker: Bruce E. Johnson, MD Professor of Medicine,
Post-PET Restaging Cancer Form National Oncologic PET Registry
 Post-PET Restaging Cancer Form National Oncologic PET Registry Facility ID #: Registry Case Number: Patient Name: Your patient had a PET scan on: mm/dd/yyyy. The PET scan was done for restaging of (cancer
Post-PET Restaging Cancer Form National Oncologic PET Registry Facility ID #: Registry Case Number: Patient Name: Your patient had a PET scan on: mm/dd/yyyy. The PET scan was done for restaging of (cancer
Harmesh Naik, MD. Hope Cancer Clinic HOW DO I MANAGE STAGE 4 NSCLC IN 2012: STATE OF THE ART
 Harmesh Naik, MD. Hope Cancer Clinic HOW DO I MANAGE STAGE 4 NSCLC IN 2012: STATE OF THE ART Goals Discuss treatment options for stage 4 lung cancer: New and old Discuss new developments in personalized
Harmesh Naik, MD. Hope Cancer Clinic HOW DO I MANAGE STAGE 4 NSCLC IN 2012: STATE OF THE ART Goals Discuss treatment options for stage 4 lung cancer: New and old Discuss new developments in personalized
Non-Small Cell Lung Cancer Therapies
 Non-Small Cell Lung Cancer Therapies Guest Expert: Roy, MD, PhD Assistant Professor of Therapeutic Radiology Scott, MD Assistant Professor of Medical Oncology www.wnpr.org www.yalecancercenter.org Welcome
Non-Small Cell Lung Cancer Therapies Guest Expert: Roy, MD, PhD Assistant Professor of Therapeutic Radiology Scott, MD Assistant Professor of Medical Oncology www.wnpr.org www.yalecancercenter.org Welcome
Non-Small Cell Lung Cancer
 Non-Small Cell Lung Cancer John delcharco, MD (Statistics based on CVMC data 2009-2013) Statistics Lung cancer is the leading cause of cancer deaths in the United States. The American Cancer Society estimates
Non-Small Cell Lung Cancer John delcharco, MD (Statistics based on CVMC data 2009-2013) Statistics Lung cancer is the leading cause of cancer deaths in the United States. The American Cancer Society estimates
SMALL. 1-800-298-2436 LungCancerAlliance.org
 Understanding series SMALL CELL LUNG CANCER 1-800-298-2436 LungCancerAlliance.org A guide for the patient I TABLE OF CONTENTS ANATOMY OF THE LUNGS The following image shows different parts that make up
Understanding series SMALL CELL LUNG CANCER 1-800-298-2436 LungCancerAlliance.org A guide for the patient I TABLE OF CONTENTS ANATOMY OF THE LUNGS The following image shows different parts that make up
Klaus Junker, MD; Kathrin Langner, MD; Folker Klinke, MD; Ulrich Bosse, MD; and Michael Thomas, MD
 Grading of Tumor Regression in Non-small Cell Lung Cancer* Morphology and Prognosis Klaus Junker, MD; Kathrin Langner, MD; Folker Klinke, MD; Ulrich Bosse, MD; and Michael Thomas, MD Objective: Different
Grading of Tumor Regression in Non-small Cell Lung Cancer* Morphology and Prognosis Klaus Junker, MD; Kathrin Langner, MD; Folker Klinke, MD; Ulrich Bosse, MD; and Michael Thomas, MD Objective: Different
What If I Have a Spot on My Lung? Do I Have Cancer? Patient Education Guide
 What If I Have a Spot on My Lung? Do I Have Cancer? Patient Education Guide A M E R I C A N C O L L E G E O F C H E S T P H Y S I C I A N S Lung cancer is one of the most common cancers. About 170,000
What If I Have a Spot on My Lung? Do I Have Cancer? Patient Education Guide A M E R I C A N C O L L E G E O F C H E S T P H Y S I C I A N S Lung cancer is one of the most common cancers. About 170,000
CLINICAL TRIALS SHOULD YOU PARTICIPATE? by Gwen L. Nichols, MD
 CLINICAL TRIALS SHOULD YOU PARTICIPATE? by Gwen L. Nichols, MD Gwen L. Nichols, M.D., is currently the Oncology Site Head of the Roche Translational Clinical Research Center at Hoffman- LaRoche. In this
CLINICAL TRIALS SHOULD YOU PARTICIPATE? by Gwen L. Nichols, MD Gwen L. Nichols, M.D., is currently the Oncology Site Head of the Roche Translational Clinical Research Center at Hoffman- LaRoche. In this
Facing Lung Cancer? Learn why da Vinci Surgery may be your best treatment option for lung cancer.
 Facing Lung Cancer? Learn why da Vinci Surgery may be your best treatment option for lung cancer. The Condition: Lung Cancer The lung is the organ that moves oxygen through your body. You have two lungs
Facing Lung Cancer? Learn why da Vinci Surgery may be your best treatment option for lung cancer. The Condition: Lung Cancer The lung is the organ that moves oxygen through your body. You have two lungs
Management of stage III A-B of NSCLC. Hamed ALHusaini Medical Oncologist
 Management of stage III A-B of NSCLC Hamed ALHusaini Medical Oncologist Global incidence, CA cancer J Clin 2011;61:69-90 Stage III NSCLC Includes heterogeneous group of patients with differences in the
Management of stage III A-B of NSCLC Hamed ALHusaini Medical Oncologist Global incidence, CA cancer J Clin 2011;61:69-90 Stage III NSCLC Includes heterogeneous group of patients with differences in the
Post-recurrence survival in completely resected stage I non-small cell lung cancer with local recurrence
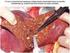 Post- survival in completely resected stage I non-small cell lung cancer with local J-J Hung, 1,2,3 W-H Hsu, 3 C-C Hsieh, 3 B-S Huang, 3 M-H Huang, 3 J-S Liu, 2 Y-C Wu 3 See Editorial, p 185 c A supplementary
Post- survival in completely resected stage I non-small cell lung cancer with local J-J Hung, 1,2,3 W-H Hsu, 3 C-C Hsieh, 3 B-S Huang, 3 M-H Huang, 3 J-S Liu, 2 Y-C Wu 3 See Editorial, p 185 c A supplementary
REPORT PERSPECTIVES IN LUNG CANCER 2010 AMSTERDAM
 REPORT PERSPECTIVES IN LUNG CANCER 2010 AMSTERDAM Valerie Van Damme, Isabelle Wauters, Johan Vansteenkiste Univ. Hospital Leuven and Leuven Lung Cancer Group Introduction Perspectives in Lung Cancer (PILC)
REPORT PERSPECTIVES IN LUNG CANCER 2010 AMSTERDAM Valerie Van Damme, Isabelle Wauters, Johan Vansteenkiste Univ. Hospital Leuven and Leuven Lung Cancer Group Introduction Perspectives in Lung Cancer (PILC)
Future Directions in Clinical Research. Karen Kelly, MD Associate Director for Clinical Research UC Davis Cancer Center
 Future Directions in Clinical Research Karen Kelly, MD Associate Director for Clinical Research UC Davis Cancer Center Outline 1. Status of Cancer Treatment 2. Overview of Clinical Research at UCDCC 3.
Future Directions in Clinical Research Karen Kelly, MD Associate Director for Clinical Research UC Davis Cancer Center Outline 1. Status of Cancer Treatment 2. Overview of Clinical Research at UCDCC 3.
CLINICAL POLICY Department: Medical Management Document Name: Opdivo Reference Number: CP.PHAR.121 Effective Date: 07/15
 Page: 1 of 6 IMPORTANT REMINDER This Clinical Policy has been developed by appropriately experienced and licensed health care professionals based on a thorough review and consideration of generally accepted
Page: 1 of 6 IMPORTANT REMINDER This Clinical Policy has been developed by appropriately experienced and licensed health care professionals based on a thorough review and consideration of generally accepted
Mesothelioma. 1. Introduction. 1.1 General Information and Aetiology
 Mesothelioma 1. Introduction 1.1 General Information and Aetiology Mesotheliomas are tumours that arise from the mesothelial cells of the pleura, peritoneum, pericardium or tunica vaginalis [1]. Most are
Mesothelioma 1. Introduction 1.1 General Information and Aetiology Mesotheliomas are tumours that arise from the mesothelial cells of the pleura, peritoneum, pericardium or tunica vaginalis [1]. Most are
Accelerated hemithoracic radiation followed by extrapleural pneumonectomy for malignant pleural mesothelioma
 Accelerated hemithoracic radiation followed by extrapleural pneumonectomy for malignant pleural mesothelioma Marc de Perrot, Ronald Feld, Natasha B Leighl, Andrew Hope, Thomas K Waddell, Shaf Keshavjee,
Accelerated hemithoracic radiation followed by extrapleural pneumonectomy for malignant pleural mesothelioma Marc de Perrot, Ronald Feld, Natasha B Leighl, Andrew Hope, Thomas K Waddell, Shaf Keshavjee,
HAVE YOU BEEN NEWLY DIAGNOSED with DCIS?
 HAVE YOU BEEN NEWLY DIAGNOSED with DCIS? Jen D. Mother and volunteer. Diagnosed with DCIS breast cancer in 2012. An educational guide prepared by Genomic Health This guide is designed to educate women
HAVE YOU BEEN NEWLY DIAGNOSED with DCIS? Jen D. Mother and volunteer. Diagnosed with DCIS breast cancer in 2012. An educational guide prepared by Genomic Health This guide is designed to educate women
clinical trials patient-assisted research studies
 LUNG CANCER TREATMENTS What you need to know about... clinical trials patient-assisted research studies foreword About LUNGevity LUNGevity is the largest national lung cancer-focused nonprofit, changing
LUNG CANCER TREATMENTS What you need to know about... clinical trials patient-assisted research studies foreword About LUNGevity LUNGevity is the largest national lung cancer-focused nonprofit, changing
U.S. Department of Health and Human Services Food and Drug Administration Center for Drug Evaluation and Research (CDER)
 Guidance for Industry Pathological Complete Response in Neoadjuvant Treatment of High-Risk Early-Stage Breast Cancer: Use as an Endpoint to Support Accelerated Approval U.S. Department of Health and Human
Guidance for Industry Pathological Complete Response in Neoadjuvant Treatment of High-Risk Early-Stage Breast Cancer: Use as an Endpoint to Support Accelerated Approval U.S. Department of Health and Human
Lung cancer forms in tissues of the lung, usually in the cells lining air passages.
 Scan for mobile link. Lung Cancer Lung cancer usually forms in the tissue cells lining the air passages within the lungs. The two main types are small-cell lung cancer (usually found in cigarette smokers)
Scan for mobile link. Lung Cancer Lung cancer usually forms in the tissue cells lining the air passages within the lungs. The two main types are small-cell lung cancer (usually found in cigarette smokers)
Lung Cancer & Mesothelioma 2011-2015
 Lung Cancer & Mesothelioma 2011-2015 Annex G Mesothelioma 1. The vision for mesothelioma services is set out in the Mesothelioma Framework issued by DH on 26 February 2007 (supported by the British Thoracic
Lung Cancer & Mesothelioma 2011-2015 Annex G Mesothelioma 1. The vision for mesothelioma services is set out in the Mesothelioma Framework issued by DH on 26 February 2007 (supported by the British Thoracic
Hodgkin Lymphoma Disease Specific Biology and Treatment Options. John Kuruvilla
 Hodgkin Lymphoma Disease Specific Biology and Treatment Options John Kuruvilla My Disclaimer This is where I work Objectives Pathobiology what makes HL different Diagnosis Staging Treatment Philosophy
Hodgkin Lymphoma Disease Specific Biology and Treatment Options John Kuruvilla My Disclaimer This is where I work Objectives Pathobiology what makes HL different Diagnosis Staging Treatment Philosophy
Please see the LUCADA data manual v3.1.3, available in the downloads section
 National Lung Cancer Audit Frequently Asked Questions What dataset should be used? Please see the LUCADA data manual v3.1.3, available in the downloads section What does LUCADA stand for? LUCADA stands
National Lung Cancer Audit Frequently Asked Questions What dataset should be used? Please see the LUCADA data manual v3.1.3, available in the downloads section What does LUCADA stand for? LUCADA stands
Treatment of Metastatic Non-Small Cell Lung Cancer: A Systematic Review of Comparative Effectiveness and Cost Effectiveness
 Treatment of Metastatic Non-Small Cell Lung Cancer: A Systematic Review of Comparative Effectiveness and Cost Effectiveness Investigators: Paul G. Shekelle, MD, PhD, Director Alicia R. Maher, MD Clinical
Treatment of Metastatic Non-Small Cell Lung Cancer: A Systematic Review of Comparative Effectiveness and Cost Effectiveness Investigators: Paul G. Shekelle, MD, PhD, Director Alicia R. Maher, MD Clinical
Is the third-line chemotherapy feasible for non-small cell lung cancer? A retrospective study
 Turkish Journal of Cancer Volume 34, No.1, 2004 19 Is the third-line chemotherapy feasible for non-small cell lung cancer? A retrospective study MUSTAFA ÖZDO AN, MUSTAFA SAMUR, HAKAN BOZCUK, ERKAN ÇOBAN,
Turkish Journal of Cancer Volume 34, No.1, 2004 19 Is the third-line chemotherapy feasible for non-small cell lung cancer? A retrospective study MUSTAFA ÖZDO AN, MUSTAFA SAMUR, HAKAN BOZCUK, ERKAN ÇOBAN,
How To Treat Mesothelioma With A Tumor Stem Cell Inhibitor
 FAK INHIBITOR DEFACTINIB (VS-6063) TARGETS MESOTHELIOMA CANCER STEM CELLS Rationale for maintenance therapy after conventional therapy Jonathan Pachter, Ph.D. Vice President of Research, Verastem, Inc.
FAK INHIBITOR DEFACTINIB (VS-6063) TARGETS MESOTHELIOMA CANCER STEM CELLS Rationale for maintenance therapy after conventional therapy Jonathan Pachter, Ph.D. Vice President of Research, Verastem, Inc.
Activity of pemetrexed in thoracic malignancies
 Activity of pemetrexed in thoracic malignancies Results of phase III clinical studies of pemetrexed in malignant pleural mesothelioma and non-small cell lung cancer show benefit P emetrexed (Alimta) is
Activity of pemetrexed in thoracic malignancies Results of phase III clinical studies of pemetrexed in malignant pleural mesothelioma and non-small cell lung cancer show benefit P emetrexed (Alimta) is
National Clinical Trials Network Groups Update Fall 2014
 National Clinical Trials Network Groups Update Fall 2014 Walter J Curran, Jr, MD An NRG Oncology Group Chair Executive Director Winship Cancer Institute of Emory University Atlanta, GA NCTN Groups Update
National Clinical Trials Network Groups Update Fall 2014 Walter J Curran, Jr, MD An NRG Oncology Group Chair Executive Director Winship Cancer Institute of Emory University Atlanta, GA NCTN Groups Update
Malignant Mesothelioma State of the Art
 Malignant Mesothelioma State of the Art Paul Baas The Netherlands Cancer Institute August 12, 2011, Carlsbad, CA Summary Diagnosis; epithelial type subdivided Pleiomorphic vs other Staging: IASLC-IMIG
Malignant Mesothelioma State of the Art Paul Baas The Netherlands Cancer Institute August 12, 2011, Carlsbad, CA Summary Diagnosis; epithelial type subdivided Pleiomorphic vs other Staging: IASLC-IMIG
RH de Boer 1, IE Smith 1, U Pastorino 2, MER O Brien 1, F Ramage 1, S Ashley 1 and P Goldstraw 2
 British Journal of Cancer (1999) 79(9/10), 1514 1518 1999 Cancer Research Campaign Article no. bjoc.1998.0241 Pre-operative chemotherapy in early stage resectable non-small-cell lung cancer: a randomized
British Journal of Cancer (1999) 79(9/10), 1514 1518 1999 Cancer Research Campaign Article no. bjoc.1998.0241 Pre-operative chemotherapy in early stage resectable non-small-cell lung cancer: a randomized
Corso Integrato di Clinica Medica ONCOLOGIA MEDICA AA 2010-2011 LUNG CANCER. VIII. THERAPY. V. SMALL CELL LUNG CANCER Prof.
 Corso Integrato di Clinica Medica ONCOLOGIA MEDICA AA 2010-2011 LUNG CANCER. VIII. THERAPY. V. SMALL CELL LUNG CANCER Prof. Alberto Riccardi SMALL CELL LUNG CARCINOMA Summary of treatment approach * limited
Corso Integrato di Clinica Medica ONCOLOGIA MEDICA AA 2010-2011 LUNG CANCER. VIII. THERAPY. V. SMALL CELL LUNG CANCER Prof. Alberto Riccardi SMALL CELL LUNG CARCINOMA Summary of treatment approach * limited
Treatment of Metastatic Non-Small Cell Lung Cancer: A Systematic Review of Comparative Effectiveness and Cost-Effectiveness
 Department of Veterans Affairs Health Services Research & Development Service Treatment of Metastatic Non-Small Cell Lung Cancer: A Systematic Review of Comparative Effectiveness and Cost-Effectiveness
Department of Veterans Affairs Health Services Research & Development Service Treatment of Metastatic Non-Small Cell Lung Cancer: A Systematic Review of Comparative Effectiveness and Cost-Effectiveness
Post-operative intrapleural chemotherapy for mesothelioma
 Post-operative intrapleural chemotherapy for mesothelioma Robert Kratzke, MD John Skoglund Chair for Lung Cancer Research Section of Heme-Onc-Transplant University of Minnesota Medical School Efficacy
Post-operative intrapleural chemotherapy for mesothelioma Robert Kratzke, MD John Skoglund Chair for Lung Cancer Research Section of Heme-Onc-Transplant University of Minnesota Medical School Efficacy
Malignant pleural mesothelioma P/D vs. EPP
 3 rd International Thoracic Oncology Congress Dresden, September 13 15, 2012 Malignant pleural mesothelioma P/D vs. EPP Walter Weder, MD Professor of Surgery Dokumentenname Datum Seite 1 Extrapleural Pneumonectomy
3 rd International Thoracic Oncology Congress Dresden, September 13 15, 2012 Malignant pleural mesothelioma P/D vs. EPP Walter Weder, MD Professor of Surgery Dokumentenname Datum Seite 1 Extrapleural Pneumonectomy
CHAPTER 6: TREATMENT FOR SMALL CELL LUNG CANCER
 CHAPTER 6: TREATMENT FOR SMALL CELL LUNG CANCER INTRODUCTION This chapter provides an overview of treatment for small cell lung cancer (SCLC). Treatment options are presented based on the extent of disease.
CHAPTER 6: TREATMENT FOR SMALL CELL LUNG CANCER INTRODUCTION This chapter provides an overview of treatment for small cell lung cancer (SCLC). Treatment options are presented based on the extent of disease.
2009 Cancer Committee
 2009 Cancer Committee Lung (non-small cell) Cancer Study Stage IIIA and IIIB John Clouse, M.D. Alicia Stark, RHIT Jeff Robinson Table of Cont ent s Table of Contents... 1 Lung (non-small cell) Cancer Facts.2
2009 Cancer Committee Lung (non-small cell) Cancer Study Stage IIIA and IIIB John Clouse, M.D. Alicia Stark, RHIT Jeff Robinson Table of Cont ent s Table of Contents... 1 Lung (non-small cell) Cancer Facts.2
BNC105 CANCER CLINICAL TRIALS REACH KEY MILESTONES CLINICAL PROGRAM TO BE EXPANDED
 ASX ANNOUNCEMENT 3 August 2011 ABN 53 075 582 740 BNC105 CANCER CLINICAL TRIALS REACH KEY MILESTONES CLINICAL PROGRAM TO BE EXPANDED Data from renal cancer trial supports progression of the trial: o Combination
ASX ANNOUNCEMENT 3 August 2011 ABN 53 075 582 740 BNC105 CANCER CLINICAL TRIALS REACH KEY MILESTONES CLINICAL PROGRAM TO BE EXPANDED Data from renal cancer trial supports progression of the trial: o Combination
LOOKING FORWARD PUMA BIOTECHNOLOGY, INC. 2014 ANNUAL REPORT
 LOOKING FORWARD PUMA BIOTECHNOLOGY, INC. 2014 ANNUAL REPORT Puma Biotechnology, Inc. is a development stage biopharmaceutical company that acquires and develops innovative products for the treatment of
LOOKING FORWARD PUMA BIOTECHNOLOGY, INC. 2014 ANNUAL REPORT Puma Biotechnology, Inc. is a development stage biopharmaceutical company that acquires and develops innovative products for the treatment of
Guidance for Industry
 Guidance for Industry Cancer Drug and Biological Products Clinical Data in Marketing Applications U.S. Department of Health and Human Services Food and Drug Administration Center for Drug Evaluation and
Guidance for Industry Cancer Drug and Biological Products Clinical Data in Marketing Applications U.S. Department of Health and Human Services Food and Drug Administration Center for Drug Evaluation and
Study on Melanoma - New Research for Cancer Patients
 A service of the U.S. National Institutes of Health Trial record 7 of 203 for: Melanoma Analysis Previous Study Return to List Next Study Expression Analysis of Specific Markers in Non-small Cell Lung
A service of the U.S. National Institutes of Health Trial record 7 of 203 for: Melanoma Analysis Previous Study Return to List Next Study Expression Analysis of Specific Markers in Non-small Cell Lung
Lung cancer is not just one disease. There are two main types of lung cancer:
 1. What is lung cancer? 2. How common is lung cancer? 3. What are the risk factors for lung cancer? 4. What are the signs and symptoms of lung cancer? 5. How is lung cancer diagnosed? 6. What are the available
1. What is lung cancer? 2. How common is lung cancer? 3. What are the risk factors for lung cancer? 4. What are the signs and symptoms of lung cancer? 5. How is lung cancer diagnosed? 6. What are the available
REPORT ASCO 2002 ORLANDO : LUNG CANCER Johan F. Vansteenkiste, MD, PhD, Univ. Hospital and Leuven Lung Cancer Group
 REPORT ASCO 2002 ORLANDO : LUNG CANCER Johan F. Vansteenkiste, MD, PhD, Univ. Hospital and Leuven Lung Cancer Group In the 2002 edition of the ASCO meeting, a total of 315 abstracts in the field of respiratory
REPORT ASCO 2002 ORLANDO : LUNG CANCER Johan F. Vansteenkiste, MD, PhD, Univ. Hospital and Leuven Lung Cancer Group In the 2002 edition of the ASCO meeting, a total of 315 abstracts in the field of respiratory
The National Clinical Lung Cancer Audit (LUCADA)
 The National Clinical Lung Cancer Audit (LUCADA) DATA MANUAL Title: Version: 3.1.5 Date: September 2013 LUCADA Lung Cancer Audit VERSION HISTORY Version Date Issued Brief Summary of Change Owner s Name
The National Clinical Lung Cancer Audit (LUCADA) DATA MANUAL Title: Version: 3.1.5 Date: September 2013 LUCADA Lung Cancer Audit VERSION HISTORY Version Date Issued Brief Summary of Change Owner s Name
Chapter 7: Lung Cancer
 Chapter 7: Lung Cancer Contents Chapter 7: Lung Cancer... 1 Small Cell... 2 Good PS + Limited stage... 2 Cisplatin/etoposide... 2 Concurrent chemotherapy + XRT... 2 Good / Intermediate PS... 2 Carboplatin
Chapter 7: Lung Cancer Contents Chapter 7: Lung Cancer... 1 Small Cell... 2 Good PS + Limited stage... 2 Cisplatin/etoposide... 2 Concurrent chemotherapy + XRT... 2 Good / Intermediate PS... 2 Carboplatin
People Living with Cancer
 Patient Guide ASCOInformation for People Living with Cancer ADVANCED LUNG CANCER TREATMENT Recommendations of the American Society of Clinical Oncology Welcome The American Society of Clinical Oncology
Patient Guide ASCOInformation for People Living with Cancer ADVANCED LUNG CANCER TREATMENT Recommendations of the American Society of Clinical Oncology Welcome The American Society of Clinical Oncology
