Standards and datasets for reporting cancers. Dataset for the histological reporting of mesothelioma. April 2013
|
|
|
- Philomena Cook
- 8 years ago
- Views:
Transcription
1 Standards and datasets for reporting cancers Dataset for the histological reporting of mesothelioma April 2013 Authors Professor Andrew G Nicholson, Royal Brompton Hospital and Harefield NHS Trust Professor John Gosney, Royal Liverpool Hospital, Liverpool Professor Michael Sheaff, Barts Health NHS Trust, London Dr Salli Muller, University Hospitals of Leicester NHS Trust, Leicester Unique document number Document name G134 Dataset for histological reporting of mesothelioma Version number 1 Produced by Professor Andrew G Nicholson, Professor John Gosney, Professor Michael Sheaff and Dr Salli Muller, on behalf of the College s Cancer Services Working Group. All authors are consultant cellular pathologists and specialise in thoracic pathology. AGN is a member of the pathology panel of International Association for the Study of Lung Cancer (IASLC) and a member of the IASLC Staging Committee. AGN and JG are members of the Lung Cancer and Mesothelioma Advisory Group for the Department of Health. MS is a member of the International Mesothelioma Interest Group. Date active April 2013 Date for review April 2014 Comments In accordance with the College s pre-publications policy, this document was put on The Royal College of Pathologists website for consultation from 6 February to 6 March Forty-two items of feedback were received and the authors considered them and amended the document as appropriate. Please publications@rcpath.org if you wish to see the responses and comments. Dr Suzy Lishman Acting Director of Communications The Royal College of Pathologists 2 Carlton House Terrace, London, SW1Y 5AF Tel: Fax: Web: Registered charity in England and Wales, no , The Royal College of Pathologists This work is copyright. You may download, display, print and reproduce this document for your personal, non-commercial use. Apart from any use as permitted under the Copyright Act 1968 or as set out above, all other rights are reserved. Requests and inquiries concerning reproduction and rights should be addressed to The Royal College of Pathologists at the above address. First published: PSU V7 Final
2 Contents Foreword Introduction Clinical information required on the specimen request form Preparation of specimens before dissection Specimen handling and block selection Considerations for microscopy Core data items Non-core data items Diagnostic coding and staging Reporting of cytology specimens Reporting of frozen sections Prognostic and predictive markers Criteria for audit of the dataset References Appendix A TNM staging of mesothelioma Appendix B SNOMED codes Appendix C Reporting proforma for mesothelioma biopsy and cytology specimens Appendix D Reporting proforma for mesothelioma resection specimens Appendix E Summary table Explanation of levels of evidence Appendix F AGREE compliance monitoring sheet NICE has accredited the process used by The Royal College of Pathologists to produce its Cancer Datasets and Tissue Pathways guidance. Accreditation is valid for 5 years from July More information on accreditation can be viewed at For full details on our accreditation visit: PSU V7 Final
3 Foreword The cancer datasets published by The Royal College of Pathologists (RCPath) are a combination of textual guidance, educational information and reporting proformas. The datasets enable pathologists to grade and stage cancers in an accurate, consistent manner in compliance with international standards and provide prognostic information, thereby allowing clinicians to provide a high standard of care for patients and appropriate management for specific clinical circumstances. It may rarely be necessary or even desirable to depart from the guidelines in the interests of specific patients and special circumstances. The clinical risk of departing from the guidelines should be assessed by the relevant multidisciplinary team (MDT); just as adherence to the guidelines may not constitute defence against a claim of negligence, so a decision to deviate from them should not necessarily be deemed negligent. Each dataset contains core data items that are mandated for inclusion in the Cancer Outcomes and Services Dataset (COSD previously the National Cancer Data Set) in England. Core data items are items that are supported by robust published evidence and are required for cancer staging, optimal patient management and prognosis. Core data items meet the requirements of professional standards (as defined by the Information Standards Board for Health and Social Care [ISB]) and it is recommended that at least 90% of reports on cancer resections should record a full set of core data items. Other, non-core, data items are described. These may be included to provide a comprehensive report or to meet local clinical or research requirements. All data items should be clearly defined to allow the unambiguous recording of data. The following stakeholder groups that have been consulted: British Thoracic Oncology Group British Thoracic Society Society for Cardiothoracic Surgery in Great Britain and Ireland. Evidence for the data items in the dataset is derived from consensus of recognised experts, in particular recent guidelines from an internationally convened group of pathologists with a particular interest in mesothelioma, many of whom are part of the International Mesothelioma Interest Group (IMIG), together with review of current literature. Evidence has been graded using modified SIGN guidance see Appendix E. No major organisational changes or cost implications have been identified that would hinder the implementation of the dataset. A formal revision cycle for all cancer datasets takes place on a three-yearly basis. However, each year, the College will ask the authors of the dataset, in conjunction with the relevant sub-specialty adviser to the College, to consider whether or not the dataset needs to be updated or revised. A full consultation process will be undertaken if major revisions are required, i.e. revisions to core data items (the only exception being changes to international tumour grading and staging schemes that have been approved by the Specialty Advisory Committee on Cellular Pathology and affiliated professional bodies; these changes will be implemented without further consultation). If minor revisions or changes to non-core data items are required, an abridged consultation process will be undertaken whereby a short note of the proposed changes will be placed on the College website for two weeks for Fellows attention. If Fellows do not object to the changes, the short notice of change will be incorporated into the dataset and the full revised version (incorporating the changes) will replace the existing version on the College website. All changes will be documented in the data control section of the relevant dataset. This dataset has been reviewed by the WGCS and is being placed on the College website for consultation with the membership from 6 March to 3 April All comments received from the PSU V7 Final
4 WGCS and membership will be addressed by the authors to the satisfaction of the WGCS Chair and the Acting Director of Communications. This dataset was developed without external funding to the writing group. The College requires the authors of datasets to provide a list of potential conflicts of interest; these are monitored by the Professional Standards Unit and are available on request. The authors of this document have declared that there are no conflicts of interest. 1 Introduction Although mesotheliomas may present in the peritoneum and other sites, they most commonly arise in the pleura and this dataset is limited to the reporting of mesothelioma at this site. It is one of the most important occupational diseases, with incidence steadily rising due to its association with exposure to asbestos. It is estimated that numbers in the United Kingdom will continue to rise until around 2020 and only then decline. Because most mesotheliomas are not completely resectable, guidelines for diagnosis from the Royal College have previously been incorporated within the tissue pathway document for thoracic diseases. However, with requirements for all neoplastic disease to be dealt with in cancer datasets, together with rare cases undergoing a variety of surgical resections, (radical pleurectomy, pleuropneumonectomy), a specific cancer dataset is now required. The pleura is a common site for metastatic disease as well as for other rarer primary tumours, such as sarcomas. Although the latter may be considered in the differential diagnosis, this document deals only with the data that are required for mesothelioma. Features in both biopsy and resection specimens should be reported according the following guidelines, as data are important in: a) deciding on the most appropriate treatment for particular patients, including the need and choice of adjuvant therapy b) providing prognostic information to clinicians and patients c) providing more reliable staging than using clinical data alone d) monitoring clinical effectiveness of therapeutic trials e) providing accurate data for cancer registration. International guidelines on the reporting of mesotheliomas have been published by an invited group of pathologists under the aegis of IMIG. 1 The reporting form and guidance in the following pages and the data herein are in part a reflection of this evidence-based work, together with the International Association for the Study of Lung Cancer s (IASLC) recommendations for staging, i.e. the 7 th edition TNM. 2 The purpose of this document is to define the core data that must be recorded for all patients with a histological diagnosis of mesothelioma. These are guidelines that are intended to help pathologists provide local clinicians with the necessary information to manage their patients effectively. Consistency in reporting and staging is improved by the use of standard terminology for example, precise definition of the various subtypes of mesothelioma according to the WHO 2004 classification, 3 together with accurate definition of anatomic parameters related to staging. Given the complexity of the thorax, when faced with the rare occurrence of a resection specimen, discussion with the surgeon is not infrequently required to ensure that information about the pathological staging is accurately delivered. PSU V7 Final
5 Target users and health benefits of this guideline The target primary users of the dataset are trainee and consultant cellular pathologists and, on their behalf, the suppliers of IT products to laboratories. The secondary users are surgeons and oncologists, cancer registries and the National Cancer Intelligence Network. Standardised cancer reporting and multidisciplinary team (MDT) working reduce the risk of histological misdiagnosis and help to ensure that clinicians have all of the relevant pathological information required for tumour staging, management and prognosis. Collection of standardised cancer specific data also provides information for healthcare providers, epidemiologists, and facilitates international benchmarking and research. 2 Clinical information required on the specimen request form Name, date of birth, hospital, hospital number, NHS or CHI number, procedure, specimen type, date of procedure, surgeon/physician should be provided. In addition, the laterality and procedure (biopsy, core needle biopsy, thoracoscopic [VATS] biopsy, thoracotomy, incisional biopsy, pleurectomy, or extrapleural pneumonectomy) should be documented. Details of any previous biopsy or cytology, any previous malignancy, previous treatment such as neoadjuvant chemotherapy and/or radiotherapy must also be recorded. Any exposure to asbestos must be documented, if known. If a diagnostic frozen section was performed, this must be recorded and the intraoperative diagnosis must be documented. 3 Preparation of specimens before dissection The majority of specimens are biopsies and therefore require no more than formalin fixation before processing. The use of electron microscopy has largely been superseded by immunohistochemistry, although the selection of a small piece of tissue for fixation in glutaraldehyde may be undertaken before placing in formalin, if this investigation is going to be undertaken. Small biopsies should be processed in their entirety, with consideration given to using multiple cassettes as cases not infrequently require extensive immunohistochemistry. Debulking specimens (e.g. pleurectomy) should be fixed for 24 hours and then sampled thoroughly. Pleuropneumonectomy specimens are ideally placed in formalin after inflation of the lung via the airways in similar fashion to that undertaken for lung cancer resections. Close correlation with the surgeon is recommended prior to dissection, in order to identify areas of concern regarding completeness of resection and relevant anatomic structures (pericardium, diaphragm, mediastinal fat, etc). [Expert opinion is that good communication between surgeon and pathologist improves the accuracy of determining completeness of resection. Level of evidence D.] 4 Specimen handling and block selection The overall size of biopsies should be measured and documented, and any identifiable tissues included in the specimen (pleura, chest wall adipose tissue and/or skeletal muscle, rib[s], diaphragm, lymph nodes, mediastinal structures, etc.) should be documented. For surgical biopsies, specimens should be sectioned perpendicular to the pleural surface, because orientation in this plane facilitates the diagnosis of desmoplastic variants of mesothelioma. This is because it enables better assessment of variations in cellularity, which are obscured by cross-cutting. In relation to radical pleurectomy and pleuropneumonectomy specimens, the distribution of disease should be described (distribution: diffuse, nodular, localised/solitary), together with PSU V7 Final
6 the extent of pleural involvement (localised, subtotal, circumferential), together with involvement of the fissures and interlobular septa. If there is a dominant tumour mass, its size should be measured and its location identified. If present, additional nodules within the lung or patterns of spread within the lung should be noted. The distance to the nearest resection margin should be documented (lateral soft tissue [chest wall] margin, bronchus, pulmonary vessels, mediastinal structures if included, diaphragm), inking margins where appropriate. As discussed above, this not infrequently requires discussion with the surgeon prior to dissection. In relation to radical pleurectomy specimens, it is important to identify and sample appropriate areas to stage the specimen according to the current TNM guidelines, in particular to identify and sample pericardium and diaphragm as well as the pleura. Abnormalities within the lung parenchyma (e.g. fibrosis, tumour involvement either as nodules or through direct spread) should be noted, as should non-neoplastic abnormalities in pleura and mediastinal tissues (e.g. pleural plaques), although these are not viewed as core items. Asbestos bodies should be looked for. This may be facilitated by normal thickness Perls staining or 25 micrometre-thick unstained sections. In radical pleurectomies, any adherent lung tissue needs to be identified and sampled for this purpose. Finally, if feasible and there is appropriate consent, banking frozen fresh tumour for future research is recommended. [The basis for block selection is extrapolated from the need to provide microscopic confirmation or evaluation of prognostic and predictive factors Level of evidence C.] 5 Considerations for microscopy Histological type Histological typing of mesothelioma is recorded according to the WHO 2004 classification with initial subdivision into epithelioid, biphasic and sarcomatoid variants. Further subdivision into patterns may also be undertaken as some have clinical relevance such as pleomorphic variants, 4 although this is not viewed as a core data item. [Histopathological type is important for clinical management and prognosis, with strength of evidence varying for different types Levels of evidence B D.] If a neoplasm is present, the usual distinction to be made is between mesothelioma and metastatic adenocarcinoma, since these are by far the most common malignant neoplasms at this site. A diastase-pas or combined Alcian Blue/PAS stain for epithelial mucins may therefore be of use. Staining for acidic (connective tissue) mucins alone (Alcian blue +/- hyaluronidase) can also be of value but has largely been superseded by immunohistochemistry. Diagnosis, however, may be impossible with the small amount of tissue usually present in a pleural needle biopsy and further large biopsies may be required, especially for distinguishing reactive from neoplastic infiltrates. In the latter situation, broad spectrum cytokeratins (e.g. AE1/3, MNF116, CAM 5.2) may be useful in identifying extent of invasion, including invasion into fat, when this is difficult to appreciate on H&E. Clinical and radiological features are often invaluable in difficult cases, ideally through multidisciplinary discussion. Other neoplasms also arise in or spread to the pleura. 5 The distinction between epithelioid mesothelioma and metastatic adenocarcinoma cannot be made with confidence on morphological grounds alone and immunohistochemistry is mandatory. Currently none of the antigens indicative of mesothelial or glandular (adenocarcinomatous) differentiation is sufficiently sensitive or specific, so a panel is recommended. This will vary according to the preference of the individual pathologist, but PSU V7 Final
7 recommended markers of mesothelial differentiation include cytokeratins of classes 5 and 6, calretinin, N-cadherin, Wilm s Tumour-1(WT1), D2-40 and thrombomodulin. 1, 6 However, it must be emphasised that the specificity and sensitivity for mesothelioma using these antibodies is significantly reduced in poorly differentiated epithelioid neoplasms, and these data should not be interpreted in isolation from other data. 7 For poorly differentiated epithelioid neoplasms, the use of several broad spectrum cytokeratins may be necessary. Suitable markers of glandular differentiation include epithelial glycoprotein (BerEp4 antibody), CEA and the CD15 antigen. Further immunohistochemistry (e.g. TTF-1, cytokeratin subclasses, hormone receptors) may be required to further define the nature of metastatic adenocarcinoma. A broader panel of antibodies is required to distinguish sarcomatoid mesothelioma from sarcomas (primary and metastatic), and no antibody is 100% specific or sensitive. The most consistently useful is cytokeratin staining, which is positive in 80 90% of sarcomatoid mesotheliomas Genetic analysis may be of diagnostic value in identifying some sarcomas (e.g. X:18 translocation for synovial sarcoma). 11 Referral to regional or national experts is recommended in complex and difficult cases. 6 Core data items 6.1 Clinical Name, date of birth, hospital, hospital number, NHS/CHI number, specimen type, procedure, date of procedure, surgeon/physician should be supplied. Laterality and type of procedure must be documented. Neoadjuvant treatment should be documented, if undertaken. 6.2 Relationship of tumour to other intra-thoracic structures The location of the tumour in the thorax, as well as its relationship to adherent structures, should be recorded. In particular, areas of likely invasion that pertain to staging should be assessed (chest wall, diaphragm, pericardium, lung, great vessels, pericardium, lymph nodes). Separate tumour nodules in the main resection specimen or separately submitted samples (e.g. separate lung or pleural nodules) should also be documented. [Extent of invasion forms part of established staging criteria Level of evidence B.] 6.3 Size of tumour If a single localised mass, the maximum diameter of tumour should be measured to the nearest millimetre. Ideally, three dimensions should be recorded. If there is a dominant mass, this should be measured in similar fashion, with description of other localised nodules or extent of more diffuse confluent disease. 6.4 Pathological Histological type should be stated (epithelioid, biphasic, sarcomatoid (desmoplastic variant if present). Given the need for ancillary investigations to make the diagnosis, the immunohistochemistry panel used should be documented, this being at least two mesothelium-associated markers and two epithelium-associated markers for epithelioid and biphasic tumours (discussed in section 5). For sarcomatoid variants, due to the wide differential diagnosis, the full repertoire of antibodies used should be listed. PSU V7 Final
8 As well as involvement by tumour, background lung should be assessed for the presence of asbestos bodies, although if identified, their presence does not contribute to the diagnosis of mesothelioma, only to its causation. Asbestosis should also be documented, if present. Bronchial and vascular margins of the lung should also be sampled. 6.5 Resections following therapy Gross preparation of a resected specimen after preoperative (neoadjuvant) therapy should follow the same principles outlined for primarily resected specimens. However, it is likely that some of the tumor will have become necrotic and more sections will need to be examined in order to have a valid representation of the histologic appearance. A percentage of remaining viable tumour should be reported. 6.6 Lymph node spread If sampled, the presence or absence of tumour should be recorded. The location of involved nodes (ipsilateral bronchopulmonary and/or hilar lymph nodes (N1), metastasis in the subcarinal lymph nodes and/or ipsilateral internal mammary or mediastinal lymph nodes (N2), metastasis and contralateral mediastinal, internal mammary, or hilar nodes and/or the ipsilateral or contralateral supraclavicular or scalene lymph nodes (N3) must be indicated. 6.7 Margins Any area where there is concern about completeness of resection should be sampled by the pathologist, ideally after discussion with the surgeon, with subsequent reporting on whether or not the margins are clear. Excision will rarely be complete in radical pleurectomies. [Staging criteria above are known to provide important prognostic data that govern postsurgical management Level of evidence B.] 6.8 Metastases The presence or absence of metastases should be documented. 7 Non-core data items Various additional parameters have been recommended, but as yet there is insufficient evidence with regard to influencing patient management for them to be included as core items. They may be prospectively recorded at a local level, according to needs and interest. Further subtyping according to pattern may also be undertaken (tubulopapillary, solid, rhabdoid, pleomorphic, deciduoid, etc.), although this is not viewed as a core element. Abnormalities within the lung parenchyma (e.g. fibrosis, tumour involvement either as nodules or through direct spread) may be noted, as may non-neoplastic abnormalities in pleura and mediastinal tissues (e.g. pleural plaques). The presence of asbestos bodies and pleural plaques should be documented. Extracapsular spread from involved lymph nodes may also be documented Some cases may also have ancillary mutation analysis which, if clinically useful, can be documented in the pathology report. PSU V7 Final
9 8 Diagnostic coding and staging The 7 th TNM staging system is recommended for all resected mesotheliomas (Appendix A). The site, histological diagnosis and procedure should be coded using SNOMED (Appendix B). [Recommendation based on UK expert opinion and those of International Mesothelioma Interest Group Level of evidence D.] 9 Reporting of cytology specimens As with biopsies, cytological findings should be correlated with the clinical and imaging findings to establish whether the available cytological material is sufficient to render a specific diagnosis or a clinically relevant differential diagnosis. If a pleural cytology specimen is positive or suspicious for malignancy, and there is no other specimen, then material should undergo the same ancillary investigations as for biopsies in terms of the differential diagnosis, which ideally is via a cell pellet for histology as this allows preservation of residual material. Identification of an epithelial phenotype will allow a definitive diagnosis of metastatic carcinoma. Identification of a mesothelial phenotype will allow further management decisions in terms of a definitive diagnosis of mesothelioma or further sampling, dependent on the clinical scenario (Appendix D). [Recommendation based on collective opinion of experts Level of evidence D.] 10 Reporting of frozen sections Biopsies of pleura are frequently sent for frozen section, although there must not be an expectation of a definitive diagnosis due to the requirements for ancillary investigations. However, a diagnosis of malignancy can usually be made, which allows the surgeon to undertake intra-operative decisions, such as whether or not to undertake pleurodesis. [Recommendation based on collective opinion of experts Level of evidence D.] 11 Prognostic and predictive markers At present, neither predictive nor prognostic immunohistochemical or molecular markers are recommended for routine use. 12 Criteria for audit of the dataset The following standards are suggested as some of criteria that might be used in periodic reviews of the lung cancer pathology service. Completeness of histopathology reports, expressed as average proportion of the core data items recorded, or as proportion of the reports that successfully include 100% of the items; the standard is that all contain 100% of the items. Specificity and sensitivity of antibodies in diagnostic use and proposed new markers when available. PSU V7 Final
10 Inter- and intra-observer studies in relation to epithelioid, biphasic and sarcomatoid variants. Accuracy of cytology diagnosis via histology correlation. In addition the following audits are recommended by the RCPath as key performance indicators ( SNOMED T, M and P coding: standard 95 % by April 2012 and 100% by April 2014 core data items in report: standard 90% excisions using template or proforma and including dataset information: standard 80% by April 2012 and 90% by April 2014 diagnostic biopsies reported within 7 days: standard 80% by April 2012 and 90% by April 2014 All histopathology specimens reported within 10 calendar days: standard 80% by April 2012 and 90% by April The availability of pathology reports and data at MDT meetings: standard: 90% of cases discussed at MDT meetings where biopsies or resections have been taken should have pathology reports/core data available for discussion at the time of the meeting standard: 90% of cases where pathology has been reviewed for the MDT meeting should have the process of review recorded. 13 References 1. Husain AN, Colby TV, Ordonez NG, Krausz T, Borczuk A, Cagle PT et al. Guidelines for pathologic diagnosis of malignant mesothelioma: a consensus statement from the International Mesothelioma Interest Group. Arch Pathol Lab Med 2009;133: Goldstraw P (editor). Staging Manual in Thoracic Oncology. International Association for the Study of Lung Cancer. Florida, USA: Rx Press, World Health Organization Classification of Tumours. Tumours of the Lung, Pleura, Thymus and Heart. Lyons: IARC Press, Kadota K, Suzuki K, Sima CS, Rusch VW, Adusumilli PS, Travis WD. Pleomorphic epithelioid diffuse malignant pleural mesothelioma: a clinicopathological review and conceptual proposal to reclassify as biphasic or sarcomatoid mesothelioma. J Thorac Oncol 2011;6: Attanoos RL, Gibbs AR. 'Pseudomesotheliomatous' carcinomas of the pleura: a 10-year analysis of cases from the Environmental Lung Disease Research Group, Cardiff. Histopathology 2003;43: Ordonez NG. What are the current best immunohistochemical markers for the diagnosis of epithelioid mesothelioma? A review and update. Hum Pathol 2007;38: Miettinen M, Sarlomo-Rikala M. Expression of calretinin, thrombomodulin, keratin 5, and mesothelin in lung carcinomas of different types - An immunohistochemical analysis of 596 tumors in comparison with epithelioid mesotheliomas of the pleura. Am J Surg Pathol 2003; 27: PSU V7 Final
11 8. Attanoos RL, Dojcinov SD, Webb R, Gibbs AR. Anti-mesothelial markers in sarcomatoid mesothelioma and other spindle cell neoplasms. Histopathology 2000;37: Cagle PT, Truong LD, Roggli VL, Greenberg SD. Immunohistochemical differentiation of sarcomatoid mesotheliomas from other spindle cell neoplasms. Am J Clin Pathol 1989; 92: Montag AG, Pinkus GS, Corson JM. Keratin protein immunoreactivity of sarcomatoid and mixed types of diffuse malignant mesothelioma: an immunoperoxidase study of 30 cases. Hum Pathol 1988;19: Weinbreck N, Vignaud JM, Begueret H, Burke L, Benhattar J, Guillou L et al. SYT-SSX fusion is absent in sarcomatoid mesothelioma allowing its distinction from synovial sarcoma of the pleura. Mod Pathol 2007;20: PSU V7 Final
12 Appendix A TNM staging of mesothelioma (from Staging Manual in Thoracic Oncology, 2009) 2 T ptx pt0 Primary tumour Primary tumour cannot be assessed) No evidence of primary tumour) pt1a Tumour is limited to ipsilateral parietal with or without mediastinal, with or without diaphragmatic pleural involvement. No involvement of the visceral pleura pt1b Tumour is limited to ipsilateral parietal with or without mediastinal, with or without diaphragmatic pleural involvement. Tumour involves the visceral pleura pt2 pt3 pt4 Tumour involves each of the ipsilateral pleural surfaces with at least one of the following features: - invasion of diaphragmatic muscle - extension of tumour from visceral pleura from visceral pleura into underlying lung parenchyma Tumour involves all of the ipsilateral pleural surfaces with at least one of the following: - invasion of the endothoracic fascia - invasion into mediastinal fat - solitary completely resectable focus of tumour invading the soft tissues of the chest wall - non-transmural involvement of the pericardium Tumour involves all of the ipsilateral pleural surfaces with at least one of the following: - diffuse or multifocal invasion of the soft tissues of the chest wall - diffuse extension or multifocal masses of tumour within the chest wall, with or without associated rib destruction - direct transdiaphragmatic extension of tumour to the peritoneum - direct extension of tumour into mediastinal organs - direct extension of tumour to the contralateral pleura - direct extension of tumour into the spine - tumour extending through to the internal surface of the pericardium, with or without a pericardial effusion or tumour involving myocardium N pnx pn0 pn1 pn2 pn3 Regional lymph nodes Regional lymph nodes cannot be assessed No regional lymph node metastasis Metastasis in ipsilateral bronchopulmonary and/or hilar lymph nodes Metastasis in the subcarinal or ipsilateral mediastinal lymph nodes, including the ipsilateral internal mammary and peridiaphragmatic lymph nodes Metastasis in contralateral mediastinal, contralateral internal mammary, ipsilateral or contralateral supraclavicular lymph nodes PSU V7 Final
13 M pm0 pm1 Distant metastasis No distant metastasis Distant metastasis T3 describes locally advanced but potentially resectable tumour T4 describes locally advanced, technically unresectable tumour Stage grouping Stage I T1 N0 M0 Stage Ia T1a N0 M0 Stage Ib T1b N0 M0 Stage II T2 N0 M0 Stage III T1, T2 N1 M0 T1, T2 N2 M0 T3 N0, N1, N2 M0 Stage IV T4 Any N M0 Any T N3 M0 Any T Any N M1 PSU V7 Final
14 Appendix B SNOMED codes 3 Topography Pleura T29000 Morphology Mesothelioma (diffuse or localised) M9050/3 Sarcomatoid (inc. desmoplastic) mesothelioma M9051/3 Epithelioid mesothelioma M9052/3 Biphasic mesothelioma M9053/3 Well-differentiated papillary mesothelioma M9052/1 Adenomatoid tumour M9054/0 Procedure Local P codes should be recorded. At present, P codes vary according to the SNOMED system in use in different institutions. PSU V7 Final
15 Appendix C Reporting proforma for mesothelioma biopsy/cytology specimens Surname. Forenames.. Date of birth.. Sex... Hospital.. Hospital no.. NHS/CHI no.. Date of receipt.. Date of reporting.... Report no... Pathologist. Surgeon... Lab no... Previous treatment (neoadjuvant chemotherapy/radiotherapy) Yes No Specimen origin Pleura Lung Other.... Sample type* (more than one box may be ticked) Biopsy Cytology Pleural biopsy Core needle biopsy VATS biopsy Open biopsy Lymph node biopsy Specify site(s)..... Other site(s) Details... Pleural effusion Pericardial effusion Other Details.. FNA Details.. Microscopic features Histological type of mesothelioma Epithelioid Biphasic Sarcomatoid Desmoplastic variant Yes No Ancillary investigations D-PAS mucin staining Positive Negative Alcian Blue mucin staining Positive Negative Immunohistochemistry (list antibodies used minimum of four recommended) Calretinin Positive Negative Cytokeratin 5/6 Positive Negative BerEP4 Positive Negative CEA Positive Negative (Other:... Positive Negative ) Comments: SNOMED codes: Signature.... Date.../..../... PSU V7 Final
16 Appendix D Reporting proforma for mesothelioma resection specimens Surname. Forenames.. Date of birth.. Sex... Hospital.. Hospital no.. NHS/CHI no.. Date of receipt.. Date of reporting.... Report no... Pathologist. Surgeon... Lab no... Previous treatment (neoadjuvant chemotherapy/radiotherapy) Yes No Laterality Right Left Specimen type Pleurectomy Radical pleurectomy Local chest wall/pleural resection Pleuropneumonectomy Additional nodules (specify number and sites) Submitted material Parietal pleura Yes No Visceral pleura Yes No Diaphragm Yes No Endothoracic fascia Yes No Lung Yes No Details... Mediastinal fat Yes No Chest wall Yes No Pericardium Yes No Rib Yes No Peritoneum Yes No Details... Contralateral pleura Yes No Spine Yes No Histological type of mesothelioma Epithelioid Biphasic Sarcomatoid Desmoplastic variant Yes No Tumour size (if localised).mm Ancillary investigations D-PAS mucin staining Positive Negative Alcian Blue mucin staining Positive Negative Immunohistochemistry (list antibodies used minimum of four recommended) Calretinin Positive Negative Cytokeratin 5/6 Positive Negative BerEP4 Positive Negative CEA Positive Negative (Other:... Positive Negative ) Macroscopic/microscopic features Tumour involves parietal pleura without involvement of the ipsilateral visceral pleura Yes No N/A Tumour involves parietal pleura with involvement of the ipsilateral visceral pleura Yes No N/A Direct invasion of but not through the diaphragm Yes No N/A Direct invasion of lung parenchyma Yes No N/A Direct invasion of endothoracic fascia Yes No N/A Direct invasion of mediastinal fat Yes No N/A Localised focus of tumour invading the soft tissue of the chest wall Yes No N/A PSU V7 Final
17 Direct invasion of but not through the pericardium Yes No N/A Diffuse or multiple foci of the tumour invading the soft tissue of the chest wall Yes No N/A Direct invasion of rib Yes No N/A Direct invasion of the peritoneum through the diaphragm Yes No N/A Direct invasion of great vessels/oesophagus/trachea/other(mediastinal organ) Yes No N/A Extension into contralateral pleura Yes No N/A Direct invasion of the spine Yes No N/A Direct involvement through the pericardium Yes No N/A Direct invasion of the myocardium Yes No N/A Lymph node involvement Ipsilateral bronchopulmonary and/or hilar lymph nodes Not submitted Submitted Involved Subcarinal or ipsilateral mediastinal lymph nodes, including the ipsilateral internal mammary and peridiaphragmatic lymph nodes Not submitted Submitted Involved Contralateral mediastinal, contralateral internal mammary, ipsilateral or contralateral supraclavicular lymph nodes Not submitted Submitted Involved If neoadjuvant therapy, % of viable tumour on cross-section... Margins Excision complete (R0) Yes No N/A Microscopic involvement (R1) Yes No N/A Macroscopic involvement (R2) Yes No N/A Sites of involvement if R1 or R2: Closest margin if excision complete:... distance...mm Site(s) of incomplete resection Metastases Unknown Absent Present Details: Background lung (if sampled) Asbestos bodies Yes No N/A Asbestosis Yes No N/A Summary of pathological staging (select highest stage from above data): SNOMED code: Comments: Signature.... Date.../..../... PSU V7 Final
18 Appendix E Summary table Explanation of levels of evidence (modified from Palmer K et al. BMJ 2008; 337:1832) Level of evidence Level A Level B Level C Level D Good practice point (GPP) Nature of evidence At least one high-quality meta-analysis, systematic review of randomised controlled trials or a randomised controlled trial with a very low risk of bias and directly attributable to the target cancer type, or A body of evidence demonstrating consistency of results and comprising mainly well-conducted meta-analyses, systematic reviews of randomised controlled trials or randomised controlled trials with a low risk of bias, directly applicable to the target cancer type. A body of evidence demonstrating consistency of results and comprising mainly high-quality systematic reviews of case-control or cohort studies and high-quality case-control or cohort studies with a very low risk of confounding or bias and a high probability that the relation is causal and which are directly applicable to the target cancer type or, Extrapolation evidence from studies described in A. A body of evidence demonstrating consistency of results and including wellconducted case-control or cohort studies and high quality case-control or cohort studies with a low risk of confounding or bias and a moderate probability that the relation is causal and which are directly applicable to the target cancer type or, Extrapolation evidence from studies described in B. Non-analytic studies such as case reports, case series or expert opinion or, Extrapolation evidence from studies described in C. Recommended best practice based on the clinical experience of the authors of the writing group PSU V7 Final
19 Appendix F AGREE compliance monitoring sheet The cancer datasets of The Royal College of Pathologists comply with the AGREE standards for good quality clinical guidelines. The sections of this dataset that indicate compliance with each of the AGREE standards are indicated in the table. AGREE Standard Scope and purpose 1 The overall objective(s) of the guideline is (are) specifically described 1 2 The clinical question(s) covered by the guidelines is (are) specifically described 1 3 The patients to whom the guideline is meant to apply are specifically described 1 Stakeholder involvement 4 The guideline development group includes individuals from all the relevant professional groups 5 The patients views and preferences have been sought n/a Section of dataset 6 The target users of the guideline are clearly defined Foreword 2 7 The guideline has been piloted among target users 1 Rigour of development 8 Systematic methods were used to search for evidence Foreword 9 The criteria for selecting the evidence are clearly described Foreword 10 The methods used for formulating the recommendations are clearly described Foreword 11 The health benefits, side effects and risks have been considered in formulating the recommendations 12 There is an explicit link between the recommendations and the supporting evidence 2 Foreword 4 9,13 13 The guideline has been externally reviewed by experts prior to its publication Foreword 14 A procedure for updating the guideline is provided Foreword Clarity of presentation 15 The recommendations are specific and unambiguous The different options for management of the condition are clearly presented Key recommendations are easily identifiable The guideline is supported with tools for application Appendices A D Applicability 19 The potential organisational barriers in applying the recommendations have been discussed 20 The potential cost implications of applying the recommendations have been considered 21 The guideline presents key review criteria for monitoring and/audit purposes 12 Editorial independence Foreword Foreword 22 The guideline is editorially independent from the funding body Foreword 23 Conflicts of interest of guideline development members have been recorded Foreword *The Lay Advisory Committee (LAC) of The Royal College of Pathologists has advised the Director of Communications that there is no reason to consult directly with patients or the public regarding this dataset because it is technical in nature and intended to guide pathologists in their practice. The authors will refer to the LAC for further advice if necessary. PSU V7 Final
Recommendations for the Reporting of Pleural Mesothelioma
 Recommendations for the Reporting of Pleural Mesothelioma Association of Directors of Anatomic and Surgical Pathology * DOI: 10.1309/6A30YQHBMTHEJTEM It has been evident for decades that pathology reports
Recommendations for the Reporting of Pleural Mesothelioma Association of Directors of Anatomic and Surgical Pathology * DOI: 10.1309/6A30YQHBMTHEJTEM It has been evident for decades that pathology reports
Update on Mesothelioma
 November 8, 2012 Update on Mesothelioma Intro incidence and nomenclature Update on Classification Diagnostic specimens Morphologic features Epithelioid Histology Biphasic Histology Immunohistochemical
November 8, 2012 Update on Mesothelioma Intro incidence and nomenclature Update on Classification Diagnostic specimens Morphologic features Epithelioid Histology Biphasic Histology Immunohistochemical
Extrapleural Pneumonectomy for Malignant Mesothelioma: Pro. Joon H. Lee 9/17/2012
 Extrapleural Pneumonectomy for Malignant Mesothelioma: Pro Joon H. Lee 9/17/2012 Malignant Pleural Mesothelioma (Epidemiology) Incidence: 7/mil (Japan) to 40/mil (Australia) Attributed secondary to asbestos
Extrapleural Pneumonectomy for Malignant Mesothelioma: Pro Joon H. Lee 9/17/2012 Malignant Pleural Mesothelioma (Epidemiology) Incidence: 7/mil (Japan) to 40/mil (Australia) Attributed secondary to asbestos
Protocol for the Examination of Specimens From Patients With Malignant Pleural Mesothelioma
 Protocol for the Examination of Specimens From Patients With Malignant Pleural Mesothelioma Based on AJCC/UICC TNM, 7th edition Protocol web posting date: February 1, 2011 Procedures Resection Authors
Protocol for the Examination of Specimens From Patients With Malignant Pleural Mesothelioma Based on AJCC/UICC TNM, 7th edition Protocol web posting date: February 1, 2011 Procedures Resection Authors
Objectives. Mylene T. Truong, MD. Malignant Pleural Mesothelioma Background
 Imaging of Pleural Tumors Mylene T. Truong, MD Imaging of Pleural Tumours Mylene T. Truong, M. D. University of Texas M.D. Anderson Cancer Center, Houston, TX Objectives To review tumors involving the
Imaging of Pleural Tumors Mylene T. Truong, MD Imaging of Pleural Tumours Mylene T. Truong, M. D. University of Texas M.D. Anderson Cancer Center, Houston, TX Objectives To review tumors involving the
Thoracic Mesothelium. Protocol applies to malignant thoracic mesothelioma. Procedures Cytology (No Accompanying Checklist) Incisional Biopsy Resection
 Thoracic Mesothelium Protocol applies to malignant thoracic mesothelioma. Protocol revision date: January 2004 Based on AJCC/UICC TNM, 6 th edition Procedures Cytology (No Accompanying Checklist) Incisional
Thoracic Mesothelium Protocol applies to malignant thoracic mesothelioma. Protocol revision date: January 2004 Based on AJCC/UICC TNM, 6 th edition Procedures Cytology (No Accompanying Checklist) Incisional
INTERNATIONAL ASSOCIATION FOR THE STUDY OF LUNG CANCER Prospective Mesothelioma Staging Project
 INTERNATIONAL ASSOCIATION FOR THE STUDY OF LUNG CANCER Prospective Mesothelioma Staging Project Data Forms and Fields in CRAB Electronic Data Capture System - Reduced Set - Pivotal data elements for developing
INTERNATIONAL ASSOCIATION FOR THE STUDY OF LUNG CANCER Prospective Mesothelioma Staging Project Data Forms and Fields in CRAB Electronic Data Capture System - Reduced Set - Pivotal data elements for developing
Diagnosis of Mesothelioma Pitfalls and Practical Information
 Diagnosis of Mesothelioma Pitfalls and Practical Information Mary Beth Beasley, M.D. Mt Sinai Medical Ctr Dept of Pathology One Gustave L Levy Place New York, NY 10029 (212) 241-5307 mbbeasleymd@yahoo.com
Diagnosis of Mesothelioma Pitfalls and Practical Information Mary Beth Beasley, M.D. Mt Sinai Medical Ctr Dept of Pathology One Gustave L Levy Place New York, NY 10029 (212) 241-5307 mbbeasleymd@yahoo.com
Pathology of lung cancer
 Pathology of lung cancer EASO COURSE ON LUNG CANCER AND MESOTHELIOMA DAMASCUS (SYRIA), MAY 3-4, 2007 Gérard ABADJIAN MD Pathologist Associate Professor, Saint Joseph University Pathology Dept. Hôtel-Dieu
Pathology of lung cancer EASO COURSE ON LUNG CANCER AND MESOTHELIOMA DAMASCUS (SYRIA), MAY 3-4, 2007 Gérard ABADJIAN MD Pathologist Associate Professor, Saint Joseph University Pathology Dept. Hôtel-Dieu
MALIGNANT MESOTHELIOMA UPDATE ON PATHOLOGY AND IMMUNOHISTOCHEMISTRY
 MALIGNANT MESOTHELIOMA CLASSIFICATION MALIGNANT MESOTHELIOMA UPDATE ON PATHOLOGY AND IMMUNOHISTOCHEMISTRY Sisko Anttila, MD, PhD Jorvi Hospital Laboratory of Pathology Helsinki University Hospital Espoo,
MALIGNANT MESOTHELIOMA CLASSIFICATION MALIGNANT MESOTHELIOMA UPDATE ON PATHOLOGY AND IMMUNOHISTOCHEMISTRY Sisko Anttila, MD, PhD Jorvi Hospital Laboratory of Pathology Helsinki University Hospital Espoo,
MALIGNANT MESOTHELIOMA UPDATE ON PATHOLOGY AND IMMUNOHISTOCHEMISTRY
 MALIGNANT MESOTHELIOMA UPDATE ON PATHOLOGY AND IMMUNOHISTOCHEMISTRY Sisko Anttila, MD, PhD Jorvi Hospital Laboratory of Pathology Helsinki University Hospital Espoo, Finland 2nd Nordic Conference on Applied
MALIGNANT MESOTHELIOMA UPDATE ON PATHOLOGY AND IMMUNOHISTOCHEMISTRY Sisko Anttila, MD, PhD Jorvi Hospital Laboratory of Pathology Helsinki University Hospital Espoo, Finland 2nd Nordic Conference on Applied
Case of the. Month October, 2012
 Case of the Month October, 2012 Case The patient is a 47-year-old male with a 3-week history of abdominal pain. A CT scan of the abdomen revealed a suggestion of wall thickening at the tip of the appendix
Case of the Month October, 2012 Case The patient is a 47-year-old male with a 3-week history of abdominal pain. A CT scan of the abdomen revealed a suggestion of wall thickening at the tip of the appendix
Mesothelioma. 1. Introduction. 1.1 General Information and Aetiology
 Mesothelioma 1. Introduction 1.1 General Information and Aetiology Mesotheliomas are tumours that arise from the mesothelial cells of the pleura, peritoneum, pericardium or tunica vaginalis [1]. Most are
Mesothelioma 1. Introduction 1.1 General Information and Aetiology Mesotheliomas are tumours that arise from the mesothelial cells of the pleura, peritoneum, pericardium or tunica vaginalis [1]. Most are
How To Write Lung Cancer Data Standards For Nhs Scotland
 For reference only Do Not Use For more information contact: cdsis@nhs.net Lung Cancer Pathology Data Standards June 2008 National Clinical Dataset Development Programme (NCDDP) Support Team Information
For reference only Do Not Use For more information contact: cdsis@nhs.net Lung Cancer Pathology Data Standards June 2008 National Clinical Dataset Development Programme (NCDDP) Support Team Information
PATHOLOGY OF THE PLEURA: Mesothelioma and mimickers Necessity of Immunohistochemistry. M. Praet
 PATHOLOGY OF THE PLEURA: Mesothelioma and mimickers Necessity of Immunohistochemistry M. Praet Pathology of the Pleura Normal serosa: visceral and parietal layers Inflammation Neoplasia: Primary: mesothelioma
PATHOLOGY OF THE PLEURA: Mesothelioma and mimickers Necessity of Immunohistochemistry M. Praet Pathology of the Pleura Normal serosa: visceral and parietal layers Inflammation Neoplasia: Primary: mesothelioma
Notice of Faculty Disclosure
 The Diagnosis of Malignant Mesothelioma Andrew Churg, MD Department of Pathology University of British Columbia Vancouver, BC, Canada achurg@mail.ubc.ca Notice of Faculty Disclosure In accordance with
The Diagnosis of Malignant Mesothelioma Andrew Churg, MD Department of Pathology University of British Columbia Vancouver, BC, Canada achurg@mail.ubc.ca Notice of Faculty Disclosure In accordance with
بسم هللا الرحمن الرحيم
 بسم هللا الرحمن الرحيم Updates in Mesothelioma By Samieh Amer, MD Professor of Cardiothoracic Surgery Faculty of Medicine, Cairo University History Wagner and his colleagues (1960) 33 cases of mesothelioma
بسم هللا الرحمن الرحيم Updates in Mesothelioma By Samieh Amer, MD Professor of Cardiothoracic Surgery Faculty of Medicine, Cairo University History Wagner and his colleagues (1960) 33 cases of mesothelioma
The develpemental origin of mesothelium
 Mesothelioma Tallinn 14.12.06 Henrik Wolff Finnish Institute of Occupational Health The develpemental origin of mesothelium Mesodermal cavities (pleura, peritoneum and pericardium ) are lined with mesenchymal
Mesothelioma Tallinn 14.12.06 Henrik Wolff Finnish Institute of Occupational Health The develpemental origin of mesothelium Mesodermal cavities (pleura, peritoneum and pericardium ) are lined with mesenchymal
Primary -Benign - Malignant Secondary
 TUMOURS OF THE LUNG Primary -Benign - Malignant Secondary The incidence of lung cancer has been increasing almost logarithmically and is now reaching epidemic levels. The overall cure rate is very low
TUMOURS OF THE LUNG Primary -Benign - Malignant Secondary The incidence of lung cancer has been increasing almost logarithmically and is now reaching epidemic levels. The overall cure rate is very low
The National Clinical Lung Cancer Audit (LUCADA)
 The National Clinical Lung Cancer Audit (LUCADA) DATA MANUAL Title: Version: 3.1.5 Date: September 2013 LUCADA Lung Cancer Audit VERSION HISTORY Version Date Issued Brief Summary of Change Owner s Name
The National Clinical Lung Cancer Audit (LUCADA) DATA MANUAL Title: Version: 3.1.5 Date: September 2013 LUCADA Lung Cancer Audit VERSION HISTORY Version Date Issued Brief Summary of Change Owner s Name
Malignant Mesothelioma Current Approaches to a Difficult Problem. Raja M Flores, MD Thoracic Surgery Memorial Sloan-Kettering Cancer Center
 Malignant Mesothelioma Current Approaches to a Difficult Problem Raja M Flores, MD Thoracic Surgery Memorial Sloan-Kettering Cancer Center Malignant Pleural Mesothelioma Clinical Presentation Insidious
Malignant Mesothelioma Current Approaches to a Difficult Problem Raja M Flores, MD Thoracic Surgery Memorial Sloan-Kettering Cancer Center Malignant Pleural Mesothelioma Clinical Presentation Insidious
Practical Effusion Cytology
 Practical Effusion Cytology A Community Pathologist s Approach to Immunocytochemistry in Body Fluid Cytology Emily E. Volk, MD William Beaumont Hospital Troy, MI College of American Pathologists 2004.
Practical Effusion Cytology A Community Pathologist s Approach to Immunocytochemistry in Body Fluid Cytology Emily E. Volk, MD William Beaumont Hospital Troy, MI College of American Pathologists 2004.
3-F. Pathology of Mesothelioma
 3-F. Pathology of Mesothelioma Kouki Inai Professor of Department of Pathology, Graduate School of Biomedical Science, Hiroshima University Introduction Mesothelioma is a peculiar type of malignancy, which
3-F. Pathology of Mesothelioma Kouki Inai Professor of Department of Pathology, Graduate School of Biomedical Science, Hiroshima University Introduction Mesothelioma is a peculiar type of malignancy, which
Protocol for the Examination of Specimens From Patients With Tumors of the Peritoneum
 Protocol for the Examination of Specimens From Patients With Tumors of the Peritoneum Protocol applies to all primary borderline and malignant epithelial tumors and malignant mesothelial neoplasms of the
Protocol for the Examination of Specimens From Patients With Tumors of the Peritoneum Protocol applies to all primary borderline and malignant epithelial tumors and malignant mesothelial neoplasms of the
Proposed Adjustments to Pathologic Staging of Epithelial Malignant Pleural Mesothelioma Based on Analysis of 354 Cases
 Original Article Proposed Adjustments to Pathologic Staging of Epithelial Malignant Pleural Mesothelioma Based on Analysis of 354 Cases William G. Richards, PhD 1,2 ; John J. Godleski, MD 2,3 ; Beow Y.
Original Article Proposed Adjustments to Pathologic Staging of Epithelial Malignant Pleural Mesothelioma Based on Analysis of 354 Cases William G. Richards, PhD 1,2 ; John J. Godleski, MD 2,3 ; Beow Y.
PRINCESS MARGARET CANCER CENTRE CLINICAL PRACTICE GUIDELINES
 PRINCESS MARGARET CANCER CENTRE CLINICAL PRACTICE GUIDELINES LUNG SITE MESOTHELIOMA Lung Site Group Mesothelioma Date Guideline Created: April 2013 Authors: Dr. Meredith Giuliani, Dr. Andrea Bezjak 1.
PRINCESS MARGARET CANCER CENTRE CLINICAL PRACTICE GUIDELINES LUNG SITE MESOTHELIOMA Lung Site Group Mesothelioma Date Guideline Created: April 2013 Authors: Dr. Meredith Giuliani, Dr. Andrea Bezjak 1.
Case based applications part III
 Case based applications part III Los Angeles Society Of Pathologists January 25, 2014 Sanja Dacic, MD, PhD University of Pittsburgh Medical Center 1 CASE 1 A 44-year-old woman with multiple lung nodules.
Case based applications part III Los Angeles Society Of Pathologists January 25, 2014 Sanja Dacic, MD, PhD University of Pittsburgh Medical Center 1 CASE 1 A 44-year-old woman with multiple lung nodules.
Malignant Pleural Diseases Advances Clinicians Should Know F Gleeson
 Malignant Pleural Diseases Advances Clinicians Should Know F Gleeson The following relevant disclosures, conflicts of interest and/ or financial relationships exist related to this presentation: Consultant
Malignant Pleural Diseases Advances Clinicians Should Know F Gleeson The following relevant disclosures, conflicts of interest and/ or financial relationships exist related to this presentation: Consultant
Protocol applies to all primary borderline and malignant epithelial tumors, and malignant mesothelial neoplasms of the peritoneum.
 Peritoneum Protocol applies to all primary borderline and malignant epithelial tumors, and malignant mesothelial neoplasms of the peritoneum. Protocol revision date: January 2004 No AJCC/UICC staging system
Peritoneum Protocol applies to all primary borderline and malignant epithelial tumors, and malignant mesothelial neoplasms of the peritoneum. Protocol revision date: January 2004 No AJCC/UICC staging system
The Proposed New International TNM Staging System for Malignant Pleural Mesothelioma: Application to Imaging
 323.....:. #{149} :. #{149}..: #{149}. #{149}. :- : Received June 22, 1995; accepted after revision September 12, 1995. 1 Department of Radiology, Duke University Medical Center, Box 3808, Dunham, NC 2771
323.....:. #{149} :. #{149}..: #{149}. #{149}. :- : Received June 22, 1995; accepted after revision September 12, 1995. 1 Department of Radiology, Duke University Medical Center, Box 3808, Dunham, NC 2771
YOUR LUNG CANCER PATHOLOGY REPORT
 UNDERSTANDING YOUR LUNG CANCER PATHOLOGY REPORT 1-800-298-2436 LungCancerAlliance.org A GUIDE FOR THE PATIENT 1 CONTENTS What is a Pathology Report?...3 The Basics...4 Sections of a Pathology Report...7
UNDERSTANDING YOUR LUNG CANCER PATHOLOGY REPORT 1-800-298-2436 LungCancerAlliance.org A GUIDE FOR THE PATIENT 1 CONTENTS What is a Pathology Report?...3 The Basics...4 Sections of a Pathology Report...7
Seattle. Case Presentations. Case 1. 76 year old female with a history of breast cancer 12 years ago. Now presents with a pleural effusion.
 Seattle Montreal IAP September 2006 Case Presentations Allen M. Gown, M.D. Medical Director and Chief Pathologist PhenoPath Laboratories Clinical Professor of Pathology University of British Columbia Case
Seattle Montreal IAP September 2006 Case Presentations Allen M. Gown, M.D. Medical Director and Chief Pathologist PhenoPath Laboratories Clinical Professor of Pathology University of British Columbia Case
Ahmed Farouk Abd El-Hafez, MD
 Presented By Ahmed Farouk Abd El-Hafez, MD Lecturer of Cardiothoracic Surgery Assiut University Asbestos exposure : amphibole fibers especially crocidolite asbestos Nonasbestos Causes : Silicate fibers
Presented By Ahmed Farouk Abd El-Hafez, MD Lecturer of Cardiothoracic Surgery Assiut University Asbestos exposure : amphibole fibers especially crocidolite asbestos Nonasbestos Causes : Silicate fibers
Round Table with Drs. Anne Tsao and Alex Farivar, Case 2: Mesothelioma
 Round Table with Drs. Anne Tsao and Alex Farivar, Case 2: Mesothelioma I d like to welcome everyone, thanks for coming out to our lunch with experts. The faculty today are great people in the thoracic
Round Table with Drs. Anne Tsao and Alex Farivar, Case 2: Mesothelioma I d like to welcome everyone, thanks for coming out to our lunch with experts. The faculty today are great people in the thoracic
Plueral Malignancy: Radiologic-pathologic
 Plueral Malignancy: Radiologic-pathologic Correlation Ritu R. Gill, MD Pleural Malignancies: Radiologic-Pathologic Correlation Ritu R Gill MD Brigham and Women s Hospital Boston, Massachusetts Pleural
Plueral Malignancy: Radiologic-pathologic Correlation Ritu R. Gill, MD Pleural Malignancies: Radiologic-Pathologic Correlation Ritu R Gill MD Brigham and Women s Hospital Boston, Massachusetts Pleural
Lung Carcinomas New 2015 WHO Classification. Spasenija Savic Pathology
 Lung Carcinomas New 2015 WHO Classification Spasenija Savic Pathology ***EXPECTED SPRING 2015*** This authoritative, concise reference book provides an international standard for oncologists and pathologists
Lung Carcinomas New 2015 WHO Classification Spasenija Savic Pathology ***EXPECTED SPRING 2015*** This authoritative, concise reference book provides an international standard for oncologists and pathologists
Malignant Mesothelioma
 Malignant Malignant mesothelioma is a tumour originating from mesothelial cells. 85 95% of mesotheliomas are caused by asbestos exposure. It occurs much more commonly in the chest (malignant pleural mesothelioma)
Malignant Malignant mesothelioma is a tumour originating from mesothelial cells. 85 95% of mesotheliomas are caused by asbestos exposure. It occurs much more commonly in the chest (malignant pleural mesothelioma)
Malignant Mesothelioma
 Malignant mesothelioma is a tumour originating from mesothelial cells. 85 95% of mesotheliomas are caused by asbestos exposure. It occurs much more commonly in the chest (malignant pleural mesothelioma)
Malignant mesothelioma is a tumour originating from mesothelial cells. 85 95% of mesotheliomas are caused by asbestos exposure. It occurs much more commonly in the chest (malignant pleural mesothelioma)
Treatment of mesothelioma in Bloemfontein, South Africa
 European Journal of Cardio-thoracic Surgery 24 (2003) 434 440 www.elsevier.com/locate/ejcts Treatment of mesothelioma in Bloemfontein, South Africa W.J. de Vries*, M.A. Long Cardiothoracic Department,
European Journal of Cardio-thoracic Surgery 24 (2003) 434 440 www.elsevier.com/locate/ejcts Treatment of mesothelioma in Bloemfontein, South Africa W.J. de Vries*, M.A. Long Cardiothoracic Department,
ASBESTOS EXPOSURE AND SARCOMATOID MALIGNANT PLEURAL MESOTHELIOMA Gorantla Sambasivarao 1, Namballa Usharani 2, Tupakula Suresh Babu 3
 ASBESTOS EXPOSURE AND SARCOMATOID MALIGNANT PLEURAL MESOTHELIOMA Gorantla Sambasivarao 1, Namballa Usharani 2, Tupakula Suresh Babu 3 HOW TO CITE THIS ARTICLE: Gorantla Sambasivarao, Namballa Usharani,
ASBESTOS EXPOSURE AND SARCOMATOID MALIGNANT PLEURAL MESOTHELIOMA Gorantla Sambasivarao 1, Namballa Usharani 2, Tupakula Suresh Babu 3 HOW TO CITE THIS ARTICLE: Gorantla Sambasivarao, Namballa Usharani,
A Cytokeratin- and Calretinin-negative Staining Sarcomatoid Malignant Mesothelioma
 A Cytokeratin- and Calretinin-negative Staining Sarcomatoid Malignant Mesothelioma MICHAEL G. HURTUK and MICHELE CARBONE Cardinal Bernadin Cancer Center, Cancer Immunology Program, Department of Pathology,
A Cytokeratin- and Calretinin-negative Staining Sarcomatoid Malignant Mesothelioma MICHAEL G. HURTUK and MICHELE CARBONE Cardinal Bernadin Cancer Center, Cancer Immunology Program, Department of Pathology,
Neoplasms of the LUNG and PLEURA
 Neoplasms of the LUNG and PLEURA 2015-2016 FCDS Educational Webcast Series Steven Peace, BS, CTR September 19, 2015 2015 Focus o Anatomy o SSS 2000 o MPH Rules o AJCC TNM 1 Case 1 Case Vignette HISTORY:
Neoplasms of the LUNG and PLEURA 2015-2016 FCDS Educational Webcast Series Steven Peace, BS, CTR September 19, 2015 2015 Focus o Anatomy o SSS 2000 o MPH Rules o AJCC TNM 1 Case 1 Case Vignette HISTORY:
SMALL CELL LUNG CANCER
 Protocol for Planning and Treatment The process to be followed in the management of: SMALL CELL LUNG CANCER Patient information given at each stage following agreed information pathway 1. DIAGNOSIS New
Protocol for Planning and Treatment The process to be followed in the management of: SMALL CELL LUNG CANCER Patient information given at each stage following agreed information pathway 1. DIAGNOSIS New
Research Article Frequency of Surgery in Black Patients with Malignant Pleural Mesothelioma
 Disease Markers Volume 2015, Article ID 282145, 5 pages http://dx.doi.org/10.1155/2015/282145 Research Article Frequency of Surgery in Black Patients with Malignant Pleural Mesothelioma Emanuela Taioli,
Disease Markers Volume 2015, Article ID 282145, 5 pages http://dx.doi.org/10.1155/2015/282145 Research Article Frequency of Surgery in Black Patients with Malignant Pleural Mesothelioma Emanuela Taioli,
Male. Female. Death rates from lung cancer in USA
 Male Female Death rates from lung cancer in USA Smoking represents an interesting combination of an entrenched industry and a clearly drug-induced cancer Tobacco Use in the US, 1900-2000 5000 100 Per Capita
Male Female Death rates from lung cancer in USA Smoking represents an interesting combination of an entrenched industry and a clearly drug-induced cancer Tobacco Use in the US, 1900-2000 5000 100 Per Capita
Académie internationale de Pathologie - Division arabe XX ème congrès 24-26 novembre 2008 Alger. Immunohistochemistry in malignant mesotheliomas
 Académie internationale de Pathologie - Division arabe XX ème congrès 24-26 novembre 2008 Alger Immunohistochemistry in malignant mesotheliomas Françoise Thivolet-Béjui Groupement Hospitalier Est Lyon-Bron
Académie internationale de Pathologie - Division arabe XX ème congrès 24-26 novembre 2008 Alger Immunohistochemistry in malignant mesotheliomas Françoise Thivolet-Béjui Groupement Hospitalier Est Lyon-Bron
Chapter 2 Staging of Breast Cancer
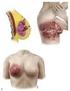 Chapter 2 Staging of Breast Cancer Zeynep Ozsaran and Senem Demirci Alanyalı 2.1 Introduction Five decades ago, Denoix et al. proposed classification system (tumor node metastasis [TNM]) based on the dissemination
Chapter 2 Staging of Breast Cancer Zeynep Ozsaran and Senem Demirci Alanyalı 2.1 Introduction Five decades ago, Denoix et al. proposed classification system (tumor node metastasis [TNM]) based on the dissemination
Sternotomy and removal of the tumor
 Sternotomy and removal of the tumor All thymomas originate from epithelial thymic cells 4% of them consist of a pure population of epithelial cells Most have mixed populations of lymphoid cells to a
Sternotomy and removal of the tumor All thymomas originate from epithelial thymic cells 4% of them consist of a pure population of epithelial cells Most have mixed populations of lymphoid cells to a
Pleural Mesothelioma: An Institutional Experience of 66 Cases
 The Korean Journal of Pathology 2014; 48: 91-99 ORIGINAL ARTICLE Pleural Mesothelioma: An Institutional Experience of 66 Cases Soomin Ahn In Ho Choi Joungho Han Jhingook Kim 1 Myung-Ju Ahn 2 Departments
The Korean Journal of Pathology 2014; 48: 91-99 ORIGINAL ARTICLE Pleural Mesothelioma: An Institutional Experience of 66 Cases Soomin Ahn In Ho Choi Joungho Han Jhingook Kim 1 Myung-Ju Ahn 2 Departments
Report series: General cancer information
 Fighting cancer with information Report series: General cancer information Eastern Cancer Registration and Information Centre ECRIC report series: General cancer information Cancer is a general term for
Fighting cancer with information Report series: General cancer information Eastern Cancer Registration and Information Centre ECRIC report series: General cancer information Cancer is a general term for
Clinicopathological Study on Malignant Pleural Mesotheliomas
 Table of Contents Clinicopathological Study on Malignant Pleural Mesotheliomas PL-4-03 Kenzo Hiroshima Kenzo Hiroshima 1, Akira Iyota 1, Kiyoshi Sibuya 1, Toshikazu Yusa 2, Takehiko Fujisawa 1 and Yukio
Table of Contents Clinicopathological Study on Malignant Pleural Mesotheliomas PL-4-03 Kenzo Hiroshima Kenzo Hiroshima 1, Akira Iyota 1, Kiyoshi Sibuya 1, Toshikazu Yusa 2, Takehiko Fujisawa 1 and Yukio
Rotation Specific Goals & Objectives: University Health Network-Princess Margaret Hospital/ Sunnybrook Breast/Melanoma
 Rotation Specific Goals & Objectives: University Health Network-Princess Margaret Hospital/ Sunnybrook Breast/Melanoma Medical Expert: Breast Rotation Specific Competencies/Objectives 1.0 Medical History
Rotation Specific Goals & Objectives: University Health Network-Princess Margaret Hospital/ Sunnybrook Breast/Melanoma Medical Expert: Breast Rotation Specific Competencies/Objectives 1.0 Medical History
Understanding your pathology report
 Understanding your pathology report 2 Contents Contents Introduction 3 What is a pathology report? 3 Waiting for your results 4 What s in a pathology report? 4 Information about your breast cancer 5 What
Understanding your pathology report 2 Contents Contents Introduction 3 What is a pathology report? 3 Waiting for your results 4 What s in a pathology report? 4 Information about your breast cancer 5 What
A 70-year old Man with Pleural Effusion
 Mesothelioma Diagnosis: Pitfalls and Latest Updates S Klebe and DW Henderson Recommendations Indisputable malignant cells on cytomorphological criteria which demonstrate a mesothelial phenotype, which
Mesothelioma Diagnosis: Pitfalls and Latest Updates S Klebe and DW Henderson Recommendations Indisputable malignant cells on cytomorphological criteria which demonstrate a mesothelial phenotype, which
L Lang-Lazdunski, A Bille, S Marshall, R Lal, D Landau, J Spicer
 Pleurectomy/decortication, hyperthermic pleural lavage with povidone-iodine and systemic chemotherapy in malignant pleural mesothelioma. A 10-year experience. L Lang-Lazdunski, A Bille, S Marshall, R Lal,
Pleurectomy/decortication, hyperthermic pleural lavage with povidone-iodine and systemic chemotherapy in malignant pleural mesothelioma. A 10-year experience. L Lang-Lazdunski, A Bille, S Marshall, R Lal,
Changes in Breast Cancer Reports After Second Opinion. Dr. Vicente Marco Department of Pathology Hospital Quiron Barcelona. Spain
 Changes in Breast Cancer Reports After Second Opinion Dr. Vicente Marco Department of Pathology Hospital Quiron Barcelona. Spain Second Opinion in Breast Pathology Usually requested when a patient is referred
Changes in Breast Cancer Reports After Second Opinion Dr. Vicente Marco Department of Pathology Hospital Quiron Barcelona. Spain Second Opinion in Breast Pathology Usually requested when a patient is referred
Please see the LUCADA data manual v3.1.3, available in the downloads section
 National Lung Cancer Audit Frequently Asked Questions What dataset should be used? Please see the LUCADA data manual v3.1.3, available in the downloads section What does LUCADA stand for? LUCADA stands
National Lung Cancer Audit Frequently Asked Questions What dataset should be used? Please see the LUCADA data manual v3.1.3, available in the downloads section What does LUCADA stand for? LUCADA stands
Outline. Workup for metastatic breast cancer. Metastatic breast cancer
 Metastatic breast cancer Immunostain Update: Diagnosis of metastatic breast carcinoma, emphasizing distinction from GYN primary 1/3 of breast cancer patients will show metastasis 1 st presentation or 20-30
Metastatic breast cancer Immunostain Update: Diagnosis of metastatic breast carcinoma, emphasizing distinction from GYN primary 1/3 of breast cancer patients will show metastasis 1 st presentation or 20-30
A Practical Guide to Advances in Staging and Treatment of NSCLC
 A Practical Guide to Advances in Staging and Treatment of NSCLC Robert J. Korst, M.D. Director, Thoracic Surgery Medical Director, The Blumenthal Cancer Center The Valley Hospital Objectives Revised staging
A Practical Guide to Advances in Staging and Treatment of NSCLC Robert J. Korst, M.D. Director, Thoracic Surgery Medical Director, The Blumenthal Cancer Center The Valley Hospital Objectives Revised staging
Malignant Mesothelioma Diagnosed by Bronchoscopic Biopsy
 CASE REPORT http://dx.doi.org/10.4046/trd.2015.78.3.297 ISSN: 1738-3536(Print)/2005-6184(Online) Tuberc Respir Dis 2015;78:297-301 Malignant Mesothelioma Diagnosed by Bronchoscopic Biopsy Yeon-Hee Park,
CASE REPORT http://dx.doi.org/10.4046/trd.2015.78.3.297 ISSN: 1738-3536(Print)/2005-6184(Online) Tuberc Respir Dis 2015;78:297-301 Malignant Mesothelioma Diagnosed by Bronchoscopic Biopsy Yeon-Hee Park,
Clinical cases in Malignant Pleural Mesothelioma: Adherence to the ESMO Clinical Practice Guidelines
 Clinical cases in Malignant Pleural Mesothelioma: Adherence to the ESMO Clinical Practice Guidelines Wieneke Buikhuisen The Netherlands Cancer Institute Amsterdam The Netherlands Case (1) Male, 56 year
Clinical cases in Malignant Pleural Mesothelioma: Adherence to the ESMO Clinical Practice Guidelines Wieneke Buikhuisen The Netherlands Cancer Institute Amsterdam The Netherlands Case (1) Male, 56 year
Recommendations for cross-sectional imaging in cancer management, Second edition
 www.rcr.ac.uk Recommendations for cross-sectional imaging in cancer management, Second edition Breast cancer Faculty of Clinical Radiology www.rcr.ac.uk Contents Breast cancer 2 Clinical background 2 Who
www.rcr.ac.uk Recommendations for cross-sectional imaging in cancer management, Second edition Breast cancer Faculty of Clinical Radiology www.rcr.ac.uk Contents Breast cancer 2 Clinical background 2 Who
Mesothelioma: Questions and Answers
 CANCER FACTS N a t i o n a l C a n c e r I n s t i t u t e N a t i o n a l I n s t i t u t e s o f H e a l t h D e p a r t m e n t o f H e a l t h a n d H u m a n S e r v i c e s Mesothelioma: Questions
CANCER FACTS N a t i o n a l C a n c e r I n s t i t u t e N a t i o n a l I n s t i t u t e s o f H e a l t h D e p a r t m e n t o f H e a l t h a n d H u m a n S e r v i c e s Mesothelioma: Questions
Uses and Abuses of Pathology in Asbestos-exposed Populations
 Uses and Abuses of Pathology in Asbestos-exposed Populations Jerrold L. Abraham, MD Department of Pathology State University of New York Upstate Medical University Syracuse, NY, 13210 USA The term: Asbestosis,
Uses and Abuses of Pathology in Asbestos-exposed Populations Jerrold L. Abraham, MD Department of Pathology State University of New York Upstate Medical University Syracuse, NY, 13210 USA The term: Asbestosis,
WORKPLACE SAFETY AND INSURANCE APPEALS TRIBUNAL DECISION NO. 1557/14
 WORKPLACE SAFETY AND INSURANCE APPEALS TRIBUNAL DECISION NO. 1557/14 BEFORE: M. Crystal: Vice-Chair HEARING: August 20, 2014 at Toronto Written DATE OF DECISION: December 4, 2014 NEUTRAL CITATION: 2014
WORKPLACE SAFETY AND INSURANCE APPEALS TRIBUNAL DECISION NO. 1557/14 BEFORE: M. Crystal: Vice-Chair HEARING: August 20, 2014 at Toronto Written DATE OF DECISION: December 4, 2014 NEUTRAL CITATION: 2014
More than 2,500 people are diagnosed with mesothelioma in the UK each year.
 This information is an extract from the booklet Understanding mesothelioma. You may find the full booklet helpful. We can send you a free copy see page 5. Contents Introduction Pleural mesothelioma Peritoneal
This information is an extract from the booklet Understanding mesothelioma. You may find the full booklet helpful. We can send you a free copy see page 5. Contents Introduction Pleural mesothelioma Peritoneal
Digital Health: Catapulting Personalised Medicine Forward STRATIFIED MEDICINE
 Digital Health: Catapulting Personalised Medicine Forward STRATIFIED MEDICINE CRUK Stratified Medicine Initiative Somatic mutation testing for prediction of treatment response in patients with solid tumours:
Digital Health: Catapulting Personalised Medicine Forward STRATIFIED MEDICINE CRUK Stratified Medicine Initiative Somatic mutation testing for prediction of treatment response in patients with solid tumours:
International Journal of Case Reports in Medicine
 International Journal of Case Reports in Medicine Vol. 2013 (2013), Article ID 409830, 15 minipages. DOI:10.5171/2013.409830 www.ibimapublishing.com Copyright 2013 Andrew Thomas Low, Iain Smith and Simon
International Journal of Case Reports in Medicine Vol. 2013 (2013), Article ID 409830, 15 minipages. DOI:10.5171/2013.409830 www.ibimapublishing.com Copyright 2013 Andrew Thomas Low, Iain Smith and Simon
How To Prepare A Meeting For A Health Care Conference
 THORACIC ONCOLOGY MULTIDISCIPLINARY TEAM MEETINGS: OPERATIONAL POLICY EXECUTIVE SUMMARY 1. All patients in Lothian with thoracic malignancies should be discussed at designated times in pathway (see App
THORACIC ONCOLOGY MULTIDISCIPLINARY TEAM MEETINGS: OPERATIONAL POLICY EXECUTIVE SUMMARY 1. All patients in Lothian with thoracic malignancies should be discussed at designated times in pathway (see App
Clinical Commissioning Policy: Proton Beam Radiotherapy (High Energy) for Paediatric Cancer Treatment
 Clinical Commissioning Policy: Proton Beam Radiotherapy (High Energy) for Paediatric Cancer Treatment Reference: NHS England xxx/x/x 1 Clinical Commissioning Policy: Proton Beam Radiotherapy (High Energy)
Clinical Commissioning Policy: Proton Beam Radiotherapy (High Energy) for Paediatric Cancer Treatment Reference: NHS England xxx/x/x 1 Clinical Commissioning Policy: Proton Beam Radiotherapy (High Energy)
National Bowel Cancer Audit Report 2008 Public and Executive Summary
 National Bowel Cancer Audit Report 2008 Public and Executive Summary Prepared in association with: Healthcare Quality Improvement Partnership HQIP Association of Coloproctology of Great Britain and Ireland
National Bowel Cancer Audit Report 2008 Public and Executive Summary Prepared in association with: Healthcare Quality Improvement Partnership HQIP Association of Coloproctology of Great Britain and Ireland
MESOTHELIOMA. Not Just a Late Night Commercial. Graciela Hoal, RN, MSN, ACNP-BC
 MESOTHELIOMA Not Just a Late Night Commercial Graciela Hoal, RN, MSN, ACNP-BC Saturday Session Thoracic Surgery Nurse Practitioner Greater Los Angeles Veteran Affairs Objectives Course Objectives: Discuss
MESOTHELIOMA Not Just a Late Night Commercial Graciela Hoal, RN, MSN, ACNP-BC Saturday Session Thoracic Surgery Nurse Practitioner Greater Los Angeles Veteran Affairs Objectives Course Objectives: Discuss
Lung Cancer Treatment Guidelines
 Updated June 2014 Derived and updated by consensus of members of the Providence Thoracic Oncology Program with the aid of evidence-based National Comprehensive Cancer Network (NCCN) national guidelines,
Updated June 2014 Derived and updated by consensus of members of the Providence Thoracic Oncology Program with the aid of evidence-based National Comprehensive Cancer Network (NCCN) national guidelines,
III II II 111111111111111111111111111111111111111111 1 1111111111111 III. ge: /ears sex: Male Access
 UUID : 10F97F63 - EE6F-448D - 98E1-8AAA21F55753 TCGA - TS-A7PB-01A-PR Redacted III II II 111111111111111111111111111111111111111111 1 1111111111111 III Date Receive III 11111111111111111 11111111111 II
UUID : 10F97F63 - EE6F-448D - 98E1-8AAA21F55753 TCGA - TS-A7PB-01A-PR Redacted III II II 111111111111111111111111111111111111111111 1 1111111111111 III Date Receive III 11111111111111111 11111111111 II
How To Treat Mesothelioma
 Mesothelioma in the UK Liz Darlison Mesothelioma UK The National Macmillan Mesothelioma Resource Centre June 2010 Mesothelioma Applied Research Foundation - 2010 www.curemeso.org Contents Size of Mesothelioma
Mesothelioma in the UK Liz Darlison Mesothelioma UK The National Macmillan Mesothelioma Resource Centre June 2010 Mesothelioma Applied Research Foundation - 2010 www.curemeso.org Contents Size of Mesothelioma
IASLC Mesothelioma Staging Project - Reduced Set of Data Elements for Proposals for Change to 8th Edition TNM 7/7/2013
 Page 1 of 16 Patient Characteristics form (3 variables) Disease Description form (6 variables) Pre-treatment/Evaluative TNM form - Pre-Treatment/Evaluative Thickness and Effusions form - Pre-Treatment/Evaluative
Page 1 of 16 Patient Characteristics form (3 variables) Disease Description form (6 variables) Pre-treatment/Evaluative TNM form - Pre-Treatment/Evaluative Thickness and Effusions form - Pre-Treatment/Evaluative
Cytology : first alert of mesothelioma? Professor B. Weynand, UCL Yvoir, Belgium
 Cytology : first alert of mesothelioma? Professor B. Weynand, UCL Yvoir, Belgium Introduction 3 cavities with the same embryologic origin the mesoderme Pleura Exudates Pleura Peritoneum Pericardium 22%
Cytology : first alert of mesothelioma? Professor B. Weynand, UCL Yvoir, Belgium Introduction 3 cavities with the same embryologic origin the mesoderme Pleura Exudates Pleura Peritoneum Pericardium 22%
The evolving pathology of solitary fibrous tumours. Luciane Dreher Irion MREH / CMFT / NSOPS
 The evolving pathology of solitary fibrous tumours Luciane Dreher Irion MREH / CMFT / NSOPS Historical review Haemangiopericytoma (HPC) first described primarily as a soft tissue vascular tumour of pericytic
The evolving pathology of solitary fibrous tumours Luciane Dreher Irion MREH / CMFT / NSOPS Historical review Haemangiopericytoma (HPC) first described primarily as a soft tissue vascular tumour of pericytic
9. Discuss guidelines for follow-up post-thyroidectomy for cancer (labs/tests) HH
 9. Discuss guidelines for follow-up post-thyroidectomy for cancer (labs/tests) HH Differentiated thyroid cancer expresses the TSH receptor on the cell membrane and responds to TSH stimulation by increasing
9. Discuss guidelines for follow-up post-thyroidectomy for cancer (labs/tests) HH Differentiated thyroid cancer expresses the TSH receptor on the cell membrane and responds to TSH stimulation by increasing
Malignant pleural mesothelioma: outcome of limited surgical management
 Interactive Cardiovascular and Thoracic Surgery 2 (2003) 30 34 Institutional review Thoracic general Malignant pleural mesothelioma: outcome of limited surgical management Peter G. Phillips a, George Asimakopoulos
Interactive Cardiovascular and Thoracic Surgery 2 (2003) 30 34 Institutional review Thoracic general Malignant pleural mesothelioma: outcome of limited surgical management Peter G. Phillips a, George Asimakopoulos
Immunohistochemical differentiation of metastatic tumours
 Immunohistochemical differentiation of metastatic tumours Dr Abi Wheal ST1. TERA 3/2/14 Key points from a review article written by Daisuke Nonaka Intro Metastatic disease is the initial presentation in
Immunohistochemical differentiation of metastatic tumours Dr Abi Wheal ST1. TERA 3/2/14 Key points from a review article written by Daisuke Nonaka Intro Metastatic disease is the initial presentation in
Understanding Pleural Mesothelioma
 Understanding Pleural Mesothelioma UHN Information for patients and families Read this booklet to learn about: What is pleural mesothelioma? What causes it? What are the symptoms? What tests are done to
Understanding Pleural Mesothelioma UHN Information for patients and families Read this booklet to learn about: What is pleural mesothelioma? What causes it? What are the symptoms? What tests are done to
Today s Topics. Tumors of the Peritoneum in Women
 Today s Topics Tumors of the Peritoneum in Women Charles Zaloudek, M.D. Department of Pathology 505 Parnassus Ave., M563 University of California, San Francisco San Francisco, CA USA charles.zaloudek@ucsf.edu
Today s Topics Tumors of the Peritoneum in Women Charles Zaloudek, M.D. Department of Pathology 505 Parnassus Ave., M563 University of California, San Francisco San Francisco, CA USA charles.zaloudek@ucsf.edu
20 Diagnostic Cytopathology, Vol 36, No 1 ' 2007 WILEY-LISS, INC.
 Utility of WT-1, p63, MOC31, Mesothelin, and Cytokeratin (K903 and CK5/6) Immunostains in Differentiating Adenocarcinoma, Squamous Cell Carcinoma, and Malignant Mesothelioma in Effusions Robert T. Pu,
Utility of WT-1, p63, MOC31, Mesothelin, and Cytokeratin (K903 and CK5/6) Immunostains in Differentiating Adenocarcinoma, Squamous Cell Carcinoma, and Malignant Mesothelioma in Effusions Robert T. Pu,
Original Article Differential diagnosis of sarcomatoid mesothelioma from true sarcoma and sarcomatoid carcinoma using immunohistochemistry
 Pathology International 2008; 58: 75 83 doi:10.1111/j.1440-1827.2007.02193.x Original Article Differential diagnosis of sarcomatoid mesothelioma from true sarcoma and sarcomatoid carcinoma using immunohistochemistry
Pathology International 2008; 58: 75 83 doi:10.1111/j.1440-1827.2007.02193.x Original Article Differential diagnosis of sarcomatoid mesothelioma from true sarcoma and sarcomatoid carcinoma using immunohistochemistry
Rare Thoracic Tumours
 Rare Thoracic Tumours 1. Epithelial Tumour of Trachea 1 1.1 General Results Table 1. Epithelial Tumours of Trachea: Incidence, Trends, Survival Flemish Region 2001-2010 Both Sexes Incidence Trend EAPC
Rare Thoracic Tumours 1. Epithelial Tumour of Trachea 1 1.1 General Results Table 1. Epithelial Tumours of Trachea: Incidence, Trends, Survival Flemish Region 2001-2010 Both Sexes Incidence Trend EAPC
Understanding Your Surgical Options for Lung Cancer
 Information Booklet for Patients Understanding Your Surgical Options for Lung Cancer Understanding Lung Cancer If you have just been diagnosed with lung cancer, this booklet will serve as an informational
Information Booklet for Patients Understanding Your Surgical Options for Lung Cancer Understanding Lung Cancer If you have just been diagnosed with lung cancer, this booklet will serve as an informational
Malignant Pleural Mesothelioma in Singapore
 RESEARCH COMMUNICATION C SP Yip 1, HN Koong 2, CM Loo 3, KW Fong 1* Abstract Aim: To examine the clinical characteristics and outcomes of malignant pleural mesothelioma (MPM) in Singapore. Methods and
RESEARCH COMMUNICATION C SP Yip 1, HN Koong 2, CM Loo 3, KW Fong 1* Abstract Aim: To examine the clinical characteristics and outcomes of malignant pleural mesothelioma (MPM) in Singapore. Methods and
PROTOCOL OF THE RITA DATA QUALITY STUDY
 PROTOCOL OF THE RITA DATA QUALITY STUDY INTRODUCTION The RITA project is aimed at estimating the burden of rare malignant tumours in Italy using the population based cancer registries (CRs) data. One of
PROTOCOL OF THE RITA DATA QUALITY STUDY INTRODUCTION The RITA project is aimed at estimating the burden of rare malignant tumours in Italy using the population based cancer registries (CRs) data. One of
Disclosures. Learning Objectives. Effusion = Confusion. Diagnosis Of Serous Cavity Effusions - Beware The Mesothelial Cell!
 Disclosures Diagnosis Of Serous Cavity Effusions - Beware The Mesothelial Cell! No Relevant Financial Relationships with Commercial Interests Syed Z. Ali, M.D. Syed Z. Ali, M.D. Associate Professor of
Disclosures Diagnosis Of Serous Cavity Effusions - Beware The Mesothelial Cell! No Relevant Financial Relationships with Commercial Interests Syed Z. Ali, M.D. Syed Z. Ali, M.D. Associate Professor of
Mesothelioma. Malignant Pleural Mesothelioma
 Mesothelioma William G. Richards, PhD Brigham and Women s Hospital Malignant Pleural Mesothelioma 2,000-3,000 cases per year (USA) Increasing incidence Asbestos (50-80%, decreasing) 30-40 year latency
Mesothelioma William G. Richards, PhD Brigham and Women s Hospital Malignant Pleural Mesothelioma 2,000-3,000 cases per year (USA) Increasing incidence Asbestos (50-80%, decreasing) 30-40 year latency
TUMORS OF THE TESTICULAR ADNEXA and SPERMATIC CORD
 TUMORS OF THE TESTICULAR ADNEXA and SPERMATIC CORD Victor E. Reuter, MD Memorial Sloan-Kettering Cancer Center reuterv@mskcc.org 66 th Annual Pathology Seminar California Society of Pathologists Short
TUMORS OF THE TESTICULAR ADNEXA and SPERMATIC CORD Victor E. Reuter, MD Memorial Sloan-Kettering Cancer Center reuterv@mskcc.org 66 th Annual Pathology Seminar California Society of Pathologists Short
RENAL CANCER PATHOLOGY WHAT REALLY MATTERS? STEWART FLEMING UNIVERSITY OF DUNDEE
 RENAL CANCER PATHOLOGY WHAT REALLY MATTERS? STEWART FLEMING UNIVERSITY OF DUNDEE MAJOR PARADIGM SHIFT IN EARLY 1990S IN UNDERSTANDING RENAL CANCER Molecular differential pathology of renal cell tumours
RENAL CANCER PATHOLOGY WHAT REALLY MATTERS? STEWART FLEMING UNIVERSITY OF DUNDEE MAJOR PARADIGM SHIFT IN EARLY 1990S IN UNDERSTANDING RENAL CANCER Molecular differential pathology of renal cell tumours
The New International Staging System Lung Cancer
 The New International Staging System Lung Cancer Valerie W. Rusch, MD Chief, Thoracic Surgery Memorial Sloan-Kettering Cancer Center Chair, Lung and Esophagus Task Force, American Joint Commission on Cancer
The New International Staging System Lung Cancer Valerie W. Rusch, MD Chief, Thoracic Surgery Memorial Sloan-Kettering Cancer Center Chair, Lung and Esophagus Task Force, American Joint Commission on Cancer
Diseases. Inflammations Non-inflammatory pleural effusions Pneumothorax Tumours
 Pleura Visceral pleura covers lungs and extends into fissures Parietal pleura limits mediastinum and covers dome of diaphragm and inner aspect of chest wall. Two layers between them (pleural cavity) contains
Pleura Visceral pleura covers lungs and extends into fissures Parietal pleura limits mediastinum and covers dome of diaphragm and inner aspect of chest wall. Two layers between them (pleural cavity) contains
Lung Cancer & Mesothelioma 2011-2015
 Lung Cancer & Mesothelioma 2011-2015 Annex G Mesothelioma 1. The vision for mesothelioma services is set out in the Mesothelioma Framework issued by DH on 26 February 2007 (supported by the British Thoracic
Lung Cancer & Mesothelioma 2011-2015 Annex G Mesothelioma 1. The vision for mesothelioma services is set out in the Mesothelioma Framework issued by DH on 26 February 2007 (supported by the British Thoracic
Diagnostic Challenge. Department of Pathology,
 Cytology of Pleural Fluid as a Diagnostic Challenge Paavo Pääkkö,, MD, PhD Chief Physician and Head of the Department Department of Pathology, Oulu University Hospital,, Finland Oulu University Hospital
Cytology of Pleural Fluid as a Diagnostic Challenge Paavo Pääkkö,, MD, PhD Chief Physician and Head of the Department Department of Pathology, Oulu University Hospital,, Finland Oulu University Hospital
Renal Cell Carcinoma: Advances in Diagnosis B. Iványi, MD
 Renal Cell Carcinoma: Advances in Diagnosis B. Iványi, MD Department of Pathology University of Szeged, Hungary ISUP Vancouver Classification of Renal Neoplasia Am J Surg Pathol 37:14691489, 2013 13 histologic
Renal Cell Carcinoma: Advances in Diagnosis B. Iványi, MD Department of Pathology University of Szeged, Hungary ISUP Vancouver Classification of Renal Neoplasia Am J Surg Pathol 37:14691489, 2013 13 histologic
Malignant Mesothelioma
 Malignant Mesothelioma What is malignant mesothelioma? Malignant mesothelioma is a cancer that starts in cells in the linings of certain parts of the body, especially in the linings of the chest or abdomen.
Malignant Mesothelioma What is malignant mesothelioma? Malignant mesothelioma is a cancer that starts in cells in the linings of certain parts of the body, especially in the linings of the chest or abdomen.
