55 What are the rules for promoting medications to consumers in the U.S.?
|
|
|
- Deirdre Price
- 9 years ago
- Views:
Transcription
1 Chapter 55 What are the rules for promoting medications to consumers in the U.S.? The U.S. and New Zealand are the only two countries that allow the directto-consumer (DTC) advertising of prescription medications. The promotion of over-the-counter (OTC) products and dietary supplements to consumers is allowed in this country and in other countries. The rules and regulations in the U.S. for DTC advertising are different for prescription medicines, OTC drugs, and dietary supplements. The rules are also somewhat different for print and broadcast promotions. What are the current regulations? Two federal agencies are responsible for overseeing this promotion, and these agencies use different standards to distinguish truthful advertising from false or misleading advertising. These agencies are the FDA and the Federal Trade Commission (FTC). The regulation of advertising, including drug advertising, must not violate the First Amendment right of free speech guaranteed by the U.S. Constitution. The concept of First Amendment protection for religious and political speech is well recognized. The Supreme Court has ruled that truthful commercial speech such as advertising, as long as it is not false or misleading, also is entitled to protection under the Constitution. Public safety is the primary reason for regulatory restrictions on medication promotion. Drug advertising that overemphasizes product benefits and downplays the medication s potential harms may present a significant public health concern. The regulations for prescription drug advertising require ads to contain fair balance information, whether for DTC promotions or ads targeting the medical professions. In the past, this meant that print ads would have to appear with the text of a drug s professional product labeling, or a brief summary of the professional label. This essentially prohibited television or radio advertising of medications to consumers because all the label information would have to be read on the radio or appear on the TV screen. In the late 1990s, the FDA opened the door for DTC television and radio broadcast advertising by allowing the use of a major statement of 184
2 185
3 medication risks instead of the professional product label. The advertisement, either print or broadcast, has to direct the consumer to a medication s professional product label printed in a magazine or manufacturer s Web site. From the consumers perspective, the problem with the current interpretation of the regulations is that professional product labels are not written for them. This information is written in dense technical jargon in small type, and it assumes the reader is a health professional. The FDA has never written regulations directly addressing DTC prescription medication advertising. Instead, the agency has applied the regulations that govern directto-professional advertising to DTC advertising. The regulatory oversight of OTC medication promotion is the responsibility of the FTC. The FTC standards for OTC drug promotion require advertising to be truthful and non-deceptive, require the advertiser to have evidence to back up their claims, and require that the ads cannot be unfair. The evidence standard requires competent and reliable scientific evidence. The FDA s standard for substantial evidence is technically and legally much stricter. What is the FDA s role? The FDA is responsible for overseeing all prescription medication advertising in the U.S. This regulatory responsibility resides with the FDA s Division of Drug Marketing, Advertising, and Communications (DDMAC). This small organization operates with limited resources but has the enormous task of ensuring truthful, balanced and accurate communication of prescription medication information and advertising. DDMAC regulates all print and broadcast prescription medication advertising targeting both consumers and health professionals. When DDMAC finds that a company is distributing false or misleading advertising in violation of laws and regulations, a Notice of Violation or Warning Letter may be issued to that company. These notices can be found on the FDA s Web site, at www. fda.gov/iceci/enforcementactions/warningletters/default.htm. In cases of repeated advertising violations, DDMAC can require companies to distribute correctional advertisements, though this option is rarely used. Newly enacted legislation will give DDMAC the authority to review DTC ads prior to their dissemination to ensure accuracy and fair balance. What is the argument for and against DTC advertising? Before discussing the pros and cons of DTC advertising, it must be noted that DTC advertising should not be analyzed in isolation. DTC and direct-toprofessional promotions are two parts of a carefully planned marketing strategy for prescription medications. 186
4 One of the main arguments for DTC advertising is that it is educational for consumers. Proponents of DTC prescription medication advertising, primarily pharmaceutical manufacturers, maintain that medication advertising gives consumers the information they need to make informed decisions about their treatment options. Another argument put forth by those in favor of DTC advertising is that it drives consumers to consult their physicians with questions about the advertised medications. It is argued that previously undetected health problems may be found at these appointments. Opponents of DTC advertising argue that an important educational message about the safety and effectiveness of prescription medications can not be communicated in a 30- or 60-second television or radio advertisement. The dictionary defines the purpose of advertising as calling attention to a product by emphasizing its desirable qualities and to arouse a desire to buy the product. In the view of opponents, the sole purpose of DTC advertising is to sell. Advertising is one-sided and does not allow for fully informed decisions about medications. This risks needless adverse drug reactions and substantial economic harm to individuals and the health-care system because costly medications may be prescribed when less expensive, safer, and more effective alternatives are available. What is the experience? DCT advertising has been a remarkable economic success for the pharmaceutical industry. The top four most frequently prescribed prescription drugs in the U.S. in 2007 also were among the top drugs in terms of spending on DTC advertising: atorvastatin (Lipitor), montelukast (Singulair), escitalopram (Lexapro) and esomeprazole (Nexium). They accounted for sales approaching $16 billion in The power of advertising is evident from noting that atorvastatin is a cholesterol-lowering statin drug. There is now a less-expensive generic version of simvastatin (Zocor) available to lower cholesterol. The biggest market for Singulair is to treat hayfever, but it may be less effective for this use than OTC loratadine (Claritin). Escitalopram, an antidepressant, and esomeprazole, used for stomach acid reflux, have the same chemical structures as older drugs citalopram (Celexa), and OTC omeprazole (Prilosec), respectively. A 2002 survey by the American Association for Retired People of 1,250 persons age 45 or older reported that 91 percent had seen a commercial ad for a medication on TV. Of them, 19 percent subsequently asked for the advertised medication, and 11 percent had received a prescription. DTC advertising has grown from slightly less than $1 billion in 1996 to over $4 billion spent in DTC advertising is a very good investment 187
5 for manufacturers. Every dollar spent on DTC advertising results in a sales increase of as much as $6, according to a Congressional estimate. Many of the most heavily advertised medications may not be that important therapeutically. COX-2 drugs including rofecoxib (Vioxx), valdecoxib (Bextra), and celecoxib (Celebrex) were heavily promoted as safer drugs for arthritis and pain with a lower risk of GI tract toxicity, ulcers and hemorrhage than older drugs known as non-steroidal anti-inflammatory drugs (NSAIDs). Rofecoxib and valdecoxib were removed from the market because of unacceptable heart risks. A national survey of COX-2 prescribing found 63 percent of the growth in the use of these drugs was in patients who could have used older, less expensive NSAIDs. Key messages 3 Promotion of prescription medications is regulated. 3 DTC advertising generates strong patient demand, which physicians have difficulty controlling. 3 DTC advertising appears to increase the sale of newer, more costly medications when less expensive, equally effective older drugs are available. 3 The U.S. is one of just two countries in the world which allow DTC advertising of prescription medications. 3 Promotion of OTC medications is less restricted than promotion of prescription medications. 188
Potential Savings from Generic Drugs in Upstate New York
 T H E F A C T S A B O U T Potential Savings from Generic Drugs in Upstate New York $880 Million in Potential Savings for Upstate New York Counties Finger Lakes Region $141 million Western New York Region
T H E F A C T S A B O U T Potential Savings from Generic Drugs in Upstate New York $880 Million in Potential Savings for Upstate New York Counties Finger Lakes Region $141 million Western New York Region
Introduction to Compliance with FDA Labeling and Advertising Requirements
 Introduction to Compliance with FDA Labeling and Advertising Requirements Second Annual Pharmaceutical Industry Regulatory and Compliance Summit Dick Kenny FDA History Basic function of government Oldest
Introduction to Compliance with FDA Labeling and Advertising Requirements Second Annual Pharmaceutical Industry Regulatory and Compliance Summit Dick Kenny FDA History Basic function of government Oldest
Interplay Between FDA Advertising and Promotion Enforcement Activities, Product Liability, and Consumer Fraud Litigation
 Interplay Between FDA Advertising and Promotion Enforcement Activities, Product Liability, and Consumer Fraud Litigation Leslie M. Tector Quarles & Brady LLP September 30, 2014 Objectives Which federal
Interplay Between FDA Advertising and Promotion Enforcement Activities, Product Liability, and Consumer Fraud Litigation Leslie M. Tector Quarles & Brady LLP September 30, 2014 Objectives Which federal
DIRECT-TO-CONSUMER PRESCRIPTION DRUG ADVERTISING
 DIRECT-TO-CONSUMER PRESCRIPTION DRUG ADVERTISING American College of Physicians A Position Paper 2006 DIRECT-TO-CONSUMER PRESCRIPTION DRUG ADVERTISING A Position Paper of the American College of Physicians
DIRECT-TO-CONSUMER PRESCRIPTION DRUG ADVERTISING American College of Physicians A Position Paper 2006 DIRECT-TO-CONSUMER PRESCRIPTION DRUG ADVERTISING A Position Paper of the American College of Physicians
Scott Byrne & Rachel Weeks G400: Pharma Report 10/22/09 Direct to Consumer Advertising: The Affect on the Pharmaceutical Industry
 Scott Byrne & Rachel Weeks G400: Pharma Report 10/22/09 Direct to Consumer Advertising: The Affect on the Pharmaceutical Industry A Glance at the Pharmaceutical Industry The pharmaceutical industry is
Scott Byrne & Rachel Weeks G400: Pharma Report 10/22/09 Direct to Consumer Advertising: The Affect on the Pharmaceutical Industry A Glance at the Pharmaceutical Industry The pharmaceutical industry is
Script/Notes for PowerPoint Presentation. Medication Use Safety Training for Seniors (MUST for Seniors)
 Script/Notes for PowerPoint Presentation Medication Use Safety Training for Seniors (MUST for Seniors) Instructions: You can use the following script to help you prepare your remarks to your organization
Script/Notes for PowerPoint Presentation Medication Use Safety Training for Seniors (MUST for Seniors) Instructions: You can use the following script to help you prepare your remarks to your organization
Copyright. Jeremiah J. Kelly (2015). All rights reserved. Further dissemination without express written consent strictly prohibited.
 Copyright. Jeremiah J. Kelly (2015). All rights reserved. Further dissemination without express written consent strictly prohibited. Sources of Regulation 502(n) added to the FD&C Act in the Kefauver-Harris
Copyright. Jeremiah J. Kelly (2015). All rights reserved. Further dissemination without express written consent strictly prohibited. Sources of Regulation 502(n) added to the FD&C Act in the Kefauver-Harris
Over the Counter Drugs (OTCs): Considerations for Physical Therapy Practice in Canada
 Background Over the Counter Drugs (OTCs): Considerations for Physical Therapy Practice in Canada The use of medications or drugs by non-physician health professionals is evolving and is linked to collaboration
Background Over the Counter Drugs (OTCs): Considerations for Physical Therapy Practice in Canada The use of medications or drugs by non-physician health professionals is evolving and is linked to collaboration
The Swedish Pharmaceutical Reimbursement System
 The Swedish Pharmaceutical Reimbursement System January 2007 www.lfn.se The Swedish Pharmaceutical Reimbursement System - A brief overview Sweden made some major changes to its reimbursement system in
The Swedish Pharmaceutical Reimbursement System January 2007 www.lfn.se The Swedish Pharmaceutical Reimbursement System - A brief overview Sweden made some major changes to its reimbursement system in
Choosing Pain Medicine for Osteoarthritis. A Guide for Consumers
 Choosing Pain Medicine for Osteoarthritis A Guide for Consumers Fast Facts on Pain Relievers Acetaminophen (Tylenol ) works on mild pain and has fewer risks than other pain pills. Prescription (Rx) pain
Choosing Pain Medicine for Osteoarthritis A Guide for Consumers Fast Facts on Pain Relievers Acetaminophen (Tylenol ) works on mild pain and has fewer risks than other pain pills. Prescription (Rx) pain
Guidance for Industry
 Guidance for Industry Consumer-Directed Broadcast Advertisements Questions and Answers U.S. Department of Health and Human Services Food and Drug Administration Center for Drug Evaluation and Research
Guidance for Industry Consumer-Directed Broadcast Advertisements Questions and Answers U.S. Department of Health and Human Services Food and Drug Administration Center for Drug Evaluation and Research
DUVAL CLIENT ALERT December 2009
 1 P a g e DUVAL CLIENT ALERT December 2009 In this DUVAL CLIENT ALERT, we list practical ways medical device companies can lawfully communicate about their products prior to FDA clearance or approval.
1 P a g e DUVAL CLIENT ALERT December 2009 In this DUVAL CLIENT ALERT, we list practical ways medical device companies can lawfully communicate about their products prior to FDA clearance or approval.
Amgen GLOBAL CORPORATE COMPLIANCE POLICY
 1. Scope Applicable to all Amgen Inc. and subsidiary or affiliated company staff members, consultants, contract workers and temporary staff worldwide ( Covered Persons ). Consultants, contract workers,
1. Scope Applicable to all Amgen Inc. and subsidiary or affiliated company staff members, consultants, contract workers and temporary staff worldwide ( Covered Persons ). Consultants, contract workers,
2. ABOUT THE HUMAN GENETICS COMMISSION (HGC) 4. WHAT ARE THE CURRENT LAWS/LEGISLATION AROUND GENETIC TESTING?
 Genes direct: Ensuring the effective oversight of genetic tests supplied directly to the public (A report by the Human Genetics Commission, March 2003) SUMMARY: Below is a five-page summary of the UK Human
Genes direct: Ensuring the effective oversight of genetic tests supplied directly to the public (A report by the Human Genetics Commission, March 2003) SUMMARY: Below is a five-page summary of the UK Human
Cosmetics Regulation in the United States and the European Union:
 Cosmetics Regulation in the United States and the European Union: Different Pathways to the Same Result By Richard Kingham and Lucas E. Beirne Introduction It is sometimes suggested that cosmetic products
Cosmetics Regulation in the United States and the European Union: Different Pathways to the Same Result By Richard Kingham and Lucas E. Beirne Introduction It is sometimes suggested that cosmetic products
US Health Statistics: Americans Most Over-Prescribed Country in the World
 US Health Statistics: Americans Most Over-Prescribed Country in the World A vitally important story reported in the April 15, 1998, issue of the Journal of the American Medical Association (JAMA), sums
US Health Statistics: Americans Most Over-Prescribed Country in the World A vitally important story reported in the April 15, 1998, issue of the Journal of the American Medical Association (JAMA), sums
Medication Guide Plavix (PLAV-iks) (clopidogrel bisulfate) tablets
 Medication Guide Plavix (PLAV-iks) (clopidogrel bisulfate) tablets Read this Medication Guide before you start taking Plavix and each time you get a refill. There may be new information. This Medication
Medication Guide Plavix (PLAV-iks) (clopidogrel bisulfate) tablets Read this Medication Guide before you start taking Plavix and each time you get a refill. There may be new information. This Medication
What is Rheumatoid Arthritis?
 Name: A Case Study in Pharmaceutical Research Date: Class Work: Part I Aim: How do Biologists Select their Projects? What is Rheumatoid Arthritis? Rheumatoid arthritis (rue-ma-toyd arth-write-tis) is a
Name: A Case Study in Pharmaceutical Research Date: Class Work: Part I Aim: How do Biologists Select their Projects? What is Rheumatoid Arthritis? Rheumatoid arthritis (rue-ma-toyd arth-write-tis) is a
Principles of Good Practice for Advertising and Promotion of Animal Health Products. A Voluntary Guideline Developed by the Animal Health Institute
 Principles of Good Practice for Advertising and Promotion of Animal Health Products A Voluntary Guideline Developed by the Animal Health Institute The Animal Health Institute is the U.S. trade association
Principles of Good Practice for Advertising and Promotion of Animal Health Products A Voluntary Guideline Developed by the Animal Health Institute The Animal Health Institute is the U.S. trade association
DOUBLE PATENTING CONSIDERATIONS by Mark Cohen
 DOUBLE PATENTING CONSIDERATIONS by Mark Cohen The Federal Circuit recently issued an important decision with respect to restriction practice and obviousness double patenting in Pfizer Inc. v. Teva Pharmaceuticals
DOUBLE PATENTING CONSIDERATIONS by Mark Cohen The Federal Circuit recently issued an important decision with respect to restriction practice and obviousness double patenting in Pfizer Inc. v. Teva Pharmaceuticals
Guidance for Industry Direct-to-Consumer Television Advertisements FDAAA DTC Television Ad Pre- Dissemination Review Program
 Guidance for Industry Direct-to-Consumer Television Advertisements FDAAA DTC Television Ad Pre- Dissemination Review Program DRAFT GUIDANCE This guidance document is being distributed for comment purposes
Guidance for Industry Direct-to-Consumer Television Advertisements FDAAA DTC Television Ad Pre- Dissemination Review Program DRAFT GUIDANCE This guidance document is being distributed for comment purposes
Healthcare Law Compliance Guide (commonly known as the White Guide)
 Pfizer Inc 235 East 42nd Street New York, NY 10017 www.pfizer.com Dear Colleagues: At Pfizer, we are committed to upholding the highest standards when we interact with physicians, healthcare organizations,
Pfizer Inc 235 East 42nd Street New York, NY 10017 www.pfizer.com Dear Colleagues: At Pfizer, we are committed to upholding the highest standards when we interact with physicians, healthcare organizations,
Articles Presented. Journal Presentation. Dr Albert Lo. Dr Albert Lo
 * This presentation is prepared by the author in one s personal capacity for the purpose of academic exchange and does not represent the views of his/her organisations on the topic discussed. Journal Presentation
* This presentation is prepared by the author in one s personal capacity for the purpose of academic exchange and does not represent the views of his/her organisations on the topic discussed. Journal Presentation
Health Council of Canada
 What are the Public Health Implications? Direct-to-Consumer Advertising of Prescription Drugs in Canada Barbara Mintzes, PhD Centre for Health Services and Policy Research University of British Columbia
What are the Public Health Implications? Direct-to-Consumer Advertising of Prescription Drugs in Canada Barbara Mintzes, PhD Centre for Health Services and Policy Research University of British Columbia
Health Products and Food Branch. www.hc-sc.gc.ca
 Health Products and Food Branch 1 HEALTH CANADA PART C DIVISION 5 - DRUGS FOR CLINICAL TRIALS INVOLVING HUMAN SUBJECTS Part C - Division 5 2 Part A: Part B: Part C: Part D: Part E: Part G: Part J: Administration
Health Products and Food Branch 1 HEALTH CANADA PART C DIVISION 5 - DRUGS FOR CLINICAL TRIALS INVOLVING HUMAN SUBJECTS Part C - Division 5 2 Part A: Part B: Part C: Part D: Part E: Part G: Part J: Administration
Direct-to-Consumer Prescription Drug Advertising The European Commission s Proposals for Legislative Change
 Direct-to-Consumer Prescription Drug Advertising The European Commission s Proposals for Legislative Change Health Action International (HAI-Europe), December 2001 The European Union currently forbids
Direct-to-Consumer Prescription Drug Advertising The European Commission s Proposals for Legislative Change Health Action International (HAI-Europe), December 2001 The European Union currently forbids
The Unintended Effects of
 The Unintended Effects of Healthcare Reform TOM SUROVY, PRINCIPLE COMPLIANCE ATTORNEY CONTENTS CHILD-ONLY POLICIES... 3 PRESCRIPTIONS FOR NONPRESCRIPTION OVER-THE-COUNTER DRUGS... 4 COMMISSIONS FOR INSURANCE
The Unintended Effects of Healthcare Reform TOM SUROVY, PRINCIPLE COMPLIANCE ATTORNEY CONTENTS CHILD-ONLY POLICIES... 3 PRESCRIPTIONS FOR NONPRESCRIPTION OVER-THE-COUNTER DRUGS... 4 COMMISSIONS FOR INSURANCE
Peptic Ulcer. Anatomy The stomach is a hollow organ. It is located in the upper abdomen, under the ribs.
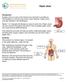 Peptic Ulcer Introduction A peptic ulcer is a sore in the lining of your stomach or duodenum. The duodenum is the first part of your small intestine. Peptic ulcers may also develop in the esophagus. Nearly
Peptic Ulcer Introduction A peptic ulcer is a sore in the lining of your stomach or duodenum. The duodenum is the first part of your small intestine. Peptic ulcers may also develop in the esophagus. Nearly
2015 Travelers Prescription Drug Plan Blue Cross Blue Shield Plan and United Healthcare Choice Plus Plan
 2015 Travelers Prescription Drug Plan Blue Cross Blue Shield Plan and United Healthcare Choice Plus Plan Plan Details, Programs, and Policies Table of Contents Click on the links below to be taken to that
2015 Travelers Prescription Drug Plan Blue Cross Blue Shield Plan and United Healthcare Choice Plus Plan Plan Details, Programs, and Policies Table of Contents Click on the links below to be taken to that
Ask Your Doctor if There May Be a SMARTER CHOICE
 If you have osteoarthritis, rheumatoid arthritis or ankylosing spondylitis, Could Your NSAID Pain Medicine Be Hurting Your Stomach? Ask Your Doctor if There May Be a SMARTER CHOICE 1 of 8 Making Smart
If you have osteoarthritis, rheumatoid arthritis or ankylosing spondylitis, Could Your NSAID Pain Medicine Be Hurting Your Stomach? Ask Your Doctor if There May Be a SMARTER CHOICE 1 of 8 Making Smart
PHARMACY BENEFIT DESIGN CONSIDERATIONS
 PHARMACY BENEFIT DESIGN CONSIDERATIONS Is your pharmacy benefit designed for your employees or the big drug companies? The pharmacy (or prescription) benefit is one of the most sought after benefits by
PHARMACY BENEFIT DESIGN CONSIDERATIONS Is your pharmacy benefit designed for your employees or the big drug companies? The pharmacy (or prescription) benefit is one of the most sought after benefits by
Goals & Objectives. Drug Development & the FDA Pharmacy 309. Outline. An History of Disasters. Be able to describe
 Drug Development & the FDA Pharmacy 309 Tom Hazlet, Pharm.D., Dr.P.H. 616-2732 thazlet@u... Goals & Objectives Be able to describe the major regulatory events in the drug development process the concepts
Drug Development & the FDA Pharmacy 309 Tom Hazlet, Pharm.D., Dr.P.H. 616-2732 thazlet@u... Goals & Objectives Be able to describe the major regulatory events in the drug development process the concepts
APMI CODE OF PRACTICE ON ADVERTISING OF MEDICINAL PRODUCTS ISSUE 1
 APMI CODE OF PRACTICE ON ADVERTISING OF MEDICINAL PRODUCTS ISSUE 1 August 2010 1. Introduction... 2 2. Scope... 3 3. Definitions... 4 4. General rules of advertising of medicinal products... 6 5. Advertising
APMI CODE OF PRACTICE ON ADVERTISING OF MEDICINAL PRODUCTS ISSUE 1 August 2010 1. Introduction... 2 2. Scope... 3 3. Definitions... 4 4. General rules of advertising of medicinal products... 6 5. Advertising
MEMORANDUM. The Officers and Directors of the Company. Publicity Before and After Filing an IPO Registration Statement
 MEMORANDUM TO: FROM: The Officers and Directors of the Company Fenwick & West LLP DATE: December 15, 2011 RE: Publicity Before and After Filing an IPO Registration Statement United States federal securities
MEMORANDUM TO: FROM: The Officers and Directors of the Company Fenwick & West LLP DATE: December 15, 2011 RE: Publicity Before and After Filing an IPO Registration Statement United States federal securities
Attitudes and Beliefs About the Use of Over-the-Counter Medicines: A Dose of Reality
 Attitudes and Beliefs About the Use of Over-the-Counter Medicines: A Dose of Reality A National Survey of Consumers and Health Professionals Prepared for: National Council on Patient Information and Education
Attitudes and Beliefs About the Use of Over-the-Counter Medicines: A Dose of Reality A National Survey of Consumers and Health Professionals Prepared for: National Council on Patient Information and Education
DRAFT GUIDANCE. This guidance document is being distributed for comment purposes only.
 Providing Regulatory Submissions in Electronic and Non-Electronic Format Promotional Labeling and Advertising Materials for Human Prescription Drugs Guidance for Industry DRAFT GUIDANCE This guidance document
Providing Regulatory Submissions in Electronic and Non-Electronic Format Promotional Labeling and Advertising Materials for Human Prescription Drugs Guidance for Industry DRAFT GUIDANCE This guidance document
The Role of Advertising in America
 The Role of Advertising in America An Overview by the Association of National Advertisers (ANA) Introduction Advertising occupies a major place in American society. Linked to the bedrock principles that
The Role of Advertising in America An Overview by the Association of National Advertisers (ANA) Introduction Advertising occupies a major place in American society. Linked to the bedrock principles that
Guidance for Industry
 Guidance for Industry Toll-Free Number Labeling and Related Requirements for Over-the-Counter and Prescription Drugs Marketed With Approved Applications Small Entity Compliance Guide U.S. Department of
Guidance for Industry Toll-Free Number Labeling and Related Requirements for Over-the-Counter and Prescription Drugs Marketed With Approved Applications Small Entity Compliance Guide U.S. Department of
Prescription Drugs and Mass Media Advertising, 2000
 N O V E M B E R 2 0 0 1 Prescription Drugs and Mass Media Advertising, 2000 A research report by The National Institute for Health Care Management Research and Educational Foundation C O N T E N T S 1225
N O V E M B E R 2 0 0 1 Prescription Drugs and Mass Media Advertising, 2000 A research report by The National Institute for Health Care Management Research and Educational Foundation C O N T E N T S 1225
For a Healthier America: Reducing Prescription Drug Misuse and Abuse
 For a Healthier America: Reducing Prescription Drug Misuse and Abuse The misuse and abuse of prescription medicines is a growing public health problem. In addition to the tragic toll on families and communities,
For a Healthier America: Reducing Prescription Drug Misuse and Abuse The misuse and abuse of prescription medicines is a growing public health problem. In addition to the tragic toll on families and communities,
EPA s proposed hazardous waste pharmaceutical regulations
 EPA s proposed hazardous waste pharmaceutical regulations November 4, 2015 By J. Timothy Ramsey The United States Environmental Protection Agency ( EPA ) published proposed regulations in the Federal Register
EPA s proposed hazardous waste pharmaceutical regulations November 4, 2015 By J. Timothy Ramsey The United States Environmental Protection Agency ( EPA ) published proposed regulations in the Federal Register
Advertising guidelines for products registered under the ACVM Act
 Advertising guidelines for products registered under the ACVM Act ACVM guideline 1. Introduction 2. Advertising 3. Advertising DOs and DON Ts 4. Restricted veterinary medicines (RVMs) 5. Advertising conditions
Advertising guidelines for products registered under the ACVM Act ACVM guideline 1. Introduction 2. Advertising 3. Advertising DOs and DON Ts 4. Restricted veterinary medicines (RVMs) 5. Advertising conditions
A Middle Class in the United States? Behind the Counter (BTC) Drugs. Frederick H. Branding, R.Ph., J.D. Rahul Narula
 A Middle Class in the United States? Behind the Counter (BTC) Drugs Frederick H. Branding, R.Ph., J.D. Rahul Narula FDA s Regulation of Drugs Federal Food, Drug and Cosmetic Act (FDCA) passed by Congress
A Middle Class in the United States? Behind the Counter (BTC) Drugs Frederick H. Branding, R.Ph., J.D. Rahul Narula FDA s Regulation of Drugs Federal Food, Drug and Cosmetic Act (FDCA) passed by Congress
Direct-to-Consumer Prescription Drug Advertising: Understanding Its Consequences
 Direct-to-Consumer Prescription Drug Advertising: Understanding Its Consequences by Jisu Huh and Lee B. Becker James M. Cox Jr. Center for International Mass Communication Training and Research Grady College
Direct-to-Consumer Prescription Drug Advertising: Understanding Its Consequences by Jisu Huh and Lee B. Becker James M. Cox Jr. Center for International Mass Communication Training and Research Grady College
POLICY STATEMENT. Guidelines for Refractive Surgery Advertising. Statement of Purpose:
 POLICY STATEMENT Guidelines for Refractive Surgery Advertising Statement of Purpose: These guidelines are designed to assist ophthalmologists in providing truthful, informative advertising of refractive
POLICY STATEMENT Guidelines for Refractive Surgery Advertising Statement of Purpose: These guidelines are designed to assist ophthalmologists in providing truthful, informative advertising of refractive
Cancer Treatment Moringa Oleifera for Cancer Prevention or Treatment
 Cancer Treatment Moringa Oleifera for Cancer Prevention or Treatment As we learn more about cancer, we are empowered to use more of the tools which nature has created for us to help battle this terrible
Cancer Treatment Moringa Oleifera for Cancer Prevention or Treatment As we learn more about cancer, we are empowered to use more of the tools which nature has created for us to help battle this terrible
Thinking About Complementary & Alternative Medicine
 Thinking About Complementary & Alternative Medicine A guide for people with cancer PATIENT & family EDUCATION U.S. DEPARTMENT OF HEALTH AND HUMAN SERVICES National Institutes of Health National Cancer
Thinking About Complementary & Alternative Medicine A guide for people with cancer PATIENT & family EDUCATION U.S. DEPARTMENT OF HEALTH AND HUMAN SERVICES National Institutes of Health National Cancer
OPINION NO. 90-04 March 16, 1990
 ..,, OPINION NO. 90-04 March 16, 1990 FACTS: The inquiring attorney asks about the propriety of mailing to members of the bar and others a professional announcement regarding his election to membership
..,, OPINION NO. 90-04 March 16, 1990 FACTS: The inquiring attorney asks about the propriety of mailing to members of the bar and others a professional announcement regarding his election to membership
medicineupdate Tramadol for pain Asking the right questions about new medicines Page Section 1: What tramadol is 1 Section 2: What tramadol is for 1
 medicineupdate Asking the right questions about new medicines Tramadol for pain Page Section 1: What tramadol is 1 Section 2: What tramadol is for 1 Section 3: Who can take tramadol 2 Section 4: What does
medicineupdate Asking the right questions about new medicines Tramadol for pain Page Section 1: What tramadol is 1 Section 2: What tramadol is for 1 Section 3: Who can take tramadol 2 Section 4: What does
Direct-to-Consumer (DTC) Pharmaceutical Marketing: Impacts and Policy Implications
 SPNHA Review Volume 7 Issue 1 Article 5 10-19-2011 Direct-to-Consumer (DTC) Pharmaceutical Marketing: Impacts and Policy Implications Dayna M. Porter Grand Valley State University Follow this and additional
SPNHA Review Volume 7 Issue 1 Article 5 10-19-2011 Direct-to-Consumer (DTC) Pharmaceutical Marketing: Impacts and Policy Implications Dayna M. Porter Grand Valley State University Follow this and additional
COMMISSION AUTHORIZED
 V900033 UNITED STATES OF AMERICA FEDERAL TRADE COMMISSION WASHINGTON. D.C. 20S80 COMMISSION AUTHORIZED April 17, 1990 Bruce Hamilton Executive Director State Bar of Arizona 363 North First Avenue Phoenix,
V900033 UNITED STATES OF AMERICA FEDERAL TRADE COMMISSION WASHINGTON. D.C. 20S80 COMMISSION AUTHORIZED April 17, 1990 Bruce Hamilton Executive Director State Bar of Arizona 363 North First Avenue Phoenix,
University of Cincinnati College of Medicine
 University of Cincinnati College of Medicine Policy Policy and Guidelines for Industry Relationships Policy and Guidelines for Industry Relationships FAQ Date Updated Page 5/14/2008 2 -- 8 NB: The Institute
University of Cincinnati College of Medicine Policy Policy and Guidelines for Industry Relationships Policy and Guidelines for Industry Relationships FAQ Date Updated Page 5/14/2008 2 -- 8 NB: The Institute
Survey of Clinical Trial Awareness and Attitudes
 Survey of Clinical Trial Awareness and Attitudes Purpose of the Study Over $2 billion per year is spent on recruiting patients for clinical trials, yet slow patient enrollment is the primary cause of delay
Survey of Clinical Trial Awareness and Attitudes Purpose of the Study Over $2 billion per year is spent on recruiting patients for clinical trials, yet slow patient enrollment is the primary cause of delay
GOVERNMENT OF THE DISTRICT OF COLUMBIA Department of Health
 GOVERNMENT OF THE DISTRICT OF COLUMBIA Department of Health Prescription Drug Marketing Costs A Guide for Pharmaceutical Manufacturers and Labelers Published by the District of Columbia Department of Health
GOVERNMENT OF THE DISTRICT OF COLUMBIA Department of Health Prescription Drug Marketing Costs A Guide for Pharmaceutical Manufacturers and Labelers Published by the District of Columbia Department of Health
IMPORTANCE OF THE ENFORCEMENT OF THE LAW AND EU DIRECTIVES CONCERNING TRANSLATION OF PHARMACEUTICALS LEAFLETS
 IMPORTANCE OF THE ENFORCEMENT OF THE LAW AND EU DIRECTIVES CONCERNING TRANSLATION OF PHARMACEUTICALS LEAFLETS Qendro Gentiana Planetary University, Faculty of Medical Sciences, Department of Pharmacy,
IMPORTANCE OF THE ENFORCEMENT OF THE LAW AND EU DIRECTIVES CONCERNING TRANSLATION OF PHARMACEUTICALS LEAFLETS Qendro Gentiana Planetary University, Faculty of Medical Sciences, Department of Pharmacy,
PRACTICAL HELP FROM THE ARTHRITIS FOUNDATION. www.arthritis.org 800-283-7800. Psoriatic Arthritis
 Psoriatic Arthritis WHAT IS PSORIATIC ARTHRITIS? Psoriatic (sore-ee-aah-tick) arthritis is a condition that causes pain and swelling in joints and scaly patches on the skin. Psoriatic arthritis occurs
Psoriatic Arthritis WHAT IS PSORIATIC ARTHRITIS? Psoriatic (sore-ee-aah-tick) arthritis is a condition that causes pain and swelling in joints and scaly patches on the skin. Psoriatic arthritis occurs
CORNERSTONE THERAPEUTICS INC. SECOND AMENDED AND RESTATED CODE OF BUSINESS CONDUCT AND ETHICS
 CORNERSTONE THERAPEUTICS INC. SECOND AMENDED AND RESTATED CODE OF BUSINESS CONDUCT AND ETHICS This Second Amended and Restated Code of Business Conduct and Ethics (the Code ) sets forth legal and ethical
CORNERSTONE THERAPEUTICS INC. SECOND AMENDED AND RESTATED CODE OF BUSINESS CONDUCT AND ETHICS This Second Amended and Restated Code of Business Conduct and Ethics (the Code ) sets forth legal and ethical
ACTIVELY MANAGED DRUG SOLUTIONS. for maintenance and specialty medication. Actively Managed Drug Solutions is not available in the province of Quebec
 ACTIVELY MANAGED DRUG SOLUTIONS for maintenance and specialty medication Actively Managed Drug Solutions is not available in the province of Quebec ARE YOU UNDERESTIMATING THE IMPACT OF CHRONIC DISEASE?
ACTIVELY MANAGED DRUG SOLUTIONS for maintenance and specialty medication Actively Managed Drug Solutions is not available in the province of Quebec ARE YOU UNDERESTIMATING THE IMPACT OF CHRONIC DISEASE?
Direct-to-Consumer Advertising of Prescription Drugs: Exposure and Response
 Direct-to-Consumer Advertising of Prescription Drugs: Exposure and Response November 2010 Direct-to-Consumer Advertising of Prescription Drugs: Exposure and Response Report Prepared by Helen Brown COPYRIGHT
Direct-to-Consumer Advertising of Prescription Drugs: Exposure and Response November 2010 Direct-to-Consumer Advertising of Prescription Drugs: Exposure and Response Report Prepared by Helen Brown COPYRIGHT
Coventry Health Care of Georgia, Inc. Coventry Health and Life Insurance Company
 Coventry Health Care of Georgia, Inc. Coventry Health and Life Insurance Company PRESCRIPTION DRUG RIDER This Prescription Drug Rider is an attachment to the Coventry Health Care of Georgia, Inc. ( Health
Coventry Health Care of Georgia, Inc. Coventry Health and Life Insurance Company PRESCRIPTION DRUG RIDER This Prescription Drug Rider is an attachment to the Coventry Health Care of Georgia, Inc. ( Health
U.S. Department of Health and Human Services Food and Drug Administration Center for Drug Evaluation and Research (CDER) September 2009 OTC
 Guidance for Industry Labeling of Nonprescription Human Drug Products Marketed Without an Approved Application as Required by the Dietary Supplement and Nonprescription Drug Consumer Protection Act: Questions
Guidance for Industry Labeling of Nonprescription Human Drug Products Marketed Without an Approved Application as Required by the Dietary Supplement and Nonprescription Drug Consumer Protection Act: Questions
AAD ADVERTISING STANDARDS
 Approved: Board of Directors 11/14/09 AAD ADVERTISING STANDARDS The American Academy of Dermatology and AAD Association (collectively, the Academy ) seeks to advance the science and art of dermatology
Approved: Board of Directors 11/14/09 AAD ADVERTISING STANDARDS The American Academy of Dermatology and AAD Association (collectively, the Academy ) seeks to advance the science and art of dermatology
The History of the Legal And Regulatory Issues Surrounding Pharmacy Compounding
 The History of the Legal And Regulatory Issues Surrounding Pharmacy Compounding T.C. Spencer Pryor Partner, Alston & Bird LLP FDA & CDC: Collaboration & Challenges FSMA, Food Outbreaks, Pharmacy Compounding
The History of the Legal And Regulatory Issues Surrounding Pharmacy Compounding T.C. Spencer Pryor Partner, Alston & Bird LLP FDA & CDC: Collaboration & Challenges FSMA, Food Outbreaks, Pharmacy Compounding
How To Weigh Data From A Study
 PRINCIPLES ON RESPONSIBLE SHARING OF TRUTHFUL AND NON-MISLEADING INFORMATION ABOUT MEDICINES WITH HEALTH CARE PROFESSIONALS AND PAYERS INTRODUCTION In the era of data-driven medicine, where all parties
PRINCIPLES ON RESPONSIBLE SHARING OF TRUTHFUL AND NON-MISLEADING INFORMATION ABOUT MEDICINES WITH HEALTH CARE PROFESSIONALS AND PAYERS INTRODUCTION In the era of data-driven medicine, where all parties
