GUIDELINES FOR THE ADMINISTRATION OF DRUGS THROUGH ENTERAL FEEDING TUBES
|
|
|
- Hortense Lang
- 9 years ago
- Views:
Transcription
1 GUIDELINES FOR THE ADMINISTRATION OF DRUGS THROUGH ENTERAL FEEDING TUBES 2 nd Edition October 2003 NJWATT/PHARM/Review Date October 2004
2 Disclaimer Representations in this guide are believed to be true and accurate. The Primary Care Trusts, NHS Trusts, and their employees, or agents accept no liability for loss of any nature to persons, organisations, or institutions which may arise as a result of errors. Users of this guide are reminded that crushing medication or opening a capsule to aid administration results in unlicensed administration. It is recommended that unlicensed administration methods are authorised by the prescriber and documented in writing. 2
3 Contents Legal aspects of unlicensed administration 4 General Principles 5 Administration Instructions 9 Interactions with feed 10 Jejunostomy Tubes 11 List of preparations 14 Bibliography 24 3
4 UNLICENSED ADMINISTRATION Although nurses, midwives and health visitors are advised not to crush medication there will be occasions where the patient is unable to swallow solid oral dosage forms, or medication is required to be administered via enteral tube. In many cases an alternative route of administration or suitable liquid formulation is available and should be used in the first instance. If alternative formulations or methods of administration are unavailable and the medication is still indicated then it may be necessary to crush tablets or open capsules. The practitioner involved should bring this to the attention of the prescriber (and the pharmacist). Authorisation for this unlicensed administration together with the reasons why must be documented in the patients medical record by the prescriber. Crushing medication or opening capsules prior to administration usually results in unlicensed administration. Under the Medicines Act 1968 only Medical and Dental practitioners can authorise the administration of unlicensed medicines. It may therefore be illegal to open a capsule or crush a tablet before administration without the authorisation of the prescriber. Where such administration results in harm to the patient then the manufacturer has no liability for any harm that ensues and if the unlicensed administration is unauthorised by the prescribing doctor then liability may lie solely with the administering nurse. Where unlicensed administration is authorised by a prescriber then the liability is shared between the prescriber and the administering nurse. If this is on the advice on a pharmacist then the liability is shared between all three professionals. The balance of this liability would be assessed in a court of law on an individual case basis. In practice providing the prescriber has authorised the administration and advice has been taken from a pharmacist regarding the suitability of a preparation for unlicensed administration then the nurse would usually be seen as having acted in an appropriate and professional manner. Providing these steps have been taken any adverse effects resulting from the unlicensed administration are unlikely to occur. It should be borne in mind that usually it is in the best interests of the patient to administer the drug even if this is in an unlicensed manner. The consequences of not giving the medication usually outweigh any risk associated with unlicensed administration. Nursing staff should take reasonable steps to ensure that unlicensed administration does not cause harm to the patient even if this is at the request of a prescriber. Advice from a pharmacist or referral to these guidelines should be the absolute minimum action undertaken. Within the acute Trust pharmacy advice is available 24 hours a day via the on call pharmacist. Any adverse effects, including problems with feeding tubes, should be reported to the prescriber and pharmacist immediately. 4
5 GENERAL PRINCIPLES When administering medication via enteral tube consideration needs to be taken regarding drug formulation, interactions with feed, type of tube, site of placement, and site of drug absorption. Usually drugs are not licensed for enteral administration and there is little information available in the literature on any aspects of enteral drug administraton. Most recommendations are therefore theoretical. CHOICE OF ROUTE Where a drug is needed but the oral route is unavailable or available by tube only, consideration should first be given to alternative methods of administration. Intravenous administration is often an option but requires trained staff to administer, is associated with increased risk to the patient from complications and is usually more expensive. Other routes for consideration include transdermal, rectal, buccal or sublingual, nebulisation, vaginal, as well as alternative parenteral routes im, iv, sc. THE NEED FOR MEDICATION SHOULD BE REVIEWED BEFORE SEARCHING FOR AN ALTERNATIVE ROUTE In patients who are very unwell previous medication may no longer be necessary both in the short and long term and prescribed items should be reviewed and stopped if no longer indicated or of benefit to the patient. Patients undergoing surgery who are nil by mouth can often have medication discontinued temporarily without harm e.g. statins or replaced by an alternative agent e.g. sc low molecular weight heparin in place of warfarin. Sometimes an alternative preparation from the same or even a different therapeutic group may be more suitable eg calcium antagonists for hypertension are not available as liquid formulation but diuretics or beta blockers are. Where drugs need to be continued and no other route is available then enteral administration may need to be considered. Pharmacists should identify patients receiving medication via the enteral route and be proactive in reviewing their medication and in advising nursing staff / medical staff to facilitate drug administration. Equally nursing staff should request pharmacy advice in patients where drug administration via the enteral route is deemed necessary and refer to current policies and guidelines where available. Sometimes in patients where the enteral route is not available e.g. after surgery where there is free drainage or suction where aspirates may be large it may be feasible (after consultation with medical staff) to suspend drainage or suction for long enough to facilitate administration and absorption. It should be understood that in such circumstances absorption may be at best be variable and drug effects should be closely monitored. Prokinetic agents may be used to encourage the restoration of normal gut function e.g. metoclopramide, erythromycin, or domperidone. POTENTIAL PROBLEMS May include: unavailibility of suitable formulation, blocking of tubes, diarrhoea due to hyperosmolar formulation, and drug interactions (between different drugs, between drug and feeds, between drug and tube) 5
6 Tube types Fine bore tubes are unsuitable for some thick liquid formulations eg lansoprazole suspension sachets, cholestyramine. Granules may also block fine bore tubes. Tube Placement There are more likely to be problems with drug absorption where the tube is placed beyond the stomach e.g. percutaneous endoscopic jejunostomy (PEJ) tubes. Staff should be aware of situations in which the drug may be administered beyond the drugs main site of absorption eg digoxin, cephalexin, ketoconazole, phenytoin and other anticonvulsants. In such circumstances it is important to monitor for signs of reduced drug efficacy. Therapeutic drug monitoring may be necessary. When a tube is placed beyond the stomach sterile water must be used for flushing and drug administration. FORMULATIONS Liquid formulations Drugs for administration via enteral feeding tubes should ideally be liquids or disperible/soluble formulations. Not all drugs are commercially available as liquids but pharmacy may be able to organise certain liquid preparations on request if feeding is likely to be long term. In addition to proprietory products listed in the BNF liquid medicines may also be available as a 'special' from other hospitals or manufacturers. In exceptional circumstances extemporaneously dispensed liquid formulations may be manufactured 'in house' by the hospital pharmacy. It should be noted that whilst the use of specials may be suitable to solve a short term problem in the acute setting this may not always be the most appropriate or cost effective method for long term use especially where patients are to be transferred to primary care. Even when a medicine is available as a liquid formulation several other factors may need to be considered. E.g. lansoprazole suspension is too viscous for administration via fine bore tube. Some manufacturers advise dilution of suspension prior to administration through tubes. Augmentin suspension should be diluted to half strength to avoid 'caking'.the administration of sucralfate suspension has been associated with 'bezoar' formation in patients with impaired gastric emptying. Diazepam suspension is not recommended for enteral tube administration as it is absorbed onto plastic tubing. When transferring a patient to a liquid formulation the use of a different salt may require dosage adjustment due to differing bioavailibilities e.g. phenytoin, mebeverine, and chlormethiazole. Liquid formulations of high osmolality particularly those containing sorbitol can be associated with osmotic diarrhoea, bloating, stomach cramps, and delayed gastric emptying. This is more likely to be a problem when a lot of medicines are given as liquid formulations in high volumes. Many liquids have an osmolality as high as 1000 mosm/kg compared with the normal osmolality of GI secretions which is mosm/kg. This is not usually a problem where small volumes are administered e.g. digoxin elixir, but may be avoided by dilution with water immediately prior to administration. The sorbitol content of most liquid medicines is not readily available but can be obtained where necessary from individual manufacturers. Cumulative sorbitol doses above 20g may cause particularly severe adverse effects. Administration of hyperosmolar solutions may be particularly problematical when administered through a jejunostomy tube. 6
7 Generally elixirs or suspensions should be used rather that rather than syrups. s because of their low ph are prone to clumping in contact with GI feeds. Where a liquid preparation is unavailable or unsuitable soluble or dispersible tablets may be available. Often tablets not specifically marketed as dispersible will disperse in water. Again advice should be sought from pharmacy. Solid Dosage Forms tablets and capsules Sometimes it may be necessary to crush tablets or open capsules prior to flushing down enteral tubes. Not all solid dosage forms are suitable for crushing. Generally plain uncoated tablets will either disperse in water or can be crushed to a fine powder for dispersal in water. Film coated or sugar coated tablets can usually be crushed as the coating is designed to improve appearance and to mask taste. These obviously do not matter if being administered via enteral tube. The following formulations should not be administered through enteral feeding tubes without pharmacy advice Enteric or other coated tablets Enteric coatings are designed to protect drugs from attack by stomach acid or to protect the stomach from irritation by the drug. Beyond the stomach the coating breaks down to allow drug absorption. Enteric coated tablets should not be crushed as this destroys the coating resulting in inactivation of drug or increased risk of adverse effect. Controlled, slow, or extended release products These products are formulated to allow a gradual release of drug as the preparation passes down the GI tract. This may enable less frequent dosing or minimise side effects due to peak concentrations of drug in the body. Crushing controlled release formulations may destroy their controlled release properties resulting in adverse effects and shortening duration of action. Deaths have been reported as a result of crushing controlled release morphine preparations. These types of preparation must never be crushed. Some controlled release products are formulated as granules inside a capsule. Similarly these granules must not be crushed but may be suitable for administration via enteral tube. Buccal/ Sublingual tablets should not be crushed and administered via an enteral tube as this can result in reduced effectiveness due to increased first pass metabolism at the liver. Hard gelatin capsules may often be opened and the contents mixed with water prior to tube administration. Some capsules contain enteric coated granules eg lansoprasole, or granules with a slow release coating eg Slophylline and should not be crushed prior to administration. The administration of such granules intact may block fine bore tubes. The contents of some soft gelatin capsules may be withdrawn and administered via tube eg nifedipine. Doxycycline capsules contain an irritant salt and should not be opened to facilitate administration. Dispersible tablets should be used instead. 7
8 Injections Occasionally parenteral formulations may be given orally e.g. vancomycin injection but this is usually an expensive option and not all injectable formulations are suitable for this route. eg gentamcin is not absorbed, amiodarone injection is not suitable because of its high polyethylene glycol content. Advice should be sought from pharmacy. Cytotoxics Advice must be sought from pharmacy regarding the administration of cytotoxic drugs via enteral tube. The document Guidelines for the Handling and Administration of Cytotoxic Drugs should also be referred to. Oral Syringes Where possible oral syringes should be used to draw up and administer medicines. Fatalities have been reported when medicines intended for oral administration have accidently been given intravenously because they were drawn up in IV syringes. Ideally oral syringes should be incapable of attachment to IV lines or needles by virtue of their design. 8
9 Practical Aspects of Administration 1) Medications should not be added directly to nutrition formulations. 2) Liquid dosage forms should be used where available (Check with pharmacy re liquid availibility.) Most liquid formulations should be diluted with 30ml sterile water to reduce their osmolarity prior to administration. Some liquid medicines however should not be diluted. Please refer to list of preparations. 3) If tablets must be used the nurse should first check to see whether the tablet can be crushed. If so the tablet should be crushed into a fine powder and mixed with 10 to 15ml of water. Generally sustained release or enteric coated medication should not be crushed. If in doubt advice should be sought from pharmacy. 4) If capsules can be opened then they should be opened and the powder mixed with 10 to 15ml of sterile water. 5) In certain circumstances medication formulated for injection may be administered orally (unlicensed use). Advice on suitability should be sought from pharmacy. 6) The tube should be flushed with 30ml of sterile water prior to administration of the drug. 7) The medication should be remixed immediately prior to administration by swirling the medication in the cup. In the case of liquid formulations these should be shaken prior to withdrawal from the bottle. 8) The medication should be drawn up into a needleless oral or catheter tipped syringe. 9) When more than one medication is administered then 5ml of sterile water should be administered between drugs to avoid drug interactions. Medications should never be mixed together in a syringe. Smaller volumes may be necessary in babies and children. 10) After administration the tube should be flushed with 30ml of sterile water and clamped. 11) The amount of water used should be documented on fluid balance sheets. 9
10 Medicines for which there are documented interactions with enteral nutrition Where there is doubt about compatibility further information should be obtained from pharmacy. Medication Problem Recommendation Carbamazepine Decreased absorption. Hold enteral nutrition 2hr before and 2hrs after dose. May adhere to tubing. Monitor levels. Suppositories are available. If used the dose will need to be adjusted Ciprofloxacin Flucloxacillin Absorption decreased by 25-50% due to chelation with divalent cations in feed Absorption decreased by presence of feed Hydralazine Decreased absorption Adjust dose. Monitor BP Hold enteral nutrition 1hr before and 2 hrs after dose or administer higher dose. Consider IV therapy Hold enteral nutrition 1 hr before and 2 hr after dose or administer higher dose Penicillin V Phenytoin sodium Sucralfate Tetracyclines Theophylline Thyroxine Warfarin Unpredictable absorption May be reduced by 30-80% May form phenytoincalcium or phenytoinprotein complexes with feed Absorption may be reduced by 75% Binds to protein in the feed Absorption reduced by calcium in feed Absorption decreased may be reduced by 60-70% Reduced absorption. Particularly with soya based products. May interact with Vit K and soya proteins in feed. Stop feed 1 hr prior to and 2 hours after administration, or use higher doses, or use amoxycillin. Usually given once daily so administer during feeding break or stop feed 2 hours before and after dose Use alternative agent e.g. H2 antagonist, or PPI. Consider alternative antibiotic or feeding regimen. Discuss with medical staff and pharmacy. Stop feed 1 hr before and 2 hrs after administration. This may be impractical due to frequency of administration of non SR liquid preparation. Monitor levels. Monitor thyroid function. Avoid soya based products. Monitor INR closely including when changing from or back to normal diet. In problem patients consider non soya based feed. 10
11 Holding enteral nutrition in order to administer medications may become problematic. For products that require enteral nutrition to be held for one hour before and one or two hours after drug administration then the following procedure should be adopted. 1) If enteral nutrition is administered in bolus doses then the drug should be scheduled during the off periods. 2) If enteral nutrition is administered continuously and the drug is available in an alternative dosage form (eg suppository, injection) the oral dosage form should be switched to a therapeutically equivalent dosage form and the patients clinical response to therapy monitored. 3) If enteral nutrition is administered continuously and the drug is not available in an alternative dosage form, or the patient does not have intravenous access, enteral nutrition should be withheld for 1 hour before and 1 hour after drug administration. Drug levels should be monitored where appropriate and the patient s clinical response to therapy monitored. If it is not feasable to hold enteral feeding then the oral dosage should be increased by the amount expected to be lost as a result of the interaction. e.g. phenytoin 300mg daily may be changed to 400mg- 500mg daily because of an expected 70-80% reduction in absorption when combined with enteral nutrition and phenytoin levels should be monitored. Advice on dosage adjustments can be obtained from pharmacy. Clinical response and/or drug serum levels should be monitored where appropriate. Administration of drugs via jejunostomy tube There are no drugs licensed for intrajejunal administration in the UK and little data on absorpion of drugs via this route. Other routes or alternative methods of treatment should therefore be sought wherever possible. Clinical response and drug levels should be monitored as a guide to treatment success. The normal secretions in the jejunum has an osmolality of 285±10 mosm/kg. When a hypertonic solution is rapidly infused via a jejunostomy tube there is an influx of water and electrolytes into the small intestine leading to an osmotic diarrhoea. Many liquid drug formulations have osmolalities exceeding 1000 mosm/kg particularly those sugar free preparations containing sorbitol. In circumstances where this is likely to be a problem the following may be considered. 1) Sterile water should be used to flush jejunostomy tubes and disperse tablets 2) Use disperible tablets or finely crushed tablets dispersed in water as an alternative to liquid formulations 3) If the parenteral form is isotonic or hypotonic this may be used as an alternative to liquid formulations. Check with pharmacy re suitability. Not all injections can be given in this manner. 4) Only where a drug has been shown to be compatible with enteral feed may a liquid preparation be mixed with feeding solution to reduce osmolality. In practice there is little data regarding compatibility with feeds and most feed manufacturers do not recommend this. 11
12 Drugs for which there is documented advice for administration via jejunostomy tube. Medication Amoxycillin Aspirin Cephalexin Cimetidine Ciprofloxacin Digoxin Flucloxacillin Frusemide Ketoconazole Loperamide Nifedipine Omeprazole Paracetamol Phenytoin Ranitidine Comments Administer reconstituted injection as suspension has high osmolality Use dispersible tablets Administration by this route may result in decreased plasma concentrations. Company data suggests sufficient absorption via this route. Use crushed tablets dispersed in water. Absorption may be less than conventional oral administration. Do not administer at same time as feed. Monitor clinical response or use IV route. Liquid formulation is hypertonic. Injection may be given via tube Elixir and injection have high osmolalities. Use crushed tablets dispersed in water. Monitor levels and adjust dose as necessary Use reconstituted injection (as amoxycillin). Absorption decreased by food so stop feed at time of administration Possibly reduced absorption when given by this route. Monitor clinical effect and adjust dose as necessary. Not recommended as a low ph is required for absorption Loperamide is thought to act locally in the colon so should be suitable for JEJ tube administration. The commercial liquid formulation should be suitable for JEJ tube administration. Absorption thought to occur in small intestine. Administer contents of capsule immediately as liquid is very sensitive to light. Monitor clinical effect eg Blood pressure Use omeprazole MUPS tabs Good absorption when administered by this route. Do not mix with enteral feed. Monitor levels. Use injection rather than liquid preparation 12
13 Is the drug prescribed via the NG/PEG route? May wish to consider alter-native routes of administra-tion, eg topical, buccal, transdermal, rectal Yes If not appropriate No Is the drug available in tablet form? Is there a commercially available liquid preparation? (check BNF) No Is the drug available in capsule form? Yes No Yes Is the sorbitol content and osmolality of the liquid suitable for NG/PEG administration and are there no known interactions/incompatibilities with feed? Use liquid Yes Is there a commercially available soluble/dispersible tablet? No Is it a hard gelatin capsule that can be opened? Yes No No Can the tablet be dispersed? No Can the tablet be crushed? Do not crush if: Enteric coated Slow/modified Release Seek pharmacy advice if: Cytotoxic Hormone Prostaglandin analogue No Can the pharmacy dispensary prepare a solution/suspension? No Can the injection be used? No CONSIDER ALTERNATIVE No Can the capsule contents be flushed down the tube with water? Refer to guidelines. No Can the capsule contents be removed with a syringe and needle? Refer to guidelines Remember to confirm unlicensed administration with prescriber NJWATT/PHARM/Review Date October 2004
14 List of Preparations This list is intended to be used as a guide to the range of medicines which are available as liquid preparations and which solid dosage forms can be successfully given via tube. This may be useful when prescribing for patients with swallowing difficulties as well as tube administration. Inclusion in this list does not always mean that liquids are suitable for enteral administration. A minority of medicines althougth formulated as liquids may not be suitable for tube administration e.g. lansoprazole suspension. Many tablets not marketed as soluble or dispersible may disperse in water to form a suspension which is suitable for administration via enteral tube or to patients with swallowing difficulties. Tablets should only be dispersed immediately prior to, or shortly before administration. Some hard gelatin capsules may be pulled apart and and the contents dispersed in water prior to administration. Drug Strength Comments Acetazolamide Aciclovir Aledronate Alfacalcidol 200mg/5ml Dispersible tablets 2 microgram/ml Approx 100 nanograms per drop Disperses in water with fine powder sediment. Injection may be given orally. No liquid formulation available. Ask Pharmacy for advice. Alfuzosin Crush tablets and disperse in water Allopurinol Disperses in water Aluminium Hydroxide Mixture May interact with feed to form protein-albumin complexes 4% w/w Amantadine 50mg/5ml. Contains sorbitol. Amiloride 5mg/5ml. Aminophylline Phyllocontin tabs should not be crushed. Transfer patient to theophylline (See below) or contact pharmacy for advice. Amiodarone Disperses in water. Injection is not suitable for oral administration because of high polyethylene glycol content. Amitriptyline Oral Soln 25mg/5ml 50mg/5ml. Amlodipine Amoxapine Amoxycillin Anastrazole Asasantin Ascorbic acid Aspirin Atenolol Atorvastatin Augmentin Azathioprine 125mg/5ml 250mg/5ml 25mg/5ml Dispersible tabs 250/125mg 125/31/5ml 250/62/5ml Disperses in water. Give immediately as light sensitive Crush tablets and disperse in water. Powder for reconstitution with water. Capsules may be opened and contents dispersed in water. Crush tablet in water. Give immediately. Open capsules. Administer granules and dispersible aspirin. Granules must not be crushed. May block fine bore ng tubes Crush tablets and disperse in water Effervescent tablets available Dispersible tablets available. Enteric coated tablets should not be crushed. Suppositories may also be available. Crush and disperse in water. Poorly soluble slurry may block tube.. Dilute to half strength with water prior to administration via enteral tube. Disperses in water. Take cytotoxic precautions if crushing tablets. If oral route not feasible injection may be available. Contact pharmacy for advice. NJWATT/PHARM/Review Date October 2004
15 Baclofen Liquid 5mg/5ml Balsalazide Open capsule and disperse contents in water. May block fine bore tubes. Bendrofluazide Tablets disperse in water. Benzhexol Liquid formulation may be available. Contact pharmacy for advice. Benztropine Injection can be used orally Contact pharmacy for advice. Bezafibrate Crush tablets and disperse in water. Do not crush Belazip mono. Contact pharmacy for advice. Bisoprolol Crush tablets and disperse in water. Bisacodyl Suppositries available. Bromocriptine Crush tablets and disperse in water Bumetanide 1mg/5ml. Buspirone Crush tablets and disperse in water. Calcichew Calcichew D3 Calcichew D3 Forte Crush tabs and disperse in water. Change to alternative preparation eg. Calcium Sandoz syrup. Cacit D3 sachets may be available. Ask pharmacy for advice. Calcium Sandoz 8.1mmol/15ml Contains sucrose Calcium Resonium Caffeine citrate Mixture 10mg/ml Contact pharmacy for advice. may be available. Captopril Disperses in water with fine powder sediment Carbamazepine. 100mg/5ml Shake well before use. Contains sorbitol. Dilute before use with equal volume of water. Use suppositories as an alternative. Suppositories only licensed for 7 days use. 100mg orally is approximately equivalent to 125mg suppository. Retard formulation should not be crushed. Carbimazole Crush tablets and disperse in water. Carvediol Crush and disperse in water Cephalexin 125mg/5ml Powder for reconstitution with water. Cetirizine Oral Solution Sugar Free 5mg/5ml Chloral Hydrate Elixir 143mg/5ml 500mg/5ml Chlordiazepoxide Open capsules and mix powder with water. Chlormethiazole 250mg/5ml Store in fridge. 250mg of edisylate salt is equivalent to one capsule. Chlorothiazide 250mg/5ml Chlorthalidone Chlorpheniramine Chlorpromazime Cimetidine Cinnarizine Ciclosporin Ciclosporin (Sandimmun) 2mg/5ml 25mg/5ml 100mg/5ml 200mg/5ml Oral solution. (Neoral)100mg/ 5ml Oral soln 100mg/5ml Disperses with fine powder sediment. may be available. Crush tablets and disperse in water.. Must not be administered with grapefruit or grapefruit juice. Solution may be mixed with orange juice or apple juice to improve taste. Administer immediately. Monitor ciclosporin level when changing between preparations. Must not be administered with grapefruit or grapefruit juice. To mask taste solution may be mixed with cold milk, cold chocolate drink, cola, or orange juice immediately before taking. Do not use plastic cup. Available on a named patient basis only 15
16 Ciprofibrate Ciprofloxacin Citalopram Clarithromycin Clindamycin Clobazam Clonazepam Clomipramine Clonidine Clopidogel Clozapine Co-beneldopa (Madopar) 125mg/5ml Dispersible Tablet 62.5mg, 125mg Oral drops may be available. Contact pharmacy. Crush and disperse tablets in water. Disperses in water. may be available. Absorbed in the duodenum. When administered via the jejunum absorption may be incomplete. Interacts with feed, see above. Oral drops are available but are not directly bioequivalent to the tablets. 8mg (4 drops) of liquid may be considered to be therapeutically equivalent to a 10mg citalopram tablet. Tablets may be crushed and dispersed in water. Granules for reconstitution Disperse contents of capsule in water prior to administration. Open capsule and mix contents with water. Special suspension may be available. Please as pharmacy for advice. Disperses in water. may be available may be available. Contact pharmacy for advice. Open capsules and mix contents with water. Catapres injection may be given orally. Please contact pharmacy for advice. Plavix tablets may be crushed and dispersed in water. Special suspension may be available. Contact pharmacy for advice. Faster onset of action compared to plain tablets. Dose and dosage frequency may need adjustment when changing from other forms. Give at same time each day in relation to feeding. Madopar capsules should not be opened Co-careldopa (Sinemet) Tabs disperse in water. CR formulations should not be crushed. Dose and dosage frequency may need adjustment when changing from other forms. Give at same time each day in relation to feeding Co-codamol 8mg/500mg Available as effervescent tablets. Note high sodium content. Paracetamol available as suppositories or liquid. Co-danthramer, Strong Co-danthrusate Codeine phosphate Colchicine Co-trimoxazole Linctus 15mg/5ml 25mg/5ml 240mg/5ml Change to Co-danthramer suspension Crush tablets and disperse in water Contains sorbitol Creon Cyclizine Cyproterone Dantrolene Dapsone Demeclocycline Dexamethsone Desmopressin 480mg/5ml Oral Soln. 2mg/5ml Contains sucrose Contents of capsules may be administered in liquid without crushing. For patients with swallowing difficulties mix with fluid or soft food. Sachets may be available. Contact pharmacy for advice. Crush tablets and disperse in water. Give immediately as light sensitive. Disperses in water Open capsules and mix with fruit juice. Crush and disperse tablets in water Open capsules and flush contents with water. Tablets may be crushed. Consider nasal spray or sc injection 16
17 Diazepam Diclofenac Digoxin Dihydrocodeine Diltiazem Dipyridamole Oral solution. 2mg/5ml 5mg/5ml Dispersible tabs 50mg 50microgram in 1ml Elixir 10mg/5ml Capsules Tablets Various strengths Oral 50mg/5ml Some preparations contain sorbitol. Drug binds to PVC tubing so efficacy may be reduced. Rectal dosage form available. Suppositories also available. When changing from tablets patients on larger oral doses may need dose adjustment. Monitor plasma levels. Bioavailability of the liquid is 80% and the tablets 70%. A 62.5microgram tablet is equivalent to 50 microgram (1ml) of elixir) Do not dilute. May be given IV as an alternative. If so the dose must be reduced by 20-30% Monitor plasma levels Martindale brand contains sucrose. Capsules may be opened and granules administered without crushing. Modified release tablets should not be crushed.. Intact granules from a modified release capsule may be given but may block fine bore tubes. Granules must not be crushed. Docusate sodium Oral Solution 50mg/5ml Domperidone 1mg/ml Contains sorbitol. Suppositories also available Donepezil Crush tablet and disperse in water. Dothiepin Oral solution. Contains sorbitol. 25mg/5ml Capsules may be opened and contents dispersed in water. Doxazosin Crush and disperse tablet in sterile water. Do not use tap water. Do not crush Doxazosin XL Doxepin Open capsules and mix contents with water Doxycycline Dispersible tab Do not open capsules. Use dispersible tablet. 100mg Enalapril Entacapone Erythromycin Ethambutol Ethamsylate Ethosuximide Etidronate Felodipine Fenofibrate Ferrous sulphate Flucloxacillin Flecainide Fluconazole Fludrocortisone 125mg/5ml 250mg/5ml 250mg/5ml 125mg/5ml 250mg/5ml 50mg/5ml 200mg/5ml Disperses slowly in water with stirring Crush and disperse tablet in water. May stain the tube orange so flush with plenty of water. For oral administration it may be given in jam, honey or orange juice. Powder for reconstitution Crush tablet and disperse in water. may be available contact pharmacy for advice. Dicynene tablets may be crushed and dispersed in water. Tablet disperses freely in water. Feed should be stopped 2 hours before and after administration. Do not crush as will lose sustained release properties. Lipantil Micro capsules may be opened and the contents dispersed in water. Use sodium feredetate (ironedetate) elixir. 190mg/5ml equivalent to 27.5mg iron/5ml Powder for reconstitution. Crush tablets or use injection orally. Do not mix with alkali solutions, sulphate, phosphate or chloride ions. Use sterile water, not tap water. may be available from Penn pharmaceuticals. Powder for reconstitution with water. Disperses in water 17
18 Fluoxetine Flupenthixol Folic acid Frusemide Fusidic Acid Gabapentin Galantamine Glibenclamide Gliclazide Glipemiride Glipizide Griseofulvin Haloperidol Hydralazine Hydrocortisone Hydroxychloroquine Hydroxyzine Hyoscine butylbromide Hyoscine hydrobromide Ibuprofen Imipramine Indapamide Indomethacin Irbesartan Isoniazid Isosorbide mononitrate Itraconazole Ketoconazole Lamotrigine Lansoprazole Leflunomide Levetiracetam Liquid 20mg/5ml 2.5mg/5ml Oral solution 20mg/5ml 40mg/5ml Paediatric liq 5mg/5ml 250mg/5ml Oral suspension 125mg/5ml Liquid 2mg/ml 10mg/5ml 100mg/5ml 25mg/5ml 25mg/5ml Elixir 50mg/5ml Oral liquid 50mg/5ml Dispersible tabs 5mg,25mg, 100mg Does not contain sorbitol. Fluoxetine is a severe eye irritant. Administer with care. Crush tablets in fruit juice and give immediately Sugar Free Fucidic acid suspension is incompletely absorbed and doses recommended for suspension are proportionately higher than for sodium fusidate tablets. Open capsules and disperse contents in water. Contents of capsules can be sprinkled on food for patients with swallowing difficulties. Use immediately as drug is rapidly hydrolysed. If given orally fruit juice may be used to mask unpleasant taste. Crush and disperse tablet in water. Crush tablets and mix with water. Monitor blood glucose. Consider insulin. Disperses in water. May be a risk of increased absorption. Monitor blood glucose. Consider insulin as above. Not recommended. Crushing may effect bioavailibility. As for gliclazide. Sugar Free Crush and disperse in water. May be increased rate of absorption so monitor blood glucose. Disperses in water. Crush and disperse tablet in water. Tablets may be crushed and dispersed in water. Injection can be given orally. Consider Patches. also available. Tofranil syrup now discontinued. Alternative liquid preparation may be available. Contact pharmacy for advice. Crush and disperse tablets in water. Do not crush Natrilix SR. may not be available. Use suppositories as alternative management option. Use alternative NSAID e.g. Diclofenac, ibuprofen. Crush and disperse tablet in water. Disperses in water. Increased rate of absorption may lead to increased side effects. Do not take with food Crush tablet and disperse in water. The use of lansoprazole suspension via enteral feeding tube is not recommended due to its high viscosity and adherence to tubing. Use lansoprazole fast tabs or consider changing to omeprazole MUPS tabs. Crush and disperse in water. Kepra tablets may be crushed and dispersed in water. Manufacturers recommend half a glass. 18
19 Levomepromazine Lithium Lisinopril Lofepramine Loperamide Loratadine Lorazepam Lormetazepam Madopar Mebendazole Mebeverine Medroxyprogesterone Mefenamic Acid Megestrol Menadiol sodium phosphate Metformin Methyldopa Mesalazine Metoclopramide Metolozone Metoprolol Metronidazole Mirtazapine Misoprostol Moclobamide Morphine Sulphate Oral solution Lithium citrate 509mg/5ml 70mg/5ml 1mg/5ml 5mg/5ml 100mg/5ml Liquid 50mg/5ml 50mg/5ml 500mg/5ml Oral soln. 5mg/5ml 200mg/5ml Oral solution 10mg/5ml 100mg/5ml Injection may be given orally. Contact pharmacy for advice. A special suspension may be available from Boots specials unit. Modified release tablets should not be crushed. Lithium carbonate 200mg is broadly equivalent to lithium citrate 509mg. Plasma levels should be monitored when changing between preparations. Adjustment of dose and frequency may be necessary. Tablets slowly disperse in water Sugar Free Does not contain sorbitol. Should not be diluted as this may reduce efficacy. Crush and disperse in water. Sublingual tablet may be available. Please ask pharmacy for advice. Crush tablets and mix with water. Consider alternative benzodiazepine. See Co-beneldopa 15ml mebeverine pamoate 50mg/5ml is broadly equivalent to 135mg mebeverine hydrochloride. Provera tablets may be crushed and dispersed in water. Crush and disperse tablets in water. Crush and disperse in water. is unlicensed. may be available. Contact pharmacy for advice. Consider alternative anti-hypertensive. Suppositories available or change to alternative product eg sulphasalazine. Crush and disperse in water. Increased bioavailibility may cause postural hypotension. Monitor patient carefully. Disperses very slowly in water. Consider change to atenolol which is available as a liquid. Requires activation by stomach acid. Do not administer via nasojejunal or jejunostomy tube. Presence of enteral feed may interfere with activation. Use tablets dispersed in water instead or suppositories or administer 1 hour before or 2 hours after feed. Crush tablet and disperse in water. Liquid preparation may be available. Contact pharmacy for advice. Tablets may be crushed and mixed with water. Tablets may be crushed and mixed with water. Morphine sulphate controlled release products must never be crushed for administration via feeding tube as this destroys the modified release mechanism resulting in highly dangerous peak and trough levels. The granules from contolled release morhine capsules can be administered via PEG tube but extreme care must be taken to avoid inadvertent crushing or damage to the coating on the granules. For this reason more frequent administration of non controlled release morphine (Oramorph) is the preferred option. Administration of the granules from Zomorph capsules is licensed providing the tube has a diameter of more than 16 french gauge. If morphine is given via NJ or jejunostomy tube dose adjustment may be necessary because of reduced transit time and extent of absorption. 19
20 Nabilone Nicardipine Naproxen Nicorandil Nifedipine Nimodipine Nitrazepam Nitrofurantoin Norethisterone Omeprazole Olanzapine Olsalazine Ondansetron Orphenadrine Oxybutynin Oxycodone Oxytetracycline Pancreatic enzymes Paracetamol Paroxetine Penicillin V Pergolide Perindopril Phenelzine Phenobarbitone 125mg/5ml Tablets 30mg 2.5mg/5ml MUPS tablets 10mg, 20mg 4mg/5ml Oral Soln 50mg/5ml Elixir 2.5mg/5ml Dispersible tablets Suppositories All various strengths Liquid 10mg/5ml Oral solution 125mg/5ml 250mg/5ml Elixir 15mg/5ml Open capsule and immediately flush contents with water. Liquid suspension may be available. Contact pharmacy for advice. Contains 1.7mmol sodium in 5ml Crush tablet and disperse in water. Give immediately. Controlled release Coracten capsules may be opened and the contents given. Do not crush the granules. Granules may block fine bore tubes. Change to a long acting calcium antagonist eg amlodipine. Give liquid contents of nifedipine capsules using 1ml syringe and needle. Pull back plunger while squeezing capsule. May need more than one capsule to obtain required dose. 5mg = 0.17ml, 10mg = 0.34ml. Monitor for side effects. Eg Hypotension. Flush liquid down tube using saline not water. Must be given as a tds dosing schedule. This is extremely light sensitive and must be given immediately. Crush tablets and administer immediately or give IV. IV infusion is not suitable for oral administration as it contains ethanol and polyethylene glycol. Open and disperse Macrodantin capsules. Disperses slowly in water. Place tablet in syringe. Fill with 10ml water and 5ml air. Shake for 2 minutes to disperse tablet. Granules may block tubes smaller than 7F. For alternative management options please contact pharmacy for advice. MUPS tablets must not be crushed. Use Zyprexia Velotabs Open capsule disperse contents in water. Melt tablets may also be available. Suppositories may also be available. Warning: Do not crush controlled release formulations. Contact pharmacy for advice. Use alternative tetracycline if possible eg. doxycycline dispersible tabs. Capsules of tetracycline may be available. Contact pharmacy for advice. Seek advice from pharmacy. Soluble tablets are preferable to suspension. is hyperosmolar and may cause diarrhoea when administered via the NJ or J routes. Soluble tabs contain 388mg sodium per tablet. Suppositories are an alternative management option. Powder for reconstitution with water. Disperse tablet in water. May take up to 15 minutes for disintegration. Crush and disperse tablet in water. Crush and disperse in water. Contains alcohol 38% Special alcohol free suspension may be available. Please ask pharmacy for advice. 20
21 Phenytoin Phytomenandione Piroxicam Pizotifen Pramipexole Pravastatin Prazosin Prednisolone Prochlorperazine Procyclidine Proguanil Primidone Promazine Promethazine Propafenone Propylthiouracil Propranolol Pseudoephedrine Pyrazinamide Pyridoxine Pyridostigmine Quetiapine Quinine Sulphate Ramapril Ranitidine Reboxetine Rifampicin Riluzole Rivastigmine Risperidone Rofecoxib Salbutalmol Selegiline Senna Sertraline Sinemet Simvasatin Sodium feredetate (ironedetate) 30mg/5ml 90mg/5ml Elixir 250 mcg/5ml 5mg/5ml 2.5mg/5ml 250mg/5ml 50mg/5ml Elixir 5mg/5ml Elixir 30mg/5ml 150mg/10ml Effervescent tabs 150mg 100mg/5ml Liquid 1mg/ml 2mg/5ml 7.5mg/5ml Elixir 190mg/5ml Phenytoin suspension and capsules are not equivalent. 90mg suspension is approximately equivalent to 100mg tablets or capsules. Diluting suspension decreases its viscosity and aids administration. is hyper-osmolar and may cause diarrhoa when administered via the NJ and J routes. Phenytoin interacts with feeds. See above. Konakion MM injection may be given orally. Consider alternative NSAID available as a liquid. Crush and disperse in water. Crush and disperse in water. Crush tablets and mix with water. Use soluble tabs Suppositories also available. Crush and disperse in water. May not be available. Contact pharmacy for advice. Crush tablets and administer orally in 5% glucose. Crush and disperse tablet in water. Liquid may be available from pharmacy. Do not crush modified release preparations. Contact pharmacy for advice on conversion to liquid formulation. Consider alternative beta blocker. Crush tablets and administer with water. Stop feed half an hour before and after administration. Crush tablets and flush with water. Disperses slowly in water. Crush and disperse in water. Crush and disperse in lager volume of water. Sugar coating should dissolve. Open capsules and flush contents with water.. Ranitidine syrup contains sorbitol and may cause diarrhoea. Crush and disperse tablet in water. Stop feed 30 minutes before and after administration. Crush and disperse in water. Open Exelon capsules and disperse contents in water. May be diluted with mineral water, orange juice or black coffee. Liquid formulation may be available. Contact pharmacy for advice. Do not crush modified release tablets. If changing from modified release to liquid dosage frequency will need to be increased. Liquid or oral lyophilisates may be available. Contact pharmacy for advice. Crush and disperse in water. See Co-careldopa Crush tablets and administer in water immediately. Sugar Free Equivalent to 27.5mg iron/5ml 21
22 Sodium fusidate Sodium clodronate Sodium picosulphate Sodium valproate Sotalol Spironolactone Sucralfate Sulfasalazine Sulpiride Tacrolimus Tamoxifen Tamsulosin Temazepam Tetrabenazine Tetracycline Theophylline Thiamine Thioridazine Elixir 5mg/5ml Oral liquid 200mg/5ml Oral suspension 25mg/5ml Dispersible Tablets 1g/5ml 250mg/5ml Oral soln 200mg/5ml Oral Soln 10mg/5ml Oral Soln 10mg/5ml Nuelin 60mg/5ml 25mg/5ml See Fusidic acid above Open capsules and administer with water. Stop feeds for 2 hours before and after administration.. Contains sorbitol. If patient on Epilim chrono will need to adjust dosage frequency. Ask pharmacy for advice. Crush tablets and administer with water. Interacts with enteral feeds. Forms insoluble complexes with protein in enteral feeds. May block tubes or cause intestinal obstruction (bezoar formation). Avoid combined administration with enteral feed. Use alternative agent. Contact pharmacy for advice. Open capsule and administer granules with water. Do not crush the granules. Crush tablets and disperse in water. Consider alternative antibiotic. If tetracycline in indicated doxycycline dispersible tablets are available. Increase in dosage frequency is required if transferring from a SR formulation to a short acting formulation. Sustained release theophylline tablets must not be crushed. Slophylline capsules may be opened and the granules administered without crushing (granules may block the tube). Nuelin tablets are dispersible in water and may be used as an alternative to Nuelin liquid. Theophylline doses may need to be adjusted when changing between preparations. Monitor theophylline levels. Theophylline interacts with feeds. See above. Crush tablets and administer with water. Thyroxine Disperses in water after crushing. Tiagabine Crush and disperse in water. Tizanidine Crush and disperse in water. Tolbutamide Crush and disperse in water. Monitor blood glucose. Consider alternative sulphonylurea which can be more easily crushed. Tolterodine Crush and disperse in water. Do not crush Detrusitol XL Topiramate Crush tablets and administer with water. Tramadol Open capsule and administer contents with water. Tranexamic acid Injection may be given orally after dilution, or suspension made by pharmacy. Contact pharmacy for advice Trazodone 50mg/5ml. Trifluorperazine Liquid formulation may be available. Contact pharmacy for advice. Trimeprazine 7.5mg/5ml 30mg/5ml Does not contain sorbitol. Trimethoprim Ursodeoxycholic acid Valsartan 50mg/5ml Contains sorbitol. Ursofalk suspension may be available. Contact pharmacy for advice. Open capsules and give contents in water immediately as it is not very stable. 22
23 Vancomycin Venlafaxine Verapamil Vigabatrin Vitamin B Preparations Vitamin E (Ephynal) Warfarin Zolpidem Zuclopenthixol Oral soln 40mg/5ml Sachet 500mg 500mg/5ml Disperses in water. Use injection orally for administration via enteral tube. NB for treatment of antibiotic associated colitis only. Oral vancomycin is not suitable for the treatment of systemic infections since it is not significantly absorbed. Efexor tablets can be crushed and dispersed in water. Efexor XL capsules can be opened and the contents administered with water. Do not crush the granules.. If patient taking modified release verapamil. contact pharmacy for advice on dose and dosage frequency adjustment. Contact pharmacy for advice. Effects of warfarin can be reduced by vitamin K in enteral feeds. Monitor INR and adjust dose as necessary. Crush and disperse tablet in water. Clopixol tablets may be crushed and dispersed in water. Consider depot injection. 23
24 Bibliography O Hare M, Fair R, Administering Drugs Through Enteral Feeding Tubes The Royal Hospitals Belfast Adams D Administration of Drugs through a jejunostomy tube. An overview of compatibility and efficacy B J Int Care 1994;4: Cerrulli J, Malone M, Assessment of drug related problems in clinical nutrition patients J Parent Ent Nutr 1999;23: E.K.Kelly, Hannawa T Techniques for administering oral medications to critical care patients receiving continuous enteral nutrition Am J Health-Syst Pharm 1999;56: A-Z guide to administration of drugs via nasogastric/peg Tube Matthews E, Stockton L, Thomson F Thomson FC, Naysmith MR, Lindsay A, Managing drug therapy in patients receiving enteral and parenteral nutrition Hospital Pharmacist 2000;7: Reeves V J, Hopkins S, The administration of medication via a percutaneous endoscopic gastrostomy tube Broomfield Hospital, Mid Essex Hospitals Services NHS Trust 24
Medicines & Enteral Feeding Tubes A DELA V IDICKI-MASTILOVICH C LINICAL P HARMACIST R OYAL C HILDREN S H OSPITAL
 Medicines & Enteral Feeding Tubes A DELA V IDICKI-MASTILOVICH C LINICAL P HARMACIST R OYAL C HILDREN S H OSPITAL Introduction Types of Tubes Choosing a formulation Crushing tablets/opening capsules Administration
Medicines & Enteral Feeding Tubes A DELA V IDICKI-MASTILOVICH C LINICAL P HARMACIST R OYAL C HILDREN S H OSPITAL Introduction Types of Tubes Choosing a formulation Crushing tablets/opening capsules Administration
PROJECT LIST GENERIC PRODUCTS
 PROJECT LIST GENERIC PRODUCTS Acetylcysteine, Effervescent tablets 200 mg, 600 mg Alendronate sodium, Tablets 10, 70 mg Alfuzosin,Tablets 2.5mg Alfuzosin, ER Tablets 10 mg Ambroxol, Effervescent tablets
PROJECT LIST GENERIC PRODUCTS Acetylcysteine, Effervescent tablets 200 mg, 600 mg Alendronate sodium, Tablets 10, 70 mg Alfuzosin,Tablets 2.5mg Alfuzosin, ER Tablets 10 mg Ambroxol, Effervescent tablets
$10.00 PRESCRIPTION PROGRAM DETAILS
 $10.00 PRESCRIPTION PROGRAM DETAILS 1. The $10.00 program applies only to certain generic drugs at commonly prescribed 90 day usage dosages. (See list). 2. The Program may change without notification and
$10.00 PRESCRIPTION PROGRAM DETAILS 1. The $10.00 program applies only to certain generic drugs at commonly prescribed 90 day usage dosages. (See list). 2. The Program may change without notification and
$4, 30-day $10, 90-day
 $4 Prescriptions - Choose from hundreds of generic drugs and over the counter medications. Free Home Delivery Mailed right to your home Free shipping Prescription Program includes up to a 30-day supply
$4 Prescriptions - Choose from hundreds of generic drugs and over the counter medications. Free Home Delivery Mailed right to your home Free shipping Prescription Program includes up to a 30-day supply
Appendix 10: Administering drugs via feeding tubes
 Appendix 10: Administering drugs via feeding tubes Administering drugs via feeding tubes is generally an unlicensed activity. There is little published data and most recommendations are theoretical and/or
Appendix 10: Administering drugs via feeding tubes Administering drugs via feeding tubes is generally an unlicensed activity. There is little published data and most recommendations are theoretical and/or
GUIDANCE ON THE ADMINISTRATION OF DRUGS VIA ENTERAL FEEDING TUBES V 3.0
 GUIDANCE ON THE ADMINISTRATION OF DRUGS VIA ENTERAL FEEDING TUBES V 3.0 1 st June 2013 Table of Contents Table of Contents... 2 1. Introduction... 3 2. Purpose of this Policy/Procedure... 3 3. Scope...
GUIDANCE ON THE ADMINISTRATION OF DRUGS VIA ENTERAL FEEDING TUBES V 3.0 1 st June 2013 Table of Contents Table of Contents... 2 1. Introduction... 3 2. Purpose of this Policy/Procedure... 3 3. Scope...
PREFERRED GENERIC DRUG LIST
 These discount programs are NOT health insurance policies and are not intended as a substitute for insurance. The programs do not qualify as a minimum creditable coverage under Massachusetts law or where
These discount programs are NOT health insurance policies and are not intended as a substitute for insurance. The programs do not qualify as a minimum creditable coverage under Massachusetts law or where
Each un coated tablet contains: Nimesulide. Each un coated tablet contains: Nimesulide Paracetamol
 Sr No. Generic Name Composition Claim 1 Tabs. 100/ 2 + Para Tabs. 3 + Tizanidine Tabs. 4 + Tabs. 5 +Serratio Tabs. 6 +Serratio Tabs. 7 Aceclo Tabs. IP 8 Aceclo + Para Tabs. 9 Aceclo + Para + Chlorzoxazone
Sr No. Generic Name Composition Claim 1 Tabs. 100/ 2 + Para Tabs. 3 + Tizanidine Tabs. 4 + Tabs. 5 +Serratio Tabs. 6 +Serratio Tabs. 7 Aceclo Tabs. IP 8 Aceclo + Para Tabs. 9 Aceclo + Para + Chlorzoxazone
Retail Prescription Program Drug List
 Retail Prescription Program Drug List Price Matters New Men s Health Category Convenience Free Home Delivery Our 4 prescriptions have saved our customers over 3 billion The program is available to everyone,
Retail Prescription Program Drug List Price Matters New Men s Health Category Convenience Free Home Delivery Our 4 prescriptions have saved our customers over 3 billion The program is available to everyone,
612 Program Midtown Express Pharmacy
 ALENDRONATE SOD TAB 35MG (max 1 per week) $37.00 $70.00 ALENDRONATE SOD TAB 70MG (max 1 per week) $37.00 $70.00 ALLOPURINOL TAB 100MG $20.00 $38.00 ALLOPURINOL TAB 300MG $20.00 $38.00 AMITRIPTYLINE TAB
ALENDRONATE SOD TAB 35MG (max 1 per week) $37.00 $70.00 ALENDRONATE SOD TAB 70MG (max 1 per week) $37.00 $70.00 ALLOPURINOL TAB 100MG $20.00 $38.00 ALLOPURINOL TAB 300MG $20.00 $38.00 AMITRIPTYLINE TAB
All Wales Prescription Writing Standards
 All Wales Prescription Writing Standards These standards should be read in conjunction with completing the All Wales Medication Chart e- learning package, available on the Learning@NHSWales internet site
All Wales Prescription Writing Standards These standards should be read in conjunction with completing the All Wales Medication Chart e- learning package, available on the Learning@NHSWales internet site
The 365-day period begins with the first dispensing transaction for each Ontario Drug Benefit (ODB) recipient on or after October 1, 2015.
 Ontario Public Drug Programs, Ministry of Health and Long-Term Care Chronic-use Medications List by In accordance with subsection 18 (11.1) of Ontario Regulation 201/96 made under the Ontario Drug Benefit
Ontario Public Drug Programs, Ministry of Health and Long-Term Care Chronic-use Medications List by In accordance with subsection 18 (11.1) of Ontario Regulation 201/96 made under the Ontario Drug Benefit
Home Delivery Prescription Program Drug List
 Home Delivery Prescription Program Drug List Low-cost prescriptions, right in your mailbox. Now you can have your generic prescriptions mailed right to your home, no matter where you live. Because we think
Home Delivery Prescription Program Drug List Low-cost prescriptions, right in your mailbox. Now you can have your generic prescriptions mailed right to your home, no matter where you live. Because we think
Directory of Generic Medications Eligible for Rx Savings Program Flat Fees
 Directory of Generic Medications Eligible for Rx Savings Program Flat Fees CONNECTICUT VERSION If you re already enrolled in the FREE* Rx Savings Program, use this guide to find your best choices. And,
Directory of Generic Medications Eligible for Rx Savings Program Flat Fees CONNECTICUT VERSION If you re already enrolled in the FREE* Rx Savings Program, use this guide to find your best choices. And,
1 Manipulation of formulae and dilutions
 1 Manipulation of formulae and dilutions This first chapter of questions is specifically designed to cover a range of numeracy skills. It will give the user further experience in the skills of addition,
1 Manipulation of formulae and dilutions This first chapter of questions is specifically designed to cover a range of numeracy skills. It will give the user further experience in the skills of addition,
Generic Pharmacy Discount
 Generic Pharmacy Discount 83 Park Place Blvd Suite 101 Clearwater, FL 33759 Fourth Quarter 2014 727.461.6044 800.314.0088 Pharmacies Generic Discount Program Information Enclosed information provided
Generic Pharmacy Discount 83 Park Place Blvd Suite 101 Clearwater, FL 33759 Fourth Quarter 2014 727.461.6044 800.314.0088 Pharmacies Generic Discount Program Information Enclosed information provided
. ก ก 1. Actifed tab/syr. Triprolidine : should be stored in tight, light-resistant Pseudoepedrine : Triprolidine (p.25) Pseudoepedrine (p.
 ก ก. 2553 (p.807) 3. Adrenaline inj. 1mg/ml (Epinephrine inj.) ( Druginfor. p.629) 4. Aminophylline/ Theophylline inj. (Drug infor. Ed. 11 th p.1339) 5. Amlodipine + HCTZ (Moduretic). ก ก 1. Actifed tab/syr.
ก ก. 2553 (p.807) 3. Adrenaline inj. 1mg/ml (Epinephrine inj.) ( Druginfor. p.629) 4. Aminophylline/ Theophylline inj. (Drug infor. Ed. 11 th p.1339) 5. Amlodipine + HCTZ (Moduretic). ก ก 1. Actifed tab/syr.
Common Medication for People Receiving Haemodialysis
 Common Medication for People Receiving Haemodialysis Delivering the best in care UHB is a no smoking Trust To see all of our current patient information leaflets please visit www.uhb.nhs.uk/patient-information-leaflets.htm
Common Medication for People Receiving Haemodialysis Delivering the best in care UHB is a no smoking Trust To see all of our current patient information leaflets please visit www.uhb.nhs.uk/patient-information-leaflets.htm
Information and Guidance on the Prescribing and Use of Unlicensed Pharmaceutical Specials
 Information and Guidance on the Prescribing and Use of Unlicensed Pharmaceutical Specials Contents Introduction 3 Scope 3 What is a pharmaceutical special? 3 The clinical and legal differences 5 Processes
Information and Guidance on the Prescribing and Use of Unlicensed Pharmaceutical Specials Contents Introduction 3 Scope 3 What is a pharmaceutical special? 3 The clinical and legal differences 5 Processes
Doncaster & Bassetlaw Medicines Formulary
 Doncaster & Bassetlaw Medicines Formulary Section 4.9: Drugs Used in Parkinsonism and related Disorders Co-Beneldopa 12.5/50, 25/100 and 50/200 (Madopar) Capsules Co-Beneldopa 12.5/50 and 25/100 Dispersible
Doncaster & Bassetlaw Medicines Formulary Section 4.9: Drugs Used in Parkinsonism and related Disorders Co-Beneldopa 12.5/50, 25/100 and 50/200 (Madopar) Capsules Co-Beneldopa 12.5/50 and 25/100 Dispersible
Bariatric surgery patients and their medicines
 Bulletin 54 April 2014 2.0 Bariatric surgery patients and their medicines There has been a steady increase in bariatric surgery 1 since the National Institute of Health and Care Excellence (NICE) published
Bulletin 54 April 2014 2.0 Bariatric surgery patients and their medicines There has been a steady increase in bariatric surgery 1 since the National Institute of Health and Care Excellence (NICE) published
Berkshire Healthcare NHS Foundation Trust Becky White CHS Pharmacist April 2013
 Berkshire Healthcare NHS Foundation Trust Becky White CHS Pharmacist April 2013 Access to palliative care drugs out of hours Agreement set up with local community pharmacy s to hold stock of commonly prescribed
Berkshire Healthcare NHS Foundation Trust Becky White CHS Pharmacist April 2013 Access to palliative care drugs out of hours Agreement set up with local community pharmacy s to hold stock of commonly prescribed
Community Pharmacists in NHS Rotherham
 SERVICE LEVEL AGREEMENT TO ENABLE COMMUNITY PHARMACISTS IN NHS ROTHERHAM TO PROVIDE PALLIATIVE CARE DRUGS AS LOCAL ENHANCED SERVICE PREPARED BY: NHS Rotherham CCG Medicines Management Team on behalf of
SERVICE LEVEL AGREEMENT TO ENABLE COMMUNITY PHARMACISTS IN NHS ROTHERHAM TO PROVIDE PALLIATIVE CARE DRUGS AS LOCAL ENHANCED SERVICE PREPARED BY: NHS Rotherham CCG Medicines Management Team on behalf of
Palliative Care Drug Plan (Plan P) Formulary List of drugs PharmaCare covers
 Palliative Care Drug Plan (Plan P) Formulary List of drugs PharmaCare covers Important Notes: Last Updated: May 11, 2015 Pharmacists must submit a claim on PharmaNet at the time of purchase to enable coverage.
Palliative Care Drug Plan (Plan P) Formulary List of drugs PharmaCare covers Important Notes: Last Updated: May 11, 2015 Pharmacists must submit a claim on PharmaNet at the time of purchase to enable coverage.
Drugs to consider prescribing by brand name or where brands should not be switched
 Index Page Index Page Adrenaline (epinephrine) prefilled syringes 2 Hormone replacement therapy oral 3 Alprostadil injection 4 Human papillomavirus vaccine 5 Aminophylline modified release 2 Insulins 3
Index Page Index Page Adrenaline (epinephrine) prefilled syringes 2 Hormone replacement therapy oral 3 Alprostadil injection 4 Human papillomavirus vaccine 5 Aminophylline modified release 2 Insulins 3
Pharmaceutical Issues when Crushing, Opening or Splitting Oral Dosage Forms June 2011
 Pharmaceutical Issues when Crushing, Opening or Splitting Oral Dosage Forms June 2011 Introduction It is important to recognise the potential consequences of manipulating a medicinal product. Changing
Pharmaceutical Issues when Crushing, Opening or Splitting Oral Dosage Forms June 2011 Introduction It is important to recognise the potential consequences of manipulating a medicinal product. Changing
Acute management of Parkinson s
 Acute management of Parkinson s Fife Parkinson s Service 2013 1 Contents 1. Introduction 2. On admission to hospital 3. If Patient has compromised swallow or is nil by mouth 4. Conversion charts if patient
Acute management of Parkinson s Fife Parkinson s Service 2013 1 Contents 1. Introduction 2. On admission to hospital 3. If Patient has compromised swallow or is nil by mouth 4. Conversion charts if patient
Measuring Generic Efficiency in Part D
 Measuring in Part D Rates among Medicare Part D Generic efficiency rate is a measurement of the total number of prescrip=ons filled as a generic for products with a direct generic subs=tute within a therapeu=c
Measuring in Part D Rates among Medicare Part D Generic efficiency rate is a measurement of the total number of prescrip=ons filled as a generic for products with a direct generic subs=tute within a therapeu=c
Basic Medication Administration Exam RN (BMAE-RN) Study Guide
 Basic Medication Administration Exam RN (BMAE-RN) Study Guide Review correct procedure and precautions for the following routes of administration: Ear drops Enteral feeding tube Eye drops IM, subcut injections
Basic Medication Administration Exam RN (BMAE-RN) Study Guide Review correct procedure and precautions for the following routes of administration: Ear drops Enteral feeding tube Eye drops IM, subcut injections
Always take this medicine exactly as described in this leaflet or as your doctor, pharmacist or nurse have told you.
 leaflet: Information for the user Macrogol 4000 10 g powder for oral solution in sachet Macrogol 4000
leaflet: Information for the user Macrogol 4000 10 g powder for oral solution in sachet Macrogol 4000
DRUG CALCULATIONS. Mathematical accuracy is a matter of life and death. [Keighley 1984]
![DRUG CALCULATIONS. Mathematical accuracy is a matter of life and death. [Keighley 1984] DRUG CALCULATIONS. Mathematical accuracy is a matter of life and death. [Keighley 1984]](/thumbs/39/19297070.jpg) DRUG CALCULATIONS Mathematical accuracy is a matter of life and death. [Keighley 1984] At the end of this practice sheet, you will be able to: Perform simple arithmetical tasks. Accurately calculate drug
DRUG CALCULATIONS Mathematical accuracy is a matter of life and death. [Keighley 1984] At the end of this practice sheet, you will be able to: Perform simple arithmetical tasks. Accurately calculate drug
NEW SCHEDULE H1 INSERTED IN DRUGS AND COSMETICS RULES :
 LOCOST 85, GIDC Estate, Por-Ramangamdi, N.H.8, Dist. Vadodara - 391 243 Gujarat, India. Tel. No.: (91-265) 2830009 Fax No.: (P.P.) 2831446 E-mail: [email protected] Website: www.locostindia.com A Public
LOCOST 85, GIDC Estate, Por-Ramangamdi, N.H.8, Dist. Vadodara - 391 243 Gujarat, India. Tel. No.: (91-265) 2830009 Fax No.: (P.P.) 2831446 E-mail: [email protected] Website: www.locostindia.com A Public
Chapter 4: Central nervous system
 Page 1 of 15 Menu Pharmacy Home Page Chapter 4: Central nervous system POPAM The GHT Formulary applies to the treatment of adults only Policies & Forms To search the chapter click 'Ctrl' + 'F' and type
Page 1 of 15 Menu Pharmacy Home Page Chapter 4: Central nervous system POPAM The GHT Formulary applies to the treatment of adults only Policies & Forms To search the chapter click 'Ctrl' + 'F' and type
PREPARATIONS: Adrenaline 1mg in 1ml (1:1000) Adrenaline 100micrograms in 1ml (1:10,000)
 ADRENALINE Acute hypotension Via a CENTRAL venous line Initially 100-300 nanograms/kg/minute 0.1-0.3 microgram/kg/minute adjusted according to response up to a maximum of 1.5 micrograms/kg/minute. Increase
ADRENALINE Acute hypotension Via a CENTRAL venous line Initially 100-300 nanograms/kg/minute 0.1-0.3 microgram/kg/minute adjusted according to response up to a maximum of 1.5 micrograms/kg/minute. Increase
Anti Protozoal. Albendazole Oral Suspension USP Albendazole Tablets USP 400 mg Anti Malarial
 ANTI-INFECTIVES Anti Biotics Levofloxacin Tablets 250, 500, 750 mg Ofloxacin Tablets 200, 400 mg Sparfloxacin Tablets 200 mg Ciprofloxacin Tablets USP 250, 500 mg Ofloxacin & Ornidazole Tablets Nitrofurantoin
ANTI-INFECTIVES Anti Biotics Levofloxacin Tablets 250, 500, 750 mg Ofloxacin Tablets 200, 400 mg Sparfloxacin Tablets 200 mg Ciprofloxacin Tablets USP 250, 500 mg Ofloxacin & Ornidazole Tablets Nitrofurantoin
Nurse Initiated Medications Procedure
 1. Purpose This Procedure is performed as a means of ensuring the safe administration of therapeutic medication to patients in accordance with all legislative and regulatory requirements. 2. Application
1. Purpose This Procedure is performed as a means of ensuring the safe administration of therapeutic medication to patients in accordance with all legislative and regulatory requirements. 2. Application
A Guide to pain relief medicines For patients receiving Palliative Care
 A Guide to pain relief medicines For patients receiving Palliative Care 1 Which pain medicines are you taking? Contents Page No. Amitriptyline 8 Codeine 9 Co-codamol 10 Co-dydramol 11 Diclofenac (Voltarol
A Guide to pain relief medicines For patients receiving Palliative Care 1 Which pain medicines are you taking? Contents Page No. Amitriptyline 8 Codeine 9 Co-codamol 10 Co-dydramol 11 Diclofenac (Voltarol
Service Specification for NHS Community Pharmacy Palliative Care Drugs Stockist Scheme
 Service Specification for NHS Community Pharmacy Palliative Care Drugs Stockist Scheme 1. This agreement is between NHS England North Midlands (Derbyshire/ Nottinghamshire only) (the Commissioner) Birch
Service Specification for NHS Community Pharmacy Palliative Care Drugs Stockist Scheme 1. This agreement is between NHS England North Midlands (Derbyshire/ Nottinghamshire only) (the Commissioner) Birch
Order No. Date Surgery DRUG ORDER SHEET. Page 1. Quantity Unit Total Ordered Cost Cost. Order No. Date
 Order No. Surgery Page 1 Aciclovir 800mg (35) Tabs Aciclovir 400mg (56) Tabs Adrenaline 1:10000 Aerospacer adult Aerospacer child/baby with mask Amitriptiline 10mg Amoxicillin 125/5 syrup 100 mls Amoxicillin
Order No. Surgery Page 1 Aciclovir 800mg (35) Tabs Aciclovir 400mg (56) Tabs Adrenaline 1:10000 Aerospacer adult Aerospacer child/baby with mask Amitriptiline 10mg Amoxicillin 125/5 syrup 100 mls Amoxicillin
Nursing 113. Pharmacology Principles
 Nursing 113 Pharmacology Principles 1. The study of how drugs enter the body, reach the site of action, and are removed from the body is called a. pharmacotherapeutics b. pharmacology c. pharmacodynamics
Nursing 113 Pharmacology Principles 1. The study of how drugs enter the body, reach the site of action, and are removed from the body is called a. pharmacotherapeutics b. pharmacology c. pharmacodynamics
Pharmacological Management of Dementia
 Pharmacological Management of Dementia Christopher Sullivan Lead Clinical Pharmacist South and West Devon Devon Partnership Trust Overview Management of cognitive symptoms Management of non-cognitive symptoms
Pharmacological Management of Dementia Christopher Sullivan Lead Clinical Pharmacist South and West Devon Devon Partnership Trust Overview Management of cognitive symptoms Management of non-cognitive symptoms
CICLOSPORIN. What are the aims of this leaflet?
 CICLOSPORIN What are the aims of this leaflet? This leaflet has been written to help you understand more about ciclosporin. It tells you what it is, how it works, how it is used to treat skin conditions,
CICLOSPORIN What are the aims of this leaflet? This leaflet has been written to help you understand more about ciclosporin. It tells you what it is, how it works, how it is used to treat skin conditions,
Use of the Injectable Medicines Guide website in clinical areas - IntraVENOUS medicine monographs (January 2013)
 Use of the Injectable Medicines Guide website in clinical areas - IntraVENOUS medicine monographs (January 2013) Introduction The IntraVENOUS medicines section of the Injectable Medicines Guide website
Use of the Injectable Medicines Guide website in clinical areas - IntraVENOUS medicine monographs (January 2013) Introduction The IntraVENOUS medicines section of the Injectable Medicines Guide website
Acetylcholinesterase Inhibitors and Memantine Clinical Indication: Treatment of Dementia in Alzheimer s Disease (AD)
 SHARED CARE PROTOCOL AND INFORMATION FOR GPS Acetylcholinesterase Inhibitors and Memantine Clinical Indication: Treatment of Dementia in Alzheimer s Disease (AD) Version: 3 Date Approved: June 2011 Review
SHARED CARE PROTOCOL AND INFORMATION FOR GPS Acetylcholinesterase Inhibitors and Memantine Clinical Indication: Treatment of Dementia in Alzheimer s Disease (AD) Version: 3 Date Approved: June 2011 Review
PPP 1. Continuation of a medication to ensure continuity of care
 PRESCRIBING POLICIES: 4.7 PHARMACIST AUTHORITY The College of Pharmacists of BC Professional Practice Policy (PPP) 58 Medication Management (Adapting a Prescription) became effective April 1, 2009. The
PRESCRIBING POLICIES: 4.7 PHARMACIST AUTHORITY The College of Pharmacists of BC Professional Practice Policy (PPP) 58 Medication Management (Adapting a Prescription) became effective April 1, 2009. The
Clinical Practice Guidelines: Nurse Practitioner, Palliative Care Service
 Clinical Practice Guidelines: Nurse Practitioner, Palliative Care Service Purpose of Role The Nurse Practitioner (NP) will provide comprehensive palliative care management and advanced nursing care in
Clinical Practice Guidelines: Nurse Practitioner, Palliative Care Service Purpose of Role The Nurse Practitioner (NP) will provide comprehensive palliative care management and advanced nursing care in
Basic Medication Administration Exam LPN/LVN (BMAE-LPN/LVN) Study Guide
 Basic Medication Administration Exam LPN/LVN (BMAE-LPN/LVN) Study Guide Review correct procedure and precautions for the following routes of administration: Ear drops Enteral feeding tube Eye drops IM,
Basic Medication Administration Exam LPN/LVN (BMAE-LPN/LVN) Study Guide Review correct procedure and precautions for the following routes of administration: Ear drops Enteral feeding tube Eye drops IM,
BC Cancer Agency & Canadian Cancer Society Financial Support Drug Program (FSDP) for Cancer Patients. Drug Benefit List. Updated February 15, 2016
 BC Cancer Agency & Canadian Cancer Society Financial Support Drug Program (FSDP) for Cancer Patients Drug Benefit List Updated February 15, 2016 The FSDP will operate following rules established by the
BC Cancer Agency & Canadian Cancer Society Financial Support Drug Program (FSDP) for Cancer Patients Drug Benefit List Updated February 15, 2016 The FSDP will operate following rules established by the
MEDICINES MANAGEMENT STANDARD OPERATING PROCEDURE (MMSOP018) Preparation of Medication Administration Record (MAR) Charts
 MEDICINES MANAGEMENT STANDARD OPERATING PROCEDURE (MMSOP018) Preparation of Medication Administration Record (MAR) Charts Any deviation in practice from this procedure must be discussed with the Community
MEDICINES MANAGEMENT STANDARD OPERATING PROCEDURE (MMSOP018) Preparation of Medication Administration Record (MAR) Charts Any deviation in practice from this procedure must be discussed with the Community
GUIDELINES ON THE MANAGEMENT OF PAIN DUE TO CANCER IN ADULTS
 GUIDELINES ON THE MANAGEMENT OF PAIN DUE TO CANCER IN ADULTS Bristol Palliative Care Collaborative Contact Numbers: Hospital Specialist Palliative Care Teams: Frenchay 0117 340 6692 Southmead 0117 323
GUIDELINES ON THE MANAGEMENT OF PAIN DUE TO CANCER IN ADULTS Bristol Palliative Care Collaborative Contact Numbers: Hospital Specialist Palliative Care Teams: Frenchay 0117 340 6692 Southmead 0117 323
PHOSPHATE-SANDOZ Tablets (High dose phosphate supplement)
 1 PHOSPHATE-SANDOZ Tablets (High dose phosphate supplement) PHOSPHATE-SANDOZ PHOSPHATE-SANDOZ Tablets are a high dose phosphate supplement containing sodium phosphate monobasic. The CAS registry number
1 PHOSPHATE-SANDOZ Tablets (High dose phosphate supplement) PHOSPHATE-SANDOZ PHOSPHATE-SANDOZ Tablets are a high dose phosphate supplement containing sodium phosphate monobasic. The CAS registry number
PREFERRED GENERIC DRUG LIST
 ALLERGIES & COLD AND FLU Preferred generic drugs MARCH 2013 supply* for $5 supply* for $10 and $15 * The day supply is based upon the average dispensing patterns for the specific drug and strength. 90-
ALLERGIES & COLD AND FLU Preferred generic drugs MARCH 2013 supply* for $5 supply* for $10 and $15 * The day supply is based upon the average dispensing patterns for the specific drug and strength. 90-
NSW Higher School Certificate Senior Science 9.2 Lifestyle Chemistry
 NSW Higher School Certificate Senior Science 9.2 Lifestyle Chemistry Section 5 Drug Solubility 9.2 Lifestyle Chemistry Section 5 ::: Drug Solubility 9.2.5 The solubility of drugs has an effect on the way
NSW Higher School Certificate Senior Science 9.2 Lifestyle Chemistry Section 5 Drug Solubility 9.2 Lifestyle Chemistry Section 5 ::: Drug Solubility 9.2.5 The solubility of drugs has an effect on the way
END OF LIFE MEDICINES INFORMATION PACK
 END OF LIFE MEDICINES INFORMATION PACK Advice on end of life medication is available from the nursing and medical team at St Nicholas Hospice Care - telephone 01284 766133. Many drugs used in palliative
END OF LIFE MEDICINES INFORMATION PACK Advice on end of life medication is available from the nursing and medical team at St Nicholas Hospice Care - telephone 01284 766133. Many drugs used in palliative
Ward 29 guide to the safe preparation and administration of intravenous (IV) antibiotics at home
 Ward 29 guide to the safe preparation and administration of intravenous (IV) antibiotics at home Contents Page Important contact numbers 1 General information on preparing and administering IV antibiotics
Ward 29 guide to the safe preparation and administration of intravenous (IV) antibiotics at home Contents Page Important contact numbers 1 General information on preparing and administering IV antibiotics
Common medicines given to neurosurgery patients on discharge from hospital
 Common medicines given to neurosurgery patients on discharge from hospital This leaflet contains brief information about some of the medicines we commonly supply on discharge to patients who have been
Common medicines given to neurosurgery patients on discharge from hospital This leaflet contains brief information about some of the medicines we commonly supply on discharge to patients who have been
Notice from the Executive Officer: Supporting Sustainability and Access for the Ontario Drug Benefit Program
 Ontario Public Drug Programs, Ministry of Health and Long-Term Care Notice from the Executive Officer: Supporting Sustainability and Access for the Ontario Drug Benefit Program Responsible management of
Ontario Public Drug Programs, Ministry of Health and Long-Term Care Notice from the Executive Officer: Supporting Sustainability and Access for the Ontario Drug Benefit Program Responsible management of
Section II When you are finished with this section, you will be able to: Define medication (p 2) Describe how medications work (p 3)
 Section II When you are finished with this section, you will be able to: Define medication (p 2) Describe how medications work (p 3) List the different medication effects (p5) List the ways that medications
Section II When you are finished with this section, you will be able to: Define medication (p 2) Describe how medications work (p 3) List the different medication effects (p5) List the ways that medications
Excerpt from Food-Medication Interactions 13th edition by Zaneta M. Pronsky, MS, RD, LDN, FADA INTRODUCTION
 Excerpt from Food-Medication Interactions 13th edition by Zaneta M. Pronsky, MS, RD, LDN, FADA INTRODUCTION The study of the absorption, distribution, metabolism and excretion of drugs is referred to as
Excerpt from Food-Medication Interactions 13th edition by Zaneta M. Pronsky, MS, RD, LDN, FADA INTRODUCTION The study of the absorption, distribution, metabolism and excretion of drugs is referred to as
Members enjoy more Pharmacy savings *
 Sam s Plus Members enjoy more Pharmacy savings 5 prescription drugs available for FREE Generic medications: Donepezil, Pioglitazone, Escitalopram, Finasteride and Vitamin D2 50,000IU are $ 0 for a 30-day
Sam s Plus Members enjoy more Pharmacy savings 5 prescription drugs available for FREE Generic medications: Donepezil, Pioglitazone, Escitalopram, Finasteride and Vitamin D2 50,000IU are $ 0 for a 30-day
National Patient Safety Agency. Risk Assessment of Injectable Medicines. STEP 1 Local Risk Factor Assessment. STEP 2 Product Risk Factor Assessment
 NPSA Injectable Medicines Risk Assessment Tool National Patient Safety Agency Risk Assessment of Injectable Medicines STEP 1 Local Risk Factor Assessment. Carry out a baseline assessment in a near patient
NPSA Injectable Medicines Risk Assessment Tool National Patient Safety Agency Risk Assessment of Injectable Medicines STEP 1 Local Risk Factor Assessment. Carry out a baseline assessment in a near patient
Extra Value Drug List. *
 List. * Brand Diabetes Levemir 100 units/ml Vial 10mL $122.29 Levemir FlexPen 100 units/ml 15mL $203.03 NovoLog 100 units/ml Vial 10mL $116.84 NovoLog FlexPen Syringe 15mL $222.41 NovoLog 100 units/ml
List. * Brand Diabetes Levemir 100 units/ml Vial 10mL $122.29 Levemir FlexPen 100 units/ml 15mL $203.03 NovoLog 100 units/ml Vial 10mL $116.84 NovoLog FlexPen Syringe 15mL $222.41 NovoLog 100 units/ml
PATIENT INFORMATION LEAFLET. Forceval Junior Capsules
 PATIENT INFORMATION LEAFLET Forceval Junior Capsules Read all of this leaflet carefully because it contains important information for you or your child. Please note this leaflet has been written as if
PATIENT INFORMATION LEAFLET Forceval Junior Capsules Read all of this leaflet carefully because it contains important information for you or your child. Please note this leaflet has been written as if
Assistance. Teaching Plan. With Self-Administered Medication
 Assistance With Self-Administered Medication Teaching Plan Note to leader: Complete Page 4 of the learning guide and insert it in the learning guide before making copies of the lesson for each learner.
Assistance With Self-Administered Medication Teaching Plan Note to leader: Complete Page 4 of the learning guide and insert it in the learning guide before making copies of the lesson for each learner.
Alharith Hassan. Q 10 Method of Shelf-life estimation. Methods of Chemical stabilisation 11/20/2015
 Q 10 Method of Shelf-life estimation Q 10 approach is an old concept that could be useful for estimating the shelf-life at room temperature of products recommended for cold storage. Calculations are based
Q 10 Method of Shelf-life estimation Q 10 approach is an old concept that could be useful for estimating the shelf-life at room temperature of products recommended for cold storage. Calculations are based
June 2010 Pharmacy Professional
 f e at u r e s p e c i a l s Dealing with specials Responding to members requests the Society presents practical guidance for pharmacists on professional responsibilities when dealing with the supply of
f e at u r e s p e c i a l s Dealing with specials Responding to members requests the Society presents practical guidance for pharmacists on professional responsibilities when dealing with the supply of
Medication Guide. Serious loss of body fluid (dehydration) and changes in blood salts (electrolytes) in your blood.
 Medication Guide MoviPrep (moo-vee-prěp) (PEG 3350, sodium sulfate, sodium chloride, potassium chloride, sodium ascorbate, and ascorbic acid for oral solution) Read this Medication Guide before you start
Medication Guide MoviPrep (moo-vee-prěp) (PEG 3350, sodium sulfate, sodium chloride, potassium chloride, sodium ascorbate, and ascorbic acid for oral solution) Read this Medication Guide before you start
Good practice for drug calculations
 Good practice for drug calculations A step-by-step guide for nurses, doctors and all other healthcare professionals Pharmacyservices 7048 Drug Cal Guide A6 Update_AW.indd 1 22/07/2014 09:55 2 Contents
Good practice for drug calculations A step-by-step guide for nurses, doctors and all other healthcare professionals Pharmacyservices 7048 Drug Cal Guide A6 Update_AW.indd 1 22/07/2014 09:55 2 Contents
MEDICINE ADMINISTRATION WORKBOOK (YEAR 3; Semester 1)
 MEDICINE ADMINISTRATION WORKBOOK (YEAR 3; Semester 1) STUDENT S NAME: CLASS: PERSONAL TUTOR S NAME: MENTOR S SIGNATURE: DATE: Placement 1 MENTOR S SIGNATURE: DATE: Placement 2 To confirm that Mentor is
MEDICINE ADMINISTRATION WORKBOOK (YEAR 3; Semester 1) STUDENT S NAME: CLASS: PERSONAL TUTOR S NAME: MENTOR S SIGNATURE: DATE: Placement 1 MENTOR S SIGNATURE: DATE: Placement 2 To confirm that Mentor is
CENTRAL NERVOUS SYSTEM MANAGEMENT OF PARKINSON S DISEASE
 MANAGEMENT CENTRAL NERVOUS SYSTEM MANAGEMENT OF PARKINSON S DISEASE Parkinson s Disease is classically determined by the triad or rest tremor (usually starting in one arm) with bradykinesia (slowing of
MANAGEMENT CENTRAL NERVOUS SYSTEM MANAGEMENT OF PARKINSON S DISEASE Parkinson s Disease is classically determined by the triad or rest tremor (usually starting in one arm) with bradykinesia (slowing of
Your A-Z of Pain Relief A guide to pain relief medicines. We care, we discover, we teach
 Your A-Z of Pain Relief A guide to pain relief medicines We care, we discover, we teach Which pain medicines are you taking? Abstral (see Fentanyl Instant Tablets) Amitriptyline 5 Brufen (see Ibuprofen)
Your A-Z of Pain Relief A guide to pain relief medicines We care, we discover, we teach Which pain medicines are you taking? Abstral (see Fentanyl Instant Tablets) Amitriptyline 5 Brufen (see Ibuprofen)
Review Group: Mental Health Operational Medicines Management Group. Signature Signature Signature. Review Date: December 2014
 Mental Health NHS Grampian Mental Health Service Staff Guidance For The Prescribing Of Vitamin Supplementation During In-Patient Admission (Mental Health) For Alcohol Withdrawal Co-ordinators: Consultant
Mental Health NHS Grampian Mental Health Service Staff Guidance For The Prescribing Of Vitamin Supplementation During In-Patient Admission (Mental Health) For Alcohol Withdrawal Co-ordinators: Consultant
paediatric nursing: calculation skills
 Calculation skills p a e d i a t r i c n u r s i n g : c a l c u l a t i o n s k i l l s Written by: Meriel Hutton RGN, BA, CertEd, PhD, Senior Associate Dean (Undergraduate Studies, Nursing and Midwifery),
Calculation skills p a e d i a t r i c n u r s i n g : c a l c u l a t i o n s k i l l s Written by: Meriel Hutton RGN, BA, CertEd, PhD, Senior Associate Dean (Undergraduate Studies, Nursing and Midwifery),
DRUG CALCULATIONS FOR REGISTERED NURSES ADULT SERVICES
 Serving the people of north east Essex DRUG CALCULATIONS FOR REGISTERED NURSES ADULT SERVICES A Programmed Approach - It is essential you read this pack & practice the questions prior to your drug calculation
Serving the people of north east Essex DRUG CALCULATIONS FOR REGISTERED NURSES ADULT SERVICES A Programmed Approach - It is essential you read this pack & practice the questions prior to your drug calculation
**Form 1: - Consultant Copy** Telephone Number: Fax Number: Email: Author: Dr Bernard Udeze Pharmacist: Claire Ault Date of issue July 2011
 Effective Shared Care Agreement for the treatment of Dementia in Alzheimer s Disease Donepezil tablets / orodispersible tablets (Aricept / Aricept Evess ) These forms (1 and 2) are to be completed by both
Effective Shared Care Agreement for the treatment of Dementia in Alzheimer s Disease Donepezil tablets / orodispersible tablets (Aricept / Aricept Evess ) These forms (1 and 2) are to be completed by both
How To Take A Strong Opioid Painkiller
 Using strong painkillers for cancer pain This information is an extract from the booklet Controlling cancer pain. You may find the full booklet helpful. We can send you a copy free see page 8. Contents
Using strong painkillers for cancer pain This information is an extract from the booklet Controlling cancer pain. You may find the full booklet helpful. We can send you a copy free see page 8. Contents
Tube Feeding at Home Gravity or Syringe Feeding
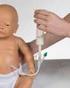 Tube Feeding at Home Gravity or Syringe Feeding Adapted from the former Taming the Feeding Tube, North Shore Hospital. 1 Where do I find information in this booklet? Subject Page How do I contact my health
Tube Feeding at Home Gravity or Syringe Feeding Adapted from the former Taming the Feeding Tube, North Shore Hospital. 1 Where do I find information in this booklet? Subject Page How do I contact my health
Pharmacy Savings Program
 Pharmacy Savings Program SELECT GENERICS DRUG LIST The Pharmacy Savings Program provides you with savings on select generic medications included on this list. The prices for these select generic medications
Pharmacy Savings Program SELECT GENERICS DRUG LIST The Pharmacy Savings Program provides you with savings on select generic medications included on this list. The prices for these select generic medications
PHARMACISTS AMENDMENTS TO PRESCRIPTIONS
 PHARMACISTS AMENDMENTS TO PRESCRIPTIONS May 2016 Version 2.3 Table of Contents 1. Introduction... 3 2. Purpose of this Policy... 3 3. Scope... 3 4. Definitions/Glossary... 3 5. Ownership and Responsibilities...
PHARMACISTS AMENDMENTS TO PRESCRIPTIONS May 2016 Version 2.3 Table of Contents 1. Introduction... 3 2. Purpose of this Policy... 3 3. Scope... 3 4. Definitions/Glossary... 3 5. Ownership and Responsibilities...
PATIENT INFORMATION LEAFLET. Calcium Sandoz Syrup calcium glubionate and calcium lactobionate
 PATIENT INFORMATION LEAFLET Calcium Sandoz Syrup calcium glubionate and calcium lactobionate Read all of this leaflet carefully before you start taking this medicine Keep this leaflet. You may need to
PATIENT INFORMATION LEAFLET Calcium Sandoz Syrup calcium glubionate and calcium lactobionate Read all of this leaflet carefully before you start taking this medicine Keep this leaflet. You may need to
Hyperosmolar Non-Ketotic Diabetic State (HONK)
 Hyperosmolar Non-Ketotic Diabetic State (HONK) University Hospitals of Leicester NHS Trust Guidelines for Management of Acute Medical Emergencies Management is largely the same as for diabetic ketoacidosis
Hyperosmolar Non-Ketotic Diabetic State (HONK) University Hospitals of Leicester NHS Trust Guidelines for Management of Acute Medical Emergencies Management is largely the same as for diabetic ketoacidosis
Usual total daily dosage *Indicates usual starting dose in mg/kg/day (mg of AED per kg of the child s weight per day) Drug (Generic Name)
 Table of Anti Epileptic Drugs (AEDs) used in the treatment of Epilepsy in Children under 12. IMPORTANT All the tables below are guidelines only, giving average daily dose ranges. Treatment will generally
Table of Anti Epileptic Drugs (AEDs) used in the treatment of Epilepsy in Children under 12. IMPORTANT All the tables below are guidelines only, giving average daily dose ranges. Treatment will generally
Clinically Significant Cardiovascular. Drug Interactions. April 30, 2014 Michael A Militello, PharmD, BCPS Cardiovascular Clinical Pharmacist
 Clinically Significant Cardiovascular Drug Interactions April 30, 2014 Michael A Militello, PharmD, BCPS Cardiovascular Clinical Pharmacist Introduction Drug interactions represent 3-5% of preventable
Clinically Significant Cardiovascular Drug Interactions April 30, 2014 Michael A Militello, PharmD, BCPS Cardiovascular Clinical Pharmacist Introduction Drug interactions represent 3-5% of preventable
UnitedHealthcare Group Medicare Advantage (PPO)
 Your Plan Explained UnitedHealthcare Group Medicare Advantage (PPO) UHEX11MP3230855_001 Y0066_100616_09113 Your Medicare. This brochure explains your Medicare Advantage plan, a type of health plan also
Your Plan Explained UnitedHealthcare Group Medicare Advantage (PPO) UHEX11MP3230855_001 Y0066_100616_09113 Your Medicare. This brochure explains your Medicare Advantage plan, a type of health plan also
LDU Maths, Stats and Numeracy Support. Metric Conversions
 Metric Conversions Here are some metric weights arranged in size order, starting with the biggest:- Kg (kilogram) g (gram) mg (milligram) mcg (microgram) Each one is a thousand (1000) times smaller than
Metric Conversions Here are some metric weights arranged in size order, starting with the biggest:- Kg (kilogram) g (gram) mg (milligram) mcg (microgram) Each one is a thousand (1000) times smaller than
Prompting, assisting and administration of medication in a care setting: guidance for professionals
 Prompting, assisting and administration of medication in a care setting: guidance for professionals Publication date: March 2015 Publication code HCR-0315-092 Page 1 of 6 Background Any care service provision
Prompting, assisting and administration of medication in a care setting: guidance for professionals Publication date: March 2015 Publication code HCR-0315-092 Page 1 of 6 Background Any care service provision
Following minor gynaecological surgery
 Following minor gynaecological surgery Exceptional healthcare, personally delivered n Following your operation you should have an adult to take you home and remain with you overnight. Transport home should
Following minor gynaecological surgery Exceptional healthcare, personally delivered n Following your operation you should have an adult to take you home and remain with you overnight. Transport home should
DRUG FOOD INTERACTIONS
 ANTIHYPERTENSIVES Natural licorice in large amounts can aggravate high blood pressure and can lower potassium levels. Natural licorice should be avoided with all antihypertensive medications. (Read food
ANTIHYPERTENSIVES Natural licorice in large amounts can aggravate high blood pressure and can lower potassium levels. Natural licorice should be avoided with all antihypertensive medications. (Read food
ROUTES OF DRUG ADMINISTRATION
 ROUTES OF DRUG ADMINISTRATION Review Article P. Verma (1) *, A.S. Thakur (1), K. Deshmukh (2), Dr. A.K. Jha (1) S. Verma (2) Address for Correspondence 1. Shri Shankaracharya Institute of Pharmaceutical
ROUTES OF DRUG ADMINISTRATION Review Article P. Verma (1) *, A.S. Thakur (1), K. Deshmukh (2), Dr. A.K. Jha (1) S. Verma (2) Address for Correspondence 1. Shri Shankaracharya Institute of Pharmaceutical
Pharmacokinetics: Do we know what we are doing? Prof Dana Niehaus Department of Psychiatry University of Stellenbosch
 Pharmacokinetics: Do we know what we are doing? Prof Dana Niehaus Department of Psychiatry University of Stellenbosch Content Pharmacokinetics (What the body does to the drug) Absorption Distribution Metabolism
Pharmacokinetics: Do we know what we are doing? Prof Dana Niehaus Department of Psychiatry University of Stellenbosch Content Pharmacokinetics (What the body does to the drug) Absorption Distribution Metabolism
RECONSTITUTING MEDICATIONS: HOW TO FLUFF UP MEDICATIONS
 RECONSTITUTING MEDICATIONS: HOW TO FLUFF UP MEDICATIONS After the completion of this module you will be able to: Define medication reconstitution. Read a medication label. Reconstitute a medication. Calculate
RECONSTITUTING MEDICATIONS: HOW TO FLUFF UP MEDICATIONS After the completion of this module you will be able to: Define medication reconstitution. Read a medication label. Reconstitute a medication. Calculate
Gastrostomy Feeding CARE GUIDELINES FOR PATIENTS & CARERS. Accessory items. Freka UK Funnel Adapter. Fixation Plate CH9. Male Luer Lock Adapter
 Accessory items Freka UK Funnel Adapter Fixation Plate CH9 Code: 7755681 Code: 7903002 Male Luer Lock Adapter CH9 Fixation Plate CH15 Code: 7981311 Code: 7904002 Male Luer Lock Adapter CH15 Code: 798137Y
Accessory items Freka UK Funnel Adapter Fixation Plate CH9 Code: 7755681 Code: 7903002 Male Luer Lock Adapter CH9 Fixation Plate CH15 Code: 7981311 Code: 7904002 Male Luer Lock Adapter CH15 Code: 798137Y
Health (Drugs and Poisons) Regulation 1996. Drug Therapy Protocol Rural and Isolated Practice Area Endorsed Nurse
 Health (Drugs and Poisons) Regulation 1996 Drug Therapy Protocol Rural and Isolated Practice Area Endorsed Nurse Health Protection Unit Medicines Regulation and Quality PO Box 21 Fortitude Valley BC QLD
Health (Drugs and Poisons) Regulation 1996 Drug Therapy Protocol Rural and Isolated Practice Area Endorsed Nurse Health Protection Unit Medicines Regulation and Quality PO Box 21 Fortitude Valley BC QLD
patient group direction
 DICLOFENAC v01 1/8 DICLOFENAC PGD Details Version 1.0 Legal category Staff grades Approved by POM Paramedic (Non-ECP) Nurse (Non-ECP) Emergency Care Practitioner (Paramedic) Emergency Care Practitioner
DICLOFENAC v01 1/8 DICLOFENAC PGD Details Version 1.0 Legal category Staff grades Approved by POM Paramedic (Non-ECP) Nurse (Non-ECP) Emergency Care Practitioner (Paramedic) Emergency Care Practitioner
INTRAVENOUS DRUG QUICKGUIDE
 INTRAVENOUS DRUG QUICKGUIDE GENERAL NOTES IMPORTANT THIS GUIDE IS AN AIDE MEMOIRE ONLY, AND SHOULD NOT REPLACE YOUR PROFESSIONAL REQUIREMENT TO USE MANUFACTUER S LICENSED INFORMATION TO INFORM YOUR PREPARATION
INTRAVENOUS DRUG QUICKGUIDE GENERAL NOTES IMPORTANT THIS GUIDE IS AN AIDE MEMOIRE ONLY, AND SHOULD NOT REPLACE YOUR PROFESSIONAL REQUIREMENT TO USE MANUFACTUER S LICENSED INFORMATION TO INFORM YOUR PREPARATION
Understanding ph and Osmolarity. Marc Stranz, PharmD
 Understanding ph and Osmolarity Marc Stranz, PharmD Outline ph and osmolarity tolerance guidelines, Definitions of ph and osmolarity, ph and osmolarity in vitro data, ph and osmolarity of common infusions,
Understanding ph and Osmolarity Marc Stranz, PharmD Outline ph and osmolarity tolerance guidelines, Definitions of ph and osmolarity, ph and osmolarity in vitro data, ph and osmolarity of common infusions,
PACKAGE LEAFLET: INFORMATION FOR THE USER. VITAMINE B12 STEROP 1mg/1ml Solution for injection / oral solution. Cyanocobalamin
 PACKAGE LEAFLET: INFORMATION FOR THE USER VITAMINE B12 STEROP 1mg/1ml Solution for injection / oral solution Cyanocobalamin Read all of this leaflet carefully before you start using this medicine because
PACKAGE LEAFLET: INFORMATION FOR THE USER VITAMINE B12 STEROP 1mg/1ml Solution for injection / oral solution Cyanocobalamin Read all of this leaflet carefully before you start using this medicine because
