Experiment C-31 Color Absorption
|
|
|
- Lizbeth Scott
- 7 years ago
- Views:
Transcription
1 1 Experiment C-31 Color Absorption Objectives To understand the concepts of light waves and color. To investigate how red, green and blue liquids absorb light of different wavelengths. To learn about colorimeter applications. Modules and Sensors PC + NeuLog application USB-200 module NUL-219 Colorimeter logger sensor Equipment and Accessories 50 ml beaker 3 Plastic cuvette (included with 3 the sensor) The items above (except for the cuvette) are included in the NeuLog Utility accessories, UTL-KIT. Materials 20 ml of red food coloring (1:40 dilution) 20 ml of green food coloring (1:40 dilution) 20 ml of blue food coloring (1:40 dilution)
2 2 Introduction In order to understand color concepts, it is first necessary to understand light concepts. Visible light waves are seen by the human eye as different colors and they are characterized by their wavelength, which is the distance between any two corresponding points on successive waves. The length of the wave determines the amount of energy it has; the shorter the wavelength, the higher the energy. Color may be also defined in more subjective terms, as something perceived by an individual. When light is reflected from an object, cones and rods in the retina of the eye respond to the light and the brain interprets the information received as color. Light can be directly absorbed by an object, reflected at the surface or transmitted through it. The electromagnetic spectrum which is visible to us is in the range of nm. When visible light with an energy distribution similar to sunlight (light of all colors) completely reflects from an object, this light appears white to the human eye. When the object completely absorbs all the light, it is recognized as black. When we project light through a transparent colored liquid, some light is absorbed and some is transmitted (and reflected). A red liquid transmits mostly red light and absorbs the rest. This principle is used in colorimetry. Colorimetry is a procedure in which a solution is analyzed by passing selected wavelengths of light (in the visible range) through a solution and measuring the amount of light that is transmitted. Then the absorbed light can be calculated.
3 3 The quality (wavelengths) of the absorbed light depends on the molecule dissolved in the solvent. This may help to identify a solute. The quantity of the absorbed light depends on the concentration of the absorbing solute (see experiment C-28, Beer-Lambert law). This helps when you want to determine the concentration of a solute in a solution. In this experiment we will investigate the absorbance of red, green, and blue food coloring after irradiating light in red (640 nm), green (524 nm) and blue (470 nm) wavelengths. Procedure Experiment setup 1. Set up the experiment as shown in the picture below. 2. Make sure you have three 50 ml beakers, each containing 20 ml of diluted food coloring (red, green and blue). It is recommended to dilute the food coloring by 40 (depends on the initial concentration). Make sure you also have three cuvettes.
4 4 Sensor setup 3. Connect the USB-200 module to the PC. 4. Check that the colorimeter sensor is connected to the USB-200 module. Note: The following application functions are explained in short. It is recommended to practice the NeuLog application functions (as described in the user manual) beforehand. 5. Run the NeuLog application and check that the colorimeter sensor is identified.
5 5 Settings 6. Click on the On-line Experiment icon in the NeuLog main icon bar. 7. Click on the Sensor's Module box. 8. Click on the Range button. 9. Select the "Red Abs" button to change the sensor's mode to absorbance of red wavelengths. 10. This experiment is done in single step mode so the experiment duration and sample rate will not be set.
6 6 Testing and measurements 11. Pour the red food coloring into the cuvette so that it will fill a third of it. 12. Place the cuvette in the colorimeter and click on the Single Step icon wavelength. in order to measure absorbance in the red 13. Click on the Table icon on the bottom part of the screen. A table will be displayed for data record. 14. Change the "Manual values" column name to "Light/Liquid" (clicking on the title of the column will allow you to do so). 15. Fill the suitable name in the "Light/Liquid" column, for this measurement it will be "Red/Red".
7 16. Fill in the following table with your measurement: 7 Liquid Red food coloring Green food coloring Blue food coloring Red Light irradiated Green Blue 17. Click on the Erase Experiment icon. 18. Click on the sensor's Module box and change the irradiated light to green. 19. Repeat the measurement while filling the suitable name in the "Light/Liquid" column, for this measurement it will be "Green/Red". 20. Repeat the measurements according to table above.
8 8 21. This is an example of irradiating the red food coloring with red, green and blue light. We can see that the red liquid does not absorb any of the red light (A=0). However, it absorbs green and blue light (A=0.83 and A=0.69 respectively). Our results are compatible with the concepts discussed in the introduction. When we project light through a transparent colored liquid, some light is absorbed (the colors that we do not see) and the rest is transmitted and reflected (the colors that we do see).
9 9 Summary questions 1. Explain the results of the green and blue food coloring measurements. 2. If the liquids were more diluted, how would it affect the results? Explain. 3. What is the best light color (of the colorimeter) for determining the concentration of your red, green and blue food coloring? 4. According to your results, which wavelengths (in terms of colors) do you think chlorophylls use in photosynthesis?
Experiment P-17 Magnetic Field Strength
 1 Experiment P-17 Magnetic Field Strength Objectives To learn about basic properties of magnets. To study the relationship between magnetic field strength and the distance from the magnet. Modules and
1 Experiment P-17 Magnetic Field Strength Objectives To learn about basic properties of magnets. To study the relationship between magnetic field strength and the distance from the magnet. Modules and
2 Spectrophotometry and the Analysis of Riboflavin
 2 Spectrophotometry and the Analysis of Riboflavin Objectives: A) To become familiar with operating the Platereader; B) to learn how to use the Platereader in determining the absorption spectrum of a compound
2 Spectrophotometry and the Analysis of Riboflavin Objectives: A) To become familiar with operating the Platereader; B) to learn how to use the Platereader in determining the absorption spectrum of a compound
Beer's Law: Colorimetry of Copper(II) Solutions
 Exercise 11 Page 1 Illinois Central College CHEMISTRY 130 Name: Beer's Law: Colorimetry of Copper(II) Solutions Objectives In this experiment, we will use Beer's Law to determine the unknown concentrations
Exercise 11 Page 1 Illinois Central College CHEMISTRY 130 Name: Beer's Law: Colorimetry of Copper(II) Solutions Objectives In this experiment, we will use Beer's Law to determine the unknown concentrations
Determining the Free Chlorine Content of Swimming Pool Water. HOCl H + + OCl. Evaluation copy
 Determining the Free Chlorine Content of Swimming Pool Water Computer 33 Physicians in the nineteenth century used chlorine water as a disinfectant. Upon the discovery that certain diseases were transmitted
Determining the Free Chlorine Content of Swimming Pool Water Computer 33 Physicians in the nineteenth century used chlorine water as a disinfectant. Upon the discovery that certain diseases were transmitted
NEULOG HEART RATE AND PULSE LOGGER SENSOR GUIDE
 NeuLog heart rate and pulse logger sensor NUL-208 The NeuLog pulse sensor can be used for any science experiment which requires a human heart rate or pulse measurement such as in the fields of Biology,
NeuLog heart rate and pulse logger sensor NUL-208 The NeuLog pulse sensor can be used for any science experiment which requires a human heart rate or pulse measurement such as in the fields of Biology,
Determining the Quantity of Iron in a Vitamin Tablet. Evaluation copy
 Determining the Quantity of Iron in a Vitamin Tablet Computer 34 As biochemical research becomes more sophisticated, we are learning more about the role of metallic elements in the human body. For example,
Determining the Quantity of Iron in a Vitamin Tablet Computer 34 As biochemical research becomes more sophisticated, we are learning more about the role of metallic elements in the human body. For example,
Appendix C. Vernier Tutorial
 C-1. Vernier Tutorial Introduction: In this lab course, you will collect, analyze and interpret data. The purpose of this tutorial is to teach you how to use the Vernier System to collect and transfer
C-1. Vernier Tutorial Introduction: In this lab course, you will collect, analyze and interpret data. The purpose of this tutorial is to teach you how to use the Vernier System to collect and transfer
The Determination of an Equilibrium Constant
 The Determination of an Equilibrium Constant Computer 10 Chemical reactions occur to reach a state of equilibrium. The equilibrium state can be characterized by quantitatively defining its equilibrium
The Determination of an Equilibrium Constant Computer 10 Chemical reactions occur to reach a state of equilibrium. The equilibrium state can be characterized by quantitatively defining its equilibrium
A Beer s Law Experiment
 A Beer s Law Experiment Introduction There are many ways to determine concentrations of a substance in solution. So far, the only experiences you may have are acid-base titrations or possibly determining
A Beer s Law Experiment Introduction There are many ways to determine concentrations of a substance in solution. So far, the only experiences you may have are acid-base titrations or possibly determining
1051-232 Imaging Systems Laboratory II. Laboratory 4: Basic Lens Design in OSLO April 2 & 4, 2002
 05-232 Imaging Systems Laboratory II Laboratory 4: Basic Lens Design in OSLO April 2 & 4, 2002 Abstract: For designing the optics of an imaging system, one of the main types of tools used today is optical
05-232 Imaging Systems Laboratory II Laboratory 4: Basic Lens Design in OSLO April 2 & 4, 2002 Abstract: For designing the optics of an imaging system, one of the main types of tools used today is optical
INTRODUCTION TO SPECTROMETRY
 Experiment 11 INTRODUCTION TO SPECTROMETRY Portions adapted by Ross S. Nord and Colleagues, Chemistry Department, Eastern Michigan University from ANAL 043, written by Donald F. Clemens and Warren A. McAllister,
Experiment 11 INTRODUCTION TO SPECTROMETRY Portions adapted by Ross S. Nord and Colleagues, Chemistry Department, Eastern Michigan University from ANAL 043, written by Donald F. Clemens and Warren A. McAllister,
EXPERIMENT 11 UV/VIS Spectroscopy and Spectrophotometry: Spectrophotometric Analysis of Potassium Permanganate Solutions.
 EXPERIMENT 11 UV/VIS Spectroscopy and Spectrophotometry: Spectrophotometric Analysis of Potassium Permanganate Solutions. Outcomes After completing this experiment, the student should be able to: 1. Prepare
EXPERIMENT 11 UV/VIS Spectroscopy and Spectrophotometry: Spectrophotometric Analysis of Potassium Permanganate Solutions. Outcomes After completing this experiment, the student should be able to: 1. Prepare
Photosynthesis and Light in the Ocean Adapted from The Fluid Earth / Living Ocean Heather Spalding, UH GK-12 program
 Photosynthesis and Light in the Ocean Adapted from The Fluid Earth / Living Ocean Heather Spalding, UH GK-12 program Algae, like your Halimeda, and plants live in very different environments, but they
Photosynthesis and Light in the Ocean Adapted from The Fluid Earth / Living Ocean Heather Spalding, UH GK-12 program Algae, like your Halimeda, and plants live in very different environments, but they
Electromagnetic Radiation (EMR) and Remote Sensing
 Electromagnetic Radiation (EMR) and Remote Sensing 1 Atmosphere Anything missing in between? Electromagnetic Radiation (EMR) is radiated by atomic particles at the source (the Sun), propagates through
Electromagnetic Radiation (EMR) and Remote Sensing 1 Atmosphere Anything missing in between? Electromagnetic Radiation (EMR) is radiated by atomic particles at the source (the Sun), propagates through
Spectrophotometry and the Beer-Lambert Law: An Important Analytical Technique in Chemistry
 Spectrophotometry and the Beer-Lambert Law: An Important Analytical Technique in Chemistry Jon H. Hardesty, PhD and Bassam Attili, PhD Collin College Department of Chemistry Introduction: In the last lab
Spectrophotometry and the Beer-Lambert Law: An Important Analytical Technique in Chemistry Jon H. Hardesty, PhD and Bassam Attili, PhD Collin College Department of Chemistry Introduction: In the last lab
Absorbance Spectrophotometry: Analysis of FD&C Red Food Dye #40
 Absorbance Spectrophotometry: Analysis of FD&C Red Food Dye #40 Note: there is a second document that goes with this one! 2046 - Absorbance Spectrophotometry - Calibration Curve Procedure. The second document
Absorbance Spectrophotometry: Analysis of FD&C Red Food Dye #40 Note: there is a second document that goes with this one! 2046 - Absorbance Spectrophotometry - Calibration Curve Procedure. The second document
Using the Spectrophotometer
 Using the Spectrophotometer Introduction In this exercise, you will learn the basic principals of spectrophotometry and and serial dilution and their practical application. You will need these skills to
Using the Spectrophotometer Introduction In this exercise, you will learn the basic principals of spectrophotometry and and serial dilution and their practical application. You will need these skills to
MAKING SENSE OF ENERGY Electromagnetic Waves
 Adapted from State of Delaware TOE Unit MAKING SENSE OF ENERGY Electromagnetic Waves GOALS: In this Part of the unit you will Learn about electromagnetic waves, how they are grouped, and how each group
Adapted from State of Delaware TOE Unit MAKING SENSE OF ENERGY Electromagnetic Waves GOALS: In this Part of the unit you will Learn about electromagnetic waves, how they are grouped, and how each group
Photosynthesis and Cellular Respiration. Stored Energy
 Photosynthesis and Cellular Respiration Stored Energy What is Photosynthesis? plants convert the energy of sunlight into the energy in the chemical bonds of carbohydrates sugars and starches. SUMMARY EQUATION:
Photosynthesis and Cellular Respiration Stored Energy What is Photosynthesis? plants convert the energy of sunlight into the energy in the chemical bonds of carbohydrates sugars and starches. SUMMARY EQUATION:
2C: One in a Million. Part 1: Making solutions. Name: Section: Date: Materials
 Name: Section: Date: 2C: One in a Million Drinking water can contain up to 1.3 parts per million (ppm) of copper and still be considered safe. What does parts per million mean? Both living things and the
Name: Section: Date: 2C: One in a Million Drinking water can contain up to 1.3 parts per million (ppm) of copper and still be considered safe. What does parts per million mean? Both living things and the
Name Class Date. spectrum. White is not a color, but is a combination of all colors. Black is not a color; it is the absence of all light.
 Exercises 28.1 The Spectrum (pages 555 556) 1. Isaac Newton was the first person to do a systematic study of color. 2. Circle the letter of each statement that is true about Newton s study of color. a.
Exercises 28.1 The Spectrum (pages 555 556) 1. Isaac Newton was the first person to do a systematic study of color. 2. Circle the letter of each statement that is true about Newton s study of color. a.
Color and Light. DELTA SCIENCE READER Overview... 125 Before Reading... 126 Guide the Reading... 127 After Reading... 133
 Color and Light T ABLE OF CONTENTS ABOUT DELTA SCIENCE MODULES Program Introduction................... iii Teacher s Guide..................... iv Delta Science Readers............... vi Equipment and
Color and Light T ABLE OF CONTENTS ABOUT DELTA SCIENCE MODULES Program Introduction................... iii Teacher s Guide..................... iv Delta Science Readers............... vi Equipment and
Upon completion of this lab, the student will be able to:
 1 Learning Outcomes EXPERIMENT B4: CHEMICAL EQUILIBRIUM Upon completion of this lab, the student will be able to: 1) Analyze the absorbance spectrum of a sample. 2) Calculate the equilibrium constant for
1 Learning Outcomes EXPERIMENT B4: CHEMICAL EQUILIBRIUM Upon completion of this lab, the student will be able to: 1) Analyze the absorbance spectrum of a sample. 2) Calculate the equilibrium constant for
8.2 Cells and Energy. What is photosynthesis? Photosynthesis takes place in the chloroplasts. CHAPTER 8. Solar cells and chloroplasts
 CHAPTER 8 CELL PROCESSES 8.2 Cells and Energy To stay alive, you need a constant supply of energy. You need energy to move, think, grow, and even sleep. Where does that energy come from? It all starts
CHAPTER 8 CELL PROCESSES 8.2 Cells and Energy To stay alive, you need a constant supply of energy. You need energy to move, think, grow, and even sleep. Where does that energy come from? It all starts
COLORIMETER. Description 0358BT. Figure 1. The Colorimeter
 COLORIMETER Description 0358BT Figure 1. The Colorimeter Introduction The Colorimeter is designed to determine the concentration of a solution by analyzing its color intensity. Monochromatic light from
COLORIMETER Description 0358BT Figure 1. The Colorimeter Introduction The Colorimeter is designed to determine the concentration of a solution by analyzing its color intensity. Monochromatic light from
Effect of Light Colors on Bean Plant Growth
 Effect of Light Colors on Bean Plant Growth Teacher Edition Grade: Grades 6-8 Delaware State Science Standard: Science Standard 6 - Life Processes Strand: Structure/Function Relationship Strand: Matter
Effect of Light Colors on Bean Plant Growth Teacher Edition Grade: Grades 6-8 Delaware State Science Standard: Science Standard 6 - Life Processes Strand: Structure/Function Relationship Strand: Matter
THE NATURE OF LIGHT AND COLOR
 THE NATURE OF LIGHT AND COLOR THE PHYSICS OF LIGHT Electromagnetic radiation travels through space as electric energy and magnetic energy. At times the energy acts like a wave and at other times it acts
THE NATURE OF LIGHT AND COLOR THE PHYSICS OF LIGHT Electromagnetic radiation travels through space as electric energy and magnetic energy. At times the energy acts like a wave and at other times it acts
Evaluation copy. Case File 9. A Killer Cup of Coffee? GlobalTech manager dies
 Case File 9 Killer Cup of Coffee: Using colorimetry to determine concentration of a poison Determine the concentration of cyanide in the solution. A Killer Cup of Coffee? SOUTH PAINTER, Tuesday: It was
Case File 9 Killer Cup of Coffee: Using colorimetry to determine concentration of a poison Determine the concentration of cyanide in the solution. A Killer Cup of Coffee? SOUTH PAINTER, Tuesday: It was
Lab #11: Determination of a Chemical Equilibrium Constant
 Lab #11: Determination of a Chemical Equilibrium Constant Objectives: 1. Determine the equilibrium constant of the formation of the thiocyanatoiron (III) ions. 2. Understand the application of using a
Lab #11: Determination of a Chemical Equilibrium Constant Objectives: 1. Determine the equilibrium constant of the formation of the thiocyanatoiron (III) ions. 2. Understand the application of using a
Review Vocabulary spectrum: a range of values or properties
 Standards 7.3.19: Explain that human eyes respond to a narrow range of wavelengths of the electromagnetic spectrum. 7.3.20: Describe that something can be seen when light waves emitted or reflected by
Standards 7.3.19: Explain that human eyes respond to a narrow range of wavelengths of the electromagnetic spectrum. 7.3.20: Describe that something can be seen when light waves emitted or reflected by
What s in the Mix? Liquid Color Spectroscopy Lab (Randy Landsberg & Bill Fisher)
 What s in the Mix? Liquid Color Spectroscopy Lab (Randy Landsberg & Bill Fisher) Introduction: There is more to a color than a name. Color can tell us lots of information. In this lab you will use a spectrophotometer
What s in the Mix? Liquid Color Spectroscopy Lab (Randy Landsberg & Bill Fisher) Introduction: There is more to a color than a name. Color can tell us lots of information. In this lab you will use a spectrophotometer
The Determination of an Equilibrium Constant
 The Determination of an Equilibrium Constant Chemical reactions occur to reach a state of equilibrium. The equilibrium state can be characterized by quantitatively defining its equilibrium constant, K
The Determination of an Equilibrium Constant Chemical reactions occur to reach a state of equilibrium. The equilibrium state can be characterized by quantitatively defining its equilibrium constant, K
To determine the mass of iron in one adult dose of either a ferrous sulfate or. ferrous gluconate iron supplement using a colorimetric technique.
 Lab: Colorimetric Analysis of Iron in Iron Supplements Purpose To determine the mass of iron in one adult dose of either a ferrous sulfate or ferrous gluconate iron supplement using a colorimetric technique.
Lab: Colorimetric Analysis of Iron in Iron Supplements Purpose To determine the mass of iron in one adult dose of either a ferrous sulfate or ferrous gluconate iron supplement using a colorimetric technique.
University of Wisconsin Chemistry 524 Spectroscopic Applications (GFAA, ICP, UV/Vis, Fluorescence)
 University of Wisconsin Chemistry 524 Spectroscopic Applications (GFAA, ICP, UV/Vis, Fluorescence) For this laboratory exercise, you will explore a variety of spectroscopic methods used in an analytical
University of Wisconsin Chemistry 524 Spectroscopic Applications (GFAA, ICP, UV/Vis, Fluorescence) For this laboratory exercise, you will explore a variety of spectroscopic methods used in an analytical
UV/VIS/IR SPECTROSCOPY ANALYSIS OF NANOPARTICLES
 UV/VIS/IR SPECTROSCOPY ANALYSIS OF NANOPARTICLES SEPTEMBER 2012, V 1.1 4878 RONSON CT STE K SAN DIEGO, CA 92111 858-565 - 4227 NANOCOMPOSIX.COM Note to the Reader: We at nanocomposix have published this
UV/VIS/IR SPECTROSCOPY ANALYSIS OF NANOPARTICLES SEPTEMBER 2012, V 1.1 4878 RONSON CT STE K SAN DIEGO, CA 92111 858-565 - 4227 NANOCOMPOSIX.COM Note to the Reader: We at nanocomposix have published this
TEACHER ACTIVITY GUIDE
 Page 1/5 TEACHER ACTIVITY GUIDE EFFECT OF HEAT & ph ON COLOR & TEXTURE OF GREEN VEGETABLES Taken from IFT Experiments in Food Science Series Color plays a key role in establishing consumer acceptability
Page 1/5 TEACHER ACTIVITY GUIDE EFFECT OF HEAT & ph ON COLOR & TEXTURE OF GREEN VEGETABLES Taken from IFT Experiments in Food Science Series Color plays a key role in establishing consumer acceptability
Principle of Thermal Imaging
 Section 8 All materials, which are above 0 degrees Kelvin (-273 degrees C), emit infrared energy. The infrared energy emitted from the measured object is converted into an electrical signal by the imaging
Section 8 All materials, which are above 0 degrees Kelvin (-273 degrees C), emit infrared energy. The infrared energy emitted from the measured object is converted into an electrical signal by the imaging
Chemistry 111 Lab: Intro to Spectrophotometry Page E-1
 Chemistry 111 Lab: Intro to Spectrophotometry Page E-1 SPECTROPHOTOMETRY Absorption Measurements & their Application to Quantitative Analysis study of the interaction of light (or other electromagnetic
Chemistry 111 Lab: Intro to Spectrophotometry Page E-1 SPECTROPHOTOMETRY Absorption Measurements & their Application to Quantitative Analysis study of the interaction of light (or other electromagnetic
Shimadzu UV-VIS User s Guide
 Shimadzu UV-VIS User s Guide 1) Push the F4 button on the UV-VIS instrument keypad. This will enable PC control. Push the F4 Button 2) Log into the UV-VIS software with your username and password. 3) After
Shimadzu UV-VIS User s Guide 1) Push the F4 button on the UV-VIS instrument keypad. This will enable PC control. Push the F4 Button 2) Log into the UV-VIS software with your username and password. 3) After
Using Spectrophotometers to Examine Photosynthetic Rates Under Various Qualities of Light
 Purdue GK-12 Lesson Plan 2006-07 Using Spectrophotometers to Examine Photosynthetic Rates Under Various Qualities of Light Purdue University GK-12 2006-2007 Lead developer and contact: Amanda Deering Purdue
Purdue GK-12 Lesson Plan 2006-07 Using Spectrophotometers to Examine Photosynthetic Rates Under Various Qualities of Light Purdue University GK-12 2006-2007 Lead developer and contact: Amanda Deering Purdue
Experiment 2 Kinetics II Concentration-Time Relationships and Activation Energy
 2-1 Experiment 2 Kinetics II Concentration-Time Relationships and Activation Energy Introduction: The kinetics of a decomposition reaction involving hydroxide ion and crystal violet, an organic dye used
2-1 Experiment 2 Kinetics II Concentration-Time Relationships and Activation Energy Introduction: The kinetics of a decomposition reaction involving hydroxide ion and crystal violet, an organic dye used
Measuring Manganese Concentration Using Spectrophotometry
 Measuring Manganese Concentration Using Spectrophotometry Objectives To use spectroscopy to determine the amount of Manganese is an unknown sample. Scenario Your have just joined a "Green Team" at SMC
Measuring Manganese Concentration Using Spectrophotometry Objectives To use spectroscopy to determine the amount of Manganese is an unknown sample. Scenario Your have just joined a "Green Team" at SMC
Overview. What is EMR? Electromagnetic Radiation (EMR) LA502 Special Studies Remote Sensing
 LA502 Special Studies Remote Sensing Electromagnetic Radiation (EMR) Dr. Ragab Khalil Department of Landscape Architecture Faculty of Environmental Design King AbdulAziz University Room 103 Overview What
LA502 Special Studies Remote Sensing Electromagnetic Radiation (EMR) Dr. Ragab Khalil Department of Landscape Architecture Faculty of Environmental Design King AbdulAziz University Room 103 Overview What
SYNTHESIS AND ANALYSIS OF SILVER/GOLD NANOPARTICLES
 SYNTHESIS AND ANALYSIS OF SILVER/GOLD NANOPARTICLES Background Shelby Hatch and George Schatz Northwestern University, Evanston, IL 60208 All physical and chemical properties are size dependent, and the
SYNTHESIS AND ANALYSIS OF SILVER/GOLD NANOPARTICLES Background Shelby Hatch and George Schatz Northwestern University, Evanston, IL 60208 All physical and chemical properties are size dependent, and the
Measuring Protein Concentration through Absorption Spectrophotometry
 Measuring Protein Concentration through Absorption Spectrophotometry In this lab exercise you will learn how to homogenize a tissue to extract the protein, and then how to use a protein assay reagent to
Measuring Protein Concentration through Absorption Spectrophotometry In this lab exercise you will learn how to homogenize a tissue to extract the protein, and then how to use a protein assay reagent to
Shielding Ourselves from Ultraviolet Light
 Shielding Ourselves from Ultraviolet Light Introduction The purpose of this experiment is to examine the effect of passing ultraviolet (UV) light through a variety of materials to determine how well each
Shielding Ourselves from Ultraviolet Light Introduction The purpose of this experiment is to examine the effect of passing ultraviolet (UV) light through a variety of materials to determine how well each
Can Traffic Accidents be eliminated by Robots?
 Can Traffic Accidents be eliminated by Robots? Elementary Science and Technology Grade 7 Teaching- learning Module for Unit Light and Sound Abstract This modules leads to a decision making activity related
Can Traffic Accidents be eliminated by Robots? Elementary Science and Technology Grade 7 Teaching- learning Module for Unit Light and Sound Abstract This modules leads to a decision making activity related
After a wave passes through a medium, how does the position of that medium compare to its original position?
 Light Waves Test Question Bank Standard/Advanced Name: Question 1 (1 point) The electromagnetic waves with the highest frequencies are called A. radio waves. B. gamma rays. C. X-rays. D. visible light.
Light Waves Test Question Bank Standard/Advanced Name: Question 1 (1 point) The electromagnetic waves with the highest frequencies are called A. radio waves. B. gamma rays. C. X-rays. D. visible light.
Application Note: Absorbance
 Units Units Theory of absorbance Light absorption occurs when atoms or molecules take up the energy of a photon of light, thereby reducing the transmission of light as it is passed through a sample. Light
Units Units Theory of absorbance Light absorption occurs when atoms or molecules take up the energy of a photon of light, thereby reducing the transmission of light as it is passed through a sample. Light
Green Heating. Pupil Research Brief. Teachers Notes. Syllabus Coverage Subject Knowledge and Understanding. Route through the Brief UPIL ESEARCHER
 R P UPIL ESEARCHER Green Heating I NITIATIVE Pupil Research Brief Teachers Notes Syllabus Coverage Subject Knowledge and Understanding all types of electromagnetic radiation form a continuous spectrum
R P UPIL ESEARCHER Green Heating I NITIATIVE Pupil Research Brief Teachers Notes Syllabus Coverage Subject Knowledge and Understanding all types of electromagnetic radiation form a continuous spectrum
From lowest energy to highest energy, which of the following correctly orders the different categories of electromagnetic radiation?
 From lowest energy to highest energy, which of the following correctly orders the different categories of electromagnetic radiation? From lowest energy to highest energy, which of the following correctly
From lowest energy to highest energy, which of the following correctly orders the different categories of electromagnetic radiation? From lowest energy to highest energy, which of the following correctly
Experiment 13H THE REACTION OF RED FOOD COLOR WITH BLEACH 1
 Experiment 13H FV 1/25/2011(2-run) THE REACTION OF RED FOOD COLOR WITH BLEACH 1 PROBLEM: Determine the rate law for the chemical reaction between FD&C Red Dye #3 and sodium hypochlorite. LEARNING OBJECTIVES:
Experiment 13H FV 1/25/2011(2-run) THE REACTION OF RED FOOD COLOR WITH BLEACH 1 PROBLEM: Determine the rate law for the chemical reaction between FD&C Red Dye #3 and sodium hypochlorite. LEARNING OBJECTIVES:
Computer Vision: Machine Vision Filters. Computer Vision. Optical Filters. 25 August 2014
 Computer Vision Optical Filters 25 August 2014 Copyright 2001 2014 by NHL Hogeschool, Van de Loosdrecht Machine Vision BV and Klaas Dijkstra All rights reserved j.van.de.loosdrecht@nhl.nl, jaap@vdlmv.nl,
Computer Vision Optical Filters 25 August 2014 Copyright 2001 2014 by NHL Hogeschool, Van de Loosdrecht Machine Vision BV and Klaas Dijkstra All rights reserved j.van.de.loosdrecht@nhl.nl, jaap@vdlmv.nl,
Drop Counter Sensor Product Number: ENDRP-AD100
 imagine explore learn Drop Counter Sensor Product Number: ENDRP-AD100 Overview The Drop Counter sensor is an optical sensor that accurately records the number of drops of titrant added during a titration.
imagine explore learn Drop Counter Sensor Product Number: ENDRP-AD100 Overview The Drop Counter sensor is an optical sensor that accurately records the number of drops of titrant added during a titration.
A. Incorrect! No, while this statement is correct, it is not the best answer to the question.
 Biochemistry - Problem Drill 18: Photosynthesis No. 1 of 10 1. What is photosynthesis? Select the best answer. (A) Photosynthesis happens in the chloroplasts. (B) Light absorption by chlorophyll induces
Biochemistry - Problem Drill 18: Photosynthesis No. 1 of 10 1. What is photosynthesis? Select the best answer. (A) Photosynthesis happens in the chloroplasts. (B) Light absorption by chlorophyll induces
Reaction Stoichiometry and the Formation of a Metal Ion Complex
 Reaction Stoichiometry and the Formation of a Metal Ion Complex Objectives The objectives of this laboratory are as follows: To use the method of continuous variation to determine the reaction stoichiometry
Reaction Stoichiometry and the Formation of a Metal Ion Complex Objectives The objectives of this laboratory are as follows: To use the method of continuous variation to determine the reaction stoichiometry
1 Laboratory #5: Grating Spectrometer
 SIMG-215-20061: LABORATORY #5 1 Laboratory #5: Grating Spectrometer 1.1 Objective: To observe and measure the spectra of different light sources. 1.2 Materials: 1. OSA optics kit. 2. Nikon digital camera
SIMG-215-20061: LABORATORY #5 1 Laboratory #5: Grating Spectrometer 1.1 Objective: To observe and measure the spectra of different light sources. 1.2 Materials: 1. OSA optics kit. 2. Nikon digital camera
Outline. Quantizing Intensities. Achromatic Light. Optical Illusion. Quantizing Intensities. CS 430/585 Computer Graphics I
 CS 430/585 Computer Graphics I Week 8, Lecture 15 Outline Light Physical Properties of Light and Color Eye Mechanism for Color Systems to Define Light and Color David Breen, William Regli and Maxim Peysakhov
CS 430/585 Computer Graphics I Week 8, Lecture 15 Outline Light Physical Properties of Light and Color Eye Mechanism for Color Systems to Define Light and Color David Breen, William Regli and Maxim Peysakhov
Phosphates (ortho- and total)
 INTRODUCTION Phosphates (ortho- and total) Phosphorus is an essential nutrient for all aquatic plants and algae. Only a very small amount is needed, however, so an excess of phosphorus can easily occur.
INTRODUCTION Phosphates (ortho- and total) Phosphorus is an essential nutrient for all aquatic plants and algae. Only a very small amount is needed, however, so an excess of phosphorus can easily occur.
Setting Up the MimioCapture Device
 Setting Up the MimioCapture Device The MimioCapture device enables you to use dry erase markers to draw on the whiteboard and record your writing without the need of a projector. Using MimioStudio Ink
Setting Up the MimioCapture Device The MimioCapture device enables you to use dry erase markers to draw on the whiteboard and record your writing without the need of a projector. Using MimioStudio Ink
Computer Vision. Color image processing. 25 August 2014
 Computer Vision Color image processing 25 August 2014 Copyright 2001 2014 by NHL Hogeschool and Van de Loosdrecht Machine Vision BV All rights reserved j.van.de.loosdrecht@nhl.nl, jaap@vdlmv.nl Color image
Computer Vision Color image processing 25 August 2014 Copyright 2001 2014 by NHL Hogeschool and Van de Loosdrecht Machine Vision BV All rights reserved j.van.de.loosdrecht@nhl.nl, jaap@vdlmv.nl Color image
Reaction of Blue Food Dye with Bleach
 Exercise 2 Reaction of Blue Food Dye with Bleach 2 Introduction In the experiment, you will study the rate of the reaction of FD&C Blue #1 (Blue #1 is denoted by E number E133 in food stuff) with sodium
Exercise 2 Reaction of Blue Food Dye with Bleach 2 Introduction In the experiment, you will study the rate of the reaction of FD&C Blue #1 (Blue #1 is denoted by E number E133 in food stuff) with sodium
GETTING TO KNOW YOUR 4G USB
 GETTING TO KNOW YOUR Telstra Pre-paid 4G USB LET S GET THIS SHOW ON THE ROAD WHAT S INSIDE Your Telstra Pre-Paid 4G USB has been tested to the highest standards to deliver you the best possible coverage
GETTING TO KNOW YOUR Telstra Pre-paid 4G USB LET S GET THIS SHOW ON THE ROAD WHAT S INSIDE Your Telstra Pre-Paid 4G USB has been tested to the highest standards to deliver you the best possible coverage
1. Three-Color Light. Introduction to Three-Color Light. Chapter 1. Adding Color Pigments. Difference Between Pigments and Light. Adding Color Light
 1. Three-Color Light Chapter 1 Introduction to Three-Color Light Many of us were taught at a young age that the primary colors are red, yellow, and blue. Our early experiences with color mixing were blending
1. Three-Color Light Chapter 1 Introduction to Three-Color Light Many of us were taught at a young age that the primary colors are red, yellow, and blue. Our early experiences with color mixing were blending
Experiment #5: Qualitative Absorption Spectroscopy
 Experiment #5: Qualitative Absorption Spectroscopy One of the most important areas in the field of analytical chemistry is that of spectroscopy. In general terms, spectroscopy deals with the interactions
Experiment #5: Qualitative Absorption Spectroscopy One of the most important areas in the field of analytical chemistry is that of spectroscopy. In general terms, spectroscopy deals with the interactions
INF-USB2 and SI-USB Quick Start Guide
 INF-USB2 and SI-USB Quick Start Guide Please follow these instructions carefully. DO NOT connect the INF-USB2 or SI-USB module to your computer before running the setup program. After running Setup and
INF-USB2 and SI-USB Quick Start Guide Please follow these instructions carefully. DO NOT connect the INF-USB2 or SI-USB module to your computer before running the setup program. After running Setup and
Austin Peay State University Department of Chemistry Chem 1111. The Use of the Spectrophotometer and Beer's Law
 Purpose To become familiar with using a spectrophotometer and gain an understanding of Beer s law and it s relationship to solution concentration. Introduction Scientists use many methods to determine
Purpose To become familiar with using a spectrophotometer and gain an understanding of Beer s law and it s relationship to solution concentration. Introduction Scientists use many methods to determine
Zetasizer Nano-ZS User Instructions
 Zetasizer Nano-ZS User Instructions 1. Activate the instrument computer by logging in to the central login system in the service corridor. If needed, log in to the local instrument computer Username: zetasizer.
Zetasizer Nano-ZS User Instructions 1. Activate the instrument computer by logging in to the central login system in the service corridor. If needed, log in to the local instrument computer Username: zetasizer.
Review Questions Photosynthesis
 Review Questions Photosynthesis 1. Describe a metabolic pathway. In a factory, labor is divided into small individual jobs. A carmaker, for example, will have one worker install the front windshield, another
Review Questions Photosynthesis 1. Describe a metabolic pathway. In a factory, labor is divided into small individual jobs. A carmaker, for example, will have one worker install the front windshield, another
CS 325 Computer Graphics
 CS 325 Computer Graphics 01 / 25 / 2016 Instructor: Michael Eckmann Today s Topics Review the syllabus Review course policies Color CIE system chromaticity diagram color gamut, complementary colors, dominant
CS 325 Computer Graphics 01 / 25 / 2016 Instructor: Michael Eckmann Today s Topics Review the syllabus Review course policies Color CIE system chromaticity diagram color gamut, complementary colors, dominant
CPI Links Content Guide & Five Items Resource
 CPI Links Content Guide & Five Items Resource Introduction The following information should be used as a companion to the CPI Links. It provides clarifications concerning the content and skills contained
CPI Links Content Guide & Five Items Resource Introduction The following information should be used as a companion to the CPI Links. It provides clarifications concerning the content and skills contained
VMobile Desktop Talk User Guide
 VMobile Desktop Talk User Guide 0 1. Description 1.1. VMobile Desktop Talk (mentioned in this document as Software) provides Voice over IP service for personal computers, working under Windows OS. 1.2.
VMobile Desktop Talk User Guide 0 1. Description 1.1. VMobile Desktop Talk (mentioned in this document as Software) provides Voice over IP service for personal computers, working under Windows OS. 1.2.
1. At which temperature would a source radiate the least amount of electromagnetic energy? 1) 273 K 3) 32 K 2) 212 K 4) 5 K
 1. At which temperature would a source radiate the least amount of electromagnetic energy? 1) 273 K 3) 32 K 2) 212 K 4) 5 K 2. How does the amount of heat energy reflected by a smooth, dark-colored concrete
1. At which temperature would a source radiate the least amount of electromagnetic energy? 1) 273 K 3) 32 K 2) 212 K 4) 5 K 2. How does the amount of heat energy reflected by a smooth, dark-colored concrete
Separation and Identification of Plant Pigments Dr. Gergens - SD Mesa College
 Separation and Identification of Plant Pigments Dr. Gergens - SD Mesa College PURPOSE In this experiment, the photosynthetic pigments common to all flowering plants will be extracted by liquidliquid extraction.
Separation and Identification of Plant Pigments Dr. Gergens - SD Mesa College PURPOSE In this experiment, the photosynthetic pigments common to all flowering plants will be extracted by liquidliquid extraction.
Beckman Coulter DTX 880 Multimode Detector Bergen County Technical Schools Stem Cell Lab
 Beckman Coulter DTX 880 Multimode Detector Bergen County Technical Schools Stem Cell Lab Room 213 Beckman Coulter DTX 880 Multimode Detector Information The Beckman Coulter DTX 880 Multimode Detector is
Beckman Coulter DTX 880 Multimode Detector Bergen County Technical Schools Stem Cell Lab Room 213 Beckman Coulter DTX 880 Multimode Detector Information The Beckman Coulter DTX 880 Multimode Detector is
Call Center - Agent Application User Manual
 Forum 700 Call Center Agent Application User Manual Legal notice: Belgacom and the Belgacom logo are trademarks of Belgacom. All other trademarks are the property of their respective owners. The information
Forum 700 Call Center Agent Application User Manual Legal notice: Belgacom and the Belgacom logo are trademarks of Belgacom. All other trademarks are the property of their respective owners. The information
3. USB DRIVER. 3-1. Installation of USB driver. 3-1-1. Driver for Infinity SD Data Logger. INFINITY Series Acquisition Tools Manual
 3. USB DRIVER 3-1. Installation of USB driver Before you launch this software, the driver should be once surely registered. Access between the instrument and a PC through the USB cable supplied by us as
3. USB DRIVER 3-1. Installation of USB driver Before you launch this software, the driver should be once surely registered. Access between the instrument and a PC through the USB cable supplied by us as
Macs are not directly compatible with Noetix.
 Those that use Macintosh computers to access Noetix cannot directly access Noetix through http://www.sandiego.edu/noetix. If one is on a Mac and goes to Noetix directly in a web browser, the page is likely
Those that use Macintosh computers to access Noetix cannot directly access Noetix through http://www.sandiego.edu/noetix. If one is on a Mac and goes to Noetix directly in a web browser, the page is likely
Jackson Systems E-commerce Guide
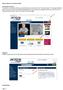 Accessing E-commerce To access the e-commerce feature, go to jacksonsystems.com and click on Contractor Login in the upper right hand corner. If you do not have a username and password, you can request
Accessing E-commerce To access the e-commerce feature, go to jacksonsystems.com and click on Contractor Login in the upper right hand corner. If you do not have a username and password, you can request
Renewable Energy Monitor User Manual And Software Reference Guide. sales@fuelcellstore.com (979) 703-1925
 Renewable Energy Monitor User Manual And Software Reference Guide sales@fuelcellstore.com (979) 703-1925 1 Introducing the Horizon Renewable Energy Monitor The Renewable Energy Monitor is an educational
Renewable Energy Monitor User Manual And Software Reference Guide sales@fuelcellstore.com (979) 703-1925 1 Introducing the Horizon Renewable Energy Monitor The Renewable Energy Monitor is an educational
Light Waves and Matter
 Name: Light Waves and Matter Read from Lesson 2 of the Light Waves and Color chapter at The Physics Classroom: http://www.physicsclassroom.com/class/light/u12l2a.html MOP Connection: Light and Color: sublevel
Name: Light Waves and Matter Read from Lesson 2 of the Light Waves and Color chapter at The Physics Classroom: http://www.physicsclassroom.com/class/light/u12l2a.html MOP Connection: Light and Color: sublevel
Cary 100 Bio UV-Vis Operating Instructions 09/25/2012 S.V.
 1234 Hach Hall 515-294-5805 www.cif.iastate.edu Cary 100 Bio UV-Vis Operating Instructions 09/25/2012 S.V. Location: Contact: 1240 Hach Hall Steve Veysey, 1234 Hach Hall Safety All researchers working
1234 Hach Hall 515-294-5805 www.cif.iastate.edu Cary 100 Bio UV-Vis Operating Instructions 09/25/2012 S.V. Location: Contact: 1240 Hach Hall Steve Veysey, 1234 Hach Hall Safety All researchers working
Use of Micropipettes
 Use of Micropipettes Prior to lab you should understand: The function of micropipettes in the laboratory Basic parts of micropipette What volumes are measured with P, P and P1 micopipettors How to read
Use of Micropipettes Prior to lab you should understand: The function of micropipettes in the laboratory Basic parts of micropipette What volumes are measured with P, P and P1 micopipettors How to read
DSM155 Digital LED Light Meter User s Manual DSM155
 DSM155 Digital LED Light Meter User s Manual DSM155 CONTENTS TABLE OF CONTENTS......3 LIMITED WARRANTY AND LIMITATION OF LIABILITY......3 Out of Box....3 Accessories....3 Safety Information....3 Certification....5
DSM155 Digital LED Light Meter User s Manual DSM155 CONTENTS TABLE OF CONTENTS......3 LIMITED WARRANTY AND LIMITATION OF LIABILITY......3 Out of Box....3 Accessories....3 Safety Information....3 Certification....5
Energy Pathways in Earth s Atmosphere
 BRSP - 10 Page 1 Solar radiation reaching Earth s atmosphere includes a wide spectrum of wavelengths. In addition to visible light there is radiation of higher energy and shorter wavelength called ultraviolet
BRSP - 10 Page 1 Solar radiation reaching Earth s atmosphere includes a wide spectrum of wavelengths. In addition to visible light there is radiation of higher energy and shorter wavelength called ultraviolet
Luminol Test PROCESS SKILLS SCIENCE TOPICS VOCABULARY
 EXPERIMENT: LUMINOL TEST Luminol Test Visitors mix a solution of luminol with fake blood (hydrogen peroxide) to produce a reaction that gives off blue light. OBJECTIVES: Visitors learn that some chemical
EXPERIMENT: LUMINOL TEST Luminol Test Visitors mix a solution of luminol with fake blood (hydrogen peroxide) to produce a reaction that gives off blue light. OBJECTIVES: Visitors learn that some chemical
TO INVESTIGATE THE PROTECTION OF SUNCREAMS AGAINST UV RAYS
 TO INVESTIGATE THE PROTECTION OF SUNCREAMS AGAINST UV RAYS Aim: Our aim for this project is to collect data on the effectiveness of different sunscreens against UVA and UVB light, a) In natural sunlight
TO INVESTIGATE THE PROTECTION OF SUNCREAMS AGAINST UV RAYS Aim: Our aim for this project is to collect data on the effectiveness of different sunscreens against UVA and UVB light, a) In natural sunlight
THE EFFECT OF COLOUR FILTERS ON SOLAR PANELS. Katie Fitzgerald Expo Project Grade 7
 THE EFFECT OF COLOUR FILTERS ON SOLAR PANELS Katie Fitzgerald Expo Project Grade 7 OBSERVATION By using a solar light instead of electricity, one can assist in lightening the load on our environment. By
THE EFFECT OF COLOUR FILTERS ON SOLAR PANELS Katie Fitzgerald Expo Project Grade 7 OBSERVATION By using a solar light instead of electricity, one can assist in lightening the load on our environment. By
GENERAL SCIENCE LABORATORY 1110L Lab Experiment 6: Ohm s Law
 GENERAL SCIENCE LABORATORY 1110L Lab Experiment 6: Ohm s Law OBJECTIVES: To verify Ohm s law, the mathematical relationship among current, voltage or potential difference, and resistance, in a simple circuit.
GENERAL SCIENCE LABORATORY 1110L Lab Experiment 6: Ohm s Law OBJECTIVES: To verify Ohm s law, the mathematical relationship among current, voltage or potential difference, and resistance, in a simple circuit.
Studying Photosynthetic Electron Transport through the Hill Reaction
 Studying Photosynthetic Electron Transport through the Hill Reaction In this lab exercise you will learn techniques for studying electron transport. While our research system will be chloroplasts and photosynthetic
Studying Photosynthetic Electron Transport through the Hill Reaction In this lab exercise you will learn techniques for studying electron transport. While our research system will be chloroplasts and photosynthetic
STAAR Science Tutorial 30 TEK 8.8C: Electromagnetic Waves
 Name: Teacher: Pd. Date: STAAR Science Tutorial 30 TEK 8.8C: Electromagnetic Waves TEK 8.8C: Explore how different wavelengths of the electromagnetic spectrum such as light and radio waves are used to
Name: Teacher: Pd. Date: STAAR Science Tutorial 30 TEK 8.8C: Electromagnetic Waves TEK 8.8C: Explore how different wavelengths of the electromagnetic spectrum such as light and radio waves are used to
LinQ Mobile Software User s Guide
 LinQ Mobile Software User s Guide Trademarks SMART Board, LinQ and the SMART logo are either registered trademarks or trademarks of SMART Technologies Inc. Windows is either a registered trademark or a
LinQ Mobile Software User s Guide Trademarks SMART Board, LinQ and the SMART logo are either registered trademarks or trademarks of SMART Technologies Inc. Windows is either a registered trademark or a
Introduction to Infrared Thermography. Jeff Beard IT 570
 Introduction to Infrared Thermography Jeff Beard IT 570 Fall 2007 Contents Welcome...3 OBJECTIVES...3 Section 1: Infrared Thermography Basics...4 1.1 INFRARED THERMOGRAPHY TERMINOLOGY...5 PRACTICE ACTIVITY
Introduction to Infrared Thermography Jeff Beard IT 570 Fall 2007 Contents Welcome...3 OBJECTIVES...3 Section 1: Infrared Thermography Basics...4 1.1 INFRARED THERMOGRAPHY TERMINOLOGY...5 PRACTICE ACTIVITY
Chemistry 118 Laboratory University of Massachusetts Boston Beer s Law
 Name: LEARNING GOALS: Chemistry 118 Laboratory University of Massachusetts Boston Beer s Law 1. Become familiar with the concept of concentration and molarity. 2. Become familiar with making dilutions
Name: LEARNING GOALS: Chemistry 118 Laboratory University of Massachusetts Boston Beer s Law 1. Become familiar with the concept of concentration and molarity. 2. Become familiar with making dilutions
Service Desk. (Ver.Oct.2012)
 Service Desk Application (Ver.Oct.2012) Overview-1 The new Service desk (Ver.Oct.2012) is a functional upgrade over the Ticket based Helpdesk currently in-use (Ver.Jan 2012) The application has two components:
Service Desk Application (Ver.Oct.2012) Overview-1 The new Service desk (Ver.Oct.2012) is a functional upgrade over the Ticket based Helpdesk currently in-use (Ver.Jan 2012) The application has two components:
GETTING TO KNOW YOUR PRE-PAID TELSTRA WI-FI 4G
 GETTING TO KNOW YOUR PRE-PAID TELSTRA WI-FI 4G LET S GET THIS SHOW ON THE ROAD You must be excited about your brand new Pre-Paid Telstra Wi-Fi 4G. This guide will help you get connected as quickly and
GETTING TO KNOW YOUR PRE-PAID TELSTRA WI-FI 4G LET S GET THIS SHOW ON THE ROAD You must be excited about your brand new Pre-Paid Telstra Wi-Fi 4G. This guide will help you get connected as quickly and
Inking in MS Office 2013
 VIRGINIA TECH Inking in MS Office 2013 Getting Started Guide Instructional Technology Team, College of Engineering Last Updated: Fall 2013 Email tabletteam@vt.edu if you need additional assistance after
VIRGINIA TECH Inking in MS Office 2013 Getting Started Guide Instructional Technology Team, College of Engineering Last Updated: Fall 2013 Email tabletteam@vt.edu if you need additional assistance after
Select Correct USB Driver
 Select Correct USB Driver Windows often installs updated drivers automatically, and defaults to this latest version. Not all of these drivers are compatible with our software. If you are experiencing communications
Select Correct USB Driver Windows often installs updated drivers automatically, and defaults to this latest version. Not all of these drivers are compatible with our software. If you are experiencing communications
Colorimetric Determination of Iron in Vitamin Tablets
 Cautions: 6 M hydrochloric acid is corrosive. Purpose: To colorimetrically determine the mass of iron present in commercial vitamin tablets using a prepared calibration curve. Introduction: Iron is considered
Cautions: 6 M hydrochloric acid is corrosive. Purpose: To colorimetrically determine the mass of iron present in commercial vitamin tablets using a prepared calibration curve. Introduction: Iron is considered
F re Alarm Network etwo (FAN) T rou rou h Virt Vi ual u Private Network etwo (VPN) Dr. Dr Sa S mir H. Abdull--Ja J u a wad
 Fire Alarm Network (FAN) Through Virtual Private Network (VPN) By Dr. Samir H. Abdul-Jauwad Director General (Acting) Environment, Health and Safety Department July 07, 2013 1 Outline Introduction of Virtual
Fire Alarm Network (FAN) Through Virtual Private Network (VPN) By Dr. Samir H. Abdul-Jauwad Director General (Acting) Environment, Health and Safety Department July 07, 2013 1 Outline Introduction of Virtual
