Corticosteroids for COPD Exacerbation
|
|
|
- Sibyl Horn
- 9 years ago
- Views:
Transcription
1 PL Detail-Document # This PL Detail-Document gives subscribers additional insight related to the Recommendations published in PHARMACIST S LETTER / PRESCRIBER S LETTER August 2013 Corticosteroids for COPD Exacerbation Background Systemic corticosteroids are indicated for exacerbations of chronic obstructive pulmonary disease (COPD) to improve lung function and oxygenation, hasten recovery, and reduce risk of treatment failure and relapse. 1 But clinicians may question which corticosteroid should be used, at what dose and duration, and by what route. This article reviews information to help answer these questions, including a new study about duration of therapy. Which corticosteroid should be used? Guidelines specifically mention prednisone, prednisolone, and methylprednisolone for COPD exacerbations. 1,2 Prednisone is commonly used due to cost, available strengths, limited mineralocorticoid activity, intermediate duration of action, and once-daily dosing. 3 What route should be used? The oral route is preferred over intravenous administration unless the patient cannot tolerate oral intake. 1,2 What dose should be used? A dose of prednisone 25 to 50 mg once daily or its equivalent is recommended. 1,2,4 For equivalent corticosteroid doses and other practical information on corticosteroid use (including when and how to taper), see our PL Detail-Document, Using Oral Corticosteroids: a Toolbox. What duration is sufficient? A duration of ten to 14 days is recommended in international guidelines (seven to 14 days, Canadian Thoracic Society recommendations), but is not based on high-quality evidence. 1,2,4 A placebo-controlled study compared a fiveday course of steroids to a 14-day course. The first dose was methylprednisolone 40 mg IV (for ease of administration to patients in distress), followed by prednisone 40 mg once daily for four or 13 more days. Three hundred fourteen patients presenting to the emergency department with a COPD exacerbation, a 20 or greater pack-year smoking history, and no history of asthma were randomized. Most patients had severe or very severe disease. The primary outcome measure was time to next exacerbation within 180 days. Almost all randomized patients were admitted to the hospital. There was no difference between groups in exacerbation rate, time to exacerbation, survival, or need for mechanical ventilation. Five-day therapy was associated with a shorter hospital stay (eight vs nine days; p=0.04). Incidence of hyperglycemia, new or worsening hypertension, heart failure, infection, insomnia, or psychiatric symptoms was not reduced by shortterm therapy. 5 Conclusion A five-day course of prednisone 40 mg once daily is a reasonable option for COPD exacerbations [Evidence level A; high-quality RCT]. 5 Limiting the duration of individual courses of therapy may reduce side effects associated with high cumulative doses of corticosteroids (e.g., fractures). 6 Users of this PL Detail-Document are cautioned to use their own professional judgment and consult any other necessary or appropriate sources prior to making clinical judgments based on the content of this document. Our editors have researched the information with input from experts, government agencies, and national organizations. Information and internet links in this article were current as of the date of publication. Copyright 2013 by Therapeutic Research Center
2 (PL Detail-Document #290801: Page 2 of 2) Levels of Evidence In accordance with the trend towards Evidence-Based Medicine, we are citing the LEVEL OF EVIDENCE for the statements we publish. Level Definition A High-quality randomized controlled trial (RCT) High-quality meta-analysis (quantitative systematic review) B Nonrandomized clinical trial Nonquantitative systematic review Lower quality RCT Clinical cohort study Case-control study Historical control Epidemiologic study C Consensus Expert opinion D Anecdotal evidence In vitro or animal study Adapted from Siwek J, et al. How to write an evidence-based clinical review article. Am Fam Physician 2002;65: American Thoracic Society/European Respiratory Society. Standards for the diagnosis and management of patients with COPD. oc.pdf. (Accessed July 4, 2013). 3. PL Detail-Document, Using Oral Corticosteroids: a Toolbox. Pharmacist's Letter/Prescriber s Letter. May O'Donnell DE, Hernandez P, Kaplan A, et al. Canadian Thoracic Society recommendations for management of chronic obstructive pulmonary disease-2008 update-highlights for primary care. Can Respir J 2008;15(Suppl A):1A-8A. 5. Leuppi JD, Schuetz P, Bingisser R, et al. Shortterm vs conventional glucocorticoid therapy in acute exacerbations of chronic obstructive pulmonary disease: the REDUCE randomized clinical trial. JAMA 2013;309: Niewoehner DE, Erbland ML, Deupree RH, et al. Effect of systemic glucocorticoids on exacerbations of chronic obstructive pulmonary disease. N Engl J Med 1999;340: Project Leader in preparation of this PL Detail- Document: Melanie Cupp, Pharm.D., BCPS References 1. Global Strategy for Diagnosis, Management, and Prevention of COPD, Global Initiative for Chronic Obstructive Lung Disease (GOLD) _Report_2013_Feb20.pdf. (Accessed July 4, 2013). Cite this document as follows: PL Detail-Document, Corticosteroids for COPD Exacerbation. Pharmacist s Letter/Prescriber s Letter. August Evidence and Recommendations You Can Trust 3120 West March Lane, Stockton, CA ~ TEL (209) ~ FAX (209) Copyright 2013 by Therapeutic Research Center Subscribers to the Letter can get PL Detail-Documents, like this one, on any topic covered in any issue by going to or
3 Background Patients with chronic obstructive pulmonary disease (COPD) are often candidates for betablocker therapy for coronary artery disease, heart failure, and other indications. However, clinicians may be hesitant to prescribe beta-blockers in this population. The concern is that beta-blockers, especially noncardioselective agents, could cause bronchoconstriction or reduce the efficacy of beta- 2 agonists such as albuterol. But new evidence suggests beta-blockers might improve survival and decrease exacerbations in COPD patients. This article discusses beta-blocker safety in COPD, reviews the new studies, and gives practical advice regarding beta-blocker use by COPD patients in light of this new evidence of benefit. Beta-Blocker Safety in COPD Labeling for noncardioselective beta-blockers (e.g., timolol, carvedilol [e.g., Coreg], labetalol, nadolol, propranolol) generally contraindicate or warns against use in COPD. Furthermore, reviews and guidelines often consider COPD to be a contraindication to beta-blocker use based on cases of bronchospasm with use of noncardioselective agents. Cardioselective agents have 20-fold or greater affinity for beta-1 receptors than beta-2 receptors. They should therefore be safer for use in COPD. 1 A meta-analysis was performed to clarify the effect of cardioselective beta-blockers on pulmonary function in patients with asthma and COPD responsive to beta-2 agonists. Studies included in the analysis were randomized, placebo-controlled, and blinded. Outcomes examined included change in FEV1 from baseline, number of symptomatic patients, and need for inhaled beta-2 agonist rescue. Cardioselective beta-blockers studied included atenolol (Tenormin), metoprolol (e.g., Lopressor), bisoprolol (Zebeta [U.S.]), and acebutolol (Sectral; with intrinsic sympathomimetic activity PL Detail-Document # This PL Detail-Document gives subscribers additional insight related to the Recommendations published in PHARMACIST S LETTER / PRESCRIBER S LETTER August 2013 Beta-Blockers and COPD [ISA]). Other agents included but not available in the U.S. or Canada were practolol, celiprolol (with ISA), and xamoterol (with ISA). Single doses of these agents decreased FEV1 by 7.46%, but FEV1 increased 4.63% compared to placebo after administration of a beta-2 agonist. Respiratory symptoms were not increased by beta-blocker use. In the studies in which the beta-blocker was continued for days or weeks, FEV1 was not affected. For agents without ISA, FEV1 decreased by 3.22% compared to placebo (95% CI to 1.36). After beta-2 agonist use, there was a 12% increase in FEV1 compared to placebo (95% CI 4.12 to 19.87). Respiratory symptoms and need for beta-2 agonist rescue were not affected by beta-blocker use. So although betablockers may decrease FEV1 initially, with continued use, FEV1, symptoms, and rescue inhaler use are not adversely affected. In fact, response to beta-2 agonists seemed to improve compared to placebo. It is important to note that most of the patients in this study were relatively young (range 19.5 to 65.1 years of age, mean 40.1 years of age) with mild to moderate obstruction. 1 Five randomized, blinded, placebo-controlled trials published between January 1966 and May 2011 were included in a meta-analysis to examine the effect of beta-blockers on FEV1 and response to beta-2 agonists in COPD patients. 2 Betablockers used in the studies were propranolol, metoprolol, atenolol, celiprolol, and bisoprolol. Use of a nonselective beta-blocker was associated with a decrease in FEV1 of 0.14 L (p< ) and a 13.42% decrease in beta-2 agonist responsiveness (p< ). Use of cardioselective beta-blockers was associated with a decrease in FEV1 of 0.03 L (p=0.04) and had no effect on beta-2 agonist response. Three studies in this analysis actually showed an association between beta-blocker use and increased responsiveness to beta-agonists. 2 A cohort study was undertaken to examine the long-term effect of beta-blockers on mortality and Copyright 2013 by Therapeutic Research Center
4 exacerbations in COPD patients. 3 The cohort consisted of 2230 patients 45 years or older with a COPD diagnosis. The cohort was taken from the electronic medical records of 23 general medical practices in the Netherlands. The study period was from January 1, 1995, through Outcomes were mortality and first COPD exacerbation. Mean age of the patients at the start of the study was 64.8 years (SD 11.2 years). Of the cohort, 29.8% of patients received a betablocker (24.4% cardioselective). Survival rate was higher for beta-blocker users (HR 0.68, 95% CI 0.56 to 0.83). Also, risk of exacerbation was lower in beta-blocker users (HR 0.71, 95% CI 0.6 to 0.83). The hazard ratios were adjusted for confounders, including age, gender, smoking, cardiovascular diseases, diabetes, hypertension, respiratory and cardiovascular medication use, and need for pulmonologist referral. Subgroup analyses revealed that patients who required pulmonologist referral did not have a survival benefit associated with beta-blocker use. Study limitations include possible COPD misdiagnosis, which could have overestimated benefit, or incomplete correction of confounding by indication, which would have underestimated benefit. Also healthier patients may have been selected to receive a beta-blocker. 3 A retrospective cohort of 412 COPD patients from two academic primary care practices was examined to determine the effect of beta-blocker use on COPD exacerbations. 4 Of those that were beta-blocker users (n=166), 75% were taking a cardioselective agent. COPD medications were similar between groups, but beta-blocker users were more likely to have diabetes, hypertension, or cardiovascular disease. Beta-blocker users were less likely to have a COPD exacerbation than nonusers (OR 0.61, 95% CI 0.40 to 0.93). Although beta-blocker users were less likely than nonusers to experience a mild exacerbation, there was no difference between groups in regard to moderate or severe exacerbations. There was no difference in results among users of cardioselective vs nonselective agents; however, the number of patients receiving nonselective agents was small. 4 Data from a Swedish national database was used to examine the effect of cardiovascular drugs, including beta-blockers, on mortality in severe COPD. Patients started on oxygen between October 1, 2005 and June 30, 2009 were (PL Detail-Document #290801: Page 2 of 4) followed prospectively until death, withdrawal of oxygen, or December 31, Almost all patients receiving a beta-blocker (n=829) were receiving a cardioselective agent. Beta-blocker use was associated with increased mortality (HR 1.19, 95% CI 1.04 to 1.37). The negative effect of beta-blockers appeared to be highest in men, patients not receiving a long-acting betaagonist/inhaled corticosteroid, and in patients with cardiovascular disease. A strength of this study is its focus only on patients with severe COPD. Confounding by indication and inability to definitively determine adherence are weaknesses. 5 The association between beta-blocker use and hospitalization or mortality was examined in patients with left ventricular systolic dysfunction (LVSD) and COPD registered in the OPTIMIZE- HF (Organized Program to Initiate Lifesaving Treatment in Hospitalized Patients with Heart Failure) cohort (n=722). 6 About 40% of the patients were receiving a cardioselective agent, most commonly metoprolol. Among COPD patients, those using a cardioselective betablocker had a 60- to 90-day mortality or hospitalization rate similar to that of COPD patients not using a beta-blocker (43.6% vs 44.1%). Those using a noncardioselective agent actually had a lower event rate (37.7%). Sixtyday mortality rate was 6.2% in patients receiving a cardioselective beta-blocker, 5% in those receiving a nonselective beta-blocker, and 7.8% in patients receiving no beta-blocker. After adjusting for confounders, the HRs for 60-day mortality were 0.53 (0.25 to 1.13) for cardioselective agent use vs no beta-blocker, and 0.47 (0.25 to 0.89) for nonselective agent use vs no beta-blocker. HRs for 60- to 90-day mortality or hospitalization were 1.19 (0.75 to 1.87) for cardioselective agent use vs no beta-blocker and 0.89 (0.59 to 1.35) for noncardioselective agent use vs no beta-blocker. 6 Commentary Evidence suggests that COPD patients achieve a mortality benefit from beta-blocker use postmyocardial infarction and prior to major vascular surgery [Evidence level B; case-control studies]. 7,8 Beta-blockers might also benefit COPD itself [Evidence level B; cohort studies]. 3,4 The mechanism may involve treatment of diagnosed or undiagnosed concomitant disease states such as heart failure or ischemic heart disease. 3 Other Copyright 2013 by Therapeutic Research Center
5 potential mechanisms include improvement in exercise capacity (by reducing heart rate), reduction in lung inflammation (animal data), and up-regulation of pulmonary beta-receptors. 2,9 Overall, evidence supports using a betablocker in COPD patients when needed for compelling indications (e.g., ischemic heart disease, heart failure), especially those with milder COPD. Publication bias cannot be ruled out, however. 5 It may be prudent to choose a cardioselective agent (e.g., atenolol, bisoprolol, nebivolol, and to a lesser extent, metoprolol) to maximize margin of safety. 2 For example, metoprolol or bisoprolol may be safer choices than carvedilol for COPD patients with heart failure [Evidence level B; lower quality RCT]. 10 Start with a low dose and monitor patients closely for worsening COPD. Keep in mind that bisoprolol loses its cardioselectivity at 20 mg daily and metoprolol at total daily doses of over 100 mg. 11,12 Cardioselectivity could be lost if even lower doses of metoprolol are used with a 2D6 inhibitor (e.g., bupropion). 13 Users of this PL Detail-Document are cautioned to use their own professional judgment and consult any other necessary or appropriate sources prior to making clinical judgments based on the content of this document. Our editors have researched the information with input from experts, government agencies, and national organizations. Information and internet links in this article were current as of the date of publication. Levels of Evidence In accordance with the trend towards Evidence-Based Medicine, we are citing the LEVEL OF EVIDENCE for the statements we publish. Level Definition A High-quality randomized controlled trial (RCT) High-quality meta-analysis (quantitative systematic review) B Nonrandomized clinical trial Nonquantitative systematic review Lower quality RCT Clinical cohort study Case-control study Historical control Epidemiologic study C Consensus Expert opinion D Anecdotal evidence In vitro or animal study Adapted from Siwek J, et al. How to write an evidence-based clinical review article. Am Fam Physician 2002;65: (PL Detail-Document #290801: Page 3 of 4) Project Leader in preparation of this PL Detail- Document: Melanie Cupp, Pharm.D., BCPS References 1. Salpeter SR, Ormiston TM, Salpeter EE. Cardioselective beta-blockers in patients with reactive airway disease: a meta-analysis. Ann Intern Med 2002;137: Ni Y, Shi G, Wan H. Use of cardioselective B- blockers in patients with chronic obstructive pulmonary disease: a meta-analysis of randomized, placebo-controlled blinded trials. J Int Med Res 2012;40: Rutten FH, Zuithoff NP, Hak E, et al. Beta-blockers may reduce mortality and risk of exacerbations in patients with chronic obstructive pulmonary disease. Arch Intern Med 2010;170: Farland MZ, Peters CJ, Williams JD, et al. B- blocker use and incidence of chronic obstructive pulmonary disease exacerbations. Ann Pharmacother 2013;47: Ekstrom MP, Hermansson AB, Strom KE. Effects of cardiovascular drugs on mortality in severe chronic obstructive pulmonary disease. Am J Respir Crit Care Med 2013;187: Mentz RJ, Wojdyla D, Fiuzat M, et al. Association of beta-blocker use and selectivity with outcomes in patients with heart failure and chronic obstructive pulmonary disease (from OPTIMIZE-HF). Am J Cardiol 2013;111: Gottlieb SS, McCarter RJ, Vogel RA. Effect of beta-blockade on mortality among high-risk and low-risk patients after myocardial infarction. N Engl J Med 1998;339: van Gestel YR, Hoeks SE, Sin DD, et al. Impact of cardioselective beta-blockers on mortality in patients with chronic obstructive pulmonary disease and atherosclerosis. Am J Respir Crit Care Med 2008;178: Sin DD, Man SF. A curious case of beta-blockers in chronic obstructive pulmonary disease. Arch Intern Med 2010;170: Jabbour A, Macdonald PS, Keogh AM, et al. Differences between beta-blockers in patients with chronic heart failure and chronic obstructive pulmonary disease: a randomized crossover trial. Am J Coll Cardiol 2010;55: Product information for Zebeta. Duramed Pharmaceuticals, Inc. Pomona, NY November AHFS Drug Information. Metoprolol succinate/metoprolol tartrate. Bethesda, MD: American Society of Health-System Pharmacists; p Copyright 2013 by Therapeutic Research Center
6 (PL Detail-Document #290801: Page 4 of 4) 13. Product information for Lopressor. Novartis Pharmaceuticals Corporation. East Hanover, NJ January Cite this document as follows: PL Detail-Document, Beta-Blockers and COPD. Pharmacist s Letter/Prescriber s Letter. August Evidence and Recommendations You Can Trust 3120 West March Lane, Stockton, CA ~ TEL (209) ~ FAX (209) Copyright 2013 by Therapeutic Research Center Subscribers to the Letter can get PL Detail-Documents, like this one, on any topic covered in any issue by going to or
7 PL Detail-Document # This PL Detail-Document gives subscribers additional insight related to the Recommendations published in PHARMACIST S LETTER / PRESCRIBER S LETTER January 2015 Inhalers for COPD (Last modified January 2016) The following table lists inhalers available for the treatment of COPD, their role in therapy of stable disease, dosing frequency, and cost. In general, treatment starts with as-needed use of a short-acting bronchodilator and/or scheduled use of a long-acting bronchodilator. Beta-2 agonists and anticholinergics can be combined to improve efficacy and/or limit side effects. The addition of a corticosteroid should be reserved for patients with severe or very severe disease not controlled with a long-acting bronchodilator due to risks, including corticosteroid-associated pneumonia. 1 Abbreviations: COPD = chronic obstructive pulmonary disease; LABA = long-acting beta-2 agonists; LAAC = long-acting anticholinergic a. May differ from product labeling. b. Wholesale cost for 30-day supply of highest strength, of generic if available. For short-acting agents, cost is for 200 inhalations. c. Only long-acting inhalers specifically approved for COPD are included. Medication c Role in Stable COPD Dosing Frequency a Cost b Short-acting Bronchodilators Beta-2 agonists As-needed, for symptoms. 1 Four times daily, and/or asneeded 2 U.S.: Can combine beta-2 agonists and Albuterol (Salbutamol) ipratropium to improve efficacy ProAir HFA $53.02 (ProAir HFA, Proventil HFA, and/or limit side effects. 1 Proventil HFA $64.75 Ventolin HFA [U.S.]; Airomir, LABA or LAAC preferred to regular Ventolin HFA $60.79 Ventolin Diskus, Ventolin HFA, use of short-acting bronchodilator. 1 Canada: $6.48 generics [Canada]) Levalbuterol U.S.: $68.24 (Xopenex HFA [U.S. only]) Terbutaline Canada: $8.35 (Bricanyl Turbuhaler [Canada]) Anticholinergic U.S.: $ Ipratropium (Atrovent HFA) Canada: $20.44 Combination Beta-2 agonist/ Anticholinergic Albuterol/ipratropium (Combivent Respimat) Alternative to short-acting beta-2 agonist plus ipratropium as separate inhalers. Four times daily (in place of longacting bronchodilator), and/or asneeded 2 U.S.: Combivent Respimat $ Canada: Combivent Respimat $31.00 Copyright 2015 by Therapeutic Research Center
8 (PL Detail-Document #310107: Page 2 of 4) Medication c Role in Stable COPD Dosing Frequency a Cost b Long-acting Bronchodilators Long-Acting Beta-2 agonists (LABAs) First-line option for mild or moderate disease. 1 Can combine LABA and LAAC. 1 Twice daily 3 U.S.: $ Canada: $54.58 Formoterol (Foradil Aerolizer [U.S.], Foradil [Canada]) Indacaterol (Arcapta Neohaler [U.S.], Onbrez Breezhaler [Canada]) Olodaterol (Striverdi Respimat) Salmeterol (Serevent Diskus [U.S.]; Serevent Diskus, Serevent Diskhaler [Canada only]) Long-Acting Anticholinergics (LAACs) Aclidinium (Tudorza Pressair [U.S.], Tudorza Genuair [Canada]) Once daily 1 U.S.: $ Canada: $50.22 Once daily 4,14 U.S.: $ Canada: approved by Health Canada but not marketed at time of publication Twice daily 3 U.S.: $ Canada: Serevent Diskus $60.59; Serevent Diskhaler $60.59 for disks, $5.90 for Diskhaler Twice daily 5,6 U.S.: $ Canada: $57.35 Glycopyrrolate (Seebri Neohaler [U.S.], Seebri Breezhaler [Canada]) Tiotropium (Spiriva HandiHaler, Spiriva Respimat) Umeclidinium (Incruse Ellipta ) U.S.: Twice daily 26 Canada: Once daily 7 U.S.: $ Canada: $57.35 Once daily 15,16 U.S.: Spiriva HandiHaler $315.68; Respimat $ Canada: Spiriva HandiHaler $70.20; Respimat $70.20 Once daily 8,17 U.S.: $ Canada: $64.80 Copyright 2015 by Therapeutic Research Center
9 (PL Detail-Document #310107: Page 3 of 4) Medication c Role in Stable COPD Dosing Frequency a Cost b Long-acting Bronchodilators Combination LABA/LAAC First-line option for mild or moderate Formoterol/Aclidinium (Duaklir Genuair [Canada]) disease. 1 Can combine LABA and LAAC. 1 Twice daily 18 Canada: $80.03 Indacaterol/Glycopyrrolate (Utibron Neohaler [U.S.], Ultibro Breezhaler [Canada]) Olodaterol/Tiotropium (Stiolto Respimat [U.S.], Inspiolto Respimat [Canada]) Vilanterol/Umeclidinium (Anoro Ellipta) U.S.: Twice daily. 27 Canada: Once daily 9 U.S.: $ Canada: $86.84 Once daily 24,25 U.S.: $ Canada: $68.04 Once daily 10,11 U.S.: $ Canada: $87.48 Combination LABA/ Corticosteroid Formoterol/Budesonide (Symbicort [U.S.]; Symbicort Turbuhaler [Canada]) For severe or very severe COPD with frequent exacerbations despite use of LABA and/or LAAC. 1 Can combine with LAAC. 1 Twice daily 19,20 U.S.: $ Canada: $90.61 Salmeterol/Fluticasone propionate (Advair Diskus, Advair HFA [U.S.]; Advair Diskus, Advair [Canada]) Vilanterol/Fluticasone furoate (Breo Ellipta) Twice daily U.S.: $ Canada: Advair Diskus $149.39; Advair $ Once daily 12,13 U.S.: $ Canada: $ Users of this PL Detail-Document are cautioned to use their own professional judgment and consult any other necessary or appropriate sources prior to making clinical judgments based on the content of this document. Our editors have researched the information with input from experts, government agencies, and national organizations. Information and internet links in this article were current as of the date of publication. Copyright 2015 by Therapeutic Research Center
10 (PL Detail-Document #310107: Page 4 of 4) Project Leader in preparation of this PL Detail- Document: Melanie Cupp, Pharm.D., BCPS (original); Neeta Bahal O Mara, Pharm.D., BCPS (January 2015 update); Annette Murray, BScPharm (January 2016 update) References 1. Global Initiative for Chronic Obstructive Lung Disease (GOLD). Global strategy for the diagnosis, management, and prevention of chronic obstructive pulmonary disease (updated 2016). Report% pdf. (Accessed January 8, 2016). 2. American Thoracic Society/European Respiratory Society. Standards for the diagnosis and management of patients with COPD. (Last reviewed February 2015). (Accessed January 8, 2016). 3. Criner GJ, Bourbeau J, Diekemper RL, et al. Prevention of acute exacerbations of COPD: American College of Chest Physicians and Canadian Thoracic Society guideline. Chest 2015;147: Product information for Striverdi Respimat. Boehringer Ingelheim. Ridgefield, CT July Product information for Tudorza Pressair. Forest Pharmaceuticals, Inc. St. Louis, MO December Product monograph for Tudorza Genuair. Almirall Limited. Mississauga, ON L5N 8C3. July Product monograph for Seebri Breezhaler. Novartis Canada. Dorval, QC H9S 1A9. August Product information for Incruse Ellipta. GlaxoSmithKline. Research Triangle Park, NC June Product monograph for Ultibro Breezhaler. Novartis Canada. Dorval, QC H9S 1A9. August Product information for Anoro Ellipta. GlaxoSmithKline. Research Triangle Park, NC December Product monograph for Anoro Ellipta. GlaxoSmithKline. Mississauga, ON L5N 6L4. November Product information for Breo Ellipta. GlaxoSmithKline. Research Triangle Park, NC September Product monograph for Breo Ellipta. GlaxoSmithKline Inc. Mississauga, ON L5N 6L4. October Product monograph for Striverdi Respimat. Boehringer Ingelheim Canada. Burlington, ON L7L 5H4. June Product information for Spiriva Respimat. Boehringer Ingelheim. Ridgefield, CT September Product monograph for Spiriva Respimat. Boehringer Ingelheim Canada. Burlington, ON L7L 5H4. December Product monograph for Incruse Ellipta. GlaxoSmithKline. Mississauga, ON L5N 6L4. January Product monograph for Duaklir Genuair. AstraZeneca Canada. Mississauga, ON L4Y 1M4. April Product information for Symbicort. AstraZeneca. Wilmington, DE May Product monograph for Symbicort Turbuhaler. AstraZeneca Canada. Mississauga, ON L4Y 1M4. April Product information for Advair Diskus. GlaxoSmithKline. Research Triangle Park, NC July Product information for Advair HFA. GlaxoSmithKline. Research Triangle Park, NC December Product monograph for Advair, Advair Diskus. GlaxoSmithKline. Mississauga, ON L5N 6L4. July Product information for Stiolto Respimat. Boehringer Ingelheim. Ridgefield, CT June Product monograph for Inspiolto Respimat. Boehringer Ingelheim Canada. Burlington, ON L7L 5H4. May Product information for Seebri Neohaler. Novartis Pharmaceuticals Corp. East Hanover, NJ October Product information for Utibron Neohaler. Novartis Pharmaceuticals Corp. East Hanover, NJ October Cite this document as follows: PL Detail-Document, Inhalers for COPD. Pharmacist s Letter/Prescriber s Letter. January Evidence and Recommendations You Can Trust 3120 West March Lane, Stockton, CA ~ TEL (209) ~ FAX (209) Copyright 2015 by Therapeutic Research Center Subscribers to the Letter can get PL Detail-Documents, like this one, on any topic covered in any issue by going to or
11 Detail-Document # This Detail-Document accompanies the related article published in PHARMACIST S LETTER / PRESCRIBER S LETTER May 2010 ~ Volume 26 ~ Number Using Oral Corticosteroids: a Toolbox (Last update July 2013) Corticosteroids have a variety of uses. Among these are the treatment of inflammatory, autoimmune, and endocrine disorders, and symptom control in cancer patients. Regardless of the indication, a few questions commonly arise, such as which corticosteroid should be used for a given indication? How do the corticosteroids compare in regard to potency, duration of effect, and glucocorticoid vs mineralocorticoid effect? Does the corticosteroid require tapering? If so, how quickly can it be tapered? The following tables can be used to help answer these common clinical questions. Oral Corticosteroid Comparison 1,7,12,19 Drug c Equivalent Dose Duration of Action a Mineralocorticoid activity? Cortisone b 25 mg Short ++ Dexamethasone 0.75 mg Long 0 Hydrocortisone 20 mg Short ++ Methylprednisolone 4 mg Intermediate 0 Prednisolone 5 mg Intermediate (12 to 24 hours) + Prednisone b 5 mg Intermediate (12 to 24 hours) + a. Approximate duration of anti-inflammatory effect. Prednisone is comparator. b. Cortisone and prednisone are prodrugs converted in the liver to active cortisol and prednisolone, respectively. c. Fludrocortisone (Florinef) not included as it has purely mineralocorticoid effects and is therefore not considered interchangeable with above listed corticosteroids. Common Corticosteroid Indications d Indication Preferred Corticosteroid Rationale Adrenal insufficiency Hydrocortisone (plus fludrocortisone if additional mineralocorticoid activity needed) 2,3 Traditional replacement of choice. 3 Hydrocortisone provides both mineralocorticoid and glucocorticoid activity. However, prednisone or dexamethasone provide more physiologic (i.e., longer acting) glucocorticoid replacement. 3 Asthma Prednisone or prednisolone 4 Commonly used. Less mineralocorticoid activity than hydrocortisone. 4 Brain tumors Dexamethasone 5 Long history of use, ease of use, clinical comfort. 5 Chronic obstructive pulmonary disease (COPD) Methylprednisolone, prednisone 6 Mentioned in GOLD guidelines. 6 Copyright 2010 by Therapeutic Research Center Pharmacist s Letter / Prescriber s Letter ~ P.O. Box 8190, Stockton, CA ~ Phone: ~ Fax: ~
12 (Detail-Document #260507: Page 2 of 3) Common Corticosteroid Indications d Indication Preferred Corticosteroid Rationale Connective tissue and rheumatic disorders (e.g., lupus, rheumatoid arthritis) Prednisone 1,19 Well-absorbed, intermediate duration of action, cheap, variety of strengths and dosage forms. Can also use methylprednisolone or prednisolone. 1 Single morning dose mimics physiologic cortisol secretion and minimizes HPA axis suppression. However, twice-daily dosing may reduce side Nausea and vomiting (radiation, Dexamethasone chemotherapy, postoperative, pregnancy) Poison ivy/oak/sumac Prednisone taper starting with 1 mg/kg/day or 30 to 60 mg per day (0.5 mg/kg/day for children), total therapy effects and improve efficacy. 19 Most commonly used/studied. Standard regimen. 9 Do not use pre-packaged corticosteroid taper; dose too low and duration of duration 14 to 21 days. 8-10,16 therapy too short. 8 d. In general, hydrocortisone is chosen only when sodium and water retention are desired (e.g., for treatment of adrenal insufficiency). 1 Intermediate-acting agents (prednisone, prednisolone, methylprednisolone) given once daily may minimize HPA-axis suppression. 7 Prednisone is commonly used due to cost, available strengths, and clinical convention/comfort. Dexamethasone is used for certain indications based on clinical experience. Oral liquid prednisolone formulations are often used in children. Corticosteroid Tapering to Prevent Disease Flare and/or Adrenal Insufficiency e Consider Tapering If Consider Not Tapering If Treating poison ivy/oak/sumac 8,9 Course lasts less than 2 to 3 weeks (and no reason for taper such as those described in column to left). 12,14 Disease flare is of concern (e.g., autoimmune disease, rheumatoid Treating asthma or COPD flare for 1 to 2 weeks (patient not on systemic arthritis) 11 steroids prior to flare) 11 Patient frail or very ill (e.g., severe hematologic, inflammatory, or Patient being treated for allergic reaction which has resolved 12 immune disease) 12 Treating disease flare in patient taking systemic steroids prior to flare 13 Reason for stopping corticosteroid is severe corticosteroid adverse effect (weigh severity of adverse effect vs risks of discontinuation) 12 Patient has cushingoid symptoms (e.g., moon face, buffalo hump) 12 e. Necessity of tapering and tapering regimens not well-studied. Dose/duration necessary to cause significant HPA axis suppression unknown. 1 Expect some suppression with prednisone doses >7.5 mg daily (more so with >20 mg daily) for >3 weeks. 14,18 General recommendations include: (1) taper by 5% to 20% every 1 to 2 weeks, depending in underlying illness; (2) increase dose and slow taper if patient complains of flu-like symptoms or disease flare; (3) consider switching to a shorter-acting agent (prednisone), consolidating multiple daily doses to a single morning dose, and converting to alternate day dosing, (4) consider use of ACTH test to confirm adrenal recovery, (5) consider increased dose or supplemental hydrocortisone in times of stress. 1,12,15,17 Copyright 2010 by Therapeutic Research Center Pharmacist s Letter / Prescriber s Letter ~ P.O. Box 8190, Stockton, CA ~ Phone: ~ Fax: ~
13 (Detail-Document #260507: Page 3 of 3) Users of this document are cautioned to use their own professional judgment and consult any other necessary or appropriate sources prior to making clinical judgments based on the content of this document. Our editors have researched the information with input from experts, government agencies, and national organizations. Information and Internet links in this article were current as of the date of publication. Project Leader in preparation of this Detail- Document: Melanie Cupp, Pharm.D., BCPS References 1. Gong WC. Connective tissue disorders: the clinical use of corticosteroids. In: Koda-Kimble MA, Young LY, Alldredge BK, editors. Applied therapeutics: the clinical use of drugs. 9 th ed. Philadelphia, PA: Lippincott Williams & Wilkins; Bornstein SR. Predisposing factors for adrenal insufficiency. N Engl J Med 2009;360: Schneider EF. Endocrine disorders. In: Mueller BA, Bertch KE, Dunsworth D, et al., editors. Pharmacotherapy self-assessment program. 4 th ed. Kansas City, MO: American College of Clinical Pharmacy; Gibson PG. Corticosteroids-clinical applications: exacerbations of asthma in adults. Australian Prescriber 1996;19: Nahaczewski AE, Fowler SB, Hariharan S. Dexamethasone therapy in patients with brain tumors-a focus on tapering. J Neurosci Nurs 2004;36: Global Strategy for Diagnosis, Management and Prevention of COPD, Global Initiative for Chronic Obstructive Lung Disease (GOLD) Report_2013_Feb20.pdf. (Accessed July 11, 2013). 7. Chen SW. Rheumatic disorders. In: Koda-Kimble MA, Young LY, Alldredge BK, editors. Applied therapeutics: the clinical use of drugs. 9 th ed. Philadelphia, PA: Lippincott Williams & Wilkins; Brodell RT, Williams L. Taking the itch out of poison ivy. Are you prescribing the right medication? Postgrad Med 1999;106: Lee NP, Arriola ER. Poison ivy, oak, and sumac dermatitis. West J Med 1999;171: Gladman AC. Toxicodendron dermatitis: poison ivy, oak, and sumac. Wilderness Environ Med 2006;17: Richter B, Neises G, Clar C. Glucocorticoid withdrawal schemes in chronic medical disorders. A systematic review. Endocrinol Metab Clin North Am 2002;31: Alves C, Robazzi TCV, Mendonca M. Withdrawal from glucocorticoid therapy: clinical practice recommendations. J Pediatr (Rio J) 2008;84: Tapering after short courses of corticosteroids: is it necessary? Pharmacist s Letter/Prescriber s Letter 2003;19(12): Cooper MS, Stewart PM. Corticosteroid insufficiency in acutely ill patients. New Engl J Med 2003;348: Common oral medications that may need tapering. Pharmacist s Letter/Prescriber s Letter 2008;24(12): Guin JD. Treatment of toxicodendron dermatitis (poison ivy and poison oak). Skin Therapy Lett 2001;6: Firestein GS, Budd RC, Harris ED, et al, editors. Kelley s textbook of rheumatology. 8 th ed. Philadelphia, PA: W.B. Saunders Company; The Merck Manuals Online Medical Library. The Merck Manual for Healthcare Professionals. Prednisone. April sional/lexicomp/prednisone.html. (Accessed July 11, 2013). 19. Chen SW, O Callaghan R, Reta AM. Rheumatoid arthritis. In: Alldredge BK, Corelli RL, Ernst ME, et al., editors. Applied therapeutics: the clinical use of drugs. 10 th ed. Philadelphia, PA: Lippincott Williams & Wilkins; Cite this Detail-Document as follows: Using oral corticosteroids: a toolbox. Pharmacist s Letter/Prescriber s Letter 2010;26(5): Evidence and Advice You Can Trust 3120 West March Lane, P.O. Box 8190, Stockton, CA ~ TEL (209) ~ FAX (209) Copyright 2010 by Therapeutic Research Center Subscribers to Pharmacist s Letter and Prescriber s Letter can get Detail-Documents, like this one, on any topic covered in any issue by going to or
GEORGIA MEDICAID FEE-FOR-SERVICE ASTHMA and COPD AGENTS PA SUMMARY
 GEORGIA MEDICAID FEE-FOR-SERVICE ASTHMA and COPD AGENTS PA SUMMARY Preferred Anticholinergics and Combinations Atrovent HFA (ipratropium) Combivent Respimat (ipratropium/albuterol) Ipratropium neb inhalation
GEORGIA MEDICAID FEE-FOR-SERVICE ASTHMA and COPD AGENTS PA SUMMARY Preferred Anticholinergics and Combinations Atrovent HFA (ipratropium) Combivent Respimat (ipratropium/albuterol) Ipratropium neb inhalation
Before prescribing for COPD management, the patient should have had appropriate assessment, including spirometry, as per NICE guidelines.
 Formulary Guidance for Management of COPD patients Before prescribing for COPD management, the patient should have had appropriate assessment, including spirometry, as per NICE guidelines. For inhaler
Formulary Guidance for Management of COPD patients Before prescribing for COPD management, the patient should have had appropriate assessment, including spirometry, as per NICE guidelines. For inhaler
Medications for Managing COPD in Hospice Patients. Jim Joyner, PharmD, CGP Director of Clinical Operations Outcome Resources
 Medications for Managing COPD in Hospice Patients Jim Joyner, PharmD, CGP Director of Clinical Operations Outcome Resources Goal of medications in COPD Decrease symptoms and/or complications Reduce frequency
Medications for Managing COPD in Hospice Patients Jim Joyner, PharmD, CGP Director of Clinical Operations Outcome Resources Goal of medications in COPD Decrease symptoms and/or complications Reduce frequency
medicineupdate to find out more about this medicine
 medicineupdate Asking the right questions about new medicines Seretide for chronic obstructive pulmonary disease What this medicine is 1 What this medicine treats 2 Other medicines available for this condition
medicineupdate Asking the right questions about new medicines Seretide for chronic obstructive pulmonary disease What this medicine is 1 What this medicine treats 2 Other medicines available for this condition
Sponsor Novartis Pharmaceuticals
 Clinical Trial Results Database Page 1 Sponsor Novartis Pharmaceuticals Generic Drug Name Indacaterol Therapeutic Area of Trial Chronic Obstructive Pulmonary Disease (COPD) Indication studied: COPD Study
Clinical Trial Results Database Page 1 Sponsor Novartis Pharmaceuticals Generic Drug Name Indacaterol Therapeutic Area of Trial Chronic Obstructive Pulmonary Disease (COPD) Indication studied: COPD Study
Initiation and Adjustment of Insulin Regimens for Type 2 Diabetes
 PL Detail-Document #300128 This Detail-Document accompanies the related article published in PHARMACIST S LETTER / PRESCRIBER S LETTER January 2014 Initiation and Adjustment of Insulin Regimens for Type
PL Detail-Document #300128 This Detail-Document accompanies the related article published in PHARMACIST S LETTER / PRESCRIBER S LETTER January 2014 Initiation and Adjustment of Insulin Regimens for Type
Treatment of Hypertension: JNC 8 and More
 PL Detail-Document #300201 This PL Detail-Document gives subscribers additional insight related to the Recommendations published in PHARMACIST S LETTER / PRESCRIBER S LETTER February 2014 Treatment of
PL Detail-Document #300201 This PL Detail-Document gives subscribers additional insight related to the Recommendations published in PHARMACIST S LETTER / PRESCRIBER S LETTER February 2014 Treatment of
Prevention of Acute COPD exacerbations
 December 3, 2015 Prevention of Acute COPD exacerbations George Pyrgos MD 1 Disclosures No funding received for this presentation I have previously conducted clinical trials with Boehringer Ingelheim. Principal
December 3, 2015 Prevention of Acute COPD exacerbations George Pyrgos MD 1 Disclosures No funding received for this presentation I have previously conducted clinical trials with Boehringer Ingelheim. Principal
YOU VE BEEN REFERRED TO AN ASTHMA SPECIALIST...
 YOU VE BEEN REFERRED TO AN ASTHMA SPECIALIST... ...HERE S WHAT TO EXPECT You have been referred to an allergist because you have or may have asthma. The health professional who referred you wants you to
YOU VE BEEN REFERRED TO AN ASTHMA SPECIALIST... ...HERE S WHAT TO EXPECT You have been referred to an allergist because you have or may have asthma. The health professional who referred you wants you to
AECOPD: Management and Prevention
 AECOPD: Management and Prevention Neil MacIntyre MD Duke University Medical Center Durham NC AECOPD: Management and Prevention AECOPD: Definitions and impact Acute management of AECOPD Preventing AECOPD.
AECOPD: Management and Prevention Neil MacIntyre MD Duke University Medical Center Durham NC AECOPD: Management and Prevention AECOPD: Definitions and impact Acute management of AECOPD Preventing AECOPD.
PLAN OF ACTION FOR. Physician Name Signature License Date
 PLAN OF ACTION FOR Patient s copy (patient s name) I Feel Well Lignes I feel short directrices of breath: I cough up sputum daily. No Yes, colour: I cough regularly. No Yes I Feel Worse I have changes
PLAN OF ACTION FOR Patient s copy (patient s name) I Feel Well Lignes I feel short directrices of breath: I cough up sputum daily. No Yes, colour: I cough regularly. No Yes I Feel Worse I have changes
RES/006/APR16/AR. Speaker : Dr. Pither Sandy Tulak SpP
 RES/006/APR16/AR Speaker : Dr. Pither Sandy Tulak SpP Definition of Asthma (GINA 2015) Asthma is a common and potentially serious chronic disease that imposes a substantial burden on patients, their families
RES/006/APR16/AR Speaker : Dr. Pither Sandy Tulak SpP Definition of Asthma (GINA 2015) Asthma is a common and potentially serious chronic disease that imposes a substantial burden on patients, their families
Prof. Florian Gantner. Vice President Respiratory Diseases Research Boehringer Ingelheim
 Prof. Florian Gantner Vice President Respiratory Diseases Research Boehringer Ingelheim Research and Development in Practice: COPD Chronic Obstructive Pulmonary Disease (COPD) Facts Main cause of COPD
Prof. Florian Gantner Vice President Respiratory Diseases Research Boehringer Ingelheim Research and Development in Practice: COPD Chronic Obstructive Pulmonary Disease (COPD) Facts Main cause of COPD
Chronic obstructive pulmonary disease (COPD)
 Chronic obstructive pulmonary disease (COPD) Chronic obstructive pulmonary disease (COPD) is the name for a group of lung diseases including chronic bronchitis, emphysema and chronic obstructive airways
Chronic obstructive pulmonary disease (COPD) Chronic obstructive pulmonary disease (COPD) is the name for a group of lung diseases including chronic bronchitis, emphysema and chronic obstructive airways
This clinical study synopsis is provided in line with Boehringer Ingelheim s Policy on Transparency and Publication of Clinical Study Data.
 abcd Clinical Study for Public Disclosure This clinical study synopsis is provided in line with s Policy on Transparency and Publication of Clinical Study Data. The synopsis which is part of the clinical
abcd Clinical Study for Public Disclosure This clinical study synopsis is provided in line with s Policy on Transparency and Publication of Clinical Study Data. The synopsis which is part of the clinical
Medical Policy An independent licensee of the Blue Cross Blue Shield Association
 Afrezza Page 1 of 6 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: Afrezza (human insulin) Prime Therapeutics will review Prior Authorization requests Prior Authorization
Afrezza Page 1 of 6 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: Afrezza (human insulin) Prime Therapeutics will review Prior Authorization requests Prior Authorization
Quality Measures for Pharmacies
 PL Detail-Document #320101 This PL Detail-Document gives subscribers additional insight related to the Recommendations published in PHARMACIST S LETTER / PRESCRIBER S LETTER January 2016 Quality for Pharmacies
PL Detail-Document #320101 This PL Detail-Document gives subscribers additional insight related to the Recommendations published in PHARMACIST S LETTER / PRESCRIBER S LETTER January 2016 Quality for Pharmacies
COPD and Asthma Differential Diagnosis
 COPD and Asthma Differential Diagnosis Chronic Obstructive Pulmonary Disease (COPD) is the third leading cause of death in America. Learning Objectives Use tools to effectively diagnose chronic obstructive
COPD and Asthma Differential Diagnosis Chronic Obstructive Pulmonary Disease (COPD) is the third leading cause of death in America. Learning Objectives Use tools to effectively diagnose chronic obstructive
COPD Prescribing Guidelines
 South Staffordshire Area Prescribing Group COPD Prescribing Guidelines Inhaler choices in this guideline are different from previous versions produced by the APG. It is not expected patients controlled
South Staffordshire Area Prescribing Group COPD Prescribing Guidelines Inhaler choices in this guideline are different from previous versions produced by the APG. It is not expected patients controlled
COPD PROTOCOL CELLO. Leiden
 COPD PROTOCOL CELLO Leiden May 2011 1 Introduction This protocol includes an explanation of the clinical picture, diagnosis, objectives and medication of COPD. The Cello way of working can be viewed on
COPD PROTOCOL CELLO Leiden May 2011 1 Introduction This protocol includes an explanation of the clinical picture, diagnosis, objectives and medication of COPD. The Cello way of working can be viewed on
Kevin Saunders MD CCFP Rivergrove Medical Clinic Wellness Institute @ SOGH April 17 2013
 Kevin Saunders MD CCFP Rivergrove Medical Clinic Wellness Institute @ SOGH April 17 2013 Family physician with Rivergrove Medical Clinic Practice in the north end since 1985 Medical Director of the Wellness
Kevin Saunders MD CCFP Rivergrove Medical Clinic Wellness Institute @ SOGH April 17 2013 Family physician with Rivergrove Medical Clinic Practice in the north end since 1985 Medical Director of the Wellness
Doncaster & Bassetlaw Medicines Formulary
 Doncaster & Bassetlaw Medicines Formulary Section 3.2: Corticosteroids Beclometasone 50, 100 and 250micrograms/dose Clickhaler Clenil Modulite (Beclometasone CFC free) 50, 100, and 250micrograms/dose MDI
Doncaster & Bassetlaw Medicines Formulary Section 3.2: Corticosteroids Beclometasone 50, 100 and 250micrograms/dose Clickhaler Clenil Modulite (Beclometasone CFC free) 50, 100, and 250micrograms/dose MDI
A list of FDA-approved testosterone products can be found by searching for testosterone at http://www.accessdata.fda.gov/scripts/cder/drugsatfda/.
 FDA Drug Safety Communication: FDA cautions about using testosterone products for low testosterone due to aging; requires labeling change to inform of possible increased risk of heart attack and stroke
FDA Drug Safety Communication: FDA cautions about using testosterone products for low testosterone due to aging; requires labeling change to inform of possible increased risk of heart attack and stroke
Kortverk. betaagonist. Grönmarkerat är rekommenderat i första hand vid KOL resp astma för vuxna och barn >7år. Antikolinergika.
 Aerobec och Autohaler spray 50 och 100 µg/dos Anoro Ellipta 55/22 µg/dos Airflusal Forspiro 50/250 och 50/500 µg/dos Vilanterol Umeklidinium Airomir 0,1mg/dos Autohaler spray 0,1mg/dos Airsalb 0,1mg/ dos
Aerobec och Autohaler spray 50 och 100 µg/dos Anoro Ellipta 55/22 µg/dos Airflusal Forspiro 50/250 och 50/500 µg/dos Vilanterol Umeklidinium Airomir 0,1mg/dos Autohaler spray 0,1mg/dos Airsalb 0,1mg/ dos
Bronchodilators in COPD
 TSANZSRS Gold Coast 2015 Can average outcomes in COPD clinical trials guide treatment strategies? Long live the FEV1? Christine McDonald Dept of Respiratory and Sleep Medicine Austin Health Institute for
TSANZSRS Gold Coast 2015 Can average outcomes in COPD clinical trials guide treatment strategies? Long live the FEV1? Christine McDonald Dept of Respiratory and Sleep Medicine Austin Health Institute for
Understanding COPD. An educational health series from
 Understanding COPD An educational health series from Our Mission since 1899 is to heal, to discover, and to educate as a preeminent healthcare institution. We serve by providing the best integrated and
Understanding COPD An educational health series from Our Mission since 1899 is to heal, to discover, and to educate as a preeminent healthcare institution. We serve by providing the best integrated and
Stacie L. Penkova, PharmD, MHSA, BCPS Clinical Pharmacy Manager Critical Care Pharmacy Specialist Drug Information Coordinator Pharmacology Summit
 Stacie L. Penkova, PharmD, MHSA, BCPS Clinical Pharmacy Manager Critical Care Pharmacy Specialist Drug Information Coordinator Pharmacology Summit July 26, 2014 Objectives Classify asthma by severity Prescribe
Stacie L. Penkova, PharmD, MHSA, BCPS Clinical Pharmacy Manager Critical Care Pharmacy Specialist Drug Information Coordinator Pharmacology Summit July 26, 2014 Objectives Classify asthma by severity Prescribe
James F. Kravec, M.D., F.A.C.P
 James F. Kravec, M.D., F.A.C.P Chairman, Department of Internal Medicine, St. Elizabeth Health Center Chair, General Internal Medicine, Northeast Ohio Medical University Associate Medical Director, Hospice
James F. Kravec, M.D., F.A.C.P Chairman, Department of Internal Medicine, St. Elizabeth Health Center Chair, General Internal Medicine, Northeast Ohio Medical University Associate Medical Director, Hospice
Pulmonary Rehabilitation in Newark and Sherwood
 Pulmonary Rehabilitation in Newark and Sherwood With exception of smoking cessation pulmonary rehabilitation is the single most effective intervention for any patient with COPD. A Cochrane review published
Pulmonary Rehabilitation in Newark and Sherwood With exception of smoking cessation pulmonary rehabilitation is the single most effective intervention for any patient with COPD. A Cochrane review published
The patient s response to therapy within the first hour in the Emergency Room is one of the most reliable ways to predict need for hospitalization.
 Emergency Room Asthma Management Algorithm The Emergency Room Asthma Management Algorithm is to be used for any patient seen in the Emergency Room with the diagnosis of asthma. (The initial history should
Emergency Room Asthma Management Algorithm The Emergency Room Asthma Management Algorithm is to be used for any patient seen in the Emergency Room with the diagnosis of asthma. (The initial history should
ECG may be indicated for patients with cardiovascular risk factors
 eappendix A. Summary for Preoperative ECG American College of Cardiology/ American Heart Association, 2007 A1 2002 A2 European Society of Cardiology and European Society of Anaesthesiology, 2009 A3 Improvement,
eappendix A. Summary for Preoperative ECG American College of Cardiology/ American Heart Association, 2007 A1 2002 A2 European Society of Cardiology and European Society of Anaesthesiology, 2009 A3 Improvement,
Disclosures. Consultant and Speaker for Biogen Idec, TEVA Neuroscience, EMD Serrono, Mallinckrodt, Novartis, Genzyme, Accorda Therapeutics
 Mitzi Joi Williams, MD Neurologist MS Center of Atlanta, Atlanta, GA Disclosures Consultant and Speaker for Biogen Idec, TEVA Neuroscience, EMD Serrono, Mallinckrodt, Novartis, Genzyme, Accorda Therapeutics
Mitzi Joi Williams, MD Neurologist MS Center of Atlanta, Atlanta, GA Disclosures Consultant and Speaker for Biogen Idec, TEVA Neuroscience, EMD Serrono, Mallinckrodt, Novartis, Genzyme, Accorda Therapeutics
Drug therapy SHORT-ACTING BETA AGONISTS SHORT-ACTING ANTICHOLINERGICS LONG-ACTING BETA AGONISTS LONG-ACTING ANTICHOLINERGICS
 Drug therapy 6 6.1 What is the role of bronchodilators in COPD? 52 SHORT-ACTING BETA AGONISTS 6.2 How do short-acting beta agonists work? 52 6.3 What are the indications for their use? 52 6.4 What is the
Drug therapy 6 6.1 What is the role of bronchodilators in COPD? 52 SHORT-ACTING BETA AGONISTS 6.2 How do short-acting beta agonists work? 52 6.3 What are the indications for their use? 52 6.4 What is the
Clinical Guideline. Recommendation 3: For stable COPD patients with respiratory symptoms
 Clinical Guideline Diagnosis and Management of Stable Chronic Obstructive Pulmonary Disease: A Clinical Practice Guideline Update from the American College of Physicians, American College of Chest Physicians,
Clinical Guideline Diagnosis and Management of Stable Chronic Obstructive Pulmonary Disease: A Clinical Practice Guideline Update from the American College of Physicians, American College of Chest Physicians,
MEDICATION GUIDE. SYMBICORT 80/4.5 (budesonide 80 mcg and formoterol fumarate dihydrate 4.5 mcg) Inhalation Aerosol
 MEDICATION GUIDE SYMBICORT 80/4.5 (budesonide 80 mcg and formoterol fumarate dihydrate 4.5 mcg) Inhalation Aerosol SYMBICORT 160/4.5 (budesonide 160 mcg and formoterol fumarate dihydrate 4.5 mcg) Inhalation
MEDICATION GUIDE SYMBICORT 80/4.5 (budesonide 80 mcg and formoterol fumarate dihydrate 4.5 mcg) Inhalation Aerosol SYMBICORT 160/4.5 (budesonide 160 mcg and formoterol fumarate dihydrate 4.5 mcg) Inhalation
Clinical Guideline. Recommendation 3: For stable COPD patients with respiratory symptoms
 Clinical Guideline Diagnosis and Management of Stable Chronic Obstructive Pulmonary Disease: A Clinical Practice Guideline Update from the American College of Physicians, American College of Chest Physicians,
Clinical Guideline Diagnosis and Management of Stable Chronic Obstructive Pulmonary Disease: A Clinical Practice Guideline Update from the American College of Physicians, American College of Chest Physicians,
COPD. (Chronic Obstructive Pulmonary Disease) (Emphysema) (Chronic Bronchitis) Education For Our Community
 COPD (Chronic Obstructive Pulmonary Disease) (Emphysema) (Chronic Bronchitis) Education For Our Community Chronic Obstructive Pulmonary Disease (COPD) Definition Chronic obstructive pulmonary disease (COPD)
COPD (Chronic Obstructive Pulmonary Disease) (Emphysema) (Chronic Bronchitis) Education For Our Community Chronic Obstructive Pulmonary Disease (COPD) Definition Chronic obstructive pulmonary disease (COPD)
Chronic Obstructive Pulmonary Disease (COPD) Admission Order Set
 Patient Name: PHN: Page 1/1 Admit to Dr: Notified Consult: Dr: Family Dr: Precautions: Contact Droplet Enhanced Droplet Airborne - Reason: _ Code Status: Full Resuscitation or Consults: Reason: Dietician
Patient Name: PHN: Page 1/1 Admit to Dr: Notified Consult: Dr: Family Dr: Precautions: Contact Droplet Enhanced Droplet Airborne - Reason: _ Code Status: Full Resuscitation or Consults: Reason: Dietician
Management of exacerbations in chronic obstructive pulmonary disease in Primary Care
 Management of exacerbations in chronic obstructive pulmonary disease in Primary Care Acute exacerbations of chronic obstructive pulmonary disease (COPD) are associated with significant morbidity and mortality.
Management of exacerbations in chronic obstructive pulmonary disease in Primary Care Acute exacerbations of chronic obstructive pulmonary disease (COPD) are associated with significant morbidity and mortality.
Medical management of CHF: A New Class of Medication. Al Timothy, M.D. Cardiovascular Institute of the South
 Medical management of CHF: A New Class of Medication Al Timothy, M.D. Cardiovascular Institute of the South Disclosures Speakers Bureau for Amgen Background Chronic systolic congestive heart failure remains
Medical management of CHF: A New Class of Medication Al Timothy, M.D. Cardiovascular Institute of the South Disclosures Speakers Bureau for Amgen Background Chronic systolic congestive heart failure remains
Inhaled and Oral Corticosteroids
 Inhaled and Oral Corticosteroids Corticosteroids (steroids) are medicines that are used to treat many chronic diseases. Corticosteroids are very good at reducing inflammation (swelling) and mucus production
Inhaled and Oral Corticosteroids Corticosteroids (steroids) are medicines that are used to treat many chronic diseases. Corticosteroids are very good at reducing inflammation (swelling) and mucus production
Achieving Quality and Value in Chronic Care Management
 The Burden of Chronic Disease One of the greatest burdens on the US healthcare system is the rapidly growing rate of chronic disease. These statistics illustrate the scope of the problem: Nearly half of
The Burden of Chronic Disease One of the greatest burdens on the US healthcare system is the rapidly growing rate of chronic disease. These statistics illustrate the scope of the problem: Nearly half of
Irish Association for Emergency Medicine (IAEM) submission to the National COPD Strategy
 31 st Irish Association for Emergency Medicine (IAEM) submission to the National COPD Strategy 1 Introduction Chronic obstructive pulmonary disease (COPD) is an important disease for patients, the health
31 st Irish Association for Emergency Medicine (IAEM) submission to the National COPD Strategy 1 Introduction Chronic obstructive pulmonary disease (COPD) is an important disease for patients, the health
Marilyn Borkgren-Okonek, APN, CCNS, RN, MS Suburban Lung Associates, S.C. Elk Grove Village, IL
 Marilyn Borkgren-Okonek, APN, CCNS, RN, MS Suburban Lung Associates, S.C. Elk Grove Village, IL www.goldcopd.com GLOBAL INITIATIVE FOR CHRONIC OBSTRUCTIVE LUNG DISEASE GLOBAL STRATEGY FOR DIAGNOSIS, MANAGEMENT
Marilyn Borkgren-Okonek, APN, CCNS, RN, MS Suburban Lung Associates, S.C. Elk Grove Village, IL www.goldcopd.com GLOBAL INITIATIVE FOR CHRONIC OBSTRUCTIVE LUNG DISEASE GLOBAL STRATEGY FOR DIAGNOSIS, MANAGEMENT
On completion of this chapter you should be able to: discuss the stepwise approach to the pharmacological management of asthma in children
 7 Asthma Asthma is a common disease in children and its incidence has been increasing in recent years. Between 10-15% of children have been diagnosed with asthma. It is therefore a condition that pharmacists
7 Asthma Asthma is a common disease in children and its incidence has been increasing in recent years. Between 10-15% of children have been diagnosed with asthma. It is therefore a condition that pharmacists
Concept Series Paper on Disease Management
 Concept Series Paper on Disease Management Disease management is the concept of reducing health care costs and improving quality of life for individuals with chronic conditions by preventing or minimizing
Concept Series Paper on Disease Management Disease management is the concept of reducing health care costs and improving quality of life for individuals with chronic conditions by preventing or minimizing
Pathway for Diagnosing COPD
 Pathway for Diagnosing Visit 1 Registry Clients at Risk Patient presents with symptoms suggestive of Exertional breathlessness Chronic cough Regular sputum production Frequent bronchitis ; wheeze Occupational
Pathway for Diagnosing Visit 1 Registry Clients at Risk Patient presents with symptoms suggestive of Exertional breathlessness Chronic cough Regular sputum production Frequent bronchitis ; wheeze Occupational
Testosterone; What s all the hype? KRISTEN WYRICK, LTCOL,USAFR, MC USUHS, FAMILY MEDICINE JOINT BASE LANGLEY-EUSTIS
 Testosterone; What s all the hype? KRISTEN WYRICK, LTCOL,USAFR, MC USUHS, FAMILY MEDICINE JOINT BASE LANGLEY-EUSTIS The faces of Low Testosterone What your patients are seeing Pharmacy Industry Testosterone
Testosterone; What s all the hype? KRISTEN WYRICK, LTCOL,USAFR, MC USUHS, FAMILY MEDICINE JOINT BASE LANGLEY-EUSTIS The faces of Low Testosterone What your patients are seeing Pharmacy Industry Testosterone
EMS Information Bulletin- #060
 BUREAU OF EMERGENCY MEDICAL SERVICES EMS Information Bulletin- #060 DATE: October 27, 2008 SUBJECT: TO: FROM: Continuous Positive Airway Pressure for Basic Life Support Pennsylvania EMS Organizations &
BUREAU OF EMERGENCY MEDICAL SERVICES EMS Information Bulletin- #060 DATE: October 27, 2008 SUBJECT: TO: FROM: Continuous Positive Airway Pressure for Basic Life Support Pennsylvania EMS Organizations &
Glucocorticoids, Inhaled Therapeutic Class Review (TCR)
 Glucocorticoids, Inhaled Therapeutic Class Review (TCR) July 31, 2015 No part of this publication may be reproduced or transmitted in any form or by any means, electronic or mechanical, including photocopying,
Glucocorticoids, Inhaled Therapeutic Class Review (TCR) July 31, 2015 No part of this publication may be reproduced or transmitted in any form or by any means, electronic or mechanical, including photocopying,
Two Days of Dexamethasone Versus 5 Days of Prednisone in the Treatment of Acute Asthma: A Randomized Controlled Trial
 GENERAL MEDICINE/ORIGINAL RESEARCH Two Days of Dexamethasone Versus 5 Days of Prednisone in the Treatment of Acute Asthma: A Randomized Controlled Trial Joel Kravitz, MD, FRCPSC, Paul Dominici, MD, Jacob
GENERAL MEDICINE/ORIGINAL RESEARCH Two Days of Dexamethasone Versus 5 Days of Prednisone in the Treatment of Acute Asthma: A Randomized Controlled Trial Joel Kravitz, MD, FRCPSC, Paul Dominici, MD, Jacob
Compare the physiologic responses of the respiratory system to emphysema, chronic bronchitis, and asthma
 Chapter 31 Drugs Used to Treat Lower Respiratory Disease Learning Objectives Describe the physiology of respirations Compare the physiologic responses of the respiratory system to emphysema, chronic bronchitis,
Chapter 31 Drugs Used to Treat Lower Respiratory Disease Learning Objectives Describe the physiology of respirations Compare the physiologic responses of the respiratory system to emphysema, chronic bronchitis,
Standardizing the measurement of drug exposure
 Standardizing the measurement of drug exposure The ability to determine drug exposure in real-world clinical practice enables important insights for the optimal use of medicines and healthcare resources.
Standardizing the measurement of drug exposure The ability to determine drug exposure in real-world clinical practice enables important insights for the optimal use of medicines and healthcare resources.
Severe asthma Definition, epidemiology and risk factors. Mina Gaga Athens Chest Hospital
 Severe asthma Definition, epidemiology and risk factors Mina Gaga Athens Chest Hospital Difficult asthma Defined as asthma, poorly controlled in terms of chronic symptoms, with episodic exacerbations,
Severe asthma Definition, epidemiology and risk factors Mina Gaga Athens Chest Hospital Difficult asthma Defined as asthma, poorly controlled in terms of chronic symptoms, with episodic exacerbations,
Co-morbiditeit associeert met respiratoire aandoeningen louter omwille van de leeftijd. COPD patiënten hebben veel meer kans op longkanker dan rokers
 Academisch centrum huisartsgeneeskunde Pneumologie Juni 2012 Prof W Janssens Co-morbiditeit associeert met respiratoire aandoeningen louter omwille van de leeftijd. COPD patiënten hebben veel meer kans
Academisch centrum huisartsgeneeskunde Pneumologie Juni 2012 Prof W Janssens Co-morbiditeit associeert met respiratoire aandoeningen louter omwille van de leeftijd. COPD patiënten hebben veel meer kans
Tests. Pulmonary Functions
 Pulmonary Functions Tests Static lung functions volumes Dynamic lung functions volume and velocity Dynamic Tests Velocity dependent on Airway resistance Resistance of lung tissue to change in shape Dynamic
Pulmonary Functions Tests Static lung functions volumes Dynamic lung functions volume and velocity Dynamic Tests Velocity dependent on Airway resistance Resistance of lung tissue to change in shape Dynamic
5. Treatment of Asthma in Children
 Treatment of sthma in hildren 5. Treatment of sthma in hildren 5.1 Maintenance Treatment 5.1.1 rugs Inhaled Glucocorticoids. Persistent wheezing in children under the age of three can be controlled with
Treatment of sthma in hildren 5. Treatment of sthma in hildren 5.1 Maintenance Treatment 5.1.1 rugs Inhaled Glucocorticoids. Persistent wheezing in children under the age of three can be controlled with
DERBYSHIRE JOINT AREA PRESCRIBING COMMITTEE (JAPC) MANAGEMENT of Atrial Fibrillation (AF)
 DERBYSHIRE JOINT AREA PRESCRIBING COMMITTEE (JAPC) MANAGEMENT of Atrial Fibrillation (AF) Key priorities Identification and diagnosis Treatment for persistent AF Treatment for permanent AF Antithrombotic
DERBYSHIRE JOINT AREA PRESCRIBING COMMITTEE (JAPC) MANAGEMENT of Atrial Fibrillation (AF) Key priorities Identification and diagnosis Treatment for persistent AF Treatment for permanent AF Antithrombotic
HEdis Code Quick Reference Guide Disease Management Services
 HEdis Code Quick Reference Guide Disease Management Services Respiratory Conditions Appropriate Testing for Children With Pharyngitis (ages 2-18) [Commercial, Medicaid] Appropriate Treatment (no antibiotic)
HEdis Code Quick Reference Guide Disease Management Services Respiratory Conditions Appropriate Testing for Children With Pharyngitis (ages 2-18) [Commercial, Medicaid] Appropriate Treatment (no antibiotic)
Department of Surgery
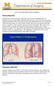 What is emphysema? 2004 Regents of the University of Michigan Emphysema is a chronic disease of the lungs characterized by thinning and overexpansion of the lung-like blisters (bullae) in the lung tissue.
What is emphysema? 2004 Regents of the University of Michigan Emphysema is a chronic disease of the lungs characterized by thinning and overexpansion of the lung-like blisters (bullae) in the lung tissue.
Omega-3 fatty acids improve the diagnosis-related clinical outcome. Critical Care Medicine April 2006;34(4):972-9
 Omega-3 fatty acids improve the diagnosis-related clinical outcome 1 Critical Care Medicine April 2006;34(4):972-9 Volume 34(4), April 2006, pp 972-979 Heller, Axel R. MD, PhD; Rössler, Susann; Litz, Rainer
Omega-3 fatty acids improve the diagnosis-related clinical outcome 1 Critical Care Medicine April 2006;34(4):972-9 Volume 34(4), April 2006, pp 972-979 Heller, Axel R. MD, PhD; Rössler, Susann; Litz, Rainer
Prior Authorization Guideline
 Prior Authorization Guideline Guideline: PDP IBT Inj - Vivitrol Therapeutic Class: Central Nervous System Agents Therapeutic Sub-Class: Opiate Antagonist Client: 2007 PDP IBT Inj Approval Date: 2/20/2007
Prior Authorization Guideline Guideline: PDP IBT Inj - Vivitrol Therapeutic Class: Central Nervous System Agents Therapeutic Sub-Class: Opiate Antagonist Client: 2007 PDP IBT Inj Approval Date: 2/20/2007
CARDIO/PULMONARY MEDICINE FOR PRIMARY CARE. Las Vegas, Nevada Bellagio March 4 6, 2016. Participating Faculty
 CARDIO/PULMONARY MEDICINE FOR PRIMARY CARE Las Vegas, Nevada Bellagio March 4 6, 2016 Participating Faculty Friday, March 4th: 7:30 am - 8:00 am Registration and Hot Breakfast 8:00 am - 9:00 am Pulmonary
CARDIO/PULMONARY MEDICINE FOR PRIMARY CARE Las Vegas, Nevada Bellagio March 4 6, 2016 Participating Faculty Friday, March 4th: 7:30 am - 8:00 am Registration and Hot Breakfast 8:00 am - 9:00 am Pulmonary
Exploratory data: COPD and blood eosinophils. David Price: 9.23-9.35am
 Exploratory data: COPD and blood eosinophils David Price: 9.23-9.35am Blood Eosinophilia in COPD The reliability and utility of blood eosinophils as a marker of disease burden, healthcare resource utilisation
Exploratory data: COPD and blood eosinophils David Price: 9.23-9.35am Blood Eosinophilia in COPD The reliability and utility of blood eosinophils as a marker of disease burden, healthcare resource utilisation
ESCMID Online Lecture Library. by author
 Do statins improve outcomes of patients with sepsis and pneumonia? Jordi Carratalà Department of Infectious Diseases Statins for sepsis & community-acquired pneumonia Sepsis and CAP are major healthcare
Do statins improve outcomes of patients with sepsis and pneumonia? Jordi Carratalà Department of Infectious Diseases Statins for sepsis & community-acquired pneumonia Sepsis and CAP are major healthcare
Medication and Devices for Chronic Obstructive Pulmonary Disease (COPD)
 Medication and Devices for Chronic Obstructive Pulmonary Disease (COPD) Patients with COPD take a wide variety of medicines to manage their symptoms these include: Inhaled Short Acting Bronchodilators
Medication and Devices for Chronic Obstructive Pulmonary Disease (COPD) Patients with COPD take a wide variety of medicines to manage their symptoms these include: Inhaled Short Acting Bronchodilators
Topic: New Treatment = Better Outcome?
 Session on COPD: Novel Concepts and Promising New Drugs Topic: New Treatment = Better Outcome? Through a CME Grant sponsored by New Treatment = Better Outcome? Tim S. Trinidad, MD Disclosure Present: COPD
Session on COPD: Novel Concepts and Promising New Drugs Topic: New Treatment = Better Outcome? Through a CME Grant sponsored by New Treatment = Better Outcome? Tim S. Trinidad, MD Disclosure Present: COPD
The largest clinical study of Bayer's Xarelto (rivaroxaban) Wednesday, 14 November 2012 07:38
 Bayer HealthCare has announced the initiation of the COMPASS study, the largest clinical study of its oral anticoagulant Xarelto (rivaroxaban) to date, investigating the prevention of major adverse cardiac
Bayer HealthCare has announced the initiation of the COMPASS study, the largest clinical study of its oral anticoagulant Xarelto (rivaroxaban) to date, investigating the prevention of major adverse cardiac
Asthma POEMs. Patient Orientated Evidence that Matters
 ASTHMA POEMs Asthma POEMs Patient Orientated Evidence that Matters Developed by the Best Practice Advocacy Centre Level 8, 10 George Street PO Box 6032 Dunedin Phone 03 4775418 Fax 03 4772622 Acknowledgement
ASTHMA POEMs Asthma POEMs Patient Orientated Evidence that Matters Developed by the Best Practice Advocacy Centre Level 8, 10 George Street PO Box 6032 Dunedin Phone 03 4775418 Fax 03 4772622 Acknowledgement
Shared Care Guideline-Use of Donepezil, Galantamine, Rivastigmine and Memantine in Dementia
 Shared Care Guideline-Use of Donepezil, Galantamine, Rivastigmine and Memantine in Dementia Version: 3.0 Ratified by: Medicines Committee Date ratified: 16 th November 2011 Name of originator/author: James
Shared Care Guideline-Use of Donepezil, Galantamine, Rivastigmine and Memantine in Dementia Version: 3.0 Ratified by: Medicines Committee Date ratified: 16 th November 2011 Name of originator/author: James
February 25, 2014. Dear Dr. Hamburg:
 February 25, 2014 Margaret A. Hamburg, M.D. Commissioner Food and Drug Administration Department of Health and Human Services WO 2200 10903 New Hampshire Avenue Silver Spring, MD 20993-0002 Division of
February 25, 2014 Margaret A. Hamburg, M.D. Commissioner Food and Drug Administration Department of Health and Human Services WO 2200 10903 New Hampshire Avenue Silver Spring, MD 20993-0002 Division of
CURRICULUM VITAE KIRK GEORGE VOELKER, M.D.
 PERSONAL INFORMATION DATE OF BIRTH December 29, 1961 PLACE OF BIRTH OFFICE ADDRESS E-MAIL Buffalo, NY. 1537 State St. 34236 (941) 330-1696 [email protected] ACADEMIC RECORD UNDERGRADUATE MEDICAL SCHOOL
PERSONAL INFORMATION DATE OF BIRTH December 29, 1961 PLACE OF BIRTH OFFICE ADDRESS E-MAIL Buffalo, NY. 1537 State St. 34236 (941) 330-1696 [email protected] ACADEMIC RECORD UNDERGRADUATE MEDICAL SCHOOL
Sponsor. Novartis Generic Drug Name. Vildagliptin. Therapeutic Area of Trial. Type 2 diabetes. Approved Indication. Investigational.
 Clinical Trial Results Database Page 1 Sponsor Novartis Generic Drug Name Vildagliptin Therapeutic Area of Trial Type 2 diabetes Approved Indication Investigational Study Number CLAF237A2386 Title A single-center,
Clinical Trial Results Database Page 1 Sponsor Novartis Generic Drug Name Vildagliptin Therapeutic Area of Trial Type 2 diabetes Approved Indication Investigational Study Number CLAF237A2386 Title A single-center,
Clinical Quality Measure Crosswalk: HEDIS, Meaningful Use, PQRS, PCMH, Beacon, 10 SOW
 Clinical Crosswalk: HEDIS, Meaningful Use, PQRS, PCMH, Beacon, 10 SOW NQF 0105 PQRS 9 NQF 0002 PQRS 66 Antidepressant Medication Management Appropriate Testing for Children with Pharyngitis (2-18 years)
Clinical Crosswalk: HEDIS, Meaningful Use, PQRS, PCMH, Beacon, 10 SOW NQF 0105 PQRS 9 NQF 0002 PQRS 66 Antidepressant Medication Management Appropriate Testing for Children with Pharyngitis (2-18 years)
Guidance to support the stepwise review of combination inhaled corticosteroid therapy for adults ( 18yrs) in asthma
 Guidance to support the stepwise review of combination inhaled corticosteroid therapy for adults ( 18yrs) in asthma Important Complete asthma control needs to be achieved for at least 12 weeks before attempting
Guidance to support the stepwise review of combination inhaled corticosteroid therapy for adults ( 18yrs) in asthma Important Complete asthma control needs to be achieved for at least 12 weeks before attempting
COPD. What is COPD? How many people have COPD in Canada? Who gets COPD?
 What is COPD? COPD stands for Chronic Obstructive Pulmonary Disease. It is a long-term lung disease that makes it difficult for air to move into and out of the lungs. COPD is used to describe a few lung
What is COPD? COPD stands for Chronic Obstructive Pulmonary Disease. It is a long-term lung disease that makes it difficult for air to move into and out of the lungs. COPD is used to describe a few lung
Acknowledgements. PAH in Children: Natural History. The Sildenafil Saga
 The Sildenafil Saga David L. Wessel, MD Executive Vice President Chief Medical Officer Ikaria Distinguished Professor of Critical Care Children s National Medical Center Washington, DC, USA Acknowledgements
The Sildenafil Saga David L. Wessel, MD Executive Vice President Chief Medical Officer Ikaria Distinguished Professor of Critical Care Children s National Medical Center Washington, DC, USA Acknowledgements
Position Statement from the Irish Thoracic Society on the treatment of Idiopathic Pulmonary Fibrosis
 BACKGROUND Position Statement from the Irish Thoracic Society on the treatment of Idiopathic Pulmonary Fibrosis Idiopathic Pulmonary Fibrosis (IPF) is a rare, chronic and fatal disease characterised by
BACKGROUND Position Statement from the Irish Thoracic Society on the treatment of Idiopathic Pulmonary Fibrosis Idiopathic Pulmonary Fibrosis (IPF) is a rare, chronic and fatal disease characterised by
How Can We Get the Best Medication History?
 How Can We Get the Best Medication History? Stephen Shalansky, Pharm.D., FCSHP Pharmacy Department, St. Paul s Hospital Faculty of Pharmaceutical Sciences, UBC How Are We Doing Now? Completeness of Medication
How Can We Get the Best Medication History? Stephen Shalansky, Pharm.D., FCSHP Pharmacy Department, St. Paul s Hospital Faculty of Pharmaceutical Sciences, UBC How Are We Doing Now? Completeness of Medication
Journal Club: Niacin in Patients with Low HDL Cholesterol Levels Receiving Intensive Statin Therapy by the AIM-HIGH Investigators
 Journal Club: Niacin in Patients with Low HDL Cholesterol Levels Receiving Intensive Statin Therapy by the AIM-HIGH Investigators Shaikha Al Naimi Doctor of Pharmacy Student College of Pharmacy Qatar University
Journal Club: Niacin in Patients with Low HDL Cholesterol Levels Receiving Intensive Statin Therapy by the AIM-HIGH Investigators Shaikha Al Naimi Doctor of Pharmacy Student College of Pharmacy Qatar University
25-hydroxyvitamin D: from bone and mineral to general health marker
 DIABETES 25 OH Vitamin D TOTAL Assay 25-hydroxyvitamin D: from bone and mineral to general health marker FOR OUTSIDE THE US AND CANADA ONLY Vitamin D Receptors Brain Heart Breast Colon Pancreas Prostate
DIABETES 25 OH Vitamin D TOTAL Assay 25-hydroxyvitamin D: from bone and mineral to general health marker FOR OUTSIDE THE US AND CANADA ONLY Vitamin D Receptors Brain Heart Breast Colon Pancreas Prostate
New Cholesterol Guidelines: Carte Blanche for Statin Overuse Rita F. Redberg, MD, MSc Professor of Medicine
 New Cholesterol Guidelines: Carte Blanche for Statin Overuse Rita F. Redberg, MD, MSc Professor of Medicine Disclosures & Relevant Relationships I have nothing to disclose No financial conflicts Editor,
New Cholesterol Guidelines: Carte Blanche for Statin Overuse Rita F. Redberg, MD, MSc Professor of Medicine Disclosures & Relevant Relationships I have nothing to disclose No financial conflicts Editor,
Measure #257 (NQF 1519): Statin Therapy at Discharge after Lower Extremity Bypass (LEB) National Quality Strategy Domain: Effective Clinical Care
 Measure #257 (NQF 1519): Statin Therapy at Discharge after Lower Extremity Bypass (LEB) National Quality Strategy Domain: Effective Clinical Care 2016 PQRS OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY
Measure #257 (NQF 1519): Statin Therapy at Discharge after Lower Extremity Bypass (LEB) National Quality Strategy Domain: Effective Clinical Care 2016 PQRS OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY
Package leaflet: Information for the patient. Bricanyl Tablets 5 mg terbutaline sulfate
 Package leaflet: Information for the patient Bricanyl Tablets 5 mg terbutaline sulfate Read all of this leaflet carefully before you start taking this medicine because it contains important information
Package leaflet: Information for the patient Bricanyl Tablets 5 mg terbutaline sulfate Read all of this leaflet carefully before you start taking this medicine because it contains important information
Inhibit terminal acid secretion from parietal cells by blocking H + /K + - ATPase pump
 Chris J. Taylor, Pharm.D., BCPS Clinical Pharmacist Phoenix VA Health Care System Review pharmacology of PPIs Discuss possible association between PPI use and development of the following: Pneumonia (community-acquired
Chris J. Taylor, Pharm.D., BCPS Clinical Pharmacist Phoenix VA Health Care System Review pharmacology of PPIs Discuss possible association between PPI use and development of the following: Pneumonia (community-acquired
SYNOPSIS. Risperidone: Clinical Study Report CR003274
 SYNOPSIS Protocol No: CR003274 Title of Study: An Open-Label, Long-Term Trial of Risperidone Long-Acting Microspheres in the Treatment of Subjects Diagnosed with Schizophrenia Coordinating Investigator:
SYNOPSIS Protocol No: CR003274 Title of Study: An Open-Label, Long-Term Trial of Risperidone Long-Acting Microspheres in the Treatment of Subjects Diagnosed with Schizophrenia Coordinating Investigator:
Longitudinal Modeling of Lung Function in Respiratory Drug Development
 Longitudinal Modeling of Lung Function in Respiratory Drug Development Fredrik Öhrn, PhD Senior Clinical Pharmacometrician Quantitative Clinical Pharmacology AstraZeneca R&D Mölndal, Sweden Outline A brief
Longitudinal Modeling of Lung Function in Respiratory Drug Development Fredrik Öhrn, PhD Senior Clinical Pharmacometrician Quantitative Clinical Pharmacology AstraZeneca R&D Mölndal, Sweden Outline A brief
PRESCRIBING FOR SMOKING CESSATION. (Adapted from the Self-Limiting Conditions Independent Study Program for Manitoba Pharmacists)
 PRESCRIBING FOR SMOKING CESSATION (Adapted from the Self-Limiting Conditions Independent Study Program for Manitoba Pharmacists) Acknowledgements The Self-Limiting Conditions Independent Study Program
PRESCRIBING FOR SMOKING CESSATION (Adapted from the Self-Limiting Conditions Independent Study Program for Manitoba Pharmacists) Acknowledgements The Self-Limiting Conditions Independent Study Program
Objectives COPD. Chronic Obstructive Pulmonary Disease (COPD) 4/19/2011
 Objectives Discuss assessment findings and treatment for: Chronic Obstructive Pulmonary Disease Bronchitis Emphysema Asthma Anaphylaxis Other respiratory issues Provide some definitions Chronic Obstructive
Objectives Discuss assessment findings and treatment for: Chronic Obstructive Pulmonary Disease Bronchitis Emphysema Asthma Anaphylaxis Other respiratory issues Provide some definitions Chronic Obstructive
This clinical study synopsis is provided in line with Boehringer Ingelheim s Policy on Transparency and Publication of Clinical Study Data.
 abcd Clinical Study for Public Disclosure This clinical study synopsis is provided in line with s Policy on Transparency and Publication of Clinical Study Data. The synopsis which is part of the clinical
abcd Clinical Study for Public Disclosure This clinical study synopsis is provided in line with s Policy on Transparency and Publication of Clinical Study Data. The synopsis which is part of the clinical
CCHCS Care Guide: Asthma
 GOALS SHORTNESS OF BREATH, WHEEZE, COUGH NIGHT TIME AWAKENINGS ACTIVITY INTERFERENCE SABA* USE FOR SYMPTOM CONTROL FEV1* OR PEAK FLOW EXACERBATIONS REQUIRING ORAL STEROIDS < 2 DAYS / WEEK 2 TIMES / MONTH
GOALS SHORTNESS OF BREATH, WHEEZE, COUGH NIGHT TIME AWAKENINGS ACTIVITY INTERFERENCE SABA* USE FOR SYMPTOM CONTROL FEV1* OR PEAK FLOW EXACERBATIONS REQUIRING ORAL STEROIDS < 2 DAYS / WEEK 2 TIMES / MONTH
New and Emerging Immunotherapies for Multiple Sclerosis: Oral Agents
 New and Emerging Immunotherapies for Multiple Sclerosis: Oral Agents William Tyor, M.D. Chief, Neurology Atlanta VA Medical Center Professor, Department of Neurology Emory University School of Medicine
New and Emerging Immunotherapies for Multiple Sclerosis: Oral Agents William Tyor, M.D. Chief, Neurology Atlanta VA Medical Center Professor, Department of Neurology Emory University School of Medicine
Prior Authorization Guideline
 Prior Authorization Guideline Guideline: CSD - Suboxone Therapeutic Class: Central Nervous System Agents Therapeutic Sub-Class: Analgesics and Antipyretics (Opiate Partial Agonists) Client: County of San
Prior Authorization Guideline Guideline: CSD - Suboxone Therapeutic Class: Central Nervous System Agents Therapeutic Sub-Class: Analgesics and Antipyretics (Opiate Partial Agonists) Client: County of San
Not All Clinical Trials Are Created Equal Understanding the Different Phases
 Not All Clinical Trials Are Created Equal Understanding the Different Phases This chapter will help you understand the differences between the various clinical trial phases and how these differences impact
Not All Clinical Trials Are Created Equal Understanding the Different Phases This chapter will help you understand the differences between the various clinical trial phases and how these differences impact
Comparison/Control Groups. Mary Foulkes, PhD Johns Hopkins University
 This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike License. Your use of this material constitutes acceptance of that license and the conditions of use of materials on this
This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike License. Your use of this material constitutes acceptance of that license and the conditions of use of materials on this
