The Management of a Global Quality System in a Central Laboratory
|
|
|
- Morris Hines
- 9 years ago
- Views:
Transcription
1 The Management of a Quality ystem in a Central aboratory Mario Zanata Manager OP dministration, Regulatory Training and rchives Rome, 4 th October 2005 genda Company Introduction Company Quality ssurance Management in a Central aboratory. 1
2 Covance Drug Development ervices stablished industry's first Central aboratory ervices business in Covance Central aboratory ervices (CC) Fully dedicated to clinical trials within the Drug development Industry. Dedicated to testing specimens, resulting and reporting data. aboratory ervices ogistics Data Management Project Management 2
3 Covance Central aboratory ocations ocation stablished Operate in 6 locations, serving all continents Indianapolis, Indiana Geneva, witzerland ingapore Zeist, Netherlands Cape Town, outh frica Johannesburg, outh frica ydney, ustralia ão Paulo, Brazil CC: a Company Organisation Responsabilities Processes nalytical Methods Combinable Data G O B H D Worldwide responsiblity for a specific department. nsures processes and procedures are harmonized. liminates local (site level) specificities and inconsistencies. Implement Processes. 3
4 CC Quality ssurance Vision upport and ensure full compliance and operational excellence of global CC activities through: Proactive Quality uditing, Quality Tracking Management and Q and Regulatory Training Programs Full Compliance to Regulatory Requirements by: everaging the understanding of compliance, quality, regulatory risk and providing direction and strategy Q Management at CC means Main Components 6: ICH GCP aboratory ccreditations OP ystem dministration uditing rchiving Quality Tracking and Management QC xternal Programs Regulatory and Q Training G O B H D Q Core Functions Quality Compliance Quality Tracking & Management Quality ssurance Operations 4
5 Quality Compliance Objective: nsures global processes are implemented, compliant to internal OPs/ystems and are adequately implemented and interpreted. Conduct audits Report findings Facilitate implementation of corrective and preventative actions. Responsible for Regulatory and Inspection preparedness. Host ponsor udits uditing Team Q uditor Training Program Quality Tracking Management (QTM) Objective: Function within Q that manages and oversees the Quality Complaint Management (CP) process for CC, ensures root cause is identified and corrective and preventative actions are implemented. Central repository that collects, tracks and trends quality issues (external customer complains and internal service failures). QTM to ensure causal factors are identified and corrective and preventative actions are implemented. QTM reports are presented and reviewed monthly with the CC xecutive Committee. 5
6 Quality ssurance Operations OPs dministration Regulatory Training rchives Quality ssurance Operations OP dministration OP dministration centralized in one site Unique OP dministration Database OPs available to users on the Intranet Centralized Communication H R D R P O N I B I I T I Heads are responsible for implementation of OPs for their departments OP Team responsible for the administration of the OP Process 6
7 Quality ssurance Operations Regulatory and Q Training Objective: nhance Q and Regulatory awareness throughout Training on Q strategic topics Trainings provided to all employees. Training Catalogue ICH GCP principles Q Introduction for new hires Q Delegates OP Process Training Files Documentation Practices Quality ssurance Operations rchives Objectives nhance rchiving requirements for: Physical security on the archives Record retention duration valuates and implements a rchiving strategy 7
8 Quality ssurance network xternal support to Q activities Q delegates iaison with departments Coordination and control of Training Files upport to the Implementation of OPs Participation with Q informational meetings Contact person between Q department and the other departments Conclusions Q Compliance QTM Organisation Responsabilities Processes Q Operations nalytical Methods Combinable Data 8
ROLES, RESPONSIBILITIES AND DELEGATION OF DUTIES IN CLINICAL TRIALS OF MEDICINAL PRODUCTS
 ROLES, RESPONSIBILITIES AND DELEGATION OF DUTIES IN CLINICAL TRIALS OF MEDICINAL PRODUCTS STANDARD OPERATING PROCEDURE NO SOP 09 DATE RATIFIED 4/7/13 NEXT REVIEW DATE 4/7/14 POLICY STATEMENT/KEY OBJECTIVES:
ROLES, RESPONSIBILITIES AND DELEGATION OF DUTIES IN CLINICAL TRIALS OF MEDICINAL PRODUCTS STANDARD OPERATING PROCEDURE NO SOP 09 DATE RATIFIED 4/7/13 NEXT REVIEW DATE 4/7/14 POLICY STATEMENT/KEY OBJECTIVES:
The Concept of Quality in Clinical Research. Dorota Śwituła Senior Clinical Quality Assurance Advisor
 The Concept of Quality in Clinical Research Dorota Śwituła Senior Clinical Quality Assurance Advisor 1 Agenda What is quality? How we define quality in clinical research? The standard components of a Quality
The Concept of Quality in Clinical Research Dorota Śwituła Senior Clinical Quality Assurance Advisor 1 Agenda What is quality? How we define quality in clinical research? The standard components of a Quality
U.S. Food and Drug Administration
 U.S. Food and Drug Administration Notice: Archived Document The content in this document is provided on the FDA s website for reference purposes only. It was current when produced, but is no longer maintained
U.S. Food and Drug Administration Notice: Archived Document The content in this document is provided on the FDA s website for reference purposes only. It was current when produced, but is no longer maintained
An Overview of Clinical Archiving. Russell Joyce Records & Information Manager
 An Overview of Clinical Archiving Russell Joyce Records & Information Manager Objectives To understand the Historical context of archiving Importance of archiving Regulatory & legal frameworks Archiving
An Overview of Clinical Archiving Russell Joyce Records & Information Manager Objectives To understand the Historical context of archiving Importance of archiving Regulatory & legal frameworks Archiving
New Investigator Collaborations and Interactions: Regulatory
 Your Health and Safety... Our priority Votre santé et votre Securité notre priorité New Investigator Collaborations and Interactions: Regulatory NCIC Clinical Trials Group New Investigator Clinical Trials
Your Health and Safety... Our priority Votre santé et votre Securité notre priorité New Investigator Collaborations and Interactions: Regulatory NCIC Clinical Trials Group New Investigator Clinical Trials
Harmonizing Change Control Processes Globally
 Quality & Compliance Associates, LLC Harmonizing Change Control Processes Globally President Quality & Compliance Associates, LLC When We Deal In A Global Environment, How Do We Design A System That Addresses
Quality & Compliance Associates, LLC Harmonizing Change Control Processes Globally President Quality & Compliance Associates, LLC When We Deal In A Global Environment, How Do We Design A System That Addresses
ASTRAZENECA GLOBAL POLICY QUALITY AND REGULATORY COMPLIANCE
 ASTRAZENECA GLOBAL POLICY QUALITY AND REGULATORY COMPLIANCE THIS POLICY OUTLINES THE TOP LEVEL REQUIREMENTS TO SUPPORT PRODUCT QUALITY IN THE DEVELOPMENT, MANUFACTURE AND DISTRIBUTION OF ACTIVE PHARMACEUTICAL
ASTRAZENECA GLOBAL POLICY QUALITY AND REGULATORY COMPLIANCE THIS POLICY OUTLINES THE TOP LEVEL REQUIREMENTS TO SUPPORT PRODUCT QUALITY IN THE DEVELOPMENT, MANUFACTURE AND DISTRIBUTION OF ACTIVE PHARMACEUTICAL
& SOFTWARE TESTING @ GSS
 CMK C FNC CN, NC & FW NG @ G C:55, M:0, :100, K:0 C:96, M:82, :0, K:0 C:91, M:52, :2, K:6 C:0, M:0, :0, K:40 C26, M:19, :18, K:4 Global oftech olutions (G) believes in delivering quality service to their
CMK C FNC CN, NC & FW NG @ G C:55, M:0, :100, K:0 C:96, M:82, :0, K:0 C:91, M:52, :2, K:6 C:0, M:0, :0, K:40 C26, M:19, :18, K:4 Global oftech olutions (G) believes in delivering quality service to their
Data Management Unit Research Institute for Health Sciences, Chiang Mai University
 Data Management Unit Research Institute for Health Sciences, Chiang Mai University Clinical Data Management is the process of handling data from clinical trials. The inherent goal of any clinical data
Data Management Unit Research Institute for Health Sciences, Chiang Mai University Clinical Data Management is the process of handling data from clinical trials. The inherent goal of any clinical data
INTERIM SITE MONITORING PROCEDURE
 INTERIM SITE MONITORING PROCEDURE 1. PURPOSE The purpose of this SOP is to describe the interim monitoring procedures conducted at Institution, according to GCP and other applicable local regulations.
INTERIM SITE MONITORING PROCEDURE 1. PURPOSE The purpose of this SOP is to describe the interim monitoring procedures conducted at Institution, according to GCP and other applicable local regulations.
This unit will provide the structure needed to achieve successful supervision and oversight of the study. We will explain the importance of an
 Music No audio Please review information shown to understand the general course navigation and available resources. Resources, such as the audio script, example forms, and website links, can be accessed
Music No audio Please review information shown to understand the general course navigation and available resources. Resources, such as the audio script, example forms, and website links, can be accessed
Presented by Rosemarie Bell 24 April 2014
 Global Good Manufacturing Practice A Comparability Study to Link Good Manufacturing Practice Standards for World Wide Compliance Within the Cellular Therapy Industry Presented by Rosemarie Bell 24 April
Global Good Manufacturing Practice A Comparability Study to Link Good Manufacturing Practice Standards for World Wide Compliance Within the Cellular Therapy Industry Presented by Rosemarie Bell 24 April
Funded by the European Union
 Funded by the uropean Union Date: 12/07/2010 Tender number 07R01/20/11 Project no. uropeaid/126969/c/r/yu Project title: Improving tructural Capacity of the erbian tatistical Office in view of approximating
Funded by the uropean Union Date: 12/07/2010 Tender number 07R01/20/11 Project no. uropeaid/126969/c/r/yu Project title: Improving tructural Capacity of the erbian tatistical Office in view of approximating
Clinical Trial Oversight: Ensuring GCP Compliance, Patient Safety and Data Integrity
 Clinical Trial Oversight: Ensuring GCP Compliance, Patient Safety and Data Integrity Michelle Quaye Regulatory Manager, Advanced Therapy Trials University College London Overview The Principles of Good
Clinical Trial Oversight: Ensuring GCP Compliance, Patient Safety and Data Integrity Michelle Quaye Regulatory Manager, Advanced Therapy Trials University College London Overview The Principles of Good
Core Training Outline
 Core Training Outline Modules: 1) Study Design 2) PI Oversight 3) Financial Management 4) Study Operations 5) Recruitment & Retention 6) Informed Consent 7) Investigational Products 8) Subject Safety 9)
Core Training Outline Modules: 1) Study Design 2) PI Oversight 3) Financial Management 4) Study Operations 5) Recruitment & Retention 6) Informed Consent 7) Investigational Products 8) Subject Safety 9)
GCP - Records Managers Association
 GCP - Records Managers Association Guidance on the Scanning and Destruction of Paper Records 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 28 29 30 31 32 33 34 The introduction
GCP - Records Managers Association Guidance on the Scanning and Destruction of Paper Records 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 28 29 30 31 32 33 34 The introduction
LIBRARY GUIDE: Clinical Medical Device
 LIBRARY GUIDE: Clinical Medical Device Table of Contents Overview... 3 Clinical Curriculum Core Program Course Descriptions: A Tour of Health Canada (PHDV89)...5 A Tour of Health Europe (PHDV90)...5 A
LIBRARY GUIDE: Clinical Medical Device Table of Contents Overview... 3 Clinical Curriculum Core Program Course Descriptions: A Tour of Health Canada (PHDV89)...5 A Tour of Health Europe (PHDV90)...5 A
The role, duties and responsibilities of clinical trials personnel Monitoring: rules and recommendations
 The role, duties and responsibilities of clinical trials personnel Monitoring: rules and recommendations Maria Luisa Paoloni OPBG Clinical & Research Services Monitoring and Responsible of monitoring:
The role, duties and responsibilities of clinical trials personnel Monitoring: rules and recommendations Maria Luisa Paoloni OPBG Clinical & Research Services Monitoring and Responsible of monitoring:
REGULATIONS FOR THE POSTGRADUATE DIPLOMA IN CLINICAL RESEARCH METHODOLOGY (PDipClinResMethodology)
 452 REGULATIONS FOR THE POSTGRADUATE DIPLOMA IN CLINICAL RESEARCH METHODOLOGY (PDipClinResMethodology) (See also General Regulations) M.57 Admission requirements To be eligible for admission to the courses
452 REGULATIONS FOR THE POSTGRADUATE DIPLOMA IN CLINICAL RESEARCH METHODOLOGY (PDipClinResMethodology) (See also General Regulations) M.57 Admission requirements To be eligible for admission to the courses
GCP Inspection of Ti Trial Master Files
 Safeguarding public health GCP Inspection of Ti Trial Master Files DIA, 12th Conference on European Electronic Document Management/ EDM Crown copyright 2011 The materials featured within these MHRA presentation
Safeguarding public health GCP Inspection of Ti Trial Master Files DIA, 12th Conference on European Electronic Document Management/ EDM Crown copyright 2011 The materials featured within these MHRA presentation
RECOMMENDATION ON THE CONTENT OF THE TRIAL MASTER FILE AND ARCHIVING
 RECOMMENDATION ON THE CONTENT OF THE TRIAL MASTER FILE AND ARCHIVING July 2006 TABLE OF CONTENTS Page 1. Introduction 2 2. Scope 2 3. Documents to be archived 2 4. Quality of essential documents 10 5.
RECOMMENDATION ON THE CONTENT OF THE TRIAL MASTER FILE AND ARCHIVING July 2006 TABLE OF CONTENTS Page 1. Introduction 2 2. Scope 2 3. Documents to be archived 2 4. Quality of essential documents 10 5.
Health Canada s GCP Compliance Program. GCP Information Sessions November 2010
 Your Health and Safety... Our priority Votre santé et votre Securité notre priorité Health Canada s GCP Compliance Program GCP Information Sessions November 2010 Objective To describe the role that Health
Your Health and Safety... Our priority Votre santé et votre Securité notre priorité Health Canada s GCP Compliance Program GCP Information Sessions November 2010 Objective To describe the role that Health
Using CITI Program Content: Good Clinical Practice (GCP)
 CONTENT AUTHOR Jaime A. Arango, EdD, CIP CITI Program at the University of Miami INTRODUCTION Like all CITI Program educational materials, the components of the Good Clinical Practice (GCP) series can
CONTENT AUTHOR Jaime A. Arango, EdD, CIP CITI Program at the University of Miami INTRODUCTION Like all CITI Program educational materials, the components of the Good Clinical Practice (GCP) series can
CLINICAL RESEARCH GENERIC TASK DESCRIPTIONS
 Purpose Purpose Purpose Primary responsibility for implementation, coordination, evaluation, communication and/or management of research studies. May involve more than one study or multiple sites within
Purpose Purpose Purpose Primary responsibility for implementation, coordination, evaluation, communication and/or management of research studies. May involve more than one study or multiple sites within
How To Improve Your Salary At The Finance Sector Union Of Ustralia
 Secure PY @ FSU Summary Proposed lassification and Pay Model: Improved Proposal During 2013/14 enterprise bargaining negotiations FSU members raised concerns with the broken pay model at. In the 2014 greement,
Secure PY @ FSU Summary Proposed lassification and Pay Model: Improved Proposal During 2013/14 enterprise bargaining negotiations FSU members raised concerns with the broken pay model at. In the 2014 greement,
ICH Q10 Pharmaceutical Quality System (PQS)
 ICH Q10 Pharmaceutical Quality System (PQS) International Conference on Harmonisation of Technical Requirements for Registration of Pharmaceuticals for Human Use ICH Q10 PQS Guideline Background Objectives
ICH Q10 Pharmaceutical Quality System (PQS) International Conference on Harmonisation of Technical Requirements for Registration of Pharmaceuticals for Human Use ICH Q10 PQS Guideline Background Objectives
Quality Management Systems
 Management ystems Introduction An organisation will benefit from establishing an effective quality management system (QM). he cornersne of a quality organisation is the concept of the cusmer and supplier
Management ystems Introduction An organisation will benefit from establishing an effective quality management system (QM). he cornersne of a quality organisation is the concept of the cusmer and supplier
STANDARD OPERATING PROCEDURE FOR RESEARCH. Management of Essential Documents and Trial Folders
 STANDARD OPERATING PROCEDURE FOR RESEARCH Management of Essential Documents and Trial Folders Author Linda Ward Author s Job Title QA Coordinator Division Department Version number 2 Ref SOP/CLN/001/2
STANDARD OPERATING PROCEDURE FOR RESEARCH Management of Essential Documents and Trial Folders Author Linda Ward Author s Job Title QA Coordinator Division Department Version number 2 Ref SOP/CLN/001/2
Excellence Through Your Client Relationship
 xcellence hrough Your Client Relationship Partnerships: Logitas is an escrow agent, partner of oftware editors and users, to design, control and preserve accurate deposits of sources. Contact: For any
xcellence hrough Your Client Relationship Partnerships: Logitas is an escrow agent, partner of oftware editors and users, to design, control and preserve accurate deposits of sources. Contact: For any
Barnett International and CHI's Inaugural Clinical Trial Oversight Summit June 4-7, 2012 Omni Parker House Boston, MA
 Barnett International and CHI's Inaugural Clinical Trial Oversight Summit June 4-7, 2012 Omni Parker House Boston, MA This presentation is the property of DynPort Vaccine Company LLC, a CSC company, and
Barnett International and CHI's Inaugural Clinical Trial Oversight Summit June 4-7, 2012 Omni Parker House Boston, MA This presentation is the property of DynPort Vaccine Company LLC, a CSC company, and
Establishment of a Quality Systems approach to Clinical Site Management An Overview of Standard Operating Procedures (SOPs)
 Establishment of a Quality Systems approach to Clinical Site Management An Overview of Standard Operating Procedures (SOPs) Cornelia Kamp, MBA Executive Director Strategic Initiatives Tim Hackett Director
Establishment of a Quality Systems approach to Clinical Site Management An Overview of Standard Operating Procedures (SOPs) Cornelia Kamp, MBA Executive Director Strategic Initiatives Tim Hackett Director
GLP vs GMP vs GCP Dominique Pifat, Ph.D., MBA The Biologics Consulting Group [email protected]
 GLP vs GMP vs GCP Dominique Pifat, Ph.D., MBA The Biologics Consulting Group [email protected] Common Misconception Good Laboratory Practices 1) A quality system concerned with the organizational process
GLP vs GMP vs GCP Dominique Pifat, Ph.D., MBA The Biologics Consulting Group [email protected] Common Misconception Good Laboratory Practices 1) A quality system concerned with the organizational process
Guidance to Research Ethics Committees on Initial Facility Assessment
 Guidance to Research Ethics Committees on Initial Facility Assessment Introduction One of the roles of a Research Ethics Committee is to provide an opinion on a proposed study, based on the suitability
Guidance to Research Ethics Committees on Initial Facility Assessment Introduction One of the roles of a Research Ethics Committee is to provide an opinion on a proposed study, based on the suitability
R&D Administration Manager. Research and Development. Research and Development
 Document Title: Document Number: Patient Recruitment SOP031 Staff involved in development: Job titles only Document author/owner: Directorate: Department: For use by: RM&G Manager, R&D Administration Manager,
Document Title: Document Number: Patient Recruitment SOP031 Staff involved in development: Job titles only Document author/owner: Directorate: Department: For use by: RM&G Manager, R&D Administration Manager,
Global regulatory affairs role in the biopharmaceutical industry
 CHAPTER TWO Global affairs role in the biopharmaceutical industry 2.1 Overview 2.2 Global affairs organization 2.3 Role of global affairs 2.4 Key functions and activities 2.5 Global strategy 2.6 Global
CHAPTER TWO Global affairs role in the biopharmaceutical industry 2.1 Overview 2.2 Global affairs organization 2.3 Role of global affairs 2.4 Key functions and activities 2.5 Global strategy 2.6 Global
16 Rankings On First Page. 30 Total Keywords. KEYWORD RANKINGS We are tracking Benchmark Date and Current Ranking. Ranking Changes Improved
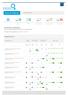 Keyword Rankings harmonygroup.co.za 0 Total Keywords 8 Rankings On First Page 6 Rankings On First Page 7 Rankings On Second Page 7 Ranking Changes Improved NA Ranking Changes Declined KEYWORD RANKINGS
Keyword Rankings harmonygroup.co.za 0 Total Keywords 8 Rankings On First Page 6 Rankings On First Page 7 Rankings On Second Page 7 Ranking Changes Improved NA Ranking Changes Declined KEYWORD RANKINGS
Essential Documents for Clinical Trial Research. Centre for Health Evaluation and Outcome Sciences Manager, Clinical Research Development Office
 Essential Documents for Clinical Trial Research Erin Cherban, MSc., CCRP Centre for Health Evaluation and Outcome Sciences Manager, Clinical Research Development Office Document Examples See the following
Essential Documents for Clinical Trial Research Erin Cherban, MSc., CCRP Centre for Health Evaluation and Outcome Sciences Manager, Clinical Research Development Office Document Examples See the following
Testing Automated Manufacturing Processes
 Testing Automated Manufacturing Processes (PLC based architecture) 1 ❶ Introduction. ❷ Regulations. ❸ CSV Automated Manufacturing Systems. ❹ PLCs Validation Methodology / Approach. ❺ Testing. ❻ Controls
Testing Automated Manufacturing Processes (PLC based architecture) 1 ❶ Introduction. ❷ Regulations. ❸ CSV Automated Manufacturing Systems. ❹ PLCs Validation Methodology / Approach. ❺ Testing. ❻ Controls
Procurement Training & Certification Catalogue 2015
 Procurement raining & Certification Catalogue 2015 tandard raining Courses Page Public Procurement, Level 2-4 Advanced Certificate in Public Procurement, Level 3-5 trategic Diploma in Public Procurement,
Procurement raining & Certification Catalogue 2015 tandard raining Courses Page Public Procurement, Level 2-4 Advanced Certificate in Public Procurement, Level 3-5 trategic Diploma in Public Procurement,
JRO RMG RSS SOP 12. Standard Operating Procedure (SOP) for Study Closedown and Archiving Royal Free site specific SOP
 Office Location: 1 st Floor Maple House 149 Tottenham Court Road London W1T 7DN Joint Research Office Tel No. 0845 1555 000 Web-sites: www.uclh.nhs.uk; www.ucl.ac.uk/jro Postal Address: UCL, Gower Street
Office Location: 1 st Floor Maple House 149 Tottenham Court Road London W1T 7DN Joint Research Office Tel No. 0845 1555 000 Web-sites: www.uclh.nhs.uk; www.ucl.ac.uk/jro Postal Address: UCL, Gower Street
QUALITY CONTROL AND QUALITY ASSURANCE IN CLINICAL RESEARCH
 QUALITY CONTROL AND QUALITY ASSURANCE IN CLINICAL RESEARCH Martin Valania, Executive Director, Corporate QA and Compliance Introduction Pharmaceutical companies recognize the benefits of carefully managing
QUALITY CONTROL AND QUALITY ASSURANCE IN CLINICAL RESEARCH Martin Valania, Executive Director, Corporate QA and Compliance Introduction Pharmaceutical companies recognize the benefits of carefully managing
NEWCASTLE CLINICAL TRIALS UNIT STANDARD OPERATING PROCEDURES
 NEWCASTLE CLINICAL TRIALS UNIT STANDARD OPERATING PROCEDURES SOP details SOP title: SOP number: SOP category: Document Control GE-003 General SOP history Version number: 01 Version date: 1 March 2013 Effective
NEWCASTLE CLINICAL TRIALS UNIT STANDARD OPERATING PROCEDURES SOP details SOP title: SOP number: SOP category: Document Control GE-003 General SOP history Version number: 01 Version date: 1 March 2013 Effective
Archiving of Research Documentation
 Suspension, Termination & Completion Standard Operating Procedure VERSION / REVISION: 2.0 EFFECTIVE DATE: 28 05 12 REVIEW DATE: 28 05 14 AUTHOR(S): CONTROLLER: APPROVED BY: Clinical Trials Manager; Recruitment
Suspension, Termination & Completion Standard Operating Procedure VERSION / REVISION: 2.0 EFFECTIVE DATE: 28 05 12 REVIEW DATE: 28 05 14 AUTHOR(S): CONTROLLER: APPROVED BY: Clinical Trials Manager; Recruitment
TEMPLATE DATA MANAGEMENT PLAN
 TEMPLATE DATA MANAGEMENT PLAN ICRIN (QM sub group) Version: XX Date: XXXXXXX Page 1 of 6 1.0 Document Ownership The Data Management Plan (DMP) will be initiated and subsequently owned by the Data Manager
TEMPLATE DATA MANAGEMENT PLAN ICRIN (QM sub group) Version: XX Date: XXXXXXX Page 1 of 6 1.0 Document Ownership The Data Management Plan (DMP) will be initiated and subsequently owned by the Data Manager
History and Principles of Good Clinical Practice
 History and Principles of Good Clinical Practice Cristina E. Torres, Ph.D. Social Science Professor, UPM-NIH FERCAP Coordinator ICH: International Conference on Harmonization GCP: Good Clinical Practices
History and Principles of Good Clinical Practice Cristina E. Torres, Ph.D. Social Science Professor, UPM-NIH FERCAP Coordinator ICH: International Conference on Harmonization GCP: Good Clinical Practices
The Global Guideline for GCP Audit
 The Global Guideline for GCP Audit Author: JSQA: Hiroyasu Yamashita, e-mail: [email protected] Reviewed and Acknowledged by: BARQA: Patricia Fitzgerald, e-mail: [email protected]
The Global Guideline for GCP Audit Author: JSQA: Hiroyasu Yamashita, e-mail: [email protected] Reviewed and Acknowledged by: BARQA: Patricia Fitzgerald, e-mail: [email protected]
Standard Operating Procedure on Training Requirements for staff participating in CTIMPs Sponsored by UCL
 Page 1 of 10 Standard Operating Procedure on Training Requirements for staff participating in CTIMPs Sponsored by UCL SOP ID Number: Effective Date:01/08/2012 Version Number & Date of Authorisation: V02,
Page 1 of 10 Standard Operating Procedure on Training Requirements for staff participating in CTIMPs Sponsored by UCL SOP ID Number: Effective Date:01/08/2012 Version Number & Date of Authorisation: V02,
gsop-32-02 - Vendor Assessment SOP page 1 of 10
 gsop-32-02 - Vendor Assessment SOP page 1 of 10 Hertfordshire Hospitals R&D Consortium Incorporating West Herts Hospitals NHS Trust and East & North Herts NHS Trust VENDOR ASSESSMENT Research & Development
gsop-32-02 - Vendor Assessment SOP page 1 of 10 Hertfordshire Hospitals R&D Consortium Incorporating West Herts Hospitals NHS Trust and East & North Herts NHS Trust VENDOR ASSESSMENT Research & Development
INTRODUCTION. This book offers a systematic, ten-step approach, from the decision to validate to
 INTRODUCTION This book offers a systematic, ten-step approach, from the decision to validate to the assessment of the validation outcome, for validating configurable off-the-shelf (COTS) computer software
INTRODUCTION This book offers a systematic, ten-step approach, from the decision to validate to the assessment of the validation outcome, for validating configurable off-the-shelf (COTS) computer software
Audit Committee Charter
 Audit Committee Charter 1. Purpose. The Audit Committee of the Board of Directors shall assist the Board in fulfilling its oversight responsibility with respect to: Abbott s accounting and financial reporting
Audit Committee Charter 1. Purpose. The Audit Committee of the Board of Directors shall assist the Board in fulfilling its oversight responsibility with respect to: Abbott s accounting and financial reporting
Product Lifecycle Management in the Medical Device Industry. An Oracle White Paper Updated January 2008
 Product Lifecycle Management in the Medical Device Industry An Oracle White Paper Updated January 2008 Product Lifecycle Management in the Medical Device Industry PLM technology ensures FDA compliance
Product Lifecycle Management in the Medical Device Industry An Oracle White Paper Updated January 2008 Product Lifecycle Management in the Medical Device Industry PLM technology ensures FDA compliance
RD SOP17 Research data management and security
 RD SOP17 Research data management and security Version Number: V2 Name of originator/author: Dr Andy Mee, R&I Manager Name of responsible committee: R&I Committee Name of executive lead: Medical Director
RD SOP17 Research data management and security Version Number: V2 Name of originator/author: Dr Andy Mee, R&I Manager Name of responsible committee: R&I Committee Name of executive lead: Medical Director
The Study Site Master File and Essential Documents
 The Study Site Master File and Essential Documents Standard Operating Procedure Office of Health and Medical Research Queensland Health SOP reference: 002 Version number: 1 Effective date: 01 June 2010
The Study Site Master File and Essential Documents Standard Operating Procedure Office of Health and Medical Research Queensland Health SOP reference: 002 Version number: 1 Effective date: 01 June 2010
Barriers and Solutions for the Expansion of Clinical Research in Latin America
 Barriers and Solutions for the Expansion of Clinical Research in Latin America A Pharmaceutical Company s Perspective E. Isola DIA S. Paulo Sep 06 Outline Regulatory Culture Ethical issues Sponsors Patients
Barriers and Solutions for the Expansion of Clinical Research in Latin America A Pharmaceutical Company s Perspective E. Isola DIA S. Paulo Sep 06 Outline Regulatory Culture Ethical issues Sponsors Patients
Regulations for Handling Samples and Laboratory Testing from R&D through Phase III Clinical Trials
 Regulations for Handling Samples and Laboratory Testing from R&D through Phase III Clinical Trials Ron Hinkel, Director Quality Systems BioReliance Inc. Patti Rossman, President Globiox Purpose Keep pace
Regulations for Handling Samples and Laboratory Testing from R&D through Phase III Clinical Trials Ron Hinkel, Director Quality Systems BioReliance Inc. Patti Rossman, President Globiox Purpose Keep pace
Reflection paper on GCP compliance in relation to trial master files (paper and/or electronic) for management, audit and inspection of clinical trials
 1 2 3 15 June 2015 EMA/INS/GCP/636736/2012 Good Clinical Practice Inspectors Working Group (GCP IWG) 4 5 6 7 Reflection paper on GCP compliance in relation to trial master files (paper and/or electronic)
1 2 3 15 June 2015 EMA/INS/GCP/636736/2012 Good Clinical Practice Inspectors Working Group (GCP IWG) 4 5 6 7 Reflection paper on GCP compliance in relation to trial master files (paper and/or electronic)
Integrated Information System for Food grains Management IISFM*
 Integrated Information ystem for Foodgrains Management IIFM Integrated Information ystem for Food grains Management IIFM* 1 Ranjna Nagpal, r ech Director, NIC, NC of Delhi [email protected], [email protected]
Integrated Information ystem for Foodgrains Management IIFM Integrated Information ystem for Food grains Management IIFM* 1 Ranjna Nagpal, r ech Director, NIC, NC of Delhi [email protected], [email protected]
Strategic HR Value Proposition
 Strategic HR Value Proposition Lise Ann Ebert, MSL, PHR, SHRM-CP Director of Human Resources The Indiana Rail Road Company Building a Road Map for Doing More with Less as a Strategic Business Partner Purpose
Strategic HR Value Proposition Lise Ann Ebert, MSL, PHR, SHRM-CP Director of Human Resources The Indiana Rail Road Company Building a Road Map for Doing More with Less as a Strategic Business Partner Purpose
Adventist HealthCare, Inc.
 IRB POLICY ON HUMAN RESEARCH PROTECTION (HRP) AND GOOD CLINICAL PRACTICE (GCP) TRAINING Collaborative Institutional Training Initiative (CITI) Requirements at Adventist Healthcare, Inc. I. Required Human
IRB POLICY ON HUMAN RESEARCH PROTECTION (HRP) AND GOOD CLINICAL PRACTICE (GCP) TRAINING Collaborative Institutional Training Initiative (CITI) Requirements at Adventist Healthcare, Inc. I. Required Human
Clinical Training Management
 Clinical Training Management Learning and Compliance for Clinical Research Helping to Fuel the Growth of CROs and Service Providers According to clinical researcher CenterWatch, the FDA and global regulatory
Clinical Training Management Learning and Compliance for Clinical Research Helping to Fuel the Growth of CROs and Service Providers According to clinical researcher CenterWatch, the FDA and global regulatory
Information Governance Policy
 Information Governance Policy Version: 4 Bodies consulted: Caldicott Guardian, IM&T Directors Approved by: MT Date Approved: 27/10/2015 Lead Manager: Governance Manager Responsible Director: SIRO Date
Information Governance Policy Version: 4 Bodies consulted: Caldicott Guardian, IM&T Directors Approved by: MT Date Approved: 27/10/2015 Lead Manager: Governance Manager Responsible Director: SIRO Date
ENTERPRISE RISK MANAGEMENT POLICY
 ENTERPRISE RISK MANAGEMENT POLICY TABLE OF CONTENTS 1. Purpose... 33 2. Scope... 33 3. Policy... 33 4. Procedure... 33 5. Responsibility and Authority... 44 6. Review of Policy... 55 7. Availability of
ENTERPRISE RISK MANAGEMENT POLICY TABLE OF CONTENTS 1. Purpose... 33 2. Scope... 33 3. Policy... 33 4. Procedure... 33 5. Responsibility and Authority... 44 6. Review of Policy... 55 7. Availability of
Document Management Solution (EDMS)
 Implementing TrackWise with an Electronic Document Management Solution (EDMS) Alex Kotikovsky Product Manager Sparta Systems Gilad Kigel Manager, Solutions Consulting Sparta Systems Agenda Document Management
Implementing TrackWise with an Electronic Document Management Solution (EDMS) Alex Kotikovsky Product Manager Sparta Systems Gilad Kigel Manager, Solutions Consulting Sparta Systems Agenda Document Management
Annex A. Levels 1 5 of the Clinical Research Coordinator Track in the Clinical Research WSQ Framework
 Annex A Levels 1 5 of the Clinical Research Coordinator Track in the Clinical Research WSQ Framework WSQ Higher Certificate in Clinical Research (Clinical Research Coordinators) Apply Drug Development
Annex A Levels 1 5 of the Clinical Research Coordinator Track in the Clinical Research WSQ Framework WSQ Higher Certificate in Clinical Research (Clinical Research Coordinators) Apply Drug Development
CONTROLLED DOCUMENT- DO NOT COPY STANDARD OPERATING PROCEDURE. STH Investigator
 Research Department STANDARD OPERATING PROCEDURE STH Investigator Archiving of Essential Documentation Generated During Clinical Research SOP Number A127 Version Number V1.3 Effective Date Author Zoe Whiteley
Research Department STANDARD OPERATING PROCEDURE STH Investigator Archiving of Essential Documentation Generated During Clinical Research SOP Number A127 Version Number V1.3 Effective Date Author Zoe Whiteley
Good Clinical Practices Its origins. GCP Information Sessions November 2010
 Your Health and Safety... Our priority Votre santé et votre Securité notre priorité Good Clinical Practices Its origins GCP Information Sessions November 2010 Objective To briefly describe the evolution
Your Health and Safety... Our priority Votre santé et votre Securité notre priorité Good Clinical Practices Its origins GCP Information Sessions November 2010 Objective To briefly describe the evolution
Quality Management in Clinical Trials
 CLINICAL CASE STUDY SERIES Quality Management in Clinical Trials Clinical trials are conducted to collect the data necessary to provide information for academia, industry, and regulators to make decisions
CLINICAL CASE STUDY SERIES Quality Management in Clinical Trials Clinical trials are conducted to collect the data necessary to provide information for academia, industry, and regulators to make decisions
Comparing GCP Requirements for Medical Device Clinical Trials in the US and Japan
 REGULATORY MANAGER Comparing GCP Requirements for Medical Device Clinical Trials in the US and Japan By Harmonization-by-Doing Working Group 4 Introduction The convergence of US and Japanese medical device
REGULATORY MANAGER Comparing GCP Requirements for Medical Device Clinical Trials in the US and Japan By Harmonization-by-Doing Working Group 4 Introduction The convergence of US and Japanese medical device
Business Plan 2015-2017
 Business Plan 2015-2017 safetycodes.ab.ca Table of Contents Introduction 4 Strategic Priorities 5 Organizational Structure 7 Business Plan Goals 8 2015 Financials 13 2015-2017 Business Plan Page 3 Introduction
Business Plan 2015-2017 safetycodes.ab.ca Table of Contents Introduction 4 Strategic Priorities 5 Organizational Structure 7 Business Plan Goals 8 2015 Financials 13 2015-2017 Business Plan Page 3 Introduction
CONTROLLED DOCUMENT. Uncontrolled Copy. RDS014 Research Related Archiving. University Hospitals Birmingham NHS Foundation Trust
 University Hospitals Birmingham NHS Foundation Trust CONTROLLED DOCUMENT RDS014 Research Related Archiving CATEGORY: CLASSIFICATION: PURPOSE Controlled Document Number: Version Number: 1 Controlled Document
University Hospitals Birmingham NHS Foundation Trust CONTROLLED DOCUMENT RDS014 Research Related Archiving CATEGORY: CLASSIFICATION: PURPOSE Controlled Document Number: Version Number: 1 Controlled Document
Regulatory Writing Clinical Project Management Strategic Communications
 Regulatory Writing Clinical Project Management Strategic Communications FLEXIBLE. STRATEGIC. INFORMED. COMPLIANT. For over 15 years, Niche Science and Technology has been providing outstanding medical
Regulatory Writing Clinical Project Management Strategic Communications FLEXIBLE. STRATEGIC. INFORMED. COMPLIANT. For over 15 years, Niche Science and Technology has been providing outstanding medical
How Industry Can Partner with FDA in Defining a Risk- Based Monitoring Program
 How Industry Can Partner with FDA in Defining a Risk- Based Monitoring Program Presenter: Jan Holladay Pierre, MPH Quality Principal Leader DynPort Vaccine Company LLC, A CSC Company This presentation
How Industry Can Partner with FDA in Defining a Risk- Based Monitoring Program Presenter: Jan Holladay Pierre, MPH Quality Principal Leader DynPort Vaccine Company LLC, A CSC Company This presentation
DAIDS Bethesda, MD USA. POLICY Requirements for Clinical Quality Management Plans
 CHANGE SUMMARY: This policy and associated appendices have been reviewed for accuracy and updated to meet 508 compliance guidelines. Key changes to the policy require all sites to submit a bi-annual Clinical
CHANGE SUMMARY: This policy and associated appendices have been reviewed for accuracy and updated to meet 508 compliance guidelines. Key changes to the policy require all sites to submit a bi-annual Clinical
The anglo american Safety way. Safety Management System Standards
 The anglo american Safety way Safety Management System Standards 2 The Anglo American Safety Way CONTENTS Introduction 04 Anglo American Safety Framework 05 Safety in anglo american 06 Monitoring and review
The anglo american Safety way Safety Management System Standards 2 The Anglo American Safety Way CONTENTS Introduction 04 Anglo American Safety Framework 05 Safety in anglo american 06 Monitoring and review
OECD Series on Principles of GLP and Compliance Monitoring Number 8 (Revised)
 Unclassified ENV/JM/MONO(99)24 ENV/JM/MONO(99)24 Or. Eng. Unclassified Organisation de Coopération et de Développement Economiques OLIS : 14-Sep-1999 Organisation for Economic Co-operation and Development
Unclassified ENV/JM/MONO(99)24 ENV/JM/MONO(99)24 Or. Eng. Unclassified Organisation de Coopération et de Développement Economiques OLIS : 14-Sep-1999 Organisation for Economic Co-operation and Development
ICH Q 10 Pharmaceutical Quality System PQS DIA China Annual Meeting, May 2010
 ICH Q 10 Pharmaceutical Quality System PQS DIA China Annual Meeting, May 2010 Joseph C. Famulare Head of External Relations and Collaboration Pharma Global Technical Operations Global Quality, F. Hoffmann-La
ICH Q 10 Pharmaceutical Quality System PQS DIA China Annual Meeting, May 2010 Joseph C. Famulare Head of External Relations and Collaboration Pharma Global Technical Operations Global Quality, F. Hoffmann-La
Good Clinical Practice 101: An Introduction
 Good Clinical Practice 101: An Introduction Presented by: Lester Jao Lacorte, MD Medical Officer Commissioner s Fellow Division of Bioresearch Monitoring Office of Compliance Center for Devices and Radiological
Good Clinical Practice 101: An Introduction Presented by: Lester Jao Lacorte, MD Medical Officer Commissioner s Fellow Division of Bioresearch Monitoring Office of Compliance Center for Devices and Radiological
No. 706. Page 1 of 5. Issue Date 4/21/2014
 Subject: ROUTINE MONITORING VISITS Standard Operating Procedure Prepared By: Beaumont Research Coordinating Center, Research Institute PURPOSE Prior Issue Date 8/15/2011 No. 706 Issue Date 4/21/2014 Page
Subject: ROUTINE MONITORING VISITS Standard Operating Procedure Prepared By: Beaumont Research Coordinating Center, Research Institute PURPOSE Prior Issue Date 8/15/2011 No. 706 Issue Date 4/21/2014 Page
DATA MANAGEMENT IN CLINICAL TRIALS: GUIDELINES FOR RESEARCHERS
 Reference Number: UHB 139 Version Number: 2 Date of Next Review: 14 Apr 2018 Previous Trust/LHB Reference Number: N/A DATA MANAGEMENT IN CLINICAL TRIALS: GUIDELINES FOR RESEARCHERS Introduction and Aim
Reference Number: UHB 139 Version Number: 2 Date of Next Review: 14 Apr 2018 Previous Trust/LHB Reference Number: N/A DATA MANAGEMENT IN CLINICAL TRIALS: GUIDELINES FOR RESEARCHERS Introduction and Aim
SUBJECT: FRAUD AND ABUSE POLICY: CP 6018
 SUBJECT: FRAUD AND ABUSE POLICY: Department of Origin: Compliance & Audit Responsible Position: Vice President of Compliance and Audit Date(s) of Review and Revision: 07/10; 04/11; 11/11; 02/12; 6/12;
SUBJECT: FRAUD AND ABUSE POLICY: Department of Origin: Compliance & Audit Responsible Position: Vice President of Compliance and Audit Date(s) of Review and Revision: 07/10; 04/11; 11/11; 02/12; 6/12;
LIBRARY GUIDE: Clinical Pharmaceutical
 LIBRARY GUIDE: Clinical Pharmaceutical Table of Contents Overview...2 Course Descriptions Core Knowledge: A Tour of the FDA (PHDV60)...4 A Tour of Health Canada (PHDV89)...4 A Tour of Health Europe (PHDV90)...4
LIBRARY GUIDE: Clinical Pharmaceutical Table of Contents Overview...2 Course Descriptions Core Knowledge: A Tour of the FDA (PHDV60)...4 A Tour of Health Canada (PHDV89)...4 A Tour of Health Europe (PHDV90)...4
Needs, Providing Solutions
 Identifying Needs, Providing Solutions 1 I n d u s t r y The growth of medical research and the countless innovations coming from the pharmaceutical, biotechnology and medical device industry, has improved
Identifying Needs, Providing Solutions 1 I n d u s t r y The growth of medical research and the countless innovations coming from the pharmaceutical, biotechnology and medical device industry, has improved
Clinical Research Nurse (CRN)
 1 JOB IDENTIFICATION Job title: Responsible to: Department: Division/directorate: Job reference no.: No. of CRNs: Last update: (CRN) Senior Research Nurse Consultant/ Unit Manager /PI Add local specifications
1 JOB IDENTIFICATION Job title: Responsible to: Department: Division/directorate: Job reference no.: No. of CRNs: Last update: (CRN) Senior Research Nurse Consultant/ Unit Manager /PI Add local specifications
ORACLE AGILE PLM FOR THE MEDICAL DEVICE INDUSTRY
 ORACLE AGILE PLM FOR THE MEDICAL DEVICE INDUSTRY Enterprise PLM is a strategic approach to managing the lifecycle of a product throughout its full value chain: from initial requirements gathering through
ORACLE AGILE PLM FOR THE MEDICAL DEVICE INDUSTRY Enterprise PLM is a strategic approach to managing the lifecycle of a product throughout its full value chain: from initial requirements gathering through
STANDARD OPERATING PROCEDURE FOR DATA RETENTION
 STANDARD OPERATING PROCEDURE FOR DATA RETENTION SOP Number: GCP DM 505.02 Version Number & Date: 2 nd Version; 25 Apr 2013 Superseded Version Number & Date (if applicable): 1 st Version; 17 June 2010 Effective
STANDARD OPERATING PROCEDURE FOR DATA RETENTION SOP Number: GCP DM 505.02 Version Number & Date: 2 nd Version; 25 Apr 2013 Superseded Version Number & Date (if applicable): 1 st Version; 17 June 2010 Effective
TrackWise - Quality Management System
 TrackWise - Quality Management System Focus area: Electronic Management of CAPA Systems in the Regulated Industry May 11, 2007 Yaniv Vardi VP, Operations Sparta Systems Europe, Ltd. Agenda Sparta Systems
TrackWise - Quality Management System Focus area: Electronic Management of CAPA Systems in the Regulated Industry May 11, 2007 Yaniv Vardi VP, Operations Sparta Systems Europe, Ltd. Agenda Sparta Systems
Essential Conditions and Standards for Initial Registration
 Essential Conditions and Standards for Initial Registration Registering Bodies Australian Skills Quality Authority Tel: 1300 701 801 www.asqa.gov.au Victoria Victorian Registration and Qualifications Authority
Essential Conditions and Standards for Initial Registration Registering Bodies Australian Skills Quality Authority Tel: 1300 701 801 www.asqa.gov.au Victoria Victorian Registration and Qualifications Authority
Health Canada s GCP Compliance Program Part 2. GCP Information Sessions November 2010
 Your Health and Safety... Our priority Votre santé et votre Securité notre priorité Health Canada s GCP Compliance Program Part 2 GCP Information Sessions November 2010 Inspection Program Main objectives
Your Health and Safety... Our priority Votre santé et votre Securité notre priorité Health Canada s GCP Compliance Program Part 2 GCP Information Sessions November 2010 Inspection Program Main objectives
How To Write A Pca Dss Compliance Solution For Gameplan Group Ltd
 PCI Compliance reporting solution This document describes GamePlan s PCI DSS compliance solution and its ability to assist organisations to be compliant with the regulatory requirements of the Payment
PCI Compliance reporting solution This document describes GamePlan s PCI DSS compliance solution and its ability to assist organisations to be compliant with the regulatory requirements of the Payment
Job Description. 5. To ensure recruitment, retention and achievement of learners are fully supported and facilitated.
 Job Description Post Number: Job Title: Responsible to: Responsible for: TBC Curriculum Leader Principal Performance and management of a team of tutors/lecturers within the curriculum area in agreement
Job Description Post Number: Job Title: Responsible to: Responsible for: TBC Curriculum Leader Principal Performance and management of a team of tutors/lecturers within the curriculum area in agreement
2016 ICRM CRM Exam Prep Product Material. Part 6: Sample Business Case
 2016 ICRM CRM Exam Prep Product Material Part 6: Sample Business Case Pharma Testing Company (PTC), Inc. Topic: Comprehensive Records Management Subtopics: Compliance, Litigation, Storage, Record Retention
2016 ICRM CRM Exam Prep Product Material Part 6: Sample Business Case Pharma Testing Company (PTC), Inc. Topic: Comprehensive Records Management Subtopics: Compliance, Litigation, Storage, Record Retention
