Deep Brain Stimulation for Essential Tremor and Parkinson s Disease (NCD )
|
|
|
- Cathleen Haynes
- 10 years ago
- Views:
Transcription
1 Policy Number Reimbursement Policy Approved By UnitedHealthcare Medicare Reimbursement Policy Committee Current Approval Date 06/10/2015 IMPORTANT NOTE ABOUT THIS REIMBURSEMENT POLICY This policy is applicable to UnitedHealthcare Medicare Advantage Plans offered by UnitedHealthcare and its affiliates. You are responsible for submission of accurate claims. This reimbursement policy is intended to ensure that you are reimbursed based on the code or codes that correctly describe the health care services provided. UnitedHealthcare reimbursement policies use Current Procedural Terminology (CPT *), Centers for Medicare and Medicaid Services (CMS), or other coding guidelines. References to CPT or other sources are for definitional purposes only and do not imply any right to reimbursement. This reimbursement policy applies to all health care services billed on CMS 1500 forms and, when specified, to those billed on UB04 forms (CMS 1450). Coding methodology, industry-standard reimbursement logic, regulatory requirements, benefits design and other factors are considered in developing reimbursement policy. This information is intended to serve only as a general resource regarding UnitedHealthcare s reimbursement policy for the services described and is not intended to address every aspect of a reimbursement situation. Accordingly, UnitedHealthcare may use reasonable discretion in interpreting and applying this policy to health care services provided in a particular case. Further, the policy does not address all issues related to reimbursement for health care services provided to UnitedHealthcare enrollees. Other factors affecting reimbursement may supplement, modify, or, in some cases, supersede this policy. These factors may include, but are not limited to legislative mandates, the physician or other provider contracts, and/or the enrollee s benefit coverage documents. Finally, this policy may not be implemented exactly the same way on the different electronic claims processing systems used by UnitedHealthcare due to programming or other constraints; however, UnitedHealthcare strives to minimize these variations. UnitedHealthcare may modify this reimbursement policy at any time by publishing a new version of the policy on this Website. However, the information presented in this policy is accurate and current as of the date of publication. *CPT copyright 2010 (or such other date of publication of CPT) American Medical Association. All rights reserved. CPT is a registered trademark of the American Medical Association. Proprietary information of UnitedHealthcare. Copyright 2015 United HealthCare Services, Inc. Table of Contents Application...2 Summary...2 Overview...2 Reimbursement Guidelines...3 CPT/HCPCS Codes...4 ICP/PCS Codes...5 References Included (but not limited to):...7 CMS NCD(s)...7 CMS Article...7 CMS Benefit Policy Manual...7 CMS Claims Processing Manual...7 MLN Matters...7 UnitedHealthcare Medicare Advantage Coverage Summaries...7 UnitedHealthcare Medical Policies...7 Others...7 History...7 Proprietary information of UnitedHealthcare. Copyright 2015 United HealthCare Services, Inc. Page 1
2 Application Reimbursement Policy This reimbursement policy applies to services reported using the Health Insurance Claim Form CMS-1500 or its electronic equivalent or its successor form, and services reported using facility claim form CMS-1450 or its electronic equivalent or its successor form. This policy applies to all products, all network and non-network physicians, and other health care professionals. The HCPCS/CPT code(s) may be subject to Correct Coding Initiative (CCI) edits. This policy does not take precedence over CCI edits. Please refer to the CCI for correct coding guidelines and specific applicable code combinations prior to billing UnitedHealthcare. It is not enough to link the procedure code to a correct, payable ICD-9-CM diagnosis code. The diagnosis must be present for the procedure to be paid. Compliance with the provisions in this policy is subject to monitoring by pre-payment review and/or post-payment data analysis and subsequent medical review. The effective date of changes/additions/deletions to this policy is the committee meeting date unless otherwise indicated. CPT codes and descriptions are copyright 2010 American Medical Association (or such other date of publication of CPT). All rights reserved. CPT is a registered trademark of the American Medical Association. Applicable FARS/DFARS restrictions apply to Government use. Fee schedules, relative value units, conversion factors, and/or related components are not assigned by the AMA, are not part of CPT, and the AMA is not recommending their use. The AMA does not directly or indirectly practice medicine or dispense medical services. The AMA assumes no liability for data contained or not contained herein. Current Dental Terminology (CDT), including procedure codes, nomenclature, descriptors, and other data contained therein, is copyright by the American Dental Association, 2002, All rights reserved. CDT is a registered trademark of the American Dental Association. Applicable FARS/DFARS apply. Summary Overview Essential tremor (ET) is a progressive, disabling action tremor (also referred to as a kinetic or postural tremor) which most often affects the hands during sustained arm extension or during voluntary motion such as writing or pouring. ET may also affect the head, voice, and legs and affects more than one million patients in the U.S. Although ET may start at any age, there appears to be a bimodal distribution peaking in the second and sixth decades. ET affects men and women equally and while its precise pathogenesis is unknown, it does occur genetically in some families as an autosomal dominant trait. Pharmacotherapy is the first line treatment of ET and may improve function by reducing the severity of tremor. However, certain patients do not adequately respond to or cannot tolerate these medications. Thalamic Deep Brain Stimulation (DBS) may be helpful for carefully selected individuals with marked tremor causing significant functional disability that is refractory to optimal medical therapy. Parkinson's disease (PD) is an age-related neurodegenerative disorder, characterized by tremor, rigidity, bradykinesia (gradual loss of spontaneous movement) and progressive postural instability. PD effects up to one million Americans. The primary underlying abnormality in PD is the progressive loss of dopamineproducing cells in the brain, leading to an imbalance of dopamine and acetylcholine, the normal neurotransmitters in the corpus striatum. The most common form, idiopathic PD, begins most often between ages forty five to sixty five, with average onset at age fifty-eight. The cause of PD remains unknown, although proposed theories include the role of genetic and certain environmental factors. For patients who become unresponsive to pharmacological treatments and/or have intolerable drug side effects, DBS may be helpful; DBS requires the stereotactic placement of an indwelling electrode in the brain. This treatment for PD supports observations that high-frequency stimulation of the affected neurons induces functional inhibition in target regions of the brain. DBS thus simulates the effect of an ablative surgical lesion but, unlike lessoning surgery, DBS adjusts the implanted electrode can be re-positioned (or removed). The mechanism of action remains unknown. Possible mechanisms include release of local inhibitory neurotransmitters, depolarization blockade, or jamming of abnormal neuron firing patterns. The device currently used for DBS is the Active system developed by Medtronic, Inc. (Minneapolis, MN). The system consists of several implantable and nonimplantable components (listed below), including a quadripolar electrode (four contact sites arranged along the distal edge) which is stereotactically implanted into the targeted structure. Stimulation parameters, including electrode contact site selection, stimulation pulse amplitude, frequency, and width adjusts to optimize symptom relief. Proprietary information of UnitedHealthcare. Copyright 2015 United HealthCare Services, Inc. Page 2
3 Reimbursement Guidelines Medicare will cover unilateral or bilateral thalamic ventralis intermedius nucleus (VIM) deep brain stimulation (DBS) for the treatment of essential tremor (ET) and/or Parkinsonian tremor and unilateral or bilateral subthalamic nucleus (STN) or globus pallidus interna (GPi) DBS for the treatment of Parkinson's disease (PD) only under the following conditions: Medicare will only consider DBS devices to be reasonable and necessary if they are Food and Drug Administration (FDA) approved devices for DBS or devices used in accordance with FDA approved protocols governing Category B Investigational Device Exemption (IDE) DBS clinical trials. For thalamic VIM DBS to be considered reasonable and necessary, patients must meet all of the following criteria: a. Diagnosis of ET based on postural or kinetic tremors of hand(s) without other neurologic signs, or diagnosis of idiopathic PD (presence of at least 2 cardinal PD features (tremor, rigidity, or bradykinesia)) which is of a tremor-dominant form b. Marked disabling tremor of at least level three or four on the Fahn-Tolosa-Marin Clinical Tremor Rating Scale (or equivalent scale) in the extremity intended for treatment, causing significant limitation in daily activities despite optimal medical therapy c. Willingness and ability to cooperate during conscious operative procedure, as well as during post-surgical evaluations, adjustments of medications and stimulator settings For STN or GPi DBS to be considered reasonable and necessary, patients must meet all of the following criteria: a. Diagnosis of PD based on the presence of at least two cardinal PD features (tremor, rigidity, or bradykinesia) b. Advanced idiopathic PD as determined by the use of Hoehn and Yahr stage or Unified Parkinson's Disease Rating Scale (UPDRS) part III motor subscale c. L-dopa responsive with clearly defined "on" periods d. Persistent disabling Parkinson's symptoms or drug side effects (e.g., dyskinesias, motor fluctuations, or disabling "off" periods) despite optimal medical therapy e. Willingness and ability to cooperate during conscious operative procedure, as well as during post-surgical evaluations, adjustments of medications and stimulator settings DBS is not reasonable and necessary and is not covered for ET or PD patients with any of the following: a. Non-idiopathic Parkinson's disease or "Parkinson's Plus" syndromes b. Cognitive impairment, dementia, or depression, which worsens or interferes with the patient's ability to benefit from DBS c. Current psychosis, alcohol abuse or other drug abuse. d. Structural lesions such as basal ganglionic stroke, tumor, or vascular malformation as etiology of the movement disorder e. Previous movement disorder surgery within the affected basal ganglion f. Significant medical, surgical, neurologic or orthopedic co-morbidities contraindicating DBS surgery or stimulation Patients who undergo DBS implantation should not be exposed to diathermy (deep heat treatment including shortwave diathermy, microwave diathermy and ultrasound diathermy) or any type of MRI, which may adversely affect the DBS system or adversely affect the brain around the implanted electrodes. DBS should be performed with extreme caution in patients with cardiac pacemakers or other electronically controlled implants, which may adversely affect or be affected by the DBS system. For DBS lead implantation to be considered reasonable and necessary, providers and facilities must meet all of the following criteria: Neurosurgeons must: o Be properly trained in the procedure; o Have experience with the surgical management of movement disorders, including DBS therapy; and Proprietary information of UnitedHealthcare. Copyright 2015 United HealthCare Services, Inc. Page 3
4 o Have experience performing stereotactic neurosurgical procedures. Operative teams must have training and experience with DBS systems, including knowledge of anatomical and neurophysiological characteristics for localizing the targeted nucleus, surgical and/or implantation techniques for the DBS system, and operational and functional characteristics of the device. Physicians specializing in movement disorders must be involved in both patient selection and postprocedure care. Hospital medical centers must have: o Brain imaging equipment (MRI and/or CT) for pre-operative stereotactic localization and targeting of the surgical site(s); o Operating rooms with all necessary equipment for stereotactic surgery; and o Support services necessary for care of patients undergoing this procedure and any potential complications arising intraoperatively or postoperatively. CPT/HCPCS Codes Code Description Twist drill, burr hole, craniotomy, or craniectomy with stereotactic implantation of neurostimulator electrode array in subcortical site (e.g., thalamus, globus pallidus, subthalamic nucleus, periventricular, periaqueductal gray), without use of intraoperative microelectrode recording; first array Twist drill, burr hole, craniotomy, or craniectomy with stereotactic implantation of nucleus, periventricular, periaqueductal gray), without use of intraoperative microelectrode recording; each additional array (List separately in addition to primary procedure) Twist drill, burr hole, craniotomy, or craniectomy with stereotactic implantation of nucleus, periventricular, periaqueductal gray), with use of intraoperative microelectrode recording; first array Twist drill, burr hole, craniotomy, or craniectomy with stereotactic implantation of nucleus, periventricular, periaqueductal gray), with use of intraoperative microelectrode recording; each additional array (List separately in addition to primary procedure) Revision or removal of intracranial neurostimulator electrodes Insertion or replacement of cranial neurostimulator pulse generator or receiver, direct or inductive coupling; with connection to a single electrode array Insertion or replacement of cranial neurostimulator pulse generator or receiver, direct or inductive coupling; with connection to 2 or more electrode arrays Twist drill, burr hole, craniotomy, or craniectomy with stereotactic implantation of nucleus, periventricular, periaqueductal gray), with use of intraoperative microelectrode recording; each additional array (List separately in addition to primary procedure) Functional cortical and subcortical mapping by stimulation and/or recording of electrodes on brain surface, or of depth electrodes, to provoke seizures or identify vital brain structures; initial hour of attendance by a physician or other qualified health care professional Functional cortical and subcortical mapping by stimulation and/or recording of electrodes on brain surface, or of depth electrodes, to provoke seizures or identify vital brain structures; each additional hour of attendance by a physician or other qualified health care professional (List separately in addition to code for primary procedure) Proprietary information of UnitedHealthcare. Copyright 2015 United HealthCare Services, Inc. Page 4
5 95970 Electronic analysis of implanted neurostimulator pulse generator system (eg, rate, pulse output modulation, cycling, impedance and patient compliance measurements); simple or complex brain, spinal cord, or peripheral (i.e., cranial nerve, peripheral nerve, sacral nerve, neuromuscular) neurostimulator pulse generator/transmitter, without reprogramming Electronic analysis of implanted neurostimulator pulse generator system (eg, rate, pulse output modulation, cycling, impedance and patient compliance measurements); simple spinal cord, or peripheral (i.e., peripheral nerve, sacral nerve, neuromuscular) neurostimulator pulse generator/transmitter, with intraoperative or subsequent programming Electronic analysis of implanted neurostimulator pulse generator system (eg, rate, pulse output modulation, cycling, impedance and patient compliance measurements); complex spinal cord, or peripheral (i.e., peripheral nerve, sacral nerve, neuromuscular) (except cranial nerve) neurostimulator pulse generator/transmitter, with intraoperative or subsequent programming, first hour Electronic analysis of implanted neurostimulator pulse generator system (eg, rate, pulse output modulation, cycling, impedance and patient compliance measurements); complex spinal cord, or peripheral (i.e., peripheral nerve, sacral nerve, neuromuscular) (except cranial nerve) neurostimulator pulse generator/transmitter, with intraoperative or subsequent programming, each additional 30 minutes after first hour (List separately in addition to code for primary procedure) Electronic analysis of implanted neurostimulator pulse generator system (eg, rate, pulse amplitude and duration, battery status, electrode selectability and polarity, impedance and patient compliance measurements), complex deep brain neurostimulator pulse generator/transmitter, with initial or subsequent programming; first hour Electronic analysis of implanted neurostimulator pulse generator system (eg, rate, pulse amplitude and duration, battery status, electrode selectability and polarity, impedance and patient compliance measurements), complex deep brain neurostimulator pulse generator/transmitter, with initial or subsequent programming; each additional 30 minutes after first hour (List separately in addition to code for primary procedure L8681 Patient programmer (external) for use with implantable programmable neurostimulator pulse generator, replacement only L8685 Implantable neurostimulator pulse generator, single array, rechargeable, includes extension L8686 Implantable neurostimulator pulse generator, single array, nonrechargeable, includes extension L8687 Implantable neurostimulator pulse generator, dual array, rechargeable, includes extension L8688 Implantable neurostimulator pulse generator, dual array, nonrechargeable, includes extension L8679 Implantable Neurostimulator, Pulse Generator, Any Type (new code effective 01/01/2014) (Replaces HCPCS codes L8685, L8686, L8687, and L8688) ICP/PCS Codes ICP Code Description PCS Code Description Implantation or replacement of intracranial neurostimulator lead(s) 00H00MZ Brain, Open Proprietary information of UnitedHealthcare. Copyright 2015 United HealthCare Services, Inc. Page 5
6 00H03MZ 00H04MZ 00H60MZ 00H63MZ 00H64MZ 00HE0MZ 00HE3MZ 00HE4MZ 00P00MZ 00P03MZ 00P04MZ 00P60MZ 00P63MZ 00P64MZ 00PE0MZ 00PE3MZ 00PE4MZ 00PEXMZ 00H03MZ Brain, Percutaneous Brain, Percutaneous Endoscopic Cerebral Ventricle, Open Endoscopic Cranial Nerve, Open Cranial Nerve, Percutaneous Cranial Nerve, Percutaneous Endoscopic Brain, Open Brain, Percutaneous Brain, Percutaneous Endoscopic Cerebral Ventricle, Open Endoscopic Cranial Nerve, Open Cranial Nerve, Percutaneous Cranial Nerve, Percutaneous Endoscopic Cranial Nerve, External Brain, Percutaneous Proprietary information of UnitedHealthcare. Copyright 2015 United HealthCare Services, Inc. Page 6
7 References Included (but not limited to): CMS NCD(s) NCD Parkinson s Disease Reference NCDs: NCD Vagus Nerve Stimulation NCD Electrical Nerve Stimulators CMS Article One article CMS Benefit Policy Manual Chapter 14; 10 Coverage of Medical Devices Chapter 15; 260 Ambulatory Surgical Center Services CMS Claims Processing Manual Chapter 12; 90.3 Physicians Services Performed in Ambulatory Surgical Centers (ASC) Chapter 14; 10 General ASC Information Chapter 20; 110 General Billing Requirements - for DME, Prosthetics, Orthotic Devices, and Supplies Chapter 32; 50 Parkinson's Disease MLN Matters Calendar Year (CY) 2014 Update for Durable Medical Equipment, Prosthetics, Orthotics, and Supplies, MM8531, January 01, 2014 UnitedHealthcare Medicare Advantage Coverage Summaries Parkinson s Disease UnitedHealthcare Reimbursement Policies NCD Vagus Nerve Stimulation UnitedHealthcare Medical Policies Deep Brain Stimulation Others Decision Memo for Deep Brain Stimulation for Parkinson's Disease, CMS Website Technology Assessment: Bilateral Deep Brain Stimulation (DBS) Of The Subthalamic Nucleus (STN) Or The Globus Pallidus Interna (Gpi) For Treatment Of Advanced Parkinson's Disease, CMS Website History Date Revisions 06/10/2015 Annual Review for MRP Committee presentation and approval 05/28/2014 Annual Review for MRP Committee presentation and approval 05/08/2013 Removed some of the current criteria (ICPs, HCPCS codes L8685-L8688) Added ICP Kept CPT Codes /07/2013 Removed CPT/HCPCS Codes: E0752-Implantable neurostimulator electrode, each (Expired 01/01/2006) E0756-Implantable neurostimulator pulse generator (Expired 01/01/2006) Twist drill, burr hole, craniotomy, or craniectomy for stereotactic implantation of one neurostimulator array in subcortical site (e.g., thalamus, globus pallidus, subthalamic nucleus, periventricular, periaqueductal gray) (Expired 01/01/2004) 11/30/2011 No change Proprietary information of UnitedHealthcare. Copyright 2015 United HealthCare Services, Inc. Page 7
National Coverage Determination. Vagus Nerve Stimulation (VNS)
 National Coverage Determination Vagus Nerve Stimulation (VNS) Number NEURO-004 Contractor Name Wisconsin Physicians Service Insurance Corporation AMA CPT Copyright Statement CPT codes, descriptions and
National Coverage Determination Vagus Nerve Stimulation (VNS) Number NEURO-004 Contractor Name Wisconsin Physicians Service Insurance Corporation AMA CPT Copyright Statement CPT codes, descriptions and
Diabetes Outpatient Self-Management Training (NCD 40.1)
 Policy Number 40.1 Approved By UnitedHealthcare Medicare Reimbursement Policy Committee Current Approval Date 02/11/2015 IMPORTANT NOTE ABOUT THIS REIMBURSEMENT POLICY This policy is applicable to UnitedHealthcare
Policy Number 40.1 Approved By UnitedHealthcare Medicare Reimbursement Policy Committee Current Approval Date 02/11/2015 IMPORTANT NOTE ABOUT THIS REIMBURSEMENT POLICY This policy is applicable to UnitedHealthcare
Routine Costs in a Clinical Trial (NCD 310.1)
 Policy Number 310.1 Approved By UnitedHealthcare Medicare Committee Current Approval Date 12/16/2015 IMPORTANT NOTE ABOUT THIS REIMBURSEMENT POLICY This policy is applicable to UnitedHealthcare Medicare
Policy Number 310.1 Approved By UnitedHealthcare Medicare Committee Current Approval Date 12/16/2015 IMPORTANT NOTE ABOUT THIS REIMBURSEMENT POLICY This policy is applicable to UnitedHealthcare Medicare
Coverage and Authorization Services is available to respond to your coding questions toll-free at 800-292-2903.
 For Urinary Control Commonly Billed Codes October 2010 Medtronic provides this information for your convenience only. It is not intended as a recommendation regarding clinical practice. It is the responsibility
For Urinary Control Commonly Billed Codes October 2010 Medtronic provides this information for your convenience only. It is not intended as a recommendation regarding clinical practice. It is the responsibility
New Patient Visit. UnitedHealthcare Medicare Reimbursement Policy Committee
 New Patient Visit Policy Number NPV04242013RP Approved By UnitedHealthcare Medicare Committee Current Approval Date 12/16/2015 IMPORTANT NOTE ABOUT THIS REIMBURSEMENT POLICY This policy is applicable to
New Patient Visit Policy Number NPV04242013RP Approved By UnitedHealthcare Medicare Committee Current Approval Date 12/16/2015 IMPORTANT NOTE ABOUT THIS REIMBURSEMENT POLICY This policy is applicable to
Contractor Number 11302. Oversight Region Region IV
 Local Coverage Determination (LCD): Spinal Cord Stimulators for Chronic Pain (L32549) Contractor Information Contractor Name Palmetto GBA opens in new window Contractor Number 11302 Contractor Type MAC
Local Coverage Determination (LCD): Spinal Cord Stimulators for Chronic Pain (L32549) Contractor Information Contractor Name Palmetto GBA opens in new window Contractor Number 11302 Contractor Type MAC
Incident To Services
 Policy Number INT04242013RP Approved By Incident To Services UnitedHealthcare Medicare Committee Current Approval Date 11/18/2015 IMPORTANT NOTE ABOUT THIS REIMBURSEMENT POLICY This policy is applicable
Policy Number INT04242013RP Approved By Incident To Services UnitedHealthcare Medicare Committee Current Approval Date 11/18/2015 IMPORTANT NOTE ABOUT THIS REIMBURSEMENT POLICY This policy is applicable
Local Coverage Determination (LCD): Spinal Cord Stimulation (Dorsal Column Stimulation) (L34705)
 Local Coverage Determination (LCD): Spinal Cord Stimulation (Dorsal Column Stimulation) (L34705) Contractor Information Contractor Name Novitas Solutions, Inc. LCD Information Document Information LCD
Local Coverage Determination (LCD): Spinal Cord Stimulation (Dorsal Column Stimulation) (L34705) Contractor Information Contractor Name Novitas Solutions, Inc. LCD Information Document Information LCD
REIMBURSEMENT POLICY CMS-1500 Physical Medicine & Rehabilitation: Multiple Therapy Procedure Reduction Policy 2/13/2013
 Policy Number REIMBURSEMENT POLICY CMS-1500 Physical Medicine & Rehabilitation: Multiple Therapy Procedure Reduction Policy 2013R0121C Annual Approval Date 2/13/2013 Approved By National Reimbursement
Policy Number REIMBURSEMENT POLICY CMS-1500 Physical Medicine & Rehabilitation: Multiple Therapy Procedure Reduction Policy 2013R0121C Annual Approval Date 2/13/2013 Approved By National Reimbursement
Observation Care Evaluation and Management Codes Policy
 Policy Number REIMBURSEMENT POLICY Observation Care Evaluation and Management Codes Policy 2016R0115A Annual Approval Date 3/11/2015 Approved By Payment Policy Oversight Committee IMPORTANT NOTE ABOUT
Policy Number REIMBURSEMENT POLICY Observation Care Evaluation and Management Codes Policy 2016R0115A Annual Approval Date 3/11/2015 Approved By Payment Policy Oversight Committee IMPORTANT NOTE ABOUT
Physical Medicine & Rehabilitation: Maximum Combined Frequency per Day Policy
 REIMBURSEMENT POLICY Policy Number Physical Medicine & Rehabilitation: Maximum Combined Frequency per Day Policy 2015R0101B Annual Approval Date 7/8/2015 Approved By Payment Policy Oversight Committee
REIMBURSEMENT POLICY Policy Number Physical Medicine & Rehabilitation: Maximum Combined Frequency per Day Policy 2015R0101B Annual Approval Date 7/8/2015 Approved By Payment Policy Oversight Committee
Physical Medicine & Rehabilitation: Multiple Therapy Procedure Reduction Policy
 Policy Number 2015R0121C Physical Medicine & Rehabilitation: Procedure Reduction Policy Annual Approval Date 3/11/2015 Approved By Payment Policy Oversight Committee IMPORTANT NOTE ABOUT THIS REIMBURSEMENT
Policy Number 2015R0121C Physical Medicine & Rehabilitation: Procedure Reduction Policy Annual Approval Date 3/11/2015 Approved By Payment Policy Oversight Committee IMPORTANT NOTE ABOUT THIS REIMBURSEMENT
Pressure Reducing Support Surfaces
 Policy Number AIR06012011RP Approved By UnitedHealthcare Medicare Committee Current Approval Date 05/27/2015 IMPORTANT NOTE ABOUT THIS REIMBURSEMENT POLICY This policy is applicable to UnitedHealthcare
Policy Number AIR06012011RP Approved By UnitedHealthcare Medicare Committee Current Approval Date 05/27/2015 IMPORTANT NOTE ABOUT THIS REIMBURSEMENT POLICY This policy is applicable to UnitedHealthcare
Assistant Surgeon Policy
 Policy Number 2015R5000D Annual Approval Date Assistant Surgeon Policy 11/12/2014 Approved By REIMBURSEMENT POLICY Payment Policy Oversight Committee IMPORTANT NOTE ABOUT THIS REIMBURSEMENT POLICY You
Policy Number 2015R5000D Annual Approval Date Assistant Surgeon Policy 11/12/2014 Approved By REIMBURSEMENT POLICY Payment Policy Oversight Committee IMPORTANT NOTE ABOUT THIS REIMBURSEMENT POLICY You
Corporate Medical Policy
 Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: spinal_cord_stimulation 3/1980 10/2015 10/2016 10/2015 Description of Procedure or Service Spinal cord stimulation
Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: spinal_cord_stimulation 3/1980 10/2015 10/2016 10/2015 Description of Procedure or Service Spinal cord stimulation
Ambulance Policy. Approved By 7/8/2015
 Ambulance Number 2015R0123H Annual Approval Date 7/8/2015 Approved By Payment Oversight Committee IMPORTANT NOTE ABOUT THIS REIMBURSEMENT POLICY You are responsible for submission of accurate claims. This
Ambulance Number 2015R0123H Annual Approval Date 7/8/2015 Approved By Payment Oversight Committee IMPORTANT NOTE ABOUT THIS REIMBURSEMENT POLICY You are responsible for submission of accurate claims. This
Preventive Medicine and Screening Policy
 REIMBURSEMENT POLICY Policy Number 2015R0013C Preventive Medicine and Screening Policy Annual Approval Date 3/11/2015 Approved By Payment Policy Oversight Committee IMPORTANT NOTE ABOUT THIS REIMBURSEMENT
REIMBURSEMENT POLICY Policy Number 2015R0013C Preventive Medicine and Screening Policy Annual Approval Date 3/11/2015 Approved By Payment Policy Oversight Committee IMPORTANT NOTE ABOUT THIS REIMBURSEMENT
Local Coverage Article: Cardiovascular Stress Testing (A53123)
 Local Coverage Article: Cardiovascular Stress Testing (A53123) Contractor Information Contractor Name Novitas Solutions, Inc. Article Information General Information Article ID A53123 Original ICD-9 Article
Local Coverage Article: Cardiovascular Stress Testing (A53123) Contractor Information Contractor Name Novitas Solutions, Inc. Article Information General Information Article ID A53123 Original ICD-9 Article
Motor dysfunction 2: Spinal cord injury and subcortical motor disorders ANATOMY REVIEW: Basal Ganglia
 Motor dysfunction 2: Spinal cord injury and subcortical motor disorders ANATOMY REVIEW: Basal Ganglia A group of subcortical nuclei caudate, putamen, globus pallidus Caudate & Putamen = Neostriatum caudate
Motor dysfunction 2: Spinal cord injury and subcortical motor disorders ANATOMY REVIEW: Basal Ganglia A group of subcortical nuclei caudate, putamen, globus pallidus Caudate & Putamen = Neostriatum caudate
PARKINSON S DISEASE IN LONG-TERM-CARE SETTINGS
 PARKINSON S DISEASE IN LONG-TERM-CARE SETTINGS De Anna Looper, RN CHPN Corporate Clinical Consultant / Legal Nurse Consultant Carrefour Associates L.L.C. PARKINSON S DISEASE IN LONG-TERM-CARE SETTINGS
PARKINSON S DISEASE IN LONG-TERM-CARE SETTINGS De Anna Looper, RN CHPN Corporate Clinical Consultant / Legal Nurse Consultant Carrefour Associates L.L.C. PARKINSON S DISEASE IN LONG-TERM-CARE SETTINGS
Intra-operative Nerve Monitoring Coding Guide. March 1, 2011
 Intra-operative Nerve Monitoring Coding Guide March 1, 2011 Please direct any questions to: Patty Telgener, RN Vice President, Reimbursement Services Emerson Consultants (303) 526-7604 (office) (303) 570-2159
Intra-operative Nerve Monitoring Coding Guide March 1, 2011 Please direct any questions to: Patty Telgener, RN Vice President, Reimbursement Services Emerson Consultants (303) 526-7604 (office) (303) 570-2159
KYPHON. Reimbursement Guide. Physician Reimbursement. Balloon Kyphoplasty Procedure. ICD-9-CM Diagnosis Codes. CPT Codes and Payment
 KYPHON Balloon Kyphoplasty Procedure Reimbursement Guide ICD-9-CM Diagnosis Codes Providers should report the ICD-9-CM diagnosis code that most accurately describes the patient s condition. Please refer
KYPHON Balloon Kyphoplasty Procedure Reimbursement Guide ICD-9-CM Diagnosis Codes Providers should report the ICD-9-CM diagnosis code that most accurately describes the patient s condition. Please refer
Radiology Multiple Imaging Reduction Policy
 Policy Number 2015R0085C Radiology Multiple Imaging Reduction Policy Annual Approval Date 7/8/2015 Approved By Payment Policy Oversight Committee IMPORTANT NOTE ABOUT THIS You are responsible for submission
Policy Number 2015R0085C Radiology Multiple Imaging Reduction Policy Annual Approval Date 7/8/2015 Approved By Payment Policy Oversight Committee IMPORTANT NOTE ABOUT THIS You are responsible for submission
ANESTHESIA - Medicare
 ANESTHESIA - Medicare Policy Number: UM14P0008A2 Effective Date: August 19, 2014 Last Reviewed: January 1, 2016 PAYMENT POLICY HISTORY Version DATE ACTION / DESCRIPTION Version 2 January 1, 2016 Under
ANESTHESIA - Medicare Policy Number: UM14P0008A2 Effective Date: August 19, 2014 Last Reviewed: January 1, 2016 PAYMENT POLICY HISTORY Version DATE ACTION / DESCRIPTION Version 2 January 1, 2016 Under
Supply Policy. Approved By 1/27/2014
 Supply Policy Policy Number 2014R0006A Annual Approval Date 1/27/2014 Approved By National Reimbursement Forum United HealthCare Community & State Payment Policy Committee IMPORTANT NOTE ABOUT THIS You
Supply Policy Policy Number 2014R0006A Annual Approval Date 1/27/2014 Approved By National Reimbursement Forum United HealthCare Community & State Payment Policy Committee IMPORTANT NOTE ABOUT THIS You
Modifier Reference Policy
 Policy Number 2016R0111C Annual Approval Date Modifier Reference Policy 11/11/2015 Approved By Payment Policy Oversight Committee IMPORTANT NOTE ABOUT THIS REIMBURSEMENT POLICY You are responsible for
Policy Number 2016R0111C Annual Approval Date Modifier Reference Policy 11/11/2015 Approved By Payment Policy Oversight Committee IMPORTANT NOTE ABOUT THIS REIMBURSEMENT POLICY You are responsible for
 Epimed Would Like To Congratulate The 20th Annual Budapest Conference The New Shape of Pain Relief SmalleST. ThiNNeST. CoNTouRed design. Precision Novi is the world s smallest, thinnest 16 contact primary
Epimed Would Like To Congratulate The 20th Annual Budapest Conference The New Shape of Pain Relief SmalleST. ThiNNeST. CoNTouRed design. Precision Novi is the world s smallest, thinnest 16 contact primary
ABC s of Parkinson s Disease 4/29/15 Karen Parenti, MS, PsyD
 ABC s of Parkinson s Disease 4/29/15 Karen Parenti, MS, PsyD What is Parkinson s Disease? Parkinson's disease is a progressive disorder of the nervous system that affects movement. It develops gradually,
ABC s of Parkinson s Disease 4/29/15 Karen Parenti, MS, PsyD What is Parkinson s Disease? Parkinson's disease is a progressive disorder of the nervous system that affects movement. It develops gradually,
EXHIBIT C BOSTON SCIENTIFIC PRODUCTS AVAILABLE FOR NIH BRAIN INITIATIVE Page 1 of 9
 Page 1 of 9 NOTE: All systems and products should be considered as investigational-use only in the context of the NIH BRAIN Initiative. VERCISE DEEP BRAIN STIMULATION SYSTEM Description: 16-channel rechargeable
Page 1 of 9 NOTE: All systems and products should be considered as investigational-use only in the context of the NIH BRAIN Initiative. VERCISE DEEP BRAIN STIMULATION SYSTEM Description: 16-channel rechargeable
Intrathecal Baclofen for CNS Spasticity
 Intrathecal Baclofen for CNS Spasticity Last Review Date: November 13, 2015 Number: MG.MM.ME.31bC5 Medical Guideline Disclaimer Property of EmblemHealth. All rights reserved. The treating physician or
Intrathecal Baclofen for CNS Spasticity Last Review Date: November 13, 2015 Number: MG.MM.ME.31bC5 Medical Guideline Disclaimer Property of EmblemHealth. All rights reserved. The treating physician or
Suppliers are to follow The Health Plan requirements for precertification, as applicable.
 Eye Prostheses Adopted from the National Government Services website. For any item to be covered by The Health Plan, it must: 1. Be eligible for a defined Medicare or Health Plan benefit category 2. Be
Eye Prostheses Adopted from the National Government Services website. For any item to be covered by The Health Plan, it must: 1. Be eligible for a defined Medicare or Health Plan benefit category 2. Be
Telemedicine Policy Annual Approval Date
 Policy Number 2016R0046A Telemedicine Policy Annual Approval Date 4/08/2015 Approved By REIMBURSEMENT POLICY CMS-1500 Payment Policy Oversight Committee IMPORTANT NOTE ABOUT THIS REIMBURSEMENT POLICY You
Policy Number 2016R0046A Telemedicine Policy Annual Approval Date 4/08/2015 Approved By REIMBURSEMENT POLICY CMS-1500 Payment Policy Oversight Committee IMPORTANT NOTE ABOUT THIS REIMBURSEMENT POLICY You
Guide to Deep Brain Stimulation Therapy
 Parkinson s Disease: Guide to Deep Brain Stimulation Therapy By: Michael S. Okun, MD Medical Director, National Parkinson Foundation Center of Excellence Co-Director, Center for Movement Disorders and
Parkinson s Disease: Guide to Deep Brain Stimulation Therapy By: Michael S. Okun, MD Medical Director, National Parkinson Foundation Center of Excellence Co-Director, Center for Movement Disorders and
Intraoperative Nerve Monitoring Coding Guide. March 1, 2010
 Intraoperative Nerve Monitoring Coding Guide March 1, 2010 Please direct any questions to: Kim Brew Manager Reimbursement and Therapy Access Medtronic ENT (904) 279-7569 Rev 9/10 KB TO OUR PARTNERS IN
Intraoperative Nerve Monitoring Coding Guide March 1, 2010 Please direct any questions to: Kim Brew Manager Reimbursement and Therapy Access Medtronic ENT (904) 279-7569 Rev 9/10 KB TO OUR PARTNERS IN
Advanced Monitoring Parameters 2015 Quick Guide to Hospital Coding, Coverage and Payment
 Advanced Monitoring Parameters 2015 Quick Guide to Hospital Coding, Coverage and Payment The information in this quick guide is provided by our Healthcare Economics Department, which supports Respiratory
Advanced Monitoring Parameters 2015 Quick Guide to Hospital Coding, Coverage and Payment The information in this quick guide is provided by our Healthcare Economics Department, which supports Respiratory
Modifier Reference Policy
 Policy Number 2015R0111C Annual Approval Date Modifier Reference Policy 11/12/2014 Approved By Payment Policy Oversight Committee IMPORTANT NOTE ABOUT THIS REIMBURSEMENT POLICY You are responsible for
Policy Number 2015R0111C Annual Approval Date Modifier Reference Policy 11/12/2014 Approved By Payment Policy Oversight Committee IMPORTANT NOTE ABOUT THIS REIMBURSEMENT POLICY You are responsible for
Ambulatory Surgery Center Coding and Payment Guide 2015
 Targeted Drug Delivery Ambulatory Surgery Center Coding and Payment Guide 2015 Flowonix Medical has compiled this coding information for your convenience. This information is gathered from third party
Targeted Drug Delivery Ambulatory Surgery Center Coding and Payment Guide 2015 Flowonix Medical has compiled this coding information for your convenience. This information is gathered from third party
CODING. Neighborhood Health Plan 1 Provider Payment Guidelines
 CODING Policy The terms of this policy set forth the guidelines for reporting the provision of care rendered by NHP participating providers, including but not limited to use of standard diagnosis and procedure
CODING Policy The terms of this policy set forth the guidelines for reporting the provision of care rendered by NHP participating providers, including but not limited to use of standard diagnosis and procedure
Professional/Technical Component Policy
 Policy Number 2015R0012C Professional/Technical Component Policy Annual Approval Date 1/27/2014 Approved By Payment Policy Oversight Committee IMPORTANT NOTE ABOUT THIS REIMBURSEMENT POLICY You are responsible
Policy Number 2015R0012C Professional/Technical Component Policy Annual Approval Date 1/27/2014 Approved By Payment Policy Oversight Committee IMPORTANT NOTE ABOUT THIS REIMBURSEMENT POLICY You are responsible
Hyperbaric Oxygen Therapy (NCD 20.29)
 Policy Number Reimbursement Policy 20.29 Approved By UnitedHealthcare Medicare Reimbursement Policy Committee Current Approval Date 10/08/2014 IMPORTANT NOTE ABOUT THIS REIMBURSEMENT POLICY This policy
Policy Number Reimbursement Policy 20.29 Approved By UnitedHealthcare Medicare Reimbursement Policy Committee Current Approval Date 10/08/2014 IMPORTANT NOTE ABOUT THIS REIMBURSEMENT POLICY This policy
Global Surgery Fact Sheet
 DEPARTMENT OF HEALTH AND HUMAN SERVICES Centers for Medicare & Medicaid Services R Global Surgery Fact Sheet Fact Sheet Definition of a Global Surgical Package Medicare established a national definition
DEPARTMENT OF HEALTH AND HUMAN SERVICES Centers for Medicare & Medicaid Services R Global Surgery Fact Sheet Fact Sheet Definition of a Global Surgical Package Medicare established a national definition
WELLCARE CLAIM PAYMENT POLICIES
 WellCare and Harmony Health Plan s claim payment policies are based on publicly distributed guidelines from established industry sources such as the Centers for Medicare and Medicaid Services (CMS), the
WellCare and Harmony Health Plan s claim payment policies are based on publicly distributed guidelines from established industry sources such as the Centers for Medicare and Medicaid Services (CMS), the
Rotator Cuff Repair Surgical Procedures
 Rotator Cuff Repair Surgical Procedures 2011 Reimbursement and Coding Reference Guide for Physicians and Hospitals This coding reference guide is intended to illustrate the common CPT * codes, ICD-9 CM
Rotator Cuff Repair Surgical Procedures 2011 Reimbursement and Coding Reference Guide for Physicians and Hospitals This coding reference guide is intended to illustrate the common CPT * codes, ICD-9 CM
Incontinence Supplies Policy
 REIMBURSEMENT POLICY Policy Number 2016R7111A Incontinence Supplies Policy Annual Approval Date 3/11/2015 Approved By Payment Policy Oversight Committee IMPORTANT NOTE ABOUT THIS REIMBURSEMENT POLICY You
REIMBURSEMENT POLICY Policy Number 2016R7111A Incontinence Supplies Policy Annual Approval Date 3/11/2015 Approved By Payment Policy Oversight Committee IMPORTANT NOTE ABOUT THIS REIMBURSEMENT POLICY You
Occupational Therapy Program
 Health Care Authority Occupational Therapy Program Billing Instructions [WAC 182-545-0300] About This Publication This publication supersedes all previous Agency Occupational Therapy Program Billing Instructions
Health Care Authority Occupational Therapy Program Billing Instructions [WAC 182-545-0300] About This Publication This publication supersedes all previous Agency Occupational Therapy Program Billing Instructions
PSYCHOLOGICAL AND NEUROPSYCHOLOGICAL TESTING
 Status Active Medical and Behavioral Health Policy Section: Behavioral Health Policy Number: X-45 Effective Date: 01/22/2014 Blue Cross and Blue Shield of Minnesota medical policies do not imply that members
Status Active Medical and Behavioral Health Policy Section: Behavioral Health Policy Number: X-45 Effective Date: 01/22/2014 Blue Cross and Blue Shield of Minnesota medical policies do not imply that members
Physical Therapy Program
 Health and Recovery Services Administration Physical Therapy Program Billing Instructions ProviderOne Readiness Edition [WAC 388-545-0500] About This Publication This publication supersedes all previous
Health and Recovery Services Administration Physical Therapy Program Billing Instructions ProviderOne Readiness Edition [WAC 388-545-0500] About This Publication This publication supersedes all previous
Status Active. Assistant Surgeons. This policy addresses reimbursement for assistant surgical procedures during the same operative session.
 Status Active Reimbursement Policy Section: Surgery/Interventional Procedure Policy Number: RP - Surgery/Interventional Procedure - 001 Assistant Surgeons Effective Date: June 1, 2015 Assistant Surgeons
Status Active Reimbursement Policy Section: Surgery/Interventional Procedure Policy Number: RP - Surgery/Interventional Procedure - 001 Assistant Surgeons Effective Date: June 1, 2015 Assistant Surgeons
Stuart B Black MD, FAAN Chief of Neurology Co-Medical Director: Neuroscience Center Baylor University Medical Center at Dallas
 Billing and Coding in Neurology and Headache Stuart B Black MD, FAAN Chief of Neurology Co-Medical Director: Neuroscience Center Baylor University Medical Center at Dallas CPT Codes vs. ICD Codes Category
Billing and Coding in Neurology and Headache Stuart B Black MD, FAAN Chief of Neurology Co-Medical Director: Neuroscience Center Baylor University Medical Center at Dallas CPT Codes vs. ICD Codes Category
US Reimbursement Guide
 US Reimbursement Guide The information with this notice is general reimbursement information only. It is not legal advice, nor is it about how to code, complete or submit any particular claim for payment.
US Reimbursement Guide The information with this notice is general reimbursement information only. It is not legal advice, nor is it about how to code, complete or submit any particular claim for payment.
Coding and Payment Guide for the Physical Therapist. An essential coding, billing, and reimbursement resource for the physical therapist
 Coding and Payment Guide for the Physical Therapist An essential coding, billing, and reimbursement resource for the physical therapist 2011 Contents Introduction...1 Coding Systems... 1 HCPCS Level II
Coding and Payment Guide for the Physical Therapist An essential coding, billing, and reimbursement resource for the physical therapist 2011 Contents Introduction...1 Coding Systems... 1 HCPCS Level II
Durable Medical Equipment, Orthotics and Prosthetics Multiple Frequency Policy
 Policy Number REIMBURSEMENT POLICY Durable Medical Equipment, Orthotics and Prosthetics Multiple Frequency Policy 2015R0109C Annual Approval Date 7/8/2015 Approved By Payment Policy Oversight Committee
Policy Number REIMBURSEMENT POLICY Durable Medical Equipment, Orthotics and Prosthetics Multiple Frequency Policy 2015R0109C Annual Approval Date 7/8/2015 Approved By Payment Policy Oversight Committee
Chapter 28. Drug Treatment of Parkinson s Disease
 Chapter 28 Drug Treatment of Parkinson s Disease 1. Introduction Parkinsonism Tremors hands and head develop involuntary movements when at rest; pin rolling sign (finger and thumb) Muscle rigidity arthritis
Chapter 28 Drug Treatment of Parkinson s Disease 1. Introduction Parkinsonism Tremors hands and head develop involuntary movements when at rest; pin rolling sign (finger and thumb) Muscle rigidity arthritis
Modifier Reference PAYMENT POLICY ID NUMBER: 10-011. Original Effective Date: 05/14/10. Revised: 05/31/12 DESCRIPTION:
 Private Property of Florida Blue. This payment policy is Copyright 2012, Florida Blue. All Rights Reserved. You may not copy or use this document or disclose its contents without the express written permission
Private Property of Florida Blue. This payment policy is Copyright 2012, Florida Blue. All Rights Reserved. You may not copy or use this document or disclose its contents without the express written permission
ICD-10 Coding for Audiology
 ICD-10 Coding for Audiology Mary Sue Fino-Szumski, Ph.D., M.B.A. Vanderbilt University School of Medicine Vanderbilt Bill Wilkerson Center Department of Hearing and Speech Sciences Disclosure Financial
ICD-10 Coding for Audiology Mary Sue Fino-Szumski, Ph.D., M.B.A. Vanderbilt University School of Medicine Vanderbilt Bill Wilkerson Center Department of Hearing and Speech Sciences Disclosure Financial
Local Coverage Article: Endovascular Repair of Aortic Aneurysms (A53124)
 Local Coverage Article: Endovascular Repair of Aortic Aneurysms (A53124) Contractor Information Contractor Name Novitas Solutions, Inc. Article Information General Information Article ID A53124 Original
Local Coverage Article: Endovascular Repair of Aortic Aneurysms (A53124) Contractor Information Contractor Name Novitas Solutions, Inc. Article Information General Information Article ID A53124 Original
CODING SHEETS CHRONIC INTRACTABLE SPASTICITY. Effective January 1, 2009 CODMAN 3000 NEUROMODULATION AND ONCOLOGY REIMBURSEMENT HOTLINE
 CODING SHEETS CHRONIC INTRACTABLE SPASTICITY Effective January 1, 2009 CODMAN 3000 NEUROMODULATION AND ONCOLOGY REIMBURSEMENT HOTLINE Phone: 800-609-1108 Email: [email protected] Fax: 303-703-1572 CODMAN
CODING SHEETS CHRONIC INTRACTABLE SPASTICITY Effective January 1, 2009 CODMAN 3000 NEUROMODULATION AND ONCOLOGY REIMBURSEMENT HOTLINE Phone: 800-609-1108 Email: [email protected] Fax: 303-703-1572 CODMAN
Coding and Payment Guide for Anesthesia Services. An essential coding, billing, and reimbursement resource for anesthesiology and pain management
 Coding and Payment Guide for Anesthesia Services An essential coding, billing, and reimbursement resource for anesthesiology and pain management 2011 Contents Introduction...1 Coding Systems... 1 Claim
Coding and Payment Guide for Anesthesia Services An essential coding, billing, and reimbursement resource for anesthesiology and pain management 2011 Contents Introduction...1 Coding Systems... 1 Claim
Telemedicine Policy. Approved By 1/27/2014
 REIMBURSEMENT POLICY Policy Number 2015R0046F Annual Approval Date Telemedicine Policy 1/27/2014 Approved By National Reimbursement Forum United HealthCare Community & State Payment Policy Committee IMPORTANT
REIMBURSEMENT POLICY Policy Number 2015R0046F Annual Approval Date Telemedicine Policy 1/27/2014 Approved By National Reimbursement Forum United HealthCare Community & State Payment Policy Committee IMPORTANT
Spinal Cord Stimulation (SCS) Therapy: Fact Sheet
 Spinal Cord Stimulation (SCS) Therapy: Fact Sheet What is SCS Therapy? Spinal cord stimulation (SCS) may be a life-changing 1 surgical option for patients to control their chronic neuropathic pain and
Spinal Cord Stimulation (SCS) Therapy: Fact Sheet What is SCS Therapy? Spinal cord stimulation (SCS) may be a life-changing 1 surgical option for patients to control their chronic neuropathic pain and
How To Get A Dbs Therapy For Obsessive Compulsive Disorder
 Sample Appeal Letters Appeals Payers who deny coverage usually do so for one of these three reasons: The therapy is investigational / experimental The therapy is not medically necessary The therapy is
Sample Appeal Letters Appeals Payers who deny coverage usually do so for one of these three reasons: The therapy is investigational / experimental The therapy is not medically necessary The therapy is
PARKINSON S DISEASE INTRODUCTION. Parkinson s disease is defined as a disease of the nervous system that affects voluntary movement.
 PARKINSON S DISEASE INTRODUCTION Parkinson s disease is a disorder of the brain and the nervous system. It is one of the more common neurological diseases in people over the age of 60, and it is more common
PARKINSON S DISEASE INTRODUCTION Parkinson s disease is a disorder of the brain and the nervous system. It is one of the more common neurological diseases in people over the age of 60, and it is more common
OBSERVATION CARE EVALUATION AND MANAGEMENT CODES
 REIMBURSEMENT POLICY OBSERVATION CARE EVALUATION AND MANAGEMENT CODES Policy Number: ADMINISTRATIVE 232.8 T0 Effective Date: April, 205 Table of Contents APPLICABLE LINES OF BUSINESS/PRODUCTS... APPLICATION...
REIMBURSEMENT POLICY OBSERVATION CARE EVALUATION AND MANAGEMENT CODES Policy Number: ADMINISTRATIVE 232.8 T0 Effective Date: April, 205 Table of Contents APPLICABLE LINES OF BUSINESS/PRODUCTS... APPLICATION...
Local Coverage Determination (LCD): Special EEG Tests (L33699) Contractor Information
 Local Coverage Determination (LCD): Special EEG Tests (L33699) Contractor Information Contractor Name First Coast Service Options, Inc. Document Information LCD ID L33699 LCD Title Special EEG Tests Original
Local Coverage Determination (LCD): Special EEG Tests (L33699) Contractor Information Contractor Name First Coast Service Options, Inc. Document Information LCD ID L33699 LCD Title Special EEG Tests Original
Transmittal 55 Date: MAY 5, 2006. SUBJECT: Changes Conforming to CR3648 for Therapy Services
 CMS Manual System Pub 100-03 Medicare National Coverage Determinations Department of Health & Human Services (DHHS) Centers for Medicare & Medicaid Services (CMS) Transmittal 55 Date: MAY 5, 2006 Change
CMS Manual System Pub 100-03 Medicare National Coverage Determinations Department of Health & Human Services (DHHS) Centers for Medicare & Medicaid Services (CMS) Transmittal 55 Date: MAY 5, 2006 Change
Physician Coding and Payment Guide 2015
 Targeted Drug Delivery Physician Coding and Payment Guide 2015 Flowonix Medical has compiled this coding information for your convenience. This information is gathered from third party sources and is subject
Targeted Drug Delivery Physician Coding and Payment Guide 2015 Flowonix Medical has compiled this coding information for your convenience. This information is gathered from third party sources and is subject
Contractor Information. LCD Information. Local Coverage Determination (LCD): HbA1c (L32939) Contract Number 11202
 Local Coverage Determination (LCD): HbA1c (L32939) Contractor Information Contractor Name Palmetto GBA opens in new window Contract Number 11202 Contract Type MAC - Part B LCD Information Document Information
Local Coverage Determination (LCD): HbA1c (L32939) Contractor Information Contractor Name Palmetto GBA opens in new window Contract Number 11202 Contract Type MAC - Part B LCD Information Document Information
Status Active. Reimbursement Policy Section: Anesthesia Services Policy Number: RP - Anesthesia - 001 Anesthesia Effective Date: June 1, 2015
 Status Active Reimbursement Policy Section: Anesthesia Services Policy Number: RP - Anesthesia - 001 Anesthesia Effective Date: June 1, 2015 Anesthesia Policy Description: Definitions: This policy addresses
Status Active Reimbursement Policy Section: Anesthesia Services Policy Number: RP - Anesthesia - 001 Anesthesia Effective Date: June 1, 2015 Anesthesia Policy Description: Definitions: This policy addresses
REHABILITATION SERVICES (OUTPATIENT)
 REHABILITATION SERVICES (OUTPATIENT) Protocol: MSC028 Effective Date: March 1, 2016 Table of Contents Page COMMERCIAL COVERAGE RATIONALE... 1 DEFINITIONS... 2 APPLICABLE CODES... 4 REFERENCES... 7 POLICY
REHABILITATION SERVICES (OUTPATIENT) Protocol: MSC028 Effective Date: March 1, 2016 Table of Contents Page COMMERCIAL COVERAGE RATIONALE... 1 DEFINITIONS... 2 APPLICABLE CODES... 4 REFERENCES... 7 POLICY
Billing and Coding Guidance Co-morbidities associated with morbid obesity
 Billing and Coding Guidance Co-morbidities associated with morbid obesity AMA CPT / ADA CDT / AHA NUBC Copyright Statement CPT only copyright 2002-2014 American Medical Association. All Rights Reserved.
Billing and Coding Guidance Co-morbidities associated with morbid obesity AMA CPT / ADA CDT / AHA NUBC Copyright Statement CPT only copyright 2002-2014 American Medical Association. All Rights Reserved.
How To Cover Occupational Therapy
 Guidelines for Medical Necessity Determination for Occupational Therapy These Guidelines for Medical Necessity Determination (Guidelines) identify the clinical information MassHealth needs to determine
Guidelines for Medical Necessity Determination for Occupational Therapy These Guidelines for Medical Necessity Determination (Guidelines) identify the clinical information MassHealth needs to determine
Brain Cancer. This reference summary will help you understand how brain tumors are diagnosed and what options are available to treat them.
 Brain Cancer Introduction Brain tumors are not rare. Thousands of people are diagnosed every year with tumors of the brain and the rest of the nervous system. The diagnosis and treatment of brain tumors
Brain Cancer Introduction Brain tumors are not rare. Thousands of people are diagnosed every year with tumors of the brain and the rest of the nervous system. The diagnosis and treatment of brain tumors
CODING SHEETS CHRONIC INTRACTABLE PAIN MANAGEMENT. Effective January 1, 2011 CODMAN 3000 NEUROMODULATION AND ONCOLOGY REIMBURSEMENT HOTLINE
 CODING SHEETS CHRONIC INTRACTABLE PAIN MANAGEMENT Effective January 1, 2011 CODMAN 3000 NEUROMODULATION AND ONCOLOGY REIMBURSEMENT HOTLINE Phone: 800-609-1108 Email: [email protected] Fax: 303-703-1572
CODING SHEETS CHRONIC INTRACTABLE PAIN MANAGEMENT Effective January 1, 2011 CODMAN 3000 NEUROMODULATION AND ONCOLOGY REIMBURSEMENT HOTLINE Phone: 800-609-1108 Email: [email protected] Fax: 303-703-1572
Sacral Nerve Neuromodulation/Stimulation
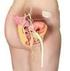 MEDICAL POLICY POLICY RELATED POLICIES POLICY GUIDELINES DESCRIPTION SCOPE BENEFIT APPLICATION RATIONALE REFERENCES CODING APPENDIX HISTORY Sacral Nerve Neuromodulation/Stimulation Number 7.01.69 Effective
MEDICAL POLICY POLICY RELATED POLICIES POLICY GUIDELINES DESCRIPTION SCOPE BENEFIT APPLICATION RATIONALE REFERENCES CODING APPENDIX HISTORY Sacral Nerve Neuromodulation/Stimulation Number 7.01.69 Effective
Empire BlueCross BlueShield Professional Reimbursement Policy
 Subject: Evaluation and Management Services and Related Modifiers -25 & 57 NY Policy: 0026 Effective: 8/19/2013 1/31/2014 Coverage is subject to the terms, conditions, and limitations of an individual
Subject: Evaluation and Management Services and Related Modifiers -25 & 57 NY Policy: 0026 Effective: 8/19/2013 1/31/2014 Coverage is subject to the terms, conditions, and limitations of an individual
Local Coverage Article: Venipuncture Necessitating Physician s Skill for Specimen Collection Supplemental Instructions Article (A50852)
 Local Coverage Article: Venipuncture Necessitating Physician s Skill for Specimen Collection Supplemental Instructions Article (A50852) Contractor Information Contractor Name CGS Administrators, LLC Article
Local Coverage Article: Venipuncture Necessitating Physician s Skill for Specimen Collection Supplemental Instructions Article (A50852) Contractor Information Contractor Name CGS Administrators, LLC Article
Local Coverage Determination (LCD): Medicine: Autonomic Function Tests (L34500)
 Local Coverage Determination (LCD): Medicine: Autonomic Function Tests (L34500) Contractor Information Contractor Name Cahaba Government Benefit Administrators, LLC LCD Information Document Information
Local Coverage Determination (LCD): Medicine: Autonomic Function Tests (L34500) Contractor Information Contractor Name Cahaba Government Benefit Administrators, LLC LCD Information Document Information
Parkinson's s disease - a
 Parkinson's Disease Parkinson's s disease - a progressive disorder of the nervous system that affects movement. The most common perception of Parkinson s is the patient having tremors. Hands shaking, inability
Parkinson's Disease Parkinson's s disease - a progressive disorder of the nervous system that affects movement. The most common perception of Parkinson s is the patient having tremors. Hands shaking, inability
Vaccination (Immunization)
 Policy Number IMM10012009SC Approved By Vaccination (Immunization) UnitedHealthcare Medicare Committee Current Approval Date 04/08/2015 IMPORTANT NOTE ABOUT THIS REIMBURSEMENT POLICY This policy is applicable
Policy Number IMM10012009SC Approved By Vaccination (Immunization) UnitedHealthcare Medicare Committee Current Approval Date 04/08/2015 IMPORTANT NOTE ABOUT THIS REIMBURSEMENT POLICY This policy is applicable
2015 Subcutaneous Implantable Defibrillator (the S-ICD TM System) Coding Guide Rhythm Management
 2015 Subcutaneous Implantable Defibrillator (the S-ICD TM System) Coding Guide Rhythm Management Contents Physician Coding... 2 Hospital Outpatient Coding and Payment... 3 Ambulatory Surgery Center (ASC)
2015 Subcutaneous Implantable Defibrillator (the S-ICD TM System) Coding Guide Rhythm Management Contents Physician Coding... 2 Hospital Outpatient Coding and Payment... 3 Ambulatory Surgery Center (ASC)
NOVOSTE BETA-CATH SYSTEM
 HOSPITAL INPATIENT AND OUTPATIENT BILLING GUIDE FOR THE NOVOSTE BETA-CATH SYSTEM INTRAVASCULAR BRACHYTHERAPY DEVICE This guide is intended solely for use as a tool to help hospital billing staff resolve
HOSPITAL INPATIENT AND OUTPATIENT BILLING GUIDE FOR THE NOVOSTE BETA-CATH SYSTEM INTRAVASCULAR BRACHYTHERAPY DEVICE This guide is intended solely for use as a tool to help hospital billing staff resolve
Kimberly Anderson-Erisman, PhD Director of Education University of Miami & Miami Project to Cure Paralysis
 Webinar title: Direct Nerve Stimulation What is it really and what is the potential? Presenter/presenters: Kimberly Anderson-Erisman, PhD Director of Education University of Miami & Miami Project to Cure
Webinar title: Direct Nerve Stimulation What is it really and what is the potential? Presenter/presenters: Kimberly Anderson-Erisman, PhD Director of Education University of Miami & Miami Project to Cure
Instructions & Forms for Submitting Claims to Medicare
 Instructions & Forms for Submitting Claims to Medicare The Centers for Medicare and Medicaid services have issued a national coverage policy for the WalkAide. CMS will cover patients who have a diagnosis
Instructions & Forms for Submitting Claims to Medicare The Centers for Medicare and Medicaid services have issued a national coverage policy for the WalkAide. CMS will cover patients who have a diagnosis
istent Trabecular Micro-Bypass Stent Reimbursement Guide
 istent Trabecular Micro-Bypass Stent Reimbursement Guide Table of Contents Overview Coding 3 4 Coding Overview Procedure Coding Device Coding Additional Coding Information Coverage Payment 10 11 Payment
istent Trabecular Micro-Bypass Stent Reimbursement Guide Table of Contents Overview Coding 3 4 Coding Overview Procedure Coding Device Coding Additional Coding Information Coverage Payment 10 11 Payment
ESSURE REIMBURSEMENT GUIDE
 ESSURE REIMBURSEMENT GUIDE A CODING AND COVERAGE RESOURCE Indication Essure is indicated for women who desire permanent birth control (female sterilization) by bilateral occlusion of the fallopian tubes.
ESSURE REIMBURSEMENT GUIDE A CODING AND COVERAGE RESOURCE Indication Essure is indicated for women who desire permanent birth control (female sterilization) by bilateral occlusion of the fallopian tubes.
UnitedHealthcare Medicare Solutions Notification/Prior Authorization Requirements Effective Jan. 1, 2016
 This list represents our advance notification/prior authorization review requirements as referenced in the UnitedHealthcare Physician, Health Care Professional, Facility and Ancillary Provider 2016 Administrative
This list represents our advance notification/prior authorization review requirements as referenced in the UnitedHealthcare Physician, Health Care Professional, Facility and Ancillary Provider 2016 Administrative
Medicare 101: Basics of CPT. Part B Provider Outreach and Education February 11, 2015
 Medicare 101: Basics of CPT Part B Provider Outreach and Education February 11, 2015 Housekeeping Tips When you called in, did you enter your attendee code? Dial-in number: 1-800-791-2345 Attendee (participant)
Medicare 101: Basics of CPT Part B Provider Outreach and Education February 11, 2015 Housekeeping Tips When you called in, did you enter your attendee code? Dial-in number: 1-800-791-2345 Attendee (participant)
Coding for the Contraceptive Implant and IUDs
 LARC Quick Coding Guide 2012 UPDATE Coding for the Contraceptive Implant and IUDs CRECT CODING can result in more appropriate compensation for services. To help practices receive appropriate payment for
LARC Quick Coding Guide 2012 UPDATE Coding for the Contraceptive Implant and IUDs CRECT CODING can result in more appropriate compensation for services. To help practices receive appropriate payment for
NeuroPace. RNS System Patient Manual
 NeuroPace RNS System Patient Manual CAUTION: Federal law restricts this device to sale by or on the order of a physician. 01/2013 DN 1013555 Rev 2 1 2013 NeuroPace, Inc. 2 This Manual supports: RNS Neurostimulator
NeuroPace RNS System Patient Manual CAUTION: Federal law restricts this device to sale by or on the order of a physician. 01/2013 DN 1013555 Rev 2 1 2013 NeuroPace, Inc. 2 This Manual supports: RNS Neurostimulator
SAME DAY/SAME SERVICE
 SAME DAY/SAME SERVICE REIMBURSEMENT POLICY Policy Number: ADMINISTRATIVE 7. T0 Effective Date: June, 20 Table of Contents APPLICABLE LINES OF BUSINESS/PRODUCTS... APPLICATION... OVERVIEW... REIMBURSEMENT
SAME DAY/SAME SERVICE REIMBURSEMENT POLICY Policy Number: ADMINISTRATIVE 7. T0 Effective Date: June, 20 Table of Contents APPLICABLE LINES OF BUSINESS/PRODUCTS... APPLICATION... OVERVIEW... REIMBURSEMENT
Class Action Settlement Recap
 Class Action Settlement Recap Enhancements to Claim Payment Policy, Processing and Payment Disclosure, and an Appeals Process for Class Action Settlement Providers The following enhancements are effective
Class Action Settlement Recap Enhancements to Claim Payment Policy, Processing and Payment Disclosure, and an Appeals Process for Class Action Settlement Providers The following enhancements are effective
MEDICAL CONTESTED CASE HEARING NO 12090 M6-12-38043-01 DECISION AND ORDER
 MEDICAL CONTESTED CASE HEARING NO 12090 M6-12-38043-01 DECISION AND ORDER This case is decided pursuant to Chapter 410 of the Texas Workers Compensation Act and Rules of the Division of Workers Compensation
MEDICAL CONTESTED CASE HEARING NO 12090 M6-12-38043-01 DECISION AND ORDER This case is decided pursuant to Chapter 410 of the Texas Workers Compensation Act and Rules of the Division of Workers Compensation
Ms. Jackson is the Manager of Health Finance and Reimbursement, Division of Health Policy and Practice Services, Washington, DC.
 Electrodiagnostic Testing with Same Day Evaluation Management By: Shane J. Burr, MD; Scott I. Horn, DO; Jenny J. Jackson, MPH, CPC; Joseph P. Purcell, DO Dr. Burr practices general inpatient and outpatient
Electrodiagnostic Testing with Same Day Evaluation Management By: Shane J. Burr, MD; Scott I. Horn, DO; Jenny J. Jackson, MPH, CPC; Joseph P. Purcell, DO Dr. Burr practices general inpatient and outpatient
