Malignant pleural mesothelioma (MPM) is a malignancy
|
|
|
- Colin Lyons
- 8 years ago
- Views:
Transcription
1 Original Article Is Radiotherapy Useful for Treating Pain in Mesothelioma? A Phase II Trial Nicholas MacLeod, FRCR,* Anthony Chalmers, PhD, Noelle O Rourke, MD, Karen Moore, BSc, Jonathan Sheridan, FRCR, Lynn McMahon, BSc, Caroline Bray, MSc, Jon Stobo, MSc, Alan Price, FRCP, Marie Fallon, MD,* and Barry J. Laird, MD* Introduction: Radiotherapy is often used to treat pain in malignant pleural mesothelioma (MPM), although there is limited evidence to support this. The aim of this trial was to assess the role of radiotherapy for the treatment of pain in MPM. Methods: A multicentre, single arm phase II trial was conducted. Eligible patients fulfilled the following criteria: pathological or radiological diagnosis of MPM; pain secondary to MPM; radiotherapy indicated for pain control; and more than 18 years of age. Patients had assessments of pain and other symptoms at baseline and then received 20 Gy in five daily fractions. Key follow-up points were 5 and 12 weeks posttreatment. The primary end point measure was assessment of pain at the site of radiotherapy at 5 weeks. Secondary end points included effects on quality of life, breathlessness, fatigue, mood, toxicity, and the radiological response. Results: Forty patients were recruited from three UK oncology centers. Fourteen patients had a clinically meaningful improvement in their pain 5 weeks post radiotherapy (intention to treat), with five patients having a complete improvement. On the basis of a complete case analysis of the 30 patients assessable at week 5, 47% (confidence intervals, ) of patients alive at week 5 had an improvement in their pain. There was no improvement in other key symptoms or quality of life. Conclusions: Radiotherapy for pain control in MPM is an effective treatment in a proportion of patients. Future studies examining differing radiotherapy regimens with a view to improving response rates are warranted. *Edinburgh Cancer Research Centre, University of Edinburgh, Edinburgh, United Kingdom; Beatson West of Scotland Cancer Centre, Glasgow, United Kingdom; University of Glasgow, Glasgow, United Kingdom; and European Palliative Care Research Centre, Norwegian University of Science and Technology, Trondheim, Norway. M Fallon and BJ Laird are joint senior authors. The clinical trial was funded by grants from the June Hancock Mesothelioma Research Fund and the Beatson Cancer Charity. We would also like to thank Dr Matthew Hatton. Clinical Trial Registration: ISRCTN Disclosure: The authors declare no conflict of interest. Address for correspondence: Nicholas MacLeod, MRCP, Edinburgh Cancer Research Centre, University of Edinburgh, Edinburgh, United Kingdom. Nicholas.macleod@ggc.scot.nhs.uk DOI: /JTO Copyright 2015 by the International Association for the Study of Lung Cancer. This is an open-access article distributed under the terms of the Creative Commons Attribution-Non Commercial-No Derivatives 3.0 License, where it is permissible to download and share the work provided it is properly cited. The work cannot be changed in any way or used commercially. ISSN: /15/ Key Words: Radiotherapy, Pain, Mesothelioma. (J Thorac Oncol. 2015;10: 00 00) Malignant pleural mesothelioma (MPM) is a malignancy affecting the pleural lining. Exposure to asbestos is the most common etiological factor, and although it is a relatively rare malignancy, the incidence of MPM in the United Kingdom is among the highest in the world and is set to rise further in the next few years. 1,2 Its rarity is in contrast to its clinical effect. The median survival is 12 months, and lack of effective treatments means that it represents one of the main therapeutic challenges in Oncology. 3 5 As a result, the vast majority of patients are symptomatic of the disease and die from the condition. MPM carries a high symptom burden, and pain is the most common. Various analgesics, including strong opioids, nonsteroidal anti-inflammatory drugs, anticonvulsants, local anesthetic agents, and steroids, are used. Despite these, pain control is often suboptimal. 6 Over the last three decades, radiotherapy has been used as a key analgesic modality in MPM. Although this is widely accepted as a therapeutic option, a recent systematic review reported that there is limited evidence to support the role of radiotherapy in treating pain in MPM. 7 Studies to date have used a wide variety of doses and fractionation regimens, which have resulted in no clear consensus on the optimal radiotherapy regimen. Moreover, there have been no prospective studies using validated end points, although a previous study from Glasgow suggested some short-term benefit. 8 As a result, little can be drawn from these data in terms of informing practice. On the basis of the current evidence, which is mainly retrospective, the response rates using radiotherapy to treat pain in MPM vary from 0% to 69%. There is a need for prospective studies using standard radiotherapy regimens and validated end points, which examine radiotherapy for treating pain in MPM. Therefore, the SYSTEMS study was conducted to assess the role of radiotherapy for pain control in MPM using standard radiotherapy regimens, controlled background analgesia, and validated assessment tools. MATERIALS AND METHODS A multicenter, single arm, phase II study was conducted. The study had independent ethics committee approval (UK 12/ Journal of Thoracic Oncology Volume 10, Number 6, June
2 MacLeod et al. Journal of Thoracic Oncology Volume 10, Number 6, June 2015 WS/0134) and was conducted in accordance with the Declaration of Helsinki. It was registered at the ISRCTN database ( ). There was one major protocol modification, which was an amendment to include positron emission tomography computed tomography (PET-CT) scanning to assess a possible role in radiotherapy planning in MPM. These findings will be presented elsewhere. This was discussed and approved by the Trial Steering Committee and occurred after 19 patients had been consented. Patients Eligible patients fulfilled the following criteria: 18 years of age or over; histological or agreed clinical and radiological diagnosis of MPM by thoracic oncology multidisciplinary team; due to receive radiotherapy (indication pain resulting from MPM); Eastern Cooperative Oncology Group performance status 0 2; CT scan within the previous 8 weeks; and had pain 4 of 10 (worst pain in the previous 24 hours) on a 0 10 visual analogue scale. Patients who had received radiotherapy or chemotherapy in the preceding 6 weeks that was likely to alter pain during the duration of the trial or planned chemotherapy during the trial period were excluded. The trial was conducted in three regional cancer centers in the United Kingdom (Beatson West of Scotland Cancer Centre, Edinburgh Cancer Centre, and Weston Park Hospital NHS Trust, Sheffield). Trial Design Before consent, all patients were assessed and had their current analgesia optimized. The aim of this was to stabilize analgesia before radiotherapy, with the purpose of minimizing the likelihood of changes in analgesia during the trial. This was undertaken to enable, where possible, the true effect of radiotherapy to be seen. Consenting patients would have stable but poorly controlled pain, necessitating the need for radiotherapy. After obtaining written informed consent, all baseline assessments were performed. Patients then underwent radiotherapy planning and subsequently started treatment a maximum of 7 days from trial baseline. Radiotherapy Wire markings were placed on the skin to outline the outer limits of the painful area. Radiotherapy was planned using CT simulation either by CT field placement or by voluming of the tumor using an Eclipse workstation. All planning CTs were captured on LightSpeed Simulator LS RT 16 GE Medical CT scanner (GE Medical systems, Crawley, United Kingdom) using a 120 kv automatic ma modulation range of mas with 50 cm Dual Field of View. When the target area was contoured, the gross tumor volume was defined as the volume of tumor, which was felt to be responsible for the pain. This was then grown to a planning target volume (PTV) by 1 2 cm, subject to the discretion of the treating clinician. All patients were planned to receive 20 Gy in five fractions to the area of pain. During the trial, assessments were performed at weeks 1, 5, and 12 after the start of radiotherapy. Patients were also contacted by telephone every week to monitor for any adverse events and assess analgesia. End Points The primary end point was level of pain 5 weeks after radiotherapy. An improvement in pain was defined as a 30% reduction in pain from baseline. Pain was assessed using the brief pain inventory (BPI), which has been extensively validated in cancer pain. An improvement of 30% is accepted as a clinically meaningful improvement in pain in studies of analgesic interventions. 9 Opioid analgesic use was recorded and is reported in morphine equivalent daily dose (MEDD) to allow comparison between different opioid types and to enable the effect of changes in analgesia with respect to changes in pain after radiotherapy. Secondary end points assessed the effect of radiotherapy at weeks 1, 5, and 12 on the following: pain response at weeks 1 and 12; dyspnea assessed using a numerical rating scale; mood assessed using the Hospital Anxiety and Depression Scale; quality of life (QoL) using the EORTC QLQ-30; fatigue using the Fatigue Severity Scale (FSS); night sweats assessed using an numerical rating scale; radiotherapy toxicity as per common toxicity criteria for adverse events version 4.0; and the effect of radiotherapy on tumor bulk assessed by comparing CT scans at week 12 with baseline CT using the modified response evaluation criteria in solid tumors. 10 Statistical Considerations The statistical analysis was based on the objective of showing the proportion of patients for whom radiotherapy was an effective means of treating pain in MPM at 5 weeks post radiotherapy. A sample size of 40 patients was calculated to enable the proportion of responders to be estimated within 15%, depending on the true underlying proportion. An intention to treat (ITT) approach was used. The secondary end points were exploratory in nature. All statistical analyses were performed using SPSS version 21.0 (Chicago, IL). Unless otherwise stated, means and standard deviations (SD) are used, and 95% confidence intervals (CI) are reported throughout. Patients who met the primary end point are termed responders and patients who did not are termed nonresponders. RESULTS Patient disposition is shown in the CONSORT diagram in Figure 1. From June 2012 to December 2013, 40 patients consented to the trial. Of these, 37 patients started radiotherapy, with 35 completing their prescribed course. All sites of pain were in the chest. Patient demographics are shown in Table 1. Thirty-five patients were male, and the median age (interquartile range [IQR]) was ( ) years. The most common histological type was epithelioid in 21 patients (56.8%), and the majority of patients were performance status 1 or 2. The median survival from the time of trial registration was 93 days (CI, ). However, this differed depending on histological subtypes epithelioid 124 days (83 165) versus sarcomatoid 65 (37 93), p = The mean (SD) time from initial diagnosis to study entry was days (274.16). The median (IQR) baseline BPI score was 57 ( ), and the median (IQR) baseline opioid dose was 55 mg (25 210). 2 Copyright 2015 by the International Association for the Study of Lung Cancer
3 Journal of Thoracic Oncology Volume 10, Number 6, June 2015 Using Radiotherapy to Treat Pain in Mesothelioma FIGURE 1. Patient disposition. At the time of primary end point assessment (week 5), 30 patients were evaluable. Three patients did not start radiotherapy, two patients failed to complete radiotherapy, two patients had died before week 5, and three further patients had deteriorated to the point that they were no longer able to complete the assessment (Fig. 1). The primary end point, based on an ITT analysis, was met by 14 patients (35%) who had a clinically significant improvement in pain 5 weeks post radiotherapy. Nine patients (22.5%) had an improvement of 60% in BPI score with five patients (12.5%) having a complete response (100% improvement in BPI). Therefore, based on a complete case analysis of 30 evaluable patients at week 5, 47% (CI, ) of patients responded to the radiotherapy. Of the 14 patients who responded to radiotherapy, eight had epithelioid histology, four had sarcomatoid, and two had mixed histology. As a percentage of the total number of each of these histological subtypes, 38% of epithelioid patients responded, 40% of sarcomatoid, and 66.6% of mixed histology. There was no statistically significant difference between histological subtypes in terms of response. At weeks 1 and 12, the pain response rate was 27.5% (CI, 14 6% to 43.9%) and 15.0% (CI, %), respectively, on an ITT analysis. On the basis of a complete case analysis, the proportion of pain responders at week 1 was 36.7% (CI, %) and at week 12 was 33.3% (CI, %). Although 32 patients completed the week 1 assessment, two of them had incomplete data and so were not evaluable. Eighteen patients were evaluable at week 12. Figure 2 shows changes in opioid dose and pain (BPI) per responder status. Only four responders had an increase in their opioid dose between study baseline and end point, and in only one patient was this >20 mg (MEDD). There was no difference in mean opioid dose between baseline and end point in the responders, p = There was no difference in the percentage change from baseline MEDD at week 1 (p = 0.577) or week 5 (p = 0.355) between responders and nonresponders. Of the 14 responders, nine were on simple and eight on adjuvant analgesics at baseline. A similar proportion (16 of 24) of the nonresponders was on simple analgesics at baseline. Although a slightly higher proportion (17 of 24) of nonresponders was on adjuvant analgesics at baseline compared with responders, this was not statistically significant, p = Throughout the duration of the study, only one patient was started on an adjuvant analgesic. Therefore, the improvement in pain is likely due to radiotherapy rather than analgesia. There was no change in global QoL for patients throughout the study when they are taken as a whole group. However, there was a trend suggesting an improvement in QoL in responders and a decline in global QoL in nonresponders, although this was not statistically significant. The Copyright 2015 by the International Association for the Study of Lung Cancer 3
4 MacLeod et al. Journal of Thoracic Oncology Volume 10, Number 6, June 2015 TABLE 1. Patient Demographics Characteristic n % Mean SD Median IQR Age Male ECOG Time from diagnosis to trial entry (days) Mesothelioma Histology Epithelioid Sarcomatoid Mixed Other Not available Metastases Present Absent Unknown Previous anticancer therapy for MPM Chemotherapy Radiotherapy SD, standard deviation; IQR, interquartile range; ECOG, Eastern Cooperative Oncology Group. median improvement in QoL in responders was (IQR, to 41.67) compared with a median decline of (IQR, to 0.00) in nonresponders. In terms of specific QoL components, there was a worsening of fatigue, appetite loss, and nausea/vomiting scores. Fatigue scores at week 12 (p = 0.040) and nausea/vomiting at week 1 (p = 0.017) had significantly increased. There were, however, improvements in pain, dyspnea, insomnia, and constipation. Pain scores at weeks 1 (p = 0.005) and 5 (p = 0.034) and dyspnea at week 1 (p = 0.037) were significantly lower. There was no significant difference between responders and nonresponders in the change in dyspnea score at week 5 (responders: median change, 0; IQR, 33 to 33; nonresponders: 16.67, IQR 33 to 0; p = 0.203). The greater improvement in nonresponders may be due to baseline dyspnea score being higher than for responders, although not significantly so (p = 0.148). Only one patient had a cough, which was recorded as grade 2 at all times including baseline. The effect of radiotherapy on other key symptoms is shown in Table 2. There was no significant change in any other secondary end points with the exception of night sweats, which improved by week 5 (p = 0.01). In Table 3, the percentage of patients with the most common symptoms and likely side effects from radiotherapy are reported. Only one patient had a delay in delivery of their radiotherapy because of radiotherapy induced odynophagia, however, this patient had an apical tumor, and their larynx was within the radiotherapy field. The patient had been given additional analgesia as prophylactic cover but had not taken it. On commencing the analgesia, the patient s odynophagia improved considerably, and he completed the radiotherapy. Changes in disease bulk, assessed using CT, are shown in Table 4. Only 18 patients were alive and well enough to undergo the week 12 CT. Of these, there was only one partial response as assessed by the modified response evaluation criteria in solid tumors criteria for assessment of response in MPM. 10 None of the five patients who had radiological progressive disease had a drop in their BPI score. The median PTV was cm 3 (IQR, ). There was no difference between the median PTV for responders cm 3 (IQR ) and non responders cm 3 (IQR, ) suggesting that the size of the PTV does not correlate with the magnitude of response. Median survival of responders was 106 days (95% CI, days). Median survival was slightly lower in nonresponders at 93 days (95% CI, days), but the difference was not statistically significant (p = 0.465). DISCUSSION This is the largest trial to date that examines the role of radiotherapy in MPM and the first to use validated assessment tools in this setting. The findings support that radiotherapy is an effective treatment for a proportion of patients with MPM-related pain with 35% of assessable patients experiencing a clinically meaningful improvement in their pain. Of these, 12.5% had a complete improvement in their pain. There was no association between pain response and 4 Copyright 2015 by the International Association for the Study of Lung Cancer
5 Journal of Thoracic Oncology Volume 10, Number 6, June 2015 Using Radiotherapy to Treat Pain in Mesothelioma FIGURE 2. A, Waterfall plot of percentage change from baseline to week 5 in total BPI score and (B) corresponding raw change from baseline to week 5 in morphine equivalent daily dose. The dotted line indicates a 30% reduction from baseline BPI score, the response criterion. Note, a further 10 patients were classed as nonresponders, but have missing baseline and/or week 5 BPI total score. improvement in any other symptoms, and therefore, palliative radiotherapy in MPM should only be considered for pain control. There has been a lack of previous work in this area, with which to compare our findings. The only other prospective study that has examined radiotherapy in MPM reported 22 patients who were treated with hemithoracic irradiation using Cobalt-60 machines at a dose of 30 Gy in 10 fractions. Pain scores improved in 13 patients 1 month after radiotherapy with no increase in analgesic requirements. Validated pain assessment tools were not used in this study, though none were available at the time. 8 The findings of the present study are, therefore, of interest and provide evidence to support radiotherapy as a useful modality for treating pain in MPM. Of particular note was that in 12.5% of patients a complete analgesic response was noted. This provides grounds for optimism for future work in this area. Future work should examine dose escalation, both in terms of total dose and dose per fraction, because the likelihood of improving outcomes by increasing dose TABLE 2. Symptom Assessments Between Trial Baseline and Other Timepoints Symptom (score) Baseline mean (SD) Week 1 mean (SD) P Value Week 5 mean (SD) P Value Week 12 mean (SD) P Value Dyspnea (0 10) 4.46 (2.47) 4.19 (2.84) (2.35) (3.08) Sweats (0 10) 3.44 (3.58) 3.16 (3.29) (3.02) (2.74) HADS anxiety (0 21) 5.86 (4.17) 4.90 (4.15) (4.58) (4.93) HADS depression (0 21) 6.86 (3.41) 6.97 (4.09) (4.15) (3.55) Fatigue (0 63) (11.88) (13.71) (11.29) > (13.76) HADS, hospital anxiety and depression scale. Copyright 2015 by the International Association for the Study of Lung Cancer 5
6 MacLeod et al. Journal of Thoracic Oncology Volume 10, Number 6, June 2015 TABLE 3. Common Toxicity Criteria for Adverse Event Grades at Each Trial Timepoint Baseline Week 1 Week 5 Week 12 n % n % n % n % Anorexia Grade 0/ Grade Grade Dyspnea Grade 0/ Grade Grade Fatigue Grade 0/ Grade Grade Not assessed/ deceased Hyperhidrosis Grade 0/ Grade Pain Grade 0/ Grade Grade Pleuritic pain Grade 0/ Grade Grade per fraction varies between tumor types and is determined by the α/β ratio of the tumor. Although rapidly proliferating squamous cell carcinomas, such as head and neck or cervical cancer, have high α/β ratios and benefit from treatment with small doses per fraction, many nonsquamous tumors with lower proliferation rates have low α/β ratios and hence benefit from higher doses per fraction. 11,12 Although there are TABLE 4. Computed Tomography Response at 12 Weeks Evaluated as per Modified Recist 1.1 Assessment of overall response in target lesions n % CR PR SD PD Not evaluable Total CR, complete response; PR, partial response; SD, stable disease, PD, progressive disease. little data from which to estimate the α/β ratio for MPM, its nonsquamous histology, relatively low proliferation index, mesenchymal origin, and apparent radioresistance are all consistent with a low α/β value. This hypofractionated approach would also have the advantage of reducing hospital visits and delivering palliative treatment in a timely fashion, which are clearly important in patients with limited survival, as is the limited toxicity seen here. Therefore, dose escalation studies, ideally delivered using advanced radiotherapy techniques, such as intensity modulated radiotherapy, to help provide adequate coverage of bulky areas of disease, while sparing critical normal tissues including lung and would seem the obvious next step. There are certain characteristics of the study population that should be highlighted. Only 35% patients had received prior chemotherapy, which is lower than that would be anticipated for patients with MPM. However, the median age of patients in the study was 71 years compared with 60 years in previous studies examining the use of chemotherapy in MPM. 4,5 Furthermore, an epidemiological study showed that, over a 4-year period, only 54 of 146 patients were considered 6 Copyright 2015 by the International Association for the Study of Lung Cancer
7 Journal of Thoracic Oncology Volume 10, Number 6, June 2015 Using Radiotherapy to Treat Pain in Mesothelioma fit for chemotherapy and of that, only 28 (18%) received chemotherapy. 13 Therefore, the figure reported in the present study appears to be representative of the population from which the study patients were recruited. The percentage of patients in the study with epithelioid histology was 56.8%. This is perhaps lower than that would have been anticipated and is certainly lower than either of the two phase III chemotherapy trials that showed a survival advantage for cisplatin in combination with an antifolate agent. 4,5 Sarcomatoid histology, seen in 27% of patients in this study compared with between 1% and 8% in those chemotherapy studies, is associated with a significantly worse prognosis than epithelioid histology. 5 This would suggest that patients receiving radiotherapy for pain control are a very different population from those studied in previous chemotherapy trials. Despite the improvement in pain, there was no improvement in QoL or other symptoms, although there was a trend toward improved QoL in those who responded to radiotherapy. There may be many explanations for this. Primarily, these patients are near the end of life as shown by the median survival of 3.1 months in this trial. QoL naturally deteriorates during this time. In addition, multiple symptoms coexist in MPM, such as dyspnea and fatigue, and these are unlikely to be influenced by an improvement in pain. Similar results have been found in chemotherapy studies in MPM where no QoL improvements have been observed. 3 This may also reflect a generic problem associated with attempts to study QoL outcomes in patients with advanced cancers in which patient attrition and general deterioration make it very difficult to detect treatment-related changes in QoL. The SYSTEMS trial has limitations. The most obvious of these is the small sample size. However, this was designed as a single arm observational trial, and the main aim was to inform future, larger scale studies; this has been achieved. Another limitation was the very high attrition rate within the trial with only 75% of patients being evaluable 5 weeks after radiotherapy. This highlights the poor survival of these patients and the fact that, by the time most patients with MPM develop significant, uncontrolled chest pain, and they are usually at an advanced stage of their illness. Another potential limitation was the choice of radiotherapy regimen. This is no consensus on the standard radiotherapy technique for treating patients with MPM with palliative intent, so a rather conservative dose and regimen was selected because it was widely used in the study centers. We cannot comment on whether pain improvement persisted beyond 12 weeks as this timepoint was the end of the study. CONCLUSION Palliative radiotherapy can be an effective method for treating pain in MPM, and in a proportion of patients is associated with dramatic improvements in pain. These findings provide a foundation for current practice and highlight that radiotherapy studies in complex tumors, such as MPM, are feasible. Dose escalation studies that aim to increase response rates are now eagerly awaited. REFERENCES 1. McElvenny DM, Darnton AJ, Price MJ, Hodgson JT. Mesothelioma mortality in Great Britain from 1968 to Occup Med (Lond) 2005;55: Hodgson JT, McElvenny DM, Darnton AJ, Price MJ, Peto J. The expected burden of mesothelioma mortality in Great Britain from 2002 to Br J Cancer 2005;92: Muers MF, Stephens RJ, Fisher P, et al.; MS01 Trial Management Group. Active symptom control with or without chemotherapy in the treatment of patients with malignant pleural mesothelioma (MS01): a multicentre randomised trial. Lancet 2008;371: van Meerbeeck JP, Gaafar R, Manegold C, et al. Randomized phase III study of cisplatin with or without raltitrexed in patients with malignant pleural mesothelioma: an intergroup study of the European Organisation for Research and Treatment of Cancer Lung Cancer Group and the National Cancer Institute of Canada. J Clin Oncol 2005; 23: Vogelzang NJ, Rusthoven JJ, Symanowski J, et al. Phase III study of pemetrexed in combination with cisplatin versus cisplatin alone in patients with malignant pleural mesothelioma. J Clin Oncol 2003;21: Price A. What is the role of radiotherapy in malignant pleural mesothelioma? Oncologist 2011;16: Macleod N, Price A, O Rourke N, Fallon M, Laird B. Radiotherapy for the treatment of pain in malignant pleural mesothelioma: a systematic review. Lung Cancer 2014;83: Bissett D, Macbeth FR, Cram I. The role of palliative radiotherapy in malignant mesothelioma. Clin Oncol (R Coll Radiol) 1991;3: Farrar JT, Portenoy RK, Berlin JA, Kinman JL, Strom BL. Defining the clinically important difference in pain outcome measures. Pain 2000;88: Byrne MJ, Nowak AK. Modified RECIST criteria for assessment of response in malignant pleural mesothelioma. Ann Oncol 2004;15: Adelstein DJ, Li Y, Adams GL, et al. An intergroup phase III comparison of standard radiation therapy and two schedules of concurrent chemoradiotherapy in patients with unresectable squamous cell head and neck cancer. J Clin Oncol 2003;21: Rose PG, Bundy BN, Watkins EB, et al. Concurrent cisplatin-based radiotherapy and chemotherapy for locally advanced cervical cancer. N Engl J Med 1999;340: Chapman A, Mulrennan S, Ladd B, Muers MF. Population based epidemiology and prognosis of mesothelioma in Leeds, UK. Thorax 2008;63: Copyright 2015 by the International Association for the Study of Lung Cancer 7
the standard of care 2009 5/1/2009 Mesothelioma: The standard of care take home messages PILC 2006 Jan.vanmeerbeeck@ugent.be Brussels, March 7, 2009
 Mesothelioma: The standard of care Jan.vanmeerbeeck@ugent.be Brussels, March 7, 2009 take home messages PILC 2006 All patients should receive adequate palliation of dyspnea and pain before starting chemotherapy
Mesothelioma: The standard of care Jan.vanmeerbeeck@ugent.be Brussels, March 7, 2009 take home messages PILC 2006 All patients should receive adequate palliation of dyspnea and pain before starting chemotherapy
NCCN Non-Small Cell Lung Cancer V.1.2011 Update Meeting 07/09/10
 Guideline Page and Request NSCL-3 Stage IA, margins positive delete the recommendation for chemoradiation. Stage IB, IIA, margins positive delete the recommendation for chemoradiation + Stage IIA, Stage
Guideline Page and Request NSCL-3 Stage IA, margins positive delete the recommendation for chemoradiation. Stage IB, IIA, margins positive delete the recommendation for chemoradiation + Stage IIA, Stage
L Lang-Lazdunski, A Bille, S Marshall, R Lal, D Landau, J Spicer
 Pleurectomy/decortication, hyperthermic pleural lavage with povidone-iodine and systemic chemotherapy in malignant pleural mesothelioma. A 10-year experience. L Lang-Lazdunski, A Bille, S Marshall, R Lal,
Pleurectomy/decortication, hyperthermic pleural lavage with povidone-iodine and systemic chemotherapy in malignant pleural mesothelioma. A 10-year experience. L Lang-Lazdunski, A Bille, S Marshall, R Lal,
Scottish Medicines Consortium
 Scottish Medicines Consortium pemetrexed 500mg infusion (Alimta ) No. (192/05) Eli Lilly 8 July 2005 The Scottish Medicines Consortium has completed its assessment of the above product and advises NHS
Scottish Medicines Consortium pemetrexed 500mg infusion (Alimta ) No. (192/05) Eli Lilly 8 July 2005 The Scottish Medicines Consortium has completed its assessment of the above product and advises NHS
Malignant Mesothelioma State of the Art
 Malignant Mesothelioma State of the Art Paul Baas The Netherlands Cancer Institute August 12, 2011, Carlsbad, CA Summary Diagnosis; epithelial type subdivided Pleiomorphic vs other Staging: IASLC-IMIG
Malignant Mesothelioma State of the Art Paul Baas The Netherlands Cancer Institute August 12, 2011, Carlsbad, CA Summary Diagnosis; epithelial type subdivided Pleiomorphic vs other Staging: IASLC-IMIG
SMALL CELL LUNG CANCER
 Protocol for Planning and Treatment The process to be followed in the management of: SMALL CELL LUNG CANCER Patient information given at each stage following agreed information pathway 1. DIAGNOSIS New
Protocol for Planning and Treatment The process to be followed in the management of: SMALL CELL LUNG CANCER Patient information given at each stage following agreed information pathway 1. DIAGNOSIS New
Mesothelioma. 1. Introduction. 1.1 General Information and Aetiology
 Mesothelioma 1. Introduction 1.1 General Information and Aetiology Mesotheliomas are tumours that arise from the mesothelial cells of the pleura, peritoneum, pericardium or tunica vaginalis [1]. Most are
Mesothelioma 1. Introduction 1.1 General Information and Aetiology Mesotheliomas are tumours that arise from the mesothelial cells of the pleura, peritoneum, pericardium or tunica vaginalis [1]. Most are
Introduction Objective Methods Results Conclusion
 Introduction Objective Methods Results Conclusion 2 Malignant pleural mesothelioma (MPM) is the most common form of mesothelioma a rare cancer associated with long latency period (i.e. 20 to 40 years),
Introduction Objective Methods Results Conclusion 2 Malignant pleural mesothelioma (MPM) is the most common form of mesothelioma a rare cancer associated with long latency period (i.e. 20 to 40 years),
Disease/Illness GUIDE TO ASBESTOS LUNG CANCER. What Is Asbestos Lung Cancer? www.simpsonmillar.co.uk Telephone 0844 858 3200
 GUIDE TO ASBESTOS LUNG CANCER What Is Asbestos Lung Cancer? Like tobacco smoking, exposure to asbestos can result in the development of lung cancer. Similarly, the risk of developing asbestos induced lung
GUIDE TO ASBESTOS LUNG CANCER What Is Asbestos Lung Cancer? Like tobacco smoking, exposure to asbestos can result in the development of lung cancer. Similarly, the risk of developing asbestos induced lung
Malignant Mesothelioma
 Malignant Malignant mesothelioma is a tumour originating from mesothelial cells. 85 95% of mesotheliomas are caused by asbestos exposure. It occurs much more commonly in the chest (malignant pleural mesothelioma)
Malignant Malignant mesothelioma is a tumour originating from mesothelial cells. 85 95% of mesotheliomas are caused by asbestos exposure. It occurs much more commonly in the chest (malignant pleural mesothelioma)
Malignant Mesothelioma
 Malignant mesothelioma is a tumour originating from mesothelial cells. 85 95% of mesotheliomas are caused by asbestos exposure. It occurs much more commonly in the chest (malignant pleural mesothelioma)
Malignant mesothelioma is a tumour originating from mesothelial cells. 85 95% of mesotheliomas are caused by asbestos exposure. It occurs much more commonly in the chest (malignant pleural mesothelioma)
Activity of pemetrexed in thoracic malignancies
 Activity of pemetrexed in thoracic malignancies Results of phase III clinical studies of pemetrexed in malignant pleural mesothelioma and non-small cell lung cancer show benefit P emetrexed (Alimta) is
Activity of pemetrexed in thoracic malignancies Results of phase III clinical studies of pemetrexed in malignant pleural mesothelioma and non-small cell lung cancer show benefit P emetrexed (Alimta) is
Summary of treatment benefits
 Risk Management Plan PEMETREXED Powder for concentrate for Solution for infusion Pemetrexed is also indicated as monotherapy for the maintenance treatment of locally advanced or metastatic non small cell
Risk Management Plan PEMETREXED Powder for concentrate for Solution for infusion Pemetrexed is also indicated as monotherapy for the maintenance treatment of locally advanced or metastatic non small cell
Report on New Patented Drugs - Alimta
 Report on New Patented Drugs - Alimta Under its transparency initiative, the PMPRB publishes the results of the reviews of new patented drugs by Board Staff, for purposes of applying the PMPRB s Excessive
Report on New Patented Drugs - Alimta Under its transparency initiative, the PMPRB publishes the results of the reviews of new patented drugs by Board Staff, for purposes of applying the PMPRB s Excessive
How To Treat A Cancer With A Radical
 Management of mesothelioma Jan.vanmeerbeeck@ugent.be Amsterdam, March 6, 2010 1 management Palliation Symptomatic care Pain Breathlessness Radiotherapy Chemotherapy Surgery Radical (intention to cure)
Management of mesothelioma Jan.vanmeerbeeck@ugent.be Amsterdam, March 6, 2010 1 management Palliation Symptomatic care Pain Breathlessness Radiotherapy Chemotherapy Surgery Radical (intention to cure)
A new score predicting the survival of patients with spinal cord compression from myeloma
 A new score predicting the survival of patients with spinal cord compression from myeloma (1) Sarah Douglas, Department of Radiation Oncology, University of Lubeck, Germany; sarah_douglas@gmx.de (2) Steven
A new score predicting the survival of patients with spinal cord compression from myeloma (1) Sarah Douglas, Department of Radiation Oncology, University of Lubeck, Germany; sarah_douglas@gmx.de (2) Steven
GUIDELINES FOR THE MANAGEMENT OF LUNG CANCER
 GUIDELINES FOR THE MANAGEMENT OF LUNG CANCER BY Ali Shamseddine, MD (Coordinator); as04@aub.edu.lb Fady Geara, MD Bassem Shabb, MD Ghassan Jamaleddine, MD CLINICAL PRACTICE GUIDELINES FOR THE TREATMENT
GUIDELINES FOR THE MANAGEMENT OF LUNG CANCER BY Ali Shamseddine, MD (Coordinator); as04@aub.edu.lb Fady Geara, MD Bassem Shabb, MD Ghassan Jamaleddine, MD CLINICAL PRACTICE GUIDELINES FOR THE TREATMENT
January 2013 LONDON CANCER NEW DRUGS GROUP RAPID REVIEW. Summary. Contents
 LONDON CANCER NEW DRUGS GROUP RAPID REVIEW Paclitaxel albumin (Abraxane ) as a substitute for docetaxel/paclitaxel for cancer Paclitaxel albumin (Abraxane ) as a substitute for docetaxel/ paclitaxel for
LONDON CANCER NEW DRUGS GROUP RAPID REVIEW Paclitaxel albumin (Abraxane ) as a substitute for docetaxel/paclitaxel for cancer Paclitaxel albumin (Abraxane ) as a substitute for docetaxel/ paclitaxel for
Post-operative intrapleural chemotherapy for mesothelioma
 Post-operative intrapleural chemotherapy for mesothelioma Robert Kratzke, MD John Skoglund Chair for Lung Cancer Research Section of Heme-Onc-Transplant University of Minnesota Medical School Efficacy
Post-operative intrapleural chemotherapy for mesothelioma Robert Kratzke, MD John Skoglund Chair for Lung Cancer Research Section of Heme-Onc-Transplant University of Minnesota Medical School Efficacy
Prior Authorization Guideline
 Prior Authorization Guideline Guideline: PS Inj - Alimta Therapeutic Class: Antineoplastic Agents Therapeutic Sub-Class: Antifolates Client: PS Inj Approval Date: 8/2/2004 Revision Date: 12/5/2006 I. BENEFIT
Prior Authorization Guideline Guideline: PS Inj - Alimta Therapeutic Class: Antineoplastic Agents Therapeutic Sub-Class: Antifolates Client: PS Inj Approval Date: 8/2/2004 Revision Date: 12/5/2006 I. BENEFIT
Adjuvant Therapy Non Small Cell Lung Cancer. Sunil Nagpal MD Director, Thoracic Oncology Jan 30, 2015
 Adjuvant Therapy Non Small Cell Lung Cancer Sunil Nagpal MD Director, Thoracic Oncology Jan 30, 2015 No Disclosures Number of studies Studies Per Month 12 10 8 6 4 2 0 1 2 3 4 5 6 7 8 9 10 11 12 1 2 3
Adjuvant Therapy Non Small Cell Lung Cancer Sunil Nagpal MD Director, Thoracic Oncology Jan 30, 2015 No Disclosures Number of studies Studies Per Month 12 10 8 6 4 2 0 1 2 3 4 5 6 7 8 9 10 11 12 1 2 3
Screening, early referral and treatment for asbestos related cancer
 Screening, early referral and treatment for asbestos related cancer Marc de Perrot, MD, MSc, FRCSC Toronto Mesothelioma Research Program University of Toronto Asbestos related diseases Mesothelioma Lung
Screening, early referral and treatment for asbestos related cancer Marc de Perrot, MD, MSc, FRCSC Toronto Mesothelioma Research Program University of Toronto Asbestos related diseases Mesothelioma Lung
Maintenance therapy in in Metastatic NSCLC. Dr Amit Joshi Associate Professor Dept. Of Medical Oncology Tata Memorial Centre Mumbai
 Maintenance therapy in in Metastatic NSCLC Dr Amit Joshi Associate Professor Dept. Of Medical Oncology Tata Memorial Centre Mumbai Definition of Maintenance therapy The U.S. National Cancer Institute s
Maintenance therapy in in Metastatic NSCLC Dr Amit Joshi Associate Professor Dept. Of Medical Oncology Tata Memorial Centre Mumbai Definition of Maintenance therapy The U.S. National Cancer Institute s
Surgical therapy of. who should be operated
 SAMO Interdisciplinary Workshop on Chest Tumors Lucerne, 13th and 14th January 2012 Surgical therapy of mesothelioma, who should be operated Walter Weder MD Professor of Surgery University Hospital Zurich
SAMO Interdisciplinary Workshop on Chest Tumors Lucerne, 13th and 14th January 2012 Surgical therapy of mesothelioma, who should be operated Walter Weder MD Professor of Surgery University Hospital Zurich
Accelerated hemithoracic radiation followed by extrapleural pneumonectomy for malignant pleural mesothelioma
 Accelerated hemithoracic radiation followed by extrapleural pneumonectomy for malignant pleural mesothelioma Marc de Perrot, Ronald Feld, Natasha B Leighl, Andrew Hope, Thomas K Waddell, Shaf Keshavjee,
Accelerated hemithoracic radiation followed by extrapleural pneumonectomy for malignant pleural mesothelioma Marc de Perrot, Ronald Feld, Natasha B Leighl, Andrew Hope, Thomas K Waddell, Shaf Keshavjee,
TITLE: Comparison of the dosimetric planning of partial breast irradiation with and without the aid of 3D virtual reality simulation (VRS) software.
 SAMPLE CLINICAL RESEARCH APPLICATION ABSTRACT: TITLE: Comparison of the dosimetric planning of partial breast irradiation with and without the aid of 3D virtual reality simulation (VRS) software. Hypothesis:
SAMPLE CLINICAL RESEARCH APPLICATION ABSTRACT: TITLE: Comparison of the dosimetric planning of partial breast irradiation with and without the aid of 3D virtual reality simulation (VRS) software. Hypothesis:
Malignant Pleural Mesothelioma in Singapore
 RESEARCH COMMUNICATION C SP Yip 1, HN Koong 2, CM Loo 3, KW Fong 1* Abstract Aim: To examine the clinical characteristics and outcomes of malignant pleural mesothelioma (MPM) in Singapore. Methods and
RESEARCH COMMUNICATION C SP Yip 1, HN Koong 2, CM Loo 3, KW Fong 1* Abstract Aim: To examine the clinical characteristics and outcomes of malignant pleural mesothelioma (MPM) in Singapore. Methods and
Malignant Mesothelioma: an Update
 Malignant Mesothelioma: an Update Nico van Zandwijk Asbestos Diseases Research Institute Bernie Banton Centre University of Sydney Australia Physicians Week RACP 19-5-2009 Health Risks of Asbestos Fibers
Malignant Mesothelioma: an Update Nico van Zandwijk Asbestos Diseases Research Institute Bernie Banton Centre University of Sydney Australia Physicians Week RACP 19-5-2009 Health Risks of Asbestos Fibers
Research Article Malignant Pleural Mesothelioma Outcomes in the Era of Combined Platinum and Folate Antimetabolite Chemotherapy
 Lung Cancer International Volume 2015, Article ID 590148, 7 pages http://dx.doi.org/10.1155/2015/590148 Research Article Malignant Pleural Mesothelioma Outcomes in the Era of Combined Platinum and Folate
Lung Cancer International Volume 2015, Article ID 590148, 7 pages http://dx.doi.org/10.1155/2015/590148 Research Article Malignant Pleural Mesothelioma Outcomes in the Era of Combined Platinum and Folate
REPORT ASCO 2002 ORLANDO : LUNG CANCER Johan F. Vansteenkiste, MD, PhD, Univ. Hospital and Leuven Lung Cancer Group
 REPORT ASCO 2002 ORLANDO : LUNG CANCER Johan F. Vansteenkiste, MD, PhD, Univ. Hospital and Leuven Lung Cancer Group In the 2002 edition of the ASCO meeting, a total of 315 abstracts in the field of respiratory
REPORT ASCO 2002 ORLANDO : LUNG CANCER Johan F. Vansteenkiste, MD, PhD, Univ. Hospital and Leuven Lung Cancer Group In the 2002 edition of the ASCO meeting, a total of 315 abstracts in the field of respiratory
Mesothelioma. Malignant Pleural Mesothelioma
 Mesothelioma William G. Richards, PhD Brigham and Women s Hospital Malignant Pleural Mesothelioma 2,000-3,000 cases per year (USA) Increasing incidence Asbestos (50-80%, decreasing) 30-40 year latency
Mesothelioma William G. Richards, PhD Brigham and Women s Hospital Malignant Pleural Mesothelioma 2,000-3,000 cases per year (USA) Increasing incidence Asbestos (50-80%, decreasing) 30-40 year latency
Radiotherapy in locally advanced & metastatic NSC lung cancer
 Radiotherapy in locally advanced & metastatic NSC lung cancer Dr Raj Hegde. MD. FRANZCR Consultant Radiation Oncologist. William Buckland Radiotherapy Centre. Latrobe Regional Hospital. Locally advanced
Radiotherapy in locally advanced & metastatic NSC lung cancer Dr Raj Hegde. MD. FRANZCR Consultant Radiation Oncologist. William Buckland Radiotherapy Centre. Latrobe Regional Hospital. Locally advanced
Clinical cases in Malignant Pleural Mesothelioma: Adherence to the ESMO Clinical Practice Guidelines
 Clinical cases in Malignant Pleural Mesothelioma: Adherence to the ESMO Clinical Practice Guidelines Wieneke Buikhuisen The Netherlands Cancer Institute Amsterdam The Netherlands Case (1) Male, 56 year
Clinical cases in Malignant Pleural Mesothelioma: Adherence to the ESMO Clinical Practice Guidelines Wieneke Buikhuisen The Netherlands Cancer Institute Amsterdam The Netherlands Case (1) Male, 56 year
Malignant pleural mesothelioma P/D vs. EPP
 3 rd International Thoracic Oncology Congress Dresden, September 13 15, 2012 Malignant pleural mesothelioma P/D vs. EPP Walter Weder, MD Professor of Surgery Dokumentenname Datum Seite 1 Extrapleural Pneumonectomy
3 rd International Thoracic Oncology Congress Dresden, September 13 15, 2012 Malignant pleural mesothelioma P/D vs. EPP Walter Weder, MD Professor of Surgery Dokumentenname Datum Seite 1 Extrapleural Pneumonectomy
Radiation Therapy in the Treatment of
 Lung Cancer Radiation Therapy in the Treatment of Lung Cancer JMAJ 46(12): 537 541, 2003 Kazushige HAYAKAWA Professor and Chairman, Department of Radiology, Kitasato University School of Medicine Abstract:
Lung Cancer Radiation Therapy in the Treatment of Lung Cancer JMAJ 46(12): 537 541, 2003 Kazushige HAYAKAWA Professor and Chairman, Department of Radiology, Kitasato University School of Medicine Abstract:
Radioterapia panencefalica. Umberto Ricardi
 Radioterapia panencefalica Umberto Ricardi Background Systemic disease to the brain is unfortunately a quite common event Radiotherapy, especially with the great technical development during the past decades,
Radioterapia panencefalica Umberto Ricardi Background Systemic disease to the brain is unfortunately a quite common event Radiotherapy, especially with the great technical development during the past decades,
Clinical Spotlight in Breast Cancer
 2015 European Oncology Congress in Vienna Clinical Spotlight in Breast Cancer Reference Slide Deck Abstract #1815 Impact of Palbociclib Plus Fulvestrant on Global QOL, Functioning, and Symptoms Compared
2015 European Oncology Congress in Vienna Clinical Spotlight in Breast Cancer Reference Slide Deck Abstract #1815 Impact of Palbociclib Plus Fulvestrant on Global QOL, Functioning, and Symptoms Compared
THE SECRETS OF OUR SUCCESS
 THE SECRETS OF OUR SUCCESS QUALITY OF LIFE STUDIES OF THE NCIC Andrea Bezjak, MDCM, MSc,, FRCPC Chair, NCIC CTG QOL Committee Outline of the Presentation Can we consider NCIC CTG QOL activities a success?
THE SECRETS OF OUR SUCCESS QUALITY OF LIFE STUDIES OF THE NCIC Andrea Bezjak, MDCM, MSc,, FRCPC Chair, NCIC CTG QOL Committee Outline of the Presentation Can we consider NCIC CTG QOL activities a success?
Temporal Trends in Demographics and Overall Survival of Non Small-Cell Lung Cancer Patients at Moffitt Cancer Center From 1986 to 2008
 Special Report Temporal Trends in Demographics and Overall Survival of Non Small-Cell Lung Cancer Patients at Moffitt Cancer Center From 1986 to 2008 Matthew B. Schabath, PhD, Zachary J. Thompson, PhD,
Special Report Temporal Trends in Demographics and Overall Survival of Non Small-Cell Lung Cancer Patients at Moffitt Cancer Center From 1986 to 2008 Matthew B. Schabath, PhD, Zachary J. Thompson, PhD,
Cancer Treatments Subcommittee of PTAC Meeting held 18 September 2015. (minutes for web publishing)
 Cancer Treatments Subcommittee of PTAC Meeting held 18 September 2015 (minutes for web publishing) Cancer Treatments Subcommittee minutes are published in accordance with the Terms of Reference for the
Cancer Treatments Subcommittee of PTAC Meeting held 18 September 2015 (minutes for web publishing) Cancer Treatments Subcommittee minutes are published in accordance with the Terms of Reference for the
Kanıt: Klinik çalışmalarda ZYTIGA
 mkdpk de Sonunda Gerçek İlerleme! Kanıt: Klinik çalışmalarda ZYTIGA Dr. Sevil Bavbek 5. Türk Tıbbi Onkoloji Kongresi Mart 214, Antalya Endocrine therapies Adrenals Testis Abiraterone Orteronel Androgen
mkdpk de Sonunda Gerçek İlerleme! Kanıt: Klinik çalışmalarda ZYTIGA Dr. Sevil Bavbek 5. Türk Tıbbi Onkoloji Kongresi Mart 214, Antalya Endocrine therapies Adrenals Testis Abiraterone Orteronel Androgen
How To Treat Mesothelioma
 Mesothelioma in the UK Liz Darlison Mesothelioma UK The National Macmillan Mesothelioma Resource Centre June 2010 Mesothelioma Applied Research Foundation - 2010 www.curemeso.org Contents Size of Mesothelioma
Mesothelioma in the UK Liz Darlison Mesothelioma UK The National Macmillan Mesothelioma Resource Centre June 2010 Mesothelioma Applied Research Foundation - 2010 www.curemeso.org Contents Size of Mesothelioma
Principal Investigator: Valerie W. Rusch, MD, FACS, Chief, Thoracic Surgery Memorial Sloan-Kettering Cancer Center
 Protocol 1101-1088 Phase I study of intra-pleural administration of GL-ONC1 in patients with malignant pleural effusion: primary, metastases and mesothelioma Principal Investigator: Valerie W. Rusch, MD,
Protocol 1101-1088 Phase I study of intra-pleural administration of GL-ONC1 in patients with malignant pleural effusion: primary, metastases and mesothelioma Principal Investigator: Valerie W. Rusch, MD,
بسم هللا الرحمن الرحيم
 بسم هللا الرحمن الرحيم Updates in Mesothelioma By Samieh Amer, MD Professor of Cardiothoracic Surgery Faculty of Medicine, Cairo University History Wagner and his colleagues (1960) 33 cases of mesothelioma
بسم هللا الرحمن الرحيم Updates in Mesothelioma By Samieh Amer, MD Professor of Cardiothoracic Surgery Faculty of Medicine, Cairo University History Wagner and his colleagues (1960) 33 cases of mesothelioma
Goals and Objectives: Breast Cancer Service Department of Radiation Oncology
 Goals and Objectives: Breast Cancer Service Department of Radiation Oncology The breast cancer service provides training in the diagnosis, management, treatment, and follow-up of breast malignancies, including
Goals and Objectives: Breast Cancer Service Department of Radiation Oncology The breast cancer service provides training in the diagnosis, management, treatment, and follow-up of breast malignancies, including
Population-based survival for malignant mesothelioma after introduction of novel chemotherapy
 Eur Respir J 2012; 40: 185 189 DOI: 10.1183/09031936.00153611 CopyrightßERS 2012 Population-based survival for malignant mesothelioma after introduction of novel chemotherapy Ronald A.M. Damhuis*, Caroline
Eur Respir J 2012; 40: 185 189 DOI: 10.1183/09031936.00153611 CopyrightßERS 2012 Population-based survival for malignant mesothelioma after introduction of novel chemotherapy Ronald A.M. Damhuis*, Caroline
Everolimus plus exemestane for second-line endocrine treatment of oestrogen receptor positive metastatic breast cancer
 LONDON CANCER NEWS DRUGS GROUP RAPID REVIEW Everolimus plus exemestane for second-line endocrine treatment of oestrogen receptor positive metastatic breast cancer Everolimus plus exemestane for second-line
LONDON CANCER NEWS DRUGS GROUP RAPID REVIEW Everolimus plus exemestane for second-line endocrine treatment of oestrogen receptor positive metastatic breast cancer Everolimus plus exemestane for second-line
How valuable is a cancer therapy? It depends on who you ask.
 How valuable is a cancer therapy? It depends on who you ask. Comparing and contrasting the ESMO Magnitude of Clinical Benefit Scale with the ASCO Value Framework in Cancer Ram Subramanian Kevin Schorr
How valuable is a cancer therapy? It depends on who you ask. Comparing and contrasting the ESMO Magnitude of Clinical Benefit Scale with the ASCO Value Framework in Cancer Ram Subramanian Kevin Schorr
The epidemiology and treatment of mesothelioma in South East England 1985 2002
 The epidemiology and treatment of mesothelioma in South East England 1985 2002 V Mak, E Davies, V Putcha, B Choodari-Oskooei, H Møller King s College London, Thames Cancer Registry, London, UK Correspondence
The epidemiology and treatment of mesothelioma in South East England 1985 2002 V Mak, E Davies, V Putcha, B Choodari-Oskooei, H Møller King s College London, Thames Cancer Registry, London, UK Correspondence
Effects of Chemotherapy on Quality of Life for Patients with Lung Cancer
 CLINICAL STUDY Effects of Chemotherapy on Quality of Life for Patients with Lung Cancer Ahmet Bircan, MD 1 ; M. Bahad r Berktafl, MD 1 ; Hülya Bay z, MD 1 ; Nihal Baflay, MD 1 ; Sema Bircan, MD 2 ; Mine
CLINICAL STUDY Effects of Chemotherapy on Quality of Life for Patients with Lung Cancer Ahmet Bircan, MD 1 ; M. Bahad r Berktafl, MD 1 ; Hülya Bay z, MD 1 ; Nihal Baflay, MD 1 ; Sema Bircan, MD 2 ; Mine
Guidance on competencies for management of Cancer Pain in adults
 Guidance on competencies for management of Cancer Pain in adults Endorsed by: Contents Introduction A: Core competencies for practitioners in Pain Medicine B: Competencies for practitioners in Pain Medicine
Guidance on competencies for management of Cancer Pain in adults Endorsed by: Contents Introduction A: Core competencies for practitioners in Pain Medicine B: Competencies for practitioners in Pain Medicine
Pre-workshop exercise
 Setting research priorities for mesothelioma workshop 10 th November 2014 Pre-workshop exercise Your individual ranking of unanswered questions about the diagnosis, treatment and care of mesothelioma Please
Setting research priorities for mesothelioma workshop 10 th November 2014 Pre-workshop exercise Your individual ranking of unanswered questions about the diagnosis, treatment and care of mesothelioma Please
Seton Medical Center Hepatocellular Carcinoma Patterns of Care Study Rate of Treatment with Chemoembolization 2007 2012 N = 50
 General Data Seton Medical Center Hepatocellular Carcinoma Patterns of Care Study Rate of Treatment with Chemoembolization 2007 2012 N = 50 The vast majority of the patients in this study were diagnosed
General Data Seton Medical Center Hepatocellular Carcinoma Patterns of Care Study Rate of Treatment with Chemoembolization 2007 2012 N = 50 The vast majority of the patients in this study were diagnosed
SBRT (Elekta), 45 Gy in fractions of 3 Gy 3x/week for 5 weeks (N=22) vs.
 Uitgangsvraag 6: Wat is de plaats van stereotactische radiotherapiebehandeling (SBRT) bij HCC patiënten? Primaire studies I Study ID II Method III Patient characteristics IV Intervention(s) V Results primary
Uitgangsvraag 6: Wat is de plaats van stereotactische radiotherapiebehandeling (SBRT) bij HCC patiënten? Primaire studies I Study ID II Method III Patient characteristics IV Intervention(s) V Results primary
Small Cell Lung Cancer
 Small Cell Lung Cancer Types of Lung Cancer Non-small cell carcinoma (NSCC) (87%) Adenocarcinoma (38%) Squamous cell (20%) Large cell (5%) Small cell carcinoma (13%) Small cell lung cancer is virtually
Small Cell Lung Cancer Types of Lung Cancer Non-small cell carcinoma (NSCC) (87%) Adenocarcinoma (38%) Squamous cell (20%) Large cell (5%) Small cell carcinoma (13%) Small cell lung cancer is virtually
How To Prepare A Meeting For A Health Care Conference
 THORACIC ONCOLOGY MULTIDISCIPLINARY TEAM MEETINGS: OPERATIONAL POLICY EXECUTIVE SUMMARY 1. All patients in Lothian with thoracic malignancies should be discussed at designated times in pathway (see App
THORACIC ONCOLOGY MULTIDISCIPLINARY TEAM MEETINGS: OPERATIONAL POLICY EXECUTIVE SUMMARY 1. All patients in Lothian with thoracic malignancies should be discussed at designated times in pathway (see App
Rare Thoracic Tumours
 Rare Thoracic Tumours 1. Epithelial Tumour of Trachea 1 1.1 General Results Table 1. Epithelial Tumours of Trachea: Incidence, Trends, Survival Flemish Region 2001-2010 Both Sexes Incidence Trend EAPC
Rare Thoracic Tumours 1. Epithelial Tumour of Trachea 1 1.1 General Results Table 1. Epithelial Tumours of Trachea: Incidence, Trends, Survival Flemish Region 2001-2010 Both Sexes Incidence Trend EAPC
Management of spinal cord compression
 Management of spinal cord compression (SUMMARY) Main points a) On diagnosis, all patients should receive dexamethasone 10mg IV one dose, then 4mg every 6h. then switched to oral dose and tapered as tolerated
Management of spinal cord compression (SUMMARY) Main points a) On diagnosis, all patients should receive dexamethasone 10mg IV one dose, then 4mg every 6h. then switched to oral dose and tapered as tolerated
Malignant Pleural Diseases Advances Clinicians Should Know F Gleeson
 Malignant Pleural Diseases Advances Clinicians Should Know F Gleeson The following relevant disclosures, conflicts of interest and/ or financial relationships exist related to this presentation: Consultant
Malignant Pleural Diseases Advances Clinicians Should Know F Gleeson The following relevant disclosures, conflicts of interest and/ or financial relationships exist related to this presentation: Consultant
Extrapleural Pneumonectomy for Malignant Mesothelioma: Pro. Joon H. Lee 9/17/2012
 Extrapleural Pneumonectomy for Malignant Mesothelioma: Pro Joon H. Lee 9/17/2012 Malignant Pleural Mesothelioma (Epidemiology) Incidence: 7/mil (Japan) to 40/mil (Australia) Attributed secondary to asbestos
Extrapleural Pneumonectomy for Malignant Mesothelioma: Pro Joon H. Lee 9/17/2012 Malignant Pleural Mesothelioma (Epidemiology) Incidence: 7/mil (Japan) to 40/mil (Australia) Attributed secondary to asbestos
Protein kinase C alpha expression and resistance to neo-adjuvant gemcitabine-containing chemotherapy in non-small cell lung cancer
 Protein kinase C alpha expression and resistance to neo-adjuvant gemcitabine-containing chemotherapy in non-small cell lung cancer Dan Vogl Lay Abstract Early stage non-small cell lung cancer can be cured
Protein kinase C alpha expression and resistance to neo-adjuvant gemcitabine-containing chemotherapy in non-small cell lung cancer Dan Vogl Lay Abstract Early stage non-small cell lung cancer can be cured
SAKK Lung Cancer Group. Current activities and future projects
 SAKK Lung Cancer Group Current activities and future projects SAKK Lung Cancer Group Open group of physicians interested in lung cancer Mostly Medical Oncologists, but also Thoracic Surgeons Radiation
SAKK Lung Cancer Group Current activities and future projects SAKK Lung Cancer Group Open group of physicians interested in lung cancer Mostly Medical Oncologists, but also Thoracic Surgeons Radiation
London Cancer. Mesothelioma Lung Protocols
 London Cancer Mesothelioma Lung Protocols Version 0.9 Contents 1. Staging... 3 2. Mesothelioma Summary of Chemotherapy Protocols... 4 3. Mesothelioma Chemotherapy Protocols... 7 3.1. Pemetrexed (Alimta
London Cancer Mesothelioma Lung Protocols Version 0.9 Contents 1. Staging... 3 2. Mesothelioma Summary of Chemotherapy Protocols... 4 3. Mesothelioma Chemotherapy Protocols... 7 3.1. Pemetrexed (Alimta
Please see the LUCADA data manual v3.1.3, available in the downloads section
 National Lung Cancer Audit Frequently Asked Questions What dataset should be used? Please see the LUCADA data manual v3.1.3, available in the downloads section What does LUCADA stand for? LUCADA stands
National Lung Cancer Audit Frequently Asked Questions What dataset should be used? Please see the LUCADA data manual v3.1.3, available in the downloads section What does LUCADA stand for? LUCADA stands
MesoPDT. Photodynamic Therapy for malignant pleural mesothelioma ONCO-THAI. Image Assisted Laser Therapy for Oncology
 MesoPDT Photodynamic Therapy for malignant pleural mesothelioma ONCO-THAI Image Assisted Laser Therapy for Oncology Unité Inserm ONCO-THAI «Image Assisted Laser Therapy for Oncology» Inserm ONCO-THAI "Image
MesoPDT Photodynamic Therapy for malignant pleural mesothelioma ONCO-THAI Image Assisted Laser Therapy for Oncology Unité Inserm ONCO-THAI «Image Assisted Laser Therapy for Oncology» Inserm ONCO-THAI "Image
Lung Cancer Treatment Guidelines
 Updated June 2014 Derived and updated by consensus of members of the Providence Thoracic Oncology Program with the aid of evidence-based National Comprehensive Cancer Network (NCCN) national guidelines,
Updated June 2014 Derived and updated by consensus of members of the Providence Thoracic Oncology Program with the aid of evidence-based National Comprehensive Cancer Network (NCCN) national guidelines,
Guidance for Industry
 Guidance for Industry Cancer Drug and Biological Products Clinical Data in Marketing Applications U.S. Department of Health and Human Services Food and Drug Administration Center for Drug Evaluation and
Guidance for Industry Cancer Drug and Biological Products Clinical Data in Marketing Applications U.S. Department of Health and Human Services Food and Drug Administration Center for Drug Evaluation and
POLICY A. INDICATIONS
 Alimta (pemetrexed) Line(s) of Business: HMO; PPO; QUEST Integration Akamai Advantage Original Effective Date: 09/01/2007 Current Effective Date: 10/01/2015 POLICY A. INDICATIONS The indications below
Alimta (pemetrexed) Line(s) of Business: HMO; PPO; QUEST Integration Akamai Advantage Original Effective Date: 09/01/2007 Current Effective Date: 10/01/2015 POLICY A. INDICATIONS The indications below
Corso Integrato di Clinica Medica ONCOLOGIA MEDICA AA 2010-2011 LUNG CANCER. VIII. THERAPY. V. SMALL CELL LUNG CANCER Prof.
 Corso Integrato di Clinica Medica ONCOLOGIA MEDICA AA 2010-2011 LUNG CANCER. VIII. THERAPY. V. SMALL CELL LUNG CANCER Prof. Alberto Riccardi SMALL CELL LUNG CARCINOMA Summary of treatment approach * limited
Corso Integrato di Clinica Medica ONCOLOGIA MEDICA AA 2010-2011 LUNG CANCER. VIII. THERAPY. V. SMALL CELL LUNG CANCER Prof. Alberto Riccardi SMALL CELL LUNG CARCINOMA Summary of treatment approach * limited
A new score predicting the survival of patients with spinal cord compression from myeloma
 A new score predicting the survival of patients with spinal cord compression from myeloma (1) Sarah Douglas, Department of Radiation Oncology, University of Lubeck, Germany; sarah_douglas@gmx.de (2) Steven
A new score predicting the survival of patients with spinal cord compression from myeloma (1) Sarah Douglas, Department of Radiation Oncology, University of Lubeck, Germany; sarah_douglas@gmx.de (2) Steven
Sonneveld, P; de Ridder, M; van der Lelie, H; et al. J Clin Oncology, 13 (10) : 2530-2539 Oct 1995
 Comparison of Doxorubicin and Mitoxantrone in the Treatment of Elderly Patients with Advanced Diffuse Non-Hodgkin's Lymphoma Using CHOP Versus CNOP Chemotherapy. Sonneveld, P; de Ridder, M; van der Lelie,
Comparison of Doxorubicin and Mitoxantrone in the Treatment of Elderly Patients with Advanced Diffuse Non-Hodgkin's Lymphoma Using CHOP Versus CNOP Chemotherapy. Sonneveld, P; de Ridder, M; van der Lelie,
Clinical Commissioning Policy: Proton Beam Radiotherapy (High Energy) for Paediatric Cancer Treatment
 Clinical Commissioning Policy: Proton Beam Radiotherapy (High Energy) for Paediatric Cancer Treatment Reference: NHS England xxx/x/x 1 Clinical Commissioning Policy: Proton Beam Radiotherapy (High Energy)
Clinical Commissioning Policy: Proton Beam Radiotherapy (High Energy) for Paediatric Cancer Treatment Reference: NHS England xxx/x/x 1 Clinical Commissioning Policy: Proton Beam Radiotherapy (High Energy)
Clinical Management Guideline Management of locally advanced or recurrent Renal cell carcinoma. Protocol for Planning and Treatment
 Protocol for Planning and Treatment The process to be followed in the management of: LOCALLY ADVANCED OR METASTATIC RENAL CELL CARCINOMA Patient information given at each stage following agreed information
Protocol for Planning and Treatment The process to be followed in the management of: LOCALLY ADVANCED OR METASTATIC RENAL CELL CARCINOMA Patient information given at each stage following agreed information
Clinical Study Report
 An Open, Multi-Center, Phase II Clinical Trial to Evaluate Efficacy and Safety of Taxol (), UFT, and Leucovorin in Patients with Advanced Gastric Cancer Clinical Study Report 4F, No. 156, Jiankang Rd.,
An Open, Multi-Center, Phase II Clinical Trial to Evaluate Efficacy and Safety of Taxol (), UFT, and Leucovorin in Patients with Advanced Gastric Cancer Clinical Study Report 4F, No. 156, Jiankang Rd.,
Advances In Chemotherapy For Hormone Refractory Prostate Cancer. TAX 327 study results & SWOG 99-16 study results presented at ASCO 2004
 Ronald de Wit Rotterdam Cancer Institute The Netherlands Advances In Chemotherapy For Hormone Refractory Prostate Cancer TAX 327 study results & SWOG 99-16 study results presented at Slide 1 Prostate Cancer
Ronald de Wit Rotterdam Cancer Institute The Netherlands Advances In Chemotherapy For Hormone Refractory Prostate Cancer TAX 327 study results & SWOG 99-16 study results presented at Slide 1 Prostate Cancer
7. Prostate cancer in PSA relapse
 7. Prostate cancer in PSA relapse A patient with prostate cancer in PSA relapse is one who, having received a primary treatment with intent to cure, has a raised PSA (prostate-specific antigen) level defined
7. Prostate cancer in PSA relapse A patient with prostate cancer in PSA relapse is one who, having received a primary treatment with intent to cure, has a raised PSA (prostate-specific antigen) level defined
Management of Non-Small Cell Lung Cancer Guide for General Practitioners
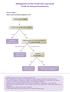 Management of n-small Cell Lung Cancer Guide for General Practitioners Clinical Stage I Cancer only in one lobe of lung and
Management of n-small Cell Lung Cancer Guide for General Practitioners Clinical Stage I Cancer only in one lobe of lung and
Best supportive care: Do we know what it is?
 Best supportive care: Do we know what it is? Angela Boland Rumona Dickson Barbara Jack James Stevenson Edge Hill University Faculty of Health www.liv.ac.uk/lrig Collaborative partners Liverpool Reviews
Best supportive care: Do we know what it is? Angela Boland Rumona Dickson Barbara Jack James Stevenson Edge Hill University Faculty of Health www.liv.ac.uk/lrig Collaborative partners Liverpool Reviews
Background. t 1/2 of 3.7 4.7 days allows once-daily dosing (1.5 mg) with consistent serum concentration 2,3 No interaction with CYP3A4 inhibitors 4
 Abstract No. 4501 Tivozanib versus sorafenib as initial targeted therapy for patients with advanced renal cell carcinoma: Results from a Phase III randomized, open-label, multicenter trial R. Motzer, D.
Abstract No. 4501 Tivozanib versus sorafenib as initial targeted therapy for patients with advanced renal cell carcinoma: Results from a Phase III randomized, open-label, multicenter trial R. Motzer, D.
Columbia University Mesothelioma Applied Research Foundation - 2009 - www.curemeso.org. Mesothelioma Center www.mesocenter.org
 Columbia University Mesothelioma Center www.mesocenter.org Multimodal clinical trials, treatment (surgery, radiation, chemotherapy) Peritoneal mesothelioma program Immunotherapy translational, experimental
Columbia University Mesothelioma Center www.mesocenter.org Multimodal clinical trials, treatment (surgery, radiation, chemotherapy) Peritoneal mesothelioma program Immunotherapy translational, experimental
Mesothelioma mortality in Great Britain 1968-2009. Summary 2. Overall scale of disease including trends 3. Region 6. Occupation 7
 Health and Safety Executive Mesothelioma Mesothelioma mortality in Great Britain 1968-2009 Contents Summary 2 Overall scale of disease including trends 3 Region 6 Occupation 7 Estimation of the future
Health and Safety Executive Mesothelioma Mesothelioma mortality in Great Britain 1968-2009 Contents Summary 2 Overall scale of disease including trends 3 Region 6 Occupation 7 Estimation of the future
REPORT PERSPECTIVES IN LUNG CANCER 2010 AMSTERDAM
 REPORT PERSPECTIVES IN LUNG CANCER 2010 AMSTERDAM Valerie Van Damme, Isabelle Wauters, Johan Vansteenkiste Univ. Hospital Leuven and Leuven Lung Cancer Group Introduction Perspectives in Lung Cancer (PILC)
REPORT PERSPECTIVES IN LUNG CANCER 2010 AMSTERDAM Valerie Van Damme, Isabelle Wauters, Johan Vansteenkiste Univ. Hospital Leuven and Leuven Lung Cancer Group Introduction Perspectives in Lung Cancer (PILC)
Guidance for Industry FDA Approval of New Cancer Treatment Uses for Marketed Drug and Biological Products
 Guidance for Industry FDA Approval of New Cancer Treatment Uses for Marketed Drug and Biological Products U.S. Department of Health and Human Services Food and Drug Administration Center for Drug Evaluation
Guidance for Industry FDA Approval of New Cancer Treatment Uses for Marketed Drug and Biological Products U.S. Department of Health and Human Services Food and Drug Administration Center for Drug Evaluation
Case Number: RT2009-114(M) Potential Audiences: Intent Doctor, Oncology Special Nurse, Resident Doctor
 Extensive-Stage Small Cell Lung Cancer Post Full-Course Chemotherapy with Residual Locoregional Cancer Disease: the Role and Treatment Consideration of Radiotherapy Case Number: RT2009-114(M) Potential
Extensive-Stage Small Cell Lung Cancer Post Full-Course Chemotherapy with Residual Locoregional Cancer Disease: the Role and Treatment Consideration of Radiotherapy Case Number: RT2009-114(M) Potential
4.8 Lung cancer. 4.8.5 In the UK, three fractionation regimens are most commonly used:
 4.8 Lung cancer 4.8.1 In 2005, both NICE (National Institute for Health and Clinical Excellence) and SIGN (Scottish Intercollegiate Guidelines Network) published guidelines on the management of lung cancer.
4.8 Lung cancer 4.8.1 In 2005, both NICE (National Institute for Health and Clinical Excellence) and SIGN (Scottish Intercollegiate Guidelines Network) published guidelines on the management of lung cancer.
Management of low grade glioma s: update on recent trials
 Management of low grade glioma s: update on recent trials M.J. van den Bent The Brain Tumor Center at Erasmus MC Cancer Center Rotterdam, the Netherlands Low grades Female, born 1976 1 st seizure 2005,
Management of low grade glioma s: update on recent trials M.J. van den Bent The Brain Tumor Center at Erasmus MC Cancer Center Rotterdam, the Netherlands Low grades Female, born 1976 1 st seizure 2005,
Current Status and Perspectives of Radiation Therapy for Breast Cancer
 Breast Cancer Current Status and Perspectives of Radiation Therapy for Breast Cancer JMAJ 45(10): 434 439, 2002 Masahiro HIRAOKA, Masaki KOKUBO, Chikako YAMAMOTO and Michihide MITSUMORI Department of Therapeutic
Breast Cancer Current Status and Perspectives of Radiation Therapy for Breast Cancer JMAJ 45(10): 434 439, 2002 Masahiro HIRAOKA, Masaki KOKUBO, Chikako YAMAMOTO and Michihide MITSUMORI Department of Therapeutic
Table of Contents. Data Supplement 1: Summary of ASTRO Guideline Statements. Data Supplement 2: Definition of Terms
 Definitive and Adjuvant Radiotherapy in Locally Advanced Non-Small-Cell Lung Cancer: American Society of Clinical Oncology Clinical Practice Guideline Endorsement of the American Society for Radiation
Definitive and Adjuvant Radiotherapy in Locally Advanced Non-Small-Cell Lung Cancer: American Society of Clinical Oncology Clinical Practice Guideline Endorsement of the American Society for Radiation
ARTICLE IN PRESS. European Journal of Radiology xxx (2009) xxx xxx. Contents lists available at ScienceDirect. European Journal of Radiology
 European Journal of Radiology xxx (2009) xxx xxx Contents lists available at ScienceDirect European Journal of Radiology journal homepage: www.elsevier.com/locate/ejrad Three-dimensional evaluation of
European Journal of Radiology xxx (2009) xxx xxx Contents lists available at ScienceDirect European Journal of Radiology journal homepage: www.elsevier.com/locate/ejrad Three-dimensional evaluation of
Management of Postmenopausal Women with T1 ER+ Tumors: Options and Tradeoffs. Case Study. Surgery. Lumpectomy and Radiation
 Management of Postmenopausal Women with T1 ER+ Tumors: Options and Tradeoffs Michael Alvarado, MD Associate Professor of Surgery University of California San Francisco Case Study 59 yo woman with new palpable
Management of Postmenopausal Women with T1 ER+ Tumors: Options and Tradeoffs Michael Alvarado, MD Associate Professor of Surgery University of California San Francisco Case Study 59 yo woman with new palpable
Radiotherapy in Plasmacytoma and Myeloma. David Cutter Multiple Myeloma NSSG Annual Meeting 14 th September 2015
 Radiotherapy in Plasmacytoma and Myeloma David Cutter Multiple Myeloma NSSG Annual Meeting 14 th September 2015 Contents Indications for radiotherapy: Palliation in Multiple Myeloma Solitary Bone Plasmacytoma
Radiotherapy in Plasmacytoma and Myeloma David Cutter Multiple Myeloma NSSG Annual Meeting 14 th September 2015 Contents Indications for radiotherapy: Palliation in Multiple Myeloma Solitary Bone Plasmacytoma
Articles. Funding Cancer Research UK and the Medical Research Council (UK).
 Active symptom control with or without chemotherapy in the treatment of patients with malignant pleural mesothelioma (MS1): a multicentre randomised trial Martin F Muers, Richard J Stephens, Patricia Fisher,
Active symptom control with or without chemotherapy in the treatment of patients with malignant pleural mesothelioma (MS1): a multicentre randomised trial Martin F Muers, Richard J Stephens, Patricia Fisher,
Before, Frank's immune cells could
 Before, Frank's immune cells could barely recognize a prostate cancer cell. Now, they are focused on it. Stimulate an immune response against advanced prostate cancer Extend median survival beyond 2 years
Before, Frank's immune cells could barely recognize a prostate cancer cell. Now, they are focused on it. Stimulate an immune response against advanced prostate cancer Extend median survival beyond 2 years
Treatment Algorithms for the Management of Lung Cancer in NSW Guide for Clinicians
 Treatment Algorithms for the Management of Lung Cancer in NSW Guide for Clinicians Background The Cancer Institute New South Wales Oncology Group Lung (NSWOG Lung) identified the need for the development
Treatment Algorithms for the Management of Lung Cancer in NSW Guide for Clinicians Background The Cancer Institute New South Wales Oncology Group Lung (NSWOG Lung) identified the need for the development
GRANT APPLICATION FORM: ORGANISING AN ESO EDUCATIONAL EVENT
 GRANT APPLICATION FORM: ORGANISING AN ESO EDUCATIONAL EVENT 1) TOPIC OF THE EVENT MALIGNANT PLEURAL DISEASE 2) SUGGESTED TITLE : Approach to Pleural Cancer: State-of-the-Art 3) TYPE OF EVENT Course Native
GRANT APPLICATION FORM: ORGANISING AN ESO EDUCATIONAL EVENT 1) TOPIC OF THE EVENT MALIGNANT PLEURAL DISEASE 2) SUGGESTED TITLE : Approach to Pleural Cancer: State-of-the-Art 3) TYPE OF EVENT Course Native
BNC105 CANCER CLINICAL TRIALS REACH KEY MILESTONES CLINICAL PROGRAM TO BE EXPANDED
 ASX ANNOUNCEMENT 3 August 2011 ABN 53 075 582 740 BNC105 CANCER CLINICAL TRIALS REACH KEY MILESTONES CLINICAL PROGRAM TO BE EXPANDED Data from renal cancer trial supports progression of the trial: o Combination
ASX ANNOUNCEMENT 3 August 2011 ABN 53 075 582 740 BNC105 CANCER CLINICAL TRIALS REACH KEY MILESTONES CLINICAL PROGRAM TO BE EXPANDED Data from renal cancer trial supports progression of the trial: o Combination
296 cohort patient study. May 2015. Spirometry-monitored deep breathing technique to increase the accuracy of radiotherapy treatment
 breath-hold radiotherapy for breast cancer: Cancer Partners UK s approach to improving outcomes in left-sided breast cancer radiotherapy - an evidence - based review 296 cohort patient study May 2015 Overview
breath-hold radiotherapy for breast cancer: Cancer Partners UK s approach to improving outcomes in left-sided breast cancer radiotherapy - an evidence - based review 296 cohort patient study May 2015 Overview
Objectives. Mylene T. Truong, MD. Malignant Pleural Mesothelioma Background
 Imaging of Pleural Tumors Mylene T. Truong, MD Imaging of Pleural Tumours Mylene T. Truong, M. D. University of Texas M.D. Anderson Cancer Center, Houston, TX Objectives To review tumors involving the
Imaging of Pleural Tumors Mylene T. Truong, MD Imaging of Pleural Tumours Mylene T. Truong, M. D. University of Texas M.D. Anderson Cancer Center, Houston, TX Objectives To review tumors involving the

 Understanding Clinical Trials HR =.6 (CI :.51.7) p
Understanding Clinical Trials HR =.6 (CI :.51.7) p