Selection of Cell Lines for Manufacturing Therapeutic Antibodies by Flow Cytometric Cell Sorting. Andrew Racher Lonza Biologics
|
|
|
- Cathleen Summers
- 8 years ago
- Views:
Transcription
1 Selection of Cell Lines for Manufacturing Therapeutic Antibodies by Flow Cytometric Cell Sorting Andrew Racher Lonza Biologics
2 Structure of Talk 1. The Problem 2. Possible Solutions 3. Solution Chosen 4. Results 5/26/2005 / 2
3 The Problem Organisations developing therapeutic antibody drugs need to complete these activities as rapidly as possible whilst still meeting, for example: Economic criteria, e.g. exceeding a minimum productivity Regulatory criteria, e.g. clonal cell line How can the duration of cell line construction and development, a critical path activity, be reduced yet still meet these other criteria? 5/26/2005 / 3
4 Selection of high producing clonal GS-CHO cell lines Conventional cloning Transfection Suspension evaluation Cloning Suspension evaluation PSS Time (weeks) To isolate high producing GS-CHO cell lines that fit manufacturing process need to evaluate transfectants in suspension culture before cloning Ensures good starting point for cloning Elapsed time is 40 weeks 5/26/2005 / 4
5 Possible solutions How can programme length be shortened? Duration can be reduced by moving straight from the transfection stage to the cloning stage Conventional cloning Transfection Suspension evaluation Cloning Suspension evaluation PSS Time (weeks) 5/26/2005 / 5
6 Consequence of omitting a screening round (1) To ensure several cell lines with the desired growth and productivity characteristics are selected: screen more cell lines in each round Fewer rounds but, as p(good cell line) does not change, need to screen more cell lines per round No. of cell lines = p(good cell line) * no. screened 5/26/2005 / 6
7 Consequence of omitting a screening round (2) Alternatively, the pool of cell lines can be enriched for those with desired characteristics prior to each stage For example: Glutamine Synthetase (GS) Expression System indirectly enriches for high producers at transfection stage by use of the GS inhibitor MSX Use second method to enrich directly for high producers between transfection and cloning stages Antibody (mg/l) µm 50 µm Selection conditions - MSX concentration 5/26/2005 / 7
8 Rapid enrichment of high producer sub-populations Number of flow cytometric-based assays developed that can identify and isolate high producing recombinant cell lines including: Gel Microdrop (Powell & Weaver, 1990, Biotechnol 8:333) Cellular affinity matrix (Holmes & Al-Rubeai, 1999, J Immunol Meth 230:141; Borth et al, 2001, Biotechnol Bioeng 71:266) Membrane trapping of secreted proteins (Brezinsky et al, 2003, J Immunol Meth 277:141) 5/26/2005 / 8
9 Approach taken with GS-cell lines 5/26/2005 / 9
10 Approach taken with GS-cell lines Affinity-matrix surface capture method for identifying high producing cells in transfectant pools Developed from method of Holmes & Al- Rubeai (1999) Fluorescence signal proportional to amount of secreted antibody Sort using FACS to enrich for high producers within population 5/26/2005 / 10
11 Affinity-matrix surface capture (AMSC): construction, capture and detection capture molecule (biotinylated Protein A) free, fluorochrome-labelled detection antibody neutravidin bridge captured, secreted antibody biotinylated-cell surface cell intracellular antibody EP A, Lonza 5/26/2005 / 11
12 Affinity-matrix surface capture (AMSC) biotinylated Protein A fluorochrome-labelled detection antibody neutravidin bridge secreted antibody biotinylated-cell surface cell EP A, Lonza 5/26/2005 / 12
13 Analysis of AMSC-labelled GS-CHO cells producing a recombinant antibody Fluorescent signals for antibody-producing GS-CHO cells were substantially higher than for non-producing cells 5/26/2005 / 13
14 The questions: Does high fluorescence correlate with high productivity? More accurately, specific production rate not product concentration Do cell lines generated by both the AMSC and conventional methods produce similar antibody concentrations in a model of the production process? 5/26/2005 / 14
15 Outline of process flows for AMSC and conventional methods 5/26/2005 / 15
16 Conventional cell line construction method Transfect host cells with vector 96 well plates, single colonies per well cell lines Quantitative productivity assessment Static culture Suspension (Erlenmeyer flask) culture cell lines Adapt to chemically defined medium cell lines 5-10 cell lines Preliminary quantitative assessment Select cell lines to clone Fed-batch assessment of growth, productivity and product quality Clone 5/26/2005 / 16
17 AMSC-cloning cell line construction method Transfect host cells with vector Transfectant pools, T-flasks Analyse pools by AMSC method Bulk sorts Enriched transfectant pools Quantitative productivity assessment Clone Static culture Suspension (Erlenmeyer flask) culture Select cell lines 5-10 cell lines cell lines Preliminary quantitative assessment cell lines Quantitative cell lines productivity assessment Adapt to chemically defined medium Fed-batch assessment of growth, productivity and product quality 5/26/2005 / 17
18 Comparison of AMSC-cloning and conventional methods - experimental approach (1) Conventional AMSC-cloning Starting point: wells with one colony Starting point: transfectant pools 40 wks duration 22 transfection + 18 cloning 31 wks duration Endpoint: clonal, high producing cell lines Endpoint: clonal, high producing cell lines 5/26/2005 / 18
19 Results 5/26/2005 / 19
20 Comparison of AMSC-cloning and conventional methods - experimental approach (2) Cell line CHOK1SV transfected with GS expression vector encoding HC and LC genes for a human IgG Conventional High producing non-clonal cell lines isolated from single, independent transfectant colonies Cell lines cloned to generate high producing clonal cell lines AMSC-cloning Transfectant pools enriched for high producing cell lines Pools for cloning chosen on basis of their SPR value SPR = antibody produced per cell over 48 hour period Productivities of cell lines compared at 4 points 5/26/2005 / 20
21 Does high fluorescence correlate with high productivity? Cell populations sorted from high and low signal regions Sorted populations expanded and SPRs determined The mean SPRs from the two regions are different (p = 0.05) High fluorescence correlates with high productivity Sorting enriched the population for high producers 5/26/2005 / 21
22 Conventional vs. AMSC-cloning: static culture (1) STATIC CULTURE SUSPENSION (Erlenmeyer flask culture) Conventional Transfect host cells with vector cell lines cell lines cell lines 5-10 cell lines Single colonies per well Productivity assessment (quantitative) Adapt to chemicallydefined medium Preliminary productivity assessment (quantitative) Fed-batch assessment of growth, productivity and product quality Select cell lines for cloning STATIC CULTURE SUSPENSION (Erlenmeyer flask culture) AMSC-cloning Transfect host cells with vector Analysis by AMSC Enrich transfectant pools Clone cell lines cell lines cell lines 5-10 cell lines Select cell lines for further analysis Pools in T-flasks Bulk sorts Productivity assessment (quantitative) Productivity assessment (quantitative) Adapt to chemicallydefined medium Preliminary productivity assessment (quantitative) Fed-batch assessment of growth, productivity and product quality 22 + cloning (18) = 40 wks 31 wks 5/26/2005 / 22
23 Conventional vs. AMSC-cloning: static culture (2) AMSC-cloning: populations from high signal region cloned; productivity of clones determined in small-scale static cultures Conventional: transfectants screened to remove low producers; productivity of survivors assessed in small-scale static cultures Are the mean antibody concentration values of the two sets of cell lines generated by the two methods equal? 5/26/2005 / 23
24 Conventional vs. AMSC-cloning: static culture (3) Distributions of productivities from the two methods are similar The mean values for antibody concentrations from the two methods are equal p = /26/2005 / 24
25 Conventional vs. AMSC-cloning: batch, suspension culture (1) STATIC CULTURE SUSPENSION (Erlenmeyer flask culture) Conventional Transfect host cells with vector cell lines cell lines cell lines 5-10 cell lines Single colonies per well Productivity assessment (quantitative) Adapt to chemicallydefined medium Preliminary productivity assessment (quantitative) Fed-batch assessment of growth, productivity and product quality Select cell lines for cloning STATIC CULTURE SUSPENSION (Erlenmeyer flask culture) AMSC-cloning Transfect host cells with vector Analysis by AMSC Enrich transfectant pools Clone cell lines cell lines cell lines 5-10 cell lines Select cell lines for further analysis Pools in T-flasks Bulk sorts Productivity assessment (quantitative) Productivity assessment (quantitative) Adapt to chemicallydefined medium Preliminary productivity assessment (quantitative) Fed-batch assessment of growth, productivity and product quality 22 + cloning (18) = 40 wks 31 wks 5/26/2005 / 25
26 Conventional vs. AMSC-cloning: batch, suspension culture (2) For both methods, antibody concentration in static culture used to rank cell lines. High ranked cell lines progressed Adapted to suspension culture growth Growth and productivity assessed in cultures operated in batch mode 5/26/2005 / 26
27 Conventional vs. AMSC-cloning: batch, suspension culture (3) Distributions of productivities from the two methods are not similar The mean values for antibody concentrations from the two methods are different Conventional 192 mg/l AMSC pool 136 mg/l 5/26/2005 / 27
28 Conventional vs. AMSC-cloning: fed-batch, suspension culture (1) STATIC CULTURE SUSPENSION (Erlenmeyer flask culture) Conventional Transfect host cells with vector cell lines cell lines cell lines 5-10 cell lines Single colonies per well Productivity assessment (quantitative) Adapt to chemicallydefined medium Preliminary productivity assessment (quantitative) Fed-batch assessment of growth, productivity and product quality Select cell lines for cloning STATIC CULTURE SUSPENSION (Erlenmeyer flask culture) AMSC-cloning Transfect host cells with vector Analysis by AMSC Enrich transfectant pools Clone cell lines cell lines cell lines 5-10 cell lines Select cell lines for further analysis Pools in T-flasks Bulk sorts Productivity assessment (quantitative) Productivity assessment (quantitative) Adapt to chemicallydefined medium Preliminary productivity assessment (quantitative) Fed-batch assessment of growth, productivity and product quality 22 + cloning (18) = 40 wks 31 wks 5/26/2005 / 28
29 Conventional vs. AMSC-cloning: fed-batch, suspension culture (2) Antibody concentration in batch culture used to rank cell lines High ranked cell lines progressed Growth and productivity assessed in cultures operated in fed-batch mode Key screening stage for selecting cell lines for use in cgmp manufacturing processes Erlenmeyer flask model of production bioreactor Uses same system of medium and feeds 5/26/2005 / 29
30 Conventional vs. AMSC-cloning: fed-batch, suspension culture (3) 5/26/2005 / 30
31 Conventional vs. AMSC-cloning: fed-batch, suspension culture (4) The mean antibody concentrations for sets of cell lines created by the two methods are equal p = 0.57 The mean Qp values for sets of cell lines created by the two methods are equal p = 0.65 Sets of cell lines generated by both the AMSC and conventional methods produced similar antibody concentrations in a model of the production process 5/26/2005 / 31
32 Conventional vs. AMSC-cloning: clonal cell lines (1) STATIC CULTURE SUSPENSION (Erlenmeyer flask culture) Conventional Transfect host cells with vector cell lines cell lines cell lines 5-10 cell lines Single colonies per well Productivity assessment (quantitative) Adapt to chemicallydefined medium Preliminary productivity assessment (quantitative) Fed-batch assessment of growth, productivity and product quality Select cell lines for cloning STATIC CULTURE SUSPENSION (Erlenmeyer flask culture) AMSC-cloning Transfect host cells with vector Analysis by AMSC Enrich transfectant pools Clone cell lines cell lines cell lines 5-10 cell lines Select cell lines for further analysis Pools in T-flasks Bulk sorts Productivity assessment (quantitative) Productivity assessment (quantitative) Adapt to chemicallydefined medium Preliminary productivity assessment (quantitative) Fed-batch assessment of growth, productivity and product quality 22 + cloning (18) = 40 wks 31 wks 5/26/2005 / 32
33 Conventional vs. AMSC-cloning: clonal cell lines (2) Antibody concentration used to rank cell lines in successive screening rounds Static then batch culture High ranked cell lines progressed Growth and productivity assessed in cultures operated in fed-batch mode Key screening stage Erlenmeyer flask model of production bioreactor 5/26/2005 / 33
34 Conventional vs. AMSC-cloning: clonal cell lines (3) 5/26/2005 / 34
35 Conventional vs. AMSC-cloning: fed-batch, suspension culture (4) Relationship between mean antibody concentration for daughter clones and parent does 95% CI for clone set include value of parent? Set A, no Set B, yes Only set A was different to its parent Relationship between mean specific production rate for daughter clones and parent does 95% CI for clone set include value of parent? Set A, yes Set B, yes Both sets were similar to their parents 5/26/2005 / 35
36 Conventional vs. AMSC-cloning: fed-batch, suspension culture (5) Relationship between the mean antibody concentrations for sets of cell lines created by the two methods Conventional = clone set B = AMSC clone set A p [ 0.05 Relationship between the mean specific production rates for sets of cell lines created by the two methods Conventional = clone set A = AMSC clone set B p [ /26/2005 / 36
37 Conventional vs. AMSC-cloning: fed-batch, suspension culture (6) When panels of cell lines were compared in the key screening stage (fed-batch culture), results indicate that: Cloning did not give systematic improvement in productivity characteristics between daughter clones and parent Cloning did not result in systematic improvement of productivity characteristics between clone sets A and B compared and the cell line sets Conventional and AMSC Overall, the data do not indicate a systematic improvement in productivity between clonal cell lines generated by the conventional method and the AMSC-cloning method Exceptions can be found 5/26/2005 / 37
38 Impact of using AMSC-cloning approach Both methods generated a panel of cell lines having similar productivities Enrichment of transfectant pool before cloning has shortened screening step Time saving = 9 weeks Conventional cloning Transfection Suspension evaluation Cloning Suspension evaluation PSS AMSC-cloning Transfection AMSC-sort & expansion Cloning Suspension evaluation PSS Time from start (weeks) 5/26/2005 / 38
39 Summary Cell line construction and selection methods were run in parallel using a conventional protocol and a flow cytometric protocol Cytometric method enriched cell populations for high producers For the key selection stage, evaluation in fed-batch suspension culture, mean antibody concentration and mean specific production rate were similar The productivities of the panels of candidate cell lines were generally similar AMSC-cloning method reduces timeline by 9 weeks Potential for further improvement if qualify FACS for single cell sorting 5/26/2005 / 39
40 Acknowledgements Cell Culture Process Development Group, LB, Slough Assay Development Group, LB, Slough 5/26/2005 / 40
ESTABLISHMENT OF CELL LINES FOR MANUFACTURING RECOMBINANT ANTIBODIES. Dr Andrew Racher
 ESTABLISHMENT OF CELL LINES FOR MANUFACTURING RECOMBINANT ANTIBODIES Dr Andrew Racher Lonza Biologics plc, 2004 Structure of talk Issues Selection of a high producing cell line Selection for process compatibility
ESTABLISHMENT OF CELL LINES FOR MANUFACTURING RECOMBINANT ANTIBODIES Dr Andrew Racher Lonza Biologics plc, 2004 Structure of talk Issues Selection of a high producing cell line Selection for process compatibility
The Importance of Developing a High Yield of Product
 European Antibody Congress Lyon, 3 rd November 2005 The Importance of Developing a High Yield of Product John Birch, Lonza Biologics plc Monoclonal Antibodies A Success Story Fastest growing segment of
European Antibody Congress Lyon, 3 rd November 2005 The Importance of Developing a High Yield of Product John Birch, Lonza Biologics plc Monoclonal Antibodies A Success Story Fastest growing segment of
Improving GS-CHO Cell Line Selection: Reducing Time to Clinic
 Cell Line Development and Engineering 2-6 March 2009, Berlin Improving GS-CHO Cell Line Selection: Reducing Time to Clinic Adrian Haines, 2009 Lonza Biologics plc, Slough, UK Disclaimer Certain matters
Cell Line Development and Engineering 2-6 March 2009, Berlin Improving GS-CHO Cell Line Selection: Reducing Time to Clinic Adrian Haines, 2009 Lonza Biologics plc, Slough, UK Disclaimer Certain matters
The use of FACS for the selection of cell lines with superior productivity characteristics
 ESACT-UK, Cambridge 2007 The use of FACS for the selection of cell lines with superior productivity characteristics Jonathan Welsh Synopsis Why use FACS in bioprocess development Issues regarding use of
ESACT-UK, Cambridge 2007 The use of FACS for the selection of cell lines with superior productivity characteristics Jonathan Welsh Synopsis Why use FACS in bioprocess development Issues regarding use of
Accelerating Antibody Process Development: Exploring the Synergies Between Engineered Host Cells and Process Development
 Pharma&Biotech Accelerating Antibody Process Development: Exploring the Synergies Between Engineered Host Cells and Process Development James Rance PhD Head of Development Services Singapore Lonza Biologics
Pharma&Biotech Accelerating Antibody Process Development: Exploring the Synergies Between Engineered Host Cells and Process Development James Rance PhD Head of Development Services Singapore Lonza Biologics
Secretion Assay Cell Screening Service Gel Microdrop
 Cell Screening Service Gel Microdrop The Cell Screening Service is comprised of five phases: 1) Evaluation of the Cell Line after encapsulation; 2) Assessment of Reagents; 3) Secretion Assay Optimization;
Cell Screening Service Gel Microdrop The Cell Screening Service is comprised of five phases: 1) Evaluation of the Cell Line after encapsulation; 2) Assessment of Reagents; 3) Secretion Assay Optimization;
How To Develop A Cell Line
 Advances in The Development of Biopharmaceuticals The application of modern technologies and services to the development of Biopharmaceutical processes Dr Jonathan H Dempsey Process Science Fellow Invitrogen
Advances in The Development of Biopharmaceuticals The application of modern technologies and services to the development of Biopharmaceutical processes Dr Jonathan H Dempsey Process Science Fellow Invitrogen
EMABling Antibody Production Platform
 EMABling Antibody Production Platform An industrial solution for the production of therapeutic antibodies with high cytotoxic activity B IOMANUFACTURING EMABling : A fully integrated development platform
EMABling Antibody Production Platform An industrial solution for the production of therapeutic antibodies with high cytotoxic activity B IOMANUFACTURING EMABling : A fully integrated development platform
Accelerating drug development to FTIH: Potential of new expression technologies
 Accelerating drug development to FTIH: Potential of new expression technologies Lekan Daramola Associate Director Biopharmaceutical Development, Cell Culture & Fermentation Sciences CMC Strategy Forum
Accelerating drug development to FTIH: Potential of new expression technologies Lekan Daramola Associate Director Biopharmaceutical Development, Cell Culture & Fermentation Sciences CMC Strategy Forum
A customizable ADCC assay service for antibodies & fusion proteins.
 Antibody Dependent Cell- Mediated Cytotoxicity (ADCC) Assay A customizable ADCC assay service for antibodies & fusion proteins. Our ADCC assay service accurately detects cell lysis based on LDH-release.
Antibody Dependent Cell- Mediated Cytotoxicity (ADCC) Assay A customizable ADCC assay service for antibodies & fusion proteins. Our ADCC assay service accurately detects cell lysis based on LDH-release.
Potelligent CHOK1SV. European Antibody Congress 30 Nov - 02 Dec, Geneva, Switzerland. Alison Porter, 2009 Lonza Biologics plc, Slough, UK
 Potelligent European Antibody Congress 30 Nov - 02 Dec, Geneva, Switzerland Alison Porter, 2009 Lonza Biologics plc, Slough, UK Disclaimer Certain matters discussed in this presentation may constitute
Potelligent European Antibody Congress 30 Nov - 02 Dec, Geneva, Switzerland Alison Porter, 2009 Lonza Biologics plc, Slough, UK Disclaimer Certain matters discussed in this presentation may constitute
Exciting Trends in Bioprocessing
 Exciting Trends in Bioprocessing Alfred Doig and Susan Dana Jones, Ph.D. April 20, 2015 BioProcess Technology Consultants, Inc. 12 Gill Street, Suite 5450 Woburn, MA 01801 Exciting Trends in Bioprocessing
Exciting Trends in Bioprocessing Alfred Doig and Susan Dana Jones, Ph.D. April 20, 2015 BioProcess Technology Consultants, Inc. 12 Gill Street, Suite 5450 Woburn, MA 01801 Exciting Trends in Bioprocessing
Introduction to flow cytometry
 Introduction to flow cytometry Flow cytometry is a popular laser-based technology. Discover more with our introduction to flow cytometry. Flow cytometry is now a widely used method for analyzing the expression
Introduction to flow cytometry Flow cytometry is a popular laser-based technology. Discover more with our introduction to flow cytometry. Flow cytometry is now a widely used method for analyzing the expression
Luca Romagnoli, Ph.D. Business Development Manager
 Modelli innovativi di produzione per lo sviluppo di un processo altamente qualitativo di farmaci biologici Luca Romagnoli, Ph.D. Business Development Manager BIOLOGICAL DRUGS - SOURCES Monoclonal antibodies
Modelli innovativi di produzione per lo sviluppo di un processo altamente qualitativo di farmaci biologici Luca Romagnoli, Ph.D. Business Development Manager BIOLOGICAL DRUGS - SOURCES Monoclonal antibodies
HuCAL Custom Monoclonal Antibodies
 HuCAL Custom Monoclonal HuCAL Custom Monoclonal Antibodies Highly Specific, Recombinant Antibodies in 8 Weeks Highly Specific Monoclonal Antibodies in Just 8 Weeks HuCAL PLATINUM (Human Combinatorial Antibody
HuCAL Custom Monoclonal HuCAL Custom Monoclonal Antibodies Highly Specific, Recombinant Antibodies in 8 Weeks Highly Specific Monoclonal Antibodies in Just 8 Weeks HuCAL PLATINUM (Human Combinatorial Antibody
Valentina Gualato, Ph.D. Process Development Scientist
 COMPANY PRESENTATION Quality and Innovation Valentina Gualato, Ph.D. Process Development Scientist MISSION areta international is a biotech company dedicated to the contract development and manufacturing
COMPANY PRESENTATION Quality and Innovation Valentina Gualato, Ph.D. Process Development Scientist MISSION areta international is a biotech company dedicated to the contract development and manufacturing
ELITE Custom Antibody Services
 ELITE Custom Antibody Services ELITE Custom Antibody Services Experience, confidence, and understanding As a manufacturer and service provider, we have the experience, confidence, and understanding to
ELITE Custom Antibody Services ELITE Custom Antibody Services Experience, confidence, and understanding As a manufacturer and service provider, we have the experience, confidence, and understanding to
BioProcessing J O U R N A L. Trends and Developments in BioProcess Technology. Volume 9 Issue 1 ISSN 1538-8786
 A Production of ISBioTech Summer 2010 Issue BioProcessing J O U R N A L Trends and Developments in BioProcess Technology Volume 9 Issue 1 ISSN 1538-8786 Trends & Developments in BioProcess Technology Novel
A Production of ISBioTech Summer 2010 Issue BioProcessing J O U R N A L Trends and Developments in BioProcess Technology Volume 9 Issue 1 ISSN 1538-8786 Trends & Developments in BioProcess Technology Novel
The Leading Gene Through Screen CRO
 The Leading Gene Through Screen CRO Molecular Biology Services Molecular Biology drives modern drug discovery. Drug screening, target validation and structural biology processes frequently require recombinant
The Leading Gene Through Screen CRO Molecular Biology Services Molecular Biology drives modern drug discovery. Drug screening, target validation and structural biology processes frequently require recombinant
Genes to Proteins to Antibodies
 Genes to Proteins to Antibodies About Us Fusion Antibodies is a CRO established in 2001 as a spin-out from Queen s University Belfast. The company building is situated in a charming area of Springbank
Genes to Proteins to Antibodies About Us Fusion Antibodies is a CRO established in 2001 as a spin-out from Queen s University Belfast. The company building is situated in a charming area of Springbank
Towards energy-based dynamic optimization of monoclonal antibody producing GS-NS0 Cultures
 20 th European Symposium on Computer Aided Process Engineering ESCAPE20 S. Pierucci and G. Buzzi Ferraris (Editors) 2010 Elsevier B.. All rights reserved. Towards energy-based dynamic optimization of monoclonal
20 th European Symposium on Computer Aided Process Engineering ESCAPE20 S. Pierucci and G. Buzzi Ferraris (Editors) 2010 Elsevier B.. All rights reserved. Towards energy-based dynamic optimization of monoclonal
HuCAL Custom Monoclonal Antibodies
 HuCAL Custom Monoclonal Antibodies Highly Specific Monoclonal Antibodies in just 8 Weeks PROVEN, HIGHLY SPECIFIC, HIGH AFFINITY ANTIBODIES IN 8 WEEKS WITHOUT HuCAL PLATINUM IMMUNIZATION (Human Combinatorial
HuCAL Custom Monoclonal Antibodies Highly Specific Monoclonal Antibodies in just 8 Weeks PROVEN, HIGHLY SPECIFIC, HIGH AFFINITY ANTIBODIES IN 8 WEEKS WITHOUT HuCAL PLATINUM IMMUNIZATION (Human Combinatorial
Chapter 3.2» Custom Monoclonal
 198 3 3.2 Custom Monoclonal 199 Mouse monoclonal antibody development Chapter 3.2» Custom Monoclonal 200 In vitro monoclonals expression service 201 Mouse monoclonal antibody additional services 202 Magnetic
198 3 3.2 Custom Monoclonal 199 Mouse monoclonal antibody development Chapter 3.2» Custom Monoclonal 200 In vitro monoclonals expression service 201 Mouse monoclonal antibody additional services 202 Magnetic
Integrated Protein Services
 Integrated Protein Services Custom protein expression & purification Version DC04-0012 Expression strategy The first step in the recombinant protein generation process is to design an appropriate expression
Integrated Protein Services Custom protein expression & purification Version DC04-0012 Expression strategy The first step in the recombinant protein generation process is to design an appropriate expression
Animal Cell Culture. Third Edition. A Practical Approach OXJORD VNIVVRSITY 1'RVSS
 Animal Cell Culture Third Edition A Practical Approach Edited by John R. W. Masters 3rd Floor Research Laboratories, University College London OXJORD VNIVVRSITY 1'RVSS Contents List of protocols page xiii
Animal Cell Culture Third Edition A Practical Approach Edited by John R. W. Masters 3rd Floor Research Laboratories, University College London OXJORD VNIVVRSITY 1'RVSS Contents List of protocols page xiii
Catalent Biologics & Clinical Supplies The SMART Solution
 Catalent Biologics & Clinical Supplies The SMART Solution Advanced Technology and Integrated Solutions From DNA to Clinical Supply & Cold Chain Distribution Dr. Florian Schwaak Account Manager Germany
Catalent Biologics & Clinical Supplies The SMART Solution Advanced Technology and Integrated Solutions From DNA to Clinical Supply & Cold Chain Distribution Dr. Florian Schwaak Account Manager Germany
3 Chapter Three: Material and methods (clone creation, upstream and downstream process)
 3 Chapter Three: Material and methods (clone creation, upstream and downstream process) 3.1 Model proteins and CHO cell cultures Two recombinant produced, CHO-cell-derived model-glycoproteins, a less glycosylated
3 Chapter Three: Material and methods (clone creation, upstream and downstream process) 3.1 Model proteins and CHO cell cultures Two recombinant produced, CHO-cell-derived model-glycoproteins, a less glycosylated
Dr.ssa Maria Luisa Nolli CEO
 IL RUOLO DELLE PICCOLE E MEDIE INDUSTRIE NELLA PROMOZIONE DELLA MEDICINA TRASLAZIONALE Quality and Innovation Dr.ssa Maria Luisa Nolli CEO INTRODUCTION ARETA INTERNATIONAL 12 years of experience in contract
IL RUOLO DELLE PICCOLE E MEDIE INDUSTRIE NELLA PROMOZIONE DELLA MEDICINA TRASLAZIONALE Quality and Innovation Dr.ssa Maria Luisa Nolli CEO INTRODUCTION ARETA INTERNATIONAL 12 years of experience in contract
Challenges in the cgmp Manufacturing of hescs: Lessons Learned from Monoclonal Antibodies
 Challenges in the cgmp Manufacturing of hescs: Lessons Learned from Monoclonal Antibodies ISCT 2011 Annual Meeting Rotterdam, The Netherlands May 18 21, 2011 BioProcess Technology Consultants www.bptc.com
Challenges in the cgmp Manufacturing of hescs: Lessons Learned from Monoclonal Antibodies ISCT 2011 Annual Meeting Rotterdam, The Netherlands May 18 21, 2011 BioProcess Technology Consultants www.bptc.com
Molecular Biology Techniques: A Classroom Laboratory Manual THIRD EDITION
 Molecular Biology Techniques: A Classroom Laboratory Manual THIRD EDITION Susan Carson Heather B. Miller D.Scott Witherow ELSEVIER AMSTERDAM BOSTON HEIDELBERG LONDON NEW YORK OXFORD PARIS SAN DIEGO SAN
Molecular Biology Techniques: A Classroom Laboratory Manual THIRD EDITION Susan Carson Heather B. Miller D.Scott Witherow ELSEVIER AMSTERDAM BOSTON HEIDELBERG LONDON NEW YORK OXFORD PARIS SAN DIEGO SAN
High yield antibody production in a disposable WAVE Bioreactor
 High yield antibody production in a Process intensification using perfusion culture Christian Kaisermayer 1, Jianjun Yang 2 1 GE Healthcare Europe GmbH, Vienna, Austria (Christian.Kaisermayer@ge.com),
High yield antibody production in a Process intensification using perfusion culture Christian Kaisermayer 1, Jianjun Yang 2 1 GE Healthcare Europe GmbH, Vienna, Austria (Christian.Kaisermayer@ge.com),
CD3/TCR stimulation and surface detection Determination of specificity of intracellular detection of IL-7Rα by flow cytometry
 CD3/TCR stimulation and surface detection Stimulation of HPB-ALL cells with the anti-cd3 monoclonal antibody OKT3 was performed as described 3. In brief, antibody-coated plates were prepared by incubating
CD3/TCR stimulation and surface detection Stimulation of HPB-ALL cells with the anti-cd3 monoclonal antibody OKT3 was performed as described 3. In brief, antibody-coated plates were prepared by incubating
On the origin of giant multinuclear Reed-Sternberg cells and the role of CD4 T cells in Hodgkin lymphoma
 On the origin of giant multinuclear Reed-Sternberg cells and the role of CD4 T cells in Hodgkin lymphoma Uber die Entstehung von multinuklearen Reed-Sternberg Riesenzellen und die Rolle von CD4 T-Zellen
On the origin of giant multinuclear Reed-Sternberg cells and the role of CD4 T cells in Hodgkin lymphoma Uber die Entstehung von multinuklearen Reed-Sternberg Riesenzellen und die Rolle von CD4 T-Zellen
Cell Culture Technology for Pharmaceutical and Cellular Therapies
 Cell Culture Technology for Pharmaceutical and Cellular Therapies Sadettin S. Ozturk, Ph.D. Centocor Inc. 200 Great Valley Parkway, Malvern, PA 19355 Outline 1. Introduction to Monoclonal Antibodies 2.
Cell Culture Technology for Pharmaceutical and Cellular Therapies Sadettin S. Ozturk, Ph.D. Centocor Inc. 200 Great Valley Parkway, Malvern, PA 19355 Outline 1. Introduction to Monoclonal Antibodies 2.
CONTENT. Chapter 1 Review of Literature. List of figures. List of tables
 Abstract Abbreviations List of figures CONTENT I-VI VII-VIII IX-XII List of tables XIII Chapter 1 Review of Literature 1. Vaccination against intracellular pathogens 1-34 1.1 Role of different immune responses
Abstract Abbreviations List of figures CONTENT I-VI VII-VIII IX-XII List of tables XIII Chapter 1 Review of Literature 1. Vaccination against intracellular pathogens 1-34 1.1 Role of different immune responses
MAB Solut. MABSolys Génopole Campus 1 5 rue Henri Desbruères 91030 Evry Cedex. www.mabsolut.com. is involved at each stage of your project
 Mabsolus-2015-UK:Mise en page 1 03/07/15 14:13 Page1 Services provider Department of MABSolys from conception to validation MAB Solut is involved at each stage of your project Creation of antibodies Production
Mabsolus-2015-UK:Mise en page 1 03/07/15 14:13 Page1 Services provider Department of MABSolys from conception to validation MAB Solut is involved at each stage of your project Creation of antibodies Production
Technologies for Monoclonal Antibody Production
 Technologies for Monoclonal Antibody Production First we help you bridge the distance between upstream and downstream. Then we help you shorten it. Monoclonal antibodies for therapeutic, diagnostic and
Technologies for Monoclonal Antibody Production First we help you bridge the distance between upstream and downstream. Then we help you shorten it. Monoclonal antibodies for therapeutic, diagnostic and
Important Topics in the Expression of Recombinant Antibodies from CHO Cells
 Cell Line Development and Engineering Workshop, Prague, March 2008 Important Topics in the Expression of Recombinant Antibodies from CHO Cells Robert Young/Lonza Biologics/Mar 2008 Disclaimer Certain matters
Cell Line Development and Engineering Workshop, Prague, March 2008 Important Topics in the Expression of Recombinant Antibodies from CHO Cells Robert Young/Lonza Biologics/Mar 2008 Disclaimer Certain matters
Integrated Protein Services
 Integrated Protein Services Custom protein expression & purification Last date of revision June 2015 Version DC04-0013 www.iba-lifesciences.com Expression strategy The first step in the recombinant protein
Integrated Protein Services Custom protein expression & purification Last date of revision June 2015 Version DC04-0013 www.iba-lifesciences.com Expression strategy The first step in the recombinant protein
The immune response Antibodies Antigens Epitopes (antigenic determinants) the part of a protein antigen recognized by an antibody Haptens small
 The immune response Antibodies Antigens Epitopes (antigenic determinants) the part of a protein antigen recognized by an antibody Haptens small molecules that can elicit an immune response when linked
The immune response Antibodies Antigens Epitopes (antigenic determinants) the part of a protein antigen recognized by an antibody Haptens small molecules that can elicit an immune response when linked
Guideline for Generation of Stable Cell Lines Technical Reference Guide
 BioResearch Guideline for Generation of Stable Cell Lines Technical Reference Guide 1. Background Stable, long-term expression of a gene of interest can be either achieved by eukaryotic vectors that harbor
BioResearch Guideline for Generation of Stable Cell Lines Technical Reference Guide 1. Background Stable, long-term expression of a gene of interest can be either achieved by eukaryotic vectors that harbor
Geniron. Custom Antibody Services. Serum services Antibody Monoclonal. Purification Antibody Mono Y Genetic Immunization Genbody Polyclonal Antibody
 Geniron Custom Antibody Services Serum services Antibody Monoclonal Purification Antibody Mono Y Genetic Immunization Genbody Polyclonal Antibody Geniron Poly Y WE PROVIDE OUR SERVICES TO With Expertise
Geniron Custom Antibody Services Serum services Antibody Monoclonal Purification Antibody Mono Y Genetic Immunization Genbody Polyclonal Antibody Geniron Poly Y WE PROVIDE OUR SERVICES TO With Expertise
Retrospective Analysis of a Host Cell Protein Perfect Storm: Identifying Immunogenic Proteins and Fixing the Problem
 Retrospective Analysis of a Host Cell Protein Perfect Storm: Identifying Immunogenic Proteins and Fixing the Problem Kevin Van Cott, Associate Professor Dept. of Chemical and Biomolecular Engineering Nebraska
Retrospective Analysis of a Host Cell Protein Perfect Storm: Identifying Immunogenic Proteins and Fixing the Problem Kevin Van Cott, Associate Professor Dept. of Chemical and Biomolecular Engineering Nebraska
Monash Antibody Technologies Facility
 Monash Antibody Technologies Facility The Monash Antibody Technologies Facility: A high throughput monoclonal antibody production platform for phospho-kinomics Tecan Symposium Zürich September 2007 What
Monash Antibody Technologies Facility The Monash Antibody Technologies Facility: A high throughput monoclonal antibody production platform for phospho-kinomics Tecan Symposium Zürich September 2007 What
LFB GROUP & SANOFI combine their bioproduction capabilities to provide integrated CMO services for biopharmaceuticals
 LFB GROUP & SANOFI combine their bioproduction capabilities to provide integrated CMO services for biopharmaceuticals MAbLaunch TM is a joint bioproduction platform combining LFB Biomanufacturing (LFB
LFB GROUP & SANOFI combine their bioproduction capabilities to provide integrated CMO services for biopharmaceuticals MAbLaunch TM is a joint bioproduction platform combining LFB Biomanufacturing (LFB
Quantitative proteomics background
 Proteomics data analysis seminar Quantitative proteomics and transcriptomics of anaerobic and aerobic yeast cultures reveals post transcriptional regulation of key cellular processes de Groot, M., Daran
Proteomics data analysis seminar Quantitative proteomics and transcriptomics of anaerobic and aerobic yeast cultures reveals post transcriptional regulation of key cellular processes de Groot, M., Daran
2.1.2 Characterization of antiviral effect of cytokine expression on HBV replication in transduced mouse hepatocytes line
 i 1 INTRODUCTION 1.1 Human Hepatitis B virus (HBV) 1 1.1.1 Pathogenesis of Hepatitis B 1 1.1.2 Genome organization of HBV 3 1.1.3 Structure of HBV virion 5 1.1.4 HBV life cycle 5 1.1.5 Experimental models
i 1 INTRODUCTION 1.1 Human Hepatitis B virus (HBV) 1 1.1.1 Pathogenesis of Hepatitis B 1 1.1.2 Genome organization of HBV 3 1.1.3 Structure of HBV virion 5 1.1.4 HBV life cycle 5 1.1.5 Experimental models
Understanding the immune response to bacterial infections
 Understanding the immune response to bacterial infections A Ph.D. (SCIENCE) DISSERTATION SUBMITTED TO JADAVPUR UNIVERSITY SUSHIL KUMAR PATHAK DEPARTMENT OF CHEMISTRY BOSE INSTITUTE 2008 CONTENTS Page SUMMARY
Understanding the immune response to bacterial infections A Ph.D. (SCIENCE) DISSERTATION SUBMITTED TO JADAVPUR UNIVERSITY SUSHIL KUMAR PATHAK DEPARTMENT OF CHEMISTRY BOSE INSTITUTE 2008 CONTENTS Page SUMMARY
Chapter 8. Summary and Perspectives
 Chapter 8 Summary and Perspectives 131 Chapter 8 Summary Overexpression of the multidrug resistance protein MRP1 confer multidrug resistance (MDR) to cancer cells. The contents of this thesis describe
Chapter 8 Summary and Perspectives 131 Chapter 8 Summary Overexpression of the multidrug resistance protein MRP1 confer multidrug resistance (MDR) to cancer cells. The contents of this thesis describe
Custom Antibody Services
 Custom Antibody Services Custom service offerings DNA sequence Plasmid Peptide Structure Protein Peptide Small molecule Cells Spleen Lymphocytes Antigen Preparation Immunization Fusion & Subcloning Expansion
Custom Antibody Services Custom service offerings DNA sequence Plasmid Peptide Structure Protein Peptide Small molecule Cells Spleen Lymphocytes Antigen Preparation Immunization Fusion & Subcloning Expansion
CFSE Cell Division Assay Kit
 CFSE Cell Division Assay Kit Item No. 10009853 Customer Service 800.364.9897 * Technical Support 888.526.5351 www.caymanchem.com TABLE OF CONTENTS GENERAL INFORMATION 3 Materials Supplied 4 Precautions
CFSE Cell Division Assay Kit Item No. 10009853 Customer Service 800.364.9897 * Technical Support 888.526.5351 www.caymanchem.com TABLE OF CONTENTS GENERAL INFORMATION 3 Materials Supplied 4 Precautions
Title: Mapping T cell epitopes in PCV2 capsid protein - NPB #08-159. Date Submitted: 12-11-09
 Title: Mapping T cell epitopes in PCV2 capsid protein - NPB #08-159 Investigator: Institution: Carol Wyatt Kansas State University Date Submitted: 12-11-09 Industry summary: Effective circovirus vaccines
Title: Mapping T cell epitopes in PCV2 capsid protein - NPB #08-159 Investigator: Institution: Carol Wyatt Kansas State University Date Submitted: 12-11-09 Industry summary: Effective circovirus vaccines
LFB GROUP & SANOFI combine their bioproduction capabilities to provide integrated CMO services for biopharmaceuticals
 LFB GROUP & SANOFI combine their bioproduction capabilities to provide integrated CMO services for biopharmaceuticals MAbLaunch TM is a joint bioproduction platform combining LFB Biomanufacturing (LFB
LFB GROUP & SANOFI combine their bioproduction capabilities to provide integrated CMO services for biopharmaceuticals MAbLaunch TM is a joint bioproduction platform combining LFB Biomanufacturing (LFB
Practical Cell Analysis
 Practical Cell Analysis Dimitri Pappas Dept of Chemistry & Biochemistry, Texas Tech University, USA WILEY A John Wiley and Sons, Ltd, Publication Contents Preface Acknowledgments xiii xix 1 Getting Started
Practical Cell Analysis Dimitri Pappas Dept of Chemistry & Biochemistry, Texas Tech University, USA WILEY A John Wiley and Sons, Ltd, Publication Contents Preface Acknowledgments xiii xix 1 Getting Started
Overview of Phase 1 Oncology Trials of Biologic Therapeutics
 Overview of Phase 1 Oncology Trials of Biologic Therapeutics Susan Jerian, MD ONCORD, Inc. February 28, 2008 February 28, 2008 Phase 1 1 Assumptions and Ground Rules The goal is regulatory approval of
Overview of Phase 1 Oncology Trials of Biologic Therapeutics Susan Jerian, MD ONCORD, Inc. February 28, 2008 February 28, 2008 Phase 1 1 Assumptions and Ground Rules The goal is regulatory approval of
Chapter 18: Applications of Immunology
 Chapter 18: Applications of Immunology 1. Vaccinations 2. Monoclonal vs Polyclonal Ab 3. Diagnostic Immunology 1. Vaccinations What is Vaccination? A method of inducing artificial immunity by exposing
Chapter 18: Applications of Immunology 1. Vaccinations 2. Monoclonal vs Polyclonal Ab 3. Diagnostic Immunology 1. Vaccinations What is Vaccination? A method of inducing artificial immunity by exposing
In Vitro And In Vivo Production Of Antibodies
 In Vitro And In Vivo Production Of Antibodies OCTOBER 27, 2015 BY ADMIN Overview An antibody is a protein normally produced by the B cells of the immune system. Their original purpose is to identify and
In Vitro And In Vivo Production Of Antibodies OCTOBER 27, 2015 BY ADMIN Overview An antibody is a protein normally produced by the B cells of the immune system. Their original purpose is to identify and
BBSRC TECHNOLOGY STRATEGY: TECHNOLOGIES NEEDED BY RESEARCH KNOWLEDGE PROVIDERS
 BBSRC TECHNOLOGY STRATEGY: TECHNOLOGIES NEEDED BY RESEARCH KNOWLEDGE PROVIDERS 1. The Technology Strategy sets out six areas where technological developments are required to push the frontiers of knowledge
BBSRC TECHNOLOGY STRATEGY: TECHNOLOGIES NEEDED BY RESEARCH KNOWLEDGE PROVIDERS 1. The Technology Strategy sets out six areas where technological developments are required to push the frontiers of knowledge
Your partner in immunology
 Your partner in immunology Expertise Expertise Reactivity Reactivity Quality Quality Advice Advice Who are we? Specialist of antibody engineering Covalab is a French biotechnology company, specialised
Your partner in immunology Expertise Expertise Reactivity Reactivity Quality Quality Advice Advice Who are we? Specialist of antibody engineering Covalab is a French biotechnology company, specialised
3 months 1.5 months 1.5 months. 1 month
 Rabbit monoclonal antibody (Mab) is secreted by the plasma B-cell of the rabbit. Traditional generation of rabbit Mab relies on a rabbit myeloma for B- cell fusion (
Rabbit monoclonal antibody (Mab) is secreted by the plasma B-cell of the rabbit. Traditional generation of rabbit Mab relies on a rabbit myeloma for B- cell fusion (
Introduction to Bioprocessing
 Introduction to Bioprocessing Cambridge Healthtech Institute Peptalk Palm Springs, CA Presented by Susan Dana Jones and Sheila Magil BioProcess Technology Consultants www.bptc.com BioProcess Technology
Introduction to Bioprocessing Cambridge Healthtech Institute Peptalk Palm Springs, CA Presented by Susan Dana Jones and Sheila Magil BioProcess Technology Consultants www.bptc.com BioProcess Technology
High Throughput Robotic Production of Monoclonal Antibodies at MATF. Ed Nice, Daniel Layton, Caroline Laverty, Natasha Dodge,
 High Throughput Robotic Production of Monoclonal Antibodies at MATF Ed Nice, Daniel Layton, Caroline Laverty, Natasha Dodge, Bronwyn Briers and Jean Tang Monash Antibody Technology Facility Clayton Victoria
High Throughput Robotic Production of Monoclonal Antibodies at MATF Ed Nice, Daniel Layton, Caroline Laverty, Natasha Dodge, Bronwyn Briers and Jean Tang Monash Antibody Technology Facility Clayton Victoria
Triskel: a strategic consulting firm for biopharmaceutical companies
 BioStart : Manufacturing and operations in Biotech Strategic stakes and technology evolution in recombinant protein production APFL May 14 th, 2009 François LAWNY Triskel Integrated Services Geneva, Switzerland
BioStart : Manufacturing and operations in Biotech Strategic stakes and technology evolution in recombinant protein production APFL May 14 th, 2009 François LAWNY Triskel Integrated Services Geneva, Switzerland
Protein-responsive ribozyme switches in eukaryotic cells
 Protein-responsive ribozyme switches in eukaryotic cells Andrew B. Kennedy, James V. Vowles, Leo d Espaux, and Christina D. Smolke Presented by Marianne Linz and Jennifer Thornton March 11, 2015 Synthetic
Protein-responsive ribozyme switches in eukaryotic cells Andrew B. Kennedy, James V. Vowles, Leo d Espaux, and Christina D. Smolke Presented by Marianne Linz and Jennifer Thornton March 11, 2015 Synthetic
Notch 1 -dependent regulation of cell fate in colorectal cancer
 Notch 1 -dependent regulation of cell fate in colorectal cancer Referees: PD Dr. Tobias Dick Prof. Dr. Wilfried Roth http://d-nb.info/1057851272 CONTENTS Summary 1 Zusammenfassung 2 1 INTRODUCTION 3 1.1
Notch 1 -dependent regulation of cell fate in colorectal cancer Referees: PD Dr. Tobias Dick Prof. Dr. Wilfried Roth http://d-nb.info/1057851272 CONTENTS Summary 1 Zusammenfassung 2 1 INTRODUCTION 3 1.1
Protein Expression. A Practical Approach J. HIGGIN S
 Protein Expression A Practical Approach S. J. HIGGIN S B. D. HAMES List of contributors Abbreviations xv Xvi i 1. Protein expression in mammalian cell s Marlies Otter-Nilsson and Tommy Nilsso n 1. Introduction
Protein Expression A Practical Approach S. J. HIGGIN S B. D. HAMES List of contributors Abbreviations xv Xvi i 1. Protein expression in mammalian cell s Marlies Otter-Nilsson and Tommy Nilsso n 1. Introduction
Validated Cell-Based Assays for Rapid Screening and Functional Characterization of Therapeutic Monoclonal Antibodies
 Validated Cell-Based Assays for Rapid Screening and Functional Characterization of Therapeutic Monoclonal Antibodies Abhishek Saharia, Ph. D. Senior Product Manager Validation Assays Drug Discovery Evolution
Validated Cell-Based Assays for Rapid Screening and Functional Characterization of Therapeutic Monoclonal Antibodies Abhishek Saharia, Ph. D. Senior Product Manager Validation Assays Drug Discovery Evolution
INTERNATIONAL CONFERENCE ON HARMONISATION OF TECHNICAL REQUIREMENTS FOR REGISTRATION OF PHARMACEUTICALS FOR HUMAN USE Q5B
 INTERNATIONAL CONFERENCE ON HARMONISATION OF TECHNICAL REQUIREMENTS FOR REGISTRATION OF PHARMACEUTICALS FOR HUMAN USE ICH HARMONISED TRIPARTITE GUIDELINE QUALITY OF BIOTECHNOLOGICAL PRODUCTS: ANALYSIS
INTERNATIONAL CONFERENCE ON HARMONISATION OF TECHNICAL REQUIREMENTS FOR REGISTRATION OF PHARMACEUTICALS FOR HUMAN USE ICH HARMONISED TRIPARTITE GUIDELINE QUALITY OF BIOTECHNOLOGICAL PRODUCTS: ANALYSIS
Use of the ambr 250 in combination with high-throughput design and analysis tools for rapid, scalable USP development
 Use of the ambr 250 in combination with high-throughput design and analysis tools for rapid, scalable USP development Authors: Dr Fern Slingsby and Dr Simon Dewar Upstream Process Development FUJIFILM
Use of the ambr 250 in combination with high-throughput design and analysis tools for rapid, scalable USP development Authors: Dr Fern Slingsby and Dr Simon Dewar Upstream Process Development FUJIFILM
THE His Tag Antibody, mab, Mouse
 THE His Tag Antibody, mab, Mouse Cat. No. A00186 Technical Manual No. TM0243 Update date 01052011 I Description.... 1 II Key Features. 2 III Storage 2 IV Applications.... 2 V Examples - ELISA..... 2 VI
THE His Tag Antibody, mab, Mouse Cat. No. A00186 Technical Manual No. TM0243 Update date 01052011 I Description.... 1 II Key Features. 2 III Storage 2 IV Applications.... 2 V Examples - ELISA..... 2 VI
1.Gene Synthesis. 2.Peptide & Phospho-P. Assembly PCR. Design & Synthesis. Advantages. Specifications. Advantages
 1.Gene Synthesis Assembly PCR Looking for a cdna for your research but could not fish out the gene through traditional cloning methods or a supplier? Abnova provides a gene synthesis service via assembly
1.Gene Synthesis Assembly PCR Looking for a cdna for your research but could not fish out the gene through traditional cloning methods or a supplier? Abnova provides a gene synthesis service via assembly
TransIT -2020 Transfection Reagent
 Quick Reference Protocol, MSDS and Certificate of Analysis available at mirusbio.com/5400 INTRODUCTION TransIT -2020 Transfection Reagent is a high-performance, animal-origin free, broad spectrum reagent
Quick Reference Protocol, MSDS and Certificate of Analysis available at mirusbio.com/5400 INTRODUCTION TransIT -2020 Transfection Reagent is a high-performance, animal-origin free, broad spectrum reagent
PX Therapeutics : the partner for early stage biotherapeutics development Biotuesday, May 5 2009
 PX Therapeutics : the partner for early stage biotherapeutics development Biotuesday, May 5 2009 Christelle Dagoneau, PhD Business Development Director Company Profile Protein expert incorporated in 2000
PX Therapeutics : the partner for early stage biotherapeutics development Biotuesday, May 5 2009 Christelle Dagoneau, PhD Business Development Director Company Profile Protein expert incorporated in 2000
博 士 論 文 ( 要 約 ) A study on enzymatic synthesis of. stable cyclized peptides which. inhibit protein-protein interactions
 博 士 論 文 ( 要 約 ) 論 文 題 目 A study on enzymatic synthesis of stable cyclized peptides which inhibit protein-protein interactions ( 蛋 白 質 間 相 互 作 用 を 阻 害 する 安 定 な 環 状 化 ペプチドの 酵 素 合 成 に 関 する 研 究 ) 氏 名 張 静 1
博 士 論 文 ( 要 約 ) 論 文 題 目 A study on enzymatic synthesis of stable cyclized peptides which inhibit protein-protein interactions ( 蛋 白 質 間 相 互 作 用 を 阻 害 する 安 定 な 環 状 化 ペプチドの 酵 素 合 成 に 関 する 研 究 ) 氏 名 張 静 1
Manufacturing process of biologics
 Manufacturing process of biologics K. Ho Afssaps, France 2011 ICH International Conference on Harmonisation of Technical Requirements for Registration of Pharmaceuticals for Human Use 2011 ICH 1 Disclaimer:
Manufacturing process of biologics K. Ho Afssaps, France 2011 ICH International Conference on Harmonisation of Technical Requirements for Registration of Pharmaceuticals for Human Use 2011 ICH 1 Disclaimer:
FCAP Array v3.0 Software: A New Tool to Analyze BD Cytometric Bead Array (CBA) Data. Monisha Sundarrajan, PhD BD Biosciences
 FCAP Array v3.0 Software: A New Tool to Analyze BD Cytometric Bead Array (CBA) Data Monisha Sundarrajan, PhD BD Biosciences 23 13795 00 Agenda Introduction Overview of BD CBA bead based immunoassays FCAP
FCAP Array v3.0 Software: A New Tool to Analyze BD Cytometric Bead Array (CBA) Data Monisha Sundarrajan, PhD BD Biosciences 23 13795 00 Agenda Introduction Overview of BD CBA bead based immunoassays FCAP
Development and validation of neutralising anti-drug antibody (Nabs) assays
 Development and validation of neutralising anti-drug antibody (Nabs) assays Clare Kingsley Sector Manager, Bioanalytical Sciences, Quotient Bioresearch EBF 2012 Overview Anti-drug antibody and neutralising
Development and validation of neutralising anti-drug antibody (Nabs) assays Clare Kingsley Sector Manager, Bioanalytical Sciences, Quotient Bioresearch EBF 2012 Overview Anti-drug antibody and neutralising
Supplementary Materials for
 www.sciencesignaling.org/cgi/content/full/7/339/ra80/dc1 Supplementary Materials for Manipulation of receptor oligomerization as a strategy to inhibit signaling by TNF superfamily members Julia T. Warren,
www.sciencesignaling.org/cgi/content/full/7/339/ra80/dc1 Supplementary Materials for Manipulation of receptor oligomerization as a strategy to inhibit signaling by TNF superfamily members Julia T. Warren,
Analyzing antibody sequence for recombinant antibody expression. Hangxing Yu, Ph.D Senior Scientist, GenScript May 20, 2015
 Analyzing antibody sequence for recombinant antibody expression Hangxing Yu, Ph.D Senior Scientist, GenScript May 20, 2015 Presentation Outline 1 2 3 4 Antibody basics, structure and function Antibody
Analyzing antibody sequence for recombinant antibody expression Hangxing Yu, Ph.D Senior Scientist, GenScript May 20, 2015 Presentation Outline 1 2 3 4 Antibody basics, structure and function Antibody
Eden Biodesign ebook Monoclonal Antibody Production: Building the Platform
 Eden Biodesign ebook Monoclonal Antibody Production: Building the Platform CHAPTER 1: Overview CHAPTER 2: Challenges CHAPTER 3: Purification Methodology CHAPTER 4: Results CHAPTER 5: About Eden Biodesign
Eden Biodesign ebook Monoclonal Antibody Production: Building the Platform CHAPTER 1: Overview CHAPTER 2: Challenges CHAPTER 3: Purification Methodology CHAPTER 4: Results CHAPTER 5: About Eden Biodesign
Transfection-Transfer of non-viral genetic material into eukaryotic cells. Infection/ Transduction- Transfer of viral genetic material into cells.
 Transfection Key words: Transient transfection, Stable transfection, transfection methods, vector, plasmid, origin of replication, reporter gene/ protein, cloning site, promoter and enhancer, signal peptide,
Transfection Key words: Transient transfection, Stable transfection, transfection methods, vector, plasmid, origin of replication, reporter gene/ protein, cloning site, promoter and enhancer, signal peptide,
specific B cells Humoral immunity lymphocytes antibodies B cells bone marrow Cell-mediated immunity: T cells antibodies proteins
 Adaptive Immunity Chapter 17: Adaptive (specific) Immunity Bio 139 Dr. Amy Rogers Host defenses that are specific to a particular infectious agent Can be innate or genetic for humans as a group: most microbes
Adaptive Immunity Chapter 17: Adaptive (specific) Immunity Bio 139 Dr. Amy Rogers Host defenses that are specific to a particular infectious agent Can be innate or genetic for humans as a group: most microbes
Advanced BioDesign Outlines Solutions. Antibody Overview. by Advanced BioDesign. Project Start. Immunogenicity. Selecting Your Antigen
 Advanced BioDesign Outlines Solutions by Advanced BioDesign Antibody Overview Launching an immunisation programme is an important experimental step that needs care. With Advanced BioDesign, you may develop
Advanced BioDesign Outlines Solutions by Advanced BioDesign Antibody Overview Launching an immunisation programme is an important experimental step that needs care. With Advanced BioDesign, you may develop
Facility construction and start up for commercial scale manufacturing of monoclonal antibodies - A case study
 24 th Interphex, Japan, Technical Conference June 29 th, 2011 Michael Brown Facility construction and start up for commercial scale manufacturing of monoclonal antibodies - A case study Lonza AG Financially
24 th Interphex, Japan, Technical Conference June 29 th, 2011 Michael Brown Facility construction and start up for commercial scale manufacturing of monoclonal antibodies - A case study Lonza AG Financially
Advances inthe Development. of Therapeutic
 Advances inthe Development of Therapeutic Monoclonal Antibodies Susan Dana Jones, Francisco J. Castillo, Howard L. Levine ABSTRACT Monoclonal antibodies (MAbs) and related products are a dominant component
Advances inthe Development of Therapeutic Monoclonal Antibodies Susan Dana Jones, Francisco J. Castillo, Howard L. Levine ABSTRACT Monoclonal antibodies (MAbs) and related products are a dominant component
BaculoDirect Baculovirus Expression System Free your hands with the BaculoDirect Baculovirus Expression System
 BaculoDirect Baculovirus Expression System Free your hands with the BaculoDirect Baculovirus Expression System The BaculoDirect Baculovirus Expression System gives you: Unique speed and simplicity High-throughput
BaculoDirect Baculovirus Expression System Free your hands with the BaculoDirect Baculovirus Expression System The BaculoDirect Baculovirus Expression System gives you: Unique speed and simplicity High-throughput
"Small and large molecules bioproduction by mammalian and microbial fermentation"
 "Small and large molecules bioproduction by mammalian and microbial fermentation" H.-P. Meyer Bio/PharMOS 2004-23-25 March 2004 Symposium B Business Diversification in Life Science "Small and large molecules
"Small and large molecules bioproduction by mammalian and microbial fermentation" H.-P. Meyer Bio/PharMOS 2004-23-25 March 2004 Symposium B Business Diversification in Life Science "Small and large molecules
Call 2014: High throughput screening of therapeutic molecules and rare diseases
 Call 2014: High throughput screening of therapeutic molecules and rare diseases The second call High throughput screening of therapeutic molecules and rare diseases launched by the French Foundation for
Call 2014: High throughput screening of therapeutic molecules and rare diseases The second call High throughput screening of therapeutic molecules and rare diseases launched by the French Foundation for
HUMORAL IMMUNE RE- SPONSES: ACTIVATION OF B CELLS AND ANTIBODIES JASON CYSTER SECTION 13
 SECTION 13 HUMORAL IMMUNE RE- SPONSES: ACTIVATION OF B CELLS AND ANTIBODIES CONTACT INFORMATION Jason Cyster, PhD (Email) READING Basic Immunology: Functions and Disorders of the Immune System. Abbas,
SECTION 13 HUMORAL IMMUNE RE- SPONSES: ACTIVATION OF B CELLS AND ANTIBODIES CONTACT INFORMATION Jason Cyster, PhD (Email) READING Basic Immunology: Functions and Disorders of the Immune System. Abbas,
Application Note No. 2 / July 2012. Quantitative Assessment of Cell Quality, Viability and Proliferation. System
 Application Note No. 2 / July 2012 Quantitative Assessment of Cell Quality, Viability and Proliferation System Quantitative Assessment of Cell Quality, Viability and Proliferation Introduction In vitro
Application Note No. 2 / July 2012 Quantitative Assessment of Cell Quality, Viability and Proliferation System Quantitative Assessment of Cell Quality, Viability and Proliferation Introduction In vitro
Cellular Implants as mini-factories for the production of therapeutic antibodies against Altzheimer s disease. rapport research week Jonas Schiffmann
 Cellular Implants as mini-factories for the production of therapeutic antibodies against Altzheimer s disease rapport research week Jonas Schiffmann Supported by Aurélien Lathuilière, Bernard Schneider
Cellular Implants as mini-factories for the production of therapeutic antibodies against Altzheimer s disease rapport research week Jonas Schiffmann Supported by Aurélien Lathuilière, Bernard Schneider
Amaxa Mouse T Cell Nucleofector Kit
 Amaxa Mouse T Cell Nucleofector Kit For T cells isolated from C57BL/6 & BALB/c mice Evaluated for murine T cells isolated from C57BL/6 & BALB/c mice This protocol is designed for murine lymphocytes or
Amaxa Mouse T Cell Nucleofector Kit For T cells isolated from C57BL/6 & BALB/c mice Evaluated for murine T cells isolated from C57BL/6 & BALB/c mice This protocol is designed for murine lymphocytes or
Cytell Cell Imaging System
 Cytell Cell Imaging System Simplify your cell analysis and start changing tomorrow today www.gelifesciences.com/cytell The Cytell Cell Imaging System is an intuitive cell analysis instrument that streamlines
Cytell Cell Imaging System Simplify your cell analysis and start changing tomorrow today www.gelifesciences.com/cytell The Cytell Cell Imaging System is an intuitive cell analysis instrument that streamlines
Cloning GFP into Mammalian cells
 Protocol for Cloning GFP into Mammalian cells Studiepraktik 2013 Molecular Biology and Molecular Medicine Aarhus University Produced by the instructors: Tobias Holm Bønnelykke, Rikke Mouridsen, Steffan
Protocol for Cloning GFP into Mammalian cells Studiepraktik 2013 Molecular Biology and Molecular Medicine Aarhus University Produced by the instructors: Tobias Holm Bønnelykke, Rikke Mouridsen, Steffan
How To Get A Grant From Kinesis
 - The collaboration initiative to move drug candidates forward Introduction What are we offering? How? Why apply? Terms Background For grant applications and to attract venture capital start-up companies
- The collaboration initiative to move drug candidates forward Introduction What are we offering? How? Why apply? Terms Background For grant applications and to attract venture capital start-up companies
Assay Qualification Template for Host Cell Protein ELISA
 Assay Qualification Template for Host Cell Protein ELISA Introduction: With ever increasing importance being placed on host cell protein (HCP) removal during the purification process, it is critical to
Assay Qualification Template for Host Cell Protein ELISA Introduction: With ever increasing importance being placed on host cell protein (HCP) removal during the purification process, it is critical to
Overview. Transcriptional cascades. Amazing aspects of lineage plasticity. Conventional (B2) B cell development
 Overview B cell development Transcriptional cascades Amazing aspects of lineage plasticity Conventional (B2) B cell development What happens to an autoreactive B cell? B1 vs B2 cells Key anatomical sites
Overview B cell development Transcriptional cascades Amazing aspects of lineage plasticity Conventional (B2) B cell development What happens to an autoreactive B cell? B1 vs B2 cells Key anatomical sites
Protein Analysis. -Detection and quantification. Toby M Holmes
 Protein Analysis -Detection and quantification Toby M Holmes Clinical Research Unit UCD school of Medicine and Medical Sciences Mater Misericordiae University Hospital Dublin Protein structure General
Protein Analysis -Detection and quantification Toby M Holmes Clinical Research Unit UCD school of Medicine and Medical Sciences Mater Misericordiae University Hospital Dublin Protein structure General
Efficient Multi-Well Protein Purification Strategies
 Application Note PN 33576 Efficient Multi-Well Protein Purification Strategies Introduction Many tools and techniques are available today for protein purification. Development of a purification process
Application Note PN 33576 Efficient Multi-Well Protein Purification Strategies Introduction Many tools and techniques are available today for protein purification. Development of a purification process
Supplementary Figure 1.
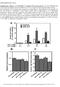 Supplementary Figure 1. (A) MicroRNA 212 enhances IS from pancreatic β-cells. INS-1 832/3 β-cells were transfected with precursors for mirnas 212, 375, or negative control oligonucleotides. 48 hrs after
Supplementary Figure 1. (A) MicroRNA 212 enhances IS from pancreatic β-cells. INS-1 832/3 β-cells were transfected with precursors for mirnas 212, 375, or negative control oligonucleotides. 48 hrs after
