Guidelines for the Diagnosis and Treatment of Malignant Pleural Mesothelioma
|
|
|
- Harold Walters
- 8 years ago
- Views:
Transcription
1 Guidelines for the Diagnosis and Treatment of Malignant Pleural Mesothelioma Supported by:
2 Asbestos Diseases Research Institute 2013 Authors: Organising Committee Publisher: Asbestos Diseases Research Institute Published: July 2013 ISBN print ISBN electronic This work is copyright. Apart from any use as permitted under the Copyright Act 1968, no part may be reproduced by any process without prior written permission from The Asbestos Diseases Research Institute. Requests and enquiries concerning reproduction and rights should be addressed to: Asbestos Diseases Research Institute PO Box 3628 Rhodes NSW 2138 Australia Disclaimer This document is a general guide to appropriate practice, to be followed subject to the clinician s judgment and the patient s preference in each individual case. The guidelines are designed to provide information to assist in decision-making. They are based on the best evidence available at time of compilation. The guidelines are not meant to be prescriptive. Conflict of interest The development of these clinical practice guidelines has been undertaken by a working party of experts convened by the Asbestos Diseases Research Institute. Of the 47 experts involved five declared a potential conflict of interest. Advisory boards, and participation in educational meetings organised by the Pharma industry were mentioned as a source of potential conflicts of interests. The Guidelines Steering Committee reviewing the conflict of interest declarations concluded that the methodological rigor used for the evidence review and the involvement of many experts has effectively managed any conflict of interest real or perceived. None of the experts involved in the development of the Guidelines for the Diagnosis and Treatment of Malignant Pleural Mesothelioma has received remuneration for their activities. Three working party members, who were involved in reviewing and grading the available evidence, have received a modest compensation for the hundreds of hours spent on quality control. Suggested citation Organising Committee. Guidelines for the Diagnosis and Treatment of Malignant Pleural Mesothelioma. Asbestos Diseases Research Institute; Sydney: Publication Approval These guidelines were approved by the Chief Executive Officer of the National Health and Medical Research Council (NHMRC) on 2 July 2013, under Section 14A of the National Health and Medical Research Council Act In approving these guidelines the NHMRC considers that they meet the NHMRC standard for clinical practice guidelines. This approval is valid for a period of 5 years. NHMRC is satisfied that they are based on the systematic identification and synthesis of the best available scientific evidence and make clear recommendations for health professionals practising in an Australian health care setting. The NHMRC expects that all guidelines will be reviewed no less than once every five years. This publication reflects the views of the authors and not necessarily the views of the Australian Government.
3 Table of contents Foreword 2 Executive summary 4 Summary of recommendations 5 Consensus based recommendations 9 Clinical practice points Introduction Diagnosis Assessment Active anti-cancer treatment Palliative and supportive care Models of care 66 Appendix A: Future research areas 72 Appendix B: Committee details 72 Appendix C: Overview of guideline development process 79 Appendix D: NHMRC Evidence Statement Form 79 Appendix E: Abbreviations 80 Appendix F: Glossary 81 Appendix G: Conflict of Interest 85 Appendix H: Acknowledgments 85 References 86 Sponsorship/support The development of these Guidelines was made possible by a generous donation from the Biaggio Signorelli Foundation; a Cancer Institute NSW grant and a contribution from Cancer Council NSW.
4 Foreword 2 Malignant Pleural Mesothelioma (MPM), the asbestos-induced neoplasm originating in the mesothelial lining of the lung cavities represents significant diagnostic and therapeutic challenges for clinicians in Australia. Very seldom diagnosed prior to the advent of widespread asbestos mining in the early to mid-twentieth century, it has sharply risen in incidence over the last five decades. According to the most recent Australian Institute of Health and Welfare data, there were 666 cases of malignant mesothelioma diagnosed in Australia in 2009 and around 90% of them originated in the pleura. Malignant pleural mesothelioma is almost always a fatal disease and the prognosis can only be modestly influenced by oncological treatments. The diagnostic process can be complex, with highly specialised advice frequently required to arrive at a definite diagnosis. Treatment varies from therapeutic nihilism to radical combined-modality treatment approaches. Although the disease and its management have a huge impact on the social, emotional, and material wellbeing of patients and families, supportive and palliative care pathways appear to be under-developed. The development of guidelines under the auspices of the Asbestos Diseases Research Institute has been undertaken in response to these circumstances. The guidelines organize the diagnostic and assessment process along the lines of the scientific evidence available, and provide for tailoring treatment on the basis of each patient s characteristics. Considerable emphasis has been placed on investigating and addressing supportive and palliative care needs in MPM, however the volume and quality of evidence specific to MPM available in these domains was disappointingly small. MPM is almost exclusively a man-made disease and Australia has one of the highest burdens of MPM on a population basis in the world. For the experts involved in collating and assessing the literature on the management of MPM for these guidelines, the level of active Australian research in areas such as diagnostic techniques, prognostic assessment, advanced radiotherapy techniques, and surgical outcomes has been a source of gratification. Many of these developments remain in the research and development phase. These Guidelines for the Diagnosis and Treatment of Malignant Pleural Mesothelioma systematise the approach to the management of MPM based on the best available evidence in accordance with standards to the assessment of evidence developed by The National Health and Medical Research Council in 2011(1). The Asbestos Diseases Research Institute, and the national team of experts involved in the preparation of the Guidelines, intends that they be a source of reference for health practitioners and consumers, because optimal management, by adherence to best practice guidelines, will improve the quality of life for each patient with malignant pleural mesothelioma and their confidence in the treatment approach.
5 Guidelines for the Diagnosis and Treatment of Malignant Pleural Mesothelioma The development of Guidelines for the Diagnosis and Treatment of Malignant Pleural Mesothelioma has drawn on contributions from a large number of people. Particular thanks are due to the Steering Committee members who took responsibility for drafting each section of the Guidelines, to librarians Suzanne Bakker, Jeremy Cullis and Yaping Liu for the retrieval of relevant literature, to Christopher Clarke, Henry Marshall and Steven Leong for their detailed assessment and grading of evidence, and to Ms Victoria Keena of the Asbestos Diseases Research Institute whose energy and commitment over an extended period has been a source of strength to all. Many of these contributions were voluntary. All were beyond the strict call of duty. The reward for this effort will be in seeing these guidelines used widely leading to better outcomes for patients with MPM. 3 Andrew Penman Chair Guidelines, Steering Committee Nico van Zandwijk Director Asbestos Diseases Research Institute
6 Executive summary 4 Malignant mesothelioma is an aggressive tumour originating in the serosal membranes that line the thoracic and abdominal cavities. More than 90% of reported mesothelioma cases occur in the pleura. The occurrence of malignant mesothelioma is typically related to exposure to mineral fibres such as asbestos and erionite. The World Health Organization (WHO) has recognised asbestos as one of the most important occupational carcinogens and in 2010 upgraded its global estimate of asbestos-related diseases to 107,000 annual deaths. Australia, as one of the largest consumers of asbestos worldwide in the post-world War II period, has one of the highest incidences of malignant mesothelioma. The current epidemic of malignant mesothelioma is closely associated with past occupational exposure. Asbestos, however, persists in our natural and built environments, and it is important that we continue to minimise exposure to it by all reasonable means. There are indications that in Australia the diagnostic and treatment practices for malignant pleural mesothelioma are not equally distributed, with considerable expertise concentrated in some hospitals and lacking in others. Moreover, there are no guidelines that specifically consider diagnosis and treatment of this almost invariably fatal disease in the Australian context. These evidence-based guidelines have been developed by a multidisciplinary team of experts (volunteers) that is encouraging improved management of malignant pleural mesothelioma through evidencebased decision making. Guidelines are guides and not rules. A good approach is to be fully aware of appropriate guidelines before making management decisions.
7 Guidelines for the Diagnosis and Treatment of Malignant Pleural Mesothelioma Summary of recommendations Chapter 2 - Diagnosis Recommendations Grade* Page 1. CT-guided core biopsy or VAT-guided pleural biopsy is recommended A 27 depending on the clinical circumstances to obtain adequate tissue for histological analysis including immunohistochemistry, and has high sensitivity and specificity for the diagnosis of malignant pleural mesothelioma. 2. Cytological recognition of an atypical mesothelial proliferation in pleural C 27 effusion fluid from patients may be sufficient for diagnosis in some patients when correlated with the clinical background and imaging studies, and when biopsy is considered inadvisable or unnecessary. 3. It should be standard histopathological practice to subtype mesotheliomas B 28 into epithelial (epithelioid), sarcomatoid and biphasic types (and other rare variants) and the distinction between epithelial versus sarcomatoid mesothelioma carries prognostic significance. 4. A panel of immunohistochemical markers should be used for pathologic B 30 diagnosis of malignant pleural mesothelioma. 5. The immunohistochemical panels should contain positive (mesothelial) B 30 and negative (carcinoma-related) markers for malignant mesotheliomas with an epithelioid component and include at least one cytokeratin marker, at least two mesothelial markers and at least two carcinomarelated markers. 6. For pleural mesothelioma-like tumours with an epithelial component, it B 30 is recommended that immunolabelling for both calretinin and TTF-1 is routinely carried out. 7. Additional markers should be added when tumours other than lung B 30 cancer enter into the differential diagnosis. 8. The immunoprofile of sarcomatoid mesotheliomas including desmoplastic B 30 mesothelioma is more restricted than that for mesotheliomas with an epithelial component, with variable expression of markers such as cytokeratin 5/6, calretinin, WT1 and podoplanin (D2-40). Labelling for cytokeratins is important and can facilitate assessment of invasion. However, cytokeratin-negative sarcomatoid mesotheliomas are recognised. 9. Tissue invasion should be demonstrated by histology or imaging studies to B 32 diagnose malignant mesothelioma definitively. 10. Measurement of the blood SMRP level is not recommended for routine clinical diagnosis. B 32 5 *Grade of recommendation A = Body of evidence can be trusted to guide practice B = Body of evidence can be trusted to guide practice in most situations C = Body of evidence provides some support for recommendation(s) but care should be taken in its application D = Body of evidence is weak and recommendation must be applied with caution.
8 Chapter 3 - Assessment 6 Recommendations Grade* Page 11. The TNM system should be used for disease staging in mesothelioma. B Patients with suspected or confirmed malignant pleural mesothelioma A 36 diagnosis should be assessed for therapeutic planning with CT of the thorax and abdomen with contrast enhancement. 13. CT or ultrasonography should be used to guide biopsy and drainage of B 36 pleural effusion. 14. FDG-PET is a more sensitive modality than CT to detect possible lymph A 37 node involvement and distant metastatic disease, and should be performed when the presence of disease in these sites will influence a management plan. 15. FDG-PET-CT should be used in preference to FDG-PET where available. A MRI should not be part of a routine assessment of patients with B 38 mesothelioma. 17. MRI with gadolinium enhancement can be useful in specialised situations C 38 where it is important to delineate tumour extension in the diaphragm, endothoracic fascia, chest wall or through iatrogenic tumour seeding. 18. Mediastinoscopy is recommended as an additional staging procedure for B 39 patients being considered for radical surgery in order to exclude N2 level nodal disease or to confirm pathological involvement where imaging is equivocal. 19. The addition of EUS-FNA and or EBUS is feasible in mesothelioma and may C 39 identify additional N2, T4, and M1 disease. 20. Bilateral thoracoscopy and laparoscopy with peritoneal lavage may B 39 identify additional M1 disease or sarcomatoid histology and taking the potential morbidity associated with radical surgery into account extended (surgical) staging should be considered for all patients with malignant pleural mesothelioma before resection. 21. Baseline prognostic assessment should include evaluation of important 41 patient, clinical, biological and imaging factors. a. Epithelioid histological type and performance status 1 are relatively A favourable prognostic factors. b. Male sex, weight loss and chest pain are unfavourable prognostic B factors. c. Elevated white cell count is an unfavourable prognostic factor. B d. Other markers of inflammation also confer an unfavourable prognosis. C e. Measurement of either SUVmax or TGV by FDG-PET provides prognostic information in patients with MPM. C
9 Guidelines for the Diagnosis and Treatment of Malignant Pleural Mesothelioma Recommendations Grade* Page 22. During treatment: 42 a. Assessment of treatment response using quantitative FDG-PET B parameters is predictive of survival outcome. b. Nodal stage 1, minimal residual disease and epithelioid histology are A favourable prognostic factors. c. Increasing serum SMRP levels during treatment are an unfavourable B prognostic marker. 23. Following suspected recurrence: 42 a. FDG-PET-CT should be performed when a diagnosis of recurrence B after previous radical surgical therapy is equivocal on other imaging modalities. b. Measurement of SUVmax on FDG-PET-CT following post-surgical C relapse is predictive of survival outcome. 24. Pleurodesis status should be known when interpreting results of CT or B 43 FDG PET imaging. 25. The extent of pre-treatment evaluation, including radiological evaluation C 43 and assessment of clinical and laboratory prognostic factors should be considered in the context of potential and appropriate management options. 26. In patients being considered for radical treatment, assessment should C 43 include pulmonary and cardiac function testing and evaluation of psychological status and co-morbidities. 27. Pre-treatment evaluation of patients considered for chemotherapy should include assessment of co-morbidities and general fitness. C 43 7
10 Chapter 4 Active anti-cancer treatment 8 Recommendations Grade* Page 28. Combination chemotherapy (pemetrexed and cisplatin or carboplatin) A 46 rather than single drug treatment should be used as first-line systemic treatment for malignant pleural mesothelioma. 29. Thoracoscopic pleurodesis is an effective treatment option to control B 48 recurrent malignant pleural effusions in mesothelioma. 30. If the thoracoscopic pleurodesis is not appropriate or fails, palliative C 48 pleurectomy/decortication should be considered for symptom control. 31. Only patients with favourable prognostic features, and favourable A 51 histology and staging, should be referred for consideration of radical treatment involving extensive cytoreductive surgery. 32. Radical surgical approaches should be restricted to institutions with B 51 significant surgical experience and high volume of cases. 33. Extensive cytoreductive surgery should only be used as part of B 51 multimodality treatment. 34. Mesothelioma is sensitive to moderately high radiation doses and C 52 radiotherapy is advocated for palliation of symptomatic tumour masses arising from the pleural cavity or metastases in other locations. 35. For doses greater than 50 Gy, advanced radiotherapy technologies with C 53 strict constraints for contralateral lung doses are recommended to avoid excessive toxicity. 36. The administration of prophylactic radiotherapy following pleural interventions in patients with mesothelioma has no significant effect on changing the disease course and is not recommended. C 54 Chapter 5 Palliative and supportive care Recommendations Grade* Page 37. Pleurodesis should be used to prevent recurrent pleural effusions. B Regular oral low dose, sustained release opioids should be given to reduce the intensity of breathlessness. B 58
11 Guidelines for the Diagnosis and Treatment of Malignant Pleural Mesothelioma Consensus based recommendations Chapter 3 - Assessment Consensus based recommendation i: Routine mediastinoscopy and other invasive procedures are not indicated in patients receiving supportive care or palliative management with chemotherapy. Page 39 Chapter 4 - Active Anti-Cancer Treatment Consensus based recommendation ii: Immunologically based and targeted therapies for patients with malignant mesothelioma should be restricted to clinical trials. Page 47 Clinical practice points 9 Chapter 2 - Diagnosis Clinical practice points a: VAT is not only the gold standard for securing biopsy tissue for the pathological diagnosis, but it also allows effective drainage of pleural effusion and talc pleurodesis. b: It is recommended that unless loculation of the fluid or other physical constraints prevent adequate sampling of the effusion fluid a minimum of 100 ml of effusion fluid and preferably the entire volume of fluid is submitted for cytology (after sampling of small volumes for biochemical and microbiological assessment). Such sampling is advocated to allow recovery of sufficient numbers of cells for cell block sections and immunohistochemical studies. Page c: The anatomical site and extent of lesions should be determined. 31 d: When tissue invasion cannot be identified, the lesion should be designated as an atypical mesothelial proliferation. 32 Chapter 3 - Assessment Clinical practice point e: New-generation spiral CT should be used in imaging malignant pleural mesothelioma. Page 37
12 Chapter 4 Active anti-cancer treatment Clinical practice points f: A multidisciplinary team with sufficient experience should provide advice on the suitability of patients for trimodality therapy and the ongoing treatment strategy adopted. g: Patients whose MPM progresses despite induction (neoadjuvant) chemotherapy should not be offered cytoreductive surgery followed by hemithoracic radiotherapy. Page Chapter 5 Palliative and supportive care 10 Clinical practice points Page h: Patients with malignant mesothelioma should be referred to a palliative care 57 specialist in a timely manner, and on the basis of their needs. i: The WHO principles of cancer pain management for patients with malignant 57 mesothelioma should be followed. j: A specialist palliative care physician should be involved early as part of the 57 multidisciplinary oncology team for patients with refractory or unresponsive pain. k: Palliative radiotherapy should be considered for patients with painful chest wall 57 infiltration or nodules. l: In order to tailor information to a person s individual needs at a particular point in 59 time, it is necessary to: give clear information specific to the individual repeat and summarise important information encourage questions actively check the person s understanding, and provide additional written/ audiovisual information. m: Patients should be screened for psychological distress and unmet needs. 61 n: Patients and carers should be referred to appropriate counseling services when required. 62 o: Information, guidance and emotional support should be provided for carers. 62 p: Consultations should be provided with specialist nurses trained in the care of patients with malignant pleural mesothelioma. q: Practitioners dealing with MPM patients should be aware that legal remedies are available and the patient should be advised of this upon diagnosis
13 Guidelines for the Diagnosis and Treatment of Malignant Pleural Mesothelioma Chapter 6 Models of care Clinical practice points r: A multidisciplinary team approach will ensure consistency in patient management through the development of a multidisciplinary care plan that will guide patient treatment throughout their illness and provide support for their carers. s: Treating specialists and/or the MDT should establish communication with the patient s GP as soon as possible after diagnosis, and keep them informed about their patient s changing needs and whom they should contact for expert advice. t: Nurse care coordinators are important members of the MDT. They provide support and information to patients with mesothelioma, ensure timely and appropriate referrals, help navigate the patient through their disease journey and coordinate their multidisciplinary care. u: Where mesothelioma-specific treatment options, including surgery, are not available in a given centre, medical teams should refer patients to centres offering expert mesothelioma care for discussion of all potential treatment options and care planning. v: The frequency and type of follow-up should be determined by individual patient symptoms, the stage of the disease and the treatment goals. CT scanning is the most useful investigation for evaluating disease progress. w: Allied health professionals are important members of the MDT and contribute to symptom management and improved quality of life in patients with malignant mesothelioma. Page
14 1.0 Introduction 1.1 Background Malignant mesothelioma is an aggressive tumour that originates in the serosal membranes that line the thoracic and abdominal cavities. This disease has become an important health issue over recent years, with Australia having one of the highest reported incidences (2-4). More than 90% of reported cases of mesothelioma occur in the pleura, compared with 4 7% affecting the peritoneum, and fewer than 1% jointly occurring in the pericardium and tunica vaginalis testis (2, 4, 5). Even rarer cases have been recorded as apparently primary ovarian mesotheliomas (6, 7). The occurrence of malignant mesothelioma is typically related to exposure to mineral fibres such as asbestos and erionite (8-10). Asbestos is a collection of naturally occurring crystalline hydrated silicates that are resistant to high temperatures and humidity. Asbestos fibres are biopersistent (retained in the human body) and can be detected as asbestos bodies in the lung many years after inhalation (11). 12 The World Health Organization (WHO) has recognised asbestos as one of the most important occupational carcinogens and in 2010 upgraded its global estimate of asbestosrelated diseases to 107,000 annual deaths (12). 1.2 History of mesothelioma The first studies on the association between asbestos and malignant mesothelioma appeared in the 1950s. Weiss case report of asbestosis and pleural malignancy and Van der Schoot s paper describing three insulation workers with malignant disease were the first of many to be published (13, 14). Wagner confirmed the association between asbestos and malignant mesothelioma through his work in the 1950s in South Africa, a country that mined all three commercial types of asbestos (15). Because most asbestos exposure occurred in the work environment, malignant mesothelioma has traditionally been considered an occupational disease. Paraoccupational malignant mesothelioma has been described in households of asbestos workers in which cohabitants had been exposed via contaminated clothes (16). The term environmental malignant mesothelioma has been used to describe disease identified in people living close to asbestos mines or factories or when people have been exposed to asbestos or asbestos-like material present in the soil (17, 18). Other factors have been recognised as potential causes of malignant mesothelioma. Radiotherapy to the chest has been reported but the number of patients with this association is limited (19). The role of SV40 (one of the simian viruses) viral infection as an important etiologic cofactor in malignant mesothelioma remains under discussion (20, 21). Exposure to asbestos is more common in occupations with a predominantly male workforce, which explains why the current incidence of malignant mesothelioma is higher among men than women. Most mesothelioma patients have been primary asbestos workers or people who handled raw asbestos in the mining, milling, transportation and manufacturing of the material. As this high-risk occupational exposure has been limited by the total ban on the use of asbestos products in Australia, the exposure-mix may change to include a greater proportion of people who have been exposed in non-occupational settings.
15 Guidelines for the Diagnosis and Treatment of Malignant Pleural Mesothelioma A dose-response relationship between cumulative asbestos exposure (increased levels or duration of exposure, or both) and malignant mesothelioma has been demonstrated (22). A safe threshold of cumulative exposure, below which there is no increased risk, has not been defined (23). The latency period, or the period between first exposure to asbestos and the diagnosis of mesothelioma, shows a wide range (20 60 years) and there are indications that the latency in Australia has increased in recent years (24). The median age at diagnosis of malignant mesothelioma in Australia is slightly above 70 years, with many patients presenting with co-morbidities (4). 1.3 Incidence of malignant mesothelioma Variation in the incidence of malignant mesothelioma is reported in different parts of the world. For example, seven people per million in Japan have been diagnosed with malignant mesothelioma compared with 40 people per million in Australia. These differences are largely attributable to the amount of asbestos consumed in a certain period (25). Australia, as one of the largest consumers of asbestos worldwide in the post-world War II period, has one of the highest incidences of malignant mesothelioma. Around 660 new cases of malignant mesothelioma were documented in 2007 and, in terms of mortality, this disease is approaching the numbers of deaths caused by multiple myeloma and ovarian cancer. 13 There is also regional variation in the incidence of malignant mesothelioma. For example, in Australia the highest reported incidence has been in men in Western Australia. This variation is largely attributable to occupational exposure associated with crocidolite mining in Wittenoom (3). Most deaths caused by malignant mesothelioma in Australia and other developed countries are due to occupational exposure to asbestos. The frequency of cases attributable to occupational exposure may have begun to decline owing to stringent control of asbestos use and handling. Asbestos, however, persists in our natural and built environments, and it is important that we continue to minimise exposure to it by all reasonable means. Among mesothelioma patients who do not have a history of occupational exposure, there is now a high proportion of people with a history of home renovation, in which exposure to asbestos might have occurred (26). Research is needed to determine if asbestos exposure explains this high proportion. It is important also that we remain alert to sources of possible exposure to asbestos in the community and control any such exposure as it is identified. Data on the incidence and mortality of malignant mesothelioma in Aboriginal and Torres Strait Islanders and culturally and linguistically diverse groups has not been reliably estimated due to the lack of recorded ethnicity. However, from July 2010, all new cases of malignant mesothelioma diagnosed in Australia are monitored by the Australian Mesothelioma Registry.
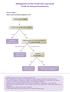
16 1.4 Clinical need for these Guidelines A recent study highlighted the lack of standardisation or adherence to guidelines during diagnosis, treatment, and surveillance of cancer patients as one of the major barriers to providing high quality cancer care (27). According to the US Institute of Medicine (28), high quality health care must be: based on the best evidence efficient safe from avoidable errors delivered in a timely manner patient-centred equitable. 14 There is scant data available on the current medical practices for patients with malignant pleural mesothelioma in Australia. A report on 295 patients diagnosed with malignant mesothelioma in the 1980s found considerable variation in practice (29). There are indications that diagnostic and treatment practices are not equally distributed, with considerable expertise concentrated in some hospitals and lacking in others. Several clinical guidelines for malignant pleural mesothelioma have been published recently (21, 30-35). All were collated by experts but none of them used a systematic analysis of the literature retrieved through general search terms and PICO (patient, intervention, comparison, outcome) questions as required by the Australian National Health and Medical Research Council (NHMRC)(1). Moreover, there are no guidelines that specifically consider diagnosis and treatment of malignant pleural mesothelioma in the Australian context. To address this gap, a team of experts decided to write guidelines based on a systematic review of the available literature. These guidelines are based on a systematic review of the literature executed according to the NHMRC guidelines development plan (36). Primum non nocere was regarded a primary issue when formulating the guideline recommendations. In addition we have drafted five scenarios that have assisted us in selecting the most important PICO questions. Scenario A (Figure 1) is based on the most common presentation of patients with malignant mesothelioma those presenting with a pleural effusion. Scenario B depicts another (less frequent) pathway, when a patient presents with a pleural mass (Figure 2). In scenario C the assessment journey of patients with a pathologically confirmed diagnosis is outlined (Figure 3) and scenario D deals with treatment choices for malignant mesothelioma patients after diagnosis and assessment (Figure 4). Scenario E (Figure 5) depicts the second-line treatment choices. PICO questions were formulated according to these scenarios and literature searches were based on these PICO questions (see Tables ). The evidence found in the literature searches was graded to produce evidence-based recommendations applicable to the Australian clinical context. Although the cutoff date of the literature review was 31st October 2011, a few exceptions (eight) were made to include prominent articles that were published after this date, adding important new information. These guidelines will provide a benchmark for the evaluation of current patterns of care for patients with malignant pleural mesothelioma.
17 Guidelines for the Diagnosis and Treatment of Malignant Pleural Mesothelioma Scenario A: Presentation and Diagnosis Scenario A: Presentation and Diagnosis Diagnostic pleural aspiration Alternate diagnosis (not MPM) Pleural effusion requiring evaluation Suspicious for MPM - not fit - declines Pathological confirmation required Procedures: -VATS -Open Biopsy -TTNA -Mediastinoscopy -E(B)US FNA - Other Pathologically confirmed MPM 15 Not requiring pathological confirmation Figure 1. Scenario A. The most common presentation of a patient with malignant mesothelioma MPM Malignant Pleural Mesothelioma VATS Video-Assisted Thoracoscopic Surgery TTNA Trans-Thoracic Needle Aspiration E(B)US FNA Endobronchial or Esophageal Endoscopic Ultrasound-guided Fine Needle Aspiration
18 Scenario B: Presentation and Diagnosis Scenario B: Presentation and Diagnosis TTNA - Fine needle -- core No MPM 16 Pleural thickening or pleural mass requiring evaluation Pathologically confirmed MPM Open biopsy other biopsy VATS No MPM Figure 2. Scenario B. Pathway of a patient presenting with pleural thickening or pleural mass. MPM Malignant Pleural Mesothelioma TTNA Trans-Thoracic Needle Aspiration
19 Guidelines for the Diagnosis and Treatment of Malignant Pleural Mesothelioma Scenario C: Assessment additional investigations Scenario C: Assessment additional investigations Imaging -CT -PET -Other Pathologically confirmed MPM Requires staging and/or preoperative evaluation Functional / Performance -ECOG -Organ / -Cardiopulmonary function -QoL TNM stage + fitness MDT 17 Does not require staging or further evaluation LABS Figure 3. Scenario C. The journey of the MPM patient with a pathologically confirmed diagnosis. MPM Malignant Pleural Mesothelioma CT Computer tomography PET Positron emission tomography ECOG Performance Status QoL Quality of Life TNM Tumour, Node, Metastasis MDT Multidisciplinary Team
20 Scenario D: Treatment choices! Scenario D: Treatment choices 18 Pathologically confirmed MPM MDT & patient preference Anti-cancer Rx indicated Anti-cancer Rx options: -Trimodality -Chemo -RT -Surgery -other Not fit / not suitable / declines anti-cancer Rx Symptom relief options: -Pain -Dyspnoea -Malaise -other Figure 4. Scenario D. Treatment choices for MPM patients after diagnosis and assessment. MPM Malignant Pleural Mesothelioma MDT - Multidisciplinary Team Rx Therapy RT - Radiotherapy
بسم هللا الرحمن الرحيم
 بسم هللا الرحمن الرحيم Updates in Mesothelioma By Samieh Amer, MD Professor of Cardiothoracic Surgery Faculty of Medicine, Cairo University History Wagner and his colleagues (1960) 33 cases of mesothelioma
بسم هللا الرحمن الرحيم Updates in Mesothelioma By Samieh Amer, MD Professor of Cardiothoracic Surgery Faculty of Medicine, Cairo University History Wagner and his colleagues (1960) 33 cases of mesothelioma
Malignant Mesothelioma
 Malignant Malignant mesothelioma is a tumour originating from mesothelial cells. 85 95% of mesotheliomas are caused by asbestos exposure. It occurs much more commonly in the chest (malignant pleural mesothelioma)
Malignant Malignant mesothelioma is a tumour originating from mesothelial cells. 85 95% of mesotheliomas are caused by asbestos exposure. It occurs much more commonly in the chest (malignant pleural mesothelioma)
Malignant Mesothelioma
 Malignant mesothelioma is a tumour originating from mesothelial cells. 85 95% of mesotheliomas are caused by asbestos exposure. It occurs much more commonly in the chest (malignant pleural mesothelioma)
Malignant mesothelioma is a tumour originating from mesothelial cells. 85 95% of mesotheliomas are caused by asbestos exposure. It occurs much more commonly in the chest (malignant pleural mesothelioma)
Objectives. Mylene T. Truong, MD. Malignant Pleural Mesothelioma Background
 Imaging of Pleural Tumors Mylene T. Truong, MD Imaging of Pleural Tumours Mylene T. Truong, M. D. University of Texas M.D. Anderson Cancer Center, Houston, TX Objectives To review tumors involving the
Imaging of Pleural Tumors Mylene T. Truong, MD Imaging of Pleural Tumours Mylene T. Truong, M. D. University of Texas M.D. Anderson Cancer Center, Houston, TX Objectives To review tumors involving the
Mesothelioma: Questions and Answers
 CANCER FACTS N a t i o n a l C a n c e r I n s t i t u t e N a t i o n a l I n s t i t u t e s o f H e a l t h D e p a r t m e n t o f H e a l t h a n d H u m a n S e r v i c e s Mesothelioma: Questions
CANCER FACTS N a t i o n a l C a n c e r I n s t i t u t e N a t i o n a l I n s t i t u t e s o f H e a l t h D e p a r t m e n t o f H e a l t h a n d H u m a n S e r v i c e s Mesothelioma: Questions
Malignant Mesothelioma: an Update
 Malignant Mesothelioma: an Update Nico van Zandwijk Asbestos Diseases Research Institute Bernie Banton Centre University of Sydney Australia Physicians Week RACP 19-5-2009 Health Risks of Asbestos Fibers
Malignant Mesothelioma: an Update Nico van Zandwijk Asbestos Diseases Research Institute Bernie Banton Centre University of Sydney Australia Physicians Week RACP 19-5-2009 Health Risks of Asbestos Fibers
L Lang-Lazdunski, A Bille, S Marshall, R Lal, D Landau, J Spicer
 Pleurectomy/decortication, hyperthermic pleural lavage with povidone-iodine and systemic chemotherapy in malignant pleural mesothelioma. A 10-year experience. L Lang-Lazdunski, A Bille, S Marshall, R Lal,
Pleurectomy/decortication, hyperthermic pleural lavage with povidone-iodine and systemic chemotherapy in malignant pleural mesothelioma. A 10-year experience. L Lang-Lazdunski, A Bille, S Marshall, R Lal,
Mesothelioma. 1. Introduction. 1.1 General Information and Aetiology
 Mesothelioma 1. Introduction 1.1 General Information and Aetiology Mesotheliomas are tumours that arise from the mesothelial cells of the pleura, peritoneum, pericardium or tunica vaginalis [1]. Most are
Mesothelioma 1. Introduction 1.1 General Information and Aetiology Mesotheliomas are tumours that arise from the mesothelial cells of the pleura, peritoneum, pericardium or tunica vaginalis [1]. Most are
INTERNATIONAL ASSOCIATION FOR THE STUDY OF LUNG CANCER Prospective Mesothelioma Staging Project
 INTERNATIONAL ASSOCIATION FOR THE STUDY OF LUNG CANCER Prospective Mesothelioma Staging Project Data Forms and Fields in CRAB Electronic Data Capture System - Reduced Set - Pivotal data elements for developing
INTERNATIONAL ASSOCIATION FOR THE STUDY OF LUNG CANCER Prospective Mesothelioma Staging Project Data Forms and Fields in CRAB Electronic Data Capture System - Reduced Set - Pivotal data elements for developing
Asbestos Related Diseases
 Asbestos Related Diseases Asbestosis Mesothelioma Lung Cancer Pleural Disease Asbestosis and Mesothelioma (LUNG CANCER) Support Group 1800 017 758 www.amsg.com.au ii Helping you and your family through
Asbestos Related Diseases Asbestosis Mesothelioma Lung Cancer Pleural Disease Asbestosis and Mesothelioma (LUNG CANCER) Support Group 1800 017 758 www.amsg.com.au ii Helping you and your family through
Asbestos Related Diseases. Asbestosis Mesothelioma Lung Cancer Pleural Disease. connecting raising awareness supporting advocating
 Asbestos Related Diseases Asbestosis Mesothelioma Lung Cancer Pleural Disease connecting raising awareness supporting advocating 1800 017 758 www.asbestosassociation.com.au Asbestos lagging was widely
Asbestos Related Diseases Asbestosis Mesothelioma Lung Cancer Pleural Disease connecting raising awareness supporting advocating 1800 017 758 www.asbestosassociation.com.au Asbestos lagging was widely
Clinical cases in Malignant Pleural Mesothelioma: Adherence to the ESMO Clinical Practice Guidelines
 Clinical cases in Malignant Pleural Mesothelioma: Adherence to the ESMO Clinical Practice Guidelines Wieneke Buikhuisen The Netherlands Cancer Institute Amsterdam The Netherlands Case (1) Male, 56 year
Clinical cases in Malignant Pleural Mesothelioma: Adherence to the ESMO Clinical Practice Guidelines Wieneke Buikhuisen The Netherlands Cancer Institute Amsterdam The Netherlands Case (1) Male, 56 year
Disease/Illness GUIDE TO ASBESTOS LUNG CANCER. What Is Asbestos Lung Cancer? www.simpsonmillar.co.uk Telephone 0844 858 3200
 GUIDE TO ASBESTOS LUNG CANCER What Is Asbestos Lung Cancer? Like tobacco smoking, exposure to asbestos can result in the development of lung cancer. Similarly, the risk of developing asbestos induced lung
GUIDE TO ASBESTOS LUNG CANCER What Is Asbestos Lung Cancer? Like tobacco smoking, exposure to asbestos can result in the development of lung cancer. Similarly, the risk of developing asbestos induced lung
Surgeons Role in Symptom Management. A/Prof Cliff K. C. Choong Consultant Thoracic Surgeon Latrobe Regional Hospital GIPPSLAND
 Surgeons Role in Symptom Management A/Prof Cliff K. C. Choong Consultant Thoracic Surgeon Latrobe Regional Hospital GIPPSLAND Conditions PLEURAL Pleural effusion Pneumothorax ENDOBRONCHIAL Haemoptysis
Surgeons Role in Symptom Management A/Prof Cliff K. C. Choong Consultant Thoracic Surgeon Latrobe Regional Hospital GIPPSLAND Conditions PLEURAL Pleural effusion Pneumothorax ENDOBRONCHIAL Haemoptysis
Treatment Algorithms for the Management of Lung Cancer in NSW Guide for Clinicians
 Treatment Algorithms for the Management of Lung Cancer in NSW Guide for Clinicians Background The Cancer Institute New South Wales Oncology Group Lung (NSWOG Lung) identified the need for the development
Treatment Algorithms for the Management of Lung Cancer in NSW Guide for Clinicians Background The Cancer Institute New South Wales Oncology Group Lung (NSWOG Lung) identified the need for the development
Cytology : first alert of mesothelioma? Professor B. Weynand, UCL Yvoir, Belgium
 Cytology : first alert of mesothelioma? Professor B. Weynand, UCL Yvoir, Belgium Introduction 3 cavities with the same embryologic origin the mesoderme Pleura Exudates Pleura Peritoneum Pericardium 22%
Cytology : first alert of mesothelioma? Professor B. Weynand, UCL Yvoir, Belgium Introduction 3 cavities with the same embryologic origin the mesoderme Pleura Exudates Pleura Peritoneum Pericardium 22%
SMALL CELL LUNG CANCER
 Protocol for Planning and Treatment The process to be followed in the management of: SMALL CELL LUNG CANCER Patient information given at each stage following agreed information pathway 1. DIAGNOSIS New
Protocol for Planning and Treatment The process to be followed in the management of: SMALL CELL LUNG CANCER Patient information given at each stage following agreed information pathway 1. DIAGNOSIS New
ASBESTOS EXPOSURE AND SARCOMATOID MALIGNANT PLEURAL MESOTHELIOMA Gorantla Sambasivarao 1, Namballa Usharani 2, Tupakula Suresh Babu 3
 ASBESTOS EXPOSURE AND SARCOMATOID MALIGNANT PLEURAL MESOTHELIOMA Gorantla Sambasivarao 1, Namballa Usharani 2, Tupakula Suresh Babu 3 HOW TO CITE THIS ARTICLE: Gorantla Sambasivarao, Namballa Usharani,
ASBESTOS EXPOSURE AND SARCOMATOID MALIGNANT PLEURAL MESOTHELIOMA Gorantla Sambasivarao 1, Namballa Usharani 2, Tupakula Suresh Babu 3 HOW TO CITE THIS ARTICLE: Gorantla Sambasivarao, Namballa Usharani,
International Journal of Case Reports in Medicine
 International Journal of Case Reports in Medicine Vol. 2013 (2013), Article ID 409830, 15 minipages. DOI:10.5171/2013.409830 www.ibimapublishing.com Copyright 2013 Andrew Thomas Low, Iain Smith and Simon
International Journal of Case Reports in Medicine Vol. 2013 (2013), Article ID 409830, 15 minipages. DOI:10.5171/2013.409830 www.ibimapublishing.com Copyright 2013 Andrew Thomas Low, Iain Smith and Simon
What is Mesothelioma?
 What is Mesothelioma? Mesothelioma is a rare type of cancer that develops in the mesothelial cells found in one s body. These cells form membranous linings that surround and protect the body s organs and
What is Mesothelioma? Mesothelioma is a rare type of cancer that develops in the mesothelial cells found in one s body. These cells form membranous linings that surround and protect the body s organs and
Lung Cancer & Mesothelioma 2011-2015
 Lung Cancer & Mesothelioma 2011-2015 Annex G Mesothelioma 1. The vision for mesothelioma services is set out in the Mesothelioma Framework issued by DH on 26 February 2007 (supported by the British Thoracic
Lung Cancer & Mesothelioma 2011-2015 Annex G Mesothelioma 1. The vision for mesothelioma services is set out in the Mesothelioma Framework issued by DH on 26 February 2007 (supported by the British Thoracic
Mesothelioma. Malignant Pleural Mesothelioma
 Mesothelioma William G. Richards, PhD Brigham and Women s Hospital Malignant Pleural Mesothelioma 2,000-3,000 cases per year (USA) Increasing incidence Asbestos (50-80%, decreasing) 30-40 year latency
Mesothelioma William G. Richards, PhD Brigham and Women s Hospital Malignant Pleural Mesothelioma 2,000-3,000 cases per year (USA) Increasing incidence Asbestos (50-80%, decreasing) 30-40 year latency
Extrapleural Pneumonectomy for Malignant Mesothelioma: Pro. Joon H. Lee 9/17/2012
 Extrapleural Pneumonectomy for Malignant Mesothelioma: Pro Joon H. Lee 9/17/2012 Malignant Pleural Mesothelioma (Epidemiology) Incidence: 7/mil (Japan) to 40/mil (Australia) Attributed secondary to asbestos
Extrapleural Pneumonectomy for Malignant Mesothelioma: Pro Joon H. Lee 9/17/2012 Malignant Pleural Mesothelioma (Epidemiology) Incidence: 7/mil (Japan) to 40/mil (Australia) Attributed secondary to asbestos
J of Evidence Based Med & Hlthcare, pissn- 2349-2562, eissn- 2349-2570/ Vol. 2/Issue 33/Aug. 17, 2015 Page 5063
 PERITONEAL MALIGNANT MESOTHELIOMA: A RARE S. R. Dhamotharan 1, S. Shanthi Nirmala 2, F. Celine Foustina Mary 3, M. Arul Raj Kumar 4, R. Vinothprabhu 5 HOW TO CITE THIS ARTICLE: S. R. Dhamotharan, S. Shanthi
PERITONEAL MALIGNANT MESOTHELIOMA: A RARE S. R. Dhamotharan 1, S. Shanthi Nirmala 2, F. Celine Foustina Mary 3, M. Arul Raj Kumar 4, R. Vinothprabhu 5 HOW TO CITE THIS ARTICLE: S. R. Dhamotharan, S. Shanthi
B10/S/a Cancer: Malignant Mesothelioma (Adult)
 B10/S/a 2013/14 NHS STANDARD CONTRACT FOR CANCER: MALIGNANT MESOTHELIOMA (ADULT) SECTION B PART 1 - SERVICE SPECIFICATIONS Service Specification No. Service Commissioner Lead Provider Lead Period Date
B10/S/a 2013/14 NHS STANDARD CONTRACT FOR CANCER: MALIGNANT MESOTHELIOMA (ADULT) SECTION B PART 1 - SERVICE SPECIFICATIONS Service Specification No. Service Commissioner Lead Provider Lead Period Date
Understanding Pleural Mesothelioma
 Understanding Pleural Mesothelioma UHN Information for patients and families Read this booklet to learn about: What is pleural mesothelioma? What causes it? What are the symptoms? What tests are done to
Understanding Pleural Mesothelioma UHN Information for patients and families Read this booklet to learn about: What is pleural mesothelioma? What causes it? What are the symptoms? What tests are done to
How To Treat Mesothelioma
 Mesothelioma in the UK Liz Darlison Mesothelioma UK The National Macmillan Mesothelioma Resource Centre June 2010 Mesothelioma Applied Research Foundation - 2010 www.curemeso.org Contents Size of Mesothelioma
Mesothelioma in the UK Liz Darlison Mesothelioma UK The National Macmillan Mesothelioma Resource Centre June 2010 Mesothelioma Applied Research Foundation - 2010 www.curemeso.org Contents Size of Mesothelioma
Asbestos Diseases. What Is Asbestos?
 1 Asbestos Diseases What Is Asbestos? Asbestos is a term applied to a group of minerals formed into rock and mined in a similar way to coal. In this form, asbestos is made up of strong, fine and flexible
1 Asbestos Diseases What Is Asbestos? Asbestos is a term applied to a group of minerals formed into rock and mined in a similar way to coal. In this form, asbestos is made up of strong, fine and flexible
GUIDELINES FOR THE MANAGEMENT OF LUNG CANCER
 GUIDELINES FOR THE MANAGEMENT OF LUNG CANCER BY Ali Shamseddine, MD (Coordinator); as04@aub.edu.lb Fady Geara, MD Bassem Shabb, MD Ghassan Jamaleddine, MD CLINICAL PRACTICE GUIDELINES FOR THE TREATMENT
GUIDELINES FOR THE MANAGEMENT OF LUNG CANCER BY Ali Shamseddine, MD (Coordinator); as04@aub.edu.lb Fady Geara, MD Bassem Shabb, MD Ghassan Jamaleddine, MD CLINICAL PRACTICE GUIDELINES FOR THE TREATMENT
Mesothelioma. 1995-2013, The Patient Education Institute, Inc. www.x-plain.com ocft0101 Last reviewed: 03/21/2013 1
 Mesothelioma Introduction Mesothelioma is a type of cancer. It starts in the tissue that lines your lungs, stomach, heart, and other organs. This tissue is called mesothelium. Most people who get this
Mesothelioma Introduction Mesothelioma is a type of cancer. It starts in the tissue that lines your lungs, stomach, heart, and other organs. This tissue is called mesothelium. Most people who get this
Asbestos and your lungs
 This information describes what asbestos is and the lung conditions that are caused by exposure to it. It also includes information about what to do if you have been exposed to asbestos, and the benefits
This information describes what asbestos is and the lung conditions that are caused by exposure to it. It also includes information about what to do if you have been exposed to asbestos, and the benefits
Survey of Mesothelioma Associated with Asbestos Exposure in Japan
 The research and development and the dissemination projects related to the 13 fields of occupational injuries and illnesses Survey of Mesothelioma Associated with Asbestos Exposure in Japan Clinical characteristics
The research and development and the dissemination projects related to the 13 fields of occupational injuries and illnesses Survey of Mesothelioma Associated with Asbestos Exposure in Japan Clinical characteristics
Lung Cancer Treatment Guidelines
 Updated June 2014 Derived and updated by consensus of members of the Providence Thoracic Oncology Program with the aid of evidence-based National Comprehensive Cancer Network (NCCN) national guidelines,
Updated June 2014 Derived and updated by consensus of members of the Providence Thoracic Oncology Program with the aid of evidence-based National Comprehensive Cancer Network (NCCN) national guidelines,
Diagnostic Challenge. Department of Pathology,
 Cytology of Pleural Fluid as a Diagnostic Challenge Paavo Pääkkö,, MD, PhD Chief Physician and Head of the Department Department of Pathology, Oulu University Hospital,, Finland Oulu University Hospital
Cytology of Pleural Fluid as a Diagnostic Challenge Paavo Pääkkö,, MD, PhD Chief Physician and Head of the Department Department of Pathology, Oulu University Hospital,, Finland Oulu University Hospital
Round Table with Drs. Anne Tsao and Alex Farivar, Case 2: Mesothelioma
 Round Table with Drs. Anne Tsao and Alex Farivar, Case 2: Mesothelioma I d like to welcome everyone, thanks for coming out to our lunch with experts. The faculty today are great people in the thoracic
Round Table with Drs. Anne Tsao and Alex Farivar, Case 2: Mesothelioma I d like to welcome everyone, thanks for coming out to our lunch with experts. The faculty today are great people in the thoracic
PRACTICE FRAMEWORK AND COMPETENCY STANDARDS FOR THE PROSTATE CANCER SPECIALIST NURSE
 PRACTICE FRAMEWORK AND COMPETENCY STANDARDS FOR THE PROSTATE CANCER SPECIALIST NURSE MARCH 2013 MONOGRAPHS IN PROSTATE CANCER OUR VISION, MISSION AND VALUES Prostate Cancer Foundation of Australia (PCFA)
PRACTICE FRAMEWORK AND COMPETENCY STANDARDS FOR THE PROSTATE CANCER SPECIALIST NURSE MARCH 2013 MONOGRAPHS IN PROSTATE CANCER OUR VISION, MISSION AND VALUES Prostate Cancer Foundation of Australia (PCFA)
Update on Mesothelioma
 November 8, 2012 Update on Mesothelioma Intro incidence and nomenclature Update on Classification Diagnostic specimens Morphologic features Epithelioid Histology Biphasic Histology Immunohistochemical
November 8, 2012 Update on Mesothelioma Intro incidence and nomenclature Update on Classification Diagnostic specimens Morphologic features Epithelioid Histology Biphasic Histology Immunohistochemical
HEALTH CARE FOR EXPOSURE TO ASBESTOS. 2010 The SafetyNet Centre for Occupational Health and Safety Research Memorial University www.safetynet.mun.
 HEALTH CARE FOR PATIENTS WITH EXPOSURE TO ASBESTOS 2010 The SafetyNet Centre for Occupational Health and Safety Research Memorial University www.safetynet.mun.ca HEALTH CARE FOR PATIENTS WITH EXPOSURE
HEALTH CARE FOR PATIENTS WITH EXPOSURE TO ASBESTOS 2010 The SafetyNet Centre for Occupational Health and Safety Research Memorial University www.safetynet.mun.ca HEALTH CARE FOR PATIENTS WITH EXPOSURE
FREQUENTLY ASKED QUESTIONS about asbestos related diseases
 FREQUENTLY ASKED QUESTIONS about asbestos related diseases 1. What are the main types of asbestos lung disease? In the human body, asbestos affects the lungs most of all. It can affect both the spongy
FREQUENTLY ASKED QUESTIONS about asbestos related diseases 1. What are the main types of asbestos lung disease? In the human body, asbestos affects the lungs most of all. It can affect both the spongy
Malignant Mesothelioma
 Malignant Mesothelioma What is malignant mesothelioma? Malignant mesothelioma is a cancer that starts in cells in the linings of certain parts of the body, especially in the linings of the chest or abdomen.
Malignant Mesothelioma What is malignant mesothelioma? Malignant mesothelioma is a cancer that starts in cells in the linings of certain parts of the body, especially in the linings of the chest or abdomen.
Management of Non-Small Cell Lung Cancer Guide for General Practitioners
 Management of n-small Cell Lung Cancer Guide for General Practitioners Clinical Stage I Cancer only in one lobe of lung and
Management of n-small Cell Lung Cancer Guide for General Practitioners Clinical Stage I Cancer only in one lobe of lung and
Principal Investigator: Valerie W. Rusch, MD, FACS, Chief, Thoracic Surgery Memorial Sloan-Kettering Cancer Center
 Protocol 1101-1088 Phase I study of intra-pleural administration of GL-ONC1 in patients with malignant pleural effusion: primary, metastases and mesothelioma Principal Investigator: Valerie W. Rusch, MD,
Protocol 1101-1088 Phase I study of intra-pleural administration of GL-ONC1 in patients with malignant pleural effusion: primary, metastases and mesothelioma Principal Investigator: Valerie W. Rusch, MD,
Malignant Mesothelioma
 Malignant Mesothelioma What is malignant mesothelioma? Malignant mesothelioma is a cancer that starts in cells in the linings of certain parts of the body, especially in the linings of the chest or abdomen.
Malignant Mesothelioma What is malignant mesothelioma? Malignant mesothelioma is a cancer that starts in cells in the linings of certain parts of the body, especially in the linings of the chest or abdomen.
Sternotomy and removal of the tumor
 Sternotomy and removal of the tumor All thymomas originate from epithelial thymic cells 4% of them consist of a pure population of epithelial cells Most have mixed populations of lymphoid cells to a
Sternotomy and removal of the tumor All thymomas originate from epithelial thymic cells 4% of them consist of a pure population of epithelial cells Most have mixed populations of lymphoid cells to a
Surgery. Wedge resection only part of the lung, not. not a lobe, is removed. Cancer Council NSW
 The treatment you receive will depend on your lung cancer type, for example, whether you have a non-small cell lung cancer Adenocarcinoma or Squamous cell carcinoma, and if this is a sub-type with a mutation.
The treatment you receive will depend on your lung cancer type, for example, whether you have a non-small cell lung cancer Adenocarcinoma or Squamous cell carcinoma, and if this is a sub-type with a mutation.
Malignant Pleural Mesothelioma in Singapore
 RESEARCH COMMUNICATION C SP Yip 1, HN Koong 2, CM Loo 3, KW Fong 1* Abstract Aim: To examine the clinical characteristics and outcomes of malignant pleural mesothelioma (MPM) in Singapore. Methods and
RESEARCH COMMUNICATION C SP Yip 1, HN Koong 2, CM Loo 3, KW Fong 1* Abstract Aim: To examine the clinical characteristics and outcomes of malignant pleural mesothelioma (MPM) in Singapore. Methods and
How To Prevent Asbestos Related Diseases
 BD5.3 Report of Working Groups Elimination of Asbestos-related Diseases ICOH 2012 March 18, 2012 Cancun Report of WG Elimination of Asbestos-related Diseases Dr. Sherson mail to ICOH President of 7 December
BD5.3 Report of Working Groups Elimination of Asbestos-related Diseases ICOH 2012 March 18, 2012 Cancun Report of WG Elimination of Asbestos-related Diseases Dr. Sherson mail to ICOH President of 7 December
Asbestos Health Risks. Dr Andrew Pengilley Acting Chief Health Officer
 Asbestos Health Risks Dr Andrew Pengilley Acting Chief Health Officer Asbestos Asbestos is a name given to several different fibrous minerals Three main commercial types are Chrysotile (white asbestos)
Asbestos Health Risks Dr Andrew Pengilley Acting Chief Health Officer Asbestos Asbestos is a name given to several different fibrous minerals Three main commercial types are Chrysotile (white asbestos)
Rotation Specific Goals & Objectives: University Health Network-Princess Margaret Hospital/ Sunnybrook Breast/Melanoma
 Rotation Specific Goals & Objectives: University Health Network-Princess Margaret Hospital/ Sunnybrook Breast/Melanoma Medical Expert: Breast Rotation Specific Competencies/Objectives 1.0 Medical History
Rotation Specific Goals & Objectives: University Health Network-Princess Margaret Hospital/ Sunnybrook Breast/Melanoma Medical Expert: Breast Rotation Specific Competencies/Objectives 1.0 Medical History
This factsheet aims to outline the characteristics of some rare lung cancers, and highlight where each type of lung cancer may be different.
 There are several different kinds of lung cancer, often referred to as lung cancer subtypes. Some of these occur more often than others. In this factsheet we will specifically look at the subtypes of cancers
There are several different kinds of lung cancer, often referred to as lung cancer subtypes. Some of these occur more often than others. In this factsheet we will specifically look at the subtypes of cancers
How To Treat Lung Cancer At Cleveland Clinic
 Treatment Guide Lung Cancer Management The Chest Cancer Center at Cleveland Clinic, which includes specialists from the Respiratory Institute, Taussig Cancer Institute and Miller Family Heart & Vascular
Treatment Guide Lung Cancer Management The Chest Cancer Center at Cleveland Clinic, which includes specialists from the Respiratory Institute, Taussig Cancer Institute and Miller Family Heart & Vascular
Occupational respiratory diseases due to Asbestos. Dirk Dahmann, IGF, Bochum
 Occupational respiratory diseases due to Asbestos Dirk Dahmann, IGF, Bochum Contents Introduction Diseases Further Effects Preventive Strategies Conclusion Asbestos minerals Woitowitz, 2003 Imports (+
Occupational respiratory diseases due to Asbestos Dirk Dahmann, IGF, Bochum Contents Introduction Diseases Further Effects Preventive Strategies Conclusion Asbestos minerals Woitowitz, 2003 Imports (+
How To Prepare A Meeting For A Health Care Conference
 THORACIC ONCOLOGY MULTIDISCIPLINARY TEAM MEETINGS: OPERATIONAL POLICY EXECUTIVE SUMMARY 1. All patients in Lothian with thoracic malignancies should be discussed at designated times in pathway (see App
THORACIC ONCOLOGY MULTIDISCIPLINARY TEAM MEETINGS: OPERATIONAL POLICY EXECUTIVE SUMMARY 1. All patients in Lothian with thoracic malignancies should be discussed at designated times in pathway (see App
Recommendations for cross-sectional imaging in cancer management, Second edition
 www.rcr.ac.uk Recommendations for cross-sectional imaging in cancer management, Second edition Breast cancer Faculty of Clinical Radiology www.rcr.ac.uk Contents Breast cancer 2 Clinical background 2 Who
www.rcr.ac.uk Recommendations for cross-sectional imaging in cancer management, Second edition Breast cancer Faculty of Clinical Radiology www.rcr.ac.uk Contents Breast cancer 2 Clinical background 2 Who
Treating Mesothelioma - A Quick Guide
 Treating Mesothelioma - A Quick Guide Contents This is a brief summary of the information on Treating mesothelioma from CancerHelp UK. You will find more detailed information on the website. In this information
Treating Mesothelioma - A Quick Guide Contents This is a brief summary of the information on Treating mesothelioma from CancerHelp UK. You will find more detailed information on the website. In this information
Pre-workshop exercise
 Setting research priorities for mesothelioma workshop 10 th November 2014 Pre-workshop exercise Your individual ranking of unanswered questions about the diagnosis, treatment and care of mesothelioma Please
Setting research priorities for mesothelioma workshop 10 th November 2014 Pre-workshop exercise Your individual ranking of unanswered questions about the diagnosis, treatment and care of mesothelioma Please
Understanding Pleural Mesothelioma
 Understanding Pleural Mesothelioma A guide for people with cancer, their families and friends Cancer information For information & support, call Understanding Pleural Mesothelioma A guide for people with
Understanding Pleural Mesothelioma A guide for people with cancer, their families and friends Cancer information For information & support, call Understanding Pleural Mesothelioma A guide for people with
Malignant Pleural Diseases Advances Clinicians Should Know F Gleeson
 Malignant Pleural Diseases Advances Clinicians Should Know F Gleeson The following relevant disclosures, conflicts of interest and/ or financial relationships exist related to this presentation: Consultant
Malignant Pleural Diseases Advances Clinicians Should Know F Gleeson The following relevant disclosures, conflicts of interest and/ or financial relationships exist related to this presentation: Consultant
Malignant Mesothelioma State of the Art
 Malignant Mesothelioma State of the Art Paul Baas The Netherlands Cancer Institute August 12, 2011, Carlsbad, CA Summary Diagnosis; epithelial type subdivided Pleiomorphic vs other Staging: IASLC-IMIG
Malignant Mesothelioma State of the Art Paul Baas The Netherlands Cancer Institute August 12, 2011, Carlsbad, CA Summary Diagnosis; epithelial type subdivided Pleiomorphic vs other Staging: IASLC-IMIG
Recommendations for the Reporting of Pleural Mesothelioma
 Recommendations for the Reporting of Pleural Mesothelioma Association of Directors of Anatomic and Surgical Pathology * DOI: 10.1309/6A30YQHBMTHEJTEM It has been evident for decades that pathology reports
Recommendations for the Reporting of Pleural Mesothelioma Association of Directors of Anatomic and Surgical Pathology * DOI: 10.1309/6A30YQHBMTHEJTEM It has been evident for decades that pathology reports
How To Understand How Cancer Works
 Mesothelioma Understanding your diagnosis Mesothelioma Understanding your diagnosis When you first hear that you have cancer, you may feel alone and afraid. You may be overwhelmed by the large amount of
Mesothelioma Understanding your diagnosis Mesothelioma Understanding your diagnosis When you first hear that you have cancer, you may feel alone and afraid. You may be overwhelmed by the large amount of
Lessons learned from the Western Australian experience with mesothelioma
 Lessons learned from the Western Australian experience with mesothelioma Alison Reid, Western Australian Institute for Medical Research In partnership with Nick de Klerk, Nola Olsen, Jan Sleith, Geoffrey
Lessons learned from the Western Australian experience with mesothelioma Alison Reid, Western Australian Institute for Medical Research In partnership with Nick de Klerk, Nola Olsen, Jan Sleith, Geoffrey
Malignant Pleural Mesothelioma. NCCN Guidelines for Patients
 Malignant Pleural Mesothelioma NCCN Guidelines for Patients Presented with support from the national law firm of Baron & Budd Also available at NCCN.com About this booklet Its purpose Learning that you
Malignant Pleural Mesothelioma NCCN Guidelines for Patients Presented with support from the national law firm of Baron & Budd Also available at NCCN.com About this booklet Its purpose Learning that you
Case of the. Month October, 2012
 Case of the Month October, 2012 Case The patient is a 47-year-old male with a 3-week history of abdominal pain. A CT scan of the abdomen revealed a suggestion of wall thickening at the tip of the appendix
Case of the Month October, 2012 Case The patient is a 47-year-old male with a 3-week history of abdominal pain. A CT scan of the abdomen revealed a suggestion of wall thickening at the tip of the appendix
Screening, early referral and treatment for asbestos related cancer
 Screening, early referral and treatment for asbestos related cancer Marc de Perrot, MD, MSc, FRCSC Toronto Mesothelioma Research Program University of Toronto Asbestos related diseases Mesothelioma Lung
Screening, early referral and treatment for asbestos related cancer Marc de Perrot, MD, MSc, FRCSC Toronto Mesothelioma Research Program University of Toronto Asbestos related diseases Mesothelioma Lung
Malignant Mesothelioma Current Approaches to a Difficult Problem. Raja M Flores, MD Thoracic Surgery Memorial Sloan-Kettering Cancer Center
 Malignant Mesothelioma Current Approaches to a Difficult Problem Raja M Flores, MD Thoracic Surgery Memorial Sloan-Kettering Cancer Center Malignant Pleural Mesothelioma Clinical Presentation Insidious
Malignant Mesothelioma Current Approaches to a Difficult Problem Raja M Flores, MD Thoracic Surgery Memorial Sloan-Kettering Cancer Center Malignant Pleural Mesothelioma Clinical Presentation Insidious
Summary of treatment benefits
 Risk Management Plan PEMETREXED Powder for concentrate for Solution for infusion Pemetrexed is also indicated as monotherapy for the maintenance treatment of locally advanced or metastatic non small cell
Risk Management Plan PEMETREXED Powder for concentrate for Solution for infusion Pemetrexed is also indicated as monotherapy for the maintenance treatment of locally advanced or metastatic non small cell
The Need for Accurate Lung Cancer Staging
 The Need for Accurate Lung Cancer Staging Peter Baik, DO Thoracic Surgery Cancer Treatment Centers of America Oklahoma Osteopathic Association 115th Annual Convention Financial Disclosures: None 2 Objectives
The Need for Accurate Lung Cancer Staging Peter Baik, DO Thoracic Surgery Cancer Treatment Centers of America Oklahoma Osteopathic Association 115th Annual Convention Financial Disclosures: None 2 Objectives
MesoPDT. Photodynamic Therapy for malignant pleural mesothelioma ONCO-THAI. Image Assisted Laser Therapy for Oncology
 MesoPDT Photodynamic Therapy for malignant pleural mesothelioma ONCO-THAI Image Assisted Laser Therapy for Oncology Unité Inserm ONCO-THAI «Image Assisted Laser Therapy for Oncology» Inserm ONCO-THAI "Image
MesoPDT Photodynamic Therapy for malignant pleural mesothelioma ONCO-THAI Image Assisted Laser Therapy for Oncology Unité Inserm ONCO-THAI «Image Assisted Laser Therapy for Oncology» Inserm ONCO-THAI "Image
How To Treat A Cancer With A Radical
 Management of mesothelioma Jan.vanmeerbeeck@ugent.be Amsterdam, March 6, 2010 1 management Palliation Symptomatic care Pain Breathlessness Radiotherapy Chemotherapy Surgery Radical (intention to cure)
Management of mesothelioma Jan.vanmeerbeeck@ugent.be Amsterdam, March 6, 2010 1 management Palliation Symptomatic care Pain Breathlessness Radiotherapy Chemotherapy Surgery Radical (intention to cure)
British Thoracic Society Standards of Care Committee
 250 Thorax 2001;56:250 265 BTS statement Statement on malignant mesothelioma in the United Kingdom British Thoracic Society Standards of Care Committee Correspondence to: Dr J Wiggins, Wexham Park Hospital,
250 Thorax 2001;56:250 265 BTS statement Statement on malignant mesothelioma in the United Kingdom British Thoracic Society Standards of Care Committee Correspondence to: Dr J Wiggins, Wexham Park Hospital,
Asbestos and the diseases it causes
 Asbestos and the diseases it causes October 2013 Liz Darlison Mesothelioma UK University Hospitals of Leicester Contents What is asbestos Why is it such an issue in the UK Disease Statistics Asbestos Related
Asbestos and the diseases it causes October 2013 Liz Darlison Mesothelioma UK University Hospitals of Leicester Contents What is asbestos Why is it such an issue in the UK Disease Statistics Asbestos Related
MALIGNANT PLEURAL MESOTHELIOMA TREATED IN CLINIC FOR PULMONARY DISEASES AND TUBERCULOSIS PODHRASTOVI IN TEN-YEAR PERIOD ( FROM 1998 TO 2007 )
 & MALIGNANT PLEURAL MESOTHELIOMA TREATED IN CLINIC FOR PULMONARY DISEASES AND TUBERCULOSIS PODHRASTOVI IN TEN-YEAR PERIOD ( FROM 1998 TO 2007 ) Vesna Čukić*, Aida Ustamujić, Vladimir Lovre, Hasan Žutić,
& MALIGNANT PLEURAL MESOTHELIOMA TREATED IN CLINIC FOR PULMONARY DISEASES AND TUBERCULOSIS PODHRASTOVI IN TEN-YEAR PERIOD ( FROM 1998 TO 2007 ) Vesna Čukić*, Aida Ustamujić, Vladimir Lovre, Hasan Žutić,
PRINCESS MARGARET CANCER CENTRE CLINICAL PRACTICE GUIDELINES
 PRINCESS MARGARET CANCER CENTRE CLINICAL PRACTICE GUIDELINES LUNG SITE MESOTHELIOMA Lung Site Group Mesothelioma Date Guideline Created: April 2013 Authors: Dr. Meredith Giuliani, Dr. Andrea Bezjak 1.
PRINCESS MARGARET CANCER CENTRE CLINICAL PRACTICE GUIDELINES LUNG SITE MESOTHELIOMA Lung Site Group Mesothelioma Date Guideline Created: April 2013 Authors: Dr. Meredith Giuliani, Dr. Andrea Bezjak 1.
PLEURAL MESOTHELIOMA
 PLEURAL MESOTHELIOMA PLEURAL MESOTHELIOMA By: Domenico Galetta Institute of Oncology Giovanni Paolo II I.R.C.C.S. Oncology Hospital of Bari, Italy Silvia Novello University of Torino, Italy San Luigi Hospital
PLEURAL MESOTHELIOMA PLEURAL MESOTHELIOMA By: Domenico Galetta Institute of Oncology Giovanni Paolo II I.R.C.C.S. Oncology Hospital of Bari, Italy Silvia Novello University of Torino, Italy San Luigi Hospital
Case Report Pleural Mesothelioma Presenting as Periumbilical Metastasis: The First Clinical Documentation
 Volume 2013, Article ID 198729, 4 pages http://dx.doi.org/10.1155/2013/198729 Case Report Pleural Mesothelioma Presenting as Periumbilical Metastasis: The First Clinical Documentation R. F. Falkenstern-Ge,
Volume 2013, Article ID 198729, 4 pages http://dx.doi.org/10.1155/2013/198729 Case Report Pleural Mesothelioma Presenting as Periumbilical Metastasis: The First Clinical Documentation R. F. Falkenstern-Ge,
7. Prostate cancer in PSA relapse
 7. Prostate cancer in PSA relapse A patient with prostate cancer in PSA relapse is one who, having received a primary treatment with intent to cure, has a raised PSA (prostate-specific antigen) level defined
7. Prostate cancer in PSA relapse A patient with prostate cancer in PSA relapse is one who, having received a primary treatment with intent to cure, has a raised PSA (prostate-specific antigen) level defined
What If I Have a Spot on My Lung? Do I Have Cancer? Patient Education Guide
 What If I Have a Spot on My Lung? Do I Have Cancer? Patient Education Guide A M E R I C A N C O L L E G E O F C H E S T P H Y S I C I A N S Lung cancer is one of the most common cancers. About 170,000
What If I Have a Spot on My Lung? Do I Have Cancer? Patient Education Guide A M E R I C A N C O L L E G E O F C H E S T P H Y S I C I A N S Lung cancer is one of the most common cancers. About 170,000
Avastin: Glossary of key terms
 Avastin: Glossary of key terms Adenocarcinoma Adenoma Adjuvant therapy Angiogenesis Anti-angiogenics Antibody Antigen Avastin (bevacizumab) Benign A form of carcinoma that originates in glandular tissue.
Avastin: Glossary of key terms Adenocarcinoma Adenoma Adjuvant therapy Angiogenesis Anti-angiogenics Antibody Antigen Avastin (bevacizumab) Benign A form of carcinoma that originates in glandular tissue.
Lung Cancer Multidisciplinary Meeting Toolkit. National Lung Cancer Working Group
 Lung Cancer Multidisciplinary Meeting Toolkit National Lung Cancer Working Group September 2014 Contents Introduction 1 Multidisciplinary meetings 1 Toolkit for implementing high-quality lung cancer MDMs
Lung Cancer Multidisciplinary Meeting Toolkit National Lung Cancer Working Group September 2014 Contents Introduction 1 Multidisciplinary meetings 1 Toolkit for implementing high-quality lung cancer MDMs
The National Clinical Lung Cancer Audit (LUCADA)
 The National Clinical Lung Cancer Audit (LUCADA) DATA MANUAL Title: Version: 3.1.5 Date: September 2013 LUCADA Lung Cancer Audit VERSION HISTORY Version Date Issued Brief Summary of Change Owner s Name
The National Clinical Lung Cancer Audit (LUCADA) DATA MANUAL Title: Version: 3.1.5 Date: September 2013 LUCADA Lung Cancer Audit VERSION HISTORY Version Date Issued Brief Summary of Change Owner s Name
Accelerated hemithoracic radiation followed by extrapleural pneumonectomy for malignant pleural mesothelioma
 Accelerated hemithoracic radiation followed by extrapleural pneumonectomy for malignant pleural mesothelioma Marc de Perrot, Ronald Feld, Natasha B Leighl, Andrew Hope, Thomas K Waddell, Shaf Keshavjee,
Accelerated hemithoracic radiation followed by extrapleural pneumonectomy for malignant pleural mesothelioma Marc de Perrot, Ronald Feld, Natasha B Leighl, Andrew Hope, Thomas K Waddell, Shaf Keshavjee,
The develpemental origin of mesothelium
 Mesothelioma Tallinn 14.12.06 Henrik Wolff Finnish Institute of Occupational Health The develpemental origin of mesothelium Mesodermal cavities (pleura, peritoneum and pericardium ) are lined with mesenchymal
Mesothelioma Tallinn 14.12.06 Henrik Wolff Finnish Institute of Occupational Health The develpemental origin of mesothelium Mesodermal cavities (pleura, peritoneum and pericardium ) are lined with mesenchymal
Mesothelioma and Asbestos
 CANCER INFORMATION FACTSHEET Mesothelioma and Asbestos The information in this factsheet will help you to understand more about mesothelioma. It is an agreed view on this cancer by medical experts. We
CANCER INFORMATION FACTSHEET Mesothelioma and Asbestos The information in this factsheet will help you to understand more about mesothelioma. It is an agreed view on this cancer by medical experts. We
OBJECTIVES By the end of this segment, the community participant will be able to:
 Cancer 101: Cancer Diagnosis and Staging Linda U. Krebs, RN, PhD, AOCN, FAAN OCEAN Native Navigators and the Cancer Continuum (NNACC) (NCMHD R24MD002811) Cancer 101: Diagnosis & Staging (Watanabe-Galloway
Cancer 101: Cancer Diagnosis and Staging Linda U. Krebs, RN, PhD, AOCN, FAAN OCEAN Native Navigators and the Cancer Continuum (NNACC) (NCMHD R24MD002811) Cancer 101: Diagnosis & Staging (Watanabe-Galloway
The Center for Prostate Cancer. Personalized Treatment. Clinical Excellence.
 The Center for Prostate Cancer Personalized Treatment. Clinical Excellence. The Center for Prostate Cancer Leaders in Prostate Cancer Treatment and Research The Center for Prostate Cancer at the North
The Center for Prostate Cancer Personalized Treatment. Clinical Excellence. The Center for Prostate Cancer Leaders in Prostate Cancer Treatment and Research The Center for Prostate Cancer at the North
Na#onal Asbestos Forum 2013: Advance in Medical Research on Asbestos- Related Diseases
 Na#onal Asbestos Forum 2013: Advance in Medical Research on Asbestos- Related Diseases Professor Nico van Zandwijk Asbestos Diseases Research Ins#tute Content List of Asbestos- Related Diseases Epidemiology
Na#onal Asbestos Forum 2013: Advance in Medical Research on Asbestos- Related Diseases Professor Nico van Zandwijk Asbestos Diseases Research Ins#tute Content List of Asbestos- Related Diseases Epidemiology
Guidelines for Management of Renal Cancer
 Guidelines for Management of Renal Cancer Date Approved by Network Governance July 2012 Date for Review July 2015 Changes Between Versions 2 and 3 Section 5 updated bullets 5.3 and 5.4 Section 6 updated
Guidelines for Management of Renal Cancer Date Approved by Network Governance July 2012 Date for Review July 2015 Changes Between Versions 2 and 3 Section 5 updated bullets 5.3 and 5.4 Section 6 updated
There must be an appropriate administrative structure for each residency program.
 Specific Standards of Accreditation for Residency Programs in Radiation Oncology 2015 VERSION 3.0 INTRODUCTION The purpose of this document is to provide program directors and surveyors with an interpretation
Specific Standards of Accreditation for Residency Programs in Radiation Oncology 2015 VERSION 3.0 INTRODUCTION The purpose of this document is to provide program directors and surveyors with an interpretation
Uses and Abuses of Pathology in Asbestos-exposed Populations
 Uses and Abuses of Pathology in Asbestos-exposed Populations Jerrold L. Abraham, MD Department of Pathology State University of New York Upstate Medical University Syracuse, NY, 13210 USA The term: Asbestosis,
Uses and Abuses of Pathology in Asbestos-exposed Populations Jerrold L. Abraham, MD Department of Pathology State University of New York Upstate Medical University Syracuse, NY, 13210 USA The term: Asbestosis,
Mesothelioma and Asbestos
 Cancer information factsheet Mesothelioma and Asbestos The information in this factsheet will help you to understand more about mesothelioma. It is an agreed view on this cancer by medical experts. We
Cancer information factsheet Mesothelioma and Asbestos The information in this factsheet will help you to understand more about mesothelioma. It is an agreed view on this cancer by medical experts. We
MESOTHELIOMA. Not Just a Late Night Commercial. Graciela Hoal, RN, MSN, ACNP-BC
 MESOTHELIOMA Not Just a Late Night Commercial Graciela Hoal, RN, MSN, ACNP-BC Saturday Session Thoracic Surgery Nurse Practitioner Greater Los Angeles Veteran Affairs Objectives Course Objectives: Discuss
MESOTHELIOMA Not Just a Late Night Commercial Graciela Hoal, RN, MSN, ACNP-BC Saturday Session Thoracic Surgery Nurse Practitioner Greater Los Angeles Veteran Affairs Objectives Course Objectives: Discuss
Neoplasms of the LUNG and PLEURA
 Neoplasms of the LUNG and PLEURA 2015-2016 FCDS Educational Webcast Series Steven Peace, BS, CTR September 19, 2015 2015 Focus o Anatomy o SSS 2000 o MPH Rules o AJCC TNM 1 Case 1 Case Vignette HISTORY:
Neoplasms of the LUNG and PLEURA 2015-2016 FCDS Educational Webcast Series Steven Peace, BS, CTR September 19, 2015 2015 Focus o Anatomy o SSS 2000 o MPH Rules o AJCC TNM 1 Case 1 Case Vignette HISTORY:
Optimal care pathway for people with lung cancer
 Optimal care pathway for people with lung cancer Optimal care pathway for people with lung cancer To receive this publication in an accessible format phone 03 9096 2136 using the National Relay Service
Optimal care pathway for people with lung cancer Optimal care pathway for people with lung cancer To receive this publication in an accessible format phone 03 9096 2136 using the National Relay Service
Guideline for the Imaging of Patients Presenting with Breast Symptoms incorporating the guideline for the use of MRI in breast cancer
 Guideline for the Imaging of Patients Presenting with Breast Symptoms incorporating the guideline for the use of MRI in breast cancer Version History Version Date Summary of Change/Process 0.1 09.01.11
Guideline for the Imaging of Patients Presenting with Breast Symptoms incorporating the guideline for the use of MRI in breast cancer Version History Version Date Summary of Change/Process 0.1 09.01.11
PET POSITIVE PLEURAL PLAQUES DECADES AFTER PLEURODESIS: MESOLTHELIOMA? Ellen A. Middleton 1. Jonathan C. Daniel 2. Kenneth S.
 PET POSITIVE PLEURAL PLAQUES DECADES AFTER PLEURODESIS: MESOLTHELIOMA? Ellen A. Middleton 1 Jonathan C. Daniel 2 Kenneth S. Knox 1 Kathleen Williams 1 Departments of Medicine 1 and Surgery 2, University
PET POSITIVE PLEURAL PLAQUES DECADES AFTER PLEURODESIS: MESOLTHELIOMA? Ellen A. Middleton 1 Jonathan C. Daniel 2 Kenneth S. Knox 1 Kathleen Williams 1 Departments of Medicine 1 and Surgery 2, University
Small cell lung cancer
 Small cell lung cancer Small cell lung cancer is a disease in which malignant (cancer) cells form in the tissues of the lung. The lungs are a pair of cone-shaped breathing organs that are found within
Small cell lung cancer Small cell lung cancer is a disease in which malignant (cancer) cells form in the tissues of the lung. The lungs are a pair of cone-shaped breathing organs that are found within
CANCERS OF THE PERITONEUM PERITONEAL MESOTHELIOMA PREFERRED MODEL OF CARE AND CRITERIA FOR REFERENCE CENTRES
 CANCERS OF THE PERITONEUM PERITONEAL MESOTHELIOMA PREFERRED MODEL OF CARE AND CRITERIA FOR REFERENCE CENTRES Coordinator: Alex Kartheuser (Colorectal Surgery, Cliniques universitaires St-Luc, UCL) Authors
CANCERS OF THE PERITONEUM PERITONEAL MESOTHELIOMA PREFERRED MODEL OF CARE AND CRITERIA FOR REFERENCE CENTRES Coordinator: Alex Kartheuser (Colorectal Surgery, Cliniques universitaires St-Luc, UCL) Authors
