Le Calcium, C est la Vie: Calcium Makes Waves 1
|
|
|
- Daniella Lewis
- 8 years ago
- Views:
Transcription
1 Plant Physiology, May 1999, Vol. 120, pp. 1 6, American Society of Plant Physiologists Update on Regulation Le Calcium, C est la Vie: Calcium Makes Waves 1 Anthony Trewavas* Institute of Cell and Molecular Biology, University of Edinburgh, Kings Building, Mayfield Road, Edinburgh EH9 3JH, Scotland, United Kingdom Every reader of this article knows that he or she started life as a single cell. Less familiar is the debt we owe to calcium in our earliest seconds. Penetration of the egg by the paternal sperm initiated an epigenetic calcium wave that moved quickly as a hollow band across the cytoplasm. In the wake of this calcium wave, processes were activated that led to cell division, differentiation, growth, and our eventual appearance as mature adults. As it is said in France, Le calcium, c est la vie. A calcium wave marked the onset of our existence, and will quite probably mark our demise: Irreversible failure of calcium-wave generation in the heart is the most common cause of death. Therefore, calcium waves are a life-and-death issue. So much depends on the intricacies and cellular patterning of the simple ion that is calcium. How much is understood about calcium waves? What is the mechanism of calciumwave formation? What is their significance? What specific role could calcium waves perform in plant cells? Some profound insights come from considering these questions. The true significance of calcium waves may lie in constructing a cellular intelligence. CALCIUM IS NOT A MOBILE ION IN THE CYTOPLASM In 1957, Hodgkin and Keynes performed a simple but seminal experiment, injecting small portions of the recently developed 45 Ca and 42 K into squid axons. Several hours later, they transversely sectioned the axon and examined the distribution of these isotopes. Whereas 42 K had uniformly diffused throughout the axon, 45 Ca had remained at the site of injection (Fig. 1), showing that calcium is not freely mobile in the cytoplasm. If calcium does move within the cytoplasm, mechanisms other than diffusion are responsible. Two facts can account for the above observations. Calcium binds to many proteins that are attached to the cytoskeleton or to membrane surfaces. Estimates suggest a cytoplasmicbinding capacity per cell of 0.1 to 0.5 mm calcium (Mahlo et al., 1998). Calcium-dependent calcium-atpases rapidly pump excess calcium into organelles, vesicles, and, in plant cells, out into the cell wall. The vacuole is a major repository 1 This report was supported in part by the Biotechnological and Biological Sciences Research Council. * Corresponding author; trewavas@ed.ac.uk; fax of plant-cell intracellular calcium and must make an unusual contribution to cell-calcium signaling. Other important intracellular calcium stores are the ER, the mitochondria, and possibly the Golgi vesicles. Intracellular stores and the cell wall may contain 0.1 to 10.0 mm calcium. The calcium remaining in the cytoplasm after protein binding and organelle uptake is between 10 and 200 nm. This is called resting calcium. There are pronounced electrochemical concentration gradients of calcium between the cytoplasm and the cell wall and between the cytoplasm and the intracellular vesicles and organelles. CALCIUM CHANNELS CONNECT THE STORES OF CALCIUM WITH THE CYTOPLASM Calcium-channel proteins permit the flow of calcium between the cytoplasm (with its very low concentrations) and the other cellular compartments (with much higher concentrations). Channels open when cells are signaled, and calcium enters the cytoplasm down its electrochemical gradient. Families of channels are known to exist, some activated by membrane potential, others by membrane stretch, and still others by various kinds of signals, including second messengers. A single channel can transmit 10 6 atoms of calcium per second. Elevation of calcium at the cytoplasmic channel mouth can be rapid: calcium concentrations there can reach 0.1 mm. However, channels rapidly close when the cytoplasmic level increases and the activation of calciumdependent calcium-atpases causes calcium to move back to the intracellular stores and into the cell wall. Transient elevations of calcium (sometimes called spikes) may last anywhere from a few seconds to many minutes in plant cells, depending on the characteristics of the stimulating signal. HOW TO MAKE CALCIUM MORE MOBILE IN THE CYTOPLASM The observations by Hodgkin and Keynes (1957) make it difficult to understand how calcium waves can move. If calcium is not mobile in the cytoplasm, then channel opening would limit calcium elevations to the domain of the channel mouth. Although a wave might be initiated, it could not move. The discovery of the role of inositol phos- Abbreviations: IP 3, inositol-1,4,5-trisphosphate; PIP 2, phosphatidylinositol-4,5-bisphosphate.
2 2 Trewavas Plant Physiol. Vol. 120, 1999 HOW TO MAKE THE CALCIUM WAVE MOVE Figure 1. Diagrammatic summary of the Hodgkin and Keynes (1957) experiment. Solid line, 45 Ca; dotted line, 42 K. phates in the generation of calcium waves provided a partial answer to this dilemma. Phospholipase C, which is bound to the plasma membrane and activated by signals, is present as several isoforms in plant cells and many isoforms in animal cells. The substrate for phospholipase C, PIP 2, is synthesized from precursors in the plasma membrane. When phospholipase C is activated, PIP 2 is hydrolyzed to diglyceride and IP 3. Unlike calcium, IP 3 is freely mobile in the cytoplasm. IP 3 -sensitive channels located in the membranes of organelles, vesicles, and the plasma membrane are opened, enabling calcium to enter the cytoplasm. Alexandre et al. (1990) and Gilroy et al. (1990) established the IP 3 mobilization of calcium in plant cells a decade ago; more recently, Allen et al. (1995) demonstrated IP 3 -binding (and heparin-sensitive) channels. When an IP 3 -dependent channel opens, the calcium concentration surrounding adjacent channels increases (Fig. 2B). The calcium-binding sites of these channels will be occupied and will briefly open, enabling the opening of others. Calcium is therefore responsible for inducing further calcium release, and this release underpins wave movement. This wave is not a forward movement of calcium, but a forward movement of calcium release. IP 3 - induced inactivation of channels causes the direction of calcium-wave movement to be away from the point of origin (Fig. 3A). IP 3 -sensitive channels are located in membranes, and the calcium wave thus moves across the cytoplasmic membrane face. Many calcium-dependent proteins (e.g. calmodulin and calcium/calmodulin-dependent protein kinases) are bound to the membrane surface and will therefore be activated (Collinge and Trewavas, 1989). Processes downstream from calcium signaling (e.g. opening of potassium and chloride channels and calcium-dependent gene expression) will be induced. The domains on the membrane surface are difficult to examine, but new technology with targeted calcium probes is providing new insights (Knight et al., 1996). Other second messengers (e.g. ADP-Rib) can also mobilize calcium from intracellular stores (Allen et al., 1995). Another layer of complexity in calcium signaling is therefore added. HOW TO MAKE A CALCIUM WAVE Although these observations helped our understanding, they did not resolve the question of calcium-wave construction and movement (Fig. 2A). Elevation of IP 3 and its rapid diffusion throughout the cytoplasm would simply open all intracellular IP 3 -dependent channels; no spatial or kinetic distinction between different regions of the cell would be detected and no calcium wave would be observed. Only with purification and kinetic analysis of the IP 3 -sensitive channel was the conundrum resolved: Marchant and Taylor (1997) found that the IP 3 channel needs both calcium and IP 3 to open. When IP 3 binds to the channel, a calcium-binding site is briefly exposed. If calcium is absent, the channel rapidly inactivates (Fig. 2A); if calcium is present, the calciumbinding site is occupied and the channel is opened. But the binding of calcium serves only to delay eventual IP 3 - induced channel inactivation. Protein phosphorylation probably inactivates the channel, but the mechanism is not yet understood. Mobilization of calcium through a single IP 3 -dependent channel is therefore brief and self-limiting. However, IP 3 -sensitive channels act as a kind of coincidence counter of local calcium and IP 3 concentrations, safeguarding against spontaneous opening. Kinetic analysis of the IP 3 channel indicates flexibility in the requirements for calcium and IP 3. High concentrations of calcium need only low concentrations of IP 3 to open the channel; conversely, high concentrations of IP 3 need only low concentrations of calcium. Figure 2. The effect of IP 3 and calcium on the opening of IP 3 - sensitive channels. A, Closed-channel binding only IP 3 and an openchannel binding both IP 3 and calcium. B, Illustration of how an open channel opens an adjacent channel by locally increasing calcium.
3 Le Calcium, C est la Vie: Calcium Makes Waves 3 In the presence of optimal cellular IP 3 concentrations, a full calcium wave is likely. The wave will move from its initiation site progressively throughout the cell. The specific characteristics of initiation sites are not known but might reflect the clustering of receptors or an unusual density of channels. However, there are many situations in which the wave will be truncated and elevation of calcium will be limited to particular regions of the membrane surface. First, IP 3 is rapidly hydrolyzed by specific phosphatases. If the initial stimulating signal is weak, IP 3 elevations will be low and will rapidly decline below the critical threshold. The calcium wave will be confined to specific cellular regions and can be described as clouds or puffs. Second, spatial separation of phospholipase C from IP 3 - specific phosphatases will generate transient gradients of IP 3. Only where IP 3 exceeds a critical threshold will spatially constrained increases of calcium be detected. Third, the calcium contents of regions of the ER are known to vary. In regions that contain insufficient stored calcium, wave propagation will cease. The final wave will have a complex topology composed of high and low calcium concentrations and regions where no calcium elevation is detectable. Spatial differentiation of calciumdependent proteins on the membrane surface enables many different combinations of downstream proteins to be activated. Fourth, if calcium-dependent calcium-atpases are clustered on the membrane surface, a complex topological calcium wave will again result. And, finally, whether waves jump from one membrane surface to another depends on the distance between them and the strength of the initial signal. The wave-initiation site determines the region in which the wave starts. Figure 4 illustrates the expected spatial distribution of calcium elevation as a result of truncated waves induced by different signals. CALCIUM WAVES IN PLANT CELLS Figure 3. Comparison of the information flow through a calcium cloud and a simple neuronal network. A, Generation of a wavefront of calcium release through a number of IP 3 -sensitive channels from a single activated channel (black arrows). B, Diversification of a single electrical signal through a neuronal network with the signal impacting on a single neuron (black arrows). CONSTRAINTS ON CALCIUM-WAVE FORMATION AND LIFETIME IP 3 channels are located in the vacuole membrane (tonoplast) and almost certainly in the ER and plasma membrane of plant cells. Calcium waves can be induced in pollen tubes by photolytic release of IP 3 from loaded, caged IP 3 (Franklin-Tong et al., 1996). The wave initiates in a cellular region containing the nucleus and ER and moves to the tip within 1 min via a relay of IP 3 -sensitive channels. The absence of a vacuole in this region of the pollen tube indicates that the tonoplast is not involved in the transmission of this calcium wave. Some of these IP 3 -sensitive channels are found in the plasma membrane. Caged IP 3 photolysis in pollen tubes containing heparin (to inhibit binding of IP 3 to the channel) permits wave initiation; however, the wave is rapidly truncated and fails to reach the tip. A calcium wave in pollen tubes can also be induced by localized photolytic release of calcium in just the cellular region containing the nucleus and ER (Mahlo and Trewavas, 1996). These observations confirm that calcium and IP 3 can separately activate plant IP 3 -sensitive channels. Furthermore, a wave-initiation site has been clearly identified. Calcium waves have also been induced by suddenly reduc- Figure 4. Anticipated changes in cytosolic calcium by a truncated calcium wave initiated by different signals in different parts of the cell. Spatial differences should initiate different molecular and physiological changes.
4 4 Trewavas Plant Physiol. Vol. 120, 1999 ing the plasma membrane potential of guard cells and subjecting Fucus rhizoids to hypo-osmotic shock, and in both cases the wave was initiated near the cell periphery (Taylor et al., 1996; Grabov and Blatt, 1998). Clouds of calcium confined to discrete cellular regions have been imaged in guard cells induced to close with different signals. The spatial location of the cloud differed according to the signal used (Gilroy et al., 1991). DIFFERENT SIGNALS WILL INDUCE CALCIUM WAVES OF DIFFERENT SHAPE, FORM, AND KINETICS IN PLANT CELLS Different signals do not uniformly activate phospholipase C. Furthermore, the strength of any signal should quantitatively modify cellular IP 3 concentrations. Variable kinetics in the development of the calcium wave will result. Because a number of channels must open within the vicinity of each other and within a certain time period for calcium-wave initiation, a lag period may also be detected. Lag periods may be common when IP 3 concentrations are low. Many factors (e.g. channel density, channel state, intracellular store replenishment, other second messengers, and the age and state of development of the cell) may also contribute to the final kinetics of the wave. Table I summarizes the measured kinetics of calcium transients in tobacco seedlings after initiation by different signals. Each signal does indeed induce unique kinetics and therefore should induce a unique topology of calcium elevation (the so-called fingerprint) and activate unique combinations of calcium-dependent proteins. Some cellular states and signals will enable repetitive wave initiation. Repetition may lead to oscillations of calcium, which have been observed in guard cells, root hairs, pollen tubes, and tobacco seedlings (for refs., see Trewavas and Mahlo, 1997). NEURAL NETWORKS PROVIDE ORGANISMS WITH INTELLIGENCE Figure 3B indicates that there is a formal equivalence in character, structure, and information flow between a neural network and a calcium wave. Neural networks, both Table I. Characteristics of the calcium transient in tobacco seedlings after the imposition of different signals All data were obtained on young tobacco seedlings transformed with the calcium-sensitive, luminescent protein aequorin. Further details are provided by Mahlo et al. (1998). Signal Lag Period Rise Time Total Transient s Oxidative shock Wind Cold shock Hyperosmotic shock Anoxia Elicitors Blue light Heat shock several min 10 min 30 min real and artificial, have five important properties that enable them to act intelligently: (a) they are spatially structured; (b) individual neurons can act as coincidence controllers, passing or blocking specific signals arriving coincidentally from different neurons and thus from different signals (protein kinases control information flow through neurons; Abel et al., 1998); (c) synchronization among the membrane potentials of many neurons leads to rhythmic oscillations (e.g. -rhythms) in membrane potential, which are modified by further signaling; (d) neural networks learn ; and (e) memory of previous signals can be accessed and used to modify current signal information. Learning and memory are interlinked. Networks can learn by reinforcing a neural pathway connecting the signal and the response. Reinforcement can occur either by increasing the numbers of dendrites (connections) or by increasing the strength of existing connections between adjacent neurons in the pathway. The latter process is thought to involve integrins (Grotweil et al., 1998). The consequence of learning is an enormous acceleration in information flux between the signal and the elements initiating the desired response. THE CALCIUM WAVE IS ANALOGOUS TO AN OPERATING NEURAL NETWORK If the calcium signaling system has a formal equivalence to a neural network, it should be able to compute, remember, and learn even though it is confined to single cells. Although admittedly more limited than a complex neural network, calcium waves most certainly provide for some aspects of intelligent behavior by plants. Even simple neural nets involving no more than 12 to 14 neurons have good computational properties (Lewis and Kristan, 1998), and a calcium system should do better. The parallels between neural networks and calcium waves are striking: the calcium wave is spatially structured. The IP 3 -sensitive channel can act as a coincidence counter (specific directions of calcium release can be propagated whereas others are inhibited) and channel inactivation can be regulated by protein kinases that regulate coincidence signaling. Oscillations in cellular calcium represent the cooperative integration of the behavior of many individual IP 3 -sensitive channels. When plant cells are signaled, they increase the cellular constituents concerned with calcium signaling. The cellular content of phospholipase C, calmodulin, calcium-activated protein kinases and other kinases, cadp-rib, PIP 2, and IP 3 are all increased by signaling (Braam and Davis, 1990; Drobak and Watkins, 1994; Urao et al., 1994; Hirayama et al., 1995; Wu et al., 1997). However else these data might be regarded, this is a clear example of cellular learning. The consequence of these changes is an accelerated information flux through calcium-dependent pathways and is analogous to reinforcement. So far as I am aware, this property of increasing the cellular content of signal components is unique to plants. Some of the early difficulties in detecting inositol phosphates such as PIP 2 and IP 3 can be traced to using unstimulated cells for investigation. ABA increases the cellular
5 Le Calcium, C est la Vie: Calcium Makes Waves 5 accumulations of cadp-rib, and the wall removal that forms protoplasts also elevates IP 3. The changes in these constituents are often slow, taking many hours, which of course emphasizes the similarity to neural network learning (Drobak and Watkins, 1994; Wu et al., 1997). In an animal cell, for example, complete elevations of inositol phosphates tend to occur in 0.1 s or less. Another similarity between neural networks and calcium waves is that cells are able to access a memory because a similar calcium signal in different cell types initiates different effects. Cells know what they are and where they are. They are able to remember previous experience and incorporate this information to modify current signals. Important aspects of this memory reside in the cell wall because cell wall removal resets many aspects of development in algae and higher plants (Pennell, 1998). A calcium-based memory can result from exhaustion of calcium stores from certain areas of the ER. The final topology of any calcium wave passing over this ER surface would reflect variations in ER-stored calcium. A unique combination of effector proteins would thus be activated. However, this memory would be time-limited because cellular membranes are dynamic in characteristics and structure. In the cell wall, on the other hand, calcium-pectate secretion could be targeted to discrete wall areas during prior signaling, growth, and development. Calcium concentrations outside the plasma membrane would be clustered in a spatially unique pattern. The development of a calcium wave on the plasma membrane surface would be propagated only in those regions where calcium was sufficiently high. The final geometry of the wave would represent the integration of the present signal modified by a stable, longterm memory. Protein distribution on the plant plasma membrane has been observed to be highly differentiated. If plasma membrane effector proteins (e.g. calmodulin) are clustered in specific regions, novel physiological outcomes result. Verdus et al. (1997) observed that exogenous calcium can modify a long-term memory controlling regeneration in plant cells. Bands of wall material have been observed to be laid down in growing pollen tubes (Pierson et al., 1995), suggesting that localized pectin secretion is feasible. WHY DO PLANTS NEED AN INTELLIGENT COMPUTATIONAL SYSTEM? Intelligence is not usually associated with plant behavior. But intelligence is not the same as sentience. Both Darwin and Sachs have commented on the similarity between plant cell signal transduction and neural network behavior: In several respects light seems to act on plants in nearly the same manner as it does on animals by means of the nervous system (Darwin, 1880); and I have repeatedly had cause to refer to certain resemblances between the phenomena of irritability in the vegetable kingdom and those of the animal body (Sachs, 1881). What particular problems faced by plants require intelligent behavior? Wild seedlings must grow where they land. The external environment is composed of probably 17 distinct constituents and, being variable in intensity even from minute to minute, generates an almost infinite variety of environmental states. There are probably as many internal plant signals that either pass through or are perceived at the plasma membrane (Trewavas and Mahlo, 1997). In responding intelligently to this multiplicity of signals, plants have become masters of phenotypic and physiological plasticity, which allows them to cope with the variable circumstances that surround them. Behavioral plasticity surely demands a cellular system of considerable computing power if plants are to survive the signal morass in which they find themselves. The ubiquity of calcium involvement in plant-cell signal transduction suggests that calcium forms the basis of the intelligent system controlling plasticity. Many examples of phenotypic plasticity exist. I have chosen one example, root branching, to illustrate this article; however, the reader can construct additional scenarios for any of the multiple phenotypically plastic responses to water, light, herbivory, pests, wind, temperature, minerals, soil factors, and so on. Leaf abscission and herbivory provide excellent examples (Addicott, 1982; Karban and Baldwin, 1997). There are far too many environmental situations for simple genetic control to work; so epigenetic control must be the ultimate regulator. In the laboratory Drew et al. (1973) have shown that the primary roots of cereals produce a profusion of lateral roots when growing in layers of soil rich in nitrate or phosphate. Some plants can efficiently mine the soil, much as humans would. But in wild grasses much other information that will modify the outcome has to be taken into account (Trewavas, 1992): the age of the root, the state of development of the pericycle, whether other laterals have already formed (grasses produce a mat of secondary roots that efficiently exploit the surrounding soil), the quality of the soil (e.g. balance, excess of other minerals, or compactness), the availability of water (which may override other factors), the state of the shoot (etiolated, shaded or green, damaged, or eaten), nearness of the shoot, wind sway, carbohydrate levels, availability of growth factors and amino acids, nearness of or damage by parasites, ethylene, carbon dioxide and oxygen in the soil, flowering, and so on. Each of these variables known to modify root branching does so quantitatively. The combination of possible environmental circumstances that pericycle cells must assess is enormous, and the amount of information that has to be computed rapidly mounts up; but after assessment, the numbers of pericycle cells to be induced or root primordia to be formed will be decided. The decision will be intelligent, geared in some way to an optimal solution for survival of the particular individual plant. As plants represent 99% or more of the biomass of the earth, the computing power has some degree of accuracy! CALCIUM SIGNALING IS AN EMERGENT PROPERTY Cellular molecules, cells, tissues, whole plants, populations, and ecosystems are frequently arranged in a hierarchy. At each level of the hierarchy, the connections between the constituents generate the level above. The 50,000
6 6 Trewavas Plant Physiol. Vol. 120, 1999 or so cellular molecular species represent the lowest level. Interactions between these molecules construct a complex network perhaps best described as analogous to a very large, badly woven fishing net. Like all networks, the connections form molecular collectives that generate new emergent properties. Cellular behavior is emergent behavior, as are cytoskeletal dynamics, cell division, cell growth, and cell development. Emergent properties are not predictable from the most detailed analysis of the constituents, because by definition it is the interactions between the molecules that are critical. Emergent properties can be observed, but understanding is another matter. Undoubtedly, the spatial calcium signal is a primary emergent characteristic in its control of metabolism and gene expression. However, every cell is unique in certain molecular characteristics, so the internal calcium signal in each cell is represented by a unique topology (Trewavas and Malho, 1997). We may never fully understand the complex information implicit in the topological calcium signal that is generated. But, then again, 20 years ago we understood nothing at all of calcium in plant cells and now knowledge increases on a daily basis. Life is complex, and le calcium, c est la vie suggests that calcium signaling is as infinitely complex as life itself. ACKNOWLEDGMENTS Many people have contributed to this discussion. Errors and omissions are my own responsibility. Received January 15, 1999; accepted February 17, LITERATURE CITED Abel T, Martin KC, Batsch D, Kandel ER (1998) Memory suppressor genes: inhibitory constraints on the storage of long-term memory. Science 279: Addicott FT (1982) Abscission. University of California Press. Berkeley, CA Alexandre J, Lassales JP, Kado RT (1990) Opening of calcium channels in isolated red beet root vacuole membrane by inositol 1,4,5 trisphosphate. Nature 343: Allen GJ, Muir SR, Sanders D (1995) Release of calcium from individual plant vacuoles by both InsP 3 and cyclic ADP-ribose. Science 268: Braam J, Davis RW (1990) Rain, wind and touch induced expression of calmodulin and calmodulin related genes in Arabidopsis thaliana. Cell 63: Collinge M, Trewavas AJ (1989) Location of calmodulin in the pea plasma membrane. J Biol Chem 364: Darwin C (1880) The Power of Movement in Plants. John Murray, London Drew MC, Saker LR, Ashley TW (1973) Nutrient supply and the growth of the seminal root system in barley. J Exp Bot 24: Drobak BK, Watkins PAC (1994) IP 3 production in plant cells: stimulation by the venom peptides mellitin and mastaporan. Biochem Biophys Res Commun 205: Franklin-Tong VE, Drobak BK, Allan AC, Watkins PAC, Trewavas AJ (1996) Growth of pollen tubes of Papaver rheoas is regulated by a slow-moving calcium wave propagated by inositol triphosphosphate. Plant Cell 8: Gilroy S, Fricker M, Read ND, Trewavas AJ (1991) Role of calcium in signal transduction of Commelina guard cells. Plant Cell 3: Gilroy S, Read ND, Trewavas AJ (1990) Elevation of stomatal cytosol calcium by photolysis of loaded caged probes initiates stomatal closure. Nature 346: Grabov A, Blatt MR (1998) Membrane voltage initiates calcium waves and potentiates calcium increase with abscisic acid in guard cells. Proc Natl Acad Sci USA 95: Grotwiel MS, Beck CDO, Wu KH, Zhu X, David RL (1998) Integrin-mediated short term memory in Drosophila. Nature 391: Hirayama T, Ohto C, Mizoguchi T, Shinozaki K (1995) A gene encoding a phosphatidylinositol-specific phospholipase C is induced by dehydration and salt stress in Arabidopsis thaliana. Proc Natl Acad Sci USA 92: Hodgkin AL, Keynes RD (1957) Movements of labeled calcium in giant squid axons. J Physiol 138: Karban R, Baldwin IT (1997) Induced Responses to Herbivory. The University of Chicago Press, Chicago, IL Knight H, Trewavas AJ, Knight MR (1996) Cold calcium signalling in Arabidopsis involves two cellular pools and a change in calcium signature after acclimation. Plant Cell 8: Lewis JE, Kristan WB (1998) A neuronal network for computing population vectors in the leech. Nature 391: Malho R, Moutinho A, Van der Luit A, Trewavas AJ (1998) Spatial characteristics of calcium signalling: the calcium wave as a basic unit in plant cell calcium signalling. Phil Trans R Soc Lond B 3: Malho R, Trewavas AJ (1996) Localized apical increases of cytosolic free calcium control pollen tube orientation. Plant Cell 8: Marchant JS, Taylor CW (1997) Cooperative activation of IP 3 receptors by sequential binding of IP 3 and calcium safeguards against spontaneous activity. Curr Biol 7: Pennell R (1998) Cell walls: structures and signals. Curr Opin Plant Biol 1: Pierson ES, Li YQ, Zhang HQ, Willemse MTM, Linskens HF, Cresti M (1995) Pulsatory growth of pollen tubes: investigation of a possible relationship with the periodic distribution of wall components. Acta Bot Neerl 44: Sachs JV (1881) Lectures on the Physiology of Plants. Clarendon Press, Oxford, UK Taylor AR, Manison NFH, Fernandez C, Wood J, Brownlee C (1996) Spatial organisation of calcium signalling involved in volume control of the Fucus rhizoid. Plant Cell 8: Trewavas AJ (1992) Growth substances in context: a decade of sensitivity. Biochem Soc Trans 20: Trewavas AJ, Malho R (1997) Signal perception and transduction, the origin of the phenotype. Plant Cell 9: Urao T, Katagiri T, Mizoguchi T, Yamaguchi-Shinozaki K, Hayashida N, Shinozaki K (1994) Two genes that encode calciumdependent protein kinases are induced by drought and high salt stresses in Arabidopsis thaliana. Mol Gen Genet 244: Verdus MC, Thellier M, Ripoli C (1997) Storage of environmental signals in flax: their morphogenetic effect as enabled by a transient depletion of calcium. Plant J 12: Wu Y, Kuzma J, Marechal E, Graeff R, Lee HC, Foster R, Chua NM (1997) Abscisic acid signalling through cyclic ADP-ribose in plants. Science 278:
Keystone Review Practice Test Module A Cells and Cell Processes. 1. Which characteristic is shared by all prokaryotes and eukaryotes?
 Keystone Review Practice Test Module A Cells and Cell Processes 1. Which characteristic is shared by all prokaryotes and eukaryotes? a. Ability to store hereditary information b. Use of organelles to control
Keystone Review Practice Test Module A Cells and Cell Processes 1. Which characteristic is shared by all prokaryotes and eukaryotes? a. Ability to store hereditary information b. Use of organelles to control
Cell and Membrane Practice. A. chromosome B. gene C. mitochondrion D. vacuole
 Name: ate: 1. Which structure is outside the nucleus of a cell and contains N?. chromosome. gene. mitochondrion. vacuole 2. potato core was placed in a beaker of water as shown in the figure below. Which
Name: ate: 1. Which structure is outside the nucleus of a cell and contains N?. chromosome. gene. mitochondrion. vacuole 2. potato core was placed in a beaker of water as shown in the figure below. Which
Actions of Hormones on Target Cells Page 1. Actions of Hormones on Target Cells Page 2. Goals/ What You Need to Know Goals What You Need to Know
 Actions of Hormones on Target Cells Graphics are used with permission of: Pearson Education Inc., publishing as Benjamin Cummings (http://www.aw-bc.com) Page 1. Actions of Hormones on Target Cells Hormones
Actions of Hormones on Target Cells Graphics are used with permission of: Pearson Education Inc., publishing as Benjamin Cummings (http://www.aw-bc.com) Page 1. Actions of Hormones on Target Cells Hormones
Anatomy and Physiology Placement Exam 2 Practice with Answers at End!
 Anatomy and Physiology Placement Exam 2 Practice with Answers at End! General Chemical Principles 1. bonds are characterized by the sharing of electrons between the participating atoms. a. hydrogen b.
Anatomy and Physiology Placement Exam 2 Practice with Answers at End! General Chemical Principles 1. bonds are characterized by the sharing of electrons between the participating atoms. a. hydrogen b.
Chapter-21b: Hormones and Receptors
 1 hapter-21b: Hormones and Receptors Hormone classes Hormones are classified according to the distance over which they act. 1. Autocrine hormones --- act on the same cell that released them. Interleukin-2
1 hapter-21b: Hormones and Receptors Hormone classes Hormones are classified according to the distance over which they act. 1. Autocrine hormones --- act on the same cell that released them. Interleukin-2
1.1.2. thebiotutor. AS Biology OCR. Unit F211: Cells, Exchange & Transport. Module 1.2 Cell Membranes. Notes & Questions.
 thebiotutor AS Biology OCR Unit F211: Cells, Exchange & Transport Module 1.2 Cell Membranes Notes & Questions Andy Todd 1 Outline the roles of membranes within cells and at the surface of cells. The main
thebiotutor AS Biology OCR Unit F211: Cells, Exchange & Transport Module 1.2 Cell Membranes Notes & Questions Andy Todd 1 Outline the roles of membranes within cells and at the surface of cells. The main
Biological cell membranes
 Unit 14: Cell biology. 14 2 Biological cell membranes The cell surface membrane surrounds the cell and acts as a barrier between the cell s contents and the environment. The cell membrane has multiple
Unit 14: Cell biology. 14 2 Biological cell membranes The cell surface membrane surrounds the cell and acts as a barrier between the cell s contents and the environment. The cell membrane has multiple
PART I: Neurons and the Nerve Impulse
 PART I: Neurons and the Nerve Impulse Identify each of the labeled structures of the neuron below. A. B. C. D. E. F. G. Identify each of the labeled structures of the neuron below. A. dendrites B. nucleus
PART I: Neurons and the Nerve Impulse Identify each of the labeled structures of the neuron below. A. B. C. D. E. F. G. Identify each of the labeled structures of the neuron below. A. dendrites B. nucleus
ANIMATED NEUROSCIENCE
 ANIMATED NEUROSCIENCE and the Action of Nicotine, Cocaine, and Marijuana in the Brain Te a c h e r s G u i d e Films for the Humanities & Sciences Background Information This program, made entirely of
ANIMATED NEUROSCIENCE and the Action of Nicotine, Cocaine, and Marijuana in the Brain Te a c h e r s G u i d e Films for the Humanities & Sciences Background Information This program, made entirely of
Diabetes and Insulin Signaling
 Diabetes and Insulin Signaling NATIONAL CENTER FOR CASE STUDY TEACHING IN SCIENCE by Kristy J. Wilson School of Mathematics and Sciences Marian University, Indianapolis, IN Part I Research Orientation
Diabetes and Insulin Signaling NATIONAL CENTER FOR CASE STUDY TEACHING IN SCIENCE by Kristy J. Wilson School of Mathematics and Sciences Marian University, Indianapolis, IN Part I Research Orientation
Lecture 8. Protein Trafficking/Targeting. Protein targeting is necessary for proteins that are destined to work outside the cytoplasm.
 Protein Trafficking/Targeting (8.1) Lecture 8 Protein Trafficking/Targeting Protein targeting is necessary for proteins that are destined to work outside the cytoplasm. Protein targeting is more complex
Protein Trafficking/Targeting (8.1) Lecture 8 Protein Trafficking/Targeting Protein targeting is necessary for proteins that are destined to work outside the cytoplasm. Protein targeting is more complex
7 Answers to end-of-chapter questions
 7 Answers to end-of-chapter questions Multiple choice questions 1 B 2 B 3 A 4 B 5 A 6 D 7 C 8 C 9 B 10 B Structured questions 11 a i Maintenance of a constant internal environment within set limits i Concentration
7 Answers to end-of-chapter questions Multiple choice questions 1 B 2 B 3 A 4 B 5 A 6 D 7 C 8 C 9 B 10 B Structured questions 11 a i Maintenance of a constant internal environment within set limits i Concentration
Cellular Calcium Dynamics. Jussi Koivumäki, Glenn Lines & Joakim Sundnes
 Cellular Calcium Dynamics Jussi Koivumäki, Glenn Lines & Joakim Sundnes Cellular calcium dynamics A real cardiomyocyte is obviously not an empty cylinder, where Ca 2+ just diffuses freely......instead
Cellular Calcium Dynamics Jussi Koivumäki, Glenn Lines & Joakim Sundnes Cellular calcium dynamics A real cardiomyocyte is obviously not an empty cylinder, where Ca 2+ just diffuses freely......instead
Plant Growth & Development. Growth Stages. Differences in the Developmental Mechanisms of Plants and Animals. Development
 Plant Growth & Development Plant body is unable to move. To survive and grow, plants must be able to alter its growth, development and physiology. Plants are able to produce complex, yet variable forms
Plant Growth & Development Plant body is unable to move. To survive and grow, plants must be able to alter its growth, development and physiology. Plants are able to produce complex, yet variable forms
Section 7-3 Cell Boundaries
 Note: For the past several years, I ve been puzzling how to integrate new discoveries on the nature of water movement through cell membranes into Chapter 7. The Section below is a draft of my first efforts
Note: For the past several years, I ve been puzzling how to integrate new discoveries on the nature of water movement through cell membranes into Chapter 7. The Section below is a draft of my first efforts
Six major functions of membrane proteins: Transport Enzymatic activity
 CH 7 Membranes Cellular Membranes Phospholipids are the most abundant lipid in the plasma membrane. Phospholipids are amphipathic molecules, containing hydrophobic and hydrophilic regions. The fluid mosaic
CH 7 Membranes Cellular Membranes Phospholipids are the most abundant lipid in the plasma membrane. Phospholipids are amphipathic molecules, containing hydrophobic and hydrophilic regions. The fluid mosaic
Functional Biology of Plants
 Brochure More information from http://www.researchandmarkets.com/reports/2252012/ Functional Biology of Plants Description: Functional Biology of Plants provides students and researchers with a clearly
Brochure More information from http://www.researchandmarkets.com/reports/2252012/ Functional Biology of Plants Description: Functional Biology of Plants provides students and researchers with a clearly
Chapter 36: Resource Acquisition & Transport in Vascular Plants
 Chapter 36: Resource Acquisition & Transport in Vascular Plants 1. Overview of Transport in Plants 2. Transport of Water & Minerals 3. Transport of Sugars 1. Overview of Transport in Plants H 2 O CO 2
Chapter 36: Resource Acquisition & Transport in Vascular Plants 1. Overview of Transport in Plants 2. Transport of Water & Minerals 3. Transport of Sugars 1. Overview of Transport in Plants H 2 O CO 2
Cell Structure & Function!
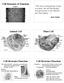 Cell Structure & Function! Chapter 3! The most exciting phrase to hear in science, the one that heralds new discoveries, is not 'Eureka!' but 'That's funny.! -- Isaac Asimov Animal Cell Plant Cell Cell
Cell Structure & Function! Chapter 3! The most exciting phrase to hear in science, the one that heralds new discoveries, is not 'Eureka!' but 'That's funny.! -- Isaac Asimov Animal Cell Plant Cell Cell
Chapter 39: Plant Responses to Internal and External Signals
 Name Period Concept 39.1 Signal transduction pathways link signal reception to response This concept brings together the general ideas on cell communication from Chapter 11 with specific examples of signal
Name Period Concept 39.1 Signal transduction pathways link signal reception to response This concept brings together the general ideas on cell communication from Chapter 11 with specific examples of signal
CHAPTER 5 SIGNALLING IN NEURONS
 5.1. SYNAPTIC TRANSMISSION CHAPTER 5 SIGNALLING IN NEURONS One of the main functions of neurons is to communicate with other neurons. An individual neuron may receive information from many different sources.
5.1. SYNAPTIC TRANSMISSION CHAPTER 5 SIGNALLING IN NEURONS One of the main functions of neurons is to communicate with other neurons. An individual neuron may receive information from many different sources.
Ions cannot cross membranes. Ions move through pores
 Ions cannot cross membranes Membranes are lipid bilayers Nonpolar tails Polar head Fig 3-1 Because of the charged nature of ions, they cannot cross a lipid bilayer. The ion and its cloud of polarized water
Ions cannot cross membranes Membranes are lipid bilayers Nonpolar tails Polar head Fig 3-1 Because of the charged nature of ions, they cannot cross a lipid bilayer. The ion and its cloud of polarized water
2. Fill in the blank. The of a cell is like a leader, directing and telling the different parts of the cell what to do.
 1. Plant and animal cells have some similarities as well as differences. What is one thing that plant and animal cells have in common? A. cell wall B. chlorophyll C. nucleus D. chloroplasts 2. Fill in
1. Plant and animal cells have some similarities as well as differences. What is one thing that plant and animal cells have in common? A. cell wall B. chlorophyll C. nucleus D. chloroplasts 2. Fill in
Cell Structure and Function. Eukaryotic Cell: Neuron
 Cell Structure and Function Eukaryotic Cell: Neuron Cell Structure and Function Eukaryotic Cells: Blood Cells Cell Structure and Function Prokaryotic Cells: Bacteria Cell Structure and Function All living
Cell Structure and Function Eukaryotic Cell: Neuron Cell Structure and Function Eukaryotic Cells: Blood Cells Cell Structure and Function Prokaryotic Cells: Bacteria Cell Structure and Function All living
BSC 2010 - Exam I Lectures and Text Pages. The Plasma Membrane Structure and Function. Phospholipids. I. Intro to Biology (2-29) II.
 BSC 2010 - Exam I Lectures and Text Pages I. Intro to Biology (2-29) II. Chemistry of Life Chemistry review (30-46) Water (47-57) Carbon (58-67) Macromolecules (68-91) III. Cells and Membranes Cell structure
BSC 2010 - Exam I Lectures and Text Pages I. Intro to Biology (2-29) II. Chemistry of Life Chemistry review (30-46) Water (47-57) Carbon (58-67) Macromolecules (68-91) III. Cells and Membranes Cell structure
Mechanisms of Hormonal Action Bryant Miles
 Mechanisms of ormonal Action Bryant Miles Multicellular organisms need to coordinate metabolic activities. Complex signaling systems have evolved using chemicals called hormones to regulate cellular activities.
Mechanisms of ormonal Action Bryant Miles Multicellular organisms need to coordinate metabolic activities. Complex signaling systems have evolved using chemicals called hormones to regulate cellular activities.
Review of the Cell and Its Organelles
 Biology Learning Centre Review of the Cell and Its Organelles Tips for most effective learning of this material: Memorize the names and structures over several days. This will help you retain what you
Biology Learning Centre Review of the Cell and Its Organelles Tips for most effective learning of this material: Memorize the names and structures over several days. This will help you retain what you
The Cell Teaching Notes and Answer Keys
 The Cell Teaching Notes and Answer Keys Subject area: Science / Biology Topic focus: The Cell: components, types of cells, organelles, levels of organization Learning Aims: describe similarities and differences
The Cell Teaching Notes and Answer Keys Subject area: Science / Biology Topic focus: The Cell: components, types of cells, organelles, levels of organization Learning Aims: describe similarities and differences
7. A selectively permeable membrane only allows certain molecules to pass through.
 CHAPTER 2 GETTING IN & OUT OF CELLS PASSIVE TRANSPORT Cell membranes help organisms maintain homeostasis by controlling what substances may enter or leave cells. Some substances can cross the cell membrane
CHAPTER 2 GETTING IN & OUT OF CELLS PASSIVE TRANSPORT Cell membranes help organisms maintain homeostasis by controlling what substances may enter or leave cells. Some substances can cross the cell membrane
Parts of the Nerve Cell and Their Functions
 Parts of the Nerve Cell and Their Functions Silvia Helena Cardoso, PhD [ 1. Cell body] [2. Neuronal membrane] [3. Dendrites] [4. Axon] [5. Nerve ending] 1. Cell body The cell body (soma) is the factory
Parts of the Nerve Cell and Their Functions Silvia Helena Cardoso, PhD [ 1. Cell body] [2. Neuronal membrane] [3. Dendrites] [4. Axon] [5. Nerve ending] 1. Cell body The cell body (soma) is the factory
IIn our high tech world, one of the hottest areas of development
 Topic 1.1 Why are cells important? Key Concepts Studying cells helps us understand how organisms function. Cellular organelles work together to carry out life functions. Cellular processes enable organisms
Topic 1.1 Why are cells important? Key Concepts Studying cells helps us understand how organisms function. Cellular organelles work together to carry out life functions. Cellular processes enable organisms
4. Biology of the Cell
 4. Biology of the Cell Our primary focus in this chapter will be the plasma membrane and movement of materials across the plasma membrane. You should already be familiar with the basic structures and roles
4. Biology of the Cell Our primary focus in this chapter will be the plasma membrane and movement of materials across the plasma membrane. You should already be familiar with the basic structures and roles
Electron Transport Generates a Proton Gradient Across the Membrane
 Electron Transport Generates a Proton Gradient Across the Membrane Each of respiratory enzyme complexes couples the energy released by electron transfer across it to an uptake of protons from water in
Electron Transport Generates a Proton Gradient Across the Membrane Each of respiratory enzyme complexes couples the energy released by electron transfer across it to an uptake of protons from water in
CHAPTER XV PDL 101 HUMAN ANATOMY & PHYSIOLOGY. Ms. K. GOWRI. M.Pharm., Lecturer.
 CHAPTER XV PDL 101 HUMAN ANATOMY & PHYSIOLOGY Ms. K. GOWRI. M.Pharm., Lecturer. Types of Muscle Tissue Classified by location, appearance, and by the type of nervous system control or innervation. Skeletal
CHAPTER XV PDL 101 HUMAN ANATOMY & PHYSIOLOGY Ms. K. GOWRI. M.Pharm., Lecturer. Types of Muscle Tissue Classified by location, appearance, and by the type of nervous system control or innervation. Skeletal
Questions on The Nervous System and Gas Exchange
 Name: Questions on The Nervous System and Gas Exchange Directions: The following questions are taken from previous IB Final Papers on Topics 6.4 (Gas Exchange) and 6.5 (Nerves, hormones and homeostasis).
Name: Questions on The Nervous System and Gas Exchange Directions: The following questions are taken from previous IB Final Papers on Topics 6.4 (Gas Exchange) and 6.5 (Nerves, hormones and homeostasis).
MCAS Biology. Review Packet
 MCAS Biology Review Packet 1 Name Class Date 1. Define organic. THE CHEMISTRY OF LIFE 2. All living things are made up of 6 essential elements: SPONCH. Name the six elements of life. S N P C O H 3. Elements
MCAS Biology Review Packet 1 Name Class Date 1. Define organic. THE CHEMISTRY OF LIFE 2. All living things are made up of 6 essential elements: SPONCH. Name the six elements of life. S N P C O H 3. Elements
Name Date Period. 2. When a molecule of double-stranded DNA undergoes replication, it results in
 DNA, RNA, Protein Synthesis Keystone 1. During the process shown above, the two strands of one DNA molecule are unwound. Then, DNA polymerases add complementary nucleotides to each strand which results
DNA, RNA, Protein Synthesis Keystone 1. During the process shown above, the two strands of one DNA molecule are unwound. Then, DNA polymerases add complementary nucleotides to each strand which results
Cell Unit Practice Test #1
 ell Unit Practice Test #1 Name: ate: 1. Which organelle is primarily concerned with the conversion of potential energy of organic compounds into suitable form for immediate use by the cell?. mitochondria.
ell Unit Practice Test #1 Name: ate: 1. Which organelle is primarily concerned with the conversion of potential energy of organic compounds into suitable form for immediate use by the cell?. mitochondria.
Student name ID # 2. (4 pts) What is the terminal electron acceptor in respiration? In photosynthesis? O2, NADP+
 1. Membrane transport. A. (4 pts) What ion couples primary and secondary active transport in animal cells? What ion serves the same function in plant cells? Na+, H+ 2. (4 pts) What is the terminal electron
1. Membrane transport. A. (4 pts) What ion couples primary and secondary active transport in animal cells? What ion serves the same function in plant cells? Na+, H+ 2. (4 pts) What is the terminal electron
Neurophysiology. 2.1 Equilibrium Potential
 2 Neurophysiology 2.1 Equilibrium Potential An understanding of the concepts of electrical and chemical forces that act on ions, electrochemical equilibrium, and equilibrium potential is a powerful tool
2 Neurophysiology 2.1 Equilibrium Potential An understanding of the concepts of electrical and chemical forces that act on ions, electrochemical equilibrium, and equilibrium potential is a powerful tool
7.2 Cells: A Look Inside
 CHAPTER 7 CELL STRUCTURE AND FUNCTION 7.2 Cells: A Look Inside Imagine a factory that makes thousands of cookies a day. Ingredients come into the factory, get mixed and baked, then the cookies are packaged.
CHAPTER 7 CELL STRUCTURE AND FUNCTION 7.2 Cells: A Look Inside Imagine a factory that makes thousands of cookies a day. Ingredients come into the factory, get mixed and baked, then the cookies are packaged.
COMPARISON OF PLANT AND ANIMAL CELLS SIMILARITIES IN PLANT & ANIMAL CELLS
 COMPARISON OF PLANT AND ANIMAL CELLS Cells vary widely in structure and function, even within the same organism. The human body, for example, has more than 200 different types of cells, each with a specialized
COMPARISON OF PLANT AND ANIMAL CELLS Cells vary widely in structure and function, even within the same organism. The human body, for example, has more than 200 different types of cells, each with a specialized
Bi 360: Midterm Review
 Bi 360: Midterm Review Basic Neurobiology 1) Many axons are surrounded by a fatty insulating sheath called myelin, which is interrupted at regular intervals at the Nodes of Ranvier, where the action potential
Bi 360: Midterm Review Basic Neurobiology 1) Many axons are surrounded by a fatty insulating sheath called myelin, which is interrupted at regular intervals at the Nodes of Ranvier, where the action potential
The Making of the Fittest: Evolving Switches, Evolving Bodies
 OVERVIEW MODELING THE REGULATORY SWITCHES OF THE PITX1 GENE IN STICKLEBACK FISH This hands-on activity supports the short film, The Making of the Fittest:, and aims to help students understand eukaryotic
OVERVIEW MODELING THE REGULATORY SWITCHES OF THE PITX1 GENE IN STICKLEBACK FISH This hands-on activity supports the short film, The Making of the Fittest:, and aims to help students understand eukaryotic
RAD 223. Radiography physiology. Lecture Notes. First lecture: Cell and Tissue
 RAD 223 Radiography physiology Lecture Notes First lecture: Cell and Tissue Physiology: the word physiology derived from a Greek word for study of nature. It is the study of how the body and its part work
RAD 223 Radiography physiology Lecture Notes First lecture: Cell and Tissue Physiology: the word physiology derived from a Greek word for study of nature. It is the study of how the body and its part work
Hormones & Chemical Signaling
 Hormones & Chemical Signaling Part 2 modulation of signal pathways and hormone classification & function How are these pathways controlled? Receptors are proteins! Subject to Specificity of binding Competition
Hormones & Chemical Signaling Part 2 modulation of signal pathways and hormone classification & function How are these pathways controlled? Receptors are proteins! Subject to Specificity of binding Competition
B2 1 Cells, Tissues and Organs
 B2 Cells, Tissues and Organs 5 minutes 5 marks Page of 7 Q. The diagram shows a bacterium. On the drawing, name the structures labelled A, B, C and D. (Total 4 marks) Q2. (a) The diagrams show cells containing
B2 Cells, Tissues and Organs 5 minutes 5 marks Page of 7 Q. The diagram shows a bacterium. On the drawing, name the structures labelled A, B, C and D. (Total 4 marks) Q2. (a) The diagrams show cells containing
the plant & animal cell
 6.1 Basic unit of life Biology Biology Structure & functions of 06 the plant & animal cell In 1665, Robert Hooke observed a section of a cork using a microscope prepared by him. He discovered a structure
6.1 Basic unit of life Biology Biology Structure & functions of 06 the plant & animal cell In 1665, Robert Hooke observed a section of a cork using a microscope prepared by him. He discovered a structure
Chapter 3. Cellular Structure and Function Worksheets. 39 www.ck12.org
 Chapter 3 Cellular Structure and Function Worksheets (Opening image copyright by Sebastian Kaulitzki, 2010. Used under license from Shutterstock.com.) Lesson 3.1: Introduction to Cells Lesson 3.2: Cell
Chapter 3 Cellular Structure and Function Worksheets (Opening image copyright by Sebastian Kaulitzki, 2010. Used under license from Shutterstock.com.) Lesson 3.1: Introduction to Cells Lesson 3.2: Cell
CELLS: PLANT CELLS 20 FEBRUARY 2013
 CELLS: PLANT CELLS 20 FEBRUARY 2013 Lesson Description In this lesson we will discuss the following: The Cell Theory Terminology Parts of Plant Cells: Organelles Difference between plant and animal cells
CELLS: PLANT CELLS 20 FEBRUARY 2013 Lesson Description In this lesson we will discuss the following: The Cell Theory Terminology Parts of Plant Cells: Organelles Difference between plant and animal cells
CELL MEMBRANES, TRANSPORT, and COMMUNICATION. Teacher Packet
 AP * BIOLOGY CELL MEMBRANES, TRANSPORT, and COMMUNICATION Teacher Packet AP* is a trademark of the College Entrance Examination Board. The College Entrance Examination Board was not involved in the production
AP * BIOLOGY CELL MEMBRANES, TRANSPORT, and COMMUNICATION Teacher Packet AP* is a trademark of the College Entrance Examination Board. The College Entrance Examination Board was not involved in the production
Muscles How muscles contract - The Sliding Filament Theory
 Muscles How muscles contract - The Sliding Filament Theory A muscle contains many muscle fibers A muscle fiber is a series of fused cells Each fiber contains a bundle of 4-20 myofibrils Myofibrils are
Muscles How muscles contract - The Sliding Filament Theory A muscle contains many muscle fibers A muscle fiber is a series of fused cells Each fiber contains a bundle of 4-20 myofibrils Myofibrils are
Chapter 5 Organelles. Lesson Objectives List the organelles of the cell and their functions. Distinguish between plant and animal cells.
 Chapter 5 Organelles Lesson Objectives List the organelles of the cell and their functions. Distinguish between plant and animal cells. Check Your Understanding What is a cell? How do we visualize cells?
Chapter 5 Organelles Lesson Objectives List the organelles of the cell and their functions. Distinguish between plant and animal cells. Check Your Understanding What is a cell? How do we visualize cells?
Plant Responses to Environmental Cues Tropisms, Photoperiodism, and Plant Hormones
 Plant Responses to Environmental Cues Tropisms, Photoperiodism, and Plant Hormones Plant Responses to Environmental Cues Phototropism - plant growth response to light shoots bend toward light - positive
Plant Responses to Environmental Cues Tropisms, Photoperiodism, and Plant Hormones Plant Responses to Environmental Cues Phototropism - plant growth response to light shoots bend toward light - positive
1. f. Students know usable energy is captured from sunlight by chloroplasts and is stored through the synthesis of sugar from carbon dioxide.
 1. The fundamental life processes of plants and animals depend on a variety of chemical reactions that occur in specialized areas of the organism s cells. As a basis for understanding this concept: 1.
1. The fundamental life processes of plants and animals depend on a variety of chemical reactions that occur in specialized areas of the organism s cells. As a basis for understanding this concept: 1.
BIOL 305L Laboratory Two
 Please print Full name clearly: Introduction BIOL 305L Laboratory Two Osmosis, because it is different in plants! Osmosis is the movement of solvent molecules through a selectively permeable membrane into
Please print Full name clearly: Introduction BIOL 305L Laboratory Two Osmosis, because it is different in plants! Osmosis is the movement of solvent molecules through a selectively permeable membrane into
Resting membrane potential ~ -70mV - Membrane is polarized
 Resting membrane potential ~ -70mV - Membrane is polarized (ie) Electrical charge on the outside of the membrane is positive while the electrical charge on the inside of the membrane is negative Changes
Resting membrane potential ~ -70mV - Membrane is polarized (ie) Electrical charge on the outside of the membrane is positive while the electrical charge on the inside of the membrane is negative Changes
An Overview of Cells and Cell Research
 An Overview of Cells and Cell Research 1 An Overview of Cells and Cell Research Chapter Outline Model Species and Cell types Cell components Tools of Cell Biology Model Species E. Coli: simplest organism
An Overview of Cells and Cell Research 1 An Overview of Cells and Cell Research Chapter Outline Model Species and Cell types Cell components Tools of Cell Biology Model Species E. Coli: simplest organism
Date: Student Name: Teacher Name: Jared George. Score: 1) A cell with 1% solute concentration is placed in a beaker with a 5% solute concentration.
 Biology Keystone (PA Core) Quiz Homeostasis and Transport - (BIO.A.4.1.1 ) Plasma Membrane, (BIO.A.4.1.2 ) Transport Mechanisms, (BIO.A.4.1.3 ) Transport Facilitation Student Name: Teacher Name: Jared
Biology Keystone (PA Core) Quiz Homeostasis and Transport - (BIO.A.4.1.1 ) Plasma Membrane, (BIO.A.4.1.2 ) Transport Mechanisms, (BIO.A.4.1.3 ) Transport Facilitation Student Name: Teacher Name: Jared
Investigating cells. Cells are the basic units of living things (this means that all living things are made up of one or more cells).
 SG Biology Summary notes Investigating cells Sub-topic a: Investigating living cells Cells are the basic units of living things (this means that all living things are made up of one or more cells). Cells
SG Biology Summary notes Investigating cells Sub-topic a: Investigating living cells Cells are the basic units of living things (this means that all living things are made up of one or more cells). Cells
The Lipid Bilayer Is a Two-Dimensional Fluid
 The Lipid Bilayer Is a Two-Dimensional Fluid The aqueous environment inside and outside a cell prevents membrane lipids from escaping from bilayer, but nothing stops these molecules from moving about and
The Lipid Bilayer Is a Two-Dimensional Fluid The aqueous environment inside and outside a cell prevents membrane lipids from escaping from bilayer, but nothing stops these molecules from moving about and
tissues are made of cells that work together, organs are )
 Study Guide Cells Unit Test Matching. Write the letter of the correct response on the line. You may use the responses more than once. A. proteins B. simple carbohydrates C. complex carbohydrates D. lipids
Study Guide Cells Unit Test Matching. Write the letter of the correct response on the line. You may use the responses more than once. A. proteins B. simple carbohydrates C. complex carbohydrates D. lipids
CORAL REEF ECOLOGY. Objectives Students will learn that coral is an animal. Students will learn the differences between plant and animal cells.
 CORAL REEF ECOLOGY Concepts Scientific method Difference between plants and animals cells HCPS III Benchmarks SC 4.1.2 SC 4.4.1 Duration 2 hours Source Material PRISM Vocabulary Cell Cell Membrane Cytosol
CORAL REEF ECOLOGY Concepts Scientific method Difference between plants and animals cells HCPS III Benchmarks SC 4.1.2 SC 4.4.1 Duration 2 hours Source Material PRISM Vocabulary Cell Cell Membrane Cytosol
Cellular Structure and Function
 Chapter Test A CHAPTER 7 Cellular Structure and Function Part A: Multiple Choice In the space at the left, write the letter of the term or phrase that best answers each question. 1. Which defines a cell?
Chapter Test A CHAPTER 7 Cellular Structure and Function Part A: Multiple Choice In the space at the left, write the letter of the term or phrase that best answers each question. 1. Which defines a cell?
The Cell: Organelle Diagrams
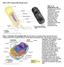 The Cell: Organelle Diagrams Fig 7-4. A prokaryotic cell. Lacking a true nucleus and the other membrane-enclosed organelles of the eukaryotic cell, the prokaryotic cell is much simpler in structure. Only
The Cell: Organelle Diagrams Fig 7-4. A prokaryotic cell. Lacking a true nucleus and the other membrane-enclosed organelles of the eukaryotic cell, the prokaryotic cell is much simpler in structure. Only
Photosynthesis and Cellular Respiration. Stored Energy
 Photosynthesis and Cellular Respiration Stored Energy What is Photosynthesis? plants convert the energy of sunlight into the energy in the chemical bonds of carbohydrates sugars and starches. SUMMARY EQUATION:
Photosynthesis and Cellular Respiration Stored Energy What is Photosynthesis? plants convert the energy of sunlight into the energy in the chemical bonds of carbohydrates sugars and starches. SUMMARY EQUATION:
Bacterial (Prokaryotic) Cell. Common features of all cells. Tour of the Cell. Eukaryotic Cell. Plasma Membrane defines inside from outside
 www.denniskunkel.com Tour of the Cell www.denniskunkel.com Today s Topics Properties of all cells Prokaryotes and Eukaryotes Functions of Major Cellular Organelles Information, Synthesis&Transport,, Vesicles
www.denniskunkel.com Tour of the Cell www.denniskunkel.com Today s Topics Properties of all cells Prokaryotes and Eukaryotes Functions of Major Cellular Organelles Information, Synthesis&Transport,, Vesicles
2007 7.013 Problem Set 1 KEY
 2007 7.013 Problem Set 1 KEY Due before 5 PM on FRIDAY, February 16, 2007. Turn answers in to the box outside of 68-120. PLEASE WRITE YOUR ANSWERS ON THIS PRINTOUT. 1. Where in a eukaryotic cell do you
2007 7.013 Problem Set 1 KEY Due before 5 PM on FRIDAY, February 16, 2007. Turn answers in to the box outside of 68-120. PLEASE WRITE YOUR ANSWERS ON THIS PRINTOUT. 1. Where in a eukaryotic cell do you
Plant and Animal Cells
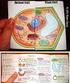 Plant and Animal Cells a. Explain that cells take in nutrients in order to grow, divide and to make needed materials. S7L2a b. Relate cell structures (cell membrane, nucleus, cytoplasm, chloroplasts, and
Plant and Animal Cells a. Explain that cells take in nutrients in order to grow, divide and to make needed materials. S7L2a b. Relate cell structures (cell membrane, nucleus, cytoplasm, chloroplasts, and
Compartmentalization of the Cell. Objectives. Recommended Reading. Professor Alfred Cuschieri. Department of Anatomy University of Malta
 Compartmentalization of the Cell Professor Alfred Cuschieri Department of Anatomy University of Malta Objectives By the end of this session the student should be able to: 1. Identify the different organelles
Compartmentalization of the Cell Professor Alfred Cuschieri Department of Anatomy University of Malta Objectives By the end of this session the student should be able to: 1. Identify the different organelles
The microscope is an important tool.
 KEY CONCEPT Microscopes allow us to see inside the cell. BEFORE, you learned Some organisms are unicellular and some are multicellular A microscope is necessary to study most cells The cell theory describes
KEY CONCEPT Microscopes allow us to see inside the cell. BEFORE, you learned Some organisms are unicellular and some are multicellular A microscope is necessary to study most cells The cell theory describes
Homeostasis and Transport Module A Anchor 4
 Homeostasis and Transport Module A Anchor 4 Key Concepts: - Buffers play an important role in maintaining homeostasis in organisms. - To maintain homeostasis, unicellular organisms grow, respond to the
Homeostasis and Transport Module A Anchor 4 Key Concepts: - Buffers play an important role in maintaining homeostasis in organisms. - To maintain homeostasis, unicellular organisms grow, respond to the
Muscle Tissue. Muscle Physiology. Skeletal Muscle. Types of Muscle. Skeletal Muscle Organization. Myofibril Structure
 Muscle Tissue Muscle Physiology Chapter 12 Specially designed to contract Generates mechanical force Functions locomotion and external movements internal movement (circulation, digestion) heat generation
Muscle Tissue Muscle Physiology Chapter 12 Specially designed to contract Generates mechanical force Functions locomotion and external movements internal movement (circulation, digestion) heat generation
pathway that involves taking in heat from the environment at each step. C.
 Study Island Cell Energy Keystone Review 1. Cells obtain energy by either capturing light energy through photosynthesis or by breaking down carbohydrates through cellular respiration. In both photosynthesis
Study Island Cell Energy Keystone Review 1. Cells obtain energy by either capturing light energy through photosynthesis or by breaking down carbohydrates through cellular respiration. In both photosynthesis
Which of the following can be determined based on this model? The atmosphere is the only reservoir on Earth that can store carbon in any form. A.
 Earth s Cycles 1. Models are often used to explain scientific knowledge or experimental results. A model of the carbon cycle is shown below. Which of the following can be determined based on this model?
Earth s Cycles 1. Models are often used to explain scientific knowledge or experimental results. A model of the carbon cycle is shown below. Which of the following can be determined based on this model?
Partnerships Implementing Engineering Education Worcester Polytechnic Institute Worcester Public Schools Supported by: National Science Foundation
 Cells & Heredity: 6.C.2 Design a Cell Travel Brochure Grade Level 6 Sessions Seasonality Instructional Mode(s) Team Size WPS Benchmarks MA Frameworks Key Words 15-50 minutes N/A Individual N/A 06.SC.LS.05
Cells & Heredity: 6.C.2 Design a Cell Travel Brochure Grade Level 6 Sessions Seasonality Instructional Mode(s) Team Size WPS Benchmarks MA Frameworks Key Words 15-50 minutes N/A Individual N/A 06.SC.LS.05
Transport in Plants Notes AP Biology Mrs. Laux 3 levels of transport occur in plants: 1. Uptake of water and solutes by individual cells
 3 levels of transport occur in plants: 1. Uptake of water and solutes by individual cells -for photosynthesis and respiration -ex: absorption of H 2 O /minerals by root hairs 2. Short distance cell-to-cell
3 levels of transport occur in plants: 1. Uptake of water and solutes by individual cells -for photosynthesis and respiration -ex: absorption of H 2 O /minerals by root hairs 2. Short distance cell-to-cell
AP Biology Essential Knowledge Student Diagnostic
 AP Biology Essential Knowledge Student Diagnostic Background The Essential Knowledge statements provided in the AP Biology Curriculum Framework are scientific claims describing phenomenon occurring in
AP Biology Essential Knowledge Student Diagnostic Background The Essential Knowledge statements provided in the AP Biology Curriculum Framework are scientific claims describing phenomenon occurring in
Energy Flow in the Pond Teacher s Guide February 2011
 Energy Flow in the Pond Teacher s Guide February 2011 Grades: 6, 7 & 8 Time: 3 hours With the pond as a model, students explore how energy that originates from the sun keeps changing shape and form as
Energy Flow in the Pond Teacher s Guide February 2011 Grades: 6, 7 & 8 Time: 3 hours With the pond as a model, students explore how energy that originates from the sun keeps changing shape and form as
Unit 5 Photosynthesis and Cellular Respiration
 Unit 5 Photosynthesis and Cellular Respiration Advanced Concepts What is the abbreviated name of this molecule? What is its purpose? What are the three parts of this molecule? Label each part with the
Unit 5 Photosynthesis and Cellular Respiration Advanced Concepts What is the abbreviated name of this molecule? What is its purpose? What are the three parts of this molecule? Label each part with the
Journal of Virginia Science Education. Exposing Student Misconceptions about Cellular Structure: A Curriculum Topic Study. Abbie Martin, M.Ed.
 Exposing Student Misconceptions about Cellular Structure: A Curriculum Topic Study Introduction Abbie Martin, M.Ed. Curriculum topic study allows teachers to investigate a specific concept in math or science
Exposing Student Misconceptions about Cellular Structure: A Curriculum Topic Study Introduction Abbie Martin, M.Ed. Curriculum topic study allows teachers to investigate a specific concept in math or science
Copyright 2000-2003 Mark Brandt, Ph.D. 54
 Pyruvate Oxidation Overview of pyruvate metabolism Pyruvate can be produced in a variety of ways. It is an end product of glycolysis, and can be derived from lactate taken up from the environment (or,
Pyruvate Oxidation Overview of pyruvate metabolism Pyruvate can be produced in a variety of ways. It is an end product of glycolysis, and can be derived from lactate taken up from the environment (or,
* Is chemical energy potential or kinetic energy? The position of what is storing energy?
 Biology 1406 Exam 2 - Metabolism Chs. 5, 6 and 7 energy - capacity to do work 5.10 kinetic energy - energy of motion : light, electrical, thermal, mechanical potential energy - energy of position or stored
Biology 1406 Exam 2 - Metabolism Chs. 5, 6 and 7 energy - capacity to do work 5.10 kinetic energy - energy of motion : light, electrical, thermal, mechanical potential energy - energy of position or stored
Lab 4: Osmosis and Diffusion
 Lab 4: Osmosis and Diffusion The plasma membrane enclosing every cell is the boundary that separates the cell from its external environment. It is not an impermeable barrier, but like all biological membranes,
Lab 4: Osmosis and Diffusion The plasma membrane enclosing every cell is the boundary that separates the cell from its external environment. It is not an impermeable barrier, but like all biological membranes,
Chapter 12. Temperature Regulation. Temperature Regulation. Heat Balance. An Overview of Heat Balance. Temperature Regulation. Temperature Regulation
 Chapter 12 Body core temperature regulation Critical for: Cellular structures Metabolic pathways Too high Protein structure of cells destroyed Too low Slowed metabolism Cardiac arrhythmias Homeothermic
Chapter 12 Body core temperature regulation Critical for: Cellular structures Metabolic pathways Too high Protein structure of cells destroyed Too low Slowed metabolism Cardiac arrhythmias Homeothermic
7.2 Cell Structure. Lesson Objectives. Lesson Summary. Cell Organization Eukaryotic cells contain a nucleus and many specialized structures.
 7.2 Cell Structure Lesson Objectives Describe the structure and function of the cell nucleus. Describe the role of vacuoles, lysosomes, and the cytoskeleton. Identify the role of ribosomes, endoplasmic
7.2 Cell Structure Lesson Objectives Describe the structure and function of the cell nucleus. Describe the role of vacuoles, lysosomes, and the cytoskeleton. Identify the role of ribosomes, endoplasmic
CHAPTER 5.1 5.2: Plasma Membrane Structure
 CHAPTER 5.1 5.2: Plasma Membrane Structure 1. Describe the structure of a phospholipid molecule. Be sure to describe their behavior in relationship to water. 2. What happens when a collection of phospholipids
CHAPTER 5.1 5.2: Plasma Membrane Structure 1. Describe the structure of a phospholipid molecule. Be sure to describe their behavior in relationship to water. 2. What happens when a collection of phospholipids
Plasma Membrane hydrophilic polar heads
 The Parts of the Cell 3 main parts in ALL cells: plasma membrane, cytoplasm, genetic material this is about the parts of a generic eukaryotic cell Plasma Membrane -is a fluid mosaic model membrane is fluid
The Parts of the Cell 3 main parts in ALL cells: plasma membrane, cytoplasm, genetic material this is about the parts of a generic eukaryotic cell Plasma Membrane -is a fluid mosaic model membrane is fluid
Effect Of Amino Acids On Plants
 Effect Of Amino Acids On Plants Agriculture production is a very intensive business and is related to better quality and better yield leading to better profitability Every farmer s dreams to achieve this
Effect Of Amino Acids On Plants Agriculture production is a very intensive business and is related to better quality and better yield leading to better profitability Every farmer s dreams to achieve this
How To Understand The Human Body
 Introduction to Biology and Chemistry Outline I. Introduction to biology A. Definition of biology - Biology is the study of life. B. Characteristics of Life 1. Form and size are characteristic. e.g. A
Introduction to Biology and Chemistry Outline I. Introduction to biology A. Definition of biology - Biology is the study of life. B. Characteristics of Life 1. Form and size are characteristic. e.g. A
Cell Membrane Structure (and How to Get Through One)
 Cell Membrane Structure (and How to Get Through One) A cell s membrane is a wall of sorts that defines the boundaries of a cell. The membrane provides protection and structure for the cell and acts as
Cell Membrane Structure (and How to Get Through One) A cell s membrane is a wall of sorts that defines the boundaries of a cell. The membrane provides protection and structure for the cell and acts as
Jan Baptisa van Helmont (1648)
 Instructions To help you navigate these slides, you should set your viewer to display thumbnails of these slides. On many viewers, this can be done by pressing the F4 key. The slides should be viewed in
Instructions To help you navigate these slides, you should set your viewer to display thumbnails of these slides. On many viewers, this can be done by pressing the F4 key. The slides should be viewed in
Class 10 NCERT Science Text Book Chapter 7 Control and Coordination
 Class 10 NCERT Science Text Book Chapter 7 Control and Coordination Question 1: What is the difference between a reflex action and walking? A reflex action is a rapid, automatic response to a stimulus.
Class 10 NCERT Science Text Book Chapter 7 Control and Coordination Question 1: What is the difference between a reflex action and walking? A reflex action is a rapid, automatic response to a stimulus.
BIOLOGICAL MEMBRANES: FUNCTIONS, STRUCTURES & TRANSPORT
 BIOLOGICAL MEMBRANES: FUNCTIONS, STRUCTURES & TRANSPORT UNIVERSITY OF PNG SCHOOL OF MEDICINE AND HEALTH SCIENCES DISCIPLINE OF BIOCHEMISTRY AND MOLECULAR BIOLOGY BMLS II / B Pharm II / BDS II VJ Temple
BIOLOGICAL MEMBRANES: FUNCTIONS, STRUCTURES & TRANSPORT UNIVERSITY OF PNG SCHOOL OF MEDICINE AND HEALTH SCIENCES DISCIPLINE OF BIOCHEMISTRY AND MOLECULAR BIOLOGY BMLS II / B Pharm II / BDS II VJ Temple
Nerves and Nerve Impulse
 Nerves and Nerve Impulse Terms Absolute refractory period: Period following stimulation during which no additional action potential can be evoked. Acetylcholine: Chemical transmitter substance released
Nerves and Nerve Impulse Terms Absolute refractory period: Period following stimulation during which no additional action potential can be evoked. Acetylcholine: Chemical transmitter substance released
Introduction to the Cell: Plant and Animal Cells
 Introduction to the Cell: Plant and Animal Cells Tissues, Organs, and Systems of Living Things Cells, Cell Division, and Animal Systems and Plant Systems Cell Specialization Human Systems All organisms
Introduction to the Cell: Plant and Animal Cells Tissues, Organs, and Systems of Living Things Cells, Cell Division, and Animal Systems and Plant Systems Cell Specialization Human Systems All organisms
3.1 Cells and cell function
 BTEC s own resources 3.1 Cells and cell function In this section: P1 How you are made Key terms Tissue a group of similar cells acting together to perform a particular function. Epithelial cells one of
BTEC s own resources 3.1 Cells and cell function In this section: P1 How you are made Key terms Tissue a group of similar cells acting together to perform a particular function. Epithelial cells one of
Chapter 2: Cell Structure and Function pg. 70-107
 UNIT 1: Biochemistry Chapter 2: Cell Structure and Function pg. 70-107 Organelles are internal structures that carry out specialized functions, interacting and complementing each other. Animal and plant
UNIT 1: Biochemistry Chapter 2: Cell Structure and Function pg. 70-107 Organelles are internal structures that carry out specialized functions, interacting and complementing each other. Animal and plant
SOME Important Points About Cellular Energetics by Dr. Ty C.M. Hoffman
 SOME Important Points About Cellular Energetics by Dr. Ty C.M. Hoffman An Introduction to Metabolism Most biochemical processes occur as biochemical pathways, each individual reaction of which is catalyzed
SOME Important Points About Cellular Energetics by Dr. Ty C.M. Hoffman An Introduction to Metabolism Most biochemical processes occur as biochemical pathways, each individual reaction of which is catalyzed
Cells & Cell Organelles
 Cells & Cell Organelles The Building Blocks of Life H Biology Types of cells bacteria cells Prokaryote - no organelles Eukaryotes - organelles animal cells plant cells Cell size comparison Animal cell
Cells & Cell Organelles The Building Blocks of Life H Biology Types of cells bacteria cells Prokaryote - no organelles Eukaryotes - organelles animal cells plant cells Cell size comparison Animal cell
