! Elements:! Isotopes:! ! Isotope Notation:!
|
|
|
- Noel Harrell
- 7 years ago
- Views:
Transcription
1 Lesson 27: The Nucleus Part 1: Elements and Isotopes The atom is made up of three kinds of subatomic particles: Particle Mass (kg) Charge (C) Proton Neutron Electron Elements: Isotopes: Isotope Notation: Example: Uranium-235 The atomic mass unit: The atomic mass unit is an alternative unit of mass used for very small particles. The atomic mass unit is, by definition, the average mass of a nucleon in the carbon-12 isotope. Example: Convert the mass of the proton, neutron and electron into atomic mass units Proton Neutron Electron Uranium-238
2 Your Physics 30 periodic table: The masses given on the periodic table were found using a mass spectrometer: Velocity Selector Mass Spectrometer (ion separator) B1 B2
3 Example: A singly charged ion passes through the velocity selector of a mass spectrometer undeflected. Then they enter the separation region and are deflected with a radius of 8.12 m. What is the mass in atomic mass units? Part 2: Mass Defect and Nuclear Binding Energy The mass of each isotope for each element has been measured precisely using a mass spectrometer. But when scientists predict what the mass of each isotope should be, based on the number of protons and neutrons found in the nucleus, they find a discrepancy between the predicted amount and the actual measured value. Example: The mass defect: Isotope Symbol Protons Neutrons Predicted Mass (u) Actual Mass (u) Oxygen Oxygen Oxygen
4 Where did the mass go? When a nucleus forms, a certain amount of energy is needed to hold all the nucleons together. Einstein s Formula: For Oxygen-16, the nuclear binding energy is: Example: What is the nuclear binding energy for the helium-4 nucleus if the measured mass of helium is u? Units of energy: Convert joules to ev: Convert ev to MeV:
5 Another alternative unit of mass: MeV/c 2 The MeV/c 2 is another alternative unit of mass used in nuclear and particle physics. This unit tells us the amount of MeV that would be released if the entire mass of a particle were converted into energy. Example: The mass of lithium-7 is u. What is the mass of the lithium nucleus in MeV/c 2? Units of Mass: Convert kg to u: Convert kg to MeV/c 2 : Practice Problems: 1. What is the binding energy in one atom of strontium-86 ( u)? What is the binding energy per nucleon? ( MeV, MeV/nucleon) 2. Isotopes are separated by a mass spectrometer. Ions are accelerated through a potential difference and then allowed to pass through a velocity selector. The velocity selector is composed of a uniform T magnetic field and a uniform electric field perpendicular to each other. If the electric field is produced using parallel plates that are 1.50 cm apart, what is the potential difference between the plates that will permit singly charged ions with a speed of 4.20 x 10 5 m/s to pass undeflected through the selector? (252 V) 3. Single charged ions pass undeflected through the velocity selector of a mass spectrometer. This velocity selector has a magnetic field of T and an electric field of 7000 V/m perpendicular to each other. These ions then enter a separation region where the magnetic field is the same as in the velocity selector. If the radius of the deflected ions is 8.13 mm, what is the mass of each ion? (1.16 x kg) 4. An ion source contains two isotopes of magnesium: 25 ( u) and 12Mg ( u). These ions travel undeflected through the velocity selector (B = T, E = 4.60 x 10 5 V/m) of a mass spectrometer. If both ions are doubly charged, how far apart are the ions in the ion detector (B = T)? ( m) 24 12Mg 5. A singly charged carbon ion travels in a circular path (r = 11.3 cm) through the ion?
6 separation region (B = T) of a mass spectrometer. If the velocity selector has a magnetic field of T and an electric field of 7.50 x 10 4 V/m perpendicular to each other, what is the mass number of the carbon isotope? (13) 6. Determine the mass defect of an atom of cobalt-59 which has a measured atomic mass of Express your answer in atomic mass units and kilograms. ( u, x kg) 7. Two isotopes of chlorine occur in nature. Their natural abundance is measured with a mass spectrometer. The chlorine-35 isotope has an atomic mass of u and a natural abundance of %. The chlorine-37 isotope has anatomic mass of and a natural abundance of %. By a calculation of your own, verify that the value of on the periodic table is a weighted average of the isotopes. 8. Find the binding energy in MeV for lithium-7 (atomic mass = u). ( MeV) 9. For radium-226 (atomic mass = u) obtain (a) the mass defect, (b) the binding energy in MeV, and (c) the binding energy per nucleon. ( u, MeV, MeV/nucleon)
F321 THE STRUCTURE OF ATOMS. ATOMS Atoms consist of a number of fundamental particles, the most important are... in the nucleus of an atom
 Atomic Structure F32 TE STRUCTURE OF ATOMS ATOMS Atoms consist of a number of fundamental particles, the most important are... Mass / kg Charge / C Relative mass Relative Charge PROTON NEUTRON ELECTRON
Atomic Structure F32 TE STRUCTURE OF ATOMS ATOMS Atoms consist of a number of fundamental particles, the most important are... Mass / kg Charge / C Relative mass Relative Charge PROTON NEUTRON ELECTRON
Chapter Five: Atomic Theory and Structure
 Chapter Five: Atomic Theory and Structure Evolution of Atomic Theory The ancient Greek scientist Democritus is often credited with developing the idea of the atom Democritus proposed that matter was, on
Chapter Five: Atomic Theory and Structure Evolution of Atomic Theory The ancient Greek scientist Democritus is often credited with developing the idea of the atom Democritus proposed that matter was, on
2 The Structure of Atoms
 CHAPTER 4 2 The Structure of Atoms SECTION Atoms KEY IDEAS As you read this section, keep these questions in mind: What do atoms of the same element have in common? What are isotopes? How is an element
CHAPTER 4 2 The Structure of Atoms SECTION Atoms KEY IDEAS As you read this section, keep these questions in mind: What do atoms of the same element have in common? What are isotopes? How is an element
Objectives 404 CHAPTER 9 RADIATION
 Objectives Explain the difference between isotopes of the same element. Describe the force that holds nucleons together. Explain the relationship between mass and energy according to Einstein s theory
Objectives Explain the difference between isotopes of the same element. Describe the force that holds nucleons together. Explain the relationship between mass and energy according to Einstein s theory
2 ATOMIC SYSTEMATICS AND NUCLEAR STRUCTURE
 2 ATOMIC SYSTEMATICS AND NUCLEAR STRUCTURE In this chapter the principles and systematics of atomic and nuclear physics are summarised briefly, in order to introduce the existence and characteristics of
2 ATOMIC SYSTEMATICS AND NUCLEAR STRUCTURE In this chapter the principles and systematics of atomic and nuclear physics are summarised briefly, in order to introduce the existence and characteristics of
CHARGED PARTICLES & MAGNETIC FIELDS - WebAssign
 Name: Period: Due Date: Lab Partners: CHARGED PARTICLES & MAGNETIC FIELDS - WebAssign Purpose: Use the CP program from Vernier to simulate the motion of charged particles in Magnetic and Electric Fields
Name: Period: Due Date: Lab Partners: CHARGED PARTICLES & MAGNETIC FIELDS - WebAssign Purpose: Use the CP program from Vernier to simulate the motion of charged particles in Magnetic and Electric Fields
............... [2] At the time of purchase of a Strontium-90 source, the activity is 3.7 10 6 Bq.
![............... [2] At the time of purchase of a Strontium-90 source, the activity is 3.7 10 6 Bq. ............... [2] At the time of purchase of a Strontium-90 source, the activity is 3.7 10 6 Bq.](/thumbs/40/21837699.jpg) 1 Strontium-90 decays with the emission of a β-particle to form Yttrium-90. The reaction is represented by the equation 90 38 The decay constant is 0.025 year 1. 90 39 0 1 Sr Y + e + 0.55 MeV. (a) Suggest,
1 Strontium-90 decays with the emission of a β-particle to form Yttrium-90. The reaction is represented by the equation 90 38 The decay constant is 0.025 year 1. 90 39 0 1 Sr Y + e + 0.55 MeV. (a) Suggest,
( + and - ) ( - and - ) ( + and + ) Atoms are mostly empty space. = the # of protons in the nucleus. = the # of protons in the nucleus
 Atoms are mostly empty space Atomic Structure Two regions of every atom: Nucleus - is made of protons and neutrons - is small and dense Electron cloud -is a region where you might find an electron -is
Atoms are mostly empty space Atomic Structure Two regions of every atom: Nucleus - is made of protons and neutrons - is small and dense Electron cloud -is a region where you might find an electron -is
Instructors Guide: Atoms and Their Isotopes
 Instructors Guide: Atoms and Their Isotopes Standards Connections Connections to NSTA Standards for Science Teacher Preparation C.3.a.1 Fundamental structures of atoms and molecules. C.3.b.27 Applications
Instructors Guide: Atoms and Their Isotopes Standards Connections Connections to NSTA Standards for Science Teacher Preparation C.3.a.1 Fundamental structures of atoms and molecules. C.3.b.27 Applications
MASS DEFECT AND BINDING ENERGY
 MASS DEFECT AND BINDING ENERGY The separate laws of Conservation of Mass and Conservation of Energy are not applied strictly on the nuclear level. It is possible to convert between mass and energy. Instead
MASS DEFECT AND BINDING ENERGY The separate laws of Conservation of Mass and Conservation of Energy are not applied strictly on the nuclear level. It is possible to convert between mass and energy. Instead
CHEM 1411 Chapter 5 Homework Answers
 1 CHEM 1411 Chapter 5 Homework Answers 1. Which statement regarding the gold foil experiment is false? (a) It was performed by Rutherford and his research group early in the 20 th century. (b) Most of
1 CHEM 1411 Chapter 5 Homework Answers 1. Which statement regarding the gold foil experiment is false? (a) It was performed by Rutherford and his research group early in the 20 th century. (b) Most of
Magnetic Fields and Forces. AP Physics B
 Magnetic ields and orces AP Physics acts about Magnetism Magnets have 2 poles (north and south) Like poles repel Unlike poles attract Magnets create a MAGNETIC IELD around them Magnetic ield A bar magnet
Magnetic ields and orces AP Physics acts about Magnetism Magnets have 2 poles (north and south) Like poles repel Unlike poles attract Magnets create a MAGNETIC IELD around them Magnetic ield A bar magnet
For convenience, we may consider an atom in two parts: the nucleus and the electrons.
 Atomic structure A. Introduction: In 1808, an English scientist called John Dalton proposed an atomic theory based on experimental findings. (1) Elements are made of extremely small particles called atoms.
Atomic structure A. Introduction: In 1808, an English scientist called John Dalton proposed an atomic theory based on experimental findings. (1) Elements are made of extremely small particles called atoms.
Main properties of atoms and nucleus
 Main properties of atoms and nucleus. Atom Structure.... Structure of Nuclei... 3. Definition of Isotopes... 4. Energy Characteristics of Nuclei... 5. Laws of Radioactive Nuclei Transformation... 3. Atom
Main properties of atoms and nucleus. Atom Structure.... Structure of Nuclei... 3. Definition of Isotopes... 4. Energy Characteristics of Nuclei... 5. Laws of Radioactive Nuclei Transformation... 3. Atom
ANSWER KEY : BUILD AN ATOM PART I: ATOM SCREEN Build an Atom simulation ( http://phet.colorado.edu/en/simulation/build an atom )
 ANSWER KEY : PART I: ATOM SCREEN Build an Atom simulation ( http://phet.colorado.edu/en/simulation/build an atom ) 1. Explore the Build an Atom simulation with your group. As you explore, talk about what
ANSWER KEY : PART I: ATOM SCREEN Build an Atom simulation ( http://phet.colorado.edu/en/simulation/build an atom ) 1. Explore the Build an Atom simulation with your group. As you explore, talk about what
Electrons in Atoms & Periodic Table Chapter 13 & 14 Assignment & Problem Set
 Electrons in Atoms & Periodic Table Name Warm-Ups (Show your work for credit) Date 1. Date 2. Date 3. Date 4. Date 5. Date 6. Date 7. Date 8. Electrons in Atoms & Periodic Table 2 Study Guide: Things You
Electrons in Atoms & Periodic Table Name Warm-Ups (Show your work for credit) Date 1. Date 2. Date 3. Date 4. Date 5. Date 6. Date 7. Date 8. Electrons in Atoms & Periodic Table 2 Study Guide: Things You
47374_04_p25-32.qxd 2/9/07 7:50 AM Page 25. 4 Atoms and Elements
 47374_04_p25-32.qxd 2/9/07 7:50 AM Page 25 4 Atoms and Elements 4.1 a. Cu b. Si c. K d. N e. Fe f. Ba g. Pb h. Sr 4.2 a. O b. Li c. S d. Al e. H f. Ne g. Sn h. Au 4.3 a. carbon b. chlorine c. iodine d.
47374_04_p25-32.qxd 2/9/07 7:50 AM Page 25 4 Atoms and Elements 4.1 a. Cu b. Si c. K d. N e. Fe f. Ba g. Pb h. Sr 4.2 a. O b. Li c. S d. Al e. H f. Ne g. Sn h. Au 4.3 a. carbon b. chlorine c. iodine d.
2014 Spring CHEM101 Ch1-2 Review Worksheet Modified by Dr. Cheng-Yu Lai,
 Ch1 1) Which of the following underlined items is not an intensive property? A) A chemical reaction requires 3.00 g of oxygen. B) The density of helium at 25 C is 1.64 10-4 g/cm3. C) The melting point
Ch1 1) Which of the following underlined items is not an intensive property? A) A chemical reaction requires 3.00 g of oxygen. B) The density of helium at 25 C is 1.64 10-4 g/cm3. C) The melting point
Basics of Nuclear Physics and Fission
 Basics of Nuclear Physics and Fission A basic background in nuclear physics for those who want to start at the beginning. Some of the terms used in this factsheet can be found in IEER s on-line glossary.
Basics of Nuclear Physics and Fission A basic background in nuclear physics for those who want to start at the beginning. Some of the terms used in this factsheet can be found in IEER s on-line glossary.
Test Bank - Chapter 4 Multiple Choice
 Test Bank - Chapter 4 The questions in the test bank cover the concepts from the lessons in Chapter 4. Select questions from any of the categories that match the content you covered with students. The
Test Bank - Chapter 4 The questions in the test bank cover the concepts from the lessons in Chapter 4. Select questions from any of the categories that match the content you covered with students. The
Objectives. PAM1014 Introduction to Radiation Physics. Constituents of Atoms. Atoms. Atoms. Atoms. Basic Atomic Theory
 PAM1014 Introduction to Radiation Physics Basic Atomic Theory Objectives Introduce and Molecules The periodic Table Electronic Energy Levels Atomic excitation & de-excitation Ionisation Molecules Constituents
PAM1014 Introduction to Radiation Physics Basic Atomic Theory Objectives Introduce and Molecules The periodic Table Electronic Energy Levels Atomic excitation & de-excitation Ionisation Molecules Constituents
Magnetism. d. gives the direction of the force on a charge moving in a magnetic field. b. results in negative charges moving. clockwise.
 Magnetism 1. An electron which moves with a speed of 3.0 10 4 m/s parallel to a uniform magnetic field of 0.40 T experiences a force of what magnitude? (e = 1.6 10 19 C) a. 4.8 10 14 N c. 2.2 10 24 N b.
Magnetism 1. An electron which moves with a speed of 3.0 10 4 m/s parallel to a uniform magnetic field of 0.40 T experiences a force of what magnitude? (e = 1.6 10 19 C) a. 4.8 10 14 N c. 2.2 10 24 N b.
Physics 1104 Midterm 2 Review: Solutions
 Physics 114 Midterm 2 Review: Solutions These review sheets cover only selected topics from the chemical and nuclear energy chapters and are not meant to be a comprehensive review. Topics covered in these
Physics 114 Midterm 2 Review: Solutions These review sheets cover only selected topics from the chemical and nuclear energy chapters and are not meant to be a comprehensive review. Topics covered in these
Atomic Theory Part 1
 Atomic Theory Part 1 Reading: Ch 2 sections 1 6, 8 Homework: Chapter 2: 39, 47, 43, 49, 51*, 53, 55, 57, 71, 73, 77, 99, 103 (optional) * = important homework question The Atomic Theory (John Dalton, 1803)
Atomic Theory Part 1 Reading: Ch 2 sections 1 6, 8 Homework: Chapter 2: 39, 47, 43, 49, 51*, 53, 55, 57, 71, 73, 77, 99, 103 (optional) * = important homework question The Atomic Theory (John Dalton, 1803)
Chapter 2 Atoms, Ions, and the Periodic Table
 Chapter 2 Atoms, Ions, and the Periodic Table 2.1 (a) neutron; (b) law of conservation of mass; (c) proton; (d) main-group element; (e) relative atomic mass; (f) mass number; (g) isotope; (h) cation; (i)
Chapter 2 Atoms, Ions, and the Periodic Table 2.1 (a) neutron; (b) law of conservation of mass; (c) proton; (d) main-group element; (e) relative atomic mass; (f) mass number; (g) isotope; (h) cation; (i)
9/13/2013. However, Dalton thought that an atom was just a tiny sphere with no internal parts. This is sometimes referred to as the cannonball model.
 John Dalton was an English scientist who lived in the early 1800s. Dalton s atomic theory served as a model for how matter worked. The principles of Dalton s atomic theory are: 1. Elements are made of
John Dalton was an English scientist who lived in the early 1800s. Dalton s atomic theory served as a model for how matter worked. The principles of Dalton s atomic theory are: 1. Elements are made of
Chemistry CP Unit 2 Atomic Structure and Electron Configuration. Learning Targets (Your exam at the end of Unit 2 will assess the following:)
 Chemistry CP Unit 2 Atomic Structure and Electron Learning Targets (Your exam at the end of Unit 2 will assess the following:) 2. Atomic Structure and Electron 2-1. Give the one main contribution to the
Chemistry CP Unit 2 Atomic Structure and Electron Learning Targets (Your exam at the end of Unit 2 will assess the following:) 2. Atomic Structure and Electron 2-1. Give the one main contribution to the
Nuclear Structure. particle relative charge relative mass proton +1 1 atomic mass unit neutron 0 1 atomic mass unit electron -1 negligible mass
 Protons, neutrons and electrons Nuclear Structure particle relative charge relative mass proton 1 1 atomic mass unit neutron 0 1 atomic mass unit electron -1 negligible mass Protons and neutrons make up
Protons, neutrons and electrons Nuclear Structure particle relative charge relative mass proton 1 1 atomic mass unit neutron 0 1 atomic mass unit electron -1 negligible mass Protons and neutrons make up
Matter. Atomic weight, Molecular weight and Mole
 Matter Atomic weight, Molecular weight and Mole Atomic Mass Unit Chemists of the nineteenth century realized that, in order to measure the mass of an atomic particle, it was useless to use the standard
Matter Atomic weight, Molecular weight and Mole Atomic Mass Unit Chemists of the nineteenth century realized that, in order to measure the mass of an atomic particle, it was useless to use the standard
Unit 3 Study Guide: Electron Configuration & The Periodic Table
 Name: Teacher s Name: Class: Block: Date: Unit 3 Study Guide: Electron Configuration & The Periodic Table 1. For each of the following elements, state whether the element is radioactive, synthetic or both.
Name: Teacher s Name: Class: Block: Date: Unit 3 Study Guide: Electron Configuration & The Periodic Table 1. For each of the following elements, state whether the element is radioactive, synthetic or both.
Chapter 19 Magnetic Forces and Fields
 Chapter 19 Magnetic Forces and Fields Student: 3. The magnetism of the Earth acts approximately as if it originates from a huge bar magnet within the Earth. Which of the following statements are true?
Chapter 19 Magnetic Forces and Fields Student: 3. The magnetism of the Earth acts approximately as if it originates from a huge bar magnet within the Earth. Which of the following statements are true?
Chapter 1: Moles and equations. Learning outcomes. you should be able to:
 Chapter 1: Moles and equations 1 Learning outcomes you should be able to: define and use the terms: relative atomic mass, isotopic mass and formula mass based on the 12 C scale perform calculations, including
Chapter 1: Moles and equations 1 Learning outcomes you should be able to: define and use the terms: relative atomic mass, isotopic mass and formula mass based on the 12 C scale perform calculations, including
Atomic Structure Chapter 5 Assignment & Problem Set
 Atomic Structure Name Warm-Ups (Show your work for credit) Date 1. Date 2. Date 3. Date 4. Date 5. Date 6. Date 7. Date 8. Atomic Structure 2 Study Guide: Things You Must Know Vocabulary (know the definition
Atomic Structure Name Warm-Ups (Show your work for credit) Date 1. Date 2. Date 3. Date 4. Date 5. Date 6. Date 7. Date 8. Atomic Structure 2 Study Guide: Things You Must Know Vocabulary (know the definition
Write an equation, including state symbols, for the ionisation of indium that requires the minimum energy.(1)
 MINI MOCK Questions Unit 1 Atomic Structure AS Chemistry Q1. Indium is in Group 3 in the Periodic Table and exists as a mixture of the isotopes 113 In and 115 In. (a) Use your understanding of the Periodic
MINI MOCK Questions Unit 1 Atomic Structure AS Chemistry Q1. Indium is in Group 3 in the Periodic Table and exists as a mixture of the isotopes 113 In and 115 In. (a) Use your understanding of the Periodic
ATOMS A T O M S, I S O T O P E S, A N D I O N S. The Academic Support Center @ Daytona State College (Science 120, Page 1 of 39)
 ATOMS A T O M S, I S O T O P E S, A N D I O N S The Academic Support Center @ Daytona State College (Science 120, Page 1 of 39) THE ATOM All elements listed on the periodic table are made up of atoms.
ATOMS A T O M S, I S O T O P E S, A N D I O N S The Academic Support Center @ Daytona State College (Science 120, Page 1 of 39) THE ATOM All elements listed on the periodic table are made up of atoms.
Phys222 Winter 2012 Quiz 4 Chapters 29-31. Name
 Name If you think that no correct answer is provided, give your answer, state your reasoning briefly; append additional sheet of paper if necessary. 1. A particle (q = 5.0 nc, m = 3.0 µg) moves in a region
Name If you think that no correct answer is provided, give your answer, state your reasoning briefly; append additional sheet of paper if necessary. 1. A particle (q = 5.0 nc, m = 3.0 µg) moves in a region
CHAPTER 4: ATOMS AND ELEMENTS
 CHAPTER 4: ATOMS AND ELEMENTS Problems: 1-70 then after Chapter 9, complete 71-94, 103-104, 107-108, 113-114 4.1 Experiencing Atoms at Tiburon atom: smallest identifiable unit of an element All matter
CHAPTER 4: ATOMS AND ELEMENTS Problems: 1-70 then after Chapter 9, complete 71-94, 103-104, 107-108, 113-114 4.1 Experiencing Atoms at Tiburon atom: smallest identifiable unit of an element All matter
100 cm 1 m. = 614 cm. 6.14 m. 2.54 cm. 1 m 1 in. 1 m. 2.54 cm 1ft. 1 in = 242 in. 614 cm. 242 in 1 ft. 1 in. 100 cm = 123 m
 Units and Unit Conversions 6. Define the problem: If the nucleus were scaled to a diameter of 4 cm, determine the diameter of the atom. Develop a plan: Find the accepted relationship between the size of
Units and Unit Conversions 6. Define the problem: If the nucleus were scaled to a diameter of 4 cm, determine the diameter of the atom. Develop a plan: Find the accepted relationship between the size of
Lesson 43: Alpha, Beta, & Gamma Decay
 Lesson 43: Alpha, Beta, & Gamma Decay The late 18s and early 19s were a period of intense research into the new nuclear realm of physics. In 1896 Henri Becquerel found that a sample of uranium he was doing
Lesson 43: Alpha, Beta, & Gamma Decay The late 18s and early 19s were a period of intense research into the new nuclear realm of physics. In 1896 Henri Becquerel found that a sample of uranium he was doing
Monday 11 June 2012 Afternoon
 Monday 11 June 2012 Afternoon A2 GCE PHYSICS B (ADVANCING PHYSICS) G495 Field and Particle Pictures *G412090612* Candidates answer on the Question Paper. OCR supplied materials: Data, Formulae and Relationships
Monday 11 June 2012 Afternoon A2 GCE PHYSICS B (ADVANCING PHYSICS) G495 Field and Particle Pictures *G412090612* Candidates answer on the Question Paper. OCR supplied materials: Data, Formulae and Relationships
Multiple Choice Identify the letter of the choice that best completes the statement or answers the question.
 Introduction to Chemistry Exam 2 Practice Problems 1 Multiple Choice Identify the letter of the choice that best completes the statement or answers the question. 1.Atoms consist principally of what three
Introduction to Chemistry Exam 2 Practice Problems 1 Multiple Choice Identify the letter of the choice that best completes the statement or answers the question. 1.Atoms consist principally of what three
13C NMR Spectroscopy
 13 C NMR Spectroscopy Introduction Nuclear magnetic resonance spectroscopy (NMR) is the most powerful tool available for structural determination. A nucleus with an odd number of protons, an odd number
13 C NMR Spectroscopy Introduction Nuclear magnetic resonance spectroscopy (NMR) is the most powerful tool available for structural determination. A nucleus with an odd number of protons, an odd number
Chapter 22 Magnetism
 22.6 Electric Current, Magnetic Fields, and Ampere s Law Chapter 22 Magnetism 22.1 The Magnetic Field 22.2 The Magnetic Force on Moving Charges 22.3 The Motion of Charged particles in a Magnetic Field
22.6 Electric Current, Magnetic Fields, and Ampere s Law Chapter 22 Magnetism 22.1 The Magnetic Field 22.2 The Magnetic Force on Moving Charges 22.3 The Motion of Charged particles in a Magnetic Field
Chapter NP-5. Nuclear Physics. Nuclear Reactions TABLE OF CONTENTS INTRODUCTION OBJECTIVES 1.0 NUCLEAR REACTIONS 2.0 NEUTRON INTERACTIONS
 Chapter NP-5 Nuclear Physics Nuclear Reactions TABLE OF CONTENTS INTRODUCTION OBJECTIVES 1.0 2.0 NEUTRON INTERACTIONS 2.1 ELASTIC SCATTERING 2.2 INELASTIC SCATTERING 2.3 RADIATIVE CAPTURE 2.4 PARTICLE
Chapter NP-5 Nuclear Physics Nuclear Reactions TABLE OF CONTENTS INTRODUCTION OBJECTIVES 1.0 2.0 NEUTRON INTERACTIONS 2.1 ELASTIC SCATTERING 2.2 INELASTIC SCATTERING 2.3 RADIATIVE CAPTURE 2.4 PARTICLE
EXPERIMENT 4 The Periodic Table - Atoms and Elements
 EXPERIMENT 4 The Periodic Table - Atoms and Elements INTRODUCTION Primary substances, called elements, build all the materials around you. There are more than 109 different elements known today. The elements
EXPERIMENT 4 The Periodic Table - Atoms and Elements INTRODUCTION Primary substances, called elements, build all the materials around you. There are more than 109 different elements known today. The elements
Q27.1 When a charged particle moves near a bar magnet, the magnetic force on the particle at a certain point depends
 Q27.1 When a charged particle moves near a bar magnet, the magnetic force on the particle at a certain point depends A. on the direction of the magnetic field at that point only. B. on the magnetic field
Q27.1 When a charged particle moves near a bar magnet, the magnetic force on the particle at a certain point depends A. on the direction of the magnetic field at that point only. B. on the magnetic field
Physics 30 Worksheet #10 : Magnetism From Electricity
 Physics 30 Worksheet #10 : Magnetism From Electricity 1. Draw the magnetic field surrounding the wire showing electron current below. x 2. Draw the magnetic field surrounding the wire showing electron
Physics 30 Worksheet #10 : Magnetism From Electricity 1. Draw the magnetic field surrounding the wire showing electron current below. x 2. Draw the magnetic field surrounding the wire showing electron
Periodic Table. 1. In the modern Periodic Table, the elements are arranged in order of increasing. A. atomic number B. mass number
 Name: ate: 1. In the modern, the elements are arranged in order of increasing. atomic number. mass number. oxidation number. valence number 5. s the elements in Group I are considered in order of increasing
Name: ate: 1. In the modern, the elements are arranged in order of increasing. atomic number. mass number. oxidation number. valence number 5. s the elements in Group I are considered in order of increasing
2. John Dalton did his research work in which of the following countries? a. France b. Greece c. Russia d. England
 CHAPTER 3 1. Which combination of individual and contribution is not correct? a. Antoine Lavoisier - clarified confusion over cause of burning b. John Dalton - proposed atomic theory c. Marie Curie - discovered
CHAPTER 3 1. Which combination of individual and contribution is not correct? a. Antoine Lavoisier - clarified confusion over cause of burning b. John Dalton - proposed atomic theory c. Marie Curie - discovered
Unit 1 Practice Test. Matching
 Unit 1 Practice Test Matching Match each item with the correct statement below. a. proton d. electron b. nucleus e. neutron c. atom 1. the smallest particle of an element that retains the properties of
Unit 1 Practice Test Matching Match each item with the correct statement below. a. proton d. electron b. nucleus e. neutron c. atom 1. the smallest particle of an element that retains the properties of
Atomic Calculations. 2.1 Composition of the Atom. number of protons + number of neutrons = mass number
 2.1 Composition of the Atom Atomic Calculations number of protons + number of neutrons = mass number number of neutrons = mass number - number of protons number of protons = number of electrons IF positive
2.1 Composition of the Atom Atomic Calculations number of protons + number of neutrons = mass number number of neutrons = mass number - number of protons number of protons = number of electrons IF positive
1. Units of a magnetic field might be: A. C m/s B. C s/m C. C/kg D. kg/c s E. N/C m ans: D
 Chapter 28: MAGNETIC FIELDS 1 Units of a magnetic field might be: A C m/s B C s/m C C/kg D kg/c s E N/C m 2 In the formula F = q v B: A F must be perpendicular to v but not necessarily to B B F must be
Chapter 28: MAGNETIC FIELDS 1 Units of a magnetic field might be: A C m/s B C s/m C C/kg D kg/c s E N/C m 2 In the formula F = q v B: A F must be perpendicular to v but not necessarily to B B F must be
Charged Particle in a Magnetic Field
 Charged Particle in a Magnetic Field Consider a particle moving in an external magnetic field with its velocity perpendicular to the field The force is always directed toward the center of the circular
Charged Particle in a Magnetic Field Consider a particle moving in an external magnetic field with its velocity perpendicular to the field The force is always directed toward the center of the circular
Amount of Substance. http://www.avogadro.co.uk/definitions/elemcompmix.htm
 Page 1 of 14 Amount of Substance Key terms in this chapter are: Element Compound Mixture Atom Molecule Ion Relative Atomic Mass Avogadro constant Mole Isotope Relative Isotopic Mass Relative Molecular
Page 1 of 14 Amount of Substance Key terms in this chapter are: Element Compound Mixture Atom Molecule Ion Relative Atomic Mass Avogadro constant Mole Isotope Relative Isotopic Mass Relative Molecular
Chapter NP-1. Nuclear Physics. Atomic Nature of Matter TABLE OF CONTENTS INTRODUCTION OBJECTIVES 1.0 PROPERTIES OF SUBSTANCES
 Chapter NP-1 Nuclear Physics Atomic Nature of Matter TABLE OF CONTENTS INTRODUCTION OBJECTIVES 1.0 PROPERTIES OF SUBSTANCES 1.1 CHEMICAL AND PHYSICAL PROPERTIES 2.0 COMPOSITION OF ATOMS 2.1 ATOMIC STRUCTURE
Chapter NP-1 Nuclear Physics Atomic Nature of Matter TABLE OF CONTENTS INTRODUCTION OBJECTIVES 1.0 PROPERTIES OF SUBSTANCES 1.1 CHEMICAL AND PHYSICAL PROPERTIES 2.0 COMPOSITION OF ATOMS 2.1 ATOMIC STRUCTURE
AP2 Magnetism. (c) Explain why the magnetic field does no work on the particle as it moves in its circular path.
 A charged particle is projected from point P with velocity v at a right angle to a uniform magnetic field directed out of the plane of the page as shown. The particle moves along a circle of radius R.
A charged particle is projected from point P with velocity v at a right angle to a uniform magnetic field directed out of the plane of the page as shown. The particle moves along a circle of radius R.
EARLY ATOMIC THEORY AND STRUCTURE
 CHAPTER 5 EARLY ATOMIC THEORY AND STRUCTURE SOLUTIONS TO REVIEW QUESTIONS 1. Elements are composed of indivisable particles called atoms. Atoms of the same element have the same properties; atoms of different
CHAPTER 5 EARLY ATOMIC THEORY AND STRUCTURE SOLUTIONS TO REVIEW QUESTIONS 1. Elements are composed of indivisable particles called atoms. Atoms of the same element have the same properties; atoms of different
Mass Spectrometry. Overview
 Mass Spectrometry Overview Mass Spectrometry is an analytic technique that utilizes the degree of deflection of charged particles by a magnetic field to find the relative masses of molecular ions and fragments.2
Mass Spectrometry Overview Mass Spectrometry is an analytic technique that utilizes the degree of deflection of charged particles by a magnetic field to find the relative masses of molecular ions and fragments.2
Masses in Atomic Units
 Nuclear Composition - the forces binding protons and neutrons in the nucleus are much stronger (binding energy of MeV) than the forces binding electrons to the atom (binding energy of ev) - the constituents
Nuclear Composition - the forces binding protons and neutrons in the nucleus are much stronger (binding energy of MeV) than the forces binding electrons to the atom (binding energy of ev) - the constituents
Pearson Physics Level 30 Unit VIII Atomic Physics: Chapter 17 Solutions
 Pearson Physics Level 30 Unit VIII Atomic Physics: Chapter 17 Solutions Student Book page 831 Concept Check Since neutrons have no charge, they do not create ions when passing through the liquid in a bubble
Pearson Physics Level 30 Unit VIII Atomic Physics: Chapter 17 Solutions Student Book page 831 Concept Check Since neutrons have no charge, they do not create ions when passing through the liquid in a bubble
The Structure of the Atom
 The Structure of the Atom Copyright Glencoe/McGraw-Hill, a division of the McGraw-Hill Companies, Inc. Section 4. Early Ideas About Matter pages 02 05 Section 4. Assessment page 05. Contrast the methods
The Structure of the Atom Copyright Glencoe/McGraw-Hill, a division of the McGraw-Hill Companies, Inc. Section 4. Early Ideas About Matter pages 02 05 Section 4. Assessment page 05. Contrast the methods
Solar Energy Production
 Solar Energy Production We re now ready to address the very important question: What makes the Sun shine? Why is this such an important topic in astronomy? As humans, we see in the visible part of the
Solar Energy Production We re now ready to address the very important question: What makes the Sun shine? Why is this such an important topic in astronomy? As humans, we see in the visible part of the
******* KEY ******* Atomic Structure & Periodic Table Test Study Guide
 Atomic Structure & Periodic Table Test Study Guide VOCABULARY: Write a brief definition of each term in the space provided. 1. Atoms: smallest unit of an element that has all of the properties of that
Atomic Structure & Periodic Table Test Study Guide VOCABULARY: Write a brief definition of each term in the space provided. 1. Atoms: smallest unit of an element that has all of the properties of that
Introduction to Nuclear Physics
 Introduction to Nuclear Physics 1. Atomic Structure and the Periodic Table According to the Bohr-Rutherford model of the atom, also called the solar system model, the atom consists of a central nucleus
Introduction to Nuclear Physics 1. Atomic Structure and the Periodic Table According to the Bohr-Rutherford model of the atom, also called the solar system model, the atom consists of a central nucleus
Conceptual: 1, 3, 5, 6, 8, 16, 18, 19. Problems: 4, 6, 8, 11, 16, 20, 23, 27, 34, 41, 45, 56, 60, 65. Conceptual Questions
 Conceptual: 1, 3, 5, 6, 8, 16, 18, 19 Problems: 4, 6, 8, 11, 16, 20, 23, 27, 34, 41, 45, 56, 60, 65 Conceptual Questions 1. The magnetic field cannot be described as the magnetic force per unit charge
Conceptual: 1, 3, 5, 6, 8, 16, 18, 19 Problems: 4, 6, 8, 11, 16, 20, 23, 27, 34, 41, 45, 56, 60, 65 Conceptual Questions 1. The magnetic field cannot be described as the magnetic force per unit charge
List the 3 main types of subatomic particles and indicate the mass and electrical charge of each.
 Basic Chemistry Why do we study chemistry in a biology course? All living organisms are composed of chemicals. To understand life, we must understand the structure, function, and properties of the chemicals
Basic Chemistry Why do we study chemistry in a biology course? All living organisms are composed of chemicals. To understand life, we must understand the structure, function, and properties of the chemicals
Untitled Document. 1. Which of the following best describes an atom? 4. Which statement best describes the density of an atom s nucleus?
 Name: Date: 1. Which of the following best describes an atom? A. protons and electrons grouped together in a random pattern B. protons and electrons grouped together in an alternating pattern C. a core
Name: Date: 1. Which of the following best describes an atom? A. protons and electrons grouped together in a random pattern B. protons and electrons grouped together in an alternating pattern C. a core
Physics 112 Homework 5 (solutions) (2004 Fall) Solutions to Homework Questions 5
 Solutions to Homework Questions 5 Chapt19, Problem-2: (a) Find the direction of the force on a proton (a positively charged particle) moving through the magnetic fields in Figure P19.2, as shown. (b) Repeat
Solutions to Homework Questions 5 Chapt19, Problem-2: (a) Find the direction of the force on a proton (a positively charged particle) moving through the magnetic fields in Figure P19.2, as shown. (b) Repeat
Trends of the Periodic Table Diary
 Trends of the Periodic Table Diary Trends are patterns of behaviors that atoms on the periodic table of elements follow. Trends hold true most of the time, but there are exceptions, or blips, where the
Trends of the Periodic Table Diary Trends are patterns of behaviors that atoms on the periodic table of elements follow. Trends hold true most of the time, but there are exceptions, or blips, where the
Atomic Structure: Chapter Problems
 Atomic Structure: Chapter Problems Bohr Model Class Work 1. Describe the nuclear model of the atom. 2. Explain the problems with the nuclear model of the atom. 3. According to Niels Bohr, what does n stand
Atomic Structure: Chapter Problems Bohr Model Class Work 1. Describe the nuclear model of the atom. 2. Explain the problems with the nuclear model of the atom. 3. According to Niels Bohr, what does n stand
NOTES ON The Structure of the Atom
 NOTES ON The Structure of the Atom Chemistry is the study of matter and its properties. Those properties can be explained by examining the atoms that compose the matter. An atom is the smallest particle
NOTES ON The Structure of the Atom Chemistry is the study of matter and its properties. Those properties can be explained by examining the atoms that compose the matter. An atom is the smallest particle
Bohr s Model of the Atom
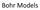 Bohr Models Bohr s Model of the Atom Focuses on electrons and their arrangement. Bohr stated that electrons move with constant speed in fixed orbits around the nucleus, like planets around a sun. Bohr
Bohr Models Bohr s Model of the Atom Focuses on electrons and their arrangement. Bohr stated that electrons move with constant speed in fixed orbits around the nucleus, like planets around a sun. Bohr
Level 3 Achievement Scale
 Unit 1: Atoms Level 3 Achievement Scale Can state the key results of the experiments associated with Dalton, Rutherford, Thomson, Chadwick, and Bohr and what this lead each to conclude. Can explain that
Unit 1: Atoms Level 3 Achievement Scale Can state the key results of the experiments associated with Dalton, Rutherford, Thomson, Chadwick, and Bohr and what this lead each to conclude. Can explain that
UNIT (2) ATOMS AND ELEMENTS
 UNIT (2) ATOMS AND ELEMENTS 2.1 Elements An element is a fundamental substance that cannot be broken down by chemical means into simpler substances. Each element is represented by an abbreviation called
UNIT (2) ATOMS AND ELEMENTS 2.1 Elements An element is a fundamental substance that cannot be broken down by chemical means into simpler substances. Each element is represented by an abbreviation called
Elements in the periodic table are indicated by SYMBOLS. To the left of the symbol we find the atomic mass (A) at the upper corner, and the atomic num
 . ATOMIC STRUCTURE FUNDAMENTALS LEARNING OBJECTIVES To review the basics concepts of atomic structure that have direct relevance to the fundamental concepts of organic chemistry. This material is essential
. ATOMIC STRUCTURE FUNDAMENTALS LEARNING OBJECTIVES To review the basics concepts of atomic structure that have direct relevance to the fundamental concepts of organic chemistry. This material is essential
PHYA5/1. General Certificate of Education Advanced Level Examination June 2011. Unit 5 Nuclear and Thermal Physics Section A
 Centre Number Surname Candidate Number For Examinerʼs Use Other Names Candidate Signature Examinerʼs Initials General Certificate of Education Advanced Level Examination June 2011 Question 1 2 Mark Physics
Centre Number Surname Candidate Number For Examinerʼs Use Other Names Candidate Signature Examinerʼs Initials General Certificate of Education Advanced Level Examination June 2011 Question 1 2 Mark Physics
1. In the general symbol cleus, which of the three letters. 2. What is the mass number of an alpha particle?
 1. In the general symbol cleus, which of the three letters Z A X for a nu represents the atomic number? 2. What is the mass number of an alpha particle? 3. What is the mass number of a beta particle? 4.
1. In the general symbol cleus, which of the three letters Z A X for a nu represents the atomic number? 2. What is the mass number of an alpha particle? 3. What is the mass number of a beta particle? 4.
PY106 Class13. Permanent Magnets. Magnetic Fields and Forces on Moving Charges. Interactions between magnetic north and south poles.
 Permanent Magnets Magnetic ields and orces on Moing Charges 1 We encounter magnetic fields frequently in daily life from those due to a permanent magnet. Each permanent magnet has a north pole and a south
Permanent Magnets Magnetic ields and orces on Moing Charges 1 We encounter magnetic fields frequently in daily life from those due to a permanent magnet. Each permanent magnet has a north pole and a south
Chemistry 1000 Lecture 2: Nuclear reactions and radiation. Marc R. Roussel
 Chemistry 1000 Lecture 2: Nuclear reactions and radiation Marc R. Roussel Nuclear reactions Ordinary chemical reactions do not involve the nuclei, so we can balance these reactions by making sure that
Chemistry 1000 Lecture 2: Nuclear reactions and radiation Marc R. Roussel Nuclear reactions Ordinary chemical reactions do not involve the nuclei, so we can balance these reactions by making sure that
SCH 3UI Unit 2 Outline Up to Quiz #1 Atomic Theory and the Periodic Table
 Lesson Topics Covered SCH 3UI Unit 2 Outline Up to Quiz #1 Atomic Theory and the Periodic Table 1 Note: History of Atomic Theory progression of understanding of composition of matter; ancient Greeks and
Lesson Topics Covered SCH 3UI Unit 2 Outline Up to Quiz #1 Atomic Theory and the Periodic Table 1 Note: History of Atomic Theory progression of understanding of composition of matter; ancient Greeks and
Quiz: Work and Energy
 Quiz: Work and Energy A charged particle enters a uniform magnetic field. What happens to the kinetic energy of the particle? (1) it increases (2) it decreases (3) it stays the same (4) it changes with
Quiz: Work and Energy A charged particle enters a uniform magnetic field. What happens to the kinetic energy of the particle? (1) it increases (2) it decreases (3) it stays the same (4) it changes with
Nuclear Physics. Nuclear Physics comprises the study of:
 Nuclear Physics Nuclear Physics comprises the study of: The general properties of nuclei The particles contained in the nucleus The interaction between these particles Radioactivity and nuclear reactions
Nuclear Physics Nuclear Physics comprises the study of: The general properties of nuclei The particles contained in the nucleus The interaction between these particles Radioactivity and nuclear reactions
3 Atomic Structure 15
 3 Atomic Structure 15 3.1 Atoms You need to be familiar with the terms in italics The diameter of the nucleus is approximately 10-15 m and an atom 10-10 m. All matter consists of atoms. An atom can be
3 Atomic Structure 15 3.1 Atoms You need to be familiar with the terms in italics The diameter of the nucleus is approximately 10-15 m and an atom 10-10 m. All matter consists of atoms. An atom can be
@ Oxford Fajar Sdn. Bhd. (008974-T) 2012. Matter. 1.1 Atoms and Molecules 1.2 Mole Concept 1.3 Stoichiometry
 1 Matter 1.1 Atoms and Molecules 1.2 Mole Concept 1.3 Stoichiometry 2 Chemistry for Matriculation Semester 1 1.1 Atoms and Molecules LEARNING OUTCOMES Describe proton, electron and neutron in terms of
1 Matter 1.1 Atoms and Molecules 1.2 Mole Concept 1.3 Stoichiometry 2 Chemistry for Matriculation Semester 1 1.1 Atoms and Molecules LEARNING OUTCOMES Describe proton, electron and neutron in terms of
Chapter 21. Magnetic Forces and Magnetic Fields
 Chapter 21 Magnetic Forces and Magnetic Fields 21.1 Magnetic Fields The needle of a compass is permanent magnet that has a north magnetic pole (N) at one end and a south magnetic pole (S) at the other.
Chapter 21 Magnetic Forces and Magnetic Fields 21.1 Magnetic Fields The needle of a compass is permanent magnet that has a north magnetic pole (N) at one end and a south magnetic pole (S) at the other.
ATOMS AND THE PERIODIC TABLE CHAPTER 3 PHYSICAL SCIENCE
 ATOMS AND THE PERIODIC TABLE CHAPTER 3 PHYSICAL SCIENCE Chapter 3 Vocabulary Words (27 words) Nucleus Atomic number Proton Mass number Neutron Isotopes Electron Atomic mass unit (amu) Energy level Average
ATOMS AND THE PERIODIC TABLE CHAPTER 3 PHYSICAL SCIENCE Chapter 3 Vocabulary Words (27 words) Nucleus Atomic number Proton Mass number Neutron Isotopes Electron Atomic mass unit (amu) Energy level Average
ATOMS: ATOMIC STRUCTURE QUESTIONS AND ANSWERS
 ATOMS: ATOMIC STRUCTURE QUESTIONS AND ANSWERS QUESTION ONE: MODELS OF THE ATOM (2011;1) At different times scientists have proposed various descriptions or models of the atom to match experimental evidence
ATOMS: ATOMIC STRUCTURE QUESTIONS AND ANSWERS QUESTION ONE: MODELS OF THE ATOM (2011;1) At different times scientists have proposed various descriptions or models of the atom to match experimental evidence
Atoms, Ions and Molecules The Building Blocks of Matter
 Atoms, Ions and Molecules The Building Blocks of Matter Chapter 2 1 Chapter Outline 2.1 The Rutherford Model of Atomic Structure 2.2 Nuclides and Their Symbols 2.3 Navigating the Periodic Table 2.4 The
Atoms, Ions and Molecules The Building Blocks of Matter Chapter 2 1 Chapter Outline 2.1 The Rutherford Model of Atomic Structure 2.2 Nuclides and Their Symbols 2.3 Navigating the Periodic Table 2.4 The
Chapter 5 TEST: The Periodic Table name
 Chapter 5 TEST: The Periodic Table name HPS # date: Multiple Choice Identify the choice that best completes the statement or answers the question. 1. The order of elements in the periodic table is based
Chapter 5 TEST: The Periodic Table name HPS # date: Multiple Choice Identify the choice that best completes the statement or answers the question. 1. The order of elements in the periodic table is based
4.1 Studying Atom. Early evidence used to develop models of atoms.
 4.1 Studying Atom Early evidence used to develop models of atoms. Democritus said that all matter consisted of extremely small particles that could NOT be divided called these particles atoms from the
4.1 Studying Atom Early evidence used to develop models of atoms. Democritus said that all matter consisted of extremely small particles that could NOT be divided called these particles atoms from the
Homework #10 (749508)
 Homework #10 (749508) Current Score: 0 out of 100 Description Homework on quantum physics and radioactivity Instructions Answer all the questions as best you can. 1. Hewitt10 32.E.001. [481697] 0/5 points
Homework #10 (749508) Current Score: 0 out of 100 Description Homework on quantum physics and radioactivity Instructions Answer all the questions as best you can. 1. Hewitt10 32.E.001. [481697] 0/5 points
UNIVERSITY OF SASKATCHEWAN Department of Physics and Engineering Physics
 UNIVERSITY OF SASKATCHEWAN Department of Physics and Engineering Physics Physics 111.6 MIDTERM TEST #4 March 15, 2007 Time: 90 minutes NAME: (Last) Please Print (Given) STUDENT NO.: LECTURE SECTION (please
UNIVERSITY OF SASKATCHEWAN Department of Physics and Engineering Physics Physics 111.6 MIDTERM TEST #4 March 15, 2007 Time: 90 minutes NAME: (Last) Please Print (Given) STUDENT NO.: LECTURE SECTION (please
Elements, Atoms & Ions
 Introductory Chemistry: A Foundation FOURTH EDITION by Steven S. Zumdahl University of Illinois Elements, Atoms & Ions Chapter 4 1 2 Elements Aims: To learn about the relative abundances of the elements,
Introductory Chemistry: A Foundation FOURTH EDITION by Steven S. Zumdahl University of Illinois Elements, Atoms & Ions Chapter 4 1 2 Elements Aims: To learn about the relative abundances of the elements,
Atoms, Ions and Molecules The Building Blocks of Matter
 Atoms, Ions and Molecules The Building Blocks of Matter Chapter 2 1 Chapter Outline 2.1 The Rutherford Model of Atomic Structure 2.2 Nuclides and Their Symbols 2.3 Navigating the Periodic Table 2.4 The
Atoms, Ions and Molecules The Building Blocks of Matter Chapter 2 1 Chapter Outline 2.1 The Rutherford Model of Atomic Structure 2.2 Nuclides and Their Symbols 2.3 Navigating the Periodic Table 2.4 The
Chem term # 1 review sheet C. 12 A. 1
 hem term # 1 review sheet Name: ate: 1. n isotope of which element has an atomic number of 6 and a mass number of 14?. carbon. magnesium. nitrogen. silicon 6. Which atoms represent different isotopes of
hem term # 1 review sheet Name: ate: 1. n isotope of which element has an atomic number of 6 and a mass number of 14?. carbon. magnesium. nitrogen. silicon 6. Which atoms represent different isotopes of
Name Block Date Ch 17 Atomic Nature of Matter Notes Mrs. Peck. atoms- the smallest particle of an element that can be identified with that element
 Name Block Date Ch 17 Atomic Nature of Matter Notes Mrs. Peck atoms- the smallest particle of an element that can be identified with that element are the building blocks of matter consists of protons and
Name Block Date Ch 17 Atomic Nature of Matter Notes Mrs. Peck atoms- the smallest particle of an element that can be identified with that element are the building blocks of matter consists of protons and
18.2 Comparing Atoms. Atomic number. Chapter 18
 As you know, some substances are made up of only one kind of atom and these substances are called elements. You already know something about a number of elements you ve heard of hydrogen, helium, silver,
As you know, some substances are made up of only one kind of atom and these substances are called elements. You already know something about a number of elements you ve heard of hydrogen, helium, silver,
Lecture 09 Nuclear Physics Part 1
 Lecture 09 Nuclear Physics Part 1 Structure and Size of the Nucleus Νuclear Masses Binding Energy The Strong Nuclear Force Structure of the Nucleus Discovered by Rutherford, Geiger and Marsden in 1909
Lecture 09 Nuclear Physics Part 1 Structure and Size of the Nucleus Νuclear Masses Binding Energy The Strong Nuclear Force Structure of the Nucleus Discovered by Rutherford, Geiger and Marsden in 1909
The Advanced Placement Examination in Chemistry. Part I Multiple Choice Questions Part II Free Response Questions Selected Questions from1970 to 2010
 The Advanced Placement Examination in Chemistry Part I Multiple Choice Questions Part II Free Response Questions Selected Questions from1970 to 2010 Atomic Theory and Periodicity Part I 1984 1. Which of
The Advanced Placement Examination in Chemistry Part I Multiple Choice Questions Part II Free Response Questions Selected Questions from1970 to 2010 Atomic Theory and Periodicity Part I 1984 1. Which of
