HEREDITARY ANGIOEDEMA MEDICATION THERAPY
|
|
|
- Alfred King
- 7 years ago
- Views:
Transcription
1 HEREDITARY ANGIOEDEMA MEDICATION THERAPY Coverage for services, procedures, medical devices drugs are dependent upon benefit eligibility as outlined in the member's specific benefit plan. This Medical Coverage Guideline must be read in its entirety to determine coverage eligibility, if any. This Medical Coverage Guideline provides information related to coverage determinations only does not imply that a service or treatment is clinically appropriate or inappropriate. The provider the member are responsible for all decisions regarding the appropriateness of care. Providers should provide BCBSAZ complete medical rationale when requesting any exceptions to these guidelines. The section identified as Description defines or describes a service, procedure, medical device or drug is in no way intended as a statement of medical necessity /or coverage. The section identified as Criteria defines criteria to determine whether a service, procedure, medical device or drug is considered medically necessary or experimental or investigational. State or federal mates, e.g., FEP program, may dictate that any drug, device or biological product approved by the U.S. Food Drug Administration (FDA) may not be considered experimental or investigational thus the drug, device or biological product may be assessed only on the basis of medical necessity. Medical Coverage Guidelines are subject to change as new information becomes available. For purposes of this Medical Coverage Guideline, the terms "experimental" "investigational" are considered to be interchangeable. BLUE CROSS, BLUE SHIELD the Cross Shield Symbols are registered service marks of the Blue Cross Blue Shield Association, an association of independent Blue Cross Blue Shield Plans. All other trademarks service marks contained in this guideline are the property of their respective owners, which are not affiliated with BCBSAZ. Description: Injectable medications used for treating individuals with hereditary angioedema (HAE) include: Berinert (C1 esterase inhibitor, human) Cinryze (C1 esterase inhibitor, human) Firazyr (icatibant) Kalbitor (ecallantide) Ruconest (C1 esterase inhibitor, recombinant) O829.3.docx Page 1 of 5
2 Criteria: See Resources section for FDA-approved dosage. Berinert: FDA approved dosage of Berinert for treatment of acute abdominal, facial or laryngeal angioedema attacks in individuals 5 years of age or older with hereditary angioedema (HAE) is considered medically necessary with documentation of no previous life-threatening immediate hypersensitivity reactions, including anaphylaxis, to Berinert or its components. Berinert for all other indications not previously listed or if above criteria not met is considered 1. Lack of final approval from the Food Drug Administration, 3. Insufficient evidence to support improvement of the net health outcome, than, established alternatives, These indications include, but are not limited to: Prophylaxis against angioedema attacks in individuals with HAE Cinryze: FDA approved dosage of Cinryze for routine prophylaxis against angioedema attacks in individuals 9 years of age or older with hereditary angioedema (HAE) is considered medically necessary with documentation of no previous life-threatening immediate hypersensitivity reactions, including anaphylaxis, to Cinryze or its components. Cinryze for all other indications not previously listed or if above criteria not met is considered 1. Lack of final approval from the Food Drug Administration, 3. Insufficient evidence to support improvement of the net health outcome, than, established alternatives, O829.3.docx Page 2 of 5
3 Criteria: (cont.) Firazyr: FDA approved dosage of Firazyr for treatment of acute attacks of angioedema in individuals 18 years of age older with hereditary angioedema (HAE) is considered medically necessary. Firazyr for all other indications not previously listed or if above criteria not met is considered 1. Lack of final approval from the Food Drug Administration, 3. Insufficient evidence to support improvement of the net health outcome, than, established alternatives, Kalbitor: FDA approved dosage of Kalbitor for treatment of acute attacks of angioedema in individuals 12 years of age older with hereditary angioedema (HAE) is considered medically necessary with documentation of no previous life-threatening immediate hypersensitivity reactions, including anaphylaxis, to Kalbitor or its components. Kalbitor for all other indications not previously listed or if above criteria not met is considered 1. Lack of final approval from the Food Drug Administration, 3. Insufficient evidence to support improvement of the net health outcome, than, established alternatives, O829.3.docx Page 3 of 5
4 Criteria: (cont.) Ruconest: FDA approved dosage of Ruconest for treatment of acute attacks in individuals 13 years of age or older with hereditary angioedema (HAE) is considered medically necessary with documentation of no previous life-threatening immediate hypersensitivity reactions, including anaphylaxis, to Ruconest or its components. Ruconest for all other indications not previously listed or if above criteria not met is considered 1. Lack of final approval from the Food Drug Administration, 3. sufficient evidence to support improvement of the net health outcome, than, established alternatives, These indications include, but are not limited to: The use in HAE individuals with laryngeal attacks Resources: Literature reviewed 09/27/16. We do not include marketing materials, poster boards nonpublished literature in our review. Berinert Package Insert: - FDA-approved indication dosage: Treatment of acute abdominal, facial or laryngeal attacks of hereditary angioedema (HAE) in adult pediatric individuals. The safety efficacy of Berinert for prophylactic therapy have not been established. For intravenous use only. Administer 20 International Units per kg body weight. Inject at a rate of approx. 4mL per minute. O829.3.docx Page 4 of 5
5 Resources: (cont.) Cinryze Package Insert: - FDA-approved indication dosage: Routine prophylaxis against angioedema attacks in individuals 13 years of age or older with hereditary angioedema (HAE). For intravenous use only. Administer 1,000 Units Intravenous every 3 or 4 days. Inject at a rate of approx 1 ml/min. Firazyr Package Insert: - FDA-approved indication dosage: Treatment of acute attacks of hereditary angioedema (HAE) in individuals 18 years of age older. Administer 30 mg injected subcutaneously in the abdominal area. If response is inadequate or symptoms recur, additional injections of 30 mg may be administered at intervals of at least 6 hours. Do not administer more than 3 injections in 24 hours. Kalbitor Package Insert: - FDA-approved indication dosage: Treatment of acute attacks of hereditary angioedema (HAE) in individuals 12 years of age older. Administer 30 mg injected subcutaneously in three 10 mg (1 ml) Injections. If an attack persists, an additional dose of 30 mg may be administered within a 24 hour period. Ruconest Package Insert: - FDA-approved indication dosage: Treatment of acute attacks in individuals 13 years of age or older with hereditary angioedema (HAE). Effectiveness has not been established in HAE patients with laryngeal attacks. For intravenous use only. Administer over approx. 5 minutes. If body weight < 84 kg, administer 50 IU per kg. If body weight > or = to 84 kg, administer 4200IU (2 vials). If the attack symptoms persist, an additional (second) dose can be administered at the recommended dose level. Do not exceed 4200 IU per dose. No more than two doses should be administered within a 24 hour period. O829.3.docx Page 5 of 5
NUVIGIL (armodafinil) oral tablet
 NUVIGIL (armodafinil) oral tablet Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit plan. This Pharmacy Coverage
NUVIGIL (armodafinil) oral tablet Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit plan. This Pharmacy Coverage
Updates to the Alberta Human Services Drug Benefit Supplement
 Updates to the Alberta Human Services Drug Benefit Supplement Effective January 1, 2016 Inquiries should be directed to: Pharmacy Services Alberta Blue Cross 10009 108 Street NW Edmonton AB T5J 3C5 Telephone
Updates to the Alberta Human Services Drug Benefit Supplement Effective January 1, 2016 Inquiries should be directed to: Pharmacy Services Alberta Blue Cross 10009 108 Street NW Edmonton AB T5J 3C5 Telephone
Updates to the Alberta Drug Benefit List. Effective January 1, 2016
 Updates to the Alberta Drug Benefit List Effective January 1, 2016 Inquiries should be directed to: Pharmacy Services Alberta Blue Cross 10009 108 Street NW Edmonton AB T5J 3C5 Telephone Number: (780)
Updates to the Alberta Drug Benefit List Effective January 1, 2016 Inquiries should be directed to: Pharmacy Services Alberta Blue Cross 10009 108 Street NW Edmonton AB T5J 3C5 Telephone Number: (780)
BRACHYTHERAPY FOR TREATMENT OF BREAST CANCER
 BRACHYTHERAPY FOR TREATMENT OF BREAST CANCER Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit plan. This
BRACHYTHERAPY FOR TREATMENT OF BREAST CANCER Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit plan. This
AUTISM SPECTRUM DISORDER TREATMENTS
 AUTISM SPECTRUM DISORDER TREATMENTS Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit plan. This Medical Coverage
AUTISM SPECTRUM DISORDER TREATMENTS Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit plan. This Medical Coverage
SEMI-IMPLANTABLE AND FULLY IMPLANTABLE MIDDLE EAR HEARING AIDS
 Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit plan. This Medical Coverage Guideline must be read in its
Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit plan. This Medical Coverage Guideline must be read in its
Medical Policy An independent licensee of the Blue Cross Blue Shield Association
 Afrezza Page 1 of 6 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: Afrezza (human insulin) Prime Therapeutics will review Prior Authorization requests Prior Authorization
Afrezza Page 1 of 6 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: Afrezza (human insulin) Prime Therapeutics will review Prior Authorization requests Prior Authorization
CONVENTIONAL AND DIGITAL HEARING AIDS
 CONVENTIONAL AND DIGITAL HEARING AIDS Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit plan. This Medical
CONVENTIONAL AND DIGITAL HEARING AIDS Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit plan. This Medical
Pharming Group NV (NYSE Euronext: PHARM)
 Pharming Group NV (NYSE Euronext: PHARM) Sijmen de Vries, MD, MBA Chief Execu?ve Officer Annual General Meeting of Shareholders Leiden, 18 June 2014. 1 Safe harbour statement The informa,on contained in
Pharming Group NV (NYSE Euronext: PHARM) Sijmen de Vries, MD, MBA Chief Execu?ve Officer Annual General Meeting of Shareholders Leiden, 18 June 2014. 1 Safe harbour statement The informa,on contained in
Update on treatment of hereditary angioedema
 Review article Update on treatment of hereditary angioedema Larisa V. Buyantseva, 1 Niti Sardana 2 and Timothy J Craig 3 Summary Background: Hereditary Angioedema (HAE) is a rare disease characterized
Review article Update on treatment of hereditary angioedema Larisa V. Buyantseva, 1 Niti Sardana 2 and Timothy J Craig 3 Summary Background: Hereditary Angioedema (HAE) is a rare disease characterized
ELECTROMYOGRAPHY (EMG), NEEDLE, NERVE CONDUCTION STUDIES (NCS) AND QUANTITATIVE SENSORY TESTING (QST)
 AND QUANTITATIVE SENSORY TESTING (QST) Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit plan. This Medical
AND QUANTITATIVE SENSORY TESTING (QST) Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit plan. This Medical
NasdaqGS: SNTS. September 5, 2012
 NasdaqGS: SNTS September 5, 2012 Safe Harbor This presentation may include forward-looking statements that are based on management s beliefs and assumptions and on information currently available to management.
NasdaqGS: SNTS September 5, 2012 Safe Harbor This presentation may include forward-looking statements that are based on management s beliefs and assumptions and on information currently available to management.
Clinical Commissioning Policy: Treatment of Acute Attacks in Hereditary Angiodema (Adult) April 2013. Reference: NHSCB/B09/P/b
 Clinical Commissioning Policy: Treatment of Acute Attacks in Hereditary Angiodema (Adult) April 2013 Reference: NHS Commissioning Board Clinical Commissioning Policy: Treatment of Acute Attacks in Hereditary
Clinical Commissioning Policy: Treatment of Acute Attacks in Hereditary Angiodema (Adult) April 2013 Reference: NHS Commissioning Board Clinical Commissioning Policy: Treatment of Acute Attacks in Hereditary
PROs and Rare Diseases
 PROs and Rare Diseases Anne Pariser, M.D. Office of New Drugs, Rare Diseases Program Center for Drug Evaluation and Research Food & Drug Administration May 21, 2013 Begin with Goals in Mind We all want
PROs and Rare Diseases Anne Pariser, M.D. Office of New Drugs, Rare Diseases Program Center for Drug Evaluation and Research Food & Drug Administration May 21, 2013 Begin with Goals in Mind We all want
One vial contains 500 U* of C1-inhibitor** After reconstitution the product contains 500 U/5 ml which correspond to a concentration of 100 U/ml.
 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Cetor 100 U/ml powder and solvent for solution for injection 2. QUALITATIVE AND QUANTITATIVE COMPOSITION One vial contains 500 U* of
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Cetor 100 U/ml powder and solvent for solution for injection 2. QUALITATIVE AND QUANTITATIVE COMPOSITION One vial contains 500 U* of
AUBAGIO (teriflunomide) oral tablet
 AUBAGIO (teriflunomide) oral tablet Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit plan. This Pharmacy
AUBAGIO (teriflunomide) oral tablet Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit plan. This Pharmacy
9/16/2014. Anti-Immunoglobulin E (IgE) Omalizumab (Xolair ) Dosing Guidance
 Disclosure Statement of Financial Interest New Therapies for Asthma Including Omalizumab and Anti-Cytokine Therapies Marsha Dangler, PharmD, BCACP Clinical Pharmacy Specialist James H. Quillen VA Medical
Disclosure Statement of Financial Interest New Therapies for Asthma Including Omalizumab and Anti-Cytokine Therapies Marsha Dangler, PharmD, BCACP Clinical Pharmacy Specialist James H. Quillen VA Medical
NasdaqGS: SNTS. July 10, 2013
 NasdaqGS: SNTS July 10, 2013 Safe Harbor This presentation may include forward-looking statements that are based on management s beliefs and assumptions and on information currently available to management.
NasdaqGS: SNTS July 10, 2013 Safe Harbor This presentation may include forward-looking statements that are based on management s beliefs and assumptions and on information currently available to management.
Vaccine Protocol Agreement. Name of Pharmacy: Address: City, State, Zip:
 Vaccine Protocol Agreement Name of Pharmacy: Address: City, State, Zip: This Vaccine Protocol Agreement (the "Protocol") authorizes the Georgia licensed pharmacists (the "Pharmacists") or nurses ( Nurses
Vaccine Protocol Agreement Name of Pharmacy: Address: City, State, Zip: This Vaccine Protocol Agreement (the "Protocol") authorizes the Georgia licensed pharmacists (the "Pharmacists") or nurses ( Nurses
Documentation Summary for Chemotherapy Administration, Nonchemotherapy Injections and Infusions
 Documentation Summary for Chemotherapy Administration, Nonchemotherapy Injections and Infusions Documentation to Support Medical Necessity of Chemotherapy Services Date: April 23, 2012 Source Information:
Documentation Summary for Chemotherapy Administration, Nonchemotherapy Injections and Infusions Documentation to Support Medical Necessity of Chemotherapy Services Date: April 23, 2012 Source Information:
Pharming Group NV (Euronext: PHARM)
 Pharming Group NV (Euronext: PHARM) Sijmen de Vries, MD, MBA Chief Executive Officer Annual General Meeting of Shareholders Leiden, 30 April 2015 1 Safe harbour statement The information contained in this
Pharming Group NV (Euronext: PHARM) Sijmen de Vries, MD, MBA Chief Executive Officer Annual General Meeting of Shareholders Leiden, 30 April 2015 1 Safe harbour statement The information contained in this
Pediatric hereditary angioedema
 The Harvard community has made this article openly available. Please share how this access benefits you. Your story matters. Citation Published Version Accessed Citable Link Terms of Use MacGinnitie, Andrew
The Harvard community has made this article openly available. Please share how this access benefits you. Your story matters. Citation Published Version Accessed Citable Link Terms of Use MacGinnitie, Andrew
Influenza Vaccine Protocol Agreement (O.C.G.A. Section 43-34-26.1)
 Influenza Vaccine Protocol Agreement (O.C.G.A. Section 43-34-26.1) This Influenza Vaccine Protocol Agreement (the "Protocol") authorizes the Georgia licensed pharmacists (the "Pharmacists") or nurses (
Influenza Vaccine Protocol Agreement (O.C.G.A. Section 43-34-26.1) This Influenza Vaccine Protocol Agreement (the "Protocol") authorizes the Georgia licensed pharmacists (the "Pharmacists") or nurses (
Billing with National Drug Codes (NDCs) Frequently Asked Questions
 Billing with National Drug Codes (NDCs) Frequently Asked Questions NDC Overview Converting HCPCS/CPT Units to NDC Units Submitting NDCs on Professional/Ancillary Claims Reimbursement Details For More Information
Billing with National Drug Codes (NDCs) Frequently Asked Questions NDC Overview Converting HCPCS/CPT Units to NDC Units Submitting NDCs on Professional/Ancillary Claims Reimbursement Details For More Information
Original Policy Date
 MP 5.01.20 Tysabri (natalizumab) Medical Policy Section Prescription Drug Issue 12:2013 Original Policy Date 12:2013 Last Review Status/Date Local Policy/12:2013 Return to Medical Policy Index Disclaimer
MP 5.01.20 Tysabri (natalizumab) Medical Policy Section Prescription Drug Issue 12:2013 Original Policy Date 12:2013 Last Review Status/Date Local Policy/12:2013 Return to Medical Policy Index Disclaimer
CSAC/EIA Health Small Group Access+ HMO 15-0 Inpatient Benefit Summary
 CSAC/EIA Health Small Group Access+ HMO 15-0 Inpatient Benefit Summary (Uniform Health Plan Benefits and Coverage Matrix) Blue Shield of California THIS MATRIX IS INTENDED TO BE USED TO HELP YOU COMPARE
CSAC/EIA Health Small Group Access+ HMO 15-0 Inpatient Benefit Summary (Uniform Health Plan Benefits and Coverage Matrix) Blue Shield of California THIS MATRIX IS INTENDED TO BE USED TO HELP YOU COMPARE
How To Take Methotrexate By Injection
 How To Take Methotrexate By Injection 1 how long does it take for methotrexate to work for abortion 2 methotrexate 15 mg hair loss 3 methotrexate injection dosage for rheumatoid arthritis 4 order methotrexate
How To Take Methotrexate By Injection 1 how long does it take for methotrexate to work for abortion 2 methotrexate 15 mg hair loss 3 methotrexate injection dosage for rheumatoid arthritis 4 order methotrexate
Bayer HealthCare, LLC Sharps Disposal Submission to CalRecycle July 1, 2014
 Bayer HealthCare, LLC Sharps Disposal Submission to CalRecycle July 1, 2014 1. A pharmaceutical manufacturer that sells or distributes a medication in California that is usually intended to be self-injected
Bayer HealthCare, LLC Sharps Disposal Submission to CalRecycle July 1, 2014 1. A pharmaceutical manufacturer that sells or distributes a medication in California that is usually intended to be self-injected
January 2012. page 1 / 11
 January 2012 page 1 / 11 Neurology Advising on this article: Jack J. Chen January 5, 2012 Ocrelizumab reduces disease activity in patients with multiple sclerosis Key Point Ocrelizumab (Roche), an investigational
January 2012 page 1 / 11 Neurology Advising on this article: Jack J. Chen January 5, 2012 Ocrelizumab reduces disease activity in patients with multiple sclerosis Key Point Ocrelizumab (Roche), an investigational
DVT/PE Management with Rivaroxaban (Xarelto)
 DVT/PE Management with Rivaroxaban (Xarelto) Rivaroxaban is FDA approved for the acute treatment of DVT and PE and reduction in risk of recurrence of DVT and PE. FDA approved indications: Non valvular
DVT/PE Management with Rivaroxaban (Xarelto) Rivaroxaban is FDA approved for the acute treatment of DVT and PE and reduction in risk of recurrence of DVT and PE. FDA approved indications: Non valvular
Page 1 of 15 Origination Date: 09/14 Revision Date(s): 10/2015, 02/2016 Developed By: Medical Criteria Committee 10/28/2015
 Moda Health Plan, Inc. Medical Necessity Criteria Subject: Actemra (tocilizumab) Page 1 of 15 Origination Date: 09/14 Revision Date(s): 10/2015, 02/2016 Developed By: Medical Criteria Committee 10/28/2015
Moda Health Plan, Inc. Medical Necessity Criteria Subject: Actemra (tocilizumab) Page 1 of 15 Origination Date: 09/14 Revision Date(s): 10/2015, 02/2016 Developed By: Medical Criteria Committee 10/28/2015
Wyckoff Administration Policy on Epinephrine Nurse, Student and or Delegate
 Wyckoff Administration Policy on Epinephrine Nurse, Student and or Delegate It is the policy of this school to apply New Jersey Law N.J.S.A. 18A: 40-12.3-12.6 in the following way: The school will provide
Wyckoff Administration Policy on Epinephrine Nurse, Student and or Delegate It is the policy of this school to apply New Jersey Law N.J.S.A. 18A: 40-12.3-12.6 in the following way: The school will provide
SECTION N: MEDICATIONS. N0300: Injections. Item Rationale Health-related Quality of Life. Planning for Care. Steps for Assessment. Coding Instructions
 SECTION N: MEDICATIONS Intent: The intent of the items in this section is to record the number of days, during the last 7 days (or since admission/reentry if less than 7 days) that any type of injection,
SECTION N: MEDICATIONS Intent: The intent of the items in this section is to record the number of days, during the last 7 days (or since admission/reentry if less than 7 days) that any type of injection,
MANAGEMENT OF COMMON SIDE EFFECTS of INH (Isoniazid), RIF (Rifampin), PZA (Pyrazinamide), and EMB (Ethambutol)
 MANAGEMENT OF COMMON SIDE EFFECTS of INH (Isoniazid), RIF (Rifampin), PZA (Pyrazinamide), and EMB (Ethambutol) 1. Hepatotoxicity: In Active TB Disease a. Background: 1. Among the 4 standard anti-tb drugs,
MANAGEMENT OF COMMON SIDE EFFECTS of INH (Isoniazid), RIF (Rifampin), PZA (Pyrazinamide), and EMB (Ethambutol) 1. Hepatotoxicity: In Active TB Disease a. Background: 1. Among the 4 standard anti-tb drugs,
PHENYLEPHRINE HYDROCHLORIDE INJECTION USP
 PRESCRIBING INFORMATION PHENYLEPHRINE HYDROCHLORIDE INJECTION USP 10 mg/ml Sandoz Canada Inc. Date of Preparation: September 1992 145 Jules-Léger Date of Revision : January 13, 2011 Boucherville, QC, Canada
PRESCRIBING INFORMATION PHENYLEPHRINE HYDROCHLORIDE INJECTION USP 10 mg/ml Sandoz Canada Inc. Date of Preparation: September 1992 145 Jules-Léger Date of Revision : January 13, 2011 Boucherville, QC, Canada
Overview of the BCBSRI Prescription Management Program
 Definitions Overview of the BCBSRI Prescription Management Program DISPENSING GUIDELINES mean: the prescription order or refill must be limited to the quantities authorized by your doctor not to exceed
Definitions Overview of the BCBSRI Prescription Management Program DISPENSING GUIDELINES mean: the prescription order or refill must be limited to the quantities authorized by your doctor not to exceed
The Janssen Biotech Support System. Getting patients started on STELARA
 The Janssen Biotech Support System Getting patients started on STELARA 3 STEPS after identifying patients appropriate for STELARA (ustekinumab) STEP 1: Determine the correct dose STELARA is administered
The Janssen Biotech Support System Getting patients started on STELARA 3 STEPS after identifying patients appropriate for STELARA (ustekinumab) STEP 1: Determine the correct dose STELARA is administered
Committee Approval Date: September 12, 2014 Next Review Date: September 2015
 Medication Policy Manual Policy No: dru361 Topic: Pradaxa, dabigatran Date of Origin: September 12, 2014 Committee Approval Date: September 12, 2014 Next Review Date: September 2015 Effective Date: November
Medication Policy Manual Policy No: dru361 Topic: Pradaxa, dabigatran Date of Origin: September 12, 2014 Committee Approval Date: September 12, 2014 Next Review Date: September 2015 Effective Date: November
Chapter 15. Reconstitution and Dosages Measured in Units. Copyright 2012, 2007 Mosby, Inc., an affiliate of Elsevier Inc. All rights reserved.
 Chapter 15 Reconstitution and Dosages Measured in Units 1 Objectives Calculating drug dosage problems that first require reconstitution of a powdered drug into a liquid form Using a proportion to solve
Chapter 15 Reconstitution and Dosages Measured in Units 1 Objectives Calculating drug dosage problems that first require reconstitution of a powdered drug into a liquid form Using a proportion to solve
NasdaqGS: SNTS. June 03, 2013
 NasdaqGS: SNTS June 03, 2013 Safe Harbor This presentation may include forward-looking statements that are based on management s beliefs and assumptions and on information currently available to management.
NasdaqGS: SNTS June 03, 2013 Safe Harbor This presentation may include forward-looking statements that are based on management s beliefs and assumptions and on information currently available to management.
Session 3 Topics. Argatroban. Argatroban. Drug Use and Adverse Effects. Laboratory Monitoring of Anticoagulant Therapy
 ~~Marshfield Labs Presents~~ Laboratory Monitoring of Anticoagulant Therapy Session 3 of 4 Michael J. Sanfelippo, M.S. Technical Director, Coagulation Services Session 3 Topics Direct Thrombin Inhibitors:
~~Marshfield Labs Presents~~ Laboratory Monitoring of Anticoagulant Therapy Session 3 of 4 Michael J. Sanfelippo, M.S. Technical Director, Coagulation Services Session 3 Topics Direct Thrombin Inhibitors:
HEALTH DEPARTMENT BILLING GUIDELINES
 HEALTH DEPARTMENT BILLING GUIDELINES Acknowledgement: Current Procedural Terminology (CPT ) is copyright 2015 American Medical Association. All Rights Reserved. No fee schedules, basic units, relative
HEALTH DEPARTMENT BILLING GUIDELINES Acknowledgement: Current Procedural Terminology (CPT ) is copyright 2015 American Medical Association. All Rights Reserved. No fee schedules, basic units, relative
XXXXX Petitioner v File No. 121439-001. Issued and entered this 27 TH day of October 2011 by R. Kevin Clinton Commissioner ORDER
 In the matter of STATE OF MICHIGAN DEPARTMENT OF ENERGY, LABOR & ECONOMIC GROWTH OFFICE OF FINANCIAL AND INSURANCE REGULATION Before the Commissioner of Financial and Insurance Regulation XXXXX Petitioner
In the matter of STATE OF MICHIGAN DEPARTMENT OF ENERGY, LABOR & ECONOMIC GROWTH OFFICE OF FINANCIAL AND INSURANCE REGULATION Before the Commissioner of Financial and Insurance Regulation XXXXX Petitioner
Beaumont Hospital Department of Nephrology and Renal Nursing. Guideline for administering Ferinject
 Beaumont Hospital Department of Nephrology and Renal Nursing Guideline Name: Guideline for administering Ferinject Guideline Number: 18 Guideline Version: a Developed By: Louise Kelly CNM 1 Renal Day Care
Beaumont Hospital Department of Nephrology and Renal Nursing Guideline Name: Guideline for administering Ferinject Guideline Number: 18 Guideline Version: a Developed By: Louise Kelly CNM 1 Renal Day Care
Corporate Medical Policy Allergy Immunotherapy (Desensitization)
 Corporate Medical Policy Allergy Immunotherapy (Desensitization) File Name: Origination: Last CAP Review: Next CAP Review: Last Review: allergy_immunotherapy 7/1979 11/2014 11/2015 10/2015 Description
Corporate Medical Policy Allergy Immunotherapy (Desensitization) File Name: Origination: Last CAP Review: Next CAP Review: Last Review: allergy_immunotherapy 7/1979 11/2014 11/2015 10/2015 Description
Blue Cross Blue Shield of Michigan
 Medicare Plus Blue Home infusion therapy Applies to: Medicare Plus Blue PPO SM Medicare Plus Blue Group PPO SM X Both Home infusion therapy Home infusion therapy is the continuous, slow administration
Medicare Plus Blue Home infusion therapy Applies to: Medicare Plus Blue PPO SM Medicare Plus Blue Group PPO SM X Both Home infusion therapy Home infusion therapy is the continuous, slow administration
MEDICATION MANUAL Policy & Procedure
 MEDICATION MANUAL Policy & Procedure TITLE: Section: Initial Management of Anaphylaxis Following Immunization Medication Specific NUMBER: MM 20-005 Date Issued: October 2009 Source: Distribution: Capital
MEDICATION MANUAL Policy & Procedure TITLE: Section: Initial Management of Anaphylaxis Following Immunization Medication Specific NUMBER: MM 20-005 Date Issued: October 2009 Source: Distribution: Capital
Guidelines for anaphylaxis emergency medication (adrenaline [epinephrine] autoinjector) prescription
![Guidelines for anaphylaxis emergency medication (adrenaline [epinephrine] autoinjector) prescription Guidelines for anaphylaxis emergency medication (adrenaline [epinephrine] autoinjector) prescription](/thumbs/33/15971578.jpg) Guidelines for anaphylaxis emergency medication (adrenaline [epinephrine] autoinjector) prescription Introduction The aim of these guidelines is to outline the appropriate prescription of adrenaline (epinephrine)
Guidelines for anaphylaxis emergency medication (adrenaline [epinephrine] autoinjector) prescription Introduction The aim of these guidelines is to outline the appropriate prescription of adrenaline (epinephrine)
STANDARD OPERATING PROCEDURE. Administration of High Dose Muscular Vitamin Supplements for Undergoing Alcohol
 STANDARD OPERATING PROCEDURE Administration of High Dose Muscular Vitamin Supplements for Undergoing Alcohol DOCUMENT CONTROL: Version: 2 Ratified by: Clinical Effectiveness Committee Date ratified: 03
STANDARD OPERATING PROCEDURE Administration of High Dose Muscular Vitamin Supplements for Undergoing Alcohol DOCUMENT CONTROL: Version: 2 Ratified by: Clinical Effectiveness Committee Date ratified: 03
PACKAGE LEAFLET: INFORMATION FOR THE USER. PARACETAMOL MACOPHARMA 10 mg/ml, solution for infusion. Paracetamol
 PACKAGE LEAFLET: INFORMATION FOR THE USER PARACETAMOL MACOPHARMA 10 mg/ml, solution for infusion Paracetamol Read all of this leaflet carefully before you start using this medicine. Keep this leaflet.
PACKAGE LEAFLET: INFORMATION FOR THE USER PARACETAMOL MACOPHARMA 10 mg/ml, solution for infusion Paracetamol Read all of this leaflet carefully before you start using this medicine. Keep this leaflet.
How Is OnabotulinumtoxinA Reimbursed For Chronic Migraine? Impact Of FDA Approval And The New CPT Code
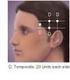 How Is OnabotulinumtoxinA Reimbursed For Chronic Migraine? Impact Of FDA Approval And The New CPT Code Effective January 1, 2013, physicians will be able to report the new CPT code 64615 when performing
How Is OnabotulinumtoxinA Reimbursed For Chronic Migraine? Impact Of FDA Approval And The New CPT Code Effective January 1, 2013, physicians will be able to report the new CPT code 64615 when performing
D( desired ) Q( quantity) X ( amount ) H( have)
 Name: 3 (Pickar) Drug Dosage Calculations Chapter 10: Oral Dosage of Drugs Example 1 The physician orders Lasix 40 mg p.o. daily. You have Lasix in 20 mg, 40 mg, and 80 mg tablets. If you use the 20 mg
Name: 3 (Pickar) Drug Dosage Calculations Chapter 10: Oral Dosage of Drugs Example 1 The physician orders Lasix 40 mg p.o. daily. You have Lasix in 20 mg, 40 mg, and 80 mg tablets. If you use the 20 mg
I B2.4. Design of the patient information leaflet for VariQuin
 (English translation of official Dutch version) I B2.4. Design of the patient information leaflet for VariQuin Information for the Patient: Read this package leaflet carefully when you have some time to
(English translation of official Dutch version) I B2.4. Design of the patient information leaflet for VariQuin Information for the Patient: Read this package leaflet carefully when you have some time to
Pharming Group NV (NYSE Euronext: PHARM)
 Pharming Group NV (NYSE Euronext: PHARM) Board of Management Sijmen de Vries, Chief Executive Officer Bruno Giannetti, Chief Operating Officer Robin Wright, Chief Financial Officer Annual General Meeting
Pharming Group NV (NYSE Euronext: PHARM) Board of Management Sijmen de Vries, Chief Executive Officer Bruno Giannetti, Chief Operating Officer Robin Wright, Chief Financial Officer Annual General Meeting
5.04.20. Perjeta. Perjeta (pertuzumab) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.04.20 Subject: Perjeta Page: 1 of 5 Last Review Date: June 19, 2015 Perjeta Description Perjeta (pertuzumab)
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.04.20 Subject: Perjeta Page: 1 of 5 Last Review Date: June 19, 2015 Perjeta Description Perjeta (pertuzumab)
GUIDELINE FOR ADMINISTRATION OF FENTANYL FOR PAIN RELIEF IN LABOUR
 GUIDELINE FOR ADMINISTRATION OF FENTANYL FOR PAIN RELIEF IN LABOUR INTRODUCTION Intravenous (IV) Fentanyl is a good option for pain management during labour and should be administered in a safe and competent
GUIDELINE FOR ADMINISTRATION OF FENTANYL FOR PAIN RELIEF IN LABOUR INTRODUCTION Intravenous (IV) Fentanyl is a good option for pain management during labour and should be administered in a safe and competent
Hospital Outpatient Coding and Billing Information Sheet for Neulasta and NEUPOGEN
 Hospital Outpatient Coding and Billing Information Sheet for Neulasta and Neulasta Delivery Kit Neulasta Prefilled Syringe For assistance contact 1-844-MYNEULASTA (1-844-696-3852) or visit www.amgenassistonline.com
Hospital Outpatient Coding and Billing Information Sheet for Neulasta and Neulasta Delivery Kit Neulasta Prefilled Syringe For assistance contact 1-844-MYNEULASTA (1-844-696-3852) or visit www.amgenassistonline.com
Don t Let Money Go to Waste. Learn to Bill Discarded Drugs Correctly.
 Don t Let Money Go to Waste. Learn to Bill Discarded Drugs Correctly. The Centers for Medicare & Medicaid Services (CMS) recently released Transmittal 1962 clarifying the use of modifier JW and how to
Don t Let Money Go to Waste. Learn to Bill Discarded Drugs Correctly. The Centers for Medicare & Medicaid Services (CMS) recently released Transmittal 1962 clarifying the use of modifier JW and how to
Antibiotic Prophylaxis for the Prevention of Infective Endocarditis and Prosthetic Joint Infections for Dentists
 PRACTICE ADVISORY SERVICE FAQ 6 Crescent Road, Toronto, ON Canada M4W 1T1 T: 416.961.6555 F: 416.961.5814 Toll Free: 1.800.565.4591 www.rcdso.org Antibiotic Prophylaxis for the Prevention of Infective
PRACTICE ADVISORY SERVICE FAQ 6 Crescent Road, Toronto, ON Canada M4W 1T1 T: 416.961.6555 F: 416.961.5814 Toll Free: 1.800.565.4591 www.rcdso.org Antibiotic Prophylaxis for the Prevention of Infective
Spanish Consensus on Bradykinin-Induced Angioedema (II)
 Spanish Consensus on Bradykinin-Induced Angioedema (II) REVIEWS Consensus Statement on the Diagnosis, Management, and Treatment of Angioedema Mediated by Bradykinin. Part II. Treatment, Follow-up, and
Spanish Consensus on Bradykinin-Induced Angioedema (II) REVIEWS Consensus Statement on the Diagnosis, Management, and Treatment of Angioedema Mediated by Bradykinin. Part II. Treatment, Follow-up, and
Shire to acquire Dyax conference call transcript. Event Date: November 2, 2015
 Shire to acquire Dyax conference call transcript Event Date: November 2, 2015 Company Participants Matthew Osborne, Head of IR Flemming Ornskov, CEO Jeff Poulton, CFO Gustav Christensen, Dyax's President
Shire to acquire Dyax conference call transcript Event Date: November 2, 2015 Company Participants Matthew Osborne, Head of IR Flemming Ornskov, CEO Jeff Poulton, CFO Gustav Christensen, Dyax's President
ALLERGENIC EXTRACT. Prescription Set of Serial Dilutions (or Maintenance Vial (s)) INSTRUCTIONS FOR USE. U.S. Government License No.
 ALLERGENIC EXTRACT Prescription Set of Serial Dilutions (or Maintenance Vial (s)) INSTRUCTIONS FOR USE U.S. Government License No. 308 Revised 07/04 PO Box 800 Lenoir, NC 28645 USA DESCRIPTION This set
ALLERGENIC EXTRACT Prescription Set of Serial Dilutions (or Maintenance Vial (s)) INSTRUCTIONS FOR USE U.S. Government License No. 308 Revised 07/04 PO Box 800 Lenoir, NC 28645 USA DESCRIPTION This set
Prescription Drug Plan
 Prescription Drug Plan The prescription drug plan helps you pay for prescribed medications using either a retail pharmacy or the mail order program. For More Information Administrative details and procedures
Prescription Drug Plan The prescription drug plan helps you pay for prescribed medications using either a retail pharmacy or the mail order program. For More Information Administrative details and procedures
Prior Authorization Guideline
 Prior Authorization Guideline Guideline: PDP IBT Inj - Vivitrol Therapeutic Class: Central Nervous System Agents Therapeutic Sub-Class: Opiate Antagonist Client: 2007 PDP IBT Inj Approval Date: 2/20/2007
Prior Authorization Guideline Guideline: PDP IBT Inj - Vivitrol Therapeutic Class: Central Nervous System Agents Therapeutic Sub-Class: Opiate Antagonist Client: 2007 PDP IBT Inj Approval Date: 2/20/2007
Chemotherapy Administration, Hydration and Therapeutic, Prophylactic, and Diagnostic Injections and Infusions
 Chemotherapy Administration, Hydration and Therapeutic, Prophylactic, and Diagnostic Injections and Infusions Table of Contents: Overview... Professional Services... 2 Services... 4 Overview Chemotherapy
Chemotherapy Administration, Hydration and Therapeutic, Prophylactic, and Diagnostic Injections and Infusions Table of Contents: Overview... Professional Services... 2 Services... 4 Overview Chemotherapy
SECTION 3. Criteria for Special Authorization of Select Drug Products. Section 3 Criteria for Special Authorization of Select Drug Products
 SECTION 3 Criteria for Special Authorization of Select Drug Products Section 3 Criteria for Special Authorization of Select Drug Products CRITERIA FOR SPECIAL AUTHORIZATION OF SELECT DRUG PRODUCTS The
SECTION 3 Criteria for Special Authorization of Select Drug Products Section 3 Criteria for Special Authorization of Select Drug Products CRITERIA FOR SPECIAL AUTHORIZATION OF SELECT DRUG PRODUCTS The
Allergy Emergency Treatment Protocol
 Allergy Emergency Treatment Protocol I. Initial evaluation of possible allergic reaction a. Cease administration of allergenic extracts b. Notify physician c. Record vital signs: blood pressure, pulse,
Allergy Emergency Treatment Protocol I. Initial evaluation of possible allergic reaction a. Cease administration of allergenic extracts b. Notify physician c. Record vital signs: blood pressure, pulse,
Reconstitution of Solutions
 Chapter 12 Reconstitution of Solutions Reconstitution Process of mixing and diluting solutions Some medications supplied in powder form and must be mixed with liquid before administration Parts of Solutions
Chapter 12 Reconstitution of Solutions Reconstitution Process of mixing and diluting solutions Some medications supplied in powder form and must be mixed with liquid before administration Parts of Solutions
GUIDELINES FOR THE USE OF PALIPERIDONE PALMITATE (Xeplion ) Version: 2
 SH CP 29 GUIDELINES FOR THE USE OF PALIPERIDONE PALMITATE (Xeplion ) Summary: Guidelines for the use of paliperidone palmitate (Xeplion ) Keywords (minimum of 5): (To assist policy search engine) Target
SH CP 29 GUIDELINES FOR THE USE OF PALIPERIDONE PALMITATE (Xeplion ) Summary: Guidelines for the use of paliperidone palmitate (Xeplion ) Keywords (minimum of 5): (To assist policy search engine) Target
Pharming Group N.V. BUY. 3 August 2015. Initiation of Coverage. Treating hereditary angioedema, one rabbit at a time.
 Pharming Group N.V. BUY Pharmaceuticals Treating hereditary angioedema, one rabbit at a time Initiation of Coverage Driven by its lead product Ruconest, we believe Dutch biotech company Pharming Group
Pharming Group N.V. BUY Pharmaceuticals Treating hereditary angioedema, one rabbit at a time Initiation of Coverage Driven by its lead product Ruconest, we believe Dutch biotech company Pharming Group
PATIENT S INSTRUCTIONS ON TAKING ALLERGY VIALS TO ANOTHER PHYSICIAN S OFFICE
 PATIENT S INSTRUCTIONS ON TAKING ALLERGY VIALS TO ANOTHER PHYSICIAN S OFFICE 1. Allergy vials should be kept refrigerated at all times. They may stay out in room temperature for about 6 hours. It would
PATIENT S INSTRUCTIONS ON TAKING ALLERGY VIALS TO ANOTHER PHYSICIAN S OFFICE 1. Allergy vials should be kept refrigerated at all times. They may stay out in room temperature for about 6 hours. It would
1.0 Abstract. Title: Real Life Evaluation of Rheumatoid Arthritis in Canadians taking HUMIRA. Keywords. Rationale and Background:
 1.0 Abstract Title: Real Life Evaluation of Rheumatoid Arthritis in Canadians taking HUMIRA Keywords Rationale and Background: This abbreviated clinical study report is based on a clinical surveillance
1.0 Abstract Title: Real Life Evaluation of Rheumatoid Arthritis in Canadians taking HUMIRA Keywords Rationale and Background: This abbreviated clinical study report is based on a clinical surveillance
Example 3: Dilantin-125 is available as 125 mg/5 ml. Dilantin-125, 0.3 g PO, is ordered. How much should the nurse administer to the patient?
 Drug Dosage & IV Rates Calculations Drug Dosage Calculations Drug dosage calculations are required when the amount of medication ordered (or desired) is different from what is available on hand for the
Drug Dosage & IV Rates Calculations Drug Dosage Calculations Drug dosage calculations are required when the amount of medication ordered (or desired) is different from what is available on hand for the
BEDFORDSHIRE AND LUTON JOINT PRESCRIBING COMMITTEE (JPC)
 BEDFORDSHIRE AND LUTON JOINT PRESCRIBING COMMITTEE (JPC) September 2014 Review date: September 2017 Bulletin 203: Tocilizumab (subcutaneous) in combination with methotrexate or as monotherapy for the treatment
BEDFORDSHIRE AND LUTON JOINT PRESCRIBING COMMITTEE (JPC) September 2014 Review date: September 2017 Bulletin 203: Tocilizumab (subcutaneous) in combination with methotrexate or as monotherapy for the treatment
Resource Document 6: Tetanus Immunization. I. Introduction
 Resource Document 6: Tetanus Immunization I. Introduction Attention must be directed to adequate tetanus prophylaxis in the multiply injured patient, particularly if open-extremity trauma is present. The
Resource Document 6: Tetanus Immunization I. Introduction Attention must be directed to adequate tetanus prophylaxis in the multiply injured patient, particularly if open-extremity trauma is present. The
Rh D Immunoglobulin (Anti-D)
 Document Number PD2006_074 Rh D Immunoglobulin (Anti-D) Publication date 29-Aug-2006 Functional Sub group Clinical/ Patient Services - Maternity Clinical/ Patient Services - Medical Treatment Population
Document Number PD2006_074 Rh D Immunoglobulin (Anti-D) Publication date 29-Aug-2006 Functional Sub group Clinical/ Patient Services - Maternity Clinical/ Patient Services - Medical Treatment Population
FDA Update on the H1N1 Flu Vaccine and Antiviral Medications
 FDA Update on the H1N1 Flu Vaccine and Antiviral Medications Beth Fabian Fritsch, R.Ph., M.B.A. Commander, U.S. Public Health Service Health Programs Coordinator Office of Special Health Issues Food and
FDA Update on the H1N1 Flu Vaccine and Antiviral Medications Beth Fabian Fritsch, R.Ph., M.B.A. Commander, U.S. Public Health Service Health Programs Coordinator Office of Special Health Issues Food and
TABLE OF CONTENTS. Home Infusion Therapy Guidelines... 2
 TABLE OF CONTENTS Home Infusion Therapy Guidelines... 2 Services normally considered eligible for benefits... 2 Description... 2 Pre-certification Requirements... 3 Billing Guidelines... 3 Home Infusion
TABLE OF CONTENTS Home Infusion Therapy Guidelines... 2 Services normally considered eligible for benefits... 2 Description... 2 Pre-certification Requirements... 3 Billing Guidelines... 3 Home Infusion
Medication Policy Manual. Topic: Betaseron, Extavia, interferon beta-1b Date of Origin: June 18, 2004
 Medication Policy Manual Policy No: dru108 Topic: Betaseron, Extavia, interferon beta-1b Date of Origin: June 18, 2004 Committee Approval Date: December 12, 2014 Next Review Date: December 2015 Effective
Medication Policy Manual Policy No: dru108 Topic: Betaseron, Extavia, interferon beta-1b Date of Origin: June 18, 2004 Committee Approval Date: December 12, 2014 Next Review Date: December 2015 Effective
ALBERTA IMMUNIZATION POLICY GUIDELINES
 ALBERTA IMMUNIZATION POLICY GUIDELINES Hepatitis Vaccines. Hepatitis A Vaccines Refer to the vaccine product monograph and the Canadian Immunization Guide for further Product monographs are available on
ALBERTA IMMUNIZATION POLICY GUIDELINES Hepatitis Vaccines. Hepatitis A Vaccines Refer to the vaccine product monograph and the Canadian Immunization Guide for further Product monographs are available on
BOTOX Injection (Onabotulinumtoxin A) for Migraine Headaches [Preauthorization Required]
![BOTOX Injection (Onabotulinumtoxin A) for Migraine Headaches [Preauthorization Required] BOTOX Injection (Onabotulinumtoxin A) for Migraine Headaches [Preauthorization Required]](/thumbs/29/13417512.jpg) BOTOX Injection (Onabotulinumtoxin A) for Migraine Headaches [Preauthorization Required] Medical Policy: MP-RX-01-11 Original Effective Date: March 24, 2011 Reviewed: Revised: This policy applies to products
BOTOX Injection (Onabotulinumtoxin A) for Migraine Headaches [Preauthorization Required] Medical Policy: MP-RX-01-11 Original Effective Date: March 24, 2011 Reviewed: Revised: This policy applies to products
National Patient Safety Goals Effective January 1, 2015
 National Patient Safety Goals Effective January 1, 2015 Goal 1 Improve the accuracy of resident identification. NPSG.01.01.01 Long Term are ccreditation Program Medicare/Medicaid ertification-based Option
National Patient Safety Goals Effective January 1, 2015 Goal 1 Improve the accuracy of resident identification. NPSG.01.01.01 Long Term are ccreditation Program Medicare/Medicaid ertification-based Option
Active Clinical Trials
 Active Clinical Trials 1. Genentech Efalizumab Protocol No. ACD4230g Title: A Phase II/III, Randomized, Open-Label, Active-Controlled, Multicenter Trial to Evaluate the Safety and Efficacy of Efalizumab
Active Clinical Trials 1. Genentech Efalizumab Protocol No. ACD4230g Title: A Phase II/III, Randomized, Open-Label, Active-Controlled, Multicenter Trial to Evaluate the Safety and Efficacy of Efalizumab
Prior Authorization Guideline
 Prior Authorization Guideline Guideline: PS Inj - Alimta Therapeutic Class: Antineoplastic Agents Therapeutic Sub-Class: Antifolates Client: PS Inj Approval Date: 8/2/2004 Revision Date: 12/5/2006 I. BENEFIT
Prior Authorization Guideline Guideline: PS Inj - Alimta Therapeutic Class: Antineoplastic Agents Therapeutic Sub-Class: Antifolates Client: PS Inj Approval Date: 8/2/2004 Revision Date: 12/5/2006 I. BENEFIT
Pharming Group NV (RUCONEST) Chemicals
 Pharming Group NV Interim Report January - September Leiden, The Netherlands, 28 October, Biotech company Pharming Group N.V. ( Pharming or the Company ) (Euronext Amsterdam: PHARM) presents its (unaudited)
Pharming Group NV Interim Report January - September Leiden, The Netherlands, 28 October, Biotech company Pharming Group N.V. ( Pharming or the Company ) (Euronext Amsterdam: PHARM) presents its (unaudited)
Aim To identify the signs and symptoms of anaphylaxis and provide emergency care.
 Community Health Policies, Procedures and Guidelines Procedures Aim To identify the signs and symptoms of anaphylaxis and provide emergency care. Equipment Adrenaline autoinjectors: o EpiPen Jr or Anapen
Community Health Policies, Procedures and Guidelines Procedures Aim To identify the signs and symptoms of anaphylaxis and provide emergency care. Equipment Adrenaline autoinjectors: o EpiPen Jr or Anapen
Or. Admin. R. 851-047-0030. OREGON ADMINISTRATIVE RULES Copyright (c) 2009 by The Oregon Secretary of State All rights reserved.
 Or. Admin. R. 851-047-0030 OREGON ADMINISTRATIVE RULES Copyright (c) 2009 by The Oregon Secretary of State All rights reserved. *** This document is current through changes published in the *** *** December
Or. Admin. R. 851-047-0030 OREGON ADMINISTRATIVE RULES Copyright (c) 2009 by The Oregon Secretary of State All rights reserved. *** This document is current through changes published in the *** *** December
Corporate Medical Policy
 Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: infusion_therapy_in_the_home 3/1998 2/2016 2/2017 2/2016 Description of Procedure or Service Home infusion
Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: infusion_therapy_in_the_home 3/1998 2/2016 2/2017 2/2016 Description of Procedure or Service Home infusion
8/6/2010. Name of medication Concentration (1:1,000 or 1mg/1ml) Expiration date
 Learning Objectives: Anaphylaxis & Epinephrine Administration by the EMT Adapted with permission from the Pilot Project for the Administration of Epinephrine by Washington EMTs With successful completion
Learning Objectives: Anaphylaxis & Epinephrine Administration by the EMT Adapted with permission from the Pilot Project for the Administration of Epinephrine by Washington EMTs With successful completion
NHS FORTH VALLEY Guidelines for use of high dose Intravenous Esomeprazole in Adults (Previously called the Hong Kong Protocol)
 NHS FORTH VALLEY Guidelines for use of high dose Intravenous Esomeprazole in Adults (Previously called the Hong Kong Protocol) Date of First Issue 10/05/2010 Approved 16/06/2010 Current Issue Date 18/11/2015
NHS FORTH VALLEY Guidelines for use of high dose Intravenous Esomeprazole in Adults (Previously called the Hong Kong Protocol) Date of First Issue 10/05/2010 Approved 16/06/2010 Current Issue Date 18/11/2015
National Patient Safety Goals Effective January 1, 2015
 National Patient Safety Goals Goal 1 Nursing are enter ccreditation Program Improve the accuracy of patient and resident identification. NPSG.01.01.01 Use at least two patient or resident identifiers when
National Patient Safety Goals Goal 1 Nursing are enter ccreditation Program Improve the accuracy of patient and resident identification. NPSG.01.01.01 Use at least two patient or resident identifiers when
Eliminating Infusion Confusion. Agenda
 Eliminating Infusion Confusion (Drug Administrations in Facility and Non-facility Settings) Presented by Maria Rita Genovese, CPC, PCS & Maryann C. Palmeter, CPC, CENTC 1 Agenda Review of CPT codes What
Eliminating Infusion Confusion (Drug Administrations in Facility and Non-facility Settings) Presented by Maria Rita Genovese, CPC, PCS & Maryann C. Palmeter, CPC, CENTC 1 Agenda Review of CPT codes What
5. Treatment of Asthma in Children
 Treatment of sthma in hildren 5. Treatment of sthma in hildren 5.1 Maintenance Treatment 5.1.1 rugs Inhaled Glucocorticoids. Persistent wheezing in children under the age of three can be controlled with
Treatment of sthma in hildren 5. Treatment of sthma in hildren 5.1 Maintenance Treatment 5.1.1 rugs Inhaled Glucocorticoids. Persistent wheezing in children under the age of three can be controlled with
IV solutions may be given either as a bolus dose or infused slowly through a vein into the plasma at a constant or zero-order rate.
 د.شيماء Biopharmaceutics INTRAVENOUS INFUSION: IV solutions may be given either as a bolus dose or infused slowly through a vein into the plasma at a constant or zero-order rate. The main advantage for
د.شيماء Biopharmaceutics INTRAVENOUS INFUSION: IV solutions may be given either as a bolus dose or infused slowly through a vein into the plasma at a constant or zero-order rate. The main advantage for
Overview of the BCBSRI Prescription Management Program
 Definitions Overview of the BCBSRI Prescription Management Program DISPENSING GUIDELINES mean: the prescription order or refill must be limited to the quantities authorized by your doctor not to exceed
Definitions Overview of the BCBSRI Prescription Management Program DISPENSING GUIDELINES mean: the prescription order or refill must be limited to the quantities authorized by your doctor not to exceed
Perfalgan 10 mg/ml, solution for infusion
 PACKAGE LEAFLET: INFORMATION FOR THE USER Perfalgan 10 mg/ml, solution for infusion Paracetamol Read all of this leaflet carefully before you start using this medicine. Keep this leaflet. You may need
PACKAGE LEAFLET: INFORMATION FOR THE USER Perfalgan 10 mg/ml, solution for infusion Paracetamol Read all of this leaflet carefully before you start using this medicine. Keep this leaflet. You may need
BCCA Protocol Summary for Advanced Therapy for Relapsed Testicular Germ Cell Cancer Using PACLitaxel, Ifosfamide and CISplatin (TIP)
 BCCA Protocol Summary for Advanced Therapy for Relapsed Testicular Germ Cell Cancer Using PACLitaxel, Ifosfamide and CISplatin (TIP) Protocol Code Tumour Group Contact Physician UGUTIP Genitourinary Dr.
BCCA Protocol Summary for Advanced Therapy for Relapsed Testicular Germ Cell Cancer Using PACLitaxel, Ifosfamide and CISplatin (TIP) Protocol Code Tumour Group Contact Physician UGUTIP Genitourinary Dr.
Growth Hormone Therapy
 Growth Hormone Therapy Policy Number: Original Effective Date: MM.04.011 05/21/1999 Line(s) of Business: Current Effective Date: HMO; PPO; QUEST 10/28/2011 Section: Prescription Drugs Place(s) of Service:
Growth Hormone Therapy Policy Number: Original Effective Date: MM.04.011 05/21/1999 Line(s) of Business: Current Effective Date: HMO; PPO; QUEST 10/28/2011 Section: Prescription Drugs Place(s) of Service:
Billing with National Drug Codes (NDCs) Frequently Asked Questions
 Billing with National Drug Codes (NDCs) Frequently Asked Questions NDC Overview Converting HCPCS/CPT Units to NDC Units Submitting NDCs on Professional Claims Reimbursement Details For More Information
Billing with National Drug Codes (NDCs) Frequently Asked Questions NDC Overview Converting HCPCS/CPT Units to NDC Units Submitting NDCs on Professional Claims Reimbursement Details For More Information
Thioctacid 600 T Solution for Injection contains 600 mg alpha-lipoic acid
 Package Leaflet: Information for the User Thioctacid 600 T Solution for Injection contains 600 mg alpha-lipoic acid For use in adults Active substance: Alpha-lipoic acid, Trometamol salt (1:1) Read all
Package Leaflet: Information for the User Thioctacid 600 T Solution for Injection contains 600 mg alpha-lipoic acid For use in adults Active substance: Alpha-lipoic acid, Trometamol salt (1:1) Read all
