Dietary Supplements and INDs. Kris West, J.D., M.S. Office of Research Compliance May 2013
|
|
|
- Bernard Riley
- 7 years ago
- Views:
Transcription
1 Dietary Supplements and INDs Kris West, J.D., M.S. Office of Research Compliance May 2013
2 Dietary Supplements and INDs When does the clinical investigation of a supplement require an IND?
3 FDA Riddle When is a dietary supplement not a dietary supplement?
4 Answer When it s a Drug!
5 Presentation Overview FDA Definitions Regulation of Dietary Supplements & Drugs When dietary supplements become drugs. Dietary supplements in approved drug form. Tools
6 DEFINITIONS Office of Research Compliance
7 FDA Definition of Dietary Supplement Product intended to supplement the diet that contains one or more of the following ingredients: vitamin; mineral; herb; botanical; amino acid; dietary substance to supplement the diet by increasing dietary intake; or concentrate, metabolite, constituent, extract or combination of any of the above ingredients. Labeled as a dietary supplement; not intended for ingestion as a conventional food or sole item of diet. Does not include an item approved as a drug or investigational new drug.
8 FDA Definition of Food Articles used for food or drink for humans or animals and components of those articles. Chewing gum.
9 FDA Definition of Drug Articles in US Pharmacopoeia, official Homoeopathic Pharmacopoeia of the United States, or official National Formulary, and their supplements. Articles intended for use in the diagnosis, cure, mitigation, treatment, or prevention of disease in man or other animals. Articles (other than food) intended to affect the structure or any function of the body of man or other animals. Components of the foregoing articles.
10 Regulation of Drugs & Dietary Supplements
11 Regulation of Drugs To be distributed in interstate commerce, including use in clinical investigations, a drug must be an FDA approved drug or be covered by an IND. Major Pathways to Approval: 505(b)(1) New Drug Application Abbreviated Investigational New Drug Application (Generics) 505(b)(2) New Drug Application for certain changes to approved drugs Over the Counter Monograph System Additional Older Pathway: GRASE Generally Recognized as Safe and Effective
12 Regulation of Dietary Supplements Dietary Supplement Health and Education Act of 1994 No FDA pre-market approval of dietary supplements, except in case of new dietary ingredient. New dietary ingredient: Manufacturer must provide FDA with evidence it relies on to substantiate safety/effectiveness. Manufacturer is responsible for determining that dietary supplements it manufactures or distributes is safe, and claims made are not false or misleading.
13 When Dietary Supplements become Drugs.
14 When are Dietary Supplements Treated as Drugs? If the dietary supplement is claimed to be for use in the diagnosis, cure, mitigation, treatment or prevention of a disease and its associated symptoms, then the dietary supplement is considered to be a drug. Dietary supplements that claim only that they affect the structure or function of the body of humans or animals are considered dietary supplements.
15 Do Clinical Investigations of Dietary Supplement Require an IND? NO IND REQUIRED IF lawfully marketed dietary supplement is studied for how it affects the structure or function of the body. AN IND IS REQUIRED IF the dietary supplement is being studied for whether it cures, treats, mitigates, prevents or diagnoses a disease or associated symptoms.
16 Clues: Dietary Supplement Types that Need an IND When these types of products are used in a clinical investigation, they generally need an IND: Product is part of a class of products intended to treat, diagnose, prevent, mitigate or cure a disease. Product is used as a substitute for a product that treats a disease. Product is used to augment a therapy or drug that is intended to treat, diagnose, prevent, mitigate or cure a disease. Product is a medical food; food for special dietary use; or infant formula.
17 Clues: Study Purposes that Need an IND When a clinical investigation has one of the following purposes, it will need an IND: Dietary supplement (DS) effects on specific disease or disease class. DS effects on signs or symptoms of a disease DS effects on condition associated with a natural state or process if the condition is uncommon or can cause serious harm. DS role in in body s response to a disease or vector of disease. Endpoint that is a disease or symptom of a disease or indication of a disease.
18 Dietary Supplements in Approved Drug Form
19 Dietary Supplements in Approved Drug Form Some dietary supplements are also FDA approved drugs, e.g., prescription form of a vitamin. If study uses a prescription form of a dietary supplement, then regular IND v. IND exempt criteria apply to the study. IND exemption criteria: FDA approved drug No intent to report to FDA as well controlled clinical study in support of new indication or label change, or for prescription drug advertising. No route of administration, dose, patient population or other factor that significantly increases risk or decreases acceptability of risk associated with use of drug. IRB approval Not intended to promote or commercialize the drug.
20 Bottomline If a dietary supplement (includes herbs, botanicals, spices, vitamins or minerals) is used in a clinical investigation that studies its effects on a disease or disease symptoms, then the dietary supplement will be considered to be a drug. Same rule applies to food, except for medical food or infant formula, which require INDs. If the drug used in the study is an unapproved drug (i.e., item does not have, or study does not use, an FDA approved prescription or OTC form), then an IND will be required to conduct the clinical investigation.
21 Tools Office of Research Compliance
22 IRB Tools Decision Tree FAQ FDA Guidances: pdf DocumentsRegulatoryInformation/DietarySupplements/ ucm htm
23 Questions Kris West Office of Research Compliance (404)
FDA s Final Compliance Policy Guide for Marketed Unapproved Drugs Is Agency Enforcement at a Crossroads, or Stuck in a Traffic Circle
 FDA s Final Compliance Policy Guide for Marketed Unapproved Drugs Is Agency Enforcement at a Crossroads, or Stuck in a Traffic Circle Kurt R. Karst Associate Hyman, Phelps & McNamara, P.C. 700 Thirteenth
FDA s Final Compliance Policy Guide for Marketed Unapproved Drugs Is Agency Enforcement at a Crossroads, or Stuck in a Traffic Circle Kurt R. Karst Associate Hyman, Phelps & McNamara, P.C. 700 Thirteenth
Marketed Unapproved Drugs: FDA to Take Immediate Enforcement Action at Any Time, Without Prior Notice
 Marketed Unapproved Drugs: FDA to Take Immediate Enforcement Action at Any Time, Without Prior Notice Kurt R. Karst Hyman, Phelps & McNamara, P.C. 700 Thirteenth Street, N.W., Suite 1200 Washington, D.C.
Marketed Unapproved Drugs: FDA to Take Immediate Enforcement Action at Any Time, Without Prior Notice Kurt R. Karst Hyman, Phelps & McNamara, P.C. 700 Thirteenth Street, N.W., Suite 1200 Washington, D.C.
Enrolled Copy H.B. 128
 1 ELECTRONIC PRESCRIBING ACT 2 2009 GENERAL SESSION 3 STATE OF UTAH 4 Chief Sponsor: Ronda Rudd Menlove 5 Senate Sponsor: Peter C. Knudson 6 Cosponsor: Evan J. Vickers 7 8 LONG TITLE 9 General Description:
1 ELECTRONIC PRESCRIBING ACT 2 2009 GENERAL SESSION 3 STATE OF UTAH 4 Chief Sponsor: Ronda Rudd Menlove 5 Senate Sponsor: Peter C. Knudson 6 Cosponsor: Evan J. Vickers 7 8 LONG TITLE 9 General Description:
Statement of purpose: This bill proposes to implement the recommendations 6
 0 Page of H. Introduced by Representative Masland of Thetford Referred to Committee on Date: Subject: Taxation; sales and use tax; rate of tax; services Statement of purpose: This bill proposes to implement
0 Page of H. Introduced by Representative Masland of Thetford Referred to Committee on Date: Subject: Taxation; sales and use tax; rate of tax; services Statement of purpose: This bill proposes to implement
Table Summary of the California Acupuncture Board Scope of Practice Summary by Kevin McNamee, D.C., L.Ac. with The Supply Center funding
 Table Summary of the California Acupuncture Board Scope of Practice Summary by Kevin McNamee, D.C., L.Ac. with The Supply Center funding A professions scope of practice is a dynamic entity which will continually
Table Summary of the California Acupuncture Board Scope of Practice Summary by Kevin McNamee, D.C., L.Ac. with The Supply Center funding A professions scope of practice is a dynamic entity which will continually
An Overview of the Regulation of Homeopathic Drug Products in the United States
 An Overview of the Regulation of Homeopathic Drug Products in the United States and the Role of the Homoeopathic Pharmacopoeia Convention of the United States J.P. Borneman, Ph.D. President, Homoeopathic
An Overview of the Regulation of Homeopathic Drug Products in the United States and the Role of the Homoeopathic Pharmacopoeia Convention of the United States J.P. Borneman, Ph.D. President, Homoeopathic
Guidance for Industry
 Guidance for Industry IND Exemptions for Studies of Lawfully Marketed Drug or Biological Products for the Treatment of Cancer U.S. Department of Health and Human Services Food and Drug Administration Center
Guidance for Industry IND Exemptions for Studies of Lawfully Marketed Drug or Biological Products for the Treatment of Cancer U.S. Department of Health and Human Services Food and Drug Administration Center
Guidance for Clinical Investigators, Sponsors, and IRBs
 Guidance for Clinical Investigators, Sponsors, and IRBs Investigational New Drug Applications (INDs) Determining Whether Human Research Studies Can Be Conducted Without an IND U.S. Department of Health
Guidance for Clinical Investigators, Sponsors, and IRBs Investigational New Drug Applications (INDs) Determining Whether Human Research Studies Can Be Conducted Without an IND U.S. Department of Health
ST 2004-01 Food Definition, Issued May, 2004; Revised June, 2007 and May, 2015
 Business Tax Division Sales & Use Tax P.O. Box 530 Columbus; Ohio 43216-0530 www.tax.ohio.gov ST 2004-01 Food Definition, Issued May, 2004; Revised June, 2007 and May, 2015 The purpose of this information
Business Tax Division Sales & Use Tax P.O. Box 530 Columbus; Ohio 43216-0530 www.tax.ohio.gov ST 2004-01 Food Definition, Issued May, 2004; Revised June, 2007 and May, 2015 The purpose of this information
Introduction to Homeopathic Medicine for the Pharmacy Professional Posttest/Rationale
 Introduction to Homeopathic Medicine for the Pharmacy Professional Posttest/Rationale 1. Homeopathic medicines are regulated by the U.S. Food and Drug Administration (FDA) under the A. Affordable Care
Introduction to Homeopathic Medicine for the Pharmacy Professional Posttest/Rationale 1. Homeopathic medicines are regulated by the U.S. Food and Drug Administration (FDA) under the A. Affordable Care
From the Text. Definitions. Definitions. Definitions. Definitions. RSPT 2217 Basic Concepts & Principles. RSPT 2217-01 Basic Concepts
 RSPT 2217 & Principles Gardenhire Chapter 1; p. 1-10 From the Text Key Terms and page 2 Orphan Drugs Table 1-1; page 6 Abbreviations Table 1-2; page 8 Aerosolized Agents Table 1-3; page 9 Drug: Pharmacology:
RSPT 2217 & Principles Gardenhire Chapter 1; p. 1-10 From the Text Key Terms and page 2 Orphan Drugs Table 1-1; page 6 Abbreviations Table 1-2; page 8 Aerosolized Agents Table 1-3; page 9 Drug: Pharmacology:
2014 Annual Report on Inspections of Establishments
 2014 Annual Report on Inspections of Establishments Table of Contents Introduction... 2 Data Collection and Definitions... 3 Section 510(h)(6)(A)(i) Number of Domestic and Foreign Establishments Registered
2014 Annual Report on Inspections of Establishments Table of Contents Introduction... 2 Data Collection and Definitions... 3 Section 510(h)(6)(A)(i) Number of Domestic and Foreign Establishments Registered
Robert Jarrin Senior Director, Government Affairs. May 22, 2013
 Robert Jarrin Senior Director, Government Affairs May 22, 2013 1 Section 201(h) FD&C Act "an instrument, apparatus, implement, machine, contrivance, implant, in vitro reagent, or other similar or related
Robert Jarrin Senior Director, Government Affairs May 22, 2013 1 Section 201(h) FD&C Act "an instrument, apparatus, implement, machine, contrivance, implant, in vitro reagent, or other similar or related
Coventry Health Care of Georgia, Inc. Coventry Health and Life Insurance Company
 Coventry Health Care of Georgia, Inc. Coventry Health and Life Insurance Company PRESCRIPTION DRUG RIDER This Prescription Drug Rider is an attachment to the Coventry Health Care of Georgia, Inc. ( Health
Coventry Health Care of Georgia, Inc. Coventry Health and Life Insurance Company PRESCRIPTION DRUG RIDER This Prescription Drug Rider is an attachment to the Coventry Health Care of Georgia, Inc. ( Health
USE BLUE OR BLACK INK ONLY. 1c. ARE YOU THE NEW OWNER OF A PREVIOUSLY REGISTERED FACILITY? Yes O No O
 FDA USE ONLY USE BLUE OR BLACK INK ONLY Date: (MM/DD/YYYY) Section 1 - TYPE OF REGISTRATION 1a. O DOMESTIC REGISTRATION O FOREIGN REGISTRATION 1b. O INITIAL REGISTRATION O UPDATE OF REGISTRATION INFORMATION
FDA USE ONLY USE BLUE OR BLACK INK ONLY Date: (MM/DD/YYYY) Section 1 - TYPE OF REGISTRATION 1a. O DOMESTIC REGISTRATION O FOREIGN REGISTRATION 1b. O INITIAL REGISTRATION O UPDATE OF REGISTRATION INFORMATION
Complementary and Alternative Medicine Products and Their Regulation by the Food and Drug Administration
 Guidance for Industry on Complementary and Alternative Medicine Products and Their Regulation by the Food and Drug Administration DRAFT GUIDANCE This guidance document is being distributed for comment
Guidance for Industry on Complementary and Alternative Medicine Products and Their Regulation by the Food and Drug Administration DRAFT GUIDANCE This guidance document is being distributed for comment
CDRH Regulated Software
 CDRH Regulated Software An Introduction John F. Murray Jr. CDRH Software Compliance Expert CDRH Regulates Software in the following areas Medical Devices Automation of Production Systems Automation of
CDRH Regulated Software An Introduction John F. Murray Jr. CDRH Software Compliance Expert CDRH Regulates Software in the following areas Medical Devices Automation of Production Systems Automation of
The Role of Complementary and Alternative Medicine
 The Role of Complementary and Alternative Medicine By Leslie Mendoza Temple, MD and Cynthia A. Holmes, PhD According to Dr. Andrew Weil, Director of the Integrative Medicine program at the University of
The Role of Complementary and Alternative Medicine By Leslie Mendoza Temple, MD and Cynthia A. Holmes, PhD According to Dr. Andrew Weil, Director of the Integrative Medicine program at the University of
What is a medical device? Medical Devices: Roadmap to Market. Kathryn Klaus, Esq.
 Medical Devices: Roadmap to Market Kathryn Klaus, Esq. The last installment of Regulatory 360 discussed the FDA organization in general where it came from and a broad overview of how it operates, as well
Medical Devices: Roadmap to Market Kathryn Klaus, Esq. The last installment of Regulatory 360 discussed the FDA organization in general where it came from and a broad overview of how it operates, as well
Guidance Notes on How to classify products as proprietary Chinese medicines. (Reference for the Trade)
 Guidance Notes on How to classify products as proprietary Chinese medicines (Reference for the Trade) Aim According to section 119 of the Chinese Medicine Ordinance (Cap. 549, Laws of Hong Kong) ( the
Guidance Notes on How to classify products as proprietary Chinese medicines (Reference for the Trade) Aim According to section 119 of the Chinese Medicine Ordinance (Cap. 549, Laws of Hong Kong) ( the
MOBILE MEDICAL APPLICATIONS
 October 7, 2013 EVOKE HEALTH POINT OF VIEW MOBILE MEDICAL APPLICATIONS FDA GUIDANCE FOR INDUSTRY FOR MORE INFORMATION: Mark McConaghy, VP, Strategy Evoke Health 267.765.4998 mark.mcconaghy@evokehealth.com
October 7, 2013 EVOKE HEALTH POINT OF VIEW MOBILE MEDICAL APPLICATIONS FDA GUIDANCE FOR INDUSTRY FOR MORE INFORMATION: Mark McConaghy, VP, Strategy Evoke Health 267.765.4998 mark.mcconaghy@evokehealth.com
Institute. Talking about Complementary and Alternative Medicine with Health Care Providers: A Workbook and Tips
 Institute National Cancer Office of Cancer Complementary and Alternative Medicine Talking about Complementary and Alternative Medicine with Health Care Providers: A Workbook and Tips U.S. DEPARTMENT OF
Institute National Cancer Office of Cancer Complementary and Alternative Medicine Talking about Complementary and Alternative Medicine with Health Care Providers: A Workbook and Tips U.S. DEPARTMENT OF
How do Patients Take THE GIFT from Mother Earth, BEST FULVIC and Humic and Fulvic Based Supplements?
 How do Patients Take THE GIFT from Mother Earth, BEST FULVIC and Humic and Fulvic Based Supplements? Patients typically start taking THE GIFT from Mother Earth. THE GIFT from Mother Earth is a highly-concentrated
How do Patients Take THE GIFT from Mother Earth, BEST FULVIC and Humic and Fulvic Based Supplements? Patients typically start taking THE GIFT from Mother Earth. THE GIFT from Mother Earth is a highly-concentrated
Regulation and Risk Management of Combination Products
 Regulation and Risk Management of Combination Products Katherine Ulman Associate Scientist Global Regulatory Compliance Manager Dow Corning Healthcare Jim Curtis Senior Specialist, Healthcare Applications
Regulation and Risk Management of Combination Products Katherine Ulman Associate Scientist Global Regulatory Compliance Manager Dow Corning Healthcare Jim Curtis Senior Specialist, Healthcare Applications
Getting Your Claims Right
 Getting Your Claims Right A guide to complying with the Nutrition, Health and Related Claims Standard of the Australia New Zealand Food Standards Code Disclaimer The information presented here has been
Getting Your Claims Right A guide to complying with the Nutrition, Health and Related Claims Standard of the Australia New Zealand Food Standards Code Disclaimer The information presented here has been
Inspections, Compliance, Enforcement, and Criminal Investigations
 Page 1 of 5 Home Inspections, Compliance, Enforcement, and Criminal Investigations Enforcement Actions Warning Letters Inspections, Compliance, Enforcement, and Criminal Investigations Jabones Pardo S.A.
Page 1 of 5 Home Inspections, Compliance, Enforcement, and Criminal Investigations Enforcement Actions Warning Letters Inspections, Compliance, Enforcement, and Criminal Investigations Jabones Pardo S.A.
Script/Notes for PowerPoint Presentation. Medication Use Safety Training for Seniors (MUST for Seniors)
 Script/Notes for PowerPoint Presentation Medication Use Safety Training for Seniors (MUST for Seniors) Instructions: You can use the following script to help you prepare your remarks to your organization
Script/Notes for PowerPoint Presentation Medication Use Safety Training for Seniors (MUST for Seniors) Instructions: You can use the following script to help you prepare your remarks to your organization
What Is CAM? What is CAM? Are complementary medicine and alternative medicine different from each other? What is integrative medicine?
 What Is CAM? There are many terms used to describe approaches to health care that are outside the realm of conventional medicine as practiced in the United States. This fact sheet explains how the National
What Is CAM? There are many terms used to describe approaches to health care that are outside the realm of conventional medicine as practiced in the United States. This fact sheet explains how the National
2016 -- H 7082 S T A T E O F R H O D E I S L A N D
 LC00 01 -- H 0 S T A T E O F R H O D E I S L A N D IN GENERAL ASSEMBLY JANUARY SESSION, A.D. 01 A N A C T RELATING TO FOOD AND DRUGS -- GENETICALLY-ENGINEERED FOODS Introduced By: Representatives Hull,
LC00 01 -- H 0 S T A T E O F R H O D E I S L A N D IN GENERAL ASSEMBLY JANUARY SESSION, A.D. 01 A N A C T RELATING TO FOOD AND DRUGS -- GENETICALLY-ENGINEERED FOODS Introduced By: Representatives Hull,
IACP Comparison of 503A and 503B The Drug Quality and Security Act of 2013
 Exempt Sections within the Food, Drug, and Cosmetic Act If a "traditional compounder", defined as a licensed pharmacist or licensed physician, meets ALL conditions within Section 503A, the compounder is
Exempt Sections within the Food, Drug, and Cosmetic Act If a "traditional compounder", defined as a licensed pharmacist or licensed physician, meets ALL conditions within Section 503A, the compounder is
Outpatient Prescription Drug Benefit
 Outpatient Prescription Drug Benefit GENERAL INFORMATION This supplemental Evidence of Coverage and Disclosure Form is provided in addition to your Member Handbook and Health Plan Benefits and Coverage
Outpatient Prescription Drug Benefit GENERAL INFORMATION This supplemental Evidence of Coverage and Disclosure Form is provided in addition to your Member Handbook and Health Plan Benefits and Coverage
(1) Cancer drug means a prescription drug that is used to treat cancer or the side effects of cancer.
 DEPARTMENT OF PUBLIC HEALTH AND ENVIRONMENT Health Promotion and Disease Prevention Services CANCER DRUG REPOSITORY PROGRAM 6 CCR 1015-10 [Editor s Notes follow the text of the rules at the end of this
DEPARTMENT OF PUBLIC HEALTH AND ENVIRONMENT Health Promotion and Disease Prevention Services CANCER DRUG REPOSITORY PROGRAM 6 CCR 1015-10 [Editor s Notes follow the text of the rules at the end of this
Complementary and Alternative Medicine Products and Their Regulation by the Food and Drug Administration
 Guidance for Industry on Complementary and Alternative Medicine Products and Their Regulation by the Food and Drug Administration DRAFT GUIDANCE This guidance document is being distributed for comment
Guidance for Industry on Complementary and Alternative Medicine Products and Their Regulation by the Food and Drug Administration DRAFT GUIDANCE This guidance document is being distributed for comment
Structure/Function Claims Small Entity Compliance Guide
 Structure/Function Claims Small Entity Compliance Guide The Food and Drug Administration has prepared this Small Entity Compliance Guide in accordance with section 212 of the Small Business Regulatory
Structure/Function Claims Small Entity Compliance Guide The Food and Drug Administration has prepared this Small Entity Compliance Guide in accordance with section 212 of the Small Business Regulatory
Guidance for Industry
 Guidance for Industry Applications Covered by Section 505(b)(2) DRAFT GUIDANCE This guidance document is being distributed for comment purposes only. Comments and suggestions regarding this draft document
Guidance for Industry Applications Covered by Section 505(b)(2) DRAFT GUIDANCE This guidance document is being distributed for comment purposes only. Comments and suggestions regarding this draft document
Guidance for Industry Time and Extent Applications for Nonprescription Drug Products
 Guidance for Industry Time and Extent Applications for Nonprescription Drug Products U.S. Department of Health and Human Services Food and Drug Administration Center for Drug Evaluation and Research (CDER)
Guidance for Industry Time and Extent Applications for Nonprescription Drug Products U.S. Department of Health and Human Services Food and Drug Administration Center for Drug Evaluation and Research (CDER)
Investigational Drugs: Investigational Drugs and Biologics
 : I. PURPOSE The purpose of this policy is to establish procedures for the proper control, storage, use and handling of investigational drugs and biologics to ensure that adequate safeguards are in place
: I. PURPOSE The purpose of this policy is to establish procedures for the proper control, storage, use and handling of investigational drugs and biologics to ensure that adequate safeguards are in place
If a child or adult needs to avoid cows milk, remember that it may be present in many foods, such as:
 Milk Allergy Cows milk allergy affects around 3 6% of (3 6 in every 100) infants and young children who usually start to have symptoms in their first few months. This causes many health problems and is
Milk Allergy Cows milk allergy affects around 3 6% of (3 6 in every 100) infants and young children who usually start to have symptoms in their first few months. This causes many health problems and is
Module 11. How to Evaluate Claims of New Heart Failure Treatments and Cures
 Module 11 How to Evaluate Claims of New Heart Failure Treatments and Cures The Heart Failure Society of America (HFSA) is a non-profit organization of health care professionals and researchers who are
Module 11 How to Evaluate Claims of New Heart Failure Treatments and Cures The Heart Failure Society of America (HFSA) is a non-profit organization of health care professionals and researchers who are
Pre-Questions. Mastering Clinical Research July 29, 2015
 Pre-Questions Mastering Clinical Research July 29, 2015 1. To be compliant with SOP 2.1 Obtaining Informed Consent for greater than minimal risk interventional clinical trial, which Licensed Professional
Pre-Questions Mastering Clinical Research July 29, 2015 1. To be compliant with SOP 2.1 Obtaining Informed Consent for greater than minimal risk interventional clinical trial, which Licensed Professional
Historical document. Guidelines for levels and kinds of evidence to support indications and claims
 Guidelines for levels and kinds of evidence to support indications and claims For Non-Registerable Medicines, including Complementary Medicines, and other Listable Medicines Version 1.1, April 2011 About
Guidelines for levels and kinds of evidence to support indications and claims For Non-Registerable Medicines, including Complementary Medicines, and other Listable Medicines Version 1.1, April 2011 About
Medical Device Software
 Medical Device Software Bakul Patel Senior Policy Advisor 1 Overview Medical devices and software Oversight principles and Current approach Trends, Challenges and opportunities Addressing challenges 2
Medical Device Software Bakul Patel Senior Policy Advisor 1 Overview Medical devices and software Oversight principles and Current approach Trends, Challenges and opportunities Addressing challenges 2
Information Sheet Guidance For IRBs, Clinical Investigators, and Sponsors
 Information Sheet Guidance For IRBs, Clinical Investigators, and Sponsors Frequently Asked Questions About Medical Devices Additional copies are available from: Office of Good Clinical Practice Office
Information Sheet Guidance For IRBs, Clinical Investigators, and Sponsors Frequently Asked Questions About Medical Devices Additional copies are available from: Office of Good Clinical Practice Office
Thinking About Complementary & Alternative Medicine
 Thinking About Complementary & Alternative Medicine A guide for people with cancer PATIENT & family EDUCATION U.S. DEPARTMENT OF HEALTH AND HUMAN SERVICES National Institutes of Health National Cancer
Thinking About Complementary & Alternative Medicine A guide for people with cancer PATIENT & family EDUCATION U.S. DEPARTMENT OF HEALTH AND HUMAN SERVICES National Institutes of Health National Cancer
Clearinghouse Rule 06-051
 Clearinghouse Rule 06-051 STATE OF WISCONSIN CHIROPRACTIC EXAMINING BOARD ------------------------------------------------------------------------------------------------------------ IN THE MATTER OF RULE-MAKING
Clearinghouse Rule 06-051 STATE OF WISCONSIN CHIROPRACTIC EXAMINING BOARD ------------------------------------------------------------------------------------------------------------ IN THE MATTER OF RULE-MAKING
Evaluating New Cancer Treatments
 Evaluating New Cancer Treatments You ve just heard about a possible new cancer treatment and you wonder if it might work for you. Your doctor hasn t mentioned it, but you want to find out more about this
Evaluating New Cancer Treatments You ve just heard about a possible new cancer treatment and you wonder if it might work for you. Your doctor hasn t mentioned it, but you want to find out more about this
Patient access to medical records
 New Law Alert No. 11 April 2002 California edition Patient access to medical records Acupuncture treatment modalities are expanded Cervical cancer screening CME required for pain management Mental health
New Law Alert No. 11 April 2002 California edition Patient access to medical records Acupuncture treatment modalities are expanded Cervical cancer screening CME required for pain management Mental health
Combination Products Regulation in the United States
 Combination Products Regulation in the United States Presenter: Scott Sardeson RAC US/EU 3M Health Care St. Paul, MN USA 1 Presentation Outline Combination products Definitions and Regulations Jurisdiction
Combination Products Regulation in the United States Presenter: Scott Sardeson RAC US/EU 3M Health Care St. Paul, MN USA 1 Presentation Outline Combination products Definitions and Regulations Jurisdiction
As You Age. Aging, Medicines, and Alcohol. A Guide to
 As You Age A Guide to Aging, Medicines, and Alcohol U.S. DEPARTMENT OF HEALTH AND HUMAN SERVICES Substance Abuse and Mental Health Services Administration Food and Drug Administration www.samhsa.gov Take
As You Age A Guide to Aging, Medicines, and Alcohol U.S. DEPARTMENT OF HEALTH AND HUMAN SERVICES Substance Abuse and Mental Health Services Administration Food and Drug Administration www.samhsa.gov Take
Finalized by the Codex Alimentarius Commission July 4, 2005 Guidelines for Vitamin and Mineral Food Supplements PREAMBLE
 Revised Version - Text of final Codex Vitamin and Mineral Guideline. Offered for Amendment on behalf of Natural Solutions Foundation and Citizens Working Group on Codex Finalized by the Codex Alimentarius
Revised Version - Text of final Codex Vitamin and Mineral Guideline. Offered for Amendment on behalf of Natural Solutions Foundation and Citizens Working Group on Codex Finalized by the Codex Alimentarius
Calcium. 1995-2013, The Patient Education Institute, Inc. www.x-plain.com nuf40101 Last reviewed: 02/19/2013 1
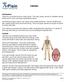 Calcium Introduction Calcium is a mineral found in many foods. The body needs calcium to maintain strong bones and to carry out many important functions. Not having enough calcium can cause many health
Calcium Introduction Calcium is a mineral found in many foods. The body needs calcium to maintain strong bones and to carry out many important functions. Not having enough calcium can cause many health
FOOD FACILITIES REGISTRATION OF. Protecting the U.S. Food Supply. What You Need to Know About
 U.S. Food and Drug Administration Protecting the U.S. Food Supply U.S. Department of Health and Human Services What You Need to Know About REGISTRATION OF FOOD FACILITIES FDA Food Security Information
U.S. Food and Drug Administration Protecting the U.S. Food Supply U.S. Department of Health and Human Services What You Need to Know About REGISTRATION OF FOOD FACILITIES FDA Food Security Information
High Blood Pressure. Dr. Rath s Cellular Health Recommendations for Prevention and Adjunct Therapy
 4 High Blood Pressure Dr. Rath s Cellular Health Recommendations for Prevention and Adjunct Therapy The Facts About High Blood Pressure Dr. Rath s Cellular Health Recommendations: - Documented Health Benefits
4 High Blood Pressure Dr. Rath s Cellular Health Recommendations for Prevention and Adjunct Therapy The Facts About High Blood Pressure Dr. Rath s Cellular Health Recommendations: - Documented Health Benefits
[DOCKET NO.96N-0002] DRAFT
![[DOCKET NO.96N-0002] DRAFT [DOCKET NO.96N-0002] DRAFT](/thumbs/26/7603707.jpg) [DOCKET NO.96N-0002] DRAFT DRAFT DOCUMENT CONCERNING THE REGULATION OF PLACENTAL/UMBILICAL CORD BLOOD STEM CELL PRODUCTS INTENDED FOR TRANSPLANTATION OR FURTHER MANUFACTURE INTO INJECTABLE PRODUCTS DECEMBER,
[DOCKET NO.96N-0002] DRAFT DRAFT DOCUMENT CONCERNING THE REGULATION OF PLACENTAL/UMBILICAL CORD BLOOD STEM CELL PRODUCTS INTENDED FOR TRANSPLANTATION OR FURTHER MANUFACTURE INTO INJECTABLE PRODUCTS DECEMBER,
Excellence in Care: Over-the-Counter Drugs Update
 Excellence in Care: Over-the-Counter Drugs Update Renee Acosta, RPh INDEPENDENT STUDY Health Professions Institute for Continuing Education Austin Community College The Austin Community College Health
Excellence in Care: Over-the-Counter Drugs Update Renee Acosta, RPh INDEPENDENT STUDY Health Professions Institute for Continuing Education Austin Community College The Austin Community College Health
Food supplements. Summary information on legislation relating to the sale of food supplements
 Food supplements Summary information on legislation relating to the sale of food supplements 1 This guidance applies to the whole of the UK and was prepared by the Department of Health in association with
Food supplements Summary information on legislation relating to the sale of food supplements 1 This guidance applies to the whole of the UK and was prepared by the Department of Health in association with
Learn More About Product Labeling
 Learn More About Product Labeling Product label The product label is developed during the formal process of review and approval by regulatory agencies of any medicine or medical product. There are specific
Learn More About Product Labeling Product label The product label is developed during the formal process of review and approval by regulatory agencies of any medicine or medical product. There are specific
What is homeopathy? HOMEOPATHIC. learn more about:
 Natural Relief What is homeopathy? learn more about: The basic principles of homeopathy How to choose the right homeopathic combination medicine How to read Hyland s Drug Facts Why choose homeopathy? Understanding
Natural Relief What is homeopathy? learn more about: The basic principles of homeopathy How to choose the right homeopathic combination medicine How to read Hyland s Drug Facts Why choose homeopathy? Understanding
Insurance coverage of medical foods for treatment of inherited metabolic disorders
 May 18, 2012 Insurance coverage of medical foods for treatment of inherited metabolic disorders Secretary s Advisory Committee on Heritable Disorders in Newborns and Children (SACHDNC) Susan A. Berry,
May 18, 2012 Insurance coverage of medical foods for treatment of inherited metabolic disorders Secretary s Advisory Committee on Heritable Disorders in Newborns and Children (SACHDNC) Susan A. Berry,
Templates. FDA Mobile Medical App Regulations. Your own sub headline This is an example text. Your Logo
 Templates FDA Mobile Medical App Regulations Your own sub headline This is an example text Your Logo FDA Oversight of Medical Devices The latest Guidance from the FDA Tom Richards MD/MS trichards0445@me.com
Templates FDA Mobile Medical App Regulations Your own sub headline This is an example text Your Logo FDA Oversight of Medical Devices The latest Guidance from the FDA Tom Richards MD/MS trichards0445@me.com
Interplay Between FDA Advertising and Promotion Enforcement Activities, Product Liability, and Consumer Fraud Litigation
 Interplay Between FDA Advertising and Promotion Enforcement Activities, Product Liability, and Consumer Fraud Litigation Leslie M. Tector Quarles & Brady LLP September 30, 2014 Objectives Which federal
Interplay Between FDA Advertising and Promotion Enforcement Activities, Product Liability, and Consumer Fraud Litigation Leslie M. Tector Quarles & Brady LLP September 30, 2014 Objectives Which federal
Guidance for Industry Labeling for Bronchodilators: Cold, Cough, Allergy, Bronchodilator,
 Reprinted from FDA s website by Guidance for Industry Labeling for Bronchodilators: Cold, Cough, Allergy, Bronchodilator, Guidance And Antiasthmatic for Drug Industry Products for Over-the-Counter Labeling
Reprinted from FDA s website by Guidance for Industry Labeling for Bronchodilators: Cold, Cough, Allergy, Bronchodilator, Guidance And Antiasthmatic for Drug Industry Products for Over-the-Counter Labeling
Role of the Investigational Drug Services (IDS) in the Management of Investigational Drugs
 Role of the Investigational Drug Services (IDS) in the Management of Investigational Drugs Charlesworth Rae, BS, PharmD, JD Investigational Drug Pharmacist July 2012 1 Outline of Presentation I. Introduction
Role of the Investigational Drug Services (IDS) in the Management of Investigational Drugs Charlesworth Rae, BS, PharmD, JD Investigational Drug Pharmacist July 2012 1 Outline of Presentation I. Introduction
SPECIAL REPORT PERSONAL TRAINING MAY BE TAX DEDUCTIBLE
 SPECIAL REPORT PERSONAL TRAINING MAY BE TAX DEDUCTIBLE PERSONAL TRAINING MAY BE TAX DEDUCTIBLE Does finding tax relief while working out sound too good to be true? Surprisingly, you may be able to financially
SPECIAL REPORT PERSONAL TRAINING MAY BE TAX DEDUCTIBLE PERSONAL TRAINING MAY BE TAX DEDUCTIBLE Does finding tax relief while working out sound too good to be true? Surprisingly, you may be able to financially
CODEX STANDARD FOR FOLLOW-UP FORMULA CODEX STAN 156-1987. This standard applies to the composition and labelling of follow-up formula.
 CODEX STAN 156-1987 Page 1 of 9 CODEX STANDARD FOR FOLLOW-UP FORMULA CODEX STAN 156-1987 1. SCOPE This standard applies to the composition and labelling of follow-up formula. It does not apply to foods
CODEX STAN 156-1987 Page 1 of 9 CODEX STANDARD FOR FOLLOW-UP FORMULA CODEX STAN 156-1987 1. SCOPE This standard applies to the composition and labelling of follow-up formula. It does not apply to foods
Spotlight: Improved Services for Clients with Qualifying Conditions
 Spotlight: Improved Services for Clients with Qualifying Conditions The new food package for clients with s provides a unique opportunity to coordinate better services for clients. Not only does it improve
Spotlight: Improved Services for Clients with Qualifying Conditions The new food package for clients with s provides a unique opportunity to coordinate better services for clients. Not only does it improve
Public Health Service 0,t!,,I I Fgod and Drug Administration, I College Park, MD 20740
 RTMENT OF HEALTH &,HUMAN.SERVICES Public Health Service 0,t!,,I I Fgod and Drug Administration, I College Park, MD 20740 Ryan Baidya, Ph.D., MBA President and CEO r BioZak, Inc. 50 E. St. John Street,
RTMENT OF HEALTH &,HUMAN.SERVICES Public Health Service 0,t!,,I I Fgod and Drug Administration, I College Park, MD 20740 Ryan Baidya, Ph.D., MBA President and CEO r BioZak, Inc. 50 E. St. John Street,
A b i l e n e I n d e p e n d e n t S c h o o l D i s t r i c t ABILENE INDEPENDENT SCHOOL DISTRICT MEDICATION ADMINISTRATION PROCEDURES
 A b i l e n e I n d e p e n d e n t S c h o o l D i s t r i c t 241 Pine Street Abilene, Texas 79601 (325) 677-1444 ABILENE INDEPENDENT SCHOOL DISTRICT MEDICATION ADMINISTRATION PROCEDURES All prescription
A b i l e n e I n d e p e n d e n t S c h o o l D i s t r i c t 241 Pine Street Abilene, Texas 79601 (325) 677-1444 ABILENE INDEPENDENT SCHOOL DISTRICT MEDICATION ADMINISTRATION PROCEDURES All prescription
DRUG INTERACTIONS: WHAT YOU SHOULD KNOW. Council on Family Health
 DRUG INTERACTIONS: WHAT YOU SHOULD KNOW Council on Family Health Drug Interactions There are more opportunities today than ever before to learn about your health and to take better care of yourself. It
DRUG INTERACTIONS: WHAT YOU SHOULD KNOW Council on Family Health Drug Interactions There are more opportunities today than ever before to learn about your health and to take better care of yourself. It
Liver Wellness Increasing Public Awareness of Liver Health
 Liver Wellness Increasing Public Awareness of Liver Health Managing Your Medications Managing Your Medications Medicines can help you feel better and keep you feeling well, but they can be hard to manage.
Liver Wellness Increasing Public Awareness of Liver Health Managing Your Medications Managing Your Medications Medicines can help you feel better and keep you feeling well, but they can be hard to manage.
Millions of Americans suffer from abdominal pain, bloating, constipation and diarrhea. Now new treatments can relieve your pain and discomfort.
 3888-IBS Consumer Bro 5/8/03 10:38 AM Page 1 TAKE THE IBS TEST Do you have recurrent abdominal pain or discomfort? YES NO UNDERSTANDING IRRITABLE BOWEL SYNDROME A Consumer Education Brochure Do you often
3888-IBS Consumer Bro 5/8/03 10:38 AM Page 1 TAKE THE IBS TEST Do you have recurrent abdominal pain or discomfort? YES NO UNDERSTANDING IRRITABLE BOWEL SYNDROME A Consumer Education Brochure Do you often
What Is Complementary and Alternative Medicine (CAM)?
 What Is Complementary and Alternative Medicine (CAM)? There are many terms used to describe approaches to health care that are outside the realm of conventional medicine as practiced in the United States.
What Is Complementary and Alternative Medicine (CAM)? There are many terms used to describe approaches to health care that are outside the realm of conventional medicine as practiced in the United States.
Medicines and You: A Guide for Older Adults
 Medicines and You: A Guide for Older Adults Council on Family Health Provided in cooperation with U.S. Department of Health and Human Services Food and Drug Administration and the Administration on Aging
Medicines and You: A Guide for Older Adults Council on Family Health Provided in cooperation with U.S. Department of Health and Human Services Food and Drug Administration and the Administration on Aging
New Chemical Entity Exclusivity Determinations for Certain Fixed- Combination Drug Products
 New Chemical Entity Exclusivity Determinations for Certain Fixed- Combination Drug Products Guidance for Industry U.S. Department of Health and Human Services Food and Drug Administration Center for Drug
New Chemical Entity Exclusivity Determinations for Certain Fixed- Combination Drug Products Guidance for Industry U.S. Department of Health and Human Services Food and Drug Administration Center for Drug
Claiming to be the Best: Understanding How to Substantiate Your Claims
 Claiming to be the Best: Understanding How to Substantiate Your Claims Michelle C. Jackson MARCH 9, 2012 1 Agenda Brief Overview of Regulatory Agencies and Self- Regulatory Bodies What Is a Claim? Claim
Claiming to be the Best: Understanding How to Substantiate Your Claims Michelle C. Jackson MARCH 9, 2012 1 Agenda Brief Overview of Regulatory Agencies and Self- Regulatory Bodies What Is a Claim? Claim
IN THE UNITED STATES IN 2014,
 Topics of Professional Interest Quality Certification Programs for Dietary Supplements IN THE UNITED STATES IN 2014, dietary supplements were a $36.7 billion industry. 1 The public has easy access to dietary
Topics of Professional Interest Quality Certification Programs for Dietary Supplements IN THE UNITED STATES IN 2014, dietary supplements were a $36.7 billion industry. 1 The public has easy access to dietary
Thomas Conroy, RPh., J.D. Director, Promotion Compliance Global Regulatory Affairs MARCH 11, 2015
 Thomas Conroy, RPh., J.D. Director, Promotion Compliance Global Regulatory Affairs MARCH 11, 2015 All slides and accompanying comments, ideas, arguments and other statements of any kind are personal to
Thomas Conroy, RPh., J.D. Director, Promotion Compliance Global Regulatory Affairs MARCH 11, 2015 All slides and accompanying comments, ideas, arguments and other statements of any kind are personal to
Food and Drug Administration
 Center for Drug Evaluation and Research (CDER) Small Business Assistance Program The Information Source for Regulated Domestic and International Small Pharmaceutical Business CAPT Brenda Stodart, USPHS
Center for Drug Evaluation and Research (CDER) Small Business Assistance Program The Information Source for Regulated Domestic and International Small Pharmaceutical Business CAPT Brenda Stodart, USPHS
SPECIAL COVERAGES. Seniors with Special Support coverage will pay the lesser of the Special Support co-payment or the $20 per prescription.
 SPECIAL COVERAGES A) SENIORS DRUG PLAN As of July 1, 2015, Saskatchewan residents who are 65 years of age and older with a reported income (Line 236) that is $65,515 or less for 2013 will be eligible for
SPECIAL COVERAGES A) SENIORS DRUG PLAN As of July 1, 2015, Saskatchewan residents who are 65 years of age and older with a reported income (Line 236) that is $65,515 or less for 2013 will be eligible for
General Wellness: Policy for Low Risk Devices. Draft Guidance for Industry and Food and Drug Administration Staff
 General Wellness: Policy for Low Risk Devices Draft Guidance for Industry and Food and Drug Administration Staff DRAFT GUIDANCE This guidance document is being distributed for comment purposes only. Document
General Wellness: Policy for Low Risk Devices Draft Guidance for Industry and Food and Drug Administration Staff DRAFT GUIDANCE This guidance document is being distributed for comment purposes only. Document
Miracle Health Claims & Dietary Supplements Add a Dose of Skepticism
 Miracle Health Claims & Dietary Supplements Add a Dose of Skepticism Federal Trade Commission consumer.ftc.gov Miracle Health Claims People spend billions of dollars a year on health-related products
Miracle Health Claims & Dietary Supplements Add a Dose of Skepticism Federal Trade Commission consumer.ftc.gov Miracle Health Claims People spend billions of dollars a year on health-related products
REPORT TO CONGRESS REQUIRED BY SECTION 918 OF THE FEDERAL FOOD, DRUG, AND COSMETIC ACT AS AMENDED BY PUBLIC LAW 111-31
 REPORT TO CONGRESS INNOVATIVE PRODUCTS AND TREATMENTS TO ACHIEVE ABSTINENCE FROM TOBACCO USE, REDUCTIONS IN CONSUMPTION OF TOBACCO, AND REDUCTIONS IN THE HARM ASSOCIATED WITH CONTINUED TOBACCO USE REQUIRED
REPORT TO CONGRESS INNOVATIVE PRODUCTS AND TREATMENTS TO ACHIEVE ABSTINENCE FROM TOBACCO USE, REDUCTIONS IN CONSUMPTION OF TOBACCO, AND REDUCTIONS IN THE HARM ASSOCIATED WITH CONTINUED TOBACCO USE REQUIRED
PMAs, 510(k)s, and Advanced IDE Topics
 PMAs, 510(k)s, and Advanced IDE Topics Kenneth J. Cavanaugh Jr., Ph.D. Scientific Reviewer Division of Cardiovascular Devices Office of Device Evaluation Center for Devices and Radiological Health kenneth.cavanaugh@fda.hhs.gov
PMAs, 510(k)s, and Advanced IDE Topics Kenneth J. Cavanaugh Jr., Ph.D. Scientific Reviewer Division of Cardiovascular Devices Office of Device Evaluation Center for Devices and Radiological Health kenneth.cavanaugh@fda.hhs.gov
Welcome to Your Personal Health Record
 Welcome to Your Personal Health Record What is the Shared Care Plan? The Shared Care Plan is a free, easy-to-use, Personal Health Record that lets you keep track of vital health information in case of
Welcome to Your Personal Health Record What is the Shared Care Plan? The Shared Care Plan is a free, easy-to-use, Personal Health Record that lets you keep track of vital health information in case of
Pharmaceutical Guidelines Overview
 Pharmaceutical Guidelines Overview Elizabeth Scofield Visa Global Brand Protection John Horton President and Founder, LegitScript August 5 & 6, 2014 Visa Public Disclaimer Notice: Please be advised that
Pharmaceutical Guidelines Overview Elizabeth Scofield Visa Global Brand Protection John Horton President and Founder, LegitScript August 5 & 6, 2014 Visa Public Disclaimer Notice: Please be advised that
MAKING INFORMED DECISIONS
 FATIGUE TREATMENTS MAKING INFORMED DECISIONS MS SERVICES SYMPTOM SERIES Fatigue treatments In order to reduce fatigue, many people use a variety of treatments including both medicine. While some people
FATIGUE TREATMENTS MAKING INFORMED DECISIONS MS SERVICES SYMPTOM SERIES Fatigue treatments In order to reduce fatigue, many people use a variety of treatments including both medicine. While some people
Bulimia Nervosa. This reference summary explains bulimia. It covers symptoms and causes of the condition, as well as treatment options.
 Bulimia Nervosa Introduction Bulimia nervosa, or bulimia, is an eating disorder. A person with bulimia eats a large amount of food in a short amount of time. To prevent weight gain, the person then purges.
Bulimia Nervosa Introduction Bulimia nervosa, or bulimia, is an eating disorder. A person with bulimia eats a large amount of food in a short amount of time. To prevent weight gain, the person then purges.
Dietitian, Nutritionist, Nutritional Therapist or Diet Expert? A comprehensive guide to roles and functions.
 Dietitian, Nutritionist, Nutritional Therapist or Diet Expert? A comprehensive guide to roles and functions. 1 Dietitians Choosing the right person to seek help and advice from can sometimes be a confusing
Dietitian, Nutritionist, Nutritional Therapist or Diet Expert? A comprehensive guide to roles and functions. 1 Dietitians Choosing the right person to seek help and advice from can sometimes be a confusing
Cosmetics Regulation in the United States and the European Union:
 Cosmetics Regulation in the United States and the European Union: Different Pathways to the Same Result By Richard Kingham and Lucas E. Beirne Introduction It is sometimes suggested that cosmetic products
Cosmetics Regulation in the United States and the European Union: Different Pathways to the Same Result By Richard Kingham and Lucas E. Beirne Introduction It is sometimes suggested that cosmetic products
Talking With Your Doctor About Multiple Myeloma: A guide to making the most of your healthcare visits
 There s a lot to think about and many important decisions to make when you or someone you care for has multiple myeloma. It s important to have an ongoing conversation with your healthcare team throughout
There s a lot to think about and many important decisions to make when you or someone you care for has multiple myeloma. It s important to have an ongoing conversation with your healthcare team throughout
483.25(i) Nutrition (F325) Surveyor Training: Interpretive Guidance Investigative Protocol
 483.25(i) Nutrition (F325) Surveyor Training: 1 With regard to the revised guidance F325 Nutrition, there have been significant changes. Specifically, F325 and F326 were merged. However, the regulatory
483.25(i) Nutrition (F325) Surveyor Training: 1 With regard to the revised guidance F325 Nutrition, there have been significant changes. Specifically, F325 and F326 were merged. However, the regulatory
PRIOR NOTICE OF IMPORTED FOOD SHIPMENTS A
 U.S. Food and Drug Administration Protecting the U.S. Food Supply U.S. Department of Health and Human Services What You Need to Know About PRIOR NOTICE OF IMPORTED FOOD SHIPMENTS A Small Entity Compliance
U.S. Food and Drug Administration Protecting the U.S. Food Supply U.S. Department of Health and Human Services What You Need to Know About PRIOR NOTICE OF IMPORTED FOOD SHIPMENTS A Small Entity Compliance
NHS GGC Vitamin D Supplementation Frequently asked Questions
 Pharmacy & Prescribing Support Unit NHS GGC Vitamin D Supplementation Frequently asked Questions In February 2012 the Scottish Government issued advice on supplements for groups at risk of vitamin D deficiency.
Pharmacy & Prescribing Support Unit NHS GGC Vitamin D Supplementation Frequently asked Questions In February 2012 the Scottish Government issued advice on supplements for groups at risk of vitamin D deficiency.
Generic and Brand Name Drugs: Understanding the Basics
 Generic and Brand Name Drugs: Understanding the Basics We ve been there. We can help. Joe has just been diagnosed with bipolar disorder. After talking to his doctor, he decides to see a therapist and go
Generic and Brand Name Drugs: Understanding the Basics We ve been there. We can help. Joe has just been diagnosed with bipolar disorder. After talking to his doctor, he decides to see a therapist and go
Circle Of Life SM : Cancer Education and Wellness for American Indian and Alaska Native Communities
 Wellness along the Cancer Journey: American Indian, Alaska Native and Complementary Healing Revised October 2015 Chapter 3: Complementary and Alternative Medicine Alternative & Native Healing Rev. 10.8.15
Wellness along the Cancer Journey: American Indian, Alaska Native and Complementary Healing Revised October 2015 Chapter 3: Complementary and Alternative Medicine Alternative & Native Healing Rev. 10.8.15
FAQs about Warfarin (brand name Coumadin )
 FAQs about Warfarin (brand name Coumadin ) What is warfarin? Warfarin is the most commonly used anticoagulant in the US. An anticoagulant is a drug used to prevent unwanted and harmful blood clots. Although
FAQs about Warfarin (brand name Coumadin ) What is warfarin? Warfarin is the most commonly used anticoagulant in the US. An anticoagulant is a drug used to prevent unwanted and harmful blood clots. Although
LUNG CANCER CLINICAL TRIALS
 UNDERSTANDING LUNG CANCER CLINICAL TRIALS 1-800-298-2436 LungCancerAlliance.org A GUIDE FOR THE PATIENT 1 TABLE OF CONTENTS INTRODUCTION TO CLINICAL TRIALS What Is a Clinical Trial?...4 Types of Clinical
UNDERSTANDING LUNG CANCER CLINICAL TRIALS 1-800-298-2436 LungCancerAlliance.org A GUIDE FOR THE PATIENT 1 TABLE OF CONTENTS INTRODUCTION TO CLINICAL TRIALS What Is a Clinical Trial?...4 Types of Clinical
NEW UPDATES IN OTC PRODUCTS: 2014 DONALD SULLIVAN, PHARMD
 NEW UPDATES IN OTC PRODUCTS: 2014 DONALD SULLIVAN, PHARMD NEW UPDATES IN OTC PRODUCTS: 2014 ACTIVITY DESCRIPTION The program will discuss recently marketed OTC products including their place in therapy,
NEW UPDATES IN OTC PRODUCTS: 2014 DONALD SULLIVAN, PHARMD NEW UPDATES IN OTC PRODUCTS: 2014 ACTIVITY DESCRIPTION The program will discuss recently marketed OTC products including their place in therapy,
Vitamin C Content of Foods
 Vitamin C Content of Foods Experiment #11 Objective: To measure the heat and alkaline stability of vitamin C and its quantity in juices or tablets. Introduction Vitamin C is an essential component of the
Vitamin C Content of Foods Experiment #11 Objective: To measure the heat and alkaline stability of vitamin C and its quantity in juices or tablets. Introduction Vitamin C is an essential component of the
Goals & Objectives. Drug Development & the FDA Pharmacy 309. Outline. An History of Disasters. Be able to describe
 Drug Development & the FDA Pharmacy 309 Tom Hazlet, Pharm.D., Dr.P.H. 616-2732 thazlet@u... Goals & Objectives Be able to describe the major regulatory events in the drug development process the concepts
Drug Development & the FDA Pharmacy 309 Tom Hazlet, Pharm.D., Dr.P.H. 616-2732 thazlet@u... Goals & Objectives Be able to describe the major regulatory events in the drug development process the concepts
