MERCAPTOPURINE (Adults)
|
|
|
- Myrtle Kennedy
- 7 years ago
- Views:
Transcription
1 Shared Care Guideline DRUG: Introduction: MERCAPTOPURINE (Adults) Indication: Disease modifying drug for Crohn s disease and ulcerative colitis. Mercaptopurine is usually used in patients who cannot tolerate azathioprine due to side effects. Licensing Information: The above are unlicensed indications. Formulations: Tablets of 50mg Dosage & administration: Doses are initiated at 0.5mg/kg/day (to the nearest 25mg) and increased slowly at 4-6 week intervals. Doses may adjusted up to mg/kg/day according to patient response and side effects. Contraindications & Warnings: Azathioprine-induced pancreatitis or severe immunosuppression. Azathioprine hypersensitivity use with caution as there may be cross sensitivity between mercaptopurine and azathioprine. A trial of mercaptopurine may be considered appropriate. TPMT (thiopurine methyltransferase) deficiency. Serious infection. Caution: moderate to severe renal insufficiency or impaired liver function. Impaired bone marrow function Considered safe in pregnancy when benefits of treatment outweigh risks of untreated disease. Normal recommendation however is to advise on appropriate contraception in men and women of child bearing potential (as per the manufacturer's recommendations) and discuss any planned pregnancy with the specialist. If unplanned pregnancy occurs continue mercaptopurine and discuss with the specialist. Breast feeding the manufacturer advises women should not breast feed. Please consult specialist in cases of breastfeeding Live attenuated vaccines should be avoided. There may be a reduced response to other vaccines given. Interactions: Allopurinol Avoid if possible. If co-administration of allopurinol is necessary this should ONLY be after consultant initiation and the dose of mercaptopurine should be reduced to one quarter of the original dose with weekly blood monitoring. Febuxostat may increase mercaptopurine levels Warfarin - Closely monitor INR. Live vaccines - Avoid. Name: Mercaptopurine Shared Care Guideline Page 1 of 5
2 Co-trimoxazole (Septrin), trimethoprim, captopril (and possibly other ACE inhibitors) Co-administration of cytostatic or myelosuppressive drugs should be avoided. Co-prescribing of sulfasalazine, mesalazine or olsalazine should be under specialist advice only as increased risk of haematological toxicity. For full list see BNF or SPC at Adverse Effects: In general side effects are considered to be dose related and most will resolve on reduction of mercaptopurine dose or on cessation of treatment. Common Leucopenia, thrombocytopenia, raised transaminases, hepatotoxicity GP to be alert to any oral ulcerations, sore throat, unexplained rash or abnormal bruising or bleeding. Pancreatitis (common in patients with inflammatory bowel disease) Uncommon Nausea (relieved by taking tablets after meals), hypersensitivity, cholestatic jaundice, alopecia, rash and anaemia Very Rare Intestinal ulceration, haematological malignancies (mercaptopurine is mutagenic and carcinogenic). For full list see SPC at of the specialist initiating treatment: General: To assess the suitability of the patient for treatment. As per national, regional and local guidelines, to ensure that the patient/carer has received counselling and understands the therapy, its benefits, limitations, continued monitoring (where applicable), adverse effects, and is aware of actions to take if adverse effects are suspected. Go through patient information leaflet with patient. Inform patients of the long term monitoring requirements and provide a monitoring booklet. Inform the GP of the information provided to the patient. To review the patient as agreed intervals and copy all relevant results to the GP Carry out disease and initial drug monitoring as listed below. Formally hand over to GP by letter and patient informed - send a copy (either electronically or paper copy) of the Shared Care Guideline to the GP and ask whether they are willing to participate in shared care. Prescribing : Issue all prescriptions for 8 weeks minimum until patient is safely established on mercaptopurine and specialist team are satisfied that it is appropriate to share care. Name: Mercaptopurine Shared Care Guideline Page 2 of 5
3 Disease & drug monitoring: Monitor bloods according to schedule: TPMT (thiopurine methyltransferase) At baseline prior to commencing treatment Every 1 to 2 weeks for the first 8 weeks at clinical discretion whilst under secondary care and then as advised by specialist team (see responsibilities of GPs- monitoring). (Note weekly monitoring if allopurinol coadministered) of other prescribers: Discuss shared care arrangement with patient. Support and advise GPs as required. Assess response to treatment and initiate any dose changes as clinically appropriate including discontinuation of treatment. General and Prescribing: To reply to the request for shared care within 2 weeks of receipt of the Consultant letter. Monitor and prescribe as recommended by the specialist. Ensure continued prescribing of mercaptopurine remains clinically appropriate at dose advised by initiating team. The GP will typically be asked to take up monitoring and prescribing of mercaptopurine 8 weeks after treatment has been initiated. Notify Consultant if treatment with mercaptopurine is discontinued. Ensure there are no drug interactions with any other medications initiated in primary care (Note: Avoid live vaccines). Disease & drug monitoring: Carry out drug monitoring as listed and communicate abnormal results to the Specialist. Urgent drug discontinuation/ referral to specialist as clinically appropriate To stop treatment on the advice of the specialist. To refer back to the Specialist if the patient s condition deteriorates. Identify adverse effects to mercaptopurine and report these to the Specialist and where appropriate to the Commission on Human Medicines/MHRA (Yellow card scheme). Unless otherwise stated by the secondary care Specialist, apply the following monitoring frequencies following handover from secondary care: After handover from the specialist Monthly for 4 months then every 3 months (unless dose changed see below or abnormal blood results) (Note weekly monitoring if allopurinol coadministered) On dose increase by hospital, GP to temporarily increase monitoring to week 2,4,8 and then if stable resume previous monitoring regimen unless informed otherwise by initial prescriber Name: Mercaptopurine Shared Care Guideline Page 3 of 5
4 At consultations Ask about oral ulceration, unexplained bruising/bleeding, rash, sore throat of the Patient / Carer: Discontinue mercaptopurine and seek advice from initiating team if: WCC: <3.5 Neutrophils: <2.0 Platelets: <150 AST or ALT: >3 times the normal range Mouth or throat ulceration Unexplained bruising or bleeding Fever, nausea, vomiting or diarrhoea Diffuse alopecia Jaundice Summary: Report any possible side effects to their GP in particular potential signs and symptoms of bone marrow suppression (e.g. unexplained bruising, bleeding, sore throat, infection etc). Practice safe exposure to the sun Ensure they have adequate supply of medication. Attend appointments and take along monitoring book. Ensure appropriate contraception. Avoid breast feeding, unless discussed and agreed with specialist Inform GP urgently if unexpected pregnancy is suspected Disease & drug monitoring: As above contact GP or initiating team if side effects develop (see adverse effects) and attend appointments including those for routine blood tests/investigations Communication: Specialist to GP: The specialist will inform the GP when they have initiated mercaptopurine and when there are any subsequent changes in treatment standard clinic letter. Send a copy (either electronically or paper copy) of the Shared Care Guideline to the GP and ask whether they are willing to participate in shared care. Inform the GP of the information provided to the patient GP to Specialist: To reply to the request for shared care within 2 weeks of receipt of the Consultant letter. Irrespective of whether you accept prescribing responsibility or not, you should inform the consultant of relevant medical information regarding the patient and changes to the patient s medication regime irrespective of indication. Notify Consultant if treatment with mercaptopurine is discontinued. Name: Mercaptopurine Shared Care Guideline Page 4 of 5
5 Contact names & details: If you have any concerns regarding individual patients, see consultant letter for medical contact details or contact one of the following: Name Title/Location Telephone / Bleep / Gastroenterology York Gastroenterology Scarborough General surgery Scarborough IBD Specialist Nurse Drs Robins and Kelly Drs Turnbull and Turvill Drs Smale and Millson Drs Mitchell and Saraj Dr A Muddu Prof J Macfie Miss C McNaught Costs: Drug Tariff Oct 2012 Mercaptopurine 50mg x 25 = References: Yorkshire Regional Guidelines on the Monitoring of Adult Patients on Disease Modifying Drugs (DMARDS) Including Biologic Therapies. 5 th Edition. March 2009 BNF 66 Puri-Nethol SPC accessed on 20 th January 2014 Document Control: This information is not inclusive of all prescribing information and potential adverse effects. Please refer to the SPC (data sheet) or BNF for further prescribing information. The original Microsoft Word file of this document is located on: York Teaching Hospital NHS Foundation Trust Pharmacy Department X:\MEDICINES INFORMATION\Shared Care Guidelines\Approved Shared Care Guidelines\MERCAPTOPURINE Shared Care Guideline V1.1 Shared Care Guidelines are also available electronically via Prepared by: Checked by: Version: Dimah Sweis / Vikki Furneaux Jane Crewe / Stuart Parkes (Pharmacy YH) Clair Ranns (Pharmacist NY and Humber Commissioning Support Unit) 1.1 (Updated February 2014 to ammend information relating to use in pregnancy, disease and drug monitoring tests, and exposure to sun.) Date of Issue / Review: January 2014 (Updated February 2014) Date for next Review: January 2016 Approved by: Drug & Therapeutics Committee, January 2014 Name: Mercaptopurine Shared Care Guideline Page 5 of 5
LEFLUNOMIDE (Adults)
 Shared Care Guideline DRUG: Introduction: LEFLUNOMIDE (Adults) Indication: Disease modifying drug for rheumatoid arthritis and psoriatic arthritis Licensing Information: Disease modifying drug for active
Shared Care Guideline DRUG: Introduction: LEFLUNOMIDE (Adults) Indication: Disease modifying drug for rheumatoid arthritis and psoriatic arthritis Licensing Information: Disease modifying drug for active
INITIATING ORAL AUBAGIO (teriflunomide) THERAPY
 FOR YOUR PATIENTS WITH RELAPSING FORMS OF MS INITIATING ORAL AUBAGIO (teriflunomide) THERAPY WARNING: HEPATOTOXICITY AND RISK OF TERATOGENICITY Severe liver injury including fatal liver failure has been
FOR YOUR PATIENTS WITH RELAPSING FORMS OF MS INITIATING ORAL AUBAGIO (teriflunomide) THERAPY WARNING: HEPATOTOXICITY AND RISK OF TERATOGENICITY Severe liver injury including fatal liver failure has been
Shared Care Guideline for the use of Leflunomide for Rheumatoid Arthritis
 Shared Care Guideline for the use of Leflunomide for Rheumatoid Section 1: Agreement for transfer of prescribing to GP Please sign this form and return it to the named consultant if you are willing to
Shared Care Guideline for the use of Leflunomide for Rheumatoid Section 1: Agreement for transfer of prescribing to GP Please sign this form and return it to the named consultant if you are willing to
Teriflunomide (Aubagio) 14mg once daily tablet
 Teriflunomide (Aubagio) 14mg once daily tablet Exceptional healthcare, personally delivered Your Consultant Neurologist has suggested that you may benefit from treatment with Teriflunomide. The decision
Teriflunomide (Aubagio) 14mg once daily tablet Exceptional healthcare, personally delivered Your Consultant Neurologist has suggested that you may benefit from treatment with Teriflunomide. The decision
Rivaroxaban: Prescribing Guidance for the treatment of provoked venous thromboembolism (VTE)
 Rivaroxaban: Prescribing Guidance for the treatment of provoked venous thromboembolism (VTE) Amber Drug Level 2 Leeds We have started your patient on rivaroxaban for the treatment of provoked VTE (deep
Rivaroxaban: Prescribing Guidance for the treatment of provoked venous thromboembolism (VTE) Amber Drug Level 2 Leeds We have started your patient on rivaroxaban for the treatment of provoked VTE (deep
Rivaroxaban: Amber Drug Guidance for the prevention of stroke and systemic embolism in patients with non-valvular AF
 Leeds Rivaroxaban: Amber Drug Guidance for the prevention of stroke and systemic embolism in patients with non-valvular AF Amber Drug Level 3 (amber drug with monitoring requirements) We have started your
Leeds Rivaroxaban: Amber Drug Guidance for the prevention of stroke and systemic embolism in patients with non-valvular AF Amber Drug Level 3 (amber drug with monitoring requirements) We have started your
Maintenance of abstinence in alcohol dependence
 Shared Care Guideline for Prescription and monitoring of Acamprosate Calcium Author(s)/Originator(s): (please state author name and department) Dr Daly - Consultant Psychiatrist, Alcohol Services Dr Donnelly
Shared Care Guideline for Prescription and monitoring of Acamprosate Calcium Author(s)/Originator(s): (please state author name and department) Dr Daly - Consultant Psychiatrist, Alcohol Services Dr Donnelly
Donepezil (Aricept ), Galantamine (Reminyl XL ), Rivastigmine (Exelon ) and Memantine (Ebixa )
 Donepezil (Aricept ), Galantamine (Reminyl XL ), Rivastigmine (Exelon ) and Memantine (Ebixa ) ESCA: For the treatment of Alzheimer s disease. SECONDARY CARE SECTION TO BE COMPLETED BY INITIATING DOCTOR
Donepezil (Aricept ), Galantamine (Reminyl XL ), Rivastigmine (Exelon ) and Memantine (Ebixa ) ESCA: For the treatment of Alzheimer s disease. SECONDARY CARE SECTION TO BE COMPLETED BY INITIATING DOCTOR
Dabigatran: Amber Drug Guidance for the prevention of stroke and systemic embolism in patients with non-valvular AF
 Leeds Dabigatran: Amber Drug Guidance for the prevention of stroke and systemic embolism in patients with non-valvular AF Amber Drug Level 3 (amber drug with monitoring requirements) We have started your
Leeds Dabigatran: Amber Drug Guidance for the prevention of stroke and systemic embolism in patients with non-valvular AF Amber Drug Level 3 (amber drug with monitoring requirements) We have started your
Adjunctive psychosocial intervention. Conditions requiring dose reduction. Immediate, peak plasma concentration is reached within 1 hour.
 Shared Care Guideline for Prescription and monitoring of Naltrexone Hydrochloride in alcohol dependence Author(s)/Originator(s): (please state author name and department) Dr Daly - Consultant Psychiatrist,
Shared Care Guideline for Prescription and monitoring of Naltrexone Hydrochloride in alcohol dependence Author(s)/Originator(s): (please state author name and department) Dr Daly - Consultant Psychiatrist,
Leflunomide Leflunomide
 Drug information Leflunomide Leflunomide This leaflet provides information on leflunomide and will answer any questions you have about the treatment. Arthritis Research UK produce and print our booklets
Drug information Leflunomide Leflunomide This leaflet provides information on leflunomide and will answer any questions you have about the treatment. Arthritis Research UK produce and print our booklets
Naltrexone Shared Care Guideline for the treatment of alcohol dependence and opioid dependance
 Naltrexone Shared Care Guideline for the treatment of alcohol dependence and opioid dependance Introduction Indication/Licensing information: Naltrexone is licensed for use as an additional therapy, within
Naltrexone Shared Care Guideline for the treatment of alcohol dependence and opioid dependance Introduction Indication/Licensing information: Naltrexone is licensed for use as an additional therapy, within
DERBYSHIRE JOINT AREA PRESCRIBING COMMITTEE (JAPC) SHARED CARE AGREEMENT FRAMEWORK
 DERBYSHIRE JOINT AREA PRESCRIBING COMMITTEE (JAPC) SHARED CARE AGREEMENT FRAMEWORK ACAMPROSATE CALCIUM (Campral EC) for alcohol abstinence ESCA: Adjunct in the treatment of chronic alcohol dependence (under
DERBYSHIRE JOINT AREA PRESCRIBING COMMITTEE (JAPC) SHARED CARE AGREEMENT FRAMEWORK ACAMPROSATE CALCIUM (Campral EC) for alcohol abstinence ESCA: Adjunct in the treatment of chronic alcohol dependence (under
Trust Guideline. for. Shared Care Guidelines for the Use of Leflunomide (Arava )
 A guideline recommended for use Trust Guideline for Shared Care Guidelines for the Use of Leflunomide (Arava ) In: By: For: East and North Herts NHS Trust (ENHT) East and North Herts PCT/ENHCCG Consultants
A guideline recommended for use Trust Guideline for Shared Care Guidelines for the Use of Leflunomide (Arava ) In: By: For: East and North Herts NHS Trust (ENHT) East and North Herts PCT/ENHCCG Consultants
METHOTREXATE Rheumatoid arthritis, Psoriasis, Crohn s Disease & Ulcerative Colitis DOCUMENT TO BE SCANNED INTO ELECTRONIC RECORDS AND FILED IN NOTES
 NHS ONEL and BHRuT NHS Trust Shared Care Guidelines METHOTREXATE Rheumatoid arthritis, Psoriasis, Crohn s Disease & Ulcerative Colitis DOCUMENT TO BE SCANNED INTO ELECTRONIC RECORDS AND FILED IN NOTES
NHS ONEL and BHRuT NHS Trust Shared Care Guidelines METHOTREXATE Rheumatoid arthritis, Psoriasis, Crohn s Disease & Ulcerative Colitis DOCUMENT TO BE SCANNED INTO ELECTRONIC RECORDS AND FILED IN NOTES
Methotrexate treatment
 Methotrexate treatment Oral methotrexate pre-treatment patient information leaflet This leaflet has been prepared to support information given to you as part of your discussions with the doctor, nurse
Methotrexate treatment Oral methotrexate pre-treatment patient information leaflet This leaflet has been prepared to support information given to you as part of your discussions with the doctor, nurse
METHOTREXATE TREATMENT
 METHOTREXATE TREATMENT This leaflet has been prepared to support information given to you as part of your discussions with the doctor, nurse or pharmacist before you start treatment with oral methotrexate.
METHOTREXATE TREATMENT This leaflet has been prepared to support information given to you as part of your discussions with the doctor, nurse or pharmacist before you start treatment with oral methotrexate.
Patient Group Direction Hospital: Bristol Royal Infirmary Department: UHBristol Thrombosis Service University Hospitals Bristol NHS Foundation Trust.
 Patient Group Direction Hospital: Bristol Royal Infirmary Department: UHBristol Thrombosis Service University Hospitals Bristol NHS Foundation Trust. This Patient Group Direction (PGD) has been written
Patient Group Direction Hospital: Bristol Royal Infirmary Department: UHBristol Thrombosis Service University Hospitals Bristol NHS Foundation Trust. This Patient Group Direction (PGD) has been written
A guide to the accelerated elimination procedure
 A guide to the accelerated elimination procedure Prescribing information and information on adverse event reporting can be found on the last page. What is the accelerated elimination procedure? An accelerated
A guide to the accelerated elimination procedure Prescribing information and information on adverse event reporting can be found on the last page. What is the accelerated elimination procedure? An accelerated
**Form 1: - Consultant Copy** Telephone Number: Fax Number: Email: Author: Dr Bernard Udeze Pharmacist: Claire Ault Date of issue July 2011
 Effective Shared Care Agreement for the treatment of Dementia in Alzheimer s Disease Donepezil tablets / orodispersible tablets (Aricept / Aricept Evess ) These forms (1 and 2) are to be completed by both
Effective Shared Care Agreement for the treatment of Dementia in Alzheimer s Disease Donepezil tablets / orodispersible tablets (Aricept / Aricept Evess ) These forms (1 and 2) are to be completed by both
GMMMG Interface Prescribing Subgroup. Shared Care Template
 GMMMG Interface Prescribing Subgroup Shared Care Template Shared Care Guideline for Selective Serotonin Reuptake Inhibitors (SSRIs) for the treatment of Obsessive Compulsive Disorder (OCD) and Body Dysmorphic
GMMMG Interface Prescribing Subgroup Shared Care Template Shared Care Guideline for Selective Serotonin Reuptake Inhibitors (SSRIs) for the treatment of Obsessive Compulsive Disorder (OCD) and Body Dysmorphic
Allopurinol Allopurinol
 Drug information Allopurinol Allopurinol This leaflet provides information on allopurinol and will answer any questions you have about the treatment. Arthritis Research UK produce and print our booklets
Drug information Allopurinol Allopurinol This leaflet provides information on allopurinol and will answer any questions you have about the treatment. Arthritis Research UK produce and print our booklets
There is a risk of renal impairment in dehydrated children and adolescents.
 PACKAGE LEAFLET: INFORMATION FOR THE USER MELFEN 200mg FILM-COATED TABLETS MELFEN 400mg FILM-COATED TABLETS Ibuprofen Read all of this leaflet carefully before you start taking this medicine because it
PACKAGE LEAFLET: INFORMATION FOR THE USER MELFEN 200mg FILM-COATED TABLETS MELFEN 400mg FILM-COATED TABLETS Ibuprofen Read all of this leaflet carefully before you start taking this medicine because it
Dorset Cardiac Centre
 P a g e 1 Dorset Cardiac Centre Patients with Atrial Fibrillation/Flutter undergoing DC Cardioversion or Ablation procedures- Guidelines for Novel Oral Anti-coagulants (NOACS) licensed for this use February
P a g e 1 Dorset Cardiac Centre Patients with Atrial Fibrillation/Flutter undergoing DC Cardioversion or Ablation procedures- Guidelines for Novel Oral Anti-coagulants (NOACS) licensed for this use February
Breast Pathway Group FEC 60 (Fluorouracil / Epirubicin / Cyclophosphamide) in Early Breast Cancer in Elderly / Frail
 Breast Pathway Group FEC 60 (Fluorouracil / Epirubicin / Cyclophosphamide) in Early Breast Cancer in Elderly / Frail Indication: Neoadjuvant or adjuvant therapy for elderly and frail patients with breast
Breast Pathway Group FEC 60 (Fluorouracil / Epirubicin / Cyclophosphamide) in Early Breast Cancer in Elderly / Frail Indication: Neoadjuvant or adjuvant therapy for elderly and frail patients with breast
Shared care protocol for the management of patients with Rheumatoid Arthritis treated with disease modifying antirheumatic drugs (DMARDs)
 Tameside Hospital NHS Foundation Trust and NHS Tameside and Glossop Shared care protocol for the management of patients with Rheumatoid Arthritis treated with disease modifying antirheumatic drugs (DMARDs)
Tameside Hospital NHS Foundation Trust and NHS Tameside and Glossop Shared care protocol for the management of patients with Rheumatoid Arthritis treated with disease modifying antirheumatic drugs (DMARDs)
Shared Care Guideline-Use of Donepezil, Galantamine, Rivastigmine and Memantine in Dementia
 Shared Care Guideline-Use of Donepezil, Galantamine, Rivastigmine and Memantine in Dementia Version: 3.0 Ratified by: Medicines Committee Date ratified: 16 th November 2011 Name of originator/author: James
Shared Care Guideline-Use of Donepezil, Galantamine, Rivastigmine and Memantine in Dementia Version: 3.0 Ratified by: Medicines Committee Date ratified: 16 th November 2011 Name of originator/author: James
Paracetamol apollo +9191 46 950 950. Paracetamol apollo +9191 46 950 950. Paracetamol
 Paracetamol apollo +9191 46 950 950 Paracetamol apollo +9191 46 950 950 Paracetamol CAS Number : 103-90-2 Molecular Weight : 151.17 g/mol Molecular Formula : C8H9NO2 Systematic (IUPAC) : N-(4- hydroxyphenyl)ethanamide
Paracetamol apollo +9191 46 950 950 Paracetamol apollo +9191 46 950 950 Paracetamol CAS Number : 103-90-2 Molecular Weight : 151.17 g/mol Molecular Formula : C8H9NO2 Systematic (IUPAC) : N-(4- hydroxyphenyl)ethanamide
IMPORTANT DRUG WARNING Regarding Mycophenolate-Containing Products
 Dear Healthcare Provider: Mycophenolate REMS (Risk Evaluation and Mitigation Strategy) has been mandated by the FDA (Food and Drug Administration) due to postmarketing reports showing that exposure to
Dear Healthcare Provider: Mycophenolate REMS (Risk Evaluation and Mitigation Strategy) has been mandated by the FDA (Food and Drug Administration) due to postmarketing reports showing that exposure to
Lung Pathway Group Nintedanib (Vargatef) in advanced Non-Small Cell Lung Cancer (NSCLC)
 Lung Pathway Group Nintedanib (Vargatef) in advanced Non-Small Cell Lung Cancer (NSCLC) Indication: In combination with docetaxel in locally advanced, metastatic or locally recurrent NSCLC of adenocarcinoma
Lung Pathway Group Nintedanib (Vargatef) in advanced Non-Small Cell Lung Cancer (NSCLC) Indication: In combination with docetaxel in locally advanced, metastatic or locally recurrent NSCLC of adenocarcinoma
Inpatient Anticoagulation Safety. To provide safe and effective anticoagulation therapy through a collaborative approach.
 Inpatient Anticoagulation Safety Purpose: Policy: To provide safe and effective anticoagulation therapy through a collaborative approach. Upon the written order of a physician, Heparin, Low Molecular Weight
Inpatient Anticoagulation Safety Purpose: Policy: To provide safe and effective anticoagulation therapy through a collaborative approach. Upon the written order of a physician, Heparin, Low Molecular Weight
Atrial Fibrillation and Anticoagulants
 York Teaching Hospital NHS Foundation Trust Atrial Fibrillation and Anticoagulants A guide to your diagnosis and treatment Information for patients, relatives and carers For more information, please contact:
York Teaching Hospital NHS Foundation Trust Atrial Fibrillation and Anticoagulants A guide to your diagnosis and treatment Information for patients, relatives and carers For more information, please contact:
Temozolomide (oral) with concurrent radiotherapy to the brain
 Temozolomide (oral) with concurrent radiotherapy to the brain Temozolomide (oral) with concurrent radiotherapy to the brain This leaflet is offered as a guide to you and your family. You will find it useful
Temozolomide (oral) with concurrent radiotherapy to the brain Temozolomide (oral) with concurrent radiotherapy to the brain This leaflet is offered as a guide to you and your family. You will find it useful
Rivaroxaban shared care guidelines for the prevention of stroke and embolism in adult patients with nonvalvular atrial fibrillation.
 South West Essex Rivaroxaban Shared Care Guideline (SCG) Rivaroxaban shared care guidelines for the prevention of stroke and embolism in adult patients with nonvalvular atrial fibrillation. Introduction
South West Essex Rivaroxaban Shared Care Guideline (SCG) Rivaroxaban shared care guidelines for the prevention of stroke and embolism in adult patients with nonvalvular atrial fibrillation. Introduction
PACKAGE LEAFLET: INFORMATION FOR THE USER. Dalacin C 150 mg Capsules. clindamycin hydrochloride. Dalacin C 150mg Capsules clindamycin hydrochloride
 PACKAGE LEAFLET: INFORMATION FOR THE USER PFIZER Dalacin C 150 mg Capsules clindamycin hydrochloride Dalacin C 150mg Capsules clindamycin hydrochloride PFIZER Read all of this leaflet carefully before
PACKAGE LEAFLET: INFORMATION FOR THE USER PFIZER Dalacin C 150 mg Capsules clindamycin hydrochloride Dalacin C 150mg Capsules clindamycin hydrochloride PFIZER Read all of this leaflet carefully before
5.07.09. Aubagio. Aubagio (teriflunomide) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.07.09 Subject: Aubagio Page: 1 of 6 Last Review Date: December 5, 2014 Aubagio Description Aubagio (teriflunomide)
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.07.09 Subject: Aubagio Page: 1 of 6 Last Review Date: December 5, 2014 Aubagio Description Aubagio (teriflunomide)
Introduction. Background to this event. Raising awareness 09/11/2015
 Introduction Primary Care Medicines Governance HSCB Background to this event New class of medicines Availability of training Increasing volume of prescriptions Reports of medication incidents Raising awareness
Introduction Primary Care Medicines Governance HSCB Background to this event New class of medicines Availability of training Increasing volume of prescriptions Reports of medication incidents Raising awareness
2. Characteristics of staff Qualifications required. Additional requirements. Continued education & training requirements
 Patient Group Direction The supply of Azithromycin 1g as a single dose by accredited Community Pharmacists to patients in receipt of a positive test result to Chlamydia trachomatis, and treatment of their
Patient Group Direction The supply of Azithromycin 1g as a single dose by accredited Community Pharmacists to patients in receipt of a positive test result to Chlamydia trachomatis, and treatment of their
Latest advice for medicines users The monthly newsletter from the MHRA and its independent advisor the Commission on Human Medicines
 Latest advice for medicines users The monthly newsletter from the MHRA and its independent advisor the Commission on Human Medicines Volume 6, Issue 10, May 2013 Drug safety advice Yellow card scheme Stop
Latest advice for medicines users The monthly newsletter from the MHRA and its independent advisor the Commission on Human Medicines Volume 6, Issue 10, May 2013 Drug safety advice Yellow card scheme Stop
MS Treatments Aubagio TM
 1 MSology Essentials Series Aubagio TM (teriflunomide) Developed by MSology with the invaluable assistance of multiple sclerosis nurse advisors: Bonnie Blain Central Alberta MS Clinic, Red Deer, Alberta
1 MSology Essentials Series Aubagio TM (teriflunomide) Developed by MSology with the invaluable assistance of multiple sclerosis nurse advisors: Bonnie Blain Central Alberta MS Clinic, Red Deer, Alberta
PATIENT INFORMATION LEAFLET. CEFALEXIN 250 mg AND 500 mg CAPSULES CEFALEXIN
 PATIENT INFORMATION LEAFLET CEFALEXIN 250 mg AND 500 mg CAPSULES CEFALEXIN Read all of this leaflet carefully before you start taking this medicine. - Keep this leaflet. You may need to read it again.
PATIENT INFORMATION LEAFLET CEFALEXIN 250 mg AND 500 mg CAPSULES CEFALEXIN Read all of this leaflet carefully before you start taking this medicine. - Keep this leaflet. You may need to read it again.
Hull & East Riding Prescribing Committee
 Hull & East Riding Prescribing Committee Guideline on Prescribing of Gonadorelin (GnRH) Analogues and Progesterone Receptor Modulators in treatment of Endometriosis or prior to Endometrial Ablation, or
Hull & East Riding Prescribing Committee Guideline on Prescribing of Gonadorelin (GnRH) Analogues and Progesterone Receptor Modulators in treatment of Endometriosis or prior to Endometrial Ablation, or
Topical Tacrolimus or Pimecrolimus for the treatment of mild, moderate or severe atopic eczema. Effective Shared Care Agreement
 Topical Tacrolimus or Pimecrolimus for the treatment of mild, moderate or severe atopic eczema. Effective Shared Care Agreement A Copy of this page signed by all three parties should be retained in the
Topical Tacrolimus or Pimecrolimus for the treatment of mild, moderate or severe atopic eczema. Effective Shared Care Agreement A Copy of this page signed by all three parties should be retained in the
This regimen has low emetogenic potential refer to local protocol None required routinely. Baseline results valid for 7 days. Results valid for 72 hrs
 Regimen : Ipilimumab for Advanced Melanoma ICD10 code Codes pre-fixed with C43. Indication Regimen detail Ipilimumab is recommended as an option for treating advanced (unresectable or metastatic) melanoma
Regimen : Ipilimumab for Advanced Melanoma ICD10 code Codes pre-fixed with C43. Indication Regimen detail Ipilimumab is recommended as an option for treating advanced (unresectable or metastatic) melanoma
Patient Information Leaflet
 Patient Information Leaflet METHOTREXATE We hope this fact sheet will provide you with some information about Methotrexate and answer some of the questions you may have. Methotrexate is available in tablet
Patient Information Leaflet METHOTREXATE We hope this fact sheet will provide you with some information about Methotrexate and answer some of the questions you may have. Methotrexate is available in tablet
Professor Andrew Wright,
 Professor Andrew Wright, Systemic treatments are drugs taken as tablets or injections that travel through the bloodstream, dampening down the immune system to reach and treat eczema all over the body.
Professor Andrew Wright, Systemic treatments are drugs taken as tablets or injections that travel through the bloodstream, dampening down the immune system to reach and treat eczema all over the body.
2 What you need to know before you have Ampiclox
 Reason for update: GDS 14 & QRD Updates Response to questions for variation update section 4.1 of SPC MHRA Submission Date: 6 November 2014 MHRA Approval Date: Text Date: October 2014 Text Issue and Draft
Reason for update: GDS 14 & QRD Updates Response to questions for variation update section 4.1 of SPC MHRA Submission Date: 6 November 2014 MHRA Approval Date: Text Date: October 2014 Text Issue and Draft
National Chlamydia Screening Programme September 2012 PATIENT GROUP DIRECTION FOR THE ADMINISTRATION OF AZITHROMYCIN FOR CHLAMYDIA TRACHOMATIS
 PATIENT GROUP DIRECTION FOR THE ADMINISTRATION OF AZITHROMYCIN FOR CHLAMYDIA TRACHOMATIS Below is a template that can be used to produce a local patient group direction (PGD) for the administration of
PATIENT GROUP DIRECTION FOR THE ADMINISTRATION OF AZITHROMYCIN FOR CHLAMYDIA TRACHOMATIS Below is a template that can be used to produce a local patient group direction (PGD) for the administration of
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE. Multiple Technology Appraisal
 NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Multiple Technology Appraisal Infliximab, adalimumab and golimumab for treating moderately to severely active ulcerative colitis after the failure of conventional
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Multiple Technology Appraisal Infliximab, adalimumab and golimumab for treating moderately to severely active ulcerative colitis after the failure of conventional
Essential Shared Care Agreement Drugs for Dementia
 Ref No. E040 Essential Shared Care Agreement Drugs for Dementia Please complete the following details: Patient s name, address, date of birth Consultant s contact details (p.3) And send One copy to: 1.
Ref No. E040 Essential Shared Care Agreement Drugs for Dementia Please complete the following details: Patient s name, address, date of birth Consultant s contact details (p.3) And send One copy to: 1.
EMEA PUBLIC STATEMENT ON LEFLUNOMIDE (ARAVA) - SEVERE AND SERIOUS HEPATIC REACTIONS -
 The European Agency for the Evaluation of Medicinal Products Post-authorisation evaluation of medicines for human use London, 12 March 2001 Doc. Ref: EMEA/H/5611/01/en EMEA PUBLIC STATEMENT ON LEFLUNOMIDE
The European Agency for the Evaluation of Medicinal Products Post-authorisation evaluation of medicines for human use London, 12 March 2001 Doc. Ref: EMEA/H/5611/01/en EMEA PUBLIC STATEMENT ON LEFLUNOMIDE
MELATONIN FOR SLEEP DISORDERS IN CHILDREN AND ADOLESCENTS WITH NEURODEVELOPMENTAL DISORDERS SHARED CARE GUIDELINES
 MELATONIN FOR SLEEP DISORDERS IN CHILDREN AND ADOLESCENTS WITH NEURODEVELOPMENTAL DISORDERS SHARED CARE GUIDELINES Version control: Version Date Main changes/comments V1 4 June 2013 First draft circulated
MELATONIN FOR SLEEP DISORDERS IN CHILDREN AND ADOLESCENTS WITH NEURODEVELOPMENTAL DISORDERS SHARED CARE GUIDELINES Version control: Version Date Main changes/comments V1 4 June 2013 First draft circulated
NHS ONEL and NELFT Shared Care Guidelines. Management of medications for Alzheimer s disease. Patient Name : Date of Birth: NHS No:
 NHS ONEL and NELFT Shared Care Guidelines Management of medications for Alzheimer s disease DOCUMENT TO BE SCANNED INTO ELECTRONIC RECORDS AND FILED IN NOTES Patient Name : Date of Birth: NHS No: Name
NHS ONEL and NELFT Shared Care Guidelines Management of medications for Alzheimer s disease DOCUMENT TO BE SCANNED INTO ELECTRONIC RECORDS AND FILED IN NOTES Patient Name : Date of Birth: NHS No: Name
Prescribing Framework for Donepezil in the Treatment and Management of Dementia
 Hull & East Riding Prescribing Committee Prescribing Framework for Donepezil in the Treatment and Management of Dementia Patients Name:.. NHS Number: Patients Address:... (Use addressograph sticker) GP
Hull & East Riding Prescribing Committee Prescribing Framework for Donepezil in the Treatment and Management of Dementia Patients Name:.. NHS Number: Patients Address:... (Use addressograph sticker) GP
Appendix D- Monoclonal Gammopathy of Undetermined Significance (MGUS)
 Local Enhanced Service: Primary care management of stable haematological patients Monoclonal Gammopathy of Undetermined Significance (MGUS) 1. Introduction Monoclonal Gammopathy of Undetermined Significance
Local Enhanced Service: Primary care management of stable haematological patients Monoclonal Gammopathy of Undetermined Significance (MGUS) 1. Introduction Monoclonal Gammopathy of Undetermined Significance
Oxford University Hospitals. NHS Trust. Department of Neurology Natalizumab (Tysabri) for Multiple Sclerosis. Information for patients
 Oxford University Hospitals NHS Trust Department of Neurology Natalizumab (Tysabri) for Multiple Sclerosis Information for patients page 2 What is Natalizumab and what is it used for? Natalizumab is an
Oxford University Hospitals NHS Trust Department of Neurology Natalizumab (Tysabri) for Multiple Sclerosis Information for patients page 2 What is Natalizumab and what is it used for? Natalizumab is an
Understanding Colitis and Crohn s Disease
 Improving life for people affected by Colitis and Crohn s Disease Understanding Colitis and Crohn s Disease 1 Understanding Colitis and Crohn s Disease Understanding Ulcerative Colitis and Crohn s Disease...
Improving life for people affected by Colitis and Crohn s Disease Understanding Colitis and Crohn s Disease 1 Understanding Colitis and Crohn s Disease Understanding Ulcerative Colitis and Crohn s Disease...
Warfarin therapy for stroke patients with atrial fibrillation
 Warfarin therapy for stroke patients with atrial fibrillation Delivering the best in care UHB is a no smoking Trust To see all of our current patient information leaflets please visit www.uhb.nhs.uk/patient-information-leaflets.htm
Warfarin therapy for stroke patients with atrial fibrillation Delivering the best in care UHB is a no smoking Trust To see all of our current patient information leaflets please visit www.uhb.nhs.uk/patient-information-leaflets.htm
Teriflunomide is the active metabolite of Leflunomide, a drug employed since 1994 for the treatment of rheumatoid arthritis (Baselt, 2011).
 Page 1 of 10 ANALYTE NAME AND STRUCTURE TERIFLUNOMIDE Teriflunomide TRADE NAME Aubagio CATEGORY Antimetabolite TEST CODE PURPOSE Therapeutic Drug Monitoring GENERAL RELEVANCY BACKGROUND sclerosis. The
Page 1 of 10 ANALYTE NAME AND STRUCTURE TERIFLUNOMIDE Teriflunomide TRADE NAME Aubagio CATEGORY Antimetabolite TEST CODE PURPOSE Therapeutic Drug Monitoring GENERAL RELEVANCY BACKGROUND sclerosis. The
Vincristine by short infusion Doxorubicin by injection Cyclophosphamide by injection Rituximab by an infusion over between 60 minutes to a few hours
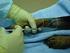 R-CHOP R-CHOP This leaflet is offered as a guide to you and your family. The possible benefits of treatment vary; for some people chemotherapy may reduce the risk of the cancer coming back, for others
R-CHOP R-CHOP This leaflet is offered as a guide to you and your family. The possible benefits of treatment vary; for some people chemotherapy may reduce the risk of the cancer coming back, for others
CLL. Handheld record. Stockport NHS foundation trust
 CLL 2015 Handheld record Contains- Patient information booklet, details of haematology clinic assessment and ongoing clinical assessment at GP surgery Stockport NHS foundation trust You have been diagnosed
CLL 2015 Handheld record Contains- Patient information booklet, details of haematology clinic assessment and ongoing clinical assessment at GP surgery Stockport NHS foundation trust You have been diagnosed
Lung Pathway Group Pemetrexed and Cisplatin in Non-Small Cell Lung Cancer (NSCLC)
 Indication: NICE TA181 First line treatment option in advanced or metastatic non-squamous NSCLC (histology confirmed as adenocarcinoma or large cell carcinoma) Performance status 0-1 Regimen details: Pemetrexed
Indication: NICE TA181 First line treatment option in advanced or metastatic non-squamous NSCLC (histology confirmed as adenocarcinoma or large cell carcinoma) Performance status 0-1 Regimen details: Pemetrexed
Oxford Anticoagulation & Thrombosis Service Deep Vein Thrombosis (DVT) and Pulmonary Embolism (PE)
 Oxford University Hospitals NHS Trust Oxford Anticoagulation & Thrombosis Service Deep Vein Thrombosis (DVT) and Pulmonary Embolism (PE) Information for people with a blood clot (thrombus) What is this
Oxford University Hospitals NHS Trust Oxford Anticoagulation & Thrombosis Service Deep Vein Thrombosis (DVT) and Pulmonary Embolism (PE) Information for people with a blood clot (thrombus) What is this
How long will it take to work? You may begin to feel better within a few days or it may take up to six weeks after your first treatment session.
 Crohn's and Colitis UK Drug Treatment Information Improving life for people affected by inflammatory bowel diseases Infliximab This information leaflet aims to answer common questions you may have if you
Crohn's and Colitis UK Drug Treatment Information Improving life for people affected by inflammatory bowel diseases Infliximab This information leaflet aims to answer common questions you may have if you
A PATIENT S GUIDE TO DEEP VEIN THROMBOSIS TREATMENT
 A PATIENT S GUIDE TO DEEP VEIN THROMBOSIS TREATMENT This medicine is subject to additional monitoring. This will allow quick identification of new safety information. If you get any side effects, talk
A PATIENT S GUIDE TO DEEP VEIN THROMBOSIS TREATMENT This medicine is subject to additional monitoring. This will allow quick identification of new safety information. If you get any side effects, talk
West of Scotland Cancer Network Chemotherapy Protocol. Cisplatin and Pemetrexed for Malignant Mesothelioma (LUWOS 0021)
 West of Scotland Cancer Network Chemotherapy Protocol Cisplatin and Pemetrexed for Malignant Mesothelioma (LUWOS 0021) Indication Palliative chemotherapy for malignant mesothelioma of the pleura Eligibility
West of Scotland Cancer Network Chemotherapy Protocol Cisplatin and Pemetrexed for Malignant Mesothelioma (LUWOS 0021) Indication Palliative chemotherapy for malignant mesothelioma of the pleura Eligibility
MS Treatments Gilenya
 1 MSology Essentials Series Gilenya (fingolimod) Developed by MSology with the invaluable assistance of multiple sclerosis nurse advisors: Trudy Campbell Dalhousie MS Research Unit, Capital Health, Halifax,
1 MSology Essentials Series Gilenya (fingolimod) Developed by MSology with the invaluable assistance of multiple sclerosis nurse advisors: Trudy Campbell Dalhousie MS Research Unit, Capital Health, Halifax,
Risk Management Plan
 Risk Management Plan Active substance: Drospirenone/ethinylestradiol Version number: 4.0 VI.2 Elements for a Public Summary VI.2.1 Overview of disease epidemiology Oral contraception Effective control
Risk Management Plan Active substance: Drospirenone/ethinylestradiol Version number: 4.0 VI.2 Elements for a Public Summary VI.2.1 Overview of disease epidemiology Oral contraception Effective control
Rheumatoid Arthritis. GP workshop 15 January 2011
 Rheumatoid Arthritis GP workshop 15 January 2011 Case 1 A 72 year old Malay woman with RA comes for routine follow up. She feels generally unwell in the last 5 days. Appetite is fair. Her joints are fine.
Rheumatoid Arthritis GP workshop 15 January 2011 Case 1 A 72 year old Malay woman with RA comes for routine follow up. She feels generally unwell in the last 5 days. Appetite is fair. Her joints are fine.
abc Trust Guideline for Use of Oral Methotrexate, Shared Care Guideline for Adults
 Trust Guideline for abc Use of Oral Methotrexate, Shared Care Guideline for Adults A guideline recommended for use In: East and North Herts NHS Trust East and North Herts PCT By: Consultants in East and
Trust Guideline for abc Use of Oral Methotrexate, Shared Care Guideline for Adults A guideline recommended for use In: East and North Herts NHS Trust East and North Herts PCT By: Consultants in East and
Salazopyrin Tablets. 500 mg. Sulfasalazine POM. 112 Tablets. Each tablet contains sulfasalazine 500 mg. Use as directed by a doctor. Oral use.
 Salazopyrin Tablets 500 mg Sulfasalazine POM 112 Tablets Each tablet contains sulfasalazine 500 mg. Use as directed by a doctor. Oral use. Store below 25 C. Read the leaflet before use. Keep out of the
Salazopyrin Tablets 500 mg Sulfasalazine POM 112 Tablets Each tablet contains sulfasalazine 500 mg. Use as directed by a doctor. Oral use. Store below 25 C. Read the leaflet before use. Keep out of the
Initiate Atorvastatin 20mg daily
 Type 2 Diabetes Patient Objectives Stopping Smoking BMI > 25 kg m² Control BP to
Type 2 Diabetes Patient Objectives Stopping Smoking BMI > 25 kg m² Control BP to
Novartis Gilenya FDO Program Clinical Protocol and Highlights from Prescribing Information (PI)
 Novartis Gilenya FDO Program Clinical Protocol and Highlights from Prescribing Information (PI) Highlights from Prescribing Information - the link to the full text PI is as follows: http://www.pharma.us.novartis.com/product/pi/pdf/gilenya.pdf
Novartis Gilenya FDO Program Clinical Protocol and Highlights from Prescribing Information (PI) Highlights from Prescribing Information - the link to the full text PI is as follows: http://www.pharma.us.novartis.com/product/pi/pdf/gilenya.pdf
Summary of the risk management plan (RMP) for Accofil (filgrastim)
 EMA/475472/2014 Summary of the risk management plan (RMP) for Accofil (filgrastim) This is a summary of the risk management plan (RMP) for Accofil, which details the measures to be taken in order to ensure
EMA/475472/2014 Summary of the risk management plan (RMP) for Accofil (filgrastim) This is a summary of the risk management plan (RMP) for Accofil, which details the measures to be taken in order to ensure
Summary of the risk management plan (RMP) for Cerdelga (eliglustat)
 EMA/743948/2014 Summary of the risk management plan (RMP) for Cerdelga (eliglustat) This is a summary of the risk management plan (RMP) for Cerdelga, which details the measures to be taken in order to
EMA/743948/2014 Summary of the risk management plan (RMP) for Cerdelga (eliglustat) This is a summary of the risk management plan (RMP) for Cerdelga, which details the measures to be taken in order to
MANAGEMENT OF COMMON SIDE EFFECTS of INH (Isoniazid), RIF (Rifampin), PZA (Pyrazinamide), and EMB (Ethambutol)
 MANAGEMENT OF COMMON SIDE EFFECTS of INH (Isoniazid), RIF (Rifampin), PZA (Pyrazinamide), and EMB (Ethambutol) 1. Hepatotoxicity: In Active TB Disease a. Background: 1. Among the 4 standard anti-tb drugs,
MANAGEMENT OF COMMON SIDE EFFECTS of INH (Isoniazid), RIF (Rifampin), PZA (Pyrazinamide), and EMB (Ethambutol) 1. Hepatotoxicity: In Active TB Disease a. Background: 1. Among the 4 standard anti-tb drugs,
Package leaflet: Information for the patient. Laxido Orange, powder for oral solution
 Package leaflet: Information for the patient Laxido Orange, powder for oral solution Read all of this leaflet carefully before you start taking this medicine because it contains important information for
Package leaflet: Information for the patient Laxido Orange, powder for oral solution Read all of this leaflet carefully before you start taking this medicine because it contains important information for
Leukapheresis for inflammatory bowel disease
 Issue date: June 2005 Leukapheresis for inflammatory bowel disease Understanding NICE guidance information for people considering the procedure, and for the public Information about NICE Interventional
Issue date: June 2005 Leukapheresis for inflammatory bowel disease Understanding NICE guidance information for people considering the procedure, and for the public Information about NICE Interventional
PACKAGE LEAFLET: INFORMATION FOR THE USER Paracetamol 500 mg Effervescent Tablets Paracetamol
 PACKAGE LEAFLET: INFORMATION FOR THE USER Paracetamol 500 mg Effervescent Tablets Paracetamol Read all of this leaflet carefully because it contains important information for you. This medicine is available
PACKAGE LEAFLET: INFORMATION FOR THE USER Paracetamol 500 mg Effervescent Tablets Paracetamol Read all of this leaflet carefully because it contains important information for you. This medicine is available
Drug Treatment Information for patients with Inflammatory Bowel Disease. Infliximab
 Drug Treatment Information for patients with Inflammatory Bowel Disease Infliximab Improving life for people affected by Colitis and Crohn s Disease National Association For Colitis and Crohn s Disease
Drug Treatment Information for patients with Inflammatory Bowel Disease Infliximab Improving life for people affected by Colitis and Crohn s Disease National Association For Colitis and Crohn s Disease
Docetaxel + Carboplatin + Trastuzumab (TCH) Adjuvant Breast Cancer
 Docetaxel + Carboplatin + Trastuzumab (TCH) Adjuvant Breast Cancer Background: A non-anthracycline based regimen for high-risk, HER 2 positive breast cancer in the adjuvant setting (BCIRG 006). Patient
Docetaxel + Carboplatin + Trastuzumab (TCH) Adjuvant Breast Cancer Background: A non-anthracycline based regimen for high-risk, HER 2 positive breast cancer in the adjuvant setting (BCIRG 006). Patient
MEDICATION GUIDE mitoxantrone (mito-xan-trone) for injection concentrate
 MEDICATION GUIDE mitoxantrone (mito-xan-trone) for injection concentrate Read this Medication Guide before you start receiving mitoxantrone and each time you receive mitoxantrone. There may be new information.
MEDICATION GUIDE mitoxantrone (mito-xan-trone) for injection concentrate Read this Medication Guide before you start receiving mitoxantrone and each time you receive mitoxantrone. There may be new information.
CICLOSPORIN. What are the aims of this leaflet?
 CICLOSPORIN What are the aims of this leaflet? This leaflet has been written to help you understand more about ciclosporin. It tells you what it is, how it works, how it is used to treat skin conditions,
CICLOSPORIN What are the aims of this leaflet? This leaflet has been written to help you understand more about ciclosporin. It tells you what it is, how it works, how it is used to treat skin conditions,
Drugs for Alcohol Dependence: Clinical Guidance and Three Way Agreement
 Drugs for Alcohol Dependence: Clinical Guidance and Three Way Agreement for County Durham In partnership with the GP, the client, and the County Durham Drug and Alcohol Service December 2015 Version 1.0
Drugs for Alcohol Dependence: Clinical Guidance and Three Way Agreement for County Durham In partnership with the GP, the client, and the County Durham Drug and Alcohol Service December 2015 Version 1.0
Breast Pathway Group AC (Doxorubicin / Cyclophosphamide) in Early Breast Cancer
 Breast Pathway Group AC (Doxorubicin / Cyclophosphamide) in Early Breast Cancer Indication: Neoadjuvant or adjuvant treatment of breast cancer Regimen details: Doxorubicin 60mg/m 2 IV Day 1 Cyclophosphamide
Breast Pathway Group AC (Doxorubicin / Cyclophosphamide) in Early Breast Cancer Indication: Neoadjuvant or adjuvant treatment of breast cancer Regimen details: Doxorubicin 60mg/m 2 IV Day 1 Cyclophosphamide
Biologic Treatments for Rheumatoid Arthritis
 Biologic Treatments Rheumatoid Arthritis (also known as cytokine inhibitors, TNF inhibitors, IL 1 inhibitor, or Biologic Response Modifiers) Description Biologics are new class of drugs that have been
Biologic Treatments Rheumatoid Arthritis (also known as cytokine inhibitors, TNF inhibitors, IL 1 inhibitor, or Biologic Response Modifiers) Description Biologics are new class of drugs that have been
Rheumatoid Arthritis monitoring of DMARDs
 www.bpac.org.nz keyword: DMARDS Rheumatoid Arthritis monitoring of DMARDs Key reviewers: Professor John Highton, Head of Section, Department of Medical and Surgical Sciences, Dunedin School of Medicine,
www.bpac.org.nz keyword: DMARDS Rheumatoid Arthritis monitoring of DMARDs Key reviewers: Professor John Highton, Head of Section, Department of Medical and Surgical Sciences, Dunedin School of Medicine,
Safety Information Card for Xarelto Patients
 Safety Information Card for Xarelto Patients 15mg Simply Protecting More Patients 20mg Simply Protecting More Patients Keep this card with you at all times Present this card to every physician or dentist
Safety Information Card for Xarelto Patients 15mg Simply Protecting More Patients 20mg Simply Protecting More Patients Keep this card with you at all times Present this card to every physician or dentist
Summary of the risk management plan (RMP) for Xultophy (insulin degludec / liraglutide)
 EMA/467911/2014 Summary of the risk management plan (RMP) for Xultophy (insulin degludec / liraglutide) This is a summary of the risk management plan (RMP) for Xultophy, which details the measures to be
EMA/467911/2014 Summary of the risk management plan (RMP) for Xultophy (insulin degludec / liraglutide) This is a summary of the risk management plan (RMP) for Xultophy, which details the measures to be
Dalacin C 150 mg Hard Capsules Clindamycin (as clindamycin hydrochloride)
 Package leaflet: Information for the patient Dalacin C 150 mg Hard Capsules Clindamycin (as clindamycin hydrochloride) Read all of this leaflet carefully before you start taking this medicine because it
Package leaflet: Information for the patient Dalacin C 150 mg Hard Capsules Clindamycin (as clindamycin hydrochloride) Read all of this leaflet carefully before you start taking this medicine because it
Humulin R (U500) insulin: Prescribing Guidance
 Leeds Humulin R (U500) insulin: Prescribing Guidance Amber Drug Level 2 We have started your patient on Humulin R (U500) insulin for the treatment of diabetic patients with marked insulin resistance requiring
Leeds Humulin R (U500) insulin: Prescribing Guidance Amber Drug Level 2 We have started your patient on Humulin R (U500) insulin for the treatment of diabetic patients with marked insulin resistance requiring
Always take this medicine exactly as described in this leaflet or as your doctor, pharmacist or nurse have told you.
 leaflet: Information for the user Macrogol 4000 10 g powder for oral solution in sachet Macrogol 4000
leaflet: Information for the user Macrogol 4000 10 g powder for oral solution in sachet Macrogol 4000
Eastern Health MS Service. Tysabri Therapy. Information for People with MS and their Families
 Eastern Health MS Service Tysabri Therapy Information for People with MS and their Families The Eastern Health MS Service has developed this information for you as a guide through what will happen to you
Eastern Health MS Service Tysabri Therapy Information for People with MS and their Families The Eastern Health MS Service has developed this information for you as a guide through what will happen to you
Mycophenolate mofetil (CellCept ): risks of miscarriage and birth defects. Patient guide. Key points to remember
 Mycophenolate (CellCept ): risks of miscarriage and birth defects Patient guide Key points to remember Mycophenolate (CellCept ) causes birth defects and miscarriages Follow the contraceptive advice given
Mycophenolate (CellCept ): risks of miscarriage and birth defects Patient guide Key points to remember Mycophenolate (CellCept ) causes birth defects and miscarriages Follow the contraceptive advice given
Multiple sclerosis disease-modifying drugs second line treatments
 Great Ormond Street Hospital for Children NHS Foundation Trust: Information for Families Multiple sclerosis disease-modifying drugs second line treatments The following information should be read in conjunction
Great Ormond Street Hospital for Children NHS Foundation Trust: Information for Families Multiple sclerosis disease-modifying drugs second line treatments The following information should be read in conjunction
ORAL ANTICOAGULANTS RIVAROXABAN (XARELTO) FOR PULMONARY EMBOLISM (PE)
 ORAL ANTICOAGULANTS RIVAROXABAN (XARELTO) FOR PULMONARY EMBOLISM (PE) Information Leaflet Your Health. Our Priority. Page 2 of 6 What Are Anticoagulants And What Do They Do? This information leaflet has
ORAL ANTICOAGULANTS RIVAROXABAN (XARELTO) FOR PULMONARY EMBOLISM (PE) Information Leaflet Your Health. Our Priority. Page 2 of 6 What Are Anticoagulants And What Do They Do? This information leaflet has
PROTOCOL TITLE: Ambulatory Initiation and Management of Warfarin for Adults
 PROTOCOL NUMBER: 7 PROTOCOL TITLE: Ambulatory Initiation and Management of Warfarin for Adults THIS PROTOCOL APPLIES TO: UW Health Clinics: all adult outpatients with an active order for warfarin TARGET
PROTOCOL NUMBER: 7 PROTOCOL TITLE: Ambulatory Initiation and Management of Warfarin for Adults THIS PROTOCOL APPLIES TO: UW Health Clinics: all adult outpatients with an active order for warfarin TARGET
Gilenya. Exceptional healthcare, personally delivered
 Gilenya Exceptional healthcare, personally delivered Your Consultant Neurologist has suggested that you may benefit from treatment with Gilenya. The decision to start this form of treatment can be difficult.
Gilenya Exceptional healthcare, personally delivered Your Consultant Neurologist has suggested that you may benefit from treatment with Gilenya. The decision to start this form of treatment can be difficult.
In Tanzania, ARVs were introduced free-of-charge by the government in 2004 and, by July 2008, almost 170,000 people were receiving the drugs.
 ANTIRETROVIRAL TREATMENT What is ART and ARV? ART is a short form for Antiretroviral Therapy (or Treatment). Antiretroviral therapy is a treatment consisting of a combination of drugs which work against
ANTIRETROVIRAL TREATMENT What is ART and ARV? ART is a short form for Antiretroviral Therapy (or Treatment). Antiretroviral therapy is a treatment consisting of a combination of drugs which work against
